
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) |
(Commission File Number) | (IRS Employer Identification No.) |
|
|
||
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number including area
code: (
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Trading Symbol | Name of each exchange on which registered | ||
| The |
Indicate by check mark whether the registrant is an emerging growth company as defined in as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b–2 of the Securities Exchange Act of 1934 (§ 240.12b–2 of this chapter).
Emerging growth
company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 1.01 Entry into Material Definitive Agreements
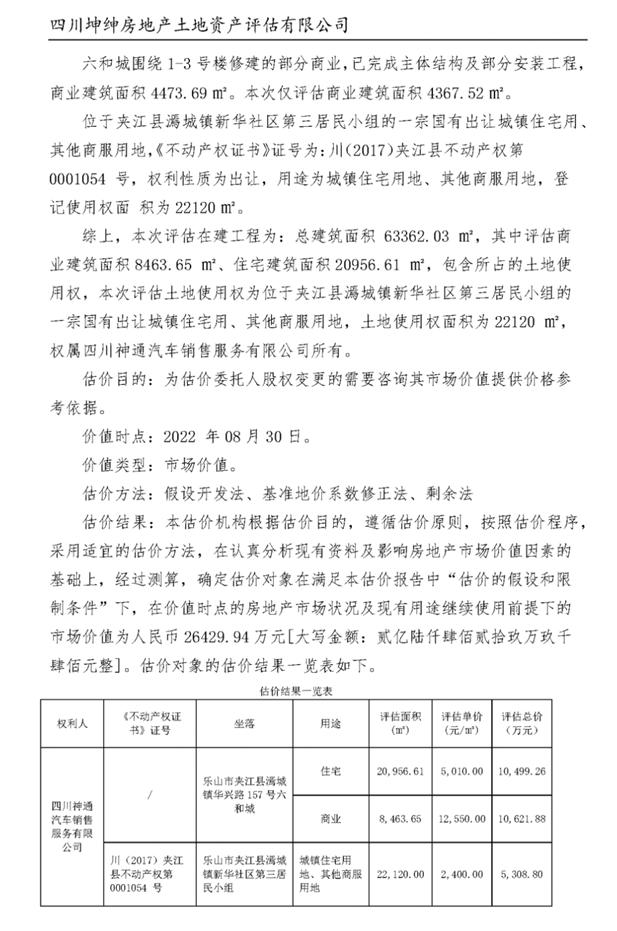
On August 14, 2023, ABVC BioPharma, Inc., a Nevada corporation, (the “Company”) entered into a cooperation agreement (the “Agreement”, the transaction contemplated therein the “Transaction”) with Zhonghui United Technology (Chengdu) Group Co., Ltd. (“中汇联和科技(成都)集团有限公司”), a Company established under the Law of People’s Republic of China (“Zhonghui”). Pursuant to the Agreement, the Company agreed to acquire 20% of the ownership of a property and the parcel of the land (the “Property”) owned by Zhonghui in Leshan, Sichuan, China. The valuation of the Property as of April 18, 2023, which was assessed by an independent third party, is estimated to be approximately CNY 264,299,400 or approximately US$37,000,000. In exchange, the Company agreed to issue to Zhonghui, an aggregate of 370,000 shares (the “Shares”) of common stock of the Company, par value $0.001 per share, at a per share price of $20.0. The Company and Zhonghui plan to jointly develop the Property into a healthcare center for senior living, long-term care, and medical care in the areas of ABVCs' special interests, such as Ophthalmology, Oncology, and Central Nervous Systems, which can be established as a base for the China market and global development.
Zhonghui agreed that the Shares will be subject to a lock-up period of one year following the closing date of this Transaction. In addition, parties agreed that, after one year following the closing of the Transaction, the market value of the Shares or the value of the Property increase or decrease, the parties will negotiate in good faith to make reasonable adjustment. For example, if the market value of the Shares is less than the value of the Property, the Company will issue more shares to Zhonghui, provided that, the Zhonghui’s ownership of the Company’s shares shall not exceed 19.99% of the then issued and outstanding shares; in return, if the value of the Property becomes less than the market value of the Shares, Zhonghui shall transfer additional ownership of the Property to the Company.
On August 17, 2023, the Company issued a press release announcing the entry into the Agreement. A copy of the press release is attached hereto as Exhibit 99.1 and is incorporated herein by reference.
The foregoing summary of the terms of the Agreement is subject to, and qualified in its entirety by, the complete Term Sheet, which is attached as an exhibit to this filing and incorporated herein by reference.
Item 9.01 Financial Statement and Exhibits
(d) Exhibits
| Exhibit No. | Description | |
| 10.1 | The Collaboration Agreement dated August 14, 2023 | |
| 99.1 | Press Release dated August 17, 2023 | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
1
SIGNATURE
Pursuant to the requirements of the Securities and Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| ABVC BioPharma, Inc. | ||
| August 17, 2023 | By: | /s/ Uttam Patil |
| Uttam Patil | ||
| Chief Executive Officer | ||
2
Exhibit 10.1
COOPERATION AGREEMENT
This COOPERATION AGREEMENT (“Agreement”, the transaction contemplated herein the “Transaction”) is entered into as of August 14, 2023, by and between ABVC BioPharma, Inc. (“ABVC”), and Zhonghui United Technology (Chengdu) Group Co., Ltd. (“中汇联和科技(成都)集团有限公司”) and its affiliated enterprises (hereinafter referred to as “ZHONGHUI”).
