Form 10-K: 0001835567-23-000018 compared to 0001835567-22-000010FALSE000183556720212022FYP2Y000183556720212022-01-0120212022-12-310001835567us-gaap:CommonClassAMember20212022-01-0120212022-12-310001835567us-gaap:WarrantMember20212022-01-0120212022-12-31000183556720212022-06-30iso4217:USD000183556720222023-03-2130xbrli:shares0001835567pear:PublicWarrantsMember2022-12-3100018355672021-12-31iso4217:USDxbrli:shares0001835567pear:ThimblePointAcquisitionCorpMemberus-gaap:ProductMember20212022-1201-03012021-12-0300018355672021-12-3100018355672020-12-312022-12-310001835567us-gaap:ProductMember2021-01-012021-12-310001835567us-gaap:ProductMemberpear:CollaborationAndLicenseRevenueMember20202022-01-0120202022-12-310001835567pear:CollaborationAndLicenseRevenueMember2021-01-012021-12-310001835567pear:CollaborationAndLicenseRevenueMemberSubscriptionSupportAndProfessionalServicesRevenueMember2020-01-0120202022-01-012022-12-310001835567pear:SubscriptionSupportAndProfessionalServicesRevenueMember2021-01-012021-12-31000183556720202021-01-012020-12-310001835567us-gaap:ConvertiblePreferredStockMemberus-gaap:PreferredStockMember2019-12-310001835567us-gaap:CommonStockMember2019-12-310001835567us-gaap:AdditionalPaidInCapitalMember2019-12-310001835567us-gaap:RetainedEarningsMember2019-12-310001835567us-gaap:AccumulatedOtherComprehensiveIncomeMember2019-12-3100018355672019-12-310001835567us-gaap:ConvertiblePreferredStockMembersrt:RestatementAdjustmentMemberus-gaap:PreferredStockMember2019-12-310001835567us-gaap:CommonStockMembersrt:RestatementAdjustmentMember2019-12-310001835567srt:RestatementAdjustmentMemberus-gaap:AdditionalPaidInCapitalMember2019-12-310001835567srt:RestatementAdjustmentMember2019-12-310001835567us-gaap:ConvertiblePreferredStockMemberus-gaap:PreferredStockMemberpear:RevisionOfPriorPeriodAdjustedBalanceMember2019-12-310001835567us-gaap:CommonStockMemberpear:RevisionOfPriorPeriodAdjustedBalanceMember2019-12-310001835567pear:RevisionOfPriorPeriodAdjustedBalanceMemberus-gaap:AdditionalPaidInCapitalMember2019-12-310001835567us-gaap:RetainedEarningsMemberpear:RevisionOfPriorPeriodAdjustedBalanceMember2019-12-310001835567pear:RevisionOfPriorPeriodAdjustedBalanceMemberus-gaap:AccumulatedOtherComprehensiveIncomeMember2019-12-310001835567pear:RevisionOfPriorPeriodAdjustedBalanceMember2019-12-310001835567us-gaap:ConvertiblePreferredStockMemberus-gaap:PreferredStockMember2020-01-012020-12-310001835567us-gaap:CommonStockMember2020-01-012020-12-310001835567us-gaap:AdditionalPaidInCapitalMember2020-01-012020-12-310001835567us-gaap:RetainedEarningsMember2020-01-012020-12-310001835567us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-01-012020-12-310001835567us-gaap:ConvertiblePreferredStockMemberus-gaap:PreferredStockMember2020-12-312021-12-310001835567us-gaap:CommonStockMember2020-12-310001835567us-gaap:AdditionalPaidInCapitalMember2020-12-310001835567us-gaap:RetainedEarningsMember2020-12-310001835567us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-12-3100018355672020-12-310001835567us-gaap:ConvertiblePreferredStockMemberPreferredStockMemberus-gaap:PreferredStockMemberConvertiblePreferredStockMember2021-01-012021-12-310001835567us-gaap:CommonStockMember2021-01-012021-12-310001835567us-gaap:AdditionalPaidInCapitalMember2021-01-012021-12-310001835567us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-01-012021-12-310001835567us-gaap:RetainedEarningsMember2021-01-012021-12-310001835567us-gaap:ConvertiblePreferredStockMemberus-gaap:PreferredStockMember2021-12-310001835567us-gaap:CommonStockMember2021-12-310001835567us-gaap:AdditionalPaidInCapitalMember2021-12-310001835567us-gaap:RetainedEarningsMember2021-12-310001835567us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-12-310001835567us-gaap:PrivatePlacementMemberus-gaap:CommonClassAMemberCommonStockMember2021-12-032021-12-032022-01-012022-12-310001835567us-gaap:PrivatePlacementMemberus-gaap:CommonClassAMemberAdditionalPaidInCapitalMember2021-12-032022-01-012022-12-310001835567us-gaap:CommonClassAMemberAccumulatedOtherComprehensiveIncomeMember2021-12-032022-01-012022-12-310001835567us-gaap:CommonClassAMemberRetainedEarningsMember2021-12-032021-12-032022-01-012022-12-310001835567pear:AnchorInvestorMemberus-gaap:CommonClassAMember2021-12-032021-12-030001835567pear:AnchorInvestorMemberus-gaap:CommonClassAMember2021-12-0300018355672021-12-032021-12-03xbrli:pure0001835567pear:COVID19Member2020-12-310001835567pear:COVID19Memberpear:AccruedCompensationAndRelatedBenefitsMember2020us-gaap:CommonStockMember2022-12-310001835567us-gaap:OtherNoncurrentLiabilitiesMemberAdditionalPaidInCapitalMember2022-12-310001835567us-gaap:RetainedEarningsMember2022-12-310001835567us-gaap:AccumulatedOtherComprehensiveIncomeMember2022-12-31pear:product0001835567pear:COVID19Member2020-12-310001835567pear:COVID19Member2021-12-310001835567pear:COVID19MemberAccruedCompensationAndRelatedBenefitsMemberpear:AccruedCompensationAndRelatedBenefitsMemberCOVID19Member2021-12-310001835567pear:COVID19Member2022-12-310001835567us-gaap:EquipmentMember2021-01-012021-12-310001835567srt:MinimumMemberus-gaap:SoftwareDevelopmentMember2021-01-0120212022-01-012022-12-310001835567us-gaap:SoftwareDevelopmentMembersrt:MaximumMemberMinimumMember2021-01-0120212022-01-012022-12-310001835567srt:MaximumMemberus-gaap:SoftwareDevelopmentMember2022-01-012022-12-310001835567us-gaap:FurnitureAndFixturesMember20212022-01-012021-12-312022-12-310001835567us-gaap:CostOfSalesMember2022-01-012022-12-310001835567us-gaap:ResearchAndDevelopmentExpenseMember2022-01-012022-12-310001835567us-gaap:AccountingStandardsUpdate201602Member2022-01-010001835567pear:PrivatePlacementWarrantsMember2021-12-312022-12-310001835567pear:PublicWarrantsMember2022-12-310001835567pear:Customer1Memberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-31xbrli:pure0001835567us-gaap:RevenueFromContractWithCustomerMemberpear:Customer2Memberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001835567pear:Customer3Memberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001835567pear:SubscriptionSupportAndProfessionalServicesRevenueMemberpear:Customer1Memberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001835567pear:Customer1Memberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001835567pear:Customer2Memberus-gaap:RevenueFromContractWithCustomerMemberpear:Customer2Memberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001835567pear:Customer3Memberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001835567us-gaap:RevenueFromContractWithCustomerMemberpear:Customer3MemberAccountsReceivableMemberpear:Customer1Memberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001835567us-gaap:AccountsReceivableMemberpear:Customer2Memberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001835567us-gaap:AccountsReceivableMemberpear:Customer3Memberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001835567us-gaap:AccountsReceivableMemberpear:Customer4Memberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001835567us-gaap:AccountsReceivableMemberpear:Customer1Memberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001835567us-gaap:AccountingStandardsUpdate201602MemberAccountsReceivableMemberpear:Customer2Memberus-gaap:SubsequentEventMembersrt:MinimumMemberCustomerConcentrationRiskMember2022-01-012021-01-012021-12-31pear:segment0001835567us-gaap:AccountingStandardsUpdate201602Memberus-gaap:SubsequentEventMembersrt:MaximumMemberCommonClassAMember20222021-0112-01030001835567pear:ThimblePointAcquisitionCorpMember2021-12-032021-12-030001835567pear:ThimblePointAcquisitionCorpMember2021-12-03pear:Tranche0001835567us-gaap:CommonClassAMemberpear:ThimblePointAcquisitionCorpMember2021-12-032021-12-030001835567pear:TrancheOneMemberpear:ThimblePointAcquisitionCorpMember2021-12-030001835567pear:TrancheTwoMemberpear:ThimblePointAcquisitionCorpMember2021-12-030001835567pear:TrancheThreeMemberpear:ThimblePointAcquisitionCorpMember2021-12-03pear:Day0001835567pear:PublicWarrantsMember2021-12-030001835567pear:PrivatePlacementWarrantsMember2021-12-030001835567pear:PrivatePlacementWarrantsMember2022-01-012022-12-3100018355672021-12-032021-12-030001835567us-gaap:AdditionalPaidInCapitalMember2021-12-032021-12-030001835567us-gaap:SellingGeneralAndAdministrativeExpensesMember2021-12-032021-12-030001835567pear:THMAPublicMemberus-gaap:PrivatePlacementMemberus-gaap:CommonClassAMember2021-12-032021-12-030001835567us-gaap:PrivatePlacementMemberus-gaap:CommonClassAMember2021-12-030001835567us-gaap:CommonClassAMember2021-12-032021-12-030001835567us-gaap:CommonClassAMemberpear:AnchorInvestorMember2021-12-032021-12-030001835567us-gaap:CommonClassAMemberpear:AnchorInvestorMember2021-12-030001835567us-gaap:CommonClassAMemberpear:THMAPublicMember2021-12-030001835567pear:THMAInitialStockholdersMemberus-gaap:CommonClassAMember2021-12-030001835567us-gaap:CommonClassAMemberpear:THMAInitialStockholdersMember2021-12-030001835567pear:THMASponsorMemberus-gaap:CommonClassAMemberForwardPurchaseAgreementToAnchorInvestorMember2021-12-030001835567pear:PIPEInvestorsAndForwardPurchaseAgreementMemberus-gaap:CommonClassAMember2021-12-030001835567pear:LegacyPearEquityholdersMemberus-gaap:CommonClassAMember2021-12-030001835567pear:PublicAndPrivatePlacementWarrantsMember2021-12-0300018355672021-12-030001835567us-gaap:MoneyMarketFundsMember2021-12-310001835567us-gaap:FairValueInputsLevel1Memberus-gaap:MoneyMarketFundsMember2022-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2022-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel2Member2022-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2022-12-310001835567us-gaap:USTreasurySecuritiesMember2022-12-310001835567us-gaap:FairValueInputsLevel1Memberus-gaap:USTreasurySecuritiesMember2022-12-310001835567us-gaap:FairValueInputsLevel2Memberus-gaap:USTreasurySecuritiesMember2022-12-310001835567us-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel3Member2022-12-310001835567us-gaap:CorporateBondSecuritiesMember2022-12-310001835567us-gaap:CorporateBondSecuritiesMemberus-gaap:FairValueInputsLevel1Member2022-12-310001835567us-gaap:CorporateBondSecuritiesMemberus-gaap:FairValueInputsLevel2Member2022-12-310001835567us-gaap:CorporateBondSecuritiesMemberus-gaap:FairValueInputsLevel3Member2022-12-310001835567us-gaap:FairValueInputsLevel1Member2022-12-310001835567us-gaap:FairValueInputsLevel2Member2022-12-310001835567us-gaap:FairValueInputsLevel3Member2022-12-310001835567us-gaap:MoneyMarketFundsMember2021-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2021-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel2Member2021-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2021-12-310001835567us-gaap:CorporateBondSecuritiesMember2021-12-310001835567us-gaap:FairValueInputsLevel1MemberCorporateBondSecuritiesMemberus-gaap:CorporateBondSecuritiesMemberFairValueInputsLevel1Member2021-12-310001835567us-gaap:CorporateBondSecuritiesMemberus-gaap:FairValueInputsLevel2Member2021-12-310001835567us-gaap:CorporateBondSecuritiesMemberus-gaap:FairValueInputsLevel3Member2021-12-310001835567us-gaap:CommercialPaperMember2021-12-310001835567us-gaap:FairValueInputsLevel1Memberus-gaap:CommercialPaperMember2021-12-310001835567us-gaap:CommercialPaperMemberFairValueInputsLevel2Memberus-gaap:FairValueInputsLevel2MemberCommercialPaperMember2021-12-310001835567us-gaap:CommercialPaperMemberus-gaap:FairValueInputsLevel3Member2021-12-310001835567us-gaap:FairValueInputsLevel1Member2021-12-310001835567us-gaap:FairValueInputsLevel2Member2021-12-310001835567us-gaap:FairValueInputsLevel3Member2021-12-310001835567us-gaap:MoneyMarketFundsMember2020-12-310001835567us-gaap:FairValueInputsLevel1Memberus-gaap:MoneyMarketFundsMember2020-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel2Member2020-12-310001835567us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2020-12-310001835567us-gaap:CorporateBondSecuritiesMember2020-12-310001835567us-gaap:FairValueInputsLevel1Memberus-gaap:CorporateBondSecuritiesMember2020-12-310001835567us-gaap:CorporateBondSecuritiesMemberus-gaap:FairValueInputsLevel2Member2020-12-310001835567us-gaap:CorporateBondSecuritiesMemberus-gaap:FairValueInputsLevel3Member2020-12-310001835567us-gaap:CommercialPaperMember2020-12-310001835567us-gaap:FairValueInputsLevel1Memberus-gaap:CommercialPaperMember2020-12-310001835567us-gaap:CommercialPaperMemberus-gaap:FairValueInputsLevel2Member2020-12-310001835567us-gaap:CommercialPaperMemberus-gaap:FairValueInputsLevel3Member2020-12-310001835567us-gaap:FairValueInputsLevel1Member2020-12-310001835567us-gaap:FairValueInputsLevel2Member2020-12-310001835567us-gaap:FairValueInputsLevel3Member2020-12-310001835567srt:MinimumMember2021srt:MinimumMember2022-12-310001835567srt:MaximumMember20212022-12-310001835567us-gaap:MeasurementInputSharePriceMemberpear:SeriesAWarrantsMember2020-12-310001835567pear:SeriesDWarrantsMemberus-gaap:MeasurementInputSharePriceMember2020-12-310001835567pear:SeriesAWarrantsMemberus-gaap:MeasurementInputExpectedTermMember2020-12-31pear:year0001835567pear:SeriesDWarrantsMemberus-gaap:MeasurementInputExpectedTermMember2020-12-310001835567pear:SeriesAWarrantsMemberus-gaap:MeasurementInputPriceVolatilityMember2020-12-310001835567pear:SeriesDWarrantsMemberus-gaap:MeasurementInputPriceVolatilityMember2020-12-310001835567us-gaap:MeasurementInputRiskFreeInterestRateMemberpear:SeriesAWarrantsMember2020-12-310001835567pear:SeriesDWarrantsMemberus-gaap:MeasurementInputRiskFreeInterestRateMember2020-12-310001835567pear:SeriesAWarrantsMemberus-gaap:WarrantMember2019-12-310001835567pear:SeriesDWarrantsMemberus-gaap:WarrantMember2019-12-310001835567pear:SeriesAWarrantsMemberus-gaap:WarrantMember2020-01-012020-12-310001835567pear:SeriesDWarrantsMemberus-gaap:WarrantMember2020-01-012020-12-310001835567pear:SeriesAWarrantsMemberus-gaap:WarrantMember2020-12-310001835567pear:SeriesDWarrantsMemberus-gaap:WarrantMember2020-12-310001835567pear:SeriesAWarrantsMemberus-gaap:WarrantMember2021-01-012021-12-310001835567pear:SeriesDWarrantsMemberus-gaap:WarrantMember2021-01-012021-12-310001835567pear:SeriesAWarrantsMemberus-gaap:WarrantMember2021-12-310001835567pear:SeriesDWarrantsMemberus-gaap:WarrantMember2021-12-310001835567us-gaap:MeasurementInputSharePriceMember2021-12-310001835567us-gaap:MeasurementInputRiskFreeInterestRateMember2022-12-310001835567us-gaap:MeasurementInputSharePriceMember2021-12-310001835567us-gaap:MeasurementInputRiskFreeInterestRateMember2022-12-310001835567us-gaap:MeasurementInputRiskFreeInterestRateMember2021-12-310001835567us-gaap:MeasurementInputExpectedTermMember20212022-12-31pear:year0001835567us-gaap:MeasurementInputExpectedTermMember2021-12-310001835567us-gaap:MeasurementInputPriceVolatilityMember2022-12-310001835567us-gaap:MeasurementInputPriceVolatilityMember2021-12-310001835567us-gaap:MeasurementInputExpectedDividendRateMember2022-12-310001835567us-gaap:MeasurementInputExpectedDividendRateMember2021-12-310001835567pear:ContingentConsiderationLiabilityMember20202021-12-310001835567pear:ContingentConsiderationLiabilityMember20212022-01-0120212022-12-310001835567pear:ContingentConsiderationLiabilityMember20212022-12-310001835567us-gaap:SoftwareAndSoftwareDevelopmentCostsMember2022-12-310001835567us-gaap:SoftwareAndSoftwareDevelopmentCostsMember2021-12-310001835567us-gaap:SoftwareAndSoftwareDevelopmentCostsMemberEquipmentMember20202022-12-310001835567us-gaap:EquipmentMember2021-12-310001835567us-gaap:EquipmentMember2020-12-310001835567us-gaap:ConstructionInProgressMember2021-12-310001835567us-gaap:ConstructionInProgressMember2020FurnitureAndFixturesMember2022-12-310001835567us-gaap:FurnitureAndFixturesMember2021-12-310001835567us-gaap:FurnitureAndFixturesMemberLeaseholdImprovementsMember20202022-12-310001835567us-gaap:LeaseholdImprovementsMember2021-12-310001835567us-gaap:LeaseholdImprovementsMemberConstructionInProgressMember20202022-12-310001835567us-gaap:LineOfCreditMemberConstructionInProgressMember2021-12-310001835567pear:PerceptiveCreditFacilityMemberus-gaap:LineOfCreditMember2020-06-300001835567pear:PerceptiveCreditFacilityTranche1Memberus-gaap:LineOfCreditMember2020-06-300001835567us-gaap:LineOfCreditMemberpear:PerceptiveCreditFacilityTranche2Member2020-06-300001835567us-gaap:LineOfCreditMemberpear:PerceptiveCreditFacilityTranche3Member2020-06-300001835567pear:PerceptiveCreditFacilityMemberus-gaap:LineOfCreditMemberus-gaap:LondonInterbankOfferedRateLIBORMemberpear:PerceptiveCreditFacilityMember2020-06-302020-06-300001835567pear:PerceptiveCreditFacilityMemberus-gaap:LineOfCreditMemberus-gaap:LondonInterbankOfferedRateLIBORMemberpear:PerceptiveCreditFacilityMember20202022-0612-30310001835567us-gaap:LineOfCreditMemberpear:PerceptiveCreditFacilityMemberpear:PerceptiveCreditFacilityMemberus-gaap:LineOfCreditMember2022-12-310001835567pear:PerceptiveCreditFacilityMemberus-gaap:LineOfCreditMember2020-12-310001835567pear:PerceptiveCreditFacilityMemberus-gaap:LineOfCreditMemberpear:DebtCovenant1Member2022-12-310001835567pear:PerceptiveCreditFacilityMemberpear:DebtCovenant2Memberus-gaap:LineOfCreditMembersrt:ScenarioForecastMember2025-03-310001835567srt:ScenarioForecastMember20212023-01-0120212023-1203-310001835567us-gaap:LineOfCreditMemberpear:DebtCovenant1Memberpear:PerceptiveCreditFacilityMembersrt:ScenarioForecastMember2020-06-302023-04-012023-06-300001835567us-gaap:LineOfCreditMemberpear:DebtCovenant2Memberpear:PerceptiveCreditFacilityMembersrt:ScenarioForecastMember2020-06-302023-07-012023-09-300001835567srt:ScenarioForecastMember2023-10-012023-12-310001835567us-gaap:SeriesCPreferredStockMember2020-06-30pear:warrant0001835567pear:SeriesCPreferredStockWarrant2Member2020-06-300001835567pear:SeriesCPreferredStockWarrant1Member2020-06-300001835567us-gaap:SeriesDPreferredStockMember2020-06-300001835567pear:SeriesD1PreferredStockMember2020-06-300001835567us-gaap:SeriesDPreferredStockMember2021-11-302021-11-300001835567pear:SeriesD1PreferredStockMember2021-11-300001835567us-gaap:CommonStockMember2021-11-300001835567us-gaap:CommonClassAMember2021-11-300001835567us-gaap:LineOfCreditMemberpear:PerceptiveCreditFacilityMember2020-06-302020-06-300001835567us-gaap:LineOfCreditMemberpear:PerceptiveCreditFacilityMember2021-12-310001835567pear:TermLoanMemberpear:SVBTermLoanMember2020-06-302020-06-300001835567pear:TermLoanMemberpear:SVBTermLoanMember2020-06-300001835567pear:TermLoanMemberpear:SVBTermLoanMember2020-09-300001835567pear:TermLoanMemberpear:SVBTermLoanMember2019-12-310001835567pear:TermLoanMemberpear:SVBTermLoanMember2018-12-310001835567us-gaap:SeriesAPreferredStockMemberpear:TermLoanMemberpear:SVBTermLoanMember2019-12-310001835567us-gaap:SeriesAPreferredStockMemberpear:TermLoanMemberpear:SVBTermLoanMember2018-12-310001835567pear:TermLoanMemberpear:SVBTermLoanMember2020-12-310001835567us-gaap:SeriesAPreferredStockMemberpear:TermLoanMemberpear:SVBTermLoanMember2016-12-310001835567pear:TermLoanMemberpear:SVBTermLoanMember2021-12-310001835567us-gaap:SeriesAPreferredStockMemberpear:SiliconValleyBankMember2021-12-010001835567pear:SiliconValleyBankMember2021-12-010001835567pear:SiliconValleyBankMember2021-12-020001835567pear:BostonMassachusettsMember2021us-gaap:LineOfCreditMember2020-06-302020-06-300001835567pear:BostonMassachusettsMember2022-12-31utr:sqft0001835567pear:BostonMassachusettsMembersrt:ScenarioForecastMember2022-01-010001835567pear:SanFranciscoCaliforniaMember20212022-12-310001835567pear:RaleighNorthCarolinaMember2021-12-31pear:license_agreement2022-12-310001835567pear:SubleaseMemberpear:BostonMassachusettsMemberus-gaap:SubsequentEventMember2023-01-010001835567pear:SubleaseMemberus-gaap:SubsequentEventMember2023-01-010001835567pear:SubleaseMemberpear:RaleighNorthCarolinaMemberus-gaap:SubsequentEventMember2023-01-060001835567pear:SubleaseMemberus-gaap:SubsequentEventMember2023-01-06pear:license_agreement0001835567pear:InventionScienceFundILLCMember2022-01-012022-12-310001835567pear:InventionScienceFundILLCMember2021-01-012021-12-310001835567pear:InventionScienceFundILLCMemberRed5GroupLLCMemberus-gaap:LicenseAgreementTermsMember2020-01-012020-12-312022-12-310001835567pear:Red5GroupLLCMemberus-gaap:LicenseAgreementTermsMember2021-12-312022-01-012022-12-310001835567pear:Red5GroupLLCMember2021-01-012021-12-310001835567pear:Red5GroupLLCMemberBeHealthSolutionsLLCMemberus-gaap:LicenseAgreementTermsMember20202021-01-0120202021-1203-310001835567pear:BeHealthSolutionsLLCMemberus-gaap:LicenseAgreementTermsMember20202022-01-0120202022-1203-310001835567pear:BeHealthSolutionsLLCMemberus-gaap:LicenseAgreementTermsMember20212022-01-0120212022-12-310001835567pear:BeHealthSolutionsLLCMemberus-gaap:LicenseAgreementTermsMember20212022-12-310001835567pear:DataFoundryCloudSubscriptionMember2021-06-172021-06-170001835567pear:DataFoundryCloudSubscriptionMember20212022-12-310001835567pear:WaypointHealthInnovationsLLCMemberus-gaap:LicenseAgreementTermsMember2022-01-012022-12-310001835567pear:WaypointHealthInnovationsLLCMemberus-gaap:LicenseAgreementTermsMember2022-12-310001835567pear:WaypointHealthInnovationsLLCMemberus-gaap:LicenseAgreementTermsMember2021-0112-0120212022-12-310001835567pear:WaypointHealthInnovationsLLCMemberSoftBankCorpMemberus-gaap:LicenseAgreementTermsMemberpear:PearTherapeuticsIncMember20212022-1203-31150001835567pear:SoftBankCorpMemberus-gaap:CollaborativeArrangementMember20192022-01-0120192022-12-310001835567pear:UpfrontPaymentMemberpear:NovartisMemberus-gaap:CollaborativeArrangementMember2018-03-012018-03-310001835567pear:NovartisMemberus-gaap:SeriesBPreferredStockMember2018-03-012018-03-310001835567pear:NovartisMemberus-gaap:SeriesBPreferredStockMember2018-03-310001835567pear:NovartisMemberpear:ReimbursementMemberus-gaap:CollaborativeArrangementMember2020-01-012020-12-310001835567pear:NovartisMemberus-gaap:CollaborativeArrangementMember2019-01-012019-12-310001835567pear:NovartisMember2018-04-01us-gaap:CollaborativeArrangementMember2018-06-300001835567pear:NovartisMemberus-gaap:CollaborativeArrangementMember2020-06-292020-06-290001835567us-gaap:SeriesAPreferredStockMember2021-12-020001835567us-gaap:SeriesBPreferredStockMember2021-12-020001835567us-gaap:SeriesCPreferredStockMember2021-12-020001835567pear:SeriesD1PreferredStockMember2021-12-020001835567pear:SeriesD2PreferredStockMember2021-12-0200018355672021-12-020001835567pear:SeriesD1PreferredStockMember2021-12-310001835567pear:SeriesD2PreferredStockMember2021-12-310001835567us-gaap:SeriesCPreferredStockMember2021-12-310001835567us-gaap:SeriesBPreferredStockMember2021-12-310001835567us-gaap:SeriesAPreferredStockMember2021-12-310001835567pear:SeriesD1PreferredStockMember2020-11-020001835567pear:SeriesD2PreferredStockMember2020-11-020001835567us-gaap:SeriesDPreferredStockMember2020-11-020001835567us-gaap:SeriesDPreferredStockMember2020-11-022020-11-0200018355672020-11-0200018355672020-11-022020-11-020001835567us-gaap:CommonStockMembersrt:ChiefExecutiveOfficerMember2020-11-022020-11-020001835567us-gaap:CommonStockMemberpear:CertainEligibleEmployeesMember2020-11-022020-11-020001835567us-gaap:CommonStockMemberpear:ChiefExecutiveOfficerAndCertainEligibleEmployeesMember2020-11-022020-11-020001835567us-gaap:CommonStockMemberpear:OtherShareholdersMember2020-11-022020-11-020001835567us-gaap:CommonStockMemberus-gaap:InvestorMember2020-11-022020-11-020001835567us-gaap:SeriesAPreferredStockMember2020-01-012020-03-310001835567us-gaap:SeriesBPreferredStockMember2020-01-012020-03-310001835567pear:SeriesAAndBPreferredStockMember2020-01-012020-03-310001835567us-gaap:SeriesDPreferredStockMember2021-02-230001835567us-gaap:SeriesDPreferredStockMember2021-02-232021-02-230001835567pear:ThimblePointAcquisitionCorpMemberus-gaap:IPOMember2021-02-040001835567pear:ThimblePointAcquisitionCorpMemberSubscriptionSupportAndProfessionalServicesRevenueMember2022-12-310001835567pear:ThimblePointAcquisitionCorpMemberus-gaap:IPOMember2021-02-040001835567us-gaap:CommonClassAMemberpear:ThimblePointAcquisitionCorpMemberpear:PublicWarrantMember2021-02-040001835567pear:ThimblePointAcquisitionCorpMemberus-gaap:CommonClassAMemberus-gaap:IPOMemberpear:PublicWarrantMemberpear:ThimblePointAcquisitionCorpMemberpear:PublicWarrantMemberus-gaap:IPOMember2021-02-040001835567pear:SharePriceMoreThanOrEqualsToUsdEighteenMemberThimblePointAcquisitionCorpMemberpear:ThimblePointAcquisitionCorpMemberSharePriceMoreThanOrEqualsToUsdEighteenMember20212022-01-0120212022-12-310001835567pear:PublicWarrantsMemberpear:ThimblePointAcquisitionCorpMember20212022-12-310001835567pear:SharePriceMoreThanOrEqualsToUsdEighteenMemberus-gaap:CommonClassAMemberpear:ThimblePointAcquisitionCorpMemberus-gaap:CommonClassAMemberpear:SharePriceMoreThanOrEqualsToUsdEighteenMember20212022-12-310001835567pear:SharePriceLessThanOrEqualsToUsdEighteenMemberpear:ThimblePointAcquisitionCorpMember20212022-12-310001835567pear:SharePriceLessThanOrEqualsToUsdEighteenMemberpear:ThimblePointAcquisitionCorpMember20212022-01-0120212022-12-310001835567pear:SharePriceLessThanOrEqualsToUsdEighteenMemberpear:ThimblePointAcquisitionCorpMemberus-gaap:CommonClassAMemberpear:ThimblePointAcquisitionCorpMember20212022-12-310001835567pear:PrivatePlacementWarrantsMember2021-0112-01032021-12-31030001835567pear:ATMOfferingMemberus-gaap:SubsequentEventMember2023-01-010001835567pear:A2013StockIncentivePlanMember2020-11-030001835567pear:A2021IncentiveAwardPlanMember2022-12-310001835567pear:A2021IncentiveAwardPlanMember2021-12-310001835567pear:A2021IncentiveAwardPlanMember2021-01-0120212022-01-012022-12-310001835567pear:A2021IncentiveAwardPlanMemberus-gaap:SubsequentEventMember2023-01-012023-01-010001835567pear:A2021IncentiveAwardPlanMember2022-01-012022-01-010001835567us-gaap:EmployeeStockOptionMember2022-01-012022-12-310001835567us-gaap:EmployeeStockOptionMember2021-01-012021-12-310001835567srt:MinimumMemberus-gaap:EmployeeStockOptionMember2020-01-0120202022-01-012022-12-310001835567srt:MaximumMemberus-gaap:EmployeeStockOptionMember2022-01-012022-12-310001835567srt:MinimumMemberus-gaap:EmployeeStockOptionMember2021-01-012021-12-310001835567srt:MaximumMemberus-gaap:EmployeeStockOptionMembersrt:MaximumMember2021-01-012021-12-310001835567srt:MinimumMemberus-gaap:EmployeeStockOptionMember2020-01-0120202022-01-012022-09-300001835567us-gaap:RestrictedStockUnitsRSUMemberpear:A2021IncentiveAwardPlanMember2022-01-012022-12-310001835567us-gaap:EmployeeStockOptionMemberRestrictedStockUnitsRSUMembersrt:MaximumMember2020pear:A2021IncentiveAwardPlanMember2022-12-310001835567us-gaap:RestrictedStockUnitsRSUMember2021-12-310001835567us-gaap:RestrictedStockUnitsRSUMember2022-01-012020-12-312022-12-310001835567us-gaap:RestrictedStockUnitsRSUMember2022-12-310001835567us-gaap:RestrictedStockUnitsRSUMember2022-07-012022-07-3100018355672022-11-142022-11-14pear:employee0001835567srt:CumulativeEffectPeriodOfAdoptionAdjustmentMember2022-01-012022-12-310001835567us-gaap:EmployeeStockMember2022-01-012022-12-310001835567us-gaap:EmployeeStockMember2021-12-310001835567us-gaap:EmployeeStockMember2022-12-310001835567us-gaap:SubsequentEventMemberus-gaap:EmployeeStockMember2023-01-012023-01-010001835567us-gaap:CostOfSalesMember2021-01-012021-12-310001835567us-gaap:CostOfSalesMember2020-01-012020-12-310001835567us-gaap:ResearchAndDevelopmentExpenseMember2021-01-012021-12-310001835567us-gaap:ResearchAndDevelopmentExpenseMemberSellingGeneralAndAdministrativeExpensesMember20202022-01-0120202022-12-310001835567us-gaap:SellingGeneralAndAdministrativeExpensesMember2021-01-012021-12-3100018355672022-01-012022-03-310001835567us-gaap:SellingGeneralAndAdministrativeExpensesMemberDomesticCountryMember20202022-01-0120202022-12-310001835567us-gaap:CommonStockMembersrt:ChiefExecutiveOfficerMemberForeignCountryMember20202022-01-0120202022-12-310001835567pear:CertainEligibleEmployeesMemberus-gaap:CommonStockMemberNetOperatingLossCarryforwardsResearchAndDevelopmentTaxCreditsAndAccruedExpensesMember20202022-01-012020-12-310001835567us-gaap:CommonStockMemberpear:ChiefExecutiveOfficerAndCertainEligibleEmployeesMember2020-12-310001835567us-gaap:CommonStockMemberpear:ChiefExecutiveOfficerAndCertainEligibleEmployeesMember2020-01-012020-12-310001835567us-gaap:EmployeeStockMember2021-01-012021-12-310001835567us-gaap:EmployeeStockMember2021-12-3100018355672021-01-012021-032022-12-310001835567pear:NetOperatingLossCarryforwardsResearchAndDevelopmentTaxCreditsAndAccruedExpensesMember2021-01-012021-12-310001835567pear:NetOperatingLossCarryforwardsResearchAndDevelopmentTaxCreditsAndAccruedExpensesMember2020-01-012020-12-310001835567us-gaap:DomesticCountryMember20212022-12-310001835567us-gaap:StateAndLocalJurisdictionMember20212022-12-310001835567us-gaap:ResearchMemberDomesticCountryMemberus-gaap:DomesticCountryMemberResearchMember20212022-12-310001835567us-gaap:StateAndLocalJurisdictionMemberus-gaap:ResearchMember20212022-12-310001835567us-gaap:ConvertiblePreferredStockMemberEmployeeStockOptionMember2021-01-012021-12-310001835567us-gaap:ConvertiblePreferredStockMember2020-01-0120202022-01-012022-12-310001835567us-gaap:EmployeeStockOptionMember2021-01-012021-12-310001835567us-gaap:EmployeeStockOptionMemberRestrictedStockMember20202022-01-0120202022-12-310001835567us-gaap:WarrantMemberRestrictedStockMember2021-01-012021-12-310001835567us-gaap:WarrantMemberpear:PrivatePlacementWarrantsMember20202022-01-0120202022-12-310001835567pear:PrivatePlacementWarrantsMember2021-01-012021-12-310001835567pear:PrivatePlacementWarrantsMemberPublicWarrantsMember20202022-01-0120202022-12-310001835567pear:PublicWarrantsMember2021-01-012021-12-310001835567pear:PublicWarrantsMemberEarnOutSharesMember20202022-01-0120202022-12-310001835567pear:EarnOutSharesMember2021-01-012021-12-310001835567pear:EarnOutSharesMember20202022-0107-012520202022-12-310001835567us-gaap:CommonStockMembersrt:ChiefExecutiveOfficerMember2020-12-012020-12-310001835567pear:CertainEligibleEmployeesMemberus-gaap:CommonStockMember2020-12-012020-12-310001835567us-gaap:CommonStockMemberpear:ChiefExecutiveOfficerAndCertainEligibleEmployeesMember2020-12-012020-12-310001835567us-gaap:CommonStockMemberpear:OtherShareholdersMember2020-12-012020-12-310001835567us-gaap:CommonStockMemberus-gaap:InvestorMember2020-12-012020-12-3107-25
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
| | | | | |
| ☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December 31, 20212022 |
OR
| | | | | |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission File Number: 001-39969
Pear Therapeutics, Inc.
(Exact name of Registrant as specified in its charter)
| | | | | | | | |
| 200 State Street, 13th Floor | |
| Boston, MA 02109 | |
| Delaware | (617) 925-7848 | 85-4103092 |
(State or other jurisdiction of
incorporation or organization) | (Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices) | (I.R.S. Employer
Identification No.) |
Securities registered pursuant to Section 12(b) of the Act: | | | | | | | | | | | | | | |
| Title of Each Class | | Trading Symbol(s) | | Name of Each Exchange on Which Registered |
Class A common stock,
par value $0.0001 per share | | PEAR | | The Nasdaq Stock Market LLC |
| Warrants, each exercisable for one share of Class A common stock for $11.50 per share | | PEARW | | The Nasdaq Stock Market LLC |
Securities registered pursuant to section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports); and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | |
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| Non-accelerated filer | ☒ | Smaller reporting company | ☒ |
| | | Emerging growth company | ☒ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☒
No ☒ The aggregate market value of the voting common shares held by non-affiliates of the registrant was approximately $272126.78 million, computed by reference to the closing sale price of the Class A common stock as reported by the Nasdaq Capital Market on June 30, 20212022, the last trading day of the registrant’s most recently completed second fiscal quarter. The Company has no non-voting common shares.
The number of shares of the registrant’s Class A common stock outstanding as of March 2130, 20222023 was 137142,836739,028169.
DOCUMENTS INCORPORATED BY REFERENCE
DOCUMENTS INCORPORATED BY REFERENCE
None. Certain information required to be provided in Part III of this Annual Report on Form 10-K will be provided by a Definitive Proxy Statement for our 2023 Annual Meeting of Stockholders (the “Proxy Statement”) to be filed with the Securities and Exchange Commission on or before May 1, 2023.
Pear Therapeutics, Inc.
Form 10-K
For the Fiscal Year Ended December 31, 20212022
TABLE OF CONTENTS
| | | | | | | | | | | |
| | | Page |
| | | |
| | PART I | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | PART II | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | PART III | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | PART IV | |
| | | |
| | | |
| | | |
References throughout this Form 10-K to “we,” “us,” the “Company,” “Pear,” or “our company” are to Pear Therapeutics, Inc. and its consolidated subsidiaries, and “Legacy Pear” refers to Pear Therapeutics (US), Inc. and its consolidated subsidiary prior to the business combination that we consummated with Thimble Point Acquisition Corp. on December 3, 2021, unless otherwise noted or the context otherwise indicates.
This document contains references to trademarks, trade names, and service marks belonging to other entities. Solely for convenience, trademarks, trade names, and service marks referred to in this Annual Report on Form 10-K may appear without the ® or TM symbols, but such references are not intended to indicate, in any way, that the applicable licensor will not assert, to the fullest extent under applicable law, its rights to these trademarks and trade names. We do not intend our use or display of other companies’ trade names, trademarks, or service marks to imply a relationship with, or endorsement or sponsorship of us by, any other company.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 1
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K (“Form 10-K
CAUTIONARY STATEMENT
In February 2023, we initiated a process to explore a range of strategic alternatives to maximize shareholder value. The Company has engaged MTS Health Partners, L.P. to act as the Company’s exclusive investment banker to assist in evaluating potential alternatives. Potential strategic alternatives that may be evaluated include a sale or merger of the Company, the sale of all or a portion of the Company’s assets and/or intellectual property, or securing additional financing or partnerships that would enable further development of our programs. There is no set timetable for this process and there can be no assurance that this process will result in the Company pursuing a transaction or that any transaction, if pursued, will be completed on attractive terms. Additionally, there can be no assurances that any particular course of action, business arrangement or transaction, or series of transactions, will be pursued, successfully consummated, or lead to increased stockholder value. If the strategic process is unsuccessful, our Board may decide to pursue a liquidation or obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection. In the event of such liquidation, bankruptcy case, or other wind-down event, holders of our securities will likely suffer a total loss of their investment.
As of the date of this filing, Perceptive Credit Holdings III, LP, as administrative agent and lender (“Perceptive”) has alleged that certain defaults or events of default have occurred and are continuing under the terms of our secured Amended and Restated Credit Agreement and Guaranty with Perceptive Credit Holdings III, LP, (“Perceptive Credit Facility”). Perceptive has not delivered any formal notice of Default or Event of Default. To the extent that any such allegations are valid, Perceptive would have certain rights and remedies under the Perceptive Credit Facility. The Company disputes the allegations and is in discussions with Perceptive to resolve this dispute and otherwise to address the Company’s obligations under the Perceptive Credit Facility. There can be no assurances that such discussions will result in any resolution, and any resolution, or the lack of any resolution, may result in Perceptive exercising remedies under the Perceptive Credit Facility.
Pear Therapeutics, Inc. cautions that trading in the Company’s securities is highly speculative and poses substantial risks. Trading prices for the Company’s securities may bear little or no relationship to the actual value realized, if any, by holders of the Company’s securities. Accordingly, the Company urges extreme caution with respect to existing and future investments in its securities.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K (“Form 10-K”) contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, that involve risks and uncertainties (some of which are beyond our control) or other assumptions that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. Many of the forward-looking statements are located in Part II, Item 7 of this Form 10-K under the heading “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” Forward-looking statements provide current expectations of future events based on certain assumptions and include any statement that does not directly relate to any historical or current fact. These risks and uncertainties include, but are not limited to, those factors described under the heading “Risk Factors”. The risks described under the heading “Risk Factors” are not exhaustive. Forward-looking statements can also be identified by words such as “future,” “anticipates,” “believes,” “estimates,” “expects,” “intends,” “plans,” “predicts,” “will,” “would,” “could,” “can,” “may,” and similar terms. Forward-looking statements are not guarantees of future performance and the Company’s actual results may differ significantly from the results discussed in the forward-looking statements. Factors that might cause such differences include, but are not limited to, those discussed in Part I, Item 1A of this Form 10-K under the heading “Risk Factors.” New risk factors emerge from time to time and it is not possible to predict all such risk factors, nor can we assess the impact of all such risk factors on our business or the extent to which any factor or combination of factors may cause actual results to differ materially from those contained in any forward-looking statements. The Company assumes no obligation to revise or update any forward-looking statements for any reason, except as required by law. YouSuch forward-looking statements involve various risks and uncertainties that could cause actual outcomes or results to differ materially from those indicated in these statements. Forward-looking statements in this Form 10-K include, but are not limited to, statements about:
▪plans and expectations for the outcome of strategic alternatives, expectations regarding our strategic alternative review process, and the timing and success of such process regarding a potential transaction;
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 2
▪beliefs about our available options and financial condition;
▪our ability to fund our planned operations for the next twelve months and our ability to continue as a going concern;
▪ expectations that our cash will be sufficient to fund our operating expenses into the future;
▪estimates for our expenses and capital requirements; and
▪our expectations regarding our ability to maintain the listing of our common stock on the National Association of Securities Dealers Automated Quotations (“Nasdaq”) Capital Market.
All of our forward-looking statements are as of the date of this Form 10-K only. In each case, actual results may differ materially from such forward-looking information. We can give no assurance that such expectations or forward-looking statements will prove to be correct. An occurrence of or any material adverse change in one or more of the risk factors or risks and uncertainties referred to in this Form 10-K or included in our other public disclosures or our other periodic reports or other documents or filings filed with or furnished to the Securities and Exchange Commission (the “SEC”), could materially and adversely affect our business, prospects, financial condition, and results of operations. Therefore, you should not place undue reliance upon ouron forward-looking statements. Any public statements or disclosures by us following this Form 10-K that modify or impact any of the forward-looking statements contained in this Form 10-K will be deemed to modify or supersede such statements in this Form 10-K.
Except as required by law, we do not intend to update or change any forward-looking statements as a result of new information, future events, or otherwise.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 23
PART I
BASIS OF PRESENTATION
References throughout this Form 10-K to “we,” “us,” the “Company,” “Pear” or “our company” are to Pear Therapeutics, Inc. (formerly known as Thimble Point Acquisition Corp.), and “Legacy Pear” refers to Pear Therapeutics (US), Inc. prior to the Business Combination, unless otherwise noted or the context otherwise indicates.
On December 3, 2021 (the “Closing”), we consummated a business combination, or the “Business Combination”(the “Business Combination”), pursuant to the terms of the business combination agreement, or “(“Business Combination Agreement””), dated June 21, 2021, by and among the Company (formerly known as Thimble Point Acquisition Corp., or “THMA”), Pear Therapeutics (US), Inc., a Delaware corporation incorporated on August 14, 2013 (“Pear US”) (formerly known as Pear Therapeutics, Inc.) and Oz Merger Sub, Inc., pursuant to which Oz Merger Sub., Inc. (a Delaware corporation and wholly-owned subsidiary of THMA, or “Merger Sub”) merged with and into Pear US, with Pear US surviving as our wholly owned subsidiary. Upon the closing of the Business Combination, THMA changed its name to Pear Therapeutics, Inc. (“Pear” or the “Company”).
Pursuant to the terms of the Business Combination Agreement, each share of Legacy Pear common stock, par value $0.0001 per share (“Legacy Pear Common Shares”) issued and outstanding immediately prior to the Closing, after giving effect to the conversion of all issued and outstanding shares of Legacy Pear preferred stock, par value $0.0001 per share (“Legacy Pear Preferred Shares”) to Legacy Pear Common Shares, were canceled and converted into the right to receive a number of shares of Class A common stock, par value $0.0001 per share of the Company (“Class A common stock”) equal to the number of shares of Legacy Pear Common Shares multiplied by the exchange ratio of approximately 1.47. In addition, all outstanding equity awards of Legacy Pear were converted into equity awards with the option to purchase Class A common stock with the same terms and conditions adjusted by the exchange ratio of approximately 1.47.
Legacy Pear is deemed the accounting predecessor and the post-company successor SEC registrant, which means Legacy Pear financial statements for previous periods are disclosed in this Form 10-K. Future period reports filed with the SEC will include Pear Therapeutics, Inc. and its subsidiaries.
See Note 3, Business Combination, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 34
ITEM 1. BUSINESS
Overview
We have initiated a process to explore a range of strategic alternatives to maximize shareholder value and have engaged professional advisors. The Company has engaged MTS Health Partners, L.P. (“MTS”) to act as the Company’s exclusive investment bank to assist in evaluating potential alternatives. Potential strategic alternatives that may be evaluated include a sale or merger of the Company, the sale of all or a portion of the Company’s assets and/or intellectual property, or securing additional financing or partnerships that would enable further development of our programs. There is no set timetable for this process, and there can be no assurance that this strategic review process will result in our pursuit of any transaction or that any transaction, if pursued, will be completed. Additionally, there can be no assurances that any particular course of action, business arrangement or transaction, or series of transactions, will be pursued, successfully consummated, or lead to increased stockholder value. If the strategic process is unsuccessful, our Board may decide to pursue a liquidation or obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection. In the event of such liquidation, bankruptcy case, or other wind-down event, holders of our securities will likely suffer a total loss of their investment.
Pear is a commercial-stage healthcare company pioneering a new class of software-based medicines, sometimes referred to as Prescription Digital Therapeutics (“PDTs”), which use software to treat diseases directly. Our vision is to advance healthcare through the widespread use of PDTs, and to be the one-stop shop for PDTs offered both by Pear and by other organizations that may choose to hostmake their products onavailable to patients, providers, and payors via our commercial platform, known as PearConnect™.
Recent global trends are converging to highlight a significant unmet need for new and innovative solutions for thedisease treatment of diseasessolutions. We believe that our software-based, data-driven solutions are well suited to satisfy this growing unmet need for the treatment of diseases, including addiction and insomnia. We believe that PDTs have the potential to become a cornerstone of the emerging digital health ecosystem and that PDTs are a transformative new generation of therapeutics.
We believe that Pear’s platform has the potential to provide for discovery, development, and commercialization of PDTs at scale. We have designed our development platform to allow for the repeated advancement of PDTs through U.S. Food and Drug Administration (“FDA”) market authorization. Over time, our commercial platform, PearConnect, is similarly designed to offer the infrastructure needed to distribute PDTs developed by us or others. Due to our first-mover advantage, we believe that Pear is positioned to be a long-term leader in this novel therapeutic class.
Pear is one of the category creators and leaders of the PDT industry, as evidenced by being the first company to receive FDA market authorizationmarket authorization from the US Food and Drug Administration (“FDA”) for a PDT. Our marketed PDTs, reSET, reSET-O, and Somryst, were among the first three PDTs authorized by FDA and address psychiatric indications, an area with significant unmet need. needs. While we continue supporting Somryst, our primary focus is fully commercializing reSET and reSET-O.
Two of Pear’s FDA-authorized PDTs, reSET and reSET-O, treat addiction, which currently affects more than 20nearly 46 million people in the United States (“US”).
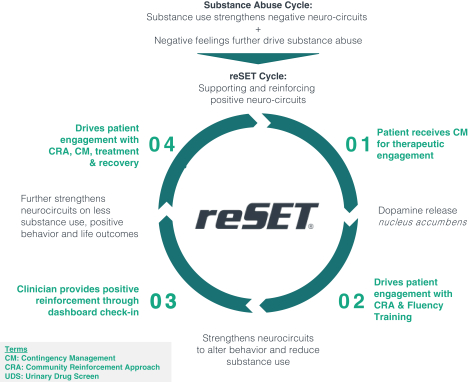
Pear’s first product, reSET, is indicated for the treatment of substance use disorder (“SUD”) as a monotherapy. To combat SUD, reSET works to enhance patient abstinence, improve patient treatment retention relative to human intervention-based alternatives, and extendextends clinicians’ reach outside of scheduled office visits. reSET’s mechanisms of action seek to directly modify addiction-related neurocircuitry and induce dopamine in the brain, a process with the potential to repair dysfunctional neurophysiology.
Pear’s second product, reSET-O, is the first PDT to receive FDA Breakthrough Designation, and is FDA-authorized for the treatment of opioid use disorder (“OUD”) in combination with buprenorphine. Approximately 15.6 million Americans suffer from OUD annually in the US, and approximately 50more than 100,000 Americans die each yearannually from an opioid overdose. To combat OUD, reSET-O works alongside buprenorphine to reduce dependence on opioids, and, similar to reSET, to improve patient treatment adherence and extend clinicians’ reach outside of scheduled office visits.
Pear’sOur third product, Somryst, is the only software-based, FDA-authorized, and guideline-recommended treatment for chronic insomnia. Chronic insomnia is estimated to affect approximately 30 million people in the US. In addition to adversely affecting quality of life, chronic insomnia can also give rise to other serious and life-threatening medical conditions. Many patients turn to sleep medications to combat their chronic insomnia. Most available sleep medications are only recommended for short-term use due to their habit-forming side effects, and, as a result, are not a long-term solution to treat chronic insomnia. We believe that the lack of a convenient and effective solution for chronic insomnia represents a significant unmet medical need that Somryst can help address to reduce the occurrence of chronic insomnia. indicated for the treatment of chronic insomnia. The Company has deprioritized commercialization efforts regarding Somryst while focusing available resources on the commercialization of reSET and reSET-O. During the year ended December 31, 2022, we determined the acquired technology used in our Somryst product that was previously capitalized in intangible assets was impaired and recorded an impairment
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 5
expense of $0.8 million, which is included in cost of revenue in the Company's consolidated statement of operations.
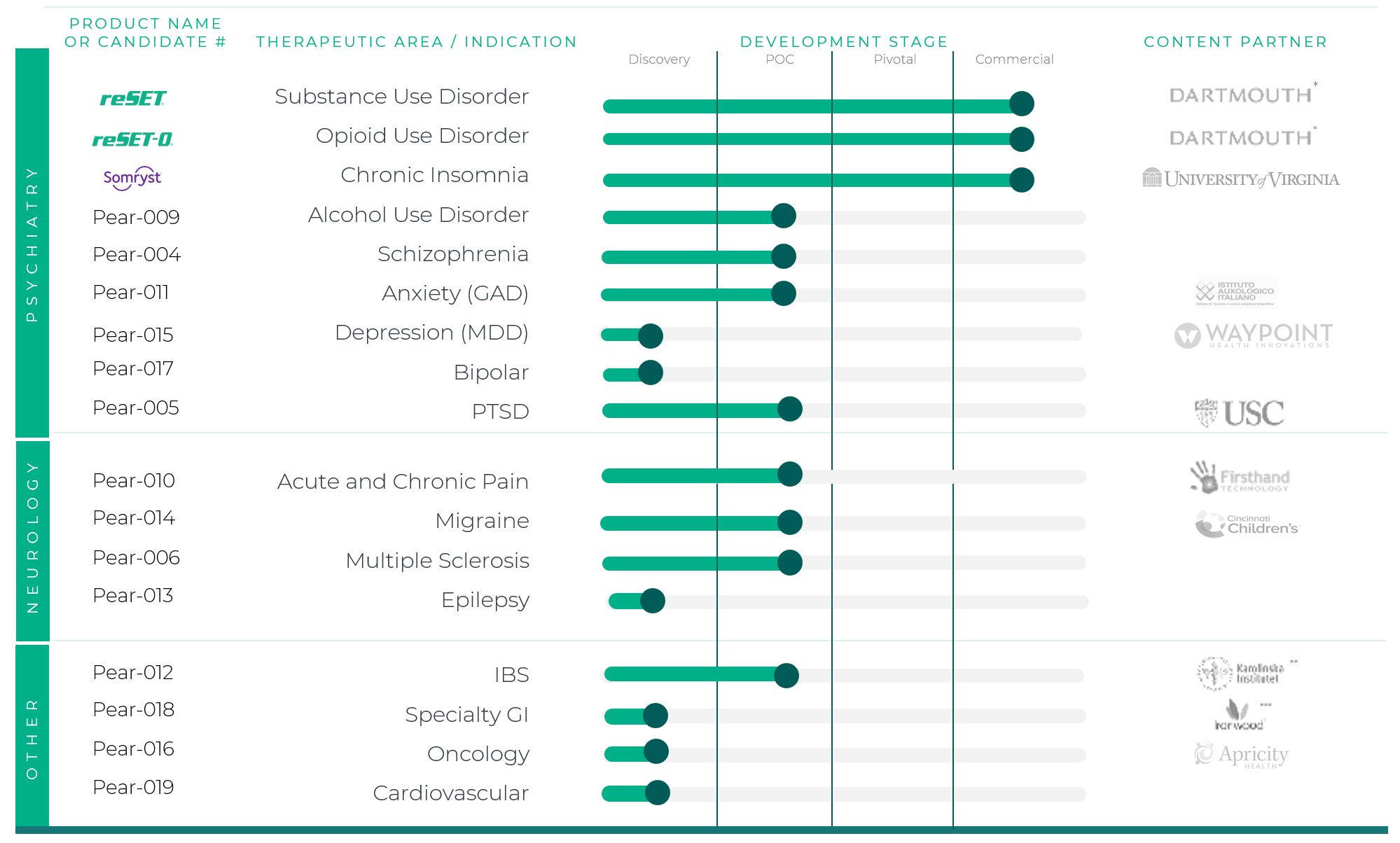
We believe that PDTs have the potential to directly treat a breadth of additional diseases beyond our initial products for the treatment of addiction and insomnia, including the diseases listed in the graphic below. To capitalize on this potential, Pear has developed a robust pipeline of PDTs for a variety of additional indications across psychiatry, neurology, and other therapeutic areas. Pear currently has a pipeline of 14 PDT product
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 4
candidates, as well as the infrastructure and knowledge base needed to continue to expand upon this pipeline, which we believe has the potential to deliver more than one hundred PDTs.
Note: Diseases show above are potential medication conditions that PDTs could address in the future. In July 2022, however, we paused investment in our pipeline candidates to focus investment on our commercial products. We believe our pipeline could provide significant long-term value for patients, providers, payors, and our shareholders if we are able to obtain additional funding or complete a strategic alternative. Due to the worsening macroeconomic environment and the longer-term outlook, in the year ended December 31, 2022, we wrote off $2.1 million of intangible assets related to acquired technology in connection with our pipeline products. We intend to restart investment in our pipeline if we are able to consummate a strategic alternative and if funds become available.
The Prescription Digital Therapeutic Opportunity
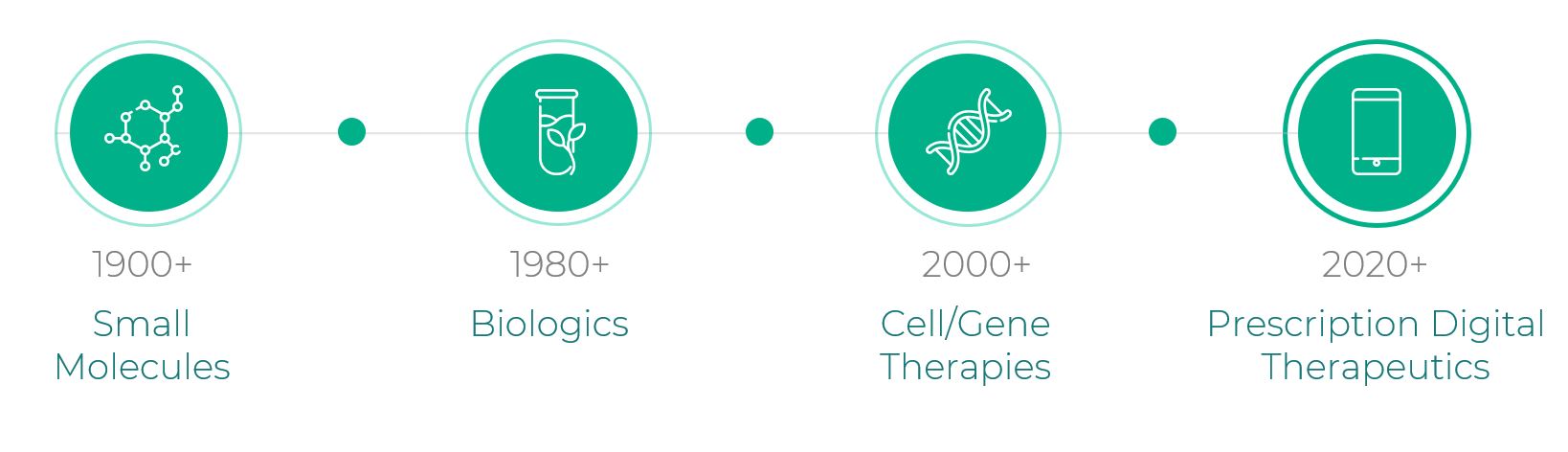
For decades, innovations have expanded the classes of therapeutics to treat disease, from small molecules starting in the 1900s to biologics starting in the late 1970s to cell and gene therapies starting around 2000. This collective innovation has resulted in consistent improvement of health outcomes around the world. Still, across numerous disease states, limited treatment options and substantial barriers to access persist. These barriers include excessive cost, conditions that are difficult to treat with drugs, geographic inaccessibility, and unfavorable side effect profiles.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 5
Pear is advancing PDTs, a novel class of therapeutics, which are software that can be prescribed by a clinician, either alone or in combination with drugs, to treat disease directly. We believe that PDTs are a transformative new class of medicines.

 Similar to pharmaceuticals, novel PDTs undergo rigorous clinical development via clinical trials designed to seek FDA authorization to safely and effectively treat disease. Similar to wellness apps, PDTs utilize digital technology to remotelydirectly interface with patients. PDTs are designed to expand access and convenience for patients, improve reach for clinicians, and reduce cost for payors by reducing and/or augmenting human intervention, providing for more efficient care, and by ideally improving clinical outcomes.
Similar to pharmaceuticals, novel PDTs undergo rigorous clinical development via clinical trials designed to seek FDA authorization to safely and effectively treat disease. Similar to wellness apps, PDTs utilize digital technology to remotelydirectly interface with patients. PDTs are designed to expand access and convenience for patients, improve reach for clinicians, and reduce cost for payors by reducing and/or augmenting human intervention, providing for more efficient care, and by ideally improving clinical outcomes. Currently, we believe the medical community is embracing the integration of software into the navigation and delivery of care. PayorsWe believe payors are increasingly recognizing the near- and long-term cost benefitbenefits of using software for the treatment of diseases. The pervasiveness of technology and growth of telehealth allows for virtual access that was not previously available. As the category creator and leader in PDTs, Pear is defining and expanding the opportunities in this market due to factors which include, but are not limited to:
•being the first mover and leader in the PDT space, defining the industry via the first three FDA-authorized products.;
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 6
•having products in major markets, with reSET and reSET-O for the treatment of addiction, and Somryst for the treatment of chronic insomnia, with the potential to address, in the aggregate, more than 50 million US patients and more than 850 million patients worldwide.;
•having a deep and broad pipeline of PDTs, with 14 product candidates with the potential to redefine care across a range of therapeutic areas via diverse mechanisms of action.
•having the first scalable end-to-end platformability to implement scalable end-to-end platforms (PearCreate and PearConnect) that are designed to discover, develop, and deliver PDTs to patients, creating the horizontal infrastructure to enable our PDTs, and potentially those of other companies, to come to market and be delivered to patients. Pear’s platform also has;
•having the potential to integrate into care delivery and payor infrastructure with virtuous network effects and modularity, facilitating speed and scale in PDT deployment.; and
•demonstrating adoption by patients, clinicians, and payors, which could be leveraged across various future opportunities.
Products and Pipeline
Products
PDTs are software applications authorized by FDA that are intended to treat disease. PDTs are designed to be prescribed by clinicians, reimbursed by third-party payors, and used by patients to improve clinical outcomes as part of a patient’s care, similar to FDA approved medications and medical devices. PDTs are authorized to deliver evidence-based mechanisms-of-action, such as cognitive behavioral therapy, contingency management, and exposure therapy, that the patient engages with on their mobile device and may be used alone or in combination with medications. The value of Pear’s FDA-authorized PDTs is supported by evidence demonstrating safety and
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 6
clinical effectiveness in randomized control trials, collected data on PDT usage and clinical outcomes in real-world data, and health economic value.
We believe PDTs can be utilized in the treatment of a wide variety of diseases. Pear’s first three FDA-authorized products address behavioral health indications:
•reSET is authorized in the US and Singapore for the treatment of substance use disorder related to alcohol, cannabis, cocaine, and stimulants (such as methamphetamine).
•reSET-O is authorized in the US for use in combination with buprenorphine in the US for the treatment of opioid use disorder and was the first PDT to receive FDA Breakthrough Designation.
•Somryst is the only software-based FDA-authorized and guideline-recommendedin the US for treatment for chronic insomnia and the first PDT submitted through FDA’s traditional 510(k) pathway while simultaneously being reviewed as part of FDA’s Software Precertification Pilot Program.of chronic insomnia.
The Company has deprioritized commercialization efforts regarding Somryst while focusing available resources on the commercialization of reSET and reSET-O. During the year ended December 31, 2022, we determined the acquired technology previously capitalized in intangible assets and used in our Somryst product offering was impaired and recorded an impairment expense of $0.8 million, which is included in cost of revenue in the Company's consolidated statement of operations.
For the year ended December 31, 2022, the majority of our revenue was generated through Access Agreements, and three customers: a state government agency, a state department, and a state, representing 18%, 14%, and 11%, respectively, of total revenue. Additionally, all of our subscription, support, and professional services revenue, which represented 11% of total revenue, came from one customer, the Medicaid program of a state government. For the year ended December 31, 2021, the majority of our revenue was generated through bulk purchases from three customers: a state government, state department and a state agency, representing 34%, 23%, and 10%, respectively, of total revenue.
As of December 31, 2022, four customers: a state government, a state department, and two state agencies, representing 33%, 21%, 20%, and 11%, respectively, of accounts receivable. As of December 31, 2021, two customers, a department of a state and the Medicaid program of a state government, represented 33% and 32% of accounts receivable, respectively.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 7
Pipeline
In July 2022, we paused investment in our pipeline to focus investment in our commercial products. We believe our pipeline could provide significant long-term value for patients, providers, payers, and our shareholders if sufficient funding were available. In addition, we would need to increase revenues, and the macroeconomic environment would need to improve before further investments in the pipeline could be made. We continue to market reSET and reSET-O and support Somryst. In the fourth quarter 2022, we recorded an impairment in our intangibles assets of $2.1 million related to certain acquired technology for Pear-015, our depression product candidate, that was capitalized during the year ended December 31, 2021 sometimes referred to as our Waypoint asset.
Our pipeline consists of 14 product candidates, including candidates in psychiatry, neurology, and outside of central nervous system therapeutic areas such as gastrointestinal (“GI”), oncology, and cardiovascular. We are initially focusing on psychiatric and neurologic conditions, which supports our strategy to commercialize our own products.
All of Pear’s product candidate development is, at present,PDT product candidate development is conducted in humans. As therapeutic candidates consisting of software, thereThere are no in vivo animal or in vitro pre-clinical or non-clinical studies. This reduces the translational risk of going from animals to humans that is encountered by many biotechnology and typical pharmaceutical companies encounter.
The pipeline chart below uses four stages of development: Discovery, Proof of Concept (“POC”), Pivotal, and Commercial. Because PDTs are regulated as medical devices, these four stages align with guidance from the Center for Devices and Radiological Health (“CDRH”), which has two broad stages of clinical development before commercialization—pre-pivotal (such as feasibility, including first-in-human) and pivotal (generates definitive evidence of the safety and effectiveness for a specified intended use). Pear breaks down the pre-pivotal clinical development stage into Discovery and POC.
Pre-commercial development at Pear is segregated into three stages: Discovery, POC, and Pivotal. Pear refers to the stage before clinical development as Discovery. Discovery is the concept stage, in which the product candidate, its mechanism(s) of action, and target patient population(s) are defined, technical capabilities and prototypes are built, and then the candidate concept is rapidly tested in iterative evaluations. Some Discovery stage programs are focused on a specific indication, and others are focused on a disease area that may be refined based on further research. POC is the early clinical development stage, in which the product candidate is being tested in human clinical trials designed to prove that the candidate concept is worthy of advancement to the Pivotal stage. POC stage activities are related to technical work, study design, planning, other operational clinical trial activities, and statistical analysis. Pivotal is the registrational phase, in which the product candidate is tested in a randomized controlled trial designed to support market authorization from a regulatory authority such as FDA. Pivotal stage activities are related to technical work, study design, planning, other operational clinical trial activities, and/or statistical analysis that are part of a regulatory submission. Commercial stage includes any products which have received market authorization from FDA.
Pear seeks out and licenseshas historically licensed content suitable for the development of PDTs that has beenthird parties have initially developed by third parties. These parties, or content partners, are primarily academics or academic institutions, and Pear’s current partners are listed in the table below.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 78
At present, other than immaterial investments, we have paused all development activities in our pipeline product candidates unless and until we consummate a strategic alternative. Below is a summary of our products, product candidates, and partners:
*Dartmouth transaction is with a researcher employed by Dartmouth. Pear has no direct contractual relationship with Dartmouth relating to this content.
**Karolinska transaction is with individual researchers who are employed by the Karolinska Institute. Pear has no direct contractual relationship with the Karolinska Institute relating to this content.
***Services agreement with Ironwood to evaluate a PDT in GI diseases.
As reflected above, Pear has licensed or otherwise acquired rights in content suitable for the development of PDTs from a variety of content partners. For further information concerning content collaborations, see the section below entitled ““License Agreements.”.”
Our agreement involving the University of Virginia relates to our license agreement with BeHealth Solutions, LLC and the relationship between Pear and the University of Virginia Patent Foundation d/b/a University of Virginia Licensing & Ventures.
Our license with Red 5 Group LLC relates to our content collaboration involving a researcher employed by Dartmouth College.
Pear licenses certain content related to the treatment of irritable bowel syndrome (“IBS”) developed by individual researchers employed by the Karolinska Institute who founded a company called Hedman-Lagerlöf Och Ljótsson Psykologi AB (“NCAB”) and assigned intellectual property rights in and to the IBS-related content to NCAB. Under Pear’s license agreement with NCAB, entered into on December 14, 2019, Pear was granted an exclusive license in the US and a non-exclusive license throughout the rest of the world to develop and commercialize digital therapeutic products incorporating NCAB’s IBS-related content, in exchange for an upfront payment in the mid-five figures, one-time milestone payments in low-six figures upon delivery to Pear of updated IBS-related content and upon first FDA approval of such a therapeutic product, and a royalty percentage in the low-single digits on net sales of such therapeutic products.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 9
Pear licenses contain content related to the treatment of stress disorders from the University of Southern California (“USC”). Under the Technology License and Distribution Agreement entered into with USC on January 11, 2016, Pear was granted worldwide exclusive rights to develop drug/software combination products incorporating that
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 8
content to treat disorders, illnesscontent to treat disorders, illness, or trauma, in addition to certain worldwide non-exclusive rights. Pear initially paid USC an upfront fee in the low-five figures and agreed to pay a royalty percentage in the low-to-mid single digits on net sales of licensed products, an annual license fee (creditable against royalty payment obligations in the same year) increasing in amount from the low-six figures in 2018 to the low-seven figures in 2021, and regulatory and commercialization milestone payments that, in the aggregate, could reach a total in the low-seven figures. On February 11, 2019, Pear notified USC of its election no longer to pay the annual license fee, thus converting the exclusive license granted by USC to a non-exclusive license.
Pear licenses certain containcontent related to the treatment of mental health conditions, including anxiety and depression from Instituto Auxologico Italiano (“IAI”). Under a software license agreement dated March 23, 2015, IAI granted Pear worldwide exclusive rights to develop drug/software combination products incorporating that content, in addition to certain worldwide non-exclusive rights. Pear agreed to pay IAI a royalty percentage in the low-to-mid single digits on net sales of licensed products, subject to an annual minimum revenue threshold in the low-six figures beginning in the fifth year of the license to maintain exclusivity; as this threshold was not met, Pear’s license from IAI is now non-exclusive. Pear has obtained additional software, documentation, and other intellectual property rights relating to the therapeutic treatment of depression from Waypoint Health Innovations, LLC (“Waypoint”) under an Assignment Agreement and Intellectual Property License Agreement dated November 30, 2021 (collectively, the “Waypoint Agreement”). Under the Waypoint Agreement, Pear acquired all rights related to a CBT-based depression app and a license to all intellectual property rights owned or controlled by Waypoint relating to the app. Pear agreed to pay an upfront payment, regulatory and commercial milestones, a royalty percentage in the low-single digits on net sales, and is required to make annual payments starting in the second half of 2022 through 2026 or until a commercial milestone payment is made under the agreement.
Pear licenses certain content related to the treatment of migraine headaches from Children’s Hospital Medical Center, d/b/a Cincinnati Children’s Hospital Medical Center (“CHMC”). Under a license agreement entered by the parties on December 17, 2019, CHMC granted a worldwide, non-exclusive license to Pear to develop and commercialize therapeutic products incorporating CHMC’s migraine-related content in exchange for an upfront payment in the mid-five figures and Pear’s agreement to pay CHMC one-time regulatory and commercial milestone payments that, in the aggregate, could reach a total in the mid-six figures, and a royalty percentage in low-single digits on net sales of such therapeutic products.
Under a services agreement dated January 9, 2020, with Apricity Health, LLC (“Apricity”), a company engaged in the business of creating digital health therapeutic solutions for improving cancer treatment, Pear has a right of first offer to negotiate terms of a license with Apricity to develop and commercialize each of the first two commercial products developed by Apricity.
Under a services agreement with Ironwood Pharmaceuticals, Inc. (“Ironwood”) dated October 25, 2019, Ironwood agreed to pay Pear a sum not to exceed the low-six figures to develop initial concepts for PDTs for treatment of gastrointestinal indications.
Under an assignment agreement dated January 15, 2019, Pear acquired rights in certain assets primarily related to the treatment of acute and chronic pain from Firsthand Technology, Inc. (“Firsthand”) (the “Firsthand Agreement”). In exchange for acquiring those assets, Legacy Pear issued Legacy Pear Common Shares to Firsthand, agreed to pay an amount capped in the low-six figures to certain creditors of Firsthand, and agreed to pay a one-time milestone payment in the mid-six figures to Firsthand upon first commercial sale of an FDA-cleared Product (as defined in the Firsthand Agreement) and to pay a royalty percentage in the low-single digits on net sales of such Products. Other than the agreements with ISF, Red 5, and BeHealth, Pear does not consider any of the foregoing agreements to be material for purposes of Item 601(b)(10) of Regulation S-K.
Pear’s PDTs and its product candidates achieve their therapeutic potential by delivering evidence-based mechanisms-of-action, such as standard behavioral treatments. For example, reSET delivers an addiction-specific form of cognitive behavioral therapy called Community Reinforcement Approach (“CRA”), fluency training, and contingency management (“CM”). Somryst delivers a disease-specific intervention called Cognitive Behavioral Therapy for Insomnia (“CBTi”), which is also the first-line treatment recommended for patients with chronic insomnia in the American College of Physicians and the American Academy of Sleep Medicine’s clinical guidelines for patients with chronic insomnia. Across Pear’s pipeline, the product candidates each deliver various forms of standardized and disease-specific behavioral treatments such
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 10
disease-specific behavioral treatments such as cognitive behavioral therapy (“CBT”), behavioral activation (“BA”), exposure therapy, and/or cognitive restructuring. Product candidates that are intended to treat diseases with standard-of-care pharmacotherapy, such as schizophrenia, for example, would make recommendations on medication usage to support overall care.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 9
Pear’s products and product candidates may be studied both in trials sponsored and conducted by Pear and trials that are conducted by non-Pear researchers in collaborative and investigator-initiated trials. In the fourth quarter of 2022 Pear is currently enrolling patients in a study that it is sponsoring and conducting on its owncompleted a study involving gamification of reSET-O, which iswas an outpatient-based, randomized-controlled, open-label study conducted at two addiction treatment programs of a gamified-version of reSET-O. This trial iswas partially supported by a grant from the National Institute on Drug Abuse. The study objectives arewere (i) to evaluate participant engagement data [Time Frame: From Week 1 to Week 8 (End of Treatment)], and (ii) to evaluate the number of active sessions per week between PEAR-008 (gamified version) and reSET-O.
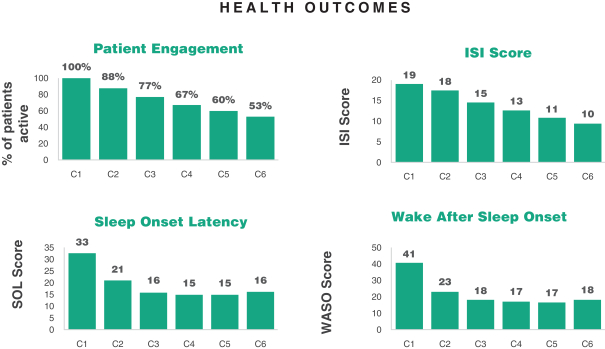
Pear temporarily paused patientcompleted enrollment inof itsthe DREAM trialStudy atin the end of 2021 to make improvements to its development platform in order to gather additional follow up data. Pear received approval for their institutional review board protocol amendment on February 2, 2022,third quarter of 2022 and will began to re-enroll patients (withLast Patient Last Visit (the an enrollment goaldate that the of 1,500 patientslast subject completed the study) in DREAM, which is an open-label, 9-week treatment, de-centralized trial to collect real-world evidence for Somrystthe fourth quarter of 2022. The study objectives are (i) to measure the change in the Insomnia Severity Index (“ISI”) [Time Frame: From baseline to Day 63 (End of Treatment) and Days 243, 428, 610, and 793 (Follow-up)], and (ii) to measure the change in the ISI total score from baseline to end of treatment and follow-up. The ISI’s total score ranges from 0 (not clinically significant) to 28 (clinically significant). Long-term follow-up assessments are ongoing. If we do not receive additional funding in the future, we may not be able to complete this study and other studies.
Key Elements of Our Growth Strategy
Our immediate strategy is to continue our strategic process to explore strategic alternatives.
We have initiated a process to explore a range of strategic alternatives to maximize shareholder value and have engaged professional advisors, including an investment bank, to act as strategic advisors for this process. Potential strategic alternatives that may be evaluated include a sale or merger of the Company, the sale of all or a portion of the Company’s assets and/or intellectual property, or securing additional financing or partnerships that would enable further development of our programs. There can be no assurance that this strategic review process will result in our pursing any transaction or that any transaction, if pursed, will be completed. If the strategic process is unsuccessful, our Board may decide to pursue a liquidation or obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection.
Our mission is to use software to treat disease directly and increase access to care for patients, including underserved constituents of the healthcare ecosystem. WeIn the event our strategic process is successful and we obtain sufficient funding, we plan to accomplish this goal over time by developing, licensing,:
•Focusing on selling reSET and acquiring PDTs, and building the preferred PDT commercialization platform for use by prescribing clinicians. We aim to be the one-stop shop for PDTs offered both by Pear and by other organizations that may choose to host their products on our commercial platform. To achieve our missionreSET-O to states in the near term. Most of our 2022 revenue came from sales of our addiction products directly to states and state agencies. We generated additional revenue in 2022 from state Medicaid reimbursement for our addiction products. By focusing on the most mature aspect of our business model in the near-term, we are pursuing the following strategies:intend to grow revenue while containing costs. We believe our pipeline and platforms provide optionality that will allow us to invest in significant long-term value-creation when our revenues and financing supports such investments.
•Capitalize on our leadership position in the PDT market. By obtaining FDA market authorization for the first three PDTs, we established ourselves as the leader and pioneer of a new category. We currently have 14 additional product candidates in our pipeline, andWith sufficient funding, we intend tobelieve we could capitalize on our leadership position via horizontal scale.
•Continue to invest in our efforts to create the primary commercial platform through which patients and clinicians access PDTs. We are pioneering new commercialization approaches for PDTs. We seek to capture the best of tech and life science approaches to create a scalable and capital-efficient model across our existing portfolio and downstream pipeline. To date, our sales, marketing, and medical affairs teams have engaged with more than 690 clinicians who have written prescriptions for one or more of our three FDA-authorized products. We expect that number will grow proportionally in response to increasing traction with payors and growth in the number of commercially available PDTs. In addition, we recently began piloting direct-to-patient engagement via our virtual care platform, which allows patients to obtain a prescription without going to a clinician’s office.
•Establish reimbursement pathwaysEstablish reimbursement pathways and payment infrastructure. . To date, we have 30 organizations across over 31.7 million individuals with healthcare coverage providingprovide access to our three FDA-authorized products via listing on formularycertain formularies, as a covered benefit, through bulk purchasepurchases, or by funding a study. Pear’s payor strategy focuses across all major payor channels, including employers, Integrated Delivery Networks (“IDNs”), pharmacy benefit managers (“PBMs”), commercial payors, and government payors including Medicaid and Medicare.
•Opportunistically license and acquire PDT product candidates and host third-party PDT products. We aspire to host products from other PDT companies on our multi-product Pear MD™ Clinician Dashboards and technology infrastructure. This has the potential to create a PDT marketplace for all patients to access safe, effective, and secure digital therapeutics. We believe we will be the acquirer and licensing partner of choice for other PDT companies and academia. Our development platform is designed to bring product candidates through development, and our commercialization platform could afford long-term commercial success. We will continue to opportunistically pursue acquisitions and licensing opportunities to grow our developmental pipeline. Pear’s experience developing, including Medicaid and Medicare. Pear works with the payor channel to cover Pear’s products through both the pharmacy and guiding PDTsmedical benefit as well as adjudication through the regulatory reviewboth pharmacy and medical operations.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 1011
process, as well as our scalable platform, provide a foundation on which Pear could potentially develop and host more than a hundred additional PDTs.
•Protect our intellectual property. In addition to securing regulatory authorization for our PDTs, we make strategic use of various intellectual property regimes: patents; copyrights; trademarks; and trade secrets. We strive to protect and enhance the proprietary technology, inventions, and improvements that are commercially important to the development of our business, including seeking, maintaining, and defending patent rights, whether developed internally or licensed from third parties.
•Expand outside the United StatesOver time, subject to obtaining necessary funding, advancing PearConnect as a key commercial platform through which patients and clinicians access PDTs. We are pioneering new commercialization approaches for PDTs. Subject to obtaining sufficient funding, we aspire to connect other product candidates from third-party PDT companies onto PearConnect. This has the potential to create a PDT marketplace for the patients of other PDT manufacturers to access safe, effective, and secure digital therapeutics.
•Subject to obtaining necessary funding, restart our efforts to opportunistically license and acquire PDT product candidates and connect third-party PDT candidates. Today, we operate only in the US. At this pointSubject to obtaining necessary funding, we intend to expandrecommence our capabilitiesefforts to support ex-US partners who would commercialize specific products in specific regions. We plan on evaluating potential partners on a geography-by-geography basisopportunistically pursue acquisitions and licensing opportunities to grow our developmental pipeline. Pear’s experience developing and guiding PDTs through the regulatory review process, as well as our scalable platforms, could provide a foundation on which Pear could potentially develop, host, and will pursue partnerships to maximize theconnect other PDTs from Pear as valuewell ofas our companythird parties.
Industry Trends Overview
Pear believes that major trends are converging to create a significant opportunity to treat disease with software, either alone or in combination with drugs, due to the following factors:
•The ongoing burden of chronic disease. Ninety percent of the $34.81 trillion in US annual healthcare expenditures are for people with chronic and mental health conditions.
•There is a pronounced shortage of clinicians. Across many disease areas there are tens of millions of patients with only a few thousand trained specialists who can provide treatment.
•There is a rapid patient and clinician adoption of digital solutions for healthcare delivery, such as telemedicine. The number of people who have usedusing telehealth has doubled from 40% prior toapproximately 39% before the COVID-19 pandemic to nearly 80% post-quarantine.
•There is pervasive use of technology. Americans spend an average of 5.4 hours per day on their mobile phones. Connection to a therapeutic delivered digitally is convenient and available for most Americans. The data captured by this engagement could also drive deeper insights into disease states and the ability to personalize treatment.
We believe the trends described above have created a moment in time when technology-based approaches are essential to, and capable of, revolutionizing healthcare. We believe that these trends have accelerated due to the COVID-19 pandemic, which resulted in fewer in-person visits and increased the adoption and use of technology and digital health experiences across consumers, clinicians, payors, and the broader healthcare system.
Our PDT Solution and Value Proposition
PDTs are prescribed by clinicians, downloaded by the patient to a smart phone, tablet, or VR headset, and used under the supervision of a prescribing clinician. PDTs have a recommended frequency and duration of use, but patients can use the product whenever they want and wherever they are. PDTs receive market authorization from regulators, like FDA, with a label that addresses both safety and effectiveness. To obtain a label from FDA, PDT companies developing novel therapies are generally required to submit data from one or more randomized, controlled trials (“RCT”) that demonstrate safety and effectiveness, and product candidates must be developed in a good manufacturing practice (“GMP”)-compliant environment.
In order for a patient to obtain a prescription, there is first an appropriate assessment and diagnosis of the patient, which is then followed by direction and ongoing guidance, as well as accountability related to product use and ongoing care managementpatient accountability related to product use
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 12
and ongoing care management by the clinician. Engagement levels for Pear’s PDTs significantly exceed that of published rates of health and wellness digital products. For patients with disease, a prescription is an enabler of access, engagement, and real-world effectiveness. PDTs integrate into existing healthcare infrastructure, allowing for a digital experience via a patient’s own clinician, enhancing the continuum of care rather than fragmenting care paradigms.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 11
We believe that PDTs will become an important segment of the digital health revolution and a vital part of the medical armamentarium because they have qualities that are similar to those of both health and wellness apps and pharmaceuticals. PDTs leverage the pervasiveness and accessibility of digital technology to improve human health. However, PDTs are always developed in a GMP-compliant environment, maintain compliance with data security and HIPAA protocols, and integrate into the standard of care. PDTs are also tested in RCTs and are FDA-authorized as safe and effective therapies for particular indications like pharmaceuticals, but they also enable clinicians to receive timely feedback regarding patient progress and therapy adherence. This patient monitoring capability afforded by PDTs may be the difference in a patient’s success during a course of treatment. The table below shows some of the key differences between PDTs and other products.


Pear reimagined what treating disease could look like from the vantage point of patients, clinicians, and payors. We set out to disrupt the healthcare system with urgency because we believe there is a growing need for the treatments that can be provided by PDTs. With our proprietary development and commercialization platform, PearConnect, we can bring product candidates to FDA for market authorization, and we can bring FDA-authorized products to patients, clinicians, and payors. We continue to refine and optimize our development capabilities, and we believe we will be able to scale our operations significantly in the near-term, allowing us to pursue authorizations for more product candidates, to self-commercialize our products inside the US, and commercialize our products outside of the US with the help of regional partners.
Pear is a platform-based, product-driven company. Our end-to-end PDT engine, which currently includes three FDA-authorized products and a pipeline of 14 product candidates, continues to produce clinical and real-world data as well as health economic outcome research (“HEOR”) data that provide evidence of Pear’s ability to enhance outcomes, increase access and reduce healthcare costs, and validate Pear’s value proposition. We believe our value proposition will magnify over time, as our model scales and we expand the reach and impact of PDTs.
We believe PDTs are poised to disrupt healthcare delivery in ways that could offer benefits to three major stakeholders:
1.Patients: PDTs provide 24/7 remote treatment access to patients that can improve outcomes and be used alongside other standard -of -care treatments. Additionally, PDTs have a relatively benign side effect profile compared to traditional pharmaceutical therapies.
2.Clinicians: PDTs provide additional treatment options and improve clinicians’ reach to patients, potentially resulting in broader patient impact. They are integrated into standard practice and may also be used alongside traditional drug-based treatments. Data from the PDT provides insight to the clinician and the clinical care team, which can facilitate enhanced care navigation and delivery. They also provide reimbursable events for interactions on the “Pear MDPearMD Clinician Dashboard,” an end-to-end patient service center. .
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 13
3.Payors: PDTs have the potential to reduce overall healthcare costs to payors by providing patients with therapies that may be more cost-effective. Additionally, PDTs may fill gaps in care across indications with high prevalence.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 12
For Patients
PDTs have the potential to be safe, effective, and accessible care options for patients with diseases and mental health disorders. These options are increasingly needed as diseases, and mental health disorders require more innovative approaches to overcome current treatment challenges and realize better patient outcomes. With Pear’s PDTs, patients have 24/7 remote access to their treatment program, and to clinicians who can monitorclinicians are able to observe progress 24/7 to inform their treatment approach, regardless of physical proximity. TheyPDTs can be integrated into standard of care and may be used alongside pharmacotherapy. Pear’s software is designed to be easy-to-use and intuitive, and it also generates data from patient usage to inform the treatment approach for each patient. Our clinical trial and real-world data also continue to demonstrate a favorable side-effect profile versus existing medications, which could foster stronger adherence to treatment programs.
The privacy provided by reSET and reSET-O, for example, offersis designed to offer access to treatment of addiction without fear of stigma. Patients also gain control over where they conduct addiction treatment and can conduct clinicianremote check-ins remotelyfor their clinician to view, which empowerempowers patients to better navigate their care. Further, we believe that our software enables a larger portion of Americans access to addiction treatment. Only a small fraction of patients with addiction are actively enrolled in treatment today,. and weWe believe that reSET and reSET-O may significantly lower treatment barriers to ongoing care and substantially increase the ability of clinicians to serve more and diverse patient needs. An analogous shift is seen in Somyrst, Pear’s treatment for chronic insomnia. Clinical guidelines indicate Cognitive Behavioral Therapy for insomnia (“CBTi”) as the first-line treatment, but there are currently fewer thanapproximately 300 certified and licensed CBTi clinicians in the US to treat a patient population of approximately 30 million. We believe that Somyrst as a therapeutic may substantially change the access paradigm, creating an ability for patients to get the treatment they need when they need it most.
For Clinicians
PDTs provide a new treatment option that clinicians can provide to their patients. Many clinicians today lack evidence-based treatment options with the potential to improve outcomes. PDTs are a standardized, evidence-based, cost-effective, and easily accessed class of treatments that can be used as a standalone treatment or in conjunction with pharmacotherapies. Not only can PDTs better enable clinicians to serve their patients, but PDTs can also improve the reach of clinicians, creating the potential for broader patient impact.
In addition, we believe Pear’s products enable richer and more effective patient data and engagement capabilities. For clinicians prescribing reSET, reSET-O, or Somryst, clinicians may use our Pear MDPearMD Clinician Dashboard and review a wealth of data-based, real-time feedback that can help them better understand and manage their patients’ treatment journey. The Pear MDPearMD Clinical Dashboard is easy to use and accessible 24/7, enabling care teams to conveniently manage populations of patients. This real-time engagement is particularly impactful in the world of addiction, in which adherence is continually decided, moment to moment, outside of the walls of the clinician’s office. The ability of a clinician to regularly monitor and engage with their patient can make the difference in successful navigation of the treatment journey.
Pear’s platformPearMD Clinician Dashboard interactions can also create reimbursable events for Pear MD Clinician Dashboard interactions.
For Payors
We believe Pear’s PDTs willhave the potential to reduce overall medical costs, support value-based care initiatives, and improve member experience. Insights and information collected by Pear’s PDTs can be integrated within care management workflows to provide payors with valuable insights that support population health management and value-based care initiatives. Pear’s PDTs can also fill gaps in specialty care across large populations, providing timely insights into patient engagement and practice performance via a dedicated platform.
Athe PearMD Clinician Dashboard.
Pear’s PearConnect platform has enabled rapid scaling of evidence generation for the benefit of provider organizations and payors to quantify product use, outcomes, and economic value. Over the past 12 months, Pear
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 14
has generated, presented and published numerous new datasets. Previously, Pear published a study of 351 patients evaluating healthcare resource utilization via insurance claims six months after reSET-O initiation found that compared to a six-month baseline there was a 62% reduction in inpatient hospitalizations and a 20% reduction in emergency room visits, resulting in a near-term cost-savings of $2,150 per patient. The durability of reSET-O’s treatment effect at nine (9) months was evaluated in a larger group of patients and was communicated in publications. One of those publication compared outcomes between reSET-O patients and
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 13
controls and found that reSET-O-treated patients experienced costs that were $2,708 lower than for controls, and which were also driven by a reduction in hospital-related utilization.
In addition, Pear published 12-month health economic data evaluating reSET-O compared to control in both national all-comer payor as well as Medicaid subpopulations. This analysis of over 1800 patients demonstrated lower health care costs for reSET-O treated patients compared to controls, as measured by actual health care claims, resulting in a health care cost reduction of an estimated $2,791 per patient in the national population, and a health care cost reduction of $3,832 per patient in the Medicaid population, driven predominantly by reduction in unique hospital encounters (emergency room, inpatient hospitalizations, and others). The study also observed an increase in medication possession ratio of buprenorphine in the reSET-O treated group compared to controls.
Pear also published real world health economic data for reSET. In 101 reSET treated patients in the real-world, claims analysis found an overall reduction in hospital encounters in the post-period compared to the pre-period, observing health care savings of an estimated $3,591 per patient in the 6–months post reSET initiation compared to the 6 months pre-reSET initiation. In addition, on May 19, 2021, Pear presented results of two analyses demonstrating the cost-effectiveness of reSET-O at the virtual annual meeting of The Professional Society for Health Economics and Outcomes Research. The machine learning analysis showed that all clusters of engagement (low, medium, and high) each experienced a very similar reduction in emergency department and inpatient stays of approximately 60%. The budget impact analysis showed that reSET-O plus treatment-as-usual (“TAU”) (i.e., transmucosal buprenorphine, face-to-face counseling and contingency management) versus TAU alone resulted in a projected net cost reduction over a five-year period. Supporting this budget impact analysis are two subsequent publications examining the cost effectiveness and cost utility of reSET-O, both showing quality adjusted life year gains from increased retention and abstinence, respectively, which were accompanied by reductions in overall costs, supporting the economic advantages of reSET-O vs. TAU.
Pear published its first health economic data evaluating Somryst. In a real-world claims analysis of patients with chronic insomnia exposed to the digital CBTi, researchers found overall reduction in hospital events in the 24 months post-treatment compared to the 24 months pre-treatment, with facility related cost-savings of an estimated $2,059 per patient in the post-treatment period. An analysis comparing treated patients compared to the control group over 24 months, presented at the EU The Professional Society for Health Economics and Outcomes Research (ISPOR) in Europe and the American Managed Care Pharmacy (AMCP) Nexus in fall 2022, observed a reduction of hospital facility services compared to the control group, resulting in health care utilization savings of an estimated $8,202 per patient.
We believe that the real-world claims analysis and health economic modeling data, together with existing RCT effectiveness data, demonstrate that reSET-O, reSET-O, and Somryst can provide compelling clinical and economic cost-savings to payors seeking a high level of care for their members.
Our Scalable Commercial Platform - PearConnect™
Pear’s commercial platform was designedOur goal is for PearConnect to bebecome an agile and scalable resource with the ability to hostoffer multiple PDTs developed by Pear and potentially by third parties. We believeHowever, to implement that the investments that we are makingvision, substantial capital will need to be expensed and substantial resources will need to be deployed. While we are not in a position today to pursue this vision, we believe if the requisite investments are made in our development engine, commercialization strategy, clinician and patient awareness, and HEOR data will enable our platform to, PearConnect could become the industry standard. We also believe that this will be an advantage not only to Pear, but also to our third-party developers that can potentially host their PDTs on Pear’s commercial platform. Through this replicable process, Pear expects to further boost its industry-leading position as the primary provider of PDTsan industry leading platform.
With this potential ability to host third-party PDTs on its platform, Pear is positioning itself not only as a leading PDT developer, but also as a centralized marketplace for patients seeking PDTs, which is a key value proposition and brand definer for Pear.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 15
Our Advantages
•Pear is pioneering the PDT industry. Pear is the first mover in the PDT industry, and as a result has advanced experience in developing and commercializing PDTs. Pear was the first company to receive FDA authorization for a PDT and the term PDT was conceived and defined during that initial authorization process. Pear currently has the greatest number of FDA-authorized PDTs and. Pear has been developing PDTs since 2013.
