THE UNITED STATES SECURITIES AND EXCHANGE COMMISSION (“SEC”) DOES NOT PASS UPON THE MERITS OF OR GIVE ITS APPROVAL TO ANY SECURITIES OFFERED OR THE TERMS OF THE OFFERING, NOR DOES IT PASS UPON THE ACCURACY OR COMPLETENESS OF ANY OFFERING CIRCULAR OR OTHER SELLING LITERATURE. THESE SECURITIES ARE OFFERED PURSUANT TO AN EXEMPTION FROM REGISTRATION WITH THE COMMISSION; HOWEVER, THE COMMISSION HAS NOT MADE AN INDEPENDENT DETERMINATION THAT THIS INVESTMENT INVOLVES A DEGREE OF RISK THAT MAY NOT BE SUITABLE FOR ALL PERSONS. ONLY THOSE INVESTORS WHO CAN BEAR THE LOSS OF A SIGNIFICANT PORTION OF THEIR INVESTMENT SHOULD PARTICIPATE IN THE INVESTMENT. (SEE “RISK FACTORS” BELOW.)
AN OFFERING STATEMENT PURSUANT TO REGULATION A RELATING TO THESE SECURITIES HAS BEEN FILED WITH THE SECURITIES AND EXCHANGE COMMISSION. INFORMATION CONTAINED IN THIS PRELIMINARY OFFERING CIRCULAR IS SUBJECT TO COMPLETION OR AMENDMENT. THESE SECURITIES MAY NOT BE SOLD NOR MAY OFFERS TO BUY BE ACCEPTED BEFORE THE OFFERING STATEMENT FILED WITH THE COMMISSION IS QUALIFIED. THIS PRELIMINARY OFFERING CIRCULAR SHALL NOT CONSTITUTE AN OFFER TO SELL OR THE SOLICITATION OF AN OFFER TO BUY NOR MAY THERE BE ANY SALES OF THESE SECURITIES IN ANY STATE IN WHICH SUCH OFFER, SOLICITATION OR SALE WOULD BE UNLAWFUL BEFORE REGISTRATION OR QUALIFICATION UNDER THE LAWS OF ANY SUCH STATE. THE COMPANY MAY ELECT TO SATISFY ITS OBLIGATION TO DELIVER A FINAL OFFERING CIRCULAR BY SENDING YOU A NOTICE WITHIN TWO BUSINESS DAYS AFTER THE COMPLETION OF THE SALE THAT CONTAINS THE URL WHERE THE FINAL OFFERING CIRCULAR OR THE OFFERING STATEMENT IN WHICH SUCH FINAL OFFERING CIRCULAR WAS FILED MAY BE OBTAINED.
THE SECURITIES OFFERED HAVE NOT BEEN APPROVED OR DISAPPROVED BY ANY STATE REGULATORY AUTHORITY NOR HAS ANY STATE REGULATORY AUTHORITY PASSED UPON OR ENDORSED THE MERITS OF THE OFFERING OR THE ACCURACY OR ADEQUACY OF THIS OFFERING CIRCULAR. ANY REPRESENTATION TO THE CONTRARY IS UNLAWFUL.
Preliminary Offering Circular
For
McGinley Orthopaedic Innovations, Inc.
A Wyoming Corporation
August 11, 2022
SECURITIES OFFERED : Equity in the form of Shares of common stock
PRICE PER SHARE : $5.25 per Share of Common Stock
MAXIMUM OFFERING AMOUNT : $40,000,000.00
MINIMUM OFFERING AMOUNT : Not Applicable (No Minimum Offering Amount)
MINIMUM INVESTMENT : $100.00
CONTACT INFORMATION :
McGinley Orthopaedic Innovations, Inc.
234 E. 1st St. Suite 242
Casper, WY 82601
(307) 315-6403
Generally, no sale may be made to you in this Offering if the aggregate purchase price you pay is more than ten (10%) percent of the greater of your annual income or net worth. Different rules apply to accredited investors and non-natural persons. Before making any representation that your investment does not exceed applicable thresholds, Investors are encouraged to review rule 251(d)(2)(i)(C) of Regulation A. For general information on investing, Investors are encouraged to refer to www.investor.gov.
McGinley Orthopaedic Innovations, Inc. (the “Company” or the “Issuer”) is an orthopedic surgical device/implant innovator and manufacturer based in Casper, Wyoming. The Company was founded as McGinley Orthopaedic Innovations, LLC in 2012. The Company converted to a Wyoming Corporation on May 13, 2022 by filing Articles of Incorporation and a Statement of Conversion with the Wyoming Secretary of State (See Exhibit 2A “Articles of Incorporation and Other Documents”).
The minimum investment amount per Investor is One Hundred Dollars ($100.00), representing Nineteen and Four Hundredths (19.04) Shares at Five Dollars and Twenty-Five Cents ($5.25) per Share. The Company is run by a board of directors, comprised of one (1) director (the “Board” collectively, “Director” when referring to a director). The day-to-day management and investment decisions of the Company are vested in the Board and in the officers of the Company (the “Officers”).
Sales of the Shares pursuant to the Offering will commence immediately upon qualification of the Offering by the Securities and Exchange Commission (the “Effective Date”) and will terminate at the discretion of the Board or twelve (12) months following the Effective Date, whichever is earlier. The maximum amount of the Offering shall not exceed Forty Million Dollars ($40,000,000) in any twelve (12) month period (“Maximum Offering Amount”) in accordance with Tier II of Regulation A as set forth under the Securities Act of 1933, as amended, (“Reg A Tier II” or “Tier II”). The Company intends to offer the Shares described herein on a continuous and ongoing basis pursuant to Rule 251(d)(3)(i)(F). Further, the acceptance of Investor subscriptions, may be briefly paused at times to allow the Company to effectively and accurately process and settle subscriptions that have been received. (See “Terms of the Offering” below.) The Company may increase the Maximum Offering Amount at its sole and absolute discretion, subject to qualification by the SEC of a post-qualification amendment.
Prior to this Offering, there has been no public market for the Shares, and none is expected to develop. The Offering price is arbitrary and does not bear any relationship to the value of the assets of the Company. The Company does not currently have plans to list any Shares on any securities market, however reserves the right to do so in the future. Investing in the Company through the purchase of Shares involves risks, some of which are set forth below. See the section titled “Risk Factors” to read about the factors an Investor should consider prior to purchasing Shares.
Investors who purchase Shares will become shareholders of the Company (“Investors” or “Shareholders” subject to the terms of the Articles of Incorporation and the Bylaws of the Company (see Exhibit 2A “Articles of Incorporation and Other Corporate Documents” and Exhibit 2B “Bylaws”) once the Company deposits the Investor’s investment into the Company’s main operating account.
The Director/Officer will receive compensation from the Company as employees (see “Risk Factors” below starting on Page 5 and, “Compensation of Directors and Officers” below). Investing in the Shares is speculative and involves substantial risks, including risk of complete loss. Prospective Investors should purchase these securities only if they can afford a complete loss of their investment (see “Risk Factors” below starting on Page 6) .
As of the date of this Offering Circular, the Company has engaged KoreConX as transfer agent in relation to this Offering. The Company has engaged Wilmington Trust as Escrow Agent for this Offering.
NO PERSON HAS BEEN AUTHORIZED IN CONNECTION WITH THIS OFFERING TO GIVE ANY INFORMATION OR TO MAKE ANY REPRESENTATIONS OTHER THAN THAT INFORMATION AND THOSE REPRESENTATIONS SPECIFICALLY CONTAINED IN THIS OFFERING CIRCULAR; ANY OTHER INFORMATION OR REPRESENTATIONS SHOULD NOT BE RELIED UPON. ANY PROSPECTIVE PURCHASER OF THE SECURITIES WHO RECEIVES ANY OTHER INFORMATION OR REPRESENTATIONS SHOULD CONTACT THE COMPANY IMMEDIATELY TO DETERMINE THE ACCURACY OF SUCH INFORMATION AND REPRESENTATIONS. NEITHER THE DELIVERY OF THIS OFFERING CIRCULAR NOR ANY SALES HEREUNDER SHALL, UNDER ANY CIRCUMSTANCES, CREATE AN IMPLICATION THAT THERE HAS BEEN NO CHANGE IN THE AFFAIRS OF THE COMPANY OR IN THE INFORMATION SET FORTH HEREIN SINCE THE DATE OF THIS OFFERING CIRCULAR SET FORTH ABOVE.
THE INFORMATION CONTAINED IN THIS OFFERING CIRCULAR HAS BEEN SUPPLIED BY THE COMPANY. THIS OFFERING CIRCULAR CONTAINS SUMMARIES OF DOCUMENTS NOT CONTAINED IN THIS OFFERING CIRCULAR, BUT ALL SUCH SUMMARIES ARE QUALIFIED IN THEIR ENTIRETY BY REFERENCES TO THE ACTUAL DOCUMENTS. COPIES OF DOCUMENTS REFERRED TO IN THIS OFFERING CIRCULAR, BUT NOT INCLUDED AS AN EXHIBIT, WILL BE MADE AVAILABLE TO QUALIFIED PROSPECTIVE INVESTORS UPON REQUEST.
RULE 251(D)(3)(I)(F) DISCLOSURE. RULE 251(D)(3)(I)((F) PERMITS REGULATION A OFFERINGS TO CONDUCT ONGOING CONTINUOUS OFFERINGS OF SECURITIES FOR MORE THAN THIRTY (30) DAYS AFTER THE QUALIFICATION DATE IF: (1) THE OFFERING WILL COMMENCE WITHIN TWO (2) DAYS AFTER THE QUALIFICATION DATE; (2) THE OFFERING WILL BE MADE ON A CONTINUOUS AND ONGOING BASIS FOR A PERIOD THAT MAY BE IN EXCESS OF THIRTY (30) DAYS OF THE INITIAL QUALIFICATION DATE; (3) THE OFFERING WILL BE IN AN AMOUNT THAT, AT THE TIME THE OFFERING CIRCULAR IS QUALIFIED, IS REASONABLY EXPECTED TO BE OFFERED AND SOLD WITHIN ONE (1) YEAR FROM THE INITIAL QUALIFICATION DATE; AND (4) THE SECURITIES MAY BE OFFERED AND SOLD ONLY IF NOT MORE THAN THREE (3) YEARS HAVE ELAPSED SINCE THE INITIAL QUALIFICATION DATE OF THE OFFERING, UNLESS A NEW OFFERING CIRCULAR IS SUBMITTED AND FILED BY THE COMPANY PURSUANT TO RULE 251(D) (3)(I)((F) WITH THE SEC COVERING THE REMAINING SECURITIES OFFERED UNDER THE PREVIOUS OFFERING; THEN THE SECURITIES MAY CONTINUE TO BE OFFERED AND SOLD UNTIL THE EARLIER OF THE QUALIFICATION DATE OF THE NEW OFFERING CIRCULAR OR THE ONE HUNDRED EIGHTY (180) CALENDAR DAYS AFTER THE THIRD ANNIVERSARY OF THE INITIAL QUALIFICATION DATE OF THE PRIOR OFFERING CIRCULAR. THE COMPANY INTENDS TO OFFER THE SHARES DESCRIBED HEREIN ON A CONTINUOUS AND ONGOING BASIS PURSUANT TO RULE 251(D)(3)(I)(F). THE COMPANY INTENDS TO COMMENCE THE OFFERING IMMEDIATELY AND NO LATER THAN TWO (2) DAYS FROM THE INITIAL QUALIFICATION DATE. THE COMPANY REASONABLY EXPECTS TO OFFER AND SELL THE SECURITIES STATED IN THIS OFFERING CIRCULAR WITHIN ONE (1) YEAR FROM THE INITIAL QUALIFICATION DATE.
The Company will commence sales of the Shares immediately upon qualification of the Offering by the SEC. The Company approximates that sales will commence during Q3 – 2022.
| Price to Public* | Commissions** | Proceeds to Other Persons*** | Proceeds to the Company | |||||||||||||
| Amount to be Raised per Share | $ | 5.25 | $ | 0.22 | 1.31 | $ | 3.71 | |||||||||
| Minimum Investment Amount | $ | 100 | $ | 4.25 | $ | 25.00 | $ | 68.25 | ||||||||
| Minimum Offering Amount | N/A | N/A | N/A | N/A | ||||||||||||
| Maximum Offering Amount | $ | 40,000,000 | $ | 1,700,000 | $ | 10,000,000 | $ | 28,300,000 | ||||||||
*The Offering price to Investors was arbitrarily determined by the Board.
** The Company is not using an underwriter for the sale of Shares. These commissions listed are those for Rialto Markets, a FINRA broker-dealer. Rialto Markets is entitled to 3% on all passive sales of securities as placement agent. If securities are sold through the efforts of Rialto Markets, 8% will be due to Rialto Markets (instead of 3%) up to a maximum of $800,000 – for potential maximum commissions of $1,700,000. The commissions due to Rialto Markets are conditional on the services provided by Rialto Markets with respect to any one sale. See “Plan of Distribution “ below.
*** The Company intends to have selling shareholders as part of this Offering. See “Plan of Distribution” which includes a section named “Selling Shareholders” below.
FORWARD LOOKING STATEMENTS
Investors should not rely on forward-looking statements because they are inherently uncertain. Investors should not rely on forward-looking statements in this Offering Circular. This Offering Circular contains forward-looking statements that involve risks and uncertainties. The use of words such as “anticipated”, “projected”, “forecasted”, “estimated”, “prospective”, “believes”, “expects,” “plans”, “future”, “intends”, “should”, “can”, “could”, “might”, “potential”, “continue”, “may”, “will”, and similar expressions identify these forward-looking statements. Investors should not place undue reliance on these forward-looking statements, which may apply only as of the date of this Offering Circular, and the Company undertakes no obligation to publicly update or revise any forward-looking information, other than as required by applicable law.
TABLE OF CONTENTS
4
The following information is only a brief summary of, and is qualified in its entirety by, the detailed information appearing elsewhere in this Offering Circular. This Offering Circular, together with the exhibits attached including, but not limited to, the Articles of Incorporation and Company Bylaws, copies of which are attached hereto as Exhibit 2A and Exhibit 2B, respectively, and should be carefully read in their entirety before any investment decision is made. If there is a conflict between the terms contained in this Offering Circular and these documents, Articles of Incorporation and Bylaws shall prevail and control, and no Investor should rely on any reference herein to the Articles of Incorporation or Bylaws without consulting the actual underlying documents.
| COMPANY INFORMATION | McGinley Orthopaedic Innovations Inc. with a principal place of business located at 234 E. 1st St. Suite 242, Casper, WY 82601. | |
| MANAGEMENT | The Company is managed by a Board of Directors. The Board is comprised of one (1) Director who also serves as the Company’s one (1) Officer. See “Risk Factors” and “Directors, Officers, and Significant Employees” below. | |
| THE OFFERING | This Offering is the first capital raise by the Issuer under Regulation A. The Company was previously operating as a Wyoming Limited Liability Company. The Company is selling Company equity in the form of Common Stock (the “Common Stock” or the “Shares”) through this Offering. The Company will use the Proceeds of this Offering to scale up its existing operations. See “Use of Proceeds” below | |
| SECURITIES BEING OFFERED | The Shares are being offered at a purchase price of $5.25 per Share. The Minimum Investment Amount for any Investor is $100.00. Upon purchase of the Shares, a Shareholder is granted (i) the right to vote on all matters subject to a Common Stock vote; and (ii) the right to receive dividends or disbursements, when the Board declares such dividends or disbursements. For a summary of the rights granted to Shareholders, see “Description of the Securities” below. | |
| COMPENSATION TO DIRECTORS | The Company pays the Director and Officer’s
salary for his role as Director and Officer. For more information on this compensation see “Compensation of the
Director and Officers” section below. The Director, Officer, and employees of the Company will not be compensated through commissions for the sale of the Shares through this Offering. | |
| PRIOR EXPERIENCE OF COMPANY MANAGEMENT | The Director/Officer and significant employees have experience in technology company finance, operations, and manufacturing. See “Directors, Officers, and Significant Employees” below. | |
| INVESTOR SUITABILITY STANDARDS | The Shares will not be sold to any person unless
they are a “Qualified Purchaser”. A Qualified Purchaser includes: (1) an “Accredited Investor” as
that term is defined in Rule 501(a) of Regulation D promulgated under the Securities Act of 1933 (the “Securities
Act”); or (2) all other Investors who meet the investment limitations set forth in Rule 251(d)(2)(i)(C) of Regulation
A. Such persons as stated in (2) above must conform with the “Limitations on Investment Amount” as described in
this Summary below. Each person acquiring Shares may be required to represent that he, she, or it is purchasing the Shares for his, her, or its own account for investment purposes and not with a view to resell or distribute the securities. Each prospective purchaser of Shares may be required to furnish such information or certification as the Company may require in order to determine whether any person or entity purchasing Shares is an Accredited Investor if such is claimed by the Investor. | |
| LIMITATIONS ON INVESTMENT AMOUNT | For Qualified Purchasers who are Accredited Investors, there is no limitation as to the amount invested through the purchase of Shares. For non-Accredited Investors, the aggregate purchase price paid to the Company for the purchase of the Shares cannot be more than 10% of the greater of the purchaser’s (1) annual income or net worth as determined under Rule 501(a) of Regulation D, if purchaser is a natural person; or (2) revenue or net assets for the purchaser’s most recently completed fiscal year if purchaser is a non-natural person. Different rules apply to Accredited Investors and non-natural persons. Each Investor should review Rule 251(d)(2)(i)(C) of Regulation A as determined under Rule 501(a) of Regulation D before purchasing the Shares. | |
| COMMISSIONS FOR SELLING Shares | The Shares will be offered and sold directly by the Company, the Board, the Officers, and Company’s employees. No commissions for selling the Shares will be paid to the Company, the Board, the Officers, or the Company’s employees. The Company is not using an underwriter for the sale of Shares. The commissions listed are those for Rialto Markets, a FINRA broker-dealer. Rialto Markets is entitled to 3% on all passive sales of securities as placement agent. If securities are sold through the efforts of Rialto Markets, 8% will be due to Rialto Markets (instead of 3%) up to a maximum of $800,000 – for potential maximum commissions of $1,700,000. The commissions due to Rialto Markets are conditional on the services provided by Rialto Markets with respect to any one sale. See “Plan of Distribution “ below. | |
| NO LIQUIDITY | There is no public market for the Shares, and none is expected to develop in the near future. Additionally, the Shares will be transferable, in accordance with Federal and state securities laws, and Wyoming law. However, the Shares will not be listed for trading on any exchange or automated quotation system. (See “Description of the Securities” below.) Prospective Investors are urged to consult their own legal advisors with respect to secondary trading of the Shares. See “Risk Factors” below. | |
| SELLING SECURITYHOLDERS | 1,904,761.9 Shares of Common Stock are being offered for the account of Selling Shareholders. This represents 4.21% of the outstanding Shares of Common Stock as of the Date of this Offering Circular. | |
| BONUS SHARES | The Company is offering Bonus Shares as part of this Offering. This includes discounts from 3% to 55% under certain circumstances. See “Plan of Distribution” below. | |
| COMPANY EXPENSES | Except as otherwise provided herein, the Company shall bear all costs and expenses associated with the Offering, the operation of the Company, including, but not limited to, the annual tax preparation of the Company's tax returns, any state and federal income tax due, accounting fees, filing fees, independent audit reports, other costs and expenses, and other advisory fees. |
The Company’s has two primary product lines, the (i) IntelliSense Drill Technologyâ Drill and accessories line of products; and the (ii) Lever Action Plate Systemâ line of products.
The IntelliSense Drill Technology® and accessories will be referred to as the “IntelliSense Drill(s)” or “IntelliSense products”. Please see “Description of the Business” below for a list of what products are included in these terms.
The Lever Action Plate Systemâ line of products will be referred to as “LAPS” or “LAPS products”.
Both “IntelliSense Drill Technology" and “Lever Action Plate System” are registered trademarks owned by the Company. This Offering Circular will not continue to incorporate the “®“ symbol to identify these registered trademarks as such for the sake of simplicity and reserve all rights.
Collectively the IntelliSense products and LAPS products will be referred to collectively as the (the “Products”).
5
The SEC requires the Company to identify risks that are specific to its business and its financial condition. The Company is still subject to all the same risks that all companies in its business, and all companies in the economy, are exposed to. These include risks relating to economic downturns, political and economic events and technological developments (such as hacking and the ability to prevent hacking). Additionally, early-stage companies are inherently riskier than more developed companies. You should consider general risks as well as specific risks when deciding whether to invest.
Risks Related to the Company and Its Business
The Company has not been profitable since its inception.
The Company had net losses of approximately $2,858,000 and $2,200,000 for the years ended December 31, 2021 and 2020, respectively, as well as negative cash flow from operations of approximately $2,055,000 and $1,485,000, respectively. See “Part F/S”, below, and the audit report included therein. There is no assurance that the Company will ever be profitable or generate sufficient revenues to operate its business or pay dividends.
The loss of any member of the Company’s management team or their inability to attract and retain highly skilled scientists, engineers, and salespeople could adversely affect company business.
Company success depends on the skills, experience and performance of key members of the Company’s management team, including the Officer and Director. The individual and collective efforts of the Company’s sole Officer/Director and its significant employees will be essential as the Company continues to develop the Company and Products.
Company research and development and manufacturing operations depend on the Company’s ability to attract and retain highly-skilled employees including scientists, technicians and engineers, marketing managers and salespersons. The Company may not be able to attract or retain a sufficient number of qualified highly-skilled employees in the future due to the competition for qualified personnel in the medical device industry. The Company also faces competition from universities and public and private research institutions in recruiting and retaining highly qualified scientific personnel. Recruiting and retention difficulties can limit the ability to support Company. Other than for the Chief Executive Officer, the Company also does not maintain “key person” insurance on any of Company employees.
The Company may not be able to scale the Company’s Products on the anticipated timetable.
The Company’s Products may be difficult to scale to a commercially viable level since it must meet expectations that it is equivalent or superior to traditional orthopedic drills and implants in terms of efficacy, safety, and cost efficiency. The Company will continue to develop and refine the Products and their manufacturing processes to ensure the Products, and future iterations of the Products, meet performance goals and cost targets. The Company may need to perform additional laboratory and clinical trials in the future, and may encounter problems and delays. If future clinical trials reveal technical defects or reveal that the Company’s Products do not meet performance goals and cost targets, the Company’s commercialization schedule could be delayed as the Company attempts to devise solutions to the defects or problems. If the Company is unable to find solutions, the Company’s business may not be as successful.
The Company may not be able to successfully execute the business plan.
In addition to the requirement to successfully develop, manufacture, and market the Products for commercial success, the Company must also raise significant amounts of capital, foster relationships with key suppliers and attract customers. There is no guarantee that the Company will be able to achieve or sustain any of the foregoing within the anticipated timeframe or at all. Even though the Company’s principals are long-time industry professionals, who have been working for some time building relationships in the orthopedics industry, the Company may exceed the budget, encounter obstacles in research and development activities, or be hindered or delayed in implementing the Company’s commercialization plans, any of which could imperil the Company’s ability to secure customer contracts and begin generating significant revenues. In
addition, any such delays or problems may require the Company to secure additional funding over and above what the Company currently anticipates it requires in order to sustain business, which the Company may not be able to raise.
The Company’s Products may not achieve market acceptance for its Products thereby reducing the chance for success.
The Company is in the early stages of selling the Products. Unanticipated events may result in lower sales than anticipated, which could force the Company to limit expenditures on research and development, advertising, and general Company requirements for improving and expanding Product offerings. The Company cannot guarantee market demand or interest in the current Products or future product offerings, which could have a material adverse effect on business, results of operations, and overall financial condition.
The Company has only two main product lines.
The Company’s primary products are the (i) IntelliSense products and (ii) Lever Action Plate System products. These are two distinct products with disparate design and manufacturing paths. The Company may sell or license either or both of these product lines. The Company’s survival in the near term depends on being able to sell these Products to a sufficient number of customers to make a profit. The Company’s current customer base is still small, and the Company will only succeed if it can attract more customers for its primary product and maintain those customers.
The Company has not yet generated profits and it may take a long time for the Company to become profitable.
The Company has not yet generated any profit from the sale of its Products. The Company is working towards having sales of the Products, but the Company anticipates that upon a successful Offering, it will take time to achieve profitability, if at all.
The Company’s ability to raise capital and to commercialize the Products may be materially impacted by the ongoing COVID-19 pandemic.
The Company’s ability to raise capital and to commercialize the Products may be materially impacted by the ongoing COVID-19 pandemic. The full impact on the economy and the capital markets in the U.S. and the rest of the world from the COVID-19 pandemic are uncertain, in terms of both scale and duration. The high level of volatility in the capital markets may make it difficult to raise funds, especially for early stage companies that involve higher risk. If the Company raises sufficient funds to commercialize its Products, the Company may have difficulty securing supplies needed or manufacturing and distribution partners. The impact of social distancing measures and related workforce reductions may negatively impact the ability of suppliers to deliver the Company to the components the Company needs for manufacture or the ability of any of the Company’s potential partners to operate effectively to meet Company requirements. The Company cannot assure an Investor that, should it raise sufficient funds, it will be able to contract with suppliers, manufacturing partners or distribution partners at a level that would allow the Company to achieve profitability, or at all.
If the market chooses to buy competing Products, the Company may fail.
Although the Company believes that its Products will be commercially viable, there is no verification by the marketplace that the Products will be accepted by or purchased by customers at the scale desired by the Company The market may choose existing competitor products to those of the Company. If the market chooses to continue to use competing products, it may be more difficult for the Company to ever become profitable which would be substantially harm the business and, possibly, cause it to fail whereby the Investors could lose their entire investment. In addition to Company dependency on their continued services, Company future success will also depend on the ability to attract and retain additional future key personnel. The Company may face intense competition for such qualified individuals from well-established and better financed competitors. The Company may not be able to attract qualified new employees or retain existing employees, which may have a material adverse effect on the Company’s results of operations and financial condition.
The medical device industry is subject to rapidly changing technology which could make the Products and other products the Company develops obsolete.
The Company’s industry is characterized by rapid technological changes, frequent new product introductions and enhancements and evolving industry standards, all of which could make the Company’s Products obsolete. The Company’s future success will depend on its ability to anticipate and keep pace with the evolving needs of the industry on a timely and cost-effective basis and to pursue new market opportunities that develop as a result of technological and scientific advances. The attractiveness of the Company’s Products partly depends on the ability to continue to improve the Products. Failure to deliver such improvements in the timelines suggested may affect the Company’s business plan and ability to obtain greater market penetration, or otherwise cause the Company to lose market share.
The Company may require additional funding to develop and commercialize the Products. If the Company is unable to secure additional financing on acceptable terms, or at all, the Company may be forced to modify its current business plan or to curtail Company planned operations.
The Company anticipates incurring significant operating losses and using significant funds for marketing the Products, and manufacturing capability scaling. The Company’s existing cash resources are insufficient to finance these operations. Accordingly, the Company will need to secure additional sources of capital to develop its business and the Products as planned. In the event that the Proceeds from this Offering are not sufficient to successfully commercialize the Products, the Company may seek to secure future capital. The Company may seek substantial additional financing through public and/or private financing, which may include equity and/or debt financings, and through other arrangements, including collaborative arrangements. As part of such efforts, the Company may seek loans from certain of their executive Officers and Director(s).
If the Company is unable to secure additional financing in the near term, the Company may be forced to: (1) curtail or abandon the Company’s existing business plans; (2) default on any debt
obligations; (3) file for bankruptcy; (4) seek to sell some or all of Company assets; and/or (4) cease business operations.
If the Company is forced to take any of these steps, the Company’s Common Stock may lose significant value or become worthless.
Any future financing may result in ownership dilution to the Company’s existing Shareholders and may grant rights to future investors more favorable than the rights currently held by the Company’s existing Shareholders.
If the Company raises additional capital by issuing equity, equity-related, or convertible securities, the economic, voting and other rights of the Company’s existing Shareholders may be diluted, and those newly-issued securities may be issued at prices that are at a significant discount to current and/or then fair market value of the Shares. In addition, any such newly issued securities may have rights superior to those of the Company’s Common Stock as offered through this Offering. If the Company obtains additional capital through collaborative arrangements, the Company may be required to relinquish rights to technologies or its Products than the Company might otherwise have or become subject to restrictive covenants that may affect business.
6
Risks Related to Development and Regulatory Clearance of The Company’s Products
The FDA, other regulatory bodies and other comparable foreign regulatory authorities each have substantial discretion in the clearance process for the Company’s Products and may either refuse to consider the Company’s application(s) for review or may form the opinion after review of the data that the Company’s application is insufficient to grant clearance or authorization for commercial use.
The Company has received the necessary FDA clearances to commercialize its existing Products. However, the FDA and other comparable foreign regulatory authorities each have substantial discretion in the clearance process and may either refuse to consider the Company’s future applications or may form the opinion after review of the Company’s application(s) that the application is insufficient to allow clearance of for human health applications. Clearance procedures vary among jurisdictions and can involve requirements and administrative review periods different from, and greater than, those in United States. Moreover, any clearances that the Company obtains may not cover all of the clinical indications for which the Company will seek clearance, or could contain significant limitations in the form of narrow indications, warnings, precautions or contra-indications with respect to conditions of use. In such an event, the Company’s ability to generate revenues from the Products would be greatly reduced and its business would be harmed.
Failure to obtain clearance to market the Company product for human health applications may limit Company prospects for growth.
The Company may require clearance from the FDA and similar agencies in other countries prior to marketing new products which are under development . The Company will need to establish, to the satisfaction of those organizations, that its new Products are safe for use. Because the Company’s Products are novel and represents a departure from the status quo, the Company cannot assure Investors that the Company will receive clearance.
7
Risks Related to the Securities and the Offering
There is no current market for the Shares.
There is no formal marketplace for the resale of the Company’s Shares and none is expected to arise for the foreseeable future. Investors should assume that they may not be able to liquidate their investment.
The Offering price of the Company’s Shares was not established on an independent basis; the actual value of an investment may be substantially less than what Investor pays for the securities.
The Company’s Board of Directors established the Offering price of the Company’s Shares on an arbitrary basis. The Offering price of the Shares bears no relationship to the book or asset values or to any other established criteria for valuing Shares. Because the Offering price is not based upon any independent valuation, the Offering price may not be indicative of the proceeds that an Investor would receive upon liquidation. Further, the Offering price may be significantly more than the price at which the Shares would trade if they were to be listed on an exchange or actively traded by broker-dealers.
McGinley Orthopaedic Innovations, Inc. has existing Patents that the Company might not be able to protect.
The Company's most valuable assets are its intellectual property. It holds 71 United States utility patents with approximately 20 pending. It is possible that competitors may attempt to misappropriate or violate intellectual property rights owned by the Company. The Company intends to protect its intellectual property portfolio from such violations, within the constraints of available resources. It is important to note that unforeseeable costs associated with such practices may consume a significant portion of capital, which could negatively affect the Company’s research and development efforts and business, in general. However, the Company has anticipated the possible need to protect its Patents from infringement and therefore has allocated Proceeds from this Offering, in the form of Working Capital, to account for these potential occurrences.
The Company is controlled by Joseph McGinley MD/PhD, Co-Founder and the sole Director and Officer
Dr. Joseph McGinley is the beneficial owner of a majority of the Company’s Shares of Common Stock as of the date of this Offering Circular. Upon a successful Offering (where the Maximum Offering Amount is raised) Dr. Jospeh McGinley will still own a majority of the Company’s voting Shares. Investors in this Offering will not have a majority of voting Shares and therefore will not have the ability to control a vote of the Shareholders without consensus from Dr. Joseph McGinley. Dr. Joseph McGinley, therefore, has complete control as to the direction of the Company. There is a disproportionate reliance on Dr. Joseph McGinley
for the operation of the Company, and therefore a risk that the direction of the Company may change if Dr. McGinley is unable to perform his duties as Director and Officer.
Bonus Shares have a potentially dilutive effect.
As part of this Offering, the Company may offer Bonus Shares to certain investors (See “Plan of Distribution” below). There is the potential that the Company sells the Shares solely through investments subject to the award of Bonus Shares. In the event the Company sells the Shares solely through investments subject to the award of Bonus Shares, the Company could raise gross Proceeds equaling $40,000,000 million or 100% of the Maximum Offering Amount. In the event the Company sells the Shares in amounts less than the total number of Shares offered, the Bonus Shares may still have a dilutive effect to Investors in this Offering.
The Company will not absorb the cost of the issuance of the Bonus Shares; to the extent any are issued, it will not reduce the Proceeds that the Company receives. The Company will issue the Bonus Shares from its authorized Shares. The issuance of these Bonus Shares will have a maximum potential dilutive effect of 50% of the Shares offered for purchase or 3,809,523.81 Bonus Shares, meaning the company is Offering a total of 11,428,571.43 Shares through this Offering.