ABVC and ZHONGHUI are sometimes referred to herein individually as a “Party,” and collectively as the “Parties.”
Due to the mutual interests and benefits of both parties to strategically cooperate and construct an integrated platform for global development of healthcare business and for the medical, pharmaceutical, biotechnology and healthcare collaboration in China market, ABVC will acquire real estate through equity transfer for the development of a large-scale health and wellness base.
Article 1: Subject Matter and Consideration
1.1 Subject Matter: The subject matter of this Agreement includes:
a) The common shares of ABVC (stock symbol: ABVC) on the Nasdaq Capital Market, with a price of twenty US dollars per share.
b) ZHONGHUI’s ownership of a property located at No. 157 Huaxing Road, Liuhe City, Jiajiang County (under construction) and a parcel of state-owned land in Xinhua Community, Third Residential Group, Liuhe City, Jiajiang County, intended for town residential and other commercial service purposes (hereinafter referred to as The Property).
As of April 18, 2023, the valuation of the Property is CNY 264,299,400 or approximately US$37,000,000 (see Exhibit A).
1.2 Consideration: The consideration for ZHONGHUI shall be as follows:
a) ABVC shall transfer 370,000 shares of common stock of ABVC (the “Shares”) to ZHONGHUI at a price of twenty dollars per share.
b) ZHONGHUI shall transfer 20% ownership of the Property to ABVC.
1.3 Transfer of Ownership: Upon completion of the transaction, ownership of the shares shall be transferred from ABVC to ZHONGHUI, and ownership of the Property shall be transferred from ZHONGHUI to ABVC.
Article 2: Exchange of The Property through ABVC Equity Transfer
2.1 Agreement to Exchange Property: ABVC and ZHONGHUI agree to exchange a 20% ownership stake in ZHONGHUI ‘s property through an equity transfer of ABVC shares.
2.2 Consideration The consideration shall be US$7,400,000, equivalent to the Shares at twenty US dollars per share. ZHONGHUI agrees the Shares shall be restricted shares and the issuance of such is exempt from registration in reliance on Regulation S promulgated under the 1933 Securities Act, as amended. In addition, Zhonghui agrees that the Share Consideration will be locked up for one year (the “Lock-up Period”) and during the Lock-up Period the proxy voting right of all the Shares will be delegated to the Chairman of ABVC.
2.3 Transfer Process: The transfer of Shares from ABVC to ZHONGHUI shall be executed in accordance with the applicable laws, regulations, and procedures governing equity transfers.
2.4 Transfer of Ownership: Upon completion of the share transfer, a 20% ownership in the Property shall be transferred from ZHONGHUI to ABVC.
2.5 Representation regarding the Valuation: Both parties acknowledge and agree that the value of the Transaction is based on the estimated value of the Property and the market value of ABVC shares at the time of the equity transfer.
2.6 Valuation Dispute: In the event of any dispute regarding the valuation or other matters related to the Transaction, the parties shall make good faith efforts to resolve the dispute through negotiation and mediation.
2.7 Taxes and Fees: Any taxes, fees, or other charges arising from the equity transfer and exchange shall be the responsibility of the respective parties as determined by applicable laws and regulations.
Article 3: Capital Protection Agreement
3.1 Agreement on Value Maintenance: One year after the execution of this Agreement, both parties will make efforts to ensure that the value of The Property and the value of the Shares shall be maintained at the same or higher value as at the closing day of this Agreement.
3.2 Adjustment Mechanism: In the event that either the value of the Property or the Shares changes on the day of the one-year anniversary of the closing date of this Agreement, both parties agree to engage in reasonable adjustments to address the disparity.
3.3 Adjustment Process: The process for making adjustments shall be determined through mutual agreement and in accordance with the applicable laws and regulations. The parties shall engage in good faith negotiations to reach a mutually acceptable solution. In case, the value of the Share Consideration is below US$7,400,000, more stock shall be issued. In case the value of The Property is below US$7,400,000, a greater ownership interest in the property shall be transfer to ABVC; provided, however, that ZHONGHUI’s ownership shall be capped at 19.99% of then issued and outstanding shares of ABVC common stock.
2
3.4 Valuation Assessment: In the event of a dispute regarding the valuation or value maintenance, the parties may engage an independent third-party expert or appraiser to assess the value and provide recommendations for adjustment, the assessment fees will be shared equally between both parties.
3.5 Amendments to the Agreement: Any amendments or modifications to this Agreement necessitated by the value adjustment shall be made in writing and signed by both parties.
3.6 Force Majeure: The provisions of this Agreement shall not apply in cases of force majeure events that are beyond the reasonable control of either party and that significantly impact the value of The Property or stocks.
