
SECURITIES
AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
__________________
SCHEDULE 14D-9
SOLICITATION/RECOMMENDATION STATEMENT
PURSUANT TO SECTION 14(d)(4) OF THE
SECURITIES EXCHANGE ACT OF 1934
(Amendment No. 3)
ELAN
CORPORATION, PLC
(Name of Subject Company)
ELAN
CORPORATION, PLC
(Names of Person Filing Statement)
______________________________
Ordinary Shares, par value €0.05
each
(Title of Class of Securities)
______________________________
G29539106
(CUSIP Number of Class of Securities)
______________________________
American Depositary Shares, each representing
one Ordinary Share
(Title of Class of Securities)
______________________________
284131208
(CUSIP Number of Class of Securities)
______________________________
|
William F. Daniel Elan Corporation, plc Treasury Building Dublin 2, Ireland 011-353-1-709-4000
Notice and Communications on Behalf of the Person Filing Statement)
|
| Copies to: |
|
Christopher T. Cox, Esq. Cadwalader, Wickersham & Taft LLP One World Financial Center New York, New York 10281 (212) 504-6000 |
| [_] | Check the box if the filing relates solely to preliminary communications made before the commencement of a tender offer. |
This
Amendment No. 3 to Schedule 14D-9 amends and supplements the Solicitation/Recommendation Statement on
Schedule 14D-9 (as amended from time to time, the “Statement”)
originally filed by Elan Corporation, plc, a public limited company organized under the laws of Ireland
(“Elan”), with the Securities and Exchange Commission on
May 15, 2013, relating to the unsolicited offer by Echo Pharma Acquisition Limited, a private limited company
incorporated under the laws of Ireland (“Royalty Pharma”), as
disclosed in the Tender Offer Statement on Schedule TO, dated May 2, 2013, as amended, to exchange each outstanding
ordinary share of €0.05 each of Elan (“Elan Shares”),
including Elan Shares represented by American Depositary Shares, for US$12.50 in cash, upon the terms and subject to the
conditions set forth in (i) the Cash Offer, dated May 2, 2013, as amended (the “Offer
Document”), and (ii) the related revised forms of acceptance and Letter
of Transmittal. Except as specifically noted herein, the information set forth in the Statement remains unchanged.
| ITEM 4. | THE SOLICITATION OR RECOMMENDATION. |
Item 4 is hereby amended and supplemented by reference to the information under the heading “Royalty Pharma Offer Substantially Undervalues Elan Today” in Part 1 of Exhibit (a)(8) hereto.
| ITEM 8. | ADDITIONAL INFORMATION. |
Section 8.1 of Exhibit (a)(1) to the Statement is hereby amended and supplemented to include the following as the penultimate paragraph thereof:
“The Theravance Agreement provides an undertaking by Elan that the shareholder circular relating to the EGM will contain a recommendation from Elan’s board of directors to its shareholders to vote in favor of the Theravance Transactions and that such recommendations will not be withdrawn, modified or altered in any way without Theravance’s prior written consent. If the Elan board were to breach this provision of the Theravance Agreement, Elan could be exposed to a claim for damages for breach of contract and such damages would not be subject to limitation or “cap” under any provision of the Theravance Agreement.”
| ITEM 9. | EXHIBITS. |
| The following exhibits are filed herewith or incorporated herein by reference: |
|
Exhibit |
Description | ||
| (a)(1)* | Response to Royalty Pharma’s offer by the board of directors of Elan, May 15, 2013 (incorporated by reference to Exhibit (a)(1) to the Solicitation/Recommendation Statement on Schedule 14D-9 of Elan Corporation, plc filed with the Commission on May 15, 2013) | ||
| (a)(5) | Royalty Participation Agreement between Theravance, Inc. and Elan Corporation, plc dated May 12, 2013 (incorporated by reference to Exhibit 99.1 of the Report of Foreign Issuer on Form 6-K of Elan Corporation, plc filed with the Commission on May 17, 2013) | ||
| (a)(6) | Share Purchase Agreement by and between Dr. Rudolf Stefan Widmann, SASR Neunundvierzigste Beteiligungsverwaltung GmbH and Elan Corporation, plc dated May 19, 2013 | ||
| (a)(7) | Transaction Agreement between Elan Pharma International Limited and Nerano Pharma Limited dated May 19, 2013 | ||
| (a)(8) | Circular to Shareholders and ADS Holders, dated May 27, 2013 | ||
| (a)(9) |
Press release, dated May 27, 2013 | ||
* Previously filed. | |||
| 2 |
SIGNATURE
After due inquiry and to the best of my knowledge and belief, I certify that the information set forth in this statement is true, complete and correct.
| ELAN CORPORATION, PLC | ||
| By: | /s/ William F. Daniel | |
| Name: William F. Daniel Title: Executive Vice President and Company Secretary | ||
Dated: May 28, 2013
| 3 |
EXHIBIT INDEX
| ITEM 9. | EXHIBITS. |
| The following exhibits are filed herewith or incorporated herein by reference: |
|
Exhibit |
Description | |
| (a)(1)* | Response to Royalty Pharma’s offer by the board of directors of Elan, May 15, 2013 (incorporated by reference to Exhibit (a)(1) to the Solicitation/Recommendation Statement on Schedule 14D-9 of Elan Corporation, plc filed with the Commission on May 15, 2013) | |
| (a)(5) | Royalty Participation Agreement between Theravance, Inc. and Elan Corporation, plc dated May 12, 2013 (incorporated by reference to Exhibit 99.1 of the Report of Foreign Issuer on Form 6-K of Elan Corporation, plc filed with the Commission on May 17, 2013) | |
| (a)(6) | Share Purchase Agreement by and between Dr. Rudolf Stefan Widmann, SASR Neunundvierzigste Beteiligungsverwaltung GmbH and Elan Corporation, plc dated May 19, 2013 | |
| (a)(7) | Transaction Agreement between Elan Pharma International Limited and Nerano Pharma Limited dated May 19, 2013 | |
| (a)(8) | Circular to Shareholders and ADS Holders, dated May 27, 2013 | |
| (a)(9) |
Press release, dated May 27, 2013
|
* Previously filed.
| 4 |
Exhibit (a)(6)
SHARE PURCHASE AGREEMENT
by and between
Dr. Rudolf Stefan Widmann
born 24.03.1957
Wintergasse 85/1b
3002 Purkersdorf
(the "Seller")
ON THE ONE HAND
and
SASR Neunundvierzigste Beteiligungsverwaltung GmbH
FN 395723 x
Tuchlauben 17
1010 Vienna
(the "Purchaser 1")
and
Elan Corporation, plc
Registered number 30356
Treasury Building
Lower Grand Canal Street
Dublin 2
Ireland
(the "Purchaser 2")
ON THE OTHER HAND
as follows:
2
Table of Contents
| 1 | Definitions | 3 |
| 2 | Recitals | 11 |
| 3 | Sale and Purchase of the Shares | 12 |
| 4 | Purchase Price and Payment | 13 |
| 5 | Representations and Warranties of the Seller | 14 |
| 6 | Representations and Warranties of the Purchasers | 30 |
| 7 | Recovery by the Purchasers | 33 |
| 8 | Conduct of Business during the Interim Period. Covenants of the Seller | 37 |
| 9 | Conditions Precedent | 40 |
| 10 | Signing and Closing | 42 |
| 11 | Purchaser 2 Guarantee | 45 |
| 12 | Confidentiality | 45 |
| 13 | Costs and Expenses | 45 |
| 14 | Governing Law and Dispute Resolution | 45 |
| 15 | Final Provisions | 46 |
3
| 1 | Definitions |
| ABGB | means the Austrian General Civil Code (Allgemeines Bürgerliches Gesetzbuch) as amended from time to time; | ||
| Agreement | means this Share Purchase Agreement; | ||
| Articles | means the Company's current articles of association (Satzung); | ||
| Audited Financial Statements 2012 |
means the Financial Statements 2012 having been audited by the Company's auditor and having received an unqualified audit opinion; | ||
| Balance Sheet Date | means 31 December 2012; | ||
| Base Purchase Price | has the meaning set out in Clause 4.1; | ||
| Best Efforts | or a similar expression shall mean a covenant of the respective Party within the meaning of § 880a first case ABGB (Verwendungszusage); | ||
| Break-up Event | has the meaning set out in Clause 9.3; | ||
| Break-up Fee | means EUR 5,000,000 (Euro five million) plus VAT, if any; | ||
| Business | means researching, developing, manufacturing, marketing, distributing and selling pharmaceutical and medicinal products, as currently conducted by the Company and the Subsidiary in the areas of non-generic, rare/orphan diseases including without limitation in the therapeutic areas of hematology, oncology, cardiology, pulmonology, neurology, psychiatry and metabolic diseases in the territories in which the Company and the Subsidiary currently conduct business; | ||
| Business Day | means any day (other than a Saturday or |
4
| Sunday) on which banks are open to the general public in Vienna, Austria and Dublin, Ireland; | |||
| Cash Purchase Price | has the meaning set out in Clause 4.2; | ||
| Claim(s) | means a demand or assertion by a claimant for compensation, payment, or reimbursement for a Loss; | ||
| Closing | has the meaning set out in Clause 10.2; | ||
| Closing Date | has the meaning set out in Clause 10.2; | ||
| Closing Confirmation Letter | has the meaning set out in Clause 10.6; | ||
| Company | means AOP Orphan Pharmaceuticals Aktiengesellschaft with corporate seat in Vienna and business address at Wilhelminenstraße 91/IIf, 1160 Vienna, Austria, registered in the companies' register of the Vienna Commercial Court under FN 237770 m; | ||
| Conditions Precedent | has the meaning set out in Clause 9.1; | ||
| Consideration Shares | means such number of validly and newly issued and fully paid-up ordinary shares of Purchaser 2 which corresponds to an amount of EUR 87,833,333 (Euro eighty seven million eight hundred thousand three hundred thirty three) divided by the closing price of Purchaser 2's ordinary shares on the Main Securities Market of the Irish Stock Exchange in Dublin on the trading day immediately prior to the Closing Date, rounded up to the nearest whole share, having a nominal value of EUR 0.05 and carrying one vote per share; | ||
| Contingent Purchase Price | has the meaning set out in Clause 4.4; | ||
| CREST | means an electronic system, which settles transfers of shares through selected exchanges in Ireland; |
5
| De Minimis Amount | has the meaning set out in Clause 7.2.1.1; | ||
| Disclosed Documents | means the documents listed in Exhibit 5.34 and contained on USB sticks as further listed and described in the screenshots attached to Exhibit 5.34 which have been delivered to the Purchasers by the Seller on or before 00:00 am CET on 18 May 2013; | ||
| EBITDA | means net income/loss for the year (Jahresüberschuss/-fehlbetrag) pursuant to § 231 para (2) no. 22 UGB plus or minus taxes on income (Steuern vom Einkommen und Ertrag) pursuant to § 231 para (2) no. 21 UGB plus extraordinary expenses (außerordentliche Aufwendungen) pursuant to § 231 para (2) no 19 UGB minus extraordinary income (außerordentliche Erträge) pursuant to § 231 para (2) no 18 UGB plus interest and similar expenses (Zinsen und ähnliche Aufwendungen) pursuant to § 231 para (2) no 15 UGB plus expenses on financial assets and current securities (Aufwendungen aus Finanzanlagen und aus Wertpapieren des Umlaufvermögens) pursuant to § 231 para (2) no 14 UGB minus income from the disposal or revaluation of financial assets and current securities (Erträge aus dem Abgang von und der Zuschreibung zu Finanzanlagen und Wertpapieren des Umlaufvermögens) pursuant to § 231 para (2) no 13 UGB minus other interest and similar income (sonstige Zinsen und ähnliche Erträge) pursuant to § 231 para (2) no 12 UGB minus income from other long-term securities and and loans (Erträge aus anderen Wertpapieren und Ausleihungen des Finanzanlagevermögens) pursuant to § 231 para (2) no 11 UGB minus income from participating interests (Erträge aus Beteiligungen) pursuant to § 231 para (2) no 10 UGB plus depreciation and amortisation (Abschreibungen) pursuant to § 231 para (2) no 7a) and no 7b) UGB; |
6
| Effective Date | means the Closing Date; | ||
| Encumbrance | means any claim, charge, mortgage, pledge, hypothecation, lien, usufruct, retention of title or other third party right or security interest of any kind, option, equitable right, power of sale, right of pre-emption, right of first refusal or an agreement, arrangement or obligation to create any of the foregoing; | ||
| EUR | means the Euro, the lawful currency of the member states of the European Union that adopt the single currency in accordance with the EC Treaty; | ||
| Financial Statements | means the Financial Statements 2010, the Financial Statements 2011 and the Financial Statements 2012 collectively; | ||
| Financial Statements 2010 | means the audited annual financial statements of the Company as of 31 December 2010, which have been drawn up in accordance with the UGB and Austrian generally accepted accounting principles; | ||
| Financial Statements 2011 | means the audited annual financial statements of the Company as of 31 December 2011, which have been drawn up in accordance with the UGB and Austrian generally accepted accounting principles; | ||
| Financial Statements 2012 | means the draft annual financial statements of the Company as of 31 December 2012, which have been drawn up in accordance with the UGB and Austrian generally accepted accounting principles; | ||
| Intellectual Property Rights | means patents, trademarks (including trade dress and service marks), internet domain names (and corresponding rights thereto), trade secrets and copyrights; | ||
| Interim Period | means the period between Signing and (including) Closing; |
7
| IP Rights Assignment Agreement |
has the meaning set out in Clause 10.4.2; | ||
| Licenses | has the meaning set out in Clause 5.27.2; | ||
| Long-Stop Date | has the meaning set out in Clause 9.2; | ||
| Loss | means any and all damages (excluding lost profit, consequential damages and indirect damages), liabilities, losses, defects, Taxes, penalties, expenses, assessments, judgements or deficiencies of any nature whatsoever (including, without limitation, reasonable attorney's fees and expenses, consultant's and investigator's fees and expenses, and other reasonable costs and expenses incident to any suit, action or proceeding); | ||
| Marketing Authorisations | means any marketing authorisation (or applications) relating to the Products and filed by or on behalf of the Company, including, but not limited to, the marketing authorisations listed in Exhibit 5.27.3; | ||
| Material Adverse Effect | means the occurrence of any of the following events: |
| (i) | the Seller ceasing to be the chief executive officer of the Company; | |||
| (ii) | the Audited Financial Statements 2012 deviating from the Financial Statements 2012; or | |||
| (iii) | any other event which materially adversely affects the state of affairs, the assets and liabilities, the financial condition, the profits, the prospects or the results of operation of the Company, the Subsidiary or the Business, |
| if and to the extent such event(s) occur(s) between Signing and Closing and, in the case of lit (ii) only, if the deviation, taken |
8
| as a whole, resulted in or can reasonably be expected to result in a decline in the EBITDA of the Company of 30 % or more between (1) the Financial Statements 2012 and (2) the Audited Financial Statements 2012; | |||
| Material Contract(s) | means an agreement or agreements to which the Company or the Subsidiary is a party and which is/are materially relevant to the Programs; | ||
| Other Occupiers | means all past or present owners, tenants, licensees or other occupiers or users (lawful or unlawful) of any Properties, other than the Company and the Subsidiary; | ||
| Parties | means Seller and Purchasers collectively; | ||
| Party | means any of the Parties individually; | ||
| Person(s) | means any natural person, corporation, firm, business trust, joint venture, association, organization, company, partnership or other business entity, or any government or any agency or political subdivision thereof; | ||
| Products | means all products currently developed, marketed, distributed, sold and/or commercialised by the Company or the Subsidiary as set out in Exhibit 1; | ||
| Programs | means any and all of Landiolol, Peg-IFN-a 2b, Anagrelide Retard and Resimmune; | ||
| Properties | means the properties owned, occupied or used by the Company as listed in Exhibit 5.15.2; | ||
| Purchase Price | means the Cash Purchase Price, the Consideration Shares and, if applicable, the Contingent Purchase Price; | ||
| Purchaser | means any of the Purchasers individually; |
9
| Purchaser 1 | means SASR Neunundvierzigste Beteiligungsverwaltung GmbH, an Austrian limited liability company with corporate seat in Vienna and business address at Tuchlauben 17, 1010 Vienna, registered in the companies' register of the Vienna Commercial Court under FN 395723 x; | ||
| Purchaser 2 | means Elan Corporation plc, an Irish public limited company with corporate seat in Dublin and business address at Treasury Building, Lower Grand Canal Street, Dublin 2, Ireland, registered in under Company Number 30356; | ||
| Purchasers | means Purchaser 1 and Purchaser 2 collectively; | ||
| Registered Share Capital | means the registered nominal share capital (Grundkapital) of the Company, amounting to EUR 200,000 (Euro two hundred thousand), fully paid-up; | ||
| Seller | means Dr. Rudolf Stefan Widmann, born on 24 March 1957 and residing at Wintergasse 85/1b, 3002 Purkersdorf, Austria; | ||
| Share | means a common share of the Company, issued to bearer (Inhaberaktie), with no par-value (Stückaktie) and representing a pro rata amount of the Registered Share Capital (anteiliger Betrag am Grundkapital) of EUR 10 (Euro ten) per share; | ||
| Shares | means a total of 20.000 (twenty thousand) no par-value bearer shares (auf Inhaber lautende Stückaktien) in the Company owned by the Seller and representing 100 % of the Company's Registered Share Capital; | ||
| Signing | has the meaning set out in Clause 10.1; | ||
| Signing Date | means the date of execution of this Agreement; |
10
| Subsidiary | means AOP Orphan Limited, an English limited liability company with corporate seat in Weybridge and business address at Midgley Snelling, Ibex House, Baker Street, Weybridge, Surrey KT13 8AH, registered in the companies register of the Companies House Cardiff under 8082765; | ||
| Subsidiary Shares | means all the shares in the Subsidiary, fully paid up, as held by the Company, corresponding to a participation in the Subsidiary of 100 %; | ||
| Tax Authority | means any official authority or public body that is competent to impose or declare Taxes; | ||
| Tax Returns | means any return, report, declaration, statement, claim for refund or information return or statement required to be filed with any Tax Authority in relation to Taxes; | ||
| Tax or Taxes | means all kinds of Austrian and/or foreign taxes, tax allocations (Steuerumlagen), charges, fees, duties, levies, penalties or other assessments whether direct or indirect, including corporate income tax, income tax, capital gains tax, wage tax, trade taxes, VAT, stamp duties and transfer taxes (Gebühren und Verkehrsteuern), provincial and community taxes (Landes- und Gemeindeabgaben) and customs duties as well as social security contributions, in each case together with any interest, late interest, penalties or additions (including late filing fees (Verspätungszuschlag) and/or late payment fees (Säumniszuschlag)) attributable thereon; | ||
| Tetmodis | means one of the Products called Tetmodis®, which contains the compound Tetrabenazine; | ||
| Third Party or Third Parties | shall mean a Person who or which is not a |
11
| Party; | |||
| Third Party Claim | has the meaning set out in Clause 7.5.1; | ||
| Third Party IP Rights | has the meaning set out in Clause 5.14.3; | ||
| Threshold | means EUR 2,000,000 (Euro two million); | ||
| Title | means the instrument that gives rise to a legal right of possession or control; | ||
| UGB | means the Austrian Enterprise Act (Unternehmensgesetzbuch) as amended from time to time; | ||
| US GAAP | means accounting principles and practices generally accepted in the United States of America, as in effect on the date of the relevant financial statements; | ||
| Vienna Rules | means the Rules of Arbitration and Conciliation of the International Arbitral Centre of the Economic Chamber Austria in Vienna, Austria. |
| 2 | Recitals |
| 2.1 | The Company is a stock corporation (Aktiengesellschaft) duly established and existing under the laws of Austria with corporate seat in Vienna and business address at Wilhelminenstraße 91/IIf, 1160 Vienna, Austria, registered in the companies' register of the Vienna Commercial Court under FN 237770 m. The Company's Registered Share Capital amounts to EUR 200,000 (Euro two hundred thousand), fully paid up. |
| 2.2 | The Company is a pharmaceutical company focussed on researching, developing, manufacturing, marketing, distributing and selling pharmaceutical and medicinal products including in the areas of hematology, oncology, cardiology, pulmonology, neurology, psychiatry and metabolic diseases. |
| 2.3 | Subject to the terms and conditions of this Agreement, the Seller wants to sell and transfer to the Purchasers the Shares, which represent 100 % of the Company's Registered Share Capital, and the Purchasers want to purchase and accept the transfer of the Shares from the Seller. |
12
| 3 | Sale and Purchase of the Shares |
| 3.1 | The Registered Share Capital is divided into 20,000 common shares (Stammaktien), issued to bearer (Inhaberaktien), with no par-value (Stückaktien), representing a pro rata amount of the Registered Share Capital (anteiliger Betrag am Grundkapital) of EUR 10 (Euro ten) per Share. The Seller owns 20,000 (twenty thousand) common shares represented by global interim certificates (Sammelzwischenscheine) numbers 1 to 8, representing shares no. 1 through 8.000, numbers 9 to 16, representing shares no. 8.001 through 16.000, numbers 17 to 18, representing shares no. 16.001 through 18.000 and numbers 19 to 20, representing shares no. 18.001 through 20.000 (collectively the "Shares"). |
| 3.2 | Subject to the terms and conditions of this Agreement, the Seller hereby undertakes to sell and, subject to the satisfaction of the Conditions Precedent, at Closing shall transfer: |
| 3.2.1 | 13,333 of the Shares to Purchaser 1 and Purchaser 1 hereby undertakes to purchase and, subject to the satisfaction of the Conditions Precedent, at Closing shall accept from the Seller such 13,333 of the Shares; and |
| 3.2.2 | 6,667 of the Shares to Purchaser 2 and Purchaser 2 hereby undertakes to purchase and, subject to the satisfaction of the Conditions Precedent, at Closing shall accept from Seller such 6,667 of the Shares. |
| 3.3 | The transfer of the global interim certificates (Sammelzwischenscheine) representing the Shares shall occur at Closing upon payment of the Base Purchase Price pursuant to Clause 4.5. At Closing, the Seller shall deliver and hand over |
| 3.3.1 | to Purchaser 1 the original(s) of the global interim certificate(s) (Sammelzwischenschein(e)) representing 13,333 Shares; and |
| 3.3.2 | to Purchaser 2 the original(s) of the global interim certificate(s) (Sammelzwischenschein(e)) representing 6,667 Shares. |
| 3.4 | The transfer of the Shares shall have economic effect as from the Closing Date (the "Effective Date") and, unless otherwise expressly stated in this Agreement, all rights (including, without limitation, the right to receive dividends or distributions declared, made or paid) and obligations, benefits and risk (including, without limitation, economic risk, force majeure risk, etc.) associated with the Shares shall pass from the Seller to the Purchasers as of the Effective Date. |
| 3.5 | Title to the Shares shall pass to the Purchasers upon completion of all Closing actions pursuant to Clause 10.4. |
| 3.6 | The Seller waives any and all pre-emption rights or other rights or restrictions on transfer in respect of the Shares or interests in the Shares to which he may be entitled under the Articles, any previous articles of association of the Company, or otherwise, enforcement of or compliance with which may or might restrict, prevent |
13
or delay the performance of the provisions of this Agreement, and shall procure the waiver of all other rights of pre-emption as may be required to give full effect to the provisions of this Agreement.
| 4 | Purchase Price and Payment |
| 4.1 | The purchase price for the Shares (the "Purchase Price") shall consist of a Cash Purchase Price pursuant to Clause 4.2, the issuance of Consideration Shares to the Seller pursuant to Clause 4.3 (collectively with the Cash Purchase Price the "Base Purchase Price") and, if applicable, a Contingent Purchase Price pursuant to Clause 4.4. |
| 4.2 | The cash purchase price for the Shares shall amount to EUR 175,666,667 (EUR one hundred seventy five million six hundred sixty six thousand six hundred sixty seven), (the "Cash Purchase Price"). |
| 4.3 | As additional consideration for the sale and transfer of the Shares to the Purchasers, the Seller shall at Closing receive (in total) such number of validly and newly (from authorized capital) issued and fully paid-up ordinary shares of Purchaser 2 which corresponds to an amount of EUR 87,833,333 (Euro eighty seven million eight hundred thousand three hundred thirty three) divided by the closing price of Purchaser 2's ordinary shares on the Main Securities Market of the Irish Stock Exchange in Dublin on the trading day immediately prior to the Closing Date, rounded up to the nearest whole share, having a nominal value of EUR 0.05, carrying one vote per share and listed on the Main Securities Market of the Irish Stock Exchange in Dublin (collectively the "Consideration Shares"). The Consideration Shares shall be delivered to the Seller at Closing in accordance with Clause 4.5.2. |
| 4.4 | Subject to the terms and conditions set forth in Exhibit 4.4, the Seller shall be entitled to a contingent purchase price, if any, (the "Contingent Purchase Price") in addition to the Base Purchase Price. The maximum amount of the Contingent Purchase Price shall be EUR 270,000,000 (Euro two hundred seventy million). |
| 4.5 | The Base Purchase Price shall be due and payable at Closing as follows: |
| 4.5.1 | the Cash Purchase Price shall be paid by Purchaser 1 to the Seller to the Seller's bank account details of which are set forth in Exhibit 4.5.1; and |
| 4.5.2 | the Consideration Shares shall be delivered by Purchaser 2 to the Seller in book entry form through CREST, to a CREST account to be opened by the Seller with a reputable credit institution/broker. The Seller shall notify Purchaser 2 at the latest 5 (five) Business Days prior to the Closing Date of the following details of his CREST account: his CREST account number, the name of the credit institution/broker where the Seller opened his CREST account, and any other details reasonably required by Purchaser 2 to deliver the Consideration Shares to the Seller through CREST. |
14
| 4.5.3 | Payment of the Cash Purchase Price and delivery of the Consideration Shares in accordance with this Clause 4.5 shall discharge the Purchasers' liability in respect of the Base Purchase Price towards the Seller. |
| 4.6 | The Contingent Purchase Price, if any, shall be due and payable by Purchaser 1 as further set forth in Exhibit 4.4. |
| 4.7 | All payments to be made pursuant to this Agreement, shall be made when due by irrevocable SWIFT wire transfer of immediately available funds, free and clear of all costs, fees, deductions and charges in EUR, excluding in respect of the Consideration Shares which shall be credited to a CREST account nominated by the Seller. |
| 5 | Representations and Warranties of the Seller |
| 5.1 | The Seller represents and warrants to the Purchasers that each of the representations and warranties as set out in this Clause 5 are correct, complete and not misleading, unless otherwise expressly stated in this Clause 5, as of Signing and Closing. References to the best knowledge, belief or awareness of Seller (or similar phrases) shall mean the actual knowledge of the Seller after due inquiry. |
| 5.2 | Organisation, Power and Authority. |
| 5.2.1 | The Seller has full power and authority to own, hold and sell the Shares and to execute and deliver this Agreement and the Shares and to consummate the transactions contemplated hereunder. |
| 5.2.2 | The Company is a stock corporation (Aktiengesellschaft), duly incorporated and organized and validly existing under the laws of Austria and has the power and the necessary licenses and approvals to own its assets and lease its properties and to carry on its Business as currently being conducted. The Company is not over-indebted, insolvent, subject to a procedure under the Companies Reorganisation Act (Unternehmensreorganisationsgesetz) or comparable procedure, nor is such insolvency, over-indebtedness or procedure threatening to the Company. |
| 5.2.3 | The Articles of the Company as currently in force are attached hereto as Exhibit 5.2.3; no shareholder resolution has been passed to amend these articles of association. All necessary meetings and other corporate actions of or by the Company, its shareholders and management and supervisory boards have been legally and properly held or taken, and all resolutions passed by the meetings of shareholders and directors have been duly recorded as required under the Austrian Stock Corporation Act (AktG) and all necessary filing have been made with the companies register. There exists no shareholder, cooperation agreement or voting arrangement or similar arrangement in relation to the Company. |
15
| 5.2.4 | The companies register excerpt of the Company attached hereto as Exhibit 5.2.4 is accurate and complete in all respects. |
| 5.3 | Shares and Registered Share Capital. |
| 5.3.1 | The total authorized, issued and outstanding share capital of the Company is EUR 200,000 (Euro two hundred thousand), fully paid up. The Registered Share Capital is divided into 20,000 common shares (Stammaktien), issued to bearer (Inhaberaktien), with no par-value (Stückaktien), representing a pro rata amount of the Registered Share Capital (anteiliger Betrag am Grundkapital) of EUR 10 (Euro ten) per Share. There exists no option, right of conversion or other issue of shares or securities, which could increase or reduce the number of the shares of the Company or the Company's Registered Share Capital. The nominal share capital of the Company has not been decreased or redeemed; there has not been any unlawful repayment of share capital (Einlagenrückgewähr). |
| 5.3.2 | The Shares correspond to a participation in the Company of 100 % (one hundred percent). The Shares are the only shares issued by the Company and each of the Shares carries the same pro rata voting rights and other rights attaching to it, including without limitation, dividend rights, as all other shares. |
| 5.3.3 | The Shares are fully paid-up and free and clear of all Encumbrances. The Seller is the sole and unrestricted legal and beneficial owner of all of the Shares. Upon Closing, the Purchasers will acquire unrestricted legal and beneficial ownership title to all Shares. |
| 5.3.4 | There are no reasons why the Purchasers may be obliged to pay supplementary contributions, other payments or capital increases to the Company or why it would be liable for unpaid capital contributions to the Seller, his predecessor(s) in interest or other shareholders. |
| 5.3.5 | Other than as set forth in the Articles, there are no outstanding stock rights, options, warrants or rights of first refusal, pre-emptive, subordination or similar rights or other rights to purchase, obtain or acquire, or any outstanding securities or obligations convertible into or exchangeable for, or any voting or other agreements with respect to, any shares of the share capital of the Company, or any other securities of the Company. The Company is not currently obligated, now or in the future, contingently or otherwise, to issue, allot, purchase or redeem any shares of the share capital or any other securities to any person or entity. There are no treasury shares of the Company. There are no outstanding bonds, debentures, notes or other indebtedness having the right to vote on any matters on which shareholders of the Company may vote. |
16
| 5.4 | All consents and approvals, including without limitation consents and approvals under applicable corporate law, for the transfer of the Shares on the terms and conditions set out in this Agreement, have been obtained by the Seller. |
| 5.5 | Except for the Subsidiary, the Company does not hold any shares or any other participations or interest in any other company, partnership, business organization or other entity. |
| 5.6 | The execution, delivery and performance of this Agreement by the Seller and the consummation of the transactions contemplated hereby do not and will not, with or without the giving of any notice or the lapse of time, or both, (i) violate, conflict with, result in the breach of, require a consent under, allow any party to prematurely terminate or accelerate the performance required by any of the terms, conditions or provisions of the Company's Articles or other governing documents of the Company or any covenant, agreement, contract or understanding to which the Seller is a party or any order, ruling, decree, judgement, arbitration award, law, rule, regulation or stipulation to which the Seller or the Company is subject; or (ii) constitute a default thereunder; or (iii) result in the creation of any Encumbrance of any nature whatsoever upon any of the properties or assets of the Company. There is no action, lawsuit, investigation or proceeding pending or threatened against the Company or the Seller before any court, arbitration panel or governmental authority which in any manner challenges or seeks to prevent, alter or delay the transactions contemplated herein. |
| 5.7 | Financials. |
| 5.7.1 | The Financial Statements have been prepared in accordance with the UGB and Austrian generally accepted accounting principles, applied on a consistent basis, maintaining the previous accounting and valuation principles, methods and rules without material changes. The Financial Statements 2010 and 2011 have been audited and received unqualified audit opinions. The Financial Statements 2012 are attached hereto as Exhibit 5.7.1. |
| 5.7.2 | The Financial Statements are true and correct, consistent in all respects with the books and records of the Company, and present a complete and accurate view of the state of affairs, the assets and liabilities (Vermögenslage), the financial condition (Finanzlage), and the results of operation (Ertragslage) of the Company for the periods referenced therein. There are no liabilities, actual or contingent, except as disclosed in the Financial Statements or incurred in the ordinary course of the Business since the Balance Sheet Date in accordance with this Agreement. Full provision for all liabilities, actual or contingent, has been made in the Financial Statements. The Company has no material capital commitments. |
| 5.7.3 | The monthly management accounts of the Company for the business year 2013 as disclosed to the Purchasers prior to Signing are true and |
17
correct, consistent in all material respects with the books and records of the Company, have been prepared on a basis consistent with that adopted in the preparation of the Financial Statements, and present – in all material respects – a true and fair view of the state of affairs, the assets and liabilities (Vermögenslage), the financial condition (Finanzlage), and the results of operation (Ertragslage) of the Company for the periods referenced therein.
| 5.8 | All accounting documents and all other records required to be kept under any applicable law and any other books, files and records of the Company are up to date and have been and are properly, accurately and completely kept. The retention periods required under any applicable law, including without limitation any commercial law and Tax law, in relation to the Company have been fully complied with |
| 5.9 | Since the Balance Sheet Date: |
| 5.9.1 | there has been no material adverse change in the Company's financial position, prospects, turnover or profits; |
| 5.9.2 | the Company's Business has been carried on in the normal course without any interruption or alteration in its nature, scope or manner, and so as to maintain the Business as a going concern; |
| 5.9.3 | the Company has not entered into any transaction, contract or arrangement, or assumed or incurred any material liabilities (including contingent liabilities) or made any payment otherwise than in the ordinary course of carrying on its Business except as set forth in Exhibit 5.9.3; |
| 5.9.4 | the Company has not entered into any unusual or onerous commitments or contracts; |
| 5.9.5 | the Company's business has not been materially and adversely affected by the loss of any important customer or source of supply, and to the best knowledge of the Seller there are no facts which are likely to give rise to any such effects; |
| 5.9.6 | no dividend or other distribution has been declared, made or paid to the Company's shareholders except as provided for in the Financial Statements 2012 and except in accordance with Clause 8.2.1; |
| 5.9.7 | no share or loan capital has been allotted or issued or transferred or agreed to be allotted, issued or transferred; |
| 5.9.8 | the Company has not borrowed any money, or entered into any loan, debt or similar arrangements; |
| 5.9.9 | the Company has not made or received any surrender relating to group relief or the benefit of advance corporation tax; |
18
| 5.9.10 | the Company has not made, or agreed to make, any capital expenditure exceeding EUR 1,000,000 (Euro one million) in aggregate, or incurred, or agreed to incur, any commitments involving capital expenditure exceeding EUR 1,000,000 (Euro one million) in aggregate; and |
| 5.9.11 | no resolution of the shareholders or any class thereof has been passed (other than any resolution constituting ordinary business passed at an annual general meeting, details of which have been provided in the Disclosed Documents). |
| 5.10 | The Company has no bank or deposit accounts, howsoever described, save as set out in Exhibit 5.10. |
| 5.11 | The audited consolidated balance sheet of the Financial Statements and the related audited consolidated statement of operations, other comprehensive (loss) income, stockholders’ capital deficiency and cash flows (and the notes thereto) were prepared in accordance with the books and records of the Company and with Austrian generally accepted accounting principles, consistently applied during the applicable period and present fairly in all material respects the consolidated financial position of the Company and the Subsidiary, the consolidated results of their operations and cash flows for the applicable period. |
| 5.12 | Taxes. |
| 5.12.1 | All Tax Returns of or with respect to any Taxes that are required to be filed by or with respect to the Company have been duly and timely filed in accordance with all applicable laws. All items required to be included in each such Tax Return have been so included and all information provided in each such Tax Return was and is true, correct and complete. |
| 5.12.2 | All Taxes that have become due with respect to the period covered by each such Tax Return whether or not reflected on the Tax Return have been timely paid in full by the Company. Further, the Company has accrued for, and made deductions and retentions in respect of, Tax as required under applicable Tax laws and, in particular, has timely discharged all liabilities in respect to Tax that have become due on or prior to Closing in accordance with all applicable laws. |
| 5.12.3 | The amount of the provisions for Tax included in the Financial Statements is adequate and fully in accordance with applicable accounting practice. Proper provision has (where applicable) been made for deferred Tax in accordance with generally accepted accounting principles. |
| 5.12.4 | As of the Signing Date, no claim, assessment, deficiency or adjustment has been asserted against the Company with respect to any Tax Return, and there are no Tax related investigations, audits (except for the Tax audit ongoing at Signing), actions, proceedings, investigations, claims or assessments pending, proposed or threatening against or with respect to |
19
the Company imposed or initiated by a Tax Authority. In case any such action has been imposed, initiated or threatened by a Tax Authority between Signing and Closing, as of the Closing Date, the Seller has duly notified the Purchasers of any such event without undue delay upon occurrence.
| 5.12.5 | The Company has maintained all records, invoices and other documents required to be maintained for Tax purposes, including but not limited to comprehensive transfer pricing documentations which are in compliance with the Austrian Transfer Pricing Guidelines, and it has fulfilled all disclosure, notification and accounting obligations. |
| 5.12.6 | No Encumbrances have been imposed upon or asserted against the Company as a result of or in connection with the failure or alleged failure to pay any Tax. |
| 5.12.7 | The Company has not received any Tax ruling or entered into any written and legally binding agreement or is currently under negotiations to enter into any such agreement with any Tax authority which would affect the Tax situation of the Company in any period ending after the Closing Date. The Company is not subject to any Tax group or Tax planning scheme. |
| 5.12.8 | The Company has complied with all Tax obligations under applicable laws, in particular with intra-group transfer pricing rules. There has been and will be no transaction involving the Seller up to and including the Closing Date which qualifies as a constructive dividend for Tax purposes (verdeckte Gewinnausschüttung). |
| 5.12.9 | The Company has not been engaged in any scheme, arrangement or other practice whatsoever designed to reduce Taxes which could be disputed by any Tax Authority or could result in any additional Taxation being levied against the Company or could render the Company liable to pay any interest, penalty or fine relating to the Taxation. |
| 5.12.10 | The Company is resident for Tax purposes in Austria and has never been resident for Tax purposes in any other jurisdiction. Exhibit 5.12.10 contains a complete and accurate list of all jurisdictions where the Company is carrying on a trade, business or activity that creates a taxable presence of the Company in that jurisdiction. With the exception of those jurisdictions referred to in Exhibit 5.12.10 and Austria, the Company has never paid Tax on income, profit or gains to any other Tax authority in any other jurisdiction. |
| 5.12.11 | The sale and transfer of the Company's entire share in Orphanidis Pharma Research GmbH with corporate seat in Vienna and business address at Wilheminenstraße 91/II f, 1160 Vienna, Austria, registered in the companies’ register of the Vienna Commercial Court under registration number |
20
FN 257583 z, to a Third Party will not result in any Tax liability of the Company or the Purchasers.
| 5.13 | Title to and Sufficiency of Assets, Encumbrances. |
| 5.13.1 | The Company has good and marketable title to the assets reflected as being owned by it in the Financial Statements. Except as stated in the Financial Statements, all assets owned by the Company are free and clear of any Encumbrances. |
| 5.13.2 | The assets owned by the Company together with all Intellectual Property Rights owned or used by the Company and assets held by the Company under hire, purchase, leasing and rental agreements comprise all the assets necessary for the continuation of the Business as now carried on. |
| 5.14 | Intellectual Property Rights. |
| 5.14.1 | Exhibit 5.14.1 contains a complete and correct list of all Patents, Trademarks (including service marks and trade dress) and Domain Names owned and used by the Company. The Intellectual Property Rights owned or used by the Company are free from all Encumbrances and are valid and enforceable. |
| 5.14.2 | The Intellectual Property Rights owned or used by the Company have not been challenged by Third Parties and no such challenge is threatening and no circumstances exist or are threatening which may lead to a withdrawal or cancellation of any of the Intellectual Property Rights owned or used by the Company, or which may otherwise adversely affect the Intellectual Property Rights owned or used by the Company. Furthermore, the Intellectual Property Rights owned or used by the Company are to the Seller's best knowledge not subject to any pending proceedings for opposition, cancellation, revocation or rectification and no such proceedings have been threatened. No Intellectual Property Rights owned or used by the Company will expire, terminate or be terminated as a result of the transactions contemplated by this Agreement. All fees necessary to maintain the Intellectual Property Rights owned or used by the Company have been paid as and when due, all necessary renewal applications have been filed. |
| 5.14.3 | The Company has valid licenses to use all patents, trademarks, software and other Intellectual Property Rights which are (i) used by the Company in order to conduct its Business and (ii) which are not owned by the Company (the "Third Party IP Rights"). |
| 5.14.4 | To the Seller's best knowledge, the utilization or sale of any and all of the assets and properties of the Company or the utilization of any Intellectual Property Right do not infringe any Intellectual Property Right of any Third Party and the Company has not received notice of any such infringement. |
21
| 5.14.5 | Neither the Seller nor any current or former employee, freelancer, consultant, adviser or (other) independent contractor of the Company, or any other party, has or, originating during the period before the Closing Date, will have any claim against the Company or Purchaser(s) in connection with the present use of the Intellectual Property Rights owned by the Company. |
| 5.14.6 | The Company has taken reasonable security measures to protect the secrecy, confidentiality and value of all Intellectual Property Rights owned or used by the Company, research, development and production processes and all trade secrets used in the operation of its Business. The Company has not disclosed any of the information or data contained in dossiers filed with any relevant regulatory authority in relation to the Marketing Authorisations to any Third Party, except to the competent authorities or bodies responsible for the regulation of dealings in medicinal products, nor has it allowed any Third Party to cross-refer to any data or information contained in the Marketing Authorisations. |
| 5.14.7 | The Company has (A) not collected any personally identifiable information from any Third Parties or, if it has, (B) has complied with all applicable laws and regulations relating to the collection, storage and onward transfer of all personally identifiable information collected by the Company or by Third Parties having authorized access to the Company’s or other records. |
| 5.14.8 | The computer equipment and the computer software programs used by the Company in the conduct of its Business are its unencumbered property or lawfully licensed, and are fit and sufficient for the purposes for which they are being used and provide sufficient processing and storage capacity. |
| 5.15 | Real Property and Premises. |
| 5.15.1 | The Company does not own any real property, buildings on Third Party land (Superädifikate) or building rights. |
| 5.15.2 | Exhibit 5.15.2 contains a complete and correct list of all the real estate leased by the Company. These lease agreements are in full force and effect. None of them have been terminated and/or can be terminated as a consequence or result of the consummation of the transaction contemplated by this Agreement. |
| 5.16 | Contractual. |
| 5.16.1 | There is no Third Party (or Third Party together with other Third Parties connected with it) on whom the Company is substantially dependent or the cessation of transactions with whom would substantially affect the Business of the Company. |
22
| 5.16.2 | The Company has not entered into and are not party to any consortium, cooperation, joint venture, partnership, framework or franchise agreement or similar. |
| 5.16.3 | The Company is not party to any agreement or arrangement which restricts its freedom to carry on Business in any part of the world in such manner as it thinks fit. |
| 5.16.4 | No distributor of the Company is or, under a contract, agreement or arrangement currently in force, will be entitled to compensation under the Austrian Commercial Agency Act (Handelsvertretergesetz). |
| 5.16.5 | The Company is not party or subject to or bound by any Material Contract: |
| 5.16.5.1 | not on an arm’s length basis in the ordinary and usual course of business; |
| 5.16.5.2 | containing covenants directly, explicitly or indirectly limiting in any respect the freedom of the Company or to compete in any line of business or with any Person or entity; |
| 5.16.5.3 | relating to pensions, profit sharing, retirement or stock option plans; or |
| 5.16.5.4 | relating to any royalty, dividend or similar arrangement based on the revenues or profits of the Company, except – with respect to royalties only – in the ordinary course of business. |
| 5.16.6 | Exhibit 5.16.6 sets forth a complete and correct list of all Material Contracts to which the Company is party. (A) Each Material Contract is in full force and effect at arm's length terms, is enforceable against the parties thereto in accordance with its terms and conditions and has not been terminated, (B) the Company is not in default under or in breach of any Material Contract which cannot be reasonably remedied or which entitles the other party/-ies to termination of that Material Contract, (C) no counter-party to any Material Contract has notified the Company or the Seller that it intends to terminate or materially or adversely change the (terms of) a Material Contract, and (D) no counter-party to a Material Contract will be entitled to terminate the respective Material Contract as a consequence of the signing of this Agreement or the consummation of the transaction contemplated hereby. |
| 5.17 | All transactions and agreements between (i) the Company and the Seller; and/or (ii) the Company and Third Parties during the three years prior to Closing have been concluded at arms' length. All payments and other consideration (A) made by the Company and (B) received by the Company have been determined and paid at arms' length. |
23
| 5.18 | There are no loans, leases or other agreements or transactions between the Company and any present or former shareholder, director, officer or employee of the Company, or any member of such officer's, director's, employee's or shareholder's immediate family, or any person or entity controlled by such officer, director, employee or shareholder or his or her immediate family. |
| 5.19 | No shareholder resolution on the distribution of profits or on the dissolution of re-serves (capital or profit reserves) has been passed within the last three years in relation to the Company other than as disclosed in the Disclosed Documents. Any undistributed profits carried forward from previous business years of the Company, including profits, if any, for the business year that ended on 31 December 2012 and attaching to the Shares (except for the dividend pursuant to Clause 8.2.1), shall be for the account of the Purchaser. |
| 5.20 | Competing Businesses. |
Neither the Seller nor any of the (other) management board members of the Company has any material right or interest, direct or indirect, in any business which is competitive with any present or proposed Business of the Company.
| 5.21 | Loans. |
| 5.21.1 | there are not outstanding any loans made by the Company to or for the Seller, director, former director or shadow director of the Company or any person, entity or trust connected with any of them; |
| 5.21.2 | there are not outstanding any debts owing to the Company by the Seller, director, former director or shadow director of the Company or any person connected with any of them; or |
| 5.21.3 | there are not outstanding any debts owing by the Company which arose outside the ordinary course of business. |
| 5.22 | Employment. |
| 5.22.1 | Exhibit 5.22.1 contains a correct and complete list of all employees with valid employment agreements with the Company at Signing and of all individuals supplying services under supply agreements with the Company at Signing, showing date of entry, seniority/position and remuneration. |
| 5.22.2 | The Company is in material compliance with all applicable laws pertaining to its employees, including, but not limited to, all laws relating to employment standards, human rights, pay equity, labour relations, occupational health and safety, privacy, workers' compensation, pensions, benefits, Taxes and contributions to MVK (Mitarbeitervorsorgekasse) as well as social security contributions. |
24
| 5.22.3 | All employees of the Company are employed on contracts without unusual or unduly onerous obligations for the employer under such terms and conditions of employment. No payment or benefit has been made or promised to be made to any employee of the Company in connection with an actual or proposed termination of his or her employment or a termination in the context, or as a consequence, of the transactions contemplated hereby other than pursuant to applicable mandatory law. |
| 5.22.4 | The contracts of the three management board members (including pension and severance pay arrangements, if any) of the Company as attached hereto as Exhibit 5.22.4 are the only agreements in force between the Company and the management board members as of Signing. These management agreements contain the entire agreements between the Company and the respective counterparty, are complete in all respects and no other or deviating or supplemental agreements, written or oral, exist or will be concluded through Closing between the Company and any management board member. |
| 5.22.5 | Other than any statutorily applicable collective bargaining agreements (Kollektivverträge), there is no collective labour agreement to or by which the Company is or was a party or otherwise bound (apart from internal guidelines and policies). |
| 5.22.6 | The Company has not concluded and is not party to any employment agreement or management board member agreement providing for a statutory or voluntary severance entitlement (Abfertigung oder Abfindung) in excess of (i) the mandatory statutory severance entitlements of the respective employee, or (ii) the mandatory severance entitlements pursuant to the collective bargaining agreement statutorily applicable to the respective employee. |
| 5.22.7 | As far as the Seller is aware, there are no labour disruptions pending or threatened against the Company. |
| 5.22.8 | All payments in relation to employment, whether standard remuneration, bonuses, premiums or payments of any other nature, including social security contributions and Taxes under any applicable law, due and payable by the Company have been made as and when due. Except as required under statutorily applicable collective bargaining agreements (Kollektivverträge) or in the ordinary course of the Company's Business, no increases of payments in relation to employment, whether standard remuneration, bonuses, premiums or compensation of any other nature have been made or agreed by the Company between the Balance Sheet Date and the Signing Date and no such increases have been or will be made, promised or committed to by or on behalf of the Company through Closing. |
25
| 5.22.9 | Without prejudice to Clause 5.7, the Company has created sufficient pro-visions in accordance with applicable law and Austrian generally accepted accounting principles for (A) any employee entitlements, including, without limitation, severance pay entitlements (Abfertigungsrückstellungen), unconsumed vacation and jubilee entitlements; (B) obligations of the Company under early retirement schemes (Vorruhestandsverträge) and/or early part-retirement schemes (Altersteilzeitverträge); and (C) all other employee-related costs, including, without limitation, bonuses. |
| 5.22.10 | True and accurate details of all bonus plans, bonus schemes and bonus principles in place at and/or applying to the Company are disclosed in Exhibit 5.22.10. There are no other bonus plans, bonus schemes, bonus principles or stock options or share incentive schemes in place, applying to or having applied to the Company. |
| 5.22.11 | There is no works council (Betriebsrat) or other employee representatives at the Company. |
| 5.22.12 | No freelancer (freier Mitarbeiter) or consultant (Berater) of the Company could, under any applicable law, claim to be, or may be considered by any competent authority to be an employee of the Company. |
| 5.22.13 | All employees newly employed with the Company since 2006 and currently employed with the Company have executed employment agreements that oblige such current and/or former employees to protect the Company's confidential information. As to Intellectual Property Rights, such employees have executed agreements assigning such rights to the Company. |
| 5.23 | Pensions, Sickness, Work Safety. |
| 5.23.1 | The Company has no pension or sickness fund and to the Seller's best knowledge, has complied with all applicable rules and regulations relating to work safety and occupational health in all material respects. During the course of their employment, to the Seller's best knowledge the employees of the Company have not been and are not exposed to anything which may have contributed or may contribute to the occurrence of a disability or latent occupational diseases. |
| 5.23.2 | The Company has not incurred and will not incur any costs or liability in respect of any pension liability arising out of employment or relating to any events before the Closing Date, except as (fully) accrued in the Financial Statements 2012. |
| 5.24 | Litigation; Defects of Products. |
| 5.24.1 | The Company is not involved, whether as defendant, claimant or otherwise, in any litigation, arbitration, mediation or other dispute resolution mechanism or as a party in any administrative proceedings and to the |
26
Seller's best knowledge, no such proceedings are threatening by or against the Company. The Company has fully complied with and has performed all of its material obligations under any judgements, settlements, decrees, decision of or agreements with any authority, court or arbitration panel. No governmental, administrative, regulatory or other official investigation or inquiry concerning the Company is in progress or pending and there are no circumstances that may lead to any such investigation or inquiry, excluding Tax audits.
| 5.24.2 | The Company has not manufactured, imported, sold or supplied products which are or were, or will become, in any material respect faulty or defective or a danger to a purchaser, a consumer, the public or any section of the public at large, or which, as far as the Seller is aware, do not comply in any material respect with any warranties or representations expressly or impliedly made (whether pursuant to statute or otherwise) by the Company, or with all applicable laws, regulations, standards and requirements. There are no outstanding claims against the Company in respect of |
| 5.24.2.1 | defects in any of the Products or any part of the packaging or labelling of any of the Products; |
| 5.24.2.2 | delays in delivery of any Products to customers of the Company; or |
| 5.24.2.3 | deficiencies in the safety or efficacy of any Products or otherwise relating to liability for goods or services supplied or to be supplied by the Company, |
and to the Seller's best knowledge no such claims are likely or threatened.
| 5.25 | Programs. |
| 5.25.1 | The Company holds all material regulatory and administrative permits, approvals, consents, Licenses, authorisations and similar instruments legally required to conduct the Programs. |
| 5.25.2 | The clinical trials conducted by, or on behalf of, the Company in relation to the Programs were and, if still pending, are being conducted in accordance with all applicable regulatory requirements and no breaches of any applicable regulatory requirements have occurred and there are no unresolved safety issues with respect to these Programs, except as disclosed in Exhibit 5.25.2. |
| 5.25.3 | The clinical trials undertaken and being undertaken in connection with the Programs have resulted in no fatalities or serious illnesses to any individuals participating in such clinical trials, except as disclosed in Exhibit 5.25.2. |
27
| 5.25.4 | The Seller has provided the Purchasers with full details of the Programs, including all studies, tests and trials relating to the Programs, and has no knowledge of any adverse studies, tests or trials relating to any of the Programs, except for customary amendment of protocols implemented from time to time. |
| 5.25.5 | The Company has not received any notice or correspondence from any governmental agency requiring the termination or suspension of any clinical trials conducted by, or on behalf of, the Company, or after the commencement of any clinical trial, any modification of such clinical trial conducted by, or on behalf of, the Company. |
| 5.25.6 | To the Seller's best knowledge, there are no outstanding or threatened claims against the Company in respect of any matters arising in connection with the Programs, including without limitation from the clinical trials. |
| 5.25.7 | No third party has terminated or threatened to terminate any contract, licence or other arrangement relating to a Program and entered into with the Company, or the Subsidiary. |
| 5.26 | The Company has adequate insurance coverage, customary for the scope of its Business, including where such coverage is required in order to comply with any legislative, regulatory, contractual or other obligations. |
| 5.27 | Legal Compliance and Regulatory. |
| 5.27.1 | To the Seller's best knowledge, the Company and the Subsidiary have carried on the Business in all material respects in compliance with applicable laws, regulations, Licenses and by-laws in Austria and all other countries, and to the Seller's best knowledge there is no investigation or enquiry by, or order, decree or judgment of, any court, governmental agency or regulatory body outstanding against the Company or the Subsidiary. To the Seller's best knowledge, the Company has in all material respects complied with all conditions, orders and requirements imposed by any authority. |
| 5.27.2 | The Company and the Subsidiary hold all material regulatory and administrative permits, approvals, consents, licenses and similar instruments (including without limitation all Marketing Authorisations (and applications) ("Licenses") legally required to (be qualified to) conduct their Business as presently conducted, for the ownership of its assets, Intellectual Property Rights and properties and the commercialisation of the Products. All Licenses are in full force and effect, subject to their terms. The Seller is not aware that any of the Licenses might be amended, challenged, revoked or not renewed or that further conditions as to the Business of the Company as currently conducted might be imposed. |
28
| 5.27.3 | Exhibit 5.27.3 contains a correct and complete list of all Marketing Authorisations material to the Business as currently conducted. |
| 5.27.4 | The Company has timely filed all material reports, registrations, statements and certifications that it was required to file with any competent governmental authority and with respect to which a failure by the Company to timely file would have a material impact on the Business. |
| 5.27.5 | The Company is not subject to any cease-and-desist or other order or enforcement action issued by, or is a party to any written agreement or understanding with, any governmental authority that currently restricts or affects in any material respect the conduct of its Business (or that, as a result of completion of this Agreement, would restrict in any material respect the conduct of the business of the Purchasers' group). |
| 5.27.6 | To the Seller's best knowledge, there are no and there have been no actions or omissions of the Seller or the Company that may lead to any material liabilities, penalties, fines, damages or any other loss of the Company under any laws that would not have incurred if the business activities of the Company had been lawfully conducted. |
| 5.27.7 | The Company has not manufactured or procured the manufacture of, sold or supplied in the course of the Business any Product which is or was or, to the Seller's best knowledge, will become defective or dangerous. |
| 5.27.8 | There have been: |
| 5.27.8.1 | no decisions by any governmental or regulatory authority indicating that any of the Products is in any way defective, unsafe for normal use, or fails to meet applicable standards required by such governmental or regulatory authorities; |
| 5.27.8.2 | no material outstanding unresolved safety issues with respect to the Products; and |
| 5.27.8.3 | no recalls initiated by the Company or ordered by any governmental or regulatory body with respect to the Products within 4 (four) years prior to the Closing Date except with respect to Busulvex; |
and, to the Seller's best knowledge, no such circumstances are likely or threatened.
| 5.27.9 | Each of the management board members of the Company who are required to be registered, licensed or qualified with any governmental authority are, in each case to the extent required, duly registered, licensed and qualified for that purpose, and such registrations, licenses and qualifications are in full force and effect. None of the management board members |
29
of the Company has been subject to any order or decree or judgment disqualifying him from holding the office of director or any other position within the Company.
| 5.27.10 | None of the management board members of the Company has been convicted of any criminal offence or been subject to any criminal proceedings. |
| 5.27.11 | To the Seller's best knowledge, no payment prohibited by law has been made, directly or indirectly, by or on behalf of the Company to any business relations such as customers or suppliers, any public official or any domestic or foreign government, a political party or its officers, or any candidate for public office and has in all material respects complied with all applicable laws and regulations, including, without limitation, those implementing international conventions, relating to improper payments and has in every instance acted consistently with principles of good conduct and business ethics. |
| 5.28 | The Seller has not given any guarantees, suretyships or any other form of comfort or support in favour of the Company or any of the Company's obligations. The Company is not a party to and has not entered into any contracts between business enterprises as defined in § 238 AktG, nor granted or issued any comfort letters, suretyships, guarantees or any other liabilities or rights (including, without limitation, rights in rem) in favour of other Person's and/or entities' obligations, including, without limitation, obligations of the Seller. |
| 5.29 | The Company has not received, applied for or used any public grants (Zuschüsse), allowances (Zulagen), aids, public support or other subsidies (Subventionen) in whatever form, other than those disclosed to the Purchasers in writing prior to Signing such as for Moli in 2008 and the fact that the Company has received research premium (Forschungsprämie). |
| 5.30 | No broker, agent, adviser or other person acting on behalf of the Seller is or will be entitled to any commission or fee from Purchaser or the Company in connection with any of the transactions contemplated by this Agreement |
| 5.31 | The Company has not given a power of attorney or any other authority (express, implied or ostensible) which is still outstanding or effective to any Person to enter into any contract or commitment or do anything on its behalf, other than any authority to employees to enter into routine trading contracts in the normal course of their duties. |
| 5.32 | The Company has not sold or otherwise disposed of any shares or assets in circumstances such that it is or may be still subject to any liability (whether contingent or otherwise) under any representation, warranty or indemnity given or agreed to be given on or in connection with such sale or disposal. |
30
| 5.33 | Subsidiary and Representative Offices. |
| 5.33.1 | The Company is the direct sole and unrestricted legal and beneficial owner of the Subsidiary Shares and the Subsidiary Shares are fully paid up, free and clear of all Encumbrances. The nominal share capital of the Subsidiary has not been decreased or redeemed; there has not been any unlawful repayment of share capital (Einlagenrückgewähr). |
| 5.33.2 | The Subsidiary is a private company limited by shares, duly incorporated and organized and validly existing under the laws of England and Wales. |
| 5.33.3 | The Subsidiary is not over-indebted, insolvent, subject to a reorganization or comparable procedure, nor is such insolvency, over-indebtedness or procedure threatening to the Subsidiary. |
| 5.33.4 | The Subsidiary does not hold any shares or any other participations or interest in any other company, partnership, business organization or other entity. The Subsidiary does not control the management of any other company, partnership, entity or business organization, nor does it participate in such a control. |
| 5.33.5 | There are no outstanding liabilities between the Seller and any Subsidiary. |
| 5.33.6 | Attached hereto as Exhibit 5.33.6 is a correct and complete list of all representative offices and area managers of the Company. |
| 5.34 | All Disclosed Documents are listed in Exhibit 5.34; in addition, Exhibit 5.34 contains printouts of screenshots listing the Disclosed Documents contained on the USB sticks delivered to the Purchasers. All information disclosed to Purchasers and/or their advisers in the course of their due diligence or otherwise, including in the Exhibits to this Clause 5 and the Disclosed Documents and all responses provided to questions of the Purchasers and/or their advisers, including, without limitation, information disclosed with respect to assets, liabilities, rights and obligations of the Company as well as the circumstances referring to the representations and warranties is in all material respects correct, complete and not misleading. No circumstances are known to the Seller that were not disclosed to the Purchasers and/or their advisors in writing prior to Signing and that could make the information disclosed incorrect, incomplete or misleading in a material respect. |
| 6 | Representations and Warranties of the Purchasers |
| 6.1 | Each Purchaser, unless otherwise expressly stated in this Clause 6, individually (and not on a joint and several basis) represents and warrants that the representations and warranties as set out in this Clause 6 are correct, complete and not misleading, unless otherwise expressly stated in this Clause 6, as of Signing and Closing. |
31
| 6.2 | Each Purchaser has full power and authority to purchase the Shares and to execute and deliver this Agreement and consummate the transactions contemplated thereby including all necessary corporate approvals (except for the shareholder approval pursuant to Clause 9.1.2). Each Purchaser has been duly incorporated and organized and validly exists under the laws of its respective jurisdiction of incorporation. Each Purchaser has the power and the necessary licenses and approvals to carry on its business as currently being conducted. |
| 6.3 | Subject to Clause 9.1.2, (A) this Agreement has been duly executed by each Purchaser and constitutes a legal, valid and binding obligation of each Purchaser enforceable against each Purchaser in accordance with its terms; and (B) all Third Party consents and approvals, including without limitation, consents and approvals under applicable corporate law for the transfer of the Shares on the terms and conditions set out in this Agreement have been obtained. |
| 6.4 | Purchaser 2 represents and warrants that at Closing: |
| 6.4.1 | The Consideration Shares will be duly issued in accordance with Irish law and credited as fully paid. Each of the Consideration Shares will constitute one ordinary share of Purchaser 2, each having a nominal value of EUR 0.05, carrying one vote per share. The Consideration Shares will be admitted to trading on the Main Securities Market of the Irish Stock Exchange in Dublin on the Closing Date. |
| 6.4.2 | The Consideration Shares are not subject to any rights of any person and are freely transferable under the articles of association of Purchaser 2, the laws of the Republic of Ireland and, to the best knowledge of Purchaser 2, other applicable laws. The Consideration Shares will have ISIN IE0003072950 and will be fungible with the shares of Purchaser 2 currently traded on the New York Stock Exchange and the Main Securities Market of the Irish Stock Exchange in Dublin. No authorizations, approvals, consents or licenses from any governmental authority or regulatory body or agency, including, without limitation, any bank regulatory authority, body or agency, are required to be obtained by Purchaser 2 to effect dividend payments declared and payable on, or in respect of, the Consideration Shares. The Consideration Shares shall carry the right to receive dividends declared, made or paid after the Closing Date (save that they shall not rank for any dividend or other distribution of the Purchaser 2 declared made, or paid by reference to a record date before the Closing Date). |
| 6.5 | Claims for breach of any of the Purchasers' representations and warranties relating to Clause 6.2 through Clause 6.4 inclusive shall expire within 15 (fifteen) years from the Closing Date. |
32
| 6.6 | Notice of Breach. |
| 6.6.1 | If the Seller become aware of any matter or circumstance that may give rise to a Claim against the Purchasers under this Agreement, the Seller shall as soon as reasonably practicable but in any event within 3 (three) months of becoming so aware give notice in writing to the Purchaser, setting out such information readily available to the Seller as is reasonably necessary to enable the Purchasers to assess the merits of the Claim and to act to preserve evidence. |
| 6.6.2 | Failure to give notice within the period specified in Clause 6.6.1 shall not prejudice the Seller except to the extent that the Purchasers' liability is thereby increased, in which case the Purchasers shall not be liable for the amount of that increase. |
| 6.7 | Third Party Claims. |
| 6.7.1 | If a Claim notified pursuant to Clause 6.6.1 is a result of or in connection with a Third Party Claim, then: |
| 6.7.1.1 | subject to Clause 6.7.2 and subject to the Purchasers indemnifying the Seller against any and all Loss, the Seller shall take such action as the Purchasers may reasonably request, to avoid, dispute, deny, defend, resist, appeal, compromise or contest such claim; |
| 6.7.1.2 | the Seller may admit, compromise, dispose of or settle such claim if the Purchasers unreasonably delay making a request pursuant to Clause 6.7.1.1, provided that the Seller has notified the Purchasers of its intention to deal with such claim; |
| 6.7.1.3 | if the Purchasers make a request pursuant to Clause 6.7.1.1, subject to Clause 6.7.2, the Seller shall take all reasonable steps to ensure that the Purchasers are provided on reasonable notice with all material correspondence and documentation relating to the claim as the Purchasers may reasonably request, subject to the Purchasers agreeing in such form as the Seller may reasonably require to keep all such correspondence and information confidential and to use it only for the purpose of dealing with the relevant claim. |
| 6.7.2 | The Seller shall not be required to take any action or provide any information to the Purchasers which the Seller, in its absolute discretion, reasonably considers may be unduly onerous or materially prejudicial to it. |
| 6.8 | The Seller shall not be entitled to claim that any fact, matter or circumstance causes any of the Purchasers' representations or warranties pursuant to Clause 6.1 through Clause 6.4 inclusive to be breached or renders any of these |
33
representations or warranties misleading if it has been expressly and specifically disclosed in this Agreement or an Exhibit to Clause 6 of this Agreement and only matters so disclosed in this Agreement or an Exhibit to this Agreement shall qualify the Purchasers' representations and warranties pursuant to Clause 6.1 through Clause 6.4 inclusive, notwithstanding any investigations the Seller or its advisors have conducted in the course of due diligence, the contract negotiations and notwithstanding any information the Seller or its advisors have received or obtained otherwise.
| 7 | Recovery by the Purchasers |
| 7.1 | Breach of the Seller's representation and warranties. |
| 7.1.1 | If and to the extent that (i) any representation or warranty made by the Seller under this Agreement is incorrect, incomplete or misleading or (ii) the Seller violates, contravenes or breaches any covenant, agreement or obligation of the Seller under or pursuant to this Agreement, the Seller shall put the Purchaser(s), the Company or the Subsidiary, as the case may be, into the position they would have been in the absence of such breach (restitution in kind; Naturalrestitution). If the Seller is unable to achieve this position within 3 (three) months after having been notified by a Purchaser of the breach (Clause 7.4), the Purchasers shall be entitled to (i) claim payment of the amount of the Loss on a Euro for Euro basis pursuant to Clause 7.1.2 or (ii) in the case of a breach of representation or warranty (re-)establish the represented and/or warranted status at the cost and expense of the Seller. |
| 7.1.2 | If and to the extent the Purchasers claim payment of the amount of the Loss, any payment to be received by the Purchaser(s) hereunder shall be treated as a reduction of the Purchase Price and shall be settled primarily by way of deduction from and/or set-off against any unpaid part of the Purchase Price, in particular from or against the Contingent Purchase Price, if any. The Purchasers shall be entitled to set-off pursuant to this Clause 7.1.2 once a Claim has been (i) determined by an arbitral award rendered by an arbitral tribunal in accordance with Clause 14.2, such award to be final and enforceable, or (ii) settled by written agreement between the Parties. To the extent any Loss cannot be satisfied by set-off from or against the Contingent Purchase Price, if any, the Seller shall promptly reimburse the Purchasers in cash. If and to the extent a payment of an amount of a Loss is not treated as a reduction of the Purchase Price, the Seller shall indemnify the Purchasers from any adverse income Tax (Ertragssteuern) consequences arising out of our in connection with such reimbursement and/or VAT payable thereon, if any. |
| 7.1.3 | If and to the extent a defect (Mangel) is the result of an obligation or liability of the Company, which non-existence has been represented or warranted, Purchasers' claim pursuant to this Clause 7 shall be equal to an |
34
amount which (a) Purchaser(s), the Company or the Subsidiary have to pay in order to fully settle, discharge and satisfy this obligation or liability.
| 7.1.4 | The Parties agree and acknowledge that the Purchasers shall not be precluded from raising damage claims and/or pursuing any other legal remedies available under applicable laws, including without limitation, claims based on error (Irrtum), fraudulent misrepresentations, deceit or force (within the meaning of § 870 ABGB) out of or in connection with this Agreement; damages and any other claims shall be determined and calculated pursuant to applicable laws. The right to void (anfechten) or rescind (aufheben) this Agreement pursuant to § 934 ABGB (Verkürzung über die Hälfte) and the Purchasers' right to rescission (Wandlung) shall be excluded. |
| 7.1.5 | The Purchasers shall not be entitled to recover from the Seller under this Agreement more than once in respect of the same Loss suffered. |
| 7.2 | Financial Limitations. |
| 7.2.1 | In relation to claims for a breach of any of the Seller's representations and warranties, the Purchasers shall only be entitled to a Claim: |
| 7.2.1.1 | if the individual Claim exceeds an amount of EUR 200,000 (Euro two hundred thousand) (the "De Minimis Amount"); and |
| 7.2.1.2 | once the aggregate amount of all Loss(es) which in the individual case exceed the De Minimis Amount exceeds EUR 2,000,000 (Euro two million) (the "Threshold"). If the Threshold is exceeded, the Purchasers shall be entitled to claim the full amount and not only the excess above the Threshold. |
The De Minimis Amount and the Threshold shall not apply with respect to Claims of the Purchasers relating to the representations and warranties set forth in Clauses 5.2, 5.3 and/or Clause 5.12 (Taxes).
| 7.2.2 | The aggregate liability of the Seller for Claims relating to a breach of the Seller's representations and warranties shall not exceed 40 % (forty percent) of the Base Purchase Price, except for Claims relating to Clauses 5.2 and/or 5.3, where the Seller's maximum liability shall not exceed an amount equal to 100 % (one hundred percent) of the Base Purchase Price. The aggregate liability of the Seller for Claims relating to a breach of the Seller's representations and warranties shall in no event exceed an amount equal to 100 % (one hundred percent) of the Base Purchase Price. |
35
| 7.3 | Time Limitations. |
| 7.3.1 | The statute of limitations for all Claims for breach of any of Seller's representations and warranties, except for the Seller's representations and warranties relating to (i) Organisation, Power and Authority and Shares and Share Capital (Clauses 5.2 and 5.3), (ii) Clause 5.12 (Taxes), (iii) Intellectual Property Rights (Clause 5.14) and (iv) claims based on error (Irrtum), fraudulent misrepresentation, deceit or force within the meaning of § 870 ABGB shall expire within 12 (twelve) months from the Closing Date. |
| 7.3.2 | The statute of limitations for all Claims for breach of any of Seller's representations and warranties relating to Clause 5.12 (Taxes) shall not expire before 6 (six) months after all Taxes have been finally charged and assessed for periods until the Closing Date inclusive and, if a Tax audit has been conducted for the period up to the Closing Date inclusive, not before 6 (six) months after the legally binding completion (Rechtskraft) of the Tax audit. For the purposes of this Clause 7.3.2, a legally binding completion (Rechtskraft) of a Tax audit will only be given when all proceedings pending at the Constitutional Court (Verfassungsgerichtshof) and/or the Administrative Court (Verwaltungsgerichtshof) have also been completed. |
| 7.3.3 | Claims for breach of any of Seller's representations and warranties relating to Intellectual Property Rights (Clause 5.14) shall expire within 36 (thirty six) months from the Closing Date. |
| 7.3.4 | Claims for breach of any of Seller's representations and warranties relating to Organisation, Power and Authority and Shares and Share Capital (Clauses 5.2 and 5.3) shall expire within 15 (fifteen) years from the Closing Date. |
| 7.3.5 | The statute of limitations for Purchasers' claims based on error (Irrtum), fraudulent misrepresentation, deceit or force within the meaning of § 870 ABGB, shall expire within 7 (seven) years from the Closing Date. |
| 7.4 | Notice of Breach. |
| 7.4.1 | If the Purchasers become aware of any matter or circumstance that may give rise to a Claim against the Seller under this Agreement, the Purchasers shall as soon as reasonably practicable but in any event within 3 (three) months of becoming so aware give notice in writing to the Seller, setting out such information readily available to the Purchasers as is reasonably necessary to enable the Seller to assess the merits of the claim and to act to preserve evidence. A notice given by only one Purchaser in connection with this Agreement shall be deemed given on behalf of both Purchasers, unless indicated otherwise in such notice. |
| 7.4.2 | Failure to give notice within the period specified in Clause 7.4.1 shall not prejudice the Purchasers except to the extent that the Seller's liability is |
36
thereby increased, in which case the Seller shall not be liable for the amount of that increase.
| 7.5 | Third Party Claims. |
| 7.5.1 | If a claim notified pursuant to Clause 7.4.1 is a result of or in connection with a claim (including, without limitation, any claim for breach of any of the Seller's representations and warranties relating to Taxes within the meaning of Clause 5.12) by or liability to a third party, including, without limitation, any (Tax) Authority, in respect of which indemnification may be sought pursuant to this Agreement (a "Third Party Claim"), then: |
| 7.5.1.1 | subject to Clause 7.5.2 and subject to the Seller indemnifying the Purchasers against any and all Loss, the Purchasers shall, or Purchasers shall procure that the Company, or the Subsidiary, shall, take such action as the Seller may reasonably request, to avoid, dispute, deny, defend, resist, appeal, compromise or contest such Third Party Claim; |
| 7.5.1.2 | the Purchasers may admit, compromise, dispose of or settle such Third Party Claim if the Seller unreasonably delays making a request pursuant to Clause 7.5.1.1, provided that the Purchasers have notified the Seller of their intention to deal with such Third Party Claim; |
| 7.5.1.3 | if the Seller makes a request pursuant to Clause 7.5.1.1, subject to Clause 7.5.2, the Purchasers shall, and the Purchasers shall procure that the Company, or the Subsidiary, shall, take all reasonable steps to ensure that the Seller is provided on reasonable notice with all material correspondence and documentation relating to the Third Party Claim as the Seller may reasonably request, subject to the Seller agreeing in such form as the Purchasers may reasonably require to keep all such correspondence and information confidential and to use it only for the purpose of dealing with the relevant Third Party Claim. |
| 7.5.2 | The Purchasers shall not be required to take any action or provide any information to a Seller which a Purchaser, in its absolute discretion, reasonably considers may be unduly onerous or materially prejudicial to it, the Company or the Subsidiary or to its, the Company's, the Subsidiary's or Purchaser 2's group's businesses. |
| 7.6 | The Purchasers shall not be entitled to claim that any fact, matter or circumstance causes any of the Seller's representations or warranties pursuant to Clause 5 to be breached or renders any of these representations or warranties misleading if it has been fairly and accurately disclosed in the Disclosed Documents, this Agreement |
37
or an Exhibit to Clause 5 of this Agreement in such manner to enable the Purchaser to identify the scope and nature of such liability and to make a reasonable assessment of the impact of such liability and only matters so disclosed in the Disclosed Documents, this Agreement or an Exhibit to Clause 5 of this Agreement shall qualify the Seller's representations and warranties pursuant to Clause 5, notwithstanding any investigations the Purchasers or their advisors have conducted in the course of a due diligence, the contract negotiations and notwithstanding any information the Purchasers or their advisors have received or obtained otherwise.
| 7.7 | The restrictions and limitations of the Seller's liability pursuant to Clause 7.6 shall not apply to Third Party Claims made as a result of a breach of any of the representations and warranties relating to Clause 5.12 (Taxes). |
| 7.8 | The Seller shall not be liable for a Claim if and to the extent: |
| 7.8.1 | specific provisions (Rückstellungen) were made for the matter or circumstance(s) in the Audited Financial Statements 2012; |
| 7.8.2 | the matter has been approved in writing in advance by the Purchasers during the Interim Period pursuant to Clause 8.1; |
| 7.8.3 | such Claim arises or is increased (but in the latter case only to the extent of the amount of the increase) (i) as a result of any change after the Closing Date in any legislation or Tax rate which (was) is not in force before and/or at the Closing Date; or |
| 7.8.4 | there is an immediate (i.e. within the same financial year) relief from Tax for the Purchasers, the Company or the Subsidiary, as the case may be. |
| 8 | Conduct of Business during the Interim Period. Covenants of the Seller |
| 8.1 | The Seller hereby covenants that during the Interim Period he shall, except in the ordinary course of the Business as currently conducted or with the prior written consent of the Purchasers, such consent not to be unreasonably or arbitrarily withheld or delayed: |
| 8.1.1 | cause the Company and Subsidiary not to terminate, amend, enter into or agree to enter into any material contract, or assume or incur any liabilities or make any payment or waive or release any rights of value, or modify any agreement or enter into or agree to enter into any other agreement or commitment which is not capable of being terminated prior to 31 December 2013 and which provides for annual obligations of the Company and Subsidiary in excess of EUR 2,000,000 (Euro two million); |
| 8.1.2 | promptly notify the Purchasers in writing of the existence or occurrence of (i) any fact, event or condition which comes to his attention, which may |
38
materially alter the accuracy or completeness of any representation or warranty contained in this Agreement, or result in any such representation or warranty being incorrect or misleading; or (ii) any breach of any covenant, agreement or obligation of the Seller hereunder or of the occurrence of any event that may make the satisfaction of the Conditions Precedent impossible, impractical or unrealistic;
| 8.1.3 | cause the Company and Subsidiary to maintain in full force and effect, at the Company's and Subsidiary's cost, their existing insurance coverage; |
| 8.1.4 | cause the Company and Subsidiary not to sell, lease or alienate any of their assets except in the ordinary course of business, nor cause any Encumbrance to exist in respect thereof; |
| 8.1.5 | cause the Company and Subsidiary not to make any material change in the level of borrowing (except for the utilization of existing credit lines in line with past practice) and not to enter into any new loan, guarantee, surety or any other financing facilities; |
| 8.1.6 | cause the Company and Subsidiary not to enter into any factoring of accounts receivables; |
| 8.1.7 | from the Signing Date, except with the prior written consent of the Purchasers, cause the Company and Subsidiary not to modify the terms of employment, including salary, wages and fringe benefits, of any of their employees, not to hire any employee(s) nor terminate the services of any employees, unless in the ordinary course of business; |
| 8.1.8 | use all available reasonable means (alle zumutbar zur Verfügung stehenden Möglichkeiten ausschöpfen) to obtain each consent or agreement of each person necessary for the fulfilment of its obligations under this Agreement and the consummation of the transactions contemplated thereby; |
| 8.1.9 | cause the Company and Subsidiary to pay or withhold, as the case may be, in a timely manner all Taxes required to be paid by them after the Signing Date and on or before the Closing Date and duly and timely prepare and file, in a manner consistent with past practice and applicable laws, all Tax Returns required to be filed after the Signing Date and on or before the Closing Date; |
| 8.1.10 | not enter into discussions or negotiations with any person relating to the sale of any shares in the Company or Subsidiary or any of the Company's or Subsidiary's assets (as to assets other than in the ordinary course of business) or the issuance of any shares, or other securities of any nature, kind or description, of the Company or Subsidiary, nor enter into any discussions or negotiations with any person relating to a merger, spin-off, contribution, sale or other disposition of substantially the whole of the |
39
Company or the Subsidiary or the acquisition of or the obtaining of control over any other company or entity;
| 8.1.11 | not take any actions which would alter the nature or form of the Shares as shares issued to bearer (Inhaberaktien); and |
| 8.1.12 | cause the Company and Subsidiary to maintain adequate levels of working capital in accordance with past practice. |
| 8.2 | In addition, the Seller hereby covenants that during the Interim Period there has not been and will not be, without limitation: |
| 8.2.1 | any declaration or authorisation or payment, open or hidden, of any annual or interim dividend or other distribution (whether in cash, stock, or in kind) to the Seller, including but not limited to loans, considerations other than at arm's length terms and in the normal course of business, nor any reduction of the Company's Registered Share Capital, except as set forth in and subject to the terms and conditions of Exhibit 8.2.1; or |
| 8.2.2 | any appointment of the auditor (Abschlussprüfer) of the Company for any calendar year following the calendar year having ended on 31 December 2012, without the prior written consent of the Purchasers. |
| 8.3 | During the Interim Period, the Seller shall (to the extent permitted by applicable law) cause the Company to: |
| 8.3.1 | grant the Purchasers free and unrestricted access, upon reasonable notice and during customary business hours to the books and records of the Company, provided that the Purchasers do not unreasonably interfere with the operation of the Company's business; |
| 8.3.2 | furnish the Purchasers with such financial, operating and other data and information in respect of the Company as the Purchasers shall from time to time reasonably request, and |
| 8.3.3 | furnish the Purchasers with copies of all such contracts, books and records, documents, data and information as the Purchasers shall from time to time reasonably request. |
| 8.4 | Restrictive Covenants. |
| 8.4.1 | The Seller undertakes, except in continuation of his work for or service to the Company, not to directly or indirectly until the expiry of (A) 3 (three) years from the Closing Date; and (B) 1 (one) year from the Seller ceasing to work for, or provide services to, the Company: |
40
| 8.4.1.1 | be involved, as employee or as manager in any activity relating to the Business as currently operated by the Company or the Subsidiary; |
| 8.4.1.2 | solicit or accept any business relating to the Company's or the Subsidiary's Business from any of the Company's or the Subsidiary's present or prospective customers or induce any of its present or prospective customers or other business partners to terminate their business relationship with the Company or the Subsidiary; |
| 8.4.1.3 | in any way discredit in public, through the media or otherwise, the Company or the Subsidiary or any of the Company's or the Subsidiary’s staff or present or prospective customers or any of its business dealings in a way that could be expected to have a negative impact on the business of the Company; |
| 8.4.1.4 | solicit, endeavour to entice away from the Company or the Subsidiary or otherwise try to induce to leave the Company or the Subsidiary any person who is as at the date hereof employed or, to the knowledge of the Seller scheduled to be employed by the Company or the Subsidiary or is a member of a corporate body of or a regular advisor to the Company or the Subsidiary. |
| 8.4.2 | The Purchasers shall be entitled to set off Claims pursuant to this Clause 8.4 against the Contingent Purchase Price, if any. The Purchasers shall be entitled to set-off pursuant to this Clause 8.4.2 once a Claim has been (i) determined by an arbitral award rendered by an arbitral tribunal in accordance with Clause 14.2, such award to be final and enforceable, or (ii) settled by written agreement between the Parties. |
| 9 | Conditions Precedent |
| 9.1 | The obligations of the Seller and the Purchasers to consummate the sale and transfer of the Shares as contemplated by this Agreement are subject to the satisfaction of all of the following conditions precedent (aufschiebende Bedingungen; collectively the "Conditions Precedent") having been fulfilled on or before the Closing Date in accordance with this Agreement: |
| 9.1.1 | Absence of a Material Adverse Effect, respectively of events or circumstances that, individually or in the aggregate, could reasonably be expected to have a Material Adverse Effect; |
41
| 9.1.2 | the passing of an ordinary resolution approving the transactions contemplated by this Agreement by Purchaser 2's shareholders at a general meeting of Purchaser 2; |
| 9.1.3 | delivery to the Purchasers of the original of a written confirmation duly executed by the Seller that the representations and warranties as set out in Clause 5 are correct, complete and not misleading, unless otherwise expressly stated in Clause 5, as of Closing; |
| 9.1.4 | receipt by the Purchasers of written evidence that the following Third Parties have irrevocably and unconditionally consented to the consummation of the transactions contemplated by this Agreement or confirmed that no consent is required: United Therapeutics (Remodulin® (Treprostonil)), Ferrer Internacional S.A. (Adasuve®) and PharmaEssentia Corporation (Peg-IFN-a 2b); |
| 9.1.5 | delivery by the Seller and the Company to the Purchasers of the Audited Financial Statements 2012; and |
| 9.1.6 | preparation of financial statements of the Company as at 31 December 2012 prepared in accordance with US GAAP, to the reasonable satisfaction of the Purchasers. |
| 9.2 | The Purchasers shall be responsible for the satisfaction of the Conditions Precedent pursuant to Clause 9.1.2, the Seller shall be responsible for the satisfaction of the Conditions Precedent pursuant to Clauses 9.1.1, 9.1.3, 9.1.4 and 9.1.5 and the Parties shall jointly be responsible to the satisfaction of the Conditions Precedent pursuant to Clause 9.1.6. Following Signing, (i) Purchaser 2 will appoint PwC PricewaterhouseCoopers (or such other accounting firm as reasonably selected by Purchaser 2) to assist (at Purchaser 2's cost and expense) the Company in the reconciliation of the Financial Statements 2012 with US GAAP; and (ii) KPMG (or such other accounting firm as reasonably selected by Purchaser 2) shall be appointed by the Company to audit (at Purchaser 2's cost and expense) these financial statements of the Company as at 31 December 2012 prepared in accordance with US GAAP. The Seller does not make or give a representation or warranty as to the compliance of these financial statements of the Company with US GAAP. Each of the Parties agrees to act in good faith and to use its respective best efforts to procure that each of the Conditions Precedent is fulfilled as soon as reasonably possible, but at the latest on 1 August 2013 (the "Long-Stop Date"). |
| 9.3 | If Closing does not occur until the Long-Stop Date due to a failure by the Purchasers to obtain the approval of Purchaser 2's shareholders pursuant to Clause 9.1.2 (the "Break-up Event"), Purchaser 2 shall be liable to pay to the Seller the Break-Up Fee as lump sum reimbursement for the Seller's costs and expenses in connection with the transactions contemplated by this Agreement (including but not limited to fees and costs for legal, financial and tax advice). Purchaser 2 shall notify the Seller of the occurrence of a Break-up Event, if any, within 30 (thirty) |
42
Business Days from the date of its general meeting of shareholders. The Break-up Fee, if any, shall be due and payable within 30 (thirty) days from the date of such notice. Except for the Seller's right to terminate the agreement pursuant to Clause 10.3, any further or other Claims of the Seller, including without limitation for damages (including indirect or consequential damages and lost profit) or losses as well as claims based on culpa in contrahendo shall be excluded in a Break-up Event.