8
On May 13, 2022 the Company converted from a Wyoming limited liability company, McGinley Orthopaedic Innovations, LLC, to a Wyoming corporation, McGinley Orthopaedic Innovations, Inc. This conversion was executed through the filing of a Statement of Conversion with the Wyoming Secretary of State (see Exhibit 2A “Articles of Incorporation and Other Corporate Documents”). The Shareholders of the Company received a 1:1 conversion of their LLC Membership Units to Common Stock.
The proportions of ownership by the Director and Officer remained the same as prior to the conversion. However, on various dates additional Shares were accrued but not issued to the Director or Officer within the past calendar year pursuant to the table below:
| NAME | DATE ACCRUED | NUMBER OF SHARES ISSUED/ACCRUED | VALUE OF SHARES ISSUED/ACCRUED | |||||||
| Joseph McGinley | 3/31/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 6/17/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 9/24/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 12/31/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 2/17/2022 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 5/15/2022 | 51,282.05 | $ | 1.95 | ||||||
| Total Shares Issued/Accrued | 83,333.35 | $ | 162,500.03 | |||||||
All of the Shares above were accrued at the price of $1.95 per Share. When the above persons desire to have the Shares issued, they will be required to pay $1.95 to receive the Shares.
The Company may engage in other financings including future equity raises. In the event the Company sells equity securities subsequent to an Investor’s purchase of Shares through this Offering or future offerings, the Investor’s proportionate ownership of the Company will be diluted.
9
The Offering will be made through general solicitation, direct solicitation, and marketing efforts whereby Investors will be directed to the investment website (invest.mcginleyorthopaedics.com) to invest. The Company has engaged Rialto Markets, an independent FINRA broker-dealer to assist with the Share sales in exchange for a 3% commission fee on the aggregate sales. The Offering is conducted on a best-efforts basis. No Commissions or any other renumeration for the Share sales will be provided to the Company, the Directors, any Officer, or any employee of the Company, relying on the safe harbor from broker-dealer registration set forth in Rule 3a4-1 under the Securities Exchange Act of 1934, as amended.
The Company will not limit or restrict the sale of the Shares during this 12-month Offering. No market exists for the Shares and no market is anticipated or intended to exist in the near future, therefore there is no plan to stabilize the market for any securities to be offered.
Directors, Officers, and employees of the Company are primarily engaged in the Company’s business of developing, manufacturing, and selling medical devices, and none of them are, or have ever been, brokers nor dealers of securities. The Directors, Officers, and employees will not be compensated in connection with the sale of securities through this Offering. The Company believes that the Directors, Officers, and employees are associated persons of the Company not deemed to be brokers under Exchange Act Rule 3a4-1 because: (1) no Director, Officer, or employee is subject to a statutory disqualification, as that term is defined in section 3(a)(39) of Exchange Act at the time of their participation; (2) no Director, Officer, or employee will be compensated in connection with his participation by the payment of commissions or by other renumeration based either directly or indirectly on transactions in connection with the sale of securities through this Offering; (3) no Director, Officer, or employee is an associated person of a broker or dealer; (4) the Directors, Officers, and employees primarily perform substantial duties for the Company other than the sale or promotion of securities; (5) no Director, Officer, or employee has acted as a broker or dealer within the preceding twelve months of the date of this Offering Circular; (6) no Director, Officer, or employee will participate in selling this Offering after more than twelve months from the Effective Date of the Offering.
Rialto Markets LLC (“Rialto”) has agreed to act as placement agent to assist in connection with this Offering. Rialto is not purchasing or selling any securities offered by this Offering Circular, nor is it required to arrange the purchase or sale of any specific number or dollar amount of securities. However, Rialto has agreed to use their best efforts to arrange for the sale of the Shares offered through this Offering Circular. In addition, Rialto may engage other brokers to sell the securities on their behalf. Rialto will receive compensation for all passive sales of the Shares offered and sold through pursuant to this Offering at a rate of 3% of the gross Proceeds for a maximum of $900,000. The Company may pay Rialto 8% of the gross Proceeds from the sale of up to $10,000,000 in Common Stock resulting from the direct selling efforts of Rialto not to exceed $800,000.
In the event that Rialto’s targeted selling efforts lead to sales of up to $10,000,000 in Shares of Common Stock, Rialto will be entitled to 8% of the gross Proceeds from the sale of such Shares of Common Stock not to exceed $800,000. If Rialto’s efforts lead to all $10,000,000, the maximum commissions to be charged would be $1,700,000. The $1,700,000 is made up of 8% of $10,000,000, or $800,000, and 3% on the remaining $30,000,000 for $900,000. There will not be any commissions charged at a combined 11%.
The Company will also publicly market the Offering using general solicitation through methods that include emails to potential Investors, the internet, social media, and any other means of widespread communication.
The Offering Circular will be furnished to prospective investors via download 24 hours per day, 7 days per week on the Company’s website at invest.mcginleyorthopaedics.com and via of the EDGAR filing system. The following table shows the total discounts and commissions payable to Rialto in connection with this Offering by the Company.
In the event that Rialto’s targeted selling efforts lead to sales up to $10,000,000 in Shares of Common Stock, Rialto will be entitled to 8.0% of the Gross Proceeds from the sale of such shares of Common Stock not to exceed $800,000.
| Price Per Share | Total Offering | |||||||
| Public Offering Price | $ | 5.25 | $ | 40,000,000 | ||||
| Placement Agent Commissions | $ | 0.22* | $ | 1,700,000* | ||||
| Proceeds, Before Expenses | $ | 4.90 | $ | 38,300,000 | ||||
*this represents the maximum potential commissions due to Rialto, the commissions actually due may be less than this number conditional on the success of Rialto’s targeted sales efforts.
10
Other Terms
Rialto has also agreed to perform the following services in exchange for the compensation discussed above:
- Act as lead broker for the Offering, coordinating efforts of parties involved and providing regulatory guidance;
- Manage the back-end process of the Offering Platform technology that Investors use to invest in the Offering;
- Reviewing marketing materials if requested;
- Performing AML/KYC checks on all Investors, and;
- Providing other financial advisory services normal and customary for Regulation A offerings and coordinate with the Company’s registered transfer agent and legal representatives.
In addition to the commissions described above, the Company will also pay $6,500 to Rialto for out-of-pocket accountable expenses paid prior to commencing the Offering. This fee will be used for the purpose of coordinating filings with FINRA (Form 5110). In addition, the Company will pay Rialto $10,000 consulting fee upon the issuance of the FINRA No Objection Letter and a $5,000 Blue Sky filing service fee for managing the filings required for Blue Sky regulations. The Company will forward the fees required for state notice filing fees, estimated to be approximately $13,000. Assuming the full amount of the offering is raised and that Rialto's targeted selling efforts lead to sales of $10,000,000, the Company estimates that the total commissions of the Offering payable by the Company to Rialto will be approximately $1,700,000. Maximum expected out of pocket expenses total $34,500.
11
| Selling Shareholders | Common Shares Held Prior to Offering | Common Shares Offered for Sale | Common Shares Held After Offering | |||||||||
| Achreja, Jivan Singh | 148,470.35 | 113,917.72 | 34,552.64 | |||||||||
| Anderson, David | 25,641.03 | 20,512.82 | 5,128.21 | |||||||||
| Becker, Garry | 13,661.20 | 1,400.00 | 12,261.20 | |||||||||
| Bishop, David F. | 38,461.54 | 10,000.00 | 28,461.54 | |||||||||
| Biswal, Sandip | 191,380.16 | 38,276.03 | 153,104.13 | |||||||||
| Brookstein, David | 15,337.42 | 12,269.94 | 3,067.48 | |||||||||
| Brus, Cary and Karen | 110,000.00 | 40,000.00 | 70,000.00 | |||||||||
| Chan , Bryan | 10,000.00 | 8,000.00 | 2,000.00 | |||||||||
| Connor , Kathleen | 70,333.69 | 5,000.00 | 65,333.69 | |||||||||
| Cook, Thomas | 10,000.00 | 5,000.00 | 5,000.00 | |||||||||
| Cross Jr., Gregory H. | 15,337.42 | 13,803.68 | 1,533.74 | |||||||||
| Cubin, Eric | 137,356.42 | 120,888.54 | 16,467.88 | |||||||||
| Cubin, Jonna | 47,500.00 | 42,750.00 | 4,750.00 | |||||||||
| Cundy, Bradley A. | 10,928.96 | 8,743.17 | 2,185.79 | |||||||||
| Davis, Tad | 111,336.01 | 30,000.00 | 81,336.01 | |||||||||
| D'elia, Joshua D. | 32,269.94 | 29,042.95 | 3,226.99 | |||||||||
| DeVore, Robert | 27,322.40 | 10,000.00 | 17,322.40 | |||||||||
| Domsic, James D. | 46,283.54 | 25,000.00 | 21,283.54 | |||||||||
| Domsic, Joseph J. | 35,875.38 | 15,000.00 | 20,875.38 | |||||||||
| Duerloo, Brian | 34,013.60 | 15,000.00 | 19,013.60 | |||||||||
| Edwards, Bill | 13,661.20 | 6,830.60 | 6,830.60 | |||||||||
| Ekis, James L. | 12,269.94 | 6,000.00 | 6,269.94 | |||||||||
| Ellis, Dennis | 13,661.20 | 10,928.96 | 2,732.24 | |||||||||
| Emery, Richard | 21,857.92 | 17,486.34 | 4,371.58 | |||||||||
| Finch, Mark L. | 17,006.80 | 1,700.00 | 15,306.80 | |||||||||
| Flaherty, Michael | 486,072.16 | 125,000.00 | 361,072.16 | |||||||||
| French, Dan | 10,000.00 | 9,000.00 | 1,000.00 | |||||||||
| Gillem, Michael T. | 41,821.72 | 20,910.86 | 20,910.86 | |||||||||
| Giriech, Michael | 12,269.94 | 11,042.95 | 1,226.99 | |||||||||
| Goodreau, Randall G. | 15,337.42 | 7,500.00 | 7,837.42 | |||||||||
| Graham, Travis | 15,337.42 | 13,803.68 | 1,533.74 | |||||||||
| Grunfeld, Robert | 26,455.03 | 21,164.02 | 5,291.01 | |||||||||
| Hays, Michael | 22,354.50 | 11,628.84 | 10,725.66 | |||||||||
| Healey, Nicholas | 22,269.94 | 5,567.49 | 16,702.45 | |||||||||
| Higgins, Patrick | 163,934.43 | 131,147.54 | 32,786.89 | |||||||||
| Hogan, Brian | 15,337.42 | 13,803.68 | 1,533.74 | |||||||||
Selling Shareholders (continued)
| Selling Shareholders | Common Shares Held Prior to Offering | Common Shares Offered For Sale | Common Shares Held After Offering | |||||||||
| Hogan, John F. | 82,344.23 | 25,000.00 | 57,344.23 | |||||||||
| Ivanov, Oleg | 50,000.00 | 45,000.00 | 5,000.00 | |||||||||
| Jackson, Lisa | 10,000.00 | 9,000.00 | 1,000.00 | |||||||||
| King , Jackie | 17,006.80 | 15,306.12 | 1,700.68 | |||||||||
| Kozin M.D., William | 50,674.85 | 10,000.00 | 40,674.85 | |||||||||
| Larsen , Dr. Aaron | 20,000.00 | 4,000.00 | 16,000.00 | |||||||||
| Larsen , Jerrold L. | 50,000.00 | 15,000.00 | 35,000.00 | |||||||||
| Larsen , Nicholas | 20,000.00 | 5,000.00 | 15,000.00 | |||||||||
| Larsen , Ryan | 20,000.00 | 18,000.00 | 2,000.00 | |||||||||
| Leachtenauer, Paul C. | 141,601.81 | 15,000.00 | 126,601.81 | |||||||||
| Leaseke , Paul | 30,463.92 | 15,000.00 | 15,463.92 | |||||||||
| Logan, Particia | 10,000.00 | 9,000.00 | 1,000.00 | |||||||||
| Mamot, Keith | 20,000.00 | 18,000.00 | 2,000.00 | |||||||||
| Mamot, Michael | 10,000.00 | 9,000.00 | 1,000.00 | |||||||||
| Mansell, Donna J. | 13,661.20 | 10,928.96 | 2,732.24 | |||||||||
| McConnell, Rita | 13,661.20 | 1,400.00 | 12,261.20 | |||||||||
| Mills, Andy | 27,210.88 | 6,802.72 | 20,408.16 | |||||||||
| Moulds, Elizabeth | 136,612.02 | 50,000.00 | 86,612.02 | |||||||||
| Novick, Robert | 100,000.00 | 10,000.00 | 90,000.00 | |||||||||
| Patnaik, Goutam | 112,881.07 | 22,577.00 | 90,304.07 | |||||||||
| Porter , Peg Connor | 46,012.27 | 4,600.00 | 41,412.27 | |||||||||
| Poullos, Peter | 94,027.80 | 18,000.00 | 76,027.80 | |||||||||
| Power, Joanne | 19,125.68 | 15,300.54 | 3,825.14 | |||||||||
| Quinn, Brian R. | 16,802.72 | 3,500.00 | 13,302.72 | |||||||||
| Reiger, Mark A. | 13,661.20 | 10,928.96 | 2,732.24 | |||||||||
| Robinson, Jimmy D. | 22,269.94 | 20,000.00 | 2,269.94 | |||||||||
| Rudd, Gary | 20,464.48 | 6,800.00 | 13,664.48 | |||||||||
| Sadeghipour, Keyanoush | 38,201.08 | 10,000.00 | 28,201.08 | |||||||||
| Schilling, William C. | 16,802.72 | 15,122.45 | 1,680.27 | |||||||||
| Schlidt, Robert | 225,270.44 | 100,000.00 | 125,270.44 | |||||||||
| Six, Randall G. | 13,661.20 | 10,928.96 | 2,732.24 | |||||||||
| Sulser, Daniel F. | 1,827,626.37 | 100,000.00 | 1,727,626.37 | |||||||||
| Tarka, Christopher J. | 26,455.03 | 21,164.02 | 5,291.01 | |||||||||
| Tarka, Elizabeth Ann | 26,455.03 | 21,164.02 | 5,291.01 | |||||||||
| Tobin, Robert | 179,829.26 | 149,208.43 | 30,620.83 | |||||||||
| Tomlinson, David J. | 13,661.20 | 10,928.96 | 2,732.24 | |||||||||
| Toussaint, R. James | 25,641.03 | 20,512.82 | 5,128.21 | |||||||||
| Walker, Thomas J. | 17,006.80 | 15,306.12 | 1,700.68 | |||||||||
| Wells, Donald | 54,948.04 | 15,200.00 | 39,748.04 | |||||||||
| Wilkinson DPM, Michael P. | 40,000.00 | 20,000.00 | 20,000.00 | |||||||||
| Workman, Ernest | 93,926.02 | 23,962.02 | 69,964.00 | |||||||||
| Grand Total | 5,809,122.38 | 1,904,761.90 | 3,904,360.48 |
The Company is offering 1,904,761.90 Shares for the accounts of 74 Selling Shareholders, as described in the table above. This represents approximately 4.21% of the outstanding Common Shares as of the date of this Offering Circular. The Shares on account of Selling Shareholders will not be sold from the beginning of the Offering but in accordance with the schedule described below:
- The first $5,000,000 of the net Proceeds (defined below) will be allocated 100% to the Company;
- The next $5,000,000 of the net Proceeds will be allocated 80% to the Company and 20% to the accounts of Selling Shareholders who purchased Shares for cash in one or more of the Company’s prior offerings; and,
- The remaining $30,000,000 of the net Proceeds will be allocated 70% to the Company and 30% to the accounts of all of the Selling Shareholders.
The term “net Proceeds” refers to the gross Proceeds received by the Company less any commissions and other Offering fees. (See discussion on this “Plan of Distribution” above).
Bonus Share Program
Certain Investors will be eligible to receive additional Shares of Common Stock (“Bonus Shares”) depending upon the amount invested by such Investors. The Company will not absorb the cost of the issuance of the Bonus Shares; to the extent any are issued, it will not reduce the Proceeds that the Company receives. The Company will issue the Bonus Shares from its authorized Shares. The issuance of these Bonus Shares will have a maximum potential dilutive effect of 55% consisting of (1) the Bonus Shares issued pursuant to investment amount for a maximum of 50% (3,809,523.81 Bonus Shares); and, (2) Bonus Shares issued pursuant to time of investment for a maximum of 5% (380,952.38 Bonus Shares) - meaning the Company is Offering a total of 11,809,523.81 Shares through this Offering.
Bonus Shares issued pursuant to investment amount
The following table describes the ratio of Bonus Shares due to an Investor based on the size of initial investment:
| A | B | |
| Investment Amount | Ratio of Bonus Shares | |
| $5,000 - $24,999.99 | 3% | |
| $25,000- $49,999.99 | 5% | |
| $50,000 - $99,999.99 | 10% | |
| $100,000 - $499,999.99 | 15% | |
| $500,000 - $999,999.99 | 25% | |
| $1,000,000 and above | 50% | |
For clarity, the number of Bonus Shares owed to an Investor as per the table above equals the number of Shares owed to the Investor upon purchase at the stated Investment Amount ranges in column A multiplied by the percentage in column B. For example, if an Investor purchases $5,250 worth of Shares, the Investor will be awarded 30 Bonus Shares, for a total of 1,030 Shares.
Bonus Shares issued pursuant to time of investment
In addition to the Bonus Shares issued pursuant to investment amount – if an Investor invests within the first six weeks following the start of the Offering, that Investor will be granted a bonus of 5% Bonus Shares. These Bonus Shares are in addition to any Bonus Shares issued pursuant to investment amount. For example, if an Investor invests $5,250 within the first six weeks of the Offering, that Investor will receive 1,080 Shares.
12
| 25% | 50% | 75% | 100% | |||||||||||||
| 1. Marketing/Sales/Distribution | $ | 2,500,000 | $ | 3,500,000 | $ | 6,000,000 | $ | 7,000,000 | ||||||||
| 2. Finance/Administrative | $ | 300,000 | $ | 500,000 | $ | 1,000,000 | $ | 1,750,000 | ||||||||
| 3. Product Development | $ | 200,000 | $ | 500,000 | $ | 1,500,000 | $ | 1,500,000 | ||||||||
| 4. Quality/Compliance | $ | 100,000 | $ | 200,000 | $ | 500,000 | $ | 750,000 | ||||||||
| 5. Planning/Partnerships/Acquisitions | $ | 100,000 | $ | 300,000 | $ | 2,000,000 | $ | 5,000,000 | ||||||||
| 6. Investor Acquisition | $ | 1,000,000 | $ | 2,000,000 | $ | 3,000,000 | $ | 4,000,000 | ||||||||
| 7. Working Capital | $ | 4,400,000 | $ | 8,000,000 | $ | 8,000,000 | $ | 8,000,000 | ||||||||
| 8. PPE Expansion | $ | 400,000 | $ | 1,000,000 | $ | 1,000,000 | $ | 2,000,000 | ||||||||
| 9. Proceeds to Selling Shareholders | $ | 1,000,000 | $ | 4,000,000 | $ | 7,000,000 | $ | 10,000,000 | ||||||||
| Total | $ | 10,000,000 | $ | 20,000,000 | $ | 30,000,000 | $ | 40,000,000 |
Upon successful Offering whereby the Company receives gross Proceeds equaling $40,000,000, the Company expects to use the Proceeds as shown in (a) the categories defined in the left-hand column of the table above and (b) in the amounts shown in the table’s right-most column. Deployment of partial Proceeds are shown in the middle columns labeled 25%, 50%, and 75% from left to right. Investment in each of the categories and amounts shown in the table above are anticipated to be as follows.
THE COMPANY RESERVES THE RIGHT TO CHANGE ITS USE OF THE PROCEEDS AT ANY TIME, AT THE SOLE DISCRETION OF THE COMPANY, ITS DIRECTOR(S), OR OFFICERS WITHOUT PRIOR NOTICE TO SHAREHOLDERS OR POTENTIAL INVESTORS.
1. Marketing/Sales/Distribution/Customer Service $7,000,000
The Company anticipates to devote up to $7,00,000 of the Proceeds in the branding, marketing, selling, distribution, and customer service/training of its products to hospitals, surgical centers, and individual orthopedic surgeons. This will include the expansion of the Company’s sales force, support employees, educational materials, marketing materials, and online and in-person advertising.
2. Finance/Administration $1,750,000
The Company will use up to $1,750,000 of the Proceeds for staffing and equipping its finance/accounting and administrative departments. Finance and accounting will require resources in treasury, control, intangible asset accounting, external audit, and accounting systems. Administration requires investment in human resources, public relations, risk management, legal, and information technology.
3. Product Development $1,500,000
A competitive strength of the Company is its intellectual property: patents and proprietary knowhow. The Company has numerous new products in various stages of development that require investment to bring them to market. For this purpose, the Company intends to increase its current level of product development spending up to $1,500,000 of the Proceeds.
4. Quality Assurance/Compliance $750,000
Quality assurance and regulatory compliance are major responsibilities that the Company owes to its customers, government regulators (e.g., FDA), and international standards setting organizations. The strong programs already operative in the company will need to additional resources to expand as the Company grows. The Company expects to spend $750,000 on these activities.
5. Planning/Partnerships/Acquisitions $5,000,000
A key source of growth for the Company in coming years will involve licensing, product development partnerships and possible acquisitions of various technologies. The Company anticipates spending as much as $5,000,000 on these activities. No such technologies have been identified as of the date of this Offering Circular.
6. Investor Acquisition $4,000,000
The Company’s cost of presenting itself to potential Investors nationally and internationally, and of properly administering their investments in the Company, is significant. The Company expects that up to $4,000,000 of the Proceeds will be required to compensate to investment intermediaries, transfer agents, market makers, attorneys, and other advisors involved is completing a successful offering.
7. Addition to Working Capital $8,000,000
Anticipated growth in the Company’s revenue, production and marketing/selling operations, staffing, and possibly partnering/acquisition opportunities will require the Company to hold significant cash reserves. The Company expects to set aside up to $8,000,000 for cash reserves and working capital.
8. Property, Plant, and Equipment Expansion $2,000,000
As demand for the Company’s Products grows, its manufacturing facilities will also increase the size and complexity. The Company anticipates investing up to $2,000,000 of the Proceeds for plant expansion and new equipment.
9. Proceeds to Selling Shareholders $10,000,000
The Company anticipates up to $10,000,000 of the Proceeds to go to selling shareholders. See “Selling Shareholders” above.
13
Corporate History
McGinley Orthopaedic Innovations, Inc. (the “Company”) is an orthopedic surgical device/implant innovator and manufacturer based in Casper, Wyoming. The Company was founded as McGinley Orthopaedic Innovations, LLC in 2012. The Company converted to a Wyoming Corporation on May 13, 2022 by filing Articles of Incorporation and Statement of Conversion with the Wyoming Secretary of State (See Exhibit 2A “Certificate of Incorporation and Other Documents”).
Summary of the Company
The Company was established in 2012 with the goal of increasing patient safety and physician confidence through technological advances in the orthopedic field. The Company has developed and is marketing patented, FDA-cleared IntelliSense Drill Technology, a set of orthopedic power tools and accessories, and the Lever Action Plate System bone fixation implant system. The Company aims to make its growing product lines the “standard of care” in orthopedic surgery worldwide. Its mission is three-fold: increase patient safety, improve patient care, and reduce surgical cost.
The patented and proprietary sensing, navigation, and robotic capabilities of the IntelliSense Drills were cleared for commercial sale by the FDA on February 20, 2015 through clearance by FDA of the Company’s 510(k). The IntelliSense Drills exhibit capabilities far beyond those available to surgeons in the legacy devices offered by any other power tool manufacturer. “Legacy” devices is a reference to the current status quo products of the orthopedic surgery industry.
The Lever Action Plate System was cleared for commercial sale by the FDA on June 25, 2020 through clearance by FDA of the Company’s 510(k). The Company believes that the LADS will revolutionize wrist surgery and dramatically improve patient outcomes.
The Company currently owns issued or pending patents and has already attracted substantial interest from leading orthopedic device makers in the U.S. The Company is engaged in partnering discussions with several firms related to technology development, licensing, commercialization, and distribution in various segments of the national and global orthopedic power tool and implant markets.
The Company has two wholly-owned subsidiaries: DS Manufacturing LLC and McGinley Engineered Solutions, LLC. DS Manufacturing LLC, has a DBA as McGinley Manufacturing, is a wholly-owned subsidiary of the Company, and operates a custom machine and fabrication shop with a fully integrated engineering team. Capabilities include custom engineering and design, high precision turning, milling, welding, machining, replacement part fabrication, CNC plasma cutting, powder coating, as well as 5-axis capability. McGinley Manufacturing also manufactures parts of varying sizes, including small scale pieces for precision hand tools to medium-sized pieces for hard rock mining projects for the mining and oil services industries.
McGinley Engineered Solutions, LLC is a wholly-owned subsidiary of the Company and holds title to all of the patents assigned to the Company.
IntelliSense Drill Technology


IntelliSense Technology includes series of surgical drills that have proprietary sensing, navigation, and robotic capabilities far beyond those available to surgeons in the devices offered by any other surgical power tool manufacturer.
The Company’s first products utilizing IntelliSense Technology received FDA 510(k) clearance in February 2015 and has been used in over 1,000 surgical procedures at over 25 orthopedic surgical centers and teaching hospitals across the United States. The hand-held, electric, surgical drill is designed for routine use in many common orthopedic procedures that require the placement of hardware and bone drilling. Unique design features of IntelliSense Drills include (1) an ergonomic and distinctive drill housing; (2) an integrated and automated depth measurement component, a
chuck uniquely designed to engage the Company’s proprietary quick-release drill bits; (3) integrated lighting; (4) an electronic control unit that provides power, engages sensors, collects and analyzes data, and displays information in real time on a touchscreen visible to the entire surgical team. The IntelliSense Drill’s high-torque motor and a broad range depth sensor is suited to large/small bone drilling and fracture fixations. Furthermore, the IntelliSense Drill is significantly smaller, lighter, easier to use, and more accurate than other standard non-sensing orthopedic drills in service today.
IntelliSense Drill Technology has been reported by physicians to reduce the number of wasted screws, plates, and other consumables and may shorten the duration of a typical orthopedic surgery by 20 minutes or more.
Features of the IntelliSense Drill:
- The Drills include an integrated and automated depth measurement component that, together with a microprocessor, has several modes. The main feature of the IntelliSense Drill Technology is the depth measuring component that stops the Drill once the desired depth has been achieved. This significantly reduces the risk of drilling too deep into the bone and “plunging”, or drilling through the bone. The measuring device will stop the drilling automatically depending on which mode the user selects. These modes include (1) Bicortical (Drill stops upon breaching the second cortex); (2) Freehand (Drill operates as a standard orthopedic drill while including depth measurement); and (3) Set Depth (Drill stops at a predetermined depth), and Multicortex (Drill stops upon breaching a preset number of cortices).
- Orthopedic drill bits are notoriously difficult to sterilize. Minimizing infection risk depends on the consistent use of a new drill bit for each surgery. Preventing surgical site infection remains one of the key patient safety goal standards per the National Patient Safety Goals Joint Commission (NPSG.07.05.01). To assure the IntelliSense Drill bits are proprietary, the Company has designed a patented interlock sensitive to autoclave sterilization which prevents Drill bit reuse.
- LED lights mounted to the front of the Drill housing that illuminates the operative site. It can be activated throughout the procedure, only during active drilling, or turned off. Enhanced visibility is achieved without cumbersome, outside sources such as headlamps, ceiling-hung, and roll-around lights that cast shadows.
IntelliSense Drills Accessories

The accessories include a keyed chuck, keyless chuck, standard AO chuck, and a patented universal pin driver. While these chucks allow the IntelliSense Drill to be used with a wide range of generic products, the benefits of the IntelliSense Drill Technology - in particular the auto-stop and depth measurement features – will not function while using the generic products. Replacement of the Drill bits will drive a secondary and constant revenue stream for the Company.
The Equalizer®:

A companion tool to the IntelliSense Drill, the patented Equalizer, determines the size and length differences that are manufactured into plate and screw sets developed by different manufacturers. With some plate systems, the plates, screws, and depth gauges are manufactured for exclusivity within its own system. Using a depth gauge from one company will give accurate results for plates and screw sets made by that company, but may be completely unreliable for plates and screws made by a different company because of their proprietary offsets. These systems do not give an actual measurement, but only a screw size number; therefore, they are incompatible with the IntelliSense Drill, which is a true measurement device. The Equalizer was developed to eliminate the issue of manufactured plate offsets. The Equalizer can determine the offset of a plate needed for a surgical procedure, and that offset can be input to the IntelliSense Drill for compatibility with all plating systems.
The Revolver® Universal Drill Bit Guide System

The patented Revolver revolutionizes conventional drill bit guides by replacing multiple standard drill bit guides with one 10-piece kit. It features a rotating chamber that adjusts to multiple drill bit diameters from 1.0mm to 5.0mm. It has a range of attachments that extend the length of the guide up to 220mm for percutaneous applications. It has a non-slip tip providing stability on the bone and a comfortable, ergonomic handle. The surgeon’s hand placement is safely away from the drilling point. The Revolver is universal with most standard drill bits. Some of its benefits include cost savings by eliminating numerous conventional drill bit guides, ease of inventory management and sterilization, reducing surgical time in switching and locating various guides and requires little to no training.
14
IntelliSense Pins
The pin technology currently used to transfix bone for traction or fixation has remained unchanged for decades. The Company has developed a new pin design to be used with the IntelliSense Pin Pilot (a specialized drill bit), with the goal of reducing heat generation while maintaining proper fixation. IntelliSense Pins will be required when using the IntelliSense Pin Pilot.
Advantages of the IntelliSense Drill Technology
Speed of IntelliSense Drill Technology
Hospitals rigorously schedule operating rooms to maximize utilization of scarce resources. According to studies from Akron General Hospital and Northwestern Memorial Hospital, typical Level 1 Trauma Center Operating Room cost in the range of $20.93 to $97.00 per minute. According to a 2005 study of 100 U.S. hospitals, the average charge for the Operating Room was $62.00 per minute, not including extra resources specific to the procedure or provider fees and anesthesia. The time savings from the automatic measuring features of IntelliSense can add one procedure per day of operating room utilization.
An average trauma case uses 10 fixation screws. Surgeons spend one minute (or more) per screw to stop, measure and re-drill each hole (10 mins x $62/min = $620).
Competing Technology for Measuring Drilling Depth
Fluoroscopy is used to confirm screw measurements. For each image, the team must move the fluoroscope to the patient, position the machine arm over the incision, and move team members away from the radiation. Then they take the image, move the equipment back out of the way, and finally reposition the team and resume drilling. In a trauma setting, this takes approximately four to eight minutes per case; in a spine procedure, 25-40 minutes. Using the lower trauma figure, IntelliSense® will save OR time costs attributable only to fluoroscopy by approximately $496.
Less Screw Replacement
On average, when a screw is incorrectly sized, it takes five to ten minutes to remove and replace the screw and additional time to re-image the new screw, according to a surgeon at a hospital in New York. The Company estimates (statistically) the cost of screw replacement at $370 per typical trauma procedure.
Fewer Wasted Screws
Published literature indicates a screw error rate requiring removal and replacement of between 20% and 24.7% that is directly attributable to measurement error. Based on a minimum of six screws used per orthopedic surgery, the Company estimates 1 to 2 screw(s) requiring replacement per case. Fracture fixation screws cost approximately $35 to $150; uni-axial pedicle screws for certain spine surgeries cost up to $1,000 and multi-axial pedicle screws may cost up to $1,500. When surgeons recognize an incorrect screw length during surgery and replace it with one of proper length, hospitals often times pay for the error time, costing hundreds of dollars.