3.7 Governing Law and Jurisdiction: This Agreement shall be governed by and construed in accordance with the laws of the jurisdiction in the State of New York. Any disputes arising from or in connection with this Agreement shall be subject to the exclusive jurisdiction of the courts in that jurisdiction.
Article 4: Mutual Goals
4.1 Development of a Large Health and Wellness Base: Both ABVC and ZHONGHUI agree to collaborate on the development of a large-scale health and wellness base. The goal is to establish a comprehensive facility that offers a wide range of health and wellness services to promote a healthy lifestyle and well-being. Specifically, a healthcare center for senior living, long term care and medical care in the areas of ABVC’s special interests, such as Ophthalmology, Oncology and Central Nervous Systems, will be established as the base for China market as well as global development.
4.2 Construction of an Integrated Platform for Industry-Academia-Research Collaboration and Rural Revitalization: Both parties aim to build an integrated platform that facilitates collaboration between industry, academia, and research. This platform seeks to contribute to rural revitalization by promoting innovation, knowledge exchange, and sustainable
development in rural areas.
Article 5: Termination, Replacement and Amendment
5.1 Both parties aim to complete the transfer of the Property by September 30th, 2023. If unable to do so, both parties shall negotiate and determine a subsequent transfer date.
3
5.2 If there are ownership restrictions, such as seizure or mortgage, affecting The Property held by ZHONGHUI, preventing the transfer or transaction, ABVC agrees that ZHONGHUI can replace such assets with other project assets that are legally valid, feasible for execution, tradable, and meet the requirements of ABVC. The specific details of asset replacement shall be determined through further negotiation and documented in a supplementary agreement or attachment.
5.3 The Property for the final transaction can be adjusted based on the legal due diligence of this transaction, considering The Property and equity situations of both parties, as well as the transaction structure. Any adjustments will be documented through a supplementary agreement or attachment.
5.4 In case the due diligence results are inconsistent with the disclosures or records in this agreement and its attachments, or if discrepancies are found with ABVC’s statements or warranties, or if undisclosed debts, costs, expenses, expenditures, or other matters are discovered, or if there are legal/political defects in The Property that hinder the transfer of rights, ABVC reserves the right to terminate this cooperation.
5.5 If, based on reasonable analysis and judgment through due diligence, ABVC determines that the transaction’s objectives are difficult to achieve or pose significant obstacles to the transaction, ABVC reserves the right to terminate this cooperation.
5.6 Composition: Supplementary agreements or appended documents related to assets and equity transfer subsequently signed by both parties shall constitute valid integral parts of this Agreement. The rights and obligations of both parties shall be subject to the terms and conditions stipulated in this agreement and in any supplementary agreements or appended documents subsequently signed.
5.7 Amendment: The terms and conditions in this Agreement can be further amended in writing upon the mutual agreement of both parties.
5.8 Termination: This Agreement shall become invalid or terminated and shall no longer have any effect. However, the provisions of Sections 3.7, Sections 5, Sections 6, and Sections 7 shall remain in effect after the termination or invalidation of this Agreement.
4
Article 6: Confidentiality
6.1 Duty of Care: Both parties agree to exercise due care in maintaining the confidentiality of any relevant data or information. This duty of confidentiality shall not be affected by the termination or expiration of this Agreement.
6.2 Non-Disclosure Obligation: Both parties shall keep all confidential information received from the other party confidential and shall not disclose it to any third party without prior written consent, unless required by law or authorized by the disclosing party.
6.3 Return or Destruction of Confidential Information: Upon the expiration, termination, or at the request of either party, the receiving party shall promptly return to the disclosing party all confidential information received, along with any copies or reproductions thereof. Alternatively, the receiving party shall follow the instructions of the disclosing party regarding the destruction of such confidential information.
6.4 Survival of Confidentiality Obligations: The obligations of confidentiality and non-disclosure shall survive the expiration or termination of this Agreement and shall continue to be binding on both parties.
6.5 Exceptions: The obligations of confidentiality shall not apply to information that: (a) was already known to the receiving party prior to its disclosure by the disclosing party; (b) is or becomes publicly available without breach of this Agreement; (c) is received from a third party without breach of any confidentiality obligation; or (d) is independently developed by the receiving party without reference to the disclosing party’s confidential information.
Article 7: Copies of the Agreement
7.1 Two original copies of this Agreement have been prepared, with each party holding one original copy.
7.2 After signing, each party shall retain the original copy held by them as valid evidence between the parties.
7.3 Copies, reproductions, or electronic files of this Agreement shall have the same legal effect and may be used for communication between the parties.
Article 8: Language.
The Agreement is in both English and Chinese, which both have binding effects. If there is any conflict between the English and Chinese language, English language prevails.
