
Building a high-performing, growth-focused, sterile injectable CDMO March 2026

2 Fiscal Year Change On August 1, 2025, our Board of Directors approved a change in the Company’s fiscal year that ended on the last Sunday of May to a fiscal year that corresponds with the calendar year, ending on December 31st, effective for the fiscal period beginning May 26, 2025, and ending December 31, 2025 (the “Fiscal Year Change”). The Fiscal Year Change is applied on a prospective basis and does not adjust operating results for prior periods. References in this presentation to “FY 2025” refer to our prior fiscal year ending on May 25, 2025, and references in this presentation to “transition period” refer to the approximately seven-month period from May 26, 2025, through December 31, 2025. For more information regarding the Fiscal Year Change and results for this period, please refer to our filings with the with the Securities and Exchange Commission (“SEC”) , including, but not limited to, the Annual Report on Form 10-KT for the transition period ended December 31, 2025, available on our website at www.lifecore.com and at www.sec.gov.

3 Important Information Regarding Forward-Looking Statements This presentation contains forward-looking statements regarding future events and our future results that are subject to the safe harbor created under the Private Securities Litigation Reform Act of 1995 and other safe harbors under the Securities Act of 1933 and the Securities Exchange Act of 1934. Words such as “anticipate”, “estimate”, “expect”, “project”, “aim,” “designed to,” “plan”, “intend”, “believe”, “may”, “might”, “will”, “should”, “can have”, “likely” and similar expressions are used to identify forward-looking statements. In addition, all statements regarding our future financial and operating performance and strategy, including our goals of achieving a 12+% revenue CAGR and increasing Adjusted EBITDA margins to more than 25% in the mid-term; positioning of the Company for sustained, long-term growth; key initiatives that are expected to continue to drive margin improvement; expected benefits of our new ERP system; financial guidance for 2026 and longer-term outlook; three-pronged strategy for growth comprised of maximizing our existing customer business, advancing programs currently within our late-stage development pipeline towards commercialization, and winning impactful new business that will continue to fill our project pipeline; anticipated revenue growth and improved capacity utilization; the future diversification of our customer base and reduction of dependency on any one customer; visibility and nature of leading revenue indicators; a medical device program expected to contribute >50% of projected commercial pipeline revenue by 2030; launch timelines from our late-stage development portfolio; continued efficiency and cost containment discipline; significant inflection point in existing commercial customer demand beginning in 2027; and use of cash resources or need to raise additional financing in 2026 or in the near-term, are forward-looking statements. All forward- looking statements involve certain risks and uncertainties that could cause actual results to differ materially, including such factors as, among others, the timing and amount of future expenses, revenue, net income (loss), Adjusted EBITDA, cash flow and capital requirements, and timing and availability of and the need for additional financing; our ability to maintain or expand our relationships with our current customers, including the impact of changes in consumer demand for the products we manufacture for our customers; our ability to grow and diversify our business with new customers, including the potential loss of development customers if they do not receive required funding or regulatory approvals or for other reasons; our ability to comply with covenants under our credit agreements and to pay required interest and principal payments when due; our ability to fund any redemptions of shares of the outstanding Series A Convertible Preferred Stock if requested by holders in accordance with their terms; our ability to raise additional capital for ongoing needs, including through equity financing, debt financing, collaborations, strategic alliances or licensing arrangements; the impact of macroeconomic events or circumstances on our operations and financial performance, including inflation, tariffs, interest rates, social unrest and global instability; the performance of our third-party suppliers; pharmaceutical industry market forces that may impact our customers’ success and continued demand for the products we produce for those customers; our ability to recruit or retain key scientific, technical, business development, and management personnel and our executive officers; our ability to comply with stringent U.S. and foreign government regulation in the manufacture of pharmaceutical products, including current Good Manufacturing Practice, or cGMP; the outcome and cost of existing and any new litigation or regulatory proceedings; and other risk factors set forth from time to time in the company’s filings with the SEC, including, but not limited to, the Annual Report on Form 10-KT for the transition period ended December 31, 2025 (the “December 2025 10-KT”). For additional information about factors that could cause actual results to differ materially from those described in the forward-looking statements, please refer to our filings with the SEC, including the risk factors contained in the December 2025 10-KT. Forward-looking statements represent management’s current expectations as of the date hereof and are inherently uncertain. Except as required by law, we do not undertake any obligation to update forward-looking statements made by us to reflect subsequent events or circumstances.

4 Non-GAAP Financial Measures This presentation contains non-GAAP financial information, including Adjusted EBITDA and free cash flow. The Company has included a reconciliation of Adjusted EBITDA to net (loss) income and operating cash flows to free cash flow, the most directly comparable financial measures calculated in accordance with GAAP. We define Adjusted EBITDA as net (loss) income as determined under GAAP excluding (i) interest expense, net of interest income, (ii) income tax expense (benefit), (iii) depreciation and amortization, (iv) stock-based compensation, (v) change in fair value of debt derivatives, (vi) financing fees (non-interest), (vii) loss on sale or disposal of assets, (viii) reorganization costs, (ix) restructuring (recoveries) costs, (x) franchise tax equivalent to income tax, (xi) contract cancellation costs, (xii) loss (income) from discontinued operations, (xiii) stockholder activist settlement costs, and (xiv) start-up costs. Free cash flow reduces operating cash flows by capital expenditures. See slide entitled “Reconciliation of Non-GAAP Financial Measures” in this presentation for the company’s definition of Adjusted EBITDA and free cash flow for the fiscal year ended May 25, 2025, and for the 2025 transition period (from May 26, 2025, to December 31, 2025) and reconciliations thereof to net (loss) income and operating cash flows, respectively, for each such period. The company has disclosed these non-GAAP financial measures to supplement its consolidated financial statements presented in accordance with GAAP. These non-GAAP financial measures exclude/include certain items that are included in the company’s results reported in accordance with GAAP because we believe they are not reflective of our core operations or indicative of our ongoing operations. Management believes these non-GAAP financial measures provide useful additional information to investors about trends in the company’s operations and are useful for period-over-period comparisons. Management uses Adjusted EBITDA and free cash flow, in addition to GAAP financial measures, to monitor trends in the company’s operations, understand and compare operating results across accounting periods, for financial and operational decision making, for planning and forecasting purposes, and with respect to Adjusted EBITDA as a measure of performance for compensation decisions. These non-GAAP financial measures should not be considered in isolation or as a substitute for the comparable GAAP measures. In addition, these non-GAAP financial measures may not be the same as similar measures provided by other companies due to the potential differences in methods of calculation and items being excluded/included. These non- GAAP financial measures should be read in conjunction with the company’s consolidated financial statements presented in accordance with GAAP.