Mitigated Drill Plunging
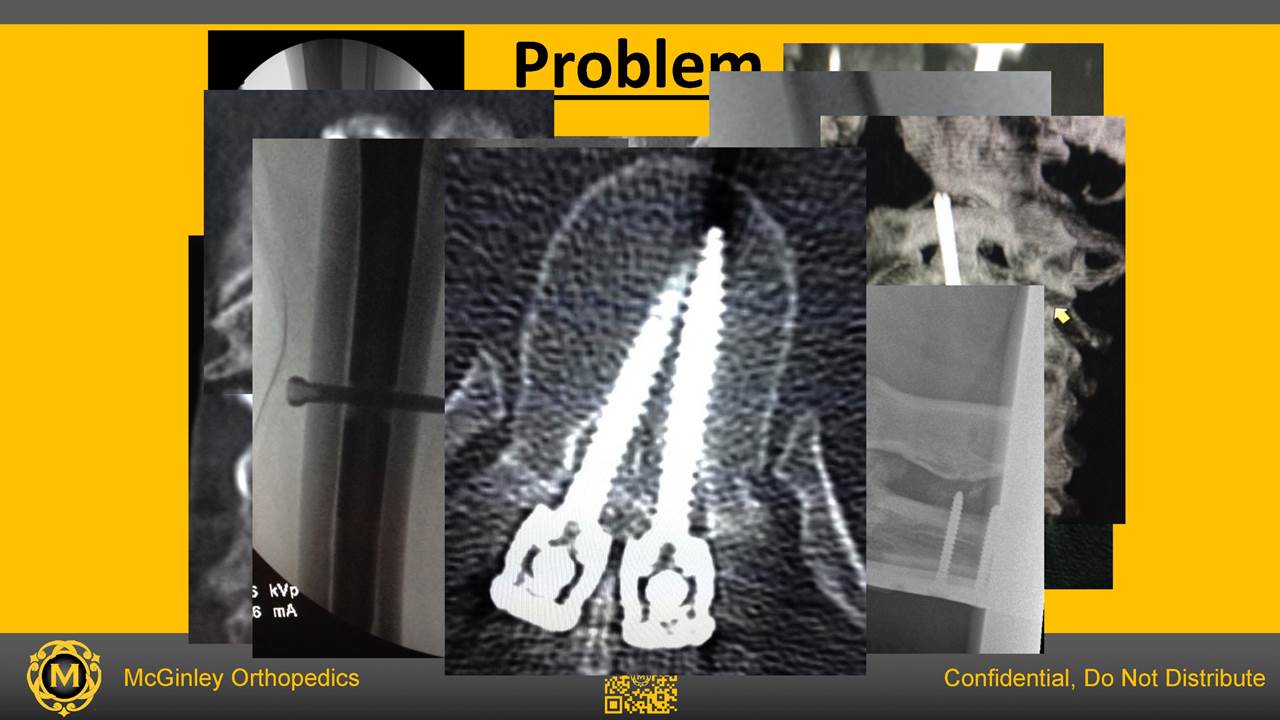
When surgeons drill too far through a bone blood vessels, nerves, tendons, and organs such as the spinal cord can be damaged. Plunging is a common and under-reported problem. In an informal experiment at a major trade show in 2015, over 150 surgeons were asked to drill holes in saw bones and avoid plunging. The surgeons averaged a 6.67mm plunge with a standard deviation of 3.32mm. Many critical organs and structures lie within 7mm of a bone. This simple experiment shows that, despite frequent denials, surgeons often plunge even when they consciously try not to. In the worst cases, plunging too far can "wind up" nerves and vessels in an instant, causing serious injury or even death
Fewer Screw Injuries
Measurement errors may result in excessively long screws that protrude through bones or short screws causing an unstable plate long screws can rub or erode tissue causing pain or harm as seen in the testimonial IntelliSense Drill commercial. Especially dangerous for patients is a spinal pedicle screw that is too long as it can compress the outer wall of the aorta threatening to erode and puncture the vessel. It is potentially fatal
Automatic and Speedy Measuring
To avoid plunging and screw length errors, orthopedic surgeons interrupt the surgical procedure several times to measure each hole. Errors in screw length are common in orthopedic surgeries. Average trauma surgeries consume up to ten screws, so interrupting the surgical process to measure hole depth consumes significant amounts of very expensive operating room time
Lever Action Plate System® for Distal Radius Fractures
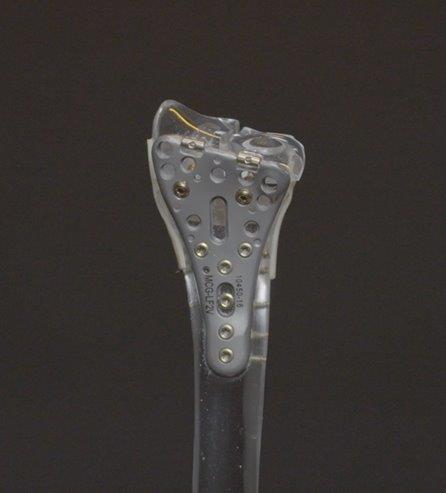
The Company’s second major product is the Lever Action Plate System for Distal Radius Fractures. The Lever Action Plate System received FDA 510(k) clearance in June 2020. The Company acquired exclusive rights to manufacture this technology along with associated issued patents.


The Lever Action Plate System® is a novel solution for distal radius fractures, a wide range of wrist fractures that affect the joint. This innovative plate system features proprietary beams that align volar tilt, an angle of the bones in the wrist. The beams are inserted into the bone fragment and with the turn of a screw, the beams and the fragment elevated into the surgeon’s desired placement. An improvement on the current, generic plates available on the market, the system has optional variable angle screws of varying sizes with patented locking technology. The contour of the plate is designed to adhere comfortably to the watershed line, a theoretical line on the radius in relation to tendons and nerves.
Since FDA clearance, Company engineers have partnered with physicians to perfect the design and bring the device to market. The plates are currently manufactured by Affiliate McGinley Manufacturing. The Company reserves the right to use third party manufacturers for the manufacture of any of the Products, including the Drills and Lever Action Plate Systems.
15
Clinical Collaborations for the Company’s Products
The Company supports and collaborates on independent and joint research projects with several academic and medical institutions across the country. Such collaborations enable the Company to get its products into the hands of leading medical researchers and refine the IntelliSense Technology. The institutions the Company collaborate with include:
- University of Wyoming;
- New York University
- Shriners Hospital for Children - Philadelphia
- Temple University
- University of Pennsylvania Medical Center - Hamot Hospital
- University of Southern California
Intellectual Property
The Company’s intellectual property assets consist of patents, FDA 510(k) clearances, proprietary knowledge in orthopedics and sports medicine, trade secrets, mechanical and electrical engineering know-how, radiology know-how, registered trademarks, and copyrights.
Patents Summary
The Company currently owns a total of 71 patents issued by United States Patent Office (“USPTO”) and foreign jurisdictions. Approximately 20 more patents are in process of preparation and filing with various jurisdictions.
The Company continues to invest significantly in (i) research & development directed at solving longstanding problems in orthopedic surgery, and in (ii) protecting its discoveries and innovations via the patent process.
FDA 510(k) Clearances
The Company has applied for and received two 510(k) clearances from the FDA. These 510(k) clearances were for the IntelliSense Drills and accessories and (ii) the Lever Action Plate System bone fixation implant system.
Patent/Product Categories
The Company’s issued and pending US patents, and their foreign registrations, can be grouped into six product categories or families. These categories, and the number of the Company’s US and foreign registrations that are core technologies for each, are as follows:
· IntelliSense Drill and Related Systems/Improvements/Features 39
· Orthopedic Implants 9
· IntelliSense Pin Pilot, Driver and Accessories 10
· IntelliSense© Bone Saw and Related Systems/Improvements/Features 8
· High Speed Burr 3
· Hand Held Surgical Device Navigation System 2
Total Issued Patents: 71
16
Affiliates
DS Manufacturing LLC d/b/a McGinley Manufacturing
In April of 2015, the Company acquired DS Manufacturing LLC, its primary supplier of machined parts and conducts business as, by filing a “doing business as”, McGinley Manufacturing LLC (“McGinley Manufacturing”). McGinley Manufacturing has ten (10) employees and revenue exceeding $1.0 million. McGinley Manufacturing operates a custom machine and fabrication shop with a fully integrated engineering team. Capabilities include custom engineering and design, high precision turning, milling, welding, machining, replacement part fabrication, CNC plasma cutting, powder coating, as well as 5-axis capability. McGinley Manufacturing also manufactures parts of varying sizes, including small scale pieces for precision hand tools to medium-sized pieces for hard rock mining projects for the mining and oil services industries.
McGinley Engineered Solutions LLC
McGinley Engineered Solutions, LLC is a wholly-owned subsidiary of the Company, and holds title to all of the patents used by the Company. This is the sole purpose of McGinley Engineered Solutions LLC.
TriOpportunity Investment Group LLC
TriOpportunity Investment Group LLC is owned by Dr. Joseph McGinley and his wife, Diane McGinley. TriOpportunity Investment Group LLC is controlled by Dr. Joseph McGinley. TriOpportunity Investment Group LLC owns the building from which the Company leases its offices.
17
Markets for Company Products
IntelliSense Drill Technology Market
The Company’s market opportunity exists in the over five million orthopedic surgeries performed annually in the United States alone, a number that grows each year. Each of these surgeries is time consuming, costly, and potentially risky for patients and surgical teams. In 2019, orthopedic surgeries consumed over six million drill bits worldwide (approximately four million in the U.S.), at a value exceeding $650 million. Sale of consumables associated with orthopedic surgeries (drill bits, pins, saw blades, high speed burrs), estimated to be 10.1 million in 2021, are expected to grow in parallel with drill sales to 14.5 million by 2027. The Company estimates U.S. sales of power drills – relatively low in volume but high in margin – to be approximately 24,000 units in 2020. The bulk of current annual sales volume is driven by replacement or updating of old equipment, and by restocking of consumables such as drill bits, pins, saw blades, burrs, and batteries. The Company’s global unit sales volume should increase as the IntelliSense Products are adopted by hospitals as the necessary standard of care in orthopedic surgery and the obsolete devices currently in use are replaced. The Company’s IntelliSense Products address a market in the U.S. alone exceeding $1 billion annually.
Customer Profile
The Company will market the IntelliSense Drills to orthopedic surgeons, surgical centers, and hospitals throughout the United States and globally. The main value proposition derives from the increased safety of using the IntelliSense Drill Technology versus the current ‘dumb’ drills which are the current status quo.
Studies show that waste of implants (primarily plates and fixation screws) occurs in 12% to 30% of orthopedic surgical cases at a cost exceeding $162 million annually (U.S.). Globally, preventable orthopedic surgical errors cost in excess of $1.5 billion per year. This cost falls on patients, health care providers, and health insurers. The sensors, software, and real-time monitoring and measurement capabilities of IntelliSense Drills may dramatically reduce orthopedic surgical errors, waste, and their associated economic costs.
The IntelliSense Drill Technology assists surgeons and patients in numerous ways. It may possibly make trauma and spine surgeries safer and less costly by (i) reducing the risk of surgeons drilling through bones (i.e. “plunging”) into patient’s arteries, veins, nerves, and vital organs; (ii) giving surgeons precise depth measurement and accurate screw sizing in real time; (iii) reducing or eliminating the need for x-rays during surgery, and (iv) precisely recording data for each bone
penetration drilled by the surgeon. Independent studies show that use of IntelliSense Drill Technology can reduce the cost of orthopedic surgery by approximately $1,850 per procedure.
In contrast to IntelliSense Drills, conventional orthopedic power tools and current “standard of care” surgical procedures are obsolete. Conventional devices lack the sensors, software, real-time data recording, and artificial intelligence embedded in Company devices. Existing “standard of care” orthopedic surgical practices force surgeons to rely on “feel” to avoid plunging into the patient’s blood vessels, nerves, tendons and other structures and organs.
18
Lever Action Plate System Market
The LAPS is a major advance in the repair of complex wrist fractures. This innovative plate system features proprietary beams that align volar tilt, an angle of the bones in the wrist. In the global plates and screws market, plates account for 83%, or $4.3 billion in 2020 and are expected to reach $5.6 billion by 2027.
Wrist fractures are the most common upper extremity fracture experienced in the U.S. population. The market size of volar distal radius plates is estimated to by 1.1 million units with a CAGR of 5.8% over the next five years. Volar distal radius plates make up 92% of the market, while dorsal plates are only 8%.
This innovative technology addresses a real and prevalent need in orthopedics. There are around 67 upper extremity fractures per 10,000 people annually in the U.S.; 25% of these fractures occur in the distal radius and ulna. In addition to the ability to restore volar tilt, the Lever Action Plate System has optional variable angle screws with patented locking technology. This hybrid type of plate and screw system gives surgeons flexibility to decide which product type that best suits the needs of the patient.
The global market for fracture fixation products, including plate systems, is estimated to reach $12.1 billion by 2025. This estimate is driven by an increasing incidence of fractures in the elderly population and a rapid adoption of internal fixation devices for small bones. The fracture fixation market is made up of both external and internal fixation devices, which include frames, pins, plates, intramedullary nails, cables, staples, and wires.
The leading competitors for the fracture fixation market are Johnson & Johnson, Stryker, Zimmer Biomet, and Smith & Nephew. Implanted medical devices are one of the most profitable businesses in the domestic healthcare industry.
Distal radius fractures account for about 20% of all fractures treated in emergency departments and 8% to 15% of all bony injuries in adults. A complication rate of 15% has been reported with traditional volar plating and post-op complication rates can be as high as 80%.
Distribution of the Company’s Products
The Company’s marketing and sales efforts are focused on (i) building the Company’s sales “funnel”; (ii) empowering patients to advocate directly for IntelliSense Technology and LAPS use;
(iii) rolling out a nationwide team of experienced medical device sales representatives; and, (iv) adding distributors to support the nationwide sales team.
The Company’s management and nationwide sales and distribution team have established a large number of evaluation-for-purchase trials at leading orthopedic hospitals and medical schools across the country. The key selling points emphasize clinical, economic, and patient outcome benefits to the hospital, surgeons, and staff.
To date, the Company has focused its sales efforts on teaching institutions and hospitals with the most reputable surgeons. Leading orthopedic surgeons are demonstrating IntelliSense and LAPS Products to residents, fellows, and interns, thus building future customers. Several of these surgeons have become champions of the Products and have aided in word-of-mouth efforts.
The Company will use the Proceeds to focus its sales and distribution team on national group purchase organizations (“GPOs”) and ambulatory surgery centers, the most rapidly growing segment among providers of orthopedic surgery.
The Company uses a traditional regional sales and distribution strategy to affect sales of its Products. The Company has four regional
representatives on its internal sales team. The Company will continue to recruit qualified individuals to the sales team in coming
quarters. As sales opportunities in discrete geographic area increase, additional distributors will be brought on board.
The Company has priced the its Products competitively with industry standards. In addition to outright sale of the IntelliSense Drills, the Company offers hospitals convenient lease-to-own and per-use rental arrangements that eliminate the need for capital purchases that must endure the time-consuming consideration approval of the hospitals’ capital acquisition committees prior to purchase.
19
Announced Products
The Company is developing new products based on its issued and pending patents that will, when introduced in the market over the coming five years, greatly increase the Company’s market opportunity and growth rate. These new products (and the product families of which they are a part) include the following:
· IntelliSense Drill for Spine (Drill)
• IntelliSense Drill with Navigation (Navigation)
• IntelliSense MicroDrill with Navigation (Drill)
· Lever Action Plates® for Tibial Plateau Fracture (Implants)
• Independent Navigation for Orthopedic Devices (Navigation)
• IntelliSense Bone Saw (Saw)
· Ankle Fixation Plate System (Implants)
The Company has announced the above products to the general public. As of the Date of this Offering Circular, none of these announced products are ready for commercial distribution.
Several of these following products either do not require 510(k) clearance or they will be covered under the Company’s current 510(k) as predicate devices.
IntelliSense Cannulated Drill Bit: Cannulated drill bits provide the surgeon with the ability to drill a hole over a guide wire to help insure proper trajectory. These drill bits are often used in lower extremity procedures and will provide expanded use of the IntelliSense Drill Technology. The Company anticipates that this product will be commercially available in Q4-2022.
IntelliSense Pin Pilot: The Pin Pilot revolutionizes pin/wire drivers and employs technological advances for pin and K-wire placement. With IntelliSense Drill Technology, this device can auto-stop after breaching a predetermined number of cortices, subchondral or endosteal, while simultaneously providing depth measurement and cortical edge detection.
The Company anticipates this product will be covered by the Company’s existing 510(k) for the IntelliSense Drill. The Company anticipates that this product will be commercially available in Q4-2022.
IntelliSense Spine (Specialized Products and Software): IntelliSense Spine embodies patented and proprietary algorithms in software controlling the IntelliSense Drill and adapts the latter to surgery or the spine. The IntelliSense Drill Technology will include four activities, three spine locations, and six modes of operation, including endosteal and subchondral modes. The Company believes that no 510(k) is needed for this product and anticipates commercial sales to begin in 2023.
IntelliSense Drill and MicroDrill with Navigation: IntelliSense Drill with Navigation will provide the surgeon real-time point-of-contact navigation. This navigation system may also be an accessory to conventional drills. The MicroDrill with Navigation is a miniaturized device employing all features of the IntelliSense Drill plus revolutionary navigation software, for use in specialized orthopedic surgeries requiring the utmost delicacy and precision (e.g., fingers and spine surgeries).
The Company anticipates this product may be covered by the Company’s existing 510(k) for the IntelliSense drill. In the event the current 510(k) does not cover this product as a predicate device, the Company will submit a new 510(k) application prior to commercial sales. The Company anticipates that this product will be commercially available in 2024.
IntelliSense Bone Saw: The IntelliSense Bone Saw employs Company patented and proprietary technology in an orthopedic saw device. This product addresses the pressing need for greatly improved precision in joint replacement surgeries.
The Company anticipates this product may be covered by the Company’s existing 510(k) for the IntelliSense drill. In the event the current 510(k) does not cover this product as a predicate device, the Company will submit a new 510(k) clearance application prior to commercial sales. The Company anticipates that this product will be commercially available in 2024.
Ankle Fusion Implants: Ankle fusion is a surgical procedure used to treat ankle pain due to severe degenerative arthritis. This surgery involves removing the inflamed cartilage and surgically joining
two or more ankle bones together. The Company is working with a partner surgeon in the early stage of product development for a unique product that can be used for these procedures.
The Company does not believe any 510(k) will cover this product as a predicate device. The Company will submit a new 510(k) clearance application prior to commercial sales. The Company anticipates that this product will be commercially available in 2024.
Special Characteristics of the Company’s Operations and Competing Products/Procedures
The Company is subject to regulation of its Products before, during, and after clearance by the FDA Products for commercial distribution of the Products
20
Facilities Regulation
Regulation of the Company as a Device Manufacturer
The Company may outsource some of manufacturing of the Company’s Products, in part, to third party manufacturers.
Quality System Regulation and Good Manufacturing: Medical Devices
After clearance of the Products by the FDA through the 510(k) process, the Company will be subject to FDA Quality Systems Regulation (“QS Regulation”) and Good Manufacturing Practices” (“GMPs”). Manufacturers must establish and follow quality systems to help ensure that their medical device products consistently meet applicable requirements and specifications known as current good manufacturing practices. Continued Compliance with these QS Regulations and GMPs is an ongoing duty of the Company. On February 23, 2022, the FDA proposed a regulation which would incorporate the international standard specific for medical device quality management systems set by the International Organization for Standardization (“ISO”), ISO 13485:2016 Medical Devices – Quality Management Systems – Requirements for Regulatory Purposes. The Company intends to use contract manufacturers and McGinley Manufacturing for manufacturing of its Products. All of these contract manufacturers will be required by the Company to be ISO 13485:2016, and thus compliant with FDA GMP.
The Company does not anticipate that this proposed regulation will affect the operations of the Company, since the Company intends on complying with ISO 13485:2016 prior to the proposed rule change in development of its facilities.
The Company has also directed McGinley Manufacturing to maintain certification with AS 9100-Aerospace Management Standard.
Employees
The Company, not including its subsidiaries, has nine (9) full time employees and one (1) part-time employee.
21
MANAGEMENT’S DISCUSSION AND ANALYSIS OF THE FINANCIAL CONDITION OF THE ISSUER
The following is a discussion of the financial condition of the Company. On May 13, 2022 the Company executed a conversion from McGinley Orthopaedic Innovations, LLC, a Wyoming limited liability company, to McGinley Orthopaedic Innovations, Inc., a Wyoming corporation. This conversion occurred after the end of FY2021 on December 31, 2021. Therefore, all figures and disclosures in this section will be those of McGinley Orthopaedic Innovations, LLC. For this reason, references to the “Company” will include McGinley Orthopaedic Innovations, LLC and McGinley Orthopaedic Innovations, Inc. only throughout this section.
Components of Results of Operations
The following discussion of the Company’s financial conditions and results of operations should be read in conjunction with the financial statements and the related notes included elsewhere in this Offering Statement. The following discussion contains forward-looking statements that reflect the Company’s plans, estimates and beliefs. The Company’s actual results could differ significantly from those discussed in the forward-looking statements. Unless otherwise indicated, the latest results discussed below are as of December 31, 2021.
Revenue/Net Loss from Operating Activities
The Company is, and has been, solely focused on development and commercialization of its Products and development of new related products. As of the date of this Offering Circular, the Company has never been profitable.
In 2021 the Company’s revenues were $1,352,296, an increase of $335,000 (33%) over revenues of $1,017,289 in 2020. The reason for this increase was due to medical sales and mining sales increasing after the economic effects caused by COVID-19 recovered, and the launch of the LAPS product line.
In FY2021 the Company had a net loss of $2,857,806. In FY2020 the Company’s net loss was $2,199,694. Net loss increased in 2021 by $658,000 (33%) over 2020. The reason for this increase was due to operating expenses increasing by $837,000 (26%) and other income being reduced by $49,000 (11 %).
Loss from Operations increased to $3,273,899 in 2021 from $2,664,432 in 2020, an increase of $613,000 (23%). The reason for this increase was due to operating expenses increasing by $837,000.
Operating Expenses
The $613,000 increase in Operating Loss in 2021 over 2020 was driven primarily by an increase of $837,000 in Selling, General and Administrative expenses (“SG&A”) in 2021 over 2020. The Company classifies its SG&A expenses as wages and benefits, general office, research and
development, professional fees, rent, insurance, advertising, other expenses, dues and subscriptions, depreciation and amortization, taxes and licenses, computer and internet, and postage and shipping. Over 80% of SG&A in 2021 was accounted for by wages and benefits, general office, Research & Development, professional (legal and accounting) fees, and travel and entertainment. The reason for this increase in SG&A expenses was due to an increase in the in-house sales team, rent and insurance for a new location and 2020 spending being generally lower due to Covid-19.
Wages and Benefits
In FY2021, the Company spent $2,338,656 on wages, benefits, contractor and other compensation expenses. In FY2020, the Company spent $1,890,629 on wages, benefits, contractor and other compensation. The increase of $448,000 (24%) in 2021 over 2020 was driven primarily by enlargement of the sales force. The Company expects that significantly increased spending in all personnel categories – particularly in marketing and selling expense – will follow a successful Offering by the Company, as discussed in Use of Proceeds discussed elsewhere in this document.
General Office Expenses
In FY2021, the Company spent $494,047 on general office expenses. In FY2020, the Company spent $398,757 on general office expenses. The increase of $95,000 (24%) in 2021 over 2020 was driven primarily by (1) a $15,000 increase in office supplies related to the Company’s new office location; (2) a $41,000 increase in tradeshow spending due to COVID-19 restrictions being lifted; and, (3) $42,000 used in recruiting a Vice President of Sales.
Research and Development Expense
The Company’s research and development efforts are focused on the continued development of new products related to the Company’s IntelliSense Technology and LAPS Products. Research and development expenses consist primarily of materials, equipment, and manufacturing costs to design, prototype, develop, and test the Company’s new products, and to acquire the parts, tools and equipment needed to do so.
In FY2021, the Company spent $305,331 on research and development expenses. In FY2020, the Company spent $353,460 on research and development expenses. The decrease of $48,000 (14%) from 2020 to 2021 was due primarily to the need to reallocate resources to the Company’s enlarged sales force.
The Company anticipates that annual increases in R&D expenses will resume upon a successful Offering by the Company, as discussed in Use of Proceeds discussed elsewhere in this Offering Circular.
Professional fees
In FY2021, the Company spent $249,910 on professional fees. In FY2020, the Company spent $184,685 on professional fees. The increase of $65,225 (35%) in 2021 over 2020 was driven
primarily by an increase in spending for the contract CFO, patent attorney, and engineering/quality assurance programs. The Company anticipates that annual increases in professional fees will continue upon a successful Offering by the Company, as discussed in Use of Proceeds discussed elsewhere in this document
Travel and Entertainment
In FY2021, the Company spent $148,282 on travel and entertainment. In FY2020, the Company spent $63,423 on travel and entertainment. The increase of $84,850 (234%) in 2021 over 2020 was driven primarily by primarily by an increase in the internal sales force and Covid 19 restrictions being lifted. The Company anticipates that increases in travel and entertainment professional fees will continue upon a successful Offering by the Company, as discussed in Use of Proceeds discussed elsewhere in this document.
Membership Units/Accrued Membership Units
Between August 15, 2019 and March 31, 2022, the Company issued 573,633.16 Membership Units and accrued 353,846.15 Accrued Membership Units for future issuance to members of the Company’s Board of Advisors, pursuant to Board of Advisor agreements executed by each Board of Advisors member. These Units are discussed in the Company’s financial statements in Note I to the balance sheet.
Liquidity and Capital Resources
As of December 31, 2021, the Company had cash of $1,624,273, working capital of $3,067,379, and total assets of $4,983,251. On December 31, 2020, the Company had cash of $2,026,934, working capital of $3,368,779, and total assets of $5,121,205. From December 31, 2020 to December 31, 2021, cash decreased by $402,661 (20 %), working capital decreased by $301,400 ( 9 %), and total assets decreased by $ 137,954 (3 %).
The Company has a $1,000,000 line of credit with a bank which matures February 8, 2024. Interest is subject to change on a monthly basis, calculated at the prime plus a variable rate (3.50% as of December 31, 2021). There was no outstanding balance on the line of credit as of December 31, 2021 and 2020.
The Company has additional capital requirements during the current fiscal year 2022 to be used in expanding its marketing and sales team and program, adding additional skills to its management team, and undertaking other initiatives as summarized in “Use of Proceeds” above. The Company is seeking additional capital via the sale of its securities in this Offering. The Company’s Offering is for a maximum of 7,619,047.62 Shares (not including Bonus Shares) at price of $5.25 per Share, with potential aggregate gross Proceeds of $40,000,000.
There can be no assurance that the Company will be successful in acquiring additional funding at levels sufficient to fund its future capital requirements. If the Company is unable to raise additional capital in sufficient amounts or on terms acceptable to it, the Company may have to significantly reduce, delay, scale back or discontinue the development of its technologies and patents or discontinue operations completely.
22
The Company owns real property through its wholly-owned subsidiary DS Manufacturing d/b/a McGinley Manufacturing. The property is an 8400 sq.ft. manufacturing plant located at 585 E Birch St Glenrock, WY 82637. The plant is a highly-specialized and highly-technical manufacturing facility capable of manufacturing all of the Company’s Products. In some occurrences, the Company must use third-party contract manufacturer for processes such as heat treating, anodizing, and heat treatment. The facility is and will continue to be ISO 13485:2016 and AS 9100 certified.
23
DIRECTORS, OFFICERS AND SIGNIFICANT EMPLOYEES
Directors
| Name | Position | Age | Term of Office | Approximate Hours per week | ||||||||||
| Dr. Joseph McGinley | Director | 48 | June 2012 - Present | Full Time | ||||||||||
Officers
| Name | Position | Age | Term of Office | Approximate Hours per week | ||||||||
| Dr. Joseph McGinley | CEO/President/Treasurer/Secretary | 48 | June 2012 - Present | Full Time | ||||||||
Significant Employees
| Name | Position | Age | Term of Office | Approximate Hours per week | ||||||||||
| Dr. Richard C. McGinity | Director of Finance | 78 | July 2016 - Present | Full Time | ||||||||||
| Diane McGinley | Director of Operations | 46 | June 2012 - Present | Full Time | ||||||||||
| Ben Warren | Vice President- McGinley Manufacturing | 55 | June 2015-Present | Full Time | ||||||||
| Adam Johnson | Director of Engineering | 46 | June 2012- Present | Full Time | ||||||||
| Michael Hays | Manager of Finances | 58 | June 2015 | Full Time | ||||||||
| Travis Sides | Operations Manager | 48 | August 2015-Present | Full Time |
24
Directors/Officers
Dr. Joseph McGinley: Founder, Chairman of the Board of Directors, Chief Executive Officer, President, Treasurer, Secretary
Full Time
Duties of Dr. Joseph McGinley as CEO/President, Treasurer and Secretary
The duties of the CEO and President are set by the Bylaws.
The President is the Chief Executive Officer and President of the Company, subject to the control of the Board of Directors, has responsibility for the conduct and management of the business and fiscal affairs of the Company and the general supervision of its property, business interests, and agents. The President, or a person designated by the President, must preside at all meetings of the Shareholders and Directors, unless otherwise ordered by the Board of Directors. The President may sign, with the Secretary or any other proper officer of the Company authorized by the Board of Directors, certificates for Shares of the Company and deeds, mortgages, bonds, contracts, or other instruments that the Board of Directors has authorized to be executed, except in cases where the signing and execution thereof is expressly delegated by the Board of Directors or the Bylaws to some other Officer or agent of the Company, or is required by law to be otherwise signed or executed.
The Treasurer is responsible for the administration of the financial affairs of the Company and has charge and custody of, and is responsible for, all funds and securities of the Company. The Treasurer also gives receipts for moneys due and payable to the Company from any source whatsoever, and deposits all such money in the name of the Company in such banks, trust companies, or other depositories as selected by the board of directors, and, if required by the Board of Directors, will give a bond for the faithful discharge of the Treasurer’s duties in such sums and with such security or surety as the Board of Directors may require.
The Secretary keeps or causes to be kept a book of minutes of all meetings of Directors and Shareholders, a stock transfer book, or a duplicate stock transfer book, showing the names of the Shareholders and their addresses, the number and classes of Shares held by each, the number and date of certificates issued for such Shares, if any, and the number and date of cancellation of certificates surrendered for cancellation, if any. The Secretary also gives or causes to be given notice of the meetings of the Shareholders and Board of Directors as is required by the Bylaws, and, when requested or required, certifies any records of the Company.
Significant Employees
Dr. Richard McGinity: Director of Finance
Dr. Richard McGinity joined the Company as Director of Finance in July 2016, and is a member of its Board of Advisors. Dr. McGinity holds degrees from Princeton University (AB), Harvard University (MBA, DBA), and is President Emeritus of the University of Wyoming. Prior to joining the University of Wyoming faculty in 2007 as Daniels Chair of Business Ethics, Dr. McGinity was a general partner in a Boston-based venture capital fund and subsequently launched his own investment banking firm serving technology firms and their founders. He has extensive public and private transaction structuring and execution experience, has served on numerous boards of publicly-traded and private companies in the technology, energy, and banking industries, and is an “audit committee financial expert” as defined in Sarbanes-Oxley Section 407. He deployed three times to Vietnam as a Navy pilot, earning two Air Medals.
Duties of Dr. Richard McGinity as Director of Finance
The duties of the Director of Finances of the Company include oversight of the three corporate functions of (i) accounting and control, (ii) economic strategy and forecasting, and (iii) investor and financial community relations. The latter includes compliance with financial accounting standards, all financial disclosure and regulatory reporting requirements, and state and federal tax regulations. The Director of Finance serves at the direction of the CEO and President Dr. Joseph McGinley.
Diane McGinley M.S.: Director of Operations
Diane McGinley joined the Company in 2012 upon the Company’s inception. She is a graduate of Ohio University, with an M.S. in Education from Gwynedd Mercy College. Her early career was as an educator, curriculum director, and instructional facilitator. She has experience in corporate finance and governance as a board member of Hilltop Bank in Casper, WY, in which she serves as a member of seven committees: Assets and Liabilities, Trust, Trust Audit, Board Credit, Compensation, Marketing and Building.
Duties of Diane McGinley, MS
Diane McGinley oversees human resources, general operations and marketing and those departments’ personnel.
Ben Warren: Vice President of Manufacturing, McGinley Manufacturing
Ben Warren has more than 22 years of experience in the machining industry. He established DS Manufacturing, Inc. in 2009 to serve the hard rock mining industry with after-market parts. In 2015, he partnered with the Company to form McGinley Manufacturing, a full-service machining and manufacturing facility for the medical, oil and gas, and mining industries. Ben now oversees all manufacturing operations for the Company at its plant in Glenrock, WY.
Duties of Ben Warren as Vice President of Manufacturing, McGinley Manufacturing
The duties of the Vice President of McGinley Manufacturing are to oversee production, operations, and personnel at the facility. He is to ensure that quality systems certification requirements are being met and followed. He is in charge of ensuring quality of products, meeting OSHA requirements for safety and is the direct supervisor to the manufacturing operations manager.
Adam Johnson: Director of Engineering
Adam Johnson has over 22 years of experience in custom engineering design and manufacturing. His experience includes positions in production facility development, machine design, composite lumber manufacturing, business development, and continuous improvement training. He holds a Bachelor of Science degree in Mechanical Engineering from Colorado State University.
Duties of Adam Johnson as Director of Engineering
The duties of the Director of Engineering include overseeing the engineering department. He is responsible for creating engineer work flows, document submissions to the FDA, ensuring adherence to the quality system and managing personnel. He also oversees product specialists and their work in the field with end user customers.