5
Signature Page
Party A: ABVC BioPharma, Inc
Name: Uttam Yashwant Patil, Ph.D. Title: CEO
Address: 44370 Old Warm Springs Blvd.– Fremont, California 94538 USA
Party B: 中 汇 联 和 科 技 ( 成 都 ) 集 團 有 限 公 司
Name:孙 文 成
Title: CHAIRMAN
Addess:中国(四川)成都青羊区順城大街249号西星大厦7楼
6
Exhibit A

7

8
9

10
Exhibit 99.1

ABVC Executes Cooperation Agreement for Strategic Investments
Fremont, CA, August 17, 2023 – ABVC BioPharma, Inc. (NASDAQ: ABVC) ("Company"), a clinical-stage biopharmaceutical company developing therapeutic solutions in Oncology/Hematology, Neurology, and Ophthalmology, announced today that it had entered into a Cooperation Agreement to exchange a 20% ownership stake in real estate property of Zhonghui United Technology (Chengdu) Group Co., Ltd. (“中汇联和科技(成都)集团有限公司”) and its affiliated enterprises (“Zhonghui”).
Under the terms of the Cooperation Agreement, the estimated value of the transaction is approximately $7.4 million, equivalent to 370,000 shares of ABVC common stock at $20 per share.
"We are extremely proud to have executed the Cooperation Agreement with Zhonghui; this transaction builds on ABVC's commitment to grow its biopharma industry and to provide best-in-class innovations in research, development, and manufacturing of pharmaceuticals, biotechnology-based food and medicines, medical devices, biomedical technologies, and nutraceuticals," said Dr. Uttam Patil, Chief Executive Officer of ABVC. "As per the current Cooperation Agreement, ABVC and Zhonghui agree to collaborate on developing a large-scale health and wellness base. The goal is to establish a comprehensive facility that offers a wide range of health and wellness services to promote a healthy lifestyle and well-being. Specifically, a healthcare center for senior living, long-term care, and medical care in the areas of ABVCs' special interests, such as Ophthalmology, Oncology, and Central Nervous Systems, is planned to be established as the base for the China market and global development. Both parties aim to build an integrated industry, academia, and research collaboration platform. This platform will also seek to contribute to rural revitalization by promoting innovation, knowledge exchange, and sustainable development in rural areas. Our strong partnership with leading institutions and expertise in Oncology/Hematology, Neurology, and Ophthalmology will aid us in global expansion."
About ABVC BioPharma
ABVC BioPharma is a clinical-stage biopharmaceutical company with an active pipeline of six drugs and one medical device (ABV-1701/Vitargus®) under development. For its drug products, the Company utilizes in-licensed technology from its network of world-renowned research institutions to conduct proof-of-concept trials through Phase II of clinical development. The Company's network of research institutions includes Stanford University, the University of California at San Francisco, and Cedars-Sinai Medical Center. For Vitargus®, the Company intends to conduct global clinical trials through Phase III.
1
Forward-Looking Statements
This press release contains "forward-looking statements." The words may precede such statements "intends," "may," "will," "plans," "expects," "anticipates," "projects," "predicts," "estimates," "aims," "believes," "hopes," "potential," or similar words. Forward-looking statements are not guarantees of future performance, are based on certain assumptions, and are subject to various known and unknown risks and uncertainties, many of which are beyond the Company's control, and cannot be predicted or quantified, and, consequently, actual results may differ materially from those expressed or implied by such forward-looking statements. None of the outcomes expressed herein are guaranteed. Such risks and uncertainties include, without limitation, risks and uncertainties associated with (i) our inability to manufacture our product candidates on a commercial scale on our own, or in collaboration with third parties; (ii) difficulties in obtaining financing on commercially reasonable terms; (iii) changes in the size and nature of our competition; (iv) loss of one or more key executives or scientists; and (v) difficulties in securing regulatory approval to proceed to the next level of the clinical trials or to market our product candidates. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's filings with the Securities and Exchange Commission (SEC), including the Company's Annual Report on Form 10-K and its Quarterly Reports on Form 10-Q. Investors are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. The Company assumes no obligation to publicly update or revise its forward-looking statements as a result of new information, future events or otherwise.