5 Lifecore at a Glance 400 Employees Inclusive, Performance- Driven Culture Fully integrated CDMO offering development and fill/finish of sterile injectable pharmaceuticals Leader in Sodium Hyaluronate (HA) Global Regulatory Capabilities Founded in 1965 • Non-GAAP Measure. See disclaimers on slides 2, 3 & 4, and “Reconciliation of Non-GAAP Financial Measures” slide ** The estimate was based on historical fiscal year 2025 revenues, projected development pipeline, and new business pricing, volume and other assumptions Approx. 248,000 Sq. Ft. Facility $300M Annual Production Capacity** 20+ Commercial Products 2026 Financial Guidance and Business Profile $120-$125M Projected 2026 Revenue $20.5-$25M Projected 2026 Adj. EBITDA* 17%-20% Projected 2026 Adj. EBITDA Margin*

6 Campus Overview Site 1 – HQ (Lyman Blvd.) 150,000 sqft Site 2 (Lakeview Drive) 78,000 sqft Site 3 (Shelby Court) 20,000 sqft Manufacturing Operations • Sodium hyaluronate manufacturing (fermentation) • Drug and medical device formulation and filling • Secondary packaging • Microbiology and analytical quality control laboratories • Warehousing: 6,400 ft2 CRT; 1,500 ft2 cooler • Distribution Development Operations • Pilot laboratory Manufacturing Operations • Final packaging • Warehousing: 16,400 ft2 CRT; 4,000 sqft cooler • Distribution • Quality control laboratory • Particulate lab Development Operations • Analytical development laboratory Manufacturing Operations • Receipt, inspection, and warehousing of raw materials and components • 10,000 ft2 CRT; 1,795 ft2 cooler • Storage and distribution of finished goods • Potential for future expansion (120,000 ft2 available) 248,000sqft ~400State-of-the-art facilities, within 2 square miles Employees
7 Executing Our Strategy to Drive Sustainable Growth Strong commercial foundation with long-term customer relationships High-potential late-stage development pipeline representing significant future recurring revenue opportunity Revamped commercial strategy positioned to drive impactful growth over the mid- to long-term Disciplined cost structure approach designed to unlock additional value via organizational efficiency, strategic investments, and enhanced procurement Experienced and proven leadership team with deep expertise in the CDMO industry

8 Financial Highlights Comparison to comparable prior-year period Recent Developments • Ended December 2025 with over $38.9 million in liquidity, including cash of $17.5 million and availability under our revolver of $21.4 million • Cash from operations of $7.3 million and free cash flow of $3.6 million during transition period • Improved workforce productivity in manufacturing by more than 20% over the past 18 months • Significant improvement in Operating expenses with $11.1 million reduction, period-over-period • Ongoing preparation for existing commercial customer demand in 2027 including new HA specification for Asian market and completion of aseptic stability batches • Five new programs signed with new customers during fiscal transition period, including one late- state GLP-1 program, two commercial site transfer and two early-stage programs 7-month Transition Period December 2025 $75.5M Revenues $18.0M Net Loss $13.1M Adjusted EBITDA* * Non-GAAP measure. See disclaimers on slides 2, 3 & 4, and “Reconciliation of Non-GAAP Financial Measures” slide 7-month Period December 2024 $63.0M Revenues $30.6M Net Loss $2.6M Adjusted EBITDA* (Unaudited)