Michael Hays: Manager of Accounting and Finance
Michael Hays joined the Company in 2015 and has over 32 years of experience in corporate accounting, with over 19 years in the manufacturing industries and more than five years in healthcare accounting. He holds a degree in finance from the University of Wyoming.
Duties of Michael Hays as Manager of Accounting and Finance
The duties of the Manager of Accounting and Finance include responsibility for the Company’s accounting, treasury, and bank relationship activities.
Travis Sides: Manufacturing Operations Manager
Tavis Sides joined the Company in 2020 and has over 17 years of sales, safety, and quality experience. He enjoys challenging work and has worked on many projects that improved work conditions for his team members. He graduated with an Associates degree in Psychology from Casper College in 2000.
Duties of Travis Sides as Manufacturing Operations Manager
The duties of Manufacturing Operation Manager include creating a production schedule, monitoring and adjusting manufacturing work flows, customer relationships including recruiting new customers and overseeing machinists and other facility personnel. This position has responsibility roles in the quality system certifications.
Family Relationship Disclosure
Dr. Joseph McGinley and Diane McGinley are husband and wife.
25
COMPENSATION OF DIRECTORS AND OFFICERS
Directors
| Name | Capacity of Compensation | Cash Compensation | Other Compensation | Total Compensation | ||||||||||||
| Dr. Joseph McGinley | Director | $ | 20,000 | $ | 162,500.03* | $ | 182,500.03 | |||||||||
Officers
| Name | Capacity of Compensation | Cash Compensation | Other Compensation | Total Compensation | ||||||||||
| Dr. Joseph McGinley | CEO, President, Treasurer, Secretary | $ | 25,000 | $ | 0 | $ | 25,000 | |||||||
Dr. Joseph McGinley’s compensation in his capacities as an Officer and as a Director are segregated and are Dr. Joseph McGinley’s compensation in his capacities as an Officer and as a Director are segregated and are reflected in the above tables.
*The Director accrued the following Shares that are accrued but not issued to the Director within the past calendar year pursuant to the table below:
| NAME | DATE ACCRUED | NUMBER OF SHARES ISSUED/ACCRUED | VALUE OF SHARES ISSUED/ACCRUED | |||||||
| Joseph McGinley | 3/31/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 6/17/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 9/24/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 12/31/2021 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 2/17/2022 | 6,410.26 | $ | 1.95 | ||||||
| Joseph McGinley | 5/15/2022 | 51,282.05 | $ | 1.95 | ||||||
| Total Shares Issued/Accrued | 83,333.35 | $ | 162,500.03 | |||||||
All of the Shares above were accrued at the price of $1.95 per Share. When the above persons desire to have the Shares issued, they will be required to pay $1.95 to receive the Shares.
Aggregate Compensation to Directors
$207,500.03
There are no anticipated increases to compensation for Directors and Officers planned by the Company at this time.
SECURITY OWNERSHIP OF MANAGEMENT AND CERTAIN SECURITYHOLDERS
| Title of Class | Name and Address of Beneficial Owner | Amount of Shares Owned | Amount and nature of beneficial ownership acquirable | Percent of Class | ||||||||||
| Common Stock | Dr. Joseph McGinley | 30,267,901.81 | 83,333.35* | 67.05% | ||||||||||
(1) Dr. Joseph McGinley 234 E. 1st St. Suite 242 Casper, WY 82601. None of the Shares beneficially owned or acquirable by Dr. Joseph McGinley are being offered for sale in this Offering.
* Dr. Joseph McGinley is the beneficial owner of these Shares pursuant to accrual of those Shares (See “Dilution” above)
26
INTEREST OF MANAGEMENT AND OTHERS IN CERTAIN TRANSACTIONS
Related Party Operating Lease
TriOpportunity Investment Group LLC is owned by Dr. Joseph McGinley and his wife, Diane McGinley. TriOpportunity Investment Group LLC is controlled by Dr. Joseph McGinley. TriOpportunity Investment Group LLC owns the building from which the Company leases its offices. The Company is a tenant in this building located at 234 E. 1st St. Suite 242 Casper, WY 82601. The Company has payables due to TriOpportunity Investment Group LLC
The Company has an operating lease for office space from TriOpportunity Investment Group LLC which began in January 2021. The lease is for seven years and expires in December 2027. Rent expense is recognized on a straight-line basis over the term of the lease. The lease is also subject to a three percent minimum annual increase. In connection with the related party lease, approximately $102,000 was paid to the lessor for specific leasehold improvements requested by the Company.
Future minimum rental payments under non-cancellable operating leases (with initial or remaining lease terms in excess of one year) for each of the five years subsequent to December 31, 2021 are:
2022 - $113,221
2023 - $113,280
2024 - $113,341
2025 - $113,403
Thereafter - $113,467
Total $ 566,712
Total combined rent expense associated with the operating leases amounted to approximately $114,000 and $0 for the years ended December 31, 2021 and 2020, respectively.
27
The Company has one class of authorized stock, Common Stock. The following is a description of some of the rights associated with the Common Stock.
Dividends
The Board may authorize, and the Company may make, distributions (including dividends on its outstanding shares) in the manner and on the terms and conditions provided by law and the Articles of Incorporation.
Before payment of any dividend, there may be set aside out of any funds of the Company available for dividends such sum or sums as the Board, in their absolute discretion, think proper as a reserve or reserves to meet contingencies, or for equalizing dividends, or for repairing or maintaining any property of the Company, or for such other purpose as the Board shall think conducive to the interests of the Company.
Voting
Shareholders are only permitted to vote on the following matters:
- Removal of a Director
- Election of Directors
- Termination and dissolution of the Company
- Amendment of the Articles of Incorporation
- Any other matter requiring a vote of the Shareholders in the Articles of Incorporation or the Wyoming Business Corporation Act
Each Share is entitled to one vote.
Preemptive rights
There are no preemptive rights for Shareholders of the Company. This includes no right to purchase any classes of stock, option, warrants, or any other security convertible into Company stock.
Redemption Provisions
The Articles of Incorporation and Bylaws do not provide for redemption rights.
Sinking Fund Provisions
The Articles of Incorporation and Bylaws do not contain sinking fund provisions.
Liability to Further Calls
Shareholders are not required to participate in future capital calls.
Restrictions on Alienability
There are no restrictions of alienability for the Shares.
Drag Along Rights
In the event 65% of the shareholders and the Board approve and agree to a sale, merger, or reclassification of the Company where an excess of 50% of the Company’s voting share or capital is transferred, the Shareholders will have a drag-along right to participate in the sale.
28
Part F/S
Consolidated Financial Statements and Report of
Independent Certified Public Accountants
McGinley Orthopaedic Innovations, LLC
December 31, 2021 and 2020
REPORT OF INDEPENDENT CERTIFIED PUBLIC ACCOUNTANTS
To the Members of
McGinley Orthopaedic Innovations, LLC:
Opinion
We have audited the accompanying consolidated financial statements of McGinley Orthopaedic Innovations, LLC (the “Company”), which comprise of the consolidated balance sheets as of December 31, 2021 and 2020, and the related consolidated statements of operations, changes in members’ equity, and cash flows for the years then ended, and the related notes to the consolidated financial statements.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2021 and 2020, and the results of its operations and cash flows for the years then ended in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”).
Basis of Opinion
We conducted our audits in accordance with auditing standards generally accepted in the United States of America (“U.S. GAAS”). Our responsibilities under those standards are further described in the Auditor’s Responsibilities for the Audit of the Consolidated Financial Statements section of our report. We are required to be independent of the Company and to meet our other ethical responsibilities in accordance with the relevant ethical requirements relating to our audits. We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our audit opinion.
Substantial Doubt about the Company’s Ability to Continue as a Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note C to the consolidated financial statements, the Company had net losses of approximately $2,878,000 and $2,205,000 for the years ended December 31, 2021 and 2020, respectively, as well as negative cash flow from operations of approximately $2,055,000 and $1,485,000, respectively. Management’s evaluation of the events and conditions and management’s plans regarding those matters are also described in Note C. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty. Our opinion is not modified with respect to that matter.
Responsibilities of Management for the Consolidated Financial Statements
Management is responsible for the preparation and fair presentation of the consolidated financial statements in accordance with U.S. GAAP, and for the design, implementation, and maintenance of internal control relevant to the preparation and fair presentation of consolidated financial statements that are free from material misstatement, whether due to fraud or error.
In preparing the consolidated financial statements, management is required to evaluate whether there are conditions or events, considered in the aggregate, that raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date that the consolidated financial statements are available to be issued.
Auditor’s Responsibilities for the Audit of the Consolidated Financial Statements
Our objectives are to obtain reasonable assurance about whether the consolidated financial statements as a whole are free from material misstatement, whether due to fraud or error, and to issue an auditor’s report that includes our opinion. Reasonable assurance is a high level of assurance but is not absolute assurance and therefore is not a guarantee that an audit conducted in accordance with U.S. GAAS will always detect a material misstatement when it exists. The risk of not detecting a material misstatement resulting from fraud is higher than for one resulting from error, as fraud may involve collusion, forgery, intentional omissions, misrepresentations, or the override of internal control. Misstatements are considered material if there is a substantial likelihood that, individually or in the aggregate, they would influence the judgement made by a reasonable user based on the consolidated financial statements.
In performing an audit accordance with U.S. GAAS, we:
Exercise professional judgement and maintain professional skepticism throughout the audit.
Identity and assets the risks of material misstatement of the consolidated financial statements, whether due to fraud or error, and design and perform audit procedures responsive to those risks. Such procedures include examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements.
Obtain an understanding of internal control relevant to the audit in order to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control. Accordingly, no such opinion is expressed.
Evaluate the appropriateness of accounting policies used and the reasonableness of significant accounting estimates made by management, as well as evaluate the overall presentation of the consolidated financial statements.
Conclude whether, in our judgement, there are conditions or events, considered in the aggregate that raise substantial doubt about the Company’s ability to continue as a going concern for a reasonable period of time.
We are required to communicate with those charged with governance regarding, among other matters, the planned scope and timing of the audit, significant audit findings, and certain internal control related matters that we identified during the audit.
s//: Assurance Dimensions
Jacksonville, Florida
July 13, 2022
McGinley Orthopaedic Innovations, LLC
Consolidated Balance Sheets
As of December 31, 2021 and 2020
| ASSETS | 2021 | 2020 | ||||||
| Current assets: | ||||||||
| Cash | $ | 1,624,273 | $ | 2,026,934 | ||||
| Accounts receivable, net | 137,360 | 63,311 | ||||||
| Due from related parties | 72,394 | 6,658 | ||||||
| Inventories | 1,355,011 | 1,373,094 | ||||||
| Prepaid expenses | 101,414 | 74,672 | ||||||
| Total current assets | 3,290,452 | 3,544,669 | ||||||
| Property and equipment, net | 1,190,846 | 1,073,317 | ||||||
| Intangible assets, net | 501,953 | 503,219 | ||||||
| Total assets | $ | 4,983,251 | $ | 5,121,205 | ||||
| LIABILITIES AND MEMBERS’ EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 129,292 | $ | 57,861 | ||||
| Accrued liabilities | 93,781 | 53,742 | ||||||
| Notes payable – related parties | — | 64,287 | ||||||
| Total liabilities | 223,073 | 175,890 | ||||||
| Members’ equity: | ||||||||
| Paid-in capital | 22,867,040 | 21,082,704 | ||||||
| Member units to be issued | 888,333 | — | ||||||
| Accumulated deficit | (18,995,195 | ) | (16,137,389 | ) | ||||
| Total members’ equity | 4,760,178 | 4,945,315 | ||||||
| Total liabilities and members’ equity | $ | 4,983,251 | $ | 5,121,205 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
McGinley Orthopaedic Innovations, LLC
Consolidated Statements of Operations
For the Years Ended December 31, 2021 and 2020
| 2021 | 2020 | |||||||
| Revenues | $ | 1,352,296 | $ | 1,017,289 | ||||
| Cost of goods sold | 555,276 | 448,127 | ||||||
| Gross margin | 797,020 | 569,162 | ||||||
| Selling, general and administrative expenses: | ||||||||
| Wages and benefits | 2,338,656 | 1,890,629 | ||||||
| General office | 494,047 | 398,757 | ||||||
| Research and development | 305,331 | 353,460 | ||||||
| Professional fees | 249,910 | 184,685 | ||||||
| Travel & entertainment | 148,282 | 63,423 | ||||||
| Rent | 115,416 | 26,189 | ||||||
| Insurance | 107,643 | 102,857 | ||||||
| Advertising | 73,610 | 33,421 | ||||||
| Other expenses | 65,240 | 34,343 | ||||||
| Dues & subscriptions | 55,233 | 19,538 | ||||||
| Depreciation and amortization | 50,966 | 58,513 | ||||||
| Taxes & licenses | 24,967 | 33,831 | ||||||
| Computer & interest | 24,659 | 24,301 | ||||||
| Postage & shipping | 16,959 | 9,647 | ||||||
| Total selling, general and administrative expenses | 4,070,919 | 3,233,594 | ||||||
| Loss from operations | (3,273,899 | ) | (2,664,432 | ) | ||||
| Other income: | ||||||||
| Gain on extinguishment of debt | 316,500 | 316,500 | ||||||
| Interest income | 3,630 | 8,179 | ||||||
| Miscellaneous income | 95,963 | 140,059 | ||||||
| Total other income | 416,093 | 464,738 | ||||||
| Net loss before income taxes | (2,857,806 | ) | (2,199,694 | ) | ||||
| Provision for income taxes | — | — | ||||||
| Net loss | $ | (2,857,806 | ) | $ | (2,199,694 | ) | ||
The accompanying notes are an integral part of these consolidated financial statements.
McGinley Orthopaedic Innovations, LLC
Consolidated Statements of Operations
For the Years Ended December 31, 2021 and 2020
| Membership Units | Membership Units | Member Units to be issued | Accumulated deficit | Total | ||||||||||||||||
| Units | Amount | |||||||||||||||||||
| Balance at December 31, 2019 | 42,398,572 | $ | 19,057,422 | $ | — | $ | (13,937,695 | ) | $ | 5,119,727 | ||||||||||
| Issuance of membership units, net of issuance costs | 646,688 | 1,205,278 | — | — | 1,205,278 | |||||||||||||||
| Issuance of membership units for loan repayment | 39,440 | 64,287 | — | — | 64,287 | |||||||||||||||
| Stock-based compensation expense | 390,986 | 755,717 | — | — | 755,717 | |||||||||||||||
| Net loss | — | — | — | (2,199,694 | ) | (2,199,694 | ) | |||||||||||||
| Balance at December 31, 2020 | 43,475,686 | 21,082,704 | — | (16,137,389 | ) | 4,945,315 | ||||||||||||||
| Issuance of membership units, net of issuance costs | 903,846 | 1,720,049 | — | — | 1,720,049 | |||||||||||||||
| Issuance of membership units for loan repayment | 39,440 | 64,287 | — | — | 64,287 | |||||||||||||||
| Stock-based compensation expense | — | — | 888,333 | — | 888,333 | |||||||||||||||
| Net loss | — | — | — | (2,857,806 | ) | (2,857,806 | ) | |||||||||||||
| Balance at December 31, 2021 | 44,418,972 | $ | 22,867,040 | $ | 888,333 | $ | (18,995,195 | ) | $ | 4,760,178 | ||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
McGinley Orthopaedic Innovations, LLC
Consolidated Statements of Cash Flows
For the Years Ended December 31, 2021 and 2020
| 2021 | 2020 | |||||||
| Cash flows from operating activities: | ||||||||
| Net loss | $ | (2,857,806 | ) | $ | (2,199,694 | ) | ||
| Adjustments to reconcile net loss to net cash used by operating activities: | ||||||||
| Depreciation and amortization | 202,141 | 199,714 | ||||||
| Stock-based compensation expense | 888,333 | 755,717 | ||||||
| Gain on extinguishment of debt | (316,500 | ) | (316,500 | ) | ||||
| Changes in assets and liabilities: | ||||||||
| Accounts receivable, net | (74,049 | ) | 77,110 | |||||
| Inventories | 18,083 | 47,936 | ||||||
| Prepaid expenses | (26,741 | ) | 45,096 | |||||
| Accounts payable and accrued liabilities | 111,470 | (94,590 | ) | |||||
| Net cash used by operating activities | (2,055,069 | ) | (1,485,211 | ) | ||||
| Cash flows from investing activities: | ||||||||
| Purchases of property and equipment | (281,489 | ) | (35,126 | ) | ||||
| Investment in intangible assets | (36,915 | ) | (44,732 | ) | ||||
| Net advances to related parties | (65,736 | ) | (2,997 | ) | ||||
| Net cash used by investing activities | (384,140 | ) | (82,855 | ) | ||||
| Cash flows from financing activities: | ||||||||
| Proceeds from PPP loans | 316,500 | 316,500 | ||||||
| Proceeds from membership units issued | 1,762,501 | 1,217,239 | ||||||
| Capital raise costs | (42,452 | ) | (11,961 | ) | ||||
| Net cash provided by financing activities | 2,036,549 | 1,521,778 | ||||||
| Net change in cash | (402,660 | ) | (46,288 | ) | ||||
| Cash at beginning of year | 2,026,933 | 2,073,222 | ||||||
| Cash at end of year | $ | 1,624,273 | $ | 2,026,934 | ||||
| Supplemental and non-cash disclosures | ||||||||
| Cash paid for income taxes | $ | — | $ | — | ||||
| Cash paid for interest | $ | — | $ | — | ||||
| Issuance of member units for payment on note payable | $ | 64,287 | $ | 64,287 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
McGinley Orthopaedic Innovations, LLC
Notes to the Consolidated Financial Statements
December 31, 2021 and 2020
Notes A – Nature of Business
McGinley Orthopaedic Innovations, LLC (“MOI”) is a Wyoming limited liability corporation. MOI was established in 2012 with the goal of increasing patient safety and physician confidence through technological advances in the orthopedic field. MOI has developed and is marketing patented, FDA-approved Intellisense Drill Technology®, an orthopedic power tool, and the Lever Action Plate System® bone fixation implant system.
DS Manufacturing LLC, known commonly as McGinley Manufacturing, is a wholly-owned subsidiary of MOI, and operates a custom machine and fabrication shop with a fully integrated engineering team. Capabilities include custom engineering and design, high precision turning, milling, welding, machining, replacement part fabrication, CNC plasma cutting, powder coating, as well as 5-axis capability. McGinley Manufacturing also manufactures parts of varying sizes, including small scal pieces for precision hand tools to medium-sized pieces for hard rock mining projects for the mining and oil services industries.
McGinley Engineered Solutions, LLC is a wholly-owned subsidiary of MOI, and holds title to all of the patents assigned to MOI.
Note B – Summary of Significant Accounting Policies
Principles of Consolidation
The accompanying consolidated financial statements and related notes include MOI and its wholly owned subsidiaries described in Note A. Hereinafter, they are collectively referred to as the Company. All significant inter-company accounts and transactions have been eliminated in consolidation.
Basis of Accounting
The Company prepares its consolidated financial statements using the accrual methods of accounting in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”).
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the amounts reported in the consolidated financial statements and accompanying notes. Such estimates include inventory reserves for obsolescence, allowance for uncollectible receivables, depreciable lives of property and equipment and costs of patents applied for. Actual results could differ from those estimates.
Recent Accounting Pronouncements
The Company periodically reviews new accounting standards that issued as Accounting Standards Updates (“ASU”) by the Financial Accounting Standards Board (“FASB”). The Company carefully considers all new pronouncements that alter previous U.S. GAAP, and has identified the following new accounting standards that it believes merits further discussion. Other accounting standards that have been issued or proposed by FASB that do not require adoption until a future date are not expected to have a material impact on the consolidated financial statements upon adoption.
The Company adopted Accounting Standards Codification (“ASC”) 606.Revenue from Contracts with Customers as of January 1, 2020 using the modified retrospective method for all contracts with customers. The Adoption did not result in an impact on operating retained earnings. No significant judgements were made in the application of the guidance in ASC 606.
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842). This guidance amends the existing accounting standards for lease accounting, including requiring lessees to recognize most leases on their balance sheet. Under ASU 2020-05, ASU 2016-02 is effective for years beginning on or after December 15, 2021. Management is evaluating the impact of this ASU on the Company’s financial reporting.
Cash
The Company deposits cash with financial institutions which management believes are of high credit quality. The Company does not believe it is exposed to significant risk. At December 31, 2021 and 2020, there were no cash equivalents.
Accounts Receivable
Accounts receivable are stated at the amount management expects to collect for outstanding balances. Management provides for probable uncollectible accounts through an allowance for doubtful accounts. The allowance is estimated based on historical performance and management’s assessment of the current status of individual accounts. Management estimated an allowance of approximately $5,000 and $7,000 at December 31, 2021 and 2020, respectively, which is based on an analysis of individual trade accounts, historical experience with customers and general economic conditions.
Inventory
Inventories are valued on the lower of cost or market. Inventory is valued at the lower of first-in, first-out cost or net realizable value, which is the estimated selling prices in the ordinary course of business, less reasonably predictable costs of completion, disposal, and transportation. The Company has established the policy of capitalizing manufacturing materials and supplies, labor, overhead, and expenses allocable to finished goods and assemblies for the inventory valuation. The Company also carries expendable supplies inventory. This includes expendable items that are consumed in the manufacturing process.
As of December 31, 2021 and 2020, management has recorded a reserve of approximately $1,327,000 for both years. Inventories consisted of the following at December 31:
| 2021 | 2020 | |||||||
| Materials and supplies | $ | 201,953 | $ | 163,695 | ||||
| Work in process and finished goods | 2,479,665 | 2,536,006 | ||||||
| Inventory reserve | (1,326,607 | ) | (1,326,607 | ) | ||||
| Total inventories | $ | 1,355,011 | $ | 1,373,094 | ||||
Property and Equipment
Property and equipment are recorded at cost. Depreciation is calculated using the straight-line method over the estimated useful lives of the assets. Maintenance and repairs are charged to expense as incurred. Betterments and renewals are capitalized. When property and equipment are sold or otherwise disposed of, the asset account and related accumulated depreciation account are relieved, and any gain or loss is included in operations. The straight-line method of depreciation is used over the following estimated useful lives:
| Years | ||||
| Machinery | 5-15 | |||
| Leasehold improvements | 5-15 | |||
| Computers and software | 5 | |||
| Furniture and equipment | 5-15 | |||
| Buildings | 39 | |||
| Vehicles | 5 | |||
| Land | 10 | |||
Additions and improvements are capitalized, while replacements, maintenance, and repairs, which do not improve or extend the life of the respective assets, are expensed as incurred. Gains and losses from the disposition of assets are recorded in the year of disposition.
Intangible Assets
Intangible assets consist of patents and patents pending. Patents and patents pending are being amortized on the straight-line method over twenty years, but not exceeding the initial expiration date of the patent.
Impairment of Long-Lived Assets
Long-lived assets such as property and equipment, and intangible assets subject to amortization are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. If this review reveals an indicator of impairment, as determined based on estimated undiscounted cash flows, the carrying amounts of the related long-lived assets are adjusted to fair value. Management has determined that there has been no impairment in the carrying value of its long-lived assets as of December 31, 2021 and 2020.
Revenue Recognition
All revenues from exchange transactions are recorded in accordance with ASC 606, which is recognized when: (i) a contract with a customer has been identified, (ii) the performance obligation(s) in the contract have been identified, (iii) the transaction price has been determined, (iv) the transaction price has been allocated to each performance obligation in the contract, and (v) the Company has satisfied the applicable performance obligation at a point in time or over time.
The Company’s revenues are primarily generated from the sale of products, and represent a single performance obligation and are earned at a point in time when the goods have been shipped. Payment for the remaining customer’s is due between 15-30 days. The Company’s revenues also contain drill system rentals to customers and are recognized on a per use and/or monthly basis. All revenue is earned in the United States.
The Company’s revenue by type are summarized as follows for the year ended December 31:
| 2021 | 2020 | |||||||
| Product sales | $ | 1,219,511 | $ | 1,004,696 | ||||
| Drill system rentals | 132,785 | 12,593 | ||||||
| Total revenues | $ | 1,352,296 | $ | 1,017,289 | ||||
Research and Development
Research and development costs that do not meet the criteria for capitalization are expensed as incurred. Research and development expenses include compensation, employee benefits, and incentive-unit-based compensation for employees, as well as fees paid to outside consultants. Nonrefundable advanced payments, for goods and services that will be used in future research development activities are expensed when the activity is performed or when the goods have been received, rather than when payment is made, in accordance with ASC 730, Research and Development.
Stock-based Compensation
For stock-based compensation awards, the Company measures compensation costs for these awards to employees and non-employees based upon the fair value of the award on the date of grant.
Income Taxes
Effective January 1, 2016, the Company elected to be taxed as a C corporation. The Company files a consolidated income tax return with its subsidiaries in the U.S. Federal jurisdiction. Generally, the Company’s tax returns remain open for three years for Federal income tax examination. At December 31, 2021, the Company believes there are no significant uncertain tax positions or liabilities, or interest and penalties associated with uncertain tax positions.
The Company’s income tax expenses consists of the following components: current and deferred. Current income tax expense reflects taxes to be paid or refunded for the current period by applying the provisions of the enacted tax law to the taxable income or excess of deductions over revenues. The Company determines deferred income taxes using the liability (or balance sheet) method. Under this method, the net deferred tax asset or liability is based on the tax effects of the differences between the book and tax bases of assets and liabilities, and enacted changes in tax rates and laws are recognized in the period in which they occur.
Deferred income tax expenses results from changes in deferred tax assets and liabilities between periods. Deferred tax assets are recognized if it is more likely than not, based on the technical merits, that the tax position will be realized or sustained upon examination. The term more likely than not means a likelihood of more than 50 percent; the terms examined and upon examination also include resolution of the related appeals or litigation processes, if any. A tax position that meets the more-likely-than-not recognition threshold is initially and subsequently measured as the largest amount of tax benefit that has a greater than 50 percent likelihood of being realized upon settlement with a taxing authority that has full knowledge of all relevant information. The determination of whether or not a tax position has met the more-likely-than-not recognition threshold considers the facts, circumstances, and information available at the reporting date and is subject to management’s judgement. Deferred tax assets are reduced by a valuation allowance if, based on the weight of evidence available, it is more likely than not that some portion or all of a deferred tax asset will not be realized.
Note C – Going Concern
The accompanying consolidated financial statements have been prepared assuming the Company will continue as a going concern. For the years ended December 31, 2021 and 2020, the Company incurred net losses of approximately $2,878,000 and $2,205,000, respectively, and had negative cash flows from operations in the amount of approximately $2,055,000 and $1,485,000, respectively. As of December 31, 2021 and 2020, the Company had an accumulated deficit of approximately $19,021,000 and $16,143,000, respectively. These matters raise substantial doubt about the Company’s ability to continue as a going concern.
The ability of the Company to continue as a going concern is dependent upon increasing sales and obtaining additional capital and financing. Managements intends to attempt to raise funds by way of private offerings. While the Company believes in the viability of its strategy to increase sales and in its ability to raise additional funds, there can be no assurances to that effect. The financial statements do not include adjustments to reflect the possible effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from the outcome of this uncertainty.
Note D – Property and Equipment
Property and equipment consisted of the following as of December 31:
| 2021 | 2020 | |||||||
| Machinery | $ | 1,555,335 | $ | 1,448,200 | ||||
| Leasehold improvements | 152,752 | 39,202 | ||||||
| Computers and software | 75,612 | 68,603 | ||||||
| Furniture and equipment | 46,341 | 30,834 | ||||||
| Buildings | 27,482 | — | ||||||
| Vehicles | 20,244 | 20,244 | ||||||
| Land | 10,806 | — | ||||||
| 1,888,572 | 1,607,083 | |||||||
| Less: accumulated depreciation | (697,726 | ) | (533,766 | ) | ||||
| Property and equipment, net | $ | 1,190,846 | $ | 1,073,317 | ||||
For the years ended December 31, 2021 and 2020, the depreciation expense was approximately $164,000 and $154,000 respectively. The expense is split between cost of goods sold and selling, general and administrative expenses. Depreciation expense included in costs of goods sold was approximately $151,000 and $141,000, respectively.
Note E – Intangible Assets
Intangible assets consisted of the following as of December 31:
| 2021 | 2020 | Useful Life (years) | ||||||||||
| Intellectual property | $ | 802,580 | $ | 765,667 | 20 | |||||||
| Less: accumulated amortization | (300,627 | ) | (262,448 | ) | ||||||||
| Intangible assets, net | $ | 501,953 | $ | 503,219 | ||||||||
Amortization expense charged to operations for the years ended December 31, 2021 and 2020 was approximately $38,000 and $46,000, respectively. The following is a schedule of the estimated amortization expense for intangible assets over the remaining useful life:
| Years ending December 31, | ||||||
| 2022 | 35,908 | |||||
| 2023 | 35,908 | |||||
| 2024 | 35,908 | |||||
| 2025 | 35,908 | |||||
| 2026 | 35,515 | |||||
| Thereafter | 322,806 | |||||
| $ | 501,953 | |||||
Note F – Note Payable – Related Parties
On April 17, 2015 the Company entered into agreement to purchase DS Manufacturing. The purchase agreement called for the Company to purchase DS Manufacturing for a total of $450,010, which was to be paid in membership units of the Company over six years. The balance of the note payable as of December 31, 2021 and 2020, was zero and $64,287 each year. These payments represent non-cash financing payments. As of December 31, 2021, the note payable was paid in full.
Note G – Notes Payable – Paycheck Protection Program Loans
In March 2020, Congress established the Paycheck Protection Program (“PPP”) to provide relief to small businesses during the coronavirus pandemic (“COVID-19”) as part of the Coronavirus Aid, Relief, and Economic Security Act. The legislation authorized Treasury to use the Small Business Association’s 7(a) small business lending program to fund forgivable loans that qualifying businesses could spend to cover payroll, mortgage interest, rent, and utilities during the “Covered Period” defined as the 24-week period starting on the date the PPP loan proceeds are received. Upon meeting certain criteria as specified in the PPP program, the loan is eligible for partial or total forgiveness. The Company received two PPP loans totaling $316,500 in April 2020, and two PPP2 loans of $316,500 in January 2021. These loans were fully forgiven in the respective year they were received, and are reflected as a gain on extinguishment of debt on the consolidated statements of operations.
Note H – Line of Credit
The Company has a $1,000,000 line of credit with a bank which matures February 8, 2024. Interest is subject to change on a monthly basis, calculated at the prime plus a variable rate (3.50% as of December 31, 2021). There was no outstanding balance on the line of credit as of December 31, 2021 and 2020.
McGinley Orthopaedic Innovations, LLC
Notes to the Consolidated Financial Statements
December 31, 2021 and 2020
Note I – Member’s Equity
The Company has the ability to issue an unlimited number of membership units. As of December 31, 2021 and 2020, the Company had 44,418,972 and 43,475,686 membership units issued and outstanding. All membership units have equal voting rights and allocations of profits and losses.
For the years ended December 31, 2021 and 2020, the Company issued 903,846 and 646,688 membership units for cash of approximately $1,720,000 and $1,205,000, respectively, net of issuance costs.
Accrued Member Units
For the years ended December 31, 2021 and 2020, the Company awarded the nine Board of Advisor members 455,556 and 390,986 shares of member units, respectively, valued at $1.95 per member unit, which was based on the most recent issuance price. This compensation for management services was reported as stock-based expense in the amount of $888,333 and $755,717, respectively. These costs are included in wages and benefits on the consolidated statements of operations. As of December 31, 2021, the 455,556 member units earned by the nine Board of Advisor members had not been issued and are included in member units to be issued on the consolidated balance sheets.
As of December 31, 2021, the Company was committed to issuing the following member units to the Board of Advisors related to their agreements. The Company estimated the value of the member units based on the then current issuance price. These have not been accrued as of December 31, 2021 as the Board of Advisor must continue to participate as an advisor to earn the awards.
| Year-ended | Awarded units | Estimated value of the units | ||||||||
| 2022 | 146,368 | $ | 285,417 | |||||||
Note J – Income Taxes
No provision or benefit for federal or state income taxes has been reflected for the year ended December 31, 2021 and 2020 since the Company reported losses and has established a valuation allowance against the total net deferred tax asset.
Significant components of the Company’s net deferred tax assets are as follows as of December 31:
| 2021 | 2020 | |||||||
| Net operating loss carryforward | $ | 2,127,185 | $ | 1,810,683 | ||||
| Property and equipment | (84,562 | ) | (86,646 | ) | ||||
| Inventory reserve | 278,587 | 278,587 | ||||||
| Intangible assets | 10,424 | 12,399 | ||||||
| Stock-based compensation | 186,550 | — | ||||||
| Valuation allowance | (2,518,184 | ) | (2,015,023 | ) | ||||
| Net deferred tax asset | $ | — | $ | — | ||||
At December 31, 2021 and 2020, the Company had federal net operating loss carryforwards of approximately $10,129,000 and $8,622,000, respectively. The federal net operating losses beginning in 2018 may be carried forward indefinitely but are limited to 80% of the taxable income in any one tax period. The deferred tax assets has been adjusted to account for the limitation. The pre-2018 net operating losses are approximately $2,378,000 and can be carried back two years and forward 20 years, and will begin expiring in 2036. The Company’s federal and state blended tax rate was 21% for the years ended December 31, 2021 and 2020, respectively.
