
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||

| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |||||||
| Title of Each Class | Trading Symbol(s) | Name of Each Exchange Where Registered | |||||||||||||||
| The | |||||||||||||||||
| x | Accelerated filer | ☐ | ||||||||||||
| Non-accelerated filer | ☐ | Smaller reporting company | ||||||||||||
| Emerging growth company | ||||||||||||||
| Page | ||||||||
F- 1 | ||||||||
| Product | Indication | Collaborator | Major Markets | |||||||||||||||||
 | RMS in the U.S. RRMS in the E.U. | None | U.S. France Germany Italy Japan Spain U.K. | |||||||||||||||||
 | RMS in the U.S. | Alkermes Pharma Ireland Limited, a subsidiary of Alkermes plc (Alkermes) | U.S. | |||||||||||||||||
 | RMS | None | U.S. France Germany Italy Japan Spain | |||||||||||||||||
 | RMS in the U.S. RRMS in the E.U. | None | U.S. France Germany Italy Spain U.K. | |||||||||||||||||
 | RMS RRMS in the E.U. Crohn's disease in the U.S. | None | U.S. France Germany Italy Spain U.K. | |||||||||||||||||
 | Walking ability for patients with MS | Acorda Therapeutics, Inc. (Acorda) | France Germany | |||||||||||||||||
| Product | Indication | Collaborator | Major Markets | |||||||||||||||||
 | SMA | Ionis Pharmaceuticals Inc. (Ionis) | U.S. Brazil Canada France Germany Italy Japan Spain Turkey | |||||||||||||||||
| Product | Indication | Major Markets | ||||||||||||
 | Rheumatoid arthritis Juvenile idiopathic arthritis Psoriatic arthritis Axial spondyloarthritis Plaque psoriasis Paediatric plaque psoriasis | France Germany Italy Spain U.K. | ||||||||||||
 | Rheumatoid arthritis Juvenile idiopathic arthritis Axial spondyloarthritis Psoriatic arthritis Psoriasis Paediatric plaque psoriasis Hidradenitis suppurativa Adolescent hidradenitis suppurativa Crohn’s disease Paediatric Crohn's disease Ulcerative colitis Uveitis Paediatric Uveitis | France Germany U.K. | ||||||||||||
 | Rheumatoid arthritis Crohn’s disease Paediatric Crohn’s disease Ulcerative colitis Paediatric ulcerative colitis Ankylosing spondylitis Psoriatic arthritis Psoriasis | France Germany Italy | ||||||||||||
| Product | Indication | Major Markets | ||||||||||||
 | Non-Hodgkin's lymphoma CLL Rheumatoid arthritis Two forms of ANCA-associated vasculitis Pemphigus vulgaris | U.S. Canada | ||||||||||||
 | Non-Hodgkin's lymphoma CLL | U.S. | ||||||||||||
 | In combination with chlorambucil for previously untreated CLL Follicular lymphoma In combination with chemotherapy followed by GAZYVA alone for previously untreated follicular lymphoma | U.S. | ||||||||||||
 | RMS PPMS | U.S. Australia Germany Switzerland | ||||||||||||
| Product | Indication | Collaborator | Major Markets | |||||||||||||||||
 | Moderate to severe plaque psoriasis | None | Germany | |||||||||||||||||
| Product | Territory | Patent No. | General Subject Matter | Patent Expiration(1) | ||||||||||||||||||||||
| TECFIDERA | U.S. | 8,399,514 | Methods of treatment | 2028(2) | ||||||||||||||||||||||
| Europe | 1131065 | Formulations of dialkyl fumarates and their use for treating autoimmune diseases | 2024(3) | |||||||||||||||||||||||
| Europe | 2137537 | Methods of use | 2028(4) | |||||||||||||||||||||||
| PLEGRIDY | U.S. | 7,446,173 | Polymer conjugates of interferon beta-1a | 2022 | ||||||||||||||||||||||
| U.S. | 8,524,660 | Methods of treatment | 2023 | |||||||||||||||||||||||
| U.S. | 8,017,733 | Polymer conjugates of interferon beta-1a | 2027 | |||||||||||||||||||||||
| Europe | 1656952 | Polymer conjugates of interferon-beta-1a and uses thereof | 2024(5) | |||||||||||||||||||||||
| Europe | 1476181 | Polymer conjugates of interferon-beta-1a and uses thereof | 2023(6) | |||||||||||||||||||||||
| TYSABRI | U.S. | 7,807,167 | Methods of treatment | 2023 | ||||||||||||||||||||||
| U.S. | 9,493,567 | Methods of treatment | 2027 | |||||||||||||||||||||||
| Europe | 1485127 | Methods of use | 2023(2) | |||||||||||||||||||||||
| Europe | 2676967 | Methods of use | 2027 | |||||||||||||||||||||||
| FAMPYRA | Europe | 1732548 | Sustained-release aminopyridine compositions for increasing walking speed in patients with MS | 2025(7) | ||||||||||||||||||||||
| Europe | 2377536 | Sustained-release aminopyridine compositions for treating MS | 2025(8) | |||||||||||||||||||||||
| VUMERITY | U.S. | 8,669,281 | Compounds and pharmaceutical compositions | 2033 | ||||||||||||||||||||||
| U.S. | 9,090,558 | Methods of treatment | 2033 | |||||||||||||||||||||||
| U.S. | 10,080,733 | Crystalline forms, pharmaceutical compositions and methods of treatment | 2033 | |||||||||||||||||||||||
| SPINRAZA | U.S. | 7,101,993 | Oligonucleotides containing 2’-O-modified purines | 2023 | ||||||||||||||||||||||
| U.S. | 7,838,657 | SMA treatment via targeting of SMN2 splice site inhibitory sequences | 2027 | |||||||||||||||||||||||
| U.S. | 8,110,560 | SMA treatment via targeting of SMN2 splice site inhibitory sequences | 2025 | |||||||||||||||||||||||
| U.S. | 8,361,977 | Compositions and methods for modulation of SMN2 splicing | 2030 | |||||||||||||||||||||||
| U.S. | 8,980,853 | Compositions and methods for modulation of SMN2 splicing | 2030 | |||||||||||||||||||||||
| U.S. | 9,717,750 | Compositions and methods for modulation of SMN2 splicing | 2030 | |||||||||||||||||||||||
| U.S. | 9,926,559 | Compositions and methods for modulation of SMN2 splicing | 2034 | |||||||||||||||||||||||
| U.S. | 10,266,822 | SMA treatment via targeting of SMN2 splice site inhibitory sequences | 2025 | |||||||||||||||||||||||
| U.S. | 10,436,802 | Methods for Treating Spinal Muscular Atrophy | 2035 | |||||||||||||||||||||||
| Europe | 1910395 | Compositions and methods for modulation of SMN2 splicing | 2026(9) | |||||||||||||||||||||||
| Europe | 2548560 | Compositions and methods for modulation of SMN2 splicing | 2026(10) | |||||||||||||||||||||||
| Europe | 3305302 | Compositions and methods for modulation of SMN2 splicing | 2030 | |||||||||||||||||||||||
| Europe | 3308788 | Compositions and methods for modulation of SMN2 splicing | 2026 | |||||||||||||||||||||||
| Europe | 3449926 | Compositions and methods for modulation of SMN2 splicing | 2030 | |||||||||||||||||||||||
| Product | Territory | Expected Expiration | ||||||||||||
| TECFIDERA | E.U. | 2024 | ||||||||||||
| PLEGRIDY | U.S. | 2026 | ||||||||||||
| E.U. | 2024 | |||||||||||||
| FAMPYRA | E.U. | 2021 | ||||||||||||
| SPINRAZA | U.S. | 2023 | ||||||||||||
| E.U. | 2029 | |||||||||||||
| Competing Product | Competitor | |||||||
| AUBAGIO (teriflunomide) | Sanofi Genzyme | |||||||
| BETASERON/BETAFERON (interferon-beta-1b) | Bayer Group | |||||||
| COPAXONE (glatiramer acetate) | Teva Pharmaceuticals Industries Ltd. | |||||||
| EXTAVIA (interferon-beta-1b) | Novartis AG | |||||||
| GILENYA (fingolimod) | Novartis AG | |||||||
| GLATOPA (glatiramer acetate) | Sandoz, a division of Novartis AG | |||||||
| LEMTRADA (alemtuzumab) | Sanofi Genzyme | |||||||
| MAVENCLAD (cladribine) | EMD Serono | |||||||
| MAYZENT (siponimod) | Novartis AG | |||||||
| OCREVUS (ocrelizumab) | Genentech | |||||||
| REBIF (interferon-beta-1) | EMD Serono | |||||||
| ZEPOSIA (ozanimod) | BMS | |||||||
| BAFIERTAM (monomethyl fumarate) | Banner Life Sciences | |||||||
| KESIMPTA (ofatumumab) | Novartis AG | |||||||
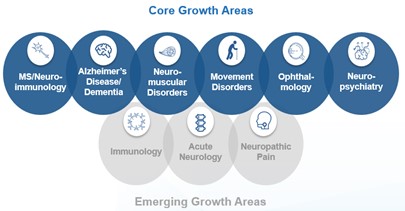
| Core Growth Areas | MS and Neuroimmunology | BIIB061 (oral remyelination) - MS | Phase 1 | |||||||||||||||||||||||
| BIIB091 (BTK inhibitor) - MS | Phase 1 | |||||||||||||||||||||||||
| BIIB107 (anti-VLA4) - MS | Phase 1 | |||||||||||||||||||||||||
| Alzheimer's Disease and Dementia | Aducanumab (Aβ mAb)* - Alzheimer's | Filed in U.S., E.U. and Japan | ||||||||||||||||||||||||
BAN2401 (lecanemab)* - Alzheimer's | Phase 3 | |||||||||||||||||||||||||
| BIIB092 (gosuranemab) - Alzheimer's | Phase 2 | |||||||||||||||||||||||||
| BIIB076 (anti-tau mAb) - Alzheimer's | Phase 1 | |||||||||||||||||||||||||
| BIIB080 (tau ASO) - Alzheimer's | Phase 1 | |||||||||||||||||||||||||
| Neuromuscular Disorders, including SMA and ALS | BIIB067 (tofersen) - ALS | Phase 3 | ||||||||||||||||||||||||
BIIB078 (IONIS-C9Rx)# - ALS | Phase 1 | |||||||||||||||||||||||||
BIIB105 (ataxin-2 ASO)# - ALS | Phase 1 | |||||||||||||||||||||||||
| BIIB100 (XP01 inhibitor) - ALS | Phase 1 | |||||||||||||||||||||||||
| BIIB110 (ActRIIA/B ligand trap) - SMA | Phase 1 | |||||||||||||||||||||||||
| Parkinson's Disease and Movement Disorders | BIIB124 (SAGE-324)* - Essential Tremor | Phase 2 | ||||||||||||||||||||||||
BIIB094 (ION859)# - Parkinson's | Phase 1 | |||||||||||||||||||||||||
| BIIB118 (CK1 inhibitor) - ISWRD in Parkinson's | Phase 1 | |||||||||||||||||||||||||
BIIB101 (ION464)# - Multiple System Atrophy | Phase 1 | |||||||||||||||||||||||||
BIIB122 (DNL151)* - Parkinson's | Phase 1 | |||||||||||||||||||||||||
| Ophthalmology | BIIB111 (timrepigene emparvovec) - CHM | Phase 3 | ||||||||||||||||||||||||
| BIIB112 (RPGR gene therapy) - XLRP | Phase 2/3 | |||||||||||||||||||||||||
| Neuropsychiatry | BIIB125 (zuranolone)* - PPD | Phase 3 | ||||||||||||||||||||||||
| BIIB125 (zuranolone)* - MDD | Phase 3 | |||||||||||||||||||||||||
| BIIB104 (AMPA PAM) - CIAS | Phase 2 | |||||||||||||||||||||||||
| Emerging Growth Areas | Immunology | Dapirolizumab pegol (anti-CD40L)* - SLE | Phase 3 | |||||||||||||||||||||||
| BIIB059 (anti-BDCA2) - CLE/SLE | Phase 2 | |||||||||||||||||||||||||
| Acute Neurology | BIIB093 (glibenclamide IV) - LHI^ Stroke | Phase 3 | ||||||||||||||||||||||||
TMS-007# - Acute Ischemic Stroke | Phase 2 | |||||||||||||||||||||||||
| BIIB093 (glibenclamide IV) - Brain Contusion | Phase 2 | |||||||||||||||||||||||||
| Neuropathic Pain | BIIB074 (vixotrigine) - Trigeminal Neuralgia | Phase 2 | ||||||||||||||||||||||||
| BIIB074 (vixotrigine) - Small Fiber Neuropathy | Phase 2 | |||||||||||||||||||||||||
| BIIB095 (Nav 1.7) - Neuropathic Pain | Phase 1 | |||||||||||||||||||||||||
| Biosimilars | SB11 (referencing LUCENTIS)* | Filed in U.S. and E.U. | ||||||||||||||||||||||||
SB15 (referencing EYLEA)* | Phase 3 | |||||||||||||||||||||||||
| Facility | Drug Substance Manufactured | |||||||
| RTP, NC | AVONEX PLEGRIDY TYSABRI Other* | |||||||
| Officer | Current Position | Age | Year Joined Biogen | |||||||||||||||||
| Michel Vounatsos | Chief Executive Officer | 59 | 2016 | |||||||||||||||||
| Susan H. Alexander | Executive Vice President, Chief Legal Officer and Secretary | 64 | 2006 | |||||||||||||||||
| Michael R. McDonnell | Executive Vice President and Chief Financial Officer | 57 | 2020 | |||||||||||||||||
| Alphonse Galdes, Ph.D. | Executive Vice President, Pharmaceutical Operations and Technology | 68 | 1995 | |||||||||||||||||
| Ginger Gregory, Ph.D. | Executive Vice President and Chief Human Resources Officer | 53 | 2017 | |||||||||||||||||
| Chirfi Guindo | Executive Vice President, Global Product Strategy and Commercialization | 55 | 2017 | |||||||||||||||||
| Robin C. Kramer | Senior Vice President, Chief Accounting Officer | 55 | 2018 | |||||||||||||||||
| Alfred W. Sandrock, Jr., M.D., Ph.D. | Executive Vice President, Research and Development | 63 | 1998 | |||||||||||||||||
| Michel Vounatsos | |||||
| Experience | |||||
| Mr. Vounatsos has served as our Chief Executive Officer and as a member of our Board of Directors since January 2017. Prior to that, from April 2016 to December 2016, Mr. Vounatsos served as our Executive Vice President, Chief Commercial Officer. Prior to joining Biogen, Mr. Vounatsos spent 20 years at Merck & Co., Inc. (Merck), a pharmaceutical company, where he most recently served as President, Primary Care, Customer Business Line and Merck Customer Centricity. In this role, he led Merck’s global primary care business unit, a role which encompassed Merck’s cardiology-metabolic, general medicine, women’s health and biosimilars groups and developed and instituted a strategic framework for enhancing the company’s relationships with key constituents, including the most significant providers, payors and retailers and the world’s largest governments. Mr. Vounatsos previously held leadership positions across Europe and in China for Merck. Prior to that, Mr. Vounatsos held management positions at Ciba-Geigy, a pharmaceutical company. Mr. Vounatsos currently serves on the advisory board of Tsinghua University School of Pharmaceutical Sciences, on the Supervisory Board of Liryc, the Electrophysiology and Heart Modeling Institute at the University of Bordeaux, on the board of directors of N-Lorem Foundation and as a member of the MIT Presidential CEO Advisory Board. | |||||
| Public Company Boards | |||||
| l | PerkinElmer, Inc., a global scientific technology and life science research company | ||||
| Education | |||||
| l | Universite Victor Segalen, Bordeaux II, France, C.S.C.T. Certificate in Medicine | ||||
| l | HEC School of Management - Paris, M.B.A. | ||||
| Susan H. Alexander | |||||
| Experience | |||||
| Ms. Alexander has served as our Executive Vice President, Chief Legal Officer and Secretary since April 2018. Prior to that, Ms. Alexander served as our Executive Vice President, Chief Legal, Corporate Services and Secretary from March 2017 to March 2018, as our Executive Vice President, Chief Legal Officer and Secretary from December 2011 to March 2017 and as our Executive Vice President, General Counsel and Corporate Secretary from 2006 to December 2011. Prior to joining Biogen, Ms. Alexander served as the Senior Vice President, General Counsel and Corporate Secretary of PAREXEL International Corporation, a biopharmaceutical services company, from 2003 to January 2006. From 2001 to 2003 Ms. Alexander served as General Counsel of IONA Technologies, a software company. From 1995 to 2001 Ms. Alexander served as Counsel at Cabot Corporation, a specialty chemicals and performance materials company. Prior to that, Ms. Alexander was a partner at the law firms of Hinckley, Allen & Snyder and Fine & Ambrogne. | |||||
| Public Company Boards | |||||
| l | Invacare Corporation, a medical and healthcare product company | ||||
| Education | |||||
| l | Wellesley College, B.A. | ||||
| l | Boston University School of Law, J.D. | ||||
| Michael R. McDonnell | |||||
| Experience | |||||
Mr. McDonnell has served as our Executive Vice President and Chief Financial Officer since August 2020. Prior to joining Biogen, Mr. McDonnell served as Executive Vice President and Chief Financial Officer of IQVIA Holdings Inc., a leading global provider of advanced analytics, technology solutions and contract research services to the life sciences industry, from December 2015 until July 2020. Prior to that, Mr. McDonnell served as the Executive Vice President and Chief Financial Officer of Intelsat, a leading global provider of satellite services, from November 2008 to December 2015, as Executive Vice President and Chief Financial Officer of MCG Capital Corporation, a publicly-held commercial finance company, from September 2004 until October 2008 and as MCG Capital Corporation’s Chief Operating Officer from August 2006 until October 2008. Before joining MCG Capital Corporation, Mr. McDonnell served as Executive Vice President and Chief Financial Officer for EchoStar Communications Corporation (f/k/a DISH Network Corporation), a direct-to-home satellite television operator, from July 2004 until August 2004 and as its Senior Vice President and Chief Financial Officer from August 2000 to July 2004. Mr. McDonnell spent 14 years at PricewaterhouseCoopers LLP, including 4 years as a partner. Mr. McDonnell has a Bachelor of Science degree in accounting from Georgetown University and is a certified public accountant. | |||||
| Education | |||||
| l | Georgetown University, B.S. Accounting | ||||
| Alphonse Galdes, Ph.D. | |||||
| Experience | |||||
| Dr. Galdes has served as our Executive Vice President, Pharmaceutical Operations and Technology since September 2019. Since joining Biogen in 1995, Dr. Galdes has held several senior executive positions, including most recently as Senior Vice President, Asset Development and Portfolio Management from November 2015 to September 2019 and Senior Vice President, Technical Development from October 2010 to November 2015. Dr. Galdes was a Rhodes Scholar at Oxford University and performed post-doctoral research work at the Department of Biological Chemistry at Harvard Medical School. | |||||
| Education | |||||
| l | University of Malta, B.Sc. Chemistry and Biology | ||||
| l | University of Malta, M.Sc. Biochemistry | ||||
| l | Oxford University, Ph.D. Biochemistry | ||||
| Ginger Gregory, Ph.D. | |||||
| Experience | |||||
| Dr. Gregory has served as our Executive Vice President and Chief Human Resources Officer since July 2017. Prior to joining Biogen, Dr. Gregory served as Executive Vice President and Chief Human Resources Officer at Shire PLC, a global specialty biopharmaceutical company, from February 2014 to April 2017. Prior to that, Dr. Gregory held executive-level human resources positions for several multinational companies across a variety of industries, including Dunkin’ Brands Group Inc., a restaurant holding company, where she served as Chief Human Resource Officer, Novartis AG, a pharmaceutical company, where she was the division head of Human Resources for Novartis Vaccines and Diagnostics, Novartis Consumer Health and Novartis Institutes of BioMedical Research and Novo Nordisk A/S, a pharmaceutical company, where she served as Senior Vice President, Corporate People & Organization at the company’s headquarters in Copenhagen, Denmark. Earlier in her career, Dr. Gregory held a variety of human resources generalist and specialist positions at BMS, a pharmaceutical company, and served as a consultant with Booz Allen & Hamilton, an information technology consulting company, in the area of organization change and effectiveness. | |||||
| Education | |||||
| l | University of Massachusetts, B.A. Psychology | ||||
| l | The George Washington University, Ph.D. Psychology | ||||
| Chirfi Guindo | |||||
| Experience | |||||
| Mr. Guindo has served as our Executive Vice President, Global Product Strategy and Commercialization since February 2019. Prior to that, Mr. Guindo served as our Executive Vice President and Head of Global Marketing, Market Access and Customer Innovation from November 2017 to February 2019. Prior to joining Biogen, Mr. Guindo spent 27 years in the global pharmaceutical industry and held several leadership positions at Merck, a pharmaceutical company, in Canada, the U.S., France, Africa and the Netherlands. He worked in several disciplines including Finance, Sales & Marketing, General Management and Global Strategy/Product Development in specialty, acute and hospital care. Most recently Mr. Guindo was Vice President and Managing Director and President and Managing Director of Merck Canada from October 2014 to November 2017. From January 2011 to October 2014 he was Vice President and General Manager, Global HIV Franchise at Merck. | |||||
| Education | |||||
| l | Ecole Central de Paris (France), Engineering | ||||
| l | Stern School of Business, New York University, M.B.A. Finance/Economics | ||||
| Robin C. Kramer | |||||
| Experience | |||||
| Ms. Kramer has served as our Senior Vice President, Chief Accounting Officer since December 2020. Prior to that, Ms. Kramer served as our Vice President, Chief Accounting Officer from November 2018 to December 2020. Prior to joining Biogen, Ms. Kramer served as the Senior Vice President and Chief Accounting Officer of Hertz Global Holdings, Inc., a car rental company, from May 2014 to November 2018. Prior to that, Ms. Kramer was an audit partner at Deloitte & Touche LLP (Deloitte), a professional services firm, from 2007 to 2014, including serving in Deloitte's National Office Accounting Standards and Communications Group from 2007 to 2010. From 2005 to 2007 Ms. Kramer served as Chief Accounting Officer of Fisher Scientific International, Inc., a laboratory supply and biotechnology company, and from 2004 to 2005 Ms. Kramer served as Director, External Reporting, Accounting and Control for the Gillette Company, a personal care company. Ms. Kramer also held partner positions in the public accounting firms of Ernst & Young LLP and Arthur Anderson LLP. Ms. Kramer is a licensed certified public accountant (CPA) in Massachusetts. She is a member of the Massachusetts Society of CPAs and the American Institute of CPAs. Ms. Kramer currently serves on the board of directors of Samsung Bioepis and on the board of directors of the Center for Women and Enterprise. Ms. Kramer previously served as a Board Member for the Massachusetts State Board of Accountancy from September 2011 to December 2015 and Probus Insurance Company Europe DAC from 2016 to 2018. | |||||
| Public Company Boards | |||||
| l | Armata Pharmaceuticals, Inc., a biotechnology company | ||||
| Education | |||||
| l | Salem State University, B.B.A. Accounting | ||||
| Alfred W. Sandrock, Jr., M.D., Ph.D. | |||||
| Experience | |||||
| Dr. Sandrock has served as our Executive Vice President, Research and Development since September 2019. Prior to that, Dr. Sandrock served as our Chief Medical Officer from October 2017 to January 2020, as our Executive Vice President, Chief Medical Officer Neurology and Neurodegeneration from October 2015 to October 2017, as our Chief Medical Officer and Group Senior Vice President from April 2013 to October 2015 and as our Chief Medical Officer and Senior Vice President of Development Sciences from February 2012 to April 2013. Prior to that, Dr. Sandrock held several other senior executive positions since joining Biogen in 1998, including Senior Vice President of Neurology Research and Development and Vice President of Clinical Development, Neurology. | |||||
| Education | |||||
| l | Stanford University, B.A. Human Biology | ||||
| l | Harvard Medical School, M.D. | ||||
| l | Harvard University, Ph.D. Neurobiology | ||||
| l | Massachusetts General Hospital, internship in Medicine, residency and chief residency in Neurology and clinical fellowship in Neuromuscular Disease and Clinical Neurophysiology (electromyography) | ||||
| Period | Total Number of Shares Purchased (#) | Average Price Paid per Share ($) | Total Number of Shares Purchased as Part of Publicly Announced Programs (#) | Approximate Dollar Value of Shares That May Yet Be Purchased Under Our Programs ($ in millions) | |||||||||||||||||||
| October 2020 | — | $ | — | — | $ | 5,000.0 | |||||||||||||||||
| November 2020 | — | $ | — | — | $ | 5,000.0 | |||||||||||||||||
| December 2020 | 1,620,969 | $ | 246.77 | — | $ | 4,600.0 | |||||||||||||||||
| Total | 1,620,969 | $ | 246.77 | ||||||||||||||||||||
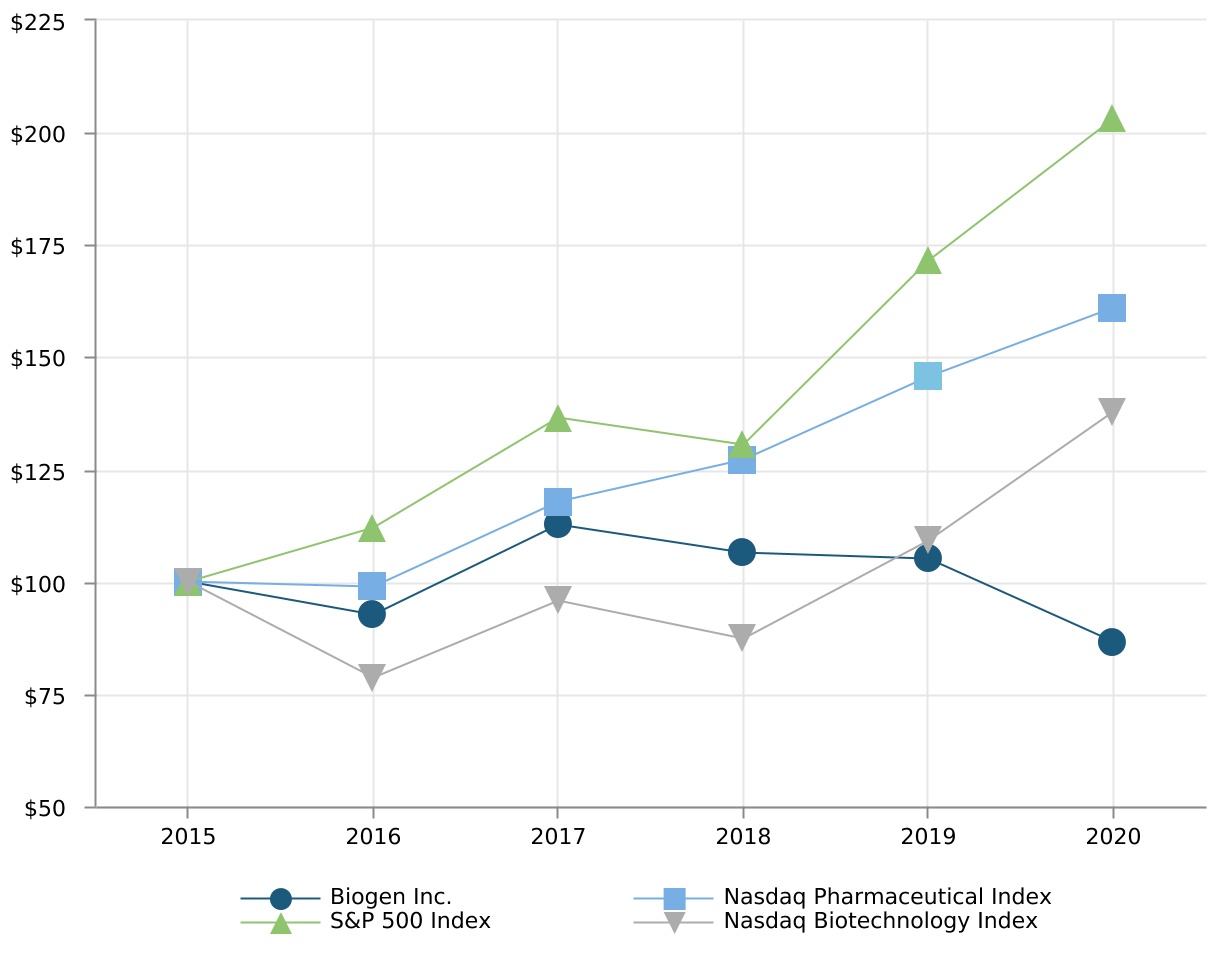
| 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | |||||||||||||||||||||||||||||||||
| Biogen Inc. | $100.00 | $92.57 | $112.74 | $106.49 | $105.01 | $86.65 | ||||||||||||||||||||||||||||||||
| Nasdaq Pharmaceutical Index | $100.00 | $98.91 | $117.83 | $127.20 | $145.65 | $160.97 | ||||||||||||||||||||||||||||||||
| S&P 500 Index | $100.00 | $111.96 | $136.40 | $130.42 | $171.49 | $203.04 | ||||||||||||||||||||||||||||||||
| Nasdaq Biotechnology Index | $100.00 | $78.65 | $95.69 | $87.21 | $109.11 | $137.94 | ||||||||||||||||||||||||||||||||
Our results of operations are summarized as follows: | |||||||||||||||||||||||||||||
| For the Years Ended December 31, | |||||||||||||||||||||||||||||
| (In millions, except per share amounts) | 2020 | 2019 | 2018 | 2017 | 2016 | ||||||||||||||||||||||||
| Results of Operations | |||||||||||||||||||||||||||||
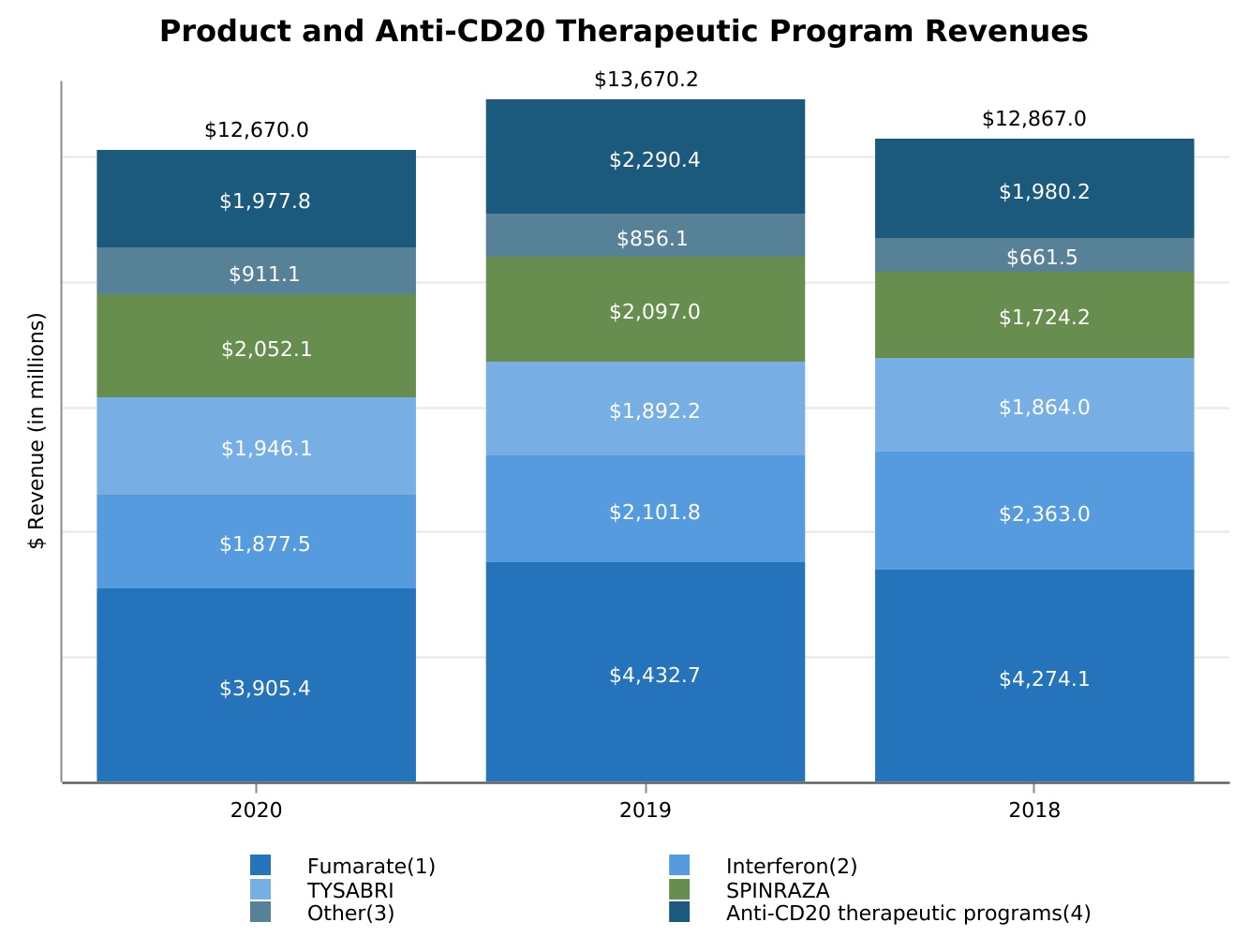
| Product revenues, net | $ | 10,692.2 | $ | 11,379.8 | $ | 10,886.8 | $ | 10,354.7 | $ | 9,817.9 | |||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | 1,977.8 | 2,290.4 | 1,980.2 | 1,559.2 | 1,314.5 | ||||||||||||||||||||||||
| Other revenues | 774.6 | 707.7 | 585.9 | 360.0 | 316.4 | ||||||||||||||||||||||||
| Total revenues | 13,444.6 | 14,377.9 | 13,452.9 | 12,273.9 | 11,448.8 | ||||||||||||||||||||||||
| Total cost and expenses | 8,894.5 | 7,335.3 | 7,564.3 | 6,928.1 | 6,297.1 | ||||||||||||||||||||||||
| Income from operations | 4,550.1 | 7,042.6 | 5,888.6 | 5,345.8 | 5,151.7 | ||||||||||||||||||||||||
| Other income (expense), net | 497.4 | 83.3 | 11.0 | (217.0) | (218.7) | ||||||||||||||||||||||||
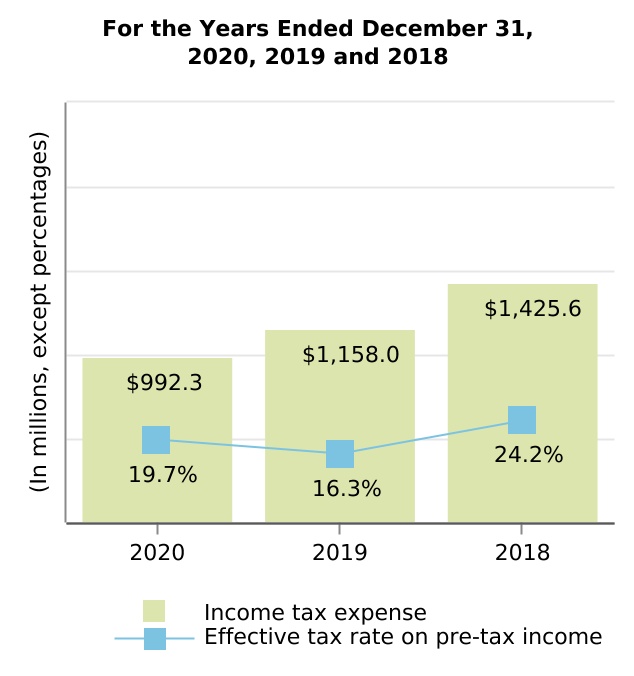
| Income before income tax expense and equity in loss of investee, net of tax | 5,047.5 | 7,125.9 | 5,899.6 | 5,128.8 | 4,933.0 | ||||||||||||||||||||||||
| Income tax expense | 992.3 | 1,158.0 | 1,425.6 | 2,458.7 | 1,237.3 | ||||||||||||||||||||||||
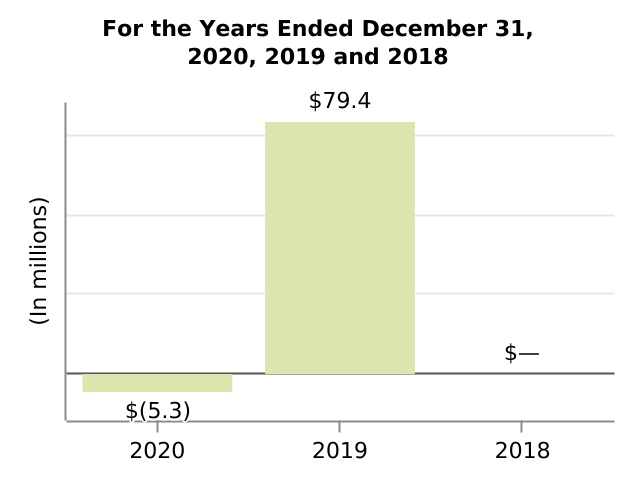
| Equity in loss of investee, net of tax | (5.3) | 79.4 | — | — | — | ||||||||||||||||||||||||
| Net income | 4,060.5 | 5,888.5 | 4,474.0 | 2,670.1 | 3,695.7 | ||||||||||||||||||||||||
| Net income (loss) attributable to noncontrolling interests, net of tax | 59.9 | — | 43.3 | 131.0 | (7.1) | ||||||||||||||||||||||||
| Net income attributable to Biogen Inc. | $ | 4,000.6 | $ | 5,888.5 | $ | 4,430.7 | $ | 2,539.1 | $ | 3,702.8 | |||||||||||||||||||
| Diluted Earnings Per Share | |||||||||||||||||||||||||||||
| Diluted earnings per share attributable to Biogen Inc. | $ | 24.80 | $ | 31.42 | $ | 21.58 | $ | 11.92 | $ | 16.93 | |||||||||||||||||||
| Weighted-average shares used in calculating diluted earnings per share attributable to Biogen Inc. | 161.3 | 187.4 | 205.3 | 213.0 | 218.8 | ||||||||||||||||||||||||
| Our financial condition is summarized as follows: | |||||||||||||||||||||||||||||
| As of December 31, | |||||||||||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | 2017 | 2016 | ||||||||||||||||||||||||
| Financial Condition | |||||||||||||||||||||||||||||
| Cash, cash equivalents and marketable securities | $ | 3,382.2 | $ | 5,884.0 | $ | 4,913.9 | $ | 6,746.3 | $ | 7,724.5 | |||||||||||||||||||
| Total assets | $ | 24,618.9 | $ | 27,234.3 | $ | 25,288.9 | $ | 23,652.6 | $ | 22,876.8 | |||||||||||||||||||
| Notes payable, less current portion | $ | 7,426.2 | $ | 4,459.0 | $ | 5,936.5 | $ | 5,935.0 | $ | 6,512.7 | |||||||||||||||||||
| Total Biogen Inc. shareholders’ equity | $ | 10,700.3 | $ | 13,343.2 | $ | 13,039.6 | $ | 12,612.8 | $ | 12,140.1 | |||||||||||||||||||
| For the Years Ended December 31, | % Change | $ Change | ||||||||||||||||||||||||||||||||||||||||||
| 2020 vs. 2019 | 2019 vs. 2018 | 2020 vs. 2019 | 2019 vs. 2018 | |||||||||||||||||||||||||||||||||||||||||
| (In millions, except percentages) | 2020 | 2019 | 2018 | |||||||||||||||||||||||||||||||||||||||||
| Product revenues, net: | ||||||||||||||||||||||||||||||||||||||||||||
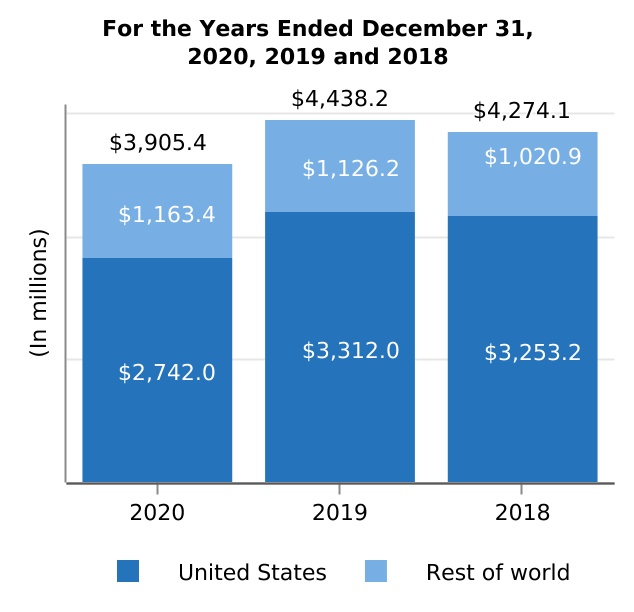
| United States | $ | 5,900.1 | $ | 6,713.8 | $ | 6,800.5 | (12.1) | % | (1.3) | % | $ | (813.7) | $ | (86.7) | ||||||||||||||||||||||||||||||
| Rest of world | 4,792.1 | 4,666.0 | 4,086.3 | 2.7 | 14.2 | 126.1 | 579.7 | |||||||||||||||||||||||||||||||||||||
| Total product revenues, net | 10,692.2 | 11,379.8 | 10,886.8 | (6.0) | 4.5 | (687.6) | 493.0 | |||||||||||||||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | 1,977.8 | 2,290.4 | 1,980.2 | (13.6) | 15.7 | (312.6) | 310.2 | |||||||||||||||||||||||||||||||||||||
| Other revenues | 774.6 | 707.7 | 585.9 | 9.5 | 20.8 | 66.9 | 121.8 | |||||||||||||||||||||||||||||||||||||
| Total revenues | $ | 13,444.6 | $ | 14,377.9 | $ | 13,452.9 | (6.5) | % | 6.9 | % | $ | (933.3) | $ | 925.0 | ||||||||||||||||||||||||||||||
| For the Years Ended December 31, | % Change | $ Change | ||||||||||||||||||||||||||||||||||||||||||
| 2020 vs. 2019 | 2019 vs. 2018 | 2020 vs. 2019 | 2019 vs. 2018 | |||||||||||||||||||||||||||||||||||||||||
| (In millions, except percentages) | 2020 | 2019 | 2018 | |||||||||||||||||||||||||||||||||||||||||
| Multiple Sclerosis (MS): | ||||||||||||||||||||||||||||||||||||||||||||
| Fumarate* | $ | 3,905.4 | $ | 4,438.2 | $ | 4,274.1 | (12.0) | % | 3.8 | % | $ | (532.8) | $ | 164.1 | ||||||||||||||||||||||||||||||
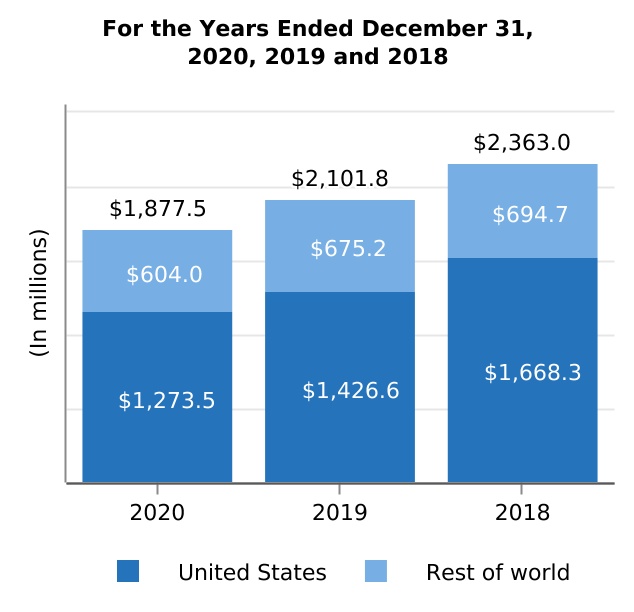
| Interferon** | 1,877.5 | 2,101.8 | 2,363.0 | (10.7) | (11.1) | (224.3) | (261.2) | |||||||||||||||||||||||||||||||||||||
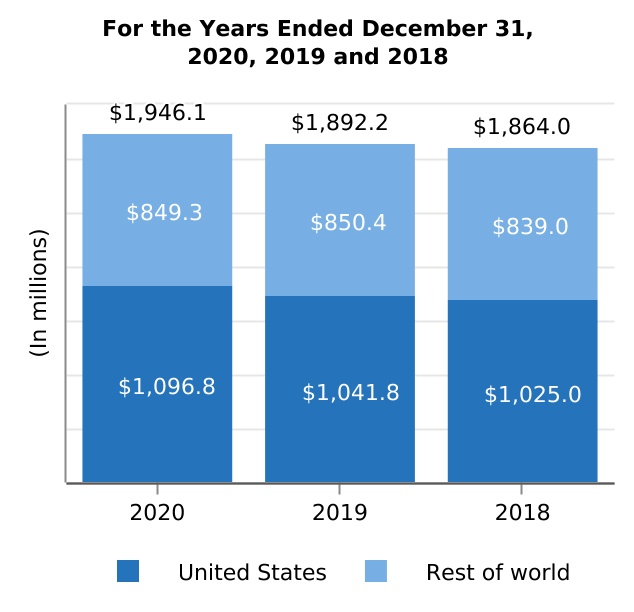
| TYSABRI | 1,946.1 | 1,892.2 | 1,864.0 | 2.8 | 1.5 | 53.9 | 28.2 | |||||||||||||||||||||||||||||||||||||
| FAMPYRA | 103.1 | 97.1 | 92.7 | 6.2 | 4.7 | 6.0 | 4.4 | |||||||||||||||||||||||||||||||||||||
| ZINBRYTA | — | — | 1.4 | — | nm | — | (1.4) | |||||||||||||||||||||||||||||||||||||
| Subtotal: MS product revenues | 7,832.1 | 8,529.3 | 8,595.2 | (8.2) | (0.8) | (697.2) | (65.9) | |||||||||||||||||||||||||||||||||||||
| Spinal Muscular Atrophy: | ||||||||||||||||||||||||||||||||||||||||||||
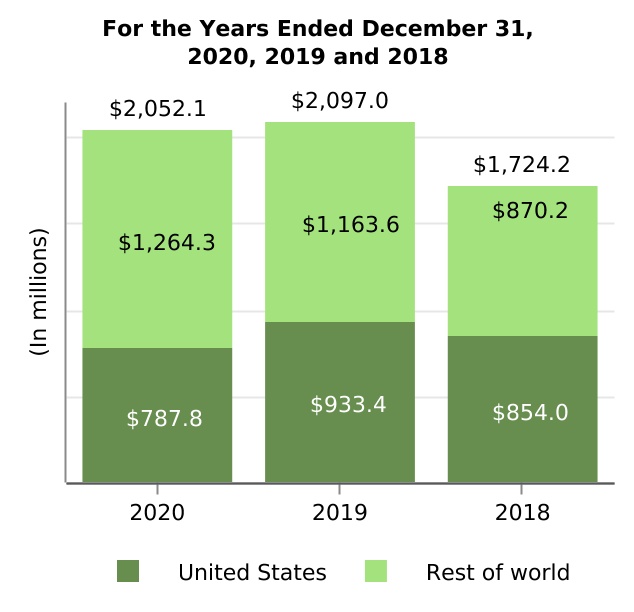
| SPINRAZA | 2,052.1 | 2,097.0 | 1,724.2 | (2.1) | 21.6 | (44.9) | 372.8 | |||||||||||||||||||||||||||||||||||||
| Biosimilars: | ||||||||||||||||||||||||||||||||||||||||||||
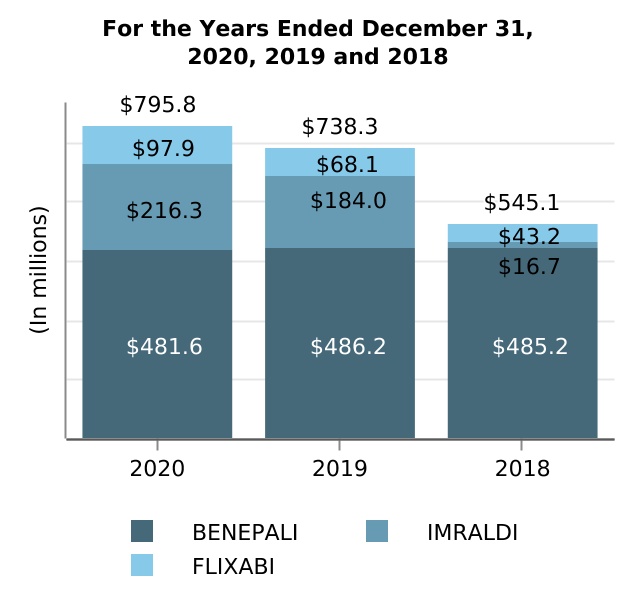
| BENEPALI | 481.6 | 486.2 | 485.2 | (0.9) | 0.2 | (4.6) | 1.0 | |||||||||||||||||||||||||||||||||||||
| IMRALDI | 216.3 | 184.0 | 16.7 | 17.6 | nm | 32.3 | 167.3 | |||||||||||||||||||||||||||||||||||||
| FLIXABI | 97.9 | 68.1 | 43.2 | 43.8 | 57.6 | 29.8 | 24.9 | |||||||||||||||||||||||||||||||||||||
| Subtotal: Biosimilar product revenues | 795.8 | 738.3 | 545.1 | 7.8 | 35.4 | 57.5 | 193.2 | |||||||||||||||||||||||||||||||||||||
| Other: | ||||||||||||||||||||||||||||||||||||||||||||
| FUMADERM | 12.2 | 15.2 | 22.3 | (19.7) | (31.8) | (3.0) | (7.1) | |||||||||||||||||||||||||||||||||||||
| Total product revenues, net | $ | 10,692.2 | $ | 11,379.8 | $ | 10,886.8 | (6.0) | % | 4.5 | % | $ | (687.6) | $ | 493.0 | ||||||||||||||||||||||||||||||
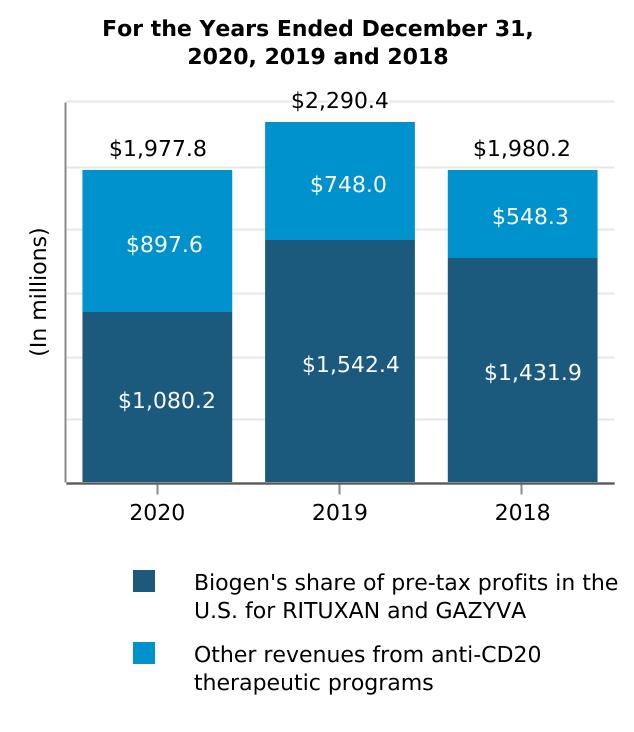
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Product revenues, net | $ | 3,334.1 | $ | 4,747.4 | $ | 4,484.3 | ||||||||||||||
| Cost and expenses | 433.0 | 622.7 | 669.6 | |||||||||||||||||
| Pre-tax profits in the U.S. | $ | 2,901.1 | $ | 4,124.7 | $ | 3,814.7 | ||||||||||||||
| Biogen's share of pre-tax profits | $ | 1,080.2 | $ | 1,542.4 | $ | 1,431.9 | ||||||||||||||
| For the Years Ended December 31, | % Change | $ Change | ||||||||||||||||||||||||||||||||||||||||||
| 2020 vs. 2019 | 2019 vs. 2018 | 2020 vs. 2019 | 2019 vs. 2018 | |||||||||||||||||||||||||||||||||||||||||
| (In millions, except percentages) | 2020 | 2019 | 2018 | |||||||||||||||||||||||||||||||||||||||||
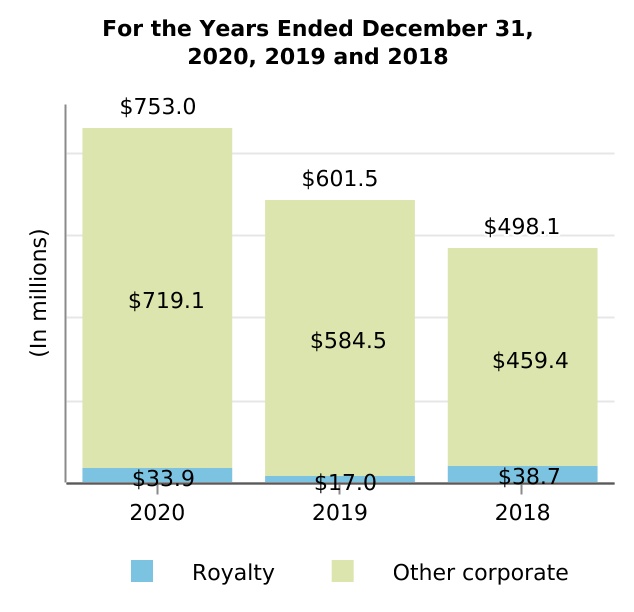
| Revenues from collaborative and other relationships | $ | 21.6 | $ | 106.2 | $ | 87.8 | (79.7) | % | 21.0 | % | $ | (84.6) | $ | 18.4 | ||||||||||||||||||||||||||||||
| Other royalty and corporate revenues | 753.0 | 601.5 | 498.1 | 25.2 | 20.8 | 151.5 | 103.4 | |||||||||||||||||||||||||||||||||||||
| Total other revenues | $ | 774.6 | $ | 707.7 | $ | 585.9 | 9.5 | % | 20.8 | % | $ | 66.9 | $ | 121.8 | ||||||||||||||||||||||||||||||
| For the Years Ended December 31, | % Change | $ Change | ||||||||||||||||||||||||||||||||||||||||||
| 2020 vs. 2019 | 2019 vs. 2018 | 2020 vs. 2019 | 2019 vs. 2018 | |||||||||||||||||||||||||||||||||||||||||
| (In millions, except percentages) | 2020 | 2019 | 2018 | |||||||||||||||||||||||||||||||||||||||||
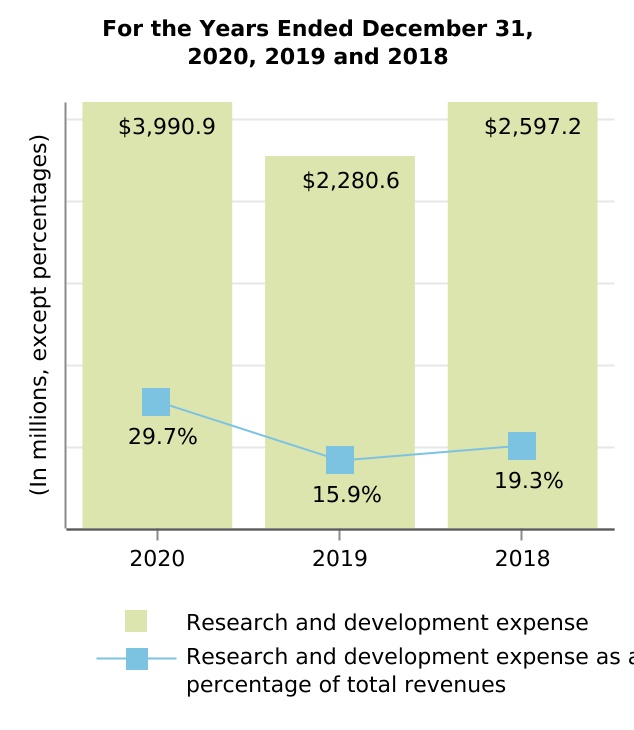
| Cost of sales, excluding amortization and impairment of acquired intangible assets | $ | 1,805.2 | $ | 1,955.4 | $ | 1,816.3 | (7.7) | % | 7.7 | % | $ | (150.2) | $ | 139.1 | ||||||||||||||||||||||||||||||
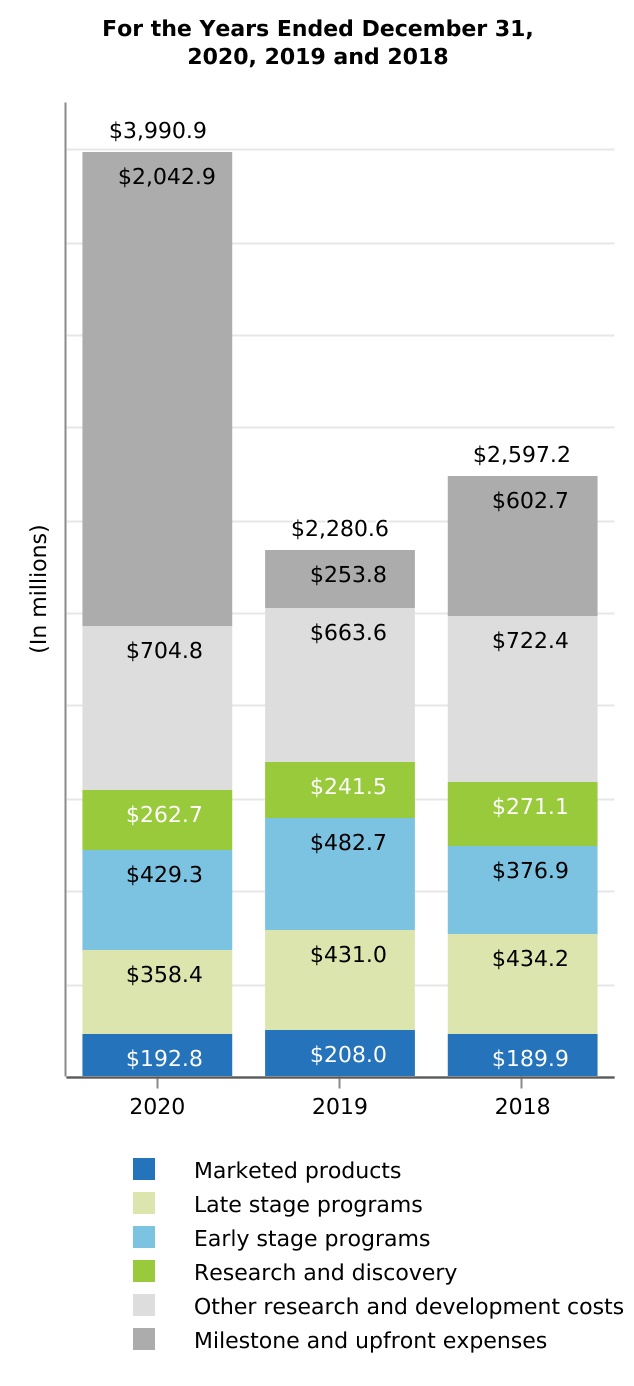
| Research and development | 3,990.9 | 2,280.6 | 2,597.2 | 75.0 | (12.2) | 1,710.3 | (316.6) | |||||||||||||||||||||||||||||||||||||
| Selling, general and administrative | 2,504.5 | 2,374.7 | 2,106.3 | 5.5 | 12.7 | 129.8 | 268.4 | |||||||||||||||||||||||||||||||||||||
| Amortization and impairment of acquired intangible assets | 464.8 | 489.9 | 747.3 | (5.1) | (34.4) | (25.1) | (257.4) | |||||||||||||||||||||||||||||||||||||
| Collaboration profit (loss) sharing | 232.9 | 241.6 | 185.0 | (3.6) | 30.6 | (8.7) | 56.6 | |||||||||||||||||||||||||||||||||||||
| (Gain) loss on divestiture of Hillerød, Denmark manufacturing operations | (92.5) | 55.3 | — | nm | nm | (147.8) | 55.3 | |||||||||||||||||||||||||||||||||||||
| (Gain) loss on fair value remeasurement of contingent consideration | (86.3) | (63.7) | (12.3) | 35.5 | 417.9 | (22.6) | (51.4) | |||||||||||||||||||||||||||||||||||||
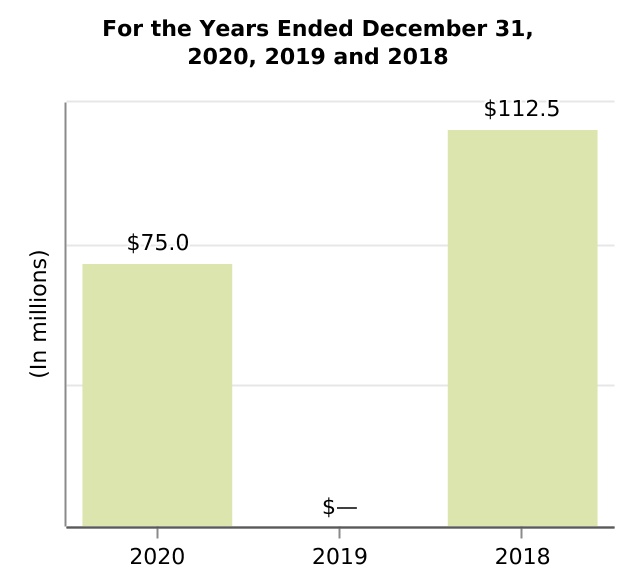
| Acquired in-process research and development | 75.0 | — | 112.5 | nm | nm | 75.0 | (112.5) | |||||||||||||||||||||||||||||||||||||
| Restructuring charges | — | 1.5 | 12.0 | nm | (87.5) | (1.5) | (10.5) | |||||||||||||||||||||||||||||||||||||
| Total cost and expenses | $ | 8,894.5 | $ | 7,335.3 | $ | 7,564.3 | 21.3 | % | (3.0) | % | $ | 1,559.2 | $ | (229.0) | ||||||||||||||||||||||||||||||

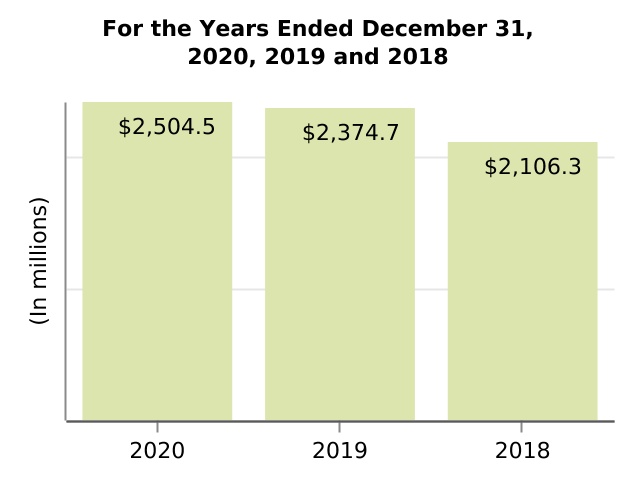
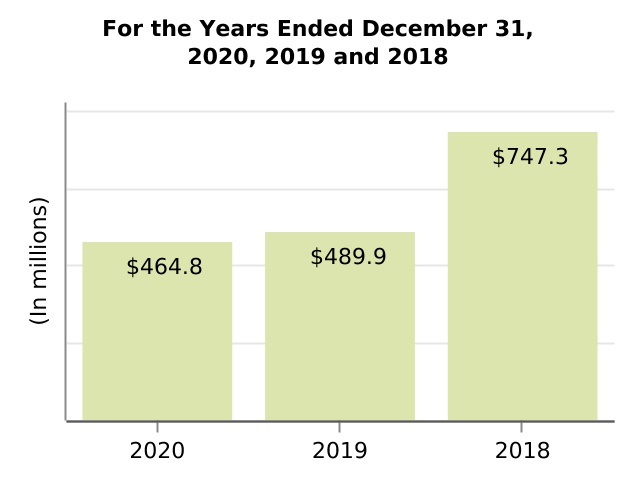
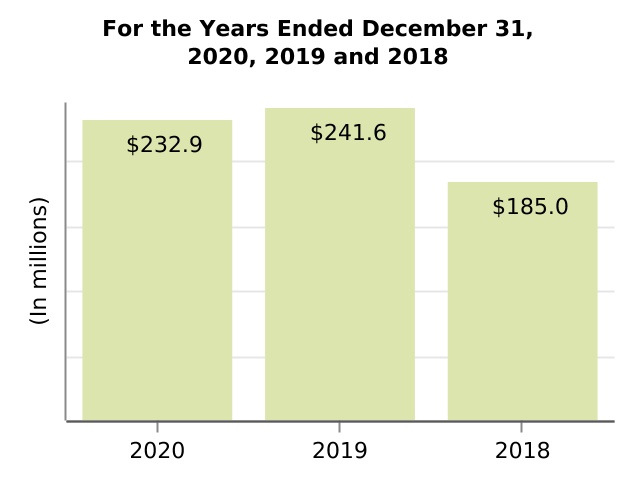
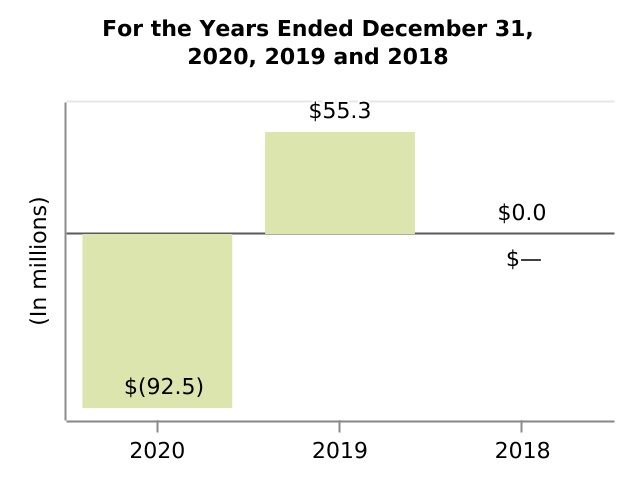
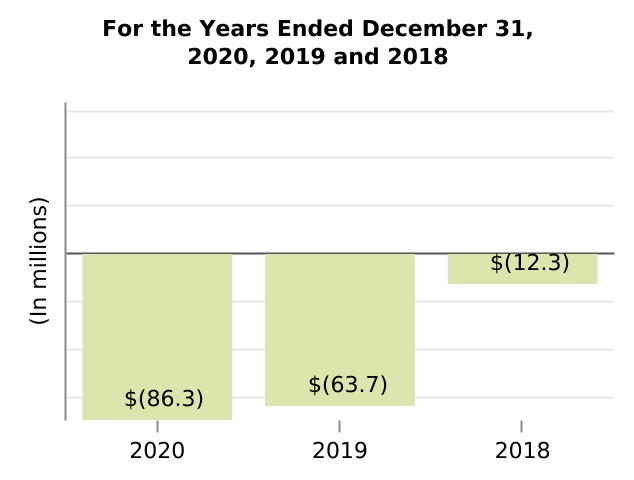
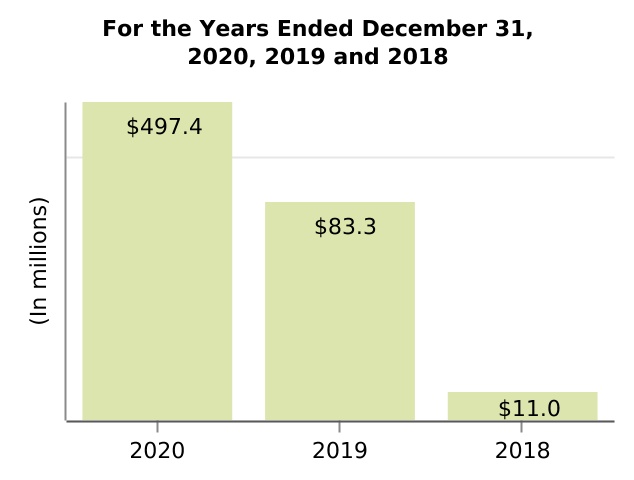
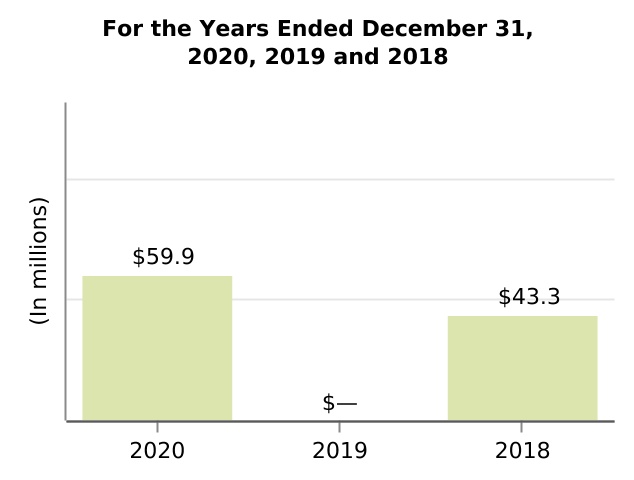
| As of December 31, | % Change | |||||||||||||||||||
| (In millions, except percentages) | 2020 | 2019 | 2020 vs. 2019 | |||||||||||||||||
| Financial assets: | ||||||||||||||||||||
| Cash and cash equivalents | $ | 1,331.2 | $ | 2,913.7 | (54.3) | % | ||||||||||||||
| Marketable securities — current | 1,278.9 | 1,562.2 | (18.1) | |||||||||||||||||
| Marketable securities — non-current | 772.1 | 1,408.1 | (45.2) | |||||||||||||||||
| Total cash, cash equivalents and marketable securities | $ | 3,382.2 | $ | 5,884.0 | (42.5) | % | ||||||||||||||
| Borrowings: | ||||||||||||||||||||
| Current portion of notes payable | $ | — | $ | 1,495.8 | nm | |||||||||||||||
| Notes payable | 7,426.2 | 4,459.0 | 66.5 | |||||||||||||||||
| Total borrowings | $ | 7,426.2 | $ | 5,954.8 | 24.7 | % | ||||||||||||||
| Working Capital: | ||||||||||||||||||||
| Current assets | $ | 6,887.1 | $ | 8,381.8 | (17.8) | % | ||||||||||||||
| Current liabilities | (3,742.2) | (4,863.8) | (23.1) | |||||||||||||||||
| Total working capital | $ | 3,144.9 | $ | 3,518.0 | (10.6) | % | ||||||||||||||
| For the Years Ended December 31, | % Change | |||||||||||||||||||||||||||||||
| 2020 vs. 2019 | 2019 vs. 2018 | |||||||||||||||||||||||||||||||
| (In millions, except percentages) | 2020 | 2019 | 2018 | |||||||||||||||||||||||||||||
| Net cash flows provided by operating activities | $ | 4,229.8 | $ | 7,078.6 | $ | 6,187.7 | (40.2) | % | 14.4 | % | ||||||||||||||||||||||
| Net cash flows provided by (used in) investing activities | (608.6) | 470.5 | (2,046.3) | (229.4) | % | nm | ||||||||||||||||||||||||||
| Net cash flows used in financing activities | (5,272.7) | (5,860.4) | (4,472.0) | 10.0 | (31.0) | |||||||||||||||||||||||||||
| Payments Due by Period | ||||||||||||||||||||||||||||||||
| (In millions) | Total | Less than 1 Year | 1 to 3 Years | 3 to 5 Years | After 5 Years | |||||||||||||||||||||||||||
Non-cancellable operating leases (1)(2) | $ | 387.8 | $ | 70.3 | $ | 123.6 | $ | 89.6 | $ | 104.3 | ||||||||||||||||||||||
Long-term debt obligations (3) | 11,853.0 | 279.1 | 1,512.9 | 2,218.0 | 7,843.0 | |||||||||||||||||||||||||||
Purchase and other obligations (4) | 1,248.0 | 398.3 | 420.0 | 424.6 | 5.1 | |||||||||||||||||||||||||||
| Defined benefit obligation | 151.2 | — | — | — | 151.2 | |||||||||||||||||||||||||||
| Total contractual obligations | $ | 13,640.0 | $ | 747.7 | $ | 2,056.5 | $ | 2,732.2 | $ | 8,103.6 | ||||||||||||||||||||||
| Financial Statements | Page Number | |||||||
| Consolidated Statements of Income | F-2 | |||||||
| Consolidated Statements of Comprehensive Income | F-3 | |||||||
| Consolidated Balance Sheets | F-4 | |||||||
| Consolidated Statements of Cash Flows | F-5 | |||||||
| Consolidated Statements of Equity | F-6 | |||||||
| Notes to Consolidated Financial Statements | F-9 | |||||||
| Report of Independent Registered Public Accounting Firm | F-79 | |||||||
| Exhibit No. | Description | |||||||
| 2.1† | ||||||||
| 2.2 | ||||||||
| 3.1 | ||||||||
| 3.2 | ||||||||
| 3.3 | ||||||||
| 4.1 | ||||||||
| 4.1 | ||||||||
| 4.2 | ||||||||
| 4.3 | ||||||||
| 4.4+ | ||||||||
| 10.1 | ||||||||
| 10.2 | ||||||||
| 10.3† | ||||||||
| 10.4† | ||||||||
| 10.5 | ||||||||
| 10.6* | ||||||||
| 10.7* | ||||||||
| 10.8* | ||||||||
| 10.9* | ||||||||
| 10.10* | ||||||||
| 10.11* | ||||||||
| 10.12* | ||||||||
| Exhibit No. | Description | |||||||
| 10.13* | ||||||||
| 10.14* | ||||||||
| 10.15* | ||||||||
| 10.16* | ||||||||
| 10.17* | ||||||||
| 10.18* | ||||||||
| 10.19* | ||||||||
| 10.20* | ||||||||
| 10.21* | ||||||||
| 10.22* | ||||||||
| 10.23* | ||||||||
| 10.24* | ||||||||
| 10.25* | ||||||||
| 10.26* | ||||||||
| 10.27* | ||||||||
| 10.28* | ||||||||
| 10.29* | ||||||||
| 10.30* | ||||||||
| 10.31* | ||||||||
| 10.32* | ||||||||
| 10.33* | ||||||||
| 10.34* | ||||||||
| Exhibit No. | Description | |||||||
| 10.35* | ||||||||
| 10.36* | ||||||||
| 10.37* | ||||||||
| 10.38* | ||||||||
| 10.39* | ||||||||
| 10.40* | ||||||||
| 10.41*+ | ||||||||
| 10.42* | ||||||||
| 10.43* | ||||||||
| 10.44* | ||||||||
| 21+ | ||||||||
| 23+ | ||||||||
| 31.1+ | ||||||||
| 31.2+ | ||||||||
| 32.1++ | ||||||||
| 101++ | The following materials from Biogen Inc.’s Annual Report on Form 10-K for the year ended December 31, 2020, formatted in iXBRL (Inline Extensible Business Reporting Language): (i) the Consolidated Statements of Income, (ii) the Consolidated Statements of Comprehensive Income, (iii) the Consolidated Balance Sheets, (iv) the Consolidated Statements of Cash Flows, (v) the Consolidated Statements of Equity and (vi) Notes to Consolidated Financial Statements. | |||||||
| * | Management contract or compensatory plan or arrangement. | ||||
| † | Confidential treatment has been granted or requested with respect to portions of this exhibit. | ||||
| + | Filed herewith. | ||||
| ++ | Furnished herewith. | ||||
| BIOGEN INC. | |||||
| By: | /S/ MICHEL VOUNATSOS | ||||
| Michel Vounatsos | |||||
| Chief Executive Officer | |||||
| Name | Capacity | Date | ||||||||||||
/S/ MICHEL VOUNATSOS | Director and Chief Executive Officer (principal executive officer) | February 3, 2021 | ||||||||||||
| Michel Vounatsos | ||||||||||||||
/S/ MICHAEL R. MCDONNELL | Executive Vice President and Chief Financial Officer (principal financial officer) | February 3, 2021 | ||||||||||||
| Michael R. McDonnell | ||||||||||||||
/S/ ROBIN C. KRAMER | Senior Vice President, Chief Accounting Officer (principal accounting officer) | February 3, 2021 | ||||||||||||
| Robin C. Kramer | ||||||||||||||
/S/ STELIOS PAPADOPOULOS | Director and Chairman of the Board of Directors | February 3, 2021 | ||||||||||||
| Stelios Papadopoulos | ||||||||||||||
/S/ ALEXANDER J. DENNER | Director | February 3, 2021 | ||||||||||||
| Alexander J. Denner | ||||||||||||||
/S/ CAROLINE D. DORSA | Director | February 3, 2021 | ||||||||||||
| Caroline D. Dorsa | ||||||||||||||
/S/ WILLIAM A. HAWKINS | Director | February 3, 2021 | ||||||||||||
| William A. Hawkins | ||||||||||||||
/S/ NANCY L. LEAMING | Director | February 3, 2021 | ||||||||||||
| Nancy L. Leaming | ||||||||||||||
/S/ JESUS B. MANTAS | Director | February 3, 2021 | ||||||||||||
| Jesus B. Mantas | ||||||||||||||
/S/ RICHARD C. MULLIGAN | Director | February 3, 2021 | ||||||||||||
| Richard C. Mulligan | ||||||||||||||
/S/ ROBERT W. PANGIA | Director | February 3, 2021 | ||||||||||||
| Robert W. Pangia | ||||||||||||||
/S/ BRIAN S. POSNER | Director | February 3, 2021 | ||||||||||||
| Brian S. Posner | ||||||||||||||
/S/ ERIC K. ROWINSKY | Director | February 3, 2021 | ||||||||||||
| Eric K. Rowinsky | ||||||||||||||
/S/ STEPHEN A. SHERWIN | Director | February 3, 2021 | ||||||||||||
| Stephen A. Sherwin | ||||||||||||||
| Page Number | ||||||||
| Consolidated Statements of Income | F-2 | |||||||
| Consolidated Statements of Comprehensive Income | F-3 | |||||||
| Consolidated Balance Sheets | F-4 | |||||||
| Consolidated Statements of Cash Flows | F-5 | |||||||
| Consolidated Statements of Equity | F-6 | |||||||
| Notes to Consolidated Financial Statements | F-9 | |||||||
| Report of Independent Registered Public Accounting Firm | F-80 | |||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| 2020 | 2019 | 2018 | ||||||||||||||||||
| Revenues: | ||||||||||||||||||||
| Product, net | $ | $ | $ | |||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | ||||||||||||||||||||
| Other | ||||||||||||||||||||
| Total revenues | ||||||||||||||||||||
| Cost and expenses: | ||||||||||||||||||||
| Cost of sales, excluding amortization and impairment of acquired intangible assets | ||||||||||||||||||||
| Research and development | ||||||||||||||||||||
| Selling, general and administrative | ||||||||||||||||||||
| Amortization and impairment of acquired intangible assets | ||||||||||||||||||||
| Collaboration profit (loss) sharing | ||||||||||||||||||||
| (Gain) loss on divestiture of Hillerød, Denmark manufacturing operations | ( | |||||||||||||||||||
| (Gain) loss on fair value remeasurement of contingent consideration | ( | ( | ( | |||||||||||||||||
| Acquired in-process research and development | ||||||||||||||||||||
| Restructuring charges | ||||||||||||||||||||
| Total cost and expenses | ||||||||||||||||||||
| Income from operations | ||||||||||||||||||||
| Other income (expense), net | ||||||||||||||||||||
| Income before income tax expense and equity in loss of investee, net of tax | ||||||||||||||||||||
| Income tax expense | ||||||||||||||||||||
| Equity in (income) loss of investee, net of tax | ( | |||||||||||||||||||
| Net income | ||||||||||||||||||||
| Net income (loss) attributable to noncontrolling interests, net of tax | ||||||||||||||||||||
| Net income attributable to Biogen Inc. | $ | $ | $ | |||||||||||||||||
| Net income per share: | ||||||||||||||||||||
| Basic earnings per share attributable to Biogen Inc. | $ | $ | $ | |||||||||||||||||
| Diluted earnings per share attributable to Biogen Inc. | $ | $ | $ | |||||||||||||||||
| Weighted-average shares used in calculating: | ||||||||||||||||||||
| Basic earnings per share attributable to Biogen Inc. | ||||||||||||||||||||
| Diluted earnings per share attributable to Biogen Inc. | ||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| 2020 | 2019 | 2018 | ||||||||||||||||||
| Net income attributable to Biogen Inc. | $ | $ | $ | |||||||||||||||||
| Other comprehensive income: | ||||||||||||||||||||
| Unrealized gains (losses) on securities available for sale, net of tax | ( | ( | ||||||||||||||||||
| Unrealized gains (losses) on cash flow hedges, net of tax | ( | ( | ||||||||||||||||||
| Gains (losses) on net investment hedges, net of tax | ( | |||||||||||||||||||
| Unrealized gains (losses) on pension benefit obligation, net of tax | ( | ( | ||||||||||||||||||
| Currency translation adjustment | ( | |||||||||||||||||||
| Total other comprehensive income (loss), net of tax | ( | |||||||||||||||||||
| Comprehensive income attributable to Biogen Inc. | ||||||||||||||||||||
| Comprehensive income (loss) attributable to noncontrolling interests, net of tax | ( | |||||||||||||||||||
| Comprehensive income | $ | $ | $ | |||||||||||||||||
| As of December 31, | |||||||||||
| 2020 | 2019 | ||||||||||
| ASSETS | |||||||||||
| Current assets: | |||||||||||
| Cash and cash equivalents | $ | $ | |||||||||
| Marketable securities | |||||||||||
| Accounts receivable, net | |||||||||||
| Due from anti-CD20 therapeutic programs | |||||||||||
| Inventory | |||||||||||
| Other current assets | |||||||||||
| Total current assets | |||||||||||
| Marketable securities | |||||||||||
| Property, plant and equipment, net | |||||||||||
| Operating lease assets | |||||||||||
| Intangible assets, net | |||||||||||
| Goodwill | |||||||||||
| Deferred tax asset | |||||||||||
| Investments and other assets | |||||||||||
| Total assets | $ | $ | |||||||||
| LIABILITIES AND EQUITY | |||||||||||
| Current liabilities: | |||||||||||
| Current portion of notes payable | $ | $ | |||||||||
| Taxes payable | |||||||||||
| Accounts payable | |||||||||||
| Accrued expenses and other | |||||||||||
| Total current liabilities | |||||||||||
| Notes payable | |||||||||||
| Deferred tax liability | |||||||||||
| Long-term operating lease liabilities | |||||||||||
| Other long-term liabilities | |||||||||||
| Total liabilities | |||||||||||
| Commitments, contingencies and guarantees (Notes 21 and 22) | |||||||||||
| Equity: | |||||||||||
| Biogen Inc. shareholders’ equity | |||||||||||
| Preferred stock, par value $0.001 per share | |||||||||||
| Common stock, par value $0.0005 per share | |||||||||||
| Additional paid-in capital | |||||||||||
| Accumulated other comprehensive loss | ( | ( | |||||||||
| Retained earnings | |||||||||||
| Treasury stock, at cost; 23.8 million and 23.8 million shares, respectively | ( | ( | |||||||||
| Total Biogen Inc. shareholders’ equity | |||||||||||
| Noncontrolling interests | ( | ( | |||||||||
| Total equity | |||||||||||
| Total liabilities and equity | $ | $ | |||||||||
| For the Years Ended December 31, | |||||||||||||||||
| 2020 | 2019 | 2018 | |||||||||||||||
| Cash flows from operating activities: | |||||||||||||||||
| Net income | $ | $ | $ | ||||||||||||||
| Adjustments to reconcile net income to net cash flows from operating activities: | |||||||||||||||||
| Depreciation and amortization | |||||||||||||||||
| Impairment of intangible assets | |||||||||||||||||
| Acquired in-process research and development | |||||||||||||||||
| Share-based compensation | |||||||||||||||||
| Contingent consideration | ( | ( | ( | ||||||||||||||
| (Gain)/loss on divestiture of Hillerod, Denmark manufactuing operations | ( | ||||||||||||||||
| Deferred income taxes | |||||||||||||||||
| Unrealized (gain) loss on strategic investments | ( | ( | ( | ||||||||||||||
| Loss on equity method investment | ( | ||||||||||||||||
| Other | |||||||||||||||||
| Changes in operating assets and liabilities, net: | |||||||||||||||||
| Accounts receivable | ( | ||||||||||||||||
| Due from anti-CD20 therapeutic programs | ( | ||||||||||||||||
| Inventory | ( | ( | ( | ||||||||||||||
| Accrued expenses and other current liabilities | |||||||||||||||||
| Income tax assets and liabilities | ( | ||||||||||||||||
| Other changes in operating assets and liabilities, net | ( | ( | ( | ||||||||||||||
| Net cash flows provided by operating activities | |||||||||||||||||
| Cash flows from investing activities: | |||||||||||||||||
| Proceeds from sales and maturities of marketable securities | |||||||||||||||||
| Purchases of marketable securities | ( | ( | ( | ||||||||||||||
| Contingent consideration paid related to Fumapharm AG acquisition | ( | ( | |||||||||||||||
| Acquisition of Nightstar Therapeutics plc, net of cash acquired | ( | ||||||||||||||||
| Purchase of Ionis Pharmaceuticals, Inc. stock | ( | ||||||||||||||||
| Purchase of Sangamo Therapeutics, Inc. stock | ( | ||||||||||||||||
| Purchase of Denali Therapeutics Inc. stock | ( | ||||||||||||||||
| Purchase of Sage Therapeutics, Inc. stock | ( | ||||||||||||||||
| Proceeds from divesiture of Hillerod, Denmark manufacturing operations | |||||||||||||||||
| Purchases of property, plant and equipment | ( | ( | ( | ||||||||||||||
| Acquired in-process research and development | ( | ( | |||||||||||||||
| Acquisitions of intangible assets | ( | ( | ( | ||||||||||||||
| Investment in Samsung Bioepis | ( | ||||||||||||||||
| Proceeds from sales of strategic investments | |||||||||||||||||
| Other | ( | ||||||||||||||||
| Net cash flows provided by (used in) investing activities | ( | ( | |||||||||||||||
| Cash flows from financing activities: | |||||||||||||||||
| Purchase of treasury stock | ( | ( | ( | ||||||||||||||
| Payments related to issuance of stock for share-based compensation arrangements, net | ( | ||||||||||||||||
| Proceeds from borrowings | |||||||||||||||||
| Repayments of borrowings | ( | ( | |||||||||||||||
| Net contribution (distribution) to noncontrolling interest | ( | ( | |||||||||||||||
| Contingent consideration payments | ( | ||||||||||||||||
| Other | ( | ||||||||||||||||
| Net cash flows (used in) financing activities | ( | ( | ( | ||||||||||||||
| Net increase (decrease) in cash and cash equivalents | ( | ( | |||||||||||||||
| Effect of exchange rate changes on cash and cash equivalents | ( | ||||||||||||||||
| Cash and cash equivalents, beginning of the year | |||||||||||||||||
| Cash and cash equivalents, end of the year | $ | $ | $ | ||||||||||||||
| Preferred stock | Common stock | Additional paid-in capital | Accumulated other comprehensive loss | Retained earnings | Treasury stock | Total Biogen Inc. shareholders’ equity | Noncontrolling interests | Total equity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2019 | $ | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | — | — | — | — | — | ( | — | — | — | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distribution to noncontrolling interest | — | — | — | — | — | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Capital contribution from noncontrolling interest | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the 2020 Share Repurchase Program, at cost | — | — | — | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the 2020 Share Repurchase Program, at cost | — | — | ( | — | ( | — | ( | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the December 2019 Share Repurchase Program, at cost | — | — | — | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the December 2019 Share Repurchase Program, at cost | — | — | ( | — | ( | — | ( | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the March 2019 Share Repurchase Program, at cost | — | — | — | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the March 2019 Share Repurchase Program, at cost | — | — | ( | — | ( | — | ( | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan | — | — | — | — | ( | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Compensation expense related to share-based payments | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | — | — | — | ( | — | — | — | — | ( | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2020 | $ | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Preferred stock | Common stock | Additional paid-in capital | Accumulated other comprehensive loss | Retained earnings | Treasury stock | Total Biogen Inc. shareholders’ equity | Noncontrolling interests | Total equity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2018 | $ | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | — | — | — | — | — | — | — | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Capital contribution from noncontrolling interest | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the March 2019 Share Repurchase Program, at cost | — | — | — | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the March 2019 Share Repurchase Program, at cost | — | — | ( | ( | — | ( | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the 2018 Share Repurchase Program, at cost | — | — | — | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the 2018 Share Repurchase Program, at cost | — | — | ( | ( | — | ( | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan | — | — | — | — | ( | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Compensation related to share-based payments | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2019 | $ | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Preferred stock | Common stock | Additional paid-in capital | Accumulated other comprehensive loss | Retained earnings | Treasury stock | Total Biogen Inc. shareholders’ equity | Noncontrolling interests | Total equity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2017 | $ | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | — | — | — | — | — | — | — | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Capital contribution from noncontrolling interests | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distribution to noncontrolling interests | — | — | — | — | — | — | — | — | — | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the 2018 Share Repurchase Program, at cost | — | — | — | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the 2018 Share Repurchase Program, at cost | — | — | ( | ( | — | ( | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the 2016 Share Repurchase Program, at cost | — | — | — | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the 2016 Share Repurchase Program, at cost | — | — | ( | ( | — | ( | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan | — | — | ( | — | — | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Compensation expense related to share-based payments | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adoption of new accounting guidance | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2018 | $ | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asset Category | Useful Lives | ||||
| Land | Not depreciated | ||||
| Buildings | 15 to 40 years | ||||
| Leasehold Improvements | Lesser of the useful life or the term of the respective lease | ||||
| Furniture and Fixtures | 5 to 7 years | ||||
| Machinery and Equipment | 5 to 20 years | ||||
| Computer Software and Hardware | 3 to 5 years | ||||
| For the Years Ended December 31, | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | 2018 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (In millions) | United States | Rest of World | Total | United States | Rest of World | Total | United States | Rest of World | Total | |||||||||||||||||||||||||||||||||||||||||||||||
| Multiple Sclerosis (MS): | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fumarate* | $ | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||
| Interferon** | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TYSABRI | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FAMPYRA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ZINBRYTA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Subtotal: MS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spinal Muscular Atrophy: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SPINRAZA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biosimilars: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BENEPALI | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IMRALDI | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FLIXABI | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Subtotal: Biosimilars | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FUMADERM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total product revenues | $ | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2020 | ||||||||||||||||||||||||||
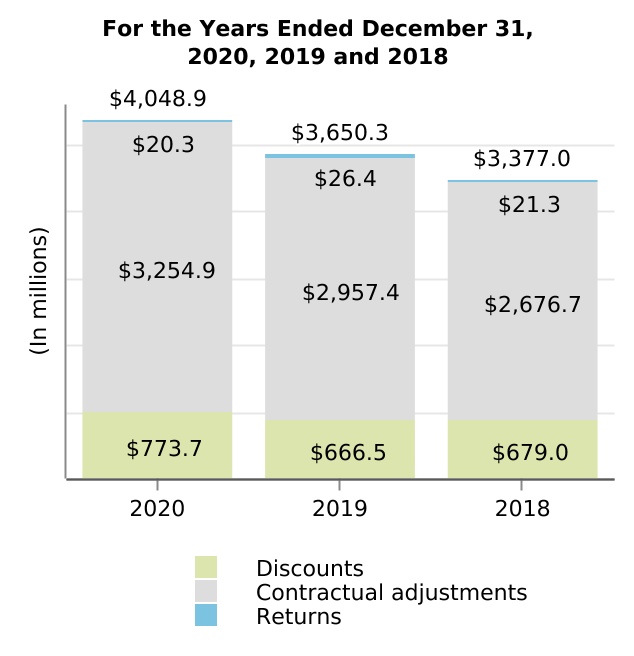
| (In millions) | Discounts | Contractual Adjustments | Returns | Total | ||||||||||||||||||||||
| Beginning balance | $ | $ | $ | $ | ||||||||||||||||||||||
| Current provisions relating to sales in current year | ||||||||||||||||||||||||||
| Adjustments relating to prior years | ( | ( | ( | |||||||||||||||||||||||
| Payments/returns relating to sales in current year | ( | ( | ( | |||||||||||||||||||||||
| Payments/returns relating to sales in prior years | ( | ( | ( | ( | ||||||||||||||||||||||
| Ending balance | $ | $ | $ | $ | ||||||||||||||||||||||
| December 31, 2019 | ||||||||||||||||||||||||||
| (In millions) | Discounts | Contractual Adjustments | Returns | Total | ||||||||||||||||||||||
| Beginning balance | $ | $ | $ | $ | ||||||||||||||||||||||
| Current provisions relating to sales in current year | ||||||||||||||||||||||||||
| Adjustments relating to prior years | ( | ( | ||||||||||||||||||||||||
| Payments/returns relating to sales in current year | ( | ( | ( | ( | ||||||||||||||||||||||
| Payments/returns relating to sales in prior years | ( | ( | ( | ( | ||||||||||||||||||||||
| Ending balance | $ | $ | $ | $ | ||||||||||||||||||||||
| December 31, 2018 | ||||||||||||||||||||||||||
| (In millions) | Discounts | Contractual Adjustments | Returns | Total | ||||||||||||||||||||||
| Beginning balance | $ | $ | $ | $ | ||||||||||||||||||||||
| Current provisions relating to sales in current year | ||||||||||||||||||||||||||
| Adjustments relating to prior years | ( | ( | ( | ( | ||||||||||||||||||||||
| Payments/returns relating to sales in current year | ( | ( | ( | ( | ||||||||||||||||||||||
| Payments/returns relating to sales in prior years | ( | ( | ( | ( | ||||||||||||||||||||||
| Ending balance | $ | $ | $ | $ | ||||||||||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Reduction of accounts receivable | $ | $ | ||||||||||||
| Component of accrued expenses and other | ||||||||||||||
| Total revenue-related reserves | $ | $ | ||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Biogen's share of pre-tax profits in the U.S. for RITUXAN and GAZYVA | $ | $ | $ | |||||||||||||||||
| Other revenues from anti-CD20 therapeutic programs | ||||||||||||||||||||
| Total revenues from anti-CD20 therapeutic programs | $ | $ | $ | |||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Revenues from collaborative and other relationships: | ||||||||||||||||||||
| Revenues earned under our technical development agreement, manufacturing service agreements and royalty revenues on biosimilar products with Samsung Bioepis | $ | $ | $ | |||||||||||||||||
| Other revenues from collaborative and other relationships | ( | |||||||||||||||||||
| Other royalty and corporate revenues: | ||||||||||||||||||||
| Royalty | ||||||||||||||||||||
| Other corporate | ||||||||||||||||||||
| Total other revenues | $ | $ | $ | |||||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Raw materials | $ | $ | ||||||||||||
| Work in process | ||||||||||||||
| Finished goods | ||||||||||||||
| Total inventory | $ | $ | ||||||||||||
| As of December 31, 2020 | As of December 31, 2019 | ||||||||||||||||||||||||||||||||||||||||
| (In millions) | Estimated Life | Cost | Accumulated Amortization | Net | Cost | Accumulated Amortization | Net | ||||||||||||||||||||||||||||||||||
| Completed technology | 4-28 years | $ | $ | ( | $ | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||
| In-process research and development | Indefinite until commercialization | — | — | ||||||||||||||||||||||||||||||||||||||
| Trademarks and trade names | Indefinite | — | — | ||||||||||||||||||||||||||||||||||||||
| Total intangible assets | $ | $ | ( | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||
| (In millions) | As of December 31, 2020 | |||||||
| 2021 | $ | |||||||
| 2022 | ||||||||
| 2023 | ||||||||
| 2024 | ||||||||
| 2025 | ||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Goodwill, beginning of year | $ | $ | ||||||||||||
| Increase to goodwill | ||||||||||||||
| Elimination of goodwill allocated to Hillerød, Denmark manufacturing operations | ( | |||||||||||||
| Other | ||||||||||||||
| Goodwill, end of year | $ | $ | ||||||||||||
| As of December 31, 2020 | ||||||||||||||||||||||||||
| (In millions) | Total | Quoted Prices in Active Markets (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | ||||||||||||||||||||||
| Assets: | ||||||||||||||||||||||||||
| Cash equivalents | $ | $ | $ | $ | ||||||||||||||||||||||
| Marketable debt securities: | ||||||||||||||||||||||||||
| Corporate debt securities | ||||||||||||||||||||||||||
| Government securities | ||||||||||||||||||||||||||
| Mortgage and other asset backed securities | ||||||||||||||||||||||||||
| Marketable equity securities | ||||||||||||||||||||||||||
| Derivative contracts | ||||||||||||||||||||||||||
| Plan assets for deferred compensation | ||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||||||||||||
| Liabilities: | ||||||||||||||||||||||||||
| Derivative contracts | $ | $ | $ | $ | ||||||||||||||||||||||
| Contingent consideration obligations | ||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||||||||||||
| As of December 31, 2019 | ||||||||||||||||||||||||||
| (In millions) | Total | Quoted Prices in Active Markets (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | ||||||||||||||||||||||
| Assets: | ||||||||||||||||||||||||||
| Cash equivalents | $ | $ | $ | $ | ||||||||||||||||||||||
| Marketable debt securities: | ||||||||||||||||||||||||||
| Corporate debt securities | ||||||||||||||||||||||||||
| Government securities | ||||||||||||||||||||||||||
| Mortgage and other asset backed securities | ||||||||||||||||||||||||||
| Marketable equity securities | ||||||||||||||||||||||||||
| Derivative contracts | ||||||||||||||||||||||||||
| Plan assets for deferred compensation | ||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||||||||||||
| Liabilities: | ||||||||||||||||||||||||||
| Derivative contracts | $ | $ | $ | $ | ||||||||||||||||||||||
| Contingent consideration obligations | ||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||||||||||||
| As of December 31, 2020 | ||||||||||||||||||||||||||||||||
| (In millions) | Fair Value | Valuation Technique | Unobservable Input | Range | Weighted Average | |||||||||||||||||||||||||||
| Liabilities: | ||||||||||||||||||||||||||||||||
| Contingent consideration obligation | $ | Discounted cash flow | Discount rate | |||||||||||||||||||||||||||||
| Expected timing of achievement of development milestones | 2021 to 2025 | — | ||||||||||||||||||||||||||||||
| As of December 31, 2020 | ||||||||||||||||||||
| (In millions) | Beginning Book Value | Impairment | Ending Book Value | |||||||||||||||||
| BIIB111 intangible asset | $ | $ | ( | $ | ||||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
2.900% Senior Notes due September 15, 2020(1) | $ | $ | ||||||||||||
| 3.625% Senior Notes due September 15, 2022 | ||||||||||||||
| 4.050% Senior Notes due September 15, 2025 | ||||||||||||||
| 2.250% Senior Notes due May 1, 2030 | ||||||||||||||
| 5.200% Senior Notes due September 15, 2045 | ||||||||||||||
| 3.150% Senior Notes due May 1, 2050 | ||||||||||||||
| Total | $ | $ | ||||||||||||
| As of December 31, | |||||||||||||||||
| (In millions) | 2020 | 2019 | |||||||||||||||
| Fair value, beginning of year | $ | $ | |||||||||||||||
| Changes in fair value | ( | ( | |||||||||||||||
| Payments and other | |||||||||||||||||
| Fair value, end of year | $ | $ | |||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Commercial paper | $ | $ | ||||||||||||
| Overnight reverse repurchase agreements | ||||||||||||||
| Money market funds | ||||||||||||||
| Short-term debt securities | ||||||||||||||
| Total | $ | $ | ||||||||||||
| As of December 31, 2020 | ||||||||||||||||||||||||||
| (In millions) | Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Fair Value | ||||||||||||||||||||||
| Corporate debt securities | ||||||||||||||||||||||||||
| Current | $ | $ | $ | ( | $ | |||||||||||||||||||||
| Non-current | ( | |||||||||||||||||||||||||
| Government securities | ||||||||||||||||||||||||||
| Current | ||||||||||||||||||||||||||
| Non-current | ||||||||||||||||||||||||||
| Mortgage and other asset backed securities | ||||||||||||||||||||||||||
| Current | ||||||||||||||||||||||||||
| Non-current | ( | |||||||||||||||||||||||||
| Total marketable debt securities | $ | $ | $ | ( | $ | |||||||||||||||||||||
| Marketable equity securities, current | $ | $ | $ | $ | ||||||||||||||||||||||
| Marketable equity securities, non-current | $ | $ | $ | ( | $ | |||||||||||||||||||||
| As of December 31, 2019 | ||||||||||||||||||||||||||
| (In millions) | Amortized Cost | Gross Unrealized Gains | Gross Unrealized Losses | Fair Value | ||||||||||||||||||||||
| Corporate debt securities | ||||||||||||||||||||||||||
| Current | $ | $ | $ | $ | ||||||||||||||||||||||
| Non-current | ||||||||||||||||||||||||||
| Government securities | ||||||||||||||||||||||||||
| Current | ||||||||||||||||||||||||||
| Non-current | ( | |||||||||||||||||||||||||
| Mortgage and other asset backed securities | ||||||||||||||||||||||||||
| Current | ||||||||||||||||||||||||||
| Non-current | ( | |||||||||||||||||||||||||
| Total marketable debt securities | $ | $ | $ | ( | $ | |||||||||||||||||||||
| Marketable equity securities, non-current | $ | $ | $ | ( | $ | |||||||||||||||||||||
| As of December 31, 2020 | As of December 31, 2019 | |||||||||||||||||||||||||
| (In millions) | Estimated Fair Value | Amortized Cost | Estimated Fair Value | Amortized Cost | ||||||||||||||||||||||
| Due in one year or less | $ | $ | $ | $ | ||||||||||||||||||||||
| Due after one year through five years | ||||||||||||||||||||||||||
| Due after five years | ||||||||||||||||||||||||||
| Total marketable debt securities | $ | $ | $ | $ | ||||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Proceeds from maturities and sales | $ | $ | $ | |||||||||||||||||
| Realized gains | ||||||||||||||||||||
| Realized losses | ||||||||||||||||||||
| Notional Amount As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Euro | $ | $ | ||||||||||||
| British pound | ||||||||||||||
| Total foreign currency forward contracts | $ | $ | ||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||||||||||||||||||||||||||
Net Gains/(Losses) Reclassified from AOCI into Operating Income (in millions) | Net Gains/(Losses) Recognized in Operating Income (in millions) | |||||||||||||||||||||||||||||||||||||||||||
| Location | 2020 | 2019 | 2018 | Location | 2020 | 2019 | 2018 | |||||||||||||||||||||||||||||||||||||
| Revenues | $ | $ | $ | ( | Revenues | $ | ( | $ | $ | |||||||||||||||||||||||||||||||||||
| Operating expenses | ( | Operating expenses | ( | |||||||||||||||||||||||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net Gains/(Losses) Recognized in Other Comprehensive Income (Effective Portion) (in millions) | Net Gains/(Losses) Recognized in Other Comprehensive Income (Amounts Excluded from Effectiveness Testing) (in millions) | Net Gains/(Losses) Recognized in Net Income (Amounts Excluded from Effectiveness Testing) (in millions) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Location | 2020 | 2019 | 2018 | Location | 2020 | 2019 | 2018 | Location | 2020 | 2019 | 2018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gains (losses) on net investment hedge | $ | ( | $ | $ | ( | Gains (losses) on net investment hedge | $ | $ | $ | Other income (expense) | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| As of December 31, | ||||||||||||||||||||
| (In millions) | Balance Sheet Location | 2020 | 2019 | |||||||||||||||||
| Cash Flow Hedging Instruments: | ||||||||||||||||||||
| Asset derivative instruments | Other current assets | $ | $ | |||||||||||||||||
| Liability derivative instruments | Accrued expenses and other | |||||||||||||||||||
| Other long-term liabilities | ||||||||||||||||||||
| Net Investment Hedging Instruments: | ||||||||||||||||||||
| Asset derivative instruments | Other current assets | |||||||||||||||||||
| Liability derivative instruments | Accrued expenses and other | |||||||||||||||||||
| Fair Value Hedging Instruments | ||||||||||||||||||||
| Liability derivative instruments | Accrued expenses and other | |||||||||||||||||||
| Other Derivative Instruments: | ||||||||||||||||||||
| Asset derivative instruments | Other current assets | |||||||||||||||||||
| Liability derivative instruments | Accrued expenses and other | |||||||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Land | $ | $ | ||||||||||||
| Buildings | ||||||||||||||
| Leasehold improvements | ||||||||||||||
| Machinery and equipment | ||||||||||||||
| Computer software and hardware | ||||||||||||||
| Furniture and fixtures | ||||||||||||||
| Construction in progress | ||||||||||||||
| Total cost | ||||||||||||||
| Less: accumulated depreciation | ( | ( | ||||||||||||
| Total property, plant and equipment, net | $ | $ | ||||||||||||
| As of December 31, | |||||||||||||||||
| (In millions) | Balance sheet location | 2020 | 2019 | ||||||||||||||
| Assets: | |||||||||||||||||
| Operating lease assets | Operating lease assets | $ | $ | ||||||||||||||
| Liabilities | |||||||||||||||||
| Current operating lease liabilities | $ | $ | |||||||||||||||
| Non-current operating lease liabilities | Long-term operating lease liabilities | ||||||||||||||||
| Total operating lease liabilities | $ | $ | |||||||||||||||
| For the Years Ended December 31, | |||||||||||||||||
| (In millions) | Income Statement Location | 2020 | 2019 | ||||||||||||||
| Operating lease cost | Research and development | $ | $ | ||||||||||||||
| Selling, general and administrative | |||||||||||||||||
| Variable lease cost | Research and development | ||||||||||||||||
| Selling, general and administrative | |||||||||||||||||
| Sublease income | Selling, general and administrative | ( | ( | ||||||||||||||
| Other (income) expense, net | ( | ( | |||||||||||||||
| Net lease cost | $ | $ | |||||||||||||||
(In millions) | As of December 31, 2020 | |||||||
| 2021 | $ | |||||||
| 2022 | ||||||||
| 2023 | ||||||||
| 2024 | ||||||||
| 2025 | ||||||||
| Thereafter | ||||||||
| Total lease payments | $ | |||||||
| Less: interest | ||||||||
| Present value of operating lease liabilities | $ | |||||||
| As of December 31, | ||||||||||||||
| 2020 | 2019 | |||||||||||||
| Weighted average remaining lease term in years | ||||||||||||||
| Weighted average discount rate | % | % | ||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Cash paid for amounts included in the measurement of lease liabilities | $ | $ | ||||||||||||
| Operating lease assets obtained in exchange for lease obligations | ||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Current portion: | ||||||||||||||
2.900% Senior Notes due September 15, 2020(1) | $ | $ | ||||||||||||
| Current portion of notes payable | $ | $ | ||||||||||||
| Non-current portion: | ||||||||||||||
| 3.625% Senior Notes due September 15, 2022 | $ | $ | ||||||||||||
| 4.050% Senior Notes due September 15, 2025 | ||||||||||||||
| 2.250% Senior Notes due May 1, 2030 | ||||||||||||||
| 5.200% Senior Notes due September 15, 2045 | ||||||||||||||
| 3.150% Senior Notes due May 1, 2050 | ||||||||||||||
| Non-current portion of notes payable | $ | $ | ||||||||||||
| (In millions) | As of December 31, 2020 | |||||||
| 2021 | $ | |||||||
| 2022 | ||||||||
| 2023 | ||||||||
| 2024 | ||||||||
| 2025 | ||||||||
| 2026 and thereafter | ||||||||
| Total | $ | |||||||
| As of December 31, 2020 | As of December 31, 2019 | As of December 31, 2018 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| (In millions) | Authorized | Issued | Outstanding | Authorized | Issued | Outstanding | Authorized | Issued | Outstanding | ||||||||||||||||||||||||||||||||||||||||||||
| Common stock | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2020 | ||||||||||||||||||||||||||||||||||||||
| (In millions) | Unrealized Gains (Losses) on Securities Available for Sale, net of tax | Unrealized Gains (Losses) on Cash Flow Hedges, net of tax | Gains (Losses) on Net Investment Hedge, Net of Tax | Unfunded Status of Postretirement Benefit Plans, net of tax | Currency Translation Adjustments | Total | ||||||||||||||||||||||||||||||||
| Balance, December 31, 2019 | $ | $ | $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||||||||
| Other comprehensive income (loss) before reclassifications | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||
| Amounts reclassified from accumulated other comprehensive income (loss) | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Net current period other comprehensive income (loss) | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||
| Balance, December 31, 2020 | $ | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||||||
| December 31, 2019 | ||||||||||||||||||||||||||||||||||||||
| (In millions) | Unrealized Gains (Losses) on Securities Available for Sale, net of tax | Unrealized Gains (Losses) on Cash Flow Hedges, net of tax | Gains (Losses) on Net Investment Hedge, Net of Tax | Unfunded Status of Postretirement Benefit Plans, net of tax | Currency Translation Adjustments | Total | ||||||||||||||||||||||||||||||||
| Balance, December 31, 2018 | $ | ( | $ | $ | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||||||||
| Other comprehensive income (loss) before reclassifications | ( | |||||||||||||||||||||||||||||||||||||
| Amounts reclassified from accumulated other comprehensive income (loss) | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||
| Net current period other comprehensive income (loss) | ( | ( | ||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2019 | $ | $ | $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||||||||
| December 31, 2018 | ||||||||||||||||||||||||||||||||||||||
| (In millions) | Unrealized Gains (Losses) on Securities Available for Sale, net of tax | Unrealized Gains (Losses) on Cash Flow Hedges, net of tax | Gains (Losses) on Net Investment Hedge, Net of Tax | Unfunded Status of Postretirement Benefit Plans, net of tax | Currency Translation Adjustments | Total | ||||||||||||||||||||||||||||||||
| Balance, December 31, 2017 | $ | ( | $ | ( | $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||||||
| Amount reclassified, net of tax, upon adoption of ASU 2016-01 | ||||||||||||||||||||||||||||||||||||||
| Balance, January 1, 2018 | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||
| Other comprehensive income (loss) before reclassifications | ( | ( | ||||||||||||||||||||||||||||||||||||
| Amounts reclassified from accumulated other comprehensive income (loss) | ( | |||||||||||||||||||||||||||||||||||||
| Net current period other comprehensive income (loss) | ( | ( | ||||||||||||||||||||||||||||||||||||
| Balance, December 31, 2018 | $ | ( | $ | $ | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||||||||
| (In millions) | Income Statement Location | Amounts Reclassified from Accumulated Other Comprehensive Income | ||||||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||||||||
| 2020 | 2019 | 2018 | ||||||||||||||||||||||||
| Gains (losses) on securities available for sale | Other income (expense) | $ | ( | $ | $ | ( | ||||||||||||||||||||
| Income tax benefit (expense) | ( | |||||||||||||||||||||||||
| Gains (losses) on cash flow hedges | Revenues | ( | ||||||||||||||||||||||||
| Operating expenses | ( | |||||||||||||||||||||||||
| Other income (expense) | ||||||||||||||||||||||||||
| Income tax benefit (expense) | ( | ( | ||||||||||||||||||||||||
| Gains (losses) on net investment hedge | Other Income (expense) | |||||||||||||||||||||||||
| Total reclassifications, net of tax | $ | $ | $ | ( | ||||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Numerator: | ||||||||||||||||||||
| Net income attributable to Biogen Inc. | $ | $ | $ | |||||||||||||||||
| Denominator: | ||||||||||||||||||||
| Weighted average number of common shares outstanding | ||||||||||||||||||||
| Effect of dilutive securities: | ||||||||||||||||||||
| Time-vested restricted stock units | ||||||||||||||||||||
| Market stock units | ||||||||||||||||||||
| Performance stock units settled in stock | ||||||||||||||||||||
| Dilutive potential common shares | ||||||||||||||||||||
| Shares used in calculating diluted earnings per share | ||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Research and development | $ | $ | $ | |||||||||||||||||
| Selling, general and administrative | ||||||||||||||||||||
| Subtotal | ||||||||||||||||||||
| Capitalized share-based compensation costs | ( | ( | ( | |||||||||||||||||
| Share-based compensation expense included in total cost and expenses | ||||||||||||||||||||
| Income tax effect | ( | ( | ( | |||||||||||||||||
| Share-based compensation expense included in net income attributable to Biogen Inc. | $ | $ | $ | |||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Market stock units | $ | $ | $ | |||||||||||||||||
| Time-vested restricted stock units | ||||||||||||||||||||
| Cash settled performance units | ( | |||||||||||||||||||
| Performance units | ( | |||||||||||||||||||
| Performance stock units settled in stock | ||||||||||||||||||||
| Performance stock units settled in cash | ||||||||||||||||||||
| Employee stock purchase plan | ||||||||||||||||||||
| NST stock options | ||||||||||||||||||||
| Subtotal | ||||||||||||||||||||
| Capitalized share-based compensation costs | ( | ( | ( | |||||||||||||||||
| Share-based compensation expense included in total cost and expenses | $ | $ | $ | |||||||||||||||||
| Shares | Weighted Average Exercise Price | ||||||||||
| Outstanding at December 31, 2019 | $ | ||||||||||
| Granted | |||||||||||
| Exercised | ( | ||||||||||
| Cancelled | |||||||||||
| Outstanding at December 31, 2020 | $ | ||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Tax benefit realized for stock options | $ | $ | $ | |||||||||||||||||
| Cash received from the exercise of stock options | ||||||||||||||||||||
| December 31, 2020 | ||||||||||||||
| Shares | Weighted Average Grant Date Fair Value | |||||||||||||
| Unvested at December 31, 2019 | $ | |||||||||||||
Granted (1) | ||||||||||||||
| Vested | ( | |||||||||||||
| Forfeited | ( | |||||||||||||
| Unvested at December 31, 2020 | $ | |||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| 2020 | 2019 | 2018 | ||||||||||||||||||
| Expected dividend yield | ||||||||||||||||||||
| Range of expected stock price volatility | 37.8% - 44.1% | 31.2% - 33.6% | 27.5% - 32.4% | |||||||||||||||||
| Range of risk-free interest rates | 1.41% - 1.48% | 2.46% - 2.53% | 1.9% - 2.3% | |||||||||||||||||
| 30 calendar day average stock price on grant date | $257.83 - $325.40 | $228.59 - $331.18 | $279.47 - $346.76 | |||||||||||||||||
| Weighted-average per share grant date fair value | $ | $ | $ | |||||||||||||||||
| Shares | |||||
| Unvested at December 31, 2019 | |||||
| Granted | |||||
| Vested | ( | ||||
| Forfeited | |||||
| Unvested at December 31, 2020 | |||||
| Shares | |||||
| Unvested at December 31, 2019 | |||||
| Granted | |||||
| Vested | ( | ||||
| Forfeited | |||||
| Unvested at December 31, 2020 | |||||
| Shares | Weighted Average Grant Date Fair Value | ||||||||||
| Unvested at December 31, 2019 | $ | ||||||||||
Granted (1) | |||||||||||
| Vested | |||||||||||
| Forfeited | ( | ||||||||||
| Unvested at December 31, 2020 | $ | ||||||||||
| Shares | |||||
| Unvested at December 31, 2019 | |||||
Granted (1) | |||||
| Vested | ( | ||||
| Forfeited | ( | ||||
| Unvested at December 31, 2020 | |||||
| Shares | Weighted Average Grant Date Fair Value | ||||||||||
| Unvested at December 31, 2019 | $ | ||||||||||
Granted (1) | |||||||||||
| Vested | ( | ||||||||||
| Forfeited | ( | ||||||||||
| Unvested at December 31, 2020 | $ | ||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions, except share amounts) | 2020 | 2019 | 2018 | |||||||||||||||||
| Shares issued under the 2015 ESPP | ||||||||||||||||||||
| Cash received under the 2015 ESPP | $ | $ | $ | |||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Income before income taxes (benefit): | ||||||||||||||||||||
| Domestic | $ | $ | $ | |||||||||||||||||
| Foreign | ||||||||||||||||||||
| Total | $ | $ | $ | |||||||||||||||||
| Income tax expense (benefit): | ||||||||||||||||||||
| Current: | ||||||||||||||||||||
| Federal | $ | $ | $ | |||||||||||||||||
| State | ||||||||||||||||||||
| Foreign | ||||||||||||||||||||
| Total | ||||||||||||||||||||
| Deferred: | ||||||||||||||||||||
| Federal | $ | ( | $ | $ | ( | |||||||||||||||
| State | ( | ( | ( | |||||||||||||||||
| Foreign | ( | |||||||||||||||||||
| Total | ||||||||||||||||||||
| Total income tax expense | $ | $ | $ | |||||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Deferred tax assets: | ||||||||||||||
| Tax credits | $ | $ | ||||||||||||
| Inventory, other reserves and accruals | ||||||||||||||
| Intangibles, net | ||||||||||||||
| Net operating loss | ||||||||||||||
| Share-based compensation | ||||||||||||||
| Other | ||||||||||||||
| Valuation allowance | ( | ( | ||||||||||||
| Total deferred tax assets | $ | $ | ||||||||||||
| Deferred tax liabilities: | ||||||||||||||
| Purchased intangible assets | $ | ( | $ | ( | ||||||||||
| GILTI | ( | ( | ||||||||||||
| Tax credits | ( | ( | ||||||||||||
| Depreciation, amortization and other | ( | ( | ||||||||||||
| Total deferred tax liabilities | $ | ( | $ | ( | ||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| 2020 | 2019 | 2018 | ||||||||||||||||||
| Statutory rate | % | % | % | |||||||||||||||||
| State taxes | ||||||||||||||||||||
| Taxes on foreign earnings | ( | ( | ( | |||||||||||||||||
| Credits and net operating loss utilization | ( | ( | ( | |||||||||||||||||
| Purchased intangible assets | ||||||||||||||||||||
| Divestiture of Denmark manufacturing operations | ( | |||||||||||||||||||
| Internal reorganization of certain intellectual property rights | ( | |||||||||||||||||||
| TECFIDERA impairment | ||||||||||||||||||||
| GILTI | ||||||||||||||||||||
| U.S. tax reform | ||||||||||||||||||||
| Swiss tax reform | ( | |||||||||||||||||||
| Other | ( | |||||||||||||||||||
| Effective tax rate | % | % | % | |||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Beginning balance at January 1, | $ | $ | $ | |||||||||||||||||
| Additions based on tax positions related to the current period | ||||||||||||||||||||
| Additions for tax positions of prior periods | ||||||||||||||||||||
| Reductions for tax positions of prior periods | ( | ( | ( | |||||||||||||||||
| Statute expirations | ( | ( | ( | |||||||||||||||||
| Settlement refund (payment) | ( | |||||||||||||||||||
| Ending balance ar December 31, | $ | $ | $ | |||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Cash paid during the year for: | ||||||||||||||||||||
| Interest | $ | $ | $ | |||||||||||||||||
| Income taxes | ||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Interest income | $ | $ | $ | |||||||||||||||||
| Interest expense | ( | ( | ( | |||||||||||||||||
| Gain (loss) on investments, net | ||||||||||||||||||||
| Foreign exchange gains (losses), net | ( | ( | ( | |||||||||||||||||
| Other, net | ( | ( | ||||||||||||||||||
| Total other income (expense), net | $ | $ | $ | |||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Net gains (losses) recognized during the period on equity securities | $ | $ | $ | |||||||||||||||||
| Less: Net gains (losses) recognized on equity securities sold during the period and on capital distributions | ( | |||||||||||||||||||
| Unrealized gains (losses) recognized during the period on equity securities | $ | $ | $ | |||||||||||||||||
| As of December 31, | ||||||||||||||
| (In millions) | 2020 | 2019 | ||||||||||||
| Revenue-related reserves for discounts and allowances | $ | $ | ||||||||||||
| Collaboration expenses | ||||||||||||||
| Employee compensation and benefits | ||||||||||||||
| Royalties and licensing fees | ||||||||||||||
| Derivative liabilities | ||||||||||||||
| Current portion of contingent consideration obligations | ||||||||||||||
| Other | ||||||||||||||
| Total accrued expenses and other | $ | $ | ||||||||||||
| Until GAZYVA First Non-CLL FDA Approval | % | ||||
| After GAZYVA First Non-CLL FDA Approval until First GAZYVA Threshold Date | % | ||||
| After First GAZYVA Threshold Date until Second GAZYVA Threshold Date | % | ||||
| After Second GAZYVA Threshold Date | % | ||||
| Until First GAZYVA Threshold Date | % | ||||
| After First GAZYVA Threshold Date until Second GAZYVA Threshold Date | % | ||||
| After Second GAZYVA Threshold Date | % | ||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Biogen's share of pre-tax profits in the U.S. for RITUXAN and GAZYVA, including the reimbursement of selling and development expenses | $ | $ | $ | |||||||||||||||||
| Other revenues from anti-CD20 therapeutic programs | ||||||||||||||||||||
| Total revenues from anti-CD20 therapeutic programs | $ | $ | $ | |||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Total development expense incurred by the collaboration related to the advancement of BAN2401 and elenbecestat | $ | $ | $ | |||||||||||||||||
| Biogen's share of BAN2401 and elenbecestat development expense reflected in research and development expense in our consolidated statements of income | ||||||||||||||||||||
| Total sales and marketing expense incurred by the collaboration | ||||||||||||||||||||
| Biogen's share of BAN2401 and elenbecestat sales and marketing expense reflected in selling, general and administrative expense in our consolidated statements of income | ||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Total aducanumab development expense | $ | $ | $ | |||||||||||||||||
| Biogen's share of aducanumab development expense reflected in research and development expense in our consolidated statements of income | ||||||||||||||||||||
| Total aducanumab sales and marketing expense incurred by the collaboration | ||||||||||||||||||||
| Biogen's share of aducanumab sales and marketing expense reflected in selling, general and administrative expense in our consolidated statements of income | ||||||||||||||||||||
| Total aducanumab collaboration third party milestone expense | ||||||||||||||||||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | ||||||||||||||||||||
| For the Years Ended December 31, | ||||||||||||||||||||
| (In millions) | 2020 | 2019 | 2018 | |||||||||||||||||
| Total UCB development expense | $ | $ | $ | |||||||||||||||||
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | ||||||||||||||||||||
| December 31, 2020 | ||||||||||||||||||||||||||||||||
| (In millions) | U.S. | Europe | Asia | Other | Total | |||||||||||||||||||||||||||
| Product revenues from external customers | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | ||||||||||||||||||||||||||||||||
| Other revenues from external customers | ||||||||||||||||||||||||||||||||
| Long-lived assets | ||||||||||||||||||||||||||||||||
| December 31, 2019 | ||||||||||||||||||||||||||||||||
| (In millions) | U.S. | Europe | Asia | Other | Total | |||||||||||||||||||||||||||
| Product revenues from external customers | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | ||||||||||||||||||||||||||||||||
| Other revenues from external customers | ||||||||||||||||||||||||||||||||
| Long-lived assets | ||||||||||||||||||||||||||||||||
| December 31, 2018 | ||||||||||||||||||||||||||||||||
| (In millions) | U.S. | Europe | Asia | Other | Total | |||||||||||||||||||||||||||
| Product revenues from external customers | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | ||||||||||||||||||||||||||||||||
| Other revenues from external customers | ||||||||||||||||||||||||||||||||
| Long-lived assets | ||||||||||||||||||||||||||||||||
| (In millions, except per share amounts) | First Quarter | Second Quarter | Third Quarter | Fourth Quarter | Total Year | ||||||||||||||||||||||||
| 2020 | |||||||||||||||||||||||||||||
| Product revenues, net | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Other revenues | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Total revenues | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
Gross profit (1) | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Net income | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Net income attributable to Biogen Inc. | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Net income per share: | |||||||||||||||||||||||||||||
| Basic earnings per share attributable to Biogen Inc. | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Diluted earnings per share attributable to Biogen Inc. | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Weighted-average shares used in calculating: | |||||||||||||||||||||||||||||
| Basic earnings per share attributable to Biogen Inc. | |||||||||||||||||||||||||||||
| Diluted earnings per share attributable to Biogen Inc. | |||||||||||||||||||||||||||||
| (In millions, except per share amounts) | First Quarter | Second Quarter | Third Quarter | Fourth Quarter | Total Year | ||||||||||||||||||||||||
| 2019 | |||||||||||||||||||||||||||||
| Product revenues, net | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Other revenues | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Total revenues | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
Gross profit (1) | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Net income | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Net income attributable to Biogen Inc. | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Net income per share: | |||||||||||||||||||||||||||||
| Basic earnings per share attributable to Biogen Inc. | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Diluted earnings per share attributable to Biogen Inc. | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Weighted-average shares used in calculating: | |||||||||||||||||||||||||||||
| Basic earnings per share attributable to Biogen Inc. | |||||||||||||||||||||||||||||
| Diluted earnings per share attributable to Biogen Inc. | |||||||||||||||||||||||||||||
| SUBSIDIARY | STATE OR OTHER JURISDICTION OF INCORPORATION OR ORGANIZATION | |||||||
| Biogen Foundation Inc. | Massachusetts | |||||||
| Biogen MA Inc. | Massachusetts | |||||||
| Biogen Realty Corporation | Massachusetts | |||||||
| Biogen Realty Limited Partnership | Massachusetts | |||||||
| Biogen U.S. Corporation | Massachusetts | |||||||
| Biogen U.S. Limited Partnership | Massachusetts | |||||||
| Biogen (RTP) Realty LLC | Delaware | |||||||
| Biogen Chesapeake LLC | Delaware | |||||||
| Biogen Holding I LLC | Delaware | |||||||
| Biogen Holding II LLC | Delaware | |||||||
| Biogen Manufacturing Holding LLC | Delaware | |||||||
| Biogen New Ventures Inc. | Delaware | |||||||
| Biogen SRO Inc. | Delaware | |||||||
| Biogen Therapeutics Inc. | Delaware | |||||||
| Biogen U.S. Pacific LLC | Delaware | |||||||
| Biogen U.S. West Corporation | Delaware | |||||||
| Conforma Therapeutics Corporation | Delaware | |||||||
| Stromedix, Inc. | Delaware | |||||||
| Nightstar, Inc. | Delaware | |||||||
| Biogen (Argentina) SRL | Argentina | |||||||
| Biogen Australia PTY Ltd | Australia | |||||||
| Biogen Austria GmbH | Austria | |||||||
| Biogen Belgium N.V./S.A. | Belgium | |||||||
| Biogen International Holding Limited | Bermuda | |||||||
| Biogen (Bermuda) Technologies Ltd. | Bermuda | |||||||
| Biogen Brasil Produtos Farmaceuticos LTDA | Brazil | |||||||
| Biogen Canada Inc. | Canada | |||||||
| Biogen Chile SpA | Chile | |||||||
| Biogen Biotechnology (Shanghai) Co., Ltd | China | |||||||
| BIIB Colombia S.A.S. | Colombia | |||||||
| Biogen Pharma d.o.o. | Croatia | |||||||
| Biogen (Czech Republic) s.r.o. | Czech Republic | |||||||
| Biogen (Denmark) A/S | Denmark | |||||||
| Biogen Estonia OU | Estonia | |||||||
| Biogen Finland OY | Finland | |||||||
| Biogen France S.A.S. | France | |||||||
| Biogen GmbH | Germany | |||||||
| Biogen Hong Kong Limited | Hong Kong | |||||||
| Biogen Hungary KFT | Hungary | |||||||
| Biogen Idec Biotech India Pvt. Ltd. | India | |||||||
| Biogen Idec (Ireland) Ltd. | Ireland | |||||||
| Nightstar Europa Limited | Ireland | |||||||
| Biogen Italia S.R.L. | Italy | |||||||
| Biogen Japan Ltd. | Japan | |||||||
| Biogen Korea LLC | Korea | |||||||
| Biogen Latvia SIA | Latvia | |||||||
| Biogen Lithuania UAB | Lithuania | |||||||
| Biogen Luxembourg Holding S.a.r.l. | Luxembourg | |||||||
| Biogen Mexico S. de R.L. de C.V. | Mexico | |||||||
| Biogen Mexico Servicios S. de R.L. de C.V. | Mexico | |||||||
| Biogen NZ Biopharma Ltd. | New Zealand | |||||||
| Biogen Norway AS | Norway | |||||||
| Biogen Poland Sp. z.o.o. | Poland | |||||||
| Biogen Portugal Sociedade Farmaceutica, Unipessoal Lda. | Portugal | |||||||
| Biogen Slovakia s.r.o. | Slovak Republic | |||||||
| Biogen Pharma, farmacevtska in biotehnoloska druzba, d.o.o. | Slovenia | |||||||
| Biogen Spain, S.L. | Spain | |||||||
| Biogen Sweden AB | Sweden | |||||||
| Biogen International GmbH | Switzerland | |||||||
| Biogen International Neuroscience GmbH | Switzerland | |||||||
| Biogen Management Services GmbH | Switzerland | |||||||
| Biogen Swiss Investments GmbH | Switzerland | |||||||
| Biogen Swiss Manufacturing GmbH | Switzerland | |||||||
| Biogen Switzerland AG | Switzerland | |||||||
| Biogen Switzerland Holdings GmbH | Switzerland | |||||||
| Eidetica Biopharma GmbH | Switzerland | |||||||
| Biogen Taiwan Limited | Taiwan | |||||||
| Biogen B.V. | The Netherlands | |||||||
| Biogen Netherlands B.V. | The Netherlands | |||||||
| Biogen Idec Limited | United Kingdom | |||||||
| Biogen Idec Research Ltd. | United Kingdom | |||||||
| Convergence Pharmaceuticals Limited | United Kingdom | |||||||
| Convergence Pharmaceuticals Holdings Ltd. | United Kingdom | |||||||
| Old Convergence Pharmaceuticals Limited | United Kingdom | |||||||
| Silver Acquisition Co. Ltd. | United Kingdom | |||||||
| Nightstar Therapeutics Limited | United Kingdom | |||||||
| NightstaRx Limited | United Kingdom | |||||||
| Tungsten Bidco Limited | United Kingdom | |||||||
| Biogen Idec Uruguay SA | Uruguay | |||||||
| Date: February 3, 2021 | /s/ Michel Vounatsos | |||||||
| Michel Vounatsos | ||||||||
| Chief Executive Officer | ||||||||
| Date: February 3, 2021 | /s/ Michael R. McDonnell | |||||||
| Michael R. McDonnell | ||||||||
| Executive Vice President and | ||||||||
| Chief Financial Officer | ||||||||
| Date: February 3, 2021 | /s/ Michel Vounatsos | |||||||
| Michel Vounatsos | ||||||||
| Chief Executive Officer | ||||||||
| [principal executive officer] | ||||||||
| Date: February 3, 2021 | /s/ Michael R. McDonnell | |||||||
| Michael R. McDonnell | ||||||||
| Executive Vice President and | ||||||||
| Chief Financial Officer | ||||||||
| [principal financial officer] | ||||||||
Consolidated Statements of Comprehensive Income - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Statement of Comprehensive Income [Abstract] | |||
| Net income attributable to Biogen Inc. | $ 4,000.6 | $ 5,888.5 | $ 4,430.7 |
| Other comprehensive income: | |||
| Unrealized gains (losses) on securities available for sale, net of tax | (2.8) | 8.2 | (3.9) |
| Unrealized gains (losses) on cash flow hedges, net of tax | (186.8) | (26.9) | 139.2 |
| Gains (losses) on net investment hedges, net of tax | (33.6) | 21.6 | 3.5 |
| Unrealized gains (losses) on pension benefit obligation, net of tax | (33.5) | (1.5) | 5.5 |
| Currency translation adjustment | 92.9 | 103.8 | (67.8) |
| Total other comprehensive income (loss), net of tax | (163.8) | 105.2 | 76.5 |
| Comprehensive income attributable to Biogen Inc. | 3,836.8 | 5,993.7 | 4,507.2 |
| Comprehensive income (loss) attributable to noncontrolling interests, net of tax | 60.9 | (0.4) | 42.9 |
| Comprehensive income | $ 3,897.7 | $ 5,993.3 | $ 4,550.1 |
Consolidated Balance Sheets (Parenthetical) - $ / shares shares in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Statement of Financial Position [Abstract] | ||
| Preferred stock, par value (in dollars per share) | $ 0.001 | $ 0.001 |
| Common stock, par value (in dollars per share) | $ 0.0005 | $ 0.0005 |
| Treasury stock at cost, shares (in shares) | 23.8 | 23.8 |
Consolidated Statements of Equity - USD ($) shares in Millions, $ in Millions |
Total |
Cumulative Effect, Period of Adoption, Adjustment |
Preferred stock |
Common stock |
Additional paid-in capital |
Accumulated other comprehensive loss |
Accumulated other comprehensive loss
Cumulative Effect, Period of Adoption, Adjustment
|
Retained earnings |
Retained earnings
Cumulative Effect, Period of Adoption, Adjustment
|
Treasury stock |
Total Biogen Inc. shareholders’ equity |
Total Biogen Inc. shareholders’ equity
Cumulative Effect, Period of Adoption, Adjustment
|
Noncontrolling interests |
2020 Share Repurchase Program |
2020 Share Repurchase Program
Common stock
|
2020 Share Repurchase Program
Additional paid-in capital
|
2020 Share Repurchase Program
Retained earnings
|
2020 Share Repurchase Program
Treasury stock
|
2020 Share Repurchase Program
Total Biogen Inc. shareholders’ equity
|
December 2019 Share Repurchase Program |
December 2019 Share Repurchase Program
Common stock
|
December 2019 Share Repurchase Program
Additional paid-in capital
|
December 2019 Share Repurchase Program
Retained earnings
|
December 2019 Share Repurchase Program
Treasury stock
|
December 2019 Share Repurchase Program
Total Biogen Inc. shareholders’ equity
|
March 2019 Share Repurchase Program |
March 2019 Share Repurchase Program
Common stock
|
March 2019 Share Repurchase Program
Additional paid-in capital
|
March 2019 Share Repurchase Program
Retained earnings
|
March 2019 Share Repurchase Program
Treasury stock
|
March 2019 Share Repurchase Program
Total Biogen Inc. shareholders’ equity
|
2018 Share Repurchase Program |
2018 Share Repurchase Program
Common stock
|
2018 Share Repurchase Program
Additional paid-in capital
|
2018 Share Repurchase Program
Retained earnings
|
2018 Share Repurchase Program
Treasury stock
|
2018 Share Repurchase Program
Total Biogen Inc. shareholders’ equity
|
2016 Share Repurchase Program |
2016 Share Repurchase Program
Common stock
|
2016 Share Repurchase Program
Additional paid-in capital
|
2016 Share Repurchase Program
Retained earnings
|
2016 Share Repurchase Program
Treasury stock
|
2016 Share Repurchase Program
Total Biogen Inc. shareholders’ equity
|
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beginning balance (in shares) at Dec. 31, 2017 | 0.0 | 235.3 | 23.8 | ||||||||||||||||||||||||||||||||||||||||
| Beginning balance at Dec. 31, 2017 | $ 12,598.1 | $ 106.1 | $ 0.0 | $ 0.1 | $ 97.8 | $ (318.4) | $ 1.5 | $ 15,810.4 | $ 104.6 | $ (2,977.1) | $ 12,612.8 | $ 106.1 | $ (14.7) | ||||||||||||||||||||||||||||||
| Increase (Decrease) in Stockholders' Equity [Roll Forward] | |||||||||||||||||||||||||||||||||||||||||||
| Net income | 4,474.0 | 4,430.7 | 4,430.7 | 43.3 | |||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | 76.1 | 76.5 | 76.5 | (0.4) | |||||||||||||||||||||||||||||||||||||||
| Capital contribution from noncontrolling interest | 13.8 | 0.0 | 13.8 | ||||||||||||||||||||||||||||||||||||||||
| Distribution to noncontrolling interests | (50.0) | 0.0 | (50.0) | ||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the share repurchase program, at cost (in shares) | (4.3) | (4.3) | (10.5) | (10.5) | |||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the share repurchase program, at cost | $ (1,352.6) | $ (1,352.6) | $ (1,352.6) | $ (3,000.0) | $ (3,000.0) | $ (3,000.0) | |||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the share repurchase program, at cost (in shares) | 4.3 | 4.3 | 10.5 | 10.5 | |||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the share repurchase program, at cost | $ 0.0 | $ 0.0 | $ (92.8) | $ (1,259.8) | $ 1,352.6 | 0.0 | $ 0.0 | $ 0.0 | $ (171.1) | $ (2,828.9) | $ 3,000.0 | $ 0.0 | |||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans (in shares) | 0.2 | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans | 41.2 | $ 0.0 | 41.2 | 41.2 | |||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan (in shares) | 0.3 | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan | (43.8) | $ 0.0 | (43.8) | (43.8) | |||||||||||||||||||||||||||||||||||||||
| Compensation expense related to share-based payments | 168.7 | 168.7 | 168.7 | ||||||||||||||||||||||||||||||||||||||||
| Ending balance (in shares) at Dec. 31, 2018 | 0.0 | 221.0 | 23.8 | ||||||||||||||||||||||||||||||||||||||||
| Ending balance at Dec. 31, 2018 | 13,031.6 | $ 0.0 | $ 0.1 | 0.0 | (240.4) | 16,257.0 | $ (2,977.1) | 13,039.6 | (8.0) | ||||||||||||||||||||||||||||||||||
| Increase (Decrease) in Stockholders' Equity [Roll Forward] | |||||||||||||||||||||||||||||||||||||||||||
| Net income | 5,888.5 | 5,888.5 | 5,888.5 | ||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | 104.8 | 105.2 | 105.2 | (0.4) | |||||||||||||||||||||||||||||||||||||||
| Capital contribution from noncontrolling interest | 4.3 | 0.0 | 4.3 | ||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the share repurchase program, at cost (in shares) | (14.7) | (14.7) | (8.9) | (8.9) | |||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the share repurchase program, at cost | $ (3,720.9) | $ (3,720.9) | $ (3,720.9) | $ (2,147.4) | $ (2,147.4) | (2,147.4) | |||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the share repurchase program, at cost (in shares) | 14.7 | 14.7 | 8.9 | 8.9 | |||||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the share repurchase program, at cost | $ 0.0 | $ 0.0 | $ (121.5) | $ (3,599.4) | $ 3,720.9 | 0.0 | $ 0.0 | $ 0.0 | $ (110.5) | $ (2,036.9) | $ 2,147.4 | $ 0.0 | |||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans (in shares) | 0.2 | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans | 40.8 | $ 0.0 | 40.8 | 40.8 | |||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan (in shares) | 0.4 | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan | (53.8) | $ 0.0 | (53.8) | (53.8) | |||||||||||||||||||||||||||||||||||||||
| Compensation expense related to share-based payments | 191.2 | 191.2 | 191.2 | ||||||||||||||||||||||||||||||||||||||||
| Ending balance (in shares) at Dec. 31, 2019 | 0.0 | 198.0 | 23.8 | ||||||||||||||||||||||||||||||||||||||||
| Ending balance at Dec. 31, 2019 | 13,339.1 | $ 0.0 | $ 0.1 | 0.0 | (135.2) | 16,455.4 | $ (2,977.1) | 13,343.2 | (4.1) | ||||||||||||||||||||||||||||||||||
| Increase (Decrease) in Stockholders' Equity [Roll Forward] | |||||||||||||||||||||||||||||||||||||||||||
| Net income | 4,060.5 | 4,000.6 | 4,000.6 | 59.9 | |||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | (162.8) | (163.8) | (163.8) | 1.0 | |||||||||||||||||||||||||||||||||||||||
| Capital contribution from noncontrolling interest | 4.0 | 0.0 | 4.0 | ||||||||||||||||||||||||||||||||||||||||
| Distribution to noncontrolling interests | (75.0) | (75.0) | |||||||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the share repurchase program, at cost (in shares) | (1.6) | (1.6) | (16.7) | (16.7) | (4.1) | (4.1) | |||||||||||||||||||||||||||||||||||||
| Repurchase of common stock pursuant to the share repurchase program, at cost | $ (400.0) | $ (400.0) | $ (400.0) | $ (5,000.0) | $ (5,000.0) | $ (5,000.0) | $ (1,279.1) | $ (1,279.1) | (1,279.1) | ||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the share repurchase program, at cost (in shares) | 1.6 | 1.6 | 16.7 | 16.7 | 4.1 | 4.1 | |||||||||||||||||||||||||||||||||||||
| Retirement of common stock pursuant to the share repurchase program, at cost | $ 0.0 | $ (60.8) | $ (339.2) | $ 400.0 | $ 0.0 | $ (121.3) | $ (4,878.7) | $ 5,000.0 | $ 0.0 | $ 0.0 | $ (71.0) | $ (1,208.1) | $ 1,279.1 | $ 0.0 | |||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans (in shares) | 0.2 | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock option and stock purchase plans | 49.3 | 49.3 | 0.0 | 49.3 | |||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan (in shares) | 0.4 | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock under stock award plan | (53.7) | 0.0 | (53.7) | (53.7) | |||||||||||||||||||||||||||||||||||||||
| Compensation expense related to share-based payments | 204.5 | 204.5 | 204.5 | ||||||||||||||||||||||||||||||||||||||||
| Other | (0.7) | (0.7) | (0.7) | ||||||||||||||||||||||||||||||||||||||||
| Ending balance (in shares) at Dec. 31, 2020 | 0.0 | 176.2 | 23.8 | ||||||||||||||||||||||||||||||||||||||||
| Ending balance at Dec. 31, 2020 | $ 10,686.1 | $ 0.0 | $ 0.1 | $ 0.0 | $ (299.0) | $ 13,976.3 | $ (2,977.1) | $ 10,700.3 | $ (14.2) |
Summary of Significant Accounting Policies |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Accounting Policies [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of Significant Accounting Policies | Summary of Significant Accounting Policies References in these notes to "Biogen," the "company," "we," "us" and "our" refer to Biogen Inc. and its consolidated subsidiaries. Business Overview Biogen is a global biopharmaceutical company focused on discovering, developing and delivering worldwide innovative therapies for people living with serious neurological and neurodegenerative diseases as well as related therapeutic adjacencies. Our core growth areas include multiple sclerosis (MS) and neuroimmunology; Alzheimer’s disease and dementia; neuromuscular disorders, including spinal muscular atrophy (SMA) and amyotrophic lateral sclerosis (ALS); movement disorders, including Parkinson's disease; ophthalmology; and neuropsychiatry. We are also focused on discovering, developing and delivering worldwide innovative therapies in our emerging growth areas of immunology; acute neurology; and neuropathic pain. In addition, we commercialize biosimilars of advanced biologics. We support our drug discovery and development efforts through the commitment of significant resources to discovery, research and development programs and business development opportunities. Our marketed products include TECFIDERA, VUMERITY, AVONEX, PLEGRIDY, TYSABRI and FAMPYRA for the treatment of MS; SPINRAZA for the treatment of SMA; and FUMADERM for the treatment of severe plaque psoriasis. We have certain business and financial rights with respect to RITUXAN for the treatment of non-Hodgkin's lymphoma, chronic lymphocytic leukemia (CLL) and other conditions; RITUXAN HYCELA for the treatment of non-Hodgkin's lymphoma and CLL; GAZYVA for the treatment of CLL and follicular lymphoma; OCREVUS for the treatment of primary progressive MS (PPMS) and relapsing MS (RMS); and other potential anti-CD20 therapies pursuant to our collaboration arrangements with Genentech, Inc. (Genentech), a wholly-owned member of the Roche Group. For additional information on our collaboration arrangements with Genentech, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Our innovative drug development and commercialization activities are complemented by our biosimilar business that expands access to medicines and reduces the cost burden for healthcare systems. Through our agreements with Samsung Bioepis Co., Ltd. (Samsung Bioepis), our joint venture with Samsung BioLogics Co., Ltd. (Samsung BioLogics), we market and sell BENEPALI, an etanercept biosimilar referencing ENBREL, IMRALDI, an adalimumab biosimilar referencing HUMIRA, and FLIXABI, an infliximab biosimilar referencing REMICADE, in certain countries in Europe and have an option to acquire exclusive rights to commercialize these products in China. Additionally, we have exclusive rights to commercialize two potential ophthalmology biosimilar products, SB11, a proposed ranibizumab biosimilar referencing LUCENTIS, and SB15, a proposed aflibercept biosimilar referencing EYLEA, in major markets worldwide, including the United States (U.S.), Canada, Europe, Japan and Australia. For additional information on our collaboration arrangements with Samsung Bioepis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Consolidation Our consolidated financial statements reflect our financial statements, those of our wholly-owned subsidiaries and those of certain variable interest entities where we are the primary beneficiary. For consolidated entities where we own or are exposed to less than 100.0% of the economics, we record net income (loss) attributable to noncontrolling interests in our consolidated statements of income equal to the percentage of the economic or ownership interest retained in such entities by the respective noncontrolling parties. Intercompany balances and transactions are eliminated in consolidation. In determining whether we are the primary beneficiary of a variable interest entity, we apply a qualitative approach that determines whether we have both (1) the power to direct the economically significant activities of the entity and (2) the obligation to absorb losses of, or the right to receive benefits from, the entity that could potentially be significant to that entity. These considerations impact the way we account for our existing collaborative relationships and other arrangements. We continuously assess whether we are the primary beneficiary of a variable interest entity as changes to existing relationships or future transactions may result in us consolidating or deconsolidating one or more of our collaborators or partners. Use of Estimates The preparation of our consolidated financial statements requires us to make estimates, judgments and assumptions that may affect the reported amounts of assets, liabilities, equity, revenues and expenses and related disclosure of contingent assets and liabilities. On an ongoing basis we evaluate our estimates, judgments and methodologies. We base our estimates on historical experience and on various other assumptions that we believe are reasonable, the results of which form the basis for making judgments about the carrying values of assets, liabilities and equity and the amount of revenues and expenses. Actual results may differ from these estimates. The length of time and full extent to which the COVID-19 pandemic directly or indirectly impacts our business, results of operations and financial condition, including sales, expenses, reserves and allowances, manufacturing, clinical trials, research and development costs and employee-related amounts, depends on future developments that are highly uncertain, subject to change and are difficult to predict, including as a result of new information that may emerge concerning COVID-19 and the actions taken to contain or treat COVID-19 as well as the economic impact on local, regional, national and international customers and markets. We have made estimates of the impact of COVID-19 within our condensed consolidated financial statements and there may be changes to those estimates in future periods. Revenue Recognition In May 2014 the Financial Accounting Standards Board (FASB) issued Accounting Standards Update (ASU) No. 2014-09, Revenue from Contracts with Customers (Topic 606), which supersedes all existing revenue recognition requirements, including most industry specific guidance. This standard requires a company to recognize revenues when it transfers goods or services to customers in an amount that reflects the consideration that the company expects to receive for those goods or services. This standard became effective for us on January 1, 2018, and was adopted using the modified retrospective method. The adoption of this standard as of January 1, 2018, did not change our revenue recognition. We recognize revenues when our customer obtains control of promised goods or services, in an amount that reflects the consideration which we expect to receive in exchange for those goods or services. We recognize revenues following the five-step model prescribed under the FASB Accounting Standards Codification (ASC) 606, Revenue from Contracts with Customers: (i) identify contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenues when (or as) we satisfy the performance obligation. Product Revenues In the U.S., we sell our products primarily to wholesale distributors and specialty pharmacy providers. In other countries, we sell our products primarily to wholesale distributors, hospitals, pharmacies and other third-party distribution partners. These customers subsequently resell our products to health care providers and patients. In addition, we enter into arrangements with health care providers and payors that provide for government-mandated or privately-negotiated discounts and allowances related to our products. Product revenues are recognized when the customer obtains control of our product, which occurs at a point in time, typically upon delivery to the customer. We expense incremental costs of obtaining a contract as and when incurred if the expected amortization period of the asset that we would have recognized is one year or less or the amount is immaterial. Reserves for Discounts and Allowances Product revenues are recorded net of reserves established for applicable discounts and allowances that are offered within contracts with our customers, health care providers or payors, including those associated with the implementation of pricing actions in certain of the international markets in which we operate. Our process for estimating reserves established for these variable consideration components do not differ materially from our historical practices. Product revenue reserves, which are classified as a reduction in product revenues, are generally characterized in the following categories: discounts, contractual adjustments and returns. These reserves are based on estimates of the amounts earned or to be claimed on the related sales and are classified as reductions of accounts receivable (if the amount is payable to our customer) or a liability (if the amount is payable to a party other than our customer). Our estimates of reserves established for variable consideration are calculated based upon a consistent application of our methodology utilizing the expected value method. These estimates reflect our historical experience, current contractual and statutory requirements, specific known market events and trends, industry data and forecasted customer buying and payment patterns. The transaction price, which includes variable consideration reflecting the impact of discounts and allowances, may be subject to constraint and is included in the net sales price only to the extent that it is probable that a significant reversal of the amount of the cumulative revenues recognized will not occur in a future period. Actual amounts may ultimately differ from our estimates. If actual results vary, we adjust these estimates, which could have an effect on earnings in the period of adjustment. Discounts include trade term discounts and wholesaler incentives. Trade term discounts and wholesaler incentives primarily relate to estimated obligations for credits to be granted to wholesalers for remitting payment on their purchases within established incentive periods and credits to be granted to wholesalers for compliance with various contractually-defined inventory management practices, respectively. We determine these reserves based on our historical experience, including the timing of customer payments. Contractual adjustments primarily relate to Medicaid and managed care rebates, pharmacy rebates, co-payment (copay) assistance, Veterans Administration (VA) and Public Health Service (PHS) discounts, specialty pharmacy program fees and other governmental rebates or applicable allowances. •Medicaid rebates relate to our estimated obligations to states under established reimbursement arrangements. Rebate accruals are recorded in the same period the related revenue is recognized, resulting in a reduction of product revenue and the establishment of a liability which is included in other current liabilities. Our liability for Medicaid rebates consists of estimates for claims that a state will make for the current quarter, claims for prior quarters that have been estimated for which an invoice has not been received, invoices received for claims from the prior quarters that have not been paid and an estimate of potential claims that will be made for inventory that exists in the distribution channel at period end. •Governmental rebates or chargebacks, including VA and PHS discounts, represent our estimated obligations resulting from contractual commitments to sell products to qualified healthcare providers at prices lower than the list prices we charge to wholesalers which provide those products. The wholesaler charges us for the difference between what the wholesaler pays for the products and the ultimate selling price to the qualified healthcare providers. Rebate and chargeback reserves are established in the same period as the related revenue is recognized, resulting in a reduction in product revenue and accounts receivable. Chargeback amounts are generally determined at the time of resale to the qualified healthcare provider from the wholesaler, and we generally issue credits for such amounts within a few weeks of the wholesaler notifying us about the resale. Our reserves for VA, PHS and chargebacks consist of amounts that we expect to issue for inventory that exists at the wholesalers that we expect will be sold to qualified healthcare providers and chargebacks that wholesalers have claimed for which we have not issued a credit. •Managed care rebates represent our estimated obligations to third parties, primarily pharmacy benefit managers. Rebate accruals are recorded in the same period the related revenue is recognized, resulting in a reduction of product revenue and the establishment of a liability which is included in accrued expenses and other current liabilities. These rebates result from performance-based goals, formulary position and price increase limit allowances (price protection). The calculation of the accrual for these rebates is based on an estimate of the coverage patterns and the resulting applicable contractual rebate rate(s) to be earned over a contractual period. •Copay assistance represents financial assistance to qualified patients, assisting them with prescription drug co-payments required by insurance. The calculation of the accrual for copay is based on an estimate of claims and the cost per claim that we expect to receive associated with inventory that exists in the distribution channel at period end. •Pharmacy rebates represent our estimated obligations resulting from contractual commitments to sell products to specific pharmacies. Rebate accruals are recorded in the same period the related revenue is recognized, resulting in a reduction of product revenue and the establishment of a liability which is included in accrued expenses and other current liabilities. These rebates result from contracted discounts on product purchased or product dispensed. The calculation of the accrual for these rebates is based on an estimate of the pharmacy’s buying or dispensing patterns and the resulting applicable contractual rebate rate(s) to be earned over the contractual period. •Other governmental rebates, non-U.S. pharmaceutical taxes or applicable allowances primarily relate to mandatory rebates and discounts in international markets where government-sponsored healthcare systems are the primary payors for healthcare. Product return reserves are established for returns expected to be made by wholesalers and are recorded in the period the related revenue is recognized, resulting in a reduction to product revenues. In accordance with contractual terms, wholesalers are permitted to return product for reasons such as damaged or expired product. The majority of wholesaler returns are due to product expiration. Expired product return reserves are estimated through a comparison of historical return data to their related sales on a production lot basis. Historical rates of return are determined for each product and are adjusted for known or expected changes in the marketplace specific to each product. In addition to discounts, rebates and product returns, we also maintain certain customer service contracts with distributors and other customers in the distribution channel that provide us with inventory management, data and distribution services, which are generally reflected as a reduction of revenues. To the extent we can demonstrate a separable benefit and fair value for these services we classify these payments in selling, general and administrative expenses. Revenues from Anti-CD20 Therapeutic Programs Our collaboration with Genentech is within the scope of ASC 808, Collaborative Agreements, which provides guidance on the presentation and disclosure of collaborative arrangements. For purposes of this footnote, we refer to RITUXAN and RITUXAN HYCELA collectively as RITUXAN. Our share of the pre-tax co-promotion profits on RITUXAN and GAZYVA and royalty revenues on the sale of OCREVUS resulted from an exchange of a license. As we do not have future performance obligations under the license or collaboration agreement, revenues are recognized as the underlying sales occur. Revenues from anti-CD20 therapeutic programs consist of: (i) our share of pre-tax profits and losses in the U.S. for RITUXAN and GAZYVA; and (ii) other revenues from anti-CD20 therapeutic programs, which primarily consist of our share of pre-tax co-promotion profits on RITUXAN in Canada and royalty revenues on sales of OCREVUS. Pre-tax co-promotion profits on RITUXAN and GAZYVA are calculated and paid to us by Genentech and the Roche Group. Pre-tax co-promotion profits consist of net sales to third-party customers less applicable costs to manufacture, third-party royalty expenses, distribution, selling and marketing expenses and joint development expenses incurred by Genentech and the Roche Group. Our share of the pre-tax profits on RITUXAN and GAZYVA include estimates that are based on information received from Genentech and the Roche Group. These estimates are subject to change and actual results may differ. We recognize royalty revenues on sales of OCREVUS based on our estimates from third party and market research data of OCREVUS sales occurring during the corresponding period. Differences between actual and estimated royalty revenues will be adjusted for in the period in which they become known, which is generally expected to be the following quarter. For additional information on our relationship with Genentech, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Other Revenues Royalty Revenues We recognize royalty revenues related to sales by our licensees of products covered under patents that we own. Collaborative and Other Relationships We have a number of significant collaborative and other third-party relationships for revenues and for the development, regulatory approval, commercialization and marketing of certain of our products and product candidates. Where we are the principal on sales transactions with third parties, we recognize revenues, cost of sales and operating expenses on a gross basis in their respective lines in our consolidated statements of income. Where we are not the principal on sales transactions with third parties, we record our share of the revenues, cost of sales and operating expenses on a net basis in collaborative and other relationships included in other revenues in our consolidated statements of income. Our development and commercialization arrangements with Genentech and Samsung Bioepis represent collaborative arrangements as each party is an active participant in one or more joint operating activities and is exposed to significant risks and rewards of these arrangements. These arrangements resulted from an exchange of a license and utilize the sales and usage based royalty exception. Therefore, revenues relating to royalties or profit-sharing amounts received are recognized as the underlying sales occur. For additional information on our collaboration arrangements with Genentech and Samsung Bioepis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Other Corporate Revenues We record other corporate revenues primarily from amounts earned under contract manufacturing agreements. Revenues under contract manufacturing agreements are recognized when the customer obtains control of the product, which may occur at a point in time or over time depending on the terms and conditions of the agreement. Fair Value Measurements We have certain financial assets and liabilities recorded at fair value which have been classified as Level 1, 2 or 3 within the fair value hierarchy as described in the accounting standards for fair value measurements. •Level 1 — Fair values are determined utilizing quoted prices (unadjusted) in active markets for identical assets or liabilities that we have the ability to access; •Level 2 — Fair values are determined by utilizing quoted prices for identical or similar assets and liabilities in active markets or other market observable inputs such as interest rates, yield curves, foreign currency spot rates and option pricing valuation models; and •Level 3 — Prices or valuations that require inputs that are both significant to the fair value measurement and unobservable. The majority of our financial assets have been classified as Level 2. Our financial assets (which include our cash equivalents, marketable debt securities and certain of our marketable equity securities, derivative contracts and plan assets for deferred compensation) have been initially valued at the transaction price and subsequently valued, at the end of each reporting period, utilizing third-party pricing services or option pricing valuation models. The pricing services utilize industry standard valuation models, including both income and market-based approaches and observable market inputs to determine value. These observable market inputs include reportable trades, benchmark yields, credit spreads, broker/dealer quotes, bids, offers, current spot rates and other industry and economic events. We validate the prices provided by our third-party pricing services by understanding the models used, obtaining market values from other pricing sources and analyzing pricing data in certain instances. The option pricing valuation models use assumptions within the model, including the term, stock price volatility, constant maturity risk-free interest rate and dividend yield. After completing our validation procedures, we did not adjust or override any fair value measurements provided by our pricing services as of December 31, 2020 and 2019. Other Assets and Liabilities The carrying amounts reflected in our consolidated balance sheets for current accounts receivable, due from anti-CD20 therapeutic programs, other current assets, accounts payable and accrued expenses and other, approximate fair value due to their short-term maturities. Cash and Cash Equivalents We consider only those investments that are highly liquid, readily convertible to cash and that mature within three months from date of purchase to be cash equivalents. As of December 31, 2020 and 2019, cash equivalents were comprised of money market funds, commercial paper, overnight reverse repurchase agreements and other debt securities with maturities less than 90 days from the date of purchase. Accounts Receivable The majority of our accounts receivable arise from product sales and primarily represent amounts due from our wholesale and other third-party distributors, public hospitals, pharmacies and other government entities and have standard payment terms that generally require payment within 30 to 90 days. We do not adjust our receivables for the effects of a significant financing component at contract inception if we expect to collect the receivables in one year or less from the time of sale. In countries where we have experienced a pattern of payments extending beyond our contractual payment term and we expect to collect receivables greater than one year from the time of sale, we have assessed whether the customer has a significant financing component and discounted our receivables and reduced related revenues over the period of time that we estimate those amounts will be paid using the country’s market-based borrowing rate for such period. The related receivables are classified at the time of sale as non-current assets. We accrete interest income on these receivables, which is recorded as a component of other income (expense), net in our consolidated statements of income. We provide reserves against accounts receivable for estimated losses that may result from a customer's inability to pay. Amounts determined to be uncollectible are charged or written-off against the reserve. Concentration of Credit Risk Financial instruments that potentially subject us to concentrations of credit risk include cash and cash equivalents, investments, derivatives and accounts receivable. We attempt to minimize the risks related to cash and cash equivalents and investments by investing in a broad and diverse range of financial instruments as previously defined by us. We have established guidelines related to credit ratings and maturities intended to safeguard principal balances and maintain liquidity. Our investment portfolio is maintained in accordance with our investment policy, which defines allowable investments, specifies credit quality standards and limits the credit exposure of any single issuer. We minimize credit risk resulting from derivative instruments by choosing only highly rated financial institutions as counterparties. Concentrations of credit risk with respect to receivables, which are typically unsecured, are somewhat mitigated due to the wide variety of customers and markets using our products, as well as their dispersion across many different geographic areas. We monitor the financial performance and creditworthiness of our customers so that we can properly assess and respond to changes in their credit profile. We continue to monitor these conditions and assess their possible impact on our business. Marketable Securities and Other Investments Marketable Debt Securities Available-for-sale marketable debt securities are recorded at fair market value and unrealized gains and losses are included in accumulated other comprehensive income (loss) in equity, net of related tax effects, unless the security has experienced a credit loss, we have determined that we have the intent to sell the security or we have determined that it is more likely than not that we will have to sell the security before its expected recovery. Realized gains and losses are reported in other income (expense), net on a specific identification basis. Marketable Equity Securities and Venture Capital Funds Our marketable equity securities are recorded at fair market value and, beginning January 1, 2018, unrealized gains and losses are included in other income (expense), net in our consolidated statements of income. Prior to January 1, 2018, unrealized gains and losses were included in accumulated other comprehensive income (loss) in equity, net of related tax effects. Our marketable equity securities represent investments in publicly traded equity securities and are included in investments and other assets in our consolidated balance sheets. Our investments in venture capital funds are recorded at net asset value, which approximates fair value, and, beginning January 1, 2018, unrealized gains and losses are included in other income (expense), net in our consolidated statements of income. Prior to January 1, 2018, these investments were accounted for under the cost method of accounting. The underlying investments of the venture capital funds in which we invest are in equity securities of certain biotechnology companies and are included in investments and other assets in our consolidated balance sheets. Non-Marketable Equity Securities We also invest in equity securities of companies whose securities are not publicly traded and where fair value is not readily available. These investments are recorded using either the equity method of accounting or the cost minus impairment adjusted for changes in observable prices, depending on our ownership percentage and other factors that suggest we have significant influence. We monitor these investments to evaluate whether any increase or decline in their value has occurred, based on the implied value of recent company financings, public market prices of comparable companies and general market conditions. These investments are included in investments and other assets in our consolidated balance sheets. Evaluating Marketable Debt Securities for Other-than-Temporary Impairments We conduct periodic reviews to identify and evaluate each investment that has an unrealized loss, in accordance with the meaning of other-than-temporary impairment. An unrealized loss exists when the current fair value of an individual security is less than its amortized cost basis. Unrealized losses on available-for-sale debt securities that are determined to be temporary, and not related to credit loss, are recorded, net of tax, in accumulated other comprehensive income. For available-for-sale debt securities with unrealized losses, management performs an analysis to assess whether we intend to sell or whether we would more likely than not be required to sell the security before the expected recovery of the amortized cost basis. Where we intend to sell a security, or may be required to do so, the security’s decline in fair value is deemed to be other-than-temporary and the full amount of the unrealized loss is reflected in earnings as an impairment loss. Regardless of our intent to sell a security, we perform additional analysis on all securities with unrealized losses to evaluate losses associated with the creditworthiness of the security. Credit losses are identified where we do not expect to receive cash flows sufficient to recover the amortized cost basis of a security. Equity Method of Accounting In circumstances where we have the ability to exercise significant influence over the operating and financial policies of a company in which we have an investment, we utilize the equity method of accounting for recording investment activity. In assessing whether we exercise significant influence, we consider the nature and magnitude of our investment, the voting and protective rights we hold, any participation in the governance of the other company and other relevant factors such as the presence of a collaborative or other business relationship. Under the equity method of accounting, we record in our consolidated statements of income our share of income or loss of the other company. If our share of losses exceeds the carrying value of our investment, we will suspend recognizing additional losses and will continue to do so unless we commit to providing additional funding. Inventory Inventories are stated at the lower of cost or net realizable value with cost based on the first-in, first-out method. We classify our inventory costs as long-term when we expect to utilize the inventory beyond our normal operating cycle and include these costs in investments and other assets in our consolidated balance sheets. Inventory that can be used in either the production of clinical or commercial products is expensed as research and development costs when identified for use in a clinical manufacturing campaign. Capitalization of Inventory Costs We capitalize inventory costs associated with our products prior to regulatory approval, when, based on management’s judgment, future commercialization is considered probable and the future economic benefit is expected to be realized. We consider numerous attributes in evaluating whether the costs to manufacture a particular product should be capitalized as an asset. We assess the regulatory approval process and where the particular product stands in relation to that approval process, including any known safety or efficacy concerns, potential labeling restrictions and other impediments to approval. We evaluate our anticipated research and development initiatives and constraints relating to the product and the indication in which it will be used. We consider our manufacturing environment including our supply chain in determining logistical constraints that could hamper approval or commercialization. We consider the shelf life of the product in relation to the expected timeline for approval and we consider patent related or contract issues that may prevent or delay commercialization. We also base our judgment on the viability of commercialization, trends in the marketplace and market acceptance criteria. Finally, we consider the reimbursement strategies that may prevail with respect to the product and assess the economic benefit that we are likely to realize. We expense previously capitalized costs related to pre-approval inventory upon a change in such judgment, due to, among other potential factors, a denial or significant delay of approval by necessary regulatory bodies. At December 31, 2020, we capitalized approximately $93.8 million of pre-launch inventory for aducanumab, an anti-amyloid beta antibody candidate for the potential treatment of Alzheimer's disease that we are developing in collaboration with Eisai Co., Ltd. (Eisai). If aducanumab does not receive regulatory approval in the U.S., we would expense this inventory as research and development expense and, under the terms of our collaboration agreement with Eisai to jointly develop and commercialize aducanumab, Eisai would reimburse us for 45.0% of the costs. Obsolescence and Unmarketable Inventory At each reporting period we review our inventories for excess or obsolescence and write-down obsolete or otherwise unmarketable inventory to its estimated net realizable value. If the actual net realizable value is less than that estimated by us, or if it is determined that inventory utilization will further diminish based on estimates of demand, additional inventory write-downs may be required. Additionally, our products are subject to strict quality control and monitoring that we perform throughout the manufacturing process. In the event that certain batches or units of product no longer meet quality specifications, we will record a charge to cost of sales to write-down any unmarketable inventory to its estimated net realizable value. In all cases, product inventory is carried at the lower of cost or its estimated net realizable value. Amounts written-down due to unmarketable inventory are charged to cost of sales. Property, Plant and Equipment Property, plant and equipment are carried at cost, subject to reviews for impairment whenever events or changes in circumstances indicate that the carrying amount of the asset may not be recoverable. The cost of normal, recurring or periodic repairs and maintenance activities related to property, plant and equipment are expensed as incurred. The cost for planned major maintenance activities, including the related acquisition or construction of assets, is capitalized if the repair will result in future economic benefits. Interest costs incurred during the construction of major capital projects are capitalized until the underlying asset is ready for its intended use, at which point the interest costs are amortized as depreciation expense over the life of the underlying asset. We also capitalize certain direct and incremental costs associated with the validation effort required for licensing by regulatory agencies of new manufacturing equipment for the production of a commercially approved drug. These costs primarily include direct labor and material and are incurred in preparing the equipment for its intended use. The validation costs are either amortized over the life of the related equipment or expensed as cost of sales when the product produced in the validation process is sold. In addition, we capitalize certain internal use computer software development costs. If the software is an integral part of production assets, these costs are included in machinery and equipment and are amortized on a straight-line basis over the estimated useful lives of the related software, which generally range from to five years. We generally depreciate or amortize the cost of our property, plant and equipment using the straight-line method over the estimated useful lives of the respective assets, which are summarized as follows:
When we dispose of property, plant and equipment, we remove the associated cost and accumulated depreciation from the related accounts in our consolidated balance sheets and include any resulting gain or loss in our consolidated statements of income. Leases In February 2016 the FASB issued ASU No. 2016-02, Leases (Topic 842), a new standard issued to increase transparency and comparability among organizations related to their leasing activities. This standard established a right-of-use model that requires all lessees to recognize right-of-use assets and lease liabilities on their balance sheet that arise from leases as well as provide disclosures with respect to certain qualitative and quantitative information related to a company's leasing arrangements to meet the objective of allowing users of financial statements to assess the amount, timing and uncertainty of cash flows arising from leases. The FASB subsequently issued the following amendments to ASU 2016-02 that have the same effective date and transition date: ASU No. 2018-01, Leases (Topic 842): Land Easement Practical Expedient for Transition to Topic 842, ASU No. 2018-10, Codification Improvements to Topic 842, Leases, ASU No. 2018-11, Leases (Topic 842): Targeted Improvements, ASU No. 2018-20, Narrow-Scope Improvement for Lessors, and ASU No. 2019-01, Leases (Topic 842): Codification Improvements. We adopted these amendments with ASU 2016-02 (collectively, the new leasing standards) effective January 1, 2019. We adopted the new leasing standards using the modified retrospective transition approach, as of January 1, 2019, with no restatement of prior periods or cumulative adjustment to retained earnings. Upon adoption, we elected the package of transition practical expedients, which allowed us to carry forward prior conclusions related to whether any expired or existing contracts are or contain leases, the lease classification for any expired or existing leases and initial direct costs for existing leases. We also elected the practical expedient to not reassess certain land easements and made an accounting policy election to not recognize leases with an initial term of 12 months or less within our consolidated balance sheets and to recognize those lease payments on a straight-line basis in our consolidated statements of income over the lease term. Upon adoption of the new leasing standards we recognized an operating lease asset of approximately $463.0 million and a corresponding operating lease liability of approximately $526.0 million, which are included in our consolidated balance sheets. The adoption of the new leasing standards did not have an impact on our consolidated statements of income. We determine if an arrangement is a lease at contract inception. Operating lease assets represent our right to use an underlying asset for the lease term and operating lease liabilities represent our obligation to make lease payments arising from the lease. Operating lease assets and liabilities are recognized at the commencement date of the lease based upon the present value of lease payments over the lease term. When determining the lease term, we include options to extend or terminate the lease when it is reasonably certain that we will exercise that option. We use the implicit rate when readily determinable and use our incremental borrowing rate when the implicit rate is not readily determinable based upon the information available at the commencement date in determining the present value of the lease payments. Our incremental borrowing rate is determined using a secured borrowing rate for the same currency and term as the associated lease. The lease payments used to determine our operating lease assets may include lease incentives, stated rent increases and escalation clauses linked to rates of inflation when determinable and are recognized in our operating lease assets in our consolidated balance sheets. Our lease agreements may include both lease and non-lease components, which we account for as a single lease component when the payments are fixed. Variable payments included in the lease agreement are expensed as incurred. For certain equipment leases, such as vehicles, we apply a portfolio approach to effectively account for the operating lease assets and liabilities. Our operating leases are reflected in operating lease assets, accrued expenses and other and in long-term operating lease liabilities in our consolidated balance sheets. Lease expense for minimum lease payments is recognized on a straight-line basis over the lease term. We also have real estate lease agreements which are subleased to third parties. Operating leases for which we are the sublessor are included in accrued expenses and other and other long-term liabilities in our consolidated balance sheets. We recognize sublease income on a straight-line basis over the lease term in our consolidated statements of income. For additional information on the adoption of the new leasing standards, please read Note 11, Leases, to these consolidated financial statements. Intangible Assets Our intangible assets consist of completed technology (comprised of acquired and in-licensed rights and patents, developed technology, out-licensed patents), in-process research and development (IPR&D) acquired after January 1, 2009, trademarks and trade names. Our intangible assets are recorded at fair value at the time of their acquisition and are stated in our consolidated balance sheets net of accumulated amortization and impairments, if applicable. Intangible assets related to acquired and in-licensed rights and patents, developed technology and out-licensed patents are amortized over their estimated useful lives using the economic consumption method if anticipated future revenues can be reasonably estimated. The straight-line method is used when revenues cannot be reasonably estimated. Amortization is recorded within amortization and impairment of acquired intangible assets in our consolidated statements of income. Acquired and in-licensed rights and patents primarily relate to our acquisition of all remaining rights to TYSABRI from Elan Pharma International Ltd. (Elan), an affiliate of Elan Corporation, plc. Acquired and in-licensed rights and patents also include other amounts related to our other marketed products and programs acquired through business combinations. Developed technology primarily relates to our AVONEX product, which was recorded in connection with the merger of Biogen, Inc. and IDEC Pharmaceuticals Corporation in 2003. We amortize the intangible assets related to our TYSABRI, AVONEX, SPINRAZA, VUMERITY and TECFIDERA (rest of world) products using the economic consumption method based on revenues generated from the products underlying the related intangible assets. An analysis of the anticipated lifetime revenues of our TYSABRI, AVONEX, SPINRAZA, VUMERITY and TECFIDERA (rest of world) products is performed annually during our long-range planning cycle and whenever events or changes in circumstances would significantly affect the anticipated lifetime revenues of our TYSABRI, AVONEX, SPINRAZA, VUMERITY and TECFIDERA (rest of world) products. Intangible assets related to trademarks, trade names and IPR&D prior to commercialization are not amortized because they have indefinite lives; however, they are subject to review for impairment. We review our intangible assets with indefinite lives for impairment annually, as of October 31, and whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable. Acquired In-process Research and Development (IPR&D) Acquired IPR&D represents the fair value assigned to research and development assets that have not reached technological feasibility. The value assigned to acquired IPR&D is determined by estimating the costs to develop the acquired technology into commercially viable products, estimating the resulting revenues from the projects and discounting the net cash flows to present value. The revenues and costs projections used to value acquired IPR&D are, as applicable, reduced based on the probability of success of developing a new drug. Additionally, the projections consider the relevant market sizes and growth factors, expected trends in technology and the nature and expected timing of new product introductions by us and our competitors. The rates utilized to discount the net cash flows to their present value are commensurate with the stage of development of the projects and uncertainties in the economic estimates used in the projections. Upon the acquisition of IPR&D, we complete an assessment of whether our acquisition constitutes the purchase of a single asset or a group of assets. We consider multiple factors in this assessment, including the nature of the technology acquired, the presence or absence of separate cash flows, the development process and stage of completion, quantitative significance and our rationale for entering into the transaction. If we acquire a business as defined under applicable accounting standards, then the acquired IPR&D is capitalized as an intangible asset. If we acquire an asset or group of assets that do not meet the definition of a business under applicable accounting standards, then the acquired IPR&D is expensed on its acquisition date. Future costs to develop these assets are recorded to research and development expense as they are incurred. When performing our impairment assessment, we calculate the fair value using the same methodology as described above. If the carrying value of our acquired IPR&D exceeds its fair value, then the intangible asset is written down to its fair value. Changes in estimates and assumptions used in determining the fair value of our acquired IPR&D could result in an impairment. Impairments are recorded within amortization and impairment of acquired intangible assets in our consolidated statements of income. Assets that have been previously impaired, including our vixotrigine (BIIB074) program for the potential treatment of neuropathic pain, such as trigeminal neuralgia (TGN), could become further impaired in the future. Goodwill Goodwill represents the difference between the purchase price and the fair value of the identifiable tangible and intangible net assets when accounted for using the purchase method of accounting. Goodwill is not amortized, but is reviewed for impairment. Goodwill is reviewed for impairment annually, as of October 31, and whenever events or changes in circumstances indicate that the carrying value of the goodwill may not be recoverable. We compare the fair value of our reporting unit to its carrying value. If the carrying value of the net assets assigned to the reporting unit exceeds the fair value of our reporting unit, we would record an impairment loss equal to the difference. As described in Note 24, Segment Information, to these consolidated financial statements, we operate in one operating segment, which is our only reporting unit. Impairment of Long-Lived Assets Long-lived assets to be held and used, including property, plant and equipment, and definite-lived intangible assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of the assets or asset group may not be recoverable. Determination of recoverability is based on an estimate of undiscounted future cash flows resulting from the use of the asset and its eventual disposition. In the event that such cash flows are not expected to be sufficient to recover the carrying amount of the assets, the assets are written-down to their fair values. Long-lived assets to be disposed of are carried at fair value less costs to sell. Contingent Consideration The consideration for our acquisitions often includes future payments that are contingent upon the occurrence of a particular event or events. We record an obligation for such contingent payments at fair value on the acquisition date. We estimate the fair value of contingent consideration obligations through valuation models that incorporate probability-adjusted assumptions related to the achievement of the milestones and thus likelihood of making related payments. We revalue our contingent consideration obligations each reporting period. Changes in the fair value of our contingent consideration obligations are recognized in our consolidated statements of income. Changes in the fair value of the contingent consideration obligations can result from changes to one or multiple inputs, including adjustments to the discount rates, changes in the amount or timing of expected expenditures associated with product development, changes in the amount or timing of cash flows and reserves associated with products upon commercialization, changes in the assumed achievement or timing of any cumulative sales-based and development milestones, changes in the probability of certain clinical events and changes in the assumed probability associated with regulatory approval. Discount rates in our valuation models represent a measure of the credit risk associated with settling the liability. The period over which we discount our contingent obligations is based on the current development stage of the product candidates, our specific development plan for that product candidate adjusted for the probability of completing the development step and when the contingent payments would be triggered. In estimating the probability of success, we utilize data regarding similar milestone events from several sources, including industry studies and our own experience. These fair value measurements are based on significant inputs not observable in the market. Significant judgment is employed in determining the appropriateness of these assumptions as of the acquisition date and for each subsequent period. Derivative Instruments and Hedging Activities Cash Flow and Fair Value Derivative Instruments We recognize all derivative instruments as either assets or liabilities at fair value in our consolidated balance sheets. Changes in the fair value of our derivative instruments are recognized each period in current earnings or accumulated other comprehensive income (loss), depending on whether the derivative instrument is designated as part of a hedge transaction and, if so, the type of hedge transaction. We classify the cash flows from these instruments in the same category as the cash flows from the hedged items. We do not hold or issue derivative instruments for trading or speculative purposes. We assess at inception and on an ongoing basis, whether the derivative instruments that are used in hedging transactions are highly effective in offsetting the changes in cash flows or fair values of the hedged items. We exclude the forward points portion of the derivative instruments used in a hedging transaction from the effectiveness test and record the fair value gain or loss related to this portion each period in our consolidated statements of income in the same line as the underlying hedged item. If we determine that a forecasted transaction is no longer probable of occurring, we discontinue hedge accounting for the affected portion of the hedge instrument, and any related unrealized gain or loss on the contract is recognized in current earnings. Net Investment Derivative Instruments We are exposed to the impact of foreign exchange fluctuations on our investment in the equity of Samsung Bioepis, which is denominated in a currency other than the U.S. dollar, and could adversely impact the U.S. dollar value of this investment. Using derivative instruments, we have hedged our net investment position to mitigate the effects of foreign exchange fluctuations. We recognize these designated net investment hedges as either assets or liabilities, at fair value, in our consolidated balance sheets. We hedge the changes in the spot exchange rate in accumulated other comprehensive income (loss) and exclude changes to the forward rate and amortize the forward points in other income (expense), net in our consolidated statements of income over the term of the contract. We classify the cash flows from these instruments in the same category as the cash flows from the hedged items. For additional information on our derivative instruments and hedging activities, please read Note 9, Derivative Instruments, to these consolidated financial statements. Translation of Foreign Currencies The functional currency for most of our foreign subsidiaries is their local currency. For our non-U.S. subsidiaries that transact in a functional currency other than the U.S. dollar, assets and liabilities are translated at current rates of exchange at the balance sheet date. Income and expense items are translated at the average foreign currency exchange rates for the period. Adjustments resulting from the translation of the financial statements of our foreign operations into U.S. dollars are excluded from the determination of net income and are recorded in accumulated other comprehensive income, a separate component of equity. For subsidiaries where the functional currency of the assets and liabilities differ from the local currency, non-monetary assets and liabilities are translated at the rate of exchange in effect on the date assets were acquired while monetary assets and liabilities are translated at current rates of exchange as of the balance sheet date. Income and expense items are translated at the average foreign currency rates for the period. Translation adjustments of these subsidiaries are included in other income (expense), net in our consolidated statements of income. Royalty Cost of Sales We make royalty payments to a number of third parties under license or purchase agreements associated with our acquisition of intellectual property. These royalty payments are typically calculated as a percentage (royalty rate) of the sales of our products in a particular year. That royalty rate may remain constant, increase or decrease within each year based on the total amount of sales during the annual period. Each quarterly period, we estimate our total royalty obligation for the full year and recognize the proportional amount as cost of sales based on actual quarterly sales as a percentage of full year estimated sales. For example, if the level of net sales in any calendar year increases the royalty rate within the year, we will record our cost of sales at an even rate over the year, based on the estimated blended royalty rate. Accounting for Share-Based Compensation Our share-based compensation programs grant awards that have included stock options, restricted stock units that vest based on stock performance known as market stock units (MSUs), performance-vested restricted stock units that settle in cash (CSPUs), time-vested restricted stock units (RSUs), performance-vested restricted stock units that can be settled in cash or shares of our common stock (PUs) at the sole discretion of the Compensation and Management Development Committee of our Board of Directors, performance-vested stock units that settle in stock or cash (PSUs) and shares issued under our employee stock purchase plan (ESPP). Compensation expense is recognized based on the estimated fair value of the awards at grant date. We recognize compensation expense for the number of awards expected to vest after taking into consideration an estimate of award forfeitures over the requisite service period, which is generally the vesting period. Where awards are made with non-substantive vesting periods (for instance, where a portion of the award vests upon retirement eligibility), we estimate and recognize expense based on the period from the grant date to the date the employee becomes retirement eligible. The fair values of our MSUs are estimated using a lattice model with a Monte Carlo simulation. We apply an accelerated attribution method to recognize share-based compensation expense over the applicable service period for our MSUs. The probability of actual shares expected to be earned is considered in the grant date valuation, therefore the expense is not adjusted to reflect the actual units earned. The fair values of our RSUs are based on the market value of our stock on the date of grant. Compensation expense for RSUs is recognized straight-line over the applicable service period. We apply an accelerated attribution method to recognize share-based compensation expense when accounting for our CSPUs, PUs and PSUs that settle in cash, and the fair value of the liability is remeasured at the end of each reporting period through expected settlement. Compensation expense associated with CSPUs, PUs and PSUs that settle in cash are based upon the stock price and the number of units expected to be earned after assessing the probability that certain performance criteria will be met and the targeted payout level associated with the performance criteria expected to be achieved. Cumulative adjustments are recorded each quarter to reflect changes in the stock price and estimated outcome of the performance-related conditions until the date results are determined and settled. If performance criteria are not met or not expected to be met, any compensation expense previously recognized to date associated with the awards will be reversed. The fair values of PSUs that settle in stock are based upon the stock price on the date of grant. Compensation expense is recognized for the number of units expected to be earned after assessing the probability that certain performance criteria will be met and the targeted payout level associated with the performance criteria expected to be achieved. Cumulative adjustments are recorded each quarter to reflect the estimated outcome of the performance-related conditions until the date results are determined and settled. If performance criteria are not met or not expected to be met, any compensation expense previously recognized to date associated with the awards will be reversed. Research and Development Expenses Research and development expenses consist of expenses incurred in performing research and development activities, which include compensation and benefits, facilities and overhead expenses, clinical trial expenses and fees paid to contract research organizations (CROs), clinical supply and manufacturing expenses, write-offs of inventory that was previously capitalized in anticipation of product launch and determined to no longer be realizable and other outside expenses and upfront fees and milestones paid to third-party collaborators. Research and development expenses are expensed as incurred. Upfront and milestone payments made to third-party collaborators are expensed as incurred up to the point of regulatory approval. Milestone payments made upon regulatory approval are capitalized and amortized over the remaining useful life of the related product. Payments we make for research and development services prior to the services being rendered are recorded as prepaid assets in our consolidated balance sheets and are expensed as the services are provided. We also accrue the costs of ongoing clinical trials associated with programs that have been terminated or discontinued for which there is no future economic benefit at the time the decision is made to terminate or discontinue the program. From time to time, we enter into development agreements in which we share expenses with a collaborative partner. We record payments received from our collaborative partners for their share of the development costs as a reduction of research and development expense, except as discussed in Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Because an initial indication has been approved for both RITUXAN and GAZYVA, expenses incurred by Genentech in the ongoing development of RITUXAN and GAZYVA are not recorded as research and development expense, but rather reduce our share of profits recorded as a component of revenues from anti-CD20 therapeutic programs. For collaborations with commercialized products, if we are the principal, we record revenues and the corresponding operating costs in their respective line items in our consolidated statements of income. If we are not the principal, we record operating costs as a reduction of revenue. Selling, General and Administrative Expenses Selling, general and administrative expenses are primarily comprised of compensation and benefits associated with sales and marketing, finance, human resources, legal, information technology and other administrative personnel, outside marketing, advertising and legal expenses and other general and administrative costs. Advertising costs are expensed as incurred. For the years ended December 31, 2020, 2019 and 2018, advertising costs totaled $111.8 million, $79.2 million and $90.2 million, respectively. Income Taxes The provision for income taxes includes federal, state, local and foreign taxes. Income taxes are accounted for under the asset and liability method. Deferred tax assets and liabilities are recognized for the estimated future tax consequences of temporary differences between the financial statement carrying amounts and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the year in which the temporary differences are expected to be recovered or settled. We evaluate the realizability of our deferred tax assets and establish a valuation allowance when it is more likely than not that all or a portion of deferred tax assets will not be realized. We recognize deferred taxes associated with our global intangible low-taxed income (GILTI) tax calculations. The income tax consequences from the intra-entity transfers of inventory within our consolidated group, both current and deferred, are recorded as a prepaid tax or deferred charge and recognized through our consolidated statements of income when the inventory is sold to a third party. In October 2016 the FASB issued ASU No. 2016-16, Income Taxes (Topic 740): Intra-Entity Transfer of Assets Other Than Inventory. This standard eliminates the deferral of the tax effects of intra-entity asset transfers other than inventory. As a result, the income tax consequences from the intra-entity transfer of an asset other than inventory and associated changes to deferred taxes will be recognized when the transfer occurs. We adopted this standard on January 1, 2018, using the modified retrospective method, through a cumulative-effect adjustment to retained earnings as of that date. Upon adoption, we recognized additional net deferred tax assets of approximately $0.5 billion, offset by a corresponding net increase to retained earnings of approximately $0.5 billion. In the fourth quarter of 2018, when we elected to begin recognizing deferred taxes on the GILTI tax calculation, we recorded an additional deferred tax liability of $0.4 billion with a corresponding reduction to our retained earnings as these differences are related to intra-entity transactions. We will recognize incremental deferred income tax expense thereafter as these deferred tax assets and liabilities are utilized. We account for uncertain tax positions using a “more likely than not” threshold for recognizing and resolving uncertain tax positions. We evaluate uncertain tax positions on a quarterly basis and consider various factors including, but not limited to, changes in tax law, the measurement of tax positions taken or expected to be taken in tax returns, the effective settlement of matters subject to audit, information obtained during in process audit activities and changes in facts or circumstances related to a tax position. We also accrue for potential interest and penalties related to unrecognized tax benefits in income tax expense. Contingencies We are currently involved in various claims and legal proceedings. Loss contingency provisions are recorded if the potential loss from any claim, asserted or unasserted, or legal proceeding is considered probable and the amount can be reasonably estimated or a range of loss can be determined. These accruals represent management’s best estimate of probable loss. Disclosure also is provided when it is reasonably possible that a loss will be incurred or when it is reasonably possible that the amount of a loss will exceed the recorded provision. On a quarterly basis, we review the status of each significant matter and assess its potential financial exposure. Significant judgment is required in both the determination of probability and as to whether an exposure is reasonably estimable. Because of uncertainties related to these matters, accruals are based only on the best information available at the time. As additional information becomes available, we reassess the potential liability related to pending claims and litigation and may change our estimates. Legal costs associated with legal proceedings are expensed when incurred. Earnings per Share Basic earnings per share is computed by dividing undistributed net income attributable to Biogen Inc. by the weighted-average number of common shares outstanding during the period. Diluted earnings per share is computed based on the treasury method by dividing net income by the weighted-average number of common shares outstanding during the period plus potentially dilutive common equivalent shares outstanding. New Accounting Pronouncements From time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that we adopt as of the specified effective date. Unless otherwise discussed below, we do not believe that the adoption of recently issued standards have or may have a material impact on our consolidated financial statements or disclosures. Leases In February 2016 the FASB issued the new leasing standards to increase transparency and comparability among organizations related to their leasing activities. For additional information on the adoption of the new leasing standards, please read the section titled Lease above, and Note 11, Leases, to these consolidated financial statements. Credit Losses In June 2016 the FASB issued ASU No. 2016-13, Financial Instruments - Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments. The FASB subsequently issued amendments to ASU 2016-13, which have the same effective date and transition date of January 1, 2020. These standards require that credit losses be reported using an expected losses model rather than the incurred losses model that is currently used, and establishes additional disclosures related to credit risks. For available-for-sale debt securities with unrealized losses, these standards now require allowances to be recorded instead of reducing the amortized cost of the investment. These standards limit the amount of credit losses to be recognized for available-for-sale debt securities to the amount by which carrying value exceeds fair value and requires the reversal of previously recognized credit losses if fair value increases. Based on the composition of our investment portfolio, accounts receivable and other financial assets, current market conditions and historical credit loss activity, the adoption of these standards did not have a material impact on our consolidated financial position and results of operations and related disclosures. Debt Securities In March 2017 the FASB issued ASU No. 2017-08, Receivables - Nonrefundable Fees and Other Costs (Subtopic 310-20): Premium Amortization on Purchased Callable Debt Securities. This standard amends the amortization period for certain purchased callable debt securities held at a premium by shortening the amortization period to the earliest call date. This standard became effective for us on January 1, 2019, and was adopted using a modified retrospective transition approach. The adoption of this standard did not result in a significant adjustment to our marketable debt securities. Fair Value Measurements In August 2018 the FASB issued ASU No. 2018-13, Fair Value Measurement (Topic 820): Disclosure Framework - Changes to the Disclosure Requirements for Fair Value Measurement. This standard modifies certain disclosure requirements on fair value measurements. This standard became effective for us on January 1, 2020. The adoption of this standard did not have a material impact on our disclosures. Derivative Instruments and Hedging Activities In October 2018 the FASB issued ASU No. 2018-16, Derivatives and Hedging (Topic 815): Inclusion of the Secured Overnight Financing Rate (SOFR) Overnight Index Swap (OIS) Rate as a Benchmark Interest Rate for Hedge Accounting Purposes. This standard permits use of the OIS rate based on the SOFR as a U.S. benchmark interest rate for hedge accounting purposes under ASC 815, Derivatives and Hedging. This standard became effective for us on January 1, 2019, and did not have an impact on our consolidated results of operations or financial position. Collaborative Arrangements In November 2018 the FASB issued ASU No. 2018-18, Collaborative Arrangements (Topic 808): Clarifying the Interaction between Topic 808 and Topic 606. This standard makes targeted improvements for collaborative arrangements as follows: •Clarifies that certain transactions between collaborative arrangement participants should be accounted for as revenue under ASC 606, Revenue from Contracts with Customers, when the collaborative arrangement participant is a customer in the context of a unit of account. In those situations, all the guidance in ASC 606 should be applied, including recognition, measurement, presentation and disclosure requirements; •Adds unit-of-account guidance to ASC 808, Collaborative Arrangements, to align with the guidance in ASC 606 (that is, a distinct good or service) when an entity is assessing whether the collaborative arrangement or a part of the arrangement is within the scope of ASC 606; and •Precludes a company from presenting transactions with collaborative arrangement participants that are not directly related to sales to third parties with revenue recognized under ASC 606 if the collaborative arrangement participant is not a customer. This standard became effective for us on January 1, 2020. A retrospective transition approach is required for either all contracts or only for contracts that are not completed at the date of initial application of ASC 606, with a cumulative adjustment to opening retained earnings, as of January 1, 2018. The adoption of this standard did not have a material impact on our consolidated financial position, results of operations and related disclosures.
|
||||||||||||||||||||||||||||||||||||||||||||||||
Acquisitions |
12 Months Ended |
|---|---|
Dec. 31, 2020 | |
| Business Combinations [Abstract] | |
| Acquisitions | Acquisitions BIIB118 Acquisition In March 2020 we acquired BIIB118 (CK1 inhibitor), a novel CNS-penetrant small molecule inhibitor of casein kinase 1, for the potential treatment of patients with behavioral and neurological symptoms across various psychiatric and neurological diseases from Pfizer Inc. (Pfizer). We are developing BIIB118 for the potential treatment of irregular sleep wake rhythm disorder in Parkinson’s disease and plan to develop BIIB118 for the potential treatment of sundowning in Alzheimer's disease. In connection with this acquisition, we made an upfront payment of $75.0 million to Pfizer, which was accounted for as an asset acquisition and recorded as acquired IPR&D in our consolidated statements of income as BIIB118 has not yet reached technological feasibility. We may also pay Pfizer up to $635.0 million in potential additional development and commercialization milestone payments as well as tiered royalties in the high single digits to sub-teens. Acquisition of Nightstar Therapeutics plc In June 2019 we completed our acquisition of all of the outstanding shares of Nightstar Therapeutics plc (NST), a clinical-stage gene therapy company focused on adeno-associated virus treatments for inherited retinal disorders. As a result of this acquisition, we added two mid- to late-stage clinical assets, as well as preclinical programs, in ophthalmology. These assets include BIIB111 (timrepigene emparvovec), which is in Phase 3 development for the potential treatment of choroideremia, a rare, degenerative, X-linked inherited retinal disorder that leads to blindness and currently has no approved treatments, and BIIB112 (RPGR gene therapy), which is in Phase 2/3 development for the potential treatment of X-linked retinitis pigmentosa, which is a rare inherited retinal disease with no currently approved treatments. Under the terms of the acquisition, we paid NST shareholders $25.50 in cash for each issued and outstanding NST share, which totaled $847.6 million. In addition, we paid $4.6 million in cash for equity compensation, which is attributable to pre-combination services and is reflected as a component of the total purchase price paid. The fair value of equity compensation attributable to the post-combination service period was $26.2 million, of which $18.4 million was recognized as a charge to selling, general and administrative expense with the remaining $7.8 million as a charge to research and development expense in our consolidated statements of income. These amounts were associated with the accelerated vesting of stock options previously granted to NST employees and were fully paid in cash as of June 30, 2019. We funded this acquisition through available cash and accounted for it as an acquisition of a business. We finalized purchase accounting for this acquisition in the fourth quarter of 2019. The fair value of the IPR&D programs acquired was determined through a probability adjusted discounted cash flow analysis utilizing a discount rate of 12.5%. We recorded IPR&D assets for BIIB111 and BIIB112 at their initial fair values of $480.0 million and $220.0 million, respectively. Some of the more significant assumptions utilized in our asset valuations included the estimated net cash flows for each year for each asset or product, including net revenues, cost of sales, research and development and other operating expenses, the potential regulatory and commercial success risks, competitive trends impacting the asset and each cash flow stream as well as other factors. These fair value measurements were based on significant inputs not observable in the market and thus represent Level 3 fair value measurements. We recognized goodwill in relation to the fair value associated with NST workforce's expertise and early research in retinal disorders. We also recognized goodwill in relation to the establishment of a deferred tax liability for the acquired IPR&D intangible assets, which have no tax basis. This deferred tax liability is net of the related impacts on the deferred taxes for GILTI. Goodwill that is tax deductible for GILTI purposes is approximately $60.9 million as of December 31, 2020. Pro forma results of operations as a result of this acquisition have not been presented as this acquisition is not material to our consolidated statements of income. Subsequent to June 7, 2019, the acquisition date, our results of operations include the results of operations of NST. BIIB100 Acquisition In January 2018 we acquired BIIB100 (XP01 inhibitor) from Karyopharm Therapeutics Inc. (Karyopharm). BIIB100 is a Phase 1 investigational oral compound for the potential treatment of certain neurological and neurodegenerative diseases, primarily in ALS. BIIB100 is a novel therapeutic candidate that works by inhibiting a protein known as XPO1, with the goal of reducing inflammation and neurotoxicity, along with increasing neuroprotective responses. We accounted for this transaction as an asset acquisition as the value being acquired primarily relates to a single asset. In connection with the closing of this transaction, we made an upfront payment of $10.0 million to Karyopharm, which was recorded as acquired IPR&D in our consolidated statements of income as BIIB100 had not yet reached technological feasibility. We may also pay Karyopharm up to $207.0 million in additional milestone payments as well as tiered royalties on potential net commercial sales in the mid-single digit to low-teen percentages. BIIB104 Acquisition In April 2018 we acquired BIIB104 (AMPA) from Pfizer. BIIB104 is a first-in-class, Phase 2b ready AMPA receptor potentiator for cognitive impairment associated with schizophrenia. AMPA receptors mediate fast excitatory synaptic transmission in the central nervous system, a process which can be disrupted in a number of neurological and psychiatric diseases, including schizophrenia. We accounted for this transaction as an asset acquisition as the value being acquired primarily relates to a single asset. In connection with the closing of this transaction, we made an upfront payment of $75.0 million to Pfizer, which was recorded as acquired IPR&D in our consolidated statements of income as BIIB104 had not yet reached technological feasibility. We may also pay Pfizer up to $515.0 million in total development and commercialization milestone payments as well as tiered royalties on potential net commercial sales in the low to mid-teen percentages. BIIB110 Acquisition In July 2018 we acquired BIIB110 (ActRIIA/B ligand trap) and ALG-802 from AliveGen Inc. (AliveGen). BIIB110 and ALG-802 represent novel ways of targeting the myostatin pathway. We initially plan to study BIIB110 in multiple neuromuscular indications, including SMA and ALS. We accounted for this transaction as an asset acquisition as the value being acquired primarily relates to a single asset. In connection with the closing of this transaction, we made an upfront payment of $27.5 million to AliveGen, which was recorded as acquired IPR&D in our consolidated statements of income as BIIB110 had not yet reached technological feasibility. We may also pay AliveGen up to $535.0 million in additional development and commercialization milestones.
|
Divestitures |
12 Months Ended |
|---|---|
Dec. 31, 2020 | |
| Divestitures [Abstract] | |
| Divestitures | Divestitures Divestiture of Hillerød, Denmark Manufacturing Operations In August 2019 we completed the sale of all of the outstanding shares of our subsidiary that owned our biologics manufacturing operations in Hillerød, Denmark to FUJIFILM Corporation (FUJIFILM). Upon the closing of this transaction, we received approximately $881.9 million in cash, which may be adjusted based on other contractual terms, which are discussed below. We determined that the operations disposed of in this transaction did not meet the criteria to be classified as discontinued operations under the applicable guidance. As part of this transaction, we provided FUJIFILM with certain minimum batch production commitment guarantees. There is a risk that the minimum contractual batch production commitments will not be met. Based upon current estimates we do not expect to incur an adverse commitment obligation associated with such guarantees. We may further adjust this estimate based upon changes in business conditions, which may result in the increase or reduction of this adverse commitment obligation in subsequent periods. We also may be obligated to indemnify FUJIFILM for liabilities that existed relating to certain business activities incurred prior to the closing of this transaction. In addition, we may earn certain contingent payments based on future manufacturing activities at the Hillerød facility. For the disposition of a business, our policy is to recognize contingent consideration when the consideration is realizable. We currently believe the probability of earning these payments is remote and therefore we did not include these contingent payments in our calculation of the fair value of the operations. As part of this transaction, we entered into certain manufacturing services agreements with FUJIFILM pursuant to which FUJIFILM will use the Hillerød facility to produce commercial products for us, such as TYSABRI, as well as other third-party products. For the year ended December 31, 2019, we recognized a total net loss of approximately $124.2 million related to the transaction in our consolidated statements of income. This loss included a pre-tax loss of $55.3 million, which was recorded in loss on divestiture of Hillerød, Denmark manufacturing operations. The loss recognized was based on exchange rates and business conditions on the closing date of this transaction, and included costs to sell our Hillerød, Denmark manufacturing operations of approximately $11.2 million and our estimate of the fair value of adverse commitments of approximately $74.0 million, primarily associated with the guarantee of future minimum batch production at the Hillerød facility. We also recorded a tax expense of $68.9 million related to this transaction. In addition, upon the closing of this transaction, we sold to FUJIFILM $41.8 million of raw materials that were remaining at the Hillerød facility on the closing date of this transaction. These materials were sold at cost, which approximates fair value. During the year ended December 31, 2020, we reduced our estimate of the fair value of the adverse commitment associated with the guarantee of future batch production by approximately $62.0 million based on our current manufacturing forecasts. Additionally, we recorded a reduction to our pre-tax loss of approximately $30.5 million due to a refund of interest paid associated with a tax matter. Our estimate of the fair value of the adverse commitments is a Level 3 measurement and is based on forecasted batch production at the Hillerød facility.
|
Revenues |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revenue from Contract with Customer [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revenues | Revenues Product Revenues Revenues by product are summarized as follows:
*Fumarate includes TECFIDERA and VUMERITY. VUMERITY became commercially available in the U.S. in November 2019. **Interferon includes AVONEX and PLEGRIDY. We recognized revenues from two wholesalers accounting for 30.5% and 15.3% of gross product revenues in 2020, 30.0% and 17.2% of gross product revenues in 2019 and 32.0% and 18.4% of gross product revenues in 2018, respectively. As of December 31, 2020, two wholesale distributors individually accounted for approximately 21.1% and 8.5% of net accounts receivable associated with our product sales, as compared to 24.1% and 13.9% as of December 31, 2019, respectively. An analysis of the change in reserves for discounts and allowances is summarized as follows:
The total reserves above, which are included in our consolidated balance sheets, are summarized as follows:
Revenues from Anti-CD20 Therapeutic Programs Revenues from anti-CD20 therapeutic programs are summarized in the table below. For purposes of this footnote, we refer to RITUXAN and RITUXAN HYCELA collectively as RITUXAN.
Approximately 14.7%, 15.9% and 14.7% of our total revenues in 2020, 2019 and 2018, respectively, were derived from our collaboration arrangements with Genentech. For additional information on our collaboration arrangements with Genentech, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Other Revenues Other revenues are summarized as follows:
Other corporate revenues primarily reflect amounts earned under contract manufacturing agreements with our strategic customers, including Bioverativ Inc. (Bioverativ). During the years ended December 31, 2020, 2019 and 2018, we recognized $48.6 million, $383.2 million and $206.7 million, respectively, in revenues under the manufacturing and supply agreement with Bioverativ entered into in connection with the spin-off of our hemophilia business. During the third quarter of 2019, we amended our agreement with a contract manufacturing customer pursuant to which we licensed certain of our manufacturing-related intellectual property to the customer. In the second quarter of 2020, the customer received regulatory approval for its product that is being manufactured using certain of our manufacturing-related intellectual property. As a result we are entitled to $500.0 million in a series of three payments. The first payment became due upon a regulatory approval of such product and was received during the second quarter of 2020. Subsequent payments are due on the first and second anniversaries of the regulatory approval. Other corporate revenues for the year ended December 31, 2020, reflect $346.2 million related to the delivery of the license for certain of our manufacturing-related intellectual property under the amended agreement discussed above and the performance of manufacturing product supply services for such customer. We have allocated the remaining $153.8 million of the $500.0 million transaction price to the performance of manufacturing product supply services for the customer, which we expect to perform through 2026. The value allocated to the manufacturing services was based on expected demand for supply and the fair value of comparable manufacturing and development services. For additional information on our collaboration arrangements with Samsung Bioepis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Inventory |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inventory Disclosure [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inventory | Inventory The components of inventory are summarized as follows:
Inventory amounts written down as a result of excess, obsolescence, unmarketability or other reasons are charged to cost of sales, and totaled $26.6 million, $52.2 million and $41.9 million for the years ended December 31, 2020, 2019 and 2018, respectively. Divestiture of Hillerød, Denmark Manufacturing Operations In August 2019 we completed the sale of all of the outstanding shares of our subsidiary that owned our biologics manufacturing operations in Hillerød, Denmark to FUJIFILM. This transaction included the sale of $14.0 million of work in process inventory. In addition, we sold to FUJIFILM approximately $41.8 million of raw materials that were remaining at the Hillerød facility on the closing date of this transaction. These materials were sold at cost, which approximates fair value. For additional information on the divestiture of our Hillerød, Denmark manufacturing operations, please read Note 3, Divestitures, to these consolidated financial statements.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Intangible Assets and Goodwill |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Goodwill and Intangible Assets Disclosure [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intangible Assets and Goodwill | Intangible Assets and Goodwill Intangible Assets Intangible assets, net of accumulated amortization, impairment charges and adjustments are summarized as follows:
Amortization and Impairments Amortization and impairments of acquired intangible assets totaled $464.8 million, $489.9 million and $747.3 million for the years ended December 31, 2020, 2019 and 2018, respectively. Amortization of acquired intangible assets, excluding impairment charges, totaled $255.1 million, $274.0 million and $381.2 million for the years ended December 31, 2020, 2019 and 2018, respectively. The decrease in amortization of acquired intangible assets, excluding impairment charges, over the three years was primarily due to a lower rate of amortization for acquired intangible assets. For the year ended December 31, 2020, amortization and impairment of acquired intangible assets reflects the impact of a $115.0 million impairment charge related to BIIB111, which was obtained as part of our acquisition of NST, a $75.4 million impairment charge related to BIIB054 (cinpanemab) and a $19.3 million impairment charge related to one of our other IPR&D intangible assets. For the year ended December 31, 2019, amortization and impairments of acquired intangible assets reflects the impact of a $215.9 million impairment charge related to certain IPR&D assets associated with the Phase 2b study of BG00011 (STX-100) for the potential treatment of idiopathic pulmonary fibrosis (IPF), which was discontinued in the third quarter of 2019. For the year ended December 31, 2018, amortization and impairments of acquired intangible assets reflects the impact of a $189.3 million impairment charge related to certain IPR&D assets associated with our vixotrigine program, as discussed below, and a $176.8 million impairment charge related to our U.S. license to Forward Pharma A/S' (Forward Pharma) intellectual property, including Forward Pharma's intellectual property related to TECFIDERA. Completed Technology Completed technology primarily relates to our acquisition of all remaining rights to TYSABRI from Elan as well as other amounts related to our other marketed products and programs acquired through business combinations. IPR&D Related to Business Combinations IPR&D represents the fair value assigned to research and development assets that we acquired as part of a business combination and had not yet reached technological feasibility at the date of acquisition. Included in IPR&D balances are adjustments related to foreign currency exchange rate fluctuations. We review amounts capitalized as acquired IPR&D for impairment annually, as of October 31, and whenever events or changes in circumstances indicate to us that the carrying value of the assets might not be recoverable. The carrying value associated with our IPR&D assets as of December 31, 2020 and 2019, relates to the various IPR&D programs we acquired in connection with our acquisitions of NST and Convergence Pharmaceuticals Holdings Ltd. (Convergence). The majority of the balance relates to our acquisition of NST in June 2019 whereby we acquired IPR&D programs with an estimated fair value of approximately $585.0 million as of December 31, 2020. For additional information on our acquisition of NST, please read Note 2, Acquisitions, to these consolidated financial statements. BIIB111 During the fourth quarter of 2020 we began experiencing third-party manufacturing delays that may impact our timeline for a potential filing of a Biologics License Application (BLA) for BIIB111 for regulatory approval by up to one year. In addition, we determined that forecasted costs associated with advancing the BIIB111 program through Phase 3 development and potential commercialization will exceed our original estimates. We reassessed the fair value of the program based on these changes in assumptions and determined that the program was partially impaired. We recognized an impairment charge of $115.0 million during the fourth quarter of 2020, which resulted in a reduction of the IPR&D asset from $480.0 million to $365.0 million. BIIB054 In February 2021 we announced that we discontinued development of BIIB054 as a potential treatment of Parkinson's disease as our Phase 2 SPARK study did not meet its primary or secondary endpoints. Although we made this determination in February 2021, it was based on conditions that existed as of December 31, 2020. As a result, we recognized an impairment charge of approximately $75.4 million during the fourth quarter of 2020 to reduce the fair value of the related IPR&D intangible asset to zero. The IPR&D impairment charges were included in amortization and impairment of acquired intangible assets and the gain resulting from the remeasurement of our contingent consideration obligation was recorded in (gain) loss on fair value remeasurement of contingent consideration in our consolidated statements of income. The fair value of the intangible assets and contingent consideration obligations were based on a probability-adjusted discounted cash flow calculation using Level 3 fair value measurements and inputs including estimated revenues, costs and probabilities of success. Vixotrigine In the periods since we acquired vixotrigine, there have been numerous delays in the initiation of Phase 3 studies for the potential treatment of TGN as we engaged with the U.S. Food and Drug Administration (FDA) regarding the design of the Phase 3 studies and awaited data and insights from mid-stage clinical trials of vixotrigine in other indications that have since been completed. The fair value of the TGN asset is not significantly in excess of carrying value. As of December 31, 2020, the carrying value associated with our vixotrigine IPR&D assets was $177.5 million. Estimated Future Amortization of Intangible Assets The estimated future amortization of finite-lived intangible assets for the next five years is expected to be as follows:
Goodwill The following table provides a roll forward of the changes in our goodwill balance:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Fair Value Measurements |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair Value Disclosures [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair Value Measurements | Fair Value Measurements The tables below present information about our assets and liabilities that are regularly measured and carried at fair value and indicate the level within the fair value hierarchy of the valuation techniques we utilized to determine such fair value:
There have been no material impairments of our assets measured and carried at fair value during the years ended December 31, 2020 and 2019. In addition, there have been no changes in valuation techniques during the years ended December 31, 2020 and 2019. The fair value of Level 2 instruments classified as cash equivalents and marketable debt securities was determined through third-party pricing services. The fair value of Level 2 instruments classified as marketable equity securities represents our investments in Sangamo Therapeutics, Inc. (Sangamo) common stock, Denali Therapeutics Inc. (Denali) common stock and Sage Therapeutics, Inc. (Sage) common stock and are valued using an option pricing valuation model as the investments are each subject to certain holding period restrictions. For additional information on our investments in Sangamo, Denali and Sage common stock, please read Note 8, Financial Instruments, to these consolidated financial statements. Our investments in marketable equity securities also include shares of Ionis Pharmaceuticals, Inc. (Ionis) common stock acquired in June 2018. Our shares of Ionis common stock were initially subject to certain holding period restrictions that have since expired. The fair value of this investment was a Level 1 measurement as of December 31, 2020. For additional information on our collaboration arrangements with Ionis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. For a description of our validation procedures related to prices provided by third-party pricing services and our option pricing valuation model, please read Note 1, Summary of Significant Accounting Policies - Fair Value Measurements, to these consolidated financial statements. The following table summarizes the significant unobservable inputs in the fair value measurement of our contingent consideration obligations as of December 31, 2020:
The weighted average discount rate was calculated based on the relative fair value of our contingent consideration obligations. In addition, we apply various probabilities of technological and regulatory success, ranging from 39.9% to certain probability, to the valuation models to estimate the fair values of our contingent consideration obligations. Nonrecurring Fair Value Measurements In addition to assets and liabilities that are recorded at fair value on a recurring basis, we record assets and liabilities at fair value on a nonrecurring basis as required by accounting principles generally accepted in the U.S. (U.S. GAAP). Generally, assets are recorded at fair value on a nonrecurring basis as a result of impairment charges. The gains or losses on assets measured at fair value on a nonrecurring basis, are summarized as follows:
For the year ended December 31, 2020, we recorded a partial impairment charge of $115.0 million related to BIIB111. For additional information, please read Note 6, Intangible Assets and Goodwill, to these consolidated financial statements. Debt Instruments The fair values of our debt instruments, which are Level 2 liabilities, are summarized as follows:
(1) Our 2.900% Senior Notes due September 15, 2020, were redeemed in full in May 2020 using the net proceeds from the issuance on April 30, 2020, of our senior unsecured notes for an aggregate principal amount of $3.0 billion. For additional information, please read Note 12, Indebtedness, to these consolidated financial statements. The fair values of each of our series of Senior Notes were determined through market, observable and corroborated sources. For additional information related to our Senior Notes, please read Note 12, Indebtedness, to these consolidated financial statements. Contingent Consideration Obligations In connection with our acquisitions of Convergence and Biogen International Neuroscience GmbH (BIN), we agreed to make additional payments based upon the achievement of certain milestone events. The following table provides a roll forward of the fair values of our contingent consideration obligations, which includes Level 3 measurements:
As of December 31, 2020 and 2019, approximately $110.3 million and $197.7 million, respectively, of the fair value of our total contingent consideration obligations was reflected as a component of other long-term liabilities in our consolidated balance sheets with the remaining balance reflected as a component of accrued expenses and other. For the year ended December 31, 2020, changes in the fair value of our contingent consideration obligations were primarily due to our discontinuing development of BIIB054 for the potential treatment of Parkinson's disease, resulting in a reduction of our contingent consideration obligations of $51.0 million as well as other changes in the probability and the expected timing of the achievement of certain remaining developmental milestones, changes in the interest rates used to revalue our contingent consideration liabilities and the passage of time. For the year ended December 31, 2019, changes in the fair value of our contingent consideration obligations were primarily due to the discontinuation of the Phase 2b study of BG00011 for the potential treatment of IPF resulting in a reduction of our contingent consideration obligations of $61.2 million as well as other changes in the probability and expected timing of achievement of certain developmental milestones, a decrease in interest rates used to revalue our contingent consideration liabilities and the passage of time. The fair values of the contingent consideration liabilities were based on a probability-adjusted discounted cash flow calculation using Level 3 fair value measurements and inputs. For additional information on the valuation techniques and inputs utilized in the valuation of our financial assets and liabilities, please read Note 1, Summary of Significant Accounting Policies, to these consolidated financial statements. Convergence Pharmaceuticals Holdings Limited In connection with our acquisition of Convergence in February 2015 we recorded a contingent consideration obligation of $274.5 million. As of December 31, 2020 and 2019, the fair value of this contingent consideration obligation was $259.8 million and $244.6 million, respectively. Our most recent valuation was determined based upon net cash flow projections of $400.0 million, probability weighted and discounted using a rate of 0.6%, which is a measure of the credit risk associated with settling the liability. Biogen International Neuroscience GmbH In connection with our acquisition of BIN in December 2010 we recorded a contingent consideration obligation of $81.2 million. We discontinued further development of BIIB054 for the potential treatment of Parkinson's disease based on the results of a Phase 2 study of BIIB054. Additionally, during the third and fourth quarters of 2020 we discontinued other programs related to our acquisition of BIN for which we had immaterial contingent consideration obligations. As a result, the fair value of the contingent consideration obligations related to our acquisition of BIN has been adjusted to zero, resulting in a gain of $101.5 million for the year ended December 31, 2020. Acquired IPR&D The fair values of the acquired IPR&D assets were based on a probability-adjusted discounted cash flow calculation using Level 3 fair value measurements and inputs including estimated revenues and probabilities of success. These assets are tested for impairment annually until commercialization, after which time the acquired IPR&D will be amortized over its estimated useful life using the economic consumption method. In connection with our acquisition of BIN, we recognized a $110.9 million acquired IPR&D intangible asset. We discontinued further development of BIIB054 for the potential treatment of Parkinson's disease and recognized an impairment charge of $75.4 million during the fourth quarter of 2020 to reduce the fair value of the IPR&D intangible asset to zero. In connection with our acquisition of Stromedix Inc., we recognized a $219.2 million acquired IPR&D intangible asset. During the third quarter of 2019 we discontinued the Phase 2b study of BG00011 for the potential treatment of IPF and recognized an impairment charge of $215.9 million to reduce the fair value of the IPR&D intangible asset to zero. In connection with our acquisition of Convergence, we recognized a $424.6 million acquired IPR&D intangible asset. During the third quarter of 2018 we recognized impairment charges related to certain IPR&D assets associated with our vixotrigine program totaling $189.3 million. For additional information on our IPR&D intangible assets, including a discussion of our most significant assumptions, please read Note 6, Intangible Assets and Goodwill, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Financial Instruments |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Investments, All Other Investments [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Financial Instruments | Financial Instruments The following table summarizes our financial assets with maturities of less than 90 days from the date of purchase included in cash and cash equivalents in our consolidated balance sheets:
The carrying values of our commercial paper, including accrued interest, overnight reverse repurchase agreements, money market funds and our short-term debt securities approximate fair value due to their short-term maturities. Our marketable equity securities gains (losses) are recorded in other income (expense), net in our consolidated statements of income. The following tables summarize our marketable debt and equity securities, classified as available for sale:
Summary of Contractual Maturities: Available-for-Sale Debt Securities The estimated fair value and amortized cost of our marketable debt securities available-for-sale by contractual maturity are summarized as follows:
The average maturity of our marketable debt securities available-for-sale as of December 31, 2020 and 2019, was approximately 11 months and 14 months, respectively. Proceeds from Marketable Debt Securities The proceeds from maturities and sales of marketable debt securities and resulting realized gains and losses are summarized as follows:
Realized losses for the year ended December 31, 2020, 2019 and 2018, primarily relate to sales of corporate bonds, agency mortgage-backed securities and other asset-backed securities. Strategic Investments As of December 31, 2020 and 2019, our strategic investment portfolio was comprised of investments totaling $2,024.6 million and $393.9 million, respectively, which are included in investments and other assets in our consolidated balance sheets. Our strategic investment portfolio includes investments in equity securities of certain biotechnology companies, which are reflected within our disclosures included in Note 7, Fair Value Measurements, to these consolidated financial statements, venture capital funds where the underlying investments are in equity securities of certain biotechnology companies and non-marketable equity securities. The increase in our strategic investment portfolio for the year ended December 31, 2020, was primarily due to our purchases of Sage, Denali and Sangamo common stock, as discussed below. These purchases were reflected as net cash flows used in investing activities within the consolidated statement of cash flows. Sage Therapeutics, Inc. In November 2020 we entered into a global collaboration and license agreement with Sage. In connection with the closing of this collaboration in December 2020 we purchased approximately 6.2 million shares of Sage common stock. This investment is classified as a Level 2 marketable equity security due to certain holding period restrictions and is remeasured each reporting period and carried at fair value. The effects of certain holding period restrictions on the investment are estimated using an option pricing valuation model. The most significant assumptions within the model are the term of the restrictions and the stock price volatility, which is based upon historical volatility of similar companies. We also use a constant maturity risk free-interest rate to match the remaining term of the restrictions on our investment in Sage common stock and a dividend yield of zero based upon the fact that Sage and similar companies generally have not historically granted cash dividends. For additional information on our collaboration agreement with Sage, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Denali Therapeutics Inc. In August 2020 we entered into a collaboration and license agreement with Denali. As part of this collaboration we purchased approximately 13 million shares of Denali common stock in September 2020. This investment is classified as a Level 2 marketable equity security due to certain holding period restrictions and is remeasured each reporting period and carried at fair value. The effects of certain holding period restrictions on the investment are estimated using an option pricing valuation model. The most significant assumptions within the model are the term of the restrictions and the stock price volatility, which is based upon historical volatility of similar companies. We also use a constant maturity risk free-interest rate to match the remaining term of the restrictions on our investment in Denali common stock and a dividend yield of zero based upon the fact that Denali and similar companies generally have not historically granted cash dividends. For additional information on our collaboration agreement with Denali, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Sangamo Therapeutics, Inc. In February 2020 we entered into a collaboration and license agreement with Sangamo. In connection with the closing of this transaction in April 2020 we purchased approximately 24 million shares of Sangamo common stock. This equity method investment will be remeasured each reporting period and carried at fair value due to our election of the fair value option. The effects of certain holding period restrictions on the investment are estimated using an option pricing valuation model. The most significant assumptions within the model are the term of the restrictions and the stock price volatility, which is based upon historical volatility of similar companies. We also use a constant maturity risk free-interest rate to match the remaining term of the restrictions on our investment in Sangamo common stock and a dividend yield of zero based upon the fact that Sangamo and similar companies generally have not historically granted cash dividends. For additional information on our collaboration agreement with Sangamo, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Samsung Bioepis In June 2018 we exercised our option under our joint venture agreement with Samsung BioLogics to increase our ownership percentage in Samsung Bioepis from approximately 5.0% to approximately 49.9%. The share purchase transaction was completed in November 2018 and, upon closing, we paid 759.5 billion South Korean won ($676.6 million) to Samsung BioLogics. As of December 31, 2020 and 2019, the carrying value of our investment in Samsung Bioepis totaled 673.8 billion South Korean won ($620.2 million) and 670.8 billion South Korean won ($580.2 million), respectively, which is classified as a component of investments and other assets within our consolidated balance sheets. For additional information on our collaboration arrangements with Samsung Bioepis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Derivative Instruments |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Derivative Instruments and Hedging Activities Disclosure [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Derivative Instruments | Derivative Instruments Foreign Currency Forward Contracts - Hedging Instruments Due to the global nature of our operations, portions of our revenues and operating expenses are recorded in currencies other than the U.S. dollar. The value of revenues and operating expenses measured in U.S. dollars is therefore subject to changes in foreign currency exchange rates. In order to mitigate these changes, we use foreign currency forward contracts to lock in exchange rates associated with a portion of our forecasted international revenues and operating expenses. Foreign currency forward contracts in effect as of December 31, 2020 and 2019, had durations of 1 to 24 months and 1 to 15 months, respectively. These contracts have been designated as cash flow hedges and unrealized gains or losses on the portion of these foreign currency forward contracts that are included in the effectiveness test are reported in accumulated other comprehensive income (loss) (referred to as AOCI in the table below). Realized gains and losses of such contracts are recognized in revenues when the sale of product in the currency being hedged is recognized and in operating expenses when the expense in the currency being hedged is recorded. We recognize all cash flow hedge reclassifications from accumulated other comprehensive income (loss) and fair value changes of excluded portions in the same line item in our consolidated statements of income that has been impacted by the hedged item. The notional value of foreign currency forward contracts that were entered into to hedge forecasted revenues and operating expenses is summarized as follows:
The pre-tax portion of the fair value of these foreign currency forward contracts that were included in accumulated other comprehensive income (loss) in total equity reflected net losses of $212.5 million as of December 31, 2020, net gains of $0.5 million as of December 31, 2019, and net gains of $27.3 million as of December 31, 2018. We expect the net losses of $212.5 million to be settled over the next 24 months, of which $175.2 million of these losses are expected to be settled over the next 12 months, with any amounts in accumulated other comprehensive income (loss) to be reported as an adjustment to revenues or operating expenses. We consider the impact of our and our counterparties’ credit risk on the fair value of the contracts as well as the ability of each party to execute its contractual obligations. As of December 31, 2020 and 2019, credit risk did not materially change the fair value of our foreign currency forward contracts. The following table summarizes the effect of foreign currency forward contracts designated as hedging instruments in our consolidated statements of income:
Interest Rate Contracts - Hedging Instruments We have entered into interest rate lock contracts or interest rate swap contracts on certain borrowing transactions to manage our exposure to interest rate changes and to reduce our overall cost of borrowing. Interest Rate Swap Contracts In connection with the issuance of our 2.90% Senior Notes due September 15, 2020, we entered into interest rate swaps with an aggregate notional amount of $675.0 million, which were set to expire on September 15, 2020. The interest rate swap contracts were designated as hedges of the fair value changes in our 2.90% Senior Notes attributable to changes in interest rates. The carrying value of our 2.90% Senior Notes as of December 31, 2019, included approximately $2.3 million related to changes in the fair value of these interest rate swap contracts. In May 2020 we settled our interest rate swap contracts, in conjunction with our early redemption of our 2.90% Senior Notes, resulting in a gain of approximately $3.3 million for the year ended December 31, 2020, which was recorded as a component of interest expense in our consolidated statements of income. Net Investment Hedges - Hedging Instruments In February 2012 we entered into a joint venture agreement with Samsung BioLogics establishing an entity, Samsung Bioepis, to develop, manufacture and market biosimilar products. In June 2018 we exercised our option under our joint venture agreement to increase our ownership percentage in Samsung Bioepis from approximately 5.0% to approximately 49.9%. The share purchase transaction was completed in November 2018 and, upon closing, we paid 759.5 billion South Korean won ($676.6 million) to Samsung BioLogics. Our investment in the equity of Samsung Bioepis is exposed to the currency fluctuations in the South Korean won. In order to mitigate these currency fluctuations between the U.S. dollar and South Korean won, we have entered into foreign currency forward contracts. Foreign currency forward contracts in effect as of December 31, 2020, had remaining durations of 10 months. These contracts have been designated as net investment hedges. We recognize changes in the spot exchange rate in accumulated other comprehensive income (loss). The pre-tax portion of the fair value of these foreign currency forward contracts that were included in accumulated other comprehensive income (loss) in total equity reflected net losses of $21.2 million and $1.5 million as of December 31, 2020 and 2019, respectively. We exclude fair value changes related to the forward rate from our hedging relationship and will amortize the forward points in other income (expense), net in our consolidated statements of income over the term of the contract. The pre-tax portion of the fair value of the forward points that were included in accumulated other comprehensive income (loss) in total equity reflected gains of $0.2 million and $2.9 million as of December 31, 2020 and 2019, respectively. The following table summarizes the effect of our net investment hedges in our consolidated financial statements:
For additional information on our collaboration arrangements with Samsung Bioepis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Foreign Currency Forward Contracts - Other Derivative Instruments We also enter into other foreign currency forward contracts, usually with durations of one month or less, to mitigate the foreign currency risk related to certain balance sheet positions. We have not elected hedge accounting for these transactions. The aggregate notional amount of these outstanding foreign currency contracts was $1,158.0 million and $793.8 million as of December 31, 2020 and 2019, respectively. Net gains of $30.1 million, net losses of $5.9 million and net gains of $2.0 million related to these contracts were recorded as a component of other income (expense), net for the years ended December 31, 2020, 2019 and 2018, respectively. Summary of Derivative Instruments While certain of our derivative instruments are subject to netting arrangements with our counterparties, we do not offset derivative assets and liabilities in our consolidated balance sheets. The amounts in the table below would not be substantially different if the derivative assets and liabilities were offset. The following table summarizes the fair value and presentation in our consolidated balance sheets of our outstanding derivative instruments, including those designated as hedging instruments:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Property, Plant and Equipment |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Property, Plant and Equipment [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Property, Plant and Equipment | Property, Plant and Equipment Property, plant and equipment are recorded at historical cost, net of accumulated depreciation. Components of property, plant and equipment, net are summarized as follows:
Depreciation expense totaled $201.9 million, $190.6 million and $269.4 million for the years ended December 31, 2020, 2019 and 2018, respectively. For the years ended December 31, 2020, 2019 and 2018, we capitalized interest costs related to construction in progress totaling approximately $65.2 million, $68.8 million and $54.0 million, respectively. Solothurn, Switzerland Manufacturing Facility In order to support our future growth and drug development pipeline, we are building a large-scale biologics manufacturing facility in Solothurn, Switzerland. We expect this facility to be partially operational during the first half of 2021. Upon completion, this facility will include 393,000 square feet related to a large-scale biologics manufacturing facility, 290,000 square feet of warehouse, utilities and support space and 51,000 square feet of administrative space. As of December 31, 2020 and 2019, we had approximately $1.8 billion and $1.9 billion, respectively, capitalized as construction in progress related to this facility. For the year ended December 31, 2020, we placed approximately $256.8 million of fixed assets in service related to this facility. Divestiture of Hillerød, Denmark Manufacturing Operations In August 2019 we completed the sale of all of the outstanding shares of our subsidiary that owned our biologics manufacturing operations in Hillerød, Denmark to FUJIFILM. This transaction included $631.5 million of property, plant and equipment, which was primarily comprised of $312.5 million for buildings and $287.3 million for machinery and equipment. For additional information on the divestiture of our Hillerød, Denmark manufacturing operations, please read Note 3, Divestitures, to these consolidated financial statements.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Leases |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Leases [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Leases | Leases We lease real estate, including laboratory and office space, and certain equipment. Our leases have remaining lease terms ranging from less than one year to ten years. Certain leases include one or more options to renew, exercised at our sole discretion, with renewal terms that can extend the lease term from one year to six years. In addition, we sublease certain real estate to third parties. Our sublease portfolio consists of operating leases, with remaining lease terms ranging from four years to eight years. Our subleases do not include an option to renew as they are coterminous with our operating leases. All of our leases qualify as operating leases. The following table summarizes the presentation in our consolidated balance sheets of our operating leases:
The following table summarizes the effect of lease costs in our consolidated statements of income:
Variable lease cost primarily related to operating expenses, taxes and insurance associated with our operating leases. As these costs are generally variable in nature, they are not included in the measurement of the operating lease asset and related lease liability. The minimum lease payments for the next five years and thereafter is expected to be as follows:
The weighted average remaining lease term and weighted average discount rate of our operating leases are as follows:
Supplemental disclosure of cash flow information related to our operating leases included in cash flows provided by operating activities in our consolidated statements of cash flows is as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Indebtedness |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Debt Disclosure [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indebtedness | Indebtedness Our indebtedness is summarized as follows:
(1) Our 2.900% Senior Notes due September 15, 2020, were redeemed in full in May 2020 using the net proceeds from the issuance on April 30, 2020, of our senior unsecured notes for an aggregate principal amount of $3.0 billion, as discussed below. 2020 Senior Notes On April 30, 2020, we issued senior unsecured notes for an aggregate principal amount of $3.0 billion (2020 Senior Notes) as of December 31, 2020, consisting of the following: •$1.5 billion aggregate principal amount of 2.25% Senior Notes due May 1, 2030, valued at 99.973% of par; and •$1.5 billion aggregate principal amount of 3.15% Senior Notes due May 1, 2050, valued at 99.174% of par. Our 2020 Senior Notes are senior unsecured obligations and may be redeemed at our option at any time at 100.0% of the principal amount plus accrued interest and, until a specified period before maturity, a specified make-whole amount. Our 2020 Senior Notes contain a change-of-control provision that, under certain circumstances, may require us to purchase our 2020 Senior Notes at a price equal to 101.0% of the principal amount plus accrued and unpaid interest to the date of repurchase. We incurred approximately $24.4 million of costs associated with this offering which have been recorded as a reduction to the carrying amount of the debt on our consolidated balance sheet. These costs will be amortized as additional interest expense using the effective interest rate method over the period from issuance through maturity. The discounts will be amortized as additional interest expense over the period from issuance through maturity using the effective interest rate method. Interest on our 2020 Senior Notes is payable May 1 and November 1 of each year, commencing November 1, 2020. 2015 Senior Notes The following is a summary of our currently outstanding senior secured notes issued in 2015 (the 2015 Senior Notes) as of December 31, 2020: •$1.0 billion aggregate principal amount of 3.625% Senior Notes due September 15, 2022, valued at 99.920% of par; •$1.75 billion aggregate principal amount of 4.05% Senior Notes due September 15, 2025, valued at 99.764% of par; and •$1.75 billion aggregate principal amount of 5.20% Senior Notes due September 15, 2045, valued at 99.294% of par. The original costs associated with this offering of approximately $47.5 million have been recorded as a reduction to the carrying amount of the debt in our consolidated balance sheets. These costs along with the discounts will be amortized as additional interest expense using the effective interest rate method over the period from issuance through maturity. Our 2015 Senior Notes are senior unsecured obligations and may be redeemed at our option at any time at 100.0% of the principal amount plus accrued interest and a specified make-whole amount. Our 2015 Senior Notes contain a change of control provision that may require us to purchase the notes at a price equal to 101.0% of the principal amount plus accrued and unpaid interest to the date of purchase under certain circumstances. On September 15, 2015, we issued $1.5 billion aggregate principal amount of 2.90% Senior Notes due September 15, 2020, at 99.792% of par. Our 2.90% Senior Notes were senior unsecured obligations. In connection with the 2.90% Senior Notes, we entered into interest rate swap contracts where we received a fixed rate and paid a variable rate. In May 2020 we used the net proceeds from the sale of our 2020 Senior Notes to redeem our 2.90% Senior Notes prior to their maturity and recognized a net pre-tax charge of $9.4 million upon the extinguishment of these notes. This charge, which was recognized in interest expense in other income (expense), net in our consolidated statements of income for the year ended December 31, 2020, reflects the payment of a $12.7 million early call premium and the write off of remaining unamortized original debt issuance costs and discount balances, partially offset by a $3.3 million gain related to the settlement of the associated interest rate swap contracts. For additional information on our interest rate contracts, please read Note 9, Derivative Instruments, to these consolidated financial statements. 2020 Credit Facility In January 2020 we entered into a $1.0 billion, five-year senior unsecured revolving credit facility under which we are permitted to draw funds for working capital and general corporate purposes. The terms of the revolving credit facility include a financial covenant that requires us not to exceed a maximum consolidated leverage ratio. This revolving credit facility replaced the revolving credit facility that we entered into in August 2015. As of December 31, 2020, we had no outstanding borrowings and were in compliance with all covenants under this facility. Debt Maturity The total gross payments due under our debt arrangements are as follows:
The fair value of our debt is disclosed in Note 7, Fair Value Measurements, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Equity |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equity [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equity | Equity Preferred Stock We have 8.0 million shares of Preferred Stock authorized, of which 1.75 million shares are authorized as Series A, 1.0 million shares are authorized as Series X junior participating and 5.25 million shares are undesignated. Shares may be issued without a vote or action of shareholders from time to time in classes or series with the designations, powers, preferences and the relative, participating, optional or other special rights of the shares of each such class or series and any qualifications, limitations or restrictions thereon as set forth in the instruments governing such shares. Any such Preferred Stock may rank prior to common stock as to dividend rights, liquidation preference or both, and may have full or limited voting rights and may be convertible into shares of common stock. No shares of Preferred Stock were issued and outstanding during 2020, 2019 and 2018. Common Stock The following table describes the number of shares authorized, issued and outstanding of our common stock as of December 31, 2020, 2019 and 2018:
Share Repurchases In October 2020 our Board of Directors authorized a program to repurchase up to $5.0 billion of our common stock (2020 Share Repurchase Program). Our 2020 Share Repurchase Program does not have an expiration date. All share repurchases under our 2020 Share Repurchase Program will be retired. Under our 2020 Share Repurchase Program, we repurchased and retired approximately 1.6 million shares of our common stock at a cost of approximately $400.0 million during the year ended December 31, 2020. In December 2019 our Board of Directors authorized a program to repurchase up to $5.0 billion of our common stock (December 2019 Share Repurchase Program), which was completed as of September 30, 2020. All shares repurchased under our December 2019 Share Repurchase Program were retired. Under our December 2019 Share Repurchase Program, we repurchased and retired approximately 16.7 million shares of our common stock at a cost of approximately $5.0 billion during the year ended December 31, 2020. In March 2019 our Board of Directors authorized a program to repurchase up to $5.0 billion of our common stock (March 2019 Share Repurchase Program), which was completed as of March 31, 2020. All shares repurchased under our March 2019 Share Repurchase Program were retired. Under our March 2019 Share Repurchase Program, we repurchased and retired approximately 4.1 million and 14.7 million shares of our common stock at a cost of approximately $1.3 billion and $3.7 billion during the years ended December 31, 2020 and 2019, respectively. In August 2018 our Board of Directors authorized a program to repurchase up to $3.5 billion of our common stock (2018 Share Repurchase Program), which was completed as of June 30, 2019. All share repurchases under our 2018 Share Repurchase Program were retired. Under our 2018 Share Repurchase Program, we repurchased and retired approximately 8.9 million and 4.3 million shares of our common stock at a cost of approximately $2.1 billion and $1.4 billion during the years ended December 31, 2019 and 2018, respectively. In July 2016 our Board of Directors authorized a program to repurchase up to $5.0 billion of our common stock (2016 Share Repurchase Program), which was completed as of June 30, 2018. All share repurchases under our 2016 Share Repurchase Program were retired. Under our 2016 Share Repurchase Program, we repurchased and retired approximately 10.5 million shares of common stock at a cost of approximately $3.0 billion during the year ended December 31, 2018. Amounts paid to repurchase shares in excess of their par value are allocated between additional paid-in capital and retained earnings, with payments in excess of our additional paid-in-capital balance recorded as a reduction to retained earnings. Accumulated Other Comprehensive Income (Loss) The following tables summarize the changes in accumulated other comprehensive income (loss), net of tax by component:
The following table summarizes the amounts reclassified from accumulated other comprehensive income:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Earnings per Share |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Earnings Per Share [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Earnings per Share | Earnings per Share Basic and diluted earnings per share are calculated as follows:
Amounts excluded from the calculation of net income per diluted share because their effects were anti-dilutive were insignificant. Earnings per share for the years ended December 31, 2020, 2019 and 2018, reflects the repurchase of approximately 22.4 million shares, 23.6 million shares and 14.8 million shares of our common stock, respectively, under our share repurchase programs. For additional information on our share repurchase programs, please read Note 13, Equity, to these consolidated financial statements.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Share-Based Payments |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based Payment Arrangement [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share-Based Payments | Share-Based Payments Share-Based Compensation Expense The following table summarizes share-based compensation expense included in our consolidated statements of income:
The following table summarizes share-based compensation expense associated with each of our share-based compensation programs:
As of December 31, 2020, unrecognized compensation cost related to unvested share-based compensation was approximately $207.6 million, net of estimated forfeitures. We expect to recognize the cost of these unvested awards over a weighted-average period of 1.9 years. Share-Based Compensation Plans We have three share-based compensation plans pursuant to which awards are currently being made: (i) the Biogen Inc. 2006 Non-Employee Directors Equity Plan (2006 Directors Plan); (ii) the Biogen Inc. 2017 Omnibus Equity Plan (2017 Omnibus Equity Plan); and (iii) the Biogen Inc. 2015 Employee Stock Purchase Plan (2015 ESPP). Directors Plan In May 2006 our shareholders approved the 2006 Directors Plan for share-based awards to our directors. Awards granted from the 2006 Directors Plan may include stock options, shares of restricted stock, RSUs, stock appreciation rights and other awards in such amounts and with such terms and conditions as may be determined by a committee of our Board of Directors, subject to the provisions of the 2006 Directors Plan. We have reserved a total of 1.6 million shares of common stock for issuance under the 2006 Directors Plan. The 2006 Directors Plan provides that awards other than stock options and stock appreciation rights will be counted against the total number of shares reserved under the plan in a 1.5-to-1 ratio. In June 2015 our shareholders approved an amendment to extend the term of the 2006 Directors Plan until June 2025. Omnibus Plan In June 2017 our shareholders approved the 2017 Omnibus Equity Plan for share-based awards to our employees. Awards granted from the 2017 Omnibus Equity Plan may include stock options, shares of restricted stock, RSUs, performance shares, stock appreciation rights and other awards in such amounts and with such terms and conditions as may be determined by a committee of our Board of Directors, subject to the provisions of the 2017 Omnibus Equity Plan. Shares of common stock available for grant under the 2017 Omnibus Equity Plan consist of 8.0 million shares reserved for this purpose, plus shares of common stock that remained available for grant under the Biogen Idec Inc. 2008 Omnibus Equity Plan (2008 Omnibus Equity Plan) as of June 7, 2017, or that could again become available for grant if outstanding awards under the 2008 Omnibus Equity Plan as of June 7, 2017, are cancelled, surrendered or terminated in whole or in part. The 2017 Omnibus Equity Plan provides that awards other than stock options and stock appreciation rights will be counted against the total number of shares available under the plan in a 1.5-to-1 ratio. We have not made any awards pursuant to the 2008 Omnibus Equity Plan since our shareholders approved the 2017 Omnibus Equity Plan, and do not intend to make any awards pursuant to the 2008 Omnibus Equity Plan in the future, except that unused shares under the 2008 Omnibus Equity Plan have been carried over for use under the 2017 Omnibus Equity Plan. Stock Options We currently do not grant stock options to our employees or directors. Outstanding stock options previously granted to our employees and directors generally have a 10-year term and vest over a period of between and four years, provided the individual continues to serve at Biogen through the vesting dates. Options granted under all plans are exercisable at a price per share not less than the fair market value of the underlying common stock on the date of grant. As of December 31, 2020, all outstanding options were exercisable. The following table summarizes our stock option activity:
The total intrinsic values of options exercised in 2020, 2019 and 2018 totaled $2.9 million, $4.2 million and $4.0 million, respectively. The following table summarizes the amount of tax benefit realized for stock options and cash received from the exercise of stock options:
Market Stock Units (MSUs) MSUs awarded to employees prior to 2014 vested in four equal annual increments beginning on the first anniversary of the grant date. Participants may ultimately earn between zero and 150.0% of the target number of units granted based on actual stock performance. MSUs awarded to employees in 2014 and thereafter vest in three equal annual increments beginning on the first anniversary of the grant date, and participants may ultimately earn between zero and 200.0% of the target number of units granted based on actual stock performance. The vesting of these awards is subject to the respective employee’s continued employment. The number of MSUs granted represents the target number of units that are eligible to be earned based on the attainment of certain market-based criteria involving our stock price. The number of MSUs earned is calculated at each annual anniversary from the date of grant over the respective vesting periods, resulting in multiple performance periods. Accordingly, additional MSUs may be issued or currently outstanding MSUs may be cancelled upon final determination of the number of awards earned. The following table summarizes our MSU activity:
(1) MSUs granted during 2020 include awards granted in conjunction with our annual awards made in February 2020 and MSUs granted in conjunction with the hiring of employees. These grants reflect the target number of shares eligible to be earned at the time of grant. MSUs granted in 2020 also reflect an adjustment based upon the final performance multiplier in relation to shares granted in 2019, 2018 and 2017. We value grants of MSUs using a lattice model with a Monte Carlo simulation. This valuation methodology utilizes several key assumptions, the 30 calendar day average closing stock price on the date of grant for MSUs, expected volatility of our stock price, risk-free rates of return and expected dividend yield. The assumptions used in our valuation are summarized as follows:
The fair values of MSUs vested in 2020, 2019 and 2018 totaled $26.9 million, $32.5 million and $26.9 million, respectively. Cash Settled Performance Units (CSPUs) CSPUs awarded to employees vest in three equal annual increments beginning on the first anniversary of the grant date. The vesting of these awards is subject to the respective employee’s continued employment with such awards settled in cash. The number of CSPUs granted represents the target number of units that are eligible to be earned based on the attainment of certain performance measures established at the beginning of the performance period, which ends on December 31 of each year. Participants may ultimately earn between zero and 200.0% of the target number of units granted based on the degree of actual performance metric achievement. Accordingly, additional CSPUs may be issued or currently outstanding CSPUs may be cancelled upon final determination of the number of units earned. CSPUs are classified as liability awards and will be settled in cash based on the 30 calendar day average closing stock price through each vesting date, once the actual vested and earned number of units is known. Since no shares are issued, these awards do not dilute equity. The following table summarizes our CSPU activity:
The cash paid in settlement of CSPUs vested in 2020, 2019 and 2018 totaled $3.8 million, $10.6 million and $15.1 million, respectively. Performance-vested Restricted Stock Units (PUs) PUs are granted to certain employees in the form of RSUs that may be settled in cash or shares of our common stock at the sole discretion of the Compensation and Management Development Committee of our Board of Directors. These awards are structured and accounted for the same way as the CSPUs, and vest in three equal annual increments beginning on the first anniversary of the grant date. The number of PUs granted represents the target number of units that are eligible to be earned based on the attainment of certain performance measures established at the beginning of the performance period, which ends on December 31 of each year. Participants may ultimately earn between zero and 200.0% of the target number of units granted based on the degree of actual performance metric achievement. Accordingly, additional PUs may be issued or currently outstanding PUs may be cancelled upon final determination of the number of units earned. PUs settling in cash are based on the 30 calendar day average closing stock price through each vesting date once the actual vested and earned number of units is known. The following table summarizes our PU activity:
All PUs that vested in 2020, 2019 and 2018 were settled in cash totaling $3.4 million, $10.4 million and $17.0 million, respectively. Performance Stock Units (PSUs) PSUs Settled in Stock During the first quarter of 2018 we began granting awards for performance-vested RSUs that will settle in stock. PSUs awarded to employees have a three-year performance period and vest on the third anniversary of the grant date. The vesting of these awards is subject to the respective employee’s continued employment. The number of PSUs granted represents the target number of units that are eligible to be earned based on the achievement of cumulative three-year performance measures established at the beginning of the performance period, which ends on December 31 of the third year of the performance period. Participants may ultimately earn between zero and 200.0% of the target number of PSUs granted based on the degree of achievement of the applicable performance metric. Accordingly, additional PSUs may be issued or currently outstanding PSUs may be cancelled upon final determination of the number of units earned. The following table summarizes our PSUs that settle in stock activity:
(1) PSUs settled in stock granted in 2020 include awards granted in conjunction with our annual awards made in February 2020 and PSUs granted in conjunction with the hiring of employees. These grants reflect the target number of shares eligible to be earned at the time of grant. PSUs Settled in Cash During the first quarter of 2018 we began granting awards for performance-vested restricted stock units that will settle in cash. PSUs awarded to employees have performance periods and vest on the third anniversary of the grant date. The vesting of these awards is subject to the respective employee’s continued employment. The number of PSUs granted represents the target number of units that are eligible to be earned based on the achievement of annual performance measures established when the performance objectives are defined, which will be at the beginning of each year and will end on December 31 of such year. Participants may ultimately earn between zero and 200.0% of the target number of PSUs granted based on the degree of achievement of the applicable performance metric. Accordingly, additional PSUs may be issued or currently outstanding PSUs may be cancelled upon final determination of the number of units earned. PSUs are classified as liability awards and will be settled in cash based on the 30 calendar day average closing stock price through the vesting date, once the actual vested and earned number of PSUs is determined. Since no shares are issued, these awards do not dilute equity. The following table summarizes our PSUs that settle in cash activity:
(1) PSUs settled in cash granted in 2020 include awards granted in conjunction with our annual awards made in February 2020 and PSUs granted in conjunction with the hiring of employees. These grants reflect the target number of shares eligible to be earned at the time of grant. Time-Vested Restricted Stock Units (RSUs) RSUs awarded to employees generally vest no sooner than one-third per year over three years on the anniversary of the date of grant, or upon the third anniversary of the date of the grant, provided the employee remains continuously employed with us, except as otherwise provided in the plan. Shares of our common stock will be delivered to the employee upon vesting, subject to payment of applicable withholding taxes. RSUs awarded to directors for service on our Board of Directors vest on the first anniversary of the date of grant, provided in each case that the director continues to serve on our Board of Directors through the vesting date. Shares of our common stock will be delivered to the director upon vesting and are not subject to any withholding taxes. The following table summarizes our RSU activity:
(1) RSUs granted in 2020 primarily represent RSUs granted in conjunction with our annual awards made in February 2020 and awards made in conjunction with the hiring of new employees. RSUs granted in 2020 also include approximately 10,000 RSUs granted to our Board of Directors. RSUs granted in 2019 and 2018 had weighted average grant date fair values of $304.44 and $316.32, respectively. The fair values of RSUs vested in 2020, 2019 and 2018 totaled $140.5 million, $131.5 million and $111.7 million, respectively. Employee Stock Purchase Plan (ESPP) In June 2015 our shareholders approved the 2015 ESPP. The maximum aggregate number of shares of our common stock that may be purchased under the 2015 ESPP is 6.2 million. The following table summarizes our ESPP activity:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Income Taxes |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income Tax Disclosure [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income Taxes | Income Taxes Income Tax Expense Income before income tax expense and the income tax expense consist of the following:
2017 Tax Act The Tax Cuts and Jobs Act of 2017 (2017 Tax Act) resulted in significant changes to the U.S. corporate income tax system. These changes include a federal statutory rate reduction from 35.0% to 21.0%, the elimination or reduction of certain domestic deductions and credits and limitations on the deductibility of interest expense and executive compensation. The 2017 Tax Act also transitions international taxation from a worldwide system to a modified territorial system, which has the effect of subjecting certain earnings of our foreign subsidiaries and collaborations to immediate U.S. taxation as GILTI or Subpart F income, includes a one-time mandatory deemed repatriation tax on accumulated foreign subsidiaries' previously untaxed foreign earnings (the Transition Toll Tax) and base erosion prevention measures on U.S. earnings and reduces the effective tax rate on income that comes from U.S. exports, called Foreign Derived Intangible Income. These changes became effective in 2018. During the year ended December 31, 2018, we recognized a net reduction of $34.6 million in our estimated Transition Toll Tax, an expense of $12.7 million to remeasure our deferred tax balances, an expense of $135.8 million related to establishing deferred taxes for GILTI and an expense of $11.0 million to reflect other aspects of the 2017 Tax Act. Transition Toll Tax The 2017 Tax Act eliminated the deferral of U.S. income tax on the historical unrepatriated earnings by imposing the Transition Toll Tax. The Transition Toll Tax was assessed on our share of our foreign corporations' accumulated foreign earnings that were not previously taxed. Earnings in the form of cash and cash equivalents were taxed at a rate of 15.5% and all other earnings were taxed at a rate of 8.0%. As of December 31, 2020 and 2019, we have accrued income tax liabilities of $697.0 million under the Transition Toll Tax. Of the amounts accrued as of December 31, 2020, $62.0 million is expected to be paid within one year. The Transition Toll Tax will be paid over an eight--year period, which started in 2018, and does not accrue interest. Unremitted Earnings At December 31, 2020, we considered our earnings not to be permanently reinvested outside the U.S. and therefore recorded deferred tax liabilities associated with an estimate of the total withholding taxes expected as a result of our repatriation of earnings. Other than for earnings, we are permanently reinvested for book/tax basis differences of approximately $1.5 billion as of December 31, 2020, primarily arising through the impacts of purchase accounting. These permanently reinvested basis differences could reverse through sales of the foreign subsidiaries, as well as various other events, none of which were considered probable as of December 31, 2020. The residual U.S. tax liability, if these differences reverse, would be between $300.0 million and $400.0 million as of December 31, 2020. Coronavirus Aid, Relief and Economic Security Act In response to the COVID-19 pandemic, the Coronavirus Aid, Relief and Economic Security Act (CARES Act) was signed into law in the U.S. in March 2020. The CARES Act adjusted a number of provisions of the tax code, including the calculation and eligibility of certain deductions and the treatment of net operating losses and tax credits. The enactment of the CARES Act did not result in any material adjustments to our income tax provision for the year ended December 31, 2020, or to our net deferred tax assets as of December 31, 2020. TECFIDERA In June 2020 and September 2020 judgments were entered in favor of the defendants in the patent infringement proceedings relating to TECFIDERA Orange-Book listed patents pursuant to the Drug Price Competition and Patent Term Restoration Act of 1984, commonly known as the Hatch-Waxman Act, in West Virginia and Delaware. We have appealed the judgments in both actions. For additional information, please read Note 20, Litigation, to these consolidated financial statements. Multiple TECFIDERA generic entrants are now in the U.S. market and have deeply discounted prices compared to TECFIDERA. The generic competition for TECFIDERA significantly reduced our TECFIDERA revenues during the year ended December 31, 2020, and is expected to have a substantial negative impact on our TECFIDERA revenues for as long as there is generic competition. As of December 31, 2020, we have assessed the realizability of our deferred tax assets that are dependent on future expected sales of TECFIDERA in the U.S. and reduced the net value of certain deferred tax assets by approximately $1.7 billion and reduced the net value of deferred tax liabilities associated with GILTI and tax credits by approximately $1.6 billion. For the year ended December 31, 2020, the income tax expense associated with these reductions was approximately $90.3 million. Deferred Tax Assets and Liabilities Significant components of our deferred tax assets and liabilities are summarized as follows:
The change in the valuation allowance between December 31, 2020 and 2019, was primarily related to the establishment of a valuation allowance against certain deferred tax assets, the realization of which is dependent on future sales of TECFIDERA in the U.S., as discussed above. In addition to deferred tax assets and liabilities, we have recorded deferred charges related to intra-entity sales of inventory. As of December 31, 2020 and 2019, the total deferred charges were $142.2 million and $243.8 million, respectively. Tax Rate A reconciliation between the U.S. federal statutory tax rate and our effective tax rate is summarized as follows:
Changes in Tax Rate For the year ended December 31, 2020, our effective tax rate was primarily increased by the income tax expense related to the establishment of a valuation allowance against certain deferred tax assets, the realization of which is dependent on future sales of TECFIDERA in the U.S., as discussed above, and partially offset by the benefit recognized on the effective settlement of certain tax matters. Additionally, our 2019 effective tax rate benefited from an internal reorganization of certain intellectual property rights and the enactment of a new taxing regime in the country and certain cantons of Switzerland, which we refer to as Swiss Tax Reform, partially offset by tax expense related to the divestiture of our Hillerød, Denmark manufacturing operations. Although we recognized a loss on the divestiture of our Hillerød, Denmark manufacturing operations, the divestiture required us to write-off certain deferred tax assets and resulted in a taxable gain in certain jurisdictions. For additional information on the divestiture of our Hillerød, Denmark manufacturing operations, please read Note 3, Divestitures, to these consolidated financial statements. As a result of the 2019 internal reorganization of certain intellectual property rights, we recorded a deferred tax asset of $754.1 million and a deferred tax liability of $603.3 million as of December 31, 2019. For the year ended December 31, 2019, as compared to 2018, the decrease in our effective tax rate was primarily due to the combination of the internal reorganization of certain intellectual property rights and the impact of the Swiss Tax Reform. This decrease was partially offset by a $68.9 million tax expense related to the divestiture of our subsidiary that owned our Hillerød, Denmark manufacturing operations. We also had a higher effective tax rate in 2018 resulting from the unfavorable effects of the 2017 Tax Act and our sale of inventory, the tax effect of which had been included within prepaid taxes at January 1, 2018, at a higher effective tax rate than the 2018 statutory tax rate. Tax Attributes As of December 31, 2020, we had net operating losses and general business credit carry forwards for U.S. federal income tax purposes of approximately $0.6 million and $1.3 million, respectively, which begin to expire in 2026. For U.S. state income tax purposes, we had research and investment credit carry forwards of approximately $142.4 million that begin to expire in 2022. For foreign income tax purposes, we had $18.0 billion of Swiss federal net operating loss carryforwards that begin to expire in 2025 and $16.8 billion of Swiss cantonal net operating loss carryforwards that begin to expire in 2025. In assessing the realizability of our deferred tax assets, we have considered whether it is more likely than not that some portion or all of the deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. In making this determination, under the applicable financial reporting standards, we are allowed to consider the scheduled reversal of deferred tax liabilities, projected future taxable income and tax planning strategies. Based upon the level of historical taxable income and income tax liability and projections for future taxable income over the periods in which the deferred tax assets are utilizable, we believe it is more likely than not that we will realize the net benefits of the deferred tax assets of our wholly owned subsidiaries, net of the recorded valuation allowance. In the event that actual results differ from our estimates or we adjust our estimates in future periods, we may need to adjust or establish a valuation allowance, which could materially impact our consolidated financial position and results of operations. Accounting for Uncertainty in Income Taxes A reconciliation of the beginning and ending amount of our unrecognized tax benefits is summarized as follows:
Our 2020 activity reflects the impact of the effective settlement of certain tax matters. We and our subsidiaries are routinely examined by various taxing authorities. We file income tax returns in various U.S. states and in U.S. federal and other foreign jurisdictions. With few exceptions, we are no longer subject to U.S. federal tax examination for years before 2017 or state, local or non-U.S. income tax examinations for years before 2012. The U.S. Internal Revenue Service and other national tax authorities routinely examine our intercompany transfer pricing with respect to intellectual property related transactions and it is possible that they may disagree with one or more positions we have taken with respect to such valuations. Included in the balance of unrecognized tax benefits as of December 31, 2020, 2019 and 2018, are $68.8 million, $122.7 million and $109.1 million (net of the federal benefit on state issues), respectively, of unrecognized tax benefits that, if recognized, would affect the effective income tax rate in future periods. We recognize potential interest and penalties related to unrecognized tax benefits in income tax expense. In 2020, 2019 and 2018 we recognized a net interest and penalty expense of $1.0 million, $4.7 million and $2.2 million, respectively. We have accrued $21.2 million and $20.0 million for the payment of interest and penalties as of December 31, 2020 and 2019, respectively. Federal and State Uncertain Tax Positions It is reasonably possible that we will adjust the value of our uncertain tax positions related to certain transfer pricing, collaboration matters and other issues as we receive additional information from various taxing authorities, including reaching settlements with such authorities. We estimate that it is reasonably possible that our gross unrecognized tax benefits, exclusive of interest, could decrease by up to approximately $25.0 million in the next 12 months as a result of various audit closures, settlements and expiration of the statute of limitations. Accounting for Bioverativ Spin-off On February 1, 2017, in connection with the spin-off of our hemophilia business, we distributed all of the then outstanding shares of Bioverativ common stock to Biogen shareholders pursuant to a separation agreement. In March 2018 Bioverativ was acquired by Sanofi S.A. (Sanofi) and is now an indirect wholly-owned subsidiary of Sanofi. The spin-off of our hemophilia business was intended to qualify for tax-free treatment to Biogen and its shareholders under the Internal Revenue Code. Our 2017 tax return position remains open to audit. Bioverativ and Sanofi agreed to indemnify us for certain potential liabilities that may arise.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Other Consolidated Financial Statement Detail |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Organization, Consolidation and Presentation of Financial Statements [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Consolidated Financial Statement Detail | Other Consolidated Financial Statement Detail Supplemental Cash Flow Information Supplemental disclosure of cash flow information for the years ended December 31, 2020, 2019 and 2018, is as follows:
Non-cash Operating, Investing and Financing Activity In the fourth quarter of 2018 we accrued $300.0 million upon reaching $20.0 billion in total cumulative sales of FUMADERM and TECFIDERA (together, the Fumapharm Products), which was paid in the first quarter of 2019. These amounts, net of tax benefit, were accounted for as increases to goodwill in accordance with the accounting standard applicable to business combinations when we acquired Fumapharm AG. In connection with the construction of our large-scale biologics manufacturing facility in Solothurn, Switzerland, we accrued charges related to processing equipment and engineering services of approximately $12.4 million and $50.0 million in our consolidated balance sheets as of December 31, 2020 and 2019, respectively. For additional information on the construction of our manufacturing facility in Solothurn, Switzerland, please read Note 10, Property, Plant and Equipment, to these consolidated financial statements. Other Income (Expense), Net Components of other income (expense), net, are summarized as follows:
Gain (loss) on investments, net, as reflected in the table above, relate to debt securities, equity securities of certain biotechnology companies, venture capital funds where the underlying investments are in equity securities of certain biotechnology companies and non-marketable equity securities. For the year ended December 31, 2020, net unrealized and realized gains on our holdings in equity securities were approximately $681.8 million and $12.1 million, respectively, compared to net unrealized and realized gains of $150.1 million and $50.0 million in 2019. The net unrealized gains recognized during the year ended December 31, 2020, primarily reflects an increase in the fair value of Denali and Sangamo common stock of approximately $703.9 million. The following table summarizes our gain (loss) on investments, net that relates to our equity securities held as of December 31, 2020, 2019 and 2018:
Accrued Expenses and Other Accrued expenses and other consists of the following:
Other Long-term Liabilities Other long-term liabilities were $1,329.6 million and $1,348.9 million as of December 31, 2020 and 2019, respectively, and include accrued income taxes totaling $709.9 million and $803.3 million, respectively.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Collaborative and Other Relationships |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Organization, Consolidation and Presentation of Financial Statements [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Collaborative and Other Relationships | Collaborative and Other RelationshipsIn connection with our business strategy, we have entered into various collaboration agreements that provide us with rights to develop, produce and market products using certain know-how, technology and patent rights maintained by our collaborative partners. Terms of the various collaboration agreements may require us to make milestone payments upon the achievement of certain product research and development objectives and pay royalties on future sales, if any, of commercial products resulting from the collaboration. Depending on the collaborative arrangement, we may record funding receivable or payable balances with our collaboration partners, based on the nature of the cost-sharing mechanism and activity within the collaboration. Our significant collaborative arrangements are discussed below. Genentech, Inc. (Roche Group) We have certain business and financial rights with respect to RITUXAN for the treatment of non-Hodgkin's lymphoma, CLL and other conditions; RITUXAN HYCELA for the treatment of non-Hodgkin's lymphoma and CLL; GAZYVA for the treatment of CLL and follicular lymphoma; OCREVUS for the treatment of PPMS and RMS; and other potential anti-CD20 therapies pursuant to our collaboration arrangements with Genentech, a wholly-owned member of the Roche Group. For purposes of this footnote, we refer to RITUXAN and RITUXAN HYCELA collectively as RITUXAN. Our collaboration arrangements will continue in effect until we mutually agree to terminate the collaboration, except that if we undergo a change in control, as defined in our collaboration agreement, Genentech has the right to present an offer to buy the rights to RITUXAN and we must either accept Genentech’s offer or purchase Genentech’s rights on the same terms as its offer. Genentech will also be deemed concurrently to have purchased our rights to OCREVUS and any other collaboration anti-CD20 products in development in exchange for a royalty as well as our rights to GAZYVA in exchange for the compensation described in the collaboration arrangement. Our collaboration with Genentech was created through a contractual arrangement and not through a joint venture or other legal entity. RITUXAN Genentech and its affiliates are responsible for the worldwide manufacture of RITUXAN as well as all development and commercialization activities as follows: U.S. We have co-exclusively licensed our rights to develop, commercialize and market RITUXAN in the U.S. Canada We have co-exclusively licensed our rights to develop, commercialize and market RITUXAN in Canada. GAZYVA The Roche Group and its sub-licensees maintain sole responsibility for the development, manufacture and commercialization of GAZYVA in the U.S. We recognize our share of the development and commercialization expenses of GAZYVA as a reduction of our share of pre-tax profits in revenues from anti-CD20 therapeutic programs. Commercialization of GAZYVA impacts our percentage of the co-promotion profits for RITUXAN, as summarized in the table below. OCREVUS In March 2017 the FDA approved OCREVUS for the treatment of RMS and PPMS. Pursuant to the terms of our collaboration arrangements with Genentech, we receive a tiered royalty on U.S. net sales from 13.5% and increasing up to 24.0% if annual net sales exceed $900.0 million. There will be a 50.0% reduction to these royalties if a biosimilar to OCREVUS is approved in the U.S. In addition, we receive a gross 3.0% royalty on net sales of OCREVUS outside the U.S., with the royalty period lasting 11 years from the first commercial sale of OCREVUS on a country-by-country basis. OCREVUS has been approved for the treatment of RMS and PPMS in the E.U. and certain other countries. The commercialization of OCREVUS does not impact the percentage of the co-promotion profits we receive for RITUXAN or GAZYVA. Genentech is solely responsible for development and commercialization of OCREVUS and funding future costs. Genentech cannot develop OCREVUS in CLL, non-Hodgkin's lymphoma or rheumatoid arthritis. OCREVUS royalty revenues were based on our estimates from third party and market research data of OCREVUS sales occurring during the corresponding period. Differences between actual and estimated royalty revenues will be adjusted for in the period in which they become known, which is generally expected to be the following quarter. Profit-sharing Formulas RITUXAN Profit Share Our current pretax co-promotion profit-sharing formula for RITUXAN provides for a 30.0% share on the first $50.0 million of co-promotion operating profits earned each calendar year. Our share of annual co-promotion profits in excess of $50.0 million varies, as summarized in the table below, upon the following events:
First Non-CLL GAZYVA FDA Approval means the FDA’s first approval of GAZYVA in an indication other than CLL. First GAZYVA Threshold Date means the earlier of (i) the date of the First Non-CLL GAZYVA FDA approval if U.S. gross sales of GAZYVA for the preceding consecutive 12-month period were at least $150.0 million or (ii) the first day of the calendar quarter after the date of the First Non-CLL GAZYVA FDA Approval that U.S. gross sales of GAZYVA within any consecutive 12-month period have reached $150.0 million. Second GAZYVA Threshold Date means the first day of the calendar quarter after U.S. gross sales of GAZYVA within any consecutive 12-month period have reached $500.0 million. The Second GAZYVA Threshold Date can be achieved regardless of whether GAZYVA has been approved in a non-CLL indication. Our share of RITUXAN pre-tax profits in the U.S. in excess of $50.0 million for the years ended December 31, 2020, 2019 and 2018, was 37.5%. In addition, should the FDA approve an anti-CD20 product other than OCREVUS or GAZYVA that is acquired or developed by Genentech and subject to the collaboration agreement, our share of the co-promotion operating profits would be between 30.0% and 37.5% based on certain events. GAZYVA Profit Share Our current pretax profit-sharing formula for GAZYVA provides for a 35.0% share on the first $50.0 million of operating profits earned each calendar year. Our share of annual profits in excess of $50.0 million varies, as summarized in the table below, upon the following events:
Our share of GAZYVA pre-tax profits in excess of $50.0 million for the years ended December 31, 2020, 2019 and 2018, was 37.5%. In November 2017 the FDA approved GAZYVA in combination with chemotherapy, followed by GAZYVA alone, for people with previously untreated advanced follicular lymphoma. Revenues from Anti-CD20 Therapeutic Programs Revenues from anti-CD20 therapeutic programs are summarized as follows:
Prior to regulatory approval, we record our share of the expenses incurred by the collaboration for the development of anti-CD20 products in research and development expense in our consolidated statements of income. After an anti-CD20 product is approved, we record our share of the development expenses related to that product as a reduction of our share of pre-tax profits in revenues from anti-CD20 therapeutic programs. Ionis Pharmaceuticals, Inc. SPINRAZA In January 2012 we entered into a collaboration and license agreement with Ionis pursuant to which we have an exclusive, worldwide license to develop and commercialize SPINRAZA for the treatment of SMA. Under our agreement with Ionis, we make royalty payments to Ionis on annual worldwide net sales of SPINRAZA using a tiered royalty rate between 11.0% and 15.0%, which are recognized in cost of sales within our consolidated statements of income. Royalty cost of sales related to sales of SPINRAZA for the years ended December 31, 2020, 2019 and 2018, totaled $286.6 million, $293.0 million and $238.0 million, respectively. 2012 Ionis Agreement In December 2012 we entered into an agreement with Ionis for the development and commercialization of up to three gene targets. Under this agreement, Ionis is responsible for global development of any product candidate through the completion of a Phase 2 trial and we will provide advice on the clinical trial design and regulatory strategy. We have an option to license the product candidate until completion of the Phase 2 trial. If we exercise our option, we will pay a license fee of up to $70.0 million to Ionis and assume global development, regulatory and commercialization responsibilities. Ionis is eligible to receive up to $130.0 million in additional milestone payments upon the achievement of certain regulatory milestones as well as royalties on future sales if we successfully develop the product candidate after option exercise. Upon entering into this agreement, we made an upfront payment of $30.0 million to Ionis and agreed to make potential additional payments, prior to licensing, of up to $10.0 million based on the development of the selected product candidate as well as a mark-up of the cost estimate of the Phase 1 and Phase 2 trials. During 2015 we recognized this $10.0 million developmental milestone upon the selection of BIIB080 (tau ASO), which is currently in Phase 1 development for the potential treatment of Alzheimer's disease. In December 2019 we exercised our option with Ionis and obtained a worldwide, exclusive, royalty-bearing license to develop and commercialize BIIB080. In connection with the option exercise, we made a payment of $45.0 million to Ionis, which was recorded as research and development expense in our consolidated statements of income. Future payments may include additional milestone payments of up to $155.0 million and royalties on future sales in the low- to mid-teens if we successfully develop the product candidate after option exercise. 2018 Ionis Agreement In June 2018 we closed a 10-year exclusive collaboration agreement with Ionis to develop novel antisense oligonucleotide (ASO) drug candidates for a broad range of neurological diseases (2018 Ionis Agreement) for a total payment of $1.0 billion, consisting of an upfront payment of $375.0 million and the purchase of approximately 11.5 million shares of Ionis common stock at a cost of $625.0 million. Upon closing, we recorded $50.9 million of the $375.0 million upfront payment as prepaid services in our consolidated balance sheets and recognized the remaining $324.1 million as research and development expense in our consolidated statements of income. The amount recorded as prepaid services represented the value of the employee resources committed to the arrangement to provide research and discovery services over the term of the agreement. The 11.5 million shares of Ionis common stock were purchased at a premium to their fair value at the transaction closing date. The premium consisted of acquiring the shares at a price above the fair value based on the trailing 10-day weighted-average close price prior to entering into the 2018 Ionis Agreement in April 2018 and the effect of certain holding period restrictions. We recorded an asset of $462.9 million in investments and other assets in our consolidated balance sheets reflecting the fair value of the Ionis common stock as of the purchase date and a charge of $162.1 million to research and development expense in our consolidated statements of income in the second quarter of 2018 reflecting the premium paid for the Ionis common stock. Our investment in Ionis common stock is remeasured each reporting period. Changes in the fair value of our investment in Ionis common stock, including the effect of the holding period restrictions, are reflected in other income (expense), net in our consolidated statements of income. For additional information on the fair value of our investment in Ionis common stock, please read Note 7, Fair Value Measurements, to these consolidated financial statements. We have the option to license therapies arising out of the 2018 Ionis Agreement and will be responsible for the development and commercialization of such therapies. We may pay development milestones to Ionis of up to $125.0 million or $270.0 million for each program, depending on the indication plus an annual license fee, as well as royalties on potential net commercial sales. During the years ended December 31, 2020 and 2019, we incurred milestones of $11.3 million and $30.0 million, respectively, related to the advancement of neurological targets identified under the 2018 Ionis Agreement. 2017 SMA Collaboration Agreement In December 2017 we entered into a collaboration agreement with Ionis to identify new ASO drug candidates for the potential treatment of SMA. Under this agreement, we have the option to license therapies arising out of this collaboration and will be responsible for their development and commercialization of such therapies. Upon entering into this agreement, we made a $25.0 million upfront payment to Ionis and we may pay Ionis up to $260.0 million in additional development and regulatory milestone payments if new drug candidates advance to marketing approval. Upon commercialization, we may also pay Ionis up to $800.0 million in additional performance-based milestone payments and tiered royalties on potential net sales of such therapies. 2013 Long-term Strategic Research Agreement In September 2013 we entered into a six-year research collaboration agreement with Ionis under which both companies collaborate to perform discovery level research and subsequent development and commercialization activities of antisense or other therapeutics for the potential treatment of neurological diseases. Under this agreement, Ionis performs research on a set of neurological targets identified within the agreement. Ionis is eligible to receive milestone payments, license fees and royalty payments for all product candidates developed through this collaboration, with the specific amount dependent upon the modality of the product candidate advanced by us under the terms of the agreement. For non-ALS antisense product candidates, Ionis is responsible for global development through the completion of a Phase 2 trial and we provide advice on the clinical trial design and regulatory strategy. For ALS antisense product candidates, we are responsible for global development, clinical trial design and regulatory strategy. We have an option to license a product candidate until completion of the Phase 2 trial. If we exercise our option, we will pay Ionis up to a $70.0 million license fee and assume global development, regulatory and commercialization responsibilities. Ionis could receive additional milestone payments upon the achievement of certain regulatory milestones of up to $130.0 million, plus additional amounts related to the cost of clinical trials conducted by Ionis under the collaboration, and royalties on future sales if we successfully develop the product candidate after option exercise. In December 2018 we exercised our option with Ionis and obtained a worldwide, exclusive, royalty-bearing license to develop and commercialize BIIB067 (tofersen), an investigational treatment for ALS with superoxide dismutase 1 (SOD1) mutations. In connection with the option exercise, we made a payment of $35.0 million to Ionis, which was recorded as research and development in our consolidated statements of income. Future payments may include potential post-licensing milestone payments of up to $55.0 million and royalties in the low- to mid-teen percentages on potential annual worldwide net sales. We are solely responsible for the costs and expenses related to the development, manufacturing and commercialization of tofersen following the option exercise. During the years ending December 31, 2020, 2019 and 2018, we incurred milestones of $28.0 million, $20.0 million and $18.0 million, respectively, related to the advancement of programs under this agreement, which were recorded as research and development expense in our consolidated statements of income. Eisai Co., Ltd. BAN2401 Collaboration We have a collaboration agreement with Eisai to jointly develop and commercialize BAN2401, a monoclonal antibody that targets amyloid beta aggregates, and elenbecestat, the oral BACE (base amyloid cleaving enzyme) inhibitor, two Eisai product candidates for the potential treatment of Alzheimer's disease (the BAN2401 Collaboration). In September 2019 we and Eisai discontinued the global Phase 3 studies of elenbecestat in early Alzheimer's disease. Eisai serves as the global operational and regulatory lead for BAN2401 and all costs, including research, development, sales and marketing expenses, are shared equally between us and Eisai. If BAN2401 receives marketing approval, we and Eisai will co-promote BAN2401 and share profits equally. In addition, the BAN2401 Collaboration provides both parties with certain rights and obligations in the event of a change in control of either party. The BAN2401 Collaboration also provided Eisai with an option to jointly develop and commercialize aducanumab (Aducanumab Option) and an option to jointly develop and commercialize one of our anti-tau monoclonal antibodies (Anti-Tau Option). In October 2017 Eisai exercised its Aducanumab Option and we entered into a new collaboration agreement for the joint development and commercialization of aducanumab (the Aducanumab Collaboration Agreement). Eisai may exercise the Anti-Tau Option after completion of the Phase 1 clinical trial of such anti-tau monoclonal antibody. If Eisai exercises its Anti-Tau Option, we will receive an upfront payment from Eisai and will be entitled to additional development and commercial milestone payments. Eisai has not yet exercised its Anti-Tau Option. A summary of development and sales and marketing expenses related to the BAN2401 Collaboration is as follows:
Aducanumab Collaboration Agreement Under the Aducanumab Collaboration Agreement, we and Eisai will co-promote aducanumab with a region-based profit split and we lead the ongoing development of aducanumab. In March 2019, based on a pre-specified futility analysis, we discontinued the global Phase 3 trials, EMERGE and ENGAGE, designed to evaluate the efficacy and safety of aducanumab in patients with early Alzheimer's disease. A new analysis of a larger dataset from these trials, conducted in scientific collaboration with the FDA, showed that the Phase 3 EMERGE study met its pre-specified primary and secondary endpoints. In the first quarter of 2019, as a result of the decision to discontinue the Phase 3 EMERGE and ENGAGE trials following the futility analysis, we accrued and subsequently paid approximately $45.0 million related to the termination of various clinical trials and research and development contracts net of the expected 45.0% Eisai reimbursement of development costs incurred under the Aducanumab Collaboration Agreement. In October 2019 we and Eisai announced that we plan to pursue regulatory approval for aducanumab in the U.S. In July 2020 we completed the submission of a BLA for the approval of aducanumab to the FDA. For the period through March 31, 2018, we were responsible for 100.0% of development costs incurred by the collaboration for the advancement of aducanumab (aducanumab development expense). For the period April 1, 2018 through December 31, 2018, Eisai reimbursed us for 15.0% of aducanumab development expense incurred and beginning January 1, 2019, is reimbursing us for 45.0% of aducanumab development expense incurred. For the year ended December 31, 2020, we recognized net profit-sharing income of $33.8 million to reflect Eisai's 45.0% share of the $75.0 million milestone expense related to the submission of the BLA for the approval of aducanumab to the FDA. Upon commercialization, both companies will co-promote aducanumab with a region-based profit split. We will receive a 55.0% share of the potential profits (losses) in the U.S., a 68.5% share of the potential profits (losses) in the European Union (E.U.) and a 20.0% share of the potential profits (losses) in Japan and Asia, excluding China and South Korea. The two companies will continue to share equally in the potential profits (losses) in rest of world markets. Sales and marketing expense incurred before commercialization are shared in proportion to the same region-based profit split that will be utilized to co-promote aducanumab. A summary of development, sales and marketing and milestone expense related to the Aducanumab Collaboration Agreement is as follows:
In addition, we and Eisai co-promote AVONEX, TYSABRI and TECFIDERA in Japan in certain settings and Eisai distributes AVONEX, TYSABRI, TECFIDERA and PLEGRIDY in India and other Asia-Pacific markets, excluding China. UCB Pharma S.A. We have a collaboration agreement with UCB Pharma S.A. (UCB) to jointly develop and commercialize dapirolizumab pegol, an anti-CD40L pegylated Fab, for the potential treatment of systemic lupus erythematosus and other future agreed indications. Either we or UCB may propose development of dapirolizumab pegol in additional indications. If the parties do not agree to add an indication as an agreed indication to the collaboration, we or UCB may, at the sole expense of the applicable party, pursue development in such excluded indication(s), subject to an opt-in right of the non-pursuing party after proof of clinical activity. All costs incurred for agreed indications, including research, development, sales and marketing expenses, are shared equally between us and UCB. Upon marketing approval, we and UCB will co-promote dapirolizumab pegol and share profits equally. A summary of development expense related to the UCB collaboration agreement is as follows:
Alkermes In November 2017 we entered into an exclusive license and collaboration agreement with Alkermes Pharma Ireland Limited, a subsidiary of Alkermes plc (Alkermes), for VUMERITY, a novel fumarate for the treatment of RMS. In October 2019 the FDA approved VUMERITY in the U.S. for the treatment of RMS. In November 2019 VUMERITY became commercially available in the U.S. Under this agreement, we received an exclusive, worldwide license to develop and commercialize VUMERITY and we pay Alkermes a royalty of 15.0% on worldwide net commercial sales of VUMERITY. Royalties payable on net commercial sales of VUMERITY are subject, under certain circumstances, to tiered minimum annual payment requirements for a period of five years following FDA approval. Alkermes is eligible to receive royalties in the high-single digits to sub-teen double digits of annual net commercial sales upon successful development and commercialization of new product candidates, other than VUMERITY, developed under the exclusive license from Alkermes. During the fourth quarter of 2019, following the FDA's approval of VUMERITY, we paid Alkermes $155.0 million in milestone payments, which were recorded in intangible assets in our consolidated balance sheets and will be amortized over the useful life of the product. For the years ended December 31, 2020, 2019 and 2018, we recorded $32.4 million, $53.5 million and $68.7 million, respectively, in research and development expense in our consolidated statements of income related to this collaboration. Alkermes currently supplies VUMERITY to us pursuant to a supply agreement. In October 2019 we entered into a new supply agreement and amended our license and collaboration agreement with Alkermes. We have elected to initiate a technology transfer and, following a transition period, to manufacture VUMERITY or have VUMERITY manufactured by a third party we have engaged in exchange for paying an increased royalty rate to Alkermes on any portion of future worldwide net commercial sales of VUMERITY that is manufactured by us or our designee. Bristol-Myers Squibb Company In June 2017 we completed an exclusive license agreement with Bristol-Myers Squibb Company (BMS) for the development and potential commercialization of BIIB092 (gosuranemab), an antibody targeting tau, the protein that forms the deposits, or tangles, in the brain associated with Alzheimer's disease. Under this agreement, we received worldwide rights to gosuranemab and are responsible for the full development and potential commercialization of gosuranemab in Alzheimer's disease and progressive supranuclear palsy (PSP). In December 2019 we announced that the Phase 2 PASSPORT study investigating gosuranemab in individuals with PSP did not meet its primary endpoint. Based on these results, we discontinued development of gosuranemab in PSP and other primary tauopathies. We will continue our ongoing Phase 2 TANGO study of gosuranemab for mild cognitive impairment due to Alzheimer's disease or mild Alzheimer's disease, given differences in disease pathology. Upon entering into this agreement, we made an upfront payment of $300.0 million to BMS and assumed all remaining obligations to the former shareholders of iPierian, Inc. (iPierian) related to BMS’s acquisition of iPierian in 2014. We may pay BMS up to $360.0 million in additional milestone payments, and potential royalties, and we may pay the former shareholders of iPierian up to $370.0 million in remaining milestone payments as well as potential royalties on net commercial sales. For the years ended December 31, 2020, 2019 and 2018, we recorded $62.4 million, $144.0 million and $97.0 million, respectively, in research and development expense in our consolidated statements of income related to this agreement. Acorda Therapeutics, Inc. In June 2009 we entered into a collaboration and license agreement with Acorda Therapeutics, Inc. (Acorda) to develop and commercialize products containing fampridine, such as FAMPYRA, in markets outside the U.S. We are responsible for all regulatory activities and the future clinical development of related products in those markets. Under this agreement, we pay tiered royalties based on the level of ex-U.S. net sales and we may pay potential milestone payments based on the successful achievement of certain regulatory and commercial milestones, which would be capitalized as intangible assets upon achievement of the milestones and amortized utilizing an economic consumption model. The next expected milestone of $15.0 million, due if ex-U.S. net sales reach $100.0 million over a period of four consecutive quarters, was recognized during the third quarter of 2020 and capitalized within intangible assets, net in our consolidated balance sheet. Royalty payments are recognized in cost of sales within our consolidated statements of income. In connection with the collaboration and license agreement, we also entered into a supply agreement with Acorda for the commercial supply of FAMPYRA. This agreement is a sublicense arrangement of an existing agreement between Acorda and Alkermes Inc., who acquired Elan Drug Technologies, the original party to the license with Acorda. For the years ending December 31, 2020, 2019 and 2018, total cost of sales related to royalties and commercial supply of FAMPYRA reflected in our consolidated statements of income were $44.5 million, $42.0 million and $36.5 million, respectively. Sage Therapeutics, Inc. In November 2020 we entered into a global collaboration and license agreement with Sage to jointly develop and commercialize zuranolone (SAGE-217) for the potential treatment of major depressive disorder, postpartum depression and other psychiatric disorders and SAGE-324 for the potential treatment of essential tremor and other neurological disorders. In connection of the closing of this transaction in December 2020 we purchased $650.0 million of Sage common stock, or approximately 6.2 million shares at approximately $104.14 per share, which are subject to transfer restrictions. We recorded an asset in investments and other assets in our consolidated balance sheets to reflect the initial fair value of the Sage common stock acquired and a charge of approximately $209.0 million to research and development expense in our consolidated statements of income to reflect the premium paid for the Sage common stock. We also made an upfront payment of $875.0 million that was recorded as research and development expense. We may also pay Sage development and commercial milestone payments that could total up to approximately $1.6 billion if all the specified milestones set forth in this agreement are achieved. Both companies will share equal responsibility and costs for development as well as profits and losses for commercialization in the U.S. Outside of the U.S., we are responsible for development and commercialization, excluding Japan, Taiwan and South Korea, with respect to zuranolone and will pay Sage potential tiered royalties in the high teens to low twenties. Denali Therapeutics Inc. In August 2020 we entered into a collaboration and license agreement with Denali to co-develop and co-commercialize Denali's small molecule inhibitors of leucine-rich repeat kinase 2 (LRRK2) for Parkinson's disease. In addition to the LRRK2 program, we also have an exclusive option to license two preclinical programs from Denali’s Transport Vehicle platform, including its Antibody Transport Vehicle (ATV): ATV enabled anti-amyloid beta program and a second program utilizing its Transport Vehicle technology. Further, we have a right of first negotiation on two additional Transport Vehicle-enabled therapeutics, should Denali decide to seek a collaboration for such programs. As part of this collaboration we purchased approximately $465.0 million of Denali common stock in September 2020, or approximately 13 million shares at approximately $34.94 per share, which are subject to transfer restrictions. We recorded an asset in investments and other assets in our consolidated balance sheets to reflect the initial fair value of the Denali common stock acquired and a charge of approximately $41.3 million to research and development expense in our consolidated statements of income to reflect the premium paid for the Denali common stock. We also made an upfront payment of $560.0 million that was recorded as research and development expense. We may also pay Denali development and commercial milestone payments that could total up to approximately $1.1 billion if the milestones related to the LRRK2 program are achieved. Under this agreement, both companies share responsibility and costs for global development based on specified percentages and we are responsible for commercialization and will pay Denali potential tiered royalties. For the year ended December 31, 2020, we recorded $8.8 million in research and development expense in our consolidated statements of income related to this collaboration. Sangamo Therapeutics, Inc. In February 2020 we entered into a collaboration and license agreement with Sangamo to develop and commercialize ST-501 for tauopathies, including Alzheimer's disease; ST-502 for synucleinopathies, including Parkinson’s disease; a third neuromuscular disease target; and up to nine additional neurological disease targets to be identified and selected within a five-year period. The companies are leveraging Sangamo’s proprietary zinc finger protein technology delivered via adeno-associated virus to modulate the expression of key genes involved in neurological diseases. In connection with the closing of this transaction in April 2020 we purchased $225.0 million of Sangamo common stock, or approximately 24 million shares at approximately $9.21 per share, which are subject to transfer restrictions. We recorded an asset in investments and other assets in our consolidated balance sheets to reflect the initial fair value of the Sangamo common stock acquired and a charge of approximately $83.0 million to research and development expense in our consolidated statements of income to reflect the premium paid for the Sangamo common stock. We also made an upfront payment of $125.0 million that was recorded as research and development expense. We may also pay Sangamo research, development, regulatory and commercial milestone payments that could total up to approximately $2.4 billion if we select all of the targets allowed under this agreement and all the specified milestones set forth in this agreement are achieved. Of this amount, up to $80.0 million relates to the selection of targets, $1.9 billion relates to the achievement of specified research, clinical development, regulatory and first commercial sale milestones and $380.0 million relates to the achievement of specified sales-based milestones if annual worldwide net sales of licensed products reach specified levels. In addition, we will pay Sangamo tiered royalties on potential net commercial sales of any products developed under this collaboration in the high single digit to double digit sub-teen percentages. For the year ended December 31, 2020, we recorded $6.4 million in research and development expense in our consolidated statements of income related to this collaboration. Other Research and Discovery Arrangements These arrangements may include the potential for future milestone payments based on the achievement of certain clinical and commercial development payable over a period of several years. Other For the years ended December 31, 2020, 2019 and 2018, we recorded $92.1 million, $77.0 million and $48.6 million, respectively, as research and development expense in our consolidated statements of income related to other research and discovery related arrangements. Samsung Bioepis Co., Ltd. Joint Venture Agreement In February 2012 we entered into a joint venture agreement with Samsung BioLogics establishing an entity, Samsung Bioepis, to develop, manufacture and market biosimilar products. Samsung BioLogics contributed 280.5 billion South Korean won (approximately $250.0 million) for an 85.0% ownership interest in Samsung Bioepis and we contributed 49.5 billion South Korean won (approximately $45.0 million) for the remaining 15.0% ownership interest. In June 2018 we exercised our option under our joint venture agreement to increase our ownership percentage in Samsung Bioepis from approximately 5.0%, which reflected the effect of previous equity financings in which we did not participate, to approximately 49.9%. The share purchase transaction was completed in November 2018 and, upon closing, we paid 759.5 billion South Korean won ($676.6 million) to Samsung BioLogics. As of December 31, 2020, our ownership percentage remained at approximately 49.9%. We recognize our share of the results of operations related to our investment in Samsung Bioepis under the equity method of accounting one quarter in arrears when the results of the entity become available, which is reflected as equity in income (loss) of investee, net of tax in our consolidated statements of income. During 2015, as our share of losses exceeded the carrying value of our initial investment, we suspended recognizing additional losses. In the first quarter of 2019 we restarted recognizing our share of Samsung Bioepis' income (losses), and we began recognizing amortization on certain basis differences resulting from our November 2018 investment. Upon investment, the equity method of accounting requires us to identify and allocate differences between the fair value of our investment and the carrying value of our interest in the underlying net assets of the investee. These basis differences are amortized over their economic life. The total basis difference was approximately $675.0 million and relates to inventory, developed technology, IPR&D and deferred tax balances. The basis differences related to inventory were amortized, net of tax, over their estimated useful lives of 1.5 years, and the basis differences related to developed technology and IPR&D for marketed products will be amortized, net of tax, over their estimated useful lives of 15 years. The former chief executive officer (the incumbent chairman of the board) and the chief financial officer of our joint venture partner, Samsung BioLogics, is currently subject to ongoing criminal proceedings that we continue to monitor. While these proceedings could impact the operations of Samsung Bioepis and its business, we have assessed the value of our investment in Samsung Bioepis and continue to believe that the fair value of the investment is in excess of its net book value. For the year ended December 31, 2020, we recognized net income on our investment of $5.3 million, reflecting our share of income totaling $45.3 million offset by amortization of basis differences totaling $40.0 million. For the year ended December 31, 2019, we recognized net losses on our investment of $79.4 million, reflecting our share of losses totaling $1.2 million and amortization of basis differences totaling $78.2 million. As of December 31, 2020 and 2019, the carrying value of our investment in Samsung Bioepis totaled 673.8 billion South Korean won ($620.2 million) and 670.8 billion South Korean won ($580.2 million), respectively, which is classified as a component of investments and other assets within our consolidated balance sheets. 2019 Transaction In December 2019 we completed a transaction with Samsung Bioepis and secured the exclusive rights to commercialize two potential ophthalmology biosimilar products, SB11, a proposed ranibizumab biosimilar referencing LUCENTIS, and SB15, a proposed aflibercept biosimilar referencing EYLEA, in major markets worldwide, including the U.S., Canada, Europe, Japan and Australia. Samsung Bioepis will be responsible for development and will supply both products to us. In connection with this transaction, we made an upfront payment of $100.0 million to Samsung Bioepis in January 2020, of which $63.0 million was recorded as research and development expense in 2019 and $37.0 million was recorded as an intangible asset in 2019. Additionally, during the third quarter of 2020, we paid Samsung Bioepis a $15.0 million development milestone, which was included in research and development expense in our consolidated statements of income. We may pay Samsung Bioepis up to $195.0 million in additional development, regulatory and sales-based milestones. We also acquired an option to extend the term of our 2013 commercial agreement for BENEPALI, IMRALDI and FLIXABI by an additional five years, subject to payment of an option exercise fee of $60.0 million, and obtained an option to acquire exclusive rights to commercialize these products in China. 2013 Commercial Agreement In December 2013 we entered into an agreement with Samsung Bioepis to commercialize, over a 10-year term, 3 anti-tumor necrosis factor (TNF) biosimilar product candidates in Europe and in the case of BENEPALI, Japan. As discussed above, we have an option to extend this agreement by an additional five years. Under this agreement, we have made upfront and clinical development milestone payments totaling $46.0 million, which were recorded as research and development expense in our consolidated statements of income as the programs they relate to had not achieved regulatory approval. We also agreed to make additional milestone payments of $25.0 million upon regulatory approval in the E.U. for each of the three anti-TNF biosimilar product candidates. IMRALDI, an adalimumab biosimilar referencing HUMIRA, FLIXABI, an infliximab biosimilar referencing REMICADE, and BENEPALI, an etanercept biosimilar referencing ENBREL, received regulatory approval in the E.U. in August 2017, May 2016 and January 2016, respectively, and we capitalized the related milestone payments totaling $75.0 million as intangible assets, net in our consolidated balance sheets. In April 2018 we and Samsung Bioepis announced an agreement with AbbVie Inc. (AbbVie) related to the commercialization of IMRALDI. Under the terms of the agreement, AbbVie granted us and Samsung Bioepis patent licenses for the use and sale of IMRALDI in Europe, on a country-by-country basis, and we make royalty payments to AbbVie on behalf of Samsung Bioepis. We began to recognize revenues on sales of IMRALDI to third parties in Europe in the fourth quarter of 2018. We reflect revenues on sales of BENEPALI, IMRALDI and FLIXABI to third parties in product revenues, net in our consolidated statements of income and record the related cost of revenues and sales and marketing expenses in our consolidated statements of income to their respective line items when these costs are incurred. Royalty payments to AbbVie on sales of IMRALDI are recognized in cost of sales within our consolidated statements of income. We share 50% of the profit or loss related to our commercial agreement with Samsung Bioepis, which is recognized in collaboration profit (loss) sharing in our consolidated statements of income. For the years ended December 31, 2020, 2019 and 2018, we recognized a net profit-sharing expense of $266.5 million, $241.6 million and $187.4 million, respectively, to reflect Samsung Bioepis' 50% sharing of the net collaboration profits. Other Services Simultaneous with the formation of Samsung Bioepis, we also entered into a technical development services agreement, a manufacturing agreement and a license agreement with Samsung Bioepis. Under the technical development services agreement, we provided Samsung Bioepis technical development and technology transfer services, which included, but were not limited to, cell culture development, purification process development, formulation development and analytical development. Under the manufacturing agreement, we manufacture clinical and commercial quantities of bulk drug substance of biosimilar products for Samsung Bioepis pursuant to contractual terms. Following the divestiture of our Hillerød, Denmark manufacturing operations in August 2019, FUJIFILM assumed responsibility for the manufacture of clinical and commercial quantities of bulk drug substance of biosimilar products for Samsung Bioepis. We no longer recognize revenues for the manufacturing completed after the divestiture date under the manufacturing agreements with Samsung Bioepis. For additional information on the divestiture of our Hillerød, Denmark manufacturing operations, please read Note 3, Divestitures, to these consolidated financial statements. Under the license agreement, we granted Samsung Bioepis an exclusive license to use, develop, manufacture and commercialize biosimilar products created by Samsung Bioepis using Biogen product-specific technology. In exchange, we receive single digit royalties on biosimilar products developed and commercialized by Samsung Bioepis. For the years ended December 31, 2020, 2019 and 2018, we recognized $20.9 million, $106.2 million and $96.4 million, respectively, in revenues under the license, technical development services and manufacturing agreements, which is reflected in revenues from collaborative and other relationships, as a component of other revenues in our consolidated statements of income. Amounts receivable from Samsung Bioepis related to the agreements discussed above were $5.1 million and $85.0 million as of December 31, 2020 and 2019, respectively. Amounts payable to Samsung Bioepis as of December 31, 2020, were $99.0 million. Amounts payable to Samsung Bioepis as of December 31, 2019, consisted of the $100.0 million upfront payment related to the transaction we completed in December 2019, as discussed above.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Investments in Variable Interest Entities |
12 Months Ended |
|---|---|
Dec. 31, 2020 | |
| Investments in Variable Interest Entities [Abstract] | |
| Investments in Variable Interest Entities | Investments in Variable Interest Entities Consolidated Variable Interest Entities Our consolidated financial statements include the financial results of variable interest entities in which we are the primary beneficiary. The following are our significant variable interest entities. Neurimmune SubOne AG We have a collaboration and license agreement with Neurimmune SubOne AG (Neurimmune) for the development and commercialization of antibodies for the potential treatment of Alzheimer's disease, including aducanumab (as amended, the Neurimmune Agreement). We are responsible for the development, manufacturing and commercialization of all collaboration products. The Neurimmune Agreement is effective for the longer of the duration of certain patents relating to a licensed product or 12 years from the first commercial sale of a licensed product. We consolidate the results of Neurimmune as we determined that we are the primary beneficiary of Neurimmune because we have the power through the collaboration to direct the activities that most significantly impact the entity’s economic performance and we are required to fund 100.0% of the research and development costs incurred in support of the collaboration. In October 2017 we amended the terms of the Neurimmune Agreement and made a $150.0 million payment to Neurimmune in exchange for a 15.0% reduction in the previously negotiated royalty rates payable on products developed under the Neurimmune Agreement, including royalties payable on potential commercial sales of aducanumab. In May 2018 we made an additional $50.0 million payment to Neurimmune to further reduce the previously negotiated royalty rates payable on products developed under the Neurimmune Agreement, including royalties payable on potential commercial sales of aducanumab, by an additional 5.0%. Our royalty rates payable on products developed under the Neurimmune Agreement, including royalty rates payable on potential commercial sales of aducanumab, now range from the high single digits to sub-teens. As we consolidate the results of Neurimmune, we treated these payments as distributions and recognized them as charges to noncontrolling interests in the fourth quarter of 2017 and the second quarter of 2018, as applicable. Under the terms of the Neurimmune Agreement, we were required to pay Neurimmune a milestone payment of $75.0 million upon the regulatory filing with the FDA for the approval of aducanumab. During the second quarter of 2020, we paid Neurimmune $75.0 million upon the completed submission of the BLA for the approval of aducanumab to the FDA, which was recognized as a charge to noncontrolling interests for the year ended December 31, 2020. In addition, for the year ended December 31, 2020, we recognized net profit-sharing income of $33.8 million to reflect Eisai's 45.0% share of the $75.0 million milestone expense. Additionally, if aducanumab receives regulatory approval in the jurisdictions where we have submitted filings, we may pay up to approximately $200.0 million in milestones to Neurimmune in 2021, which includes $100.0 million if launched in the U.S., $50.0 million if launched in three or more countries within the E.U. and $50.0 million if launched in Japan. Milestones payable to Neurimmune are shared expenses under the Aducanumab Collaboration Agreement with Eisai. Research and development costs for which we reimburse Neurimmune are reflected in research and development expense in our consolidated statements of income. During the years ending December 31, 2020, 2019 and 2018, amounts reimbursed were immaterial. The assets and liabilities of Neurimmune are not significant to our consolidated financial position or results of operations as it is a research and development organization. We have provided no financing to Neurimmune other than contractually required amounts. Under the Aducanumab Collaboration Agreement, Eisai had an option to share in the benefit and cost associated with the royalty reductions discussed above; however, Eisai did not elect to share in the benefit and cost with respect to either the October 2017 or May 2018 royalty reductions, which will impact the amount of profits (losses) on potential commercial sales of aducanumab to be shared with Eisai. For additional information on our collaboration arrangements with Eisai, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Unconsolidated Variable Interest Entities We have relationships with various variable interest entities that we do not consolidate as we lack the power to direct the activities that significantly impact the economic success of these entities. These relationships include investments in certain biotechnology companies and research collaboration agreements. As of December 31, 2020 and 2019, the carrying value of our investments in certain biotechnology companies representing potential unconsolidated variable interest entities totaled $12.8 million and $22.7 million, respectively. Our maximum exposure to loss related to these variable interest entities is limited to the carrying value of our investments. We have also entered into research collaboration agreements with certain variable interest entities where we are required to fund certain development activities. These development activities are included in research and development expense in our consolidated statements of income as they are incurred. We have provided no financing to these variable interest entities other than previously contractually required amounts.
|
Litigation |
12 Months Ended |
|---|---|
Dec. 31, 2020 | |
| Loss Contingency, Information about Litigation Matters [Abstract] | |
| Litigation | Litigation We are currently involved in various claims and legal proceedings, including the matters described below. For information as to our accounting policies relating to claims and legal proceedings, including use of estimates and contingencies, please read Note 1, Summary of Significant Accounting Policies, to these consolidated financial statements. With respect to some loss contingencies, an estimate of the possible loss or range of loss cannot be made until management has further information, including, for example, (i) which claims, if any, will survive dispositive motion practice; (ii) information to be obtained through discovery; (iii) information as to the parties' damages claims and supporting evidence; (iv) the parties’ legal theories; and (v) the parties' settlement positions. The claims and legal proceedings in which we are involved also include challenges to the scope, validity or enforceability of the patents relating to our products, pipeline or processes and challenges to the scope, validity or enforceability of the patents held by others. These include claims by third parties that we infringe their patents. An adverse outcome in any of these proceedings could result in one or more of the following and have a material impact on our business or consolidated results of operations and financial position: (i) loss of patent protection; (ii) inability to continue to engage in certain activities; and (iii) payment of significant damages, royalties, penalties and/or license fees to third parties. Loss Contingencies Aducanumab Securities Litigation We and certain current and former officers are named as defendants in an action filed by a shareholder on November 13, 2020, in the U.S. District Court for the Central District of California and an action filed by a shareholder on January 5, 2021, in the U.S. District Court for the District of Massachusetts. Both actions allege violations of federal securities laws under 15 U.S.C §78j(b) and §78t(a) and 17 C.F.R. §240.10b-5 and are seeking a declaration of the action as a class action and an award of damages, interest and attorneys' fees. An estimate of the possible loss or range of loss cannot be made at this time. No trial date has been set. IMRALDI Patent Litigation In September 2018 Fresenius Kabi Deutschland GmbH (Fresenius Kabi) commenced proceedings for damages and injunctive relief against Biogen France SAS in the Tribunal de Grande Instance de Paris, alleging that IMRALDI, the adalimumab biosimilar product of Samsung Bioepis UK Limited that Biogen has commercialized in Europe, infringes the French counterpart of European Patent No. 3 148 510 (the '510 Patent), which was issued in June 2018 and expires in May 2035. No hearing has been scheduled. In October 2018 Fresenius Kabi commenced preliminary injunction proceedings against Biogen (Denmark) Manufacturing ApS and Biogen Denmark A/S in Denmark's Maritime and Commercial High Court alleging infringement of Danish Utility Models. In June 2019 the Danish court denied Fresenius Kabi's request for a preliminary injunction. Fresenius Kabi has appealed that decision and was permitted to add a claim of infringement of the Danish counterpart of the ‘510 patent, and the appeal is pending. In July 2020 the Danish Patent Board of Appeal revoked the Danish Utility Models that were the subject of Fresenius Kabi’s October 2018 request for a preliminary injunction and Fresenius Kabi has appealed those revocations to Denmark’s Maritime and Commercial High Court. No hearing has been scheduled in that appeal. In June 2020 Fresenius Kabi commenced preliminary injunction proceedings against Biogen (Denmark) Manufacturing ApS and Biogen (Denmark) A/S in Denmark’s Maritime and Commercial High Court alleging infringement of another Danish Utility Model. A hearing has been scheduled for May 2021. In November 2018 Fresenius Kabi commenced infringement proceedings for damages and injunctive relief against Biogen GmbH in the Düsseldorf Regional Court relating to the German counterpart of the ‘510 Patent. A hearing has been set for August 2021. In July 2019 Gedeon Richter PLC (Gedeon Richter) commenced proceedings against Biogen GmbH in the Düsseldorf Regional Court alleging infringement of the German counterpart of European Patent No. 3 212 667 (the '667 Patent), which was issued in September 2018 and expires in October 2035, and seeking damages and injunctive relief. A hearing has been set for November 2021. An estimate of the possible loss or range of loss in the IMRALDI patent litigation described above cannot be made at this time. Qui Tam Litigation In July 2015 a qui tam action filed by Michael Bawduniak on behalf of the U.S. and certain states was unsealed by the U.S. District Court for the District of Massachusetts. The action alleges sales and promotional activities in violation of the federal False Claims Act and state law counterparts and seeks single and treble damages, civil penalties, interest, attorneys’ fees and costs. No trial date has been set. The U.S. has not made an intervention decision. An estimate of the possible loss or range of loss cannot be made at this time. Dispute with Former Convergence Shareholders In November and December 2019 Shareholder Representative Services LLC, on behalf of the former shareholders of Convergence, sent us correspondence asserting claims of $200.0 million for alleged breach of the contract under which we acquired Convergence. We dispute the claims. Dispute with Jacobs Switzerland GmbH Jacobs Switzerland GmbH, the general contractor for the construction of our large-scale biologics manufacturing facility in Solothurn, Switzerland, claimed additional payments were due for construction costs. We have reached an agreement in principle to resolve the claim. Other Matters Petition for Inter Partes Review In July 2018 Mylan Pharmaceuticals, Inc. (Mylan) filed a petition that was granted by the U.S. Patent Trial and Appeal Board (PTAB) for inter partes review of our U.S. Patent No. 8,399,514 (the '514 Patent). The '514 Patent includes claims covering treatment of MS with 480 mg of dimethyl fumarate per day as provided for in our TECFIDERA label. In February 2020 the PTAB issued a final written decision upholding the patentability of the ‘514 Patent and in April 2020 Mylan filed an appeal in the U.S. Court of Appeals for the Federal Circuit (the Federal Circuit), which is pending. Hatch-Waxman Act Litigation relating to TECFIDERA Orange-Book Listed Patents In 2017 to 2020, we filed patent infringement proceedings relating to TECFIDERA Orange-Book listed patents pursuant to the Drug Price Competition and Patent Term Restoration Act of 1984, commonly known as the Hatch-Waxman Act (the Delaware Actions), against Accord Healthcare Inc., Alkem Laboratories Ltd., Amneal Pharmaceuticals LLC, Cipla Limited, Graviti Pharmaceuticals Pvt. Ltd., Hetero USA, Inc., Lupin Atlantis Holdings SA, Macleods Pharmaceuticals, Ltd., MSN Laboratories Pvt. Ltd., Pharmathen S.A., Prinston Pharmaceutical Inc., Sandoz Inc., Shilpa Medicare Limited, Slayback Pharma LLC, Sun Pharmaceutical Industries, Ltd., Sun Pharmaceutical Industries, Inc., Sun Pharma Global FZE, Torrent Pharmaceuticals Ltd., TWi Pharmaceuticals, Inc., Windlas Healthcare Pvt. Ltd. and Zydus Pharmaceuticals (USA) Inc. (collectively, the Delaware Defendants) in the U.S. District Court for the District of Delaware (the Delaware Court) and against Mylan in the U.S. District Court for the Northern District of West Virginia (the West Virginia Court). On June 22, 2020, the West Virginia Court entered judgment for Mylan that the asserted claims of the ‘514 Patent are invalid for lack of written description. We appealed the judgment to the Federal Circuit and the appeal is pending. The Delaware Court entered judgment for the Delaware Defendants on the grounds that the judgment of the West Virginia Court applies to the Delaware Actions under principles of collateral estoppel. We have appealed the judgments and the appeal is pending. European Patent Office Oppositions In 2016 the European Patent Office (EPO) revoked our European Patent No. 2 137 537, which covers the treatment of MS with 480 mg of dimethyl fumarate as provided for in our TECFIDERA label. We have appealed to the Technical Boards of Appeal of the EPO and a hearing date has been set for January 2022. In March 2018 the EPO revoked Forward Pharma's European Patent No. 2 801 355, which expires in October 2025. Forward Pharma has filed an appeal to the Technical Boards of Appeal of the EPO and a hearing has been set for September 2021. TYSABRI Patent Revocation Matters In November 2017 Bioeq GMBH, affiliated with the Polpharma Group, brought an action in the Polish Patent Office seeking to revoke Polish Patent No. 215263 (the Polish '263 Patent), which corresponds to our European Patent No. 1 485 127 (the E.U. '127 Patent) and covers administration of natalizumab (TYSABRI) to treat MS. The Polish '263 Patent expires in February 2023. A hearing was held in January 2021 and a decision is pending. In August 2020 a related entity, Polpharma Biologics S.A., also brought an action seeking to revoke the Polish ‘263 Patent. No hearing has been set in this matter. Swiss Pharma International AG, also affiliated with the Polpharma Group, filed actions in the District Court of the Hague, Netherlands (January 2016), the German Patents Court (March 2016) and the Commercial Court of Rome (November 2017) seeking to invalidate the Dutch, German and Italian counterparts, respectively, of the E.U. '127 Patent, which also cover administration of natalizumab (TYSABRI) to treat MS and expire in February 2023. The Dutch and German counterparts were ruled invalid. The decision in the Dutch action was affirmed on appeal and the German appeal has been withdrawn. No hearing has been set in the Italian action. Annulment Proceedings in General Court of the European Union relating to TECFIDERA Pharmaceutical Works Polpharma SA (Polpharma) and Mylan Ireland Ltd. (Mylan Ireland) have each filed applications in the General Court of the European Union (Polpharma in October 2018 and Mylan Ireland in November 2020) seeking to annul decisions of the European Medicines Agency (EMA) refusing to validate Polpharma’s and Mylan Ireland’s respective applications to market a generic version of TECFIDERA. The EMA’s refusals were on the grounds that TECFIDERA benefits from regulatory data protection. Biogen and the European Commission were granted leave to intervene in support of the EMA in the case brought by Polpharma. That case was heard in July 2020 and we are awaiting a decision. We intend to seek leave to intervene in support of the EMA in the case brought by Mylan Ireland. No hearing has been set in that matter. Product Liability and Other Legal Proceedings We are also involved in product liability claims and other legal proceedings generally incidental to our normal business activities. While the outcome of any of these proceedings cannot be accurately predicted, we do not believe the ultimate resolution of any of these existing matters would have a material adverse effect on our business or financial condition.
|
Commitments and Contingencies |
12 Months Ended |
|---|---|
Dec. 31, 2020 | |
| Commitments and Contingencies Disclosure [Abstract] | |
| Commitments and Contingencies | Commitments and Contingencies Royalty Payments TYSABRI In 2013 we acquired from Elan full ownership of all remaining rights to TYSABRI that we did not already own or control. Under the acquisition agreement, we are obligated to make contingent payments to Elan of 18.0% on annual worldwide net commercial sales up to $2.0 billion and 25.0% on annual worldwide net commercial sales that exceed $2.0 billion. Royalty payments to Elan and other third parties are recognized as cost of sales in our consolidated statements of income. Elan was acquired by Perrigo Company plc (Perrigo) in December 2013 and Perrigo subsequently sold its rights to these payments to a third-party effective January 2017. SPINRAZA In 2016 we exercised our option to develop and commercialize SPINRAZA from Ionis. Under our agreement with Ionis, we make royalty payments to Ionis on annual worldwide net commercial sales of SPINRAZA using a tiered royalty rate between 11.0% and 15.0%, which are recorded as cost of sales in our consolidated statements of income. For additional information on our collaboration arrangements with Ionis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. VUMERITY In October 2019 the FDA approved VUMERITY for the treatment of RMS. Under our agreement with Alkermes, we make royalty payments to Alkermes on worldwide net commercial sales of VUMERITY using a royalty rate of 15.0%, which are recorded as cost of sales in our consolidated statements of income. Royalties payable on net commercial sales of VUMERITY are subject, under certain circumstances, to tiered minimum annual payment requirements for a period of five years following FDA approval. For additional information on our collaboration arrangement with Alkermes, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Contingent Consideration related to Business Combinations In connection with our acquisition of Convergence, we agreed to make additional payments based upon the achievement of certain milestone events. As the acquisition of Convergence occurred after January 1, 2009, we recognized the contingent consideration liabilities associated with this transaction at their fair value on the acquisition date and revalue the remaining obligations each reporting period. We may pay up to approximately $400.0 million in remaining milestones related to these acquisitions. Fumapharm AG In 2006 we acquired Fumapharm AG. As part of this acquisition we acquired the Fumapharm Products. We were required to make contingent payments to former shareholders of Fumapharm AG and holders of their rights based on the attainment of certain cumulative sales levels of Fumapharm Products and the level of total net sales of Fumapharm Products in the prior 12-month period, as defined in the acquisition agreement, until such time as the cumulative sales level reached $20.0 billion, at which time no further contingent payments were due. During the first quarter of 2019 we paid the final $300.0 million contingent payment as we achieved the $20.0 billion cumulative sales levels related to the Fumapharm Products in the fourth quarter of 2018. Contingent Development, Regulatory and Commercial Milestone Payments Based on our development plans as of December 31, 2020, we could trigger potential future milestone payments to third parties of up to approximately $10.2 billion, including approximately $1.9 billion in development milestones, approximately $1.3 billion in regulatory milestones and approximately $7.0 billion in commercial milestones, as part of our various collaborations, including licensing and development programs. Payments under these agreements generally become due and payable upon achievement of certain development, regulatory or commercial milestones. Because the achievement of these milestones was not considered probable as of December 31, 2020, such contingencies have not been recorded in our financial statements. Amounts related to contingent milestone payments are not considered contractual obligations as they are contingent on the successful achievement of certain development, regulatory or commercial milestones. If certain clinical and commercial milestones are met, we may pay up to $86.2 million in milestones in 2021 under our current agreements. Additionally, if aducanumab receives regulatory approval in the jurisdictions where we have submitted filings, we may pay up to $200.0 million in milestones to Neurimmune in 2021, which includes $100.0 million if launched in the U.S., $50.0 million if launched in three or more countries within the E.U. and $50.0 million if launched in Japan. Milestones payable to Neurimmune are shared expenses under the Aducanumab Collaboration Agreement with Eisai. During the second quarter of 2020 we paid Neurimmune $75.0 million upon the completed submission of the BLA for the approval of aducanumab to the FDA, which was recognized as a charge to noncontrolling interests for the year ended December 31, 2020. Other Funding Commitments As of December 31, 2020, we have several ongoing clinical studies in various clinical trial stages. Our most significant clinical trial expenditures are to CROs. The contracts with CROs are generally cancellable, with notice, at our option. We recorded accrued expenses of approximately $21.7 million in our consolidated balance sheet for expenditures incurred by CROs as of December 31, 2020. We have approximately $593.0 million in cancellable future commitments based on existing CRO contracts as of December 31, 2020. As part of the sale of our Hillerød, Denmark manufacturing operations to FUJIFILM, we provided FUJIFILM with certain minimum batch production commitment guarantees. There is a risk that the minimum contractual batch production commitments will not be met. Based upon current estimates we do not expect to incur an adverse commitment obligation associated with such guarantees. We developed this estimate using a probability-weighted estimate of future manufacturing activity and may further adjust this estimate based upon changes in business conditions, which may result in the increase or reduction of this adverse commitment obligation in subsequent periods. Tax Related Obligations We exclude liabilities pertaining to uncertain tax positions from our summary of contractual obligations as we cannot make a reliable estimate of the period of cash settlement with the respective taxing authorities. As of December 31, 2020, we have approximately $79.6 million of liabilities associated with uncertain tax positions. As of December 31, 2020 and 2019, we have accrued income tax liabilities of $697.0 million, respectively, under the Transition Toll Tax. Of the amounts accrued as of December 31, 2020, $62.0 million is expected to be paid within one year. The Transition Toll Tax will be paid over an eight--year period, which started in 2018, and does not accrue interest. For additional information on the Transition Toll Tax, please read Note 16, Income Taxes, to these consolidated financial statements. Solothurn, Switzerland Manufacturing Facility In order to support our future growth and drug development pipeline, we are building a large-scale biologics manufacturing facility in Solothurn, Switzerland. We expect this facility to be partially operational during the first half of 2021. As of December 31, 2020, we had contractual commitments of approximately $9.3 million related to the construction of this facility.
|
Guarantees |
12 Months Ended |
|---|---|
Dec. 31, 2020 | |
| Guarantees [Abstract] | |
| Guarantees | Guarantees As of December 31, 2020 and 2019, we did not have significant liabilities recorded for guarantees. We enter into indemnification provisions under our agreements with other companies in the ordinary course of business, typically with business partners, contractors, clinical sites and customers. Under these provisions, we generally indemnify and hold harmless the indemnified party for losses suffered or incurred by the indemnified party as a result of our activities. These indemnification provisions generally survive termination of the underlying agreement. The maximum potential amount of future payments we could be required to make under these indemnification provisions is unlimited. However, to date we have not incurred material costs to defend lawsuits or settle claims related to these indemnification provisions. As a result, the estimated fair value of these agreements is minimal. Accordingly, we have no liabilities recorded for these agreements as of December 31, 2020 and 2019.
|
Employee Benefit Plans |
12 Months Ended |
|---|---|
Dec. 31, 2020 | |
| Retirement Benefits [Abstract] | |
| Employee Benefit Plans | Employee Benefit Plans We sponsor various retirement and pension plans. Our estimates of liabilities and expenses for these plans incorporate a number of assumptions, including expected rates of return on plan assets and interest rates used to discount future benefits. 401(k) Savings Plan We maintain a 401(k) Savings Plan, which is available to substantially all regular employees in the U.S. over the age of 21. Participants may make voluntary contributions. We make matching contributions according to the 401(k) Savings Plan’s matching formula. All matching contributions and participant contributions vest immediately. The 401(k) Savings Plan also holds certain transition contributions on behalf of participants who previously participated in the Biogen, Inc. Retirement Plan. The expense related to our 401(k) Savings Plan primarily consists of our matching contributions. Expense related to our 401(k) Savings Plan totaled $44.3 million, $44.8 million and $42.2 million for the years ended December 31, 2020, 2019 and 2018, respectively. Deferred Compensation Plan We maintain a non-qualified deferred compensation plan, known as the Supplemental Savings Plan (SSP), which allows a select group of management employees in the U.S. to defer a portion of their compensation. The SSP also provides certain credits to highly compensated U.S. employees that are paid by the company. These credits are known as the Restoration Match. The deferred compensation amounts are accrued when earned. Such deferred compensation is distributable in cash in accordance with the rules of the SSP. Deferred compensation amounts under such plan as of December 31, 2020 and 2019, totaled approximately $120.0 million and $114.6 million, respectively, and are included in other long-term liabilities in our consolidated balance sheets. The SSP also holds certain transition contributions on behalf of participants who previously participated in the Biogen, Inc. Retirement Plan. The Restoration Match and participant contributions vest immediately. Distributions to participants can be either in one lump sum payment or annual installments as elected by the participants. Pension Plans Our retiree benefit plans include defined benefit plans for employees in our affiliates in Switzerland and Germany as well as other insignificant defined benefit plans in certain other countries where we maintain an operating presence. Our Swiss plan is a government-mandated retirement fund that provides employees with a minimum investment return. The minimum investment return is determined annually by the Swiss government and was 1.00% in 2020, 2019 and 2018. Under the Swiss plan, both we and certain of our employees with annual earnings in excess of government determined amounts are required to make contributions into a fund managed by an independent investment fiduciary. Employer contributions must be in an amount at least equal to the employee’s contribution. Minimum employee contributions are based on the respective employee’s age, salary and gender. As of December 31, 2020 and 2019, the Swiss plan had an unfunded net pension obligation of $75.7 million and $42.9 million, respectively, and plan assets that totaled $170.0 million and $127.1 million, respectively. In 2020, 2019 and 2018 we recognized expense totaling $15.5 million, $14.7 million and $14.8 million, respectively, related to our Swiss plan, of which $2.6 million, $1.2 million and $1.3 million, respectively, was included in other income (expense), net. The obligations under the German plans are unfunded and totaled $75.5 million and $59.6 million as of December 31, 2020 and 2019, respectively. Net periodic pension cost related to the German plans totaled $6.2 million, $5.1 million and $5.3 million for the years ended December 31, 2020, 2019 and 2018, respectively, of which $2.0 million, $1.4 million and $1.5 million, respectively, was included in other income (expense), net.
|
Segment Information |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Segment Reporting [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Segment Information | Segment Information We operate as one operating segment, focused on discovering, developing and delivering worldwide innovative therapies for people living with serious neurological and neurodegenerative diseases as well as related therapeutic adjacencies. Our Chief Executive Officer (CEO), as the chief operating decision-maker, manages and allocates resources to the operations of our company on a total company basis. Our research and development organization is responsible for the research and discovery of new product candidates and supports development and registration efforts for potential future products. Our pharmaceutical, operations and technology organization manages the development of the manufacturing processes, clinical trial supply, commercial product supply, distribution, buildings and facilities. Our commercial organization is responsible for U.S. and international development of our commercial products. The company is also supported by corporate staff functions. Managing and allocating resources on a total company basis enables our CEO to assess the overall level of resources available and how to best deploy these resources across functions, therapeutic areas and research and development projects that are in line with our long-term company-wide strategic goals. Consistent with this decision-making process, our CEO uses consolidated, single-segment financial information for purposes of evaluating performance, forecasting future period financial results, allocating resources and setting incentive targets. Enterprise-wide disclosures about product revenues, other revenues and long-lived assets by geographic area are presented below. Revenues are primarily attributed to individual countries based on location of the customer or licensee. Geographic Information The following tables contain certain financial information by geographic area:
Other As of December 31, 2020, 2019 and 2018, approximately $2,180.6 million, $2,028.8 million and $1,748.5 million, respectively, of our long-lived assets were related to the construction of our large-scale biologics manufacturing facility in Solothurn, Switzerland. In August 2019 we completed the sale of all of the outstanding shares of our subsidiary that owned our biologics manufacturing operations in Hillerød, Denmark to FUJIFILM. As of December 31, 2018, approximately $646.5 million of our long-lived assets were related to our manufacturing facility in Hillerød, Denmark. For additional information on our large-scale biologics manufacturing facility in Solothurn, Switzerland, please read Note 10, Property, Plant and Equipment, to these consolidated financial statements. For additional information on the divestiture of our Hillerød, Denmark manufacturing operations, please read Note 3, Divestitures, to these consolidated financial statements.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quarterly Financial Data (Unaudited) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quarterly Financial Information Disclosure [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quarterly Financial Data (Unaudited) | Quarterly Financial Data (Unaudited)
(1) Gross profit is calculated as total revenues less cost of sales, excluding amortization and impairment of acquired intangible assets.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Summary of Significant Accounting Policies (Policies) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Accounting Policies [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||
| Business Overview | Our marketed products include TECFIDERA, VUMERITY, AVONEX, PLEGRIDY, TYSABRI and FAMPYRA for the treatment of MS; SPINRAZA for the treatment of SMA; and FUMADERM for the treatment of severe plaque psoriasis. We have certain business and financial rights with respect to RITUXAN for the treatment of non-Hodgkin's lymphoma, chronic lymphocytic leukemia (CLL) and other conditions; RITUXAN HYCELA for the treatment of non-Hodgkin's lymphoma and CLL; GAZYVA for the treatment of CLL and follicular lymphoma; OCREVUS for the treatment of primary progressive MS (PPMS) and relapsing MS (RMS); and other potential anti-CD20 therapies pursuant to our collaboration arrangements with Genentech, Inc. (Genentech), a wholly-owned member of the Roche Group. For additional information on our collaboration arrangements with Genentech, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Our innovative drug development and commercialization activities are complemented by our biosimilar business that expands access to medicines and reduces the cost burden for healthcare systems. Through our agreements with Samsung Bioepis Co., Ltd. (Samsung Bioepis), our joint venture with Samsung BioLogics Co., Ltd. (Samsung BioLogics), we market and sell BENEPALI, an etanercept biosimilar referencing ENBREL, IMRALDI, an adalimumab biosimilar referencing HUMIRA, and FLIXABI, an infliximab biosimilar referencing REMICADE, in certain countries in Europe and have an option to acquire exclusive rights to commercialize these products in China. Additionally, we have exclusive rights to commercialize two potential ophthalmology biosimilar products, SB11, a proposed ranibizumab biosimilar referencing LUCENTIS, and SB15, a proposed aflibercept biosimilar referencing EYLEA, in major markets worldwide, including the United States (U.S.), Canada, Europe, Japan and Australia. For additional information on our collaboration arrangements with Samsung Bioepis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Consolidation | Our consolidated financial statements reflect our financial statements, those of our wholly-owned subsidiaries and those of certain variable interest entities where we are the primary beneficiary. For consolidated entities where we own or are exposed to less than 100.0% of the economics, we record net income (loss) attributable to noncontrolling interests in our consolidated statements of income equal to the percentage of the economic or ownership interest retained in such entities by the respective noncontrolling parties. Intercompany balances and transactions are eliminated in consolidation. In determining whether we are the primary beneficiary of a variable interest entity, we apply a qualitative approach that determines whether we have both (1) the power to direct the economically significant activities of the entity and (2) the obligation to absorb losses of, or the right to receive benefits from, the entity that could potentially be significant to that entity. These considerations impact the way we account for our existing collaborative relationships and other arrangements. We continuously assess whether we are the primary beneficiary of a variable interest entity as changes to existing relationships or future transactions may result in us consolidating or deconsolidating one or more of our collaborators or partners.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Use of Estimates | The preparation of our consolidated financial statements requires us to make estimates, judgments and assumptions that may affect the reported amounts of assets, liabilities, equity, revenues and expenses and related disclosure of contingent assets and liabilities. On an ongoing basis we evaluate our estimates, judgments and methodologies. We base our estimates on historical experience and on various other assumptions that we believe are reasonable, the results of which form the basis for making judgments about the carrying values of assets, liabilities and equity and the amount of revenues and expenses. Actual results may differ from these estimates. The length of time and full extent to which the COVID-19 pandemic directly or indirectly impacts our business, results of operations and financial condition, including sales, expenses, reserves and allowances, manufacturing, clinical trials, research and development costs and employee-related amounts, depends on future developments that are highly uncertain, subject to change and are difficult to predict, including as a result of new information that may emerge concerning COVID-19 and the actions taken to contain or treat COVID-19 as well as the economic impact on local, regional, national and international customers and markets. We have made estimates of the impact of COVID-19 within our condensed consolidated financial statements and there may be changes to those estimates in future periods.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Revenue Recognition | In May 2014 the Financial Accounting Standards Board (FASB) issued Accounting Standards Update (ASU) No. 2014-09, Revenue from Contracts with Customers (Topic 606), which supersedes all existing revenue recognition requirements, including most industry specific guidance. This standard requires a company to recognize revenues when it transfers goods or services to customers in an amount that reflects the consideration that the company expects to receive for those goods or services. This standard became effective for us on January 1, 2018, and was adopted using the modified retrospective method. The adoption of this standard as of January 1, 2018, did not change our revenue recognition. We recognize revenues when our customer obtains control of promised goods or services, in an amount that reflects the consideration which we expect to receive in exchange for those goods or services. We recognize revenues following the five-step model prescribed under the FASB Accounting Standards Codification (ASC) 606, Revenue from Contracts with Customers: (i) identify contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenues when (or as) we satisfy the performance obligation. Product Revenues In the U.S., we sell our products primarily to wholesale distributors and specialty pharmacy providers. In other countries, we sell our products primarily to wholesale distributors, hospitals, pharmacies and other third-party distribution partners. These customers subsequently resell our products to health care providers and patients. In addition, we enter into arrangements with health care providers and payors that provide for government-mandated or privately-negotiated discounts and allowances related to our products. Product revenues are recognized when the customer obtains control of our product, which occurs at a point in time, typically upon delivery to the customer. We expense incremental costs of obtaining a contract as and when incurred if the expected amortization period of the asset that we would have recognized is one year or less or the amount is immaterial. Reserves for Discounts and Allowances Product revenues are recorded net of reserves established for applicable discounts and allowances that are offered within contracts with our customers, health care providers or payors, including those associated with the implementation of pricing actions in certain of the international markets in which we operate. Our process for estimating reserves established for these variable consideration components do not differ materially from our historical practices. Product revenue reserves, which are classified as a reduction in product revenues, are generally characterized in the following categories: discounts, contractual adjustments and returns. These reserves are based on estimates of the amounts earned or to be claimed on the related sales and are classified as reductions of accounts receivable (if the amount is payable to our customer) or a liability (if the amount is payable to a party other than our customer). Our estimates of reserves established for variable consideration are calculated based upon a consistent application of our methodology utilizing the expected value method. These estimates reflect our historical experience, current contractual and statutory requirements, specific known market events and trends, industry data and forecasted customer buying and payment patterns. The transaction price, which includes variable consideration reflecting the impact of discounts and allowances, may be subject to constraint and is included in the net sales price only to the extent that it is probable that a significant reversal of the amount of the cumulative revenues recognized will not occur in a future period. Actual amounts may ultimately differ from our estimates. If actual results vary, we adjust these estimates, which could have an effect on earnings in the period of adjustment. Discounts include trade term discounts and wholesaler incentives. Trade term discounts and wholesaler incentives primarily relate to estimated obligations for credits to be granted to wholesalers for remitting payment on their purchases within established incentive periods and credits to be granted to wholesalers for compliance with various contractually-defined inventory management practices, respectively. We determine these reserves based on our historical experience, including the timing of customer payments. Contractual adjustments primarily relate to Medicaid and managed care rebates, pharmacy rebates, co-payment (copay) assistance, Veterans Administration (VA) and Public Health Service (PHS) discounts, specialty pharmacy program fees and other governmental rebates or applicable allowances. •Medicaid rebates relate to our estimated obligations to states under established reimbursement arrangements. Rebate accruals are recorded in the same period the related revenue is recognized, resulting in a reduction of product revenue and the establishment of a liability which is included in other current liabilities. Our liability for Medicaid rebates consists of estimates for claims that a state will make for the current quarter, claims for prior quarters that have been estimated for which an invoice has not been received, invoices received for claims from the prior quarters that have not been paid and an estimate of potential claims that will be made for inventory that exists in the distribution channel at period end. •Governmental rebates or chargebacks, including VA and PHS discounts, represent our estimated obligations resulting from contractual commitments to sell products to qualified healthcare providers at prices lower than the list prices we charge to wholesalers which provide those products. The wholesaler charges us for the difference between what the wholesaler pays for the products and the ultimate selling price to the qualified healthcare providers. Rebate and chargeback reserves are established in the same period as the related revenue is recognized, resulting in a reduction in product revenue and accounts receivable. Chargeback amounts are generally determined at the time of resale to the qualified healthcare provider from the wholesaler, and we generally issue credits for such amounts within a few weeks of the wholesaler notifying us about the resale. Our reserves for VA, PHS and chargebacks consist of amounts that we expect to issue for inventory that exists at the wholesalers that we expect will be sold to qualified healthcare providers and chargebacks that wholesalers have claimed for which we have not issued a credit. •Managed care rebates represent our estimated obligations to third parties, primarily pharmacy benefit managers. Rebate accruals are recorded in the same period the related revenue is recognized, resulting in a reduction of product revenue and the establishment of a liability which is included in accrued expenses and other current liabilities. These rebates result from performance-based goals, formulary position and price increase limit allowances (price protection). The calculation of the accrual for these rebates is based on an estimate of the coverage patterns and the resulting applicable contractual rebate rate(s) to be earned over a contractual period. •Copay assistance represents financial assistance to qualified patients, assisting them with prescription drug co-payments required by insurance. The calculation of the accrual for copay is based on an estimate of claims and the cost per claim that we expect to receive associated with inventory that exists in the distribution channel at period end. •Pharmacy rebates represent our estimated obligations resulting from contractual commitments to sell products to specific pharmacies. Rebate accruals are recorded in the same period the related revenue is recognized, resulting in a reduction of product revenue and the establishment of a liability which is included in accrued expenses and other current liabilities. These rebates result from contracted discounts on product purchased or product dispensed. The calculation of the accrual for these rebates is based on an estimate of the pharmacy’s buying or dispensing patterns and the resulting applicable contractual rebate rate(s) to be earned over the contractual period. •Other governmental rebates, non-U.S. pharmaceutical taxes or applicable allowances primarily relate to mandatory rebates and discounts in international markets where government-sponsored healthcare systems are the primary payors for healthcare. Product return reserves are established for returns expected to be made by wholesalers and are recorded in the period the related revenue is recognized, resulting in a reduction to product revenues. In accordance with contractual terms, wholesalers are permitted to return product for reasons such as damaged or expired product. The majority of wholesaler returns are due to product expiration. Expired product return reserves are estimated through a comparison of historical return data to their related sales on a production lot basis. Historical rates of return are determined for each product and are adjusted for known or expected changes in the marketplace specific to each product. In addition to discounts, rebates and product returns, we also maintain certain customer service contracts with distributors and other customers in the distribution channel that provide us with inventory management, data and distribution services, which are generally reflected as a reduction of revenues. To the extent we can demonstrate a separable benefit and fair value for these services we classify these payments in selling, general and administrative expenses. Revenues from Anti-CD20 Therapeutic Programs Our collaboration with Genentech is within the scope of ASC 808, Collaborative Agreements, which provides guidance on the presentation and disclosure of collaborative arrangements. For purposes of this footnote, we refer to RITUXAN and RITUXAN HYCELA collectively as RITUXAN. Our share of the pre-tax co-promotion profits on RITUXAN and GAZYVA and royalty revenues on the sale of OCREVUS resulted from an exchange of a license. As we do not have future performance obligations under the license or collaboration agreement, revenues are recognized as the underlying sales occur. Revenues from anti-CD20 therapeutic programs consist of: (i) our share of pre-tax profits and losses in the U.S. for RITUXAN and GAZYVA; and (ii) other revenues from anti-CD20 therapeutic programs, which primarily consist of our share of pre-tax co-promotion profits on RITUXAN in Canada and royalty revenues on sales of OCREVUS. Pre-tax co-promotion profits on RITUXAN and GAZYVA are calculated and paid to us by Genentech and the Roche Group. Pre-tax co-promotion profits consist of net sales to third-party customers less applicable costs to manufacture, third-party royalty expenses, distribution, selling and marketing expenses and joint development expenses incurred by Genentech and the Roche Group. Our share of the pre-tax profits on RITUXAN and GAZYVA include estimates that are based on information received from Genentech and the Roche Group. These estimates are subject to change and actual results may differ. We recognize royalty revenues on sales of OCREVUS based on our estimates from third party and market research data of OCREVUS sales occurring during the corresponding period. Differences between actual and estimated royalty revenues will be adjusted for in the period in which they become known, which is generally expected to be the following quarter. For additional information on our relationship with Genentech, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Other Revenues Royalty Revenues We recognize royalty revenues related to sales by our licensees of products covered under patents that we own. Collaborative and Other Relationships We have a number of significant collaborative and other third-party relationships for revenues and for the development, regulatory approval, commercialization and marketing of certain of our products and product candidates. Where we are the principal on sales transactions with third parties, we recognize revenues, cost of sales and operating expenses on a gross basis in their respective lines in our consolidated statements of income. Where we are not the principal on sales transactions with third parties, we record our share of the revenues, cost of sales and operating expenses on a net basis in collaborative and other relationships included in other revenues in our consolidated statements of income. Our development and commercialization arrangements with Genentech and Samsung Bioepis represent collaborative arrangements as each party is an active participant in one or more joint operating activities and is exposed to significant risks and rewards of these arrangements. These arrangements resulted from an exchange of a license and utilize the sales and usage based royalty exception. Therefore, revenues relating to royalties or profit-sharing amounts received are recognized as the underlying sales occur. For additional information on our collaboration arrangements with Genentech and Samsung Bioepis, please read Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Other Corporate Revenues We record other corporate revenues primarily from amounts earned under contract manufacturing agreements. Revenues under contract manufacturing agreements are recognized when the customer obtains control of the product, which may occur at a point in time or over time depending on the terms and conditions of the agreement.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Fair Value Measurements | We have certain financial assets and liabilities recorded at fair value which have been classified as Level 1, 2 or 3 within the fair value hierarchy as described in the accounting standards for fair value measurements. •Level 1 — Fair values are determined utilizing quoted prices (unadjusted) in active markets for identical assets or liabilities that we have the ability to access; •Level 2 — Fair values are determined by utilizing quoted prices for identical or similar assets and liabilities in active markets or other market observable inputs such as interest rates, yield curves, foreign currency spot rates and option pricing valuation models; and •Level 3 — Prices or valuations that require inputs that are both significant to the fair value measurement and unobservable. The majority of our financial assets have been classified as Level 2. Our financial assets (which include our cash equivalents, marketable debt securities and certain of our marketable equity securities, derivative contracts and plan assets for deferred compensation) have been initially valued at the transaction price and subsequently valued, at the end of each reporting period, utilizing third-party pricing services or option pricing valuation models. The pricing services utilize industry standard valuation models, including both income and market-based approaches and observable market inputs to determine value. These observable market inputs include reportable trades, benchmark yields, credit spreads, broker/dealer quotes, bids, offers, current spot rates and other industry and economic events. We validate the prices provided by our third-party pricing services by understanding the models used, obtaining market values from other pricing sources and analyzing pricing data in certain instances. The option pricing valuation models use assumptions within the model, including the term, stock price volatility, constant maturity risk-free interest rate and dividend yield. After completing our validation procedures, we did not adjust or override any fair value measurements provided by our pricing services as of December 31, 2020 and 2019. Other Assets and Liabilities The carrying amounts reflected in our consolidated balance sheets for current accounts receivable, due from anti-CD20 therapeutic programs, other current assets, accounts payable and accrued expenses and other, approximate fair value due to their short-term maturities.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Cash and Cash Equivalents | We consider only those investments that are highly liquid, readily convertible to cash and that mature within three months from date of purchase to be cash equivalents. As of December 31, 2020 and 2019, cash equivalents were comprised of money market funds, commercial paper, overnight reverse repurchase agreements and other debt securities with maturities less than 90 days from the date of purchase. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Accounts Receivable | The majority of our accounts receivable arise from product sales and primarily represent amounts due from our wholesale and other third-party distributors, public hospitals, pharmacies and other government entities and have standard payment terms that generally require payment within 30 to 90 days. We do not adjust our receivables for the effects of a significant financing component at contract inception if we expect to collect the receivables in one year or less from the time of sale. In countries where we have experienced a pattern of payments extending beyond our contractual payment term and we expect to collect receivables greater than one year from the time of sale, we have assessed whether the customer has a significant financing component and discounted our receivables and reduced related revenues over the period of time that we estimate those amounts will be paid using the country’s market-based borrowing rate for such period. The related receivables are classified at the time of sale as non-current assets. We accrete interest income on these receivables, which is recorded as a component of other income (expense), net in our consolidated statements of income. We provide reserves against accounts receivable for estimated losses that may result from a customer's inability to pay. Amounts determined to be uncollectible are charged or written-off against the reserve.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration of Credit Risk | Financial instruments that potentially subject us to concentrations of credit risk include cash and cash equivalents, investments, derivatives and accounts receivable. We attempt to minimize the risks related to cash and cash equivalents and investments by investing in a broad and diverse range of financial instruments as previously defined by us. We have established guidelines related to credit ratings and maturities intended to safeguard principal balances and maintain liquidity. Our investment portfolio is maintained in accordance with our investment policy, which defines allowable investments, specifies credit quality standards and limits the credit exposure of any single issuer. We minimize credit risk resulting from derivative instruments by choosing only highly rated financial institutions as counterparties.Concentrations of credit risk with respect to receivables, which are typically unsecured, are somewhat mitigated due to the wide variety of customers and markets using our products, as well as their dispersion across many different geographic areas. We monitor the financial performance and creditworthiness of our customers so that we can properly assess and respond to changes in their credit profile. We continue to monitor these conditions and assess their possible impact on our business. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Marketable Securities and Other Investments | Marketable Debt Securities Available-for-sale marketable debt securities are recorded at fair market value and unrealized gains and losses are included in accumulated other comprehensive income (loss) in equity, net of related tax effects, unless the security has experienced a credit loss, we have determined that we have the intent to sell the security or we have determined that it is more likely than not that we will have to sell the security before its expected recovery. Realized gains and losses are reported in other income (expense), net on a specific identification basis. Marketable Equity Securities and Venture Capital Funds Our marketable equity securities are recorded at fair market value and, beginning January 1, 2018, unrealized gains and losses are included in other income (expense), net in our consolidated statements of income. Prior to January 1, 2018, unrealized gains and losses were included in accumulated other comprehensive income (loss) in equity, net of related tax effects. Our marketable equity securities represent investments in publicly traded equity securities and are included in investments and other assets in our consolidated balance sheets. Our investments in venture capital funds are recorded at net asset value, which approximates fair value, and, beginning January 1, 2018, unrealized gains and losses are included in other income (expense), net in our consolidated statements of income. Prior to January 1, 2018, these investments were accounted for under the cost method of accounting. The underlying investments of the venture capital funds in which we invest are in equity securities of certain biotechnology companies and are included in investments and other assets in our consolidated balance sheets. Non-Marketable Equity Securities We also invest in equity securities of companies whose securities are not publicly traded and where fair value is not readily available. These investments are recorded using either the equity method of accounting or the cost minus impairment adjusted for changes in observable prices, depending on our ownership percentage and other factors that suggest we have significant influence. We monitor these investments to evaluate whether any increase or decline in their value has occurred, based on the implied value of recent company financings, public market prices of comparable companies and general market conditions. These investments are included in investments and other assets in our consolidated balance sheets. Evaluating Marketable Debt Securities for Other-than-Temporary Impairments We conduct periodic reviews to identify and evaluate each investment that has an unrealized loss, in accordance with the meaning of other-than-temporary impairment. An unrealized loss exists when the current fair value of an individual security is less than its amortized cost basis. Unrealized losses on available-for-sale debt securities that are determined to be temporary, and not related to credit loss, are recorded, net of tax, in accumulated other comprehensive income. For available-for-sale debt securities with unrealized losses, management performs an analysis to assess whether we intend to sell or whether we would more likely than not be required to sell the security before the expected recovery of the amortized cost basis. Where we intend to sell a security, or may be required to do so, the security’s decline in fair value is deemed to be other-than-temporary and the full amount of the unrealized loss is reflected in earnings as an impairment loss. Regardless of our intent to sell a security, we perform additional analysis on all securities with unrealized losses to evaluate losses associated with the creditworthiness of the security. Credit losses are identified where we do not expect to receive cash flows sufficient to recover the amortized cost basis of a security.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Equity Method of Accounting | In circumstances where we have the ability to exercise significant influence over the operating and financial policies of a company in which we have an investment, we utilize the equity method of accounting for recording investment activity. In assessing whether we exercise significant influence, we consider the nature and magnitude of our investment, the voting and protective rights we hold, any participation in the governance of the other company and other relevant factors such as the presence of a collaborative or other business relationship. Under the equity method of accounting, we record in our consolidated statements of income our share of income or loss of the other company. If our share of losses exceeds the carrying value of our investment, we will suspend recognizing additional losses and will continue to do so unless we commit to providing additional funding. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Inventory | Inventories are stated at the lower of cost or net realizable value with cost based on the first-in, first-out method. We classify our inventory costs as long-term when we expect to utilize the inventory beyond our normal operating cycle and include these costs in investments and other assets in our consolidated balance sheets. Inventory that can be used in either the production of clinical or commercial products is expensed as research and development costs when identified for use in a clinical manufacturing campaign. Capitalization of Inventory Costs We capitalize inventory costs associated with our products prior to regulatory approval, when, based on management’s judgment, future commercialization is considered probable and the future economic benefit is expected to be realized. We consider numerous attributes in evaluating whether the costs to manufacture a particular product should be capitalized as an asset. We assess the regulatory approval process and where the particular product stands in relation to that approval process, including any known safety or efficacy concerns, potential labeling restrictions and other impediments to approval. We evaluate our anticipated research and development initiatives and constraints relating to the product and the indication in which it will be used. We consider our manufacturing environment including our supply chain in determining logistical constraints that could hamper approval or commercialization. We consider the shelf life of the product in relation to the expected timeline for approval and we consider patent related or contract issues that may prevent or delay commercialization. We also base our judgment on the viability of commercialization, trends in the marketplace and market acceptance criteria. Finally, we consider the reimbursement strategies that may prevail with respect to the product and assess the economic benefit that we are likely to realize. We expense previously capitalized costs related to pre-approval inventory upon a change in such judgment, due to, among other potential factors, a denial or significant delay of approval by necessary regulatory bodies. At December 31, 2020, we capitalized approximately $93.8 million of pre-launch inventory for aducanumab, an anti-amyloid beta antibody candidate for the potential treatment of Alzheimer's disease that we are developing in collaboration with Eisai Co., Ltd. (Eisai). If aducanumab does not receive regulatory approval in the U.S., we would expense this inventory as research and development expense and, under the terms of our collaboration agreement with Eisai to jointly develop and commercialize aducanumab, Eisai would reimburse us for 45.0% of the costs. Obsolescence and Unmarketable Inventory At each reporting period we review our inventories for excess or obsolescence and write-down obsolete or otherwise unmarketable inventory to its estimated net realizable value. If the actual net realizable value is less than that estimated by us, or if it is determined that inventory utilization will further diminish based on estimates of demand, additional inventory write-downs may be required. Additionally, our products are subject to strict quality control and monitoring that we perform throughout the manufacturing process. In the event that certain batches or units of product no longer meet quality specifications, we will record a charge to cost of sales to write-down any unmarketable inventory to its estimated net realizable value. In all cases, product inventory is carried at the lower of cost or its estimated net realizable value. Amounts written-down due to unmarketable inventory are charged to cost of sales.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Property, Plant and Equipment | Property, plant and equipment are carried at cost, subject to reviews for impairment whenever events or changes in circumstances indicate that the carrying amount of the asset may not be recoverable. The cost of normal, recurring or periodic repairs and maintenance activities related to property, plant and equipment are expensed as incurred. The cost for planned major maintenance activities, including the related acquisition or construction of assets, is capitalized if the repair will result in future economic benefits. Interest costs incurred during the construction of major capital projects are capitalized until the underlying asset is ready for its intended use, at which point the interest costs are amortized as depreciation expense over the life of the underlying asset. We also capitalize certain direct and incremental costs associated with the validation effort required for licensing by regulatory agencies of new manufacturing equipment for the production of a commercially approved drug. These costs primarily include direct labor and material and are incurred in preparing the equipment for its intended use. The validation costs are either amortized over the life of the related equipment or expensed as cost of sales when the product produced in the validation process is sold. In addition, we capitalize certain internal use computer software development costs. If the software is an integral part of production assets, these costs are included in machinery and equipment and are amortized on a straight-line basis over the estimated useful lives of the related software, which generally range from to five years. We generally depreciate or amortize the cost of our property, plant and equipment using the straight-line method over the estimated useful lives of the respective assets, which are summarized as follows:
When we dispose of property, plant and equipment, we remove the associated cost and accumulated depreciation from the related accounts in our consolidated balance sheets and include any resulting gain or loss in our consolidated statements of income.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Leases | In February 2016 the FASB issued ASU No. 2016-02, Leases (Topic 842), a new standard issued to increase transparency and comparability among organizations related to their leasing activities. This standard established a right-of-use model that requires all lessees to recognize right-of-use assets and lease liabilities on their balance sheet that arise from leases as well as provide disclosures with respect to certain qualitative and quantitative information related to a company's leasing arrangements to meet the objective of allowing users of financial statements to assess the amount, timing and uncertainty of cash flows arising from leases. The FASB subsequently issued the following amendments to ASU 2016-02 that have the same effective date and transition date: ASU No. 2018-01, Leases (Topic 842): Land Easement Practical Expedient for Transition to Topic 842, ASU No. 2018-10, Codification Improvements to Topic 842, Leases, ASU No. 2018-11, Leases (Topic 842): Targeted Improvements, ASU No. 2018-20, Narrow-Scope Improvement for Lessors, and ASU No. 2019-01, Leases (Topic 842): Codification Improvements. We adopted these amendments with ASU 2016-02 (collectively, the new leasing standards) effective January 1, 2019. We adopted the new leasing standards using the modified retrospective transition approach, as of January 1, 2019, with no restatement of prior periods or cumulative adjustment to retained earnings. Upon adoption, we elected the package of transition practical expedients, which allowed us to carry forward prior conclusions related to whether any expired or existing contracts are or contain leases, the lease classification for any expired or existing leases and initial direct costs for existing leases. We also elected the practical expedient to not reassess certain land easements and made an accounting policy election to not recognize leases with an initial term of 12 months or less within our consolidated balance sheets and to recognize those lease payments on a straight-line basis in our consolidated statements of income over the lease term. Upon adoption of the new leasing standards we recognized an operating lease asset of approximately $463.0 million and a corresponding operating lease liability of approximately $526.0 million, which are included in our consolidated balance sheets. The adoption of the new leasing standards did not have an impact on our consolidated statements of income. We determine if an arrangement is a lease at contract inception. Operating lease assets represent our right to use an underlying asset for the lease term and operating lease liabilities represent our obligation to make lease payments arising from the lease. Operating lease assets and liabilities are recognized at the commencement date of the lease based upon the present value of lease payments over the lease term. When determining the lease term, we include options to extend or terminate the lease when it is reasonably certain that we will exercise that option. We use the implicit rate when readily determinable and use our incremental borrowing rate when the implicit rate is not readily determinable based upon the information available at the commencement date in determining the present value of the lease payments. Our incremental borrowing rate is determined using a secured borrowing rate for the same currency and term as the associated lease. The lease payments used to determine our operating lease assets may include lease incentives, stated rent increases and escalation clauses linked to rates of inflation when determinable and are recognized in our operating lease assets in our consolidated balance sheets. Our lease agreements may include both lease and non-lease components, which we account for as a single lease component when the payments are fixed. Variable payments included in the lease agreement are expensed as incurred. For certain equipment leases, such as vehicles, we apply a portfolio approach to effectively account for the operating lease assets and liabilities. Our operating leases are reflected in operating lease assets, accrued expenses and other and in long-term operating lease liabilities in our consolidated balance sheets. Lease expense for minimum lease payments is recognized on a straight-line basis over the lease term. We also have real estate lease agreements which are subleased to third parties. Operating leases for which we are the sublessor are included in accrued expenses and other and other long-term liabilities in our consolidated balance sheets. We recognize sublease income on a straight-line basis over the lease term in our consolidated statements of income. For additional information on the adoption of the new leasing standards, please read Note 11, Leases, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Intangible Assets | Our intangible assets consist of completed technology (comprised of acquired and in-licensed rights and patents, developed technology, out-licensed patents), in-process research and development (IPR&D) acquired after January 1, 2009, trademarks and trade names. Our intangible assets are recorded at fair value at the time of their acquisition and are stated in our consolidated balance sheets net of accumulated amortization and impairments, if applicable. Intangible assets related to acquired and in-licensed rights and patents, developed technology and out-licensed patents are amortized over their estimated useful lives using the economic consumption method if anticipated future revenues can be reasonably estimated. The straight-line method is used when revenues cannot be reasonably estimated. Amortization is recorded within amortization and impairment of acquired intangible assets in our consolidated statements of income. Acquired and in-licensed rights and patents primarily relate to our acquisition of all remaining rights to TYSABRI from Elan Pharma International Ltd. (Elan), an affiliate of Elan Corporation, plc. Acquired and in-licensed rights and patents also include other amounts related to our other marketed products and programs acquired through business combinations. Developed technology primarily relates to our AVONEX product, which was recorded in connection with the merger of Biogen, Inc. and IDEC Pharmaceuticals Corporation in 2003. We amortize the intangible assets related to our TYSABRI, AVONEX, SPINRAZA, VUMERITY and TECFIDERA (rest of world) products using the economic consumption method based on revenues generated from the products underlying the related intangible assets. An analysis of the anticipated lifetime revenues of our TYSABRI, AVONEX, SPINRAZA, VUMERITY and TECFIDERA (rest of world) products is performed annually during our long-range planning cycle and whenever events or changes in circumstances would significantly affect the anticipated lifetime revenues of our TYSABRI, AVONEX, SPINRAZA, VUMERITY and TECFIDERA (rest of world) products. Intangible assets related to trademarks, trade names and IPR&D prior to commercialization are not amortized because they have indefinite lives; however, they are subject to review for impairment. We review our intangible assets with indefinite lives for impairment annually, as of October 31, and whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Acquired In-process Research and Development (IPR&D) | Acquired IPR&D represents the fair value assigned to research and development assets that have not reached technological feasibility. The value assigned to acquired IPR&D is determined by estimating the costs to develop the acquired technology into commercially viable products, estimating the resulting revenues from the projects and discounting the net cash flows to present value. The revenues and costs projections used to value acquired IPR&D are, as applicable, reduced based on the probability of success of developing a new drug. Additionally, the projections consider the relevant market sizes and growth factors, expected trends in technology and the nature and expected timing of new product introductions by us and our competitors. The rates utilized to discount the net cash flows to their present value are commensurate with the stage of development of the projects and uncertainties in the economic estimates used in the projections. Upon the acquisition of IPR&D, we complete an assessment of whether our acquisition constitutes the purchase of a single asset or a group of assets. We consider multiple factors in this assessment, including the nature of the technology acquired, the presence or absence of separate cash flows, the development process and stage of completion, quantitative significance and our rationale for entering into the transaction. If we acquire a business as defined under applicable accounting standards, then the acquired IPR&D is capitalized as an intangible asset. If we acquire an asset or group of assets that do not meet the definition of a business under applicable accounting standards, then the acquired IPR&D is expensed on its acquisition date. Future costs to develop these assets are recorded to research and development expense as they are incurred. When performing our impairment assessment, we calculate the fair value using the same methodology as described above. If the carrying value of our acquired IPR&D exceeds its fair value, then the intangible asset is written down to its fair value. Changes in estimates and assumptions used in determining the fair value of our acquired IPR&D could result in an impairment. Impairments are recorded within amortization and impairment of acquired intangible assets in our consolidated statements of income. Assets that have been previously impaired, including our vixotrigine (BIIB074) program for the potential treatment of neuropathic pain, such as trigeminal neuralgia (TGN), could become further impaired in the future.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Goodwill | Goodwill represents the difference between the purchase price and the fair value of the identifiable tangible and intangible net assets when accounted for using the purchase method of accounting. Goodwill is not amortized, but is reviewed for impairment. Goodwill is reviewed for impairment annually, as of October 31, and whenever events or changes in circumstances indicate that the carrying value of the goodwill may not be recoverable. We compare the fair value of our reporting unit to its carrying value. If the carrying value of the net assets assigned to the reporting unit exceeds the fair value of our reporting unit, we would record an impairment loss equal to the difference. As described in Note 24, Segment Information, to these consolidated financial statements, we operate in one operating segment, which is our only reporting unit.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Impairment of Long-Lived Assets | Long-lived assets to be held and used, including property, plant and equipment, and definite-lived intangible assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of the assets or asset group may not be recoverable. Determination of recoverability is based on an estimate of undiscounted future cash flows resulting from the use of the asset and its eventual disposition. In the event that such cash flows are not expected to be sufficient to recover the carrying amount of the assets, the assets are written-down to their fair values. Long-lived assets to be disposed of are carried at fair value less costs to sell.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Contingent Consideration | The consideration for our acquisitions often includes future payments that are contingent upon the occurrence of a particular event or events. We record an obligation for such contingent payments at fair value on the acquisition date. We estimate the fair value of contingent consideration obligations through valuation models that incorporate probability-adjusted assumptions related to the achievement of the milestones and thus likelihood of making related payments. We revalue our contingent consideration obligations each reporting period. Changes in the fair value of our contingent consideration obligations are recognized in our consolidated statements of income. Changes in the fair value of the contingent consideration obligations can result from changes to one or multiple inputs, including adjustments to the discount rates, changes in the amount or timing of expected expenditures associated with product development, changes in the amount or timing of cash flows and reserves associated with products upon commercialization, changes in the assumed achievement or timing of any cumulative sales-based and development milestones, changes in the probability of certain clinical events and changes in the assumed probability associated with regulatory approval. Discount rates in our valuation models represent a measure of the credit risk associated with settling the liability. The period over which we discount our contingent obligations is based on the current development stage of the product candidates, our specific development plan for that product candidate adjusted for the probability of completing the development step and when the contingent payments would be triggered. In estimating the probability of success, we utilize data regarding similar milestone events from several sources, including industry studies and our own experience. These fair value measurements are based on significant inputs not observable in the market. Significant judgment is employed in determining the appropriateness of these assumptions as of the acquisition date and for each subsequent period. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Derivative Instruments and Hedging Activities | Cash Flow and Fair Value Derivative Instruments We recognize all derivative instruments as either assets or liabilities at fair value in our consolidated balance sheets. Changes in the fair value of our derivative instruments are recognized each period in current earnings or accumulated other comprehensive income (loss), depending on whether the derivative instrument is designated as part of a hedge transaction and, if so, the type of hedge transaction. We classify the cash flows from these instruments in the same category as the cash flows from the hedged items. We do not hold or issue derivative instruments for trading or speculative purposes. We assess at inception and on an ongoing basis, whether the derivative instruments that are used in hedging transactions are highly effective in offsetting the changes in cash flows or fair values of the hedged items. We exclude the forward points portion of the derivative instruments used in a hedging transaction from the effectiveness test and record the fair value gain or loss related to this portion each period in our consolidated statements of income in the same line as the underlying hedged item. If we determine that a forecasted transaction is no longer probable of occurring, we discontinue hedge accounting for the affected portion of the hedge instrument, and any related unrealized gain or loss on the contract is recognized in current earnings. Net Investment Derivative Instruments We are exposed to the impact of foreign exchange fluctuations on our investment in the equity of Samsung Bioepis, which is denominated in a currency other than the U.S. dollar, and could adversely impact the U.S. dollar value of this investment. Using derivative instruments, we have hedged our net investment position to mitigate the effects of foreign exchange fluctuations. We recognize these designated net investment hedges as either assets or liabilities, at fair value, in our consolidated balance sheets. We hedge the changes in the spot exchange rate in accumulated other comprehensive income (loss) and exclude changes to the forward rate and amortize the forward points in other income (expense), net in our consolidated statements of income over the term of the contract. We classify the cash flows from these instruments in the same category as the cash flows from the hedged items. For additional information on our derivative instruments and hedging activities, please read Note 9, Derivative Instruments, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Translation of Foreign Currencies | The functional currency for most of our foreign subsidiaries is their local currency. For our non-U.S. subsidiaries that transact in a functional currency other than the U.S. dollar, assets and liabilities are translated at current rates of exchange at the balance sheet date. Income and expense items are translated at the average foreign currency exchange rates for the period. Adjustments resulting from the translation of the financial statements of our foreign operations into U.S. dollars are excluded from the determination of net income and are recorded in accumulated other comprehensive income, a separate component of equity. For subsidiaries where the functional currency of the assets and liabilities differ from the local currency, non-monetary assets and liabilities are translated at the rate of exchange in effect on the date assets were acquired while monetary assets and liabilities are translated at current rates of exchange as of the balance sheet date. Income and expense items are translated at the average foreign currency rates for the period. Translation adjustments of these subsidiaries are included in other income (expense), net in our consolidated statements of income. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Royalty Cost of Sales | We make royalty payments to a number of third parties under license or purchase agreements associated with our acquisition of intellectual property. These royalty payments are typically calculated as a percentage (royalty rate) of the sales of our products in a particular year. That royalty rate may remain constant, increase or decrease within each year based on the total amount of sales during the annual period. Each quarterly period, we estimate our total royalty obligation for the full year and recognize the proportional amount as cost of sales based on actual quarterly sales as a percentage of full year estimated sales. For example, if the level of net sales in any calendar year increases the royalty rate within the year, we will record our cost of sales at an even rate over the year, based on the estimated blended royalty rate. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Accounting for Share-Based Compensation | Our share-based compensation programs grant awards that have included stock options, restricted stock units that vest based on stock performance known as market stock units (MSUs), performance-vested restricted stock units that settle in cash (CSPUs), time-vested restricted stock units (RSUs), performance-vested restricted stock units that can be settled in cash or shares of our common stock (PUs) at the sole discretion of the Compensation and Management Development Committee of our Board of Directors, performance-vested stock units that settle in stock or cash (PSUs) and shares issued under our employee stock purchase plan (ESPP). Compensation expense is recognized based on the estimated fair value of the awards at grant date. We recognize compensation expense for the number of awards expected to vest after taking into consideration an estimate of award forfeitures over the requisite service period, which is generally the vesting period. Where awards are made with non-substantive vesting periods (for instance, where a portion of the award vests upon retirement eligibility), we estimate and recognize expense based on the period from the grant date to the date the employee becomes retirement eligible. The fair values of our MSUs are estimated using a lattice model with a Monte Carlo simulation. We apply an accelerated attribution method to recognize share-based compensation expense over the applicable service period for our MSUs. The probability of actual shares expected to be earned is considered in the grant date valuation, therefore the expense is not adjusted to reflect the actual units earned. The fair values of our RSUs are based on the market value of our stock on the date of grant. Compensation expense for RSUs is recognized straight-line over the applicable service period. We apply an accelerated attribution method to recognize share-based compensation expense when accounting for our CSPUs, PUs and PSUs that settle in cash, and the fair value of the liability is remeasured at the end of each reporting period through expected settlement. Compensation expense associated with CSPUs, PUs and PSUs that settle in cash are based upon the stock price and the number of units expected to be earned after assessing the probability that certain performance criteria will be met and the targeted payout level associated with the performance criteria expected to be achieved. Cumulative adjustments are recorded each quarter to reflect changes in the stock price and estimated outcome of the performance-related conditions until the date results are determined and settled. If performance criteria are not met or not expected to be met, any compensation expense previously recognized to date associated with the awards will be reversed. The fair values of PSUs that settle in stock are based upon the stock price on the date of grant. Compensation expense is recognized for the number of units expected to be earned after assessing the probability that certain performance criteria will be met and the targeted payout level associated with the performance criteria expected to be achieved. Cumulative adjustments are recorded each quarter to reflect the estimated outcome of the performance-related conditions until the date results are determined and settled. If performance criteria are not met or not expected to be met, any compensation expense previously recognized to date associated with the awards will be reversed.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Research and Development Expenses | Research and development expenses consist of expenses incurred in performing research and development activities, which include compensation and benefits, facilities and overhead expenses, clinical trial expenses and fees paid to contract research organizations (CROs), clinical supply and manufacturing expenses, write-offs of inventory that was previously capitalized in anticipation of product launch and determined to no longer be realizable and other outside expenses and upfront fees and milestones paid to third-party collaborators. Research and development expenses are expensed as incurred. Upfront and milestone payments made to third-party collaborators are expensed as incurred up to the point of regulatory approval. Milestone payments made upon regulatory approval are capitalized and amortized over the remaining useful life of the related product. Payments we make for research and development services prior to the services being rendered are recorded as prepaid assets in our consolidated balance sheets and are expensed as the services are provided. We also accrue the costs of ongoing clinical trials associated with programs that have been terminated or discontinued for which there is no future economic benefit at the time the decision is made to terminate or discontinue the program. From time to time, we enter into development agreements in which we share expenses with a collaborative partner. We record payments received from our collaborative partners for their share of the development costs as a reduction of research and development expense, except as discussed in Note 18, Collaborative and Other Relationships, to these consolidated financial statements. Because an initial indication has been approved for both RITUXAN and GAZYVA, expenses incurred by Genentech in the ongoing development of RITUXAN and GAZYVA are not recorded as research and development expense, but rather reduce our share of profits recorded as a component of revenues from anti-CD20 therapeutic programs. For collaborations with commercialized products, if we are the principal, we record revenues and the corresponding operating costs in their respective line items in our consolidated statements of income. If we are not the principal, we record operating costs as a reduction of revenue.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Selling, General and Administrative Expenses | Selling, general and administrative expenses are primarily comprised of compensation and benefits associated with sales and marketing, finance, human resources, legal, information technology and other administrative personnel, outside marketing, advertising and legal expenses and other general and administrative costs. Advertising costs are expensed as incurred. For the years ended December 31, 2020, 2019 and 2018, advertising costs totaled $111.8 million, $79.2 million and $90.2 million, respectively.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Income Taxes | The provision for income taxes includes federal, state, local and foreign taxes. Income taxes are accounted for under the asset and liability method. Deferred tax assets and liabilities are recognized for the estimated future tax consequences of temporary differences between the financial statement carrying amounts and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the year in which the temporary differences are expected to be recovered or settled. We evaluate the realizability of our deferred tax assets and establish a valuation allowance when it is more likely than not that all or a portion of deferred tax assets will not be realized. We recognize deferred taxes associated with our global intangible low-taxed income (GILTI) tax calculations. The income tax consequences from the intra-entity transfers of inventory within our consolidated group, both current and deferred, are recorded as a prepaid tax or deferred charge and recognized through our consolidated statements of income when the inventory is sold to a third party. In October 2016 the FASB issued ASU No. 2016-16, Income Taxes (Topic 740): Intra-Entity Transfer of Assets Other Than Inventory. This standard eliminates the deferral of the tax effects of intra-entity asset transfers other than inventory. As a result, the income tax consequences from the intra-entity transfer of an asset other than inventory and associated changes to deferred taxes will be recognized when the transfer occurs. We adopted this standard on January 1, 2018, using the modified retrospective method, through a cumulative-effect adjustment to retained earnings as of that date. Upon adoption, we recognized additional net deferred tax assets of approximately $0.5 billion, offset by a corresponding net increase to retained earnings of approximately $0.5 billion. In the fourth quarter of 2018, when we elected to begin recognizing deferred taxes on the GILTI tax calculation, we recorded an additional deferred tax liability of $0.4 billion with a corresponding reduction to our retained earnings as these differences are related to intra-entity transactions. We will recognize incremental deferred income tax expense thereafter as these deferred tax assets and liabilities are utilized. We account for uncertain tax positions using a “more likely than not” threshold for recognizing and resolving uncertain tax positions. We evaluate uncertain tax positions on a quarterly basis and consider various factors including, but not limited to, changes in tax law, the measurement of tax positions taken or expected to be taken in tax returns, the effective settlement of matters subject to audit, information obtained during in process audit activities and changes in facts or circumstances related to a tax position. We also accrue for potential interest and penalties related to unrecognized tax benefits in income tax expense.