9 We Serve Large and Growing Markets with Strong Tailwinds Global Injectable CDMO $10B Market1 +10% CAGR Acceleration of US-based Manufacturing Global CDMO $120B Market1 +8% CAGR 50%+ of Annual US Drug Approvals are Injectables2 GLP-1 $47B Market3 Expected to Increase 10X 1. Jefferies September 2024 PBOA - 8th Annual Meeting Uncovering Life Sciences Investment Trends /J. Miller October 2024 – Outsourcing Includes drug product (finished dose form), drug substance (active pharmaceutical ingredients (API) 2. William Blair Equity Research August 2024 – Percent of FDA Approvals for 2023 and YTD as of July 31, 2024 3. Markets and Markets July 2024- GLP-1 Analogues Market Size, Share & Trends 2032

10 $90M Invested over Previous Five Years • Significant growth CapEx complete – enables execution of mid-term plan • State-of-the-art, 5-head isolator filler – ~100% increase in annual production capacity* – Full isolator technology, state-of-the-art containment – Significantly expanded available capacity – Broad capability: vials, syringes & cartridges – Strengthens compliance – ~25 million annual unit production capacity * Based on estimates derived from internal testing and historical capacity data. Significant Investment in Capabilities Supporting Growth

11 The Lifecore Difference Technical Expertise Decades of proven experience in complex injectables Integrated Model Development to commercialization Quality Multi-compendial regulatory system

Our Journey 12 Position Company for Growth • Expand capabilities and capacity • Refine and rebuild development pipeline • Revamp commercial strategy • Stabilize and right-size business • Implement performance- driven culture Sustained Growth • Achieve CAGR and margin targets • Grow commercialized program revenue • Continue pipeline commercialization • Expand and advance the development pipeline to unlock the next wave of commercial growth Drive Growth • Increase production to address doubling of fill finish demand for largest customer • Support commercialization of late-stage pipeline • Strengthen development pipeline w/ new programs • Drive margin improvement through operational and cost-containment initiatives 2024 – 2026 2027 – 2028 2029 +

13 Aggressive and Achievable Growth Strategy • Strong commercial foundation • High-potential late-stage development pipeline • Revamped commercial strategy • Disciplined cost structure approach • Experienced and proven leadership team Targeting 12% Revenue CAGR and Adjusted EBITDA Margins of 25%+ by 2029
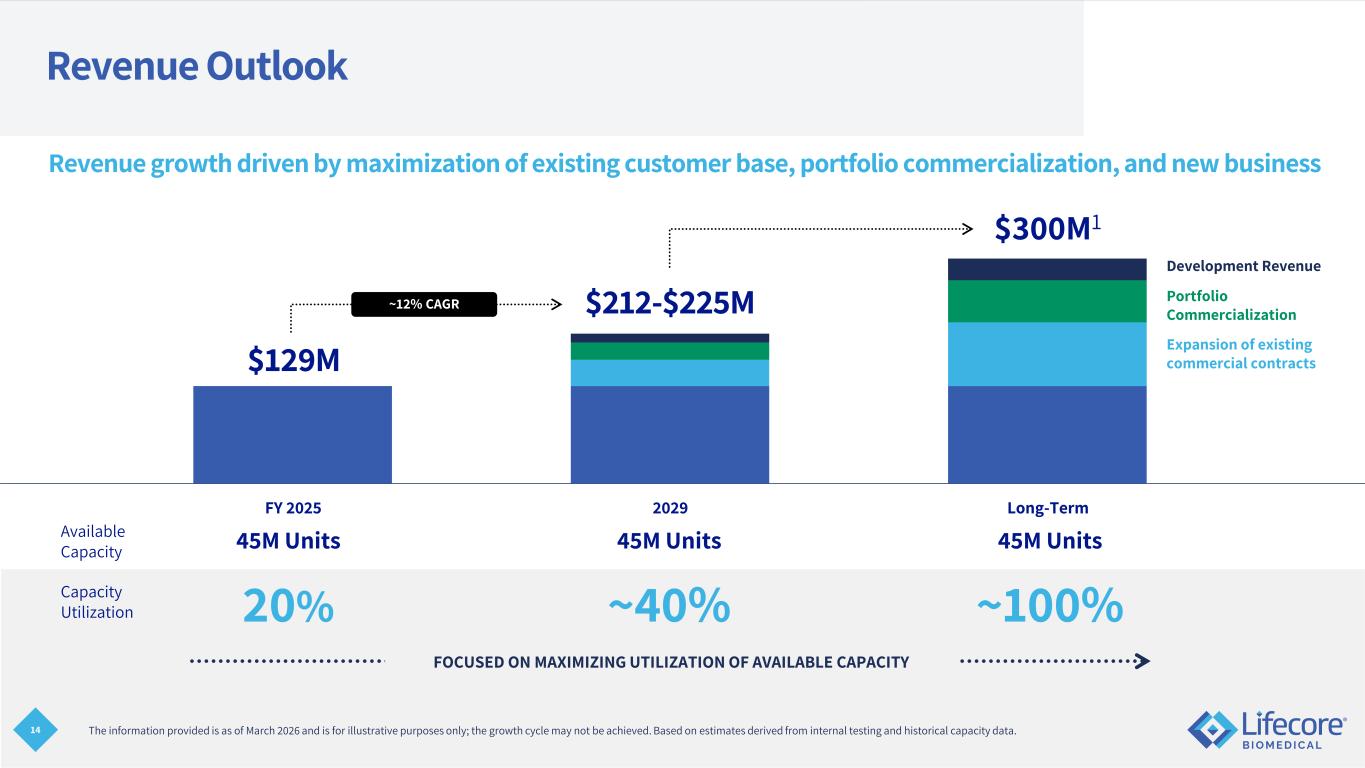
14 FOCUSED ON MAXIMIZING UTILIZATION OF AVAILABLE CAPACITY Revenue Outlook Expansion of existing commercial contracts Portfolio Commercialization Development Revenue Revenue growth driven by maximization of existing customer base, portfolio commercialization, and new business 45M Units 45M Units 45M Units 20% ~40% ~100% Available Capacity Capacity Utilization The information provided is as of March 2026 and is for illustrative purposes only; the growth cycle may not be achieved. Based on estimates derived from internal testing and historical capacity data. FY 2025 2029 Long-Term ~12% CAGR $129M $212-$225M $300M1
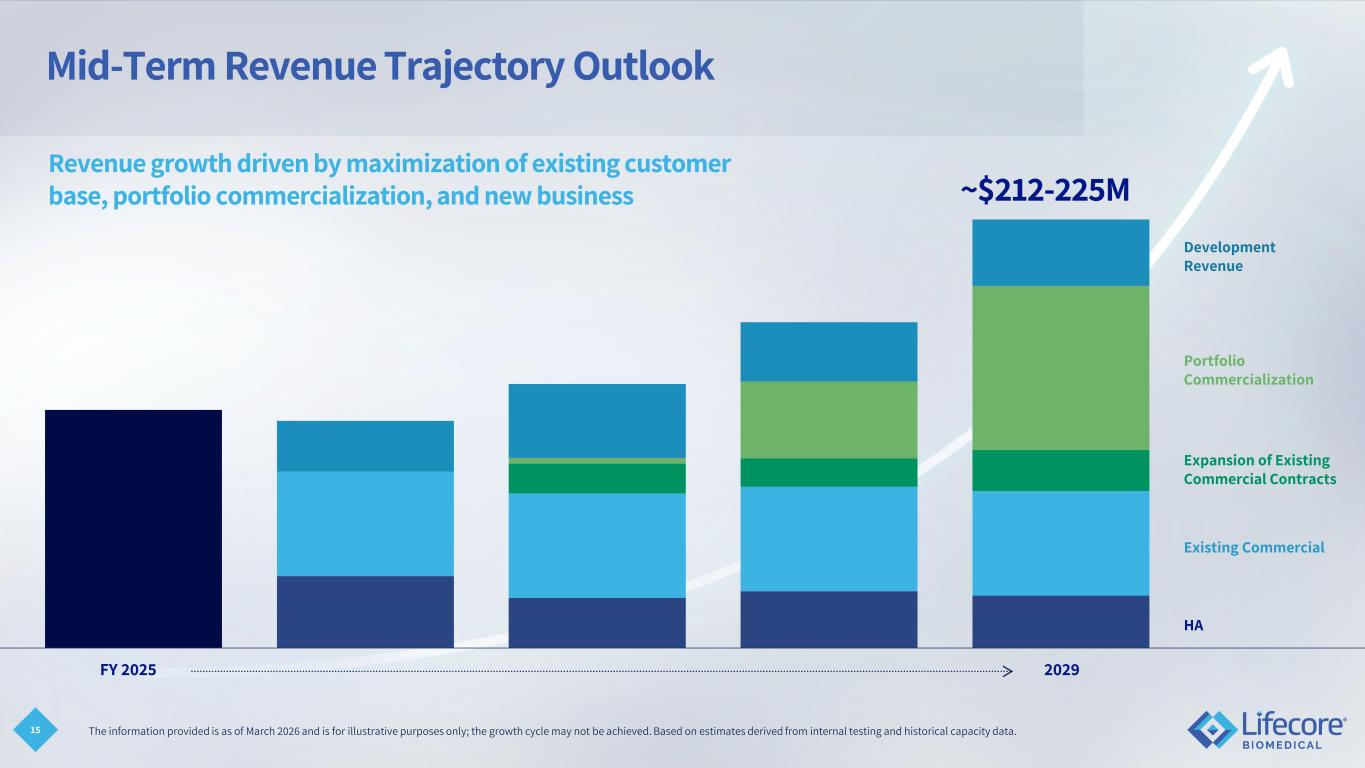
15 Mid-Term Revenue Trajectory Outlook Revenue growth driven by maximization of existing customer base, portfolio commercialization, and new business FY 2025 2029 ~$212-225M HA Existing Commercial Expansion of Existing Commercial Contracts Portfolio Commercialization Development Revenue The information provided is as of March 2026 and is for illustrative purposes only; the growth cycle may not be achieved. Based on estimates derived from internal testing and historical capacity data.