The entire balance of the deferred tax asset has been offset by a valuation allowance since there may be limitations on the amount of net operating loss carryforwards which can be used in future years, and utilization of net operating loss carryforwards cannot be sufficiently assured at December 31, 2021 and 2020.
Note K – Related Party Transactions
Related Party Receivables
The Company has receivables due from various related parties of approximately $72,000 and $7,000 as of December 31, 2021 and 2020, respectively.
Related Party Payables
The Company has payables due to various related parties of approximately $6,000 and $9,000 as of December 31, 2021 and 2020, respectively. These are included in accounts payable on the accompanying consolidated balance sheets.
Related Party Operating Lease
The Company has an operating lease for office space from a related party which began in January 2021. The lease is for seven years and expires in December 2027. Rent expense is recognized on a straight-line basis over the term of the lease. The lease is also subject to a three percent minimum annual increase. In connection with the related party lease, approximately $102,000 was paid to the lessor for specific leasehold improvements requested by the Company.
Future minimum rental payments under non-cancellable operating leases (with initial or remaining lease terms in excess of one year) for each of the five years subsequent to December 31, 2021 are:
| 2022 | $ | 113,221 | ||||
| 2023 | 113,280 | |||||
| 2024 | 113,341 | |||||
| 2025 | 113,403 | |||||
| Thereafter | 113,467 | |||||
| Total | $ | 566,712 |
Total combined rent expense associated with the operating leases amounted to approximately $114,000 and $0 for the years ended December 31, 2021 and 2020, respectively.
Note L – Concentrations and Commitments
Operating Leases
The Company leases office space under operating leases agreements that expire at various dates through July 2021. Rent expense was recognized on a straight-line basis over the term of the lease. Total combined rent expense associated with the operating leases amounted to approximately $64,000 and $20,000 for the years ended December 31, 2021 and 2020, respectively. Upon termination of these lease agreements, the Company leased office space with a related party as discussed at Note J.
COVID-19
Due to the COVID-19 outbreak in 2020, the Company was not closed at any point. Additionally, there were no staff furloughed or terminated due to COVID-19. Overall, the Company was not significantly affected by COVID-19.
Due to the level of risk this virus may have on the global economy, it is at least reasonably possible that it could have an impact on the operations of the Company in the near term that could materially impact the Company’s consolidated financial statements, however, management does not believe there will be any future impact. In addition, with vacancies and therapeutics coming to market, these will also help to mitigate any potential future losses.
Note M – Subsequent Events
On May 13, 2022, the Company filed to be a Wyoming corporation. The new name of the Company is McGinley Orthopaedic Innovations, Inc. The Company’s membership units converted to common stock, which have a par value of $0.001. The Board also established that the Company is authorized to issue 100,000,000 shares of common stock.
Between the balance sheet date for the year ending December 31, 2021 and July 13, 2022, the date the consolidated financial statements were available to be issued, the Copany issued 722,821 member units raising additional capital of approximately $1,410,000.
Subsequent events have been evaluated through July 13, 2022, which is the date the consolidated financial statements were available to be issued.
29
Exhibit Index
Exhibit 2A: Certificate of Incorporation and Other Documents
Exhibit 2B: Bylaws
Exhibit 4. Subscription Agreement
Exhibit 8. Escrow Agreement
Exhibit 11. Written Expert Consent Letter of Assurance Dimensions
Exhibit 12. Legal Opinion of Trae O’Neil High
Exhibit 13: Testing the Waters Materials
30
Signature Page
Pursuant to the requirements of Regulation A, the issuer certifies that it has reasonable grounds to believe that it meets all of the requirements for filing on Form 1-A and has duly caused this Offering Statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Casper, WY, on August 11, 2022.
(Exact name of the Issuer as specified in its Charter)
McGinley Orthopaedic Innovations, Inc.
234 E. 1st St. Suite 242
Casper, WY 82601
(307) 315-6403
By:
s/Dr. Joseph McGinley
Director, CEO, President, Treasurer, Secretary of McGinley Orthopaedic Innovations, Inc.
(Date): August 11, 2022
Location Signed: Casper, WY
This Offering Statement has been signed by the following Officers in the capacities and on the dates indicated.
s/Dr. Joseph McGinley
CEO, President, Treasurer, Secretary of McGinley Orthopaedic Innovations, Inc.
(Date): August 11, 2022
Location Signed: Casper, WY
This Offering Statement has been signed by the following Directors in the capacities and on the dates indicated.
s/Dr. Joseph McGinley
Director of McGinley Orthopaedic Innovations, Inc.
(Date): August 11, 2022
Location Signed: Casper, WY
ARTICLES OF INCOP
OF
MCGINLEY ORTHOPAEDIC INNVOATIONS, INC.
A Wyoming Corporation
Pursuant to Wyo. Stat. §§ 17-29-1009 and 17-26-101, McGinley Orthopaedic Innovations, LLC, with Articles of Organization filed with the Secretary of State on April 27, 2012, hereby converts to a Wyoming corporation as set forth in the Statement of Conversion attached hereto and as stated as follows:
RECITALS
WHEREAS, the company was originally created on April 27, 2012, in the State of Wyoming.
WHEREAS, the original name of the limited liability company formed in the state of Wyoming is McGinley Orthopaedic Innovations, LLC, with filing ID No. 2012-000621432.
WHEREAS, the members approved the conversion of the entity from a limited liability company to a corporation by more than 65% of the Membership Interests, as required by the Operating Agreement, with 77% of the Membership Interests voting in favor of the conversion and no votes against.
WHEREAS, the name of the converted entity shall be McGinley Orthopaedic Innovations,
ARTICLE I. NAME OF THE CORPORATION
The name of the Corporation shall be: McGinley Orthopaedic Innovations, Inc.
ARTICLE II. REGISTERED AGENT
The name and physical address of the Corporation's registered agent is: Hathaway & Kunz, LLP, 2515 Warren Ave., Suite 500, Cheyenne, Wyoming 82001.
In accordance with Wyo. Stat. § 17-28-104(e), the business entity consents to service by electronic means per the requisite email address set forth in the organizer signature block below for use in the limited circumstances where the business entity has no registered agent or where the agent cannot with reasonable diligence be served.
ARTICLE III. MAILING ADDRESS
The mailing address of the Corporation is 234 E 1st Street, Suite 242, Casper, W
1
ARTICLE IV. PRINCIPAL OFFICE ADDRESS
The principal office address of the Corporation is 234 E 1st Street, Suite 242, Casper, Wyoming 82601.
ARTICLE V. AUTHORIZED STOCK
A. The corporation is authorized to issue one (1) class of common shares. Common shares shall be common stock with voting rights, as such is restricted herein. The corporation is authorized to issue an unlimited number of common shares of stock.
B. Dividends may be declared by the Board of Directors of the Corporation and paid out of any funds legally available therefor at such times and in such amounts as the Board of Directors shall determine.
C. The capital stock, after the amount of the subscription price has been paid in, shall not be subject to assessment to pay the debts of the Corporation.
D. Any stock of the Corporation may be issued for money, property, services rendered, labor done, cash advances for the Corporation, or for any other assets of value in accordance with the action of the Board of Directors, whose judgment as to value received in return therefor shall be conclusive and said stock when issued shall be fully paid and non-assessable.
E. The Board of Directors shall have the authority to impose restrictions upon the transfer of the capital stock of the Corporation from time to time as it deems necessary in the best interests of the Corporation or as required by law.
F. The Board of Directors shall have the authority to determine whether the Corporation will issue physical stock certificates.
2
ARTICLE VI. NO PREEMPTIVE RIGHTS
No holder of any stock of the Corporation shall be entitled, as a matter of right, to purchase, subscribe for or otherwise acquire any new or additional shares of stock of the Corporation of any class, or any options or warrants to purchase, subscribe for or
otherwise acquire any such new or additional shares, or any shares, bonds, notes, debentures or other securities convertible into or carrying options or warrants to purchase, subscribe for or otherwise acquire any such new or additional shares, unless specifically authorized by the Board of Directors of the Corporation.
ARTICLE VII. DIRECTORS
A. The management of the business and the conduct of the affairs of the Corporation shall be vested in its Board. The Board of this Corporation shall be known as directors, and the number of the directors may from time to time be increased or decreased in such manner as shall be permitted by this Article or determined by the Directors as set forth in the Bylaws of this Corporation. There shall not be fewer than one (1) member of the Board of Directors and no more than seven (7) members of the Board of Directors.
B. The Board is expressly empowered to adopt, amend or repeal the Bylaws of the Corporation, subject to any restrictions that may be set forth in these Articles of Incorporation.
C. Each member of the Board of Directors shall have one vote at a meeting of the Board of Directors. Matters of the Board of Directors shall be decided by a majority of the directors present at a meeting of the Board of Directors at which a quorum is present. The Board of Directors shall be expressly permitted to take action without a meeting, provided such action is evidenced by a written consent and signed by a majority of the directors.
D. The name and address of the initial director of the corporation shall be: Joseph C. McGinley, 234 E 1st Street, Suite 242, Casper, Wyoming 82601.
E. The holders of record of Common Shares shall be entitled to elect the directors of the Corporation in accordance with Article VIII. Any director elected as provided in the preceding sentence may be removed without cause by, and only by, an affirmative shareholder vote of sixty-five percent (65%) of common shares of stock eligible to vote and as in accordance with Article VIII.
ARTICLE VIII. SHAREHOLDER VOTING ON CORPORATE ACTIONS
A. Shareholders of the Corporation shall only be permitted to vote on those items set forth herein.
i) Removal of a Director;
ii) Election of Directors;
iii) Termination and dissolution of the Corporation;
iv) Amendment of these Articles, subject to Article XIII;
v) Any matter requiring a vote of the shareholders as provided in these Articles or the Wyoming Business Corporation Act.
3
B. Shareholders may vote by written consent, with or without a formal meeting or prior notice. Action taken by written consent may be taken if such consent is signed and dated by not less than the minimum number of votes that would be required to authorize or take the action at a meeting at which all shares entitled to vote on the action were present and voted, and such consents shall be delivered to the Corporation for inclusion in the minutes. Assignees or transferees of common shares of stock may not vote unless they become a holder of record with approval of the Directors of the Corporation.
C. The holder of each common share issued and outstanding shall have one vote in respect of each common share held immediately after the close of business on the record date fixed for such meeting or the effective date of such written consent and shall be entitled to notice of any stockholders' meeting in accordance with the bylaws of the Corporation.
D. Unless otherwise specified herein, on any matter subject to shareholder approval, the affirmative vote or concurrence of the holders of more than fifty percent (50%) of the total votes of the outstanding common shares of the Corporation entitled to vote thereon are required to make effective all transactions that require shareholder approval under applicable law, except that the following shall require an affirmative shareholder vote of sixty-five percent (65%) of common shares of stock eligible to vote:
i) Confess a judgment against the Corporation, or to file or consent to filing a petition for or against the corporation under any federal or state bankruptcy, insolvency, or reorganization act;
ii) Sell substantially all of the property in liquidation or cessation of the business of the Corporation before actual termination of the Corporation;
iii) Amend these Articles except as provided in Article XIII;
iv) Change or reorganize the Corporation into any other legal form;
v) Expel a shareholder and force a mandatory redemption and/or acquisition of any shareholders shares;
vi) Dissolve and liquidate the Corporation;
vii) Distribute more than 25% of the fair market value of the corporation's assets in any tax year;
E. For all other matters not specifically enumerated herein, the Board of Directors shall have full authority to manage the Corporation.
ARTICLE IX. DIRECTOR LIMITATION OF LIABILITY
A. To the fullest extent permitted by Wyoming law, specifically Wyo. Stat 17-16-202 (2021) of the Wyoming Business Corporations Act, a director of the Corporation shall not be personally liable to the Corporation or its shareholders for monetary damages for any action taken, or any failure to take any action, as a director, except liability for: (i) the amount of financial benefit received by a director to which he is not entitled; (ii) an intentional infliction of harm on the corporation or shareholders; (iii) violation of Wyo. Stat. 17-16-833; or (iv) an intentional violation of criminal law. If the Wyoming Business Corporations Act is amended after this Article is adopted to authorize corporate action further eliminating or limiting the personal liability of directors, then the liability of a director of the Corporation shall be eliminated or limited to the fullest extent permitted by the Wyoming Business Corporations Act, as so amended.
B. Any repeal or modification of the foregoing paragraph by the shareholders of the Corporation shall not adversely affect any right or protection of a director of the Corporation existing at the time of such repeal or modification.
4
ARTICLE X. INDEMNIFICATION OF DIRECTORS OFFICERS, EMPLOYEES, FIDUCIARIES AND AGENTS
A. The liability of the directors of the Corporation for monetary damages shall be eliminated to the fullest extent permissible under Wyoming law. The Corporation shall indemnify, to the fullest extent permitted by applicable law, any person, and the estate and personal representative of any such person, against all liability and expense (including attorneys' fees) incurred by reason of the fact that he is or was a director or officer of the Corporation or, while serving at the request of the Corporation as a director, officer, partner, trustee, employee, fiduciary, attorney or agent of, or in any similar managerial or fiduciary position of, another domestic or foreign corporation or other individual or entity or of an employee benefit plan. The Corporation also shall indemnify any person who is serving or has served the Corporation as director, officer, employee, fiduciary, attorney or agent, and that person's estate and personal representative, to the extent and in the manner provided in any bylaw, resolution of the shareholders or directors, contract, or otherwise, so long as such provision is legally permissible.
B. The Corporation shall advance expenses in advance of the final disposition of the case to or for the benefit of a director, officer, employee, fiduciary, or agent, who is party to a proceeding such as described in the preceding paragraph A to the maximum extent permitted by applicable law.
C. Any repeal or modification of the foregoing paragraph by the shareholders of the Corporation shall not adversely affect any right or protection of a director or officer of the Corporation or other person entitled to indemnification existing at the time of such repeal or modification.
ARTICLE XI. DRAG-ALONG
A. In the event that the holders of more than sixty-five percent (65%) of the aggregate voting power of the issued and outstanding Common Shares (the "Dragging Shareholders") and the Board of Directors approve and agree to a proposed Trade Sale (as defined below) (as so approved and agreed, the "Approved Sale"), then the Dragging Shareholders shall have the right to require all other shareholders of the Corporation (the "Dragged Shareholders") by giving a written notice (the "Approved Sale Notice") to the Corporation and each of the Dragged Shareholders which shall include (i) a description and number of the shares, other equity securities or assets of the Corporation (or its subsidiary) to be sold, (ii) the identity and address of the prospective transferee, and (iii) the consideration for which the Approved Sale is proposed to be made, subject to and upon such terms and conditions as the Dragging Shareholders may reasonably require and within thirty (30) days after giving such Approved Sale Notice:
i. in the event an Approved Sale is to be brought to a vote at a general meeting of the Corporation and to the extent such Dragged Shareholder is entitled to vote, to vote as otherwise provided herein; and/or
ii. in the event that the Approved Sale is to be effected by the sale of shares of the Corporation, to sell all shares of the Corporation beneficially held by the Dragged Shareholders in the Approved Sale (or in the event that the Dragging Shareholders are selling fewer than all of their shares held in the Corporation, shares in the same proportion as the Dragging Shareholders are selling) on the same terms and conditions as those offered to the Dragging Shareholders.
B. For the purpose of this Article XI, a "Trade Sale" means any of the following events: (i) any consolidation, amalgamation, scheme of arrangement, reorganization, merger or other transaction in which the shareholders of the Corporation (or any subsidiary of the Corporation) immediately prior to such consolidation, amalgamation, scheme of arrangement, reorganization, merger or other transaction, own less than 50% of such surviving entity's voting power or share capital in the aggregate immediately after such consolidation, amalgamation, scheme of arrangement, reorganization, merger, or other transaction, (ii) any transaction or series of related transactions in which in excess of 50% of the Corporation's (or any of its subsidiaries') voting power or share capital is transferred; or (iii) a sale, transfer, lease, exclusive licensing or other disposition of all or substantially all of the assets of the Corporation or any of its subsidiaries (whether in a single transaction or a series of related transaction).
C. In furtherance of the foregoing, each Dragged Shareholder agrees:
5
i. in the event an Approved Sale is to be brought to a vote at a general meeting, each Dragged Shareholder (to the extent it is entitled to vote at such meeting) shall be present, in person or by proxy, or by electronic means at all such meetings and be counted for the purposes of determining the presence of a quorum at such meetings and the presence of the number of votes necessary for the effectiveness of any shareholder resolutions;
ii. in the event an Approved Sale is to be brought to a vote at a general meeting, each Dragged Shareholder (to the extent it is entitled to vote at such meeting) shall vote (in person, by proxy or by action by written consent, as applicable) all shares in the Corporation as to which it has record or beneficial ownership in favor of such Approved Sale and in opposition of any and all other proposals that could reasonably be expected to delay or impair the ability of the Corporation to consummate such Approved Sale;
iii. to refrain from exercising any dissenters' rights or rights of appraisal under applicable laws (if any) at any time with respect to such Approved Sale; and
1v. to execute and deliver all related documentation and take such other action in support of the Approved Sale as shall reasonably be requested by the Dragging Shareholders.
D. Forthe avoidance of doubt, if and to the extent that there are inconsistencies between this Article XI and any other provisions of this Article, the terms of this Article XI shall prevail and control. Notwithstanding any provision in this Article, any transfer or transaction contemplated under this Article XI shall not be subject to any transfer restriction, or any prior written consent or approval of any shareholder or any director, except those specifically set forth in this Article XI.
ARTICLE XII. CONFLICTING INTEREST TRANSACTIONS
No act, contract, or other transaction between the Corporation and one or more of its directors, officers, or employees, or between the Corporation and any corporation or association of which one or more of this Corporation's officers, directors, or employees are in any way interested, shall be affected or invalidated in any way because of such fact; provided the director brings it to the attention of the Corporation and: (i) action by qualified directors disclaiming the corporation's interest in the opportunity is taken in compliance with the procedures set forth in the Wyoming Business Corporations Act, as if the decision being made concerned a director's conflicting interest transaction; or (ii) Shareholders' action disclaiming the corporation's interest in the opportunity is taken in compliance with the procedures set forth in Wyoming Business Corporations Act, as if the decision being made concerned a director's conflicting interest transaction, except that, rather than making required disclosure as defined under the Wyoming Business Corporations Act, in each case the director shall have made prior disclosure to those acting on behalf of the Corporation of all material facts concerning the business opportunity that are then known to the director.
ARTICLE XIII. AMENDMENT WITHOUT VOTE
A. The Board of Directors may, without a shareholder vote, amend any provision of these Articles or the Corporation's Bylaws and prepare and deliver any documents necessary to reflect:
i. A change in the name of the corporation or the location of the principal office of the Corporation;
ii. The admission, substitution, or termination of shareholders according to these Articles;
iii. A change that the Board of Directors in its sole discretion determines to be necessary or advantageous to qualify or to enable the corporation to continue to qualify as a corporation or to ensure that the tax treatment of the corporation does not change;
iv. A change that does not adversely affect the shareholders in any material respect or that is required or contemplated by these Articles; or
v. Any other amendments similar to the foregoing.
B. Any other amendments require the affirmative approval of sixty-five (65%) of the issued and outstanding of common shares as set forth in Section VIII(D)(iii).
6
ARTICLE XIV. ELECTRONIC TRANSACTIONS
The directors, officers, and shareholders of the Corporation agree to conduct transactions by electronic means pursuant to the Uniform Electronic Transactions Act, codified as Wyo. Stat. § 40- 21-101 ct seq. All notices, communications, and meetings may be conducted or provided through the use of any means of communication, including, for meetings, electronic transmission by which all parties participating may communicate with each other during the meeting. Wherever notice is required under these Articles, the Corporation's Bylaws, or, if applicable by law, notice may be communicated by electronic transmission, such as by email, to the shareholders entitled to such notice. Notice by electronic transmission shall constitute written notice. Such notice shall be effective when
an electronic transmission has been made to a data address, email address, or by any other electronic means provided by the shareholder.
ARTICLE XV. DIRECTOR DUTIES
In carrying out the duties of the Board, the Director(s) shall act as a fiduciary for the Shareholders. In fulfilling this fiduciary duty, the Director(s) shall act in good faith and loyalty in a manner the Director reasonably believes to be in the best interests of the Corporation and its Shareholders, and with such care as an ordinary prudent person in a like position would use under similar circumstances.
ARTICLE XVI.
The incorporators' names and addresses follow:
Joseph C. McGinley, MD., Ph.D.
234 E 1st Street, Suite 242
Casper, Wyoming 82601
mcginley@mcginleyinnovations.com
s//: Joseph McGinley
Joseph C. McGinley, MD., Ph.D.
Date: May, 112022
7
Statement of Conversion
1. Pursuant to Wyo. Stat. §§ 17-29-1009 and 17-26-101, McGinley Orthopaedic Innovations, LLC, with Articles of Organization filed with the Secretary of State on April 27, 2012, has converted to a corporation.
2. The original name of the limited liability company is McGinley Orthopaedic Innovations, LLC, with filing ID No. 2012-000621432.
3. The name of the new entity is McGinley Orthopaedic Innovations, Inc.
4. The original company was created on April 27, 2012, in the State of Wyoming.
5. The members approved the conversion of the entity from a limited liability company to a corporation by more than 65% of the Membership Interests, as required by the Operating Agreement, with 77% of the Membership Interests voting in favor of the conversion and no votes against.
DATED as of this 11th day of May 2022.
MCGINLEY ORTHOPAEDIC INNOVATIONS, INC.
BY: s//: Joseph McGinley
Incorporator
Joseph C. McGinley, M.D., Ph.D.
234 E 1st Street, Suite 242
Casper, Wyoming 82601
mcginley@mcginleyinnovations.com
8
STATE OF WYOMING
Office of the Secretary of State
I, EDWARD A. BUCHANAN, Secretary of State of the State of Wyoming, do hereby certify that the filing requirements for the issuance of this certificate have been fulfilled.
CERTIFICATE OF CONVERSION
McGinley Orthopaedic Innovations, LLC converted from a Wyoming Limited Liability Company
toMcGinley Orthopaedic Innovations, Inc. a Wyoming Profit Corporation
I have affixed hereto the Great Seal of the State of Wyoming and duly executed this official certificate at Cheyenne, Wyoming on this 13th day of May, 2022
s//: Edward A. Buchanan
Secretary of State
By: Shawn Havel
Filed Date: 05/13/2022
1
RESOLUTIONS
of
McGINLEY ORTHOPAEDIC INNOVATIONS, Inc.
The undersigned, being the sole Director of McGinley Orthopaedic Innovations, Inc. (the “Company”), and acting pursuant to his authority as set forth in the Articles of Incorporation of the Company dated May 13, 2022 (the “Articles”), hereby adopts the following resolutions:
WHEREAS, on May 13, 2022, the Company converted from a limited liability company to a corporation by filing Articles and a Statement of Conversion with the Wyoming Secretary of State; and
WHEREAS, such Articles authorize the Company to issue an unlimited number of one class of common shares of stock; and
WHEREAS, the Company now wishes to limit the common stock authorized to be issued to one hundred million (100,000,000) shares of common stock of one class;
NOW, THEREFORE, BE IT RESOLVED, that the Company’s previously authorized number of shares of common stock be limited to one hundred million (100,000,000).
IT IS FURTHER RESOLVED, that any actions taken by the Director and/or officers of the Company prior to the date of the foregoing resolutions that are within the authority conferred thereby are hereby ratified, confirmed, and approved as the acts and deeds of the Company.
IT IS FURTHER RESOLVED, that the Director and/or officers of the Company, be, and each of them hereby is, authorized to do and perform any and all such acts, including execution of any and all documents, deemed necessary or advisable to carry out the purposes of the foregoing resolutions.
Signed this ____ day of June 2022.
________________________________________
Joseph C. McGinley, Director
1
RESOLUTIONS
of
McGINLEY ORTHOPAEDIC INNOVATIONS, Inc.
The undersigned, being the sole Director of McGinley Orthopaedic Innovations, Inc. (the “Company”), and acting pursuant to his authority as set forth in the Articles of Incorporation of the Company dated May 13, 2022 (the “Articles”), hereby adopts the following resolutions:
WHEREAS, on May 13, 2022, the Company converted from a limited liability company to a corporation by filing Articles and a Statement of Conversion with the Wyoming Secretary of State; and
WHEREAS, such Articles authorize the Company to issue one class of common shares of stock; and
WHEREAS, such Articles require the capital stock of the Company to be fully paid and non-assessable; and
WHEREAS, the Company now wishes to establish the par value of such common stock at $0.001 per share;
NOW, THEREFORE, BE IT RESOLVED, that the par value of the Company’s shares of common stock shall be established at $0.001 per share.
IT IS FURTHER RESOLVED, that any actions taken by the Director and/or officers of the Company prior to the date of the foregoing resolutions that are within the authority conferred thereby are hereby ratified, confirmed, and approved as the acts and deeds of the Company.
IT IS FURTHER RESOLVED, that the Director and/or officers of the Company, be, and each of them hereby is, authorized to do and perform any and all such acts, including execution of any and all documents, deemed necessary or advisable to carry out the purposes of the foregoing resolutions.
Signed this ____ day of June 2022.
________________________________________
Joseph C. McGinley, Director
BYLAWS OF
MCGINLEY ORTHOPAEDIC INNOVATIONS, INC.
ARTICLE I
OFFICES
1.1 Principal Office. The principal office of McGinley Orthopaedic Innovations, Inc., a Wyoming corporation, (the “Corporation”) shall be located at such location within or without the State of Wyoming as is designated in its articles of incorporation or the most recent annual report filed with the Secretary of State of the State of Wyoming. Certain books and records are required by these bylaws to be maintained by the Corporation at its principal office. The Corporation may maintain such additional offices, either within or without the State of Wyoming as are necessary or convenient for the operation of its business.
1.2 Registered Agent. The Registered Agent of the Corporation shall be Hathaway & Kunz, LLP, located at 2515 Warren Ave., Suite 500, Cheyenne, Wyoming, 82001. The registered office is the office at which service of process may be served on the Corporation and may, but need not, be the same as the principal office. The registered office of the Corporation may not be changed without filing a notice of the change with Secretary of State of Wyoming.
ARTICLE II
SHAREHOLDERS
2.1 Annual Shareholders’ Meetings. The annual meeting of the shareholders will be held at a date and time as set by the Board of Directors. At the annual meeting, the shareholders must elect a board of directors and may transact any other business that may legally come before the meeting.
2.2 Remote Meeting. Shareholders may participate in a meeting of the shareholders by means of electronic communication by which all persons participating in the meeting can hear each other during the meeting. A shareholder participating in a meeting by this means is deemed to be present in person at the meeting.
2.3 Special Shareholders’ Meetings. Special meetings of the shareholders may be called by the President, the Board of Directors, or shareholders holding at least 10% of all votes entitled to be cast on any issue proposed to be considered at the special meeting. If the special meeting is called by shareholders, such shareholders must sign, date, and deliver to the secretary one or more written demands for the special meeting, describing the purpose or purposes for which it is to be held. The record date for determining shareholders entitled to demand a special meeting is the date the first shareholder signs the demand. Only business within the purpose or purposes described in the notice of the meeting may be conducted at a special meeting.
2.4 Place of Shareholders’ Meetings. Meetings of the shareholders shall be held at such place within or without the State of Wyoming as designated by the Board of Directors or the person or persons calling the meeting. The Board of Directors may, in its sole discretion, determine that the meeting shall not be held at any place, but may instead be held solely by means of electronic transmission by which all parties participating may communicate with each other during the meeting.
1
2.5 Notice of Shareholders’ Meetings.
2.5.1 Required Notice. Except as otherwise provided by law, notice, given in writing or by electronic transmission, of each meeting of stockholders shall be given not less than ten (10) nor more than sixty (60) days before the date of the meeting. Notice shall be given to each stockholder entitled to vote at such meeting and to any other shareholder entitled to receive notice of the meeting under the Wyoming Business Corporation Act (the “Act”) or the articles of incorporation. Such notice shall specify the place, if any, date and hour, and the means of remote communications, if any, by which stockholders and proxyholders may be deemed to be present and vote at any such meeting. In the case of special meetings, the notice shall include the purpose or purposes of the meeting. Notice of shareholders’ meetings may be given by electronic communication.
2.5.2 Effective Date. A notice of a meeting of the shareholders will be deemed effective at the earlier of:
1. When received;
2. When deposited in the United States mail, with postage prepaid, addressed to the shareholder at the shareholder’s address as it appears on the Corporation’s current record of shareholders;
3. Five days after deposit in the United States mail, if mailed, with postage prepaid, to the shareholder at an address where the shareholder receives mail other than the address that appears on the Corporation’s current record of shareholders; or
4. On the date shown on the return receipt if sent by registered or certified mail, return receipt requested, and the receipt is signed on behalf of the addressee.
5. Electronic transmission is effective when it has been made to the data address of the shareholder or when it has been made in a manner otherwise authorized by the shareholder.
2.5.3 Meetings for Special Purposes. If a purpose of any shareholder meeting is to consider a proposed amendment to the articles of incorporation; a plan of merger or share exchange; the sale, lease, exchange, or other disposition of all, or substantially all of the Corporation’s property; the dissolution of the Corporation; or the removal of a director, the notice must so state and must be accompanied by the items required by the Act.
2.5.4 Adjourned Meeting. When a meeting is adjourned for more than 120 days after the date fixed for the original meeting, or when a redetermination of the persons entitled to receive notice of the adjourned meeting is required by law, notice of the adjourned meeting must be given in the same manner as required for an original meeting. In all other cases, no notice of the adjournment or of the business to be transacted at the adjourned meeting need be given other than by announcement at the original meeting before adjournment.
2.5.5 Waiver of Notice. A shareholder entitled to notice may waive notice of a meeting of the shareholders by delivering a signed written waiver to the Corporation or by electronic
transmission by such shareholder. The waiver will be effective whether signed before or after the meeting. A shareholder’s attendance at a meeting waives the right to object to lack of notice or defective notice of the meeting, unless the shareholder objects to holding the meeting at the beginning of the meeting. A shareholder’s attendance at a meeting also waives objection to consideration of a particular matter at the meeting that is not within the purpose or purposes described in the meeting notice, unless the shareholder objects to considering the matter when it is presented. Any shareholder so waiving notice of such meeting shall be bound by the proceedings of any such meeting in all respects as if due notice thereof had been given.
2.6 Shareholders of Record. The persons entitled to receive notice of or to vote at any shareholders’ meeting will be those persons designated as shareholders in the stock transfer books of the Corporation at the close of business on the day prior to the date the notice is mailed or otherwise transmitted, or on such other date as determined in advance by the board of directors, which date may not be more than 70 or less than 10 days before the meeting. If the meeting is a special shareholders’ meeting demanded by shareholders, the persons entitled to receive notice of the meeting and to vote will be determined as of the date the first shareholder signs the demand, unless otherwise determined in advance by the board of directors.
2.7 Shareholders’ List. The secretary must make a complete record of the shareholders entitled to vote at each meeting, arranged in alphabetical or numerical order, showing the mailing address of each shareholder if the shareholder is identified by name, or the shareholder’s authorized means of receipt for electronic transmissions if the shareholder is identified by data address, or both the mailing address and data address. The record shall include the number of shares held by each shareholder. The shareholders’ list must be available, at the Corporation’s principal office, for inspection by any shareholder, or the shareholder’s agent or attorney, beginning two business days after notice of the meeting is given and continuing through the meeting.
2
2.8 Quorum.
2.8.1 Required Quorum. A majority of the shares entitled to vote, represented in person, by proxy, or by remote electronic communication, constitutes a quorum for the transaction of business at any shareholders’ meeting. If a person attends a meeting for the express purpose of objecting to the transaction of any business on the grounds that the meeting is not lawfully called or convened, the shares held by that person or represented by a proxy given to that person will not be included for purposes of determining whether a quorum is present. Once a share is represented for any purpose at a meeting, other than for the purpose of objecting as provided above, it is deemed present for quorum purposes for the remainder of the meeting and any adjournment thereof, unless a new notice is or must be given for the adjourned meeting or a new record date must be set for the meeting. The persons present, in person or via electronic transmission, at a duly organized meeting may continue to transact business until adjournment, notwithstanding the withdrawal of enough persons to leave less than a quorum.
2.8.2 Adjournment to Obtain Quorum. In the absence of a quorum, a majority of the shares represented in person, via electronic transmission, or by proxy may, subject to the requirements of notice and record date set forth in these bylaws, adjourn a shareholders’ meeting from time to time until a quorum attend. Any business which might have been transacted at the original meeting may be transacted at the adjourned meeting if a quorum exists.