| 9.4 | The Parties shall keep each other informed on the progress and status of the fulfilment of the Conditions Precedent and, for such purpose, each Party shall immediately notify the other Party/-ies in writing with respect to the satisfaction of a Condition Precedent for which it is responsible hereunder and shall present to the other Party/-ies documents evidencing, to the reasonable satisfaction of the other Party/-ies, the fulfilment of the respective Condition Precedent. |
| 9.5 | No Party shall take any action between Signing and Closing that is contrary to, or inconsistent with, the provisions of this Agreement or the consummation of the transactions contemplated hereby or any other actions referred to herein. In particular, no Party shall take any action impeding the fulfilment of the Conditions Precedent. |
| 9.6 | The Purchasers shall be entitled, in their sole discretion, to waive the Conditions Precedent pursuant to Clauses 9.1.1, 9.1.3, 9.1.4, 9.1.5 and 9.1.6 at any time between Signing and Closing by written notice to the Seller. |
| 10 | Signing and Closing |
| 10.1 | This Agreement is executed today in Vienna (the "Signing"; the date of Signing is referred to as the "Signing Date"). |
| 10.2 | Subject to the fulfilment or waiver of the Conditions Precedent, the sale and transfer of the Shares as contemplated in Clause 3.2 and Clause 3.3 shall be completed at 10:00 am Vienna time at the offices of Purchasers' counsel, on the 5th (fifth) business day after the last of the Conditions Precedent has been fulfilled or waived (the "Closing"; the date of the Closing is referred to as the "Closing Date") or at such other location, time or date as may be mutually agreed between the Parties in writing. |
| 10.3 | In the event that the Conditions Precedent have not been met or satisfied and Closing therefore has not taken place on or before the Long-Stop Date, the Seller and the Purchasers (acting only jointly) may (in addition to and without prejudice to all other rights and remedies available pursuant to this Agreement) terminate this Agreement by giving written notice to the other Party/-ies, unless the periods have been changed to (a) different date(s) by mutual written consent. Neither the Seller nor the Purchasers shall have a right to terminate this Agreement if they have prevented the occurrence of any of the Conditions Precedent of this Agreement or the Closing. |
43
| 10.4 | At Closing, the Parties shall cause or submit or cause to be submitted, as the case may be, the following actions and documents in the following order: |
| 10.4.1 | The Seller shall deliver to the Purchasers duly executed originals of resignation letters of Dr. Heinz Russwurm, born 01.04.1958, Mag. Heinrich Friedrich, born 06.03.1944, and Dr. Ingrid Aichinger-Widmann, born 29.09.1960, as members of the supervisory board of the Company with effect as of Closing in form and substance corresponding to the draft attached hereto as Exhibit 10.4.1; |
| 10.4.2 | the Seller shall deliver to the Purchasers 1 (one) original of an IP Rights Assignment Agreement ("IP Rights Assignment Agreement") between the Seller and the Company in form and substance corresponding to the draft attached hereto as Exhibit 10.4.2, duly executed by the Seller; |
| 10.4.3 | the Seller shall deliver to the Purchasers the duly executed original of the confirmation pursuant to Clause 9.1.3; |
| 10.4.4 | the Seller shall deliver to the Purchasers written evidence reasonably satisfactory to the Purchasers that the counterparties in respect of each of the contracts relating to the Programs have irrevocably and unconditionally consented to the consummation of the transactions contemplated by this Agreement; |
| 10.4.5 | the Seller shall deliver to the Purchasers the duly executed originals of an IP Rights Assignment Agreement in form and substance corresponding to the draft attached hereto as Exhibit 10.4.5 for each employee of the Company who was or is involved in the development of a (the) Product(s); |
| 10.4.6 | the Seller shall deliver to the Purchasers written evidence that the commercial co-operation agreement by and between the Company and Orpha Trade GmbH (FN 153303 x) dated 15 December 2010 has been extended for a 3-year term, such extension term to commence on 16 December 2013; |
| 10.4.7 | the Seller shall deliver to the Purchasers written evidence reasonably satisfactory to the Purchasers that Orpha Trade GmbH has transferred to the Company or to an affiliate (§ 228 para 3 UGB) of Purchaser 2 as Purchaser 2 may select, all of the assets, rights, licences and Intellectual Property Rights it owns or controls in relation to Tetmodis; |
| 10.4.8 | the Seller shall deliver to the Purchasers (A) a simple copy of the notarial deed de-merger agreement (Spaltungsplan oder Spaltungs- und Übernahsmvertrag) evidencing the tax-neutral (for the Company and the Purchasers) carve-out of the Company's 80 % participation in Activartis Biotech GmbH with corporate seat in Vienna and business address at Zimmermannplatz 10, 1090 Vienna, Austria, registered in the companies’ |
44
register of the Vienna Commercial Court under FN 242030 b, and the loan of the Company as lender and Activartis Biotech GmbH as borrower; as well as (B) a copy of the companies' register filing of this de-merger with the competent commercial court;
| 10.4.9 | the Seller shall deliver to the Purchasers written evidence reasonably satisfactory to the Purchasers that the filings for the transfer of the Marketing Authorisations held by Orpha Devel GmbH in respect of the Products to the Company or to an affiliate (§ 228 para 3 UGB) of Purchaser 2 as Purchaser 2 may select have been initiated; |
| 10.4.10 |
| 10.4.11 | the Seller shall deliver to the Purchasers written evidence reasonably satisfactory to the Purchasers on the tax-neutral (for the Company and the Purchasers) transfer of the Company's shareholdings in (i) Novelix; (ii) Lampl Business Center Holding Company Ltd, Hong Kong; and (iii) shareholding in Pharma 21, Inc (USA) to the Seller or a Third Party; |
| 10.4.12 | the Purchasers shall deliver to the Seller evidence of the satisfaction of the Conditions Precedent pursuant to Clause 9.1.2; |
| 10.4.13 | Purchaser 1 shall issue the instruction(s) for the payment of the Cash Purchase Price pursuant to Clause 4.5.1; and Purchaser 2 shall issue the instruction for the delivery to the Seller of the Consideration Shares in accordance with Clause 4.5.2; |
| 10.4.14 | simultaneously with (Zug-um-Zug gegen) the Closing actions pursuant to Clause 10.4.13, the Seller shall deliver to the Purchasers the originals of the global interim certificates (Sammelzwischenscheine) representing the Shares pursuant to Clause 3.3; and |
| 10.4.15 | Purchaser 2 shall deliver to the Seller (A) a certificate by Purchaser 2's Company Secretary, confirming the admittance to trading of the Consideration Shares on the Main Securities Market of the Irish Stock Exchange in Dublin on the Closing Date; and (B) if applicable, a copy of its notice of the offer of the Consideration Shares to the new issues calendar (Meldung zum Emissionskalender) pursuant to § 13 Austrian Capital Markets Act. |
| 10.5 | All of the actions set forth in Clause 10.4 shall constitute the Closing hereof and will be deemed to have occurred simultaneously; non-occurrence of any of the actions set forth in Clause 10.4 will render null and void any other action with the effect that Closing has not occurred unless such action has been waived by the Party/-ies to whose benefit it had been provided for, and the Closing will then be deemed not consumed but for such waiver. |
| 10.6 | Immediately after Closing, the Seller on the one hand and the Purchasers on the other hand shall each execute a separate closing confirmation letter in form and |
45
substance corresponding to the draft attached hereto as Exhibit 10.6 (the "Closing Confirmation Letter"), confirming to the respective other Party/-ies that all Closing actions have been completed and that Closing has occurred and deliver 1 (one) duly executed original of such Closing Confirmation Letter to the respective other Party/-ies.
| 11 | Purchaser 2 Guarantee |
Purchaser 2 hereby guarantees (§ 880a second case ABGB) the complete and timely performance by Purchaser 1 (or a permitted assignee pursuant to Clause 15.5) of all obligations of Purchaser 1 (or a permitted assignee pursuant to Clause 15.5) pursuant to this Agreement.
| 12 | Confidentiality |
| 12.1 | Neither the Seller nor the Purchasers (nor any of their respective family members or affiliates) shall make any announcement in connection with the existence or subject matter of this Agreement without the prior written approval of the other Party/-ies (such approval not to be unreasonably withheld or delayed). |
| 12.2 | The restrictions in Clause 12.1 shall not apply to the extent that the announcement is required by law, by any stock exchange or any regulatory or other supervisory body or authority of competent jurisdiction, whether or not the requirement has the force of law. |
| 12.3 | The Parties understand and acknowledge that this Agreement and the transactions contemplated hereunder are confidential in nature, and each Party agrees to protect its confidentiality. |
| 13 | Costs and Expenses |
| 13.1 | Each Party shall be responsible for and bear its own costs and expenses incurred in connection with the preparation, conclusion and consummation of this Agreement and the transactions contemplated thereby, including without limitation the costs and expenses of its advisors, in particular attorneys fees, auditors fees, as well as fees of financial advisors, if any. |
| 13.2 | All charges, costs and fees which result from the filings under any merger control law and in compliance with other regulatory requirements, including, but not limited to, the charges, costs and fees of the competent merger control authorities, shall be borne by the Purchaser. |
| 14 | Governing Law and Dispute Resolution |
| 14.1 | This Agreement shall be governed by and construed in accordance with Austrian law, excluding both (i) the United Nations Convention on Contracts for the International |
46
Sale of Goods and (ii) the conflict of law rules of Austrian private international law.
| 14.2 | Any and all disputes arising out of or in connection with this Agreement, including, without limitation, a dispute as to the conclusion, validity or existence of this Agreement, shall be finally and exclusively resolved and settled under the Rules of Arbitration and Conciliation of the International Arbitral Centre of the Austrian Federal Economic Chamber as amended from time to time (the "Vienna Rules"), by 3 (three) arbitrators appointed in accordance with these rules. The language of the arbitration shall be English. The seat of the arbitration shall be Vienna, Austria. Any award and/or final decision of the arbitrators shall include a decision on costs, including, without limitation, fees of counsel. The Parties hereby submit to multiparty proceedings in accordance with the Rules and the Purchasers undertake to nominate the same arbitrator whenever they are respondents in proceedings. |
| 14.3 | Notwithstanding Clause 14.2, the Parties agree that, in addition to any and all other remedies that may be available under this Agreement, each Party shall be entitled to request injunctive relief or to initiate injunction proceedings at the (ordinary) courts of competent jurisdiction. |
| 15 | Final Provisions |
| 15.1 | Each of the Parties shall from time to time execute such documents and perform such acts as any Party may reasonably require to transfer the Shares to Purchasers and to give any Party the full benefit of this Agreement. |
| 15.2 | This Agreement has been drawn up in English and the English language shall therefore be the governing language. Terms to which a German translation has been added shall be interpreted throughout this Agreement in the meaning assigned to them by the German translation. |
| 15.3 | This Agreement is executed in 3 (three) counterparts, one for the Seller and one for each Purchaser, each of which shall be deemed an original and all of which together shall constitute one and the same instrument. |
| 15.4 | This Agreement constitutes the entire agreement and understanding of the Parties relating to the subject matter hereof and supersedes all prior agreements and understandings, whether oral or written, relating to the subject matter hereof. Amendments to and modifications of this Agreement shall be valid only if agreed upon in writing and signed by all Parties hereto; this formal requirement may only be waived in writing. |
47
| 15.5 | Except as expressly provided for in this Agreement, neither this Agreement nor any of the rights, interests or obligations hereunder shall be assigned by any of the Parties without the prior written consent of the other Party/-ies. The Purchasers shall be entitled to assign their rights and obligations out of or in connection with this Agreement, respectively their contractual position hereunder (Vertragsübernahme) to any affiliated undertaking (verbundenes Unternehmen) of the Purchasers within the meaning of § 228 para 3 UGB. In any case, Purchaser 2 shall be entitled to contribute, transfer and/or assign any and all of its rights under or in connection with this Agreement, respectively its contractual position hereunder (Vertragsübernahme), to Purchaser 1. |
| 15.6 | All Exhibits to this Agreement are an integral part of this Agreement. All references in this Agreement to an Exhibit shall be deemed to be references to a Clause of this Agreement unless the context otherwise requires. |
| 15.7 | Clause and subsection headings of this Agreement are for ease of reference only and do not affect the substance, meaning or interpretation of any provision hereof. Words denoting the singular include the plural and vice versa, words denoting any one gender include all genders. All references to legal provisions shall include references to any statutory modification or re-enactment thereof, whether before or after the Signing. |
| 15.8 | The Parties to this Agreement have participated jointly in the negotiation and drafting of this Agreement. If any ambiguity or question of intent or interpretation arises, this Agreement shall be construed as if drafted jointly by all Parties hereto, and no presumption or burden of proof shall arise favouring or disfavouring any Party by virtue of the authorship of any provision of this Agreement. |
| 15.9 | The failure of any Party to enforce or to exercise, at any time or for any period of time any term of or any right or remedy arising pursuant to or under this Agreement shall not constitute, and shall not be construed as, a waiver of such term or right or remedy and shall in no way affect that Party's right to enforce or exercise it later, provided that such right is not time barred or precluded. Any waiver to this effect must be explicitly in writing. |
| 15.10 | If any provision of this Agreement shall be held to be illegal, invalid or unenforceable, in whole or in part, such provision or part shall to that extent be deemed not to form a part of this Agreement but the legality, validity or enforceability of the remainder of this Agreement shall not be affected. The illegal, invalid or unenforceable provision, in whole or in part, shall be replaced by such valid and enforceable provision which corresponds to the original intention and purpose of the illegal, invalid or unenforceable provision. |
| 15.11 | Any notice(s) required or permitted in connection with this Agreement or by law (a "Notice") shall be given in writing in English by an authorized representative of the relevant Party and shall be delivered by hand, sent to the recipient by certified or registered mail or by international courier service (such as DHL, UPS or the |
48
like), and advance facsimile transmission to the address set forth below or an address to be provided by the relevant Party in writing and by certified or registered mail, postage prepaid, with reference to this Clause 15.11 to the other Party. Any Notice shall be effective upon receipt and shall be deemed to have been received:
| 15.11.1 | at the time of delivery, if delivered by hand, registered or certified mail or courier; |
| 15.11.2 | at the time of transmission in legible form, if delivered by fax. |
If to Seller:
PRIVATE & CONFIDENTIAL
Dr. Rudolf Widmann
Wintergasse 85/1b
3002 Purkersdorf
Austria
If to Purchasers:
Elan Corporation, plc
Attn: The General Counsel
Treasury Building
Lower Grand Canal Street
Dublin 2, Ireland
Fax: +353 1 709 4713
With a copy (which shall not constitute Notice) to:
A&L Goodbody Solicitors
Attn: Alan Casey
IFSC
North Wall Quay
Dublin 1, Ireland
Fax: +353 1 649 2649
[Remainder of this page intentionally left blank. List of Exhibits to follow.]
49
List of Exhibits
| Exhibit 1 | List of all Products; | |
| Exhibit 4.4 | Contingent Purchase Price; | |
| Exhibit 4.5.1 | Seller's bank account; | |
| Exhibit 5.2.3 | Current Articles (Satzung) of the Company; | |
| Exhibit 5.2.4 | Current Companies' Register excerpt of the Company; | |
| Exhibit 5.7.1 | Financial Statements 2012; | |
| Exhibit 5.9.3 | Transactions outside the ordinary course of Business since 31 December 2012; | |
| Exhibit 5.10 | List of Company bank accounts; | |
| Exhibit 5.12.10 | List of jurisdictions with taxable presence of the Company; | |
| Exhibit 5.14.1 | List of Patents, Trademarks (including service marks and trade dress) and Domain Names owned and used by the Company; | |
| Exhibit 5.15.2 | List of all real property leased by the Company; | |
| Exhibit 5.16.6 | List of all Material Contracts; | |
| Exhibit 5.22.1 | List of Employees; | |
| Exhibit 5.22.4 | Copies of the Management Board Member Agreements; | |
| Exhibit 5.22.10 | Details of all Company bonus plans; | |
| Exhibit 5.25.2 | Clinical Trials; | |
| Exhibit 5.27.3 | List of all material Marketing Authorisations; | |
| Exhibit 5.33.6 | List of representative offices and area managers; | |
| Exhibit 5.34 | List of Disclosed Documents and Screenshots; | |
| Exhibit 8.2.1 | Permitted Dividend; |
50
| Exhibit 10.4.1 | Supervisory board resignation letters (agreed form); | |
| Exhibit 10.4.2 | IP Rights Assignment Agreement Seller (agreed form); | |
| Exhibit 10.4.5 | IP Rights Assignment Agreement Employees (agreed form); | |
| Exhibit 10.6 | Closing Confirmation Letters (agreed form). |
[Remainder of this page intentionally left blank. Signature page to follow.]
Vienna, this 19 May 2013
For the Seller
| /s/ Dr. Rudolf Stefan Widmann |
Dr. Rudolf Stefan Widmann
born 24.03.1957
For SASR Neunundvierzigste Beteiligungsverwaltung GmbH
John Given, General Counsel
acting under PoA dated 16 May 2013
For Elan Corporation, plc
John Given, General Counsel
acting under PoA dated 16 May 2013
Vienna, this 19 May 2013
For the Seller
Dr. Rudolf Stefan Widmann
born 24.03.1957
For SASR Neunundvierzigste Beteiligungsverwaltung GmbH
| /s/ John Given |
John Given, General Counsel
acting under PoA dated 16 May 2013
For Elan Corporation, plc
| /s/ John Given |
John Given, General Counsel
acting under PoA dated 16 May 2013























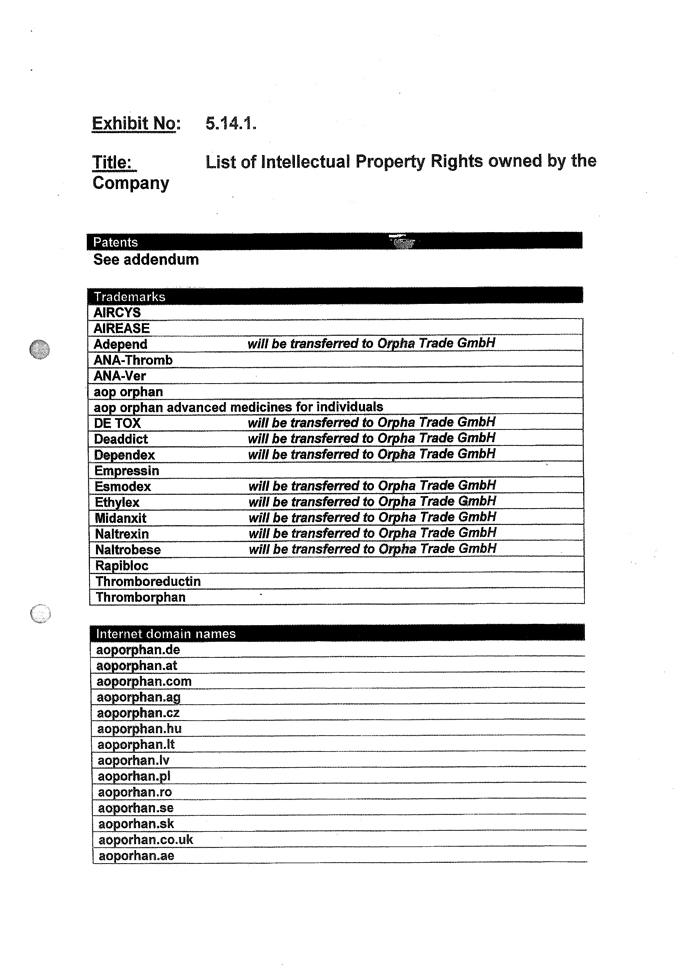
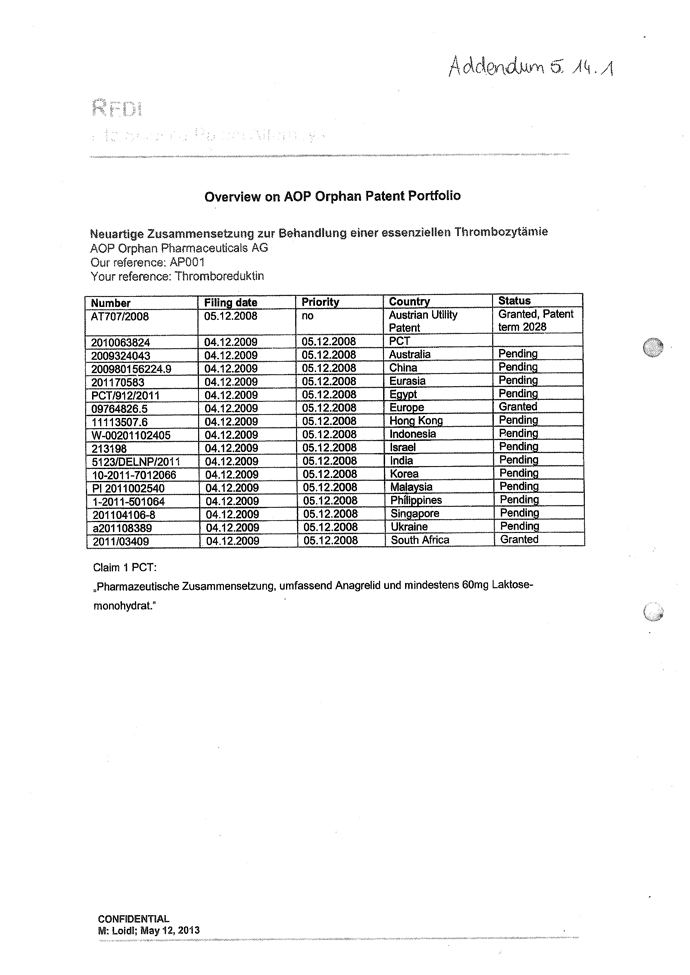










































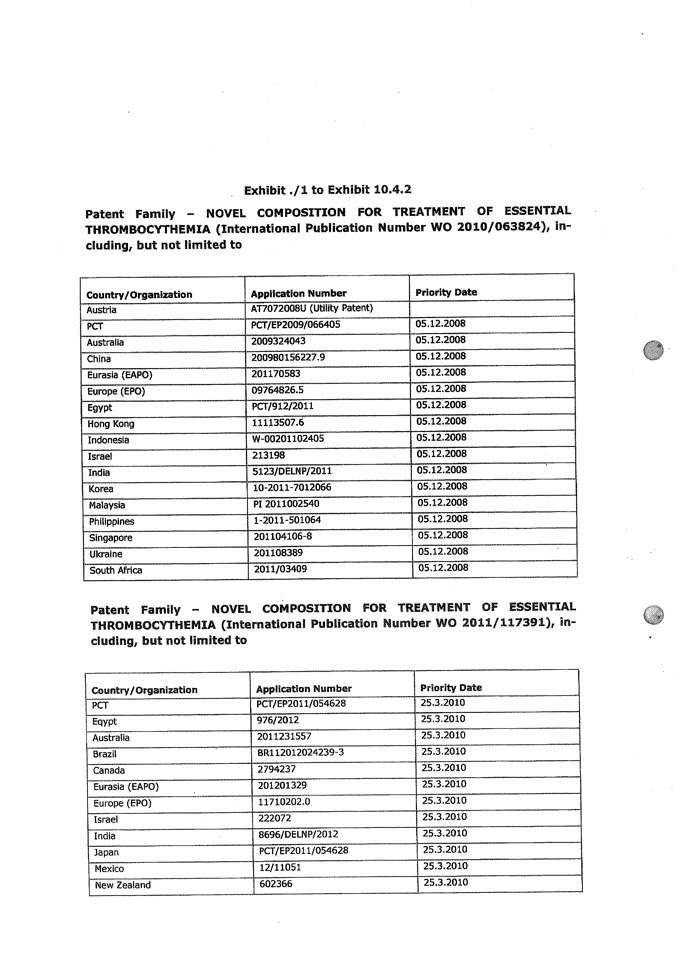













Exhibit No: Title: List of all Products Product Thromboreductin Remodulin . Tetmodis Britlofex ·· Busilvex . IVHebex Canemes Ospolot .. Oxymetholone ·Empress in Vitamin E Tadim . Promixin IV Tadim Inhale Wilfactin Hemoleven Adasuve Staccato Exhibit No: 4.4 Title: Contigent Purchase Price See addendum Exhibit 4.4 1 Definitions 1.1 Unless expressly otherwise defined in Clause 1.2 of this Exhibit 4.4, defined terms used herein shall have the meaning given to them in Clause 1 (Definitions) of the Agreement. 1.2 In addition, the following defined terms shall have the following meaning: Anagrelide Retard Program Clinical Failure EMA Existing Molecule landiolol IPrrogram M6201730/6 means a clinical development program for a new formulation (e.g., for extended release) of Anagrelide, i.e. Anagrelide retard, aimed at submission for regulatory approval via a line extension for the currently marketed formulation Thromboreductin (Anagrelide) in the current Indication, and or achieving a new indication for the treatment of essential thrombo ·Cythaemia In a new subset of patients (e.g., "At risk" Patients With Essential Thrombocythaemia (ARETA)) in addition to the current indication of Thromboreductin ; means a clinical event or clinical evidence which does not support progression or continued development of a Program on the grounds of safety or efficacy; means the European Medicines Agency as established by Regulation (EC) No 726/2004 of the European Parliament and of the Council of 31 March 2004; means a molecule for an Orphan Drug candidate currently held by the Company; means a landiolol pharmaceutical/ preclinical and clinical program to allow regulatory submission in the in Milestone Payment(s) New Molecule North America Rights Peg IFN a 2b Program Phase II Phase UI Orphan Drug M6201730/6 2. dication control of heart rate for various clinical conditions; has the meaning set out in Clause 2.2 of this Exhibit 4.4; means a molecule in Phase II or Phase III of clinical development for an Orphan Drug candidate not currently held by the Company and as agreed upon to Purchaser 2 such consent not to be unreasonably withheld; means exclusive development and commercial rights to North America (i.e. the United States of America and Canada); means a Peg IFN a 2b (e.g., AOP2014) pharmaceutical, preclinical and clinical program to allow regulatory submission in the Indication polycythaemla vera; means a well controlled, closely monitored, clinical study conducted to evaluate the dose dependent effectiveness of a drug product for a particular Indication or Indications In patients with the disease or condition under study and to determine the common short term side effects and risks associated with such drug product; means as set forth In 21 Code of Federal Regulations Section 312.21 of the United States, as amended or as defined by the relevant government health authority (or successor agency thereof) in any country of the world whose approval is necessary to market and sell a drug product In the relevant country of the world; means a drug product that Is tended for the safe and effective treatment, diagnosis or prevention of a rare disease/disorder that affects fewer than 200,000 people In the United States, or that affects more than 200,000 persons but are not expected to recover the costs of developing and marketing of such treatment drug; In the European Union not more than 5 out of 10.000 patents are affected In a rare disease; Regulatory Acceptance means validated by a regulatory authority, i.e. by the EMA (centralised procedure) or national regulatory authorities (national/mutual recognition or decentrallsed procedure) submissions of regulatory dossiers upon application by the Company; Reslmmune Program means a bi functional anti CD3· . diphtheria toxin fusion protein (e.g., recombinant anti CD3 bl slnglechain Fv dlphtherla toxin fusion protein, A dmDT390 blsFv) pharmaceutical, preclinical and clinical program to allow regulatory submission in the Indication cutaneous or peripheral T cell Lymphoma. 2. Payment of the contingent Purchase Price 2.1 Following Closing, the Seller shall use his Best Efforts to obtain North America Rights to the Peg IFN a 2b Program for the Company. 2..2 Subject to the terms and conditions of the Agreement and this Exhibit 4.4, the Seller shall be entitled to the Contingent Purchase Price, If any, as follows: 2.2.1 2.2.2 2.2.3 M6201730/6 If Regulatory Acceptance has been obtained for the Landlolol Program on or before 31 December 2016, an amount of EUR 65,000,000 (Euro sixty five miiUon) shall be payable; if Regulatory Acceptance has been obtained for the Anagrelide Retard Program on or before 31 December 2017, an amount of EUR 35,000,000 (Euro thirty five million) shall be payable; if Regulatory Acceptance has been obtained for the Peg IFN*Ot 2b Program l . on or before 31 December 20171 an amount of EUR 85,000,000 () (Euro eighty five million) shall be payable 4 2. 2.4 If Regulatory Acceptance has been obtained for the Resimmune Program on or before 31 December 2017, an amount of EUR 40,000,000 (Euro forty million) shall be payable; and/or 2.2.5 If, on or before 31 December 2017, the Company either: (i} acquires by way of In licence or otherwise the development and commercial right to New Molecules; or (II) achieves Regulatory Acceptance of an Existing Molecule; then, an amount of EUR 15,000,000 (Euro fifteen million) per Existing Molecule or New Molecule (as the case may be) shall be payable subject always to a cap of EUR 45,000,000 (i.e. a cap of 3 such molecules). Each potential payment pursuant to this Clause 2.2 of this Exhibit 4.4 Is individually also referred to as a "Milestone Payment" and collectively they are also referred to as the "Milestone Payments". 2..3 Each Milestone Payment shall be contingent upon the Seller being, at the time when the respective Milestone Payment becomes due and payable pursuant to Clause 2.4 of this Exhibit 4.4, a member of the management board (Mitglled des Vorstands} of the Company, appointed by the supervisory board (Aufsichtsrat) of the Company. If the Seller's appointment Is terminated by the Company without cause or due to the Seller's inability to continue his work for the Company due to complete disability prior to 31 December 2017 and (A} a Program Is abandoned by the Company for reasons other than Clinical Failure, the Seller's 75 % entitlement pursuant to Clause 2.7 of this Exhibit 4.4 shall remain unaffected; or (B) In the case of Clauses 2.2.1 through 2.2.4 of this Exhibit 4.4 only, Regulatory Acceptance Is obtained, the Seller's entitlement to the Milestone Payments shall remain unaffected. 2.4 If and to the extent triggered pursuant to Clause 2.2 of this Exhibit 4.4, each Milestone Payment shall be due and payable within 90 (ninety) days from (A) Regulatory Acceptance; or (B) receipt by Purchaser 1 of the Seller's notice pursuant to Clause 3.1 of this Exhibit 4.4, whichever Is the later. 2.5 Each Milestone Payment shall be separate and Independent of any and all other Milestone Payments, If any. In case one or more Milestone Payments are not achieved (e.g. In case of failure to achieve Regulatory Acceptance for a Program prior to a deadline as set forth In Clause 2.2 of this Exhibit 4.4), all of the Seller's rights and claims to such Milestone Payment shall be terminated and forfeited (I.e. the Seller shall In particular not be able to compensate for underachievement with respect to one Milestone Payment by overachievement with respect to another Milestone Payment). 2.6 If and to the extent due, a Milestone Payment shall be payable by Purchaser 1 to the Seller's bank account set forth in Exhibit 4.5.1 of the Agreement or such other bank account of the Seller as the Seller may from time to time notify to the Purchaser 1 In writing pursuant to Clause 15.11 of the Agreement. . 5. 2.7 With respect to Clauses 2.2.1 through 2 2.4 of this Exhibit 4.4 only: In the event that a Program .Is abandoned by the Company for reasons other than Clinical Fallure. on of prior to 31 Decernber.2017; the Seller wm be entitled to a payment equivalent to 75·% (seventy".flve ·percent) of the respective Milestone Paymen which would have been payable to the Seller had that Program obtained Regula tory Acceptance. 2.8 The Contingent Purchase Price, If any, shall in no event exceed an amount of EUR 270,000,000 (Euro two hundred seventy million). 3 Seller's Notice 3.1 The Seller undertakes to notify Purchaser 1 In writing pursuant to Clause 15.11 of the Agreement without undue delay but in any case within 5 (five} Business Days of: 3.1.1 3.1.2 3.1.3 M6201730/6 Regulatory Acceptance in the case of Clauses 2.2.1 through 2.2.4; acquisition of a New Molecule and/or Regulatory Acceptance of an Existing Molecule In the case of Clause 2.2.5; and having obtained North America Rights in the case of Clause 2.1. Exhibit No: 5.2.3 Tftfe: .,. Curren.t Articles· of Asso iation (Satzung) of the , \I See addendum Satzung der AOP Orphan Pharmaceuticals Aktiengesellschaft 1. GrUnderin Stand per 03.02.2011] AOP Orphan Pharma Research AG (vormals AOP Orphan Pharmaceuticals Aktie ngesellschaft) 2. Firma Die Firma der Gesellschaft lautet: AOP Orphan Pharmaceuticals Aktiengesellschaft 3. Sitz und Zweigniederlassungen 3.1 Die Gesellschaft hat ihren Sitz in Wien. 3.2 Die Gesellschaft kann Zweigniederlassungen im In und Ausland errichten. 4. Gegenstand des Vertrages Gegenstand des Unternehmens ist 4.1 der Handel mit Waren aller Art, insbesondere von Arzneimitteln und diagnostischen Produkten; 4.2 die Entwicklung und Herstellung von Arzneimitteln und diagnostischen Produkten; 4.3 der Erwerb und die Verwaltung von Beteiligungen an anderen Gesellschaften im lnund Ausland, insbesondere von solchen, die in gleichen oder ahnlichen Geschaftszweigen tatig sind; 4.4 die Erwerbung von Rechten an Medikamenten und diagnostischen Produkten. I I I 2 4.5 Dari.iber hinaus ist die Gesellschaft zu allen Geschaften und Ma nahmen berechtigt, die zur Erreichung des Gesellschaftszweckes notwendig und nOtzlich erscheinen 4.6 Bankgeschafte nach dem BWG sind ausgeschlossen. 5.. Dauer und Geschaftsjahr der Gesellschaft 5.1 Die Gesellschaft wird auf unbestimmte Zeit errichtet. 5.2 Die Geschaftsjahre beginnen mit dem 01. Janner und enden mit dem darauffolgenden 31. Dezember. 6. Veroffentlichungen Veroffentlichungen der Gesellschaft erfolgen durch Einschaltung in der .. Wiener Zeitung". 7.1 7. Grundkapital und Aktien Hohe und StOcke lung Das Grundkapital der Gesellschaft betrag € 200.000,00 (zweihunderttausend) und ist zerlegt in 20.000 (zwanzigtausend) Stock StOckaktien. 7.2 Gattung der Aktien 7.3 Die Aktien Iauten auf lnhaber. Trifft im Faile einer Kapitalerhohung der ErhOhungsbeschluss keine Bestimmung daruber, ob die Aktien auf den lnhaber oder auf Namen Iauten, so Iauten sie ebenfalls auf den lnhaber. Ausgabe der Aktien Ausgabebetrag und Ausgabebedingungen hat der Vorstand im Einvemehmen mit dem Aufsichtsrat festzusetzen. 7.4 Stimmrecht Das Stimmrecht wird nach Stockaktien ausgeObt; je eine Stockaktie gewahrt das Recht auf eine Stimme. r 7.5 Aktienurkunden L..f) Form und lnhalt der Aktienurkunden sowie der Gewinnanteil und ErneuerungsscheinE setzt der Vorstand im Einvernehmen mit dem· Aufsichtsrat fest; entsprechendes gilt etwa fUr auszugebende Zwischenscheine sowie Schufdverschreibungen und deren Zinsschelne .... 8. Organe Organe der Gesellschaft sind: der Vorstand der Aufsichtsrat die Hauptversammlung 8.1 Der Vorstand 8.1.1 Zusammensetzung Der Vorstand besteht aus einer, zwei oder drei Personen und wird durch den Aufsichtsrat gewahlt. Der Aufsichtsrat ist zum Widerruf der Bestellung a us Grunden im Sinne des § 75 Abs 4 des Aktiengesetzes berechtigt. 8.1.2 Vertretungsrecht Die Gesellschaft wird, wenn der Vorstand aus einer Person besteht, durch diesen, wenn er aus mehreren Personen zusammengesetzt ist, durch zwei Vorstandsmitglieder gemeinsam oder durch ein Vorstandsmitglied gemeinsam mit einem Prokur[sten vertreten. Auch bei Vorhandensein mehrerer Mltglieder des Vorstandes kann einem oder mehreren Mitgliedern die Befugnis zur alleinigen Vertretung der Gesellschaft erteilt werden. 8.1 .3 Werden mehrere Personen zu Vorstandsmitgliedern bestellt, so kann der Aufsichtsrat ein Mitglied zum Vorsitzenden des Vorstandes ernennen. 8.1.4 Mitglieder des· Vorstands sind berechtlgt, gleichzeitig bei Kapitalgesellschaften, an denen die Gesellschaft zumindest 50 % des Grund oder Stammkapita!s halt ( .. Tochtergesellschaften"), Organfunktionen, insbesondere als Vorstandsmitglieder oder GeschaftsfUhrer, zu Obernehmen und in ihrer Funktion als Vorstandsmitglieder der Gesellschaft Geschafte mit diesen Tochtergesellschaften, vertreten durch sie selbst, zu schlier.!.en (Doppelvertretung). Das handelnde Vorstandsmitglied oder die handelnden Vorstandsmitglieder haben Ober aile derartigen Geschafte im Rahmen einer (o 4 Doppelvertretung unverzuglich eine Urkunde uber den lnhalt des Geschaftes zu errichten. 8.2 Der Aufsichtsrat 8.2.1 Zusammensetzung und Funktionsperiode 8.2.1.1 Der Aufsichtsrat besteht aus mindestens drei, hochstens sechs von der Hauptversammlung gewahlten Mitgliedern 8.2.1.2 Die Bestellung des ersten Aufsichtsrates gilt bis zur Beendigung der ersten Hauptversammlung, die nach Ablauf eines Jahres seit der Eintragung der Gesellschaft in das Handelsregister zur Beschlussfassung f.lber die Entlastung stattfindet. 8.2.1.3 Danach wahrt die Funktionsperiode eines Aufsichtsratsmitgliedes bis zur Beendigung der Hauptversammlung, die Ober die Entlastung fOr das zweite Geschaftsjahr nach der Wahl beschlieBt. 8.2.1.4 8.2.1.5 8.2.1.6 8.2.2 8.2.2.'1 Ein Aufsichtsratsmitglied kann seine Funktion jederzeit unter Einhaltung einer vierzehntagige n Kundigungsfrist zurucklegen. Die Bestellung zum Aufsichtsratsmitglied kann vor Ablauf der Funktionsperiode von der Hauptversammlung widerrufen werden. Der Beschluss bedarf der einfachen Mehrheit derabgegebenen Stimmen. Der Aufsichtsrat wahlt alljahrlich in einer im Anschluss an die ordentliche Hauptversammlung abzuhaltende Sitzung, zu der es keiner besonderen Einladung bedarf, aus seiner Mitte einen Vorsitzenden und einen stellvertretenden Vorsitzenden. Findet keine Wahl im Anschluss an die ordentliche Hauptversammlung statt, so gilt der Vorsitzende und der stellvertretende Vorsitzende so lange als wiedergewahlt, bis im Anschluss an eine ordentlichen Hauptversammlung abgehaltenen Sitzung des Aufsichtsrates der Vorsitzende und/oder der stellvertretende Vorsitzende neugewahlt wird, wobei eine Wiederwahl zulassig ist. Aufgaben Der Aufsichtsrat hat die GeschaftsfOhrung des Vorstandes zu Qberwachen. 5 Zu folgenden Geschaften bedarf der Vorstand der vorherigen schriftlichen Zustimmung des Aufsichtsrates: Erwerb und Verauf1erungen von Beteiligungen (§228 HGB) sowie Erwerb, Verauflerung und Stillegung von Unternehmen und Betrieben, sofern damit eine Gegenieistung verbunden ist, die einen Betrag von € 2,000.000,00 Obersteigt. Erwerb, Verauflerung und Beiastung von Liagenschaften, sofern damit eine Gegenleistung verbunden ist, die einen Betrag von € 2,000.000,00 Obersteigt. Errichtung und Schlief!ung von Zweigniederlassungen lnvestitionen, bei denen die Anschaffungskosten € 1,000.000,00 im einzelnen und insgesamt in einem Geschaftsjahr€ 4,000.000,00 i.ibersteigen, Aufnahme von Anleihen, Darlehen und Krediten, die einen Betrag von € 1,000.000,00 im einzelnen und insgesamt in einem Geschaftsjahr € 4,000.000,00 ubersteigen, Gewahrung von Darlehen und Kreqiten, die einen Betrag von € 1,000.000,00 im einzelnen und insgesamt in einem GeschiMtsjahr € 4,000.000,00 Obersteigen, Aufnahme und Aufgabe von Geschaftszweigen und Produktionsarten, Festlegung a!lgemeiner Grundsatze der Geschaftspolitik, Festlegung von Grundsatzen uber Gewahrung von Gewinn und Umsatzbeteiligungen und Pensionszusagen an leitende Angestellte im Sinne des § 80 Abs 1 Aktiengesetz, Die Einraumung vor; Optionen auf Aktien der Gese!lschaft an Arbeitnehmer und leitende Angestellte der Gesellschaft oder eines mit ihr verbundenen Unternehmen, Erteilung der Prokura, Oas Angebot der Teilnahme der Aktionare an der Hauptversammlung im Wege elektronischer Kommunikation (8.3.3.4) sowie die Obertragung der Hauptversammlung an die nicht anwesenden Aktionare (8.3.3.5). 6 8.2.3 Ein Aufsichtsratsmitglied kann ein anderes Aufsichtsratsmitglied oder einen osterreichischen Rechtsanwalt oder offentlichen Notar schriftlich bevollmachtigen, in einer bestimmten Sitzung an seiner Stelle auch schriftlich das Stimmrecht auszuuben. 8.2.4 8.2.4.1 Beschlussfahigkeit und Entscheidungen im Aufsichtrat Der Aufsichtsrat ist beschlussfahig, wenn drei seiner Mitglieder, darunter der Vorsitzende oder sein Stellvertreter, anwesend sind. 8.2.4.2 BeschiOsse des Aufsichtsrates werden, soweit nicht ein Mheres Quorum vorgeschrieben ist, mit einfacher Mehrheit der abgegebenen Stimmen gefasst. Bei Stimmengleichheit ist in der nachsten Aufsichtsratssitzung neu abzustimmen. 8.2.5 Willenserklarungen Willenserklarungen des Aufsichtsrates werden vom Vorsitzenden oder im Faile seiner Verhinderung vom Stellvertreter abgegeben. 8.2.6. VergOtung 8.2.6.1. Die Mitglieder des Aufsichtsrates erhalten Ersatz der bei der Ausubung ihrer Tatigkeit erwachsenden baren Auslagen. 8.2.6.2. Oberdies erhalten die. Mitglieder des Aufsichtsrates eine Aufwandsentschadigung, deren Hohe die Hauptversammlung bestimmt. 8.2.6.3. Der Aufsichtsrat kann Satzungsanderungen, die nur die Fassung betreffen, beschlief!.en. 8.3 Die Hauptversammlung 8.3.1 Ort Die Hauptversammlungen der Gesellschaft finden am Sitz der Gesellschaft, an einem ihrer Zweigniederlassungen oder in einer in der Einladung zur Hauptversammlung genannten Landeshauptstadt Osterreichs statt. 8.3.2 Einberufung Die Hauptversammlungen werden vom Aufsichtsrat oder Vorstand einberufen. Die Einberufung ist unter Angabe des Gegenstandes der Hauptversammlung unter Einhaltung einer Frist von 28 Tagen bei Einberufung einer ordentlichen ltJ 8.3.3 8.3.3.1 8.3.3.2 8.3.3.3 8.3.3.4 8.3.3.5 8.3.4 . 7 Hauptversammlung, ansonsten unter Einhaltung einer Frist von 21 Tagen zu veroffentlich n. An namentlich bekannte Aktionare hat eine gesonderte schriftliche Einladung mit einem Verzeichnis der Tagesordnungspunkte unter Einhaltung der oben genannten Frist zu ergehen, wobei die Versendung eingeschrieben und an die zuletzt der Gesellschaft bekannt gegebene Adresse erfolgt. Fur den Lauf der Frist gi!t der Tag der Postaufgabe als Tag der Bekanntmachung. Ein Aktionar kann der Gesellschaft stattdessen eine elektronische Postadresse bekannt geben und in die Mitteilung der Einberufung auf diesem Weg einwilligen. Sofern der Gesellschaft alfe Aktionare namentllch bekannt sind, kann die Einberufung per Brief und elektronischer Post an Stelle der Bekanntmachung erfolgen. Teilnahme und Ausubung des Stimmrechtes Zur Teilnahme an der Hauptversammlung ist jeder Aktionar berechtigt, der bei der Gesellschaft, bei einem osterreichischen offentlichen Notar oder bei der Hauptniederlassung eines inlanG!ischen Kreditinstitutes die Aktien spatestens am siebenten Tag vor der Hauptversammlung bis zur Beendigung der Hauptversammlung hi nterlegt. Die Bescheinigung der autorisierten Stelle uber die erfolgte Hinte legung der Aktien ist vor der Hauptversammlung bei der Gesellschaft einzureichen. Sind weder Aktien noch Zwischenscheine ausgegeben, so sind die Voraussetzungen fur die Teilnahme in der Einladung zur Hauptversammlung bekannt zu geben. Der Vorstand kann vorsehen, dass Aktionare an der Hauptversammlung im Wege elektronischer Kommunikation teilnehmen und auf diese Weise einzelne oder aile Rechte ausuben konnen. Er kann den Aktionaren insbesondere eine Satellitenversammlung, eine Fernteilnahme und eine Fernabstimmung anbieten. lm Faile des Angebots einer Fernabstimmung hat der Vorstand das Verfahren zur Abstimmung zu regeln und auf welche Weise Aktionare Widerspruch erheben konnen. Der Vorstand kann vorsehen, dass die Hauptversammlung fUr die nicht anwesenden Aktionare akustisch und allenfalls auch optisch in Echtzeit Obertragen wird. Vertretung Ein Aktionar kann sich in der Hauptversammlung durch einen schriftlichen Bevollmachtigten vertreten lassen. 8.3.5 8.3.5.1 8.3.5.2 8 Vorsitz Den Vorsitz in der Hauptversammlung fOhrt der Vorsitzende des Aufsichtsrates oder sein Stellvertreter. 1st keiner von diesen erschienen oder zur Leitung der Versammlung bereit, so !eitet der zur Beurkundung beigezogene Notar die Hauptversammlung zur Wahl des Leiters.der Hauptversammlung. Der Vorsitzende der Hauptversammlung bestimmt die Reihenfolge der Verhand!ungsgegenstande sowie Art und Form der Abstimmung. 8.3.6 Beschlussfi\ihigkeit Die Hauptversammlung ist beschlussfahig, wenn zumindest 65% des Grundkapitals vertreten sind. Wird dieser Prozentsatz nicht erreicht, kann unter Einhaltung der Bestimmungen dieser Satzung eine neuerliche Hauptversammlung einberufen werden, welchs dann jedenfalls unabhangig vom vertretenen Grundkapital beschlussfahig ist. 8.3.7 Mehrheiten Die Hauptversammlung fasst ihre BeschiOsse mit einfacher Stimmenmehrheit des vertretenen stimmberechtigten Grundkapitals, soweit nicht das Gesetz zwingend eine andere Mehrheit verschreibt. 8.3.8 Die Zuschaltung von Mitgliedern des Vorstandes oder es Aufsichtsrats Ober eine optische und akustische Zweiweg Verbindung ist gestattet. Auf die selbe Weise kann auch der AbschlussprOfer den Verhandlungen zugezogen werden. 9. Jahresabschluss und Gewinnbeteifigung 9.1 Vorlagefrist Der Vorstand hat in den ersten funf Monaten des Geschaftsjahres fOr das abgelaufene Geschaftsjahr den Jahresabschluss und einen Geschaftsbericht aufzustellen und nach PrOfung durch den Abschlussprufer nebst einem Vorschlag fur die Gewinnverteilung dem Aufsichtsrat vorzulegen. Der Aufsichtsrat kann diese Frist Ober Antrag des Vorstandes aus wichtigen Grunden um zwei Monate verlangern. 9.2 Feststellung des Jahresabschlusses Die Hauptversammlung beschlieP..t alljahrlich in den ersten acht Monaten des Geschaftsjahres Ober die Entlastung des Vorstandes und des Aufsichtsrates, Ober die Verwendung des im Vorjahr erzielten Jahresergebnisses, Ober die Wahl des 9 AbschlussprOfers und in denim Gesetz vorgesehenen Fallen Ober die Feststerlung des Jahresabschlusses. 9.3 Gewinnverteiiung Soweit die Hauptversammlung keine andere V erwendung _ beschliel1t, wird der . Bilanzgewinn auf die Aktionare im Verhaltnis der auf die Aktien eingezahlten Einlagen 'verteili. Die uptversammfung kann den Blla zgewinn aber at.ich ganz ode"f.tel,lweise1 von derVerteilung ausschliel1en odereine andere Verwendung beschliel1en. Soweit die Hauptversammlung nichts anderes festsetzt, sind Dividenden binnen 30 Tag .m nach der ordentlichen Hauptversammlung zur Zahlung an die Aktionare fallig. Dividenden, die von den Aktionaren nicht innerhalb von drei Jahren nach Falligkeit in Empfang genommen werden, sind veriallen und werden den freien ROcklagen der Gesellschaft zugewiesen. 10. GrUndungskosten 10.1 Die mit der Errichtung und Regrstrierung der Gesellschaft verbundenen Kosten und Abgaben werden bis zu ein m Hochstbetrag von € 20.000,00 (zwanzigtausend) von der Gesellschaft getragen. 10.2 Die Grundungskosten ind mit der Hohe der tatsachlich aufgewendeten Betrage als Ausgaben in die erste Jahresrechnung einzustellen. 11.1 11. Sonstige Bestimmungen Weder einem Aktionar noch einem Dritten wurde ein besonderer Vorteil elngeraumt (§ 19/1 AktG). Betrifft: Handelsgericht Wien Firma: AOP Orphan Pharmaceuticals AktiengeseUschaft. FN 82506 h lch beurkunde, dass der Wortlaut der Satzung der vorgenannten Geseltschaft mit dem in der Hauptversammlung vom 3.2.2011 beschlossenen, durchgreifend neu gefassten Satzung vollkommen ubereinstimmt. M· Wien, am dritten Februar zweitausendelf. Dr. Christoph PFAFFENBERGER NOTARp TNER uno SUBSTITUT DES 0FFENTLICHEN NOTARS WERNER POJNOL . · .. JNNERE STAO'r Exhibit No: 5.2.4 Title: Current Companies' Register Excerpt of the Company See addendum REPUBLIK O'STERREICH FIRMENBUCH Grundlage dieses Aus lges i st. das Hauptbuch erganzt um Daten a us clBr sammlung. Urkunden ·· Letzte Eintragung am 12 .10. 2012 mit der.· Eintr:agungsnumrner H zustandiges Gericht. Handelsgericht Wien FIID4A 1 AOP O:r:phan Pharmaceuticals Aktiengesellschaft RECHTSFORM 1 Aktiengesellschaft SITZ in 1 polit.ischer Geme.i.nde Wien GESCHAFTSANSCHRIFT 4 WilhelrninenstraEe 91/II:E 1160 Wien GESCHAFTSZWEIG J. Pharmazeutische Industrie KAPITJ.UJ 7 EUR 200.000 .li.RT der AK'I'IEN 3 20.000 StQckaktien STICHTAG fQr JAHRES.li.BSCHioUSS 7 31. Dezember JAHRESABSCHLUSS (zuletzt. eingetragen; weitere siehe Historie) 14 zum 31.12.2011 eingereicht am 05.10.2012 VERTRETUNGSBEFUGNIS 1 D:i.e Gesellschaft wird, wenn mehrere Vor·standsmitgl:i.edeJ: bestellt sind, du:cch zwei Vm:standsmitglieder gemeinsam oder durch e.i.nes von ihnen gemeinsam mit einern Prokuristen vertreten. Der .r,ufsicht.srat kann einzelnen Vorst.andsrnit.gliedern selbstandige Vertretungsbefugnis erteilen. ART DER BEI\.2\.l'.JNTMACHTJNG 1 Die Bekanntmachungen de1:· Gesellschaft erfolgen irn Amtsblatt. zur vhener Zeitung. l Satzung vom 22.07.2003 Seite 1 von 3 Hauptversammlungsbeschluss vom 15.07.2003 d r AOP Orphan Pharmaceuticals A.ktiengese ll schaft (FN 153303 x) als ubertragende Gesellschaft. Spaltu.ng unter: (Tbertragung des Betriebes de.r. OP Orphan Pharma Research AG auf diese Gesellschaft als neu gegri.i.ndete gemass Spaltungsplan vom 6.6.2003 Hauptversamm1 ungsbeschluss vom 19. 01. 200<] Anderung der Satzung im Punkt. 5.2 Hauptversammlungsbeschluss vom 28.09.2007 Kap:i.talerhohung aus Gesellschaftsmitteln urn EUR 54.654,33 beschlossen und durchgefO.hrt. Andenmg der Satzung in den Punkten 5. 2. und 7 .1. Hauptve:csammhmgsbE;schluss vom 03.02. 2011 Neufassun9 der Satzung. VORST.I\ND A F Dr. Rudolf Widmann, geb. 24.03.1957 vertritt seit 15.08.2003 selbstlndig Dr. Georg Fischer, geb. 12.11.1958 vertritt seit 27.06.2008 gemeinsam mit dem Vors.i.t.r:;(·mden des vorstandes oder gemeinsam mit dem Vorsitzenden des Vorstandes und einem weiteren Vorstands··· mitglied . .. UFSICHTSRATSMITGisiED .B DDr. Heinz Russwu:cm, geb. 01..04.1958 Vorsitzende/r c D Mag. Heinrich Friedrich, geb. 06.03.1944 Stellvertreter/in des/der: Vcn·sitzenden Dr. Ingrid Aich.i.ng er· Widmann, geb. 29.09.1960 Mitglied Dr. Rudolf Widmann, geb. 24.03.19 i7 Winter9asse 85/lb 3 002 Purk 7.rsdorf DDr . Heinz Russwurm, gr,;b. 01.04.1958 Mag. Heinrich Friedrich, geb. 06.03.1944 Dz·. Ingrid ... l\ichinger VJidrnann: <;Jeb. 29.09.1960 Dr. Georg Fischer· geb. 12.11.1958 Si'J.en.e<Jasse 8 12/46 1220 Vien VOLI,ZUGSUBERS I CHT Handelsgericht Wien Seite 2 von 3 1 eingetrar:=_ren am 15.08. 2003 Geschi.i.ftsf;;t.ll 71 Fr 8255/03 i Antraq auf Neueintragung einex: Firma eingelangt am 29.07.2003 2 eingetragen am 19.02.2004, Gescha:Etsfall 71 Fr 820/04 i Antrag .:..uf Anderung eingelangt am 29.01.2004 3 eingetragen am 29.12.2004 Ge.schiift.s.faJ.l 71 Fr 13460/04 y Einreichunq ,Jahresa.bschluss eingelangt am .22. 1.2. 2004 4 eingetragen am 09. 0 . 2006 Geschaftsfall 71 Fr H65/06 i Antrag a.uf Andenmq · eingel<:mgt am 02 .02.2006 7 ein9etragen am 06.11.2007 Geschf>ft:sfall 71 F1· 12962/07 t Antrag auf f.,nderung ei.ngelangt. am 25.10.2007 8 ei.ngetragen am 22.07.2008 Geschi:Ht.sfa1l 71 Fr 9458/0f3 y Antrag auf .ii.nderung eingelan.gt:. am 16.07.2008 11 eingetragen .:;.m 17.02.201.1 Gescha.ftstall 7J. Fr 2587/11 w Ant.r:aq auf Anderung einge.langt a.m 15.02.2011. 14 eingetragen am 1.2.10.2012 El.ekt:roni.sche Einreichung Jahresabschluss Geschlftsfall 71 Fr 18856/12 d eingelangt. am o:: .. 10. 2012 INFORMATION DER OSTERREICHISCHEN NATIONALBANK t:um 16.05. 201.3 giiltis:J '.: Identnumme:r:: 59'76073 Seite 3 von 3 SIGNATUR l Dieses Dokument wurde elektronisch signiert. Hinweis i Auch ein Ausdruck dieses Ookuments hat I I die Beweiskraft einer Ciffentlichen UrkundePrufinforrnation ! Signatur und des Ausdrucks tinden Sie unter: Exhibit No: 5.14.1. Title: List of Intellectual Property Rights owned by the Company Patents 1fi· See addendum Overview on AOP Orphan Patent Portfolio Neuartige Zusammensetzung zur Behandh.mg einer essenzlellen Thrombozytan"lie AOP Orphan Pharmaceuticals AG Our reference: AP001 Your reference: Thromboreduktin Number Filing date .... Priority Country Status AT707/2008 05.12.2008 no Austrian Utility Granted, Patent Patent term 2028 2010063824 04.12.2009 05.12.2008 PCT 2009324043 04.12.2009 05.12.2008 Australia · Pending .. 200980156224.9 04.12.2009 05.12.2008 China Pending 201170583 04.12.2009 05.12.2008 Eurasia Pending PCT/912/2011 04.12.2009 05.12.2008 Egypt Pending 09764826.5 04.12.2009 05.12.2008 Europe Granted 11113507.6 04.12.2009 05.12.2008 Hong Kong Pending W 00201102405 04.12.2009 05.12.2008 Indonesia Pending 213198 04.12.2009 05.12.20()8 Israel Pending 5123/DELNP/2011 04.12.2009 05.12.2008 India Pending 10 2011 7012066 04.12.2009 05.12.2008 Korea Pending Pl2011002540 04.12.2009 05.12.2008 Malaysia Pending 1 2011 501064 04.12.2009 05.12.2008 Phftippines Pending 201104106 8 04.12.2009 05.12.2008 Singapore Pending a201108389 04.12.2009 05.12.2008 Ukraine Pending 2011/03409 04.12.2009 05.12.2008 South Africa Granted Claim 1 PCT: ,Pharmazeutische Zusammensetzung, umfassend Anagrelid und mindestens 60mg Laktosemonohydrat." CONFIDENTIAL M: Loidl; May 12, 2013 Novel composition for treatment of essential thrombocythemia AOP Orphan Pharmaceuticals AG Our reference: AP004 Your reference: Anathromb retard Number Filing date PrioriW Country EP 1 0157772. ·4··· 25.03.2010 no EP 2011117391 25.03.2011 25.03.2010 PCT 976/2012 25.03.2011 25.03.2010 Egypt 2011231557 25.03.2011 25.03.2010 Australia BR112012024239 3 25.03.2011 25.03.2010 Brazil 2794237 25.03.2011 25.03.2010 Canada 201201329 25.03.2011 25.03.2010 Eurasia 11710202.0 25.03.2011 25.03.2010 EP 222072 25.03.2011 25.03.2010 Israel 8696/DELNP/2012 25.03.2011 25.03.2010 India 2013 500526 25.03.2011 25.03.2010 Japan 12/11051 25.03.2011 25.03.2010 Mexico 602366 25.03.2011 25.03.2010 New Zealand 201207067 8 25.03.2011 25.03.2010 Singapore a201212238 25.03.2011 25.03.2010 Ukraine 13637014 25.03.2011 25.03.2010 USA 2012/06911 25.03.2011 25.03.2010 South Africa Claim 1 PCT: Status .... Abandoned Pending Pending Pending Pending Pending Pending Pending Pending Pending Pending Pending Pending Pending Pending Pending "A pharmaceutical composition free of gastric coating comprising anagrelide HCI, a non pH dependent polymer and a pharmaceutically acceptable water soluble acid." CONFIDENTIAL M: Loidl; Mal( 12, 2013 Pharmaceutical composition AOP Orphan Pharmaceuticals AG Our reference: AP003 Your reference: Landioloi/Cyclodextrin Number Filing dat A 2107/2007 21.12.2007 2009079679 22.12.2008 2008340179 22.12.2008 2712414 22.12.2008 200880122163X 22.12.2008 08863830.9* . 22.12.2008 5194/DELNP/201 0 22.12.2008 2010 538268 22.12.2008 58.6200 22.12.2008 _12809927 22.12.2008 Priority no 21.12.2007 . 21.12.2007 21.12.2007 21.12.2007 ' 21.12.2007 21.12.2007 21.12.2007 21.12.2007 21.12.2007 Co !!:L__ Status ·AusJrJ ... Abandoned All PCT countries .. Australia Pending_ Canada .. Penging Japan · E nding New Zealand Granted .. USA nding Validated in: AT, BE, BG, CH, CY, CZ, DE, OK, EE, ES, Fl, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, LV, MC, MT, NL, NO, PL, PT, RO, SE, Sl, SK, TR Main claims: 1. Verwendung eines Cyclodextrins und/oder eines funktionellen Cyclodextrin Derivates zur Er.Mhung der Stabilitat eines ultrakurzwirksamen 11 Adrenorezeptor Antagonisten und/oder eines pharmazeutisch akzeptablen Salzes davon in einer zur parenteralen Verabreichung geeigneten lagerstabilen wasserigen LOsung. 2. Pharmazeutische Zusammensetzung zur parenteralen Verabreichung eines ultrakurzwirksamen r..Adrenorezeptor Antagonisten in Form einer lagerstabilen Losung, im wesentlichen bestehend aus a) einem ultrakurzwirksamen 11 Adrenorezeptor Antagonisten und/oder einem pharmazeutisch akzeptablen Salz davon b) Wasser c) einem Cyclodextrin und/oder einem funktionellen CyclodextrinDerivat. NoV lfast..disintagrating comegsitions 'comprising Nabif ij AOP .Orph_an Pha.rm ceuticals AG · .. · Our;feference ·7APOo'f;t Your reference: Nabiiorie FDT Number .... Filing date Priority Country Status EP 10192497.5 25.11.2010 no EP Abandoned PCT/EP2011/070951 24.11.2011 25.11.2010 PCT PCT/EP2Q11/070951 24.11.2011 25.11.2010 United Arab Emirates Pending PCTIEP2011/070961 24.11.2011 25.11.2010 Australia Pending PCTIJSP2011l!!J;10961 24.11.2011 25.11 .. 2010 stazil Pending PCT/EP2011/070951 24.11.2011 25.11.2010 Canada Pending PCT/EP2011/070951 24.11.2011 25.11.2010 Eurasia Pending ·· 11790937.4 2:4.11.2011 25.11.2010 EP Pending PCT/EP2011/070951 24.11.2011 25.11.2010 Israel Pending 340 24.11.2011 ... 25.11.2010 India Pending PC 2.4.11,2011 25.11.,201 0 Jaoan Pending PC 11(070951 24.11.2011 25.11.2010 Mexico Pending .6()9 24.11.4011 ·· 25.11.2010 New Zealand Pending PC 01t/()7Q951 2 .11.2o11 25.11.201.0 Singapore Pending PC 2011/0'70951 ,24.11.2011 25.11.2010 USA Pending 201302703 24.11.2011 .... 25.11.2()10 South Africa Pending Claim 1 PCT: 1. Composition comprising Nabilone and randomly methylated 11 cyclodextrin (RAMEB) in the weight ratio (dry weight to dry weight) of 1 :60 1:140, preferably in the weight ratio of 1 :90 1 :11 0 and wherein Nabilone and RAMEB are comprised as an aqueous soluble complex .. 11 nr: 1. CONFIDENTIAL M: loidl; May 12, 2013 Exhibit No: 5.15.2 list of all real properties leased by the Company Representative Offices in: Romania Poland Macedonia Slovakia Czech Republic Hungary Lithuania monthly costs in EUR 800,00 1.330,21 2.613,00 1.650,00 .1.938,12 1.467,20 222,02 Exhibit No: 5.22.4 Title: Copies of the Management Board Member Agreements See addendum ANSTEtLUNGSVERTRAG abgeschlossen zwischen AOP Orphan Pharmaceutica Is Aktiengesellschaft, WilhelmlnenstraBe 91/llf, 1160 Wlen (im Folgenden nGesellschaft" genannt) einerseits und Mag. Andreas Steiner, geboren am 09.12.1959 Hofstattgasse 8/6, 1180 Wlen, (im Folgenden Norstandsmltglled" genannt) andererselts 1. Bestellung und Anstellung 1.1. Der Aufslchtsrat der Gesellschaft hat Herrn Mag. Andreas Steiner mit Beschluss vom 18.03.2013 mit Wlrkung 15.05.2013 fUr 3 (drei) Jahre zum Mltglled des Vorstandes der Gesellschaft bestellt. Herr Mag. Steiner hat diese Bestellung angenommen. 1.2. Der gegenstandliche Anstellungsvertrag 1st auf die Bestelldauer von 3 (drel) Jahren, das he16t bis zum 14.05.2016, befrlstet. Sollte der Anstellungsvertrag nicht bis spatestens 6 Monate vor Ablauf dieser 3 Jahre geki.indigt werden, so verlangert sich der Anstellungsvertrag automatlsch urn 1 (ein) weiteres Jahr, sofern der Aufsichtsrat der Gesetlschaft das Vorstandsmltglied vor .Ablauf der Funktlonsperlode zumlndest auf ein weltercs Jahr wlederbestellt. 1 .3. Der Ansteltungsvertrag regelt die vertragllchen Beziehungen zwischen dem Vorstandsmitglied und der Gesetlschaft. 2. Tl:itlgkeltsberelch, Rect'!tsgrundlagen 2.1. Das Vorstandsmltglied 1st kollektiv mit dem Vorstandsvorsftzenden, der.zeit Dr. Rudolf I:'Jidmann, oder mit dem Vorstandsvorsitzenden und einem weiteren Vorstandsmitglied zur Vertretung der Gesellschaft und zur Zelchnung berechtigt (Gesamtvertretungsbefugnls). Der Aufsichtsrat kann die Vertretungsbefugnlsse des Vorstandsmitgliedes durch Beschluss beschranken. 2.2. Das Vorstandsmltglied leltet unter eigener Verantwortung im Rahmen der Geschaftsvertellung fOr den Vorstand die Bereiche Finance & Administration und 1st lnnerhalb dieser Bereiche fiir die Einhaltung samtlicher Rechtsvorschrlften und Sicherheitsstandards, SOWle der fOr die Tatlgkelt der Gesellschaft relevallten Rlchtlinien und Empfehlungen, wie insbesondere die der European Medicines Agency (EMA) und der.lnternational Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), verantwortlich. 2.3. Das Vorstandsmitglied hat die lhm i.ibertragenen Aufgaben mit der Sorgfalt eines ordentlichen und gewlssenhaften Geschaftslelters wahrzunehmen. Grundlagen fOr die Tatigkeit des Vorstandsmitglledes sind die Bestimmungen des Aktiengesetzes, die dem Vorstandsmitglied bekannte Satzung der Gesellschaft, die verbindlichen Beschllisse des Aufslchtsrates der Gesellschaft, die Geschaftsordnung fur den Vorstand, sowie der gegenstandliche Anstellungsvertrag. Verletzungen der In dlesen Grundlagen festgehaltenen Verpfllchtungen konnen Schadenersatzansprilche der Gesellschaft begrGnden. 2 3. Geheimhaltungspfllcht 3.1. Das Vorstandsmitglled 1st verpflic:htet, ilber vertraullche Angaben Stillschweigen zu bewahren und samtliche Geschiifts und Betrlebsgehelmnlsse der Gesellschaft gegenuber jedem Dritten und, wenn dies lm Interesse der Gesellschaft geboten erschelnt, auch gegenUber Angehorlgen der Gesellschaft, sowelt dlese nicht Kraft lhrer dienstllchen Funktlon und Tatlgkelt zur Entgegennahme derartiger Mitteilungen befugt sind, geheim zu halten. Als Geschafts und Betriebsgehelmnlsse sind ohne ROckslcht auf Patentlerfllhlgkelt oder sonstlge rechtllche Schutzfahigkelt insbesondere aile lm Zuge der dlenstllchen Tlitlgkelt des Vorstandsmitglledes von der Gesellschaft ausgeObten Forschungs und Entwlcklungstatlgkelten und dadurch direkt oder indirekt gewonnenen Ergebnisse, siimtliche durchge{Qhrten Studlen, sowie aile fllr die Gesellschaft gewonnenen Arbeltsgehelmnisse, Erfahrungen und so.nstlgtln Erkenntnlsse .. zu versteh n,. deren Gehelmhaltung nach dem pfllt;htgemiiBmt ·EI'messen elnes .9eWissenhaften Gesdt ftsleiters Is geheilllhattuf\gsbedurftlg anzusehen sind undloder deren Gehefmhaltung aus anderen GrUnden lm Interesse der Gesellschaft g boten erscheint. 4. &\tg lt 4.1. 4.2. 4.3. .Z\Isi t h .zu . m I!J:J , 4.1. ten U tug E\rll!ilt d!ls \GQrstaJ1dsmJtglied J;.lhrlich · eine Grati *J(iln, deren Pm Au 5 Monat\igt}f:Y tter(<t naall MaRgabe des Err chen ·Qer Cieschaft.sz.l .( . nd der.fi)flr tll chen t t des Voi'Startdllmit!!lltedes festgelegt wlrd. Die Gest;"t\ lel.e . erden a t:. gltlo d $ A \,len GescMf,t jahres bekann b n und die Definition der p rsQflfldlen Zl!!fe vet;eJhbart. OJe Gratifikatloh Wlrd nach fofbsdlluss des Geschaftsjahres ausg,e tdt. Oer A c: r : t. qo ,4t l!e Gratrtlkatlon n1it Absc:hluss des Geschilftsjahres fur das folgende Geschliftsjahr elnseitig widerrufen. 4.4. Dem Vorstandsmltglied wird fUr die Dauer seiner Tatigkelt eln Dienstfaluzeug zur Verfligung gestellt. Fur die Beni:itzung dieses Dienstfahrzeuges gelten die D:estimmungen der AOP Orphan Company Car des Dienstfahrzeuges, welcher inkl. USt festgelegt wlrd. Ole zum wird als Anhang./1 zum Vertrag genom men. Das Vorstandsmitglled erklart, d1:1ss er sbth fOr n Fall der Anpassung dleser Rlchtllnle an aktuelle Gegebenhelten, er sich als Vorstandsmltglied ebenfalls dieser Anpassung unterwerfen wird. 5. Dienstreisen ..5.1. lm Faile der Verwendung seines Privatfahrzeuges fOr dienstllche Zwecke ist das Vorstandsmitglled zur Verrechnung von Kilometergeld an die Gesellschaft nach den jeweils steuerllch als Aufwandersatz anerkannten Hochstsatzen berechtlgt. Tagesgelder geblihren nach dem jewells steuerlich als Aut\¥andersatz anerl<annten Hochstsatzen. 6. 6.1. 3 Urlaub Der Urlaubsanspruch des Vor.standsmltglledes fichtet skh grundsatzlich nach den Bestimmungen des Urlaubsgesetzes In der Jew lls g ltenden Fa$'1iUng mit der MaBgabe, dass der Zeltpunkt des Urlaubsantrlttes mit den and ren Vorstandsmltgliedern und gegebenenfalls mit dem Aufslchtsratsvorsitzenden abwstlmmen 1st. 7. Wettbewerbsverbot, Nebentatigkelten, Konkurrenz.klausel 7.1. Fur das Vorstandsmitglied gilt das Wettbewerbsverbot des § 79 AktG. Darilber hinaus ist es dem Vorstandsmitglled verwehrt, slch an elner anderen Handelsgesellschaft oder Gesellschaft im Geschaftszwelg der Gesellschaft als Gesellschafter weder dlrekt noch lndlrekt zu betelllgen. 7.2. lm Faile elnes VerstoBes gegen das In 7.1. gere.gelta Wettbewed:.sverbot kann dle Gesellschaft Schadenersatz fordern oder stattdessen vom VofstandsmitgtiJ d rlangen, das.s er die fUr elger·lP. Rechnung gemachten Geschafte als fOr Rechnung der·GeseiiSd'laft elngegangen gelten tasse un.d dass das Vorstandsmltglled den Gesch!iften ftlr fremde Rechnung bezogene Vergutung E::: ! ; ; ;abtrete. 7 .3. Ober das bezeichnete ; Einwllligung der Gesellschaft, die der Aufslchtsrat 11rh,rlftlltrh einer anderen Tatigkelt als der durch dlesen Anstellungsvertrag geregelten, untersagt, selbst wenn dlese Tiitlgkelt nlcht konkurrlerend 1st. Der Aufslchtsrat hat die schrlftllche Elnwllligung zu erteilen, wenn durch die Titlgkelt weder die lnteressen der Gesellschaft noch die Elnsatzf hlgkelt des Der bloBe Erwerb einer Betelllgung 7.4. Das Vorstandsmltglied verpflichtet sfch, nach Beendlgung des Anstellungsvertrages durch ein Jahr lnnerhalb Osterrelchs im Geschiifts;,twelg der Gesellschaft weder selbststandlg noch unselbststandlg tatig zu seln, sofern dlese Tiitlgkeit die Forschung und Entwicklung von Arznelmlt ln fUr lndikationen betrlfft, fOr die auch AOP Forschungs und Entwlcklungsarbelt dlrekt ode r lndlrekt durch.fuhrte oder durchfUhrt. FOr den Fall der. Verletzung dleser Verelnbarung verpfttchtet slch das V¢rstandsmitglled zur Zahlung elner Vertragsstrafe In Hohe von 4 Monatsgehliltern, wobei der Bezug im letzten Monat dleses Anstellungsvertrages die Bemessungsgrundlage bitdet. Elne allenfalls ausgezahlte Gratiflkatlon 1st in diese Bemessungsgrundlage· elnzubeziehen. Dlese Verpflichtung besteht unabhangig vom Grund fUr die Beendigung des Anstellungsverhiiltnlsses. 8. Entgeltfortzahlung 8.1. Flir die Verhindcrung des Vorstandsmitgliedes an der l.eistung seiner Oicnste durch Krankheit oder Ungllicksfall gilt§ 8 Angestelltengesetz sinngemaB. 4 9. Verfall von AnsprUchen 9.1. Es wlrd vereinbart, dass das Vorstandsmitglied offene Ansprliche aus dem gegenstlindllchen Anstellungsverhaltnis bel sonstlgem Verfalllnnerhalb von 6 Monaten ab Flilligkeit bel der Gesellschaft schrlftllch geltend ·zu machen hat. Ansprilche aus dem Aufwandersatz fUr Dlenstrelsen mussen bel sonstlgem Verfalllnnerhalb von 3 Monaten ab Hilllgkelt schrlftllch gel tend gemacht werden. 9.2. Bel rechtzeltlger Geltendmachung blelben die generell maBgebllchen Verjahrungs bzw. Verfallsfrlsten gewahrt. 10. Abberufung, Riicktritt, Vertragsaufltisung 1 0.1. Der Aufslchtsrat ist berecht!gt, das Vorstandsmitglled bel Vorliegen elnes wichtigen Grundes vorzeitig von selnem Amt abzuberufen. Als wlchtlge Grllnde gelten Insbesondere, jedoch nicht ausschlleBiich, grobe Pftlchtverletzung, Unffihlgkelt zur ordnungsgem!iBen GeschaftsfOhrung und Entziehung des Vertrauens durch die Hauptversammlung. 1 0.2. Der Aufslchtsrat der Gesellschaft wlrd splitestens sechs Monate vor Ablauf der Bestelldauer tlber die Verllingerung des Anstellungsvertrages des Vorstandsmitglledes entscheiden. Hat der Aufsichtsrat nlcht mlndestens sechs Monate vor Ablauf der Bestelldauer durch Beschluss entschieden, so wlrd die Gesellschaft unbeschadet der dem Vorstandsmltglied nach dlesem Vertrag sonst zustehenden Ansprtiche die gemliB dlesem Vertrag gebllbrenden Bezi:ige fur die Dauer von sechs Monaten nach Ablauf der Mandatspertode welter auszahlen. · 1 0.3. Das Vorstandsrnltglied ist berechtlgt, unter Elnhaltung elner sechsmonatlgen Kllndlgungsfrist zum Monatsletzten von der Vorstandsfunktlon zurllckzutreten und diesen Anstellungsvertrag zu kOndlgen. Bei Vorltegen elnes wlchtlgen Grundes ist das Vorstandsmltglled berechtigt, von der Vorstandsfunktlon zuriickzutreten und den Anstellungsvertrag jederzeit ohne Elnhaltung einer frist zu kiindlgen. t 0.4. Bel Beendlgung der Vorstandsfunktlon ist die Gesellschaft berechtlgt, dlesen Anstellungsvertrag unter Elnhaltung einer sechsmonatlgen Frlst zum Monatsletzten aufzulosen. Bei Vorllegen elnes wichtlgen Grundes 1st die Gesellschaft berechtlgt, den Anstellungsvertrag jederzeit ohne Einhaltung elner Frlst zu kilndlgen. 11, Schlussbestimmungen 11.1. Ourch dlesen Vertrag werden die schuldrechtllchen Beziehungen der Vertragspartner abschlleBend geregelt. 11.2.. Anderungen UF.ld.Erganzungen dleses Vertrages bedurfen zu ihrer Wirksamkeit der Schriftform. Auch fiir das Abgehen von dlesem Formerfordernis ist die Schrlftform notwendig. 11.3. Die Unwlrksamkeit einzelner Bestimmungen dieses Vertrages berOhrt die Wirksamkeit der ilbrlgen Bestlmmungen nicht. Sofern eine Klausel dleses Vertrages mangelhaft sein sollte, insbesondere well sie gegen geltendes Recht verstoBt oder undurchfuhrbar ist, verpfllchten slch die Parteien dazu, umgehend die mangelhafte Klausel durch eine miingelfreie, sohin durch elne insbesondere rechtsgiiltige und durchfllhrbare Klausel zu ers tzen, die der ursprunglichen Klausel in wirtschaftllcher Hinslcht m<lglichst nahe kommt. 11.4. 11.5. 5 Dleser Vertr.ag wlrd in zwei Originalen errichtet, von welchen jeder der Vertragspartner elnes erhalt. Samtllche Streltlgkelten aus diesem Vertrag elnschlleBIIch der Fragen seines giiltlgen Zustandekommens bzw. seiner Nachwlrkungen werden ausschlleRiich vor dem Handelsgericht Wien entschleden. Es gilt ausschlleBilch osterrelchlsches Recht. Mag. Andreas Steiner, geb. 09.12.1959 .. ANSTEllUNGSVERTRAG abgesqhlossen zvvischen AOP ,p n PharfuaQeutiCt:llS Aktiengesellschatl Wllbelminenslraf3e 91' t1160 Wlen ,,. · (im Folgenden ,Gesellschaft" genannt) einerseits und Or. Georg Fischer, geboren am 12.11.1958 Silenegasse 8 12/46, 1220 Wien (lm Folgendan ,Vorstandsmitglled" genannt) andererseits 1. Bestellung und Anstellung ' ·c,.· '1.1. Oar Aufsichtsrat der Gesellschaft hat Harrn Dr. Fischer mit Beschluss vom 27.06.2008 zum Vorstandsmitglied bestellt. Dar mit dieser Bestellung zeitglelch abgeschlossene auf zwei Jahre befristete Anstellungsvertrag endet am 30.06.2010. 1 .2. Dar gegenstandliche Anstellungsvertrag gilt nun fOr die restliche Bestelldauer von 5 (fOnf) Jahren, das heiBt bis zum 26.06.2015, und endet, soweit in der Folge nichts anderes vareinbart wird, nach Ablauf dieser 5 (fi.inf) Jahre, ohne dass es einer KOndigung bedarf. Dar Anstellungsvertrag regelt die vertraglichen Beziehungen zwischen dem Vorstandsmitglied und dar Gesellschaft. 2. Tatlgkeitsberelch, Rechtsgrund1agen 2.1. Das Vorstandsmitglied ist kollektiv mit dem Vorstandsvorsitzenden, Herrn Dr. Rudolf Widmann, oder mit dem Vorstandsvorsitzenden und einem weiteren Vorstandsmitgiied zur Vertretung der Gesellschaft und zur Zeichnung berechtigt (Gesamtvertretungsbefugnis). Der Aufsichtsrat l<ann die Vertretungsbefugnisse des Vorstandsmitgliedes durch Beschluss beschranken. Verantwortung im Rahmen der 2 und Empfehlungen, wie insbesondere die der Euro; ean Medicines Agency (EMeA) und der International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), verantwortlich. 2.3. Das Vorstandsmitglied hat die ihm ubertragenen Aufgaben mit der Sorgfalt eines ordentlichen und gewissenhaften Geschaftsleiters wahrzunehmen. Grundlagen tQr die Tatigkeit des Vorstandsmitgliedes sind die Bestimmungen des Aktiengesetzes, die dem Vorstandsmitglied bekannte Satzuqg der Gesellschaft, die verbindlichen BeschlOsse des Aufsiohtsrates der Gesellschaft, .die Geschaftsordnung tor den Vorstar ...:, sowie der gegenstandliche Anstellungsvertrag. Verletzungen der in diesen Grundlagen festgehaltenen Verpflichtungen konnen Schadenersatzanspruche der Gesellschaft begrOnden. 3. Geheimhaltungspflicht 3. i . Das Vorstandsmitglied ist verpflichtet, Qber vertrauliche Angaben Stlllschweigen zu bewahren und samtliche Geschafts und Betriebsgeheimnisse der Gesellschaft gegenOber jedem Dritten und, wenn dies lm Interesse der Gesellschaft geboten erscheint, auch gegenuber AngehOrigen der Gesellschaft, soweit diese nicht Kratt ihrer dienstlichen Funktion und Tatigkeit zur Entgegennahme·derartiger Mitteilungen befugt sind, geheim zu halten. Als Geschafts und Betriebsgeheimnlsse sind ohne Rucksicht auf Patentierfahigkeit oder sonstige rechtliche Schutzfahigkeit insbesondere aile im Zuge der dienstlichen Tatigkeit des Vorstandsmitgliedes von der Gesellschaft ausgel!bten Forschungs und Entwicklun.gstatigkeiten und dadurch direkt oder i'1direkt gewonnenen Er\. 3bnisse, samtliche durchgefOhrten Studien, sowie aile fOr die Gesellschaft gewonnenen Arbeitsgeheimnisse, Erfahrungen und sonstigen Erkenntnisse ..::u verstehen, deren Geheimhaltung nach dem pflichtgemaBen Ermessen eines gewissenhaften Geschaftsleiters als geheimhaltungsbedurftrg nzusehen sind und/oder deren Geheimhaltung aus anderen Grunden lm Interesse der Gesellschaft geboten erscheint. 4. Entgelt 4.1. Das Vorstandsmitglied erMit' als Entgelt fOr seine gesamte Tatigkeit im Diensta der Gesellscllaft einen jahrlichen Bruttobezug von © Dr. Veronil<· Cortolezis "3 4.2. Die regelmaBige wochentliche Normalarbeitszeit betragt derzeit 38,5 Stunden. Die Einteilung der taglichen Arbeitszeit obliegt dem Vorstandsmitglied. Eine Qber diese Arbeitszeit hinausgehende Arbeitsleistung gilt als Uberstundenleistung. Das Vorstandmitglied wird Oberstunden leisten, soweit dies fOr die ErfOIIung seiner Aufgaben erforderlich ist und dem berucksichtigungswOrdige lnteressen seinerseits nicht entgegenstehen. 4.3. Das Vorstandsmitglied wird Aufzeichnungen uber die von ihm geleisteten Oberstunden fOhren und dlese Aufzeichnungen jeweils bis zum 5. des Folgemonats fOr das vorangehende Monat der Gesellschaft Obermitteln. Ein allfalliges Entgelt fOr geleistete Oberstunden (Grundlohn und SFN·Zuschlage) wird am Ende jenes Monats zur Zahlung fallig, der dem Monat folgt, in dem diese Oberstunden geleistet wurden. Oberstunden werden im AusmaB von 1 o Stunden pro Monat abgegolten. 4.4. Zusatzlich zu dem im Absatz 4.1. geregelten Bezug erhiilt das Vorstandsmitglied jahrlich eine Gratifikatlon, deren Hohe vom Aufsichtsrat jahrlich fOr das kommende Geschattsjahr nach MaBgabe des Geschaftserfolges und der personlichen Leistung des Vorstandsmltglledes festgelegt wlrd. Die Gratiflkation betragt derzeit € 24.878,80 (in Worten: Euro Vierundzwanzlgtausendachthundertaohtttndslebzlg/80) und wlrd lm ersten Quartal des darauffolgenden Geschaftsjahres ausgezahlt. 4.5. Welters erhalt das Vorstandsmitglied zusatzlich zu dem im Absatz 4.1. geregelten Bezug eine Zuwendung zu seinem Pensionskassenvertrag Nr. 2211 bel der Victoria· Volksbanken Pensionskassen AG in Hohe ...... 4.6. Dam Vorstandsmitglied wird fOr die Dauer seiner Tatlgkeit ein Dienstfahrzeug zur VerfOgung gestellt. FOr die Beni.itzung dieses Dienstfahrzeuges gelten die Bestimmungen de ;s Dienstfahrzeuges in ihrer jeweils gOitigen Fassung. Das Gesellschaft aktuellen Gegebenheiten 5. Oienstreisen 5.i. lm Faile der Verwendung seines Privatfahrzeuges fOr dienstliche Zwecke ist das Vorstandsmitglied zur Verrechnung von Kilometergeld an die Gesellschaft nach den jeweils steuerlich als Aufwandersatz anerkannten Hochstsatzen berechtigt. Tagesgelder gebOhren nach dem jeweils steuerlich als Aufwandersatz anerkannten Hochstsatzen. ©Dr. Veronika Cortolezis 4 6., .,,. Urlaub · · · 'jdB. (;,::" t:'f5er · tl lJb,anspnJG"". di ;; Vorstand mltgiietf . ridit f· steff grunds tilfch ·naof derf Bestimmungen des Urlaubsgesetzes in der jeweils geltenden Fassung mit der MaBgabe, dass der Zeitpunkt des Urlaubsantrittes mit den anderen Vorstandsmitgliedern und gegebenenfalls mit dem Aufsichtsratsvorsitzenden abzustimmen ist. 7. Wettbewerbsverbot, Nebentatigkeiten, Konkurrenzklausel 7.1. FOr das Vorstandsmitglied gilt das Wettbewarbsverbot des§ 79 AktG. DarOber hinaus ist es dem Vorstand verwehrt, sich an einer konkurrierenden Handelsgesallschaft oder Gesallschaft im GeschiUtszwelg dar Gesallschaft als Gesellschafter zu batailigan. 7.2. lm Faile alnes VerstoBes gegen das In 7.1. geregelte Wettbewerbsverbot kann die Gesallschaft vom Vorstandsmltglied Schadenersatz fordern oder sta dassan von Jhm verlangen, dass er die fOr eigene Rechnung gemachten Geschafte als fOr Rechnung der Gesellschaft elngagangen gelten lasse und dass das Vorstandsmitglied die aus den Geschaften fur fremde Rechnung bezogene VergOtung herausgebe oder seinan Anspruch auf die VargOtung an die Gesellschaft abtreta. 7 · ('14 hinaus ist dem Vorstandsmitgliad ohne · · 1 d. d A f · h 1e er u SIC . ·· · n at, die AusObung einer anderen Tatigkeit als dar durch diasen rs · · · , gereg en, untersagt, salbst wenn diese Tatigkeit nicht konkurrierend ir . Der Aufsicht ;rat hat die Einwilligung zu erteilen, wenn durch die Hitigkeit wader die lnteressen der Gesellschaft noch die Einsatzfahigkeit des Vorstandsmitgliedes fUr die Gasallschaft gefahrdet erscheinan. Der bloBe Erwarb einer Beteiligung von1··· 7.4. Dem Vorstand.smitglied ist es ausdrOcklich gestattet, tor ein nicht konkurrierendes Pharmaunternehmen als Konzasslonar zu fungieren. 8. 8.i. EntgeJtfortzahlung Fur die Verhinderung des Vorstandsmitgliedes an der Leistung seiner Dienste durch Krankheit oder UngiOcksfall gilt § B Angestelltengesetz sinngemaB ©Dr. Veronlka Cortolezis ·5· 9. Verfall von Anspri.ichen 9.1. Es wird vereinbart, dass das Vorstandsmitglied offene AnsprOche aus dem gegenstiimdlichen Anstellungsverhaltnis bei sonstigem Verfall innerhalb von 6 (sechs) Monaten ab Falligkeit bei der Gesellschaft schriftlich geltend zu machen hat. AnsprCiche aus dem Aufwandersatz fur Dienstreisen mussen bei sonstigem Verfall innerhalb von 3 (drei) Monaten ab Falligkeit schriftlich geltend gemacht warden. 9.2. Bei rechtzeitiger Geltendmachung bleiben die generell maBgeblichen Verjahrungs bzw. Verfallsfristen gewahrt. 1 o. Abberufung, Rucktritt, Vertragsauflosung 10.1. Der Aufsichtsrat ist berechtigt, das Vorstandsmitglied bei Vorllegen eines wichtigen Grundes vorzeitig von seinem Amt abzuberufen. Als wichtige GrOnde gelten insbesondere, jedoch nlcht ausschlieBiich, grobe Pflichtverletzung, Unfahigkeit zur ordnungsgemaBen GeschaftsfOhrung und Entziehung des Vertrauens duroh die Hauptversammlung. 1 0.2. Der Aufsichtsrat der Gesellschaft wird spatestens 3 (drei) Monate vor Ablauf der Bestelldauer Ober die Verlangerung des Anstellungsvertrages des Vorstandsmitgliedes entscheiden. Hat der Aufsichtsrat nicht mindestens 3 (drel) Monate vor Ablauf der Bestelldauer durch Beschluss entschieden, so wird die Gesellschaft unbeschadet der dem Vorstandsmitglied nach diesem Vertrag sonst zustehenden AnsprOche die gemaB diesem Vertrag gebuhrenden Bezuge fOr die Dauer von 3 (drei) Monaten nach Ablaut der Mandatsperiode weiter auszahlen. 1 0.3. Das Vorstandsmitglied ist berechtigt, unter Einhaltung einer sechsmonatigen KOndigungsfrist zum Monatsletzten von der Vorstandsfunktion z.urOckzutreten und diesen Anstellungsvertrag zu kOndigen. i 0.4. Bei Beendigung der Vorstandsfunktion ist die Gesellschaft berechtigt, diesen Anstellungsvertrag unter Einhaltung einer sechsmonatigen Frist zum Monatsletzten aufzul6sen. Bei Vorliegen eines wichtigen Grundes ist die Gesellschaft berechtigt, den Anstellungsvertrag jederzeit ohne Einhaltung einer Frist zu kOndigen. ©Dr. Veronika Cortolezis 6 11. Schlussbestimmungen 11.1. Durch dies n Vert.rag werden die schuldrechtlichen Beziehungen der Vertragspartner abschliel3end geregelt. 11.2. Anderungen und Erganzungen dieses Vertrages bedOrfen zu ihrer Wirksamkeit der Schriftform. Auch tor das Abgehen von diesem Formerfordernis ist die Schriftform notwendig. 11.3. Die Unwirksamkeit elnzelner Bestimmungen dieses Vertrages berOhrt die Wirksamkeit der Obrigen Bestimmungen nicht. Sofern eine Klausel dieses Vertrages mangelhaft sein sollte, insbesondere wail sle gegen geltendes Recht verstOBt oder undurchfOhrbar ist, verpflichten sich die Partelen dazu, umgehend die mangelhafte Klausel durch eine mA.ngelfreie, sohin durch eine insbesondere rechtsgOitige und durchfOhrbare Klausel zu ersetzen, die dar ursprOnglichen Klausel in wirtschaftlicher Hinsicht mOglichst nahe kommt. 11.4. Dieser Vertrag wird in zwei Originalen errichtet, von welchen jeder der Vertragspartner elnes erhalt. 11.5. Samtllche Streitigkeiten aus dlesem Vertrag einschlieBiich der Fragen seines gOitigen Zustandekommens bzw: seiner Nachwirkungen werden ausschlleBiich vor dem Handelsgericht Wien entschieden. Es gilt ausschlieBiich Osterrelohisches Recht. Wien,am fUr AOP Orphan Pharmaceuticals Aktiengesel s ft: DDr. H inz . Ru swurrn · too \t . Dr. Georg Fischer,·geb. 12.11.1958 © Dr. Veronika Cortolezis Vorstandsdienstvertrag 1. ANSTELLUNG Herr wohnhaft in Dr. Rudolf Stefan Widmann FavoritenstraBe 25/4/8, 1 040 Wien geboren am 24.03.57 in St. Johann in Tirol, osterreichische Staatsburger, verheiratet, im folgenden Dr. Widmann genannt, tritt mit 1. Janner 1997 in den Dienst der Firma AOP Orphan Pharmaceuticals AG, FavoritenstraBe 25/4/8, 1040 Wien, im folgenden kurz AOP genannt. 2. AUFGABEN Dr. Widmann wird mit der FOhrung der Gesellschaft als Vorstand betraut. Dr. Widmann wird alles dEiransetzen, die Geseltschaft erfolgreich auf, ml!l. u fQr s.e,ine Tltlgkeit wird etn Na. ran. Zusa l i ng wlrd ein Betrat zur Finanzlerung einer Vetsf¢1!\erung leii'IEJr W.afitl. geWj\hrt, sofem dies auch · einel\'l stetaedicher Aufwand fur die AOF' sieh · Dle$er Betrag wlijl jihrlich im gleichen AusmaB wle dar Die ZEtf:IIMnQ$n erfol.$0 jewells am. Lc:ttz:ten eines jedefl Kalenderm(lnates im t1Elchhinein. GrundsttZildh hat or. Widmann sefh G halt baw. aile sonsttten aus clem Vertrag resultlerenden ffnanzielten AnsprOche, wie etwa Weihnachtsremuneratlon, Unaubsbelhitfe und andere& mehr, bel AOP bzw. dessen Beauftragten zu beheben, wobei auf WUnsch des Dr. Widmann dlese Zahlungnn auch auf eln von ihm bekanntg,egebenes Konto durchgefnhrt warden. 4. ARBEITSZEIT/DIATENREGELUNG Die regelmi:iBige wochentliche Arbeltszeit beW\gt 40 Stunden. Bei mehr als 4 stundlger elnem Termin auBerhalb Wiens gelangen Diaten zur Bei Auslandsterminen wird der entsprechende 5. MEHRARBEITSSTUNDEN UNO OBERSTUNDEN Dr. Widmann ist verpflichtet, Mehrarbeitsstunden und/oder Oberstunden, die fUr die Erreichung der Untemehmensziele notwendig sind, in vertretbarem AusmaBe zu leisten. Diese werden jedoch von AOP nicht extra honoriert, sondem gelten durch die im Absatz 2 des vorliegenden Vertrages definierte Oberstundenpauschale als abgegolten. 6. SONSTIGE AKTIVITATEN Dr. Widmann ist ausschlieBiich fUr AOP tatig. Etwaige anderen Aktivitaten sind vor deren Aufnahme dam Aufsichtsrat zur Genehmigung vorzulegen. 7. URLAUB Der Urlaubsanspruch betragt 5 Wochen. 8. LAUFZEIT/KONDIGUNG Das Dienstverhaltnis wird auf unbestimmte Zeit abgeschlossen. Das Dienstverhaltnis kann von beiden Seiten unter Einhaltung elner 6 monatigen Kundigungsfrist am jeweils letzten Tag eines Kalendermonats beendet warden. 9. Wenn Dr. Widmann ohne 5I liD wenn ihm eln Verschulden an auf Ersatz des von ihm ve ans zu. Dleser allfilllge Schadenersatzanspruch wird ohne Rucksicht auf den tats#lchlich entstehenden Schadan im belderseltigen ausdrOckllchen Elnvemehmen pauschaliert, und zwar so, daB Dr. Widmann im Faile eines vorzeitlgen Austrittes ohne wichtigen Grund oder einer durch ihm verschuldeten vorzeltigen Entl i lft08«1t'KIJWI1.fli1Wfl dll ;U 0 Pten Betrag schuldet, den AOP bel einem, von thm vef!leliitaelen vorzeitlgen Austtitt an r 3 Monatsentgelte anerkennt vereinbarten Konventlonalstrafe. Die Dienstverhiltnisses filllg. 10. GEHEIMHALTUNGSKLAUSEL Dr. Widmann verpflichtet sich, sowohl wahrend des aufrechten Dienstverhaltnisses als auch Ober elnen Zeitraurn von mindestens drei Jahren nach Aufl6sung des Dienstverhaltnisses, samtliche ihm im Rahmen der Tllltigkeit fur AOP zugangllch gewordenen Firmenstrategien, ln!bnnatlonen Ober Bezugs oder Verkaufspreise, Bezugsquellen, Kooperatlonen sowie aile &onstigen Daten, die fOr eine Kon'kurrenzflrma von Interesse sein konnten, geheim zu halten und nicht an Dritte weiterzugeben. Sollte es dennoch zu einer Weitergabe solcher lnformationen an Drltte wahrend des angegebenen Zeitraumes kommen, haftet die Dr. Widmann ausdrilcklich fOr aile Folgeschaden, die AOP daraus erwachsen. 11. VERFALL VON ANSPROCHEN Es wird vereinbart, daB offene Anspruche aus dem gage sonstigem Verfall ir.nerhalb von 3 Monaten ab FEUJigkelt b warden mussen. Bei rechtzeitiger Geltendmachung bleiben di s bei acht generell maBgeblichen Verjahrungs bzw. Verfallfristen gewahrt. Dr. Widmann: GareseJAL Dr. Rudolf Widmann Wien, am 3. Februar 1997 2 AMENDMENT 01 zum Vorstandsdienstvertrag vom 03.02.1997 abgeschlossen zwischen Dr. Rudolf Widmann (im folgenden Vorstand genannt) und AOP Orphan Pharmaceuticals AG (im folgenden AOP Orphan genannt) Priam bel Dr. Rudolf Widmann und AOP Orphan haben am 03. Februar 1997 einen Vorstandsdienstvertrag abgeschlossen. Die Parteien kon:tmen nunmehr Oberein, den Vertrag wie folgt zu erganzen: 1. Elnstufung Zufolge der vereinbarten Tatigkeit und der von dem Vorstand tiekannt gegebenen Berufsjahre werden Vordienstzeiten im AusmaS von 8 Berufsjahren fOr Tatigkeiten vor BegrOndung des Dienstverhaltnisses mit AOP Orphan angerechnet. 2. Allgemelnes Die Obrigen Bestimmungen des Vorstandsdienstvertrags bleiben von dieser Vereinbarung unberOhrt. Wien, am 1 o. Marz 1998 Dr. Rudolf Widmann DDr. Heinz Russwurm Aufsichtsratsvorsitzender AMENDMENT 02 zum Vorstandsdienstvertrag vom 03.02.1997 und :z:um Amendment 01 vom 10. Mlr:z: 1998 abgeschlossen zwischen Dr. Rudolf Widmann (im folgenden Vorstand genannt) und AOIP Orphan Pharmaceuticals AG (im folgenden AOP Orphan genannt) Priam bel Dr. Rudolf Widmann und AOP Orphan haben einen Vorstandsdienstvertrag und ein Amendment Ol abgeschlossen. Im Amendment 01 wurde vereinbart, Vordlenstzelten lm AusmaB von 8 Berufsjahren anzurechnen. Die Parteien halten nunmehr fest, dass die Berechnung der 8 Berufsjahre inkorrekt erfolgt 1st und die Berechnung wie folgt richtiggestellt wird: 1. Einstufung Zufolge der vereinbarten Tatigkeit und der von dem Vorstand bekannt gegebenen Berufsjahre werden unter Bezugnahme auf EStG § 14 Abs 1/ZI 3 Vordlenstzeiten im AusmaB von 13 Berufsjahren fur Tatlgkelten vor BegrOndung des Dienstverhaltnisses mit AOP Orphan angerechnet. 