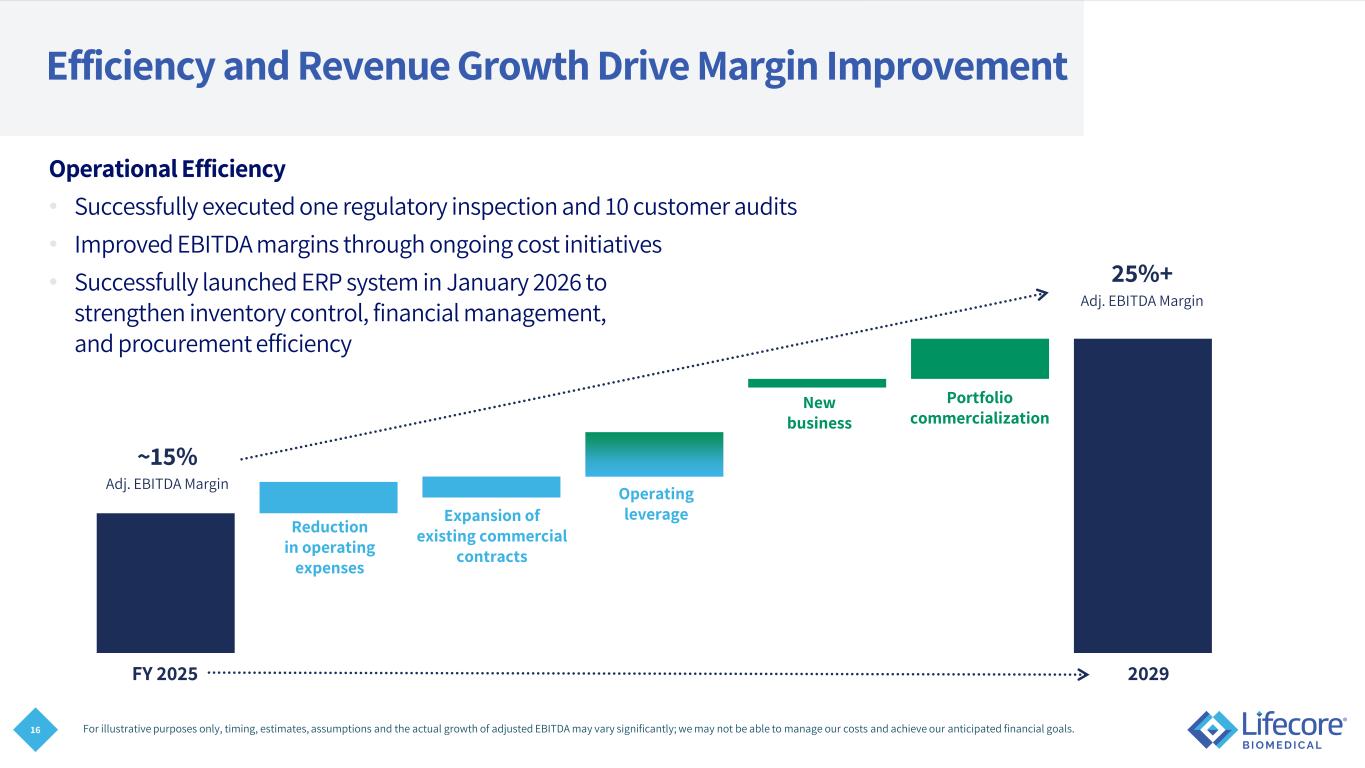
16 FY 2025 2029 Efficiency and Revenue Growth Drive Margin Improvement Operational Efficiency • Successfully executed one regulatory inspection and 10 customer audits • Improved EBITDA margins through ongoing cost initiatives • Successfully launched ERP system in January 2026 to strengthen inventory control, financial management, and procurement efficiency For illustrative purposes only, timing, estimates, assumptions and the actual growth of adjusted EBITDA may vary significantly; we may not be able to manage our costs and achieve our anticipated financial goals. ~15% 25%+ Adj. EBITDA Margin Adj. EBITDA Margin Reduction in operating expenses Expansion of existing commercial contracts Portfolio commercialization New business Operating leverage
Executing Three-Pronged Growth Strategy 17 Maximizing Existing Customer Business Advancing Programs Towards Commercialization Driving New Business
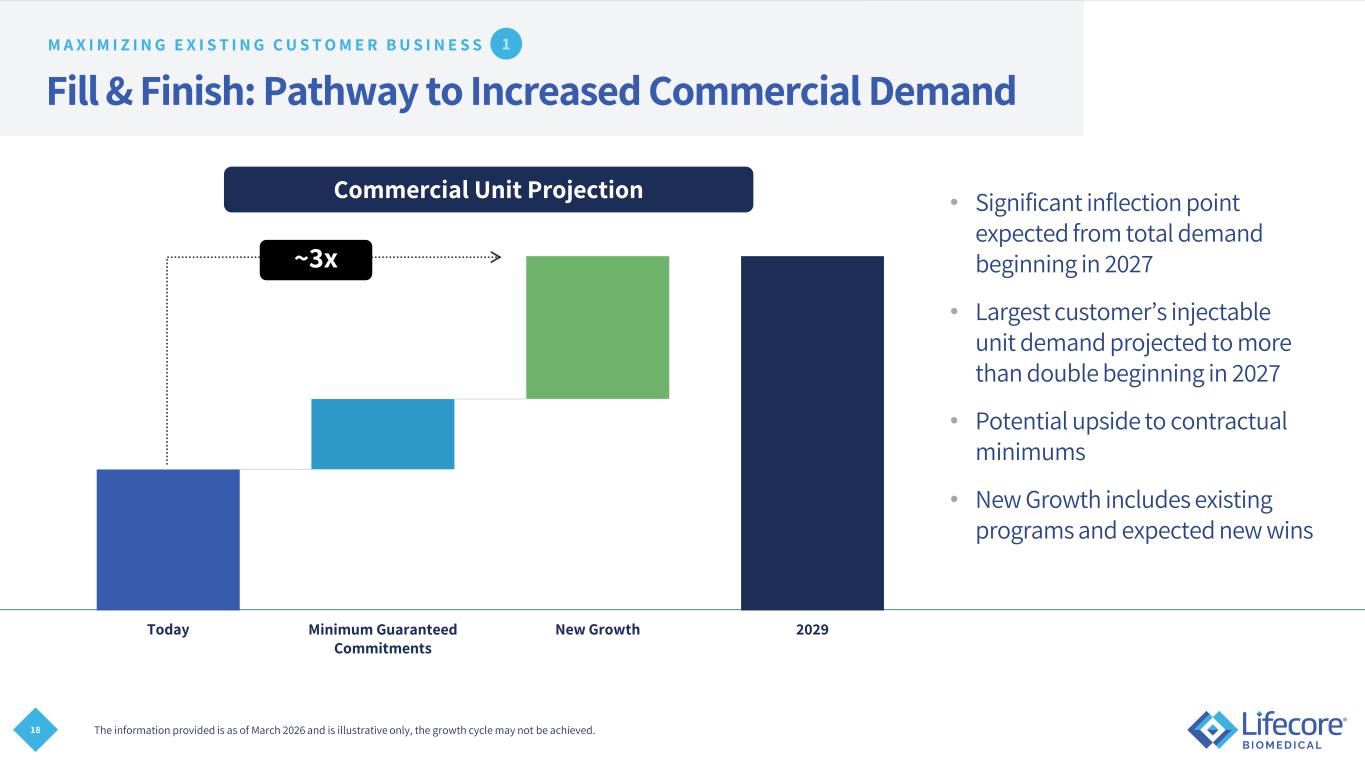
18 Fill & Finish: Pathway to Increased Commercial Demand • Significant inflection point expected from total demand beginning in 2027 • Largest customer’s injectable unit demand projected to more than double beginning in 2027 • Potential upside to contractual minimums • New Growth includes existing programs and expected new wins The information provided is as of March 2026 and is illustrative only, the growth cycle may not be achieved. M A X I M I Z I N G E X I S T I N G C U S T O M E R B U S I N E S S Today Minimum Guaranteed Commitments New Growth 2029 Commercial Unit Projection ~3x
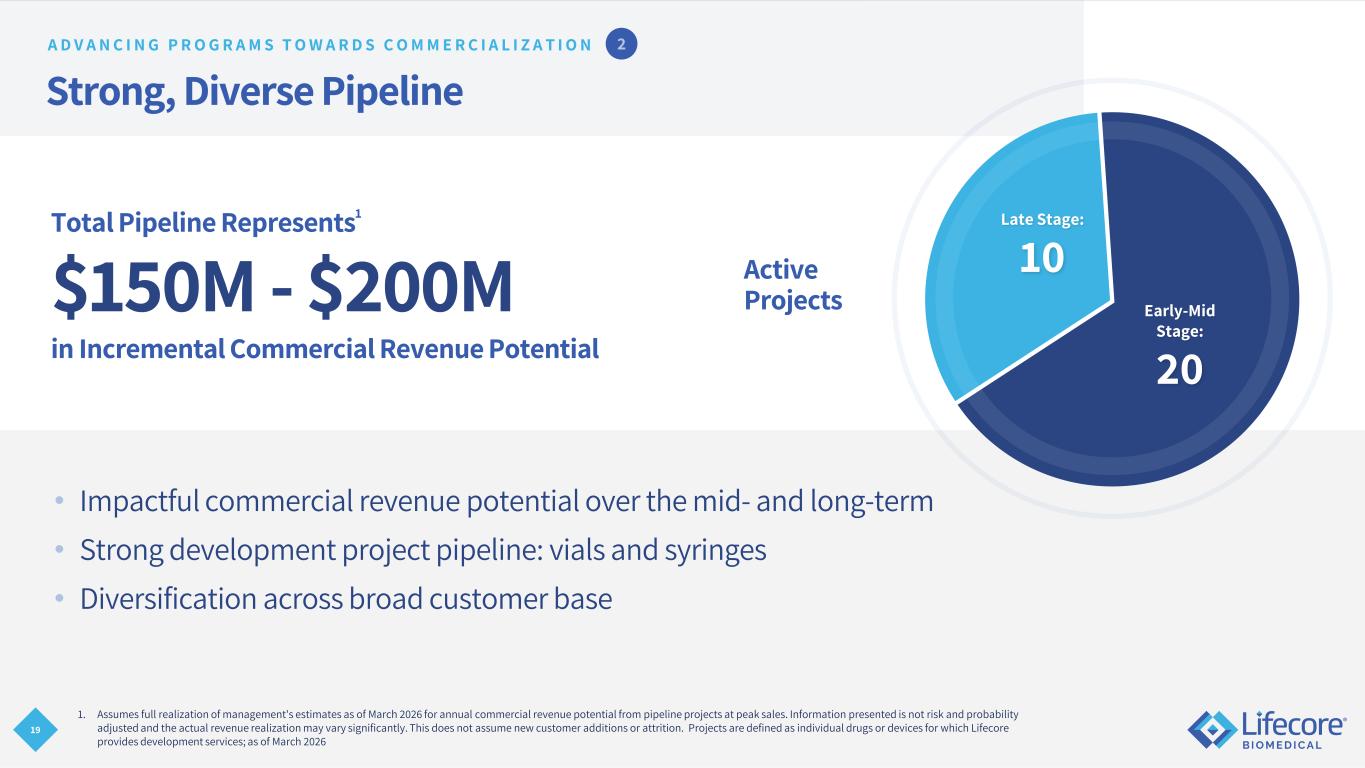
19 Strong, Diverse Pipeline • Impactful commercial revenue potential over the mid- and long-term • Strong development project pipeline: vials and syringes • Diversification across broad customer base Total Pipeline Represents1 $150M - $200M in Incremental Commercial Revenue Potential 1. Assumes full realization of management's estimates as of March 2026 for annual commercial revenue potential from pipeline projects at peak sales. Information presented is not risk and probability adjusted and the actual revenue realization