2.9 Voting of Shares.
2.9.1 One Share, One Vote. Each shareholder is entitled to one vote on each matter submitted to a vote at a meeting of the shareholders for each share of voting stock standing in the name of the shareholder on the stock transfer books of the Corporation.
2.9.2 Proxies. A shareholder may vote in person, via electronic transmission, or by proxy in writing signed by the shareholder or by the shareholder’s duly authorized attorney-in-fact. Such proxy will be effective when received by the secretary or other officer or agent of the Corporation authorized to tabulate votes. No proxy will be valid after 11 months from the date of its execution, unless a longer period is expressly provided in the proxy.
2.9.3 Shares Owned by Corporation. Shares held in the name of another corporation may be voted by such officer, agent, or proxy as the bylaws of such corporation may prescribe, or, in the absence of such provision, as the board of directors of the Corporation holding the shares may determine. However, shares held in the name of another corporation are not entitled to vote if this corporation owns, directly or indirectly, a majority of the shares entitled to vote for the directors of the other corporation.
2.9.4 Shares Held by Fiduciary. Shares held by a personal representative, administrator, executor, guardian, or conservator may be voted by that person, either in person or by proxy, without a transfer of such shares into the name of that person. Shares held in the name of a trustee may be voted by the trustee, either in person, via electronic transmission, or by proxy, but no trustee is entitled to vote shares held by that trustee without a transfer of such shares into the name of the trustee.
2.9.5 Shares Held by Receiver. Shares standing in the name of a receiver may be voted by the receiver, and shares held by or under the control of a receiver may be voted by the receiver without being transferred into the receiver’s name if authority to do so is contained in an appropriate order of the court by which the receiver was appointed.
2.9.6 Pledged Shares. A shareholder whose shares are pledged is entitled to vote such shares until the shares have been transferred into the name of the secured party, and thereafter the secured party is entitled to vote the shares so transferred.
2.9.7 Rejection of Vote. The Corporation is entitled to reject a vote, consent, waiver, or proxy appointment if the secretary or other officer or agent authorized to tabulate votes, acting in good faith, has reasonable basis for doubt about the validity of the signature on it or about the signor’s authority to sign for the shareholder.
2.9.8 Joint Owners of Shares. If shares or other securities having voting power stand of record in the names of two (2) or more persons, whether fiduciaries, members of a partnership, joint tenants, tenants in common, tenants by the entirety, or otherwise, or if two (2) or more persons have the same fiduciary relationship respecting the same shares, unless the secretary is given written notice to the contrary and is furnished with a copy of the instrument or order appointing them or creating the relationship wherein it is so provided, their acts with respect to voting or giving consent shall have the following effect: (a) if only one (1) votes, such person’s act binds all; (b) if more than one (1) votes, the act of the majority so voting binds all; (c) if more than one (1) votes, but
the vote is evenly split on any particular matter, each faction may vote the securities in question proportionally. If the instrument filed with the Secretary shows that any such tenancy is held in unequal interests, shall be a majority or even split in interest.
2.10 Shareholder Decisions.
2.10.1 Majority Vote. When a quorum is present or represented at any shareholders’ meeting in person or by proxy and entitled to vote on the subject matter, action on any matter, other than the election of directors, is approved if the votes cast favoring the action exceed the votes cast opposing the action, unless the vote of a greater number is required by law, the articles of incorporation, or these bylaws, in which case the contrary provision is controlling.
2.10.2 Election of Directors. The number of director candidates equal to the number of directors to be elected who receive the largest number of votes cast by the shares entitled to vote in the election at a shareholders’ meeting at which a quorum is present will be elected to serve as members of the board of directors.
2.10.3 Shareholder Action by Consent. Any action required or permitted to be taken at a shareholders’ meeting may be taken without a meeting if a consent in writing or electronic transmission, setting forth the action so taken, is signed by the shareholders entitled to vote holding not less than the minimum number of shares that would be necessary to authorize or take such action at a meeting with respect to the subject matter thereof and the consent is delivered to the Corporation for inclusion in the minute book. If the act to be taken requires that notice be given to nonvoting shareholders, the Corporation must give the nonvoting shareholders written notice of the proposed action not more than ten (10) days after the action is taken, which notice must contain or be accompanied by the same material that would have been required if a formal meeting had been called to consider the action. A consent signed under this section has the effect of a meeting vote and may be described as such in any document. If more than one counterpart of a consent is signed by shareholders, all counterparts will be considered one document. If action is taken by less than unanimous written consent of the voting shareholders, the corporation shall give its nonconsenting voting shareholders written notice of the action not more than ten (10) days after written consents sufficient to take the action have been delivered to the corporation.
3
ARTICLE III
DIRECTORS
3.1 Corporate Powers. The business and affairs of the Corporation will be managed and controlled by the board of directors. All corporate powers must be exercised by, or under the authority of, the board of directors.
3.2 Number, Tenure, and Qualifications.
3.2.1 Number. The authorized number of directors of the corporation shall be fixed by resolution of the Board of Directors from time to time. The board of directors will consist of at least one (1), but no more than seven (7) individuals. If only one director is named in these bylaws or is serving at any time, words in the plural referring to directors will include the singular.
3.2.2 Term. Each of the initial directors will hold office until the first annual
shareholders’ meeting, and each director elected thereafter will hold office until the next annual shareholders’ meeting following his or her election. Despite the expiration of a director’s term, he or she will continue to serve until the election and qualification of his or her successor. However, the term of a director will terminate immediately on the director’s death, resignation, or removal, and the director will not continue to serve.
3.2.3 Qualifications. Unless required by the articles of incorporation, it is not necessary that directors be residents of the State of Wyoming or shareholders of the Corporation.
3.3 Annual Meetings. A regular annual meeting of the board of directors will be held immediately after the adjournment of the annual shareholders’ meeting and in the same place, if any, as the shareholders’ meeting.
3.4 Special Meetings. Special meetings of the board of directors may be called at any time by the President or by any director for any purpose. The person calling the special meeting of the board of directors may fix any place, if any, for holding the meeting within the county in which the Corporation has its principal place of business. Special meetings may be held in other locations with the consent of all directors.
3.5 Notice of Board of Directors Meetings.
3.5.1 Annual Meetings. No notice of annual meetings of the board of directors other than these bylaws is required.
3.5.2 Special Meetings. A special meeting of the board of directors must be preceded by at least a two days’ notice of the date, time, and place, if any, of the meeting. Unless otherwise required by the articles of incorporation, the notice of a special meeting must specify the purposes of the meeting or the business to be transacted.
3.5.3 Form and Effect. Notice of a special meeting of the board of directors may be given orally, in writing, or via electronic transmission. If notice is given orally, it will be effective when communicated if communicated in a comprehensible manner. If the notice is given in writing, it will be effective at the earliest of the following:
a. When received;
b. Five days after deposit in the United States mail, with postage prepaid, addressed to the director either at the director’s business office or the address of the director that appears on the Corporation’s current records of shareholders; or
c. On the date shown on the return receipt if sent by registered or certified mail, return receipt requested, and the receipt is signed on behalf of the addressee.
d. Electronic transmission is effective when it has been made to the data address of the shareholder or when it has been made in a manner otherwise authorized by the shareholder.
3.5.4 Waiver of Notice. A director entitled to notice may waive notice of a meeting of the board of directors by a signed document delivered to the secretary. The waiver will be effective whether signed before or after the meeting. A director’s attendance at a meeting waives objection to lack of notice or defective notice of the meeting, unless the director at the beginning of the meeting, or promptly upon the director’s arrival, objects to holding the meeting, or transacting business at the meeting and does not thereafter vote for or assent to action taken at the meeting.
4
3.5.5 Quorum. A majority of the number of directors in office immediately before the meeting begins constitutes a quorum for the transaction of business at any meeting of the board of directors.
3.6 Action by Board of Directors.
3.6.1 Director’s Meeting. The affirmative vote of a majority of directors present in person or by electronic communications at a meeting at which a quorum is present will be the act of the board of directors, unless the vote of a greater number of directors is required by law, the articles of incorporation, or these bylaws.
3.6.2 Written Consent. Any action required or permitted to be taken at a meeting of the board of directors may be taken without a meeting if a consent in writing, setting forth the action so taken, is signed by a majority of the directors and delivered to the Corporation for inclusion in the minute book. A consent signed under this section has the effect of a meeting vote and may be described as such in any document. If more than one counterpart of a consent is signed by directors, all counterparts will be considered one document.
3.6.3 Remote Meetings. Any director may participate in a meeting of the directors by means of electronic communication by which all persons participating in the meeting can hear each other during the meeting. A director participating in a meeting by this means is deemed to be present in person at the meeting.
3.7 Removal of Directors. Any director may be removed with or without cause by an affirmative vote of sixty-five percent (65%) the shareholders eligible to vote. A director may only be removed by the shareholders at a special meeting called for the purpose of removing the director, and the meeting notice must state that the purpose, or one of the purposes, of the meeting is removal of the director.
3.8 Board of Directors’ Vacancies. Unless the articles of incorporation provide otherwise, if a vacancy occurs on the board of directors, including a vacancy resulting from an increase in the number of directors, the shareholders may fill the vacancy by electing a director. During such time that the shareholders fail or are unable to fill a vacancy, the board of directors may fill the vacancy. If the directors remaining in office constitute less than a quorum of the board of directors, the remaining directors may fill the vacancy by the affirmative vote of a majority of their number. The term of a director elected to fill a vacancy expires at the next annual shareholders’ meeting (although the director may continue to serve after the expiration of the term as provided in the section of these bylaws relating to directors’ terms).
3.9 Compensation of Directors. By resolution of the board of directors, the directors may be paid their expenses, if any, of attendance at each meeting of the board of directors and may
be paid a fixed sum for attendance at each meeting or a stated salary as director. No such payments shall prevent any director from serving the Corporation in any other capacity and receiving compensation for that service.
3.10 Committees. The board of directors may create one or more committees and appoint members of the board of directors to serve on them. Each committee must have two or more members, who serve at the pleasure of the board of directors. Creation of a committee and appointment of members to it must be approved by a majority of all the directors in office when the action is taken. Any such committee will have and may exercise all of the authority granted by the board of directors for the management of the Corporation except to the extent such delegation of authority is prohibited by law.
3.11 Organization. Meetings of the board of directors shall be presided over by the chair of the board, or in the chair's absence by the chief executive officer, or in the absence of both by a director chosen at the meeting. The secretary shall act as secretary of the meeting, but in the absence of the secretary, the person presiding at the meeting may appoint any person to act as secretary of the meeting.
3.12 Presumption of Assent. A director of the Corporation who is present at a meeting of the board of directors at which action on any corporate matter is taken shall be presumed to have assented to the action taken unless: (i) the director objects at the beginning of the meeting, or promptly upon the director's arrival, to holding the meeting or transacting business at the meeting; (ii) such director's dissent or abstention from the action taken is entered in the minutes of the meeting; or (iii) the director delivers written notice of the director's dissent or abstention to the presiding officer of the meeting before the adjournment of the meeting or to the Corporation within a reasonable time after adjournment of the meeting. Such right to dissent shall not apply to a director who voted in favor of such action.
5
ARTICLE IV
OFFICERS
4.1 Appointment. The officers of this corporation will be a President, a Secretary, and a Treasurer, who will be appointed by the board of directors. The board of directors may appoint additional officers or assistant officers, from time to time. The same individual may simultaneously hold more than one office unless specifically prohibited therefrom by law. An officer may, but need not, be a shareholder or a member of the board of directors.
4.2 Term of Office. Each officer will hold office until the earlier of the following:
a. The officer’s successor is appointed;
b. The officer resigns; or
c. The officer is removed in accordance with the Bylaws hereof. The designation of a specified term does not grant any contract rights to any officer.
4.3 Removal. The board of directors may remove any officer at any time, with or without cause.
4.4 Resignation. Any officer may resign at any time by giving notice in writing or by
electronic transmission to the board of directors or to the President. Any such resignation shall be effective when received by the person or persons to whom such notice is given, unless a later time is specified therein, in which event the resignation shall become effective at such later time. Unless otherwise specified in such notice, the acceptance of any such resignation shall not be necessary to make it effective. Any resignation shall be without prejudice to the rights, if any, of the corporation under any contract with the resigning officer.
4.5 Compensation. The compensation of the officers of the Corporation, if any, will be fixed by the board of directors.
4.6 Duties. Each officer has the authority and must perform the duties set forth in these bylaws and to the extent consistent with these bylaws, the duties prescribed, from time to time, by the board of directors or by direction of an officer authorized by the board of directors to prescribe the duties of other officers.
4.7 President. The President is the Chief Executive Officer of the Corporation and, subject to the control of the board of directors, has responsibility for the conduct and management of the business and fiscal affairs of the Corporation and the general supervision of its property, business interests, and agents. The President, or a person designated by the President, must preside at all meetings of the shareholders and directors, unless otherwise ordered by the board of directors. The President may sign, with the secretary or any other proper officer of the Corporation authorized by the board of directors, certificates for shares of the Corporation and deeds, mortgages, bonds, contracts, or other instruments that the board of directors has authorized to be executed, except in cases where the signing and execution thereof is expressly delegated by the board of directors or these bylaws to some other officer or agent of the Corporation, or is required by law to be otherwise signed or executed.
4.8 Vice Presidents. In the absence of the president or in the event of the president's death, inability or refusal to act, the vice president (or in the event there shall be more than one vice president, the vice presidents in the order designated at the time of their appointment, or in the absence of any designation then in the order of their appointment) shall perform the duties of the president, and when so acting shall have all the powers of and be subject to all the restrictions upon the president; and shall perform such other duties as from time to time may be assigned to the vice president by the president or by the board of directors.
4.9 Secretary. The secretary must:
4.9.1 Minute Book. Keep or cause to be kept at the principal office, or such other place as the board of directors may order, a book of minutes of all meetings of directors and shareholders showing the time and place of the meeting, whether it was a regular or special meeting, and if a special meeting, how authorized, the notice given, the names of those present at directors’ meetings, the number of shareholders present or represented at shareholders’ meetings, and the proceedings at those meetings;
4.9.2 Stock Transfer Records. Keep or cause to be kept at the principal office of the Corporation, or at the office of the Corporation’s transfer agent, a stock transfer book, or a duplicate stock transfer book, showing the names of the shareholders and their addresses, the number and classes of shares held by each, the number and date
of certificates issued for such shares, if any, and the number and date of cancellation of certificates surrendered for cancellation, if any;
4.9.3 Notices. Give or cause to be given notice of the meetings of the shareholders and board of directors as is required by these bylaws;
4.9.4 Sign Documents. Sign with the president, or a vice-president if any, certificates for shares of the Corporation, if any, the issuance of which must have been authorized by resolution of the board of directors; and
4.9.5 Certification. When requested or required, certify any records of the Corporation.
6
4.10 Treasurer. The treasurer must:
4.10.1 Financial Administration. Be responsible for the administration of the financial affairs of the Corporation and have charge and custody of, and be responsible for, all funds and securities of the Corporation;
4.10.2 Receipts and Deposits. Receive and give receipts for moneys due and payable to the Corporation from any source whatsoever, and deposit all such money in the name of the Corporation in such banks, trust companies, or other depositories as must be selected by the board of directors; and
4.10.3 Bonding. If required by the board of directors, give a bond for the faithful discharge of the treasurer’s duties in such sums and with such security or surety as the board of directors may require.
ARTICLE V
SHARES OF CAPITAL STOCK & CERTIFICATES
5.1 Issuance of Stock. Shares of capital stock of any class now or hereafter authorized, securities convertible into or exchangeable for such stock, or options or other rights to purchase such stock or securities may be issued or granted in accordance with authority granted by resolution of the Board of Directors.
5.2 Stock Certificates. Shares of the capital stock of the Corporation shall be in the form adopted by the Board of Directors, represented by certificates or uncertificated, may be signed by the Chairman of the Board of Directors, or the President or Vice President and the Treasurer or the Assistant Treasurer or the Secretary or the Assistant Secretary of the Corporation, and may be sealed with the seal of the Corporation. If the shares of capital stock of the Corporation are represented by certificates, all such certificates shall be numbered consecutively, and the name of the person owning the shares represented thereby, with the number of such shares and the date of issue, shall be entered on the books of the Corporation.
5.2.1 Form. In the case that shares of capital stock of the Corporation are represented by certificates, certificates for shares will be in such form as the board of directors may determine. Each certificate for shares must state the following upon its face:
a. The name of the Corporation and that it is organized under the laws of Wyoming;
b. The name of the person to whom it is issued;
c. The number and class of shares and the designation of the series, if any, that the certificate represents; and
d. The number of the certificate and its date of issuance.
5.2.2 Signing Certificates. The certificates must be signed by the President, or a Vice-President, and the Secretary, or an assistant secretary.
5.2.3 Shares Without Certificates. The Corporation may issue shares without certificates.
5.2.4 Legends. Legends relating to restrictions on the transfer of shares may be marked on share certificates if required by an agreement to which the Corporation is a party or by action of the board of directors.
7
5.3 Transfer of Stock. Shares of capital stock of the Corporation shall be transferred only on the books of the Corporation, by the holder of record in person, by electronic transmission, or by the holder’s duly authorized representative, and in the case of certificated shares, only upon surrender to the Corporation of the certificate for such shares duly endorsed for transfer, together with such other documents (if any) as may be required to effect such transfer. If the shares of capital stock of the Corporation are represented by certificates, the Corporation must issue a new certificate to the person or persons entitled to the shares represented by that certificate, cancel the old certificate, and record the transaction upon its stock transfer books.
5.4 Lost, Stolen, Destroyed, or Mutilated Certificates. New stock certificates may be issued to replace certificates alleged to have been lost, stolen, destroyed, or mutilated, upon such terms and conditions, including proof of loss or destruction, and the giving of a satisfactory bond of indemnity, as the Board of Directors from time to time may determine.
5.5 Regulations. The Board of Directors shall have the power and authority to make all such rules and regulations not inconsistent with these Bylaws as it may deem expedient concerning the issue, transfer, and registration of shares of capital stock of the Corporation.
5.6 Record Date for Stock. In order that the Corporation may determine the stockholders entitled to notice of or to vote at any meeting of stockholders or any adjournment thereof, or to express consent to corporate action in writing without a meeting, or entitled to receive payment of any dividend or other distribution or allotment of any rights, or entitled to exercise any rights in respect of any change, conversion or exchange of stock or for the purpose of any other lawful action, the board of directors may fix, in advance, a record date, which shall not be more than sixty (60) nor less than ten (10) days before the date of such meeting, nor more than sixty (60) days prior to any other action. A determination of stockholders of record entitled to notice of or to vote at a
meeting of stockholders shall apply to any adjournment of the meeting: provided, however, that the board of directors may fix a new record date for the adjourned meeting.
5.7 List of Shareholders. The name and address of the person to whom shares are issued, or the identifier and data address to whom shares are issued to, with the number of shares and date of issue, must be entered on the stock transfer books of the Corporation.
8
ARTICLE VI
INDEMNIFICATION
6.1 Permissible Indemnification of Directors.
6.1.1 Good Faith Conduct. Unless otherwise provided in the articles of incorporation, the Corporation may indemnify any individual made a party to a proceeding because the individual is or was a director of the Corporation against liability incurred in the proceeding, but only if the individual demonstrates he or she conducted himself or herself in good faith and reasonably believed that his or her official conduct was in the best interests of the Corporation and his or her other conduct was not opposed to the Corporation’s best interests. If the proceeding involves the director’s conduct with respect to an employee benefit plan, it is sufficient that the director reasonably believed the conduct to be in the interests of the participants or beneficiaries of the plan. In the case of indemnification in connection with any criminal proceeding, the individual must also demonstrate that the individual had no reasonable cause to believe his or her conduct was unlawful. A determination that the individual has met these standards of conduct required for permissible indemnification must be made in accordance with the procedures set forth in the Wyoming Business Corporation Act.
6.1.2 Derivative Actions. Indemnification in connection with a proceeding by or in the right of the Corporation is limited to reasonable expenses incurred by the individual in connection with the proceeding.
6.1.3 Limit on Indemnification. The Corporation may not indemnify a director in connection with a proceeding by or in the right of the Corporation in which the director is adjudged liable to the Corporation, or in connection with any other proceeding charging improper personal benefit to the director, whether or not involving action in the director’s official capacity, in which the director is adjudged liable on the basis that the director received improper personal benefit.
6.2 Mandatory Indemnification of Directors. Unless the articles of incorporation provide to the contrary, the Corporation must indemnify a director against reasonable expenses incurred by the director in connection with the proceeding if the director is wholly successful, on the merits or otherwise, in the defense of any proceeding to which the director is a party because of being a director of the Corporation.
6.3 Advances for Expenses of Directors. Unless otherwise provided in the articles of incorporation, the Corporation may pay for or reimburse the reasonable expenses incurred by a director who is a party to a proceeding in advance of final disposition of the proceeding, if:
6.3.1 The director furnishes the Corporation a written affirmation of the director’s good faith belief that his or her conduct meets the standards of conduct required for permissible
indemnification;
6.3.2 The director furnishes the Corporation a written undertaking, executed personally or on the director’s behalf, to repay the advance if it is ultimately determined that the director did not meet the standard of conduct (which undertaking must be an unlimited general obligation of the director but need not be secured and may be accepted without reference to financial ability to make repayment); and
6.3.3 Those authorizing the reimbursement determine that the facts then known would not preclude permissible indemnification.
6.4 Indemnification of Officers, Agents, and Employees. Unless otherwise provided in the articles of incorporation, the board of directors may indemnify and advance expenses to any officer, employee, or agent of the Corporation, who is not a director of the Corporation, to any extent consistent with public policy, as determined by the general or specific action of the board of directors.
9
ARTICLE VII
CORPORATE RECORDS
7.1 Maintenance of Records. The Corporation must maintain adequate and correct books, records, and accounts of its business and properties. Such records include, without limitation, recorded minutes of all meetings of its shareholders and board of directors, a record of all actions taken by the shareholders or board of directors without a meeting, and a record of all actions taken by a committee of the board of directors in place of the board of directors on behalf of the Corporation, and all additional documents subject to shareholder inspection rights provided for in these bylaws, the articles, or the Act. All of these books, records, and accounts must be kept at the Corporation’s principal office.
7.2 Shareholder Inspection Rights. If a shareholder of the Corporation gives the Corporation written demand at least five business days before the date on which the shareholder wishes to inspect and copy, a shareholder is entitled to inspect and copy, during regular business hours at the Corporation’s principal office, the following records:
7.2.1 The articles of incorporation of the Corporation and all amendments or restatements;
7.2.2 The bylaws of the Corporation and all amendments or restatements;
7.2.3 Resolutions adopted by its board of directors creating one or more classes or series of shares, and fixing their relative rights, preferences, and limitations, if shares issued pursuant to those resolutions are outstanding;
7.2.4 The minutes of all shareholders’ meetings, and records of all actions taken by shareholders without a meeting, for the past three years;
7.2.5 All written communications to shareholders generally within the past three years;
7.2.6 A list of the names and business addresses of the Corporation’s current directors and officers; and
7.2.7 The Corporation’s most recent annual report delivered to the Secretary of State.
ARTICLE VIII
DISTRIBUTIONS
The board of directors may authorize, and the Corporation may make, distributions (including dividends on its outstanding shares) in the manner and on the terms and conditions provided by law and the articles of incorporation. Before payment of any dividend, there may be set aside out of any funds of the Corporation available for dividends such sum or sums as the board of directors from time to time, in their absolute discretion, think proper as a reserve or reserves to meet contingencies, or for equalizing dividends, or for repairing or maintaining any property of the corporation, or for such other purpose as the board of directors shall think conducive to the interests of the corporation, and the board of directors may modify or abolish any such reserve in the manner in which it was created.
ARTICLE IX
CONTRACTS, LOANS, CHECKS, DEPOSITS
9.1 Contracts. The board of directors may authorize any officer or officers, agent or agents, to enter into any contract or execute and deliver any instrument in the name of and on behalf of the Corporation, and such authority may be general or confined to specific instances.
A director or officer of the Corporation shall not be disqualified by the director's office from dealing or contracting with the Corporation either as a vendor, purchaser, or otherwise. The fact that any director or officer, or any firm of which any director or officer of the Corporation is a shareholder, officer or director, is in any way interested in any transaction of the Corporation shall not make such transaction void or voidable, or require such director or officer of the Corporation to account to the Corporation for any profits therefrom, so long as the transaction/deal satisfies the requirements of Wyo. Stat. § 17-16-863 et seq.
9.2 Loans. No loans shall be made, or accepted, on behalf of the Corporation, and no evidences of indebtedness shall be issued in the Corporation's name, unless authorized by a resolution of the board of directors. Such authority may be general or confined to specific instances.
9.3 Checks, Drafts, Notes. All checks, drafts, or other orders for the payment of money, notes, or other evidences of indebtedness issued in the name of the Corporation shall be signed by such officer or officers, agent or agents of the Corporation and in such manner as shall from time to time be determined by resolution of the board of directors.
9.4 Deposits. All funds of the Corporation not otherwise employed shall be deposited from time to time to the credit of the Corporation in such banks, trust companies, or other depositories as the board of directors may select.
9.5 Voting Securities Held by the Corporation. The chief executive officer, or such other
officer or agent designated by the board of directors, shall have full power and authority on behalf of the Corporation to attend, act at, and vote at any meeting of security or interest holders of other corporations or entities in which the Corporation may hold securities or interests. At the meeting, the chief executive officer or other designated agent shall possess and exercise any and all rights and powers incident to the ownership of the securities or interest with the Corporation holds.
10
ARTICLE X
CORPORATE SEAL
The board of directors may, but is not required to, provide a corporate seal, which will be circular in form and have inscribed thereon any designation, including the name of the Corporation, the state of its incorporation, and the words “corporate seal.”
ARTICLE XI
AMENDMENTS
11.1 Board of Directors. The board of directors may amend or repeal these bylaws as provided by law or the articles of incorporation, unless the shareholders, in adopting, amending, or repealing a particular bylaw provide expressly that the board of directors may not amend or repeal that bylaw.
11.2 Shareholders. Approval by the majority of the shareholders may also amend or repeal these bylaws.
ARTICLE XII
RATIFICATION OF DEFECTIVE CORPORATE ACTION
12.1 Definitions. The following words have the meaning as used in this Article:
12.1.1 “Corporate action” means any action taken by or on behalf of the Corporation, including any action taken by the incorporator, the board of directors, a committee of the board of directors, an officer or agent of the Corporation or the shareholders.
12.1.2 “Date of the defective corporate action” means the date (or the approximate date, if the exact date is unknown) the defective corporate action was purported to have been taken.
12.1.3 “Defective corporate action” means (i) any corporate action purportedly taken that is, and at the time such corporate action was purportedly taken would have been, within the power of the Corporation, but is void or voidable due to a failure of authorization, and (ii) an overissue.
12.1.4 “Failure of authorization” means the failure to authorize, approve or otherwise effect a corporate action in compliance with the provisions of this Act, the articles of incorporation or bylaws, a corporate resolution or any plan or agreement to which the Corporation is a party, if and to the extent such failure would render such corporate action void or voidable.
12.1.5 “Overissue” means the purported issuance of:
a. shares of a class or series in excess of the number of shares of a class or series the Corporation has the power to issue under Article V at the time of such issuance; or
b. shares of any class or series that is not then authorized for issuance by the articles of incorporation.
12.1.6 “Putative shares” means the shares of any class or series (including shares issued upon exercise of rights, options, warrants or other securities convertible into shares of the Corporation, or interests with respect to such shares) that were created or issued as a result of a defective corporate action, that (i) but for any failure of authorization would constitute valid shares, or (ii) cannot be determined by the board of directors to be valid shares.
12.1.7 “Valid shares” means the shares of any class or series that have been duly authorized and validly issued in accordance with the Act, including as a result of ratification or amendment under this Article XII.
12.1.8 “Amendment effective time” with respect to any defective corporate action ratified under this Article XII means the later of:
a. the time at which the ratification of the defective corporate action is approved by the shareholders, or if approval of shareholders is not required, the time at which the notice required becomes effective; and
b. the time at which any articles of amendment filed become effective.
The amendment effective time shall not be affected by the filing or pendency of a judicial proceeding under section 12.8 or otherwise, unless otherwise ordered by the court.
11
12.2 Defective Corporate Action.
12.2.1 A defective corporate action shall not be void or voidable if ratified in accordance with section 12.3 or validated in accordance with section 12.8.
12.2.2 Ratification under section 12.3 or amendment under section 12.8 shall not be deemed to be the exclusive means of ratifying or validating any defective corporate action, and the absence or failure of ratification in accordance with this Article XII shall not, of itself, affect the validity or effectiveness of any corporate action properly ratified under common law or otherwise, nor shall it create a presumption that any such corporate action is or was a defective corporate action or void or voidable.
12.2.3 In the case of an overissue, putative shares shall be valid shares effective as of the date originally issued or purportedly issued upon:
a. the effectiveness under this Article XII and under Article XI of an amendment to the articles of incorporation authorizing, designating or creating such shares; or
b. the effectiveness of any other corporate action under this Article XII ratifying the authorization, designation or creation of such shares.
12.3 Ratification of Defective Corporate Actions.
12.3.1 To ratify a defective corporate action under this section (other than the ratification of an election of the initial board of directors under subsection (2)), the board of directors shall take action ratifying the action in accordance with section 12.4, stating:
a. the defective corporate action to be ratified and, if the defective corporate action involved the issuance of putative shares, the number and type of putative shares purportedly issued;
b. the date of the defective corporate action;
c. the nature of the failure of authorization with respect to the defective corporate action to be ratified; and
d. that the board of directors approves the ratification of the defective corporate action.
12.3.2 In the event that a defective corporate action to be ratified relates to the election of the board of directors of the Corporation under section 2.1, a majority of the persons who, at the time of the ratification, are exercising the powers of directors may take an action stating:
a. the name of the person or persons who first took action in the name of the Corporation as the initial board of directors of the Corporation;
b. the earlier of the date on which such persons first took such action or were purported to have been elected as the initial board of directors; and
c. that the ratification of the election of such person or persons as the initial board of directors is approved.
12.3.3 If any provision of the Wyoming Business Corporation Act, the articles of incorporation or bylaws, any corporate resolution or any plan or agreement to which the Corporation is a party in effect at the time action under subsection (a) is taken requires shareholder approval or would have required shareholder approval at the date of the occurrence of the defective corporate action, the ratification of the defective corporate action approved in the action taken by the directors under subsection (a) shall be submitted to the shareholders for approval in accordance with section 12.4.
12.3.4 Unless otherwise provided in the action taken by the board of directors under subsection (a), after the action by the board of directors has been taken and, if required, approved by the shareholders, the board of directors may abandon the ratification at any time before the amendment effective time without further action of the shareholders.
12
12.4 Action on Ratification.
12.4.1 The quorum and voting requirements applicable to a ratifying action by the board of directors under section 12.3(1) shall be the quorum and voting requirements applicable to
the corporate action proposed to be ratified at the time such ratifying action is taken.
12.4.2 If the ratification of the defective corporate action requires approval by the shareholders under section 12.3(3), and if the approval is to be given at a meeting, the Corporation shall notify each holder of valid and putative shares, regardless of whether entitled to vote, as of the record date for notice of the meeting and as of the date of the occurrence of defective corporate action, provided that notice shall not be required to be given to holders of valid or putative shares whose identities or addresses for notice cannot be determined from the records of the Corporation. The notice must state that the purpose, or one of the purposes, of the meeting, is to consider ratification of a defective corporate action and must be accompanied by (i) either a copy of the action taken by the board of directors in accordance with section 12.3(1) or the information required by sections 12.3(1)(a) through (1)(d), and (ii) a statement that any claim that the ratification of such defective corporate action and any putative shares issued as a result of such defective corporate action should not be effective, or should be effective only on certain conditions, shall be brought within 120 days from the applicable amendment effective time.
12.4.3 Except as provided in subsection (d) with respect to the voting requirements to ratify the election of a director, the quorum and voting requirements applicable to the approval by the shareholders required by section 12.3(3) shall be the quorum and voting requirements applicable to the corporate action proposed to be ratified at the time of such shareholder approval.
12.4.4 The approval by shareholders to ratify the election of a director requires that the votes cast within the voting group favoring such ratification exceed the votes cast opposing such ratification of the election at a meeting at which a quorum is present.
12.4.5 Putative shares on the record date for determining the shareholders entitled to vote on any matter submitted to shareholders under section 12.3(3) (and without giving effect to any ratification of putative shares that becomes effective as a result of such vote) shall neither be entitled to vote nor counted for quorum purposes in any vote to approve the ratification of any defective corporate action.