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Contingencies | We are currently involved in various claims and legal proceedings. Loss contingency provisions are recorded if the potential loss from any claim, asserted or unasserted, or legal proceeding is considered probable and the amount can be reasonably estimated or a range of loss can be determined. These accruals represent management’s best estimate of probable loss. Disclosure also is provided when it is reasonably possible that a loss will be incurred or when it is reasonably possible that the amount of a loss will exceed the recorded provision. On a quarterly basis, we review the status of each significant matter and assess its potential financial exposure. Significant judgment is required in both the determination of probability and as to whether an exposure is reasonably estimable. Because of uncertainties related to these matters, accruals are based only on the best information available at the time. As additional information becomes available, we reassess the potential liability related to pending claims and litigation and may change our estimates. Legal costs associated with legal proceedings are expensed when incurred. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Earnings per Share | Basic earnings per share is computed by dividing undistributed net income attributable to Biogen Inc. by the weighted-average number of common shares outstanding during the period. Diluted earnings per share is computed based on the treasury method by dividing net income by the weighted-average number of common shares outstanding during the period plus potentially dilutive common equivalent shares outstanding. | ||||||||||||||||||||||||||||||||||||||||||||||||
| New Accounting Pronouncements | From time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that we adopt as of the specified effective date. Unless otherwise discussed below, we do not believe that the adoption of recently issued standards have or may have a material impact on our consolidated financial statements or disclosures. Leases In February 2016 the FASB issued the new leasing standards to increase transparency and comparability among organizations related to their leasing activities. For additional information on the adoption of the new leasing standards, please read the section titled Lease above, and Note 11, Leases, to these consolidated financial statements. Credit Losses In June 2016 the FASB issued ASU No. 2016-13, Financial Instruments - Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments. The FASB subsequently issued amendments to ASU 2016-13, which have the same effective date and transition date of January 1, 2020. These standards require that credit losses be reported using an expected losses model rather than the incurred losses model that is currently used, and establishes additional disclosures related to credit risks. For available-for-sale debt securities with unrealized losses, these standards now require allowances to be recorded instead of reducing the amortized cost of the investment. These standards limit the amount of credit losses to be recognized for available-for-sale debt securities to the amount by which carrying value exceeds fair value and requires the reversal of previously recognized credit losses if fair value increases. Based on the composition of our investment portfolio, accounts receivable and other financial assets, current market conditions and historical credit loss activity, the adoption of these standards did not have a material impact on our consolidated financial position and results of operations and related disclosures. Debt Securities In March 2017 the FASB issued ASU No. 2017-08, Receivables - Nonrefundable Fees and Other Costs (Subtopic 310-20): Premium Amortization on Purchased Callable Debt Securities. This standard amends the amortization period for certain purchased callable debt securities held at a premium by shortening the amortization period to the earliest call date. This standard became effective for us on January 1, 2019, and was adopted using a modified retrospective transition approach. The adoption of this standard did not result in a significant adjustment to our marketable debt securities. Fair Value Measurements In August 2018 the FASB issued ASU No. 2018-13, Fair Value Measurement (Topic 820): Disclosure Framework - Changes to the Disclosure Requirements for Fair Value Measurement. This standard modifies certain disclosure requirements on fair value measurements. This standard became effective for us on January 1, 2020. The adoption of this standard did not have a material impact on our disclosures. Derivative Instruments and Hedging Activities In October 2018 the FASB issued ASU No. 2018-16, Derivatives and Hedging (Topic 815): Inclusion of the Secured Overnight Financing Rate (SOFR) Overnight Index Swap (OIS) Rate as a Benchmark Interest Rate for Hedge Accounting Purposes. This standard permits use of the OIS rate based on the SOFR as a U.S. benchmark interest rate for hedge accounting purposes under ASC 815, Derivatives and Hedging. This standard became effective for us on January 1, 2019, and did not have an impact on our consolidated results of operations or financial position. Collaborative Arrangements In November 2018 the FASB issued ASU No. 2018-18, Collaborative Arrangements (Topic 808): Clarifying the Interaction between Topic 808 and Topic 606. This standard makes targeted improvements for collaborative arrangements as follows: •Clarifies that certain transactions between collaborative arrangement participants should be accounted for as revenue under ASC 606, Revenue from Contracts with Customers, when the collaborative arrangement participant is a customer in the context of a unit of account. In those situations, all the guidance in ASC 606 should be applied, including recognition, measurement, presentation and disclosure requirements; •Adds unit-of-account guidance to ASC 808, Collaborative Arrangements, to align with the guidance in ASC 606 (that is, a distinct good or service) when an entity is assessing whether the collaborative arrangement or a part of the arrangement is within the scope of ASC 606; and •Precludes a company from presenting transactions with collaborative arrangement participants that are not directly related to sales to third parties with revenue recognized under ASC 606 if the collaborative arrangement participant is not a customer. This standard became effective for us on January 1, 2020. A retrospective transition approach is required for either all contracts or only for contracts that are not completed at the date of initial application of ASC 606, with a cumulative adjustment to opening retained earnings, as of January 1, 2018. The adoption of this standard did not have a material impact on our consolidated financial position, results of operations and related disclosures.
|
||||||||||||||||||||||||||||||||||||||||||||||||
Summary of Significant Accounting Policies (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Accounting Policies [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||
| Property, plant and equipment estimated useful lives | We generally depreciate or amortize the cost of our property, plant and equipment using the straight-line method over the estimated useful lives of the respective assets, which are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||
Revenues (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revenue from Contract with Customer [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revenue by product | Revenues by product are summarized as follows:
*Fumarate includes TECFIDERA and VUMERITY. VUMERITY became commercially available in the U.S. in November 2019. **Interferon includes AVONEX and PLEGRIDY.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Analysis of change in reserves | An analysis of the change in reserves for discounts and allowances is summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total reserves included in consolidated balance sheet | The total reserves above, which are included in our consolidated balance sheets, are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | Revenues from anti-CD20 therapeutic programs are summarized in the table below. For purposes of this footnote, we refer to RITUXAN and RITUXAN HYCELA collectively as RITUXAN.
Revenues from anti-CD20 therapeutic programs are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other revenues | Other revenues are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Inventory (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inventory Disclosure [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of inventory | The components of inventory are summarized as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Intangible Assets and Goodwill (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Goodwill and Intangible Assets Disclosure [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intangible assets | Intangible assets, net of accumulated amortization, impairment charges and adjustments are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Estimated future amortization of intangible assets | The estimated future amortization of finite-lived intangible assets for the next five years is expected to be as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of roll forward of the changes in goodwill | The following table provides a roll forward of the changes in our goodwill balance:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Fair Value Measurements (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair Value Disclosures [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of assets and liabilities recorded at fair value | The tables below present information about our assets and liabilities that are regularly measured and carried at fair value and indicate the level within the fair value hierarchy of the valuation techniques we utilized to determine such fair value:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of significant unobservable inputs | The following table summarizes the significant unobservable inputs in the fair value measurement of our contingent consideration obligations as of December 31, 2020:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of gains or losses on assets measured at fair value, nonrecurring basis | The gains or losses on assets measured at fair value on a nonrecurring basis, are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of fair and carrying value of debt instruments | The fair values of our debt instruments, which are Level 2 liabilities, are summarized as follows:
(1) Our 2.900% Senior Notes due September 15, 2020, were redeemed in full in May 2020 using the net proceeds from the issuance on April 30, 2020, of our senior unsecured notes for an aggregate principal amount of $3.0 billion. For additional information, please read Note 12, Indebtedness, to these consolidated financial statements.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair value of contingent consideration obligations | The following table provides a roll forward of the fair values of our contingent consideration obligations, which includes Level 3 measurements:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Financial Instruments (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Investments, All Other Investments [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of financial assets with maturities of less than 90 days included within cash and cash equivalents | The following table summarizes our financial assets with maturities of less than 90 days from the date of purchase included in cash and cash equivalents in our consolidated balance sheets:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Debt and equity securities, available-for-sale | The following tables summarize our marketable debt and equity securities, classified as available for sale:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of contractual maturities: available-for-sale securities | The estimated fair value and amortized cost of our marketable debt securities available-for-sale by contractual maturity are summarized as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Proceeds from marketable securities, excluding strategic investments | The proceeds from maturities and sales of marketable debt securities and resulting realized gains and losses are summarized as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Derivative Instruments (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Derivative Instruments and Hedging Activities Disclosure [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency forward contracts that were entered into to hedge forecasted revenue | The notional value of foreign currency forward contracts that were entered into to hedge forecasted revenues and operating expenses is summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of the effect of derivatives designated as cash flow hedging instruments on our consolidated statements of income | The following table summarizes the effect of foreign currency forward contracts designated as hedging instruments in our consolidated statements of income:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of the effect of derivatives designated as net investment hedging instruments on our consolidated statement of income | The following table summarizes the effect of our net investment hedges in our consolidated financial statements:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of the fair value for our outstanding derivatives | The following table summarizes the fair value and presentation in our consolidated balance sheets of our outstanding derivative instruments, including those designated as hedging instruments:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Property, Plant and Equipment (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Property, Plant and Equipment [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of property, plant and equipment, net | Components of property, plant and equipment, net are summarized as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Leases (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Leases [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Schedule of operating leases | All of our leases qualify as operating leases. The following table summarizes the presentation in our consolidated balance sheets of our operating leases:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Operating lease costs | The following table summarizes the effect of lease costs in our consolidated statements of income:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Operating lease liability maturity | The minimum lease payments for the next five years and thereafter is expected to be as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Operating lease weighted average remaining term and discount rate | The weighted average remaining lease term and weighted average discount rate of our operating leases are as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Operating lease supplemental cash flow disclosure | Supplemental disclosure of cash flow information related to our operating leases included in cash flows provided by operating activities in our consolidated statements of cash flows is as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Indebtedness (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Debt Disclosure [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of indebtedness | Our indebtedness is summarized as follows:
(1) Our 2.900% Senior Notes due September 15, 2020, were redeemed in full in May 2020 using the net proceeds from the issuance on April 30, 2020, of our senior unsecured notes for an aggregate principal amount of $3.0 billion, as discussed below.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total debt maturities | The total gross payments due under our debt arrangements are as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Equity (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equity [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of common stock | The following table describes the number of shares authorized, issued and outstanding of our common stock as of December 31, 2020, 2019 and 2018:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Schedule of Accumulated Other Comprehensive Income (Loss) | The following tables summarize the changes in accumulated other comprehensive income (loss), net of tax by component:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reclassification out of Accumulated Other Comprehensive Income | The following table summarizes the amounts reclassified from accumulated other comprehensive income:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Earnings per Share (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Earnings Per Share [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Basic and diluted earnings per share | Basic and diluted earnings per share are calculated as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Share-Based Payments (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based Payment Arrangement [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation expense included in consolidated statements of income | The following table summarizes share-based compensation expense included in our consolidated statements of income:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of share-based compensation expense associated with each of our share-based compensating programs | The following table summarizes share-based compensation expense associated with each of our share-based compensation programs:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock option activity | The following table summarizes our stock option activity:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tax benefit and cash received from stock option exercises | The following table summarizes the amount of tax benefit realized for stock options and cash received from the exercise of stock options:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Market stock units activity | The following table summarizes our MSU activity:
(1) MSUs granted during 2020 include awards granted in conjunction with our annual awards made in February 2020 and MSUs granted in conjunction with the hiring of employees. These grants reflect the target number of shares eligible to be earned at the time of grant. MSUs granted in 2020 also reflect an adjustment based upon the final performance multiplier in relation to shares granted in 2019, 2018 and 2017.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Assumptions used in valuation of market based stock units | The assumptions used in our valuation are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash settled performance shares activity | The following table summarizes our CSPU activity:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Performance units activity | The following table summarizes our PU activity:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Performance stock units settled in stock activity | The following table summarizes our PSUs that settle in stock activity:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Performance stock units settled in cash activity | The following table summarizes our PSUs that settle in cash activity:
(1) PSUs settled in cash granted in 2020 include awards granted in conjunction with our annual awards made in February 2020 and PSUs granted in conjunction with the hiring of employees. These grants reflect the target number of shares eligible to be earned at the time of grant.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time-vested restricted stock units activity | The following table summarizes our RSU activity:
(1) RSUs granted in 2020 primarily represent RSUs granted in conjunction with our annual awards made in February 2020 and awards made in conjunction with the hiring of new employees. RSUs granted in 2020 also include approximately 10,000 RSUs granted to our Board of Directors.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shares issued under employee stock purchase plan | The following table summarizes our ESPP activity:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Income Taxes (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income Tax Disclosure [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income before income tax provision and the income tax expense | Income before income tax expense and the income tax expense consist of the following:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of deferred tax assets and liabilities | Significant components of our deferred tax assets and liabilities are summarized as follows:
The change in the valuation allowance between December 31, 2020 and 2019, was primarily related to the establishment of a valuation allowance against certain deferred tax assets, the realization of which is dependent on future sales of TECFIDERA in the U.S., as discussed above.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reconciliation between the U.S. federal statutory tax rate and effective tax rate | A reconciliation between the U.S. federal statutory tax rate and our effective tax rate is summarized as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reconciliation of beginning and ending amount of unrecognized tax benefits | A reconciliation of the beginning and ending amount of our unrecognized tax benefits is summarized as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Other Consolidated Financial Statement Detail (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Organization, Consolidation and Presentation of Financial Statements [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Supplemental cash flow information | Supplemental disclosure of cash flow information for the years ended December 31, 2020, 2019 and 2018, is as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other income (expense), net | Components of other income (expense), net, are summarized as follows:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gain (loss) on investments | The following table summarizes our gain (loss) on investments, net that relates to our equity securities held as of December 31, 2020, 2019 and 2018:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accrued expenses and other | Accrued expenses and other consists of the following:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Collaborative and Other Relationships (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Organization, Consolidation and Presentation of Financial Statements [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Co-promotion profit sharing formula | Our share of annual co-promotion profits in excess of $50.0 million varies, as summarized in the table below, upon the following events:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pretax profit sharing formula | Our share of annual profits in excess of $50.0 million varies, as summarized in the table below, upon the following events:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revenues from anti-CD20 therapeutic programs | Revenues from anti-CD20 therapeutic programs are summarized in the table below. For purposes of this footnote, we refer to RITUXAN and RITUXAN HYCELA collectively as RITUXAN.
Revenues from anti-CD20 therapeutic programs are summarized as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of activity related to BAN2401 and Elenbecestat collaboration | A summary of development and sales and marketing expenses related to the BAN2401 Collaboration is as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of activity related to Aducanumab collaboration | A summary of development, sales and marketing and milestone expense related to the Aducanumab Collaboration Agreement is as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Summary of activity related to the UCB collaboration | A summary of development expense related to the UCB collaboration agreement is as follows:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Segment Information (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Segment Reporting [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Geographic information | The following tables contain certain financial information by geographic area:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quarterly Financial Data (Unaudited) (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quarterly Financial Information Disclosure [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quarterly Financial Data (Unaudited) |
(1) Gross profit is calculated as total revenues less cost of sales, excluding amortization and impairment of acquired intangible assets.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Divestitures (Details) - USD ($) $ in Millions |
12 Months Ended | |||
|---|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Aug. 31, 2019 |
|
| Income Statement, Balance Sheet and Additional Disclosures by Disposal Groups, Including Discontinued Operations [Line Items] | ||||
| (Gain) loss on divestiture of Hillerød, Denmark manufacturing operations | $ 92.5 | $ (55.3) | $ 0.0 | |
| Inventory raw materials sold to FUJIFILM | $ 41.8 | |||
| Denmark Manufacturing Operations | ||||
| Income Statement, Balance Sheet and Additional Disclosures by Disposal Groups, Including Discontinued Operations [Line Items] | ||||
| Consideration received for sale of Denmark manufacturing operations | $ 881.9 | |||
| Loss on divestiture of Hillerød, Denmark manufacturing operations, net of tax | 124.2 | |||
| (Gain) loss on divestiture of Hillerød, Denmark manufacturing operations | 30.5 | (55.3) | ||
| Expected costs to sell disposal group | 11.2 | |||
| Future minimum batch production for Denmark manufacturing operations | 74.0 | |||
| Tax expense on disposal group | $ 68.9 | |||
| Purchase commitment, reduction | $ 62.0 | |||
Revenues - Total Reserves for Discounts and Allowances (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Dec. 31, 2017 |
|---|---|---|---|---|
| SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Line Items] | ||||
| Revenue-related reserves for discounts and allowances | $ 1,276.0 | $ 1,198.9 | $ 1,051.3 | $ 761.6 |
| Reduction of accounts receivable | ||||
| SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Line Items] | ||||
| Revenue-related reserves for discounts and allowances | 195.4 | 197.8 | ||
| Component of accrued expenses and other | ||||
| SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Line Items] | ||||
| Revenue-related reserves for discounts and allowances | $ 1,080.6 | $ 1,001.1 |
Revenues - Revenues from Anti-CD20 Therapeutic Programs (Details) - USD ($) $ in Millions |
3 Months Ended | 12 Months Ended | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Sep. 30, 2020 |
Jun. 30, 2020 |
Mar. 31, 2020 |
Dec. 31, 2019 |
Sep. 30, 2019 |
Jun. 30, 2019 |
Mar. 31, 2019 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues from anti-CD20 therapeutic programs | $ 2,852.6 | $ 3,376.1 | $ 3,681.6 | $ 3,534.3 | $ 3,671.3 | $ 3,600.1 | $ 3,616.7 | $ 3,489.8 | $ 13,444.6 | $ 14,377.9 | $ 13,452.9 |
| Revenues from anti-CD20 therapeutic programs | |||||||||||
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues from anti-CD20 therapeutic programs | $ 419.0 | $ 560.1 | $ 478.3 | $ 520.4 | $ 600.8 | $ 595.8 | $ 576.4 | $ 517.4 | 1,977.8 | 2,290.4 | 1,980.2 |
| Genentech | |||||||||||
| Disaggregation of Revenue [Line Items] | |||||||||||
| Biogen's share of pre-tax profits in the U.S. for RITUXAN and GAZYVA | 1,080.2 | 1,542.4 | 1,431.9 | ||||||||
| Other revenues from anti-CD20 therapeutic programs | $ 897.6 | $ 748.0 | $ 548.3 | ||||||||
Revenues - Other Revenues (Details) - USD ($) $ in Millions |
3 Months Ended | 12 Months Ended | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Sep. 30, 2020 |
Jun. 30, 2020 |
Mar. 31, 2020 |
Dec. 31, 2019 |
Sep. 30, 2019 |
Jun. 30, 2019 |
Mar. 31, 2019 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues | $ 2,852.6 | $ 3,376.1 | $ 3,681.6 | $ 3,534.3 | $ 3,671.3 | $ 3,600.1 | $ 3,616.7 | $ 3,489.8 | $ 13,444.6 | $ 14,377.9 | $ 13,452.9 |
| Royalty | |||||||||||
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues | 33.9 | 17.0 | 38.7 | ||||||||
| Other corporate | |||||||||||
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues | 719.1 | 584.5 | 459.4 | ||||||||
| Total other revenues | |||||||||||
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues | $ 132.0 | $ 125.7 | $ 407.6 | $ 109.3 | $ 145.7 | $ 109.6 | $ 160.0 | $ 292.4 | 774.6 | 707.7 | 585.9 |
| Collaborative Arrangement | Samsung Bioepis | |||||||||||
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues | 20.9 | 106.2 | 96.4 | ||||||||
| Collaborative Arrangement | Other Relationships | |||||||||||
| Disaggregation of Revenue [Line Items] | |||||||||||
| Total revenues | $ 0.7 | $ 0.0 | $ (8.6) | ||||||||
Inventory - Components of Inventory (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Inventory Disclosure [Abstract] | ||
| Raw materials | $ 314.9 | $ 169.7 |
| Work in process | 544.5 | 460.0 |
| Finished goods | 209.2 | 174.5 |
| Total inventory | $ 1,068.6 | $ 804.2 |
Inventory - Narrative (Details) - USD ($) $ in Millions |
12 Months Ended | |||
|---|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Aug. 31, 2019 |
|
| Inventory [Line Items] | ||||
| Write-downs on excess, obsolete, unmarketable or other inventory | $ 26.6 | $ 52.2 | $ 41.9 | |
| Inventory raw materials sold to FUJIFILM | $ 41.8 | |||
| Work-in-process | ||||
| Inventory [Line Items] | ||||
| Inventory work-in-process sold to FUJIFILM | $ 14.0 | |||
Intangible Assets and Goodwill - Summary of Intangible Assets (Details) - USD ($) $ in Millions |
12 Months Ended | |
|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
|
| Finite-Lived Intangible Assets [Line Items] | ||
| Cost | $ 7,394.3 | $ 7,379.3 |
| Total intangible assets | 8,220.8 | 8,408.8 |
| Accumulated Amortization | (5,136.5) | (4,881.4) |
| Net | 2,257.8 | 2,497.9 |
| Total intangible assets | $ 3,084.3 | 3,527.4 |
| Minimum | ||
| Finite-Lived Intangible Assets [Line Items] | ||
| Estimated life (in years) | 4 years | |
| Maximum | ||
| Finite-Lived Intangible Assets [Line Items] | ||
| Estimated life (in years) | 28 years | |
| In-process research and development | ||
| Finite-Lived Intangible Assets [Line Items] | ||
| Cost and net | $ 762.5 | 965.5 |
| Trademarks and trade names | ||
| Finite-Lived Intangible Assets [Line Items] | ||
| Cost and net | $ 64.0 | $ 64.0 |
Intangible Assets and Goodwill - Estimated Future Amortization of Intangible Assets (Details) $ in Millions |
Dec. 31, 2020
USD ($)
|
|---|---|
| Goodwill and Intangible Assets Disclosure [Abstract] | |
| 2021 | $ 205.0 |
| 2022 | 215.0 |
| 2023 | 215.0 |
| 2024 | 225.0 |
| 2025 | $ 220.0 |
Intangible Assets and Goodwill - Changes in Goodwill (Details) - USD ($) $ in Millions |
12 Months Ended | |
|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
|
| Goodwill [Roll Forward] | ||
| Goodwill, beginning of year | $ 5,757.8 | $ 5,706.4 |
| Increase to goodwill | 0.0 | 117.5 |
| Elimination of goodwill allocated to Hillerød, Denmark manufacturing operations | 0.0 | (69.5) |
| Other | 4.3 | 3.4 |
| Goodwill, end of year | $ 5,762.1 | $ 5,757.8 |
Fair Value Measurements - Summary of Significant Unobservable Inputs (Details) $ in Millions |
Dec. 31, 2020
USD ($)
|
Dec. 31, 2019
USD ($)
|
Dec. 31, 2018
USD ($)
|
|---|---|---|---|
| Discount rate | |||
| Fair Value, Assets and Liabilities Measured on Recurring and Nonrecurring Basis [Line Items] | |||
| Unobservable Input | 0.0060 | ||
| Weighted Average | Discount rate | |||
| Fair Value, Assets and Liabilities Measured on Recurring and Nonrecurring Basis [Line Items] | |||
| Unobservable Input | 0.0060 | ||
| Fair Value, Recurring | |||
| Fair Value, Assets and Liabilities Measured on Recurring and Nonrecurring Basis [Line Items] | |||
| Fair Value | $ 259.8 | $ 346.1 | $ 409.8 |
Fair Value Measurements - Gains and Losses on Assets Measured at Fair Value (Details) $ in Millions |
7 Months Ended | 12 Months Ended | |
|---|---|---|---|
|
Dec. 31, 2020
USD ($)
|
Dec. 31, 2019
USD ($)
|
Jun. 07, 2019
USD ($)
|
|
| Finite-lived Intangible Assets [Roll Forward] | |||
| Beginning Book Value | $ 8,220.8 | $ 8,408.8 | |
| Ending Book Value | 3,084.3 | $ 3,527.4 | |
| BIIB111 | Fair Value, Nonrecurring | |||
| Finite-lived Intangible Assets [Roll Forward] | |||
| Beginning Book Value | $ 480.0 | ||
| Impairment | (115.0) | ||
| Ending Book Value | $ 365.0 |
Fair Value Measurements - Fair Value of Contingent Consideration Obligations (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation, Calculation [Roll Forward] | |||
| Contingent consideration | $ (86.3) | $ (63.7) | $ (12.3) |
| Fair value, measurements, recurring | |||
| Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation, Calculation [Roll Forward] | |||
| Fair value, beginning of year | 346.1 | 409.8 | |
| Contingent consideration | (86.3) | (63.7) | |
| Payments and other | 0.0 | 0.0 | |
| Fair value, end of year | $ 259.8 | $ 346.1 | $ 409.8 |
Financial Instruments - Summary of Financial Assets with Maturities (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Cash and Cash Equivalents [Line Items] | ||
| Cash equivalents | $ 626.9 | $ 2,541.1 |
| Commercial paper | ||
| Cash and Cash Equivalents [Line Items] | ||
| Cash equivalents | 61.1 | 384.4 |
| Overnight reverse repurchase agreements | ||
| Cash and Cash Equivalents [Line Items] | ||
| Cash equivalents | 37.4 | 368.8 |
| Money market funds | ||
| Cash and Cash Equivalents [Line Items] | ||
| Cash equivalents | 505.1 | 1,628.5 |
| Short-term debt securities | ||
| Cash and Cash Equivalents [Line Items] | ||
| Cash equivalents | $ 23.3 | $ 159.4 |
Financial Instruments - Summary of Contractual Maturities (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Estimated Fair Value | ||
| Due in one year or less | $ 1,278.9 | $ 1,562.2 |
| Due after one year through five years | 722.6 | 1,234.5 |
| Due after five years | 49.5 | 173.6 |
| Total marketable debt securities | 2,051.0 | 2,970.3 |
| Amortized Cost | ||
| Due in one year or less | 1,278.6 | 1,560.8 |
| Due after one year through five years | 721.3 | 1,230.4 |
| Due after five years | 49.2 | 173.8 |
| Amortized Cost | $ 2,049.1 | $ 2,965.0 |
Financial Instruments - Proceeds from Marketable Debt Securities (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Investments, All Other Investments [Abstract] | |||
| Proceeds from maturities and sales | $ 7,299.4 | $ 6,007.0 | $ 9,173.7 |
| Realized gains | 17.7 | 6.0 | 3.2 |
| Realized losses | $ 26.0 | $ 1.5 | $ 11.7 |
Derivative Instruments - Foreign Currency Forward Contracts Entered into Hedge Forecasted Revenues (Details) - Designated as Hedging Instrument - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Derivatives, Fair Value [Line Items] | ||
| Derivative, notional amount | $ 3,229.7 | $ 1,892.4 |
| Euro | ||
| Derivatives, Fair Value [Line Items] | ||
| Derivative, notional amount | 2,979.1 | 1,892.4 |
| British pound | ||
| Derivatives, Fair Value [Line Items] | ||
| Derivative, notional amount | $ 250.6 | $ 0.0 |
Derivative Instruments - Summary of the Effect of Derivatives Designated as Cash Flow Hedging Instruments (Details) - Foreign exchange contract - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Revenues | |||
| Derivative Instruments, Gain (Loss) [Line Items] | |||
| Net Gains/(Losses) Reclassified from AOCI into Operating Income (in millions) | $ 18.3 | $ 118.6 | $ (42.5) |
| Operating expenses | |||
| Derivative Instruments, Gain (Loss) [Line Items] | |||
| Net Gains/(Losses) Reclassified from AOCI into Operating Income (in millions) | 3.3 | (3.3) | 0.2 |
| Cash flows, revenue | Revenues | |||
| Derivative Instruments, Gain (Loss) [Line Items] | |||
| Gain (loss) on derivative instruments, net, pretax | (9.9) | 2.9 | 10.8 |
| Cash flows, operating expenses | Operating expenses | |||
| Derivative Instruments, Gain (Loss) [Line Items] | |||
| Gain (loss) on derivative instruments, net, pretax | $ 0.0 | $ 0.2 | $ (0.1) |
Derivative Instruments - Summary of the Effect of Derivatives Designated as Net Investment Hedging Instruments (Details) - Net investment hedge - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Derivative Instruments, Gain (Loss) [Line Items] | |||
| Net Gains/(Losses) Recognized in Other Comprehensive Income (Effective Portion) (in millions) | $ (35.1) | $ 25.3 | $ (3.8) |
| Net Gains/(Losses) Recognized in Other Comprehensive Income (Amounts Excluded from Effectiveness Testing) (in millions) | 4.5 | 3.3 | 0.0 |
| Other (income) expense, net | |||
| Derivative Instruments, Gain (Loss) [Line Items] | |||
| Net Gains/(Losses) Recognized in Net Income (Amounts Excluded from Effectiveness Testing) (in millions) | $ 2.9 | $ 7.0 | $ 1.5 |
Property, Plant and Equipment (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Property, Plant and Equipment [Abstract] | ||
| Land | $ 119.8 | $ 118.1 |
| Buildings | 1,025.3 | 835.0 |
| Leasehold improvements | 104.6 | 99.5 |
| Machinery and equipment | 1,027.8 | 844.5 |
| Computer software and hardware | 903.0 | 798.4 |
| Furniture and fixtures | 62.5 | 58.3 |
| Construction in progress | 1,950.8 | 2,084.4 |
| Total cost | 5,193.8 | 4,838.2 |
| Less: accumulated depreciation | (1,782.3) | (1,590.9) |
| Total property, plant and equipment, net | $ 3,411.5 | $ 3,247.3 |
Leases - Narrative (Details) |
Dec. 31, 2020 |
|---|---|
| Minimum | |
| Operating Leased Assets [Line Items] | |
| Operating lease, contract term | 1 year |
| Operating lease, renewal term | 1 year |
| Minimum | Sublease | |
| Operating Leased Assets [Line Items] | |
| Operating lease, contract term | 4 years |
| Maximum | |
| Operating Leased Assets [Line Items] | |
| Operating lease, contract term | 10 years |
| Operating lease, renewal term | 6 years |
| Maximum | Sublease | |
| Operating Leased Assets [Line Items] | |
| Operating lease, contract term | 8 years |
Leases - Schedule of Operating Leases (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Operating lease assets | $ 433.3 | $ 427.0 |
| Operating Lease, Liability, Current, Statement of Financial Position [Extensible List] | us-gaap:OtherLiabilitiesCurrent | |
| Long-term operating lease liabilities | $ 402.0 | 412.7 |
| Present value of operating lease liabilities | 485.2 | 486.3 |
| Operating lease assets | ||
| Operating lease assets | 433.3 | 427.0 |
| Accrued expenses and other | ||
| Operating lease liability, current | 83.2 | 73.6 |
| Long-term operating lease liabilities | ||
| Long-term operating lease liabilities | $ 402.0 | $ 412.7 |
Leases - Operating Lease Costs (Details) - USD ($) $ in Millions |
12 Months Ended | |
|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
|
| Net lease cost | $ 92.4 | $ 86.7 |
| Research and development | ||
| Operating lease cost | 5.2 | 6.7 |
| Variable lease cost | 1.1 | 1.2 |
| Selling, general and administrative | ||
| Operating lease cost | 93.1 | 84.6 |
| Variable lease cost | 21.1 | 23.7 |
| Sublease income | (24.2) | (25.6) |
| Other (income) expense, net | ||
| Sublease income | $ (3.9) | $ (3.9) |
Leases - Operating Lease Liability Maturity (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Leases [Abstract] | ||
| 2021 | $ 95.8 | |
| 2022 | 93.5 | |
| 2023 | 82.0 | |
| 2024 | 75.0 | |
| 2025 | 56.9 | |
| Thereafter | 129.8 | |
| Total lease payments | 533.0 | |
| Less: interest | 47.8 | |
| Present value of operating lease liabilities | $ 485.2 | $ 486.3 |
Leases - Operating Lease Weighted Average Remaining Term and Discount Rate (Details) |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Leases [Abstract] | ||
| Weighted average remaining lease term in years | 6 years 3 months 18 days | 7 years 25 days |
| Weighted average discount rate | 2.90% | 3.20% |
Leases - Operating Lease Supplemental Cash Flow Disclosure (Details) - USD ($) $ in Millions |
12 Months Ended | |
|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
|
| Leases [Abstract] | ||
| Cash paid for amounts included in the measurement of lease liabilities | $ 100.2 | $ 93.8 |
| Operating lease assets obtained in exchange for lease obligations | $ 59.0 | $ 35.9 |
Indebtedness - Total Debt Maturities (Details) $ in Millions |
Dec. 31, 2020
USD ($)
|
|---|---|
| Debt Disclosure [Abstract] | |
| 2021 | $ 0.0 |
| 2022 | 1,000.0 |
| 2023 | 0.0 |
| 2024 | 0.0 |
| 2025 | 1,750.0 |
| 2026 and thereafter | 4,750.0 |
| Total | $ 7,500.0 |
Equity - Summary of Common Stock (Details) - shares |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|---|---|---|---|
| Equity [Abstract] | |||
| Common stock authorized (in shares) | 1,000,000,000.0 | 1,000,000,000.0 | 1,000,000,000.0 |
| Common stock issued (in shares) | 176,200,000 | 198,000,000.0 | 221,000,000.0 |
| Common stock outstanding (in shares) | 152,400,000 | 174,200,000 | 197,200,000 |
Earnings per Share - Narrative (Details) - shares shares in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Earnings Per Share [Abstract] | |||
| Repurchase of common stock (in shares) | 22.4 | 23.6 | 14.8 |
Share-Based Payments - Stock Option Activity (Details) - Share-based Payment Arrangement, Option shares in Thousands |
12 Months Ended |
|---|---|
|
Dec. 31, 2020
$ / shares
shares
| |
| Shares | |
| Beginning balance, outstanding shares (in shares) | shares | 12 |
| Granted (in shares) | shares | 0 |
| Exercised (in shares) | shares | (12) |
| Cancelled (in shares) | shares | 0 |
| Ending balance, outstanding shares (in shares) | shares | 0 |
| Weighted Average Exercise Price | |
| Beginning balance, weighted average exercise price (in dollars per share) | $ / shares | $ 58.46 |
| Granted (in dollars per share) | $ / shares | 0 |
| Exercised (in dollars per share) | $ / shares | 58.46 |
| Cancelled (in dollars per share) | $ / shares | 0 |
| Ending balance, weighted average exercise price (in dollars per share) | $ / shares | $ 0 |
Share-Based Payments - Tax Benefit and Cash Received from Stock Option Exercises (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Share-based Payment Arrangement [Abstract] | |||
| Tax benefit realized for stock options | $ 2.9 | $ 2.5 | $ 2.2 |
| Cash received from the exercise of stock options | $ 0.7 | $ 0.4 | $ 0.8 |
Share-Based Payments - Market Stock Units Activity (Details) - Market stock units - $ / shares shares in Thousands |
12 Months Ended | |||||
|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
||||
| Shares | ||||||
| Beginning balance, unvested shares (in shares) | 183 | |||||
| Granted (in shares) | [1] | 133 | ||||
| Vested (in shares) | (82) | |||||
| Forfeited (in shares) | (33) | |||||
| Ending balance, unvested shares (in shares) | 201 | 183 | ||||
| Weighted Average Grant Date Fair Value | ||||||
| Beginning balance, weighted average grant date fair value (in dollars per share) | $ 378.09 | |||||
| Granted (in dollars per share) | 398.61 | [1] | $ 378.08 | $ 378.85 | ||
| Vested (in dollars per share) | 375.42 | |||||
| Forfeited (in dollars per share) | 402.62 | |||||
| Ending balance, weighted average grant date fair value (in dollars per share) | $ 388.98 | $ 378.09 | ||||
| ||||||
Share-Based Payments - Assumptions Used in Valuation of Market Based Stock Units (Details) - Market stock units - USD ($) |
12 Months Ended | |||||
|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
||||
| Share-based Compensation Arrangement by Share-based Payment Award | ||||||
| Expected dividend yield | 0.00% | 0.00% | 0.00% | |||
| Minimum range of risk-free interest rates | 1.41% | 2.46% | 1.90% | |||
| Maximum range of risk-free interest rates | 1.48% | 2.53% | 2.30% | |||
| Average stock price on grant date minimum | $ 257.83 | $ 228.59 | $ 279.47 | |||
| Average stock price on grant date maximum | $ 325.40 | $ 331.18 | $ 346.76 | |||
| Granted (in dollars per share) | $ 398.61 | [1] | $ 378.08 | $ 378.85 | ||
| Minimum | ||||||
| Share-based Compensation Arrangement by Share-based Payment Award | ||||||
| Range of expected stock price volatility | 37.80% | 31.20% | 27.50% | |||
| Maximum | ||||||
| Share-based Compensation Arrangement by Share-based Payment Award | ||||||
| Range of expected stock price volatility | 44.10% | 33.60% | 32.40% | |||
| ||||||
Share-Based Payments - Cash Settled Performance Shares Activity (Details) - Cash settled performance units shares in Thousands |
12 Months Ended |
|---|---|
|
Dec. 31, 2020
shares
| |
| Shares | |
| Beginning balance, unvested shares (in shares) | 13,000 |
| Granted (in shares) | 0 |
| Vested (in shares) | (13,000) |
| Forfeited (in shares) | 0 |
| Ending balance, unvested shares (in shares) | 0 |
Share-Based Payments - Performance Units Activity (Details) - Performance units shares in Thousands |
12 Months Ended |
|---|---|
|
Dec. 31, 2020
shares
| |
| Shares | |
| Beginning balance, unvested shares (in shares) | 11,000 |
| Granted (in shares) | 0 |
| Vested (in shares) | (11,000) |
| Forfeited (in shares) | 0 |
| Ending balance, unvested shares (in shares) | 0 |
Share-Based Payments - Performance Stock Units Settled in Stock Activity (Details) - Performance stock units settled in stock shares in Thousands |
12 Months Ended | |||
|---|---|---|---|---|
|
Dec. 31, 2020
$ / shares
shares
| ||||
| Shares | ||||
| Beginning balance, unvested shares (in shares) | shares | 111,000 | |||
| Granted (in shares) | shares | 73,000 | [1] | ||
| Vested (in shares) | shares | 0 | |||
| Forfeited (in shares) | shares | (29,000) | |||
| Ending balance, unvested shares (in shares) | shares | 155,000 | |||
| Weighted Average Grant Date Fair Value | ||||
| Beginning balance, weighted average grant date fair value (in dollars per share) | $ / shares | $ 316.39 | |||
| Granted (in dollars per share) | $ / shares | 293.35 | [1] | ||
| Vested (in dollars per share) | $ / shares | 0 | |||
| Forfeited (in dollars per share) | $ / shares | 323.42 | |||
| Ending balance, weighted average grant date fair value (in dollars per share) | $ / shares | $ 304.19 | |||
| ||||
Share-Based Payments - Performance Stock Units Settled in Cash Activity (Details) - Performance stock units settled in cash shares in Thousands |
12 Months Ended |
|---|---|
|
Dec. 31, 2020
shares
| |
| Shares | |
| Beginning balance, unvested shares (in shares) | 82,000 |
| Granted (in shares) | 63,000 |
| Vested (in shares) | (2,000) |
| Forfeited (in shares) | (23,000) |
| Ending balance, unvested shares (in shares) | 120,000 |
Share-Based Payments - Time-Vested Restricted Stock Units Activity (Details) - Time-vested restricted stock units - $ / shares |
12 Months Ended | |||||
|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
||||
| Shares | ||||||
| Beginning balance, unvested shares (in shares) | 938,000 | |||||
| Granted (in shares) | [1] | 620,000 | ||||
| Vested (in shares) | (440,000) | |||||
| Forfeited (in shares) | (100,000) | |||||
| Ending balance, unvested shares (in shares) | 1,018,000 | 938,000 | ||||
| Weighted Average Grant Date Fair Value | ||||||
| Beginning balance, weighted average grant date fair value (in dollars per share) | $ 306.55 | |||||
| Granted (in dollars per share) | 318.87 | [1] | $ 304.44 | $ 316.32 | ||
| Vested (in dollars per share) | 304.45 | |||||
| Forfeited (in dollars per share) | 314.46 | |||||
| Ending balance, weighted average grant date fair value (in dollars per share) | $ 314.46 | $ 306.55 | ||||
| Number of shares granted (in shares) | [1] | 620,000 | ||||
| Director | ||||||
| Shares | ||||||
| Granted (in shares) | [1] | 10,000 | ||||
| Weighted Average Grant Date Fair Value | ||||||
| Number of shares granted (in shares) | [1] | 10,000 | ||||
| ||||||
Share-Based Payments - Shares Issued Under Employee Stock Purchase Plan (Details) - USD ($) shares in Thousands, $ in Millions |
1 Months Ended | 12 Months Ended | ||
|---|---|---|---|---|
Jun. 30, 2015 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Employee Stock Ownership Plan (ESOP) Disclosures [Line Items] | ||||
| Shares issued under the 2015 ESPP (in shares) | 6,200 | |||
| 2015 ESPP | ||||
| Employee Stock Ownership Plan (ESOP) Disclosures [Line Items] | ||||
| Shares issued under the 2015 ESPP (in shares) | 212 | 204 | 170 | |
| Cash received under the 2015 ESPP | $ 48.6 | $ 40.4 | $ 40.5 | |
Income Taxes - Income Before Income Tax Provision and the Income Tax Expense (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Income before income taxes (benefit): | |||
| Domestic | $ 3,290.0 | $ 4,725.3 | $ 3,877.0 |
| Foreign | 1,757.5 | 2,400.6 | 2,022.6 |
| Income before income tax expense and equity in loss of investee, net of tax | 5,047.5 | 7,125.9 | 5,899.6 |
| Current: | |||
| Federal | 647.0 | 947.4 | 1,131.8 |
| State | 41.2 | 59.1 | 45.5 |
| Foreign | 155.1 | 84.4 | 140.0 |
| Total | 843.3 | 1,090.9 | 1,317.3 |
| Deferred: | |||
| Federal | (1,749.9) | 1,143.9 | (62.0) |
| State | (6.8) | (2.3) | (7.4) |
| Foreign | 1,905.7 | (1,074.5) | 177.7 |
| Total | 149.0 | 67.1 | 108.3 |
| Total income tax expense | $ 992.3 | $ 1,158.0 | $ 1,425.6 |
Income Taxes - Narrative (Details) - USD ($) $ in Millions |
12 Months Ended | |||
|---|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Dec. 31, 2017 |
|
| Tax Credit Carryforward [Line Items] | ||||
| GILTI | $ (1,143.7) | $ (1,381.6) | ||
| Income taxes paid | 62.0 | |||
| Deferred tax liabilities, temporary differences, purchase accounting, foreign subsidiaries | 1,500.0 | |||
| Total deferred charges and prepaid taxes | 142.2 | 243.8 | ||
| Valuation allowance | 1,753.9 | 1.1 | ||
| Other | 103.1 | 103.7 | ||
| Unrecognized tax benefit | 68.8 | 122.7 | $ 109.1 | |
| Net interest expense | 1.0 | 4.7 | 2.2 | |
| Unrecognized tax benefits, interest on income taxes accrued | 21.2 | 20.0 | ||
| Decrease in unrecognized tax benefits is reasonably possible | 25.0 | |||
| 2017 Tax Act | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Reduction in deferred tax liability, undistributed foreign earnings | 34.6 | |||
| Income tax expense (benefit) | 12.7 | |||
| GILTI | 135.8 | |||
| Other tax expense (benefit) | $ 11.0 | |||
| Transition toll tax liabilities | 697.0 | 697.0 | ||
| General Business | Domestic Country | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Operating loss carryforwards | 0.6 | |||
| Tax credit carryforward, amount | 1.3 | |||
| Operating loss carryforward, subject to expiration | 18,000.0 | |||
| General Business | Foreign Tax Authority | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Operating loss carryforward, subject to expiration | 16,800.0 | |||
| Research | State and Local Jurisdiction | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Tax credit carryforward, amount | 142.4 | |||
| Earnings in the form of cash and cash equivalents | 2017 Tax Act | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Transition toll tax repatriation tax rate | 15.50% | |||
| Other Foreign Earnings | 2017 Tax Act | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Transition toll tax repatriation tax rate | 8.00% | |||
| Minimum | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Residual U.S. tax liability on reversal of deferred tax liabilities on foreign subsidiaries purchase accounting | 300.0 | |||
| Maximum | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Residual U.S. tax liability on reversal of deferred tax liabilities on foreign subsidiaries purchase accounting | 400.0 | |||
| Denmark Manufacturing Operations | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Tax expense on disposal group | 68.9 | |||
| TECFIDERA | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Deferred tax assets, value, reduction in value | 1,700.0 | |||
| Deferred tax liabilities, value, reduction in value | 1,600.0 | |||
| Income tax expense, reduction in deferred tax assets and liabilities, net | $ 90.3 | |||
| Intellectual Property | ||||
| Tax Credit Carryforward [Line Items] | ||||
| Other | 754.1 | |||
| Deferred tax liabilities, other | $ 603.3 | |||
Income Taxes - Components of Deferred Tax Assets and Liabilities (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
|---|---|---|
| Deferred tax assets: | ||
| Tax credits | $ 113.4 | $ 106.6 |
| Inventory, other reserves and accruals | 165.9 | 162.0 |
| Intangibles, net | 1,546.0 | 3,380.0 |
| Net operating loss | 2,080.3 | 130.4 |
| Share-based compensation | 23.3 | 23.8 |
| Other | 103.1 | 103.7 |
| Valuation allowance | (1,753.9) | (1.1) |
| Total deferred tax assets | 2,278.1 | 3,905.4 |
| Deferred tax liabilities: | ||
| Purchased intangible assets | (396.2) | (350.3) |
| GILTI | (1,143.7) | (1,381.6) |
| Tax credits | (174.6) | (1,617.2) |
| Depreciation, amortization and other | (227.0) | (135.0) |
| Total deferred tax liabilities | $ (1,941.5) | $ (3,484.1) |
Income Taxes - Tax Rate (Details) |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Income Tax Disclosure [Abstract] | |||
| Statutory rate | 21.00% | 21.00% | 21.00% |
| State taxes | 0.70% | 0.80% | 0.60% |
| Taxes on foreign earnings | (3.30%) | (4.50%) | (1.90%) |
| Credits and net operating loss utilization | (1.20%) | (1.10%) | (0.90%) |
| Purchased intangible assets | 0.70% | 0.40% | 1.20% |
| Divestiture of Denmark manufacturing operations | (0.40%) | 1.00% | 0.00% |
| Internal reorganization of certain intellectual property rights | 0.00% | (2.10%) | 0.00% |
| TECFIDERA impairment | 1.80% | 0.00% | 0.00% |
| GILTI | 1.30% | 1.50% | 1.60% |
| U.S. tax reform | 0.00% | 0.00% | 2.10% |
| Swiss tax reform | 0.00% | (0.80%) | 0.00% |
| Other | (0.90%) | 0.10% | 0.50% |
| Effective tax rate | 19.70% | 16.30% | 24.20% |
Income Taxes - Accounting for Uncertainty in Income Taxes (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Reconciliation of Unrecognized Tax Benefits, Excluding Amounts Pertaining to Examined Tax Returns [Roll Forward] | |||
| Beginning balance at January 1, | $ 129.9 | $ 114.2 | $ 66.8 |
| Additions based on tax positions related to the current period | 1.5 | 5.3 | 0.5 |
| Additions for tax positions of prior periods | 51.7 | 17.2 | 58.7 |
| Reductions for tax positions of prior periods | (63.6) | (10.3) | (13.6) |
| Statute expirations | (7.9) | (0.1) | (2.9) |
| Settlement refund (payment) | 3.6 | 4.7 | |
| Settlement refund (payment) | (35.9) | ||
| Ending balance ar December 31, | $ 75.7 | $ 129.9 | $ 114.2 |
Other Consolidated Financial Statement Detail - Supplemental Cash Flow Information (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Organization, Consolidation and Presentation of Financial Statements [Abstract] | |||
| Interest | $ 272.7 | $ 244.2 | $ 243.2 |
| Income taxes | $ 906.7 | $ 1,064.5 | $ 1,007.1 |
Other Consolidated Financial Statement Detail - Narrative (Details) - USD ($) |
3 Months Ended | 12 Months Ended | ||
|---|---|---|---|---|
Dec. 31, 2018 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Business Acquisition [Line Items] | ||||
| Increase to goodwill | $ 0 | $ 117,500,000 | ||
| Unrealized gain (loss) on investments | (681,800,000) | 150,100,000 | ||
| Realized investment gains (losses) | 12,100,000 | 50,000,000.0 | ||
| Unrealized gains (losses) recognized during the period on equity securities | 681,800,000 | 150,100,000 | $ 128,500,000 | |
| Other long-term liabilities | 1,329,600,000 | 1,348,900,000 | ||
| Accrued income taxes | 709,900,000 | 803,300,000 | ||
| Denali and Sangamo | ||||
| Business Acquisition [Line Items] | ||||
| Unrealized gains (losses) recognized during the period on equity securities | 703,900,000 | |||
| Solothurn | ||||
| Business Acquisition [Line Items] | ||||
| Construction in progress | 12,400,000 | $ 50,000,000.0 | ||
| Fumarate | Fumapharm AG | Twenty billion | ||||
| Business Acquisition [Line Items] | ||||
| Increase to goodwill | $ 300,000,000.0 | |||
| Cumulative sales level | 20,000,000,000.0 | 20,000,000,000.0 | ||
| Fumarate | Fumapharm AG | Each additional one billion up to twenty billion | ||||
| Business Acquisition [Line Items] | ||||
| Cumulative sales level | $ 20,000,000,000.0 | $ 20,000,000,000.0 | $ 20,000,000,000.0 | |
Other Consolidated Financial Statement Detail - Other Income (Expense), Net (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Organization, Consolidation and Presentation of Financial Statements [Abstract] | |||
| Interest income | $ 42.0 | $ 120.0 | $ 112.5 |
| Interest expense | (222.5) | (187.4) | (200.6) |
| Gain (loss) on investments, net | 685.7 | 204.7 | 119.5 |
| Foreign exchange gains (losses), net | (10.7) | (7.0) | (9.9) |
| Other, net | 2.9 | (47.0) | (10.5) |
| Total other income (expense), net | $ 497.4 | $ 83.3 | $ 11.0 |
Other Consolidated Financial Statement Detail - Gain (Loss) on Investments (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Organization, Consolidation and Presentation of Financial Statements [Abstract] | |||
| Net gains (losses) recognized during the period on equity securities | $ 693.9 | $ 200.1 | $ 127.9 |
| Less: Net gains (losses) recognized on equity securities sold during the period and on capital distributions | (12.1) | (50.0) | 0.6 |
| Unrealized gains (losses) recognized during the period on equity securities | $ 681.8 | $ 150.1 | $ 128.5 |
Other Consolidated Financial Statement Detail - Accrued Expenses and Other (Details) - USD ($) $ in Millions |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Dec. 31, 2017 |
|---|---|---|---|---|
| SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Line Items] | ||||
| Revenue-related reserves for discounts and allowances | $ 1,276.0 | $ 1,198.9 | $ 1,051.3 | $ 761.6 |
| Total accrued expenses and other | 3,145.3 | 2,765.8 | ||
| Component of accrued expenses and other | ||||
| SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Line Items] | ||||
| Revenue-related reserves for discounts and allowances | 1,080.6 | 1,001.1 | ||
| Collaboration expenses | 389.9 | 281.6 | ||
| Employee-related Liabilities, Current | 333.8 | 309.1 | ||
| Accrued Royalties, Current | 218.5 | 220.9 | ||
| Derivative liabilities | 181.5 | 6.7 | ||
| Business Combination, Contingent Consideration, Liability, Current | 149.6 | 148.4 | ||
| Other Accrued Liabilities, Current | 791.4 | 798.0 | ||
| Total accrued expenses and other | $ 3,145.3 | $ 2,765.8 |
Collaborative and Other Relationships - OCREVUS (Details) - OCREVUS $ in Millions |
1 Months Ended |
|---|---|
|
Mar. 31, 2017
USD ($)
| |
| Collaborative arrangements and non-collaborative arrangement transactions | |
| Royalty operating profit threshold for highest royalty rate percentage | $ 900.0 |
| Reduction in royalty rate | 50.00% |
| Collaboration agreement term | 11 years |
| Foreign Tax Authority | |
| Collaborative arrangements and non-collaborative arrangement transactions | |
| Royalty percentage to be received | 3.00% |
| Minimum | |
| Collaborative arrangements and non-collaborative arrangement transactions | |
| Royalty percentage to be received | 13.50% |
| Maximum | |
| Collaborative arrangements and non-collaborative arrangement transactions | |
| Royalty percentage to be received | 24.00% |
Collaborative and Other Relationships - Co-promotion Profit Sharing Formula (Details) - RITUXAN |
Dec. 31, 2020 |
|---|---|
| Co-promotion profit sharing formula | |
| Until GAZYVA First Non-CLL FDA Approval | 40.00% |
| After First GA101 Threshold Date | |
| After GAZYVA First Non-CLL FDA Approval until First GAZYVA Threshold Date | 39.00% |
| After First GAZYVA Threshold Date until Second GAZYVA Threshold Date | 37.50% |
| After Second GAZYVA Threshold Date | 35.00% |
Collaborative and Other Relationships - Profit Sharing (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| RITUXAN | |||
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Percentage of co promotion operating profits first fifty million | 30.00% | ||
| Until First GAZYVA Threshold Date | 39.00% | ||
| After First GAZYVA Threshold Date until Second GAZYVA Threshold Date | 37.50% | ||
| GAZYVA | |||
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Percentage of co promotion operating profits first fifty million | 35.00% | ||
| Co-promotion operating profit threshold for Rituxan in U S and Canada to determine share of co promotion operating profit prior to amendment | $ 50.0 | ||
| Limit of gross sale of GAZYVA to be achieved in preceding 12 months under option one | 150.0 | ||
| Limit of gross sale of GAZYVA to be achieved in any 12 months under option one | 150.0 | ||
| Sales trigger gross sales threshold | $ 500.0 | ||
| Until First GAZYVA Threshold Date | 39.00% | ||
| After First GAZYVA Threshold Date until Second GAZYVA Threshold Date | 37.50% | 37.50% | 37.50% |
| New Anti Cd20 | |||
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Future percentage of co-promotion operating profits | 37.50% | 37.50% | 37.50% |
| New Anti Cd20 | Minimum | |||
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Future percentage of co-promotion operating profits | 30.00% | ||
| New Anti Cd20 | Maximum | |||
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Future percentage of co-promotion operating profits | 37.50% | ||
Collaborative and Other Relationships - Pretax Profit Sharing Formula (Details) - GAZYVA |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|---|---|---|---|
| Co-promotion profit sharing formula | |||
| Until First GAZYVA Threshold Date | 39.00% | ||
| After First GA101 Threshold Date | |||
| After First GAZYVA Threshold Date until Second GAZYVA Threshold Date | 37.50% | 37.50% | 37.50% |
| After Second GAZYVA Threshold Date | 35.00% |
Collaborative and Other Relationships - Revenues from Anti-CD20 Therapeutic Programs (Details) - USD ($) $ in Millions |
3 Months Ended | 12 Months Ended | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Sep. 30, 2020 |
Jun. 30, 2020 |
Mar. 31, 2020 |
Dec. 31, 2019 |
Sep. 30, 2019 |
Jun. 30, 2019 |
Mar. 31, 2019 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Total revenues from anti-CD20 therapeutic programs | $ 2,852.6 | $ 3,376.1 | $ 3,681.6 | $ 3,534.3 | $ 3,671.3 | $ 3,600.1 | $ 3,616.7 | $ 3,489.8 | $ 13,444.6 | $ 14,377.9 | $ 13,452.9 |
| Revenues from anti-CD20 therapeutic programs | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Total revenues from anti-CD20 therapeutic programs | $ 419.0 | $ 560.1 | $ 478.3 | $ 520.4 | $ 600.8 | $ 595.8 | $ 576.4 | $ 517.4 | 1,977.8 | 2,290.4 | 1,980.2 |
| Genentech | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Biogen's share of pre-tax profits in the U.S. for RITUXAN and GAZYVA, including the reimbursement of selling and development expenses | 1,080.2 | 1,542.4 | 1,431.9 | ||||||||
| Other revenues from anti-CD20 therapeutic programs | $ 897.6 | $ 748.0 | $ 548.3 | ||||||||
Collaborative and Other Relationships - Ionis (Details) - USD ($) shares in Millions, $ in Millions |
1 Months Ended | 12 Months Ended | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2019 |
Dec. 31, 2018 |
Jun. 30, 2018 |
Dec. 31, 2017 |
Sep. 30, 2013 |
Dec. 31, 2012 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Dec. 31, 2016 |
Dec. 31, 2015 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Cost of sales, excluding amortization and impairment of acquired intangible assets | $ 1,805.2 | $ 1,955.4 | $ 1,816.3 | ||||||||
| Research and development | 3,990.9 | 2,280.6 | 2,597.2 | ||||||||
| Ionis Pharmaceuticals | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| License fee | $ 70.0 | $ 70.0 | |||||||||
| Expected license fee and regulatory milestone payments | $ 130.0 | 130.0 | |||||||||
| Upfront payment for collaboration agreement | $ 375.0 | 30.0 | |||||||||
| Total aducanumab collaboration third party milestone expense | $ 155.0 | $ 10.0 | |||||||||
| Research and development | $ 324.1 | 11.3 | 30.0 | $ 10.0 | |||||||
| Research and development expense asset acquired | $ 45.0 | ||||||||||
| Term of collaboration agreement | 10 years | 6 years | |||||||||
| Total payment to enter collaboration agreement | $ 1,000.0 | ||||||||||
| Investment in common stock, shares purchased (in shares) | 11.5 | ||||||||||
| Purchase of common stock | $ 625.0 | ||||||||||
| Prepaid research and discovery services | 50.9 | ||||||||||
| Investment in common stock, value | 462.9 | ||||||||||
| Premium on purchase of common stock | 162.1 | ||||||||||
| Estimated additional payments upon achievement of development and commercial milestones | $ 260.0 | ||||||||||
| Upfront and milestone payments made to collaborative partner | 25.0 | ||||||||||
| Expected additional milestone payments when certain sales threshold is met | $ 800.0 | ||||||||||
| Ionis Pharmaceuticals | SOD1 | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Research and development | 28.0 | 20.0 | 18.0 | ||||||||
| Estimated additional payments upon achievement of development and commercial milestones | $ 55.0 | ||||||||||
| Upfront and milestone payments made to collaborative partner | $ 35.0 | ||||||||||
| Ionis Pharmaceuticals | SPINRAZA | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Cost of sales, excluding amortization and impairment of acquired intangible assets | $ 286.6 | $ 293.0 | $ 238.0 | ||||||||
| Ionis Pharmaceuticals | Minimum | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Estimated additional payments upon achievement of development and commercial milestones | 125.0 | ||||||||||
| Ionis Pharmaceuticals | Minimum | SPINRAZA | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Percentage of royalties as per collaboration | 11.00% | 11.00% | |||||||||
| Ionis Pharmaceuticals | Maximum | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Estimated additional payments upon achievement of development and commercial milestones | $ 270.0 | ||||||||||
| Ionis Pharmaceuticals | Maximum | SPINRAZA | |||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | |||||||||||
| Percentage of royalties as per collaboration | 15.00% | 15.00% | |||||||||
Collaborative and Other Relationships - Eisai (Details) $ in Millions |
3 Months Ended | 9 Months Ended | 12 Months Ended | 24 Months Ended | ||||
|---|---|---|---|---|---|---|---|---|
|
Jun. 30, 2020
USD ($)
|
Mar. 31, 2019
USD ($)
|
Mar. 31, 2018 |
Dec. 31, 2018 |
Dec. 31, 2020
USD ($)
product
|
Dec. 31, 2019
USD ($)
|
Dec. 31, 2018
USD ($)
|
Dec. 31, 2020 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||
| Percentage of development costs incurred | 100.00% | |||||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | $ 3.3 | $ (77.4) | $ 0.0 | |||||
| Eisai | ||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||
| Number of product candidates | product | 2 | |||||||
| Loss on contract termination | $ 45.0 | |||||||
| Percentage of future development costs related to Eisai | 15.00% | 45.00% | 45.00% | |||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | $ 33.8 | 0.0 | 0.0 | |||||
| Total aducanumab collaboration third party milestone expense | $ 75.0 | $ 75.0 | $ 0.0 | $ 0.0 | ||||
| Eisai | United States | ||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||
| Biogen share of co-promotion profits or losses | 55.00% | |||||||
| Eisai | European Union | ||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||
| Biogen share of co-promotion profits or losses | 68.50% | |||||||
| Eisai | JAPAN | ||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||
| Biogen share of co-promotion profits or losses | 20.00% | |||||||
Collaborative and Other Relationships - Summary of Activity Related to BAN2401 and Elenbecestat Collaboration (Details) - Eisai - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Total development expense incurred by the collaboration related to the advancement of BAN2401 and elenbecestat | $ 219.3 | $ 348.7 | $ 232.0 |
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | 109.6 | 174.3 | 116.0 |
| Selling, general and administrative | E2609 and BAN2401 | |||
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | 4.9 | 16.2 | 5.4 |
| Total sales and marketing expense incurred by the collaboration | $ 9.8 | $ 32.4 | $ 10.7 |
Collaborative and Other Relationships - Summary of Activity Related to Aducanumab Collaboration (Details) - USD ($) $ in Millions |
3 Months Ended | 12 Months Ended | ||
|---|---|---|---|---|
Jun. 30, 2020 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | $ 3.3 | $ (77.4) | $ 0.0 | |
| Eisai | ||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | 109.6 | 174.3 | 116.0 | |
| Total aducanumab collaboration third party milestone expense | $ 75.0 | 75.0 | 0.0 | 0.0 |
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | 33.8 | 0.0 | 0.0 | |
| Aducanumab | Research and development | Eisai | ||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Total UCB development expense | 152.0 | 179.4 | 264.8 | |
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | 83.6 | 98.7 | 234.6 | |
| Aducanumab | Selling, general and administrative | Eisai | ||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Total UCB development expense | 353.0 | 27.4 | 50.6 | |
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | $ 193.7 | $ 15.1 | $ 27.3 | |
Collaborative and Other Relationships - Summary of Activity Related to the UCB Collaboration (Details) - Research and development - UCB Pharma S.A. - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Total UCB development expense | $ 58.3 | $ 31.9 | $ 29.7 |
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | $ 29.2 | $ 16.0 | $ 14.9 |
Collaborative and Other Relationships - Alkermes (Details) - USD ($) $ in Millions |
1 Months Ended | 12 Months Ended | ||
|---|---|---|---|---|
Oct. 31, 2019 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Research and development | $ 3,990.9 | $ 2,280.6 | $ 2,597.2 | |
| Potential future milestone payments commitment to third party approximately | 10,200.0 | |||
| Alkermes | ||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Research and development | $ 32.4 | 53.5 | $ 68.7 | |
| Potential future milestone payments commitment to third party approximately | $ 155.0 | |||
| VUMERITY | Alkermes | ||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Percentage of royalties as per collaboration | 15.00% | 15.00% | ||
| Royalties payable period, post FDA approval | 5 years | |||
Collaborative and Other Relationships - Bristol Myers Squibb (Details) - USD ($) $ in Millions |
1 Months Ended | 12 Months Ended | ||
|---|---|---|---|---|
Jun. 30, 2017 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Research and development | $ 3,990.9 | $ 2,280.6 | $ 2,597.2 | |
| Bristol-Myers Squibb | ||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Research and development | $ 300.0 | 62.4 | $ 144.0 | $ 97.0 |
| Total aducanumab collaboration third party milestone expense | 360.0 | |||
| iPerian | ||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||
| Total aducanumab collaboration third party milestone expense | $ 370.0 | |||
Collaborative and Other Relationships - Acorda (Details) - Acorda - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | |||
| Expected additional milestone payments when certain sales threshold is met | $ 15.0 | ||
| Foreign sales required to trigger milestone | 100.0 | ||
| Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income | $ 44.5 | $ 42.0 | $ 36.5 |
Collaborative and Other Relationships - Other Arrangements (Details) - USD ($) $ / shares in Units, shares in Millions, $ in Millions |
1 Months Ended | 12 Months Ended | ||||
|---|---|---|---|---|---|---|
Nov. 30, 2020 |
Sep. 30, 2020 |
Feb. 29, 2020 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||
| Research and development | $ 3,990.9 | $ 2,280.6 | $ 2,597.2 | |||
| Sage Therapeutics | ||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||
| Global licensing collaboration, shares purchased, amount | $ 650.0 | |||||
| Global licensing collaboration, shares purchased (in shares) | 6.2 | |||||
| Global licensing collaboration, purchase price per share (in dollars per share) | $ 104.14 | |||||
| Research and development expense asset acquired | $ 209.0 | |||||
| Upfront and milestone payments made to collaborative partner | 875.0 | |||||
| Global licensing collaboration, development and commercial milestone payments | $ 1,600.0 | |||||
| Denali Therapeutics | ||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||
| Global licensing collaboration, shares purchased, amount | $ 465.0 | |||||
| Global licensing collaboration, shares purchased (in shares) | 13.0 | |||||
| Global licensing collaboration, purchase price per share (in dollars per share) | $ 34.94 | |||||
| Research and development expense asset acquired | $ 41.3 | |||||
| Upfront and milestone payments made to collaborative partner | 560.0 | |||||
| Global licensing collaboration, development and commercial milestone payments | $ 1,100.0 | |||||
| Research and development | 8.8 | |||||
| Sangamo Therapeutics, Inc. Agreement | ||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||
| Global licensing collaboration, shares purchased, amount | $ 225.0 | |||||
| Global licensing collaboration, shares purchased (in shares) | 24.0 | |||||
| Global licensing collaboration, purchase price per share (in dollars per share) | $ 9.21 | |||||
| Research and development expense asset acquired | $ 83.0 | |||||
| Upfront and milestone payments made to collaborative partner | 125.0 | |||||
| Global licensing collaboration, development and commercial milestone payments | 2,400.0 | |||||
| Research and development | $ 6.4 | |||||
| Term of collaboration agreement | 5 years | |||||
| Global licensing collaboration, payment, selection of targets | 80.0 | |||||
| Global licensing collaboration, payment,first milestone | 1,900.0 | |||||
| Global licensing collaboration, payment, sales based milestone | $ 380.0 | |||||
| Other research and discovery | ||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||
| Research and development | $ 92.1 | $ 77.0 | $ 48.6 | |||
Collaborative and Other Relationships - Samsung Bioepis (Details) $ in Millions, ₩ in Billions |
1 Months Ended | 3 Months Ended | 12 Months Ended | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Jan. 31, 2020
USD ($)
|
Nov. 30, 2018
USD ($)
|
Nov. 30, 2018
KRW (₩)
|
Dec. 31, 2013
USD ($)
|
Feb. 29, 2012
USD ($)
|
Dec. 31, 2020
USD ($)
|
Sep. 30, 2020
USD ($)
|
Jun. 30, 2020
USD ($)
|
Mar. 31, 2020
USD ($)
|
Dec. 31, 2019
USD ($)
|
Sep. 30, 2019
USD ($)
|
Jun. 30, 2019
USD ($)
|
Mar. 31, 2019
USD ($)
|
Dec. 31, 2020
USD ($)
product
|
Dec. 31, 2019
USD ($)
product
|
Dec. 31, 2018
USD ($)
|
Dec. 31, 2020
KRW (₩)
|
Dec. 31, 2019
KRW (₩)
|
Jun. 30, 2018 |
Feb. 29, 2012
KRW (₩)
|
|
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||||||||||||||
| Payments to increase investment in Samsung Bioepis | $ 0.0 | $ 0.0 | $ 676.6 | |||||||||||||||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | $ 3.3 | $ (77.4) | 0.0 | |||||||||||||||||
| Number of ophthalmolgy biosimilar products | product | 2 | 2 | ||||||||||||||||||
| Research and Development Expense (Excluding Acquired in Process Cost) | $ 3,990.9 | $ 2,280.6 | 2,597.2 | |||||||||||||||||
| Collaboration profit (loss) sharing | 232.9 | 241.6 | 185.0 | |||||||||||||||||
| Revenues | $ 2,852.6 | $ 3,376.1 | $ 3,681.6 | $ 3,534.3 | $ 3,671.3 | $ 3,600.1 | $ 3,616.7 | $ 3,489.8 | 13,444.6 | 14,377.9 | 13,452.9 | |||||||||
| Samsung Bioepis | ||||||||||||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||||||||||||||
| Upfront and milestone payments made to collaborative partner | $ 100.0 | |||||||||||||||||||
| Research and development expense asset acquired | 63.0 | |||||||||||||||||||
| Prepaid research and discovery services | $ 37.0 | |||||||||||||||||||
| Estimated additional payments upon achievement of development and commercial milestones | $ 195.0 | |||||||||||||||||||
| Term of collaboration agreement | 10 years | 5 years | ||||||||||||||||||
| Option exercise fee | $ 60.0 | |||||||||||||||||||
| Research and Development Expense (Excluding Acquired in Process Cost) | $ 46.0 | |||||||||||||||||||
| Expected profit share percentage | 50.00% | |||||||||||||||||||
| Collaboration profit (loss) sharing | $ 266.5 | 241.6 | 187.4 | |||||||||||||||||
| Revenues | 20.9 | 106.2 | $ 96.4 | |||||||||||||||||
| Due from related parties | 5.1 | 85.0 | 5.1 | 85.0 | ||||||||||||||||
| Due to related parties | 99.0 | 100.0 | 99.0 | 100.0 | ||||||||||||||||
| Samsung Bioepis | Development Milestones | ||||||||||||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||||||||||||||
| Total aducanumab collaboration third party milestone expense | $ 15.0 | |||||||||||||||||||
| Samsung Bioepis | ||||||||||||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||||||||||||||
| Contributions by third party | $ 250.0 | ₩ 280.5 | ||||||||||||||||||
| Joint venture ownership percentage | 85.00% | |||||||||||||||||||
| Investment in Samsung Bioepis | $ 45.0 | $ 620.2 | $ 580.2 | $ 620.2 | 580.2 | ₩ 673.8 | ₩ 670.8 | ₩ 49.5 | ||||||||||||
| Samsung Bioepis, ownership percentage | 15.00% | 49.90% | 49.90% | 49.90% | 15.00% | |||||||||||||||
| Samsung Bioepis, ownership percentage before additional purchase transaction | 5.00% | |||||||||||||||||||
| Payments to increase investment in Samsung Bioepis | $ 676.6 | ₩ 759.5 | ||||||||||||||||||
| Total basis difference | $ 675.0 | |||||||||||||||||||
| Net profits on investment | $ 5.3 | 79.4 | ||||||||||||||||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | 45.3 | 1.2 | ||||||||||||||||||
| Amortization | $ 40.0 | $ 78.2 | ||||||||||||||||||
| Total aducanumab collaboration third party milestone expense | 25.0 | |||||||||||||||||||
| Net change in acquired and in-licensed rights and patents | $ 75.0 | |||||||||||||||||||
| Samsung Bioepis | Inventories | ||||||||||||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||||||||||||||
| Equity method investment basis difference amortization period | 1 year 6 months | |||||||||||||||||||
| Samsung Bioepis | Completed technology | ||||||||||||||||||||
| Collaborative arrangements and non-collaborative arrangement transactions | ||||||||||||||||||||
| Equity method investment basis difference amortization period | 15 years | |||||||||||||||||||
Investments in Variable Interest Entities (Details) - USD ($) $ in Millions |
1 Months Ended | 3 Months Ended | 9 Months Ended | 12 Months Ended | 24 Months Ended | |||
|---|---|---|---|---|---|---|---|---|
May 31, 2018 |
Oct. 31, 2017 |
Jun. 30, 2020 |
Dec. 31, 2018 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Dec. 31, 2020 |
|
| Variable Interest Entity [Line Items] | ||||||||
| Payment to Neurimmune | $ 4.6 | $ 0.0 | $ 0.0 | |||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | 3.3 | (77.4) | 0.0 | |||||
| Potential future milestone payments commitment to third party approximately | 10,200.0 | $ 10,200.0 | ||||||
| Investment in biotechnology companies that are determined to be unconsolidated variable interest entities | 12.8 | 22.7 | 12.8 | |||||
| Twelve months | ||||||||
| Variable Interest Entity [Line Items] | ||||||||
| Potential future milestone payments commitment to third party approximately | $ 200.0 | 200.0 | ||||||
| Neurimmune | ||||||||
| Variable Interest Entity [Line Items] | ||||||||
| Term of collaboration agreement | 12 years | |||||||
| Research and development costs, percentage | 100.00% | |||||||
| Payment to Neurimmune | $ 50.0 | $ 150.0 | ||||||
| Reduction in royalty rate payable on commercial sales | 15.00% | |||||||
| Additional reduction in royalty rate payable on commercial sales | 5.00% | |||||||
| Regulatory Milestones | ||||||||
| Variable Interest Entity [Line Items] | ||||||||
| Potential future milestone payments commitment to third party approximately | $ 1,300.0 | $ 1,300.0 | ||||||
| Eisai | ||||||||
| Variable Interest Entity [Line Items] | ||||||||
| Total aducanumab collaboration third party milestone expense | $ 75.0 | 75.0 | 0.0 | 0.0 | ||||
| Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income | $ 33.8 | $ 0.0 | $ 0.0 | |||||
| Percentage of future development costs related to Eisai | 15.00% | 45.00% | 45.00% | |||||
| Neurimmune | Regulatory Milestones | ||||||||
| Variable Interest Entity [Line Items] | ||||||||
| Potential future milestone payments commitment to third party approximately | $ 100.0 | $ 100.0 | ||||||
| Aducanumab | Regulatory Milestones | European Union | ||||||||
| Variable Interest Entity [Line Items] | ||||||||
| Potential future milestone payments commitment to third party approximately | 50.0 | 50.0 | ||||||
| Aducanumab | Regulatory Milestones | JAPAN | ||||||||
| Variable Interest Entity [Line Items] | ||||||||
| Potential future milestone payments commitment to third party approximately | $ 50.0 | $ 50.0 | ||||||
Litigation - Narrative (Details) $ in Millions |
2 Months Ended |
|---|---|
|
Dec. 31, 2019
USD ($)
| |
| Loss Contingency, Information about Litigation Matters [Abstract] | |
| Claims asserted | $ 200.0 |
Commitments and Contingencies - Narrative (Details) - USD ($) |
1 Months Ended | 3 Months Ended | 12 Months Ended | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2019 |
Oct. 31, 2019 |
Dec. 31, 2012 |
Mar. 31, 2019 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
Dec. 31, 2016 |
Dec. 31, 2021 |
Dec. 31, 2013 |
|
| Business Acquisition [Line Items] | ||||||||||
| Maximum contingent consideration in the form of development and approval milestones | $ 400,000,000.0 | |||||||||
| Contingent payment | $ 300,000,000.0 | 0 | $ 0 | $ 58,200,000 | ||||||
| Potential future milestone payments commitment, approximately | 10,200,000,000 | |||||||||
| Accrued expenses | 21,700,000 | |||||||||
| Cancellable future commitments | 593,000,000.0 | |||||||||
| Liabilities associated with uncertain tax positions | 79,600,000 | |||||||||
| Income taxes paid | 62,000,000.0 | |||||||||
| Solothurn | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Contractual obligation | 9,300,000 | |||||||||
| 2017 Tax Act | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Transition toll tax liabilities | $ 697,000,000.0 | 697,000,000.0 | 697,000,000.0 | |||||||
| Twelve months | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | 200,000,000.0 | |||||||||
| Development Milestones | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | 1,900,000,000 | |||||||||
| Regulatory Milestones | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | 1,300,000,000 | |||||||||
| Commercial Milestones | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | $ 7,000,000,000.0 | |||||||||
| Ionis Pharmaceuticals | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Total aducanumab collaboration third party milestone expense | 155,000,000.0 | $ 10,000,000.0 | ||||||||
| Ionis Pharmaceuticals | Minimum | SPINRAZA | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Percentage of royalties as per collaboration | 11.00% | 11.00% | ||||||||
| Ionis Pharmaceuticals | Maximum | SPINRAZA | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Percentage of royalties as per collaboration | 15.00% | 15.00% | ||||||||
| Alkermes | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | $ 155,000,000.0 | $ 155,000,000.0 | ||||||||
| Alkermes | VUMERITY | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Percentage of royalties as per collaboration | 15.00% | 15.00% | ||||||||
| Royalties payable period, post FDA approval | 5 years | |||||||||
| Aducanumab | Forecast | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | $ 86,200,000 | |||||||||
| Aducanumab | Regulatory Milestones | European Union | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | $ 50,000,000.0 | |||||||||
| Aducanumab | Regulatory Milestones | JAPAN | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | 50,000,000.0 | |||||||||
| Neurimmune | Regulatory Milestones | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Potential future milestone payments commitment, approximately | 100,000,000.0 | |||||||||
| TYSABRI | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Future contingent payment for annual worldwide net sales up to $2.0 billion | 18.00% | |||||||||
| Future contingent payment threshold | $ 2,000,000,000.0 | |||||||||
| Future contingent payment for annual worldwide net sales that exceed $2.0 billion | 25.00% | |||||||||
| Fumapharm AG | Fumarate | Twenty billion | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Cumulative sales level | 20,000,000,000.0 | |||||||||
| Fumapharm AG | Fumarate | Each additional one billion up to twenty billion | ||||||||||
| Business Acquisition [Line Items] | ||||||||||
| Cumulative sales level | $ 20,000,000,000.0 | $ 20,000,000,000.0 | ||||||||
Employee Benefit Plans - Narrative (Details) - USD ($) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| SWITZERLAND | |||
| Defined Contribution Plan Disclosure [Line Items] | |||
| Percentage of minimum investment return | 1.00% | 1.00% | 1.00% |
| Unfunded net pension | $ 75.7 | $ 42.9 | |
| Employee benefit plan obligations | 170.0 | 127.1 | |
| Pension cost (reversal) | 15.5 | 14.7 | $ 14.8 |
| SWITZERLAND | Other (income) expense, net | |||
| Defined Contribution Plan Disclosure [Line Items] | |||
| Pension cost (reversal) | 2.6 | 1.2 | 1.3 |
| GERMANY | |||
| Defined Contribution Plan Disclosure [Line Items] | |||
| Employee benefit plan obligations | 75.5 | 59.6 | |
| Periodic pension cost | 6.2 | 5.1 | 5.3 |
| GERMANY | Other (income) expense, net | |||
| Defined Contribution Plan Disclosure [Line Items] | |||
| Periodic pension cost | $ 2.0 | 1.4 | 1.5 |
| Domestic Plan | |||
| Defined Contribution Plan Disclosure [Line Items] | |||
| Minimum qualifying age for employee benefit plan | 21 years | ||
| Expenses related to savings plan | $ 44.3 | 44.8 | $ 42.2 |
| Deferred Compensation Plan | |||
| Defined Contribution Plan Disclosure [Line Items] | |||
| Deferred compensation liability | $ 120.0 | $ 114.6 | |
Segment Information - Narrative (Details) $ in Millions |
12 Months Ended | ||
|---|---|---|---|
|
Dec. 31, 2020
USD ($)
segment
|
Dec. 31, 2019
USD ($)
|
Dec. 31, 2018
USD ($)
|
|
| Revenue from External Customer [Line Items] | |||
| Number of reportable segment | segment | 1 | ||
| Long-lived assets related to operations in Denmark | $ 3,844.8 | $ 3,674.3 | $ 3,601.2 |
| Solothurn | |||
| Revenue from External Customer [Line Items] | |||
| Long-lived assets related to operations in Denmark | $ 2,180.6 | $ 2,028.8 | 1,748.5 |
| DENMARK | |||
| Revenue from External Customer [Line Items] | |||
| Long-lived assets related to operations in Denmark | $ 646.5 |
Segment Information - Geographic Information (Details) - USD ($) $ in Millions |
3 Months Ended | 12 Months Ended | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Sep. 30, 2020 |
Jun. 30, 2020 |
Mar. 31, 2020 |
Dec. 31, 2019 |
Sep. 30, 2019 |
Jun. 30, 2019 |
Mar. 31, 2019 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | $ 2,852.6 | $ 3,376.1 | $ 3,681.6 | $ 3,534.3 | $ 3,671.3 | $ 3,600.1 | $ 3,616.7 | $ 3,489.8 | $ 13,444.6 | $ 14,377.9 | $ 13,452.9 |
| Long-lived assets | 3,844.8 | 3,674.3 | 3,844.8 | 3,674.3 | 3,601.2 | ||||||
| U.S. | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Long-lived assets | 1,496.3 | 1,493.2 | 1,496.3 | 1,493.2 | 1,152.7 | ||||||
| Europe | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Long-lived assets | 2,321.4 | 2,162.9 | 2,321.4 | 2,162.9 | 2,442.8 | ||||||
| Asia | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Long-lived assets | 16.2 | 6.2 | 16.2 | 6.2 | 3.9 | ||||||
| Other | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Long-lived assets | 10.9 | 12.0 | 10.9 | 12.0 | 1.8 | ||||||
| Product, net | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 2,301.6 | 2,690.3 | 2,795.7 | 2,904.6 | 2,924.8 | 2,894.7 | 2,880.3 | 2,680.0 | 10,692.2 | 11,379.8 | 10,886.8 |
| Product, net | U.S. | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 5,900.1 | 6,713.8 | 6,800.5 | ||||||||
| Product, net | Europe | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 3,656.4 | 3,794.5 | 3,370.3 | ||||||||
| Product, net | Asia | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 596.7 | 320.3 | 281.2 | ||||||||
| Product, net | Other | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 539.0 | 551.2 | 434.8 | ||||||||
| Revenues from anti-CD20 therapeutic programs | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 419.0 | 560.1 | 478.3 | 520.4 | 600.8 | 595.8 | 576.4 | 517.4 | 1,977.8 | 2,290.4 | 1,980.2 |
| Revenues from anti-CD20 therapeutic programs | U.S. | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 1,897.4 | 2,211.9 | 1,903.4 | ||||||||
| Revenues from anti-CD20 therapeutic programs | Europe | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 0.1 | 0.2 | 0.2 | ||||||||
| Revenues from anti-CD20 therapeutic programs | Asia | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 0.0 | 0.0 | 0.0 | ||||||||
| Revenues from anti-CD20 therapeutic programs | Other | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 80.3 | 78.3 | 76.6 | ||||||||
| Other | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | $ 132.0 | $ 125.7 | $ 407.6 | $ 109.3 | $ 145.7 | $ 109.6 | $ 160.0 | $ 292.4 | 774.6 | 707.7 | 585.9 |
| Other | U.S. | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 733.6 | 585.8 | 457.0 | ||||||||
| Other | Europe | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 8.1 | 9.7 | 32.7 | ||||||||
| Other | Asia | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | 32.9 | 112.2 | 96.2 | ||||||||
| Other | Other | |||||||||||
| Revenue, Major Customer [Line Items] | |||||||||||
| Revenues | $ 0.0 | $ 0.0 | $ 0.0 | ||||||||
Quarterly Financial Data (Unaudited) (Details) - USD ($) $ / shares in Units, shares in Millions, $ in Millions |
3 Months Ended | 12 Months Ended | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2020 |
Sep. 30, 2020 |
Jun. 30, 2020 |
Mar. 31, 2020 |
Dec. 31, 2019 |
Sep. 30, 2019 |
Jun. 30, 2019 |
Mar. 31, 2019 |
Dec. 31, 2020 |
Dec. 31, 2019 |
Dec. 31, 2018 |
|||
| Selected Quarterly Financial Information [Abstract] | |||||||||||||
| Revenues | $ 2,852.6 | $ 3,376.1 | $ 3,681.6 | $ 3,534.3 | $ 3,671.3 | $ 3,600.1 | $ 3,616.7 | $ 3,489.8 | $ 13,444.6 | $ 14,377.9 | $ 13,452.9 | ||
| Gross profit | [1] | 2,361.9 | 2,927.0 | 3,270.5 | 3,080.0 | 3,224.2 | 3,170.1 | 3,140.4 | 2,887.8 | 11,639.4 | 12,422.5 | ||
| Net income | 357.6 | 703.9 | 1,606.5 | 1,392.6 | 1,439.7 | 1,545.9 | 1,494.1 | 1,408.8 | 4,060.5 | 5,888.5 | 4,474.0 | ||
| Net income attributable to Biogen Inc. | $ 357.9 | $ 701.5 | $ 1,542.1 | $ 1,399.1 | $ 1,439.7 | $ 1,545.9 | $ 1,494.1 | $ 1,408.8 | $ 4,000.6 | $ 5,888.5 | $ 4,430.7 | ||
| Net income per share: | |||||||||||||
| Basic earnings per share attributable to Biogen Inc. (in dollars per share) | $ 2.33 | $ 4.47 | $ 9.60 | $ 8.10 | $ 8.10 | $ 8.40 | $ 7.85 | $ 7.17 | $ 24.86 | $ 31.47 | $ 21.63 | ||
| Diluted earnings per share attributable to Biogen Inc. (in dollars per share) | $ 2.32 | $ 4.46 | $ 9.59 | $ 8.08 | $ 8.08 | $ 8.39 | $ 7.85 | $ 7.15 | $ 24.80 | $ 31.42 | $ 21.58 | ||
| Weighted-average shares used in calculating: | |||||||||||||
| Basic earnings per share attributable to Biogen Inc. (in shares) | 153.7 | 156.9 | 160.6 | 172.8 | 177.8 | 184.0 | 190.3 | 196.6 | 160.9 | 187.1 | 204.9 | ||
| Diluted earnings per share attributable to Biogen Inc. (in shares) | 154.0 | 157.2 | 160.9 | 173.1 | 178.2 | 184.2 | 190.4 | 197.0 | 161.3 | 187.4 | 205.3 | ||
| Product, net | |||||||||||||
| Selected Quarterly Financial Information [Abstract] | |||||||||||||
| Revenues | $ 2,301.6 | $ 2,690.3 | $ 2,795.7 | $ 2,904.6 | $ 2,924.8 | $ 2,894.7 | $ 2,880.3 | $ 2,680.0 | $ 10,692.2 | $ 11,379.8 | $ 10,886.8 | ||
| Revenues from anti-CD20 therapeutic programs | |||||||||||||
| Selected Quarterly Financial Information [Abstract] | |||||||||||||
| Revenues | 419.0 | 560.1 | 478.3 | 520.4 | 600.8 | 595.8 | 576.4 | 517.4 | 1,977.8 | 2,290.4 | 1,980.2 | ||
| Other | |||||||||||||
| Selected Quarterly Financial Information [Abstract] | |||||||||||||
| Revenues | $ 132.0 | $ 125.7 | $ 407.6 | $ 109.3 | $ 145.7 | $ 109.6 | $ 160.0 | $ 292.4 | $ 774.6 | $ 707.7 | $ 585.9 | ||
| |||||||||||||
{
"instance": {
"biib-20201231.htm": {
"axisCustom": 10,
"axisStandard": 48,
"contextCount": 940,
"dts": {
"calculationLink": {
"local": [
"biib-20201231_cal.xml"
]
},
"definitionLink": {
"local": [
"biib-20201231_def.xml"
],
"remote": [
"http://xbrl.fasb.org/us-gaap/2020/elts/us-gaap-eedm-def-2020-01-31.xml",
"http://xbrl.fasb.org/srt/2020/elts/srt-eedm1-def-2020-01-31.xml"
]
},
"inline": {
"local": [
"biib-20201231.htm"
]
},
"labelLink": {
"local": [
"biib-20201231_lab.xml"
],
"remote": [
"http://xbrl.fasb.org/us-gaap/2020/elts/us-gaap-doc-2020-01-31.xml",
"https://xbrl.sec.gov/dei/2020/dei-doc-2020-01-31.xml"
]
},
"presentationLink": {
"local": [
"biib-20201231_pre.xml"
]
},
"referenceLink": {
"remote": [
"https://xbrl.sec.gov/dei/2020/dei-ref-2020-01-31.xml",
"http://xbrl.fasb.org/us-gaap/2020/elts/us-gaap-ref-2020-01-31.xml"
]
},
"schema": {
"local": [
"biib-20201231.xsd"
],
"remote": [
"http://xbrl.fasb.org/srt/2020/elts/srt-2020-01-31.xsd",
"http://www.xbrl.org/2003/xbrl-instance-2003-12-31.xsd",
"http://www.xbrl.org/2003/xbrl-linkbase-2003-12-31.xsd",
"http://www.xbrl.org/2003/xl-2003-12-31.xsd",
"http://www.xbrl.org/2003/xlink-2003-12-31.xsd",
"http://www.xbrl.org/dtr/type/numeric-2009-12-16.xsd",
"http://www.xbrl.org/dtr/type/nonNumeric-2009-12-16.xsd",
"http://www.xbrl.org/2005/xbrldt-2005.xsd",
"http://www.xbrl.org/2006/ref-2006-02-27.xsd",
"http://xbrl.fasb.org/srt/2020/elts/srt-types-2020-01-31.xsd",
"http://xbrl.fasb.org/us-gaap/2020/elts/us-gaap-2020-01-31.xsd",
"http://xbrl.fasb.org/us-gaap/2020/elts/us-roles-2020-01-31.xsd",
"http://xbrl.fasb.org/srt/2020/elts/srt-roles-2020-01-31.xsd",
"https://xbrl.sec.gov/country/2020/country-2020-01-31.xsd",
"http://xbrl.fasb.org/us-gaap/2020/elts/us-types-2020-01-31.xsd",
"https://xbrl.sec.gov/currency/2020/currency-2020-01-31.xsd",
"https://xbrl.sec.gov/dei/2020/dei-2020-01-31.xsd",
"http://www.xbrl.org/lrr/role/negated-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/net-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/deprecated-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/reference-2009-12-16.xsd",
"http://xbrl.fasb.org/us-gaap/2020/elts/us-parts-codification-2020-01-31.xsd"
]
}
},
"elementCount": 1124,
"entityCount": 1,
"hidden": {
"http://fasb.org/us-gaap/2020-01-31": 48,
"http://www.biogenidec.com/20201231": 6,
"http://xbrl.sec.gov/dei/2020-01-31": 4,
"total": 58
},
"keyCustom": 152,
"keyStandard": 570,
"memberCustom": 117,
"memberStandard": 95,
"nsprefix": "biib",
"nsuri": "http://www.biogenidec.com/20201231",
"report": {
"R1": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:DocumentType",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "document",
"isDefault": "true",
"longName": "000010001 - Document - Cover Page",
"role": "http://www.biogenidec.com/role/CoverPage",
"shortName": "Cover Page",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:DocumentType",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R10": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisposalGroupsIncludingDiscontinuedOperationsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210081003 - Disclosure - Divestitures",
"role": "http://www.biogenidec.com/role/Divestitures",
"shortName": "Divestitures",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisposalGroupsIncludingDiscontinuedOperationsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R100": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:WeightedAverageNumberOfSharesCommonStockSubjectToRepurchaseOrCancellation",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240764049 - Disclosure - Earnings per Share - Narrative (Details)",
"role": "http://www.biogenidec.com/role/EarningsperShareNarrativeDetails",
"shortName": "Earnings per Share - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:WeightedAverageNumberOfSharesCommonStockSubjectToRepurchaseOrCancellation",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R101": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:AllocatedShareBasedCompensationExpense",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240794050 - Disclosure - Share-Based Payments - Share-based Compensation Expense Included in Consolidation Statements of Income (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails",
"shortName": "Share-Based Payments - Share-based Compensation Expense Included in Consolidation Statements of Income (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:EmployeeServiceShareBasedCompensationTaxBenefitFromCompensationExpense",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R102": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:AllocatedShareBasedCompensationExpense",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240804051 - Disclosure - Share-Based Payments - Summary of Share-based Compensation Expense Associated with Each Share-based Compensating Programs (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails",
"shortName": "Share-Based Payments - Summary of Share-based Compensation Expense Associated with Each Share-based Compensating Programs (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfCompensationCostForShareBasedPaymentArrangementsAllocationOfShareBasedCompensationCostsByPlanTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icf69f103a98a40ce90631e9f60d5754b_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:AllocatedShareBasedCompensationExpense",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R103": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognized",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240814052 - Disclosure - Share-Based Payments - Narrative (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"shortName": "Share-Based Payments - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognized",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R104": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia7c1ad961cd346de9af30e39027d96bf_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240824053 - Disclosure - Share-Based Payments - Stock Option Activity (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails",
"shortName": "Share-Based Payments - Stock Option Activity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia7c1ad961cd346de9af30e39027d96bf_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R105": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:CashProceedsReceivedAndTaxBenefitFromShareBasedPaymentAwardsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:EmployeeServiceShareBasedCompensationTaxBenefitFromExerciseOfStockOptions",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240834054 - Disclosure - Share-Based Payments - Tax Benefit and Cash Received from Stock Option Exercises (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsTaxBenefitandCashReceivedfromStockOptionExercisesDetails",
"shortName": "Share-Based Payments - Tax Benefit and Cash Received from Stock Option Exercises (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:CashProceedsReceivedAndTaxBenefitFromShareBasedPaymentAwardsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:EmployeeServiceShareBasedCompensationTaxBenefitFromExerciseOfStockOptions",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R106": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:ScheduleOfMarketStockUnitsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i28a8afaf854c4785ba7b167d54f6f8a0_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240844055 - Disclosure - Share-Based Payments - Market Stock Units Activity (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"shortName": "Share-Based Payments - Market Stock Units Activity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:ScheduleOfMarketStockUnitsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i57041d1a814045c7be6ea6d6fd527f45_D20200101-20201231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriod",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R107": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:AssumptionsUsedInValuationOfMarketBasedStockUnitsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i57041d1a814045c7be6ea6d6fd527f45_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240854056 - Disclosure - Share-Based Payments - Assumptions Used in Valuation of Market Based Stock Units (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"shortName": "Share-Based Payments - Assumptions Used in Valuation of Market Based Stock Units (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:AssumptionsUsedInValuationOfMarketBasedStockUnitsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i57041d1a814045c7be6ea6d6fd527f45_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R108": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:ScheduleOfCashSettledPerformanceSharesActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i2b626c65959246e09aa12e28314bba0c_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240864057 - Disclosure - Share-Based Payments - Cash Settled Performance Shares Activity (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"shortName": "Share-Based Payments - Cash Settled Performance Shares Activity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:ScheduleOfCashSettledPerformanceSharesActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i2b626c65959246e09aa12e28314bba0c_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R109": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:PerformanceunitsactivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i94d4a650ffa44c1998500b69da6939ac_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240874058 - Disclosure - Share-Based Payments - Performance Units Activity (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"shortName": "Share-Based Payments - Performance Units Activity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:PerformanceunitsactivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i94d4a650ffa44c1998500b69da6939ac_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R11": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:RevenueFromContractWithCustomerTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210101004 - Disclosure - Revenues",
"role": "http://www.biogenidec.com/role/Revenues",
"shortName": "Revenues",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:RevenueFromContractWithCustomerTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R110": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:PerformanceStockUnitsSettledinStockTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i78b2e6ab84a14827b284ec4a34e34ebd_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240884059 - Disclosure - Share-Based Payments - Performance Stock Units Settled in Stock Activity (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"shortName": "Share-Based Payments - Performance Stock Units Settled in Stock Activity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:PerformanceStockUnitsSettledinStockTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i78b2e6ab84a14827b284ec4a34e34ebd_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R111": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:PerformanceStockUnitsSettledinCashTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ib2b88b6b11d44635858d6a6d1dd65fd2_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240894060 - Disclosure - Share-Based Payments - Performance Stock Units Settled in Cash Activity (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"shortName": "Share-Based Payments - Performance Stock Units Settled in Cash Activity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:PerformanceStockUnitsSettledinCashTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ib2b88b6b11d44635858d6a6d1dd65fd2_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R112": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:TimeVestedRestrictedStockUnitsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i3db70ce0acff40ddb1a8049995563df4_I20191231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240904061 - Disclosure - Share-Based Payments - Time-Vested Restricted Stock Units Activity (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails",
"shortName": "Share-Based Payments - Time-Vested Restricted Stock Units Activity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:TimeVestedRestrictedStockUnitsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i12aa11ebb73d46f6a5e8a7655a9a3d7e_D20200101-20201231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedInPeriod",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R113": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "if188b16b7c0a45a1a919112de323694b_D20150601-20150630",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:StockIssuedDuringPeriodSharesEmployeeStockPurchasePlans",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240914062 - Disclosure - Share-Based Payments - Shares Issued Under Employee Stock Purchase Plan (Details)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails",
"shortName": "Share-Based Payments - Shares Issued Under Employee Stock Purchase Plan (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"ix:continuation",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icf6d89d5466147cd8f697b7487932bfb_D20200101-20201231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:StockIssuedDuringPeriodSharesEmployeeStockPurchasePlans",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R114": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfComponentsOfIncomeTaxExpenseBenefitTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeLossFromContinuingOperationsBeforeIncomeTaxesDomestic",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240944063 - Disclosure - Income Taxes - Income Before Income Tax Provision and the Income Tax Expense (Details)",
"role": "http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails",
"shortName": "Income Taxes - Income Before Income Tax Provision and the Income Tax Expense (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfComponentsOfIncomeTaxExpenseBenefitTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeLossFromContinuingOperationsBeforeIncomeTaxesDomestic",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R115": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:DeferredtaxliabilitiesGILTItaxcalculation",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240954064 - Disclosure - Income Taxes - Narrative (Details)",
"role": "http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"shortName": "Income Taxes - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-8",
"lang": "en-US",
"name": "biib:Deferredtaxliabilitiestemporarydifferencespurchaseaccountingforeignsubsidiaries",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R116": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:DeferredTaxAssetsTaxCreditCarryforwards",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240964065 - Disclosure - Income Taxes - Components of Deferred Tax Assets and Liabilities (Details)",
"role": "http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails",
"shortName": "Income Taxes - Components of Deferred Tax Assets and Liabilities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:DeferredTaxAssetsTaxCreditCarryforwards",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R117": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240974066 - Disclosure - Income Taxes - Tax Rate (Details)",
"role": "http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails",
"shortName": "Income Taxes - Tax Rate (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R118": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfUnrecognizedTaxBenefitsRollForwardTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i4fdb048733b648df99bd7b1e5b1476ef_I20191231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:UnrecognizedTaxBenefits",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240984067 - Disclosure - Income Taxes - Accounting for Uncertainty in Income Taxes (Details)",
"role": "http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails",
"shortName": "Income Taxes - Accounting for Uncertainty in Income Taxes (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfUnrecognizedTaxBenefitsRollForwardTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ic9925b2c3e594ae0812001969da2a99b_I20171231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:UnrecognizedTaxBenefits",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R119": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfCashFlowSupplementalDisclosuresTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InterestPaidNet",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241014068 - Disclosure - Other Consolidated Financial Statement Detail - Supplemental Cash Flow Information (Details)",
"role": "http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailSupplementalCashFlowInformationDetails",
"shortName": "Other Consolidated Financial Statement Detail - Supplemental Cash Flow Information (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfCashFlowSupplementalDisclosuresTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InterestPaidNet",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R12": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:InventoryDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210181005 - Disclosure - Inventory",
"role": "http://www.biogenidec.com/role/Inventory",
"shortName": "Inventory",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:InventoryDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R120": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfGoodwillTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:GoodwillAcquiredDuringPeriod",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241024069 - Disclosure - Other Consolidated Financial Statement Detail - Narrative (Details)",
"role": "http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"shortName": "Other Consolidated Financial Statement Detail - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:UnrealizedGainLossOnInvestments",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R121": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfOtherNonoperatingIncomeExpenseTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InvestmentIncomeInterest",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241034070 - Disclosure - Other Consolidated Financial Statement Detail - Other Income (Expense), Net (Details)",
"role": "http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails",
"shortName": "Other Consolidated Financial Statement Detail - Other Income (Expense), Net (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfOtherNonoperatingIncomeExpenseTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InvestmentIncomeInterest",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R122": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:GainLossOnInvestmentsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:EquitySecuritiesFvNiGainLoss",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241044071 - Disclosure - Other Consolidated Financial Statement Detail - Gain (Loss) on Investments (Details)",
"role": "http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailGainLossonInvestmentsDetails",
"shortName": "Other Consolidated Financial Statement Detail - Gain (Loss) on Investments (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:GainLossOnInvestmentsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:EquitySecuritiesFvNiGainLoss",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R123": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:AnalysisOfAmountOfAndChangeInProductRevenueReservesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ValuationAllowancesAndReservesBalance",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241054072 - Disclosure - Other Consolidated Financial Statement Detail - Accrued Expenses and Other (Details)",
"role": "http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails",
"shortName": "Other Consolidated Financial Statement Detail - Accrued Expenses and Other (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfAccruedLiabilitiesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i45223ed4352840169e55fb7523bb7c45_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "biib:Collaborationexpensesaccrual",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R124": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ie7879fe01aa94f84adbd555a7a199d32_D20170301-20170331",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:Royaltyoperatingprofitthresholdforhighestroyaltyratepercentage",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241084073 - Disclosure - Collaborative and Other Relationships - OCREVUS (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"shortName": "Collaborative and Other Relationships - OCREVUS (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ie7879fe01aa94f84adbd555a7a199d32_D20170301-20170331",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:Royaltyoperatingprofitthresholdforhighestroyaltyratepercentage",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R125": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "id2fc03f67f5645f1b013252212df9935_I20201231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionOne",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241094074 - Disclosure - Collaborative and Other Relationships - Co-promotion Profit Sharing Formula (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"shortName": "Collaborative and Other Relationships - Co-promotion Profit Sharing Formula (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "id2fc03f67f5645f1b013252212df9935_I20201231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionOne",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R126": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "id2fc03f67f5645f1b013252212df9935_I20201231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfCoPromotionOperatingProfitsFirstFiftyMillion",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241104075 - Disclosure - Collaborative and Other Relationships - Profit Sharing (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"shortName": "Collaborative and Other Relationships - Profit Sharing (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "id2fc03f67f5645f1b013252212df9935_I20201231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfCoPromotionOperatingProfitsFirstFiftyMillion",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R127": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i771406e8a3b34c93b5f87c9b46df7655_I20201231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionOne",
"reportCount": 1,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241114076 - Disclosure - Collaborative and Other Relationships - Pretax Profit Sharing Formula (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"shortName": "Collaborative and Other Relationships - Pretax Profit Sharing Formula (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i771406e8a3b34c93b5f87c9b46df7655_I20201231",
"decimals": "3",
"lang": "en-US",
"name": "biib:PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionThree",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R128": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241124077 - Disclosure - Collaborative and Other Relationships - Revenues from Anti-CD20 Therapeutic Programs (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"shortName": "Collaborative and Other Relationships - Revenues from Anti-CD20 Therapeutic Programs (Details)",
"subGroupType": "details",
"uniqueAnchor": null
},
"R129": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:CostOfGoodsAndServicesSold",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241134078 - Disclosure - Collaborative and Other Relationships - Ionis (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"shortName": "Collaborative and Other Relationships - Ionis (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "if223f76ca1884559bc1cd766f260b0eb_D20130901-20130930",
"decimals": "-5",
"lang": "en-US",
"name": "biib:LicenseFee",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R13": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:GoodwillAndIntangibleAssetsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210221006 - Disclosure - Intangible Assets and Goodwill",
"role": "http://www.biogenidec.com/role/IntangibleAssetsandGoodwill",
"shortName": "Intangible Assets and Goodwill",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:GoodwillAndIntangibleAssetsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R130": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i12a519048d3946d6b1cae8db3bf6ea2b_D20180101-20180331",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfDevelopmentCostsIncurred",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241144079 - Disclosure - Collaborative and Other Relationships - Eisai (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"shortName": "Collaborative and Other Relationships - Eisai (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i12a519048d3946d6b1cae8db3bf6ea2b_D20180101-20180331",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfDevelopmentCostsIncurred",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R131": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:SummaryOfActivityRelatedToCollaborationWithCompanyEightTableTextBlockTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i27837e9cd10e410d9324a0de6843615f_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:ExpenseIncurredByCollaboration",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241154080 - Disclosure - Collaborative and Other Relationships - Summary of Activity Related to BAN2401 and Elenbecestat Collaboration (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"shortName": "Collaborative and Other Relationships - Summary of Activity Related to BAN2401 and Elenbecestat Collaboration (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:SummaryOfActivityRelatedToCollaborationWithCompanyEightTableTextBlockTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i27837e9cd10e410d9324a0de6843615f_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:ExpenseIncurredByCollaboration",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R132": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeLossFromEquityMethodInvestments",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241164081 - Disclosure - Collaborative and Other Relationships - Summary of Activity Related to Aducanumab Collaboration (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"shortName": "Collaborative and Other Relationships - Summary of Activity Related to Aducanumab Collaboration (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:SummaryofActivityRelatedtoAducanumabCollaborationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i523d958a043b46abb2bf95ddabf9e48d_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "biib:Expenseincurredbythecollaboration",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R133": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i307bd3b52c544217a250d6bec5afed5a_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:Expenseincurredbythecollaboration",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241174082 - Disclosure - Collaborative and Other Relationships - Summary of Activity Related to the UCB Collaboration (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails",
"shortName": "Collaborative and Other Relationships - Summary of Activity Related to the UCB Collaboration (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i307bd3b52c544217a250d6bec5afed5a_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:Expenseincurredbythecollaboration",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R134": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241184083 - Disclosure - Collaborative and Other Relationships - Alkermes (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"shortName": "Collaborative and Other Relationships - Alkermes (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i062dfb826bb543c0ab344580c2bc9f39_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R135": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241194084 - Disclosure - Collaborative and Other Relationships - Bristol Myers Squibb (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails",
"shortName": "Collaborative and Other Relationships - Bristol Myers Squibb (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i18853d4d37eb4076a332a07be0132022_D20170601-20170630",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R136": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ie154ef82e19b42d287f8709039383fa3_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:ExpectedAdditionalMilestonePaymentsWhenMeetingCertainSalesThreshold",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241204085 - Disclosure - Collaborative and Other Relationships - Acorda (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails",
"shortName": "Collaborative and Other Relationships - Acorda (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ie154ef82e19b42d287f8709039383fa3_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:ExpectedAdditionalMilestonePaymentsWhenMeetingCertainSalesThreshold",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R137": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241214086 - Disclosure - Collaborative and Other Relationships - Other Arrangements (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"shortName": "Collaborative and Other Relationships - Other Arrangements (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i7ba45b40cf69417f9bccf9942bb04760_D20201101-20201130",
"decimals": "-5",
"lang": "en-US",
"name": "biib:GlobalLicensingCollaborationAgreementAmountSharesPurchased",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R138": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:PaymentsToAcquireEquityMethodInvestments",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241224087 - Disclosure - Collaborative and Other Relationships - Samsung Bioepis (Details)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"shortName": "Collaborative and Other Relationships - Samsung Bioepis (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i897172e0b7bd4edda63f02e0d50f23aa_D20200101-20200131",
"decimals": "-5",
"lang": "en-US",
"name": "biib:UpfrontAndMilestonePaymentsMadeToCollaborativePartner",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R139": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:PaymentsToMinorityShareholders",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241244088 - Disclosure - Investments in Variable Interest Entities (Details)",
"role": "http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails",
"shortName": "Investments in Variable Interest Entities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i4fdb048733b648df99bd7b1e5b1476ef_I20191231",
"decimals": "-5",
"lang": "en-US",
"name": "biib:InvestmentInBiotechnologyCompaniesThatAreDeterminedToBeUnconsolidatedVariableInterestEntities",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R14": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueDisclosuresTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210281007 - Disclosure - Fair Value Measurements",
"role": "http://www.biogenidec.com/role/FairValueMeasurements",
"shortName": "Fair Value Measurements",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueDisclosuresTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R140": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i933bc3e4859e442d998a3136c487cd85_D20191101-20191231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LossContingencyDamagesSoughtValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241264089 - Disclosure - Litigation - Narrative (Details)",
"role": "http://www.biogenidec.com/role/LitigationNarrativeDetails",
"shortName": "Litigation - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i933bc3e4859e442d998a3136c487cd85_D20191101-20191231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LossContingencyDamagesSoughtValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R141": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationContingentConsiderationArrangementsRangeOfOutcomesValueHigh",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241284090 - Disclosure - Commitments and Contingencies - Narrative (Details)",
"role": "http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"shortName": "Commitments and Contingencies - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i2093a163235b48f4b3fa0b2166bb4321_D20190101-20190331",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:PaymentForContingentConsiderationLiabilityFinancingActivities",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R142": {
"firstAnchor": {
"ancestors": [
"biib:PercentageOfMinimumInvestmentReturn",
"biib:PercentageOfMinimumInvestmentReturn",
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i4e1ee455bd644649ba9f949ccefc1a95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfMinimumInvestmentReturn",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241314091 - Disclosure - Employee Benefit Plans - Narrative (Details)",
"role": "http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails",
"shortName": "Employee Benefit Plans - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"biib:PercentageOfMinimumInvestmentReturn",
"biib:PercentageOfMinimumInvestmentReturn",
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i4e1ee455bd644649ba9f949ccefc1a95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "biib:PercentageOfMinimumInvestmentReturn",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R143": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:NumberOfReportableSegments",
"reportCount": 1,
"unique": true,
"unitRef": "segment",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241344092 - Disclosure - Segment Information - Narrative (Details)",
"role": "http://www.biogenidec.com/role/SegmentInformationNarrativeDetails",
"shortName": "Segment Information - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:NumberOfReportableSegments",
"reportCount": 1,
"unique": true,
"unitRef": "segment",
"xsiNil": "false"
}
},
"R144": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241354093 - Disclosure - Segment Information - Geographic Information (Details)",
"role": "http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails",
"shortName": "Segment Information - Geographic Information (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfRevenueFromExternalCustomersAttributedToForeignCountriesByGeographicAreaTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "id49704cf795948148f16dbae680341c9_I20181231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:NoncurrentAssets",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R145": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "241384094 - Disclosure - Quarterly Financial Data (Unaudited) (Details)",
"role": "http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"shortName": "Quarterly Financial Data (Unaudited) (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:GrossProfit",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R15": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CashCashEquivalentsAndMarketableSecuritiesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210361008 - Disclosure - Financial Instruments",
"role": "http://www.biogenidec.com/role/FinancialInstruments",
"shortName": "Financial Instruments",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CashCashEquivalentsAndMarketableSecuritiesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R16": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DerivativeInstrumentsAndHedgingActivitiesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210431009 - Disclosure - Derivative Instruments",
"role": "http://www.biogenidec.com/role/DerivativeInstruments",
"shortName": "Derivative Instruments",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DerivativeInstrumentsAndHedgingActivitiesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R17": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210501010 - Disclosure - Property, Plant and Equipment",
"role": "http://www.biogenidec.com/role/PropertyPlantandEquipment",
"shortName": "Property, Plant and Equipment",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R18": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeasesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210541011 - Disclosure - Leases",
"role": "http://www.biogenidec.com/role/Leases",
"shortName": "Leases",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeasesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R19": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DebtDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210621012 - Disclosure - Indebtedness",
"role": "http://www.biogenidec.com/role/Indebtedness",
"shortName": "Indebtedness",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DebtDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R2": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "100010002 - Statement - Consolidated Statements of Income",
"role": "http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"shortName": "Consolidated Statements of Income",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:SellingGeneralAndAdministrativeExpense",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R20": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:StockholdersEquityNoteDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210671013 - Disclosure - Equity",
"role": "http://www.biogenidec.com/role/Equity",
"shortName": "Equity",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:StockholdersEquityNoteDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R21": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:EarningsPerShareTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210731014 - Disclosure - Earnings per Share",
"role": "http://www.biogenidec.com/role/EarningsperShare",
"shortName": "Earnings per Share",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:EarningsPerShareTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R22": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210771015 - Disclosure - Share-Based Payments",
"role": "http://www.biogenidec.com/role/ShareBasedPayments",
"shortName": "Share-Based Payments",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R23": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210921016 - Disclosure - Income Taxes",
"role": "http://www.biogenidec.com/role/IncomeTaxes",
"shortName": "Income Taxes",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R24": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:AdditionalFinancialInformationDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210991017 - Disclosure - Other Consolidated Financial Statement Detail",
"role": "http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetail",
"shortName": "Other Consolidated Financial Statement Detail",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:AdditionalFinancialInformationDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R25": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CollaborativeArrangementDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211061018 - Disclosure - Collaborative and Other Relationships",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationships",
"shortName": "Collaborative and Other Relationships",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CollaborativeArrangementDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R26": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:VariableInterestEntityDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211231019 - Disclosure - Investments in Variable Interest Entities",
"role": "http://www.biogenidec.com/role/InvestmentsinVariableInterestEntities",
"shortName": "Investments in Variable Interest Entities",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:VariableInterestEntityDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R27": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LegalMattersAndContingenciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211251020 - Disclosure - Litigation",
"role": "http://www.biogenidec.com/role/Litigation",
"shortName": "Litigation",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LegalMattersAndContingenciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R28": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CommitmentsAndContingenciesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211271021 - Disclosure - Commitments and Contingencies",
"role": "http://www.biogenidec.com/role/CommitmentsandContingencies",
"shortName": "Commitments and Contingencies",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CommitmentsAndContingenciesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R29": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:GuaranteesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211291022 - Disclosure - Guarantees",
"role": "http://www.biogenidec.com/role/Guarantees",
"shortName": "Guarantees",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:GuaranteesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R3": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLoss",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "100020003 - Statement - Consolidated Statements of Comprehensive Income",
"role": "http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome",
"shortName": "Consolidated Statements of Comprehensive Income",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:OtherComprehensiveIncomeLossAvailableForSaleSecuritiesAdjustmentNetOfTax",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R30": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PensionAndOtherPostretirementBenefitsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211301023 - Disclosure - Employee Benefit Plans",
"role": "http://www.biogenidec.com/role/EmployeeBenefitPlans",
"shortName": "Employee Benefit Plans",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PensionAndOtherPostretirementBenefitsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R31": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SegmentReportingDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211321024 - Disclosure - Segment Information",
"role": "http://www.biogenidec.com/role/SegmentInformation",
"shortName": "Segment Information",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SegmentReportingDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R32": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:QuarterlyFinancialInformationTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "211361025 - Disclosure - Quarterly Financial Data (Unaudited)",
"role": "http://www.biogenidec.com/role/QuarterlyFinancialDataUnaudited",
"shortName": "Quarterly Financial Data (Unaudited)",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:QuarterlyFinancialInformationTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R33": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:BusinessOverviewPolicyTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "220022001 - Disclosure - Summary of Significant Accounting Policies (Policies)",
"role": "http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies",
"shortName": "Summary of Significant Accounting Policies (Policies)",
"subGroupType": "policies",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:BusinessOverviewPolicyTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R34": {
"firstAnchor": {
"ancestors": [
"us-gaap:PropertyPlantAndEquipmentPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:ScheduleOfPropertyPlantAndEquipmentUsefulLivesTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230033001 - Disclosure - Summary of Significant Accounting Policies (Tables)",
"role": "http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesTables",
"shortName": "Summary of Significant Accounting Policies (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"us-gaap:PropertyPlantAndEquipmentPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:ScheduleOfPropertyPlantAndEquipmentUsefulLivesTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R35": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisaggregationOfRevenueTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230113002 - Disclosure - Revenues (Tables)",
"role": "http://www.biogenidec.com/role/RevenuesTables",
"shortName": "Revenues (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisaggregationOfRevenueTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R36": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfInventoryCurrentTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230193003 - Disclosure - Inventory (Tables)",
"role": "http://www.biogenidec.com/role/InventoryTables",
"shortName": "Inventory (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfInventoryCurrentTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R37": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:IntangibleAssetsExcludingGoodwillTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230233004 - Disclosure - Intangible Assets and Goodwill (Tables)",
"role": "http://www.biogenidec.com/role/IntangibleAssetsandGoodwillTables",
"shortName": "Intangible Assets and Goodwill (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:IntangibleAssetsExcludingGoodwillTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R38": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfFairValueAssetsAndLiabilitiesMeasuredOnRecurringBasisTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230293005 - Disclosure - Fair Value Measurements (Tables)",
"role": "http://www.biogenidec.com/role/FairValueMeasurementsTables",
"shortName": "Fair Value Measurements (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfFairValueAssetsAndLiabilitiesMeasuredOnRecurringBasisTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R39": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230373006 - Disclosure - Financial Instruments (Tables)",
"role": "http://www.biogenidec.com/role/FinancialInstrumentsTables",
"shortName": "Financial Instruments (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R4": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "100030004 - Statement - Consolidated Balance Sheets",
"role": "http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"shortName": "Consolidated Balance Sheets",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:AvailableForSaleSecuritiesDebtSecuritiesCurrent",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R40": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfNotionalAmountsOfOutstandingDerivativePositionsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230443007 - Disclosure - Derivative Instruments (Tables)",
"role": "http://www.biogenidec.com/role/DerivativeInstrumentsTables",
"shortName": "Derivative Instruments (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfNotionalAmountsOfOutstandingDerivativePositionsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R41": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230513008 - Disclosure - Property, Plant and Equipment (Tables)",
"role": "http://www.biogenidec.com/role/PropertyPlantandEquipmentTables",
"shortName": "Property, Plant and Equipment (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R42": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:LesseeLeaseTableTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230553009 - Disclosure - Leases (Tables)",
"role": "http://www.biogenidec.com/role/LeasesTables",
"shortName": "Leases (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:LesseeLeaseTableTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R43": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfDebtTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230633010 - Disclosure - Indebtedness (Tables)",
"role": "http://www.biogenidec.com/role/IndebtednessTables",
"shortName": "Indebtedness (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfDebtTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R44": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:SummaryOfCommonStockTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230683011 - Disclosure - Equity (Tables)",
"role": "http://www.biogenidec.com/role/EquityTables",
"shortName": "Equity (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:SummaryOfCommonStockTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R45": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230743012 - Disclosure - Earnings per Share (Tables)",
"role": "http://www.biogenidec.com/role/EarningsperShareTables",
"shortName": "Earnings per Share (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R46": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230783013 - Disclosure - Share-Based Payments (Tables)",
"role": "http://www.biogenidec.com/role/ShareBasedPaymentsTables",
"shortName": "Share-Based Payments (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R47": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfComponentsOfIncomeTaxExpenseBenefitTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "230933014 - Disclosure - Income Taxes (Tables)",
"role": "http://www.biogenidec.com/role/IncomeTaxesTables",
"shortName": "Income Taxes (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfComponentsOfIncomeTaxExpenseBenefitTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R48": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfCashFlowSupplementalDisclosuresTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "231003015 - Disclosure - Other Consolidated Financial Statement Detail (Tables)",
"role": "http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailTables",
"shortName": "Other Consolidated Financial Statement Detail (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfCashFlowSupplementalDisclosuresTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R49": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:CoPromotionProfitSharingFormulaTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "231073016 - Disclosure - Collaborative and Other Relationships (Tables)",
"role": "http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsTables",
"shortName": "Collaborative and Other Relationships (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:CoPromotionProfitSharingFormulaTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R5": {
"firstAnchor": null,
"groupType": "statement",
"isDefault": "false",
"longName": "100040005 - Statement - Consolidated Balance Sheets (Parenthetical)",
"role": "http://www.biogenidec.com/role/ConsolidatedBalanceSheetsParenthetical",
"shortName": "Consolidated Balance Sheets (Parenthetical)",
"subGroupType": "parenthetical",
"uniqueAnchor": null
},
"R50": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfRevenueFromExternalCustomersAttributedToForeignCountriesByGeographicAreaTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "231333017 - Disclosure - Segment Information (Tables)",
"role": "http://www.biogenidec.com/role/SegmentInformationTables",
"shortName": "Segment Information (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfRevenueFromExternalCustomersAttributedToForeignCountriesByGeographicAreaTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R51": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "231373018 - Disclosure - Quarterly Financial Data (Unaudited) (Tables)",
"role": "http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedTables",
"shortName": "Quarterly Financial Data (Unaudited) (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R52": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"biib:BusinessOverviewPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "biib:NumberOfOphthalmolgyBiosimilarProductsExclusiveRights",
"reportCount": 1,
"unitRef": "product",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240044001 - Disclosure - Summary of Significant Accounting Policies - Narrative (Details)",