may vary significantly. This does not assume new customer additions or attrition. Projects are defined as individual drugs or devices for which Lifecore provides development services; as of March 2026 Active Projects Late Stage: 10 Early-Mid Stage: 20 A D V A N C I N G P R O G R A M S T O W A R D S C O M M E R C I A L I Z A T I O N
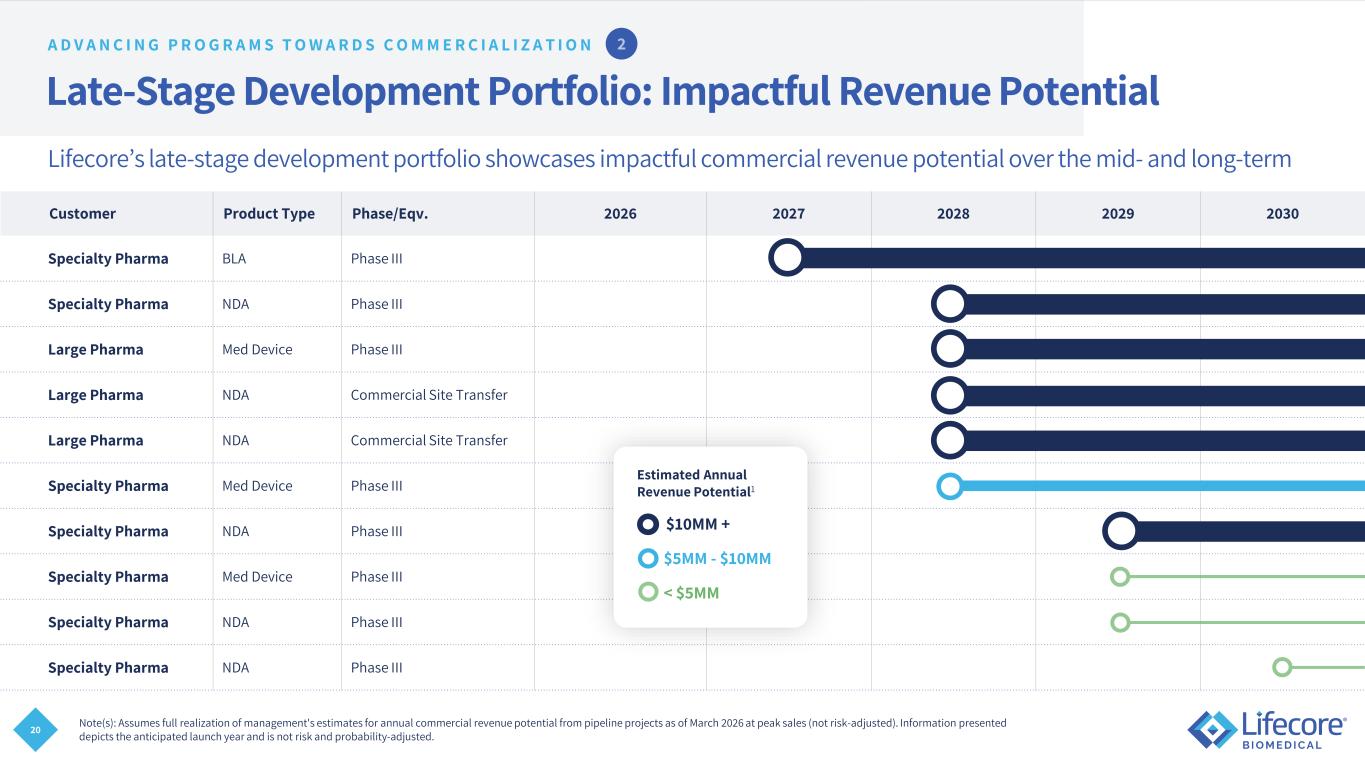
20 Late-Stage Development Portfolio: Impactful Revenue Potential Customer Product Type Phase/Eqv. 2026 2027 2028 2029 2030 Specialty Pharma BLA Phase III Specialty Pharma NDA Phase III Large Pharma Med Device Phase III Large Pharma NDA Commercial Site Transfer Large Pharma NDA Commercial Site Transfer Specialty Pharma Med Device Phase III Specialty Pharma NDA Phase III Specialty Pharma Med Device Phase III Specialty Pharma NDA Phase III Specialty Pharma NDA Phase III Note(s): Assumes full realization of management's estimates for annual commercial revenue potential from pipeline projects as of March 2026 at peak sales (not risk-adjusted). Information presented depicts the anticipated launch year and is not risk and probability-adjusted. $10MM + $5MM - $10MM < $5MM Estimated Annual Revenue Potential1 A D V A N C I N G P R O G R A M S T O W A R D S C O M M E R C I A L I Z A T I O N Lifecore’s late-stage development portfolio showcases impactful commercial revenue potential over the mid- and long-term

21 Attracting New High-Value Business Leveraging state-of-the-art capabilities Strategically expanding target market Upgrading sales/marketing strategy and talent D R I V I N G N E W B U S I N E S S • Increase in quality and quantity of business development pipeline • Recent addition of late stage GLP-1 program and two impactful commercial site transfers • Expansion into other indication areas beyond traditional focus in ophthalmology

22 Key Takeaways High-Growth Market Expected to Increase by 100% by 2030 Capital Investments Enable Clear Path to Scale Experienced Leadership & Exceptional Track Record of Success Aggressive and Achievable Growth Strategy of Both Top and Bottom Line
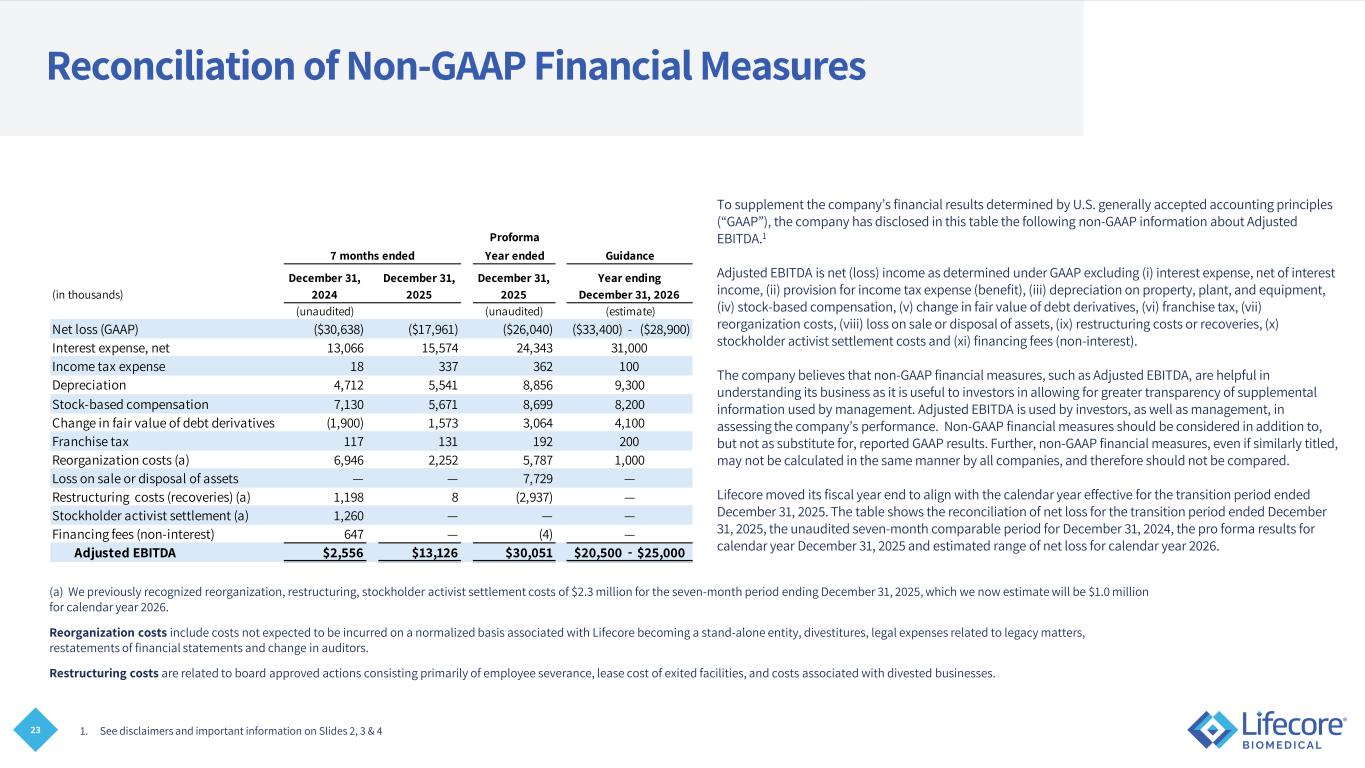