Contact:
Tom Masterson
Email: tmasterson@allelecomms.com
2
Cover |
Aug. 14, 2023 |
|---|---|
| Cover [Abstract] | |
| Document Type | 8-K |
| Amendment Flag | false |
| Document Period End Date | Aug. 14, 2023 |
| Entity File Number | 001-40700 |
| Entity Registrant Name | ABVC BIOPHARMA, INC. |
| Entity Central Index Key | 0001173313 |
| Entity Tax Identification Number | 26-0014658 |
| Entity Incorporation, State or Country Code | NV |
| Entity Address, Address Line One | 44370 Old Warm Springs Blvd. |
| Entity Address, City or Town | Fremont |
| Entity Address, State or Province | CA |
| Entity Address, Postal Zip Code | 94538 |
| City Area Code | 510 |
| Local Phone Number | 668-0881 |
| Written Communications | false |
| Soliciting Material | false |
| Pre-commencement Tender Offer | false |
| Pre-commencement Issuer Tender Offer | false |
| Title of 12(b) Security | Common Stock, par value $0.001 per share |
| Trading Symbol | ABVC |
| Security Exchange Name | NASDAQ |
| Entity Emerging Growth Company | false |
{
"instance": {
"ea183745-8k_abvcbio.htm": {
"axisCustom": 0,
"axisStandard": 0,
"baseTaxonomies": {
"http://xbrl.sec.gov/dei/2023": 22
},
"contextCount": 1,
"dts": {
"inline": {
"local": [
"ea183745-8k_abvcbio.htm"
]
},
"labelLink": {
"local": [
"abvc-20230814_lab.xml"
]
},
"presentationLink": {
"local": [
"abvc-20230814_pre.xml"
]
},
"schema": {
"local": [
"abvc-20230814.xsd"
],
"remote": [
"http://www.xbrl.org/2003/xbrl-instance-2003-12-31.xsd",
"http://www.xbrl.org/2003/xbrl-linkbase-2003-12-31.xsd",
"http://www.xbrl.org/2003/xl-2003-12-31.xsd",
"http://www.xbrl.org/2003/xlink-2003-12-31.xsd",
"http://www.xbrl.org/2005/xbrldt-2005.xsd",
"http://www.xbrl.org/2006/ref-2006-02-27.xsd",
"http://www.xbrl.org/lrr/role/negated-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/net-2009-12-16.xsd",
"https://www.xbrl.org/2020/extensible-enumerations-2.0.xsd",
"https://www.xbrl.org/dtr/type/2020-01-21/types.xsd",
"https://www.xbrl.org/dtr/type/2022-03-31/types.xsd",
"https://xbrl.fasb.org/srt/2023/elts/srt-2023.xsd",
"https://xbrl.fasb.org/srt/2023/elts/srt-roles-2023.xsd",
"https://xbrl.fasb.org/srt/2023/elts/srt-types-2023.xsd",
"https://xbrl.fasb.org/us-gaap/2023/elts/us-gaap-2023.xsd",
"https://xbrl.fasb.org/us-gaap/2023/elts/us-roles-2023.xsd",
"https://xbrl.fasb.org/us-gaap/2023/elts/us-types-2023.xsd",
"https://xbrl.sec.gov/country/2023/country-2023.xsd",
"https://xbrl.sec.gov/dei/2023/dei-2023.xsd"
]
}
},
"elementCount": 59,
"entityCount": 1,
"hidden": {
"http://xbrl.sec.gov/dei/2023": 2,
"total": 2
},
"keyCustom": 0,
"keyStandard": 22,
"memberCustom": 0,
"memberStandard": 0,
"nsprefix": "ABVC",
"nsuri": "http://abvcpharma.com/20230814",
"report": {
"R1": {
"firstAnchor": {
"ancestors": [
"span",
"b",
"p",
"body",
"html"
],
"baseRef": "ea183745-8k_abvcbio.htm",
"contextRef": "AsOf2023-08-14",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:DocumentType",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "document",
"isDefault": "true",
"longName": "00000001 - Document - Cover",
"menuCat": "Cover",
"order": "1",
"role": "http://abvcpharma.com/role/Cover",
"shortName": "Cover",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"b",
"p",
"body",
"html"
],
"baseRef": "ea183745-8k_abvcbio.htm",
"contextRef": "AsOf2023-08-14",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:DocumentType",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
}
},
"segmentCount": 0,
"tag": {
"dei_AmendmentDescription": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Description of changes contained within amended document.",
"label": "Amendment Description"
}
}
},
"localname": "AmendmentDescription",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "stringItemType"
},
"dei_AmendmentFlag": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the XBRL content amends previously-filed or accepted submission.",
"label": "Amendment Flag"
}
}
},
"localname": "AmendmentFlag",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_AnnualInformationForm": {
"auth_ref": [
"r14"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag with value true on a form if it is an annual report containing an annual information form.",
"label": "Annual Information Form"
}
}
},
"localname": "AnnualInformationForm",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_AuditedAnnualFinancialStatements": {
"auth_ref": [
"r14"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag with value true on a form if it is an annual report containing audited financial statements.",
"label": "Audited Annual Financial Statements"
}
}
},
"localname": "AuditedAnnualFinancialStatements",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_CityAreaCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Area code of city",
"label": "City Area Code"
}
}
},
"localname": "CityAreaCode",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_CountryRegion": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Region code of country",
"label": "Country Region"
}
}
},
"localname": "CountryRegion",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_CoverAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Cover page.",
"label": "Cover [Abstract]"
}
}
},
"localname": "CoverAbstract",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"xbrltype": "stringItemType"
},
"dei_CurrentFiscalYearEndDate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "End date of current fiscal year in the format --MM-DD.",
"label": "Current Fiscal Year End Date"
}
}
},
"localname": "CurrentFiscalYearEndDate",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "gMonthDayItemType"
},
"dei_DocumentAccountingStandard": {
"auth_ref": [
"r13"
],
"lang": {
"en-us": {
"role": {
"documentation": "The basis of accounting the registrant has used to prepare the financial statements included in this filing This can either be 'U.S. GAAP', 'International Financial Reporting Standards', or 'Other'.",
"label": "Document Accounting Standard"
}
}
},
"localname": "DocumentAccountingStandard",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "accountingStandardItemType"
},
"dei_DocumentAnnualReport": {
"auth_ref": [
"r11",
"r13",
"r14"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as an annual report.",
"label": "Document Annual Report"
}
}
},