•Pear leverages our data aggregation capabilities. Pear’s ability to collect data from existing PDTs, generate insights and improvements based on these data, and then quickly develop new PDTs using these insights has enabled us to bring to market the first three FDA-authorized PDTs. We believe it will enable us to continue to bring additional FDA-authorized PDTs to market across a breadth of indications.
•Pear developed a reproducible process for cost-effective development and regulatory review of PDTs. We conduct agile software development for our medical devices in a GMP-compliant environment. Due to our remote clinical trial infrastructure, we are able to conduct clinical trials without the need for external sites and develop our products iteratively, which both accelerates timelines and reduces costs. This iterative product development paradigm contrasts with traditional drug development in which a molecule that enters the clinic cannot be improved or adapted along the way.
•Pear utilizes a relatively lean and efficient salesforce to target customers at the enterprise level. Our commercialization model is built on the intersection of biotech, tech, and medical device commercialization methods. We take an enterprise-level, key-account management approach to pursuing IDNs, health systems, academic teaching hospitals, and large addiction clinics and systems. This is a capital-efficient and scalable model that allows for accelerated growth. As we secure additional
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 14
reimbursement contracts with payors, we will seek to accelerate revenue growth through increased investment in sales and marketing. Pear currently promotes reSET and reSET-O using a small specialty salesforce and targeted marketing budget. Despite our lean salesforce and budget, our commercialization efforts have resulted in over 700 clinicians across 32 states who have prescribed reSET and reSET-O over 20,000 times since launch. Pear currently boasts patient and prescriber satisfaction scores of 89% and 82%, respectively, which validates our approach. The results achieved by reSET and reSET-O provide Pear with a strong foundation to achieve scalable and sustainable sales over time. Pear currently promotes Somryst via a direct-to-consumer pilot that launched in the fourth quarter of 2020.
•Pear is building a robust IP portfolio. Pear regularly applies for patents, copyrights, and trademarks throughout the development and iteration of its products, and also judiciously maintains trade secrets. Pear protects the iterations of its proprietary technology that are commercially important to its business by filing, maintaining, and, if necessary, defending patent rights, whether developed internally or licensed from third parties. We also rely on copyright, including registrations for product source code, graphic user interfaces and other content, as well as on trademarks. Finally, Pear maintains trade secrets relating to its proprietary PDT product development platform to enable Pear to strengthen its leadership position in the PDT industry over the long-term.
•Pear has a diverse and scalable management team that is pioneering the PDT space. The discovery, development, and commercialization of PDTs as a new healthcare delivery system requires both a breadth and depth of experience. Our cross-disciplinary and cross-functional team of experts collectively synthesizes years of experience in medicine, biotech, technology, and data science. This breadth and depth of experience has enabled Pear to forge new pathways within the healthcare industry.
Our Commercialization Strategy
With three FDA-authorized products on the market, and a deep and broad pipeline of product candidates,we believe Pear is currently at ahas made significant commercial inflection pointprogress. Additionally, the PDT category continues to expand with threeeight other companies recently receiving marketing authorization. There is strong momentum in the industry, and we believe our commercialization platform has the potential to set us up as the one-stop shop for PDTs.
We are pioneering PDT commercialization
By developing PDTs as a new class of medicines, Pear pioneers innovative approaches for its commercialization. Pear’s commercialization strategy attempts to combine the most effective features of the commercialization methodologies of both tech and life sciences companies to create a scalable yet capital-efficient PDT commercialization model.
Pear deploys two distinct commercialization models. First, to capture the most effective features of traditional life sciences commercialization methodologies, Pear utilizes a conventional sales-enabled approach focused on targeting large, multi-specialty health systems to capture the most effective features of traditional life sciences commercialization methodologies. Second, to capture the most effective features of traditional tech commercialization methodologies, Pear utilizes a virtual care platformthe PearMD Clinician Dashboard to directly engage with clinicians and patients.
Pear’s platformThe PearMD Clinician Dashboard enables the collection of data characterizing patient engagement and progress during treatment with PDTs. ThisWe believe this data collection capability appeals to clinicians and payors who seek to track treatment progress. Pear’s centralized PDT platformThe dashboard is configured to provide robust data that describes patient treatment progress to clinicians and payors, thereby providing these stakeholders with insights that have the potential to improve clinical outcomes and lower costs. Clinicians can use the insights provided by Pear’s platformdashboard to enable the clinician to support a patient’s course of treatment outside of routine office visits, and during periods when the patient may be experiencing increased challenges with treatment adherence. Similarly, payors can use the data provided by Pear’s platform and during periods when the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 16
patient may be experiencing increased challenges with treatment adherence. Similarly, payors can use the data provided by Pear’s dashboard to supplement their understanding of population-level treatment outcomes, and to adjust access to PDTs accordingly. As a result, Pear’s PDT platformthe PearMD Clinician Dashboard serves as an enterprise sales tool for stakeholders looking to monitor and improve the clinical outcomes of patients through data collection and analysis.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 15
We are building patient, clinician, and payor awareness.
As the pioneer in PDTs, Pear is built to educate all participants of care delivery, including patients, clinicians and payors, about a new class of medicines. To inform and educate the public about PDTs, Pear has built and deployed sales, marketing, patient and clinician services, and medical affairs teams. These PDT specialists are dedicated to educating patients, clinicians, payors, and other stakeholders about PDTs.
As a result of the efforts of our PDT specialists, PDTs are increasingly recognized as a treatment option by the medical community, as evidenced by the increasing number of clinicians prescribing PDTs and the increasing number of PDT prescriptions being written. To date, Pear has educated more than 690hundreds of clinicians that have collectively written prescriptions for one or more of Pear’s three FDA-authorized products, and we expectbelieve that number will grow significantly over time. Once introduced to PDTs, those clinicians are equipped to provide valuable education and guidance to patients and other clinicians regarding how the use of PDTs.
We are creating access to PDTs by establishing reimbursement pathways.
The Pear team is actively working to establish reimbursement pathways for PDTs, and we believe that as a result of these efforts, payors are increasingly providing their members with access to PDTs. To date, Pear has 30had 39 payors across more than 31.7 million covered lives that provide access to its PDTs. reSET and reSET-O are currently covered as pharmacy benefits, medical benefits, and/or via direct purchase transactions. In an effort toTo onboard additional payors, Pear engages directly with payors to establish coding, coverage, and payment pathways for its PDTs and the PDT category as a whole. Pear is also exploring innovative contracting models with payors that utilize the strong correlation between patient engagement with PDTs and the resulting clinical and economic benefits. We believe that these continued payor engagement efforts will result in reimbursement for PDTs by both commercial and public payors in the US, including, but not limited to, self-insured employers, PBMs, health plans, IDNs, and public payors, including Medicaid and Medicare.
In addition to pursuing traditional payor reimbursement of PDTs, Pear also offers a PEAR Assistance Program (“PAP”) to ensure that eligible, underserved patients living with SUD or OUD have access to reSET and reSET-OPear’s PDTs. PAP is designed for SUD or OUD patients who are being treated by a licensed US clinician on an outpatient basis and have been prescribed a PDT. PAP-qualifying patients have limited or no health insurance coverage, a demonstrated qualifying financial need, and live within the US. Patients enrolled in PAP generate additional real -world and HEOR data that may be used to support the utilization and coverage of Pear’s products.
We are building end-to-end infrastructure that provides ease of use to clinicians .
To enable a high degree of accessibility to PDTs for clinicians, Pear has developed, designed, and deployed a patient service platform, called PearConnect™, designed to be a convenient one-stop shop for PDTs. PearConnect features four distinct system components, consisting of PDTs for patient use, the Pear MDPearMD Clinician Dashboard, an end-to-end patient service center, and a data analytics system configured to aggregate patient engagement, adherence, and clinical outcome data for insight generation. These components of PearConnect are intentionally designed to improve accessibility to Pear’s PDTs. PearConnect also incorporates various data privacy and security features to support the protection, quality, and integrity of each component of our platformPearConnect, including the data that is processed and stored by the platformPearConnect. Each component plays a meaningful role in ongoing and product evolution and performance, building Pear’s PDT marketplace, and enabling clinicians and payors to assess the value of each PDT on a population and individual level.
The Pear MD Clinician Dashboard displays icons representing multiple different PDTs to a viewing clinician. Via the Pear MD Clinician Dashboard, clinicians are able to learn about the PDTs offered and are able to gain valuable, timely insights into their patients’ progress over the course of a PDT prescription through the Pear MD Clinician Dashboard based on engagement, adherence, and clinical outcome data gathered and analyzed by the analytics component of the platform. If clinicians and payors deem initial PDT prescriptions a success as supported by this collected and displayed health economic data, we can easily add any new PDT products to existing agreements with clinicians and to the Pear MD Clinician Dashboard.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 16
Pear’s end-to-end patient service center provides support to patients that have been prescribed PDTs, which includes informational and technical support regarding the use of the PDT and the virtual care platform. The service makes it easier for clinicians to introduce new patients to PDTs and to Pear’s platform. PearMD Clinician Dashboard. The service makes it easier for clinicians to introduce new patients to PDTs and to Pear’s platform.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 17
The data analytics component of PearConnect collects patient engagement, adherence, and clinical outcome data, and generates insights based on this data. The insights generated by the data analytics component of PearConnect can be accessed by clinicians via the Pear MDPearMD Clinician Dashboard to inform clinicians of their patients’ progress over the course of a PDT prescription. The insights generated by the data analytics can also be used by us to incrementally evolve individual PDTs to better serve patients, and we believe we will ultimately generate improved clinical outcomes.
We are able to demonstrate the value of our PDTs with HEOR data generated based on commercial patients.
Pear’s commercial infrastructure allows for the analysis of patients’ healthcare claims data pre-, during, and post-use of Pear’s PDTs. Based on these robust analytics, Pear is able to gain insight into PDT utilization across patient groups, and to understand how use of its PDTs correlates with clinical outcomes. Data-derived insights support collaboration with clinicians, coverage and reimbursement by payors, and product enhancements.
Discovery, Strategy, and Development Platform- PearCreate™
Developing an entirelya new class of medicines led to Pear pioneering new approaches for the discovery and development of PDTs. We have invested in and created a model to progress from concept to market authorization, and we believe we have optimized that model to the point where it is now ready to scale. subject to our ability to invest in our pipeline. In July 2022, we announced that we paused substantially all investment in our pipeline.
Pear is pioneering clinical development. OurOur PearCreate platform consists of three approaches consist of: (i) an integrated virtual and decentralized clinical study platform, (ii) agile development as therapeutic candidates move through phases, and (iii) integrated health economic and real-world extension studies to facilitate regular and timely evidence generation.
First, Pear has designed and built its own integrated virtual and decentralized study platform. This technology facilitates a seamless experience for fully virtual and decentralized studies. DREAM, Pear’s remote, real-world clinical study of Somryst is the first to utilize this platformPearCreate, with recruitment, enrollment, consentsconsent, monitoring, and assessments all completed through digital and virtual means. Patients do not need to be present at a clinic or site for any step of the process. In addition to being more cost-effective, the speed of recruitment and enrollment is enhanced by facilitating fully automated screening and consenting, and by minimizing the number of patients requiring manual or direct human interaction.
Second, as Pear’s therapeutic candidates move from discovery and through subsequent stages of development, Pear iterates on the product. If data indicate that adding additional modalities or mechanisms of action to the candidate is warranted, Pear is able to refine the PDT in between subsequent studies and re-evaluate the candidate in a variety of clinical study designs. We believe deploying this agile and adaptable framework to clinical development has the potential to increase not only speed but also the probability of success.
Third, as Pear conducts pre-market studies on PearCreate, health economic endpoints are integrated and real-world extension studies are built into development plans. Registrational and even earlier stage development studies having health economic or insurance claims endpoints have the potential to facilitate earlier insights to support payor engagement. Post-randomized studies and even post-market, Pear continues to generate clinical and economic data on every patient that uses a PDT, whether it is a Pear PDT or a third-party PDT using the Pear platformPearConnect. Adaptive real-world extension studies post-registration demonstrate generalizability from RCTs, and have the ability to demonstrate that RCT outcomes are consistent in real-world use and across various clinical settings and patient populations.
Our Commercial Products
reSET
reSET is the first PDT to receive FDA market authorization and is authorized in the US and Singapore for the treatment of SUD in patients who are 18 years of age and older. reSET is the only FDA-authorized product
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 17
indicated to treat addiction to alcohol, cannabis, cocaine, and stimulants (such as methamphetamines) as no pharmacotherapy or other FDA-approved treatments for this patient population exist. reSET is a 12-week
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 18
prescription-only treatment for patients who are actively enrolled in outpatient therapy under the supervision of a clinician.
Disease Overview
Addiction is a disease of the brain that manifests as compulsive substance use, known as SUD. In the US, the disease of addiction can be characterized as an epidemic, with more than 2046 million Americans struggling with addiction based on 20192021 data.
Each year, there are approximately 13,000 overdose deaths related to stimulant use, particularly methamphetamine, and approximately 16,000 deaths related to cocaine use. Since the 1970s, there has been a significant increase in mortality related to unintentional drug poisonings, growing at 7% per year. Unfortunately, the COVID-19 pandemic has only accelerated these trends. In 2020, there was an 18% increase in drug -related overdose deaths and a 26% increase in overdoses involving cocaine.
Traditional medical guidelines recommend Cognitive Behavioral Therapy (“CBT”) and contingency management (“CM”) as best practices in treating SUD. However, there are multiple roadblocks that prevent a majority of SUD patients from ultimately receiving effective behavioral treatment, such as CBT and CM. CBT is largely conducted in outpatient settings, and is a time-intensive, location-dependent treatment methodology. This and the sheer quantity of SUD patients results in an insufficient number of trained therapists and facilities to meet demand. This results in SUD patients often receiving inconsistent treatment in inconvenient locations. Insufficient access to CBT can cause patients to drop out of therapy or not pursue therapy in the first place. Even when SUD patients are able to gain sufficient access to CBT, clinician education, training, and treatment paradigms vary. Documented discrepancies in CBT administration standards and protocols impede the ability of CBT to adequately and uniformly treat the wide range and quantity of SUD patients that require care. Furthermore, many patients may experience perceived or real stigma associated with physical attendance at CBT sessions. As a result of these many obstacles, only a fraction of SUD patients receive CBT treatment. For instance, in 2019, only 1.5% of SUD patients received formal treatment.
In addition to the challenges toof initiating and completing a first round of SUD treatment, for many patients, addiction can be a lifelong battle that often involves multiple relapses for many patients. This constant and recurring need for treatment places additional burdens on SUD patients, particularly given the currently insufficient treatment options available. There remains a significant unmet need for accessible and effective SUD treatment options.
Our Solution
To bridge this gap between disease prevalence and treatment availability, Pear has an FDA-authorized PDT that has been clinically proven to help treat addiction and extend the reach of clinicians to patients during their recovery journey. reSET is authorized for the treatment of SUD in patients 18 years or older. reSET’s mechanism of action combines addiction-specific CBT, fluency training, contingency management, and craving and trigger assessment, all provided by Pear’s software-based application. It is intended to reduce substance abuse during treatment, eventually supporting discontinuance and recovery while simultaneously improving retention rates in the outpatient treatment program.
To begin treatment, the clinician prescribes the patient the PDT. The patient then downloads the software to a smart phone, tablet, or VR headset, inputs their prescription access code, and gets access to the PDT. The PDT includes three evidence-based treatments, each of which represent a distinct mechanism of action:
•The first is an addiction-specific form of cognitive behavioral therapy, called a community reinforcement approach (“CRA”), that moves patients from actively using a substance to reducing and ultimately discontinuing use. CRA is a form of treatment that integrates cognition and learning with treatment techniques. CRA is based on the insight that cognitive, emotional, and behavioral variables are functionally interrelated. Treatment is aimed at identifying and modifying the patient’s maladaptive thought processes and problematic behaviors through cognitive restructuring and behavioral techniques
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 18
to achieve change. Disease-specific CRA targets the associated neurocircuitry for addiction, harnessing adaptive neuroplasticity. reSET is comprised of 61 interactive modules (31 core and 30 supplement modules). Core modules focus on key CRA concepts to build skills to support behavior change and prevent relapse. Supplemental modules provide more in-depth information on specific topics
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 19
relapse. Supplemental modules provide more in-depth information on specific topics, such as relationship skills and living with a disease.
•The second is fluency training, which assesses proficiency and reinforces concept mastery. Fluency training supports the ability of the patient to apply their learnings in moments of stress to facilitate their treatment and ultimate recovery.
•The third is contingency management, which is a positive reinforcement mechanism. CM induces a dopamine response directly competing with the negative response induced by substance use. It does this by rewarding healthy behaviors and key outcomes such as engagement in treatment and reduced substance use through a clinical algorithm.
CRA and fluency training strengthen affected neurocircuitry while CM induces dopamine in the nucleus accumbens repairing dysfunctional neurophysiology, driving engagement with CRA and treatment in a virtuous cycle. These integrated mechanisms of action drive patient engagement and target the pathophysiology of addiction to correct and restore neurofunction.
reSET also includes validated assessments, including substance use, triggers, and cravings supporting interactive engagement and insights to the clinical care team. All of this information is shared with the clinical care team via the Pear MDPearMD Clinician Dashboard, enabling the clinician and care team to derive insights to inform optimal patient care.
By integrating PDTs into the practice of medicine, the clinician and patient relationship may be strengthened, and data insights facilitate clinician extension for greater efficiency and effectiveness. reSET and reSET-O provide 24/7 anytime, anywhere treatment. As the patient undergoes therapy with reSET, the patient and clinician can continue to meet in person or virtually as part of their outgoing treatment therapy, and the clinician has easy access via the
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 19
Pear MDPearMD Clinician Dashboard to monitor and adjust treatment. We believe the connectivity established through our PDTs is valuable to patients and clinicians.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 20
Our Clinical Trial Data
Among patients with SUD (i.e., addiction to cocaine, cannabis, stimulants, and/or alcohol), RCT data demonstrated that adding the PDT to TAU more than doubled abstinence rates during the last month of the 12-week trial. Among all patients, adding a PDT to outpatient care improved the retention rate compared to TAU (72.2% vs 63.5%) at the end of the 12-week trial. Treatment with the PDT did not demonstrate a significant difference in unanticipated adverse events compared to TAU.
Adult men and women (N=399) entering 10 outpatient addiction treatment programs were randomly assigned to receive 12 weeks of either TAU (N=193) or reduced treatment-as-usual (“rTAU”) plus the PDT, with the intervention substituting for about two hours of standard care per week (N=206).
TAU consisted of individual and group counseling at the participating programs for 4-6 hours per week. rTAU consisted of 2 hours less than TAU of aggregate individual and group counseling. The primary outcome measures were abstinence from drugs and heavy drinking (measured by twice-weekly urine drug screens and self-report) and time to dropout from treatment.
Those in the PDT+rTAU group had a higher retention rate of 76.2% compared to 63.2% in TAU and a greater abstinence rate in the last 4 weeks of 40.3% in the PDT+rTAU group relative to 17.6% in the TAU group. This effect was more pronounced among patients who had a positive urine drug or breath alcohol screen at study entry (N=192) with an abstinence rate in the last four (4) weeks in the PDT+rTAU group of 16.1% compared to 3.2% of TAU.
reSET Pivotal Clinical Trial. Abstinence in TAU relative to rTAU + PDT.
reSET Pivotal Clinical Trial. Abstinence and Retention in TAU relative to rTAU + PDT.
| | | | | | | | | | | | | | | | | | | | |
| Outcomes | | TAU | | rTAU+reSET | | p-value |
Abstinence: All patients | | 17.6 | % | | 40.3 | % | | 0.4000 |
Abstinence: Non-abstinent at study start | | 3.2 | % | | 16.1 | % | | 1.3000 |
Retention in treatment: All patients | | 63.2 | % | | 76.2 | % | | 4.2000 |
Overall safety was in-line with expectations and no significant difference was observed in the rate of adverse events between groups (p = 0.3563).
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 2021
Evaluation of rTAU+PDT abstinence and retention compared to TAU.
| | | | | | | | | | | | | | | | | | | | |
| Outcomes | | TAU | | rTAU+PDT | | p-value |
Number of Adverse Events | | 29,000 | | 37,000 | | n/a |
% of Adverse Events | | 11.5 | % | | 14.5 | % | | 0.3563 |
Adverse events evaluated were typical of patients with SUD, including cardiovascular disease, GI events, depression, mania, suicidal behavior, suicidal thoughts and attempts. None of the adverse events in the interventional arm were adjudicated by the study investigators to be related to the PDT.
This RCT was reviewed by FDA and supported Pear’s FDA market authorization of reSET to treat patients with SUD to improve clinical outcomes.
Real World Evidence and Health Economic and Outcomes Research Data
Pear intends to publishpublished reSET Real World Evidence (“RWERWE”) and HEOR data starting with a six-month duration. In 101 reSET treated patients in the real-world, claims analysis found an overall reduction in hospital encounters in the post-treatment compared to the pre-treatment, observing health care savings of an estimated $3,591 per patient in the 6 months post reSET initiation compared to the 6 months pre-reSET initiation.
reSET-O
reSET-O is the second PDT to receive FDA authorization and the first to receive breakthrough designation in the US for the treatment of OUD in patients who are 18 years of age and older. reSET-O is a 12-week prescription-only treatment that is intended to increase retention of patients with OUD in outpatient treatment by providing CBT as an adjunct to outpatient treatment that includes transmucosal buprenorphine and CM. reSET-O is indicated only for patients who are currently under the supervision of a clinician.
Disease Overview
Opioid use disorder in the US has increased so dramatically and taken such a heavy toll on American lives over the past decade that, in 2017, the US Department of Human Health Services declared the opioid epidemic a public health emergency. Approximately 1.6 million Americans suffer from OUD annually in the US, and according to the CDC, the substance use and opioid use disorder epidemics continue to grow with unprecedented numbers of fatal opioid overdoses, in excess of 200 deaths daily (over 75,000 annually), approximately 87% of which are due to widely available, inexpensive yet pharmacologically-potent synthetic opioids. Nonfatal overdoses far outnumber fatal overdoses and place a tremendous burden the health care system through excess utilization of acute care services such as emergency department and inpatient stays, which contributes to rising health care costs. For example, every year, there are over 500,000 emergency room visits for opioid overdoses in the US. Of those patients that are discharged from the emergency room following an overdose, 24% are readmitted to the emergency room for additional emergency care within 30 days. As a result of these frequent emergency room visits, opioid overdoses are estimated to cost the US $11.0 billion in hospital costs alone. A recent study evaluated the direct healthcare cost of 2,044,467 people with OUD to be $89.1 billion, with an average cost of $43,581 per patient per year. The cost of the current opioid crisis in the US is estimated to exceed $700.0 billion.
Treatment has been shown to help patients avoid the increased risk of respiratory arrest, and organ system damage, and dramatically reduce the use of acute care services and associated high costs. However, only 11.2% of patients in the US receive treatment with MOUDMedications for Opioid Use Disorder (“MOUD”) in a given year. For those who obtain treatment, dropout rates are high, ranging between 30% and 50% at 90 days, highlighting the need for comprehensive support to keep patients engaged in their recovery. Progress in recovery helps reduce the burden on the health care system, and helps to mitigate the harm the opioid crisis causes.
The current standard of care for treating opioid addiction is medication-assisted treatment (“MAT”). MAT consists of the administration of medication, such as buprenorphine, in combination with counseling and behavioral therapy. Only a fraction of opioid-addicted patients, however, receive treatment. Similar to SUD patients, there are
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 22
significant barriers to opioid-addicted patients receiving accessible and effective care. Many patients fail to receive care as a result of care inaccessibility, cost, or patient stigmatization. It is estimated that only 19% of opioid-addicted patients receive treatment each year. In addition, many opioid-addicted patients elect to rely solely on
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 21
medication and reject counseling. For example, only 10% of opioid-addicted patients that are prescribed buprenorphine receive adjunct psychotherapy treatment. While reliance solely on the medication portion of the recommended treatment paradigm may provide a higher level of convenience for patients, adherence to only a portion of treatment decreases the overall probability of success. Therefore, due to the current lack of effective treatment options for OUD, and the continually growing number of patients, there remains a significant unmet need in the fight against OUD.
Our Solution
To bridge this gap between disease prevalence and treatment, Pear has an FDA-authorized PDT that has been clinically proven to treat OUD and extend the reach of clinicians to patients during their recovery journey. reSET-O is authorized for use in combination with buprenorphine in the US for the treatment of OUD in patients who are 18 years or older.
Similar to reSET, reSET-O is intended to increase the retention of patients with OUD in outpatient treatment by providing the same behavioral treatments. This PDT is a 12-week treatment used as an adjunct to outpatient treatment, including transmucosal buprenorphine and CM. Patients using reSET-O are required to be under the supervision of a clinician.
reSET-O has three mechanisms of action consisting of addiction-specific CRA, fluency training, and CM. reSET-O has an additional function that supports the patient’s appropriate use of buprenorphine. This function includes reminders, assessments of use, non-use, and reasons and beliefs. reSET-O also has additional content modules specific to OUD.
Similar to reSET, patients download reSET-O to their smart phone, tablet, or VR headset for discreet, convenient access to therapy, interactive treatment, learning, and support. reSET-O lets patients practice what their clinicians taught them anytime, anywhere, and report daily triggers, cravings, and substance use, so they feel more engaged and supported between appointments.
reSET-O also encourages patients with tangible rewards via its CM capabilities. These rewards can be managed by clinicians.
As the patient undergoes therapy with reSET-O, the patient and clinician continue to meet in person or virtually as part of their outgoing treatment therapy, and the clinician has real-time access via the Pear MDPearMD Clinician Dashboard to monitor and adjust treatment.
Our Clinical Trial Data
There have been multiple RCTs evaluating PDTs in patients with OUD in combination with MAT and Medications for Opioid Use Disorder (“MOUD”)MOUD. These studies have been conducted across a variety of durations and clinical care models. The primary RCT that the FDA reviewed to support the marketing authorization of reSET-O is described below.
In summary, the clinical data demonstrated that adding reSET-O alongside outpatient treatment and buprenorphine use increased the retention of patients with OUD by 14% at the end of the 12-week trial and reduced opioid use. Patients achieving no substance use by the end of treatment was 75.9% in TAU + PDT group versus 60.6% in TAU-alone (p = 0.03). Patients in the TAU + PDT group had an 82.4% retention rate compared to 68.4% in the TAU group (p = 0.02). The observed adverse events were of a type and frequency as anticipated in a large population of patients with OUD or associated with buprenorphine pharmacotherapy, particularly during the induction phase. The adverse events observed were not adjudicated to be device-related.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 23
A block-randomized, parallel, 12-week treatment trial was conducted with 170 opioid-dependent adult patients. Participants received an internet-based community reinforcement approach intervention plus CM (“CRA+”) and buprenorphine, or CM-alone plus buprenorphine.
•All patients received 30 minutes of face-to-face clinician interaction every other week.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 22
•Patients provided urine samples three times per week to objectively monitor substance use.
•Primary endpoints were:
▪retention in treatment, and
▪no substance use (during last four weeks).
Incorporation of a PDT increased retention and abstinence
| | | | | | | | | | | | | | | | | |
| | TAU + PDT | | TAU | | p-value |
Retention | 82.4 | % | | 68.4 | % | | 20.00 |
No Substance Use (last 4 weeks) | 77.3 | % | | 62.1 | % | | 30.00 |
Among patients whose primary addiction was opioids, adding reSET-O to TAU had significantly greater odds of opioid abstinence during weeks 9-12 of the 12-week trial with abstinence rates (no opioids) at 77.3% for the TAU + reSET-O treatment group versus 62.1% for the TAU group (p = 0.02). Further, adding reSET-O to outpatient therapy, buprenorphine increased retention of patients with OUD by 14% at the end of the 12-week trial, with retention found to be 82.4% in the TAU + reSET-O treatment group versus 68.4% in the TAU group (p = 0.02).
Overall safety was in line with expectations and no significant difference was observed in the rate of adverse events between groups (p = 0.42). The observed adverse events were of a type and frequency as anticipated in a large population of patients with OUD or associated with buprenorphine pharmacotherapy, particularly during the induction phase. The adverse events observed were not adjudicated to be device related.
Real World Evidence
Pear continues to generate RWE evaluating the use of reSET-O across prospective and commercial use. We plan to continue to publish and disseminate reSET-O real-world data.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 24
For example, a published real-world analysis evaluated patient engagement, usage of reSET-O, and associated outcomes of opioid use and treatment retention in those using buprenorphine MAT/MOUD. The results demonstrated that reSET-O is readily and broadly used by patients with OUD and that real-world therapeutic engagement is positively associated with abstinence and retention in treatment.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 23
The real-world observational analysis was conducted in a population of 3,144 patients with OUD who were prescribed their first 12-week prescription for reSET-O. The patients came from 30 different states and represented a wide range of demographics (15.4% of individuals were between ages 19-29, 45.5% were between 30-39, 25% were between 40-49, 11.2% were between 50-59, and 2.9% were age 60 or older).
A summary of the results is set forth below:
•Patients in the 40-49 age range had highest level of “Active” days of treatment.
•80% of patients completed at least 25% of core modules, 66% completed half of all core modules, and 49% completed all core modules across the entire cohort of patients.
•Patients exhibited usage of reSET-O across a 24-hour period, including before and after clinic hours, at times when clinicians would not traditionally be available.
•Over 70% of patients were retained in reSET-O treatment and continued to use their PDT during the last 4 weeks.
•91% of the patients were “responders”, meaning 80% of their self-reports and urine drug screens (“UDS”) were negative for illicit opioid use.
•Patient engagement on reSET-O was higher than other products as shown in the chart below.
Patient Engagement on reSET-O Compared to Other Products
(using the above-reference study for reSET-O)
Patients have also benefited from repeat prescriptions for reSET-O. Another peer-reviewed study evaluated the impact of longer-term (24 week) engagement and use of reSET-O in a sample of 3,817 patients) and found increased abstinence (86%) and retention in treatment (91%), and 94.4% met the responder rate of being abstinent for at least 80% of the weeks of treatment.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 25
Healthcare Economic & Outcomes Research
Pear continues to generate real-world evidence evaluating the use of reSET-O.
reSET-O’s impact on healthcare resource utilization has been demonstrated in two separate analyses. In the first study there was a per-patient reduction of $2,150 in the six months after reSET-O initiation vs. a six-month
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 24
baseline, and there was a $2,708 difference vs, there was a per-patient reduction of $2,150 in the six months after reSET-O initiation vs. a six-month baseline, and there was a $2,708 difference versus controls at nine months, driven largely in both instances by reductions in emergency department visits and inpatient stay-associated costs.
A follow-on analysis of this patient population examined the cost implications for payers,payors and found real-world cost reductions of $2,385 six months after product initiation (vs. the six-month baseline period) for a typical US payer after factoring in rebates. This analysis also showed that the number-needed-to-treat, i.e., the number of patients you need to treat to prevent one additional hospital-related event, was just 4.8 treated patients.
In addition, an analysis of the impact of a second prescription of reSET-O using a linear regression model found a further 27% reduction in hospital-related events with a second prescription for reSET-O (24 weeks of treatment), compared to the first (12 weeks of treatment).
In addition to clinical value, these results support the cost-savings benefit of reSET-O as an adjunct to TAU.
Somryst In 2022 Pear published 12-month health economic data, evaluating reSET-O compared to a control group in both national all-comer payor as well as Medicaid subpopulations. This analysis of over 1800 patients demonstrated lower health care costs for reSET-O treated patients compared to controls, as measured by actual health care claims, resulting in a health care cost reduction of an estimated $2,791 per patient in the national population, and a health care cost reduction of an estimated $3,832 per patient in the Medicaid population, driven predominantly by reduction in unique hospital encounters (emergency room, inpatient hospitalizations, and others).
Somryst
The Company has deprioritized commercialization efforts regarding Somryst while focusing available resources on the commercialization of reSET and reSET-O and wrote off the acquired technology previously capitalized in intangible assets used in our Somryst product offering and therefore recorded impairment expense of $0.8 million, which is included in cost of revenue in the Company's consolidated statement of operations, during the year ended December 31, 2022.
Somryst is the only non-drug FDA-authorized and guideline-recommended treatment for chronic insomnia in adults over 22 years of age. It is intended to improve patients’ symptoms of chronic insomnia. Somryst is the first PDT submitted through FDA’s traditional 510(k) pathway while simultaneously reviewed as part of the FDA’s Software Precertification Pilot Program to help build and test FDA’s Digital Health Precertification Working Model 1.0. Somryst is a nine-week prescription-only treatment. Somryst has been the subject of 29 completed clinical studies and ongoing clinical studies.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 26
Disease Overview
Insomnia is a sleep disorder that manifests as difficulty falling or remaining asleep. Insomnia can be a short-term (e.g., acute) or a long-term (e.g., chronic) condition. Chronic insomnia is classified as persistent insomnia that occurs at least three nights a week for a period of at least three months. Chronic insomnia is a relatively common medical condition in the US, and is estimated to affect approximately 30 million Americans.
Chronic insomnia adversely affects many facets of a patient’s everyday quality of life, including social, occupational, and educational. These reductions in a patient’s daily quality of life can also give rise to more serious, and life-threatening medical conditions, including depression, suicidality, hypertension, and heart attacks. For example, in patients with chronic insomnia approximately 40% have depression, and up to 35% have anxiety. Chronic insomnia also results in a plethora of more measurable detrimental consequences, including heavy costs and resource utilization strain on the healthcare system. For example, annual medical costs for patients diagnosed with insomnia are approximately 25% higher relative to those without insomnia.
Current options for the treatment of chronic insomnia are limited, and the available treatment options have thus far proven ineffective. The American Academy of Sleep Medicine and the American College of Physicians clinical guidelines recommend CBTi as the first-line treatment for patients suffering from chronic insomnia. However, CBTi is difficult to access, as fewer thanapproximately 300 certified and licensed CBTi clinicians currently exist in the US to treat a patient population of approximately 30 million.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 25
Despite the clinical guidance recommending CBTi as the first-line treatment for chronic insomnia, over 50% of chronic insomnia patients do not seek CBTi treatment, but instead rely on the use of sleep medications (e.g., “sleeping pills”), such as Ambien (zolpidem). Because most available sleep medications are only recommended for short-term use, they are not recommended as a long-term solution to chronic insomnia. Long-term use of sleep medications can yield unwanted side-effects and result in serious, adverse health consequences. Further, utilization of sleep medications can result in reliance on the medication for sleep induction, rather than addressing the underlying condition, thereby perpetuating the chronic nature of the disease.
Our Solution
We believe Somryst addresses the limitations of current standard of care by treating the underlying factors without a label risk of dependence, inappropriate use, and adverse effects of pharmacotherapy. Somryst effectively delivers all three primary mechanisms of actions provided by face-to-face CBTi (sleep restriction and consolidation, cognitive restructuring, and stimulus control), using Pear’s software-based PDT. By delivering CBTi digitally, barriers to people with chronic insomnia are reduced while receiving guideline-recommended treatment. The following is a brief description of each mechanism of action.
•Sleep restriction and consolidation reducesreduce the time it takes to fall-to-sleep and reducesreduce the amount of wakefulness in the middle of the night by consolidating the patient’s sleep through an algorithm to direct the patient’s sleep-wake pattern.
•Cognitive restructuring teaches patients to identify maladaptive thought patterns related to sleep circuits and replace and strengthen healthy neurocircuits.
•Stimulus control teaches patients to identify behaviors, thoughts, and practices that increase arousal and limit sleep and then to correct those behaviors.
Patients are prescribed Somryst by their clinician. After downloading the PDT to a smart phone, tablet, or VR headset, patients engage in fully digital, automated, and interactive exercises that move patients through the mechanisms of action to get better sleep. These interactive modules span a range of topics, including properly tracking sleep patterns and identifying stimulants that may be harming sleep quality and restructuring maladaptive thoughts about sleep. Patients use Somryst for approximately 45-60 minutes weekly, over a period of 6-9 weeks. The content and interface encouragesencourage engagement, facilitatesfacilitate active learning, and promotes behavioral change, while the algorithm supports a tailored experience. promote behavioral change, while the algorithm supports a tailored experience.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 27
Our Clinical Trial Data
Somryst provides a clinician-facing dashboard via the PearMD Clinician Dashboard that allows healthcare providers to track patient treatment and progress. The PearMD Clinician Dashboard displays information about patients’ use of Somryst, including the Insomnia Severity Index (“ISI”), the Patient Health Questionnaire 8 scores, and sleep metrics derived from nightly sleep diaries (“Sleep-Onset-Latency” or “SOL”) and Wakefulness-After-Sleep-Onset (“WASO”). The evidence base for Somryst is extensive and includes the following:
•Somryst has been examined in an aggregate of 29 completed and ongoing studies. FDA submission and authorization was supported by two RCTs with an aggregate of more than 1,400 adults with chronic insomnia. The data from the RCTs showed a 45% decrease in the severity of insomnia symptoms, a 50% decrease in depression symptoms, and nearly a 45% decrease in anxiety symptom. It has also demonstrated a durable effect on insomnia, depression, and anxiety for up to 18 months.
•In a clinical trial of 303 patients with chronic insomnia (Study 1), those on treatment demonstrated clinically meaningful improvements in insomnia severity, SOL (time to fall asleep), and WASO (time awake at night) at the end of treatment, as well as at six and 12 months follow-up compared to active control. Results of the study were published in JAMA Psychiatry.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 26
•In a separate study of 1,149 adult patients with chronic insomnia and depressive symptoms (Study 2), those on treatment for nine weeks saw a significant reduction in insomnia severity measurements compared to controls.
The majority of patients randomized to the Somryst arm no longer met clinical criteria for insomnia at the end of the nine-week treatment. Patients were examined at baseline, nine weeks, six months, 12 months, and 18 months following treatment. At treatment end, the patients who received Somryst experienced a decrease in their mean ISI score to 7.3 from baseline of mean 15.9, while the control arm had a reduction to 13.2 from 16.2 baseline. In addition, the Somryst group had a reduction in depression severity to a mean score of 3.8 from an 8.0 baseline mean. The mean score decreased to 6.2 from a mean of 7.8 at baseline in control subjects. It was observed that the clinical improvements were maintained to 18 months post-treatment. Study data, published in Lancet Psychiatry, showed decreases in anxiety symptoms and suicidal ideation.
The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (“DSM-5”) makes no distinction between primary and comorbid insomnia. This previous distinction had been of questionable relevance in clinical practice, and a diagnosis of insomnia is made if an individual meets the diagnostic criteria, despite any coexisting conditions. The International Classification of Sleep Disorders, Third Edition criteria are consistent with the changes to the DSM-5.
The DSM-5 defines insomnia as dissatisfaction with sleep quantity or quality, associated with one (or more) of the following symptoms:
•Difficulty initiating sleep;
•Difficulty maintaining sleep, characterized by frequent awakenings or problems returning to sleep after awakenings; and
•Early-morning awakening with inability to return to sleep.
Other criteria include the following:
•The sleep disturbance causes clinically significant distress or impairments in social, occupational, educational, academic, behavioral, or other important areas of functioning;
•The sleep difficulty occurs at least three nights per week;
•The sleep difficulty is present for at least three months;
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 28
•The sleep difficulty occurs despite adequate opportunity for sleep;
•The insomnia cannot be explained by and does not occur exclusively during the course of another sleep-wake disorder;
•The insomnia is not attributable to the physiological effects of a drug of abuse or medication; and
•Coexisting mental disorders and medical conditions do not adequately explain the predominant complaint of insomnia
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 27
Somryst use led to a meaningful reduction in ISI as well as reduction SOL and WASO, as shown by the UVA and GoodNight Studies.
UVA Study: ISI Score Results
Proportion of ISI responders (reduction in ISI score > 7 from baseline)

| | | | | | | | | | | | | | | | | | | | |
| Time of Assessment | | Somryst Group | | Control Group | | p-value |
End of Treatment Period (Week 9) | | 52.6 | % | | 16.9 | % | | <0.0001 |
Month 6 Follow-Up | | 59.6 | % | | 35.7 | % | | 0.0002 |
Month 12 Follow-Up | | 69.7 | % | | 43.0 | % | | <0.0001 |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 2829
GoodNight Study: ISI Score Results
Proportion of ISI responders (reduction in ISI score > 7 from baseline)
| | | | | | | | | | | | | | | | | | | | |
| Time of Assessment | | Somryst Group | | Control Group | | p-value |
End of Treatment Period (Week 9) | | 62.8 | % | | 14.0 | % | | <0.0001 |
Month 6 Follow-Up | | 56.2 | % | | 18.9 | % | | <0.0001 |
Month 12 Follow-Up | | 59.3 | % | | 25.2 | % | | <0.0001 |
Real World Evidence and Health Economics & Outcomes Research
Pear continues to generate RWE and HEOR data in patients with chronic insomnia.
A real-world deployment with 7,414 patients who utilized Somryst evaluated the benefits and health outcomes. In the real-world experience, patients utilized the product for nine weeks, consisting of six treatment modules. Data were collected on the FDA-reviewed endpoint, ISI, patient reported outcomes, and over 300,000 sleep diaries. As shown in the figures below, ISI, SOL, and WASO scores saw meaningful decreases as patients progressed through the six treatment modules.
Patient Engagement, ISI Score, SOL and WASO by completed core of the six cores in Somryst.

Pear Therapeutics, Inc. | 2022 Form 10-K |Page 30
Patient Engagement, ISI Score, SOL and WASO by completed core of the six cores in Somryst.
Pear published its first health economic data evaluating Somryst in 2022. In a real-world claims analysis of patients with chronic insomnia exposed to the digital CBTi, researchers found an overall reduction in hospital events in the 24 months post-treatment compared to the 24 months pre-treatment, with facility related cost-savings of an estimated $2,059 per patient in the post-treatment period. An analysis comparing treated patients compared to control patients over 24 months, presented at the EU The Professional Society for Health Economics and Outcomes Research (ISPOR) in Europe and the American Managed Care Pharmacy (AMCP) Nexus in fall 2022, observed a reduction of hospital facility services compared to control, resulting in health care utilization savings of an estimated $8,202 per patient.
Dynamic Evidence Generation
We believe that PDTs have an advantage over molecular therapeutics because data of product usage, patient-reported, and clinical outcomes of PDTs may be captured more rapidly. These data collected in studies as well as in commercial use facilitate regular evidence generation. Pear’s platformplatforms can facilitate linking healthcare resource utilization in the form of public and private insurance claims. Pear is able to rapidly scale evidence (clinical and health economic) and analyze across care-settings, specific populations, regions, and customers.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 2931
This evidence continuum is a key competitive advantage of Pear and its platformplatforms. Pear plans to continue to expand the evidence for its PDTs in additional RCTs, RWE, and health-economic evaluation to support continued product improvement, and to generate information for clinicians, and value for payors.
Our Development Pipeline
PDTs have the potential to treat many diverse diseases directly and enhance health outcomes for patients. We have 14Currently, we have paused the development of all the product candidates in our pipeline, including ten product candidates in psychiatry and neurology, our two initial areas of focus to support our self-commercialization strategy, and four candidates in other therapeutic areas as set forth below.. Below is a summary of the pipeline products:
*Dartmouth transaction is with a researcher employed by Dartmouth. Pear has no direct contractual relationship with Dartmouth relating to this content.
**Karolinska transaction is with individual researchers who are employed by the Karolinska Institute. Pear has no direct contractual relationship with the Karolinska Institute relating to this content.
***Services agreement with Ironwood to evaluate a PDT in GI diseases.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 3032
We are initially building a portfolio of psychiatry and then neurology-focused PDTs. We also have early-stage programsThe Company paused its investment in its pipeline of product candidates as part of a restructuring it implemented on July 25, 2022, in order to prioritize certain commercial efforts. The Company has suspended development of its pipeline candidates subject to obtaining additional funding due to increased revenue or other financing activities.
We have built a portfolio of PDT product candidates that is primarily focused on psychiatric conditions. Over time, if we are able to consummate a strategic alternative and sufficient funding is available, we could expand into other therapeutic areas. We anticipate the first two areas in GI, oncology, and cardiovascular indications. Our near-term focus iswhich we would invest if we return to expandinvesting in our leadership in psychiatry by developing PDTs for the treatment ofpipeline are Alcohol Use Disorder (“AUD”), depression, anxiety,AUD”) and schizophreniadepression. These fourtwo additions have the potential to help fill the need for additional treatment options and enhance the offering to mental and behavioral health clinicians and psychiatrists while supporting payors’ need to reduce morbidity and cost in these indications.
Additional details about our four key research and development projects in our pipeline are set forth below:
1)Alcohol Use Disorder Product Candidate
•Current Stage: POC with Breakthrough Designation from FDA
•Patient Population: Alcohol Use Disorder
•Product Candidate Number: PEAR-009
•Description of Therapeutic Modality and Method of Action (“MoA”):
i.Mobile Device
ii.PEAR-009 is a multimodal PDT candidate that delivers digital CRA, Fluency Training, CM and Behavioral Activation and validated assessments to improve clinical outcomes of AUD
•Expected Use: Alone (“monotherapy”) with potential for future combination (“combination therapy”)
•Objective: To develop a PDT that could be used alone or in combination with another medication to treat patients with AUD
•Current Status and Next Steps: This product candidate is at POC stage. Pear is currently evaluating data to determine the next steps.
•Risks and Uncertainties: There are limited therapies for AUD currently and no existing FDA-market authorized PDTs for AUD on the market. For example, it is uncertain that further clinical testing would support the safety and effectiveness of the product and its market authorization, or whether the product, if authorized, would be receive reimbursement from third party payors.
2)Schizophrenia Product Candidate
•Current Stage: POC
•Patient Population: Psychotic Spectrum Disorders including patients with schizophrenia
•Product Candidate Number: PEAR-004
•Description of Therapeutic Modality and MoAs:
i.Mobile Device
ii.Pear-004 is a multimodal PDT candidate that delivers BA, cognitive restructuring, illness self-management, cognitive behavioral therapy, and validated assessments to improve clinical outcomes in patients with schizophrenia
•Expected Use: Combination therapy with antipsychotics
•Objective: To develop a PDT that could be used in combination with pharmacotherapy to treat patients with schizophrenia
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 31
•Current Status and Next Steps This product candidate has completed the POC stage and is ready for a potential pivotal randomized clinical trial to support potential FDA submission. Pear has development plans outlining specific types of studies and purposes, but the clinical trial planning has not commenced.
•Risks and Uncertainties: There are many failures of therapeutics while in development for schizophrenia and/or psychotic spectrum disorder. There are no existing FDA-market authorized PDTs for schizophrenia on the market.
3)Anxiety Product Candidate
•Current Stage: POC
•Patient Population: Generalized Anxiety Disorder (“GAD”)
•Product Candidate Number: PEAR-011
•Description of Therapeutic Modality and MoAs:
i.Mobile Device
ii.PEAR-011 is a multimodal PDT candidate that delivers Panic Modulation (a digitized form of psychotherapy where the patient is instructed and learns to employ mechanisms of modulating their fear, anxiety or panic response to stimuli to desensitize them to future exposures to the inciting stimuli), BA, CBT, sleep hygiene and stimulus control plus medication management and validated assessments to improve clinical outcomes in patients with anxiety disorder
•Expected Use: Potential for monotherapy and combination therapy
•Objective: To develop a PDT that could be used alone and/or in combination with pharmacotherapy to treat patients with anxiety disorder
•Current Status and Next Steps: This product candidate is at the POC stage and is ready for a potential pivotal randomized clinical trial, which is the next step to support potential FDA submission. Pear has development plans outlining specific types of studies and purposes, but the clinical trial planning has not commenced.
•Risks and Uncertainties: There are many failures of therapeutics while in development for psychiatric diseases like Anxiety-Spectrum Disorders. There are no existing FDA-market authorized PDTs for anxiety on the market.
4)Depression Product Candidate
•Current Stage: Discovery
•Patient Population: Major Depressive Disorder and additional Depression populations
•Product Candidate Number: PEAR-015
•Description of Therapeutic Modality and MoAs:
i.Mobile Device
ii.PEAR-015 is a multimodal PDT candidate that delivers CBT, BA with Ecologic Momentary Assessment (repeated sampling of a patient’s behaviors and experiences on which brief interventions by the software are delivered), sleep content plus medication and validated assessments to improve clinical outcomes in patients with depression. Pear acquired two clinical
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 32
state depression product candidates in the fourth quarter of 2021 and is in the process of integrating those product candidates into PEAR-015.
•Expected Use: Potential for monotherapy and combination therapy
•Objective: To develop a PDT that could be used alone and/or in combination with pharmacotherapy to treat patients with Depression
•Current Status and Next Steps: This product candidate is at the Discovery stage. Once the new product candidates are integrated into PEAR-015, it will require clinical studies, and if promising, would then in the future be evaluated in a potential pivotal clinical trial to support potential FDA submission.
•Risks and Uncertainties: There are many failures of therapeutics while in development for psychiatric diseases like Depression. There are no existing FDA-market authorized PDTs for Depression on the market.
•Development activities for product candidates haveOur ability to initiate development activities for product candidates will a number ofbe subject to our ability to increase revenue or obtain additional funding in addition to other risks and uncertainties. All therapeutic development activities have risk and probabilities of success that can vary by disease indication. In many of the disease areas Pear is developing product candidates there is a long history of failure. The areas of mental and behavioral health or neuropsychiatry, which is a primary focus for Pear, have had notable failures across biotech, pharma, and digital health. Each of Pear’s pipeline candidates have technical, clinical, regulatory, and commercial risk. See the heading “Risk Factors—Risks Related to Pear’s Products— If the Company is not able to develop and release new products, or successful enhancements, new features and modifications to the Company’s existing products, the Company’s business, financial condition and results of operations could be materially and adversely affected”” in Part I, Item 1A of this Form 10-K for more information on risks related to product development.
Additional specific risks and uncertainties also include but are not limited to:
•each potential therapeutic must achieve levels of use by the patient in their disease population sufficient for the evidence-based treatment or mechanism of action to exert its behavioral and/or biologic effect. If levels of product use or engagement are not high enough a study may not be successful and a product candidate may not be viable;
•even if patients do engage at sufficient levels, the proposed evidence-based treatment or scientific mechanism of action may not be effective in achieving the desired clinical outcome;
•clinical trials may fail for numerous reasons including operational complexities, difficulty in measurement, challenges in enrollment, adverse events and safety events, poor endpoints, variabilities amongst sites, protocol deviations, and many others; and
•the technology, such as algorithms or engineering, may fail.
Ongoing PDT discovery Future PDT discovery
Currently, Pear is not pursuing any PDT discovery efforts. If a successful strategic transaction is consummated and sufficient funding is available, Pear could re-engage in discovery.
There are two categories of PDT’s interventional mechanisms of action. The first is delivery of neurobehavioral interventions. These are scientific and evidence-based practices, many of which we know work from face-to-face delivery, and target affected disease-specific neurocircuitry. Examples include CBT, cognitive restructuring, behavioral activation, exposure therapy, and CM. The second category is dose-optimization. In diseases where pharmacotherapy exists, PDTs can be integrated with the molecular therapeutic to assess patient response and optimize medication dose and frequency. Use cases include titrating a medication to therapeutic effect, optimizing chronic dosing, tapering medication, and recommending dosing as needed when a disease exacerbation is occurring.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 33
We believe utilizing both categories of interventional mechanisms of action will facilitate further pipeline expansion. Pear’s existing PDT candidates in neurologic diseases including epilepsy, pain, and multiple sclerosis, have potential to increase the patient populations and addressable markets.
Gastroenterology, cardiology, and oncology are particularly promising therapeutic areas. In oncology, combining dose-optimization of immuno-oncology drugs (which could target both solid or liquid tumors) with neurobehavioral mechanisms of action, multi-modal PDTs could optimize both the treatment of cancer directly and the behavioral disorders that often coincide with cancer treatment. For example, by utilizing labs, patient-reported outcomes, and sensor data, recommendations to refine the dosing and frequency of chemotherapeutic agents to reduce dose-limiting toxicities and maximize overall-survival can be integrated with CBTi for insomnia in patients undergoing treatment for cancer. Similar approaches may be deployed in GI and cardiovascular.
In addition, we willPear Therapeutics, Inc. | 2022 Form 10-K |Page 33
In addition, over time, if we are able to consummate a strategic alternative and sufficient funding is available, we could continue our strategic pursuit of enabling technologies, such as digital sensors, that may be used to enhance our products and product candidates.
We expect to significantly grow our pipeline through internal discovery efforts, in-licensing, and acquisition. We continue to seek to accumulate early-stage clinical assets and technologies for development. We believe our scalable infrastructure to discover, develop, and deliver these products to patients will allow us to be a partner of choice for development, regulatory, and commercial stage dealspotentially enhance our products and product candidates.
Competition
The pharmaceutical, biotechnology, and digital health industries are characterized by rapidly advancing technologies, intense competition, and an emphasis on proprietary products. While Pear believes that Pear’s technology, development experience, and scientific knowledge provide Pear with competitive advantages, Pear faces potential competition from many different sources, including large pharmaceutical and biotechnology companies, digital health companies, and academic institutions. To drive continual technology advantage, Pear seeks patent protection and establishes collaborative arrangements for the research, development, manufacturing, and commercialization of evidence-based therapeutics. Any products that Pear successfully develops and commercializes will compete with new therapies that may become available in the future.
Pear competes in the segments of the pharmaceutical, biotechnology, and other related markets that develop therapeutics as treatments, in particular treatments for addiction (reSET and reSET-O) and insomnia (Somryst). There are many other companies that have commercialized and/or are developing such treatments for addiction and insomnia, including large pharmaceutical and biotechnology companies such as Alkermes and their product Vivitrol, Orexo and their product Zubsolv, SandozIndivior and their product Suboxone, Braeburn and their product Brixadi, Pfizer and their product Halcion, Merck and their product Belsomra, Sunovion and their product Lunesta and Sanofi and their product Ambien.
Pear’s competitors in the digital health space, with specific focus in addiction and insomnia have created non-regulated products, are not supported by randomized controlled trials to support treatment claims, such as Dynamicare, CBT4CBTCHESS Health and Orexo and their product Modia, and Big Health and their products Sleepio, Pzizz, and CalmDaylight.
Rather, Pear believes the competitive landscape is best understood by comparing the primary mechanism of action (behavioral support/intervention or improving medication adherence and tracking); to the business model for patient acquisition (apps marketed direct-to-consumer; tech-enabled healthcare services offered to members of health plans, most often those of self-insured employers; or regulated products prescribed by providers).
While some solutions have evolved to include elements of various mechanisms such as behavioral support, reminders for medication adherence, or remote monitoring and transmission of biometric data, in Pear’s view, each has a primary mechanism for affecting disease and a clearly defined model for acquiring patients or consumers.
To Pear’s knowledge, Pear’s reSET and reSET-O are the only FDA-authorized PDTs with a direct treatment-related claim for addiction, and Pear’s Somryst is the only FDA-authorized PDT with a direct treatment-related claim for
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 34
chronic insomnia that can be prescribed by providers and reimbursed by insurance. This has required Pear to generate significant evidence to demonstrate safety, effectiveness, clinical outcomes and distinct impact on the total cost of care. While many early market entrants (in fact, nearly 360350,000 health and wellness apps are now available in Apple’s App Store) are making marketing claims related to the ability to help treat addiction and insomnia, Pear believes the landscape will change dramatically when new solutions that can be prescribed by providers and covered by insurance become broadly available.
There are a number of companies in the prescription digital therapeutics space, but none of these companies have commercialized an FDA-authorized prescription digital therapeutic to target addiction or insomnia at this time. Other than Pear, we are aware of threesix companies that have received FDA-authorization to make medical claims as prescription digital therapeutics. This includes (i) Akili Interactive Labs and their product Endeavor for ADHD, (ii) Mahana Therapeutics and their product Parallel for IBS, and(iii) Nightware for patients with PTSD that are experiencing nightmares, (iv) AppliedVR and their product RelieVRx for chronic pain, (v) Luminopia for patients with amblyopia, and (vi) metaME and their product Regulora for abdominal pain due to IBS. Other companies of which Pear is aware in the space that have not received FDA-authorization and are not authorized to make medical claims but may pursue FDA-authorization in the future include: (i) Click Therapeutics in Depression, (ii) Better Therapeutics in
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 34
Cardiometabolic, (iii) Cognoa in Autism, (iv) AppliedVR in Pain, (v) Orexo and GAIA AG in Depression, (vi) MedRhythms in Stroke, and (vii) Blue Note Therapeutics for Cancer Related Anxiety and Depression.
License Agreements
We have rights to use and exploit technologies disclosed in issued and pending patents and other therapeutic content under licenses and/or assignment agreements with other entities (collectively, “License Agreements”). We consider the commercial terms of these License Agreements, which provide for milestone and royalty payments, and their provisions regarding diligence, insurance, indemnification, and other similar matters, to be reasonable and customary for our industry.
The material License Agreements related to our three commercial products are those with The Invention Science Fund I, LLC (“ISF”), Red 5 Group, LLC (“Red 5”), and BeHealth Solutions, LLC (“BeHealth”).
The Invention Science Fund I, LLC
We entered into a contribution and license agreement with ISF in February 2015, as amended on February 28, 2018 (the “ISF Agreement”), pursuant to which ISF grants the Company certain licenses under specified patent rights to develop and commercialize licensed products either independently and/or in combination with a drug for use in connection with the treatment of central nervous system disorders. The oldest patent in the family is expected to expire in 2028.
Under the terms of the ISF Agreement, we are required to pay a minimum annual royalty, capped at low-seven figures per year. From inception through December 31, 20212022, we have paid ISF $2.6 million in minimum annual royalties, and for the year ended December 31, 20212022, recorded annual minimum royalties payable of $1.0 million. The ISF Agreement also requires us to pay a royalty percentage in the low single digits on net sales of products covered by the in-licensed patents that do not include drugs. To date, no royalties have been paid on net sales of products covered by the in-licensed patents that do not include drugs. In addition, as to sales of products covered by the in-licensed patents that do include drugs, the ISF Agreement requires us to pay either (i) a royalty percentage on net sales in the low single digits or (ii) an annual fee based on net sales. To date, no royalties have been paid on net sales of products covered by the in-licensed patents that include drugs.
Under the ISF Agreement, ISF covenants that neither it nor any of its affiliates will sue, or assert any claim against (or cause any third party to sue or assert any claim against) us for infringement of any patent covered under the ISF Agreement with respect to our permitted use of such patents under the ISF Agreement (the “Covenant”).
In consideration of the Covenant granted to us in the ISF Agreement, the ISF Agreement requires us to pay an annual covenant fee (the “Covenant Fee”) in a variable amount ranging from mid-to-high five digits to a low-six figures per year through December 2030. Through December 31, 2021, we have paid ISF $0.3 million in annual Covenant Fees.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 35
2022, we have paid ISF $0.3 million in annual Covenant Fees. The ISF Agreement will terminate upon the expiration or invalidation of all of the patents subject to the license. Additionally, ISF may terminate the ISF Agreement upon certain breaches by us of the terms of the ISF Agreement, and we may terminate the ISF Agreement 90 days after providing written notice to ISF. The Covenant, and our related obligation to pay the annual Covenant Fee, may be terminated by us 30 days after providing written notice to ISF.
Red 5 Group, LLC
In January 2015, we entered into a software license agreement with Red 5, and on March 21, 2018, the parties entered into an amended and restated software license agreement which was further amended on July 1, 2021 to clarify certain terms and increase the royalty rate by a de minimis amount (such agreement, as amended, “Amended Red 5 Group License”). Under the Amended Red 5 Group License, Red 5 grants us, inter alia, an exclusive (other than a limited reserved right for ongoing academic studies), worldwide, sublicensable, royalty-bearing license to develop and commercialize integrated products incorporating certain technology and documentation materials relating to the treatment of psychological and substance use disorders. Under the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 35
Amended Red 5 Group License, we agreed to use commercially reasonable efforts to develop integrated products in accordance with a development plan, to introduce any integrated products that gain regulatory approval into the commercial markets, to market integrated products that have gained regulatory approval following such introduction into the market, and to make integrated products that have gained regulatory approval reasonably available to the public.
In March 2018, the parties amended the original Red 5 agreement, and we subsequently paid an amendment fee in the high-six figures in consideration for expanding the scope of our exclusive license. On July 1, 2021, the parties amended the Amended Red 5 Group License to further clarify certain terms and increase the royalty rate by a de minimisde minimis amount.
To the extent achieved, we are obligated to pay Red 5 up to an aggregate of $0.4 million if certain milestones related to product regulatory approval and commercial sales are achieved in respect to a software/drug combination, which we are not currently pursuing. Each such regulatory and sales milestone is payable only once.
The Amended Red 5 Group License also requires us to pay a single digit royalty percentage on net revenue from integrated products. Through December 31, 20212022, we have recorded $1.39 million in royalties to Red 5. We are also required to make annual maintenance fee payments capped at a sum in the low-six figures per year, creditable against future royalty and milestone payment obligations. From inception through December 31, 20212022, we have recorded $1.0.4 million of annual maintenance fees under the terms of the agreement. In addition, we have a credit in the mid-six figures against future royalty and milestone payment obligations.
The Amended Red 5 Group License will continue in perpetuity unless terminated in accordance with its terms, under which Red 5 may terminate the license upon certain breaches by us of the terms of the license, and we may terminate the license 60 days after providing written notice to Red 5.
BeHealth Solutions, LLC
We entered into an assignment, license, and services agreement with BeHealth, effective March 24, 2018 (the “BeHealth Agreement”), which relates to software, documentation, and intellectual property rights in and to certain therapeutic content related to cognitive behavioral treatment for insomnia and other sleep disorders that BeHealth licensed from the University of Virginia Patent Foundation d/b/a University of Virginia Licensing & Ventures Group (“UVA LVG”).
Under the terms of the BeHealth Agreement, we paid an initial up-front fee in the mid-six figures to BeHealth, and during the year ended December 31, 2019, we paid a milestone payment to BeHealth of $0.8 million upon the FDA’s acceptance of a filing seeking marketing authorization for Somryst that was recorded as a research and development expense in the consolidated statement of operations and comprehensive loss for the period then ended. During the year ended December 31, 2020, we paid a milestone payment to BeHealth of $0.8 million upon the FDA’s marketing authorization of Somryst. The milestone payment was capitalized in other long-term assets and amortized on a straight-line basis to cost of product revenue over the estimated useful life, which was ten
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 36
years at initial recognition. During the year ended December revenue over the estimated useful life, which was ten years at initial recognition. During the year ended December 31, 2021, we recorded $1.0 million related to the achievement of a commercial milestone. Each such regulatory and sales milestone is payable only once, and no other milestone payment may become due and payable after March 24, 2028. Assuming thatIn the future, should the Company pursue sales of Somryst and we meet the remaining commercial milestones, the Company could be obligated to make payments of up to an additional $26.0 million in the aggregate upon achievement of various annual commercial milestones and a mid-to-high-single-digit percentage royalty on net sales to BeHealth.
The Company’s forecasted future revenue and expense cash flow projections indicated the carrying amounts of this intangible asset was not recoverable, and therefore the Company recorded an impairment expense of $0.8 million, which is included in cost of revenue in the Company's consolidated statement of operations, during the year ended December 31, 2022.
Because the underlying license agreement (with exclusive rights relating to the content and non-exclusive rights relating to the underlying platform) between BeHealth and UVA LVG was assigned to us, we owe a royalty percentage in the mid-single digits on net sales of integrated products to UVA LVG. We have the right to sublicense our rights under the assigned license agreement, and we will be required to pay a percentage of such sublicense
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 36
income in the low-double digits to UVA LVG. We are required to make annual minimum royalty payments in the low-five figures to UVA LVG under the assigned license agreement, which payments are creditable against royalty payments on a year-by-year basis. Through December 31, 20212022, we have recorded only de minimis royalties under the UVA LVG assigned license agreement, and we have paid de minimis royalties, $2.5 million in milestone payments, and $0.5 million in other fees to BeHealth. We have a buyout option in the assigned license agreement under which we may pay UVA LVG a lump sum of $1.0 million in exchange for a perpetual royalty-free license in the field.
The underlying license from UVA LVG will continue in perpetuity unless terminated in accordance with the terms of the assigned license agreement, under which UVA LVG may terminate the license upon certain breaches by us of the terms of the license, and we may terminate the license at any time upon 30 days’ written notice to UVA LVG.
Intellectual Property
In addition to securing regulatory authorization for our PDTs, we make strategic use of various intellectual property regimes: patents;Emerging Growth Company Status (JOBS Act)
Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can choose not to take advantage of the extended transition period and comply with the requirements that apply to non-emerging growth companies, and any such election to not take advantage of the extended transition period is irrevocable. We are an “emerging growth company” as defined in Section 2(A) of the Securities Act of 1933, as amended, and have elected to take advantage of the benefits of this extended transition period.
We expect to use this extended transition period for complying with new or revised accounting standards that have different effective dates for public business entities and nonpublic business entities until the earlier of the date we (a) are no longer an emerging growth company or (b) affirmatively and irrevocably opts out of the extended transition period provided in the JOBS Act. This may make it difficult or impossible to compare our financial results with the financial results of another public company that is either not an emerging growth company or is an emerging growth company that has chosen not to take advantage of the extended transition period exemptions because of the potential differences in accounting standards used.
In addition, we have chosen to take other exemptions and reduced reporting requirements provided by the JOBS Act and are not required to, among other things:
(a) provide an auditor’s attestation report on Pear’s system of internal control over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act;
(b) provide all of the compensation disclosure that may be required of non-emerging growth public companies under the Dodd-Frank Wall Street Reform and Consumer Protection Act; and
(c) disclose certain executive compensation-related items, such as the correlation between executive compensation and performance and comparisons of the Chief Executive Officer’s compensation to median employee compensation.
Intellectual Property
In addition to securing regulatory authorization for our PDTs, we make strategic use of various intellectual property regimes: patents, copyrights;, trademarks;, and trade secrets. We strive to protect and enhance the proprietary technology, inventions, and improvements that are commercially important to the development of our business, including seeking, maintaining, and defending patent rights, whether developed internally or licensed from third parties. We also rely on copyrights (including copyright registrations for product source code), on trademarks, and on trade secrets relating to our proprietary PDT product development platform to develop, strengthen, and maintain our proprietary position in the PDT field.
Our commercial success may depend in part on our ability to: consummate a strategic alternative, obtain and maintain patent and other proprietary protection for commercially important technology, inventions and know-how related to our business; defend and enforce our patents, copyrights, trademarks and other proprietary rights; preserve the confidentiality of our trade secrets; and operate without infringing the valid, enforceable patents, and other proprietary rights;
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 37
preserve the confidentiality of our trade secrets; and operate without infringing the valid, enforceable patents, and proprietary rights of third parties. Our ability to limit third parties from making, using, selling, offering to sell, or importing our products may depend on the extent to which we have rights under valid and enforceable licenses, patents, copyrights, or trade secrets that cover these activities. In some cases, enforcement of these rights may depend on third-party licensors or co-owners. With respect to both company-owned and licensed intellectual property, we cannot be sure that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents that may be granted to us in the future will be commercially useful in protecting our commercial products.
Patent expiration dates noted below refer to statutory expiration dates and do not take into account any potential patent term adjustment or extension that may be available.
Solely Owned Patent Assets
We exclusively own eleven patent families directed to a variety of different aspects related to PDTs. One family is directed to ensuring data security in the treatment of diseases and disorders using digital therapeutics. One US patent has issued in this family and will expire in 2038. A continuation patent application has been filed claiming the benefit of the allowed application, and, if granted, will expire in 2038. A Patent Cooperation Treaty application has been filed in this family, and national phase entry applications have been filed in Europe, Japan, Singapore, New Zealand, China, Israel, Canada, and Australia. Another family is directed to the optimization of buprenorphine
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 37
; of those, patents have issued in Canada, New Zealand, Australia, Israel and Japan. Another family is directed to the optimization of buprenorphine induction. If granted, the patent(s) in this family would expire in 2039. A Patent Cooperation Treaty application has been filed in this family, and national phase entry applications have been filed in Australia, Canada, China, Europe, Israel, Japan, New Zealand, Singapore, and South Korea. Another family is directed to the treatment of migraines. If granted, the patent(s) in this family would expire in 2040. A Patent Cooperation Treaty application has been filed in this family, and national phase entry applications have been filed in Australia, Canada, China, Europe, Israel, Japan, New Zealand, Singapore, and South Korea. Another family is directed to treating depressive symptoms associated with multiple sclerosis. If granted, the patent(s) in this family would expire in 2040. A Patent Cooperation Treaty application has been filed in this family, and national phase entry applications have been filed in Australia, Canada, China, Europe, Israel, Japan, New Zealand, Singapore, and South Korea. Another family is directed to clinical curation of crowdsourced data. If granted, the patent(s) in this family would expire in 2040. A Patent Cooperation Treaty application has been filed in this family, and national phase entry applications have been filed in Australia, Canada, China, Europe, Israel, Japan, New Zealand, Singapore, and South Korea. Another family is directed to visualizing and modifying treatment of a patient utilizing digital therapeutics. If granted, the patent(s) in this family would expire in 2040. A Patent Cooperation Treaty application has been filed in this family, but this application has lapsed. Another family is directed to generating and administering digital therapeutic placebo and shams. If granted, the patent(s) in this family would expire in 2041. A Patent Cooperation Treaty application has been filed in this family, with aand national phase entry date in January 2023applications have been filed in Australia, Canada, China, Europe, Israel, Japan, New Zealand, Singapore, and South Korea.
Another family is directed to a display screen or portion thereof with an animated graphical user interface. A US design patent has issued in this family and will expire in 2035. A European design has been registered in this family and will expire in 2024. Another family is directed to a display screen or portion thereof with a graphical user interface. A US design patent has issued in this family and will expire in 2035. A European design has been registered in this family and will expire in 2024. Another family is directed to a display screen or portion thereof with an animated graphical user interface. A US design patent has issued in this family and will expire in 2036. A European design has been registered in this family and will expire in 2024. Another family is directed to a graphical user interface. A European design has been registered in this family and will expire in 2024.
Jointly Owned Patent Assets
We jointly own two patent families directed to a variety of different aspects related to digital therapeutics. One family is directed to the treatment of depressive symptoms and disorders utilizing digital therapies. If granted, the patent(s) in this family would expire in 2040. A Patent Cooperation Treaty application has been filed in this family,
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 38
and national phase entry date applications have been filed in Australia, Canada, China, Europe, Israel, Japan, New Zealand, Singapore, and South Korea. Another family is directed to treatment utilizing antipsychotics in combination with digital therapies. If granted, the patent(s) in this family would expire in 2039. A Patent Cooperation Treaty application has been filed in this family, and national phase entry applications have been filed in Australia, Canada, China, Europe, Japan, South Korea, SingaporeIsrael, Japan, New Zealand, China, Israel, CanadaSingapore, and AustraliaSouth Korea.
Licensed Patent Assets
As discussed above in “License Agreements - ISF” above, we are the exclusive licensee within a broad field of use to a portfolio of patents directed to combinational drug and prescription digital therapy treatment. Nineteen US patents have been allowed in this portfolio, the patents expiring between 2028 and, at the latest, 2036. Those patents encompass more than 550 granted claims directed to drug/software combinations and that may potentially cover aspects of current or future PDTs. One US patent applications is pending in this portfolio. If granted, the pending patent application would expire in 2028. As exclusive licensee, we playhave played a pivotal role in the ongoing prosecution of this portfolio.
Registered Copyrights
In addition to our portfolio of utility and design patents, we hold copyright in each of our PDTs and pursue federal copyright registration where appropriate. We have registered copyright in the source code for each of our commercial products with the US Copyright Office.
Registered Trademarks
Beyond patent and copyright protection, we also protect our valuable trademarks and associated brand recognition by registering trademarks with the US Patent and Trademark Office. We own seven US trademark
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 38
and in certain foreign jurisdictions. We own seven US trademark registrations covering goods and services in multiple classes. Our federally registered trademarks are PEAR THERAPEUTICS, the Pear logo, RESET, RESET-O, and SOMRYST. CertainOur pending application to register RESET-A and one of our RESET and RESET-O trademark applications and registrations are the subject of ongoing opposition and cancellation proceedings, respectively. , and decisions in our favor in prior opposition and cancellation proceedings brought by the same entity related to other RESET and RESET-O trademark applications and registrations, respectively, are the subject of an appeal pending in a federal district court in California. We have registered PEAR THERAPEUTICS in the European Union. We have registered SOMRYST in the United Kingdom.
Government Regulation
Insurance and Coverage
In the US and markets in other countries, patients generally rely on third-party payors to reimburse all or part of the costs associated with their treatment. Adequate coverage and reimbursement from governmental healthcare programs, such as Medicare and Medicaid, and commercial payors is critical to new product acceptance. Pear’s ability to successfully commercialize Pear’s product candidates will depend in part on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from government health administration authorities, private health insurers, and other organizations. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels. The availability of coverage and extent of reimbursement by governmental and private payors is essential for most patients to be able to afford treatments. Sales of product candidates that Pear may identify will depend substantially, both domestically and abroad, on the extent to which the costs of Pear’s product candidates will be paid by health maintenance, managed care, pharmacy benefit, and similar healthcare management organizations, or reimbursed by government health administration authorities, private health coverage insurers, and other third-party payors.
There is also significant uncertainty related to the insurance coverage and reimbursement of newly approved products, and coverage may be more limited than the purposes for which the product is approved by FDA or comparable foreign regulatory authorities. In the US, a key decision-maker about reimbursement for new products is the Centers for Medicare & Medicaid Services (“CMS”), an agency within the US Department of Health and
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 39
Human Services (“HHS”). CMS decides whether and to what extent a new product will be covered and reimbursed under Medicare, and several US private payors tend to follow CMS to some degree.
In determining reimbursement, payors generally consider whether the product is:
•a covered benefit under its health plan;
•safe, effective, and medically necessary;
•appropriate for the specific patient;
•cost-effective; and
•neither experimental nor investigational.
Each payor determines whether or not it will provide coverage for a treatment, what amount it will pay the manufacturer for the treatment, and on what tier of its formulary it will be placed. The position on a payor’s list of covered drugs, biological products, and medical devices, or formulary, generally determines the co-payment that a patient will need to make to obtain the therapy and can strongly influence the adoption of such therapy by patients and clinicians. Patients who are prescribed treatments for their conditions and clinicians prescribing such services generally rely on third-party payors to reimburse all or part of the associated healthcare costs. Patients are unlikely to use Pear’s products unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of Pear’s products. There may be significant delays in obtaining such coverage and reimbursement for newly authorized products, and coverage may be more limited than the purposes for which the product is authorized by FDA.
Healthcare Laws and Regulations
Pear is subject to applicable fraud and abuse and other healthcare laws and regulations, including, without limitation, the US federal Anti-Kickback Statute and the US federal False Claims Act, which may constrain the business or financial arrangements, and relationships through which Pear sells, markets and distributes Pear’s
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 39
, and distributes Pear’s products. In particular, the promotion, sales and marketing of healthcare items and services, as well as certain business arrangements in the healthcare industry (e.g., healthcare providers, clinicians and third-party payors), are subject to extensive laws designed to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, structuring and commission(s), certain customer incentive programs, and other business arrangements generally. Pear also may be subject to patient information and privacy and security regulation by both the federal government and the states in which Pear conducts its business. The applicable federal and state healthcare laws and regulations that may affect Pear’s ability to operate include, but are not limited to:
•the federal Anti-Kickback Statute, which prohibits, among other things, persons or entities from knowingly and willfully soliciting, receiving, offering or paying any remuneration (including any kickback, bribe or rebate), directly or indirectly, overtly or covertly, in cash or in kind, to induce, or in return for, the purchase, lease, order, arrangement, or recommendation of any good, facility, item or service for which payment may be made, in whole or in part, under a federal healthcare program, such as the Medicare and Medicaid programs. A person or entity does not need to have actual knowledge of the federal Anti-Kickback Statute or specific intent to violate it to have committed a violation. Violations are subject to civil and criminal fines and penalties for each violation, plus up to three times the remuneration involved, imprisonment, and exclusion from government healthcare programs. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal False Claims Act or federal civil money penalties. On December 2, 2020, the Office of Inspector General (“OIG”) published further modifications to the federal Anti-Kickback Statute. Under the final rules, OIG added safe harbor protections under the Anti-Kickback Statute for certain coordinated care and value-based arrangements among clinicians, providers, and others. This rule, with certain exceptions, became effective January 19,
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 40
2021. Implementation of this change is currently under review by the Biden administration and may be amended or repealed. Pear continues to evaluate what effect, if any, the rule will have on Pear’s business;
•the federal civil and criminal false claims laws and civil monetary penalty laws, such as the federal False Claims Act, which impose criminal and civil penalties and authorize civil whistleblower or qui tam actions, against individuals or entities for, among other things: knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent; knowingly making, using or causing to be made or used, a false statement of record material to a false or fraudulent claim or obligation to pay or transmit money or property to the federal government or knowingly concealing or knowingly and improperly avoiding or decreasing an obligation to pay money to the federal government. A person can be held liable under the federal False Claims Act even when they do not submit claims directly to government payors if they are deemed to “cause” the submission of false or fraudulent claims. The federal False Claims Act also permits a private individual acting as a “whistleblower” to bring actions on behalf of the federal government alleging violations of the federal False Claims Act and to share in any monetary recovery;
•Applicableapplicable privacy laws and regulations, including HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (“HITECH”), and their respective implementing regulations, including the Final Omnibus Rule published in January 2013, which impose requirements on certain covered healthcare providers, health plans, and healthcare clearinghouses as well as their respective business associates, independent contractors or agents of covered entities, that perform services for them that involve the creation, maintenance, receipt, use, or disclosure of, individually identifiable health information relating to the privacy, security and transmission of individually identifiable health information. HITECH also created new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions. In addition, there may be additional federal, state and non-US laws which govern the privacy and security of health and other personal information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 40
•Thethe US federal transparency requirements under the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act of 2010, or collectively, the Affordable Care Act (“ACA”) including the provision commonly referred to as the Physician Payments Sunshine Act, and its implementing regulations, which requires applicable manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program to report annually to CMS, information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, as well as ownership and investment interests held by the physicians described above and their immediate family members. Effective January 1, 2022, these reporting obligations will extend to include transfers of value made to certain non-physician providers such as physician assistants and nurse practitioners; and
•federal consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm consumers.
•Additionally, Pear is subject to state equivalents of each of the healthcare laws and regulations described above, among others, some of which may be broader in scope and may apply regardless of the payor. Many states in the US have adopted laws similar to the federal Anti-Kickback Statute and False Claims Act, and may apply to Pear’s business practices, including, but not limited to, research, distribution, sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental payors, including private insurers. Several states also impose other marketing restrictions or require medical device manufacturers to make marketing or price disclosures to the state, and may also require that medical device manufacturers, such as Pear, maintain licenses to manufacture and/or distribute products within their state. State laws also govern the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 41
often are not preempted by HIPAA, thus complicating compliance efforts. There are ambiguities as to what is required to comply with these state requirements and if Pear fails to comply with an applicable state law requirement, Pear could be subject to penalties.
Data Privacy and Security Laws
Numerous federal and state laws and regulations govern the collection, use, disclosure, storage and transmission of personally identifiable information, including protected health information. These laws and regulations, including their interpretation by governmental agencies, are subject to frequent change and could have a negative impact on Pear’s business. In addition, in the future, industry requirements or guidance, contractual obligations, and/or legislation at both the federal and the state level may limit, forbid or regulate the use or transmission of health information outside of the US.
These varying interpretations can create complex compliance issues for Pear and Pear’s partners and potentially expose Pear to additional expense, adverse publicity and liability, any of which could adversely affect Pear’s business.
Federal and state consumer protection laws are increasingly being applied by the FTC and states’ attorneys general to regulate the collection, use, storage, and disclosure of personal or personally identifiable information, through websites or otherwise, and to regulate the presentation of website content.
The security measures that Pear and Pear’s third-party vendors and subcontractors have in place to ensure compliance with privacy and data protection laws may not protect Pear’s facilities and systems from security breaches, ransomware acts of vandalism or theft, computer viruses, misplaced or lost data, programming, and human errors or other similar events. Even though Pear provides for appropriate protections through Pear’s agreements with Pear’s third-party vendors, Pear still has limited control over their actions and practices. A breach of privacy or security of personally identifiable health information may result in an enforcement action, including criminal and civil liability, against us. Pear is not able to predict the extent of the impact such incidents may have on Pear’s business. Enforcement actions against Pear could be costly and could interrupt regular operations, which may adversely affect Pear’s business. While Pear has not received any notices of violation of the applicable privacy
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 41
and data protection lawsand data protection laws, and we believe Pear is in compliance with such laws, there can be no assurance that Pear will not receive such notices in the future.
There is ongoing concern from privacy advocates, regulators, and others regarding data privacy and security issues, and the number of jurisdictions with data privacy and security laws has been increasing. Also, there are ongoing public policy discussions regarding whether the standards for de-identification, anonymization or pseudonymization of health information are sufficient, and the risk of re-identification sufficiently small, to adequately protect patient privacy. Pear expects that there will continue to be new proposed and amended laws, regulations and industry standards concerning privacy, data protection and information security in the US, such as the California Consumer Privacy Act (“CCPA”), which went into effect on January 1, 2020 and has been amended several times. FurtherEffective January 1, 2023, a new California privacy law, the CPRA, was passed by California voters on November 3, 2020(with certain provisions having retroactive effect to January 1, 2022) went into effect. The CPRA will create additional obligations with respect to processing and storing personal information that are scheduled to take effect on January 1, 2023 (with certain provisions having retroactive effect to January 1, 2022). Additionally, a new Virginia privacy law, the VCDPA, was signed into law on March 2, 2021 and is also scheduled to take effect on January 1, 2023, and the Colorado Privacy Act (“CPA”) which was passed by the state legislature and is awaiting the governor’s signature, will take effect on July 1, 2023. The VCDPA and CPA will impose many similar obligations regarding the processing and storing of personal information as the CCPA and the CPRAcreates additional obligations with respect to processing and storing personal information. Other states in the Unites States also are considering omnibus privacy legislation, and industry organizations regularly adopt and advocate for new standards in these areas. While the CCPA, CPRA, CPA and VCDPA contain contains exceptions for certain activities involving protected health information already regulated under HIPAA, Pear cannot yet determine the impact the CCPA, CPRA, CPA, VCDPA or other such future laws, regulations and standards may have on Pear’s business.
Healthcare Legislative Reform
In both the US and certain foreign jurisdictions, there have been a number of legislative and regulatory changes to the healthcare system that could impact Pear’s ability to sell Pear’s products profitably. In particular, in 2010, the ACA was enacted, which contained a number of measures aimed at controlling healthcare costs.
Since its enactment, there have been numerous judicial, administrative, executive, and legislative challenges to certain aspects of the ACA, and Pear expects there will be additional challenges and amendments to the ACA in the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 42
future. Additionally, the Trump administration issued various Executive Orders which eliminated cost-sharing subsidies and various provisions that would impose a fiscal burden on states or a cost, fee, tax, penalty, or regulatory burden on individuals, healthcare providers, health insurers, or manufacturers of pharmaceuticals or medical devices and Congress has introduced several pieces of legislation aimed at significantly revising or repealing the ACA. Further, on December 20, 2019, President Trump signed into law the Further Consolidated Appropriations Act (H.R. 1865), which repealed the Cadillac tax, the health insurance provider tax, and the medical device excise tax. It is unclear whether the ACA will be overturned, repealed, replaced, or further amended. Pear cannot predict what affect further changes to the ACA would have on Pear’s business, especially given the new administration.
Other legislative changes have been proposed and adopted in the US since the ACA was enacted. In August 2011, the Budget Control Act of 2011, among other things, created measures for spending reductions by Congress. A Joint Select Committee on Deficit Reduction, tasked with recommending a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, was unable to reach required goals, thereby triggering the legislation’s automatic reduction to several government programs. This includes aggregate reductions of Medicare payments to providers up to 2% per fiscal year, and, due to subsequent legislative amendments, will remain in effect through 2030 unless additional congressional action is taken. The CARES ActCoronavirus Aid, Relief, and Economic Security Act, as well as subsequent legislation, suspended the sequestration payment adjustment percentage of 2% applied to all Medicare Fee-for-Service claims from May 1, 2020 through December March 31, 20202022. The Consolidated Appropriations Act, 2021, extended the suspension period to March 31, 2021. An Act to Prevent Across-the-Board Direct Spending Cuts, and for Other Purposes, signed into law on April 14, 2021, extends the suspension periodSequestration resumed at 1% for dates between April 1, 2022 and June 30, 2022, and then increased to the pre-pandemic level to December 31, 2021of 2% for dates after July 1, 2022.
There has been increasing legislative and enforcement interest in the US with respect to prescription-pricing practices. It is unclear what effect such legislative and enforcement interest may have on prescription devices.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 42
Further, it is unclear whether the Biden administration will challenge, reverse, revoke or otherwise modify the prior administration’s executive and administrative actions.
Pear expects that these and other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that Pear received for any approved device, which could have an adverse effect on customers for Pear’s product candidates. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors.
There have been, and likely will continue to be, legislative and regulatory proposals at the foreign, federal and state levels in the US directed at broadening the availability of healthcare and containing or lowering the cost of healthcare. The implementation of cost containment measures or other healthcare reforms may prevent Pear from being able to generate revenue, attain profitability or commercialize Pear’s products. Such reforms could have an adverse effect on anticipated revenue from product candidates that Pear may successfully develop and for which Pear may obtain regulatory approval and that may affect Pear’s overall financial condition and ability to develop product candidates. If Pear or any third parties Pear may engage are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if Pear or such third parties are not able to maintain regulatory compliance, Pear’s current or any future product candidates Pear may develop may lose any regulatory approval that may have been obtained and Pear may not achieve or sustain profitability.
FDA Regulation
United States
Pear’s productsPDTs are medical devices subject to extensive and ongoing regulation by FDA under the Federal Food, Drug, and Cosmetic Act (“FD&C Act”) and its implementing regulations, as well as other federal and state regulatory bodies in the US The laws and regulations govern, among other things, product design and development, preclinical and clinical testing, manufacturing, packaging, labeling, storage, recordkeeping and reporting, clearance, authorization, or approval, marketing, distribution, promotion, import and export and post-marketing surveillance. Failure to comply with applicable requirements may subject a device and/or its manufacturer to a variety of
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 43
administrative sanctions, such as issuance of warning letters, import detentions, civil monetary penalties, mandatory recalls, and/or judicial sanctions, such as product seizures, injunctions and criminal prosecution.
Generally, establishments that design and/or manufacture devices are required to register their establishments with the FDA. They also must provide the FDA with a list of the devices that they design and/or manufacture at their facilities.
The FDA enforces its requirements by market surveillance and periodic visits, both announced and unannounced, to inspect or re-inspect equipment, facilities, laboratories and processes to confirm regulatory compliance. These inspections may include the manufacturing facilities of subcontractors. Following an inspection, the FDA may issue a report, known as a Form 483, listing instances where the manufacturer has failed to comply with applicable regulations and/or procedures or, if observed violations are sufficiently severe and urgent, a warning letter. If the manufacturer does not adequately respond to a Form 483 or warning letter, the FDA make take enforcement action against the manufacturer or impose other sanctions or consequences, which may include:
•cease and desist orders;
•injunctions or consent decrees;
•civil monetary penalties;
•recall, detention or seizure of products;
•operating restrictions, partial or total shutdown of production facilities;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 43
•refusal of or delay in granting requests for 510(k) clearance, de novo authorization, or premarket approval of new products or modified products;
•withdrawing 510(k) clearances, de novo authorizations, or premarket approvals that are already granted;
•refusal to grant export approval or export certificates for devices; and
•criminal prosecution.
FDA’s Pre-market Clearance, Authorization, and Approval Requirements
Each prescription digital therapeutic Pear seeks to commercially distribute in the US will require either a prior de novo authorization, 510(k) clearance, unless it is exempt, or a Premarket Approval (“PMA”) from FDA under its medical device authorities. Generally, if a new device has a predicate that is already classified as Class II, FDA will allow that new device to be marketed under a 510(k) clearance. If there is no legally marketed predicate device and general controls alone or with special controls provide reasonable assurance of safety and effectiveness, FDA will allow the new device to be marketed under a de novo authorization; otherwise, a PMA is required. Medical devices are classified into one of three classes—Class I, Class II or Class III—depending on the degree of risk associated with each medical device and the extent of control needed to provide reasonable assurance of safety and effectiveness. Class I devices are deemed to be low risk and are subject to the general controls of the FD&C Act, such as provisions that relate to: adulteration; misbranding; registration and listing; notification; records and reports; and good manufacturing practices. Class II devices are moderate risk. They are subject to general controls and may also be subject to special controls. Special controls include, for example, performance standards, post market surveillance, patient registries and guidance documents. Devices deemed by FDA to pose the greatest risk, such as life-sustaining, life-supporting or implantable devices, or devices deemed not substantially equivalent to a previously cleared 510(k) device, are placed in Class III. A Class III device cannot be marketed in the US unless FDA approves the device after submission of a PMA. However, there are some Class III devices for which FDA has not yet called for a PMA. For these devices, the manufacturer must submit a pre-market notification and obtain 510(k) clearance in order to commercially distribute these devices. FDA can also impose sales, marketing or other restrictions on devices in order to assure that they are used in a safe and effective manner. There is no guarantee that a submission to FDA will result in a clearance, authorization, or approval. In addition, various review processprocesses and goals to which applications are currently subject are the result of Medical Device User Fee Act (“MDUFA”) IV,
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 44
which expires after September 30, 2022; negotiations for enactment of a MDUFA V law and adoption of associated goals are ongoing.
While most Class I and some Class II devices may be marketed without prior FDA authorization, most other medical devices can be legally sold within the US only if the FDA has: (i) approved a PMA application prior to marketing, generally applicable to most Class III devices; (ii) cleared the device in response to a 510(k) submission, generally applicable to some Class I and most Class II devices; or (iii) authorized the device to be marketed through the de novo classification process, generally applicable for novel Class I or II devices.
510(k) Clearance Pathway
Product marketing in the US for most Class II and a limited number of Class I devices typically follows the 510(k) premarket notification pathway. When a 510(k) clearance is required, Pear must submit a pre-market notification to FDA demonstrating that Pear’s proposed device is substantially equivalent to a legally marketed device, referred to as a “predicate device.” A predicate device may be a previously 510(k) cleared device or a Class III device that was in commercial distribution before May 28, 1976 for which the FDA has not yet called for PMA applications, or a product previously placed in Class II or Class I through the de novo classification process. To demonstrate substantial equivalence, the manufacturer must show that the proposed device has the same intended use as the predicate device, and it either has the same technological characteristics, or different technological characteristics and the information in the pre-market notification demonstrates that the device is equally safe and effective and does not raise different questions of safety and effectiveness as compared to the predicate device. FDA may require further information, including clinical data, to make a determination regarding substantial equivalence. If FDA determines that the device, or its intended use, is not substantially equivalent to a previously cleared device or use, FDA will place the device into Class III.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 44
There are three types of 510(k)s: traditional, special and abbreviated. Special 510(k)s are for devices that are modified, and the modification needs a new 510(k) but does not affect the intended use or alter the fundamental scientific technology of the device. Abbreviated 510(k)s are for devices that conform to a recognized standard. FDA has a user fee goal to apply no more than 90 calendar review days to traditional and abbreviated 510(k) submissions. During the process, FDA may issue an Additional Information request, which stops the clock. The applicant has 180 days to respond. Therefore, the total review time could be up to 270 days. The special and abbreviated 510(k)s are intended to streamline review, and FDA intends to process special 510(k)s within 30 days of receipt, although it can take longer, if FDA has questions. There are no guarantees that FDA meet its goals, although it has agreed to try to meet its 90 calendar day decision goals in 95% of 510(k) submissions for FY 2022.
After a device receives 510(k) clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, requires a new 510(k) clearance or could require a PMA approval or de novo classification. The FDA requires each manufacturer to make this determination in the first instance, but the FDA can review any such decision. If the FDA disagrees with a manufacturer’s decision not to seek a new 510(k) clearance for the modified device, the agency may retroactively require the manufacturer to seek 510(k) clearance, de novo classification, or PMA approval. The FDA also can require the manufacturer to cease marketing and/or recall the modified device until 510(k) clearance, de novo authorization, or PMA approval is obtained.
De Novo Classification
When it is determined there is no legally marketed predicate device, the de novo process provides a pathway to classify novel low- or moderate-risk medical devices for which general controls alone, or general and special controls, provide reasonable assurance of safety and effectiveness. Medical device types that FDA has not previously classified as Class I, II or III are automatically classified into Class III regardless of the level of risk they pose. The de novo classification procedure allows a manufacturer whose novel device is automatically classified into Class III to request down-classification of its medical device into Class I or Class II on the basis that the device presents low or moderate risk, rather than requiring the submission and approval of a PMA application. Prior to the enactment of the FDA Safety and Innovation Act of 2012 (“FDASIA”), a medical device could only be eligible for de novo classification if the manufacturer first submitted a 510(k) pre-market notification and received a
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 45
determination from FDA that the device was not substantially equivalent. FDASIA streamlined the de novo classification pathway by permitting manufacturers to request de novo classification directly without first submitting a 510(k) pre-market notification to FDA and receiving a not substantially equivalent determination. Under FDASIA, FDA is required to classify the device within 120 days following receipt of the de novo application. If the manufacturer seeks reclassification into Class II, the manufacturer must include a draft proposal for special controls that are necessary to provide a reasonable assurance of the safety and effectiveness of the medical device. In addition, FDA may reject the reclassification petition if it identifies a legally marketed predicate device that would be appropriate for a 510(k) or determines that the device is not low to moderate risk or that general controls would be inadequate to control the risks and special controls cannot be developed. Devices that are classified into Class I or Class II through a de novo classification request may be marketed and used as predicates for future premarket notification 510(k) submissions.