2. Allgemelnes .Ole Obrigen Bestlmmungen des Vorstandsdienstvertrags blelben von dieser Vereinbarung unberuhrt. Wien, am 11. August 2008 Dr. Rudolf Widmann· DDr. Heinz Russwurm Aufsichtsratsvorsltzender Exhibit No: 5.27.3 See addendum Product: Britlofex no exclusivity MAH Product: Busilvex Orphan drug status ends 2014 I Country Austria MAH Pierre Fabre Slovenia Pierre Fabre Hungary Pierre Fabre Product: IVHebex no exclusivity Country MAH 1 Poland LFB Product: Canemes no exclusivity Orpha Devel country Aust iMAH Product: Ospolot no exclusivity Country MAH Austria Desitin Product: Tadim, Promixin IV, Tadim Inhale no exclusivity Country MAH Austria Profile Product: Wilfactin, Hemoleven no exclusivity Product: Adasuve Staccato 10 year data exclusivity starting 2013 Country MAH Austria Alexza Czech Republic Alexza Poland Alexza Slovakia Alexza Baltics Alexza Hungary Alexza Slovenia Alexza Exhibit No: 5.33.6 Title: List of representative offices and area managers See addendum Exhibit 8.2.1 1 Definitions 1.1 UnlesS' expressly ot'fleFwise defined in Clause 1.2· of ·this Exhibit 8.2.1, d fined·" terms used herein shall have the meaning given to them in Clause 1 (Definitions) of the Agreement. 1.2 In addition, the following defined terms shall have the following meaning: Cash Closing Accounts Closing Accounts Date Dividend Instalment Due Date Marketable Securities Provisions M6244142/9 means cash, cheques and bank deposits (Kassenbestand, Schecks, Guthaben bei Kreditinstituten) pursuant to § 224 para (2) B. IV. UGB; means unaudited financial statements of the Company as of the Closing Accounts Date, comprising balance sheet and P&L account, prepared in the format of the Audited Financial Statements 2012 and in accordance with Austrian generally accepted accounting principles, including without limitation the principle of balance sheet continuity; means 30 June 2013; means 30 April, 31 July, 31 October and 31 January of a calendar year or, if these dates are not Business Days, the Business Day immediately following such date(s); means securities of the fixed assets ( Wertpapiere (Wertrechte) des Anlagevermogens) pursuant to § 224 para 2 A. III. 5. UGB and other securities and participations (sonstige Wertpapiere und Anteile) pursuant to § 224 para 2 B. III. 2 UGB; means prov1s1ons for severance payments, pensions, taxes and other provisions (ROckstelfungen fOr Abfertigungen, Pensionen, Steuerrilckstellungen und sonstige ROckstelfungen) pursuant to § 224 para (3) C. 1. through 4. UGB; 2 Working Capital 2 Dividend to Seller 1 Payment 2.1 Subject to the terms and conditions of the Agreement and this Exhibit 8.2.1, the Seller shall be entitled to a dividend by the Company as follows: 2.1.1 The Seller shall be entitled to a dividend by the Company corresponding to the amount of the Working Capital pursuant to the Closing Accounts minus an amount (the "Dividend"). The dividend resolution shall provide for the payment of the Dividend in instalments pursuant to Clause 2.1.11. 2.1.2 To determine the amount of the Dividend, the Seller shall procure that the Company draws up and prepares the Closing Accounts as of the Closing Accounts Date. The Closing Accounts shall inter alia present the Working Capital as of the Closing Accounts Date. The Closing Accounts shall be delivered to the Purchasers within 30 (thirty) business days from the Closing Accounts Date. The Closing Accounts shall be audited Purchaser 2's auditor or such other .international ac :ounting firm as the Purchaser may reasonably select within 4 (four) weeks from receipt of the Closing Accounts by the auditor and, upon completion of such audit, the audited Closing Accounts shall be submitted to the Seller and the Purchasers. 2.1.3 2.1.4 M6244142/9 Within 14 (fourteen) days following receipt by the Seller of the audited Closing Accounts, the Seller shall be entitled to review and examine the audited Closing Accounts. The Closing Accounts prepared, reviewed and audited in accordance with the provisions of this Clause 2.1 will be final and binding upon the Parties, unless a written notice of disagreement with respect thereto (a "Notice of Disagreement"), specifying in reasonable detail the nature and reasons of such disagreement, is delivered by the Seller to the Purchasers within 14 (fourteen) days following the date on which the audited Closing Accounts were delivered to the Seller. If a Notice of Disagreement is given, during a period of 30 (thirty) days following the delivery of such notice, Purchasers and Seller will attempt in good faith to resolve by mutual agreement any difference they may have with respect to any matter specified in such Notice of Disagreement and, to the extent they will be able to amicably resolve such differences, they will agree also on the adjustment( s) to be made to the audited Closing Accounts to reflect such mutual agreement. 2.1.5 If Seller and Purchasers fail to reach an agreement with respect to all matters specified in the Notice of Disagreement within such period of 30 (thirty) days, then all matters as to which agreement is not reached (the "Disputed Matters") will be submitted to and reviewed by an expert (the "Expert"), whictlshall be any reputable and leading international accounting firm {other than the Company's or the Purchasers' auditor), jointly selected by Purchasers and Seller. If, within 7 (seven) days following the expiration of the 30 (thirty) day period contemplated above, Purchasers and Seller fail to agree upon the selection of the Expert, or any Expert selected by them does not agree to perform the services called for hereunder, and in the latter case only the Parties cannot agree on an alternate Expert within a further period of 7 (seven) days, the Expert shall be selected by the then President of the Austrian Chamber of Accountants, Vienna (Kammer der Wirtschaftstreuhander), with preference being given in making such selection to any leading international accounting firm qualified to render audit services in Austria (other than the Company's or the Purchasers' auditor) which may be agreeable to perform such services. 2.1.6 2.1.7 M6244142/9 The Expert {I) shall consider ·only the Disputed Matters, (ii) shall act promptly to resolve all such matters, (iii) shall comply with the provisions of this Clause 2.1, (iv) shall be empowered to act as an expert (Schiedsgutachter) and not as an arbitrator only to the extent strictly required to resolve the Disputed Matters and (v) its determinations with respect thereto shall be fi,nal, conclusive and binding upon Purchasers and Seller and shall not be subject to appeal. The application of § 1056 ABGB is hereby excluded by the Parties. Upon resolution by the Expert of all Disputed Matters, the Expert shall prepare and deliver to the Parties, as soon as possible and in any event not later than 30 (thirty) days following its acceptance of the appointment, the audited Closing Accounts and the amount of the Working Capital, adjusted to reflect any determinations of the Expert with respect to any Disputed Matters. The audited Closing Accounts in respect of which no Notice of Disagreement has been notified by the Seller to the Purchasers or which have been adjusted as stated. above (to reflect any agreement reached between the Parties with respect to matters specified in a Notice of Disagreement and/or pursuant to any determination of the Expert with respect to any Disputed Matters) shall be final, conclusive and binding upon the Purchasers and the Seller solely for the purpose of determining the amount of the Dividend and, accordingly, shall not, per se, affect the annual financial "4. statements of the Company to be submitted to shareholders' approval and to be published ·and filed In accordance with applicable taxes; . 2.1.'8··· Neither thEf"acnieVement of ·any 'mutua'l greern.en(oi the · Pardes ·ps sef forth above, nor any determinations by the Expert may be deemed or otherwise construed to limit any rights of the Purchasers under or in connection with the Agreement. All fees and disbursements of the Expert due in connection with the provision of the services contemplated hereunder shall be shared between the Seller and the Purchaser:. in the proportion they have prevailed in or lost their case, thereby applying the provisions of Sections 41 et seq. of the Austrian Code of Civil Procedure (Zivilprozessordnung) by analogy. It is also possible for either Party to prevail in or lose its case entirety. In his Expert opinion, the Expert will also decide in which proportion the Parties shall bear the Expert's fees. The Parties shall be liable for the Expert's fees in this proportion. 2.1.9 Within 30 (thirty) days after the audited Closing Accounts have become final and binding on the Parties in accordance with this Clause 2.1, a shareholders' meeting of the Company shall be held to resolve on the distribution of the Dividend to the Seller as former shareholder of the Company. 2.1.10 Following the resolution of the Dividend pursuant to Clause 2.1.9, the Company shall pay the dividend withholding tax (KESt). 2.1.11 Subject to Clause 2.1.12 of this Exhibit 8.2.1, the Dividend shall b.e payable to the Seller by the Company in quarterly instalments on each Dividend p.stalment Due Date, beginning as of 30 September 2013. 2.1.12 Each instalment of the Dividend shall be due anc. payable by the Company 20 (twenty) Business Days after each Dividend Instalment Due Date only if and to the extent: 2.1.12.1 M6244142/9 Within 5 (five) Business Days from each Dividend Instalment Due Date, the Company's auditor has certified in writing to the Company (for the attention of the supervisory board), with a copy to Purchaser 2 that (A) on the respective Dividend Instalment Due Com ny had Cash of not less than Ius such amount of Marketable Securities which are required to be held against Provisions under Austrian generally accepted accounting principles as at such Dividend Instalment Due Date; and (B) during the quarter immediately preceding the respective Dividend Instalment Due Date the Company had and during the quarter immediately following the respective Dividend Instalment Due Date is reasonably likely to maintain a normalized Working Capital of not In case. the minimum Cash balance pursuant to sub Cii=!USe (A) above is not met on the respective Dividend Instalment Due Date, payment of the Dividend instahrrent may still be made, prOVIded that·the Seller no ifies the Company's auditor (with a copy to Purchaser 2) in wr;lting on the payment date pursuant to Clause 2.1.12 that' the minimum Cash balance pursuant to sub clause (A) above is met on the payment date and provides (a) bank statement(s) (Kontoauszug) of the Company's bank account(s) of even date evidencing the correctness of such notice; 2.1.12.2 within 10 (ten) Business Days from receipt of the auditor certificate pursuant to Clause 2.1.12.1 of this Exhibit 8.2.1, the chairman of the supervisory board of the Company, respectively, in case of the chairman's incapacity, the deputy chairman has approved the payment of the respective instalment of the Dividend. No additional approval by the chairman (deputy chairman) of the supervisory board shall be required for payment of a Dividend instalment on the basis of Seller's notice to the auditor accompanied by (a) bank statement(s) (Kontoauszug) pursuant to Clause 2.1.12.1 second sentence; 2.1.12.3 on the payment date pursuant to Clause 2.1.12 of this Exhibit 8.2.1, the Company actually has Cash of not less than us such amount of Mare Securities which are red to be held against Provisions under Austrian generally accepted accounting principles and the Dividend can be paid without requiring loan or credit financing of any kind or nature; and 2.1.12.4 the Dividend shall have been paid in full on or before 31 December 2018. If not paid in full by this date, all of the Seller's rights and Claims to the Dividend not covered by a dividend instalment payment at an earlier Dividend Instalment Due Date shall be terminated and forfeited. 2.2 If and to the extent due, the Dividend shalt be payable by the Company to the Seller's bank account set forth in Exhibit 4.5 . .1 of the Agreement or such other bank account of the Seller as the Seller may from time to time notify to the Company (with a. copy to Purchaser 2) in writing pursuant to Clause 15.11 of the Agreement. 2.3 The Seller shall not be entitled to set off Claims pursuant to this Exhibit 8.2.1 against Claims of the Company against the Seller out of or in connection with the Agreement, if any. M6244142/9 Exhibit No: 1 0.4.1 See addendum G [Address] AOP Orphan Pharmaceuticals Aktiengesellschaft Attn: Management Board (Vorstand) Wilheminenstra6e 91/Ilf 1160 Vienna Austria Resignation as Member of the Supervisory Board Dear Sirs, SPA Exhibit 10.4.1 Vienna __ ·· · 2013 Herewith, I resign as member of the supervisory board of AOP Orphan Pharmaceuticals Aktiengesellschaft with corporate seat in Vienna and business address at WilheminenstraBe 91/IIf, 1160 Vienna, Austrfa, registered in the companies' register of the Vienna Commercial Court under FN 237770 m ("Company"). Such resignation shall be effective as of [Closing Date] . . I confirm that I have no claims towards the Company at the time of resignation. Kindly acknowledge my resignation as member of the supervisory board of the Company and file my resignation with the companies' register. I trust that my discharge (Entlastung) for the busin 'SS year ending on 31 December 2012 will be resolved upon in the upcoming annu<..l general meeting of the Company. Kind regards For AOP Orphan Pharmaceuticals AG Dr. Rudolf Widmann [Address] AOP Orphan Pharmaceuticals Aktiengesellschaft Attn: Management Board ( Vorstand) Wilhemim:. traBe 91/IIf 1160 Vienna Austria Resignation as Member of the Supervisory Board Dear Sirs, SPA Exhibit 10.4.1 Vienna · · 2013 Herewith, I resign as member of the supervisory board of AOP Orphan Pharmaceuticals Aktiengesellschaft with corporate seat in Vienna and business address at WilheminenstraBe 91/Ilf, 1160 Vienna, Austria, registered in the companies' register of the Vienna Commercial Court under FN 237770 m ("Company"). Such resignation shall be effective as of [Closing Date] . . I confirm that I have no claims towards the Company at the time of resignation. Kil"dly acknowledge my resignation as member of the supervisory board of the Company and file my resignation with the companies' register. I trust that my discharge (Ent/astung) for the business year ending on 31 December 2012 will be resolved upon in the upcoming annual general meeting of the Company. Kind regards For AOP Orphan Pharmaceuticals AG Received and acknowledged on __ . __ . 2013 Dr. Rudolf Widmann [Address] AOP Orphan Pharmaceuticals Aktiengesellschaft Attn: Management Board ( Vorstand) WilheminenstraBe 91/IIf 1160 Vienna Austria Resignation :.: s Member of the Supervisory Board Dear Sirs, SPA Exhibit 10.4.1 Vienna 2013 Herewith, I resign as member of the supervisory board of AOP Orphan Pharmaceuticals Aktiengesellschaft with corporate seat in Vienna and business address at WilheminenstraBe 91/IIf, 1160 Vienna, Austria, registered in the companies' register of the Vienna Commercial Court under FN 237770 m ("Company"). Such resignation shall be effective as of [Closing Date]. I confirm that I have no claims towards the Company at the time of resignation, Kindly acknowledge my resignation as member of the supervisory board of the Company and file my resignation with the companies' register. I trust that my discharge (Entlastung) for the business year ending on 31 December 2012 will be resolved upon in the upcoming annual general meeting of the Company. Kind regards For AOP Orphan Pharmaceuticals AG Rec iveda:od acknowledged on 2013 Dr. Rudolf Widmann Exhibit No: 1 0.4.2 Title Rights Assignment Agreement between Dr. Rudolf Widmann and AOP Orphan Pharmaceuticals Aktiengesellschaft See addendum Exhibit 1 0.4.2 ASSIGNMENT OF RIGHTS 1. Creations: Dr. Rudolf Widmann, born 24.3.1957, (hereinafter "Director") is acting and employed as director of AOP Orphan Pharmaceuticals AG, FN237770m, (hereinafter "AOP"). Director herewith confirms that all inventions and technical improvements, patentable or not, and all designs, discoveries, new or improved products, business methods, PR and advertising materials as well as other production and distribution methods and know how and other creative work which he created, developed or practically exploited during his term as director of AOP, alone or in co operation with third parties and which relate to AOP and its affiliates or the research and development activities of AOP and its affiliates or which result from the activities performed by the director for AOP (hereinafter "Creations") unrestrictedly and irrevocably belong to AOP. 2. Transfer of Rights to the Creations: Director expressly confirms and acknowledges herewith the in every respect comprehensive and irrevocable assignment/transfer of Rights to the Creations including but not limited to the Rights set forth in Exhibit ./1, to AOP. This assignment/transfer includes, but is not limited to, to the extent permissible under the applicable laws all rights of inventions or technological improvements which Director has made during his work for AOP or due to experiences made or work done by AOP. 3. Subsequent Transfer of Rights: To the ement that AOP has not already been comprehensively assigned the Rights to the Creation and Prior Creations as set forth above in Sec. 1 and 2, Director herewith explicitly assigns and grants such rights to AOP. Such assignment of rights shall be exclusive, thus also excluding any further rights of Director. 4. Scope of Transferred Rights: AOP therefore has in particular the irrevocable, exclusive, unrestricted as to time, content and territory (worldwide) rights to exploit all Rights to the Creations and Prlor Creations especially the right file applications for registered rights (in particular patents, utility patents, SPCs) to entire and/or partial publication, reproduction, dissemination, processing, translation, sending, intangible reproduction, making available to the public and any and all other use possible now or in the future (irrespective whether known today) as well as the right to exert the rights according to § 20 and 21 (1) Austrian Copyright Protection Act. Therefore, AOP is also entitled to assert all rights in Creations and Prior Creations, in particular to sue for any (past, present and/or future) infringements thereof and to collect for all (past, present and/or future) damages arising from such infringements. AOP is in particular also entitled to assign these rights entirely or partly to third parties, to grant sublicenses respectively rights of exploitation and/or exploitation permissions. AOP is not obliged to use the Creations and Prior Creations, in particular, § 29 Austrian Copyright Protection Act shall not apply. To the extent permissible under the applicable laws, Director waives the right to be named. 2 5. Assistance: Director is obliged to assist with procurement of rights (in particular patents) for the Creations and Prior Creations, in particular by duly executing any additional documents required for procurement of such registered right s. Insofar as any·intellectual property·fights (including, but. not limited to patents, utility patents, trademarks, designs, copyrights) owned 9r inlicensed by the Director necessary or useful for the current business of AOP ("Related IP Rights") have not already been assigned/transferred by the Assignment of Rights as set out in Sec. 1. to 5. above, Director grants AOP an irrevocable, non exclusive, unrestricted as to time, content and territory (worldwide) license in such Related IP Rights. 7. Confirmation of Power of Disposition: Director expressly confirms not to be restricted in the assignment of Rights hereunder by any prior assignment of rights to any third party and that no rights of thi:·d parties are in conflict with and/or prevent the assignment hereunder. 8. Full Compensation: Director expressly declares and confirms that this comprehensive transfer/assignment/licensing of Rights has· already been fully and finally compensated by the remuneration paid by AOP and/or Orpha to Director. Place, Date Signature Exhibit ./1 to Exhibit 10.4.2 Patent Family NOVEL COMPOSITION FOR TREATMENT OF ESSENTIAL THROMBOCYTHEMIA (International Publication Number WO 2010/063824), including, but not limited to Country /Organization Application Number Priority Date Austria AT7072008U (Utility Patent) ' PCT PCT /EP2009/066405········· 05.12.2008 Australia 2009324043 05.12.2008 Aw. China ··2oo980 156227.9 05.12.2008 Eurasia (EAPO) 201170583 05.12.2008 Europe (EPO) 09764826.5 05.12.2008 Egypt PCT/912/2011 05.12 2008 Hong Kong 11113507.6 05.12.2008 Indonesia W 00201102405 05.12.2008 Israel 213198 05.12.2008 ... India 5123/0ELNP/2011 05.12.2008 Korea 10 2011 7012066 05.12.2008 Malaysia PI 2011002540 05.12.2008 Philippines 1 2011 501064 05.12.2008 Singapore 201104106 8 05.12.2008 Ukraine 201108389 05.12.2008 .. South Africa 2011/03409 05.12.2008 ........ Patent Family NOVEL COMPOSITION FOR TREATMENT OF ESSENTIAL THROMBOCYTHEMIA (International Publication Number WO 2011/117391), including, but not limited to Country I Organization Application Number Priority Date PCT PCT/EP2011/054628 25.3.2010 Eqypt 976/2012 25.3.2010 Australia 2011231557 25.3.2010 Brazil BR112012024239 3 25 .. 3.2010 Canada 2794237 '''2'5.3.2010 Eurasia (EAPO) 201201329 25.3.2010 25:3.2010 Europe (EPO) 11710202.0 Israel 222072 25.3.2010 India 8696/DELNP/2012 25.3.2010 Japan PCT/EP2011/054628 25.3.2010 Mexico 12/11051 25.3.2010 New Zealand 602366 25.3.2010 Singapore USA I Patent Family PHARMACEUTICAL COMPOSITION (FOR THE PARENTERAL ADMINISTRATION OF AN ULTRASHORT ACTING BETA ADRENORECEPTOR ANTAGONIST) (International Publication Number WO 2009/079679), including, but not limited to Country /Organization Application Number Priority Date PCT/ AT2008/000470 PCT 21.12.2007 · .. f 21":·12.2007 ·· ·· Australia AU 2008340179 Canada 2712414 21.12.2oo:r 2iT2:·2oo1 China CN200880122163X Europe (EPO) EP08863830.9 21.12.2007 .. 5194/DELNP/2010 21.12.2oo? India Japan 2010 538268 21.12.2007 New Zealand 586200 21.12.2007 USA US12809927 21.12.2007 Spain ES08863830T 21.12.2007 Denmark DK08863830T 21.12.2007 HRP2012104ST Croatia 21.12.2007 Portugal PT08863830T 21.12.2007 Validated in: AT, BE, BG, CH, CY, CZ, DE, DK, EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, LV, MC, MT, Nl, NO, PL, PT, RO, SE, SI, SK, TR Patent Family THERAPEUTIC USE OF PROTEIN POLYMER CONJUGATES (International Publication Number WO 2011/072138), including, but not limited to Country /Organization Application Number Priority Date PCT PCT/US2010/059714 10.12.2009 "Ja'Paii JP2012543282 10.12.2009 Europe (EPo)" EP10836693 10.12.2009 China CN201080056068 10.12.2009 Korea KR1020127015843 10.12.2009 Israel IL220249A 10.12.2009 10:12.2009 Singapore SG2012038964A Australia AU2010328067 10.12.2009 Mexico MX2012006132 10.12.2009 ARP100104587A """iii"12.2009 Armenia Canada CA2782624 10.12.2009 USA usi296440SA 10.12.2009 Patent Fam lv Therapeutic Use o Protem"Potvmer Conjugates iJ f Country /Organization Application Number Priority Date us 2009285411 US2010964408A 12.10.2009 PCT W02010US59714A 12.10.2009 Canada CA2782624A 12.10.2009 Exhibit No: 1 0.4.5 Jitle: IP Rights Assignment Agreement for employees of AOP Orphan Pharmaceuticals Aktiengesellschaft See addendum ASSIGNMENT OF RIGHTS between born _._.19_ (the "Employee") and AOP Orphan Pharmaceuticals AG (hereinafter .,AOP"; together with the Employee the "Parties" ). 1. Employee is involved at AOP inter alia in research activities and the creation of creations (in particular research tasks and results, development , procurementand distribution Know How, improvements, as well as all registered and unregistered intellectual property rights, in particular copyrights, trademarks, designs, utility patents and patents as well as applications for such patents; such creations already created in course of his employment as well as all future creations to be created during his employment, including employee inventions, will be referred to as .,Creations" hereinafter. Employee shall immediately and comprehensively disclose each Creation, including past and future Creations to AOP. 2. Employee hereby declares and confirms that he/she has assigned/transferred all in each respect compreh.ensive rights, irrespective of their legal nature under the current and future legal situation (pursuant to Austrian, foreign and/or international legal system), in particular all property and intellectual property rights in the Creations, to AOP. This assignment/transfer is exclusive, thus excluding further rights of third parties, including Employee. This assignment/transfer is irrevocable and therefore continues to have effect after termination of the agreement. AOP therefore has in particular the irrevocable, exclusive, unrestricted as to time, content and territory (worldwide) rights to the Creations especially the right to entire and/or partial publication, reproduction, dissemination, processing, sending, intangible reproduction, making available to the public and any and all other use possible now or in the future (irrespective whether known today) as well as the right to exert the rights according to § 20 and 21 (1) Austrian Copyright Protection Act. AOP is in particular also entitled to assign these rights entirely or partly to third parties, to grant sublicenses respectively rights of exploitation and/or exploi tation permissions and to obtain registired rights (in particular designs, patents, utility. patents and/er trademaFks).ior the Creations.··AOP is noh0bliged to use the 3. Until application for a registered right, in particular until a patent or utility patet\t application is made, both Parties are obliged to maintain absolute confidentiality. 4, Employee expressly warrants that he/she is not restricted in the assignment/ transfer of rights hereunder and that no rights of third parties are in conflict with and/or prevent the assignment hereunder. 5. This comprehensive transfer/assignment of Rights is already fully and finally compensated by the remuneration agreed on. Vienna, Rechteinraumung abgeschlossen zwischen geboren _._.19_ (der "Mitarbeiter"/die ,Mitarbeiterin") und AOP Orphan Pharmaceuticals AG (im Folgenden AOP; zusammen mit der dem Mitarbeiter die "Parteien" ). 1. Der Mitarbeiter/die Mitarbeiterin ist bei AOP unter anderem mit Forschungstatigkeiten und der Schaffung von Leistungen (insbesondere Forschungsarbeiten und ergebnissen, Entwicklungs , Einkaufs und Vertriebs Know How, Know How, Verbesserungen, sowie aile eingetragenen oder nicht eingetragenen lmmaterialgoterrechte, insbesondere Urheberrechte, Marken, Designs, Gebrauchsmuster und Patente sowie Anmeldungen von solchen Rechten) befasst (diese vom Mitarbeiter/ von der Mitarbeiterin bisher schon im Rahmen des Vertragsverhaltnisses geschaffenen sowie die noch im Rahmen des Vertragsverhaltnisses zu schaffenden Leistungen, einschlieP.,Iich allfalliger Diensterfindungen, werden im Folgenden insgesamt kurz ,Leistungen" genannt). Der Mitarbeiter/die Mitarbeiterin · ist verpflichtet, jede Leistung, einschlieBiich fOr die Vergangenheit und die Zukunft, unverzOglich und vollstandig AOP offenzulegen. 2, Der Mitarbeiter/die Mitarbeiterin erklart und bestatigt hiermit, dass er/sie AOP samtliche, in jeder Hinsicht umfassenden Rechte, welcher Rechtsnatur nach derzeitiger und kunftiger Rechtslage (nach 6sterreichischer, auslandischer und/oder internationaler Rechtsordnung) auch immer, insbesondere aile Eigentums und immaterialgOterrechtlichen Rechte an den Leistungen eingeraumt hat. Diese Rechtseinra.umung ist ausschliel11ich und schlieBt daher aile anderen, auch den Mitarbeiter/die Mitarbeiterin selbst, aus. Diese Rechtseinraumung ist unwiderruflich und wirkt daher auch uber eine allfallige Beendigung des Vertragsverhaltnisses hinaus. AOP hat daher insbesondere die unwiderruflichen, ausschlieP.,Iichen, zeitlich, sachlich und raumlich unbeschrankten (weltweiten) Werknutzungsrechte an den Leistungen, insbesondere zur ganzlichen und/oder teilweisen Veroffentlichung, Vervielfaltigung, Verbreitung, Bearbeitung, Sendung, unk6rperlichen dergabe und zur Zuganglichmachung an die Qffentlichkeit sowie jeder sonstigen derzeit oder kOnftig moglichen Nutzung (gleichgOitig ob heute schon bekannt) sowie auch die Berechtigung zur AusObung der in§ 20 und § 21 Abs 1 UrhG bezeichneten Rechte (Urheberbezeichnung und Werkschutz). AOP ist insbesondere auch berechtigt, diese Rechte ganz oder teilweise an Dritte zu Obertragen, Sublizenzen zu erteilen bzw Werknutzungsrechte und/oder Werknutzungsbewilligungen einzuraumen und auch registrierte Schutzrechte (insbesondere Geschmacksmuster, Patente, Gebrauchsmuster und/oder Marken) fOr die Leistungen zu erwerben. AOP ist nicht zur Nutzung der Leistungen sowie zur Nennung des Mitarbeiters/der Mitarbeiterin bei der Nutzung der Leistungen verpflichtet. 3. Bis zur Anmeldung eines Schutzrechtes, insbesondere bei Patent oder Gebrauchsmusteranmeldungen, sind beide Parteien zur absoluten Verschwiegenheit verpflichtet. 4. Der Mitarbeiter/die Mitarbeiterin steht ausdrOcklich dafOr ein, in der Verfugung Ober die Rechte gemai?> dieser Vereinbarung nicht durch Rechtseinraumungen an Dritte beschrankt zu sein und bestatigt, dass auch sonstige Rechte Dritter dieser Vereinbarung nicht entgegen stehen. 5. Diese umfassende Rechtseinraumung ist durch das vereinbarte Entgelt zur Ganze und endgOitig abgegolten. Wien, am Exhibit No: 10.6 Title: Closing Confirmation letter See addendum Dr. Rudolf Stefan Widmann Wintergasse 85/1b 3002 Purkersdorf Austria PERSONALLY DELIVERED To: Elan Corporation, pic Attn: The General Counsel Treasury Building Lower Grand Canal Street Dublin 2, Ireland Fax: +353 1 709 4713 With a copy to: A&L Goodbody Solicitors Attn: Alan Casey IFSC North Wall Quay Dublin 1, Ireland Fax: +353 1 649 2649 Closing Confirmation letter Dear Sirs Exhibit 10.6 This Closing Confirmation Letter relates to the share purchase agreement (the "SPA") entered into by and between SASR Neunundvierzigste Beteiligungsverwaltung GmbH and you, Elan Corporation, pic, (together the "Purchasers") on the one hand and me, Dr. Rudolf Widmann (the "Seller"), on the other hand on [181 1 with respect to the sale and purchase of the Shares (as defined in the SPA). Abbreviations and capitalized terms used herein shall have the meaning given to them in the SPA, unless defined otherwise herein. References to clauses contained herein shall be references to clauses of the SPA, unless expressly indicated otherwise herein. 1. Conditions Precedent to be fulfiUed by the Purchasers The SPA was entered into conditional upon satisfaction by the Purchasers of all of the following Conditions Precedent: Clause 9.1.2: The passing of an ordinary resolution approving the transactions contemplated by the SPA by Purchaser 2's shareholders at a general meeting of Purchaser 2. Exhibit 10.6 I confirm receipt of satisfactory written evidence that this Condition Precedent has been fulfilled on [ date]. Actions taken at Closing by the Purchasers · that the following actions have been taken by the Purchasers at Closing in accordance with Clause 10.4 of the SPA: 2.1 The Purchasers have delivered evidence of the satisfaction of the Condition Precedent pursuant to Clause 9.1.2; 2.2 Purchaser 1 has issued the instructlon(s) for the payment of the Cash Purchase Price pursuant to Clause 4.5.1 and Clause 4.5.2; 2.3 Purchaser 2 has issued the instruction for the delivery to the Seller of the Consideration Shares in accordance with Clause 4.5.3; and 2.4 Purchaser 2 has submitted (A) the filing with the Irish Stock Exchange as regards the admittance to trading of the Consideration Shares on the Main Securities Market of the Irish Stock Exchange in Dublin; and (B) a copy of its notice of the offer of the Consideration Shares to the new Issues calendar (Me/dung zum Emissionskalender) pursuant to § 13 Austrian Capital Markets Act. 3. Seller's Closing Confirmation I, the Seller, therefore acknowledge and confirm that all actions required to be taken by the Purchasers to constitute the Closing of the SPA have been taken" In accordance with the SPA and that Closing has thus occurred today, on the date last written below. 4. General Provisions Clauses 13 through 15 of the SPA are hereby incorporated into this Closing Confirmation Letter by reference and shall apply to this Closing Confirmation Letter mutatis mutandis as if set out herein in full. _________ ,this_._. 2013 Dr. Rudolf Widmann, born 24.03.1957 Elan Corporation, pic Treasury Building Lower Grand Canal Street Dublin 2, Ireland SASR Neunundvierzigste Beteiligungsverwaltung GmbH Tuchlauben 17 1010 Vienna Austria PERSONALLY DELIVERED PRIVATE 8t CONFIDENTIAL Dr. Rudolf Widmann Wintergasse 85/1b 3002 Purkersdorf Austria Fax: +43 [I] Closing Confirmation Letter Dear Dr Widmann Exhibit 10,6 This Closing Confirmation Letter relates to the ·share purchase agreement (the "SPA") entered into by and between you, Dr. Rudolf Widmann (the "Seller"), on the one hand and us, SASR Neunundvierzigste Beteiligungsverwaltung GmbH and Evergreen Corporation, pic, (together the "Purchasers"), on the other hand on [11111 with respect to the sale and purchase of the Shares (as defined in the SPA). Abbreviations and capitalized terms used herein shall have the meaning given to them in the SPA, unless defined otherwise herein. References to clauses contained herein shan be references to clauses of the SPA, unless expressly indicated otherwise herein. 1. Conditions Precedent to be fulfilled by the Seller The SPA was entered into conditional upon satisfaction by the Seller of all of the following Conditions Precedent, to the extent not waived by Purchasers pursuant to Clause 9.6: 1.1 Clause 9 .1.1: Absence of a Material Adverse Effect We confirm receipt of receipt of satisfactory written evidence from the Seller that no Material Adverse Effect, respectively events or circumstances that, individually or in the aggregate, could reasonably be expected to have a Material Adverse Effect have occurred. Exhibit 10.6 2 1.2 Clause 9.1.3: Seller's representations and warranties We confirm receipt of the original of the written confirmation duly executed by the Seller that the representations and warranties as set out in Clause 5 are correct, complete and not misleading, unless otherwise expressly stated in Clause 5, as of Closing. 1.3 Clause 9.1.4: Receipt of irrevocable and unconditional consent by Third Parties We confirm receipt of written evidence reasonably satisfactory that the following Third Parties have irrevocably and unconditionally consented to the consummation of the transactions contemplated by the SPA or confirm that no consent is required: United Therapeutics (Remodulin (Treprostonil)), Ferrer Internacional S.A. {Adasuve ), Alexza Pharmaceuticals, Inc (Advasuve ) and PharmaEssentia Corporation {Peg IFN a. 2b). 1.4 Clause 9.1.5: Company's financial statements 2012 We confirm receipt of the Audited Financial Statements 2012. 2. Actions taken at Closing by the Seller We confirm that the following documents have been presented and delivered to ·us and that the following actions have been taken by the Seller at Closing in accordance with Clause 10.4 of the SPA: 2.1 The Seller has delivered duly executed originals of resignation letters of as members of the supervisory board of the Company with effect as of Closing in form and substance corresponding to the draft attached to the SPA as Exhibit 10.4.1. 2.2 The Seller has delivered 1 (one) original of an IP Rights Assignment Agreement between the Seller and the Company in form and substance corresponding to the draft attached to the SPA as Exhibit 10.4.2, duly executed by the Seller. 2.3 The Seller has delivered the duly executed original of the confirmation pursuant to Clause 9.1.3. 2.4 The Seller has delivered written evidence reasonably satisfactory to the Purchasers that the counterparties in respect of each of the contracts relating to the Programs have irrevocably and unconditionally consented to the consummation of the transactions contemplated by the SPA. 2.5 The Seller t1as delivered the duly executed originals of an IP Rights Assignment Agreement in form and substance corresponding to the draft attached to the SPA as Exhibit 10.4.5 for each employee of the Company who was or is involved in the development of a (the) Product(s). Exhibit 10.6 3 2.6 The Seller has delivered 1 (one) original of the Cooperation and Distribution Agreement by and between the Company and Orpha Trade GmbH (FN 153303 x) in form and substance corresponding to the draft attached to the SPA as Exhibit 10.4.6, duly executed on behalf of Orpha Trade GmbH and the Company. 2.7 The Seller has delivered written evidence reasonably satisfactory to the Purchasers that Orpha Trade GmbH has transferred to the Company all of the assets, rights, licenses and Intellectual Property Rights it owns or controls in relation to Tetmodis. 2.8 The Seller has delivered a simple copy of the notarial deed de merger agreement (Spa/tungsplan oder Spaltungs und Abtretungsvertrag) evidencing the taxneutral (for the Company and the Purchasers) carve out of the Company's 80 % participation in Activartis Biotech GmbH with corporate seat in Vienna and business address at Zimmermannplatz 10, 1090 Vienna, Austria, registered in the companies' register of the Vienna Commercial Court under FN 242030 b and of the loan to which the Company as lender and Activartis Biotech GmbH as borrower are parties. 2.9 The Seller has delivered written evidence reasonably satisfactory to the Purchasers that the Market Authorisations held by Orpha Devel GmbH in respect of the Products have been initiated. 2.10 The Seller has delivered written evidence reasonably satisfactory to the Purchasers that the Company's shareholdings in Novelix, Pharma 21, Inc (USA) and Lampl Business Center Holding Company Ltd, Hong Kong, have been transferred to a third party in a tax neutral manner for the Company and the Purchaser. 2.11 The Seller has delivered (i) to Purchaser 1 the originals of the global interim certificates (Sammelzwischenscheine) numbers [ ) to [ ] and numbers [ ] to [ ]; and (ii) to Purchaser 2 the originals of the global interim certificates (Sammelzwischenscheine) numbers [ ] to [ ] and numbers [ ] to[ .. ], in each case representing the Shares pursuant to Clause 3.3. 3. Purchasers' Closing Confirmation We, the Purchasers, therefore acknowledge and confirm that all actions required to be taken by the Seller to constitute the Closing of the SPA have been taken in accordance with the SPA and that Closing has thus occurred today, on the date last written below. 4. General Provisions Clauses 13 through 16 of the SPA are hereby incorporated into this Closing Confirmation Letter by reference and shall apply to this Closing Confirmation Letter mutatis mutandis as if set out herein in full. Exhibit 10.6 4,this ·_.2013 For SASR Neunundvierzigste Beteiligungsverwaltung GmbH [ ], born [ ] For Elan Corporation, pic [ ], born [ ]
Exhibit (a)(7)
EXECUTION COPY
Dated 19 May 2013
ELAN PHARMA international limited
and
nerano pharma limited
TRANSACTION AGREEMENT
relating to the formation of
speranza therapeutics limted
(the Company)
and related matters
A&L Goodbody

THIS TRANSACTION AGREEMENT is dated 19 May 2013 and made between:
| (1) | Elan Pharma International Limited, a private limited company incorporated in Ireland, (registered no. 222276), whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2 (EPIL); and |
| (2) | Nerano Pharma Limited a limited liability company incorporated in Ireland, (registered no. 527689), whose registered office is at 45 Fitzwilliam Square, Dublin 2 (Nerano). |
(EPIL and Nerano hereinafter may be referred to individually as a Party or collectively as the Parties).
| A. | The Company was incorporated on 13 May 2013 in Ireland under registration number 527543 and has at the date hereof an authorised capital of 1,000,000 shares of US$0.01 each which is $10,000 (ten thousand dollars). |
| B. | Irish OpCo was incorporated on 17 August 2011 in Ireland as an indirect wholly owned subsidiary of Elan under registration number 502444 and has at the date hereof an authorised capital of $1,000,000 (one million US dollars). |
| C. | The Parties have agreed to enter into this Agreement to set forth the terms and conditions relating to the consummation of the Transaction. |
In consideration of the sum of €1.00 this day paid by Nerano to EPIL (the receipt and adequacy of which is hereby acknowledged) IT IS HEREBY AGREED as follows:
| 1. | Definitions and Interpretation: |
| 1.1. | In this Agreement, unless the context otherwise requires: |
an Affiliate of any person means:
| (a) | any holding company or subsidiary of that person and any subsidiary of any such holding company; and |
| (b) | any other individual, company, body corporate, partnership or other entity which; |
| (i) | is Controlled by that person; |
| (ii) | Controls that person; or |
| (iii) | is Under Common Control with that person; |
Agreement means this Transaction Agreement;
the Ancillary Agreements shall have the meaning given in clause 2.2;
Business Day means any day on which banks are generally open for business in Dublin;
the Business Transfer Agreement means the agreement to be made between EPIL and Irish OpCo pursuant to which Irish OpCo agrees to purchase certain assets of EPIL related to the D5 Business, substantially in the form of Exhibit A to this Agreement;
Business Plan means the agreed form business plan relating to the Group, to be agreed between the Parties within 90 days after Completion, using their commercially reasonable efforts, which is to constitute the “Business Plan” for the purposes of the Shareholders' Agreement;
Clinical Plan means the plan that sets out the clinical goals of the Company in relation to the D5 Business, as agreed by EPIL and Nerano and set out at Exhibit E to this Agreement;
| 1 |
the Company means Speranza Therapeutics Limited (registered no. 527543) whose registered office is McCann FitzGerald, Riverside One, Sir John Rogerson's Quay, Dublin 2;
Completion means completion of this Agreement pursuant to clause 3;
Confidential Information means:
| (a) | any and all information which is or was received or obtained as a result of entering into or performing, or which is or was supplied by or on behalf of a party in the negotiations leading to, this Agreement; and |
| (b) | the existence of, or any of the terms or provisions of, any of the Transaction Documents. |
Control means the power of a person (whether alone or together with any other person or persons) to secure directly or indirectly, including through one or more intermediaries, that the affairs of another person are conducted in accordance with the wishes of the first-mentioned person, either by means of the holding of shares or the possession of voting power directly or indirectly in or in relation to that or any other person, or by virtue of any powers conferred by any applicable laws or regulations or the constitutional or other documentation regulating or managing the affairs of that or any other person, or otherwise, and the expressions Controlled, Controls and Under Common Control shall be construed accordingly;
D5 Business means the research, development, manufacture and commercialisation of a Product;
Derivative means any compounds identified, obtained, developed, created, synthesized, generated, designed or resulting from, based upon, containing or incorporating the chemical structure of Scyllo-Inositol as those compounds are generically and/or specifically disclosed and claimed in the Licensed Patent Rights;
Elan means Elan Corporation, plc, a public limited company incorporated in Ireland (registered no. 30356), whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2;
Elan Offer Period means the "offer period" (as such term is defined under the Irish Takeover Panel Act, 2007, Takeover Rules) in which Elan was placed by virtue of a Rule 2.4 Announcement by Royalty Pharma dated 25 February 2013;
Elan Shareholder Approval means the passing of an ordinary resolution by Elan's shareholders as may be required by Rule 21 of the Irish Takeover Panel Act, 2007, Takeover Rules;
Elan's Solicitors means A&L Goodbody, North Wall Quay, IFSC, Dublin 1;
the Employees means those persons whose employment is to transfer to Irish OpCo or the Subsidiary in connection with the Transaction;
ES1 means Elan's indirect wholly owned subsidiary, Elan Science One Limited (registered no. 460037);
Executive Employment Agreement means the employment or services agreement to be entered into between Irish OpCo and Seamus Mulligan on Completion;
Group means the Company and any subsidiary of the Company hereafter established or acquired;
Ireland means Ireland excluding Northern Ireland;
Irish OpCo means Elan Science Nine Limited (in the process of changing its name to Speranza Biopharma Limited) an indirect wholly owned subsidiary of Elan (registered number 502444);
Licensed Patent Rights shall have the meaning ascribed to it in the Collaboration Agreement (as amended) between Waratah Pharmaceuticals Inc. and EPIL dated 25 September 2006;
| 2 |
Loan Agreements means the loan agreements of even date herewith under which Nerano is providing Irish OpCo with a ten year interest-free loan of $20,000,000 to fund development of the D5 Business, and EPIL is providing Irish OpCo with the ten year interest-free loan for $7,000,000 to fund development of the D5 Business;
Product means any pharmaceutical preparation or dosage form containing Scyllo-Inositol or a Derivative;
the Royalty and Option Agreement means the agreement to be made between EPIL and Irish OpCo to regulate the commercialisation of the Products, substantially in the form of Exhibit B to this Agreement;
Scyllo-Inositol is the compound “AZD-103”, as more specifically disclosed in the Licensed Patent Rights;
the Shareholders' Agreement means the agreement to be made between, inter alia, ES1 and Nerano to regulate the parties relationship as shareholders in the Company, substantially in the form of Exhibit C to this Agreement;
the Share Purchase Agreement means the agreement to be made between EPIL and the Company for the sale and purchase of 100% of the issued share capital of Irish OpCo, substantially in the form of Exhibit D to this Agreement;
the Subsidiary means Elan's indirect wholly-owned subsidiary, Speranza Therapeutics Corp., a limited liability company incorporated in the State of Delaware (registered no. 5332090);
the Transaction means, collectively, the transactions contemplated to be consummated pursuant to the Transaction Documents;
the Transaction Documents means this Agreement, the Share Purchase Agreement, the Shareholders' Agreement, the Business Transfer Agreement, the Royalty and Option Agreement, and the Ancillary Agreements; and
the Transitional Services Agreement means the agreement to be made between EPIL and the Company in relation to the provision by EPIL and/or its Affiliates of certain transitional services to the Group for a six month period after Completion (or such other period as may be agreed between EPIL and Nerano).
| 1.2. | Unless the context otherwise requires: |
| 1.2.1. | words importing the singular include the plural and vice versa, words importing the masculine include the feminine, and words importing persons include corporations; |
| 1.2.2. | reference to writing or similar expressions includes transmission by telecopier or comparable means of communication; |
| 1.2.3. | references to Acts, statutory instruments and other legislation are to legislation operative in Ireland and to such legislation amended, extended or re-enacted (whether before or after the date of this Agreement), and include any legislation of which it is a re-enactment (whether with or without modification) and also include any subordinate legislation made under that legislation; and |
| 1.2.4. | reference to any document includes that document as amended or supplemented, whether before or after the date of this Agreement. |
| 3 |
| 2. | Actions Prior To Completion |
| 2.1. | The Parties covenant and agree to take, or to cause their respective Affiliates to take, the following actions from and after the date hereof and prior to Completion (or the date of termination hereof, in the event that the conditions set forth in clause 3 are not satisfied or waived in accordance with the terms hereof): |
| 2.1.1. | EPIL shall procure that save in respect of the matters expressly required by the Transaction Documents, neither Irish OpCo nor the Subsidiary will carry out any business or trading activities or incur any liability or obligation from the date of its incorporation or formation until Completion; and |
| 2.1.2. | Nerano shall procure that save in respect of the matters expressly required by the Transaction Documents, the Company will not carry out any business or trading activities or incur any liability or obligation from execution of this Agreement until Completion. |
| 2.2. | The Parties covenant and agree to use their commercially reasonable efforts from the date of this Agreement to agree the final form of the following agreements, each of which is to be executed and delivered at Completion (the Ancillary Agreements): |
| 2.2.1. | the Loan Agreements; |
| 2.2.2. | individual Employment Agreements, between Irish OpCo or the Subsidiary (as appropriate) and the Employees; |
| 2.2.3. | the Executive Employment Agreement; |
| 2.2.4. | Memorandum and Articles of the Company and the Subsidiary; |
| 2.2.5. | the Business Plan; |
| 2.2.6. | the Transitional Services Agreement; and |
| 2.2.7. | any matters which a Transaction Document contemplates is to be agreed at the time of execution thereof with such other documents, instruments and agreements as the Parties may determine are necessary or advisable to implement the Transaction. |
| 2.3. | The Parties shall not, and shall procure that their respective Affiliates shall not, prior to the Effective Time unless otherwise required by law |
| 2.3.1. | increase or offer, promise or agree to increase the remuneration (howsoever defined but including salary and any variable and/or incentive arrangements) or the severance arrangements in respect of any of the Employees; |
| 2.3.2. | increase or make offers of employment so as to increase the number of the Employees; or |
| 2.3.3. | replace or offer to redeploy any of the Employees. |
| 3. | Completion |
| 3.1. | Completion shall take place on the fulfilment of the conditions set forth in clause 3.4 of this Agreement at the offices of Elan's Solicitors. |
| 3.2. | It is agreed that: |
| 4 |
| 3.2.1. | EPIL shall use all commercially reasonable endeavours to ensure that the condition set forth in clause 3.4.3 of this Agreement is fulfilled at least five Business Days prior to 28 June 2013, which is the target date for Completion (the “Target Completion Date”); and |
| 3.2.2. | EPIL and Nerano shall use all commercially reasonable endeavours to ensure that the condition set forth in clause 3.4.4 of this Agreement is fulfilled at least five Business Days prior to the Target Completion Date. |
| 3.3. | In any event, if the conditions in clause 3.4 are not fulfilled or waived by the Parties on or before 1 August 2013, either party may at any time prior to the fulfilment of the conditions, rescind this Agreement by notice to the other, and this will not prejudice the other rights and remedies of the rescinding Party under this Agreement. |
| 3.4. | Conditions: |
The conditions referred to in this clause 3 are:
| 3.4.1. | all approvals and actions of or by all relevant governmental, regulatory or administrative authorities that are necessary to consummate the transactions contemplated by this Agreement and required to be obtained prior to Completion by applicable law shall have been obtained; |
| 3.4.2. | no injunction or restraining order shall have been issued by any court of competent jurisdiction and be in effect which restrains or prohibits, and no governmental, regulatory or administrative authority of competent jurisdiction shall have enacted or issued any law, rule, regulation or order that remains in effect and that has the effect of making illegal or otherwise restraining, enjoining or prohibiting, consummation of the transactions contemplated by this Agreement; |
| 3.4.3. | the Elan Shareholder Approval shall have been obtained, or the Elan Offer Period shall have ended; and |
| 3.4.4. | the Parties shall have agreed the final form of each of the Ancillary Agreements (other than the Business Plan). |
| 3.5. | Completion Obligations: |
Upon Completion:
| 3.5.1. | each of the Parties shall cause its respective Affiliates to deliver to the other Party the executed Transaction Documents to which it is a Party, other than the Employment Agreements which shall be delivered within three Business Days of Completion; and |
| 3.5.2. | the completion obligations set forth in each of the Transaction Documents (if any) shall be performed in accordance with the terms thereof. |
| 3.6. | Post Completion: |
In accordance with the terms of the Shareholders Agreement, each of the Parties shall cause its respective Affiliates to use commercially reasonable efforts to agree the terms of the Business Plan within 90 Day of Completion. The terms of the Business Plan shall be in accordance with the provisions of the Clinical Plan. Until the Business Plan is agreed, the D5 Business shall be carried out in accordance with the Clinical Plan.
| 4. | Warranties |
| 5 |
| 4.1. | Each Party hereto warrants and represents to and undertakes with the other Party as at the date of this Agreement that: |
| 4.1.1. | it is duly incorporated and validly existing under the laws of its jurisdiction of incorporation; |
| 4.1.2. | it has full power and authority to enter into this Agreement, which constitutes legal obligations which are valid and binding on it in accordance with their terms, and to perform the obligations expressed to be assumed by it, and has taken all necessary corporate action to authorise the execution, delivery and performance of this Agreement; and |
| 4.1.3. | it has duly executed this Agreement. |
| 5. | Miscellaneous: |
| 5.1. | Entire Agreement: |
This Agreement and the agreements referenced herein supersede all prior representations, arrangements, understandings and agreements, and set out the entire, complete and exclusive agreement and understanding between the Parties.
| 5.2. | Remedies Cumulative: |
The provisions of this Agreement and the rights and remedies of the Parties are independent, cumulative and are without prejudice and in addition to any other rights or remedies which a Party may have whether arising under common law, statute, custom or otherwise. The exercise by a Party of any one right or remedy under this Agreement or at law or in equity will not (unless expressly provided in this Agreement or at law or in equity) operate so as to hinder or prevent the exercise by that Party of any other right or remedy.
| 5.3. | Notices: |
Any notice or other communication to be given or served under this Agreement shall be in writing, addressed to the relevant party and expressed to be a notice or communication under this Agreement and, without prejudice to the validity of another method of service, may be delivered or sent by pre-paid post or facsimile addressed as follows:
| (1) | to Elan: |
William F. Daniel
Company Secretary
Elan Pharma International Limited
Treasury Building
Lower Grand Canal St.
Dublin 2
With a copy to:
Alan Casey
A&L Goodbody
North Wall Quay,
IFSC, Dublin 1
Fax: +353 1 6492649
| (2) | to Nerano: |
| 6 |
Seamus Mulligan
Nerano Pharma Limited
45 Fitzwilliam Square
Dublin 2
With a copy to:
Ben Gaffikin
McCann FitzGerald,
Sir John Rogerson's Quay, Dublin 2,
Fax: +353 1 8290010
or to such other address or facsimile number as the addressee may have previously substituted by notice.
| 5.3.2. | A notice or other communication will be deemed to have been duly served or given: |
| (1) | in the case of delivery, at the time of delivery; |
| (2) | in the case of posting, 48 hours after posting (and proof that the envelope containing the notice or communication was properly addressed, prepaid, and posted by airmail (where applicable) will be sufficient evidence that the notice or other communication has been duly served or given); or |
| (3) | in the case of facsimile, upon transmission, subject to the correct code or facsimile number being received on the transmission report, |
but if a notice is given or served at business premises other than between 9.00 am and 5.00 pm on a Business Day, it will be deemed to be given or served on the next following Business Day.
| 5.3.3. | A party giving or serving a notice or other communication under this Agreement by facsimile shall also give or serve a copy by post, but without prejudice to the validity and effectiveness of the service by facsimile. |
| 5.3.4. | All notices or other communications shall be in the English language. |
| 5.4. | Confidentiality: |
| 5.4.1. | Each of the parties to this Agreement undertakes with each other for the benefit of the other party that they shall preserve the confidentiality of, and not directly or indirectly reveal, report, publish, disclose, transfer or use for their own or any other purposes Confidential Information except: |
| (1) | in the circumstances set out in clause 5.4.2; |
| (2) | to the extent otherwise expressly permitted by this Agreement; or |
| (3) | with the prior written consent of the other parties to this Agreement and of the party to whose affairs such Confidential Information relates. |
| 5.4.2. | A party is permitted to disclose Confidential Information: |
| 7 |
| (1) | where the Confidential Information is in or, after the date of this Agreement, enters the public domain otherwise than as a result of: (i) a breach by that party of its obligations in this clause or (ii) a breach by the person who disclosed that Confidential Information of a confidentiality obligation where the party is or was aware of such breach; |
| (2) | if and to the extent that the party discloses Confidential Information to any person: |
| (a) | in compliance with any requirement of law; |
| (b) | in response to a requirement of any securities exchange, regulatory or governmental authority or court having jurisdiction over that party; or |
| (c) | in order to obtain Tax or other clearances or consents from the Revenue Commissioners or other relevant Tax or regulatory authorities, |
provided that any such information disclosable pursuant to sub-clauses (a), (b) or (c) may, to the fullest extent permitted by law, be disclosed only after prior consultation with the other parties to this Agreement.
| 5.5. | Assignment: |
None of the parties to this Agreement may assign any of its rights under this Agreement without the prior written consent of each of the other parties except that any party is entitled to assign the benefit of any provision of this Agreement to an Affiliate.
| 5.6. | Costs and Expenses |
Each Party shall be responsible for their own legal fees, costs and expenses incurred in connection with the negotiation, preparation and execution of this Agreement.
| 5.7. | Counterparts |
This Agreement may be executed in any number of counterparts and by the different Parties on separate counterparts each of which when executed and delivered shall constitute an original, all such counterparts together constituting one and the same instrument. The expression "counterpart" shall include any executed copy of this Agreement transmitted by facsimile.
| 5.8. | Binding on Successors |
Subject to clause 5.5, the provisions of this Agreement shall enure to the benefit of and be binding upon the respective successors and personal representatives of the Parties hereto.
| 5.9. | Governing Law and Jurisdiction |
This Agreement shall in all respects (including the formation thereof and performance thereunder) be governed by and construed in accordance with the laws of Ireland. The Parties to this Agreement agree to submit to the exclusive jurisdiction of the Courts of Ireland in relation to any disputes or proceedings arising out of or in connection with this Agreement.
[remainder of page intentionally left blank]
| 8 |
EXHIBIT A
Business Transfer Agreement
Agreed Form
SUBJECT TO CONTRACT / CONTRACT DENIED
Dated [·] 2013
ELAN PHARMA INTERNATIONAL LIMITED
(Vendor)
and
SPERANZA BIOPHARMA LIMITED
(Purchaser)
BUSINESS TRANSFER AGREEMENT
relating to the D5 Business
A & L GOODBODY
THIS BUSINESS TRANSFER AGREEMENT is dated [·] 2013 and made between
| (1) | ELAN PHARMA INTERNATIONAL LIMITED (registered no. 222276) having its registered office at Treasury Building, Lower Grand Canal Street, Dublin 2 (the Vendor); and |
| (2) | SPERANZA BIOPHARMA LIMITED a company incorporated in Ireland (Registered No. 502444) whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2, Ireland (the Purchaser). |
RECITALS:
| A. | The Vendor carries on the D5 Business (as hereinafter defined) utilising the Assets. |
| B. | The Vendor has agreed to sell and the Purchaser has agreed to purchase the Assets upon the terms and subject to the conditions of this Agreement. |
IT IS HEREBY AGREED as follows:
| 1. | PRELIMINARY |
| 1.1. | Definitions: In this Agreement and in the Schedules unless the context otherwise requires: |
1963 Act means the Companies Act, 1963;
1990 Act means the Companies Act, 1990;
Affiliate of a company shall mean any other company controlling, controlled by or under common control with such company;
Assets means the assets listed in Clause 2.1 including all of the Intellectual Property Rights owned by and/or licensed to the Vendor in respect of or related exclusively to the D5 Business, other than for the avoidance of doubt the Excluded Assets, all of which are to be acquired by or novated in favour of the Purchaser pursuant to this Agreement;
Business Contracts means all contracts, agreements, arrangements and understandings of every kind relating to the D5 Business, including but not limited to those set out in Schedule 3;
Business Day means any day on which banks are generally open for business in Dublin excluding Saturdays and Sundays;
Capital Contribution means the capital contribution in the sum of $63 million to be made by Elan Science One Limited to the Purchaser on the date hereof;
Completion means completion of the sale and purchase of the Assets pursuant to this Agreement;
Confidential Information means any and all information received, developed or obtained as a result of or in connection with (i) the operation of the D5 Business (ii) the Assets and/or (ii) entering into or performing, or supplied by or on behalf of a party in the negotiations leading to, this Agreement and which in each case relates to:
| (a) | the D5 Business; |
| (b) | the provisions of this Agreement; |
| (c) | the negotiations relating to this Agreement; |
| (d) | the subject matter of this Agreement; or |
| (e) | Irish OpCo Group. |
D5 Business means the research, development, manufacture and commercialisation of a Product;
Derivative means any compounds identified, obtained, developed, created, synthesized, generated, designed or resulting from, based upon, containing or incorporating the chemical structure of Scyllo-Inositol as those compounds are generically and/or specifically disclosed and claimed in the Licensed Patent Rights;
Effective Time means the time at which this Agreement is executed;
Elan Business means all businesses and other activities carried on by any member of the Elan Group;
Elan Employees means those individuals employed exclusively or predominantly in the Elan Business and the term Elan Employee shall be construed accordingly;
Elan Group means Elan Corporation, plc and its subsidiary undertakings from time to time;
Employees means those employees of any company within the Elan Group employed solely in connection with the Assets whose employment it has been agreed will transfer to the Irish OpCo Group on Completion;
Employment Liabilities means costs (including the cost of wages, salaries, accrued leave and holiday entitlements, accrued bonus entitlements and other remuneration or benefits), redundancy costs, expenses, taxation, PRSI payments, health contributions, levies, losses, claims, demands, actions, fines, penalties, awards, liabilities, expenses (including reasonable legal expenses);
Encumbrance includes any adverse claim or right or third party right or interest; any equity; any option or right of pre-emption or right to acquire or restrict; any mortgage, charge, assignment, hypothecation, pledge, lien or security interest or arrangement of whatsoever nature; any reservation of title; any hire purchase, lease or instalment purchase agreement and any other encumbrance, priority or security interest or similar arrangement of whatever nature;
EPIL Loan Agreement means the loan agreement entered into on the date hereof under which EPIL is providing the Purchaser with the ten year interest-free loan for $7,000,000 to fund development of the D5 Business;
Equipment means all equipment such as Blackberries, printers and laptops used by the Employees in connection with the D5 Business;
Excluded Assets means those assets of the Vendor set out in Schedule 1 which are not being sold hereunder;
Information means all customer and supplier records and correspondence (including, in each case, the full name and the address and addresses for delivery or collection or for provision of services) relating to the D5 Business, all books of account relating to the D5 Business and other records of the D5 Business for the six years prior to Completion, all deeds and documents evidencing title to the Assets (duly stamped where appropriate), all sales and technical literature
and all other documents, computer records and programs relating to or required for the due and efficient carrying on of the D5 Business and all files and other relevant information relating to the Employees;
Intellectual Property means any and all rights pertaining to inventions, discoveries, trade secrets, confidential business information, financial, marketing and business data, concepts, ideas and improvements to existing technology (in each case whether or not written down or otherwise converted to tangible form), patents, patent applications, patent disclosures, designs, trade marks, service marks, trade names, logos, rights in any domain names, algorithms, database rights, goodwill, copyright(s), moral rights, know-how, reputation, modes, get-up, computer programs and analogous property, plans, models, and all other forms of industrial or intellectual property (in each case in any part of the world and whether or not registered or registerable and to the fullest extent thereof and for the full period therefor and all extensions and renewals thereof), all applications for registration of the foregoing and all rights and interest, present and future thereto and therein;
Intellectual Property Rights means such Intellectual Property owned by and/or licensed to the Vendor used exclusively in connection with the D5 Business;
Ireland means Ireland excluding Northern Ireland and Irish will be construed accordingly;
Irish OpCo Group means the Purchaser and its subsidiaries, any holding company of the Purchaser and any subsidiary of any such holding company and references to Irish OpCo Group Company and member of the Irish OpCo Group shall be construed accordingly;
Licensed Patent Rights means the patents listed at Part B of Schedule 2, being patents and/or patent applications that are licensed to the Vendor;
Listed Business Contracts means the Business Contracts listed in Schedule 3;
Owned Patents means the patents in Part A of Schedule 2 to this Agreement, being patents and/or patent applications that are owned by the Vendor;
Pre-Effective Time Claims means all claims by any person outstanding against the Vendor as at the Effective Time or arising by reason of any act or omission by the Vendor before the Effective Time;
Pre-Effective Time Liabilities means all liabilities (whether actual or contingent), outgoings and expenses incurred by or on behalf of the Vendor in respect of the D5 Business and/or the Assets before the Effective Time, including, without limitation, all creditors and all wages, accrued holiday pay, bonuses and other outgoings in respect of the Employees;
Product means any pharmaceutical preparation or dosage form containing Scyllo-Inositol or a Derivative;
Scyllo-Inositol is the compound “AZD-103”, as more specifically disclosed in the Licensed Patent Rights;
Taxation and Tax means all forms of taxation, duties, imposts and levies whether of Ireland or elsewhere, including (but without limitation) income tax, corporation tax, corporation profits tax, advance corporation tax, capital gains tax, capital acquisitions tax, residential property tax, wealth tax, value added tax, customs and other import and export duties, excise duties, vehicle registration tax, stamp duty, capital duty, social insurance, social welfare or other similar contributions and other amounts corresponding thereto whether payable in Ireland or elsewhere, and any costs, interest, surcharge, penalty or fine relating to the same and any penalties chargeable for non-compliance with any statutory provisions or regulations in connection therewith
and the word taxation shall be construed accordingly;
TCA means the Taxes Consolidation Act, 1997;
Transfer Regulations means the European Communities (Protection of Employees on Transfer of Undertakings) Regulations, 2003 and any equivalent or similar laws which are applicable in any other jurisdiction in which persons assigned to the D5 Business are engaged; and
VAT means value added tax within the meaning of the Value-Added Tax Consolidation Act 2010.
| 1.2. | The Schedules referred to in this Agreement form an integral part of this Agreement, and references to this Agreement include reference to them. |
| 1.3. | Headings are inserted for convenience only and do not affect the construction of this Agreement. |
| 1.4. | All references in this Agreement to costs, charges or expenses include any value added tax or similar tax charged or chargeable on them. |
| 1.5. | Unless expressly stated in this Agreement or the context otherwise requires, in this Agreement: |
| 1.5.1. | references to persons are deemed to include references to natural persons, firms, partnerships, companies, corporations, associations, bodies corporate, trusts and investment funds (in each case whether or not having a separate legal personality) but references to individuals are deemed to be references to natural persons only; |
| 1.5.2. | words importing the singular include the plural and vice versa and words importing the masculine include references to the feminine and neuter and vice versa; |
| 1.5.3. | reference to writing or similar expressions includes transmission by facsimile or electronic means; |
| 1.5.4. | a word or phrase the definition of which is contained or referred to in section 2 of the 1963 Act has the meaning attributed to it by that definition; |
| 1.5.5. | references to Acts, statutory instruments and other legislation are to legislation operative in Ireland and to such legislation, modified, consolidated, amended or re-enacted (whether before or after the date of this Agreement) and any subordinate legislation made under that legislation; |
| 1.5.6. | reference to any Irish legal term, concept, legislation or regulation (including, those for any action, remedy, method of judicial proceeding, document, statute, court official, governmental authority or agency) or any accounting term or concept, in respect of any jurisdiction other than Ireland will be construed as a reference to the term or concept which most nearly corresponds to it in that jurisdiction; |
| 1.5.7. | reference to this Agreement includes this Agreement as amended or supplemented from time to time; and |
| 1.5.8. | “including” and “includes” mean including or includes without limiting the generality of the foregoing. |
| 2. | SALE OF ASSETS |
| 2.1. | Sale and Purchase: Subject to the terms of this Agreement, the Vendor shall sell as legal and beneficial owner and the Purchaser shall purchase with effect from the Effective Time in reliance on the provisions of this Agreement free from all Encumbrances and together with all rights now |
or hereafter attaching thereto, the Assets including:
| 2.1.1. | the Owned Patents; |
| 2.1.2. | all of the Vendor's right, title and interest in the Licensed Patent Rights; |
| 2.1.3. | the Intellectual Property Rights; |
| 2.1.4. | subject to clause 4 the full benefit subject to the burden (so far as the same is capable of assignment) of the Business Contracts and all of the Vendor’s pending contracts, engagements and all orders in connection with the D5 Business; |
| 2.1.5. | the Investigational New Drug Applications (IND) related to the Product; |
| 2.1.6. | any related goodwill of the D5 Business and to represent the Purchaser as carrying on the D5 Business in continuation of that carried on by the Vendor and in succession thereto; |
| 2.1.7. | the Information; |
| 2.1.8. | the Equipment; and |
| 2.1.9. | the right, title and interest of the Vendor in all other assets which pertain or are used in connection the D5 Business except the Excluded Assets. |
| 2.2. | Other Benefits: |
| 2.2.1. | If the Vendor receives any sums or benefits in respect of any of the Assets it shall forthwith pay or cause to be paid (as the case may be) to the Purchaser as soon as practicable and in any event within ten Business Days of receipt thereof, to a bank account to be nominated by the Purchaser. |
| 2.2.2. | The Vendor shall forthwith upon the execution hereof notify the interest of the Purchaser to the insurers of the Assets and the D5 Business and, at the expense of the Purchaser, keep in force its existing insurance policies in respect of the Assets until 180 days after Completion. |
| 2.3. | The Consideration: |
The consideration for the said sale of the Assets shall be satisfied by the payment of $1.00 from the Purchaser to the Vendor, the adequacy and sufficiency of which is hereby acknowledged by the Parties (the Consideration).
| 2.4. | Purchase of all the Assets: The Purchaser will not be obliged to complete the purchase of any of the Assets unless the purchase of all of the Assets is completed simultaneously. |
| 2.5. | Value Added Tax (VAT): |
| 2.5.1. | The parties shall use their best endeavours to ensure that by virtue of the provisions of sections 20(2)(c) and 26(2) of the Value-Added Tax Consolidation Act 2010 no VAT will be chargeable in respect of the sale and purchase of the Assets hereby agreed to be made. |
| 2.5.2. | Notwithstanding Clause 2.5.1, the Purchaser shall not be liable to pay to the Vendor or to any other person any amount representing VAT in respect of the sale of the Assets unless the Vendor demonstrates to the satisfaction of the Purchaser that the provisions of sections 20(2)(c) and 26(2) of the Value-Added Tax Consolidation Act 2010 do not apply. |
For the avoidance of doubt, written confirmation from the Irish Revenue Commissioners on this point shall be treated as demonstration to the satisfaction of the Purchaser.
| 2.5.3. | Subject to Clause 2.5.2, if VAT is properly chargeable in respect of the sale of the Assets and the Vendor is accountable in respect of the payment of the VAT to the VAT authorities, the Vendor shall deliver to the Purchaser a VAT invoice prepared in a format which is in accordance with relevant VAT legislation. On receipt of this VAT invoice the Purchaser shall pay to the Vendor the amount of VAT properly arising in respect of the sale of the Assets. |
| 2.5.4. | Notwithstanding Clause 2.5.2, if following the payment of any amount of VAT by the Purchaser to the Vendor in respect of the sale of the Assets it is established that VAT was incorrectly chargeable, the Vendor shall pay to the Purchaser the amount of any VAT incorrectly charged. For the avoidance of doubt, only written confirmation from the Irish Revenue Commissioners shall be treated as establishing that VAT was incorrectly chargeable. |
| 2.5.5. | In the event that the Vendor will incur a clawback of VAT in relation to or arising from the sale, the parties hereby exercise a joint option to tax within the meaning of section 94(5) Value-Added Tax Consolidation Act 2010. |
| 3. | COMPLETION AND CONDITIONS |
| 3.1. | Completion: Completion shall take place on the Effective Time at such address as the parties may agree. |
| 3.2. | Actions at Completion: Upon Completion: |
| 3.2.1. | the Vendor shall deliver to the Purchaser: |
| (1) | the Information; |
| (2) | duly executed assignments in the agreed form of the Intellectual Property Rights executed, where reasonably required by the Purchaser, in the presence of a Notary Public; |
| (3) | the Equipment, title to which passes by delivery at the place(s) where it is located; |
| (4) | subject to clause 4, duly executed deeds of assignment or novation for all Listed Business Contracts; |
| (5) | as far as is reasonably practicable, possession of the other Assets; |
| 3.2.2. | the Vendor shall, and shall use all reasonable endeavours to procure that all other necessary parties (if any), execute and deliver to the Purchaser all such assurances and things as may be necessary to vest in the Purchaser good and marketable title to the Assets (including deeds of assignment in relation to the Intellectual Property Rights) including such evidence of corporate power and proper execution as the Purchaser may reasonably require; |
| 3.2.3. | the Vendor and the Purchaser shall comply with the terms of clause 5 (in relation to the Employees); |
| 3.2.4. | the Vendor shall procure the payment of the Capital Contribution by ES1 to the Purchaser; |
| 3.2.5. | the Vendor and the Purchaser shall execute and deliver the EPIL Loan Agreement; and |
| 3.2.6. | the Vendor shall provide the Purchaser with details of the Vendor's Irish tax reference number (including evidence satisfactory to the Purchaser allowing it to verify the accuracy of the number provided ) which the Purchaser requires for the purposes of making the relevant filing in connection with the discharge of the liability to stamp duty arising on the transfer of the Assets. |
| 3.3. | Payments at Completion: The Purchaser shall on Completion pay the sum of $1.00 to the Vendor in accordance with clause 2.3.1 of this Agreement. |
| 4. | Business Contracts |
| 4.1. | Subject to the provisions of sub-clauses 4.2, 4.3 and 4.4 after the Effective Time the Purchaser shall: |
| 4.1.1. | be entitled to the benefit of the Business Contracts; |
| 4.1.2. | perform all the Vendor’s obligations under the Business Contracts; and |
| 4.1.3. | indemnify and keep indemnified the Vendor on demand against each loss, liability and cost which the Vendor incurs as a result of a failure on the part of the Purchaser to perform those obligations or any breach by the Purchaser of any law, directive, regulation or the like, applicable to any of the Business Contracts, to the extent that the loss, liability or cost is attributable to the Purchaser’s act or omission after the Effective Time. |
| 4.2. | Nothing in this Agreement shall: |
| 4.2.1. | require the Purchaser to perform any obligation falling due for performance or which should have been performed prior to the Effective Time; |
| 4.2.2. | make the Purchaser liable for any act, neglect, default or omission in respect of any of the Business Contracts on or prior to the Effective Time; or |
| 4.2.3. | impose any obligation on the Purchaser in respect of any contract other than the Business Contracts. |
| 4.3. | At Completion the Vendor will, at the option of the Purchaser acting reasonably, in respect of each Business Contract:- |
| 4.3.1. | assign (or procure the assignment of) the Business Contract in question to the Purchaser or its nominee(s); or |
| 4.3.2. | procure the novation of the Business Contract in question so that the Purchaser or its nominee(s) will become party to the relevant Business Contract in place of the Vendor |
in each case on such terms as may be agreed to by the Purchaser acting reasonably.