"role": "http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"shortName": "Summary of Significant Accounting Policies - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"us-gaap:ConsolidationPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"lang": "en-US",
"name": "biib:InterestInSubsidiary",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R53": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"us-gaap:PropertyPlantAndEquipmentPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i1b4148f6f0e64fcc8d46dde21febd1df_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentUsefulLife",
"reportCount": 1,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240054002 - Disclosure - Summary of Significant Accounting Policies - Summary of Property, Plant and Equipment Estimated Useful Lives (Details)",
"role": "http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails",
"shortName": "Summary of Significant Accounting Policies - Summary of Property, Plant and Equipment Estimated Useful Lives (Details)",
"subGroupType": "details",
"uniqueAnchor": null
},
"R54": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ResearchAndDevelopmentInProcess",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240074003 - Disclosure - Acquisitions - Narrative (Details)",
"role": "http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"shortName": "Acquisitions - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i74d25f7562934bc8a1eebaf7750912d2_D20190601-20190630",
"decimals": "INF",
"lang": "en-US",
"name": "biib:ClinicalAssetsAcquired",
"reportCount": 1,
"unique": true,
"unitRef": "product",
"xsiNil": "false"
}
},
"R55": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:DisposalGroupNotDiscontinuedOperationLossGainOnWriteDown",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240094004 - Disclosure - Divestitures (Details)",
"role": "http://www.biogenidec.com/role/DivestituresDetails",
"shortName": "Divestitures (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ida412b4bccf941a7af64620b69dcf1ef_I20190831",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:DisposalGroupIncludingDiscontinuedOperationConsideration",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R56": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240124005 - Disclosure - Revenues - Revenues by Product (Details)",
"role": "http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"shortName": "Revenues - Revenues by Product (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:DisaggregationOfRevenueTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia7c7c39b59484f68a74bc30138f093c8_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R57": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "biib:NumberOfWholesalers",
"reportCount": 1,
"unique": true,
"unitRef": "wholesaler",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240134006 - Disclosure - Revenues - Narrative (Details)",
"role": "http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"shortName": "Revenues - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "biib:NumberOfWholesalers",
"reportCount": 1,
"unique": true,
"unitRef": "wholesaler",
"xsiNil": "false"
}
},
"R58": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i4fdb048733b648df99bd7b1e5b1476ef_I20191231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ValuationAllowancesAndReservesBalance",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240144007 - Disclosure - Revenues - Reserves for Discounts and Allowances (Details)",
"role": "http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails",
"shortName": "Revenues - Reserves for Discounts and Allowances (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:AnalysisOfAmountOfAndChangeInProductRevenueReservesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "biib:CurrentProvisionsToProductRevenueReservesRelatingToSalesInCurrentYear",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R59": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:AnalysisOfAmountOfAndChangeInProductRevenueReservesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ValuationAllowancesAndReservesBalance",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240154008 - Disclosure - Revenues - Total Reserves for Discounts and Allowances (Details)",
"role": "http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails",
"shortName": "Revenues - Total Reserves for Discounts and Allowances (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:SummaryOfProductRevenueReservesIncludedInConsolidatedBalanceSheetsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i8f40d69597314563b8de93e06ec34c1c_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:ValuationAllowancesAndReservesBalance",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R6": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ProfitLoss",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "100050006 - Statement - Consolidated Statements of Cash Flows",
"role": "http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"shortName": "Consolidated Statements of Cash Flows",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:DepreciationDepletionAndAmortization",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R60": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240164009 - Disclosure - Revenues - Revenues from Anti-CD20 Therapeutic Programs (Details)",
"role": "http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails",
"shortName": "Revenues - Revenues from Anti-CD20 Therapeutic Programs (Details)",
"subGroupType": "details",
"uniqueAnchor": null
},
"R61": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240174010 - Disclosure - Revenues - Other Revenues (Details)",
"role": "http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"shortName": "Revenues - Other Revenues (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:OtherrevenuesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i87e510b249bf46dc94c9604a2b8b9a69_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:Revenues",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R62": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfInventoryCurrentTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InventoryRawMaterialsNetOfReserves",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240204011 - Disclosure - Inventory - Components of Inventory (Details)",
"role": "http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails",
"shortName": "Inventory - Components of Inventory (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfInventoryCurrentTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InventoryRawMaterialsNetOfReserves",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R63": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InventoryWriteDown",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240214012 - Disclosure - Inventory - Narrative (Details)",
"role": "http://www.biogenidec.com/role/InventoryNarrativeDetails",
"shortName": "Inventory - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:InventoryWriteDown",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R64": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:IntangibleAssetsExcludingGoodwillTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:FiniteLivedIntangibleAssetsGross",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240244013 - Disclosure - Intangible Assets and Goodwill - Summary of Intangible Assets (Details)",
"role": "http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails",
"shortName": "Intangible Assets and Goodwill - Summary of Intangible Assets (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:IntangibleAssetsExcludingGoodwillTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:FiniteLivedIntangibleAssetsGross",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R65": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "biib:AmortizationandImpairmentofAcquiredIntangibleAssets",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240254014 - Disclosure - Intangible Assets and Goodwill - Narrative (Details)",
"role": "http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"shortName": "Intangible Assets and Goodwill - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:AmortizationOfIntangibleAssets",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R66": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleofFiniteLivedIntangibleAssetsFutureAmortizationExpenseTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:FiniteLivedIntangibleAssetsAmortizationExpenseNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240264015 - Disclosure - Intangible Assets and Goodwill - Estimated Future Amortization of Intangible Assets (Details)",
"role": "http://www.biogenidec.com/role/IntangibleAssetsandGoodwillEstimatedFutureAmortizationofIntangibleAssetsDetails",
"shortName": "Intangible Assets and Goodwill - Estimated Future Amortization of Intangible Assets (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleofFiniteLivedIntangibleAssetsFutureAmortizationExpenseTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:FiniteLivedIntangibleAssetsAmortizationExpenseNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R67": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfGoodwillTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i4fdb048733b648df99bd7b1e5b1476ef_I20191231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Goodwill",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240274016 - Disclosure - Intangible Assets and Goodwill - Changes in Goodwill (Details)",
"role": "http://www.biogenidec.com/role/IntangibleAssetsandGoodwillChangesinGoodwillDetails",
"shortName": "Intangible Assets and Goodwill - Changes in Goodwill (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfGoodwillTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "id82ee39d50af4f5587e19230de06cf02_I20181231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:Goodwill",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R68": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:AvailableForSaleSecuritiesDebtSecurities",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240304017 - Disclosure - Fair Value Measurements - Summary of Assets and Liabilities Recorded at Fair Value (Details)",
"role": "http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"shortName": "Fair Value Measurements - Summary of Assets and Liabilities Recorded at Fair Value (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfFairValueAssetsAndLiabilitiesMeasuredOnRecurringBasisTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia4e82735011b489487c0fe39eb2af15c_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:CashAndCashEquivalentsFairValueDisclosure",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R69": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia29e9f9c6a0f4c099a0d0b872c4ab3e0_I20201231",
"decimals": "4",
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationContingentConsiderationLiabilityMeasurementInput",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240314018 - Disclosure - Fair Value Measurements - Summary of Significant Unobservable Inputs (Details)",
"role": "http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails",
"shortName": "Fair Value Measurements - Summary of Significant Unobservable Inputs (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia29e9f9c6a0f4c099a0d0b872c4ab3e0_I20201231",
"decimals": "4",
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationContingentConsiderationLiabilityMeasurementInput",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R7": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i23b12db8472d48b2b53334bb866cc37b_I20171231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:SharesIssued",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "100060007 - Statement - Consolidated Statements of Equity",
"role": "http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"shortName": "Consolidated Statements of Equity",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i23b12db8472d48b2b53334bb866cc37b_I20171231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:SharesIssued",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R70": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "biib:TechnologicalAndRegulatorySuccessProbability",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240324019 - Disclosure - Fair Value Measurements - Narrative (Details)",
"role": "http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"shortName": "Fair Value Measurements - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "biib:TechnologicalAndRegulatorySuccessProbability",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R71": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:IntangibleAssetsExcludingGoodwillTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:IntangibleAssetsGrossExcludingGoodwill",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240334020 - Disclosure - Fair Value Measurements - Gains and Losses on Assets Measured at Fair Value (Details)",
"role": "http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"shortName": "Fair Value Measurements - Gains and Losses on Assets Measured at Fair Value (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueAssetsMeasuredOnNonrecurringBasisTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i892b13c4cafa42ecb4c07f03ecf1b9af_I20190607",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:IntangibleAssetsGrossExcludingGoodwill",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R72": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueByBalanceSheetGroupingTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:NotesPayableFairValueDisclosure",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240344021 - Disclosure - Fair Value Measurements - Summary of Fair and Carrying Value of Debt Instruments (Details)",
"role": "http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"shortName": "Fair Value Measurements - Summary of Fair and Carrying Value of Debt Instruments (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueByBalanceSheetGroupingTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:NotesPayableFairValueDisclosure",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R73": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityGainLossIncludedInEarnings",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240354022 - Disclosure - Fair Value Measurements - Fair Value of Contingent Consideration Obligations (Details)",
"role": "http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails",
"shortName": "Fair Value Measurements - Fair Value of Contingent Consideration Obligations (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ic89f75651f684c4e8666f45255e68b39_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityGainLossIncludedInEarnings",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R74": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:CashEquivalentsAtCarryingValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240384023 - Disclosure - Financial Instruments - Summary of Financial Assets with Maturities (Details)",
"role": "http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails",
"shortName": "Financial Instruments - Summary of Financial Assets with Maturities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:CashEquivalentsAtCarryingValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R75": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:InvestmentsClassifiedByContractualMaturityDateTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240394024 - Disclosure - Financial Instruments - Summary of Marketable Securities (Details)",
"role": "http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails",
"shortName": "Financial Instruments - Summary of Marketable Securities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:AvailableForSaleDebtSecuritiesAccumulatedGrossUnrealizedGainBeforeTax",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R76": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:InvestmentsClassifiedByContractualMaturityDateTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:AvailableForSaleSecuritiesDebtMaturitiesWithinOneYearFairValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240404025 - Disclosure - Financial Instruments - Summary of Contractual Maturities (Details)",
"role": "http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails",
"shortName": "Financial Instruments - Summary of Contractual Maturities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:InvestmentsClassifiedByContractualMaturityDateTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:AvailableForSaleSecuritiesDebtMaturitiesWithinOneYearFairValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R77": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:RealizedGainLossOnInvestmentsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:ProceedsFromSaleAndMaturityOfAvailableForSaleSecurities",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240414026 - Disclosure - Financial Instruments - Proceeds from Marketable Debt Securities (Details)",
"role": "http://www.biogenidec.com/role/FinancialInstrumentsProceedsfromMarketableDebtSecuritiesDetails",
"shortName": "Financial Instruments - Proceeds from Marketable Debt Securities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:RealizedGainLossOnInvestmentsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:DebtSecuritiesAvailableForSaleRealizedGain",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R78": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:AverageMaturityOfMarketableSecurities",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240424027 - Disclosure - Financial Instruments - Narrative (Details)",
"role": "http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"shortName": "Financial Instruments - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "biib:AverageMaturityOfMarketableSecurities",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R79": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DerivativeRemainingMaturity1",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240454028 - Disclosure - Derivative Instruments - Narrative (Details)",
"role": "http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"shortName": "Derivative Instruments - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DerivativeRemainingMaturity1",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R8": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SignificantAccountingPoliciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210011001 - Disclosure - Summary of Significant Accounting Policies",
"role": "http://www.biogenidec.com/role/SummaryofSignificantAccountingPolicies",
"shortName": "Summary of Significant Accounting Policies",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SignificantAccountingPoliciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R80": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfNotionalAmountsOfOutstandingDerivativePositionsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icfac85de776a4ccdb1c2c9949959fc1e_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:DerivativeNotionalAmount",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240464029 - Disclosure - Derivative Instruments - Foreign Currency Forward Contracts Entered into Hedge Forecasted Revenues (Details)",
"role": "http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails",
"shortName": "Derivative Instruments - Foreign Currency Forward Contracts Entered into Hedge Forecasted Revenues (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfNotionalAmountsOfOutstandingDerivativePositionsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i9548c02f9465476885ffa4b8feb4449a_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:DerivativeNotionalAmount",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R81": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDerivativeInstrumentsGainLossInStatementOfFinancialPerformanceTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i970be973b11946e2ba4be4af29d27f00_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:OtherComprehensiveIncomeLossCashFlowHedgeGainLossReclassificationBeforeTax",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240474030 - Disclosure - Derivative Instruments - Summary of the Effect of Derivatives Designated as Cash Flow Hedging Instruments (Details)",
"role": "http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"shortName": "Derivative Instruments - Summary of the Effect of Derivatives Designated as Cash Flow Hedging Instruments (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDerivativeInstrumentsGainLossInStatementOfFinancialPerformanceTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i970be973b11946e2ba4be4af29d27f00_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:OtherComprehensiveIncomeLossCashFlowHedgeGainLossReclassificationBeforeTax",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R82": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfNetInvestmentHedgesInAccumulatedOtherComprehensiveIncomeLossTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ie215c6f8d19a49b09e42d9e80421319e_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:TranslationAdjustmentForNetInvestmentHedgeNetOfTaxPeriodIncreaseDecrease",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240484031 - Disclosure - Derivative Instruments - Summary of the Effect of Derivatives Designated as Net Investment Hedging Instruments (Details)",
"role": "http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"shortName": "Derivative Instruments - Summary of the Effect of Derivatives Designated as Net Investment Hedging Instruments (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfNetInvestmentHedgesInAccumulatedOtherComprehensiveIncomeLossTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ie215c6f8d19a49b09e42d9e80421319e_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:TranslationAdjustmentForNetInvestmentHedgeNetOfTaxPeriodIncreaseDecrease",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R83": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDerivativeInstrumentsInStatementOfFinancialPositionFairValueTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia2c6f6c468f64ddcb921d54c39e33bcb_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:DerivativeFairValueOfDerivativeAsset",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240494032 - Disclosure - Derivative Instruments - Summary of the Fair Value for our Outstanding Derivatives (Details)",
"role": "http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"shortName": "Derivative Instruments - Summary of the Fair Value for our Outstanding Derivatives (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDerivativeInstrumentsInStatementOfFinancialPositionFairValueTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ia2c6f6c468f64ddcb921d54c39e33bcb_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:DerivativeFairValueOfDerivativeAsset",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R84": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Land",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240524033 - Disclosure - Property, Plant and Equipment (Details)",
"role": "http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails",
"shortName": "Property, Plant and Equipment (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Land",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R85": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Depreciation",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240534034 - Disclosure - Property, Plant and Equipment - Narrative (Details)",
"role": "http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"shortName": "Property, Plant and Equipment - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:Depreciation",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R86": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ic4652a3a2e7940719a658dd16032604d_I20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseTermOfContract",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240564035 - Disclosure - Leases - Narrative (Details)",
"role": "http://www.biogenidec.com/role/LeasesNarrativeDetails",
"shortName": "Leases - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ic4652a3a2e7940719a658dd16032604d_I20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseTermOfContract",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R87": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeaseRightOfUseAsset",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240574036 - Disclosure - Leases - Schedule of Operating Leases (Details)",
"role": "http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails",
"shortName": "Leases - Schedule of Operating Leases (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:LesseeLeaseTableTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i1b65f5d0f33646d9bc123cea10741758_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:OperatingLeaseRightOfUseAsset",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R88": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:LeaseCostTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LeaseCost",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240584037 - Disclosure - Leases - Operating Lease Costs (Details)",
"role": "http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails",
"shortName": "Leases - Operating Lease Costs (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:LeaseCostTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LeaseCost",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R89": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240594038 - Disclosure - Leases - Operating Lease Liability Maturity (Details)",
"role": "http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails",
"shortName": "Leases - Operating Lease Liability Maturity (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R9": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "210061002 - Disclosure - Acquisitions",
"role": "http://www.biogenidec.com/role/Acquisitions",
"shortName": "Acquisitions",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R90": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:OperatingleaseweightedaverageremainingtermanddiscountrateTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeaseWeightedAverageRemainingLeaseTerm1",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240604039 - Disclosure - Leases - Operating Lease Weighted Average Remaining Term and Discount Rate (Details)",
"role": "http://www.biogenidec.com/role/LeasesOperatingLeaseWeightedAverageRemainingTermandDiscountRateDetails",
"shortName": "Leases - Operating Lease Weighted Average Remaining Term and Discount Rate (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:OperatingleaseweightedaverageremainingtermanddiscountrateTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeaseWeightedAverageRemainingLeaseTerm1",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R91": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:OperatingleasesupplementalcashflowdisclosureTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeasePayments",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240614040 - Disclosure - Leases - Operating Lease Supplemental Cash Flow Disclosure (Details)",
"role": "http://www.biogenidec.com/role/LeasesOperatingLeaseSupplementalCashFlowDisclosureDetails",
"shortName": "Leases - Operating Lease Supplemental Cash Flow Disclosure (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:OperatingleasesupplementalcashflowdisclosureTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeasePayments",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R92": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:DebtCurrent",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240644041 - Disclosure - Indebtedness - Summary of Indebtedness (Details)",
"role": "http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails",
"shortName": "Indebtedness - Summary of Indebtedness (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfDebtTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "ied6fc4314ff74454a352c1254c20f4a6_I20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:DebtCurrent",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R93": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:DebtInstrumentRedemptionPricePercentage",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240654042 - Disclosure - Indebtedness - Narrative (Details)",
"role": "http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"shortName": "Indebtedness - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:DebtInstrumentRedemptionPricePercentage",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R94": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfMaturitiesOfLongTermDebtTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240664043 - Disclosure - Indebtedness - Total Debt Maturities (Details)",
"role": "http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails",
"shortName": "Indebtedness - Total Debt Maturities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfMaturitiesOfLongTermDebtTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R95": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:PreferredStockSharesAuthorized",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240694044 - Disclosure - Equity - Narrative (Details)",
"role": "http://www.biogenidec.com/role/EquityNarrativeDetails",
"shortName": "Equity - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:PreferredStockSharesAuthorized",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R96": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:SummaryOfCommonStockTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:CommonStockSharesAuthorized",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240704045 - Disclosure - Equity - Summary of Common Stock (Details)",
"role": "http://www.biogenidec.com/role/EquitySummaryofCommonStockDetails",
"shortName": "Equity - Summary of Common Stock (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"biib:SummaryOfCommonStockTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i5e9a4ee8449a49d88a3bbe9b29cab624_I20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:CommonStockSharesAuthorized",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R97": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i4fdb048733b648df99bd7b1e5b1476ef_I20191231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:AccumulatedOtherComprehensiveIncomeLossNetOfTax",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240714046 - Disclosure - Equity - Accumulated Other Comprehensive Income (Loss) (Details)",
"role": "http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"shortName": "Equity - Accumulated Other Comprehensive Income (Loss) (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfAccumulatedOtherComprehensiveIncomeLossTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:OtherComprehensiveIncomeLossBeforeReclassificationsNetOfTax",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R98": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfOtherNonoperatingIncomeExpenseTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:NonoperatingIncomeExpense",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240724047 - Disclosure - Equity - Reclassification out of Accumulated Other Comprehensive Income (Details)",
"role": "http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails",
"shortName": "Equity - Reclassification out of Accumulated Other Comprehensive Income (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i1029aaf02d1e4e45bfea6b6f58c10380_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:NetIncomeLoss",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R99": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "i6841825fce32422eb1457cf2ad6e770d_D20201001-20201231",
"decimals": "-5",
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLoss",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "240754048 - Disclosure - Earnings per Share - Basic and Diluted Earnings Per Share (Details)",
"role": "http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails",
"shortName": "Earnings per Share - Basic and Diluted Earnings Per Share (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "biib-20201231.htm",
"contextRef": "icb3a3eb7d9cc449ebce025a800169b95_D20200101-20201231",
"decimals": "-5",
"lang": "en-US",
"name": "us-gaap:WeightedAverageNumberDilutedSharesOutstandingAdjustment",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
}
},
"segmentCount": 237,
"tag": {
"biib_A2015ESPPMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "2015 ESPP [Member]",
"label": "2015 ESPP [Member]",
"terseLabel": "2015 ESPP"
}
}
},
"localname": "A2015ESPPMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails"
],
"xbrltype": "domainItemType"
},
"biib_A2015SeniorNotesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "2015 Senior Notes",
"label": "2015 Senior Notes [Member]",
"terseLabel": "2015 Senior Notes"
}
}
},
"localname": "A2015SeniorNotesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_A2017OmnibusEquityPlanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "2017 Omnibus Equity Plan",
"label": "2017 Omnibus Equity Plan [Member]",
"terseLabel": "2017 Omnibus Equity Plan"
}
}
},
"localname": "A2017OmnibusEquityPlanMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_A2020SeniorNotesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "2020 Senior Notes",
"label": "2020 Senior Notes [Member]",
"terseLabel": "2020 Senior Notes"
}
}
},
"localname": "A2020SeniorNotesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_A225SeniorNotesDueMay12030Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "2.25% Senior Notes due May 1, 2030",
"label": "2.25% Senior Notes due May 1, 2030 [Member]",
"terseLabel": "2.250% Senior Notes due May 1, 2030"
}
}
},
"localname": "A225SeniorNotesDueMay12030Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"biib_A315SeniorNotesDueMay12050Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "3.15% Senior Notes due May 1, 2050",
"label": "3.15% Senior Notes due May 1, 2050 [Member]",
"terseLabel": "3.150% Senior Notes due May 1, 2050"
}
}
},
"localname": "A315SeniorNotesDueMay12050Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"biib_AccruedExpensesOnFundingCommitmentRelatedToClinicalResearch": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Accrued expenses on funding commitment related to clinical research.",
"label": "Accrued Expenses On Funding Commitment Related To Clinical Research",
"verboseLabel": "Accrued expenses"
}
}
},
"localname": "AccruedExpensesOnFundingCommitmentRelatedToClinicalResearch",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_AcordaMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Acorda.",
"label": "Acorda [Member]",
"terseLabel": "Acorda"
}
}
},
"localname": "AcordaMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails"
],
"xbrltype": "domainItemType"
},
"biib_AdditionalMilestonePayment": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Additional milestone payment.",
"label": "Additional Milestone Payment",
"terseLabel": "Total aducanumab collaboration third party milestone expense"
}
}
},
"localname": "AdditionalMilestonePayment",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Additionalreductioninroyaltyratepayableoncommercialsales": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Additional reduction in royalty rate payable on commercial sales",
"label": "Additional reduction in royalty rate payable on commercial sales",
"terseLabel": "Additional reduction in royalty rate payable on commercial sales"
}
}
},
"localname": "Additionalreductioninroyaltyratepayableoncommercialsales",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "percentItemType"
},
"biib_AdjustmentsToProductRevenueReservesRelatingToSalesInPriorYears": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Adjustments to product revenue reserves relating to sales in prior years.",
"label": "Adjustments To Product Revenue Reserves Relating To Sales In Prior Years",
"negatedTerseLabel": "Adjustments relating to prior years"
}
}
},
"localname": "AdjustmentsToProductRevenueReservesRelatingToSalesInPriorYears",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_AdministrativeSpaceMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Administrative Space",
"label": "Administrative Space [Member]",
"terseLabel": "Administrative Space"
}
}
},
"localname": "AdministrativeSpaceMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_AducanumabMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Aducanumab [Member]",
"label": "Aducanumab [Member]",
"terseLabel": "Aducanumab"
}
}
},
"localname": "AducanumabMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_AliveGenMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "AliveGen [Member]",
"label": "AliveGen [Member]",
"terseLabel": "AliveGen"
}
}
},
"localname": "AliveGenMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_AlkermesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Alkermes [Member]",
"label": "Alkermes [Member]",
"terseLabel": "Alkermes"
}
}
},
"localname": "AlkermesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_AmortizationandImpairmentofAcquiredIntangibleAssets": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 4.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amortization and Impairment of Acquired Intangible Assets",
"label": "Amortization and Impairment of Acquired Intangible Assets",
"terseLabel": "Amortization and impairment of acquired intangible assets"
}
}
},
"localname": "AmortizationandImpairmentofAcquiredIntangibleAssets",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_AnalysisOfAmountOfAndChangeInProductRevenueReservesTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Analysis of amount of and change in product revenue reserves.",
"label": "Analysis Of Amount Of And Change In Product Revenue Reserves [Table Text Block]",
"terseLabel": "Analysis of change in reserves"
}
}
},
"localname": "AnalysisOfAmountOfAndChangeInProductRevenueReservesTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_AssumptionsUsedInValuationOfMarketBasedStockUnitsTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Assumptions used in valuation of market based stock units.",
"label": "Assumptions Used In Valuation Of Market Based Stock Units [Table Text Block]",
"terseLabel": "Assumptions used in valuation of market based stock units"
}
}
},
"localname": "AssumptionsUsedInValuationOfMarketBasedStockUnitsTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_AvailableForSaleSecuritiesDebtMaturitiesAfterFiveYearsAmortizedCost": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": 3.0,
"parentTag": "us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "This item represents debt securities, at cost, net of adjustments made for accretion, amortization, other-than-temporary impairments, and hedging, if any, which are expected to mature after five years from the balance sheet date and which are categorized neither as held-to-maturity nor trading securities.",
"label": "Available For Sale Securities Debt Maturities After Five Years Amortized Cost",
"verboseLabel": "Due after five years"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesAfterFiveYearsAmortizedCost",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_AvailableForSaleSecuritiesDebtMaturitiesAfterFiveYearsFairValue": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": 3.0,
"parentTag": "us-gaap_AvailableForSaleSecuritiesDebtSecurities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "This item represents the fair value of debt securities, which are expected to mature after five years from the balance sheet date and which are categorized neither as held-to-maturity nor trading securities.",
"label": "Available For Sale Securities Debt Maturities After Five Years Fair Value",
"verboseLabel": "Due after five years"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesAfterFiveYearsFairValue",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_AverageMaturityOfMarketableSecurities": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Average remaining time to maturity of marketable debt securities, available-for-sale securities.",
"label": "Average Maturity Of Marketable Securities",
"verboseLabel": "Average maturity of marketable securities, months"
}
}
},
"localname": "AverageMaturityOfMarketableSecurities",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"biib_AverageStockPriceOnGrantDateInDollarsMaximum": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Average stock price on grant date in dollars maximum.",
"label": "Average Stock Price On Grant Date In Dollars Maximum",
"verboseLabel": "Average stock price on grant date maximum"
}
}
},
"localname": "AverageStockPriceOnGrantDateInDollarsMaximum",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_AverageStockPriceOnGrantDateInDollarsMinimum": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Average stock price on grant date in dollars minimum.",
"label": "Average Stock Price On Grant Date In Dollars Minimum",
"verboseLabel": "Average stock price on grant date minimum"
}
}
},
"localname": "AverageStockPriceOnGrantDateInDollarsMinimum",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_BENEPALIMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "BENEPALI [Member]",
"label": "BENEPALI [Member]",
"terseLabel": "BENEPALI"
}
}
},
"localname": "BENEPALIMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_BIIB054Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "BIIB054",
"label": "BIIB054 [Member]",
"terseLabel": "BIIB054"
}
}
},
"localname": "BIIB054Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_BIIB111Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "BIIB111 [Member]",
"label": "BIIB111 [Member]",
"terseLabel": "BIIB111"
}
}
},
"localname": "BIIB111Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_BIIB112Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "BIIB112 [Member]",
"label": "BIIB112 [Member]",
"terseLabel": "BIIB112"
}
}
},
"localname": "BIIB112Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_BIIB118Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "BIIB118",
"label": "BIIB118 [Member]",
"terseLabel": "BIIB118"
}
}
},
"localname": "BIIB118Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_BiogenIdecInternationalNeuroscienceMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Biogen idec international neuroscience.",
"label": "Biogen Idec International Neuroscience Member",
"terseLabel": "Biogen Idec International Neuroscience GmbH"
}
}
},
"localname": "BiogenIdecInternationalNeuroscienceMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_Biogenshareofcopromotionprofitsorlosses": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Biogen share of co-promotion profits or losses",
"label": "Biogen share of co-promotion profits or losses",
"terseLabel": "Biogen share of co-promotion profits or losses"
}
}
},
"localname": "Biogenshareofcopromotionprofitsorlosses",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails"
],
"xbrltype": "percentItemType"
},
"biib_BiologicsManufacturingMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Biologics Manufacturing",
"label": "Biologics Manufacturing [Member]",
"terseLabel": "Biologics Manufacturing"
}
}
},
"localname": "BiologicsManufacturingMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_BiosimilarsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Biosimilars",
"label": "Biosimilars [Member]",
"terseLabel": "Subtotal: Biosimilars"
}
}
},
"localname": "BiosimilarsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_BioverativMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Bioverativ [Member]",
"label": "Bioverativ [Member]",
"terseLabel": "Bioverativ"
}
}
},
"localname": "BioverativMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_BristolMyersSquibbMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Bristol-Myers Squibb [Member]",
"label": "Bristol-Myers Squibb [Member]",
"terseLabel": "Bristol-Myers Squibb"
}
}
},
"localname": "BristolMyersSquibbMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails"
],
"xbrltype": "domainItemType"
},
"biib_BusinessOverviewPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Describes an overview of the company and its operations.",
"label": "Business Overview [Policy Text Block]",
"terseLabel": "Business Overview"
}
}
},
"localname": "BusinessOverviewPolicyTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"biib_CashSettledPerformanceSharesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Cash settled performance shares [Member]",
"label": "Cash settled performance shares [Member]",
"terseLabel": "Cash settled performance units"
}
}
},
"localname": "CashSettledPerformanceSharesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_CashflowsoperatingexpensesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Cash flows, operating expenses [Member]",
"label": "Cash flows, operating expenses [Member]",
"terseLabel": "Cash flows, operating expenses"
}
}
},
"localname": "CashflowsoperatingexpensesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails"
],
"xbrltype": "domainItemType"
},
"biib_CashflowsrevenueMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Cash flows, revenue [Member]",
"label": "Cash flows, revenue [Member]",
"terseLabel": "Cash flows, revenue"
}
}
},
"localname": "CashflowsrevenueMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails"
],
"xbrltype": "domainItemType"
},
"biib_ClinicalAssetsAcquired": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Clinical Assets Acquired",
"label": "Clinical Assets Acquired",
"terseLabel": "Clinical assets acquired"
}
}
},
"localname": "ClinicalAssetsAcquired",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"biib_CoPromotionOperatingProfitThresholdToDetermineShareOfCoPromotionOperatingProfitPriorToAmendment": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Co promotion operating profit threshold to determine share of co promotion operating profit prior to amendment.",
"label": "Co Promotion Operating Profit Threshold To Determine Share Of Co Promotion Operating Profit Prior To Amendment",
"verboseLabel": "Co-promotion operating profit threshold for Rituxan in U S and Canada to determine share of co promotion operating profit prior to amendment"
}
}
},
"localname": "CoPromotionOperatingProfitThresholdToDetermineShareOfCoPromotionOperatingProfitPriorToAmendment",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_CoPromotionProfitSharingFormulaAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Co-promotion profit sharing formula.",
"label": "Co Promotion Profit Sharing Formula [Abstract]",
"verboseLabel": "Co-promotion profit sharing formula"
}
}
},
"localname": "CoPromotionProfitSharingFormulaAbstract",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails"
],
"xbrltype": "stringItemType"
},
"biib_CoPromotionProfitSharingFormulaTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Co-promotion profit sharing formula.",
"label": "Co Promotion Profit Sharing Formula [Table Text Block]",
"verboseLabel": "Co-promotion profit sharing formula"
}
}
},
"localname": "CoPromotionProfitSharingFormulaTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_CollaborationProfitSharing": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 6.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Payments are made to Elan for their share of rest of world net operating profits to effect an equal sharing of collaboration operating profit. These payments include the reimbursement of our portion of third-party royalties that Elan pays on behalf of the collaboration, relating to sales outside of the U.S. These amounts are reflected in the collaboration profit sharing line in our consolidated statement of income. As sales of TYSABRI outside the U.S. increase, our collaboration profit sharing expense will increase.",
"label": "Collaboration profit sharing",
"terseLabel": "Collaboration profit (loss) sharing"
}
}
},
"localname": "CollaborationProfitSharing",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"biib_Collaborationexpensesaccrual": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": 3.0,
"parentTag": "us-gaap_OtherLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Collaboration expenses accrual",
"label": "Collaboration expenses accrual",
"terseLabel": "Collaboration expenses"
}
}
},
"localname": "Collaborationexpensesaccrual",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_CommercialMilestonesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Commercial Milestones [Member]",
"label": "Commercial Milestones [Member]",
"terseLabel": "Commercial Milestones"
}
}
},
"localname": "CommercialMilestonesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ComputerSoftwareAndHardware": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 5.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Computer software and hardware",
"label": "Computer software and hardware",
"terseLabel": "Computer software and hardware"
}
}
},
"localname": "ComputerSoftwareAndHardware",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ContractManufacturingCustomerMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Contract Manufacturing Customer",
"label": "Contract Manufacturing Customer [Member]",
"terseLabel": "Contract Manufacturing Customer"
}
}
},
"localname": "ContractManufacturingCustomerMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ContractOptionExerciseFee": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Contract Option Exercise Fee",
"label": "Contract Option Exercise Fee",
"terseLabel": "Option exercise fee"
}
}
},
"localname": "ContractOptionExerciseFee",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ContractualAdjustmentsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Contractual adjustments.",
"label": "Contractual Adjustments [Member]",
"terseLabel": "Contractual Adjustments"
}
}
},
"localname": "ContractualAdjustmentsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "domainItemType"
},
"biib_ConvergencePharmaceuticalsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Convergence Pharmaceuticals [Member]",
"label": "Convergence Pharmaceuticals [Member]",
"terseLabel": "Convergence Pharmaceuticals"
}
}
},
"localname": "ConvergencePharmaceuticalsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_CorporateDebtSecuritiesCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Corporate debt securities Current.",
"label": "Corporate Debt Securities Current [Member]",
"verboseLabel": "Corporate debt securities Current"
}
}
},
"localname": "CorporateDebtSecuritiesCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_CorporateDebtSecuritiesNonCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Corporate Debt Securities Non Current.",
"label": "Corporate Debt Securities Non Current [Member]",
"verboseLabel": "Corporate debt securities Non-current"
}
}
},
"localname": "CorporateDebtSecuritiesNonCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_CumulativeSalesLevel": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Cumulative sales level",
"label": "Cumulative sales level",
"terseLabel": "Cumulative sales level"
}
}
},
"localname": "CumulativeSalesLevel",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_CumulativeSalesLevelAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Cumulative sales level [Axis]",
"label": "Cumulative sales level [Axis]",
"terseLabel": "Cumulative sales level [Axis]"
}
}
},
"localname": "CumulativeSalesLevelAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_CumulativeSalesLevelDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Domain] for Cumulative sales level [Axis]",
"label": "Cumulative sales level [Domain]",
"terseLabel": "Cumulative sales level [Domain]"
}
}
},
"localname": "CumulativeSalesLevelDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_CurrentProvisionsToProductRevenueReservesRelatingToSalesInCurrentYear": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Current provisions to product revenue reserves relating to sales in current year.",
"label": "Current Provisions To Product Revenue Reserves Relating To Sales In Current Year",
"terseLabel": "Current provisions relating to sales in current year"
}
}
},
"localname": "CurrentProvisionsToProductRevenueReservesRelatingToSalesInCurrentYear",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DebtInstrumentMakeWholeProvisionRedemptionPricePercentage": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Make Whole Provision Redemption Price, Percentage",
"label": "Debt Instrument, Make Whole Provision Redemption Price, Percentage",
"terseLabel": "Redemption percentage for change in control provision"
}
}
},
"localname": "DebtInstrumentMakeWholeProvisionRedemptionPricePercentage",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_DeferredTaxAssetLiabilityTypeAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Deferred Tax Asset/Liability Type [Axis]",
"label": "Deferred Tax Asset/Liability Type [Axis]",
"terseLabel": "Deferred Tax Asset/Liability Type [Axis]"
}
}
},
"localname": "DeferredTaxAssetLiabilityTypeAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_DeferredTaxAssetLiabilityTypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Domain] for Deferred Tax Asset/Liability Type [Axis]",
"label": "Deferred Tax Asset/Liability Type [Domain]",
"terseLabel": "Deferred Tax Asset/Liability Type [Domain]"
}
}
},
"localname": "DeferredTaxAssetLiabilityTypeDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DeferredTaxAssetsValueReductionInValue": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred Tax Assets, Value, Reduction in Value",
"label": "Deferred Tax Assets, Value, Reduction in Value",
"terseLabel": "Deferred tax assets, value, reduction in value"
}
}
},
"localname": "DeferredTaxAssetsValueReductionInValue",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DeferredTaxLiabilitiesDepreciationAmortizationOther": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 4.0,
"parentTag": "us-gaap_DeferredTaxLiabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred tax liabilities depreciation amortization other.",
"label": "Deferred Tax Liabilities Depreciation Amortization Other",
"negatedTerseLabel": "Depreciation, amortization and other"
}
}
},
"localname": "DeferredTaxLiabilitiesDepreciationAmortizationOther",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DeferredTaxLiabilitiesTaxCreditCarryforwards": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 3.0,
"parentTag": "us-gaap_DeferredTaxLiabilities",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred Tax Liabilities, Tax Credit Carryforwards",
"label": "Deferred Tax Liabilities, Tax Credit Carryforwards",
"terseLabel": "Tax credits"
}
}
},
"localname": "DeferredTaxLiabilitiesTaxCreditCarryforwards",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DeferredTaxLiabilitiesValueReductionInValue": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred Tax Liabilities, Value, Reduction in Value",
"label": "Deferred Tax Liabilities, Value, Reduction in Value",
"terseLabel": "Deferred tax liabilities, value, reduction in value"
}
}
},
"localname": "DeferredTaxLiabilitiesValueReductionInValue",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DeferredtaxliabilitiesGILTItaxcalculation": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 2.0,
"parentTag": "us-gaap_DeferredTaxLiabilities",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred tax liabilities, GILTI tax calculation",
"label": "Deferred tax liabilities, GILTI tax calculation",
"terseLabel": "GILTI"
}
}
},
"localname": "DeferredtaxliabilitiesGILTItaxcalculation",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Deferredtaxliabilitiestemporarydifferencespurchaseaccountingforeignsubsidiaries": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred tax liabilities, temporary differences, purchase accounting, foreign subsidiaries",
"label": "Deferred tax liabilities, temporary differences, purchase accounting, foreign subsidiaries",
"terseLabel": "Deferred tax liabilities, temporary differences, purchase accounting, foreign subsidiaries"
}
}
},
"localname": "Deferredtaxliabilitiestemporarydifferencespurchaseaccountingforeignsubsidiaries",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DeferredtaxliabilityMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Deferred tax liability [Member]",
"label": "Deferred tax liability [Member]",
"terseLabel": "Deferred tax liability"
}
}
},
"localname": "DeferredtaxliabilityMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DenaliAndSangamoMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Denali and Sangamo",
"label": "Denali and Sangamo [Member]",
"terseLabel": "Denali and Sangamo"
}
}
},
"localname": "DenaliAndSangamoMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DenaliTherapeuticsIncMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Denali Therapeutics Inc.",
"label": "Denali Therapeutics Inc. [Member]",
"terseLabel": "Denali Therapeutics"
}
}
},
"localname": "DenaliTherapeuticsIncMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DenmarkManufacturingOperationsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Denmark Manufacturing Operations [Member]",
"label": "Denmark Manufacturing Operations [Member]",
"terseLabel": "Denmark Manufacturing Operations"
}
}
},
"localname": "DenmarkManufacturingOperationsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DenominatorAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Denominator.",
"label": "Denominator [Abstract]",
"verboseLabel": "Denominator:"
}
}
},
"localname": "DenominatorAbstract",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "stringItemType"
},
"biib_DerivativeMaturityAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Derivative Maturity [Axis]",
"label": "Derivative Maturity [Axis]",
"terseLabel": "Derivative Maturity [Axis]"
}
}
},
"localname": "DerivativeMaturityAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_DerivativeMaturityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Domain] for Derivative Maturity [Axis]",
"label": "Derivative Maturity [Domain]",
"terseLabel": "Derivative Maturity [Domain]"
}
}
},
"localname": "DerivativeMaturityDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_Derivativesusedinnetinvestmenthedgenetoftaxexcludedcomponentincreasedecrease": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Derivatives used in net investment hedge, net of tax, excluded component increase (decrease)",
"label": "Derivatives used in net investment hedge, net of tax, excluded component increase (decrease)",
"terseLabel": "Net Gains/(Losses) Recognized in Other Comprehensive Income (Amounts Excluded from Effectiveness Testing) (in millions)"
}
}
},
"localname": "Derivativesusedinnetinvestmenthedgenetoftaxexcludedcomponentincreasedecrease",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DevelopmentMilestonesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Development Milestones [Member]",
"label": "Development Milestones [Member]",
"terseLabel": "Development Milestones"
}
}
},
"localname": "DevelopmentMilestonesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DirectorsPlanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Directors plan.",
"label": "Directors Plan [Member]",
"verboseLabel": "Directors Plan"
}
}
},
"localname": "DirectorsPlanMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DisputedClaimsRelatingToConstructionCosts": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Disputed Claims Relating to Construction Costs",
"label": "Disputed Claims Relating to Construction Costs",
"terseLabel": "Disputed claims"
}
}
},
"localname": "DisputedClaimsRelatingToConstructionCosts",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/LitigationNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_DistributorOneMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Distributor one.",
"label": "Distributor One [Member]",
"terseLabel": "Distributor One"
}
}
},
"localname": "DistributorOneMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DistributorTwoMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Distributor two.",
"label": "Distributor Two [Member]",
"terseLabel": "Distributor Two"
}
}
},
"localname": "DistributorTwoMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_DivestituresAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Divestitures [Abstract]",
"label": "Divestitures [Abstract]",
"terseLabel": "Divestitures [Abstract]"
}
}
},
"localname": "DivestituresAbstract",
"nsuri": "http://www.biogenidec.com/20201231",
"xbrltype": "stringItemType"
},
"biib_DividendYieldPercentage": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Dividend Yield Percentage",
"label": "Dividend Yield Percentage",
"terseLabel": "Dividend yield percentage"
}
}
},
"localname": "DividendYieldPercentage",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_DueFromUnconsolidatedJointBusiness": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 5.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Receivable from unconsolidated joint business for our share of the pre-tax co-promotion profits generated from our co-promotion arrangement for sales of RITUXAN.",
"label": "Due from unconsolidated joint business",
"verboseLabel": "Due from anti-CD20 therapeutic programs"
}
}
},
"localname": "DueFromUnconsolidatedJointBusiness",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"biib_E2609andBAN2401Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "E2609 and BAN2401 [Member]",
"label": "E2609 and BAN2401 [Member]",
"terseLabel": "E2609 and BAN2401"
}
}
},
"localname": "E2609andBAN2401Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails"
],
"xbrltype": "domainItemType"
},
"biib_EachAdditionalOneBillionUpToTwentyBillionMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Each additional one billion up to twenty billion [Member]",
"label": "Each additional one billion up to twenty billion [Member]",
"terseLabel": "Each additional one billion up to twenty billion"
}
}
},
"localname": "EachAdditionalOneBillionUpToTwentyBillionMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_EffectiveIncomeTaxRateReconciliationChangeinForeignTaxRatePercent": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 10.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Effective Income Tax Rate Reconciliation, Change in Foreign Tax Rate, Percent",
"label": "Effective Income Tax Rate Reconciliation, Change in Foreign Tax Rate, Percent",
"terseLabel": "Swiss tax reform"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationChangeinForeignTaxRatePercent",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"biib_EffectiveIncomeTaxRateReconciliationIntellectualPropertyRightsReorganization": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 7.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Effective Income Tax Rate Reconciliation, Intellectual Property Rights Reorganization",
"label": "Effective Income Tax Rate Reconciliation, Intellectual Property Rights Reorganization",
"terseLabel": "Internal reorganization of certain intellectual property rights"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationIntellectualPropertyRightsReorganization",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"biib_EffectiveTaxRateReconciliationGILTItax": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 8.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Effective Tax Rate Reconciliation, GILTI tax",
"label": "Effective Tax Rate Reconciliation, GILTI tax",
"terseLabel": "GILTI"
}
}
},
"localname": "EffectiveTaxRateReconciliationGILTItax",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"biib_EisaiMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Eisai [Member]",
"label": "Eisai [Member]",
"terseLabel": "Eisai"
}
}
},
"localname": "EisaiMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_EmployeeStockPurchasePlanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Employee stock purchase plan.",
"label": "Employee Stock Purchase Plan [Member]",
"verboseLabel": "Employee stock purchase plan"
}
}
},
"localname": "EmployeeStockPurchasePlanMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_EntityWidePercentageOfRevenueFromMajorDistributors": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Entity wide percentage of revenue from major distributors.",
"label": "Entity Wide Percentage Of Revenue From Major Distributors",
"terseLabel": "Percentage of revenue from major distributors"
}
}
},
"localname": "EntityWidePercentageOfRevenueFromMajorDistributors",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_EquityMethodInvestmentOwnershipPercentageBeforeAdditionalPurchaseTransaction": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Equity Method Investment, Ownership Percentage Before Additional Purchase Transaction",
"label": "Equity Method Investment, Ownership Percentage Before Additional Purchase Transaction",
"terseLabel": "Samsung Bioepis, ownership percentage before additional purchase transaction"
}
}
},
"localname": "EquityMethodInvestmentOwnershipPercentageBeforeAdditionalPurchaseTransaction",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_EquityMethodInvestmentsExpectedProfitShare": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Equity Method Investments, Expected Profit Share",
"label": "Equity Method Investments, Expected Profit Share",
"terseLabel": "Expected profit share percentage"
}
}
},
"localname": "EquityMethodInvestmentsExpectedProfitShare",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "percentItemType"
},
"biib_EquitySecuritiesAvailableForSaleAccumulatedGrossUnrealizedGainBeforeTax": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": 1.0,
"parentTag": "us-gaap_EquitySecuritiesFvNiCost",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Equity Securities, Available-for-sale, Accumulated Gross Unrealized Gain, before Tax",
"label": "Equity Securities, Available-for-sale, Accumulated Gross Unrealized Gain, before Tax",
"terseLabel": "Marketable equity securities, gross unrealized gains"
}
}
},
"localname": "EquitySecuritiesAvailableForSaleAccumulatedGrossUnrealizedGainBeforeTax",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_EquitySecuritiesAvailableForSaleAccumulatedGrossUnrealizedLossBeforeTax": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": 2.0,
"parentTag": "us-gaap_EquitySecuritiesFvNiCost",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Equity Securities, Available-for-Sale, Accumulated Gross Unrealized Loss, Before Tax",
"label": "Equity Securities, Available-for-Sale, Accumulated Gross Unrealized Loss, Before Tax",
"negatedTerseLabel": "Marketable equity securities, gross unrealized losses"
}
}
},
"localname": "EquitySecuritiesAvailableForSaleAccumulatedGrossUnrealizedLossBeforeTax",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_EquitySecuritiesCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Equity Securities, Current",
"label": "Equity Securities, Current [Member]",
"terseLabel": "Marketable equity securities, current"
}
}
},
"localname": "EquitySecuritiesCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_EquitySecuritiesNonCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Equity Securities, Non-Current",
"label": "Equity Securities, Non-Current [Member]",
"terseLabel": "Marketable equity securities, non-current"
}
}
},
"localname": "EquitySecuritiesNonCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_Equitymethodinvestmentbasisdifferenceamortizationperiod": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Equity method investment basis difference amortization period",
"label": "Equity method investment basis difference amortization period",
"terseLabel": "Equity method investment basis difference amortization period"
}
}
},
"localname": "Equitymethodinvestmentbasisdifferenceamortizationperiod",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "durationItemType"
},
"biib_EstimatedAdditionalPaymentsUponAchievementOfDevelopmentAndCommercialMilestones": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Estimated additional payments upon achievement of development and commercial milestones.",
"label": "Estimated Additional Payments Upon Achievement Of Development And Commercial Milestones",
"terseLabel": "Estimated additional payments upon achievement of development and commercial milestones"
}
}
},
"localname": "EstimatedAdditionalPaymentsUponAchievementOfDevelopmentAndCommercialMilestones",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ExpectedAdditionalMilestonePaymentsWhenMeetingCertainSalesThreshold": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Expected additional milestone payments when meeting certain sales threshold.",
"label": "Expected Additional Milestone Payments When Meeting Certain Sales Threshold",
"verboseLabel": "Expected additional milestone payments when certain sales threshold is met"
}
}
},
"localname": "ExpectedAdditionalMilestonePaymentsWhenMeetingCertainSalesThreshold",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ExpectedLicenseFeeAndRegulatoryMilestonePayments": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Expected license fee and regulatory milestone payments.",
"label": "Expected License Fee And Regulatory Milestone Payments",
"terseLabel": "Expected license fee and regulatory milestone payments"
}
}
},
"localname": "ExpectedLicenseFeeAndRegulatoryMilestonePayments",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ExpectedPaymentTimelineofMilestoneAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Expected Payment Timeline of Milestone [Axis]",
"label": "Expected Payment Timeline of Milestone [Axis]",
"terseLabel": "Expected Payment Timeline of Milestone [Axis]"
}
}
},
"localname": "ExpectedPaymentTimelineofMilestoneAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "stringItemType"
},
"biib_ExpectedPaymentTimelineofMilestoneDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Domain] for Expected Payment Timeline of Milestone [Axis]",
"label": "Expected Payment Timeline of Milestone [Domain]",
"terseLabel": "Expected Payment Timeline of Milestone [Domain]"
}
}
},
"localname": "ExpectedPaymentTimelineofMilestoneDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_ExpenseIncurredByCollaboration": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Total expense incurred by collaboration.",
"label": "Expense Incurred By Collaboration",
"terseLabel": "Total development expense incurred by the collaboration related to the advancement of BAN2401 and elenbecestat"
}
}
},
"localname": "ExpenseIncurredByCollaboration",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Expenseincurredbythecollaboration": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Expense incurred by the collaboration",
"label": "Expense incurred by the collaboration",
"terseLabel": "Total UCB development expense",
"verboseLabel": "Total sales and marketing expense incurred by the collaboration"
}
}
},
"localname": "Expenseincurredbythecollaboration",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Expensereflectedwithinstatementsofincome": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Expense reflected within statements of income",
"label": "Expense reflected within statements of income",
"terseLabel": "Biogen's share of UCB development expense reflected in research and development expense in our consolidated statements of income"
}
}
},
"localname": "Expensereflectedwithinstatementsofincome",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_FAMPYRAMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "FAMPYRA [Member]",
"label": "FAMPYRA [Member]",
"terseLabel": "FAMPYRA"
}
}
},
"localname": "FAMPYRAMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_FLIXABIMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "FLIXABI [Member]",
"label": "FLIXABI [Member]",
"terseLabel": "FLIXABI"
}
}
},
"localname": "FLIXABIMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_FUMADERMMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "FUMADERM [Member]",
"label": "FUMADERM [Member]",
"terseLabel": "FUMADERM"
}
}
},
"localname": "FUMADERMMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_FacilityLocationAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "-- None. No documentation exists for this element. --",
"label": "Facility Location [Axis]",
"terseLabel": "Facility Location [Axis]"
}
}
},
"localname": "FacilityLocationAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_FacilityLocationDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "-- None. No documentation exists for this element. --",
"label": "Facility Location [Domain]",
"terseLabel": "Facility Location [Domain]"
}
}
},
"localname": "FacilityLocationDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_FacilityTypeAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Facility Type",
"label": "Facility Type [Axis]",
"terseLabel": "Facility Type [Axis]"
}
}
},
"localname": "FacilityTypeAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_FacilityTypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Facility Type",
"label": "Facility Type [Domain]",
"terseLabel": "Facility Type [Domain]"
}
}
},
"localname": "FacilityTypeDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_FairValueIndefiniteLivedIntangibleAssetsDiscountRate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Fair Value, Indefinite Lived Intangible Assets, Discount Rate",
"label": "Fair Value, Indefinite Lived Intangible Assets, Discount Rate",
"terseLabel": "Fair value, in-process research and development, discount rate"
}
}
},
"localname": "FairValueIndefiniteLivedIntangibleAssetsDiscountRate",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_FairValueMeasurementInputs": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Fair Value Measurement Inputs",
"label": "Fair Value Measurement Inputs",
"terseLabel": "Fair value measurement and measurement inputs, recurring and nonrecurring"
}
}
},
"localname": "FairValueMeasurementInputs",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_ForeignSalesRequiredToTriggerMilestone": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Foreign sales required to trigger milestone.",
"label": "Foreign Sales Required To Trigger Milestone",
"verboseLabel": "Foreign sales required to trigger milestone"
}
}
},
"localname": "ForeignSalesRequiredToTriggerMilestone",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ForeignearningsintheformofcashandcashequivalentsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Foreign earnings in the form of cash and cash equivalents [Member]",
"label": "Foreign earnings in the form of cash and cash equivalents [Member]",
"terseLabel": "Earnings in the form of cash and cash equivalents"
}
}
},
"localname": "ForeignearningsintheformofcashandcashequivalentsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ForeignearningstypeAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Foreign earnings type [Axis]",
"label": "Foreign earnings type [Axis]",
"terseLabel": "Foreign earnings type [Axis]"
}
}
},
"localname": "ForeignearningstypeAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_ForeignearningstypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Domain] for Foreign earnings type [Axis]",
"label": "Foreign earnings type [Domain]",
"terseLabel": "Foreign earnings type [Domain]"
}
}
},
"localname": "ForeignearningstypeDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_FumapharmAgMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Fumapharm AG [Member]",
"label": "Fumapharm AG [Member]",
"terseLabel": "Fumapharm AG"
}
}
},
"localname": "FumapharmAgMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_FumarateMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Fumarate [Member]",
"label": "Fumarate [Member]",
"terseLabel": "Fumarate"
}
}
},
"localname": "FumarateMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_FutureContingentMilestoneTypesAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Future Contingent Milestone Types [Axis]",
"label": "Future Contingent Milestone Types [Axis]",
"terseLabel": "Future Contingent Milestone Types [Axis]"
}
}
},
"localname": "FutureContingentMilestoneTypesAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "stringItemType"
},
"biib_FutureContingentMilestoneTypesDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Domain] for Future Contingent Milestone Types [Axis]",
"label": "Future Contingent Milestone Types [Domain]",
"terseLabel": "Future Contingent Milestone Types [Domain]"
}
}
},
"localname": "FutureContingentMilestoneTypesDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_FutureContingentPaymentForAnnualWorldwideNetSalesThatExceed20Billion": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Future contingent payment for annual worldwide net sales that exceed $2.0 billion",
"label": "Future contingent payment for annual worldwide net sales that exceed $2.0 billion",
"terseLabel": "Future contingent payment for annual worldwide net sales that exceed $2.0 billion"
}
}
},
"localname": "FutureContingentPaymentForAnnualWorldwideNetSalesThatExceed20Billion",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_FutureContingentPaymentForAnnualWorldwideNetSalesUpTo20Billion": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Future contingent payment for annual worldwide net sales up to $2.0 billion",
"label": "Future contingent payment for annual worldwide net sales up to $2.0 billion",
"terseLabel": "Future contingent payment for annual worldwide net sales up to $2.0 billion"
}
}
},
"localname": "FutureContingentPaymentForAnnualWorldwideNetSalesUpTo20Billion",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_FutureContingentPaymentThreshold": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Future contingent payment threshold",
"label": "Future contingent payment threshold",
"terseLabel": "Future contingent payment threshold"
}
}
},
"localname": "FutureContingentPaymentThreshold",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_FutureRoyaltiesPercentageToBeReceivedOnSaleOfProduct": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Future royalties percentage to be received on sale of product.",
"label": "Future Royalties Percentage To Be Received On Sale Of Product",
"terseLabel": "Royalty percentage to be received"
}
}
},
"localname": "FutureRoyaltiesPercentageToBeReceivedOnSaleOfProduct",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails"
],
"xbrltype": "percentItemType"
},
"biib_Futurepercentageofcopromotionoperatingprofits": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Future percentage of co-promotion operating profits",
"label": "Future percentage of co-promotion operating profits",
"terseLabel": "Future percentage of co-promotion operating profits"
}
}
},
"localname": "Futurepercentageofcopromotionoperatingprofits",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "percentItemType"
},
"biib_GAZYVAMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "GAZYVA [Member]",
"label": "GAZYVA [Member]",
"terseLabel": "GAZYVA"
}
}
},
"localname": "GAZYVAMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "domainItemType"
},
"biib_GlobalLicensingCollaborationAgreementAmountSharesPurchased": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Global Licensing Collaboration Agreement, Amount, Shares Purchased",
"label": "Global Licensing Collaboration Agreement, Amount, Shares Purchased",
"terseLabel": "Global licensing collaboration, shares purchased, amount"
}
}
},
"localname": "GlobalLicensingCollaborationAgreementAmountSharesPurchased",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_GlobalLicensingCollaborationAgreementPaymentAchievementOfFirstCommercialSaleMilestones": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Global Licensing Collaboration Agreement, Payment, Achievement of First Commercial Sale Milestones",
"label": "Global Licensing Collaboration Agreement, Payment, Achievement of First Commercial Sale Milestones",
"terseLabel": "Global licensing collaboration, payment,first milestone"
}
}
},
"localname": "GlobalLicensingCollaborationAgreementPaymentAchievementOfFirstCommercialSaleMilestones",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_GlobalLicensingCollaborationAgreementPaymentAchievementOfSpecificSalesBasedMilestones": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Global Licensing Collaboration Agreement, Payment, Achievement of Specific Sales Based Milestones",
"label": "Global Licensing Collaboration Agreement, Payment, Achievement of Specific Sales Based Milestones",
"terseLabel": "Global licensing collaboration, payment, sales based milestone"
}
}
},
"localname": "GlobalLicensingCollaborationAgreementPaymentAchievementOfSpecificSalesBasedMilestones",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_GlobalLicensingCollaborationAgreementPaymentSelectionOfTargets": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Global Licensing Collaboration Agreement, Payment, Selection of Targets",
"label": "Global Licensing Collaboration Agreement, Payment, Selection of Targets",
"terseLabel": "Global licensing collaboration, payment, selection of targets"
}
}
},
"localname": "GlobalLicensingCollaborationAgreementPaymentSelectionOfTargets",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_GlobalLicensingCollaborationAgreementPurchasePricePerShare": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Global Licensing Collaboration Agreement, Purchase Price Per Share",
"label": "Global Licensing Collaboration Agreement, Purchase Price Per Share",
"terseLabel": "Global licensing collaboration, purchase price per share (in dollars per share)"
}
}
},
"localname": "GlobalLicensingCollaborationAgreementPurchasePricePerShare",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "perShareItemType"
},
"biib_GlobalLicensingCollaborationAgreementResearchDevelopmentRegulatoryAndCommercialMilestonePayments": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Global Licensing Collaboration Agreement, Research, Development, Regulatory and Commercial Milestone Payments",
"label": "Global Licensing Collaboration Agreement, Research, Development, Regulatory and Commercial Milestone Payments",
"terseLabel": "Global licensing collaboration, development and commercial milestone payments"
}
}
},
"localname": "GlobalLicensingCollaborationAgreementResearchDevelopmentRegulatoryAndCommercialMilestonePayments",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_GlobalLicensingCollaborationAgreementSharesPurchased": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Global Licensing Collaboration Agreement, Shares, Purchased",
"label": "Global Licensing Collaboration Agreement, Shares, Purchased",
"terseLabel": "Global licensing collaboration, shares purchased (in shares)"
}
}
},
"localname": "GlobalLicensingCollaborationAgreementSharesPurchased",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "sharesItemType"
},
"biib_GovernmentSecuritiesCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Government securities Current.",
"label": "Government Securities Current [Member]",
"verboseLabel": "Government securities Current"
}
}
},
"localname": "GovernmentSecuritiesCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_GovernmentSecuritiesNonCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Government Securities Non Current.",
"label": "Government Securities Non-current [Member]",
"verboseLabel": "Government securities Non-current"
}
}
},
"localname": "GovernmentSecuritiesNonCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_IMRALDIMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "IMRALDI [Member]",
"label": "IMRALDI [Member]",
"terseLabel": "IMRALDI"
}
}
},
"localname": "IMRALDIMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_IPerianMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "iPerian [Member]",
"label": "iPerian [Member]",
"terseLabel": "iPerian"
}
}
},
"localname": "IPerianMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails"
],
"xbrltype": "domainItemType"
},
"biib_IncomeLossFromEquityMethodInvestmentsNetOfTax": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 3.0,
"parentTag": "us-gaap_ProfitLoss",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Income (Loss) from Equity Method Investments, Net of Tax",
"label": "Income (Loss) from Equity Method Investments, Net of Tax",
"negatedLabel": "Equity in (income) loss of investee, net of tax"
}
}
},
"localname": "IncomeLossFromEquityMethodInvestmentsNetOfTax",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"biib_IncomeTaxExpenseReductionInDeferredTaxAssetsAndLiabilitiesNet": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Income Tax Expense, Reduction in Deferred Tax Assets and Liabilities, Net",
"label": "Income Tax Expense, Reduction in Deferred Tax Assets and Liabilities, Net",
"terseLabel": "Income tax expense, reduction in deferred tax assets and liabilities, net"
}
}
},
"localname": "IncomeTaxExpenseReductionInDeferredTaxAssetsAndLiabilitiesNet",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_IntangibleAssetsExcludingGoodwillTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Intangible assets excluding goodwill.",
"label": "Intangible Assets Excluding Goodwill [Table Text Block]",
"verboseLabel": "Intangible assets"
}
}
},
"localname": "IntangibleAssetsExcludingGoodwillTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillTables"
],
"xbrltype": "textBlockItemType"
},
"biib_InterestInSubsidiary": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Interest in subsidiary.",
"label": "Interest In Subsidiary",
"terseLabel": "Interest in subsidiary (less than given percentage)"
}
}
},
"localname": "InterestInSubsidiary",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_InterferonMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Interferon",
"label": "Interferon [Member]",
"terseLabel": "Interferon"
}
}
},
"localname": "InterferonMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_Inventorynetcurrentandnoncurrent": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Inventory, net current and noncurrent",
"label": "Inventory, net current and noncurrent",
"totalLabel": "Total inventory"
}
}
},
"localname": "Inventorynetcurrentandnoncurrent",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_InventoryrawmaterialssoldtoFUJIFILM": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Inventory raw materials sold to FUJIFILM",
"label": "Inventory raw materials sold to FUJIFILM",
"terseLabel": "Inventory raw materials sold to FUJIFILM"
}
}
},
"localname": "InventoryrawmaterialssoldtoFUJIFILM",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails",
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_InvestmentInBiotechnologyCompaniesThatAreDeterminedToBeUnconsolidatedVariableInterestEntities": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Investment in biotechnology companies that are determined to be unconsolidated variable interest entities.",
"label": "Investment in biotechnology companies that are determined to be unconsolidated variable interest entities",
"verboseLabel": "Investment in biotechnology companies that are determined to be unconsolidated variable interest entities"
}
}
},
"localname": "InvestmentInBiotechnologyCompaniesThatAreDeterminedToBeUnconsolidatedVariableInterestEntities",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_InvestmentInCommonStockSharesPurchased": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Investment In Common Stock, Shares Purchased",
"label": "Investment In Common Stock, Shares Purchased",
"terseLabel": "Investment in common stock, shares purchased (in shares)"
}
}
},
"localname": "InvestmentInCommonStockSharesPurchased",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"biib_Investmentincommonstockvalue": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Investment in common stock, value",
"label": "Investment in common stock, value",
"terseLabel": "Investment in common stock, value"
}
}
},
"localname": "Investmentincommonstockvalue",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_InvestmentsByThirdPartyInJointVentureAsPerAgreement": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Investments by third party in joint venture as per agreement",
"label": "Investments By Third Party In Joint Venture As Per Agreement.",
"terseLabel": "Contributions by third party"
}
}
},
"localname": "InvestmentsByThirdPartyInJointVentureAsPerAgreement",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_InvestmentsInVariableInterestEntitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Investments In Variable Interest Entities.",
"label": "Investments in Variable Interest Entities [Abstract]",
"terseLabel": "Investments in Variable Interest Entities [Abstract]"
}
}
},
"localname": "InvestmentsInVariableInterestEntitiesAbstract",
"nsuri": "http://www.biogenidec.com/20201231",
"xbrltype": "stringItemType"
},
"biib_IonisPharmaceuticalsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Ionis Pharmaceuticals [Member]",
"label": "Ionis Pharmaceuticals [Member]",
"terseLabel": "Ionis Pharmaceuticals"
}
}
},
"localname": "IonisPharmaceuticalsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_JointVentureOwnerShipPercentageByThirdParty": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Joint venture owner ship percentage by third party.",
"label": "Joint Venture OwnerShip Percentage By Third Party.",
"terseLabel": "Joint venture ownership percentage"
}
}
},
"localname": "JointVentureOwnerShipPercentageByThirdParty",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "percentItemType"
},
"biib_KaryopharmMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Karyopharm [Member]",
"label": "Karyopharm [Member]",
"terseLabel": "Karyopharm"
}
}
},
"localname": "KaryopharmMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_LesseeLeaseTableTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Lessee Lease Table [Table Text Block]",
"label": "Lessee Lease Table [Table Text Block]",
"terseLabel": "Schedule of operating leases"
}
}
},
"localname": "LesseeLeaseTableTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/LeasesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_LicenseFee": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "License Fee",
"label": "License Fee",
"terseLabel": "License fee"
}
}
},
"localname": "LicenseFee",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_LongtermoperatingleaseliabilitiesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Long-term operating lease liabilities [Member]",
"label": "Long-term operating lease liabilities [Member]",
"terseLabel": "Long-term operating lease liabilities"
}
}
},
"localname": "LongtermoperatingleaseliabilitiesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "domainItemType"
},
"biib_MSProductRevenuesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "MS Product Revenues [Member]",
"label": "MS Product Revenues [Member]",
"terseLabel": "Subtotal: MS"
}
}
},
"localname": "MSProductRevenuesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_MajorDistributorAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Major Distributor.",
"label": "Major Distributor [Axis]",
"terseLabel": "Major Distributor [Axis]"
}
}
},
"localname": "MajorDistributorAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_MajorDistributorDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Major Distributor.",
"label": "Major Distributor [Domain]",
"terseLabel": "Major Distributor [Domain]"
}
}
},
"localname": "MajorDistributorDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_MarketStockUnitsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Market stock units.",
"label": "Market Stock Units [Member]",
"verboseLabel": "Market stock units"
}
}
},
"localname": "MarketStockUnitsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_MaximumTargetNumberOfMarketStockUnitsGrantedBasedOnStockPerformanceInPercent": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Maximum target number of market stock units granted based on stock performance in percent.",
"label": "Maximum Target Number Of Market Stock Units Granted Based On Stock Performance In Percent",
"verboseLabel": "Percentage of earnings of the target number of units granted based on actual stock performance"
}
}
},
"localname": "MaximumTargetNumberOfMarketStockUnitsGrantedBasedOnStockPerformanceInPercent",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_MinimumQualifyingAgeForEmployeeBenefitPlan": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Minimum qualifying age for employee benefit plan.",
"label": "Minimum Qualifying Age For Employee Benefit Plan",
"terseLabel": "Minimum qualifying age for employee benefit plan"
}
}
},
"localname": "MinimumQualifyingAgeForEmployeeBenefitPlan",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"biib_MinimumTargetNumberOfMarketStockUnitsGrantedBasedOnStockPerformanceInPercent": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Minimum target number of market stock units granted based on stock performance in percent.",
"label": "Minimum Target Number Of Market Stock Units Granted Based On Stock Performance In Percent",
"verboseLabel": "Percentage of earnings of the target number of units granted based on actual stock performance"
}
}
},
"localname": "MinimumTargetNumberOfMarketStockUnitsGrantedBasedOnStockPerformanceInPercent",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_MortgageAndOtherAssetBackedSecuritiesCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Mortgage and other asset backed securities Current.",
"label": "Mortgage And Other Asset Backed Securities Current [Member]",
"verboseLabel": "Mortgage and other asset backed securities Current"
}
}
},
"localname": "MortgageAndOtherAssetBackedSecuritiesCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_MortgageAndOtherAssetBackedSecuritiesNonCurrentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Mortgage And Other Asset Backed Securities Non Current.",
"label": "Mortgage And Other Asset Backed Securities Non Current [Member]",
"verboseLabel": "Mortgage and other asset backed securities Non-current"
}
}
},
"localname": "MortgageAndOtherAssetBackedSecuritiesNonCurrentMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_NeurimmuneMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Neurimmune.",
"label": "Neurimmune [Member]",
"terseLabel": "Neurimmune",
"verboseLabel": "Neurimmune"
}
}
},
"localname": "NeurimmuneMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_NewAntiCd20Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "New Anti-CD20.",
"label": "New Anti Cd20 [Member]",
"terseLabel": "New Anti Cd20"
}
}
},
"localname": "NewAntiCd20Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "domainItemType"
},
"biib_NightstarMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Nightstar [Member]",
"label": "Nightstar [Member]",
"terseLabel": "Nightstar"
}
}
},
"localname": "NightstarMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_Noncontrollinginterestincreasedecreaseother": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Noncontrolling interest, increase (decrease) other",
"label": "Noncontrolling interest, increase (decrease) other",
"terseLabel": "Capital contribution from noncontrolling interest"
}
}
},
"localname": "Noncontrollinginterestincreasedecreaseother",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"biib_NumberOfDaysForCalculationOfAverageClosingStockPrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of days for calculation of average closing stock price.",
"label": "Number Of Days For Calculation Of Average Closing Stock Price",
"verboseLabel": "Number of days for calculation of average closing stock price"
}
}
},
"localname": "NumberOfDaysForCalculationOfAverageClosingStockPrice",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"biib_NumberOfEqualAnnualIncrements": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of equal annual increments.",
"label": "Number Of Equal Annual Increments",
"verboseLabel": "Number of equal annual increments"
}
}
},
"localname": "NumberOfEqualAnnualIncrements",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"biib_NumberOfOphthalmolgyBiosimilarProductsExclusiveRights": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of Ophthalmolgy Biosimilar Products, Exclusive Rights",
"label": "Number of Ophthalmolgy Biosimilar Products, Exclusive Rights",
"terseLabel": "Number of ophthalmolgy biosimilar products"
}
}
},
"localname": "NumberOfOphthalmolgyBiosimilarProductsExclusiveRights",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"biib_NumberOfProductCandidates": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of Product Candidates",
"label": "Number of Product Candidates",
"terseLabel": "Number of product candidates"
}
}
},
"localname": "NumberOfProductCandidates",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails"
],
"xbrltype": "integerItemType"
},
"biib_NumberOfShareBasedCompensationPlansPursuantToWhichAwardsAreCurrentlyBeingMade": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of share-based compensation plans pursuant to which awards are currently being made.",
"label": "Number Of Share Based Compensation Plans Pursuant To Which Awards Are Currently Being Made",
"verboseLabel": "Number of share-based compensation plans pursuant to which awards are currently being made"
}
}
},
"localname": "NumberOfShareBasedCompensationPlansPursuantToWhichAwardsAreCurrentlyBeingMade",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"biib_NumberOfSharesOfCommonStockForIssuance": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Total number of shares of common stock for issuance.",
"label": "Number Of Shares Of Common Stock For Issuance",
"verboseLabel": "Total number of shares of common stock for issuance (in shares)"
}
}
},
"localname": "NumberOfSharesOfCommonStockForIssuance",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"biib_NumberOfSquareFeet": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of Square Feet",
"label": "Number of Square Feet",
"terseLabel": "Number of square feet"
}
}
},
"localname": "NumberOfSquareFeet",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "areaItemType"
},
"biib_NumberOfWholesalers": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of Wholesalers",
"label": "Number of Wholesalers",
"terseLabel": "Number of wholesalers"
}
}
},
"localname": "NumberOfWholesalers",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"biib_NumeratorAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Numerator.",
"label": "Numerator [Abstract]",
"verboseLabel": "Numerator:"
}
}
},
"localname": "NumeratorAbstract",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "stringItemType"
},
"biib_OCREVUSMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "OCREVUS",
"label": "OCREVUS [Member]",
"terseLabel": "OCREVUS"
}
}
},
"localname": "OCREVUSMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails"
],
"xbrltype": "domainItemType"
},
"biib_OperatingleaseassetsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Operating lease assets [Member]",
"label": "Operating lease assets [Member]",
"terseLabel": "Operating lease assets"
}
}
},
"localname": "OperatingleaseassetsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "domainItemType"
},
"biib_OperatingleasesupplementalcashflowdisclosureTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Operating lease supplemental cash flow disclosure [Table Text Block]",
"label": "Operating lease supplemental cash flow disclosure [Table Text Block]",
"terseLabel": "Operating lease supplemental cash flow disclosure"
}
}
},
"localname": "OperatingleasesupplementalcashflowdisclosureTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/LeasesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_OperatingleaseweightedaverageremainingtermanddiscountrateTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Operating lease weighted average remaining term and discount rate [Table Text Block]",
"label": "Operating lease weighted average remaining term and discount rate [Table Text Block]",
"terseLabel": "Operating lease weighted average remaining term and discount rate"
}
}
},
"localname": "OperatingleaseweightedaverageremainingtermanddiscountrateTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/LeasesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_OtherForeignEarningsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Other Foreign Earnings [Member]",
"label": "Other Foreign Earnings [Member]",
"terseLabel": "Other Foreign Earnings"
}
}
},
"localname": "OtherForeignEarningsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_OtherRelationshipsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Other Relationships",
"label": "Other Relationships [Member]",
"terseLabel": "Other Relationships"
}
}
},
"localname": "OtherRelationshipsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"biib_OthercorporaterevenuesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Other corporate revenues [Member]",
"label": "Other corporate revenues [Member]",
"terseLabel": "Other corporate"
}
}
},
"localname": "OthercorporaterevenuesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"biib_OtherresearchanddiscoveryMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Other research and discovery [Member]",
"label": "Other research and discovery [Member]",
"terseLabel": "Other research and discovery"
}
}
},
"localname": "OtherresearchanddiscoveryMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails"
],
"xbrltype": "domainItemType"
},
"biib_OtherrevenuesTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Other revenues [Table Text Block]",
"label": "Other revenues [Table Text Block]",
"terseLabel": "Other revenues"
}
}
},
"localname": "OtherrevenuesTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_OtherrevenuesfromantiCD20therapeuticprograms": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails": {
"order": 2.0,
"parentTag": "us-gaap_Revenues",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Other revenues from anti-CD20 therapeutic programs consists of revenue on sales of RITUXAN outside the U.S., which consist of our share of pre-tax co-promotion profits in Canada and royalty revenue on sales of Ocrevus.",
"label": "Other revenues from anti-CD20 therapeutic programs",
"terseLabel": "Other revenues from anti-CD20 therapeutic programs",
"verboseLabel": "Other revenues from anti-CD20 therapeutic programs"
}
}
},
"localname": "OtherrevenuesfromantiCD20therapeuticprograms",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_PaymentTermsOfAccountsReceivable": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Payment terms of accounts receivable.",
"label": "Payment Terms Of Accounts Receivable",
"verboseLabel": "Payment terms of accounts receivable arising from product sales"
}
}
},
"localname": "PaymentTermsOfAccountsReceivable",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"biib_PaymentsReturnsAppliedAgainstProductRevenueReservesRelatingToCurrentYear": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Payments/ returns applied against product revenue reserves relating to current year.",
"label": "Payments Returns Applied Against Product Revenue Reserves Relating To Current Year",
"negatedTerseLabel": "Payments/returns relating to sales in current year"
}
}
},
"localname": "PaymentsReturnsAppliedAgainstProductRevenueReservesRelatingToCurrentYear",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_PaymentsReturnsAppliedAgainstProductRevenueReservesRelatingToPriorYear": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Payments/ returns applied against product revenue reserves relating to prior year.",
"label": "Payments Returns Applied Against Product Revenue Reserves Relating To Prior Year",
"negatedTerseLabel": "Payments/returns relating to sales in prior years"
}
}
},
"localname": "PaymentsReturnsAppliedAgainstProductRevenueReservesRelatingToPriorYear",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_PercentageOfCoPromotionOperatingProfitsFirstFiftyMillion": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of co promotion operating profits first fifty million.",
"label": "Percentage Of Co Promotion Operating Profits First Fifty Million",
"terseLabel": "Percentage of co promotion operating profits first fifty million"
}
}
},
"localname": "PercentageOfCoPromotionOperatingProfitsFirstFiftyMillion",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionOne": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of co promotion operating profits greater than first fifty million option one.",
"label": "Percentage Of Co Promotion Operating Profits Greater Than First Fifty Million Option One",
"verboseLabel": "Until GAZYVA First Non-CLL FDA Approval"
}
}
},
"localname": "PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionOne",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of co promotion operating profits greater than first fifty million option two.",
"label": "Percentage Of Co Promotion Operating Profits Greater Than First Fifty Million Option Two [Abstract]",
"verboseLabel": "After First GA101 Threshold Date"
}
}
},
"localname": "PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoAbstract",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails"
],
"xbrltype": "stringItemType"
},
"biib_PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionOne": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of co promotion operating profits greater than first fifty million option two sub option one.",
"label": "Percentage Of Co Promotion Operating Profits Greater Than First Fifty Million Option Two Sub Option One",
"terseLabel": "After GAZYVA First Non-CLL FDA Approval until First GAZYVA Threshold Date",
"verboseLabel": "Until First GAZYVA Threshold Date"
}
}
},
"localname": "PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionOne",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionThree": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of co promotion operating profits greater than first fifty million option two sub option three.",
"label": "Percentage Of Co Promotion Operating Profits Greater Than First Fifty Million Option Two Sub Option Three",
"terseLabel": "After Second GAZYVA Threshold Date",
"verboseLabel": "After Second GAZYVA Threshold Date"
}
}
},
"localname": "PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionThree",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionTwo": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of co promotion operating profits greater than first fifty million option two sub option two.",
"label": "Percentage Of Co Promotion Operating Profits Greater Than First Fifty Million Option Two Sub Option Two",
"terseLabel": "After First GAZYVA Threshold Date until Second GAZYVA Threshold Date",
"verboseLabel": "After First GAZYVA Threshold Date until Second GAZYVA Threshold Date"
}
}
},
"localname": "PercentageOfCoPromotionOperatingProfitsGreaterThanFirstFiftyMillionOptionTwoSubOptionTwo",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageOfDevelopmentCostsIncurred": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of Development Costs Incurred",
"label": "Percentage of Development Costs Incurred",
"terseLabel": "Percentage of development costs incurred"
}
}
},
"localname": "PercentageOfDevelopmentCostsIncurred",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageOfMinimumInvestmentReturn": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of minimum investment return.",
"label": "Percentage Of Minimum Investment Return",
"verboseLabel": "Percentage of minimum investment return"
}
}
},
"localname": "PercentageOfMinimumInvestmentReturn",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageOfRoyaltiesAsPerCollaboration": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of royalties as per collaboration.",
"label": "Percentage Of Royalties As Per Collaboration",
"terseLabel": "Percentage of royalties as per collaboration"
}
}
},
"localname": "PercentageOfRoyaltiesAsPerCollaboration",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageParValueOfSeniorNotes": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage Par Value Of Senior Notes",
"label": "Percentage Par Value Of Senior Notes",
"verboseLabel": "Redemption percentage par value of senior notes"
}
}
},
"localname": "PercentageParValueOfSeniorNotes",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageReceivablesOfWholesaleDistributorAccountedInConsolidatedReceivables": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage receivables of wholesale distributor accounted in consolidated receivables.",
"label": "Percentage Receivables Of Wholesale Distributor Accounted In Consolidated Receivables",
"terseLabel": "Percentage receivables of wholesale distributor accounted in consolidated receivables"
}
}
},
"localname": "PercentageReceivablesOfWholesaleDistributorAccountedInConsolidatedReceivables",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_PercentageoffuturedevelopmentcostsrelatedtoEisai": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of future development costs related to Eisai",
"label": "Percentage of future development costs related to Eisai",
"terseLabel": "Percentage of future development costs related to Eisai"
}
}
},
"localname": "PercentageoffuturedevelopmentcostsrelatedtoEisai",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_PerformanceStockUnitsSettledinCashMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Performance Stock Units Settled in Cash [Member]",
"label": "Performance Stock Units Settled in Cash [Member]",
"terseLabel": "Performance stock units settled in cash"
}
}
},
"localname": "PerformanceStockUnitsSettledinCashMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_PerformanceStockUnitsSettledinCashTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Performance Stock Units Settled in Cash [Table Text Block]",
"label": "Performance Stock Units Settled in Cash [Table Text Block]",
"terseLabel": "Performance stock units settled in cash activity"
}
}
},
"localname": "PerformanceStockUnitsSettledinCashTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_PerformanceStockUnitsSettledinStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Performance Stock Units Settled in Stock [Member]",
"label": "Performance Stock Units Settled in Stock [Member]",
"terseLabel": "Performance stock units settled in stock"
}
}
},
"localname": "PerformanceStockUnitsSettledinStockMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_PerformanceStockUnitsSettledinStockTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Performance Stock Units Settled in Stock [Table Text Block]",
"label": "Performance Stock Units Settled in Stock [Table Text Block]",
"terseLabel": "Performance stock units settled in stock activity"
}
}
},
"localname": "PerformanceStockUnitsSettledinStockTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_PerformanceUnitsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Performance Units [Member]",
"label": "Performance Units [Member]",
"terseLabel": "Performance units"
}
}
},
"localname": "PerformanceUnitsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_PerformanceunitsactivityTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Performance units activity [Table Text Block]",
"label": "Performance units activity [Table Text Block]",
"terseLabel": "Performance units activity"
}
}
},
"localname": "PerformanceunitsactivityTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_PeriodOfCollaborationAgreement": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Period Of Collaboration Agreement",
"label": "Period Of Collaboration Agreement",
"terseLabel": "Collaboration agreement term"
}
}
},
"localname": "PeriodOfCollaborationAgreement",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails"
],
"xbrltype": "durationItemType"
},
"biib_PfizerMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Pfizer [Member]",
"label": "Pfizer [Member]",
"terseLabel": "Pfizer"
}
}
},
"localname": "PfizerMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_PlanAssetsForDeferredCompensationFairValueDisclosure": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair values as of the balance sheet date of all assets of deferred compensation plans.",
"label": "Plan Assets For Deferred Compensation Fair Value Disclosure",
"verboseLabel": "Plan assets for deferred compensation"
}
}
},
"localname": "PlanAssetsForDeferredCompensationFairValueDisclosure",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_PostacquisitionequitycompensationMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Post-acquisition equity compensation [Member]",
"label": "Post-acquisition equity compensation [Member]",
"terseLabel": "Post-acquisition equity compensation"
}
}
},
"localname": "PostacquisitionequitycompensationMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_PotentialFutureMilestonePaymentsCommitmentToThirdPartyApproximately": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Potential future milestone payments commitment to third party approximately.",
"label": "Potential Future Milestone Payments Commitment To Third Party Approximately",
"terseLabel": "Potential future milestone payments commitment to third party approximately",
"verboseLabel": "Potential future milestone payments commitment, approximately"
}
}
},
"localname": "PotentialFutureMilestonePaymentsCommitmentToThirdPartyApproximately",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_PreTaxChargePaymentOfEarlyCallPremiumAndWriteOffOfRemainingUnamortizedDebtIssuanceCostsNetOfDerivativeGain": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Pre-tax Charge, Payment of Early Call Premium and Write off of Remaining Unamortized Debt Issuance Costs, Net of Derivative Gain",
"label": "Pre-tax Charge, Payment of Early Call Premium and Write off of Remaining Unamortized Debt Issuance Costs, Net of Derivative Gain",
"terseLabel": "Pre-tax charge"
}
}
},
"localname": "PreTaxChargePaymentOfEarlyCallPremiumAndWriteOffOfRemainingUnamortizedDebtIssuanceCostsNetOfDerivativeGain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Premiumonpurchaseofcommonstock": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Premium on purchase of common stock",
"label": "Premium on purchase of common stock",
"terseLabel": "Premium on purchase of common stock"
}
}
},
"localname": "Premiumonpurchaseofcommonstock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_PretaxProfitSharingFormulaTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Pretax Profit Sharing Formula [Table Text Block]",
"label": "Pretax Profit Sharing Formula [Table Text Block]",
"terseLabel": "Pretax profit sharing formula"
}
}
},
"localname": "PretaxProfitSharingFormulaTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_PurchaseCommitmentReductionOfAmountCommitted": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Purchase Commitment, Reduction of Amount Committed",
"label": "Purchase Commitment, Reduction of Amount Committed",
"terseLabel": "Purchase commitment, reduction"
}
}
},
"localname": "PurchaseCommitmentReductionOfAmountCommitted",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Purchaseofcommonstock": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Purchase of common stock",
"label": "Purchase of common stock",
"terseLabel": "Purchase of common stock"
}
}
},
"localname": "Purchaseofcommonstock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_RatioOfNumberOfSharesReservedUnderPlan": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Ratio of total number of shares reserved under the plan.",
"label": "Ratio Of Number Of Shares Reserved Under Plan",
"verboseLabel": "Ratio of total number of shares reserved under the plan"
}
}
},
"localname": "RatioOfNumberOfSharesReservedUnderPlan",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "pureItemType"
},
"biib_Reductionindeferredtaxliabilityundistributedforeignearnings": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Reduction in deferred tax liability, undistributed foreign earnings",
"label": "Reduction in deferred tax liability, undistributed foreign earnings",
"terseLabel": "Reduction in deferred tax liability, undistributed foreign earnings"
}
}
},
"localname": "Reductionindeferredtaxliabilityundistributedforeignearnings",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Reductioninroyaltyrate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Reduction in royalty rate",
"label": "Reduction in royalty rate",
"terseLabel": "Reduction in royalty rate"
}
}
},
"localname": "Reductioninroyaltyrate",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails"
],
"xbrltype": "percentItemType"
},
"biib_Reductioninroyaltyratepayableoncommercialsales": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Reduction in royalty rate payable on commercial sales",
"label": "Reduction in royalty rate payable on commercial sales",
"terseLabel": "Reduction in royalty rate payable on commercial sales"
}
}
},
"localname": "Reductioninroyaltyratepayableoncommercialsales",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "percentItemType"
},
"biib_RegulatoryMilestonesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Regulatory Milestones [Member]",
"label": "Regulatory Milestones [Member]",
"terseLabel": "Regulatory Milestones"
}
}
},
"localname": "RegulatoryMilestonesMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_ResearchAndDevelopmentCostsPercentage": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Research and Development Costs, Percentage",
"label": "Research and Development Costs, Percentage",
"terseLabel": "Research and development costs, percentage"
}
}
},
"localname": "ResearchAndDevelopmentCostsPercentage",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "percentItemType"
},
"biib_ResearchanddevelopmentassetAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Research and development asset [Axis]",
"label": "Research and development asset [Axis]",
"terseLabel": "Research and development asset [Axis]"
}
}
},
"localname": "ResearchanddevelopmentassetAxis",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"biib_ResearchanddevelopmentassetDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Domain] for Research and development asset [Axis]",
"label": "Research and development asset [Domain]",
"terseLabel": "Research and development asset [Domain]"
}
}
},
"localname": "ResearchanddevelopmentassetDomain",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ReserveforCashDiscountsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Reserve for Cash Discounts [Member]",
"label": "Reserve for Cash Discounts [Member]",
"terseLabel": "Discounts"
}
}
},
"localname": "ReserveforCashDiscountsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "domainItemType"
},
"biib_ResidualU.S.taxliabilityonreversalofdeferredtaxliabilitiesonforeignsubsidiariespurchaseaccounting": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Residual U.S. tax liability on reversal of deferred tax liabilities on foreign subsidiaries purchase accounting",
"label": "Residual U.S. tax liability on reversal of deferred tax liabilities on foreign subsidiaries purchase accounting",
"terseLabel": "Residual U.S. tax liability on reversal of deferred tax liabilities on foreign subsidiaries purchase accounting"
}
}
},
"localname": "ResidualU.S.taxliabilityonreversalofdeferredtaxliabilitiesonforeignsubsidiariespurchaseaccounting",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_RevenuesInPercentageFromAntiCD20TherapeuticPrograms": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Revenues In Percentage From Anti-CD20 Therapeutic Programs",
"label": "Revenues In Percentage From Anti-CD20 Therapeutic Programs",
"terseLabel": "Percentage of total revenues"
}
}
},
"localname": "RevenuesInPercentageFromAntiCD20TherapeuticPrograms",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_RevenuesfromantiCD20therapeuticprogramsTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Revenues from anti-CD20 therapeutic programs",
"label": "Revenues from anti-CD20 therapeutic programs [Table Text Block]",
"terseLabel": "Revenues from anti-CD20 therapeutic programs",
"verboseLabel": "Revenues from anti-CD20 therapeutic programs"
}
}
},
"localname": "RevenuesfromantiCD20therapeuticprogramsTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsTables",
"http://www.biogenidec.com/role/RevenuesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_Revenuesfromanticd20therapeuticprogramsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Revenues from anti-cd20 therapeutic programs",
"label": "Revenues from anti-cd20 therapeutic programs [Member]",
"terseLabel": "Revenues from anti-CD20 therapeutic programs"
}
}
},
"localname": "Revenuesfromanticd20therapeuticprogramsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"biib_RituxanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "RITUXAN.",
"label": "RITUXAN [Member]",
"terseLabel": "RITUXAN"
}
}
},
"localname": "RituxanMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "domainItemType"
},
"biib_RocheGroupGenentechMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Roche group Genentech.",
"label": "Roche Group Genentech Member",
"terseLabel": "Genentech"
}
}
},
"localname": "RocheGroupGenentechMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails"
],
"xbrltype": "domainItemType"
},
"biib_RoyaltiesPayablePeriodPostFDAApproval": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Royalties Payable Period, Post FDA Approval",
"label": "Royalties Payable Period, Post FDA Approval",
"terseLabel": "Royalties payable period, post FDA approval"
}
}
},
"localname": "RoyaltiesPayablePeriodPostFDAApproval",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"biib_RoyaltyCostofSalesPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Royalty Cost of Sales [Policy Text Block]",
"label": "Royalty Cost of Sales [Policy Text Block]",
"terseLabel": "Royalty Cost of Sales"
}
}
},
"localname": "RoyaltyCostofSalesPolicyTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"biib_Royaltyoperatingprofitthresholdforhighestroyaltyratepercentage": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Royalty operating profit threshold for highest royalty rate percentage",
"label": "Royalty operating profit threshold for highest royalty rate percentage",
"terseLabel": "Royalty operating profit threshold for highest royalty rate percentage"
}
}
},
"localname": "Royaltyoperatingprofitthresholdforhighestroyaltyratepercentage",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_SOD1Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "SOD1 [Member]",
"label": "SOD1 [Member]",
"terseLabel": "SOD1"
}
}
},
"localname": "SOD1Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "domainItemType"
},
"biib_SPINRAZAMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "SPINRAZA [Member]",
"label": "SPINRAZA [Member]",
"terseLabel": "SPINRAZA"
}
}
},
"localname": "SPINRAZAMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_SageTherapeuticsIncMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Sage Therapeutics Inc.",
"label": "Sage Therapeutics Inc. [Member]",
"terseLabel": "Sage Therapeutics"
}
}
},
"localname": "SageTherapeuticsIncMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_SalesTriggerGrossSalesThreshold": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Sales trigger gross sales threshold.",
"label": "Sales Trigger Gross Sales Threshold",
"terseLabel": "Sales trigger gross sales threshold"
}
}
},
"localname": "SalesTriggerGrossSalesThreshold",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_SamsungBiosimilarAgreementMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Samsung bio-similar agreement.",
"label": "Samsung Biosimilar Agreement [Member]",
"terseLabel": "Samsung Bioepis"
}
}
},
"localname": "SamsungBiosimilarAgreementMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"biib_SangamoCommonStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Sangamo Common Stock",
"label": "Sangamo Common Stock [Member]",
"terseLabel": "Sangamo Therapeutics"
}
}
},
"localname": "SangamoCommonStockMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_SangamoTherapeuticsIncAgreementMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Sangamo Therapeutics, Inc. Agreement",
"label": "Sangamo Therapeutics, Inc. Agreement [Member]",
"terseLabel": "Sangamo Therapeutics, Inc. Agreement"
}
}
},
"localname": "SangamoTherapeuticsIncAgreementMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "domainItemType"
},
"biib_ScheduleOfCashSettledPerformanceSharesActivityTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule Of Cash Settled Performance Shares Activity.",
"label": "Schedule Of Cash Settled Performance Shares Activity [Table Text Block]",
"terseLabel": "Cash settled performance shares activity"
}
}
},
"localname": "ScheduleOfCashSettledPerformanceSharesActivityTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_ScheduleOfMarketStockUnitsActivityTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule Of Market Stock Units Activity.",
"label": "Schedule Of Market Stock Units Activity [Table Text Block]",
"terseLabel": "Market stock units activity"
}
}
},
"localname": "ScheduleOfMarketStockUnitsActivityTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_ScheduleOfPropertyPlantAndEquipmentUsefulLivesTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of property plant and equipment useful lives.",
"label": "Schedule Of Property Plant And Equipment Useful Lives [Table Text Block]",
"terseLabel": "Property, plant and equipment estimated useful lives"
}
}
},
"localname": "ScheduleOfPropertyPlantAndEquipmentUsefulLivesTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_SegmentGeographicalGroupsOfCountriesGroupOtherMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Segment geographical groups of countries group other.",
"label": "Segment Geographical Groups Of Countries Group Other [Member]",
"verboseLabel": "Other"
}
}
},
"localname": "SegmentGeographicalGroupsOfCountriesGroupOtherMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"biib_SeniorNotesFivePointTwoPercentDueTwentyFortyFiveMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Senior Notes Five Point Two Percent Due Twenty Forty Five [Member]",
"label": "Senior Notes Five Point Two Percent Due Twenty Forty Five [Member]",
"terseLabel": "5.200% Senior Notes due September 15, 2045"
}
}
},
"localname": "SeniorNotesFivePointTwoPercentDueTwentyFortyFiveMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"biib_SeniorNotesFourPointZeroFivePercentDueTwentyTwentyFiveMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Senior Notes Four Point Zero Five Percent Due Twenty Twenty Five [Member]",
"label": "Senior Notes Four Point Zero Five Percent Due Twenty Twenty Five [Member]",
"terseLabel": "4.050% Senior Notes due September 15, 2025"
}
}
},
"localname": "SeniorNotesFourPointZeroFivePercentDueTwentyTwentyFiveMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"biib_SeniorNotesThreePointSixTwoFivePercentDueTwentyTwentyTwoMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Senior Notes Three Point Six Two Five Percent Due Twenty Twenty Two [Member]",
"label": "Senior Notes Three Point Six Two Five Percent Due Twenty Twenty Two [Member]",
"terseLabel": "3.625% Senior Notes due September 15, 2022"
}
}
},
"localname": "SeniorNotesThreePointSixTwoFivePercentDueTwentyTwentyTwoMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"biib_SeniorNotesTwoPointNinePercentDueTwentyTwentyMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Senior Notes Two Point Nine Percent Due Twenty Twenty [Member]",
"label": "Senior Notes Two Point Nine Percent Due Twenty Twenty [Member]",
"terseLabel": "2.900% Senior Notes due September 15, 2020"
}
}
},
"localname": "SeniorNotesTwoPointNinePercentDueTwentyTwentyMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"biib_SeriesXPreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series X preferred stock.",
"label": "Series X Preferred Stock [Member]",
"terseLabel": "Series X junior participating"
}
}
},
"localname": "SeriesXPreferredStockMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ShareBasedCompensationExpenseIncludedInCostsAndExpenses": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Share based compensation expense included in costs and expenses.",
"label": "Share Based Compensation Expense Included In Costs And Expenses",
"terseLabel": "Share-based compensation expense included in total cost and expenses"
}
}
},
"localname": "ShareBasedCompensationExpenseIncludedInCostsAndExpenses",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ShareOfCoPromotionProfits": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails": {
"order": 1.0,
"parentTag": "us-gaap_Revenues",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Share of co promotion profits.",
"label": "Share Of Co Promotion Profits",
"terseLabel": "Biogen's share of pre-tax profits in the U.S. for RITUXAN and GAZYVA, including the reimbursement of selling and development expenses",
"verboseLabel": "Biogen's share of pre-tax profits in the U.S. for RITUXAN and GAZYVA"
}
}
},
"localname": "ShareOfCoPromotionProfits",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ShareRepurchaseProgram1Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Repurchase Program 1",
"label": "Share Repurchase Program 1 [Member]",
"terseLabel": "2016 Share Repurchase Program"
}
}
},
"localname": "ShareRepurchaseProgram1Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ShareRepurchaseProgram2Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Repurchase Program 2",
"label": "Share Repurchase Program 2 [Member]",
"terseLabel": "2018 Share Repurchase Program"
}
}
},
"localname": "ShareRepurchaseProgram2Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ShareRepurchaseProgram3Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Repurchase Program 3",
"label": "Share Repurchase Program 3 [Member]",
"terseLabel": "March 2019 Share Repurchase Program"
}
}
},
"localname": "ShareRepurchaseProgram3Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ShareRepurchaseProgram4Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Repurchase Program 4",
"label": "Share Repurchase Program 4 [Member]",
"terseLabel": "December 2019 Share Repurchase Program"
}
}
},
"localname": "ShareRepurchaseProgram4Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ShareRepurchaseProgram5Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Repurchase Program 5",
"label": "Share Repurchase Program 5 [Member]",
"terseLabel": "2020 Share Repurchase Program"
}
}
},
"localname": "ShareRepurchaseProgram5Member",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_SharesIssuedUnderEmployeeStockPurchasePlanTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Shares issued under employee stock purchase plan.",
"label": "Shares Issued Under Employee Stock Purchase Plan [Table Text Block]",
"terseLabel": "Shares issued under employee stock purchase plan"
}
}
},
"localname": "SharesIssuedUnderEmployeeStockPurchasePlanTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_ShorttermderivativeMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Short-term derivative [Member]",
"label": "Short-term derivative [Member]",
"terseLabel": "Short-term derivative"
}
}
},
"localname": "ShorttermderivativeMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_SolothurnMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Solothurn [Member]",
"label": "Solothurn [Member]",
"terseLabel": "Solothurn"
}
}
},
"localname": "SolothurnMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_StrategicInvestmentPortfolio": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Strategic investment portfolio.",
"label": "Strategic Investment Portfolio",
"verboseLabel": "Strategic investment portfolio"
}
}
},
"localname": "StrategicInvestmentPortfolio",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_StrategicInvestmentsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Strategic investments.",
"label": "Strategic Investments [Member]",
"terseLabel": "Strategic Investments"
}
}
},
"localname": "StrategicInvestmentsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_StromedixMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Stromedix.",
"label": "Stromedix [Member]",
"verboseLabel": "Stromedix, Inc."
}
}
},
"localname": "StromedixMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_SubleaseMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Sublease [Member]",
"label": "Sublease [Member]",
"terseLabel": "Sublease"
}
}
},
"localname": "SubleaseMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/LeasesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_SummaryOfActivityRelatedToCollaborationWithCompanyEightTableTextBlockTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Summary Of Activity Related To Collaboration With Company Eight [Table Text Block] [Table Text Block]",
"label": "Summary Of Activity Related To Collaboration With Company Eight [Table Text Block] [Table Text Block]",
"terseLabel": "Summary of activity related to BAN2401 and Elenbecestat collaboration"
}
}
},
"localname": "SummaryOfActivityRelatedToCollaborationWithCompanyEightTableTextBlockTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_SummaryOfActivityRelatedToTheUCBCollaborationTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Summary of Activity Related to the UCB Collaboration",
"label": "Summary of Activity Related to the UCB Collaboration [Table Text Block]",
"terseLabel": "Summary of activity related to the UCB collaboration"
}
}
},
"localname": "SummaryOfActivityRelatedToTheUCBCollaborationTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_SummaryOfCommonStockTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Summary of common stock.",
"label": "Summary Of Common Stock [Table Text Block]",
"terseLabel": "Summary of common stock"
}
}
},
"localname": "SummaryOfCommonStockTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EquityTables"
],
"xbrltype": "textBlockItemType"
},
"biib_SummaryOfProductRevenueReservesIncludedInConsolidatedBalanceSheetsTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Summary of total product revenue reserves included in consolidated balance sheets.",
"label": "Summary Of Product Revenue Reserves Included In Consolidated Balance Sheets [Table Text Block]",
"terseLabel": "Total reserves included in consolidated balance sheet"
}
}
},
"localname": "SummaryOfProductRevenueReservesIncludedInConsolidatedBalanceSheetsTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesTables"
],
"xbrltype": "textBlockItemType"
},
"biib_SummaryofActivityRelatedtoAducanumabCollaborationTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "[Table Text Block] for Summary of Activity Related to Aducanumab Collaboration [Table]",
"label": "Summary of Activity Related to Aducanumab Collaboration [Table Text Block]",
"terseLabel": "Summary of activity related to Aducanumab collaboration"
}
}
},
"localname": "SummaryofActivityRelatedtoAducanumabCollaborationTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_TECFIDERAMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "TECFIDERA",
"label": "TECFIDERA [Member]",
"terseLabel": "TECFIDERA"
}
}
},
"localname": "TECFIDERAMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_TGNMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "TGN",
"label": "TGN [Member]",
"terseLabel": "TGN"
}
}
},
"localname": "TGNMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_TechnologicalAndRegulatorySuccessProbability": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Technological and Regulatory Success, Probability",
"label": "Technological and Regulatory Success, Probability",
"terseLabel": "Technological and regulatory success, probability"
}
}
},
"localname": "TechnologicalAndRegulatorySuccessProbability",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_Termofcollaborationagreement": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Term of collaboration agreement",
"label": "Term of collaboration agreement",
"terseLabel": "Term of collaboration agreement"
}
}
},
"localname": "Termofcollaborationagreement",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "durationItemType"
},
"biib_ThresholdOfGrossSalesOfToBeAchievedInPrecedingTwelveConsecutiveMonthsUnderOptionOne": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Threshold of gross sales of to be achieved in preceding twelve consecutive months under option one.",
"label": "Threshold Of Gross Sales Of To Be Achieved In Preceding Twelve Consecutive Months Under Option One",
"verboseLabel": "Limit of gross sale of GAZYVA to be achieved in preceding 12 months under option one"
}
}
},
"localname": "ThresholdOfGrossSalesOfToBeAchievedInPrecedingTwelveConsecutiveMonthsUnderOptionOne",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_ThresholdOfGrossSalesToBeAchievedInAnyTwelveConsecutiveMonthsUnderOptionOne": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Threshold of gross sales to be achieved in any twelve consecutive months under option one.",
"label": "Threshold Of Gross Sales To Be Achieved In Any Twelve Consecutive Months Under Option One",
"verboseLabel": "Limit of gross sale of GAZYVA to be achieved in any 12 months under option one"
}
}
},
"localname": "ThresholdOfGrossSalesToBeAchievedInAnyTwelveConsecutiveMonthsUnderOptionOne",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_TimeVestedRestrictedStockUnitsActivityTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Time Vested Restricted Stock Units Activity.",
"label": "Time Vested Restricted Stock Units Activity [Table Text Block]",
"terseLabel": "Time-vested restricted stock units activity"
}
}
},
"localname": "TimeVestedRestrictedStockUnitsActivityTableTextBlock",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"biib_TimeVestedRestrictedStockUnitsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Time vested restricted stock units.",
"label": "Time Vested Restricted Stock Units [Member]",
"verboseLabel": "Time-vested restricted stock units"
}
}
},
"localname": "TimeVestedRestrictedStockUnitsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "domainItemType"
},
"biib_Totaldeferredchargesandprepaidtaxes": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Total deferred charges and prepaid taxes",
"label": "Total deferred charges and prepaid taxes",
"terseLabel": "Total deferred charges and prepaid taxes"
}
}
},
"localname": "Totaldeferredchargesandprepaidtaxes",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Totalpaymenttoentercollaborationagreement": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Total payment to enter collaboration agreement",
"label": "Total payment to enter collaboration agreement",
"terseLabel": "Total payment to enter collaboration agreement"
}
}
},
"localname": "Totalpaymenttoentercollaborationagreement",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_TransitionTollTaxRepatriationTaxRate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Transition Toll Tax Repatriation Tax Rate",
"label": "Transition Toll Tax Repatriation Tax Rate",
"terseLabel": "Transition toll tax repatriation tax rate"
}
}
},
"localname": "TransitionTollTaxRepatriationTaxRate",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"biib_TwelvemonthsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Twelve months [Member]",
"label": "Twelve months [Member]",
"terseLabel": "Twelve months"
}
}
},
"localname": "TwelvemonthsMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"biib_TwentybillionMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Twenty billion [Member]",
"label": "Twenty billion [Member]",
"terseLabel": "Twenty billion"
}
}
},
"localname": "TwentybillionMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_TysabriProductMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "TYSABRI product [Member]",
"label": "TYSABRI product [Member]",
"terseLabel": "TYSABRI"
}
}
},
"localname": "TysabriProductMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"biib_U.S.2017TaxActMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "U.S. 2017 Tax Act",
"label": "U.S. 2017 Tax Act [Member]",
"terseLabel": "2017 Tax Act"
}
}
},
"localname": "U.S.2017TaxActMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_UCBPharmaSAMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "UCB Pharma S.A.",
"label": "UCB Pharma S.A. [Member]",
"terseLabel": "UCB Pharma S.A."
}
}
},
"localname": "UCBPharmaSAMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails"
],
"xbrltype": "domainItemType"
},
"biib_UndesignatedPreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Undesignated preferred stock.",
"label": "Undesignated Preferred Stock [Member]",
"terseLabel": "Undesignated"
}
}
},
"localname": "UndesignatedPreferredStockMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_UpfrontAndMilestonePaymentsMadeToCollaborativePartner": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Total upfront and milestone payments made to collaborative partner.",
"label": "Upfront And Milestone Payments Made To Collaborative Partner",
"terseLabel": "Upfront and milestone payments made to collaborative partner"
}
}
},
"localname": "UpfrontAndMilestonePaymentsMadeToCollaborativePartner",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_Upfrontpaymentforcollaborationagreement": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Upfront payment for collaboration agreement",
"label": "Upfront payment for collaboration agreement",
"terseLabel": "Upfront payment for collaboration agreement"
}
}
},
"localname": "Upfrontpaymentforcollaborationagreement",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails"
],
"xbrltype": "monetaryItemType"
},
"biib_VUMERITYMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "VUMERITY [Member]",
"label": "VUMERITY [Member]",
"terseLabel": "VUMERITY"
}
}
},
"localname": "VUMERITYMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_WarehouseUtilitiesAndSupportSpaceMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Warehouse, Utilities and Support Space",
"label": "Warehouse, Utilities and Support Space [Member]",
"terseLabel": "Warehouse, Utilities and Support Space"
}
}
},
"localname": "WarehouseUtilitiesAndSupportSpaceMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_WeightedAveragePeriodToRecognizeCostOfNonvestedAwards": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted-average period to recognize the cost of nonvested awards.",
"label": "Weighted Average Period To Recognize Cost Of Nonvested Awards",
"verboseLabel": "Weighted-average period to recognize the cost of unvested awards"
}
}
},
"localname": "WeightedAveragePeriodToRecognizeCostOfNonvestedAwards",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"biib_WorkinprocessMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Work-in-process",
"label": "Work-in-process [Member]",
"terseLabel": "Work-in-process"
}
}
},
"localname": "WorkinprocessMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"biib_ZINBRYTAMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "ZINBRYTA [Member]",
"label": "ZINBRYTA [Member]",
"terseLabel": "ZINBRYTA"
}
}
},
"localname": "ZINBRYTAMember",
"nsuri": "http://www.biogenidec.com/20201231",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"country_CH": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "SWITZERLAND",
"terseLabel": "SWITZERLAND"
}
}
},
"localname": "CH",
"nsuri": "http://xbrl.sec.gov/country/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"country_DE": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "GERMANY",
"terseLabel": "GERMANY"
}
}
},
"localname": "DE",
"nsuri": "http://xbrl.sec.gov/country/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"country_DK": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "DENMARK",
"terseLabel": "DENMARK"
}
}
},
"localname": "DK",
"nsuri": "http://xbrl.sec.gov/country/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"country_JP": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "JAPAN",
"terseLabel": "JAPAN"
}
}
},
"localname": "JP",
"nsuri": "http://xbrl.sec.gov/country/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"country_US": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "UNITED STATES",
"terseLabel": "United States",
"verboseLabel": "U.S."