FDA has a user fee goal to review a de novo request in 150 calendar review days (which FDA agreed to try and meet for 70% of De Novo submissions in FY 2022). During the process, FDA may issue an Additional Information request, which stops the clock. The applicant has 180 days to respond. Therefore, the total review time could be as long as 330 days.
Pre-market Approval Pathway
A Class III product not eligible for either 510(k) clearance or de novo classification must follow the PMA approval pathway. The PMA application process is generally more expensive, rigorous, lengthy, demanding, and uncertain than the 510(k) premarket notification or de novo process. A PMA application must be supported by extensive data, including but not limited to technical, preclinical, clinical trials, manufacturing, marketing history, design, controls, and labeling to demonstrate to FDA’s satisfaction reasonable assurance of safety and effectiveness of the device.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 45
FDA may convene an advisory panel of experts from outside FDA to review and evaluate the application and provide recommendations to FDA as to the safety and effectiveness of the device. Although FDA is not bound by the advisory panel decision, the panel’s recommendations are important to FDA’s overall decision-making process. In addition, FDA may conduct a preapproval inspection of the manufacturing facility to ensure compliance with the Quality System Regulation (“QSR”), which imposes elaborate testing, control, documentation, and other quality assurance requirements. The agency also may inspect one or more clinical sites to assure compliance with FDA’s regulations.
FDA has a user fee goal to review a PMA in 180 calendar review days, if the submission does not require advisory committee input, or 320 review days if the submission does require advisory committee input. FDA has agreed to try to meet this goal in most cases for FY 2022, though it is not guaranteed. During the process, FDA may issue a major deficiency letter, which stops the review clock. The applicant has up to 180 days to respond. Therefore, the total review time could be up to 360 days, if the submission does not require advisory committee input, or 500 days if the submission does require advisory committee input.
Upon completion of the PMA review, FDA may: (i) approve the PMA which authorizes commercial marketing with specific prescribing information for one or more indications, which can be more limited than those originally sought and can include post-approval conditions that the FDA believes necessary to ensure the safety and effectiveness of the device including, among other things, restrictions on labeling, promotion, sale and distribution; (ii) issue an approvable letter which indicates FDA’s belief that the PMA is approvable and states what additional information FDA requires, or the post-approval commitments that must be agreed to prior to approval; (iii) issue a not approvable letter which outlines steps required for approval, but which are typically more onerous than those in an approvable letter, and may require additional clinical trials that are often expensive and time consuming and can delay approval for months or even years; or (iv) deny the application. If FDA issues an approvable or not approvable letter, the applicant has 180 days to respond, after which FDA’s review clock is reset.
Even after approval of a PMA, a new PMA or PMA supplement may be required in the event of a modification to the device, its labeling or its manufacturing process. Supplements to a PMA often require the submission of the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 46
same type of information required for an original PMA, except that the supplement is generally limited to that information needed to support the proposed change from the product covered by the original PMA.
Breakthrough Devices
The Breakthrough Devices Program is a voluntary program intended to expedite the review, development, assessment and review of certain medical devices that provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating human diseases or conditions for which no approved or cleared treatment exists or that offer significant advantages over existing approved or cleared alternatives. Submissions for devices designated as Breakthrough Devices will receive priority review, meaning that the review of the submission is placed at the top of the appropriate review queue and receives additional review resources, as needed and available. Although Breakthrough Device designation or access to any other expedited program may expedite the development or clearance/authorization/approval process, it does not change the standards for clearance/authorization/approval. Designation for any expedited review procedure does not ensure that we will ultimately obtain regulatory clearance or approval for such product.
Clinical Trials
Clinical trials are almost always required to support a PMA, are often required for a de novo authorization, and are sometimes required for 510(k) clearance. Clinical trials may also be conducted or continued to satisfy post-approval requirements for devices with PMAs. In the US, for significant risk devices, these trials require submission of an application for an investigational device exemption (“IDE”) to FDA prior to initiating clinical trials. The IDE application must be supported by appropriate data, such as animal and laboratory testing results, showing it is safe to test the device in humans and that the testing protocol is scientifically sound. The IDE must be approved in advance by FDA for a specific number of patients at specified study sites. A 30-day waiting period after the submission of each IDE is required prior to the commencement of clinical testing in humans. If the FDA disapproves the IDE within this 30-day period, the clinical trial proposed in the IDE may not begin. During the trial, the clinical
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 46
trial sponsor must comply with FDA’s IDE requirements for investigator selection, trial monitoring, reporting and recordkeeping. The investigators must obtain patient informed consent, rigorously follow the investigational plan and study protocol, control the disposition of investigational devices and comply with all reporting and recordkeeping requirements. Clinical trials for significant risk devices may not begin until the IDE application is approved by FDA and the appropriate institutional review boards (“IRBs”) at the clinical trial sites. An IRB is an appropriately constituted group that has been formally designated to review and monitor medical research involving subjects and which has the authority to approve, require modifications in, or disapprove research to protect the rights, safety and welfare of human research subjects. A nonsignificant risk device does not require FDA approval of an IDE; however, the clinical trial must still be conducted in compliance with various requirements of FDA’s IDE regulations and be approved by an IRB at the clinical trials sites. FDA or the IRB at each site at which a clinical trial is being performed may withdraw approval of a clinical trial at any time for various reasons, including a belief that the risks to study subjects outweigh the benefits or a failure to comply with FDA or IRB requirements. Even if a trial is completed, the results of clinical testing may not demonstrate the safety and effectiveness of the device, may be equivocal or may otherwise not be sufficient to obtain approval, authorization, or clearance of the product.
Sponsors of applicable clinical trials of devices are required to register with www.clinicaltrials.gov, a public database of clinical trial information. Information related to the device, patient population, phase of investigation, study sites and investigators and other aspects of the clinical trial is made public as part of the registration.
Although the QSR does not fully apply to investigational devices, the requirement for controls on design and development does apply. The clinical trial sponsor also must manufacture the investigational device in conformity with the quality controls described in the IDE application and any conditions of IDE approval that the FDA may impose with respect to manufacturing.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 47
Ongoing Regulation by FDA
Even after a device receives clearance, authorization, or approval and is placed on the market, numerous regulatory requirements apply. These include:
•establishment registration and device listing;
•the QSR, which requires manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation, and other quality assurance procedures during all aspects of the manufacturing, design, development, distribution, and labeling process;
•labeling regulations and FDA prohibitions against the promotion of products for uncleared, unapproved, or “off-label” uses and other requirements related to promotional activities;
•medical device reporting regulations, which require that manufactures report to FDA if their device may have caused or contributed to a death or serious injury, or if their device malfunctioned and the device or a similar device marketed by the manufacturer would be likely to cause or contribute to a death or serious injury if the malfunction were to recur;
•corrections and removal reporting regulations, which require that manufacturesmanufacturers report to FDA field corrections or removals if undertaken to reduce a risk to health posed by a device or to remedy a violation of the FD&C Act that may present a risk to health; and
•post market surveillance regulations, which apply to certain Class II or III devices when necessary to protect the public health or to provide additional safety and effectiveness data for the device.
FDA regulations require manufacturers to register with FDA and to list the devices they market. Additionally, the California Department of Health Services (“CDHS”) requires manufacturers to register within the state. Following these registrations, FDA and CDHS inspect manufacturers on a routine basis for compliance with the QSR and applicable state regulations. These regulations require that Pear manufacture Pear’s products and maintain related documentation in a prescribed manner with respect to manufacturing, testing and control activities. Other states
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 47
might also require non-resident licenses concerning distribution of devices into their states. Pear is also subject to other federal, state and local laws and regulations relating to safe working conditions, laboratory and manufacturing practices. Failure to comply with applicable regulatory requirements can result in enforcement action by FDA or state authorities, which may include any of the following sanctions:
•warning or untitled letters, fines, injunctions, consent decrees and civil penalties;
•customer notifications, voluntary or mandatory recall or seizure of Pear’s products;
•operating restrictions, partial suspension or total shutdown of production;
•delay in processing submissions or applications for new products or modifications to existing products;
•withdrawing clearance, authorization, and/or approvals that have already been granted; and
•criminal prosecution.
The medical device reporting laws and regulations require manufacturers to provide information to FDA when they receive or otherwise become aware of information that reasonably suggests their devices may have caused or contributed to a death or serious injury as well as a device malfunction that likely would cause or contribute to death or serious injury if the malfunction were to recur. Failure to properly identify reportable events or to file timely reports, as well as failure to address each of the observations to FDA’s satisfaction, can subject a manufacturer to warning letters, recalls, or other sanctions and penalties.
Advertising, marketing and promotional activities for devices are also subject to FDA and US Federal Trade Commission (“FTC”) oversight and must comply with the statutory standards of the FD&C Act and the FDA’s implementing regulations, and the Federal Trade Commission Act and FTC’s implementing regulations. The FDA’s
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 48
and FTC’s oversight authority review of marketing and promotional activities encompasses, but is not limited to, direct-to-consumer advertising, healthcare provider-directed advertising and promotion, sales representative communications to healthcare professionals, promotional programming and promotional activities involving electronic media. The FDA also regulates industry-sponsored scientific and educational activities that make representations regarding product safety or efficacy in a promotional context.
Manufacturers of medical devices are permitted to promote products solely for the uses and indications set forth in the approved or cleared product labeling, and all claims must be truthful, non-misleading, and adequately substantiated. In addition, although healthcare providers may use medical devices in an off-label manner in accordance with the practice of medicine, FDA prohibits marketed devices from being marketed for off-label uses and regulates the advertising of certain devices as well. FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability, including substantial monetary penalties and criminal prosecution, including False Claims Act liability for products covered under the federal health care programs.
Finally, newly discovered or developed safety or effectiveness data may require changes to a marketed product’s labeling, including the addition of new warnings and contraindications, and also may require the implementation of other risk management measures. Also, new government requirements, including those resulting from new legislation, may be established, or FDA’s policies may change, which could delay or prevent regulatory clearance or approval of Pear’s products under development.
Pear has three FDA-authorized products: reSET, reSET-O, and Somryst. Pear’s first product, reSET, obtained a de novo authorization as a Class II device. Pear’s second and third products, reSET-O and Somryst, obtained 510(k) clearance as Class II devices.
Human Capital
The Company believes it has a talented, motivated, and dedicated team, and. Management is committed to supporting the development of all of its team members and to continuously building on its strong culture that operates at the
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 48
intersection of biology and software technology. As of December 31, 2021continuously building on its strong culture that operates at the intersection of biology and software technology. As of December 31, 2022, we had approximately 300200 employees, that includes, primarily full-time employees but also part-time employees and interns. We also engage consultants and temporary workers when needed. No employees were represented by labor unions or subject to collective bargaining agreements.
On July 25, 2022, the Company restructured of its operations and executed a reduction in workforce of 25 full-time employees. On November 14, 2022, the Company announced a second reduction in workforce, further reducing our headcount by approximately 59 employees.
Workplace Practices and Policies
The Company is committed to providing a workplace free of harassment or discrimination based on race, color, religion, sex, sexual orientation, gender identity or expression, pregnancy, age, national origin, disability status, genetic information, protected veteran status, or any other characteristic protected by law. The Company is an equal opportunity employer committed to inclusion and diversity.
In managing our business, we strive to develop and implement policies and programs that support our business goals, maintain competitiveness, promote shared fiscal responsibility among our company and our employees, strategically align talent within our organization and reward performance, while also managing the costs of such policies and programs. Our employees are supported with training and development opportunities to pursue their career paths and to ensure compliance with our policies. We adhere to our code of business conduct and ethics (the "Code of Business Conduct and Ethics"), which sets forth a commitment to our stakeholders, including our employees, to operate with integrity and mutual respect.
During the COVID-19 pandemic, we have taken and continue to take necessary actions to safeguard the health of our employees. Steps we have taken include enhancing office safety measures, encouraging hygiene practices advised by health authorities, restricting non-essential business travel
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 49
advised by health authorities, restricting non-essential business travel at certain times during the pandemic, requiring proof of COVID-19 vaccinations, including booster shots as applicable, for employees, and third-party personnel who wish to visit our officesvisitor protocols, and flexibility in remote working for all of our employees, which has continued to evolve due to the resurgence of infections as a result of mutations of the virus. We continue to actively monitor risks related to COVID-19 and proper application of our safety protocol to remain aligned with federal, state, and local laws, regulations, and guidelines. We are committed to providing consistent, transparent communication to employees around safe practices, quarantine and testing protocols, vaccine availability, and policies and procedures for safely returning to office work for those employees. We believe our technology platform will continue to support the effectiveness of our employees that choose to work remotely. Our employees and their families are also supported with access to a variety of flexible and convenient health and welfare programs, including benefits that support their physical and mental health.
Inclusion and Diversity
The Company is committed to actively recruiting from diverse backgrounds, providing training and development opportunities, fostering an inclusive culture, and ensuring equitable pay for employees, and is continuing to focus on increasing diverse representation at every level of the Company. As of December 31, 20212022, our board isof directors was comprised of three female directors and four male directors, and our executive team iswas comprised of three female officers and four male officers, and our leadership team is.
Two female officers departed the Company, Kathy Jeffery, our Chief People Officer, departed on February 17, 2023, and Julia Strandberg, our Chief Commercial Officer, departed on March 10, 2023.
As of December 31, 2022, our leadership team was comprised of six femalesseven women and five males. six men; one male vice president departed the Company in March 2023. Three female vice presidents and one male vice president will be departing the Company in April 2023.
Compensation, Benefits, Growth, and Development
Pear seeks to attract, develop, and retain talented professionals from the healthcare and tech sectors. We utilize a pay-for-performance compensation structure that involves a combination of salary, bonus (which includes a personal component and a company component, the proportions of which are based on the level of the employee), and stock-based compensation awards. We also have a robust rewards and recognition program. We seek to develop all of our employees, which we refer to as “Pearmates”, with a learning and development program. We strive to stretch our highest potential employees by promoting from within where possible.
Pear has a number of Employee Resource Groups: employee-led, voluntary groups, based around a shared interest or goal, and aligned with the mission and objectives of the company. The following are our current Employee Resource Groups and their missions:
•DEI Committee - to enable a diverse, equitableinclusive, and inclusiveengaging organization that embraces and leveragescapable of leveraging our differences and enhances a sense of belonging forto outperform, innovate, and lead the all Pearmates;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 49
way in enhancing diversity, equity, and inclusion within healthcare; •Pear Women - to support the women of Pear by providing a safe space to discuss their thoughts and concerns, with an emphasis on career growth;
•Pear Impact Committee - to create empathy by engaging employees to deepen their understanding of the patients we serve with love, care, and compassion& Allies - to create and maintain a strong environment that focuses on development, mentoring, leadership, and empowering women’s success at Pear and to be the leading digital health company for women employees;
•Employee Engagement Council - a cross-functional and cross-geographic team empowered to improve the employee experience at Pear; and
•Peargivers - to support employees whoFun Committee: To support Pearmates by encouraging them to take a moment out of their day to have caregiver responsibilities with resources and socioemotional support to enable their maximized contributions to Pear.some fun!
Engagement
The Company believes that open and honest communication among Pearmates, managers, and leadership fosters an open, collaborative work environment where everyone can participate, develop and thrive. Pearmates are encouraged to come to their managers with questions, feedback or concerns, and we regularly conduct surveys , or concerns, and we regularly conduct surveys
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 50
that gauge employee sentiment in areas like the Company culture, communication, leadership, work environment, well-being, rewards and recognition, compensation and benefits, and career development.
Available Information
We file annual, quarterly, and current reports, proxy statements, and other information with the SEC. The SEC maintains an Internet website at www.sec.gov that contains reports, proxy and information statements and other information about issuers, like us, that file electronically with the SEC. We also maintain a website at https://peartherapeutics.com/. We make available, free of charge, on our investor relations website at https://investors.peartherapeutics.com/financial-information/sec-filings, our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to these reports as soon as reasonably practicable after electronically filing or furnishing those reports to the SEC. Information contained on our website is not a part of or incorporated by reference into this prospectus and the inclusion of our website and investor relations website addresses in this prospectus is an inactive textual reference onlyor any other report we file with or furnish to the SEC.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 5051
ITEM 1A. RISK FACTORS
Summary of Risk Factors
Below is a summary of the principal factors that make an investment in our Class A common stock speculative or risky. This summary does not address all of the risks that we face. Additional discussion of the risks summarized in this risk factor summary, and other risks that we face, can be found below under the heading “Risk Factors” and should be carefully considered, together with other information in this Form 10-K and our other filings with the SEC before making an investment decision regarding our Class A common stock.
Risks relating to our Strategic Alternative Process, Potential Strategic Transaction, and Financial Position:
•We have decided to seek a strategic acquisition, business combination, partnership or other change of control transaction, and there is no guarantee that this strategic path will be successful.
• If we are unable to successfully complete a strategic acquisition, we may be forced to cease operations altogether or file for bankruptcy protection.
•We may not realize any additional value in a strategic transaction.
•If we are successful in completing a strategic transaction, we may be exposed to other operational and financial risks.
•We may not fully realize the expected cost savings and/or operating efficiencies from our restructuring activities and our ability to consummate a strategic transaction depends on our ability to retain our employees required to consummate such transaction.
•We may become involved in securities litigation that could divert management’s attention and harm the company’s business, and insurance coverage may not be sufficient to cover all costs and damages.
• We have identified conditions and events that raise substantial doubt about our ability to continue as a going concern, we need substantial funding, and if we are unable to raise capital on favorable terms, our business, financial condition, and results of operation could be materially and adversely affected.
•We have incurred net losses in every year since our inception and anticipate that we will continue to incur net losses for the foreseeable future.
•Our credit agreement with Perceptive Credit Holdings III, LP restricts our current and future operations, particularly our ability to respond to changes or to take certain actions.
•Perceptive claims that certain events of default have occurred under the Perceptive Credit Facility. Upon the occurrence of an Event of Default, Perceptive may declare the full amount outstanding under the Perceptive Credit Agreement due and payable, which would materially and adversely affect our ability to continue operations.
•The amount of our future losses is uncertain and our quarterly and annual operating results may fluctuate significantly or fall below the expectations of investors or securities analysts, each of which may cause our stock price to fluctuate or decline.
Risks relating to our business and industry include:
• The failure of our prescription digital therapeutics to achieve and maintain market acceptance and adoption by patients and physicians would cause our business, financial condition and results of operationoperations to be materially and adversely affected.
• The insurance coverage and reimbursement status of novel products, such as prescription digital therapeutics, is uncertain and only a limited number of healthcare insurers have agreed to reimburse purchases of our products. Failure to obtain or maintain adequate coverage and reimbursement for our products would substantially impair our ability to generate revenue.
• The market for prescription digital therapeutics is new, rapidly evolving, and increasingly competitive, as the healthcare industry in the US is undergoing significant structural change, which makes it difficult to forecast demand for our products.
• Our product and product candidates represent novel and innovative solutions, and negative perception of any product or product candidate that we develop could adversely affect our ability to conduct our business, obtain marketing authorizations, or identify regulatory pathways to market for such product candidate.
• Our future depends on the continued contributions of our senior management team and our ability to attract and retain other highly qualified personnel; in particular, Corey McCann, our President and Chief Executive Officer, and Christopher Guiffre, our Chief Financial Officer and Chief Operating Officer, are critical to our future vision and strategic direction.
• A limited number of healthcare insurers have agreed to reimburse purchases of our products, and there is no assurance that additional .
• We rely significantly upon Access Agreements from third-parties for the sale of our products and, if the opportunities for Access Agreements decline, such reliance could adversely affect our results.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 52
•A limited number of healthcare insurers have agreed to reimburse purchases of our products, and there is no assurance that healthcare insurers will agree to reimburse purchases of our products in the future.
• If we are unable to expand our marketing infrastructure, we may fail to increase the usage of our products and platform to meet our forecasts.
• Our products are made available via the Apple Store and the Google Play Store and supported by third-party infrastructure. If our ability to access those markets or access necessary third-party infrastructure was stopped or otherwise restricted or limited, it would materially and adversely affect our business.
• We face competition and new products may emerge that provide different or better alternatives for treatment of the conditions that our products are authorized to treat. Many of our current and future competitors have or will have significantly more resources.
Risks relating to our financial position include:
• We have a history of significant losses, anticipate increasing expenses in the future, and may not be able to achieve or maintain profitability.
•Our credit agreement with Perceptive restricts our current and future operations, particularly our ability to respond to changes or to take certain actions.
• Due to the resources required for the development of our pipeline, and depending on our ability to access capital, we will have to prioritize the development of certain product candidates over others. We may fail to expend our limited resources on product candidates that may have been more profitable or for which there is a greater likelihood of success, which would cause our business, financial condition and results of operations to be materially and adversely affected.
• We will need substantial additional funding, If we are unable to raise capital when needed or on terms favorable to us, our business, financial condition, and results of operation could be materially and adversely affected.
Risks relating to our intellectual property and technology include:
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 51
Risks relating to our intellectual property and technology include:
• Limitations on our ability to maintain or obtain patent protection and/or the patent rights relating to our products and product candidates may limit our ability to prevent third parties from competing against us.
• We in-license patents and content from third parties to develop our products and product candidates. If we had a dispute or fail to comply with obligations in the agreements with a third-party licensor, we could lose rights that are important to our business, or it could materially and adversely affect our ability to commercialize the product or product candidate affected by the dispute.
Risks relating to our products include:
• Our current product candidates are in various stages of development. Our product candidates may fail in development or suffer delays that adversely affect their commercial viability. If we fail to maintain clearance, de novo classification or authorization to market our products for expanded indications, or if we are delayed in obtaining such marketing authorizations, our business, prospects, results of operations and financial condition could be materiallyThe success of our products or any new products depends on several factors, including regulatory review timelines, timely completion, competitive pricing, adequate quality testing, integration with new and existing technologies in our products and third-party collaborators’ technologies and adversely affectedoverall market acceptance.
Risks relating to our regulatory compliance and legal matters include:
• We operate in a highly regulated industry and are subject to a wide range of federal, state and local laws, rules and regulations, including US Food and Drug Administration (“FDAFDA”) and Federal Trade Commission (“FTC”) regulatory requirements and laws pertaining to fraud and abuse in healthcare, that affect nearly all aspects of our operations. Failure to comply with these laws, rules and regulations, or to obtain and maintain required licenses, could subject Pear to enforcement actions, including substantial civil and criminal penalties, and might require Pear to recall or withdraw a product from the market or cease operations. Any of the foregoing could materially and adversely affect our business, financial condition and results of operations.
• Security breaches, ransomware attacks and other disruptions to our information technology structure could compromise our information, disrupt our business and expose us to significant liability, which would cause our business and reputation to suffer, and we may be unable to maintain and scale theour technology underlying our offerings.
• The regulatory framework for digital health products is constantly evolving. Increasingly stringent regulatory requirements could create barriers to our development and introduction of new products. Conversely, in the event that regulatory requirements are lowered, competitors could potentially enter the prescription digital therapeutic market and compete against us more easily. Either of the foregoing could materially harm our business.
• Premarket clearances, authorizations, and approvals for new or significantly modified devices could be denied or significantly delayed.
Risks relating to our financial reporting include:
•As a public reporting company, we will beare subject to rules and regulations established from time to time by the SEC regarding our internal control over financial reporting. If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, investors may lose confidence in the accuracy of our financial reports, which would harm our business and the trading price of our Class A common stock. Our management will be required to evaluate the effectiveness of our internal control over financial reporting.
• Our management has identified certain internal control deficiencies that constitute material weaknesses. If we fail to maintain an effective system of disclosure controls and internal control over financial reporting, our ability to produce timely and accurate financial statements or comply with applicable regulations could be impaired.
Risks relating to ownership of our Class A common stock and Warrants:
• The exercise of Warrants for our stock would increase the number of shares eligible for future resale in the public market and result in dilution to our stockholders. Such dilution will increase if more of our shares are redeemed.
• We may redeem unexpired Warrants prior to their exercise at a time that is disadvantageous to you, thereby making your Warrants worthless.
• Future resales of the Class A common stock may cause the market price of our securities to drop significantly, even if our business is doing well.
• If we fail to satisfy the continued listing requirements of Nasdaq such as the corporate governance requirements or the minimum closing bid price requirement, Nasdaq may take steps to delist our securities.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 52
Risk Factors
You should carefully consider the risks and uncertainties described below, together with the information included elsewhere in this Annual Report on Form 10-K and other documents we file with the SEC. The risks and uncertainties described below are those that we have identified as material, but are not the only risks and uncertainties facing us. Our business is also subject to general risks and uncertainties that affect many other companies, such as overall US and non-US economic and industry conditions including a global economic slowdown, geopolitical events, changes in laws or accounting rules, fluctuations in interest and exchange rates, terrorism, international conflicts, major health concerns, natural disasters or other disruptions of expected economic and business conditions. Additional risks and uncertainties not currently known to us or that we currently believe are immaterial also may impair our business operations and liquidity.
This section should be read in conjunction with Part II, Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the consolidated financial statements and accompanying notes in Part II, Item 8, Financial Statements, of this Form 10-K.
Risks Related to Our Business and Industry
The failure of our prescription digital therapeutics to achieve and maintain market acceptance and adoption by patients and physicians would cause our business, financial condition and results of operation53
Risk Factors
You should carefully consider the risks and uncertainties described below, together with the information included elsewhere in this Annual Report on Form 10-K and other documents we file with the SEC. The risks and uncertainties described below are those that we have identified as material, but are not the only risks and uncertainties facing us. Our business is also subject to general risks and uncertainties that affect many other companies, such as overall US and non-US economic and industry conditions including a global economic slowdown, geopolitical events, changes in laws or accounting rules, fluctuations in interest and exchange rates, terrorism, international conflicts, major health concerns, natural disasters or other disruptions of expected economic and business conditions. Additional risks and uncertainties not currently known to us or that we currently believe are immaterial also may impair our business operations and liquidity.
This section should be read in conjunction with Part II, Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the consolidated financial statements and accompanying notes in Part II, Item 8, Financial Statements, of this Form 10-K.
Risks Related to Strategic Alternative Process, Potential Strategic Transaction, and Financial Position
We have decided to seek a strategic acquisition, business combination, partnership or other change of control transaction, and there is no guarantee that this strategic path will be successful.
We have engaged MTS Health Partners, L.P. (“MTS”) as our advisor to assist with the exploration of strategic alternatives. MTS is providing a range of advisory services aimed to enhance stockholder value. The alternatives to be considered may include, but are not limited to, the sale of all or substantially all of our assets; a strategic merger or other business combination transaction; or another change of control transaction between us and a third party. We have and expect to continue to devote substantial time and resources to exploring such strategic alternatives; however, there is no set time frame and there can be no assurance that such activities will result in any agreements or transactions that will enhance stockholder value. Evaluating a strategic alternative may divert the attention of our management from ordinary operating matters. The identification, negotiation, and completion of any such transaction may also require more time and cash resources than we anticipate. In addition, potential strategic alternatives that require stockholder approval may not be approved by our stockholders.
There can be no assurance that our process to identify and evaluate potential strategic alternatives will result in any definitive offer to consummate a strategic transaction, or if made that the terms thereof will be acceptable to us. If any definitive offer to consummate a strategic transaction is received, there can be no assurance that a definitive agreement will be executed or that, if a definitive agreement is executed, the transaction will be consummated. In addition, there can be no assurance that any transaction, involving our company and/or assets, that is consummated would enhance stockholder value.
If we are successful in completing a strategic transaction, we may be exposed to other operational and financial risks, including increased near-term or long-term expenditures, exposure to unknown liabilities, incurrence of substantial debt, higher-than-expected acquisition and integration costs, write-downs of assets or impairment charges, increased amortization expenses, difficulty and cost in combining the operations and personnel of any merged businesses with our operations and personnel, impairment of relationships with key suppliers or customers of any acquired businesses due to changes in management and ownership, and inability to retain key employees of our company or any acquired businesses. If a strategic alternative is not completed, we may be subject to a number of material risks or suffer a number of consequences that may adversely affect our business, financial results, and operations.
If we are unable to successfully complete a strategic transaction, we may be forced to cease operations altogether or file for bankruptcy protection.
There can be no assurance that any of our plans will be successful or that additional capital will be available to us on reasonable terms, or at all, when needed or that we will successfully complete a strategic transaction. If we do not maintain governmental approval for our products, or if we are unsuccessful in our commercial efforts to sell our products, our business would experience significant harm. If we are unable to obtain sufficient additional
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 54
capital or to conclude a successful strategic transaction, we may be forced to defer, reduce or eliminate significant planned expenditures, restructure, curtail, or eliminate some or all of our commercial operations, dispose of technology or assets including intangible assets, conclude a strategic transaction that is unfavorable to stockholders, enter into arrangements that may require us to relinquish rights to certain of our products or product candidates, technologies or potential markets, delay or stop ongoing clinical trials, cease operations altogether or file for bankruptcy protection.
Our board of directors remains dedicated to diligently deliberating upon and making informed decisions that the directors believe are in the best interests of the company and its stockholders. There can be no assurance, however, that the company’s current strategic direction, or the board’s evaluation of strategic alternatives, will result in any initiatives, agreements, transactions or plans that will further enhance stockholder value.
In addition, given the substantial restructuring of our operations and reduction in force over the last year, it may be difficult to evaluate our current business and future prospects on the basis of historical operating performance.
We may not realize any additional value in a strategic transaction.
Potential counterparties in a strategic transaction involving our company may place minimal or no value on our assets. Further, the continued commercialization of our existing products, the development and any potential commercialization of our product candidates will require substantial additional cash to fund the costs associated with conducting the necessary preclinical and clinical testing and obtaining regulatory approval. Consequently, any potential counterparty in a strategic transaction involving our company may choose not to spend additional resources and continue to commercialize our existing products, development of our product candidates and may attribute little or no value, in such a transaction, to those product candidates.
If we are successful in completing a strategic transaction, we may be exposed to other operational and financial risks.
Although there can be no assurance that a strategic transaction will result from the process we have undertaken to identify and evaluate strategic alternatives, the negotiation and consummation of any such transaction will require significant time on the part of our management, and the diversion of management’s attention may disrupt our business. The negotiation and consummation of any such transaction may also require more time or greater cash resources than we anticipate and expose us to other operational and financial risks, including:
• inability to retain key employees of our company or any merged business;
•increased near-term and long-term expenditures;
• exposure to unknown liabilities;
• higher than expected acquisition or integration costs;
• incurrence of substantial debt or dilutive issuances of equity securities to fund future operations;
• write-downs of assets or incurrence of non-recurring, impairment or other charges;
• increased amortization expenses;
• difficulty and cost in combining the operations and personnel of any acquired business with our operations and personnel;
• impairment of relationships with key suppliers or customers of any acquired business due to changes in management and ownership; and
• possibility of future litigation.
Any of the foregoing risks could have a material adverse effect on our business, financial condition and prospects.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 55
We may not fully realize the expected cost savings and/or operating efficiencies from our restructuring activities and our ability to consummate a strategic transaction depends on our ability to retain our employees required to consummate such transaction.
On July 25, 2022 and November 14, 2022, we restructured our operations to narrow our near-term business focus and reduce our workforce by approximately 9% and 22% of our then headcount, respectively, primarily due to the ongoing decline in the macroeconomic environment. Our restructuring included external and internal cost reductions in almost all areas of our business. We focused and will continue to focus cost reductions on pipeline candidates, discovery programs, business development, and our dual platform in order to prioritize certain commercial efforts at that time. The reductions in workforce decreased overall headcount by a total of approximately 84 employees. Our headcount has reduced from approximately 300 as of June 30, 2022 to approximately 200 as of December 31, 2022.
We believe these changes were needed to streamline our organization and reallocate our resources to better align with our current strategic goals, including our current focus on pursuing strategic alternatives. However, these expense reduction measures have and may continue to yield unintended consequences and costs, such as the loss of institutional knowledge and expertise, attrition beyond our intended reductions in workforce, a reduction in morale among our remaining employees, and the risk that we may not achieve the anticipated benefits, all of which may have an adverse effect on our results of operations or financial condition. See Note 15, “Restructuring Charges” in the accompanying notes to the consolidated financial statements included in Part II, Item 8, of this Form 10-K and the “Restructuring and Reductions in Workforce” under Recent Events within Part II Item 7 for further discussion of our current restructuring activities and future anticipated cost savings.
Our ability to consummate a strategic transaction depends upon our ability to retain our employees required to consummate such a transaction, the loss of whose services may adversely impact the ability to consummate such transaction. In February 2023, Pear engaged an investment bank to explore the potential for an acquisition, company sale, merger, divestiture of assets, licensing, or other strategic transactions and/or seeking additional financing. There is no set timetable for this process and there can be no assurance that this process will result in the Company pursuing a transaction or that any transaction, if pursued, will be completed on attractive terms. If the Company is unable to complete a transaction, it may be required to seek a reorganization, liquidation, or other restructuring. In addition, our cash conservation activities, as well as the announcement that we are seeking strategic alternatives, may yield unintended consequences, such as attrition beyond our planned reductions in workforce that took place during 2022 and reduced employee morale, which may cause remaining employees to seek alternative employment. Two officers departed the Company, Kathy Jeffery, our Chief People Officer, departed on February 17, 2023, and Julia Strandberg, our Chief Commercial Officer, departed on March 10, 2023 to pursue other opportunities. In addition, our Vice President, Market Affairs, departed the Company in March 2023 to pursue other opportunities. In addition, four vice presidents will be departing the Company in April 2023. Our ability to successfully complete a strategic transaction depends in large part on our ability to retain certain of our remaining personnel. If we are unable to successfully retain our remaining personnel, we are at risk of a disruption to our exploration and consummation of a strategic alternative as well as business operations.
We may become involved in securities litigation that could divert management’s attention and harm the company’s business, and insurance coverage may not be sufficient to cover all costs and damages.
In the past, securities litigation has often followed certain significant business transactions, such as the sale of a company or announcement of any other strategic transaction. We may be exposed to such litigation even if no wrongdoing occurred. Litigation is usually expensive and diverts management’s attention and resources, which could adversely affect our business and cash resources and our ability to consummate a potential strategic transaction or the ultimate value our stockholders receive in any such transaction.
We have identified conditions and events that raise substantial doubt about our ability to continue as a going concern, we need substantial additional funding, and if we are unable to raise capital when needed or on terms favorable to us, our business, financial condition, and results of operation could be materially and adversely affected.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 56
We may be forced to wind-down our operations if we are unable to consummate a strategic transaction and/or obtain sufficient funding.
As of December 31, 2022, we had cash and cash equivalents totaling $48.3 million, and there is substantial doubt about our ability to continue as a growing concern. Based on our current operating plans, we do not have sufficient cash and cash equivalents to fund our operating expenses and capital expenditures for at least the next 12 months from the filing date of this Annual Report on Form 10-K.
In addition, we regularly maintain cash balances at third-party financial institutions in excess of the Federal Deposit Insurance Corporation insurance limit of $250,000, such as amounts to secure the letter of credit related to the lease of our San Francisco office in the amount of $0.4 million. On March 10, 2023, the issuer of the landlord’s letter of credit, Silicon Valley Bank, was placed into receivership with the FDIC. Since that date the obligations of Silicon Valley Bank have been assumed by First-Citizens Bank & Trust Company. The landlord has demanded a replacement letter of credit by no later than April 21, 2023. While we monitor the cash balances in our operating accounts on a daily basis and adjust the balances as appropriate, these balances could also be impacted from time to time. While the cash and cash equivalents are spread across various third party financial institutions, they are generally accessed through one main financial institution. Should that financial institution cease to exist, or is subject to other adverse conditions in the financial or credit markets there could be a material adverse effect on our business if we are not able to immediately access the funds in other third-party financial institutions. We can provide no assurance that access to our invested cash and cash equivalents will not be impacted by adverse conditions in the financial and credit markets.
In addition, our estimates of cash and cash equivalents needed to fund operation may prove to be wrong, and we could use our available capital resources sooner than expected. We require additional capital to sustain our operations, including our development programs.
On January 3, 2023, we entered into an At The Market Offering Agreement (the “ATM Agreement”) with H.C. Wainwright & Co., LLC (“Wainwright”) and Virtu Americas LLC (“Virtu” and, collectively with Wainwright, the “Managers” and each, a “Manager”), pursuant to which the Company may offer and sell, from time to time through the Managers, shares of the Company’s Class A common stock, par value $0.0001 per share (“Common Stock”), for aggregate gross proceeds of up to $150 million (the “Shares”). To date, we have sold an aggregate of 843,281 shares of Class A common stock pursuant to the ATM Agreement resulting in net proceeds of approximately $1.0 million. Our ability to raise additional funds under the ATM Agreement is subject to market conditions and other factors that may be outside of our control.
In February 2023 we began working with MTS Health Partners, L.P. (“MTS”), an investment bank, to assist with the exploration of strategic alternatives that may include, but are not limited to, the sale of all or substantially all of our assets; a strategic merger or other business combination transaction; or another change of control transaction between us and a third party.
We may seek additional funds through equity or debt financings or through collaborations, licensing transactions or other sources that may be identified through our strategic process. However, there can be no assurance that we will be able to complete any such transactions on acceptable terms or otherwise. The failure to obtain sufficient funds on commercially acceptable terms when needed would have a material adverse effect on our business, results of operations, and financial condition. These factors raise substantial doubt about our ability to continue as a going concern.
We do not currently have any commitments for future funding or additional capital. As such, we have paused or significantly scaled back the development or commercialization of our future product candidates or other research and development initiatives and our commercial activities are not being sufficiently funded. If we are unable to complete a strategic transaction or raise additional capital in sufficient amounts, we will not be able to continue our business and we may need to file for bankruptcy protection.
We have incurred net losses in every year since our inception and anticipate that we will continue to incur net losses for the foreseeable future.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 57
We are not profitable and have incurred significant net losses since our inception. We incurred net losses of $75.5 million and $65.1 million for the years ended December 31, 2022 and 2021, respectively. As of December 31, 2022, we had an accumulated deficit of $323.5 million. If we are able to continue as a going concern, which would require a transaction such as a strategic alternative, we expect to continue to incur significant losses for the foreseeable future. We expect to incur significant losses and negative cash flow from operations for the foreseeable future. We face a variety of challenges and risks that we will need to address and manage as we pursue our strategy, including our ability to achieve adequate payor coverage, develop and retain an effective sales force, and more broadly our workforce, achieve market acceptance of PDTs among physicians, patients and third-party payors, and in the future when adequate capital is available, to expand the use of PDTs to additional therapeutic indications. Because of the numerous risks and uncertainties associated with our commercialization efforts to increase adoption of our FDA approval we are unable to predict the timing or amount of increased expenses, or when, if ever, we will be able to achieve or maintain profitability. We expect to continue to incur substantial net losses and negative cash flows from operations as we commercialize our three existing products. We intend to continue to make targeted investments in building our US commercial infrastructure.
Based on our recurring losses and expectations to incur significant expenses and negative cash flow for the foreseeable future, our independent registered public accounting firm has included an explanatory paragraph in its report on our financial statements as of and for the years ended December 31, 2022 and December 31, 2021, expressing substantial doubt about our ability to continue as a going concern.
Further, we may incur significant costs to conduct planned clinical trials and future clinical trials. These clinical trials may be more costly than we expect, and if we do not achieve the benefits anticipated from these clinical trials, or if the realization of these benefits is delayed, they may not result in increased revenue or growth in our business. We also expect our operating costs to increase as a result of becoming a public company and will continue to increase as we grow our business. These efforts may prove more expensive than we currently anticipate, and our expenses may exceed revenues for the foreseeable future and we may not achieve profitability.
Additionally, inflation has the potential to adversely affect our business, financial condition and results of operations by increasing our overall costs. The existence of inflation in the economy has resulted in, and may continue to result in, higher costs, supply shortages, increased costs of labor and other similar effects. As a result of inflation, we have experienced and may continue to experience, cost increases. Although we may take measures to mitigate the impact of inflation, if these measures are not effective our business, financial condition and results of operations could be adversely affected.
To date, we have financed our operations principally from the closing of the Business Combination with Thimble Point Acquisition Corp. (“THMA”), the sale of Legacy Pear convertible preferred stock, payments received in connection with collaboration agreements, and proceeds from borrowings under a credit facility. Historically the revenue from product sales and collaboration agreements have not covered the full cost of our operations. Our cash flow from operations was negative for the years ended December 31, 2022 and 2021. We may not generate positive cash flow from operations or achieve profitability for the foreseeable future. Our limited operating history may make it difficult for you to evaluate our current business and future prospects. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis.
Given our limited operating history, our ability to achieve revenues sufficient to cover our operating costs may not be achieved. If we are not able to scale and grow the business to achieve significant product sales, it would materially and adversely affect our business, financial condition, and results of operations. Our failure to achieve or maintain profitability would negatively impact the value of our common stock.
Our credit agreement with Perceptive Credit Holdings III, LP restricts our current and future operations, particularly our ability to respond to changes or to take certain actions.
Our secured Amended and Restated Credit Agreement and Guaranty with Perceptive Credit Holdings III, LP, as lender (the “Perceptive Credit Facility”) is collateralized by substantially all of our assets, including our intellectual property, and imposes significant operating and financial restrictions and limit our ability and our other restricted subsidiaries’ ability to, among other things:
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 58
•incur additional indebtedness for borrowed money and guarantee indebtedness;
•pay dividends or make other distributions in respect of, or repurchase or redeem, capital stock;
•make loans, guarantees, investments and acquisitions;
•sell or otherwise dispose of assets;
•incur liens;
•enter into transactions with affiliates;
•enter into agreements restricting our subsidiaries’ ability to pay dividends;
•consolidate, merge or incur a change of control; and
•incur a material adverse change in our business condition (financial or otherwise), operations, performance or property.
As a result of these covenants and restrictions, we are and will be limited in how we conduct our business, and we may be unable to raise additional debt or equity financing to compete effectively or to take advantage of new business opportunities. In addition, the Perceptive Credit Facility requires us to comply with a minimum consolidated revenue covenant (measured on a trailing twelve-month basis) and maintain a minimum aggregate cash balance of $5.0 million in one or more accounts pledged to our lenders. The operating and financial restrictions and covenants in the Perceptive Credit Facility, as well as any future financing agreements that we may enter into, may restrict our ability to finance our operations, engage in business activities, or expand or fully pursue our business strategies. Our ability to comply with these covenants may be affected by events beyond our control, and we may not be able to meet those covenants. On March 25, 2022, we amended the Perceptive Credit Facility to adjust certain covenants under the agreement. The amendment included reducing the required minimum trailing 12-month revenue for the fiscal quarter ending March 31, 2022, through the fiscal quarter ending March 31, 2025. In the past we have received waivers with respect to certain financial covenants in our credit agreement.
We were not in compliance with the trailing twelve month revenue covenants as of December 31, 2022, and will not be in compliance with the trailing twelve month revenue covenants for the quarter ended March 31, 2023. However we requested and were granted a waiver on the trailing 12-month revenue requirement for both the quarter ended December 31, 2022 and March 31, 2023, respectively. In addition, on February 28, 2023, the Company also obtained a waiver pertaining to the existence of a “going concern” qualification in the accompanying opinion of the Company’s auditors in this Form 10-K and any resulting event of default.
We cannot guarantee that we will be able to maintain compliance with the covenants under the Perceptive Credit Facility in the future and, if we fail to do so, that we will be able to obtain waivers from the lenders or amend the covenants.
Our failure to comply with the restrictive covenants described above could, and our interest payment default will, result in an event of default under the credit agreement. An event of default will also occur if, among other things, a material adverse change in our business, operations or condition occurs, which could potentially include a material impairment of the prospect of our repayment of any portion of the amounts we owe under the credit agreement. In the case of a continuing event of default under the credit agreement, the lenders could elect to declare all amounts outstanding to be immediately due and payable, proceed against the collateral in which we granted the lenders a security interest under the credit agreement, or otherwise exercise the rights of a secured creditor. If we are forced to refinance these borrowings on less favorable terms, our business, results of operations and financial condition could be adversely affected and If we are unable to obtain sufficient additional capital or to conclude a successful strategic transaction, we may be forced to defer, reduce or eliminate significant planned expenditures, restructure, curtail, or eliminate some or all of our commercial operations, dispose of technology or assets, conclude a strategic transaction that is unfavorable to stockholders, enter into arrangements that may
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 59
require us to relinquish rights to certain of our product candidates, technologies or potential markets, delay or stop ongoing clinical trials, cease operations altogether or file for bankruptcy protection. See Note 7, Indebtedness, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for additional information regarding our Perceptive Credit Facility.
Perceptive claims that certain events of default have occurred under the Perceptive Credit Facility. Upon the occurrence of an Event of Default, Perceptive may declare the full amount outstanding under the Perceptive Credit Agreement due and payable, which would materially and adversely affect our ability to continue operations.
Perceptive has alleged that certain events of default have occurred and are continuing under our secured Perceptive Credit Facility. Perceptive has not delivered any formal notice of an Event of Default. We dispute the allegations and are in discussions with Perceptive to resolve this dispute and otherwise to address our obligations under the Perceptive Credit Facility.
There can be no assurances that our discussions with Perceptive will result in any resolution or that we will be able to resolve the alleged events of default or that any resolution, or the lack of any resolution, will not result in Perceptive exercising remedies under the Perceptive Credit Facility. In the case of a continuing Event of Default under the Perceptive Credit Facility, Perceptive could elect to declare all amounts outstanding to be immediately due and payable, proceed against the collateral in which we granted the lenders a security interest, or otherwise exercise the rights of a secured creditor. There can be no assurances that Perceptive will not declare a Default and attempt to accelerate the payment of all amounts due thereunder. If the acceleration were to occur, we could also be required to pay a prepayment penalty of $3.6 million.
The amount of our future losses is uncertain and our quarterly and annual operating results may fluctuate significantly or fall below the expectations of investors or securities analysts, each of which may cause our stock price to fluctuate or decline.
Our quarterly and annual operating results may fluctuate significantly in the future due to a variety of factors, many of which are outside of our control and may be difficult to predict, including the following:
•our ability to consummate a strategic alternative;
•an event of default under our Perceptive Credit Facility, which Perceptive has alleged currently exists;
•any change in the competitive landscape of our industry, including consolidation among our competitors or partners or as a result of COVID-19;
•our ability to attract, hire and retain qualified personnel;
• the level of demand for our products from patients, health care providers and payors;
•the risk/benefit profile, cost and reimbursement policies with respect to our products;
•the changing and volatile US and global economic environments; and
•future accounting pronouncements or changes in our accounting policies.
The cumulative effects of these factors could result in large fluctuations and unpredictability in our quarterly and annual operating results. As a result, comparing our operating results on a period-to-period basis may not be meaningful. This variability and unpredictability could also result in our failing to meet the expectations of industry or financial analysts or investors for any period. If our operating results or revenue fall below the expectations of analysts or investors or below any forecasts we may provide to the market, or if the forecasts we provide to the market are below the expectations of analysts or investors, the price of our common stock could decline substantially. Such a stock price decline could occur even when we have met any previously publicly stated guidance we may provide.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 60
Risks Related to Our Business and Industry
The failure of our prescription digital therapeutics to achieve and maintain market acceptance and adoption by patients and physicians would cause our business, financial condition and results of operations to be materially and adversely affected.
Our current business strategy is highly dependent on our prescription digital therapeutics, or PDTs, achieving and maintaining broad market acceptance by patients and physicians. Market acceptance and adoption of our PDTs depends on educating people with chronic conditions, as well as self-insured employers, commercial and government payors, health plans and physicians and other government entities, as to the distinct features, therapeutic benefits, cost savings, and other advantages of our PDTs as compared to competitive products or other currently available methodologies. If we are not successful in demonstrating to existing or potential patients and prescribers the benefits of our products, or if we are not able to achieve the support of patients, healthcare providers and payors for our products, our sales may decline or we may fail to increase our sales in line with our forecasts.
Achieving and maintaining market acceptance of our products could be negatively impacted by many factors, including:
•the failure of reSET, reSET-O and Somryst to achieve wide acceptance among people with substance use disorder, opioid use disorder and chronic insomnia, self-insured employers, commercial and government payors, health plans, physicians and other government entities, and key opinion leaders in the treatment community;
•lack of additional evidence or peer-reviewed publication of clinical or real world evidence supporting the effectiveness, safety, cost-savings or other advantages of our products over competitive products or other currently available methodologies;
•perceived risks associated with the use of our products or similar products or technologies generally;
•our ability to secure and maintain FDA and other regulatory clearance, authorization or approval for our products;
•the introduction of competitive products and the rate of acceptance of those products as compared to our products; and
•results of clinical, real world and health economics and outcomes research (“HEOR”)HEOR studies relating to chronic condition products or similar competitive products.
In addition, our products may be perceived by patients and healthcare providers to be more complicated or less effective than traditional approaches, and people may be unwilling to change their current health regimens.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 53
Moreover, we believe that healthcare providers tend to be slow to change their medical treatment practices because of perceived liability risks arising from the use of new products and the uncertainty of third-party reimbursement. Accordingly, healthcare providers may not recommend our products until there is sufficient evidence to convince them to alter their current approach.
The insurance coverage and reimbursement status of novel products, such as prescription digital therapeutics, is uncertain and only a limited number of healthcare insurers have agreed to reimburse purchases of our products. Failure to obtain or maintain adequate coverage and reimbursement for our products would substantially impair our ability to generate revenue.
In the US, patients generally rely on third-party payors to reimburse all or part of the costs associated with their treatment. Adequate coverage and reimbursement from governmental healthcare programs, such as Medicare and Medicaid, and commercial payors is critical to the ability of patients to afford treatments and achieve new product acceptance. Our ability to successfully commercialize our products will depend in part on the extent to which coverage and adequate Currently, there is no Medicare benefit category for prescriptions digital therapeutics, and it is unlikely that there will be such a benefit category unless and until Congress passes federal legislation. Our ability to successfully commercialize our products will depend in part on the extent to which coverage and adequate
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 61
reimbursement for these products and related treatments will be available from government health administration authorities, private health insurers and other organizations. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels. The availability of coverage and extent of reimbursement by governmental and private payors is essential for most patients to be able to afford treatments. Sales of products, and of product candidates that we may identify, will depend substantially on the extent to which the costs to users of such products will be paid by health maintenance, managed care, pharmacy benefit and similar healthcare management organizations, or reimbursed by government health administration authorities, private health coverage insurers and other third-party payors. If coverage and adequate reimbursement is not available, or is available only to limited levels, we may not be able to successfully commercialize our products. Even if coverage is provided, the approved reimbursement amount may not be high enough to allow us to establish or maintain pricing sufficient to achieve profitability.
There is also significant uncertainty related to, and there may be significant delays in obtaining, the insurance coverage and reimbursement of newly cleared, authorized, or approved products and coverage may be more limited than the purposes for which the device is cleared, authorized, or approved by the FDA or comparable foreign regulatory authorities. In the US, the principal decisions about reimbursement for new medicines or medical devices are typically made by the Centers for Medicare & Medicaid Services (“CMSCMS”), an agency within the US. Department of Health and Human Services (“HHSHHS”). FDA clearance or authorization provides no assurance of coverage or reimbursement by any payor. CMS decides whether and to what extent a new medicine or medical device will be covered and reimbursed under Medicare, and private payors tend to follow CMS to a substantial degree.
Factors payors consider in determining reimbursement are based on whether the product is:
•a covered benefit under its health plan;
•safe, effective and medically necessary;
•supported by robust clinical data from well-controlled clinical research;
•appropriate for the specific patient;
•cost-effective; and
•neither experimental nor investigational.
Each payor determines whether or not it will provide coverage for a treatment, what amount it will pay the manufacturer for the treatment and on what tier of its formulary the treatment will be placed. The position of a treatment on a payor’s list of covered drugs, biological products, and medical devices, or formulary, generally determines the co-payment that a patient will need to make to obtain the treatment and can strongly influence the adoption of such treatment by patients and physicians. Patients who are prescribed treatments for their conditions and providers prescribing such services generally rely on third-party payors to reimburse all or part of
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 54
the associated healthcare costs. Patients are unlikely to use our products unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of our products.
Moreover, eligibility for coverage and reimbursement does not imply that a productour products will be paid for in all cases or at a rate that covers our costs, including research, development, intellectual property, manufacturemanufacturing, marketing, salesales and distribution expenses. Interim reimbursement levels for new products, if applicable, may also not be sufficient to cover our costs and may not be made permanent. Reimbursement rates may vary according to the use of the productour products and the clinical setting in which itthey isare used, may be based on reimbursement levels already set for lower cost products and may be incorporated into existing payments for other services. Net prices for products may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors, by any future laws limiting prices and by any future relaxation of laws that presently restrict imports of products from countries where they may be sold at lower prices than in the US.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 62
Third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular drugs or devices. We cannot be sure that coverage and reimbursement will be available for all products that we commercialize and, if reimbursement is available, what the level of reimbursement will be. Inadequate coverage and reimbursement may impact the demand for, or the price of, any product for which we obtain marketing approval. If coverage and adequate reimbursement are not available, or are available only at limited levels, we may not be able to successfully commercialize our products.
In addition, in some foreign countries, the proposed pricing for a prescription device must be approved before it may be lawfully marketed. The requirements governing medical product pricing vary widely from country to country. For example, the European Union provides options for its Member States to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials that compare the cost effectiveness of a particular product candidate to currently available therapies. A Member State may approve a specific price for the medicinal products or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceuticals or medical devices will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the European Union do not follow price structures of the US and generally prices tend to be significantly lower. While we are not currently marketing or selling our products in any country other than the US, including the European Union or any of its Member States, in the event that we choose to do so in the future, we will need to comply with such requirements.
The market for prescription digital therapeutics is new, rapidly evolving, and increasingly competitive, as the healthcare industry in the US is undergoing significant structural change, which makes it difficult to forecast demand for our products.
The market for our PDTs is new and rapidly evolving, and it is uncertain whether it will achieve and sustain high levels of demand and market adoption. Our future financial performance will depend on growth in this market and on our ability to adapt to emerging demands of our customers. It is difficult to predict the future growth rate and size of our target market. The healthcare industry in the US is undergoing significant structural change and is rapidly evolving. We believe demand for our products has been driven in large part by rapidly growing costs in the traditional healthcare system, the movement toward patient-centricity and personalized healthcare, and advances in technology. Widespread acceptance of personalized healthcare is critical to our future growth and success. A reduction in the growth of personalized healthcare could reduce the demand for our PDTs and result in a lower revenue growth rate or decreased revenue.
If our assumptions regarding these uncertainties are incorrect or change in reaction to changes in our markets, or if we do not manage or address these risks successfully, our results of operations could differ materially from our expectations, and our business could suffer.
Our productproducts and product candidates representare novel and innovative potential therapeutic areas, and negative perception of any productof our products or product candidate that we develop could adversely affect our ability to conduct our
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 55
business, obtain marketing authorizations or identify alternate regulatory pathways to market for such product candidatecandidates could adversely affect our ability to conduct our business, obtain marketing authorizations or identify alternate regulatory pathways to market for such product candidates.
Our productproducts and product candidates are considered relatively new and novel therapeutic approaches. Our success will depend upon physicians who specialize in the treatment of diseases targeted by our products and product candidates prescribing potential treatments that involve the use of our products and product candidates in lieu of, or in addition to, existing treatments with which they are more familiar and for which greater clinical data may be available. Access will also depend on consumer acceptance and adoption of products that are commercialized. In addition, responses by the US, state or foreign governments to negative public perception or ethical concerns may result in new legislation or regulations that could limit our ability to develop or commercialize any product candidates, obtain or maintain marketing authorization, identify alternate regulatory pathways to market or otherwise achieve profitability.
Negative publicity concerning our products or the PDT market as a whole, could limit market acceptance of our products and product candidates. If patients and healthcare providers have a negative perception of PDTs, then a market for our products and product candidates may not develop at all, or it may develop more slowly than we expect. Our success will depend to a substantial extent on the willingness of healthcare providers to prescribe our products, the extent to which coverage and adequate reimbursement for these products and product candidates and related treatments will be available from government health administration authorities, private health insurers and other organizations and our ability to demonstrate the value of our products and product candidates to existing and potential patients and prescribers. Similarly, negative publicity regarding patient confidentiality and privacy in the context of technology-enabled healthcare or concerns experienced by our competitors could limit market acceptance of PDTs.
Our future depends on the continued contributions of our senior management team and our ability to attract and retain other highly qualified personnel; in particular, Corey McCann, our President and Chief Executive Officer, and Christopher Guiffre, our Chief Financial Officer and Chief Operating Officer, are critical to our future vision and strategic direction.
Our success depends in large part on our ability to attract and retain high-quality management in sales, market access, product development, software engineering, marketing, operations, finance and support functions, especially in the Boston area and the San Francisco Bay area. We compete for qualified technical personnel with other life sciences and information technology companies. . We compete for qualified technical personnel with other life sciences and information technology companies.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 63
Competition for qualified employees is intense in our industry, particularly for software engineers, and the loss of even a few qualified employees, or an inability to attract, train, retain and motivate additional highly skilled employees required for the planned expansion of our business could harm our operating results and impair our ability to grow. The loss of one or more of our key employees, such as Julia Strandberg, our Chief Commercial Officer, in March 2023, who left to pursue other opportunities, and any failure to have in place and execute an effective succession plan for key executives, could seriously harm our business. In addition, our Chief People Officer departed in February 2023 and one of our vice presidents departed the Company in March 2023 to pursue other opportunities. An additional four vice presidents will be departing the Company in April 2023.
As we continue to grow, weWe may be unable to continue to attract or retain the personnel we need to maintain our competitive position. To attract, train and retain key personnel, we use various measures, including competitive compensation and benefit packages (including an equity incentive program), which may require significant investment. These measures may not be enough to attract and retain the personnel we require to operate our business effectively and efficiently.
Moreover, ifdecreases in the value of our Class A common stock will impact the perceived value of ouremployee equity awards declines, itwhich may materially and adversely affect our ability to attract and retain key employees. If we do not maintain the necessary personnel to accomplish our business objectives, we may experience staffing constraints that materially and adversely affect our ability to support our programs and operations.
Many of our employees mayEmployees may also receive proceeds from sales of our equity in the public markets, which may reduce their motivation to continue to work for us.
In addition, our future also depends on the continued contributions of our senior management team and other key personnel, each of whom would be difficult to replace. In particular, Corey McCann, our President and Chief Executive Officer, and Christopher Guiffre, our Chief Financial Officer and Chief Operating Officer, are critical to our future vision and strategic direction. We rely on our executive team in the areas of operations, research and development, commercial, and general and administrative functions. Although we have entered into employment
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 56
agreements or offer letters with our key employees, these agreements have no specific duration and constituteOur key employees are at-will employment, and we do not maintain key person life insurance for some of our key employees.
In addition, from time to time, there may be changes in our senior management team that may be disruptive to our business, such as the departure of Julia Strandberg, our Chief Commercial Officer, in March 2023 who left to pursue other opportunities. If our senior management team, including any new hires that we may make, fails to work together effectively and to execute our plans and strategies on a timely basis, our business, results of operations and financial condition could be harmed.
AWe limited number of healthcare insurers have agreed to reimburse purchases of our products, and there is no assurance that additional healthcare insurers will agree to reimburse purchases of our products in the futurerely significantly upon Access Agreements from third-parties for the sale of our products and, if the opportunities for Access Agreements decline, such reliance could adversely affect our results.
While we anticipate reimbursement will become a more prominent portion of our overall revenue over time, we are currently reliant upon Access Agreements to generate revenue for the products we provide. Until we can consistently rely on the more conventional reimbursement pathways (e.g., those utilized by drug manufacturers), our revenue will be primarily driven by these Access Agreements. Generating revenue from Access Agreements has risk because they typically have a lengthy sales and contracting process. In addition, each Access Agreement represents a relatively large percentage of quarterly revenue and any failure or delay in the sales or contracting process for a single Access Agreement can have a disproportionate impact on the Pear’s revenue performance for the given period. Finally, Access Agreements tend to be funded by governmental resources which adds risk to completing any agreement in a timely manner.
To date, a limited number of healthcare insurersThese agreements have agreed to reimburse purchases of reSET, reSET-O, and Somryst. Wevarying terms and generally may depend upon revenue from sales of reSET, reSET-O, and Somryst,be revised or terminated for various reasons generally by Pear and in turn on reimbursement from third-party payors for such products. The amount that we receive in payment for our products may be materially and adversely affected by factors we do not control, including federal or state regulatory or legislative changes, and cost-containment decisions and changes in reimbursement schedules of third-party payors. Any reduction or elimination ofcertain circumstances by the Customer. Any failure to maintain existing Access Agreements or enter into these paymentsnew Access Agreements with less favorable terms than currently in place could have a material adverse effect on our business, prospects, results of operations and financial condition. In addition, there can be no assurance that we will be able to generate sufficient revenue from Access Agreements to become profitable.
Additionally, the reimbursement process is complex and can involve lengthy delays. AlsoSimilarly, our revenue could be materially and disproportionately impacted by the purchasing decisions of this limited customer base. In the future, third-party payors may reject, in whole or in part, requests for reimbursement based on determinations that certain amounts are not reimbursable under plan coverage, that services provided were not medically necessary, that additional supporting documentation is necessary, or for other reasons. Retroactive adjustments by third-party payors may be difficult or cost-prohibitive to appeal, and such changes could materially reduce the actual amount we receive. Delays and uncertainties in the reimbursement processour Access Agreement customers may decide to purchase less product from us than they have in the past, may be out of our control and mayalter purchasing patterns at any time with limited notice, or may decide not to
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 64
continue to purchase our products at all, any of which could cause our revenue to decline materially and adversely affect our business, prospects, results of operations andmaterially harm our financial condition.
IfThere wecan are unable to expand our marketing infrastructure, we may fail to increase the usage of our products and platform to meet our forecasts.
Following the termination of our commercialization agreement with Sandoz Inc. we became solely responsible for the marketing expenses relating to reSET and reSET-O in October 2019. As a result, we have only limited experience marketing our products and engaging customers at our current scale. Our financial condition and results of operations are and will continue to be highly dependent on the ability of our marketing function to adequately promote, market, and attract customers to our products and platform in a manner that complies with applicable laws and regulations and at a cost that does not exceed our current budget allocated to marketing.
If we are unable to expand our marketing capabilities, we may not be able to effectively expand the scope of our ability to attract new customers. Relatedly, if any of our advertising platforms significantly increase their advertising fees, our ability to expand our marketing reach will be greatly impeded. Any such failure could adversely affect our reputation, revenue, and results of operations.
Failure to adequately expand our direct sales force may impede our growth.
We believebe no assurance that state or federal entities will continue to provide grants to support these Access Agreements. Any discontinuance or reduction in government or private party grants could have a significant and adverse effect on these types of agreements, and as a result could have a material and adverse effect on our business, financial condition, or results of operations. Relatedly, states are beginning to receive proceeds from independent and multi-state settlement agreements with pharmaceutical companies that our future growth will dependwere involved in part on the continued developmentdistribution and sale of our direct sales force and its ability to obtain new customers and to manage our existing customers. Identifying and recruiting qualified personnel and training and managing a geographically dispersed sales team requires significant time, expense, and attention. It can take six months or longer before a new sales representative is fully trained and productive. Our business may be adversely affected if our efforts to expand and train our direct sales force do not generate a corresponding increase in revenue. In particular, if we are unable to hire, develop, and retain sufficient numbers of productive direct sales personnel or if new direct sales personnel are unable to achieve desired productivity levels in a reasonable period of time, sales of our services will suffer, and our growth will be impeded.
prescription opioids. Individual states have broad discretion for how these settlement funds may be used to prevent opioid abuse and how to distribute the funds. States are creating legislatively appointed bodies to oversee these funds (e.g., New York’s Opioid Settlement Fund, Tennessee’s Opioid Abatement Fund, Nebraska’s Opioid Recovery Fund, etc.). A state’s decision not to allocate settlement funds to Access Agreements involving our products, while allocating such funds to other opioid prevention efforts, could have a material and adverse effect on our business, financial condition, or results of operations.
Our products are made available via the Apple App Store and the Google Play Store and supported by third-party infrastructure. If our ability to access those markets or access necessary third-party infrastructure was stopped or otherwise restricted or limited, it would materially and adversely affect our business.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 57
, it would materially and adversely affect our business. Our PDTs are exclusively accessed through and depend on the Apple App Store and the Google Play Store. Both Apple and Google have broad discretion to make changes to their operating systems or payment services or change the manner in which their mobile operating systems function and their respective terms and conditions applicable to the distribution of our PDTs and to interpret their respective terms and conditions in ways that may limit, eliminate or otherwise interfere with our products, our ability to distribute our products through their stores, our ability to update our products, including to make bug fixes or other feature updates or upgrades, the features we provide, the manner in which we market our products and our ability to access native functionality or other aspects of mobile devices. To the extent either or both of them do so, our business, financial condition and results of operations would be materially and adversely affected.
There is no guarantee that the third-party infrastructure that currently support our PDTs will continue to support them or, if it does not, that other alternatives will be available. We will continue to be dependent on third-party mobile operating systems, technologies, networks and standards that we do not control, such as the Android and iOS operating systems, and any changes, bugs, technical or regulatory issues in such systems, our current relationships with carriers or future relationships with mobile manufacturers, or in their terms of service or policies that degrade our PDTs’ functionality, reduce or eliminate our ability to distribute our PDTs, limit our ability to deliver high quality PDTs, or impose fees or other charges related to delivering our offerings, could adversely affect our product usage and revenue.
We rely upon third party providers of cloud-based infrastructure to host our platform. Any disruption in the operations of these third-party providers, limitations on capacity or interference with our use could have a material adverse effect on our business, prospects, results of operations and financial condition.
Our platform’s technological infrastructure is implemented using third-party hosting services, such as Amazon Web Services. We have no control over any of these third parties, and we cannot guarantee that such third-party providers will not experience system interruptions, outages or delays, or deterioration in their performance. We need to be able to access our computational platform at any time, without interruption or degradation of performance. Our hosted platform depends on protecting the virtual cloud infrastructure hosted by third-party hosting services by maintaining our configuration, architecture, features, and interconnection specifications, as well as protecting the information stored in these virtual data centers, which is transmitted by third-party Internet service providers. We have experienced, and expect that in the future we may again experience interruptions, delays and outages in service and availability from time to time due to a variety of factors, including infrastructure changes, human or software errors, hosting disruptions and capacity constraints. Any limitation on the capacity of our third-party hosting services could adversely affect our business, financial condition, and results of operations. In addition, any incident affecting our third-party hosting services’ infrastructure, which may be caused by cyber-attacks, natural disasters, fire, flood, severe storm, earthquake, power loss, telecommunications failures, terrorist
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 65
or other attacks, and other disruptive events beyond our control, could negatively affect our cloud-based solutions. A prolonged service disruption affecting our cloud-based solutions could damage our reputation or otherwise harm our business. We may also incur significant costs for using alternative equipment or taking other actions in preparation for, or in reaction to, events that damage the third-party hosting services we use.
In the event that our service agreements with our third-party hosting services are terminated, or there is a lapse of service, elimination of services or features that we utilize, interruption of Internet service provider connectivity, or damage to such facilities, we could experience interruptions in access to our platform as well as significant delays and additional expense in arranging or creating new facilities and services and/or re-architecting our hosted software solutions for deployment on a different cloud infrastructure service provider, which could have a material adverse effect on our business, prospects, results of operations and financial condition.
We rely on a limited number of third party digital pharmacies for the fulfillment of prescriptions. This reliance on increases the risk that we could have a disruption in the fulfillment of prescriptions, which could have a material and adverse effect on our reputation, business, results of operations and financial condition.
We do not currently own or operate any pharmacy, nor are we licensed to perform pharmacy fulfillment services. We rely, and expect to continue to rely, on a limited number of third parties for the fulfillment of prescriptions.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 58
This reliance increases the risk that we could have a disruption in the fulfillment of prescriptions which could delay, preventThis reliance increases the risk that we could have a disruption in the fulfillment of prescriptions which could delay, prevent, or impair the distribution and sale of our products.
Pharmacies are subject to state and federal laws and regulations. We do not control the standards and processes of, and will be completely dependent on, our digital pharmacies for compliance with federal and state law and regulations. If our digital pharmacies fail to maintain regulatory compliance, we may need to find alternative pharmacies with the capability to fulfill prescriptions for PDTs. In addition, we have no control over the ability of our digital pharmacies to maintain adequate quality control, quality assurance, and qualified personnel. If a regulatory authority finds deficiencies with or withdraws required pharmacy licenses in the future, we may need to find alternative pharmacies with the capability to fulfill prescriptions for PDTs, which would significantly impact our ability to fulfill, distribute, and sell our products. We may be unable to establish any agreements with other digital pharmacies or to do so on acceptable terms. Even if we are able to establish agreements with other digital pharmacies, reliance on a limited number of digital pharmacies entails additional risks, including:
•the possible breach of the services agreement by the third party; and
•the possible termination or nonrenewal of the agreement by the third party at a time that is costly or inconvenient for us.
There are a limited number of digital pharmacies that have the capability to distribute PDTs and that might be capable of fulfilling prescriptions for our products.
If our current digital pharmacies cannot perform as agreed, we may be required to replace such digital pharmacies. We may incur added costs and delays in identifying and qualifying any such replacements. If the agreement with any of our third -party pharmacies is terminated, if any third -party pharmacy is unable to perform in accordance with the terms of the agreement, or if the services of any third -party pharmacy is terminated for any reason, it could have a material adverse effect on our business, prospects, results of operations, and financial condition.
Our current and anticipated future dependence upon others for the fulfillment of prescriptions for our product candidates or products may adversely affect our future profit margins and our ability to distribute any products on a timely and competitive basis.
We face significant competition and new products may emerge that provide different or better alternatives for treatment of the conditions that our products are authorized to treat. Many of our current and future competitors have or will have significantly more resources.
Our ability to achieve our strategic objectives will depend, among other things, on our ability to develop and commercialize products for the treatment of chronic conditions that are effective and safe, offer distinct features, are easy-to-use, provide measurable and meaningful cost savings to payors, and are more appealing than available alternatives. Our competitors, as well as a number of other companies, within and outside the healthcare industry,
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 66
are pursuing new delivery devices, delivery technologies, sensing technologies, procedures, drugs, and other therapies for the monitoring and treatment of chronic conditions. Any technological breakthroughs in monitoring, treatment or prevention could reduce the potential market for our products, which would significantly reduce our sales.
The introduction by competitors of products that are or claim to be superior to our products may create market confusion, which may make it difficult for potential customers to differentiate the benefits of our products over competitive products. In addition, the entry of new PDTs to the market which treat the same or similar chronic conditions to our products may lead some of our competitors to employ pricing strategies that could materially and adversely affect the pricing of our products. If a competitor develops a product that competes with or is perceived to be superior to our products, or if a competitor employs strategies that place downward pressure on pricing within our industry, our sales may decline significantly or may not increase in line with our forecasts, either of which would materially and adversely affect our business, financial condition and results of operations.
While our market is in an early stage of development, it is evolving rapidly and becoming increasingly competitive, and we expect it to attract increased competition. We currently face competition from a range of companies. Our competitors include both enterprise companies who are focused on or may enter the healthcare industry, including initiatives and partnerships launched by these large companies, and from private companies that offer
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 59
solutions for specific chronic conditions. We compete with pharmaceutical and biotechnology companies that are developing treatments for addiction and insomnia, including Alkermes and their product Vivitrol, Orexo and their product Zubsolv, Sandoz and their product Suboxone, Braeburn and their product Brixadi, Pfizer and their product Halcion, Merck and their product Belsomra, Sunovion and their product Lunesta and Sanofi and their product Ambien. In the digital health space we compete with companies that have created non-regulated products to treat addiction and insomnia such as Dynamicare, CBT4CBT and, Pzizz, Headspace, Calm, Orexo and their product Modia, and Big Health and their product Sleepio, Pzizz, Headspace,. These and Calm. Theseother companies, which may offer their solutions at lower prices, are continuing to develop additional products and becoming more sophisticated and effective. Competition from wellness apps, which are not authorized by the FDA but may attract consumers for other reasons, and from other parties will result in continued pricing pressures, which are likely to lead to price declines in certain product segments, which could negatively impact our sales, profitability and market share.
Our ability to compete effectively depends on our ability to distinguish our company and our solution from our competitors and their products, and includes factors such as:
•FDA authorization;
•effectiveness and safety;
•robust and well-controlled clinical research;
•long-term outcomes;
•ease of use and convenience;
•price;
•greater name and brand recognition;
•information security standards;
•greater market penetration;
•larger sales forces;
•larger marketing budgets;
•access to significantly greater financial, human, technical and other resources;
•breadth, depth, and effectiveness of offerings;
•FDA compliance, quality, and reliability of solutions; and
•healthcare provider, government agency and insurance carrier acceptance.
Some of our competitors may have, or new competitors or alliances may emerge that have, greater name and brand recognition, greater market share, a larger customer base, more widely adopted proprietary technologies, greater marketing expertise, larger sales forces, or significantly greater resources than we do and may be able to offer solutions competitive with ours at a more attractive price than we can. Further, our current or potential competitors may be acquired by third parties with greater available resources. As a result, our competitors may be able to respond more quickly and effectively than we can to new or changing opportunities, technologies, standards or customer requirements and may have the ability to initiate or withstand substantial price competition. In addition, our competitors may in the future establish cooperative relationships with vendors of complementary products, technologies or services to increase the availability of their solutions in the marketplace. Our competitors could also be better positioned to serve certain segments of our market, which could create additional price pressure. In light of these factors, even if our products are more effective than those of our competitors, current or potential customers may accept competitive products in lieu of purchasing our products. If
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 60
we are unable to successfully compete, our business, financial condition, and results of operations could be materially and adversely affected.
Any failure to offer high-we are unable to successfully compete, our business, financial condition, and results of operations could be materially and adversely affected.
A limited number of healthcare insurers have agreed to reimburse purchases of our products, and there is no assurance that additional healthcare insurers will agree to reimburse purchases of our products in the future.
To date, a limited number of healthcare insurers have agreed to reimburse purchases of reSET, reSET-O, and Somryst. We depend upon revenue from sales of reSET, reSET-O, and Somryst, and in turn on reimbursement from third-party payors for such products. The amount that we receive as payment for our products may be materially and adversely affected by factors cannot control, including federal or state regulatory or legislative changes, and
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 67
cost-containment decisions and changes in reimbursement schedules of third-party payors. Any reduction or elimination of these payments could have a material adverse effect on our business, prospects, results of operations and financial condition.
Additionally, the reimbursement process is complex and can involve lengthy delays. Also, third-party payors may reject, in whole or in part, requests for reimbursement based on determinations that certain amounts are not reimbursable under plan coverage, that services provided were not medically necessary, that additional supporting documentation is necessary, or for other reasons. Retroactive adjustments by third-party payors may be difficult or cost-prohibitive to appeal, and such changes could materially reduce the actual amount we receive. Delays and uncertainties in the reimbursement process may be out of our control and may materially and adversely affect our business, prospects, results of operations and financial condition.
If we are unable to expand our marketing infrastructure, we may fail to increase the usage of our products and platform to meet our forecasts.
We began commercializing our products in October 2019. As a result, we have limited experience marketing our products and engaging customers at our current scale. Our financial condition and results of operations are and will continue to be highly dependent on the ability of our marketing function to adequately promote, market, and attract customers to our products and platform in a manner that complies with applicable laws and regulations and at a cost that does not exceed our current budget allocated to marketing.
If we are unable to expand our marketing capabilities, which will be impacted by our available resources, we may not be able to effectively expand the scope of our ability to attract new customers. Relatedly, if any of our advertising platforms significantly increase their advertising fees, our ability to expand our marketing reach will be greatly impeded. Any such failure could adversely affect our reputation, revenue, and results of operations.
Failure to adequately expand our direct sales force may impede our growth.
We believe that our future growth will depend in part on the continued development of our direct sales force and its ability to obtain new customers and to manage our existing customers. Identifying and recruiting qualified personnel and training and managing a geographically dispersed sales team requires significant time, expense, and attention. In March 2023, our Chief Commercial Officer and VP, Market Access both resigned. It can take six months or longer before a new sales representative is fully trained and productive. Our business may be adversely affected if our efforts to expand and train our direct sales force do not generate a corresponding increase in revenue. In particular, if we are unable to hire, develop, and retain sufficient numbers of productive direct sales personnel or if new direct sales personnel are unable to achieve desired productivity levels in a reasonable period of time, sales of our services will suffer, and our growth will be impeded.
Any failure to offer high quality patient support may adversely affect our relationships with our existing and prospective patients, and in turn our business, results of operations and financial condition.
In implementing and using our products, our patients will depend on our patient support to resolve issues in a timely manner. We may be unable to respond quickly enough to accommodate short-term increases in demand for patient support. Increased patient demand for support could increase costs and adversely affect our results of operations and financial condition. Any failure to maintain high-quality patient support, or a market perception that we do not maintain high-quality patient support, could adversely affect patient satisfaction or the willingness of physicians to prescribe our products, and in turn our business, results of operations, and financial condition.
Acquisitions and strategic alliances could distract management and expose us to financial, execution and operational risks that could have a detrimental effect on our business.
We intend to continue to pursue acquisitions or licenses of technology to, among other things, expand the number of products we provide as well as the features within those products. We cannot guarantee that we will identify suitable candidates for acquisition or licensing, that the transactions will be completed on acceptable terms, or that we will be able to integrate newly acquired or licensed technology into our existing business. The acquisition and integration of another technology would divert management attention from other business activities, including our core business. This diversion, together with other difficulties we may incur in integrating newly acquired or licensed technology, could have a material adverse effect on our business, financial condition and results of operations. In addition, we may borrow money or issue capital stock to finance such transactions. Such borrowings might not be available on terms as favorable to us as our current borrowing terms and may increase our leverage, and the issuance of capital stock (or securities exchangeable therefore) could dilute the interests of our stockholders.
We have experienced rapid growth since inception which may not be indicative of our future growth and, if we continue to grow rapidly, we may not be able to manage our growth effectively.
Since launching reSET in 2019, we have experienced rapid growth and we continue to rapidly and significantly expand our operations. For example, our full-time employee headcount has grown from 9 employees as of December 31, 2018 to 299 employees as of December 31, 2021. This expansion increases the complexity of our business and places significant strain on our management, personnel, operations, systems, technical performance, financial resources, and internal financial control and reporting functions. We may not be able to manage growth effectively, which could damage our reputation, limit our growth, and negatively affect our operating results.
The growth and expansion of our business creates significant challenges for our management, operational, and financial resources. In the event of continued growth of our operations or in the number of our third-party relationships, our information technology systems, and our internal controls and procedures may not be adequate to support our operations. To effectively manage our growth, we must continue to improve our operational, financial, and management processes and systems and to effectively expand, train, and manage our employee base. As our organization continues to grow and we are required to implement more complex organizational management structures, we may find it increasingly difficult to maintain the benefits of our corporate culture, including our ability to quickly develop and launch new and innovative products. This could negatively affect our business performance.
We continue to experience growth in our headcount and operations, which will continue to place significant demands on our management and our operational and financial infrastructure. As we continue to grow, we must effectively integrate, develop and motivate a large number of new employees, and we must maintain the beneficial aspects of our corporate culture. To attract top talent, we have had to offer, and believe we will need to continue to offer, highly competitive compensation packages before we can validate the productivity of those employees. In addition, fluctuations in the price of our common stock may make it more difficult or costly to use equity compensation to motivate, incentivize and retain our employees. We face significant competition for talent from other healthcare, technology and high-growth companies, which include both large enterprises and privately-
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 61
held companies. We may not be able to hire new employees quickly enough to meet our needs. If we fail to effectively manage our hiring needs and successfully integrate our new hires, our efficiency and ability to meet our forecasts and our employee morale, productivity and retention could suffer, and our business, results of operations and financial condition could be materially and adversely affected.
If we cannot maintain our corporate culture, we could lose the innovation, collaborationIf we cannot maintain our corporate culture, we could lose the innovation, collaboration, and focus on the mission that contribute to our business.
We believe that our culture has been and will continue to be a critical contributor to our success. We expect to continue to hire aggressively as we expand, and we believe our corporate culture has been crucial in our success and our ability to attract highly skilled personnel. If we do not continue to develop our corporate culture or maintain and preserve our core values as we grow and evolve both in the US and internationally, we may be unable to foster the innovation, curiosity, creativity, focus on execution, teamwork and the facilitation of critical knowledge transfer and knowledge sharing we believe we need to support our growth. Moreover, liquidity available to our employee equityholders could lead to disparities of wealth among our employees, which could adversely impact relations among employees and our culture in general. Our anticipated headcount growth and our status as a public company may result in a change to our corporate culture, which could harm our business. Our status as a
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 68
public company may result in a change to our corporate culture, which could harm our business. Our reductions in force may yield unintended consequences and costs, such as attrition beyond the intended reduction in force, the distraction of employees and reduced employee morale, which could, in turn, adversely impact productivity, including through a loss of continuity, loss of accumulated knowledge and/or inefficiency during transitional periods. Any of these impacts could also adversely affect our reputation as an employer, make it more difficult for us to hire new employees in the future and increase the risk that we may not achieve the anticipated benefits from the restructuring.
The COVID-19 pandemic has hadBusiness or economic disruptions or and continues to have an adverse impact on our business, operations,global health concerns could seriously harm our business.
Broad-based business or economic disruptions could adversely affect our business and the markets and communities in which we operate.
Insale of our products. For example, in March 2020, the World Health Organization declared COVID-19 a global pandemic. This pandemic, whichoutbreak resulted in extended shutdowns of certain businesses throughout the world. While the COVID-19 pandemic has continued to spread, and the related adverse public health developments, including orders to shelter-in-place, travel restrictions, and mandated business closures, havenot materially adversely affected workforces, organizations, governments, customers, economiesour financial results and business operations through December 31, 2022, COVID-19 continues to present risks to the Company, and financial markets globally, leadingwe continue to an economic downturn and increased market volatility. It has also disrupted the normal operationsclosely monitor the impact of many businesses, including ours. This outbreak, as well as intensified measures undertaken to contain the spread ofthe pandemic on all aspects of our business. Global health concerns, such as the COVID-19 pandemic, could decreasecan impact healthcare industry spending for our products, adversely affect demand for our products, affect the ability of our sales team to travel to potential customers and the ability of our professional services teams to conduct in-person services and trainings, impact expected spending from new customers, negatively impact collections of accounts receivable, and harmwhich could have a material adverse effect on our business, and our results of operations, and financial condition.
Further, the sales cycle for a new customer of our products could lengthen, resulting in a potentially longer delay between increasing operating expenses and the generation of corresponding revenue, if any. We cannot predict with any certainty whether and to what degree the disruption caused by the COVID-19 pandemic and reactions thereto will continue and expect to face difficulty accurately predicting our internal financial forecasts. The pandemic also resulted in a shift in our workforce to a hybrid model in which employees in one of our three offices are working both remotely and onsite, and we anticipate we will continue to use this model going forward, which can increase the risk of a cybersecurity incident. In addition, our workforce has deep domain knowledge across a range of healthcare, technology, and general business, which was partially achieved by having certain of our employees working remotely across the US, which also increases the risk of a cyber security incident. Further, we continue to shift to assist new and existing customers who may also be working remotely or under hybrid models. It is not possible for us to predict the duration or magnitude of the adverse results of the outbreak and its effects on our business, results of operations, or financial condition at this timeoperation and financial condition.
Changes in funding or disruption at the FDA, the SEC and other government agencies caused by funding shortages or global health concerns could hinder their ability to hire and retain key leadership and other personnel, or otherwise prevent new or modified products from being developed, reviewed or commercialized in a timely manner or at all, or otherwise prevent those agencies from performing normal business functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA to review and grant marketing authorization for new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory and policy changes and other events that may otherwise affect the FDA’s ability to perform routine functions. Average review times at FDA have fluctuated in recent years as a result.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 62
In addition, government funding of the SEC and other government agencies on which our operations may rely, including those that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies, may also slow the time necessary for new digital therapeutics to be reviewed and/or granted marketing authorization by necessary government agencies, which would adversely affect our business. For example, in recent years, including for 35 days beginning on December 22, 2018, the US government shut down several times and certain regulatory agencies, such as the FDA and the SEC, had to furlough critical employees and stop critical activities.
If a prolonged government shutdown occurs, or if global health concerns continue to prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews or other regulatory activities, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Future government shutdowns or delays could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Risks Related to Our Financial Position
We have a history of significant losses, anticipate increasing expenses in the future, and may not be able to achieve or maintain profitability.
We have incurred significant net losses since our inception. We incurred net losses of $65.1 million and $97.0 million for the years ended December 31, 2021 and 2020, respectively. As of December 31, 2021, we had an accumulated deficit of $248.0 million. We expect to incur significant losses and negative cash flow from operations for the foreseeable future as we incur significant expenses in research and development, including developing our pipeline of product candidates, working to achieve and maintain market acceptance by physicians and patients, and incur significant sales and marketing costs, including expanding our marketing channels and operations as we grow our US commercial sales force and expand our marketing efforts to increase adoption of our FDA approved products as well as growing and enhancing our platform offering of products. Further, we will be making the necessary investments in our human capital to scale our business. Based on our recurring losses and expectations to incur significant expenses and negative cash flow for the foreseeable future, our independent registered public accounting firm has included an explanatory paragraph in its report on our financial statements as of and for the years ended December 31, 2021 and December 31, 2020 expressing substantial doubt about our ability to continue as a going concern.
Further, we will incur significant costs to conduct planned clinical trials and future clinical trials. These clinical trials may be more costly than we expect, and if we do not achieve the benefits anticipated from these clinical trials, or if the realization of these benefits is delayed, they may not result in increased revenue or growth in our business. We also expect our operating costs to increase as a result of becoming a public company and will continue to increase as we grow our business. These efforts may prove more expensive than we currently anticipate, and our expenses may exceed revenues for the foreseeable future and we may not achieve profitability.
To date, we have financed our operations principally from the closing of the Business Combination with THMA, the sale of Legacy Pear convertible preferred stock, payments received in connection with collaboration agreements, and proceeds from borrowings under a credit facility. Historically the revenue from product sales and collaboration agreements have not covered the full cost of our operations. Our cash flow from operations was negative for the years ended December 30, 2021 and 2020. We may not generate positive cash flow from operations or achieve profitability for the foreseeable future. Our limited operating history may make it difficult for you to evaluate our current business and future prospects. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis.
Given our limited operating history, our ability to achieve revenues sufficient to cover our operating costs may not be achieved. If we are not able to scale and grow the business to achieve significant product sales, it would materially and adversely affect our business, financial condition, and results of operations. Our failure to achieve or maintain profitability would negatively impact the value of our common stock.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 63
Our credit agreement with Perceptive restricts our current and future operations, particularly our ability to respond to changes or to take certain actions.
Our credit agreement with Perceptive is collateralized by substantially all of our assets, including our intellectual property, and imposes significant operating and financial restrictions and limit our ability and our other restricted subsidiaries’ ability to, among other things:
•incur additional indebtedness for borrowed money and guarantee indebtedness;
•pay dividends or make other distributions in respect of, or repurchase or redeem, capital stock;
•make loans, guarantees, investments and acquisitions;
•sell or otherwise dispose of assets;
•incur liens;
•enter into transactions with affiliates;
•enter into agreements restricting our subsidiaries’ ability to pay dividends;
•consolidate, merge or incur a change of control; and
•incur a material adverse change in our business condition (financial or otherwise), operations, performance or property.
As a result of these covenants and restrictions, we are and will be limited in how we conduct our business, and we may be unable to raise additional debt or equity financing to compete effectively or to take advantage of new business opportunities. In addition, our senior secured credit facility requires us to comply with a minimum consolidated revenue covenant (measured on a trailing twelve-month basis) and maintain a minimum aggregate cash balance of $5.0 million in one or more accounts pledged to our lenders. The operating and financial restrictions and covenants in the Perceptive Credit Facility, as well as any future financing agreements that we may enter into, may restrict our ability to finance our operations, engage in business activities, or expand or fully pursue our business strategies. Our ability to comply with these covenants may be affected by events beyond our control, and we may not be able to meet those covenants. For example, in the past we have received waivers with respect to certain financial covenants in our credit agreement. In addition, on March 25, 2022, we amended the Perceptive Credit Facility to adjust certain covenants under the agreement. The amendment included reducing the required minimum trailing 12-month revenue for the fiscal quarter ending March 31, 2022 through the fiscal quarter ending March 31, 2025. We cannot guarantee that we will be able to maintain compliance with these covenants in the future and, if we fail to do so, that we will be able to obtain waivers from the lenders or amend the covenants.
Our failure to comply with the restrictive covenants described above could result in an event of default under the credit agreement. An event of default will also occur if, among other things, a material adverse change in our business, operations or condition occurs, which could potentially include a material impairment of the prospect of our repayment of any portion of the amounts we owe under the credit agreement. In the case of a continuing event of default under the credit agreement, the lenders could elect to declare all amounts outstanding to be immediately due and payable, proceed against the collateral in which we granted the lenders a security interest under the credit agreement, or otherwise exercise the rights of a secured creditor. If we are forced to refinance these borrowings on less favorable terms, our business, results of operations and financial condition could be adversely affected. See Note 7 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for additional information regarding our Perceptive Credit Facility.
Due to the resources required for the development of our pipeline, and depending on our ability to access capital, we will have to prioritize the development of certain product candidates over others. We may fail to expend our limited resources on product candidates that may have been more profitable or for which there is a
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 64
greater likelihood of success, which would cause our business, financial condition, and results of operations to be materially and adversely affected.
We currently have three FDA-authorized or cleared products, reSET, reSET-O, and Somryst, as well as several other product candidates that are at various stages of development. We seek to maintain a process of prioritization and resource allocation to maintain an optimal balance between aggressively pursuing our more advanced clinical-stage product candidates and ensuring the development of new product candidates.
Due to the significant resources required for the development of our product candidates, we must decide which product candidates to pursue and advance and the amount of resources to allocate to each. Our decisions concerning the allocation of research, development, collaboration, management, and financial resources toward particular product candidates or therapeutic areas may not lead to the development of any viable commercial products and may divert resources away from better opportunities or cause us to relinquish valuable rights to such product candidates through collaboration, licensing, or other royalty arrangements in cases in which it would have been advantageous for us to invest additional resources to retain sole development and commercialization rights. If we make incorrect determinations regarding the viability or market potential of any of our product candidates or misreads trends in the healthcare industry, our business, financial condition, and results of operations would be materially and adversely affected.
Furthermore, there can be no assurances of our successful commercialization of other products in our development pipeline. In addition, there can be no assurances that we will be able to raise additional funds in a timely manner, on acceptable terms, if needed to support commercial operations resulting in a material detrimental effect on our ability to become profitable and accordingly be a material factor to the detriment of our ability to continue as a going concern as well as having a material adverse effect on our business, results of operations, financial condition, and cash flow and ability to operate in the future.
We will need substantial additional funding, and If we are unable to raise capital when needed or on terms favorable to us, our business, financial condition, and results of operation could be materially and adversely affected.
We have consumed substantial amounts of capital to date, and we expect to incur net losses over the next several years as we continue to develop our business, direct market our products, and make investments in our human capital in order to grow our business. We expect to continue to spend substantial amounts to continue the development of our pipeline of product candidates, to complete our currently planned clinical trials and future clinical trials, to achieve and maintain market acceptance by physicians and patients, expand our marketing channels and operations, grow our US commercial sales force, grow and enhance our platform offering of products, and make the necessary investments in human capital to scale our business. Other unanticipated costs may arise in the course of our development efforts. If we are able to gain marketing clearance, authorization, or approval for additional product candidates, we will require significant additional amounts of funding in order to launch and commercialize such additional product candidates. We cannot reasonably estimate the actual amounts necessary to successfully complete the development and commercialization of any product candidate we develop and may need substantial additional funding in the future to complete the development and commercialization of our existing and any future product candidates. Our future need for additional funding depends on many factors, including:
•the scope, progress, results, and costs of researching and developing our current product candidates, as well as other additional product candidates we may develop and pursue in the future;
•the timing of, and the costs involved in, obtaining marketing clearance, authorization, or approvals for our product candidates and any other additional product candidates we may develop and pursue in the future;
•the number of future product candidates that we may pursue and their development requirements;
•the costs of commercialization activities for our product candidates, including the costs and timing of establishing product sales, marketing, and distribution capabilities;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 65
•revenue received from commercial sales of our current products and, subject to receipt of regulatory clearance, authorization, or approval, revenue, if any, received from commercial sales of our product candidates;
•the extent to which our in-licenses or acquires rights to other products, product candidates, or technologies;
•our investment in our human capital required to grow the business and the associated costs as we expand our research and development, and establish a commercial infrastructure;
•the costs of preparing, filing, and prosecuting patent applications, maintaining, and protecting our intellectual property rights, including enforcing and defending intellectual property-related claims; and
•the costs of operating as a public company.
We cannot be certain that additional funding will be available on acceptable terms, or at all. If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, reduce, or terminate our product development programs or plans for commercialization. Further, if we raise additional capital in the form of capital stock (or securities exchangeable therefore), such issuances could dilute the interests of our stockholders.
As of December 31, 2021, we had cash and cash equivalents totaling $169.6 million and there is substantial doubt about our ability to continue as a growing concern. We do not currently have any commitments for future funding or additional capital. If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, scale back, or discontinue the development or commercialization of our products or future product candidates or other research and development initiatives or commercial activities. We may need to seek collaborators for our products and any future product candidates at an earlier stage than otherwise would be desirable or on terms that are less favorable than might otherwise be available or relinquish or license on unfavorable terms our rights to our products and any future product candidates in markets where we otherwise would seek to pursue development or commercialization ourselves. Any of the above events could significantly harm our business, prospects, financial condition, and results of operations and cause the price of our common stock to decline.