| 4.4. | Insofar as the consent of a third party is required to comply with the terms of clause 4.3 in respect of a Business Contract, the Vendor shall use all reasonable endeavours to procure such consent as soon as reasonably practicable at its own cost. Until such Business Contract is assigned or novated in accordance with clause 4.3: |
| 4.4.1. | the Vendor shall hold it in trust for the Purchaser absolutely and account to the Purchaser promptly after receipt by it for the value of any benefit of the Business Contract that arises (or relates to the period) after Completion; |
| 4.4.2. | the Vendor must enforce the Business Contract against any counterparty to it in the manner that the Purchaser reasonably directs (and promptly following such direction) from time to time, at the expense of the Purchaser; |
| 4.4.3. | the Purchaser shall, to the extent it lawfully can, perform all the obligations of the Vendor under the Business Contract, to be discharged after the Effective Time, if subcontracting is permissible and lawful under the Business Contract; |
| 4.4.4. | the Purchaser shall indemnify and keep indemnified the Vendor on demand against each loss, liability and cost which the Vendor incurs as a result of any failure on the part of the Purchaser to perform the obligations in clause 4.4.3; |
| 4.4.5. | if such subcontracting is not permissible, the Vendor shall perform all of its obligations under the Business Contract at the Purchaser’s expense; and |
| 4.4.6. | the Vendor shall (so far as it lawfully may) give all reasonable assistance to the Purchaser to enable the Purchaser to enforce its rights under the Business Contract; |
| 4.5. | This Agreement shall not constitute any assignment or an attempted assignment of any Business Contract if the assignment or attempted assignment would constitute a breach of the Business Contract. |
| 4.6. | This clause 4 does not affect the Purchaser’s rights and remedies against the Vendor in respect of a Business Contract which the Vendor warrants is or was assignable. |
| 4.7. | If any Business Contract is being novated, then this Agreement does not act as a contract or agreement for the sale of any estate or interest in the Business Contracts nor does it operate to assign any of the Business Contracts. Instead the benefit of the Business Contracts will be novated to the Purchaser pursuant to the deed of novation which is to be entered into between the parties. |
| 5. | EMPLOYEES |
| 5.1. | Safeguarding of Employees' Rights: The Vendor shall discharge all of its obligations (if any) in relation to the Employees arising under or by virtue of the Transfer Regulations. |
| 5.2. | Evidence and Information: The Vendor shall both before and after Completion furnish to the Purchaser such evidence and information as the Purchaser may from time to time require in relation to the discharge by the Vendor of the Vendor's said obligations together with such other information as the Purchaser may from time to time require in relation to the intentions, proposals, obligations and actions of the Vendor in connection with contracts of employment, employment relationships or collective agreements and any other matters in connection therewith relating to any of the Employees. |
| 5.3. | Other Obligations: The Vendor shall perform and shall be fully liable and responsible for all obligations and liabilities whatsoever pertaining to matters arising prior to Completion arising by virtue of or in connection with contracts of employment, employment relationships and/or collective agreements in existence at any time prior to Completion. |
| 5.4. | Transfer of Employees: The Vendor shall prior to Completion comply where appropriate with sections 9(2) and 20 of the Redundancy Payments Act, 1967 and shall facilitate the Purchaser in this regard so that continuity, where appropriate, is maintained. |
| 5.5. | Indemnity: The Vendor shall indemnify and keep indemnified each Irish OpCo Group Company on demand against any and all Employment Liabilities arising from: |
| 5.5.1. | a claim made against an Irish OpCo Group Company by any Employee, any person claiming to be an employee, former employee or contractor of the D5 Business or any other person by reason of the Transfer Regulations in circumstances where such claim relates to any act or omission by the Vendor or any other event or occurrence related to or arising out of the employment of such person in each case prior to the Effective Time for which the Irish OpCo Group Company is or may be liable by reason of the Transfer Regulations; |
| 5.5.2. | any cause of action of whatsoever nature by any third party against an Irish OpCo Group Company for which the Irish OpCo Group Company is or may be liable by reason of the Transfer Regulations applying to the sale of the Assets and the Business; |
| 5.6. | Redundancy and retention plan: Subject to the terms of any redundancy and retention package which has been agreed by the Vendor and Nerano Pharma Limited on or prior to the date of this Agreement (the Redundancy and Retention Program), the Vendor shall indemnify and keep indemnified each Irish OpCo Group Company on demand against: |
| 5.6.1. | any and all amounts which an Irish OpCo Group Company pays to any Employee in the event that any Irish OpCo Group Company makes the Employee redundant within 18 months of the Effective Time, provided that the amount which the Vendor shall be required to pay in respect of an Employee shall not exceed the cost of the severance package that the Vendor would have paid to the relevant Employee if his position had been made redundant immediately prior to the Effective Time; and |
| 5.6.2. | the amount due to the Employee as a result of the Employee meeting the retention targets applicable to him in the Redundancy and Retention Program, provided that in no circumstances shall the aggregate liability of the Vendor pursuant to this clause 5.6.2 exceed US$1,200,000. |
provided always that in no circumstances shall the aggregate liability of the Vendor pursuant to this clause 5.6 exceed US$3,000,000;
| 5.7. | If the provisions of the Transfer Regulations operate so as to transfer the employment of a person other than an Employee to an Irish OpCo Group Company: |
| 5.7.1. | the Irish OpCo Group Company shall, on becoming aware of the application of such provisions to such person, inform the Vendor and the Vendor shall within 14 days provide all relevant information to the Irish OpCo Group Company relating to such person and his employment and during that period shall consult with the Irish OpCo Group Company in good faith as to the appropriate steps to be taken by the Vendor and the Irish OpCo Group Company in relation to that person; and |
| 5.7.2. | the Vendor covenants with the Purchaser to pay to the Irish OpCo Group Company on demand an amount equal to: |
| (1) | any and all Employment Liabilities arising out of the employment of such person by the Irish OpCo Group Company or the termination of his/her employment during the period of two years from that person’s Transfer Date, regardless of when a claim is made by such person; |
| (2) | any and all Employment Liabilities arising out of the termination of employment of any of the Irish OpCo Group Company’s employees (who were employed prior to the Effective Time) as a consequence of the transfer to the Irish OpCo Group Company of such person; |
To the extent permitted by law the Purchaser shall use its reasonable endeavours to minimise the amounts payable to the Irish OpCo Group Company pursuant to this clause 5.7.
| 5.8. | Save in respect of those matters where the Vendor is expressly obliged in this clause 5 to indemnify the Irish OpCo Group, the Purchaser shall indemnify and keep indemnified the Vendor on demand against any and all Employment Liabilities arising from a claim made by any Employees where such claim relates to any act or omission by an Irish OpCo Group Company or any other event or occurrence related to or arising out of the employment of such person in each case after the Effective Time. |
| 5.9. | The Vendor agrees to assign or otherwise hold for the Purchaser with effect from the Effective Time the benefit of any confidentiality or other undertakings or restrictions given to the Vendor by any present or former employees of the Vendor in the D5 Business where such undertakings or restrictions or the benefit thereof are not novated to or otherwise vested in an Irish OpCo Group Company by virtue of the Transfer Regulations and accordingly the Vendor hereby agrees pending formal assignment or novation of the same and at the request of the Purchaser to take such steps, actions and proceedings as the Purchaser shall reasonably require to enforce such undertakings and restrictions (or any of them) for the benefit and at the cost of the Purchaser. |
| 6. | LIMIT OF PARTIES’ LIABILITIES |
| 6.1. | The Vendor: |
| 6.1.1. | remains responsible for all Pre-Effective Time Liabilities; |
| 6.1.2. | remains responsible for all Pre-Effective Time Claims; |
| 6.1.3. | shall promptly pay all Pre-Effective Time Liabilities and promptly settle all Pre-Effective Time Claims; and |
| 6.1.4. | shall indemnify and keep indemnified the Purchaser on demand against each loss, liability and reasonable cost (a Specified Claim) which the Purchaser incurs: |
| (1) | in connection with the ownership or operation of the D5 Business or the Assets before the Effective Time; or |
| (2) | as a result of the Vendor’s failure to comply with its obligations under clause 6.1.1, 6.1.2 or 6.1.3. |
| 6.2. | Subject to clause 5, the Purchaser: |
| 6.2.1. | is responsible for all liabilities incurred by it in connection with the D5 Business and the Assets after the Effective Time; |
| 6.2.2. | shall indemnify and keep indemnified the Vendor on demand against each loss, liability and reasonable cost (a Specified Claim) which the Vendor incurs: |
| (1) | in connection with the ownership or operation of the D5 Business or the Assets after the Effective Time; or |
| (2) | as a result of the Purchaser’s failure to comply with its obligations under clause 6.2.1. |
| 6.3. | If a party (the Indemnified Party) becomes aware of a matter which might give rise to a Specified |
Claim the following provisions shall apply:
| 6.3.1. | The Indemnified Party shall notify the other party (the Indemnifier) immediately of the matter (stating in reasonable detail the nature of the matter and, if practicable, the amount claimed) and consult with the Indemnifier with respect to the matter. If the matter has become the subject of proceedings, the Indemnified Party shall notify the Indemnifier within sufficient time to enable the Indemnifier to have time to contest the proceedings before final judgment. |
| 6.3.2. | The Indemnified Party shall: |
| (1) | take any action and institute any proceedings, and give any information and assistance, as the Indemnifier or its insurers may reasonably request to: |
| (a) | dispute, resist, appeal, compromise, defend, remedy or mitigate the matter; or |
| (b) | enforce against a person (other than the Indemnifier) the Indemnified Party’s or its insurers’ rights in relation to the matter; and |
| (2) | in connection with proceedings related to the matter (other than against the Indemnifier) use advisers chosen by the Indemnifier or its insurers and, if the Indemnifier requests, allow the Indemnifier or its insurers the exclusive conduct of the proceedings, |
| (3) | in each case the Indemnifier indemnifies the Indemnified Party for all reasonable costs incurred as a result of a request or choice by the Indemnifier or its insurers. |
| 6.3.3. | The Indemnified Party may only admit liability in respect of or settle the matter if it has first obtained the Indemnifier’s written consent (not to be unreasonably withheld or delayed). |
| 7. | NON SOLICITATION |
The Vendor undertakes with the Purchaser that the Vendor shall not, and shall procure that each of its Affiliates shall not, without the prior written consent of the Purchaser, directly or indirectly, employ, solicit or contact with a view to his/her employment any Employee for a period of two years after Completion.
| 8. | MISCELLANEOUS PROVISIONS |
| 8.1. | Confidentiality |
| 8.1.1. | The Vendor undertakes with the Purchaser that it shall preserve the confidentiality of, and not directly or indirectly reveal, report, publish, disclose, transfer or use for its own or any other purposes Confidential Information except: |
| (1) | in the circumstances set out in clause 8.1.2; |
| (2) | to the extent otherwise expressly permitted by this Agreement; or |
| (3) | with the prior written consent of the Purchaser. |
| 8.1.2. | The Vendor is permitted to disclose Confidential Information: |
| (1) | to an Affiliate; |
| (2) | where the Confidential Information, before it is furnished to the Vendor, is in the public domain; |
| (3) | where the Confidential Information, after it is furnished to the Vendor, enters the public domain otherwise than as a result of: (i) a breach by the Vendor of its obligations in this clause 8.1 or (ii) a breach by the person who disclosed that Confidential Information of a confidentiality obligation where the Vendor is aware of such breach; |
| (4) | if and to the extent that the Vendor discloses Confidential Information to any person: |
| (a) | in compliance with any requirement of law; |
| (b) | in response to a requirement of any securities exchange, regulatory or governmental authority or court having jurisdiction over the relevant Vendor; or |
| (c) | in order to obtain Tax or other clearances or consents from the Revenue Commissioners or other relevant taxing or regulatory authorities, |
provided that any such information disclosable pursuant to paragraphs (a), (b) or (c) may be disclosed only to the extent required by law.
| 8.1.3. | The restrictions contained in this clause 8.1 will continue to apply after Completion and will apply in relation to the existence and provisions of this Agreement and information relating to the Purchaser regardless of whether or not Completion occurs. |
| 8.2. | Severability |
All the terms and provisions of this Agreement are distinct and severable, and if any term or provision is held or declared to be unenforceable, illegal or void in whole or in part by any court, regulatory authority or other competent authority it will to that extent only be deemed not to form part of this Agreement, and the enforceability, legality and validity of the remainder of this Agreement will not in any event be affected. The parties shall then use all reasonable endeavours to agree a term or provision to replace the unenforceable, illegal or void term or provision which is legal and enforceable and which has an effect that is near as possible to the intended effect of the term or provision to be replaced.
| 8.3. | Whole Agreement |
This Agreement (together with any documents to be executed pursuant to the terms of this Agreement) supersedes all prior representations, arrangements, understandings and agreements, and sets out the entire, complete and exclusive agreement and understanding between the parties. The rights of the Purchaser under this Agreement are independent, cumulative and without prejudice to all other rights available to it whether as a matter of common law, statute, custom or otherwise.
| 8.4. | Survival |
The provisions of this Agreement which have not been performed at Completion will remain in full force and effect notwithstanding Completion.
| 8.5. | Remedies Cumulative |
The provisions of this Agreement and the rights and remedies of the parties are cumulative and
are without prejudice and in addition to any rights or remedies which a party may have at law or in equity. The exercise by a party of any one right or remedy under this Agreement or at law or in equity will not (unless expressly otherwise provided in this Agreement or at law or in equity) operate so as to hinder or prevent the exercise by that party of any other right or remedy.
| 8.6. | Waiver |
Any liability to the Purchaser under this Agreement may be wholly or partially released, varied, compounded or compromised by the Purchaser in its absolute discretion as regards the Vendor or any other party without in any way prejudicing or affecting its rights against any other party under the same or a similar liability, whether joint and several or otherwise. A waiver by the Purchaser of any breach by any party of any of the terms, provisions or conditions of this Agreement, or the acquiescence of the Purchaser in any act (whether commission or omission) which but for such acquiescence would be a breach, will not constitute a general waiver of the term, provision or condition or of any subsequent act which is inconsistent with it.
| 8.7. | Further Assurance |
Without limiting the generality of the foregoing, to the extent that the Purchaser or the Vendor discover following Completion that:
| 8.7.1. | any Asset that was intended to be assigned and transferred pursuant to this Agreement was not assigned and transferred at Completion, the Vendor shall (and shall procure that any other necessary parties shall) execute and do all such documents, acts and things as may reasonably be required to assign and transfer to the Purchaser the legal and beneficial ownership in such Asset; and/or |
| 8.7.2. | any asset that was not an Asset intended to be transferred pursuant to this Agreement was assigned and transferred at Completion, the Purchaser shall (and shall procure that any other parties shall) execute and do all such documents, acts and things as may reasonably be required to assign and transfer to the Vendor the legal and beneficial ownership in such asset; and/or |
| 8.7.3. | any liability that was intended to be assumed by the Purchaser pursuant to this Agreement was not assumed by the Purchaser at Completion, the parties shall (and shall procure that any other necessary parties shall) execute and do all such documents, acts and things as may reasonably be required for the Purchaser to assume such liability; and/or |
| 8.7.4. | any liability that was not intended to be assumed by the Purchaser pursuant to this Agreement was assumed by the Purchaser at Completion, the parties shall (and shall procure that any other parties shall) execute and do all such documents, acts and things as may reasonably be required for such liability to be assumed by the Vendor. |
| 8.8. | Notices |
Any notice or other communication to be given or served under this Agreement shall be in writing, addressed to the relevant party and expressed to be a notice or communication under this Agreement and, without prejudice to the validity of another method of service, may be delivered or sent by pre-paid post or facsimile addressed to the recipient at the address given above or to its fax number at that address.
| 8.9. | Counterparts |
This Agreement may be executed in any number of counterparts, and by the several parties to it on separate counterparts, each of which when so executed will constitute an original but all of
which together will evidence the same agreement.
| 8.10. | Governing Law and Jurisdiction |
This Agreement and all relationships created by it will in all respects be governed by and construed in accordance with Irish law. The Irish courts will have exclusive jurisdiction to settle any dispute which may arise out of or in connection with this Agreement or its performance.
| 8.11. | Succession and Assignment |
This Agreement may not be assigned without the consent of the other party except to an Affiliate. This Agreement will be binding upon and enure for the benefit of the permitted assigns and, where applicable, successors in title, administrators, executors and personal representatives of the parties. The benefit of any provision of this Agreement may be enforced by the beneficial owners for the time being of the D5 Business or any of the Assets.
None of the parties to this Agreement may assign any of its rights under this Agreement without the prior written consent of each of the other parties except that any party is entitled to assign the benefit of any provision of this Agreement to an Affiliate.
| 8.12. | Variations |
This Agreement may not be released, varied, discharged, amended or supplemented, except by an instrument in writing executed by each party or a duly authorised representative of each party.
| 8.13. | Financial Transfers |
The Vendor declares for the purpose of the Financial Transfers Act, 1992 that it is not resident in any jurisdiction to which financial transfers (within the meaning of that Act) are restricted by order of the Minister for Finance in accordance with the provisions of that Act and does not hold any of the Assets, and will not receive any part of the Consideration, as nominee for any persons so resident, and the Purchaser declares for the purpose of that Act that it is not so resident, is not acquiring the Assets as nominee for any persons so resident and that the Purchaser is not to its knowledge controlled directly or indirectly by persons so resident.
| 8.14. | Recitals |
The parties acknowledge that matters referred to in the recitals will not, in any way, give rise to any liability or cause of action against the Purchaser.
SCHEDULE 1
Excluded Assets
For the avoidance of doubt, the Excluded Assets of the Vendor means:
| (1) | all those assets and records of the Vendor which do not exclusively form part of the D5 Business; |
| (2) | all cash and bank accounts of the Vendor; and |
| (3) | the statutory books of the Vendor. |
SCHEDULE 2
| A. | Owned Patents |
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00667-US-NP | US | FLG | 13 Oct 2011 | 13/273,069 | ||||||
| 00667-US-PSP | US | EXP | 13 Oct 2010 | 61/455,089 | ||||||
| 00667-WO-PCT | WO | PUB | 13 Oct 2011 | PCT/US2011/056109 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00681-US-PSP | US | EXP | 03 Jun 2011 | 61/520,031 | ||||||
| 00681-US-PSP[3] | US | FLG | 31 Mar 2012 | 61/618,680 | ||||||
| 00681A1-US | US | FLG | 04 Jun 2012 | 13/488,363 | ||||||
| 00681A1W1-PCT | WO | FLG | 04 Jun 2012 | PCT/US2012/40789 | ||||||
| 00681A2 | US | FLG | 30 Sep 2011 | 61/541,333 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00692A1 | US | FLG | 15 Feb 2010 | 61/304,581 | ||||||
| 00692A1W1-AU | AU | DES | ||||||||
| 00692A1W1-CA | CA | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-CN | CN | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-EA | EA | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-EP | EP | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-JP | JP | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-NZ | NZ | DES | ||||||||
| 00692A1W1-PCT | WO | PUB | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00695-A1-US | US | FLG | 28 Sep 2012 | 13/573,640 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00700-prov1 | US | FLG | 09 Jan 2013 | 61/750,722 |
| B. | Licensed Patent Rights |
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00592-CA-PCT | CA | FLG | 13 Oct 2006 | 2626005 | ||||||
| 00592-EP-EPT | EP | FLG | 13 Oct 2006 | 06850456.2 | ||||||
| 00592-JP-PCT | JP | PUB | 13 Oct 2006 | 2008-535136 | ||||||
| 00592-US-CNT[4] | US | ABD | 11 Nov 2009 | 12/616,496 | ||||||
| 00592-US-NP[2] | US | ABD | 13 Oct 2006 | 11/580,026 | ||||||
| 00592-US-PCT[3] | US | FLG | 13 Oct 2006 | 12/090,130 | ||||||
| 00592-US-PSP | US | EXP | 13 Oct 2005 | 60/725,634 | ||||||
| 00592-WO-PCT | WO | FLG | 13 Oct 2006 | PCT/US2006/039872 | ||||||
| 00592-WO-PCT[2] | WO | OPEN | 13 Oct 2006 | PCT/IB2006/004181 | ||||||
| 00592-WO-PCT[3] | WO | PUB | 13 Oct 2006 | PCT/CA2006/001679 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00593-AU-PCT | AU | GRT | 17 Nov 2005 | 2005 306 531 | 2005 306 531 | |||||
| 00593-BR-PCT | BR | FLG | 17 Nov 2005 | PI0517733-2 | ||||||
| 00593-CA-PCT | CA | FLG | 17 Nov 2005 | 2588423 | ||||||
| 00593-CN-PCT | CN | FLG | 17 Nov 2005 | 200580046701.8 | ||||||
| 00593-EP-EPD[2] | EP | ABD | 17 Nov 2005 | 10000985.1 | ||||||
| 00593-EP-EPT | EP | ABD | 17 Nov 2005 | 05808109.2 | ||||||
| 00593-IN-PCD | IN | FLG | 17 Nov 2005 | 90/KOLNP/2012 | ||||||
| 00593-IN-PCT | IN | FLG | 17 Nov 2005 | 1887/KOLNP/2007 | ||||||
| 00593-JP-PCT | JP | PUB | 17 Nov 2005 | 2007-541601 | ||||||
| 00593-MX-PCT | MX | FLG | 17 Nov 2005 | MX/a/2007/005870 | ||||||
| 00593-PH-PCT | PH | FLG | 17 Nov 2005 | 1-2007-501053 | ||||||
| 00593-US-CNT[4] | US | ABD | 20 Aug 2007 | 11/841,773 | ||||||
| 00593-US-CNT[5] | US | ABD | 20 Aug 2007 | 11/841,745 | ||||||
| 00593-US-CNT[6] | US | ABD | 20 Aug 2007 | 11/841,763 | ||||||
| 00593-US-CNT[7] | US | ABD | 20 Aug 2007 | 11/841,730 | ||||||
| 00593-US-CNT[8] | US | ABD | 22 May 2008 | 12/125,498 | ||||||
| 00593-US-CNT[9] | US | PUB | 15 Sep 2010 | 12/882,250 | ||||||
| 00593-US-NP[3] | US | ABD | 17 Nov 2005 | 11/280,818 | ||||||
| 00593-US-PCT[2] | US | ABD | 17 Nov 2005 | 11/667,822 | ||||||
| 00593-US-PSP | US | EX-C | 17 Nov 2004 | 60/628,840 | ||||||
| 00593-WO-PCT | WO | PUB | 17 Nov 2005 | PCT/CA2005/001744 | ||||||
| 00593-ZA-DIV[2] | ZA | FLG | 17 Nov 2005 | 2008/08231 | ||||||
| 00593-ZA-PCD | ZA | FLG | 17 Nov 2005 | 2012/02636 | ||||||
| 00593-ZA-PCT | ZA | GRT | 17 Nov 2005 | 2007/04872 | 2007/04872 | |||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00594-AT-EPT | AT | GRT | 27 Feb 2004 | 04715226.9 | E 432 694 | |||||
| 00594-AU-DIV | AU | FLG | 27 Feb 2004 | 2009251035 | ||||||
| 00594-AU-PCT[2] | AU | GRT | 27 Feb 2004 | 2004216544 | 2004216544 | |||||
| 00594-BE-EPT | BE | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-BG-EPT | BG | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-BR-PCT | BR | FLG | 27 Feb 2004 | PI 0407910-8 | ||||||
| 00594-CA-PCT | CA | FLG | 27 Feb 2004 | 2516563 | ||||||
| 00594-CH-EPT | CH | GRT | 27 Feb 2004 | 04715226.9 | 1608350 |
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00594-CN-DIV | CN | FLG | 27 Feb 2004 | 200810146321.7 | ||||||
| 00594-CN-DIV[3] | CN | ABD | 27 Feb 2004 | N/A | ||||||
| 00594-CN-PCT[2] | CN | GRT | 27 Feb 2004 | 20048011335.8 | ZL 200480011335.8 | |||||
| 00594-CY-EPT | CY | GRT | 27 Feb 2004 | 04715226.9 | CY1109338 | |||||
| 00594-CZ-EPT | CZ | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-DE-EPT | DE | GRT | 27 Feb 2004 | 04715226.9 | 602004021362.4-08 | |||||
| 00594-DK-EPT | DK | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-EE-EPT | EE | GRT | 27 Feb 2004 | 04715226.9 | E003407 | |||||
| 00594-EP-EPD | EP | PUB | 27 Feb 2004 | 09005295.2 | ||||||
| 00594-EP-EPD[2] | EP | PUB | 27 Feb 2004 | 10010477.7 | ||||||
| 00594-EP-EPT[3] | EP | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-ES-EPT | ES | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-FI-EPT | FI | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-FR-EPT | FR | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-GB-EPT | GB | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-GR-EPT | GR | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-HU-EPT | HU | GRT | 27 Feb 2004 | 04715226.9 | E006366 | |||||
| 00594-IE-EPT | IE | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-IL-PCD[1] | IL | FLG | 27 Feb 2004 | 220065 | ||||||
| 00594-IL-PCT | IL | FLG | 27 Feb 2004 | 170476 | ||||||
| 00594-IT-EPT | IT | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-JP-PCD | JP | PUB | 27 Feb 2004 | 2010-294145 | ||||||
| 00594-JP-PCT[2] | JP | GRT | 27 Feb 2004 | 2006-501433 | 4999453 | |||||
| 00594-LU-EPT | LU | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-NL-EPT | NL | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-PT-EPT | PT | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-RO-EPT | RO | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-SE-EPT | SE | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-SI-EPT | SI | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-SK-EPT | SK | GRT | 27 Feb 2004 | 04715226.9 | E 6042 | |||||
| 00594-TR-EPT | TR | GRT | 27 Feb 2004 | 04715226.9 | TR 2009 06231 T4 | |||||
| 00594-US-CIP[5] | US | PUB | 27 Feb 2004 | 10/547,286 | ||||||
| 00594-US-CNT[6] | US | ABD | 03 Mar 2009 | 12/396,515 | ||||||
| 00594-US-CNT[7] | US | PUB | 29 Feb 2012 | 13/408,337 | ||||||
| 00594-US-NP[4] | US | GRT | 26 Feb 2004 | 10/787,621 | 7,521,481 | |||||
| 00594-US-PSP | US | EX-C | 27 Feb 2003 | 60/451,363 | ||||||
| 00594-US-PSP[2] | US | EX-C | 17 Nov 2003 | 60/520,958 | ||||||
| 00594-US-PSP[3] | US | EX-C | 19 Nov 2003 | 60/523,534 | ||||||
| 00594-WO-PCT | WO | PUB | 27 Feb 2004 | PCT/CA2004/00272 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00601-CA-PCT | CA | ABD | 16 Feb 2007 | 2642647 | ||||||
| 00601-EP-EPT | EP | ABD | 16 Feb 2007 | 07789484.8 | ||||||
| 00601-JP-PCT | JP | ABD | 16 Feb 2007 | 2008-554886 | ||||||
| 00601-US-NP[2] | US | ABD | 16 Feb 2007 | 11/707,156 | ||||||
| 00601-US-PSP | US | EX-C | 17 Feb 2006 | 60/774,818 | ||||||
| 00601-WO-PCT[2] | WO | ABD | 16 Feb 2007 | PCT/IB2007/001929 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. |
| 00615-CA-PCT | CA | ABD | 18 May 2007 | 2652449 | ||||||
| 00615-EP-EPT | EP | ABD | 18 May 2007 | 07719822.4 | ||||||
| 00615-US-PCT[4] | US | PUB | 18 May 2007 | 12/301,155 | ||||||
| 00615-US-PSP | US | EX-C | 19 May 2006 | 60/801,848 | ||||||
| 00615-US-PSP[2] | US | EX-C | 12 Oct 2006 | 60/851,256 | ||||||
| 00615-US-PSP[3] | US | EX-C | 13 Nov 2006 | 60/858,548 | ||||||
| 00615-WO-PCT | WO | ABD | 18 May 2007 | PCT/CA2007/000900 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00627-US-PCT[2] | US | ABD | 20 Sep 2007 | 12/438,572 | ||||||
| 00627-US-PSP | US | EX-C | 21 Sep 2006 | 60/846,366 | ||||||
| 00627-WO-PCT | WO | ABD | 20 Sep 2007 | PCT/CA2007/001678 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00629-AU-PCT | AU | FLG | 09 Mar 2007 | 2007222864 | ||||||
| 00629-BR-PCT | BR | PUB | 09 Mar 2007 | PI0708725-0 | ||||||
| 00629-CA-PCT | CA | FLG | 09 Mar 2007 | 2644804 | ||||||
| 00629-CN-PCD | CN | FLG | 09 Mar 2007 | 201210257027.X | ||||||
| 00629-CN-PCT | CN | PUB | 09 Mar 2007 | 200780016972.8 | ||||||
| 00629-CO-PCT | CO | PUB | 09 Mar 2007 | 08.108.264 | ||||||
| 00629-EA-EAT | EA | FLG | 09 Mar 2007 | 200801967 | ||||||
| 00629-EP-EPT | EP | PUB | 09 Mar 2007 | 07710726.6 | ||||||
| 00629-ID-PCT | ID | FLG | 09 Mar 2007 | W00200803173 | ||||||
| 00629-IL-PCT | IL | FLG | 09 Mar 2007 | 193970 | ||||||
| 00629-IN-PCT | IN | PUB | 09 Mar 2007 | 3723/KOLNP/2008 | ||||||
| 00629-JP-PCT | JP | FLG | 09 Mar 2007 | 2008-557568 | ||||||
| 00629-KR-PCT | KR | FLG | 09 Mar 2007 | 1-2008-7024122 | ||||||
| 00629-MX-PCT | MX | FLG | 09 Mar 2007 | MX/a/2008/011553 | ||||||
| 00629-NZ-PCT | NZ | GRT | 09 Mar 2007 | 571,181 | 571,181 | |||||
| 00629-PH-PCT | PH | FLG | 09 Mar 2007 | 1-2008-502019 | ||||||
| 00629-US-PCT[2] | US | PUB | 09 Mar 2007 | 12/282,030 | ||||||
| 00629-US-PSP | US | EX-C | 09 Mar 2006 | 60/780,526 | ||||||
| 00629-WO-PCT | WO | PUB | 09 Mar 2007 | PCT/CA2007/000395 | ||||||
| 00629-ZA-DIV | ZA | ALL | 09 Mar 2007 | 2009/08303 | ||||||
| 00629-ZA-PCT[2] | ZA | GRT | 09 Mar 2007 | 2008/07993 | 2008/07993 | |||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00632-CA-PCT | CA | FLG | 22 Nov 2007 | 2670405 | ||||||
| 00632-EP-EPT | EP | PUB | 22 Nov 2007 | 07845583.9 | ||||||
| 00632-JP-PCT | JP | PUB | 22 Nov 2007 | 2009-537460 | ||||||
| 00632-US-PCT | US | PUB | 22 Nov 2007 | 12/445,164 | ||||||
| 00632-WO-PCT | WO | PUB | 22 Nov 2007 | PCT/CA2007/002118 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00633-CA-PCT | CA | FLG | 11 Apr 2008 | 2683580 | ||||||
| 00633-US-PCT[3] | US | PUB | 11 Apr 2008 | 12/594,947 | ||||||
| 00633-US-PSP | US | EX-C | 12 Apr 2007 | 60/923,061 | ||||||
| 00633-US-PSP[2] | US | EX-C | 12 Apr 2007 | 60/923,001 | ||||||
| 00633-WO-PCT | WO | PUB | 11 Apr 2008 | PCT/CA2008/000685 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00634-CA-PCT | CA | FLG | 11 Apr 2008 | 2683546 | ||||||
| 00634-US-PCT[3] | US | PUB | 11 Apr 2008 | 12/594,950 | ||||||
| 00634-US-PSP | US | EX-C | 12 Apr 2007 | 60/923,150 | ||||||
| 00634-US-PSP[2] | US | EX-C | 12 Apr 2007 | 60/923,044 | ||||||
| 00634-WO-PCT | WO | PUB | 11 Apr 2008 | PCT/CA2008/000683 |
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00635-AU-PCT | AU | FLG | 11 Apr 2008 | 2008 238 577 | ||||||
| 00635-CA-PCT | CA | FLG | 11 Apr 2008 | 2683607 | ||||||
| 00635-EP-EPT | EP | PUB | 11 Apr 2008 | 08733752.3 | ||||||
| 00635-JP-PCT | JP | ABD | 11 Apr 2008 | 2010-502395 | ||||||
| 00635-US-PCT[2] | US | PUB | 11 Apr 2008 | 12/594,951 | ||||||
| 00635-US-PSP | US | EX-C | 12 Apr 2007 | 60/922,998 | ||||||
| 00635-WO-PCT | WO | FLG | 11 Apr 2008 | PCT/CA2008/000703 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00636-CA-PCT | CA | FLG | 11 Apr 2008 | 2683548 | ||||||
| 00636-US-PCT[3] | US | PUB | 11 Apr 2008 | 12/594,943 | ||||||
| 00636-US-PSP | US | EX-C | 12 Apr 2007 | 60/923,002 | ||||||
| 00636-US-PSP[2] | US | EX-C | 12 Apr 2007 | 60/923,120 | ||||||
| 00636-WO-PCT | WO | FLG | 11 Apr 2008 | PCT/CA2008/000684 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00643-AU-PCT | AU | FLG | 09 Oct 2009 | 2009 301 603 | ||||||
| 00643-CA-PCT | CA | FLG | 09 Oct 2009 | 2740124 | ||||||
| 00643-EP-EPT[1] | EP | PUB | 09 Oct 2009 | 09818730.5 | ||||||
| 00643-JP-PCT | JP | PUB | 09 Oct 2009 | 2011-530339 | ||||||
| 00643-NZ-PCT | NZ | FLG | 09 Oct 2009 | 592277 | ||||||
| 00643-US-NP[2] | US | PUB | 09 Oct 2009 | 12/576,957 | ||||||
| 00643-US-PSP | US | EX-C | 09 Oct 2008 | 61/104,094 | ||||||
| 00643-WO-PCT | WO | PUB | 09 Oct 2009 | PCT/CA2009/001448 | ||||||
| 00643-ZA-PCT | ZA | FLG | 09 Oct 2009 | 2011/02640 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00650-AR-NP | AR | FLG | 15 Sep 2008 | P090103535 | ||||||
| 00650-AU-PCT | AU | FLG | 15 Sep 2009 | 2009290612 | ||||||
| 00650-CA-PCT[2] | CA | FLG | 15 Sep 2009 | 2737163 | ||||||
| 00650-EP-EPT[2] | EP | PUB | 15 Sep 2009 | 09792556.4 | ||||||
| 00650-JP-PCT[2] | JP | PUB | 15 Sep 2009 | 2011-527043 | ||||||
| 00650-TW-NP | TW | PUB | 15 Sep 2009 | 98131051 | ||||||
| 00650-US-NP[2] | US | PUB | 15 Sep 2009 | 12/560,113 | ||||||
| 00650-US-PSP | US | EX-C | 15 Sep 2008 | 61/097,023 | ||||||
| 00650-VE-NP | VE | FLG | 15 Sep 2008 | N/A | ||||||
| 00650-WO-PCT | WO | FLG | 15 Sep 2009 | PCT/US2009/057003 | ||||||
| 00650-WO-PCT[2] | WO | PUB | 15 Sep 2009 | PCT/US2009/056985 | ||||||
SCHEDULE 3
Business Contracts
| 1. | Collaboration Agreement between Waratah Pharmaceuticals Inc. and Elan Pharma International Limited dated 25 September 2006 (as amended); |
| 2. | Master Services and Alliance Agreement between PPD Development, LP and Elan Pharmaceuticals, Inc. dated 1 August 2011; and |
| 3. | Master Services Agreement between Clinical Financial Services, LLC and Elan Pharmaceuticals, Inc. dated 2 April 2012. |
IN WITNESS whereof this Agreement has been entered into on the date and year first herein written.
| SIGNED for and on behalf of | |
| ELAN PHARMA INTERNATIONAL LIMITED | |
| Director | |
| SIGNED for and on behalf of | |
| SPERANZA BIOPHARMA LIMITED | |
| Director |
EXHIBIT B
Royalty and Option Agreement
Agreed Form – Subject to Contract/ Contract Denied
Dated May 2013
ELAN PHARMA INTERNATIONAL LIMITED
and
SPERANZA BIOPHARMA LIMITED
OPTION AND ROYALTY DEED OF AGREEMENT
A & L GOODBODY
THIS OPTION AND ROYALTY DEED (the Agreement) is dated [·] 2013 and made between
| (1) | ELAN PHARMA INTERNATIONAL LIMITED (registered no. 222276) having its registered office at Treasury Building, Grand Canal Street Lower, Dublin 2 (the Company); and |
| (2) | SPERANZA BIOPHARMA LIMITED a company incorporated in Ireland (Registered No. 502444) whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2 (Newco). |
Recitals:
| A. | Pursuant to a Business Transfer Agreement dated the date hereof between the Company and Newco (the Business Transfer Agreement), the Company has agreed to transfer all assets relating to the Product (as defined below) to Newco. |
| B. | Newco has agreed to grant to the Company the option described in Clause 2 and to pay the Royalty described in Clause 6 to the Company. |
IT IS HEREBY AGREED as follows:
| 1. | PRELIMINARY |
| 1.1. | Definitions: In this Agreement and in the Schedules unless the context otherwise requires: |
Affiliate means another person that directly or indirectly controls, is controlled by, or is under common control with, such first person, where control means the possession, directly or indirectly, of the power to direct or cause the direction of the business, affairs or management of a person, whether through the ownership of voting securities, by contract, as trustee or executor, or otherwise;
Application means, without limitation, Down Syndrome, Bipolar Disorder and/or Alzheimer's agitation and/or any other potential future use or application which is now known (or may in the future be known) in respect of the Patents and/or Know-How and/or any Product;
Business Days means any day other than a Saturday or Sunday or a bank or public holiday in the Republic of Ireland;
Commercialisation means any and all activities constituting importing, manufacturing, marketing, distributing, offering for sale and/or selling a Product and shall include but is not be limited to:
| (a) | sales; |
| (b) | loaning, leasing, letting on hire or sales on hire purchase; |
| (c) | supply; |
| (d) | putting into use; or |
| (e) | Promotion |
as well as activities required to fulfil on-going regulatory obligations, including adverse event reporting. When used as a verb, Commercialise shall mean to engage in Commercialisation;
Confidential Information means know-how, trade secrets, inventions (including, where applicable, information in relation to the Patents, Know-How and/or any other potential or actual patent applications covering inventions), data, information, and any improvements, modifications, derivations, or compilations thereto that is owned, licensed by or controlled by the disclosing party, provided however, that Confidential Information shall not include any information which is:
| 2 |
| (1) | already known to the receiving party at the time of disclosure, as evidenced by such party's written records, provided such information was not obtained directly or indirectly by the receiving party from the disclosing party pursuant to a confidentiality agreement; |
| (2) | publicly known prior to or after disclosure, through no default of the receiving party; |
| (3) | disclosed in good faith to the receiving party by a third party, lawfully and contractually entitled to make such disclosure; or |
| (4) | is independently discovered without the aid or application of the Confidential Information as shall be evidenced by the written records of the receiving party. |
Derivative means any compounds identified, obtained, developed, created, synthesized, generated, designed or resulting from, based upon, containing or incorporating the chemical structure of Scyllo-Inositol as those compounds are generically and/or specifically disclosed and claimed in the Licensed Patent Rights;
D5 Business means the research, development, manufacture and/or Commercialisation of a Product;
Effective Date means the date of this Agreement;
Elan Group means the Company and its Subsidiaries from time to time;
European Option Territories means the countries listed in Part A of Schedule 3;
Fair Market Value means the fair value price, licence, distribution and/or sales fee and/or a royalty or any of the foregoing (or similar) as may be applicable to the Commercialisation of the Patents and/or Know-How and/or any Product (in any way) on the open market in the Other Territories, on the basis of an arm's length transaction entered into by Newco (or any Affiliate of Newco).
Field means any use or Application or potential future use or application which is now known (or may in the future be known) in respect of the Patents and/or Know-How and/or any Product;
Generic Product a drug product that has the same or substantially the same labelling as the Product and, to the extent applicable, that is lawfully substitutable by pharmacists or other dispensers of pharmaceuticals for the Product when the Product is prescribed;
Generics Territory means any country listed in Part C of Schedule 3;
Group means Newco and its Subsidiaries from time to time;
Know-How means any and all rights related to the Product and /or the Patents, owned, licensed or controlled by Newco to include any scientific, pharmaceutical or technical information, data, discovery, invention (whether patentable or not), know-how, substances, techniques, processes, systems, formulations, designs and expertise which is not generally known to the public;
Licensed Patent Rights means the Patents listed in Part B of Schedule 1;
Milestone Events means the events described in clauses 5.1.1 and 5.1.2 and "Milestone Event" means either one of them;
| 3 |
Net Income means any amount paid to NewCo or any of its Affiliates by a third party who is not an Affiliate of NewCo in respect of the use, sale, licence, rental and/or supply of the Patents and/or Know How and/or any Product, less provisions for the following:
| (1) | reasonable and bona fide returns, credits, refunds, rebates, chargebacks, wholesalers or distribution fees, retroactive price adjustments, and any other substantially similar allowances which effectively reduce the net selling price; |
| (2) | reasonable and bona fide transportation and distribution charges or allowances, including freight pickup allowances actually paid or incurred by NewCo, or any of its Affiliates (excluding amounts reimbursed by third party customers); and |
| (3) | any tax (excluding taxes on income), excise or other governmental charges upon or measured by the production, sale, transportation, delivery or use of the Product actually paid by NewCo or any of its Affiliates. |
Non-European Option Territories means the countries listed in Part B of Schedule 3;
Option Territories means the European Option Territories and the Non-European Option Territories, and Option Territory shall mean any of them;
Other Territories means all other countries, nations, principalities or jurisdictions anywhere in the world which are not Option Territories, together with any Option Territory which does not form part of the Selected Option Territories, as those terms are defined under this Agreement;
Owned Patents means the Patents listed in Part A of Schedule 1;
Patents means any and all rights under any and all patent applications and/or patents anywhere in the world, now existing, currently pending or hereafter filed or obtained or licensed by Newco relating to the Product and any sub-divisions, divisions or extensions of same, including but not limited to Owned Patents and the Licensed Patent Rights;
Product means any pharmaceutical preparation or dosage form containing Scyllo-Inositol or a Derivative thereof;
Promotion means those activities, including, without limitation, detailing normally undertaken by a pharmaceutical company's sales force to implement marketing plans and strategies aimed at encouraging the appropriate use of a particular Product in a specific indication. When used as a verb, Promote shall mean to engage in such activities;
Realisation has the meaning given to it in the shareholders' agreement made between, inter alia, Elan Science One Limited and Nerano Pharma Limited to regulate the parties' relationship as shareholders in Speranza Therapeutics Limited;
Regulatory Approval means first approval to manufacture, market and sell a Product issued by either the US Food and Drug Administration and/or the European Medicines Agency;
Relevant Territory means a country within the Other Territories.
Royalty shall mean 3% of any Net Income received by Newco (or any Affiliate of Newco) at any time as a result of and/or due to the use, sale, license, rental and/or supply of the Patents and/or Know-How and/or any Product (in any way) from the Effective Date:
| (a) | anywhere in the world (in circumstances where the Licence Option is not taken up by the Company in accordance with clause 2 herein); or |
| (b) | in the Other Territories (in circumstances where the Licence Option is taken up by the |
| 4 |
| Company in accordance with clause 2 herein); |
and the term "Royalties" shall be construed accordingly;
Scyllo-Inositol is the compound “AZD-103”, as more specifically disclosed in the Licensed Patent Rights;
Selected Option Territories shall have the meaning given to that term in Clause 2.4;
Subsidiary means in relation to either party, each and any subsidiary or holding company of that Party and each and any subsidiary of a holding company of that party or any business entity from time to time controlling, controlled by, or under common control with, either party.
Trigger Event shall occur in relation to a Relevant Territory when the following occurs:
| (a) | the circumstances referred to in clause 6.12.2 apply; |
| (b) | a third party (other than a Newco Subsidiary) sells a Generic Product in a Generics Territory; |
| (c) | the Generic Product does not infringe a Valid Patent Claim; and |
| (d) | the last Valid Patent Claim in that Relevant Territory has expired; |
Valid Patent Claim means any claim of an issued and unexpired Patent which has not been held permanently revoked, unenforceable, unpatentable or invalid by a decision of a court or government agency of competent jurisdiction, which decision is unappealable or unappealed within the time allowed for appeal, and which has not been abandoned, disclaimed, denied or expressly admitted by the holder of the patent or supplementary protection certification to any person to be invalid or unenforceable through reissue or disclaimer or otherwise;
Waratah means Waratah Pharmaceuticals Inc, a US company with offices at Suit 220, 101 Colleget Street, Toronto, Ontario, M5G 1L7 and/or its successors or assigns under the Waratah Agreement; and
Waratah Agreement means the Collaboration Agreement between Elan Pharma International Limited and Waratah Pharmaceuticals Inc. dated 25 September 2006 in the form as amended as of the Effective Date of this Agreement.
| 1.2. | The Schedules referred to in this Agreement form an integral part of this Agreement, and references to this Agreement include reference to them. |
| 1.3. | Headings are inserted for convenience only and do not affect the construction of this Agreement. |
| 1.4. | All references in this Agreement to costs, charges or expenses include any value added tax or similar tax charged or chargeable on them. |
| 1.5. | Unless expressly stated in this Agreement or the context otherwise requires, in this Agreement: |
| 1.5.1. | references to persons are deemed to include references to natural persons, firms, partnerships, companies, corporations, associations, bodies corporate, trusts and investment funds (in each case whether or not having a separate legal personality) but references to individuals are deemed to be references to natural persons only; |
| 1.5.2. | words importing the singular include the plural and vice versa and words importing the masculine include references to the feminine and neuter and vice versa; |
| 5 |
| 1.5.3. | reference to writing or similar expressions includes transmission by facsimile or electronic means; |
| 1.5.4. | a word or phrase the definition of which is contained or referred to in section 2 of the 1963 Act has the meaning attributed to it by that definition; |
| 1.5.5. | references to Acts, statutory instruments and other legislation are to legislation operative in Ireland and to such legislation, modified, consolidated, amended or re-enacted (whether before or after the date of this Agreement) and any subordinate legislation made under that legislation; |
| 1.5.6. | reference to any Irish legal term, concept, legislation or regulation (including, those for any action, remedy, method of judicial proceeding, document, statute, court official, governmental authority or agency) or any accounting term or concept, in respect of any jurisdiction other than Ireland will be construed as a reference to the term or concept which most nearly corresponds to it in that jurisdiction; |
| 1.5.7. | reference to this Agreement includes this Agreement as amended or supplemented from time to time; and |
| 1.5.8. | “including” and “includes” mean including or includes without limiting the generality of the foregoing. |
| 2. | COMMERCIALISATION OPTION |
| 2.1. | From the Effective Date until one (1) year after the date on which the first Regulatory Approval in respect of any Application in relation to any Product is granted (the Option Period), Newco shall, in consideration of the mutual covenants of the parties set forth herein and other lawful consideration, grant to the Company an exclusive option (the Licence Option) to receive an exclusive, royalty-free, fully paid up, unrestricted, sub-licensable, assignable, perpetual licence/sub-licence whereby Newco will grant the Company a licence/sub-licence to the Patents and the Know-How to Commercialise Products in the Selected Option Territories in the Fields (or any specific field) on the terms contained in the template agreement attached at Schedule 2 of this Agreement ("Licence") and on the basis of the binding terms of this Agreement. |
| 2.2. | If the Company does not exercise the Licence Option, the Company shall be entitled to elect to negotiate in good faith exclusively with Newco during the Option Period an alternative form of collaboration or commercialisation agreement such as, but not limited to, a co-promotion or co-marketing arrangement for further development of the Products in the Fields in the Option Territories. |
| 2.3. | If the Company decides to exercise the Licence Option, the Company shall provide Newco with written notice of the exercise of the Licence Option substantively in the form attached at Schedule 4 of this Agreement (Licence Option Exercise Notice), at any time during the Option Period. |
| 2.4. | The Company shall specify in the Licence Option Exercise Notice the Option Territories in which it wishes to be granted the Licence Option, such countries to be referred to herein as the Selected Option Territories. Any Option Territory which is not a Selected Option Territory shall fall to be considered from the date of service of the Licence Option Exercise Notice as forming part of the Other Territories for the purposes of this Agreement. |
| 2.5. | The Company shall be responsible for procuring consents (if any) that are required for the exercise of the Licence Option in respect of the sub-licence of the Licensed Patent Rights granted under clause 2.1. |
| 6 |
| 2.6. | Newco will provide the Company with all reasonable assistance as the Company may request in respect of the requirement (if any) to obtain consent pursuant to clause 2.5 above. |
| 2.7. | Subject to any mechanisms agreed between the parties on or prior to the Effective Date and to the parties being reasonably satisfied that the following is in compliance with applicable law, where the Selected Option Territories include European Option Territories, the Company agrees and undertakes that it will acquire all Products which it requires for Commercialisation in all Fields in those European Option Territories from NewCo, and the parties agree to negotiate in good faith in order to agree the terms upon which NewCo will supply the Company with Products in those circumstances. Such terms will take account, where relevant, of trade dynamics, distribution control, patient access and pharmaceutical controls and applicable laws. |
| 2.8. | During the Option Period, Newco will not negotiate in any form, directly or indirectly, with any other corporation, entity or person in relation to the subject matter of Clause 2.1 and Clause 2.2, nor provide any information relating thereto to third parties, save with the prior written consent of the Company. |
| 2.9. | For the avoidance of doubt, the Company shall not pay or be obliged to pay any royalty or other revenue to Newco arising from any Commercialisation by the Company of the Patents, Know-How and/or Products in the Option Territories. |
| 2.10. | If, during the Option Period, the Company decides not to exercise the License Option, it shall promptly notify NewCo in writing of its decision, whereupon clauses 2.1 and 2.2 of this Agreement shall cease to apply and NewCo shall be free immediately to Commercialise the Products in the Field on a worldwide basis subject to the Royalty payments to the Company in accordance with clause 5.1. |
| 3. | NON-COMPETITION |
| 3.1. | During the Option Period and for the duration of any licence or sub-licence entered into between the parties pursuant to the exercise of the Licence Option, Newco shall not Commercialise the Products in the Fields in the Option Territories. |
| 3.2. | Nothing in this Agreement shall prevent Newco, either alone or in association with a third party, from Commercialising the Products in the Other Territories. |
| 4. | COSTS |
| 4.1. | Both parties shall pay and cover in full their own costs arising from the exercise by the Company of the Licence Option and/or arising from the terms contained in clause 5 of this Agreement. |
| 5. | MILESTONES |
| 5.1. | Newco shall, within thirty (30) days on the occurrence of the following Milestone Events (or either of them) pay to the Company: |
| 5.1.1. | two hundred million US dollars ($200,000,000) on a Realisation or (other than in respect of an intra-Group reorganisation) the legal, beneficial or legal and beneficial sale, transfer, assignment, licence, sub-licence, novation, distribution or other disposal of all or substantially all of the D5 Business by the Group subject to deduction of any milestone amount paid pursuant to the Waratah Agreement after the Effective Date; and |
| 5.1.2. | two hundred million US dollars ($200,000,000) on the attainment by the Group of annual worldwide Net Income of one and a half billion US dollars ($1,500,000,000). For the avoidance of doubt this Milestone as set forth in this clause 5.1.2 shall be paid to the Company only once on the attainment of this Milestone Event. |
| 7 |
| 6. | ROYALTIES |
| 6.1. | Newco shall, from the Effective Date, on a quarterly basis, pay the Company the Royalty. |
| 6.2. | Newco will use all reasonable endeavours to achieve Fair Market Value in respect of any use, sale, license, rental and/or supply of the Patents and/or Know-How and/or any Product in the Other Territories and without limiting the foregoing, will not offer or make any unreasonable or non-customary discounts in respect of the use, sale, licence, rental and/or supply of the Patents and/or Know-How and/or Product in the Other Territories. |
| 6.3. | On the occurrence of a Trigger Event with respect to a Relevant Territory, the Royalty together with any other royalty payable by Newco to any member of the Elan Group shall, upon the first day of the first quarter thereafter, be reduced by 50% in respect of any Net Income solely and exclusively related to any Commercialisation in the Relevant Territory. |
| 6.4. | Assuming the conditions as set forth in 6.3 above are met, the reductions in Royalty as set forth in clause 6.3 are only applicable on a Relevant Territory by Relevant Territory basis in the Other Territories. |
| 6.5. | For the avoidance of doubt, a Royalty accrues on the date when the Patents, Know-How and/or Product is used, sold, licensed, rented and/or supplied, the date it is supplied being the earliest of when it is invoiced, paid for, used, installed or delivered. |
| 6.6. | The Royalty shall be payable under this Agreement even if some part of the manufacture, supply, licence, rental or putting into use of the Patents, Know-How and/or Product by Newco takes place in a part of the Other Territory where there is no Patent or where the Product does not fall within the scope of any Patent. |
| 6.7. | The Royalty and other sums payable under this agreement are exclusive of VAT (or similar tax). If laws, rules or regulations require withholding of income taxes or other taxes imposed upon any payments to be made under this Agreement, the affected party shall make such withholding payments as required and subtract such withholding payments from such payments, except to the extent that the such taxes are greater than they would have been had the original payor (i.e., the Company or Newco, as the case may be) not (A) transferred its rights or obligations under this Agreement to any non-Irish resident (for tax purposes) person or entity, or (B) changed its jurisdiction of incorporation or residence for tax purposes, in either of which case the payor shall pay the payee such additional amounts so that after all deduction or withholding in respect of taxes, the payee receives an amount equal to the sum it would have received had no such transfer or change occurred. The affected party shall submit appropriate proof of payment of the withholding taxes to the other party within a reasonable period of time not exceeding thirty (30) days after filing and paying such taxes. Each party will reasonably provide the necessary documentation to enable the other party to apply for declaration of reduced tax rates or exemption from withholding taxes on royalty payments under the relevant tax treaties, if applicable. Either or both parties, as the case may be, will apply for such reduced tax rates or exemptions from withholding taxes, if applicable. Each party will also reasonably provide any and all relevant documents to the other party in order that such other party can reduce or eliminate any withholding tax or claim the withheld taxes according to the relevant tax treaties and according to local tax laws. |
| 6.8. | The Royalty and any other sums payable under this Agreement shall be paid in euro to the credit of a bank account to be designated in writing by the Company. |
| 6.9. | The Royalty payable under this Agreement shall be paid within 30 days of the end of each successive quarterly period and for the purpose of converting the local currency of Newco (in which such Royalty arises) into euro, the rate of exchange to be applied shall be the rate of |
| 8 |
exchange applied by the Dublin bankers to the Company for the purchase of euro with such foreign currency as at the close of business on the date when the relevant payment first becomes due.
| 6.10. | In the event of any delay in paying any sum due under this Agreement by the due date, Newco shall pay to the Company interest (calculated on a daily basis) on the overdue payment from the date when such payment was due to the date of actual payment at a rate of 1% over the prime rate of the Governor and Company of the Bank of Ireland from time to time. |
| 6.11. | The parties hereto acknowledge that NewCo shall (subject to the terms of clause 2, the Licence Option and any Licence) be free to acquire or to license or sub-license any intellectual property which it considers to be necessary in order to obtain Regulatory Approval in respect of any Application, or to Commercialise in any of the Fields and the parties shall use all reasonable endeavours to minimise the amount of any royalties under any such licences. |
| 6.12. | The parties shall procure that at no time shall the aggregate sum of all royalties which are required to be paid by NewCo to third parties (including the Company) in relation to Commercialisation, where the obligation to pay royalties exists at the Effective Date of this Agreement: |
| 6.12.1. | exceed 18% of Net Income, for so long as Waratah is receiving a royalty pursuant to the Waratah Agreement; or |
| 6.12.2. | exceed 15% of Net Income for so long as the Company (or any Subsidiary of the Company) is the only party entitled to receive a royalty from Newco; and |
| 6.12.3. | where such royalties which are required to be so paid by NewCo exceed the limits set out in 6.12.1 or 6.12.2 (whichever is applicable), the Royalty payable to the Company pursuant to this Agreement shall be reduced to the extent necessary to ensure that this limit is not exceeded. |
| 6.13. | For the avoidance of doubt, for the duration of this Agreement, the Company's Royalty shall never be reduced to less than 3% of Net Income. |
| 7. | AUDIT |
| 7.1. | At the same time as payment of the Royalty falls due, Newco shall submit or cause to be submitted to the Company a statement in writing recording the calculation of the Royalty payable and in particular: |
| 7.1.1. | the annual period for which the Royalties were calculated; |
| 7.1.2. | the number of Patents and/or Products and/or details of Know-How used, sold, licensed, rented and/or supplied during the annual period; |
| 7.1.3. | the number of Products manufactured during the annual period but not yet used, sold, licensed, rented and/or supplied; |
| 7.1.4. | the gross sales price of each Product used, sold, licensed, rented and/or supplied during the annual period; |
| 7.1.5. | the amount of Royalties due and payable; and |
| 7.1.6. | (subject to clause 6.9) the amount of any withholding or other income taxes deductible or due to be deducted from the amount of Royalties due and payable. |
| 9 |
| 7.2. | Newco shall keep proper records and books of account showing the description and price of any Products, Patents and/or Know-How which have been used, sold, licensed, rented and/or supplied. Such records and books shall be kept separate from any records and books not relating solely to the use, sale, license, rental and/or supply of the Products, Patents and/or Know-How and be open on reasonable advance written notice during normal business hours to inspection and audit by the Company (or its authorised representative) up to twice per calendar year, who shall be entitled to take copies of or extracts from them. If such inspection or audit should reveal a discrepancy in the Royalties paid from those payable under this Agreement, Newco shall immediately make up the shortfall and, where the discrepancy exceeds 10% of the amounts paid, reimburse the Company in respect of any professional charges incurred for such audit or inspection. Such right of inspection of the Company shall remain in effect for a period of one year after any agreed upon termination of this agreement. |
| 7.3. | If Newco is prohibited by a governmental authority in any country from making any payment due under this Agreement then Newco shall, within the prescribed period for making such payment, use all reasonable endeavours to secure from such authority permission to make such payment and shall make it within 7 days of receiving such permission. If such permission is not received within thirty (30) days of Newco making a request for such permission then, at the option of the Company, Newco shall either deposit the payment in the currency of the relevant country in a bank account designated by the Company within such country, or make the payment to an Affiliate of the Company designated by the Company and having an office in the relevant country or in another country designated by the Company. |
| 7.4. | Within thirty (30) days of the end of each calendar year, Newco shall submit to the Company a written statement certified by Newco's auditors of the aggregate gross sales, licence and/or rental price of any Products, Patents and/or Know-How released due to the Commercialisation by Newco of any Products, Patents and/or Know-How in that year and the amount due to be paid for that year under clause 6 and this clause 7. If such statement shows that the Royalty amount paid by Newco during the calendar year was less than the amount due, Newco shall pay to the Company within seven days of the submission of the statement an amount equivalent to the difference between the Royalty paid and the amount due. |
| 7.5. | The provisions of this clause 7 shall remain in effect notwithstanding termination or expiry of this agreement until the settlement of all subsisting claims by the Company. |
| 7.6. | Where Newco has granted any sub-licence of any Products, Patents and/or Know-How, Newco shall include, in its Royalty, payments in respect of all activities which are carried out by or on behalf of a sub-licensee, and shall include records of such activities in the statements it submits pursuant to this clause 7. |
| 8. | TERM AND TERMINATION |
| 8.1. | The rights and obligations contained in clause 5, clause 6 and clause 7 of this Agreement (the Royalty Agreement) and any other terms of this Agreement which are relevant to give effect to the Royalty Agreement shall be perpetual and shall not be terminated by either party for any reason unless the parties otherwise agree in writing to terminate the Royalty Agreement at any time. |
| 8.2. | Clauses 2 and 3 of this Agreement (the Licence Option Agreement) shall come into force on the Effective Date and shall remain in effect until the expiry of the Option Period (the Option Term), unless the Company has earlier terminated the Licence Option Agreement in accordance with Clause 8.3 below. |
| 8.3. | The Company shall be entitled to terminate the Licence Option Agreement at any time by serving 30 days written notice on Newco which termination will, for the avoidance of doubt have no effect on the other terms of this Agreement. |
| 10 |
| 8.4. | Termination of the Licence Option Agreement shall not release either of the parties from any other liability which at the time of termination has already accrued to the other party, nor affect in any way the survival of any other right, duty or obligation of the parties which is expressly stated (or which by implication must) survive such termination. |
| 9. | CONFIDENTIAL INFORMATION/ANNOUNCEMENTS |
| 9.1. | Upon execution of this Agreement, and thereafter during the term hereof, at such times as the parties shall mutually agree, each party may disclose to the others, in confidence Confidential Information necessary for fulfilment of obligations, or useful to the activities, contemplated by this Agreement. |
| 9.2. | Except as specifically authorised or permitted by this Agreement, each party shall, keep confidential and not disclose to others (except its Affiliates), and use only as permitted hereunder, all of the Confidential Information owned by the other parties after this Agreement's expiration or termination. |
| 9.3. | Save as otherwise specifically provided herein, each party shall disclose Confidential Information of the other parties only to those employees, representatives and agents requiring knowledge thereof in connection with fulfilling the party's obligations or exercise of that party's rights under this Agreement. Each party further agrees to (i) inform all such employees, representatives and agents of the terms and provisions of this Agreement relating to Confidential Information and their duties hereunder, and (ii) obtain their agreement hereto as a condition of receiving Confidential Information, provided that such agreement shall be deemed given in respect of such employees, representatives and agents that, at the time of disclosure, are under existing obligations of confidentiality no less onerous than those contained herein covering such disclosure. Each party shall exercise the same standard of care as it would itself exercise in relation to its own confidential information (but in no event less than a reasonable standard of care) to protect and preserve the proprietary and confidential nature of the Confidential Information disclosed to it by the other parties. |
| 9.4. | Notwithstanding the provisions of this Clause 9, Confidential Information may be |
| 9.4.1. | published if and to the extent such publication has been approved in writing by each of the parties; or |
| 9.4.2. | disclosed to the extent required by applicable laws or binding regulations or as ordered by a court or other regulatory body having binding and competent jurisdiction, provided that if a party becomes legally required to disclose any Confidential Information of the other party hereunder, the receiving party shall give the disclosing party prompt notice of such requirement to enable the disclosing party to seek a protective order or other appropriate remedy concerning any such disclosure. The receiving party shall (at the cost and expense of the disclosing party) fully co-operate with the disclosing party in connection with the disclosing party's efforts to obtain any such order or other remedy. If any such order or other remedy does not fully preclude disclosure, the receiving party shall make such disclosure only to the extent that such disclosure is legally required. |
| 9.5. | The parties agree that the obligations of this Clause 9 are necessary and reasonable in order to protect the parties' respective businesses, and each party agrees that monetary damages would be inadequate to compensate a party for any breach by the other party of its covenants and agreements set forth herein. |
| 9.6. | The parties agree that any such violation or threatened violation may cause irreparable injury to a party and that, in addition to any other remedies that may be available, in law and equity or otherwise, each party shall be entitled to seek injunctive relief against the threatened breach of the |
| 11 |
provisions of this Clause 9, or a continuation of any such breach by the other party, specific performance and other equitable relief to redress such breach together with damages and reasonable counsel fees and expenses to enforce its rights hereunder.
| 9.7. | Subject to Clause 2.5, Clause 9.3 and Clause 9.4.2, neither party shall have the right to disclose to third parties the existence of this Agreement or any of the terms and conditions hereof without the prior written consent of the other party. In the event that either party wishes to make an announcement concerning the Agreement, that party will seek the consent of the other party, which consent shall not be unreasonably withheld or delayed. The terms of any such announcement shall be agreed in good faith. |
| 10. | MISCELLANEOUS PROVISIONS |
| 10.1. | Severability |
All the terms and provisions of this Agreement are distinct and severable, and if any term or provision is held or declared to be unenforceable, illegal or void in whole or in part by any court, regulatory authority or other competent authority it will to that extent only be deemed not to form part of this Agreement, and the enforceability, legality and validity of the remainder of this Agreement will not in any event be affected. The parties shall then use all reasonable endeavours to agree a term or provision to replace the unenforceable, illegal or void term or provision which is legal and enforceable and which has an effect that is near as possible to the intended effect of the term or provision to be replaced.
| 10.2. | Whole Agreement |
This Agreement (together with any documents to be executed pursuant to the terms of this Agreement) supersedes all prior representations, arrangements, understandings and agreements, and sets out the entire, complete and exclusive agreement and understanding between the parties.
| 10.3. | Remedies Cumulative |
The provisions of this Agreement and the rights and remedies of the parties are cumulative and are without prejudice and in addition to any rights or remedies which a party may have at law or in equity. The exercise by a party of any one right or remedy under this Agreement or at law or in equity will not (unless expressly otherwise provided in this Agreement or at law or in equity) operate so as to hinder or prevent the exercise by that party of any other right or remedy.
| 10.4. | Waiver |
A waiver by any party or parties of any breach of any of the terms, provisions, conditions or covenants of this Agreement or the acquiescence of any party or parties in any act (whether commission or omission) which but for such acquiescence would be a breach, will not constitute a general waiver of the term, provision, condition or covenant or of any subsequent act which is inconsistent with it.
| 10.5. | Notices |
Any notice or other communication to be given or served under this Agreement shall be in writing, addressed to the relevant party and expressed to be a notice or communication under this Agreement and, without prejudice to the validity of another method of service, may be delivered or sent by registered post or facsimile addressed to the recipient at the address given above or to its fax number at that address.
| 12 |
| 10.6. | Counterparts |
This Agreement may be executed in any number of counterparts, and by the several parties to it on separate counterparts, each of which when so executed will constitute an original but all of which together will evidence the same agreement.
| 10.7. | Governing Law and Jurisdiction |
This Agreement and all relationships created by it will in all respects be governed by and construed in accordance with Irish law. The Irish courts will have exclusive jurisdiction to settle any dispute which may arise out of or in connection with this Agreement or its performance.
| 10.8. | Succession and Assignment |
| 10.8.1. | None of the parties to this Agreement may assign any of its rights (including in respect of the Patents) under this Agreement without the prior written consent of each of the other parties except that, for the avoidance of doubt, the Company may assign, novate, or transfer any of its rights hereunder to any Company Affiliate. |
| 10.8.2. | This Agreement will be binding on and enure for the benefit of the permitted assigns and permitted transferees or substitutes of the Company and/or Newco and references to the Company and/or Newco will be construed accordingly. |
| 10.9. | Variations |
This Agreement may not be released, varied, discharged, amended or supplemented, except by an instrument in writing executed by each party or a duly authorised representative of each party.
| 13 |
SCHEDULE 1
Patents
Part A – Owned Patents
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00667-US-NP | US | FLG | 13 Oct 2011 | 13/273,069 | ||||||
| 00667-US-PSP | US | EXP | 13 Oct 2010 | 61/455,089 | ||||||
| 00667-WO-PCT | WO | PUB | 13 Oct 2011 | PCT/US2011/056109 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00681-US-PSP | US | EXP | 03 Jun 2011 | 61/520,031 | ||||||
| 00681-US-PSP[3] | US | FLG | 31 Mar 2012 | 61/618,680 | ||||||
| 00681A1-US | US | FLG | 04 Jun 2012 | 13/488,363 | ||||||
| 00681A1W1-PCT | WO | FLG | 04 Jun 2012 | PCT/US2012/40789 | ||||||
| 00681A2 | US | FLG | 30 Sep 2011 | 61/541,333 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00692A1 | US | FLG | 15 Feb 2010 | 61/304,581 | ||||||
| 00692A1W1-AU | AU | DES | ||||||||
| 00692A1W1-CA | CA | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-CN | CN | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-EA | EA | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-EP | EP | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-JP | JP | FLG | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| 00692A1W1-NZ | NZ | DES | ||||||||
| 00692A1W1-PCT | WO | PUB | 14 Feb 2011 | PCT/US2011/024731 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00695-A1-US | US | FLG | 28 Sep 2012 | 13/573,640 | ||||||
| 14 |
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00700-prov1 | US | FLG | 09 Jan 2013 | 61/750,722 |
Part B – Licenced Patent Rights
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00592-CA-PCT | CA | FLG | 13 Oct 2006 | 2626005 | ||||||
| 00592-EP-EPT | EP | FLG | 13 Oct 2006 | 06850456.2 | ||||||
| 00592-JP-PCT | JP | PUB | 13 Oct 2006 | 2008-535136 | ||||||
| 00592-US-CNT[4] | US | ABD | 11 Nov 2009 | 12/616,496 | ||||||
| 00592-US-NP[2] | US | ABD | 13 Oct 2006 | 11/580,026 | ||||||
| 00592-US-PCT[3] | US | FLG | 13 Oct 2006 | 12/090,130 | ||||||
| 00592-US-PSP | US | EXP | 13 Oct 2005 | 60/725,634 | ||||||
| 00592-WO-PCT | WO | FLG | 13 Oct 2006 | PCT/US2006/039872 | ||||||
| 00592-WO-PCT[2] | WO | OPEN | 13 Oct 2006 | PCT/IB2006/004181 | ||||||
| 00592-WO-PCT[3] | WO | PUB | 13 Oct 2006 | PCT/CA2006/001679 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00593-AU-PCT | AU | GRT | 17 Nov 2005 | 2005 306 531 | 2005 306 531 | |||||
| 00593-BR-PCT | BR | FLG | 17 Nov 2005 | PI0517733-2 | ||||||
| 00593-CA-PCT | CA | FLG | 17 Nov 2005 | 2588423 | ||||||
| 00593-CN-PCT | CN | FLG | 17 Nov 2005 | 200580046701.8 | ||||||
| 00593-EP-EPD[2] | EP | ABD | 17 Nov 2005 | 10000985.1 | ||||||
| 00593-EP-EPT | EP | ABD | 17 Nov 2005 | 05808109.2 | ||||||
| 00593-IN-PCD | IN | FLG | 17 Nov 2005 | 90/KOLNP/2012 | ||||||
| 00593-IN-PCT | IN | FLG | 17 Nov 2005 | 1887/KOLNP/2007 | ||||||
| 00593-JP-PCT | JP | PUB | 17 Nov 2005 | 2007-541601 | ||||||
| 00593-MX-PCT | MX | FLG | 17 Nov 2005 | MX/a/2007/005870 | ||||||
| 00593-PH-PCT | PH | FLG | 17 Nov 2005 | 1-2007-501053 | ||||||
| 00593-US-CNT[4] | US | ABD | 20 Aug 2007 | 11/841,773 | ||||||
| 00593-US-CNT[5] | US | ABD | 20 Aug 2007 | 11/841,745 | ||||||
| 00593-US-CNT[6] | US | ABD | 20 Aug 2007 | 11/841,763 | ||||||
| 00593-US-CNT[7] | US | ABD | 20 Aug 2007 | 11/841,730 | ||||||
| 00593-US-CNT[8] | US | ABD | 22 May 2008 | 12/125,498 | ||||||
| 00593-US-CNT[9] | US | PUB | 15 Sep 2010 | 12/882,250 | ||||||
| 00593-US-NP[3] | US | ABD | 17 Nov 2005 | 11/280,818 | ||||||
| 00593-US-PCT[2] | US | ABD | 17 Nov 2005 | 11/667,822 | ||||||
| 00593-US-PSP | US | EX-C | 17 Nov 2004 | 60/628,840 | ||||||
| 00593-WO-PCT | WO | PUB | 17 Nov 2005 | PCT/CA2005/001744 | ||||||
| 00593-ZA-DIV[2] | ZA | FLG | 17 Nov 2005 | 2008/08231 | ||||||
| 00593-ZA-PCD | ZA | FLG | 17 Nov 2005 | 2012/02636 | ||||||
| 00593-ZA-PCT | ZA | GRT | 17 Nov 2005 | 2007/04872 | 2007/04872 | |||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00594-AT-EPT | AT | GRT | 27 Feb 2004 | 04715226.9 | E 432 694 | |||||
| 00594-AU-DIV | AU | FLG | 27 Feb 2004 | 2009251035 | ||||||
| 00594-AU-PCT[2] | AU | GRT | 27 Feb 2004 | 2004216544 | 2004216544 | |||||
| 00594-BE-EPT | BE | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-BG-EPT | BG | GRT | 27 Feb 2004 | 04715226.9 | 1608350 |
| 15 |
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00594-BR-PCT | BR | FLG | 27 Feb 2004 | PI 0407910-8 | ||||||
| 00594-CA-PCT | CA | FLG | 27 Feb 2004 | 2516563 | ||||||
| 00594-CH-EPT | CH | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-CN-DIV | CN | FLG | 27 Feb 2004 | 200810146321.7 | ||||||
| 00594-CN-DIV[3] | CN | ABD | 27 Feb 2004 | N/A | ||||||
| 00594-CN-PCT[2] | CN | GRT | 27 Feb 2004 | 20048011335.8 | ZL 200480011335.8 | |||||
| 00594-CY-EPT | CY | GRT | 27 Feb 2004 | 04715226.9 | CY1109338 | |||||
| 00594-CZ-EPT | CZ | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-DE-EPT | DE | GRT | 27 Feb 2004 | 04715226.9 | 602004021362.4-08 | |||||
| 00594-DK-EPT | DK | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-EE-EPT | EE | GRT | 27 Feb 2004 | 04715226.9 | E003407 | |||||
| 00594-EP-EPD | EP | PUB | 27 Feb 2004 | 09005295.2 | ||||||
| 00594-EP-EPD[2] | EP | PUB | 27 Feb 2004 | 10010477.7 | ||||||
| 00594-EP-EPT[3] | EP | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-ES-EPT | ES | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-FI-EPT | FI | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-FR-EPT | FR | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-GB-EPT | GB | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-GR-EPT | GR | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-HU-EPT | HU | GRT | 27 Feb 2004 | 04715226.9 | E006366 | |||||
| 00594-IE-EPT | IE | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-IL-PCD[1] | IL | FLG | 27 Feb 2004 | 220065 | ||||||
| 00594-IL-PCT | IL | FLG | 27 Feb 2004 | 170476 | ||||||
| 00594-IT-EPT | IT | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-JP-PCD | JP | PUB | 27 Feb 2004 | 2010-294145 | ||||||
| 00594-JP-PCT[2] | JP | GRT | 27 Feb 2004 | 2006-501433 | 4999453 | |||||
| 00594-LU-EPT | LU | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-NL-EPT | NL | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-PT-EPT | PT | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-RO-EPT | RO | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-SE-EPT | SE | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-SI-EPT | SI | GRT | 27 Feb 2004 | 04715226.9 | 1608350 | |||||
| 00594-SK-EPT | SK | GRT | 27 Feb 2004 | 04715226.9 | E 6042 | |||||
| 00594-TR-EPT | TR | GRT | 27 Feb 2004 | 04715226.9 | TR 2009 06231 T4 | |||||
| 00594-US-CIP[5] | US | PUB | 27 Feb 2004 | 10/547,286 | ||||||
| 00594-US-CNT[6] | US | ABD | 03 Mar 2009 | 12/396,515 | ||||||
| 00594-US-CNT[7] | US | PUB | 29 Feb 2012 | 13/408,337 | ||||||
| 00594-US-NP[4] | US | GRT | 26 Feb 2004 | 10/787,621 | 7,521,481 | |||||
| 00594-US-PSP | US | EX-C | 27 Feb 2003 | 60/451,363 | ||||||
| 00594-US-PSP[2] | US | EX-C | 17 Nov 2003 | 60/520,958 | ||||||
| 00594-US-PSP[3] | US | EX-C | 19 Nov 2003 | 60/523,534 | ||||||
| 00594-WO-PCT | WO | PUB | 27 Feb 2004 | PCT/CA2004/00272 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00601-CA-PCT | CA | ABD | 16 Feb 2007 | 2642647 | ||||||
| 00601-EP-EPT | EP | ABD | 16 Feb 2007 | 07789484.8 | ||||||
| 00601-JP-PCT | JP | ABD | 16 Feb 2007 | 2008-554886 | ||||||
| 00601-US-NP[2] | US | ABD | 16 Feb 2007 | 11/707,156 | ||||||
| 00601-US-PSP | US | EX-C | 17 Feb 2006 | 60/774,818 |
| 16 |
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00601-WO-PCT[2] | WO | ABD | 16 Feb 2007 | PCT/IB2007/001929 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00615-CA-PCT | CA | ABD | 18 May 2007 | 2652449 | ||||||
| 00615-EP-EPT | EP | ABD | 18 May 2007 | 07719822.4 | ||||||
| 00615-US-PCT[4] | US | PUB | 18 May 2007 | 12/301,155 | ||||||
| 00615-US-PSP | US | EX-C | 19 May 2006 | 60/801,848 | ||||||
| 00615-US-PSP[2] | US | EX-C | 12 Oct 2006 | 60/851,256 | ||||||
| 00615-US-PSP[3] | US | EX-C | 13 Nov 2006 | 60/858,548 | ||||||
| 00615-WO-PCT | WO | ABD | 18 May 2007 | PCT/CA2007/000900 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00627-US-PCT[2] | US | ABD | 20 Sep 2007 | 12/438,572 | ||||||
| 00627-US-PSP | US | EX-C | 21 Sep 2006 | 60/846,366 | ||||||
| 00627-WO-PCT | WO | ABD | 20 Sep 2007 | PCT/CA2007/001678 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00629-AU-PCT | AU | FLG | 09 Mar 2007 | 2007222864 | ||||||
| 00629-BR-PCT | BR | PUB | 09 Mar 2007 | PI0708725-0 | ||||||
| 00629-CA-PCT | CA | FLG | 09 Mar 2007 | 2644804 | ||||||
| 00629-CN-PCD | CN | FLG | 09 Mar 2007 | 201210257027.X | ||||||
| 00629-CN-PCT | CN | PUB | 09 Mar 2007 | 200780016972.8 | ||||||
| 00629-CO-PCT | CO | PUB | 09 Mar 2007 | 08.108.264 | ||||||
| 00629-EA-EAT | EA | FLG | 09 Mar 2007 | 200801967 | ||||||
| 00629-EP-EPT | EP | PUB | 09 Mar 2007 | 07710726.6 | ||||||
| 00629-ID-PCT | ID | FLG | 09 Mar 2007 | W00200803173 | ||||||
| 00629-IL-PCT | IL | FLG | 09 Mar 2007 | 193970 | ||||||
| 00629-IN-PCT | IN | PUB | 09 Mar 2007 | 3723/KOLNP/2008 | ||||||
| 00629-JP-PCT | JP | FLG | 09 Mar 2007 | 2008-557568 | ||||||
| 00629-KR-PCT | KR | FLG | 09 Mar 2007 | 1-2008-7024122 | ||||||
| 00629-MX-PCT | MX | FLG | 09 Mar 2007 | MX/a/2008/011553 | ||||||
| 00629-NZ-PCT | NZ | GRT | 09 Mar 2007 | 571,181 | 571,181 | |||||
| 00629-PH-PCT | PH | FLG | 09 Mar 2007 | 1-2008-502019 | ||||||
| 00629-US-PCT[2] | US | PUB | 09 Mar 2007 | 12/282,030 | ||||||
| 00629-US-PSP | US | EX-C | 09 Mar 2006 | 60/780,526 | ||||||
| 00629-WO-PCT | WO | PUB | 09 Mar 2007 | PCT/CA2007/000395 | ||||||
| 00629-ZA-DIV | ZA | ALL | 09 Mar 2007 | 2009/08303 | ||||||
| 00629-ZA-PCT[2] | ZA | GRT | 09 Mar 2007 | 2008/07993 | 2008/07993 | |||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00632-CA-PCT | CA | FLG | 22 Nov 2007 | 2670405 | ||||||
| 00632-EP-EPT | EP | PUB | 22 Nov 2007 | 07845583.9 | ||||||
| 00632-JP-PCT | JP | PUB | 22 Nov 2007 | 2009-537460 | ||||||
| 00632-US-PCT | US | PUB | 22 Nov 2007 | 12/445,164 | ||||||
| 00632-WO-PCT | WO | PUB | 22 Nov 2007 | PCT/CA2007/002118 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00633-CA-PCT | CA | FLG | 11 Apr 2008 | 2683580 | ||||||
| 00633-US-PCT[3] | US | PUB | 11 Apr 2008 | 12/594,947 |
| 17 |
| 00633-US-PSP | US | EX-C | 12 Apr 2007 | 60/923,061 | ||||||
| 00633-US-PSP[2] | US | EX-C | 12 Apr 2007 | 60/923,001 | ||||||
| 00633-WO-PCT | WO | PUB | 11 Apr 2008 | PCT/CA2008/000685 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00634-CA-PCT | CA | FLG | 11 Apr 2008 | 2683546 | ||||||
| 00634-US-PCT[3] | US | PUB | 11 Apr 2008 | 12/594,950 | ||||||
| 00634-US-PSP | US | EX-C | 12 Apr 2007 | 60/923,150 | ||||||
| 00634-US-PSP[2] | US | EX-C | 12 Apr 2007 | 60/923,044 | ||||||
| 00634-WO-PCT | WO | PUB | 11 Apr 2008 | PCT/CA2008/000683 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00635-AU-PCT | AU | FLG | 11 Apr 2008 | 2008 238 577 | ||||||
| 00635-CA-PCT | CA | FLG | 11 Apr 2008 | 2683607 | ||||||
| 00635-EP-EPT | EP | PUB | 11 Apr 2008 | 08733752.3 | ||||||
| 00635-JP-PCT | JP | ABD | 11 Apr 2008 | 2010-502395 | ||||||
| 00635-US-PCT[2] | US | PUB | 11 Apr 2008 | 12/594,951 | ||||||
| 00635-US-PSP | US | EX-C | 12 Apr 2007 | 60/922,998 | ||||||
| 00635-WO-PCT | WO | FLG | 11 Apr 2008 | PCT/CA2008/000703 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00636-CA-PCT | CA | FLG | 11 Apr 2008 | 2683548 | ||||||
| 00636-US-PCT[3] | US | PUB | 11 Apr 2008 | 12/594,943 | ||||||
| 00636-US-PSP | US | EX-C | 12 Apr 2007 | 60/923,002 | ||||||
| 00636-US-PSP[2] | US | EX-C | 12 Apr 2007 | 60/923,120 | ||||||
| 00636-WO-PCT | WO | FLG | 11 Apr 2008 | PCT/CA2008/000684 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00643-AU-PCT | AU | FLG | 09 Oct 2009 | 2009 301 603 | ||||||
| 00643-CA-PCT | CA | FLG | 09 Oct 2009 | 2740124 | ||||||
| 00643-EP-EPT[1] | EP | PUB | 09 Oct 2009 | 09818730.5 | ||||||
| 00643-JP-PCT | JP | PUB | 09 Oct 2009 | 2011-530339 | ||||||
| 00643-NZ-PCT | NZ | FLG | 09 Oct 2009 | 592277 | ||||||
| 00643-US-NP[2] | US | PUB | 09 Oct 2009 | 12/576,957 | ||||||
| 00643-US-PSP | US | EX-C | 09 Oct 2008 | 61/104,094 | ||||||
| 00643-WO-PCT | WO | PUB | 09 Oct 2009 | PCT/CA2009/001448 | ||||||
| 00643-ZA-PCT | ZA | FLG | 09 Oct 2009 | 2011/02640 | ||||||
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. | |||||
| 00650-AR-NP | AR | FLG | 15 Sep 2008 | P090103535 | ||||||
| 00650-AU-PCT | AU | FLG | 15 Sep 2009 | 2009290612 | ||||||
| 00650-CA-PCT[2] | CA | FLG | 15 Sep 2009 | 2737163 | ||||||
| 00650-EP-EPT[2] | EP | PUB | 15 Sep 2009 | 09792556.4 | ||||||
| 00650-JP-PCT[2] | JP | PUB | 15 Sep 2009 | 2011-527043 | ||||||
| 00650-TW-NP | TW | PUB | 15 Sep 2009 | 98131051 | ||||||
| 00650-US-NP[2] | US | PUB | 15 Sep 2009 | 12/560,113 | ||||||
| 00650-US-PSP | US | EX-C | 15 Sep 2008 | 61/097,023 | ||||||
| 00650-VE-NP | VE | FLG | 15 Sep 2008 | N/A | ||||||
| 00650-WO-PCT | WO | FLG | 15 Sep 2009 | PCT/US2009/057003 | ||||||
| 00650-WO-PCT[2] | WO | PUB | 15 Sep 2009 | PCT/US2009/056985 | ||||||
| 18 |
Subject to Contract/ Contract Denied
SCHEDULE 2
Template Patent and Know-How Licence Agreement
THIS AGREEMENT is dated [DATE] and made between
| (1) | SPERANZA BIOPHARMA LIMITED a company incorporated in Ireland (Registered No. 502444) whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2 (Licensor); and |
| (2) | ELAN PHARMA INTERNATIONAL LIMITED (registered no. 222276) having its registered office at Treasury Building, Grand Canal Street Lower, Dublin 2 (the Licensee). |
Recitals
| A. | The Licensee has, pursuant to the Option and Royalty Agreement entered into with the Licensor dated (the "OR Agreement") been granted an Option to obtain a licence in relation to the Patents (as defined below), Know-How (as defined below) and Product (as defined below); |
| B. | The Licensee has now decided to exercise his Licence Option in respect of the Patents, Know-How and Products with respect to the Field; and |
| C. | The Licensor has now agreed to grant, and the Licensee has agreed to take, a licence on the terms and subject to the conditions set out in this agreement. |
Agreed terms
| 1. | Interpretation |
| 1.1. | The definitions and rules of interpretation in this clause apply in this agreement. |
Affiliate means another person that directly or indirectly controls, is controlled by, or is under common control with, such first person, where control means the possession, directly or indirectly, of the power to direct or cause the direction of the business, affairs or management of a person, whether through the ownership of voting securities, by contract, as trustee or executor, or otherwise;
Application means, without limitation, Down Syndrome, Bipolar Disorder and/or Alzheimer's agitation and/or any other potential future use or application which is now known (or may in the future be known) in respect of the Patents and/or Know-How and/or any Product;
Business Transfer Agreement means the Business Transfer Agreement dated 2013 between the Licensor and the Licensee whereby the Licensee transferred all assets relating to the Product to the Licensor.