23 Reconciliation of Non-GAAP Financial Measures To supplement the company’s financial results determined by U.S. generally accepted accounting principles (“GAAP”), the company has disclosed in this table the following non-GAAP information about Adjusted EBITDA.1 Adjusted EBITDA is net (loss) income as determined under GAAP excluding (i) interest expense, net of interest income, (ii) provision for income tax expense (benefit), (iii) depreciation on property, plant, and equipment, (iv) stock-based compensation, (v) change in fair value of debt derivatives, (vi) franchise tax, (vii) reorganization costs, (viii) loss on sale or disposal of assets, (ix) restructuring costs or recoveries, (x) stockholder activist settlement costs and (xi) financing fees (non-interest). The company believes that non-GAAP financial measures, such as Adjusted EBITDA, are helpful in understanding its business as it is useful to investors in allowing for greater transparency of supplemental information used by management. Adjusted EBITDA is used by investors, as well as management, in assessing the company’s performance. Non-GAAP financial measures should be considered in addition to, but not as substitute for, reported GAAP results. Further, non-GAAP financial measures, even if similarly titled, may not be calculated in the same manner by all companies, and therefore should not be compared. Lifecore moved its fiscal year end to align with the calendar year effective for the transition period ended December 31, 2025. The table shows the reconciliation of net loss for the transition period ended December 31, 2025, the unaudited seven-month comparable period for December 31, 2024, the pro forma results for calendar year December 31, 2025 and estimated range of net loss for calendar year 2026. 1. See disclaimers and important information on Slides 2, 3 & 4 (a) We previously recognized reorganization, restructuring, stockholder activist settlement costs of $2.3 million for the seven-month period ending December 31, 2025, which we now estimate will be $1.0 million for calendar year 2026. Reorganization costs include costs not expected to be incurred on a