Our estimates may prove to be wrong, and we could use our available capital resources sooner than expected. Further, changing circumstances, some of which are beyond our control, could cause us to consume capital significantly faster than anticipated, and we may need to seek additional funds sooner than planned. If adequate funds are not available on acceptable terms, we may not be able to successfully execute our business plan or continue our business.
The amount of our future losses is uncertain and our quarterly and annual operating results may fluctuate significantly or fall below the expectations of investors or securities analysts, each of which may cause our stock price to fluctuate or decline.
Our quarterly and annual operating results may fluctuate significantly in the future due to a variety of factors, many of which are outside of our control and may be difficult to predict, including the following:
•the timing and success or failure of clinical trials for our product candidates or competing product candidates, or any other change in the competitive landscape of our industry, including consolidation among our competitors or partners or as a result of COVID-19;
•our ability to successfully recruit and retain subjects for clinical trials, and any delays caused by difficulties in such efforts, including as a result of COVID-19;
•our ability to obtain marketing authorization for our product candidates and the timing and scope of any such marketing authorizations we may receive;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 66
•the timing and cost of, and level of investment in, research and development activities relating to our product candidates, which may change from time to time;
•our ability to attract, hire and retain qualified personnel;
•expenditures that we will or may incur to develop additional product candidates;
•the level of demand for our products and product candidates, should such product candidates receive marketing authorizations, which may vary significantly;
•the risk/benefit profile, cost and reimbursement policies with respect to our products and product candidates, if granted marketing authorization, and existing and potential future therapeutics that compete with our product candidates;
•the changing and volatile US and global economic environments; and
•future accounting pronouncements or changes in our accounting policies.
The cumulative effects of these factors could result in large fluctuations and unpredictability in our quarterly and annual operating results. As a result, comparing our operating results on a period-to-period basis may not be meaningful. This variability and unpredictability could also result in our failing to meet the expectations of industry or financial analysts or investors for any period. If our operating results or revenue fall below the expectations of analysts or investors or below any forecasts we may provide to the market, or if the forecasts we provide to the market are below the expectations of analysts or investors, the price of our common stock could decline substantially. Such a stock price decline could occur even when we have met any previously publicly stated guidance we may provide.
Risks Related to Our Intellectual Property and Technology
Limitations on our ability to maintain or obtain patent protection and/or the patent rights relating to our products and product candidates may limit our ability to prevent third parties from competing against us.
Our success depends, in part, on our ability to obtain and maintain patent protection (including utility patents and design patents) for our products and product features, including back-end architecture and graphical user interfaces. Our success further depends on our ability to obtain copyright registrations for our products’ source In addition, government funding of the SEC and other government agencies on which our operations may rely, including those that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies, may also slow the time necessary for new digital therapeutics to be reviewed and/or granted marketing authorization by necessary government agencies, which would adversely affect our business. For example, in recent years, including for 35 days beginning on December 22, 2018, the US government shut down several times and certain regulatory agencies, such as the FDA and the SEC, had to furlough critical employees and stop critical activities.
If a prolonged government shutdown occurs, or if global health concerns continue to prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews or other regulatory activities, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Future government shutdowns or delays could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Risks Related to Our Intellectual Property and Technology
Limitations on our ability to maintain or obtain patent protection and/or the patent rights relating to our products and product candidates may limit our ability to prevent third parties from competing against us.
Our success depends, in part, on our ability to obtain and maintain patent protection (including utility patents and design patents) for our products and product features, including back-end architecture and graphical user interfaces. Our success further depends on our ability to obtain copyright registrations for our products’ source
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 69
code; to obtain and maintain trademark protection for our product names and other key marks; to preserve our trade secrets and know-how; and to operate without infringing the intellectual property rights of others.
We cannot assure investors that we will continue to innovate and file new patent applications, or that if filed any future patent applications will result in granted patents. We cannot assure you that any of our currently pending patent applications will result in issued patents, that any current or future patents will not be challenged, invalidated or circumvented, that the scope of any of our patents will exclude competitors, or that the patent rights granted to us will provide us any competitive advantage or protect our products. The patent position of PDT companies, including ours, is generally uncertain and involves complex legal and factual considerations and, therefore, validity and enforceability cannot be predicted with certainty. Patents may be challenged, deemed unenforceable, invalidated or circumvented. We will be able to protect our proprietary rights from unauthorized use by third parties only to the extent that our proprietary technologies and any future products are covered by valid and enforceable patents and/or copyrights or are effectively maintained as trade secrets.
Any patents we have obtained or do obtain may be challenged in the US Patent and Trademark Office (“USPTO”) or in federal courts, and may be invalidated or otherwise found unenforceable. Both the patent application process and the process of managing patent disputes can be time-consuming and expensive. If we (either alone or with a patent licensor or co-owner, as the case may require) were to initiate legal proceedings against a third party to enforce a patent related to one of our products, the defendant in such litigation could counterclaim that our (or our licensors’) patent is invalid and/or unenforceable. In patent litigation in the US, defendant counterclaims alleging invalidity and/or unenforceability are commonplace, as are validity challenges by the defendant against the subject patent or other patents before the USPTO. Grounds for a validity challenge could be an alleged failure
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 67
to meet any of several statutory requirements, including lack of novelty, obviousness or non-enablement, failure to meet the written description requirement, indefiniteness, and/or failure to claim patent-eligible subject matter. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent intentionally withheld material information from the USPTO, or made a misleading statement, during prosecution, or otherwise committed fraud on the USPTO. Additional grounds for an unenforceability assertion include an allegation of misuse or anticompetitive use of patent rights, and an allegation of incorrect inventorship with deceptive intent. Third parties may also raise similar claims before the USPTO even outside the context of litigation. The outcome is unpredictable following legal assertions of invalidity and unenforceability. With respect to the validity question, for example, we cannot be certain that no invalidating prior art existed of which we (or a patent licensor or co-owner) and the patent examiner were unaware during prosecution. These assertions may also be based on information known to us or the USPTO. If a defendant or third party were to prevail on a legal assertion of invalidity and/or unenforceability, we would lose at least part, and perhaps all, of the claims of the challenged patent. Such a loss of patent protection would or could have a material adverse impact on our business.
The standards that the USPTO (and foreign equivalents) use to grant patents are not always applied predictably or uniformly and can change. There is also no uniform, worldwide policy regarding the subject matter and scope of claims granted or allowable in utility patents. Accordingly, we do not know the degree of future protection for our proprietary rights or the breadth of claims that will be allowed in any patents issued to us (or to a patent licensor) or to others.
There can be no assurance that our technology will not be found in the future to infringe upon the rights of others or be infringed upon by others. Moreover, patent applications are in some cases maintained in secrecy until patents are issued. The publication of discoveries in the scientific or patent literature frequently occurs substantially later than the date(s) on which the underlying discoveries were made and patent applications were filed. Because patents can take many years to issue, there may be currently pending applications of which we are unaware that may later result in issued patents that our products or product candidates infringe. For example, pending applications may exist that provide support or can be amended to provide support for a claim that results in an issued patent that our products infringe. In such a case, others may assert infringement claims against us, and should we be found to infringe upon their patents or otherwise impermissibly utilize their intellectual property, we might be subject to injunctive relief and/or forced to pay damages, potentially including treble damages, if we are found to have willfully infringed such parties’ patent rights. In addition to any damages we might have to pay, we may be required to obtain licenses from the holders of this intellectual property. We may fail to obtain any of these licenses of intellectual property rights on commercially reasonable terms (and even if we are able to obtain a
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 70
license, it may be non-exclusive, in which case our competitors would potentially have access to the same technologies licensed to us as non-exclusive licensees). In that event, we may be required to expend significant time and resources to develop or license replacement technology. If we are unable to do so, we may be unable to develop or commercialize the affected products or product candidates, which could materially harm our business, and the third parties owning such intellectual property rights could seek either an injunction prohibiting our sales, or, with respect to our sales, an obligation on our part to pay royalties and/or other forms of compensation. Conversely, we may not always be able to successfully pursue our claims against others that infringe intellectual property rights in our technology. Thus, the proprietary nature of our technology or technology licensed by us may not provide adequate protection against competitors.
In addition to patents, we rely on copyrights to protect our products’ source code. We also rely on trademarks and trade names to differentiate our products from those of others and to protect the recognition of our company and products in the marketplace. We also rely on trade secrets, know-how, and proprietary knowledge that we seek to protect, in part, through confidentiality agreements with employees, consultants and others. We cannot assure you, however, that our proprietary information will not be shared or accessed without authorization, that our confidentiality agreements will not be breached, that we will have adequate remedies for any breach, or that our trade secrets will not otherwise become known to or independently developed by competitors.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 68
Changes to the patent law in the US and other jurisdictions could diminish the value of patents in general and may impact the validity, scope or enforceability of our patent rights, thereby impairing our ability to protect our products and product candidates.
As is the case with other digital therapeutic companies, our success is dependent on intellectual property, particularly patents and trade secrets. Obtaining and enforcing patents in the digital therapeutic industry involve both technological and legal complexity and are therefore costly, time consuming, and inherently uncertain. Our patent rights, their associated costs, and the enforcement or defense of such patent rights may be affected by developments or uncertainty in the patent statute, patent case law or USPTO rules and regulations. Changes in either the patent laws or interpretation of the patent laws could increase the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of our issued patents.
For example, the US Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. Additionally, there have been recent proposals for additional changes to the patent laws of the US and other countries that, if adopted, could impact our ability to obtain patent protection for our proprietary technology or our ability to enforce rights in our proprietary technology. Depending on future actions by the US Congress, the US courts, the USPTO and the relevant law-making bodies in other countries, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce any patents that we may obtain in the future.
In addition, it is uncertain whether the World Trade Organization (the “WTO”) will waive certain intellectual property protections now or in the future on certain technologies. It is unknown if such a waiver would be limited to patents or would include other forms of intellectual property including trade secrets and confidential know-how. We cannot be certain that any of our current or future product candidates or technologies would not be subject to an intellectual property waiver by the WTO. We also cannot be certain that any of our current or future intellectual property rights would be eliminated, narrowed, or weakened by such a waiver. Given the uncertain future actions by the WTO and other countries and jurisdictions around the world, including the US, it is unpredictable how our current or future intellectual property rights or how our current or future business would be impacted.
If our trademarks and trade names are not adequately protected, we may not be able to build name recognition in our markets of interest and our competitive position may be harmed.
The registered or unregistered trademarks or trade names that we own may be challenged, infringed, circumvented, declared generic, lapsed or determined to be infringing on or dilutive of other marks. We may not be able to protect our rights in these trademarks and trade names, which we need in order to build name recognition with the public. In addition, third parties have filed, and may in the future file, for registration of trademarks similar or identical to our trademarks, thereby impeding our ability to build brand identity and possibly leading to market confusion. If they succeed in registering such trademarks and/or in developing common law rights in such trademarks, and if we are not successful in challenging such third-party rights, we may not be able to use these trademarks to develop brand recognition of our services. In addition, there could be potential trade name or trademark infringement claims brought by owners of other registered trademarks or trademarks that incorporate variations of our registered or unregistered trademarks or trade names. If we are unable to establish or protect our trademarks and trade names, or if we are unable to build name recognition based on our trademarks and trade names, we may not be able to compete effectively, which could harm our competitive position, business, financial condition, results of operations and prospects.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 71
Accusations of infringement of third-party intellectual property rights could materially and adversely affect our business.
There has been substantial litigation in the areas in which we operate regarding intellectual property rights, and we may be sued for infringement from time to time in the future. Also, in some instances, we have agreed to indemnify third parties for expenses and liability resulting from claimed intellectual property infringement. From time to time, we may receive requests for indemnification in connection with allegations of intellectual property
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 69
infringement and we may choose, or be required, to assume the defense and/or reimburse third parties for their expenses, settlement and/or liability. We cannot assure you that we will be able to settle any future claims or, if we are able to settle any such claims, that the settlement will be on terms favorable to us. Our broad range of technology may increase the likelihood that third parties will claim that we infringe their intellectual property rights.
We may in the future receive notices of allegations of infringement, misappropriation or misuse of other parties’ proprietary rights. Furthermore, regardless of their merits, accusations and litigation of this nature may require significant time and expense to defend, may negatively affect customer relationships, may divert management’s attention away from other aspects of our operations and, upon resolution, may have a material adverse effect on our business, results of operations, financial condition, and cash flows.
Certain technology necessary for us to provide our solutions may, in fact, be patented by other parties either now or in the future. If such technology were validly patented by a third party, we may have to negotiate a license for the use of that technology. We may not be able to negotiate such a license at a price that is acceptable to us or at all. The existence of such a patent, or our inability to negotiate a license for any such technology on acceptable terms, could force us to cease using the technology and cease offering products incorporating the technology, which could materially and adversely affect our business and results of operations.
If we, or any of our products, were found to be infringing on the intellectual property rights of any third party, we could be subject to liability for such infringement, which could be material. We could also be prohibited from using or selling certain products, prohibited from using certain processes, or required to redesign certain products, each of which could have a material adverse effect on our business and results of operations.
These and other outcomes may: result in the loss of a substantial number of existing customers or prohibit the acquisition of new customers; cause us to pay license fees for intellectual property we are deemed to have infringed; cause us to incur costs and devote valuable technical resources to redesigning our products; cause our cost of product revenues to increase; cause us to accelerate expenditures to preserve existing revenues; materially and adversely affect our brand in the marketplace and cause a substantial loss of goodwill; cause us to change our business methods or products; and require us to cease certain business operations or offering certain products or features.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In addition to patent protection, we rely heavily upon know-how and trade secret protection, as well as non-disclosure agreements and invention assignment agreements with our employees, consultants and third parties, to protect our confidential and proprietary information, especially where we do not believe patent protection is appropriate or obtainable. It is our policy to require our employees and consultants to execute confidentiality agreements upon the commencement of employment or consulting relationships with us. These agreements provide that all confidential information concerning our business or financial affairs developed or made known to the individual or entity during the course of the party’s relationship with us is to be kept confidential and not disclosed to third parties except in specific circumstances. In the case of employees, the agreements provide that all inventions conceived or completed by the individual, and which are related to our current or planned business or research and development or made during normal working hours, on our premises or using our equipment or proprietary information, are our exclusive property. In addition to contractual measures, we try to protect the confidential nature of our proprietary information using physical and technological security measures. Such measures may not, for example, in the case of misappropriation of a trade secret by an employee or third party with authorized access, provide adequate protection for our proprietary information. Our security measures may,
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 72
for example, not prevent an employee or consultant from misappropriating our trade secrets and providing them to a competitor, and recourse we take against such misconduct may not provide an adequate remedy to protect our interests fully.
Enforcing a claim that a party illegally disclosed or misappropriated a trade secret can be difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, trade secrets may be independently developed by
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 70
others in a manner that could prevent legal recourse by us. If any of our confidential or proprietary information, such as our trade secrets, were to be disclosed or misappropriated, or if any such information were to be independently developed by a competitor, our competitive position could be harmed. In addition, courts outside the US may be less willing to protect trade secrets. Thus, we may not be able to meaningfully protect our trade secrets outside the US.
If we choose to go to court to stop a third party from using any of our trade secrets, we may incur substantial costs. These lawsuits may consume our time and other resources even if we are successful. Although we take steps to protect our proprietary information and trade secrets, including through contractual means with our employees and consultants, third parties may independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets or disclose our technology.
Third parties may assert that our employees, consultants, collaborators or partners have wrongfully used or disclosed confidential information or misappropriated trade secrets.
As is common in the digital health, technology, or pharmaceutical industries, we employ individuals who were previously employed at universities or other digital health, technology, or pharmaceutical companies, including our competitors or potential competitors. Although no claims against us are currently pending, and although we try to ensure that our employees and consultants do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that we or our employees or consultants have inadvertently or otherwise used or disclosed intellectual property, including trade secrets or other proprietary information, of a former employer or other third party. Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. This risk is similarly applicable with respect to claims by third parties against any current or future licensors.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on product candidates in all countries throughout the world is expensive. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as laws in the US. For instance, following Russia’s invasion of Ukraine in February 2022, the US government levied sanctions against Russia, and Russia responded by issuing a decree that removes protections for some patent holders who are registered in unfriendly countries, including the US. Accordingly, the USPTO has terminated its engagement with officials from intellectual property agencies in Russia. This reaction from a globally significant nation sets an unwieldy precedent, wherein other countries may now be expected to respond to political sanctions by extinguishing the intellectual property rights of those it deems “unfriendly.” Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the US where we have issued patents, or from selling or importing products made using our inventions in other jurisdictions. Competitors may also use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where we do not have patent protection or where we have patent protection but where enforcement is not as strong as that in the US. These products may compete with our products, and our patents or other intellectual property rights may not be effective or sufficient to prevent such competition.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries do not favor the enforcement of patents, trade secrets and other intellectual property rights, particularly those relating to digital health, pharmaceutical and biopharmaceutical products, which could make it difficult for us, our licensors, or our licensees to stop the infringement of our patents or marketing of competing products against third parties in violation of our proprietary
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 73
rights generally. The initiation of proceedings for infringement by third parties or by third parties to challenge the scope or validity of our patent rights in foreign jurisdictions could also result in substantial cost and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and any related patent applications at risk of not issuing, and could provoke third parties to assert claims against us, our licensors, or our licensees. We may not prevail in any lawsuits that we initiate or that are initiated against us, and the damages or other remedies awarded in lawsuits that we initiate, if any, may not be commercially meaningful.
Many countries, including European Union countries, India, Japan and China, have compulsory licensing laws under which a patent owner may be compelled under specified circumstances to grant licenses to third parties. In addition, many countries limit the enforceability of patents against government agencies or government contractors. In those countries, we may have limited remedies if patents are infringed or if we are compelled to
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 71
grant a license to a third party, which could materially diminish the value of those patents. This could limit our potential revenue opportunities. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
We are party to and may, in the future, enter into collaborations, in-licensing arrangements, joint ventures, or strategic alliances with third parties that may not result in the development of commercially viable products or the generation of significant or any future revenues.
In the ordinary course of our business, we have and may continue to enter into collaborations, in-licensing arrangements, joint ventures, or strategic alliances to develop new PDTs and to pursue new markets. Proposing, negotiating, and implementing collaborations, in-licensing arrangements, joint ventures, and strategic alliances may be a lengthy and complex process. Other companies, including those with substantially greater financial, marketing, sales, technology or other business resources, may compete with us for these opportunities or arrangements. We may not identify, secure or complete any such transactions or arrangements in a timely manner, on a cost-effective basis, on acceptable terms, or at all. We have limited institutional knowledge and experience with respect to these business development activities, and we may also not realize the anticipated benefits of any such transaction or arrangement. In particular, these collaborations may not result in the development of products that achieve commercial success or result in significant revenues and could be terminated prior to developing any products.
Additionally, we may not be in a position to exercise sole decision-making authority regarding the transaction or arrangement, which could create the potential risk of creating impasses on decisions, and our collaborators may have economic or business interests or goals that are, or that may become, inconsistent with our business interests or goals. It is possible that conflicts may arise with our collaborators, such as conflicts concerning the achievement of performance milestones, or the interpretation of significant terms under any agreement, such as those related to financial obligations or the ownership or control of intellectual property developed during the collaboration. IfSome of our current license agreements impose various development, diligence, commercialization or sublicensing, and other obligations. In spite of our efforts, a current or future licensor might conclude that we have materially breached our obligations under such license agreements and seek to terminate the license agreements, thereby removing or limiting our ability to develop and commercialize products and technology covered by these license agreements. Furthermore, if any conflicts arise with our current or future collaborators, they may act in their self-interest, which may be adverse to our best interest, and they may breach their obligations to us. In addition, we have limited control over the amount and timing of resources that our current collaborators or any future collaborators devote to our collaborators’ or our future products. Disputes between us and our collaborators may result in litigation or arbitration which would increase our expenses and divert the attention of our management. The agreements under which we may license intellectual property or technology from third parties may be complex, and certain provisions in such agreements may be susceptible to multiple interpretations. Further, these transactions and arrangements are contractual in nature and may be terminated or dissolved under the terms of the applicable agreements and, in such event, we may not continue to have rights to the products relating to such transaction or arrangement or may need to purchase such rights at a premium.
We in-license patents and content from third parties to develop our products and product candidates. If we had a dispute or fail to comply with obligations in the agreements with a third-party licensor, we could lose rights that are important to our business, or it could materially and adversely affect our ability to commercialize the product or product candidate affected by the dispute.
We license certain intellectual property that is important to our business, and in the future we may enter into additional agreements that provide us with licenses to valuable intellectual property or technology. Some of our current license agreements impose If any of these various development, diligence, commercialization or sublicensing, and other obligations, including payments in connection with the achievement of specified milestones, on us in order to maintain the licenses. In spite of our efforts, a current or future licensor might conclude that we have materially breached our obligations under such license agreements and seek to terminate the license agreements, thereby removing or limiting our ability to develop and commercialize products and technology covered by these license agreements. If these in-licenses arecollaborations were terminated, or if the underlying patent rights licensed thereunder fail to provide the intended exclusivity, competitors or other third parties would have the freedom to seek marketing authorization of, and to market, products identical to ours and we may be required to cease our development and commercialization of certain of our product candidates. Any of the foregoing could have a material adverse effect on our business, prospects, results of operations and financial condition.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 72
Licensing intellectual property involves complex legal, business and scientific issues. Disputes may arise between us and our licensors regarding intellectual property subject to a license agreement, including:
•the scope of rights granted under the license agreement and other interpretation-related issues;
•amount of royalty payments under the license agreement;
•whether and to what extent our technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement;
•our right to sublicense patent and other rights to collaborators and other third parties;
•our diligence obligations with respect to the use of the licensed technology in relation to our development and commercialization of our products, and what activities satisfy those diligence obligations; and
•the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our collaborators.
If disputes over licensed intellectual property prevent or impair our ability to maintain the licensing arrangements on acceptable terms, we may be unable to successfully develop and commercialize the affected product, or the dispute may have an adverse effect on our results of operation.
The agreements under which we may license intellectual property or technology from third parties may be complex, and certain provisions in such agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could narrow what we believe to be the scope of our rights to the relevant intellectual property or technology, or increase what we believe to be our financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, prospects, results of operations and financial condition. Moreover, if disputes over intellectual property that we have licensed prevent or impair our ability to maintain our licensing arrangements on commercially acceptable terms, we may be unable to successfully develop and commercialize the affected product candidates, which could have a material adverse effect on our business, prospects, results of operations and financial condition.
We or our licensors may be subject to claims challenging the inventorship or ownership of the patents and other intellectual property that we own or license now or in the future.
We or our licensors may be subject to claims that former employees, consultants, or other third parties have an ownership interest in the patents and intellectual property that we own or in-license or that we may own or in-license in the future. While it is our policy to require our employees and consultants who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own or such assignments may not be self-executing or may be breached. Our licensors may face similar obstacles. We or our licensors could be subject to ownership disputes arising, for example, from conflicting obligations of employees, consultants or others who are involved in developing our product candidates. Litigation may be necessary to defend against any claims challenging inventorship or ownership. If we or our licensors fail in defending any such claims, we may have to pay monetary damages and may lose valuable intellectual property rights, such as exclusive ownership of, or right to use, intellectual property, which could adversely impact our business, results of operations and financial condition.
Our products utilize third-party open source software components, which may pose particular risks to our proprietary software, technologies, products and services in a manner that could negatively affect our business.
We have chosen, and we may choose in the future, to use open source software in our products. We use various software composition tools, including Veracode and Snyk
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 74
of, and to market, products identical to ours and we may be required to cease our development and commercialization of certain of our product candidates.
Our products utilize third-party open source software components, which may pose particular risks to our proprietary software, technologies, products and services in a manner that could negatively affect our business.
We have chosen, and we may choose in the future, to use open source software in our products. We use various software composition tools, including Veracode and SonarQube, which are designed to monitor risks related to licenses and vulnerabilities related to open-source software. Use and distribution of open source software may entail greater risks than use of third-party commercial software, as open source licensors generally do not provide warranties or other contractual protections regarding infringement claims or the quality of the code. Some open source licenses may contain requirements that we make available source code for modifications or derivative
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 73
works we create based upon the type of open source software we use. If we combine our proprietary software with open source software in a certain manner, we could, under certain open source licenses, be required to release the source code of our proprietary software to the public. This would allow our competitors to create similar products with less development effort and time and ultimately could result in a loss of product sales.
Although we intend to monitor any use of open source software to avoid subjecting our products to conditions we do not intend, the terms of many open source licenses have not been interpreted by US courts, and there is a risk that any such licenses could be construed in a way that could impose unanticipated conditions or restrictions on our ability to commercialize our products. Moreover, there is no assurance that our processes for controlling our use of open source software in our products will be effective. If we are held to have breached the terms of an open source software license, we could be required to seek licenses from third parties to continue offering our products on terms that are not economically feasible, to re-engineer our products, to discontinue the sale of our products if re-engineering could not be accomplished on a timely basis, or to make generally available, in source code form, our proprietary code, any of which could materially and adversely affect our business, operating results and financial condition.
Risks Relating to Our Products Some of our products and research initiatives may be partially supported by government grant awards, which may or may not be available to us in the future or may subject us to federal regulations such as “march-in” rights, certain reporting requirements, and a preference for US manufacturing.
We have received various forms of grant funding from the National Institute on Drug Abuse (NIDA) to support the clinical and commercial development activities of reSET and reSET-O in substance use disorder and opioid use disorder, respectively. To fund a portion of our future research initiatives and development programs, we may apply for additional grant funding from NIDA or other governmental agencies. However, funding by these governmental agencies may be significantly reduced or eliminated in the future for a number of reasons. For example, some programs are subject to a yearly appropriations process in Congress. In addition, we may not receive full funding under current or future grants because of budgeting constraints of the agency administering the program or unsatisfactory progress on the study being funded. Therefore, we cannot be certain that we will receive any future grant funding from any government agencies, or, that if received, we will receive the full amount of the particular grant award. Any such reductions could delay the development of our products and research initiatives.
Moreover, patent rights on inventions conceived or first actually reduced to practice in the performance of work under a US government funding agreement, or subject inventions, are subject to the Bayh-Dole Act of 1980, or Bayh-Dole Act. US government rights in subject inventions include a non-exclusive, non-transferable, irrevocable, paid up license to practice or have practiced for or on behalf of the US the subject invention throughout the world. In addition, the US government has the right to require us to grant exclusive, partially exclusive, or non-exclusive licenses to any subject inventions to a third party if the government determines that: (1) adequate steps have not been taken to commercialize the subject invention; (2) government action is necessary to meet public health or safety needs; or (3) government action is necessary to meet requirements for public use under federal regulations, which we refer to as march-in rights. The US government also has the right to take title to subject inventions if we fail, or the applicable licensor fails, to disclose the subject invention to the government, elect title, and file a patent application within specified time limits or if we cease to prosecute the patent application or will allow a patent to
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 75
lapse. Each subject invention is also subject to certain reporting requirements, compliance with which may require us, or the applicable licensor, to expend substantial resources. In addition, the Bayh Dole Act requires that any exclusive license to use or sell products embodying the subject invention or produced through use of the subject invention in the US include a requirement that such licensed products will be manufactured substantially in the US. The manufacturing preference requirement can be waived if the patent owner can show that reasonable but unsuccessful efforts have been made to grant licenses on similar terms to potential licensees that would be likely to manufacture substantially in the US or that under the circumstances domestic manufacture is not commercially feasible.
As a result of any funding from NIDA, or if we enter into future arrangements involving federal government funding and conceive or first actually reduce to practice an invention in the performance of work under such funding, patent rights in any such subject inventions may be subject to the applicable provisions of the Bayh-Dole Act. To the extent any of our current or future patent rights relate to subject inventions, the provisions of the Bayh-Dole Act may similarly apply. Any exercise by the federal government of certain of its rights under the Bayh-Dole Act could harm our competitive position, business, financial condition, results of operations, and prospects.
Risks Relating to Our Products
The success of any enhancements or improvements to our products or any new products depends on several factors, including regulatory review timelines, timely completion, competitive pricing, adequate quality testing, integration with new and existing technologies in our products and third-party collaborators’ technologies and overall market acceptance.
We expect that the PDT market, as with many technology markets, will be characterized by rapid technological change, frequent new product and service introductions and enhancements, changing customer demands, and evolving industry standards. As an initial matter, a significant portion of our market may not have access to smartphones or other technology necessary to utilize our PDTs. In addition, the introduction of products and services embodying new technologies could quickly make existing products and services obsolete and unmarketable. Additionally, changes in laws and regulations could impact the usefulness of our products and could necessitate changes or modifications to our products to accommodate such changes. We invest substantial resources in researching and developing new products and enhancing our existing products by incorporating additional features, improving functionality, and adding other improvements to meet our patients’ evolving needs. If we are unable to develop and release new products, or successful enhancements, new features and modifications to our existing products, our business, financial condition and results of operations could be materially and adversely affected. We may not succeed in developing, marketing and delivering on a timely and cost-effective basis enhancements or improvements to our products or any new products that respond to continued changes in market demands or new customer requirements, and any enhancements or improvements to our products or any new products may not achieve market acceptance. Since developing our products is complex, the timetable for the release of new products and enhancements to existing products is difficult to predict, and we may not offer new products and updates as rapidly as our users require or expect. Any new products that we develop or acquire may not be introduced in a timely or cost-effective manner, may contain errors or defects, or may not achieve the broad market acceptance necessary to generate significant or any revenue.
The introduction of new products and products by competitors, the development of entirely new technologies to replace existing offerings or shifts in healthcare benefits trends could make our products obsolete or materially and adversely affect our business, financial condition and results of operations. We may experience difficulties with software development, industry standards, design or marketing that could delay or prevent our development, introduction or implementation of new products, enhancements, additional features or capabilities. If patients and healthcare providers do not widely adopt our products, we may not be able to realize a return on our investment. If we do not accurately anticipate patient demand or we are unable to develop, license or acquire new features and capabilities on a timely and cost-effective basis, or if such enhancements do not achieve market acceptance, it could result in adverse publicity, loss of revenue or market acceptance or claims by patients or healthcare providers brought against us, each of which could have a material and adverse effect on our reputation, business, results of operations and financial condition.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 76
Our products may cause undesirable side effects or have other properties that could limit their commercial potential.
If we or others identify undesirable side effects directly or indirectly caused by our products, a number of potentially significant negative consequences could result, including:
•regulatory authorities may withdraw clearance, authorization, or approvals of such product;
•regulatory authorities may require additional warnings on the product’s label;
•we may be required to issue safety communications to patients or healthcare providers that outline the risks of such side effects;
•we could be sued and held liable for harm caused to patients; and
•our reputation may suffer.
Any of these events could prevent us from achieving or maintaining market acceptance of the particular product or product candidate and, as a result of negative impacts to our reputation, our other products or product candidates and could significantly harm our business, results of operations and prospects.
Our current product candidates are in various stages of development. Our product candidates may fail in development or suffer delays that adversely affect their commercial viability. If we fail to maintain clearance, de novo classification or authorization to market our product candidates for expanded indications, or if we are delayed in obtaining such marketing authorizations, our business, prospects, results of operations and financial condition could be materially and adversely affected.
The process of seeking FDA marketing authorization is expensive and time consuming. There can be no assurance that marketing authorization will be granted. If we are not successful in obtaining timely clearance, de novo classification or approval of our product candidates, we may never be able to generate significant revenue and may be forced to cease operations. The FDA can delay, limit or deny for many reasons, including:
•We may not be able to demonstrate to the FDA’s satisfaction that our product candidates meet the applicable regulatory standards for clearance, de novo classification, or authorization, as applicable;
•The FDA may disagree that our clinical data supports the label and use that we are seeking; and
•The FDA may disagree that the data from our preclinical or pilot studies and clinical trials is sufficient to support marketing authorization.
Obtaining marketing authorization from the FDA or any foreign regulatory authority could result in unexpected and significant costs for us and consume management’s time and other resources. The FDA could ask us to supplement our submissions, collect additional nonclinical data, conduct additional clinical trials, prepare additional manufacturing data or information or engage in other time-consuming actions, or it could simply deny our applications. In addition, if granted marketing authorization, we will be required to obtain additional FDA authorizations or clearances prior to making certain modifications to our devices. Further, FDA may impose other restrictions on our marketing authorizations, or we may lose marketing authorization, if post-market data demonstrates safety issues or lack of effectiveness. If we are unable to obtain and maintain the necessary marketing authorizations to market our products, our financial condition may be adversely affected, and our ability to grow domestically and internationally would likely be limited. Additionally, even if granted marketing authorization, our products may not receive marketing authorization for the indications that are necessary or desirable for successful commercialization or profitability. This could have a material adverse effect on our business, prospects, results of operations and financial condition.
Our products may cause undesirable side effects or have other properties that could limit their commercial potential.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 74
If we or others identify undesirable side effects directly or indirectly caused by our products, a number of potentially significant negative consequences could result, including:
•regulatory authorities may withdraw clearance, authorization, or approvals of such product;
•regulatory authorities may require additional warnings on the product’s label;
•we may be required to issue safety communications to patients or healthcare providers that outline the risks of such side effects;
•we could be sued and held liable for harm caused to patients; and
•our reputation may suffer.
Any of these events could prevent us from achieving or maintaining market acceptance of the particular product or product candidate and, as a result of negative impacts to our reputation, our other products or product candidates and could significantly harm our business, results of operations and prospects.
If we are unable to develop and release new products, or successful enhancements, new features and modifications to our existing products, our business, financial condition and results of operations could be materially and adversely affected.
We expect that the PDT market, as with many technology markets, will be characterized by rapid technological change, frequent new product and service introductions and enhancements, changing customer demands, and evolving industry standards. As an initial matter, a significant portion of our market may not have access to smartphones or other technology necessary to utilize our PDTs. In addition, the introduction of products and services embodying new technologies could quickly make existing products and services obsolete and unmarketable. Additionally, changes in laws and regulations could impact the usefulness of our products and could necessitate changes or modifications to our products to accommodate such changes. We invest substantial resources in researching and developing new products and enhancing our existing products by incorporating additional features, improving functionality, and adding other improvements to meet our patients’ evolving needs. The success of any enhancements or improvements to our products or any new products depends on several factors, including regulatory review timelines, timely completion, competitive pricing, adequate quality testing, integration with new and existing technologies in our products and third-party collaborators’ technologies and overall market acceptance. We may not succeed in developing, marketing and delivering on a timely and cost-effective basis enhancements or improvements to our products or any new products that respond to continued changes in market demands or new customer requirements, and any enhancements or improvements to our products or any new products may not achieve market acceptance. Since developing our products is complex, the timetable for the release of new products and enhancements to existing products is difficult to predict, and we may not offer new products and updates as rapidly as our users require or expect. Any new products that we develop or acquire may not be introduced in a timely or cost-effective manner, may contain errors or defects, or may not achieve the broad market acceptance necessary to generate significant or any revenue.
The introduction of new products and products by competitors, the development of entirely new technologies to replace existing offerings or shifts in healthcare benefits trends could make our products obsolete or materially and adversely affect our business, financial condition and results of operations. We may experience difficulties with software development, industry standards, design or marketing that could delay or prevent our development, introduction or implementation of new products, enhancements, additional features or capabilities. If patients and healthcare providers do not widely adopt our products, we may not be able to realize a return on our investment. If we do not accurately anticipate patient demand or we are unable to develop, license or acquire new features and capabilities on a timely and cost-effective basis, or if such enhancements do not achieve market acceptance, it could result in adverse publicity, loss of revenue or market acceptance or claims by patients or healthcare providers brought against us, each of which could have a material and adverse effect on our reputation, business, results of operations and financial condition.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 75
Clinical trials of any of our products or product candidates may fail to produce results necessary to support regulatory clearance or authorization. Clinical trials of any of our products or product candidates may fail to produce results necessary to support regulatory clearance or authorization.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 77
We incur substantial expense for, and devote significant time to, clinical trials but cannot be certain that the trials will ever result in commercial gains. We may experience significant setbacks in clinical trials, even after earlier clinical trials showed promising results, and failure can occur at any time during the clinical development process. Any of our products may malfunction or may produce undesirable adverse effects that could cause us, institutional review boards (“IRBs”) or regulatory authorities to interrupt, delay or halt clinical trials. We, IRBs, the FDA, or another regulatory authority may suspend or terminate clinical trials at any time to avoid exposing trial participants to unacceptable health risks. Our clinical trials may produce negative or inconclusive results or may demonstrate a lack of effect of our product candidates. Additionally, the FDA may disagree with our interpretation of the data from our pilot studies and clinical trials, or may find the clinical trial design, conduct or results inadequate to demonstrate safety or effectiveness, and may require us to pursue additional clinical trials, which could further delay the clearance or authorization of our product candidates. If we are unable to demonstrate the safety and effectiveness of product candidates in our clinical trials, we will be unable to obtain the regulatory clearances or authorizations we need to commercialize new products.
In addition, to the extent that additional information regarding products being studied in clinical trials could translate to currently cleared or authorized products, such as information on new side effects, those results may impact existing clearances and authorizations, and required contraindications, warnings or precautions in product labeling.
Interim, “topline” and preliminary data from clinical trials of our products or product candidates may change as more patient data becomes available and are subject to confirmation, audit, and verification procedures that could result in material changes in the final data.
From time to time, we may publicly disclose preliminary or topline data from our pilot studies and clinical trials, which is based on a preliminary analysis of then-available data, and the results and related findings and conclusions are subject to change following a more comprehensive review of the data related to the particular study or trial. We also make assumptions, estimations, calculations, and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully and carefully evaluate all data. As a result, the topline or preliminary results that we report may differ from future results of the same studies, or different conclusions or considerations may qualify such results, once additional data have been received and fully evaluated. Topline data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, topline data should be viewed with caution until the final data are available. From time to time, we may also disclose interim data from our clinical trials. Interim or preliminary data from clinical trials are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment and treatment continues and more patient data become available or as patients from our clinical trials continue other treatments for their disease. Adverse differences between preliminary or interim data and final data could significantly harm our business prospects. Further, disclosure of interim data by us or by our competitors could result in volatility in the price of our Class A Common Sharescommon stock.
Further, third parties, including regulatory agencies, may not accept or agree with our assumptions, estimates, calculations, conclusions or analyses or may interpret or weigh the importance of data differently, which could impact the potential of the particular program, the likelihood of marketing authorization or clearance or commercialization of the particular product candidate, the commercial success of any product for which we may have already obtained authorization or clearance, and our company in general. In addition, the information we choose to publicly disclose regarding a particular study or clinical trial is derived from information that is typically extensive, and you or others may not agree with what we determine is material or otherwise appropriate information to include in our disclosure.
If the interim, topline, or preliminary data that we report differ from actual results, or if others, including regulatory authorities, disagree with the conclusions reached, our ability to obtain clearance, authorization, or
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 76
approval for, and commercialize, our product candidates may be harmed, which could harm our business, operating results, prospects or financial condition. approval for, and commercialize, our product candidates may be harmed, which could harm our business, operating results, prospects or financial condition.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 78
reSET-O is used in combination with buprenorphine which exposes us to additional risks.
reSET-O is FDA-authorized for treatment of OUD in combination with buprenorphine. We are subject to the risk that the FDA could revoke approval of buprenorphine or that safety, efficacy, manufacturing or supply issues could arise with buprenorphine. This could materially and adversely affect reSET-O’s commercial success. Furthermore, if buprenorphine and/or buprenorphine/Naloxone are utilized less due to other medications being used more frequently this could have a negative impact on the use of reSET-O.
Risks Related to Our Regulatory Compliance and Legal Matters
We operate in a highly regulated industry and are subject to a wide range of federal, state and local laws, rules and regulations, including FDA regulatory requirements and laws pertaining to fraud and abuse in healthcare, that affect nearly all aspects of our operations. Failure to comply with these laws, rules and regulations, or to obtain and maintain required licenses, could subject us to enforcement actions, including substantial civil and criminal penalties, and might require us to recall or withdraw a product from the market or cease operations. Any of the foregoing could materially and adversely affect our business, financial condition and results of operations.
We and our products are subject to extensive regulation in the US, including by the FDA. The regulations to which we are subject are complex. The FDA regulates, among other things, with respect to medical devices: design, development and manufacturing; testing, labeling, content and language of instructions for use; clinical trials; product safety; medical device cybersecurity; pre-market clearance, authorization, and approval; establishment registration and device listing; marketing, sales and distribution; complaint handling; record keeping procedures; advertising and promotion; recalls and field safety corrective actions; post-market surveillance, including reporting of deaths or serious injuries and malfunctions that, if they were to recur, could lead to death or serious injury; post-market studies; and product import and export. The FDA monitors compliance with these applicable regulatory requirements through periodic unannounced inspections as well as various other channels, such as reviewing post-market surveillance and recall reports, monitoring advertising and promotional practices on-line and at trade shows, and reviewing trade complaints submitted by competitors or other third parties. We do not know whether we will pass any future inspections for FDA compliance, or whether the FDA might identify compliance concern(s) through other channels of information. Failure to comply with applicable regulations could jeopardize our ability to sell our products and result in enforcement-related actions such as: FDA Form 483s; untitled or warning letters; clinical holds on research; fines; injunctions; civil penalties; termination of distribution; recalls or seizures of products; delays in the introduction of products into the market; total or partial suspension of production; refusal to grant future clearances, authorizations, or approvals; withdrawals or suspensions of current clearances or marketing authorizations, resulting in prohibitions on the sale and distribution of our products; and in the most serious cases, criminal penalties. Any of these sanctions could result in higher than anticipated costs or lower than anticipated sales and have a material adverse effect on our reputation, business, financial condition and results of operations.
The FDA and the Federal Trade Commission (“FTC”)FTC also regulate the advertising and promotion of our products to ensure that the claims we make are consistent with our regulatory authorizations, that there is adequate and reasonable data to substantiate the claims and that our promotional labeling and advertising is neither false nor misleading. If the FDA or FTC determines that any of our advertising or promotional claims are false, misleading, not substantiated or not permissible, we may be subject to enforcement actions, including untitled or warning letters, and we may be required to revise our promotional claims and make other corrections or restitutions. We also may be subject to fines, or other regulatory, civil, or criminal sanctions.
Additional federal and state healthcare laws and regulations that may affect our ability to conduct business include, without limitation:
•the federal Anti-Kickback Statute, which prohibits, among other things, any person from knowingly and willfully offering, soliciting, receiving or providing remuneration, directly or indirectly, in exchange for or to induce either the referral of an individual for, or the purchase, order or recommendation of, any good or service for which payment may be made under federal healthcare programs, such as CMS programs;
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 79
•the federal civil false claims and civil monetary penalties laws, including, without limitation, the federal False Claims Act, which prohibits, among other things, individuals or entities from knowingly presenting, or causing to be presented, false claims, or knowingly using false statements, to obtain payment from the federal government;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 77
•federal criminal laws that prohibit executing a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters;
•the federal Civil Monetary Penalties Law prohibits, among other things, offering or transferring remuneration to a federal healthcare beneficiary that a person knows or should know is likely to influence the beneficiary’s decision to order or receive items or services reimbursable by the government from a particular provider or supplier;
•the federal Physician Payment Sunshine Act, or Open Payments, created under the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act of 2010, or collectively the Affordable Care Act (“ACA”), and its implementing regulations, which requires manufacturers of drugs, medical devices, biologicals and medical supplies for which payment is available under Medicare, Medicaid, or the Children’s Health Insurance Program to report annually to CMS information related to payments or other transfers of value made to licensed physicians and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members;
•Health Insurance Portability Administration and Accountability Act of 1996 (“HIPAA”), as amended by the Health Information Technology for Economic and Clinical Health Act, and its implementing regulations, which impose certain requirements relating to the privacy, security and transmission of protected health information (“PHI”), on certain healthcare providers, health plans and healthcare clearinghouses, and their business associates that access or otherwise process individually identifiable health information on their behalf; HIPAA also created criminal liability for knowingly and willfully falsifying or concealing a material fact or making a materially false statement in connection with the delivery of or payment for healthcare benefits, items or services;
•medical device regulations pursuant to the federal Food, Drug and Cosmetic Act which require, among other things, pre-market clearance, authorization, or approval; compliant labeling; medical device adverse event reporting; establishment registration and device listing; reporting of corrections and removals; and quality system requirements;
•state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payor, including commercial insurers, and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and are in addition to requirements under HIPAA, thus complicating compliance efforts; and
•state laws governing the corporate practice of medicine and other healthcare professions and related fee-splitting laws.
Our employees, consultants and commercial collaborators may engage in misconduct or other improper activities, including non-compliance with such regulatory standards and requirements.
Because of the breadth of these laws and the narrowness of available statutory and regulatory exemptions, it is possible that some of our activities could be subject to challenge under one or more of such laws. Any action brought against us for violations of these laws or regulations, even if successfully defended, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. We may be subject to private “qui tam” actions brought by individual whistleblowers on behalf of the federal or state governments, with potential liability under the federal False Claims Act including mandatory treble damages and significant per-claim penalties.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 80
Although we have adopted policies and procedures designed to comply with these laws and regulations and conduct internal reviews of our compliance with these laws, our compliance is also subject to governmental review. The growth of our business and sales organization including future expansion outside of the US may increase the potential of violating these laws or our internal policies and procedures. The risk of our being found in violation of these or other laws and regulations is further increased by the fact that many have not been fully
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 78
interpreted by the regulatory authorities or the courts, and their provisions are open to a variety of interpretations. Any action brought against us for violation of these or other laws or regulations, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. If our operations are found to be in violation of any of the federal, state and foreign laws described above or any other current or future fraud and abuse or other healthcare laws and regulations that apply to us, we may be subject to penalties, including significant criminal, civil and administrative penalties, damages and fines, disgorgement, additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of noncompliance with these laws, imprisonment for individuals and exclusion from participation in government programs, such as Medicare and Medicaid, as well as contractual damages and reputational harm. We could also be required to curtail or cease our operations. Any of the foregoing consequences could seriously harm our business, financial condition and results of operations.
We willOur employees, independent contractors, consultants and collaborators may engage in misconduct or other improper activities, including non-compliance with regulatory standards and requirements, which could cause significant liability for us and harm our reputation.
We are exposed to the risk that our employees, independent contractors, consultants and collaborators may engage in fraudulent conduct or other illegal activity. It is not always possible to identify and deter misconduct by employees and other third parties, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. Misconduct by those parties could include intentional, reckless and/or negligent conduct or disclosure of unauthorized activities to us that violates:
•FDA regulations or similar regulations of comparable non-US regulatory authorities, including those laws requiring the reporting of true, complete and accurate information to such authorities;
•manufacturing standards;
•federal and state healthcare fraud and abuse laws and regulations and similar laws and regulations established and enforced by comparable non-US regulatory authorities; and
•laws that require the reporting of financial information or data accurately.
In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Activities subject to these laws may involve the improper use or misrepresentation of information obtained in the course of clinical trials, creating fraudulent data in our clinical trials or illegal misappropriation of product materials, which could result in regulatory sanctions and serious harm to our reputation. It is not always possible to identify and deter misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws, standards or regulations. Additionally, we are subject to the risk that a person or government could allege such fraud or other misconduct, even if none occurred. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and results of operations, including the imposition of civil, criminal and administrative penalties, damages, monetary fines, disgorgement, integrity oversight and reporting obligations, possible exclusion from participation in Medicare, Medicaid and other federal healthcare
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 81
programs, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could have a material adverse effect on our ability to operate our business and our results of operations.
We could incur significant liability if it is determined that we are promoting any “off-label” uses of our products.
Although our products are marketed for the specific therapeutic uses for which the devices were designed and our personnel are trained not to promote our products for uses outside of the FDA-cleared or authorized indications for use, known as “off-label uses,” we cannot, however, prevent a physician from using our products in ways, when in the physician’s independent professional medical judgment, he or she deems it appropriate. There may be increased risk of harm to patients if primary care physicians attempt to use our products off-label. Furthermore, theThe use of our products for indications other than those authorized, cleared, or approved by the FDA or authorized by any foreign regulatory body may not effectively treat such conditions, which could harm our reputation in the marketplace among primary care physicians and patients.
If the FDA or any foreign regulatory body determines that our promotional materials or training constitute promotion of an off-label use, it could request that we modify our training or promotional materials or subject us to regulatory or enforcement actions, including the issuance or imposition of an untitled letter or warning letter, injunction, seizure, civil fine or criminal penalties. It is also possible that other federal, state or foreign enforcement authorities might take action under other regulatory authority, such as false claims laws, based on our off-label promotion having caused submission of false (non-reimbursable) claims, for any products for which we obtain government reimbursement, if they consider our business activities to constitute promotion of an off-label use, which could result in significant penalties, including, but not limited to, criminal, civil and administrative penalties, damages, fines, disgorgement, exclusion from participation in government healthcare programs and the curtailment of our operations. In addition, certain jurisdictions have “all payor” false claims act laws that extend penalties for false claims beyond those submitted to government programs.
In addition, physicians may misuse our products with their patients if they are not adequately trained, potentially leading to injury and an increased risk of product liability. If our products are misused, we may become subject to costly litigation by our patients or their patients. As described above, product liability claims could divert management’s attention from our core business, be expensive to defend and result in sizeable damage awards against us that may not be covered by insurance.
We face potential product liability exposure, and, if claims brought against us are successful, we could incur substantial liabilities.
Our business exposes us to potential product liability claims that are inherent in the design, manufacture, testing and sale of medical devices. We could become the subject of product liability lawsuits alleging that component failures, manufacturing flaws, design defects or inadequate disclosure of product-related risks or product-related information resulted in an unsafe condition, injury or death to patients. In addition, thephysicians may misuse our products with their patients if they are not adequately trained, potentially leading to injury and an increased risk of product liability. The misuse of our products, or the failure of physicians or patients to adhere to operating guidelines, could cause significant harm to patients, including death, which could result in product liability claims. Product liability lawsuits and claims, safety alerts or product recalls, with or without merit, could cause us to incur substantial costs, and could place a significant strain on our financial resources, divert the attention of management from our core business, harm our reputation and materially and adversely affect our ability to attract and retain patients, any of which could have a material adverse effect on our business, financial condition and results of operations.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 79
Although we maintain third-party product liability insurance coverage, it is possible that claims against us may exceed the coverage limits of our insurance policies. Even if any product liability loss is covered by an insurance policy, these policies typically have substantial deductibles for which we are responsible. Product liability claims in excess of applicable insurance coverage could have a material adverse effect on our business, financial condition and results of operations. In addition, any product liability claim brought against us, with or without merit, could result in an increase of our product liability insurance premiums. Insurance coverage varies in cost and can be difficult to obtain, and we cannot guarantee that we will be able to obtain insurance coverage in the future on terms acceptable to us or at all.
Additionally, from time to time we may enter into agreements pursuant to which we indemnify third parties for certain claims relating to our products. These indemnification obligations may require us to pay significant sums of money for claims that are covered by these indemnification obligations. We are not currently subject to any product liability claims; however, any future product liability claims against us, regardless of their merit, may result
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 82
in negative publicity about us that could ultimately harm our reputation and could have a material adverse effect on our business, financial condition and results of operations.
Healthcare reform and other governmental and private payor initiatives may have an adverse effect upon, and could prevent, our products’ or product candidates’ commercial success.
In the US and in certain foreign jurisdictions, there have been a number of legislative and regulatory changes to the healthcare system that could impact our ability to sell our products profitably, such as the ACA.
Since its enactment, there have been numerous judicial, administrative, executive, and legislative challenges to certain aspects of the ACA, and we expect there will be additional challenges and amendments to the ACA in the future. For example, various portions of the ACA are currently undergoing legal and constitutional challenges in the US Supreme Court. Additionally, the former Trump administration issued various Executive Orders which eliminated cost-sharing subsidies and various provisions that would impose a fiscal burden on states or a cost, fee, tax, penalty or regulatory burden on individuals, healthcare providers, health insurers, or manufacturers of pharmaceuticals or medical devices and Congress has introduced several pieces of legislation aimed at significantly revising or repealing the ACA. Further, on December 20, 2019, the Further Consolidated Appropriations Act (H.R. 1865), which repeals the Cadillac tax, the health insurance provider tax, and the medical device excise tax, was signed into law. It is unclear whether the ACA will be overturned, repealed, replaced, or further amended. We cannot predict what affect further changes to the ACA would have on our business, especially under the Biden administration.
Other legislative changes have been proposed and adopted in the US since the ACA was enacted. In August 2011, the Budget Control Act of 2011, among other things, created measures for spending reductions by Congress. A Joint Select Committee on Deficit Reduction, tasked with recommending a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, was unable to reach required goals, thereby triggering the legislation’s automatic reduction to several government programs. This includes aggregate reductions of Medicare payments to providers up to 2% per fiscal year, and, due to subsequent legislative amendments, will remain in effect through 2030 unless additional congressional action is taken. Pursuant to the Coronavirus Aid, Relief, and Economic Security Act (the “CARES Act”), as well as subsequent legislation, these reductions were suspended from May 1, 2020 through March 31, 2021 due to the COVID-19 pandemic2022. Sequestration resumed at 1% for dates between April 1, 2022 and June 30, 2022, and then increased to the pre-pandemic level of 2% for dates after July 1, 2022. Proposed legislation, if passed, would extend thisintroduce another suspension until the end of the pandemic.
There has been increasing legislative and enforcement interest in the US with respect to prescription-pricing practices. Specifically, there have been several recent US Congressional inquiries and proposed federal and state legislation designed to, among other things, bring more transparency to drug pricing, reduce the cost of prescription drugs under Medicare, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drugs. The HHS has already started the process of soliciting feedback on some of these measures and, at the same time, is immediately implementing others under its existing authority. It is unclear what effect such legislative and enforcement interest may have on prescription devices. Further, it is unclear whether the Biden administration will challenge, reverse, revoke or otherwise modify the prior administration’s executive and administrative actions.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 80
We expect that these and other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we receive for any cleared, authorized, or approved device, which could have an adverse effect on patients for our products or product candidates. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors.
There have been, and likely will continue to be, legislative and regulatory proposals at the foreign, federal and state levels in the US directed at broadening the availability of healthcare and containing or lowering the cost of healthcare. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our products. Such reforms could have an adverse effect on anticipated revenue from product candidates that we may successfully develop and for which we may obtain
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 83
regulatory clearance, authorization, or approval and that may affect our overall financial condition and ability to develop product candidates. If we or any third parties we may engage are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we or such third parties are not able to maintain regulatory compliance, our current or any future product candidates we may develop may lose any regulatory clearance, authorization, or approval that may have been obtained and we may not achieve or sustain profitability.
Noncompliance with billing and documentation requirements could result in non-payment or subject us to billing or other compliance investigations by government authorities or private insurers.
Payors typically have differing and complex billing and documentation requirements. If we fail to comply with these payor-specific requirements, we may not be paid for our services or payment may be substantially delayed or reduced. Moreover, federal and state laws, rules and regulations impose substantial penalties, including criminal and civil fines, monetary penalties, exclusion from participation in government healthcare programs and imprisonment, on entities or individuals (including any individual corporate officers or physicians deemed responsible) that fraudulently or wrongfully bill government-funded programs or other third-party payors for healthcare services.
From time to time in the ordinary course of business, governmental agencies and private insurers also conduct audits of healthcare companies like us. Such audits could result in the incurrence of additional costs and diversion of management’s time and attention. In addition, such audits could trigger repayment demands based on findings that our products were billed in a manner that violated applicable billing requirements. We cannot predict whether any future audits, inquiries or investigations, or the public disclosure of such matters, likely would negatively impact our business, financial condition, results of operations, cash flows and the trading price of our securities.
We are subject to data privacy and security laws and regulations governing our collection, use, disclosure, or storage of personally identifiable information, including protected health information and payment card data, which may impose restrictions on us and our operations. Any actual or perceived noncompliance with such laws and regulations may result in penalties, regulatory action, loss of business or unfavorable publicity.
Numerous federal and state laws and regulations govern the collection, use, disclosure, storage and transmission of personally identifiable information (“PII”), including PHI, and information related to treatment for substance use disorders. These laws and regulations, including their interpretation by governmental agencies, are subject to frequent change and could have a negative impact on our business. In addition, in the future, industry requirements or guidance, contractual obligations, and/or legislation at both the federal and the state level may limit, forbid or regulate the use or transmission of health information outside of the US.
These varying interpretations can create complex compliance issues for us and our partners and potentially expose us to additional expense, adverse publicity and liability, any of which could materially and adversely affect our business.
Federal and state consumer protection laws are increasingly being applied by the FTC and states’ attorneys general to regulate the collection, use, storage and disclosure of PII, through websites or otherwise, and to regulate the presentation of website content. In addition, other laws, such as the Confidentiality of Substance Use Disorder Patient Records regulations at 42 C.F.R. Part 2, limit the potential use of in substance use disorder treatment-related data in non-treatment-based setting, such as administrative or criminal hearings related to the patient, and include associated restrictions on disclosure of information.
The security measures that we and our third-party vendors and subcontractors have in place to ensure compliance with privacy and data protection laws may not protect our facilities and systems from security breaches, acts of vandalism or theft, computer viruses, misplaced or lost data, programming and human errors or other similar events. Even though we provide for appropriate protections through our agreements with our third-party vendors, we still have limited control over their actions and practices. A breach of privacy or security of PII or PHI may result in an enforcement action, including criminal and civil liability, against us. We are not able to predict the extent of the impact such incidents may have on our business. Enforcement actions against us could be costly and could interrupt regular operations, which may materially and adversely affect our business. While we have not received
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 84
any notices of violation of the applicable privacy and data protection laws and believe we are in compliance with such laws, there can be no assurance that we will not receive such notices in the future.
There is ongoing concern from privacy advocates, regulators and others regarding data privacy and security issues, and the number of jurisdictions with data privacy and security laws has been increasing. Also, there are ongoing public policy discussions regarding whether the standards for de-identification, anonymization or
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 81
, or pseudonymization of health information are sufficient, and the risk of re-identification sufficiently small, to adequately protect patient privacy. We expect there will continue to be new proposed and amended laws, regulations and industry standards concerning privacy, data protection and information security in the US, such as the California Consumer Privacy Act (“CCPA”), which went into effect on January 1, 2020 and has been amended several times. Further, a new California privacy law, the California Privacy Rights Act (“CPRA”), was passed by California voters on November 3, 2020, and in March 2021, Virginia passed a new privacy law, the Consumer Data Protection Act (“VCDPA”), similar to the CPRA. The CPRA will create additional obligations with respect to processing and storing personal information and the VCDPA is scheduled to take effect on January 1, 2023 (with certain provisions of the CPRA having retroactive effect to January 1, 2022). In addition New York’s Stop Hacks and Improve Electronic Data Security Act (“SHIELD Act”), requires any person or business owning or licensing computerized data that includes the private information of a resident of New York to implement and maintain reasonable safeguards to protect the security, confidentiality and integrity of the private information. Other U.S., and has been amended several times. Other US states also are considering omnibus privacy legislation and industry organizations regularly adopt and advocate for new standards in these areas. While the CCPA and CPRA containcontains exceptions for certain activities involving PHI under HIPAA, we cannot yet determine the impact the CCPA, CPRA, VCDPA or other such future laws, regulations and standards may have on our business.
Future laws, regulations, standards, obligations, amendments, and changes in the interpretation of existing laws, regulations, standards and obligations could impair our or our customers’ ability to collect, use or disclose information relating to patients or consumers, including information derived therefrom, which could decrease demand for our products, increase our costs and impair our ability to maintain and grow our customer base and increase our revenue. Accordingly, we may find it necessary or desirable to fundamentally change our business activities and practices or to expend significant resources to modify our software or platform and otherwise adapt to these changes.
Further, our patients may expect us to comply with more stringent privacy and data security requirements than those imposed by laws, regulations, or self-regulatory requirements, and we may be obligated contractually to comply with additional or different standards relating to our handling or protection of data.
Any failure or perceived failure by us to comply with federal or state laws or regulations, industry standards or other legal obligations, or any actual or suspected privacy or security incident, whether or not resulting in unauthorized access to, or acquisition, release or transfer of PII or other data, may result in governmental enforcement actions and prosecutions, private litigation, fines and penalties or adverse publicity and could cause our customers to lose trust in us, which could have an adverse effect on our reputation and business. We may be unable to make such changes and modifications in a commercially reasonable manner or at all, and our ability to develop new products could be limited. Any of these developments could harm our business, financial condition and results of operations. Privacy and data security concerns, whether valid or not valid, may inhibit retention of our products by existing customer or adoption of our products by new customers.
Security breaches, ransomware attacks and other disruptions to our information technology structure could compromise our information, disrupt itsour business, and expose us to significant liability, which would cause our business and reputation to suffer and we may be unable to maintain and scale theour technology underlying our offerings.
In the ordinary course of our business, we collect, store, use and disclose sensitive data, including PHI and other types of PII. We also process and store, and use additional third parties to process and store, sensitive information including intellectual property and other proprietary business information, including that of our patients. Patient information is encrypted but not always de-identified. We manage and maintain our platform and data utilizing a combination of managed data center systems and cloud-based computing center systems.
We are highly dependent on information technology networks and systems, including the internet, to securely process, transmit and store this critical information. Security breaches of this infrastructure, including physical or electronic break-ins, computer viruses, attacks by hackers and similar breaches, and employee or contractor error, negligence or malfeasance, can create system disruptions, shutdowns or unauthorized disclosure or modifications of confidential information, causing member PHI and other PII to be accessed or acquired without authorization or
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 82
to become publicly available. We utilize third-party service providers for important aspects of the collection, storage and transmission of customer, user and patient information, and other confidential and sensitive information, and therefore rely on third parties to manage functions that have material cybersecurity risks.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 85
Because of the sensitivity of the PHI, other PII, and other confidential information we and our service providers collect, store, transmit, and otherwise process, the security of our technology platform and other aspects of our services, including those provided or facilitated by our third-party service providers, are important to our operations and business strategy. We take certain administrative, physical and technological safeguards to address these risks, such as by requiring outsourcing subcontractors who handle customer, user and patient information for us to enter into agreements that contractually obligate those subcontractors to use reasonable efforts to safeguard PHI, other PII, and other sensitive information. Measures taken to protect our systems, those of our subcontractors, or the PHI, other PII, or other sensitive data we or our subcontractors process or maintain, may not adequately protect us from the risks associated with the collection, storage and transmission of such information. Although we take steps to help protect confidential and other sensitive information from unauthorized access or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers or viruses, failures or breaches due to third-party action, employee negligence or error, malfeasance or other disruptions.
A security breach or privacy violation that leads to disclosure or unauthorized use or modification of, or that prevents access to or otherwise impacts the confidentiality, security, or integrity of, member information, including PHI or other PII, or other sensitive information we or our subcontractors maintain or otherwise process, could harm our reputation, compel us to comply with breach notification laws, cause us to incur significant costs for remediation, fines, penalties, notification to individuals and for measures intended to repair or replace systems or technology and to prevent future occurrences, potential increases in insurance premiums, and require us to verify the accuracy of database contents, resulting in increased costs or loss of revenue. If we are unable to prevent such security breaches or privacy violations or implement satisfactory remedial measures, or if it is perceived that we have been unable to do so, our operations could be disrupted, we may be unable to provide access to our platform, and could suffer a loss of customers or users or a decrease in the use of our platform, and we may suffer loss of reputation, adverse impacts on customer, user and investor confidence, financial loss, governmental investigations or other actions, regulatory or contractual penalties, and other claims and liability. In addition, security breaches and other inappropriate access to, or acquisition or processing of, information can be difficult to detect, and any delay in identifying such incidents or in providing any notification of such incidents may lead to increased harm.
Any such breach or interruption of our systems, or those of any of our third-party information technology partners, could compromise our networks or data security processes and sensitive information could be inaccessible or could be accessed by unauthorized parties, publicly disclosed, lost or stolen. Any such interruption in access, improper access, disclosure or other loss of information could result in legal claims or proceedings, liability under laws and regulations that protect the privacy of member information or other personal information, such as HIPAA, the CCPA, other state data breach laws and regulatory penalties. Unauthorized access, loss or dissemination could also disrupt our operations, including our ability to perform our services, provide member assistance services, conduct research and development activities, collect, process, and prepare company financial information, provide information about our current and future solutions and engage in other user and clinician education and outreach efforts. Any such breach could also result in the compromise of our trade secrets and other proprietary information, which could materially and adversely affect our business and competitive position. While we maintain insurance covering certain security and privacy damages and claim expenses, we may not carry insurance or maintain coverage sufficient to compensate for all liability and in any event, insurance coverage would not address the reputational damage that could result from a security incident.
International privacy and data security concerns and laws could result in additional costs and may prevent us from successfully expanding our business internationally.
Internationally, virtually every major jurisdiction has established its own data security and privacy legal framework. For instance, in May 2018, the European General Data Protection Regulation, or GDPR, came into effect and established requirements applicable to the handling of personal data and may result in fines up to €20 million (approximately $22 million based on current exchange rates) or up to 4% of annual global revenue in the preceding
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 83
financial year, whichever is higher, and other administrative penalties. In many European jurisdictions enforcement actions and consequences for non-compliance are also rising21.4 million based on current exchange rates) or up to 4% of annual global revenue in the preceding financial year, whichever is higher, and other administrative penalties. In many European jurisdictions enforcement actions and consequences for non-compliance are also rising. Our approach with respect to the GDPR and other data protection legislation may be subject to further evaluation and change, our compliance measures may not be fully adequate and may require modification, we may expend significant time and cost in developing
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 86
and maintaining a privacy governance program, data transfer or localization mechanisms, or other processes or measures to comply with these legal frameworks when looking to expand or business outside of the US.
Jurisdictions outside of the US and Europe are also considering and/or enacting comprehensive data protection legislation. Cross-border data transfers and other future developments regarding local data residency could increase the cost and complexity of delivering our services in some markets and may lead to governmental enforcement actions, litigation, fines, and penalties or adverse publicity, which could adversely affect our business and financial position, could greatly increase our cost of providing our products and services, require significant changes to our operations, or even prevent us from offering certain services in specific jurisdictions.
Several Asian countries have constitutional protections for, or have adopted legislation protecting, individuals’ personal information. For instance, the Personal Information Protection Law (“PIPL”) of the People’s Republic of China (“PRC”), was adopted on August 20, 2021, and went into effect on November 1, 2021. The PIPL shares similarities with the GDPR, including extraterritorial application, data minimization, data localization, and purpose limitation requirements, and obligations to provide certain notices and rights to citizens of the PRC. The PIPL allows for fines of up to CN¥ 50 million (approximately $8 million based on current exchange rates) or 5% of a covered company’s revenue in the prior year. Our approach with respect to the GDPR, the PIPL, and other data protection legislation may be subject to further evaluation and change, our compliance measures may not be fully adequate and may require modification, we may expend significant time and cost in developing and maintaining a privacy governance program, data transfer or localization mechanisms, or other processes or measures to comply with these legal frameworks when looking to expand or business outside of the US.
We also continue to see jurisdictions imposing data localization laws, which may require personal information of citizens of a jurisdiction to be, among other data processing operations, initially collected, stored, and modified locally within such jurisdiction. These regulations may inhibit our ability to expand into those markets without significant additional costs.
Because of the breadth of these data protection laws and the narrowness of their exceptions and safe harbors, it is possible that international expansion could subject our business or data protection policies to challenge under one or more of such laws. The scope and enforcement of each of these laws is uncertain and subject to rapid change in the current environment of heightened regulatory focus on data privacy and security issues. If our operations are found to be in violation of any of the data protection laws described above or any other laws that apply to us, we may be subject to penalties, including, but not limited to, criminal, civil and administrative penalties, damages, fines, disgorgement, individual imprisonment, possible exclusion from participation in government healthcare programs, injunctions, private qui tam actions brought by individual whistleblowers in the name of the government, class action litigation and the curtailment or restructuring of our operations, as well as additional reporting obligations and oversight if we become subject to a corrective action plan or other agreement to resolve allegations of non-compliance with these laws, any of which could adversely affect our ability to operate our business and our results of operations.
Our PearConnect platform may not operate properly, which could damage our reputation, subject us to claims, or require us to divert application of our resources from other purposes, any of which could harm our business and growth.
Our PearConnect platform provides patients and physicians with the ability to, among other things, provide (i) access to the PearMD Clinician Dashboard; (ii) an end-to-end patient service center; and (iii) a data analytics system configured to aggregate patient engagement, adherence, and clinical outcome data for insight generation. Proprietary software development is time-consuming, expensive, complex, and may involve unforeseen difficulties. We may encounter technical obstacles, and it is possible that we may discover additional problems that prevent PearConnect from operating properly. We continue to implement software with respect to a number of new applications and services. The operation of our technology also depends in part on the performance of third-party service providers. If our technology platform does not function reliably or fails to achieve provider, partner or payor expectations in terms of performance, we may be required to divert resources allocated for other business purposes to address these issues, may suffer reputational harm, lose or fail to grow our customer base, and may be subject to liability claims.
Our patient service center uses text and voice calls to communicate with healthcare providers, patients and prospective patients, and we are subject to various marketing and advertising laws including the Telephone Consumer Protection Act of 1991 (“TCPA”). If we fail to comply with applicable laws, including the TCPA, we may be subject to significant liabilities.
Our patient service center uses short message service (“SMS”), text messages and telephone calls to communicate with healthcare providers, patients and prospective patients. The actual or perceived improper sending of text messages or the making of telephone calls may subject us to potential risks, including liabilities or claims relating to consumer protection laws. Numerous class-action suits under federal and state laws have been filed in recent years against companies who conduct SMS texting programs or make unwanted telephone calls, with many resulting in multi-million-dollar settlements to the plaintiffs. Any future such litigation against us could be costly and time-consuming to defend. For example, the Telephone Consumer Protections Act of 1991 (“TCPA”)
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 87
and time-consuming to defend. For example, the TCPA, a federal statute that protects consumers from unwanted telephone calls, faxes, and text messages, restricts telemarketing and the use of automated SMS text messages without proper consent. Additionally, state regulators may determine that telephone calls to our patients are subject to state telemarketing regulations. Federal or state regulatory authorities or private litigants may claim that the notices and disclosures we provide, form of consents
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 84
we obtain, or our SMS texting practices are not adequate or violate applicable law. This may in the future result in civil claims against us. The scope and interpretation of the laws that are or may be applicable to the delivery of text messages are continuously evolving and developing. If we do not comply with these laws or regulations or if we become liable under these laws or regulations, we could face direct liability, could be required to change some portions of our business model, could face negative publicity, and our business, financial condition, and results of operations could be materially and adversely affected. Even an unsuccessful challenge of our SMS texting or telephone calling practices by our customers, regulatory authorities, or other third parties could result in negative publicity and could require a costly response from and defense by us.
We may be subject to governmental investigation, litigation and other proceedings, including intellectual property disputes, which are costly to defend and could materially harm our business and results of operations.
We may be party to government investigations, lawsuits and legal proceedings in the normal course of business. These matters are often expensive and disruptive to normal business operations. We may face allegations, lawsuits and regulatory inquiries, audits and investigations regarding data privacy, security, labor and employment, consumer protection and intellectual property infringement, including claims related to privacy, patents, publicity, trademarks, copyrights and other rights. A portion of the technologies we use incorporates open source software, and we may face claims claiming ownership of open source software or patents related to that software, rights to our intellectual property or breach of open source license terms, including a demand to release material portions of our source code or otherwise seeking to enforce the terms of the applicable open source license. We may also face allegations or litigation related to our acquisitions, securities issuances or business practices, including public disclosures about our business. Litigation and regulatory proceedings, and particularly the patent infringement and class action matters we could face, may be protracted and expensive, and the results are difficult to predict. Certain of these matters may include speculative claims for substantial or indeterminate amounts of damages and include claims for injunctive relief. Additionally, our litigation costs could be significant. Adverse outcomes with respect to litigation or any of these legal proceedings may result in significant settlement costs or judgments, penalties and fines, or require us to modify our solution or require us to stop offering certain features, all of which could negatively impact our business. We may also become subject to periodic audits, which would likely increase our regulatory compliance costs and may require us to change our business practices, which could negatively impact our results of operations. Managing legal proceedings, litigation and audits, even if we achieve favorable outcomes, is time-consuming and diverts management’s attention from our business.
The results of regulatory proceedings, litigation, claims, and audits cannot be predicted with certainty, and determining reserves for pending litigation and other legal, regulatory and audit matters requires significant judgment. There can be no assurance that our expectations will prove correct, and even if these matters are resolved in our favor or without significant cash settlements, these matters, and the time and resources necessary to litigate or resolve them, could harm our reputation, business, financial condition, results of operations and the market price of our Class A common stock.
Our commercialization efforts to date have focused almost exclusively on the US. Our ability to enter other foreign markets will depend, among other things, on itsour ability to navigate various regulatory regimes with which itwe doesdo not have experience, which could delay or prevent the growth of our operations outside of the US.
To date, our commercialization efforts have focused almost exclusively on the US. Expanding our business to attract customers in countries other than the US is an element of our long-term business strategy. Our ability to continue to expand our business and to attract talented employees and customers in various international markets will require considerable management attention and resources and is subject to the particular challenges of supporting a rapidly growing business in an environment of multiple languages, cultures, customs, legal systems, alternative dispute resolution systems, regulatory systems and commercial infrastructures. Entering new international markets will be expensive, our ability to successfully gain market acceptance in any particular market
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 88
is uncertain and the distraction of our senior management team could harm our business, financial condition and results of operation.
Sales of our products outside of the US are subject to foreign regulatory requirements that vary widely from country to country. In addition, the FDA regulates exports of medical devices from the US. While the regulations of
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 85
some countries may not impose barriers to marketing and selling our products or only require notification, others require that we obtain the marketing authorization of a specified regulatory body. Complying with foreign regulatory requirements, including obtaining registrations or marketing authorizations, can be expensive and time-consuming, and we may not receive regulatory authorizations, clearances or approvals in each country in which we may plan to market our products or we may be unable to do so on a timely basis. The time required to obtain registrations or marketing authorizations, if required by other countries, may be longer than that required for FDA clearance, authorization, or approval, and requirements for such registrations and marketing authorizations may significantly differ from FDA requirements. If we modify our products, we may need to apply for additional regulatory authorizations before we are permitted to sell the modified product. In addition, we may not continue to meet the quality and safety standards required to maintain the authorizations that we have received. If we are unable to maintain our authorizations in a particular country, we will no longer be able to sell the applicable product in that country. Regulatory de novo classification, clearance or approval by the FDA does not ensure registration or marketing authorization by regulatory authorities in other countries, and registration or marketing authorization by one or more foreign regulatory authorities does not ensure registration or marketing authorization by regulatory authorities in other foreign countries or by the FDA. A failure or delay in obtaining registration or marketing authorization in one country may have a negative effect on the regulatory process in others.
Doing business internationally involves a number of additional risks, including:
•multiple, conflicting and changing laws and regulations such as tax laws, privacy and data protection laws and regulations, export and import restrictions, employment laws, regulatory requirements and other governmental approvals, permits and licenses;
•requirements to maintain data and the processing of that data on servers located within the US or in such countries;
•protecting and enforcing our intellectual property rights;
•converting our products as well as the accompanying instructional and marketing materials to conform to the language and customs of different countries;
•complexities associated with managing multiple payor reimbursement regimes, and government payors;
•competition from companies with significant market share in our market and with a better understanding of user preferences;
•financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the effect of local and regional financial pressures on demand and payment for our products and services and exposure to foreign currency exchange rate fluctuations;
•natural disasters, political and economic instability, including wars, terrorism, political unrest, outbreak of disease (including the recent coronavirus outbreak), boycotts, curtailment of trade, and other market restrictions; and
•regulatory and compliance risks that relate to maintaining accurate information and control over activities subject to regulation under the US Foreign Corrupt Practices Act (the “FCPA”), and comparable laws and regulations in other countries.
These risks and uncertainties may impact our ability to enter foreign markets, which could delay or prevent the growth of our operations outside of the US, and have a material adverse effect on our business, prospects, results of operations and financial condition.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 86
The regulatory framework for digital health products is constantly evolving. Increasingly stringent regulatory requirements could create barriers to our development and introduction of new products. Conversely, in the event that regulatory requirements are lowered, competitors could potentially enter the prescription digital therapeutic market and compete with us more easily. Either of the foregoing could materially harm our business.
Our PDTs are novel and represent a new category of therapeutics for which the regulatory framework continues to evolve. Our ability to develop and introduce new products will depend, in part, on our ability to comply with these complex requirements, which include regulations related to product design, development and manufacturing; testing, labeling, content and language of instructions for use; clinical trials; product safety; pre market clearance, authorization, and approval; establishment registration and device listing; and marketing, sales and distribution. If, however, the regulatory framework for digital health products simplifies and the requirements that we and others are required to comply with are lowered, it could result in the increased competition and the introduction by competitors of products that are or claim to be superior to our products. For example, due to the COVID-19 public health emergency, the FDA issued, “Enforcement Policy for Digital Health Devices For Treating Psychiatric Disorders During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency,” which allows for marketing of certain digital therapeutics without premarket clearance, authorization, or approval so long as certain criteria are met. FDA is considering their treatment of digital health devices once the COVID-19 emergency declarations
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 89
end. Actions could include the requirement to submit documentation to obtain FDA authorization, reduced documentation to obtain authorization based on market experience, or market withdrawal. A number of these actions could adversely affect our business. Similarly, competitors using our products as predicates for 510(k)s may successfully argue that they should be required to submit substantially less data to support approval of their product than was required for our products based on FDA’s growing familiarity with the technology. As a result, we are subject to risks related to the developing regulatory landscape applicable to our PDTs that could have a material adverse effect on our business and results of operations.
Our products may face competition from digital health products that are marketed without regulatory clearance, authorization, or approval. Regulators have broad discretion in determining whether to enforce regulatory requirements, and may decide not to remove uncleared or unapproved products that compete with our products, which could materially and adversely impact our business.
Our PDTs, reSET, reSET-O and Somryst, have been authorized or cleared by the FDA after completion of clinical trials and related regulatory review. The FDA and other regulators have broad discretion in determining whether to enforce these requirements, however, which could result in uncleared or unapproved products entering the marketplace. If uncleared or unapproved products are allowed to compete with our products, we will face increased competition from parties who have fewer barriers to enter our industry. This increased competition could have a material, adverse effect on our business, results of operations and financial condition.
Premarket clearances, authorizations, and approvals for new or significantly modified devices could be denied or significantly delayed.
Under FDA regulations, unless exempt, a new medical device may only be commercially distributed after it has received 510(k) clearance, is authorized through the de novo classification process, or is the subject of a premarket approval (“PMA”). The FDA will clear marketing of a medical device through the 510(k) process if it is demonstrated that the new product is substantially equivalent to another legally marketed product not subject to a PMA. Sometimes, premarket submissions must be supported by clinical data. Our ability to enroll patients in clinical trials could be impacted by the COVID-19 outbreak, as many patients are electing or being asked to delay procedures at this time. The PMA process typically is more costly, lengthy and stringent than the 510(k) process and usually requires more substantial clinical studies.
The FDA may not authorize marketing via de novo classification or clear our 510(k) applications on a timely basis or at all. For example, the COVID-19 outbreak could affect the FDA’s ability to review applications or supplements. Such delays or refusals, regardless of the cause, could have a material adverse effect on our business, financial condition, and results of operations. The FDA may also change its clearance and authorization policies, adopt additional regulations or revise existing regulations, or take other actions which may prevent or delay authorization or clearance of our products.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 87
Material modifications to our devices may require new 510(k) clearance, de novo classification, PMA approval, or PMA supplement approval, or may require us to cease marketing or recall the modified devices until clearances, authorizations, or approvals are obtained.
Material modifications to the intended use or technological characteristics of our devices may require new 510(k) clearance, de novo classification, PMA approval, or PMA supplement approval, or may require us to cease marketing or recall the modified devices until clearances, authorizations, or approvals are obtained. Any modification to a 510(k)-cleared device that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, design, or manufacture, requires a new 510(k) clearance or, possibly, a de novo or a PMA. The FDA requires every manufacturer to make and document this determination in the first instance. A manufacturer may determine that a modification could not significantly affect safety or effectiveness and does not represent a major change in its intended use, so that no new 510(k) clearance is necessary. The FDA may review any manufacturer’s decision and may not agree with our decisions regarding whether new clearances, authorization, or approvals are necessary. The FDA may also on its own initiative determine that a new clearance, authorization, or approval is required.
We have modified some of our cleared and authorized devices and have determined based on our review and interpretation of the applicable FDA guidance that in certain instances new 510(k) clearances are not required. If
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 90
the FDA disagrees with our determination and requires us to submit new 510(k) clearances for modifications to our previously cleared or authorized products for which we have concluded that new clearances are unnecessary, we may be required to cease marketing or to recall the modified product until we obtain clearance, authorization, or approval. In these circumstances, we may be subject to significant enforcement actions, regulatory fines, or penalties, which could require us to redesign our products and harm our operating results. In addition, unlike traditional hardware devices, we are exposed to this risk more frequently based on the number of changes associated with software to improve performance, introduce new features, and correct issues.
Products may be subject to product recalls. A recall of our products, either voluntarily or at the direction of the FDA or another governmental authority, or the discovery of serious safety issues with our products, could materially and adversely affect us.
The FDA and similar foreign governmental authorities have the authority to require the recall of commercialized products in the event of material deficiencies or defects in their design or manufacture or in the event that a product poses an unacceptable risk to health.
The FDA’s authority to require a recall for medical devices must be based on a finding that there is reasonable probability that the device would cause serious injury or death. We may also decide to voluntarily recall our products. A government-mandated or voluntary recall could occur as a result of an unacceptable risk to health, component failures, malfunctions, manufacturing errors, design or labeling defects or other deficiencies and issues. For example., on May 20, 2021, we initiated a voluntary correction of reSET and reSET-O due to a software defect related to contingency management. This recall was reportable to the FDA and is in-process as of the date of this filing. Recalls of any of our products would divert managerial and financial resources and could materially and adversely affect our reputation and business, which could impair our ability to produce our products in a cost-effective and timely manner in order to meet our customers’ demands. We may also be subject to liability claims, be required to bear other costs, or take other actions that could materially and adversely affect our business, results of operations and financial condition.
Companies are required to maintain certain records of recalls and corrections, even if they are not reportable to the FDA. We may initiate voluntary recalls or corrections for our products in the future that we determine do not require notification of the FDA. If the FDA disagrees with our determinations, they could require us to report those actions as recalls and we may be subject to enforcement action.
We are required to report certain malfunctions, deaths, and serious injuries associated with our products, which can result in voluntary corrective actions or agency enforcement actions.
Under the FDA’s medical device reporting (“MDR”) regulations, we are required to report to the FDA when information from any source suggests that our product may have caused or contributed to a death or serious injury or that our
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 88
product has malfunctioned and, if the malfunction were to recur, would likely cause or contribute to death or serious injury. We have notproduct has malfunctioned and, if the malfunction were to recur, would likely cause or contribute to death or serious injury. In November 2022, been required towe reported our makefirst any such reports to dateMDR incident in which a patient suffered a relapse. If we fail to report these events to the FDA within the required timeframes, or at all, the FDA could take enforcement action against us.
Any adverse event involving our products, whether in the US or abroad, could result in future voluntary corrective actions, such as recalls, including corrections or customer notifications, or agency action, such as inspection or enforcement actions. If malfunctions do occur, we may be unable to correct the malfunctions adequately or prevent further malfunctions, in which case we may need to cease manufacture and distribution of the affected products, initiate voluntary recalls, and redesign the products. Regulatory authorities may also take actions against us, such as ordering recalls, imposing fines, or seizing the affected products. Any corrective action, whether voluntary or involuntary, will require the dedication of our time and capital, distract management from operating our business, and may harm our reputation and financial results.
If we fail to comply with the FDA’s Quality System Regulation (“QSR”), or any applicable foreign equivalent, our operations could be interrupted, and our potential product sales and operating results could suffer.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 91
We are required to comply with the FDA’s QSR, which delineates, among other things, the design controls, document controls, purchasing controls, identification and traceability, production and process controls, acceptance activities, nonconforming product requirements, corrective and preventive action requirements, labeling and packaging controls, handling, storage, distribution and installation requirements, complaint handling, records requirements, servicing requirements, and statistical techniques potentially applicable to the production of our medical devices. We are also subject to the regulations of foreign jurisdictions if we market products overseas.
The FDA enforces the QSR through periodic and announced or unannounced inspections of manufacturing facilities. OurBoth our San Francisco facility hasand Boston facilities have been inspected by the FDA and other designated auditing organizations, and we anticipate that we will be subject to additional future inspections. If our facilities or processes are found to be in non-compliance or fail to take satisfactory corrective action in response to adverse QSR inspectional findings, the FDA could take legal or regulatory enforcement actions against us and/or our products, including but not limited to the cessation of sales or the initiation of a recall of distributed products, which could impair our ability to produce our products in a cost-effective and timely manner in order to meet our customers’ demands. We may also be required to bear other costs or take other actions that may have a negative impact on our future sales and our ability to generate profits.
The FDA’s, other comparable state governmental agencies’, and non-US regulatory agencies’ statutes, regulations, policies or interpretations may change, and additional government regulation or statutes may be enacted, which could increase regulatory requirements, or delay, suspend, prevent marketing of any cleared, authorized, or approved products or necessitate the recall of distributed products. We cannot predict the likelihood, nature or extent of adverse governmental regulation that might arise from future legislative or administrative action, either in the US or abroad.
The medical device industry has been under heightened FDA scrutiny as the subject of government investigations and enforcement actions. If our operations and activities are found to be in violation of any FDA lawsregulations or any other governmental regulations that apply to us, we may be subject to penalties, including civil and criminal penalties, damages, fines and other legal and/or agency enforcement actions. Any penalties, damages, fines, or curtailment or restructuring of our operations or activities could materially and adversely affect our ability to operate our business and our financial results. The risk of us being found in violation of FDA lawsregulations is increased by the fact that many of these lawsregulations are broad and their provisions are open to a variety of interpretations. Any action against us for violation of these lawsregulations, even if we successfully defend ourselves against that action and its underlying allegations, could cause us to incur significant legal expenses and divert management’s attention from the operation of our business. Where there is a dispute with a federal or state governmental agency that cannot be resolved to the mutual satisfaction of all relevant parties, we may determine that the costs, both real and contingent, are not justified by the commercial returns to us from maintaining the dispute or the product.
Various claims, design features, or performance characteristics of our medical devices that we regarded as permitted by the FDA without new marketing clearance, authorization, or approval may be challenged by the FDA
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 89
products that we regarded as permitted by the FDA without new marketing clearance, authorization, or approval may be challenged by the FDA or state or foreign regulators. The FDA or state or foreign regulatory authorities may find that certain claims, design features, or performance characteristics, in order to be made or included in the products, may have to be supported by further clinical studies and marketing clearances, authorizations, or approvals, which could be lengthy, costly, and possibly unobtainable.
Risks Related to Our Financial Reporting
We rely on assumptions, estimates, internally developed software, and data from third parties to calculate our key performance indicators and other business metrics to deliver timely and accurate information in order to accurately report our financial results in the timeframe and manner required by law, and real or perceived inaccuracies in these metrics may harm our reputation and negatively affect our business.
We need to receive timely, accurate, and complete information from our internal company data that has not been independently verified utilizing internally developed software and third party software in order to accurately report our financial results on a timely basis. If the information that we receive is not accurate, our consolidated
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 92
financial statements may be materially incorrect and may require restatement. While these numbers are based on what we believe to be reasonable calculations for the applicable period of measurement, there are inherent challenges in measuring such information. In addition, our measurement of certain metrics may differ from estimates published by third parties or from similarly-titled metrics of our competitors due to differences in methodology and as a result our results may not be comparable to our competitors. As a result, we may have difficulty completing accurate and timely financial disclosures, which could have an adverse effect on our business.
Our results of operations and financial condition are subject to management’s accounting judgments and estimates, as well as changes in accounting policies.
The preparation of our financial statements requires us to make estimates and assumptions affecting the reported amounts of our assets, liabilities, revenues, expenses and earnings. If these estimates or assumptions are incorrect, it could have a material adverse effect on our results of operations or financial condition. We have identified several accounting policies as being critical to the fair presentation of our financial condition and results of operations because they involve major aspects of our business and require us to make judgments about matters that are inherently uncertain. These policies are described under the section entitled “Management’s Discussion and Analysis of Financial Conditions and Results of Operations” and should be considered in conjunction with our audited consolidated financial statements and notes thereto included in Part II, Item 8, Financial Statements, of this Form 10-K. The implementation of new accounting requirements or other changes to Generally Accepted Accounting Principles in the US, or GAAP, could have a material adverse effect on our reported results of operations and financial condition.
As a public reporting company, we will beare subject to rules and regulations established from time to time by the SEC regarding our internal control over financial reporting. If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, investors may lose confidence in the accuracy of our financial reports, which would harm our business and the trading price of our common stock. Our management will beis required to evaluate the effectiveness of our internal control over financial reporting.
As a public reporting company, we are subject to the reporting requirements of the Exchange Act, the Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley Act”), the Dodd-Frank Wall Street Reform and Consumer Protection Act, and the rules and regulations established by the SEC and Nasdaq. These rules and regulations require, among other things, that we establish and periodically evaluate procedures with respect to our internal control over financial reporting. Reporting obligations as a public company place a considerable strain on our financial and management systems, processes and controls, as well as on our personnel, including senior management. In addition, as a public company, we are required to document and test our internal control over financial reporting pursuant to Section 404 of the Sarbanes-Oxley Act so that our management can certify as to the effectiveness of our internal control over financial reporting. Management’s initial certification under Section 404 of the Sarbanes-Oxley Act will be required withis included in Part our annual report on Form 10-K for the year ending December 31, 2022.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 90
II, Item 9A, Controls and Procedures, of this Form 10-K. In support of such certifications, we have and will be required to documentdocumented and make significant changes and enhancements, including potentially hiring additional personnel, to improve our internal control over financial reporting. Likewise, our independent registered public accounting firm will not be required to attest to the effectiveness of our internal control over financial reporting until our first annual report is required to be filed with the SEC following the date we are no longer an emerging growth company.
To achieve compliance with Section 404 within the prescribed period, and in order to maintain and improve the effectiveness of our internal controls over financial reporting, we have expended, and anticipate we will continue to expend, significant resources, including accounting-related costs and significant management oversight. If any of these new or improved controls and systems do not perform as expected, we may experience further deficiencies in our controls.
We have identified gaps in our internal control environment in the past and cannot provide assurances that there will not be material weaknesses or significant deficiencies in our internal control over financial reporting in the future. Any failure to maintain internal control over financial reporting could severely inhibit our ability to
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 93
accurately report our financial condition or results of operations. If we are unable to conclude that our internal control over financial reporting is effective or if our independent registered public accounting firm determines we have a material weakness or significant deficiency in our internal control over financial reporting, we could lose investor confidence in the accuracy and completeness of our financial reports, the market price of shares of our common stock could decline, and we could be subject to sanctions or investigations by Nasdaq, the SEC, or other regulatory authorities. Failure to remedy any material weakness in our internal control over financial reporting, or to implement or maintain other effective control systems required of public companies, could also restrict our future access to the capital markets.
Our management has identified certain internal control deficiencies that constitute material weaknesses. If we fail to maintain an effective system of disclosure controls and internal control over financial reporting, our ability to produce timely and accurate financial statements or comply with applicable regulations could be impaired.
We have identified material weaknesses in our internal control over financial reporting. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements will not be prevented or detected on a timely basis. If we are unable to remediate these material weaknesses, or if we identify additional material weaknesses in the future or otherwise fail to maintain an effective system of internal controls, we may not be able to accurately or timely report our financial condition or results of operations, which may adversely affect investor confidence in us and, as a result, our stock price.
As of December 31, 2021 and 20202022, we did not design or maintain an effective control environment as we did not maintain a sufficient complement of accounting and financial reporting resources commensurate with our financial reporting requirements. This resulted in the following material weaknesses:
•we did not have sufficient accounting and reporting resources to ensure adequate segregation of duties,
•we did not design, implement,As a result, we did not design, implement and maintain adequate information systems controls, including access and change management controls, and
•we did not design, implement, and maintain controls to ensure the accurate and timely reporting of material effective control activities to ensure the accurate and timely reporting of transactions, including the capitalization ofrevenue and capitalized software costs and capital stock valuations performed by us or our advisorstransactions.
These control deficiencies could result in a misstatement in our accounts or disclosures that would result in a material misstatement to our financial statements that would not be prevented or detected. Accordingly, we determined that these control deficiencies constitute material weaknesses.
In addition, theyit could cause us to fail to meet our reporting obligations or may result in a restatement of our financial statements for prior periods. Any failure to implement and maintain effective internal control over financial reporting also could materially and adversely affect the results of periodic management evaluations and
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 91
annual independent registered public accounting firm attestation reports regarding the effectiveness of our internal control over financial reporting that we will eventually be required to include in our periodic reports that will be filed with the SEC.
Our management has concluded that these material weaknesses are due to the fact that, prior to the Business Combination, more fully described in Note 3 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K, we were a private company with limited resources. We did not have the necessary business processes and related internal controls, or the appropriate resources or level of experience and technical expertise, that would be required to oversee financial reporting processes or to address the accounting and financial reporting requirements. These material weaknesses will not be considered remediated until management designs and implements effective controls that operate for a sufficient period of time and management has concluded, through testing, that these controls are effective. Our management is in the process of developing a remediation plan, and we cannot assure you that the measures that we implement will fully address the material weaknesses and deficiencies in our internal control over financial reporting or that we may conclude that they have been fully remediated.