Commercialisation means any and all activities constituting importing, manufacturing, marketing, distributing, offering for sale and/or selling a Product and shall include but is not be limited to:
| (a) | sales; |
| (b) | loaning, leasing, letting on hire or sales on hire purchase; |
| (c) | supply; |
| (d) | putting into use; or |
| (e) | Promotion |
as well as activities required to fulfil on-going regulatory obligations, including adverse event reporting. When used as a verb, Commercialise shall mean to engage in Commercialisation;
Confidential Information means know-how, trade secrets, inventions (including, where applicable, information in relation to the Patents, Know-How and/or any other potential or actual patent applications covering inventions), data, information, and any improvements, modifications, derivations, or compilations thereto that is owned, licensed by or controlled by the disclosing party, provided however, that Confidential Information shall not include any information which is:
| (1) | already known to the receiving party at the time of disclosure, as evidenced by such party's written records, provided such information was not obtained directly or indirectly by the receiving party from the disclosing party pursuant to a confidentiality agreement; |
| (2) | publicly known prior to or after disclosure, through no default of the receiving party; |
| (3) | disclosed in good faith to the receiving party by a third party, lawfully and contractually entitled to make such disclosure; or |
| (4) | is independently discovered without the aid or application of the Confidential Information as shall be evidenced by the written records of the receiving party. |
Derivative means any compounds identified, obtained, developed, created, synthesized, generated, designed or resulting from, based upon, containing or incorporating the chemical structure of Scyllo-Inositol as those compounds are generically and/or specifically disclosed and claimed in the Patents;
Effective Date: the date of this agreement;
European Selected Option Territories means the countries listed in Part A of Schedule 2
Field: means any use or Application or potential future use or application which is now known (or may in the future be known) in respect of the Patents and/or Know-How and/or any Product;
Improvement: any improvement, enhancement or modification to the technology that is the subject of the Patents or the Licensed Know-How or any Product or its method of manufacture.
Licensed Know-How means any and all rights related to the Product and /or the Patents, owned, licensed or controlled by the Licensor to include any scientific, pharmaceutical or technical information, data, discovery, invention (whether patentable or not), know-how, substances, techniques, processes, systems, formulations, designs and expertise which is not generally known to the public;
Non-European Selected Option Territories means the countries listed in Part B of Schedule 2.
OR Agreement means the agreement entered into between Elan Pharma International Limited and Speranza Biopharma Limited;
Other Territories means all other countries, nations, principalities or jurisdictions anywhere in the world which are not Selected Option Territories, as defined under this Agreement;
Patents means any and all rights under any and all patent applications and/or patents anywhere in the world, now existing, currently pending or hereafter filed or obtained or licensed by Newco relating to the Product and any sub-divisions, divisions or extensions of same and includes, but is not limited to, the patents and patent applications set forth in Schedule 1;
Product means any pharmaceutical preparation or dosage form containing Scyllo-Inositol or a Derivative thereof;
Rights: the Patents, Licensed Know-How and Improvement(s) and all intellectual property rights of whatever nature (whether registered or unregistered) related or connected to same, to include the right to Commercialise any Product in the Field;
Scyllo-Inositol is the compound “AZD-103”, as more specifically disclosed in the Patents;
Selected Option Territories means the countries listed in Schedule 2; and
Transferred Assets means any of the Patents and/or Licensed Know-How the subject of the Business Transfer Agreement, transferred by the Licensee to the Licensor pursuant to, and in the form acquired by the Licensor at the date of, the Business Transfer Agreement and for the avoidance of doubt, before any Improvements were made and/or Products were developed by the Licensor.
| 1.2. | Clause and Schedule headings shall not affect the interpretation of this agreement. |
| 1.3. | The Schedules form part of this agreement and shall have effect as if set out in full in the body of this agreement. Any reference to this agreement includes the Schedules. |
| 1.4. | References to clauses and Schedules are to the clauses and Schedules of this agreement. |
| 1.5. | Unless the context otherwise requires, words in the singular include the plural and in the plural include the singular. |
| 1.6. | Unless the context otherwise requires, a reference to one gender shall include a reference to the other genders. |
| 1.7. | A reference to a statute or statutory provision is a reference to it as amended, extended or re-enacted from time to time; provided that, as between the parties, no such amendment, extension or re-enactment shall apply for the purposes of this agreement to the extent that it would impose any new or extended obligation, liability or restriction on, or otherwise adversely affect the rights of, any party. This clause does not, however, apply in relation to taxation. |
| 1.8. | A reference to a statute or statutory provision shall include any subordinate legislation made from time to time under that statute or statutory provision. |
| 1.9. | Writing or written includes faxes but not e-mail. |
| 1.10. | Any words following the terms including, include, in particular or any similar expression shall be construed as illustrative and shall not limit the sense of the words preceding those terms. |
| 1.11. | A person includes a natural person, corporate or unincorporated body (whether or not having separate legal personality) and that person's legal representatives, successors and permitted assigns. |
| 2. | Grant |
| 2.1. | The Licensor hereby grants to the Licensee an exclusive, sub-licensable, royalty-free, fully paid up, unrestricted and perpetual licence of the Rights for the Commercialisation of the Rights in the Selected Option Territories in the Field. |
| 2.2. | The Licensor undertakes not to exploit or Commercialise the Rights in the Selected Option Territories in the Field under any circumstances and for any purpose, or to grant others the right to do so for the duration of this Agreement. |
| 2.3. | Notwithstanding the foregoing the Licensee may elect within 12 months from the Effective Date of this Agreement, not to Commercialise the Patents and/or Know-How and/or Product in the Field in some or in all of the Selected Option Territories. The Licensee will provide the Licensor with written notice of such a decision at any time before a period of six months has elapsed from the date of this Agreement and any such countries, in which the Licensee will not be proceeding to Commercialise the Patents and/or Know-How and/or Products in the Field, will on that basis be deemed to be part of the Other Territories as regards Commercialisation in the Field from the date of that notice for the purposes of the "Royalty" as defined in the OR Agreement. |
| 3. | Provision of know-how |
| 3.1. | The Licensor shall make available to the Licensee such know-how relating to the manufacture of Products as the Licensor is at liberty to disclose and, in the opinion of the Licensor, is reasonably necessary for such manufacture. |
| 3.2. | The know-how supplied by the Licensor under clause 3.1 shall, where it has been identified by describing and recording it when provided to the Licensee, be deemed to be part of the Licensed Know-how. |
| 4. | Improvements |
| 4.1. | If the Licensee makes, devises, discovers, or otherwise acquires rights in, any Improvement: |
| 4.1.1. | all rights, title and interest in such Improvements anywhere in the world (subject to clause 4.3 below) shall vest in the Licensor; and |
| 4.1.2. | to the extent that it is not prohibited by law or by any obligation to any other person, the Licensee shall promptly notify the Licensor in writing of any non-sensitive or non-confidential |
details of the Improvement which the Licensee deems appropriate.
| 4.2. | Information provided by the Licensee to the Licensor under clause 4.1 shall be subject to the provisions of clause 7. |
| 4.3. | Any Improvements vesting in the Licensor under clause 4.1 or otherwise shall form part of the Rights licensed to the Licensee in accordance with clause 2.1. |
| 5. | Recordal of licence |
| 5.1. | The Licensee shall, at its own cost (and discretion) record the licence granted to it in clause 2.1 in the relevant or applicable patent registries or any other relevant or applicable registries anywhere in the world. |
| 5.2. | The Licensor shall provide reasonable assistance, at the Licensee's cost, to enable the Licensee to register its rights in accordance with clause 5.1. |
| 6. | Protection and Defence of the Rights |
| 6.1. | The Licensor and the Licensee shall immediately notify each other in writing, giving full particulars, if any of the following matters come to their attention: |
| 6.1.1. | any actual, suspected or threatened infringement of any of the Rights; |
| 6.1.2. | any actual, suspected or threatened unauthorised disclosure, misappropriation or misuse of the Rights; |
| 6.1.3. | any person applies for, or is granted, a patent by reason of which that person may be, or has been, granted, rights which conflict with any of the Rights granted to the Licensee under this agreement; |
| 6.1.4. | any actual or threatened claim or counterclaim that any of the Rights is invalid; |
| 6.1.5. | any actual or threatened opposition to any of the Rights; |
| 6.1.6. | any claim or counterclaim made or threatened that exploitation of any of the Rights infringe the rights of any third party (and/or any actual or threatened litigation or demand in relation to same); |
| 6.1.7. | any application is made for a compulsory licence under any Rights; or |
| 6.1.8. | any other form of attack, charge or claim to which the Rights may be subject; |
the matters referred to in clauses 6.14 – 6.1.8 (inclusive) to be hereafter referred to as a Claim, and these matters collectively to be referred to as Claims.
| 6.2. | Subject to Clause 6.3, the Licensor shall indemnify, defend and hold harmless the Licensee in relation to any Claim made against or which involves the Licensee anywhere in the world and the Licensor shall indemnify, defend and hold harmless the Licensee in relation to any costs (to include legal costs), damages and/or any other award made by a court, tribunal, arbitrator or otherwise resulting in respect a Claim, provided that the Company shall use all reasonable endeavours to mitigate any loss or damage that NewCo may incur in consequence of a matter giving rise to a right to an indemnity under this clause 6.2. |
| 6.3. | The indemnity provided for in clause 6.2: |
| 6.3.1. | shall not apply to Claims which relate to: |
| (1) | the Transferred Assets; or |
| (2) | any Improvements which are devised or discovered by the Licensee in the manner envisaged by clause 4.1 above. |
| 6.4. | The indemnity provided for in clause 6.2 shall be subject to a maximum aggregate cap in respect of all Claims arising in the Non-European Selected Option Territories of €10,000,000 but for the avoidance of doubt, the indemnity in clause 6.2 will remain uncapped as regards any Claims arising in European Selected Option Territories. |
| 6.5. | In circumstances where any of the matters referred to in clause 6.1.1 – clause 6.1.3 arise in the Selected Option Territories: |
| 6.5.1. | the Licensee may, without prejudice to the terms of clause 6.2, in its absolute discretion decide what action, if any, to take in the Selected Option Territories in respect of same, to include defence or commencement of proceedings if the Licensee deems it necessary and the Licensee shall have exclusive control over, and conduct of, all such proceedings in relation to the Rights in the Selected Option Territories; |
| 6.5.2. | the Licensor shall not make any admissions other than to the Licensee and shall provide (at the Licensee's cost) the Licensee with all reasonable assistance that it may require in the conduct of all such proceedings in relation to the Rights in the Selected Option Territories; and |
| 6.5.3. | the Licensee shall be entitled to retain all sums, damages and/or costs recovered in any such actions or proceedings in the Selected Option Territories for its own account. |
| 6.6. | The Licensee may require the Licensor to lend its name to any proceedings in respect of any of the circumstances described in clause 6.1.1 – 6.1.3, in the Selected Option Territories (at the Licensee's cost). |
| 6.7. | In circumstances where a Claim is made in the Other Territories: |
| 6.7.1. | the Licensor shall, in its absolute discretion decide what action, if any, to take in the Other Territories in respect of same, to include defence or commencement of proceedings if the Licensor deems it necessary and the Licensor shall have exclusive control over, and conduct of, all Claims and/or other proceedings in relation to the Rights in the Other Territories; |
| 6.7.2. | the Licensee shall not make any admissions other than to the Licensor and shall provide (at the Licensor's cost) the Licensor with all reasonable assistance that it may require in the conduct of any Claims or proceedings in relation to the Rights in the Other Territories; and |
| 6.7.3. | the Licensor shall be entitled to retain all sums, damages and/or costs recovered in any such actions in the Other Territories for its own account. |
| 6.8. | The parties agree to co-operate with each-other in relation to any of the matters listed in clause 6.1 and to keep each other informed of any actions taken or decisions which either party makes with respect to those matters from time to time. |
| 7. | Confidential Information |
| 7.1. | Upon execution of this Agreement, and thereafter during the term hereof, at such times as the parties shall mutually agree, each party may disclose to the others, in confidence Confidential Information necessary or useful to the activities contemplated by this Agreement. |
| 7.2. | Except as specifically authorised or permitted by this Agreement, each party shall, keep confidential and not disclose to others (except its Affiliates), and use only as permitted hereunder, all of the Confidential Information owned by the other parties after this Agreement's expiration or termination. |
| 7.3. | Save as otherwise specifically provided herein, each party shall disclose Confidential Information of the other parties only to those employees, representatives and agents requiring knowledge thereof in connection with fulfilling the party's obligations or exercise of that party's rights under this Agreement. Each party further agrees to (i) inform all such employees, representatives and agents of the terms and provisions of this Agreement relating to Confidential Information and their duties hereunder, and (ii) obtain their agreement hereto as a condition of receiving Confidential Information, provided that such agreement shall be deemed given in respect of such employees, representatives and agents that, at the time of disclosure, are under existing obligations of confidentiality no less onerous than those contained herein covering such disclosure. Each party shall exercise the same standard of care as it would itself exercise in relation to its own confidential information (but in no event less than a reasonable standard |
of care) to protect and preserve the proprietary and confidential nature of the Confidential Information disclosed to it by the other parties.
| 7.4. | Notwithstanding the provisions of this Clause 7, Confidential Information may be: |
| 7.4.1. | published if and to the extent such publication has been approved in writing by each of the parties; or |
| 7.4.2. | disclosed to the extent required by applicable laws or binding regulations or as ordered by a court or other regulatory body having binding and competent jurisdiction, provided that if a party becomes legally required to disclose any Confidential Information of the other party hereunder, the receiving party shall give the disclosing party prompt notice of such requirement to enable the disclosing party to seek a protective order or other appropriate remedy concerning any such disclosure. The receiving party shall (at the cost and expense of the disclosing party) fully co-operate with the disclosing party in connection with the disclosing party's efforts to obtain any such order or other remedy. If any such order or other remedy does not fully preclude disclosure, the receiving party shall make such disclosure only to the extent that such disclosure is legally required. |
| 7.5. | The parties agree that the obligations of this Clause 7 are necessary and reasonable in order to protect the parties' respective businesses, and each party agrees that monetary damages would be inadequate to compensate a party for any breach by the other party of its covenants and agreements set forth herein. |
| 7.6. | The parties agree that any such violation or threatened violation shall cause irreparable injury to a party and that, in addition to any other remedies that may be available, in law and equity or otherwise, each party shall be entitled to seek injunctive relief against the threatened breach of the provisions of this Clause 7, or a continuation of any such breach by the other party, specific performance and other equitable relief to redress such breach together with damages and reasonable counsel fees and expenses to enforce its rights hereunder. |
| 7.7. | Subject to Clause 7.3 and Clause 7.4.2, neither party shall have the right to disclose to third parties the existence of this Agreement or any of the terms and conditions hereof without the prior written consent of the other party. In the event that either party wishes to make an announcement concerning the Agreement, that party will seek the consent of the other party, which consent shall not be unreasonably withheld or delayed. The terms of any such announcement shall be agreed in good faith. |
| 8. | Termination and Consequences of Termination |
| 8.1. | The Licensee may, within twenty four months (24) immediately following the Effective Date, terminate this Agreement (or any part of it with respect to one or more of the Selected Option Territories) on notice to the Licensor. |
| 8.2. | The Licensor shall have the right to terminate this Agreement immediately by notice in writing to Company if:- |
| 8.2.1. | Licensee is in material breach of this Agreement and, in the event of a breach capable of being remedied, fails to remedy the breach within 30 days of receipt of notice in writing of such breach; |
| 8.2.2. | a receiver, examiner or administrator is appointed of the whole or any part of Licensee’s assets or Licensee is struck off the Register of Companies in the jurisdiction where it was incorporated or an order is made or a resolution passed for winding up the other party (unless such order or resolution is part of a voluntary scheme for the reconstruction or amalgamation of the party as a solvent corporation and the resulting corporation, if a different legal person, undertakes to be bound by this Agreement); or |
| 8.2.3. | Licensee challenges the validity of the Licensor’s Patents or Licensed Know How. |
| 8.3. | On termination of this Agreement by the Licensee, the Licensee will return or destroy (at the Licensee's option) all Confidential Information and any other materials, documentation or information relating to the |
Patents and/or Know-How.
| 9. | Further assurance |
| 9.1. | The parties shall (at the Licensee's cost and expense), and shall use all reasonable endeavours to procure that any necessary third party shall, promptly execute such documents and perform such acts as may reasonably be required for the purpose of giving full effect to this agreement. |
| 9.2. | The Licensor shall pay all fees, renewal charges and/or costs necessary for the upkeep, maintenance and/or renewal of the Patents. |
| 10. | Miscellaneous Provisions |
| 10.1. | Severability |
All the terms and provisions of this Agreement are distinct and severable, and if any term or provision is held or declared to be unenforceable, illegal or void in whole or in part by any court, regulatory authority or other competent authority it will to that extent only be deemed not to form part of this Agreement, and the enforceability, legality and validity of the remainder of this Agreement will not in any event be affected. The parties shall then use all reasonable endeavours to agree a term or provision to replace the unenforceable, illegal or void term or provision which is legal and enforceable and which has an effect that is near as possible to the intended effect of the term or provision to be replaced.
| 10.2. | Whole Agreement |
This Agreement (together with any documents to be executed pursuant to the terms of this Agreement) supersedes all prior representations, arrangements, understandings and agreements, and sets out the entire, complete and exclusive agreement and understanding between the parties.
| 10.3. | Remedies Cumulative |
The provisions of this Agreement and the rights and remedies of the parties are cumulative and are without prejudice and in addition to any rights or remedies which a party may have at law or in equity. The exercise by a party of any one right or remedy under this Agreement or at law or in equity will not (unless expressly otherwise provided in this Agreement or at law or in equity) operate so as to hinder or prevent the exercise by that party of any other right or remedy.
| 10.4. | Waiver |
A waiver by any party or parties of any breach of any of the terms, provisions, conditions or covenants of this Agreement or the acquiescence of any party or parties in any act (whether commission or omission) which but for such acquiescence would be a breach, will not constitute a general waiver of the term, provision, condition or covenant or of any subsequent act which is inconsistent with it.
| 10.5. | Notices |
Any notice or other communication to be given or served under this Agreement shall be in writing, addressed to the relevant party and expressed to be a notice or communication under this Agreement and, without prejudice to the validity of another method of service, may be delivered or sent by registered post or facsimile addressed to the recipient at the address given above or to its fax number at that address.
| 10.6. | Counterparts |
This Agreement may be executed in any number of counterparts, and by the several parties to it on separate counterparts, each of which when so executed will constitute an original but all of which together will evidence the same agreement.
| 10.7. | Governing Law and Jurisdiction |
This Agreement and all relationships created by it will in all respects be governed by and construed in accordance with Irish law. The Irish courts will have exclusive jurisdiction to settle any dispute which may arise out of or in connection with this Agreement or its performance.
| 10.8. | Succession and Assignment |
| 10.8.1. | None of the parties to this Agreement may assign any of its rights under this Agreement (including in respect of the Patents) without the prior written consent of each of the other parties except that, for the avoidance of doubt, the Licensee may assign, novate, or transfer any of its rights hereunder to any Licensee Affiliate. |
| 10.8.2. | This Agreement will be binding on and enure for the benefit of the permitted assigns and permitted transferees or substitutes of the Licensee and references to the Licensee will be construed accordingly. |
| 10.9. | Variations |
This Agreement may not be released, varied, discharged, amended or supplemented, except by an instrument in writing executed by each party or a duly authorised representative of each party.
This agreement has been entered into on the date stated at the beginning of it:
Schedule 1
The Patents
[To Insert As Per the Licence Option Exercise Notice]
Schedule 2
Selected Option Territories
D5 Territories with Elan Option
Part A – European Selected Option Territories
Part B – Non-European Selected Option Territories
[To be inserted as per Licence Option Exercise Notice]
| Signed by [NAME OF DIRECTOR] | ||
| for and on behalf of [NAME OF LICENSOR] |
Director | |
| Signed by [NAME OF DIRECTOR] | ||
| for and on behalf of [NAME OF LICENSEE] |
Director | |
SCHEDULE 3
PART A
European Option Territories
| Austria |
| Bosnia and Herzegovina |
| Bulgaria |
| Croatia |
| Czech Republic |
| Estonia |
| Hungary |
| Latvia |
| Lithuania |
| Poland |
| Romania |
| Serbia |
| Slovak Republic |
| Slovenia |
| Switzerland |
| Turkey |
PART B
Non-European Option Territories
| Bahrain | Algeria |
| Iran | Burundi |
| Iraq | Djibouti |
| Israel | Egypt |
| Jordan | Ethiopia |
| Kuwait | Ghana |
| Lebanon | Ivory Coast |
| Oman | Kenya |
| Qatar | Lesotho |
| Saudi Arabia | Libya |
| Syria | Malawi |
| UAE | Morocco |
| Yemen | Nigeria |
| Rwanda | |
| Senegal | |
| South Africa | |
| Sudan | |
| Swaziland | |
| Tanzania | |
| Tunisia | |
| Uganda |
Part C
Generics Territory
| Latin America | North America | Western Europe |
| Argentina | Canada | All 27 EU Member States |
| Belize | United States of America | and Norway, |
| Bolivia | Liechtenstein and Iceland. |
| Brazil | ||
| Chile | ||
| Columbia | ||
| Costa Rica | ||
| Ecuador | ||
| El Salvador | ||
| Guatemala | ||
| Honduras | ||
| Mexico | ||
| Nicaragua | ||
| Panama | ||
| Paraguay | ||
| Peru | ||
| Uruguay | ||
| Venezuela |
SCHEDULE 4
Licence Option Exercise Notice
[TO BE EXECUTED ON ELAN PHARMA INTERNATIONAL HEADED NOTEPAPER]
[NEWCO ADDRESS]
We wish to notify you that, pursuant to the Option and Royalty Agreement between Elan Pharma International Limited ("EPIL") and [ NEWCO ], dated [ ] (the "Option Agreement"), EPIL are exercising the Licence Option (as defined in the Option Agreement) in respect of:
(i) the following Option Territories (as that term is defined in the Option Agreement):
[ INSERT THE SELECTED OPTION TERRITORIES ]
(ii) the following Patents (as defined in the Option Agreement):
[ INSERT DETAILS OF PATENTS ]
| Docket no. | CC | Status | Filing Date | Appln No. | Grant No. |
and
(iii) all related Know-How and/or Products (as those terms are defined in the Option Agreement)
IN WITNESS whereof this Agreement has been executed as a deed on the date and year first herein written.
GIVEN under the common seal
of ELAN PHARMA INTERNATIONAL LIMITED
in the presence of
| Director | |
| Director/Secretary |
GIVEN under the common seal
of SPERANZA BIOPHARMA LIMITED
in the presence of
| Director | |
| Director/Secretary |
EXHIBIT C
Shareholders' Agreement
AGREED FORM
SUBJECT TO CONTRACT / CONTRACT DENIED
Dated 2013
ELAN SCIENCE ONE LIMITED
(ES1)
and
THE PERSONS LISTED IN SCHEDULE 1
(the Management Subscribers)
and
NERANO PHARMA LIMITED
(Nerano)
and
[ • ]
(the Employee Share Nominee)
and
SPERANZA THERAPEUTICS LIMITED
(the Company)
SUBSCRIPTION AND SHAREHOLDERS AGREEMENT
in respect of Speranza Therapeutics Limited
ãA & L GOODBODY
| 1 |
THIS AGREEMENT is dated 2013 and made between:
| (1) | Elan Science One Limited (ES1), a private limited company, incorporated in Ireland (no. 460037) whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2. |
| (2) | The Persons listed in Schedule 1 (the Management Subscribers). |
| (3) | Nerano Pharma Limited (Nerano), a private limited company, incorporated in Ireland (no. 527689) whose registered office is at 45 Fitzwilliam Square, Dublin 2; |
| (4) | [ • ] (the Employee Share Nominee); |
| (5) | Speranza Therapeutics Limited, a private company limited by shares incorporated in Ireland (no. 527543) whose registered office is at McCann FitzGerald, Riverside One, Sir John Rogerson's Quay, Dublin 2 (the Company). |
(together the 'parties' and each a 'party')
RECITALS
| A. | The Company, immediately before the subscription for the Shares provided for in this Agreement, has an authorised share capital of US$10,000 divided into 1,000,000 ordinary shares of US$0.01 each, of which (i) 6,200 have been validly issued, fully paid and are legally and beneficially held by Nerano, (ii) [ • ] have been validly issued, fully paid and are legally and beneficially held by the Management Subscribers and (iii) [ • ] have been validly issued, fully paid and are legally and beneficially held by the Employee Share Nominee but which constitute Unallocated Employee Shares. |
| B. | ES1 has agreed to subscribe US$18.00 for the issue to it of the Subscription Shares on the terms and subject to the conditions contained in this Agreement. |
| C. | The parties have agreed to enter into this Agreement to record: |
| (i) | the Subscription for the Subscription Shares by ES1 on the terms and conditions set out in this Agreement; |
| (ii) | the manner in which the parties have agreed to regulate their relationship with each other as Shareholders of the Company; and |
| (iii) | the future operation and conduct of the Business of the Company. |
IT IS HEREBY AGREED as follows:
| 1. | DEFINITIONS AND INTERPRETATION |
| 1.1. | In this Agreement, unless the context otherwise requires: |
1990 Act means the Companies Act 1990;
an Affiliate of any person means:
| (a) | any holding company or subsidiary of that person and any subsidiary of any such holding company; and |
| (b) | any other individual, company, body corporate, partnership or other entity which; |
| (i) | is Controlled by that person; |
| (ii) | Controls that person; or |
| (iii) | is Under Common Control with that person; |
(provided, however, that the Company shall be considered to be an Affiliate of any Shareholder or of any other Affiliate of any Shareholder);
| 2 |
Agreement means this subscription and shareholder's agreement;
Alteration of Share Capital means the issue, allotment, redemption or purchase by the Company of any Shares, loan stock, debentures, or other securities or instruments convertible into Shares or debentures of the Company, or any share warrants in respect of any Shares, or the grant by the Company of any option or right in respect of any of the foregoing, in the capital of the Company (including any shares, debentures, securities convertible into shares or debentures or share warrants previously issued or allotted by it), the alteration by the Company in any way of the rights attaching to any class of Shares, or the reorganisation, reduction, consolidation, subdivision or conversion of its Shares capital in any way;
Annual Budget means the itemised individual and consolidated revenue and capital budgets of the Company for the next following year, showing proposed revenues, expenses and capital expenditure, trading and cash flow figures and all anticipated material commitments for each such period to be in such form and to contain such other information as the Board may reasonably require;
Articles means the articles of association for the time being of the Company (an agreed form of which shall be adopted on or before Completion in substitution for and to the exclusion of the existing articles of association of the Company);
Asset Sale means the disposal (whether by sale, transfer, lease or otherwise and whether through a single transaction or a series of connected transactions) to any person and/or Connected Persons of that person of the legal and/or beneficial interest in or title to the whole or a substantial part of the undertaking and assets of the Company, for which purposes a substantial part means a proportion representing or accounting for 75 per cent or more of the total consolidated assets or gross consolidated turnover of the Company as determined by reference to the latest available audited or consolidated accounts of the Company (or, if no such accounts are available, as determined by the Board, acting reasonably;
Auditors means the auditors for the time being of the Company;
Board means the board of Directors of the Company as from time to time constituted;
Business means the D5 Business, more particularly described in the Business Plan which may be updated and amended in accordance with the terms of this Agreement from time to time;
Business Day means any day on which banks are generally open for business in Dublin, excluding Saturdays and Sundays;
Business Plan means the business purpose plan of the Company, to be agreed by the parties in accordance with the provisions of Schedule 8, paragraph 4;
Business Transfer Agreement means an agreement entered into on the date hereof between EPIL and Irish OpCo under which EPIL transferred the D5 Business to Irish OpCo;
Capital Contribution means the sum of $63,000,000 which ES1 contributed to Irish OpCo on the date hereof to fund the development of the D5 Business;
Chairman means the chairman for the time being of the Board;
Clinical Plan means the plan that sets out the clinical goals of the Company in relation to the D5 Business, as agreed by ES1 and Nerano prior to the Completion of this Agreement, as set out at Schedule 14;
Completion means completion of this Agreement in accordance with clause 3;
Confidential Information means:
any and all information:
| (a) | which is or was used in or otherwise relates to the business, customers or financial or other affairs of the Company; or |
| 3 |
| (b) | in respect of which the Company is bound by an obligation of confidence to a third party; or |
| (c) | which is or was received or obtained as a result of entering into or performing, or which is or was supplied by or on behalf of a party in the negotiations leading to, this Agreement; and |
the existence of, or any of the terms or provisions of, any of the Transaction Documents;
Connected Person means in relation to a person, any other person (a) who is connected for the purposes of Section 10 of the Taxes Consolidation Act 1997 to the first-mentioned person; and/or (b) who, together with the first-mentioned person, constitute Persons Acting in Concert; and/or (c) any Affiliate of the first-mentioned person;
Control means the power of a person (whether alone or together with any other person or persons) to secure directly or indirectly, including through one or more intermediaries, that the affairs of another person are conducted in accordance with the wishes of the first-mentioned person, either by means of the holding of shares or the possession of voting power directly or indirectly in or in relation to that or any other person, or by virtue of any powers conferred by any applicable laws or regulations or the constitutional or other documentation regulating or managing the affairs of that or any other person, or otherwise, and the expressions Controlled, Controls and Under Common Control shall be construed accordingly;
Controlling Interest means an Interest in Shares, or Interests in Shares, representing in aggregate over 50 per cent of the total voting rights of all members of the Company for the time being;
D5 Business means the research, development, manufacture and commercialization of a Product;
Deed of Adherence means a deed in the form set out in Schedule 13 (Deed of Adherence) providing for the person entering into it to adhere in full to the terms of this Agreement as if such person was party to this Agreement as Shareholder;
Derivative means any compounds identified, obtained, developed, created, synthesized, generated, designed or resulting from, based upon, containing or incorporating the chemical structure of Scyllo-Inositol as those compounds are generically and/or specifically disclosed and claimed in the Licensed Patent Rights;
Directors mean the directors of the Company from time to time, each a Director;
Dispute means any suit, action, proceedings and/or any dispute or difference which may arise out of or in connection with or which may relate in any way to any of the Transaction Documents (including but not limited to any suit, action, proceedings, dispute or difference relating to the formation, interpretation or performance of any of the Transaction Documents) or any dispute or difference which may arise out of or in connection with or which may relate in any way to any non-contractual obligations of any nature (including those to which Regulation (EC) No. 864/2007 applies) between the parties or any of them and Disputes shall be construed accordingly;
Effective Date means the date of this Agreement;
Emoluments means emoluments of every description including, without limitation, salaries, directors' fees, bonuses or commissions (by whatever name called), profit shares under any incentive scheme, pension contributions payable by the Company, and benefits in kind of any description as quantified for income tax purposes;
Encumbrance includes any interest or equity of any person (including any right to acquire or option) or any mortgage, charge, pledge, lien, assignment, hypothecation, security interest (including any created by law), title retention or other security agreement or arrangement or a rental, hire purchase, credit sale or other agreement for payment on deferred terms;
EPIL means Elan Pharma International Limited, a private limited company incorporated in Ireland (Company No. 222276) whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2;
| 4 |
Executive Employment Agreement means the employment agreement to be entered into between Irish OpCo and Seamus Mulligan on Completion;
Group means the Company and each of the Subsidiaries and any other subsidiary from time to time of the Company (whether incorporated before or after the date of this Agreement), and Group Company means any of them, as the case may be;
Industry Expert has the meaning given in clause 10.1;
Initial Funding means the Capital Contribution and the Shareholder Loans;
Interest in Shares shall, with respect to any Share or Shares, have the meaning given to that term in Section 54 of the 1990 Act, and, for the avoidance of doubt, includes:
| (a) | any right to control the voting or other rights attributable to any Share or Shares, disregarding any conditions or restrictions to which the exercise of any such right may be subject; |
| (b) | notwithstanding the provisions of sub-section (10) of Section 54 of the 1990 Act or any other provision of the Companies Acts, any option over, and/or right to subscribe for, any Share or Shares; and |
| (c) | any right or entitlement howsoever described (whether absolute or conditional) to receive, or to direct the payment or receipt of, any dividend or other distribution referable to any Share or Shares; |
and interests and interested shall be construed accordingly;
Investors means Nerano and ES1 and Investor means either of them;
Ireland means Ireland (excluding Northern Ireland) and Irish will be construed accordingly;
Irish OpCo means Speranza Biopharma Limited, a limited liability company (Company No.502444) with and address at Treasury Building, Lower Grand Canal Street, Dublin 2;
Licensed Patent Rights shall have the meaning ascribed to it in the collaboration agreement (as amended) between Waratah Pharmaceuticals Inc. and EPIL dated 25 September 2006;
Listing means any application for admission to listing or trading of all or any part of the share capital, debentures, notes or other securities of the Company, on any stock exchange, trading facility or securities market (including any “grey”, unregulated or unofficial market) in Ireland, the United Kingdom, any member state of the European Union, the United States of America, or any other jurisdiction, including without limitation the Main Securities Market of the Irish Stock Exchange, the main market for listed securities of the London Stock Exchange, AIM, the ESM, the New York Stock Exchange, and the market known as NASDAQ;
Loan Agreements means the loan agreements of even date herewith under which Nerano is providing Irish OpCo with a ten year interest-free loan of $20,000,000 to fund development of the D5 Business, and EPIL is providing Irish OpCo with a ten year interest-free loan for $7,000,000 to fund development of the D5 Business;
Management Representative shall have the meaning ascribed to it in clause 5.2 of this Agreement;
Material means, unless otherwise expressed, material in the context of the business, assets, liabilities or financial condition of the Company taken as a whole and Materially will be construed accordingly;
Memorandum means the memorandum of association of the Company as amended from time to time;
Option Agreement means the option and royalty deed of agreement entered into by EPIL and Irish OpCo on the date hereof under which EPIL is granted the option to commercialize the Products in certain territories and to receive a royalty in relation to the Products from Irish OpCo;
| 5 |
Persons Acting in Concert means persons who, pursuant to an agreement or undertaking (whether formal or informal) actively co-operate, through the acquisition by any of them of any Shares or Interests in Shares, to obtain or consolidate a Controlling Interest in the Company and persons coming within the definition of Persons Acting in Concert set out in the Takeovers Rules in force at the date of this Agreement, save that the parties hereto will not be Persons Acting in Concert solely by virtue of having entered into any of the Transaction Documents;
Post-Termination Provisions means the provisions in clause 12.5 (Announcements), clause 12.6 (Confidentiality), clause 12.8 (Costs and Expenses), clause 12.10 (Whole Agreement), clause 12.13 (Remedies Cumulative), clause 12.16 (Notices), clause 12.19 (Governing Law), clause 12.20 (Jurisdiction), paragraph 4.1.2 of Schedule 10 (Transfer of Shares).
Proceedings means suits, actions or proceedings arising out of or in connection with or relating in any way to any Dispute;
Product means any pharmaceutical preparation or dosage form containing Scyllo-Inositol or a Derivative;
Proposing Transferee shall have the meaning ascribed to such term in Schedule 10 (Transfer of Shares);
Proposing Transferor shall have the meaning ascribed to such term in Schedule 10 (Transfer of Shares);
Realisation means a Share Sale, an Asset Sale or a Listing;
Registered Shareholder means any person who is registered as a holder of a Share or Shares in the register of members of the Company from time to time;
Regulatory Approval means any necessary approval required by any competent governmental, regulatory or supervisory authority or agency in Ireland or elsewhere;
Sale Price shall have the meaning ascribed to such term in Schedule 10 (Transfer of Shares);
Sale Share Completion Date shall have the meaning ascribed to such term in Schedule 10 (Transfer of Shares);
Sale Shares shall have the meaning ascribed to such term in Schedule 10 (Transfer of Shares);
Scyllo-Inositol is the compound “AZD-103”, as more specifically disclosed in the Licensed Patent Rights, the structure of which is set out in Schedule 16;
Share means any ordinary share for the time being in issue in the capital of the Company and Shares shall be construed accordingly;
Share Purchase Agreement means an agreement of even date herewith between EPIL and the Company under which EPIL has transferred the entire issued share capital of Irish OpCo to the Company;
Share Sale means the transfer or other disposal (whether through a single transaction or a series of connected transactions, including by way of a scheme or arrangement under the Companies Acts) of the legal and/or beneficial interest in or title to 50% or more of the Shares to a person and/or any Connected Persons of that person;
Shareholders means any person for the time being holding a legal or beneficial interest of any kind, howsoever described, whether absolute or conditional, in Shares, including the Registered Shareholders and any person holding an Interest in Shares, and Shareholder means any one of them. For the purposes of this Agreement, the Management Subscribers shall be considered as one Shareholder, represented by the Management Representative in accordance with clause 5.2 of this Agreement;
Shareholder Group, in relation to any Shareholder which is a body corporate, means that Shareholder, any holding company of that Shareholder and any subsidiary of any such holding company;
| 6 |
Shareholder Loans means the loans granted on the date hereof pursuant to the Loan Agreements;
Subscriber means ES1;
Subscription means the subscription by ES1 for the Subscription Shares pursuant to this Agreement;
Subscription Amount means the sum of US$18.00 to be paid by ES1 on Completion for the Subscription Shares;
Subscription Shares means 1,800 ordinary shares of US$0.01 each in the capital of the Company to be subscribed for by ES1 pursuant to clause 2;
Subsidiaries means the companies listed in Schedule 3;
Tax means all forms of taxation, duties, imposts and levies whether of Ireland or elsewhere, including (but without limitation) income tax, corporation tax, corporation profits tax, advance corporation tax, capital gains tax, capital acquisitions tax, residential property tax, wealth tax, value added tax, dividend withholding tax, deposit interest retention tax, customs and other import and export duties, excise duties, stamp duty, capital duty, social insurance, social welfare or other similar contributions and other amounts corresponding thereto whether payable in Ireland or elsewhere, and any interest, surcharge, penalty or fine in connection therewith, and the word taxation shall be construed accordingly;
Termination Event means a clinical event (including any results of a clinical trial) or determination or action by a regulatory body relating to Products or the Business which means that either (a) there is no reasonable prospect of the Group achieving the targets set out in the Business Plan within the time frame contemplated by the Business Plan or (b) the Business is no longer commercially viable;
Transaction Documents means:
| (a) | this Agreement; |
| (b) | the Business Transfer Agreement; |
| (c) | the Share Purchase Agreement; |
| (d) | the Option Agreement; |
| (e) | the Loan Agreements; |
| (f) | all documents or agreements, connected with or ancillary to any of the documents or agreements referred to at paragraphs (a) to (e) above, which are in the agreed form; and |
| (g) | any other document or agreement, including any side letters, which are entered into, or to be entered into, on Completion (or at any time during the period from the date of this Agreement up to, including and after the date of Completion) by some or all of the parties to this Agreement in connection with or which are ancillary to any of the foregoing documents or agreements; |
Transfer Notice shall have the meaning ascribed to such term in Schedule 10 (Transfer of Shares);
Unallocated Employee Shares means the [ • ] Shares held by the Employee Share Nominee as at the date of this Agreement, together with any Shares which are acquired by the Employee Share Nominee pursuant to paragraph 2 of Schedule 10, which are to be transferred to existing or future employees or contractors of the Group in the manner contemplated by clause 6.5;
and cognate terms will be construed accordingly.
| 1.2. | Unless otherwise provided, references to recitals, clauses, paragraphs, schedules and annexures are to recitals, clauses, paragraphs, schedules and annexures contained in this Agreement and reference to this Agreement includes reference to its schedules. |
| 1.3. | Headings and the contents page are inserted for convenience only and do not affect the construction of this Agreement. |
| 7 |
| 1.4. | A document is in the agreed form if it is in the form of a draft agreed between and initialled by or on behalf of the parties hereto (or such of the parties as are party to it) on or before the date hereof. |
| 1.5. | All indemnities, obligations, covenants and representations arising under this Agreement, given or entered into by more than one person are given or entered into severally. |
| 1.6. | If any action or duty to be taken or performed under any of the provisions of this Agreement would fall to be taken or performed on a day which is not a Business Day, such action or duty shall be taken or performed on the Business Day next following such day. |
| 1.7. | Unless expressly stated in this Agreement or the context otherwise admits or requires, any reference in this Agreement to: |
| 1.7.1. | a word or phrase the definition of which is contained or referred to in Section 2 of the Companies Act 1963 has the meaning thereby attributed to it; |
| 1.7.2. | the singular includes the plural and vice versa, any gender includes the other gender, and he or she, his or hers, him or her, where the context so admits, includes it or its, as the case may be, and vice versa; |
| 1.7.3. | a person is deemed to include a reference to a natural person, firm, partnership, company, corporation, association, body corporate, trust, investment fund, government, state or agency (in each case whether or not having a separate legal personality), wheresoever incorporated or resident, but a reference to an individual is deemed to be a reference to a natural person only; |
| 1.7.4. | any provision of law is a reference to that provision as amended, substituted, extended or re-enacted; |
| 1.7.5. | any Irish legal term, concept, legislation or regulation (including, without limitation, any action, remedy, method of judicial proceeding, document, statute, court, court official, governmental authority or agency or any accounting term or concept) in respect of any jurisdiction other than Ireland will be construed as a reference to the term, concept, legislation or regulation which most nearly corresponds to it in that jurisdiction; |
| 1.7.6. | a regulation includes any regulation, rule, official directive, request or guideline (whether or not having the force of law) of any governmental, intergovernmental or supranational body, agency, department or regulatory, self-regulatory or other authority or organisation; |
| 1.7.7. | any phrase introduced by the terms including or includes or in particular or any similar expression is to be construed as illustrative and without limitation; |
| 1.7.8. | costs, charges or expenses include any value added tax or similar tax charged or chargeable in respect thereof; |
| 1.7.9. | time are references to Irish time; |
| 1.7.10. | a person includes his or its successors, personal representatives, permitted assigns and permitted transferees or substitutes as appropriate; |
| 1.7.11. | writing or similar expressions includes, where the context so admits, transmission by fax; |
| 1.7.12. | this Agreement, any other document or any specified provision of this Agreement or any other document is a reference to this Agreement, to such other document or to such provision as in force for the time being and as amended, extended, varied, assigned, novated, restated or supplemented from time to time; |
| 1.7.13. | words such as hereby, hereunder, hereto, hereof and herein and other words commencing with here shall unless the context clearly indicates to the contrary refer to the whole of this Agreement and not to any particular section, clause, paragraph or schedule of this Agreement; and |
| 1.7.14. | a party or the parties is a reference to a party or the parties to this Agreement. |
| 8 |
| 1.8. | It is acknowledged and agreed by the parties that the provisions of this Agreement have been negotiated, drafted and settled jointly by and on behalf of the parties and accordingly if any question arises at any time as to the meaning, intent or interpretation of any provision or provisions of this Agreement, no presumption or burden of proof shall arise in favour of or against any party solely as a result of the authorship of any of the provisions of this Agreement. |
| 2. | SUBSCRIPTION FOR SHARES |
| 2.1. | Subscription for, and Issue of, Subscription Shares: Subject to the terms and conditions set out in this Agreement, on Completion, ES1 shall subscribe for, and the Company shall allot and issue, the Subscription Shares to ES1, credited as fully paid. |
| 2.2. | Consideration for Issue: The consideration for the issue of the Subscription Shares to be subscribed for by ES1 pursuant to clause 2.1 shall be the payment, on Completion, by ES1 of the amount of the Subscription Amount. |
| 2.3. | Simultaneous Completion: ES1 will not be obliged to complete the subscription for any of the Subscription Shares to be subscribed for pursuant to clause 2.1 unless the subscription of all such Subscription Shares is completed simultaneously. |
| 2.4. | Waiver of Pre-emption Rights: Nerano and the Management Subscribers hereby waive any and all pre-emption rights or other rights or restrictions on the issue of the Subscription Shares to which they may be entitled under the Articles, any previous articles of association of the Company, this Agreement or otherwise, the enforcement of or compliance with which may or might restrict, prevent or delay the subscription, allotment and issue of the Subscription Shares to be subscribed pursuant to this clause 2 (and shall procure the waiver of all other rights of pre-emption as may be required to give full effect to this Agreement) and hereby consent to such Subscription, allotment and issue and to any and all variations of the rights attaching to the Shares held by them as a consequence of the issue of the Subscription Shares. |
| 3. | COMPLETION |
| 3.1. | Completion: Completion shall take place on the Effective Date at such address as the parties may agree. |
| 3.2. | Obligations of Shareholders and the Company at Completion: Upon Completion, each of the parties shall carry out all the obligations set out in Schedule 5 (Completion Obligations). |
| 4. | WARRANTIES |
| 4.1. | Company Warranties: The Company warrants to each of the Investors as at the date of this Agreement that each of the warranties set out in Schedule 4 is true and accurate and not misleading. |
| 4.2. | Parties’ Warranties: Each party warrants to each of the other parties that: |
| 4.2.1. | it / he has full power, legal capacity and authority: |
| (1) | to enter into, execute and deliver this Agreement; and |
| (2) | to perform its obligations set out in this Agreement. |
| 4.2.2. | the obligations set out in this Agreement constitute legal obligations that are valid, binding on and enforceable against it / him in accordance with its respective terms. |
| 4.2.3. | neither entering into this Agreement nor performing the obligations referred to in it has resulted or will result in the breach of any obligation of it / him under: |
| (1) | (in the case of a party which is a corporate body) its memorandum or articles of association, statutes, by-laws or other terms of charter or corporate regulation; |
| (2) | any law or any order, judgment or decree of any court or governmental agency; or |
| 9 |
| (3) | any contract, undertaking or agreement. |
| 5. | MANAGEMENT AND GOVERNANCE OF THE COMPANY |
| 5.1. | Board of Directors: Subject as provided for in this Agreement, the business of the Company shall be undertaken and transacted by the Board who at all times shall act in the best interests of the Company and its Shareholders, and the Company shall not enter into any contract, transaction or arrangement whereby its business would be controlled otherwise than by the Board. The Board and its committees shall be constituted and governed in accordance with the provisions set out in Schedule 6 (Board Composition and Governance). |
| 5.2. | Management Representative: |
| 5.2.1. | The Management Subscribers shall be represented in all matters in relation to the Group, the Business and in all matters governed by this Agreement, by a representative chosen from amongst their number who will be the Management Representative. |
| 5.2.2. | Where any matter set out in this Agreement requires the consent, vote or nomination of the Management Subscribers, the Management Representative shall cast one vote, consent or nomination on behalf of all the Management Subscribers, based on the will of the majority of the Management Subscribers. This vote shall represent all the Shares held by the Management Subscribers. For example, if the Management Subscribers hold in aggregate 2,000 Shares, the Management Representative shall vote the entirety of the 2,000 Shares based on the will of the majority (in holdings of Shares) of the Management Subscribers. |
| 5.3. | Shareholder Information and Access Rights: The Investors shall be entitled to the information and access rights set out in Schedule 7 (Information and Access Rights). |
| 5.4. | Shareholder Covenants Regarding Business Management and Corporate Governance: Each of the parties hereto undertakes to each other that each shall comply with the provisions set out in Schedule 8 (Shareholder Covenants), and shall procure (and shall use all such voting and other rights to which they are entitled, whether arising under statute, at common law, in equity, under contract or otherwise, to procure), insofar as it is possible having regard to their respective rights and powers in respect of the Company, that all such provisions shall be complied with in full by the Company and that the business and affairs of the Company shall at all times be managed and carried out in accordance with such provisions. |
| 5.5. | Reserved Matters: Each of the parties hereto undertake with each other that each shall procure (and shall use all such voting and other rights to which they are entitled, whether arising under statute, at common law, in equity, under contract or otherwise, to procure), as far as it is possible having regard to their respective rights and powers in respect of the Company, that the Company shall not do any of the matters set out in Schedule 9 (Reserved Matters) without the prior written consent of each of the Investors. |
| 6. | TRANSFER OF SHARES |
| 6.1. | Transfers Prohibited Except in Accordance with this Agreement: Notwithstanding any other provision of this Agreement or the Articles, no Shareholder shall: |
| 6.1.1. | pledge, mortgage (whether by way of fixed or floating charge) or otherwise create any Encumbrance over or in respect of its legal or beneficial interest in any of its Shares or any Interest in Shares; |
| 6.1.2. | sell, transfer or otherwise dispose of any of its Shares or any Interest in Shares; |
| 6.1.3. | enter into any agreement, arrangement, or understanding (whether legally binding or not) in respect of the votes attached to any of its Shares or Interest in Shares; or |
| 6.1.4. | agree, whether or not subject to any condition precedent or subsequent, to do any of the foregoing; |
otherwise than in accordance with the provisions of this Agreement (and in particular the provisions of
| 10 |
Schedule 10 (Transfer of Shares)) and the Articles, and the Board shall refuse to register any transfer which is not so in accordance.
| 6.2. | Restrictions on Transfer: The parties agree to be bound by the provisions regarding the transfer and transmission of Shares and related matters set out in Schedule 10 (Transfer of Shares). |
| 6.3. | Transfers of Shares and Interests in Shares: |
| 6.3.1. | For the avoidance of doubt, the parties agree that the provisions of this clause 6 and of Schedule 10 (Transfer of Shares) regarding the transfer and transmission of Shares shall also apply mutatis mutandis to any Interest in Shares held by any person. |
| 6.3.2. | Each Shareholder shall at the written request of the Board promptly inform, and keep informed, the Company in writing (and in any event within 7 days of being requested in writing to do so by the Board) as to the beneficial ownership and control of such Shareholder and the beneficial ownership and control of Shares and Interests in Shares held by such Shareholder. |
| 6.4. | Transfer of Entire Interest: Any transfer of Shares or Interests in Shares contemplated under this Agreement shall be deemed to carry an obligation to transfer the entire legal and beneficial interest in such Shares or Interests in Shares as the case may be, free from any lien, charge or other Encumbrance of any nature whatsoever (other than any security granted in favour of a licensed bank where the licensed bank in question has given its consent in writing to the transfer). |
| 6.5. | Issue of Shares to Management: |
| 6.5.1. | The Investors and the Employee Share Nominee agree and acknowledge that all of the Unallocated Employee Shares will be transferred to employees and/or contractors of the Group identified in the manner contemplated by this clause 6.5 within a period of 12 months following their acquisition by the Employee Share Nominee (the Allocation Period). |
| 6.5.2. | From time to time during the Allocation Period of any Shares, and following consultation with the Management Representative, Nerano may nominate: |
| (1) | an employee or contractor to whom it proposes that Unallocated Employee Shares be transferred (a Nominated Employee); and |
| (2) | the number of Unallocated Employee Shares that it proposes be transferred to the Nominated Employee (the Nominated Allocation). |
| 6.5.3. | Promptly after a nomination pursuant to clause 6.5.2 (a Nomination), the Board shall consider the Nomination. Following such consideration, the Board shall either: |
| (1) | confirm the Nomination; |
| (2) | modify the Nominated Allocation specified in the Nomination; or |
| (3) | reject the Nomination. |
| 6.5.4. | If the Board: |
| (1) | confirms a Nomination, it will issue a written notice to the Employee Share Nominee instructing it to transfer the Nominated Allocation to the Nominated Employee as soon as reasonably practicable; or |
| (2) | modifies a Nomination, it will issue a written notice to Employee Share Nominee instructing it to transfer the Nominated Allocation (as modified) to the Nominated Employee as soon as reasonably practicable, |
(any such notice being an Allocation Notice).
| 6.5.5. | The Employee Share Nominee undertakes to the Company and to ES1 that, as soon as reasonably practicable following receipt of an Allocation Notice, subject to clause 6.3, it will make the transfer referred to in the Allocation Notice using Unallocated Employee Shares for |
| 11 |
this purpose
| 6.5.6. | Subject to law, the Company shall discharge any liability to Tax of the Employee Share Nominee which arises from the holding or transfer of any Unallocated Employee Shares. |
| 6.5.7. | For the avoidance of doubt, no Shares shall be transferred to a Nominated Employee unless that Nominated Employee has first executed a Deed of Adherence in compliance with clause 9.3. |
| 7. | SHARE ISSUANCES |
| 7.1. | Pre-emption on Issue of Shares: The parties agree to be bound by the provisions set out in Schedule 11 (Pre-emption on Issue of Shares) regarding pre-emption in respect of the future issue of Shares. |
| 7.2. | Anti-Dilution Rights: The parties agree to be bound by the provisions set out in Schedule 12 (Anti-Dilution) regarding anti-dilution in respect of the future issue of Shares. |
| 8. | ADDITIONAL FUNDING REQUIREMENTS |
| 8.1. | BP Funding Situation means a situation where the Group requires additional funding (whether immediately or in the reasonably foreseeable future) in order to have sufficient finance so as to permit it to complete the clinical activities and/or obtain the regulatory approvals which are necessary for the Group to achieve the targets set out in the Business Plan. |
| 8.2. | If the Board, acting reasonably and in good faith, determines that a BP Funding Situation has occurred, it may give written notice to this effect (a BP Funding Notice) to each of the Investors, which notice shall: |
| 8.2.1. | state, with reasonable specificity, the clinical activities and/or regulatory approvals and/or other circumstances which give rise to the BP Funding Situation; |
| 8.2.2. | provide reasonable details of the costs incurred by the Group since incorporation and estimates of the likely costs to be incurred by the Group in achieving clinical activities and/or regulatory approvals and/or other circumstances which give rise to the BP Funding Situation; and |
| 8.2.3. | state, with reasonable specificity, the amount of additional funds, which in no circumstances may exceed, either individually or in aggregate with amounts contributed by the Investors pursuant to previous BP Funding Notices, US$10,000,000, which is required to fund the Group so that it has sufficient finance to complete the activities and the obtaining of the approvals referred to in the BP Funding Notice (the Required BP Funding). |
| 8.3. | At any time within 20 Business Days of service of a BP Funding Notice, an Investor may: |
| 8.3.1. | request, and the Company shall provide as soon as reasonably possible and in any event within 5 Business Days, additional information reasonably requested by that Investor to determine the validity of the BP Funding Notice; and/or |
| 8.3.2. | serve written notice on the Company (copied to the other Investor) stating that it does not accept that a BP Funding Situation has occurred (a BP Dispute Notice). If a BP Dispute Notice is served by an Investor, the Company shall refer the question of whether a BP Funding Situation has occurred to an Industry Expert who shall act in the manner specified in clause 10. |
| 8.4. | If the Board serves a BP Funding Notice and either: |
| 8.4.1. | neither Investor serves a BP Dispute Notice in accordance with clause 8.3; or |
| 8.4.2. | an Investor serves a BP Dispute Notice in accordance with clause 8.3 but the Industry Expert determines in accordance with clause 10 that a BP Funding Situation has occurred, |
as soon as reasonably practicable (and, in any event, no later than the 30 Business Days after the service of the BP Funding Notice, or, where a BP Dispute Notice is served, no later than 10 Business Days after the determination of the Industry Expert):
| 12 |
| (1) | ES1 shall provide to the Company an amount in cash equal to 7/9 of the Required BP Funding; and |
| (2) | Nerano shall provide to the Company an amount in cash equal to 2/9 of the Required BP Funding. |
| 8.5. | Any Required BP Funding which is to be provided to the Company by the Investors in accordance with this clause 8 shall be provided in the form of either loans or capital contributions by the Investors as may be agreed between the Investors. If the Investors fail to so agree, the Required BP Funding shall be provided in the form of interest-free loans to the Company which are substantially in the same form as the Shareholder Loans. |
| 9. | TERMINATION EVENT |
| 9.1. | If at any time it determines that a Termination Event has occurred, an Investor may give written notice to this effect (a Cessation Notice) to the other Investor. Within 20 Business Days of receipt of a Cessation Notice, the recipient shall by notice in writing to the other Investor (a Response Notice) confirm that either: |
| 9.1.1. | it agrees that a Termination Event has occurred; |
| 9.1.2. | it does not agree that a Termination Event has occurred. |
If the recipient Investor fails to serve a Response Notice within 20 Business Days of receipt of a Cessation Notice, it shall be deemed to have accepted that a Cessation Notice has occurred.
| 9.2. | If an Investor serves a Response Notice stating that it does not agree that a Cessation Notice, the Investors shall refer the question of whether a Cessation Dispute has occurred to an Industry Expert who shall act in accordance with clause 10. |
| 9.3. | If the Investors agree, or an Industry Expert determines, that a Termination Event has occurred, the parties shall take all actions to procure the winding-up of the Company on the basis that the assets available for distribution following discharge of all liabilities other than the Shareholder Loans (the Net Proceeds) shall be used to repay the Shareholders Loans and distributed to the Investors on the following basis: |
| 9.3.1. | if the Termination Event occurs before the date which is 15 months after Completion: |
| (1) | first, the Shareholder Loans shall be repaid in full (or pro-rata in the event of a shortfall); and |
| (2) | thereafter, the remaining Net Proceeds shall be paid to ES1; |
| 9.3.2. | if the Termination Event occurs on or after the date 15 months after Completion |
| (1) | 2/9 of the Net Proceeds shall be used in repaying the balance owing on the Shareholder Loan advanced by Nerano (and the remaining balance shall be written-off); and |
| (2) | 7/9 of the Net Proceeds shall be used in repaying the balance owing on the Shareholder Loan advanced by ES1 and/or paid to ES1 in such form as it may require. |
| 10. | INDUSTRY EXPERT |
| 10.1. | An Industry Expert shall be an expert in the pharmaceutical industry agreed by the Investors or, failing agreement within five Business Days, nominated by the President for the time being of the Law Society of Ireland. |
| 10.2. | The Industry Expert shall act on the following basis: |
| 10.2.1. | the Industry Expert shall act as an expert and not as an arbitrator and the provisions of the |
| 13 |
Arbitration Acts 2010 shall not apply to him or his determination;
| 10.2.2. | the Industry Expert is to make a decision on the dispute and notify the parties of his decision within 20 Business days of his appointment; |
| 10.2.3. | the parties shall respectively provide all the information relating to the Group and its business which the Industry Expert reasonably requires; |
| 10.2.4. | the decision of the Industry Expert is, in the absence of fraud or manifest error, final and binding on the parties; |
| 10.2.5. | the costs of the Industry Expert shall be paid by the Company. |
| 11. | TERMINATION |
| 11.1. | Duration of Agreement: Except as otherwise provided herein, this Agreement shall continue in full force and effect without limit in point of time until the earliest of the following events, whereupon it shall terminate and be of no further force or effect, subject always to clauses, 7, 8.3 and Schedule 10: |
| 11.1.1. | the completion of a Realisation (save that if a Realisation is an Asset Sale it shall terminate at the winding up of the Company and a distribution of its assets in accordance with the Articles); or |
| 11.1.2. | subject to clause 5.5, the passing of an effective resolution for the winding up of the Company or the making of a binding court order for the winding up of the Company. |
| 11.2. | Termination as regards any Shareholder: Subject to compliance with the provision of this Agreement, this Agreement will cease to have effect and shall terminate as regards any Shareholder who validly ceases to hold or own beneficially any (i) Shares or (ii) Interests in Shares (Interests and each an Interest) on and with effect from the date upon which the Shareholder ceases to hold or own beneficially the last of any such Interests. |
| 11.3. | Effect of Termination: The termination of this Agreement shall not affect, and shall be without prejudice to, any party’s rights or obligations which may have accrued to it against any other party before such termination. The termination of this Agreement shall not affect the continuance in force after such termination of the Post-Termination Provisions and any provisions hereof which are expressed to continue in force or which are by their nature capable of enforcement against any party by any other party after the termination of this Agreement. |
| 12. | MISCELLANEOUS PROVISIONS |
| 12.1. | Relationship with Articles: As between the Shareholders, in the event of any conflict or any ambiguity arising between: |
| 12.1.1. | any of the provisions of this Agreement; and |
| 12.1.2. | any of the provisions of the Memorandum and/or the Articles |
the provisions of this Agreement shall prevail, and accordingly each Shareholder shall exercise all voting and other rights and powers available to it so as to give effect to the provisions of this Agreement and to pass any members’ resolution which is required to ensure that a provision of this Agreement is effective in accordance with its terms.
| 12.2. | Group Companies: Each of the parties hereto undertakes with each other that each shall procure (and shall use all such voting and other rights to which they are entitled, whether arising under statute, at common law, in equity, under contract or otherwise, to procure), as far as it is possible having regard to their respective rights and powers in respect of the Company and each other Group Company, that each of the provisions, conditions and obligations undertaken in this Agreement with respect to the Company (including, but not limited to, clause 5, the provisions of Schedule 6 (Board Composition and Governance) with regard to the rights of Shareholders with respect to the appointment of directors shall be deemed to apply mutatis mutandis to, and be observed, performed and implemented in respect of, each Group Company, and that each Group Company shall, to the extent it is able to do so by law, observe, perform and implement the provisions and conditions in this Agreement to which the Company |
| 14 |
is subject under this Agreement, as if, where the context so admits, any reference to:
| 12.2.1. | the Company included a reference to such other Group Company; |
| 12.2.2. | the Board included a reference to the board of directors for the time being of such other Group Company: |
| 12.2.3. | the Chairman included a reference to the chairman for the time being of the board of directors of such other Group Company; |
| 12.2.4. | the Directors (or any Director) included a reference to the directors (or any director) for the time being of such other Group Company; |
| 12.2.5. | such other similar term included a reference to such similar term in the context of such other Group Company. |
| 12.3. | New Shareholders: |
| 12.3.1. | For the avoidance of doubt, it is a condition precedent to the right of any Shareholder to transfer or dispose of any Shares, any Interest in Shares, or to the allotment or issue of any unissued Shares of the Company, that the transferee or allottee (if not already bound by the terms of this Agreement) (the New Shareholder) executes a Deed of Adherence in the following capacity |
| (1) | if the New Shareholder is an employee of a member of the Group or it is proposed that he should become one, a Management Subscriber; |
| (2) | if the New Shareholder is an investor and the Investors consent in writing, an Investor; and |
| (3) | in all other cases, a party |
| 12.3.2. | The Investors may determine, notwithstanding clause 12.3.1 above, that the New Shareholder should enter into a Deed of Adherence in a different capacity to that required by clause 12.3.1 and may also agree such amendments to the Deed of Adherence as they consider appropriate in the circumstances. |
| 12.3.3. | A New Shareholder who enters into a deed of adherence as a Management Subscriber or an Investor shall have all the rights and obligations as if he were named in this Agreement as a Management Subscriber or Investor (as the case may be). |
| 12.4. | Statutory Powers: The Company shall not be bound by any provision of this Agreement to the extent it would constitute an unlawful fetter on any statutory power of the Company, but such provision shall remain valid, and shall be binding on, and enforceable by and against, all other parties in respect of whom it applies. |
| 12.5. | Announcements: |
| 12.5.1. | No party shall make any announcement to shareholders, employees, customers or suppliers, or to securities markets or other authorities or to the media or otherwise, regarding the subject-matter of the Transaction Documents or any term or provision of any of them without the prior written approval of the other parties to this Agreement. |
| 12.5.2. | Clause 12.5.1 will not apply if, and to the extent that, such announcement is required by any law applicable to the party making the announcement, or by: |
| (1) | bona fide contractual arrangements with unrelated third parties in existence at the date of this Agreement, details of which have been fully disclosed in writing to the other parties to this Agreement on or before the date of this Agreement; or |
| (2) | any stock or securities exchange, regulatory or governmental authority or court having jurisdiction over the party making the announcement, whether or not the requirement has the force of law; |
| 15 |
provided that any such announcement may only be made after prior consultation with the other parties to this Agreement to the fullest extent permitted by law.
| 12.5.3. | If any party proposes to make an announcement pursuant to this clause, it shall provide copies of that proposed announcement to each of the other parties to this Agreement before the announcement is made unless this is not reasonably practicable, in which case, a copy of the announcement shall be so provided to each party as soon as reasonably practicable. |
| 12.5.4. | Each party undertakes to provide all such information known to it or which on reasonable enquiry ought to be known to it in relation to the Group as may reasonably be required by any other party for the purposes of complying with the requirements of law or any securities exchange, regulatory or governmental authority or court having jurisdiction over the party that requires the information. |
| 12.6. | Confidentiality: |
| 12.6.1. | Each of the parties to this Agreement undertakes with each other for the benefit of the other parties and as agent and trustee for the Company that they shall preserve the confidentiality of, and not directly or indirectly reveal, report, publish, disclose, transfer or use for their own or any other purposes Confidential Information except: |
| (1) | in the circumstances set out in clause 12.6.2; |
| (2) | to the extent otherwise expressly permitted by this Agreement; or |
| (3) | with the prior written consent of the other parties to this Agreement and of the party to whose affairs such Confidential Information relates. |
| 12.6.2. | A party is permitted to disclose Confidential Information: |
| (1) | where the Confidential Information is in or, after the date of this Agreement, enters the public domain otherwise than as a result of: (i) a breach by that party of its obligations in this clause or (ii) a breach by the person who disclosed that Confidential Information of a confidentiality obligation where the party is or was aware of such breach; |
| (2) | if and to the extent that the party discloses Confidential Information to any person: |
| (a) | in compliance with any requirement of law; |
| (b) | in response to a requirement of any securities exchange, regulatory or governmental authority or court having jurisdiction over that party; or |
| (c) | in order to obtain Tax or other clearances or consents from the Revenue Commissioners or other relevant Tax or regulatory authorities, |
provided that any such information disclosable pursuant to sub-clauses (a), (b) or (c) may, to the fullest extent permitted by law, be disclosed only after prior consultation with the other parties to this Agreement.
| 12.7. | Assignment: |
| 12.7.1. | None of the parties to this Agreement may assign any of its rights under this Agreement without the prior written consent of each of the other parties except to any person to which it has transferred Shares in accordance with the terms of this Agreement. |
| 12.7.2. | Subject to clause 12.7.1, this Agreement will be binding on and enure for the benefit of the personal representatives, permitted assigns and permitted transferees or substitutes to each of the parties and references to the parties will be construed accordingly. |
| 12.8. | Costs and Expenses: Each party to this Agreement will pay its own costs and expenses of and incidental to this Agreement and the Transaction Documents, and the implementation thereof. |
| 12.9. | Severability: All the terms and provisions of this Agreement and of each of the Transaction Documents |
| 16 |
are distinct and severable, and if any term or provision is held or declared to be unenforceable, illegal or void in whole or in part by any court, regulatory authority or other competent authority it will, to that extent only, be deemed not to form part of this Agreement, and the enforceability, legality and validity of the remainder of this Agreement will not in any event be affected. The parties shall then use all reasonable endeavours to agree to replace the unenforceable, illegal or void term or provision with a term or provision which is legal and enforceable and which has an effect that is as near as possible to the intended effect of the term or provision to be replaced.
| 12.10. | Whole Agreement: The Transaction Documents (including the documents and instruments referred to therein) supersede all prior representations, arrangements, understandings and agreements between the parties relating to the subject-matter thereof, and set forth the entire, complete and exclusive agreement and understanding between the parties relating to the subject-matter thereof. |
| 12.11. | Survival: This Agreement (other than any obligations which have already been fully performed) remains in full force and effect following Completion. |
| 12.12. | Breach: Unless specifically provided to the contrary in this Agreement, any Material breach by any party of the covenants, agreements, commitments or undertakings contained in this Agreement that is not remedied within a reasonable time shall entitle any party at any time to exercise any of its rights to terminate this Agreement and/or to institute proceedings for injunctive relief, rescission or damages or to pursue such other remedies as may be available to it in respect of any such breach whether provided for under this Agreement or otherwise. |
| 12.13. | Remedies Cumulative: The provisions of this Agreement and the rights and remedies of the parties are independent, cumulative and are without prejudice and in addition to any other rights or remedies which a party may have whether arising under statute, at common law, in equity, under contract, by virtue of custom or otherwise. The exercise by a party of any one right or remedy under this Agreement, under statute, at law or in equity will not (unless expressly provided in this Agreement, under statute, at law or in equity) operate so as to hinder or prevent the exercise by that party of any other right or remedy. |
| 12.14. | Waiver: A waiver by any party or parties of any breach of any of the terms, provisions, conditions or covenants of this Agreement or the acquiescence of any party or parties in any act (whether commission or omission) which but for such acquiescence would be a breach, will not constitute a general waiver of the term, provision, condition or covenant or of any subsequent act which is inconsistent with it. |
| 12.15. | Further Assurance: At any time after the date of this Agreement, each party hereto will use reasonable endeavours to procure that any documents are executed and any acts and things are done as may reasonably be required by any other party and at the sole cost and expense of such other party, for the purposes of ensuring that the Subscription takes place, the Subscription Shares are allotted and issued, and the obligations of all parties upon Completion are fully and properly satisfied and discharged. |
| 12.16. | Notices: |
| 12.16.1. | Any notice or other communication to be given or served under this Agreement shall be in writing, addressed to the relevant party and expressed to be a notice or communication under this Agreement and, without prejudice to the validity of another method of service (subject to clause 12.16.3), may be delivered or sent by pre-paid registered post or facsimile addressed as follows: |
to ES1:
Address: Treasury Building, Lower Grand Canal Street, Dublin 2
For the attention of: William F. Daniel
With a copy to: Alan Casey, A&L Goodbody, IFSC, Dublin 1.
to [The Management Representative]:
Address:
Fax:
For the attention of:
| 17 |
With a copy to:
to Nerano:
Address: 45 Fitzwilliam Square, Dublin 2
For the attention of: Seamus Mulligan
to the Company:
Address: McCann FitzGerald, Riverside One, Sir John Rogerson's Quay, Dublin 2
For the attention of: Seamus Mulligan
or to such other address or facsimile number as the addressee may have previously substituted by notice.
| 12.16.2. | A notice or other communication will be deemed to have been duly served or given: |
| (1) | in the case of delivery, at the time of delivery; |
| (2) | in the case of posting, 48 hours after posting (and proof that the envelope containing the notice or communication was properly addressed, prepaid and registered will be sufficient evidence that the notice or other communication has been duly served or given); or |
| (3) | in the case of facsimile, upon transmission, subject to the correct code or facsimile number being received on the transmission report |
but if a notice is given or served at business premises other than between 9.00 am and 5.30 pm on a Business Day, it will be deemed to be given or served on the next following Business Day.
| 12.16.3. | A party giving or serving a notice or other communication under this Agreement by facsimile shall also give or serve a copy by post, but without prejudice to the validity and effectiveness of the service by facsimile. |
| 12.16.4. | All notices or other communications shall be in the English language. |
| 12.17. | Counterparts: This Agreement may be executed in any number of counterparts, and by the several parties to it on separate counterparts, each of which when so executed will constitute an original but all of which together will evidence the same agreement. |
| 12.18. | No Partnership: Nothing in this Agreement is intended, or shall be deemed, to establish any partnership of any kind between the parties, constitute any party the agent of another party, or authorise any party to make or enter into any commitments for or on behalf of any other party. |
| 12.19. | Governing Law: Each of the Transaction Documents and all relationships created by them and arising out of or in connection with them, together with all Disputes, will in all respects be governed by and construed in accordance with Irish law. |
| 12.20. | Jurisdiction: |
| 12.20.1. |
| (1) | the Irish courts will have exclusive jurisdiction to hear, settle and/or decide any Dispute; |
| (2) | the parties agree that the Irish courts are the most appropriate and convenient courts to hear and decide any Dispute and therefore that they will not argue to the contrary. |
| 12.20.2. | The documents which initiate any Proceedings and any other documents required in relation to those Proceedings may be served: |
| 18 |
| (1) | according to the methods of service set out in clause 12.16; or |
| (2) | in any other manner allowed by law; |
and this clause 12.20.2 will apply wherever Proceedings are initiated.
| 19 |
SCHEDULE 1
Management Subscribers1
Names of Management Subscribers to be inserted when confirmed
| 20 |
SCHEDULE 2
The Company
|
Name
|
Speranza Therapeutics Limited
|
|
Date of Incorporation
|
13 May 2013 |
|
Country of Incorporation
|
Ireland |
|
Registered Number
|
527453 |
|
Authorised Share Capital
|
US$10,000 divided into 1,000,000 ordinary shares of US$0.01 each. |
|
Issued Share Capital prior
|
1 share issued at $0.01 |
|
Registered Office
|
Riverside One, Sir John Rogerson's Quay, Dublin 2 |
|
Principal Place of Business
|
Ireland |
|
Directors
|
Seamus Mulligan James Skehan
|
|
Secretary
|
James Skehan |
|
Financial Year End
|
l |
|
Last Accounts Filed
|
N/A |
|
Last Annual Return Filed
|
N/A |
|
Annual Return Date
|
13 November |
|
Auditors
|
l |
|
Charges and Debentures
|
None |
|
Location of Statutory Registers
|
At the registered office of the company |
| Tax Residence |
Ireland
|
| 21 |
SCHEDULE 3
Subsidiaries
|
Name
|
Speranza Biopharma Limited |
|
Date of Incorporation
|
17 August 2011 |
|
Country of Incorporation
|
Ireland |
|
Registered Number
|
502444 |
|
Authorised Share Capital
|
US$1,000,000 divided into 1,000,000 ordinary shares of US$1.00 each |
|
Issued Share Capital prior
|
100 shares of US$1.00 each |
|
Registered Office |
Treasury Building, Lower Grand Canal Street, Dublin 2
|
| Principal Place of Business |
Ireland
|
|
Directors
|
Seamus Mulligan James Skehan
|
|
Secretary
|
James Skehan |
|
Financial Year End
|
31 December |
|
Last Accounts Filed
|
31 December 2011 |
|
Last Annual Return Filed
|
30 September 2012 |
|
Annual Return Date
|
30 September
|
|
Auditors
|
KPMG |
|
Charges and Debentures
|
None |
|
Location of Statutory Registers
|
At the registered office of the company |
| Tax Residence |
Ireland
|
| 22 |
|
Name
|
Speranza Therapeutics Corp. |
|
Date of Incorporation
|
May 8, 2013 |
|
Country of Incorporation
|
USA (Delaware) |
|
Registered Number
|
5332090 |
|
Registered Office
|
Registered office in state of domicile: 160 Greentree Drive, Suite 101
|
|
Principal Place of Business |
300 Technology Square Cambridge, MA 02139
|
|
Officers
|
[Seamus Mulligan] [ • ]
|
|
Financial Year End
|
31 December |
|
Last Accounts Filed
|
N/A |
|
Last Annual Return Filed
|
N/A |
|
Annual Return Date
|
N/A |
|
Auditors
|
KPMG |
|
Charges and Debentures
|
None |
|
Location of Statutory Registers |
300 Technology Square
|
| Tax Residence | Massachusetts is State in which principal place of business is located and upon qualification, will be subject to MA franchise tax; corporation will also qualify to do business in California and be subject to CA franchise tax; and is subject to franchise tax in Delaware (state of domicile) |
| 23 |
SCHEDULE 4
Warranties
| 1. | SHARES, GROUP COMPANIES AND ACCURACY OF INFORMATION |
| 1.1. | Group Structure: The contents of recitals A and of Schedules 2 and 3 are true and accurate in all respects and in particular (without limitation) Nerano is the legal and beneficial owners of the Shares, which are free from Encumbrances, and there is no agreement, arrangement or understanding to create or give any Encumbrance over or in respect of any of the Shares; |
| 1.2. | Rights to Share Capital: No person has the right to call for the allotment, issue, sale or transfer of any Share or loan capital of the Company under any option or other agreement or obligation (including conversion rights and rights of pre-emption) and there are no claims or Encumbrances on or over Shares of the Company (and there is no agreement, arrangement or understanding to create or give any Encumbrance over or in respect of any of the shares of the Company) and the Shares constitute the whole of the issued and allotted share capital of the Company. All rights and interests of every kind existing in respect of the shares of Group Companies are valid and enforceable by action or legal proceeding or otherwise. |
| 2. | TRADING HISTORY |
With the exceptions of any actions required to enter into the Transaction Documents, the Company has not traded.
| 24 |
SCHEDULE 5
Completion Obligations
| 1. | Shareholders Resolutions: Nerano undertakes with ES1 to cause resolutions of the Shareholders to be passed: |
| 1.1. | adopting the agreed form Articles in substitution for and to the exclusion of the existing articles of association of the Company; |
| 1.2. | altering the Memorandum to the agreed form Memorandum in substitution for and to the exclusion of the existing memorandum of association of the Company.2 |
| 2. | Board Meetings: Nerano undertakes to cause a board meeting of the Company to be held at which: |
| 2.1. | the Subscription Shares are allotted and issued in accordance with the terms of this Agreement and the name of ES1 is entered into the register of members as the registered holders of the Subscription Shares; and |
| 2.2. | the entering into of each of the Transaction Documents required by the Company to be entered into is approved. |
| 3. | Transaction Documents to be Executed: Each of the parties undertake with each other to cause each of the Transaction Documents to be entered into between all the relevant parties thereto. |
| 4. | Documents to be Delivered to the Subscriber: Each of the parties undertake with each other to cause the following documents to be delivered to the Subscriber: |
| 4.1. | certificates under the common seal of the Company in respect of all Subscription Shares together with certified copy extracts from each of the register of members showing the Subscribers as the registered holder the Subscription Shares; |
| 4.2. | original copies of each of the Transaction Documents; |
| 4.3. | a copy of the resolutions required to be passed pursuant to paragraph 1 of this Schedule, certified by the secretary of the Company; |
| 4.4. | a copy of the minutes of each of the board meetings required to be held pursuant to paragraph 2 of this Schedule, certified by the secretary of the Company. |
2 M&A to be agreed pre-completion
| 25 |
SCHEDULE 6
Board Composition and Governance
| 1. | Number of Directors: The parties shall procure that no persons are appointed to be a Director otherwise than in accordance with this Agreement and the parties further undertake not to appoint or permit any person to act as a de facto director of the Company. |
| 2. | Appointment of Directors: |
| 2.1. | For so long as Nerano holds not less than 50 per cent. of the issued Shares, it shall be entitled: |
| 2.1.1. | to appoint such number of persons as Directors as it may determine in its absolute discretion (each, a Nerano Director), subject to any limit on the number of Directors imposed by the Articles; and |
| 2.1.2. | remove from office any Director and any director of any member of the Group |
| 2.2. | For so long as Nerano holds Shares which constitute less than 50 per cent. of the issued Shares, it shall be entitled to appoint three persons as Directors (each, a Nerano Director) and to remove any person so appointed. |
| 2.3. | Any appointment or removal pursuant to this clause shall be made by notice in writing served on the Company. |
| 2.4. | Notwithstanding any provision of the Articles, each Nerano Director and person appointed to the board of directors of a member of the Group by Nerano shall be entitled to appoint any person to be an alternate director, shall not be required to hold any share qualification, shall not be subject to retirement by rotation and, save as permitted or required by the Articles, shall not be removed except by the Nerano. |
| 2.5. | Immediately upon Nerano nominating a person for appointment or requesting the removal of a Director pursuant to paragraph 2.1.2 or paragraph 2.2, the Company and the other Shareholders shall join with Nerano in executing all such documents and doing all such acts and things as shall be necessary or desirable to forthwith cause such person to be so appointed or removed as the case may be. |
| 2.6. | The Shareholders acknowledge and agree that on and from Completion, the Directors of the Company shall be: |
Seamus Mulligan (who shall also be the Chairman); and James Skehan.
| 3. | Board Voting: |
| 3.1. | Resolutions of the Board and any committee shall be decided by a simple majority of the votes cast by the Directors (or their duly appointed alternates) present at the meeting of the Board or committee (as the case may be) for or against the resolution, unless otherwise specified under the terms of this Agreement. |
| 3.2. | Each Director shall have one vote. In the event of an equality of votes on any resolution proposed at a properly convened meeting of the Board, the Chairman (if any) shall have a second or casting vote. |
| 4. | Notice of Board Meetings: |
| 4.1. | Reasonable notice shall be given to each of the Directors of all meetings of the Board. |
| 4.2. | A meeting of the Board may be held at shorter notice than set out in this paragraph, or without notice, with the unanimous consent of the Directors. |
| 4.3. | Notice of each Board meeting shall contain, inter alia, an agenda specifying in reasonable detail the matters to be discussed at the relevant meeting, shall be accompanied by any relevant papers for discussion at such meeting (including the draft minutes of the previous meeting) and may be sent by post, courier, facsimile or other electronic communication. |
| 26 |
| 5. | Frequency and Location of Board Meetings: |
Board meetings shall be held at regular intervals as agreed between the Directors. Meetings of the Board and any committee of the Board shall be held at the location determined by the Board from time to time.
| 6. | Quorum at Meetings: No business shall be transacted at a meeting of the Board or of a committee of the Board unless at the time there is a quorum of two Directors present. |
| 7. | Minutes of Meetings: Drafts of the minutes of every meeting of the Board and of any committee of the Board shall be sent to each of the Directors within a reasonable time of the meeting (being not more than 10 Business Days after the meeting. |
| 8. | Alternate Directors: |
| 8.1. | Any Director (other than an alternate Director) shall be entitled, at any time and from time to time by notice in writing to the Company, to appoint any person (including any other Director) to be his alternate to attend and vote at meetings of the Board in his place, on such terms and subject to such conditions as shall be set out in the appointment, and to remove from office any alternate so appointed and to appoint another in his place, provided that only one such alternate may be appointed at any time. |
| 8.2. | An alternate Director shall cease to be an alternate Director if his appointor ceases for any reason to be a Director. |
| 8.3. | An alternate Director shall be entitled to receive notices of all meetings of Directors, and to attend, to be counted in the quorum for and to vote as a Director at any such meeting at which the Director appointing him is not personally present and generally to perform all functions of his appointor as a Director in the absence of such appointor including, without prejudice to the generality of the foregoing, power to sign any written resolution. |
| 8.4. | For the avoidance of doubt, the appointment of an alternate will not require to be approved by the Board. |
| 9. | Chairman: |
| 9.1. | The Directors, acting by simple majority, shall appoint any one of the Directors to be the Chairman of the Board and shall be entitled, at any time and from time to time, to remove from office as Chairman any Director so appointed and to appoint another in his place. |
| 9.2. | The Chairman will preside as chairman at every meeting of the Board, or, if there is no such chairman or if he is not present, any Director (or his alternate) present shall, with the agreement of a simple majority of the Directors present, act as chairman. |
| 10. | Information may be passed to Shareholders: Notwithstanding the provisions of clause 12.6 (Confidentiality) of this Agreement, each Director and any alternate of such Director shall be entitled to disclose to the Shareholder or Shareholders responsible for nominating him, and to any member of such Shareholder’s Group any information concerning the Company or the business of the Company which may come into his possession as Director or alternate. Each such Shareholder shall be entitled to disclose to any other member of such Shareholder’s Group any information concerning the Company or the business of the Company which may come into his possession as Shareholder or from any such Director or alternate, and to disclose any such information to the extent such disclosure is required by law or by any regulatory body to which such Shareholder is subject PROVIDED ALWAYS that such Shareholder’s Group shall use its best endeavours to procure that any third party shall first enter into a written agreement with the Company agreeing to be bound by the confidentiality provisions of clause 12.6 (Confidentiality) of this Agreement with regard to any Confidential Information it receives as if such third party were a party to this Agreement. |
| 11. | Board Expenses: The Company shall reimburse all reasonable and vouched out-of-pocket expenses properly incurred by all members of the Board (including any duly appointed alternates) as members of the Board (and not in any other capacity) in connection with the business of the Company. |
| 12. | Conflicts of interest: |
| 27 |
Each of the parties hereto agrees and declares that any Director shall, for so long as he shall hold the office of director of the Company, be entitled, in the performance of his duties as director:-
| 12.1. | to represent the views of the Shareholder responsible for nominating him as Director at all meetings of the Board or any committee thereof; and |
| 12.2. | provided that this will not conflict to a material extent with his fiduciary duties to the Company, to take into account the interests of, and any instructions given to him by or on behalf of, such nominating Shareholder; |
and any such representations or actions on the part of such Director shall be deemed not to constitute any breach of duty, breach of trust or other conflict of interest on the part of the Director.