12.4.6 If the approval under this section of putative shares would result in an overissue, in addition to the approval required by section 12.3, approval of an amendment to the articles of incorporation under Article XI to increase the number of shares of an authorized class or series or to authorize the creation of a class or series of shares so there would be no overissue shall also be required.
12.5 Notice Requirements.
12.5.1 Unless shareholder approval is required under section 12.3(3), prompt notice of an action taken under section 12.3 shall be given to each holder of valid and putative shares, regardless of whether entitled to vote, as of (i) the date of such action by the board of directors and (ii) the date of the defective corporate action ratified, provided that notice shall not be required to be given to holders of valid and putative shares whose identities or addresses for notice cannot be determined from the records of the Corporation.
12.5.2 The notice must contain (i) either a copy of the action taken by the board of directors in accordance with section 12.3(1) or (2) or the information required by sections 12.3(1)(a)
through (1)(d) or sections 12.3(2)(a) through (2)(c), as applicable, and (ii) a statement that any claim that the ratification of the defective corporate action and any putative shares issued as a result of such defective corporate action should not be effective, or should be effective only on certain conditions, shall be brought within 120 days from the applicable amendment effective time.
12.5.3 No notice under this section is required with respect to any action required to be submitted to shareholders for approval under section 12.3(3) if notice is given in accordance with section 12.4(2).
12.5.4 A notice required by this section may be given in any manner permitted by section 2.5 and, for any corporation subject to the reporting requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, may be given by means of a filing or furnishing of such notice with the United States Securities and Exchange Commission.
12.6 Effect of Ratification. From and after the amendment effective time, and without regard to the 120-day period during which a claim may be brought under section 12.8:
12.6.1 Each defective corporate action ratified in accordance with section 12.3 shall not be void or voidable as a result of the failure of authorization identified in the action taken under section 12.3(1) or (2) and shall be deemed a valid corporate action effective as of the date of the defective corporate action;
12.6.2 The issuance of each putative share or fraction of a putative share purportedly issued pursuant to a defective corporate action identified in the action taken under section 12.3 shall not be void or voidable, and each such putative share or fraction of a putative share shall be deemed to be an identical share or fraction of a valid share as of the time it was purportedly issued; and
12.6.3 Any corporate action taken subsequent to the defective corporate action ratified in accordance with this Article XII in reliance on such defective corporate action having been validly effected and any subsequent defective corporate action resulting directly or indirectly from such original defective corporate action shall be valid as of the time taken.
13
12.7 Filings.
12.7.1 If the defective corporate action ratified under this Article XII would have required under any other section of the Act a filing in accordance with the Act, then, regardless of whether a filing was previously made in respect of such defective corporate action and in lieu of a filing otherwise required by this Act, the Corporation shall file articles of amendment in accordance with this section, and such articles of amendment shall serve to amend or substitute for any other filing with respect to such defective corporate action required by this Act.
12.7.2 The articles of amendment must set forth:
a. the defective corporate action that is the subject of the articles of amendment (including, in the case of any defective corporate action involving the issuance of putative shares, the number and type of putative shares issued and the date or dates upon which such putative shares were purported to have been issued);
b. the date of the defective corporate action;
c. the nature of the failure of authorization in respect of the defective corporate action;
d. a statement that the defective corporate action was ratified in accordance with section 12.3, including the date on which the board of directors ratified such defective corporate action and the date, if any, on which the shareholders approved the ratification of such defective corporate action; and
e. the information required by subsection (3).
12.7.3 The articles of amendment must also contain the following information:
a. if a filing was previously made in respect of the defective corporate action and no changes to such filing are required to give effect to the ratification of such defective corporate action in accordance with section 1.47, the articles of amendment must set forth (i) the name, title and filing date of the filing previously made and any articles of correction to that filing and (ii) a statement that a copy of the filing previously made, together with any articles of correction to that filing, is attached as an exhibit to the articles of amendment;
b. if a filing was previously made in respect of the defective corporate action and such filing accordance with section 12.3, the articles of amendment must set forth (i) the name, title and filing date of the filing previously made and any articles of correction to that filing and (ii) a statement that a filing containing all of the information required to be included under the applicable section or sections of the Act to give effect to such defective corporate action is attached as an exhibit to the articles of amendment, and (iii) the date and time that such filing is deemed to have become effective; or
c. if a filing was not previously made in respect of the defective corporate action and the defective corporate action ratified under section 12.3 would have required a filing under any other section of the Act, the articles of amendment must set forth (i) a statement that a filing containing all of the information required to be included under the applicable section or sections of the Act to give effect to such defective corporate action is attached as an exhibit to the articles of amendment, and (ii) the date and time that such filing is deemed to have become effective.
14
ARTICLE XIII
MISCELLANEOUS
13.1 Articles of Incorporation. In the event of any conflict between the provisions of the articles of incorporation and the bylaws, the provisions of the articles of incorporation shall govern.
13.2 Severability. If any provision of these bylaws shall be held to be invalid, illegal, unenforceable, or in conflict with the articles of incorporation, then such provision shall nonetheless be enforced to the maximum extent possible consistent with such holding and the remaining provisions of these bylaws (including without limitation, all portions of any section of these bylaws containing any such provision held to be invalid, illegal, unenforceable, or in conflict with the articles of incorporation) that
are not themselves invalid, illegal, unenforceable, or in conflict with the articles of incorporation shall remain in full force and effect.
13.3 Fiscal Year. The fiscal year of the Corporation shall be fixed by resolution of the board of directors consistent with the period authorized or required by the Internal Revenue Code.
15
CERTIFICATE OF DIRECTOR(S)
I, Joseph McGinley, hereby certify that I am the Director of McGinley Orthopaedic Innovations, Inc., a Wyoming corporation and that the foregoing Bylaws, comprising twenty-one (21) pages, including this page, have been adopted by the Board of Directors as the Bylaws and governing instrument of the Corporation as of the ____ day of 2022.
IN WITNESS WHEREOF, the undersigned have executed this Certificate on the ____ day of ______________ 2022.
MCGINLEY ORTHOPAEDIC INNOVATIONS, INC.
________________________
Joseph McGinley, Director
MCGINLEY ORTHOPAEDIC INNOVATIONS, INC., A WYOMING CORPORATION
COMMON STOCK SHARES
REGULATION A+ SUBSCRIPTION AGREEMENT
Investing in securities represented by shares of common stock (“Shares”) of McGinley Orthopaedic Innovations, Inc. (the “Company”) involves significant risks. This investment is suitable only for persons who can afford to lose their entire investment and such investment could be illiquid for an indefinite period of time. No public market currently exists for the Shares, and if a public market develops following this offering, it may not continue.
The Shares have not been registered under the Securities Act of 1933, as amended (the “Securities Act”), or any state securities or blue-sky laws and are being offered and sold in reliance on exemptions from the registration requirements of the Securities Act and state securities or blue-sky laws. Although an offering statement has been filed with the Securities and Exchange Commission (the “SEC”), that offering statement does not include the same information that would be included in a registration statement under the Securities Act. The Shares have not been approved or disapproved by the SEC, any state securities commission or other regulatory authority, nor have any of the foregoing authorities passed upon the merits of this offering or the adequacy or accuracy of the offering circular or any other materials or information made available to subscriber in connection with this offering, through the online website platform https://www. [ ], (the “Platform” or “Portal”) or the SEC’s EDGAR website at www.sec.gov.
No sale may be made to persons in this offering who are not “accredited investors” if the aggregate purchase price is more than 10% of the greater of such investors’ annual income or net worth. The Company is relying on the representations and warranties set forth by each subscriber in this Subscription Agreement and the other information provided by subscriber in connection with this offering to determine compliance with this requirement.
Prospective investors may not treat the contents of the Subscription Agreement, the offering circular or any of the other materials available (collectively, the “Offering Materials”) or any prior or subsequent communications from the Company or any of its affiliates, officers, employees or agents as investment, legal or tax advice. In making an investment decision, investors must rely on their own examination of the Company and the terms of this offering, including the merits and the risks involved. Each prospective investor should consult the investor’s own counsel, accountant and other professional advisor as to investment, legal, tax and other related matters concerning the investor’s proposed investment.
The Company reserves the right in its sole discretion and for any reason whatsoever to modify, amend and/or withdraw all or a portion of the offering and/or accept or reject in whole or in part any prospective investment in the Shares or to allot to any prospective investor less than the amount of Shares such investor desires to purchase.
Except as otherwise indicated, the Offering Materials speak as of their date. Neither the delivery nor the purchase of the Shares shall, under any circumstances, create any implication that there has been no change in the affairs of the Company since that date.
This agreement (“Subscription Agreement,” or “Agreement”) is made as of the date set forth below by and between the undersigned (“Subscriber”) and the Company and is intended to set forth certain representations, covenants and agreements between Subscriber and the Company with respect to the offering (the “Offering”) for sale by the Company of the Shares as described in the Company’s Offering Circular dated [ ], 2022 (the “Offering Circular”), a copy of which has been delivered to Subscriber. The Shares are also referred to herein as the “Securities.”
SUBSCRIPTION
1. Subscription. Subject to the terms and conditions hereof, Subscriber hereby irrevocably subscribes for and agrees to purchase from the Company the number of Shares set forth on the Subscription Agreement Signature Page, and the Company agrees to sell such Shares to Subscriber at a purchase price of $5.25 per Share for the total amount set forth on the Subscription Agreement Signature Page (the “Purchase Price”), subject to the Company’s right to sell to Subscriber such lesser number of Shares as the Company may, in its sole discretion, deem necessary or desirable.
2. Delivery of Subscription Amount; Acceptance of Subscription; Delivery of Securities. Subscriber understands and agrees that this Subscription is made subject to the following terms and conditions:
a. Contemporaneously with the execution and delivery of this Agreement through the Platform, Subscriber shall pay the Purchase Price for the Shares in the form of ACH debit transfer, wire transfer, or credit card. Your subscription is irrevocable. The escrow agent (the “Escrow Agent”) appointed by the Company will maintain all such funds for Subscriber’s benefit until the earliest to occur of: (i) the Closing (as defined below), (ii) the rejection of such subscription or (iii) the termination of the Offering by the Company in its sole discretion;
b. Payment of the Purchase Price shall be made by Subscriber via the Portal, and shall be held in escrow by the Escrow Agent until Closing, after which time, the funds tendered by Subscriber will be available to the Company;
c. This subscription shall be deemed to be accepted only when this Agreement has been signed by an authorized officer or agent of the Company (the “Closing”), and the deposit of the payment of the Purchase Price for clearance will not be deemed an acceptance of this Agreement;
d. The Company shall have the right to reject this subscription, in whole or in part;
e. The payment of the Purchase Price (or, in the case of rejection of a portion of the Subscriber’s subscription, the part of the payment relating to such rejected portion) will be returned promptly, without interest or deduction, if Subscriber’s subscription is rejected in whole or in part or if the Offering is withdrawn or canceled;
f. Subscriber shall receive notice and evidence of the digital entry (or other manner of record) of the number of the Shares owned by Subscriber reflected on the books and records of the Company and verified by the company’s transfer agent (the “Transfer Agent”), which books and records shall bear a notation that the Shares were sold in reliance upon Regulation A;
g. The Offering is described in the Offering Circular, that is available through the online website platform https://www.[ ], or the SEC’s EDGAR website at www.sec.gov. Please read this Agreement, the Offering Circular, and the Exhibits attached to the Offering Circular including, but not limited to, the Articles of Incorporation of McGinley Orthopaedic Innovations, Inc. (the “Articles”) and its bylaws (the “Bylaws”). While they are subject to change, as described below, the Company advises you to print and retain a copy of these documents for your records.
REPRESENTATIONS AND WARRANTIES OF SUBSCRIBER
By executing this Subscription Agreement, Subscriber (and, if Subscriber is purchasing the Securities subscribed for hereby in a fiduciary capacity, the person or persons for whom Subscriber is so purchasing) represents and warrants, which representations and warranties are true and complete in all material respects as of the date of the Closing:
3. Requisite Power and Authority. Such Subscriber has all necessary power and authority under all applicable provisions of law to execute and deliver this Subscription Agreement. All action on Subscriber’s part required for the lawful execution and delivery of this Subscription Agreement has been or will be effectively taken prior to the Closing. Upon execution and delivery, this Subscription Agreement will be a valid and binding obligation of Subscriber, enforceable in accordance with its terms, except (a) as limited by applicable bankruptcy, insolvency, reorganization, moratorium or other laws of general application affecting enforcement of creditors’ rights and (b) as limited by general principles of equity that restrict the availability of equitable remedies.
4. Investment Representations. Subscriber understands that the Securities have not been registered under the Securities Act. Subscriber also understands that the Securities are being offered and sold pursuant to an exemption from registration contained in the Securities Act based in part upon Subscriber’s representations contained in this Agreement. Subscriber is purchasing the Shares for Subscriber’s own account. Subscriber has received and reviewed this Agreement, The Offering Circular, the Articles and the Bylaws. Subscriber and/or Subscriber’s advisors, who are not affiliated with and not compensated directly or indirectly by the Company or an affiliate thereof, have such knowledge and experience in business and financial matters as will enable them to utilize the information which they have received in connection with the Offering to evaluate the merits and risks of an investment, to make an informed investment decision and to protect Subscriber’s own interest in connection with an investment in the Shares.
5. Illiquidity and Continued Economic Risk. Subscriber acknowledges and agrees that there is no ready public market for the Securities and that there is no guarantee that a market for
their resale will ever exist. Subscriber must bear the economic risk of this investment indefinitely and the Company has no obligation to list the Securities on any market or take any steps (including registration under the Securities Act or the Securities Exchange Act of 1934, as amended) with respect to facilitating trading or resale of the Securities. Subscriber acknowledges that Subscriber is able to bear the economic risk of losing Subscriber’s entire investment in the Securities. Subscriber also understands that an investment in the Company involves significant risks and has taken full cognizance of and understands all of the risk factors relating to the purchase of Securities.
6. Accredited Investor Status or Investment Limits. Subscriber represents that either:
a. Subscriber is an “accredited investor” within the meaning of Rule 501 of Regulation D under the Securities Act; or
b. The Purchase Price set out below, on the signature page of this Agreement, together with any other amounts previously used to purchase Securities in this Offering, does not exceed 10% of the greater of the Subscriber’s annual income or net worth. Subscriber represents that to the extent it has any questions with respect to its status as an accredited investor, or the application of the investment limits, it has sought professional advice.
7. Additional Subscriber Information; Payment Information. Subscriber agrees to provide any additional documentation the Company may reasonably request, including documentation as may be required by the Company to form a reasonable basis that the Subscriber qualifies as an “accredited investor” as that term is defined in Rule 501 under Regulation D promulgated under the Act, or otherwise as a “qualified purchaser” as that term is defined in Regulation A promulgated under the Act, or as may be required by the securities administrators or regulators of any state, to confirm that the Subscriber meets any applicable minimum financial suitability standards and has satisfied any applicable maximum investment limits. Subscriber acknowledges that Subscriber’s responses to questions on the Platform are true, complete and accurate in all respects. Payment information provided by Subscriber through the Platform is true, accurate and correct and such payment information shall be deemed to be a part of this Agreement as if and to the same extent that such information was set forth herein.
8. Company Information. Subscriber has read the Offering Circular filed with the SEC, including the section titled “Risk Factors.” Subscriber understands that the Company is subject to all the risks that apply to early-stage companies, whether or not those risks are explicitly set out in the Offering Circular. Subscriber acknowledges that no representations or warranties have been made to Subscriber, or to Subscriber’s advisors or representative, by the Company or others with respect to the business or prospects of the Company or its financial condition.
9. Neither the Company nor the Platform is an Investment Advisor. Subscriber understands that neither the Company nor the Platform is registered under the Investment Company Act of 1940 or the Investment Advisers Act of 1940.
10. Valuation. Subscriber acknowledges that the price of the Securities was set by the Company on the basis of the Company’s internal valuation and no warranties are made as to value.
Subscriber further acknowledges that future offerings of Securities may be made at lower valuations, with the result that the Subscriber’s investment will bear a lower valuation.
11. Domicile. Subscriber maintains Subscriber’s domicile (and is not a transient or temporary resident) at the address shown on the signature page and provided on the Platform.
12. Power of Attorney. Any power of attorney of the Subscriber granted in favor of the Company has been executed by the Subscriber in compliance with the laws of the state, province or jurisdiction in which such agreements were executed.
13. No Brokerage Fees. Other than commissions payable to Rialto Markets, a licensed broker-dealer, as placement agent, there are no claims for brokerage commission, finders’ fees or similar compensation in connection with the transactions contemplated by this Subscription Agreement or related documents based on any arrangement or agreement binding upon Subscriber. Subscriber will indemnify and hold the Company harmless against any liability, loss or expense (including, without limitation, reasonable attorneys’ fees and out-of-pocket expenses) arising in connection with any such claim.
14. Foreign Investors. If Subscriber is not a United States person (as defined by Section 7701(a)(30) of the Internal Revenue Code of 1986, as amended), Subscriber hereby represents that it has satisfied itself as to the full observance of the laws of its jurisdiction in connection with any invitation to subscribe for the Securities or any use of this Subscription Agreement, including (a) the legal requirements within its jurisdiction for the purchase of the Securities, (b) any foreign exchange restrictions applicable to such purchase, (c) any governmental or other consents that may need to be obtained, and (d) the income tax and other tax consequences, if any, that may be relevant to the purchase, holding, redemption, sale, or transfer of the Securities. Subscriber’s subscription and payment for and continued beneficial ownership of the Securities will not violate any applicable securities or other laws of the Subscriber’s jurisdiction.
15. Terms and Conditions of the Platform. Subscriber acknowledges that it has read, understands and agrees to the terms and conditions, privacy policy and disclaimers on the Platform.
16. Transfer Restrictions. Subscriber acknowledges and agrees that the Shares may be subject to restrictions on transfer pursuant to applicable federal and state laws, the Company’s Articles, and this Agreement. The Shares may not be sold, offered for sale, pledged or hypothecated in the absence of a registration statement in effect with respect to the Shares under the Securities Act or an opinion of legal counsel satisfactory to the Company that such registration is not required or unless the Shares are sold pursuant to Rule 144 or Rule 144A of the Securities Act. The Shares shall bear a digital or physical restrictive legend in substantially the following form (and a stop transfer order may be placed against transfer of such certificates or instruments):
THE SECURITIES EVIDENCED BY THIS CERTIFICATE HAVE NOT BEEN REGISTERED UNDER THE SECURITES ACT OF 1933 AND THEY ARE SUBJECT TO SIGNIFICANT RESTRICTIONS ON TRANSFER PURSUANT TO APPLICABLE FEDERAL AND STATE LAWS, THE COMPANY’S ARTICLES OF INCORPORATION AND THE SUBSCRIPTION AGREEMENT PURSUANT TO WHICH THESE SECURITIES WERE ORIGINALLY SOLD.
THEY MAY NOT BE SOLD, OFFERED FOR SALE, PLEDGED OR HYPOTHECATED IN THE ABSENCE OF A REGISTRATION STATEMENT IN EFFECT WITH RESPECT TO THE SECURITIES UNDER SUCH ACT OR AN OPINION OF COUNSEL SATISFACTORY TO THE COMPANY THAT SUCH REGISTRATION IS NOT REQUIRED OR UNLESS SOLD PURSUANT TO RULE 144 OR RULE 144A OF SUCH ACT.
ANY PURPORTED TRANSFER IN VIOLATION OF SUCH PROVISIONS SHALL BE VOID, AB INITIO.
SURVIVAL; INDEMNIFICATION
17. Survival; Indemnification. All representations, warranties and covenants contained in this Agreement and the indemnification contained herein shall survive (a) the acceptance of this Agreement by the Company, (b) changes in the transactions, documents and instruments described herein which are not material or which are to the benefit of Subscriber, and (c) the death or disability of Subscriber. Subscriber acknowledges the meaning and legal consequences of the representations, warranties and covenants herein, and that the Company has relied upon such representations, warranties and covenants in determining Subscriber’s qualification and suitability to purchase the Securities. Subscriber hereby agrees to indemnify, defend and hold harmless the Company, its officers, directors, employees, agents and controlling persons, from and against any and all losses, claims, damages, liabilities, expenses (including attorneys’ fees and disbursements), judgments or amounts paid in settlement of actions arising out of or resulting from the untruth of any representation of Subscriber herein or the breach of any warranty or covenant herein by Subscriber. Notwithstanding the foregoing, however, no representation, warranty, covenant or acknowledgment made herein by Subscriber shall in any manner be deemed to constitute a waiver of any rights granted to it under the Securities Act or state securities laws.
MISCELLANEOUS PROVISIONS
18. Caption and Headings. The Article and Section headings throughout this Agreement are for convenience of reference only and shall in no way be deemed to define, limit or add to any provision of this Agreement.
19. Notification of Changes. Subscriber agrees and covenants to notify the Company immediately upon the occurrence of any event prior to the consummation of this Offering that would cause any representation, warranty, covenant or other statement contained in this Agreement to be false or incorrect or of any change in any statement made herein occurring prior to the consummation of this Offering.
20. Assignability. This Agreement is not assignable by Subscriber, and may not be modified, waived or terminated except by an instrument in writing signed by the party against whom enforcement of such modification, waiver or termination is sought.
21. Binding Effect. Except as otherwise provided herein, this Agreement shall be binding upon and inure to the benefit of the parties and their heirs, executors, administrators, successors, legal representatives and assigns, and the agreements, representations, warranties and
acknowledgments contained herein shall be deemed to be made by and be binding upon such heirs, executors, administrators, successors, legal representatives and assigns.
22. Obligations Irrevocable. The obligations of Subscriber shall be irrevocable, except with the consent of the Company, until the consummation or termination of the Offering.
23. Entire Agreement; Amendment. This Agreement states the entire agreement and understanding of the parties relating to the matters contained herein, superseding all prior contracts or agreements, whether oral or written. No amendment of the Agreement shall be made without the express written consent of the parties.
24. Severability. The invalidity or unenforceability of any particular provision of this Agreement shall not affect any other provision hereof, which shall be construed in all respects as if such invalid or unenforceable provision were omitted.
25. Notices. All notices and communications to be given or otherwise made to the Subscriber shall be deemed to be sufficient if sent by electronic mail to such address as set forth for the Subscriber at the records of the Company (or that you submitted to us via the Platform). You shall send all notices or other communications required to be given hereunder to the Company via email at [ ] (with a copy to be sent concurrently via prepaid certified mail to: McGinley Orthopaedic Innovations, Inc., [ ], Attention: Investor Relations. Any such notice or communication shall be deemed to have been delivered and received on the first business day following that on which the electronic mail has been sent (assuming that there is no error in delivery). As used in this Section, “business day” shall mean any day other than a day on which banking institutions in the State of Wyoming are legally closed for business.
26. Counterparts. This Agreement may be executed in any number of counterparts, each of which when so executed and delivered shall be deemed to be an original and all of which together shall be deemed to be one and the same agreement.
27. Digital Signatures. Digital (“electronic”) signatures, often referred to as an “e-signature”, enable paperless contracts and help speed up business transactions. The 2001 E-Sign Act was meant to ease the adoption of electronic signatures. The mechanics of this Subscription Agreement’s electronic signature include your signing this Agreement below by typing in your name, with the underlying software recording your IP address, your browser identification, the timestamp, and a securities hash within an SSL encrypted environment. This electronically signed Subscription Agreement will be available to both you and the Company, as well as any associated brokers, so they can store and access it at any time, and it will be stored and accessible on the Platform and hosting provider, including backups. You and the Company each hereby consents and agrees that electronically signing this Agreement constitutes your signature, acceptance and agreement as if actually signed by you in writing. Further, all parties agree that no certification authority or other third-party verification is necessary to validate any electronic signature; and that the lack of such certification or third-party verification will not in any way affect the enforceability of your signature or resulting contract between you and the Company. You understand and agree that your e-signature executed in conjunction with the electronic submission of this Subscription Agreement shall be legally binding and such transaction shall be considered authorized by you.
By signing electronically below, you agree your electronic signature is the legal equivalent of your manual signature on this Subscription Agreement and you consent to be legally bound by this Subscription Agreement’s terms and conditions. Alternatively, you may opt-out of this provision by printing a copy of this Agreement, signing it manually and returning it to the Company and, if your subscription is accepted, the Company will manually countersign it and return a countersigned copy to you via email.
28. Consent to Electronic Delivery of Tax Documents. Please read this disclosure about how the Company will provide certain documents that it is required by the Internal Revenue Service (the “IRS”) to send to you (“Tax Documents”) in connection with your Shares. A Tax Document provides important information you need to complete your tax returns. Tax Documents include Form 1099 and/or Schedule K-1. Occasionally, the Company is required to send you CORRECTED Tax Documents. Additionally, the Company may include inserts with your Tax Documents. The Company is required to send Tax Documents to you in writing, which means in paper form. When you consent to electronic delivery of your Tax Documents, you will be consenting to delivery of Tax Documents, including these corrected Tax Documents and inserts, electronically instead of in paper form. By executing this Agreement on the Platform, you are consenting in the affirmative that the Company may send Tax Documents to you electronically and acknowledging that you are able to access Tax Documents from the site. If you subsequently withdraw consent to receive Tax Documents electronically, a paper copy will be provided. Your consent to receive the Tax Documents electronically continues for every tax year until you withdraw your consent. You can withdraw your consent before the Tax Document is furnished by mailing a letter including your name, mailing address, effective tax year, and indicating your intent to withdraw consent to the electronic delivery of Tax Documents to: McGinley Orthopaedic Innovations, Inc., [ ], Attention: Investor Relations. If you withdraw consent to receive Tax Documents electronically, a paper copy will be provided. You Must Keep Your E-mail Address Current with the Company. You must promptly notify the Company of a change of your email address. If your mailing address, email address, telephone number or other contact information changes, you may also provide updated information by contacting the Company at the address provided in this Section 28, or via phone at [ ] or email at [ ].
29. Electronic Delivery of Information. Subscriber and the Company each hereby agrees that all current and future notices, confirmations and other communications regarding this Agreement and future communications in general between the parties, may be made by email, sent to the email address of record as set forth in this Agreement or as otherwise from time to time changed or updated and disclosed to the other party, without necessity of confirmation of receipt, delivery or reading, and such form of electronic communication is sufficient for all matters regarding the relationship between the parties. If any such electronically sent communication fails to be received for any reason, including but not limited to such communications being diverted to the recipients spam filters by the recipients email service provider, or due to a recipient’s change of address, or due to technology issues by the recipients service provider, the parties agree that the burden of such failure to receive is on the recipient and not the sender, and that the sender is under no obligation to resend communications via any other means, including but not limited to postal service or overnight courier, and that such communications shall for all purposes, including legal and regulatory, be deemed to have been delivered and received. No physical, paper documents will be sent to you, and if you desire physical documents then you agree to be satisfied by directly and
personally printing, at your own expense, the electronically sent communication(s) and maintaining such physical records in any manner or form that you desire.
[SIGNATURE APPEARS ONLINE]
ESCROW AGREEMENT
This ESCROW AGREEMENT (this “Agreement”) dated as of this 4th day of January 2022 by and among Company, a Delaware corporation (the “Company”), having an address at ______; Rialto Markets, LLC, having an address at 42 Broadway, Ste 12-129, NY, NY 10004 (“Placement Agent”), and WILMINGTON TRUST, NATIONAL ASSOCIATION (the “Escrow Agent”), with its principal corporate trust office at 99 Wood Avenue South, 10th Floor, Iselin, NJ 08830. The Company and the Placement Agent, each a “Party,” are collectively referred to as “Parties” and individually, a “Party.”
All capitalized terms not herein defined shall have the meaning ascribed to them in that certain Subscription Agreement, dated as of or about _______, 2022, as amended or supplemented from time-to-time, including all attachments, schedules and exhibits thereto (the “Subscription Agreement”).
W I T N E S S E T H:
WHEREAS, the Company proposes to sell (the “Financing Transaction”) a maximum of ____________shares of our common stock, par value __________(“Common Stock”), at an offering price of $____________ per share (the “Shares”) for an offering amount of $______; provided, a minimum of gross proceeds of $___________ raised, in a public offering (the “Offering”) to investors (each, an “Investor”); and
WHEREAS, subject to all conditions to closing being satisfied or waived, the closing(s) of the Offering shall take place from time to time until the earlier of (a) the date which is one year after this Offering being qualified by the U.S. Securities and Exchange Commission (the “SEC” or the “Commission”), or (b) the date on which this Offering is earlier terminated by the Company in its sole discretion (the “Termination Date”) (the earlier of (a) or (b), the “Final Termination Date”); and
WHEREAS, there is no minimum offering amount and all funds shall only be returned to the potential Investors in the event the Offering is not consummated or if the Company, in its sole discretion, rejects all or a part of a particular potential Investor’s subscription; and
WHEREAS, in connection with the Financing Transaction contemplated by the Subscription Agreement, the Company entered into a Placement Agent Agreement between the Company and the Placement Agent, and certain other agreements, documents, instruments and certificates necessary to carry out the purposes thereof, including without limitation the Subscription Agreement (collectively, the “Transaction Documents”); and
WHEREAS, the Company and Placement Agent desire to establish an escrow account with the Escrow Agent into which the Company and Placement Agent shall instruct the Investors to deposit checks or make a wire transfer for the payment of money made payable to the order of “WILMINGTON TRUST, N.A. as Escrow Agent for ____,” and the Escrow Agent is willing to accept said checks and other instruments for the payment of money in accordance with the terms hereinafter set forth; and
WHEREAS,
the Company and Placement Agent represent and warrant to the Escrow Agent that they have not stated to any individual or entity
that the Escrow Agent’s duties will include anything other than those duties stated in this Agreement; and
WHEREAS, THE ISSUER AND THE PLACEMENT AGENT UNDERSTAND THAT THE ESCROW AGENT, BY ACCEPTING THE APPOINMTMENT AND DESIGNATION AS ESCROW AGENT HEREUNDER, IN NO WAY ENDORSES THE MERITS OF THE OFFERING OF THE SECURITIES. THE ISSUER AND THE PLACEMENT AGENT AGREE TO NOTIFY ANY PERSON ACTING ON ITS BEHALF THAT THE ESCROW AGENT’S POSITION AS ESCROW AGENT DOES NOT CONSTITUTE SUCH AN ENDORSEMENT, AND TO PROHIBIT SAID PERSONS FROM THE USE OF THE ESCROW AGENT’S NAME AS AN ENDORSER OF SUCH OFFERING. The Issuer and the Placement Agent further agree to include with any sales literature, in which the Escrow Agent’s name appears and which is used in connection with such offering, a statement to the effect that the Escrow Agent in no way endorses the merits of the offering; and
WHEREAS, the Company and Placement Agent represent and warrant to the Escrow Agent that a copy of each document that has been delivered to the Investor and third parties that include Escrow Agent’s name and duties, has been attached hereto as Schedule I.
NOW, THEREFORE, IT IS AGREED as follows:
Article 1
ESCROW DEPOSIT
Section 1.1 Delivery of Escrow Funds.
(a) Placement Agent and the Company shall instruct the Investor to deliver to Escrow Agent checks made payable to the order of “WILMINGTON TRUST, N.A. as Escrow Agent for ______ Escrow”, or wire transfer to:
Wilmington Trust Company
ABA #: 031100092
A/C #: TBD
A/C Name:
Attn: Boris Treyger
International Wires:
M&T
Buffalo, New York
ABA: 022000046
SWIFT: MANTUS33
Beneficiary Bank: Wilmington Trust
Beneficiary ABA: 031100092
A/C #: TBD
A/C Name:
All such checks and wire transfers remitted to the Escrow Agent shall be accompanied by information identifying each Investor, subscription, the Investor’s social security or taxpayer identification number and address. In the event the Investor’s address and/or social security number or taxpayer identification number are not provided to Escrow Agent by the Investor, then Placement Agent and/or the Company agree to promptly upon request provide Escrow Agent with such information in writing. The checks or wire transfers shall be deposited into a non interest-bearing account at WILMINGTON TRUST, NATIONAL ASSOCIATION entitled “WILMINGTON TRUST, N.A. as Escrow Agent ______” (the “Escrow Account”).
Investors can also send funds using credit or debit card by utilizing the Invoice and Pay® service offered by M&T Bank by visiting the portal listed below:
https://www.sampleportal.mtb.com”
(b) The collected funds deposited into the Escrow Account are referred to as the “Escrow Funds.”
(c) The Escrow Agent shall have no duty or responsibility to enforce the collection or demand payment of any funds deposited into the Escrow Account. If, for any reason, any check deposited into the Escrow Account shall be returned unpaid to the Escrow Agent, the sole duty of the Escrow Agent shall be to return the check to the Investor and advise the Company and Placement Agent promptly thereof.
(d) All funds received by the Escrow Agent shall be held only in non-interest bearing bank accounts at WILMINGTON TRUST, NATIONAL ASSOCIATION.