We are continuing to develop and refine our disclosure controls and other procedures that are designed to ensure information required to be disclosed by us in the reports we will file with the SEC is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms and that information required to be disclosed in reports under the Exchange Act is accumulated and communicated to our principal executive and financial officers. For example, we hired additional personnel in our finance group, including a Vice President, Corporate Controller and Director, SEC Reporting and Technical AccountingAny failure to develop or maintain effective controls or any difficulties encountered as a result of the implementation or improvements could harm our results of operations or cause us to fail to meet our reporting obligations and may result in a restatement of our financial statements for prior periods. Any failure to implement and maintain effective internal control over financial reporting also could materially and adversely affect the results of periodic management evaluations and annual independent registered public accounting firm attestation reports regarding the effectiveness of our internal control over financial reporting that we will eventually be required to include in our periodic reports that will be filed with the SEC.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 94
We are continuing to develop and refine our disclosure controls and other procedures that are designed to ensure information required to be disclosed by us in the reports we will file with the SEC is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms and that information required to be disclosed in reports under the Exchange Act is accumulated and communicated to our principal executive and financial officers. For example, we hired additional personnel in our finance group, including a Vice President, Chief Accounting Officer and staff with adequate US GAAP and SEC reporting experience to address complex US GAAP technical accounting issues and to prepare and review the financial statements and related disclosures in accordance with US GAAP and SEC reporting requirements. In addition, we implemented a new enterprise resource planning system to replace the former general ledger package. Any failure to develop or maintain effective controls or any difficulties encountered as a result of the implementation or improvements could harm our results of operations or cause us to fail to meet our reporting obligations and may result in a restatement of our financial statements for prior periods. Any failure to implement and maintain effective internal control over financial reporting also could materially and adversely affect the results of periodic management evaluations and annual independent registered public accounting firm attestation reports regarding the effectiveness of our internal control over financial reporting that we will eventually be required to include in our periodic reports that will be filed with the SEC.
The material weaknesses remain unremediated as of December 31, 2021As a former shell company, we will face certain disadvantages relative to companies that pursue a traditional initial public offering.
THMA was a special purpose acquisition company (“SPAC”), a form of shell company under the SEC rules. Shell companies are more highly regulated than non-shell operating companies and face significant additional restrictions on their activities under federal securities laws. As a result of the Business Combination, we ceased to be a shell company. However, companies that were formerly shell companies continue to face disadvantages under SEC rules, including (a) the inability to qualify as a “well-known seasoned issuer” and file automatically effective registration statements for three years after ceasing to be a shell company, (b) the inability to “incorporate by reference” information in certain registration statements filed under the Securities Act of 1933, as amended (the “Securities Act”) for a period of three years after ceasing to be a shell company, (c) the inability to use most free writing prospectuses until at least three years after a qualifying business combination, and (d) exclusion from certain safe harbors for offering-related communications under the Securities Act for three years after ceasing to be a shell company, including for research reports and certain communications in connection with business combinations. We expect that these disadvantages will make it more challenging and expensive, and create greater risks and delays, for both us and our stockholders to offer securities. These challenges may make our securities less attractive than those of companies that are not former shell companies and may raise our relative cost of capital.
Some members of our management team have limited experience in operating a public company.
Some of our executive officers have limited experience in the management of a publicly traded company. Our management team may not successfully or effectively manage our transition to a public company that will be subject to significant regulatory oversight and reporting obligations under federal securities laws. Certain executives’ limited experience in dealing with the increasingly complex laws pertaining to public companies could be a significant disadvantage as they will likely need to devote an increasing amount of their time to these activities, resulting in less time being devoted to the management and growth of our Companybusiness. We may not have adequate personnel with the appropriate level of knowledge, experience, and training in the accounting policies, practices or internal controls over financial reporting required of public companies.
We could be subject to additional tax liabilities and our ability to use our net operating loss carryforwards and other tax attributes may be limited.
We have incurred net operating losses (“NOLsNOLs”) since our inception and may never achieve or sustain profitability. Generally, for US federal income tax purposes, unused NOLs will carry forward. However, NOL carryforwards generated prior to January 1, 2018, are subject to expiration for US federal income tax purposes. As of December 31, 20212022, we had federal NOL carryforwards of approximately $218294.9 million, of which $17.4 million will begin to expire in 2034. In addition, under the Internal Revenue Code of 1986, as amended, or the Code, limits the ability to deduct NOL carryforward generated after December 31, 20176 million, of which $17.4 million will begin to expire in 2034. In addition, under the Internal Revenue Code of 1986, as amended, or the Code, limits the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 95
ability to deduct NOL carryforward generated after December 31, 2017, and all future NOL carryforwards to 80% of taxable income. These NOL limitations may limit or delay in part the use of NOL carryforwards, if or, when we cease operating at a loss. It is uncertain whether and to what extent applicable state tax laws will conform to this rule. As
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 92
of December of December 31, 20212022, we also had research and development tax credits of $68.23 million, which may be available to offset future income tax liabilities. The research and development tax credit carryforwards would begin to expire in 2037. As of December 31, 20212022, we also had state research and development tax credits of $1.39 million, which may be available to offset future income tax liabilities. The state research and development tax credit carryforwards would begin to expire in 2030.
In general, under Code Sections 382 and 383, if a corporation undergoes an “ownership change” (generally defined as a greater than 50% change by value in its equity ownership by certain stockholders over a three-year period), the corporation’s ability to use its pre-ownership change NOLs, carryforwards and other pre-ownership change tax attributes, such as research tax credits, to offset its post-ownership change income or taxes may be limited. Similar provisions of state tax law may also apply to limit the use of our state NOL carryforwards and other state tax attributes. We have not performed an analysis to determine whether our past issuances of stock and other changes in our stock ownership may have resulted in one or more “ownership changes” under these rules. In addition, future changes in our stock ownership may materially limit our ability to utilize our NOL carryforwards and other tax attributes under these rules. As a result, even if we earn net taxable income in the future, we may be unable to use a material portion of our NOL carryforwards and other tax attributes, which could materially and adversely affect our future cash flows.
There is also a risk that regulatory changes, such as suspensions on the use of NOL or other unforeseen reasons, may result in our existing NOL carryforwards expiring or otherwise becoming unavailable to offset future taxable income. For these reasons, we may not be able to utilize all or a material portion of our NOL carryforwards and other tax attributes, even if we attain profitability. For example, a temporary suspension of the use of certain net operating losses and tax credits has been enacted in California, and otherIllinois. Other states may enact suspensions as well. If we are limited in our ability to use our NOLs in future years in which we have taxable income, we will pay more taxes than if we were able to fully utilize our NOLs. This could materially and adversely affect our results of operations.
Risks Related to Ownership of our Class A common stock and Warrants
The terms of the Warrants may be amended in a manner adverse to a holder if holders of at least 50% of the then outstanding Public Warrants approve of such amendment.
The Private Placement Warrants and Public Warrants were issued in registered form under the Warrant Agreement, between us and Continental Stock Transfer & Trust Company, our WarrantThe rules dealing with US federal, state, and local income taxation are constantly under review by persons involved in the legislative process and by the Internal Revenue Service and the US Treasury Department. Changes to tax laws (which changes may have retroactive application) could adversely affect us or holders of our common stock. In recent years, many such changes have been made and changes are likely to continue to occur in the future. Future changes in tax laws could have a material adverse effect on our business, cash flow, financial condition or results of operations. We urge investors to consult with their legal and tax advisers regarding the implications of potential changes in tax laws on an investment in our common stock.
Risks Related to Ownership of our Class A common stock and Warrants
Our stock price is currently trading below $1.00 per share and, if it continues to trade below $1.00 per share, our Class A common stock may be subject to delisting from the Nasdaq Capital Market.
Our Class A common stock is currently trading at below $1.00 per share. Pursuant to Nasdaq Listing Rule 5550(a)(2) listed securities must maintain a minimum bid price of $1.00 per share, and Nasdaq Listing Rule 5810(c)(3)(A) provides that a failure to meet the minimum bid price requirement exists if the deficiency continues for a period of 30 consecutive business days. If our stock price remains below $1.00 for 30 consecutive business days we will receive a deficiency notice from Nasdaq and will be provided a period of 180 calendar days to regain compliance with the minimum bid price requirement. To regain compliance, the closing bid price of our Class A common stock must meet or exceed $1.00 per share for a minimum of ten consecutive business days during this 180-day period.
If we do not regain compliance within the allotted compliance period, including any extensions that may be granted by Nasdaq, Nasdaq will provide notice that the Company’s common stock will be subject to delisting. The Company would then be entitled to appeal Nasdaq’s determination, but there can be no assurance that Nasdaq would grant the Company’s request for continued listing.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 96
Such a delisting would likely have a negative effect on the price of the securities and would impair your ability to sell or purchase the securities when you wish to do so. In the event of a delisting, we can provide no assurance that any action taken by us to restore compliance with listing requirements would allow our securities to become listed again, stabilize the market price or improve the liquidity of our securities, prevent our securities from dropping below the Nasdaq minimum bid price requirement or prevent future non-compliance with Nasdaq’s listing requirements.
Additionally, if our securities are not listed on, or become delisted from, Nasdaq for any reason, trading our common stock could be conducted only in the over-the-counter (“OTC”) market or on an electronic bulletin board established for unlisted securities such as the OTC Bulletin Board, an inter-dealer automated quotation system for equity securities that is not a national securities exchange, the liquidity and price of our securities may be more limited than if we were quoted or listed on Nasdaq or another national securities exchange. You may be unable to sell your securities unless a market can be established or sustained. As a delisted security, our common stock would be subject to SEC rules as a “penny stock,” which impose additional disclosure requirements on broker-dealers. The regulations relating to penny stocks, coupled with the typically higher cost per trade to the investor of penny stocks due to factors such as broker commissions generally representing a higher percentage of the price of a penny stock than of a higher-priced stock, would further limit the ability of investors to trade in our common stock. In addition, delisting could harm our ability to raise capital through alternative financing sources on terms acceptable to us, or at all, and may result in the potential loss of confidence by investors, suppliers, customers and employees and fewer business development opportunities. For these reasons and others, delisting would adversely affect the liquidity, trading volume and price of our common stock, causing the value of an investment in us to decrease and having an adverse effect on our business, financial condition and results of operations, including our ability to attract and retain qualified employees and to raise capital.
We intend to monitor the closing bid price of our Class A common stock and consider options to resolve the noncompliance with the minimum bid price requirement, including by seeking approval from our stockholders to effect a reverse stock split of the issued and outstanding shares of our Class A common stock.
There can be no assurance that the Company will be able to comply with the minimum bid price requirement or will otherwise be in compliance with other Nasdaq listing criteria.
If we implement a reverse stock split, liquidity of our stock may be adversely affected.
We may seek approval from our stockholders to effect a reverse stock split of the issued and outstanding shares of our Class A common stock. However, there can be no assurance that the reverse stock split would be approved by our stockholders. Further, there can be no assurance that the market price per new share of common stock after the reverse stock split will remain unchanged or increase in proportion to the reduction in the number of old shares of common stock outstanding before the reverse stock split. The liquidity of the shares of our common stock may be affected adversely by any reverse stock split given the reduced number of shares of common stock that will be outstanding following the reverse stock split, especially if the market price of our common stock does not increase as a result of the reverse stock split.
Following any reverse stock split, the resulting market price of our common stock may not attract new investors and may not satisfy the investing requirements of those investors. Although we believe that a higher market price of common stock may help generate greater or broader investor interest, there can be no assurance that a reverse stock split will result in a share price that will attract new investors, including institutional investors. In addition, there can be no assurance that the market price of our common stock will satisfy the investing requirements of those investors. As a result, the trading liquidity of our common stock may not necessarily improve.
The terms of the Warrants may be amended in a manner adverse to a holder if holders of at least 50% of the then outstanding Public Warrants approve of such amendment.
The Private Placement Warrants and Public Warrants were issued in registered form under the Warrant Agreement, between us and Continental Stock Transfer & Trust Company, our warrant and transfer agent (“ContinentalContinental”). The Warrant Agreement provides that the terms of the Warrants may be amended without the consent of any holder to cure any ambiguity or correct any defective provision, but requires the approval by the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 97
holders of at least 50% of the then outstanding Public Warrants to make any change that adversely affects the interests of the registered holders of Public Warrants. Accordingly, we may amend the terms of the Public Warrants in a manner adverse to a holder if holders of at least 50% of the then outstanding Public Warrants approve of such amendment. Although our ability to amend the terms of the Public Warrants with the consent of at least 50% of the then outstanding Public Warrants is unlimited, examples of such amendments could be amendments to, among other things, increase the exercise price of the Warrants, shorten the exercise period or decrease the number of our Class A common stock purchasable upon exercise of a Warrant.
The exercise of Warrants for our stock would increase the number of shares eligible for future resale in the public market and result in dilution to our stockholders. Such dilution will increase if more of our shares are redeemed.
As of December 31, 20212022, we had Warrants to purchase an aggregate of 14,213,277267 shares of our Class A common stock outstanding, comprising 9,199,944934 Public Warrants sold as part of the units in the closing of the initial public offering of THMA, which closed on February 4, 2021 (the “Initial Public Offering”) (whether they were purchased in the Initial Public Offering or thereafter in the open market), and 5,013,333 Private Placement Warrants issued to LJ10 LLC, a Delaware limited liability company (the “Sponsor”) in a private placement simultaneously with the Initial Public Offering. These Warrants are exercisable at any time through December 4, 2026. The likelihood that the Warrants will be exercised increases if the trading price of shares of our stock exceeds the exercise price of the Warrants. The exercise price of these Warrants is $11.50 per share.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 93
There is no guarantee that the Warrants will ever be in the money after they become exercisable and prior to their expiration, and as such, the Warrants may expire worthless.
To the extent the Warrants are exercised, additional Class A common stock will be issued, which will result in dilution to the holders of our stock and increase the number of shares eligible for resale in the public market. Holders of Warrants do not have a right to redeem the Warrants. Sales of substantial numbers of shares issued upon the exercise of Warrants in the public market or the potential that such Warrants may be exercised could also adversely affect the market price of our stock.
We may redeem unexpired Warrants prior to their exercise at a time that is disadvantageous to you, thereby making your Warrants worthless.
We will have the ability to redeem the Public Warrants in whole and not in part at any time prior to their expiration, at a price of $0.01 per Warrant, if, and only if, the last reported sales price of the Class A common stock equals or exceeds $18.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within a 30 trading-day period ending on the third trading day prior to the date we send the notice of redemption to the Warrant holders. If and when the Warrants become redeemable by us, we may exercise our redemption right even if we are unable to register or qualify the underlying securities for sale under all applicable state securities laws. Redemption of the outstanding Warrants as described above could force you to: (1) exercise your Warrants and pay the exercise price therefor at a time when it may be disadvantageous for you to do so; (2) sell your Warrants at the then-current market price when you might otherwise wish to hold your Warrants; or (3) accept the nominal redemption price which, at the time the outstanding Warrants are called for redemption, we expect would be substantially less than the market value of your Warrants. The Private Placement Warrants are not redeemable by us in such a case so long as they are held by the Sponsor or its permitted transferees. The Sponsor, or its permitted transferees, has the option to exercise the Private Placement Warrants on a cashless basis and has certain registration rights.
In addition, we will have the ability to redeem the outstanding Warrants in whole and not in part for shares of our stock at any time after they become exercisable and prior to their expiration, at a price of $0.10 per warrant if, among other things, (i) the closing price of our Class A common stock equals or exceeds $10.00 per public share (as adjusted for adjustments to the number of shares issuable upon exercise or the exercise price of a Warrant) on the trading day prior to the date on which we send the notice of redemption to the Warrant holders and (ii) the closing price of our Class A common stock (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which we send the notice of redemption to the Warrant holders is less than $18.00 per share (as adjusted for share splits, share dividends, reorganizations, recapitalizations and the like). If this occurs, then the Private
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 98
Placement Warrants must also concurrently be called for redemption on the same terms (except with respect to a holder’s ability to cashless exercise its Warrants) as the outstanding Public Warrants. In such a case, the holders will be able to exercise their Warrants prior to redemption for a number of Class A common stock determined based on the redemption date and the fair market value of the Class A common stock. The value received upon exercise of the Warrants (1) may be less than the value the holders would have received if they had exercised their Warrants at a later time where the underlying share price is higher and (2) may not compensate the holders for the value of the Warrants.
Future resales of our Class A common stockstock may cause the market price of our securities to drop significantly, even if our business is doing well.
Subject to customary exceptions, the Sponsor, parties to the Registration Rights Agreement as amended and restated as of December 3, 2021, certain holders of Private Investment in Public Equity (“PIPE”)PIPE Shares and certain other holders of our Class A common stock arewere subject to lock-up provisions in which they agreed not to sell or otherwise dispose of any our Class A common stock or any other equity securities of Pear convertible into or exercisable or exchangeable for our Class A common stock held by any of them for a certain period of 180 days after the Closing)time.
However, following the expiration of eachNow since these lock-up periods have expired, the applicable stockholders willare not be restricted from selling our Class A common stock held by them, other than by applicable securities laws. As such, sales of a substantial
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 94
number of shares of our common stock in the public market could occur at any time after the expiration of the applicable lock-up periods restricted from selling our Class A common stock held by them, other than by applicable securities laws. As such, sales of a substantial number of shares of our common stock in the public market could occur at any time. These sales, or the perception in the market that the holders of a large number of shares intend to sell shares, could reduce the market price of the Class A common stock.
As restrictions on resale end and registration statements are available for use, the sale or possibility of sale of these shares could have the effect of increasing the volatility in our share price or the market price of our stock could decline if the holders of currently restricted shares sell them or are perceived by the market as intending to sell them.
The market price and trading volume of our Class A common stock may be volatile and you could decline significantly.lose all or part of your investment.
The trading price of our common stock is likely to be highly volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond our control, including limited trading volume. Since our common stock began trading on The Nasdaq Capital Market, our stock price has traded at prices as low as $0.20 per share and as high as $14.60 per share through March 30, 2023.
The stock markets, including Nasdaq on which we list the Class A common stock, have from time to time experienced significant price and volume fluctuations. Even if an active, liquid and orderly trading market develops and is sustained for our stock, the market price of our stock may be volatile and could decline significantly. In addition, the trading volume in our stock may fluctuate and cause significant price variations to occur. If the market price of our stock declines significantlyfurther, you may be unable to resell your shares at an attractive price (or at all). We cannot assure you that the market price of our stock will not fluctuate widely or decline significantly in the future in response to a number of factors, including, among others, the following:
•the realization of any of the risk factors presented in this Form 10-K;
•our announcement seeking strategic alternatives, and a potential transaction;
•failure to comply with the requirements of Nasdaq;
•the inclusion, exclusion or removal of our common stock from any stock market indices;
•additions and departures of key personnel;
•changes in the industries in which we operate;
•developments involving our competitors;
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 99
•changes in laws and regulations affecting our business;
•actual or anticipated differences in our estimates, or in the estimates of analysts, for our revenues, Adjusted EBITDA, results of operations, level of indebtedness, liquidity, or financial condition;
•additions and departures of key personnel;
•failure to comply with the requirements of Nasdaq;
•failure to comply with the Sarbanes-Oxley Act or other laws or regulations;
•future issuances, sales, resales or repurchases or anticipated issuances, sales, resales or repurchases, of our securities;
•publication of research reports by securities analysts about us or our competitors or our industry;
•the public’s reaction to our press releases, our other public announcements and our filings with the SEC;
•actions by stockholders, including the sale by the PIPE investors of any of their shares of our stock;
•the performance and market valuations of other similar companies;
•commencement of, or involvement in, litigation involving us;
•broad disruptions in the financial markets, including sudden disruptions in the credit markets;
•speculation in the press or investment community;
•actual, potential or perceived control, accounting, or reporting problems;
•changes in accounting principles, policies, and guidelines;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 95
•other events or factors, including those resulting from infectious diseases, health epidemics and pandemics (including the ongoing COVID-19 public health emergency), natural disasters, war, acts of terrorism, or responses to these events;
•actual or anticipated fluctuations in our quarterly financial results or the quarterly financial results of companies perceived to be similar to us;
•changes in the market’s expectations about our results of operations;
•our results of operations failing to meet the expectation of securities analysts or investors in a particular period;
•any major change in our Board of Directors or management;
•sales of substantial amounts of the shares of common stock by our directors, executive officers or significant stockholders or the perception that such sales could occur; and
•general economic and political conditions such as recessions, interest rates, fuel prices, international currency fluctuations, and acts of war or terrorism.
The stock market has recently experienced extreme price and volume fluctuations. The market prices of securities have experienced fluctuations that often have been unrelated or disproportionate to their companies’ operating results. We believe that the recent volatility and our current market price in part reflect market and trading dynamics unrelated to our underlying business, and we do not know how long these dynamics will last. In the past, securities class-action litigation has often been instituted against companies following periods of volatility in the market price of their shares. This type of litigation could result in substantial costs and divert our management’s attention and resources, which could have a material adverse effect on us.
Our stock price may be exposed to additional risks because our business became a public company through a “de-SPAC” transaction. There has been increased focus by government agencies on transactions such as the Business Combination in the last year, and we expect that increased focus to continue. As a result, we may be subject to
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 100
increased scrutiny by the SEC and other government agencies and holders of our securities, which could adversely affect the price of our Class A common stock. Broad market and industry factors may materially harm the market price of our securities irrespective of our operating performance. The stock market in general, and Nasdaq in particular, have experienced price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of the particular companies affected. The trading prices and valuations of these stocks, and of our securities, may not be predictable. A loss of investor confidence in the market for retail stocks or the stocks of other companies which investors perceive to be similar to us could materially and adversely affect our business, prospects, financial condition and results of operations. A decline in the market price of our securities also could adversely affect our ability to issue additional securities and our ability to obtain additional financing in the future.
The requirements of being a public company may strain our resources and divert management’s attention, and the increases in legal, accounting and compliance expenses may be greater than we anticipate.
As a public company we will incur significant legal, accounting, and other expenses that we did not incur as a private company. We are subject to the reporting requirements of the Exchange Act and are required to comply with the applicable requirements of the Sarbanes-Oxley Act and the Dodd-Frank Wall Street Reform and Consumer Protection Act, as well as the rules and regulations subsequently implemented by the SEC and the listing standards of Nasdaq, including changes in corporate governance practices and the establishment and maintenance of effective disclosure and financial controls. Compliance with these rules and regulations can be burdensome. Our management and other personnel devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations will increaseincreased our historical legal and financial compliance costs and will make some activities more time-consuming and costly. For example, we expect that these rules and regulations may make it difficult and expensive for us to obtain director and officer liability insurance and could also make it more difficult for us to attract and retain qualified members of our Board of Directors as compared to when we were a private company. In particular, we In particular, we have incurred and expect to continue to incur significant expenses and devote substantial management effort toward ensuring compliance with the requirements of Section 404 of the Sarbanes-Oxley Act, which will increase when we are no longer an “emerging growth company.” We may need to hire additional accounting and financial staff, and engage outside consultants, all with appropriate public company experience and technical accounting knowledge and maintain an internal audit function, which will increase our operating expenses. Moreover, we could incur additional compensation costs in the event that we decide to pay cash compensation closer to that of other public companies, which would increase our general and administrative expenses and could materially and adversely affect our profitability. We cannot predict or estimate the amount of additional costs we may incur or the timing of such costs.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 96
cannot predict or estimate the amount of additional costs we may incur or the timing of such costs. We are an emerging growth company within the meaning of the Securities Act, and if we take advantage of certain exemptions from disclosure requirements available to emerging growth companies, this could make our securities less attractive to investors and may make it more difficult to compare our performance with other public companies.
We are an “emerging growth company” within the meaning of the Securities Act of 1933, as amended (the “Securities Act”), as modified by the Jumpstart Our Business Startups Act of 2012 (the “JOBS ActJOBS Act”), and may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies for as long as we continue to be an emerging growth company, including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. As a result, our stockholders may not have access to certain information they may deem important. We will remain an emerging growth company until the earliest of (i) the day we are deemed to be a large accelerated filer, which, in addition to certain other criteria, means the market value of our common equity that is held by non-affiliates exceeds $700 million as of the end of the prior fiscal year’s second fiscal quarter, (ii) the last day of the fiscal year in which we have total annual gross revenue of $1.07 billion or more during such fiscal year, (iii) the date on which we have issued more than $1 billion in non-convertible debt in the prior three-year period and (iv) December 31, 2026 (the last day of the fiscal year ending after the fifth anniversary of the THMA IPOInitial Public Offering). Investors may find our securities less attractive because we will rely on these exemptions. If some investors find our securities less attractive as a result of our reliance on these exemptions, the trading prices of our securities may be lower than they otherwise would be, there may be a less active trading market for our securities and the trading prices of our securities may be more volatile.
In addition, Section 107 of the JOBS Act also provides that an emerging growth company can take advantage of the exemption from complying with new or revised accounting standards provided in Section 7(a)(2)(B) of the Securities Act as long as we are an emerging growth company. An emerging growth company can therefore delay
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 101
the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected not to opt out of such an extended transition period and, therefore, we may not be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies. This may make comparison of our financial statements with another public company which is neither an emerging growth company nor an emerging growth company which has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
The exclusive forum provision in our Certificate of Incorporation may have the effect of discouraging lawsuits against our directors and officers.
Our Second Amended and Restated Certificate of Incorporation (our “Certificate of IncorporationCertificate of Incorporation”) provides that unless we consent to the selection of an alternative forum, the Court of Chancery of the State of Delaware will, to the fullest extent permitted by applicable law, be the sole and exclusive forum for (i) any derivative action brought by a stockholder on behalf of us, (ii) any claim of breach of a fiduciary duty owed by any of our directors, officers, stockholders, or employees, (iii) any claim against us arising under our Certificate of Incorporation, our Amended and Restated Bylaws (the “BylawsBylaws”) or the Delaware General Corporation Law, as may be amended from time to time (the “DGCLDGCL”) and (iv) any claim against us governed by the internal affairs doctrine. The Certificate of Incorporation designates the US District Court for the District of Delaware as the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act of 1933, as amended.
This exclusive forum provision does not apply to claims under the Exchange Act, but does apply to other state and federal law claims including actions arising under the Securities Act. Section 22 of the Securities Act, however, created concurrent jurisdiction for federal and state courts over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder. Accordingly, there is uncertainty as to whether a court would enforce such a forum selection provision as written in connection with claims arising under the Securities Act.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 97
Although we believe this provision benefits us by providing increased consistency in the application of Delaware law in the types of lawsuits to which it applies, this choice of forum provision may have the effect of increasing costs for investors to bring a claim against us and our directors and officers and of limiting a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or any of our directors, officers, other employees or stockholders, which may discourage (but not prevent) lawsuits with respect to such claims.
Provisions in our Certificate of Incorporation and Delaware law may inhibit a takeover of us, which could limit the price investors might be willing to pay in the future for the Class A common stock and could entrench management.
Our Certificate of Incorporation contains provisions that may discourage unsolicited takeover proposals that stockholders may consider to be in their best interests. These provisions include three-year director terms and the ability of our Board of Directors to designate the terms of and issue new series of preferred shares, which may make more difficult the removal of management and may discourage transactions that otherwise could involve payment of a premium over prevailing market prices for our securities.
Section 203 of the DGCL affects the ability of an “interested stockholder” to engage in certain business combinations, for a period of three years following the time that the stockholder becomes an “interested stockholder.” We elected in our Certificate of Incorporation not to be subject to Section 203 of the DGCL. Nevertheless, our Certificate of Incorporation contains provisions that have the same effect as Section 203 of the DGCL, except that it provides that affiliates of our Sponsor and their transferees will not be deemed to be “interested stockholders,” regardless of the percentage of our voting stock owned by them, and will therefore not be subject to such restrictions. These Certificate of Incorporation provisions may limit the ability of third parties to acquire control of our company.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 102
Claims for indemnification by our directors and officers may reduce our available funds to satisfy successful third-party claims against us and may reduce the amount of money available to us.
Our Certificate of Incorporation and Bylaws provide that we will indemnify our directors and officers, in each case to the fullest extent permitted by Delaware law. In addition, as permitted by Section 145 of the DGCL, our Bylaws and ourthe indemnification agreements that we entered into with our directors and officers provide that:
•We will indemnify our directors and officers for serving in those capacities or for serving other business enterprises at our request, to the fullest extent permitted by Delaware law. Delaware law provides that a corporation may indemnify such person if such person acted in good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of the registrant and, with respect to any criminal proceeding, had no reasonable cause to believe such person’s conduct was unlawful;
•We may, in our discretion, indemnify employees and agents in those circumstances where indemnification is permitted by applicable law;
•We are required to advance expenses, as incurred, to our directors and officers in connection with defending a proceeding, except that such directors or officers shall undertake to repay such advances if it is ultimately determined that such person is not entitled to indemnification;
•We are not obligated pursuant to our Bylaws to indemnify a person with respect to proceedings initiated by that person against Auroraus or our other indemnitees, except with respect to proceedings authorized by our Board of Directors or brought to enforce a right to indemnification;
•the rights conferred in our Bylaws are not exclusive, and we are authorized to enter into indemnification agreements with our directors, officers, employees and agents and to obtain insurance to indemnify such persons; and
•We may not retroactively amend our amended and restated bylaw provisions to reduceprovisions of our Bylaws to amend our indemnification obligations to directors, officers, employees and agents.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 98
We do not intend to pay dividends for the foreseeable future, if ever.
We have never declared or paid any cash dividends on our capital stock and do not intend to pay any cash dividends in the foreseeable future. We expect to retain future earnings, if any, to fund the development and growth of our business. Any future determination to pay dividends on our capital stock will be at the discretion of our Board of Directors. Accordingly, investors must rely on sales of the Class A common stock after price appreciation, which may never occur, as the only way to realize any future gains on their investments.
We may be subject to securities litigation, which is expensive, and could divert management attention.
The market price of our common stock may be volatile and, in the past, companies that have experienced volatility in the market price of their stock have been subject to securities class action litigation. We may be the target of this type of litigation in the future. Securities litigation against us could result in substantial costs and divert management’s attention from other business concerns, which could seriously harm its business.
If securities or industry analysts do not publish or cease publishing research or reports about us, our business, or the market in which we operate, or if they change their recommendations regarding our securities adversely, the price and trading volume of our securities could decline.
The trading market for our securities will be influenced by the research and reports that industry or securities analysts may publish about us, our business, market or competitors. If any of the analysts who may cover us change their recommendation regarding our shares of common stock adversely, or provide more favorable relative recommendations about our competitors, the price of our common stock would likely decline. If any analyst who may cover us were to cease our coverage of us or fail to regularly publish reports on it, we could lose visibility in the financial markets, which in turn could cause our share price or trading volume to decline.
Future issuances of debt securities and equity securities may adversely affect us, including the market price of our stock and may be dilutive to existing stockholders.
In the future, we may incur debt or issue equity ranking senior to theour Class A common stock. Those securities will generally have priority upon liquidation. Such securities also may be governed by an indenture or other instrument containing covenants restricting its operating flexibility. Additionally, any convertible or exchangeable securities that we issue in the future may have rights, preferences and privileges more favorable than those of the Class A
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 103
common stock. Because our decision to issue debt or equity in the future will depend on market conditions and other factors beyond our control, we cannot predict or estimate the amount, timing, nature or success of our future capital raising efforts. As a result, future capital raising efforts may reduce the market price of our stock and be dilutive to existing stockholders.
Our failure to meet the continued listing requirements of Nasdaq could result in a delisting of our securities.
If we fail to satisfy the continued listing requirements of Nasdaq such as the corporate governance requirements or the minimum closing bid price requirement, Nasdaq may take steps to delist our securities. Such a delisting would likely have a negative effect on the price of the securities and would impair your ability to sell or purchase the securities when you wish to do so. In the event of a delisting, we can provide no assurance that any action taken by us to restore compliance with listing requirements would allow our securities to become listed again, stabilize the market price or improve the liquidity of our securities, prevent our securities from dropping below the Nasdaq minimum bid price requirement or prevent future non-compliance with Nasdaq’s listing requirements. Additionally, if our securities are not listed on, or become delisted from, Nasdaq for any reason, trading our common stock could be conducted only in the over-the-counter (“OTC”) market or on an electronic bulletin board established for unlisted securities such as the OTC Bulletin Board, an inter-dealer automated quotation system for equity securities that is not a national securities exchange, the liquidity and price of our securities may be more limited than if we were quoted or listed on Nasdaq or another national securities exchange. You may be unable to sell your securities unless a market can be established or sustained.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 99104
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 2. PROPERTIES
Our corporate headquarters is located in Boston, Massachusetts, where we lease approximately 19,000 square feet pursuant a lease that expires on June 1, 2028. to a lease that expires on June 1, 2028. Effective January 1, 2023, the Company entered into a sublease agreement with a third party to sublease 7,218 square feet of office space. The sublease term is for one year with an option to renew for two additional years in one-year increments.
We also lease approximately 17,000 square feet of office space in San Francisco, California pursuant to a lease that expires on July 31, 2025. We use these facilities forOur San Francisco office space is currently available to sublease as we continue consolidating our footprint.
We use the Boston facility for our executive and principal offices for sales, marketing, engineering, product, finance, legal, human resources, information technology, engineering, product, sales and marketing, and other administrative functions. and we use the San Francisco facility for primarily engineering and product activities.
In addition, we lease approximately 7,700 square feet of office space in Raleigh, North Carolina, pursuant to a lease that expires on May 31, 2026. We use this facility to houseEffective January 6, 2023, the Company subleased the entirety of the Raleigh, North Carolina office. The term of the sublease arrangement commenced on January 6, 2023, and expire twelve months thereafter, with an option to renew our Patient Support Center and various other personnel. for one twelve-month period.
The Company believes its existing facilities are adequate for its current requirements.
See Note 8, Leases, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for additional information regarding our specific leaseholds.
ITEM 3. LEGAL PROCEEDINGS
We are not currently subject to any material legal proceedings, nor, to our knowledge, is any material legal proceeding threatened against us or any of our officers or directors in their corporate capacity. From time to time, we may become involved in litigation or legal proceedings relating to claims arising from the ordinary course of business. Although the outcomes of these legal proceedings are inherently difficult to predict, we do not expect the resolution of these proceedings to have a material adverse effect on our financial position, results of operations, or cash flow.
ITEM 4. MINE SAFETY DISCLOSURES
Not Applicable.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 100105
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS, AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our Class A common stock and public warrantsPublic Warrants trade on The Nasdaq Stock Market LLC under the symbols PEAR and PEARW, respectively (formerly THMA and THMAW, respectively).
Holders
As of March 130, 20222023, there were approximately 86168 shareholders of record of our Class A common stock and 2two warrant holders of record.
The number of record holders is based upon the actual number of holders registered on our books at such date and does not include holders of shares in street name or persons, partnerships, associations, corporations, or other entities identified in security position listings maintained by depository trust companies.
Dividend PolicyDividends
We have never declared or paid any cash dividends on our Class A common stock, and we do not anticipate paying cash dividends in the foreseeable future.
Securities Authorized for Issuance Under Equity Compensation Plans
For information regarding securities authorized for issuance under our employee stock-based compensation plans, see Part III, Item 12, Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters of this Form 10-K.
Recent Sales of Unregistered Securities and Use of Proceeds
The information required has been previously disclosed in our Current Report on Form 8-K filed with the Securities and Exchange Commission on December 8, 2021.
Issuer Purchases of Equity SecuritiesPurchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
Stock performance graph
We are a smaller reporting company, as defined by Rule 12b-2 of the Exchange act, and are not required to provide a performance graph.
ITEM 6. Selected Financial DataReserved
Not applicable.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 106
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
TThis Annual Report on Form 10-K (“his discussion may containForm 10-K”) contains forward-looking statements based upon current expectations that involve risks and uncertainties. Pear’s actual results may differ materially from those anticipated in these forward-looking statements as a result of various factors, including those set forth under in the “Risk Factors” section included in Part 1I, Item 1A of this Form 10-K. All references to years, unless otherwise noted, refer to our fiscal years, which end on December 31. For purposes of this section, all references to “we,” “us,” “our,” “Pear,” or the “Company” refer to Pear Therapeutics, Inc. and its consolidated subsidiaries.
The following discussion and analysis should also be read in conjunction with the accompanying consolidated financial statements included in Part II, Item 8 of this Form 10-K. This section of this Form 10-K generally discussesdiscusses 2022 and 2021 financial condition, and 2020 itemsresults of operations and year-to-year comparisons between 2021 and 2020.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 1012022 and 2021. For discussion of 2021 items and year-over-year comparisons between 2021 and 2020 that are not included in this 2022 Form 10-K, refer to “Item 7. – Management’s Discussion and Analysis of Financial Condition and Results of Operations” found in our Form 10-K for the year ended December 31, 2021, that was filed with the Securities and Exchange Commission on March 29, 2022.
OverviewOverview
Due to the fact that we were unable to generate sufficient cash flows from operations, obtain funding to sustain operations, or reduce or stabilize expenses to the point where we could have realized a net positive cash flow, management and our board of directors determined that it was in the best interests of the stockholders to seek a strategic alternative so that we could continue to operate. If the strategic process is unsuccessful, our Board may decide to pursue a liquidation or obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection.
Further, Perceptive has alleged that certain defaults or events of default have occurred and are continuing under the terms of our Perceptive Credit Facility. Perceptive has not delivered any formal notice of Default or Event of Default. To the extent that any such allegations are valid, Perceptive would have certain rights and remedies under the Perceptive Credit Facility. The Company disputes the allegations and is in discussions with Perceptive to resolve this dispute and otherwise to address the Company’s obligations under the Perceptive Credit Facility. There can be no assurances that such discussions will result in any resolution, and any resolution, or the lack of any resolution, may result in Perceptive exercising remedies under the Perceptive Credit Facility.
The Company cautions that trading in the Company’s securities is highly speculative and poses substantial risks. Trading prices for the Company’s securities may bear little or no relationship to the actual value realized, if any, by holders of the Company’s securities. In the event of liquidation, bankruptcy or other wind-down event, holders of our securities will likely suffer a total loss of their investment. Accordingly, the Company urges extreme caution with respect to existing and future investments in its securities.
Pear is a commercial-stage healthcare company pioneering a new class of medicine, referred to as PDTs, which use software to treat diseasesdisease. Our vision is to advance healthcare through the widespread use of PDTs.
Recent global trends are converging to highlight a significant unmet need for new and innovative solutions for the treatment of diseases. We believe our PDTs are well suited to satisfy this growing unmet need for the treatment of diseases, including addiction and insomnia.
Pear is a leader in the PDT industry. Our marketed PDTs, reSET, reSET-O, and Somryst, were among the first three PDTs authorized by FDA. We believe PDTs have the potential to improve clinical outcomes, facilitate improved care coordination, improve practice efficiency, and provide data tracking over time.
Two of our FDA-authorized PDTs are for the treatment of addiction, which currently affects more than 20 million people in the USTwo of our FDA-authorized PDTs are for the treatment of addiction. Our first product, reSET, is indicated for the treatment of SUDsubstance use disorder (“SUD”) as a monotherapy. Our second product, reSET-O, is indicated for the treatment of OUDopioid use disorder (“OUD”) in combination with buprenorphine.
Our third product, Somryst, is indicated for the treatment of chronic insomnia, which currently affects more. The Company has deprioritized commercialization efforts regarding Somryst while focusing available resources on the commercialization of reSET and reSET-O and therefore recorded an impairment expense of $0.8 million related to the acquired technology, which is included in than approximately 30 million people in the US.
cost of revenue in the Company's consolidated statement of operations, during the year ended December 31, 2022.
See “Recent Events” below for further details.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 107
Operating Segments
We operate our business in a single segment and as one reporting unit, which is how our chief operating decision maker (who is our president and chief executive officer) reviews financial performance and allocates resources.
Factors Affecting Our Performance and Results of Operations
We believe that our performance and future success depend on many factors that present significant opportunities for us but also pose risks and challenges, including those discussed more fully under the heading “Risk Factors” in Part I, Item 1A of this Form 10-K.
In February 2023, our strategic focus shifted to identifying and evaluating of a range of potential strategic alternatives designed to maximize stockholder value while we continue to operate the Company and serve our patients.
Key Business Metrics
We monitor the key non-financial operating performance metrics to help us evaluate our business, identify trends affecting our business, formulate business plans, and make strategic decisions. We began using these metrics in 2021 as we began to generate product revenue, primarily from sales of reSET and reSET-O. The metrics include the following:
Prescriptions WrittenA.Total Prescriptions -in a given period is (a) the imputed number of prescriptions based on revenue recognized under Access Agreements, plus (b) the number of prescriptions written bywhich a clinician for one of Pear’s three commercial products, reSET®, reSET-O®, and Somryst®.are not imputed under Access Agreements.
B.Fulfillment Rate - the percentage of prescriptions written by a clinician forin a given period one of Pear’s three commercial products that result in patients downloading and accessing therapeutic software content.
Covered Lives - the estimated number of persons with access to Pear’s PDTs by either listing on a formulary or on a fee schedule as a covered benefit with payors, including self-insured employers, IDNs, PBMs, commercial payors, and government payors. Covered lives is a proxy metric for payment rate, which is the percentage of fulfilled prescriptions for which Pear receives payment. As we areis (a) the number of prescriptions for which either a patient commences therapy or earlythere inis the development of the PDT category, most payors do not yet cover PDTs; therefore, Pear does not yet receive payment for most fulfilled prescriptions.a contractual payment obligation and revenue has been recognized divided by (b) Total Prescriptions. (Total Prescriptions times Fulfillment Rate equals Fulfilled Prescriptions.)
To illustrate how prescriptions written, fulfillment rate, and covered lives relate to product revenue, we believe it is helpful to provide a hypothetical scenario of 100 prescriptions written with a 50% fulfillment rate and a 50% payment rate. We use covered lives as a metric that is expected to impact payment rate because, as we grow the number of covered livesC.Payment Rate in a given period is (a) the number of prescriptions for our three products,which the payment rate should increase. In this hypothetical scenario, Pear has 50 filled prescriptions, of which 25 are paid. Product revenue for the 100 prescriptions written would be
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 102
calculated by multiplying (a) 25 paid prescriptions by (b) the wholesale acquisition cost (WAC)company receives payment divided by (b) Fulfilled Prescriptions. (Fulfilled Prescriptions times Payment Rate equals Paid Prescriptions.)
D.Average Selling Price, or “ASP”, in a given period is the average price received by the Company per script for which the respective prescription, reduced by (c) any applicable discounts and rebates as well as estimated contingency management costs, if applicable.
Factors Affecting Our Performance and Results of Operations
We believe that our performance and future success depend on many factors that present significant opportunities for us, but also pose risks and challenges, including those discussed more fully under the heading “Risk Factors” in Part I, Item 1A of this Form 10-K.Company receives payment.
| | | | | | | | | |
| Key Performance Operating Metric | | | Year Ended December 31, 2022 |
| | | |
| Total Prescriptions | | | 45,000+ |
| Fulfillment Rate | | | 53% |
| Payment Rate | | | 41% |
| Average Selling Price (ASP) | | | $1,195 |
Product Revenue
We generate product revenue from the sale of our three FDA-authorized PDTs: reSET, reSET-O, and Somryst. We began our efforts to self-commercialize reSET and reSET-O in Q4 2019 and Somryst in Q4 2020. Through at least 2023, salesSales of our existing productsreSET and reSET-O are not expected to reduce our continued net losses.operating losses over time, but we cannot predict when we will achieve profitability. While we continue to support Somryst, our primary focus is on reSET and reSET-O.
We enter into arrangements with health care providers and payers enter into agreements with health care providers and payors and state and local governments to provide prescriptions which provide for volume-based discounts and other discounts, and in certain circumstances, value-based rebates (“Access Agreement”). We also enter into arrangements with health care providers and payors that provide for government-mandated and/or privately negotiated rebates and discounts with respect to the purchase of our productproducts. A portion of the product revenue is recognized when the products are made available to the customer (under a bulk ordervia Access Agreements) or when a prescription is fulfilled (via third-party reimbursement), and athe portion of the product revenue related to the clinician’s access to our proprietary clinician dashboard, PearMD, is deferred and recognized ratably over the remaining term of the PDT or if purchased under acontract (if purchased via an Access Agreement) or
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 108
the prescription duration (if purchased via third party reimbursement). When sold under an bulk purchase agreement is deferred and recognized ratablyAccess Agreement and implementation services are included, we recognize the implementation services as control is transferred to the customer, generally over the term of the contract. See Note 2, Revenue Recognition, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K.
If our development efforts for our PDT product candidates are successful and result in regulatory marketing authorization, we may generate revenue in the future from product sales or payment from collaboration or license agreements that we may enter into with third parties or any combination thereof. We cannot predict if, when, or to what extent we will generate revenue from the commercialization and sale of our product candidates. We may never succeed in obtaining regulatory approval for any of the product candidates that we currently have under development or potential future product candidates. remaining term of the contract.
Product revenue from our existing three FDA-authorized PDTs, as well as potential future product candidates, is and will be impacted by the many factors, including the following variables: coverage and reimbursement by payors, patient and clinician adoption of PDTs, pricing, reimbursement, contingency management, and product mix. (related only to reSET and reSET-O), and product mix. In the future, if we obtain additional financing, sales from future product candidates are expected to be impacted by similar variables.
PatientAlthough we provide products and Clinician Adoption of PDTs — To continue to grow our business, we will need to execute on our current business strategy of achieving and maintaining broad market acceptance of our PDTs by patients and physicians. Market acceptance and adoption of our PDTs depends on educating people with chronic conditions, as well as self-insured employers, commercial and government payors, health plans and physicians, and other government entities, as to the distinct features, therapeutic benefits, cost savings, and other advantages of our PDTs as compared to competitive products or other currently available methodologies. We have a market access team focused educating payors on the clinical outcomes and value proposition ofservices to many different types of customers, a significant portion of our product revenue is generated from awards under various US government, state, and local governments programs or grants. As a result of long sales cycle and the dependency on government or external funding, our product revenue can be unpredictable and fluctuate materially from period to period. We cannot predict the future level of demand for our products and to seekservices that towill secure favorable coverage policies and to maximizebe generated by these customers or the covered lives that have reimbursementfuture demand for our products by expanding approvals with IDNs, PBMs, and other payers. If we are not successful in demonstrating to existing or potential patients and prescribers the benefitsin the end-user marketplace. Our customer concentration exposes us to the risk of changes in the business condition of any of our products,major customers or if we arebroader changes in not able to achieve the support of patients, healthcare providers, and payors for our products, our sales may decline, or we may fail to increase our revenue.US government spending for opioid and substance abuse treatments.
Pricing — In the future, we expect to grow the number of commercially available PDTs in our product portfolio, offering a broad range of PDTs spanning multiple price points. PDTs may be subject to competition which may impact our pricing, and in addition, our products may be subject to legislative prescription-pricing practices.
Reimbursement Coverage and Reimbursement by Payors— As of December 31, 2021 , we have more than 30 organizations across more than 31.7 million covered lives providing access to our three FDA-authorized products via listing on formulary, as a covered benefit, bulk purchase, or funding a study. Pear’s payor strategy focuses across all major payor channels, including
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 103
employers, IDNs, PBMsOur payor strategy focuses across all major payor channels, including employers, Integrated Delivery Networks (“IDNs”), pharmacy benefit managers (“PBMs”), commercial payors, and government payors, including Medicaid and Medicare. We expect to increase our number of payors, and the pricing for such payors may vary as net prices for our products may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and can be subject to customary discounts and rebates. In addition, some of our products may be subject to certain customer incentive programs. Even if coverage is provided, the approved reimbursement amount may not be high enough to allow us to establish or maintain pricing sufficient to achieve profitability. As of December 31, 2021, approximately 21% of activated prescriptions (prescriptions written multiplied by fulfilled prescription rate) were paid prescriptions. In the future, as our market access team educates payors on the clinical attributes of our products, we expect our products to secure favorable coverage policies and to maximize the covered lives that have reimbursement for our products.
Contingency Management — Patient and Clinician Adoption of PDTs—To continue to grow our business, we need additional funding and once obtained we will need to execute our current business strategy of achieving and maintaining broad market acceptance of our PDTs by patients and physicians. Market acceptance and adoption of our PDTs depend on educating patients, self-insured employers, commercial and government payors, health plans and physicians, and other government entities, as to the distinct features, therapeutic benefits, cost savings, and other advantages of our PDTs as compared to competitive products or other currently available treatment options.
Pricing—In the future, assuming that we have sufficient operating capital, we expect to grow the number of commercially available PDTs in our product portfolio, offering a broad range of PDTs spanning multiple price points. PDTs may be subject to competition, which may impact our pricing, and in certain circumstances, we have offered significant discounted pricing in connection with pilot programs. In addition, our products may be subject to legislative prescription-pricing practices. Further, we continue to collect additional data to enhance product performance and bolster health economics and outcomes research (“HEOR”) and associated cost savings for payors. Our average selling price could decline over time as we engage in larger volume transactions that extend over multiple years and provide for larger volume discounts.
Contingency Management—Costs related to clinically-validated rewards that patients earn as they complete treatment goals within our reSET and reSET-O PDTs isare recorded as contra revenue.
ProductRevenue Mix — Sales of certain products and subscription, support, and professional services have, or are expected to have, higher gross margins than others. As a result, our financial performance depends, in part, on the mix of products we sell during a given period.
Collaboration and License Revenue
As of December 31, 2021, we do not have collaboration and licensing agreements from which we would recognize significant collaboration and licensing revenue. Our historical collaboration and license revenue is from Novartis Institute of Biomedical Research (“NIBR”). In June 2020, NIBR terminated their collaboration agreement with the Company, or the NIBR Agreement, related to our schizophrenia and multiple sclerosis product candidate development programs. We will not recognize any revenue under these agreements in the future.
If our development efforts for our PDT candidates are successful and result in regulatory marketing authorization or collaboration or license agreements with third parties, we may generate revenue in the future from collaboration or license agreements that we may enter into with third parties. We cannot predict if, when, or to what extent we may enter into future licensing or collaboration agreements. Further, we may never succeed in obtaining regulatory approval for any of our product candidates that are currently under development.
Cost of Productand subscription, support, and professional services that we sell during a given period.
Cost of Revenue
Cost of product revenue consists primarily of costs that are closely correlated or directly related to the delivery of the Company’s products, including pharmacy costs, royalties paid under license agreements related to our commercialized products, our products, including pharmacy costs, royalties paid under license agreements related to our commercialized products,
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 109
amortization of milestone payments capitalized related to commercialized products, hosting costs, and personnel-related costs, including salaries and bonuses, employee benefits, and stock-based compensation attributable to employees in a particular function. We expect the cost of product and associated with our implementation services. In addition, it includes subcontracted costs such as software license fees, technology, and service fees directly related to our subscription and service fee revenue.
During the quarter ended December 31, 2022, the Company’s forecasted future revenue and expense cash flow projections for Somryst indicated the carrying amounts of the intangible asset for Somryst was not fully recoverable, and therefore the Company recorded impairment expense of $0.8 million, which is included in cost of revenue in the Company's consolidated statement of operations, during the year ended December 31, 2022.
We expect the cost of revenue to increase as we further commercialize our products and increase the volume of prescriptions filled. However, we expect our cost of revenue to decrease as a percentage of total revenue over the longer-term, subject to the expected revenue growth. The majority of our cost of revenue does not fluctuate directly with increases or decreases in revenue.
Research and Development Expenses
As of December 31, 2021July 25, 2022, we have multiple product candidatespaused most investments in our pipeline, and we have incurred and will continue to incur significant research and development, or R&D, costs for their development. Developing PDTs requires a significant investment of resources over a prolonged period of time, and a core part of our strategy is to continue making sustained investments in this area. We have chosen to leverage our platform to initially focus on advancing our PDTs in the area of psychiatry. We expect our R&D expenses will increase in order to conserve cash. In addition, we anticipate that our personnel costs will decline as a result of the reductions in workforce that occurred in July and November 2022. We expect our R&D expenses will decrease substantially as we continue to investin 2023 in the development ofconnection with our pipeline of product candidates, future clinical development activities, and testing of our product candidatescontinued cost cutting measures.
R&D expenses consist of costs incurred in performing R&D activities, which include:
•expenses incurred in connection with the development of our pipeline of PDTs;
•costs in connection with third-party licensing agreements, including development and regulatory milestones;
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 104
expenses incurred to enhance our products; •costs in connection with third-party licensing agreements, including development and regulatory milestones;
•personnel-related expenses, including salaries, bonuses, benefits, and stock-based compensation for employees engaged in R&D functions;
•cost of clinical trials and studies;
•expenses incurred in connection with the discovery and development of our PDTs, including under agreements with third parties, such as consultants;
•expenses incurred under agreements with consultants who supplement our internal capabilities, including software development; and
•facilities, depreciation, and other expenses, which include direct and allocated expenses, such as rent and maintenance of facilities, insurance, and other operating costs for space and costs directly related to R&D functions.;
EachDue to the worsening macroeconomic environment and the longer-term outlook of the general markets and limited cash to fund further development of our product candidates has technical, clinical, regulatory, and commercial risk, including those discussed more fully under the heading “Risk Factors” in Part I, Item 1A of this Form 10-Kwe wrote off $2.1 million of intangible assets related to acquired technology, in the year ended December 31, 2022.
We expense R&D costs as incurred and do not track the costs at a project level. Advance payments made for goods or services to be received in the future for use in R&D activities are recorded as prepaid expenses. The prepaid amounts are expensed as the benefits are consumed. In the early phases of development, our R&D costs are often devoted to product platform and proof-of-concept studies that are not necessarily allocable to a specific product. The process of conducting the necessary clinical research to obtain regulatory approval is costly and time-consuming, and the successful development of our product candidates is highly uncertain. As a result, we are
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 110
unable to determine the duration and completion costs of our R&D projects, the costs of related clinical development, or when and to what extent, we will generate revenue from the commercialization and sale of any of our product candidates.
At this time we have paused all investment in our product candidates and delayed certain enhancements to our PearConnect platform. Each of our product candidates has technical, clinical, regulatory, and commercial risk, including those discussed more fully under the heading “Risk Factors” in Part I, Item 1A of this Form 10-K.
Selling, General, and Administrative Expenses
Selling, general, and administrative, or SG&A, expenses consist primarily of compensation for personnel, including stock-based compensation related to commercial, marketing, executive, finance and accounting, information technology, corporate and business development, and human resource functions. Other SG&A expenses include marketing initiatives, market research, and analysis, conferences and trade shows, travel expenses, professional services fees (including legal, patent, accounting, audit, tax, and consulting fees), insurance costs, amortization of certain internal-use software, general corporate expenses, and allocated facilities-related expenses, including rent and maintenance of facilities.
We expect SG&A expenses to continue to increase in absolute dollarsdecrease as we increase potential customers’ awareness and our sales and marketing functions to support existing products and future product launches. In addition, the Company expects to increase expenditures to expand our infrastructure to both drive and support the anticipated growth of the Company as well as additional expenses related to legal, accounting, information technology, investor and public relations, regulatory, and tax-related services associated with maintaining compliance with Nasdaq and SEC requirements, director and officer insurance costs, and other expenses associated with being a public company and implementing additional controls over financial reporting.reduce spending primarily on personnel-related expenses and certain commercial efforts in connection with our restructuring activities, including the reductions in workforce that occurred in July and November 2022.
Interest and other incomeOther Income (expenseExpense), net
Interest expense includes interest due under the Perceptive Credit Facility (as defined below)our secured Amended and Restated Credit Agreement and Guaranty with Perceptive Credit Holdings III, LP (the “Perceptive Credit Facility”), as administrative agent for the lenders, which we refer to as the Perceptive Credit Facility, and accretion of the debt discount on the Perceptive Credit Facility as well as the change in the fair value of our derivative liabilities and earn-out liabilities that occurred during the period. In addition, it includes the accretion of the interest of the seller financing in connection with the Waypoint asset acquired in November 2021. See Note 8 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.We expect interest expense to increase as interest rates such as the London Interbank Offered Rate (“LIBOR”) and the Secured Overnight Financing Rate (“SOFR”) increase.
Interest income consists of interest earned on cash balances held in interest-bearing accounts. We expect our interest income will fluctuate based on rising interest rates, our cash balances on hand, the timing and ability to raise additional funds, as well as the amount of expenditures for our commercial products and R&D for our product candidates and ongoing business operations.
Loss on issuance of convertible preferred stock
In December 2020 and February 2021, the Company issued shares of Series D-1 and D-2 convertible preferred stock to investors. The shares are recorded at their estimated fair market value on the date of issuance. In
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 105
connection with the Series D-1 preferred stock and D-2 preferred stock financing, the Company recorded a loss on the issuance of convertible preferred stock for the years ended December 31, 2021 and 2020, of $2.1 million and $16.8 million, respectively, which represents the amount by which the estimated fair value of the shares exceeded the sale price.
Loss/gain on extinguishment of debt
In connection with the extinguishment of the SVB Term Loan (as defined below), in June 2020, we recorded a loss on extinguishment of debt of $1.0 million for the year ended December 31, 2020.
Income taxes
Deferred income taxes are provided for the effects of temporary differences between the amounts of assets and liabilities recognized for financial reporting purposes and the amounts recognized for income tax purposes. Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. We regularly assess the need to record a valuation allowance against net deferred tax assets if, based upon the available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized. Accordingly, we have recorded a valuation allowance of $66.7 million and $40.3 million, as of December 31, 2021 and 2020, respectively. The future realization of deferred tax assets is subject to the existence of sufficient taxable income of the appropriate character (e.g., ordinary income or capital gain) as provided under the carryforward provisions of tax law. We consider the scheduled reversal of deferred tax liabilities (including the effect in available carryback and carryforward periods), future projected taxable income (including the character and jurisdiction of such income), and tax-planning strategies in making this assessment. Unrecognized tax benefits arise when the estimated benefit recorded in the financial statements differs from the amounts taken or expected to be taken in a tax return because of the considerations described above. As of December 31, 2021, we had no unrecognized tax benefits.
Financial Highlights
Year-over-year product revenue grew significantly, and R&D for our product candidates and ongoing business operations.
Financial Highlights
Year-over-year product revenue grew by approximately 178% to $10.4 million from $3.7 million primarily due to an increase in sales of reSET and reSET-O, which increased approximately 24x year over yearunder Access Agreements. Year-over-year total revenue declined due to non-recurring $9collaboration revenue grew by approximately 90% to $0.9 million from $0.5 million primarily due to the development work completed on a Japanese-language digital therapeutic for the treatment of sleep/wake disorders for the Japanese market in collaboration with SoftBank Corp. that was completed in 2022. We also recognized subscription, support, and professional services revenue of $1.24 million of collaboration revenue recognized infor the year ended December 31, 20202022, under an agreement terminated in March 2020 anda new product offering under whicha no further revenue willpilot offering of a be recognizednew product by the Medicaid program of a state government .
We incurred a net losslosses of $6575.15 million and $97.0 million for the years ended December 31, 2021, and 2020, respectively, representing a period-over-period decrease of $31.9 million or 32.9%. This decrease was primarily due to a change in the fair value of the earn-out liabilities of $47.065.1 million for the year ended December 31, 2021, and a $14.8 million decrease in loss on issuance of convertible preferred stock, offset by an $8.8 million decrease in collaboration revenues. In addition, we had a $2.62022 and 2021, respectively, representing a period-over-period increase in our net loss of $10.3 million or 15.9%. This increase in the net loss was primarily due to a $22.7 million increase in personnel-related expenses, primarily related to new hires as we expanded our commercial team from an average of 59 people in the year ended December 31, 2020, to an averageincreased headcount during the first half of 2022 compared to 2021, and severance costs associated with the July and November 2022 reductions in workforce, partially offset by $8.5 million increase in total revenue period over period and a gain related to the change in fair value of 94 peoplethe Public Warrants and the Private Placement Warrants of $6.4 million for the year ended December 31, 2021, and as we prepared to become a public company. Further, we had a $5.0 million increase in marketing-related expenses, a $4.1 million increase in professional fees, and a $22022, compared to a $0.3 million loss for the year ended December 31, 2021. We had an average of 245 full-time employees for the year ended December 31, 2021, and an average of 267 full-time employees for the year ended December 31, 2022. We had a $3.4 million increase in interest expensecosts related to the borrowing of $30.0 million under the Perceptive Credit Facility on June 30, 2020. In June 2020, the Company recorded a $1.0 million loss on extinguishment of debt. being a public company period over period, primarily related to our directors’ and officers’ insurance.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 111
To date, we have funded our operations primarily with proceeds from sales of Legacy Pear’s convertible preferred stock, proceeds as a result of the business combinationBusiness Combination, payments received in connection with collaboration and license agreements, product sales,our revenue contracts and proceeds from borrowings under various credit facilities. Since our inception, weWe have received gross cash proceeds of $175.3 million as a result of the business combination (see Note 3Business Combination (see Note 3, Business Combination, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K), and gross cash proceeds of $268.2 million from sales of our Legacy Pear’s convertible preferred stock,; andwe currently have $30.0 million of debt outstanding under a credit facility. During the year ended December the Perceptive Credit Facility.
Recent Events
Exploring Strategic Alternatives
We have engaged MTS Health Partners, L.P. (“MTS”) as our advisor to assist with the exploration of strategic alternatives. MTS is providing a range of advisory services aimed to enhance stockholder value. The alternatives to be considered may include, but are not limited to, the sale of all or substantially all of our assets; a strategic merger or other business combination transaction; or another change of control transaction between us and a third party. If the strategic process is unsuccessful, our Board may decide to pursue a liquidation or obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection. In the event of such liquidation, bankruptcy case, or other wind-down event, holders of our securities will likely suffer a total loss of their investment.
Waiver of Debt Covenants and Negotiations with Lender
The Company was not in compliance with the minimum revenue covenant for the quarter ending December 31, 2020, we completed a tender offer to purchase $322022, of $18.0 million and was in aggregate principal amount of our common stock in connection with the Legacy Pear Series D-2 preferred stock offering that was conducted in conjunction with the Legacy Pear Series D-1 stock offering.compliance with the minimum cash balance covenant requirement of $5.0 million. The Company obtained a waiver of the minimum trailing twelve (12) month revenue requirement for the quarter ended December 31, 2022 and March 31, 2023, respectively. The Company also obtained a waiver pertaining to the existence of a “going concern” qualification in the accompanying opinion of the Company’s auditors in its Annual Report on Form 10-K and any resulting event of default.
However, as of the date of this filing, Perceptive has alleged that certain defaults or events of default have occurred and are continuing under the terms of the Perceptive Credit Facility. Perceptive has not delivered any formal notice of Default or Event of Default. To the extent that any such allegations are valid, Perceptive would have certain rights and remedies under the Perceptive Credit Facility. The Company disputes the allegations and is in discussions with Perceptive to resolve this dispute and otherwise to address the Company’s obligations under the Perceptive Credit Facility. There can be no assurances that such discussions will result in any resolution, and any resolution, or the lack of any resolution, may result in Perceptive exercising remedies under the Perceptive Credit Facility. If the acceleration of the debt were to occur, we could also be required to pay a prepayment penalty of $3.6 million.
At-the-Market (ATM) Offering
On January 3, 2023, the Company entered into an ATM offering agreement (the “ATM Agreement”) with H.C. Wainwright & Co., LLC (“Wainwright”) and Virtu Americas LLC (“Virtu” and, collectively with Wainwright, the “Managers” and each, a “Manager”), pursuant to which the Company may offer and sell, from time to time through the Managers, shares of the Company’s Class A common stock. The ATM Agreement authorized an aggregate gross proceeds of up to $150.0 million. Sales of common stock through the Manager could be made by any method that is deemed an “at-the-market” offering as defined in Rule 415 promulgated under the Securities Act of 1933, as amended, including by means of ordinary brokers’ transactions at market prices, in block transactions or as otherwise agreed by the Company and the Managers. The Company will pay the designated Manager a commission of up to 3.0% of the aggregate gross proceeds from any Shares sold by the designated Manager and provide the Managers with customary indemnification and contribution rights, including for liabilities under the Securities Act. The Company also will reimburse the Managers for certain specified expenses in connection with entering into the ATM Agreement.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 106112
Recent Events
Business Combination
On December 3, 2021 (the “Closing Date”), we consummated a business combination, pursuant to the terms ofAs of March 31, 2023, the Company has the business combination agreement (“Business Combination Agreement”) dated June 21, 2021. Upon the consummation of the Business Combination, Oz Merger Sub, a newly formed subsidiary of THMA, merged with and into Pear, with Pear surviving. THMA, was renamed Pear Therapeutics, Inc (collectively, “Pear”) and Pear Therapeutics, Inc. was renamed Pear Therapeutics (US), Inc. (“Legacy Pear”). Legacy Pear is deemed the accounting predecessor and the post-company successor SEC registrant, which means Legacy Pear financial statements for previous periods will be disclosed in this Form 10-K. Future period reports filed with the SEC will include Pear Therapeutics, Inc. and its subsidiaries.
The business combination was accounted for as a reverse recapitalization. Under this method of accounting, THMA was treated as the acquired company for financial statement reporting purposes. The most significant change in the post-combination Company’s reported financial position and results was an increase in cash of $175.0 million. We paid $32.8 million in transaction costs relating to the business combination. We recorded a liability related to the Public and Private Placement Warrants of $16.5 million and $95.4 million related to the earn-out shares that holders of Legacy Pear Common Shares and Legacy Pear Preferred Shares prior on the Closing Date who received the contingent right to receive up to 12,395,625 additional shares ofsold 843,281 shares of its Class A common stock (the “Earn-Out Shares”) upon the achievement of certain earn-out targetsunder the ATM Agreement resulting in proceeds to the Company of $1.0 million, net of offering costs.
Restructuring, Reductions in Workforce, and Paused Investment in our Pipeline Product Candidates
On July 25, 2022, the Company restructured its operations to narrow its near-term business focus and reduce its workforce due to the macroeconomic environment. As a result of the restructuring, the Company incurred a charge of $0.9 million primarily associated with the severance and health insurance expenses related to 25 full-time employees, representing approximately 9% of full-time employee at the time of the restructuring and reduction in workforce. In connection with this restructuring, we reduced spending on our pipeline candidates, discovery programs, business development, and the Company's dual platform in order to prioritize certain of its commercial efforts and will continue to reduce costs in each of these areas. The direct costs associated with the first restructuring and reduction in workforce were recorded in the quarter ended September 30, 2022.
As a consequence of the business combination, we became the successor to an SEC-registered and Nasdaq-listed companyOn November 14, 2022, we announced a second reduction in workforce, further reducing our headcount by approximately 59 employees, or approximately 22% of our full-time employees as of September 30, 2022, due to the worsening macroeconomic environment. The Company recorded $2.5 million in severance charges in connection with the second reduction in workforce, related to severance payments, employee benefits, and related costs, in the fourth quarter of 2022. In addition, the Company recorded a stock-based compensation and we have hired additional personnel and implemented procedures and processescorresponding payroll tax expense benefit of $0.1 million related to address public company regulatory requirements and customary practices. We expect to incur additional annual expenses as a public company formodifications of equity awards for employees impacted by the reduction in workforce.
The estimated costs that the Company expects to incur in connection with the reduction in forces and cost saving initiatives are subject to a number of assumptions, and actual results may differ significantly from these estimates. The Company may also incur additional costs not currently contemplated due to events that may occur as a result of, among other things, directors’or that are associated with, the reductions in workforce and officers’ liability insurance, director fees,cost saving initiatives. In the future, there may and additional internal and external accounting and legal and administrative resources, including increased audit and legal feesalso be incremental one-time charges associated with non-work force related cost savings actions.
Economic Conditions (Impact of COVID-19)
In March 2020On January 30, 2023, the World Health Organization declared the global outbreak of COVID-19 to be a pandemic. The pandemic has significantly impacted the economic conditions in the US, as federal, state and local governments react to the public health crisis, creating significant uncertainties inBiden Administration announced it will end the public health emergency (and national emergency) declarations related to COVID-19 on May 11, 2023. While the US economy. The downstream impact of various lockdown ordersCOVID-19 pandemic has not materially adversely affected our financial results and related economic pullback affect our business and our customers to varying degrees. There Company is business operations through December 31, 2022, COVID-19 continues to present risks to the Company, and we continue to closely monitoringmonitor the impact of COVID-19the pandemic on all aspects of its business, including how it will impact its customers, patients, employees, suppliers, vendors, and business partners. The Company isour business. We are unable to predict the specific impact that COVID-19 may have on its business, financial position, and operations moving forward due to the numerous uncertainties. Any estimates made herein may change as new events occur and additional information is obtained, and actual results could differ materially from any estimates made herein under different assumptions or conditions.
For further details see the information under the heading “Risk Factors” in Part I, Item 1A in this Form 10-K. Pear is unable to predict the full impact that the COVID-19 pandemic will have on its future results of operations, liquidity,(including the emergence of new variants) will have on our financial position and operating results in the future.
Impact of Inflation
We are experiencing rising costs for certain inflation-sensitive operating expenses, such as labor, and certain service providers that are heavily dependent on labor. We do not believe these impacts were material to net income during the year ended December 31, 2022. However, significant sustained inflation driven by the macroeconomic environment or other factors could negatively impact our margins, profitability, and results of operations in future periods.
FL Medicaid Coverage
Effective January 1, 2023, reSET® and financial condition duereSET-O® were added to numerous uncertainties, includingFlorida Medicaid Preferred Drug List (PDL). reSET and reSET-O, the duration of the pandemic and the actions that may be taken by government authorities across the US. However, COVID-19 is not expected to result in any significant changes in costs going forward. As a result of COVID -19 pandemic we shifted our workforce to a hybrid model in which employees in one of our three offices work both remotely and onsite, and we anticipate we will continue to use this model going forward. In addition, our workforce has deep domain knowledge across a range of healthcare, technology, and general business, which was partially achieved by having certain of our employees working remotely across the US. Pear will continue to monitor the performance of its business and assess the impacts of COVID-19only FDA-authorized PDTs for the treatment of substance use disorder and opioid use disorder, respectively.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 107113
Results of Operations
The tabletables and discussion below present the results for the periods indicated:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Year Ended December 31, | | Change |
| (in thousands, except percentages) | | 2022 | | 2021 | | $ | | % |
| Revenues | | | | | | | | |
| Product revenue | | $ | 10,417 | | | $ | 3,748 | | | $ | 6,669 | | | 178 | % |
| Collaboration and license revenue | | 872 | | | 460 | | | 412 | | | 90 | % |
| Subscription, support, and professional services revenue | | $ | 1,405 | | | $ | — | | | 1,405 | | | * |
| Total revenues | | 12,694 | | | 4,208 | | | 8,486 | | | 202 | % |
| Cost and operating expenses: | | | | | | | | |
| Cost of revenue | | 8,182 | | | 5,233 | | | 2,949 | | | 56 | % |
| Research and development | | 48,311 | | | 37,041 | | | 11,270 | | | 30 | % |
| Selling, general, and administrative | | 79,551 | | | 67,619 | | | 11,932 | | | 18 | % |
| Total cost and operating expenses | | 136,044 | | | 109,893 | | | 26,151 | | | 24 | % |
Loss from operations | | (123,350) | | | (105,685) | | | (17,665) | | | (17) | % |
| Other income (expenses): | | | | | | | | |
| Interest and other expenses, net | | $ | (3,892) | | | (4,144) | | | 252 | | | 6 | % |
Change in estimated fair value of earn-out liability | | 45,339 | | | 47,038 | | | (1,699) | | | (4) | % |
Change in estimated fair value of warrant liabilities | | 6,412 | | | (298) | | | 6,710 | | | * |
| | | | | | | | |
Loss on issuance of Legacy Pear convertible preferred stock | | $ | — | | | (2,053) | | | 2,053 | | | * |
| Total other income (expense) | | 47,859 | | | 40,543 | | | 7,316 | | | 18 | % |
| Net loss | | $ | (75,491) | | | $ | (65,142) | | | $ | (10,349) | | | 16 | % |
__________________
* Percentage change not meaningful.
Product revenue—Product revenue for the year ended December 31, 20212022, was $10.4 million, compared to $3.7 million, an for the year ended December 31, 2021. The increase of $3.6 million, or 24x, compared to the prior yearwas primarily driven by increased bulk sales of reSET and reSET-O under Access Agreements.
Collaboration and license revenue—Collaboration and license revenue was $0.5 million for the year ended December 31, 2021, compared to $9for the year ended December 31, 2022, was $0.9 million, compared to $0.5 million for the year ended December 31, 2021, primarily due to the development work completed on a Japanese-language digital therapeutic for the treatment of sleep/wake disorders for the Japanese market in collaboration with SoftBank Corp. that was completed in 2022. See Part III Item 13, “Certain Relationships and Related Transactions, and Director Independence” below for information regarding the related party nature of the agreement.
Subscription, support, and professional services revenue - Subscription, support, and professional services revenue for the year ended December 31, 2022, was $1.4 million, under a pilot offering of a new product by the Medicaid program of a state government.
Cost of revenue—Cost of revenue for the year ended December 31, 2022, was $8.2 million, compared to $5.2 million for the year ended December 31, 2020. For the year ended December 31, 2020, we recognized $9.2 million in connection with the NIBR Agreement, which was terminated in June 2020. See Note 9 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Cost of product revenue—Cost of product revenue for the year ended December 31, 2021, was approximately $5.2 million, an increase of approximately $32021. This increase of $2.59 million compared to the prior yearwas primarily due to an increased product revenue as discussed above.
Due tonumber of customers and clinics served associated with our increased commercialization efforts, we saw an increase in our pharmacy costs,Access Agreements, and minimum royalties related to licensing agreements for commercialized products, pharmacy, and hosting costs for our PDTs. In additionAdditionally, in November 2020, the Company launched Somryst, which further increased our cost of product revenue.
Research and development—R&D expenses were $37.0 million and $28.1 million for the years ended December 31, 2021 and 2020, respectively. The increase of $9.0 million was primarily due to the following:
•An increase of $3.9 million of personnel-we recorded $0.8 million of impairment expense on the intangible assets related coststo and an increase of $1.0 million of recruiting fees as we continued shifting our software development work from external to internal resources, and coinciding with an increase in average R&D headcount from 88acquired technology used in our Somryst commercial product for the year ended December 31, 2020 to 107 for the year ended December 31, 2021; and 31, 2022. Several factors positively contributed to an increased efficiency of implementing our products in clinics during 2022, including economies of scale and focused
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 108114
•go-to market efforts resulting in increased productivity of our implementation team. Cost of revenue represented 64.5% and 124.4% of total revenue for the years ended December 31, 2022 and 2021, respectively. We expect cost of revenue in 2023 to decrease as a percentage of revenue as revenue increases from further economies of scale.
Research and developmentIncreases of $2.1 million of consultant costs, $1.0 million of professional and recruiting fees, and $1.0 of software expenses. —R&D expenses for the year ended December 31, 2022, were $48.3 million, compared to $37.0 million for the year ended December 31, 2021. The increase of $11.3 million was primarily due to an increase of $8.7 million of personnel-related costs from higher average headcount during the first half of 2022 and subsequent severance costs from the July and November 2022 reductions in workforce, and $2.1 million of impairment of a previously acquired intangible asset. We anticipate decreases in R&D costs compared to prior periods as a result of the restructuring and reductions in workforce as well as us pausing any further investment in our pipeline of product candidates.
Selling, general, and administrative—SG&A expenses were $67.6 million and $56.2 million for the years ended December 31, 2021 and 2020, respectively. The increase of $11.4 million was primarily due to further building out of our commercial operations, which resulted in the following increases:
•$5.3 million in personnel-related costs as a result of an increase in average headcount from approximately 90 in 2020 to approximately 120 in 2021, primarily the commercial team;
•$5.0 million in marketing and advertising costs as a result of targeted media and market awareness, education, and advocacy initiatives;
•$2.2 million in professional fees, including increases in legal and accounting fees as a result of becoming a public company;
•$1.4 million of transaction costs directly incurred in connection with the Business Combination;
•$1.3 million of depreciation and amortization expense, primarily related to amortization of software used in our patient support center;
•$1.2 million of software related expenses; and
•$1.1 million of license and subscription costs.
The results for the year ended December 31, 2020, included afor the year ended December 31, 2022, were $79.6 million, compared to $67.6 million for the year ended December 31, 2021. The increase of $711.29 million stock compensation charge from the tender offer used to purchase and re-sell Legacy Pear shareswas primarily due to an increase of $12.1 million in personnel-related costs from higher average headcount during the first half of 2022 and subsequent severance costs from the July and November 2022 reductions in workforce. These increases were offset by reduction in our marketing spend. We anticipate decreases in SG&A costs compared to prior periods as parta result of the Legacy Pear Series D preferred share issuance.restructuring and reductions in workforce, reduced marketing spend as well as reductions in our travel expenses and cost savings related to the departure of our Chief Commercial Officer .
Interest and other (expense) income, net—Interest and other income (expense), net was, for the year ended December 31, 2022, was an expense of $3.9 million compared to an expense of $4.1 million for the year ended December 31, 2021,. as compared to expense of $1.8 million for the year ended December 31, 2020. The increase in interest expense was primarily related to $4.3 million of interest related to the Perceptive Credit Facility that closed on June 30, 2020, compared to $2.5 million of interest under the Perceptive Credit Facility and SVB Term Loan for the year ended December 31, 2020. For the year ended December 31, 2020, the Company also recognized $0.7 million of interest income on cash This decrease is mainly the result of $1.1 million of interest income on cash equivalents and investments. short-term investments as a result of higher investment balances due to the proceeds of the Business Combination that closed in December 2021 and a $0.6 million gain from the change in the fair value of the embedded debt derivative recorded during year ended December 31, 2022.
Change in fair value of earn-out liabilities—For the year ended December 31, 20212022, the Companywe recognized a $4745.03 million gain as a result of the change in fair value of the earn-out liabilities from its initial recognition on, compared to a gain of $47.0 million for the December 3, 2021, the date of the Merger, untilyear ended December 31, 2021 primarily as a result of the fluctuations in the underlying share price of our Class A common stock.
Change in fair value of warrant liabilities—For the year ended December 31, 2022, we recognized a gain of $6.4 million related to the Public Warrants and the Private Placement Warrants. We recognized a $0.3 million loss for the year ended December 31, 2021, related to the Legacy Pear Warrants, which were exercised in 2021 prior to the business combination, for the year ended December 31, 2021, the Company recognized a $8.3 million loss due to the change in fair value of outstanding warrant liabilities, compared to a $0.8 million loss on Legacy Pear Warrants for the year ended December 31, 2020. The loss on the Legacy Pear Warrants for the year ended December 31, 2021 was offset by a gain on the Public and Private Placement Warrants $8.0 million, which were issued by THMA prior to the business combinationBusiness Combination.
Loss on issuance of extinguishment of debt—In June 2020, the Company paid the outstanding principal on the SVB Term Loan prior to the maturity date resulting in a loss on extinguishment of debt of $1.0 million for the year ended December 31, 2020. See Note 7 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Loss on issuance of legacyLegacy Pear convertible preferred stock—In December 2020, the Company issued shares of Legacy Pear Series D-1 and D-2 convertible preferred stock. The shares are recorded at their estimated fair market value on the date of issuance. In connection with the Legacy Pear D-1 and D-2 Preferred Stock Financing, the Company recorded a loss of $16.8 million, net of issuance costs, for the year ended December 31, 2020, which represents the amount by which the estimated fair value of the shares exceeded the sale price. In February 2021, the Company issued additionalIn February 2021, the Company issued shares of Legacy Pear Series D-1 Preferred Stock. The shares are recorded at their
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 109
estimated fair market value on the date of issuance. In connection with the Legacy Pear Series D-1 Preferred Stock, the Companywere recorded at their estimated fair market value on the date of issuance. In connection with the Legacy Pear Series D-1 Preferred Stock, we recorded a loss of $2.1 million for the year ended December 31, 2021, which represents the amount by which the estimated fair value of the shares exceeded the sale price, net of issuance costs. See Note 10 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Income tax—We did not incur income tax expenses for the yearsyear ended December 31, 2022 and 2021 or 2020. Given our lack of prior earnings history, we have a full valuation allowance primarily related to our net operating loss and R&D credit carryforwards that we do not consider more likely than not to be realized.
Liquidity and Capital Resources
Since our inception, our primary sources of capital have been proceeds from sales of Exploring Strategic Alternatives
We require substantial additional capital to sustain our operations and pursue our growth strategy, including the development of our product candidates. The Company has engaged MTS as our investment bank to assist with the exploration of strategic alternatives that may include, but are not limited to, the sale of all or substantially all of our assets; a strategic merger or other business combination transaction; or another change of control transaction between us and a third party. If a strategic process is unsuccessful, our Board may decide to pursue a liquidation or
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 115
obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection. These factors raise substantial doubt about our ability to continue as a going concern. For more information, refer to “—Liquidity and Capital Resources” below and Note 1, Nature of the Business, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K.
Sources of Liquidity
Since our inception, our primary sources of capital have been proceeds from sales of Legacy Pear convertible preferred stock, payments received in connection with collaboration agreements, payments received on our revenue contracts, include the sale of our products,, proceeds from borrowings under various credit facilities, and the business combination that occurred December 3, 2021. See Recent Events in Part II, Item 7 of this Form 10-K and Note 3Business Combination. See Note 3, Business Combination, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
We have three commercial products: reSET, reSET-O, and Somryst. The revenue from the sale of these products at the present time is not sufficient to cover the operating costs incurred. Our ability to achieve sufficient revenue to cover our costs is highly dependent on our PDTs achieving and maintaining broad market acceptance by patients and physicians and obtaining reimbursement from third-party payors. The Company has incurred recurring losses from inception and anticipates net losses and negative operating cash flows for the near future. For the years ended December 31, 2021 and 2020, we incurred net operating losses of $65.1 million and $97.0 million, respectively.
As of December 31, 2021 and 2020, we had an accumulated deficit of $248.0 million and $182.8 million, respectively. As of December 31, 2021 and 2020, we had outstanding debt of $27.0 million and $26.3 million, net of debt issuance costs, respectively. Our cash flows may fluctuate and are difficult to forecast and will depend on many factors. As of December 31, 2021Our cash flows may fluctuate and are difficult to forecast and will depend on many factors. As of December 31, 2022 and 2020December 31, 2021, we had cash and cash equivalents of $16948.6 million and $110.9 million, respectively.
Our primary uses of capital are, and we expect will continue to be for the near future, funding operating activities, including expanding our commercial operations and further development of our product pipeline. We have in the past and we expect in the future to capitalize labor costs related to the development of our internal-use software. We may also pursue acquisitions, investments, joint ventures, and other strategic transactions.
In the future, we will need to raise additional capital to pursue our growth strategy and support continuing operations. Until such time as we can generate significant revenue to fund operations, we expect to seek additional capital from the issuance of equity, debt, or other capital transactions. If sufficient funds on acceptable terms are not available when needed, we will be required to significantly reduce our operating expenses. Further, we may be unable to increase our revenue, raise additional funds, or enter into such other agreements or arrangements when needed on favorable terms, or at all. If we fail to raise capital or enter into such agreements as and when needed, we may have to significantly delay, scale back or discontinue the development and commercialization of one or more of our product candidates and other strategic initiatives. We are also subject to various covenants related to the Perceptive Credit Facility, and given the substantial doubt about our ability to continue as a going concern, there is a risk that we may not meet our covenants in the future. As of December 31, 2021 and 2020, we concluded that these circumstances raise substantial doubt about our ability to continue as a going concern.
Cash and cash equivalents
As of December 31, 2021 we had $169.6 million of cash and cash equivalents. Our future capital requirements may vary from those currently planned and will depend on various factors, including the timing and extent of R&D spending and spending on other strategic business initiatives, including expanding our commercial operations.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 110
Liquidity Risks
We expect to incur substantial additional expenditures in the near term to support our ongoing activities, including as a result of operating as a public company. We expect to continue to incur net losses for the foreseeable future. Our ability to fund our product development and clinical operations as well as commercialization of our product candidates will depend on the amount and timing of cash available to fund operations. Our future liquidity and capital funding requirements will depend on numerous factors, including:
•our revenue growth;
•the ability to obtain third-party payor reimbursement for our current products;
•the amount and timing of sales and other revenues from our product candidates, if approved, including the sales price and the availability of coverage and adequate third-party payor reimbursement;
•our commercial activities, including sales and marketing;
•our R&D efforts;
•the emergence and effect of competing or complementary products;
•the outcome, timing, and cost of meeting regulatory requirements established by the FDA, or comparable foreign regulatory authorities;
•the progress, timing, scope, and costs of our preclinical studies, clinical trials, potential future clinical trials, and other related activities;
•the costs of commercialization activities for any of our product candidates that receive marketing authorization, including the costs and timing of establishing product sales, marketing and hosting capabilities, or entering into strategic collaborations with third parties to leverage or access these capabilities;
•the cash requirements of developing our programs and our ability and willingness to finance their continued development;
•the cash requirements of any future discovery of product candidates;
•our ability to retain our current employees and the need and ability to hire additional management and sales, technical and medical personnel;
•the time and cost necessary to respond to technological and market developments, including other products that may compete with one or more of our product candidates;
•debt service requirements;
•the extent to which we acquire or invest in business, products, or technology; and
•the impact of the COVID-19 pandemic.
A change in the outcome of any of these or other variables with respect to the development of any of our product candidates could significantly change the costs and timing associated with the sale of our products or the development of product candidates. Further, our operating plans may change in the future, and we may need additional funds to meet operational needs and capital requirements associated with such operating plans. See the information under the heading “Risk Factors” included in Part I, Item 1A this Form 10-K.
Because of the numerous risks and uncertainties associated with the development and commercialization of our product candidates, we are unable to estimate the amounts of increased capital outlays and operating expenditures associated with our current and anticipated product development programs.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 111
Funding Requirements
Please see the risks associated with our substantial capital requirements explained more fully under the heading “Risk Factors-The Company will need substantial additional funding, and if it is unable to raise capital when needed or on terms favorable to the Company, the Company’s business, financial condition, and results of operation could be materially and adversely affected” in Part I, Item 1A of this Form 10-K.
Debt Financing and Covenants
3 million and $169.6 million, respectively. Based on our current operating plans inclusive of the July 2022 restructuring plan and reduction in force, the November 2022 reduction in force and further cost saving initiatives as well as strategic review process, we have sufficient cash and cash equivalents to fund our operating expenses and capital expenditures into the second quarter of 2023.
Debt Financing and CovenantsBorrowings under our $50.0 million—On June 30, 2020, the Company entered into the Perceptive Credit Facility with Perceptive. Outstanding borrowings under our secured Perceptive Credit Facility were $30.0 million as of December 31, 2021 and 2020, which was used to extinguish the former SVB Term Loan and for general business purposes December 31, 2022 and December 31, 2021. The Perceptive Credit Facility matures in June 2025. We are required to payPrior to February 1, 2023, the Perceptive Credit Facility bore interest through maturity at a variable rate of interest based upon the one-month LIBOR rate plus 11.0%, subject to a LIBOR floor of 1.0%. As of December 31, 20212022, the annual interest rate was 1215.0%1%. On January 13, 2023, the Company and Perceptive executed the First Amendment to the Perceptive Credit Facility, which replaced LIBOR with the Secured Overnight Financing Rate (“SOFR”), effective February 1, 2023. As of and for the year ended December 31, 2022, the effect of switching from LIBOR to SOFR would not have been material to the Company’s consolidated financial statements. The Company is required to make interest-only payments until May 31, 2024, after which point the Company will be required to make monthly payments of principal equal to 3.0% of the then outstanding principal until maturity on June 30, 2025.
The Perceptive Credit Facility is secured by substantially all of the assets of the Company, including our intellectual property. The Perceptive Credit Facility requires the Company to (i) maintain a minimum aggregate cash balance of $5.0 million in one or more controlled accounts, and (ii) as of the last day of each fiscal quarter commencing with the fiscal quarter ending March 31, 2022, report revenues for the trailing 12-month period that exceed the amounts that range from $518.80 million for the fiscal quarter ending March 31, 2022, to $125.0 million for the fiscal quarter ending March 31, 2025. For the quarter ending December 31, 2022, the trailing 12-month period revenue requirement was $18.0 million. The Perceptive Credit Facility contains various affirmative and negative covenants that limit the Company’s ability to engage in specified types of transactions. The Company was not in compliance with the covenantstrailing twelve month revenue covenant under the Perceptive Credit Agreement as of December 31, 2021.
See Note 7 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information. In the future, we may seek to obtain other additional sources of financing, including incurring term debt or issuing equity or debt securities.
As of December 31, 2021 and 2020, we had $0Facility as of December 31, 2022, however we requested and were granted a waiver on the trailing 12-month revenue requirement for both the quarter ended December 31, 2022 and March 31, 2023, respectively. In addition, on February 28, 2023, the Company obtained a waiver pertaining to the existence of a “going concern” qualification in the accompany opinion of the Company’s auditors in this Form 10-K and any resulting event of default.
As of the date of this filing, Perceptive has alleged that certain defaults or events of default have occurred and are continuing under the terms of the Perceptive Credit Facility. Perceptive has not delivered any formal notice of Default or Event of Default. To the extent that any such allegations are valid, Perceptive would have certain rights and remedies under the Perceptive Credit Facility. The Company disputes the allegations and is in discussions with Perceptive to resolve this dispute and otherwise to address the Company’s obligations under the Perceptive Credit Facility. There can be no assurances that such discussions will result in any resolution, and any resolution, or the lack of any resolution, may result in Perceptive exercising remedies under the Perceptive Credit Facility. As of the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 116
date of this filing, if the acceleration of the debt were to occur, we could also be required to pay a prepayment penalty of $3.6 million.
As of December 31, 2022 and December 31, 2021, we had outstanding debt of $27.4 million and $27.0 million, net of debt issuance costs of $2.6 million and $3.0 million, respectively. See Note 7, Indebtedness, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Product Sales—We have three commercial products: reSET, reSET-O, and Somryst. The revenue from the sale of these products at the present time is not sufficient to cover the operating costs incurred. Our ability to achieve sufficient revenue to cover our costs is highly dependent on our PDTs achieving and maintaining broad market acceptance by patients and physicians and obtaining reimbursement from third-party payors.
Subscription, Support, and Profession Services Revenue—In the fourth quarter of 2022, we entered into a 16-month pilot program to allow eligible Medicaid members in that state take part in a structured 24-week outpatient program that will be followed by at least six months of additional recovery support services. Under the pilot we will deliver, implement, and manage this program, specifically by managing the electronic tracking and distribution of incentives to the states Medicaid members who participate in this contingency management pilot. The contract is for an amount up to $4.0 million plus reimbursements for the contingency management. We recognized $1.4 million under the terms of this contract for year ended December 31, 2022.
Employee Stock Purchase Plan—Under the Company’s 2021 Employee Stock Purchase Plan (the “2021 ESPP”), 359,910 shares of the Company’s Class A common stock were issued for total consideration of $0.4 million during the year ended December 31, 2022.
At-The-Market Equity Offering—On January 3, 2023, the Company entered into an ATM Agreement with the Managers, pursuant to which the Company may offer and sell, from time to time through the Managers, shares of the Company’s Class A common stock for aggregate gross proceeds of up to $150 million. As of March 31, 2023, the Company has sold 843,281 shares of its Class A common stock under the ATM Agreement resulting in proceeds to the Company of $1.0 million, net of offering costs.
In the future, we may seek to obtain other additional sources of financing, including incurring term debt or issuing equity or debt securities.
Historical Cash Flows
We have incurred recurring losses from inception and anticipate net losses and negative operating cash flows for the near future. For the year ended December 31, 2022 and 2021, we incurred net operating losses of $75.5 million and $65.1 million, respectively.
As of December 31, 2022 and December 31, 2021, we had an accumulated deficit of $323.5 million and $248.0 million, respectively.
Capital Resources
Our primary uses of capital are, and we expect will continue to be for the near future, funding operating activities. We have in the past and we expect in the future to capitalize labor costs related to the development of our internal-use software.
We need to raise additional capital to pursue our growth strategy and support continuing operations. Until such time as we can generate significant revenue to fund operations, we expect to seek additional capital from the issuance of equity, debt, or other capital transactions or a strategic transaction. On July 25, 2022, we restructured our business operations to narrow our near-term business focus and decreased our workforce to reduce our operating expenses. We announced a further reduction in workforce on November 14, 2022. Despite our recent restructuring, we need to raise capital to support our operations absent a strategic transaction. In February 2023, we hired an investment bank to explore strategic alternatives. We may be unable to complete a strategic transaction, increase our revenue, raise additional funds, or enter into such other agreements or arrangements
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 117
when needed on favorable terms, or at all, our Board may decide to pursue a liquidation or obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection.
As of the date of this filing, Perceptive has alleged that certain defaults or events of default have occurred and are continuing under the terms of the Perceptive Credit Facility. Perceptive has not delivered any formal notice of Default or Event of Default. To the extent that any such allegations are valid, Perceptive would have certain rights and remedies under the Perceptive Credit Facility. The Company disputes the allegations and is in discussions with Perceptive to resolve this dispute and otherwise to address the Company’s obligations under the Perceptive Credit Facility. There can be no assurances that such discussions will result in any resolution, and any resolution, or the lack of any resolution, may result in Perceptive exercising remedies under the Perceptive Credit Facility.
We are also subject to various covenants related to the Perceptive Credit Facility, as well as the material adverse clause there is substantial doubt about our ability to continue as a going concern. As of December 31, 2022, we concluded that the above circumstances raise substantial doubt about our ability to continue as a going concern. See Note 7, Indebtedness, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Cash and Cash Equivalents
As of December 31, 2022, we had $48.4 million in a letter of credit outstanding in connection with our leased property in San Francisco, California3 million of cash and cash equivalents. In July 2022 and November 2022 we completed reductions in force and took measures to significantly reduce our operating costs, including suspension of the development of our pipeline product candidates. As noted elsewhere in this report, including Note 1, Nature of the Business, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K there is substantial doubt as to our ability to fund our planned operations for the next twelve months and to continue to operate as a going concern.