"localname": "DocumentAnnualReport",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentFiscalPeriodFocus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Fiscal period values are FY, Q1, Q2, and Q3. 1st, 2nd and 3rd quarter 10-Q or 10-QT statements have value Q1, Q2, and Q3 respectively, with 10-K, 10-KT or other fiscal year statements having FY.",
"label": "Document Fiscal Period Focus"
}
}
},
"localname": "DocumentFiscalPeriodFocus",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "fiscalPeriodItemType"
},
"dei_DocumentFiscalYearFocus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "This is focus fiscal year of the document report in YYYY format. For a 2006 annual report, which may also provide financial information from prior periods, fiscal 2006 should be given as the fiscal year focus. Example: 2006.",
"label": "Document Fiscal Year Focus"
}
}
},
"localname": "DocumentFiscalYearFocus",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "gYearItemType"
},
"dei_DocumentPeriodEndDate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "For the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.",
"label": "Document Period End Date"
}
}
},
"localname": "DocumentPeriodEndDate",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "dateItemType"
},
"dei_DocumentPeriodStartDate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The start date of the period covered in the document, in YYYY-MM-DD format.",
"label": "Document Period Start Date"
}
}
},
"localname": "DocumentPeriodStartDate",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "dateItemType"
},
"dei_DocumentQuarterlyReport": {
"auth_ref": [
"r12"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as an quarterly report.",
"label": "Document Quarterly Report"
}
}
},
"localname": "DocumentQuarterlyReport",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentRegistrationStatement": {
"auth_ref": [
"r0"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as a registration statement.",
"label": "Document Registration Statement"
}
}
},
"localname": "DocumentRegistrationStatement",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentShellCompanyEventDate": {
"auth_ref": [
"r13"
],
"lang": {
"en-us": {
"role": {
"documentation": "Date of event requiring a shell company report.",
"label": "Document Shell Company Event Date"
}
}
},
"localname": "DocumentShellCompanyEventDate",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "dateItemType"
},
"dei_DocumentShellCompanyReport": {
"auth_ref": [
"r13"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true for a Shell Company Report pursuant to section 13 or 15(d) of the Exchange Act.",
"label": "Document Shell Company Report"
}
}
},
"localname": "DocumentShellCompanyReport",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentTransitionReport": {
"auth_ref": [
"r15"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as a transition report.",
"label": "Document Transition Report"
}
}
},
"localname": "DocumentTransitionReport",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentType": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.",
"label": "Document Type"
}
}
},
"localname": "DocumentType",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "submissionTypeItemType"
},
"dei_DocumentsIncorporatedByReferenceTextBlock": {
"auth_ref": [
"r3"
],
"lang": {
"en-us": {
"role": {
"documentation": "Documents incorporated by reference.",
"label": "Documents Incorporated by Reference [Text Block]"
}
}
},
"localname": "DocumentsIncorporatedByReferenceTextBlock",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "textBlockItemType"
},
"dei_EntityAddressAddressLine1": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Address Line 1 such as Attn, Building Name, Street Name",
"label": "Entity Address, Address Line One"
}
}
},
"localname": "EntityAddressAddressLine1",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressAddressLine2": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Address Line 2 such as Street or Suite number",
"label": "Entity Address, Address Line Two"
}
}
},
"localname": "EntityAddressAddressLine2",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressAddressLine3": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Address Line 3 such as an Office Park",
"label": "Entity Address, Address Line Three"
}
}
},
"localname": "EntityAddressAddressLine3",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressCityOrTown": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the City or Town",
"label": "Entity Address, City or Town"
}
}
},
"localname": "EntityAddressCityOrTown",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressCountry": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "ISO 3166-1 alpha-2 country code.",
"label": "Entity Address, Country"
}
}
},
"localname": "EntityAddressCountry",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "countryCodeItemType"
},
"dei_EntityAddressPostalZipCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Code for the postal or zip code",
"label": "Entity Address, Postal Zip Code"
}
}
},
"localname": "EntityAddressPostalZipCode",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressStateOrProvince": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the state or province.",
"label": "Entity Address, State or Province"
}
}
},
"localname": "EntityAddressStateOrProvince",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "stateOrProvinceItemType"
},
"dei_EntityBankruptcyProceedingsReportingCurrent": {
"auth_ref": [
"r6"
],
"lang": {
"en-us": {
"role": {
"documentation": "For registrants involved in bankruptcy proceedings during the preceding five years, the value Yes indicates that the registrant has filed all documents and reports required to be filed by Section 12, 13 or 15(d) of the Securities Exchange Act of 1934 subsequent to the distribution of securities under a plan confirmed by a court; the value No indicates the registrant has not. Registrants not involved in bankruptcy proceedings during the preceding five years should not report this element.",
"label": "Entity Bankruptcy Proceedings, Reporting Current"
}
}
},
"localname": "EntityBankruptcyProceedingsReportingCurrent",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntityCentralIndexKey": {
"auth_ref": [
"r2"
],
"lang": {
"en-us": {
"role": {