}
}
},
"localname": "US",
"nsuri": "http://xbrl.sec.gov/country/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"currency_AllCurrenciesDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "All Currencies [Domain]",
"terseLabel": "All Currencies [Domain]"
}
}
},
"localname": "AllCurrenciesDomain",
"nsuri": "http://xbrl.sec.gov/currency/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"currency_EUR": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Euro Member Countries, Euro",
"verboseLabel": "Euro"
}
}
},
"localname": "EUR",
"nsuri": "http://xbrl.sec.gov/currency/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"currency_GBP": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "United Kingdom, Pounds",
"terseLabel": "British pound"
}
}
},
"localname": "GBP",
"nsuri": "http://xbrl.sec.gov/currency/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"dei_AmendmentFlag": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the XBRL content amends previously-filed or accepted submission.",
"label": "Amendment Flag",
"terseLabel": "Amendment Flag"
}
}
},
"localname": "AmendmentFlag",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "booleanItemType"
},
"dei_CityAreaCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Area code of city",
"label": "City Area Code",
"terseLabel": "City Area Code"
}
}
},
"localname": "CityAreaCode",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "normalizedStringItemType"
},
"dei_CoverAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Cover page.",
"label": "Cover [Abstract]",
"terseLabel": "Cover [Abstract]"
}
}
},
"localname": "CoverAbstract",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"xbrltype": "stringItemType"
},
"dei_CurrentFiscalYearEndDate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "End date of current fiscal year in the format --MM-DD.",
"label": "Current Fiscal Year End Date",
"terseLabel": "Current Fiscal Year End Date"
}
}
},
"localname": "CurrentFiscalYearEndDate",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "gMonthDayItemType"
},
"dei_DocumentAnnualReport": {
"auth_ref": [
"r865",
"r866",
"r867"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as an annual report.",
"label": "Document Annual Report",
"terseLabel": "Document Annual Report"
}
}
},
"localname": "DocumentAnnualReport",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentFiscalPeriodFocus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Fiscal period values are FY, Q1, Q2, and Q3. 1st, 2nd and 3rd quarter 10-Q or 10-QT statements have value Q1, Q2, and Q3 respectively, with 10-K, 10-KT or other fiscal year statements having FY.",
"label": "Document Fiscal Period Focus",
"terseLabel": "Document Fiscal Period Focus"
}
}
},
"localname": "DocumentFiscalPeriodFocus",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "fiscalPeriodItemType"
},
"dei_DocumentFiscalYearFocus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "This is focus fiscal year of the document report in YYYY format. For a 2006 annual report, which may also provide financial information from prior periods, fiscal 2006 should be given as the fiscal year focus. Example: 2006.",
"label": "Document Fiscal Year Focus",
"terseLabel": "Document Fiscal Year Focus"
}
}
},
"localname": "DocumentFiscalYearFocus",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "gYearItemType"
},
"dei_DocumentPeriodEndDate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The end date of the period reflected on the cover page if a periodic report. For all other reports and registration statements containing historical data, it is the date up through which that historical data is presented. If there is no historical data in the report, use the filing date. The format of the date is YYYY-MM-DD.",
"label": "Document Period End Date",
"terseLabel": "Document Period End Date"
}
}
},
"localname": "DocumentPeriodEndDate",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "dateItemType"
},
"dei_DocumentTransitionReport": {
"auth_ref": [
"r868"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as a transition report.",
"label": "Document Transition Report",
"terseLabel": "Document Transition Report"
}
}
},
"localname": "DocumentTransitionReport",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentType": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.",
"label": "Document Type",
"terseLabel": "Document Type"
}
}
},
"localname": "DocumentType",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "submissionTypeItemType"
},
"dei_DocumentsIncorporatedByReferenceTextBlock": {
"auth_ref": [
"r863"
],
"lang": {
"en-us": {
"role": {
"documentation": "Documents incorporated by reference.",
"label": "Documents Incorporated by Reference [Text Block]",
"terseLabel": "Documents Incorporated by Reference"
}
}
},
"localname": "DocumentsIncorporatedByReferenceTextBlock",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "textBlockItemType"
},
"dei_EntityAddressAddressLine1": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Address Line 1 such as Attn, Building Name, Street Name",
"label": "Entity Address, Address Line One",
"terseLabel": "Entity Address, Address Line One"
}
}
},
"localname": "EntityAddressAddressLine1",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressCityOrTown": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the City or Town",
"label": "Entity Address, City or Town",
"terseLabel": "Entity Address, City or Town"
}
}
},
"localname": "EntityAddressCityOrTown",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressPostalZipCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Code for the postal or zip code",
"label": "Entity Address, Postal Zip Code",
"terseLabel": "Entity Address, Postal Zip Code"
}
}
},
"localname": "EntityAddressPostalZipCode",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressStateOrProvince": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the state or province.",
"label": "Entity Address, State or Province",
"terseLabel": "Entity Address, State or Province"
}
}
},
"localname": "EntityAddressStateOrProvince",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "stateOrProvinceItemType"
},
"dei_EntityCentralIndexKey": {
"auth_ref": [
"r869"
],
"lang": {
"en-us": {
"role": {
"documentation": "A unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK.",
"label": "Entity Central Index Key",
"terseLabel": "Entity Central Index Key"
}
}
},
"localname": "EntityCentralIndexKey",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "centralIndexKeyItemType"
},
"dei_EntityCommonStockSharesOutstanding": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate number of shares or other units outstanding of each of registrant's classes of capital or common stock or other ownership interests, if and as stated on cover of related periodic report. Where multiple classes or units exist define each class/interest by adding class of stock items such as Common Class A [Member], Common Class B [Member] or Partnership Interest [Member] onto the Instrument [Domain] of the Entity Listings, Instrument.",
"label": "Entity Common Stock, Shares Outstanding",
"terseLabel": "Entity Common Stock, Shares Outstanding"
}
}
},
"localname": "EntityCommonStockSharesOutstanding",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "sharesItemType"
},
"dei_EntityCurrentReportingStatus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' whether registrants (1) have filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that registrants were required to file such reports), and (2) have been subject to such filing requirements for the past 90 days. This information should be based on the registrant's current or most recent filing containing the related disclosure.",
"label": "Entity Current Reporting Status",
"terseLabel": "Entity Current Reporting Status"
}
}
},
"localname": "EntityCurrentReportingStatus",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "All the names of the entities being reported upon in a document. Any legal structure used to conduct activities or to hold assets. Some examples of such structures are corporations, partnerships, limited liability companies, grantor trusts, and other trusts. This item does not include business and geographical segments which are included in the geographical or business segments domains.",
"label": "Entity [Domain]",
"terseLabel": "Entity [Domain]"
}
}
},
"localname": "EntityDomain",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"dei_EntityEmergingGrowthCompany": {
"auth_ref": [
"r869"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate if registrant meets the emerging growth company criteria.",
"label": "Entity Emerging Growth Company",
"terseLabel": "Entity Emerging Growth Company"
}
}
},
"localname": "EntityEmergingGrowthCompany",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "booleanItemType"
},
"dei_EntityFileNumber": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Commission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.",
"label": "Entity File Number",
"terseLabel": "Entity File Number"
}
}
},
"localname": "EntityFileNumber",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "fileNumberItemType"
},
"dei_EntityFilerCategory": {
"auth_ref": [
"r869"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate whether the registrant is one of the following: Large Accelerated Filer, Accelerated Filer, Non-accelerated Filer. Definitions of these categories are stated in Rule 12b-2 of the Exchange Act. This information should be based on the registrant's current or most recent filing containing the related disclosure.",
"label": "Entity Filer Category",
"terseLabel": "Entity Filer Category"
}
}
},
"localname": "EntityFilerCategory",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "filerCategoryItemType"
},
"dei_EntityIncorporationStateCountryCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Two-character EDGAR code representing the state or country of incorporation.",
"label": "Entity Incorporation, State or Country Code",
"terseLabel": "Entity Incorporation, State or Country Code"
}
}
},
"localname": "EntityIncorporationStateCountryCode",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "edgarStateCountryItemType"
},
"dei_EntityInteractiveDataCurrent": {
"auth_ref": [
"r870"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).",
"label": "Entity Interactive Data Current",
"terseLabel": "Entity Interactive Data Current"
}
}
},
"localname": "EntityInteractiveDataCurrent",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityPublicFloat": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and asked price of such common equity, as of the last business day of the registrant's most recently completed second fiscal quarter.",
"label": "Entity Public Float",
"terseLabel": "Entity Public Float"
}
}
},
"localname": "EntityPublicFloat",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "monetaryItemType"
},
"dei_EntityRegistrantName": {
"auth_ref": [
"r869"
],
"lang": {
"en-us": {
"role": {
"documentation": "The exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC.",
"label": "Entity Registrant Name",
"terseLabel": "Entity Registrant Name"
}
}
},
"localname": "EntityRegistrantName",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityShellCompany": {
"auth_ref": [
"r869"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the registrant is a shell company as defined in Rule 12b-2 of the Exchange Act.",
"label": "Entity Shell Company",
"terseLabel": "Entity Shell Company"
}
}
},
"localname": "EntityShellCompany",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "booleanItemType"
},
"dei_EntitySmallBusiness": {
"auth_ref": [
"r869"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicates that the company is a Smaller Reporting Company (SRC).",
"label": "Entity Small Business",
"terseLabel": "Entity Small Business"
}
}
},
"localname": "EntitySmallBusiness",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "booleanItemType"
},
"dei_EntityTaxIdentificationNumber": {
"auth_ref": [
"r869"
],
"lang": {
"en-us": {
"role": {
"documentation": "The Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS.",
"label": "Entity Tax Identification Number",
"terseLabel": "Entity Tax Identification Number"
}
}
},
"localname": "EntityTaxIdentificationNumber",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "employerIdItemType"
},
"dei_EntityVoluntaryFilers": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.",
"label": "Entity Voluntary Filers",
"terseLabel": "Entity Voluntary Filers"
}
}
},
"localname": "EntityVoluntaryFilers",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityWellKnownSeasonedIssuer": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Is used on Form Type: 10-K, 10-Q, 8-K, 20-F, 6-K, 10-K/A, 10-Q/A, 20-F/A, 6-K/A, N-CSR, N-Q, N-1A.",
"label": "Entity Well-known Seasoned Issuer",
"terseLabel": "Entity Well-known Seasoned Issuer"
}
}
},
"localname": "EntityWellKnownSeasonedIssuer",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "yesNoItemType"
},
"dei_IcfrAuditorAttestationFlag": {
"auth_ref": [
"r865",
"r866",
"r867"
],
"lang": {
"en-us": {
"role": {
"label": "ICFR Auditor Attestation Flag",
"terseLabel": "ICFR Auditor Attestation Flag"
}
}
},
"localname": "IcfrAuditorAttestationFlag",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "booleanItemType"
},
"dei_LegalEntityAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The set of legal entities associated with a report.",
"label": "Legal Entity [Axis]",
"terseLabel": "Legal Entity [Axis]"
}
}
},
"localname": "LegalEntityAxis",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"dei_LocalPhoneNumber": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Local phone number for entity.",
"label": "Local Phone Number",
"terseLabel": "Local Phone Number"
}
}
},
"localname": "LocalPhoneNumber",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "normalizedStringItemType"
},
"dei_Security12bTitle": {
"auth_ref": [
"r862"
],
"lang": {
"en-us": {
"role": {
"documentation": "Title of a 12(b) registered security.",
"label": "Title of 12(b) Security",
"terseLabel": "Title of 12(b) Security"
}
}
},
"localname": "Security12bTitle",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "securityTitleItemType"
},
"dei_SecurityExchangeName": {
"auth_ref": [
"r864"
],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the Exchange on which a security is registered.",
"label": "Security Exchange Name",
"terseLabel": "Security Exchange Name"
}
}
},
"localname": "SecurityExchangeName",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "edgarExchangeCodeItemType"
},
"dei_TradingSymbol": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Trading symbol of an instrument as listed on an exchange.",
"label": "Trading Symbol",
"terseLabel": "Trading Symbol"
}
}
},
"localname": "TradingSymbol",
"nsuri": "http://xbrl.sec.gov/dei/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CoverPage"
],
"xbrltype": "tradingSymbolItemType"
},
"srt_AsiaMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Asia [Member]",
"terseLabel": "Asia"
}
}
},
"localname": "AsiaMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"srt_ConsolidatedEntitiesAxis": {
"auth_ref": [
"r611",
"r612",
"r619",
"r620",
"r856"
],
"lang": {
"en-us": {
"role": {
"label": "Consolidated Entities [Axis]",
"terseLabel": "Consolidated Entities [Axis]"
}
}
},
"localname": "ConsolidatedEntitiesAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "stringItemType"
},
"srt_ConsolidatedEntitiesDomain": {
"auth_ref": [
"r611",
"r612",
"r619",
"r620"
],
"lang": {
"en-us": {
"role": {
"label": "Consolidated Entities [Domain]",
"terseLabel": "Consolidated Entities [Domain]"
}
}
},
"localname": "ConsolidatedEntitiesDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"srt_CumulativeEffectPeriodOfAdoptionAdjustedBalanceMember": {
"auth_ref": [
"r183",
"r188",
"r294",
"r523",
"r524",
"r525",
"r567",
"r568"
],
"lang": {
"en-us": {
"role": {
"label": "Cumulative Effect, Period of Adoption, Adjusted Balance [Member]",
"terseLabel": "Cumulative Effect, Period of Adoption, Adjusted Balance"
}
}
},
"localname": "CumulativeEffectPeriodOfAdoptionAdjustedBalanceMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "domainItemType"
},
"srt_CumulativeEffectPeriodOfAdoptionAdjustmentMember": {
"auth_ref": [
"r183",
"r188",
"r294",
"r523",
"r524",
"r525",
"r567",
"r568"
],
"lang": {
"en-us": {
"role": {
"label": "Cumulative Effect, Period of Adoption, Adjustment [Member]",
"terseLabel": "Cumulative Effect, Period of Adoption, Adjustment"
}
}
},
"localname": "CumulativeEffectPeriodOfAdoptionAdjustmentMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_CumulativeEffectPeriodOfAdoptionAxis": {
"auth_ref": [
"r183",
"r188",
"r294",
"r523",
"r524",
"r525",
"r567",
"r568"
],
"lang": {
"en-us": {
"role": {
"label": "Cumulative Effect, Period of Adoption [Axis]",
"terseLabel": "Cumulative Effect, Period of Adoption [Axis]"
}
}
},
"localname": "CumulativeEffectPeriodOfAdoptionAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"srt_CumulativeEffectPeriodOfAdoptionDomain": {
"auth_ref": [
"r183",
"r188",
"r294",
"r523",
"r524",
"r525",
"r567",
"r568"
],
"lang": {
"en-us": {
"role": {
"label": "Cumulative Effect, Period of Adoption [Domain]",
"terseLabel": "Cumulative Effect, Period of Adoption [Domain]"
}
}
},
"localname": "CumulativeEffectPeriodOfAdoptionDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_CurrencyAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Currency [Axis]",
"terseLabel": "Currency [Axis]"
}
}
},
"localname": "CurrencyAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails"
],
"xbrltype": "stringItemType"
},
"srt_DirectorMember": {
"auth_ref": [
"r260"
],
"lang": {
"en-us": {
"role": {
"label": "Director [Member]",
"terseLabel": "Director"
}
}
},
"localname": "DirectorMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "domainItemType"
},
"srt_EquityMethodInvesteeNameDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Investment, Name [Domain]",
"terseLabel": "Investment, Name [Domain]",
"verboseLabel": "Equity Method Investee, Name [Domain]"
}
}
},
"localname": "EquityMethodInvesteeNameDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_EuropeMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Europe [Member]",
"terseLabel": "Europe"
}
}
},
"localname": "EuropeMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"srt_MajorCustomersAxis": {
"auth_ref": [
"r258",
"r393",
"r397",
"r816"
],
"lang": {
"en-us": {
"role": {
"label": "Customer [Axis]",
"terseLabel": "Customer [Axis]"
}
}
},
"localname": "MajorCustomersAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails"
],
"xbrltype": "stringItemType"
},
"srt_MaximumMember": {
"auth_ref": [
"r484",
"r490",
"r727",
"r728",
"r729",
"r730",
"r731",
"r732",
"r752",
"r813",
"r817"
],
"lang": {
"en-us": {
"role": {
"label": "Maximum [Member]",
"terseLabel": "Maximum",
"verboseLabel": "Maximum"
}
}
},
"localname": "MaximumMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails",
"http://www.biogenidec.com/role/LeasesNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"srt_MinimumMember": {
"auth_ref": [
"r484",
"r490",
"r727",
"r728",
"r729",
"r730",
"r731",
"r732",
"r752",
"r813",
"r817"
],
"lang": {
"en-us": {
"role": {
"label": "Minimum [Member]",
"terseLabel": "Minimum",
"verboseLabel": "Minimum"
}
}
},
"localname": "MinimumMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails",
"http://www.biogenidec.com/role/LeasesNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"srt_NameOfMajorCustomerDomain": {
"auth_ref": [
"r258",
"r393",
"r397",
"r816"
],
"lang": {
"en-us": {
"role": {
"label": "Customer [Domain]",
"terseLabel": "Customer [Domain]"
}
}
},
"localname": "NameOfMajorCustomerDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails"
],
"xbrltype": "domainItemType"
},
"srt_ProductOrServiceAxis": {
"auth_ref": [
"r254",
"r393",
"r395",
"r754",
"r812",
"r814"
],
"lang": {
"en-us": {
"role": {
"label": "Product and Service [Axis]",
"terseLabel": "Product and Service [Axis]"
}
}
},
"localname": "ProductOrServiceAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "stringItemType"
},
"srt_ProductsAndServicesDomain": {
"auth_ref": [
"r254",
"r393",
"r395",
"r754",
"r812",
"r814"
],
"lang": {
"en-us": {
"role": {
"label": "Product and Service [Domain]",
"terseLabel": "Product and Service [Domain]"
}
}
},
"localname": "ProductsAndServicesDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"srt_RangeAxis": {
"auth_ref": [
"r427",
"r484",
"r490",
"r727",
"r728",
"r729",
"r730",
"r731",
"r732",
"r752",
"r813",
"r817"
],
"lang": {
"en-us": {
"role": {
"label": "Statistical Measurement [Axis]",
"terseLabel": "Statistical Measurement [Axis]"
}
}
},
"localname": "RangeAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails",
"http://www.biogenidec.com/role/LeasesNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "stringItemType"
},
"srt_RangeMember": {
"auth_ref": [
"r427",
"r484",
"r490",
"r727",
"r728",
"r729",
"r730",
"r731",
"r732",
"r752",
"r813",
"r817"
],
"lang": {
"en-us": {
"role": {
"label": "Statistical Measurement [Domain]",
"terseLabel": "Statistical Measurement [Domain]"
}
}
},
"localname": "RangeMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails",
"http://www.biogenidec.com/role/LeasesNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"srt_ScenarioForecastMember": {
"auth_ref": [
"r485"
],
"lang": {
"en-us": {
"role": {
"label": "Forecast [Member]",
"terseLabel": "Forecast"
}
}
},
"localname": "ScenarioForecastMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_ScenarioUnspecifiedDomain": {
"auth_ref": [
"r485"
],
"lang": {
"en-us": {
"role": {
"label": "Scenario [Domain]",
"terseLabel": "Scenario [Domain]"
}
}
},
"localname": "ScenarioUnspecifiedDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_ScheduleOfEquityMethodInvestmentEquityMethodInvesteeNameAxis": {
"auth_ref": [
"r290"
],
"lang": {
"en-us": {
"role": {
"label": "Investment, Name [Axis]",
"terseLabel": "Equity Method Investee, Name [Axis]",
"verboseLabel": "Investment, Name [Axis]"
}
}
},
"localname": "ScheduleOfEquityMethodInvestmentEquityMethodInvesteeNameAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"srt_SegmentGeographicalDomain": {
"auth_ref": [
"r255",
"r256",
"r393",
"r396",
"r815",
"r847",
"r848",
"r849",
"r850",
"r851",
"r852",
"r853",
"r854"
],
"lang": {
"en-us": {
"role": {
"label": "Geographical [Domain]",
"terseLabel": "Geographical [Domain]"
}
}
},
"localname": "SegmentGeographicalDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails",
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_StatementGeographicalAxis": {
"auth_ref": [
"r255",
"r256",
"r393",
"r396",
"r815",
"r840",
"r847",
"r848",
"r849",
"r850",
"r851",
"r852",
"r853",
"r854",
"r855"
],
"lang": {
"en-us": {
"role": {
"label": "Geographical [Axis]",
"terseLabel": "Geographical [Axis]"
}
}
},
"localname": "StatementGeographicalAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails",
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"srt_StatementScenarioAxis": {
"auth_ref": [
"r358",
"r485",
"r720"
],
"lang": {
"en-us": {
"role": {
"label": "Scenario [Axis]",
"terseLabel": "Scenario [Axis]"
}
}
},
"localname": "StatementScenarioAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"srt_TitleOfIndividualAxis": {
"auth_ref": [
"r260",
"r719"
],
"lang": {
"en-us": {
"role": {
"label": "Title of Individual [Axis]",
"terseLabel": "Title of Individual [Axis]"
}
}
},
"localname": "TitleOfIndividualAxis",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "stringItemType"
},
"srt_TitleOfIndividualWithRelationshipToEntityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Title of Individual [Domain]",
"terseLabel": "Title of Individual [Domain]"
}
}
},
"localname": "TitleOfIndividualWithRelationshipToEntityDomain",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "domainItemType"
},
"srt_ValuationAndQualifyingAccountsDisclosureLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Line Items]",
"terseLabel": "SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Line Items]"
}
}
},
"localname": "ValuationAndQualifyingAccountsDisclosureLineItems",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails",
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails",
"http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "stringItemType"
},
"srt_ValuationAndQualifyingAccountsDisclosureTable": {
"auth_ref": [
"r178"
],
"lang": {
"en-us": {
"role": {
"label": "SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Table]",
"terseLabel": "SEC Schedule, 12-09, Valuation and Qualifying Accounts Disclosure [Table]"
}
}
},
"localname": "ValuationAndQualifyingAccountsDisclosureTable",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails",
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails",
"http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "stringItemType"
},
"srt_WeightedAverageMember": {
"auth_ref": [
"r727",
"r729",
"r732"
],
"lang": {
"en-us": {
"role": {
"label": "Weighted Average [Member]",
"terseLabel": "Weighted Average"
}
}
},
"localname": "WeightedAverageMember",
"nsuri": "http://fasb.org/srt/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AOCIAttributableToParentNetOfTaxRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "AOCI Attributable to Parent, Net of Tax [Roll Forward]",
"terseLabel": "AOCI Attributable to Parent, Net of Tax [Roll Forward]"
}
}
},
"localname": "AOCIAttributableToParentNetOfTaxRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AccountingPoliciesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Accounting Policies [Abstract]",
"terseLabel": "Accounting Policies [Abstract]"
}
}
},
"localname": "AccountingPoliciesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_AccountsPayableCurrent": {
"auth_ref": [
"r64"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 1.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Accounts Payable, Current",
"verboseLabel": "Accounts payable"
}
}
},
"localname": "AccountsPayableCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccountsReceivableMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Due from customers or clients for goods or services that have been delivered or sold.",
"label": "Accounts Receivable [Member]",
"terseLabel": "Reduction of accounts receivable"
}
}
},
"localname": "AccountsReceivableMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccountsReceivableNetCurrent": {
"auth_ref": [
"r23",
"r44",
"r261",
"r262"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 4.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after allowance for credit loss, of right to consideration from customer for product sold and service rendered in normal course of business, classified as current.",
"label": "Accounts Receivable, after Allowance for Credit Loss, Current",
"verboseLabel": "Accounts receivable, net"
}
}
},
"localname": "AccountsReceivableNetCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccruedIncomeTaxesNoncurrent": {
"auth_ref": [
"r39",
"r760",
"r789"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying amount as of the balance sheet date of the unpaid sum of the known and estimated amounts payable to satisfy all domestic and foreign income tax obligations due beyond one year or the operating cycle, whichever is longer. Alternate captions include income taxes payable, noncurrent.",
"label": "Accrued Income Taxes, Noncurrent",
"terseLabel": "Accrued income taxes"
}
}
},
"localname": "AccruedIncomeTaxesNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccruedLiabilitiesMember": {
"auth_ref": [
"r69"
],
"lang": {
"en-us": {
"role": {
"documentation": "This item represents obligations incurred and payable, pertaining to costs that are statutory in nature, are incurred on contractual obligations, or accumulate over time and for which invoices have not yet been received or will not be rendered.",
"label": "Accrued Liabilities [Member]",
"terseLabel": "Accrued expenses and other"
}
}
},
"localname": "AccruedLiabilitiesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccruedRoyaltiesCurrent": {
"auth_ref": [
"r29",
"r30",
"r69"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": 2.0,
"parentTag": "us-gaap_OtherLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of obligations incurred through that date and payable for royalties. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Accrued Royalties, Current",
"terseLabel": "Accrued Royalties, Current"
}
}
},
"localname": "AccruedRoyaltiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccumulatedDefinedBenefitPlansAdjustmentMember": {
"auth_ref": [
"r93",
"r98",
"r100",
"r618"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated other comprehensive (income) loss related to defined benefit plans attributable to the parent.",
"label": "Accumulated Defined Benefit Plans Adjustment Attributable to Parent [Member]",
"terseLabel": "Unfunded Status of Postretirement Benefit Plans, net of tax"
}
}
},
"localname": "AccumulatedDefinedBenefitPlansAdjustmentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccumulatedDepreciationDepletionAndAmortizationPropertyPlantAndEquipment": {
"auth_ref": [
"r61",
"r341"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 2.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentNet",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of accumulated depreciation, depletion and amortization for physical assets used in the normal conduct of business to produce goods and services.",
"label": "Accumulated Depreciation, Depletion and Amortization, Property, Plant, and Equipment",
"negatedTerseLabel": "Less: accumulated depreciation"
}
}
},
"localname": "AccumulatedDepreciationDepletionAndAmortizationPropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccumulatedGainLossNetCashFlowHedgeParentMember": {
"auth_ref": [
"r98",
"r108",
"r617"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated other comprehensive income (loss) from gain (loss) of derivative instrument designated and qualifying as cash flow hedge included in assessment of hedge effectiveness, attributable to parent.",
"label": "Accumulated Gain (Loss), Net, Cash Flow Hedge, Parent [Member]",
"terseLabel": "Unrealized Gains (Losses) on Cash Flow Hedges, net of tax",
"verboseLabel": "Gains (losses) on cash flow hedges"
}
}
},
"localname": "AccumulatedGainLossNetCashFlowHedgeParentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccumulatedNetUnrealizedInvestmentGainLossMember": {
"auth_ref": [
"r90",
"r91",
"r92",
"r98",
"r100"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated unrealized gain (loss) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), attributable to parent.",
"label": "AOCI, Accumulated Gain (Loss), Debt Securities, Available-for-sale, Parent [Member]",
"terseLabel": "Unrealized Gains (Losses) on Securities Available for Sale, net of tax",
"verboseLabel": "Gains (losses) on securities available for sale"
}
}
},
"localname": "AccumulatedNetUnrealizedInvestmentGainLossMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccumulatedOtherComprehensiveIncomeLossDerivativeQualifyingAsHedgeExcludedComponentIncludingPortionAttributableToNoncontrollingInterestMember": {
"auth_ref": [
"r98",
"r660"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated other comprehensive income (loss) from increase (decrease) in value of excluded component of derivative hedge, including portion attributable to noncontrolling interest.",
"label": "Accumulated Other Comprehensive Income (Loss), Derivative Qualifying as Hedge, Excluded Component, Including Portion Attributable to Noncontrolling Interest [Member]",
"terseLabel": "Gains (Losses) on Net Investment Hedge, Net of Tax",
"verboseLabel": "Gains (losses) on net investment hedge"
}
}
},
"localname": "AccumulatedOtherComprehensiveIncomeLossDerivativeQualifyingAsHedgeExcludedComponentIncludingPortionAttributableToNoncontrollingInterestMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccumulatedOtherComprehensiveIncomeLossLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Accumulated Other Comprehensive Income (Loss) [Line Items]",
"terseLabel": "Accumulated Other Comprehensive Income (Loss) [Line Items]"
}
}
},
"localname": "AccumulatedOtherComprehensiveIncomeLossLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AccumulatedOtherComprehensiveIncomeLossNetOfTax": {
"auth_ref": [
"r47",
"r96",
"r97",
"r98",
"r792",
"r822",
"r823"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 4.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated change in equity from transactions and other events and circumstances from non-owner sources, net of tax effect, at period end. Excludes Net Income (Loss), and accumulated changes in equity from transactions resulting from investments by owners and distributions to owners. Includes foreign currency translation items, certain pension adjustments, unrealized gains and losses on certain investments in debt and equity securities, other than temporary impairment (OTTI) losses related to factors other than credit losses on available-for-sale and held-to-maturity debt securities that an entity does not intend to sell and it is not more likely than not that the entity will be required to sell before recovery of the amortized cost basis, as well as changes in the fair value of derivatives related to the effective portion of a designated cash flow hedge.",
"label": "Accumulated Other Comprehensive Income (Loss), Net of Tax",
"periodEndLabel": "AOCI, ending balance",
"periodStartLabel": "AOCI, beginning balance",
"verboseLabel": "Accumulated other comprehensive loss"
}
}
},
"localname": "AccumulatedOtherComprehensiveIncomeLossNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccumulatedOtherComprehensiveIncomeLossTable": {
"auth_ref": [
"r686",
"r687",
"r688",
"r689",
"r690",
"r692"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about components of accumulated other comprehensive income (loss).",
"label": "Accumulated Other Comprehensive Income (Loss) [Table]",
"terseLabel": "Accumulated Other Comprehensive Income (Loss) [Table]"
}
}
},
"localname": "AccumulatedOtherComprehensiveIncomeLossTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AccumulatedOtherComprehensiveIncomeMember": {
"auth_ref": [
"r95",
"r98",
"r100",
"r180",
"r181",
"r182",
"r618",
"r818",
"r819"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated increase (decrease) in equity from transactions and other events and circumstances from non-owner sources, attributable to the parent. Excludes net income (loss), and accumulated changes in equity from transactions resulting from investments by owners and distributions to owners.",
"label": "AOCI Attributable to Parent [Member]",
"verboseLabel": "Accumulated other comprehensive loss"
}
}
},
"localname": "AccumulatedOtherComprehensiveIncomeMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccumulatedTranslationAdjustmentMember": {
"auth_ref": [
"r88",
"r98",
"r100",
"r618",
"r687",
"r688",
"r689",
"r690",
"r692"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated other comprehensive income (loss) resulting from foreign currency translation adjustments, foreign currency transactions designated and effective as economic hedges of a net investment in a foreign entity and intra-entity foreign currency transactions that are of a long-term-investment nature, attributable to the parent.",
"label": "Accumulated Foreign Currency Adjustment Attributable to Parent [Member]",
"terseLabel": "Currency Translation Adjustments"
}
}
},
"localname": "AccumulatedTranslationAdjustmentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AdditionalFinancialInformationDisclosureTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosures of supplemental information, including descriptions and amounts, related to the balance sheet, income statement, and/or cash flow statement.",
"label": "Additional Financial Information Disclosure [Text Block]",
"verboseLabel": "Other Consolidated Financial Statement Detail"
}
}
},
"localname": "AdditionalFinancialInformationDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetail"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_AdditionalPaidInCapital": {
"auth_ref": [
"r45",
"r526"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 3.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Excess of issue price over par or stated value of the entity's capital stock and amounts received from other transactions involving the entity's stock or stockholders. Includes adjustments to additional paid in capital. Some examples of such adjustments include recording the issuance of debt with a beneficial conversion feature and certain tax consequences of equity instruments awarded to employees. Use this element for the aggregate amount of additional paid-in capital associated with common and preferred stock. For additional paid-in capital associated with only common stock, use the element additional paid in capital, common stock. For additional paid-in capital associated with only preferred stock, use the element additional paid in capital, preferred stock.",
"label": "Additional Paid in Capital",
"verboseLabel": "Additional paid-in capital"
}
}
},
"localname": "AdditionalPaidInCapital",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AdditionalPaidInCapitalMember": {
"auth_ref": [
"r180",
"r181",
"r182",
"r523",
"r524",
"r525"
],
"lang": {
"en-us": {
"role": {
"documentation": "Excess of issue price over par or stated value of the entity's capital stock and amounts received from other transactions involving the entity's stock or stockholders.",
"label": "Additional Paid-in Capital [Member]",
"verboseLabel": "Additional paid-in capital"
}
}
},
"localname": "AdditionalPaidInCapitalMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "domainItemType"
},
"us-gaap_AdjustmentForAmortization": {
"auth_ref": [
"r151",
"r329"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate amount of recurring noncash expense charged against earnings in the period to allocate the cost of assets over their estimated remaining economic lives.",
"label": "Amortization",
"negatedTerseLabel": "Amortization"
}
}
},
"localname": "AdjustmentForAmortization",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AdjustmentsToReconcileNetIncomeLossToCashProvidedByUsedInOperatingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Adjustments to Reconcile Net Income (Loss) to Cash Provided by (Used in) Operating Activities [Abstract]",
"verboseLabel": "Adjustments to reconcile net income to net cash flows from operating activities:"
}
}
},
"localname": "AdjustmentsToReconcileNetIncomeLossToCashProvidedByUsedInOperatingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "stringItemType"
},
"us-gaap_AdvertisingExpense": {
"auth_ref": [
"r531"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount charged to advertising expense for the period, which are expenses incurred with the objective of increasing revenue for a specified brand, product or product line.",
"label": "Advertising Expense",
"terseLabel": "Advertising costs"
}
}
},
"localname": "AdvertisingExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AllocatedShareBasedCompensationExpense": {
"auth_ref": [
"r493",
"r517",
"r527"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expense for award under share-based payment arrangement. Excludes amount capitalized.",
"label": "Share-based Payment Arrangement, Expense",
"negatedTerseLabel": "Compensation expense related to share-based payments",
"verboseLabel": "Share-based compensation expense"
}
}
},
"localname": "AllocatedShareBasedCompensationExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AllocatedShareBasedCompensationExpenseNetOfTax": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after tax, of expense for award under share-based payment arrangement.",
"label": "Share-based Payment Arrangement, Expense, after Tax",
"verboseLabel": "Subtotal"
}
}
},
"localname": "AllocatedShareBasedCompensationExpenseNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AmortizationOfIntangibleAssets": {
"auth_ref": [
"r151",
"r320",
"r329"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate expense charged against earnings to allocate the cost of intangible assets (nonphysical assets not used in production) in a systematic and rational manner to the periods expected to benefit from such assets. As a noncash expense, this element is added back to net income when calculating cash provided by or used in operations using the indirect method.",
"label": "Amortization of Intangible Assets",
"verboseLabel": "Amortization of acquired intangible assets"
}
}
},
"localname": "AmortizationOfIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis": {
"auth_ref": [
"r207"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of antidilutive security.",
"label": "Antidilutive Securities [Axis]",
"terseLabel": "Antidilutive Securities [Axis]"
}
}
},
"localname": "AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Antidilutive Securities Excluded from Computation of Earnings Per Share [Line Items]",
"terseLabel": "Antidilutive Securities Excluded from Computation of Earnings Per Share"
}
}
},
"localname": "AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AntidilutiveSecuritiesNameDomain": {
"auth_ref": [
"r207"
],
"lang": {
"en-us": {
"role": {
"documentation": "Incremental common shares attributable to securities that were not included in diluted earnings per share (EPS) because to do so would increase EPS amounts or decrease loss per share amounts for the period presented.",
"label": "Antidilutive Securities, Name [Domain]",
"terseLabel": "Antidilutive Securities, Name [Domain]"
}
}
},
"localname": "AntidilutiveSecuritiesNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AociDerivativeQualifyingAsHedgeExcludedComponentAfterTax": {
"auth_ref": [
"r661"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after tax, of accumulated gain (loss) from increase (decrease) in value of excluded component of derivative hedge.",
"label": "AOCI, Derivative Qualifying as Hedge, Excluded Component, after Tax",
"terseLabel": "Derivative qualifying as hedge, excluded component, after tax"
}
}
},
"localname": "AociDerivativeQualifyingAsHedgeExcludedComponentAfterTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ArrangementsAndNonarrangementTransactionsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Collaborative arrangement and arrangement other than collaborative applicable to revenue-generating activity or operations.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Domain]",
"terseLabel": "Collaborative Arrangement and Arrangement Other than Collaborative [Domain]"
}
}
},
"localname": "ArrangementsAndNonarrangementTransactionsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AssetBackedSecuritiesMember": {
"auth_ref": [
"r280",
"r428"
],
"lang": {
"en-us": {
"role": {
"documentation": "Securities that are primarily serviced by the cash flows of a discrete pool of receivables or other financial assets for example, but not limited to, credit card receivables, car loans, recreational vehicle loans, and mobile home loans.",
"label": "Asset-backed Securities [Member]",
"verboseLabel": "Mortgage and other asset backed securities"
}
}
},
"localname": "AssetBackedSecuritiesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_Assets": {
"auth_ref": [
"r163",
"r239",
"r244",
"r250",
"r293",
"r611",
"r619",
"r680",
"r758",
"r788"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying amounts as of the balance sheet date of all assets that are recognized. Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events.",
"label": "Assets",
"totalLabel": "Total assets"
}
}
},
"localname": "Assets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AssetsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Assets [Abstract]",
"verboseLabel": "ASSETS"
}
}
},
"localname": "AssetsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "stringItemType"
},
"us-gaap_AssetsCurrent": {
"auth_ref": [
"r24",
"r25",
"r83",
"r163",
"r293",
"r611",
"r619",
"r680"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 1.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying amounts as of the balance sheet date of all assets that are expected to be realized in cash, sold, or consumed within one year (or the normal operating cycle, if longer). Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events.",
"label": "Assets, Current",
"totalLabel": "Total current assets"
}
}
},
"localname": "AssetsCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AssetsCurrentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Assets, Current [Abstract]",
"verboseLabel": "Current assets:"
}
}
},
"localname": "AssetsCurrentAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "stringItemType"
},
"us-gaap_AssetsFairValueDisclosure": {
"auth_ref": [
"r663"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of probable future economic benefits obtained or controlled by an entity as a result of past transactions or events.",
"label": "Assets, Fair Value Disclosure",
"verboseLabel": "Total"
}
}
},
"localname": "AssetsFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AssetsFairValueDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Assets, Fair Value Disclosure [Abstract]",
"verboseLabel": "Assets:"
}
}
},
"localname": "AssetsFairValueDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AvailableForSaleDebtSecuritiesAccumulatedGrossUnrealizedGainBeforeTax": {
"auth_ref": [
"r273"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": 1.0,
"parentTag": "us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before tax, of unrealized gain in accumulated other comprehensive income (AOCI) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Debt Securities, Available-for-sale, Accumulated Gross Unrealized Gain, before Tax",
"terseLabel": "Gross Unrealized Gains"
}
}
},
"localname": "AvailableForSaleDebtSecuritiesAccumulatedGrossUnrealizedGainBeforeTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleDebtSecuritiesAccumulatedGrossUnrealizedLossBeforeTax": {
"auth_ref": [
"r274"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": 2.0,
"parentTag": "us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before tax, of unrealized loss in accumulated other comprehensive income (AOCI) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Debt Securities, Available-for-sale, Accumulated Gross Unrealized Loss, before Tax",
"negatedLabel": "Gross Unrealized Losses"
}
}
},
"localname": "AvailableForSaleDebtSecuritiesAccumulatedGrossUnrealizedLossBeforeTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis": {
"auth_ref": [
"r271",
"r300"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amortized cost of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Debt Securities, Available-for-sale, Amortized Cost",
"totalLabel": "Amortized Cost"
}
}
},
"localname": "AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtMaturitiesAfterOneThroughFiveYearsAmortizedCost": {
"auth_ref": [
"r278"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": 2.0,
"parentTag": "us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amortized cost of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), with single maturity date and allocated without single maturity date, maturing in second through fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Debt Securities, Available-for-Sale, Amortized Cost, Maturity, Allocated and Single Maturity Date, after Year One Through Five",
"verboseLabel": "Due after one year through five years"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesAfterOneThroughFiveYearsAmortizedCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtMaturitiesAfterOneThroughFiveYearsFairValue": {
"auth_ref": [
"r275",
"r278",
"r783"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": 2.0,
"parentTag": "us-gaap_AvailableForSaleSecuritiesDebtSecurities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), with single maturity date and allocated without single maturity date, maturing in second through fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Debt Securities, Available-for-Sale, Fair Value, Maturity, Allocated and Single Maturity Date, after Year One Through Five",
"verboseLabel": "Due after one year through five years"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesAfterOneThroughFiveYearsFairValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtMaturitiesSingleMaturityDateAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Debt Securities, Available-for-sale, Maturity, Allocated and Single Maturity Date, Fair Value [Abstract]",
"terseLabel": "Estimated Fair\u00a0Value"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesSingleMaturityDateAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtMaturitiesSingleMaturityDateAmortizedCostBasisAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Debt Securities, Available-for-sale, Maturity, Allocated and Single Maturity Date, Amortized Cost [Abstract]",
"terseLabel": "Amortized Cost"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesSingleMaturityDateAmortizedCostBasisAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtMaturitiesWithinOneYearAmortizedCost": {
"auth_ref": [
"r277"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": 1.0,
"parentTag": "us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amortized cost of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), with single maturity date and allocated without single maturity date, maturing in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Debt Securities, Available-for-Sale, Amortized Cost, Maturity, Allocated and Single Maturity Date, Year One",
"verboseLabel": "Due in one year or less"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesWithinOneYearAmortizedCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtMaturitiesWithinOneYearFairValue": {
"auth_ref": [
"r275",
"r277",
"r782"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": 1.0,
"parentTag": "us-gaap_AvailableForSaleSecuritiesDebtSecurities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), with single maturity date and allocated without single maturity date, maturing in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Debt Securities, Available-for-Sale, Fair Value, Maturity, Allocated and Single Maturity Date, Year One",
"verboseLabel": "Due in one year or less"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtMaturitiesWithinOneYearFairValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtSecurities": {
"auth_ref": [
"r268",
"r272",
"r300",
"r765"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": 3.0,
"parentTag": "us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Debt Securities, Available-for-sale",
"terseLabel": "Fair Value",
"totalLabel": "Total marketable debt securities"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtSecurities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofContractualMaturitiesDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtSecuritiesCurrent": {
"auth_ref": [
"r270",
"r300"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 3.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), classified as current.",
"label": "Debt Securities, Available-for-sale, Current",
"verboseLabel": "Marketable securities"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtSecuritiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtSecuritiesNoncurrent": {
"auth_ref": [
"r270",
"r300"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 2.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), classified as noncurrent.",
"label": "Debt Securities, Available-for-sale, Noncurrent",
"verboseLabel": "Marketable securities"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtSecuritiesNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AwardTypeAxis": {
"auth_ref": [
"r494",
"r521"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of award under share-based payment arrangement.",
"label": "Award Type [Axis]",
"terseLabel": "Award Type [Axis]"
}
}
},
"localname": "AwardTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BalanceSheetLocationAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by location on balance sheet (statement of financial position).",
"label": "Balance Sheet Location [Axis]",
"terseLabel": "Balance Sheet Location [Axis]"
}
}
},
"localname": "BalanceSheetLocationAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails",
"http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BalanceSheetLocationDomain": {
"auth_ref": [
"r639",
"r643"
],
"lang": {
"en-us": {
"role": {
"documentation": "Location in the balance sheet (statement of financial position).",
"label": "Balance Sheet Location [Domain]",
"terseLabel": "Balance Sheet Location [Domain]"
}
}
},
"localname": "BalanceSheetLocationDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails",
"http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_BuildingMember": {
"auth_ref": [
"r340"
],
"lang": {
"en-us": {
"role": {
"documentation": "Facility held for productive use including, but not limited to, office, production, storage and distribution facilities.",
"label": "Building [Member]",
"terseLabel": "Buildings"
}
}
},
"localname": "BuildingMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_BuildingsAndImprovementsGross": {
"auth_ref": [
"r27",
"r340"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 2.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated depreciation of building structures held for productive use including addition, improvement, or renovation to the structure, including, but not limited to, interior masonry, interior flooring, electrical, and plumbing.",
"label": "Buildings and Improvements, Gross",
"terseLabel": "Buildings"
}
}
},
"localname": "BuildingsAndImprovementsGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessAcquisitionAcquireeDomain": {
"auth_ref": [
"r480",
"r486"
],
"lang": {
"en-us": {
"role": {
"documentation": "Identification of the acquiree in a material business combination (or series of individually immaterial business combinations), which may include the name or other type of identification of the acquiree.",
"label": "Business Acquisition, Acquiree [Domain]",
"terseLabel": "Business Acquisition, Acquiree [Domain]"
}
}
},
"localname": "BusinessAcquisitionAcquireeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_BusinessAcquisitionAxis": {
"auth_ref": [
"r480",
"r486",
"r585",
"r586"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by business combination or series of individually immaterial business combinations.",
"label": "Business Acquisition [Axis]",
"terseLabel": "Business Acquisition [Axis]"
}
}
},
"localname": "BusinessAcquisitionAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BusinessAcquisitionContingentConsiderationLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Business Acquisition, Contingent Consideration [Line Items]",
"terseLabel": "Business Acquisition, Contingent Consideration [Line Items]"
}
}
},
"localname": "BusinessAcquisitionContingentConsiderationLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BusinessAcquisitionLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Business Acquisition [Line Items]",
"terseLabel": "Business Acquisition [Line Items]"
}
}
},
"localname": "BusinessAcquisitionLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BusinessAcquisitionSharePrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Price of a single share of a number of saleable stocks paid or offered to be paid in a business combination.",
"label": "Business Acquisition, Share Price",
"terseLabel": "Price per share"
}
}
},
"localname": "BusinessAcquisitionSharePrice",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationArrangementsChangeInAmountOfContingentConsiderationAsset1": {
"auth_ref": [
"r150",
"r598"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in the value of right to a contingent consideration asset.",
"label": "Business Combination, Contingent Consideration Arrangements, Change in Amount of Contingent Consideration, Asset",
"terseLabel": "Reduction in contingent consideration"
}
}
},
"localname": "BusinessCombinationContingentConsiderationArrangementsChangeInAmountOfContingentConsiderationAsset1",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationArrangementsRangeOfOutcomesValueHigh": {
"auth_ref": [
"r596"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "For contingent consideration arrangements recognized in connection with a business combination, this element represents an estimate of the high-end of the potential range (undiscounted) of the consideration which may be paid.",
"label": "Business Combination, Contingent Consideration Arrangements, Range of Outcomes, Value, High",
"terseLabel": "Maximum contingent consideration in the form of development and approval milestones"
}
}
},
"localname": "BusinessCombinationContingentConsiderationArrangementsRangeOfOutcomesValueHigh",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationLiability": {
"auth_ref": [
"r591",
"r592",
"r595"
],
"calculation": {
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails": {
"order": 1.0,
"parentTag": "us-gaap_LiabilitiesFairValueDisclosure",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liability recognized arising from contingent consideration in a business combination.",
"label": "Business Combination, Contingent Consideration, Liability",
"periodEndLabel": "Fair value, end of year",
"periodStartLabel": "Fair value, beginning of year",
"terseLabel": "Fair Value",
"verboseLabel": "Contingent consideration obligations"
}
}
},
"localname": "BusinessCombinationContingentConsiderationLiability",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationLiabilityCurrent": {
"auth_ref": [
"r591",
"r593"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": 5.0,
"parentTag": "us-gaap_OtherLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liability recognized arising from contingent consideration in a business combination, expected to be settled within one year or the normal operating cycle, if longer.",
"label": "Business Combination, Contingent Consideration, Liability, Current",
"terseLabel": "Business Combination, Contingent Consideration, Liability, Current"
}
}
},
"localname": "BusinessCombinationContingentConsiderationLiabilityCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationLiabilityMeasurementInput": {
"auth_ref": [
"r668"
],
"lang": {
"en-us": {
"role": {
"documentation": "Value of input used to measure contingent consideration liability from business combination.",
"label": "Business Combination, Contingent Consideration, Liability, Measurement Input",
"terseLabel": "Unobservable Input"
}
}
},
"localname": "BusinessCombinationContingentConsiderationLiabilityMeasurementInput",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "decimalItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationLiabilityNoncurrent": {
"auth_ref": [
"r591",
"r593"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liability recognized arising from contingent consideration in a business combination, expected to be settled beyond one year or the normal operating cycle, if longer.",
"label": "Business Combination, Contingent Consideration, Liability, Noncurrent",
"terseLabel": "Contingent consideration obligations"
}
}
},
"localname": "BusinessCombinationContingentConsiderationLiabilityNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationDisclosureTextBlock": {
"auth_ref": [
"r601"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for a business combination (or series of individually immaterial business combinations) completed during the period, including background, timing, and recognized assets and liabilities. The disclosure may include leverage buyout transactions (as applicable).",
"label": "Business Combination Disclosure [Text Block]",
"verboseLabel": "Acquisitions"
}
}
},
"localname": "BusinessCombinationDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Acquisitions"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_BusinessCombinationRecognizedIdentifiableAssetsAcquiredAndLiabilitiesAssumedIndefiniteLivedIntangibleAssets": {
"auth_ref": [
"r587"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of assets, excluding financial assets and goodwill, that lack physical substance, having a projected indefinite period of benefit, acquired at the acquisition date.",
"label": "Business Combination, Recognized Identifiable Assets Acquired and Liabilities Assumed, Indefinite-Lived Intangible Assets",
"terseLabel": "Intangible assets"
}
}
},
"localname": "BusinessCombinationRecognizedIdentifiableAssetsAcquiredAndLiabilitiesAssumedIndefiniteLivedIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationSeparatelyRecognizedTransactionsAxis": {
"auth_ref": [
"r583"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosures related to transactions that are recognized separately from the acquisition of assets and assumptions of liabilities in the business combination by type of transaction.",
"label": "Business Combination, Separately Recognized Transactions [Axis]",
"terseLabel": "Business Combination, Separately Recognized Transactions [Axis]"
}
}
},
"localname": "BusinessCombinationSeparatelyRecognizedTransactionsAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BusinessCombinationSeparatelyRecognizedTransactionsDomain": {
"auth_ref": [
"r583"
],
"lang": {
"en-us": {
"role": {
"documentation": "Type of transaction that is recognized separately from the acquisition of assets and assumptions of liabilities in a business combination by transaction.",
"label": "Business Combination, Separately Recognized Transactions [Domain]",
"terseLabel": "Business Combination, Separately Recognized Transactions [Domain]"
}
}
},
"localname": "BusinessCombinationSeparatelyRecognizedTransactionsDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_BusinessCombinationSeparatelyRecognizedTransactionsExpensesAndLossesRecognized": {
"auth_ref": [
"r584"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The expenses and losses recorded for each transaction with the acquiree that was recognized separately from the acquisition of assets and assumptions of liabilities in the business combination.",
"label": "Business Combination, Separately Recognized Transactions, Expenses and Losses Recognized",
"terseLabel": "Fair-value post-combination equity compensation",
"verboseLabel": "NST stock options"
}
}
},
"localname": "BusinessCombinationSeparatelyRecognizedTransactionsExpensesAndLossesRecognized",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Business Combinations [Abstract]",
"terseLabel": "Business Combinations [Abstract]"
}
}
},
"localname": "BusinessCombinationsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_BusinessCombinationsPolicy": {
"auth_ref": [
"r158",
"r582"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for completed business combinations (purchase method, acquisition method or combination of entities under common control). This accounting policy may include a general discussion of the purchase method or acquisition method of accounting (including for example, the treatment accorded contingent consideration, the identification of assets and liabilities, the purchase price allocation process, how the fair values of acquired assets and liabilities are determined) and the entity's specific application thereof. An entity that acquires another entity in a leveraged buyout transaction generally discloses the accounting policy followed by the acquiring entity in determining the basis used to value its interest in the acquired entity, and the rationale for that accounting policy.",
"label": "Business Combinations Policy [Policy Text Block]",
"terseLabel": "Contingent Consideration"
}
}
},
"localname": "BusinessCombinationsPolicy",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CashAndCashEquivalentsFairValueDisclosure": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Cash and Cash Equivalents, Fair Value Disclosure",
"verboseLabel": "Cash equivalents"
}
}
},
"localname": "CashAndCashEquivalentsFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashAndCashEquivalentsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Cash and Cash Equivalents [Line Items]",
"terseLabel": "Cash and Cash Equivalents [Line Items]"
}
}
},
"localname": "CashAndCashEquivalentsLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CashAndCashEquivalentsPolicyTextBlock": {
"auth_ref": [
"r33",
"r154",
"r158"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for cash and cash equivalents, including the policy for determining which items are treated as cash equivalents. Other information that may be disclosed includes (1) the nature of any restrictions on the entity's use of its cash and cash equivalents, (2) whether the entity's cash and cash equivalents are insured or expose the entity to credit risk, (3) the classification of any negative balance accounts (overdrafts), and (4) the carrying basis of cash equivalents (for example, at cost) and whether the carrying amount of cash equivalents approximates fair value.",
"label": "Cash and Cash Equivalents, Policy [Policy Text Block]",
"terseLabel": "Cash and Cash Equivalents"
}
}
},
"localname": "CashAndCashEquivalentsPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CashCashEquivalentsAndMarketableSecuritiesTextBlock": {
"auth_ref": [
"r287"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure of cash, cash equivalents, and debt and equity securities, including any unrealized or realized gain (loss).",
"label": "Cash, Cash Equivalents, and Marketable Securities [Text Block]",
"verboseLabel": "Financial Instruments"
}
}
},
"localname": "CashCashEquivalentsAndMarketableSecuritiesTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstruments"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents": {
"auth_ref": [
"r146",
"r153",
"r156"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 2.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash and cash equivalents, and cash and cash equivalents restricted to withdrawal or usage. Excludes amount for disposal group and discontinued operations. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Cash, Cash Equivalents, Restricted Cash and Restricted Cash Equivalents",
"periodEndLabel": "Cash and cash equivalents, end of the year",
"periodStartLabel": "Cash and cash equivalents, beginning of the year",
"verboseLabel": "Cash and cash equivalents"
}
}
},
"localname": "CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseExcludingExchangeRateEffect": {
"auth_ref": [
"r146",
"r685"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in cash and cash equivalents, and cash and cash equivalents restricted to withdrawal or usage; excluding effect from exchange rate change. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Cash, Cash Equivalents, Restricted Cash and Restricted Cash Equivalents, Period Increase (Decrease), Excluding Exchange Rate Effect",
"totalLabel": "Net increase (decrease) in cash and cash equivalents"
}
}
},
"localname": "CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseExcludingExchangeRateEffect",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashEquivalentsAtCarryingValue": {
"auth_ref": [
"r57"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation.",
"label": "Cash Equivalents, at Carrying Value",
"verboseLabel": "Cash equivalents"
}
}
},
"localname": "CashEquivalentsAtCarryingValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashFlowHedgingMember": {
"auth_ref": [
"r635"
],
"lang": {
"en-us": {
"role": {
"documentation": "Hedge of the exposure to variability in the cash flows of a recognized asset or liability, or of a forecasted transaction, that is attributable to a particular risk.",
"label": "Cash Flow Hedging [Member]",
"terseLabel": "Cash flow hedge"
}
}
},
"localname": "CashFlowHedgingMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CashProceedsReceivedAndTaxBenefitFromShareBasedPaymentAwardsTableTextBlock": {
"auth_ref": [
"r522"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of cash received from exercise of stock options and similar instruments granted under share-based payment arrangements and tax benefit from exercise of stock options.",
"label": "Cash Proceeds Received and Tax Benefit from Share-based Payment Awards [Table Text Block]",
"terseLabel": "Tax benefit and cash received from stock option exercises"
}
}
},
"localname": "CashProceedsReceivedAndTaxBenefitFromShareBasedPaymentAwardsTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ClassOfStockDomain": {
"auth_ref": [
"r161",
"r163",
"r197",
"r201",
"r202",
"r204",
"r206",
"r217",
"r218",
"r219",
"r293",
"r680"
],
"lang": {
"en-us": {
"role": {
"documentation": "Share of stock differentiated by the voting rights the holder receives. Examples include, but are not limited to, common stock, redeemable preferred stock, nonredeemable preferred stock, and convertible stock.",
"label": "Class of Stock [Domain]",
"terseLabel": "Class of Stock [Domain]"
}
}
},
"localname": "ClassOfStockDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ClassOfStockLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Class of Stock [Line Items]",
"terseLabel": "Class of Stock [Line Items]"
}
}
},
"localname": "ClassOfStockLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CollaborativeArrangementDisclosureTextBlock": {
"auth_ref": [
"r602",
"r603",
"r605"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for collaborative arrangements in which the entity is a participant, including a) information about the nature and purpose of such arrangements; b) its rights and obligations thereunder; c) the accounting policy for collaborative arrangements; and d) the income statement classification and amounts attributable to transactions arising from the collaborative arrangement between participants.",
"label": "Collaborative Arrangement Disclosure [Text Block]",
"verboseLabel": "Collaborative and Other Relationships"
}
}
},
"localname": "CollaborativeArrangementDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationships"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CollaborativeArrangementMember": {
"auth_ref": [
"r604"
],
"lang": {
"en-us": {
"role": {
"documentation": "Contractual arrangement that involves two or more parties that both: (i) actively participate in a joint operating activity and (ii) are exposed to significant risks and rewards that depend on the commercial success of the joint operating activity.",
"label": "Collaborative Arrangement [Member]",
"terseLabel": "Collaborative Arrangement"
}
}
},
"localname": "CollaborativeArrangementMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CollaborativeArrangementsAndNoncollaborativeArrangementTransactionsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Line Items]",
"terseLabel": "Collaborative arrangements and non-collaborative arrangement transactions"
}
}
},
"localname": "CollaborativeArrangementsAndNoncollaborativeArrangementTransactionsLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CommercialPaperMember": {
"auth_ref": [
"r364"
],
"lang": {
"en-us": {
"role": {
"documentation": "Unsecured promissory note (generally negotiable) that provides institutions with short-term funds.",
"label": "Commercial Paper [Member]",
"verboseLabel": "Commercial paper"
}
}
},
"localname": "CommercialPaperMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CommitmentsAndContingencies": {
"auth_ref": [
"r74",
"r357",
"r769",
"r797"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 3.0,
"parentTag": "us-gaap_LiabilitiesAndStockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Represents the caption on the face of the balance sheet to indicate that the entity has entered into (1) purchase or supply arrangements that will require expending a portion of its resources to meet the terms thereof, and (2) is exposed to potential losses or, less frequently, gains, arising from (a) possible claims against a company's resources due to future performance under contract terms, and (b) possible losses or likely gains from uncertainties that will ultimately be resolved when one or more future events that are deemed likely to occur do occur or fail to occur.",
"label": "Commitments and Contingencies",
"verboseLabel": "Commitments, contingencies and guarantees (Notes 21 and 22)"
}
}
},
"localname": "CommitmentsAndContingencies",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CommitmentsAndContingenciesDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Commitments and Contingencies Disclosure [Abstract]",
"terseLabel": "Commitments and Contingencies Disclosure [Abstract]"
}
}
},
"localname": "CommitmentsAndContingenciesDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_CommitmentsAndContingenciesDisclosureTextBlock": {
"auth_ref": [
"r354",
"r355",
"r356",
"r361"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for commitments and contingencies.",
"label": "Commitments and Contingencies Disclosure [Text Block]",
"verboseLabel": "Commitments and Contingencies"
}
}
},
"localname": "CommitmentsAndContingenciesDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingencies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CommitmentsAndContingenciesPolicyTextBlock": {
"auth_ref": [
"r158",
"r362",
"r841",
"r842"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for commitments and contingencies, which may include policies for recognizing and measuring loss and gain contingencies.",
"label": "Commitments and Contingencies, Policy [Policy Text Block]",
"terseLabel": "Contingencies"
}
}
},
"localname": "CommitmentsAndContingenciesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CommonStockMember": {
"auth_ref": [
"r180",
"r181"
],
"lang": {
"en-us": {
"role": {
"documentation": "Stock that is subordinate to all other stock of the issuer.",
"label": "Common Stock [Member]",
"verboseLabel": "Common stock"
}
}
},
"localname": "CommonStockMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "domainItemType"
},
"us-gaap_CommonStockParOrStatedValuePerShare": {
"auth_ref": [
"r43"
],
"lang": {
"en-us": {
"role": {
"documentation": "Face amount or stated value per share of common stock.",
"label": "Common Stock, Par or Stated Value Per Share",
"verboseLabel": "Common stock, par value (in dollars per share)"
}
}
},
"localname": "CommonStockParOrStatedValuePerShare",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheetsParenthetical"
],
"xbrltype": "perShareItemType"
},
"us-gaap_CommonStockSharesAuthorized": {
"auth_ref": [
"r43"
],
"lang": {
"en-us": {
"role": {
"documentation": "The maximum number of common shares permitted to be issued by an entity's charter and bylaws.",
"label": "Common Stock, Shares Authorized",
"terseLabel": "Common stock authorized (in shares)"
}
}
},
"localname": "CommonStockSharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquitySummaryofCommonStockDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CommonStockSharesIssued": {
"auth_ref": [
"r43"
],
"lang": {
"en-us": {
"role": {
"documentation": "Total number of common shares of an entity that have been sold or granted to shareholders (includes common shares that were issued, repurchased and remain in the treasury). These shares represent capital invested by the firm's shareholders and owners, and may be all or only a portion of the number of shares authorized. Shares issued include shares outstanding and shares held in the treasury.",
"label": "Common Stock, Shares, Issued",
"terseLabel": "Common stock issued (in shares)"
}
}
},
"localname": "CommonStockSharesIssued",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquitySummaryofCommonStockDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CommonStockSharesOutstanding": {
"auth_ref": [
"r43",
"r372"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares of common stock outstanding. Common stock represent the ownership interest in a corporation.",
"label": "Common Stock, Shares, Outstanding",
"terseLabel": "Common stock outstanding (in shares)"
}
}
},
"localname": "CommonStockSharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquitySummaryofCommonStockDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CommonStockValue": {
"auth_ref": [
"r43"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 2.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate par or stated value of issued nonredeemable common stock (or common stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable common shares, par value and other disclosure concepts are in another section within stockholders' equity.",
"label": "Common Stock, Value, Issued",
"verboseLabel": "Common stock, par value $0.0005 per share"
}
}
},
"localname": "CommonStockValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CompensationAndRetirementDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Retirement Benefits [Abstract]",
"terseLabel": "Retirement Benefits [Abstract]"
}
}
},
"localname": "CompensationAndRetirementDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_CompensationRelatedCostsPolicyTextBlock": {
"auth_ref": [
"r157",
"r499"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for salaries, bonuses, incentive awards, postretirement and postemployment benefits granted to employees, including equity-based arrangements; discloses methodologies for measurement, and the bases for recognizing related assets and liabilities and recognizing and reporting compensation expense.",
"label": "Compensation Related Costs, Policy [Policy Text Block]",
"terseLabel": "Accounting for Share-Based Compensation"
}
}
},
"localname": "CompensationRelatedCostsPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ComponentsOfDeferredTaxAssetsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Components of Deferred Tax Assets [Abstract]",
"terseLabel": "Deferred tax assets:"
}
}
},
"localname": "ComponentsOfDeferredTaxAssetsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ComponentsOfDeferredTaxLiabilitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Components of Deferred Tax Liabilities [Abstract]",
"verboseLabel": "Deferred tax liabilities:"
}
}
},
"localname": "ComponentsOfDeferredTaxLiabilitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ComprehensiveIncomeNetOfTax": {
"auth_ref": [
"r103",
"r105",
"r106",
"r116",
"r775",
"r806"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome_1": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of increase (decrease) in equity from transactions and other events and circumstances from net income and other comprehensive income, attributable to parent entity. Excludes changes in equity resulting from investments by owners and distributions to owners.",
"label": "Comprehensive Income (Loss), Net of Tax, Attributable to Parent",
"totalLabel": "Comprehensive income attributable to Biogen Inc."
}
}
},
"localname": "ComprehensiveIncomeNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ComprehensiveIncomeNetOfTaxAttributableToNoncontrollingInterest": {
"auth_ref": [
"r103",
"r105",
"r115",
"r609",
"r610",
"r628",
"r774",
"r805"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome_1": {
"order": 1.0,
"parentTag": "us-gaap_ComprehensiveIncomeNetOfTax",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of increase (decrease) in equity from transactions and other events and circumstances from net income (loss) and other comprehensive income (loss), attributable to noncontrolling interests. Excludes changes in equity resulting from investments by owners and distributions to owners.",
"label": "Comprehensive Income (Loss), Net of Tax, Attributable to Noncontrolling Interest",
"verboseLabel": "Comprehensive income (loss) attributable to noncontrolling interests, net of tax"
}
}
},
"localname": "ComprehensiveIncomeNetOfTaxAttributableToNoncontrollingInterest",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ComprehensiveIncomeNetOfTaxIncludingPortionAttributableToNoncontrollingInterest": {
"auth_ref": [
"r103",
"r105",
"r114",
"r608",
"r628",
"r773",
"r804"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome_1": {
"order": 2.0,
"parentTag": "us-gaap_ComprehensiveIncomeNetOfTax",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of increase (decrease) in equity from transactions and other events and circumstances from net income and other comprehensive income. Excludes changes in equity resulting from investments by owners and distributions to owners.",
"label": "Comprehensive Income (Loss), Net of Tax, Including Portion Attributable to Noncontrolling Interest",
"terseLabel": "Comprehensive income"
}
}
},
"localname": "ComprehensiveIncomeNetOfTaxIncludingPortionAttributableToNoncontrollingInterest",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ConcentrationRiskCreditRisk": {
"auth_ref": [
"r222",
"r786"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for credit risk.",
"label": "Concentration Risk, Credit Risk, Policy [Policy Text Block]",
"terseLabel": "Concentration of Credit Risk"
}
}
},
"localname": "ConcentrationRiskCreditRisk",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ConsolidationPolicyTextBlock": {
"auth_ref": [
"r158",
"r613"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy regarding (1) the principles it follows in consolidating or combining the separate financial statements, including the principles followed in determining the inclusion or exclusion of subsidiaries or other entities in the consolidated or combined financial statements and (2) its treatment of interests (for example, common stock, a partnership interest or other means of exerting influence) in other entities, for example consolidation or use of the equity or cost methods of accounting. The accounting policy may also address the accounting treatment for intercompany accounts and transactions, noncontrolling interest, and the income statement treatment in consolidation for issuances of stock by a subsidiary.",
"label": "Consolidation, Policy [Policy Text Block]",
"terseLabel": "Consolidation"
}
}
},
"localname": "ConsolidationPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ConstructionInProgressGross": {
"auth_ref": [
"r340"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 7.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of structure or a modification to a structure under construction. Includes recently completed structures or modifications to structures that have not been placed into service.",
"label": "Construction in Progress, Gross",
"terseLabel": "Construction in progress"
}
}
},
"localname": "ConstructionInProgressGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ConstructionPayableCurrentAndNoncurrent": {
"auth_ref": [
"r768",
"r798"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of obligations incurred and payable for the acquisition of merchandise, materials, supplies and services pertaining to construction projects such as a housing development or factory expansion not classified as trade payables.",
"label": "Construction Payable",
"terseLabel": "Construction in progress"
}
}
},
"localname": "ConstructionPayableCurrentAndNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ContingentConsiderationByTypeAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of contingent consideration.",
"label": "Contingent Consideration by Type [Axis]",
"terseLabel": "Contingent Consideration by Type [Axis]"
}
}
},
"localname": "ContingentConsiderationByTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ContingentConsiderationTypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Description of contingent payment arrangement.",
"label": "Contingent Consideration Type [Domain]",
"terseLabel": "Contingent Consideration Type [Domain]"
}
}
},
"localname": "ContingentConsiderationTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ContractualObligation": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of contractual obligation, including but not limited to, long-term debt, capital lease obligations, operating lease obligations, purchase obligations, and other commitments.",
"label": "Contractual Obligation",
"terseLabel": "Contractual obligation"
}
}
},
"localname": "ContractualObligation",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CorporateDebtSecuritiesMember": {
"auth_ref": [
"r428",
"r474",
"r824"
],
"lang": {
"en-us": {
"role": {
"documentation": "Debt securities issued by domestic or foreign corporate business, banks and other entities with a promise of repayment.",
"label": "Corporate Debt Securities [Member]",
"verboseLabel": "Corporate debt securities"
}
}
},
"localname": "CorporateDebtSecuritiesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CostOfGoodsAndServicesSold": {
"auth_ref": [
"r121",
"r754"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 1.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate costs related to goods produced and sold and services rendered by an entity during the reporting period. This excludes costs incurred during the reporting period related to financial services rendered and other revenue generating activities.",
"label": "Cost of Goods and Services Sold",
"terseLabel": "Cost of sales, excluding amortization and impairment of acquired intangible assets"
}
}
},
"localname": "CostOfGoodsAndServicesSold",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CostsAndExpenses": {
"auth_ref": [
"r118"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 2.0,
"parentTag": "us-gaap_OperatingIncomeLoss",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Total costs of sales and operating expenses for the period.",
"label": "Costs and Expenses",
"totalLabel": "Total cost and expenses"
}
}
},
"localname": "CostsAndExpenses",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CostsAndExpensesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Costs and Expenses [Abstract]",
"verboseLabel": "Cost and expenses:"
}
}
},
"localname": "CostsAndExpensesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "stringItemType"
},
"us-gaap_CreditFacilityAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of credit facility. Credit facilities provide capital to borrowers without the need to structure a loan for each borrowing.",
"label": "Credit Facility [Axis]",
"terseLabel": "Credit Facility [Axis]"
}
}
},
"localname": "CreditFacilityAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CreditFacilityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Type of credit facility. Credit facilities provide capital to borrowers without the need to structure a loan for each borrowing.",
"label": "Credit Facility [Domain]",
"terseLabel": "Credit Facility [Domain]"
}
}
},
"localname": "CreditFacilityDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CurrentFederalTaxExpenseBenefit": {
"auth_ref": [
"r165",
"r562",
"r571"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 1.0,
"parentTag": "us-gaap_CurrentIncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of current federal tax expense (benefit) pertaining to income (loss) from continuing operations.",
"label": "Current Federal Tax Expense (Benefit)",
"terseLabel": "Federal"
}
}
},
"localname": "CurrentFederalTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CurrentForeignTaxExpenseBenefit": {
"auth_ref": [
"r168",
"r562"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 3.0,
"parentTag": "us-gaap_CurrentIncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of current foreign income tax expense (benefit) pertaining to income (loss) from continuing operations.",
"label": "Current Foreign Tax Expense (Benefit)",
"terseLabel": "Foreign"
}
}
},
"localname": "CurrentForeignTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CurrentIncomeTaxExpenseBenefit": {
"auth_ref": [
"r165",
"r562",
"r571",
"r573"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 1.0,
"parentTag": "us-gaap_IncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of current income tax expense (benefit) pertaining to taxable income (loss) from continuing operations.",
"label": "Current Income Tax Expense (Benefit)",
"totalLabel": "Total"
}
}
},
"localname": "CurrentIncomeTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CurrentIncomeTaxExpenseBenefitContinuingOperationsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Current Income Tax Expense (Benefit), Continuing Operations [Abstract]",
"terseLabel": "Current:"
}
}
},
"localname": "CurrentIncomeTaxExpenseBenefitContinuingOperationsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CurrentStateAndLocalTaxExpenseBenefit": {
"auth_ref": [
"r165",
"r562",
"r571"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 2.0,
"parentTag": "us-gaap_CurrentIncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of current state and local tax expense (benefit) pertaining to income (loss) from continuing operations.",
"label": "Current State and Local Tax Expense (Benefit)",
"terseLabel": "State"
}
}
},
"localname": "CurrentStateAndLocalTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DebtCurrent": {
"auth_ref": [
"r66"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 4.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of short-term debt and current maturity of long-term debt and capital lease obligations due within one year or the normal operating cycle, if longer.",
"label": "Debt, Current",
"terseLabel": "Current portion of notes payable",
"verboseLabel": "Current portion of notes payable"
}
}
},
"localname": "DebtCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DebtDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Debt Disclosure [Abstract]",
"terseLabel": "Debt Disclosure [Abstract]"
}
}
},
"localname": "DebtDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_DebtDisclosureTextBlock": {
"auth_ref": [
"r368"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for information about short-term and long-term debt arrangements, which includes amounts of borrowings under each line of credit, note payable, commercial paper issue, bonds indenture, debenture issue, own-share lending arrangements and any other contractual agreement to repay funds, and about the underlying arrangements, rationale for a classification as long-term, including repayment terms, interest rates, collateral provided, restrictions on use of assets and activities, whether or not in compliance with debt covenants, and other matters important to users of the financial statements, such as the effects of refinancing and noncompliance with debt covenants.",
"label": "Debt Disclosure [Text Block]",
"verboseLabel": "Indebtedness"
}
}
},
"localname": "DebtDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Indebtedness"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DebtInstrumentAxis": {
"auth_ref": [
"r36",
"r37",
"r38",
"r759",
"r761",
"r787"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of debt instrument, including, but not limited to, draws against credit facilities.",
"label": "Debt Instrument [Axis]",
"terseLabel": "Debt Instrument [Axis]"
}
}
},
"localname": "DebtInstrumentAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DebtInstrumentFaceAmount": {
"auth_ref": [
"r697",
"r699"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Face (par) amount of debt instrument at time of issuance.",
"label": "Debt Instrument, Face Amount",
"terseLabel": "Aggregate principal amount",
"verboseLabel": "Senior notes aggregate principal amount"
}
}
},
"localname": "DebtInstrumentFaceAmount",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DebtInstrumentInterestRateStatedPercentage": {
"auth_ref": [
"r71"
],
"lang": {
"en-us": {
"role": {
"documentation": "Contractual interest rate for funds borrowed, under the debt agreement.",
"label": "Debt Instrument, Interest Rate, Stated Percentage",
"terseLabel": "Interest rate on senior notes",
"verboseLabel": "Interest rate on senior notes"
}
}
},
"localname": "DebtInstrumentInterestRateStatedPercentage",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_DebtInstrumentLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Debt Instrument [Line Items]",
"terseLabel": "Debt Instrument"
}
}
},
"localname": "DebtInstrumentLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DebtInstrumentNameDomain": {
"auth_ref": [
"r72"
],
"lang": {
"en-us": {
"role": {
"documentation": "The name for the particular debt instrument or borrowing that distinguishes it from other debt instruments or borrowings, including draws against credit facilities.",
"label": "Debt Instrument, Name [Domain]",
"terseLabel": "Debt Instrument, Name [Domain]"
}
}
},
"localname": "DebtInstrumentNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DebtInstrumentRedemptionPricePercentage": {
"auth_ref": [
"r784"
],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage price of original principal amount of debt at which debt can be redeemed by the issuer.",
"label": "Debt Instrument, Redemption Price, Percentage",
"terseLabel": "Redemptions percentage of 2015 notes"
}
}
},
"localname": "DebtInstrumentRedemptionPricePercentage",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_DebtInstrumentTable": {
"auth_ref": [
"r72",
"r166",
"r373",
"r376",
"r377",
"r378",
"r696",
"r697",
"r699",
"r785"
],
"lang": {
"en-us": {
"role": {
"documentation": "A table or schedule providing information pertaining to long-term debt instruments or arrangements, including identification, terms, features, collateral requirements and other information necessary to a fair presentation. These are debt arrangements that originally required repayment more than twelve months after issuance or greater than the normal operating cycle of the company, if longer.",
"label": "Schedule of Long-term Debt Instruments [Table]",
"terseLabel": "Schedule of Long-term Debt Instruments [Table]"
}
}
},
"localname": "DebtInstrumentTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DebtInstrumentTerm": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Period of time between issuance and maturity of debt instrument, in PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Debt Instrument, Term",
"terseLabel": "Debt instrument, term"
}
}
},
"localname": "DebtInstrumentTerm",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_DebtSecuritiesAvailableForSaleRealizedGain": {
"auth_ref": [
"r285"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of realized gain on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Debt Securities, Available-for-sale, Realized Gain",
"verboseLabel": "Realized gains"
}
}
},
"localname": "DebtSecuritiesAvailableForSaleRealizedGain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsProceedsfromMarketableDebtSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DebtSecuritiesAvailableForSaleRealizedLoss": {
"auth_ref": [
"r285"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of realized loss on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Debt Securities, Available-for-sale, Realized Loss",
"terseLabel": "Realized losses"
}
}
},
"localname": "DebtSecuritiesAvailableForSaleRealizedLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsProceedsfromMarketableDebtSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DebtSecuritiesMember": {
"auth_ref": [
"r287"
],
"lang": {
"en-us": {
"role": {
"documentation": "Debt instrument issued by corporations, governments and governmental agencies, municipalities, and other institutions.",
"label": "Debt Securities [Member]",
"verboseLabel": "Short-term debt securities"
}
}
},
"localname": "DebtSecuritiesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DecreaseInUnrecognizedTaxBenefitsIsReasonablyPossible": {
"auth_ref": [
"r543"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of decrease reasonably possible in the next twelve months for the unrecognized tax benefit.",
"label": "Decrease in Unrecognized Tax Benefits is Reasonably Possible",
"terseLabel": "Decrease in unrecognized tax benefits is reasonably possible"
}
}
},
"localname": "DecreaseInUnrecognizedTaxBenefitsIsReasonablyPossible",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredCompensationLiabilityClassifiedNoncurrent": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate carrying value as of the balance sheet date of the liabilities for all deferred compensation arrangements payable beyond one year (or the operating cycle, if longer).",
"label": "Deferred Compensation Liability, Classified, Noncurrent",
"terseLabel": "Deferred compensation liability"
}
}
},
"localname": "DeferredCompensationLiabilityClassifiedNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredFederalIncomeTaxExpenseBenefit": {
"auth_ref": [
"r165",
"r563",
"r571"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 1.0,
"parentTag": "us-gaap_DeferredIncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred federal income tax expense (benefit) pertaining to income (loss) from continuing operations.",
"label": "Deferred Federal Income Tax Expense (Benefit)",
"terseLabel": "Federal"
}
}
},
"localname": "DeferredFederalIncomeTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredForeignIncomeTaxExpenseBenefit": {
"auth_ref": [
"r165",
"r563",
"r571"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 3.0,
"parentTag": "us-gaap_DeferredIncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred foreign income tax expense (benefit) pertaining to income (loss) from continuing operations.",
"label": "Deferred Foreign Income Tax Expense (Benefit)",
"terseLabel": "Foreign"
}
}
},
"localname": "DeferredForeignIncomeTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredIncomeTaxAssetsNet": {
"auth_ref": [
"r537",
"r538"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 7.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after allocation of valuation allowances and deferred tax liability, of deferred tax asset attributable to deductible differences and carryforwards, with jurisdictional netting.",
"label": "Deferred Income Tax Assets, Net",
"terseLabel": "Deferred tax asset"
}
}
},
"localname": "DeferredIncomeTaxAssetsNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredIncomeTaxChargesMember": {
"auth_ref": [
"r857",
"r858",
"r859",
"r860"
],
"lang": {
"en-us": {
"role": {
"documentation": "Rate action of a regulator resulting in capitalization or accrual of income taxes.",
"label": "Deferred Income Tax Charge [Member]",
"terseLabel": "Deferred Income Tax Charge"
}
}
},
"localname": "DeferredIncomeTaxChargesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DeferredIncomeTaxExpenseBenefit": {
"auth_ref": [
"r151",
"r165",
"r563",
"r571",
"r572",
"r573"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 11.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
},
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 2.0,
"parentTag": "us-gaap_IncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred income tax expense (benefit) pertaining to income (loss) from continuing operations.",
"label": "Deferred Income Tax Expense (Benefit)",
"totalLabel": "Total",
"verboseLabel": "Deferred income taxes"
}
}
},
"localname": "DeferredIncomeTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredIncomeTaxExpenseBenefitContinuingOperationsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Deferred Income Tax Expense (Benefit), Continuing Operations [Abstract]",
"terseLabel": "Deferred:"
}
}
},
"localname": "DeferredIncomeTaxExpenseBenefitContinuingOperationsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DeferredIncomeTaxLiabilitiesNet": {
"auth_ref": [
"r537",
"r538"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 3.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after deferred tax asset, of deferred tax liability attributable to taxable differences with jurisdictional netting.",
"label": "Deferred Income Tax Liabilities, Net",
"verboseLabel": "Deferred tax liability"
}
}
},
"localname": "DeferredIncomeTaxLiabilitiesNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredStateAndLocalIncomeTaxExpenseBenefit": {
"auth_ref": [
"r165",
"r563",
"r571"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 2.0,
"parentTag": "us-gaap_DeferredIncomeTaxExpenseBenefit",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred state and local tax expense (benefit) pertaining to income (loss) from continuing operations.",
"label": "Deferred State and Local Income Tax Expense (Benefit)",
"terseLabel": "State"
}
}
},
"localname": "DeferredStateAndLocalIncomeTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsGoodwillAndIntangibleAssets": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 3.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences from intangible assets including goodwill.",
"label": "Deferred Tax Assets, Goodwill and Intangible Assets",
"terseLabel": "Intangibles, net"
}
}
},
"localname": "DeferredTaxAssetsGoodwillAndIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsInventory": {
"auth_ref": [
"r560",
"r561"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 2.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences from inventory.",
"label": "Deferred Tax Assets, Inventory",
"terseLabel": "Inventory, other reserves and accruals"
}
}
},
"localname": "DeferredTaxAssetsInventory",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsNet": {
"auth_ref": [
"r555"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences and carryforwards.",
"label": "Deferred Tax Assets, Net of Valuation Allowance",
"totalLabel": "Total deferred tax assets"
}
}
},
"localname": "DeferredTaxAssetsNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsOperatingLossCarryforwards": {
"auth_ref": [
"r560",
"r561"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 4.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible operating loss carryforwards.",
"label": "Deferred Tax Assets, Operating Loss Carryforwards",
"terseLabel": "Net operating loss"
}
}
},
"localname": "DeferredTaxAssetsOperatingLossCarryforwards",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsOperatingLossCarryforwardsSubjectToExpiration": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible operating loss carryforwards that are subject to expiration dates.",
"label": "Deferred Tax Assets, Operating Loss Carryforwards, Subject to Expiration",
"terseLabel": "Operating loss carryforward, subject to expiration"
}
}
},
"localname": "DeferredTaxAssetsOperatingLossCarryforwardsSubjectToExpiration",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsOther": {
"auth_ref": [
"r560",
"r561"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 6.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before allocation of valuation allowance, of deferred tax asset attributable to deductible temporary differences, classified as other.",
"label": "Deferred Tax Assets, Other",
"terseLabel": "Other"
}
}
},
"localname": "DeferredTaxAssetsOther",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsTaxCreditCarryforwards": {
"auth_ref": [
"r558",
"r560",
"r561"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 1.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before allocation of a valuation allowances, of deferred tax assets attributable to deductible tax credit carryforwards including, but not limited to, research, foreign, general business, alternative minimum tax, and other deductible tax credit carryforwards.",
"label": "Deferred Tax Assets, Tax Credit Carryforwards",
"terseLabel": "Tax credits"
}
}
},
"localname": "DeferredTaxAssetsTaxCreditCarryforwards",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsTaxDeferredExpenseCompensationAndBenefitsShareBasedCompensationCost": {
"auth_ref": [
"r560",
"r561"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 5.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences from share-based compensation.",
"label": "Deferred Tax Assets, Tax Deferred Expense, Compensation and Benefits, Share-based Compensation Cost",
"terseLabel": "Share-based compensation"
}
}
},
"localname": "DeferredTaxAssetsTaxDeferredExpenseCompensationAndBenefitsShareBasedCompensationCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsValuationAllowance": {
"auth_ref": [
"r554"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 7.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax assets for which it is more likely than not that a tax benefit will not be realized.",
"label": "Deferred Tax Assets, Valuation Allowance",
"negatedTerseLabel": "Valuation allowance",
"verboseLabel": "Valuation allowance"
}
}
},
"localname": "DeferredTaxAssetsValuationAllowance",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilities": {
"auth_ref": [
"r538",
"r555"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after deferred tax asset, of deferred tax liability attributable to taxable differences without jurisdictional netting.",
"label": "Deferred Tax Liabilities, Net",
"negatedTotalLabel": "Total deferred tax liabilities"
}
}
},
"localname": "DeferredTaxLiabilities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilitiesGoodwillAndIntangibleAssetsGoodwill": {
"auth_ref": [
"r560",
"r561",
"r599",
"r600"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax liability attributable to taxable temporary differences from goodwill.",
"label": "Deferred Tax Liabilities, Goodwill",
"terseLabel": "Deferred tax liabilities, goodwill"
}
}
},
"localname": "DeferredTaxLiabilitiesGoodwillAndIntangibleAssetsGoodwill",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilitiesGoodwillAndIntangibleAssetsIntangibleAssets": {
"auth_ref": [
"r560",
"r561"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails": {
"order": 1.0,
"parentTag": "us-gaap_DeferredTaxLiabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax liability attributable to taxable temporary differences from intangible assets other than goodwill.",
"label": "Deferred Tax Liabilities, Intangible Assets",
"negatedTerseLabel": "Purchased intangible assets"
}
}
},
"localname": "DeferredTaxLiabilitiesGoodwillAndIntangibleAssetsIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesComponentsofDeferredTaxAssetsandLiabilitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilitiesOther": {
"auth_ref": [
"r560",
"r561"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax liability attributable to taxable temporary differences classified as other.",
"label": "Deferred Tax Liabilities, Other",
"terseLabel": "Deferred tax liabilities, other"
}
}
},
"localname": "DeferredTaxLiabilitiesOther",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilitiesUndistributedForeignEarnings": {
"auth_ref": [
"r560"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax liability attributable to taxable temporary differences from undistributed earnings of subsidiary and other recognized entity not within country of domicile. Includes, but is not limited to, other basis differences.",
"label": "Deferred Tax Liabilities, Undistributed Foreign Earnings",
"terseLabel": "Transition toll tax liabilities"
}
}
},
"localname": "DeferredTaxLiabilitiesUndistributedForeignEarnings",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DefinedBenefitPlanBenefitObligation": {
"auth_ref": [
"r406"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of actuarial present value of benefits attributed to service rendered by employee for defined benefit plan.",
"label": "Defined Benefit Plan, Benefit Obligation",
"verboseLabel": "Employee benefit plan obligations"
}
}
},
"localname": "DefinedBenefitPlanBenefitObligation",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DefinedBenefitPlanFundedStatusOfPlan": {
"auth_ref": [
"r405",
"r426",
"r474"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of funded (unfunded) status of defined benefit plan, measured as difference between fair value of plan assets and benefit obligation. Includes, but is not limited to, overfunded (underfunded) status.",
"label": "Defined Benefit Plan, Funded (Unfunded) Status of Plan",
"verboseLabel": "Unfunded net pension"
}
}
},
"localname": "DefinedBenefitPlanFundedStatusOfPlan",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DefinedBenefitPlanNetPeriodicBenefitCost": {
"auth_ref": [
"r439",
"r464",
"r474",
"r475"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of net periodic benefit cost (credit) for defined benefit plan.",
"label": "Defined Benefit Plan, Net Periodic Benefit Cost (Credit)",
"verboseLabel": "Periodic pension cost"
}
}
},
"localname": "DefinedBenefitPlanNetPeriodicBenefitCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DefinedContributionPlanCostRecognized": {
"auth_ref": [
"r478"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cost for defined contribution plan.",
"label": "Defined Contribution Plan, Cost",
"terseLabel": "Expenses related to savings plan"
}
}
},
"localname": "DefinedContributionPlanCostRecognized",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DefinedContributionPlanDisclosureLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Defined Contribution Plan Disclosure [Line Items]",
"terseLabel": "Defined Contribution Plan Disclosure [Line Items]"
}
}
},
"localname": "DefinedContributionPlanDisclosureLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DefinedContributionPlanTable": {
"auth_ref": [
"r478"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about defined contribution pension plans or defined contribution other postretirement plans, separately for pension plans and other postretirement benefit plans.",
"label": "Defined Contribution Plan [Table]",
"terseLabel": "Defined Contribution Plan [Table]"
}
}
},
"localname": "DefinedContributionPlanTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_Depreciation": {
"auth_ref": [
"r151",
"r339"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of expense recognized in the current period that reflects the allocation of the cost of tangible assets over the assets' useful lives. Includes production and non-production related depreciation.",
"label": "Depreciation",
"terseLabel": "Depreciation expense"
}
}
},
"localname": "Depreciation",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DepreciationDepletionAndAmortization": {
"auth_ref": [
"r151",
"r236"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 4.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate expense recognized in the current period that allocates the cost of tangible assets, intangible assets, or depleting assets to periods that benefit from use of the assets.",
"label": "Depreciation, Depletion and Amortization",
"verboseLabel": "Depreciation and amortization"
}
}
},
"localname": "DepreciationDepletionAndAmortization",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DerivativeContractTypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Financial instrument or contract with one or more underlyings, notional amount or payment provision or both, and the contract can be net settled by means outside the contract or delivery of an asset.",
"label": "Derivative Contract [Domain]",
"terseLabel": "Derivative Contract Type [Domain]"
}
}
},
"localname": "DerivativeContractTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DerivativeFairValueOfDerivativeAsset": {
"auth_ref": [
"r84",
"r86",
"r641",
"r734"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value, before effects of master netting arrangements, of a financial asset or other contract with one or more underlyings, notional amount or payment provision or both, and the contract can be net settled by means outside the contract or delivery of an asset. Includes assets elected not to be offset. Excludes assets not subject to a master netting arrangement.",
"label": "Derivative Asset, Fair Value, Gross Asset",
"terseLabel": "Asset derivative instruments"
}
}
},
"localname": "DerivativeFairValueOfDerivativeAsset",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DerivativeFairValueOfDerivativeLiability": {
"auth_ref": [
"r84",
"r86",
"r641",
"r734"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value, before effects of master netting arrangements, of a financial liability or contract with one or more underlyings, notional amount or payment provision or both, and the contract can be net settled by means outside the contract or delivery of an asset. Includes liabilities elected not to be offset. Excludes liabilities not subject to a master netting arrangement.",
"label": "Derivative Liability, Fair Value, Gross Liability",
"terseLabel": "Liability derivative instruments"
}
}
},
"localname": "DerivativeFairValueOfDerivativeLiability",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DerivativeInstrumentRiskAxis": {
"auth_ref": [
"r640",
"r642",
"r648",
"r654"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of derivative contract.",
"label": "Derivative Instrument [Axis]",
"terseLabel": "Derivative Instrument [Axis]"
}
}
},
"localname": "DerivativeInstrumentRiskAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DerivativeInstrumentsAndHedgingActivitiesDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Derivative Instruments and Hedging Activities Disclosure [Abstract]",
"terseLabel": "Derivative Instruments and Hedging Activities Disclosure [Abstract]"
}
}
},
"localname": "DerivativeInstrumentsAndHedgingActivitiesDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_DerivativeInstrumentsAndHedgingActivitiesDisclosureTextBlock": {
"auth_ref": [
"r662"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for derivative instruments and hedging activities including, but not limited to, risk management strategies, non-hedging derivative instruments, assets, liabilities, revenue and expenses, and methodologies and assumptions used in determining the amounts.",
"label": "Derivative Instruments and Hedging Activities Disclosure [Text Block]",
"verboseLabel": "Derivative Instruments"
}
}
},
"localname": "DerivativeInstrumentsAndHedgingActivitiesDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstruments"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DerivativeInstrumentsGainLossByHedgingRelationshipAxis": {
"auth_ref": [
"r637",
"r640",
"r648"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of hedging relationship.",
"label": "Hedging Relationship [Axis]",
"terseLabel": "Hedging Relationship [Axis]"
}
}
},
"localname": "DerivativeInstrumentsGainLossByHedgingRelationshipAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DerivativeInstrumentsGainLossByHedgingRelationshipByIncomeStatementLocationByDerivativeInstrumentRiskTable": {
"auth_ref": [
"r637",
"r640",
"r648",
"r654",
"r655",
"r657",
"r659"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about the location and amount of derivative instruments and nonderivative instruments designated as hedging instruments reported before netting adjustments, and the amount of gain (loss) on derivative instruments and nonderivative instruments designated and qualified as hedging instruments.",
"label": "Derivative Instruments, Gain (Loss) [Table]",
"terseLabel": "Derivative Instruments, Gain (Loss) by Hedging Relationship, by Income Statement Location, by Derivative Instrument Risk [Table]"
}
}
},
"localname": "DerivativeInstrumentsGainLossByHedgingRelationshipByIncomeStatementLocationByDerivativeInstrumentRiskTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DerivativeInstrumentsGainLossLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Derivative Instruments, Gain (Loss) [Line Items]",
"terseLabel": "Derivative Instruments, Gain (Loss) [Line Items]"
}
}
},
"localname": "DerivativeInstrumentsGainLossLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DerivativeLiabilitiesCurrent": {
"auth_ref": [
"r85"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": 7.0,
"parentTag": "us-gaap_OtherLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value, after the effects of master netting arrangements, of a financial liability or contract with one or more underlyings, notional amount or payment provision or both, and the contract can be net settled by means outside the contract or delivery of an asset, expected to be settled within one year or normal operating cycle, if longer. Includes assets not subject to a master netting arrangement and not elected to be offset.",
"label": "Derivative Liability, Current",
"terseLabel": "Derivative liabilities"
}
}
},
"localname": "DerivativeLiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DerivativeNotionalAmount": {
"auth_ref": [
"r631",
"r633"
],
"lang": {
"en-us": {
"role": {
"documentation": "Nominal or face amount used to calculate payment on derivative.",
"label": "Derivative, Notional Amount",
"terseLabel": "Derivative, notional amount"
}
}
},
"localname": "DerivativeNotionalAmount",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DerivativeRemainingMaturity1": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Period remaining until the derivative contract matures, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Derivative, Remaining Maturity",
"terseLabel": "Range of durations of foreign currency forward contracts"
}
}
},
"localname": "DerivativeRemainingMaturity1",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_DerivativeTermOfContract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Period the derivative contract is outstanding, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Derivative, Term of Contract",
"terseLabel": "Forward contracts duration"
}
}
},
"localname": "DerivativeTermOfContract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_DerivativesFairValueLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Derivatives, Fair Value [Line Items]",
"terseLabel": "Derivatives, Fair Value [Line Items]"
}
}
},
"localname": "DerivativesFairValueLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DerivativesPolicyTextBlock": {
"auth_ref": [
"r158",
"r170",
"r630",
"r632",
"r637",
"r638",
"r656"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for its derivative instruments and hedging activities.",
"label": "Derivatives, Policy [Policy Text Block]",
"terseLabel": "Derivative Instruments and Hedging Activities"
}
}
},
"localname": "DerivativesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DesignatedAsHedgingInstrumentMember": {
"auth_ref": [
"r637"
],
"lang": {
"en-us": {
"role": {
"documentation": "Derivative instrument designated as hedging instrument under Generally Accepted Accounting Principles (GAAP).",
"label": "Designated as Hedging Instrument [Member]",
"terseLabel": "Designated as Hedging Instrument"
}
}
},
"localname": "DesignatedAsHedgingInstrumentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DevelopedTechnologyRightsMember": {
"auth_ref": [
"r589"
],
"lang": {
"en-us": {
"role": {
"documentation": "Rights to developed technology, which can include the right to develop, use, market, sell, or offer for sale products, compounds, or intellectual property.",
"label": "Developed Technology Rights [Member]",
"terseLabel": "Completed technology"
}
}
},
"localname": "DevelopedTechnologyRightsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DilutiveSecuritiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Dilutive Securities, Effect on Basic Earnings Per Share [Abstract]",
"verboseLabel": "Effect of dilutive securities:"
}
}
},
"localname": "DilutiveSecuritiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DisaggregationOfRevenueLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Disaggregation of Revenue [Line Items]",
"terseLabel": "Disaggregation of Revenue [Line Items]"
}
}
},
"localname": "DisaggregationOfRevenueLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DisaggregationOfRevenueTable": {
"auth_ref": [
"r393",
"r395",
"r396",
"r397",
"r398",
"r399",
"r400",
"r401"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about disaggregation of revenue into categories depicting how nature, amount, timing, and uncertainty of revenue and cash flows are affected by economic factor.",
"label": "Disaggregation of Revenue [Table]",
"terseLabel": "Disaggregation of Revenue [Table]"
}
}
},
"localname": "DisaggregationOfRevenueTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DisaggregationOfRevenueTableTextBlock": {
"auth_ref": [
"r393"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of disaggregation of revenue into categories depicting how nature, amount, timing, and uncertainty of revenue and cash flows are affected by economic factor.",
"label": "Disaggregation of Revenue [Table Text Block]",
"terseLabel": "Revenue by product"
}
}
},
"localname": "DisaggregationOfRevenueTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock": {
"auth_ref": [
"r529"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for share-based payment arrangement.",
"label": "Share-based Payment Arrangement [Text Block]",
"verboseLabel": "Share-Based Payments"
}
}
},
"localname": "DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPayments"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DisclosureOfCompensationRelatedCostsSharebasedPaymentsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Share-based Payment Arrangement [Abstract]",
"terseLabel": "Share-based Payment Arrangement [Abstract]"
}
}
},
"localname": "DisclosureOfCompensationRelatedCostsSharebasedPaymentsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_DiscontinuedOperationProvisionForLossGainOnDisposalNetOfTax": {
"auth_ref": [
"r2",
"r4",
"r6",
"r17"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of (gain) loss recognized for the (reversal of write-down) write-down to fair value, less cost to sell, of a discontinued operation.",
"label": "Discontinued Operation, Provision for Loss (Gain) on Disposal, Net of Tax",
"terseLabel": "Loss on divestiture of Hiller\u00f8d, Denmark manufacturing operations, net of tax"
}
}
},
"localname": "DiscontinuedOperationProvisionForLossGainOnDisposalNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DiscontinuedOperationTaxEffectOfDiscontinuedOperation": {
"auth_ref": [
"r3",
"r4",
"r5",
"r6",
"r13",
"r17",
"r539",
"r570",
"r578"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of tax expense (benefit) related to a discontinued operation. Includes, but is not limited to, tax expense (benefit) related to income (loss) from operations during the phase-out period, tax expense (benefit) related to gain (loss) on disposal, tax expense (benefit) related to gain (loss) for reversal of write-down (write-down) to fair value, less cost to sell, and tax expense (benefit) related to adjustments of a prior period gain (loss) on disposal.",
"label": "Discontinued Operation, Tax Effect of Discontinued Operation",
"terseLabel": "Tax expense on disposal group"
}
}
},
"localname": "DiscontinuedOperationTaxEffectOfDiscontinuedOperation",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DisposalGroupIncludingDiscontinuedOperationConsideration": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of consideration received or receivable for the disposal of assets and liabilities, including discontinued operation.",
"label": "Disposal Group, Including Discontinued Operation, Consideration",
"terseLabel": "Consideration received for sale of Denmark manufacturing operations"
}
}
},
"localname": "DisposalGroupIncludingDiscontinuedOperationConsideration",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DisposalGroupIncludingDiscontinuedOperationInventory1": {
"auth_ref": [
"r0",
"r1",
"r15",
"r345"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount classified as inventory attributable to disposal group held for sale or disposed of.",
"label": "Disposal Group, Including Discontinued Operation, Inventory",
"terseLabel": "Inventory work-in-process sold to FUJIFILM"
}
}
},
"localname": "DisposalGroupIncludingDiscontinuedOperationInventory1",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DisposalGroupIncludingDiscontinuedOperationOtherExpense": {
"auth_ref": [
"r14"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of other expense attributable to disposal group, including, but not limited to, discontinued operation.",
"label": "Disposal Group, Including Discontinued Operation, Other Expense",
"terseLabel": "Expected costs to sell disposal group"
}
}
},
"localname": "DisposalGroupIncludingDiscontinuedOperationOtherExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DisposalGroupIncludingDiscontinuedOperationPropertyPlantAndEquipment": {
"auth_ref": [
"r0",
"r1",
"r15",
"r345"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount classified as property, plant and equipment attributable to disposal group held for sale or disposed of.",
"label": "Disposal Group, Including Discontinued Operation, Property, Plant and Equipment",
"terseLabel": "Property, plant and equipment sold to Fujifilm"
}
}
},
"localname": "DisposalGroupIncludingDiscontinuedOperationPropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DisposalGroupNotDiscontinuedOperationLossGainOnWriteDown": {
"auth_ref": [
"r151",
"r338",
"r343"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 7.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before tax, of (gain) loss recognized for the (reversal of write-down) write-down to fair value, less cost to sell, of a disposal group. Excludes discontinued operations.",
"label": "Disposal Group, Not Discontinued Operation, Loss (Gain) on Write-down",
"negatedTerseLabel": "(Gain) loss on divestiture of Hiller\u00f8d, Denmark manufacturing operations",
"terseLabel": "(Gain) loss on divestiture of Hiller\u00f8d, Denmark manufacturing operations"
}
}
},
"localname": "DisposalGroupNotDiscontinuedOperationLossGainOnWriteDown",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/DivestituresDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DisposalGroupsIncludingDiscontinuedOperationsDisclosureTextBlock": {
"auth_ref": [
"r21",
"r350"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure related to a disposal group. Includes, but is not limited to, a discontinued operation, disposal classified as held-for-sale or disposed of by means other than sale or disposal of an individually significant component.",
"label": "Disposal Groups, Including Discontinued Operations, Disclosure [Text Block]",
"terseLabel": "Divestitures"
}
}
},
"localname": "DisposalGroupsIncludingDiscontinuedOperationsDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Divestitures"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DisposalGroupsIncludingDiscontinuedOperationsNameDomain": {
"auth_ref": [
"r480",
"r486"
],
"lang": {
"en-us": {
"role": {
"documentation": "Name of disposal group.",
"label": "Disposal Group Name [Domain]",
"terseLabel": "Disposal Group Name [Domain]"
}
}
},
"localname": "DisposalGroupsIncludingDiscontinuedOperationsNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DomesticCountryMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Designated tax department of the government that is entitled to levy and collect income taxes from the entity in its country of domicile.",
"label": "Domestic Tax Authority [Member]",
"terseLabel": "Domestic Country"
}
}
},
"localname": "DomesticCountryMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DomesticPlanMember": {
"auth_ref": [
"r479",
"r483",
"r489"
],
"lang": {
"en-us": {
"role": {
"documentation": "Location of employer sponsoring plan, designed to provide retirement benefits, determined as principal place of business. Includes, but is not limited to, defined benefit and defined contribution plans.",
"label": "Domestic Plan [Member]",
"terseLabel": "Domestic Plan"
}
}
},
"localname": "DomesticPlanMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DueFromRelatedParties": {
"auth_ref": [
"r169",
"r718",
"r766",
"r800"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "For an unclassified balance sheet, amounts due from related parties including affiliates, employees, joint ventures, officers and stockholders, immediate families thereof, and pension funds.",
"label": "Due from Related Parties",
"terseLabel": "Due from related parties"
}
}
},
"localname": "DueFromRelatedParties",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DueToRelatedPartiesCurrentAndNoncurrent": {
"auth_ref": [
"r169",
"r718",
"r767",
"r799"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying amount as of the balance sheet date of obligations due all related parties.",
"label": "Due to Related Parties",
"terseLabel": "Due to related parties"
}
}
},
"localname": "DueToRelatedPartiesCurrentAndNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EarningsPerShareAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Earnings Per Share [Abstract]",
"terseLabel": "Earnings Per Share [Abstract]",
"verboseLabel": "Net income per share:"
}
}
},
"localname": "EarningsPerShareAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "stringItemType"
},
"us-gaap_EarningsPerShareBasic": {
"auth_ref": [
"r117",
"r186",
"r187",
"r188",
"r189",
"r190",
"r194",
"r197",
"r204",
"r205",
"r206",
"r210",
"r211",
"r776",
"r807"
],
"lang": {
"en-us": {
"role": {
"documentation": "The amount of net income (loss) for the period per each share of common stock or unit outstanding during the reporting period.",
"label": "Earnings Per Share, Basic",
"verboseLabel": "Basic earnings per share attributable to Biogen Inc. (in dollars per share)"
}
}
},
"localname": "EarningsPerShareBasic",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_EarningsPerShareBasicAndDilutedAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Earnings Per Share, Basic and Diluted [Abstract]",
"terseLabel": "Net income per share:"
}
}
},
"localname": "EarningsPerShareBasicAndDilutedAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_EarningsPerShareDiluted": {
"auth_ref": [
"r117",
"r186",
"r187",
"r188",
"r189",
"r190",
"r197",
"r204",
"r205",
"r206",
"r210",
"r211",
"r776",
"r807"
],
"lang": {
"en-us": {
"role": {
"documentation": "The amount of net income (loss) for the period available to each share of common stock or common unit outstanding during the reporting period and to each share or unit that would have been outstanding assuming the issuance of common shares or units for all dilutive potential common shares or units outstanding during the reporting period.",
"label": "Earnings Per Share, Diluted",
"verboseLabel": "Diluted earnings per share attributable to Biogen Inc. (in dollars per share)"
}
}
},
"localname": "EarningsPerShareDiluted",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_EarningsPerSharePolicyTextBlock": {
"auth_ref": [
"r158",
"r207",
"r208"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for computing basic and diluted earnings or loss per share for each class of common stock and participating security. Addresses all significant policy factors, including any antidilutive items that have been excluded from the computation and takes into account stock dividends, splits and reverse splits that occur after the balance sheet date of the latest reporting period but before the issuance of the financial statements.",
"label": "Earnings Per Share, Policy [Policy Text Block]",
"terseLabel": "Earnings per Share"
}
}
},
"localname": "EarningsPerSharePolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_EarningsPerShareTextBlock": {
"auth_ref": [
"r207",
"r208",
"r209",
"r212"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for earnings per share.",
"label": "Earnings Per Share [Text Block]",
"verboseLabel": "Earnings per Share"
}
}
},
"localname": "EarningsPerShareTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShare"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_EffectOfExchangeRateOnCashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents": {
"auth_ref": [
"r685"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) from effect of exchange rate changes on cash and cash equivalents, and cash and cash equivalents restricted to withdrawal or usage; held in foreign currencies. Excludes amounts for disposal group and discontinued operations. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Effect of Exchange Rate on Cash, Cash Equivalents, Restricted Cash and Restricted Cash Equivalents",
"verboseLabel": "Effect of exchange rate changes on cash and cash equivalents"
}
}
},
"localname": "EffectOfExchangeRateOnCashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EffectiveIncomeTaxRateContinuingOperations": {
"auth_ref": [
"r540"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of current income tax expense (benefit) and deferred income tax expense (benefit) pertaining to continuing operations.",
"label": "Effective Income Tax Rate Reconciliation, Percent",
"totalLabel": "Effective tax rate"
}
}
},
"localname": "EffectiveIncomeTaxRateContinuingOperations",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 1.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of domestic federal statutory tax rate applicable to pretax income (loss).",
"label": "Effective Income Tax Rate Reconciliation, at Federal Statutory Income Tax Rate, Percent",
"verboseLabel": "Statutory rate"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationChangeInEnactedTaxRate": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 9.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to changes in the income tax rates.",
"label": "Effective Income Tax Rate Reconciliation, Change in Enacted Tax Rate, Percent",
"terseLabel": "U.S. tax reform"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationChangeInEnactedTaxRate",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationDispositionOfAssets": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 6.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying domestic federal statutory income tax rate to pretax income (loss) from continuing operations, attributable to disposition of asset. Includes, but is not limited to, intra-entity transfer of asset other than inventory.",
"label": "Effective Income Tax Rate Reconciliation, Disposition of Asset, Percent",
"terseLabel": "Divestiture of Denmark manufacturing operations"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationDispositionOfAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationForeignIncomeTaxRateDifferential": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 3.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations applicable to statutory income tax expense (benefit) outside of the country of domicile.",
"label": "Effective Income Tax Rate Reconciliation, Foreign Income Tax Rate Differential, Percent",
"verboseLabel": "Taxes on foreign earnings"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationForeignIncomeTaxRateDifferential",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationNondeductibleExpenseDepreciationAndAmortization": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 5.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to depreciation and amortization.",
"label": "Effective Income Tax Rate Reconciliation, Nondeductible Expense, Depreciation and Amortization, Percent",
"verboseLabel": "Purchased intangible assets"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationNondeductibleExpenseDepreciationAndAmortization",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationNondeductibleExpenseImpairmentLosses": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 11.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to impairment loss.",
"label": "Effective Income Tax Rate Reconciliation, Nondeductible Expense, Impairment Losses, Percent",
"terseLabel": "TECFIDERA impairment"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationNondeductibleExpenseImpairmentLosses",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationOtherAdjustments": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 12.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to other adjustments.",
"label": "Effective Income Tax Rate Reconciliation, Other Adjustments, Percent",
"verboseLabel": "Other"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationOtherAdjustments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationStateAndLocalIncomeTaxes": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 2.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations applicable to state and local income tax expense (benefit), net of federal tax expense (benefit).",
"label": "Effective Income Tax Rate Reconciliation, State and Local Income Taxes, Percent",
"verboseLabel": "State taxes"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationStateAndLocalIncomeTaxes",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationTaxCredits": {
"auth_ref": [
"r540",
"r575"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails": {
"order": 4.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": -1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to tax credits. Including, but not limited to, research credit, foreign tax credit, investment tax credit, and other tax credits.",
"label": "Effective Income Tax Rate Reconciliation, Tax Credit, Percent",
"negatedLabel": "Credits and net operating loss utilization"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationTaxCredits",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EmployeeRelatedLiabilitiesCurrent": {
"auth_ref": [
"r69"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": 1.0,
"parentTag": "us-gaap_OtherLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Total of the carrying values as of the balance sheet date of obligations incurred through that date and payable for obligations related to services received from employees, such as accrued salaries and bonuses, payroll taxes and fringe benefits. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Employee-related Liabilities, Current",
"terseLabel": "Employee-related Liabilities, Current"
}
}
},
"localname": "EmployeeRelatedLiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsCapitalizedAmount": {
"auth_ref": [
"r518"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cost capitalized for award under share-based payment arrangement.",
"label": "Share-based Payment Arrangement, Amount Capitalized",
"negatedTerseLabel": "Capitalized share-based compensation costs"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsCapitalizedAmount",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Share-based Payment Arrangement, Expensed and Capitalized, Amount [Line Items]",
"terseLabel": "Employee Service Share-based Compensation, Allocation of Recognized Period Costs [Line Items]"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationCashFlowEffectCashUsedToSettleAwards": {
"auth_ref": [
"r520"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate cash paid by the entity during the period to settle equity instruments granted under equity-based payment arrangements.",
"label": "Share-based Payment Arrangement, Cash Used to Settle Award",
"verboseLabel": "Cash settlement of awards upon vesting"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationCashFlowEffectCashUsedToSettleAwards",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognized": {
"auth_ref": [
"r519"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cost not yet recognized for nonvested award under share-based payment arrangement.",
"label": "Share-based Payment Arrangement, Nonvested Award, Cost Not yet Recognized, Amount",
"verboseLabel": "Unrecognized compensation cost related to unvested share-based compensation"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognized",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationTaxBenefitFromCompensationExpense": {
"auth_ref": [
"r517"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of tax benefit for recognition of expense of award under share-based payment arrangement.",
"label": "Share-based Payment Arrangement, Expense, Tax Benefit",
"negatedLabel": "Income tax effect"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationTaxBenefitFromCompensationExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationTaxBenefitFromExerciseOfStockOptions": {
"auth_ref": [
"r522"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of tax benefit from exercise of option under share-based payment arrangement.",
"label": "Share-based Payment Arrangement, Exercise of Option, Tax Benefit",
"verboseLabel": "Tax benefit realized for stock options"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationTaxBenefitFromExerciseOfStockOptions",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTaxBenefitandCashReceivedfromStockOptionExercisesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeStockOptionMember": {
"auth_ref": [
"r515"
],
"lang": {
"en-us": {
"role": {
"documentation": "Share-based payment arrangement granting right, subject to vesting and other restrictions, to purchase or sell certain number of shares at predetermined price for specified period of time.",
"label": "Share-based Payment Arrangement, Option [Member]",
"terseLabel": "Share-based Payment Arrangement, Option"
}
}
},
"localname": "EmployeeStockOptionMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_EmployeeStockOwnershipPlanESOPDisclosuresLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Employee Stock Ownership Plan (ESOP) Disclosures [Line Items]",
"terseLabel": "Employee Stock Ownership Plan (ESOP) Disclosures [Line Items]"
}
}
},