normalized basis associated with Lifecore becoming a stand-alone entity, divestitures, legal expenses related to legacy matters, restatements of financial statements and change in auditors. Restructuring costs are related to board approved actions consisting primarily of employee severance, lease cost of exited facilities, and costs associated with divested businesses. Proforma Year ended (in thousands) December 31, 2024 December 31, 2025 December 31, 2025 (unaudited) (unaudited) Net loss (GAAP) ($30,638) ($17,961) ($26,040) ($33,400) - ($28,900) Interest expense, net 13,066 15,574 24,343 Income tax expense 18 337 362 Depreciation 4,712 5,541 8,856 Stock-based compensation 7,130 5,671 8,699 Change in fair value of debt derivatives (1,900) 1,573 3,064 Franchise tax 117 131 192 Reorganization costs (a) 6,946 2,252 5,787 Loss on sale or disposal of assets — — 7,729 Restructuring costs (recoveries) (a) 1,198 8 (2,937) Stockholder activist settlement (a) 1,260 — — Financing fees (non-interest) 647 — (4) Adjusted EBITDA $2,556 $13,126 $30,051 $20,500 - $25,000 — 4,100 — — 1,000 — 200 Guidance Year ending December 31, 2026 31,000 100 (estimate) 8,200 7 months ended 9,300
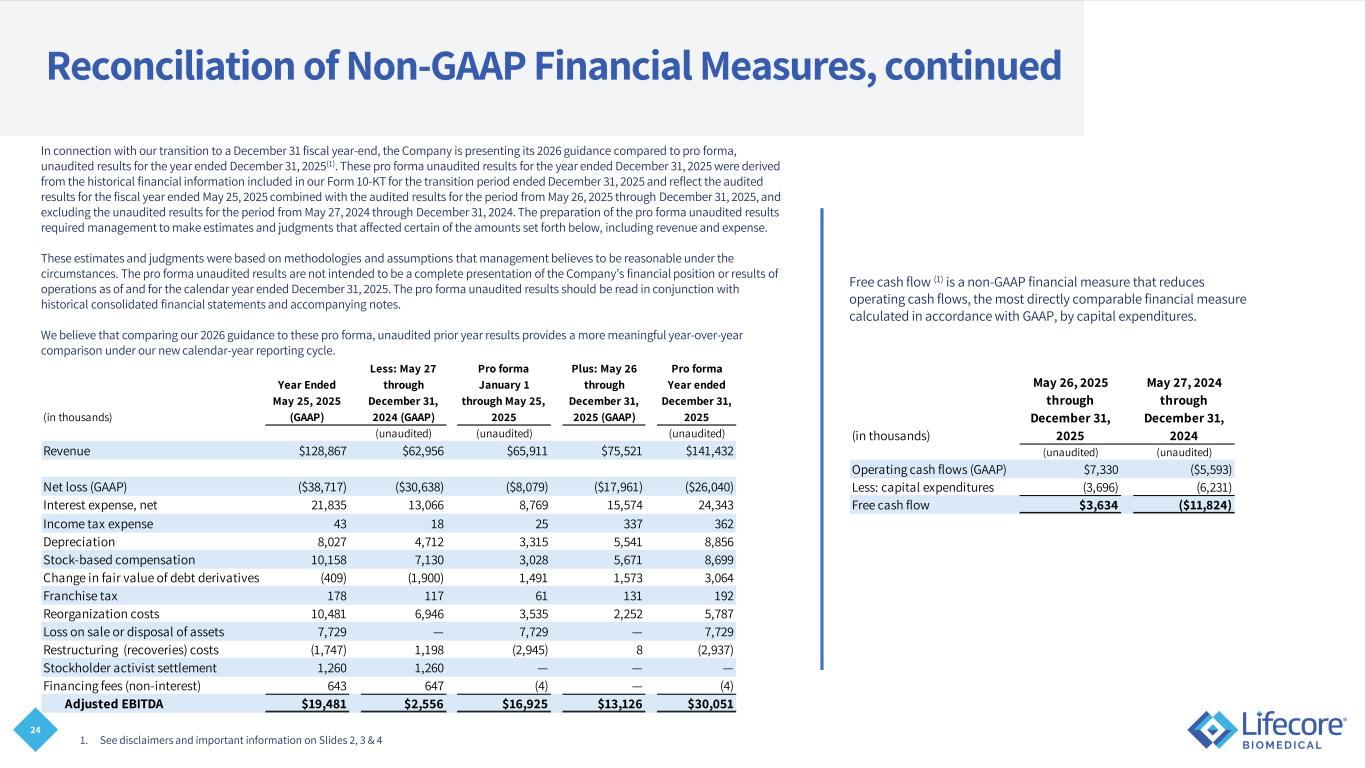
24 Reconciliation of Non-GAAP Financial Measures, continued 1. See disclaimers and important information on Slides 2, 3 & 4 In connection with our transition to a December 31 fiscal year-end, the Company is presenting its 2026 guidance compared to pro forma, unaudited results for the year ended December 31, 2025(1). These pro forma unaudited results for the year ended December 31, 2025 were derived from the historical financial information included in our Form 10-KT for the transition period ended December 31, 2025 and reflect the audited results for the fiscal year ended May 25, 2025 combined with the audited results for the period from May 26, 2025 through December 31, 2025, and excluding the unaudited results for the period from May 27, 2024 through December 31, 2024. The preparation of the pro forma unaudited results required management to make estimates and judgments that affected certain of the amounts set