Liquidity Risks
We will require additional capital in order to pursue our strategic objectives and for the Company to survive. We have hired an investment bank to assist with the exploration of a strategic alternatives. We expect to incur substantial additional expenditures in the near term to support our ongoing activities, including costs related to being a public company. Further, we expect to continue to incur net losses for the foreseeable future and have sufficient cash on hand to fund operations into the second quarter of 2023. Our ability to fund our product development and clinical operations as well as commercialization of our product candidates will depend on the amount and timing of cash available to fund operations. Our future liquidity and capital funding requirements will depend on numerous factors, including:
•our ability to retain our current employees;
•our revenue growth;
•the ability to obtain third-party payor reimbursement for our current products;
•debt service requirements;
•our commercial activities, including sales and marketing;
•our ability to reduce or contain certain costs and expenses;
•the emergence and effect of competing or complementary products;
•the outcome, timing, and cost of meeting regulatory requirements established by the FDA, or comparable foreign regulatory authorities;
•the cash requirements of developing our programs and our ability and willingness to finance their continued development;
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 118
•the time and cost necessary to respond to technological and market developments, including other products that may compete with one or more of our product candidates;
•the impact of the macroeconomic environment; and
•the impact of the COVID-19 pandemic.
Our operating plans may change in the future, and we will need additional funds to meet operational needs and capital requirements associated with such operating plans.
If we fail to obtain additional capital when needed, our ability to operate as a going concern will be harmed, and we may be required to delay, scale back or eliminate some or all of our research and development programs and commercialization efforts and/or reduce our selling, general and administrative expenses, be unable to attract and retain highly-qualified personnel, be unable to obtain and maintain contracts necessary to continue our operations and at affordable rates with competitive terms, refrain from making our contractually required payments when due (including debt payments) and/or be forced to cease operations, liquidate our assets and possibly seek bankruptcy protection.
See the information under the heading “Risk Factors” included in Part I, Item 1A this Form 10-K for risks related to our financial condition.
Funding Requirements
Please see the risks associated with our substantial capital requirements explained more fully under the heading “Risk Factors — If we are unable to successfully complete a strategic acquisition, we may be forced to cease operations altogether or file for bankruptcy protection.” in Part I, Item 1A of this Form 10-K.
Contractual Obligations, Commitments, and Contingencies other than Debt Obligations
We are party to contractual obligations involving commitments to make payments to third parties in the future. Certain contractual obligations are reflected on our Consolidated Balance Sheet as of December 31, 2022, while others are considered future obligations. Our material cash requirements as of December 31, 2022, include the following contractual obligations and commitments arising in the normal course of business, including leases, purchases commitments, and purchase obligations described in more detail below.
Leases
We lease our headquarters in Boston, Massachusetts, under a non-cancelable operating lease with an expiration date of June 1, 2028. We also lease office space in San Francisco, California, under a non-cancelable operating lease that expires on July 31, 2025, and office space in Raleigh, North Carolina, under a non-cancelable operating lease that expires on May 31, 2026.
Effective January 1, 2023, the Company subleased 7,218 square feet of space in Boston, Massachusetts with a stated term of one year and renewal options for two additional years in one year increments. Effective January 6, 2023, the Company subleased the entirety of its 7,654 sq feet of rentable space in Raleigh, North Carolina with a stated term of one year and an option to renew for one twelve-month period, at which time a 3% rate increase will apply.
See Note 8, Leases, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Unconditional purchase commitments
We enter into agreements in the normal course of business with various vendors, which are generally cancellable upon notice. Payments due upon cancellation consist only of payments for services provided or expenses incurred, including non-cancellable obligations of service providers, up to the date of cancellation.
In addition, under various licensing agreements to which we are a party,Pear Therapeutics, Inc. | 2022 Form 10-K |Page 119
On June 17, 2021, and as amended on August 3, 2021, July 29, 2022 and December 12, 2022 we entered into a non-cancelable purchase obligation for a subscription to the Palantir Foundry cloud platform, including support services, updates, and related professional services with Palantir for $9.3 million payable over three years. As of December 31, 2022, we have paid $4.4 million and have amounts due under the terms of the agreement for the years ended December 31, 2023 and 2024 of $2.3 million and $2.5 million, respectively.
License Agreements
We have various licensing agreements to which we are a party and we are obligated to pay annual license maintenance fees andunder. In addition, we may be required to make milestone payments and to pay royalties and other amounts to third parties. TheSome of the payment obligations under these agreements are contingent upon future events, such as our achievement of specified milestones or generating product revenue, and the amount, timing and likelihood of such payments are not known. Amounts related to contingent milestone payments are not considered contractual obligations as they are contingent on the successful achievement of certain milestones. These contingent milestones may not be achieved. We cannot estimate or predict when, or if, these amounts will become due.
On June 17, 2021, and later amended on August 3, 2021, the Company entered into a non-cancelable purchase obligation for a subscription to the Palantir Foundry cloud platform, including support services, updates, and related professional services with Palantir for $9.3 million payable over three years, continuing through September 30, 2024.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 112
Other license agreements require us to pay minimum annual maintenance or royalty fees. See Note 8 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
9, Commitments and Contingencies, in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Off-Balance Sheet Arrangements
As of December 31, 2022 and December 31, 2021, in connection with our leased property in San Francisco, California we had $0.4 million in a letter of credit outstanding. On March 10, 2023, the issuer of the landlord’s letter of credit, Silicon Valley Bank, was placed into receivership with the FDIC. Since that date the obligations of Silicon Valley Bank have been assumed by First-Citizens Bank & Trust Company. The landlord has demanded a replacement letter of credit by no later than April 21, 2023.
Director and Officer Indemnification
We have entered into indemnification agreements with our directors and certain officers and employees that will require us, among other things, to indemnify them against certain liabilities that may arise by reason of their status or service as directors, officers, or employees. No demands have been made upon us to provide indemnification under such agreements and there are no claims that we are aware of that could have a material effect on our consolidated financial statements.
Cash Flows
The following table provides a summary of cash flow data for each applicable period:
Operating Activities
Net cash used in operating activities was $109.0 million for | | | | | | | | | | | | | | |
| | Year Ended December 31, |
NET CASH PROVIDED BY/(USED IN) (in thousands): | | 2022 | | 2021 |
| Operating activities | | $ | (113,895) | | | $ | (109,043) | |
| Investing activities | | (9,457) | | | 2,883 | |
| Financing activities | | 2,155 | | | 164,077 | |
| Net (decrease) increase in cash, cash equivalents, and restricted cash | | $ | (121,197) | | | $ | 57,917 | |
Cash, cash equivalents and restricted cash—beginning of period | | 169,978 | | | 112,061 | |
Cash, cash equivalents and restricted cash—end of period | | $ | 48,781 | | | $ | 169,978 | |
Operating Activities
Net cash used in operating activities was $113.9 million for the year ended December 31, 2022. Net cash used in operating activities consists of a net loss of $75.5 million, adjusted for non-cash items and the effect of changes in working capital. Non-cash adjustments primarily include the change in fair value of earn-out liabilities of $45.3 million, the change in fair value of warrant liabilities of $6.4 million, and net increases in operating assets and
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 120
liabilities (working capital) of $9.8 million, partially offset by stock-based compensation of $13.5 million, depreciation of $3.3 million and an impairment of intangible assets of $2.9 million.
Our outstanding debt as of December 31, 2022, was $30.0 million. Cash interest paid for the years ended December 31, 2022, and 2021 were $3,560 and $3,709, respectively. In accordance with the lenders payment terms the December 2022 interest payment was paid in January 2023 resulting in only eleven monthly cash payments being made during the year ended December 31, 2022 as compared to twelve cash payments being made during the year ended December 31, 2021. As of December 31, 2022, we had $0.4 million of accrued interest included in accrued expenses and other current liabilities on the consolidated balance sheet related to the outstanding debt.
Net cash used in operating activities was $109.0 million for the year ended December 31, 2021. Net cash used in operating activities consists of a net loss of $65.1 million, adjusted for non-cash items and the effect of changes in working capital. Non-cash adjustments primarily include the change in fair value of earn-out liabilities of $47.0 million, million, depreciation of $1.7 million, and net changes in operating assets and liabilities (working capital) of $5.9 million, partially offset by stock-based compensation of $3.8 million, loss on issuance of convertible preferred stock of Legacy Pear of $2.1 million, and depreciation of $1.7 million and net changes in operating assets and liabilities (working capital) of $5.9 million.2.1 million.
Investing Activities
Net cash used in operatinginvesting activities was $679.95 million for 2020.the Net cash used in operating activities consists of a net lossyear ended December 31, 2022, and related primarily to the purchase of investments of $9766.0 million, adjusted for non-cash items and the effectpurchases of changes in working capital. Non-cash adjustments primarily include loss on the issuance of convertible preferred stock of Legacy Pear of $16property and equipment of $3.85 million, stock-based compensation expense of $9 offset by proceeds from the maturity and sale of investments of $60.0 million, depreciation expense of $0.4 million, loss on the extinguishment of debt of $1.0 million, and the change in fair value of Legacy Pear warrants of $0.8 million (increase in ending warrant liability).
Investing Activities.
Net cash provided by investing activities was $2.9 million for the year ended December 31, 2021, related primarily to the maturities of investments offset by the purchase of investments, $3.3 million investment in property and equipment which is primarily internal-use software, and $1.4 million in intangible assets, and a $1.0 million regulatory milestone payment.
Net cash provided by investingFinancing Activities
Net cash provided by financing activities was $582.92 million for 2020the year ended December 31, 2022, and related primarily to proceeds from the maturitiesexercise of investments offset by a regulatory milestone payment and investment in internal-use software.
Financing Activities
Through December 31, 2021, Pear has financed its operations primarily through the Business Combination with THMA (discussed above), the sale of Legacy Pear convertible preferred stock, payments received in connection with collaboration agreements, and borrowings under various credit facilitiesstock options.
Net cash provided by financing activities was $164.1 million for 2021 and related to gross proceeds from the Business Combination of $144.3 175.0 million offset by associated transaction costs of $30.7 million, and proceeds of $19.9 million from the issuance of Legacy Pear preferred convertible stock, offset by transaction costs associated with the Business Combination of $32.8 million.
Net cash provided by financing activities was $91.7 million for 2020 and primarily related to net proceeds from the issuance of Legacy Pear Series D convertible preferred stock of $111.1 million, proceeds from the drawdown on the Perceptive Credit Facility of $30.0 million, $1.0 million of borrowings under the former SVB Term Loan, and proceeds of $0.4 million from the exercise of stock options offset by the repurchase of Legacy Pear common and convertible preferred stock of $32.0 million and the payment to extinguish the former SVB Term Loan in the amount of $17.3 million.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 113
million from the issuance of Legacy Pear convertible preferred stock. Related Party Transactions
During the year ended December 31, 2020, in connection withEffective March 15, 2022, we entered into a development agreement with SoftBank Corp., an entity under common control with SVF II Cobbler (DE) LLC (a greater than 5% shareholder of Pear), to develop a Japanese-language digital therapeutic for the treatment of sleep/wake disorders for the Japanese market. See Note 10, Revenue and Contract Balances, in the accompanying notes to the Legacy Pear Series D-2 preferred stock offering, the Company completed a tender offer whereby the Company purchased 2,788,732 shares of Legacy Pear common stock from its President & Chief Executive Officer, and 41,100 Legacy Pear common shares from certain eligible employees, at a purchase price of $3.9433 per share totaling $11.2 million and resulting in a stock-based compensation expense of $7.3 million, representing the difference between the purchase price and the fair value of the Legacy Pear common stock on the date of the sale.
Certain holders of Legacy Pear Series A preferred stock had representation on the Company’s then board of directors and purchased shares of Legacy Pear Series B preferred stock. Certain holders of Legacy Pear Series A and B preferred stock had representation on the Company’s then board of directors and purchased shares of Legacy Pear Series C preferred stock. Certain holders of Legacy Pear Series A, B, and C preferred stock had representation on the Company’s then board of directors and purchased shares of Legacy Pear Series D preferred stock. Certain holders of Legacy Pear Series A, B, C, and D preferred stock had representation on the Company’s then board of directors and purchased PIPE Shares.
Emerging Growth Company Status (JOBS Act)
Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can choose not to take advantage of the extended transition period and comply with the requirements that apply to non-emerging growth companies, and any such election to not take advantage of the extended transition period is irrevocable. We are an “emerging growth company” as defined in Section 2(A) of the Securities Act of 1933, as amended, and have elected to take advantage of the benefits of this extended transition period.
We expect to use this extended transition period for complying with new or revised accounting standards that have different effective dates for public business entities and nonpublic business entities until the earlier of the date we (a) are no longer an emerging growth company or (b) affirmatively and irrevocably opts out of the extended transition period provided in the JOBS Act. This may make it difficult or impossible to compare our financial results with the financial results of another public company that is either not an emerging growth company or is an emerging growth company that has chosen not to take advantage of the extended transition period exemptions because of the potential differences in accounting standards used.
In addition, we have chosen to take other exemptions and reduced reporting requirements provided by the JOBS Act and are not required to, among other things:
(a) provide an auditor’s attestation report on Pear’s system of internal control over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act;
(b) provide all of the compensation disclosure that may be required of non-emerging growth public companies under the Dodd-Frank Wall Street Reform and Consumer Protection Act; and
(c) disclose certain executive compensation-related items such as the correlation between executive compensation and performance and comparisons of the Chief Executive Officer’s compensation to median employee compensationconsolidated financial statements included in Part II, Item 8 of this Form 10-K for further information.
Recent Accounting Pronouncements
Refer to the accompanying notes to consolidated financial statements as of and for the years ended December 31, 20212022 and 20202021, included in Part III, Item 8 of this Form 10-K for more information regarding recently issued accounting pronouncements, the timing of their adoption, and its assessment, to the extent it has made one, of their potential impact on its financial condition and results of operations.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 114
Critical Accounting Policies and Estimates
The preparation of our consolidated financial statements and related disclosures in conformity with US generally accepted accounting principles, or US GAAP, and the Company’s discussion and analysis of its financial condition and operating results require the Company’s management to make judgments, assumptions and estimates that affect the amounts reported. See Note 2 in (‘”US GAAP”), and the Company’s discussion and analysis of its financial condition
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 121
and operating results require the Company’s management to make judgments, assumptions and estimates that affect the amounts reported. These estimates and judgments affect the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for further informationreported amounts of assets, liabilities, revenues and expenses, and the related disclosure of contingent assets and liabilities. Management bases these estimates on historical experience, known trends and onevents and various other assumptions that it believeswe believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying amounts of assets and liabilities. Actual results may differ materially from these estimates if past experience or other assumptions do not turn out to be substantially accurate.
The preparation of these consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and the related disclosure of contingent assets and liabilities. We monitor our estimates on an ongoing basis for changes in facts and circumstances, and material changes in these estimates could occur in the future. Actual results may differ materially from these estimates under different assumptions or conditions, or if past experience or other assumptions do not turn out to be substantially accurate. Changes in estimates are recorded in the period in which they become known.
Legacy Pear Preferred and Common Stock Valuations
While our significant accounting policies are described in more detail in Note 2, The fair valueSummary of Legacy Pear’s preferred and Legacy Pear common stock underlying stock awards was determined by the then board of directors. Given the absence of a public trading market, the then board of directors considered numerous objective and subjective factors to determine the fair value of Legacy Pear’s common stock at each board of directors meeting in which stock awards were approved. These factors included, but were not limited to:
•the prices at which we sold our preferred stock to outside investors in arms-length transactions;
•our results of operations, financial position, and capital resources;
•contemporaneous third-party valuations common stock;
•the rights, preferences, and privileges of convertible preferred stock relative to common stock;
•the lack of marketability of common stock;
•stage and development of Pear’s business;
•the history and nature of our business, industry trends and competitive environment,
•general economic conditions; and
•the likelihood of achieving a liquidity event, such as an initial public offering or sale of Pear, given prevailing market conditions.
We determined the fair value per share of the underlying Legacy Pear preferred and Legacy Pear common stock by taking into consideration results obtained from third-party valuations and additional factors that were deemed relevant. The assumptions underlying these valuations represented management’s best estimates, which involved inherent uncertainties and the application of management’s judgment. As a result, if we had used significantly different assumptions or estimates, the fair value of our Legacy Pear preferred and Legacy Pear common stock and our stock-based compensation expense could be materially different. The fair value of the underlying Legacy Pear common stock was determined by the then board of directors until we were listed on Nasdaq on December 3, 2021.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 115
Table of ContentsSignificant Accounting Policies, to our consolidated financial statements in this Annual Report on Form 10-K, we believe that the following accounting policies are those most critical to the judgments and estimates used in the preparation of our financial statements. Revenue Recognition
We determine both product; and collaboration and license; and subscription, support, and professional services revenue recognition through the following five-step framework in accordance with ASU 2014-09, Revenue from Contracts with Customers (Topic 606) and its related amendments, or, collectively, ASC 606:
•identification of the contract, or contracts, with a customer;
•identification of the performance obligations in the contract;
•determination of the transaction price;
•allocation of the transaction price to the performance obligations in the contract; and
•recognition of revenue when, or as, Pear satisfies a performance obligation.
Performance obligations promised in a contract are identified based on the goods and services that will be transferred to the customer that are both capable of being distinct and are distinct in the context of the contract. If the contract contains a single performance obligation, the entire transaction price is allocated to the single performance obligation. To the extent a contract includes multiple promised goods and services, the Company applies judgment to determine whether promised goods and services are both capable of being distinct and distinct in the context of the contract. To the extent the goods and services are distinct, it requires an allocation of the transaction price to each performance obligation on a relative standalone selling price basis unless the transaction price is variable and meets the criteria to be allocated entirely to a performance obligation or to a distinct service that forms part of a single performance obligation. The consideration to be received is allocated among the separate performance obligations based on relative standalone selling prices. The Company typically determines standalone selling prices using an adjusted market assessment approach model, or based upon observable inputs.
The Company satisfies performance obligations either over time or at a point in time. Revenue is recognized over time if either (i) the customer simultaneously receives and consumes the benefits provided by the entity’s performance, (ii) the entity’s performance creates or enhances an asset that the customer controls as the asset is created or enhanced, or (iii) the entity’s performance does not create an asset with an alternative use to the entity and the entity has an enforceable right to payment for performance completed to date. If the entity does not satisfy a performance obligation over time, the related performance obligation is satisfied at a point in time by transferring the control of a promised good or service to a customer.
Our collaboration and license revenue to date has primarily been generated through two collaboration and license agreements: the Sandoz Agreement and the NIBR Agreement.
In October 2019, Sandoz terminated the Sandoz Agreement, and thereafter Pear assumed the responsibility to commercialize its two lead products, reSET and reSET-O. In October 2019, we began our efforts to self-commercialize reSET and reSET-O. In addition, in June 2020, NIBR terminated their collaboration agreement with the Company related to our schizophrenia and multiple sclerosis product candidate development programs. We will not recognize any revenue under these agreements in the future.
To date, while we have recognized product revenue from the sale of our PDTs, reSET, reSET-O, and Somryst, and although we expect to continue to recognize revenue from the sale of these PDTs, we do not expect to generate significant revenue until 2023. in the foreseeable future.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 122
Valuation of Earn-Out Liabilities
As discussed in Notes 1, 3, and 4 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K, on December 3, 2021, we completed a Business Combination. In connection with the Business Combination and pursuant to the Business Combination Agreement, eligible Legacy Pear Shareholders are entitled to receive an aggregate of 12,395,62512,395,625 shares of the Company’s Class A common stock (“Earn-Out SharesEarn-Out Shares”) upon the Company achieving certain earn-out triggering events. At the time of closing, the Company recorded the earn-out liabilities upon issuance at its then estimated fair value of the Earn-Out Shares. The fair
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 116
value of the Earn-Out Shares was derived utilizing a Monte Carlo Simulation Method (“MCSMvalue of the Earn-Out Shares was derived utilizing a Monte Carlo Simulation Method (“MCSM”) to assess the likelihood of a triggering event occurring in the specified time frame. Under each scenario, the occurrence of a triggering event is tracked. If one of the thresholds is reached, then the underlying fair value of the common stock at the time of the trigger event is utilized to determine the fair value of the Earn-Out Shares that would be issued. The mean value of the shares estimated over the course of numerous iterations are calculated to arrive at the fair value of the Earn-Out shares. The MCSM utilizes a number of inputs which include the trading price and volatility of the underlying common stock, expected term, risk-free interest rates, expected date of a qualifying event, and qualifying traded share price within the specified time frame. The determination of the fair value of these financial instruments is complex and highly judgmental due to the significant estimation required. In particular, the fair value estimate was sensitive to certain assumptions, such as the volatility of underlying shares. The earn-out liabilities to the Legacy Pear shareholders are measured at the end of each reporting period at fair value with changes in fair value reported in our statement of operations. As of the Closing Date, a liability for fair value of the Earn-Out Shares was estimated and recorded at $95.4 million. As of December 31, 2022, the estimated fair value decreased to $3.0 million, mainly due to changes in the underlying value of the shares of Pear common stock, which was $1.18 on December 31, 2022.
If the applicable triggering event is achieved for a tranche, the Company will account for the Earn-Out Shares for such tranche as issued and outstanding common stock.
Stock-Based Compensation
Accounting for stock-based compensation is a critical accounting policy due to the broad-based equity awards provided to employees at all levels and the use of equity awards as part of the strategy to retain employees as a result of a change of control events. Pear issues stock-based compensation to employees and non-employees, generally in the form of stock options and starting in 2022, in restricted stock units, or RSUs. Compensation expenses for stock options are recognized on a straight-line basis over the requisite service period, which is generally the vesting period. Forfeitures are accounted for when they occur. The fair value of the stock options is measured on the grant date using the Black-Scholes model, which utilizes the following estimates as inputs:
•Expected volatility: determined based on an average of the historical volatility of a peer group of similar public companies;
•Expected term: as our employee stock options have “plain vanilla” characteristics, the expected term is determined using the simplified method, which represents the average of the contractual term of the option and the weighted average vesting period of the option; the contractual life of the option is used for the expected life of non-employee stock options;
•Risk-free interest rate: based upon the US Treasury yield curve in effect at the time of grant; and
•Expected dividend yield: based on our history and current expectation of not paying dividends in the foreseeable future.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
As a “smaller reporting company,” as defined by Rule 12b‑2 of the Exchange Act we are not required to provide information under this informationitem.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 117123
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
| | | | | | | | |
Index to Consolidated Financial Statements | | Page |
| | |
| | |
| | |
| | |
| | |
| | |
All financial statement schedules have been omitted, since the required information is not applicable or is not present in amounts sufficient to require submission of the schedule, or because the information required is included in the consolidated financial statements and accompanying notes.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 118124
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Stockholders and the Board of Directors of Pear Therapeutics, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Pear Therapeutics, Inc. and its subsidiaries (the "Company") as of December 31, 20212022 and 20202021, the related consolidated statements of operations and comprehensive loss, stockholders' equity, and cash flows, for each of the two years in the period ended December 31, 20212022 and the related notes (collectively referred to as the "financial statements"). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 20212022 and 20202021, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 20212022, in conformity with accounting principles generally accepted in the United States of America.
Going Concern
Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has experienced recurring losses from operations and recurring negative operating cash flows and may be unable to remain in compliance with certain financial covenants required under its credit facility which raiseraises substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters are described in Note 1. Management’s plans include the potential sale of the Company, or all or a portion of the Company’s assets; if management is unable to effect these plans, it may seek protection under bankruptcy or liquidation. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
| | |
/s/ Deloitte & Touche LLP |
|
Boston, Massachusetts |
March 2831, 2022 |
|
| 2023
We have served as the Company’s auditor since 2017. |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 119125
Pear Therapeutics, Inc.
CONSOLIDATED BALANCE SHEETS
(dollars in thousands, except per share amounts)
| | | | | | | | | | | |
| December 31, |
| (dollars in thousands, except per share amounts) | 2022 | | 2021 |
| Assets | | | |
Current assets: | | | |
Cash and cash equivalents | $ | 48,301 | | | $ | 169,567 | |
Short-term investments | 10,971 | | | 5,004 | |
| Restricted cash - short-term | 69 | | | — | |
Accounts receivable | 6,943 | | | 1,794 | |
Prepaid expenses and other current assets | 6,675 | | | 8,876 | |
Total current assets | 72,959 | | | 185,241 | |
Property and equipment, net | 6,076 | | | 6,255 | |
| Lease right-of-use assets | 8,879 | | | — | |
Restricted cash - long-term | 411 | | | 411 | |
| | | |
Other long-term assets | 6,804 | | | 5,253 | |
Total assets | $ | 95,129 | | | $ | 197,160 | |
Liabilities and stockholders’ equity | | | |
Current liabilities: | | | |
Accounts payable | $ | 3,263 | | | $ | 1,806 | |
Accrued expenses and other current liabilities | 16,464 | | | 17,946 | |
Operating lease liabilities - current | 1,984 | | | — | |
Deferred revenues | 284 | | | 421 | |
Debt | 27,447 | | | 26,993 | |
Total current liabilities | 49,442 | | | 47,166 | |
| Operating lease liabilities - non-current | 8,176 | | | — | |
Embedded debt derivative | 1,245 | | | 675 | |
Warrant liabilities | 2,116 | | | 8,528 | |
Earn-out liabilities | 3,024 | | | 48,363 | |
Other long-term liabilities | 541 | | | 1,994 | |
Total liabilities | 64,544 | | | 106,726 | |
Commitments and contingencies (Note 9) | | | |
| | | |
Stockholders’ equity: | | | |
Preferred stock, $0.0001 par value; 10,000,000 shares authorized as of December 31, 2022; and no shares issued and outstanding as of December 31, 2022 and December 31, 2021 | — | | | — | |
Common stock, $0.0001 par value; 690,000,000 shares authorized as of December 31, 2022; and 140,454,086 and 137,836,028 shares issued and outstanding as of December 31, 2022 and December 31, 2021, respectively | 14 | | | 14 | |
Additional paid-in capital | 354,092 | | | 338,404 | |
Accumulated deficit | (323,474) | | | (247,983) | |
Accumulated other comprehensive loss | (47) | | | (1) | |
Total stockholders’ equity | 30,585 | | | 90,434 | |
Total liabilities and stockholders’ equity | $ | 95,129 | | | $ | 197,160 | |
The accompanying notes are an integral part of these consolidated financial statements.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 120126
Pear Therapeutics, Inc.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(dollars in thousands, except per share amounts)
| | | | | | | | | | | | | | | |
| | | Year Ended December 31, |
| (dollars in thousands, except per share amounts) | | | | | 2022 | | 2021 |
Revenue | | | | | | | |
Product revenue | | | | | $ | 10,417 | | | $ | 3,748 | |
Collaboration and license revenue | | | | | 872 | | | 460 | |
| Subscription, support, and professional services revenue | | | | | 1,405 | | | — | |
Total revenue | | | | | 12,694 | | | 4,208 | |
Cost and operating expenses | | | | | | | |
Cost of revenue | | | | | 8,182 | | | 5,233 | |
Research and development | | | | | 48,311 | | | 37,041 | |
Selling, general, and administrative | | | | | 79,551 | | | 67,619 | |
Total cost and operating expenses | | | | | 136,044 | | | 109,893 | |
Loss from operations | | | | | (123,350) | | | (105,685) | |
Other income (expense): | | | | | | | |
Interest and other (expense) income, net | | | | | (3,892) | | | (4,144) | |
| Change in estimated fair value of earn-out liabilities | | | | | 45,339 | | | 47,038 | |
Change in estimated fair value of warrant liabilities | | | | | 6,412 | | | (298) | |
| | | | | | | |
Loss on issuance of Legacy Pear convertible preferred stock | | | | | — | | | (2,053) | |
Total other income | | | | | 47,859 | | | 40,543 | |
Net loss | | | | | $ | (75,491) | | | $ | (65,142) | |
Unrealized loss on short-term investments | | | | | $ | (46) | | | $ | (1) | |
Comprehensive loss | | | | | $ | (75,537) | | | $ | (65,143) | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
Net loss per share: | | | | | | | |
Basic and diluted | | | | | $ | (0.54) | | | $ | (0.57) | |
| | | | | | | |
Weighted average common shares outstanding: | | | | | | | |
Basic and diluted | | | | | 138,707,278 | | | 113,328,450 | |
| | | | | | | |
The accompanying notes are an integral part of these consolidated financial statements.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 121127
Pear Therapeutics, Inc.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(dollars in thousands)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | |
| | | | Common Stock | | Additional Paid-In Capital | | Accumulated Deficit | | Accumulated Other Comprehensive Loss | | Total Stockholders’ Equity |
| (dollars in thousands) | | | | | | Shares | | Amount | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
Balance at January 1, 2021 | | | | | | 106,721,878 | | | $ | 11 | | | $ | 269,946 | | | $ | (182,841) | | | $ | — | | | $ | 87,116 | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
Issuance of Legacy Pear Series D convertible preferred stock, net of issuance costs of $83 | | | | | | 4,503,618 | | | — | | | 21,970 | | | — | | | — | | | 21,970 | |
| Exercise of common stock options | | | | | | 1,130,921 | | | — | | | 859 | | | — | | | — | | | 859 | |
| Stock-based compensation expense | | | | | | — | | | — | | | 3,810 | | | — | | | — | | | 3,810 | |
| Exercise of Legacy Pear warrants | | | | | | 1,079,686 | | | — | | | 10,907 | | | — | | | — | | | 10,907 | |
| Reverse recapitalization, net of transaction costs (Note 3) | | | | | | 24,399,925 | | | 3 | | | 142,800 | | | — | | | — | | | 142,803 | |
| Recognition of Warrant liabilities assumed in the Merger (Note 3) | | | | | | — | | | — | | | (16,487) | | | — | | | — | | | (16,487) | |
| Recognition of Earn-out liability (Note 3) | | | | | | — | | | — | | | (95,401) | | | | | | | (95,401) | |
| Other comprehensive loss | | | | | | — | | | — | | | — | | | — | | | (1) | | | (1) | |
| Net loss | | | | | | | | — | | | — | | | (65,142) | | | — | | | (65,142) | |
| Balance at December 31, 2021 | | | | | | 137,836,028 | | | $ | 14 | | | $ | 338,404 | | | $ | (247,983) | | | $ | (1) | | | $ | 90,434 | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | |
| | | | Common Stock | | Additional Paid-In Capital | | Accumulated Deficit | | Accumulated Other Comprehensive Loss | | Total Stockholders’ Equity |
| (dollars in thousands) | | | | | | Shares | | Amount | | | | |
Balance at January 1, 2022 | | | | | | 137,836,028 | | | $ | 14 | | | $ | 338,404 | | | $ | (247,983) | | | $ | (1) | | | $ | 90,434 | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | |
| Exercise of common stock options | | | | | | 2,258,138 | | | — | | | 1,795 | | | — | | | — | | | 1,795 | |
| Exercise of common stock warrants | | | | | | 10 | | | — | | | — | | | — | | | — | | | — | |
| Issuance of common stock under employee stock purchase plan | | | | | | 359,910 | | | — | | | 360 | | | — | | | — | | | 360 | |
| Stock-based compensation expense | | | | | | — | | | — | | | 13,533 | | | — | | | — | | | 13,533 | |
| Other comprehensive loss | | | | | | — | | | — | | | — | | | — | | | (46) | | | (46) | |
| Net loss | | | | | | — | | | — | | | — | | | (75,491) | | | — | | | (75,491) | |
| Balance at December 31, 2022 | | | | | | 140,454,086 | | | $ | 14 | | | $ | 354,092 | | | $ | (323,474) | | | $ | (47) | | | $ | 30,585 | |
The accompanying notes are an integral part of these consolidated financial statements.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 122128
Pear Therapeutics, Inc.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(dollars in thousands)
| | | | | | | | | | | |
| Year Ended December 31, |
| (dollars in thousands) | 2022 | | 2021 |
Operating Activities: | | | |
Net loss | $ | (75,491) | | | $ | (65,142) | |
Adjustments to reconcile net loss to net cash used in operating activities: | | | |
Depreciation expense | 3,347 | | | 1,689 | |
Impairment of intangible asset | 2,879 | | | — | |
Amortization of intangible asset | 620 | | | 563 | |
Amortization of debt discount | 453 | | | 648 | |
| Amortization of right-of-use asset | 1,735 | | | — | |
| | | |
Accretion and amortization of interest income | (26) | | | 22 | |
Stock-based compensation expense | 13,533 | | | 3,810 | |
Loss on issuance of Legacy Pear convertible preferred stock | — | | | 2,053 | |
Change in fair value of warrants | (6,412) | | | 298 | |
| Change in fair value of earn-out liabilities | (45,339) | | | (47,038) | |
| Change in fair value of embedded derivative | 569 | | | — | |
Changes in operating assets and liabilities: | | | |
Accounts receivable | (5,150) | | | (1,192) | |
Prepaid expenses and other assets | (930) | | | (8,979) | |
Lease liabilities | (1,700) | | | — | |
Accounts payable | 4 | | | (3,094) | |
Accrued expenses, other liabilities and non-current liabilities | (1,841) | | | 7,144 | |
Deferred revenues | (146) | | | 175 | |
Net cash used in operating activities | (113,895) | | | (109,043) | |
Investing Activities: | | | |
Proceeds from maturities of short-term investments | 60,048 | | | 16,525 | |
Purchases of short-term investments | (66,035) | | | (8,014) | |
Purchases of property and equipment | (3,470) | | | (3,278) | |
Purchases of intangible assets | — | | | (1,350) | |
Milestone based license fee payment | — | | | (1,000) | |
Net cash (used in) provided by investing activities | (9,457) | | | 2,883 | |
Financing Activities: | | | |
Proceeds from issuance of Legacy Pear convertible preferred stock, net | — | | | 19,917 | |
| Proceeds from Business Combination, net of transactions costs paid | — | | | 144,301 | |
| Repayment of note assumed in the Business Combination | — | | | (1,000) | |
| | | |
| | | |
| | | |
| | | |
Proceeds from exercise of stock options | 1,795 | | | 859 | |
Proceeds from employee stock purchase plan | 360 | | | — | |
Net cash provided by financing activities | 2,155 | | | 164,077 | |
Net (decrease) increase in cash, cash equivalents and restricted cash | (121,197) | | | 57,917 | |
Cash, cash equivalents and restricted cash—beginning of period | 169,978 | | | 112,061 | |
Cash, cash equivalents and restricted cash—end of period | $ | 48,781 | | | $ | 169,978 | |
Supplemental disclosure of cash flow information: | | | |
Cash paid for interest | $ | 3,560 | | | $ | 3,709 | |
Supplemental disclosure of non-cash investing and financing information: | | | |
| | | |
| Issuance of stock upon exercise of Legacy Pear warrants | $ | — | | | $ | 10,907 | |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 123129
Pear Therapeutics, Inc.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| | | | | | | | | | | |
| Recognition of warrant liability | $ | — | | | $ | 16,487 | |
| Recognition of earn-out liabilities | $ | — | | | $ | 95,401 | |
| Note payable and net liabilities assumed in the Business Combination (excluding warrant liabilities) | $ | — | | | $ | 1,147 | |
Deferred offering and equity issuance costs included in accounts payable and accrued expenses | $ | 163 | | | $ | 700 | |
| Purchase of intangible asset with seller financing | $ | — | | | $ | 1,011 | |
| | | |
Purchases of property and equipment in accounts payable and accrued expenses | $ | 86 | | | $ | 388 | |
Purchases of other assets in accounts payable and accrued expenses | $ | 1,755 | | | $ | — | |
| | | |
| | | |
(dollars in thousands)
The accompanying notes are an integral part of these consolidated financial statements.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 124130
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
1. NATURE OF THE BUSINESS
Organization
Pear Therapeutics, Inc. and subsidiaries (the "Company" or "Pear"), is a leader in prescription digital therapeutics, or PDTs. The Company’s PDTs treat diseases with clinically validated software. The Company obtained FDA marketing authorization for its three products, reSET® (2017), reSET-O® (2018), and Somryst® (2020), all of which are sold as an integrated product with access to our proprietary clinician facing dashboard, PearMD®.
On December 3, 2021, (the “Closing Date”), Thimble Point Acquisition Corp. (“THMA”)Closing Date”), we consummated the previously announced merger pursuant to the Business Combination Agreementa business combination, or the “Business Combination”, pursuant to the terms of the business combination agreement, or “Business Combination Agreement”, dated June 21, 2021, by and among THMA, Pear Therapeutics, Inc. (now Pear Therapeutics (US), Inc.) and Oz Merger Sub, Inc., pursuant to which Oz Merger Sub, Inc. merged with and into Pear Therapeutics (US), Inc.the Company (formerly known as Thimble Point Acquisition Corp., or “THMA”), Pear Therapeutics (US), Inc., a Delaware corporation incorporated on August 14, 2013 (“Pear US” or “Legacy Pear”) (formerly known as Pear Therapeutics, Inc.) and Oz Merger Sub, Inc., pursuant to which Oz Merger Sub, Inc. (a Delaware corporation and wholly-owned subsidiary of THMA, or “Merger Sub”) merged with and into Pear US, with Pear Therapeutics (US), Inc. becomingUS surviving as our wholly owned subsidiary (the “Business Combination”). Upon the closing of the Business Combination, THMA changed its name to Pear Therapeutics, Inc. and Pear Therapeutics, Inc. changed its name. Upon the closing of the Business Combination, THMA changed its name to Pear Therapeutics, Inc. (“Pear” or the “Company”). See Note 3, Business Combination, for further information.
References throughout this Form 10-K to “we,” “us,” the “Company,” “Pear” or “our company” are to Pear Therapeutics, Inc. (formerly known as Thimble Point Acquisition Corp.) and its subsidiaries, and “Legacy Pear” refers to Pear Therapeutics (US), Inc.
In connection with the Business Combination, THMA completed the sale and issuance of 10,280,000 shares of Class A common stock in a fully committed common stock private placement at a purchase price of $10.00 per share (“PIPE Shares”) for an aggregate purchase price of $102,800 (“PIPE Investment”), and a Forward Purchase Agreement Assignment, dated as of December 2, 2021, by and among THMA, the Anchor Investor, and a Pipe Investor (the “Forward Purchase Assignment”); which closed simultaneously with the consummation of the Business Combination. Upon the closing of the Business Combination, the PIPE Shares were automatically converted into shares of the Company's Class A common stock on a one-for-one basis. In addition, all of the remaining outstanding THMA Class A common shares were separated, pursuant to their terms, into one share of Class A common stock (which totaled 832,899 shares Class A common stock, “Public Shares”) and one-third (1/3) of one redeemable warrant (and THMA’s units ceased trading on the Nasdaq). Further, LP SPAC 1 LLC (the “Anchor Investor”) purchased 6,387,026 shares of Class A common stock at a purchase price of $10.00 per share in connection with the Forward Purchase Agreement, dated as of February 1, 2021, by and between THMA and the Anchor Investor (the “Forward Purchase Agreement”), as amended from time to time, including by the Amendment to Forward Purchase Agreement dated as of June 21, 2021 and the Second Amendment to Forward Purchase Agreement dated as of November 14, 2021 (the “Amended Forward Purchase Agreement”) entered into with THMA on February 1, 2021 (“THMA Sponsor Shares”). Gross proceeds from the Merger totaled approximately $175,001 which included funds held in THMA’s trust account (after giving effect to redemptions). Transaction costs totaled approximately $32,779.
Unless otherwise indicatedprior to the Business Combination, unless otherwise noted or the context otherwise requires, references in this Form 10-Kindicates. References to the “Company”, “our”, and “Pear” refer to the consolidated operations of Pear Therapeutics, Inc. and its subsidiaries. References to THMA refer to the company prior to the consummation of the Business Combination and references to “Legacy Pear” refer to Pear Therapeutics, Inc. (now Pear Therapeutics (US), Inc.) prior to the consummation of the Business Combination.
Legacy Pear was deemed the accounting acquirer in the Business Combination. This determination was primarily based on Legacy Pear’s stockholders prior to the Business Combination having a majority of the voting power in the combined company, Legacy Pear having the ability to appoint a majority of the Board of Directors of the combined company, Legacy Pear’s existing management comprising the senior management of the combined company, Legacy Pear comprising the ongoing operations of the combined company, Legacy Pear being the larger entity based on historical revenues and business operations, and the combined company assuming Legacy Pear’s name. Accordingly, for accounting purposes, the Business Combination was treated as the equivalent of Legacy Pear issuing stock for the net assets of THMA, accompanied by a recapitalization. Under this method of accounting, THMA who was the legal acquirer, is treated as the “acquired” company for financial reporting purposes. Accordingly, the Business Combination was treated as the equivalent of Pear issuing stock for the net assets of THMA, accompanied by a recapitalization. The net assets of THMA are stated at historical cost, with no goodwill or other intangible assets recorded. The equity structure has been restated in all comparative periods up to the Closing Date to reflect the number of shares of the Company’s Class A common stock, $0.0001 par value per share,
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 125
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
issued to Legacy Pear stockholders in connection with the Business Combination. As such, the shares and corresponding capital amounts and earnings per share related to Legacy Pear’s Convertible Preferred Stock (“Legacy Convertible Preferred Stock”) and Legacy Pear common stock prior to the Business Combination have been retroactively restated as shares reflecting the exchange ratio of approximately 1.47 established in the Business Combination. Legacy Pear’s convertible preferred stock previously classified as mezzanine was retroactively adjusted, converted into Class A common stock, and reclassified to permanent as a result of the reverse recapitalization. See Note 3 for more information.
THMA, now Pear Therapeutics, Inc., a Delaware corporationThimble Point Acquisition, Corp. (“THMA”) refer to the Company prior to the consummation of the Business Combination.
THMA, now Pear Therapeutics, Inc., a Delaware corporation, was incorporated on December 1, 2020. Pear Therapeutics (US), Inc., previously known as Pear Therapeutics, Inc. (“Legacy Pear”), is a Delaware corporation incorporated on August 14, 2013. The Company is headquartered in Boston, Massachusetts.
Going Concern
The Company is subject to a number of risks and uncertainties common to early-stage technology-based companies, including, but not limited to, rapid technological changes, protection of its proprietary technology and intellectual property, commercialization of existing and new products, development by competitors of competing products, dependence on key personnel, compliance with government regulations, including compliance with the US. Food and Drug Administration, or FDA (“FDA”), and the ability to secure additional capital to fund operations.
The Company obtained FDA marketing authorization for its three products, reSET® (2017), reSET-O® (2018), and Somryst® (2020). In October 2019, after terminating our agreement with Sandoz Inc., our then collaboration partner, we began the direct commercialization of reSET® and reSET-O®.
The Company has incurred recurring losses since inception and anticipates net losses and negative operating cash flows for the near future and may be unable to remain in compliance with certain financial covenants required under its credit facility. For the year ended December 31, 2021For the year ended December 31, 2022, the Company had a net loss of $6575,142491 and, as of December 31, 20212022, had an accumulated deficit of $247323,983474. As of December 31, 20212022, the Company had $17459,571272 of cash and cash equivalents and short-term investments. Management believes the Company has sufficient cash and cash equivalents to fund operating expenses and capital expenditures into the second quarter of 2023.
While the Company has recorded revenue, revenues have been insufficient to fund operations. Accordingly, the Company has funded its operationsWe require additional capital in order to pursue our strategic objectives. In February 2023, we initiated a process to explore a range of strategic alternatives to maximize shareholder value and engaged an investment bank. Potential strategic alternatives that may be evaluated include a sale or merger of the Company, the sale of all or a portion of the Company’s assets and/or intellectual property, or securing additional financing or partnerships that would enable further development of our programs. There is no set timetable for this process, and there can be no assurance that this strategic review process will result in our pursuing any transaction or that any transaction, if pursued, will be completed. Additionally, there can be no assurances that any particular course of action, business arrangement or transaction, or series of transactions, will be pursued, successfully consummated or lead to increased stockholder value. If the strategic process is unsuccessful, our Board may decide to pursue a liquidation or obtain
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 131
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
relief under the US Bankruptcy Code. The Company has hired a financial advisor, if needed, to date through a combination of proceeds raised from equity and debt issuances, including the Business Combination. The Company’s operating costs include the cost of developing and commercializing products as well as providing research services. As a consequenceassist with filing for bankruptcy protection.
As of the date of this filing, Perceptive Credit Holdings III, LP, as administrative agent and lender (“Perceptive”) has alleged that certain defaults or events of default have occurred and are continuing under the terms of the Amended and Restated Credit Agreement and Guaranty with Perceptive Credit Holdings III, LP, (“Perceptive Credit Facility”). Perceptive has not delivered any formal notice of Default or Event of Default. To the extent that any such allegations are valid, Perceptive would have certain rights and remedies under the Perceptive Credit Facility. The Company disputes the allegations and is in discussions with Perceptive to resolve this dispute and otherwise to address the Company’s obligations under the Perceptive Credit Facility. There can be no assurances that such discussions will result in any resolution, and any resolution, or the lack of any resolution, may result in Perceptive exercising remedies under the Perceptive Credit Facility. The lender has not invoked the material adverse change clause as of the date of issuance of these financial statements. The Company is subject to various covenants related to the Perceptive Credit Facility, as defined in Note 7, Indebtedness, entered into on June 30, 2020. A breach of any of these covenants, including a material adverse change in our business, operations, or condition (financial or otherwise), could result in an event of default under the Perceptive Credit Facility, which could cause all of the outstanding indebtedness under the facility to become immediately due and payable. If the acceleration of the debt were to occur, we could also be required to pay a prepayment penalty at the time of the acceleration. As of March 31, 2023, the Company will need to raise additional equity and debt financing thatprepayment penalty is approximately $3.6 million under the terms of the Perceptive Credit Facility.
Based on its recurring losses from operations incurred since inception, our expectation of continuing operating losses for the foreseeable future, negative operating cash flows for the foreseeable future, and the need to raise additional capital to finance its future operations, as well as the risk that the Company’s ability to comply with covenants under Perceptive Credit Facility may be affected by events beyond its control, and it may not be available, if at all, at terms acceptable to the Company to fund future operations. As a resultable to meet those covenants as well as the material adverse change clause in the Perceptive Credit Facility, the Company could be required to delay, scale back or abandon some or all of its development programs and other operations, which could materially harm the Company’s business, prospects, financial condition and operating results. Management believes these uncertainties raise substantial doubt abouthas concluded that there is substantial doubt regarding the Company’s ability to continue as a going concern within one year after the date that these consolidated financial statements are issued. Because of these uncertainties, the accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern, which contemplates the realization of assets and the settlement of liabilities and commitments in the normal course of business. Due to the substantial doubt about the Company’s ability to continue operating as a going concern for twelve months from the issuance date of these financial statements and the material adverse change clause in the loan agreement with its lender, the amounts dueAs such, the amounts due under the Perceptive Credit Facility as of December 31, 20212022, have been classified as current in the consolidated balance sheet. The lender has not invoked the material adverse change clause as of the date of issuance of these financial statements. The accompanying consolidated financial statements do not reflect any other adjustments relating to the recoverability and reclassification of assets and liabilities that might be necessary if the Company is unable to continue as a going concern. The Company is subject to various covenants related to the Perceptive Credit Facility entered into on June 30, 2020 (Note 7) and given the substantial doubt about the Company’s ability to continue as a going concern, there is a risk that it may not meet its covenants in the future.
COVID-19 Related Significant Risks and Uncertainties
There continues to be uncertainties regardingOn January 30, 2023, the Biden Administration announced it will end the public health emergency (and national emergency) declarations related to COVID-19 on May 11, 2023. While the COVID-19 pandemic has not materially adversely affected our financial results and business operations through December 31, 2022, COVID-19 continues to present risks to the Company, and we continue to closely monitor the impact of the pandemic of the novel coronavirus, oron all aspects of our business. We are unable to predict the impact that COVID-19. The pandemic has significantly impacted the economic conditions in the US, as federal, state and local governments react to the (including the emergence of new variants) will have on our financial position and operating results in the future.
In response to the COVID-19 pandemic, Congress passed the Coronavirus Aid, Relief and Economic Security Act of 2020, or the “CARES Act”, which was signed into law on March 27, 2020. Under the CARES Act the Company deferred payment of the employer portion of social security taxes through the end of 2020 in the amount of $854. The Company paid 50% or $427 during the year ended December 31, 2021, and included $427 in accrued expenses and other liabilities within the consolidated balance sheet as of December 31, 2021. The Company paid the remaining balance of $427 during the year ended December 31, 2022.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 126132
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
public health crisis, creating significant uncertainties in the US economy. The downstream impact of various lockdown orders and related economic pullback affect our business and our customers to varying degrees. the Company is closely monitoring the impact of COVID-19 on all aspects of its business, including how it will impact its customers, patients, employees, suppliers, vendors, and business partners. The Company is unable to predict the specific impact that COVID-19 may have on its business, financial position, and operations moving forward due to the numerous uncertainties. Any estimates made herein may change as new events occur and additional information is obtained, and actual results could differ materially from any estimates made herein under different assumptions or conditions. The Company will continue to assess the evolving impact of COVID-19.
In response to the COVID-19 pandemic, Congress passed the Coronavirus Aid, Relief and Economic Security Act of 2020, or the CARES Act, which was signed into law on March 27, 2020. The CARES Act provides for deferred payment of the employer portion of social security taxes through the end of 2020, with 50% of the deferred amount due by December 31, 2021, and the remaining 50% due by December 31, 2022. During the year ended December 31, 2020, the Company deferred payments of $854 of social security taxes, of which $427 is included in accrued expenses and other liabilities and the remaining $427 is included in other long-term liabilities. The company paid $427 during the year ended December 31, 2021, and had $427 included in accrued expenses and other liabilities within the consolidated balance sheet as of December 31, 2021.
2. 2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation and Principles of Consolidation
The accompanying consolidated financial statements have been prepared in accordance with US generally accepted accounting principles, or GAAP“GAAP”. Any reference in these notes to applicable guidance is meant to refer to the authoritative GAAP as found in the Accounting Standards Codification, or ASC“ASC”, and Accounting Standards Update, or ASU“ASU”, of the Financial Accounting Standards Board, or FASB“FASB”.
The consolidated financial statements as of December 31, 20212022 and 20202021, and for the years ended December 31, 20212022 and 20202021, include the accounts of Pear and its wholly owned subsidiaries. All significant intercompany accounts and transactions have been eliminated in the consolidated financial statements herein.
Certain monetary amounts, percentages, and other figures included elsewhere in these consolidated financial statements have been subject to rounding adjustments. Accordingly, figures shown as totals in certain tables may not be the arithmetic aggregation of the figures that precede them, and figures expressed as percentages in the text may not total 100% or, as applicable, when aggregated may not be the arithmetic aggregation of the percentages that precede them.
Reclassification of Prior Year Presentation
Certain prior year amounts have been reclassified for consistency with the current year presentation. These reclassifications had no effect on the reported results of operations. Adjustment has been made to the Consolidated Statement of Operations for the year ended December 31, 2020, to present the change in fair value of warrant liabilities as a separate line item; this amount was included in the Interest and other (expense) income, net line item in prior years.
Certain prior year amounts have been reclassified for consistency with the current year presentation. These reclassifications had no effect on the reported results of operations.Use of Estimates Use of Estimates
The preparation of the consolidated financial statements in conformity with GAAP, requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of expenses during the reporting periods. On an ongoing basis, the Company evaluates its estimates. The Company bases its estimates on historical experience and various other assumptions that the Company believes to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from those estimates and changes in estimates are reflected in reported results in the period in which they become known.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 127
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Cash, Cash Equivalents, and Restricted Cash
The Company considers only those highly liquid investments, readily convertible to cash, that mature within 90 days from the date of purchase to be cash equivalents. The Company’s cash equivalents include money market funds, commercial paper, and overnight deposits.
The following table reconciles cash, cash equivalents, and restricted cash reported within the Company’s consolidated balance sheets to the total amounts shown in the consolidated statements of cash flows:
| | | | | | | | | | | | | | |
| | December 31, |
| Reconciliation of cash, cash equivalents, and restricted cash: | | 2022 | | 2021 |
| | | | |
Cash and cash equivalents | | $ | 48,301 | | | $ | 169,567 | |
| Restricted cash - short-term | | 69 | | | — | |
Restricted cash - long-term | | 411 | | | 411 | |
Total cash, cash equivalents, and restricted cash | | $ | 48,781 | | | $ | 169,978 | |
Investments Investments
Investments include marketable securities with maturities of less than one year or where management’s intent is to use the investments to fund current operations or to make them available for current operations. All investments in marketable securities are classified as available for sale and are reported at fair value with unrealized gains and losses excluded from earnings and reported net of tax in accumulated other comprehensive income (loss), which is a component of stockholders’ equity. Unrealized losses that are determined to be other than , which is a component of stockholders’ equity. Unrealized losses that are determined to be other than
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 133
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
temporary, based on current and expected market conditions, are recognized in earnings. Declines in fair value determined to be credit-related are charged to earnings. The cost of marketable securities sold is determined by the specific identification method.
Concentration of Credit Risk
Financial instruments that are potentially subject to a significant concentration of credit risk consist primarily of cash, cash equivalents, investments, restricted cash and accounts receivable. The Company attempts to minimize the risk related to investments by working with highly rated financial institutions that invest in a broad and diverse range of financial instruments as defined by the Company. The Company has established guidelines relative to credit ratings and maturities intended to safeguard principal balances and maintain liquidity. The Company maintains its funds in accordance with its investment policy, which defines allowable investments, specifies credit quality standards and is designed to limit credit exposure to any single issuer. Through December 31, 20212022, and the date of this filing, the Company has not experienced any losses on such deposits. One collaboration partner represented 100% of the Company’s total deferred revenue as of December 31, 2020.
Fair Value Measurements
Fair value is defined as the price that would be received from selling an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. When determining the fair value measurements for assets and liabilities required to be recorded at fair value, the Company considers the principal or most advantageous market in which it would transact and considers assumptions that market participants would use when pricing the asset or liability, such as inherent risk, transfer restrictions and risk of non-performance. The accounting standard establishes a fair value hierarchy that requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. To the extent the valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair values requires more judgment. A financial instrument’s categorization within the fair value
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 128
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
hierarchy is based upon the lowest level of input that is significant to the fair value measurement. Three levels of inputs that may be used to measure fair value are:
•Level 1: Unadjusted quoted prices in active markets for identical assets or liabilities accessible to the reporting entity at the measurement date.
•Level 2: Other than quoted prices included in Level 1 inputs that are observable for the asset or liability, either directly or indirectly, for substantially the full term of the asset or liability.
•Level 3: Unobservable inputs for the asset or liability used to measure fair value to the extent that observable inputs are not available, thereby allowing for situations in which there is little, if any, market activity for the asset or liability at the measurement date.
Property and Equipment
Property and equipment are recorded at cost less accumulated depreciation. Expenditures for repairs and maintenance are expensed as incurred. Depreciation expense is recognized using the straight-line method over the estimated useful lives, which are typically:
| | | | | | | | |
| Asset Category | | Estimated Useful Life |
Equipment | | 3 years |
Internal-use software | | 3 - 5 years |
Furniture and fixtures | | 5 years |
Leasehold improvements | | Shorter of economic useful life or
the remaining lease term |
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 134
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
The Company capitalizes costs incurred to develop internal-use software during the application development stage. Costs related to preliminary project activities and post-implementation activities are expensed as incurred. Internal and external costs incurred in connection with the development of upgrades or enhancements that result in additional functionality are also capitalized. Amortization commences when the software is available for its intended use and is amortized on a straight-line basis over the software’s estimated useful life, calculated using a mid-quarter convention.
Upon retirement or sale, the cost of the assets disposed of and the related accumulated depreciation and amortization are removed from the accounts and any resulting gain or loss is included in other income (expense) in the consolidated statements of operations and other comprehensive loss. Major replacements and improvements are capitalized. Expenditures for repairs and maintenance are expensed as incurred.
Intangible Assets
Intangible assets consist of identifiable intangible assets, including developed technology, resulting from the Company’s acquisitions. Acquired identifiable finite-lived intangible assets are amortized on a straight-line basis or accelerated method over the estimated useful lives of the assets. The basis of amortization approximates the pattern in which the assets are utilized, over their estimated useful lives.
Impairment of Long-Lived Assets and Intangible Assets Subject to Amortization
Long-livelived assets primarily include property and equipment and intangible assets, which are included in other long-term assets on the consolidated balance sheets. The Company evaluates its long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable. The recoverability of assets to be held and used is measured by a comparison of the carrying amount of an asset to the future undiscounted net cash flows expected to be generated by the asset. If such assets are considered to be impaired, the impairment to be recognized is measured by the amount by which the carrying
Pear Therapeutics, Inc.amount of the asset exceeds the fair value of the asset. Based upon this analysis, due to events occurring in the fourth quarter of 2022, the Company determined that the carrying value of certain intangible assets was not recoverable and recorded a loss on impairment of $2,879, which included $788 in cost of revenue and $2,091 in research and development in the Company's consolidated statement of operations for the year ended December 31, 2022. See Note 9, Commitments and Contingencies, for more information. The Company did not recognize any impairment losses on long-lived assets for the year ended December 31, 2021.
Leases
The Company adopted the leasing standard effective January 1, 2022, using the revised modified retrospective transition method, with comparative periods continuing to be reported under ASC 840 as it was the accounting standard in effect for such period. In the adoption of ASU 2016-02, the Company carried forward the assessment from ASC 840 of whether its contracts contain or are leases, the classification of its leases, and remaining lease terms. The Company did not elect the hindsight practical expedient upon adoption of the new standard.
The most significant impact resulting from the adoption of this new standard was the recognition of $10,614 of an operating right-of-use (“ROU”) assets and $11,860 of operating lease liabilities on the adoption date, January 1, 2022. The difference between the ROU assets and lease liabilities on the accompanying consolidated balance sheet is primarily due to the accrual for lease payments as a result of straight-line lease expense and unamortized tenant incentive liability balances. Existing deferred rent and prepaid rent amounts were removed from the consolidated balance sheets at the date of adoption. The adoption did not have a material impact to the Company's consolidated statements of operations or statement of cash flows.
The Company has made an accounting policy election to keep leases with an initial term of 12 months or less off of the balance sheet and recognize those lease payments in the consolidated statements of income on a straight-line basis over the lease term. The Company has also elected the practical expedient to not separate lease and non-lease components for all of its leases as the non-lease components are not significant to the overall lease costs. If
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 129135
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
amount of the asset exceedsan interest rate is not implicit in a lease, the Company utilizes its incremental borrowing rate for a period closely matching the lease term. Prior to the adoption of ASC 842, the fair value of the asset. The Company did not recognize any impairment losses on long-lived assets for the years ended December 31, 2021 and 2020.
Leases
Rent expense for non-cancelable operating leases, including rent escalation clauses, tenant improvement allowances, and rent-free periods when applicable, is recognizedCompany recorded rent expense on a straight-line basis over the term of the lease with the difference between required lease payments and rent expense recorded as deferred rent. The lease term begins on the commencement date as defined in the lease agreement or when the Company takes possession of or begins to control the physical use of the property, whichever is earlier. Refer to Note 8related lease.
Refer to Note 8, Leases, for further information.
Derivative Liabilities
The Company accounts for derivative financial instruments as either equity or liabilities in accordance with ASC Topic 815, Derivatives and Hedging, or ASC 815, based on the characteristics and provisions of each instrument. Embedded derivatives are required to be bifurcated from the host instruments and recorded at fair value if the derivatives are not clearly and closely related to the host instruments on the date of issuance. Derivative instrument liabilities are classified in the consolidated balance sheets as current or non-current based on whether or not net-cash settlement of the derivative instrument could be required within 12 months of the balance sheet date.
The Company’s Perceptive Credit Facility (see Note 7 for more information) contains certain features that, in accordance with ASC 815, are not clearly and closely related to the host instrument and represent derivatives liabilities that are required to be re-measured at fair value each reporting period. Additionally, Legacy Pear issued Series A and Series D preferred stock warrants, which were classified as derivative liabilities recorded at fair value. See Note 4 and Note 7 for more information. The changes in the fair value of the warrants are recorded as other income (expense) in the Company’s consolidated statements of operations and comprehensive loss.
Legacy Pear Convertible Preferred Stock
In connection with the Business Combination, all Legacy Preferred Shares were converted to Class A common stock. See Notes 1, 3, and 10 for further information.
The Company recorded shares of Legacy Pear convertible preferred stock at their respective estimated fair values on the dates of issuance, net of issuance costs. Legacy Pear’s convertible preferred stock was classified outside of stockholders’ deficit because the holders of such shares had liquidation and redemption rights in the event of a deemed liquidation event that, in certain situations, are not solely within the control of the Company, such as a merger, acquisition, and sale of all or substantially all of the Company’s assets.
See Note 4, Fair Value Measurements, and Note 7, Indebtedness, for more information.Warrant Liabilities
Management evaluates all of the Company’s financial instruments, including issued Warrants to purchase its Class A common stock, to determine if such instruments are derivatives or contain features that qualify as embedded derivatives, pursuant to ASC 480 and ASC 815-15. The classification of derivative instruments, including whether such instruments should be recorded as liabilities or as equity, is re-assessed at the end of each reporting period.
As of December 31, 20212022, there are 5,013,333 Private Placement Warrants that are exercisable to purchase shares of Class A common stock to investors as well as 9,199,944 Public Warrants934 Public Warrants (see Note 3, Business Combination, for definition). All of the Company’s outstanding Warrants are recognized as assets or liabilities in accordance with ASC 815-40 and adjusts the warrant asset or liability to fair value at each reporting period. The assets or liabilities are subject to re-measurement at each balance sheet date until exercised, and any change in fair value is recognized in the statement of operations. The Public Warrants are traded on the NASDAQNasdaq and are recorded at fair value using the closing stockwarrant price (Ticker: PEARW) as of the measurement date. The Private Placement Warrants, which have a single holder, have similar terms and are subject to substantially the same redemption features as the Public Warrants. Accordingly, the most advantageous market for the Private Placement Warrants is determined from the perspective of the holder of such warrants as
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 130
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
an asset. Since any transfer to a non-permitted transferee (i.e., to a market participant) would cause the Private Placement Warrants to become public warrants, the fair value of the Private Placement Warrants is based on the quoted price of the Public Warrants.
Earn-Out Liabilities
In connection with the Business Combination, holders of Legacy Pear Common Shares and Legacy Pear Preferred Shares received the contingent right to receive additional Class A common stock (the “Earn-Out SharesEarn-Out Shares”) upon the achievement of certain earn-out targets. As the contingent earn-out consideration contains a settlement provision that precludes it from being indexed to the Company’s stock, it is classified as a liability under ASC 480. The fair value of the contingent earn-out consideration is estimated as of the acquisition date at the present value of the expected contingent payments using a Monte Carlo simulation. The fair value estimates use unobservable inputs that reflect our own assumptions as to the Company’s ability to meet the earn-out targets and discount rates used in the calculations. The unobservable inputs are defined in ASC Topic 820, “Fair Value Measurements and Disclosures,” as Level 3 inputs.
We review the probabilities of achievement of the earn-out targets to determine the impact on the fair value of the earn-out consideration on a quarterly basis over the earn-out period. Actual results are compared to the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 136
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
estimates and probabilities of achievement used in our forecasts. Should actual results of the business increase or decrease as compared to our estimates and assumptions, the estimated fair value of the contingent earn-out consideration liability will increase or decrease, up to the contractual limit, as applicable. Changes in the estimated fair value of the contingent earn-out consideration are recorded in other (expense) income in the Consolidated Statements of Operations and Comprehensive Loss and are reflected in the period in which they are identified. Changes in the estimated fair value of the contingent earn-out consideration may materially impact or cause volatility in our operating results.
Revenue Recognition
The Company recognizes revenue in accordance with ASU 2014-09, Revenue from Contracts with Customers (Topic 606) and its related amendments, or, collectively, ASC 606.
“ASC 606”. At inception, the Company determines whether contracts are within the scope of ASC 606 or other topics. For contracts that are determined to be within the scope of ASC 606, revenue is recognized when a customer obtains control of promised goods or services. The amount of revenue recognized reflects the consideration to which the Company expects to be entitled to receive in exchange for these goods and services. To achieve this core principle, the Company applies the following five steps :
(i) identify the contract with the customer;
(ii) identify the performance obligations in the contract;
(iii) determine the transaction price;
(iv) allocate the transaction price to the performance obligations in the contract; and
(v) recognize revenue when the performance obligation is satisfied.
The Company only applies the five-step model to contracts when it determines that collection of substantially all consideration for goods or services that are transferred is probable based on the customer’s intent and ability to pay the promised consideration.
Performance obligations promised in a contract are identified based on the goods and services that will be transferred to the customer that are both capable of being distinct and are distinct in the context of the contract. To the extent a contract includes multiple promised goods and services, the Company applies judgment to determine whether promised goods and services are both capable of being distinct and distinct in the context of the contract. If these criteria are not met, the promised goods and services are accounted for as a combined performance obligation.
The transaction price is determined based on the consideration to which the Company will be entitled in exchange for transferring goods and services to the customer. To the extent the transaction price includes variable consideration, the Company estimates the amount of variable consideration that should be included in the transaction price utilizing either the expected value method or the most likely amount method, depending on the nature of the variable consideration. Variable consideration is included in the transaction price if, in management’s
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 131
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
judgment, it is probable that a significant future reversal of cumulative revenue under the contract will not occur. Any estimates, including the effect of the constraint on variable consideration, are evaluated at each reporting period for any changes. DeterminingConsideration payable to customers is recorded as a reduction of the transaction price requires significant judgment, which is discussed in further detail for each of the Company’s collaboration agreements in Note 9. In addition, none. None of the Company’s contracts as of December 31, 2022 or 2021, contained a significant financing component.
If the contract contains a single performance obligation, the entire transaction price is allocated to the single performance obligation. Contracts that contain multiple performance obligations require an allocation of the transaction price to each performance obligation on a relative standalone selling price basis unless the transaction price is variable and meets the criteria to be allocated entirely to a performance obligation or to a distinct service that forms part of a single performance obligation. The Company typically determines standalone selling prices using an adjusted market assessment approach model contained a significant financing component.
The Company satisfies performance obligations either over time or at a point in time. Revenue is recognized over time if either (i) the customer simultaneously receives and consumes the benefits provided by the entity’s performance, (ii) the entity’s performance creates or enhances an asset that the customer controls as the asset is created or enhanced, or (iii) the entity’s performance does not create an asset with an alternative use to the entity and the entity has an enforceable right to payment for performance completed to date. If the entity does not satisfy a performance obligation over time, the related performance obligation is satisfied at a point in time by transferring the control of a promised good or service to a customer.
Product Revenue Recognition—Product revenue includes sales of the Company’s products, reSET, reSET-O and Somryst, all of which are PDTs that have received marketing authorization by the FDA. reSET and reSET-O are FDA-authorized, 12-week, prescription-only therapeutics for substance use disorder, or SUD, and opioid use disorder, or OUD“SUD”, and opioid use disorder, or “OUD”, respectively, to be used as adjuncts to standard outpatient treatment. Our third product, Somryst, achieved FDA marketing authorization in March 2020, for the treatment of chronic insomnia and the Company began to commercialize Somryst in October 2020. Sales of our products include multiple performance obligations, some of which are satisfied at the point in time when the products are for the treatment of chronic insomnia and the Company began to commercialize Somryst in October 2020. Each PDT is sold as an integrated product which includes the patient prescription-only
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 137
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
therapeutics and clinician access to our proprietary clinician dashboard, PearMD, to track the patient’s progress. We enter into agreements with state and local governments, health care providers and payors, to provide prescriptions which typically provide for volume-based discounts and other discounts (collectively “Access Agreements”). We also enter into arrangements with health care providers and payors that provide for government-mandated and/or privately negotiated rebates and discounts with respect to the purchase of our products. The products can made available to the customer (under a bulk order) or when a prescription is fulfilled, and some of which are recognized over the term of the contract or over the prescription period.
For the year ended December 31, 2021, the majority of our revenue was generated through bulk purchases from three customers, all of whom were state governments or agencies, who represented 34%, 23%, and 10% respectively of total revenue.
Licensing, Milestone and Contract Revenue—Prior to 2021, the Company’s revenue has primarily been generated through collaborative research, development and commercialization agreements, under which it out-licensed certain rights to its products to third parties. The terms of these agreements generally contained multiple performance obligations, which included (i) licenses, or options to obtain licenses, to the Company’s technology and (ii) research and development activities to be performed on behalf of the collaborative partner. Payments to the Company under these arrangements typically include one or more of the following: nonrefundable, up-front license fees; funding of research and/or development efforts; development, regulatory and commercial milestone payments; royalties on net sales of licensed products; and profit-share payments. Each of these payments resulted in license and collaboration revenue, except for revenue from royalties on net sales of licensed products, which are classified as royalty revenue.be sold with and without training and implementation services.
Licenses of Intellectual Property—If the license to the Company’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Company recognizes revenue from consideration allocated to the license when the license is transferred to the customer and the customer is able to use and benefit from the licenses. For licenses that are combined with other promises, the Company utilizesThe portion of the product revenue for PDT’s is recognized when the products are made available to the customer (via Access Agreements) or when a prescription is fulfilled (via third party reimbursement), and the portion of the product revenue related to the clinician’s access to our proprietary clinician dashboard is deferred and recognized ratably over the remaining term of the contract (if purchased via an Access Agreement) or the prescription duration (if purchased via third party reimbursement). Our proprietary clinician dashboard does not meet the definition of a license to intellectual property as the customer is not able to take possession of the underlying software or contract with a third party to host the dashboard.
Training, Implementation, and Other Professional Services—Training, implementation, and professional services revenue is recognized as control of these services is transferred to customers.
Subscription and Support Revenue Recognition—Subscription and support revenues are comprised of fees that provide customers with access to software licenses and related support and updates during the term of the arrangement. Subscription and support revenues are recognized ratably over the contract terms beginning on the commencement date of each contract, which is the date the Company’s service is made available to the customer and the customer is able to use and benefit from the service.
Multiple Deliverable Arrangements—If the contract contains a single performance obligation, the entire transaction price is allocated to the single performance obligation. To the extent a contract includes multiple promised goods and services, the Company applies judgment to assess the nature of the combined performance obligation to determine whether the combined performance obligation is satisfied over time or atpromised goods and services are both capable of being distinct and distinct in the context of the contract. If these criteria are not met, the promised goods and services are accounted for as a combined performance obligation. Contracts that contain multiple performance obligations require an allocation of the transaction price to each performance obligation on a relative standalone selling price (“SSP”) basis unless the transaction price is variable and meets the criteria to be allocated entirely to a performance obligation or to a point in time and,distinct service that forms part of a single performance obligation. When goods and if over timeservices are sold separately, the appropriate method of measuring progress for purposes of recognizing revenue from nonrefundable, up-front fees. The CompanyCompany determines SSP based on observable prices, when available. When observable prices are not available, the Company determines SSP based on overall pricing objectives, which take into consideration observable data, market conditions and entity-specific factors. The Company typically determines standalone selling prices using an adjusted market assessment approach model, a cost-plus margin approach, or based upon observable prices.
Changes in the Estimated Transaction Price—The Company may occasionally recognize an adjustment in revenue in the current period for performance obligations partially or fully satisfied in the previous periods resulting from changes in estimates for the transaction price, including any changes to the Company’s assessment of whether an estimate of variable consideration is constrained. For the years ended December 31, 2022 and 2021, the impact on revenue recognized in the current period, from performance obligations partially or fully satisfied in the previous period, was not material.
Customer Concentration—For the year ended December 31, 2022, the majority of our revenue was generated through Access Agreements. For the year ended December 31, 2022, three customers a state government agency, a state department and a state, represented 18%, 14%, and 11%, respectively of total revenue. Additionally all of our subscription, support and professional services revenue, which represented 11% of total revenue, under a pilot offering of a new program by the Medicaid program of a state government. For the year ended December 31,
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 132138
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue recognition.
Research and Development Funding—Arrangements that include reimbursements of research and development costs are generally combined with other promises and are generally considered to have variable consideration. Variable consideration is included in the transaction price if, in the Company’s judgment, it is probable that a significant future reversal of cumulative revenue under the contract will not occur. If and when the Company assesses that these reimbursements are probable, the Company adjusts its estimate of the overall transaction price. Any such adjustments are recorded on a cumulative catch-up basis, which would affect collaboration revenue and earnings in the period of adjustment. Research and development funding arrangements that include milestones are recognized as described below.
Development Milestone Payments—At the inception of each arrangement that includes nonrefundable payments for contingent milestones, including preclinical research and development, clinical development and regulatory milestones, the Company evaluates whether the milestones are considered probable of being achieved and estimates the amount to be included in the transaction price using the most likely amount method. If it is probable that a significant revenue reversal would not occur, the associated milestone value is included in the transaction price. Milestone payments that are not within the control of the Company or the licensee, such as regulatory approvals, are not considered probable of being achieved until those approvals are received. The transaction price is then allocated to each performance obligation on a relative stand-alone selling price basis, for which the Company recognizes revenue as or when the performance obligation under the contract is satisfied.
At the end of each reporting period, the Company reevaluates the probability of the achievement of contingent milestones and the likelihood of a significant reversal of such milestone revenue and if necessary, adjusts its estimate of the overall transaction price. Any such adjustments are recorded on a cumulative catch-up basis, which would affect revenue and earnings in the period of adjustment. This regular reassessment may result in recognition of revenue related to a contingent milestone payment before the milestone event has been achieved.
Commercial Milestone Payments and Royalties—For arrangements that include sales-based royalties, including milestone payments based on the level of sales and the license is deemed to be the predominant item to which the royalties relate, the Company recognizes revenue at the later of (i) when the related sales occur or (ii) when the performance obligation to which some or all of the royalty has been allocated has been satisfied or partially satisfied.2021, three customers, a state division, and two state departments, represented 34%, 23%, and 10%, respectively of total revenue.
Deferred Revenue—In general, deferred revenue arises from amounts received or invoiced in advance of the culmination of the earnings process and is recognized as revenue in future periods as performance obligations are satisfied. Deferred revenue expected to be recognized within the next twelve months is classified as a current liability. Upfront payment contract liabilities resulting from the Company’s license agreements do not represent a financing component as the payment is not financing the transfer of goods or services, and the technology underlying the licenses granted reflects research and development expenses already incurred by the Company.
Commissions—During the years ended December 31, 20212022 and 20202021, the Company paid commissions to its internal sales team. The Company acts as a principal in the contracts with their partners as the Company controls the product, establishes the price, and bears the risk of non-performance. The Company records the revenue on a gross basis and commissions are recorded as a sales and marketing expense in the consolidated statements of operations and comprehensive loss. The Company recognizes its commission expense as a point-in-time expense as contract obligations are primarily completed within a one-year contract period and commissions are only paid on revenue recognized, not the total value of the contract.
Accounts Receivable and Allowance for Doubtful Accounts—In general, accounts receivable consists of amounts due from customers, net of customer allowances for cash discounts and chargebacks. Our contracts with customers have standard payment terms that generally require payment within 30 days. We analyze accounts that are past due for collectability and periodically evaluate the creditworthiness of our customers. As of December 31, 20212022 and 20202021, we determined an allowance for doubtful accounts was not required based upon our review of contractual payment terms and individual customer circumstances.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 133
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Stock-Based Compensation
The Company’s stock-based compensation program allows for grants of common stock options, restricted stock awards, and restricted stock units. Grants are awarded to employees and non-employees, including directors.
The Company accounts for stock-based compensation in accordance with ASC Topic 718, Compensation-Stock Compensation, or ASC 718“ASC 718”. ASC 718 requires all stock-based payments to employees and non-employees to be recognized as an expense in the consolidated statements of operations and comprehensive loss based on their fair values. The Company estimates the fair value of options granted using the Black-Scholes option pricing model, or Black-Scholes“Black-Scholes”. The fair value of the Company’s common stock is used to determine the fair value of restricted stock awards.
Stock-based compensation awards are subject to service-based vesting periods. Compensation expense related to awards to employees and non-employees with service-based vesting conditions are recognized on a straight-line basis based on the grant date fair value over the associated service period of the award, which is generally the vesting term. The Company classifies stock-based compensation expense in the consolidated statements of operations and comprehensive loss in the same manner in which the award recipient’s payroll costs are classified or in which the award recipient’s service payments are classified. See Note 1112, Stock-Based Compensation, for a description of the types of stock-based awards granted, the compensation expense related to such awards and detail of equity-based awards outstanding.
Black-Scholes requires inputs based on certain subjective assumptions, including (i) the expected stock price volatility, (ii) the expected term of the award, (iii) the risk-free interest rate, and (iv) expected dividends. Due to the lack of a public market for the Company’s common stock and continued lack of sufficient company-specific historical and implied volatility data, the Company has based its computation of expected volatility on the historical volatility of a representative group of public companies with similar characteristics to the Company. The historical volatility is calculated based on a period of time commensurate with the expected term assumption. The Company uses the simplified method as prescribed by the SEC Staff Accounting Bulletin No. 107, Share-Based
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 139
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Payment, to calculate the expected term for options granted to employees and non-employees, whereby the expected term equals the arithmetic average of the vesting term and the original contractual term of the options due to its lack of sufficient historical data. The risk-free interest rate is based on US Treasury securities with a maturity date commensurate with the expected term of the associated award. The expected dividend yield is assumed to be zero as the Company has never paid dividends and has no current plans to pay any dividends on its common stock. The Company recognizes forfeitures as they occur.
Due to the absence of an active market for the Company’s common stock prior to the Business Combination, the Company utilized methodologies in accordance with the framework of the American Institute of Certified Public Accountants Technical Practice Aid, Valuation of Privately-Held Company Equity Securities Issued as Compensation, to estimate the fair value of its common stock. In determining the exercise prices for stock options granted, the Company considered the estimated fair value of the common stock as of the measurement date. The estimated fair value of the common stock has been determined at each grant date based upon a variety of factors, including the illiquid nature of the common stock, arm’s-length sales of the Company’s capital stock (including convertible preferred stock), the effect of the rights and preferences of the preferred shareholders and the prospects of a liquidity event. Among other factors are the Company’s financial position and historical financial performance, the status of technological developments within the Company’s research, the composition, and ability of the current research and management team, an evaluation or benchmark of the Company’s competition, and the current business climate in the marketplace. Significant changes to the key assumptions underlying the factors used could result in different fair values of common stock at each valuation date.
Cost of Product Cost of Revenue
Cost of product revenue consists primarily of costs that are closely correlated or directly related to the delivery of the Company’s products, including pharmacy costs, royalties paid under license agreements related to our
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 134
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
commercialized products, amortization of milestone payments capitalized related to commercialized products, hosting costs, closely correlated or directly related to the delivery of the Company’s products, including pharmacy costs, royalties paid under license agreements related to our commercialized products, amortization of milestone payments capitalized related to commercialized products, hosting costs, third party purchased software and services, and personnel-related costs, including salaries and bonuses and employee benefits., employee benefits, and stock-compensation. In addition, it includes the costs directly related to the delivery of our Subscription, service, and professional Services revenue, such as third party software and third party call center.
Research and Development Costs
Research and development costs are expensed as incurred. Research and development expense primarily consists of expenses incurred in performing research and development activities, including salaries and bonuses, employee benefits, stock-based compensation, facility costs, depreciation, contract services and other outside vendors engaged in conducting development activities and clinical trials, as well as the cost of licensing technology and costs related to collaboration arrangements.
We also include in research and development expenses the costs associated with software development of PDTs to support future commercial opportunities. Development costs of software to be sold, leased, or otherwise marketed are subject to capitalization, beginning when technological feasibility for the product has been established and ending when the product is available for general release. Such costs have not been significant to date as the time lapse between technological feasibility and release is typically short and thus has been included in research and development costs. Costs incurred to enhance our existing products after the general release of the product are expensed in the period they are incurred and included in research and development costs in the accompanying consolidated statements of operations and comprehensive loss.
Milestone Payments
The Company, from time to time, will enter into strategic agreements with third parties, which give the Company rights to develop, market and/or sell PDTs, the rights to which are owned by such third parties. As a result of these agreements, the Company may be obligated to make payments to these third parties contingent upon the achievement of certain pre-determined criteria. For milestones achieved prior to marketing approval of the product, such payments are expensed as research and development. After marketing approval, any additional milestone payments are capitalized and amortized to cost of product
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 140
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
milestone payments are capitalized and amortized to cost of revenue over the remaining useful life of the asset. All capitalized milestone payments are tested for recoverability whenever events or changes in circumstances indicate that the carrying amounts may not be recoverable.
Selling, General, and Administrative Expenses
Selling, general, and administrative expenses consist primarily of personnel-related expenses, including salaries and bonuses, employee benefits, stock-based compensation and marketing expenses, costs to support the commercialization of our products, allocated facilities expenses, depreciation expenses, insurance, executive management travel and professional services expenses, including legal, talent acquisition, audit, accounting, and tax-related services. Facilities costs consist of rent and maintenance of facilities. Costs associated with advertising are expensed in the period incurred and are included in selling, general, and administrative expenses. Advertising expenses were $53,693334 and $15,561693 for the years ended December 31, 20212022 and 20202021, respectively.
Patent Costs
All patent-related costs incurred in connection with the filing and prosecuting patent applications are expensed as incurred due to the uncertainty about the recovery of the expenditure. Such amounts incurred are classified as selling, general, and administrative expenses in the accompanying consolidated statements of operations and comprehensive loss.
Income Taxes
The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the Company’s financial statements and tax returns. Deferred tax assets and liabilities are determined based upon the differences between the financial statement carrying amounts, and the tax bases of existing assets and liabilities for the loss and credit carryforwards using enacted tax rates expected to be in effect in the year in which the differences are expected to reverse. Deferred tax assets are reduced by a valuation
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 135
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
allowance if it is more likely than not that these assets may not be realized. The Company determines whether a tax position will be sustained upon examination. If it is not more likely than not that a position will be sustained, none of the benefit attributable to the position is recognized. The tax benefit to be recognized for any tax position that meets the more likely than not recognition threshold is calculated as the largest amount that is more than 50% likely of being realized upon resolution of the contingency. The Company accounts for interest and penalties related to uncertain tax positions as part of its provision for income taxes.
Comprehensive Loss
For the years ended December 31, 20212022 and 20202021, the Company’s comprehensive income (loss) consists of its net loss and unrealized gains and losses from investments.
Net Loss Per Share
The Company follows the two-class method when computing net loss per share, or EPS“EPS”, as the Company has issued shares that meet the definition of participating securities. The two-class method determines net loss per share for each class of common and participating securities according to dividends declared or accumulated and participation rights in undistributed earnings. The two-class method requires income available to common stockholders for the period to be allocated between common and participating securities based upon their respective rights to receive dividends as if all income for the period had been distributed.
Basic net loss per share attributable to common stockholders is computed by dividing the net loss attributable to common stockholders by the weighted average number of common shares outstanding for the period. Diluted net loss attributable to common stockholders is computed by adjusting net loss attributable to common stockholders to reallocate undistributed earnings based on the potential impact of dilutive securities. Diluted net loss per share attributable to common stockholders is computed by dividing the diluted net loss attributable to common stockholders by the weighted average number of common shares outstanding for the period, including potential dilutive common shares assuming the dilutive effect of common stock equivalents.
The Company’s convertible preferred stock contractually entitles the holders of such shares to participate in dividends but does not contractually require the holders of such shares to participate in losses of the Company. Accordingly, in periods in which the Company reports a net loss, such losses are not allocated to such participating securities. In periods in which the Company reports a net loss attributable to common stockholders, diluted net loss per share attributable to common stockholders is the same as basic net loss per share attributable to common stockholders, since dilutive common shares are not assumed to have been issued if their effect is anti-dilutive
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 141
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
dilutive common shares assuming the dilutive effect of common stock equivalents. The Company reported a net loss attributable to common stockholders for the years ended December 31, 20212022 and 20202021.
As the Merger has been accounted for as a reverse recapitalization, the consolidated financial statements of the merged entity reflect the continuation of the pre-merger Pear Therapeutics, Inc. financial statements; Pear Therapeutics, Inc. equity has been retroactively adjusted to the earliest period presented to reflect the legal capital of the legal acquirer, THMA. As a result, net loss per share was also retrospectively adjusted for periods ended prior to the Merger. See Note 3 for details of this recapitalization and Note 13, “Business Combination”, for details of this recapitalization and Note 14, Net Loss Per Share, for discussion of the retrospective adjustment of net loss per share.
Segment and Geographic Information
Operating segments are components of an enterprise about which separate financial information is available that is evaluated regularly by the chief operating decision-maker, or decision-making group, in deciding how to allocate resources and in assessing performance. The Company’s chief operating decision maker is its President and Chief Executive Officer, or CEO“CEO”. The Company views its operations as and manages its business in one operating segment operating exclusively in the US.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 136
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Emerging Growth Company Status
The Jumpstart Our Business Startups Act of 2012 permits an emerging growth company, or EGC“EGC”, such as us to take advantage of an extended transition period to comply with the new or revised accounting standards applicable to public companies until those standards would otherwise apply to private companies. The Company has elected to use this extended transition period under the JOBS Act until such time the Company is no longer considered to be an EGC, which means that when a standard is issued or revised, it has different applications for public or private companies, we will adopt the new or revised standard at the time private companies adopt the new or revised standard and will do so until such time that we either (i) irrevocably elect to “opt-out” of such extended transition period or (ii) no longer qualify as an EGC.
Deferred Offering Costs
Deferred offering costs include certain legal, accounting, consulting and other third-party fees incurred directly related to an anticipated business combination. The Company defers offering costs classified as a long-term asset until the closing or termination of the business combination. At the closing of the business combination, these costs are recorded in stockholders’ deficit as a reduction of additional paid-in capital. Should the business combination for which those costs relate no longer be considered probable of being consummated, all deferred offering costs will be charged to operating expenses in the statement of operations at such time.
Recently IssuedRecently Adopted Accounting Pronouncements Not Yet Adopted
In February 2016, the FASB, issued ASU 2016-02, Leases (Topic 842), which requiresLeases (Topic 842), as subsequently amended, which provides guidance requiring lessees to record most leases on the balance sheet and recognize the expenserecognize a right-of-use asset (“ROU”) and a lease liability on the balance sheet for substantially all leases, with the exception of short-term leases. Leases are classified as either finance or operating, with classification affecting the pattern of expense recognition in the statement of operations and comprehensive loss in a manner similar to current practice. ASU 2016-02 states that a lessee would recognize a lease liability for the obligation to make lease payments and a right-to-use asset for the underlying asset for the lease term. This standard is applicable to the Company for the annual reporting periods beginning after December 15, 2021. The Company adopted ASCstatements of operations. The Company’s adoption of 842 onthe standard effective January 1, 2022. See Leases above and expects the adoption of ASC 842 to incrementally increase our assets and liabilities, respectively by the right of use asset in the range of $10,500 to $11,700 and by the lease liabilities in the range of $11,500 to $12,700. The impact to the accumulated deficit is expected to be immaterial.
In June 2016, the FASB issued ASU 2016-13, Financial Instruments—Credit Losses: Measurement of Credit Losses on Financial Instruments (Topic 326)Note 8, Leases. The standard, including subsequently issued amendments, requires a financial asset measured at amortized cost basis, such as accounts receivable and certain other financial assets such as available for sale debt securities, to be presented at the net amount expected to be collected based on relevant information about past events, including historical experience, current conditions and reasonable and supportable forecasts that affect the collectability of the reported amount. The new standard is effective for annual reporting periods beginning after December 15, 2022. The Company is still evaluating the impact of ASU 2016-13 on the Company’s consolidated financial statements; however, it does not expect the impact to be material. , for further information.
In December 2019, the FASB issued ASU No. 2019-12, Income Taxes-Simplifying the Accounting for Income Taxes. ASU 2019-12 eliminates certain exceptions related to the approach for intra-period tax allocation, the methodology for calculating income taxes in an interim period and the recognition of deferred tax liabilities for outside basis differences. It also clarifies and simplifies other aspects of the accounting for income taxes. This standard is effective for annual reporting periods beginning after December 15, 2021, and interim periods within annual periods beginning after December 15, 2022. The Company is currently evaluating the potential’s adoption of the standard effective January 1, 2022, did not have a material impact that ASU 2019-12 will have onto its consolidated financial statements and related disclosures; however, it does not expect the impact to be material.
In August 2020June 2016, the FASB issued ASU 2020-06, Debt—Debt with Conversion and OtherNo. 2016-13, Financial Instruments - Credit Losses (Subtopic 470-20) and Derivatives and Hedging—Contracts in Entity’s Own Equity (SubtopicTopic 326): Measurement of Credit Losses on Financial Instruments (“ASU 2016-13”). ASU 2016-13 amends the guidance on the impairment of financial instruments. This update adds an impairment model (known as the current expected credit losses model) that is based on expected losses rather than incurred losses. Under the new guidance, an entity recognizes, as an allowance, its estimate of expected credit losses. In November 2019, the FASB issued ASU No. 2019-10, Financial Instruments - Credit Losses (Topic 326), Derivatives and Hedging (Topic 815-40). This update simplifies the accounting for convertible debt instruments by removing certain accounting separation models as well as the accounting for debt instruments with embedded conversion features that), and Leases (Topic 842). ASU 2019-10 changes the effective date of the credit loss standard (ASU 2016-13) to fiscal years beginning after December 15, 2022, including interim periods within those fiscal years for smaller reporting companies. Further, the ASU clarifies that operating lease receivables are not required towithin the scope of ASC 326-20 and should instead be accounted for as derivative instruments. The update also updates and improves the consistency of earnings per share calculations forunder
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 137142
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
convertible instruments. The amendments in this ASU are effective for fiscal years beginning after December 15, 2023, including interim periods within those fiscal yearsthe new leasing standard, ASC 842. The Company is currently evaluating the impactadopted ASU 2016-13 on January 1, 2022, and has determined that the implementationadoption of this update willguidance did not have a material impact on the Company’sits consolidated financial statements and related disclosures.
3. Business Combination
As discussed in Note 1, onOn December 3, 2021, the Company consummated a business combination pursuant to the Business Combination Agreement (“BCA”) with THMA dated June 21, 2021. The Business Combination was accounted for as a reverse recapitalization in accordance with GAAP. Under this method of accountingAccordingly, THMA, who was the legal acquirer, wasis treated as the “acquired” company for financial reporting purposes. Accordingly, the Business Combination was treated as; in other words the equivalent of Pear issuing stock for the net assets of THMA, accompanied by a recapitalization. Legacy Pear issuing stock for the net assets of THMA, accompanied by a recapitalization. In accordance with GAAP, Legacy Pear was deemed the accounting acquirer primarily based on Legacy Pear’s stockholders prior to the Business Combination having a majority of the voting power in the combined company, Legacy Pear having the ability to appoint a majority of the Board of Directors of the combined company, Legacy Pear’s existing management comprising the senior management of the combined company, Legacy Pear comprising the ongoing operations of the combined company, Legacy Pear being the larger entity based on historical revenues and business operations, and the combined company assuming Legacy Pear’s name. Under this method of accounting, the net assets of THMA are stated at historical cost, with no goodwill or other intangible assets recorded.
Upon the closing of the Business Combination, holdersPursuant to the terms of the Business Combination Agreement, each share of Legacy Pear common stock, par value $0.0001 per share (“Legacy Pear Common Shares”) issued and outstanding immediately prior to the closing of the Business Combination, after giving effect to the conversion of all issued and outstanding shares of Legacy Pear commonpreferred stock received shares of Class A common stock, par value $0.0001 per share (“Legacy Pear Preferred Shares”) to Legacy Pear Common Shares, were canceled and converted into the right to receive a number of shares of Class A common stock, inpar an amount determined by application ofvalue $0.0001 per share (“Class A common stock”) equal to the number of shares of Legacy Pear Common Shares multiplied by the exchange ratio of approximately 1.47 (the “Exchange Ratio”), which was based on Legacy Pear’s implied price per share prior toExchange Ratio”) established in the Business Combination which was based on Legacy Pear’s implied price per share prior to the Business Combination. Legacy Pear Preferred Shares previously classified as mezzanine were retroactively adjusted, converted into Class A common stock, and reclassified to permanent as a result of the reverse recapitalization. In addition, all outstanding equity awards of Legacy Pear were converted into equity awards with the option to purchase Class A common stock with the same terms and conditions adjusted by the exchange ratio of approximately 1.47. The equity structure has been restated in all comparative periods up to the Closing Date to reflect the number of shares of the Company’s Class A common stock, $0.0001 par value per share, issued to Legacy Pear stockholders in connection with the Business Combination. For periods prior to the Business Combination, the reported share and per share amounts have been retroactively converted (“Retroactive ConversionRetroactive Conversion”) by applying the Exchange Ratio. The consolidated assets, liabilities and results of operations
See Note 7, Indebtedness, for information on the Legacy Pear warrants that were exercised prior to the Business Combination are those of Legacy Pear..
In addition, holders of Legacy Pear Common Shares (and Legacy Pear Preferred Shares who converted their shares into Legacy Pear Common Shares in connection with the Merger) received the contingent right to receive up to 12,395,625 additional shares of Class A common stock (the “Earn-Out SharesEarn-Out Shares”) upon the achievement of certain earn-out targets. The holders of Legacy Pear common stockholders can contingentlyCommon Shares are eligible to receive up to 12,395,625 shares in the aggregate of additional shares of Class A common stock in three equal tranches of 4,131,875 shares respectively, upon the Company achieving $12.50, $15.00, or $17.50, respectively, as its volume-weighted average price per share for any 20 trading days within a 30 consecutive trading day period (as adjusted for share splits, reverse share splits, share dividends, reorganizations, recapitalizationsrecapitalization, reclassifications, combination, exchange of shares, or the like) during the period ending on December 3, 2026.