(e) In the event that market conditions are such that negative interest applies to amounts deposited with the Escrow Agent, the Company and Placement Agent [jointly and severally] shall be responsible for the payment of such interest and the Escrow Agent shall be entitled to deduct from amounts on deposit with it an amount necessary to pay such negative interest. For the avoidance of doubt, the indemnification protections afforded to the Escrow Agent under Section 2.2 of this Agreement shall cover any interest-related expenses (including, but not limited to, negative interest) incurred by the Escrow Agent in the performance of its duties hereunder.
Section 1.2 Release of Escrow Funds. The Escrow Funds shall be paid by the Escrow Agent in accordance with the following:
(a) In the event that the Company advises the Escrow Agent in writing that the Offering has been terminated (the “Termination Notice”), the Escrow Agent shall promptly return the funds paid by each Investor to such Investor without interest or offset.
(b) At each Closing, the Company and the Placement Agent shall provide the Escrow Agent with written instructions regarding the disbursement of the Escrow Funds in accordance with Exhibit A attached hereto and made a part hereof and signed by the Company and the Placement Agent (the “Disbursement Instructions”).
(c) If by 5:00 P.M. Eastern time on the Final Termination Date, the Escrow Agent has not received written Disbursement Instructions from the Company and Placement Agent regarding the disbursement of the Escrow Funds in the Escrow Account, if any, then the Escrow Agent shall promptly return such Escrow Funds, if any, to the Investors without interest or offset. The Escrow Funds returned to the Investors shall be free and clear of any and all claims of the Escrow Agent.
(d) The Escrow Agent shall not be required to pay any uncollected funds or any funds that are not available for withdrawal.
(e) The Placement Agent or the Company will provide the Escrow Agent with the payment instructions for each Investor, to whom the funds should be returned in accordance with this section.
(f) In the event that Escrow Agent makes any payment to any other party pursuant to this Escrow Agreement and for any reason such payment (or any portion thereof) is required to be returned to the Escrow Account or another party or is subsequently invalidated, declared to be fraudulent or preferential, set aside and/or required to be repaid to a receiver, trustee or other party under any bankruptcy or insolvency law, other federal or state law, common law or equitable doctrine, then the recipient party shall repay to the Escrow Agent upon written request the amount so paid to it.
(g) The Escrow Agent shall, in its sole discretion, comply with judgments or orders issued or process entered by any court with respect to the Escrow Amount, including without limitation any attachment, levy or garnishment, without any obligation to determine such court's jurisdiction in the matter and in accordance with its normal business practices. If the Escrow Agent complies with any such judgment, order or process, then it shall not be liable to any of the Parties or any other person by reason of such compliance, regardless of the final disposition of any such judgment, order or process.
(h) Each Party understands and agrees that Escrow Agent shall have no obligation or duty to act upon a written direction delivered to Escrow Agent for the disbursement of all or part of the Escrow Amount under this Agreement (a “Written Direction”) if such Written Direction is not
(i) in writing,
(ii) signed by representatives of both Parties listed in Schedule II to this Agreement, in each case, each such individual an “Authorized Representative” of such Party), and
(iii) delivered to, and able to be authenticated by, Escrow Agent in accordance with Section 1.4 below.
(i) Upon request by any Party, the Escrow Agent set up each Party with on-line access to the account(s) established pursuant to this Agreement, which each Party can use to view and verify transaction on such account(s).
(j) A Party may specify in a Written Direction whether such Escrow Amount shall be disbursed by way of wire transfer or check. If the written notice for the disbursement of funds does not so specify the disbursement means, Escrow Agent may disburse the Escrow Amount by wire transfer.
Section 1.3 Written Direction and Other Instruction.
(a) With respect to any Written Direction or any other notice, direction or other instruction required to be delivered by a Party to Escrow Agent under this Agreement, Escrow Agent is authorized to follow and rely upon any and all such instructions given to it from time to time if the Escrow Agent believes, in good faith, that such instruction is genuine and to have been signed by an Authorized Representative of such Party. Escrow Agent shall have no duty or obligation to verify that the person who sent such instruction is, in fact, a person duly authorized to give instructions on behalf of a Party, other than to verify that the signature of the Authorized Representative on any such instruction appears to be the signature of such person. Each Party acknowledges and agrees that it is fully informed of the protections and risks associated with the various methods of transmitting instructions to Escrow Agent, and that there may be more secure methods of transmitting instructions other than the method selected by such Party. Escrow Agent shall have no responsibility or liability for any loss which may result from (i) any action taken or not taken by Escrow Agent in good faith reliance on any such signatures or instructions, (ii) as a result of a Party’s reliance upon or use of any particular method of delivering instructions to Escrow Agent, including the risk of interception of such instruction and misuse by third parties, or (iii) any officer or Authorized Representative of a Party named in Exhibit B delivered hereunder prior to actual receipt by Escrow Agent of a more current incumbency certificate or an updated Exhibit and a reasonable time for Escrow Agent to act upon such updated or more current certificate or Exhibit .
(b) Each Party may, at any time, update Schedule II by signing and submitting to Escrow Agent an update of such Schedule. Any updated Schedule shall not be effective unless Escrow Agent countersigns a copy thereof. Escrow Agent shall be entitled to a reasonable time to act to implement any changes on an updated Schedule II.
Section 1.4 Delivery and Authentication of Written Direction.
(a) A Written Direction must be delivered to Escrow Agent by one of the delivery methods set forth in Section 4.3.
(b) Each Party and Escrow Agent hereby agree that the following security procedures will be used to verify the authenticity of a Written Direction delivered by any Party to Escrow Agent under this Agreement:
(i) The Written Direction must include the name and signature of the person delivering the disbursement request to Escrow Agent. Escrow Agent will check that the name and signature of the person identified on the Written Direction appears to be the same as the name and signature of an Authorized Representative of such Party;
(ii) Escrow Agent will make a telephone call to an Authorized Representative of the Party purporting to deliver the Written Direction (which Authorized Representative may be the same as the Authorized Representative who delivered the Written Direction) at any telephone number for such Authorized Representative as set forth on Exhibit A to obtain oral confirmation of delivery of the Written Direction. If the Written Direction is a joint written notice of the Parties, the Escrow Agent shall call back an Authorized Representative of both of those Parties; and
(iii) If the Written Direction is sent by email to Escrow Agent, Escrow Agent also shall review such email address to verify that it appears to have been sent from an email address for an Authorized Representative of one of the Parties as set forth on Schedule II, as applicable, or from an email address for a person authorized under Schedule II to email a Written Direction to Escrow Agent on behalf of the Authorized Representative).
(c) Each Party acknowledges and agrees that given its particular circumstances, including the nature of its business, the size, type and frequency of its instructions, transactions and files, internal procedures and systems, the alternative security procedures offered by Escrow Agent and the security procedures in general use by other customers and banks similarly situated, the security procedures set forth in this Section 1.4 are a commercially reasonable method of verifying the authenticity of a payment order in a Written Direction.
(d) Escrow Agent is authorized to execute, and each Party expressly agrees to be bound by any payment order in a Written Direction issued in its name (and associated funds transfer) (i) that is accepted by Escrow Agent in accordance with the security procedures set forth in this Section 1.4 , whether or not authorized by such Party and/or (ii) that is authorized by or on behalf
of such Party or for which such Party is otherwise bound under the law of agency, whether or not the security procedures set forth in this Section 1.4 were followed, and to debit the Escrow Account for the amount of the payment order. Notwithstanding anything else, Escrow Agent shall be deemed to have acted in good faith and without negligence, gross negligence or misconduct if Escrow Agent is authorized to execute the payment order under this Section 1.4 . Any action taken by Escrow Agent pursuant to this paragraph prior to Escrow Agent’s actual receipt and acknowledgement of a notice of revocation, cancellation or amendment of a Written Direction shall not be affected by such notice.
(e) The security procedures set forth in this Section 1.4 are intended to verify the authenticity of payment orders provided to Escrow Agent and are not designed to, and do not, detect errors in the transmission or content of any payment order. Escrow Agent is not responsible for detecting an error in the payment order, regardless of whether any of the Parties believes the error was apparent, and Escrow Agent is not liable for any damages arising from any failure to detect an error.
(f) When instructed to credit or pay a party by both name and a unique numeric or alpha-numeric identifier (e.g. ABA number or account number), Escrow Agent, and any other banks participating in the funds transfer, may rely solely on the unique identifier, even if it identifies a party different than the party named. Each Party agrees to be bound by the rules of any funds transfer network used in connection with any payment order accepted by Escrow Agent hereunder.
(g) Escrow Agent shall not be obliged to make any payment requested under this Escrow Agreement if it is unable to validate the authenticity of the request by the security procedures set forth in this Section 1.4 . Escrow Agent’s inability to confirm a payment order may result in a delay or failure to act on that payment order. Notwithstanding anything else in this Agreement, Escrow Agent shall not be required to treat a payment order as having been received until Escrow Agent has authenticated it pursuant to the security procedures in this Section 2.3 and shall not be liable or responsible for any losses arising in relation to such delay or failure to act.
ARTICLE 2
PROVISIONS CONCERNING THE ESCROW AGENT
Section 2.1 Acceptance by Escrow Agent. The Escrow Agent hereby accepts and agrees to perform its obligations hereunder, provided that:
(a) The Escrow Agent shall be entitled to rely upon any order, judgment, opinion, or other writing delivered to it in compliance with the provisions of this Agreement without being required to determine the authenticity or the correctness of any fact stated therein or the propriety or validity of service thereof.
(b) The Escrow Agent shall be entitled to rely on and shall not be liable for any action taken or omitted to be taken by the Escrow Agent in accordance with the advice of counsel or other professionals retained or consulted by the Escrow Agent. The Escrow Agent shall be reimbursed
as set forth in Section 2.2 for any and all compensation (fees, expenses and other costs) paid and/or reimbursed to such counsel and/or professionals. The Escrow Agent may perform any and all of its duties through its agents, representatives, attorneys, custodians, and/or nominees and shall not be responsible for the acts or omissions of such agents, representatives, attorneys, custodians or nominees appointed with due care.
(c) In the event that the Escrow Agent shall be uncertain as to its duties or rights hereunder, the Escrow Agent shall be entitled to (i) refrain from taking any action other than to keep safely the Escrow Funds until it shall be directed otherwise by a court of competent jurisdiction, or (ii) deliver the Escrow Funds to a court of competent jurisdiction.
(d) The Escrow Agent shall have no duty, responsibility or obligation to interpret or enforce the terms of any agreement other than Escrow Agent’s obligations hereunder, and the Escrow Agent shall not be required to make a request that any monies be delivered to the Escrow Account The Escrow Agent makes no representation as to the validity, value, genuineness or collectability of any security or other document or instrument held by or delivered to it.
(e) The Escrow Agent shall be obligated to perform only such duties as are expressly set forth in this Agreement. No implied covenants or obligations shall be inferred from this Agreement against the Escrow Agent, nor shall the Escrow Agent be bound by the provisions of any agreement by the Company beyond the specific terms hereof. Without limiting the foregoing, the Escrow Agent shall dispose of the Escrow Funds in accordance with the express provisions of this Agreement, and has not reviewed and shall not make, be required to make or be liable in any manner for its failure to make, any determination under any other document, or any other agreement.
(f) No term or provision of this Agreement is intended to create, nor shall any such term or provision be deemed to have created, any trust, joint venture, partnership, between or among the Escrow Agent and any of the Parties.
Section 2.2. Indemnification. Placement Agent and the Company agree, jointly and severally, to indemnify and hold the Escrow Agent and its employees, officers, directors and agents (the “Indemnified Parties”) the “Indemnified Parties”) harmless from any and against all liabilities, losses, actions, suits or proceedings at law or in equity, and any other expenses, fees or charges of any character or nature, (including, without limitation, negative interest, attorney's fees and expenses and the costs of enforcement of this Escrow Agreement or any provision thereof), which an Indemnified Party may incur or with which it may be threatened by reason of acting as or on behalf of the Escrow Agent under this Escrow Agreement or arising out of the existence of the Escrow Account, except to the extent the same shall be have been finally adjudicated to have been directly caused by the Escrow Agent's gross negligence or willful misconduct. Placement Agent and the Company agree, jointly and severally, to pay or reimburse the Escrow Agent upon request for any transfer taxes or other taxes relating to the Escrow Funds incurred in connection herewith and shall indemnify and hold harmless the Escrow Agent with respect to any amounts that it is obligated to pay in the way of such taxes. The terms of this paragraph shall survive termination of this Agreement.
Section 2.3. Limitation of Liability. the escrow agent SHALL NOT be liable, directly or indirectly, for any (i) damages, Losses or expenses arising out of the services provided hereunder, other than damages, losses or expenses which have been finally adjudicated to have DIRECTLY resulted from the escrow agent’s gross negligence or willful misconduct, or (ii) special, Indirect, PUNITIVE or consequential damages or LOSSES OF ANY KIND WHATSOEVER (INCLUDING WITHOUT LIMITATION LOST PROFITS), even if the escrow agent has been advised of the possibility of such LOSSES OR damages AND REGARDLESS OF THE FORM OF ACTION, OR (III) AMOUNT IN EXCESS OF THE ESCROW FUNDS.
Section 2.4. Resignation and Termination of the Escrow Agent. The Escrow Agent may resign at any time by giving 30 days’ prior written notice of such resignation to Placement Agent and the Company. Upon providing such notice, the Escrow Agent shall have no further obligation hereunder except to hold as depositary the Escrow Funds that it receives until the end of such 30-day period. In such event, the Escrow Agent shall not take any action, other than receiving and depositing the Investor’s checks and wire transfers in accordance with this Agreement, until the Company has designated a banking corporation, trust company, attorney or other person as successor. Upon receipt of such written designation signed by Placement Agent and the Company, the Escrow Agent shall promptly deliver the Escrow Funds to such successor and shall thereafter have no further obligations hereunder. If the Company and Placement Agent have failed to appoint a successor escrow agent prior to the expiration of thirty (30) days following the delivery of such notice of resignation or removal, the Escrow Agent shall be entitled, at its sole discretion and at the expense of the Company and/or Placement Agent, to (a) return the Escrow Funds to the Company, or (b) petition any court of competent jurisdiction for the appointment of a successor escrow agent or for other appropriate relief, and any such resulting appointment shall be binding upon the parties. In either case provided for in this paragraph, the Escrow Agent shall be relieved of all further obligations and released from all liability thereafter arising with respect to the Escrow Funds.
Section 2.5 Termination. The Company and Placement Agent may terminate the appointment of the Escrow Agent hereunder upon written notice specifying the date upon which such termination shall take effect, which date shall be at least 30 days from the date of such notice. In the event of such termination, the Company and Placement Agent shall, within 30 days of such notice, appoint a successor escrow agent and the Escrow Agent shall, upon receipt of written instructions signed by the Company and Placement Agent, turn over to such successor escrow agent all of the Escrow Funds Upon receipt of the Escrow Funds, the successor escrow agent shall become the escrow agent hereunder and shall be bound by all of the provisions hereof and the Escrow Agent shall be relieved of all further obligations and released from all liability thereafter arising with respect to the Escrow Funds and under this Agreement. If the Company has failed to appoint a successor escrow agent prior to the expiration of thirty (30) days following the delivery of the notice of termination, the Escrow Agent shall be entitled, at its sole discretion and at the expense of the Company, to (a) return the Escrow Funds to the Company, or (b) petition any court
of competent jurisdiction for the appointment of a successor escrow agent or for other appropriate relief, and any such resulting appointment shall be binding upon the parties.
Section 2.6 Compensation. Escrow Agent shall be entitled, for the duties to be performed by it hereunder, to compensation as stated in the schedule attached hereto as Schedule III, which fee shall be paid by the Company upon the signing of this Agreement. In addition, the Company shall be obligated to reimburse Escrow Agent for all fees, costs and expenses incurred or that become due in connection with this Agreement or the Escrow Account, including attorney’s fees. Neither the modification, cancellation, termination, resignation or rescission of this Agreement nor the resignation or termination of the Escrow Agent shall affect the right of Escrow Agent to retain the amount of any fee which has been paid, or to be reimbursed or paid any amount which has been incurred or becomes due, prior to the effective date of any such modification, cancellation, termination, resignation or rescission. To the extent the Escrow Agent has incurred any such expenses, or any such fee becomes due, prior to any closing, the Escrow Agent shall advise the Company and the Company shall direct all such amounts to be paid directly at any such closing. As security for the due and punctual performance of any and all of the Company’s obligations to the Escrow Agent hereunder, now or hereafter arising, the Company, hereby pledges, assigns and grants to the Escrow Agent a continuing security interest in, and a lien on and right of setoff against, the Escrow Funds and all distributions thereon, investments thereof or additions thereto. If any fees, expenses or costs incurred by, or any obligations owed to, the Escrow Agent hereunder are not promptly paid when due, the Escrow Agent may reimburse itself therefor from the Escrow Funds, and may sell, convey or otherwise dispose of any Escrow Funds for such purpose. The security interest and setoff rights of the Escrow Agent shall at all times be valid, perfected and enforceable by the Escrow Agent against the Parties and all third parties in accordance with the terms of this Escrow Agreement. The terms of this paragraph shall survive termination of this Agreement.
Section 2.7. Merger or Consolidation. Any corporation or association into which the Escrow Agent may be converted or merged, or with which it may be consolidated, or to which it may sell or transfer all or substantially all of its corporate trust business and assets as a whole or substantially as a whole, or any corporation or association resulting from any such conversion, sale, merger, consolidation or transfer to which the Escrow Agent is a party, shall be and become the successor escrow agent under this Agreement and shall have and succeed to the rights, powers, duties, immunities and privileges as its predecessor, without the execution or filing of any instrument or paper or the performance of any further act.
Section 2.8. Attachment of Escrow Funds; Compliance with Legal Orders. In the event that any Escrow Amount shall be attached, garnished or levied upon by any court order, or the delivery thereof shall be stayed or enjoined by an order of a court, or any order, judgment or decree shall be made or entered by any court order affecting the Escrow Funds , the Escrow Agent is hereby expressly authorized, in its sole discretion, to respond as it deems appropriate or to comply with all writs, orders or decrees so entered or issued, or which it is advised by legal counsel of its own choosing is binding upon it, whether with or without jurisdiction. In the event that the Escrow Agent obeys or complies with any such writ, order or decree it shall not be liable to any Party or
to any other person, firm or corporation, should, by reason of such compliance notwithstanding, such writ, order or decree be subsequently reversed, modified, annulled, set aside or vacated.
Section 2.9 Force Majeure. The Escrow Agent shall not be responsible or liable for any failure or delay in the performance of its obligation under this Agreement arising out of or caused, directly or indirectly, by circumstances beyond its reasonable control, including, without limitation, acts of God; earthquakes; fire; flood; wars; acts of terrorism; civil or military disturbances; sabotage; epidemic; pandemics; riots; interruptions, loss or malfunctions of utilities, computer (hardware or software) or communications services; accidents; labor disputes; acts of civil or military authority or governmental action; hacking, cyber-attacks or other unauthorized infiltration of Escrow Agent’s information technology infrastructure it being understood that the Escrow Agent shall use commercially reasonable efforts which are consistent with accepted practices in the banking industry to resume performance as soon as reasonably practicable under the circumstances.
Section 2.10 No Financial Obligation. Escrow Agent shall not be required to use its own funds in the performance of any of its obligations or duties or the exercise of any of its rights or powers, and shall not be required to take any action which, in Escrow Agent's sole and absolute judgment, could involve it in expense or liability unless furnished with security and indemnity which it deems, in its sole and absolute discretion, to be satisfactory.
ARTICLE 3
MISCELLANEOUS
Section 3.1. Successors and Assigns. This Agreement shall be binding on and inure to the benefit of each Party and the Escrow Agent and their respective successors and permitted assigns. No other persons shall have any rights under this Agreement. No assignment of the interest of any of the Parties shall be binding unless and until written notice of such assignment shall be delivered to the other Parties and Escrow Agent and shall require the prior written consent of the other Parties and Escrow Agent (such consent not to be unreasonably withheld).
Section 3.2. Escheat. Each Party is aware that under applicable state law, property which is presumed abandoned may under certain circumstances escheat to the applicable state. The Escrow Agent shall have no liability to any of the Parties, their respective heirs, legal representatives, successors and assigns, or any other party, should any or all of the Escrow Funds escheat by operation of law.
Section 3.3. Notices. All notices, requests, demands, and other communications required under this Escrow Agreement shall be in writing, in English, and shall be deemed to have been duly given if delivered (i) personally, (ii) by facsimile transmission with written confirmation of receipt, (iii) by overnight delivery with a reputable national overnight delivery service, (iv) by mail or by certified mail, return receipt requested, and postage prepaid, or (v) by electronic transmission; including by way of e-mail (as long as such email is accompanied by a PDF or similar version of
the relevant document bearing the signature of an Authorized Representative for the Party sending the notice) with email confirmation of receipt. If any notice is mailed, it shall be deemed given five business days after the date such notice is deposited in the United States mail. If notice is given to a party, it shall be given at the address for such party set forth below. It shall be the responsibility of the Company to notify the Escrow Agent in writing of any name or address changes. In the case of communications delivered to the Escrow Agent, such communications shall be deemed to have been given on the date received by the Escrow Agent. :
If to Placement Agent:
Rialto Markets, LLC
Name Ryan Simmons
Title Head of Operations
Company Rialto Markets LLC
Address 42 Broadway, Ste 12-129, NY, NY 1004
Telephone # (917) 279-7453
Email Address ryan@rialtomarkets.com
If to the Company:
Name
Title
Company
Address
Telephone #
Email Address
Copy:
Name
Title
Company
Address
Telephone #
Email Address
If to Escrow Agent:
WILMINGTON TRUST, NATIONAL ASSOCIATION
99 Wood Avenue South, 10th Floor
Iselin, NJ 08830
Attention: Boris Treyger
Phone: (212) 941-4416
Email: btreyger@wilmingtontrust.com
Section 3.4. Governing Law and Jurisdiction. This Agreement shall be governed by and construed in accordance with the laws of the State of Delaware. Each Party and Escrow Agent hereby consents to the exclusive personal jurisdiction of the courts located in the State of Delaware in the event of a dispute arising out of or under this Agreement. Each Party and Escrow Agent hereby irrevocably waives any objection to the laying of the venue of any suit, action or proceeding and irrevocably submits to the exclusive jurisdiction of such court in such suit, action or proceeding.
Section 3.5. Entire Agreement. This Agreement and the Exhibits attached hereto (as updated from time to time in accordance herewith) set forth the entire agreement and understanding of the parties related to the Escrow Amount. If a court of competent jurisdiction declares a provision invalid, it will be ineffective only to the extent of the invalidity, so that the remainder of the provision and Escrow Agreement will continue in full force and effect.
Section 3.6. Amendment. This Agreement may be amended, modified, superseded, rescinded, or canceled only by a written instrument executed by each of the Parties and the Escrow Agent.
Section 3.7. Waivers. The failure of any party to this Agreement at any time or times to require performance of any provision under this Agreement shall in no manner affect the right at a later time to enforce the same performance. A waiver by any party to this Agreement of any such condition or breach of any term, covenant, representation, or warranty contained in this Agreement, in any one or more instances, shall neither be construed as a further or continuing waiver of any such condition or breach nor a waiver of any other condition or breach of any other term, covenant, representation, or warranty contained in this Agreement.
Section 3.8. Headings. Section headings of this Agreement have been inserted for convenience of reference only and shall in no way restrict or otherwise modify any of the terms or provisions of this Escrow Agreement.
Section 3.9. Electronic Signatures; Facsimile Signatures; Counterparts. This Escrow Agreement may be executed in one or more counterparts. Such execution of counterparts may occur by manual signature, electronic signature, facsimile signature, manual signature transmitted by means of facsimile transmission or manual signature contained in an imaged document attached to an email transmission, and any such execution that is not by manual signature shall have the same legal effect, validity and enforceability as a manual signature. Each such counterpart executed in accordance with the foregoing shall be deemed an original, with all such counterparts
together constituting one and the same instrument. The exchange of executed copies of this Escrow Agreement or of executed signature pages to this Escrow Agreement by electronic transmission, facsimile transmission or as an imaged document attached to an email transmission shall constitute effective execution and delivery hereof. Any copy of this Escrow Agreement which is fully executed and transmitted in accordance with the terms hereof may be used for all purposes in lieu of a manually executed copy of this Escrow Agreement and shall have the same legal effect, validity and enforceability as if executed by manual signature.
Section 3.10. Waiver of Jury Trial. EACH OF THE PARTIES HERETO AND THE ESCROW AGENT EXPRESSLY WAIVES THE RIGHT TO TRIAL BY JURY IN RESOLVING ANY CLAIM OR COUNTERCLAIM RELATING TO OR ARISING OUT OF THIS AGREEMENT.
Section 3.11 Termination. This Agreement will terminate upon the Final Termination Date.
Section 3.12 Anti-Terrorism/Anti-Money Laundering Laws.
IMPORTANT INFORMATION ABOUT PROCEDURES FOR OPENING A NEW ACCOUNT - To help the United States government fight the funding of terrorism or money laundering activities, Federal law requires all financial institutions to obtain, verify, and record information that identifies each person who opens a new account. What this means for the parties to this Agreement: the Escrow Agent will ask for your name, address, date of birth, and other information that will allow the Escrow Agent to identify you (e.g., your social security number or tax identification number.) The Escrow Agent may also ask to see your driver’s license or other identifying documents (e.g., passport, evidence of formation of corporation, limited liability company, limited partnership, etc., certificate of good standing.)
[The balance of this page intentionally left blank – signature page follows]
IN WITNESS WHEREOF, the parties have duly executed this Agreement as of the date first set forth above.
(Company) Rialto Markets Inc. (Placement Agent)
By: ______________________________ By: _______________________
Name: Name: Ryan Simmons
Title: Title: Head of Operations
WILMINGTON TRUST, NATIONAL ASSOCIATION
as Escrow Agent
By: ______________________________
Name:
Title: Vice President
Schedule I
Form 1 a
EXHIBIT B
Certificate as to Authorized Signatures
of COMPANY / rialto markets llc
Company/Rialto Markets LLC hereby designates each of the following persons as its Authorized Representative for purposes of this Agreement, and confirms that the title, contact information and specimen signature of each such person as set forth below is true and correct. Each such Authorized Representative is authorized to initiate and approve transactions of all types for the Escrow Account[s] established under the Agreement to which this Exhibit A is attached, on behalf of [Company]/[Placement Agent].
| Name (print): | ||
| Specimen Signature: | ||
| Title: | ||
Telephone Number (required): If more than one, list all applicable telephone numbers. | Office: Cell: | |
E-mail (required): If more than one, list all applicable email addresses. | Email 1: Email 2: |
| Name (print): | ||
| Specimen Signature: | ||
| Title: | ||
Telephone Number (required): If more than one, list all applicable telephone numbers. | Office: Cell: | |
E-mail (required): If more than one, list all applicable email addresses. | Email 1: Email 2: |
| Name (print): | ||
| Specimen Signature: | ||
| Title: | ||
Telephone Number (required): If more than one, list all applicable telephone numbers. | Office: Cell: | |
E-mail (required): If more than one, list all applicable email addresses. | Email 1: Email 2: |
Additional Email Addresses:
The following additional email addresses also may be used by Escrow Agent to verify the email address used to send any Payment Notice to Escrow Agent:
Email 1:
Email 2:
Email 3:
COMPLETE BELOW TO UPDATE EXHIBIT A
If Company wishes to update this Exhibit A, Company must complete, sign and send to Escrow Agent an updated copy of this Exhibit A with such changes. Any updated Exhibit A shall be effective once signed by Company and Escrow Agent and shall entirely supersede and replace any prior Exhibit A to this Agreement.
[____________________________________]
By:_________________________
Name:
Title:
Date:
WILMINGTON TRUST, NATIONAL ASSOCIATION (as Escrow Agent)
By:_________________________
Name:
Title:
Date:
Schedule III
Fees of Escrow Agent
| Acceptance Fee: | Waived |
Initial Fees as they relate to Wilmington Trust acting in the capacity of Escrow Agent – includes review of the Escrow Agreement; acceptance of the Escrow appointment; setting up of Escrow Account(s) and accounting records; and coordination of receipt of Escrow Information for deposit to the Escrow Account(s). Acceptance Fee payable at time of Escrow Agreement execution.
| Escrow Agent Administration Fee: | $8,000 |
For ordinary administrative services by Escrow Agent – includes daily routine account management; monitoring claim notices pursuant to the agreement; and disbursement of Escrow Information in accordance with the agreement.
Wilmington Trust’s bid is based on the following assumptions:
· Number of Escrow Accounts to be established: 1
· Est. Term: Under 12 months
· Escrow funds remain un-invested
Out-of-Pocket Expenses: Billed At Cost
1
Exhibit A
FORM OF ESCROW DISBURSEMENT INSTRUCTIONS
AND RELEASE NOTICE
Date:
WILMINGTON TRUST, NATIONAL ASSOCIATION
99 Wood Avenue South, 10th Floor
Iselin, NJ 08830
Attention: Boris Treyger
Dear Mr./Ms _______:
In accordance with the terms of Section 1.2 of the Escrow Agreement dated as of _______, 2021 (the "Escrow Agreement"), by and between ______ (the “Company”), ______ (“Placement Agent”) and WILMINGTON TRUST, NATIONAL ASSOCIATION (the "Escrow Agent"), the Company and Placement Agent hereby direct the Escrow Agent to distribute all of the Escrow Funds (as defined in the Escrow Agreement) in accordance with the following wire instructions:
________________________: $
________________________: $
________________________: $
Very truly yours,
Company
By:__________________
Name: _________________
Title: ________________
Placement Agent
By:__________________
Name: _______________
Title: ________________
CONSENT OF INDEPENDENT AUDITOR
We consent to the use, in this Offering Circular on Form 1-A, of our independent auditor’s report dated July 13, 2022, with respect to the audited consolidated balance sheets of McGinley Orthopaedic Innovations, LLC as of December 31, 2021 and 2020, and the related consolidated statements of operations, changes in members’ equity, cash flows and related notes to the consolidated financial statements for the years then ended.
Very truly yours,
Assuarance Dimensions
/s/ Assurance Dimensions
Tampa, Florida
August 11, 2022
TRAE O’NEIL HIGH LEGAL SERVICE PC
Licensed in Texas Only
(347) 663-8063
thigh@tohlspc.com
CORPORATE ● SECURITIES ● MERGERS & ACQUISITIONS ● CONTRACTS
August 8, 2022
McGinley Orthopaedic Innovations, Inc.
Joseph C. McGinley, CEO
234 E 1st St Suite 242
Casper, WY 82601
Re: Regulation A Offering
Dear Mr. McGinley:
In connection with the Regulation A offering dated on or about August 8, 2022 (the “Offering”) of the issuance and sale by McGinley Orthopaedic Innovations, Inc. (the “Company”) of shares of common stock of the Company (the “Shares”), and up to 1,904,761.90 shares of common stock of the Company offered for resale by the selling shareholders (the “Resale Shares”), please accept this letter as my opinion as to the legality of the Shares and Resale Shares covered by the Offering Circular associated therewith.
We have examined such documents and such matters of fact and law that we have deemed necessary for the purpose of rendering the opinion set forth herein. As to questions of fact material to this opinion, we have relied on certificates or comparable documents of public officials and of officers and representatives of the Company. In rendering the opinion expressed below, we have assumed without verification the genuineness of all signatures, the legal capacity of natural persons, the authenticity of all documents submitted to us as originals, the conformity to the originals of all documents submitted to us as copies and the authenticity of the originals of such copies.
Based on the foregoing, and subject to the qualifications, assumptions and limitations stated herein, we are of the opinion that the Shares have been duly authorized and, when sold, the Shares will be validly issued, fully paid and non-assessable under the laws of the State of Wyoming,** in which the Company is incorporated. Furthermore, we are of the opinion that the Resale Shares being offered by the selling shareholders have been validly issued, fully paid and non-assessable.
Our opinion that any document is legal, valid and binding is qualified as to:
(a) limitations imposed by bankruptcy, insolvency, reorganization, arrangement, fraudulent conveyance, moratorium or other laws relating to or affecting the rights of creditors generally;
(b) rights to indemnification and contribution, which may be limited by applicable law or equitable principles; and
(c) general principles of equity, including without limitation concepts of materiality, reasonableness, good faith and fair dealing, and the possible unavailability of specific performance or injunctive relief and limitation of rights of acceleration, regardless of whether such enforceability is considered in a proceeding in equity or at law.
This opinion letter is given as of the date hereof, and we express no opinion as to the effect of subsequent events or changes in law occurring or becoming effective after the date hereof. We assume no obligation to update this opinion letter or otherwise advise you with respect to any facts or circumstances or changes in law that may hereafter occur or come to our attention (even though the change may affect the legal conclusions stated in this opinion letter).
Very truly yours,
Trae O'Neil High Legal Services PC
Trae O'Neil High, Attorney
For the Firm
**Not licensed in the State of Wyoming