forth below, including revenue and expense. These estimates and judgments were based on methodologies and assumptions that management believes to be reasonable under the circumstances. The pro forma unaudited results are not intended to be a complete presentation of the Company’s financial position or results of operations as of and for the calendar year ended December 31, 2025. The pro forma unaudited results should be read in conjunction with historical consolidated financial statements and accompanying notes. We believe that comparing our 2026 guidance to these pro forma, unaudited prior year results provides a more meaningful year-over-year comparison under our new calendar-year reporting cycle. Free cash flow (1) is a non-GAAP financial measure that reduces operating cash flows, the most directly comparable financial measure calculated in accordance with GAAP, by capital expenditures. (in thousands) (unaudited) (unaudited) Operating cash flows (GAAP) $7,330 ($5,593) Less: capital expenditures (3,696) (6,231) Free cash flow $3,634 ($11,824) May 27, 2024 through December 31, 2024 May 26, 2025 through December 31, 2025 (in thousands) (unaudited) (unaudited) (unaudited) Revenue $128,867 $62,956 $65,911 $75,521 $141,432 Net loss (GAAP) ($38,717) ($30,638) ($8,079) ($17,961) ($26,040) Interest expense, net 21,835 13,066 8,769 15,574 24,343 Income tax expense 43 18 25 337 362 Depreciation 8,027 4,712 3,315 5,541 8,856 Stock-based compensation 10,158 7,130 3,028 5,671 8,699 Change in fair value of debt derivatives (409) (1,900) 1,491 1,573 3,064 Franchise tax 178 117 61 131 192 Reorganization costs 10,481 6,946 3,535 2,252 5,787 Loss on sale or disposal of assets 7,729 — 7,729 — 7,729 Restructuring (recoveries) costs (1,747) 1,198 (2,945) 8 (2,937) Stockholder activist settlement 1,260 1,260 — — — Financing fees (non-interest) 643 647 (4) — (4) Adjusted EBITDA $19,481 $2,556 $16,925 $13,126 $30,051 Plus: May 26 through December 31, 2025 (GAAP) Less: May 27 through December 31, 2024 (GAAP) Pro forma Year ended December 31, 2025 Year Ended May 25, 2025 (GAAP) Pro forma January 1 through May 25, 2025

Thank you