Further, the Company assumed the outstanding Public Warrantswarrants to purchase 9,199,944 shares of the Company’s Class A common stock at $11.50 per share and the outstanding Private Placement Warrants,(the “Public Warrants”) and the outstanding warrants (the “Private Placement Warrants”) held by LJ10 LLC, (the “Sponsor”) (the “Private Placement WarrantsSponsor”) to purchase 5,013,333 shares of the Company’s Class A common stock at $11.50 per share. The Public Warrants and Private Placement Warrants expire five years after the completion of the Business Combination.
five years after the completion of the Business Combination.Pear Therapeutics, Inc. | 2022 Form 10-K |Page 143
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
In connection with the Business Combination, the Company incurred approximately $32,779 of equity issuance costs, consisting of underwriting, legal, and other professional fees, $31,400 of which were recorded to additional paid-in capital as a reduction of proceeds and $1,379 of which was recorded as an expense in selling, general, and administrative expenses on the consolidated statement of comprehensive income.
See Note 7 for information on the Legacy Pear warrants thatIn connection with the Business Combination, THMA completed the sale and issuance of 10,280,000 shares of Class A common stock in a fully committed common stock private placement at a purchase price of $10.00 per share (“PIPE Shares”) for an aggregate purchase price of $102,800 (“PIPE Investment”), and a Forward Purchase Agreement Assignment, dated as of December 2, 2021, by and among THMA, the Anchor Investor, and a were exercised prior to the Business Combination.
PIPE investor (the “Forward Purchase Assignment”); which closed simultaneously with the consummation of the Business Combination. Upon the closing of the Business Combination, all of the remaining outstanding THMA Class A common shares were separated, pursuant to their terms, into one share of Class A common stock (which totaled 832,899 shares Class A common stock, “Public Shares”) and one-third (1/3) of one redeemable warrant (and THMA’s units ceased trading on the Nasdaq). Further, KLP SPAC 1 LLC (the “Anchor Investor”) purchased 6,387,026 shares of Class A common stock at a purchase price of $10.00 per share in connection with the Forward Purchase Agreement, dated as of February 1, 2021, by and between THMA and the Anchor Investor (the “Forward Purchase Agreement”), as amended from time to time, including by the Amendment to Forward Purchase Agreement dated as of June 21, 2021, and the Second Amendment to Forward Purchase Agreement dated as of November 14, 2021 (the “Amended Forward Purchase Agreement”), entered into with THMA on February 1, 2021 (“THMA Sponsor Shares”). Gross proceeds from the Business Combination totaled approximately $175,001 which included funds held in THMA’s trust account (after giving effect to redemptions). Transaction costs totaled approximately $32,779.The following table reconciles the elements of the Business Combination to the consolidated statement of cash flows and the consolidated statement of changes in equity:
| | | | | | | | |
| | Year Ended
December 31, 2021 |
| Cash - THMA trust and cash (net of redemptions) | | $ | 8,331 | |
| Cash - PIPE Investors and Forward Purchase Assignment | | 102,800 | |
| Cash - Forward Purchase Agreement (Anchor Investor) | | 63,870 | |
| Gross proceeds (excluding cash due at December 31, 2021 from the Trust Account) | | 175,001 | |
| Less: transaction costs and advisory fees paid | | (30,700) | |
| Net proceeds from the Business Combination | | 144,301 | |
| Less: warrant liabilities assumed | | (16,487) | |
| Less: repayment of note assumed in the Business Combination | | (1,000) | |
| Less: accrued transaction costs at December 31, 2021 | | (700) | |
| Plus: cash receivable from Trust | | 345 | |
| Less: net liabilities assumed in the Business Combination | | (146) | |
| Reverse merger, net of transaction costs | | $ | 126,313 | |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 138144
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
The number of shares of common stock outstanding immediately following the consummation of the business combinationBusiness Combination was as follows:
| | | | | | | | |
| | Class A
Common Stock |
| THMA Public Shares | | 832,899 | |
| THMA Initial Stockholders | | 6,900,000 | |
Shares Issued pursuant to Forward Purchase Agreement to Anchor Investor | | 6,387,026 | |
| Shares Issued to PIPE Investors and Forward Purchase Assignment | | 10,280,000 | |
Legacy Pear Equityholders (1) | | 113,399,293 | |
| Total shares of common stock immediately after Business Combination | | 137,799,218 | |
(1) The number of Legacy Pear shares was determined from the shares of Legacy Pear shares outstanding immediately prior to the closing of the Business Combination converted at the Exchange Ratio of approximately 1.47. All fractional shares were rounded down.
Public Warrants and Private Placement Warrants
As of the Closing Date, the total value of the liability associated with the Public and Private Placement Warrants was $16,487 measured at fair value based on the public warrant quoted price. The Company concluded the warrants met the definition of a liability and have been classified as such on the balance sheet in accordance with the accounting policy described within Note 2. At December 31, 2021, the. The fair value of the warrant liability was $8,528. See Note 102,116 and $8,528 at December 31, 2022 and December 31, 2021, respectively. See Notes 4, Fair Value Measurements, and 11, Capital Stock, for further information on the Public and Private Placement Warrants.
Earn-Out Liabilities
The Company accounts for the potential issuance of the Earn-Out Shares as a contingent consideration arrangement, a liability for which was initially valued and recorded at $95,401, which was estimated by using a Monte Carlo Simulation Method (“MCSM”) for each earn out period. Key inputs and assumptions used in this were the Company’s stock price, expected term, volatility, the risk-free rate, and dividend yield. SomeCertain of these inputs are Level 3 assumptions that are updated each reporting period as the earn-out liabilities are recorded at fair value onat a recurring basiseach reporting date. The Company revalued the earn-out liabilities as of December 31, 2022 and December 31, 2021, and determined the fair value to be $48,3633,024 and $48,363, respectively. The change in the fair value of the earn-out liabilities were recorded in other income (expense) on the statement of operations.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 139145
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
4. FAIR VALUE MEASUREMENTS
The tables below present certain of our assets and liabilities measured at fair value categorized by the level of input used in the valuation of each asset and liability.
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | December 31, 2022 |
Description | | Total Fair Value | | Level 1 | | Level 2 | | Level 3 |
Cash equivalents: | | | | | | | | |
Money market funds | | $ | 32,921 | | | $ | 32,921 | | | $ | — | | | $ | — | |
| | | | | | | | |
| | | | | | | | |
Debt investments: | | | | | | | | |
| US Treasury bills | | 3,995 | | | 3,995 | | | — | | | — | |
Corporate bonds | | 6,976 | | | — | | | 6,976 | | | — | |
| | | | | | | | |
Total debt investments | | 10,971 | | | 3,995 | | | 6,976 | | | — | |
Total assets | | $ | 43,892 | | | $ | 36,916 | | | $ | 6,976 | | | $ | — | |
Long-term liabilities: | | | | | | | | |
| Embedded debt derivative | | $ | 1,245 | | | $ | — | | | $ | — | | | $ | 1,245 | |
Warrant liabilities | | 2,116 | | | 1,370 | | | 746 | | | — | |
Earn-out liabilities | | 3,024 | | | — | | | — | | | 3,024 | |
Total liabilities | | $ | 6,385 | | | $ | 1,370 | | | $ | 746 | | | $ | 4,269 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | December 31, 2021 |
Description | | Total Fair Value | | Level 1 | | Level 2 | | Level 3 |
Cash equivalents: | | | | | | | | |
Money market funds | | $ | 129,184 | | | $ | 129,184 | | | $ | — | | | $ | — | |
Debt investments: | | | | | | | | |
Corporate bonds | | 1,007 | | | — | | | 1,007 | | | — | |
Commercial paper | | 3,998 | | | — | | | 3,998 | | | — | |
Total debt investments | | 5,005 | | | — | | | 5,005 | | | — | |
Total assets | | $ | 134,189 | | | $ | 129,184 | | | $ | 5,005 | | | $ | — | |
Long-term liabilities: | | | | | | | | |
Embedded debt derivative | | $ | 675 | | | $ | — | | | $ | — | | | $ | 675 | |
Warrant liabilities | | 8,528 | | | 5,520 | | | 3,008 | | | |
Earn-out liabilities | | 48,363 | | | — | | | — | | | 48,363 | |
Total liabilities | | $ | 57,566 | | | $ | 5,520 | | | $ | 3,008 | | | $ | 49,038 | |
The Company evaluates transfers between levels at the end of each reporting period. There were no transfers of financial instruments between levels during the yearsyear ended December 31, 20212022 and December 31, 20202021.
Cash equivalents—Money market funds included within cash equivalents are classified within Level 1 of the fair value hierarchy because they are valued using quoted market prices in active markets.
Investments—The Company measures its investments at fair value on a recurring basis and classifies those instruments within Level 1 and Level 2 of the fair value hierarchy. US Treasury bills are classified within Level 1 of the fair value hierarchy because pricing is based on quoted market prices for identical instruments in active markets of the reporting date. Marketable securities, including corporate bonds and commercial paper, are classified within Level 2 of the fair value hierarchy because pricing inputs are other than quoted prices in active markets, which are either directly or indirectly observable as of the reporting date, and fair value is determined using models or other valuation methodologies. The Company recorded unrealized losses of $146 and $291 in other comprehensive income (loss) on short-term investments for the years ended December 31, 2021 and 2020, respectively. for the
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 140146
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Embedded debt derivative years ended December 31, 2022 and 2021, respectively, in other comprehensive income (loss) on short-term investments.
Embedded debt derivative— As described in Note 7As described in Note 7, Indebtedness, the Company concluded that the contingent put options contained in the Perceptive Credit Facility that could require mandatory repayment upon the occurrence of an event of default, change of control, and certain other events represent an embedded derivative required to be bifurcated from the debt host instrument. The embedded debt derivative is measured at fair value using a probability-weighted cash flow valuation methodology,. having aThe change in estimated fair value of the embedded derivative resulted in an expense of $675569 atfor the year ended December 31, 2020, the date2022, which is recorded in Interest and other income in the consolidated statement of issuanceoperations and December 31, 2021comprehensive loss. The determination of the fair value of an embedded debt derivative includes inputs not observable in the market and as such, represents a Level 3 measurement. The methodology utilized requires inputs based on certain subjective assumptions, specifically, probabilities of mandatory debt repayment prior to maturity ranging between 0-1050%.
Warrant liabilities — As a result of the Business Combination on December 3, 2021, the Company recorded a liability for Public and Private Placement Warrants to purchase Class A common stock in the Company’s consolidated financial statements. See Note 3, Business Combination, for further information. The Public Warrants are traded on Nasdaq and are recorded at fair value using the closing stockwarrant price as of the measurement date. The Private Placement Warrants, which have a single holder, have similar terms and are subject to substantially the same redemption features as the Public Warrants. Accordingly, the most advantageous market for the Private Placement Warrants is determined from the perspective of the holder of such warrants as an asset. Since any transfer to a non-permitted transferee (i.e., to a market participant) would cause the Private Placement Warrants to become public warrantsPublic Warrants, the fair value of the Private Placement Warrants is based on the quoted price of the Public Warrants.
As of the Closing Date, the total value of the liability associated with the Public and Private Placement Warrants was $16,487 measured at fair value based on the public warrantPublic Warrant quoted price on Nasdaq (Ticker: PEARW). The Company concluded that the warrants met the definition of a liability and have been classified as such on the balance sheet in accordance with the accounting policy described within Note 2. At December 31, 20212022, the fair value of the warrant liability wasliabilities were $82,528.
Prior to the Business Combination, Legacy Pear issued warrants to purchase Series A and Series D convertible preferred stock, which were exercised on December 1, 2021 and November 30, 2021, respectively, in connection with the Business Combination. The Company measured the fair value of the liabilities related to the Legacy Pear warrants to purchase Series A and Series D convertible preferred stock, using Level 3 inputs. As of December 31, 2020, the fair market value of the Series A and D warrants were estimated using an option pricing model (“OPM”).
The following significant assumptions used in the valuation model to estimate the fair value of the Legacy pear warrant liabilities were as follows:
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 141
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
The following table reconciles the change in the fair value of the Series A and Series D warrant liabilities valued using Level 3 inputs:
Earn-out liabilities 116.
Earn-out liabilities— Upon the closing of the Business Combination, the Earn-Out Shares were accounted for as a liability because the triggering events that determine the number of shares to be earned included events that were indexed to the common stock of the Company, with the change fair value recognized in Change in the estimated fair value of earn-out liabilities in the consolidated statement of operations.
and comprehensive loss.The estimated fair value of the Earn-out Shares was determined using a Monte Carlo Simulation Method (“MCSM”)MCSM using the following assumptions at each valuation date:
Current stock price: The stock price was based on the closing price as of the valuation date.
Risk-free interest rate: The risk-free interest rate is based on the US Treasury yield curve in effect at the time of issuance for zero-coupon US Treasury notes with maturities corresponding to the expected seven-year term of the earn-out period.
Expected term: The expected term is the contractual term of the earn-out period.
Expected volatility: The volatility rate was determined using an average of historical volatilities of selected industry peers deemed to be comparable to the Company’s business corresponding to the expected seven-year term of the awards.
Expected dividend yield: The expected dividend yield is zero as the Company currently has no history or expectation of declaring dividends in the foreseeable future.
Refer to Note 3 | | | | | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 |
| Stock price | | $1.18 | | $6.20 |
| Risk-free interest rate | | 4.07% | | 1.25% |
| Expected term (in years) | | 3.92 | | 4.92 |
| Expected volatility | | 71.70% | | 55.00% |
| Dividend yield | | —% | | —% |
Refer to Note 3, Business Combination, for more information on the triggering events of the Earn-Out Shares. The change in fair value of the earn-out liabilities resulted in other income of $4745,038339 recognized in the Consolidated Statement of Operationsconsolidated statement of operations and comprehensive loss for the year ended December 31, 20212022.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 142147
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
The following table reconciles the change in the fair value of the earn-out liabilities valued using Level 3 inputs:
| | | | | | | | |
| | Earn-Out Liabilities |
Fair value as of December 31, 2021 | | $ | 48,363 | |
Change in fair value | | (45,339) | |
Fair value as of December 31, 2022 | | $ | 3,024 | |
5. PROPERTY AND EQUIPMENT
Property and equipment, net consists of the following:
| | | | | | | | | | | |
| December 31, |
| 2022 | | 2021 |
Internal-use software | $ | 9,581 | | | $ | 6,591 | |
Equipment | 724 | | | 579 | |
Furniture and fixtures | 711 | | | 586 | |
Leasehold improvements | 779 | | | 509 | |
In process | — | | | 362 | |
Total property and equipment | 11,795 | | | 8,627 | |
Less: accumulated depreciation | (5,719) | | | (2,372) | |
Property and equipment, net | $ | 6,076 | | | $ | 6,255 | |
Depreciation expense was $13,689347 and $3811,689 for the years ended December 31, 20212022 and 20202021, respectively.
6. ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
Accrued expenses and other current liabilities consist of the following:
| | | | | | | | | | | |
| December 31, |
| 2022 | | 2021 |
Compensation and related benefits | $ | 10,075 | | | $ | 11,855 | |
| Commercial and marketing related costs | 845 | | | 1,821 | |
Professional services | 1,242 | | | 1,710 | |
Research and development costs | 708 | | | 781 | |
| | | |
| | | |
| Amount due under non-cancelable purchase obligation | 1,755 | | | — | |
Other | 1,839 | | | 1,779 | |
Total | $ | 16,464 | | | $ | 17,946 | |
7. INDEBTEDNESS
Perceptive Credit Facility
On June 30, 2020, the Perceptive Close Date, the Company entered into a Credit Agreement and Guaranty, orthe Perceptive Credit Facility, with Perceptive Credit Holdings III, LP, as administrative agent and lender with a syndicate of other lenders, collectively with Perceptive. The Perceptive Credit Facility, as amended, consists of a secured term loan facility in an aggregate amount of up to $50,000, which will be made available under the following three tranches: (i) Tranche 1 - $30,000, available at the Perceptive Closing Dateclosing on June 30, 2020; (ii) Tranche 2 - $10,000, available no earlierlater than December 31, 2021; and (iii) Tranche 3 - $10,000, available no later than December 31, 2021. The Company did not draw down on the available borrowings under Tranche 2 or Tranche 3.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 143148
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
The Perceptive Credit Facility will bearbears interest through maturity at a variable rate. Through January 31, 2023, this rate was based upon the one-month LIBOR rate plus 11.0%, subject to a LIBOR floor of 1.0%. As of December 31, 20212022, the interest rate was 1215.0%. The Company is required to make interest-only payments1%. On January 13, 2023, the Company and Perceptive executed the First Amendment to the Amended and Restated Credit Agreement and Guaranty, which replaced the LIBOR benchmark rate with the Secured Overnight Financing Rate (“SOFR”), effective February 1, 2023.
The Company is required to make interest-only payments under this facility until May 31, 2024, after which point the Company will be required to make monthly payments of principal equal to 3.0% of the then outstanding principal until maturity on June 30, 2025, or the Maturity Date“Maturity Date”. If the Company prepays the loan prior to the Maturity Date, it will be required to pay a prepayment fee guaranteeing Perceptive a 1.5 times return on any prepaid amount. As of December 31, 2022, the prepayment penalty was $4,800. A change of control, which includes a new entity or group owning more than 35.0% of the Company’s voting stock, or prior to an IPO, the failure of the existing holders to own at least 35.0% of the Company’s voting stock, trigger triggers a mandatory prepayment of the term loan. The Business Combination did not trigger this clause as existing holders retained greater than 35% of the combined Company’s voting stock. The Company paid issuance costs of $750 in connection with its entry into the Perceptive Credit Facility.
The Company concluded the contingent put options that could require mandatory repayment upon the occurrence of an event of default, change of control, and certain other events represent an embedded derivative required to be bifurcated from the debt host instrument and accounted for separately and recorded an embedded debt derivative of $1,245 and $675 as of December 31, 20202022 and December 31, 2021, respectively. Any changes to the derivative liability in future periods will be recognized as interest and other (expense) income, net in the consolidated statements of operations and comprehensive loss.
The Perceptive Credit Facility is secured by substantially all the assets of the Company, including ourits intellectual property. The Perceptive Credit Facility requires the Company to (i) maintain a minimum aggregate cash balance of $5,000 in one or more controlled accounts, and (ii) as of the last day of each fiscal quarter commencing with the fiscal quarter ending March 31, 2022, report revenuesreport revenue for the trailing twelve (12-) month consecutive period that exceed the amounts set forth in the Perceptive Credit Facility which range from $518,750000 for the fiscal quarter ending MarchDecember 31, 2022, to $125,000 for the fiscal quarter ending March 31, 2025 31, 2025. The Company was not in compliance with the minimum revenue covenant for the quarter ending December 31, 2022, of $18,000 and was in compliance with the minimum cash balance covenant requirement of $5,000. The Perceptive Credit Facility contains various affirmative and negative covenants that limit the Company’s ability to engage in specified types of transactions. The Company was in compliance with the covenants under the Perceptive Credit Facility as of December 31, 2021obtained a waiver of the minimum trailing twelve (12) month revenue requirement for the period ended December 31, 2022 and the period ending March 31, 2023. The Company also obtained a waiver pertaining to the existence of a “going concern” qualification in the accompany opinion of the Company’s auditors in its Annual Report on Form 10-K and any resulting event of default.
As of the date of this filing, Perceptive has alleged that certain defaults or events of default have occurred and are continuing under the terms of the Perceptive Credit Facility. Perceptive has not delivered any formal notice of Default or Event of Default. To the extent that any such allegations are valid, Perceptive would have certain rights and remedies under the Perceptive Credit Facility. The Company disputes the allegations and is in discussions with Perceptive to resolve this dispute and otherwise to address the Company’s obligations under the Perceptive Credit Facility. There can be no assurances that such discussions will result in any resolution, and any resolution, or the lack of any resolution, may result in Perceptive exercising remedies under the Perceptive Credit Facility.
The total minimum revenue covenant required for the next twelve months are as follows:
| | | | | | | | |
| Twelve-Month Period Ended | | Minimum Total Revenue |
| March 31, 2023 | | $ | 26,000 | |
| June 30, 2023 | | $ | 44,000 | |
| September 30, 2023 | | $ | 69,000 | |
| December 31, 2023 | | $ | 100,000 | |
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 149
Pear Therapeutics, Inc.
On the Perceptive Closing DateNOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
On June 30, 2020, Perceptive received a warrant certificate exercisable into 775,000 shares of Legacy Pear Series C preferred stock, and had the Company borrowed under Tranche 2 or Tranche 3, the Company would have been obligated to issue two additional warrants, the Additional Warrants, to Perceptive each to purchase up to 50,000 shares of Legacy Pear Series C preferred stock. In the event the Company issued Legacy Pear Series D preferred stock, Perceptive had the right to convert the Legacy Pear Series C preferred stock warrant into a warrant to purchase Legacy Pear Series D preferred stock, and the exercise price shall bewould have been automatically adjusted to equal the original per share price for Legacy Pear Series D preferred stock. On the Perceptive Closing DateJune 30, 2020, the Company issued freestanding Legacy Pear Series C preferred stock warrants to Perceptive, which were converted to Legacy Pear Series D preferred stock warrants at the time of the Legacy Pear Series D funding round. The Legacy Pear Series D preferred stock warrants were exercisable for 1,012,672 shares of Legacy Pear Series D preferred stock. The Legacy Pear Series D preferred stock warrants have an exercise price of $5.51 per share and would have expired in 2030 and were exercisable at any time prior to the ten-year anniversary of the Perceptive Closing Date of the Perceptive Credit FacilityJune 30, 2030. At issuance, the Company determined that the warrant iswarrants are liability-classified and would be remeasured at fair value each reporting period, with changes in fair value recorded in the consolidated statements of operations and comprehensive loss. The Additional Warrants would have been issued as warrants to purchase 65,333 shares of Legacy Pear Series D-1 preferred stock. On November 30, 2021, Perceptive net exercised 1,012,672 Legacy Pear Series D warrants pursuant to which Perceptive obtained 629,057 shares of Legacy Pear Series D-1 preferred stock in a cashless exercise, and subsequently converted the 629,057 shares of Legacy Pear Series D-1 preferred stock into 629,057 shares of Legacy Pear common stock which were then converted into 926,232 shares of Class A common stock as adjusted by the exchange ratio based on a per share price of $9.87 per share, the THMA closing price on June 22, 2021. See Notes 1 and 3 for more information.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 144
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
On MarchNature of the Business, and 3, Business Combination, for more information.
On June 25, 2022, we amended the Perceptive Credit Facility to adjust certain covenants under the agreement. The amendment included among other things, reducing the required minimum trailing 12-month revenue for the fiscal quarter ending March 31, 2022 through the fiscal quarter ending March 31, 2025.
On the Perceptive Closing Date30, 2020, the Company received proceeds of $28,500, net of fees and expenses of $1,500. As of December 31, 20212022, no further borrowsborrowings were taken under the Perceptive Credit Facility. The outstanding balance of the Perceptive Credit Facility was:
| | | | | | | | |
Perceptive Credit Facility | | December 31, 2022 |
Principal | | $ | 30,000 | |
Less: Debt issuance costs and discount at issuance | | (2,553) | |
Net carrying amount | | $ | 27,447 | |
As discussed in Note 1, Nature of the Business, due to the substantial doubt about the Company’s ability to continue operating as a going concern for twelve months from the issuance date of these financial statements, the amounts due as of December 31, 20212022, have been classified as current in the consolidated balance sheet. Future minimum payments, including contractual interest, under the Perceptive Credit Facility as of December 31, 20212022, are as follows:
| | | | | | | | |
| Years ending December 31, | | Amounts |
| 2023 | | $ | 3,650 | |
| 2024 | | 10,603 | |
2025 | | 24,039 | |
Total | | 38,292 | |
Less: | | |
Interest payable | | (8,292) | |
Unamortized debt issuance costs | | (2,553) | |
Current portion of long-term debt | | (27,447) | |
Long-term debt | | $ | — | |
SVB Term Loan (Extinguished June 30, 2020) 8. LEASES
On June 30, 2020, using the proceeds from the Perceptive Credit Facility, the Company paid the then outstanding principal of $14,889, outstanding interest, a termination feeAs described in Note 2, Summary of Signification Accounting Policies, the Company adopted Topic 842, Leases, as of January 1, 2022. Prior period amounts have of $169 and a final payment amount of $1,080, which was equal to 6.75% of all amounts borrowed under the term loan on its then existing loan and security agreement with Silicon Valley Bank, or SVB, or the SVB Term Loan resulting in a loss on extinguishment of debt of $998.
As part of entering into the amendments to the SVB Term Loan in 2019 and 2018, the Company issued warrants to purchase 17,019 and 28,486 shares of Legacy Pear common stock, respectively, at an exercise price of $1.60 and $1.05 per share, respectively, in cash or pursuant to the net exercise provisions of the warrants. The Company estimated the fair value of the warrants at issuance to be $27 and $30, respectively, using the Black Scholes option-pricing model. The warrants would have expired on June 28, 2029 and June 8, 2028, respectively. In addition, in connectionnot been adjusted and continue to be reported in accordance with a borrowing in 2020 the Company issued warrants to purchase 35,817 shares of common stock at an exercise price of $1.60 per share in cash or pursuant to the net exercise provisions of the warrants. The Company estimated the fair value of the warrants at issuance to be $57, using the Black Scholes option-pricing model. The warrants are automatically exercised through cashless exercise if not exercised prior to the expiration date.
In addition, in 2016 with the initial SVB Term Loan, the Company granted the lender a Legacy Pear Series A preferred stock warrant which is exercisable for 32,711 shares of Legacy Pear Series A preferred stock. The exercise price of the Legacy Pear Series A preferred stock warrant was $0.9171 per share and the Series A preferred stock
our historic accounting under Topic 840. Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 145150
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
warrant would have expired on December 22, 2026. On December 1, 2021, in connection with the Business Combination, described in Note 3, SVB net exercised warrants to purchase 32,711 Legacy Pear Series A preferred shares at $0.9171 per share and 28,486 and 52,836 Legacy pear common stock warrants at $1.05 and $1.60 per share, respectively for 81,322 shares of Legacy Pear common stock, par value $0.0001 per share, pursuant to which SVB received 30,673 Legacy Pear Series A Pear preferred shares and 73,547 Legacy Pear Common Shares, and subsequent conversion of the Legacy Pear Common Shares and Legacy Pear Series A preferred shares into 153,454 Class A common stock as adjusted by the exchange ratio of approximately 1.47 based on a per share price of $14.72, the per share consideration in connection with the merger.
8. COMMITMENTS AND CONTINGENCIES
LeasesLessee Arrangements
As of December 31, 20212022, the Company leases office space under non-cancelable operating leases in three cities,: Boston, Massachusetts, consisting of approximately 19,000 square feet that will expire on June 1, 2028, including approximately 900 square feet that the Company will taketook over on January 1, 2022, San Francisco, California, consisting of approximately 17,000 square feet that will expire on July 31, 2025, and Raleigh, North Carolina, consisting of approximately 7,700 square feet that will expire on May 31, 2026. We have the right and option to extend each of the Boston and Raleigh leases for a five year period.
We recognized rent expense of $2,798 and $2,067 for the years ended December 31, 2021 and 2020, respectively. The Company has $1,007 and $577 in deferred rent recorded within other long-term liabilities in the consolidated balance sheet as of December 31, 2021 and 2020, respectively.
Future commitments under non-cancelable lease agreements are as follows:
In addition to rent, certain leases require the Company to pay additional amounts for taxes, insurance, maintenance and other operating expenses.
Licenses Related to our Commercial Products
As of December 31, 2021In addition to rent, certain leases require the Company to pay additional amounts for taxes, insurance, maintenance, and other operating expenses.
All of the Company's leases are classified as operating leases. The Company does not have finance leases. The components of Lease ROU assets and Lease liabilities are included in the consolidated balance sheets. Operating lease liabilities are based on the net present value of the remaining lease payments over the remaining lease term. In determining the present value of lease payments, the Company used its incremental borrowing rate when measuring operating lease liabilities. As of December 31, 2022, the weighted average remaining lease term is 4.4 years and the weighted average discount rate used to determine the operating lease liability is 10%.
Cash paid for amounts included in the measurement of operating lease liabilities was $2,848 for the year ended December 31, 2022. The Company recognized of rent expense of $2,878 for the year ended December 31, 2022.
Future commitments under non-cancelable lease agreements primarily related to office space are as follows: | | | | | | | | |
| Years ending December 31, | | Lease Commitments |
| 2023 | | $ | 2,912 | |
| 2024 | | 3,176 | |
| 2025 | | 2,734 | |
| 2026 | | 1,587 | |
| 2027 | | 1,518 | |
| 2028 and thereafter | | 773 | |
| Total lease payments | | 12,700 | |
| Less: present value adjustment | | (2,540) | |
| Present value of total lease liabilities | | 10,160 | |
| Less: lease liabilities - current | | (1,984) | |
| Lease liabilities non-current | | $ | 8,176 | |
Disclosures related to periods prior to the adoption of ASC 842
For the year ended December 31, 2021, rent expense was $2,798 recorded on a straight-line basis over the lease term. Deferred rent is the difference between cash payments for rent and the expense recorded. The Company had $1,007 in deferred rent recorded within other long-term liabilities in the consolidated balance sheet as of December 31, 2021. These balances were reclassified upon the adoption of ASC 842 and are included in the respective current and long-term portion of operating lease liabilities in the consolidated balance sheets.
As of December 31, 2021, prior to the adoption of ASC 842, the estimated minimum future lease payments for the next five years and thereafter were as follows:
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 151
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
| | | | | | | | |
| Years ending December 31, | | Lease Commitments |
| 2022 | | $ | 2,809 | |
| 2023 | | 2,912 | |
| 2024 | | 3,176 | |
| 2025 | | 2,734 | |
| 2026 and thereafter | | 3,879 | |
| Total | | $ | 15,510 | |
Subleases
Effective January 1, 2023, the Company subleased 7,218 square feet of office space in Boston, Massachusetts with a stated term of one year and renewal options for two additional years in one year increments. Future minimum lease payments under sublease agreements as of December 31, 2022 are $383, excluding options to extend. This amount is not included in the lease commitment table above.
Effective January 6, 2023, the Company subleased the entirety of its 7,654 square feet of rentable space in Raleigh, North Carolina. The term of the sublease arrangement commenced on January 6, 2023 and expires twelve months thereafter. Future minimum lease payments under sublease agreements at commencement of the lease are $134, excluding options to extend. This amount is not included in the lease commitment table above. The agreement includes an option to renew for one twelve-month period, at which time a 3% rate increase will apply.
9. COMMITMENTS AND CONTINGENCIES
Licenses Related to our Commercial Products
As of December 31, 2022, the Company has four license agreements related to its commercialized products.
The Invention Science Fund I, LLC
The Company entered into a contribution and license agreement for Pharmaceutical Field of Use, or FOU“FOU”, with The Invention Science Fund I, LLC, or ISF, in February 2015, as amended on February 28, 2018, or ISF Contribution and License Agreement. The ISF Contribution and License Agreement superseded an original contribution and license agreement between the Company and ISF dated December 31, 2013. Under the ISF Contribution and License Agreement, ISF granted the Company certain licenses under specified patent rights to develop and commercialize licensed products either independently and/or with a drug combination product for use in connection with the treatment of central nervous system disorders. The ISF Contribution and License Agreement contains minimum annual royalty obligations. To the extent there are sales of a licensed product, the Company is required to pay low-
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 146
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
single-digit royalties on net revenue. For the years ended December 31, 2021 and December 31, 2020, thesingle-digit royalties on net revenue. The Company recorded minimum annual royalty fees of $1,050000 and $8001,050 to ISF for the years ended December 31, 2022 and 2021, respectively.
Red 5 Group, LLC
In January 2015, the Company entered into a software license agreement with Red 5 Group, LLC, or Red 5, and in March 2018, the parties entered into an amended and restated software license agreement, or Amended Red 5 Group License. Under the original software license agreement, Red 5 licensed to the Company certain technology and materials relating to the treatment of psychological and substance use disorders, pursuant to which the Company, received, inter alia, an exclusive, worldwide, sublicensable, royalty-bearing license to develop and commercialize integrated products incorporating the licensed technology and materials. The Company agreed to use commercially reasonable efforts to develop integrated products in accordance with the development plan, to introduce any integrated products that gain regulatory approval into the commercial markets, to market integrated products that have gained regulatory approval following such introduction into the market, and to make integrated products that have gained regulatory approval reasonably available to the public.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 152
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
In March 2018, pursuant to the Amended Red 5 Group License, the parties expanded the scope of exclusivity of the license, increased certain specified annual license maintenance fees, and required the Company to pay Red 5 an amendment fee, which was paid in April 2018. On July 1, 2021, the parties amended the Amended Red 5 Group License to further clarify certain terms and increase the royalty rate by a de minimisde minimis amount.
To the extent achieved, the Company is obligated to pay up to an aggregate of $400 if certain milestones related to product regulatory approval and commercial sales are achieved in respect to a software/drug combination, which is not currently being pursued by the Company. To the extent there are sales of an integrated product, the Company is required to pay single-digit royalties on net revenues. The Company is entitled to certain reductions and offsets against its royalty and milestone payment obligations, including the annual license maintenance fees.
On July 1, 2021, the parties amended the Amended Red 5 License Agreement to further clarify certain terms and increase the royalty rate by a de minimis amount.
The Company pays minimum annual maintenance fees to Red 5 Group, LLC, or Red 5, in connection with reSET and reSET-O. For the years ended December 31, 2021 and December 31, 2020, theThe Company recorded minimum annual maintenance fees of $250 and $103250 to Red 5 for the years ended December 31, 2022 and 2021, respectively.
BeHealth Solutions, LLC and University of Virginia Patent Foundation
In March 2018, the Company and BeHealth Solutions, LLC, or BeHealth“BeHealth”, entered into an assignment, license and services agreement, or the BeHealth Agreement“BeHealth Agreement”, as well as a consulting agreement. The BeHealth Agreement closed in June 2018 and the Company paid an up-front fee. Under the BeHealth Agreement, the Company obtained license rights to certain technology and materials relating to a therapeutic treatment for insomnia. The consulting agreement is for services to be charged on a time-and-materials basis.
During the year ended December 31, 2020, the Company paid a milestone payment to BeHealth of $750 upon the FDA’s marketing authorization of Somryst, a PDT intended for use in the treatment of adults with chronic insomnia. During September 2021, a commercial milestone under the license agreement with BeHealth was achieved and the Company paid $1,000 during the year ended December 31, 2021. There were no additional milestones achieved or paid during the year ended December 31, 2022. The milestone payments arewere initially capitalized in other long-term assets in the accompanying consolidated balance sheets and amortized on a straight-line basis to cost of product revenue over the estimated useful life of five years. While the Company continues to sell Somryst, during the quarter ended December 31, 2022, the Company’s forecasted future revenue and expense cash flow projections indicated the carrying amounts of this intangible asset may not be recoverable and therefore the Company recorded impairment expense of $788, which is included in cost of revenue in the Company's consolidated statement of operations, during the year ended December 31, 2022.
The BeHealth Agreement continues in force until the expiration of all milestone and royalty payment obligations, unless terminated earlier in accordance with its terms. The Company could be obligated to make payments of up to an additional $26,000 in the aggregate upon achievement of various commercial milestones and a mid-to-high-single-digit royalty on net sales.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 147
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
The Company pays royalties based on net revenues of the sales of Somryst to BeHealth Solutions, LLC, or BeHealth, and the University of Virginia Patent Foundation, or UVPF“UVPF”. The Company recorded de minimis royalties to BeHealth and UVPF for the years ended December 31, 20212022 and 2020, respectively2021.
Guarantees and Indemnifications
As permitted under Delaware law, the Company indemnifies its officers, directors and employees for certain events or occurrences that happen by reason of the relationship with, or position held at, the Company. In addition, the indemnification agreements entered into with our former board members, Messrs. Schwab and Lynch, also provide certain indemnification rights to the entities with which they are affiliated. The Company maintains director and officer liability insurance coverage that would generally enable it to recover a portion of any future amounts paid. The Company may also be subject to indemnification obligations by law with respect to the actions of its employees under certain circumstances and in certain jurisdictions. Further, the Company is a party to a variety of agreements in the ordinary course of business under which it may be obligated to indemnify third parties with respect to certain matters. For the years ended December 31, 2021
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 153
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
parties with respect to certain matters. For the years ended December 31, 2022 and 20202021, the Company had not experienced any losses related to these indemnification obligations, and no claims were outstanding as of December 31, 20212022. The Company does not expect significant claims related to these indemnification obligations and consequently concluded that the fair value of these obligations is negligible and no related accruals were recorded.
Unconditional Purchase Commitment
On June 17, 2021, and later amended on August 3, 2021 and December 12, 2022, the Company entered into a non-cancelable purchase obligation for a subscription to the Palantir Foundry cloud platform, including support services, updates, and related professional services with Palantir for $9,300 payable over three years, continuing through September 30, 2024. Through December 31, 2021, the Company recorded $2,983 under the terms of the agreement, $2,503 of which is included in prepaid expenses on the consolidated balance sheet as of December 31, 2021.263 payable through September 30, 2024. As of December 31, 2022, under the terms of the agreement we have paid $4,438 and the Company has the following remaining unconditional purchase commit as detailed below:
| | | | | | | | |
| For the year ending December 31, | | Payments |
| 2023 | | $ | 2,325 | |
| 2024 | | 2,500 | |
| Total | | $ | 4,825 | |
Assignment and License Agreement
In November 2021, the Company and Waypoint Health Innovations, LLC (“WaypointWaypoint”) entered into an Assignment Agreement and Intellectual Property License Agreement (collectively, the “Waypoint AgreementWaypoint Agreement”). The Waypoint Agreement closed in December 2021, under which the Company obtained software, documentation, and other intellectual property rights relating to the therapeutic treatment of depression. At the same time, the Company entered into a consulting agreement with the Chief Executive Officer of Waypoint to provide certain services to Pear to be charged on a time-and-materials basis. The Company made an upfront payment of $1,350, and is required to make annual payments starting in the second half of 2022 of $250 per year through 2026 or until a commercial milestone payment is made under the agreement. The upfront payment and the net present value of the annual payments of $1,011 were initially capitalized and recorded as an intangible asset in consolidated balance sheet at December 31, 2021closing, and will bewere being amortized over five years. The net present value of the annual payments was recognized as a seller financing liability, and classified within accrued expenses and other current liabilities and other long-term liabilities on the balance sheet. Based upon the decision to pause development of its pipeline candidates indefinitely and our second reduction in our workforce, the Company recorded impairment expense of $2,091, which is included in research and development expenses in the Company's consolidated statement of operations related to this intangible asset during the year ended December 31, 2022.
The Company will be obligated to pay mid-single digit royalties on net revenues of any commercialized products that incorporate the assets obtained under the Waypoint Agreement. Additionally, the Company could be obligated to make payments of up to an additional $2,500 in the aggregate upon achievement of certain regulatory and commercial milestones. Through December 31, 2021, no2022, no royalties have been paid to Waypoint.
Legal Proceedings
The Company is also involved from time to time in various legal proceedings arising in the normal course of business. Although the outcomes of potential legal proceedings are inherently difficult to predict, the Company does not expect the resolution of these occasional legal proceedings to have a material adverse effect on its financial position, results of operations, or cash flow.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 148154
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
910. PAST COLLABORATION AND LICENSE AGREEMENT REVENUE AND CONTRACT BALANCES
Novartis Agreement (TerminatedContract Balances
The timing of revenue recognition, invoicing, and cash collections results in trade accounts receivable and unbilled receivables (contract assets) and deferred revenue (contract liabilities). We invoice our Access Agreement customers in June 2020)
In March 2018accordance with agreed-upon contractual terms, typically at the beginning of the agreement, or at periodic intervals throughout the contract term. Invoicing may occur subsequent to revenue recognition, resulting in unbilled receivables, or in advance of services being provided, resulting in deferred revenue. Deferred revenue that will be recognized during the twelve-month period from the balance sheet date is recorded as current deferred revenue and the remaining portion is recorded as non-current deferred revenue, which is included in Other long-term liabilities in the accompanying consolidated balance sheets. The following table summarizes the balances of our contract assets and liabilities:
| | | | | | | | | | | | | | |
| | December 31, |
| | 2022 | | 2021 |
Contract assets | | | | |
Trade accounts receivable | | $ | 2,425 | | | $ | 555 | |
Unbilled receivables | | 4,518 | | | 1,239 | |
Contract liabilities | | | | |
| Deferred revenue - current | | 284 | | | 421 | |
| Deferred revenue - non-current | | 12 | | | 22 | |
During the year ended December 31, 2022, the Company recognized revenue of approximately $421 that was included in deferred revenue at December 31, 2021.
Collaboration Arrangements
On March 15, 2022, the Company entered into a collaborationproof of concept agreement with Novartis Institutes for Biomedical Research, or NIBR, or the Novartis AgreementSoftBank Corp., an entity under common control with SVF II Cobbler (DE) LLC, a greater than 5% shareholder of the Company, to develop PDTsa Japanese-language digital therapeutic for Schizophrenia and Multiple Sclerosis, or MS. The two companies worked together towards developing new treatments for patients with schizophrenia and MS to better address the full burden of their illnesses.
On March 30, 2020, NIBR terminated the collaboration agreement for convenience with the Company relative to both the schizophrenia and MS programs. The termination was subject to a 90-day notice period and was effective as of June 29, 2020.
Under the Novartis Agreement, the Company received a non-refundable up-front payment of $5,000. The agreement included research and development funding, as well as future regulatory and commercial milestones and mid-to-high single-digit royalties on net sales of the two products. Contemporaneous with the Novartis Agreement, NIBR purchased 1,158,292 shares of Legacy Pear Series B preferred stock at $4.3167 per share, resulting in aggregate proceeds to the Company of $5,000. The Company allocated the proceeds from NIBR’s purchase of Legacy Pear Series B preferred stock to stockholders’ equity since the purchase price was equal to the price paid by other Legacy Pear Series B investors and the transaction was completed within a short period following the initial Legacy Pear Series B closing. The Company invoiced NIBR and receivedthe treatment of sleep/wake disorders for the Japanese market. The Company fulfilled all of its performance obligations during 2022 and recognized approximately $3,105632 for reimbursement of research and development costscollaboration revenue during the year ended December 31, 20202022.
NIBR had a buyout option with respectSubscription, Support, and Professional Services Agreement
In December, we entered into an agreement with one customer to each of the Schizophreniaprovide, maintain, and MS products, together the Buyout Option. If the Buyout Option was exercised, the license would have converted to a fully paid, perpetual, irrevocablesupport software to calculate, disburse, track and exclusive (asreport incentives to the application) license. The Company determined that the Buyout Option was not priced at a significant discount due to the early nature of both the development efforts and the PDTs market as a whole. Accordingly, the Buyout Option did not represent a material right and was not considered a separate performance obligation.
The Company concluded that the license agreement is within the scope of ASC 606. The Company identified several material promises under the Novartis Agreement, including (i) the license of intellectual property, (ii) continued technical development, regulatory and preclinical activities of the products and (iii) participating on a JSC. The Company determined that the material promises were not distinct from one another and, therefore, combined them as a single performance obligation, or the Novartis Combined License and Development.
The NIBR initial transaction price was composed solely of the $5,000 up-front payment and the initial agreed-upon reimbursement of costs related to certain technical and clinical development activities, or the Pear Novartis Development Efforts. Any potential future payments pursuant to additional Pear Novartis Development Efforts or achievement of regulatory, net sales milestones or royalties were evaluated under the most likely amount method and were not included in the Novartis initial transaction price because the amounts were considered variable consideration and were initially fully constrained. As part of the evaluation of the constraint, the Company considered numerous factors, including that receipt of such payments was largely outside the control of the Company. These amounts were added to the transaction price when the related regulatory or net sales milestones are achieved or as relates to Pear Novartis Development Efforts, as additional amounts are formally approved by the JSC. Due to the lack of historical perspective on efforts necessary to commercialize PDTs, future reimbursements related to continued Pear Novartis Development Efforts could not be reasonably estimated beyond the amounts that were approved at the JSC annually for the succeeding 12 months. Adjustments were recorded on a cumulative catch-up basis. Due to a milestone achievement and increases in JSC approved budgets, the transaction price was increased, resulting in a cumulative catch-up impact on collaboration revenue of approximately $600customer's participants and in accordance with the customer's protocols, including a call center and certain professional services. The subscription service arrangement is cancellable with three months’ notice and fees paid to date are non-refundable. The transaction price for the subscription service arrangement, as of December 31, 2022, was $3,400, and does not include expected consideration related to professional services not yet performed, for which the Company elected to recognized revenue in the amount it has a right to invoice. The Company began performing under this agreement in December 2022, and recognized approximately $1,405 forof subscription, support and professional service revenue during the year ended December 31, 2019.2022.
As the Company identified only
11. CAPITAL STOCK
The Company’s authorized capital stock consists of (a) 690,000,000 shares of common stock, par value $0.0001 per share; and (b) 10,000,000 shares of preferred stock, par value $0.0001 per share. As of December 31, 2022, there were 140,454,086 shares of Class A common stock issued and outstanding and 14,213,267 Warrants to purchase the Company’s Class A one distinct performance obligation in the Novartis Agreement, the entirety of the NIBR transaction price was allocated to the Novartis Combined License and Development and was being common stock outstanding. As of December 31, 2022, there were no shares of preferred stock issued or outstanding.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 149
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
recognized over the expected term of the agreement, which has been estimated at five years, ratably over the performance period, which, in management’s judgment, was the best measure of progress toward satisfying the performance obligation. The Company utilizes judgment to assess the nature of the combined performance obligation and the appropriate method of measuring progress for purposes of recognizing revenue. The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue recognition.
In connection with the termination of the agreement, on the effective date of the termination, June 29, 2020, deferred revenue of $8,261 was recognized.
As of December 31, 2020, there was no remaining deferred revenue from NIBR under this agreement.
10. CAPITAL STOCK
The Company’s authorized capital stock consists of (a) 690,000,000 shares of common stock, par value $0.0001 per share; and (b) 10,000,000 shares of preferred stock, par value $0.0001 per share. As of December 31, 2021, there were 137,836,028 shares of Class A common stock issued and outstanding and 14,213,277 Warrants to purchase the Company’s Class A common stock outstanding. As of December 31, 2021, there were no shares of preferred stock issued or outstanding.
Legacy Convertible Preferred Stock
In connection with the Business Combination, the Legacy Convertible Preferred Stock was retroactively adjusted, converted into Class A common stock, and reclassified to permanent equity as a result of the reverse recapitalization. As of December 31, 2021, there is no Legacy Convertible Preferred Stock authorized, issued or outstanding. The following table summarizes details of Legacy Convertible Preferred Stock authorized, issued and outstanding immediately prior to the Business Combination:
(1) Shares authorized, shares issued and outstanding, and the conversion price/share have been adjusted to reflect the exchange of Legacy Pear’s common stock for Class A common stock at an exchange ratio of approximately 1.47 as a result of the Business Combination. See Note 3 for further information.
The relevant features of the Legacy Pear Series A preferred stock, Legacy Pear Series B preferred stock, Legacy Pear Series C preferred stock and Legacy Pear Series D preferred stock, collectively, the Legacy Preferred Stock, prior to the conversion in the Business Combination, were as follows:
Conversion—The holders of Legacy Pear preferred stock had the right, at their option at any time, to convert any such shares of Legacy Preferred Stock into fully paid and nonassessable shares of Legacy Pear common stock The conversion ratio is determined by dividing the purchase price by the conversion price, which is equal to $0.9171, $4.3167, $7.1935, $6.5388, and $3.9458 per share for the Legacy Pear Series A, B, C, D-1, and D-2 preferred stock, respectively. The conversion price was subject to change if certain dilutive events occurred. Conversion was mandatory upon the closing of a firm commitment, underwritten initial public offering in which the aggregate net proceeds to the Company were at least $100,000 and have an offering price to the public of at least $11.01 per share or upon the election of a majority of the holders of the Legacy Preferred Stock.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 150
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Anti-Dilution Features—Legacy Pear Series D preferred stock contained anti-dilutive features apart from customary adjustments for splits and reverse splits of common stock (collectively, “down round features”). When a series of convertible preferred stock contains non-standard down round features, the Company is required to adjust the conversion price in the event of future stock sales at a lower unit price. In the event down round adjustments are triggered, the values attributable to the adjustment to the convertible preferred stock conversion price are recorded as an increase to additional paid-in capital and an increase to accumulated deficit. For the years ended December 31, 2020 and 2021 no down round features were triggered.
Voting Rights—The holders of the Legacy Preferred Stock voted together with the holders of common stock as a single class and each holder of outstanding shares of Legacy Preferred Stock was entitled to cast the number of votes equal to the number of whole shares of common stock into which the shares of Legacy Preferred Stock held by such holder are convertible as of the record date for determining stockholders entitled to vote on such matter. The holders of record of the shares of Legacy Pear Series D preferred stock, exclusively and as a separate class, were entitled to elect one director of the Company. The holders of record of the shares of Legacy Pear Series B preferred stock, exclusively and as a separate class, were entitled to elect one director of the Company. The holders of record of the shares of Legacy Pear Series A preferred stock, exclusively and as a separate class, were entitled to elect three directors of the Company; and any remaining directors were elected by the holders of record of shares of Legacy Preferred Stock and common stock, voting together as a single class on an as-converted to common stock basis.
Dividends—The holders of Legacy Convertible Preferred Stock were entitled to receive dividends at the rate of 8% of the original issue price for each series of Legacy Convertible Preferred Stock payable only when, as and if, declared by the Company’s board of directors. Through the date of the Business Combination, no dividends had been declared.
Liquidation Rights—In the event of any voluntary or involuntary liquidation, dissolution, or winding-up of the affairs of the Company, each holder of a share of the Legacy Pear Series D preferred stock then outstanding was entitled to be paid out of the assets of the Company available for distribution before any payment shall be made to the holders of Legacy Pear Series C, Legacy Pear Series B, Legacy Pear Series A preferred stock and common stock an amount equal to the original purchase price per share ($6.5388 and $3.9458 for Legacy Pear Series D-1 and D-2, respectively), plus any dividends declared, but unpaid thereon. From the remaining assets, Legacy Pear Series C preferred stock then outstanding was entitled to be paid out of the assets of the Company available for distribution before any payment shall be made to the holders of Legacy Pear Series B, Legacy Pear Series A preferred stock and common stock an amount equal to $7.1935 per share, plus any dividends declared, but unpaid thereon. From the remaining assets, Legacy Pear Series B preferred stock then outstanding was entitled to be paid out of the assets of the Company available for distribution before any payment shall be made to the holders of Legacy Pear Series A preferred stock and common stock an amount equal to $4.3167 per share, plus any dividends declared, but unpaid thereon. From the remaining assets, each holder of a share of the Legacy Pear Series A preferred stock then outstanding was entitled to be paid out of the assets of the Company available for distribution before any payment shall be made to the holders of common stock an amount equal to $0.9171 per share, plus any dividends declared, but unpaid thereon. Any remaining assets were to be distributed among the holders of the shares of Preferred Stock and common stock on a pro-rata basis.
Redemption Rights—Legacy Preferred Stockholders are not entitled to any redemption rights other than those under their liquidation rights previously discussed above.
Legacy Pear Series D Convertible Preferred Stock Issuance
On November 2, 2020, the Company authorized the issuance of 13,377,998 shares of Legacy Pear Series D-1 convertible preferred stock, or the Legacy Pear Series D-1 Preferred Stock, at $4.4400 per share and the issuance of 9,861,666 shares of Legacy Pear Series D-2 convertible preferred stock, or the Legacy Pear Series D-2 Preferred Stock, at $2.6800 per share. On that date, various existing and new investors entered into the Legacy Pear Series D preferred stock purchase agreements, or the Legacy Pear Series D Agreements, which resulted in the issuance of an aggregate 20,344,538 shares of Legacy Pear Series D preferred stock for aggregate gross cash proceeds of
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 151
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
$112,000. In conjunction with the issuance of Legacy Pear Series D preferred stock, the Company incurred issuance costs totaling $768. In connection with the Legacy Pear Series D-1 Preferred Stock and Legacy Pear D-2 Preferred Stock issuance, the Company recorded a loss of $16,819, which represents the amount by which the estimated fair value of the shares exceeded the cash proceeds received.
In addition, in connection with the Legacy Pear Series D-2 Preferred Stock offering, the Company commenced a tender offer to purchase up to 8,109,888 shares of Legacy Pear common stock or securities convertible into shares of Legacy Pear common stock from certain employees with tenure over four years, former employees and other stockholders, at a purchase price of $3.9433 per share for an aggregate cost of $31,980 excluding fees and expenses of $20. The tender offer expired on December 7, 2020. In connection with the tender offer, the Company purchased 2,788,732 shares of Legacy Pear common stock from the President and CEO, and 41,100 Legacy Pear common shares from certain eligible employees, at a purchase price of $3.9433 per share totaling $11,159 and resulting in a stock-based compensation expense of $7,254, representing the difference between the purchase price and the estimated fair value of the Legacy Pear common stock on the date of the sale. The Company also repurchased 1,656,467 Legacy Pear common shares from other shareholders for a total purchase price of 6,532. In total, the Company repurchased 4,486,299 shares of Legacy Pear common stock from investors having a total purchase price of $17,691.
In addition, the Company repurchased 3,617,798 shares of Legacy Pear Series A preferred stock and 5,791 shares of Legacy Pear Series B preferred stock from certain investors having a total purchase price of $14,289, which exceeded the carrying value of the Legacy Pear preferred stock by $11,053. The excess amount paid over the carrying value for the Legacy Pear common and Legacy Pear preferred stock repurchased from investors has been recorded as a reduction to additional paid-in capital and accumulated deficit in the consolidated statement of stockholders’ equity.
On February 23, 2021, the Company issued 3,058,665 shares of Legacy Pear Series D-1 convertible preferred stock at the price of $6.5388 per share, resulting in proceeds of $20,000, or the Legacy Pear Series D-1 Extension and incurred issuance costs totaling $83. In connection with the Legacy Pear Series D-1 Extension, the Company recorded a loss of $2,053, which represents the amount by which the fair value of the Legacy Pear shares exceeded the cash proceeds received.
As a result of the Business Combination, all of the shares of Legacy Pear stock were converted into Legacy Pear common stock. Each share of Legacy Pear common stock issued and outstanding immediately prior to the closing were canceled and converted into Class A common stock at an exchange ratio of approximately 1.47. See Note 1 for more information.
Warrants to Purchase Class A Common Stock
In THMA’s initial public offering, it sold units at a price of $10.00 per unit, which consisted of one share of Class A Common Stock, $0.0001 par value, and one-half of a redeemable warrant (each, a “Public Warrants”) that entitles the holders the right to purchase one share of our Class A common stock at a price of $11.50 per share and became exercisable as of 30 days from the date of the Business Combination. The Public Warrants may only be exercised for a whole number of shares, at any time commencing 30 days after the completion of the Business Combination155
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Warrants to Purchase Class A Common Stock
In THMA’s initial public offering, it sold units at a price of $10.00 per unit, which consisted of one share of Class A Common Stock, $0.0001 par value, and one-half of a redeemable warrant (each, a “Public Warrants”) that entitles the holders the right to purchase one share of our Class A common stock at a price of $11.50 per share and became exercisable as of 30 days from the date of the Business Combination. The Public Warrants are exercisable at any time and may only be exercised for a whole number of shares. A warrant holder may exercise its Public Warrants only for a whole number of Class A common stock. This means only a whole warrant may be exercised at a given time by a warrant holder. The Public Warrants expire five years after the completion of the Business Combination, or earlier upon redemption or liquidation.
The Company may redeem the outstanding Public Warrants in whole and not in part at a price of $0.01 per Pubic Warrant upon a minimum of 30 days’ prior written notice of redemption, and only in the event that the last sale price of the closing price of the Company’s Class A common stock equals or exceeds $18.00 per share for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which notice of redemption is given. If the Company redeems the Public Warrants as described above, it will have the option to require all Public Warrant holders that wish to exercise to do so on a “cashless basis”. As of December 31, 2021,
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 152
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
the Company did not redeem the outstanding Public Warrants. As of December 31, 2021the Company did not redeem the outstanding Public Warrants. As of December 31, 2022, there were 9,199,944934 outstanding Public Warrants.
The Company may redeem the outstanding Public Warrants in whole and not in part at a price of $0.10 per Pubic Warrant once the Public Warrants become exercisable, in whole and not in part at $0.10 per Public Warrant upon a minimum of 30 days’ prior written notice of redemption, provided that holders will be able to exercise their warrants on a cashless basis prior to redemption and receive that number of shares based on the redemption date and the fair market value of the shares of Class A common stock, and the closing price of the Class A common stock equals or exceeds $10.00 per share (as adjusted) on the trading day prior to the date on which the Company sends the notice of redemption to the warrant holders.
The exercise price and number of Class A common stock issuable upon exercise of the Public Warrants may be adjusted in certain circumstances including in the event of a share dividend, extraordinary dividend or recapitalization, reorganization, merger or consolidation. However, except as described below, the Public Warrants will not be adjusted for issuances of Class A common stock at a price below its exercise price.
Simultaneously with the consummation of THMA’s initial public offering, THMA Sponsor LJ1, LLC (the “Sponsor”) purchased an aggregate of 5,013,333 warrantsPrivate Placement Warrants to purchase one share of Class A common stock at an exercise price of $11.50 (the “Private Placement Warrants”) at a price of $1.50 per warrant, generating total proceeds of $7,520 in the aggregate in a private placement.
The Warrant Agreement, dated as of February 1, 2021, by and between the Company and Continental Stock Transfer & Trust Company also obligated the Company to use its best efforts to file with the SEC a registration statement for the registration, under the Securities Act of 1933, as amended (the “Securities Act”), of the issuance of the shares of Common Stock issuable upon exercise of the Public Warrants, and to cause the same to become effective and remain effective while the Public Warrants remain outstanding. On December 23, 2021, the Company’s registration statement covering the registration of such shares became effective.
The Private Placement Warrants are identical to the Public Warrants underlying the Units sold in the Initial Public Offering, except that, so long as they are held by the Sponsor or its permitted transferees: (1) they will not be redeemable by the Company for $0.01 if the criteria listed above under “Redemption of Warrants When the Price per Class A Ordinary Share Equals or Exceeds $18.00”; (2) they (including the ordinary shares issuable upon exercise of these warrants) may not, subject to certain limited exceptions, be transferred, assigned or sold by the Sponsor until 30 days after the completion of the Business Combination; (3) they may be exercised by the holders on a cashless basis; and (4) they (including the ordinary shares issuable upon exercise of these warrants) are entitled to registration rights.
11.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 156
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
At-the-Market (ATM) Offering
On January 3, 2023, the Company entered into an ATM offering agreement (the “ATM Agreement”) with H.C. Wainwright & Co., LLC (“Wainwright”) and Virtu Americas LLC (“Virtu” and, collectively with Wainwright, the “Managers” and each, a “Manager”), pursuant to which the Company may offer and sell, from time to time through the Managers, shares of the Company’s Class A common stock. The ATM Agreement authorized an aggregate gross proceeds of up to $150,000. Sales of common stock through the Manager could be made by any method that is deemed an “at-the-market” offering as defined in Rule 415 promulgated under the Securities Act of 1933, as amended, including by means of ordinary brokers’ transactions at market prices, in block transactions or as otherwise agreed by the Company and the Managers. The Company will pay the designated Manager a commission of up to 3.0% of the aggregate gross proceeds from any Shares sold by the designated Manager and to provide the Managers with customary indemnification and contribution rights, including for liabilities under the Securities Act. The Company also will reimburse the Managers for certain specified expenses in connection with entering into the ATM Agreement. As of March 31, 2023, the Company has sold 843,281 shares of its Class A common stock under the ATM Agreement resulting in proceeds to the Company of $980, net of offering costs.
12. STOCK-BASED COMPENSATION AND BENEFIT PLANS
For the years ended December 31, 2021 and 2020, theThe Company incurred stock-based compensation expenses of $313,810533 and $93,026810 for the years ended December 31, 2022 and 2021, respectively.
Stock Incentive Plans
On December 20, 2013, Legacy Pear’s board of directors adopted the 2013 Stock Incentive Plan, or the 2013 Plan, which provided for the grant of stock options, both incentive stock options and nonqualified stock options and restricted stock, to be granted to officers, directors, consultants, and service providers. As last amended and approved by the board of directors on November 3, 2020, the Company was permitted to grant up to 16,727,451 incentive awards under the 2013 Plan.
In connection with the closing of the Business Combination, the Company adopted the 2021 Stock Option and Incentive Award Plan (the “2021 Plan”) a shareholder-approved plan that provides for broad-based equity grants to employees and certain non-employees, including executive officers and permits the granting of restricted stock units (“RSUs”), stock grants, performance based awards, stock options and stock appreciation rights, as well as cash bonus awards.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 153
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Each stock option from the 2013 Plan that was outstanding immediately prior to the Business Combination, whether vested or unvested, were cancelled and exchanged for a stock option to purchase our Class A common stock in the 2021 Plan at a ratio of approximately 1.47. The per share exercise price for each stock option was divided by the ratio of approximately 1.47.
AAs of December 31, 2022, a total of 38,891,801 shares of Class A common stock are reserved under the 2021 Plan, including a total of 32,000,000 shares of our Class A common stock are initially reserved for issuance under the 2021 Plan. The 2021 Plan provides that the number of shares reserved and available for issuance under the 2021 Plan will automatically increase each January 1, beginning on January 1, 2022 and ending in 2031, by 5% of the outstanding number of Class A common stock on the immediately preceding December 31, or such lesser amount as determined by the plan administrator (Board of Directors).the Company’s board of directors or compensation committee). On January 1, 2023 and 2022, 7,004,708 shares and 6,891,801 shares, respectively were added as available for issuance to the 2021 Plan.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 157
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Common Stock Options
All stock-based awards are measured based on the grant date fair value and are generally recognized on a straight-line basis in the Company’s consolidated statement of operations and comprehensive loss over the period during which the employee is required to perform services in exchange for the award, (generally requiring a four-year vesting period). RSUs granted under the 2021 Plan generally vest over three yearsgenerally four-years.
During the years ended December 31, 2022 and 2021, the Company granted stock options to purchase 1,016,918 and 9,541,714 shares of common stock with aggregate grant date fair values of $2,461 and $30,205, respectively, calculated using the Black-Scholes option-pricing model with the following assumptions:
| | | | | | | | | | | | | | | | | |
| | | Year Ended December 31, |
| Stock Options | | | | | 2022 | | 2021 |
Risk-free interest rate | | | | | 1.86% | | 1.01% |
Expected volatility | | | | | 65.69% | | 68.85% |
Expected term (years) | | | | | 5.42-6.57 | | 5.54-6.7 |
Expected dividend yield | | | | | —% | | —% |
| Weighted average fair value at grant date | | | | | $2.42 | | $5.31 |
The risk-free interest rate assumption is based upon observed interest rates appropriate for the term of the related stock options. The expected volatility for the Common Stock was determined based on an average of the historical volatility of a peer group of similar public companies. The expected term of stock options was calculated using the average of the contractual term of the option and the weighted-average vesting period of the option, as the Company does not have sufficient history to use an alternative method to the simplified method to calculate an expected term for employees. The Company has not paid a dividend and is not expected to pay a dividend in the foreseeable future.
The fair value of stock options that vested during the years ended December 31, 2022 and 2021 was $9,258 and $1,852, respectively.
The combined stock option activity for the year ended December 31, 2022, is as follows:
| | | | | | | | | | | | | | | | | | | | | | | |
| Stock Options | | Weighted Average Exercise Price | | Weighted-Average Remaining Contractual Life (years) | | Aggregate Intrinsic Value |
| | | | | | | |
| | | | | | | |
| Outstanding at December 31, 2021 | 19,381,975 | | | $3.02 | | 8.16 | | |
Granted | 1,016,918 | | | $4.14 | | | | |
Exercised | (2,258,138) | | | $0.79 | | | | |
Canceled and forfeited | (3,288,541) | | | $4.97 | | | | |
Outstanding at December 31, 2022 | 14,852,214 | | | $3.01 | | 7.40 | | $ | 1,694 | |
Exercisable at December 31, 2022 | 9,420,481 | | | $2.07 | | 6.72 | | $ | 1,632 | |
As of December 31, 2022, the total unrecognized compensation costs related to non-vested stock options were approximately $14,141 and are expected to be recognized over a weighted average period of 2.48 years.
Restricted Stock Units
RSUs granted under the 2021 Plan generally vest in three equal annual installments over three years, based on continued employment (service period), and are settled upon vesting in shares of the Company’s Class A common stock on a one-for-one basis.
During the years ended December 31, 2021 and 2020, the Company granted stock options, to purchase 9,541,714 and 3,848,401 shares of common stock with aggregate grant date fair values of $30,205 and $2,605, respectively.
Common Stock Options
The combined stock option activity for the year ended December 31, 2021, is as follows:
(1) Number of options and the weighted average exercise price have been adjusted to reflect the exchange of Legacy Pear’s common stock for Class A common stock at an exchange ratio of approximately 1.47 in December 2021 as a result of the Business Combination. See Note 3 for further information.
The weighted-average grant one-for-one basis. The grant-date fair value of stock options granted during the years ended December 31, 2021 and 2020, was $5.31 and $1.00 per share, respectively. The fair value of stock options that vested during the year ended December 31, 2021 was $1,852.
As of December 31, 2021, the total unrecognized compensation costs related to non-vested stock options were approximately $28,894 and are expected to be recognized overRSUs is recognized as expense on a weighted average period of 3.4 years.straight-line basis
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 154158
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Stock Compensation Expense
The assumptions used to estimate the grant date fair value usingover the requisite service period, which is generally the Black-Scholes option pricing model were as follows:
The Company has classified stock-based compensation in its consolidated statements of operations and comprehensive loss as follows:
(1) In connection with the tender offer in 2020, the Company purchased 2,788,732 shares of common stock from the CEO, and 41,100 common shares from certain eligible employees, at a purchase price of $3.9433 per share totaling $11,159 and resulting in a stock-based compensation expense of $7,254, representing the difference between the purchase price and the estimated fair value of the common stock on the date of the sale.
vesting period. The fair value of RSUs is equal to the closing price of its common stock on the date of the grant. RSU activity under the 2021 Plan for the year ended December 31, 2022 is as follows: | | | | | | | | | | | |
| Number of Shares | | Weighted Average Fair Value |
| Outstanding as of December 31, 2021 | — | | — |
| Granted | 8,168,128 | | | $ | 2.98 | |
| Forfeited and canceled | (1,026,583) | | | 3.30 | |
Outstanding as of December 31, 2022 | 7,141,545 | | $ | 2.93 | |
As of December 31, 2022, there was $15,363 of unrecognized compensation cost related to time-based RSUs which is expected to be recognized over a weighted-average period of 2.26 years.
Bonus Program Settled in RSUs
In July 2022, management announced a bonus program where certain employees could receive a range of fixed dollar amounts based on the achievement of certain performance goals, to be settled in RSUs. We awarded 294,010 RSUs, which was determined using the closing price of the Company's Common Stock on November 12, 2022, the date of the Compensation Committee final certification of the Company's performance attainment and the number of awards to be issued to each eligible employee. The awards were classified as liabilities until the number of share awards became fixed once the performance metric was achieved and the grant was approved by the Compensation Committee of the Board of Directors. The awards vest equally over three years as subsequent service is provided to the Company, except for those granted to employees who were included in the reduction in workforce on November 14, 2022 whose terms were modified to allow the first tranche to vest.
Modifications
In conjunction with the reduction in workforce announced on November 14, 2022, the Company modified the terms of certain stock option and RSU awards for approximately 59 impacted employees. These modifications included removal of service conditions for the initial vesting installment of RSU awards, and extended exercise periods for stock options. As a result of these modifications, the Company recorded an $80 reduction in stock based compensation expense for the year ended December 31, 2022.
Employee Stock Purchase Plan
In connection with the closing of the Business Combination, the Company adopted the 2021 Employee Stock Purchase Plan (the “2021 ESPP”). The 2021 ESPP is a shareholder-approved plan under which substantially all employees may voluntarily enroll to purchase the Company’s Class A common stock through payroll deductions at a price equal to 85% of the lower of the fair market values of the stock as of the beginning or the end of six-month offering periods. An employee’s payroll deductions under the 2021 ESPP are limited to 15% of the employee’s compensation and employees may not purchase more than $25,000 of stock during any calendar year.
A total ofThere were 1,800,000 shares of our Class A common stock areinitially reserved and authorized for issuance under the 2021 ESPP. In addition, theThe number of Class A common stock available for issuance under the 2021 ESPP will automatically increasedincrease each January 1 of each calendar year beginning on January 1, 2022, and ending in 2031, by the lesser of 3,600,000 shares of the Company’s Class A common stock, 5% of the outstanding number of shares of Class A common stock on the immediately preceding December 31, or such lesser amount as determined by the plan administrator. As of December 31, 20212022, a total of no5,040,090 shares haveof been issued under the 2021 ESPP.
Class A common stock are available for issuance under the 2021 ESPP. On January 1, 2023, there were 3,600,000 shares added to the 2021 ESPP. We recorded $65 of stock-based Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 155159
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
12.compensation expense related to the 2021 ESPP as of December 31, 2022. There were 359,910 shares issued under the 2021 ESPP for the year ended December 31, 2022.
INCOME TAXES
Stock-Based Compensation ExpenseThe Company has classified stock-based compensation in its consolidated statements of operations and comprehensive loss as follows:
| | | | | | | | | | | | | | | | | |
| | | Year Ended December 31, |
| | | | | 2022 | | 2021 |
Cost of revenue | | | | | $ | 375 | | | $ | 133 | |
Research and development | | | | | 5,079 | | | 1,522 | |
Selling, general, and administrative | | | | | 8,079 | | | 2,155 | |
Total stock-based compensation expense | | | | | $ | 13,533 | | | $ | 3,810 | |
| | | | | | | |
13. INCOME TAXES
New Tax Legislation
The Inflation Reduction Act was signed into law on August 16, 2022, imposing a 15% corporate alternative minimum tax on adjusted financial statement income and a 1% excise tax on stock repurchases after January 1, 2023. The Act did not have an impact on the Company’s 2022 financial statements.
The Tax Cuts and Jobs Act (“TCJA”) requires taxpayers to capitalize and amortize research and experimental (“R&D”) expenditures under section 174 for tax years beginning after December 31, 2021. This rule became effective for the Company during the year ended December 31, 2022, and resulted in the capitalization of R&D costs of $44,500 for tax purposes. The Company is amortizing these costs over 5 years if the R&D was performed in the US and over 15 years if the R&D was performed outside the US.
Tax Rate
A reconciliation of the US federal statutory income rate to the Company’s effective income tax rate is as follows:
| | | | | | | | | | | |
| Years Ended December 31, |
| 2022 | | 2021 |
Federal statutory income tax rate | 21.0 | % | | 21.0 | % |
State income taxes, net of federal benefit | 6.3 | % | | 8.2 | % |
| Change in fair value of earn-out liability | 12.6 | % | | 15.2 | % |
Permanent differences | 1.2 | % | | (2.6) | % |
Federal and state R&D tax credits | 3.7 | % | | 3.0 | % |
Other | (0.3) | % | | (1.0) | % |
Provision to return adjustment | (0.6) | % | | (3.2) | % |
Change in deferred tax asset valuation allowance | (43.9) | % | | (40.6) | % |
Effective income tax rate | — | % | | — | % |
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 160
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
Deferred Tax Assets and Liabilities
The components of the Company’s deferred tax assets and liabilities are as follows:
| | | | | | | | | | | |
| December 31, |
| 2022 | | 2021 |
Deferred tax assets: | | | |
Net operating loss carryforwards | $ | 73,853 | | | $ | 55,930 | |
Deferred revenues | 74 | | | 377 | |
R&D credit carryforwards | 9,820 | | | 7,258 | |
Accrued expenses and other | 3,067 | | | 2,796 | |
Stock-based compensation expense | 2,833 | | | 576 | |
| Lease liabilities | 2,526 | | | — | |
| Capitalized R&D expenses | 9,513 | | | — | |
Depreciation | 48 | | | — | |
| Other | 309 | | | 240 | |
| Total deferred tax assets | 102,043 | | | 67,177 | |
| | | |
| Deferred tax liabilities: | | | |
| Depreciation | — | | | (227) | |
| Amortization | (62) | | | (277) | |
| Lease right-of-use assets | (2,208) | | | — | |
Less: valuation allowance | (99,773) | | | (66,673) | |
Net deferred taxes | $ | — | | | $ | — | |
The Company had no income tax expense due to the operating loss incurred for the years ended December 31, 20212022 and 20202021. The Company’s management has evaluated the positive and negative evidence bearing upon the realizability of its deferred tax assets and has determined that it is more likely than not that the Company will nullnot recognize the benefits of the net deferred tax assets. As a result, the Company recorded a full valuation allowance at December 31, 20212022 and 20202021. The valuation allowance increased by $2633,423100 during the year ended December 31, 20212022, due to the increase in deferred tax assets, primarily due to net operating loss carryforwards, research and developmentR&D tax credits and accruedcapitalized R&D expenses. The valuation allowance increased by $2126,566423 for the year ended December 31, 20202021.
As of December 31, 20212022, the Company has unused federal and state net operating loss carryforwards, or NOLs (“NOLs”), available for carryforward of $218294,908620 and $172223,082584 respectively. The federal and state NOL carryforwards begin to expire after 2034. Approximately $201277,538250 of the federal NOLs have an indefinite carryover period. The Company also had available research and developmentR&D credits for federal and state income tax purposes of $68,229309 and $1,303912, respectively. Utilization of the NOL, research and development credits and other tax attributes may be subject to a substantial annual limitation under Section 382 of the Internal Revenue Code of 1986 due to
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 156
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
ownership change limitations that have occurred previously or could occur in the future. The Company has not yet completed an evaluation of the ownership changes through December 31, 2021R&D credits and other tax attributes may be subject to a substantial annual limitation under Section 382 of the Internal Revenue Code of 1986 due to ownership change limitations that have occurred previously or could occur in the future. The Company has not yet completed an evaluation of the ownership changes through December 31, 2022.
As of December 31, 20212022 and 20202021, the Company had no uncertain tax positions. The Company recognizes both interest and penalties associated with unrecognized tax benefits as a component of income tax expense. The Company has not recorded any interest or penalties for unrecognized tax benefits since its inception.
In the normal course of business, the Company and its subsidiaries may be periodically examined by various taxing authorities. The Company files income tax returns in the US on a federal basis and in certain US states. The associated tax filings remain subject to examination by applicable tax authorities for a certain length of time following the tax year to which those filings relate. With few exceptions, the Company is no longer subject to income tax examinations for years prior to 2017
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 161
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
following the tax year to which those filings relate. With few exceptions, the Company is no longer subject to income tax examinations for years prior to 2019.
13. 14. NET LOSS PER SHARE
For periods in which the Company reports a net loss attributable to common stockholders, potentiallyPotentially dilutive securities have been excluded from the computation of diluted net loss per share as their effects would be anti-dilutive. ThereforeFor periods in which the Company reports a net loss attributable to common stockholders, the weighted average number of common shares outstanding used to calculate both basic and diluted net loss per share attributable to common stockholders is the same. The Company excluded the following potential common shares, presented based on amounts outstanding at period end, from the computation of diluted net loss per share attributable to common stockholders because including them would have had an anti-dilutive effect:
(1) Prior period results have been adjusted to reflect the exchange of Legacy Pear common stock for Class A common stock at an exchange ratio of approximately 1.47 as a result of the Business Combination. See Note 3 for further information.
| | | | | | | | | | | | | | | | |
| | | | Year Ended December 31, |
| | | | 2022 | | 2021 |
Outstanding common stock options | | | | 14,852,214 | | | 19,381,975 | |
| Unvested restricted stock units | | | | 7,141,545 | | | — | |
| | | | | | |
| Private placement warrants to purchase common stock | | | | 5,013,333 | | | 5,013,333 | |
Public warrants to purchase common stock | | | | 9,199,934 | | | 9,199,944 | |
| Earn-Out Shares | | | | 12,395,625 | | | 12,395,625 | |
Total | | | | 48,602,651 | | | 45,990,877 | |
The following table summarizes the computation of basic and diluted net loss per share attributable to common stockholders of the Company for the yearyears ended December 31, 2022 and 2021:
| | | | | | | | | | | | | | | |
| | | Year Ended December 31, |
| | | | | 2022 | | 2021 |
Numerator: | | | | | | | |
| | | | | | | |
| | | | | | | |
| Net loss attributable to common shareholders | | | | | $ | (75,491) | | | $ | (65,142) | |
Denominator: | | | | | | | |
Weighted-average common shares outstanding for basic net loss per share (1) | | | | | 138,707,278 | | | 113,328,450 | |
Basic and diluted net loss per share attributable to common stockholders (1) | | | | | $ | (0.54) | | | $ | (0.57) | |
(1) The weighted-average common shares and thus the net loss per share calculations and potentially dilutive security amounts for all periods prior to the Business Combination have been retrospectively adjusted to the equivalent number of shares outstanding immediately after the Business Combination to effect the reverse recapitalization. Historically reported weighted average shares outstanding have been multiplied by the exchange ratio of approximately 1.47. See Note 3 for further information.
Pear Therapeutics, Inc.
15. RESTRUCTURING CHARGES
On July 25, 2022, the Company restructuring of its operations and executed a reduction in workforce of 25 full-time employees. On November 14, 2022, the Company announced a second reduction in workforce further reducing our headcount by approximately 59 employees.
As a result of these reductions in workforce, the Company recorded the following expenses, primarily related to severance, employee benefits and related costs during the year ended December 31, 2022:
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 157162
Pear Therapeutics, Inc.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(dollars in thousands, except per share amounts)
| | | | | | | | |
| Line Item in Consolidated Results of Operations | | Amount |
Research and development | | $ | 1,821 | |
Selling, general, and administrative | | 1,482 | |
| Total | | $ | 3,303 | |
14. RELATED-PARTY TRANSACTIONS
Certain holders of Legacy Pear Series A preferred stock formerly had representation on the Company’s board of directors and purchased shares of Legacy Pear Series B preferred stock pursuant to the Legacy Pear Series B Agreement. Certain holders of Legacy Pear Series A and B preferred stock formerly had representation on the Company’s board of directors and purchased shares of Legacy Pear Series C preferred stock pursuant to the Legacy Pear Series C Agreement. Certain holders of Legacy Pear Series A, B and C preferred stock formerly had representation on the Company’s board of directors and purchased shares of Legacy Pear Series D preferred stock pursuant to the Legacy Pear Series D Agreement.
In December 2020, in connection with the Legacy Pear D-2 Preferred Stock offering, the Company completed a tender offer. In connection with the tender offer, the Company purchased 2,788,732 shares of Legacy Pear common stock from the CEO, and 41,100 Legacy Pear common shares from certain eligible employees, at a purchase price of $3.9433 per share totaling $11,159 and resulting in a stock-based compensation expense of $7,254, representing the difference between the purchase price and the estimated fair value of the Legacy Pear common stock on the date of the sale. The Company also repurchased 1,656,467 Legacy Pear common shares from certain other shareholders for a total purchase price of $6,532. In total, the Company repurchased 4,486,299 shares of Legacy Pear common stock from investors having a total purchase price of $17,691The following table is a reconciliation of the beginning and ending restructuring liability for the year ended December 31, 2022:
| | | | | |
| Balance as of December 31, 2021 | $ | — | |
| Accrual and accrual adjustments | 3,303 | |
| Cash payments | (2,390) | |
| Balance as of December 31, 2022 | $ | 913 | |
The accrued severance, benefits, and associated costs are reflected in accrued expenses and other current liabilities in the consolidated balance sheet as of December 31, 2022.
15. 16. SUBSEQUENT EVENTS
The Company has completed an evaluation of all subsequentWe have evaluated events and transactions occurring after the audited balance sheet date of December 31, 2021 through the filing date of this Annual Report on Form 10-K with the SEC, to ensure that theseour consolidated financial statements include appropriate disclosure of events both recognized in thewere issued and concluded that there were no events or transactions occurring during this period that required recognition or disclosure in our consolidated financial statements as of December 31, 2021, and events which occurred subsequently but were not recognized in the consolidated financial statements. The Company has concluded that no subsequent events, except for matters described in Note 8, Leases, related to the sublease agreements, Note 7, Indebtedness, related to the alleged certain defaults or events of default that may have occurred that require disclosure, exceptand are continuing under the terms of the Perceptive Credit Facility, and as disclosed within these consolidated financial statements and as described in Note 7.Note 11, Capital Stock, related to the offerings under the ATM Agreement.
In addition, as described in Note 1, Nature of the Business, in February 2023, we initiated a process to explore a range of strategic alternatives to maximize shareholder value and have engaged professional advisors. Management can make no assurances that any particular course of action, business arrangement or transaction, or series of transactions, will be pursued, successfully consummated or lead to increased stockholder value. If the strategic process is unsuccessful, our Board may decide to pursue a liquidation or obtain relief under the US Bankruptcy Code. The Company has hired advisors to explore strategic alternatives including, if needed, filing for bankruptcy protection.
* * * * * *
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 158163
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Background and Remediation of Material Weakness
In connection with our evaluation of disclosure controls and procedures covering our consolidated financial statements as of December 31, 2021, we identified material weaknesses in our internal control over financial reporting. We have concluded that material weaknesses exist in our evaluation of disclosure controls and procedures, including internal control over financial reporting, as we do not have the necessary business processes, personnel and related internal controls to operate in a manner to satisfy the accounting and financial reporting requirements of a public company. These material weaknesses primarily manifested in improper segregation of duties, inadequate design, implementation, and maintenance of adequate information systems controls, including access and change management controls and timely recording of material transactions prior to being a public company.
We are focused on designing and implementing effective internal controls measures to improve our evaluation of disclosure controls and procedures, including internal control over financial reporting, and remediate the material weaknesses. In order to remediate these material weaknesses, we have taken and plan to take the following actions:
▪the hiring and continued hiring of additional accounting staff with public company experience; including a VP, Corporate Controller and Principal Accounting Officer and Director, SEC Reporting and Technical Accounting,
▪implemented a new enterprise resource planning system to replace the prior general ledger package,
▪implementation of the maintenance of adequate information systems controls, including access and change management controls,
▪implementation of additional review controls and processes requiring timely account reconciliation and analyses of certain transactions and accounts,
▪implemented an integrated, collaborative cloud-based audit platform application, and
▪hired a national accounting firm to assist in the design and implementation of controls and remediation of controls gaps.
These actions and planned actions are subject to ongoing evaluation by management and will require testing and validation of design and operating effectiveness of internal controls over financial reporting over future periods. We are committed to the continuous improvement of our internal control over financial reporting and will continue to review the internal controls over financial reporting.
Evaluation of Disclosure Controls and Procedures
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
“Disclosure controls and procedures”, as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), are controls and other procedures that are designed to ensure that information required to be disclosed in our reports filed or submitted under the Securities Exchange Act of 1934, as amended (the "Exchange Act")Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC's rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed in company reports filed or submitted under the Exchange Act is accumulated and communicated to management, including our Chief Executive Officerprincipal executive and Chief Financial Officer, to allow timely decisions regarding required disclosure.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 159
principal financial officers, as appropriate to allow timely decisions regarding required disclosure. As required by Rules 13a-15 and 15d-15 under the Exchange Act, our Chief Executive Officer and Chief Financial Officermanagement, with the participation of our principal executive officer and principal financial officer, carried out an evaluation of the effectiveness of the design and operation of our disclosure controls and procedures as of December 31, 20212022. Based upon their evaluation, our Chief Executive Officerprincipal executive officer and Chief Financial Officerprincipal financial officer concluded that our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) were not effective.
Management’s Annual Report on Internal Control overOver Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Internal control over financial reporting is defined in Rule 13a-15(f) or 15d-15(f) promulgated under the Exchange Act as a process designed by, or under the supervision of, the Company’s principal executive officer and principal financial officer and effected by the Company’s board of directors, management and other personnel to provide reasonable assurance regarding the reliability of our financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America.
This Form 10-K does not include a report of managementManagement conducted the assessment of the effectiveness of the Company’s assessment regarding internal control over financial reporting orbased an attestation report of the company’s registered public accounting firm due to a transition period established by rules of the Securities and Exchange Commission for newly public companies. (Instruction 1 to Item 308, Regulation S-K).
Changes in Internal Controls
None.
Limitations on Effectiveness of Internal Controls
Our management, including our Chief Executive Officer and Chief Financial Officer, does not expect that our disclosure controls and procedures oron criteria established in Internal Control— Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission in 2013. As a result of this assessment, management concluded that, as of December 31, 2022, our internal control over financial reporting will prevent all errors and all fraud. A control system, no matter how well designed and implementedwas ineffective due to the material weaknesses described below. As a result, the disclosure controls and procedures were ineffective in ensuring that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms and is accumulated and communicated to our management, including the chief executive officer and chief financial officer, as appropriate to allow timely decisions regarding disclosure.
Material Weaknesses in Internal Control Over Financial Reporting
As of December 31, 2022, we did not design or maintain an effective control environment as we did not maintain a sufficient complement of accounting and financial reporting resources commensurate with our financial reporting requirements. As a result, we did not design, implement and maintain effective control activities to ensure the accurate and timely reporting of transactions including revenue and capitalized software transactions.
Planned Remediation
We are focused on designing and implementing effective internal controls and measures to improve our evaluation of disclosure controls and procedures, including internal control over financial reporting, and remediate the
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 164
material weaknesses. In order to remediate the material weaknesses, we have taken and plan to take the following actions:
•Engage external accounting advisory consultants to provide additional depth and breadth in our technical accounting and financial reporting capabilities.
•Design and implement effective internal controls which address the review of supporting documentation and accounting conclusions reached for complex transactions.
As management continues to evaluate and work to improve our internal control over financial reporting, management may determine it is necessary to take additional measures to address the material weaknesses. However, we believe the above actions will be effective in remediating the material weaknesses and we will continue to devote significant time and attention to these remediation efforts. Until the controls have been operating for a sufficient period of time and management has concluded, through testing, that these controls are executed consistently and operating effectively, the material weaknesses described above will continue to exist
Changes in Internal Control over Financial Reporting
Except for controls modified in connection with the remediation of material weaknesses identified in 2021, there were no changes in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during the year ended December 31, 2022, that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Limitations of Effectiveness of Controls and Procedures
In designing and evaluating our disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints and the benefits of controls must be considered relative to their costs. Due to the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues, errors and instances of fraud, if any, within the company have been or will be detected.
of achieving their objectives and management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 165
ITEM 9B. OTHER INFORMATION
On March 25, 2022, the Company entered into the Third Amendment to Credit Agreement and Guaranty, dated as of March 25, 2022 (the “Third Amendment”), by and among Pear US certain subsidiaries of Pear US, and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”).
Under the Third Amendment, (i) the minimum revenue covenant was amended to reduce the minimum annual revenue requirements and (ii) the Company became a guarantor of the Pear US’s obligations. In conjunction with the Third Amendment, the parties entered into an Amended and Restated Security Agreement, dated as of March 25, 2022, by and among the Company, the Administrative Agent and the other parties thereto (the “Security Agreement”) and an Amended and Restated Intercompany Subordination Agreement, dated as of March 25, 2022, by and among the Company, the Administrative Agent and the other parties thereto (the “Subordination Agreement”), pursuant to which the Company joined as a party to each such agreement.
The above summary is not complete and is qualified in its entirety to the full text of the Third Amendment, the Security Agreement and the Intercompany Subordination Agreement, which are included as Exhibits 10.34, 10.35, and 10.36 to this Form 10-K and are incorporated herein by reference.None
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 160166
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS, AND CORPORATE GOVERNANCE
Information About Our Directors and Executive Officers
The following biographical descriptions set forth certain information with respect to our directors and our executive officers as of March 1, 2022:
Dr. Corey McCann, M.D., Ph.D., Chief Executive Officer, President and Director
Dr. McCann has served as the President, Chief Executive Officer of Pear and a Director of Pear since December 2021 and prior to this, held the same positions with Pear US since August 2013. He also served as the President of Pear Therapeutics Securities Corporation, a wholly-owned subsidiary of Pear, since December 2017. Dr. McCann has also served as a member of the board of directors of NOUS Imaging, Inc. a technology company focused on medical imaging software, since March 2020, as director of Eternia Group LLC since August 2011, and as a director of Eternia Group SRL since May 2021. Previously, Dr. McCann served on the board of Edumedics LLC (now known as SentryHealth), a health and wellness solutions technology company, from January 2013 through October 2019, and Resolute Bio, Inc., a biotechnology company developing peptide-based drugs for chronic metabolic and neurological disorders from June 2016 through June 2019. Dr. McCann received his B.S. degree in biology from The Pennsylvania State University, his M.D and Ph.D. from Washington University’s St. Louis School of Medicine, and
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 161
his Ph.D. in neuroscience from Harvard University. We believe Dr. McCann is qualified to serve as a director due to his extensive experience leading and developing Pear, as well as his technical knowledge.
Christopher Guiffre, Chief Financial Officer, Chief Operating Officer, Treasurer and Assistant Secretary
Mr. Guiffre has served as the Chief Financial Officer, Chief Operating Officer, Treasurer and Assistant Secretary of Pear since December 2021 and prior to this, held the same positions with Pear US since December 2017. He has also served as the Treasurer of Pear Therapeutics Securities Corporation, a wholly-owned subsidiary of Pear, since December 2018. From December 2017 through December 2021, he served as Interim Chief Compliance Officer at Legacy Pear. From March 2015 through July 2017, Mr. Guiffre served as the President and Chief Executive Officer of Cerulean Pharma, Inc. (now known as Dare Bioscience, Inc.) (NASDAQ: DARE), a biopharmaceutical company. Mr. Guiffre received his B.S. degree in Marketing from Babson College, his J.D. from Boston College Law School, and M.B.A. from the Boston College Carroll School of Management.
Erin K. Brenner, Chief Product Development Officer
Ms. Brenner has served as the Chief Product Development Officer of Pear since December 2021 and prior to this, held the same position with Pear US since June 2020. Ms. Brenner previously served as Legacy Pear’s Vice President of Portfolio Management from January 2019 through June 2020, and as the Senior Director, Portfolio Management from July 2018 through January 2019. Ms. Brenner worked at GE Healthcare Inc. from 2004 to 2018, including leading the development of GE’s Senographe Pristina mammography platform, and from December 2016 through July 2018, she served as the Global Mammography Product Leader for GE Healthcare. She earned her B.A. in History from Middlebury College.
Katherine Jeffery, Chief People Officer
Ms. Jeffery has served as the Chief People Officer of Pear since December 2021 and prior to this, held the same position with Pear US since June 2019. Prior to that, Ms. Jeffery served as the Vice President of People for Recurly, Inc., a subscription management software company, from January 2018 through June 2019; as a Human Resources Consultant and Leadership Coach for Next Level Resource Partners, a logistics management company, from October 2016 through January 2018, and as Vice President of Human Resources for Sportvision Inc., a broadcasting media company, from February 2011 through October 2016. Ms. Jeffery has also served as President of the board of directors of W.O.M.A.N. Inc. since June 2014, as a founding member of the Product Advisory Board of Twine’s CPOHQ since June 2020, and on the Advisory Board of Bennie, an employment benefits platform, since January 2021. Ms. Jeffery earned her B.A. degree in Liberal Arts with a concentration in Human Resources from DePaul University, and an M.S. in Communication with a concentration in Managerial Communication from Northwestern University.
Yuri Maricich, Chief Medical Officer & Head of Development
Dr. Maricich has served as Pear’s Chief Medical Officer and Head of Development since December 2021 and prior to this, held the same positions with Pear US since October 2017. Prior to that, from September 2014 through September 2017, Dr. Maricich served as the Vice President of Clinical Development of Legacy Pear. Dr. Maricich leads the Clinical/Regulatory/Quality and Medical Affairs group at Pear. He leads and manages the development programs from Discovery/TPP stage, through Translational, Clinical Development, Regulatory submission and review as well as Medical Affairs. In addition to overseeing subsequent pipeline programs across a broad-spectrum of disease areas (such as CNS/neuroscience, cardiovascular, and oncology), he led reSET (1st prescription digital therapeutic), reSET-O (1st combination drug and software treatment), and Somryst (chronic insomnia) programs. Dr. Maricich is a licensed, board-certified physician, investor, clinical developer, and strategist. His work is focused on improving patient health and our healthcare system. He has worked with and led successful teams and programs at Health & LifeScience/Biotech (HLS) firms, including Corixa (acquired by GlaxoSmithKline), Xdynia (acquired by Cavion), Cavion (acquired by Jazz Pharmaceuticals), AWS, and Pear Therapeutics (1st FDA-cleared, clinically validated digital therapeutic to treat disease). He founded a digital health firm while a medical student that used natural-language processing (NLP) to structure clinical data. Dr. Maricich is also the principal and owner of Maricich & Co., which he founded in June 2007, and a Partner at Asclepius Capital, an investment firm, since
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 162
January 2012. Dr. Maricich received his B.S. in Pre-Professional/Pre-Med and Philosophy from the University of Notre Dame, his M.D. from the University of Washington and his M.B.A. from Harvard University.
Ronan P. O’Brien, General Counsel, Chief Compliance Officer and Secretary
Mr. O’Brien has served as the General Counsel and Secretary of Pear since December 2021 and prior to this, held the same positions with Pear US since March 2018. Mr. O’Brien has also served as the Chief Compliance Officer of Pear since January 2022. Since December 2018, he has served as the Secretary of Pear Therapeutics Securities Corporation, a wholly-owned subsidiary of Pear. Previously, from September 2016 through February 2018, Mr. O’Brien served as the Vice President and Associate General Counsel for Selecta Biosciences, Inc., a clinical stage biotechnology company (NASDAQ: SELB) and from June 2014 through September 2016, he served as the Executive Director and Associate General Counsel for Cerulean Pharma Inc. (now known as Dare Bioscience, Inc.) (NASDAQ: DARE), a biopharmaceutical company. Mr. O’Brien received his B.A. degree in French Literature and English Literature as well as his J.D. from Boston University.