"documentation": "A unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK.",
"label": "Entity Central Index Key"
}
}
},
"localname": "EntityCentralIndexKey",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "centralIndexKeyItemType"
},
"dei_EntityCommonStockSharesOutstanding": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate number of shares or other units outstanding of each of registrant's classes of capital or common stock or other ownership interests, if and as stated on cover of related periodic report. Where multiple classes or units exist define each class/interest by adding class of stock items such as Common Class A [Member], Common Class B [Member] or Partnership Interest [Member] onto the Instrument [Domain] of the Entity Listings, Instrument.",
"label": "Entity Common Stock, Shares Outstanding"
}
}
},
"localname": "EntityCommonStockSharesOutstanding",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "sharesItemType"
},
"dei_EntityCurrentReportingStatus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' whether registrants (1) have filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that registrants were required to file such reports), and (2) have been subject to such filing requirements for the past 90 days. This information should be based on the registrant's current or most recent filing containing the related disclosure.",
"label": "Entity Current Reporting Status"
}
}
},
"localname": "EntityCurrentReportingStatus",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityEmergingGrowthCompany": {
"auth_ref": [
"r2"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate if registrant meets the emerging growth company criteria.",
"label": "Entity Emerging Growth Company"
}
}
},
"localname": "EntityEmergingGrowthCompany",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntityExTransitionPeriod": {
"auth_ref": [
"r19"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate if an emerging growth company has elected not to use the extended transition period for complying with any new or revised financial accounting standards.",
"label": "Elected Not To Use the Extended Transition Period"
}
}
},
"localname": "EntityExTransitionPeriod",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntityFileNumber": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Commission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.",
"label": "Entity File Number"
}
}
},
"localname": "EntityFileNumber",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "fileNumberItemType"
},
"dei_EntityFilerCategory": {
"auth_ref": [
"r2"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate whether the registrant is one of the following: Large Accelerated Filer, Accelerated Filer, Non-accelerated Filer. Definitions of these categories are stated in Rule 12b-2 of the Exchange Act. This information should be based on the registrant's current or most recent filing containing the related disclosure.",
"label": "Entity Filer Category"
}
}
},
"localname": "EntityFilerCategory",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "filerCategoryItemType"
},
"dei_EntityIncorporationStateCountryCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Two-character EDGAR code representing the state or country of incorporation.",
"label": "Entity Incorporation, State or Country Code"
}
}
},
"localname": "EntityIncorporationStateCountryCode",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "edgarStateCountryItemType"
},
"dei_EntityInteractiveDataCurrent": {
"auth_ref": [
"r16"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).",
"label": "Entity Interactive Data Current"
}
}
},
"localname": "EntityInteractiveDataCurrent",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityPrimarySicNumber": {
"auth_ref": [
"r14"
],
"lang": {
"en-us": {
"role": {
"documentation": "Primary Standard Industrial Classification (SIC) Number for the Entity.",
"label": "Entity Primary SIC Number"
}
}
},
"localname": "EntityPrimarySicNumber",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "sicNumberItemType"
},
"dei_EntityPublicFloat": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and asked price of such common equity, as of the last business day of the registrant's most recently completed second fiscal quarter.",
"label": "Entity Public Float"
}
}
},
"localname": "EntityPublicFloat",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "monetaryItemType"
},
"dei_EntityRegistrantName": {
"auth_ref": [
"r2"
],
"lang": {
"en-us": {
"role": {
"documentation": "The exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC.",
"label": "Entity Registrant Name"
}
}
},
"localname": "EntityRegistrantName",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityShellCompany": {
"auth_ref": [
"r2"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the registrant is a shell company as defined in Rule 12b-2 of the Exchange Act.",
"label": "Entity Shell Company"
}
}
},
"localname": "EntityShellCompany",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntitySmallBusiness": {
"auth_ref": [
"r2"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicates that the company is a Smaller Reporting Company (SRC).",
"label": "Entity Small Business"
}
}
},
"localname": "EntitySmallBusiness",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntityTaxIdentificationNumber": {
"auth_ref": [
"r2"
],
"lang": {
"en-us": {
"role": {
"documentation": "The Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS.",
"label": "Entity Tax Identification Number"
}
}
},
"localname": "EntityTaxIdentificationNumber",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "employerIdItemType"
},
"dei_EntityVoluntaryFilers": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.",
"label": "Entity Voluntary Filers"
}
}
},
"localname": "EntityVoluntaryFilers",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityWellKnownSeasonedIssuer": {
"auth_ref": [
"r17"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Is used on Form Type: 10-K, 10-Q, 8-K, 20-F, 6-K, 10-K/A, 10-Q/A, 20-F/A, 6-K/A, N-CSR, N-Q, N-1A.",
"label": "Entity Well-known Seasoned Issuer"
}
}
},
"localname": "EntityWellKnownSeasonedIssuer",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_Extension": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Extension number for local phone number.",