| 28 |
SCHEDULE 7
Information and Access Rights
| 1. | Accounts and Annual Budget: The Company shall prepare and furnish at its own cost to the Board and to each Investor: |
| 1.1. | not later than 30 days before the beginning of each accounting period of the Company, the Annual Budget to be agreed by the Board, setting out the revenue, expenditure and budget forecasts of the Company for the next accounting period. |
| 1.2. | as soon as reasonably practicable and in any event within 90 days after the end of the accounting period to which such audited accounts relate, the audited profit and loss account and balance sheet of the Company (and, where prepared, the audited consolidated profit and loss account and balance sheet of the Company) in respect of each accounting period, together with the relative audit and management letters and all material correspondence between the Company and the Auditors concerning the accounts; |
| 1.3. | within 30 days of the end of the relevant quarter to which they relate, the unaudited quarterly management accounts of the Company comprising, in addition to any further information or details which the Board may from time to time reasonably require, a profit and loss account, balance sheet and cash flow statement, together with a comparison, including commentary, of actual performance of the Company against its budget and against its performance during the equivalent period of the previous year; |
| 2. | Additional Information: Without prejudice to the foregoing provisions of this Schedule, the Company shall keep each Investor fully informed in writing in reasonable detail, as soon as reasonably practicable, of all Material matters of which it has become aware relating to the Business (including, without limitation, any actual or potential developments, events or circumstances, adverse or otherwise, effecting the whole or any substantial part of the Business). |
| 3. | Inspection Rights: The Company shall provide each Investor or its duly authorised representatives or agents with access at all reasonable times to all accounting records, company books, documents, memoranda and information (including any records, company books, documents, memoranda and information held in electronic form or in storage media) reasonably required by it and shall cooperate with each Shareholder and its duly authorised representatives or agents and furnish same with reasonable explanations of any matters contained within or arising from an inspection of any such records, company books, documents, memoranda and information. |
| 4. | Information may be passed to an Investor’s Shareholder Group: Each Investor shall be entitled to pass to any member of such Investor’s Shareholder Group any information concerning the Group which may come into its possession pursuant to the provisions of this Schedule or via any Director or alternate pursuant to the provisions of Schedule 6 (Board Composition and Governance), and to disclose any such information to the extent such disclosure is required by law or by any regulatory body to which such Investor is subject. |
| 29 |
SCHEDULE 8
Shareholder Covenants
| 1. | Compliance with this Agreement: Each of the parties hereto shall exercise all voting rights and powers of control available to them so as to at all times comply with and, insofar as it is within their capacity to do so, give full effect to the terms and conditions of this Agreement. |
| 2. | Compliance with Memorandum and Articles: Each of the parties hereto shall exercise all voting rights and powers of control available to them so as to comply with and give full effect to (and to ensure that the Company, complies strictly at all times with) the restrictions imposed by the Memorandum and Articles of the Company. |
| 3. | Compliance with Laws: The Company shall at all times comply with, and conduct its business in accordance with, all applicable laws, regulations, permits, licences, approvals, authorisations, clearances or consents of or by any government as revenue, judicial, or regulatory agency or authority and shall ensure that all approvals, consents and licences as may from time to time be necessary for the purpose of carrying on the business of the Company are obtained and maintained in full force and effect. |
| 4. | Business Plan: |
| 4.1. | Subject to paragraph 4.2, the Group shall carry on the Business strictly in accordance with the Business Plan which will be agreed by Nerano and ES1, each acting reasonably and in good faith, within 90 days of Completion, and shall continue in effect until the end of 2015. |
| 4.2. | The terms of the Business Plan shall be in accordance with the provisions of the Clinical Plan. Until the Business Plan is agreed, the Business shall be carried out in accordance with the Clinical Plan. |
| 4.3. | Notwithstanding any other provision of this Agreement, save with the consent of each Investor, the Business Plan shall not be amended, updated, varied, revised or replaced in whole or in part in a manner which would have a negative impact on ES1 until such amendment, update, variation, revision or replacement is approved in accordance with paragraph 7 of Schedule 9 (Reserved Matters). |
| 4.4. | In the event of any Dispute with regard to any part of, or in respect of any proposals or provisions contained in, the Business Plan at any time, each Shareholder shall (to the extent possible) procure that the Directors act in good faith and shall use its respective reasonable endeavours so as to resolve any such Dispute, and until such time as any such revised Business Plan is agreed, the provisions of the existing Business Plan shall prevail. |
| 5. | General Business Covenants: The Group shall as and from Completion carry on the Business in a proper, efficient and business-like manner in material accordance with the Business Plan and the Annual Budget and in accordance with principles of good corporate governance and for the benefit of the Group so as to generate the maximum achievable profit, and shall ensure that any expansion or alteration in the Business is carried on only by the Group and/or a wholly-owned subsidiary of a Group Company. Each party hereto shall procure that all third parties directly or indirectly under its control refrain from acting in a manner which will hinder or prevent the Group from carrying on the Business in a proper, efficient and businesslike manner, and shall generally use its reasonable endeavours to promote the Business and the interests of the Group. |
| 6. | Financial Records: The Company shall maintain proper, usual and up-to-date financial and accounting records in relation to its business and affairs and shall otherwise comply in all material respects with all relevant standard accounting practices and financial reporting standards. |
| 7. | Annual Budget: No Annual Budget shall be implemented in whole or in part until approved by the Board. In the event of any Dispute with regard to any part of, or in respect of any proposals or provisions contained in, any Annual Budget, the Board shall resolve such Dispute, and until such time as any such Annual Budget is agreed, the provisions of the last quarter of the preceding agreed Annual Budget for the year in question shall prevail (indexed by reference to the previous quarterly period of inflation for each quarter). |
| 8. | Internal Systems: The Company shall maintain proper and effective reporting systems, collation |
| 30 |
systems and other management, financial, banking, and information systems, practices and procedures.
| 9. | Insurance: The Company (or another Group Company) shall take out and maintain such insurance policies as may be determined by the Board from time to time. |
| 10. | Keyman Life Insurance: The Company (or another Group Company) shall take out and maintain in force keyman life insurance on such persons, and for such period, as may be determined by the Board from time to time. |
| 11. | New Companies Bill: Each of the Shareholders shall (and shall procure that each Group Company shall) (with respect to each Group Company, its corporate status, constitutional documents, share capital, ownership, management and control and all related matters) execute and deliver all deeds, instruments and documents and do all such acts and things as may be required following the commencement of the proposed legislation to consolidate and reform Irish company law, currently known as the Companies Bill, and/or any secondary legislation arising pursuant to it, in order to continue to give full effect to the provisions this Agreement , the Memorandum and Articles, and the memorandum and articles of association for the time being of each Group Company, including if the Company is redesignated as a Designated Activity Company. |
| 12. | Auditors: The Auditors shall be instructed to report at the expense of the Company as to the amount of the profits available for distribution by the Company for each accounting period at the same time as they sign their report on the relevant audited accounts of the Company and to certify the amount of all Emoluments paid or made to the Directors and to connected persons of the Directors during that accounting period, and generally to prepare such accounts, reports and calculations as may be required by the Articles or by this Agreement from time to time. The Auditors shall be given such assistance and information by the Company as they may reasonably require in connection with the performance of any duties imposed upon them hereunder or by the Articles or under the Companies Acts. |
| 13. | Negative Pledge: No Shareholder shall pledge, mortgage (whether by way of fixed or floating charge) or otherwise create (or allow or permit to be created) any Encumbrance over his or its legal or beneficial interest in any of the Shares or Interest in Shares, or sell, transfer or dispose of any of same or contract to do so, otherwise than in accordance with this Agreement or the Articles. |
| 14. | Employment Agreements: Save as agreed by each Investor, the Company shall procure that at Completion all of the Group's employees, including the Management Subscribers, will enter into agreed form Employment Agreements. |
| 15. | General Meetings: |
| 15.1. | Notwithstanding any provisions in the Companies Acts, at least 21 days' notice in writing shall be given to each Registered Shareholder and each Director of each annual general meeting of the Company and of any other general meeting of the Company, unless each of the Registered Shareholders approve a shorter notice period. Any notice shall include an agenda identifying in reasonable detail the matters to be discussed at the meeting together with copies of any relevant papers. If any matter is not identified in reasonable detail, the Registered Shareholders shall not vote upon it, unless all of the Registered Shareholders agree to the matter in writing. |
| 15.2. | The Chairman, or, if the Chairman is not present, any Registered Shareholder or duly appointed representative or proxy of a Registered Shareholder who is present, shall preside as chairman at every general meeting of the Company. |
| 15.3. | Registered Shareholders holding together more than 50% of the issued share capital of the Company must be present (in person or by proxy, or by a duly appointed representative, as the case may be) in order to form a quorum at a general meeting of the Company. |
| 31 |
SCHEDULE 9
Reserved Matters
Matters requiring the Prior Written Consent of ES1
| 1. | Material Change to Business Purpose: Any Material change to the business purpose as contemplated in the Business Plan; |
| 2. | Disposal: The transfer, sale or disposal by any other means of the whole or a substantial part of the business, undertaking or assets, or of the shares in, any Group Company (or any interest therein), or the entering into of any contract, agreement or understanding (whether legally binding or not) so to do; |
| 3. | Alteration of Share Capital: An alteration of Share capital by the Company which would negatively impact on ES1. |
| 4. | Alteration of Constitutional Documents: To the extent it would have a negative impact on ES1, the making of any alteration to the Memorandum or Articles or the passing or adoption of any further regulations, resolutions, agreements or arrangements inconsistent with any such documents or with this Agreement; |
| 5. | Winding Up: Prior to the Investors agreeing, or an Industry Expert determining, that a Termination Event has occurred, the passing of any resolution for the winding up of any Group Company or the doing or permitting or suffering to be done any act or thing whereby the Group Company may be wound up (whether voluntarily or compulsorily); |
| 6. | Initial Funding: Expenditure of the Initial Funding by the Company on any matter which is not either (a) directly connected to the development of the D5 Business or (b) otherwise contemplated by the Business Plan; |
| 7. | Material Change to the Business Plan: Any Material change to the Business Plan which would negatively impact on ES1. |
| 8. | New Business: Save where the Company or another Shareholder(s) has offered to (re-)purchase the Shares then held by ES1 for Fair Value (as such terms is defined in Schedule 10 to this Agreement), which offer has been refused by ES1, the acquisition, development or commercialisation of other pharmaceutical assets outside of the D5 Business save as may be contemplated by the Business Plan; |
| 9. | Distribution: The declaration or distribution of any dividend or other payment out of the distributable profits by any Group Company until such time as the Initial Funding is expended; and |
| 10. | Non-Ordinary Course Contracts: Save for the Executive Management Agreement or as contemplated in the Business Plan, the making of any contract outside the ordinary course of trading or otherwise than at arm's length terms by any Group Company; |
| 32 |
SCHEDULE 10
Transfer of Shares
| 1. | Restriction on Sale: |
| 1.1. | Notwithstanding anything else in this Agreement but subject to paragraph 4 of this Schedule: |
| (a) | save with the prior written consent of each Investor, no Investor may dispose of their Shares in any way whatsoever until the later of the end of 2015 or when the Initial Funding is expended; and |
| (b) | save with the consent of the Board or as may be required pursuant to this Agreement or the Articles, the Management Subscribers are not permitted to dispose of their Shares in any way whatsoever until the occurrence of a Realisation. |
| 1.2. | If the Board gives its consent pursuant to paragraph 1.1(b) of this Schedule to a disposal of Shares by a Management Subscriber, it may require as a condition of such consent that such Shares be transferred to Nerano and held as Unallocated Management Shares. If the Board so elects, the provisions of paragraph 3 of this Schedule shall not apply to a transfer by a Management Subscriber. |
| 2. | Compulsory transfers by Management Subscribers:3 |
| 2.1. | If a person becomes a Departing Employee then at any time after that person’s Cessation Date, the Board may serve a written notice on that person (a Compulsory Transfer Notice) requiring that all of the Shares held by each member of that person’s Departing Employee’s Group be offered for sale in accordance with this paragraph 2. Upon service of a Compulsory Transfer Notice, each member of that person’s Departing Employee’s Group who holds Shares (each a Compulsory Seller) shall be deemed to have appointed the Company as his agent for the sale of all of their Shares (together, the Compulsory Sale Shares) in accordance with this paragraph 2. |
| 2.2. | The price at which each Compulsory Sale Share may be required to be transferred pursuant to this paragraph 2 shall be determined as follows:- |
| 2.2.1. | if the reason the Departing Employee became a Departing Employee is |
| (1) | his death; or |
| (2) | his ill health or permanent disability, |
or if the Board so determines, the price shall be the higher of the Issue Price and the Fair Value at the Cessation Date; and
| 2.2.2. | if the reason the Departing Employee became a Departing Employee is not for one of the reasons set out in paragraph 2.2.1 (and the Board does not make the determination referred to therein), the price shall be the lower of the Issue Price and the Fair Value at the Cessation Date. |
| 2.3. | Upon service of a Compulsory Transfer Notice, the Board shall be entitled to offer the Compulsory Sale Shares at the price determined pursuant to paragraph 2.2 to any of the following:- |
| 2.3.1. | any full-time employee of the Group or any person who is, or is to be, offered such employment (in which latter case, the acceptance of any such invitation shall be conditional on him becoming an employee); or |
| 2.3.2. | Nerano, in which case, following transfer, the Compulsory Sale Shares shall constitute Unallocated Employee Shares and shall be dealt with in accordance with clause 6.5. |
| 2.4. | The Company and each Compulsory Seller shall use their best endeavours to ensure that, after any offer referred to in paragraph 2.3 is accepted, the completion of the transfer of the relevant Compulsory Sale Shares occurs as soon as reasonably practicable. If any Compulsory Seller fails to transfer any |
3 Transfer provisions to be replicated in the Articles.
| 33 |
Compulsory Sale Shares within two Business Days of being notified of acceptance of an offer for such Compulsory Sale Shares pursuant to paragraph 2.3, the provisions of paragraph 3.7 (references therein to the Proposing Transferor, Sale Shares, Sale Price and Proposing Transferee being read as references to the Compulsory Seller making such default, the Compulsory Sale Shares in respect of which such default is made, the price determined in accordance with paragraph 2.2 and the person who has accepted the offer made pursuant to paragraph 2.3 respectively) shall apply to the transfer of such Compulsory Shares mutatis mutandis.
| 2.5. | As from the date a Compulsory Transfer Notice is deemed to have been given pursuant to and in accordance with paragraph 2.1 until such time as the Compulsory Transfer Shares have been transferred in accordance with paragraphs 2.3 and 2.4, such Compulsory Transfer Shares shall cease to entitle the holder thereof (or any proxy) to any voting, capital or dividend rights otherwise attaching to such Compulsory Transfer Shares. |
| 2.6. | The requirements of this paragraph 2 may be waived in respect of any Departing Employee in whole or in part, and whether with or without any conditions, by the Board. |
| 2.7. | For the avoidance of doubt, the provisions of paragraph 3 and paragraph 5 shall not apply in respect of any transfer pursuant to this paragraph 2. |
| 2.8. | For the purposes of this paragraph 2 |
Cessation Date means, in relation to a person who becomes a Departing Employee:-
| (a) | where the person becomes a Departing Employee by virtue of notice given by that person’s employer, the date on which such notice expires; |
| (b) | where the person’s contract of employment is terminated by that person’s employer and a payment is made in lieu of notice, the date on which this payment is made; |
| (c) | in any other case, the date on which the person’s contract of employment or services is terminated; |
Departing Employee means:-
| (a) | any employee or director of the Company or any subsidiary of the Company who ceases to be an employee or director of the Company or any subsidiary of the Company; or |
| (b) | any individual who is not an employee or director of the Company or any subsidiary of the Company but whose services are otherwise provided to the Company or any subsidiary of the Company whose services cease to be provided to the Company or any subsidiary of the Company, |
provided that in no circumstances shall Seamus Mulligan be a Departing Employee;
Departing Employee Group means in relation to any Departing Employee:-
| (a) | the Departing Employee, in respect of all Shares then held by him; |
| (b) | any person (or their nominee) (a Departing Employee’s Associate) who, at or before the time of the allotment, issue, transfer or other acquisition of Shares to or by the Departing Employee’s Associate, is designated as the holder of shares in the Company on behalf of, in trust for, or in the place of, the Departing Employee; |
Fair Value means, in relation to any Shares which are to be transferred by a Compulsory Seller, either:-
| (a) | the amount, if any, proposed by the Board, if accepted by the Compulsory Seller; or |
| (b) | if no value is proposed by the Board, or if the value proposed by the Board is not accepted by the Compulsory Seller within 14 days of the giving or deemed giving of the relevant Transfer Notice, the fair value of the Shares as determined by the Valuer pursuant to paragraph 7 |
Issue Price means, in respect of a Share, the aggregate of the amount paid up (or credited as paid up)
| 34 |
in respect of the nominal value thereof and any share premium thereon; and
Valuer means an independent investment bank, accounting firm or corporate finance firm appointed by the Board for the purpose of valuing Shares in accordance with paragraph 7.
| 3. | Offer-round on Voluntary Transfer: |
| 3.1. | Subject always to the provisions of paragraph 1 (Restriction on Sale), paragraph 2 (Compulsory Transfer by Management Subscribers), paragraph 4 (Permitted Transfers) and paragraph 5 (Tag-Along Rights) of this Schedule and to clause 6.2 of this Agreement, a Shareholder (the Proposing Transferor) desiring to transfer any Share or Shares or any Interest in Shares (together, for the purposes of this paragraph 3, the Sale Shares, and each a Sale Share) may, if permitted to transfer Shares pursuant to paragraph 1, give notice in writing to the Company (the Transfer Notice) of his desire to transfer the Sale Shares. The Transfer Notice shall state the number of Sale Shares and the sale price per Sale Share of the Sale Shares as fixed by the Proposing Transferor (which, for the avoidance of doubt, must be in cash) (the Sale Price). The effect of the service of the Transfer Notice shall be to appoint the Company as agent for and on behalf of the Proposing Transferor for the sale of the Sale Shares. |
| 3.2. | Except in the case of a Transfer Notice deemed to have been served pursuant to paragraph 2 (Compulsory Transfers) by Compulsory Sellers (and subject always to clause 6.2 of this Agreement), the Proposing Transferor may specify in the Transfer Notice that he is willing to transfer all, but not some only, of the Sale Shares, in which case the Proposing Transferor will not be obliged to transfer any Sale Shares to the Registered Shareholders pursuant to this paragraph 3 unless offers are received for all Sale Shares. |
| 3.3. | Within 5 Business Days of receipt of the Transfer Notice by the Company, the Company shall, by notice in writing to all Registered Shareholders (the Company’s Notice), offer for sale the Sale Shares at the Sale Price and on the following terms, each of which terms shall be stated in the Company’s Notice: |
| 3.3.1. | The Sale Shares shall be transferred free from all Encumbrances and together with all rights, title and interest in or attaching to them, including any right or entitlement howsoever described (whether absolute or conditional) to receive (or to direct the payment or receipt of) any dividends or other distributions (including any dividends or distributions declared or made after the date of the Transfer Notice, or declared before but paid after such date), any right or entitlement to control the voting or other rights attributable to any such Sale Share, and any option over (and/or right to subscribe for) any such Sale Share. |
| 3.3.2. | Each of the Registered Shareholders (with the exception of the Proposing Transferor) shall be entitled to buy such proportion of Sale Shares as equals, as nearly as possible, the proportion of the total number of Shares (excluding the Sale Shares) held by him at the date of the Transfer Notice, and a Registered Shareholder shall be entitled to buy fewer Sale Shares than his proportional entitlement. |
| 3.3.3. | A Registered Shareholder may offer to buy a specified number of additional Sale Shares (his Excess Sale Shares) in the event that (as a result of one or more Registered Shareholders having chosen to decline the offer of their full proportionate entitlement of Sale Shares pursuant to paragraph 3.3.2) some of the Sale Shares remain unallocated (the Unallocated Sale Shares) following the operation of paragraph 3.3.2. |
| 3.3.4. | If the Company receives offers for a number of Shares which in aggregate exceeds the number of Sale Shares, each Registered Shareholder who offered to buy Excess Sale Shares shall be deemed (so far as practicable and without exceeding the total number of Shares which each such Registered Shareholder shall have offered to purchase) to have offered to purchase such proportion of the Unallocated Sale Shares as equals, as nearly as possible, the proportion which his Excess Sale Shares bears to the total number of Excess Sale Shares for which offers were received by the Company. |
| 3.3.5. | Any offer by a Registered Shareholder to buy some or all of the Sale Shares (including, for the avoidance of doubt, any Excess Sale Shares) shall be made in writing to the Company (each, a Purchase Notice) within 15 Business Days of the date of service of the Company’s Notice (the last day of such period being the Closing Date), failing which a Shareholder shall be deemed to have declined the offer. |
| 35 |
| 3.3.6. | Any fractional entitlements which might arise regarding the apportionment of the Sale Shares as between the Registered Shareholders shall be determined by the Board, acting reasonably. |
| 3.3.7. | On the Closing Date, the Transfer Notice and each Purchase Notice shall each become irrevocable. |
| 3.4. | Within 5 Business Days following the Closing Date, the Company shall notify in writing: |
| 3.4.1. | the Proposing Transferor of the Purchase Notices (if any) received and of the names and addresses of the Registered Shareholder or Shareholders who offered to buy Sale Shares (together, the Proposing Transferees) and, if any Sale Shares are to be sold, of the number of Sale Shares which are to be acquired by each Proposing Transferee; |
| 3.4.2. | each Proposing Transferee of the number of Sale Shares he is to acquire, or, if no Sale Shares are to be sold, of that fact; |
specifying in each such notice a place and time, between 5 and 10 Business Days after the date of such notice, on which the sale and purchase of the Sale Shares is to be completed (Sale Share Completion Date). Where any Regulatory Approval or any approval of the shareholders of the Proposing Transferor or any of the Proposing Transferees is required in respect of the sale and purchase of the Sale Shares (or any of them), the Sale Share Completion Date shall be the later of the date specified by the Company in such notice and the date upon which all such shareholder approvals and/or Regulatory Approvals have been obtained.
| 3.5. | Subject always to paragraph 3.2, upon the Sale Share Completion Date, the Proposing Transferor shall be obliged to transfer such Sale Shares, and to deliver such documents as are required to give effect to such transfer upon receipt of the Sale Price for each such Share, free from Encumbrances and together with all rights, title and interest attaching to them (including any right or entitlement howsoever described, whether absolute or conditional, to receive, or to direct the payment or receipt of, any dividends or other distributions (including any dividends or distributions declared or made after the date of the Transfer Notice, or declared before but paid after such date), any right or entitlement to control the voting or other rights attributable to any such Share, and any option over (and/or right to subscribe for) any such Share), and each such Proposing Transferee shall be obliged to acquire such Sale Shares and to pay the Sale Price for such Sale Shares to the Proposing Transferor’s solicitors by way of electronic funds transfer to a bank account nominated by the Proposing Transferor’s solicitors or in such other manner as may be agreed in writing between the parties, and the receipt of the Proposing Transferor’s solicitors will be an absolute discharge to such Proposing Transferee. |
| 3.6. | If, by the Closing Date, the Company has not received offers for all the Sale Shares, the Proposing Transferor may, within 30 Business Days following the date of the notice by the Company pursuant to paragraph 3.4.1, transfer the Sale Shares for which offers were not received (or, if the Transfer Notice stated that the Proposing Transferor was only willing to transfer to the Registered Shareholders all the Sale Shares, all the Sale Shares (except in the case of a Transfer Notice deemed to have been served pursuant to paragraph 2 (Compulsory Transfers) of this Schedule)) to any person or persons at no less than the Sale Price and otherwise on terms no more favourable to such person than those specified in the Transfer Notice, and the Board shall not be entitled to decline or refuse to register any such transfer or transfers, PROVIDED ALWAYS THAT: |
| 3.6.1. | such person or persons shall first enter into a Deed of Adherence (as soon as possible and in any event not later than the date of registration of the transfer); |
| 3.6.2. | the Board may refuse to register any transfer of Shares to a person or persons it reasonably considers (acting bona fide and in the best interests of the Company) to be in competition with, or who it reasonably believes intends to set up in competition with, the Business, or to any person or persons connected with such a person or persons (or to a nominee or nominees of either); |
| 3.6.3. | if the Proposing Transferor stipulated (being so entitled under the terms of this Agreement) in the Transfer Notice that he was only willing to transfer all the Sale Shares, the Proposing Transferor shall not be entitled to sell only some of the Sale Shares to such person or persons; |
| 3.6.4. | the Board shall refuse to register any such transfer of Shares if such transfer obliges the |
| 36 |
Proposing Transferor to procure the making of an offer pursuant to paragraph 5 (Tag Along Rights), until such offer has been made and completed in accordance with the provisions of such paragraph, unless failure to complete is otherwise than as a result of the default of the Proposing Transferor;
| 3.6.5. | the Board may require to be reasonably satisfied that those shares are being transferred under a bona fide sale for an amount equal to the Sale Price without any deduction, rebate or allowance to the purchaser and, if not so satisfied, may refuse to register the transfer (and for the avoidance of doubt the Board may require such information as it reasonably requests in order to value any non-cash consideration). |
| 3.7. | If the Proposing Transferor fails to transfer any Sale Shares in accordance with this paragraph 3, the Board may (and shall if so requested by any Proposing Transferee) authorise any Director to execute, complete and deliver as agent and attorney for and on behalf of the Proposing Transferor a transfer of the Sale Shares to each of the relevant Proposing Transferees against receipt by the Company of the Sale Price due from the Proposing Transferees concerned. The Company shall hold such sums in trust for the Proposing Transferor without any obligation to pay interest. The Company's receipt of the Sale Price due from a Proposing Transferee in respect of the Sale Shares to be acquired by him shall be a good discharge to the relevant Proposing Transferee. The Directors shall then authorise registration of the transfer. The Proposing Transferor shall in any event be obliged to deliver the certificate for the Sale Shares to be transferred by him to the Company (or, where appropriate, provide an indemnity in respect thereof in a form satisfactory to the Board) whereupon he shall be entitled to the aggregate Sale Price for the relevant Sale Shares, without interest. If such certificate relates also to any Share or Shares which the Proposing Transferor has not become bound to transfer in accordance with this paragraph 3, the Company shall issue to the Proposing Transferor a new certificate for such Share or Shares. In order to secure each Shareholder's obligations under this paragraph, each Shareholder hereby appoints the Company to act as his attorney with authority in the Shareholder's name and on his behalf to execute and sign any and all agreements, instruments, deeds or other papers and documents and to do all things in his name as the Company may in its absolute discretion consider necessary to give effect to this paragraph (but no other) and the Company shall be entitled to delegate the exercise of such authority to any Director or the secretary of the Company from time to time, provided that such delegate shall not be authorised to delegate such authority further. |
| 4. | Permitted Transfers: |
Notwithstanding any other provision of this Schedule:
| 4.1. | Any Shareholder, being a body corporate, shall be entitled to transfer any Share or Shares or any Interest in Shares (Shares for the purpose of the remainder of this paragraph 4) held by it to any Affiliate, without being obliged to first offer such Shares to the other Registered Shareholders in accordance with paragraph 3 (Offer-round on Voluntary Transfer) of this Schedule, PROVIDED THAT: |
| 4.1.1. | in any such event, any such transferee shall first enter into a Deed of Adherence (as soon as possible and in any event not later than the date of registration of the transfer); |
| 4.1.2. | on such transferee proposing to cease to be an Affiliate of the Shareholder, it shall (and the Shareholder who originally transferred the Shares hereby undertakes to procure that the transferee shall) first re-transfer all its interest in the Shares held by it or on its behalf to the original transferor under this paragraph or to another Affiliate of that original Shareholder or as otherwise may be agreed in writing by the other Shareholders. |
| 5. | Tag-Along Right: |
| 5.1. | Subject always to paragraph 1 (Restriction on Sale), paragraph 2 (Compulsory Transfers by Management Subscribers) and paragraph 4 (Permitted Transfers) of this Schedule and notwithstanding the provisions of paragraph 3 (Offer-round on Voluntary Transfer) of this Schedule, any Shareholder or Shareholders (together the Proposing Transferor), desiring to transfer to a proposed purchaser any Share or Shares or any Interest in Shares (the Sale Shares), may only do so (subject to being permitted to do so in accordance with paragraph 1) if, with respect to the Sale Shares, the Proposing Transferor observes and complies in full with the offer-round provisions set out in paragraph 3 (Offer-round on Voluntary Transfer) of this Schedule, as amended and supplemented by the following provisions of this paragraph 5. |
| 37 |
| 5.2. | The Transfer Notice served in accordance with paragraph 3.1 of this Schedule shall state the following additional information: |
| 5.2.1. | the identity of the proposed purchaser (the Buyer); |
| 5.2.2. | the price per Sale Share that the Buyer proposes to pay (for the purposes of this paragraph 5, the Sale Price); |
| 5.2.3. | the manner in which the consideration is to be paid; and |
| 5.2.4. | any other terms and conditions (including any warranties, representations or indemnities) applicable to the sale of the Sale Shares to the Buyer. |
| 5.3. | The terms upon which the Company shall offer for sale the Sale Shares at the Sale Price to the other Registered Shareholders in accordance with paragraph 3.3 of this Schedule shall include the following (which additional terms shall be stated in the Company’s Notice): |
| 5.3.1. | Where, following compliance with the provisions of paragraph 3 (Offer-round on Voluntary Transfer) of this Schedule in accordance with their terms, the Proposing Transferor is entitled, and proposes, to transfer, any or all Sale Shares to the Buyer in accordance with paragraph 3.6, then any other Registered Shareholders who wish to also offer some or all of their Shares for sale to the Buyer (together, the Tagging Shareholders) shall notify the Company in writing (the Tag Along Notice) prior to the Closing Date. The effect of the service of the Tag Along Notice shall be to appoint the Company as agent for and on behalf of the Tagging Shareholders for the sale of Shares to the Buyer pursuant to the provisions of this paragraph 5. Any other Registered Shareholder who does not serve a Tag Along Notice prior to the Closing Date shall be deemed to have specified that he does not wish to offer any of his Shares for sale to the Buyer. |
| 5.3.2. | Each Tagging Shareholder shall be entitled to offer for sale such proportion of his Shares as represents, as nearly as possible, the proportion which: |
| (1) | the total number of Sale Shares which may, following compliance with the provisions of paragraph 3, be transferred by the Proposing Transferor pursuant to and in accordance with paragraph 3.6; |
bears to:
| (2) | the total number of Shares then in issue; |
(in the case of each Tagging Shareholder, the Tagging Shares).
| 5.3.3. | The Tagging Shares shall be offered for sale to the Buyer on the same terms (including any warranties, representations or indemnities) and for the same Sale Price as the Sale Shares. |
| 5.3.4. | On the Closing Date, along with the Transfer Notice and each Purchase Notice, each Tag Along Notice, if any, shall become irrevocable. |
| 5.4. | Within 5 Business Days following the Closing Date, where some or all of the Sale Shares may, following compliance with the provisions of paragraph 3 of this Schedule, be transferred by the Proposing Transferor pursuant to and in accordance with paragraph 3.6 of this Schedule, the Company shall notify in writing (along with the other information required to be notified pursuant to paragraph 3.4 of this Schedule): |
| 5.4.1. | the Proposing Transferor of the Tag Along Notices (if any) received and of the names and addresses of each Tagging Shareholder and his respective number of Tagging Shares; |
| 5.4.2. | each Tagging Shareholder of the fact that an offer of his Tagging Shares is to be made to the Buyer. |
| 5.5. | In accordance with the provisions of paragraph 3.6, the Proposing Transferor may, within 60 Business Days following the date of the notice by the Company pursuant to paragraph 3.4.1, transfer to the Buyer the Sale Shares for which offers were not received (or, if the Transfer Notice stated that the Proposing |
| 38 |
Transferor was only willing to transfer all the Sale Shares to the Registered Shareholders, all the Sale Shares) LESS the aggregate number of Tagging Shares PROVIDED ALWAYS THAT he procures that the Buyer purchases from each Tagging Shareholder his respective Tagging Shares at the same time and on the same terms for the same Sale Price, and the Board shall not be entitled (subject to the provisions of paragraphs 3.6.1 to 3.6.5) to decline or refuse to register any such transfer or transfers. For the avoidance of doubt, the provisions of paragraph 3.7 of this Schedule shall apply in respect of the Proposing Transferor.
| 5.6. | Subject to the provisions of paragraphs 3.6.1 to 3.6.5, within 60 Business Days following the date of the notice by the Company pursuant to paragraph 3.4.1, each Tagging Shareholder shall transfer his Tagging Shares to the Buyer on the same terms as the Sale Shares and for the Sale Price, in accordance with this paragraph 5. If any Tagging Shareholder fails to transfer any Tagging Shares in accordance with this paragraph 5, the Board may (and shall if so requested by the Proposing Transferor or any other Tagging Shareholder) authorise any Director to execute, complete and deliver as agent and attorney for and on behalf of the Tagging Shareholder a transfer of the Tagging Shares to the Buyer against receipt by the Company of the Sale Price due from the Buyer. The Company shall hold such sums in trust for the Tagging Shareholder without any obligation to pay interest. The Company's receipt of the Sale Price due from the Buyer in respect of the Tagging Shares to be acquired by him shall be a good discharge to the Buyer. The Directors shall then authorise registration of the transfer. The Tagging Shareholder shall in any event be obliged to deliver the certificate for the Tagging Shares to be transferred by him to the Company (or, where appropriate, provide an indemnity in respect thereof in a form satisfactory to the Board) whereupon he shall be entitled to the aggregate Sale Price for the relevant Tagging Shares, without interest. If such certificate relates also to any Share or Shares which the Tagging Shareholder has not become bound to transfer in accordance with this paragraph 5, the Company shall issue to the Tagging Shareholder a new certificate for such Share or Shares. In order to secure each Shareholder's obligations under this paragraph, each Shareholder hereby appoints the Company to act as his attorney with authority in the Shareholder's name and on his behalf to execute and sign any and all agreements, instruments, deeds or other papers and documents and to do all things in his name as the Company may in its absolute discretion consider necessary to give effect to this paragraph (but no other) and the Company shall be entitled to delegate the exercise of such authority to any Director or the secretary of the Company from time to time, provided that such delegate shall not be authorised to delegate such authority further. |
| 6. | Drag Along Right: |
Notwithstanding any other provision of this Schedule or of this Agreement:
| 6.1. | In the event that a bona fide, arms’ length offer is made at any time to any Shareholder by any third party (being any person other than a Shareholder, or any Affiliate of any Shareholder, or any Person Acting in Concert with any Shareholder or with any Affiliate of any Shareholder) (the Third Party Buyer) for the entire issued share capital of the Company (a Third Party Offer) after the later of the end of 2015 or when the Initial Funding is expended, such Shareholder shall forthwith give notice in writing to each of the other Registered Shareholders (with a copy to the Company) of the Third Party Offer, the identity of the Third Party Buyer, full details of the terms (including any warranties, representations or indemnities sought) upon which the Third Party Offer is made together with copies of all documentation relating to the Third Party Offer, and the sale price per Share offered (the Sale Price). If (a) each of the Investors and (b) the registered holders of 85% or more of the Shares (the Consenting Shareholders) agree to accept the Third Party Offer, either unconditionally or subject to conditions acceptable to the Third Party Buyer, they together shall have the right but not the obligation (the Drag Along Right) to require the remaining Registered Shareholders (the Remaining Shareholders) to sell and transfer all of the Shares and Interests in Shares, held by them or by any person on their behalf (together the Remaining Shares) to the Third Party Buyer in accordance with, and on the terms and subject to the conditions set out in, this paragraph 6. |
| 6.2. | If the Consenting Shareholders decide to exercise the Drag Along Right, they must, within 5 Business Days of the notice referred to in paragraph 6.1, give notice in writing to each of the Remaining Shareholders (with a copy to the Company) (the Drag Along Notice), upon the service of which, each Remaining Shareholder shall be bound to sell their Remaining Shares to the Third Party Buyer on, and subject to, the following terms (unless otherwise agreed between the parties): |
| 6.2.1. | The sale of the Remaining Shares shall be conditional upon the contemporaneous (or earlier) completion of the transfer to the Third Party Buyer of all of the Shares and Interests in Shares held by the Consenting Shareholders (together, the Consenting Shareholders’ Shares). |
| 39 |
| 6.2.2. | The Remaining Shares shall be transferred at the same price, which shall be the Sale Price, and on the same terms (including as to any warranties, representations or indemnities) as the transfer of the Consenting Shareholders’ Shares, and otherwise free from all Encumbrances and together with all rights, title and interest attaching to them, including any right or entitlement howsoever described (whether absolute or conditional) to receive (or to direct the payment or receipt of) any dividends or other distributions (including any dividends or distributions declared or made after the date of the Drag Along Notice, or declared before but paid after such date), any right or entitlement to control the voting or other rights attributable to any such Share, and any option over (and/or right to subscribe for) any such Share. |
| 6.2.3. | Completion of the sale of the Remaining Shares shall take place on the date specified for that purpose by the Consenting Shareholders in the Drag Along Notice (the Share Sale Completion Date). Where any Regulatory Approval or any approval of the shareholders of any of the Consenting Shareholders or Remaining Shareholders is required in respect of the sale and purchase of the Consenting Shareholders’ Shares (or any of them) or the Remaining Shares (or any of them) respectively, the Sale Share Completion Date shall be the later of the date specified in the Drag Along Notice and the date upon which all such shareholder approvals and/or Regulatory Approvals have been obtained. Notwithstanding the foregoing, the Share Sale Completion Date shall in no event be later than 120 Business Days following the date of the Drag Along Notice. |
| 6.2.4. | The Drag Along Notice once served shall be irrevocable but shall (along with all obligations thereunder) lapse in the event that, for any reason, the transfer of any or all of Consenting Shareholders’ Shares to the Third Party Buyer does not complete within 120 Business Days of the date of the Drag Along Notice. |
| 6.2.5. | Upon the Sale Share Completion Date, each of the Remaining Shareholders shall be obliged to transfer their Remaining Shares on the terms set out in paragraph 6.2.2, and to deliver such documents as are required to give effect to such transfer, upon receipt of the Sale Price for each such Share. |
| 6.2.6. | If any Remaining Shareholder fails to transfer any Remaining Shares in accordance with this paragraph 6, the Board may (and shall if so requested by any Consenting Shareholder) authorise any Director to execute, complete and deliver as agent and attorney for and on behalf of the Remaining Shareholder a transfer of the Remaining Shares to the Third Party Buyer against receipt by the Company of the Sale Price due from the Third Party Buyer. The Company shall hold such sums in trust for the Remaining Shareholder without any obligation to pay interest. The Company's receipt of the Sale Price due from the Third Party Buyer in respect of such Remaining Shares shall be a good discharge to the Third Party Buyer. The Directors shall then authorise registration of the transfer. The Remaining Shareholder shall in any event be obliged to deliver the certificate for the Remaining Shares to be transferred by him to the Company (or, where appropriate, provide an indemnity in respect thereof in a form satisfactory to the Board) whereupon he shall be entitled to the aggregate Sale Price for the relevant Remaining Shares, without interest. In order to secure each Shareholder's obligations under this paragraph, each Shareholder hereby appoints each the Company and/or each other Shareholder to act as his attorney with authority in the Shareholder's name and on his behalf to execute and sign any and all agreements (including, without limitation, any share purchase agreement), instruments, deeds or other papers and documents and to do all things in his name as the Company may in its absolute discretion consider necessary to give effect to this paragraph 6 (but no other) and the Company shall be entitled to delegate the exercise of such authority to any Director or the secretary of the Company from time to time, provided that such delegate shall not be authorised to delegate such authority further. |
| 7. | VALUATION |
| 7.1. | If the Valuer is required to determine the price at which Shares are to be transferred pursuant to paragraph 2, such price shall be the amount the Valuer shall, on the application of the Board (which application shall be made as soon as practicable following the time it becomes apparent that a valuation pursuant to this paragraph 7 is required), certify in writing to be the price which, in its opinion, represents a fair value for such Shares as between a willing seller and a willing buyer as at the date the Compulsory Transfer Notice is given or deemed to be given. Any such determination shall not take any account of whether or not the Sale Shares comprise a majority or a minority interest in the Company or |
| 40 |
of the fact that the transfer of such Shares is restricted under these Articles or the Agreement.
| 7.2. | In so certifying, the Valuer shall act as an expert and not as arbitrator and its decision shall be conclusive and binding on the Company and upon all of its holders for the purposes of this Agreement. |
| 7.3. | The costs of the Valuer shall be borne by the Company |
| 7.4. | The Company shall:- |
| 7.4.1. | fully co-operate with the Valuer in its carrying out of a valuation pursuant to this paragraph 7; |
| 7.4.2. | comply with all reasonable requests of the Valuer; and |
| 7.4.3. | provide the Valuer with all information requested by the Valuer to enable it to carry out a valuation pursuant to this paragraph 7. |
| 41 |
SCHEDULE 11
Pre-emption on Issue of Shares
| 1. | Offer-round: Save as provided in the Articles in respect of bonus issues (and subject always to the provisions of Schedule 12 (Anti-Dilution)), no Shares in the capital of the Company shall be allotted and issued except as follows: |
| 1.1. | The Shares in the capital of the Company shall be offered for subscription to all the Registered Shareholders in proportion to the numbers of Shares held by them respectively for cash and in all respects on the same terms per share. |
| 1.2. | Such offers shall be made by notice to each Registered Shareholder stating the total number of Shares so offered, the number thereof offered to that Shareholder, the price per share and the date (being not less than 20 Business Days after the date upon which such notice is given) by which the offer, if not accepted, will be deemed to have been rejected. |
| 1.3. | Any Shares in respect of which such an offer to a Registered Shareholder is accepted shall be allotted and issued on the basis of such offer and the Registered Shareholder shall be obliged to subscribe accordingly. |
| 1.4. | If any offer is rejected or deemed to have been rejected, the Shares comprised therein shall be offered for subscription on the same terms to the Registered Shareholders who have accepted the offers made to them under paragraph 1.2 of this Schedule and so that, in the event of competition, their entitlements to subscribe will be proportionate to the numbers of Shares held by them respectively. Such further offers shall be made by notice to each such Registered Shareholder stating the total number of shares so offered, the number thereof offered to that Shareholder, and the date (being not less than 20 Business Days after the date upon which such notice is given) by which the offer, if not accepted, will be deemed to have been rejected. |
| 1.5. | To the extent that any shares so offered are still not taken up, following the expiration of the period specified in paragraph 1.4 of this Schedule, the Board may, within 60 Business Days, subject to the Shareholder approval required under clause 3 of the matters listed in Schedule 9 (Reserved Matters), allot such unallocated shares to such persons as the Board thinks proper PROVIDED ALWAYS that: |
| (1) | such shares shall be offered on terms no more favourable than such shares were first offered to the Registered Shareholders under this paragraph 1; and |
| (2) | the Board shall not offer any shares to a person or persons it reasonably considers (acting bona fide and in the best interests of the Company) to be in competition with, or who it reasonably believes intends to set up in completion with, the Business, or to any person or persons connected with such a person or persons (or to a nominee or nominees of either), or to any person or persons the admission to membership of whom would, in the opinion of a majority of the Directors bona fide held by them, be damaging to the business interests of the Group. |
| 1.6. | Any fractional entitlements and other practical difficulties which might arise regarding the apportionment of any such shares shall be determined by the Board in its sole discretion. |
| 2. | Deed of Adherence: It shall be a condition of any allotment of Shares pursuant to this Schedule that the allottee (if not already bound by the terms of this Agreement) executes a Deed of Adherence (as soon as possible and in any event not later than the date of registration of the allotment in the register of allotments of the Company), and, upon the delivery of such deed, each such allottee shall be treated as a party to this Agreement. |
| 42 |
SCHEDULE 12
Anti-Dilution
Notwithstanding anything else in this Agreement, in the event of an Alteration of Share Capital, subject to law, the Company shall issue such bonus shares to ES1 or take such other action as may be required to maintain ES1's percentage shareholding at 18% of the Company's total issued Share capital.
| 43 |
SCHEDULE 13
Deed of Adherence
THIS DEED OF ADHERENCE is made on [ • ]
BY:
[insert name of New Shareholder of [insert address of New Shareholder] (the “New Shareholder”) in favour of the persons whose names are set out in the Schedule to this deed and is supplemental to the Subscription and Shareholders’ Agreement dated [ • ] 2013 between (1) Elan Science One Limited, (2) the Management Subscribers (as therein defined) (3) Nerano Pharma Limited and (4) Speranza Therapeutics Limited (the “Agreement”).
THE NEW SHAREHOLDER UNDERTAKES AS FOLLOWS:
| 1. | New Shareholder confirms that it has read a copy of the Agreement and covenants with each person named in the Schedule to this deed to perform and be bound by all the terms of the Agreement (subject to clause 12.3 of the Agreement) as if the New Shareholder were named in the Agreement as [an Investor/a Management Subscriber/a party thereto]. |
| 2. | This deed (and any dispute, controversy, proceedings or claim of whatever nature arising out of or in any way relating to this deed or its formation) shall be governed by and construed in accordance with Irish law. |
IN WITNESS whereof this deed has been executed by the New Shareholder and is intended to be and is hereby delivered on the date first above written.
[Schedule to Deed to include all parties (including by way of earlier Deeds of Adherence) to the Agreement]
| 44 |
Schedule 14
Clinical Plan
| 45 |
Schedule 15
Scyllo-Inositol "AZD-103" Structure
| 46 |
IN WITNESS whereof this Deed has been duly executed as a deed by the parties to it on the date set out at the beginning of this Deed.
| GIVEN UNDER THE COMMON SEAL | |
| of ELAN SCIENCE ONE LIMITED | |
| in the presence of: | |
| Signature of Director | |
| Signature of Director/Secretary | |
| GIVEN UNDER THE COMMON SEAL | |
| of NERANO PHARMA LIMITED | |
| in the presence of: | |
| Signature of Director | |
| Signature of Director/Secretary | |
| GIVEN UNDER THE COMMON SEAL | |
| of [Employee Share Nominee] | |
| in the presence of: | |
| Signature of Director | |
| Signature of Director/Secretary | |
| GIVEN UNDER THE COMMON SEAL | |
| of SPERANZA THERAPUETICS LIMITED | |
| in the presence of: | |
| Signature of Director | |
| Signature of Director/Secretary |
| 47 |
EXHIBIT D
Share Purchase Agreement
Agreed Form
SUBJECT TO CONTRACT / CONTRACT DENIED
Dated 2013
ELAN PHARMA INTERNATIONAL LIMITED
(Vendor)
and
SPERANZA THERAPEUTICS LIMITED
(Purchaser)
AGREEMENT
for the purchase of the entire issued share capital of
SPERANZA BIOPHARMA LIMITED
ãA & L GOODBODY

THIS AGREEMENT is dated 2013 and made between:
| (1) | Elan Pharma International Limited, a private limited company, incorporated in Ireland (no. 222276) whose registered office is at Treasury Building, Lower Grand Canal Street, Dublin 2 (the Vendor); and |
| (2) | Speranza Therapeutics Limited, a private limited company, incorporated in Ireland (registered no. 527543) whose registered office is at McCann FitzGerald, Riverside One, Sir John Rogerson's Quay, Dublin 2 (the Purchaser). |
RECITALS
| A. | The Vendor is the legal and beneficial owner of the entire issued share capital of the Company. |
| B. | The Vendor has agreed to sell and the Purchaser has agreed to purchase and pay for the Shares on the terms and conditions contained in this Agreement. |
IT IS HEREBY AGREED as follows:
| 1. | Definitions and Interpretation |
| 1.1. | Definitions: In this Agreement, unless the context otherwise requires: |
1963 Act means the Companies Act 1963;
1990 Act means the Companies Act 1990;
an Affiliate of any person means:
| (a) | any holding company or subsidiary of that person and any subsidiary of any such holding company; and |
| (b) | any other individual, company, body corporate, partnership or other entity which; |
| (i) | is Controlled by that person; |
| (ii) | Controls that person; or |
| (iii) | is Under Common Control with that person; |
Agreement means this share purchase agreement;
Assets has the meaning given in the Business Transfer Agreement;
Breach, in relation to a Warranty, means any instance of the Warranty, when made or given, being untrue and inaccurate in any respect;
Business Contracts has the meaning given in the Business Transfer Agreement;
Business Transfer Agreement means an agreement entered into on the date hereof between the Vendor and the Company under which the Vendor transfers the D5 Business to the Company;
Clinical Plan has the meaning given in the Transaction Agreement;
Company means Elan Science Nine Limited (in the process of changing its name to Speranza Biopharma Limited, details of which are set out in Schedule 1;
| 1 |
Completion means completion of the sale and purchase of the Shares under clause 3;
Control means the power of a person (whether alone or together with any other person or persons) to secure directly or indirectly, including through one or more intermediaries, that the affairs of another person are conducted in accordance with the wishes of the first-mentioned person, either by means of the holding of shares or the possession of voting power directly or indirectly in or in relation to that or any other person, or by virtue of any powers conferred by any applicable laws or regulations or the constitutional or other documentation regulating or managing the affairs of that or any other person, or otherwise, and the expressions Controlled, Controls and Under Common Control shall be construed accordingly;
Derivative means any compounds identified, obtained, developed, created, synthesized, generated, designed or resulting from, based upon, containing or incorporating the chemical structure of Scyllo-Inositol as those compounds are generically and/or specifically disclosed and claimed in the Licensed Patent Rights;
D5 Business means the research, development, manufacture and commercialization of a Product;
Directors means those listed in Schedule 1;
Dispute means any suit, action, proceedings and/or any dispute or difference which may arise out of or in connection with or which may relate in any way to this Agreement (including but not limited to any suit, action, proceedings, dispute or difference relating to the formation, interpretation or performance of any of this Agreement) and Disputes shall be construed accordingly;
Employees means those employees of any company within the Elan Group whose employment it has been agreed will transfer in accordance with the terms of the Business Transfer Agreement;
Encumbrance includes any adverse claim or right or third party right or interest; any equity; any option or right of pre-emption or right to acquire; any mortgage, charge, assignment, hypothecation, pledge, lien or security interest or arrangement of whatsoever nature; any reservation of title; any hire purchase, lease or instalment purchase agreement and any other encumbrance, priority or security interest or similar arrangement of whatever nature;
Group means the Company and the Subsidiary;
Group Company means the Company or the Subsidiary, as the case may be;
Intellectual Property and Intellectual Property Rights have the meaning given in the Business Transfer Agreement;
Ireland means Ireland excluding Northern Ireland and Irish will be construed accordingly;
Loan Agreements means the loan agreements entered into on the date hereof under which Nerano Pharma Limited is providing the Company with a ten year interest-free loan of $20,000,000 to fund development of the D5 Business, and EPIL is providing the Company with the ten year interest-free loan for $7,000,000 to fund development of the D5 Business;
Licensed Patent Rights shall have the meaning ascribed to it in the collaboration agreement (as amended) between Waratah Pharmaceuticals Inc. and EPIL dated 25 September 2006;
Listed Business Contracts means the Business Contracts listed in Schedule 3 to the Business Transfer Agreement;
Option Agreement means the Option and Royalty Deed of Agreement entered into by the Vendor and the Company on the date hereof under which the Vendor is granted the option to commercialize the Products in certain territories and to receive a royalty in relation to the Products from the Company;
Purchaser's Solicitors means McCann FitzGerald;
Proceedings means suits, actions or proceedings arising out of or in connection with or relating in any
| 2 |
way to any Dispute;
Product means any pharmaceutical preparation or dosage form containing Scyllo-Inositol or a Derivative;
Scyllo-Inositol means the compound “AZD-103”, as more specifically disclosed in the Licensed Patent Rights;
Shareholders Agreement means the agreement entered into on the date hereof between Elan Science One Limited, New InvestorCo Limited, New HoldCo Limited and the persons listed in Schedule 1 of the Shareholders Agreement, to regulate their relationship with each other as shareholders of New HoldCo Limited;
Shares means 100 shares of $1.00 each in the capital of the Company held by the Vendor, being the entire issued share capital of the Company;
Subsidiary means the company listed in the Schedule 2;
Transaction Agreement means the transaction agreement between the Vendor and Nerano Pharma Limited dated [19] May 2013;
Transaction Documents means:
| (a) | this Agreement; |
| (b) | the Business Transfer Agreement; |
| (c) | the Shareholders' Agreement; |
| (d) | the Option Agreement; |
| (e) | the Loan Agreements; |
| (f) | all documents or agreements, connected with or ancillary to any of the documents or agreements referred to at paragraphs (a) to (e) above, which are in the agreed form; and |
| (g) | any other document or agreement, which are entered into, or to be entered into, on Completion (or at any time during the period from the date of this Agreement up to, including and after the date of Completion) by some or all of the parties to this Agreement in connection with or which are ancillary to any of the foregoing documents or agreements; |
Transitional Services Agreement has the meaning given in the Transaction Agreement;
Vendor’s Group means the Vendor and each of its Affiliates;
Warranties mean the warranties in clause 4 and Schedule 3;
Warranty Claim means a claim for a Breach of any one or more of the Warranties;
and cognate terms will be construed accordingly.
| 1.2. | The Schedules referred to in this Agreement form an integral part of this Agreement and references to this Agreement include reference to them. |
| 1.3. | Headings are inserted for convenience only and do not affect the construction of this Agreement. |
| 1.4. | Interpretation: Unless the context otherwise requires: |
| 1.4.1. | a reference to the singular includes the plural and vice versa and any gender includes the other gender; |
| 3 |
| 1.4.2. | reference to writing or similar expressions includes transmission by fax; |
| 1.4.3. | a reference to any provision of law is a reference to that provision as amended, substituted, extended or re-enacted; |
| 1.4.4. | a reference to this Agreement, any other document or any specified provision of this Agreement or any other document is a reference to this Agreement, to such other document or to such provision as in force for the time being and as amended, extended, varied, assigned, novated, restated or supplementedfrom time to time |
| 1.4.5. | all references to “indemnity” and “indemnifying” any person against any circumstance include indemnifying and keeping that person harmless from each loss, liability or reasonable cost incurred as a result of: |
| (1) | defending or settling a claim alleging such liability relating to each relevant circumstance the subject matter of the indemnity; or |
| (2) | enforcing the indemnity. |
| 1.5. | Where any Warranty is given on terms that it is to the best of the knowledge and belief of the Vendor (or any other words to this or a similar effect), the Vendor will be deemed to have the knowledge and belief which they would have had if they had made all due and careful enquiries, including (to the extent reasonably required) enquiries of all relevant third parties. |
| 2. | Sale |
| 2.1. | Sale of Shares and Consideration: The Vendor as legal and beneficial owner shall sell, and the Purchaser shall purchase, the Shares for the sum of $1.00 (the receipt and adequacy of which is hereby acknowledged)free from all liens, charges and Encumbrances and together with all rights attaching now or in the future (including the right to all dividends or distributions of whatever nature declared, paid or made). |
| 3. | Completion |
| 3.1. | Completion: Completion shall take place simultaneously on the signing of this Agreement at the offices of the Vendor's Solicitors. On Completion, all legal and beneficial right, title and interest in the Shares shall pass, free from all Encumbrances and together with all rights attaching or accruing to them now or in the future, to the Purchaser. |
| 3.2. | Completion Obligations: Upon Completion, the Vendor shall deliver to the Purchaser: |
| 3.2.1. | transfers of the Shares duly executed by the Vendor in favour of the Purchaser together with the related share certificates; |
| 3.2.2. | the registers of allotment, transfers, members and directors and minute books (properly written up to the time immediately prior to Completion), a certified copy of the memorandum and articles of association, the common seal, the certificate of incorporation and (if applicable) any certificate of incorporation on change of name of the Group; |
| 3.2.3. | insofar as they are not in the custody of the Group, all the Group's financial and accounting books and records; |
| 3.2.4. | where relevant, a copy of the minutes of the board meeting of the Group, certified by the secretary of the relevant Group Company, approving all relevant matters pursuant to this Agreement and Completion (including all share transfers, resignations of directors and secretaries and the execution of all relevant agreements); |
| 4 |
| 3.2.5. | a written acknowledgement from the Vendor on terms reasonably satisfactory to the Purchaser acknowledging on its behalf and on behalf of each member of the Vendor’s Group that (save in respect of the loan from EPIL to the Company made pursuant to a Loan Agreement)neither the Company nor the Subsidiary is indebted to the Vendor or any member of the Vendor’s Group, or any director of them; |
and shall cause any persons nominated by the Purchaser to be validly appointed as additional directors of the Group, and then cause the Directors and the secretary of the Company and the directors and secretaries of the Group to retire.
| 3.3. | Payment of Consideration: Upon the Vendor complying with clause 3.2, the Purchaser shall pay the consideration for the Shares as provided by clause 2, and the receipt of the Vendor will be absolute discharge to the Purchaser. |
| 3.4. | The Purchaser shall not be obliged to complete this Agreement unless: |
| 3.4.1. | the Vendor complies with all of its obligations in clause 3.2; and |
| 3.4.2. | the purchase of all the Shares is completed simultaneously. |
| 4. | Warranties |
| 4.1. | Warranties: The Vendor warrants to the Purchaser in the terms set out in Schedule 3, as at the date of this Agreement that each of the Warranties is true and accurate subject only to any matter expressly provided for in this Agreement. |
| 4.2. | Limitations: Notwithstanding the provisions of clause 4.1, the Vendor will have no liability in respect of a Warranty Claim; |
| 4.2.1. | unless the Purchaser gives written notice to the Vendor setting out in reasonable detail the nature of the Warranty Claim and, in so far as it is possible to do so, the amount claimed or, in any event, an estimate thereof, within 18 months following Completion; |
| 4.2.2. | where the amount that would be otherwise recoverable from the Vendor (but for this clause 4.2.2) in respect of the Warranty Claim, when aggregated with any other amount or amounts recoverable in respect of other Warranty Claims, does not exceed $250,000, and in the event that the aggregated amounts exceed $250,000 the Vendor shall be liable in respect of the total aggregated amounts and not the excess only; and |
The Vendor’s total liability in respect of all Warranty Claims shall be limited to US$15,000,000.
| 4.3. | Purchaser’s Knowledge: |
| 4.3.1. | No information of which the Purchaser or its agents or advisers has knowledge (actual or constructive) and no investigation by or on behalf of the Purchaser prevents any Warranty Claim being made by the Purchaser or operates to reduce any liability of the Vendor or the amount recoverable by the Purchaser from the Vendor. |
| 4.3.2. | The Purchaser covenants to the Vendor that, as at the date of this Agreement, it is not actually aware of any circumstances which constitute a breach of any of the Warranties. |
| 4.4. | Exclusion of limitations: Notwithstanding any other provision of this Agreement: |
| 4.4.1. | No provision of this Agreement shall limit the liability of the Vendor for any claims under paragraph 2 of the Warranties relating to the Vendor’s title to the Shares or to the status or validity of the Shares; and |
| 5 |
| 4.4.2. | There shall be no limit in amount or time on the liability of the Vendor, whether under the Warranties or under any other provision of this Agreement, in respect of any claim which arises or is delayed as a result of dishonesty, fraud, wilful misconduct or wilful concealment. |
| 4.5. | Each Warranty is independent and is not limited by a provision of this Agreement or by another Warranty (except where this Agreement provides otherwise). |
| 4.6. | Each Warranty shall continue in full force and effect notwithstanding Completion. |
| 4.7. | The Vendor undertakes not to make a claim against any Group Company and/or a director, officer or employee of any Group Company which it may have in respect of a misrepresentation, inaccuracy or omission in or from information or advice provided by any Group Company or a director, officer or employee of any Group Company for the purposes of assisting the Vendor to give a Warranty. |
| 4.8. | Basis of recovery: If there is a claim for breach of the Warranties, the Vendor shall pay the Purchaser on demand (at the Purchaser’s option) an amount equal to either: |
| 4.8.1. | the reduction caused in the value of the Shares; or |
| 4.8.2. | if: |
| (1) | the value of an asset of the Group is or becomes less than the value would have been had the breach not occurred; or |
| (2) | a Group Company is subject to or incurs a liability or an increase in a liability to which it would not have been subject or which would not have incurred had the breach not occurred, |
the reduction in the value of the asset or, as the case may be, the amount of the liability or increased liability.
| 4.9. | Indemnification: The Vendor shall indemnify the Purchaser, it directors, its officers and its Affiliates (the Indemnified Parties), and keep the Indemnified Parties indemnified, on demand against each loss, liability and reasonable cost which the Indemnified Parties (or any of them) incur, whether before or after the start of an action, arising (directly or indirectly) out of: |
| 4.9.1. | the settlement of a claim against the Vendor in respect of a breach of any provision of this Agreement or the enforcement of a settlement; and |
| 4.9.2. | legal proceedings against the Vendor in respect of a breach or an alleged breach of any provision of this Agreement in which judgment is given for the Purchaser or the enforcement of the judgment. |
| 4.9.3. | any claim by any person that the Vendor in respect of this Agreement and the parties to the Business Transfer Agreement (together, the Relevant Parties) did not have the power, capacity or authority to execute and perform those agreements and the transactions contemplated by them; |
| 4.9.4. | any claim by any person that the execution and/or performance by the Relevant Parties of this Agreement or the Business Transfer Agreement conflicted with or constituted a default or required any consent under any provision of: |
| (1) | any agreement, arrangement or instrument to which any of the Relevant Parties (or any of their respective Affiliates) is a party; or |
| (2) | any law, order, judgment, decree, regulation or any other restriction of any kind by which any of the Relevant Parties (or any of their respective Affiliates) is bound; |
| 6 |
| 4.9.5. | any claim by any person that the Company does not have legal and beneficial title to, and ownership of, the Assets which were transferred to the Company, or purportedly transferred to the Company, pursuant to the Business Transfer Agreement; |
| 4.9.6. | there not being, at the time of execution of this Agreement, sufficient persons either (i) engaged by a Group Company (whether as employees or contractors) or (ii) whose services will be provided by the Vendor and/or its Affiliates pursuant to the Transitional Services Agreement who collectively have sufficient expertise and experience, (a) to conduct the D5 Business in the manner in which it is conducted as at the date of the Transaction Agreement and (b) to achieve the goals set out in the Clinical Plan; and |
| 4.9.7. | any claim by any person that the entire benefit of each of the Listed Business Contracts was not, or could not be, transferred to the Company pursuant to the Business Transfer Agreement. |
| 4.10. | No double recovery: The Purchaser shall not be entitled to recover under this Agreement more than once in respect of the same loss. |
| 5. | Miscellaneous |
| 5.1. | Entire Agreement: This Agreement supersede all prior representations, arrangements, understandings and agreements, and set out the entire, complete and exclusive agreement and understanding between the parties. The parties further agree and declare that all extrinsic evidence, including, without limitation, pre-contractual materials and oral or other evidence pre-dating this Agreement shall not be admissible or be capable of being relied on in any way in interpreting any provision of this Agreement. No party has relied on any representation, arrangement, understanding or agreement (whether written or oral) not expressly set out or referred to in this Agreement. Notwithstanding the foregoing, this clause is without prejudice to the right of the Purchaser to make any claim or claims against the Vendor or any other person other than pursuant to this Agreement for misrepresentation. |
| 5.2. | Remedies Cumulative: The provisions of this Agreement and the rights and remedies of the parties are independent, cumulative and are without prejudice and in addition to any other rights or remedies which a party may have whether arising under statute, at common law, in equity, under contract, by virtue of custom or otherwise. The exercise by a party of any one right or remedy under this Agreement, or at law or in equity will not (unless expressly provided in this Agreement, at law or in equity) operate so as to hinder or prevent the exercise by that party of any other right or remedy. |
| 5.3. | Further Assurance: At any time after the date of this Agreement, each party hereto will use all reasonable endeavours to procure that any documents are executed and any acts and things are done as may reasonably be required by any other party and at the sole cost and expense of such other party, for the purpose of giving to that party the full benefit of the provisions of this Agreement. For the avoidance of doubt, the parties agree that completion of this Agreement is not in any way subject to or conditional upon compliance with Section 60 of the 1963 Act or with any conditions of any acquisition finance of the Purchaser. |
| 5.4. | Notices: Any notice or other communication to be given or served under this Agreement may be delivered or may be sent by pre-paid post and will be deemed to have been duly served or given (in the case of delivery) at the time of delivery or (in the case of posting) 48 hours after posting. |
| 5.5. | Counterparts: This Agreement may be executed in any number of counterparts and by the several parties to it on separate counterparts, each of which when so executed will constitute an original but all of which together will evidence the same agreement. |
| 5.6. | Governing Law: This Agreement and all relationships created by it will in all respects be governed by and construed in accordance with Irish law. |
| 5.7. | Jurisdiction: |
| 7 |
| 5.7.1. | Subject to clause 5.7.2: |
| (1) | the Irish courts will have exclusive jurisdiction to hear, settle and/or decide any Dispute; |
| (2) | the Purchaser and the Vendor agree that the Irish courts are the most appropriate and convenient courts to hear and decide any Dispute and therefore that they will not argue to the contrary. |
| 5.7.2. | This clause is for the exclusive benefit of the Vendor and it will not prevent the Vendor from initiating any Proceedings in any other court of competent jurisdiction. To the extent permitted by law, the Vendor may take concurrent Proceedings in any number of jurisdictions. |
| 8 |
SCHEDULE 1
The Company
|
Name
|
Elan Science Nine Limited (in the process of changing its name to Speranza Biopharma Limited)
| |
|
Date of Incorporation
|
17 August 2011 | |
|
Country of
|
Ireland | |
|
Registered Number
|
502444 | |
|
Authorised Share
|
US$1,000,000 divided into 1,000,000 ordinary shares of US$1.00 each | |
|
Issued Share Capital
|
100 Shares issued and fully paid | |
|
Registered Office
|
Treasury Building, Lower Grand Canal Street, Dublin 2, Ireland | |
|
Directors
|
Continuing
None
|
Not Continuing
William F. Daniel
Nigel Clerkin
Mary Sheahan
|
|
Secretary
|
William F. Daniel
| |
| 9 |
SCHEDULE 2
Subsidiary
|
Name
|
Speranza Therapeutics Corp. |
|
Date of Incorporation
|
8 May 2013 |
|
Country of
|
USA (Delaware) |
|
Registered Number
|
5332090 |
|
Registered Office
|
Registered office in state of domicile: 160 Greentree Drive, Suite 101 Dover, DE 19904
Principal place of business: Third Floor Cambridge, MA 02139
|
|
Officers
|
Christopher E. Burns, President John L. Donahue, Vice President and Secretary Gregory M. Bokar, Vice President Grainne McAleese, Chief Financial Officer and Treasurer Diana L. King, Assistant Secretary
|
| 10 |
SCHEDULE 3
Warranties
| 1. | VENDOR CAPACITY AND TITLE |
| 1.1. | Power and Authority: The Vendor has full power and authority and has taken all actions necessary to enter into and perform this Agreement, the Business Transfer Agreement and each other Transaction Document to which it is party. |
| 1.2. | Valid and Binding on Vendor: The Vendor’s obligations under this Agreement, the Business Transfer Agreement and each other Transaction Document to which it is party constitutes legal obligations which are valid and binding on the Vendor in accordance with their terms. |
| 1.3. | Company’s Business: The Group has the right, power and authority to conduct the D5 Business. |
| 1.4. | No Breach of Obligations: The execution and the performance of this Agreement and each other Transaction Document (save for the Business Transfer Agreement) will not, and the execution and performance of the Business Transfer Agreement did not: |
| 1.4.1. | conflict with or constitute a default or require any consent under any provision of: |
| (1) | any agreement or instrument to which the Vendor, the Company or any of their respective Affiliates is a party; |
| (2) | the Vendor’s or the Company’s memorandum or articles of association; or |
| (3) | any law, order, judgment, decree, regulation or any other restriction of any kind by which the Vendor, the Company or any of their respective Affiliates is bound; |
| 1.4.2. | relieve any other party to a contract with the Vendor or a Group Company of any obligation or enable that party to vary or terminate its rights or obligations under that contract; |
| 1.4.3. | result in any Group Company losing the benefit of, or having to make any repayment in respect of, any asset, grant, subsidy, licence, right or privilege which it enjoys at the date of this Agreement; or |
| 1.4.4. | result in the creation, imposition, crystallisation or enforcement of any Encumbrance on any asset of a Group Company. |
| 2. | SHARES |
| 2.1. | Group Structure: |
| 2.1.1. | The Vendor is the sole legal and beneficial owner of the Shares, which are free from Encumbrances and which constitute the whole of the issued and allotted shares of the Company and there is no agreement to create or give any Encumbrance over or in respect of any of the Shares. |
| 2.1.2. | The Company is the sole legal and beneficial owner of the entire issued share capital of the Subsidiary. |
| 2.2. | Rights to Share Capital: No person has the right to call for the allotment, issue, sale or transfer of any share or loan capital of the Company under any option or other agreement or obligation (including conversion rights and rights of pre-emption) and there are no Encumbrances on shares of the Subsidiary (and there is no agreement to create or give any Encumbrance over or in respect of any of the shares of the Subsidiary). |
| 11 |
| 2.3. | Shares Fully Paid: All of the issued shares of $1.00 each in the capital of the Company are fully paid or credited as fully paid. |
| 3. | Information |
The information set out in Schedule 1 and Schedule 2 is true, accurate and not misleading.
| 4. | TRADING |
With the exceptions of any actions required to enter into or perform the Transaction Documents, no Group Company has traded. No Group Company has incurred any liabilities other than pursuant to the terms of the Transaction Documents.
| 5. | ASSETS |
| 5.1. | All of the Assets are legally and beneficially owned solely by a Group Company free from any Encumbrance. |
| 5.2. | The assets of the Group Companies comprise all the assets, facilities and services necessary for the carrying on of the D5 Business in the manner in which it is conducted at the date of this Agreement. |
| 5.3. | No material Asset is shared with another person and the Group does not materially depend for its business on, or use, any assets, facilities or services owned or supplied by any member of the Vendor’s Group save for any information technology, human resources, legal and other forms of corporate services that are provided by members of the Vendor’s Group, details of which have been disclosed in writing to Nerano Pharma Limited1. |
| 5.4. | Each insurable Asset has, at all material times, been and is, at the date of this Agreement, insured to its full replacement value against each risk normally insured against by a prudent person operating the types of business operated by the Group. |
| 6. | INTELLECTUAL PROPERTY |
| 6.1. | As far as the Vendor is aware the registered Intellectual Property owned by a Group Company is: |
| 6.1.1. | valid and enforceable (if granted), and nothing has been done or omitted to be done by which it may cease to be valid and enforceable (if granted), and, where relevant, all appropriate renewal fees and other steps required for its prosecution, maintenance or protection have been paid and taken; and |
| 6.1.2. | not being and not likely to be, infringed or attacked or the subject of any claim for ownership or compensation by any person or, in the case of Intellectual Property which is the subject of an application for registration, not being or likely to be opposed or for any other reason likely to fail to be granted in full. |
| 6.2. | In respect of the unregistered Intellectual Property that is owned by a Group Company: |
| 6.2.1. | as far as the Vendor is aware is not the subject of a challenge; |
| 1 | Any such material services which are required for the operation of the Business and which have not been put in place prior to closing are to be provided pursuant to the transitional services agreement. |
| 12 |
| 6.2.2. | as far as the Vendor is aware is not being infringed or attacked; |
| 6.2.3. | the Vendor has not received any formal written notice of any claim for ownership or compensation by any person; and |
| 6.2.4. | as far as the Vendor is aware in the case of Intellectual Property that is the subject of an application for registration, the application is not being opposed. |
| 6.3. | As far as the Vendor is aware, if and to the extent that any Intellectual Property has been licensed to a Group Company or a Group Company has the benefit of any licence of Intellectual Property, each such licence: |
| 6.3.1. | has been validly granted and is valid and enforceable; and |
| 6.3.2. | is not subject to any right of termination. |
| 6.4. | Nothing has been done or omitted to be done by any Group Company or any other party to any such licence which would enable that licence to be terminated or which constitutes a breach of that licence. |
| 7. | business CONTRACTS |
| 7.1. | The entire benefit of each of the Listed Business Contracts was transferred to the Company pursuant to the Business Transfer Agreement. |
| 7.2. | The assignment of the benefit of the Listed Business Contracts to the Company pursuant to the Business Transfer Agreement did not: |
| 7.2.1. | conflict with or constitute a default or require any consent under any provision of Listed Business Contract; |
| 7.2.2. | relieve any other party to a Listed Business Contract of any obligation or enable that party to vary or terminate its rights or obligations under that Listed Business Contract; |
| 7.2.3. | result in any Group Company losing the benefit of, or having to make any repayment in respect of, any asset, grant, subsidy, licence, right or privilege which it enjoys at the date of this Agreement; or |
| 7.2.4. | result in the creation, imposition, crystallisation or enforcement of any Encumbrance on any asset of a Group Company. |
| 7.3. | No fact or circumstance exists which might invalidate or give rise to any ground by which any of the Business Contracts may be terminated, avoided, repudiated or the terms modified to the Company’s detriment. |
| 7.4. | No party to any Business Contract has given notice of its intention to terminate, or has sought to repudiate or disclaim, the Business Contract. |
| 7.5. | Neither a Group Company nor, as far as the Vendor is aware, any other party to a Business Contract is in default of the Business Contract and no fact or circumstance exists which might give rise to such a default. |
| 8. | EMPLOYEES |
| 8.1. | The Vendor has provided to Nerano Pharma Limited the details in writing of all of the material terms and conditions of employment of each employee of the Group, including the following: their name, age, job title/description, workplace location, grade, start date, notice period, full-time, part-time, permanent, specific purpose and/or fixed-term status, salary/wage, commission, bonuses (including the criteria for |
| 13 |
any bonuses), all other benefits, entitlements or payments (stating whether discretionary or contractual).
| 8.2. | The maximum amount payable to the Employees pursuant to the severance terms applicable to the Elan Group is no more than $3,000,000. |
| 9. | Litigation and compliance with law |
| 9.1. | The Vendor’s Group conducted the D5 Business and dealt with the Assets in all material respects in accordance with all applicable legal and administrative requirements. |
| 9.2. | As far as the Vendor is aware, no fact or circumstance exists which might give rise to a civil, criminal, arbitration, administrative, tribunal, disciplinary or other proceeding, investigation or enquiry involving the Company or the Assets or a person for whose acts or defaults the Company may be vicariously liable. |
| 9.3. | There is no outstanding judgment, decree, order, arbitral award or decision of a court, tribunal, arbitrator, regulatory body or other authority involving the D5 Business against any member of the Vendor’s Group. |
| 9.4. | None of the activities or rights of any member of the Vendor’s Group relating to the D5 Business are or were ultra vires, unauthorised, invalid, unenforceable or in material breach of contract or covenant. The Vendor has not committed any breach of statutory duty or committed any tortious or other unlawful act which could lead to a successful claim or injunction being made or granted against it involving the D5 Business. |
| 9.5. | The Group has all material licences, consents, permits, approvals, registrations and authorisations (public and private) necessary to own and operate its assets and to carry on its business in the manner in which it is being, and has from time to time been, carried on. |
| 10. | solvency |
| 10.1. | The Vendor is not, and was not immediately prior to execution of the Business Transfer Agreement, insolvent or unable to pay its debts nor has the Vendor stopped or suspended payment of its debts nor has the Vendor sought from its creditors significant extensions of time for the payment of its debts nor has the Vendor commenced negotiations with one or more of its creditors with a view to rescheduling or restructuring any of its indebtedness. |
| 10.2. | No action is being taken by the Registrar of Companies to strike the Vendor from the register of companies. |
| 14 |
IN WITNESS whereof this Agreement has been duly executed as a deed by the parties to it on the date set out at the beginning of this Agreement.
| GIVENUNDER THE COMMON SEAL | |
| of ELAN PHARMA INTERNATIONAL LIMITED | |
| in the presence of: | |
| Signature of Director | |
| Signature of Director/Secretary | |
| GIVENUNDER THE COMMON SEAL | |
| of SPERANZA THERAPEUTICS LIMITED | |
| in the presence of: | |
| Signature of Director | |
| Signature of Director/Secretary |
| 15 |
Exhibit E
Clinical Plan
IN WITNESS whereof this Agreement has been duly executed by the Parties on the date set out at the beginning of this Agreement.
SIGNED for and on behalf of ELAN PHARMA INTERNATIONAL LIMITED
in the presence of:
| /s/ Gregory M. Bokar | /s/ William F. Daniel | |
| Signature | ||
| Deputy General Counsel | Director | |
| Title |
SIGNED for and on behalf of NERANO PHARMA LIMITED
in the presence of:
| /s/ Seamus Mulligan | ||
| /s/ Brendan Murphy Sir John Rogerson's Quay Dublin 2 |
||
| Signature | ||
| Director | ||
| Title |
Transaction Agreement
Exhibit (a)(8)
THIS DOCUMENT IS IMPORTANT AND REQUIRES YOUR IMMEDIATE ATTENTION. If you are in any doubt about the action you should take, you are recommended to seek financial advice immediately from your stockbroker, bank manager, solicitor, accountant, fund manager or other appropriate independent financial adviser, who, if you are resident in Ireland, is authorised or exempted under the European Communities (Markets in Financial Instruments) Regulations (Nos 1 to 3) 2007 (as amended) of Ireland, or the Investment Intermediaries Act 1995 of Ireland or, who, if you are resident in the United Kingdom, is authorised under the Financial Services and Markets Act 2000 of the United Kingdom or who, if you are resident outside of Ireland or the UK, from an appropriately authorised independent financial advisor.
If you sell or have sold or otherwise transferred all of your Elan Shares please send this document to the purchaser or transferee or to the stockbroker, bank or other agent through whom the sale or transfer was effected for transmission as soon as possible. If you sell or have sold or otherwise transferred only part of your holding of Elan Shares you should retain this document and consult your stockbroker, bank or other agent through whom the sale or transfer was effected immediately as to the action you should take.
Shareholders resident in jurisdictions outside of Ireland or the United Kingdom should consult their legal and/or financial advisors in their respective jurisdictions to ensure compliance with local securities law.
Your attention is drawn in particular to Part 5 of this document which sets out and describes certain risk factors that you should carefully consider when deciding whether or not to vote in favour of the Resolutions to be proposed at the Extraordinary General Meeting to be held on 17 June 2013. Investors should read the whole of this document.
Unless otherwise stated, defined terms in this document have the meanings given to them in Part 9 Definitions.
ELAN CORPORATION, PLC
(Incorporated and registered in Ireland under the Companies Acts 1963 to 2012 with registered number 30356)
PROPOSED ROYALTY PARTICIPATION AGREEMENT WITH THERAVANCE
PROPOSED PURCHASE OF AOP
PROPOSED DISPOSAL OF ELND005
PROPOSED SHARE REPURCHASE PROGRAM
AND
NOTICE OF EXTRAORDINARY GENERAL MEETING
Your attention is drawn to the letter from the Chairman of Elan on pages 5 to 7 of this document, which contains the recommendation of the Board to Shareholders to vote in favour of the Resolutions described herein. You should read this document in its entirety and consider whether to vote in favour of the Resolutions in light of the information contained in this document.
Notice of an Extraordinary General Meeting of Elan, to be held at 10.00 a.m. (Irish time) on 17 June 2013 at the O’Callaghan Davenport Hotel, 8/10 Merrion Street Lower, Dublin 2, Ireland is set out at the end of this document. Shareholders will find enclosed a Form of Proxy for use at the Extraordinary General Meeting. Shareholders are requested to complete and return the Form of Proxy whether or not they intend to be present at the meeting. To be valid, Forms of Proxy should be completed and signed in accordance with the instructions printed thereon and returned by post or by hand so as to reach the Registrars, Computershare Investor Services (Ireland) Limited, Heron House, Corrig Road, Sandyford Industrial Estate, Dublin 18, Ireland, as soon as possible and, in any event, by no later than 10.00 a.m. (Irish time) on 15 June 2013. Return of a Form of Proxy will not preclude a holder of Ordinary Shares from attending and voting at the meeting in person. Holders of ADSs wishing to appoint a proxy, should complete and sign the Form of Proxy and return it to the Company’s ADS Depositary, Citibank N.A., P.O. Box 43099, Providence, Rhode Island 02940-5000, United States, as soon as possible, but in any event so as to be received no later than 3.00 p.m. (New York time) on 13 June 2013.
IMPORTANT INFORMATION: Elan, at this time, is not offering to the public any Elan Shares, or any other securities in connection with the Transactions. This document is not a prospectus, nor is it a document containing information regarded by the Central Bank of Ireland as being equivalent to that of a prospectus. This document is intended only to provide Elan Shareholders
with sufficient information so as to enable them to make an informed decision as to the manner in which to exercise their voting rights in respect of the Resolutions proposed for consideration at the Extraordinary General Meeting.
Davy and Davy Corporate Finance each of which are regulated in Ireland by the Central Bank of Ireland, are acting for Elan and no one else in relation to the matters referred to herein. In connection with such matters, Davy and Davy Corporate Finance, its affiliates and their respective directors, officers, employees and agents will not regard any other person as their client, nor will they be responsible to anyone other than Elan for providing the protections afforded to their clients or for providing advice in connection with the matters described in this document or any matter referred to herein.
Morgan Stanley & Co. International plc, which is authorised by the Prudential Regulation Authority and regulated by the Financial Conduct Authority and the Prudential Regulation Authority in the United Kingdom, is acting as financial adviser to Elan and for no one else in relation to the matters referred to herein. In connection with such matters, Morgan Stanley, its affiliates and their respective directors, officers, employees and agents will not regard any other person as their client, nor will they be responsible to anyone other than Elan for providing the protections afforded to their clients or for providing advice in connection with the matters described in this document or any matter referred to herein.
Ondra LLP, which is regulated by the Financial Conduct Authority in the United Kingdom, is acting for Elan and no one else in relation to the matters referred to herein. In connection with such matters, Ondra LLP, its affiliates and their respective directors, officers, employees and agents will not regard any other person as their client, nor will they be responsible to anyone other than Elan for providing the protections afforded to their clients or for providing advice in connection with the matters described in this document or any matter referred to herein.
Citigroup Global Markets Inc, which is a member of the Securities Investor Protection Corporation and is a registered broker-dealer regulated by the Securities and Exchange Commission and Citigroup Global Markets Limited, which is authorised by the Prudential Regulation Authority and regulated by the Prudential Regulation Authority and the Financial Conduct Authority, are acting for Elan and no one else in relation to the Revised Offer. In connection with such matters, Citigroup Global Markets Inc and Citigroup Global Markets Limited, its affiliates and their respective directors, officers, employees and agents will not regard any other person as their client, nor will they be responsible to anyone other than Elan for providing the protections afforded to their clients or for providing advice in connection with the Revised Offer described in this document.
The Company has a secondary listing on the Irish Stock Exchange. For this reason, the Company is not subject to the same on-going regulatory requirements as those which would apply to an Irish company with a primary listing on the Irish Stock Exchange including the requirement that certain transactions require the approval of shareholders. The Extraordinary General Meeting is being convened in order to comply with the requirements of the Irish Takeover Rules. For further information, Shareholders should consult their own financial adviser. The contents of this document have not been, and are not required to be, approved by the Irish Stock Exchange.
No person has been authorized to give any information or make any representations other than those contained in this Circular and, if given or made, such information or representations must not be relied on as having been so authorized. The delivery of this Circular shall not, under any circumstances, create any implication that there has been no change in the affairs of the Company since the date of this Circular or that the information in it is correct as of any subsequent time.
This document has been prepared in accordance with the requirements of Irish law and is subject to disclosure and procedural requirements that may be different from those under US law. Any financial figures included or incorporated in this document may have been prepared in accordance with non-US accounting standards that may or may not be comparable to the financial statements of a US company.
Any holder of 1% or more of any class of relevant securities of Elan or of Royalty Pharma may have disclosure obligations under Rule 8.3 of the Irish Takeover Panel Act, 1997, Takeover Rules 2007 (as amended).
This Circular contains forward-looking statements that involve substantial risks and uncertainties. You can identify these statements by the fact that they use words such as “anticipate”, “estimate”, “project”, “target”, “intend”, “plan”, “will”, “believe”, “expect” and other words and terms of similar meaning in
| 2 |
connection with any discussion of future financial performance or events. Among the factors that could cause actual results to differ materially from those described or projected herein are the following: risks related to delays or difficulties encountered in obtaining, or the failure to obtain, the approval of Elan’s Shareholders of the Theravance, AOP and ELND005 Transactions and the Share Repurchase Program, the possibility that intervening events could arise which could alter the timing, or the ability to consummate the Theravance, AOP or the ELND005 Transactions or the Share Repurchase Program even if Elan Shareholder approval is obtained, the risk that third parties could challenge any or all of the Transactions, even if the Transactions are approved by Elan Shareholders and consummated, risks that the Transactions do not provide the benefits to Elan that are anticipated, and whether Elan can satisfy the escrow conditions with respect to its debt financing; as Elan’s principal source of revenue may remain a royalty on sales of Tysabri, the potential of Tysabri, which may be severely constrained by increases in the incidence of serious adverse events (including death) associated with Tysabri (in particular, by increases in the incidence rate for cases of PML), or by competition from existing or new therapies (in particular, oral therapies), and the potential for the successful development and commercialization of products, whether internally or by acquisition, especially given the separation of the Prothena business which left Elan with no material pre-clinical research programs or capabilities; Elan’s ability to maintain sufficient cash, liquid resources, and investments and other assets capable of being monetized to meet its liquidity requirements; the success of our development activities, and R&D activities in which Elan retains an interest, including, in particular, the impact of the announced discontinuation of the development of bapineuzumab intravenous in mild to moderate Alzheimer’s disease; failure to comply with anti-kickback, bribery and false claims laws in the United States, Europe and elsewhere; difficulties or delays in manufacturing and supply of Tysabri; trade buying patterns; the impact of potential biosimilar competition, the trend towards managed care and health care cost containment, including Medicare and Medicaid; legislation and other developments affecting pharmaceutical pricing and reimbursement (including, in particular, the dispute in Italy with respect to Tysabri sales), both domestically and internationally; failure to comply with Elan’s payment obligations under Medicaid and other governmental programs; exposure to product liability (including, in particular, with respect to Tysabri) and other types of lawsuits and legal defense costs and the risks of adverse decisions or settlements related to product liability, patent protection, securities class actions, governmental investigations and other legal proceedings; Elan’s ability to protect its patents and other intellectual property; claims and concerns that may arise regarding the safety or efficacy of Elan’s product candidates; interest rate and foreign currency exchange rate fluctuations and the risk of a partial or total collapse of the euro; governmental laws and regulations affecting domestic and foreign operations, including tax obligations; whether Elan is deemed to be an investment company or a passive foreign investment company; general changes in United States and International generally accepted accounting principles; growth in costs and expenses; and the impact of acquisitions, divestitures, restructurings, product withdrawals and other unusual items. A further list and description of these risks, uncertainties and other matters can be found in Elan’s Annual Report on Form 20-F for the fiscal year ended 31 December 2012, and in its Reports of Foreign Issuer on Form 6-K filed with the SEC. Elan assumes no obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise.