"localname": "EmployeeStockOwnershipPlanESOPDisclosuresLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_EntityWideInformationRevenueFromExternalCustomerLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Revenue from External Customer [Line Items]",
"terseLabel": "Revenue from External Customer [Line Items]"
}
}
},
"localname": "EntityWideInformationRevenueFromExternalCustomerLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_EntityWideRevenueMajorCustomerLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Revenue, Major Customer [Line Items]",
"terseLabel": "Revenue, Major Customer [Line Items]"
}
}
},
"localname": "EntityWideRevenueMajorCustomerLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_EquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Equity [Abstract]",
"terseLabel": "Equity [Abstract]"
}
}
},
"localname": "EquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_EquityComponentDomain": {
"auth_ref": [
"r180",
"r181",
"r182",
"r185",
"r191",
"r193",
"r216",
"r294",
"r372",
"r379",
"r523",
"r524",
"r525",
"r567",
"r568",
"r686",
"r687",
"r688",
"r689",
"r690",
"r692",
"r818",
"r819",
"r820"
],
"lang": {
"en-us": {
"role": {
"documentation": "Components of equity are the parts of the total Equity balance including that which is allocated to common, preferred, treasury stock, retained earnings, etc.",
"label": "Equity Component [Domain]",
"terseLabel": "Equity Component [Domain]"
}
}
},
"localname": "EquityComponentDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_EquityMethodInvestmentDifferenceBetweenCarryingAmountAndUnderlyingEquity": {
"auth_ref": [
"r292"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Difference between amount at which an investment accounted for under the equity method of accounting is carried (reported) on the balance sheet and amount of underlying equity in net assets the reporting Entity has in the investee.",
"label": "Equity Method Investment, Difference Between Carrying Amount and Underlying Equity",
"terseLabel": "Total basis difference"
}
}
},
"localname": "EquityMethodInvestmentDifferenceBetweenCarryingAmountAndUnderlyingEquity",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EquityMethodInvestmentOwnershipPercentage": {
"auth_ref": [
"r290"
],
"lang": {
"en-us": {
"role": {
"documentation": "The percentage of ownership of common stock or equity participation in the investee accounted for under the equity method of accounting.",
"label": "Equity Method Investment, Ownership Percentage",
"terseLabel": "Samsung Bioepis, ownership percentage"
}
}
},
"localname": "EquityMethodInvestmentOwnershipPercentage",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EquityMethodInvestments": {
"auth_ref": [
"r58",
"r240",
"r289"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "This item represents the carrying amount on the entity's balance sheet of its investment in common stock of an equity method investee. This is not an indicator of the fair value of the investment, rather it is the initial cost adjusted for the entity's share of earnings and losses of the investee, adjusted for any distributions (dividends) and other than temporary impairment (OTTI) losses recognized.",
"label": "Equity Method Investments",
"terseLabel": "Investment in Samsung Bioepis"
}
}
},
"localname": "EquityMethodInvestments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EquityMethodInvestmentsPolicy": {
"auth_ref": [
"r58",
"r145",
"r158",
"r291",
"r680"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for equity method of accounting for investments and other interests. Investment includes, but is not limited to, unconsolidated subsidiary, corporate joint venture, noncontrolling interest in real estate venture, limited partnership, and limited liability company. Information includes, but is not limited to, ownership percentage, reason equity method is or is not considered appropriate, and accounting policy election for distribution received.",
"label": "Equity Method Investments [Policy Text Block]",
"terseLabel": "Equity Method of Accounting"
}
}
},
"localname": "EquityMethodInvestmentsPolicy",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_EquitySecuritiesFvNi": {
"auth_ref": [
"r677"
],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": 3.0,
"parentTag": "us-gaap_EquitySecuritiesFvNiCost",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of investment in equity security measured at fair value with change in fair value recognized in net income (FV-NI).",
"label": "Equity Securities, FV-NI",
"terseLabel": "Marketable equity securities, fair value"
}
}
},
"localname": "EquitySecuritiesFvNi",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EquitySecuritiesFvNiCost": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Cost of investment in equity security measured at fair value with change in fair value recognized in net income (FV-NI). Excludes equity method investment and investment in equity security without readily determinable fair value.",
"label": "Equity Securities, FV-NI, Cost",
"totalLabel": "Marketable equity securities, amortized cost"
}
}
},
"localname": "EquitySecuritiesFvNiCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EquitySecuritiesFvNiGainLoss": {
"auth_ref": [
"r288"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailGainLossonInvestmentsDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of unrealized and realized gain (loss) on investment in equity security measured at fair value with change in fair value recognized in net income (FV-NI).",
"label": "Equity Securities, FV-NI, Gain (Loss)",
"totalLabel": "Net gains (losses) recognized during the period on equity securities"
}
}
},
"localname": "EquitySecuritiesFvNiGainLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailGainLossonInvestmentsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EquitySecuritiesFvNiRealizedGainLoss": {
"auth_ref": [
"r288",
"r809"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailGainLossonInvestmentsDetails": {
"order": 1.0,
"parentTag": "us-gaap_EquitySecuritiesFvNiGainLoss",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of realized gain (loss) from sale of investment in equity security measured at fair value with change in fair value recognized in net income (FV-NI).",
"label": "Equity Securities, FV-NI, Realized Gain (Loss)",
"negatedTerseLabel": "Less: Net gains (losses) recognized on equity securities sold during the period and on capital distributions"
}
}
},
"localname": "EquitySecuritiesFvNiRealizedGainLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailGainLossonInvestmentsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EquitySecuritiesFvNiUnrealizedGainLoss": {
"auth_ref": [
"r288",
"r809"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailGainLossonInvestmentsDetails": {
"order": 2.0,
"parentTag": "us-gaap_EquitySecuritiesFvNiGainLoss",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of unrealized gain (loss) on investment in equity security measured at fair value with change in fair value recognized in net income (FV-NI).",
"label": "Equity Securities, FV-NI, Unrealized Gain (Loss)",
"terseLabel": "Unrealized gains (losses) recognized during the period on equity securities"
}
}
},
"localname": "EquitySecuritiesFvNiUnrealizedGainLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailGainLossonInvestmentsDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EuropeanUnionMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Economic and political union of member states, located primarily in Europe.",
"label": "European Union [Member]",
"terseLabel": "European Union"
}
}
},
"localname": "EuropeanUnionMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Fair Value, Assets and Liabilities Measured on Recurring and Nonrecurring Basis [Line Items]",
"terseLabel": "Fair Value, Assets and Liabilities Measured on Recurring and Nonrecurring Basis [Line Items]"
}
}
},
"localname": "FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisTable": {
"auth_ref": [
"r663",
"r664",
"r665",
"r674"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about asset and liability measured at fair value on recurring and nonrecurring basis.",
"label": "Fair Value, Recurring and Nonrecurring [Table]",
"terseLabel": "Fair Value, Assets and Liabilities Measured on Recurring and Nonrecurring Basis [Table]"
}
}
},
"localname": "FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock": {
"auth_ref": [
"r666"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of input and valuation technique used to measure fair value and change in valuation approach and technique for each separate class of asset and liability measured on recurring and nonrecurring basis.",
"label": "Fair Value Measurement Inputs and Valuation Techniques [Table Text Block]",
"terseLabel": "Summary of significant unobservable inputs"
}
}
},
"localname": "FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueAssetsMeasuredOnNonrecurringBasisTextBlock": {
"auth_ref": [
"r663",
"r664",
"r666"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of assets and liabilities by class, including financial instruments measured at fair value that are classified in shareholders' equity, if any, that are measured at fair value on a nonrecurring basis in periods after initial recognition (for example, impaired assets). Disclosures may include, but are not limited to: (a) the fair value measurements recorded and the reasons for the measurements and (b) the level within the fair value hierarchy in which the fair value measurements are categorized in their entirety (levels 1, 2, 3).",
"label": "Fair Value Measurements, Nonrecurring [Table Text Block]",
"terseLabel": "Summary of gains or losses on assets measured at fair value, nonrecurring basis"
}
}
},
"localname": "FairValueAssetsMeasuredOnNonrecurringBasisTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueAssetsMeasuredOnRecurringBasisUnobservableInputReconciliationByAssetClassDomain": {
"auth_ref": [
"r669"
],
"lang": {
"en-us": {
"role": {
"documentation": "Class of asset.",
"label": "Asset Class [Domain]",
"terseLabel": "Fair Value, Assets Measured on Recurring Basis, Unobservable Input Reconciliation, by Asset Class"
}
}
},
"localname": "FairValueAssetsMeasuredOnRecurringBasisUnobservableInputReconciliationByAssetClassDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueByAssetClassAxis": {
"auth_ref": [
"r663",
"r674"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by class of asset.",
"label": "Asset Class [Axis]",
"terseLabel": "Fair Value by Asset Class [Axis]"
}
}
},
"localname": "FairValueByAssetClassAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueByBalanceSheetGroupingTextBlock": {
"auth_ref": [
"r663",
"r679"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the fair value of financial instruments, including financial assets and financial liabilities, and the measurements of those instruments, assets, and liabilities.",
"label": "Fair Value, by Balance Sheet Grouping [Table Text Block]",
"verboseLabel": "Summary of fair and carrying value of debt instruments"
}
}
},
"localname": "FairValueByBalanceSheetGroupingTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueByFairValueHierarchyLevelAxis": {
"auth_ref": [
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r474",
"r664",
"r724",
"r725",
"r726"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by level within fair value hierarchy and fair value measured at net asset value per share as practical expedient.",
"label": "Fair Value Hierarchy and NAV [Axis]",
"terseLabel": "Fair Value Hierarchy and NAV [Axis]"
}
}
},
"localname": "FairValueByFairValueHierarchyLevelAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueByMeasurementFrequencyAxis": {
"auth_ref": [
"r663",
"r664",
"r667",
"r668",
"r675"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by measurement frequency.",
"label": "Measurement Frequency [Axis]",
"terseLabel": "Measurement Frequency [Axis]"
}
}
},
"localname": "FairValueByMeasurementFrequencyAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueDisclosuresAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Fair Value Disclosures [Abstract]",
"terseLabel": "Fair Value Disclosures [Abstract]"
}
}
},
"localname": "FairValueDisclosuresAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_FairValueDisclosuresTextBlock": {
"auth_ref": [
"r673"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for the fair value of financial instruments (as defined), including financial assets and financial liabilities (collectively, as defined), and the measurements of those instruments as well as disclosures related to the fair value of non-financial assets and liabilities. Such disclosures about the financial instruments, assets, and liabilities would include: (1) the fair value of the required items together with their carrying amounts (as appropriate); (2) for items for which it is not practicable to estimate fair value, disclosure would include: (a) information pertinent to estimating fair value (including, carrying amount, effective interest rate, and maturity, and (b) the reasons why it is not practicable to estimate fair value; (3) significant concentrations of credit risk including: (a) information about the activity, region, or economic characteristics identifying a concentration, (b) the maximum amount of loss the entity is exposed to based on the gross fair value of the related item, (c) policy for requiring collateral or other security and information as to accessing such collateral or security, and (d) the nature and brief description of such collateral or security; (4) quantitative information about market risks and how such risks are managed; (5) for items measured on both a recurring and nonrecurring basis information regarding the inputs used to develop the fair value measurement; and (6) for items presented in the financial statement for which fair value measurement is elected: (a) information necessary to understand the reasons for the election, (b) discussion of the effect of fair value changes on earnings, (c) a description of [similar groups] items for which the election is made and the relation thereof to the balance sheet, the aggregate carrying value of items included in the balance sheet that are not eligible for the election; (7) all other required (as defined) and desired information.",
"label": "Fair Value Disclosures [Text Block]",
"verboseLabel": "Fair Value Measurements"
}
}
},
"localname": "FairValueDisclosuresTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurements"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueHedgingMember": {
"auth_ref": [
"r634"
],
"lang": {
"en-us": {
"role": {
"documentation": "A hedge of the exposure to changes in the fair value of a recognized asset or liability, or of an unrecognized firm commitment, that are attributable to a particular risk.",
"label": "Fair Value Hedging [Member]",
"terseLabel": "Fair value hedge"
}
}
},
"localname": "FairValueHedgingMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueInputsLevel1Member": {
"auth_ref": [
"r428",
"r430",
"r435",
"r474",
"r664",
"r724"
],
"lang": {
"en-us": {
"role": {
"documentation": "Quoted prices in active markets for identical assets or liabilities that the reporting entity can access at the measurement date.",
"label": "Fair Value, Inputs, Level 1 [Member]",
"verboseLabel": "Quoted Prices in Active Markets (Level\u00a01)"
}
}
},
"localname": "FairValueInputsLevel1Member",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueInputsLevel2Member": {
"auth_ref": [
"r428",
"r430",
"r435",
"r474",
"r664",
"r725"
],
"lang": {
"en-us": {
"role": {
"documentation": "Inputs other than quoted prices included within level 1 that are observable for an asset or liability, either directly or indirectly, including, but not limited to, quoted prices for similar assets or liabilities in active markets, or quoted prices for identical or similar assets or liabilities in inactive markets.",
"label": "Fair Value, Inputs, Level 2 [Member]",
"verboseLabel": "Significant Other Observable Inputs (Level\u00a02)"
}
}
},
"localname": "FairValueInputsLevel2Member",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueInputsLevel3Member": {
"auth_ref": [
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r474",
"r664",
"r726"
],
"lang": {
"en-us": {
"role": {
"documentation": "Unobservable inputs that reflect the entity's own assumption about the assumptions market participants would use in pricing.",
"label": "Fair Value, Inputs, Level 3 [Member]",
"verboseLabel": "Significant Unobservable Inputs (Level\u00a03)"
}
}
},
"localname": "FairValueInputsLevel3Member",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationCalculationRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation, Calculation [Roll Forward]",
"terseLabel": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation, Calculation [Roll Forward]"
}
}
},
"localname": "FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationCalculationRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTextBlock": {
"auth_ref": [
"r669",
"r674"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the fair value measurement of liabilities using significant unobservable inputs (Level 3), a reconciliation of the beginning and ending balances, separately presenting changes attributable to the following: (1) total gains or losses for the period (realized and unrealized), segregating those gains or losses included in earnings (or changes in net assets), and gains or losses recognized in other comprehensive income (loss) and a description of where those gains or losses included in earnings (or changes in net assets) are reported in the statement of income (or activities); (2) purchases, sales, issues, and settlements (each type disclosed separately); and (3) transfers in and transfers out of Level 3 (for example, transfers due to changes in the observability of significant inputs) by class of liability.",
"label": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation [Table Text Block]",
"verboseLabel": "Fair value of contingent consideration obligations"
}
}
},
"localname": "FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueMeasurementFrequencyDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement frequency.",
"label": "Measurement Frequency [Domain]",
"terseLabel": "Measurement Frequency [Domain]"
}
}
},
"localname": "FairValueMeasurementFrequencyDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityGainLossIncludedInEarnings": {
"auth_ref": [
"r670"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 16.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
},
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 8.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of gain (loss) recognized in income from liability measured at fair value on recurring basis using unobservable input (level 3).",
"label": "Fair Value, Measurement with Unobservable Inputs Reconciliation, Recurring Basis, Liability, Gain (Loss) Included in Earnings",
"negatedLabel": "Contingent consideration",
"negatedTerseLabel": "(Gain) loss on fair value remeasurement of contingent consideration"
}
}
},
"localname": "FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityGainLossIncludedInEarnings",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityPurchases": {
"auth_ref": [
"r671"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of purchases of financial instrument classified as a liability measured using unobservable inputs that reflect the entity's own assumption about the assumptions market participants would use in pricing.",
"label": "Fair Value, Measurement with Unobservable Inputs Reconciliation, Recurring Basis, Liability, Purchases",
"verboseLabel": "Additions"
}
}
},
"localname": "FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityPurchases",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilitySettlements": {
"auth_ref": [
"r671"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of settlements of financial instrument classified as a liability measured using unobservable inputs that reflect the entity's own assumption about the assumptions market participants would use in pricing.",
"label": "Fair Value, Measurement with Unobservable Inputs Reconciliation, Recurring Basis, Liability, Settlements",
"negatedLabel": "Payments and other"
}
}
},
"localname": "FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilitySettlements",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueMeasurementsFairValueHierarchyDomain": {
"auth_ref": [
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r474",
"r724",
"r725",
"r726"
],
"lang": {
"en-us": {
"role": {
"documentation": "Categories used to prioritize the inputs to valuation techniques to measure fair value.",
"label": "Fair Value Hierarchy and NAV [Domain]",
"terseLabel": "Fair Value, Measurements, Fair Value Hierarchy [Domain]"
}
}
},
"localname": "FairValueMeasurementsFairValueHierarchyDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueMeasurementsNonrecurringMember": {
"auth_ref": [
"r663",
"r664",
"r667",
"r668",
"r672",
"r675"
],
"lang": {
"en-us": {
"role": {
"documentation": "Infrequent fair value measurement. Includes, but is not limited to, fair value adjustment for impairment of asset, liability or equity, not frequently measured at fair value.",
"label": "Fair Value, Nonrecurring [Member]",
"terseLabel": "Fair Value, Nonrecurring"
}
}
},
"localname": "FairValueMeasurementsNonrecurringMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueMeasurementsRecurringMember": {
"auth_ref": [
"r673",
"r675"
],
"lang": {
"en-us": {
"role": {
"documentation": "Frequent fair value measurement. Includes, but is not limited to, fair value adjustment for impairment of asset, liability or equity, frequently measured at fair value.",
"label": "Fair Value, Recurring [Member]",
"terseLabel": "Fair Value, Recurring",
"verboseLabel": "Fair value, measurements, recurring"
}
}
},
"localname": "FairValueMeasurementsRecurringMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueOfFinancialInstrumentsPolicy": {
"auth_ref": [
"r158",
"r676",
"r678"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for determining the fair value of financial instruments.",
"label": "Fair Value of Financial Instruments, Policy [Policy Text Block]",
"terseLabel": "Fair Value Measurements"
}
}
},
"localname": "FairValueOfFinancialInstrumentsPolicy",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValuesDerivativesBalanceSheetLocationByDerivativeContractTypeByHedgingDesignationTable": {
"auth_ref": [
"r639",
"r644",
"r657"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule that discloses the location and fair value amounts of derivative instruments (and nonderivative instruments that are designated and qualify as hedging instruments) reported in the statement of financial position.",
"label": "Fair Values Derivatives, Balance Sheet Location, by Derivative Contract Type [Table]",
"terseLabel": "Fair Values Derivatives, Balance Sheet Location, by Derivative Contract Type [Table]"
}
}
},
"localname": "FairValuesDerivativesBalanceSheetLocationByDerivativeContractTypeByHedgingDesignationTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FinancialInstrumentAxis": {
"auth_ref": [
"r280",
"r281",
"r295",
"r296",
"r297",
"r298",
"r299",
"r301",
"r302",
"r303",
"r304",
"r721",
"r722",
"r723",
"r724",
"r725",
"r726",
"r727",
"r728",
"r729",
"r730",
"r731",
"r732",
"r733",
"r735",
"r736",
"r737",
"r738",
"r739",
"r740",
"r741",
"r742",
"r743",
"r744",
"r745",
"r746",
"r747",
"r748",
"r749",
"r750",
"r751"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of financial instrument.",
"label": "Financial Instrument [Axis]",
"terseLabel": "Financial Instrument [Axis]"
}
}
},
"localname": "FinancialInstrumentAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FiniteLivedIntangibleAssetUsefulLife": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Useful life of finite-lived intangible assets, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Finite-Lived Intangible Asset, Useful Life",
"verboseLabel": "Estimated life (in years)"
}
}
},
"localname": "FiniteLivedIntangibleAssetUsefulLife",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsAccumulatedAmortization": {
"auth_ref": [
"r328"
],
"calculation": {
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails": {
"order": 2.0,
"parentTag": "us-gaap_IntangibleAssetsNetExcludingGoodwill",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated amount of amortization of assets, excluding financial assets and goodwill, lacking physical substance with a finite life.",
"label": "Finite-Lived Intangible Assets, Accumulated Amortization",
"negatedLabel": "Accumulated Amortization"
}
}
},
"localname": "FiniteLivedIntangibleAssetsAccumulatedAmortization",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsAmortizationExpenseNextTwelveMonths": {
"auth_ref": [
"r330"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of amortization for assets, excluding financial assets and goodwill, lacking physical substance with finite life expected to be recognized in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Finite-Lived Intangible Asset, Expected Amortization, Year One",
"verboseLabel": "2021"
}
}
},
"localname": "FiniteLivedIntangibleAssetsAmortizationExpenseNextTwelveMonths",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillEstimatedFutureAmortizationofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsAmortizationExpenseYearFive": {
"auth_ref": [
"r330"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of amortization for assets, excluding financial assets and goodwill, lacking physical substance with finite life expected to be recognized in fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Finite-Lived Intangible Asset, Expected Amortization, Year Five",
"terseLabel": "2025"
}
}
},
"localname": "FiniteLivedIntangibleAssetsAmortizationExpenseYearFive",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillEstimatedFutureAmortizationofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsAmortizationExpenseYearFour": {
"auth_ref": [
"r330"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of amortization for assets, excluding financial assets and goodwill, lacking physical substance with finite life expected to be recognized in fourth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Finite-Lived Intangible Asset, Expected Amortization, Year Four",
"verboseLabel": "2024"
}
}
},
"localname": "FiniteLivedIntangibleAssetsAmortizationExpenseYearFour",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillEstimatedFutureAmortizationofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsAmortizationExpenseYearThree": {
"auth_ref": [
"r330"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of amortization for assets, excluding financial assets and goodwill, lacking physical substance with finite life expected to be recognized in third fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Finite-Lived Intangible Asset, Expected Amortization, Year Three",
"verboseLabel": "2023"
}
}
},
"localname": "FiniteLivedIntangibleAssetsAmortizationExpenseYearThree",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillEstimatedFutureAmortizationofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsAmortizationExpenseYearTwo": {
"auth_ref": [
"r330"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of amortization for assets, excluding financial assets and goodwill, lacking physical substance with finite life expected to be recognized in second fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Finite-Lived Intangible Asset, Expected Amortization, Year Two",
"verboseLabel": "2022"
}
}
},
"localname": "FiniteLivedIntangibleAssetsAmortizationExpenseYearTwo",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillEstimatedFutureAmortizationofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsByMajorClassAxis": {
"auth_ref": [
"r321",
"r324",
"r328",
"r332",
"r755",
"r756"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by major type or class of finite-lived intangible assets.",
"label": "Finite-Lived Intangible Assets by Major Class [Axis]",
"terseLabel": "Finite-Lived Intangible Assets by Major Class [Axis]"
}
}
},
"localname": "FiniteLivedIntangibleAssetsByMajorClassAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsFairValueDisclosure": {
"auth_ref": [
"r663"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of assets, excluding financial assets, that lack physical substance, having a limited useful life.",
"label": "Finite-lived Intangible Assets, Fair Value Disclosure",
"terseLabel": "Intangible asset, fair value"
}
}
},
"localname": "FiniteLivedIntangibleAssetsFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsGross": {
"auth_ref": [
"r328",
"r756"
],
"calculation": {
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails": {
"order": 2.0,
"parentTag": "us-gaap_IntangibleAssetsGrossExcludingGoodwill",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before amortization of assets, excluding financial assets and goodwill, lacking physical substance with a finite life.",
"label": "Finite-Lived Intangible Assets, Gross",
"verboseLabel": "Cost"
}
}
},
"localname": "FiniteLivedIntangibleAssetsGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Finite-Lived Intangible Assets [Line Items]",
"terseLabel": "Finite-Lived Intangible Assets [Line Items]"
}
}
},
"localname": "FiniteLivedIntangibleAssetsLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsMajorClassNameDomain": {
"auth_ref": [
"r321",
"r327"
],
"lang": {
"en-us": {
"role": {
"documentation": "The major class of finite-lived intangible asset (for example, patents, trademarks, copyrights, etc.) A major class is composed of intangible assets that can be grouped together because they are similar, either by their nature or by their use in the operations of a company.",
"label": "Finite-Lived Intangible Assets, Major Class Name [Domain]",
"terseLabel": "Finite-Lived Intangible Assets, Major Class Name [Domain]"
}
}
},
"localname": "FiniteLivedIntangibleAssetsMajorClassNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsNet": {
"auth_ref": [
"r328",
"r755"
],
"calculation": {
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after amortization of assets, excluding financial assets and goodwill, lacking physical substance with a finite life.",
"label": "Finite-Lived Intangible Assets, Net",
"totalLabel": "Net"
}
}
},
"localname": "FiniteLivedIntangibleAssetsNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsPeriodIncreaseDecrease": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in carrying value of assets, excluding financial assets and goodwill, lacking physical substance with a finite life.",
"label": "Finite-Lived Intangible Assets, Period Increase (Decrease)",
"terseLabel": "Net change in acquired and in-licensed rights and patents"
}
}
},
"localname": "FiniteLivedIntangibleAssetsPeriodIncreaseDecrease",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FiniteLivedIntangibleAssetsRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Finite-lived Intangible Assets [Roll Forward]",
"terseLabel": "Finite-lived Intangible Assets [Roll Forward]"
}
}
},
"localname": "FiniteLivedIntangibleAssetsRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FinitelivedIntangibleAssetsAcquired1": {
"auth_ref": [
"r322"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase in assets, excluding financial assets, lacking physical substance with a definite life, from an acquisition.",
"label": "Finite-lived Intangible Assets Acquired",
"terseLabel": "Acquired intangible asset"
}
}
},
"localname": "FinitelivedIntangibleAssetsAcquired1",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ForeignCountryMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Designated tax departments of governments entitled to levy and collect income taxes from the entity outside the entity's country of domicile.",
"label": "Foreign Tax Authority [Member]",
"terseLabel": "Foreign Tax Authority"
}
}
},
"localname": "ForeignCountryMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ForeignCurrencyContractAssetFairValueDisclosure": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of asset contracts related to the exchange of different currencies, including, but not limited to, foreign currency options, forward contracts, and swaps.",
"label": "Foreign Currency Contract, Asset, Fair Value Disclosure",
"verboseLabel": "Derivative contracts"
}
}
},
"localname": "ForeignCurrencyContractAssetFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ForeignCurrencyContractsLiabilityFairValueDisclosure": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails": {
"order": 2.0,
"parentTag": "us-gaap_LiabilitiesFairValueDisclosure",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of liability contracts related to the exchange of different currencies, including, but not limited to, foreign currency options, forward (delivery or nondelivery) contracts, and swaps entered into.",
"label": "Foreign Currency Contracts, Liability, Fair Value Disclosure",
"verboseLabel": "Derivative contracts"
}
}
},
"localname": "ForeignCurrencyContractsLiabilityFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ForeignCurrencyTransactionGainLossBeforeTax": {
"auth_ref": [
"r681",
"r682",
"r683",
"r684"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails": {
"order": 1.0,
"parentTag": "us-gaap_NonoperatingIncomeExpense",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before tax of foreign currency transaction realized and unrealized gain (loss) recognized in the income statement.",
"label": "Foreign Currency Transaction Gain (Loss), before Tax",
"terseLabel": "Foreign exchange gains (losses), net"
}
}
},
"localname": "ForeignCurrencyTransactionGainLossBeforeTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ForeignCurrencyTransactionsAndTranslationsPolicyTextBlock": {
"auth_ref": [
"r158",
"r693"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for (1) transactions denominated in a currency other than the reporting enterprise's functional currency, (2) translating foreign currency financial statements that are incorporated into the financial statements of the reporting enterprise by consolidation, combination, or the equity method of accounting, and (3) remeasurement of the financial statements of a foreign reporting enterprise in a hyperinflationary economy.",
"label": "Foreign Currency Transactions and Translations Policy [Policy Text Block]",
"terseLabel": "Translation of Foreign Currencies"
}
}
},
"localname": "ForeignCurrencyTransactionsAndTranslationsPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ForeignExchangeContractMember": {
"auth_ref": [
"r428",
"r652"
],
"lang": {
"en-us": {
"role": {
"documentation": "Derivative instrument whose primary underlying risk is tied to foreign exchange rates.",
"label": "Foreign Exchange Contract [Member]",
"verboseLabel": "Foreign exchange contract"
}
}
},
"localname": "ForeignExchangeContractMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ForeignExchangeForwardMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Foreign exchange forward traded on an exchange (examples include but are not limited to the International Securities Exchange, Philadelphia Stock Exchange, or the Chicago Mercantile Exchange) for options or future contracts to buy or sell a certain currency, at a specified date, at a fixed exercise exchange rate.",
"label": "Foreign Exchange Forward [Member]",
"terseLabel": "Foreign Exchange Forward"
}
}
},
"localname": "ForeignExchangeForwardMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ForeignPlanMember": {
"auth_ref": [
"r479",
"r483",
"r489"
],
"lang": {
"en-us": {
"role": {
"documentation": "Location of employer sponsoring plan, designed to provide retirement benefits, not determined as principal place of business. Includes, but is not limited to, defined benefit and defined contribution plans.",
"label": "Foreign Plan [Member]",
"terseLabel": "Foreign Plan"
}
}
},
"localname": "ForeignPlanMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FurnitureAndFixturesGross": {
"auth_ref": [
"r340"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 6.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated depreciation of equipment commonly used in offices and stores that have no permanent connection to the structure of a building or utilities. Examples include, but are not limited to, desks, chairs, tables, and bookcases.",
"label": "Furniture and Fixtures, Gross",
"terseLabel": "Furniture and fixtures"
}
}
},
"localname": "FurnitureAndFixturesGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FurnitureAndFixturesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Equipment commonly used in offices and stores that have no permanent connection to the structure of a building or utilities. Examples include, but are not limited to, desks, chairs, tables, and bookcases.",
"label": "Furniture and Fixtures [Member]",
"terseLabel": "Furniture and Fixtures"
}
}
},
"localname": "FurnitureAndFixturesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_GainLossOnDerivativeInstrumentsNetPretax": {
"auth_ref": [
"r640"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate net gain (loss) on all derivative instruments recognized in earnings during the period, before tax effects.",
"label": "Gain (Loss) on Derivative Instruments, Net, Pretax",
"terseLabel": "Gain (loss) on derivative instruments, net, pretax"
}
}
},
"localname": "GainLossOnDerivativeInstrumentsNetPretax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GainLossOnForeignCurrencyDerivativeInstrumentsNotDesignatedAsHedgingInstruments": {
"auth_ref": [
"r640",
"r647"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of gain (loss) recognized in earnings in the period from the increase (decrease) in fair value of foreign currency derivatives not designated as hedging instruments.",
"label": "Gain (Loss) on Foreign Currency Derivative Instruments Not Designated as Hedging Instruments",
"verboseLabel": "Net gains (losses) of other income (expense) related to foreign currency forward contracts"
}
}
},
"localname": "GainLossOnForeignCurrencyDerivativeInstrumentsNotDesignatedAsHedgingInstruments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GainLossOnInvestmentsTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of realized and unrealized gain (loss) on investment in security.",
"label": "Gain (Loss) on Securities [Table Text Block]",
"terseLabel": "Gain (loss) on investments"
}
}
},
"localname": "GainLossOnInvestmentsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_GainLossOnSaleOfInvestments": {
"auth_ref": [
"r151"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails": {
"order": 5.0,
"parentTag": "us-gaap_NonoperatingIncomeExpense",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The net realized gain (loss) on investments sold during the period, not including gains (losses) on securities separately or otherwise categorized as trading, available-for-sale, or held-to-maturity, which, for cash flow reporting, is a component of proceeds from investing activities.",
"label": "Gain (Loss) on Sale of Investments",
"terseLabel": "Gain (loss) on investments, net"
}
}
},
"localname": "GainLossOnSaleOfInvestments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GeneralBusinessMember": {
"auth_ref": [
"r559"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tax credit carryforwards which include, but are not limited to, investment, alcohol fuels, low income housing, alternative fuels, alternative motor vehicles, hurricane relief, disabled access, renewable resources electricity production, employer social security, orphan drug, new markets, employer provided child care facilities and services, biodiesel fuels, low sulfur diesel fuel production, distilled spirits and the advanced nuclear power facilities credit.",
"label": "General Business Tax Credit Carryforward [Member]",
"terseLabel": "General Business"
}
}
},
"localname": "GeneralBusinessMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_Goodwill": {
"auth_ref": [
"r310",
"r311",
"r757"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 6.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after accumulated impairment loss of an asset representing future economic benefits arising from other assets acquired in a business combination that are not individually identified and separately recognized.",
"label": "Goodwill",
"periodEndLabel": "Goodwill, end of year",
"periodStartLabel": "Goodwill, beginning of year",
"verboseLabel": "Goodwill"
}
}
},
"localname": "Goodwill",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillChangesinGoodwillDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GoodwillAcquiredDuringPeriod": {
"auth_ref": [
"r313"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase in asset representing future economic benefits arising from other assets acquired in a business combination that are not individually identified and separately recognized resulting from a business combination.",
"label": "Goodwill, Acquired During Period",
"terseLabel": "Increase to goodwill",
"verboseLabel": "Increase to goodwill"
}
}
},
"localname": "GoodwillAcquiredDuringPeriod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillChangesinGoodwillDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GoodwillAndIntangibleAssetsDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Goodwill and Intangible Assets Disclosure [Abstract]",
"terseLabel": "Goodwill and Intangible Assets Disclosure [Abstract]"
}
}
},
"localname": "GoodwillAndIntangibleAssetsDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_GoodwillAndIntangibleAssetsDisclosureTextBlock": {
"auth_ref": [
"r336"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for goodwill and intangible assets.",
"label": "Goodwill and Intangible Assets Disclosure [Text Block]",
"verboseLabel": "Intangible Assets and Goodwill"
}
}
},
"localname": "GoodwillAndIntangibleAssetsDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwill"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_GoodwillAndIntangibleAssetsGoodwillPolicy": {
"auth_ref": [
"r158",
"r317"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for goodwill. This accounting policy also may address how an entity assesses and measures impairment of goodwill, how reporting units are determined, how goodwill is allocated to such units, and how the fair values of the reporting units are determined.",
"label": "Goodwill and Intangible Assets, Goodwill, Policy [Policy Text Block]",
"terseLabel": "Goodwill"
}
}
},
"localname": "GoodwillAndIntangibleAssetsGoodwillPolicy",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_GoodwillAndIntangibleAssetsIntangibleAssetsPolicy": {
"auth_ref": [
"r158",
"r325"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for intangible assets. This accounting policy may address both intangible assets subject to amortization and those that are not. The following also may be disclosed: (1) a description of intangible assets (2) the estimated useful lives of those assets (3) the amortization method used (4) how the entity assesses and measures impairment of such assets (5) how future cash flows are estimated (6) how the fair values of such asset are determined.",
"label": "Goodwill and Intangible Assets, Intangible Assets, Policy [Policy Text Block]",
"terseLabel": "Intangible Assets"
}
}
},
"localname": "GoodwillAndIntangibleAssetsIntangibleAssetsPolicy",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_GoodwillImpairedAccumulatedImpairmentLoss": {
"auth_ref": [
"r312",
"r316"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of accumulated impairment loss for an asset representing future economic benefits arising from other assets acquired in a business combination that are not individually identified and separately recognized.",
"label": "Goodwill, Impaired, Accumulated Impairment Loss",
"terseLabel": "Accumulated impairment losses related to goodwill"
}
}
},
"localname": "GoodwillImpairedAccumulatedImpairmentLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GoodwillLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Goodwill [Line Items]",
"terseLabel": "Goodwill [Line Items]"
}
}
},
"localname": "GoodwillLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_GoodwillOtherIncreaseDecrease": {
"auth_ref": [
"r315"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease), classified as other, of an asset representing the future economic benefits from other assets acquired in a business combination that are not individually identified and separately recognized.",
"label": "Goodwill, Other Increase (Decrease)",
"terseLabel": "Other"
}
}
},
"localname": "GoodwillOtherIncreaseDecrease",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillChangesinGoodwillDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GoodwillRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Goodwill [Roll Forward]",
"terseLabel": "Goodwill [Roll Forward]"
}
}
},
"localname": "GoodwillRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillChangesinGoodwillDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_GoodwillWrittenOffRelatedToSaleOfBusinessUnit": {
"auth_ref": [
"r314"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of divestiture of an asset representing future economic benefits arising from other assets acquired in a business combination that are not individually identified and separately recognized.",
"label": "Goodwill, Written off Related to Sale of Business Unit",
"negatedLabel": "Elimination of goodwill allocated to Hiller\u00f8d, Denmark manufacturing operations"
}
}
},
"localname": "GoodwillWrittenOffRelatedToSaleOfBusinessUnit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillChangesinGoodwillDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GrossProfit": {
"auth_ref": [
"r120",
"r163",
"r239",
"r243",
"r246",
"r249",
"r252",
"r293",
"r680"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate revenue less cost of goods and services sold or operating expenses directly attributable to the revenue generation activity.",
"label": "Gross Profit",
"verboseLabel": "Gross profit"
}
}
},
"localname": "GrossProfit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GuaranteesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Guarantees [Abstract]",
"terseLabel": "Guarantees [Abstract]"
}
}
},
"localname": "GuaranteesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_GuaranteesTextBlock": {
"auth_ref": [
"r363"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for each guarantee obligation, or each group of similar guarantee obligations, including (a) the nature of the guarantee, including its term, how it arose, and the events or circumstances that would require the guarantor to perform under the guarantee; (b) the maximum potential amount of future payments (undiscounted) the guarantor could be required to make under the guarantee; (c) the current carrying amount of the liability, if any, for the guarantor's obligations under the guarantee; and (d) the nature of any recourse provisions under the guarantee, and any assets held either as collateral or by third parties, and any relevant related party disclosure. Excludes disclosures about product warranties.",
"label": "Guarantees [Text Block]",
"verboseLabel": "Guarantees"
}
}
},
"localname": "GuaranteesTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Guarantees"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_HedgingDesignationAxis": {
"auth_ref": [
"r637",
"r655"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by designation of purpose of derivative instrument.",
"label": "Hedging Designation [Axis]",
"terseLabel": "Hedging Designation [Axis]"
}
}
},
"localname": "HedgingDesignationAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_HedgingDesignationDomain": {
"auth_ref": [
"r637"
],
"lang": {
"en-us": {
"role": {
"documentation": "Designation of purpose of derivative instrument.",
"label": "Hedging Designation [Domain]",
"terseLabel": "Hedging Designation [Domain]"
}
}
},
"localname": "HedgingDesignationDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsForeignCurrencyForwardContractsEnteredintoHedgeForecastedRevenuesDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_HedgingRelationshipDomain": {
"auth_ref": [
"r637"
],
"lang": {
"en-us": {
"role": {
"documentation": "Nature or intent of a hedge.",
"label": "Hedging Relationship [Domain]",
"terseLabel": "Hedging Relationship [Domain]"
}
}
},
"localname": "HedgingRelationshipDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ImpairmentOfIntangibleAssetsExcludingGoodwill": {
"auth_ref": [
"r151",
"r333"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 17.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of impairment loss recognized in the period resulting from the write-down of the carrying amount of an intangible asset (excluding goodwill) to fair value.",
"label": "Impairment of Intangible Assets (Excluding Goodwill)",
"terseLabel": "Impairment of intangible assets"
}
}
},
"localname": "ImpairmentOfIntangibleAssetsExcludingGoodwill",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ImpairmentOfIntangibleAssetsIndefinitelivedExcludingGoodwill": {
"auth_ref": [
"r151",
"r333"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of impairment loss resulting from write-down of assets, excluding financial assets and goodwill, lacking physical substance and having a projected indefinite period of benefit to fair value.",
"label": "Impairment of Intangible Assets, Indefinite-lived (Excluding Goodwill)",
"terseLabel": "Impairment"
}
}
},
"localname": "ImpairmentOfIntangibleAssetsIndefinitelivedExcludingGoodwill",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ImpairmentOrDisposalOfLongLivedAssetsPolicyTextBlock": {
"auth_ref": [
"r158",
"r337",
"r349"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for recognizing and measuring the impairment of long-lived assets. An entity also may disclose its accounting policy for long-lived assets to be sold. This policy excludes goodwill and intangible assets.",
"label": "Impairment or Disposal of Long-Lived Assets, Policy [Policy Text Block]",
"terseLabel": "Impairment of Long-Lived Assets"
}
}
},
"localname": "ImpairmentOrDisposalOfLongLivedAssetsPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_InProcessResearchAndDevelopmentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "In process investigation of new knowledge useful in developing new product or service or new process or technique or improvement to existing product or process, and translation of knowledge into plan or design for new product or process or for improvement to existing product or process.",
"label": "In Process Research and Development [Member]",
"verboseLabel": "In-process research and development"
}
}
},
"localname": "InProcessResearchAndDevelopmentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_InProcessResearchAndDevelopmentPolicy": {
"auth_ref": [
"r62",
"r158",
"r533"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for costs assigned to identifiable tangible and intangible assets of an acquired entity to be used in the research and development activities of the combined enterprise. An entity also may disclose the appraisal method or significant assumptions used to value acquired research and development assets.",
"label": "In Process Research and Development, Policy [Policy Text Block]",
"terseLabel": "Acquired In-process Research and Development (IPR&D)"
}
}
},
"localname": "InProcessResearchAndDevelopmentPolicy",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_IncomeLossFromContinuingOperationsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Income (Loss) from Continuing Operations, Net of Tax, Including Portion Attributable to Noncontrolling Interest [Abstract]",
"terseLabel": "Income (Loss) from Continuing Operations, Including Portion Attributable to Noncontrolling Interest [Abstract]"
}
}
},
"localname": "IncomeLossFromContinuingOperationsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesDomestic": {
"auth_ref": [
"r167",
"r574"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 1.0,
"parentTag": "us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestments",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The portion of earnings or loss from continuing operations before income taxes that is attributable to domestic operations.",
"label": "Income (Loss) from Continuing Operations before Income Taxes, Domestic",
"terseLabel": "Domestic"
}
}
},
"localname": "IncomeLossFromContinuingOperationsBeforeIncomeTaxesDomestic",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesForeign": {
"auth_ref": [
"r167",
"r574"
],
"calculation": {
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": 2.0,
"parentTag": "us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestments",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The portion of earnings or loss from continuing operations before income taxes that is attributable to foreign operations, which is defined as Income or Loss generated from operations located outside the entity's country of domicile.",
"label": "Income (Loss) from Continuing Operations before Income Taxes, Foreign",
"terseLabel": "Foreign"
}
}
},
"localname": "IncomeLossFromContinuingOperationsBeforeIncomeTaxesForeign",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestments": {
"auth_ref": [
"r167",
"r239",
"r243",
"r246",
"r249",
"r252"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 1.0,
"parentTag": "us-gaap_ProfitLoss",
"weight": 1.0
},
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of income (loss) from continuing operations before deduction of income tax expense (benefit) and income (loss) attributable to noncontrolling interest, and addition of income (loss) from equity method investments.",
"label": "Income (Loss) from Continuing Operations before Equity Method Investments, Income Taxes, Noncontrolling Interest",
"totalLabel": "Income before income tax expense and equity in loss of investee, net of tax"
}
}
},
"localname": "IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestmentsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Income (Loss) from Continuing Operations before Equity Method Investments, Income Taxes, Noncontrolling Interest [Abstract]",
"terseLabel": "Income before income taxes (benefit):"
}
}
},
"localname": "IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestmentsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeLossFromEquityMethodInvestments": {
"auth_ref": [
"r111",
"r151",
"r237",
"r289",
"r770",
"r801"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 13.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of income (loss) for proportionate share of equity method investee's income (loss).",
"label": "Income (Loss) from Equity Method Investments",
"negatedTerseLabel": "Loss on equity method investment",
"terseLabel": "Eisai's share of aducanumab milestone expense reflected in collaboration profit sharing in our consolidated statements of income"
}
}
},
"localname": "IncomeLossFromEquityMethodInvestments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeLossFromEquityMethodInvestmentsNetOfDividendsOrDistributions": {
"auth_ref": [
"r151"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "This element represents the undistributed income (or loss) of equity method investments, net of dividends or other distributions received from unconsolidated subsidiaries, certain corporate joint ventures, and certain noncontrolled corporations; such investments are accounted for under the equity method of accounting. This element excludes distributions that constitute a return of investment, which are classified as investing activities.",
"label": "Income (Loss) from Equity Method Investments, Net of Dividends or Distributions",
"terseLabel": "Net profits on investment"
}
}
},
"localname": "IncomeLossFromEquityMethodInvestmentsNetOfDividendsOrDistributions",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeLossFromIndividuallySignificantComponentDisposedOfOrHeldForSaleExcludingDiscontinuedOperationsAttributableToParentBeforeIncomeTax": {
"auth_ref": [
"r347"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 1.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before tax of income (loss) from an individually significant component disposed of or held-for-sale attributable to parent. Excludes discontinued operations.",
"label": "Income (Loss) from Individually Significant Component Disposed of or Held-for-sale, Excluding Discontinued Operations, Attributable to Parent, before Income Tax",
"negatedTerseLabel": "(Gain)/loss on divestiture of Hillerod, Denmark manufactuing operations"
}
}
},
"localname": "IncomeLossFromIndividuallySignificantComponentDisposedOfOrHeldForSaleExcludingDiscontinuedOperationsAttributableToParentBeforeIncomeTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeStatementAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Income Statement [Abstract]",
"terseLabel": "Income Statement [Abstract]"
}
}
},
"localname": "IncomeStatementAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_IncomeStatementBalanceSheetAndAdditionalDisclosuresByDisposalGroupsIncludingDiscontinuedOperationsAxis": {
"auth_ref": [
"r480",
"r486"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by name of disposal group.",
"label": "Disposal Group Name [Axis]",
"terseLabel": "Disposal Group Name [Axis]"
}
}
},
"localname": "IncomeStatementBalanceSheetAndAdditionalDisclosuresByDisposalGroupsIncludingDiscontinuedOperationsAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeStatementBalanceSheetAndAdditionalDisclosuresByDisposalGroupsIncludingDiscontinuedOperationsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Income Statement, Balance Sheet and Additional Disclosures by Disposal Groups, Including Discontinued Operations [Line Items]",
"terseLabel": "Income Statement, Balance Sheet and Additional Disclosures by Disposal Groups, Including Discontinued Operations [Line Items]"
}
}
},
"localname": "IncomeStatementBalanceSheetAndAdditionalDisclosuresByDisposalGroupsIncludingDiscontinuedOperationsLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeStatementBalanceSheetAndAdditionalDisclosuresByDisposalGroupsIncludingDiscontinuedOperationsTable": {
"auth_ref": [
"r1",
"r7",
"r8",
"r9",
"r10",
"r11",
"r12",
"r16",
"r18",
"r19",
"r20",
"r346",
"r348"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about a disposal group. Includes, but is not limited to, a discontinued operation, disposal classified as held-for-sale or disposed of by means other than sale or disposal of an individually significant component.",
"label": "Disposal Groups, Including Discontinued Operations [Table]",
"terseLabel": "Disposal Groups, Including Discontinued Operations [Table]"
}
}
},
"localname": "IncomeStatementBalanceSheetAndAdditionalDisclosuresByDisposalGroupsIncludingDiscontinuedOperationsTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DivestituresDetails",
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeStatementLocationAxis": {
"auth_ref": [
"r344"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by location in the income statement.",
"label": "Income Statement Location [Axis]",
"terseLabel": "Income Statement Location [Axis]",
"verboseLabel": "Income Statement Location [Axis]"
}
}
},
"localname": "IncomeStatementLocationAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeStatementLocationDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Location in the income statement.",
"label": "Income Statement Location [Domain]",
"terseLabel": "Income Statement Location [Domain]",
"verboseLabel": "Income Statement Location [Domain]"
}
}
},
"localname": "IncomeStatementLocationDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_IncomeTaxAuthorityAxis": {
"auth_ref": [
"r544"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by tax jurisdiction.",
"label": "Income Tax Authority [Axis]",
"terseLabel": "Income Tax Authority [Axis]"
}
}
},
"localname": "IncomeTaxAuthorityAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeTaxAuthorityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Agency, division or body classification that levies income taxes, examines tax returns for compliance, or grants exemptions from or makes other decisions pertaining to income taxes.",
"label": "Income Tax Authority [Domain]",
"terseLabel": "Income Tax Authority [Domain]"
}
}
},
"localname": "IncomeTaxAuthorityDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_IncomeTaxDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Income Tax Disclosure [Abstract]",
"terseLabel": "Income Tax Disclosure [Abstract]"
}
}
},
"localname": "IncomeTaxDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_IncomeTaxDisclosureTextBlock": {
"auth_ref": [
"r541",
"r552",
"r557",
"r569",
"r576",
"r579",
"r580",
"r581"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for income taxes. Disclosures may include net deferred tax liability or asset recognized in an enterprise's statement of financial position, net change during the year in the total valuation allowance, approximate tax effect of each type of temporary difference and carryforward that gives rise to a significant portion of deferred tax liabilities and deferred tax assets, utilization of a tax carryback, and tax uncertainties information.",
"label": "Income Tax Disclosure [Text Block]",
"verboseLabel": "Income Taxes"
}
}
},
"localname": "IncomeTaxDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxes"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_IncomeTaxExpenseBenefit": {
"auth_ref": [
"r165",
"r192",
"r193",
"r238",
"r539",
"r570",
"r577",
"r810"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 2.0,
"parentTag": "us-gaap_ProfitLoss",
"weight": -1.0
},
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of current income tax expense (benefit) and deferred income tax expense (benefit) pertaining to continuing operations.",
"label": "Income Tax Expense (Benefit)",
"terseLabel": "Income tax benefit (expense)",
"totalLabel": "Total income tax expense",
"verboseLabel": "Income tax expense"
}
}
},
"localname": "IncomeTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails",
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeTaxExpenseBenefitContinuingOperationsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Income Tax Expense (Benefit), Continuing Operations [Abstract]",
"terseLabel": "Income tax expense (benefit):"
}
}
},
"localname": "IncomeTaxExpenseBenefitContinuingOperationsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesIncomeBeforeIncomeTaxProvisionandtheIncomeTaxExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeTaxExpenseBenefitContinuingOperationsAdjustmentOfDeferredTaxAssetLiability": {
"auth_ref": [
"r564"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of income tax expense (benefit) from continuing operations attributable to an adjustment of a deferred tax liability or asset for enacted changes in tax laws or rates or a change in the tax status of the entity.",
"label": "Income Tax Expense (Benefit), Continuing Operations, Adjustment of Deferred Tax (Asset) Liability",
"terseLabel": "Income tax expense (benefit)"
}
}
},
"localname": "IncomeTaxExpenseBenefitContinuingOperationsAdjustmentOfDeferredTaxAssetLiability",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeTaxPolicyTextBlock": {
"auth_ref": [
"r107",
"r158",
"r535",
"r536",
"r552",
"r553",
"r556",
"r565",
"r843"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for income taxes, which may include its accounting policies for recognizing and measuring deferred tax assets and liabilities and related valuation allowances, recognizing investment tax credits, operating loss carryforwards, tax credit carryforwards, and other carryforwards, methodologies for determining its effective income tax rate and the characterization of interest and penalties in the financial statements.",
"label": "Income Tax, Policy [Policy Text Block]",
"terseLabel": "Income Taxes"
}
}
},
"localname": "IncomeTaxPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_IncomeTaxesPaid": {
"auth_ref": [
"r148",
"r155"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of cash paid during the current period to foreign, federal, state, and local authorities as taxes on income.",
"label": "Income Taxes Paid",
"terseLabel": "Income taxes paid"
}
}
},
"localname": "IncomeTaxesPaid",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeTaxesPaidNet": {
"auth_ref": [
"r155"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of cash paid during the current period to foreign, federal, state, and local authorities as taxes on income, net of any cash received during the current period as refunds for the overpayment of taxes.",
"label": "Income Taxes Paid, Net",
"terseLabel": "Income taxes"
}
}
},
"localname": "IncomeTaxesPaidNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailSupplementalCashFlowInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInAccountsReceivable": {
"auth_ref": [
"r150"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 7.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The increase (decrease) during the reporting period in amount due within one year (or one business cycle) from customers for the credit sale of goods and services.",
"label": "Increase (Decrease) in Accounts Receivable",
"negatedLabel": "Accounts receivable"
}
}
},
"localname": "IncreaseDecreaseInAccountsReceivable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInAccruedIncomeTaxesPayable": {
"auth_ref": [
"r150"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 8.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The increase (decrease) during the period in the amount due for taxes based on the reporting entity's earnings or attributable to the entity's income earning process (business presence) within a given jurisdiction.",
"label": "Increase (Decrease) in Income Taxes Payable",
"terseLabel": "Income tax assets and liabilities"
}
}
},
"localname": "IncreaseDecreaseInAccruedIncomeTaxesPayable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInAccruedLiabilities": {
"auth_ref": [
"r150"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 3.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The increase (decrease) during the reporting period in the aggregate amount of expenses incurred but not yet paid.",
"label": "Increase (Decrease) in Accrued Liabilities",
"verboseLabel": "Accrued expenses and other current liabilities"
}
}
},
"localname": "IncreaseDecreaseInAccruedLiabilities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInEquitySecuritiesFvNi": {
"auth_ref": [
"r143"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 6.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) of investment in equity security measured at fair value with change in fair value recognized in net income (FV-NI).",
"label": "Increase (Decrease) in Equity Securities, FV-NI",
"negatedTerseLabel": "Unrealized (gain) loss on strategic investments"
}
}
},
"localname": "IncreaseDecreaseInEquitySecuritiesFvNi",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInInventories": {
"auth_ref": [
"r150"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 15.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The increase (decrease) during the reporting period in the aggregate value of all inventory held by the reporting entity, associated with underlying transactions that are classified as operating activities.",
"label": "Increase (Decrease) in Inventories",
"negatedTerseLabel": "Inventory"
}
}
},
"localname": "IncreaseDecreaseInInventories",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInOperatingCapitalAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Increase (Decrease) in Operating Capital [Abstract]",
"verboseLabel": "Changes in operating assets and liabilities, net:"
}
}
},
"localname": "IncreaseDecreaseInOperatingCapitalAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncreaseDecreaseInOtherOperatingCapitalNet": {
"auth_ref": [
"r150"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 14.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in operating assets after deduction of operating liabilities classified as other.",
"label": "Increase (Decrease) in Other Operating Assets and Liabilities, Net",
"negatedTerseLabel": "Due from anti-CD20 therapeutic programs"
}
}
},
"localname": "IncreaseDecreaseInOtherOperatingCapitalNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInOtherOperatingLiabilities": {
"auth_ref": [
"r150"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 10.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in operating liabilities classified as other.",
"label": "Increase (Decrease) in Other Operating Liabilities",
"terseLabel": "Other changes in operating assets and liabilities, net"
}
}
},
"localname": "IncreaseDecreaseInOtherOperatingLiabilities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInStockholdersEquityRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Increase (Decrease) in Stockholders' Equity [Roll Forward]",
"terseLabel": "Increase (Decrease) in Stockholders' Equity [Roll Forward]"
}
}
},
"localname": "IncreaseDecreaseInStockholdersEquityRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncrementalCommonSharesAttributableToShareBasedPaymentArrangements": {
"auth_ref": [
"r198",
"r199",
"r200",
"r206"
],
"calculation": {
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails": {
"order": 1.0,
"parentTag": "us-gaap_WeightedAverageNumberDilutedSharesOutstandingAdjustment",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Additional shares included in the calculation of diluted EPS as a result of the potentially dilutive effect of share based payment arrangements using the treasury stock method.",
"label": "Incremental Common Shares Attributable to Dilutive Effect of Share-based Payment Arrangements",
"verboseLabel": "Stock options and employee stock purchase plan (in shares)"
}
}
},
"localname": "IncrementalCommonSharesAttributableToShareBasedPaymentArrangements",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_IndefiniteLivedIntangibleAssetsByMajorClassAxis": {
"auth_ref": [
"r323",
"r331"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type or class of assets, excluding financial assets and goodwill, lacking physical substance and having a projected indefinite period of benefit.",
"label": "Indefinite-lived Intangible Assets [Axis]",
"terseLabel": "Indefinite-lived Intangible Assets by Major Class [Axis]"
}
}
},
"localname": "IndefiniteLivedIntangibleAssetsByMajorClassAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IndefiniteLivedIntangibleAssetsExcludingGoodwill": {
"auth_ref": [
"r331"
],
"calculation": {
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails": {
"order": 1.0,
"parentTag": "us-gaap_IntangibleAssetsGrossExcludingGoodwill",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of assets, excluding financial assets and goodwill, lacking physical substance and having a projected indefinite period of benefit.",
"label": "Indefinite-lived Intangible Assets (Excluding Goodwill)",
"terseLabel": "Intangible assets (excluding goodwill)",
"verboseLabel": "Cost and net"
}
}
},
"localname": "IndefiniteLivedIntangibleAssetsExcludingGoodwill",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IndefiniteLivedIntangibleAssetsMajorClassNameDomain": {
"auth_ref": [
"r323",
"r331"
],
"lang": {
"en-us": {
"role": {
"documentation": "The major class of indefinite-lived intangible asset (for example, trade names, etc. but not all-inclusive), excluding goodwill. A major class is composed of intangible assets that can be grouped together because they are similar, either by their nature or by their use in the operations of the company.",
"label": "Indefinite-lived Intangible Assets, Major Class Name [Domain]",
"terseLabel": "Indefinite-lived Intangible Assets, Major Class Name [Domain]"
}
}
},
"localname": "IndefiniteLivedIntangibleAssetsMajorClassNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_IntangibleAssetsGrossExcludingGoodwill": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails": {
"order": 1.0,
"parentTag": "us-gaap_IntangibleAssetsNetExcludingGoodwill",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated amortization of intangible assets, excluding goodwill.",
"label": "Intangible Assets, Gross (Excluding Goodwill)",
"terseLabel": "Beginning Book Value",
"totalLabel": "Total intangible assets"
}
}
},
"localname": "IntangibleAssetsGrossExcludingGoodwill",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IntangibleAssetsNetExcludingGoodwill": {
"auth_ref": [
"r319",
"r326"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 5.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
},
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying amounts of all intangible assets, excluding goodwill, as of the balance sheet date, net of accumulated amortization and impairment charges.",
"label": "Intangible Assets, Net (Excluding Goodwill)",
"periodEndLabel": "Ending Book Value",
"terseLabel": "Intangible Assets, Net (Excluding Goodwill)",
"totalLabel": "Total intangible assets",
"verboseLabel": "Intangible assets, net"
}
}
},
"localname": "IntangibleAssetsNetExcludingGoodwill",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/FairValueMeasurementsGainsandLossesonAssetsMeasuredatFairValueDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails",
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IntellectualPropertyMember": {
"auth_ref": [
"r590"
],
"lang": {
"en-us": {
"role": {
"documentation": "Intangible asset arising from original creative thought. Include, but is not limited to, trademarks, patents, and copyrights.",
"label": "Intellectual Property [Member]",
"terseLabel": "Intellectual Property"
}
}
},
"localname": "IntellectualPropertyMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_InterestCostsCapitalized": {
"auth_ref": [
"r695"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of interest capitalized during the period.",
"label": "Interest Costs Capitalized",
"terseLabel": "Interest costs capitalized"
}
}
},
"localname": "InterestCostsCapitalized",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InterestExpense": {
"auth_ref": [
"r109",
"r235",
"r694",
"r698",
"r777"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails": {
"order": 4.0,
"parentTag": "us-gaap_NonoperatingIncomeExpense",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of the cost of borrowed funds accounted for as interest expense.",
"label": "Interest Expense",
"negatedLabel": "Interest expense"
}
}
},
"localname": "InterestExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InterestExpenseMember": {
"auth_ref": [
"r653"
],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing interest expense.",
"label": "Interest Expense [Member]",
"terseLabel": "Interest Expense"
}
}
},
"localname": "InterestExpenseMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_InterestPaidNet": {
"auth_ref": [
"r142",
"r147",
"r155"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash paid for interest, excluding capitalized interest, classified as operating activity. Includes, but is not limited to, payment to settle zero-coupon bond for accreted interest of debt discount and debt instrument with insignificant coupon interest rate in relation to effective interest rate of borrowing attributable to accreted interest of debt discount.",
"label": "Interest Paid, Excluding Capitalized Interest, Operating Activities",
"terseLabel": "Interest"
}
}
},
"localname": "InterestPaidNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailSupplementalCashFlowInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InterestRateSwapMember": {
"auth_ref": [
"r651"
],
"lang": {
"en-us": {
"role": {
"documentation": "Forward based contracts in which two parties agree to swap periodic payments that are fixed at the outset of the swap contract with variable payments based on a market interest rate (index rate) over a specified period.",
"label": "Interest Rate Swap [Member]",
"terseLabel": "Interest rate swap"
}
}
},
"localname": "InterestRateSwapMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_InventoriesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tangible personal property held for sale in the ordinary course of business, or in process of, or to be consumed in, production for sale.",
"label": "Inventories [Member]",
"terseLabel": "Inventories"
}
}
},
"localname": "InventoriesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_InventoryDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Inventory Disclosure [Abstract]",
"terseLabel": "Inventory Disclosure [Abstract]"
}
}
},
"localname": "InventoryDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_InventoryDisclosureTextBlock": {
"auth_ref": [
"r309"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for inventory. Includes, but is not limited to, the basis of stating inventory, the method of determining inventory cost, the classes of inventory, and the nature of the cost elements included in inventory.",
"label": "Inventory Disclosure [Text Block]",
"verboseLabel": "Inventory"
}
}
},
"localname": "InventoryDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Inventory"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_InventoryFinishedGoodsNetOfReserves": {
"auth_ref": [
"r51",
"r308"
],
"calculation": {
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails": {
"order": 1.0,
"parentTag": "biib_Inventorynetcurrentandnoncurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying amount, net of valuation reserves and adjustments, as of the balance sheet date of merchandise or goods held by the company that are readily available for sale.",
"label": "Inventory, Finished Goods, Net of Reserves",
"verboseLabel": "Finished goods"
}
}
},
"localname": "InventoryFinishedGoodsNetOfReserves",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InventoryLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Inventory [Line Items]",
"terseLabel": "Inventory [Line Items]"
}
}
},
"localname": "InventoryLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_InventoryNet": {
"auth_ref": [
"r22",
"r80"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 1.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after valuation and LIFO reserves of inventory expected to be sold, or consumed within one year or operating cycle, if longer.",
"label": "Inventory, Net",
"verboseLabel": "Inventory"
}
}
},
"localname": "InventoryNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InventoryPolicyTextBlock": {
"auth_ref": [
"r32",
"r81",
"r158",
"r213",
"r305",
"r307",
"r309"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of inventory accounting policy for inventory classes, including, but not limited to, basis for determining inventory amounts, methods by which amounts are added and removed from inventory classes, loss recognition on impairment of inventories, and situations in which inventories are stated above cost.",
"label": "Inventory, Policy [Policy Text Block]",
"terseLabel": "Inventory"
}
}
},
"localname": "InventoryPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_InventoryRawMaterialsNetOfReserves": {
"auth_ref": [
"r53",
"r308"
],
"calculation": {
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails": {
"order": 3.0,
"parentTag": "biib_Inventorynetcurrentandnoncurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying amount, net of valuation reserves and adjustments, as of the balance sheet date of unprocessed items to be consumed in the manufacturing or production process.",
"label": "Inventory, Raw Materials, Net of Reserves",
"verboseLabel": "Raw materials"
}
}
},
"localname": "InventoryRawMaterialsNetOfReserves",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InventoryWorkInProcessNetOfReserves": {
"auth_ref": [
"r52",
"r308"
],
"calculation": {
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails": {
"order": 2.0,
"parentTag": "biib_Inventorynetcurrentandnoncurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying amount, net of reserves and adjustments, as of the balance sheet date of merchandise or goods which are partially completed. This inventory is generally comprised of raw materials, labor and factory overhead costs, which require further materials, labor and overhead to be converted into finished goods, and which generally require the use of estimates to determine percentage complete and pricing.",
"label": "Inventory, Work in Process, Net of Reserves",
"verboseLabel": "Work in process"
}
}
},
"localname": "InventoryWorkInProcessNetOfReserves",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryComponentsofInventoryDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InventoryWriteDown": {
"auth_ref": [
"r306"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of loss from reductions in inventory due to subsequent measurement adjustments, including, but not limited to, physical deterioration, obsolescence, or changes in price levels.",
"label": "Inventory Write-down",
"verboseLabel": "Write-downs on excess, obsolete, unmarketable or other inventory"
}
}
},
"localname": "InventoryWriteDown",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InvestmentIncomeInterest": {
"auth_ref": [
"r123",
"r234"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails": {
"order": 3.0,
"parentTag": "us-gaap_NonoperatingIncomeExpense",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accretion (amortization) of purchase discount (premium) of interest income on nonoperating securities.",
"label": "Investment Income, Interest",
"terseLabel": "Interest income"
}
}
},
"localname": "InvestmentIncomeInterest",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InvestmentPolicyTextBlock": {
"auth_ref": [
"r291",
"r808"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for investment in financial asset.",
"label": "Investment, Policy [Policy Text Block]",
"terseLabel": "Marketable Securities and Other Investments"
}
}
},
"localname": "InvestmentPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_InvestmentTypeAxis": {
"auth_ref": [
"r825",
"r826",
"r827",
"r828",
"r829",
"r830",
"r831",
"r832",
"r833",
"r834",
"r835",
"r836",
"r837",
"r838",
"r839"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of investments.",
"label": "Investment Type [Axis]",
"terseLabel": "Investment Type [Axis]"
}
}
},
"localname": "InvestmentTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_InvestmentTypeCategorizationMember": {
"auth_ref": [
"r825",
"r826",
"r827",
"r828",
"r829",
"r830",
"r831",
"r832",
"r833",
"r834",
"r835",
"r836",
"r837",
"r838",
"r839"
],
"lang": {
"en-us": {
"role": {
"documentation": "Asset obtained to generate income or appreciate in value.",
"label": "Investments [Domain]",
"terseLabel": "Investments [Domain]"
}
}
},
"localname": "InvestmentTypeCategorizationMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_InvestmentsAllOtherInvestmentsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Investments, All Other Investments [Abstract]",
"terseLabel": "Investments, All Other Investments [Abstract]"
}
}
},
"localname": "InvestmentsAllOtherInvestmentsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_InvestmentsClassifiedByContractualMaturityDateTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of maturities of an entity's investments as well as any other information pertinent to the investments.",
"label": "Investments Classified by Contractual Maturity Date [Table Text Block]",
"verboseLabel": "Summary of contractual maturities: available-for-sale securities"
}
}
},
"localname": "InvestmentsClassifiedByContractualMaturityDateTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_InvestmentsFairValueDisclosure": {
"auth_ref": [
"r663"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of investment securities, including, but not limited to, marketable securities, derivative financial instruments, and investments accounted for under the equity method.",
"label": "Investments, Fair Value Disclosure",
"verboseLabel": "Marketable equity securities"
}
}
},
"localname": "InvestmentsFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_Land": {
"auth_ref": [
"r26",
"r60"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 1.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated depletion of real estate held for productive use, excluding land held for sale.",
"label": "Land",
"terseLabel": "Land"
}
}
},
"localname": "Land",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LeaseContractualTermAxis": {
"auth_ref": [
"r706"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by contractual term of lease arrangement.",
"label": "Lease Contractual Term [Axis]",
"terseLabel": "Property, Plant and Equipment, Type [Axis]"
}
}
},
"localname": "LeaseContractualTermAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LeaseContractualTermDomain": {
"auth_ref": [
"r706"
],
"lang": {
"en-us": {
"role": {
"documentation": "Contractual term of lease arrangement.",
"label": "Lease Contractual Term [Domain]",
"terseLabel": "Lease Arrangement, Type [Domain]"
}
}
},
"localname": "LeaseContractualTermDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LeaseCost": {
"auth_ref": [
"r714",
"r716"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lease cost recognized by lessee for lease contract.",
"label": "Lease, Cost",
"terseLabel": "Net lease cost"
}
}
},
"localname": "LeaseCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LeaseCostTableTextBlock": {
"auth_ref": [
"r714"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of lessee's lease cost. Includes, but is not limited to, interest expense for finance lease, amortization of right-of-use asset for finance lease, operating lease cost, short-term lease cost, variable lease cost and sublease income.",
"label": "Lease, Cost [Table Text Block]",
"terseLabel": "Operating lease costs"
}
}
},
"localname": "LeaseCostTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_LeaseholdImprovementsGross": {
"auth_ref": [
"r27",
"r340"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 3.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated depreciation of additions or improvements to assets held under a lease arrangement.",
"label": "Leasehold Improvements, Gross",
"terseLabel": "Leasehold improvements"
}
}
},
"localname": "LeaseholdImprovementsGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LeasesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Leases [Abstract]",
"terseLabel": "Leases [Abstract]"
}
}
},
"localname": "LeasesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_LegalMattersAndContingenciesTextBlock": {
"auth_ref": [
"r361"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for legal proceedings, legal contingencies, litigation, regulatory and environmental matters and other contingencies.",
"label": "Legal Matters and Contingencies [Text Block]",
"verboseLabel": "Litigation"
}
}
},
"localname": "LegalMattersAndContingenciesTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Litigation"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_LesseeLeasesPolicyTextBlock": {
"auth_ref": [
"r704"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for leasing arrangement entered into by lessee.",
"label": "Lessee, Leases [Policy Text Block]",
"terseLabel": "Leases"
}
}
},
"localname": "LesseeLeasesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityMaturityTableTextBlock": {
"auth_ref": [
"r715"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of undiscounted cash flows of lessee's operating lease liability. Includes, but is not limited to, reconciliation of undiscounted cash flows to operating lease liability recognized in statement of financial position.",
"label": "Lessee, Operating Lease, Liability, Maturity [Table Text Block]",
"terseLabel": "Operating lease liability maturity"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails_1": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease.",
"label": "Lessee, Operating Lease, Liability, to be Paid",
"totalLabel": "Total lease payments"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueAfterYearFive": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails": {
"order": 2.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease due after fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, after Year Five",
"terseLabel": "Thereafter"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueAfterYearFive",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails": {
"order": 5.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year One",
"terseLabel": "2021"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearFive": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails": {
"order": 4.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Five",
"terseLabel": "2025"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearFive",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearFour": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails": {
"order": 6.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in fourth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Four",
"terseLabel": "2024"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearFour",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearThree": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails": {
"order": 1.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in third fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Three",
"terseLabel": "2023"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearThree",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearTwo": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails": {
"order": 3.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in second fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Two",
"terseLabel": "2022"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearTwo",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityUndiscountedExcessAmount": {
"auth_ref": [
"r715"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails_1": {
"order": 2.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payments in excess of discounted obligation for lease payments for operating lease.",
"label": "Lessee, Operating Lease, Liability, Undiscounted Excess Amount",
"terseLabel": "Less: interest"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityUndiscountedExcessAmount",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseRenewalTerm": {
"auth_ref": [
"r705"
],
"lang": {
"en-us": {
"role": {
"documentation": "Term of lessee's operating lease renewal, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Lessee, Operating Lease, Renewal Term",
"terseLabel": "Operating lease, renewal term"
}
}
},
"localname": "LesseeOperatingLeaseRenewalTerm",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_LesseeOperatingLeaseTermOfContract": {
"auth_ref": [
"r705"
],
"lang": {
"en-us": {
"role": {
"documentation": "Term of lessee's operating lease, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Lessee, Operating Lease, Term of Contract",
"terseLabel": "Operating lease, contract term"
}
}
},
"localname": "LesseeOperatingLeaseTermOfContract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_LesseeOperatingLeasesTextBlock": {
"auth_ref": [
"r717"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for operating leases of lessee. Includes, but is not limited to, description of operating lease and maturity analysis of operating lease liability.",
"label": "Lessee, Operating Leases [Text Block]",
"terseLabel": "Leases"
}
}
},
"localname": "LesseeOperatingLeasesTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Leases"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_Liabilities": {
"auth_ref": [
"r68",
"r163",
"r245",
"r293",
"r612",
"r619",
"r620",
"r680"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 2.0,
"parentTag": "us-gaap_LiabilitiesAndStockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying amounts as of the balance sheet date of all liabilities that are recognized. Liabilities are probable future sacrifices of economic benefits arising from present obligations of an entity to transfer assets or provide services to other entities in the future.",
"label": "Liabilities",
"totalLabel": "Total liabilities"
}
}
},
"localname": "Liabilities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesAndStockholdersEquity": {
"auth_ref": [
"r50",
"r163",
"r293",
"r680",
"r764",
"r795"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liabilities and equity items, including the portion of equity attributable to noncontrolling interests, if any.",
"label": "Liabilities and Equity",
"totalLabel": "Total liabilities and equity"
}
}
},
"localname": "LiabilitiesAndStockholdersEquity",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesAndStockholdersEquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Liabilities and Equity [Abstract]",
"verboseLabel": "LIABILITIES AND EQUITY"
}
}
},
"localname": "LiabilitiesAndStockholdersEquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "stringItemType"
},
"us-gaap_LiabilitiesCurrent": {
"auth_ref": [
"r70",
"r163",
"r293",
"r612",
"r619",
"r620",
"r680"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 1.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Total obligations incurred as part of normal operations that are expected to be paid during the following twelve months or within one business cycle, if longer.",
"label": "Liabilities, Current",
"totalLabel": "Total current liabilities"
}
}
},
"localname": "LiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesCurrentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Liabilities, Current [Abstract]",
"verboseLabel": "Current liabilities:"
}
}
},
"localname": "LiabilitiesCurrentAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "stringItemType"
},
"us-gaap_LiabilitiesFairValueDisclosure": {
"auth_ref": [
"r663"
],
"calculation": {
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value of financial and nonfinancial obligations.",
"label": "Financial and Nonfinancial Liabilities, Fair Value Disclosure",
"totalLabel": "Total"
}
}
},
"localname": "LiabilitiesFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesFairValueDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Liabilities, Fair Value Disclosure [Abstract]",
"verboseLabel": "Liabilities:"
}
}
},
"localname": "LiabilitiesFairValueDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LiabilityForUncertainTaxPositionsCurrent": {
"auth_ref": [
"r69"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount recognized for uncertainty in income taxes classified as current.",
"label": "Liability for Uncertainty in Income Taxes, Current",
"verboseLabel": "Liabilities associated with uncertain tax positions"
}
}
},
"localname": "LiabilityForUncertainTaxPositionsCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LineOfCredit": {
"auth_ref": [
"r38",
"r761",
"r787"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The carrying value as of the balance sheet date of the current and noncurrent portions of long-term obligations drawn from a line of credit, which is a bank's commitment to make loans up to a specific amount. Examples of items that might be included in the application of this element may consist of letters of credit, standby letters of credit, and revolving credit arrangements, under which borrowings can be made up to a maximum amount as of any point in time conditional on satisfaction of specified terms before, as of and after the date of drawdowns on the line. Includes short-term obligations that would normally be classified as current liabilities but for which (a) postbalance sheet date issuance of a long term obligation to refinance the short term obligation on a long term basis, or (b) the enterprise has entered into a financing agreement that clearly permits the enterprise to refinance the short-term obligation on a long term basis and the following conditions are met (1) the agreement does not expire within 1 year and is not cancelable by the lender except for violation of an objectively determinable provision, (2) no violation exists at the BS date, and (3) the lender has entered into the financing agreement is expected to be financially capable of honoring the agreement.",
"label": "Long-term Line of Credit",
"terseLabel": "Outstanding borrowings"
}
}
},
"localname": "LineOfCredit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LineOfCreditFacilityMaximumBorrowingCapacity": {
"auth_ref": [
"r65"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Maximum borrowing capacity under the credit facility without consideration of any current restrictions on the amount that could be borrowed or the amounts currently outstanding under the facility.",
"label": "Line of Credit Facility, Maximum Borrowing Capacity",
"terseLabel": "Senior unsecured revolving credit facility maximum borrowing capacity"
}
}
},
"localname": "LineOfCreditFacilityMaximumBorrowingCapacity",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LineOfCreditMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A contractual arrangement with a lender under which borrowings can be made up to a specific amount at any point in time, and under which borrowings outstanding may be either short-term or long-term, depending upon the particulars.",
"label": "Line of Credit [Member]",
"terseLabel": "Line of Credit"
}
}
},
"localname": "LineOfCreditMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LongTermDebt": {
"auth_ref": [
"r38",
"r367",
"r761",
"r790"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 2.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after unamortized (discount) premium and debt issuance costs, of long-term debt. Includes, but not limited to, notes payable, bonds payable, debentures, mortgage loans and commercial paper. Excludes capital lease obligations.",
"label": "Long-term Debt",
"terseLabel": "Non-current portion of notes payable",
"verboseLabel": "Notes payable"
}
}
},
"localname": "LongTermDebt",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtCurrentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Long-term Debt, Current Maturities [Abstract]",
"terseLabel": "Current portion:"
}
}
},
"localname": "LongTermDebtCurrentAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalAfterYearFive": {
"auth_ref": [
"r172",
"r365"
],
"calculation": {
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails": {
"order": 6.0,
"parentTag": "us-gaap_LongTermNotesPayable",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing after fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, after Year Five",
"terseLabel": "2026 and thereafter"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalAfterYearFive",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths": {
"auth_ref": [
"r172",
"r365"
],
"calculation": {
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails": {
"order": 1.0,
"parentTag": "us-gaap_LongTermNotesPayable",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year One",
"terseLabel": "2021"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInYearFive": {
"auth_ref": [
"r172",
"r365"
],
"calculation": {
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails": {
"order": 5.0,
"parentTag": "us-gaap_LongTermNotesPayable",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year Five",
"terseLabel": "2025"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInYearFive",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInYearFour": {
"auth_ref": [
"r172",
"r365"
],
"calculation": {
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails": {
"order": 4.0,
"parentTag": "us-gaap_LongTermNotesPayable",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in fourth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year Four",
"terseLabel": "2024"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInYearFour",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInYearThree": {
"auth_ref": [
"r172",
"r365"
],
"calculation": {
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails": {
"order": 3.0,
"parentTag": "us-gaap_LongTermNotesPayable",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in third fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year Three",
"terseLabel": "2023"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInYearThree",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInYearTwo": {
"auth_ref": [
"r172",
"r365"
],
"calculation": {
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails": {
"order": 2.0,
"parentTag": "us-gaap_LongTermNotesPayable",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in second fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year Two",
"terseLabel": "2022"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInYearTwo",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtNoncurrentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Long-term Debt, Excluding Current Maturities [Abstract]",
"terseLabel": "Non-current portion:"
}
}
},
"localname": "LongTermDebtNoncurrentAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LongTermNotesPayable": {
"auth_ref": [
"r72"
],
"calculation": {
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of notes payable (with maturities initially due after one year or beyond the operating cycle if longer), excluding current portion.",
"label": "Notes Payable, Noncurrent",
"terseLabel": "Non-current notes payable",
"totalLabel": "Total"
}
}
},
"localname": "LongTermNotesPayable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails",
"http://www.biogenidec.com/role/IndebtednessTotalDebtMaturitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongtermDebtTypeAxis": {
"auth_ref": [
"r72"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of long-term debt.",
"label": "Long-term Debt, Type [Axis]",
"terseLabel": "Long-term Debt, Type [Axis]"
}
}
},
"localname": "LongtermDebtTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LongtermDebtTypeDomain": {
"auth_ref": [
"r72",
"r366"
],
"lang": {
"en-us": {
"role": {
"documentation": "Type of long-term debt arrangement, such as notes, line of credit, commercial paper, asset-based financing, project financing, letter of credit financing. These are debt arrangements that originally required repayment more than twelve months after issuance or greater than the normal operating cycle of the company, if longer.",
"label": "Long-term Debt, Type [Domain]",
"terseLabel": "Long-term Debt, Type [Domain]"
}
}
},
"localname": "LongtermDebtTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LossContingencyDamagesSoughtValue": {
"auth_ref": [
"r357",
"r359",
"r360"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The value (monetary amount) of the award the plaintiff seeks in the legal matter.",
"label": "Loss Contingency, Damages Sought, Value",
"terseLabel": "Claims asserted"
}
}
},
"localname": "LossContingencyDamagesSoughtValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LitigationNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LossContingencyInformationAboutLitigationMattersAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Loss Contingency, Information about Litigation Matters [Abstract]",
"terseLabel": "Loss Contingency, Information about Litigation Matters [Abstract]"
}
}
},
"localname": "LossContingencyInformationAboutLitigationMattersAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_LossOnContractTermination": {
"auth_ref": [
"r753"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The loss recognized on termination of a contract.",
"label": "Loss on Contract Termination",
"terseLabel": "Loss on contract termination"
}
}
},
"localname": "LossOnContractTermination",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_MachineryAndEquipmentGross": {
"auth_ref": [
"r27",
"r340"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 4.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated depreciation of tangible personal property used to produce goods and services, including, but is not limited to, tools, dies and molds, computer and office equipment.",
"label": "Machinery and Equipment, Gross",
"terseLabel": "Machinery and equipment"
}
}
},
"localname": "MachineryAndEquipmentGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_MachineryAndEquipmentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tangible personal property used to produce goods and services, including, but is not limited to, tools, dies and molds, computer and office equipment.",
"label": "Machinery and Equipment [Member]",
"terseLabel": "Machinery and Equipment"
}
}
},
"localname": "MachineryAndEquipmentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MeasurementInputDiscountRateMember": {
"auth_ref": [
"r666"
],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input using interest rate to determine present value of future cash flows.",
"label": "Measurement Input, Discount Rate [Member]",
"terseLabel": "Discount rate"
}
}
},
"localname": "MeasurementInputDiscountRateMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MeasurementInputTypeAxis": {
"auth_ref": [
"r666"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of measurement input used to determine value of asset and liability.",
"label": "Measurement Input Type [Axis]",
"terseLabel": "Measurement Input Type [Axis]"
}
}
},
"localname": "MeasurementInputTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_MeasurementInputTypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input used to determine value of asset and liability.",
"label": "Measurement Input Type [Domain]",
"terseLabel": "Measurement Input Type [Domain]"
}
}
},
"localname": "MeasurementInputTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofSignificantUnobservableInputsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MinorityInterest": {
"auth_ref": [
"r78",
"r163",
"r293",
"r680",
"r763",
"r794"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 2.0,
"parentTag": "us-gaap_StockholdersEquityIncludingPortionAttributableToNoncontrollingInterest",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Total of all stockholders' equity (deficit) items, net of receivables from officers, directors, owners, and affiliates of the entity which is directly or indirectly attributable to that ownership interest in subsidiary equity which is not attributable to the parent (that is, noncontrolling interest, previously referred to as minority interest).",
"label": "Stockholders' Equity Attributable to Noncontrolling Interest",
"verboseLabel": "Noncontrolling interests"
}
}
},
"localname": "MinorityInterest",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_MinorityInterestDecreaseFromDistributionsToNoncontrollingInterestHolders": {
"auth_ref": [
"r379"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Decrease in noncontrolling interest balance from payment of dividends or other distributions by the non-wholly owned subsidiary or partially owned entity, included in the consolidation of the parent entity, to the noncontrolling interest holders.",
"label": "Noncontrolling Interest, Decrease from Distributions to Noncontrolling Interest Holders",
"negatedTerseLabel": "Distribution to noncontrolling interests"
}
}
},
"localname": "MinorityInterestDecreaseFromDistributionsToNoncontrollingInterestHolders",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_MoneyMarketFundsMember": {
"auth_ref": [
"r428"
],
"lang": {
"en-us": {
"role": {
"documentation": "Fund that invests in short-term money-market instruments, for example, but not limited to, commercial paper, banker's acceptances, repurchase agreements, government securities, certificates of deposit, and other highly liquid securities.",
"label": "Money Market Funds [Member]",
"terseLabel": "Money market funds"
}
}
},
"localname": "MoneyMarketFundsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MovementInValuationAllowancesAndReservesRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "SEC Schedule, 12-09, Movement in Valuation Allowances and Reserves [Roll Forward]",
"terseLabel": "SEC Schedule, 12-09, Movement in Valuation Allowances and Reserves [Roll Forward]"
}
}
},
"localname": "MovementInValuationAllowancesAndReservesRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_NetCashProvidedByUsedInFinancingActivities": {
"auth_ref": [
"r146"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 1.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseExcludingExchangeRateEffect",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow (outflow) from financing activities, including discontinued operations. Financing activity cash flows include obtaining resources from owners and providing them with a return on, and a return of, their investment; borrowing money and repaying amounts borrowed, or settling the obligation; and obtaining and paying for other resources obtained from creditors on long-term credit.",
"label": "Net Cash Provided by (Used in) Financing Activities",
"totalLabel": "Net cash flows (used in) financing activities"
}
}
},
"localname": "NetCashProvidedByUsedInFinancingActivities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetCashProvidedByUsedInFinancingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Net Cash Provided by (Used in) Financing Activities [Abstract]",
"verboseLabel": "Cash flows from financing activities:"
}
}
},
"localname": "NetCashProvidedByUsedInFinancingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "stringItemType"
},
"us-gaap_NetCashProvidedByUsedInInvestingActivities": {
"auth_ref": [
"r146"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 2.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseExcludingExchangeRateEffect",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow (outflow) from investing activities, including discontinued operations. Investing activity cash flows include making and collecting loans and acquiring and disposing of debt or equity instruments and property, plant, and equipment and other productive assets.",
"label": "Net Cash Provided by (Used in) Investing Activities",
"totalLabel": "Net cash flows provided by (used in) investing activities"
}
}
},
"localname": "NetCashProvidedByUsedInInvestingActivities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetCashProvidedByUsedInInvestingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Net Cash Provided by (Used in) Investing Activities [Abstract]",
"verboseLabel": "Cash flows from investing activities:"
}
}
},
"localname": "NetCashProvidedByUsedInInvestingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "stringItemType"
},
"us-gaap_NetCashProvidedByUsedInOperatingActivities": {
"auth_ref": [
"r146",
"r149",
"r152"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 3.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseExcludingExchangeRateEffect",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow (outflow) from operating activities, including discontinued operations. Operating activity cash flows include transactions, adjustments, and changes in value not defined as investing or financing activities.",
"label": "Net Cash Provided by (Used in) Operating Activities",
"totalLabel": "Net cash flows provided by operating activities"
}
}
},
"localname": "NetCashProvidedByUsedInOperatingActivities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetCashProvidedByUsedInOperatingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Net Cash Provided by (Used in) Operating Activities [Abstract]",
"verboseLabel": "Cash flows from operating activities:"
}
}
},
"localname": "NetCashProvidedByUsedInOperatingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "stringItemType"
},
"us-gaap_NetIncomeLoss": {
"auth_ref": [
"r20",
"r101",
"r104",
"r112",
"r152",
"r163",
"r184",
"r186",
"r187",
"r188",
"r189",
"r192",
"r193",
"r203",
"r239",
"r243",
"r246",
"r249",
"r252",
"r293",
"r680",
"r771",
"r802"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": 2.0,
"parentTag": "us-gaap_ComprehensiveIncomeNetOfTax",
"weight": 1.0
},
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The portion of profit or loss for the period, net of income taxes, which is attributable to the parent.",
"label": "Net Income (Loss) Attributable to Parent",
"terseLabel": "Net income attributable to Biogen Inc.",
"totalLabel": "Net income attributable to Biogen Inc.",
"verboseLabel": "Total reclassifications, net of tax"
}
}
},
"localname": "NetIncomeLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetIncomeLossAttributableToNoncontrollingInterest": {
"auth_ref": [
"r101",
"r104",
"r192",
"r193",
"r615",
"r627"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 2.0,
"parentTag": "us-gaap_NetIncomeLoss",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of Net Income (Loss) attributable to noncontrolling interest.",
"label": "Net Income (Loss) Attributable to Noncontrolling Interest",
"terseLabel": "Net income (loss) attributable to noncontrolling interests, net of tax"
}
}
},
"localname": "NetIncomeLossAttributableToNoncontrollingInterest",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetInvestmentHedgingMember": {
"auth_ref": [
"r636"
],
"lang": {
"en-us": {
"role": {
"documentation": "Hedges of a net investment in a foreign operation.",
"label": "Net Investment Hedging [Member]",
"terseLabel": "Net investment hedge"
}
}
},
"localname": "NetInvestmentHedgingMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_NewAccountingPronouncementsPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy pertaining to new accounting pronouncements that may impact the entity's financial reporting. Includes, but is not limited to, quantification of the expected or actual impact.",
"label": "New Accounting Pronouncements, Policy [Policy Text Block]",
"terseLabel": "New Accounting Pronouncements"
}
}
},
"localname": "NewAccountingPronouncementsPolicyPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_NonUsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Countries excluding the United States of America (US).",
"label": "Non-US [Member]",
"terseLabel": "Rest of World"
}
}
},
"localname": "NonUsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_NoncontrollingInterestMember": {
"auth_ref": [
"r180",
"r181",
"r182",
"r379",
"r606"
],
"lang": {
"en-us": {
"role": {
"documentation": "This element represents that portion of equity (net assets) in a subsidiary not attributable, directly or indirectly, to the parent. A noncontrolling interest is sometimes called a minority interest.",
"label": "Noncontrolling Interest [Member]",
"verboseLabel": "Noncontrolling interests"
}
}
},
"localname": "NoncontrollingInterestMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "domainItemType"
},
"us-gaap_NoncurrentAssets": {
"auth_ref": [
"r256"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Long-lived assets other than financial instruments, long-term customer relationships of a financial institution, mortgage and other servicing rights, deferred policy acquisition costs, and deferred tax assets.",
"label": "Long-Lived Assets",
"terseLabel": "Long-lived assets related to operations in Denmark",
"verboseLabel": "Long-lived assets"
}
}
},
"localname": "NoncurrentAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails",
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NondesignatedMember": {
"auth_ref": [
"r637"
],
"lang": {
"en-us": {
"role": {
"documentation": "Derivative instrument not designated as hedging instrument under Generally Accepted Accounting Principles (GAAP).",
"label": "Not Designated as Hedging Instrument [Member]",
"terseLabel": "Not designated as hedging instrument"
}
}
},
"localname": "NondesignatedMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_NonoperatingIncomeExpense": {
"auth_ref": [
"r124"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 2.0,
"parentTag": "us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestments",
"weight": 1.0
},
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate amount of income or expense from ancillary business-related activities (that is to say, excluding major activities considered part of the normal operations of the business).",
"label": "Nonoperating Income (Expense)",
"terseLabel": "Other income (expense)",
"totalLabel": "Total other income (expense), net",
"verboseLabel": "Other income (expense), net"
}
}
},
"localname": "NonoperatingIncomeExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NotesPayableCurrent": {
"auth_ref": [
"r66"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying values as of the balance sheet date of the portions of long-term notes payable due within one year or the operating cycle if longer.",
"label": "Notes Payable, Current",
"terseLabel": "2.900% Senior Notes due September 15, 2020"
}
}
},
"localname": "NotesPayableCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NotesPayableFairValueDisclosure": {
"auth_ref": [
"r67"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of notes payable.",
"label": "Notes Payable, Fair Value Disclosure",
"verboseLabel": "Notes payable, fair value"
}
}
},
"localname": "NotesPayableFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NumberOfOperatingSegments": {
"auth_ref": [
"r229"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of operating segments. An operating segment is a component of an enterprise: (a) that engages in business activities from which it may earn revenues and incur expenses (including revenues and expenses relating to transactions with other components of the same enterprise), (b) whose operating results are regularly reviewed by the enterprise's chief operating decision maker to make decisions about resources to be allocated to the segment and assess its performance, and (c) for which discrete financial information is available. An operating segment may engage in business activities for which it has yet to earn revenues, for example, start-up operations may be operating segments before earning revenues.",
"label": "Number of Operating Segments",
"terseLabel": "Number of operating segments"
}
}
},
"localname": "NumberOfOperatingSegments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"us-gaap_NumberOfReportableSegments": {
"auth_ref": [
"r229"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of segments reported by the entity. A reportable segment is a component of an entity for which there is an accounting requirement to report separate financial information on that component in the entity's financial statements.",
"label": "Number of Reportable Segments",
"verboseLabel": "Number of reportable segment"
}
}
},
"localname": "NumberOfReportableSegments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"us-gaap_OperatingExpenseMember": {
"auth_ref": [
"r653"
],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing expenses associated with normal operations.",
"label": "Operating Expense [Member]",
"terseLabel": "Operating expenses"
}
}
},
"localname": "OperatingExpenseMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_OperatingExpenses": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Generally recurring costs associated with normal operations except for the portion of these expenses which can be clearly related to production and included in cost of sales or services. Includes selling, general and administrative expense.",
"label": "Operating Expenses",
"terseLabel": "Operating expenses"
}
}
},
"localname": "OperatingExpenses",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingIncomeLoss": {
"auth_ref": [
"r239",
"r243",
"r246",
"r249",
"r252"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 1.0,
"parentTag": "us-gaap_IncomeLossFromContinuingOperationsBeforeIncomeTaxesMinorityInterestAndIncomeLossFromEquityMethodInvestments",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The net result for the period of deducting operating expenses from operating revenues.",
"label": "Operating Income (Loss)",
"totalLabel": "Income from operations"
}
}
},
"localname": "OperatingIncomeLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseCost": {
"auth_ref": [
"r707",
"r716"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of single lease cost, calculated by allocation of remaining cost of lease over remaining lease term. Includes, but is not limited to, single lease cost, after impairment of right-of-use asset, calculated by amortization of remaining right-of-use asset and accretion of lease liability.",
"label": "Operating Lease, Cost",
"terseLabel": "Operating lease cost"
}
}
},
"localname": "OperatingLeaseCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseLiability": {
"auth_ref": [
"r701"
],
"calculation": {
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails_1": {
"order": 1.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Present value of lessee's discounted obligation for lease payments from operating lease.",
"label": "Operating Lease, Liability",
"terseLabel": "Present value of operating lease liabilities"
}
}
},
"localname": "OperatingLeaseLiability",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseLiabilityMaturityDetails",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseLiabilityCurrent": {
"auth_ref": [
"r701"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Present value of lessee's discounted obligation for lease payments from operating lease, classified as current.",
"label": "Operating Lease, Liability, Current",
"terseLabel": "Operating lease liability, current"
}
}
},
"localname": "OperatingLeaseLiabilityCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseLiabilityCurrentStatementOfFinancialPositionExtensibleList": {
"auth_ref": [
"r702"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicates line item in statement of financial position that includes current operating lease liability.",
"label": "Operating Lease, Liability, Current, Statement of Financial Position [Extensible List]",
"terseLabel": "Operating Lease, Liability, Current, Statement of Financial Position [Extensible List]"
}
}
},
"localname": "OperatingLeaseLiabilityCurrentStatementOfFinancialPositionExtensibleList",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "extensibleListItemType"
},
"us-gaap_OperatingLeaseLiabilityNoncurrent": {
"auth_ref": [
"r701"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 4.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Present value of lessee's discounted obligation for lease payments from operating lease, classified as noncurrent.",
"label": "Operating Lease, Liability, Noncurrent",
"terseLabel": "Long-term operating lease liabilities"
}
}
},
"localname": "OperatingLeaseLiabilityNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeasePayments": {
"auth_ref": [
"r703",
"r710"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow from operating lease, excluding payments to bring another asset to condition and location necessary for its intended use.",
"label": "Operating Lease, Payments",
"terseLabel": "Cash paid for amounts included in the measurement of lease liabilities"
}
}
},
"localname": "OperatingLeasePayments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseSupplementalCashFlowDisclosureDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseRightOfUseAsset": {
"auth_ref": [
"r700"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 4.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's right to use underlying asset under operating lease.",
"label": "Operating Lease, Right-of-Use Asset",
"terseLabel": "Operating lease assets"
}
}
},
"localname": "OperatingLeaseRightOfUseAsset",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseWeightedAverageDiscountRatePercent": {
"auth_ref": [
"r713",
"r716"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average discount rate for operating lease calculated at point in time.",
"label": "Operating Lease, Weighted Average Discount Rate, Percent",
"terseLabel": "Weighted average discount rate"
}
}
},
"localname": "OperatingLeaseWeightedAverageDiscountRatePercent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseWeightedAverageRemainingTermandDiscountRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_OperatingLeaseWeightedAverageRemainingLeaseTerm1": {
"auth_ref": [
"r712",
"r716"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average remaining lease term for operating lease, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Operating Lease, Weighted Average Remaining Lease Term",
"terseLabel": "Weighted average remaining lease term in years"
}
}
},
"localname": "OperatingLeaseWeightedAverageRemainingLeaseTerm1",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseWeightedAverageRemainingTermandDiscountRateDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_OperatingLeasedAssetsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Operating Leased Assets [Line Items]",
"terseLabel": "Operating Leased Assets [Line Items]"
}
}
},
"localname": "OperatingLeasedAssetsLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_OperatingLossCarryforwards": {
"auth_ref": [
"r558"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of operating loss carryforward, before tax effects, available to reduce future taxable income under enacted tax laws.",
"label": "Operating Loss Carryforwards",
"terseLabel": "Operating loss carryforwards"
}
}
},
"localname": "OperatingLossCarryforwards",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Organization, Consolidation and Presentation of Financial Statements [Abstract]",
"terseLabel": "Organization, Consolidation and Presentation of Financial Statements [Abstract]"
}
}
},
"localname": "OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_OtherAccruedLiabilitiesCurrent": {
"auth_ref": [
"r29",
"r30",
"r31",
"r69"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": 4.0,
"parentTag": "us-gaap_OtherLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expenses incurred but not yet paid classified as other, due within one year or the normal operating cycle, if longer.",
"label": "Other Accrued Liabilities, Current",
"terseLabel": "Other Accrued Liabilities, Current"
}
}
},
"localname": "OtherAccruedLiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherAssetsCurrent": {
"auth_ref": [
"r82"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 6.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of current assets classified as other.",
"label": "Other Assets, Current",
"verboseLabel": "Other current assets"
}
}
},
"localname": "OtherAssetsCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherAssetsNoncurrent": {
"auth_ref": [
"r63"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 8.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of noncurrent assets classified as other.",
"label": "Other Assets, Noncurrent",
"terseLabel": "Investments and other assets"
}
}
},
"localname": "OtherAssetsNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherCommitment": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Minimum amount of other commitment not otherwise specified in the taxonomy. Excludes commitments explicitly modeled in the taxonomy, including but not limited to, long-term and short-term purchase commitments, recorded and unrecorded purchase obligations, supply commitments, registration payment arrangements, leases, debt, product warranties, guarantees, environmental remediation obligations, and pensions.",
"label": "Other Commitment",
"verboseLabel": "Cancellable future commitments"
}
}
},
"localname": "OtherCommitment",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossAvailableForSaleSecuritiesAdjustmentNetOfTax": {
"auth_ref": [
"r90",
"r91",
"r96"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": 2.0,
"parentTag": "us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after tax and adjustment, of unrealized gain (loss) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale) and unrealized gain (loss) on investment in debt security measured at amortized cost (held-to-maturity) from transfer to available-for-sale.",
"label": "OCI, Debt Securities, Available-for-Sale, Gain (Loss), after Adjustment and Tax",
"terseLabel": "Unrealized gains (losses) on securities available for sale, net of tax"
}
}
},
"localname": "OtherComprehensiveIncomeLossAvailableForSaleSecuritiesAdjustmentNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossBeforeReclassificationsNetOfTax": {
"auth_ref": [
"r98",
"r108",
"r686",
"r688",
"r692"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax, before reclassification adjustments of other comprehensive income (loss).",
"label": "Other Comprehensive Income (Loss), before Reclassifications, Net of Tax",
"terseLabel": "Other comprehensive income (loss) before reclassifications"
}
}
},
"localname": "OtherComprehensiveIncomeLossBeforeReclassificationsNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossCashFlowHedgeGainLossAfterReclassificationAndTaxParent": {
"auth_ref": [
"r96"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": 4.0,
"parentTag": "us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after tax and reclassification, of gain (loss) from derivative instrument designated and qualifying as cash flow hedge included in assessment of hedge effectiveness, attributable to parent.",
"label": "Other Comprehensive Income (Loss), Cash Flow Hedge, Gain (Loss), after Reclassification and Tax, Parent",
"terseLabel": "Unrealized gains (losses) on cash flow hedges, net of tax"
}
}
},
"localname": "OtherComprehensiveIncomeLossCashFlowHedgeGainLossAfterReclassificationAndTaxParent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossCashFlowHedgeGainLossReclassificationBeforeTax": {
"auth_ref": [
"r96",
"r99",
"r646"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before tax, of reclassification of gain (loss) from accumulated other comprehensive income (AOCI) for derivative instrument designated and qualifying as cash flow hedge included in assessment of hedge effectiveness.",
"label": "Other Comprehensive Income (Loss), Cash Flow Hedge, Gain (Loss), Reclassification, before Tax",
"terseLabel": "Net Gains/(Losses) Reclassified from AOCI into Operating Income (in millions)"
}
}
},
"localname": "OtherComprehensiveIncomeLossCashFlowHedgeGainLossReclassificationBeforeTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossDerivativeExcludedComponentIncreaseDecreaseAfterAdjustmentsAndTax": {
"auth_ref": [
"r89",
"r645"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": 1.0,
"parentTag": "us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after tax and adjustments, of gain (loss) from increase (decrease) in value of excluded component of derivative designated and qualifying as hedge. Adjustments include, but are not limited to, reclassifications for sale and settlement, and amounts recognized under systematic and rational method.",
"label": "Other Comprehensive Income (Loss), Derivative, Excluded Component, Increase (Decrease), after Adjustments and Tax",
"terseLabel": "Gains (losses) on net investment hedges, net of tax"
}
}
},
"localname": "OtherComprehensiveIncomeLossDerivativeExcludedComponentIncreaseDecreaseAfterAdjustmentsAndTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossDerivativeExcludedComponentIncreaseDecreaseBeforeAdjustmentsAndTax": {
"auth_ref": [
"r89",
"r96",
"r645",
"r649",
"r658"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before tax and adjustments, of gain (loss) from increase (decrease) in value of excluded component of derivative designated and qualifying as hedge. Adjustments include, but are not limited to, reclassifications for sale and settlement, and amounts recognized under systematic and rational method.",
"label": "Other Comprehensive Income (Loss), Derivative, Excluded Component, Increase (Decrease), before Adjustments and Tax",
"verboseLabel": "Net Gains/(Losses) Recognized in Net Income (Amounts Excluded from Effectiveness Testing) (in millions)"
}
}
},
"localname": "OtherComprehensiveIncomeLossDerivativeExcludedComponentIncreaseDecreaseBeforeAdjustmentsAndTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossForeignCurrencyTransactionAndTranslationAdjustmentNetOfTax": {
"auth_ref": [
"r87"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": 5.0,
"parentTag": "us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax and reclassification adjustments of gain (loss) on foreign currency translation adjustments, foreign currency transactions designated and effective as economic hedges of a net investment in a foreign entity and intra-entity foreign currency transactions that are of a long-term-investment nature.",
"label": "Other Comprehensive Income (Loss), Foreign Currency Transaction and Translation Adjustment, Net of Tax",
"verboseLabel": "Currency translation adjustment"
}
}
},
"localname": "OtherComprehensiveIncomeLossForeignCurrencyTransactionAndTranslationAdjustmentNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossNetOfTax": {
"auth_ref": [
"r102",
"r105",
"r108",
"r113",
"r372",
"r686",
"r691",
"r692",
"r772",
"r803"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax and reclassification adjustments of other comprehensive income (loss).",
"label": "Other Comprehensive Income (Loss), Net of Tax",
"terseLabel": "Other comprehensive income (loss), net of tax"
}
}
},
"localname": "OtherComprehensiveIncomeLossNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent": {
"auth_ref": [
"r102",
"r105",
"r608",
"r609",
"r617"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": 1.0,
"parentTag": "us-gaap_ComprehensiveIncomeNetOfTax",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of other comprehensive income (loss) attributable to parent entity.",
"label": "Other Comprehensive Income (Loss), Net of Tax, Portion Attributable to Parent",
"totalLabel": "Total other comprehensive income (loss), net of tax",
"verboseLabel": "Net current period other comprehensive income (loss)"
}
}
},
"localname": "OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Other Comprehensive Income (Loss), Net of Tax, Portion Attributable to Parent [Abstract]",
"verboseLabel": "Other comprehensive income:"
}
}
},
"localname": "OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParentAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "stringItemType"
},
"us-gaap_OtherComprehensiveIncomeLossPensionAndOtherPostretirementBenefitPlansAdjustmentNetOfTax": {
"auth_ref": [
"r94",
"r96"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome": {
"order": 3.0,
"parentTag": "us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after tax and reclassification adjustment, of (increase) decrease in accumulated other comprehensive income for defined benefit plan.",
"label": "Other Comprehensive (Income) Loss, Defined Benefit Plan, after Reclassification Adjustment, after Tax",
"negatedLabel": "Unrealized gains (losses) on pension benefit obligation, net of tax"
}
}
},
"localname": "OtherComprehensiveIncomeLossPensionAndOtherPostretirementBenefitPlansAdjustmentNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofComprehensiveIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherCurrentAssetsMember": {
"auth_ref": [
"r639",
"r657"
],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing other current assets.",
"label": "Other Current Assets [Member]",
"terseLabel": "Other current assets"
}
}
},
"localname": "OtherCurrentAssetsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_OtherCurrentLiabilitiesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing other current liabilities.",
"label": "Other Current Liabilities [Member]",
"terseLabel": "Component of accrued expenses and other"
}
}
},
"localname": "OtherCurrentLiabilitiesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails",
"http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_OtherInventoryCapitalizedCosts": {
"auth_ref": [
"r79"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before valuation and last-in first out (LIFO) reserves, of costs capitalized in inventory classified as other, expected to be sold, or consumed within one year or operating cycle, if longer.",
"label": "Other Inventory, Capitalized Costs, Gross",
"terseLabel": "Capitalized inventory"
}
}
},
"localname": "OtherInventoryCapitalizedCosts",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherLiabilitiesCurrent": {
"auth_ref": [
"r28",
"r29",
"r69"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 3.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
},
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liabilities classified as other, due within one year or the normal operating cycle, if longer.",
"label": "Other Liabilities, Current",
"totalLabel": "Total accrued expenses and other",
"verboseLabel": "Accrued expenses and other"
}
}
},
"localname": "OtherLiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherLiabilitiesNoncurrent": {
"auth_ref": [
"r73"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 5.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liabilities classified as other, due after one year or the normal operating cycle, if longer.",
"label": "Other Liabilities, Noncurrent",
"verboseLabel": "Other long-term liabilities"
}
}
},
"localname": "OtherLiabilitiesNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherNoncashIncomeExpense": {
"auth_ref": [
"r152"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 12.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of income (expense) included in net income that results in no cash inflow (outflow), classified as other.",
"label": "Other Noncash Income (Expense)",
"negatedTerseLabel": "Other"
}
}
},
"localname": "OtherNoncashIncomeExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherNoncurrentLiabilitiesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing other noncurrent liabilities.",
"label": "Other Noncurrent Liabilities [Member]",
"terseLabel": "Other long-term liabilities"
}
}
},
"localname": "OtherNoncurrentLiabilitiesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheFairValueforourOutstandingDerivativesDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_OtherNonoperatingIncomeExpense": {
"auth_ref": [
"r125"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails": {
"order": 2.0,
"parentTag": "us-gaap_NonoperatingIncomeExpense",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of income (expense) related to nonoperating activities, classified as other.",
"label": "Other Nonoperating Income (Expense)",
"terseLabel": "Other, net"
}
}
},
"localname": "OtherNonoperatingIncomeExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailOtherIncomeExpenseNetDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherNonoperatingIncomeExpenseMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing other nonoperating income (expense).",
"label": "Other Nonoperating Income (Expense) [Member]",
"terseLabel": "Other (income) expense, net"
}
}
},
"localname": "OtherNonoperatingIncomeExpenseMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails",
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails",
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_OtherPaymentsToAcquireBusinesses": {
"auth_ref": [
"r132"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow associated with other payments to acquire businesses including deposit on pending acquisitions and preacquisition costs.",
"label": "Other Payments to Acquire Businesses",
"terseLabel": "Payments for pre-combination equity compensation"
}
}
},
"localname": "OtherPaymentsToAcquireBusinesses",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherPostretirementBenefitPlansDefinedBenefitMember": {
"auth_ref": [
"r404",
"r405",
"r406",
"r407",
"r408",
"r409",
"r410",
"r411",
"r412",
"r413",
"r414",
"r415",
"r416",
"r417",
"r418",
"r419",
"r420",
"r421",
"r422",
"r423",
"r424",
"r425",
"r426",
"r427",
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r451",
"r452",
"r454",
"r457",
"r460",
"r464",
"r465",
"r466",
"r467",
"r468",
"r469",
"r470",
"r471",
"r472",
"r473",
"r474",
"r475",
"r478",
"r479",
"r480",
"r481",
"r482",
"r483"
],
"lang": {
"en-us": {
"role": {
"documentation": "Plan designed to provide other postretirement benefits. Includes, but is not limited to, defined benefit and defined contribution plans. Excludes pension benefits.",
"label": "Other Postretirement Benefits Plan [Member]",
"terseLabel": "Deferred Compensation Plan"
}
}
},
"localname": "OtherPostretirementBenefitPlansDefinedBenefitMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_OtherTaxExpenseBenefit": {
"auth_ref": [
"r164",
"r539",
"r565"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of other income tax expense (benefit).",
"label": "Other Tax Expense (Benefit)",
"terseLabel": "Other tax expense (benefit)"
}
}
},
"localname": "OtherTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ParentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Portion of equity, or net assets, in the consolidated entity attributable, directly or indirectly, to the parent. Excludes noncontrolling interests.",
"label": "Parent [Member]",
"terseLabel": "Total Biogen Inc. shareholders\u2019 equity",
"verboseLabel": "Share-based compensation expense included in net income attributable to Biogen Inc."