"label": "Extension"
}
}
},
"localname": "Extension",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_LocalPhoneNumber": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Local phone number for entity.",
"label": "Local Phone Number"
}
}
},
"localname": "LocalPhoneNumber",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_NoTradingSymbolFlag": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a security having no trading symbol.",
"label": "No Trading Symbol Flag"
}
}
},
"localname": "NoTradingSymbolFlag",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "trueItemType"
},
"dei_OtherReportingStandardItemNumber": {
"auth_ref": [
"r13"
],
"lang": {
"en-us": {
"role": {
"documentation": "\"Item 17\" or \"Item 18\" specified when the basis of accounting is neither US GAAP nor IFRS.",
"label": "Other Reporting Standard Item Number"
}
}
},
"localname": "OtherReportingStandardItemNumber",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "otherReportingStandardItemNumberItemType"
},
"dei_PreCommencementIssuerTenderOffer": {
"auth_ref": [
"r7"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act.",
"label": "Pre-commencement Issuer Tender Offer"
}
}
},
"localname": "PreCommencementIssuerTenderOffer",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_PreCommencementTenderOffer": {
"auth_ref": [
"r8"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act.",
"label": "Pre-commencement Tender Offer"
}
}
},
"localname": "PreCommencementTenderOffer",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_Security12bTitle": {
"auth_ref": [
"r1"
],
"lang": {
"en-us": {
"role": {
"documentation": "Title of a 12(b) registered security.",
"label": "Title of 12(b) Security"
}
}
},
"localname": "Security12bTitle",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "securityTitleItemType"
},
"dei_Security12gTitle": {
"auth_ref": [
"r5"
],
"lang": {
"en-us": {
"role": {
"documentation": "Title of a 12(g) registered security.",
"label": "Title of 12(g) Security"
}
}
},
"localname": "Security12gTitle",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "securityTitleItemType"
},
"dei_SecurityExchangeName": {
"auth_ref": [
"r4"
],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the Exchange on which a security is registered.",
"label": "Security Exchange Name"
}
}
},
"localname": "SecurityExchangeName",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "edgarExchangeCodeItemType"
},
"dei_SecurityReportingObligation": {
"auth_ref": [
"r9"
],
"lang": {
"en-us": {
"role": {
"documentation": "15(d), indicating whether the security has a reporting obligation under that section of the Exchange Act.",
"label": "Security Reporting Obligation"
}
}
},
"localname": "SecurityReportingObligation",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "securityReportingObligationItemType"
},
"dei_SolicitingMaterial": {
"auth_ref": [
"r10"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act.",
"label": "Soliciting Material"
}
}
},
"localname": "SolicitingMaterial",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_TradingSymbol": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Trading symbol of an instrument as listed on an exchange.",
"label": "Trading Symbol"
}
}
},
"localname": "TradingSymbol",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "tradingSymbolItemType"
},
"dei_WrittenCommunications": {
"auth_ref": [
"r18"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act.",
"label": "Written Communications"
}
}
},
"localname": "WrittenCommunications",
"nsuri": "http://xbrl.sec.gov/dei/2023",
"presentation": [
"http://abvcpharma.com/role/Cover"
],
"xbrltype": "booleanItemType"
}
},
"unitCount": 3
}
},
"std_ref": {
"r0": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r1": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r10": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "14a",
"Subsection": "12",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r11": {
"Name": "Form 10-K",
"Number": "249",
"Publisher": "SEC",
"Section": "310",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r12": {
"Name": "Form 10-Q",
"Number": "240",
"Publisher": "SEC",
"Section": "308",
"Subsection": "a",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r13": {
"Name": "Form 20-F",
"Number": "249",
"Publisher": "SEC",
"Section": "220",
"Subsection": "f",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r14": {
"Name": "Form 40-F",
"Number": "249",
"Publisher": "SEC",
"Section": "240",
"Subsection": "f",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r15": {
"Name": "Forms 10-K, 10-Q, 20-F",
"Number": "240",
"Publisher": "SEC",
"Section": "13",
"Subsection": "a-1",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r16": {
"Name": "Regulation S-T",
"Number": "232",
"Publisher": "SEC",
"Section": "405",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r17": {
"Name": "Securities Act",
"Number": "230",
"Publisher": "SEC",
"Section": "405",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r18": {
"Name": "Securities Act",
"Number": "230",
"Publisher": "SEC",
"Section": "425",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r19": {
"Name": "Securities Act",
"Number": "7A",
"Publisher": "SEC",
"Section": "B",
"Subsection": "2",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r2": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-2",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r3": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-23",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r4": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "d1-1",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r5": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "g",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r6": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12, 13, 15d",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r7": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "13e",
"Subsection": "4c",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r8": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "14d",
"Subsection": "2b",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r9": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "15",
"Subsection": "d",
"role": "http://www.xbrl.org/2003/role/presentationRef"
}
},
"version": "2.2"
}