Nothing in this document other than the Profit Forecast (as defined in Part 8) is intended to be a profit forecast and no statement in this document should be interpreted to mean that the earnings per Elan Share for the current or future financial periods will necessarily be greater than those for the relevant preceding financial period. Capitalised terms in this document, unless otherwise stated, are defined in Part 9 of this document.
| 3 |
CONTENTS
| Page | |
| Expected Timetable of Principal Events | 4 |
| Part 1 Letter from the Chairman | 5 |
| Part 2 Description of the Transactions | 8 |
| Part 3 Questions and Answers | 12 |
| Part 4 Bond Offer and Other Matters | 15 |
| Part 5 Risk Factors | 18 |
| Part 6 Financial Impact of the Transactions | 23 |
| Part 7 Additional Information | 24 |
| Part 8 Profit Forecast and report of KPMG and the Financial Advisors | 30 |
| Part 9 Definitions | 35 |
| Appendix I Extracts from the 2011 AOP Financial Statements | 41 |
| Notice of Extraordinary General Meeting | 82 |
EXPECTED TIMETABLE OF PRINCIPAL EVENTS(1)(2)
| Issue of this document | 27 May 2013 |
| Closing of Bond Offer and placing of proceeds into Escrow | 31 May 2013 |
| Latest time and date for receipt of proxies representing | 13 June 2013 |
| registered holders of ADSs(3) | at 3.00 p.m. (New York time) |
| Latest time and date for receipt of proxies representing | 15 June 2013 |
| registered holders of Ordinary Shares(4) | at 10.00 a.m. (Irish time) |
| Extraordinary General Meeting of Elan | 17 June 2013 |
| at 10.00 a.m. (Irish time) | |
| Expected Completion of the Theravance Transaction | 18 June 2013 |
| Expected Release of proceeds of Bond Offer from Escrow(5) | by 21 June 2013 |
| Expected Completion of the ELND005 Transaction | 28 June 2013 |
| Expected Completion of the AOP Transaction | 1 August 2013 |
Full details of the timetable will be published by way of announcements on the Regulatory Information Service.
Notes:
| (1) | Each of the times and dates in the above timetable is based on current expectations and is subject to change. |
| (2) | All references in this document are to Irish time unless otherwise stated. |
| (3) | Receipt by the ADS Depositary, Citibank N.A. |
| (4) | Receipt by the Registrar, Computershare Investor Services (Ireland) Limited. |
| (5) | Assumes satisfaction of the escrow release conditions set out in Part 4 of this Circular. |
| 4 |
PART 1
LETTER FROM THE CHAIRMAN
ELAN CORPORATION, PLC
(Registered in Ireland under the Companies Acts 1963 to 2012 with registered number 30356)
| Directors: | Registered Office |
| Robert A. Ingram (Chairman) | Treasury Building |
| Kelly Martin (Executive Director, CEO) | Lower Grand Canal Street |
| Gary Kennedy (Director) | Dublin 2 |
| Patrick Kennedy (Director) | Ireland |
| Giles Kerr (Director)* | |
| Kieran McGowan (Director) | |
| Kyran McLaughlin (Director) | |
| Donal O’Connor (Director) | |
| Richard Pilnik (Director) | |
| Dennis Selkoe (Director)* | |
| Andrew Von Eschenbach (Director) |
* indicates that the director will be retiring at the 2013 Annual General Meeting to be held on 30 May 2013
URGENT: PROTECT YOUR INVESTMENT IN ELAN
VOTE “YES” ON THE RESOLUTIONS TODAY
Dear Shareholder,
On behalf of the Board of Directors, I am writing to ask for your support at the Company's Extraordinary General Meeting (EGM) to be held on June 17, 2013. This meeting is especially important and could have a major impact on the value of your investment in Elan.
ELAN HAS A PROVEN TRACK RECORD
Over the past decade, Elan has made tremendous progress on our goal of building a profitable biotechnology company with strong cash flow, de-risking our business and balance sheet, and returning value to Shareholders. Consider that Elan’s current management team and Board has:
| · | Delivered revenues of $1.2 billion(1) in 2012, 150% higher than 2004, and 2012 Adjusted EBITDA(2) of positive $194 million, versus negative $197 million in 2004 |
| · | Paid off all debt (subject to the 2021 Notes issuance), which stood at $2.4 billion in 2006 |
| · | Launched the blockbuster MS treatment Tysabri, twice |
| · | Significantly reduced clinical risk in our Alzheimer’s program through the establishment of Janssen AI with Johnson & Johnson in 2009 |
| · | Successfully divested our EDT business in 2011, generating $1 billion of capital |
| · | Completed the separation of our drug discovery business, Prothena, in 2012, providing investors additional choice and investment clarity |
| · | And – with the restructuring of our Tysabri collaboration with Biogen Idec earlier this year – returned $1 billion to shareholders through the “Dutch Auction” Tender Offer buyback, and established a cash dividend that directly links shareholder value to the long-term performance and cash flow generation of Tysabri. |
| (1) | Revenue of continuing and discontinued operations under U.S. GAAP. As a result of the restructuring of our collaboration agreement with Biogen Idec the results of Tysabri that are included in the 2012 Consolidated Income statement are presented as a discontinued operation. |
| (2) | Adjusted EBITDA on a total operations basis. |
| 5 |
WE BELIEVE ELAN’S NEW PLATFORM WILL
DELIVER SUPERIOR
VALUE FOR SHAREHOLDERS
With the completion of the Tysabri restructuring – in addition to returning $1 billion to Shareholders – we have transformed Elan’s strategic platform to provide for continued growth and value creation. The new platform is built on three strong pillars:
| 1. | Tysabri Royalty: Elan maintains a highly valuable royalty on this established blockbuster product in a large and growing therapeutic category. In 2012, global in-market net sales of Tysabri exceeded $1.6 billion and constituted approximately 12% of the global MS market by value, and the number of patients using Tysabri increased by 12% last year. We have little-to-no related R&D expenses and largely fixed corporate infrastructure. Tysabri should continue as a strong income generator for the Company and Shareholders through the dividend directly linked to Tysabri’s performance. |
| 2. | Theravance Royalty Participation: The transaction we announced on May 13, 2013 provides Elan and our Shareholders with a participation interest in potential future royalty payments related to four respiratory programs partnered by Theravance and Glaxo Group Limited, including BREOTM ELLIPTATM 100/25 mcg which was recently approved by the U.S. Food and Drug Administration as an inhaled long-term, once daily maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD). The respiratory market is expected to grow to approximately $31 billion by 2025 (+40% from 2012), driven by an aging population, increased prevalence in emerging markets, earlier treatment intervention globally and treatment guidelines recommending longer-acting therapies. Products developed by Glaxo under previous respiratory programs (in which Elan does not have a participation interest) had more than a 40% share of the respiratory market in 2012, and Glaxo is expected to maintain its leading position for years to come, with its strong late-stage pipeline in collaboration with Theravance. |
The Board believes that the Theravance Transaction provides Elan and Shareholders with potential long-term, high quality cash flow with significant after-tax margin and has contractually agreed to recommend it to Shareholders. As with Tysabri, Shareholders will benefit directly – as 20% of the payments received by Elan will be paid to Shareholders as a dividend.
As summed up by one leading analyst team: “We find the Theravance transaction impressive for: 1) immediate EPS accretion, 2) immediate return of capital to shareholders, 3) virtually 100% pure net profit, 4) long duration assets, 5) partnered with premier respiratory companies, 6) no operational risk, and 7) positive NPV.”
| 3. | AOP Orphan: The agreement to acquire Austria-based AOP Orphan, which we announced on May 20, 2013, is consistent with our strategy to diversify our business and to gain access to attractive late stage pipeline assets with geographic presence in a growing region of the world. AOP has a strong regional presence in the high growth field of orphan and rare diseases. AOP, a pioneer in this field, has been successfully growing its business in the double digits since inception and continues to focus on unmet needs in a broad array of therapeutic areas. The $80 billion Eastern and Central European pharma market, which is AOP’s focus, is expected to double in size over the next 10 years. |
On May 20, 2013, we also announced an investment in NewBridge Pharmaceuticals, a regulatory and marketing partner for the global industry in some of the world’s fastest-growing and most under-served regions of the world, particularly the Middle East and Africa.
The Board believes that AOP Orphan and NewBridge together create a highly unique business. The geographic markets in which they operate are characterized by underlying growth and demand for health care products, broad economic development and increased patient and caregiver knowledge in disease areas such as oncology, cardiovascular-pulmonology, hematology, gastroenterology, neurology and a variety of rare and orphan diseases. Both businesses are headed by strong, experienced leaders, who will remain in place.
| 6 |
CONTINUED COMMITMENT TO FINANCIAL DISCIPLINE
AND RETURNING VALUE TO SHAREHOLDERS
Our commitment to lower risk growth, capital structure and cost discipline, and returning value to shareholders is as strong as ever.
On May 20, 2013, we announced the divestiture of our ELND005 asset to an independent company, Speranza Therapeutics. This allows Elan to eliminate the operating activities associated with the development of the drug (2013 estimated spend:~ $80 million), while at the same time maintaining a share of the potential upside.
We have also announced our plan for an additional $200 million share repurchase program, which would be in addition to the $1 billion buyback already completed and the dividends linked directly to Tysabri and Theravance royalties.
ROYALTY PHARMA OFFER SUBSTANTIALLY UNDERVALUES ELAN TODAY
Despite the progress we have made and strong potential for future growth, Royalty Pharma has made an opportunistic offer to acquire Elan at $12.50 per share, and stated that if Elan’s Shareholders vote in favour of any of the compelling transactions we have announced, including Theravance and AOP, its offer will lapse. As announced on May 23, 2013 your Board of Directors, after careful review and consideration and with the assistance of its executive management team as well as outside financial and legal advisors, has determined that the Royalty Pharma revised offer substantially undervalues the company.
Your Board includes former top-tier executives at leading pharmaceutical companies and healthcare organizations, including GlaxoSmithKline, Eli Lilly & Co, Athena Neurosciences, Harvard Medical School, the former Commissioner of the FDA and Director of the National Cancer Institute – as well as highly experienced financial executives. We take our fiduciary responsibilities to Shareholders very seriously, and URGE YOU TO REJECT THE ROYALTY PHARMA OFFER.
VOTE YES ON THE RESOLUTIONS AT THE JUNE 17 SHAREHOLDER MEETING
At Elan’s EGM to be held on June 17, 2013:
| · | Vote YES on the Theravance Resolution to approve the Theravance Transaction |
| · | Vote YES on the AOP Resolution to approve the AOP Transaction |
| · | Vote YES on the ELND005 Resolution to approve the ELND005 Transaction |
| · | Vote YES on the Share Repurchase Resolution to approve the Share Repurchase Program |
The Board unanimously recommends all Shareholders to vote in favour of the Resolutions, as the Directors intend to do in respect of their own beneficial holdings, amounting in aggregate to 823,341 Elan Shares, representing approximately 0.16% of the existing issued share capital of Elan.
You can vote by telephone, by Internet, or by signing, dating and returning the enclosed Form of Proxy in the postage-paid envelope provided.
If you need assistance, or have questions, and you hold ordinary shares contact Computershare Investor Services (Ireland) Limited on the dedicated Helpline number +353 1 447 5107 or see www.eproxyappointment.com.
Holders of American Depositary Shares should contact Innisfree M&A Incorporated as follows:
Toll-free from the US and Canada: 1-877-750-9498
Free-phone from Ireland and the UK: 00-800-4664-7000
Call collect: 212-750-5833 (Banks, brokers and others)
We thank you for your continued support of Elan.
Sincerely,

Robert A. Ingram
Chairman
27 May 2013
| 7 |
PART 2
DESCRIPTION OF THE TRANSACTIONS
Transactions
As part of the first phase of investment under its new strategy, Elan has announced four strategic Transactions:
Theravance Transaction
On 12 May 2013, the Company entered into the Theravance Royalty Participation Agreement pursuant to which the Company will purchase a participation interest in potential future royalty payments related to certain Royalty Interest Products each of which is a treatment or a potential treatment for COPD and/or asthma. Under the terms of the Theravance Royalty Participation Agreement, the Company will make a one-time cash payment of $1.0 billion to Theravance in exchange for a 21% participation interest in the potential future royalty payments from the Royalty Interest Products earned by Theravance when, as and if received.
On 10 May 2013, the FDA approved BREO™ ELLIPTA™ as an inhaled long-term, once-daily maintenance treatment of airflow obstruction in patients with COPD. RELVAR™ ELLIPTA™ (the chemical equivalent of BREO ELLIPTA with a different brand name for markets outside the USA) for the treatment of COPD and asthma patients is currently under review by the European Medicines Agency and in Japan. In addition, a new drug application for ANORO™ ELLIPTA™ was accepted by the FDA in February 2013, and the Prescription Drug User Fee Act goal date was set as 18 December 2013. MABA ‘081 is an investigational, single molecule bifunctional bronchodilator with both muscarinic antagonist and beta2 receptor agonist activities, which Glaxo and Theravance have announced they plan to advance into Phase 3 during 2013. Vilanterol (VI) monotherapy, a component of BREO ELLIPTA/RELVART ELLIPTA and ANORO ELLIPTA, as a monotherapy is the fourth Royalty Interest Product. Glaxo is one of the world's largest pharmaceutical companies and a leader in the development and commercialization of respiratory products. BREO™ ELLIPTA™ and RELVAR™ ELLIPTA™ (in which Elan has a participation interest) are aimed at succeeding Glaxo's Advair(r)/Seretide™ franchise, which had reported 2012 net sales of approximately $8 billion. BREO™ ELLIPTA™ and RELVAR™ ELLIPTA™ are unique, however, in that they are a once daily therapy.
The Theravance Transaction is subject to the satisfaction or waiver of certain conditions, including (i) the approval of Elan Shareholders, (ii) the absence of laws or governmental orders that prevent the closing of the Theravance Transaction, (iii) the establishment of a Theravance subsidiary that will receive the covered royalty payments, and (iv) other customary conditions.
If Elan Shareholder approval is not obtained, Elan breaches any of its undertakings relating to the holding of the shareholder meeting, or certain other conditions are not met (including if the closing of the Theravance Transaction is prevented by a law or governmental order), then Elan will be required to pay Theravance a $10.0 million break fee.
The Theravance Royalty Participation Agreement includes an undertaking by Elan that this Circular contain a recommendation from the Board to Shareholders, to vote in favour of the Theravance Transaction and that such recommendation will not be withdrawn, modified or altered in any way without Theravance’s prior written consent. If the Company was to breach this provision of the Theravance Royalty Participation Agreement, Elan could be exposed to a claim for damages for breach of contract. Notwithstanding and independent of this contractual undertaking, the Board believes that the Theravance Transaction is in the best interests of Elan and the Shareholders, and has recommended that Shareholders vote to approve the transaction.
If Shareholder approval is obtained and the other closing conditions are met or waived, the Theravance Transaction is expected to close shortly after the EGM.
AOP Transaction
On May 19, 2013, Elan and SASR, a newly formed subsidiary of Elan, entered into the AOP Share Purchase Agreement for its pending purchase of AOP. AOP researches, develops, produces and distributes innovative drugs and administration methods for rare diseases. AOP focuses on
| 8 |
haematology and oncology, cardiology and pulmonology, neurology and psychiatry, and metabolic and genetic disorders.
AOP is currently a privately owned company and is controlled by its founder and chief executive officer, Dr. Rudolf Stefan Widmann. It is based in Vienna and its sales in Austria, Poland and the Czech Republic account for over 60% of AOP’s revenues. AOP’s EBITDA was €17.6 million (approximately $23 million) (preliminary and unaudited) for the year ended 31 December 2012 and €15.7 million (approximately $20 million) for the year ended 31 December 2011, in both cases excluding estimated amounts attributable to AOP’s current generic products business, which is not being acquired.
AOP’s five most significant products, by revenue, for the year ended 31 December 2012, were:
| Subject to transfer of |
|||||
| Name | Indication Used | License | |||
| Remodulin® | Pulmonary arterial hypertension | Yes | |||
| Thromboreductin® | Essential thrombocythemia | No | |||
| Tetmodis® | Huntington’s disease | Yes | |||
| Busilvex® | Conditioning before stem cell transplantation | Yes | |||
| Canemes® | Chemotherapy-induced nausea & vomiting (CINV) | Yes |
Sales of these five products represented approximately 97% of AOP’s revenues of €59 million (approximately $76 million) (preliminary and unaudited) in the year ended 31 December 2012 and sales of two of those, Remodulin® and Thromboreductin®, represented approximately 93% of AOP’s revenues in that year.
The consideration payable under the AOP Share Purchase Agreement consists of an initial purchase price of €263.5 million (approximately $339 million), composed of €175.7 million in cash (approximately $226 million) and €87.8 million (approximately $113 million) of Elan Ordinary Shares, each deliverable at closing. There are also potential post-close milestone payments of up to €270 million (approximately $347 million) that will become payable by Elan on the advancement of certain late stage clinical programs to filing and regulatory acceptance (2014-2018 time frame).
The AOP Share Purchase Agreement contains customary representations and warranties from the seller, Dr. Widmann, about the business and some limited representations and warranties from Elan. Dr. Widmann also undertakes not to compete with AOP for three years from closing.
Completion of the AOP Transaction is subject to a number of conditions precedent, including (i) the approval of Elan Shareholders, (ii) that there have been no material adverse events between signing of the AOP Share Purchase Agreement and closing of the AOP Transaction, (iii) obtaining certain third-party consents and (iv) delivery of audited financial statements for AOP for the year ended 31 December 2012. The AOP Transaction is subject to termination if these conditions are not fulfilled by 1 August 2013. Elan is entitled to waive any of these conditions at its sole discretion, other than the receipt of Elan Shareholder approval.
If closing does not occur by 1 August 2013 due to a failure by Elan to obtain Shareholder approval, Elan will be required to pay to Dr. Widmann a break fee of €5 million (approximately $6 million) as reimbursement for his costs and expenses in connection with the AOP Transaction.
Under the terms of the AOP Transaction, each party is responsible for its own costs and expenses. The agreement is governed by Austrian law.
The EBITDA amounts noted above are prepared under Austrian GAAP. The 2012 EBITDA is preliminary and unaudited.
ELND005 Transaction
On 19 May 2013 Elan, through its wholly owned subsidiary, EPIL, entered into the ELND005 Transaction Agreement with Nerano (an Irish incorporated company controlled by Mr. Seamus Mulligan) pursuant to which Elan agreed to transfer the intellectual property and other assets related to ELND005 (scyllo-inositol) to a newly-formed company, Speranza Biopharma.
Pursuant to the ELND005 Transaction Agreement, Elan will also transfer the Transition Collaboration Agreement to Speranza Biopharma. Elan has agreed to contribute $63.0 million to Speranza Biopharma, which may only be used to fund the ELND005 Business, and to advance a further $7.0 million to Speranza Biopharma as a 10 year interest free loan. Nerano will also extend a
| 9 |
$20.0 million 10 year interest free loan to Speranza Biopharma. Pursuant to the ELND005 Transaction Agreement, EPIL will retain liabilities and claims relating to the ELND005 Business arising prior to the closing of the ELND005 Transaction and Speranza Biopharma will assume liabilities and claims relating to the ELND005 Business arising after the closing of the ELND005 Transaction. Elan will also remain responsible for up to $3.0 million in retention bonuses or severance payments made by Speranza Biopharma within 18 months of the ELND005 Transaction.
Speranza Therapeutics owns 100% of Speranza Biopharma. Pursuant to the ELND005 Transaction Agreement, Elan will subscribe for 18% of the shares in Speranza Therapeutics. Nerano will hold 62% of Speranza Therapeutics shares and the remaining 20% will be issued to senior management who work on the ELND005 Business and who will transfer from Elan to Speranza Biopharma. Elan’s share ownership in Speranza Therapeutics cannot be diluted below 18% or increased above 18%.
Pursuant to the ELND005 Transaction Agreement, Elan will receive royalty payments in the amount of 3% of worldwide sales of all ELND005 products (subject to reduction in the case of generic competition and other limited circumstances) as well as an option to receive exclusive commercialization rights to ELND005 products in certain territories. Elan may also receive two one-time $200.0 million milestone payments on the occurrence of each of the following events: (i) any sale, transfer or other disposal of the Speranza Group or all or substantially all of its business (which milestone will be subject to a deduction for all future milestone payments paid to Transition by Speranza Biopharma, the maximum amount of which would be $93 million); and (ii) attainment by the Speranza Group of worldwide annual net income of $1.5 billion.
The ELND005 Transaction is subject to certain conditions, including (i) the approval of Elan’s Shareholders, (ii) receipt of required governmental approvals or clearances, (iii) absence of laws or governmental orders preventing completion of the ELND005 Transaction and (iv) agreement and execution of certain ancillary agreements. Either party may terminate the ELND005 Transaction Agreement if the conditions are not satisfied on or before 1 August 2013.
Nerano will nominate all members of the Speranza Therapeutics board of directors. Elan and Nerano have agreed to adopt a business purpose plan for Speranza Therapeutics, within 90 days of the consummation of the ELND005 Transaction, which will provide a general framework for Speranza Therapeutics’ business activities. However, Nerano will be responsible for all operating and financial decisions on the development and commercialization of the ELND005 compound. If the Speranza Group requires additional funding to implement the business plan, it will have the ability to require Elan and Nerano to contribute a further $10.0 million to Speranza Biopharma to complete any clinical activities or obtain any regulatory approvals necessary under the business plan, with Elan to contribute 78% of this amount and Nerano to contribute the remaining 22%.
On or before the date that is 15 months after the consummation of the ELND005 Transaction, Elan and Nerano may terminate the business because it is not viable due to a clinical event or other determination. If this occurs, the initial shareholder loans will be repaid in the amounts of $7.0 million to Elan and $20.0 million to Nerano, with any remaining surplus to be paid to Elan. At any time after the date that is 15 months after the consummation of the ELND005 Transaction, if Elan and Nerano terminate the business because it is not viable, then 22% of Speranza Biopharma’s remaining net cash will be used to repay any loan made by Nerano and 78% of the remaining net cash of Speranza Biopharma will be used to repay any loan made by Elan, with any remaining surplus to be paid to Elan.
The ELND005 Transaction Agreement prohibits Speranza Biopharma from actions not in the ordinary course of business without the consent of Elan, including the following:
| · | material changes of business purpose or the business purpose plan; |
| · | significant acquisitions or disposals of assets; |
| · | alteration of share capital or of the constitutional documents of the company; |
| · | dissolution or winding up; |
| · | use of the initial funding for matters not related to ELND005 or contemplated by the business purpose plan; and |
| · | dividends or distributions. |
Share Repurchase Program
Elan currently has Shareholder authority to conduct market purchases of its Shares and is seeking Shareholder approval at its forthcoming annual general meeting for a renewal of that authority. As
| 10 |
announced on 20 May 2013, the Company intends to launch a share repurchase program in an amount of up to $200 million (assuming the granting of the relevant authority at the annual general meeting).
The Share Repurchase Program will be conducted in accordance with relevant US and Irish securities laws, the existing Shareholder authority and any renewal of that authority at the annual general meeting to be held on 30 May 2013.
As the Company is in an Offer Period, the execution of the Share Repurchase Program in the Offer Period requires Shareholder approval under Rule 4.1(f) of the Irish Takeover Rules. Accordingly Shareholders are being asked to approve the Share Repurchase Program and the purchase of Shares by the Company in the Offer Period in accordance with existing and future Shareholder authorities granted pursuant to the provisions of Part XI of the Irish Companies Act 1990.
| 11 |
PART 3
QUESTIONS AND ANSWERS ABOUT THE PROPOSED TRANSACTIONS
Introduction
To help you understand what is involved in the Transactions the following are answers to some of the questions you may have as an Elan Shareholder regarding the Transactions. These questions and answers only highlight some of the information contained in this Circular. They may not contain all the information that is important to you. You should read carefully this entire Circular to understand fully the proposed Transactions and the voting procedures for the EGM.
| Q: | Why am I receiving this Circular? |
| A: | Elan has entered into certain agreements that are described in this Circular with respect to the Transactions. |
Pursuant to Rule 21 of the Irish Takeover Rules, during the Royalty Pharma Offer Period, Elan is required to obtain Shareholder approval of the Transactions at an EGM of the Company. In the event that the Offer Period has ended, Shareholder approval of the Transactions will not be required and the Company may decide not to proceed with the EGM in those circumstances.
This document and the enclosed materials summarize the Transactions and provide information as to how to appoint a proxy or vote your Elan Shares by mail, telephone or internet.
Your vote is very important.
Elan encourages you to submit your proxy or vote your Elan Shares by mail, telephone or internet as soon as possible.
| Q: | What are the proposals on which I am being asked to vote? |
| A: | You are being asked to vote to approve the (i) Theravance Transaction; (ii) AOP Transaction; (iii) ELND005 Transaction; and (iv) Share Repurchase Program. |
| Q: | What are the reasons for the Transactions? |
| A: | As announced on 20 May 2013, the Transactions are designed to create a balance of risk (science, molecules, regulatory and reimbursement) with the benefit of diversification (therapeutics, geographies, science and operational constructs) to produce long-term growth in income and value, that allows for participation in various parts of the industry value chain for the direct benefit of Shareholders. |
| Q: | What is the position of the Elan Board of Directors regarding the proposals being put to a vote at the EGM? |
| A: | The Elan Board of Directors believes that the Transactions are fair and in the best interests of Elan and its Shareholders. The Elan Board of Directors recommends that Elan Shareholders vote “FOR” the Resolutions to approve the Transactions. |
| Q: | When are the Transactions expected to be completed? |
| A: | As of the date of this Circular, the Transactions are expected to complete as soon as possible following the EGM and in any event by the third quarter of 2013. However, no assurance can be provided as to when or if the Transactions will complete. The required vote of Elan Shareholders to approve the Transactions at the EGM, as well as the necessary regulatory consents and approvals, must first be obtained and certain other conditions specified in the Transaction documents must be satisfied or, to the extent permissible, waived. |
| Q: | If any of the Resolutions to approve the Transactions are passed, what happens to the Royalty Pharma Offer? |
| A: | If any of the Resolutions are passed, Royalty Pharma has stated in its Revised Offer Document that its Offer will lapse and will therefore cease to be capable of acceptance. |
| 12 |
| Q: | What will happen if the Transactions are not approved by Shareholders? |
| A: | (i) | In the event the Theravance Resolution is not passed by Shareholders, the Theravance Transaction will not be put into effect and a break fee of $10 million will be payable to Theravance; |
| (ii) | In the event that the ELND005 Resolution is not passed by Shareholders, the ELND005 Transaction will not be put into effect; |
| (iii) | In the event that the AOP Resolution is not passed by Shareholders, the AOP Transaction will not be put into effect and a break fee of €5 million (approximately $6 million) will be payable to Dr. Widmann (the current shareholder of AOP); |
| (iv) | In the event that the Share Repurchase Resolution is not passed by Shareholders, the Share Repurchase Program will not be put into effect; and |
| (v) | If any of the Transactions (other than the Share Repurchase Program) are not approved the 2021 Notes issued in the Bond Offer will be redeemed and the proceeds of the Bond Offer will be released from escrow back to the 2021 Noteholders together with accrued and unpaid interest of approximately $3 million on the 2021 Notes from the Issue Date up to, but not including, the date of special mandatory redemption. |
| Q: | When and where will the EGM be held? |
| A: | Elan will hold an EGM of the Company on 17 June 2013 at 10.00 a.m. (Irish time) at the O’Callaghan Davenport Hotel, 8/10 Merrion Street Lower, Dublin 2, Ireland. |
| Q: | What vote is required to approve the Resolutions? |
| A: | Approval of the Resolutions requires the affirmative vote of a majority of the votes cast by holders of Elan Shares outstanding at the Voting Record Date and entitled to vote, assuming a quorum is present at the EGM. Consequently, as long as a quorum is present, a failure to vote, an abstention from voting or a broker non-vote will have no effect on the proposal to approve the Resolutions. |
| Q: | Is it possible to approve each of the AOP Transaction, the Theravance Transaction, the ELND005 Transaction and the Share Repurchase Program individually? |
| A: | Yes, it is possible to individually approve the Theravance Transaction, the ELND005 Transaction, the AOP Transaction and the Share Repurchase Program. Each Resolution is separate and none are interconditional. |
| Q: | Who is entitled to vote? |
| A: | The Company, pursuant to Section 134A of the Companies Act, 1963, specifies that only those holders of Ordinary Shares registered in the Register as of the Voting Record Date (or in the case of an adjournment at close of business two days before the time fixed for the adjourned meeting) shall be entitled to attend and vote at the meeting in respect of the number of shares registered in their names at the time. Changes in the Register after that time will be disregarded in determining the right of any person to attend and/or vote at the meeting. Holders of Elan Ordinary Shares of the Voting Record Date are entitled to receive notice of and to vote at the EGM and any adjournments and postponements thereof. |
If your shares are held in “street name” (by a bank, broker or other nominee), then you are not the registered owner of the shares and you should refer to the question “If my shares are held in “street name” by my bank, broker or other nominee, will my bank, broker or other nominee, vote my shares for me?” for details on how to vote your shares.
| Q: | How do I vote? |
| A: | If you are an Elan Shareholder of record, you may vote your Shares at the EGM in one of the following ways: |
| • | Proxies representing registered holders of Ordinary Shares must be received by the Registrar, Computershare Investor Services (Ireland) Limited, at Heron House, Corrig Road, Sandyford Industrial Estate, Dublin 18, Ireland, no later than 10.00 a.m. (Irish time) on 15 June 2013. Completion and return of a Form of Proxy will not preclude holders of Ordinary Shares from attending and voting at the meeting in person should they so wish. Details of how to vote over the Internet are provided on the Form of Proxy. |
| 13 |
| • | Proxies representing registered holders of ADSs must be returned so that they are received by the ADS Depositary, Citibank N.A., P.O. Box 43099, Providence, Rhode Island 02940-5000, United States, no later than 3.00 p.m. (New York time) on 13 June 2013. Details of how to vote by telephone or over the internet are provided on the Form of Proxy. |
Holders of ADSs in nominee accounts should follow the instructions provided by their bank or broker custodian.
| Q: | If my shares are held in “street name” by my bank, broker or other nominee will my bank, broker or other nominee, vote my shares for me? |
| A: | Only if you provide your bank, broker or other nominee with instructions on how to vote your shares. Therefore, you should instruct your bank, broker or other nominee to vote your shares, by following the directions your bank, broker or other nominee provides. If you do not instruct your bank, broker or other nominee, your bank, broker or other nominee will generally not have the discretion to vote your shares. |
| Q: | How many votes do I have? |
| A: | Registered holders of Ordinary Shares are entitled to one vote for each Elan Share that they own. Registered holders of ADSs will be entitled to one vote for every ADS held up until the cut-off time of 3.00 p.m. (New York time) on 13 June 2013. |
| Q: | What do I need to do now? |
| A: | After carefully reading and considering the information contained in this Circular, please fill out and sign the Form of Proxy, and then mail your completed and signed Form of Proxy in the enclosed prepaid envelope as soon as possible so that your Elan Shares may be voted at the EGM, or you may follow the instructions on the proxy card and vote your Elan Shares by telephone or over the internet. Your Form of Proxy or your telephone or internet directions will instruct the persons identified as your proxy to vote your shares at the EGM. |
If you sign and send in your Form of Proxy and do not indicate how you want to vote, your proxy will be voted “FOR” each Resolution.
| Q: | What documents have I been sent? |
| A: | This Circular, (which includes the Notice of EGM) and (i) in the case of a registered holder of Ordinary Shares a Form of Proxy; and (ii) in the case of a holder of ADSs the Depositary’s Notice and a Form of Proxy. |
| Q: | What if I have any more questions? |
| A: | If you have read this document and still have questions: |
| · | If you hold Ordinary Shares contact Computershare Investor Services (Ireland) Limited on the dedicated Shareholder Helpline number +353 1 447 5107 or see www.eproxyappointment.com. |
| · | Holders of American Depositary Shares should contact Innisfree M&A Incorporated as follows: |
Toll-free from the US and Canada: 1-877-750-9498
Free-phone from Ireland and the UK: 00-800-4664-7000
Call collect: 212-750-5833 (Banks, brokers and others)
For legal reasons the above helpline will not provide advice on the merits of the Transactions or give any financial, investment, legal or taxation advice. For financial, investment, legal or taxation advice, you should consult your own financial, investment, legal or taxation adviser who, if you are based in Ireland, is authorised or exempted under the European Communities (Markets in Financial Instruments) Regulations (Nos 1 to 3) 2007 (as amended) of Ireland, or the Investment Intermediaries Act 1995 of Ireland or, who, if you are resident in the United Kingdom, is authorised under the Financial Services and Markets Act 2000 of the United Kingdom, or who, if you are resident outside of Ireland or the UK, from an appropriately authorised independent financial advisor.
| 14 |
PART 4
BOND OFFER AND OTHER MATTERS
Bond Offer
As announced on 20 May 2013, the Company, through its wholly owned subsidiaries, Elan Finance plc and Elan Finance Corp, intends to raise $850,000,000 through the issuance of 6.25% senior notes due 2021. The 2021 Notes will mature on 15 June 2021. Upon satisfaction of the escrow release conditions (as set out below) and the release of funds from escrow, the Company expects to use the net proceeds from the Bond Offer for general corporate purposes, including working capital requirements, capital expenditures, acquisitions and share repurchases. The Company and certain of its existing and future material restricted subsidiaries (other than the Issuers, and following completion of the AOP Transaction, AOP) will fully and unconditionally guarantee the 2021 Notes on a senior unsecured basis.
The 2021 Notes will be the Company’s senior unsecured obligations and will rank equally in right of payment with all of its existing and future senior unsecured indebtedness, and senior to its existing and future subordinated indebtedness. The 2021 Notes will be effectively subordinated to all of the Company’s secured indebtedness to the extent of the value of the assets securing that indebtedness.
The guarantee of the 2021 Notes by each guarantor, including the Company, will be the senior unsecured obligation of such guarantor and will rank equally in right of payment with all existing and future senior unsecured indebtedness of such guarantor, and senior to the existing and future subordinated indebtedness of such guarantor. The guarantee of the 2021 Notes by each guarantor will be effectively subordinated to all secured indebtedness of such guarantor, to the extent of the value of the assets securing that indebtedness.
The 2021 Notes will bear interest at the rate of 6.25% per annum, paid every six months in arrears on 15 June and 15 December of each year, commencing on 15 December 2013. Interest on the 2021 Notes will accrue from the Issue Date, which is expected to be 31 May 2013.
On the Issue Date, the Issuers, the Trustee and the Escrow Agent will enter into the Escrow Agreement. Pursuant to the Escrow Agreement, the Issuers will deposit or cause to be deposited the gross proceeds of the Bond Offer, (after deduction of the Initial Purchasers’ discounts), together with additional amounts sufficient to redeem the 2021 Notes in connection with a special mandatory redemption on 21 June 2013, subject to extension until 31 July 2013, if a special mandatory redemption were to occur on such date, into a segregated escrow account, until the date that the escrow release conditions described below are satisfied or a special mandatory redemption is required.
In addition, the Escrow Agreement will provide that, on or before 20 June 2013 (unless the escrowed funds have already been released), the Issuers will deposit (or cause to be deposited) into the escrow account an amount of cash that, when taken together with the escrowed funds then held in the escrow account, will be sufficient to fund a special mandatory redemption on the second Business Day following 31 July 2013. If the Issuers fail to deposit (or cause to be deposited) such amounts into the escrow account on or before 25 June 2013, a special mandatory redemption will be triggered (as set out below).
The Issuers will be entitled to obtain release of the escrow funds upon delivery to the Escrow Agent on or before 21 June 2013, or if the escrow is extended, 31 July 2013, of an officers’ certificate to the effect that:
| · | requisite approval of the Shareholders for consummation of the Theravance Transaction, the AOP Transaction and the ELND005 Transaction has been obtained; |
| · | all conditions precedent to the consummation of the Theravance Transaction have been satisfied or waived and (ii) the Theravance Transaction will be consummated on substantially the same terms set forth in the Theravance Royalty Participation Agreement concurrently with the release of funds on deposit with the Escrow Agent; and |
| · | no default or event of default shall have occurred and be continuing under the Indenture. |
| 15 |
The Escrow Agent will release the escrowed funds to the Trustee to effect a special mandatory redemption if:
| · | the Escrow Agent has not received the officer’s certificate described above on or before 21 June 2013 or, if the escrow is extended, 31 July 2013; |
| · | the Issuers deliver a certificate to the Escrow Agent certifying that the Issuers will not pursue the consummation of the Theravance Transaction; or |
| · | the Issuers fail to timely deposit (or cause to be timely deposited) additional interest required to extend the escrow period as described above. |
The special mandatory redemption price for the Notes is equal to the sum of 100% of the gross proceeds of the Notes issued on the Issue Date together with accrued and unpaid interest on the Notes from the Issue Date to, but not including, the date of special mandatory redemption.
The Bond Offer is not subject to Shareholder approval. However, if Shareholder approval is not obtained for the Theravance Transaction, the AOP Transaction and the ELND005 Transaction, the proceeds of the Bond Offer will be released from escrow to the 2021 Noteholders together with accrued and unpaid interest on the 2021 Notes from the Issue Date up to, but not including, the date of special mandatory redemption.
In addition to the foregoing, upon the occurrence of certain change of control events of the Company, each 2021 Noteholder will have the right to require that the Issuers repurchase all or a portion (equal to $200,000 or an integral multiple of $1,000 in excess thereof) of such 2021 Noteholder’s 2021 Notes at a purchase price in cash equal to 101% of the principal amount thereof, plus accrued and unpaid interest thereon to the date of repurchase.
Pursuant to the terms of the Indenture that will govern the 2021 Notes, following a change of control event (which includes a change of control of the Company), the Company will be restricted from making certain payment transactions including the payment of dividends, the repurchase of Shares, the repayment of subordinated debt and the making of certain investments (other than permitted investments under the terms of the Indenture).
Dividend Policy
On 4 March 2013, Elan announced the initiation of a cash dividend program directly linked to Tysabri market performance, calculated as a percentage of the Tysabri royalties paid to Elan by Biogen Idec pursuant to the Tysabri Transaction. Initially, Elan expects that the percentage of Tysabri royalties to be paid to Elan Shareholders pursuant to the Tysabri Dividend Program will be 20%. Elan expects to make these dividend payments in twice-yearly installments, with the first dividend expected to be paid in the fourth quarter of 2013.
On 13 May 2013, Elan announced that, upon the close of the Theravance Transaction, Elan will initiate a cash dividend program directly linked to royalty participation interest payments received by Elan pursuant to the Theravance Transaction. Initially, Elan expects that the percentage of the royalty participation interest payments to be paid to its Shareholders pursuant to the Theravance Dividend Program will be 20%. Elan expects to make these dividend payments to its Shareholders in twice-yearly installments following the closing of the Theravance Transaction. For additional information about the Theravance Transaction, see “Theravance Transaction” set out in Part 2 of this Circular.
Debt Redemption
On 2 April 2013, Elan announced that it had issued an irrevocable notice of redemption to redeem all of the $600 million in aggregate principal amount of the 2019 Notes. The total redemption payment on the 2019 Notes was $706.7 million plus accrued and unpaid interest. The redemption was completed on 2 May 2013. In connection with the redemption, Elan will record a net charge on debt retirement of approximately $119 million in the second quarter of 2013, including a non-cash write-off of approximately $11 million related to unamortized deferred financing costs.
NewBridge Transaction
On 4 April 2013, Elan invested $40 million in preferred stock of NewBridge a speciality pharmaceutical company, headquartered in Dubai, UAE, specializing in in-licensing, acquiring, registering and commercializing drugs approved by the FDA, the European Medicines Agency and Japanese
| 16 |
Pharmaceuticals and Medical Devices Agency to treat diseases with high regional prevalence in the Middle East, Africa, Turkey and the Caspian region (the “NewBridge Transaction”).
Elan acquired a 48.3% equity interest in NewBridge and has the right to nominate two directors to the board of the company. Under the terms of the investment Elan also has an option, exercisable from 1 January 2014 to 15 March 2015, to acquire the shares of certain other significant investors for an aggregate price of approximately $240 million, which would represent, together with Elan’s current holdings, approximately 95% of the equity interest in NewBridge. Elan agreed not to acquire any additional equity of NewBridge prior to the exercise of the option without consent of the board of directors.
| 17 |
PART 5
RISK FACTORS
Risk Factors Relating to the Transactions
The following risk factors should be considered carefully by Shareholders when deciding whether or not to vote in favour of the Resolutions. The risk factors should be read in conjunction with all other information relating to the Transactions contained in this document. The risks and uncertainties set out below are those which the Directors believe are material risks relating to the Transactions. Additional risks and uncertainties are not currently known to the Directors, or which the Directors currently consider to be immaterial, may also have a material adverse effect on the Transactions.
Risks Related to the Theravance Transaction
The Theravance Transaction may not complete.
On 12 May 2013, Elan entered into the Theravance Royalty Participation Agreement for the Theravance Transaction, pursuant to which Elan will purchase a participation interest in potential future royalty payments related to the Royalty Interest Products. Under the terms of the Theravance Royalty Participation Agreement, Elan will make a one-time cash payment of $1.0 billion to Theravance in exchange for a 21% participation interest in the potential future royalty payments from the Royalty Interest Products earned by Theravance when, as and if received.
For RELVAR™ ELLIPTA™/BREO™ ELLIPTA™ and vilanterol VI monotherapy, Theravance is entitled to receive royalties from Glaxo of 15% of the first $3.0 billion of combined annual global net sales, and 5% of combined annual global net sales above $3.0 billion. If ANORO™ ELLIPTA™ is approved and commercialized, royalties on annual global net sales are upward tiering and range from the 6.5% digits to 10%. If MABA ‘081 is successfully developed and commercialized as monotherapy, Theravance is entitled to receive royalties from Glaxo of between 10% and 20% of the first $3.5 billion of annual global net sales, and 7.5% of all annual global net sales above $3.5 billion.
The Theravance Transaction is subject to the satisfaction or waiver of certain conditions, including (i) the approval of Elan Shareholders, (ii) the absence of laws or governmental orders that prevent the closing of the Theravance Transaction, (iii) the establishment of a Theravance subsidiary that will receive the covered royalty payments, and (iv) other customary conditions.
The Theravance Transaction agreement may be terminated by Theravance prior to closing if the required Elan Shareholder approval is not obtained by 18 June 2013 or if the required EGM of Elan Shareholders to approve the Theravance Transaction is not held on or before 17 June 2013. If Shareholder approval is not obtained or certain other conditions are not met, then Elan will be required to pay Theravance a $10.0 million break fee.
The Company cannot guarantee whether the closing conditions for the Theravance Transaction will be satisfied or whether any of the conditions under which Theravance is permitted to terminate the agreement will occur. As a result, the Company cannot assure you that the Theravance Transaction will be completed on a timely basis, or at all. If the Theravance Transaction is not completed, the market price of Elan Ordinary Shares could decline, Elan would nevertheless remain liable for the significant expenses incurred related to the Theravance Transaction and the $10.0 million break fee, and a special mandatory redemption will occur pursuant to the terms of the Bond Offer.
Elan’s revenues pursuant to the Theravance Transaction will be substantially dependent on revenues generated by Glaxo with respect to the Royalty Interest Products, and on Glaxo’s development, manufacturing and marketing efforts in relation to those products.
If the Theravance Transaction is consummated, the amount Elan earns will be dependent on Glaxo’s development, manufacturing and commercialization efforts in relation to the four respiratory programs involved in the Theravance Transaction. Any negative developments relating to those programs, such as safety, efficacy or reimbursement issues, the introduction or greater acceptance of competing products, or adverse regulatory or legislative developments, may reduce Glaxo’s royalty payments, if any, to Theravance, and accordingly the amount generated by Elan’s 21% participation interest therein, which would impact Elan’s revenues and adversely affect its results of operations. In the event Glaxo ceases to develop or commercialize any such products, or otherwise chooses to terminate its rights to any such products, Elan’s potential revenue stream would cease for such products.
| 18 |
The Royalty Interest Products may not generate sales revenue.
Only one product of the four potential products covered by the Theravance Royalty Participation Agreement has received regulatory approval for commercialization, and none of the programs have generated any sales revenue and there can be no assurance that they will in the future. On 10 May 2013, the FDA approved BREO™ ELLIPTA™ as an inhaled long-term, once-daily maintenance treatment of airflow obstruction in patients with COPD. However, the remaining three potential products covered by the Theravance Royalty Participation Agreement have not received FDA or other regulatory approval necessary for commercialization and there can be no assurance that they will. Sales, if any, of BREO™ ELLIPTA™, or of the other three Royalty Interest Products, if they are successfully developed and receive regulatory approval for marketing, may be adversely affected by a number of factors. The regulatory approval for BREO™ ELLIPTA™ contains restrictions on use and significant safety warnings related to asthma-related death with vilanterol based products to be provided on the product label, which could limit sales. If any of the other product candidates that contain vilanterol are successfully developed and approved for marketing, they may carry similar restrictions. Furthermore, incidence of side effects with respect to the approved product or to the three product candidates, if they are successfully developed and approved, could adversely affect sales, prompt regulatory review, require significant changes to product labels or result in market withdrawal. BREO™ ELLIPTA™ may compete, with respect to some or all indications, with existing drugs in the market, including drugs, such as Advair, sold by Glaxo. New competing products, including products developed by Theravance and/or Glaxo, may enter the market with respect to BREO™ ELLIPTA™ or any of the nonapproved Royalty Interest Products if they are approved for marketing in the future. If any competing product has a similar or more attractive profile in terms of efficacy, convenience or safety, future sales under the programs covered by the Theravance Royalty Participation Agreement could be limited. Competitive products may be developed by Theravance, Glaxo or another pharmaceutical company. In addition, the Theravance Royalty Participation Agreement does not include any other drugs made using different combinations of the chemical compounds composing the Royalty Interest Products, any other products based on partly the same molecules, or any other products developed or to be developed in the future by Theravance and/or Glaxo. For example, the Theravance Transaction does not include any royalty participation interest associated with UMEC/VI/FF, an investigational medicine also in development under the LABA collaboration between Theravance and Glaxo, nor any such interest associated with MABA ‘081 in combination with any other therapeutically active component, including an inhaled corticosteroid, or with any other MABA compound as monotherapy or in combination. Any such indications or products, if successfully developed and marketed, may compete with the Royalty Interest Products, and reduce their sales and resulting royalty payments. As a result of any such competitive products, Theravance’s and Glaxo’s management attention and resources may also be diverted from the Royalty Interest Products and their financial interest in the marketing of the Royalty Interest Products may not be wholly aligned with Elan’s.
Any or all of the above factors could lead to volatility in the number of patients who begin or continue to use the products under these programs or discontinue the use of such products in any period, or affect the prices at which the Royalty Interest Products can be sold or even affect whether the Royalty Interest Products generate any sales at all – all of which would negatively impact Elan’s revenues from the Theravance Transaction.
The patents underlying the Royalty Interest Products may be challenged or may not protect the Royalty Interest Products.
Elan does not own or control any of the intellectual property associated with the Royalty Interest Products, including the underlying delivery systems. In addition to any patents relevant to the Royalty Interest Products themselves, they are expected to be marketed for use with a delivery system developed by Glaxo. The patents covering the technology underlying or used in connection with the Royalty Interest Products may be challenged, and if successful, a challenge may prevent or decrease future sales of the patented product. Third parties may make claims of infringement against the Royalty Interest Products or other technology used in connection therewith, including delivery systems, or obtain patents that prevent the sale or use of those products, all of which may negatively impact sales and therefore Elan’s revenues.
| 19 |
Three of the Royalty Interest Products are not approved for marketing and are at different stages of development. Any delay in the development or approval of these products could decrease Elan’s expected revenues from the Theravance Transaction.
Three of the Royalty Interest Products ((1) ANORO™ ELLIPTA™, (2) MABA 081 monotherapy, and (3) vilanterol (VI) monotherapy) have not to date received the approval of any regulatory authority, whether in the United States or in any other country. Product candidates must undergo extensive non-clinical and clinical studies as a condition to regulatory approval. Product development and commercialization involve a high degree of risk. Only a small number of development programs result in the commercialization of a product. Success in preclinical work or early stage clinical trials does not ensure that later stage or larger scale clinical trials will be successful. Even if later stage clinical trials are successful, product candidates may not receive marketing approval if regulatory authorities disagree with the product candidate sponsor’s view of the data, or require additional studies.
The commencement and completion of clinical studies for those product candidates may be delayed and programs may be terminated due to many factors, including, but not limited to:
| · | lack of effectiveness of product candidates during clinical studies; |
| · | adverse events, safety issues or side effects relating to the product candidates or their formulation into medicines; |
| · | inability of Glaxo or its collaborators or licensees to manufacture or obtain from third parties materials sufficient for use in non-clinical and clinical studies; |
| · | governmental or regulatory delays and changes in regulatory requirements, policy and guidelines; |
| · | failure of Glaxo to advance product candidates through clinical development; |
| · | delays in patient enrollment and variability in the number and types of patients available for clinical studies; |
| · | difficulty in maintaining contact with patients after treatment, resulting in incomplete data; |
| · | varying regulatory requirements or interpretations of data among the FDA and foreign regulatory authorities; and |
| · | a regional disturbance where patients are enrolled in clinical trials, such as a pandemic, terrorist activities or war, political unrest or a natural disaster. |
If these studies are substantially delayed or fail to prove the safety and effectiveness of the unapproved Royalty Interest Products, these products may not receive regulatory approval and Elan’s revenue prospects will be materially harmed.
Theravance may in the future become insolvent or bankrupt or otherwise cease its activities, which may jeopardize Elan’s rights and interests under the Theravance Transaction.
Under the Theravance Royalty Participation Agreement, Theravance undertakes to assign a 21% beneficial interest in its entitlement to receive royalty payments from Glaxo for the Royalty Interest Products to an English limited liability partnership that will be a subsidiary of Theravance. An English law governed trust will be declared immediately prior to the closing of the Theravance Transaction over the royalty payments earned by the Theravance subsidiary when, as and if received, and the beneficial interest in such English law governed trust will be sold to Elan at the close of the Theravance Transaction. It is expected that, after closing of the Theravance Transaction, Theravance will direct any future amounts payable to Elan under the Theravance Royalty Participation Agreement to be made directly to a bank account controlled by the trust. However, if Theravance were to become insolvent or bankrupt, there is a risk that a court may include the royalty payments from Glaxo as assets of Theravance available to its creditors. If a court were to do so, Elan may not receive its interest in any royalty payments at the time or in the amounts foreseen by the royalty participation agreement or at all.
Risks Related to the AOP Transaction
Available financial information related to AOP is subject to significant limitations.
AOP is based in Austria and is currently a privately owned company. As a result, the available financial information regarding AOP is subject to significant limitations. In particular, EBITDA data presented in Appendix I of this Circular (and the financial information on which Elan has based its investment
| 20 |
decision in agreeing to purchase AOP) and the financial information extracted from the 2011 AOP Financial Statements, included in Appendix I have been prepared under Austrian GAAP without reconciliation to IFRS or U.S. GAAP and are, thus, not prepared on the same basis as Elan’s financial information. Conversion of this information may result in changes. In addition, AOP’s financial information at, and in respect of the year ended, 31 December 2012 has not yet been audited, and audit of that information could result in significant changes. The financial information extracted from the 2011 AOP Financial Statements present the entire AOP business and do not separately carve out the estimated amounts attributable to AOP’s generics business which is not being acquired. The EBITDA reported elsewhere in this Circular excludes approximately e6.7 million (approximately $8.5 million) of costs associated with the AOP current generics products business, which is not being acquired. In addition, the 2011 AOP Financial Statements have been prepared in German and translated to English using an external translator. Further, AOP has not been subject to auditor independence requirements that would apply to a U.S. reporting issuer, and its auditors would not be considered independent under those standards.
Finally, AOP’s historical financial information may not be representative of its results following the AOP Acquisition Transaction due to its conversion from a privately owned company to a portion of the operations of a publicly owned company. In particular, compensation expense may increase as a result of revisions to compensation arrangements with AOP’s chief executive officer as he ceases to own AOP.
If the AOP Transaction disrupts AOP’s operations or results in the loss of key employees, or if we fail to coordinate those operations effectively with our existing operations, our business and results of operations could be negatively affected.
AOP is currently a privately owned company. It is possible that the AOP Transaction and the integration of AOP with Elan’s other operations could result in the loss of key employees, errors or delays in systems implementation, the disruption of our ongoing business or the acquired business or inconsistencies in standards, controls, procedures and policies that could adversely affect our ability to maintain relationships with customers and employees or to achieve the anticipated benefits of the AOP Transaction. If any key employees, particularly AOP’s founder and chief executive officer, left, the resulting loss of access to critical knowledge, abilities and ties to research-oriented physicians could materially and adversely affect the value of AOP.
The AOP Transaction may not complete.
Completion of the AOP Transaction is subject to a number of conditions precedent, including (i) the approval of Shareholders, (ii) that there have been no material adverse events between signing of the AOP Share Purchase Agreement and completion of the AOP Transaction, (iii) the obtaining of third-party consents and (iv) delivery of audited financial statements for AOP for the year ended 31 December 2012. The AOP Transaction is subject to termination if these conditions are not fulfilled by 1 August 2013. No assurance can be given that the closing conditions for the AOP Transaction will be satisfied or whether any of the conditions under which the purchase agreement for the AOP Transaction may be terminated will not occur. As a result, the AOP Transaction may not be completed on a timely basis, or at all.
If closing does not occur by 1 August 2013 due to a failure by Elan to obtain Shareholder approval, Elan will be required to pay a break fee of E5.0 million (approximately $6 million).
A failure to complete the AOP Transaction could also result in claims for damages, negative publicity and a negative impression of Elan in the investment community. The occurrence of any of these events could materially and adversely affect Elan.
AOP’s revenues are substantially dependent on a limited number of products.
Sales of five products represent approximately 97% of AOP’s revenues and sales of two of those, Remodulin® and Thromboreductin®, represent approximately 93% of AOP’s revenues. Any negative developments relating to those products, such as safety, efficacy or reimbursement issues, the introduction or greater acceptance of competing products, or adverse regulatory or legislative developments may reduce revenues from these products and adversely affect Elan’s results of operations. In addition, as various sources of exclusivity expire in relation to relevant AOP products (including Thromboreductin® in 2014), revenues for the relevant products may fall significantly. There can be no assurance that AOP will successfully develop new products in time to replace such revenues.
| 21 |
If AOP fails to fulfil related obligations, it could lose exclusivity rights in respect of products subject to orphan drug provisions prior to scheduled expiration of those rights.
Grants of exclusive rights to commercialization of a given drug and indication combination under orphan drug provisions are subject to the on-going fulfilment of related obligations by the holder of the rights. If AOP fails to meet such obligations imposed upon AOP by its orphan drug marketing authorization, it could lose exclusivity rights in respect of products subject to orphan drug provisions prior to scheduled expiration of those rights.
Risks Related to the ELND005 Transaction
The ELND005 Transaction may not complete.
On 19 May 2013, Elan, through its wholly owned subsidiary, EPIL, entered into the ELND005 Transaction Agreement, pursuant to which Elan agreed to effect the ELND005 Transaction to transfer the intellectual property and other assets related to ELND005 to a newly-formed company, Speranza Biopharma. The ELND005 Transaction is subject to conditions and is described in more detail in Part 2 of this document. No assurance can be given that the closing conditions for the ELND005 Transaction will be satisfied on a timely basis, if at all. The ELND005 Transaction may not be completed. If the ELND005 Transaction is not completed, Elan would remain liable for the significant expenses incurred related to the ELND005 Transaction and would remain directly liable for the ongoing expenses associated with the development and commercialization of the ELND005 compound.
Following the ELND005 Transaction, Elan will continue to have an indirect economic interest in, and potential funding obligations for the ELND005 Business.
If the ELND005 Transaction is consummated, Elan will continue to have an indirect economic interest in, and potential funding obligations of up to $7.8 million for, the ELND005 Business, but will not control the ELND005 Business. Elan will be dependent for any returns on its initial funding of Speranza Biopharma, including repayment of its loans, and any future royalties from sales of ELND005 upon the efforts of Nerano to continue to develop and commercialize the ELND005 compound.
There is limited clinical data showing that ELND005 would be effective in any indication and thus, there can be no assurance that ELND005 can be successfully developed or commercialized. Even if ELND005 is successfully developed and commercialized, any negative developments relating to ELND005, such as safety, efficacy or reimbursement issues, the introduction or greater acceptance of competing products, or adverse regulatory or legislative developments, may reduce the amount of any royalties to which Elan may be entitled pursuant to the ELND005 Transaction. Similarly, if ELND005 does receive approval for commercialization, many factors could limit any future revenues and royalties due to Elan, including third party royalties, restrictions on use and safety warnings in product labels and incidence of side effects.
General Transactions
Rejection of certain of the Transactions (other than the Share Repurchase Program) by Shareholders will result in break fees being payable by Elan and rejection of any of the Transactions (other than the Share Repurchase Program) will result in the proceeds of the Bond Offer being returned to the 2021 Note holders with accrued and unpaid interest on the 2021 Notes
Under the terms of the Theravance Royalty Participation Agreement, if the Shareholders do not approve the Theravance Transaction a break fee of $10 million will be payable to Theravance. Under the terms of the AOP Share Purchase Agreement, if closing does not occur by 1 August 2013, due to a failure by Elan to obtain Shareholder approval a break fee of €5 million will also be payable to Dr. Widman. In addition, though the Bond Offer is not expressly subject to Shareholder approval, in the event that any of the Transactions (other than the Share Repurchase Program) are not approved by Shareholders, the escrow release conditions will not have been fulfilled and accordingly, the proceeds of the Bond Offer will be returned to the 2021 Note holders with accrued and unpaid interest on the 2021 Notes of approximately $3 million.
| 22 |
PART 6
FINANCIAL IMPACT OF THE TRANSACTIONS
| 1. | Financial Impact of the Transactions |
The Theravance Transaction
Under the terms of the Theravance Royalty Participation Agreement, Elan will make a one-time cash payment of $1.0 billion to Theravance in exchange for a 21% participation interest in the potential future royalty payments from the Royalty Interest Products earned by Theravance when, as and if received.
The accounting treatment of the royalty participation interest to be acquired by Elan in the Theravance Transaction depends on whether the underlying program has been approved for marketing. At closing, the Company will capitalize an asset equal to the portion of the purchase price allocated to the approved drug, BREO™ ELLIPTA™. The unapproved programs will be accounted for as in-process R&D and the Company will take a one-time charge equal to the purchase price allocated to those programs. The Company currently anticipates that the in-process R&D charge related to the Theravance Transaction is expected to be more than half the purchase price of the acquired royalty participation interest.
If Elan Shareholder approval is not obtained, Elan breaches any of its undertakings relating to the holding of the shareholder meeting, or certain other conditions are not met (including if the closing of the Theravance Transaction is prevented by a law or governmental order), then Elan will be required to pay Theravance a $10.0 million break fee.
ELND005 Transaction
Elan expects to recognize a charge of more than of $60 million upon the close of the ELND005 Transaction, related to the excess of the $63.0 million Elan contribution and the $7.0 million ten-year interest free loan over the initial fair value of Elan’s 18% equity stake in Speranza.
Assuming the completion of the ELND005 Transaction by 1 July 2013, Elan expects aggregate SG&A and R&D expenses to reduce by approximately $35 million to $45 million as a result.
The AOP Transaction
AOP’s EBITDA was €17.6 million (approximately $23 million) (preliminary and unaudited) for the year ended 31 December 2012 and €15.7 million (approximately $20 million) for the year ended 31 December 2011, in both cases excluding estimated amounts attributable to AOP’s current generic products business, which is not being acquired.
AOP’s five most significant products, by revenue, for the year ended December 31, 2012, were:
| Name | Indication Used | Subject to License | |||
| Remodulin® | Pulmonary arterial hypertension | Yes | |||
| Thromboreductin® | Essential thrombocythemia | No | |||
| Tetmodis® | Huntington’s disease | Yes | |||
| Busilvex® | Conditioning before stem cell transplantation | Yes | |||
| Canemes® | Chemotherapy-induced nausea & vomiting (CINV) | Yes |
Sales of these five products represented approximately 97% of AOP’s revenues of €59.0 million (approximately $76 million) (preliminary and unaudited) in the year ended 31 December 2012 and sales of two of those, Remodulin® and Thromboreductin®, represented approximately 93% of AOP’s revenues in that year.
The fair value of the consideration for the AOP Transaction will include an estimate of the fair value of the milestone payments at the close of the transaction. Elan expects that the total fair value of the consideration will be in excess of €380 million (approximately $488 million). The excess, if any, of the fair value of the consideration paid over the fair value of the identifiable assets acquired less the fair value of the liabilities assumed will be recognized as goodwill.
If closing does not occur by 1 August 2013 due to a failure by Elan to obtain Shareholder approval, Elan will be required to pay to Dr. Widmann a break fee of €5 million (approximately $6 million) as reimbursement for his costs and expenses in connection with the AOP Transaction.
| 23 |
PART 7
ADDITIONAL INFORMATION
| 1. | Material Contracts |
| 1.1. | AOP Transaction Documents |
AOP Share Purchase Agreement
On 19 May 2013, the Company and SASR, a newly formed subsidiary of the Company, entered into a share purchase agreement for its pending purchase of AOP Orphan Pharmaceuticals Aktiengesellschaft. AOP researches, develops, produces and distributes innovative drugs and administration methods for rare diseases. AOP focuses on haematology and oncology, cardiology and pulmonology, neurology and psychiatry, and metabolic and genetic disorders.
The consideration payable under the AOP Share Purchase Agreement consists of an initial purchase price of €263.5 million (approximately $339 million), composed of €175.7 million (approximately $226 million) in cash and €87.8 million (approximately $113 million) of Elan Ordinary Shares, each deliverable at closing. There are also potential post-close milestone payments of up to €270 million (approximately $347 million) that will become payable by Elan on the advancement of certain clinical programs to filing and regulatory acceptance.
The AOP Share Purchase Agreement contains customary representations and warranties from the seller, Dr. Widmann, about the business and some limited representations and warranties from Elan. Dr. Widmann also undertakes not to compete with AOP for three years from closing.
Completion of the AOP Transaction is subject to a number of conditions precedent, including (i) the approval of Elan Shareholders, (ii) that there have been no material adverse events between signing of the AOP Share Purchase Agreement and completion of the AOP Transaction, (iii) the obtaining of certain third-party consents and (iv) delivery of audited financial statements for AOP for the year ended 31 December 2012. The AOP Transaction is subject to termination by joint action of the purchaser and seller if these conditions are not fulfilled by 1 August 2013. These conditions, with the exception of number (i) can be waived by Elan in its sole discretion.
If closing does not occur by 1 August 2013 due to a failure by Elan to obtain Shareholder approval, Elan will be required to pay to Dr. Widmann a break fee of €5.0 million (approximately $6 million) as reimbursement for his costs and expenses in connection with the transaction. Under the terms of the AOP Transaction, each party is responsible for its own costs and expenses. The agreement is governed by Austrian law.
The Company has guaranteed the obligations of SASR arising under the AOP Share Purchase Agreement.
| 1.2. | Theravance Transaction Documents |
Theravance Royalty Participation Agreement
On 12 May 2013, the Company entered into Theravance Royalty Participation Agreement pursuant to which it will purchase a participation interest in potential future royalty payments related to the Royalty Interest Products, each of which is a treatment or a potential treatment for COPD or asthma.
Under the terms of the Theravance Royalty Participation Agreement, the Company will make a one-time cash payment of $1.0 billion to Theravance in exchange for a 21% participation interest in the potential future royalty payments from the Royalty Interest Products earned by Theravance when, as and if received.
The Theravance Transaction is subject to the satisfaction or waiver of certain conditions, including (i) the approval of Elan Shareholders, (ii) the absence of laws or governmental orders that prevent the closing of the Theravance Transaction, (iii) the establishment of a Theravance subsidiary that will receive the covered royalty payments and (iv) other customary conditions.
If Shareholder approval is not obtained, or the Company breaches any of its undertakings relating to the holding of the EGM, or certain other conditions are not met (including if the
| 24 |
closing is prevented by a law or governmental order), then Elan will be required to pay Theravance a $10.0 million break fee.
If Shareholder approval is obtained and the other closing conditions are met or waived, the Theravance Transaction is expected to close shortly after the EGM.
| 1.3. | ELND005 Transaction Documents |
ELND005 Transaction Agreement
On 19 May 2013, EPIL and Nerano entered into the ELND005 Transaction Agreement to govern the proposed ELND005 Transaction. Pursuant to that agreement, the parties agreed that completion of the ELND005 Transaction was conditional on Elan Shareholder approval having been obtained (or the Offer Period having ended), the parties agreeing the final form of certain ancillary documentation, all required governmental or regulatory approvals having being obtained and there being no injunction or order restraining the completion of the transaction. EPIL agreed that it would use all commercially reasonable endeavours to ensure that the Elan Shareholder approval would be obtained (or the Offer Period ended) by 28 June 2013. The parties agreed that they would use all commercially reasonable endeavours to ensure that they agreed the final form of certain ancillary documentation related to the ELND005 Transaction for execution and delivery on completion. Pursuant to the ELND005 Transaction Agreement, on completion, the parties or certain of their affiliates will enter into the following key agreements to give effect to the ELND005 Transaction on substantially the forms agreed at signing:
| 1.3.1. | ELND005 Business Transfer Agreement |
Pursuant to the ELND005 Business Transfer Agreement, EPIL will transfer the assets relating to the ELND005 Business to Speranza Biopharma. The consideration for the sale will be satisfied by the payment of US$1.00 by Speranza Biopharma to EPIL and its assumption of the burden of the contracts relating to the ELND005 Business. EPIL will remain liable for all liabilities and claims relating to the ELND005 Business arising prior to completion. Speranza Biopharma will be liable for all liabilities and claims relating to the Speranza Business arising after completion. Speranza Biopharma will indemnify EPIL against all losses that EPIL incurs as a result of a failure by Speranza Biopharma to perform any of the obligations assumed by it relating to the Speranza Business. EPIL will be obliged to discharge all of its obligations in relation to the Speranza employees transferring to Speranza Biopharma and to indemnify Speranza Biopharma for certain claims which may be made by any of the Speranza employees and EPIL will remain responsible for up to $3.0 million in retention bonuses or severance payments made by Speranza within 18 months of completion.
| 1.3.2. | Speranza Share Purchase Agreement |
Pursuant to the Speranza Share Purchase Agreement EPIL will transfer its 100% shareholding in Speranza Biopharma to Speranza Therapeutics for $1.00. EPIL will provide customary warranties to Speranza Therapeutics in relation to the ELND005 Business. Claims for a breach of the warranties provided by EPIL will be limited such that Speranza Therapeutics must notify EPIL of any claim for breach of warranty within eighteen months from completion and the total liability in respect of all warranty claims is limited to $15,000,000. Speranza Therapeutics cannot succeed for a warranty claim unless the amount recoverable in respect of the warranty claims exceeds $250,000 in the aggregate. EPIL has also provided indemnities to Speranza Therapeutics in respect of certain contractual and employee matters which are not subject to limitations.
| 1.3.3. | Speranza Shareholders’ Agreement |
Following completion, Elan will hold 18% of Speranza Therapeutics, 62% will be held by Nerano, and the remaining 20% will be issued to senior management who work on the ELND005 programs and who will transfer from Elan to Speranza Biopharma. On completion, the parties shall enter into the Speranza Shareholders’ Agreement and shall agree a business purpose plan for the Speranza Group within 90 days, relating to the initial clinical goals for the Speranza Business. Speranza will operate the Speranza Business in accordance with the business purpose plan which will continue in effect until the end of 2015. In order to provide funding for the development of the Speranza Business (i) Elan
| 25 |
will make a capital contribution of $63 million to Speranza Biopharma; (ii) Elan will provide Speranza Biopharma with a $7 million, ten year interest-free loan; and (iii) Nerano will provide Speranza Biopharma with a ten year $20 million interest-free loan. If the Speranza Group requires additional funding to implement the business plan, it will have the ability to require Elan and Nerano to contribute a further $10.0 million to the Speranza Group to complete any clinical activities or obtain any regulatory approvals necessary under the business plan, with Elan to contribute 78% of this amount and Nerano to contribute the remaining 22%.
If either Nerano or Elan believes that the ELND005 Business is no longer commercially viable then it can notify the other that a termination event has occurred. If it is terminated within 15 months of completion, then the two loans provided by Elan and Nerano will be repaid in full and Elan will receive the balance. If it is terminated on a date that is 15 months after completion, then the parties will receive a proportionate return of the balance of the initial funding, with Nerano receiving two-ninths and Elan receiving seven-ninths.
Elan has customary minority shareholder protection rights under the agreement to protect its investment. Elan also has anti-dilution rights whereby its shareholding cannot be diluted below 18% without its consent. The agreement contains a restriction on the parties’ ability to transfer their shares and neither Nerano nor Elan are permitted to transfer their shares, without the other’s consent, until either the later of the end of 2015 or when the initial funding is expended. There are similar transfer restrictions on the Speranza employees with respect to their shareholding. The shareholders’ agreement also contains customary offer round, drag and tag provisions.
| 1.3.4. | Speranza Royalty and Option Agreement |
On completion, Elan will be granted a 3% royalty on worldwide sales of any Speranza products and an option to commercialize Speranza products in certain territories pursuant to the royalty and option agreement. Elan will also receive two once-off $200,000,000 milestone payments on the occurrence of each of the following events: (i) any sale, transfer or other disposal of the Speranza Group or all or substantially all of its business (which milestone will be subject to a deduction for all future milestone payments paid to Transition by Speranza, the maximum amount of which would be $93 million); and (ii) attainment by the Speranza Group of worldwide annual net income of US$1,500,000,000.
Speranza Therapeutics may not commercialize in those option territories for which Elan has exercised its option. Where Elan does take up the option for only some of the option territories those countries that are not chosen will remain part of the pool of countries where Speranza may commercialize the Speranza product. When the option is exercised the parties will execute a license agreement which will provide for an exclusive license for Elan in relation to the commercialization of the Speranza products and any patents and know-how regarding same in the selected option territories.
| 1.4. | Bond Documents |
| 1.4.1. | Purchase agreement |
On 23 May 2013, Elan entered into a purchase agreement with the Subsidiary Note Guarantors, the Issuers, and the Initial Purchasers. Under the terms of the Purchase Agreement the Issuers agreed to sell to the Initial Purchasers on or about 31 May 2013 (being the closing date) US$850,000,000 principal amount of 6.25% senior notes due 2021 and the Initial Purchasers agreed to purchase the Securities from the Issuers at the purchase price upon the basis of the representations and warranties provided, and subject to the conditions set out, in the Purchase Agreement. Elan, the Issuers and the Subsidiary Note Guarantors have agreed to indemnify the Initial Purchasers against certain liabilities, including liabilities under the US Securities Act of 1933, as amended, or contribute to payments that the Initial Purchasers may be required to make in respect of indemnification.
The Securities will be issued pursuant to an Indenture, to be dated as of the 31 May 2013 among the Issuers, Elan, the Subsidiary Note Guarantors and The Bank of New York Mellon, London Branch, as trustee, and will be unconditionally guaranteed on a joint and several and unsecured basis by Elan and the Subsidiary Note Guarantors.
| 26 |
| 2. | Interests, Short Positions and Dealings in Elan Securities |
| 2.1. | Terms defined in paragraph 5.1 to 5.1.14 (inclusive) of Appendix I of the Defence Document have the same meaning in this paragraph 2, save that in this paragraph 2 the “disclosure period” means the period commencing on 14 May 2013 and ended on the Latest Practicable Date and “disclosure date” means 14 May 2013. |
| 2.2. | The following dealings took place during the dates below which were not included in paragraph 5.25 of Appendix I of the Defence Document but were publicly disclosed for the purposes of Rule 8.1 of the Irish Takeover Rules: |
| Number of | ||||||||||||
| Nature of | relevant Elan | Price/Price | ||||||||||
| Name | Date of dealing | transaction | securities | Range | ||||||||
Morgan Stanley Securities Ltd | 16 April 2013 | Acquisition of Ordinary Shares | 26,751 | $ | 11.72 | |||||||
Morgan Stanley Securities Ltd | 22 April 2013 | Sale of Ordinary Shares | 9,200 | € | 8.90 | |||||||
Morgan Stanley Securities Ltd | 28 April 2013 | Acquisition of Ordinary Shares | 9,200 | € | 9.02 | |||||||
| 2.3. | During the disclosure period, the dealings in relevant Elan securities by Morgan Stanley or persons (other than exempt market makers or exempt fund managers) controlling or under the same control as Morgan Stanley were as follows: |
| Number of | ||||||||||||
| Nature of | relevant Elan | Price/Price | ||||||||||
| Name | Date of dealing | transaction | securities | Range | ||||||||
Morgan Stanley Securities Ltd | 14 May 2013 | Acquisition of Ordinary Shares | 559 | € | 8.80 | |||||||
| 2.4. | During the disclosure period, there has been no change to the information in paragraphs 5.2 – 5.24 (inclusive) and 5.26 – 5.34 (inclusive) of Appendix I of the Defence Document. |
| 2.5. | To the best of Elan’s knowledge, the Directors, executive officers or affiliates do not intend, in respect of their own beneficial holdings of securities, to accept the Revised Offer. |
| 3. | Interests, Short Positions and Dealings in Relevant Royalty Pharma Securities |
| 3.1. | During the disclosure period, there has been no change to the information in paragraphs 6.1 – 6.6 (inclusive) of Appendix 1 of the Defence Document. |
| 4. | Consents |
| 4.1. | Davy Corporate Finance, has given and not withdrawn its written consent to the issue of this document including references to its name in the form and context which they appear and to the inclusion herein of the Financial Advisors’ report on the Profit Forecast set out in Part 8. |
| 4.2. | Morgan Stanley, has given and not withdrawn its written consent to the issue of this document including references to its name in the form and context which they appear and to the inclusion herein of the Financial Advisors’ report on the Profit Forecast set out in Part 8. |
| 4.3. | Ondra, has given and not withdrawn its written consent to the issue of this document including references to its name in the form and context which they appear and to the inclusion herein of the Financial Advisors’ report on the Profit Forecast set out in Part 8. |
| 4.4. | Citi, has given and not withdrawn its written consent to the issue of this document including references to its name in the form and context which they appear and to the inclusion herein of the Financial Advisors’ report on the Profit Forecast set out in Part 8. |
| 27 |
| 4.5. | KPMG, has given and not withdrawn its written consent to the issue of this document including references to its name in the form and context which they appear and to the inclusion herein of the Accountant’s Report on the Profit Forecast set out in Part 8. |
| 4.6. | The Company published unaudited financial information for the three month period ended 31 March 2013 in an announcement dated 24 April 2013 (“Quarterly Financial Information”). In accordance with the requirements of Rule 28.6(d) of the Irish Takeover Rules, the Financial Advisors and KPMG provided a report on the Quarterly Financial Information in Appendix II of the Defence Document. For the purposes of Rule 28.5 of the Irish Takeover Rules only, the Directors confirm that the Quarterly Financial Information remains valid. The Financial Advisers and KPMG also confirm that they have no objection to their reports on the Quarterly Financial Information continuing to apply. These confirmations are provided solely in connection with Rule 28.5 of the Irish Takeover Rules and for no other purpose. |
| 5. | Irish Takeover Rules |
The following information is included as required by Rule 21.1(b) of the Irish Takeover Rules.
On 20 May 2013, Royalty Pharma issued an announcement under the Irish Takeover Rules pursuant to which it revised its Offer to acquire the entire issued and to be issued share capital of the Company. On 23 May 2013, Royalty Pharma issued the Revised Offer Document proposing an offer price of US$12.50 for each ADS and Ordinary Share. In addition, the Revised Offer Document provides that on the Extended Closing Date (as such term is defined in the Revised Offer Document), Royalty Pharma will waive down the acceptance threshold for the Revised Offer from 90% to 50% plus one Elan Share subject to, amongst other things, the Board recommending that the Theravance Transaction is approved by Shareholders at the EGM.
As announced by the Company on 23 May 2013, the Board reiterated that it unanimously, and without reservation, recommends that you reject the Revised Offer by taking no action.
As the Company is in an Offer Period, for the purposes of the Irish Takeover Rules, the Company must obtain Shareholder approval for certain corporate actions including the Transactions.
| 6. | Documents on Display |
| 6.1. | In addition to the display documents identified in the Defence Document, copies of the following documents are available for inspection during normal business hours on any Business Day at the offices of A&L Goodbody, IFSC, North Wall Quay, Dublin 1, Ireland for as long as the Offer remains open for acceptance: |
| 6.1.1. | this document; |
| 6.1.2. | the material contracts identified at paragraph 1 of this Part 7; |
| 6.1.3. | the report of the Financial Advisors and the Profit Forecast and the consents of KPMG and the Financial Advisors, to the inclusion of the report in the form and context in which they are included in this Circular; and |
| 6.1.4. | copies of any report, letter, valuation, announcement or other document, any part of which is exhibited or referred to in any document issued by, or on behalf of, the Company during the course of the Offer and pursuant to Rule 26(b)(iv) of the Irish Takeover Rules. |
| 7. | No Material Change |
Other than (i) the Transactions and the Bond Offer which are described in this document, (ii) the material contracts set forth in paragraph 1 of this Part 7 and (iii) the interests and dealings information set forth in paragraph 2 of this Part 7, the Directors of Elan are not aware of any material change in information previously published by Elan, or on its behalf.
| 8. | Responsibility Statement |
The Directors of Elan accept responsibility for the information contained in this document. To the best of their knowledge and belief (having taken all reasonable care to ensure such is the case), the information contained in this document is in accordance with the facts and does not omit anything likely to affect the import of such information.
| 28 |
| 9. | Sources and Bases of Calculation |
The relevant bases of calculation and sources of information are set out below. Where such information is repeated in the document the underlying sources and bases are not repeated.
Unless otherwise stated in this document:
| 9.1. | All financial information relating to Elan has been extracted or derived (without any adjustments) from either the annual reports and accounts of Elan, or other information made publicly available by Elan. |
| 9.2. | All information relating to the Theravance, AOP and ELND005 Transactions, the Bond Offer, the Share Repurchase Program and the NewBridge Transaction has been extracted or derived from publicly available information and, where the context so requires, the internal records of Elan. |
| 9.3. | All information regarding the Offer is sourced from the Royalty Pharma Offer Document dated 2 May 2013, the Revised Offer Announcement, dated 20 May 2013, the Revised Offer Document dated 23 May 2013 and any other material made publicly available by Royalty Pharma. |
| 9.4. | Any references to announcements and dates of announcements have been taken from public materials and press releases made by Elan and Royalty Pharma. |
| 9.5. | Values stated throughout this document have been rounded and are stated to the given number of decimal places. |
| 9.6. | The Profit Forecast is based on internal financial information of the Company and has been reported on in accordance with the Irish Takeover Rules. |
| 9.7. | Where amounts are expressed in euro and a US dollar equivalent has been provided, all such equivalents are calculated using the average U.S. Federal Reserve non buying rate for the relevant period. |
| 9.8. | Reference in the Chairman’s letter to the data relating to the restructuring of the Tysabri collaboration with Biogen Idec has been sourced from the Company’s August 2012 investor presentation, fourth quarter 2012 earnings press release, and 20 May 2013 investor presentation, slide 4. |
| 9.9. | Reference in the Chairman’s letter to global in-market net sales of Tysabri exceeded $1.6 billion last year has been sourced from the Company’s 20-F filing, 2012, page 20. |
| 9.10. | Reference in the Chairman’s letter to the growth in the respiratory market has been taken from Wall Street research, ‘The Global Use of Medicines’: Outlook Through 2015, IMS Institute for Healthcare Information. |
| 9.11. | Reference in the Chairman’s letter to the quote by one leading analyst team which states “we find the Theravance Transaction impressive for: (1) immediate EPS accretion, (2) immediate return of capital to shareholders, (3) virtually 100% pure net profit, (4) long duration assets, (5) partnered with premier respiratory companies, (6) no operational risk, and (7) positive NPU” has been taken from the analysis issued by Jefferies LLC, company update note on USA healthcare pharmaceutical dated 13 May 2013. |
| 9.12. | Reference to the $80 billion Eastern and Central European pharma market, has been taken from ‘The World Pharmaceuticals Markets Factbook 2012’, Espicom Business Intelligence. |
| 9.13. | Reference to the GlaxoSmithKline/Theravance respiratory program having more than 40% share of the market, has been taken from the Bank of America Merrill Lynch company update on GlaxoSmithKline, ‘Respiratory pivots approaching’ dated 28 February 2013. |
| 29 |
PART 8
PROFIT FORECAST AND REPORT OF KPMG AND THE FINANCIAL ADVISORS
| 1. | BACKGROUND |
In the absence of any unforeseen circumstances and on the basis of preparation and principal assumptions set out below the Directors make the following forecast of profitability for the year ending 31 December 2013 (the Profit Forecast):
| 1.1. | Elan expects Tysabri in-market sales to increase by approximately 15% in 2013 over the $1.6 billion recorded in 2012. |
| 1.2. | Excluding Tysabri, and ELND005, from completion of the ELND005 Transaction, aggregate SG&A and R&D expenses are expected to be in the range of $130 million to $150 million. |
| 1.3. | Elan expects to contribute approximately $60-$80 million to Janssen Alzheimer Immunotherapy during 2013. |
This profit forecast was originally made by the Company as part of its guidance issued in its statement of fourth quarter and full year 2012 financial results published on 6 February 2013 and is subsequently updated to take account of the effect of the ELND005 Transaction based on the assumptions below.
As the Profit Forecast constitutes a profit forecast for the purposes of the Irish Takeover Rules, the Irish Takeover Rules requires that the Profit Forecast be reported on by Elan’s reporting accountants and financial advisors in accordance with Rule 28 of the Irish Takeover Rules.
| 2. | THE ASSUMPTIONS |
The principal assumptions on which the Profit Forecast are based are set out below.
| 2.1. | There will be no material change in the ownership and control of Elan prior to 31 December 2013. |
| 2.2. | Savings to aggregate R&D and SG&A spend in the range of $35 million to $45 million are expected post completion of the ELND005 Transaction, based on the Elan estimate of full year spend for ELND005 and assuming the completion of the ELND005 Transaction by 1 July 2013. |
| 2.3. | Save for the Theravance Transaction, which was announced by Elan on 13 May 2013 and which, on completion, is not expected to impact the validity of the Profit Forecast, save for the ELND005 Transaction which was announced by Elan on 20 May 2013 and the expected impact of which has been reflected in the updated Profit Forecast, there will be no additional material acquisitions or disposals by Elan during the profit forecast period. In respect of the AOP Transaction, which was announced by Elan on 20 May 2013, no assumption has been made in relation to completion timeline or impact on the Profit Forecast. |
| 2.4. | There will be no material restructuring activities or reduction in work force announced by Elan prior to 31 December 2013. |
| 2.5. | There will be no material adverse change in economic conditions in the markets in which the Elan Group operates. |
| 2.6. | Rates of interest, taxation, inflation and foreign currency exchange rates will not change significantly during the forecast profit period. |
| 2.7. | Any changes in relevant legislation, healthcare reimbursement and reform, governmental policy or other regulatory requirements will not materially affect the results of the Elan Group. |
| 2.8. | There will be no significant adverse impact on Tysabri in-market sales from new competing products in the multiple sclerosis market. |
| 2.9. | There will be no significant delay in the manufacture or supply of Tysabri or in the supply of raw materials for Tysabri which would adversely affect Tysabri in-market sales. |
| 2.10. | There will be no material adverse change to financial forecasts for Janssen Alzheimer Immunotherapy during the forecast period. |
| 2.11. | There will be no material unprovided costs arising from litigation, legislative or regulatory requirements of which the Elan Group is not yet aware. |
| 30 |
| 3. | ACCOUNTANT’S REPORT ON PROFIT FORECAST |
The Directors
Elan Corporation, plc
Treasury Building
Lower Grand Canal Street
Dublin 2
Ireland
| Davy Corporate Finance | Citigroup Global Markets Limited |
| Davy House | 33 Canada Square |
| 49 Dawson Street | Canary Wharf |
| Dublin 2 | London E14 5LB |
| Ireland | United Kingdom |
| Morgan Stanley & Co. International plc | Ondra LLP |
| 25 Cabot Square | 125 Old Broad Street |
| Canary Wharf | London EC2N 1AR |
| London E14 4QA | United Kingdom |
| United Kingdom |
27 May 2013
Dear Sir or Madam:
Accountant’s report on profit forecast
Elan Corporation, plc
We report on the profit forecast comprising a forecast of Tysabri in-market sales for 2013, aggregate SG&A and R&D expenses (excluding Tysabri and assuming the completion of the ELND0005 transaction on or prior to 1 July 2013) for 2013 and the expected contribution to Janssen Alzheimer Immunotherapy to be incurred by Elan Corporation, plc (the ‘Company’) and its subsidiary undertakings (the ‘Group’) for the year ending 31 December 2013 (the ‘Profit Forecast’). The Profit Forecast, and the material assumptions upon which it is based, are set out in Part 8 of the Shareholders Circular (the ‘Document’) dated 27 May 2013. This report is required by Rule 28.3(a) of the Irish Takeover Panel Act, 1997, Takeover Rules, 2007 (the ‘Takeover Rules’) and is given for the purpose of complying with that rule and for no other purpose. Accordingly, we assume no responsibility in respect of this report to the offeror or to any person connected to, or acting in concert with, the offeror, or to any other person who is seeking or may in future seek to acquire control of the Company (an ‘Alternative Offeror’) or to any person connected to, or acting in concert with, an Alternative Offeror.