}
}
},
"localname": "ParentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_PaymentForContingentConsiderationLiabilityFinancingActivities": {
"auth_ref": [
"r140"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 5.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow, not made soon after acquisition date of business combination, to settle contingent consideration liability up to amount recognized at acquisition date, including, but not limited to, measurement period adjustment and less amount paid soon after acquisition date.",
"label": "Payment for Contingent Consideration Liability, Financing Activities",
"negatedLabel": "Contingent consideration payments",
"terseLabel": "Contingent payment"
}
}
},
"localname": "PaymentForContingentConsiderationLiabilityFinancingActivities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsForProceedsFromOtherInvestingActivities": {
"auth_ref": [
"r130",
"r134",
"r171"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 4.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash (inflow) outflow from investing activities classified as other.",
"label": "Payments for (Proceeds from) Other Investing Activities",
"negatedTerseLabel": "Other"
}
}
},
"localname": "PaymentsForProceedsFromOtherInvestingActivities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsForRepurchaseOfCommonStock": {
"auth_ref": [
"r138"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 6.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow to reacquire common stock during the period.",
"label": "Payments for Repurchase of Common Stock",
"negatedLabel": "Purchase of treasury stock",
"terseLabel": "Payments for repurchase of common stock"
}
}
},
"localname": "PaymentsForRepurchaseOfCommonStock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsOfCapitalDistribution": {
"auth_ref": [
"r138"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 3.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Cash outflow to owners or shareholders, excluding ordinary dividends. Includes special dividends.",
"label": "Payments of Capital Distribution",
"negatedTerseLabel": "Net contribution (distribution) to noncontrolling interest"
}
}
},
"localname": "PaymentsOfCapitalDistribution",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsOfFinancingCosts": {
"auth_ref": [
"r141"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow for loan and debt issuance costs.",
"label": "Payments of Financing Costs",
"terseLabel": "Costs associated with the senior Nnotes offerings"
}
}
},
"localname": "PaymentsOfFinancingCosts",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireAvailableForSaleSecuritiesDebt": {
"auth_ref": [
"r126",
"r131",
"r269"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 8.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow to acquire investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Payments to Acquire Debt Securities, Available-for-sale",
"negatedLabel": "Purchases of marketable securities"
}
}
},
"localname": "PaymentsToAcquireAvailableForSaleSecuritiesDebt",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireBusinessTwoNetOfCashAcquired": {
"auth_ref": [
"r134"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 9.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow associated with a second acquisition of a business, net of the cash acquired from the purchase.",
"label": "Payments to Acquire Business Two, Net of Cash Acquired",
"negatedTerseLabel": "Acquisition of Nightstar Therapeutics plc, net of cash acquired"
}
}
},
"localname": "PaymentsToAcquireBusinessTwoNetOfCashAcquired",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireBusinessesGross": {
"auth_ref": [
"r132",
"r594"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow associated with the acquisition of business during the period. The cash portion only of the acquisition price.",
"label": "Payments to Acquire Businesses, Gross",
"verboseLabel": "Total transaction value"
}
}
},
"localname": "PaymentsToAcquireBusinessesGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireEquityMethodInvestments": {
"auth_ref": [
"r132"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 10.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow associated with the purchase of or advances to an equity method investments, which are investments in joint ventures and entities in which the entity has an equity ownership interest normally of 20 to 50 percent and exercises significant influence.",
"label": "Payments to Acquire Equity Method Investments",
"negatedTerseLabel": "Investment in Samsung Bioepis",
"terseLabel": "Payments to increase investment in Samsung Bioepis"
}
}
},
"localname": "PaymentsToAcquireEquityMethodInvestments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireInProcessResearchAndDevelopment": {
"auth_ref": [
"r132"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 7.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflows from the purchase of net carrying value allocated to in-process research and development costs and materials acquired in a business combination.",
"label": "Payments to Acquire in Process Research and Development",
"negatedTerseLabel": "Acquired in-process research and development"
}
}
},
"localname": "PaymentsToAcquireInProcessResearchAndDevelopment",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireIntangibleAssets": {
"auth_ref": [
"r133"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 5.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow to acquire asset without physical form usually arising from contractual or other legal rights, excluding goodwill.",
"label": "Payments to Acquire Intangible Assets",
"negatedTerseLabel": "Acquisitions of intangible assets"
}
}
},
"localname": "PaymentsToAcquireIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireInterestInSubsidiariesAndAffiliates": {
"auth_ref": [
"r132"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 3.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow associated with the acquisition of or advances to an entity that is related to it but not strictly controlled (for example, an unconsolidated subsidiary, affiliate, and joint venture or equity method investment) or the acquisition of an additional interest in a subsidiary (controlled entity).",
"label": "Payments to Acquire Interest in Subsidiaries and Affiliates",
"negatedTerseLabel": "Purchase of subsidiaries and affiliates"
}
}
},
"localname": "PaymentsToAcquireInterestInSubsidiariesAndAffiliates",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireOtherProductiveAssets": {
"auth_ref": [
"r133"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 6.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow for the purchase of or improvements to tangible or intangible assets, used to produce goods or deliver services, classified as other.",
"label": "Payments to Acquire Other Productive Assets",
"negatedTerseLabel": "Contingent consideration paid related to Fumapharm AG acquisition"
}
}
},
"localname": "PaymentsToAcquireOtherProductiveAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquirePropertyPlantAndEquipment": {
"auth_ref": [
"r133"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 1.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow associated with the acquisition of long-lived, physical assets that are used in the normal conduct of business to produce goods and services and not intended for resale; includes cash outflows to pay for construction of self-constructed assets.",
"label": "Payments to Acquire Property, Plant, and Equipment",
"negatedLabel": "Purchases of property, plant and equipment"
}
}
},
"localname": "PaymentsToAcquirePropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToMinorityShareholders": {
"auth_ref": [
"r141"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 7.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow to a noncontrolling interest. Includes, but not limited to, reduction of noncontrolling interest ownership. Excludes dividends paid to the noncontrolling interest.",
"label": "Payments to Noncontrolling Interests",
"negatedTerseLabel": "Payments related to issuance of stock for share-based compensation arrangements, net",
"verboseLabel": "Payment to Neurimmune"
}
}
},
"localname": "PaymentsToMinorityShareholders",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PensionAndOtherPostretirementBenefitsDisclosureTextBlock": {
"auth_ref": [
"r427",
"r429",
"r435",
"r453",
"r455",
"r456",
"r457",
"r458",
"r459",
"r474",
"r476",
"r477",
"r478",
"r491"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for retirement benefits.",
"label": "Retirement Benefits [Text Block]",
"verboseLabel": "Employee Benefit Plans"
}
}
},
"localname": "PensionAndOtherPostretirementBenefitsDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlans"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_PensionExpense": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cost (reversal of cost) for pension benefits. Excludes other postretirement benefits.",
"label": "Pension Cost (Reversal of Cost)",
"terseLabel": "Pension cost (reversal)"
}
}
},
"localname": "PensionExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PerformanceSharesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share-based payment arrangement awarded for meeting performance target.",
"label": "Performance Shares [Member]",
"terseLabel": "Performance units"
}
}
},
"localname": "PerformanceSharesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_PlanNameAxis": {
"auth_ref": [
"r494",
"r521"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by plan name for share-based payment arrangement.",
"label": "Plan Name [Axis]",
"terseLabel": "Plan Name [Axis]"
}
}
},
"localname": "PlanNameAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PlanNameDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Plan name for share-based payment arrangement.",
"label": "Plan Name [Domain]",
"terseLabel": "Plan Name [Domain]"
}
}
},
"localname": "PlanNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_PreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Preferred shares may provide a preferential dividend to the dividend on common stock and may take precedence over common stock in the event of a liquidation. Preferred shares typically represent an ownership interest in the company.",
"label": "Preferred Stock [Member]",
"verboseLabel": "Preferred stock"
}
}
},
"localname": "PreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "domainItemType"
},
"us-gaap_PreferredStockParOrStatedValuePerShare": {
"auth_ref": [
"r42"
],
"lang": {
"en-us": {
"role": {
"documentation": "Face amount or stated value per share of preferred stock nonredeemable or redeemable solely at the option of the issuer.",
"label": "Preferred Stock, Par or Stated Value Per Share",
"verboseLabel": "Preferred stock, par value (in dollars per share)"
}
}
},
"localname": "PreferredStockParOrStatedValuePerShare",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheetsParenthetical"
],
"xbrltype": "perShareItemType"
},
"us-gaap_PreferredStockSharesAuthorized": {
"auth_ref": [
"r42"
],
"lang": {
"en-us": {
"role": {
"documentation": "The maximum number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) permitted to be issued by an entity's charter and bylaws.",
"label": "Preferred Stock, Shares Authorized",
"terseLabel": "Preferred stock, shares authorized (in shares)"
}
}
},
"localname": "PreferredStockSharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_PreferredStockSharesIssued": {
"auth_ref": [
"r42"
],
"lang": {
"en-us": {
"role": {
"documentation": "Total number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) issued to shareholders (includes related preferred shares that were issued, repurchased, and remain in the treasury). May be all or portion of the number of preferred shares authorized. Excludes preferred shares that are classified as debt.",
"label": "Preferred Stock, Shares Issued",
"terseLabel": "Preferred stock, shares issued (in shares)"
}
}
},
"localname": "PreferredStockSharesIssued",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_PreferredStockSharesOutstanding": {
"auth_ref": [
"r42"
],
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate share number for all nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer) held by stockholders. Does not include preferred shares that have been repurchased.",
"label": "Preferred Stock, Shares Outstanding",
"terseLabel": "Preferred stock, shares outstanding (in shares)"
}
}
},
"localname": "PreferredStockSharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_PreferredStockValue": {
"auth_ref": [
"r42"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 1.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate par or stated value of issued nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable preferred shares, par value and other disclosure concepts are in another section within stockholders' equity.",
"label": "Preferred Stock, Value, Issued",
"verboseLabel": "Preferred stock, par value $0.001 per share"
}
}
},
"localname": "PreferredStockValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PrepaidExpenseAndOtherAssets": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of asset related to consideration paid in advance for costs that provide economic benefits in future periods, and amount of other assets.",
"label": "Prepaid Expense and Other Assets",
"terseLabel": "Prepaid research and discovery services"
}
}
},
"localname": "PrepaidExpenseAndOtherAssets",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromIssuanceOfLongTermDebt": {
"auth_ref": [
"r136"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 4.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow from a debt initially having maturity due after one year or beyond the operating cycle, if longer.",
"label": "Proceeds from Issuance of Long-term Debt",
"terseLabel": "Proceeds from borrowings"
}
}
},
"localname": "ProceedsFromIssuanceOfLongTermDebt",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromMinorityShareholders": {
"auth_ref": [
"r137"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 8.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow from a noncontrolling interest. Includes, but is not limited to, purchase of additional shares or other increase in noncontrolling interest ownership.",
"label": "Proceeds from Noncontrolling Interests",
"terseLabel": "Net contribution (distribution) to noncontrolling interest"
}
}
},
"localname": "ProceedsFromMinorityShareholders",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromPaymentsForOtherFinancingActivities": {
"auth_ref": [
"r137",
"r141",
"r171"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 2.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow (outflow) from financing activities classified as other.",
"label": "Proceeds from (Payments for) Other Financing Activities",
"terseLabel": "Other"
}
}
},
"localname": "ProceedsFromPaymentsForOtherFinancingActivities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromSaleAndMaturityOfAvailableForSaleSecurities": {
"auth_ref": [
"r126",
"r127",
"r269"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 11.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow from sale, maturity, prepayment and call of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Proceeds from Sale and Maturity of Debt Securities, Available-for-sale",
"terseLabel": "Proceeds from maturities and sales",
"verboseLabel": "Proceeds from sales and maturities of marketable securities"
}
}
},
"localname": "ProceedsFromSaleAndMaturityOfAvailableForSaleSecurities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/FinancialInstrumentsProceedsfromMarketableDebtSecuritiesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromSaleOfEquityMethodInvestments": {
"auth_ref": [
"r128"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 2.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow associated with the sale of equity method investments, which are investments in joint ventures and entities in which the entity has an equity ownership interest normally of 20 to 50 percent and exercises significant influence.",
"label": "Proceeds from Sale of Equity Method Investments",
"terseLabel": "Proceeds from sales of strategic investments"
}
}
},
"localname": "ProceedsFromSaleOfEquityMethodInvestments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromSaleOfPropertyPlantAndEquipment": {
"auth_ref": [
"r129"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 12.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow from the sale of long-lived, physical assets that are used in the normal conduct of business to produce goods and services and not intended for resale.",
"label": "Proceeds from Sale of Property, Plant, and Equipment",
"terseLabel": "Proceeds from divesiture of Hillerod, Denmark manufacturing operations"
}
}
},
"localname": "ProceedsFromSaleOfPropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromStockOptionsExercised": {
"auth_ref": [
"r135",
"r522"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow from exercise of option under share-based payment arrangement.",
"label": "Proceeds from Stock Options Exercised",
"verboseLabel": "Cash received from the exercise of stock options"
}
}
},
"localname": "ProceedsFromStockOptionsExercised",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTaxBenefitandCashReceivedfromStockOptionExercisesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProductAndServiceOtherMember": {
"auth_ref": [
"r395"
],
"lang": {
"en-us": {
"role": {
"documentation": "Article or substance produced by nature, labor or machinery and act of providing assistance, classified as other.",
"label": "Product and Service, Other [Member]",
"terseLabel": "Other",
"verboseLabel": "Total other revenues"
}
}
},
"localname": "ProductAndServiceOtherMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ProductMember": {
"auth_ref": [
"r395"
],
"lang": {
"en-us": {
"role": {
"documentation": "Article or substance produced by nature, labor or machinery.",
"label": "Product [Member]",
"terseLabel": "Product, net",
"verboseLabel": "Total product revenues"
}
}
},
"localname": "ProductMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ProfitLoss": {
"auth_ref": [
"r20",
"r101",
"r104",
"r144",
"r163",
"r184",
"r192",
"r193",
"r239",
"r243",
"r246",
"r249",
"r252",
"r293",
"r608",
"r614",
"r616",
"r627",
"r628",
"r680",
"r778"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 5.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
},
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 1.0,
"parentTag": "us-gaap_NetIncomeLoss",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The consolidated profit or loss for the period, net of income taxes, including the portion attributable to the noncontrolling interest.",
"label": "Net Income (Loss), Including Portion Attributable to Noncontrolling Interest",
"totalLabel": "Net income",
"verboseLabel": "Net income"
}
}
},
"localname": "ProfitLoss",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PropertyPlantAndEquipmentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Property, Plant and Equipment [Abstract]",
"terseLabel": "Property, Plant and Equipment [Abstract]"
}
}
},
"localname": "PropertyPlantAndEquipmentAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_PropertyPlantAndEquipmentByTypeAxis": {
"auth_ref": [
"r61",
"r342"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of long-lived, physical assets used to produce goods and services and not intended for resale.",
"label": "Long-Lived Tangible Asset [Axis]",
"terseLabel": "Property, Plant and Equipment, Type [Axis]"
}
}
},
"localname": "PropertyPlantAndEquipmentByTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PropertyPlantAndEquipmentDisclosureTextBlock": {
"auth_ref": [
"r350",
"r844",
"r845",
"r846"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for long-lived, physical asset used in normal conduct of business and not intended for resale. Includes, but is not limited to, work of art, historical treasure, and similar asset classified as collections.",
"label": "Property, Plant and Equipment Disclosure [Text Block]",
"verboseLabel": "Property, Plant and Equipment"
}
}
},
"localname": "PropertyPlantAndEquipmentDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipment"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_PropertyPlantAndEquipmentGross": {
"auth_ref": [
"r60",
"r340"
],
"calculation": {
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": 1.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures.",
"label": "Property, Plant and Equipment, Gross",
"terseLabel": "Property, plant and equipment, gross",
"totalLabel": "Total cost"
}
}
},
"localname": "PropertyPlantAndEquipmentGross",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PropertyPlantAndEquipmentLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Property, Plant and Equipment [Line Items]",
"terseLabel": "Property, Plant and Equipment [Line Items]"
}
}
},
"localname": "PropertyPlantAndEquipmentLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PropertyPlantAndEquipmentNet": {
"auth_ref": [
"r34",
"r35",
"r342",
"r796"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 3.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
},
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business to produce goods and services and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures.",
"label": "Property, Plant and Equipment, Net",
"totalLabel": "Total property, plant and equipment, net",
"verboseLabel": "Property, plant and equipment, net"
}
}
},
"localname": "PropertyPlantAndEquipmentNet",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/PropertyPlantandEquipmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PropertyPlantAndEquipmentPolicyTextBlock": {
"auth_ref": [
"r59",
"r158",
"r342",
"r844",
"r845"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for long-lived, physical asset used in normal conduct of business and not intended for resale. Includes, but is not limited to, work of art, historical treasure, and similar asset classified as collections.",
"label": "Property, Plant and Equipment, Policy [Policy Text Block]",
"terseLabel": "Property, Plant and Equipment"
}
}
},
"localname": "PropertyPlantAndEquipmentPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_PropertyPlantAndEquipmentTextBlock": {
"auth_ref": [
"r34",
"r342"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of physical assets used in the normal conduct of business and not intended for resale. Includes, but is not limited to, balances by class of assets, depreciation and depletion expense and method used, including composite depreciation, and accumulated deprecation.",
"label": "Property, Plant and Equipment [Table Text Block]",
"terseLabel": "Components of property, plant and equipment, net"
}
}
},
"localname": "PropertyPlantAndEquipmentTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_PropertyPlantAndEquipmentTypeDomain": {
"auth_ref": [
"r34",
"r340"
],
"lang": {
"en-us": {
"role": {
"documentation": "Listing of long-lived, physical assets that are used in the normal conduct of business to produce goods and services and not intended for resale. Examples include land, buildings, machinery and equipment, and other types of furniture and equipment including, but not limited to, office equipment, furniture and fixtures, and computer equipment and software.",
"label": "Long-Lived Tangible Asset [Domain]",
"terseLabel": "Property, Plant and Equipment, Type [Domain]"
}
}
},
"localname": "PropertyPlantAndEquipmentTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_PropertyPlantAndEquipmentUsefulLife": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Useful life of long lived, physical assets used in the normal conduct of business and not intended for resale, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days. Examples include, but not limited to, land, buildings, machinery and equipment, office equipment, furniture and fixtures, and computer equipment.",
"label": "Property, Plant and Equipment, Useful Life",
"verboseLabel": "Property, plant and equipment, useful life"
}
}
},
"localname": "PropertyPlantAndEquipmentUsefulLife",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_PublicUtilitiesInventoryAxis": {
"auth_ref": [
"r54"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of inventory held.",
"label": "Inventory [Axis]",
"terseLabel": "Inventory [Axis]"
}
}
},
"localname": "PublicUtilitiesInventoryAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PublicUtilitiesInventoryTypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tangible personal property that is held for sale in the ordinary course of business, in process of production for such sale or is to be currently consumed in the production of goods or services to be available for sale.",
"label": "Inventory [Domain]",
"terseLabel": "Inventory [Domain]"
}
}
},
"localname": "PublicUtilitiesInventoryTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_PurchaseCommitmentRemainingMinimumAmountCommitted": {
"auth_ref": [
"r40",
"r762",
"r791"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Minimum amount to be expended to satisfy the terms of arrangements in which the entity has agreed to expend funds to procure goods or services, excluding long-term purchase commitments or unconditional purchase obligations.",
"label": "Purchase Commitment, Remaining Minimum Amount Committed",
"terseLabel": "Future minimum batch production for Denmark manufacturing operations"
}
}
},
"localname": "PurchaseCommitmentRemainingMinimumAmountCommitted",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/DivestituresDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_QuarterlyFinancialInformationDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Quarterly Financial Information Disclosure [Abstract]",
"terseLabel": "Quarterly Financial Information Disclosure [Abstract]"
}
}
},
"localname": "QuarterlyFinancialInformationDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_QuarterlyFinancialInformationTextBlock": {
"auth_ref": [
"r215"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for quarterly financial data. Includes, but is not limited to, tabular presentation of financial information for fiscal quarters, effect of year-end adjustments, and an explanation of matters or transactions that affect comparability of the information.",
"label": "Quarterly Financial Information [Text Block]",
"verboseLabel": "Quarterly Financial Data (Unaudited)"
}
}
},
"localname": "QuarterlyFinancialInformationTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnaudited"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RealizedGainLossOnInvestmentsTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of realized gains and losses on investments reported in the statement of income.",
"label": "Realized Gain (Loss) on Investments [Table Text Block]",
"verboseLabel": "Proceeds from marketable securities, excluding strategic investments"
}
}
},
"localname": "RealizedGainLossOnInvestmentsTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RealizedInvestmentGainsLosses": {
"auth_ref": [
"r811"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of realized gain (loss) on investment.",
"label": "Realized Investment Gains (Losses)",
"terseLabel": "Realized investment gains (losses)"
}
}
},
"localname": "RealizedInvestmentGainsLosses",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ReceivablesPolicyTextBlock": {
"auth_ref": [
"r158",
"r263",
"r265",
"r266",
"r267"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for receivable. Includes, but is not limited to, accounts receivable and financing receivable.",
"label": "Receivable [Policy Text Block]",
"terseLabel": "Accounts Receivable"
}
}
},
"localname": "ReceivablesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ReclassificationAdjustmentOutOfAccumulatedOtherComprehensiveIncomeOnDerivativesLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Reclassification Adjustment out of Accumulated Other Comprehensive Income on Derivatives [Line Items]",
"terseLabel": "Reclassification Adjustment out of Accumulated Other Comprehensive Income on Derivatives [Line Items]"
}
}
},
"localname": "ReclassificationAdjustmentOutOfAccumulatedOtherComprehensiveIncomeOnDerivativesLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ReclassificationFromAccumulatedOtherComprehensiveIncomeCurrentPeriodNetOfTax": {
"auth_ref": [
"r98",
"r100",
"r108",
"r686",
"r690",
"r692"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of reclassification adjustments of other comprehensive income (loss).",
"label": "Reclassification from Accumulated Other Comprehensive Income, Current Period, Net of Tax",
"terseLabel": "Amounts reclassified from accumulated other comprehensive income (loss)"
}
}
},
"localname": "ReclassificationFromAccumulatedOtherComprehensiveIncomeCurrentPeriodNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ReclassificationOutOfAccumulatedOtherComprehensiveIncomeAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by item reclassified out of accumulated other comprehensive income (loss).",
"label": "Reclassification out of Accumulated Other Comprehensive Income [Axis]",
"terseLabel": "Reclassification out of Accumulated Other Comprehensive Income [Axis]"
}
}
},
"localname": "ReclassificationOutOfAccumulatedOtherComprehensiveIncomeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ReclassificationOutOfAccumulatedOtherComprehensiveIncomeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Item reclassified out of accumulated other comprehensive income (loss).",
"label": "Reclassification out of Accumulated Other Comprehensive Income [Domain]",
"terseLabel": "Reclassification out of Accumulated Other Comprehensive Income [Domain]"
}
}
},
"localname": "ReclassificationOutOfAccumulatedOtherComprehensiveIncomeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Identifies item reclassified out of accumulated other comprehensive income (loss).",
"label": "Reclassification out of Accumulated Other Comprehensive Income [Member]",
"terseLabel": "Reclassification out of Accumulated Other Comprehensive Income"
}
}
},
"localname": "ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ReclassificationOutOfAccumulatedOtherComprehensiveIncomeTable": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about items reclassified out of accumulated other comprehensive income (loss).",
"label": "Reclassification out of Accumulated Other Comprehensive Income [Table]",
"terseLabel": "Reclassification out of Accumulated Other Comprehensive Income [Table]"
}
}
},
"localname": "ReclassificationOutOfAccumulatedOtherComprehensiveIncomeTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ReclassificationOutOfAccumulatedOtherComprehensiveIncomeTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of information about items reclassified out of accumulated other comprehensive income (loss).",
"label": "Reclassification out of Accumulated Other Comprehensive Income [Table Text Block]",
"terseLabel": "Reclassification out of Accumulated Other Comprehensive Income"
}
}
},
"localname": "ReclassificationOutOfAccumulatedOtherComprehensiveIncomeTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ReconciliationOfUnrecognizedTaxBenefitsExcludingAmountsPertainingToExaminedTaxReturnsRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Reconciliation of Unrecognized Tax Benefits, Excluding Amounts Pertaining to Examined Tax Returns [Roll Forward]",
"terseLabel": "Reconciliation of Unrecognized Tax Benefits, Excluding Amounts Pertaining to Examined Tax Returns [Roll Forward]"
}
}
},
"localname": "ReconciliationOfUnrecognizedTaxBenefitsExcludingAmountsPertainingToExaminedTaxReturnsRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RepaymentsOfDebt": {
"auth_ref": [
"r139"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 1.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow during the period from the repayment of aggregate short-term and long-term debt. Excludes payment of capital lease obligations.",
"label": "Repayments of Debt",
"negatedTerseLabel": "Repayments of borrowings"
}
}
},
"localname": "RepaymentsOfDebt",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RepurchaseAgreementsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Investments represented by an agreement between the entity and another party for the sale and repurchase of identical or substantially the same securities at a date certain for a specified price. Such agreements are generally short-term in nature.",
"label": "Repurchase Agreements [Member]",
"verboseLabel": "Overnight reverse repurchase agreements"
}
}
},
"localname": "RepurchaseAgreementsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ResearchAndDevelopmentExpense": {
"auth_ref": [
"r532",
"r861"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate costs incurred (1) in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or (2) to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process whether intended for sale or the entity's use, during the reporting period charged to research and development projects, including the costs of developing computer software up to the point in time of achieving technological feasibility, and costs allocated in accounting for a business combination to in-process projects deemed to have no alternative future use.",
"label": "Research and Development Expense",
"terseLabel": "Research and development expense asset acquired"
}
}
},
"localname": "ResearchAndDevelopmentExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost": {
"auth_ref": [
"r532"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 2.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The costs incurred in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process whether intended for sale or the entity's use, during the reporting period charged to research and development projects, excluding in-process research and development acquired in a business combination consummated during the period. Excludes software research and development, which has a separate concept.",
"label": "Research and Development Expense (Excluding Acquired in Process Cost)",
"terseLabel": "Research and Development Expense (Excluding Acquired in Process Cost)",
"verboseLabel": "Research and development"
}
}
},
"localname": "ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ResearchAndDevelopmentExpenseMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption in which the reported facts about research and development expense have been included.",
"label": "Research and Development Expense [Member]",
"terseLabel": "Research and development",
"verboseLabel": "Research and development"
}
}
},
"localname": "ResearchAndDevelopmentExpenseMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails",
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ResearchAndDevelopmentInProcess": {
"auth_ref": [],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 9.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
},
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 5.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of purchased research and development assets that are acquired in a business combination have no alternative future use and are therefore written off in the period of acquisition.",
"label": "Research and Development in Process",
"terseLabel": "Acquired in-process research and development"
}
}
},
"localname": "ResearchAndDevelopmentInProcess",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ResearchDevelopmentAndComputerSoftwarePolicyTextBlock": {
"auth_ref": [
"r62",
"r158",
"r334",
"r335",
"r861"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for its research and development and computer software activities including the accounting treatment for costs incurred for (1) research and development activities, (2) development of computer software for internal use, (3) computer software to be sold, leased or otherwise marketed as a separate product or as part of a product or process and (4) in-process research and development acquired in a purchase business combination.",
"label": "Research, Development, and Computer Software, Policy [Policy Text Block]",
"terseLabel": "Research and Development Expenses"
}
}
},
"localname": "ResearchDevelopmentAndComputerSoftwarePolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ResearchMember": {
"auth_ref": [
"r559"
],
"lang": {
"en-us": {
"role": {
"documentation": "Research tax credit carryforwards arising from certain qualifying expenditures incurred to develop new products and processes.",
"label": "Research Tax Credit Carryforward [Member]",
"terseLabel": "Research"
}
}
},
"localname": "ResearchMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RestructuringCharges": {
"auth_ref": [
"r151",
"r351",
"r352",
"r353"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 9.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expenses associated with exit or disposal activities pursuant to an authorized plan. Excludes expenses related to a discontinued operation or an asset retirement obligation.",
"label": "Restructuring Charges",
"terseLabel": "Restructuring charges"
}
}
},
"localname": "RestructuringCharges",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RetainedEarningsAccumulatedDeficit": {
"auth_ref": [
"r46",
"r379",
"r526",
"r793",
"r821",
"r823"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 5.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cumulative amount of the reporting entity's undistributed earnings or deficit.",
"label": "Retained Earnings (Accumulated Deficit)",
"verboseLabel": "Retained earnings"
}
}
},
"localname": "RetainedEarningsAccumulatedDeficit",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RetainedEarningsMember": {
"auth_ref": [
"r180",
"r181",
"r182",
"r185",
"r191",
"r193",
"r294",
"r523",
"r524",
"r525",
"r567",
"r568",
"r818",
"r820"
],
"lang": {
"en-us": {
"role": {
"documentation": "The cumulative amount of the reporting entity's undistributed earnings or deficit.",
"label": "Retained Earnings [Member]",
"terseLabel": "Retained Earnings",
"verboseLabel": "Retained earnings"
}
}
},
"localname": "RetainedEarningsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RetirementPlanSponsorLocationAxis": {
"auth_ref": [
"r406",
"r407",
"r408",
"r409",
"r410",
"r411",
"r412",
"r413",
"r414",
"r415",
"r416",
"r417",
"r418",
"r419",
"r420",
"r421",
"r422",
"r423",
"r424",
"r425",
"r426",
"r427",
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r436",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r450",
"r451",
"r452",
"r454",
"r457",
"r461",
"r462",
"r463",
"r464",
"r465",
"r466",
"r467",
"r468",
"r469",
"r470",
"r471",
"r472",
"r473",
"r479",
"r483",
"r489"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by location of employer sponsoring plan designed to provide retirement benefits. Includes, but is not limited to, defined benefit and defined contribution plans.",
"label": "Retirement Plan Sponsor Location [Axis]",
"terseLabel": "Retirement Plan Sponsor Location [Axis]"
}
}
},
"localname": "RetirementPlanSponsorLocationAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RetirementPlanSponsorLocationDomain": {
"auth_ref": [
"r406",
"r407",
"r408",
"r409",
"r410",
"r411",
"r412",
"r413",
"r414",
"r415",
"r416",
"r417",
"r418",
"r419",
"r420",
"r421",
"r422",
"r423",
"r424",
"r425",
"r426",
"r427",
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r436",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r450",
"r451",
"r452",
"r454",
"r457",
"r461",
"r462",
"r463",
"r464",
"r465",
"r466",
"r467",
"r468",
"r469",
"r470",
"r471",
"r472",
"r473",
"r479",
"r483",
"r489"
],
"lang": {
"en-us": {
"role": {
"documentation": "Location of employer sponsoring plan designed to provide retirement benefits. Includes, but is not limited to, defined benefit and defined contribution plans.",
"label": "Retirement Plan Sponsor Location [Domain]",
"terseLabel": "Retirement Plan Sponsor Location [Domain]"
}
}
},
"localname": "RetirementPlanSponsorLocationDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RetirementPlanTypeAxis": {
"auth_ref": [
"r403",
"r404",
"r405",
"r406",
"r407",
"r408",
"r409",
"r410",
"r411",
"r412",
"r413",
"r414",
"r415",
"r416",
"r417",
"r418",
"r419",
"r420",
"r421",
"r422",
"r423",
"r424",
"r425",
"r426",
"r427",
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r436",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r451",
"r452",
"r454",
"r457",
"r460",
"r464",
"r465",
"r466",
"r467",
"r468",
"r469",
"r470",
"r471",
"r472",
"r473",
"r474",
"r475",
"r478",
"r479",
"r480",
"r481",
"r482",
"r483",
"r486",
"r487",
"r488",
"r489"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of retirement benefit plan. Includes, but is not limited to, retirement benefit arrangement for defined benefit pension and other postretirement plans, retirement benefit arrangement for defined contribution pension and other postretirement plans, and special and contractual termination benefits payable upon retirement.",
"label": "Retirement Plan Type [Axis]",
"terseLabel": "Retirement Plan Type [Axis]"
}
}
},
"localname": "RetirementPlanTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RetirementPlanTypeDomain": {
"auth_ref": [
"r403",
"r404",
"r405",
"r406",
"r407",
"r408",
"r409",
"r410",
"r411",
"r412",
"r413",
"r414",
"r415",
"r416",
"r417",
"r418",
"r419",
"r420",
"r421",
"r422",
"r423",
"r424",
"r425",
"r426",
"r427",
"r428",
"r430",
"r431",
"r432",
"r433",
"r434",
"r435",
"r436",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r451",
"r452",
"r454",
"r457",
"r460",
"r464",
"r465",
"r466",
"r467",
"r468",
"r469",
"r470",
"r471",
"r472",
"r473",
"r474",
"r475",
"r478",
"r479",
"r480",
"r481",
"r482",
"r483",
"r486",
"r487",
"r488",
"r489"
],
"lang": {
"en-us": {
"role": {
"documentation": "Type of plan designed to provide participants with retirement benefits. Includes, but is not limited to, retirement benefit arrangement for defined benefit pension and other postretirement plans, retirement benefit arrangement for defined contribution pension and other postretirement plans, and special and contractual termination benefits payable upon retirement.",
"label": "Retirement Plan Type [Domain]",
"terseLabel": "Retirement Plan Type [Domain]"
}
}
},
"localname": "RetirementPlanTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EmployeeBenefitPlansNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RevenueFromContractWithCustomerAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Revenue from Contract with Customer [Abstract]",
"terseLabel": "Revenue from Contract with Customer [Abstract]"
}
}
},
"localname": "RevenueFromContractWithCustomerAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_RevenueFromContractWithCustomerTextBlock": {
"auth_ref": [
"r384",
"r385",
"r386",
"r387",
"r388",
"r389",
"r391",
"r392",
"r394",
"r402"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure of revenue from contract with customer to transfer good or service and to transfer nonfinancial asset. Includes, but is not limited to, disaggregation of revenue, credit loss recognized from contract with customer, judgment and change in judgment related to contract with customer, and asset recognized from cost incurred to obtain or fulfill contract with customer. Excludes insurance and lease contracts.",
"label": "Revenue from Contract with Customer [Text Block]",
"terseLabel": "Revenues"
}
}
},
"localname": "RevenueFromContractWithCustomerTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Revenues"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RevenueRecognitionPolicyTextBlock": {
"auth_ref": [
"r159",
"r160"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for revenue. Includes revenue from contract with customer and from other sources.",
"label": "Revenue [Policy Text Block]",
"terseLabel": "Revenue Recognition"
}
}
},
"localname": "RevenueRecognitionPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RevenueRemainingPerformanceObligation": {
"auth_ref": [
"r390"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of transaction price allocated to performance obligation that has not been recognized as revenue.",
"label": "Revenue, Remaining Performance Obligation, Amount",
"terseLabel": "Remaining performance obligation, amount"
}
}
},
"localname": "RevenueRemainingPerformanceObligation",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_Revenues": {
"auth_ref": [
"r110",
"r163",
"r232",
"r233",
"r242",
"r247",
"r248",
"r254",
"r255",
"r258",
"r293",
"r680",
"r778"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 1.0,
"parentTag": "us-gaap_OperatingIncomeLoss",
"weight": 1.0
},
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of revenue recognized from goods sold, services rendered, insurance premiums, or other activities that constitute an earning process. Includes, but is not limited to, investment and interest income before deduction of interest expense when recognized as a component of revenue, and sales and trading gain (loss).",
"label": "Revenues",
"netLabel": "Revenues",
"terseLabel": "Revenues",
"totalLabel": "Total revenues from anti-CD20 therapeutic programs",
"verboseLabel": "Total revenues"
}
}
},
"localname": "Revenues",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails",
"http://www.biogenidec.com/role/RevenuesNarrativeDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesbyProductDetails",
"http://www.biogenidec.com/role/RevenuesRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RevolvingCreditFacilityMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Arrangement in which loan proceeds can continuously be obtained following repayments, but the total amount borrowed cannot exceed a specified maximum amount.",
"label": "Revolving Credit Facility [Member]",
"terseLabel": "Revolving Credit Facility"
}
}
},
"localname": "RevolvingCreditFacilityMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RightOfUseAssetObtainedInExchangeForOperatingLeaseLiability": {
"auth_ref": [
"r711",
"r716"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase in right-of-use asset obtained in exchange for operating lease liability.",
"label": "Right-of-Use Asset Obtained in Exchange for Operating Lease Liability",
"terseLabel": "Operating lease assets obtained in exchange for lease obligations"
}
}
},
"localname": "RightOfUseAssetObtainedInExchangeForOperatingLeaseLiability",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseSupplementalCashFlowDisclosureDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RoyaltyMember": {
"auth_ref": [
"r395"
],
"lang": {
"en-us": {
"role": {
"documentation": "Money for usage-based right to asset.",
"label": "Royalty [Member]",
"terseLabel": "Royalty"
}
}
},
"localname": "RoyaltyMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SalesMember": {
"auth_ref": [
"r653"
],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing revenue from sale of goods and services rendered in the normal course of business.",
"label": "Sales [Member]",
"verboseLabel": "Revenues"
}
}
},
"localname": "SalesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasCashFlowHedgingInstrumentsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SalesReturnsAndAllowancesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A revision in the estimated reserve needed for product returns and price or other concessions granted to customers.",
"label": "Sales Returns and Allowances [Member]",
"terseLabel": "Returns"
}
}
},
"localname": "SalesReturnsAndAllowancesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ScheduleOfAccruedLiabilitiesTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of accrued liabilities.",
"label": "Schedule of Accrued Liabilities [Table Text Block]",
"verboseLabel": "Accrued expenses and other"
}
}
},
"localname": "ScheduleOfAccruedLiabilitiesTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfAccumulatedOtherComprehensiveIncomeLossTableTextBlock": {
"auth_ref": [
"r98",
"r691",
"r692"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of accumulated other comprehensive income (loss).",
"label": "Schedule of Accumulated Other Comprehensive Income (Loss) [Table Text Block]",
"terseLabel": "Schedule of Accumulated Other Comprehensive Income (Loss)"
}
}
},
"localname": "ScheduleOfAccumulatedOtherComprehensiveIncomeLossTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTable": {
"auth_ref": [
"r207"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule for securities (including those issuable pursuant to contingent stock agreements) that could potentially dilute basic earnings per share (EPS) in the future that were not included in the computation of diluted EPS because to do so would increase EPS amounts or decrease loss per share amounts for the period presented, by Antidilutive Securities.",
"label": "Schedule of Antidilutive Securities Excluded from Computation of Earnings Per Share [Table]",
"terseLabel": "Schedule of Antidilutive Securities Excluded from Computation of Earnings Per Share [Table]"
}
}
},
"localname": "ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfAvailableForSaleSecuritiesLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Debt Securities, Available-for-sale [Line Items]",
"terseLabel": "Debt Securities, Available-for-sale [Line Items]"
}
}
},
"localname": "ScheduleOfAvailableForSaleSecuritiesLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfAvailableForSaleSecuritiesReconciliationTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the reconciliation of available-for-sale securities from cost basis to fair value.",
"label": "Schedule of Available-for-sale Securities Reconciliation [Table Text Block]",
"terseLabel": "Debt and equity securities, available-for-sale"
}
}
},
"localname": "ScheduleOfAvailableForSaleSecuritiesReconciliationTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfAvailableForSaleSecuritiesTable": {
"auth_ref": [
"r276",
"r279",
"r282",
"r283",
"r284",
"r286",
"r780",
"r781"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of available-for-sale securities which includes, but is not limited to, changes in the cost basis and fair value, fair value and gross unrealized gain (loss), fair values by type of security, contractual maturity and classification, amortized cost basis, contracts to acquire securities to be accounted for as available-for-sale, debt maturities, transfers to trading, change in net unrealized holding gain (loss) net of tax, continuous unrealized loss position fair value, aggregate losses qualitative disclosures, other than temporary impairment (OTTI) losses or other disclosures related to available for sale securities.",
"label": "Schedule of Available-for-sale Securities [Table]",
"terseLabel": "Schedule of Available-for-sale Securities [Table]"
}
}
},
"localname": "ScheduleOfAvailableForSaleSecuritiesTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfBusinessAcquisitionsByAcquisitionContingentConsiderationTable": {
"auth_ref": [
"r597"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of information about a contingent payment arrangement including the terms that will result in payment and the accounting treatment that will be followed if such contingency occurs, including the potential impact on earnings per share if the contingency is to be settled in shares of common stock of the entity. The description also may include the period over which amounts are expected to be paid, and changes in the amount since the previous reporting period. This also includes contingent options and commitments.",
"label": "Schedule of Business Acquisitions by Acquisition, Contingent Consideration [Table]",
"terseLabel": "Schedule of Business Acquisitions by Acquisition, Contingent Consideration [Table]"
}
}
},
"localname": "ScheduleOfBusinessAcquisitionsByAcquisitionContingentConsiderationTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsFairValueofContingentConsiderationObligationsDetails",
"http://www.biogenidec.com/role/FairValueMeasurementsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfBusinessAcquisitionsByAcquisitionTable": {
"auth_ref": [
"r585",
"r586"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule reflecting each material business combination (or series of individually immaterial business combinations) completed during the period, including background, timing, and recognized assets and liabilities.",
"label": "Schedule of Business Acquisitions, by Acquisition [Table]",
"terseLabel": "Schedule of Business Acquisitions, by Acquisition [Table]"
}
}
},
"localname": "ScheduleOfBusinessAcquisitionsByAcquisitionTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfCashAndCashEquivalentsTable": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of cash and cash equivalent balances. This table excludes restricted cash balances.",
"label": "Schedule of Cash and Cash Equivalents [Table]",
"terseLabel": "Schedule of Cash and Cash Equivalents [Table]"
}
}
},
"localname": "ScheduleOfCashAndCashEquivalentsTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofFinancialAssetswithMaturitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfCashAndCashEquivalentsTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of cash and cash equivalents.",
"label": "Schedule of Cash and Cash Equivalents [Table Text Block]",
"verboseLabel": "Summary of financial assets with maturities of less than 90 days included within cash and cash equivalents"
}
}
},
"localname": "ScheduleOfCashAndCashEquivalentsTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfCashFlowSupplementalDisclosuresTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of supplemental cash flow information for the periods presented.",
"label": "Schedule of Cash Flow, Supplemental Disclosures [Table Text Block]",
"terseLabel": "Supplemental cash flow information"
}
}
},
"localname": "ScheduleOfCashFlowSupplementalDisclosuresTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfCollaborativeArrangementsAndNoncollaborativeArrangementTransactionsTable": {
"auth_ref": [
"r604"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about collaborative arrangement and arrangement other than collaborative applicable to revenue-generating activity or operations.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Table]",
"terseLabel": "Collaborative Arrangement and Arrangement Other than Collaborative [Table]"
}
}
},
"localname": "ScheduleOfCollaborativeArrangementsAndNoncollaborativeArrangementTransactionsTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsCopromotionProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOCREVUSDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsPretaxProfitSharingFormulaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsProfitSharingDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsRevenuesfromAntiCD20TherapeuticProgramsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfCompensationCostForShareBasedPaymentArrangementsAllocationOfShareBasedCompensationCostsByPlanTableTextBlock": {
"auth_ref": [
"r516"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of cost recognized for award under share-based payment arrangement by plan. Includes, but is not limited to, related tax benefit.",
"label": "Share-based Payment Arrangement, Cost by Plan [Table Text Block]",
"verboseLabel": "Summary of share-based compensation expense associated with each of our share-based compensating programs"
}
}
},
"localname": "ScheduleOfCompensationCostForShareBasedPaymentArrangementsAllocationOfShareBasedCompensationCostsByPlanTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfComponentsOfIncomeTaxExpenseBenefitTableTextBlock": {
"auth_ref": [
"r565"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of income tax expense attributable to continuing operations for each year presented including, but not limited to: current tax expense (benefit), deferred tax expense (benefit), investment tax credits, government grants, the benefits of operating loss carryforwards, tax expense that results from allocating certain tax benefits either directly to contributed capital or to reduce goodwill or other noncurrent intangible assets of an acquired entity, adjustments of a deferred tax liability or asset for enacted changes in tax laws or rates or a change in the tax status of the entity, and adjustments of the beginning-of-the-year balances of a valuation allowance because of a change in circumstances that causes a change in judgment about the realizability of the related deferred tax asset in future years.",
"label": "Schedule of Components of Income Tax Expense (Benefit) [Table Text Block]",
"terseLabel": "Income before income tax provision and the income tax expense"
}
}
},
"localname": "ScheduleOfComponentsOfIncomeTaxExpenseBenefitTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfDebtTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of information pertaining to short-term and long-debt instruments or arrangements, including but not limited to identification of terms, features, collateral requirements and other information necessary to a fair presentation.",
"label": "Schedule of Debt [Table Text Block]",
"terseLabel": "Summary of indebtedness"
}
}
},
"localname": "ScheduleOfDebtTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock": {
"auth_ref": [
"r555"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of net deferred tax asset or liability recognized in an entity's statement of financial position, including the following: the total of all deferred tax liabilities, the total of all deferred tax assets, the total valuation allowance recognized for deferred tax assets.",
"label": "Schedule of Deferred Tax Assets and Liabilities [Table Text Block]",
"terseLabel": "Components of deferred tax assets and liabilities"
}
}
},
"localname": "ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfDerivativeInstrumentsGainLossInStatementOfFinancialPerformanceTextBlock": {
"auth_ref": [
"r640",
"r648",
"r655"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the location and amount of derivative instruments and nonderivative instruments designated as hedging instruments reported before netting adjustments, and the amount of gain (loss) on derivative instruments and nonderivative instruments designated and qualified as hedging instruments.",
"label": "Derivative Instruments, Gain (Loss) [Table Text Block]",
"verboseLabel": "Summary of the effect of derivatives designated as cash flow hedging instruments on our consolidated statements of income"
}
}
},
"localname": "ScheduleOfDerivativeInstrumentsGainLossInStatementOfFinancialPerformanceTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfDerivativeInstrumentsInStatementOfFinancialPositionFairValueTextBlock": {
"auth_ref": [
"r644"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the location and fair value amounts of derivative instruments (and nonderivative instruments that are designated and qualify as hedging instruments) reported in the statement of financial position.",
"label": "Schedule of Derivative Instruments in Statement of Financial Position, Fair Value [Table Text Block]",
"verboseLabel": "Summary of the fair value for our outstanding derivatives"
}
}
},
"localname": "ScheduleOfDerivativeInstrumentsInStatementOfFinancialPositionFairValueTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock": {
"auth_ref": [
"r206"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of an entity's basic and diluted earnings per share calculations, including a reconciliation of numerators and denominators of the basic and diluted per-share computations for income from continuing operations.",
"label": "Schedule of Earnings Per Share, Basic and Diluted [Table Text Block]",
"verboseLabel": "Basic and diluted earnings per share"
}
}
},
"localname": "ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock": {
"auth_ref": [
"r540"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the reconciliation using percentage or dollar amounts of the reported amount of income tax expense attributable to continuing operations for the year to the amount of income tax expense that would result from applying domestic federal statutory tax rates to pretax income from continuing operations.",
"label": "Schedule of Effective Income Tax Rate Reconciliation [Table Text Block]",
"verboseLabel": "Reconciliation between the U.S. federal statutory tax rate and effective tax rate"
}
}
},
"localname": "ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTable": {
"auth_ref": [
"r493",
"r516",
"r527"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about amount recognized for award under share-based payment arrangement. Includes, but is not limited to, amount expensed in statement of income or comprehensive income, amount capitalized in statement of financial position, and corresponding reporting line item in financial statements.",
"label": "Share-based Payment Arrangement, Expensed and Capitalized, Amount [Table]",
"terseLabel": "Share-based Payment Arrangement, Expensed and Capitalized, Amount [Table]"
}
}
},
"localname": "ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock": {
"auth_ref": [
"r493",
"r516",
"r527"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of allocation of amount expensed and capitalized for award under share-based payment arrangement to statement of income or comprehensive income and statement of financial position. Includes, but is not limited to, corresponding line item in financial statement.",
"label": "Share-based Payment Arrangement, Expensed and Capitalized, Amount [Table Text Block]",
"verboseLabel": "Share-based compensation expense included in consolidated statements of income"
}
}
},
"localname": "ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfEmployeeStockOwnershipPlanESOPDisclosuresTable": {
"auth_ref": [
"r528"
],
"lang": {
"en-us": {
"role": {
"documentation": "Describes the details pertaining to each employee stock ownership plan.",
"label": "Schedule of Employee Stock Ownership Plan (ESOP) Disclosures [Table]",
"terseLabel": "Schedule of Employee Stock Ownership Plan (ESOP) Disclosures [Table]"
}
}
},
"localname": "ScheduleOfEmployeeStockOwnershipPlanESOPDisclosuresTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfEntityWideInformationRevenueFromExternalCustomersByProductsAndServicesTable": {
"auth_ref": [
"r254"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular presentation of the description and amount of revenues from a product or service, or a group of similar products or similar services, reported from external customers during the period, if the information is not provided as part of the reportable operating segment information.",
"label": "Revenue from External Customers by Products and Services [Table]",
"terseLabel": "Revenue from External Customers by Products and Services [Table]"
}
}
},
"localname": "ScheduleOfEntityWideInformationRevenueFromExternalCustomersByProductsAndServicesTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfEntityWideRevenueByMajorCustomersByReportingSegmentsTable": {
"auth_ref": [
"r258"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure about the extent of the entity's reliance on its major customers.",
"label": "Schedule of Revenue by Major Customers, by Reporting Segments [Table]",
"terseLabel": "Schedule of Revenue by Major Customers, by Reporting Segments [Table]"
}
}
},
"localname": "ScheduleOfEntityWideRevenueByMajorCustomersByReportingSegmentsTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationGeographicInformationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfFairValueAssetsAndLiabilitiesMeasuredOnRecurringBasisTableTextBlock": {
"auth_ref": [
"r663",
"r664"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of assets and liabilities, including [financial] instruments measured at fair value that are classified in stockholders' equity, if any, that are measured at fair value on a recurring basis. The disclosures contemplated herein include the fair value measurements at the reporting date by the level within the fair value hierarchy in which the fair value measurements in their entirety fall, segregating fair value measurements using quoted prices in active markets for identical assets (Level 1), significant other observable inputs (Level 2), and significant unobservable inputs (Level 3).",
"label": "Schedule of Fair Value, Assets and Liabilities Measured on Recurring Basis [Table Text Block]",
"verboseLabel": "Summary of assets and liabilities recorded at fair value"
}
}
},
"localname": "ScheduleOfFairValueAssetsAndLiabilitiesMeasuredOnRecurringBasisTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfFiniteLivedIntangibleAssetsTable": {
"auth_ref": [
"r321",
"r327",
"r755"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of assets, excluding financial assets and goodwill, lacking physical substance with a finite life.",
"label": "Schedule of Finite-Lived Intangible Assets [Table]",
"terseLabel": "Schedule of Finite-Lived Intangible Assets [Table]"
}
}
},
"localname": "ScheduleOfFiniteLivedIntangibleAssetsTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfGoodwillTable": {
"auth_ref": [
"r317",
"r318"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of goodwill and the changes during the year due to acquisition, sale, impairment or for other reasons.",
"label": "Schedule of Goodwill [Table]",
"terseLabel": "Schedule of Goodwill [Table]"
}
}
},
"localname": "ScheduleOfGoodwillTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfGoodwillTextBlock": {
"auth_ref": [
"r317",
"r318"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of goodwill by reportable segment and in total which includes a rollforward schedule.",
"label": "Schedule of Goodwill [Table Text Block]",
"verboseLabel": "Summary of roll forward of the changes in goodwill"
}
}
},
"localname": "ScheduleOfGoodwillTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfInventoryCurrentTableTextBlock": {
"auth_ref": [
"r32",
"r54",
"r55",
"r56"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the carrying amount as of the balance sheet date of merchandise, goods, commodities, or supplies held for future sale or to be used in manufacturing, servicing or production process.",
"label": "Schedule of Inventory, Current [Table Text Block]",
"verboseLabel": "Components of inventory"
}
}
},
"localname": "ScheduleOfInventoryCurrentTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InventoryTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfMaturitiesOfLongTermDebtTableTextBlock": {
"auth_ref": [
"r365"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of maturity and sinking fund requirement for long-term debt.",
"label": "Schedule of Maturities of Long-term Debt [Table Text Block]",
"terseLabel": "Total debt maturities"
}
}
},
"localname": "ScheduleOfMaturitiesOfLongTermDebtTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IndebtednessTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfNetInvestmentHedgesInAccumulatedOtherComprehensiveIncomeLossTableTextBlock": {
"auth_ref": [
"r650"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the effective portion of gains and losses on derivative instruments (and nonderivative instruments) designated and qualifying in net investment hedges recorded in accumulated other comprehensive income (loss) during the term of the hedging relationship and reclassified into earnings during the current period.",
"label": "Schedule of Net Investment Hedges in Accumulated Other Comprehensive Income (Loss) [Table Text Block]",
"terseLabel": "Summary of the effect of derivatives designated as net investment hedging instruments on our consolidated statement of income"
}
}
},
"localname": "ScheduleOfNetInvestmentHedgesInAccumulatedOtherComprehensiveIncomeLossTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfNotionalAmountsOfOutstandingDerivativePositionsTableTextBlock": {
"auth_ref": [
"r631"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the notional amounts of outstanding derivative positions.",
"label": "Schedule of Notional Amounts of Outstanding Derivative Positions [Table Text Block]",
"verboseLabel": "Foreign currency forward contracts that were entered into to hedge forecasted revenue"
}
}
},
"localname": "ScheduleOfNotionalAmountsOfOutstandingDerivativePositionsTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfOperatingLeasedAssetsTable": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of long-lived, depreciable assets that are subject to a operating lease agreements and are used in the normal conduct of business to produce goods and services. Examples may include land, buildings, machinery and equipment, and other types of furniture and equipment including, but not limited to, office equipment, furniture and fixtures, and computer equipment and software.",
"label": "Schedule of Operating Leased Assets [Table]",
"terseLabel": "Schedule of Operating Leased Assets [Table]"
}
}
},
"localname": "ScheduleOfOperatingLeasedAssetsTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfOtherNonoperatingIncomeExpenseTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of non-operating income or non-operating expense that may include amounts earned from dividends, interest on securities, gains (losses) on securities sold, equity earnings of unconsolidated affiliates, net gain (loss) on sales of business, interest expense and other miscellaneous income or expense items.",
"label": "Schedule of Other Nonoperating Income (Expense) [Table Text Block]",
"verboseLabel": "Other income (expense), net"
}
}
},
"localname": "ScheduleOfOtherNonoperatingIncomeExpenseTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfPropertyPlantAndEquipmentTable": {
"auth_ref": [
"r61",
"r342"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about physical assets used in the normal conduct of business and not intended for resale. Includes, but is not limited to, balances by class of assets, depreciation and depletion expense and method used, including composite depreciation, and accumulated deprecation.",
"label": "Property, Plant and Equipment [Table]",
"terseLabel": "Property, Plant and Equipment [Table]"
}
}
},
"localname": "ScheduleOfPropertyPlantAndEquipmentTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/PropertyPlantandEquipmentNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfQuarterlyFinancialInformationTableTextBlock": {
"auth_ref": [
"r214"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of quarterly financial data. Includes, but is not limited to, financial information for fiscal quarters, cumulative effect of a change in accounting principle and earnings per share data.",
"label": "Quarterly Financial Information [Table Text Block]",
"verboseLabel": "Quarterly Financial Data (Unaudited)"
}
}
},
"localname": "ScheduleOfQuarterlyFinancialInformationTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfRevenueFromExternalCustomersAttributedToForeignCountriesByGeographicAreaTextBlock": {
"auth_ref": [
"r119",
"r255"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the names of foreign countries from which revenue is material and the amount of revenue from external customers attributed to those countries. An entity may also provide subtotals of geographic information about groups of countries.",
"label": "Schedule of Revenue from External Customers Attributed to Foreign Countries by Geographic Area [Table Text Block]",
"verboseLabel": "Geographic information"
}
}
},
"localname": "ScheduleOfRevenueFromExternalCustomersAttributedToForeignCountriesByGeographicAreaTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformationTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfShareBasedCompensationArrangementsByShareBasedPaymentAwardTable": {
"auth_ref": [
"r494",
"r521"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about share-based payment arrangement.",
"label": "Schedule of Share-based Compensation Arrangements by Share-based Payment Award [Table]",
"terseLabel": "Schedule of Share-based Compensation Arrangements by Share-based Payment Award [Table]"
}
}
},
"localname": "ScheduleOfShareBasedCompensationArrangementsByShareBasedPaymentAwardTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock": {
"auth_ref": [
"r500",
"r510",
"r512"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure for stock option plans. Includes, but is not limited to, outstanding awards at beginning and end of year, grants, exercises, forfeitures, and weighted-average grant date fair value.",
"label": "Share-based Payment Arrangement, Option, Activity [Table Text Block]",
"terseLabel": "Stock option activity"
}
}
},
"localname": "ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfStockByClassTable": {
"auth_ref": [
"r75",
"r161",
"r217",
"r218",
"r369",
"r370",
"r371",
"r373",
"r374",
"r375",
"r376",
"r377",
"r378",
"r379"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule detailing information related to equity by class of stock. Class of stock includes common, convertible, and preferred stocks which are not redeemable or redeemable solely at the option of the issuer. It also includes preferred stock with redemption features that are solely within the control of the issuer and mandatorily redeemable stock if redemption is required to occur only upon liquidation or termination of the reporting entity.",
"label": "Schedule of Stock by Class [Table]",
"terseLabel": "Schedule of Stock by Class [Table]"
}
}
},
"localname": "ScheduleOfStockByClassTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfUnrecognizedTaxBenefitsRollForwardTableTextBlock": {
"auth_ref": [
"r551",
"r566"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the change in unrecognized tax benefits.",
"label": "Schedule of Unrecognized Tax Benefits Roll Forward [Table Text Block]",
"terseLabel": "Reconciliation of beginning and ending amount of unrecognized tax benefits"
}
}
},
"localname": "ScheduleOfUnrecognizedTaxBenefitsRollForwardTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfVariableInterestEntitiesTable": {
"auth_ref": [
"r611",
"r612",
"r619",
"r620",
"r621",
"r622",
"r623",
"r624",
"r625"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of qualitative and quantitative information related to variable interests the entity holds, whether or not such variable interest entity (VIE) is included in the reporting entity's consolidated financial statements. Includes, but is not limited to, description of the significant judgments and assumptions made in determining whether a variable interest (as defined) held by the entity requires the variable interest entity (VIE) (as defined) to be consolidated and (or) disclose information about its involvement with the VIE, individually or in aggregate (as applicable); the nature of restrictions, if any, on the consolidated VIE's assets and on the settlement of its liabilities reported by an entity in its statement of financial position, including the carrying amounts of such assets and liabilities; the nature of, and changes in, the risks associated with involvement in the VIE; how involvement with the VIE affects the entity's financial position, financial performance, and cash flows; the lack of recourse if creditors (or beneficial interest holders) of the consolidated VIE have no recourse to the general credit of the primary beneficiary (if applicable); the terms of arrangements, giving consideration to both explicit arrangements and implicit variable interests, if any, that could require the entity to provide financial support to the VIE, including events or circumstances that could expose the entity to a loss; the methodology used by the entity for determining whether or not it is the primary beneficiary of the variable interest entity; the significant factors considered and judgments made in determining that the power to direct the activities of a VIE that most significantly impact the VIE's economic performance are shared (as defined); the carrying amounts and classification of assets and liabilities of the VIE included in the statement of financial position; the entity's maximum exposure to loss, if any, as a result of its involvement with the VIE, including how the maximum exposure is determined and significant sources of the entity's exposure to the VIE; a tabular comparison of the carrying amounts of the assets and liabilities and the entity's maximum exposure to loss; information about any liquidity arrangements, guarantees, and (or) other commitments by third parties that may affect the fair value or risk of the entity's variable interest in the VIE; whether or not the entity has provided financial support or other support (explicitly or implicitly) to the VIE that it was not previously contractually required to provide or whether the entity intends to provide that support, including the type and amount of the support and the primary reasons for providing the support; and supplemental information the entity determines necessary to provide.",
"label": "Schedule of Variable Interest Entities [Table]",
"terseLabel": "Schedule of Variable Interest Entities [Table]"
}
}
},
"localname": "ScheduleOfVariableInterestEntitiesTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleofFiniteLivedIntangibleAssetsFutureAmortizationExpenseTableTextBlock": {
"auth_ref": [
"r327"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the amount of amortization expense expected to be recorded in succeeding fiscal years for finite-lived intangible assets.",
"label": "Schedule of Finite-Lived Intangible Assets, Future Amortization Expense [Table Text Block]",
"terseLabel": "Estimated future amortization of intangible assets"
}
}
},
"localname": "ScheduleofFiniteLivedIntangibleAssetsFutureAmortizationExpenseTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SegmentReportingAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Segment Reporting [Abstract]",
"terseLabel": "Segment Reporting [Abstract]"
}
}
},
"localname": "SegmentReportingAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_SegmentReportingDisclosureTextBlock": {
"auth_ref": [
"r228",
"r230",
"r231",
"r239",
"r241",
"r246",
"r250",
"r251",
"r252",
"r253",
"r254",
"r257",
"r258",
"r259"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for reporting segments including data and tables. Reportable segments include those that meet any of the following quantitative thresholds a) it's reported revenue, including sales to external customers and intersegment sales or transfers is 10 percent or more of the combined revenue, internal and external, of all operating segments b) the absolute amount of its reported profit or loss is 10 percent or more of the greater, in absolute amount of 1) the combined reported profit of all operating segments that did not report a loss or 2) the combined reported loss of all operating segments that did report a loss c) its assets are 10 percent or more of the combined assets of all operating segments.",
"label": "Segment Reporting Disclosure [Text Block]",
"verboseLabel": "Segment Information"
}
}
},
"localname": "SegmentReportingDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SegmentInformation"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SelectedQuarterlyFinancialInformationAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Selected Quarterly Financial Information [Abstract]",
"terseLabel": "Selected Quarterly Financial Information [Abstract]"
}
}
},
"localname": "SelectedQuarterlyFinancialInformationAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_SellingGeneralAndAdministrativeExpense": {
"auth_ref": [
"r122"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome": {
"order": 3.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate total costs related to selling a firm's product and services, as well as all other general and administrative expenses. Direct selling expenses (for example, credit, warranty, and advertising) are expenses that can be directly linked to the sale of specific products. Indirect selling expenses are expenses that cannot be directly linked to the sale of specific products, for example telephone expenses, Internet, and postal charges. General and administrative expenses include salaries of non-sales personnel, rent, utilities, communication, etc.",
"label": "Selling, General and Administrative Expense",
"terseLabel": "Selling, general and administrative"
}
}
},
"localname": "SellingGeneralAndAdministrativeExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SellingGeneralAndAdministrativeExpensesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing selling, general and administrative expense.",
"label": "Selling, General and Administrative Expenses [Member]",
"terseLabel": "Selling, general and administrative",
"verboseLabel": "Selling, general and administrative"
}
}
},
"localname": "SellingGeneralAndAdministrativeExpensesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/AcquisitionsNarrativeDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharebasedCompensationExpenseIncludedinConsolidationStatementsofIncomeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SellingGeneralAndAdministrativeExpensesPolicyTextBlock": {
"auth_ref": [
"r158",
"r530"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for inclusion of significant items in the selling, general and administrative (or similar) expense report caption.",
"label": "Selling, General and Administrative Expenses, Policy [Policy Text Block]",
"terseLabel": "Selling, General and Administrative Expenses"
}
}
},
"localname": "SellingGeneralAndAdministrativeExpensesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SeniorNotesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Bond that takes priority over other debt securities sold by the issuer. In the event the issuer goes bankrupt, senior debt holders receive priority for (must receive) repayment prior to (relative to) junior and unsecured (general) creditors.",
"label": "Senior Notes [Member]",
"terseLabel": "Senior Notes"
}
}
},
"localname": "SeniorNotesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofFairandCarryingValueofDebtInstrumentsDetails",
"http://www.biogenidec.com/role/IndebtednessNarrativeDetails",
"http://www.biogenidec.com/role/IndebtednessSummaryofIndebtednessDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SeriesAPreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Outstanding nonredeemable series A preferred stock or outstanding series A preferred stock. Classified within stockholders' equity if nonredeemable or redeemable solely at the option of the issuer. Classified within temporary equity if redemption is outside the control of the issuer.",
"label": "Series A Preferred Stock [Member]",
"terseLabel": "Series A"
}
}
},
"localname": "SeriesAPreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ShareBasedCompensation": {
"auth_ref": [
"r150"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows": {
"order": 2.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of noncash expense for share-based payment arrangement.",
"label": "Share-based Payment Arrangement, Noncash Expense",
"verboseLabel": "Share-based compensation"
}
}
},
"localname": "ShareBasedCompensation",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardVestingPeriod1": {
"auth_ref": [
"r495"
],
"lang": {
"en-us": {
"role": {
"documentation": "Period over which grantee's right to exercise award under share-based payment arrangement is no longer contingent on satisfaction of service or performance condition, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Includes, but is not limited to, combination of market, performance or service condition.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Award Vesting Period",
"terseLabel": "Vesting period"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardVestingPeriod1",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeitedInPeriod": {
"auth_ref": [
"r505"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of equity-based payment instruments, excluding stock (or unit) options, that were forfeited during the reporting period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Forfeited in Period",
"negatedLabel": "Forfeited (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeitedInPeriod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeituresWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r509"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average fair value as of the grant date of equity-based award plans other than stock (unit) option plans that were not exercised or put into effect as a result of the occurrence of a terminating event.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Forfeitures, Weighted Average Grant Date Fair Value",
"verboseLabel": "Forfeited (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeituresWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriod": {
"auth_ref": [
"r507"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of grants made during the period on other than stock (or unit) option plans (for example, phantom stock or unit plan, stock or unit appreciation rights plan, performance target plan).",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Grants in Period",
"terseLabel": "Number of shares granted (in shares)",
"verboseLabel": "Granted (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriodWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r507"
],
"lang": {
"en-us": {
"role": {
"documentation": "The weighted average fair value at grant date for nonvested equity-based awards issued during the period on other than stock (or unit) option plans (for example, phantom stock or unit plan, stock or unit appreciation rights plan, performance target plan).",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Grants in Period, Weighted Average Grant Date Fair Value",
"terseLabel": "Granted (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriodWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber": {
"auth_ref": [
"r506"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of non-vested equity-based payment instruments, excluding stock (or unit) options, that validly exist and are outstanding as of the balance sheet date.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Nonvested, Number",
"periodEndLabel": "Ending balance, unvested shares (in shares)",
"periodStartLabel": "Beginning balance, unvested shares (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Nonvested, Number of Shares [Roll Forward]",
"terseLabel": "Shares"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r506"
],
"lang": {
"en-us": {
"role": {
"documentation": "Per share or unit weighted-average fair value of nonvested award under share-based payment arrangement. Excludes share and unit options.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Nonvested, Weighted Average Grant Date Fair Value",
"periodEndLabel": "Ending balance, weighted average grant date fair value (in dollars per share)",
"periodStartLabel": "Beginning balance, weighted average grant date fair value (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValueRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Nonvested, Weighted Average Grant Date Fair Value [Abstract]",
"terseLabel": "Weighted Average Grant\u00a0Date Fair Value"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValueRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedInPeriod": {
"auth_ref": [
"r508"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of equity-based payment instruments, excluding stock (or unit) options, that vested during the reporting period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Vested in Period",
"negatedLabel": "Vested (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedInPeriod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedInPeriodTotalFairValue": {
"auth_ref": [
"r511"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value of share-based awards for which the grantee gained the right by satisfying service and performance requirements, to receive or retain shares or units, other instruments, or cash.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Vested in Period, Fair Value",
"terseLabel": "Total fair value of vested awards"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedInPeriodTotalFairValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedInPeriodWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r508"
],
"lang": {
"en-us": {
"role": {
"documentation": "The weighted average fair value as of grant date pertaining to an equity-based award plan other than a stock (or unit) option plan for which the grantee gained the right during the reporting period, by satisfying service and performance requirements, to receive or retain shares or units, other instruments, or cash in accordance with the terms of the arrangement.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Equity Instruments Other than Options, Vested in Period, Weighted Average Grant Date Fair Value",
"verboseLabel": "Vested (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedInPeriodWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate": {
"auth_ref": [
"r514"
],
"lang": {
"en-us": {
"role": {
"documentation": "The estimated dividend rate (a percentage of the share price) to be paid (expected dividends) to holders of the underlying shares over the option's term.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Fair Value Assumptions, Expected Dividend Rate",
"verboseLabel": "Expected dividend yield"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedVolatilityRate": {
"auth_ref": [
"r513"
],
"lang": {
"en-us": {
"role": {
"documentation": "The estimated measure of the percentage by which a share price is expected to fluctuate during a period. Volatility also may be defined as a probability-weighted measure of the dispersion of returns about the mean. The volatility of a share price is the standard deviation of the continuously compounded rates of return on the share over a specified period. That is the same as the standard deviation of the differences in the natural logarithms of the stock prices plus dividends, if any, over the period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Fair Value Assumptions, Expected Volatility Rate",
"terseLabel": "Range of expected stock price volatility"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedVolatilityRate",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateMaximum": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The maximum risk-free interest rate assumption that is used in valuing an option on its own shares.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Fair Value Assumptions, Risk Free Interest Rate, Maximum",
"verboseLabel": "Maximum range of risk-free interest rates"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateMaximum",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateMinimum": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The minimum risk-free interest rate assumption that is used in valuing an option on its own shares.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Fair Value Assumptions, Risk Free Interest Rate, Minimum",
"verboseLabel": "Minimum range of risk-free interest rates"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateMinimum",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award [Line Items]",
"terseLabel": "Share-based Compensation Arrangement by Share-based Payment Award"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfSharesAuthorized": {
"auth_ref": [
"r497"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares authorized for issuance under share-based payment arrangement.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Number of Shares Authorized",
"terseLabel": "Omnibus Plan number of shares authorized (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfSharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisesInPeriodTotalIntrinsicValue": {
"auth_ref": [
"r511"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of accumulated difference between fair value of underlying shares on dates of exercise and exercise price on options exercised (or share units converted) into shares.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Exercises in Period, Intrinsic Value",
"verboseLabel": "Total intrinsic value of options exercised"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisesInPeriodTotalIntrinsicValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresInPeriod": {
"auth_ref": [
"r505"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of shares under options that were cancelled during the reporting period as a result of occurrence of a terminating event specified in contractual agreements pertaining to the stock option plan.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Forfeitures in Period",
"negatedLabel": "Cancelled (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresInPeriod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriod": {
"auth_ref": [
"r503"
],
"lang": {
"en-us": {
"role": {
"documentation": "Net number of share options (or share units) granted during the period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Grants in Period, Net of Forfeitures",
"terseLabel": "Granted (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber": {
"auth_ref": [
"r502",
"r521"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of options outstanding, including both vested and non-vested options.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Outstanding, Number",
"periodEndLabel": "Ending balance, outstanding shares (in shares)",
"periodStartLabel": "Beginning balance, outstanding shares (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Outstanding [Roll Forward]",
"terseLabel": "Shares"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingRollForward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePrice": {
"auth_ref": [
"r501"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average price at which grantees can acquire the shares reserved for issuance under the stock option plan.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Outstanding, Weighted Average Exercise Price",
"periodEndLabel": "Ending balance, weighted average exercise price (in dollars per share)",
"periodStartLabel": "Beginning balance, weighted average exercise price (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePriceRollforward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Outstanding, Weighted Average Exercise Price [Abstract]",
"terseLabel": "Weighted Average Exercise Price"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePriceRollforward",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardAwardTypeAndPlanNameDomain": {
"auth_ref": [
"r492",
"r498"
],
"lang": {
"en-us": {
"role": {
"documentation": "Award under share-based payment arrangement.",
"label": "Award Type [Domain]",
"terseLabel": "Award Type [Domain]"
}
}
},
"localname": "ShareBasedCompensationArrangementsByShareBasedPaymentAwardAwardTypeAndPlanNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsAssumptionsUsedinValuationofMarketBasedStockUnitsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsCashSettledPerformanceSharesActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsMarketStockUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinCashActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceStockUnitsSettledinStockActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsPerformanceUnitsActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSummaryofSharebasedCompensationExpenseAssociatedwithEachSharebasedCompensatingProgramsDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsTimeVestedRestrictedStockUnitsActivityDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsExercisesInPeriodWeightedAverageExercisePrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average price at which option holders acquired shares when converting their stock options into shares.",
"label": "Share-based Compensation Arrangements by Share-based Payment Award, Options, Exercises in Period, Weighted Average Exercise Price",
"verboseLabel": "Exercised (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsExercisesInPeriodWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsForfeituresInPeriodWeightedAverageExercisePrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average price at which grantees could have acquired the underlying shares with respect to stock options that were terminated.",
"label": "Share-based Compensation Arrangements by Share-based Payment Award, Options, Forfeitures in Period, Weighted Average Exercise Price",
"verboseLabel": "Cancelled (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsForfeituresInPeriodWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageExercisePrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average per share amount at which grantees can acquire shares of common stock by exercise of options.",
"label": "Share-based Compensation Arrangements by Share-based Payment Award, Options, Grants in Period, Weighted Average Exercise Price",
"verboseLabel": "Granted (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationAwardTrancheOneMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "First portion of award under share-based payment arrangement differentiated by vesting feature, including, but not limited to, performance measure or service period.",
"label": "Share-based Payment Arrangement, Tranche One [Member]",
"terseLabel": "Share-based Payment Arrangement, Tranche One"
}
}
},
"localname": "ShareBasedCompensationAwardTrancheOneMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ShareBasedCompensationAwardTrancheThreeMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Third portion of award under share-based payment arrangement differentiated by vesting feature, including, but not limited to, performance measure or service period.",
"label": "Share-based Payment Arrangement, Tranche Three [Member]",
"terseLabel": "Share-based Payment Arrangement, Tranche Three"
}
}
},
"localname": "ShareBasedCompensationAwardTrancheThreeMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ShareBasedCompensationAwardTrancheTwoMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Second portion of award under share-based payment arrangement differentiated by vesting feature, including, but not limited to, performance measure or service period.",
"label": "Share-based Payment Arrangement, Tranche Two [Member]",
"terseLabel": "Share-based Payment Arrangement, Tranche Two"
}
}
},
"localname": "ShareBasedCompensationAwardTrancheTwoMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ShareRepurchaseProgramAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by share repurchase program.",
"label": "Share Repurchase Program [Axis]",
"terseLabel": "Share Repurchase Program [Axis]"
}
}
},
"localname": "ShareRepurchaseProgramAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareRepurchaseProgramDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the share repurchase program.",
"label": "Share Repurchase Program [Domain]",
"terseLabel": "Share Repurchase Program [Domain]"
}
}
},
"localname": "ShareRepurchaseProgramDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardAwardVestingRightsPercentage": {
"auth_ref": [
"r495"
],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of vesting of award under share-based payment arrangement.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Award Vesting Rights, Percentage",
"terseLabel": "Vesting percentage"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardAwardVestingRightsPercentage",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardExpirationPeriod": {
"auth_ref": [
"r496"
],
"lang": {
"en-us": {
"role": {
"documentation": "Period from grant date that an equity-based award expires, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Expiration Period",
"terseLabel": "Share based payment arrangement, term"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardExpirationPeriod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_SharesIssued": {
"auth_ref": [
"r372"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares of stock issued as of the balance sheet date, including shares that had been issued and were previously outstanding but which are now held in the treasury.",
"label": "Shares, Issued",
"periodEndLabel": "Ending balance (in shares)",
"periodStartLabel": "Beginning balance (in shares)"
}
}
},
"localname": "SharesIssued",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "sharesItemType"
},
"us-gaap_SignificantAccountingPoliciesTextBlock": {
"auth_ref": [
"r179"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for all significant accounting policies of the reporting entity.",
"label": "Significant Accounting Policies [Text Block]",
"terseLabel": "Summary of Significant Accounting Policies"
}
}
},
"localname": "SignificantAccountingPoliciesTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SoftwareAndSoftwareDevelopmentCostsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Purchased software applications and internally developed software for sale, licensing or long-term internal use.",
"label": "Software and Software Development Costs [Member]",
"terseLabel": "Computer\u00a0Software\u00a0and\u00a0Hardware"
}
}
},
"localname": "SoftwareAndSoftwareDevelopmentCostsMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesSummaryofPropertyPlantandEquipmentEstimatedUsefulLivesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_StateAndLocalJurisdictionMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Designated tax department of a state or local government entitled to levy and collect income taxes from the entity.",
"label": "State and Local Jurisdiction [Member]",
"terseLabel": "State and Local Jurisdiction"
}
}
},
"localname": "StateAndLocalJurisdictionMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_StatementClassOfStockAxis": {
"auth_ref": [
"r41",
"r42",
"r43",
"r161",
"r163",
"r197",
"r201",
"r202",
"r204",
"r206",
"r217",
"r218",
"r219",
"r293",
"r372",
"r680"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by the different classes of stock of the entity.",
"label": "Class of Stock [Axis]",
"terseLabel": "Class of Stock [Axis]"
}
}
},
"localname": "StatementClassOfStockAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_StatementEquityComponentsAxis": {
"auth_ref": [
"r77",
"r180",
"r181",
"r182",
"r185",
"r191",
"r193",
"r216",
"r294",
"r372",
"r379",
"r523",
"r524",
"r525",
"r567",
"r568",
"r686",
"r687",
"r688",
"r689",
"r690",
"r692",
"r818",
"r819",
"r820"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by component of equity.",
"label": "Equity Components [Axis]",
"terseLabel": "Equity Components [Axis]"
}
}
},
"localname": "StatementEquityComponentsAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/EquityReclassificationoutofAccumulatedOtherComprehensiveIncomeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_StatementLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Statement [Line Items]",
"terseLabel": "Statement [Line Items]"
}
}
},
"localname": "StatementLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_StatementOfCashFlowsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Statement of Cash Flows [Abstract]",
"terseLabel": "Statement of Cash Flows [Abstract]"
}
}
},
"localname": "StatementOfCashFlowsAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_StatementOfFinancialPositionAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Statement of Financial Position [Abstract]",
"terseLabel": "Statement of Financial Position [Abstract]"
}
}
},
"localname": "StatementOfFinancialPositionAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_StatementOfIncomeAndComprehensiveIncomeAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Statement of Comprehensive Income [Abstract]",
"terseLabel": "Statement of Comprehensive Income [Abstract]"
}
}
},
"localname": "StatementOfIncomeAndComprehensiveIncomeAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_StatementOfStockholdersEquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Statement of Stockholders' Equity [Abstract]",
"terseLabel": "Statement of Stockholders' Equity [Abstract]"
}
}
},
"localname": "StatementOfStockholdersEquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_StatementTable": {
"auth_ref": [
"r180",
"r181",
"r182",
"r216",
"r754"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule reflecting a Statement of Income, Statement of Cash Flows, Statement of Financial Position, Statement of Shareholders' Equity and Other Comprehensive Income, or other statement as needed.",
"label": "Statement [Table]",
"terseLabel": "Statement [Table]"
}
}
},
"localname": "StatementTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofCashFlows",
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails",
"http://www.biogenidec.com/role/LeasesScheduleofOperatingLeasesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_StockIssuedDuringPeriodSharesEmployeeStockPurchasePlans": {
"auth_ref": [
"r42",
"r43",
"r372",
"r379"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares issued during the period as a result of an employee stock purchase plan.",
"label": "Stock Issued During Period, Shares, Employee Stock Purchase Plans",
"terseLabel": "Shares issued under the 2015 ESPP (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesEmployeeStockPurchasePlans",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails",
"http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockIssuedDuringPeriodSharesStockOptionsExercised": {
"auth_ref": [
"r42",
"r43",
"r372",
"r379",
"r504"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of share options (or share units) exercised during the current period.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Exercises in Period",
"negatedLabel": "Exercised (in shares)",
"terseLabel": "Issuance of common stock under stock award plan (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesStockOptionsExercised",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/ShareBasedPaymentsStockOptionActivityDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockIssuedDuringPeriodSharesTreasuryStockReissued": {
"auth_ref": [
"r43",
"r372",
"r379"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of treasury shares or units reissued. Excludes reissuance of shares or units in treasury for award under share-based payment arrangement.",
"label": "Stock Issued During Period, Shares, Treasury Stock Reissued",
"terseLabel": "Issuance of common stock under stock option and stock purchase plans (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesTreasuryStockReissued",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockIssuedDuringPeriodValueEmployeeStockPurchasePlan": {
"auth_ref": [
"r42",
"r43",
"r372",
"r379"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate change in value for stock issued during the period as a result of employee stock purchase plan.",
"label": "Stock Issued During Period, Value, Employee Stock Purchase Plan",
"verboseLabel": "Cash received under the 2015 ESPP"
}
}
},
"localname": "StockIssuedDuringPeriodValueEmployeeStockPurchasePlan",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsSharesIssuedUnderEmployeeStockPurchasePlanDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockIssuedDuringPeriodValueStockOptionsExercised": {
"auth_ref": [
"r77",
"r372",
"r379"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Value of stock issued as a result of the exercise of stock options.",
"label": "Stock Issued During Period, Value, Stock Options Exercised",
"terseLabel": "Issuance of common stock under stock award plan"
}
}
},
"localname": "StockIssuedDuringPeriodValueStockOptionsExercised",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockIssuedDuringPeriodValueTreasuryStockReissued": {
"auth_ref": [
"r42",
"r43",
"r372",
"r379",
"r381"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Value of treasury shares or units reissued. Excludes reissuance of shares or units in treasury for award under share-based payment arrangement.",
"label": "Stock Issued During Period, Value, Treasury Stock Reissued",
"terseLabel": "Issuance of common stock under stock option and stock purchase plans"
}
}
},
"localname": "StockIssuedDuringPeriodValueTreasuryStockReissued",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockRepurchaseProgramAuthorizedAmount1": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of stock repurchase plan authorized.",
"label": "Stock Repurchase Program, Authorized Amount",
"terseLabel": "Authorized amount of share repurchases"
}
}
},
"localname": "StockRepurchaseProgramAuthorizedAmount1",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockRepurchasedAndRetiredDuringPeriodShares": {
"auth_ref": [
"r42",
"r43",
"r372",
"r379"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares that have been repurchased and retired during the period.",
"label": "Stock Repurchased and Retired During Period, Shares",
"terseLabel": "Retirement of common stock pursuant to the share repurchase program, at cost (in shares)"
}
}
},
"localname": "StockRepurchasedAndRetiredDuringPeriodShares",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockholdersEquity": {
"auth_ref": [
"r43",
"r48",
"r49",
"r163",
"r264",
"r293",
"r680"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 1.0,
"parentTag": "us-gaap_StockholdersEquityIncludingPortionAttributableToNoncontrollingInterest",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Total of all stockholders' equity (deficit) items, net of receivables from officers, directors, owners, and affiliates of the entity which are attributable to the parent. The amount of the economic entity's stockholders' equity attributable to the parent excludes the amount of stockholders' equity which is allocable to that ownership interest in subsidiary equity which is not attributable to the parent (noncontrolling interest, minority interest). This excludes temporary equity and is sometimes called permanent equity.",
"label": "Stockholders' Equity Attributable to Parent",
"totalLabel": "Total Biogen Inc. shareholders\u2019 equity"
}
}
},
"localname": "StockholdersEquity",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockholdersEquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Stockholders' Equity Attributable to Parent [Abstract]",
"verboseLabel": "Biogen Inc. shareholders\u2019 equity"
}
}
},
"localname": "StockholdersEquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "stringItemType"
},
"us-gaap_StockholdersEquityIncludingPortionAttributableToNoncontrollingInterest": {
"auth_ref": [
"r163",
"r180",
"r181",
"r182",
"r185",
"r191",
"r293",
"r294",
"r379",
"r523",
"r524",
"r525",
"r567",
"r568",
"r606",
"r607",
"r626",
"r680",
"r686",
"r687",
"r692",
"r819",
"r820"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 1.0,
"parentTag": "us-gaap_LiabilitiesAndStockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of stockholders' equity (deficit), net of receivables from officers, directors, owners, and affiliates of the entity, attributable to both the parent and noncontrolling interests. Amount excludes temporary equity. Alternate caption for the concept is permanent equity.",
"label": "Stockholders' Equity, Including Portion Attributable to Noncontrolling Interest",
"periodEndLabel": "Ending balance",
"periodStartLabel": "Beginning balance",
"terseLabel": "Amount reclassified, net of tax, upon adoption of ASU 2016-01",
"totalLabel": "Total equity",
"verboseLabel": "Stockholders equity"
}
}
},
"localname": "StockholdersEquityIncludingPortionAttributableToNoncontrollingInterest",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets",
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityAccumulatedOtherComprehensiveIncomeLossDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockholdersEquityIncludingPortionAttributableToNoncontrollingInterestAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Stockholders' Equity, Including Portion Attributable to Noncontrolling Interest [Abstract]",
"verboseLabel": "Equity:"
}
}
},
"localname": "StockholdersEquityIncludingPortionAttributableToNoncontrollingInterestAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "stringItemType"
},
"us-gaap_StockholdersEquityNoteDisclosureTextBlock": {
"auth_ref": [
"r162",
"r379",
"r383"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for shareholders' equity comprised of portions attributable to the parent entity and noncontrolling interest, including other comprehensive income. Includes, but is not limited to, balances of common stock, preferred stock, additional paid-in capital, other capital and retained earnings, accumulated balance for each classification of other comprehensive income and amount of comprehensive income.",
"label": "Stockholders' Equity Note Disclosure [Text Block]",
"verboseLabel": "Equity"
}
}
},
"localname": "StockholdersEquityNoteDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/Equity"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_StockholdersEquityOther": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "This element represents movements included in the statement of changes in stockholders' equity which are not separately disclosed or provided for elsewhere in the taxonomy.",
"label": "Stockholders' Equity, Other",
"negatedTerseLabel": "Other"
}
}
},
"localname": "StockholdersEquityOther",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SubleaseIncome": {
"auth_ref": [
"r709",
"r716"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of sublease income excluding finance and operating lease expense.",
"label": "Sublease Income",
"negatedTerseLabel": "Sublease income"
}
}
},
"localname": "SubleaseIncome",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TaxCreditCarryforwardAmount": {
"auth_ref": [
"r558"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of the tax credit carryforward, before tax effects, available to reduce future taxable income under enacted tax laws.",
"label": "Tax Credit Carryforward, Amount",
"terseLabel": "Tax credit carryforward, amount"
}
}
},
"localname": "TaxCreditCarryforwardAmount",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TaxCreditCarryforwardAxis": {
"auth_ref": [
"r559"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by specific tax credit related to an unused tax credit.",
"label": "Tax Credit Carryforward [Axis]",
"terseLabel": "Tax Credit Carryforward [Axis]"
}
}
},
"localname": "TaxCreditCarryforwardAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_TaxCreditCarryforwardLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Tax Credit Carryforward [Line Items]",
"terseLabel": "Tax Credit Carryforward [Line Items]"
}
}
},
"localname": "TaxCreditCarryforwardLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_TaxCreditCarryforwardNameDomain": {
"auth_ref": [
"r559"
],
"lang": {
"en-us": {
"role": {
"documentation": "The name of the tax credit carryforward.",
"label": "Tax Credit Carryforward, Name [Domain]",
"terseLabel": "Tax Credit Carryforward, Name [Domain]"
}
}
},
"localname": "TaxCreditCarryforwardNameDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_TaxCreditCarryforwardTable": {
"auth_ref": [
"r559"
],
"lang": {
"en-us": {
"role": {
"documentation": "A listing of tax credit carryforwards available to reduce future taxable income including descriptions, amounts, expiration dates, limitations on use and the related deferred tax assets and valuation allowances.",
"label": "Tax Credit Carryforward [Table]",
"terseLabel": "Tax Credit Carryforward [Table]"
}
}
},
"localname": "TaxCreditCarryforwardTable",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_TaxesPayableCurrent": {
"auth_ref": [
"r66"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 2.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of obligations incurred and payable for statutory income, sales, use, payroll, excise, real, property and other taxes. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Taxes Payable, Current",
"verboseLabel": "Taxes payable"
}
}
},
"localname": "TaxesPayableCurrent",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TrademarksAndTradeNamesMember": {
"auth_ref": [
"r588"
],
"lang": {
"en-us": {
"role": {
"documentation": "Rights acquired through registration of a trademark to gain or protect exclusive use of a business name, symbol or other device or style, or rights either acquired through registration of a business name to gain or protect exclusive use thereof.",
"label": "Trademarks and Trade Names [Member]",
"verboseLabel": "Trademarks and trade names"
}
}
},
"localname": "TrademarksAndTradeNamesMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IntangibleAssetsandGoodwillSummaryofIntangibleAssetsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_TransfersAndServicingOfFinancialInstrumentsTypesOfFinancialInstrumentsDomain": {
"auth_ref": [
"r280",
"r281",
"r721",
"r722",
"r723",
"r724",
"r725",
"r726",
"r727",
"r728",
"r729",
"r730",
"r731",
"r732",
"r733",
"r735",
"r736",
"r737",
"r738",
"r739",
"r740",
"r741",
"r742",
"r743",
"r744",
"r745",
"r746",
"r747",
"r748",
"r749",
"r750",
"r751"
],
"lang": {
"en-us": {
"role": {
"documentation": "Instrument or contract that imposes a contractual obligation to deliver cash or another financial instrument or to exchange other financial instruments on potentially unfavorable terms and conveys a contractual right to receive cash or another financial instrument or to exchange other financial instruments on potentially favorable terms.",
"label": "Financial Instruments [Domain]",
"terseLabel": "Financial Instruments [Domain]"
}
}
},
"localname": "TransfersAndServicingOfFinancialInstrumentsTypesOfFinancialInstrumentsDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsSummaryofMarketableSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_TranslationAdjustmentForNetInvestmentHedgeNetOfTaxPeriodIncreaseDecrease": {
"auth_ref": [
"r691"
],
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in the cumulative translation adjustment from gain (loss), after tax, on foreign currency derivatives, that are designated as, and are effective as, economic hedges of a net investment in a foreign entity.",
"label": "Derivatives used in Net Investment Hedge, Net of Tax, Period Increase (Decrease)",
"terseLabel": "Net Gains/(Losses) Recognized in Other Comprehensive Income (Effective Portion) (in millions)"
}
}
},
"localname": "TranslationAdjustmentForNetInvestmentHedgeNetOfTaxPeriodIncreaseDecrease",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsSummaryoftheEffectofDerivativesDesignatedasNetInvestmentHedgingInstrumentsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TreasuryStockMember": {
"auth_ref": [
"r76",
"r381"
],
"lang": {
"en-us": {
"role": {
"documentation": "Shares of an entity that have been repurchased by the entity. This stock has no voting rights and receives no dividends. Note that treasury stock may be recorded at its total cost or separately as par (or stated) value and additional paid in capital. Classified within stockholders' equity if nonredeemable or redeemable solely at the option of the issuer. Classified within temporary equity if redemption is outside the control of the issuer.",
"label": "Treasury Stock [Member]",
"verboseLabel": "Treasury stock"
}
}
},
"localname": "TreasuryStockMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "domainItemType"
},
"us-gaap_TreasuryStockRetiredCostMethodAmount": {
"auth_ref": [
"r43",
"r372",
"r380"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of decrease of par value, additional paid in capital (APIC) and retained earnings of common and preferred stock retired from treasury when treasury stock is accounted for under the cost method.",
"label": "Treasury Stock, Retired, Cost Method, Amount",
"negatedTerseLabel": "Retirement of common stock pursuant to the share repurchase program, at cost"
}
}
},
"localname": "TreasuryStockRetiredCostMethodAmount",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TreasuryStockShares": {
"auth_ref": [
"r76",
"r381"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of common and preferred shares that were previously issued and that were repurchased by the issuing entity and held in treasury on the financial statement date. This stock has no voting rights and receives no dividends.",
"label": "Treasury Stock, Shares",
"terseLabel": "Treasury stock at cost, shares (in shares)"
}
}
},
"localname": "TreasuryStockShares",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheetsParenthetical"
],
"xbrltype": "sharesItemType"
},
"us-gaap_TreasuryStockSharesAcquired": {
"auth_ref": [
"r43",
"r372",
"r379"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares that have been repurchased during the period and are being held in treasury.",
"label": "Treasury Stock, Shares, Acquired",
"negatedLabel": "Repurchase of common stock pursuant to the share repurchase program, at cost (in shares)",
"terseLabel": "Repurchase of common stock, at cost (in shares)"
}
}
},
"localname": "TreasuryStockSharesAcquired",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity",
"http://www.biogenidec.com/role/EquityNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_TreasuryStockValue": {
"auth_ref": [
"r76",
"r381",
"r382"
],
"calculation": {
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets": {
"order": 6.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount allocated to treasury stock. Treasury stock is common and preferred shares of an entity that were issued, repurchased by the entity, and are held in its treasury.",
"label": "Treasury Stock, Value",
"negatedLabel": "Treasury stock, at cost; 23.8 million and 23.8 million shares, respectively"
}
}
},
"localname": "TreasuryStockValue",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedBalanceSheets"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TreasuryStockValueAcquiredCostMethod": {
"auth_ref": [
"r372",
"r379",
"r381"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Equity impact of the cost of common and preferred stock that were repurchased during the period. Recorded using the cost method.",
"label": "Treasury Stock, Value, Acquired, Cost Method",
"negatedLabel": "Repurchase of common stock pursuant to the share repurchase program, at cost"
}
}
},
"localname": "TreasuryStockValueAcquiredCostMethod",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofEquity"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TypeOfArrangementAxis": {
"auth_ref": [
"r604"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by collaborative arrangement and arrangement other than collaborative applicable to revenue-generating activity or operations.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Axis]",
"terseLabel": "Collaborative Arrangement and Arrangement Other than Collaborative [Axis]"
}
}
},
"localname": "TypeOfArrangementAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAcordaDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsAlkermesDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsBristolMyersSquibbDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsEisaiDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsIonisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsOtherArrangementsDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSamsungBioepisDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoAducanumabCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtoBAN2401andElenbecestatCollaborationDetails",
"http://www.biogenidec.com/role/CollaborativeandOtherRelationshipsSummaryofActivityRelatedtotheUCBCollaborationDetails",
"http://www.biogenidec.com/role/CommitmentsandContingenciesNarrativeDetails",
"http://www.biogenidec.com/role/FinancialInstrumentsNarrativeDetails",
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails",
"http://www.biogenidec.com/role/RevenuesOtherRevenuesDetails",
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_USTreasuryAndGovernmentMember": {
"auth_ref": [
"r428",
"r779",
"r824"
],
"lang": {
"en-us": {
"role": {
"documentation": "This category includes investments in debt securities issued by the United States Department of the Treasury, US Government Agencies and US Government-sponsored Enterprises. Such securities may include treasury bills (short-term maturities - one year or less), treasury notes (intermediate term maturities - two to ten years), and treasury bonds (long-term maturities - ten to thirty years), debt securities issued by the Government National Mortgage Association (Ginnie Mae) and debt securities issued by the Federal National Mortgage Association (Fannie Mae) or the Federal Home Loan Mortgage Corporation (Freddie Mac).",
"label": "US Treasury and Government [Member]",
"verboseLabel": "Government securities"
}
}
},
"localname": "USTreasuryAndGovernmentMember",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/FairValueMeasurementsSummaryofAssetsandLiabilitiesRecordedatFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_UnrealizedGainLossOnDerivatives": {
"auth_ref": [
"r151"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The net change in the difference between the fair value and the carrying value, or in the comparative fair values, of derivative instruments, including options, swaps, futures, and forward contracts, held at each balance sheet date, that was included in earnings for the period.",
"label": "Unrealized Gain (Loss) on Derivatives",
"terseLabel": "Unrealized gain (loss) on derivative instruments"
}
}
},
"localname": "UnrealizedGainLossOnDerivatives",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/DerivativeInstrumentsNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrealizedGainLossOnInvestments": {
"auth_ref": [
"r151"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of unrealized gain (loss) on investment.",
"label": "Unrealized Gain (Loss) on Investments",
"verboseLabel": "Unrealized gain (loss) on investments"
}
}
},
"localname": "UnrealizedGainLossOnInvestments",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefits": {
"auth_ref": [
"r534",
"r545"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of unrecognized tax benefits.",
"label": "Unrecognized Tax Benefits",
"periodEndLabel": "Ending balance ar December 31,",
"periodStartLabel": "Beginning balance at January 1,"
}
}
},
"localname": "UnrecognizedTaxBenefits",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsDecreasesResultingFromPriorPeriodTaxPositions": {
"auth_ref": [
"r546"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of decrease in unrecognized tax benefits resulting from tax positions taken in prior period tax returns.",
"label": "Unrecognized Tax Benefits, Decrease Resulting from Prior Period Tax Positions",
"negatedTerseLabel": "Reductions for tax positions of prior periods"
}
}
},
"localname": "UnrecognizedTaxBenefitsDecreasesResultingFromPriorPeriodTaxPositions",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsDecreasesResultingFromSettlementsWithTaxingAuthorities": {
"auth_ref": [
"r548"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of decrease in unrecognized tax benefits resulting from settlements with taxing authorities.",
"label": "Unrecognized Tax Benefits, Decrease Resulting from Settlements with Taxing Authorities",
"negatedTerseLabel": "Settlement refund (payment)"
}
}
},
"localname": "UnrecognizedTaxBenefitsDecreasesResultingFromSettlementsWithTaxingAuthorities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsIncomeTaxPenaltiesAndInterestExpense": {
"auth_ref": [
"r542"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expense for interest on an underpayment of income taxes and penalties related to a tax position claimed or expected to be claimed in the tax return.",
"label": "Unrecognized Tax Benefits, Income Tax Penalties and Interest Expense",
"terseLabel": "Net interest expense"
}
}
},
"localname": "UnrecognizedTaxBenefitsIncomeTaxPenaltiesAndInterestExpense",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsIncreasesResultingFromCurrentPeriodTaxPositions": {
"auth_ref": [
"r547"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase in unrecognized tax benefits resulting from tax positions that have been or will be taken in current period tax return.",
"label": "Unrecognized Tax Benefits, Increase Resulting from Current Period Tax Positions",
"terseLabel": "Additions based on tax positions related to the current period"
}
}
},
"localname": "UnrecognizedTaxBenefitsIncreasesResultingFromCurrentPeriodTaxPositions",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsIncreasesResultingFromPriorPeriodTaxPositions": {
"auth_ref": [
"r546"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase in unrecognized tax benefits resulting from tax positions taken in prior period tax returns.",
"label": "Unrecognized Tax Benefits, Increase Resulting from Prior Period Tax Positions",
"terseLabel": "Additions for tax positions of prior periods"
}
}
},
"localname": "UnrecognizedTaxBenefitsIncreasesResultingFromPriorPeriodTaxPositions",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsIncreasesResultingFromSettlementsWithTaxingAuthorities": {
"auth_ref": [
"r548"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase in unrecognized tax benefits resulting from settlements with taxing authorities.",
"label": "Unrecognized Tax Benefits, Increase Resulting from Settlements with Taxing Authorities",
"terseLabel": "Settlement refund (payment)"
}
}
},
"localname": "UnrecognizedTaxBenefitsIncreasesResultingFromSettlementsWithTaxingAuthorities",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsInterestOnIncomeTaxesAccrued": {
"auth_ref": [
"r542"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of interest expense accrued for an underpayment of income taxes.",
"label": "Unrecognized Tax Benefits, Interest on Income Taxes Accrued",
"terseLabel": "Unrecognized tax benefits, interest on income taxes accrued"
}
}
},
"localname": "UnrecognizedTaxBenefitsInterestOnIncomeTaxesAccrued",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsReductionsResultingFromLapseOfApplicableStatuteOfLimitations": {
"auth_ref": [
"r549"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of decrease in unrecognized tax benefits resulting from lapses of applicable statutes of limitations.",
"label": "Unrecognized Tax Benefits, Reduction Resulting from Lapse of Applicable Statute of Limitations",
"negatedTerseLabel": "Statute expirations"
}
}
},
"localname": "UnrecognizedTaxBenefitsReductionsResultingFromLapseOfApplicableStatuteOfLimitations",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesAccountingforUncertaintyinIncomeTaxesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnrecognizedTaxBenefitsThatWouldImpactEffectiveTaxRate": {
"auth_ref": [
"r550"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The total amount of unrecognized tax benefits that, if recognized, would affect the effective tax rate.",
"label": "Unrecognized Tax Benefits that Would Impact Effective Tax Rate",
"verboseLabel": "Unrecognized tax benefit"
}
}
},
"localname": "UnrecognizedTaxBenefitsThatWouldImpactEffectiveTaxRate",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/IncomeTaxesNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UseOfEstimates": {
"auth_ref": [
"r220",
"r221",
"r223",
"r224",
"r225",
"r226",
"r227"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for the use of estimates in the preparation of financial statements in conformity with generally accepted accounting principles.",
"label": "Use of Estimates, Policy [Policy Text Block]",
"terseLabel": "Use of Estimates"
}
}
},
"localname": "UseOfEstimates",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/SummaryofSignificantAccountingPoliciesPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ValuationAllowancesAndReservesBalance": {
"auth_ref": [
"r173",
"r177"
],
"calculation": {
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails": {
"order": 6.0,
"parentTag": "us-gaap_OtherLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of valuation and qualifying accounts and reserves.",
"label": "SEC Schedule, 12-09, Valuation Allowances and Reserves, Amount",
"periodEndLabel": "Ending balance",
"periodStartLabel": "Beginning balance",
"terseLabel": "Revenue-related reserves for discounts and allowances"
}
}
},
"localname": "ValuationAllowancesAndReservesBalance",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/OtherConsolidatedFinancialStatementDetailAccruedExpensesandOtherDetails",
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails",
"http://www.biogenidec.com/role/RevenuesTotalReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ValuationAllowancesAndReservesDomain": {
"auth_ref": [
"r173",
"r174",
"r175",
"r176",
"r177"
],
"lang": {
"en-us": {
"role": {
"documentation": "Valuation and qualifying accounts and reserves.",
"label": "SEC Schedule, 12-09, Valuation Allowances and Reserves [Domain]",
"terseLabel": "SEC Schedule, 12-09, Valuation Allowances and Reserves [Domain]"
}
}
},
"localname": "ValuationAllowancesAndReservesDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ValuationAllowancesAndReservesTypeAxis": {
"auth_ref": [
"r173",
"r174",
"r175",
"r176",
"r177"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by valuation and qualifying accounts and reserves.",
"label": "SEC Schedule, 12-09, Valuation Allowances and Reserves Type [Axis]",
"terseLabel": "SEC Schedule, 12-09, Valuation Allowances and Reserves Type [Axis]"
}
}
},
"localname": "ValuationAllowancesAndReservesTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/RevenuesReservesforDiscountsandAllowancesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_VariableInterestEntityDisclosureTextBlock": {
"auth_ref": [
"r629"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for a variable interest entity (VIE), including but not limited to, judgments and assumptions in determining whether to consolidate and in identifying the primary beneficiary, gain (loss) recognized on the initial consolidation of the VIE, terms of arrangements, amounts and classification of the VIE's assets and liabilities, and the entity's maximum exposure to loss.",
"label": "Variable Interest Entity Disclosure [Text Block]",
"verboseLabel": "Investments in Variable Interest Entities"
}
}
},
"localname": "VariableInterestEntityDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntities"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_VariableInterestEntityLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Variable Interest Entity [Line Items]",
"terseLabel": "Variable Interest Entity [Line Items]"
}
}
},
"localname": "VariableInterestEntityLineItems",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/InvestmentsinVariableInterestEntitiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_VariableLeaseCost": {
"auth_ref": [
"r708",
"r716"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of variable lease cost, excluded from lease liability, recognized when obligation for payment is incurred for finance and operating leases.",
"label": "Variable Lease, Cost",
"terseLabel": "Variable lease cost"
}
}
},
"localname": "VariableLeaseCost",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/LeasesOperatingLeaseCostsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_VestingAxis": {
"auth_ref": [
"r521"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by vesting schedule of award under share-based payment arrangement.",
"label": "Vesting [Axis]",
"terseLabel": "Vesting [Axis]"
}
}
},
"localname": "VestingAxis",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_VestingDomain": {
"auth_ref": [
"r521"
],
"lang": {
"en-us": {
"role": {
"documentation": "Vesting schedule of award under share-based payment arrangement.",
"label": "Vesting [Domain]",
"terseLabel": "Vesting [Domain]"
}
}
},
"localname": "VestingDomain",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ShareBasedPaymentsNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_WeightedAverageNumberDilutedSharesOutstandingAdjustment": {
"auth_ref": [
"r206"
],
"calculation": {
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails": {
"order": 2.0,
"parentTag": "us-gaap_WeightedAverageNumberOfDilutedSharesOutstanding",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "The sum of dilutive potential common shares or units used in the calculation of the diluted per-share or per-unit computation.",
"label": "Weighted Average Number Diluted Shares Outstanding Adjustment",
"totalLabel": "Dilutive potential common shares (in shares)"
}
}
},
"localname": "WeightedAverageNumberDilutedSharesOutstandingAdjustment",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_WeightedAverageNumberOfDilutedSharesOutstanding": {
"auth_ref": [
"r196",
"r206"
],
"calculation": {
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"lang": {
"en-us": {
"role": {
"documentation": "The average number of shares or units issued and outstanding that are used in calculating diluted EPS or earnings per unit (EPU), determined based on the timing of issuance of shares or units in the period.",
"label": "Weighted Average Number of Shares Outstanding, Diluted",
"terseLabel": "Diluted earnings per share attributable to Biogen Inc. (in shares)",
"totalLabel": "Diluted earnings per share attributable to Biogen Inc. (in shares)"
}
}
},
"localname": "WeightedAverageNumberOfDilutedSharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_WeightedAverageNumberOfSharesCommonStockSubjectToRepurchaseOrCancellation": {
"auth_ref": [
"r195"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares of common stock subject to repurchase or cancellation determined by relating the portion of time within a reporting period that these shares have been outstanding to the total time in that period. Common stock subject to repurchase are outstanding common shares that are contingently returnable (that is, subject to recall).",
"label": "Weighted Average Number of Shares, Common Stock Subject to Repurchase or Cancellation",
"terseLabel": "Repurchase of common stock (in shares)"
}
}
},
"localname": "WeightedAverageNumberOfSharesCommonStockSubjectToRepurchaseOrCancellation",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/EarningsperShareNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_WeightedAverageNumberOfSharesOutstandingAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Weighted Average Number of Shares Outstanding, Diluted [Abstract]",
"terseLabel": "Weighted-average shares used in calculating:",
"verboseLabel": "Weighted-average shares used in calculating:"
}
}
},
"localname": "WeightedAverageNumberOfSharesOutstandingAbstract",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_WeightedAverageNumberOfSharesOutstandingBasic": {
"auth_ref": [
"r194",
"r206"
],
"calculation": {
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails": {
"order": 1.0,
"parentTag": "us-gaap_WeightedAverageNumberOfDilutedSharesOutstanding",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Number of [basic] shares or units, after adjustment for contingently issuable shares or units and other shares or units not deemed outstanding, determined by relating the portion of time within a reporting period that common shares or units have been outstanding to the total time in that period.",
"label": "Weighted Average Number of Shares Outstanding, Basic",
"verboseLabel": "Basic earnings per share attributable to Biogen Inc. (in shares)"
}
}
},
"localname": "WeightedAverageNumberOfSharesOutstandingBasic",
"nsuri": "http://fasb.org/us-gaap/2020-01-31",
"presentation": [
"http://www.biogenidec.com/role/ConsolidatedStatementsofIncome",
"http://www.biogenidec.com/role/EarningsperShareBasicandDilutedEarningsPerShareDetails",
"http://www.biogenidec.com/role/QuarterlyFinancialDataUnauditedDetails"
],
"xbrltype": "sharesItemType"
}
},
"unitCount": 11
}
},
"std_ref": {
"r0": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222160&loc=d3e1107-107759"
},
"r1": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222160&loc=SL51721533-107759"
},
"r10": {
"Name": "Accounting Standards Codification",
"Paragraph": "4A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721663-107760"
},
"r100": {
"Name": "Accounting Standards Codification",
"Paragraph": "17B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL34724394-108580"
},
"r101": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669619-108580"
},
"r102": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669619-108580"
},
"r103": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669619-108580"
},
"r104": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669625-108580"
},
"r105": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669625-108580"
},
"r106": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=d3e557-108580"
},
"r107": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=116657188&loc=SL116659661-227067"
},
"r108": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121640130&loc=d3e1436-108581"
},
"r109": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(210.5-03(11))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r11": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721665-107760"
},
"r110": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(1))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r111": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(12))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r112": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(20))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r113": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(21))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r114": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(22))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r115": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(23))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r116": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(24))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r117": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(25))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r118": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r119": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.1)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r12": {
"Name": "Accounting Standards Codification",
"Paragraph": "5A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721671-107760"
},
"r120": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.1,2)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r121": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.2(a),(d))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r122": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.4)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r123": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.7(b))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r124": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.7)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r125": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.9)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=120395209&loc=SL114868664-224227"
},
"r126": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3151-108585"
},
"r127": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3179-108585"
},
"r128": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3179-108585"
},
"r129": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3179-108585"
},
"r13": {
"Name": "Accounting Standards Codification",
"Paragraph": "5B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721673-107760"
},
"r130": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3179-108585"
},
"r131": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3213-108585"
},
"r132": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3213-108585"
},
"r133": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3213-108585"
},
"r134": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3213-108585"
},
"r135": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3255-108585"
},
"r136": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3255-108585"
},
"r137": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3255-108585"
},
"r138": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3291-108585"
},
"r139": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3291-108585"
},
"r14": {
"Name": "Accounting Standards Codification",
"Paragraph": "5B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721673-107760"
},
"r140": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3291-108585"
},
"r141": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3291-108585"
},
"r142": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3367-108585"
},
"r143": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3444-108585"
},
"r144": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3000-108585"
},
"r145": {
"Name": "Accounting Standards Codification",
"Paragraph": "21D",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=SL94080555-108585"
},
"r146": {
"Name": "Accounting Standards Codification",
"Paragraph": "24",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3521-108585"
},
"r147": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3536-108585"
},
"r148": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3536-108585"
},
"r149": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3536-108585"
},
"r15": {
"Name": "Accounting Standards Codification",
"Paragraph": "5B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(e)",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721673-107760"
},
"r150": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3602-108585"
},
"r151": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3602-108585"
},
"r152": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3602-108585"
},
"r153": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121586364&loc=d3e3044-108585"
},
"r154": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121583591&loc=d3e4273-108586"
},
"r155": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121583591&loc=d3e4297-108586"
},
"r156": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=121583591&loc=SL98516268-108586"
},
"r157": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=84158767&loc=d3e18726-107790"
},
"r158": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=84158767&loc=d3e18780-107790"
},
"r159": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=84158767&loc=d3e18823-107790"
},
"r16": {
"Name": "Accounting Standards Codification",
"Paragraph": "5B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721673-107760"
},
"r160": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=84158767&loc=d3e18823-107790"
},
"r161": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(d))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r162": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(e)(1))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r163": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(g)(1)(ii))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r164": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(h)(1)(Note 1))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r165": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(h))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r166": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08.(e),(f))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r167": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08.(h)(1)(i))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r168": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08.(h))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r169": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08.(k)(1))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r17": {
"Name": "Accounting Standards Codification",
"Paragraph": "5C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721675-107760"
},
"r170": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08.(n))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r171": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-04(a))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24072-122690"
},
"r172": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-04.(a))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24072-122690"
},
"r173": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-09(Column B))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24092-122690"
},
"r174": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-09(Column C(1)))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24092-122690"
},
"r175": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-09(Column C(2)))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24092-122690"
},
"r176": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-09(Column D))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24092-122690"
},
"r177": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-09(Column E))",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24092-122690"
},
"r178": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-09)",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=120395691&loc=d3e24092-122690"
},
"r179": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "235",
"URI": "http://asc.fasb.org/topic&trid=2122369"
},
"r18": {
"Name": "Accounting Standards Codification",
"Paragraph": "5C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721675-107760"
},
"r180": {
"Name": "Accounting Standards Codification",
"Paragraph": "23",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=115929471&loc=d3e21914-107793"
},
"r181": {
"Name": "Accounting Standards Codification",
"Paragraph": "24",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=115929471&loc=d3e21930-107793"
},
"r182": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=115929471&loc=d3e21711-107793"
},
"r183": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=115929471&loc=d3e21728-107793"
},
"r184": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22499-107794"
},
"r185": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(3)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22499-107794"
},
"r186": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22694-107794"
},
"r187": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22694-107794"
},
"r188": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22583-107794"
},
"r189": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22595-107794"
},
"r19": {
"Name": "Accounting Standards Codification",
"Paragraph": "5D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721677-107760"
},
"r190": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22644-107794"
},
"r191": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22644-107794"
},
"r192": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22658-107794"
},
"r193": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc.fasb.org/extlink&oid=109234566&loc=d3e22663-107794"
},
"r194": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1448-109256"
},
"r195": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e2646-109256"
},
"r196": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1505-109256"
},
"r197": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1252-109256"
},
"r198": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1707-109256"
},
"r199": {
"Name": "Accounting Standards Codification",
"Paragraph": "23",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1757-109256"
},
"r2": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222160&loc=d3e957-107759"
},
"r20": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721683-107760"
},
"r200": {
"Name": "Accounting Standards Codification",
"Paragraph": "28A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1500-109256"
},
"r201": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1278-109256"
},
"r202": {
"Name": "Accounting Standards Codification",
"Paragraph": "55",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e2626-109256"
},
"r203": {
"Name": "Accounting Standards Codification",
"Paragraph": "60B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=SL5780133-109256"
},
"r204": {
"Name": "Accounting Standards Codification",
"Paragraph": "60B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=SL5780133-109256"
},
"r205": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=121326447&loc=d3e1337-109256"
},
"r206": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=6371337&loc=d3e3550-109257"
},
"r207": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=6371337&loc=d3e3550-109257"
},
"r208": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=6371337&loc=d3e3630-109257"
},
"r209": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=109243012&loc=SL65017193-207537"
},
"r21": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/subtopic&trid=2122178"
},
"r210": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=120380238&loc=d3e3842-109258"
},
"r211": {
"Name": "Accounting Standards Codification",
"Paragraph": "52",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=120380238&loc=d3e4984-109258"
},
"r212": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "260",
"URI": "http://asc.fasb.org/topic&trid=2144383"
},
"r213": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "270",
"URI": "http://asc.fasb.org/extlink&oid=116846552&loc=d3e543-108305"
},
"r214": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "270",
"URI": "http://asc.fasb.org/extlink&oid=121640914&loc=d3e1280-108306"
},
"r215": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "270",
"URI": "http://asc.fasb.org/topic&trid=2126967"
},
"r216": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "272",
"URI": "http://asc.fasb.org/extlink&oid=6828210&loc=d3e70191-108054"
},
"r217": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "272",
"URI": "http://asc.fasb.org/extlink&oid=6828210&loc=d3e70229-108054"
},
"r218": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "272",
"URI": "http://asc.fasb.org/extlink&oid=6373374&loc=d3e70434-108055"
},
"r219": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "272",
"URI": "http://asc.fasb.org/extlink&oid=6373374&loc=d3e70478-108055"
},
"r22": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6676-107765"
},
"r220": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e5967-108592"
},
"r221": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e5967-108592"
},
"r222": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e5967-108592"
},
"r223": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6161-108592"
},
"r224": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6191-108592"
},
"r225": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6061-108592"
},
"r226": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6132-108592"
},
"r227": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6143-108592"
},
"r228": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8657-108599"
},
"r229": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8672-108599"
},
"r23": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6676-107765"
},
"r230": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8721-108599"
},
"r231": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8721-108599"
},
"r232": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r233": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r234": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r235": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r236": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r237": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r238": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r239": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8736-108599"
},
"r24": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6676-107765"
},
"r240": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8813-108599"
},
"r241": {
"Name": "Accounting Standards Codification",
"Paragraph": "26",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8844-108599"
},
"r242": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8906-108599"
},
"r243": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8906-108599"
},
"r244": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8906-108599"
},
"r245": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8906-108599"
},
"r246": {
"Name": "Accounting Standards Codification",
"Paragraph": "31",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8924-108599"
},
"r247": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8933-108599"
},
"r248": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8933-108599"
},
"r249": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8933-108599"
},
"r25": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6801-107765"
},
"r250": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8933-108599"
},
"r251": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8933-108599"
},
"r252": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8933-108599"
},
"r253": {
"Name": "Accounting Standards Codification",
"Paragraph": "34",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e8981-108599"
},
"r254": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e9031-108599"
},
"r255": {
"Name": "Accounting Standards Codification",
"Paragraph": "41",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e9038-108599"
},
"r256": {
"Name": "Accounting Standards Codification",
"Paragraph": "41",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e9038-108599"
},
"r257": {
"Name": "Accounting Standards Codification",
"Paragraph": "41",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e9038-108599"
},
"r258": {
"Name": "Accounting Standards Codification",
"Paragraph": "42",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=120311839&loc=d3e9054-108599"
},
"r259": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "280",
"URI": "http://asc.fasb.org/topic&trid=2134510"
},
"r26": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6812-107765"
},
"r260": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=121593590&loc=d3e4647-111522"
},
"r261": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=121593590&loc=d3e4428-111522"
},
"r262": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=121593590&loc=d3e4531-111522"
},
"r263": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=121611835&loc=d3e5033-111524"
},
"r264": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 4.E)",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=27010918&loc=d3e74512-122707"
},
"r265": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=84159169&loc=d3e10133-111534"
},
"r266": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=84159169&loc=d3e10149-111534"
},
"r267": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=84159169&loc=d3e10178-111534"
},
"r268": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121553693&loc=d3e26610-111562"
},
"r269": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121553693&loc=d3e26853-111562"
},
"r27": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6812-107765"
},
"r270": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121553693&loc=d3e26626-111562"
},
"r271": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27161-111563"
},
"r272": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(aa)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27161-111563"
},
"r273": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27161-111563"
},
"r274": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27161-111563"
},
"r275": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27161-111563"
},
"r276": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27161-111563"
},
"r277": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27198-111563"
},
"r278": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27198-111563"
},
"r279": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27198-111563"
},
"r28": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6904-107765"
},
"r280": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27232-111563"
},
"r281": {
"Name": "Accounting Standards Codification",
"Paragraph": "5A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=SL120269820-111563"
},
"r282": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27290-111563"
},
"r283": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27337-111563"
},
"r284": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27340-111563"
},
"r285": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27357-111563"
},
"r286": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)-(d)",
"Topic": "320",
"URI": "http://asc.fasb.org/extlink&oid=121645371&loc=d3e27357-111563"
},
"r287": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "320",
"URI": "http://asc.fasb.org/topic&trid=2196928"
},
"r288": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "321",
"URI": "http://asc.fasb.org/extlink&oid=120269885&loc=SL75117546-209714"
},
"r289": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "323",
"URI": "http://asc.fasb.org/extlink&oid=109237563&loc=d3e33749-111570"
},
"r29": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6911-107765"
},
"r290": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "323",
"URI": "http://asc.fasb.org/extlink&oid=114001798&loc=d3e33918-111571"
},
"r291": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "323",
"URI": "http://asc.fasb.org/extlink&oid=114001798&loc=d3e33918-111571"
},
"r292": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(3)",
"Topic": "323",
"URI": "http://asc.fasb.org/extlink&oid=114001798&loc=d3e33918-111571"
},
"r293": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "323",
"URI": "http://asc.fasb.org/extlink&oid=114001798&loc=d3e33918-111571"
},
"r294": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121646688&loc=SL121648383-210437"
},
"r295": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121599337&loc=SL82919244-210447"
},
"r296": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121599337&loc=SL82919249-210447"
},
"r297": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121599337&loc=SL82919253-210447"
},
"r298": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121599337&loc=SL82919258-210447"
},
"r299": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121599337&loc=SL82919230-210447"
},
"r3": {
"Name": "Accounting Standards Codification",
"Paragraph": "3A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222160&loc=SL51721523-107759"
},
"r30": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e6935-107765"
},
"r300": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121558606&loc=SL82898722-210454"
},
"r301": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121582814&loc=SL82922888-210455"
},
"r302": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121582814&loc=SL82922895-210455"
},
"r303": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121582814&loc=SL82922900-210455"
},
"r304": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "30",
"Topic": "326",
"URI": "http://asc.fasb.org/extlink&oid=121590138&loc=SL82922954-210456"
},
"r305": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "330",
"URI": "http://asc.fasb.org/extlink&oid=116847112&loc=d3e4492-108314"
},
"r306": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "330",
"URI": "http://asc.fasb.org/extlink&oid=116847112&loc=d3e4542-108314"
},
"r307": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "330",
"URI": "http://asc.fasb.org/extlink&oid=116847112&loc=d3e4556-108314"
},
"r308": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.BB)",
"Topic": "330",
"URI": "http://asc.fasb.org/extlink&oid=27011343&loc=d3e100047-122729"
},
"r309": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "330",
"URI": "http://asc.fasb.org/topic&trid=2126998"
},
"r31": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=121566466&loc=d3e7018-107765"
},
"r310": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=99380562&loc=d3e13770-109266"
},
"r311": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=120320667&loc=SL49117168-202975"
},
"r312": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=121556970&loc=d3e13816-109267"
},
"r313": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=121556970&loc=d3e13816-109267"
},
"r314": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=121556970&loc=d3e13816-109267"
},
"r315": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=121556970&loc=d3e13816-109267"
},
"r316": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=121556970&loc=d3e13816-109267"
},
"r317": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=121556970&loc=d3e13816-109267"
},
"r318": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=121556970&loc=d3e13854-109267"
},
"r319": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=6388964&loc=d3e16212-109274"
},
"r32": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=6361739&loc=d3e7789-107766"
},
"r320": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=6388964&loc=d3e16225-109274"
},
"r321": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16265-109275"
},
"r322": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(1)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16265-109275"
},
"r323": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16265-109275"
},
"r324": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(d)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16265-109275"
},
"r325": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16265-109275"
},
"r326": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "((a)(1),(b))",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r327": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r328": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(1)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r329": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(2)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r33": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(1))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r330": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(3)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r331": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r332": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(d)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r333": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16373-109275"
},
"r334": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "30",
"SubTopic": "40",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=118172244&loc=d3e17916-109280"
},
"r335": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "50",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=16397303&loc=d3e19347-109286"
},
"r336": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "350",
"URI": "http://asc.fasb.org/topic&trid=2144416"
},
"r337": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "05",
"SubTopic": "10",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226317&loc=d3e202-110218"
},
"r338": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226348&loc=d3e2443-110228"
},
"r339": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r34": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(13))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r340": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r341": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r342": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r343": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226691&loc=d3e2941-110230"
},
"r344": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226691&loc=d3e2941-110230"
},
"r345": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226691&loc=d3e2941-110230"
},
"r346": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226691&loc=d3e2941-110230"
},
"r347": {
"Name": "Accounting Standards Codification",
"Paragraph": "3A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226691&loc=SL51724579-110230"
},
"r348": {
"Name": "Accounting Standards Codification",
"Paragraph": "3A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226691&loc=SL51724579-110230"
},
"r349": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.CC)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=27011434&loc=d3e125687-122742"
},
"r35": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(14))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r350": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "360",
"URI": "http://asc.fasb.org/topic&trid=2155823"
},
"r351": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "420",
"URI": "http://asc.fasb.org/extlink&oid=6394359&loc=d3e17939-110869"
},
"r352": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.P.3)",
"Topic": "420",
"URI": "http://asc.fasb.org/extlink&oid=115931487&loc=d3e140864-122747"
},
"r353": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.P.4(b))",
"Topic": "420",
"URI": "http://asc.fasb.org/extlink&oid=115931487&loc=d3e140904-122747"
},
"r354": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "440",
"URI": "http://asc.fasb.org/extlink&oid=121559207&loc=d3e25336-109308"
},
"r355": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "440",
"URI": "http://asc.fasb.org/extlink&oid=121559207&loc=d3e25336-109308"
},
"r356": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "440",
"URI": "http://asc.fasb.org/topic&trid=2144648"
},
"r357": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "450",
"URI": "http://asc.fasb.org/extlink&oid=121557415&loc=d3e14326-108349"
},
"r358": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "450",
"URI": "http://asc.fasb.org/extlink&oid=121557415&loc=d3e14615-108349"
},
"r359": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "450",
"URI": "http://asc.fasb.org/extlink&oid=121557415&loc=d3e14435-108349"
},
"r36": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(19))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r360": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "450",
"URI": "http://asc.fasb.org/extlink&oid=121557415&loc=d3e14557-108349"
},
"r361": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "450",
"URI": "http://asc.fasb.org/topic&trid=2127136"
},
"r362": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "460",
"URI": "http://asc.fasb.org/extlink&oid=68068213&loc=d3e12565-110249"
},
"r363": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "460",
"URI": "http://asc.fasb.org/topic&trid=2155896"
},
"r364": {
"Name": "Accounting Standards Codification",
"Paragraph": "12A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=99376301&loc=SL5988623-112600"
},
"r365": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=6802200&loc=d3e1835-112601"
},
"r366": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=6802200&loc=SL6230698-112601"
},
"r367": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=120520924&loc=SL6031897-161870"
},
"r368": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "470",
"URI": "http://asc.fasb.org/topic&trid=2208564"
},
"r369": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(CFRR 211.02)",
"Topic": "480",
"URI": "http://asc.fasb.org/extlink&oid=65877616&loc=d3e177068-122764"
},
"r37": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(20))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r370": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=65888546&loc=d3e21300-112643"
},
"r371": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21553-112644"
},
"r372": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21463-112644"
},
"r373": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21475-112644"
},
"r374": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21484-112644"
},
"r375": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21488-112644"
},
"r376": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21506-112644"
},
"r377": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21521-112644"
},
"r378": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=109259400&loc=d3e21538-112644"
},
"r379": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.3-04)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=120397183&loc=d3e187085-122770"
},
"r38": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(22))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r380": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=120397183&loc=d3e187085-122770"
},
"r381": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=6405813&loc=d3e23239-112655"
},
"r382": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=6405834&loc=d3e23315-112656"
},
"r383": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "505",
"URI": "http://asc.fasb.org/topic&trid=2208762"
},
"r384": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130551-203045"
},
"r385": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130554-203045"
},
"r386": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130554-203045"
},
"r387": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130554-203045"
},
"r388": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130554-203045"
},
"r389": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130554-203045"
},
"r39": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(24))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r390": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130556-203045"
},
"r391": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130556-203045"
},
"r392": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130558-203045"
},
"r393": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130545-203045"
},
"r394": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121604090&loc=SL49130550-203045"
},
"r395": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121551570&loc=SL49130690-203046-203046"
},
"r396": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121551570&loc=SL49130690-203046-203046"
},
"r397": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121551570&loc=SL49130690-203046-203046"
},
"r398": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121551570&loc=SL49130690-203046-203046"
},
"r399": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121551570&loc=SL49130690-203046-203046"
},
"r4": {
"Name": "Accounting Standards Codification",
"Paragraph": "3B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222160&loc=SL51721525-107759"
},
"r40": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(25))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r400": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121551570&loc=SL49130690-203046-203046"
},
"r401": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=121551570&loc=SL49130690-203046-203046"
},
"r402": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "606",
"URI": "http://asc.fasb.org/topic&trid=49130388"
},
"r403": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "15",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "712",
"URI": "http://asc.fasb.org/extlink&oid=6410066&loc=d3e79218-111664"
},
"r404": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "15",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "712",
"URI": "http://asc.fasb.org/extlink&oid=6410066&loc=d3e79218-111664"
},
"r405": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=109237824&loc=d3e1703-114919"
},
"r406": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r407": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(1)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r408": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(10)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r409": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(2)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r41": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(27))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r410": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(3)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r411": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(4)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r412": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(5)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r413": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(6)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r414": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(7)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r415": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(8)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r416": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(9)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r417": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r418": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r419": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r42": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(28))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r420": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r421": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(4)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r422": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(5)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r423": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(6)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r424": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(7)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r425": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(8)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r426": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r427": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(i)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r428": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(ii)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r429": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iii)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r43": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(29))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r430": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(01)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r431": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r432": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)(A)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r433": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)(B)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r434": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)(C)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r435": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(03)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r436": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(e)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r437": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(f)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r438": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r439": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r44": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(3))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r440": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)(1)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r441": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)(2)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r442": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)(3)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r443": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)(4)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r444": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)(5)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r445": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)(6)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r446": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)(7)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r447": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(i)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r448": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(j)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r449": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(k)(1)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r45": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(1))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r450": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(k)(2)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r451": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(k)(3)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r452": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(k)(4)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r453": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(l)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r454": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(n)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r455": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(o)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r456": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(p)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r457": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(q)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r458": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(r)(1)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r459": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(r)(2)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e1928-114920"
},
"r46": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(3))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r460": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2410-114920"
},
"r461": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2417-114920"
},
"r462": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2417-114920"
},
"r463": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2439-114920"
},
"r464": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r465": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(1)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r466": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(2)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r467": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(3)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r468": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(4)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r469": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(5)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r47": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(4))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r470": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(6)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r471": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(7)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r472": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2709-114920"
},
"r473": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118255775&loc=d3e2919-114920"
},
"r474": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118257860&loc=d3e4179-114921"
},
"r475": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=118257860&loc=d3e4587-114921"
},
"r476": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=21916913&loc=d3e273930-122802"
},
"r477": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "60",
"Subparagraph": "(c)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=6414203&loc=d3e39689-114964"
},
"r478": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "70",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=49170846&loc=d3e28014-114942"
},
"r479": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "80",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=29639808&loc=d3e29008-114946"
},
"r48": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r480": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450702-114947"
},
"r481": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(b)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450702-114947"
},
"r482": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(c)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450702-114947"
},
"r483": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450702-114947"
},
"r484": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(d)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450657-114947"
},
"r485": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(f)(3)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450657-114947"
},
"r486": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450673-114947"
},
"r487": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(b)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450673-114947"
},
"r488": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(c)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450673-114947"
},
"r489": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(b)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450691-114947"
},
"r49": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(31))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r490": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "80",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=35742348&loc=SL14450788-114948"
},
"r491": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "715",
"URI": "http://asc.fasb.org/topic&trid=2235017"
},
"r492": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5047-113901"
},
"r493": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5047-113901"
},
"r494": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5047-113901"
},
"r495": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r496": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r497": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(3)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r498": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a),(g)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r499": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b),(f(1))",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r5": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222160&loc=d3e1012-107759"
},
"r50": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(32))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r500": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r501": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(i)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r502": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(i)-(ii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r503": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r504": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r505": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(3)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r506": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(i)-(ii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r507": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r508": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r509": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(3)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r51": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(6)(a)(1))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r510": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r511": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r512": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r513": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(ii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r514": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(iii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r515": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r516": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r517": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(1)(i)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r518": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(1)(ii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r519": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(i)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r52": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(6)(a)(3))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r520": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(k)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r521": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r522": {
"Name": "Accounting Standards Codification",
"Paragraph": "2A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=SL79508275-113901"
},
"r523": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r524": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(f)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r525": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(f)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r526": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(g)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r527": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 14.F)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=115993241&loc=d3e301413-122809"
},
"r528": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=109244661&loc=d3e17540-113929"
},
"r529": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "718",
"URI": "http://asc.fasb.org/topic&trid=2228938"
},
"r53": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(6)(a)(4))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r530": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "35",
"Subparagraph": "(a)",
"Topic": "720",
"URI": "http://asc.fasb.org/extlink&oid=6420018&loc=d3e36677-107848"
},
"r531": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "35",
"Subparagraph": "(b)",
"Topic": "720",
"URI": "http://asc.fasb.org/extlink&oid=6420018&loc=d3e36677-107848"
},
"r532": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "730",
"URI": "http://asc.fasb.org/extlink&oid=6420194&loc=d3e21568-108373"
},
"r533": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "730",
"URI": "http://asc.fasb.org/extlink&oid=6420387&loc=d3e23199-108380"
},
"r534": {
"Name": "Accounting Standards Codification",
"Paragraph": "10B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=120406818&loc=SL37586934-109318"
},
"r535": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=120406818&loc=d3e32247-109318"
},
"r536": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=120406818&loc=d3e32280-109318"
},
"r537": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=120406818&loc=d3e31917-109318"
},
"r538": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=120406818&loc=d3e31931-109318"
},
"r539": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32672-109319"
},
"r54": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(6)(a))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r540": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32687-109319"
},
"r541": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32705-109319"
},
"r542": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32718-109319"
},
"r543": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)(3)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32718-109319"
},
"r544": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32718-109319"
},
"r545": {
"Name": "Accounting Standards Codification",
"Paragraph": "15A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=SL6600010-109319"
},
"r546": {
"Name": "Accounting Standards Codification",
"Paragraph": "15A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=SL6600010-109319"
},
"r547": {
"Name": "Accounting Standards Codification",
"Paragraph": "15A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=SL6600010-109319"
},
"r548": {
"Name": "Accounting Standards Codification",
"Paragraph": "15A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(3)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=SL6600010-109319"
},
"r549": {
"Name": "Accounting Standards Codification",
"Paragraph": "15A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(4)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=SL6600010-109319"
},
"r55": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(6)(b))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r550": {
"Name": "Accounting Standards Codification",
"Paragraph": "15A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=SL6600010-109319"
},
"r551": {
"Name": "Accounting Standards Codification",
"Paragraph": "15A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=SL6600010-109319"
},
"r552": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32809-109319"
},
"r553": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32840-109319"
},
"r554": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32537-109319"
},
"r555": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32537-109319"
},
"r556": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32847-109319"
},
"r557": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32857-109319"
},
"r558": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32559-109319"
},
"r559": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32559-109319"
},
"r56": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(6)(c))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r560": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32621-109319"
},
"r561": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32632-109319"
},
"r562": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32639-109319"
},
"r563": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32639-109319"
},
"r564": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32639-109319"
},
"r565": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=84230637&loc=d3e32639-109319"
},
"r566": {
"Name": "Accounting Standards Codification",
"Paragraph": "217",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121610041&loc=d3e36027-109320"
},
"r567": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)(2)",
"Topic": "740"
},
"r568": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)(3)",
"Topic": "740"
},
"r569": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB TOPIC 6.I.5.Q1)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330036-122817"
},
"r57": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.1)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r570": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB TOPIC 6.I.7)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330036-122817"
},
"r571": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.7)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330036-122817"
},
"r572": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.Fact.1)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330036-122817"
},
"r573": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.Fact.2)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330036-122817"
},
"r574": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.Fact.3)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330036-122817"
},
"r575": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.Fact.4)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330036-122817"
},
"r576": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 11.C)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=116825942&loc=d3e330215-122817"
},
"r577": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=120385591&loc=d3e38679-109324"
},
"r578": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=120385591&loc=d3e38679-109324"
},
"r579": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "270",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=6424409&loc=d3e44925-109338"
},
"r58": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.12)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r580": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=6424122&loc=d3e41874-109331"
},
"r581": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "740",
"URI": "http://asc.fasb.org/topic&trid=2144680"
},
"r582": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "05",
"SubTopic": "10",
"Subparagraph": "(a)-(d)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=6909625&loc=d3e227-128457"
},
"r583": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=79982066&loc=d3e1392-128463"
},
"r584": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)(3)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=79982066&loc=d3e1392-128463"
},
"r585": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=79982066&loc=d3e1392-128463"
},
"r586": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=79982066&loc=d3e1486-128463"
},
"r587": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=121647850&loc=d3e4845-128472"
},
"r588": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=121598580&loc=d3e5263-128473"
},
"r589": {
"Name": "Accounting Standards Codification",
"Paragraph": "38",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=121598580&loc=d3e5504-128473"
},
"r59": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.13(a))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r590": {
"Name": "Accounting Standards Codification",
"Paragraph": "44",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=121598580&loc=d3e5558-128473"
},
"r591": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "30",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=6911189&loc=d3e6408-128476"
},
"r592": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=116859824&loc=d3e6819-128478"
},
"r593": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "30",
"Subparagraph": "b",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=116859824&loc=d3e6819-128478"
},
"r594": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(b)(1)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e6927-128479"
},
"r595": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(c)(1)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e6927-128479"
},
"r596": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(c)(3)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e6927-128479"
},
"r597": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "c",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e6927-128479"
},
"r598": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(1)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e7008-128479"
},
"r599": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "740",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=121554379&loc=d3e9972-128506"
},
"r6": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=d3e1361-107760"
},
"r60": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.13)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r600": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "740",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=121554379&loc=d3e9979-128506"
},
"r601": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "805",
"URI": "http://asc.fasb.org/topic&trid=2303972"
},
"r602": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434"
},
"r603": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434"
},
"r604": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434"
},
"r605": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "808",
"URI": "http://asc.fasb.org/topic&trid=5833765"
},
"r606": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=108774443&loc=SL4568447-111683"
},
"r607": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=108774443&loc=SL4568740-111683"
},
"r608": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=108774443&loc=SL4569616-111683"
},
"r609": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=108774443&loc=SL4569643-111683"
},
"r61": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.14)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r610": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=108774443&loc=SL4613674-111683"
},
"r611": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=116870748&loc=SL6758485-165988"
},
"r612": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=116870748&loc=SL6758485-165988"
},
"r613": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=d3e5614-111684"
},
"r614": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684"
},
"r615": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684"
},
"r616": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684"
},
"r617": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(3)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684"
},
"r618": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c),(3)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684"
},
"r619": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bb)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=121559654&loc=d3e5710-111685"
},
"r62": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.15)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r620": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=121559654&loc=d3e5710-111685"
},
"r621": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=121559654&loc=d3e5710-111685"
},
"r622": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=121559654&loc=d3e5728-111685"
},
"r623": {
"Name": "Accounting Standards Codification",
"Paragraph": "5A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=121559654&loc=SL6759159-111685"
},
"r624": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=121559654&loc=d3e5747-111685"
},
"r625": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=121559654&loc=SL6228884-111685"
},
"r626": {
"Name": "Accounting Standards Codification",
"Paragraph": "4I",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=120409616&loc=SL4590271-111686"
},
"r627": {
"Name": "Accounting Standards Codification",
"Paragraph": "4J",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=120409616&loc=SL4591551-111686"
},
"r628": {
"Name": "Accounting Standards Codification",
"Paragraph": "4K",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=120409616&loc=SL4591552-111686"
},
"r629": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "810",
"URI": "http://asc.fasb.org/topic&trid=2197479"
},
"r63": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.17)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r630": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5579240-113959"
},
"r631": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5579245-113959"
},
"r632": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5579245-113959"
},
"r633": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5580258-113959"
},
"r634": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)(i)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=d3e41620-113959"
},
"r635": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)(ii)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=d3e41620-113959"
},
"r636": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)(iii)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=d3e41620-113959"
},
"r637": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=d3e41620-113959"
},
"r638": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=d3e41638-113959"
},
"r639": {
"Name": "Accounting Standards Codification",
"Paragraph": "4A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5618551-113959"
},
"r64": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19(a))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r640": {
"Name": "Accounting Standards Codification",
"Paragraph": "4A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5618551-113959"
},
"r641": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a),(c)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624163-113959"
},
"r642": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624163-113959"
},
"r643": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624163-113959"
},
"r644": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624163-113959"
},
"r645": {
"Name": "Accounting Standards Codification",
"Paragraph": "4C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bb)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624171-113959"
},
"r646": {
"Name": "Accounting Standards Codification",
"Paragraph": "4C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624171-113959"
},
"r647": {
"Name": "Accounting Standards Codification",
"Paragraph": "4C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624171-113959"
},
"r648": {
"Name": "Accounting Standards Codification",
"Paragraph": "4C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624171-113959"
},
"r649": {
"Name": "Accounting Standards Codification",
"Paragraph": "4CCC",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL109998896-113959"
},
"r65": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19(b),22(b))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r650": {
"Name": "Accounting Standards Codification",
"Paragraph": "4CCC",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL109998896-113959"
},
"r651": {
"Name": "Accounting Standards Codification",
"Paragraph": "4D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624177-113959"
},
"r652": {
"Name": "Accounting Standards Codification",
"Paragraph": "4D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624177-113959"
},
"r653": {
"Name": "Accounting Standards Codification",
"Paragraph": "4D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624177-113959"
},
"r654": {
"Name": "Accounting Standards Codification",
"Paragraph": "4D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624177-113959"
},
"r655": {
"Name": "Accounting Standards Codification",
"Paragraph": "4E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=SL5624181-113959"
},
"r656": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121590274&loc=d3e41675-113959"
},
"r657": {
"Name": "Accounting Standards Codification",
"Paragraph": "182",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121582272&loc=SL5629052-113961"
},
"r658": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121577181&loc=SL110061190-113977"
},
"r659": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "25",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121577467&loc=d3e76258-113986"
},
"r66": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19,20)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r660": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121549185&loc=d3e80784-113994"
},
"r661": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(d)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=121549185&loc=d3e80784-113994"
},
"r662": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "815",
"URI": "http://asc.fasb.org/topic&trid=2229140"
},
"r663": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r664": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r665": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bb)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r666": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r667": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)(1)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r668": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)(2)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r669": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r67": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19,20,22)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r670": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r671": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r672": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r673": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19207-110258"
},
"r674": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=d3e19279-110258"
},
"r675": {
"Name": "Accounting Standards Codification",
"Paragraph": "6A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=117815213&loc=SL6742756-110258"
},
"r676": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "60",
"SubTopic": "10",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=7493716&loc=d3e21868-110260"
},
"r677": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=99377092&loc=SL75136599-209740"
},
"r678": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=121572278&loc=d3e13279-108611"
},
"r679": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=121572278&loc=d3e13467-108611"
},
"r68": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19-26)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r680": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=75031198&loc=d3e14064-108612"
},
"r681": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=120253306&loc=d3e28228-110885"
},
"r682": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "20",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=121605123&loc=d3e30226-110892"
},
"r683": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=109240200&loc=d3e30690-110894"
},
"r684": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=6450222&loc=d3e30840-110895"
},
"r685": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "230",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=98513438&loc=d3e33268-110906"
},
"r686": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32136-110900"
},
"r687": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r688": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r689": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(c)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r69": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.20)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r690": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(d)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r691": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r692": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=6450520&loc=d3e32583-110901"
},
"r693": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "830",
"URI": "http://asc.fasb.org/topic&trid=2175825"
},
"r694": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=6450988&loc=d3e26243-108391"
},
"r695": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=6450988&loc=d3e26243-108391"
},
"r696": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=119993939&loc=d3e28541-108399"
},
"r697": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=119993939&loc=d3e28551-108399"
},
"r698": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=119993939&loc=d3e28555-108399"
},
"r699": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=114775985&loc=d3e28878-108400"
},
"r7": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=d3e1361-107760"
},
"r70": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.21)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r700": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121603541&loc=SL77918627-209977"
},
"r701": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121603541&loc=SL77918627-209977"
},
"r702": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121603541&loc=SL77918631-209977"
},
"r703": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121603541&loc=SL77918643-209977"
},
"r704": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918666-209980"
},
"r705": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(3)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918673-209980"
},
"r706": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918673-209980"
},
"r707": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r708": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r709": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(e)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r71": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.22(a)(1))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r710": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(1)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r711": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(2)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r712": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(3)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r713": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(4)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r714": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918686-209980"
},
"r715": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121609121&loc=SL77918701-209980"
},
"r716": {
"Name": "Accounting Standards Codification",
"Paragraph": "53",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=121568110&loc=SL77918982-209971"
},
"r717": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"SubTopic": "20",
"Topic": "842",
"URI": "http://asc.fasb.org/subtopic&trid=77888251"
},
"r718": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864"
},
"r719": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39599-107864"
},
"r72": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.22)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r720": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "855",
"URI": "http://asc.fasb.org/extlink&oid=6842918&loc=SL6314020-165662"
},
"r721": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)(i)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r722": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)(ii)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r723": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r724": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r725": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r726": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r727": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r728": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r729": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r73": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.24)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r730": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r731": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r732": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r733": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r734": {
"Name": "Accounting Standards Codification",
"Paragraph": "4D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=SL51823488-111719"
},
"r735": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=66007379&loc=d3e113888-111728"
},
"r736": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=109249958&loc=SL34722452-111729"
},
"r737": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122625-111746"
},
"r738": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122625-111746"
},
"r739": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122625-111746"
},
"r74": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.25)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r740": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(4)(i)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122625-111746"
},
"r741": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r742": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r743": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r744": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(4)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r745": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(5)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r746": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(6)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r747": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(7)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r748": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(b)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r749": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(e)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r75": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.28,29)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r750": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(e)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r751": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(e)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=116651436&loc=d3e122739-111746"
},
"r752": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "910",
"URI": "http://asc.fasb.org/extlink&oid=119991564&loc=SL119991595-234733"
},
"r753": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "275",
"Topic": "912",
"URI": "http://asc.fasb.org/extlink&oid=84167019&loc=d3e54681-109401"
},
"r754": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 11.L)",
"Topic": "924",
"URI": "http://asc.fasb.org/extlink&oid=6472922&loc=d3e499488-122856"
},
"r755": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "926",
"URI": "http://asc.fasb.org/extlink&oid=120154696&loc=d3e54445-107959"
},
"r756": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "340",
"Topic": "928",
"URI": "http://asc.fasb.org/extlink&oid=6473545&loc=d3e61844-108004"
},
"r757": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(10)(1))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r758": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(11))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r759": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(13))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r76": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29,30)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r760": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(15)(1))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r761": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(16))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r762": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(17))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r763": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(22))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r764": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(23))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r765": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(6))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r766": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.10(3))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r767": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.15(3),(4))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r768": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.15(5))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r769": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.17)",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r77": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29-31)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r770": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(13)(f))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r771": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(22))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r772": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(23))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r773": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(24))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r774": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(25))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r775": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(26))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r776": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(27))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r777": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04.9)",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r778": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "235",
"Subparagraph": "(SX 210.9-05(b)(2))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399901&loc=d3e537907-122884"
},
"r779": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Subparagraph": "(b)",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120252992&loc=d3e62557-112803"
},
"r78": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.31)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r780": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120252992&loc=d3e62557-112803"
},
"r781": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120252992&loc=d3e62586-112803"
},
"r782": {
"Name": "Accounting Standards Codification",
"Paragraph": "3A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Subparagraph": "(a)",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120252992&loc=SL120269850-112803"
},
"r783": {
"Name": "Accounting Standards Codification",
"Paragraph": "3A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Subparagraph": "(b)",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120252992&loc=SL120269850-112803"
},
"r784": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "470",
"Subparagraph": "e",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=75038535&loc=d3e64711-112823"
},
"r785": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "470",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=75038535&loc=d3e64711-112823"
},
"r786": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "825",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=108315417&loc=d3e61044-112788"
},
"r787": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(16))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r788": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(12))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r789": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(15)(b)(1))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r79": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.6(a)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r790": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(16))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r791": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(19))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r792": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(23)(a)(3))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r793": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(23)(a)(4))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r794": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(24))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r795": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(25))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r796": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(8))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r797": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.(a),19)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r798": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.15(a))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r799": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.17)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r8": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=d3e1474-107760"
},
"r80": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.6(a))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r800": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.3)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r801": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(10))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r802": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(18))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r803": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(19))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r804": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(20))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r805": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(21))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r806": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(22))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r807": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(23))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r808": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(3)(b))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r809": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(3)(d))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r81": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.6(b))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r810": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(9))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r811": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04.3(a))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r812": {
"Name": "Accounting Standards Codification",
"Paragraph": "4H",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=116884468&loc=SL65671331-158438"
},
"r813": {
"Name": "Accounting Standards Codification",
"Paragraph": "7A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Subparagraph": "(d)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121643868&loc=SL117782755-158439"
},
"r814": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(a)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121639165&loc=SL117783719-158441"
},
"r815": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(b)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121639165&loc=SL117783719-158441"
},
"r816": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(c)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121639165&loc=SL117783719-158441"
},
"r817": {
"Name": "Accounting Standards Codification",
"Paragraph": "29F",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121639165&loc=SL117819544-158441"
},
"r818": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121370832&loc=SL117420844-207641"
},
"r819": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(f)(1)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121370832&loc=SL117420844-207641"
},
"r82": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.8)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r820": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(f)(2)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121370832&loc=SL117420844-207641"
},
"r821": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(g)(2)(i)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121370832&loc=SL117420844-207641"
},
"r822": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(g)(2)(ii)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121370832&loc=SL117420844-207641"
},
"r823": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(h)(2)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121370832&loc=SL117420844-207641"
},
"r824": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(e)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=121641442&loc=d3e19393-158473"
},
"r825": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column B)(Footnote 1))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r826": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column B)(Footnote 6))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r827": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column B)(Footnote 7))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r828": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column C)(Footnote 1))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r829": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column C)(Footnote 6))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r83": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.9)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r830": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column C)(Footnote 7))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r831": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column D)(Footnote 1))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r832": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column D)(Footnote 6))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r833": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-12B(Column D)(Footnote 7))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611197-123010"
},
"r834": {
"Name": "Accounting Standards Codification",
"Paragraph": "5D",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-13D(Column B)(Footnote 2))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=SL120429264-123010"
},
"r835": {
"Name": "Accounting Standards Codification",
"Paragraph": "5D",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-13D(Column C)(Footnote 2))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=SL120429264-123010"
},
"r836": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-15(Column A))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611379-123010"
},
"r837": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-15(Column B))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611379-123010"
},
"r838": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-15(Column C))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611379-123010"
},
"r839": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "320",
"Subparagraph": "(SX 210.12-15(Column D))",
"Topic": "946",
"URI": "http://asc.fasb.org/extlink&oid=120401592&loc=d3e611379-123010"
},
"r84": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=51824906&loc=SL20225862-175312"
},
"r840": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "310",
"Subparagraph": "(SX 210.12-29(Footnote 4))",
"Topic": "948",
"URI": "http://asc.fasb.org/extlink&oid=120402547&loc=d3e617274-123014"
},
"r841": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "450",
"Topic": "954",
"URI": "http://asc.fasb.org/extlink&oid=6491354&loc=d3e6049-115624"
},
"r842": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "450",
"Topic": "954",
"URI": "http://asc.fasb.org/extlink&oid=6491354&loc=d3e6052-115624"
},
"r843": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "740",
"Topic": "954",
"URI": "http://asc.fasb.org/extlink&oid=6491622&loc=d3e9504-115650"
},
"r844": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "360",
"Subparagraph": "(d)",
"Topic": "958",
"URI": "http://asc.fasb.org/extlink&oid=120429125&loc=d3e99779-112916"
},
"r845": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "360",
"Topic": "958",
"URI": "http://asc.fasb.org/extlink&oid=120429125&loc=d3e99893-112916"
},
"r846": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "360",
"Topic": "958",
"URI": "http://asc.fasb.org/extlink&oid=120429125&loc=SL120174063-112916"
},
"r847": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column B))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r848": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column C))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r849": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column D))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r85": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=51824906&loc=SL20225862-175312"
},
"r850": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column E))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r851": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column F))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r852": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column G))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r853": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column H))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r854": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column I))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r855": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Footnote 2))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r856": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Footnote 4))",
"Topic": "970",
"URI": "http://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r857": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "340",
"Topic": "980",
"URI": "http://asc.fasb.org/extlink&oid=121557228&loc=d3e43603-110378"
},
"r858": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "405",
"Topic": "980",
"URI": "http://asc.fasb.org/extlink&oid=6500807&loc=d3e48068-110394"
},
"r859": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "740",
"Topic": "980",
"URI": "http://asc.fasb.org/extlink&oid=6501382&loc=d3e54053-110423"
},
"r86": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=99393222&loc=SL20226000-175313"
},
"r860": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "740",
"Topic": "980",
"URI": "http://asc.fasb.org/extlink&oid=6501382&loc=d3e54136-110423"
},
"r861": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "985",
"URI": "http://asc.fasb.org/extlink&oid=6501960&loc=d3e128462-111756"
},
"r862": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b"
},
"r863": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-23"
},
"r864": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "d1-1"
},
"r865": {
"Name": "Form 10-K",
"Number": "249",
"Publisher": "SEC",
"Section": "310"
},
"r866": {
"Name": "Form 20-F",
"Number": "249",
"Publisher": "SEC",
"Section": "220",
"Subsection": "f"
},
"r867": {
"Name": "Form 40-F",
"Number": "249",
"Publisher": "SEC",
"Section": "240",
"Subsection": "f"
},
"r868": {
"Name": "Forms 10-K, 10-Q, 20-F",
"Number": "240",
"Publisher": "SEC",
"Section": "13",
"Subsection": "a-1"
},
"r869": {
"Name": "Regulation 12B",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-2"
},
"r87": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r870": {
"Name": "Regulation S-T",
"Number": "232",
"Publisher": "SEC",
"Section": "405"
},
"r88": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a),(b),(c)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r89": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(dd)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r9": {
"Name": "Accounting Standards Codification",
"Paragraph": "3A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721659-107760"
},
"r90": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r91": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r92": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(h)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r93": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(i),(j),(k)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r94": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(i-k)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r95": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669646-108580"
},
"r96": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=d3e637-108580"
},
"r97": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=d3e681-108580"
},
"r98": {
"Name": "Accounting Standards Codification",
"Paragraph": "14A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=SL7669686-108580"
},
"r99": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=121641772&loc=d3e689-108580"
}
},
"version": "2.1"
}