Responsibilities
It is the responsibility of the directors of the Company (the ‘Directors’) to prepare the Profit Forecast in accordance with the requirements of the Takeover Rules.
It is our responsibility to form an opinion as required by the Takeover Rules as to the proper compilation of the profit forecast and to report that opinion to you.
Save for any responsibility which we may have to those persons to whom this report is expressly addressed, to the fullest extent permitted by law we do not assume any responsibility and will not accept any liability to any other person for any loss suffered by any such other person as a result of, arising out of, or in accordance with this report or our statement, required by and given solely for the purposes of complying with Rule 28.4 of the Takeover Rules, consenting to its inclusion in the Document.
Basis of Preparation of the Profit Forecast
The Profit Forecast has been prepared on the basis stated on page 30 of the Document and is based on the unaudited interim financial results for the three months ended 31 March 2013, and a forecast for the nine months to 31 December 2013. The Profit Forecast is required to be presented on a basis consistent with the accounting policies of the Group.
| 31 |
Basis of opinion
We conducted our work in accordance with Standards for Investment Reporting issued by the Auditing Practices Board of the United Kingdom and Ireland. Our work included evaluating the basis on which the historical financial information included in the Profit Forecast has been prepared and considering whether the Profit Forecast has been accurately computed based upon the disclosed assumptions and the accounting policies of the Group. Whilst the assumptions upon which the Profit Forecast are based are solely the responsibility of the Directors, we considered whether anything came to our attention to indicate that any of the assumptions adopted by the Directors which, in our opinion, are necessary for a proper understanding of the Profit Forecast have not been disclosed or if any material assumption made by the Directors appears to us to be unrealistic.
We planned and performed our work so as to obtain the information and explanations we considered necessary in order to provide us with reasonable assurance that the Profit Forecast has been properly compiled on the basis stated.
Since the Profit Forecast and the assumptions on which it is based relate to the future and may therefore be affected by unforeseen events, we can express no opinion as to whether the actual results reported will correspond to those shown in the Profit Forecast and differences may be material.
Our work has not been carried out in accordance with auditing or other standards and practices generally accepted in the United States of America or other jurisdictions and accordingly should not be relied upon as if it had been carried out in accordance with those standards and practices.
Opinion
In our opinion the Profit Forecast, so far as the accounting policies and calculations are concerned, has been properly compiled on the basis of the assumptions made by the Directors and the basis of accounting used is consistent with the accounting policies of the Group.
Yours faithfully
KPMG
Chartered Accountants
Dublin, Ireland
| 32 |
| 4. | REPORT OF THE FINANCIAL ADVISORS |
| Morgan Stanley & Co. | Davy Corporate | Citigroup Global | Ondra LLP |
| International plc | Finance | Markets Limited | 125 Old Broad Street |
| 25 Cabot Square | Davy House, | Citigroup Centre | London EC2N 1AR |
| London | 49 Dawson Street | 33 Canada Square | United Kingdom |
| E14 4AD | Dublin 2, Ireland | Canary Wharf | |
| United Kingdom | London E14 5LB | ||
| United Kingdom | |||
| Registered in England and | Registered in Ireland | Registered in England and | Registered in England and |
| Wales No. 2068222 | No. 127823 | Wales No. 01763297 | Wales No. OC340822 |
| Authorised by the Prudential | Regulated by the Central | Authorised by the Prudential | Regulated by the Financial |
| Regulation Authority and | Bank of Ireland | Regulation Authority and | Conduct Authority |
| regulated by the Financial | regulated by the Financial | ||
| Conduct Authority and the | Conduct Authority | ||
| Prudential Regulation | |||
| Authority |
Board of Directors
Elan Corporation, plc
Treasury Building
Lower Grand Canal Street
Dublin 2
Ireland
27 May 2013
Dear Sirs
Report on the Profit Forecast of Elan Corporation, plc (the “Company”)
We refer to the profit forecast of the Company and its subsidiaries for the 12 months ending 31 December 2013 which provides that:
| 1.1. | Elan expects Tysabri in-market sales to increase by approximately 15% in 2013 over the $1.6 billion recorded in 2012. |
| 1.2. | Excluding Tysabri, and ELND005, from completion of the ELND005 Transaction, as such term is defined in the circular to shareholders dated 27 May 2013 (the “Circular”) aggregate SG&A and R&D expenses are expected to be in the range of $130 million to $150 million. |
| 1.3 | Elan expects to contribute approximately $60-$80 million to Janssen Alzheimer Immunotherapy during 2013 (collectively the “Profit Forecast”). |
The material assumptions upon which the Profit Forecast is based are set out in Part 8 to the Circular.
We have discussed the Profit Forecast and the bases and assumptions on which it is made with you and with KPMG, the Company’s reporting accountants. We have also discussed the accounting policies and bases of calculation for the Profit Forecast with you and with KPMG. We have also considered KPMG’s letter of 27 May 2013 addressed to you and to us on these matters. We have relied upon the accuracy and completeness of all the financial and other information discussed with us and have assumed such accuracy and completeness for the purposes of providing this letter.
On the basis of the foregoing, we consider that the Profit Forecast, for which you, as directors of the Company are solely responsible, has been made with due care and consideration.
This letter is provided to you solely in connection with Rule 28.3(a) and Rule 28.4 of the Irish Takeover Rules and for no other purpose. No person other than the directors of the Company can rely on the contents of this letter and to the fullest extent permitted by law, we exclude all liability to any other person other than to you, the directors of the Company, in respect of this letter or the work undertaken in connection with this letter.
Yours faithfully,
| Morgan Stanley & Co. | Davy Corporate | Citigroup Global | Ondra LLP |
| International plc | Finance | Markets Limited | |
| 33 |
| 5. | QUARTERLY FINANCIAL INFORMATION |
The Financial Advisers and KPMG also confirm that they have no objection to their reports on the Quarterly Financial Information, as set out in Appendix II of the Defence Document, continuing to apply. These confirmations are provided solely in connection with Rule 28.5 of the Irish Takeover Rules and for no other purpose.
| 34 |
PART 9
DEFINITIONS
The following definitions apply throughout this document, unless the context requires otherwise:
| "2011 AOP Financial Statements" | extracts from the audited financial statements of AOP for the year ended 31 December 2011, prepared under Austrian GAAP |
| "2019 Notes" | the $600 million in aggregate principal amount of the then outstanding 6.25% senior notes due 2019 issued in October 2012 by the Company’s wholly-owned subsidiaries, Elan Finance public limited company and Elan Finance Corp |
| "2021 Noteholders" | the holders of 2021 Notes |
| "2021 Notes" | Senior notes due 2021 to be issued pursuant to the Bond Offer |
| "Act" | the Irish Companies Acts 1963-2012 and every statutory extension, modification or re-enactment thereof from time to time in force |
| "ADR(s)" | American depositary receipt(s) representing ADS(s) |
| "ADSs" or "American Depositary Shares" | American depositary share(s), each of which represents one Ordinary Share in Elan |
| "ADS Depositary" or "Depositary" | Citibank, N.A., in its capacity as depositary bank for the ADSs pursuant to the Deposit Agreement |
| "ADS Holders" | holders of ADSs |
| "AOP" or "AOP Orphan" | AOP Orphan Pharmaceuticals Aktiengesellschaft, an Austrian pharmaceutical company specialising in orphan and rare diseases |
| "AOP Resolution" | the ordinary resolution with respect to the approval of the AOP Transaction |
| "AOP Share Purchase Agreement" | share purchase agreement dated 19 May 2013 between Elan, Dr. Rudolf Stefan Widmann and SASR to purchase the entire issued share capital of AOP Orphan |
| "AOP Transaction" | the agreement by the Company to purchase the entire issued share capital of AOP Orphan |
| "Biogen Idec" | Biogen Idec Inc. |
| "Board" or "Directors" | the board of directors of Elan from time to time |
| "Bond Offer" | the offer of the 2021 Notes by Elan Finance plc and Elan Finance Corp. as more particularly described in a preliminary offering memorandum dated 20 May 2013 |
| "Business Days" | a day, not being a Saturday, Sunday or public holiday, on which banks in Dublin and New York are open for business |
| "Central Bank" | the Central Bank of Ireland |
| "Circular" | this document dated 27 May 2013 |
| "Citi" | Citigroup Global Markets Inc. and, or, Citigroup Global Markets Limited, as relevant in the context |
| "Citibank" | Citibank N.A. |
| "COPD" | chronic obstructive pulmonary disease |
| 35 |
| "CREST" | the relevant system (as defined in the Companies Act 1990 (Uncertificated Securities) Regulations 1996 of Ireland (as amended)), enabling title to securities to be evidenced and transferred in dematerialised form operated by CrestCo Limited |
| "Davy" | J&E Davy, trading as Davy, registered office Davy House, 49 Dawson Street, Dublin 2, Ireland |
| "Davy Corporate Finance" | an Irish incorporated unlimited liability company with registered number 127823 and having its registered office at Davy House, 49 Dawson Street, Dublin 2, Ireland |
| "Debt Redemption" | the redemption of all of the $600 million in aggregate principal amount of the 2019 Notes |
| "Defence Document" | the document dated 15 May 2013 issued by the Company to its Shareholders in response to the Royalty Pharma Offer |
| "Deposit Agreement" | means the Amended and Restated Deposit Agreement dated 3 February 2012, among Elan, Citibank and the holders and beneficial owners of ADSs issued thereunder |
| "Depositary" | Citibank, N.A. |
| "Depositary's Notice" | the Depositary’s notice of the EGM to holders of ADSs |
| "EBITDA" | earnings before interest, taxes, depreciation and amortization |
| "EDT" | Elan Drug Technologies |
| "Elan" or the "Company" | Elan Corporation, plc, a public limited company incorporated in Ireland with registered number 30356 |
| "Elan Group" | Elan and its subsidiaries and subsidiary undertakings and where the context so requires, its associated undertakings |
| "Elan Share(s)" or "Share(s)" | Elan Ordinary Shares and Elan ADSs |
| "ELND005 Business" or "Speranza Business" | the research, development, manufacture and commercialization of ELND005 |
| "ELND005 Business Transfer Agreement" | the business transfer agreement between EPIL and Speranza Biopharma pursuant to which EPIL agrees to transfer all of the assets relating to the ELND005 Business to Speranza Biopharma and to be entered into on completion of the ELND005 Transaction |
| "ELND005 Resolution" | the ordinary resolution with respect to the approval of the ELND005 Transaction |
| "ELND005 Royalty and Option Agreement" or "Speranza Royalty and Option Agreement" | the royalty and option agreement between EPIL and Speranza Biopharma in order to regulate the commercialisation of certain ELND005 products to be entered into on completion of the ELND005 Transaction |
| "ELND005 Share Purchase Agreement" or "Speranza Share Purchase Agreement" | the share purchase agreement between EPIL and Speranza Therapeutics to be entered into on completion of the ELND005 Transaction and pursuant to which EPIL agrees to sell the entire issued share capital of Speranza Biopharma to Speranza Therapeutics |
| "ELDN005 Shareholders Agreement" or "Speranza Shareholders' Agreement" | the subscription and shareholders’ agreement between ES1, Nerano, Speranza Therapeutics and ELND005 management to regulate the parties’ relationship as shareholders of Speranza Therapeutics and to be entered into on completion of the ELND005 Transaction |
| "ELND005 Transaction" | the disposal by the Company of the ELND005 Business |
| 36 |
| "ELND005 Transaction Agreement" | the transaction agreement dated 19 May 2013 between Elan and Nerano to dispose of the ELND005 Business |
| "EPIL" | Elan Pharma International Limited |
| "ES1" | Elan Science One Limited |
| "Escrow Agent" | the Bank of New York Mellon |
| "Escrow Agreement" | an escrow agreement among the Bank of New York Mellon, London Branch, as trustee, the Escrow Agent and the Issuers, pursuant to which the Issuers will deposit the gross proceeds of the Bond Offer (after deduction of the Initial Purchasers discounts), together with additional amounts sufficient to redeem the 2021 Notes in connection with a special mandatory redemption on 21 June 2013 subject to extension until 31 July 2013, if a special mandatory redemption were to occur on such date, into a segregated escrow account |
| "Euro" or "€" | the legal currency of Ireland |
| "Exchange Act" | Securities Exchange Act of 1934, as amended |
| "Extraordinary General Meeting" or "EGM" | the extraordinary general meeting of the Company to be held on 17 June 2013 at 10.00 a.m. (Irish time) at the O’Callaghan Davenport Hotel, 8/10 Merrion Street Lower, Dublin 2, Ireland (and any adjournment thereof) convened by way of the Notice |
| "FDA" | United States Food and Drug Administration |
| "Financial Advisors" | Citi, Davy Corporate Finance, Ondra and Morgan Stanley |
| "Form of Proxy" | the form of proxy to be mailed with the Notice for use by Shareholders in connection with the EGM |
| "Glaxo" | Glaxo Group Limited |
| "Guarantors" | Elan and the Subsidiary Note Guarantors |
| "Indenture" | Indenture governing the 2021 Notes |
| "Initial Purchasers" | Morgan Stanley & Co. LLC and J&E Davy |
| "Investment Company" | as that term is described in the Investment Company Act of 1940 |
| "Irish Stock Exchange" | The Irish Stock Exchange Limited |
| "Irish Takeover Rules" or "Takeover Rules" | the Irish Takeover Panel Act 1997, Takeover Rules 2007 (as amended) |
| "Issue Date" | the date of issue of the 2021 Notes |
| "Issuers" | Elan Finance plc and Elan Finance Corp. in connection with the 2021 Notes |
| "Janssen AI" | Janssen Alzheimer Immunotherapy |
| "Latest Practicable Date" | 23 May 2013, being the latest practicable date before the publication of this document |
| "Listing Rules" | the listing rules of the Irish Stock Exchange |
| "Morgan Stanley" | Morgan Stanley & Co. International plc |
| "MS" | Multiple Sclerosis |
| "Nerano" | Nerano Pharma Limited |
| "NewBridge" | NewBridge Pharmaceuticals |
| 37 |
| "Notice" | the notice of Extraordinary General Meeting set out at the end of this document |
| "Offer Period" | the period commencing on, and including, 25 February 2013 and ending on such date as the Irish Takeover Rules provide or the Irish Takeover Panel otherwise may decide |
| "Ondra" | Ondra LLP, trading as Ondra Partners |
| "Ordinary Shareholder" | a holder of Ordinary Shares |
| "Ordinary Shares" | ordinary shares of €0.05 each in the capital of the Company |
| "Overseas Shareholder" | a Shareholder who is resident in, or a citizen of jurisdictions outside Ireland or custodians, nominees or trustees for persons who are citizens or nationals of, or residents in jurisdictions outside Ireland |
| "Panel" or "Irish Takeover Panel" | the Irish Takeover Panel |
| "Prothena" | Prothena Corporation plc |
| "Purchase Agreement" | the purchase agreement dated 23 May 2013 among Elan, its subsidiaries, Elan Finance public limited company, Elan Finance Corp, and certain of Elan’s other subsidiaries and Morgan Stanley & Co. LLC whereby the Issuers agree to sell the securities to Morgan Stanley & Co. LLC, as representative of the Initial Purchasers |
| "R&D" | research and development |
| "Register" | the register of members of the Company |
| "Registrars" | the Company’s registrar, being Computershare Investor Services (Ireland) Limited, Heron House, Corrig Road, Sandyford Industrial Estate, Dublin 18, Ireland |
| "Regulatory Information Service" | any of the services set out in Schedule 12 of Appendix 2 of the Listing Rules and/or the company announcement of The Irish Stock Exchange |
| "Resolutions" | the Theravance Resolution, the ELND005 Resolution, the AOP Resolution and the Share Repurchase Resolution, each being a “Resolution” |
| "Revised Offer" | the revision to the Royalty Pharma Offer the details of which are set forth in the Revised Offer Announcement and in the Revised Offer Document |
| "Revised Offer Announcement" | the announcement by Royalty Pharma dated 20 May 2013 revising the Royalty Pharma Offer |
| "Revised Offer Document" | the revised offer document issued by Royalty Pharma dated 23 May 2013 |
| "Royalty Interest Products" | the four respiratory programs under partnership between Theravance and Glaxo, being (1) RELVAR™ ELLIPTA™/BREO™ ELLIPTA™, (2) ANORO™ ELLIPTA™, (3) MABA ’081, and (4) vilanterol (VI) monotherapy |
| "Royalty Pharma" | RP Management, LLC or Echo Pharma Acquisition Limited as the context so requires |
| "Royalty Pharma Offer" or "Offer" | the offer made by Royalty Pharma on 2 May 2013 to acquire the entire issued and to be issued share capital of Elan as revised by the Revised Offer Announcement (on the terms, and subject to the conditions, set out in the Royalty Pharma Offer Document and the Revised Offer Document) including where the context so |
| 38 |
| requires, any subsequent revision, variation, extension or renewal of such offer | |
| "Royalty Pharma Offer Document" | the document dated 2 May 2013 sent by Royalty Pharma to Shareholders containing the terms and conditions of the Offer |
| "Royalty Pharma Offer Period" or "Offer Period" | or the period commencing on, and including, 25 February 2013 and ending on such date as the Irish Takeover Rules provide or the Irish Takeover Panel otherwise may decide |
| "SASR" | SASR Neunundvierzigste Beteiligungsverwaltung GmbH |
| "SEC" | the United States Securities and Exchange Commission |
| "Securities" | US$850,000,000 principal amount of 6.25% senior notes due 2021 |
| "SG&A" | selling, general and administrative |
| "Shareholders" | holders of Ordinary Shares and/or ADSs |
| "Shareholder Helpline" | the dedicated helpline telephone number +353 1 447 5107 for Elan Ordinary Shareholders |
| "Share Repurchase Program" | a share repurchase program with respect to the Shares of the Company, in an amount of up to $200 million |
| "Share Repurchase Resolution" | the ordinary resolution with respect to the approval of the Share Repurchase Program |
| "Speranza Biopharma" | Speranza Biopharma Limited |
| "Speranza Group" or "Speranza" | Speranza Therapeutics Limited together with Speranza Biopharma Limited |
| "Speranza Therapeutics" | Speranza Therapeutics Limited |
| "Subsidiary Note Guarantors" | certain of Elan’s subsidiaries (as set out in Schedule II to the Purchase Agreement) |
| "Tender Offer" | the repurchase by the Company of 88,888,888 Ordinary Shares (including Ordinary Shares represented by ADSs) from Davy for $11.25 each for an aggregate purchase price of approximately $1 billion |
| "Theravance" | Theravance, Inc. |
| "Theravance Dividend Program" | a cash dividend program directly linked to royalty participation interest payments received by Elan pursuant to the Theravance Transaction |
| "Theravance Resolution" | the ordinary resolution with respect to the approval of the Theravance Transaction set out in the Notice |
| "Theravance Royalty Participation Agreement" | the Royalty participation agreement between Elan and Theravance dated 12 May 2013 pursuant to which Elan will purchase a participation interest in potential future royalty payments |
| "Theravance Transaction" | the transaction with Theravance pursuant to which Elan will make a one-time cash payment of $1.0 billion to Theravance in exchange for a 21% participation interest in the potential future royalty payments from the Royalty Interest Products earned by Theravance, when, as and if received |
| "Transactions" | the AOP Transcation, the Theravance Transaction, the ELND005 Transaction, the Share Repurchase Program, each being a "Transaction" |
| 39 |
| "Transition" | Transition Therapeutics, Inc. |
| "Transition Collaboration Agreement" | collaboration agreement between Elan and Waratah Pharmaceuticals dated 25 September 2006, as amended |
| "Trustee" | The Bank of New York Mellon, London Branch |
| "Tysabri Dividend Program" | a cash dividend program directly linked to Tysabri market performance, calculated as a percentage of the Tysabri royalties paid to Elan by Biogen Idec pursuant to the Tysabri Transaction |
| "Tysabri Transaction" | the transfer to Biogen Idec International Holding Ltd., an affiliate of Biogen Idec, of all of Elan’s interest in the intellectual property and other assets related to the development, manufacturing and commercialization of Tysabri |
| "uncertificated" or "in uncertificated form" | with respect to Ordinary Shares, recorded on the Register as being held in uncertificated form in CREST and title to which, by virtue of the Uncertified Securities Regulations, may be transferred by means of CREST |
| "United States" or "US" | the United States of America, its territories and possessions, any State of the United States and the District of Columbia |
| "US dollar" or "USD" or "US$" | the lawful currency of the United States |
| "Voting Record Date" | close of business on 15 June 2013 |
| 40 |
Appendix I
| 1. | AOP FINANCIALS |
The extracts from the AOP 2011 Financial Statements, as set out in this Appendix I, which have been prepared and audited under Austrian GAAP, present the entire AOP business and do not separately carve out the estimated amounts attributable to AOP’s current generics business which is not been acquired. The EBITDA reported elsewhere in this Circular excludes approximately €6.7 million (approximately $8.5 million) of costs associated with the AOP current generics products business, which is not being acquired. Please refer to “Part 5 – Risk Factors – Available financial information related to AOP is subject to significant limitations” for a further discussion on how the AOP 2011 Financial Statements were prepared.
| 41 |
| 2. | AOP Financials |
AOP Orphan Pharmaceuticals AG
Balance sheet
as at 31.12.2011
| 31.12.2011 | 31.12.2011 | 31.12.2010 | ||||||||||||
| ASSETS | EUR | EUR | EUR | |||||||||||
| A. | Fixed assets | |||||||||||||
| I. | Intangible assets | |||||||||||||
| 1. Industrial property rights and similar rights/benefits | 284,287.07 | 48,707.28 | ||||||||||||
| II. | Tangible assets | |||||||||||||
| 1. Installations in third-party buildings | 426,197.31 | 471,719.80 | ||||||||||||
| 2. Other facilities, factory and office equipment | 1,007,883.62 | 895,406.17 | ||||||||||||
| 1,434,080.93 | 1,367,125.97 | |||||||||||||
| III. | Financial assets | |||||||||||||
| 1. Investments in affiliated companies | 35,001.00 | 35,001.00 | ||||||||||||
| 2. Participations | 56,647.37 | 56,647.17 | ||||||||||||
| 3. Investment securities (book-entry securities) of the fixed assets | 1,939,834.22 | 2,698,792.78 | ||||||||||||
| 4. Other loans | 1,197,264.72 | 1,044,360.67 | ||||||||||||
| 3,228,747.11 | 3,834,801.62 | |||||||||||||
| 4,947,115.11 | 5,250,634.87 | |||||||||||||
| B. | Current assets | |||||||||||||
| I. | Stocks | |||||||||||||
| 1. Raw materials and supplies | 2,864,820.08 | 2,104,186.94 | ||||||||||||
| 2. Unfinished products | 509,212.60 | 222,185.10 | ||||||||||||
| 3. Finished products and goods | 6,700,207.94 | 4,695,835.90 | ||||||||||||
| 10,074,240.62 | 7,022,207.94 | |||||||||||||
| II. | Receivables and other financial assets | |||||||||||||
| 1. Trade accounts receivable | 13,489,933.58 | 12,326,989.19 | ||||||||||||
| 2. Receivables from affiliated companies | 720.00 | 9,166.39 | ||||||||||||
| 3. Other receivables and financial assets | 3,653,464.23 | 5,084,555.98 | ||||||||||||
| 17,144,117.81 | 17,420,711.56 | |||||||||||||
| III. | Securities and shares | |||||||||||||
| 1. Other securities and shares | 1,566,652.47 | 1,456,354.07 | ||||||||||||
| IV. | Cash on hand and at banks | 8,857,652.94 | 4,430,597.93 | |||||||||||
| 37,642,663.84 | 30,329,871.50 | |||||||||||||
| C. | Accrued and deferred items | 598,399.03 | 336,900.73 | |||||||||||
| TOTAL ASSETS | 43,188,177.98 | 35,917,407.10 | ||||||||||||
| 31.12.2011 | 31.12.2011 | 31.12.2010 | ||||||||||||
| LIABILITIES | EUR | EUR | EUR | |||||||||||
| A. | Shareholders’ equity | |||||||||||||
| I. | Share capital | 200,000.00 | 200,000.00 | |||||||||||
| II. | Capital reserves | |||||||||||||
| 1. Free | 980,627.20 | 980,627.20 | ||||||||||||
| III. | Retained earnings | |||||||||||||
| 1. Legal reserves | 20,000.00 | 20,000.00 | ||||||||||||
| 2. Other reserves (free reserves) | 2,802,750.86 | 2,802,750.86 | ||||||||||||
| 2,822,750.86 | 2,822,750.86 | |||||||||||||
| IV. | Unappropriated profit (unappropriated loss) | 22,499,035.67 | 18,579,365.43 | |||||||||||
| of which accumulated profit | 18,579,365.43 | 14,604,746.06 | ||||||||||||
| 26,502,413.73 | 22,582,743.49 | |||||||||||||
| B. | Reserves | |||||||||||||
| 1. Reserves for severance payments | 258,679.10 | 251,485.22 | ||||||||||||
| 2. Reserves for pensions | 441,523.81 | 371,171.98 | ||||||||||||
| 3. Tax reserves | 0.00 | 848,355.49 | ||||||||||||
| 4. Other provisions | 5,216,977.26 | 5,379,224.45 | ||||||||||||
| 5,917,180.17 | 6,850,237.14 | |||||||||||||
| C. | Liabilities | |||||||||||||
| 1. Liabilities to banks | 118,186.37 | 334,999.99 | ||||||||||||
| 2. Trade accounts payable | 8,213,181.72 | 4,412,324.38 | ||||||||||||
| 3. Liabilities to affiliated companies | 400,800.00 | 105,600.00 | ||||||||||||
| 4. Other liabilities | 2,036,415.99 | 1,631,502.10 | ||||||||||||
| of which taxes | 149,599.81 | 180,580.34 | ||||||||||||
| of which as part of social security contributions | 36,921.72 | 36,690.56 | ||||||||||||
| 10,768,584.08 | 6,484,426.47 | |||||||||||||
| D. | Accrued and deferred items | 0.00 | 0.00 | |||||||||||
| TOTAL LIABILITIES | 43,188,177.98 | 35,917,407.10 | ||||||||||||
| [Signature] [Signature] |
| 42 |
| AOP Orphan Pharmaceuticals AG | ||||||||||
| Profit and loss statement | ||||||||||
| 01.01.2011 to 31.12.2011 | ||||||||||
| 2011 (EUR) | 2010 (EUR) | |||||||||
| 1. | Sales | 57,072,918.32 | 45,024,583.22 | |||||||
| 2. | Change to the stock of finished and unfinished products | 565,471.51 | (192,238.00 | ) | ||||||
| 3. | Other operating income | |||||||||
| a) Income from the disposal of fixed assets | 16,546.31 | 50.00 | ||||||||
| b) Income from the releasing of reserves | 1,198,252.69 | 404,559.42 | ||||||||
| c) Other | 2,681,689.19 | 1,689,398.77 | ||||||||
| 3,896,488.19 | 2,094,008.19 | |||||||||
| 4. | Expenses for material and other manufacturing services purchased | |||||||||
| a) Cost of materials | 19,201,693.60 | 13,956,297.79 | ||||||||
| b) Expenses for services purchased | 3,213,913.78 | 2,115,544.21 | ||||||||
| 22,415,607.38 | 16,071,842.00 | |||||||||
| 5. | Personnel expenses | |||||||||
| a) Salaries | 5,901,212.13 | 4,437,179.40 | ||||||||
| b) Expenses for severance payments | 53,982.75 | 71,264.15 | ||||||||
| c) Expenses for pensions | 77,071.83 | 73,454.71 | ||||||||
| d) Expenses for statutory social insurance contributions, payroll-related levies and compulsory contributions | 1,227,701.72 | 940,128.55 | ||||||||
| e) Other social benefits | 104,306.19 | 60,107.84 | ||||||||
| 7,364,274.62 | 5,582,134.65 | |||||||||
| 6. | Depreciations | |||||||||
| a) of fixed tangible and intangible assets | 577,982.42 | 465,947.15 | ||||||||
| 7. | Other operating expenses | |||||||||
| a) Taxes other than income tax and earnings tax | 794,113.78 | 208,889.97 | ||||||||
| b) Other | 21,969,522.18 | 17,865,340.27 | ||||||||
| 22,763,635.96 | 18,074,230.24 | |||||||||
| 8. | Subtotal from Lines 1 to 7 (operating profit) | 8,413,377.64 | 6,732,199.37 | |||||||
| 9. | Income from other securities and loans in financial assets | 82,691.59 | 60,830.77 | |||||||
| 10. | Other interest and similar income | |||||||||
| Interest income from bank deposits | 45,162.98 | 46,786.03 | ||||||||
| Income from securities held as current assets | 0.00 | 0.00 | ||||||||
| Interest income from loans issued | 54,764.22 | 8,627.81 | ||||||||
| 99,927.20 | 55,413.84 | |||||||||
| 11. | Income from the disposal and appreciation in value of financial assets and securities held as current assets | |||||||||
| Income from the disposal of current asset securities | 373,316.16 | 1,620.66 | ||||||||
| Income from option business | 30,423.71 | 30,637.48 | ||||||||
| 403,739.87 | 32,258.14 | |||||||||
| 12. | Expenses from financial assets and securities held as current assets | |||||||||
| Expenses from financial assets and securities held as current assets | 3,955,428.75 | 1,433,985.92 | ||||||||
| of which depreciations on financial assets | 2,565,883.95 | 1,412,584.96 | ||||||||
| of which depreciations on securities held as current assets | 387,430.20 | 14,826.68 | ||||||||
| Expenses from option business | 39,279.60 | 164,776.25 | ||||||||
| 3,994,708.35 | 1,598,762.17 | |||||||||
| 43 |
| Profit and loss statement | ||||||||||
| 01.01.2011 to 31.12.2011 | ||||||||||
| 2011 (EUR) | 2010 (EUR) | |||||||||
| 13. | Interest and similar expenses | |||||||||
| Debit interest | 208,575.87 | 51,310.47 | ||||||||
| 14. | Subtotal from Lines 9 to 13 (financial income) | (3,616,925.56 | ) | (1,501,569.89 | ) | |||||
| 15. | Earnings from normal business activities | 4,796,452.08 | 5,230,629.48 | |||||||
| 16. | Taxes on income and profit | 876,781.84 | 1,256,010.11 | |||||||
| 17. | Annual net profit | 3,919,670.24 | 3,974,619.37 | |||||||
| 18. | Release of retained earnings | |||||||||
| a) Legal reserves | 0.00 | 0.00 | ||||||||
| 19. | Annual profit | 3,919,670.24 | 3,974,619.37 | |||||||
| 20. | Earnings carried forward from the previous year | 18,579,365.43 | 14,604,746.06 | |||||||
| 21. | Balance sheet profit | 22,499,035.67 | 18,579,365.43 | |||||||
[Signature] [Signature]
| 44 |
NOTES
Financial statements for the year ending 31 December 2011
ACCOUNTING AND VALUATION METHODS
General principles
The financial statements were prepared in compliance with generally accepted accounting principles and in compliance with the general standard of providing the most accurate possible representation of the company’s net worth, financial position and earnings.
The annual financial statements were compiled according to the principle of completeness.
The principle of individual valuation was observed when valuing the various assets and debts, and the going concern principle was assumed.
The principle of prudence was taken into account by reporting only those earnings actually realised as at the balance sheet date. All recognisable risks and impending losses were taken into account.
Fixed assets
Intangible assets
Acquired intangible assets were valued at the purchasing or manufacturing cost and, where subject to depreciation, reduced in value according to the planned depreciation model.
Scheduled depreciation is calculated using the straight-line method and is based on the following useful lives:
| Asset group | Useful life |
| IT software | 2-5 years |
| Internet presence | 3 years |
Property, plant and equipment
Tangible assets were valued at the purchasing or manufacturing cost and, where subject to depreciation, reduced in value according to the planned depreciation model.
Scheduled depreciation is calculated using the straight-line method and is based on the following useful lives for each individual asset group:
| Asset group | Useful life |
| Investment in third-party buildings | 4-10 years |
| Plant and office equipment | 3-10 years |
| Vehicles | 4 years |
Low-value assets for the financial year were written down immediately and in full in the year of acquisition.
Financial assets
Financial assets were valued at the cost of acquisition and, where necessary, subjected to unscheduled write-downs.
Unscheduled write-downs were only made when the impairment is expected to be permanent.
Current assets
Inventories
Inventories were valued at the purchasing or manufacturing cost or at the lower fair value.
Social security contributions and interest on borrowed capital were not taken into account when determining manufacturing costs.
Receivables and other assets
Receivables and other assets were recognised at their nominal value.
| 45 |
Receivables in foreign currency were valued either at the rate of the transaction date or, if lower, at the exchange rate of the balance sheet date.
In the case of discernible specific risks, the lower applicable value was used.
Current asset securities were valued at the cost of acquisition or at the lower stock exchange price at the balance sheet date.
Provisions
Provision for severance payments
Severance provisions were calculated according to actuarial principles based on an interest rate of 4% and the legal retirement age.
No deduction for fluctuation was applied when calculating provisions.
Pension provisions
Pension provisions were calculated according to actuarial principles based on the present value method, using an interest rate of 5%.
Other provisions
All risks recognisable at the date of preparation of the financial statement, together with contingent liabilities of uncertain amount or unclear cause, were taken into account in the amount of other provisions according to the principle of reasonable business appraisal.
Liabilities
Liabilities are calculated at the repayable amount in accordance with the principle of prudence.
| 46 |
NOTES ON THE BALANCE SHEET
Development of fixed assets
The development of the individual items included in fixed assets and the analysis of depreciation and amortisation charges (Sect. 226, Para. 1) are shown in the assets schedule (Appendix IV).
Inventories
Inventories are as follows. Figures from the previous year are shown in brackets:
| Change in inventories (in EUR) | Final inventory 31.12 (in EUR) | |
| Raw materials | + 760,633.14 | 2,864,820.08 |
| (2010: - 151,356.50) | (2010: 2,104,186.94) | |
| Unfinished products | + 286,972.80 | 509,212.60 |
| (2010: + 47,545.11) | (2010: 222,185.10) | |
| Finished products | + 278,498.71 | 1,713,138.61 |
| (2010: - 239,783.11) | (2010: 1,434,782.78) | |
| Merchandise | + 1,732,195.51 | 4,987,069.33 |
| (2010: + 1,232,846.57) | (2010: 3,261,053.12) |
Receivables and other assets
The table below shows the remaining terms of the assets on the balance sheet:
| Total (EUR) |
Of which due within 1 year (EUR) |
Of which due within 1 to 5 years (EUR) | |
| Trade receivables | 13,489,933.58 | 12,642,900.58 | 847,033.00 |
| (2010: 12,326,989.19) | (2010: 12,326,989.19) | (2010: 0.00) | |
| Receivables from affiliated | 720.00 | 720.00 | 0.00 |
| companies | (2010: 9,166.39) | (2010: 9,166.39) | (2010: 0.00) |
| Other receivables and assets | 3,653,464.23 | 3,103,838.28 | 549,625.95 |
| (2010: 5,084,555.98) | (2010: 2,816,427.11) | (2010 2,268,127.87) | |
| Total receivables | 17,144,117.81 | 15,747,458.86 | 1,396,658.95 |
| (2010: 17,420,711.56) | (2010: 14,054,088.59) | (2010: 2,268,127.87) |
Other receivables and assets
The item “Other receivables and assets” includes significant amounts that will only produce cash additions/outflows after the balance sheet date. These relate to an advance payment for a research project, receivables from tax authorities and the research premiums for 2010 and 2011.
Equity
The company has positive equity of EUR 26,502,413.73 (2010: EUR 22,582,743.49).
The share capital of €200,000.00 is divided into 20,000 shares of nominal value €10.00.
Provisions
An analysis of provisions is shown in Appendix V.
| 47 |
Liabilities
The following table provides the terms of liabilities on the balance sheet:
| Of which due | Of which due | ||
| Total | within 1 year | within 1 to 5 years | |
| (EUR) | (EUR) | (EUR) | |
| Liabilities due to credit institutions | 118,186.37 | 118,186.37 | 0.00 |
| (2010: 334,999.99) | (2010: 223,000.00) | (2010: 111,999.99) | |
| Liabilities from trade receivables | 8,213,181.72 | 8,213,181.72 | 0.00 |
| (2010: 4,412,324.38) | (2010: 4,412,324.38) | (2010: 0.00) | |
| Liabilities due to affiliated | |||
| companies | 400,800.00 | 400,800.00 | 0.00 |
| (2010: 105,600.00) | (2010: 105,600.00) | (2010: 0.00) | |
| Other liabilities | 2,036,415.99 | 2,036,415,99 | 0.00 |
| (2010: 1,631,502.10) | (2010: 1,631,502.10) | (2010: 0.00) | |
| Total liabilities | 10,768,584.08 | 10,768,584.08 | 0.00 |
| (2010: 6,484,426.47) | (2010: 6,312,426.48) | (2010: 111,999.99) |
Other liabilities
The item “Other liabilities” includes significant amounts that will be recorded as expenses but will only produce cash additions/outflows in the following year. These relate to premiums in accordance with the premium agreements for 2011.
| 48 |
NOTES TO THE PROFIT AND LOSS STATEMENT
The income statement was structured according to the nature of expense method detailed in Sect. 231, Para. 2.
Sales
The breakdown of sales revenue by geographical market is as follows:
| Sales in Austria | EUR | 20,633,896.59 | ||
| Sales in the EU (other than Austria) | EUR | 30,126,889.76 | ||
| Sales in third countries (non-EU) | EUR | 6,312,131.97 | ||
| Total sales: | EUR | 57,072,918.32 |
Expenses for severance payments
In 2011, a total of EUR 53,982.75 was spent on severance payments, of which EUR 46,788.87 was for contributions to company-sponsored employee provision funds.
Information about the effects of income and revenue taxes on the POA
Taxes on income and revenue totalled EUR 876,781.84 and impacted the profit on ordinary activities by 18.28%.
OTHER MANDATORY DISCLOSURES
Information on participating companies
The information in accordance with Sect. 238 line 2 may be omitted as the reporting company owns less than 20% of the shares.
Number of employees
The average number of employees arranged according to wage earners and salaried staff (Sect. 239, Para. 1, line 1) is as follows:
| 2011 | 2010 | |||||||
| Wage earners | 0 | 0 | ||||||
| Salaried staff | 98 | 73 | ||||||
| Total | 98 | 73 | ||||||
Information on the members of the Board of Directors
| Management: | Name | since |
| Dr Georg Fischer | 27.06.2008 | |
| Dr Christoph Wachter | 27.06.2008 | |
| Dr Rudolf Widmann | 15.08.2003 |
Information on the members of the Supervisory Board
In the 2011 business year, the following people were members of the Supervisory Board.
Dr Heinz Russwurm (Chairman)
Mag Heinrich Friedrich (Deputy Chairman)
Dr Ingrid Aichinger-Widmann
Vienna, September 2012
| [Signature] | [Signature] | |
| Dr Rudolf Widmann CEO | ||
| 24.09.2012 |
| 49 |
| AOP Orphan Pharmaceuticals AG |
Statement of assets as at 31.12.2011 |
| Change in purchasing and manufacturing costs | Depreciation trend | Book values | ||||||||||||||||||||||||||||||||||||||||||||||||
| As at | Reclassi- | As at | As at | As at | As at | As at | ||||||||||||||||||||||||||||||||||||||||||||
| 01.01.2011 | Addition | fication | Disposal | 31.12.2011 | 01.01.2011 | Addition | Disposal | Attribution | 31.12.2011 | 31.12.2010 | 31.12.2011 | |||||||||||||||||||||||||||||||||||||||
| in EUR | in EUR | in EUR | in EUR | in EUR | in EUR | in EUR | in EUR | in EUR | in EUR | in EUR | in EUR | |||||||||||||||||||||||||||||||||||||||
| A. | Fixed assets | |||||||||||||||||||||||||||||||||||||||||||||||||
| I. | Intangible assets | |||||||||||||||||||||||||||||||||||||||||||||||||
| Industrial property rights and similar | ||||||||||||||||||||||||||||||||||||||||||||||||||
| 1. | Rights and benefits | |||||||||||||||||||||||||||||||||||||||||||||||||
| Data processing program | 125,696.85 | 197,937.66 | 0.00 | 7,181.00 | 316,453.51 | 76,989.37 | 41,911.57 | 7,181.00 | 0.00 | 111,720.14 | 48,707.28 | 204,733.37 | ||||||||||||||||||||||||||||||||||||||
| Online presence | 0.00 | 80,750.00 | 0.00 | 0.00 | 80,750.00 | 0.00 | 1,195.30 | 0.00 | 0.00 | 1,196.30 | 0.00 | 79,553.70 | ||||||||||||||||||||||||||||||||||||||
| 125,696.85 | 278,687.66 | 0.00 | 7,181.00 | 397,203.51 | 76,989.57 | 43,107.87 | 7,181.00 | 0.00 | 112,916.44 | 48,707.28 | 204,287.07 | |||||||||||||||||||||||||||||||||||||||
| II. | Tangible assets | |||||||||||||||||||||||||||||||||||||||||||||||||
| 1. | Installations in third-party buildings (Baul. lnv. Frmd. Belr.-u. Geschg.) | 562,867.70 | 12,481.65 | 0.00 | 0.00 | 575,349.35 | 91,147.90 | 58,004.14 | 0.00 | 0.00 | 149,152.04 | 471,719.80 | 426,197.31 | |||||||||||||||||||||||||||||||||||||
| 2. | Other equipment, factory and office equipment Vehicles | 1,050,885.67 | 454,929.20 | 0.00 | 162,626.05 | 1,343,188.82 | 463,731.64 | 281,969.32 | 137,104.43 | 0.00 | 603,596.73 | 587,153.83 | 734,592.09 | |||||||||||||||||||||||||||||||||||||
| Office machinery, EDP systems | 425,496.63 | 50,245.24 | 0.00 | 76,356.12 | 399,385.75 | 205,858.03 | 71,160.66 | 75,300.04 | 0.00 | 201,718.65 | 219,638.60 | 197,667.10 | ||||||||||||||||||||||||||||||||||||||
| Office equipment | 210,785.42 | 8,394.93 | 0.00 | 17,624.18 | 201,556.17 | 122,171.68 | 20,498.67 | 16,738.61 | 0.00 | 125,931.74 | 88,613.74 | 75,624.43 | ||||||||||||||||||||||||||||||||||||||
| Low-value assets | 0.00 | 103,241.76 | 0.00 | 103,241.76 | 0.00 | 0.00 | 103,241.76 | 103,241.76 | 0.00 | 0.00 | 0.00 | 0.00 | ||||||||||||||||||||||||||||||||||||||
| 1,687,167.72 | 616,811.13 | 0.00 | 359,848.11 | 1,944,130.74 | 791,761.55 | 476,870.41 | 332,384.84 | 0.00 | 936,247.12 | 895,406.17 | 1,007,883.62 | |||||||||||||||||||||||||||||||||||||||
| 2,250,035.42 | 629,292.73 | 0.00 | 359,848.11 | 2,519,480.03 | 882,909.45 | 534,874.55 | 332,384.84 | 0.00 | 1,085,399.16 | 1,367,125.97 | 1,434,030.93 | |||||||||||||||||||||||||||||||||||||||
| III. | Financial assets | |||||||||||||||||||||||||||||||||||||||||||||||||
| 1. | Investments in affiliated companies | |||||||||||||||||||||||||||||||||||||||||||||||||
| Investments in related companies | 35,001.00 | 0.00 | 0.00 | 0.00 | 35,001.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 35,001.00 | 35,001.00 | ||||||||||||||||||||||||||||||||||||||
| 2. | Participations | |||||||||||||||||||||||||||||||||||||||||||||||||
| Other participations | 56,647.17 | 0.00 | 0.00 | 0.00 | 56,647.17 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 56,647.17 | 56,647.17 | ||||||||||||||||||||||||||||||||||||||
| 3. | Fixed asset securities | 4,152,670.34 | 2,057,250.00 | 0.00 | 2,488,356.10 | 3,721,564.24 | 1,453,877.55 | 368,085.10 | 40,232.64 | 0.00 | 1,781,730.02 | 2,698,792.78 | 1,939,834.22 | |||||||||||||||||||||||||||||||||||||
| 4. | Other loans | |||||||||||||||||||||||||||||||||||||||||||||||||
| Loans | 837,360.67 | 29,798.60 | 0.00 | 121,094.40 | 746,064.87 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 837,360.67 | 746,064.87 | ||||||||||||||||||||||||||||||||||||||
| Loans issued | 207,000.00 | 2,441,998.72 | 0.00 | 0.00 | 2,648,998.72 | 0.00 | 2,197,798.85 | 0.00 | 0.00 | 2,197,798.85 | 207,000.00 | 451,199.87 | ||||||||||||||||||||||||||||||||||||||
| 1,044,360.67 | 2,471,797.32 | 0.00 | 121,094.40 | 3,395,063.59 | 0.00 | 2,197,798.85 | 0.00 | 0.00 | 2,197,798.85 | 1,044,360.67 | 1,197,264.74 | |||||||||||||||||||||||||||||||||||||||
| 5,203,679.18 | 4,529,047.32 | 0.00 | 2,609,450.50 | 7,208,276.00 | 1,453,877.56 | 2,565,883.95 | 40,232.64 | 0.00 | 3,979,528.87 | 3,834,801.62 | 3,228,747.13 | |||||||||||||||||||||||||||||||||||||||
| TOTAL ANALYSIS OF ASSETS | 7,664,411.45 | 5,437,027.76 | 0.00 | 2,976,479.61 | 10,124,959.60 | 2,413,776.58 | 3,143,866.37 | 379,798.48 | 0.00 | 5,177,844.47 | 5,250,634.87 | 4,947,115.13 | ||||||||||||||||||||||||||||||||||||||
| [Signature] [Signature] |
| 24.09.2012 |
| Notes/Appendix IV |
| 50 |
| AOP Orphan Pharmaceuticals AG |
Reserves as at 31.12.2011 |
| As at | As at | |||||||||||||||||||
| 31.12.2010 | Use | Liquidation | Allocation | 31.12.2011 | ||||||||||||||||
| (in EUR) | (in EUR) | (in EUR) | (in EUR) | (in EUR) | ||||||||||||||||
| Reserves for severance payments | 251,485.22 | 0.00 | 0.00 | 7,193.88 | 258,679.10 | |||||||||||||||
| Reserves for pensions | 371,171.98 | 0.00 | 0.00 | 70,351.83 | 441,523.81 | |||||||||||||||
| Tax reserves | 848,355.49 | 848,355.49 | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Reserves for procedural costs | 142,000.00 | 0.00 | 20,000.00 | 566,953.10 | 688,953.10 | |||||||||||||||
| Reserve for bonuses | 60,779.77 | 60,779.77 | 0.00 | 187,533.54 | 187,533.54 | |||||||||||||||
| Reserve for holidays | 156,008.35 | 0.00 | 0.00 | 21,888.76 | 177,897.11 | |||||||||||||||
| Reserve for anniversary bonuses | 85,298.21 | 0.00 | 0.00 | 16,310.59 | 101,608.80 | |||||||||||||||
| Reserve for audit expenses | 45,000.00 | 0.00 | 0.00 | 0.00 | 45,000.00 | |||||||||||||||
| Reserve for contingent losses | 373,448.27 | 0.00 | 5,295.20 | 945,000.00 | 1,313,153.07 | |||||||||||||||
| Reserves for research & development | 4,516,689.85 | 1,117,273.60 | 1,156,374.72 | 459,790.11 | 2,702,831.64 | |||||||||||||||
| Total reserves | 6,850,237.14 | 2,026,408.86 | 1,181,669.92 | 2,275,021.81 | 5,917,180.17 | |||||||||||||||
| [Signature] [Signature] | |
| 24.09.2012 | |
| Notes/Appendix V |
| 51 |
Appendix 1a
Summary of uncorrected misstatements
Company: AOP Orphan Pharmaceuticals AG
Reporting date: 31.12.2011
| Balance sheet | Income statement | Tax effect | ||||
| Basis of | ||||||
| Description of difference | Target | Credit | Target | Credit | assessment | Tax |
| Error in the annual statement | ||||||
| Incorrect identity in the annual statement | ||||||
| Deviations in assessments and valuations | ||||||
| Total errors and deviations: | ||||||
| Tax effect (tax rate... %) | ||||||
| [signature] [signature] | |
| [signature] | |
| 24.09.2012 |
| 52 |
| 4. | Audit opinion |
Report on the annual financial statements
We have audited the enclosed annual financial statements of
AOP Orphan Pharmaceuticals AG, Vienna,
for the financial year 1 January 2011 to 31 December 2011 including the accounts. These annual financial statements include the balance sheet to 31 December 2011, the profit and loss statement for the financial year ending 31 December 2011, and the notes.
Our responsibility and liability is limited in accordance with Sect. 275(2) UGB (Liability regulations for auditing small- and medium-sized companies) to the company and third parties in the amount of 2 million euros.
Responsibility of legal representative for the annual financial statements and accounts
The legal representatives of the company are responsible for the accounts and for the preparation the annual financial statements, which must provide the most accurate possible representation of the asset, financial and earnings position of the company in compliance with Austrian company regulations. This responsibility includes the formation, implementation and maintenance of an internal control system, insofar as this is important for preparing the annual financial statements and giving the most accurate possible presentation of the company’s assets, financial and earnings position, so as to avoid material misstatements, whether through intentional or unintentional error; the selection and application of suitable accounting and valuation methods; and the use of estimates which appear appropriate based on the given framework conditions.
Responsibility of the auditors and description of the nature and scope of the statutory audit
We are responsible for providing an opinion on these annual financial statements based on our audit. We have carried out our audit in accordance with the legal regulations and principles of proper accounting in Austria. These principles require us to abide by a code of conduct and plan and execute the audit such that we can form an opinion with adequate certainty as to whether the annual financial statements are free from material misstatement.
An audit involves the performance of tests to obtain proof regarding the amounts and other information stated in the annual financial statements. The selection of tests is at the discretion of the auditor, based on his assessment of the risk of occurrence of material misstatements, whether due to intentional or unintentional error. When carrying out this risk assessment, the auditor takes into account the internal control system, insofar as this is of significance for the preparing of the annual financial statements and the conveying of an accurate image of the assets, financial and earnings position of the company, so as to define appropriate tests based on the framework conditions, but not to give a judgement on the effectiveness of the company’s internal controls. The audit further involves assessing the appropriateness of the accounting and valuation methods applied and the material estimates carried out by the legal representatives, as well as an evaluation of the overall presentation of the annual financial statements.
We are of the opinion that we have obtained sufficient and suitable proof for our audit to give a sufficiently reliable basis for our audit opinion.
Audit opinion
Our audit revealed no objections. Based on the findings obtained from our audit, we believe that the annual financial statements satisfy the legal regulations and supplementary requirements of the Articles of Association, and give the most accurate possible presentation of the asset and financial position of the company as at 31 December 2011 and the earnings position of the company for the financial year 1 January 2011 to 31 December 2011, in compliance with the Austrian principles of proper accounting.
Statements on the management report
The management report is to be reviewed on the basis of legal regulations to ensure that it mirrors the annual financial statements and that the information contained in the management report does not give rise to an incorrect representation of the company’s position. The audit opinion must also contain a statement as to whether the management report is in compliance with the annual financial statements.
| 53 |
In our opinion the management report is in compliance with the annual financial statements.
Vienna, 24.09.2012
[Signature]
SMP
Wirtschaftsprüfungsgesellschaft m.b.H. [Audit firm]
The publication or disclosure of the annual financial statements with our audit opinion may only be carried out in the version confirmed by us. This audit opinion refers exclusively to the German version and full annual financial statements including management report. Any other versions shall be subject to the provisions of Sect. 281(2) UGB.
| 54 |







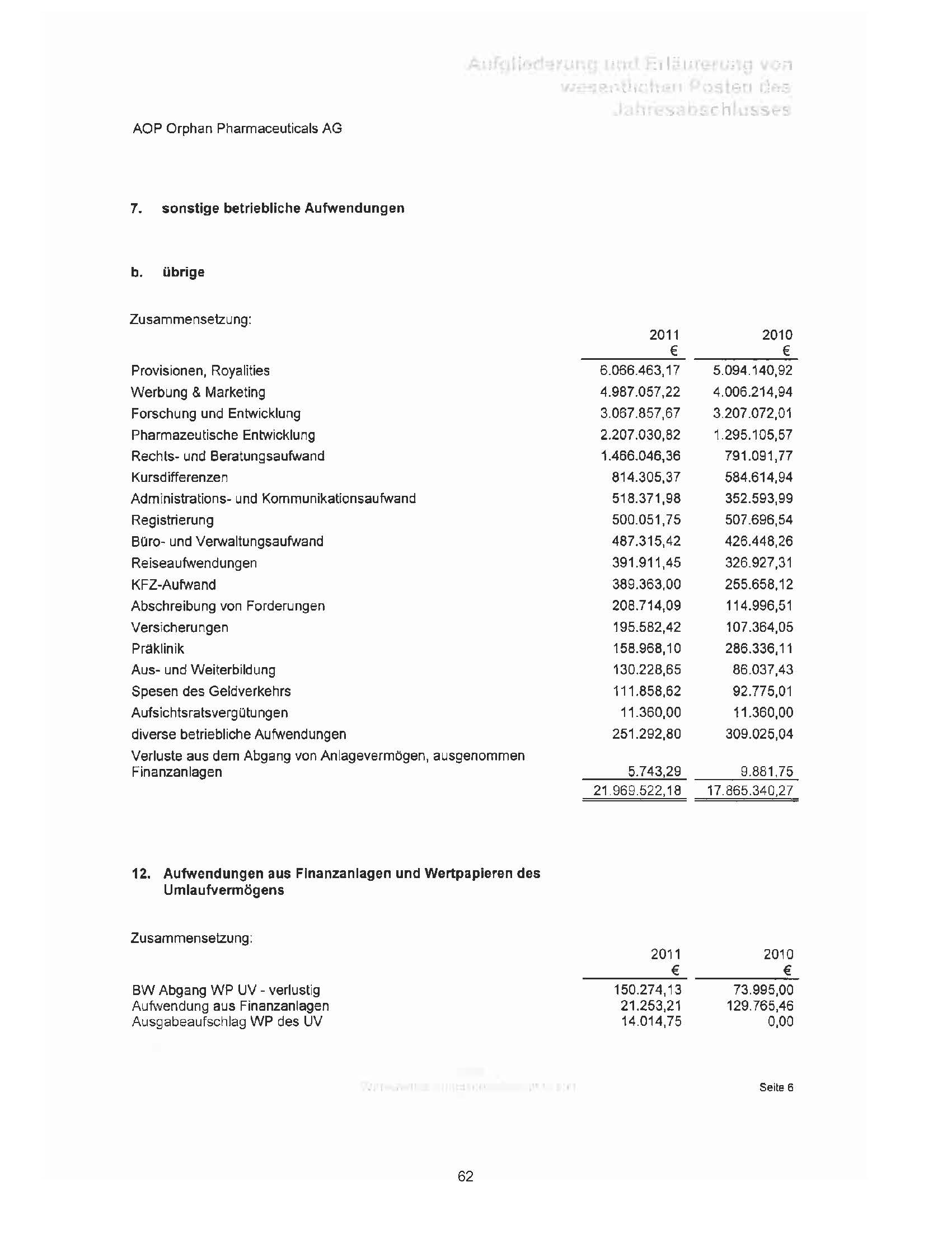










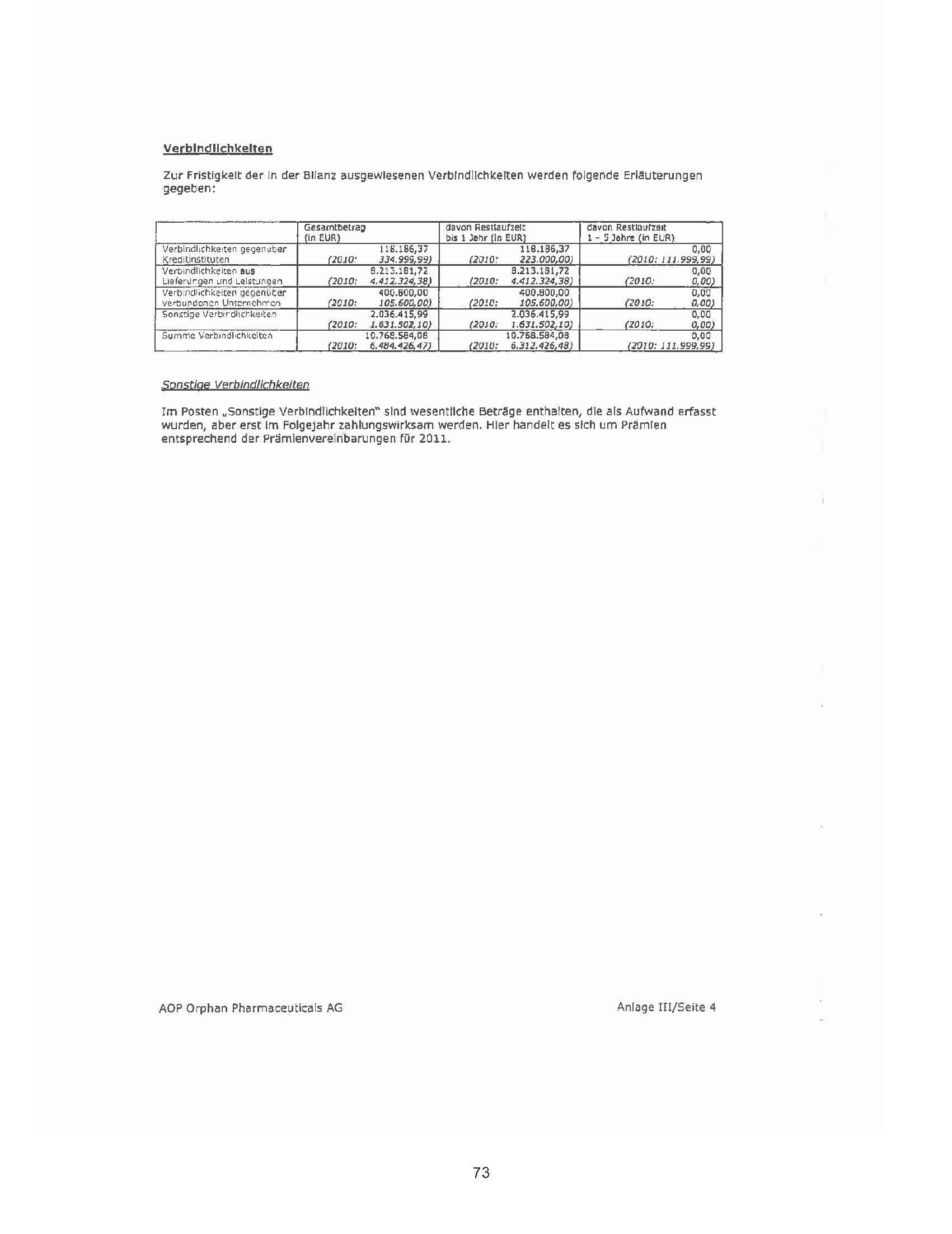



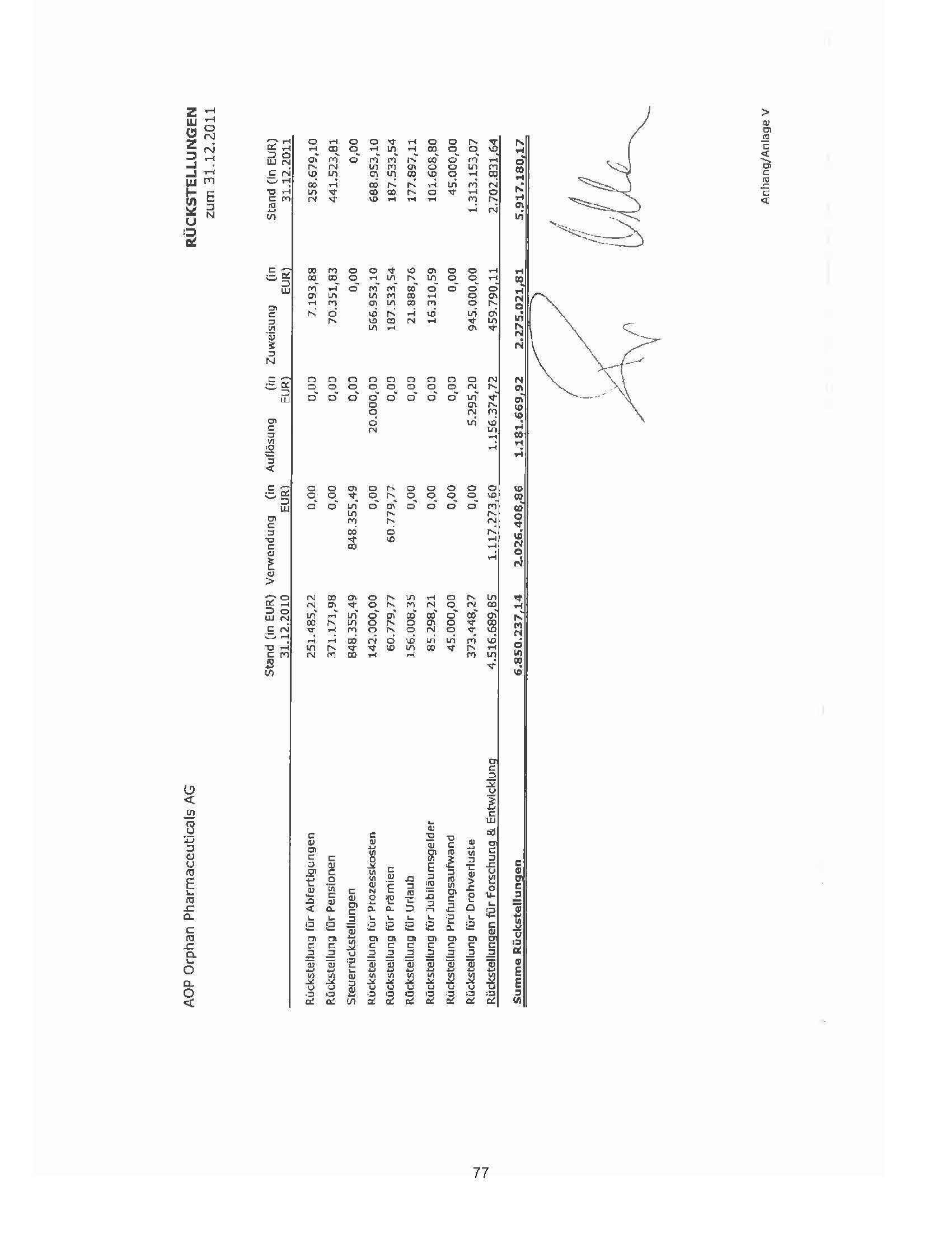




NOTICE OF EXTRAORDINARY GENERAL MEETING
Elan Corporation, plc
(Registered in Ireland under the Companies Acts 1963 to 2012 with registered number 30356)
NOTICE IS HEREBY GIVEN that an extraordinary general meeting (the “Meeting”) of Elan Corporation, plc (“Elan” or the “Company”) will be held at The O’Callaghan Davenport Hotel, 8/10 Merrion Street Lower, Dublin 2, Ireland on 17 June 2013 at 10.00 a.m. (Irish time) to consider and, if thought fit, pass the following Resolutions:
AS ORDINARY RESOLUTIONS
Resolution 1
“THAT the entry by the Company into the Theravance Transaction, (as described and defined in the circular to Elan Shareholders dated 27 May 2013 of which this Notice forms part) (the “Circular”), be and is hereby approved and the Directors, or a duly authorised committee of the Directors, be and are hereby authorised to carry the Theravance Transaction into effect (with such non-material amendments as they shall deem necessary or appropriate) and in connection therewith the Directors of the Company be and are hereby authorised to do or procure to be done any such acts and things on behalf of the Company and its subsidiaries as they consider necessary or expedient for the purpose of giving effect to the foregoing.”
Resolution 2
“THAT the entry by the Company into the AOP Transaction, (as described and defined in the Circular), be and is hereby approved and the Directors, or a duly authorised committee of the Directors, be and are hereby authorised to carry the AOP Transaction into effect (with such non-material amendments as they shall deem necessary or appropriate) and in connection therewith the Directors of the Company be and are hereby authorised to do or procure to be done any such acts and things on behalf of the Company and its subsidiaries as they consider necessary or expedient for the purpose of giving effect to the foregoing.”
Resolution 3
“THAT the entry by the Company into the ELND005 Transaction, (as described and defined in the Circular), be and is hereby approved and the Directors, or a duly authorised committee of the Directors, be and are hereby authorised to carry the ELND005 Transaction into effect (with such non-material amendments as they shall deem necessary or appropriate) and in connection therewith the Directors of the Company be and are hereby authorised to do or procure to be done any such acts and things on behalf of the Company and its subsidiaries as they consider necessary or expedient for the purpose of giving effect to the foregoing.”
Resolution 4
“THAT the entry by the Company into the Share Repurchase Program, (as described and defined in the Circular), be and is hereby approved and the Directors, or a duly authorised committee of the Directors, be and are hereby authorised to carry the Share Repurchase Program into effect (with such non-material amendments as they shall deem necessary or appropriate) and in connection therewith the Directors of the Company be and are hereby authorised to do or procure to be done any such acts and things on behalf of the Company and its subsidiaries as they consider necessary or expedient for the purpose of giving effect to the foregoing.”
By order of the Board

William F. Daniel
Company Secretary
Dated 27 May 2013
Registered Office:
Elan Corporation, plc,
Treasury Building,
Lower Grand Canal Street,
Dublin 2,
Ireland
| 82 |
NOTES FOR HOLDERS OF ORDINARY SHARES
AND FOR INFORMATION PURPOSES ONLY FOR ADS HOLDERS
| 1. | Total voting rights |
At 23 May 2013, being the latest practicable date prior to the publication of this Notice of Extraordinary General Meeting (EGM), the Company’s issued share capital consisted of 510,727,975 Ordinary Shares. The Ordinary Shares carry one vote each. Therefore, the total number of voting rights of the Company at 23 May 2013, was of 510,727,975.
| 2. | Conditions for participating in the meeting |
Every member, irrespective of how many shares they hold, has the right to attend, speak, ask questions relating to the agenda and to vote at the EGM. Completion of a Form of Proxy will not affect your right to attend, speak, ask questions relating to the agenda and vote at the EGM in person. The right to participate in the EGM is subject to the registration of the shares on the Voting Record Date (as set out in note 4 below). For the EGM on 17 June 2013, the Voting Record Date is close of business on 15 June 2013 (or in the case of an adjournment at close of business two days before the time fixed for the adjourned meeting). Changes to entries in the Register after that time will be disregarded in determining the right of any person to attend, speak, ask questions relating to the agenda and/or vote at the meeting.
If you are a registered shareholder, your shareholder reference number is to be found on your Form of Proxy. You will need to use your shareholder reference number and your PIN number to lodge your vote online via the Registrar’s website.
| 3. | Appointment of proxy |
If you cannot attend the EGM in person, you may appoint a proxy (or proxies) to attend, speak, ask questions relating to the agenda and vote on your behalf. For this purpose a personalised Form of Proxy is sent to each member. A member entitled to attend and vote at the above meeting is entitled to appoint one or more proxies to attend, speak and vote on his/her behalf. A proxy need not be a member of the Company. You may appoint the Chairman of the Company or another individual as your proxy. You may appoint a proxy by completing your Form of Proxy, making sure to sign and date the form at the bottom and return it in the pre-paid envelope provided. Forms of Proxy, to be valid, must reach the Registrars to the Company, Computershare Services (Ireland) Limited, Heron House, Sandyford Industrial Estate, Dublin 18, Ireland not later than 10.00 a.m. (Irish time) on 15 June 2013. If you are appointing someone other than the Chairman as your proxy, then you must fill in the details of your representative at the meeting in the box located underneath the wording “I/We hereby appoint the Chairman of the EGM OR the following person” on the Form of Proxy.
Alternatively, you may appoint a proxy electronically, by visiting the website of the Company’s Registrars at www.eproxyappointment.com . You will need your shareholder reference number and your PIN number, which can be found on the lower section of your Form of Proxy.
If you appoint the Chairman or another person as a proxy to vote on your behalf, please make sure to indicate how you wish your votes to be cast by ticking the relevant boxes on your Form of Proxy. If you do not indicate how you wish your proxy to vote (or where additional Resolution or procedural matters are put to the meeting) your proxy may vote or abstain as he or she sees fit. Completing and returning a Form of Proxy will not preclude you from attending and voting at the meeting should you so wish.
| 4. | Voting Record Date for EGM |
The Company, pursuant to Section 134A of the Companies Act, 1963, specifies that only those shareholders registered in the Register as at close of business on 15 June 2013 (or in the case of an adjournment at close of business two days before the time fixed for the adjourned meeting) shall be entitled to attend and vote at the meeting in respect of the number of shares registered in their names at the time. Changes in the Register after that time will be disregarded in determining the right of any person to attend and/or vote at the meeting.
| 5. | How to exercise your voting rights |
As a shareholder, you have several ways to exercise your right to vote:
| 5.1. | By attending the EGM in person; |
| 5.2. | By appointing the Chairman or another person as a proxy to vote on your behalf; |
| 5.3. | By appointing a proxy via the CREST System if you hold your shares in CREST. |
In the case of joint holders, the vote of the senior holder who tenders a vote, whether in person or by proxy, will be accepted to the exclusion of the votes of the other registered holder(s) and, for this purpose, seniority will be determined by the order in which the names stand in the register of members.
| 6. | Tabling draft Resolution |
If you or a group of members hold at least 3% of the issued share capital, representing at least 3% of the total voting rights, of the Company, you or the group of members acting together have the right to table a draft resolution for an item already on the agenda of the EGM subject to any contrary provision in company law.
In order to exercise this right, the text of the draft resolution and evidence of your identity and shareholding must be received by post by the Company Secretary at Elan Corporation, plc, Treasury Building, Lower Grand Canal Street, Dublin 2, Ireland or by email to GeneralMeeting@Elan.com within sufficient time so that it may be dispatched by the Company within the minimum notice period required for the resolution by the Companies Acts unless expressly provided otherwise in the Company’s Articles of Association. A resolution cannot be included in the EGM agenda unless the above requirements are complied with and it is received at either of these addresses by this deadline. Furthermore, members are reminded that there are provisions in company law which impose other conditions on the right of members to propose a resolution at the general meeting of a company.
| 83 |
| 7. | Members’ right to ask questions |
Members have a right to ask questions related to items on the EGM agenda and to have such questions answered by the Company subject to any reasonable measures the Company may take to ensure the identification of members. An answer is not required where: (i) to give an answer would interfere unduly with the preparation for the meeting or the confidentiality and business interests of the Company, (ii) the answer has already been given on the Company’s internet site in a question and answer forum, (iii) it appears to the Chairman of the meeting that it is undesirable in the interests of good order of the meeting that the question be answered or (iv) to give an answer will cause the Company to breach its obligations under the Irish Takeover Rules or any other relevant legislation.
| 8. | How to request/inspect documentation relating to the meeting |
The Circular and the notice of the extraordinary general meeting were issued on 27 May 2013. These documents are also available on the Company’s website, www.elan.com.
Should you wish to be sent copies of documents relating to the meeting, you may request this by telephoning the Company’s Registrars on +353 1 4475107 or by writing to the Company Secretary at the address set out above.
The Memorandum and Articles of Association of the Company may be inspected during normal business hours on any normal working day at the registered office of the Company, Treasury Building, Lower Grand Canal Street, Dublin 2, Ireland, up to and including the date of the EGM and at the EGM itself.
| 84 |
Exhibit (a)(9)
|
For Immediate Release
|
|
|
Investor Relations |
Media Relations |
|
Chris Burns Ph: + 1-800-252-3526 David Marshall Ph:+ 353-1-709-4444
|
Emer Reynolds Ph: + 353-1-709-4022 Jonathan Birt/FTI Consulting Ph: +44-751-559-7858 Jamie Tully/Sard Verbinnen & Co Ph: +1-212-687-8080
|
ELAN ANNOUNCES PUBLICATION OF NOTICE OF
EXTRAORDINARY GENERAL MEETING
DUBLIN, Ireland – May 27, 2013 - Elan Corporation, plc (NYSE: ELN) (“Elan” or the “Company”) today publishes the Notice (including details of the transactions) of an Extraordinary General Meeting of the Company (the “EGM”). The Notice of the EGM together with a Form of Proxy will be issued to shareholders today. The EGM will be held at The O’Callaghan Davenport Hotel, 8/10 Merrion Street Lower, Dublin 2, Ireland on Monday, June 17, 2013 at 10.00 a.m. Details of the business to be transacted at the EGM are contained in the Notice. The Notice will be available on the Company’s website www.elan.com. Copies of the Notice will be submitted to the Irish Stock Exchange and will be available for inspection at the below address.
Company Announcements Office,
Irish Stock Exchange,
28 Anglesea Street,
Dublin 2,
Ireland.
About Elan
Elan is a biotechnology company, headquartered in Ireland, committed to making a difference in the lives of patients and their families by dedicating itself to bringing innovations in science to fill significant unmet medical needs that continue to exist around the world. For additional information about Elan, please visit http://www.elan.com.
The Directors of Elan accept responsibility for the information contained in this announcement. To the best of their knowledge and belief (having taken all reasonable care to ensure such is the case); the information contained in this announcement is in accordance with the facts and does not omit anything likely to affect the import of such information.
Any holder of 1% or more of any class of relevant securities of Elan or of Royalty Pharma may have disclosure obligations under Rule 8.3 of the Irish Takeover Panel Act, 1997, Takeover Rules 2007 (as amended).
Forward Looking Statements
This press release contains forward-looking statements that involve substantial risks and uncertainties. You can identify these statements by the fact that they use words such as “anticipate”, “estimate”, “project”, “target”, “intend”, “plan”, “will”, “believe”, “expect” and other words and terms of similar meaning in connection with any discussion of future financial performance or events. Among the factors that could cause actual results to differ materially from those described or projected herein are the following: risks related to delays or difficulties encountered in obtaining, or the failure to obtain, the approval of Elan’s shareholders for the Theravance, AOP and ELND005 transactions, the possibility that intervening events could arise which could alter the timing, or the ability to consummate the Theravance, AOP or the ELND005 transactions even if Elan shareholder approval is obtained, the risk that third parties could challenge any or all of the transactions, even if the transactions are approved by Elan shareholders and consummated, risks that the transactions do not provide the benefits to Elan that are anticipated, whether Elan can successfully access the capital markets to raise debt financing and, as Elan’s principal source of revenue may remain a royalty on sales of Tysabri, the potential of Tysabri, which may be severely constrained by increases in the incidence of serious adverse events (including death) associated with Tysabri (in particular, by increases in the incidence rate for cases of PML), or by competition from existing or new therapies (in particular, oral therapies), and the potential for the successful development and commercialization of products, whether internally or by acquisition, especially given the separation of the Prothena business which left Elan with no material pre-clinical research programs or capabilities; Elan’s ability to maintain sufficient cash, liquid resources, and investments and other assets capable of being monetized to meet its liquidity requirements; the success of our development activities, and research and development activities in which Elan retains an interest, including, in particular, the impact of the announced discontinuation of the development of bapineuzumab intravenous in mild to moderate Alzheimer’s disease; failure to comply with anti-kickback, bribery and false claims laws in the United States, Europe and elsewhere; difficulties or delays in manufacturing and supply of Tysabri; trade buying patterns; the impact of potential biosimilar competition, the trend towards managed care and health care cost containment, including Medicare and Medicaid; legislation and other developments affecting pharmaceutical pricing and reimbursement (including, in particular, the dispute in Italy with respect to Tysabri sales), both domestically and internationally; failure to comply with Elan’s payment obligations under Medicaid and other governmental programs; exposure to product liability (including, in particular, with respect to Tysabri) and other types of lawsuits and legal defense costs and the risks of adverse decisions or settlements related to product liability, patent protection, securities class actions, governmental investigations and other legal proceedings; Elan’s ability to protect its patents and other intellectual property; claims and concerns that may arise regarding the safety or efficacy of Elan’s product candidates; interest rate and foreign currency exchange rate fluctuations and the risk of a partial or total collapse of the euro; governmental laws and regulations affecting domestic and foreign operations, including tax obligations; whether Elan is deemed to be an Investment Company or a Passive Foreign Investment Company; general changes in United States and International generally accepted accounting principles; growth in costs and expenses; and the impact of acquisitions, divestitures, restructurings, product withdrawals and other unusual items. A further list and description of these risks, uncertainties and other matters can be found in Elan’s Annual Report on Form 20-F for the fiscal year ended December 31, 2012, and in its Reports of Foreign Issuer on Form 6-K filed with the SEC. Elan assumes no obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise.
###
May 28, 2013
Via EDGAR
Securities and Exchange Commission
100 F. Street, N.E.
Washington, D.C. 20549
Attention: Perry J. Hindin
| Re: | Elan Corporation, plc Schedule 14D-9 filed May 15, 2013, as amended May 21, 2013 File No. 005-43481 |
Ladies and Gentlemen:
On behalf of our client, Elan Corporation, plc (“Elan” or the “Company”), we are submitting this letter in response to the written comments of the staff (the “Staff”) of the Securities and Exchange Commission (the “Commission”), dated May 21, 2013 (the “Comment Letter”), with respect to the Solicitation/Recommendation Statement on Schedule 14D-9, as amended (SEC File No. 005-43481) (the “Schedule 14D-9”), filed by Elan with the Commission on May 15, 2013. In connection with this letter responding to the Staff’s comments, we are filing Amendment No. 3 to the Schedule 14D-9 with the Commission.
Set forth below are the headings and text of the comments raised in the Comment Letter, followed by Elan’s responses thereto.
General
| 1. | Based on sections 3.3(a)(i), 6.8, 6.10, 7.2(d) and 7.3(a) of Elan’s Royalty Participation Agreement with Theravance, and given Royalty Pharma’s Rule 2.5 announcement issued on May 20, 2013 that its revised offer is conditioned on Elan shareholders voting against the Theravance transaction and all transactions announced in Elan’s press release filed on May 20, 2013 as Exhibit 99.5 to Elan’s Form 6-K, please amend the Schedule 14D-9 to disclose, if true, that: |
| · | the Theravance Agreement requires Elan’s board of directors to recommend the Theravance transaction and not to change or withdraw such recommendation without Theravance’s prior written consent, irrespective of (i) adverse developments affecting the advisability of Elan’s shareholders approving the Theravance agreement and/or (ii) a determination by the Elan board of directors that the Theravance transaction is not in the best interest of the Elan shareholders; |
| · | Elan’s board of directors cannot recommend Royalty Pharma’s Offer at any price given that the Offer is conditioned on Elan shareholders voting against the Theravance transaction; and |
| · | the failure of Elan’s board of directors to comply with its obligation to recommend the Theravance transaction, or the decision of the Elan's board of directors to change or withdraw such an approval recommendation, could expose Elan to a Theravance claim for damages beyond the $10 million termination fee set forth in the agreement. |
It appears from the terms of Elan’s agreement with Theravance that Elan’s board of directors must support the Theravance transaction no matter the terms of an alternative transaction. If true, please so state and explain how this is consistent with the Elan board’s fiduciary duties to its shareholders. If you disagree with our understanding, please provide support for your conclusion and disclose the circumstances under which the Elan board could support a competing transaction.
Response
Elan supplementally advises the Staff that:
| 1. | Rule 21 of the Irish Takeover Panel Act, 1997, 2007 (as amended) (“Rule 21”) requires that the transactions (the “Theravance Transactions”) contemplated by the Royalty Participation Agreement between Theravance, Inc. and Elan dated May 12, 2013 (the “Theravance Agreement”) be approved by the affirmative vote of the majority of outstanding ordinary shares of Elan represented at a shareholders’ meeting where a quorum is present. Elan has called an extraordinary general meeting of its shareholders to be held June 17, 2013 for this purpose, and to approve certain other transactions entered into by Elan and other matters which also require shareholder approval pursuant to Rule 21 (the “EGM”). |
| 2. | Section 6.8 of the Theravance Agreement provides an undertaking by Elan that the shareholder circular relating to the EGM will contain a recommendation from Elan’s board of directors (the “Elan Board”) to its shareholders to vote in favor of the Theravance Transactions and that such recommendation will not be withdrawn, modified or altered in any way without Theravance’s prior written consent. |
| 3. | If the Elan Board were to fail to comply with its undertaking in Section 6.8 of the Theravance Agreement, such failure to comply - like the failure to comply with any other provision of the Theravance Agreement - could expose Elan to a Theravance claim for damages beyond the $10 million termination payment set forth in Section 7.3(b) of the Theravance Agreement. |
Elan further supplementally advises the Staff that, notwithstanding Royalty Pharma’s suggestions to the Staff, there is no provision of the Theravance Agreement, nor any requirement of Irish law relating to fiduciary duties or otherwise, which prevents the Elan Board from recommending both an offer from Royalty Pharma and the Theravance Transactions should the Elan Board choose to do so.
Elan does not believe that an appropriate framework for analysis is to consider, as the Staff’s comment appears to suggest, the Royalty Pharma revised offer to be an “alternative transaction” to the Theravance Agreement, as if both the Theravance Transactions and the Royalty Pharma revised offer were alternative change of control transactions. To the contrary, the Theravance Transactions are an investment by Elan in a participation interest in a royalty stream, not dissimilar to Elan’s current business of owning a Tysabri royalty. Elan entered into the Theravance Transactions in a negotiated transaction with an arm’s length party in furtherance of Elan’s business and objectives and negotiated terms that it considered, in the totality of the circumstances, to be reasonable. At the time Elan entered into the Theravance Transactions, Royalty Pharma had made an inadequate offer of $10.25 plus a right worth, at most, an additional $1.00 per share, for a maximum offer of $11.25 per share. As Royalty Pharma dabbled with clearly inadequate offers, Elan continued to build its business and executed the Theravance Transactions in furtherance thereof.
Subsequent to the announcement of the Theravance Transactions, Royalty Pharma revised its offer and voluntarily limited its ability to consummate its revised offer by electing to condition its revised offer on Elan shareholder rejection of the Theravance Transactions and other matters which require shareholder approval under Rule 21. It appears to Elan that Royalty Pharma has chosen to attempt to deprive shareholders of the opportunity to both accept the Royalty Pharma revised offer and approve the Theravance Transactions and such other matters. The shareholders can decide for themselves if they wish to approve the Theravance Transactions and such other matters (and will do so at the EGM on June 17); but in any event, Royalty Pharma’s machinations with respect to the conditionality of its revised offer do not somehow convert the Theravance Transactions into an “alternative transaction” implicating fiduciary obligations in a change of control context with the respect to the Theravance Transactions. As noted above, the Elan Board remains able to both continue to recommend the Theravance Transactions and, if it chooses to do so, recommend the Royalty Pharma revised offer.
| -2- |
Elan has filed today Amendment No. 3 the Schedule 14D-9 (the “Amended Schedule 14D-9”). Elan has amended paragraph 8.1 of Exhibit (A)(1) to the Schedule 14D-9 to include the following as the penultimate paragraph thereof:
“The Theravance Agreement provides an undertaking by Elan that the shareholder circular relating to the EGM will contain a recommendation from Elan’s board of directors to its shareholders to vote in favor of the Theravance Transactions and that such recommendation will not be withdrawn, modified or altered in any way without Theravance’s prior written consent. If the Elan board were to breach this provision of the Theravance Agreement, Elan could be exposed to a claim for damages for breach of contract and such damages would not be subject to limitation or “cap” under any provision of the Theravance Agreement.”
In addition, Elan supplementally advises the Staff that it intends to file its shareholder circular with respect to the EGM, which was first distributed to shareholders yesterday, as an exhibit to the Amended Schedule 14D-9.
In addition to the aforementioned responses to the Staff’s comments, the Company acknowledges that:
| · | the Company is responsible for the adequacy and accuracy of the disclosure in the filing; |
| · | Staff comments or changes to disclosure in response to comments will not foreclose the Commission from taking any action with respect to the filing; and |
| · | the Company may not assert Staff comments as a defense in any proceeding initiated by the Commission or any person under the federal securities laws of the United States. |
* * *
Please do not hesitate to contact me at 212-504-6888 with any questions or comments you may have.
Very truly yours,
/s/ Christopher T. Cox
Christopher T. Cox
| cc: | William F. Daniel, |
| Elan Corporation, plc |
| -3- |