Julia M. Strandberg, Chief Commercial Officer
Ms. Strandberg has served as Pear’s Chief Commercial Officer since December 2021 and prior to this, held the same position with Pear US since July 2019. Prior to her position at Pear, Ms. Strandberg served as the Senior Vice President of GHX, LLC from May 2019 through July 2019, as President of Edison Consulting LLC from February 2019 through May 2019, and as Vice President and General Manager of Health Informatics and Monitoring at Medtronic plc from January 2015 through January 2019. Ms. Strandberg received her B.S. in Chemical Engineering from Purdue University, and her M.B.A. from the University of Minnesota’s Carlson School of Management.
Alison Bauerlein, Director
Ms. Bauerlein has been a member of the Board since December 2021 and is a member of both the Audit Committee and Compensation Committee. Ms. Bauerlein is a co-founder of Inogen, Inc. (Nasdaq: INGN). Most recently, she served as Inogen’s Chief Financial Officer and Executive Vice President, Finance from March 2014 until December 2021. Ms. Bauerlein also serves on the board of directors of Gelesis Holdings, Inc, (NYSE: GLS), a biotherapeutics company, as well as Koya Medical, Inc, and Equinox Ophthalmic, Inc, which are private companies. Ms. Bauerlein has over 20 years of experience in treasury, finance, accounting, risk management as well as strategic and tactical cost analysis and forecasting. Ms. Bauerlein received a B.A. degree in Economics/Mathematics with high honors from the University of California, Santa Barbara.
Jorge Gomez, Director
Mr. Gomez has been a member of the Board since December 2021 and prior to this, was a member of the board of directors of Pear US since August 2021. Mr. Gomez is the chair of the Audit Committee. Mr. Gomez has been the Executive Vice President and Chief Financial Officer of Dentsply Sirona, Inc. (Nasdaq: XRAY), a manufacturer of professional dental products and technologies, since August 2019. Mr. Gomez previously served as Chief Financial Officer of Cardinal Health, Inc. (NYSE: CAH), a multinational healthcare services company, from January 2018 to August 2019. At Cardinal Health, he was responsible for financial activities across the enterprise, including financial strategy, capital deployment, treasury, investor relations, tax, accounting, and external reporting. From July 2015 to December 2017, he was Chief Financial Officer of Cardinal Health’s Medical segment and from February 2012 to June 2015, he was Chief Financial Officer of Cardinal Health’s Pharmaceutical segment. Prior to that, Mr. Gomez served as treasurer and corporate controller at Cardinal Health. Before joining Cardinal Health, Mr. Gomez held multiple executive and leadership roles at General Motors Company (NYSE: GM), including assignments in New York, Singapore, Belgium, and Brazil. He began his career with the Smurfit-Kappa Group plc. (OTCMKTS: SMFTF) in the company’s finance development program. Currently, he also serves on the Board of Directors for Xylem, Inc. (NYSE: XYL), a global water management company. Mr. Gomez earned a bachelor’s degree in Electrical Engineering from the National University of Colombia and a master’s degree in Business Administration from the University of Hartford.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 163
Zack Lynch, Director
Mr. Lynch has been a member of the Board since December 2021 and prior to this, was a member of the board of directors of Pear US since December 2015. Mr. Lynch serves on the Audit Committee, Compensation Committee and Nominating and Corporate Governance Committee. Since July 2015, Mr. Lynch has served as the Managing Partner of JAZZ Venture Partners, a venture capital firm that focuses on investments at the intersection of digital technology and life sciences. Mr. Lynch also currently serves on the board of various life sciences companies, including Thread Robotics Inc., a medical device and services company focusing on fertility care since June 2020; Swing Therapeutics, Inc., a company focused on creating digital therapeutics for people with chronic conditions since October 2019; and Pinpoint Predictive, Inc., a company that provides behavioral risk assessments using artificial intelligence designed for insurance companies since September 2018. Mr. Lynch has also served on the board of JobGet Inc., an online and mobile application based job platform since January 2021 and served on the board of Pymetrics, Inc., a company focused on utilizing artificial intelligence and behavioral science in hiring practices, from September 2016 to March 2018. Mr. Lynch is the founder and Chairperson of the Neurotechnology Industry Organization, a global trade association representing companies involved in neuroscience, brain research institutes and patient advocacy groups and the founder and curator of the Experiential Technology and NeuroGaming Conference and Expo. Mr. Lynch co-founded NeuroInsights, a market research and investment advisory firm that advises global organizations on the impact of neurotechnology on business, government and society, and developed the NASDAQ NeuroInsights Neurotech Index (Nasdaq: NERV), a stock tracking index for neuroscience companies. Mr. Lynch served on the advisory boards of the McGovern Institute for Brain Research at MIT, Center for Neuroeconomic Studies at Claremont Graduate University and the Center for Neuroscience and Society at the University of Pennsylvania. Mr. Lynch has a B.S. in Evolutionary Biology, B.S. in Environmental Science and an M.A. in Economic Geography, all from UCLA.
Kirthiga Reddy, Director
Ms. Reddy has been a member of the Board since December 2021 and is the chair of the Nominating and Corporate Governance Committee. She previously served as a board observer on the Legacy Pear board of directors beginning December 2020 through November 23, 2021. Ms. Reddy brings over 20 years of experience leading technology-driven transformations and is a co-founder and since October 2018, has served as Investment Counsel for F7 Ventures, a female-led seed investment fund focused on enabling human operations and the investment themes of connected communities, future of work, and physical and mental health. Previously, she served as the Investment Partner at SoftBank Investment Advisers, a private equity firm headquartered in London (SBIA), and served on the Investment Committee for the SoftBank Vision Fund Emerge program, a global accelerator for companies led by underrepresented founders. From July 2010 to March 2018, Ms. Reddy held various executive roles at Facebook, Inc. (Nasdaq: FB). At Facebook, Ms. Reddy first served as the Managing Director for India and South Asia, and subsequently focused on emerging and high-growth markets including Mexico, Brazil, Indonesia, South Africa and the Middle East. Additionally, Ms. Reddy has served as a member of the board of directors of several companies, including Collective Health, Inc. since December 2019, where she also serves on the compensation and audit committees, WeWork Inc. since February 2020, and Fungible, Inc. since April 2021. Ms. Reddy has also served on the Investment Advisory Council for Neythri Futures Fund, a South Asian female-led stage-agnostic tech fund since March 2021. Ms. Reddy holds an MBA from Stanford University, an M.S. in Computer Engineering from Syracuse University and a B.E. in Computer Science from Marathwada University, India. She served on Stanford Business School Management Board from September 2014 to April 2019, including serving as Chair from September 2018 to April 2019.
Nancy Schlichting, Director
Ms. Schlichting has been a member of the Board since December 2021 and prior to this, was a member of the board of directors of Pear US since January 2021. From June 2003 until January 2017, Ms. Schlichting served as the Chief Executive Officer of the Henry Ford Health System, where she helped lead a $4.7 billion integrated healthcare system. Ms. Schlichting has also served on the board of directors of Walgreens Boots Alliance, Inc. (Nasdaq: WBA) since 2006 and chairs its Compensation and Leadership Development Committee. Ms. Schlichting has served on the board of directors of the Duke University Health System since 2016, where she serves as vice-chair, and has also served on the board of directors of Encompass Health since 2018, and Hill-Rom Holdings, Inc. since 2017,
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 164
where she also serves as the Chairperson of the Compensation and Management Development Committee. Ms. Schlichting also serves as the Trustee for a number of organizations, including The Kresge Foundation since 2003, and Duke University since 2018. In addition, Ms. Schlichting has served as a director and Vice Chair of the Detroit Symphony Orchestra since 2016. Ms. Schlichting earned her B.A. from Duke University and her M.B.A. from Cornell University in healthcare administration and accounting.
Andrew J. Schwab, Director
Mr. Schwab has been a member of the Board since December 2021 and prior to this, was a member of the board of directors of Pear US since June 2014. Mr. Schwab has served as the co-Chief Executive Officer and as a member the board of directors of 5:01 Acquisition Corp. (Nasdaq: FVAM) since its inception in August 2020. Mr. Schwab is a Founding Partner and Managing Member of 5AM Venture Management, LLC and also a Managing Member of 5AM Partners IV, LLC and 5AM Opportunities I (GP), LLC, the general partner entities of major shareholders of Pear. Prior to founding 5AM Venture Management, LLC in 2002, Mr. Schwab was a Principal at Bay City Capital where he was involved with companies such as Cubist Pharmaceuticals, Inc., PTC Therapeutics, Inc., Symyx Technologies, Inc. and Syrrx, Inc. Previously, Mr. Schwab was Vice President of Business Development at Digital Gene Technologies, Inc. and a Vice President in the life science investment banking group of Montgomery Securities. At 5AM, he has led the firm’s investments in and served on the boards of Bird Rock Bio, Inc., BlueLight Therapeutics, Inc, Camp4 Therapeutics Corporation, Cleave Therapeutics, Inc., DVS Sciences, Inc. (which was acquired by Fluidigm Corporation), EnLiven Therapeutics, Inc., Escient Pharmaceuticals, Inc., Flexion Therapeutics, Inc. (Nasdaq: FLXN), Ikaria, Inc. (which was acquired by Mallinckrodt plc and spun-out Bellerophon Therapeutics, Inc. (Nasdaq: BLPH)), Ilypsa, Inc. (which was acquired by Amgen, Inc.), Miikana Therapeutics, Inc. (which was acquired by EntreMed, Inc.), Novome Biotechnologies, Inc., Panomics Inc. (which was acquired by Affymetrix, Inc.), Precision NanoSystems, Inc. (which was acquired by Danaher Corporation), Purigen Biosystems, Inc., Synosia Therapeutics Holding AG (which was acquired by Biotie Therapies Corp.), Rarecyte, Inc., The Assay Depot (d.b.a. Scientist.com), TMRW Life Sciences, Inc. and Viveve Medical, Inc. (Nasdaq: VIVE). Mr. Schwab also currently serves on the boards of trustees of the California Academy of Sciences and Davidson College. Mr. Schwab earned a B.S. degree with Honors in Genetics & Ethics from Davidson College.
Code of Business Conduct and Ethics; Corporate Governance Guidelines
We have adopted a written Code of Business Conduct and Ethics that applies to all of our employees, officers and directors, including our principal executive officer, principal financial officer, principal accounting officer, or persons performing similar functions, as well as corporate Governance Guidelines. Copies of the Code of Ethics and Corporate Governance Guidelines can be found on the Investors (Investors → Corporate Governance → Governance Overview) section of our website at www.peartherapeutics.com. A copy of the Code of Business Conduct and Ethics may also be obtained, free of charge, upon a request directed to: Pear Therapeutics, Inc., 200 State Street, 13th Floor, Boston, Massachusetts 02109, Attention: General Counsel. We intend to disclose any amendments to the Code of Business Conduct and Ethics, or any waivers of its requirements, on our website to the extent required by the applicable rules and exchange requirements. The information on any of our websites is deemed not to be incorporated in this Form 10-K or to be part of this Form 10-K.
Director Nominations
The Board consists of seven members and is divided into three classes. At each annual general meeting of stockholders, the successors to directors whose terms then expire will be elected to serve from the time of election and qualification until the third annual meeting following the election. Stockholders may also recommend candidates for election to the Board, as described below. The Board screens potential director candidates and considers criteria including experience, qualifications, attributes, skills, diversity and other characteristics in the context of the current make-up of the Board and the needs of the Board given the circumstances of the Company.
The Board values the input of stockholders in identifying director candidates. Accordingly, the Board considers recommendations for director candidates submitted by stockholders using substantially the same criteria it applies to recommendations from directors and members of management. Any such nominations should be submitted to the Board by mail in care of the Company’s Secretary, at 200 State Street, 13th Floor, Boston, Massachusetts
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 165
02109 and be accompanied by the information required by the Bylaws. The written recommendation should be submitted withinThe information required by this item is incorporated by reference to our Definitive Proxy Statement for our 2023 Annual Meeting of Stockholders which will be filed with the time frame described in the Bylaws.
Audit Committee and Audit Committee Financial Expert
We have a separately-designated audit committee established in accordance with Section 3(a)(58)(A) of the Exchange Act, which currently consists of Mr. Gomez, Ms. Bauerlein, and Mr. Lynch. Each member of the Audit Committee is independent under the applicable Nasdaq listing rules and Rule 10A-3(b)(1) of the Exchange Act. Mr. Gomez serves as the chair of the Audit Committee. The Board has determined that Mr. Gomez and Ms. Bauerlein are each an “audit committee financial expert” within the meaning of SEC regulations. The Board has also determined that each member of the proposed Audit Committee has the requisite financial expertise required under the applicable requirements of Nasdaq.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 166
ITEM 11. EXECUTIVE COMPENSATION
Executive Summary
2021 Highlights
On December 3, 2021, the Business Combination closed and we transitioned into becoming a public company. In connection with the Business Combination, we engaged Frederic W. Cook & Co., Inc. as our independent compensation consultant to help structure a competitive executive compensation program from a public company perspective.
As a general matter, our executive compensation program consists of annual base salary, target cash bonus opportunities, and equity-based long-term incentives. We will continue to evaluate, develop, and grow our executive compensation program as we progress as a public company.
Executive Compensation Objectives
The Company is committed to providing a fair and market competitive executive compensation programs that will attract, retain and reward high-performing employees. Our compensation package is tied to the contributions of the individual and the achievement of organizational goals. Below are the objectives of our program:
•Attract, retain, and motivate superior executive talent;
•Provide incentives that reward the achievement of performance goals that directly correlate to the enhancement of shareholder value, as well as facilitate executive retention; and
•Reinforce Pear’s goal of recruiting and retaining a highly motivated workforce to support the overall growth and performance of the Company.
Compensation Governance and Best Practices
We are committed to having strong governance standards with respect to our compensation programs, procedures and practices. Our key compensation practices include the following:
| | | | | | | | |
What We Do | | What We Do Not Do |
•Emphasize the use of equity compensation to promote executive retention and reward long-term value creation •Maintain equity granting guidelines •Engage an independent compensation consultant to advise our Compensation Committee •Develop a relevant peer group to benchmark compensation | | •Do not guarantee annual salary increases •Do not grant uncapped cash incentives or guaranteed equity compensation •Do not provide significant perquisites. •Do not provide any compensation-related tax gross-ups |
Summary Compensation Table
The following table sets forth information regarding compensation earned by our President and Chief Executive Officer and our two next most highly paid executive officers who served during fiscal year 2021, as well as the compensation earned by Elon S. Boms, who served as the Chief Executive Officer of THMA until his resignation upon the closing of the Business Combination. We refer to these individuals as our named executive officers, or
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 167
“NEOs”. The compensation set forth below for Dr. McCann, Mr. Guiffre, and Ms. Strandberg includes compensation from Legacy Pear during fiscal years 2021 and 2020.
_________________
(1)The amounts reported in the “Option Awards” column reflect the aggregate grant date fair value of stock options awarded during fiscal years 2020 and 2021 computed in accordance with the provisions of Financial Accounting Standards Board Accounting Standards Codification Topic 718, or ASC 718. See Note 11 in the accompanying notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for assumptions underlying the valuation of equity awards.
(2)The amounts shown in the “Non-Equity Incentive Plan Compensation” column represent amounts earned in fiscal years 2020 and 2021 but paid in February of the following year based on the achievement of corporate and individual performance objectives set by the Board. In addition, “Non-Equity Incentive Plan Compensation” for Ms. Strandberg includes $16,798 and $66,462 in commissions earned by Ms. Strandberg for fiscal years 2020 and 2021, respectively, as a member of our commercial team.
(3)“All Other Compensation” includes:
(i)for Dr. McCann, (a) $64,935 and $49,950 during 2020 and 2021, respectively, representing payment for a lease to occupy an apartment in San Francisco, California and (b) $9,975 during 2020 representing employer matching contributions under our 401(k) plan;
(ii)for Mr. Guiffre, (a) $8,345 during 2020 representing employer matching contributions under our 401(k) plan and (b) $6,420 and $5,885 during 2020 and 2021, respectively, representing a transportation benefit; and
(iii)for Ms. Strandberg, (a) $27,000 representing payment for a lease to occupy an apartment in Boston, Massachusetts, which payments were made through March 2020, and (b) $6,550 representing employer matching contributions under our 401(k) plan.
(4)Mr. Boms was the Chief Executive Officer of THMA until December 2021, at which time he resigned from his position upon the closing of the Business Combination.
Narrative Disclosure to Summary Compensation Table
Base Salaries
We use base salaries to recognize the experience, skills, knowledge and responsibilities required of all our employees, including our NEOs. Base salaries are reviewed annually, typically in connection with our annual performance review process, and adjusted from time to time to realign salaries with market levels after taking into account individual responsibilities, performance and experience. For the year ended December 31, 2021, the annual base salaries for each of Dr. McCann, Mr. Guiffre and Ms. Strandberg were $450,000, $396,229 and $366,062, respectively. The compensation committee sets the salary and bonus for each NEO, aside from the President & Chief Executive Officer’s which is set by the Board based on recommendation from the Compensation Committee, and the compensation is subject to periodic review and adjustment. Dr. McCann’s base salary for 2022 is $550,000, Mr. Guiffre’s base salary for 2022 is $435,000 and Ms. Strandberg’s base salary for 2022 is $415,000.
Equity Compensation
We believe that equity award grants provide our executives with a strong link to our long-term performance, create an ownership culture and help to align the interests of our executives and our stockholders. In addition, we believe that equity award grants with a time-based vesting feature promote executive retention because this feature incentivizes our executive officers to remain in our employment during the vesting period. Accordingly, we typically grant equity awards to our executives, including our NEOs, in the ordinary course of business on a discretionary basis. During the year ended December 31, 2021, we granted options to purchase shares of common
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 168
stock of Legacy Pear to Mr. Guiffre and Ms. Strandberg, as described in more detail in the “Outstanding Equity Awards at 2021 Fiscal Year End” table.
Non-Equity Incentive Plan Compensation
We pay cash incentive compensation to reward our executives for their performance over the fiscal year, based on performance goals established by the Board. Incentive compensation is determined by the Board based on two components: corporate performance and individual performance. Dr. McCann’s bonus is based on 100% corporate performance and 0% individual performance, and Mr. Guiffre’s and Ms. Strandberg’s bonuses are based on 80% corporate performance and 20% individual performance. For the year ended December 31, 2021, the target bonus for Dr. McCann was equal to 50% percent of his base salary, the target bonus for Mr. Guiffre for the year ended December 31, 2021 was 45% percent of his base salary, and the target bonus for Ms. Strandberg for the year ended December 31, 2021 was 45% percent of her base salary. In addition to the incentive compensation described above, Ms. Strandberg earned $62,461 in commissions for the year ended December 31, 2021 as a member of the commercial team.
Each NEO is eligible to participate in any annual incentive compensation programs as may be established from time to time by the Board. For 2022, Dr. McCann is eligible to receive a target annual performance bonus of 60% of his annual base salary, Mr. Guiffre is eligible to receive a target annual performance bonus of 50% of his annual base salary, and Ms. Strandberg is eligible to receive a target annual performance bonus of 50% of her annual base salary.
Outstanding Equity Awards at 2021 Fiscal Year End
The following table sets forth information regarding outstanding equity awards held by each of our NEOs as of December 31, 2021. All equity awards set forth in the table below were originally granted under the Legacy Pear 2013 Stock Incentive Plan (the “2013 Plan”). In connection with the Business Combination, each option to purchase shares of common stock of Legacy Pear outstanding under the 2013 Plan were cancelled in exchange for an option to purchase Class A common stock under the Pear Therapeutics, Inc. 2021 Stock Option and Incentive Plan. Mr. Boms, who served as the Chief Executive Officer of THMA until his resignation upon the closing of the Business Combination, held no outstanding equity-based awards as of December 31, 2021.
__________________
(1)The shares underlying these stock options vest over four years with 25% of the shares vesting on the first anniversary of the vesting commencement date, and the remaining shares vesting in 36 equal monthly installments on the last day of each calendar month following the first anniversary (for the avoidance of doubt, inclusive of the calendar month in which the first anniversary falls), subject to the executive’s continued service. The vesting commencement date is January 26, 2021.
(2)The shares underlying these stock options vest over four years with 25% of the shares vesting on the first anniversary of the vesting commencement date, and the remaining shares vesting in 36 equal monthly installments on the last day of each calendar month following the first anniversary (for the avoidance of doubt, inclusive of the calendar month in which the first anniversary falls), subject to the executive’s continued service. The vesting commencement date is March 24, 2020.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 169
(3)The shares underlying these stock options vest over four years with 25% of the shares vesting on the first anniversary of the vesting commencement date, and the remaining shares vesting in 36 equal monthly installments on the last day of each calendar month following the first anniversary (for the avoidance of doubt, inclusive of the calendar month in which the first anniversary falls), subject to the executive’s continued service. The vesting commencement date is March 14, 2019.
(4)The shares underlying these stock options vest over four years with 25% of the shares vesting on the first anniversary of the vesting commencement date, and the remaining shares vesting in 36 equal monthly installments thereafter, subject to the executive’s continued service. The vesting commencement date is December 7, 2017.
(5)The shares underlying these stock options vest over four years with 25% of the shares vesting on the first anniversary of the vesting commencement date, and the remaining shares vesting in 36 equal monthly installments on the last day of each calendar month following the first anniversary (for the avoidance of doubt, inclusive of the calendar month in which the first anniversary falls), subject to the executive’s continued service. The vesting commencement date is July 22, 2019.
Potential Payments Upon Termination or Change of Control
The employment of each of our NEO’s is at-will. Each NEO has entered into a management retention agreement which provides that if the NEO is terminated “for cause” or resigns without “good reason” (as such terms are defined in the management retention agreement), the NEO is paid their accrued but unpaid vacation pay, earned but unpaid salary, and reimbursement for any business expense (collectively, the “accrued obligations”). If the NEO is terminated within 60 days before a “change in control date” (as such term is defined in the management retention agreement) or during the one year following the change in control (such period is defined in the management retention agreement as the “protection period”) and the NEO is terminated by us without cause or by the NEO for good reason, the NEO will receive payments that equal the accrued obligations, 12 months of the NEO’s base salary as of the termination date (paid in a lump sum within two weeks of the termination date), continued medical, life insurance and benefits to the same extent the NEO participated prior to the termination date for 12 months following the termination date (unless similar benefits are offered sooner by a new employer). In addition, any “equity awards” (as such term is defined in the management retention agreement) that have not been terminated will automatically accelerate and become fully exercisable and all restrictions and conditions on the NEO’s outstanding stock awards and other equity based awards will immediately lapse as of the termination date, and if an equity award has been terminated, the NEO will receive a cash amount equal to the amount he or she would have received had all such terminated equity awards been fully vested. Finally, if the NEO is terminated without cause or resigns with good reason outside of the protection period, the NEO will receive the accrued obligations, six months of the NEO’s base salary as of the termination date (paid in a lump sum within two weeks of the termination date) and medical, life insurance, disability and other benefits to the same extent the NEO participated prior to the termination date for six months following the termination date (unless similar benefits are offered sooner by a new employer).
Director Compensation
The following table presents information regarding compensation awarded to, earned by or paid to our non-employee directors in connection with their service on the Board during fiscal year 2021 (including compensation for serving on the board of directors of THMA and Legacy Pear. Other than as set forth in the table and described more fully below, we did not pay any compensation, make any equity awards to, or pay any other compensation to any of the non-employee members of the Board. Prior to the Business Combination, THMA did not pay compensation to the members of the board of directors for their service on the board of directors of THMA. Corey McCann, our President and Chief Executive Officer, did not receive any compensation for his service as a member of the Board during 2021 or 2020. Dr. McCann’s compensation for service as an employee for fiscal years 2021 and 2020 are presented above in Item 11, “Executive Compensation.”
Director Compensation Table—2021
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 170
__________________
(1)The amounts reported in the “Option awards” column reflect the aggregate grant date fair value of share-based compensation awarded during the year computed in accordance with the provisions of Financial Accounting Standards Board Accounting Standards Codification, or ASC, Topic 718. See Note 11 in the notes to the consolidated financial statements included in Part II, Item 8 of this Form 10-K for assumptions underlying the valuation of equity awards.
The table below shows the aggregate number of Class A common stock option awards held as of December 31, 2021 by each of our current non-employee directors who was serving as of that date.
Non-Employee Director Compensation Policy
Non-employee members of the Board shall be eligible to receive cash and equity compensation, as set forth in the Non-Employee Director Compensation Policy, as described below:
In addition to cash compensation, non-employee directors elected or appointed to our board of directors will be granted a one-time stock option award to purchase 70,560 shares of our Class A common stock on the date of such director’s election or appointment to the board of directors, which will vest in equal annual installments over three years, subject to continued service through such vesting dates. On the day of our annual meeting of stockholders each year, we may grant each non-employee director an annual stock option or a restricted stock award.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
Securities and Exchange Commission no later than 120 days after the end of our fiscal year (the “Proxy Statement”).
ITEM 11. EXECUTIVE COMPENSATION
The information required by this item is incorporated by reference to our Proxy Statement.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
Securities Authorized for Issuance Under Equity Compensation Plans
We have two equity compensation plans under which awards are currently authorized for issuance, the Pear Therapeutics, Inc. 2021 Stock Option and Incentive Plan (the “2021 Plan”) and the Pear Therapeutics, Inc. 2021 Employee Stock Purchase Plan (the “2021 ESPP”). In connection with the consummation of the Business Combination, each option that was unexercised and outstanding under the Legacy Pear Therapeutics, Inc. 2013 Stock Incentive Plan as of immediately prior to the effective time of the Business Combination was cancelled in exchange for an option to purchase Class A common stock under the 2021 Plan. Both the 2021 Plan and 2021 ESPP were approved by our stockholders. The following table provides information regarding securities authorized for issuance as of December 31, 2021 under our equity compensation plans.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 171
(1) Consists of Class A common stock issuable upon exercise of outstanding options issued under the 2021 Plan.
(2) Consists of 12,222,812 Class A common stock reserved for future issuance under the 2021 Plan and 1,800,000 shares of Class A common stock reserved for future issuance under the 2021 ESPP. The 2021 Plan provides that the number of shares reserved and available for issuance under the 2021 Plan will automatically increase each January 1, beginning on January 1, 2022 and ending in 2031, by 5% of the outstanding number of Class A common stock on the immediately preceding December 31, or such lesser amount as determined by the plan administrator; on January 1, 2022, 6,891,801 shares of Class A common stock were added to the 2021 Plan pursuant to the evergreen formula. The 2021 ESPP provides that the number of Class A common stock are reserved and available for issuance under the plan will automatically increase each January 1, beginning on January 1, 2022, by the lesser of 3,600,000 shares of Class A common stock, 5% of the outstanding number of Class A common stock on the immediately preceding December 31, or such lesser amount as determined by the Compensation Committee, as plan administrator; on January 1, 2022, 3,600,000 shares of Class A common stock were added to the 2021 ESPP pursuant to the evergreen formula.
Certain Beneficial Owners and Management
The following table sets forth information regarding the beneficial ownership of our Class A common stock as of March 1, 2022:
•each person known by Pear to be the beneficial owner of more than 5% of the Class A common stock;
•each of Pear’s NEOs and directors; and
•all of Pear’s executive officers and directors as a group.
Beneficial ownership is determined according to the rules of the SEC, which generally provide that a person has beneficial ownership of a security if he, she or it possesses sole or shared voting or investment power over that security, including options and restricted stock units that are currently exercisable or vested or that will become exercisable or vest within 60 days. Unless otherwise indicated, Pear believes that all persons named in the table below have sole voting and investment power with respect to the voting securities beneficially owned by them.
The following beneficial ownership of our Class A common stock is based on 137,836,028 shares of Class A common stock issued and outstanding as of the March 1, 2022:
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 172
__________________
*Indicates beneficial ownership less than 1%.
(1)Unless otherwise noted, the business address of each of the directors and executive officers is c/o Pear Therapeutics, Inc., 200 State Street, 13th Floor, Boston, Massachusetts 02109.
(2)Consists of (i) 8,157,565 shares of Class A common stock held directly by Corey McCann; (ii) 1,685,913 shares of Class A common stock held by The Corey M. McCann Irrevocable Trust of 2021 (the “McCann Irrevocable Trust”), and (iii) 986,517 shares of Class A common stock held by The McCann Family Trust (the “McCann Family Trust,” and, together with the McCann Irrevocable Trust, the “McCann Trusts”). Mia E. Moll, the Trustee of the McCann Irrevocable Trust, is the sister of Corey McCann and holds voting and investment control over the shares held by the McCann Irrevocable Trust. Mia E. Moll acts as Investment Advisor and Concord Trust Company, LLC acts as Trustee of the McCann Family Trust; the Trustee acts at the direction of a distribution committee, whose actions over the shares of Class A common stock held by the McCann Family Trust are subject to the approval of Corey McCann pursuant to the trust agreement. By virtue of the foregoing, Corey McCann may be deemed to be the beneficial owner of the shares of Class A common stock held by each of the McCann Trusts. Dr. McCann disclaims beneficial ownership of the shares of Class A common stock held by each of the McCann Trusts, except to the extent of any pecuniary interest therein. The business address of the McCann Irrevocable Trust and Mia E. Moll is c/o Bass, Doherty & Finks, P.C., 1380 Soldiers Field Road, Boston, MA 02135-1023, and the business address of the McCann Family Trust and Cancord Trust Company, LLC is 3 Executive Park Drive, Suite 302, Bedford, NH 03110.
(3)Consists of options to purchase shares of Class A common stock which have vested or which will vest within 60 days of the March 1, 2022.
(4)Mr. Boms was the Chief Executive Officer of THMA until December 2021, at which time he resigned from his position upon the closing of the Business Combination.
(5)Consists of 10,766,587 shares of Class A common stock identified in footnote 12 over which Mr. Lynch may be deemed to beneficially own and options to purchase up to 29,448 shares of Class A common stock which have vested or which will vest within 60 days of the March 1, 2022. Mr. Lynch disclaims beneficial ownership of the shares identified in footnote 12 except to the extent of his pecuniary interest therein.
(6)Consists of 18,631,771 shares of Class A common stock identified in footnote 8 over which Mr. Schwab may be deemed to beneficially own. Mr. Schwab disclaims beneficial ownership of the shares identified in footnote 8 except to the extent of his pecuniary interest therein.
(7)Consists of 24,903,573 shares of Class A common stock held by TLS Beta Pte. Ltd. and 1,900,000 shares of Class A common stock held by Elbrus Investments Pte. Ltd. Each of TLS Beta Pte. Ltd. and Elbrus Investments Pte. Ltd. is a direct wholly-owned subsidiary of Temasek Life Sciences Private Limited, which in turn is a direct wholly-owned subsidiary of Fullerton Management Pte Ltd, which in turn is a direct wholly-owned subsidiary of Temasek Holdings (Private) Limited. The address of each of the foregoing entities is 60B Orchard Road, #06-18 Tower 2, The Atrium@Orchard, Singapore.
(8)Consists of 14,648,641shares of Class A common stock held by 5AM Ventures IV, L.P. (“Ventures IV”), 610,361 shares of Class A common stock held by 5AM Co-Investors IV, L.P. (“Co-Investors IV”), and 3,372,769 shares of Class A common stock held by 5AM Opportunities I, L.P. (“Opportunities I”). 5AM Partners IV, LLC (“Partners IV”) is the sole general partner of Ventures IV and Co-Investors IV. Dr. John Diekman, Andrew Schwab and Dr. Scott M. Rocklage, are the managing members of Partners IV and, along with Partners IV, have shared voting and investment power over the shares beneficially owned by Ventures IV and Co-Investors IV. Andrew Schwab, one of our directors, is an affiliate of Ventures IV, Co-Investors IV and Opportunities I. Each of Partners IV, Dr. Diekman, Mr. Schwab and Dr. Rocklage disclaim beneficial ownership of such shares except to the extent of its or their recurring interest therein. 5AM Opportunities I (GP), LLC is the general partner of Opportunities I and may be deemed to have sole investment and voting power over the shares held by Opportunities I. Andrew Schwab and Dr. Kush Parmar are the managing members of 5AM Opportunities I (GP), LLC, and may be deemed to share voting and dispositive power over the shares held by Opportunities I. The address of all entities affiliated with 5AM Ventures is 501 2nd Street, Suite 350, San Francisco, CA 94107.
(9)Consists of 12,100,078 shares of Class A common stock held by SVF II Cobbler (DE) LLC (“SVF”). SoftBank Vision Fund II-2 L.P. is the managing member of SVF II AIV (DE) LLC, which is the sole member of SVF. SB Global Advisers Limited (“SBGA”) has been appointed as manager and is responsible for making all decisions related to the acquisition, structuring, financing and disposal of SoftBank Vision Fund II-2 L.P.’s investments, including as held by SVF. Spencer Collins, Rajeev Misra, and Neil Hadley are the directors of SBGA. As a result of these relationships, each of these entities and individuals may be deemed to share beneficial ownership of the securities held of record by SVF. Each of them disclaims any such beneficial ownership. The registered address for each of SVF and SVF II Holdings (DE) LLC is c/o Corporation Service Company, 251 Little Falls Drive, Wilmington, DE 19808. The registered address of SoftBank Vision Fund II-2 L.P. is c/o
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 173
Crestbridge Limited, 47 Esplanade, St. Helier, Jersey, JE1 0BD. The business address of SB Global Advisers Limited is 69 Grosvenor Street, London W1K 3JP, England, United Kingdom.
(10)Consists of 6,540,000 shares of Class A common stock and 5,013,333 Private Placement Warrants held by LJ10 LLC, which are exercisable for 5,013,333 shares of Class A common stock within 60 days of March 1, 2022. Elon S. Boms and two other managers are the three managers of LJ10 LLC’s board of managers. Any action by LJ10 LLC with respect to the shares, including voting and dispositive decisions, requires at least a majority vote of the managers of the board of managers. Under the so-called “rule of three”, because voting and dispositive decisions are made by a majority of the managers, none of the managers is deemed to be a beneficial owner of securities held by LJ10 LLC, even those in which such manager may hold a pecuniary interest. Accordingly, none of the managers on LJ10 LLC’s board of managers is deemed to have or share beneficial ownership of the shares held by LJ10 LLC. LJ10 LLC and KLP SPAC 1 LLC may be deemed to be members of a “group,” within the meaning of Section 13(d)(3) of the Act, comprised of LJ10 LLC and KLP SPAC 1 LLC. The business address of LJ10 LLC is 195 Church Street, 15th Floor, New Haven, Connecticut 06510.
(11)Consists of 10,859,050 shares of Class A common stock held by Arboretum Ventures IV, L.P. (“AV IV”). Arboretum Investment Manager IV, LLC (“AIM IV”) is the general partner of AV IV. Jan L. Garfinkle, Timothy B. Petersen and Paul McCreadie are the managing members of AIM IV and share voting and dispositive power with respect to the shares held by AV IV. Ms. Garfinkle and Messrs. Petersen and McCreadie disclaim beneficial ownership of the shares held by AV IV, except to the extent of their pecuniary interest therein. The address of the principal place of business of each of these entities and individuals is 303 Detroit Street, Suite 301, Ann Arbor, Michigan 48104.
(12)Consists of 9,681,673 shares of Class A common stock held by JAZZ Human Performance Technology Fund, L.P. (“Jazz Technology”) and 1,084,914 shares of Class A common stock held by JAZZ Human Performance Opportunity Fund, L.P (“Jazz Opportunity”). Jazz Human Performance Technology GP, LLC (“Jazz Technology GP”) is the general partner of Jazz Technology. Voting and dispositive decisions with respect to the shares held by Jazz Technology are exercised collectively by the managing members of Jazz Technology GP: Andrew Firlik, John Harris, Zack Lynch and John Spinale. JAZZ Human Performance Opportunity GP, LLC (“Jazz Opportunity GP”) is the general partner of Jazz Opportunity. Voting and dispositive decisions with respect to the shares held by Jazz Opportunity are exercised collectively by the managing members of Jazz Opportunity GP: Andrew Firlik, John Harris, Zack Lynch and John Spinale. Mr. Lynch, one of our directors, is an affiliate of Jazz Technology and Jazz Opportunity. The address of the principal place of business of each of these entities and individuals is 548 Market Street, #27799, San Francisco, CA 94104.
(13)Consists of 5,502,171 shares of Class A common stock held by Kaiser Permanente Group Trust (“Kaiser Trust”) and 3,001,447 shares of Class A common stock held by Kaiser Foundation Hospitals (“Kaiser Hospitals”). The Kaiser Permanente Retirement Plans Investment Committee has discretionary authority to manage and control Kaiser Trust assets. The address for Kaiser Trust and Kaiser Hospitals is One Kaiser Plaza, The Ordway Building, Oakland, California 94612.
(14)Excludes the contingent right to receive additional Class A common stock upon the achievement of certain earn-out targets.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 174
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The information required by this item is incorporated by reference to our Proxy Statement.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
Policies and Procedures for Approval of Related Person Transactions
We have adopted a formal written policy that became effective upon the completion of the Business Combination which provides that our officers, directors, nominees for election as directors, beneficial owners of more than 5% of any class of our common stock or preferred stock, any member of the immediate family of any of the foregoing persons and any firm, corporation or other entity in which any of the foregoing persons is employed or is a general partner or principal or in a similar position or in which such person has a 5% or greater beneficial ownership interest, where the amount involved in the transaction exceeds or is reasonably expected to exceed $120,000 in a single fiscal year and the related party has or will have a direct or indirect interest in the transaction (other than solely as a result of being a director or less than 10% beneficial owner of another entity), are not permitted to enter into a related party transaction with us without the approval of our Audit Committee, subject to certain exceptions. This written policy on transactions with related persons is in conformity with the requirements for issuers having publicly held common stock that is listed on Nasdaq.
The Audit Committee is responsible for reviewing and approving any related person transactions. In reviewing any related person transaction, the Audit Committee will take into account, among other factors that it deems appropriate, whether the related person transaction is on terms no less favorable to Pear than terms generally available in a transaction with an unaffiliated third party under the same or similar circumstances and the extent of the related person’s interest in the related person transaction.
Relationships and Transactions with Directors, Executive Officers, and Significant Stockholders
In addition to the executive officer and director compensation arrangements discussed above under “Executive Compensation”, since January 1, 2020, the following is a description of each transaction since January 1, 2020 and each currently proposed transaction in which:
•we, THMA or Legacy Pear have been or are to be a participant;
•the amounts involved exceeded or exceeds $120,000; and
•any of our directors, executive officers or holders of more than 5% of our outstanding capital stock, or any immediate family member of, or person sharing the household with, any of these individuals or entities, had or will have a direct or indirect material interest.
Certain Relationships and Related Person Transactions of Pear (formerly THMA)
SoftBank Agreement
Effective March 15, 2022, we entered into a development agreement with SoftBank Corp., an entity under common control with SVF II Cobbler (DE) LLC (a greater than 5% shareholder of Pear), to develop a Japanese-language digital therapeutic for the treatment of sleep/wake disorders for the Japanese market.
Founder Shares and Private Placement Warrants
On December 7, 2020, the Sponsor purchased 5,750,000 shares of Class B common stock, par value $0.0001 per share, of THMA (“THMA Class B Common Shares”) (“Founder Shares”) for an aggregate purchase price of $25,000, or approximately $0.004 per share. In January 2021, the Sponsor transferred 50,000 Founder Shares to each of Michael J. Christenson, Meghan M. FitzGerald, and Henry S. Miller, THMA’s independent directors, and an aggregate of 150,000 Founder Shares to THMA’s Advisors, in each case, at approximately the same share price initially paid by the Sponsor. On February 2, 2021, THMA effected a 1.2-to-1 forward stock split with respect to its Founder Shares, resulting in the Sponsor holding 6,900,000 Founder Shares, each THMA independent director holding 60,000 Founder Shares and THMA’s Advisors holding an aggregate of 180,000 Founder Shares. Simultaneously with the consummation of its initial public offering on February 4, 2021, THMA consummated the private placement of an aggregate of 5,013,333 Private Placement Warrants to the Sponsor at a price of $1.50 per Private Placement Warrant, generating total proceeds of $7,520,000.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 175
Subscription Agreements
In connection with the execution of the Business Combination Agreement, on June 21, 2021, THMA entered into separate PIPE Subscription Agreements whereby the Pipe Investors agreed to purchase and THMA agreed to sell, an aggregate of 9,780,000 PIPE Shares, for a purchase price of $10.00 per share and an aggregate purchase price of $97.8 million. Pursuant to the PIPE Subscription Agreements, Pear gave certain registration rights to the PIPE Investors with respect to the PIPE Shares. The sale of the PIPE Shares was consummated concurrently with the closing of the Business Combination.
Sponsor Support Agreement
In connection with the execution of the Business Combination Agreement, the Sponsor and THMA’s directors, advisors and officers (collectively, the “Sponsor Agreement Parties”) entered into the Sponsor Support Agreement with THMA and Pear US, pursuant to which each Sponsor Support Agreement Party agreed to, among other things, (i) vote at any meeting of the stockholders of THMA all of its THMA Class A Common Shares and THMA Class B Common Shares in favor of the transactions and the adoption of the Business Combination Agreement; (ii) appoint THMA as each Sponsor Agreement Party’s proxy in the event such Sponsor Agreement Party fails to fulfill its obligations under the Sponsor Agreement; (iii) be bound by certain other covenants and agreements related to the Merger; (iv) subject certain of the Sponsor’s THMA Class B Shares (the “Earn-Out Shares”) and Sponsor’s Private Placement Warrants (the “Earn-Out Warrants”) to post-closing vesting conditions and (v) be bound by certain transfer restrictions with respect to THMA Common Shares during the period between the date of the Sponsor Support Agreement and the Closing, subject to certain exceptions set forth in the Sponsor Agreement. The Sponsor Agreement also provided that the Sponsor Agreement Parties agreed to waive their redemption rights in connection with the consummation of the Business Combination with respect to any THMA Common Shares held by them.
Pursuant to the Sponsor Agreement, the Sponsor also agreed, subject to certain exceptions, not to transfer 1,269,600 Founder Shares held by it and to have 922,453 Private Placement Warrants held in trust, in each case, until such securities become released upon the achievement of certain performance-based milestones under the Sponsor Agreement. The Founder Shares held by the Sponsor’s directors and Advisors were not subject to vesting or forfeiture.
On November 14, 2021, the parties entered into an amendment to the Sponsor Support Agreement pursuant to which, among other things, the Sponsor Earn-Out Shares and Earn-Out Warrants shall no longer be subject to vesting conditions, in each case as consideration for the Sponsor agreeing to enter into the Backstop (as defined below).
Forward Purchase Agreement
In connection with the consummation of its initial public offering, THMA entered into the Forward Purchase Agreement with the KLP SPAC 1 LLC (the “Anchor Investor”), that provided for the purchase, in the aggregate, of 5,000,000 Original Forward Purchase Units, consisting of one Forward Purchase Share and one-third of one Forward Purchase Warrant, at a purchase price of $10.00 per unit, or an aggregate purchase price of $50,000,000, in a private placement concurrently with the closing of THMA’s initial business combination.
In connection with the execution of the Business Combination Agreement, the Anchor Investor entered into the Amendment to Forward Purchase Agreement with THMA, pursuant to which, effective as of immediately prior to the Closing, the Forward Purchase Agreement was amended to (i) eliminate the sale of warrants to purchase THMA Class A Common Shares and (ii) instead provide exclusively for the sale of such number of THMA Class A Common Shares equal to the sum of (x) 2,300,000 and (y) such additional THMA Class A Common Shares as the Anchor Investor elected to purchase up to the lesser of (A) the number of Public Shares redeemed by THMA’s Public Stockholders and (B) 2,700,000, in each case, for a purchase price of $10.00 per THMA Class A Common Share.
On November 14, 2021, THMA entered into an amendment to the Amended Forward Purchase Agreement, pursuant to which, among other things, the Anchor Investor increased its binding forward purchase commitment
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 176
(the “Backstop”), subject to cutback, from $23.0 million to up to $73.0 million and removed any optional element to the Backstop. The Backstop was subject to certain reductions if gross transaction proceeds exceeded each of $175.0 million and $250.0 million, respectively. The Anchor Investor was permitted to offer PIPE Investors the opportunity to participate in the Backstop in an amount up to $23.0 million, subject to cutback if proceeds exceeded $175.0 million. As consideration for committing to fund the Backstop, THMA and Legacy Pear agreed to release the Sponsor Earn-Out Shares and Earn-Out Warrants from certain earn-out conditions in connection with the Business Combination, with a certain number of such securities being transferred to each Subscriber that participated in the Backstop.
On December 2, 2021, THMA entered into the Forward Purchase Agreement Assignment pursuant to which, among other things, the Anchor Investor assigned its right to purchase 500,000 THMA Class A Common Shares to a PIPE Investor.
An entity affiliated with the Pritzker Vlock Family Office holds an indirect economic interest in each of the Sponsor and the Anchor Investor. Elon Boms, who was the Chief Executive Officer and director of THMA until the closing of the Business Combination, is a Managing Director of the Pritzker Vlock Family Office and a manager of the Anchor Investor. Steve Benson, who was THMA’s Chief Operating Officer and director until the closing of the Business Combination, is a Venture Partner with the Pritzker Vlock Family Office. Joseph Iannotta, who was THMA’s Chief Financial Officer until the closing of the Business Combination, is the Controller of the Pritzker Vlock Family Office. Messrs Boms, Benson, and Iannotta led and assisted in, respectively, the evaluation of THMA’s business combination targets, including Legacy Pear, and the negotiation of THMA’s Business Combination with Legacy Pear.
Administrative Support
Prior to the closing of the Business Combination, THMA utilized office space at 195 Church Street, 15th Floor, New Haven, Connecticut 06510 from the Sponsor. On February 2, 2021, THMA began paying to the Sponsor $10,000 per month for office space, secretarial, and administrative services provided to members of their management team. Upon completion of the Business Combination, THMA ceased paying these monthly fees. For the year ended December 31, 2021, THMA incurred and paid $100,000 of such fees.
Related Party Loans and Advances
On December 7, 2020, THMA issued an unsecured promissory note to the Sponsor, pursuant to which THMA was permitted to borrow up to an aggregate principal amount of $300,000. Such promissory note was non-interest bearing and payable on the earlier of December 31, 2021 and the completion of the Initial Public Offering. The outstanding balance under such promissory note of $136,833 was repaid at the closing of THMA’s initial public offering on February 4, 2021.
On June 21, 2021, THMA issued an unsecured promissory Note (the “2021 Note”) in the principal amount of $1,000,000 to the Sponsor in exchange for up to $1 million in working capital loans. The 2021 Note was non-interest bearing and payable upon the consummation of the Business Combination. Upon completion of the Business Combination, THMA repaid such loaned amounts out of the proceeds of the Trust Account released to THMA.
Registration Rights Agreement
THMA entered into a registration rights agreement with the Sponsor, the Anchor Investor and THMA’s directors, Advisors and officers with respect to the Private Placement Warrants, the THMA Class A Common Shares issued to the Anchor Investor pursuant to the Forward Purchase Agreement and the THMA Class A Common Shares issued upon exercise of the foregoing and upon conversion of the Founder Shares.
In connection with the consummation of the Business Combination, we entered into the Amended and Restated Registration Rights Agreement with certain Pear stockholders, pursuant to which we agreed to register for resale, pursuant to Rule 415 under the Securities Act, the Class A Common Shares and other equity securities of Pear that are held by the parties thereto from time to time.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 177
The Amended and Restated Registration Rights Agreement grants the Anchor Investor, the Sponsor, and certain of our stockholders the right to require, subject to certain conditions and limitations, that we register for resale, securities held by such stockholders and certain “piggyback” registration rights with respect to registrations initiated by us. Pursuant to the PIPE Subscription Agreements, the PIPE Investors were also granted registration rights which required us to file a registration statement registering the resale of the PIPE Shares. The registration of Class A Common Shares pursuant to the exercise of the registration rights provided under the Registration Rights Agreement and the PIPE Subscription Agreements enable the applicable Pear stockholders to resell such shares without restriction under the Securities Act. We bear the expenses incurred in connection with the filing of any registration statements pursuant to the Registration Rights Agreement and the PIPE Subscription Agreements and indemnify the stockholders against certain liabilities.
Certain Relationships and Related Person Transactions of Legacy Pear
Legacy Pear Series D-1 and D-2 Preferred Stock Financing
From December 7, 2020 through February 25, 2021, Legacy Pear sold an aggregate of 15,293,315 Legacy Pear Series D-1 Preferred Shares at a purchase price of approximately $6.5388 per share, for an aggregate purchase price of $99,999,928.30 (the “Legacy Pear Series D-1 Preferred Stock Financing”). The participants in the Legacy Pear Series D-1 Preferred Stock Financing included persons affiliated with members of Legacy Pear’s board of directors and persons that held more than 5% of Legacy Pear’s outstanding capital stock. The following table summarizes purchases of Legacy Pear Series D-1 Preferred Shares from Legacy Pear by such related persons:
(1) Elena Viboch is a former member of the board of directors of Legacy Pear and an affiliate of SVF II AIV (DE) LLC (“Softbank”). Softbank currently holds more than 5% of Pear’s outstanding capital stock. Ms. Viboch does not serve on the Pear Board.
(2) TLS Beta Pte. Ltd. (“Temasek”) currently holds more than 5% of Pear’s outstanding capital stock.
(3) Andrew Schwab is a former member of the board of directors of Legacy Pear and an affiliate of 5AM Opportunities I, L.P. (“5AM Opportunities”). 5AM Opportunities and its affiliated entities, 5AM Ventures IV, L.P. (“5AM Ventures”) and 5AM Co-Investors IV, L.P. (“5AM Co-Investors”), currently hold more than 5% of Pear’s outstanding capital stock. Mr. Schwab is also an affiliate of each of 5AM Ventures and 5AM Co-Investors. Mr. Schwab is currently a member of the Pear Board.
(4) Timothy Petersen is a former member of the board of directors of Legacy Pear and an affiliate of Arboretum Ventures IV, L.P. (“Arboretum”). Arboretum currently holds more than 5% of Pear’s outstanding capital stock. Mr. Petersen does not serve on the Pear Board.
(5) Zack Lynch is a former member of the board of directors of Legacy Pear and an affiliate of JAZZ Human Performance Technology Fund, L.P. and JAZZ Human Performance Opportunity Fund, L.P. (collectively, “JAZZ”). JAZZ currently holds more than 5% of Pear’s outstanding capital stock. Mr. Lynch is currently a member of the Pear Board.
On December 8, 2020, Legacy Pear sold an aggregate of 8,109,888 Legacy Pear Series D-2 Preferred Shares at a purchase price of approximately $3.9458 per share, for an aggregate purchase price of $31,999,996.08 (the “Legacy Pear Series D-2 Preferred Stock Financing”). The participants in the Legacy Pear Series D-2 Preferred Stock Financing included persons affiliated with members of Legacy Pear’s board of directors and persons that held more than 5% of Legacy Pear’s outstanding capital stock. The following table summarizes purchases of Legacy Pear Series D-2 Preferred Shares from Legacy Pear by such related persons:
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 178
__________________
(1) Elena Viboch is a former member of the board of directors of Legacy Pear and an affiliate of Softbank. Softbank currently holds more than 5% of Pear’s outstanding capital stock. Ms. Viboch does not serve on the Pear Board.
(2) Temasek currently holds more than 5% of Pear’s outstanding Class A common stock.
In connection with the Legacy Pear Series D-1 Preferred Stock Financing and the Legacy Pear Series D-2 Preferred Stock Financing, Legacy Pear repurchased (i) 2,788,732 Legacy Pear Common Shares from Corey McCann, Pear’s Chief Executive Officer, for $10,996,806.90, (ii) 480,000 Legacy Pear Common Shares and 1,920,000 Legacy Pear Series A Preferred Shares from 5AM Ventures for $9,463,920, (iii) 20,000 Legacy Pear Common Shares and 80,000 Legacy Pear Series A Preferred Shares from 5AM Co-Investors for $394,330 and (iv) 1,000,000 Legacy Pear Series A Preferred Shares from Arboretum for $3,943,300, each pursuant to a tender offer by Legacy Pear that closed on December 9, 2020.
Pear Stockholder Support Agreements
In connection with the execution of the Business Combination Agreement, certain equityholders of Legacy Pear representing the requisite votes necessary to approve the Merger (such stockholders, the “Pear Supporting Equityholders”) entered into stockholder support agreements with THMA and Legacy Pear (the “Stockholder Supporting Agreements”), pursuant to which each Supporting Pear Equityholder agreed to, among other things, (a) vote all of its Legacy Pear Common Shares and Legacy Pear Preferred Shares (or any securities convertible into or exercisable or exchangeable for Legacy Pear Common Shares and Legacy Pear Preferred Shares) in favor of the approval and adoption of the Business Combination Agreement, the Ancillary Agreements to which Legacy Pear was party and the transactions contemplated thereby (including the Merger) and (b) be bound by certain other covenants and agreements related to the Merger.
Investors’ Rights Agreement
Legacy Pear was party to a Third Amended and Restated Investors’ Rights Agreement, dated as of November 3, 2020 (the “IRA”), as amended by that certain Omnibus Amendment dated as of February 23, 2021, which granted registration rights and information rights, among other things, to certain holders of Legacy Pear’s capital stock, including entities affiliated with Temasek, Softbank, Arboretum, JAZZ, 5AM Ventures, 5AM Co-Investors and 5AM Opportunities. The IRA terminated upon the closing of the Business Combination.
Right of First Refusal and Co-Sale Agreement
Pursuant to certain agreements with its stockholders, including the Third Amended and Restated Right of First Refusal and Co-Sale Agreement, dated as of November 3, 2020 (the “ROFR Agreement”), as amended by that certain Omnibus Amendment dated as of February 23, 2021, Legacy Pear had the right to purchase shares of Legacy Pear’s capital stock that certain of its stockholders proposed to sell to other parties. Certain holders of Legacy Pear’s capital stock that were party to the ROFR Agreement, included Temasek, Softbank, Arboretum, JAZZ, 5AM Ventures, 5AM Co-Investors and 5AM Opportunities. The ROFR Agreement terminated upon the closing of the Business Combination.
Voting Agreement
Legacy Pear was party to the Third Amended and Restated Voting Agreement, dated as of November 3, 2020 (the “Voting Agreement”), as amended by that certain Omnibus Amendment dated as of February 23, 2021, pursuant to which certain holders of Legacy Pear’s capital stock, including Temasek, Softbank, Arboretum, JAZZ, 5AM Ventures, 5AM Co-Investors and 5AM Opportunities, agreed to vote their shares of Legacy Pear’s capital stock in favor of certain matters, including with respect to the election of directors. The Voting Agreement terminated upon the closing of the Business Combination.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 179
Director and Officer Indemnification
Legacy Pear’s certificate of incorporation and bylaws provided for indemnification and advancement of expenses for its directors and officers to the fullest extent permitted by the DGCL, subject to certain limited exceptions. Legacy Pear also entered into indemnification agreements with each of its directors. Following the Business Combination, Pear entered into new indemnification agreements with each of the directors and officers of Pear. The indemnification agreements entered into by Pear Board members Messrs. Schwab and Lynch also provided certain indemnification rights to the entities with which they are affiliated.
Director Independence
Our Board has determined that none of the members of our Board other than Dr. McCann has a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director and that each of the members of our Board other than Mr. McCann is “independent” as that term is defined under the rules of Nasdaq. Our Board has also determined that all members of our Audit Committee, Compensation Committee and Nominating and Corporate Governance Committee are independent and satisfy the relevant SEC and Nasdaq independence requirements for such committees. The transactions, relationships or arrangements described above were considered by the Board in determining director independence.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
Independent Registered Public Accounting FirmThe information required by this item is incorporated by reference to our Proxy Statement.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The information required by this Feesitem and Services
Deloitte was appointed asis incorporated by reference to our independent registered accounting firm in December 2021 and served as the independent registered public accounting firm of Legacy Pear since 2017.
The following table presents the aggregate fees billed by Deloitte & Touche LLP to us (including Legacy Pear) for the years ended:
Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Registered Public Accounting Firm
Our Audit Committee’s policy is to pre‑approve all audit and permissible non‑audit services provided by our independent registered public accounting firm, the scope of services provided by our independent registered public accounting firm, and the fees for the services to be performed. These services may include audit services, audit‑related services, tax services, and other services. Pre‑approval is detailed as to the particular service or category of services and is generally subject to a specific budget. Our independent registered public accounting firm and management are required to periodically report to the audit committee regarding the extent of services provided by our independent registered public accounting firm in accordance with this pre‑approval, and the fees for the services performed to dateProxy Statement.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 180167
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULE
(a) Documents filed as part of this report
(1) All financial statements
| | | | | | | | |
Index to Consolidated Financial Statements | | Page |
| | |
| | |
| | |
| | |
| | |
| | |
(2) Financial Statement Schedules
All financial statement schedules have been omitted, since the required information is not applicable or is not present in amounts sufficient to require submission of the schedule, or because the information required is included in the consolidated financial statements and accompanying notes included in this Form 10-K.
(3) Exhibits required by Item 601 of Regulation S-K
EXHIBIT INDEX
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | Incorporated by Reference |
| Exhibit Number | | Exhibit Description | | Schedule/
Form | | File No. | | Exhibit | | Filed Date/
Period End Date |
| | | | | | | | | | |
| 2.1 | | | | 8-K | | 001-39969 | | 2.1 | | June 22, 2021 |
| | | | | | | | | | |
3.1 | | | | 8-K | | 001-39969 | | 3.1 | | December 8, 2021 |
| | | | | | | | | | |
3.2 | | | | 8-K | | 001-39969 | | 3.2 | | December 8, 2021 |
| | | | | | | | | | |
| 4.1 | | | | S-1 | | 333-252150 | | 4.2 | | January 15, 2021 |
| | | | | | | | | | |
| 4.2 | | | | S-1 | | 333-252150 | | 4.3 | | January 15, 2021 |
| | | | | | | | | | |
| 4.3 | | | | 8-K | | 001-39969 | | 4.1 | | February 4, 2021 |
| | | | | | | | | | |
| 4.4 | | | | 10-K | | 001-39969 | | 4.4 | | March 29, 2022 |
| | | | | | | | | | |
10.1 | | | | 8-K | | 001-39969 | | 10.1 | | December 8, 2021 |
| | | | | | | | | | |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 181168
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 10.2 | | | | 8-K | | 001-39969 | | 10.1 | | June 22, 2021 |
| | | | | | | | | | |
| 10.3 | | | | 8-K | | 001-39969 | | 10.2 | | November 15, 2021 |
| | | | | | | | | | |
| 10.4 | | | | 8-K | | 001-39969 | | 10.4 | | June 22, 2021 |
| | | | | | | | | | |
10.5 | | | | 8-K | | 001-39969 | | 10.8 | | December 8, 2021 |
| | | | | | | | | | |
10.6 | | | | 8-K | | 001-39969 | | 10.9 | | December 8, 2021 |
| | | | | | | | | | |
10.7 | | | | 8-K | | 001-39969 | | 10.10 | | December 8, 2021 |
| | | | | | | | | | |
| 10.8 | | | | 8-K | | 001-39969 | | 10.11 | | December 8, 2021 |
| | | | | | | | | | |
| 10.9 | | | | 8-K | | 001-39969 | | 10.12 | | December 8, 2021 |
| | | | | | | | | | |
10.10 | | | | 8-K | | 001-39969 | | 10.13 | | December 8, 2021 |
| | | | | | | | | | |
| 10.11 | | | | 8-K | | 001-39969 | | 10.14 | | December 8, 2021 |
| | | | | | | | | | |
| 10.12 | | | | 8-K | | 001-39969 | | 10.15 | | December 8, 2021 |
| | | | | | | | | | |
| 10.13 | | | | S-4/A | | 001-39969 | | 10.19 | | October 25, 2021 |
| | | | | | | | | | |
| 10.14 | | | | 8-K | | 001-39969 | | 10.19 | | December 8, 2021 |
| | | | | | | | | | |
| 10.15 | | | | S-4/A | | 001-39969 | | 10.20 | | October 25, 2021 |
| | | | | | | | | | |
| 10.16 | | | | S-4/A | | 001-39969 | | 10.21 | | October 25, 2021 |
| 10.17 | | | | S-4/A | | 001-39969 | | 10.22 | | October 25, 2021 |
| | | | | | | | | | |
| 10.18 | | | | S-4/A | | 001-39969 | | 10.23 | | October 25, 2021 |
| | | | | | | | | | |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 182169
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 10.19 | | | | S-4/A | | 001-39969 | | 10.24 | | October 25, 2021 |
| | | | | | | | | | |
| 10.20 | | | | S-4/A | | 001-39969 | | 10.28 | | October 25, 2021 |
| | | | | | | | | | |
| 10.21 | | | | S-1 | | 333-261876 | | 10.26 | | December 23, 2021 |
| | | | | | | | | | |
| 10.22 | | | | S-4/A | | 001-39969 | | 10.31 | | October 25, 2021 |
| | | | | | | | | | |
| 10.23 | | | | S-4/A | | 001-39969 | | 10.32 | | October 25, 2021 |
| | | | | | | | | | |
| 10.24 | | | | S-4/A | | 001-39969 | | 10.33 | | October 25, 2021 |
| | | | | | | | | | |
| 10.25 | | | | S-4/A | | 001-39969 | | 10.34 | | October 25, 2021 |
| | | | | | | | | | |
| 10.26 | | | | 8-K | | 001-39969 | | 10.31 | | December 8, 2021 |
| | | | | | | | | | |
10.27 | | | | 8-K | | 001-39969 | | 10.32 | | December 8, 2021 |
| | | | | | | | | | |
| 10.28 | | | | 10-K | | 001-39969 | | 10.33 | | March 29, 2022 |
| | | | | | | | | | |
| 10.29 | | | | 10-K | | 001-39969 | | 10.34 | | March 29, 2022 |
| | | | | | | | | | |
| 10.30 | | Amended and Restated Security Agreement, dated as of March 25, 2022, by and among Pear Therapeutics, Inc., Pear Therapeutics (US), Inc., Perceptive Credit Holdings III, LP, as administrative agent, and the other parties thereto* | | 10-K | | 001-39969 | | 10.35 | | March 29, 2022 |
| | | | | | | | | | |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 183170
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 10.31 | | Amended and Restated Intercompany Subordination Agreement, dated as of March 25, 2022, by and among Pear Therapeutics, Inc., Pear Therapeutics (US), Inc., Perceptive Credit Holdings III, LP, as administrative agent, and the other parties thereto* | | 10-K | | 001-39969 | | 10.36 | | March 29, 2022 |
| | | | | | | | | | |
| 10.32 | | | | 8-K | | 001-39969 | | 10.1 | | June 15, 2022 |
| | | | | | | | | | |
| 10.33 | | | | 8-K | | 001-39969 | | 1.1 | | January 3, 2023 |
| | | | | | | | | | |
| 10.34 | | | | | | | | | | |
| | | | | | | | | | |
| 10.35 | | | | | | | | | | |
| | | | | | | | | | |
| 10.36 | | First Amendment to Amended and Restated Credit Agreement and Guaranty, dated as of January 13, 2023, by and among Pear Therapeutics, Inc., Pear Therapeutics (US), Inc., Perceptive Credit Holdings III, LP, as administrative agent, and the other parties thereto | | | | | | | | |
| | First Amendment to Amended and Restated Credit Agreement and Guaranty | | | | | | | | |
| 21.1 | | | | 8-K | | 001-39969 | | 21.1 | | December 8, 2021 |
| | | | | | | | | | |
| 23.1 | | | | | | | | | | |
| | | | | | | | | | |
| 31.1 | | | | | | | | | | |
| | | | | | | | | | |
| 31.2 | | | | | | | | | | |
| | | | | | | | | | |
| 32.1 | | | | | | | | | | |
| | | | | | | | | | |
| 101.INS | | Inline XBRL Instance Document | | | | | | | | |
| | | | | | | | | | |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema Document | | | | | | | | |
| | | | | | | | | | |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase Document | | | | | | | | |
| | | | | | | | | | |
| 101.DEF | | Inline XBRL Taxonomy Definition Linkbase Document | | | | | | | | |
| | | | | | | | | | |
| 101.LAB | | Inline XBRL Taxonomy Label Linkbase Document | | | | | | | | |
| | | | | | | | | | |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase Document | | | | | | | | |
Pear Therapeutics, Inc. | 2022 Form 10-K |Page 171
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | | | | | | | | |
__________________
*Previously filed
**Certain exhibits and schedules to this Exhibit have been omitted in accordance with Regulation S-K Item 601(b)(2). The Company agrees to furnish supplementally a copy of all omitted exhibits and schedules to the Securities and Exchange Commission upon its request.
+ Certain confidential portions (indicated by brackets and asterisks) have been omitted from this exhibit.
Pear Therapeutics, Inc.| 2021 Form 10-K |Page 184
‡ Management contract or compensatory plan or arrangement. ITEM 16. FORM 10-K SUMMARY
None.
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 185172
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| PEAR THERAPEUTICS, INC. |
| | | | | | | | | | | | |
| Date: March 2831, 20222023 | By: | /s/ Corey M. McCann |
| | Name: | Dr. Corey M. McCann |
| | Title: | President and Chief Executive Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, this Annual Report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| | | | | | | | | | | | | | |
| Signature | | Title | | Date |
| | | | |
| /s/ Dr. Corey M. McCann | |
Chief Executive Officer, President and Director | | March 2831, 20222023 |
| Dr. Corey M. McCann | | (Principal Executive Officer) | |
| | | | |
/s/ Christopher D.T. Guiffre | | Chief Financial Officer and Chief Operating Officer | | March 2831, 20222023 |
| Christopher D.T. Guiffre | | (Principal Financial Officer) | |
| | | | |
/s/ Ellen E. Snow | | Vice President, Corporate ControllerChief Accounting Officer | | March 2831, 20222023 |
| Ellen E. Snow | | (Principal Accounting Officer) | |
| | | | |
/s/ Alison Bauerlein | | Director | | March 2831, 20222023 |
| Alison Bauerlein | | |
| | | | |
/s/ Jorge M. Gomez | | Director | | March 28, 2022 |
| Jorge M. Gomez | | |
| | | | |
| /s/ Zack LynchPaul Mango | | Director | | March 2831, 20222023 |
| Zack LynchPaul Mango | | | | |
| /s/ Shivakumar Rajaraman | | Director | | March 31, 2023 |
| Shivakumar Rajaraman | |
| | |
| /s/ Kirthiga Reddy | | Director | | March 2831, 20222023 |
| Kirthiga Reddy | | |
| | | | |
| /s/ Nancy M. Schlichting | | Director | | March 28, 2022 |
| Nancy M. Schlichting | | |
| | | | |
/s/ Andrew SchwabNancy M. Schlichting | | Director | | March 28, 202231, 2023 |
Nancy M. Schlichting | | | | |
| /s/ Timothy A. Wicks | | Director | | March 31, 2023 |
| Andrew SchwabTimothy A. Wicks | | | | |
Pear Therapeutics, Inc. | 20212022 Form 10-K |Page 186173
Exhibit 10.34
Execution VersionCertain identified information has been omitted from this exhibit because it is not material and of the type that the registrant treats as private or confidential. [***] indicates that information has been omitted.
THIRD AMENDMENT TO CREDIT AGREEMENT AND GUARANTY200 State Street, 13th floor Boston, MA 02109
December 17, 2022
This THIRD AMENDMENT TO CREDIT AGREEMENT AND GUARANTY, dated as of March 25, 2022
Katherine Jeffery
[***]
[***]
The purpose of this letter agreement (this “Agreement”), is made by andto confirm the among PEAR THERAPEUTICS, INC., a Delaware corporation (f/k/a THIMBLE POINT ACQUISITION CORP.)terms of your separation of employment from Pear Therapeutics (US), Inc. (“HoldingsPear” or the “Company”), PEAR THERAPEUTICS (US),including Pear’s offer to provide you with certain pay and benefits in exchange for your agreeing to the general release of claims and certain other commitments provided for below. This Agreement shall be effective on the eighth (8ᵗʰ) INC.,day a Delaware corporation (f/k/a PEAR THERAPEUTICS, INC.)after the date you sign it (the “BorrowerEffective Date”), certain Subsidiaries of the Borrower, and PERCEPTIVE CREDIT HOLDINGS III,at which time it shall become final and binding on all parties, unless you revoke this Agreement as provided in Section 11(h). You will have until 45 days after the Separation Date to consider whether to LP,sign a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”) and as the Majority Lender. this Agreement.
RECITALS
WHEREAS, reference is made to the Credit Agreement1.Separation of Employment. Your last day with the Company including your ability to access Company systems and Guaranty, dated assoftware will be March 31, 2023, or your date of June 30, 2020, among the Borrower, certain Subsidiaries of the Borrower from time to time party thereto, the lenders from time to time party theretoresignation, whichever occurs first (the “LendersSeparation Date”). andSo the Administrative Agent (as subsequently amended, supplemented or otherwise modified, from time to time, including pursuant to this Agreement, the “Credit Agreement”);
WHERAS, after giving effect to the transactions contemplated by the Business Combination Agreement, dated as of June 21, 2021, by and among Holdings, Oz Merger Sub, Inc. and the Borrower, the Borrower is a wholly-owned Subsidiary of Holdings;
WHEREAS, subject to the terms and the conditions set forth in this Agreement, the Borrower has requested that the Credit Agreement be amended and restated as provided herein, in order to, among other things, provide for Holdings as a party thereunder;
WHEREAS, the Administrative Agent and the Majority Lenders are willing to do so on the terms and subject tolong as you remain an Executive Officer, the Company will continue to list you on the Company’s website until the conditions set forth in this Agreement; and
WHEREAS, the Lender party hereto constitutes the Majority LendersSeparation Date.
2.Payments. Regardless of whether you sign this Agreement or allow it to become effective, you will receive on the Separation Date:
(a)Payment reflecting pay for any time worked through the Separation Date that has not already been paid through Pear’s regular payroll process; and
(b)Payment of all accrued and unused Paid Time Off (PTO).
NOW, THEREFORE, for good and valuable consideration (including the Borrower’s covenants and agreements set forth below), the parties hereto hereby agree as follows:
ARTICLE I3.Separation Pay and Benefits.
AMENDMENTS TO CREDIT AGREEMENT
Section 1.01. Defined Terms. Unless otherwise defined hereinIf you sign and allow this Agreement to or the context otherwise requires, capitalized terms used inbecome effective, comply with this Agreement (including the preambles and recitals hereto and hereof) shall have the meanings ascribed to such termsand with the agreement referenced in Section 9(b) below), Pear will provide you with the following (the “Separation Pay and Benefits”):
(a)a one-time lump-sum payment in the Credit Agreement.
Section 1.02. Amendment to Credit Agreement. As of, andgross amount of Three Hundred Forty-Eight Thousand Five Hundred Dollars and 00/100 Cents ($348,500.00), subject to the occurrence of, the Effective Date, the Credit Agreement is hereby amended and restated in its entirety to read as set forth in Exhibit A hereto (the Credit Agreement, as so amended and restated, the “Amended and Restated Credit Agreement”). Each of the existing Exhibits to the Credit Agreement will remain in full force and effect. The parties hereto agree that as of, and subject to the occurrence of, the Effective Date, the waiver set forth in Section 1.02 of the Waiver, Amendment and applicable taxes and other withholdings as required by law, to be paid within thirty days after the Effective Date;
Agreement, dated as of June(b)continued eligibility for the Company’s health & dental insurance through the Separation Date, in addition to eligibility for the COBRA benefit described in Section 5(b) below; and
(c)the right to receive additional vesting of any Restricted Stock Units (“RSUs”) that were previously granted to you but have not currently vested, which RSUs will be amended to provide that: (i) the RSUs will remain outstanding through February 29, 2024, and will vest on any vesting date to occur prior to that date, notwithstanding your termination of employment, and
(ii) the remaining portion of the RSUs that would not vest on or before February 29, 2024 will terminate on the Effective Date.
4.Change of Salary. Notwithstanding your Separation Pay and Benefits described above, as of January 111, 2021, by and among the Borrower, the Administrative Agent and the Lenders shall have no further force and effect.
Section 1.03. No Other Waivers, Amendments or other Modifications Implied or Intended2023, your salary will be Five Thousand Dollars and 00/100 Cents ($5,000.00) per month, subject to applicable taxes and other withholdings as required by law, payable bi-weekly in arrears.
5.Unemployment Insurance and COBRA.
(a)You may seek unemployment benefits following the separation of your employment from Pear. You acknowledge that any decision regarding eligibility for and amounts of unemployment benefits are made by the relevant government agency, not by Pear.
(b)Providing that you remain eligible under the provisions of Consolidated Omnibus Budget Reconciliation Act of 1985 (COBRA), and upon timely completion of the appropriate forms, for the period commencing on April 1, 2023, the Company will pay your COBRA group health insurance premiums (as such premiums are due) and your eligible dependents until the earlier of (i) March 31, 2024, or (ii) the date when you become eligible for substantially equivalent health insurance coverage in connection with new employment or self-employment. The “qualifying event” under COBRA shall be deemed to have occurred on March 31, 2023.
6.. Any term or provision hereofEquity. You acknowledge that as of the Separation Date you are vested in that number of stock options, RSUs and/or shares of equity of Pear Therapeutics, Inc. pursuant to the contrary notwithstanding, the waivers, amendments2021 Stock Option and other modification contemplated hereby relate solely to the specific sections or provisions ofIncentive Plan (the “Plan”), as set forth in the Credit Agreement expressly referenced herein and relate solelyyour account with Shareworks/Morgan Stanley @ Work. Except as to the periods or transactions expressly referenced herein, and noset forth herein, you further acknowledge that all vesting shall terminate immediately upon the Separation Date and any otherand or additional waiver, amendment or other modification is made, implied or intended to be madeall rights and obligations with respect to any other term or provision of any Loan Document, or any Obligation of any Obligor or any rights or remedies of any Secured Party under any Loan Document, whether now existing or occurring in the future (including any future Default), including, but not limited to, the right to declare all or any portion of the outstanding principal amount of the Loansvested shares, whether they are options or RSUs, shall be governed solely by the terms of the Plan and your stock option and/or restricted stock award agreement(s). Notwithstanding the foregoing, if you timely execute and return to Pear the Option Amendment Agreement attached hereto as Exhibit D, then (i) your stock options will continue to vest through February 29, 2024 (notwithstanding your termination of employment), (ii) the period for exercising your stock options, to the extent that they have vested as of February 29, 2024, will be extended until March 31, 2024, and (iii) as a result of such amendment, any stock options that were “incentive stock options” will no longer qualify as such and will automatically become non-statutory stock options. Please note that the executed Option Amendment Agreement must be returned to Pear by no later than one week after the date hereof, if you wish to have your stock options amended as
described in the preceding sentence. You acknowledge and agree that, except as specifically set forth in Section 3(c) of this Agreement, you do not now have, and will not in the future have, rights to vest in any other equity plans (of whatever name or kind) that you participated in or were eligible to participate in during your employment with the Company. In addition, in exchange for the consideration received hereunder, you hereby irrevocably waive any right to receive a Q3 Performance Bonus Award of RSUs for which you were otherwise eligible.
7.Second Severance. In addition, after your Separation Date, you will be eligible for a second severance payment in a one-time lump-sum payment in the amount of One Thousand Dollars and 00/100 Cents ($1,000.00), subject to applicable taxes and other withholdings as required by law (the “Second Severance”), to be paid in exchange for a duly executed release agreement reasonably satisfactory to the Company (the “Second Release”). The Company shall pay you the Second Severance within fifteen days after you have signed and delivered the Second Release to the Company’s General Counsel.
8.Acknowledgements. You acknowledge and agree that this Agreement and the Separation Pay and Benefits to be provided to you herein are not intended to, and shall not constitute, a severance plan and shall confer no benefit on anyone other than Pear and you. You acknowledge and agree that the pay and benefits provided for herein are not otherwise due or owing to you under any offer letter or other agreement (oral or written) or any Pear policy or practice. You further acknowledge that, except for (i) any unpaid regular wages (including accrued and unused vacation) earned through the last day of employment, (ii) any expenses for which you submit required documentation within the timeframe requested by the Company and which will be paid consistent with the Company’s usual practice, and (iii) any payments you may become eligible for pursuant to the terms of this Agreement, you have been paid and provided all wages, vacation pay, holiday pay, bonus, commission, equity, business expenses, and any other form of compensation or benefit that may be due to you now or which would have become due in the future in connection with your employment with or separation of employment from Pear. You hereby agree that this Agreement supersedes any rights you may have under any other severance agreement or policy with the Company including, without limitation, the Severance and Change in Control Plan, and you hereby waive any such rights. You affirm that you have no known workplace injuries or occupational diseases relating to your employment and that you have been provided and/or have not been denied any leave to which you were entitled under any family or medical leave law or otherwise.
9.Confidentiality; Non-Disparagement; Cooperation; Other Assurances.
(a)You will return to Pear within five (5) days after the Separation Date, without modifying, damaging or disabling the same, and not retain any copies of: (i) all originals and copies of any proprietary or confidential information and trade secrets of the Company, whether in print, electronic or other form; (ii) all originals and copies of Company, customer, supplier and collaborator files, written materials, records and other documents, whether in print, electronic or other form, whether made by you or coming into your possession during the course of your employment with the Company; (iii) all identification cards, keys, security passes or other means of access to Company facilities; (iv) all credit cards, telephone cards, telephones, computer or other office equipment; (v) any other property of the Company in your possession, custody or control,
including all hardware (e.g., personal computers, laptops, thumb drives/CDs/DVDs, information stored on thumb drives/CDs/DVDs); and (vi) all usernames and passwords and any other access credentials maintained by you. You further agree that if you discover any other Company materials in your possession after the Separation Date, you immediately will return such materials to the Company. When you sign this Release, you shall also sign the Declaration attached as Exhibit A.
(b)You hereby reaffirm your obligations set forth in the Employee Nondisclosure, Non-solicitation, and Assignment of Intellectual Property Agreement between Pear and you (the “Covenants Agreement”), in substantially the form attached hereto as Exhibit
B. The Company will provide a copy of your signed Covenants Agreement upon your request. You further agree to abide by any and all common law and/or statutory obligations relating to the protection and non-disclosure of Pear’s trade secrets and/or confidential and proprietary documents and information. Consistent with the Defend Trade Secrets Act of 2016, Pear hereby notifies you of the following provisions of the DTSA: “(1) An individual shall not be held criminally or civilly liable under any Federal or State trade secret law for the disclosure of a trade secret that (A) is made (i) in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney; and (ii) solely for the purpose of reporting or investigating a suspected violation of law; or (B) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. (2) An individual who files a lawsuit for retaliation by an employer for reporting a suspected violation of law may disclose the trade secret to the attorney of the individual and use the trade secret information in the court proceeding, if the individual (A) files any document containing the trade secret under seal; and (B) does not disclose the trade secret, except pursuant to court order.” Nothing in this Agreement shall be construed to authorize, or limit liability for, an act that is otherwise prohibited by law, such as the unlawful access of material by unauthorized means.
(c)Except as to your ongoing obligations, You agree that all information relating in any way to the subject matter of this Agreement, including the existence and provisions of this Agreement, will be held confidential by you and will not be publicized or disclosed to any person (other than an immediate member of your family or your legal counsel, accountant or financial advisor, provided that any such individual to whom disclosure is made shall be bound by these confidentiality obligations), other than a state or federal tax authority or government agency to which disclosure is mandated by applicable state or federal law. Furthermore, you agree that you will not discuss this Agreement with any other Pear employee, including your peers, except to notify individuals that you will be leaving Pear on the Separation Date. Notwithstanding the foregoing, if you are a California resident, nothing in this Agreement prevents you from discussing or disclosing information about unlawful acts in the workplace, such as harassment or discrimination or any other conduct that you have reason to believe is unlawful.
(d)You agree that you will not make any statements that are professionally or personally disparaging, defamatory or false about, or adverse to, the interests of Pear (including its officers, directors, advisors, investors and employees) including, but not limited to, any statements that disparage any person, product, service, finances, financial condition, capability or any other aspect of the business of Pear, and that you will not engage in any conduct which could reasonably be expected to result in professional or personal harm to the reputation of Pear (including its officers, directors, advisors, investors and employees). Nothing herein shall prohibit
or bar you from providing truthful testimony in any legal proceeding or in communicating with any governmental agency or representative or from making any truthful disclosure required, authorized or permitted under law; provided however that if you anticipate or are required to make disclosure pursuant to this subparagraph (iii), you shall inform the Company’s General Counsel, in advance of any disclosure, at least ten (10) days prior to such disclosure whenever possible, and where not possible, with as much advance notice as practicable. Nothing in this Agreement shall limit the rights of any government agency or any party’s right of access to, participation in or cooperation with any government agency or its actions. Notwithstanding the foregoing, if you are a California resident, and in accordance with applicable law, you acknowledge and agree that the foregoing does not prohibit the disclosure of factual information (except for the amount of financial consideration provided for herein) related to civil or administrative claims regarding sexual harassment, sex discrimination, sexual assault, discrimination or harassment on the basis of any other statutorily protected category, or retaliation relating to any of the foregoing. You also hereby acknowledge and agree that as of the Effective Date, no such civil or administrative action has been filed by you or anyone on your behalf against the Company.
(e)You agree that you will make yourself reasonably available to Pear (including its attorneys), (either by telephone or videoconference or, if the Company believes necessary, in person) to assist the Company in any matter reasonably relating to the services performed by you during your employment with the Company including, but not limited to, transitioning your duties to others and ensuring all documentation is fully recorded.
(f)You represent and warrant that there are no outstanding charges, complaints, claims, grievances or actions of any nature whatsoever previously filed or brought by you or on your behalf against any of the Released Parties pending before any federal, state or local court or administrative body. You also represent and warrant that you have provided Pear with written notice of any and all concerns regarding suspected ethical and compliance issues or violations including, without limitation, anything covered by the Code of Business Conduct and Ethics, on the part of Pear, other employees, or any Released Parties.
(g)You acknowledge that the Separation Pay and Benefits provided for herein are being offered based on your representations that you have not engaged in any fraudulent or unlawful conduct, and that you have fully disclosed to the Company all material information relating to your job duties for the Company.
(h)Any breach of this Section 9 including, without limitation, your obligations under the Covenants Agreement, will constitute a material breach of this Agreement and, in addition to any other legal or equitable remedy available to Pear, will entitle Pear to withhold payment of or recover any monies or other consideration (including rights to a further tranche of vesting of RSUs) provided to you or expended on your behalf under Section 3 of this Agreement.
10.Release of Claims.
(a)You hereby acknowledge and agree that by signing this Agreement and accepting the pay and benefits provided for in this Agreement, you are waiving your right to assert any Claim (as defined below) against the Company any and all of its investors, advisors, affiliates and
subsidiaries, and all entities related to any of the foregoing, and each of the directors, officers, employees, agents, successors and assigns of any of the foregoing (collectively, the “Released Parties”) arising from acts or omissions that occurred on or before the date on which you sign this Agreement. You agree that you are making this release of Claims on behalf of yourself, your representatives, agents, estate, heirs, attorneys, insurers, servants, spouse, executors, administrators, successors, and assigns, and any other person, entity, and (to the extent allowed by law) government agency acting on your behalf. Your waiver and release is intended to bar any form of legal claim, lawsuit, charge, complaint or any other form of action (jointly referred to as “Claims”) against the Released Parties seeking money or any other form of relief, including but not limited to equitable relief (whether declaratory, injunctive or otherwise), damages or any other form of monetary recovery (including but not limited to back pay, front pay, compensatory damages, emotional distress damages, punitive damages, attorneys’ fees and any other costs). You understand that there could be unknown or unanticipated Claims resulting from your employment with Pear and the termination thereof and you agree that such Claims are intended to be, and are, included in this waiver and release. You also understand that your waiver and release includes all Claims under any statute, ordinance, regulation, executive order, common law, constitution and/or other source of law or any state, country and/or locality, including but not limited to the United States, the State of Nevada, the Commonwealth of Massachusetts, the State of California, the State of North Carolina, and/or any other state or locality where you worked for the Company (collectively and individually referred to as “Law”).
Without limiting the foregoing general waiver and release, except for Claims resulting from the failure of the Company to perform its obligations under this Agreement, you specifically waive and release the Released Parties from any Claims arising from or related to your employment relationship with the Company or the termination thereof, including without limitation: (i) Claims under any Law concerning discrimination, harassment, retaliation, or other fair employment practices, including but not limited to the Massachusetts Anti-Discrimination and Anti- Harassment Law (M.G.L. Chapter 151B), the Massachusetts Sexual Harassment Law (M.G.L. c. 214 § 1C), the Massachusetts Equal Pay Act (M.G.L. c. 149 § 105A), the Massachusetts Civil Rights Act (M.G.L. c. 12, §§ 11H, 111), the Massachusetts Equal Rights Act (M.G.L, c. 93, §§ 102, 103), the California Fair Employment and Housing Act (Cal. Gov. Code, Title 2, Division 3, Part 2.8), the California Constitution, the California Labor Code (including all laws and regulations relating to payment of wages, overtime or any other compensation or benefit), the California Family Rights Act (Cal. Gov. Code §§ 12945.2 and 19702.3), the Age Discrimination in Employment Act (29 U.S.C. § 621 et seq.), Title VII of the Civil Rights Act of 1964 (42 U.S.C. § 2000e et seq.), 42 U.S.C. § 1981, the Americans with Disabilities Act (42 U.S.C. § 12101 et seq.), the Equal Pay Act of 1963, the Employee Retirement Income Security Act (ERISA), the Age Discrimination in Employment Act (ADEA), the Older Workers Benefit Protection Act (OWBPA), the Occupational Health and Safety Act of 1970 (OSHA), the Fair Credit Reporting Act (FCRA), the Worker Adjustment and Retraining Notification (WARN) Act, the Genetic Information Nondiscrimination Act (GINA), the Uniform Services Employment and Reemployment Rights Act (USERRA), each as they may have been amended through the date you sign this Agreement; (ii) Claims under any Law relating to wages, hours, whistleblowing, leaves of absence or any other terms and conditions of employment, including, but not limited to, the Massachusetts Payment of Wages Law (M.G.L. c. 149 §§ 148 and 150), Massachusetts General Laws Chapter 149 in its entirety, Massachusetts General Laws Chapter 151 in its entirety
(including but not limited to the minimum wage and other Obligations to be immediately due and payable, imposing a default rate of interest in respect of the Obligations in accordance with Section 3.02(b) of the Amended and Restated Credit Agreement or pursuing any or all other rights and remedies available to any Secured Party as a secured party under the UCC, the Security Documentsovertime provisions), the California Labor Code (including all laws and regulations relating to payment of wages, overtime or any other Loan Document.
ARTICLE II
CONDITIONS PRECEDENT
Section 2.01. Conditions to Effectiveness of this Agreement. The amendments and other modifications set forth in this Agreement shall become effective upon the prior or simultaneous satisfaction of each of the following conditions in a manner reasonably satisfactory to the Administrative Agent (the date when all such conditions are so satisfied being the “Effective Date”):
(a) Counterparts. The Administrative Agent shall have received counterparts of (i) this Agreement, (ii) the Amended and Restated Credit Agreement and (iii) each other Loan Document required to be delivered pursuant to Section 6.03 of the Amended and Restated Credit Agreement as a condition precedent to the effectiveness thereof, in each case executed by each party thereto.
(b) Amended and Restated Credit Agreement Conditions. All conditions to effectiveness set forth in Section 6.02 of the Amended and Restated Credit Agreement shall have been satisfied.
(c) Costs and Expenses, etc. The Agent shall have received all fees, costs and expenses due and payable pursuant to Section 14.03 of the Amended and Restated Credit Agreement (including without limitation the reasonable fees and expenses of Morrison & Foerster LLP, counsel to the Administrative Agent, which have been invoiced prior to the Effective Date), together with any other fees separately agreed to by the Borrower and the Agent and the Lenders.
(d) Satisfactory Legal Form. All documents executed or submitted pursuant hereto by or on behalf of the Borrower or any of its Subsidiaries, shall be reasonably satisfactory in form and substance to the Agent and its counsel, and the Agent and its counsel shall have received all
information, approvals, resolutions, opinions, documents or instruments as the Agent or its counsel shall have reasonably requested.
ARTICLE III
REPRESENTATIONS AND WARRANTIES
Section 3.01. Representations and Warranties. To induce the Administrative Agent and the Majority Lender to enter into this Agreement, each Obligor party hereto represents and warrants to the Administrative Agent and the Majority Lender as set forth below.
(a) Validity, etc. This Agreement and each Loan Document (after giving effect to this Agreement on the Effective Date) each constitutes the legal, valid and binding obligation of each Obligor party thereto, enforceable in accordance with its respective terms, subject to the effects of bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium and other similar laws relating to or affecting creditors’ rights generally, general equitable principles (whether considered in a proceeding in equity or at law) and an implied covenant of good faith and fair dealing.
(b) Representations and Warranties, etc. Immediately prior to, and immediately after giving effect to this Agreement on the Effective Date, the following statements shall be true and correct:
(i) the representations and warranties set forth in the Amended and Restated Credit Agreement and each other Loan Document that are qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct; and
(ii) the representations and warranties set forth in the Amended and Restated Credit Agreement and each other Loan Document that are not qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct in all material respects.
(c) Authorizations and Approvals. No authorization or approval or other action by, and no notice or filing with, any Governmental Authority or any other Person (other than those that have been duly obtained or made and which are in full force and effect) is required for the due execution and delivery of this Agreement and performance of this Agreement and the Amended and Restated Credit Agreement (as amended hereby) by any Obligor party to this Agreement.
(d) No Defaults. No Default or Event of Default shall have occurred and be continuing.
ARTICLE IV
CONFIRMATION AND COVENANTS
Section 4.01. Reaffirmation. Each Obligor hereby consents to the amendment and other agreements and modifications to the Credit Agreement and other Loan Documents contemplated pursuant to this Agreement and hereby agrees that, after giving effect to this Agreement on the Effective Date, the Credit Agreement and each other Loan Document, and all Obligations thereunder (including the guarantees made pursuant to any Guaranty), are and shall continue to be in full force and effect and the same are hereby ratified in all respects, except that upon the occurrence of the Effective Date, all references in such Loan Documents to the “Credit
Agreement”, “Loan Documents”, “thereunder”, “thereof”, or words of similar import shall mean the Amended and Restated Credit Agreement and the other Loan Documents, as amended or otherwise modified by this Agreement. Each Obligor further consents and agrees that, notwithstanding the effectiveness of this Agreement, all Liens and security interests granted and created under or pursuant to any Loan Document shall continue to be valid and perfected to the same extent as immediately prior to the Effective Date.
Section 4.02. Validity, etc. Each Obligor hereby represents and warrants, that immediately after giving effect to this Agreement on the Effective Date, each Loan Document to which it is a party continues to be a legal, valid and binding obligation of such Obligor, enforceable against such Person in accordance with its terms, subject to the effects of bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium and other similar laws relating to or affecting creditors’ rights generally, general equitable principles (whether considered in a proceeding in equity or at law) and an implied covenant of good faith and fair dealing.
ARTICLE V
MISCELLANEOUS
Section 5.01. Miscellaneous, etc. In addition to the foregoing, the parties hereto agree as follows:
(a) Severability. In case any provision of or obligation under this Agreement shall be invalid, illegal or unenforceable in any jurisdiction, the validity, legality and enforceability of the remaining provisions or obligations, or of such provision or obligation in any other jurisdiction, shall not in any way be affected or impaired thereby.
(b) Integration. This Agreement, together with the other Loan Documents, incorporates all negotiations of the parties hereto with respect to the subject matter hereof and is the final expression and agreement of the parties hereto with respect to the subject matter hereof and supersedes any and all prior agreements, written or oral, with respect thereto.
(c) Cross-References; Headings. References in this Agreement to any Article or Section are, unless otherwise specified, to such Article or Section of this Agreement. Headings and captions used in this Agreement are included for convenience of reference only and shall not be given any substantive effect.
(d) Loan Document Pursuant to Credit Agreement. This Agreement shall constitute a Loan Document executed pursuant to the Credit Agreement and shall (unless otherwise expressly indicated therein) be construed, administered and applied in accordance with all of the terms and provisions of the Amended and Restated Credit Agreement, including Article XIV thereof and all rules of interpretation set forth in Article I thereof. The failure of any Obligor to perform its obligations hereunder shall constitute a Default pursuant to Section 11.01(d) of the Amended and Restated Credit Agreement.
(e) Successors and Assigns. This Agreement shall be binding upon and inure to the benefitcompensation or benefit), the California Family Rights Act (Cal. Gov. Code §§ 12945.2), the Family and Medical Leave Act of 1993 (29 U.S.C. § 2601 et seq.), all applicable federal, state or local laws relating to sick time and/or other family or medical leave, each as they may have been amended through the date you sign this Agreement. You specifically acknowledge that you are waiving any Claims for unpaid wages under these and other Laws; (iii) Claims under any local, state or federal common law theory; (iv) Claims arising under the Company’s policies or benefit plans, including, without limitation, any Claims under the Company’s Severance and Change in Control Plan adopted by the Company’s Board of Directors on June 14, 2022, and (v) Claims arising under other Law.
(b)In addition to the foregoing, you hereby agree that you are waiving all rights under section 1542 of the Civil Code of the State of California. Section 1542 provides that: “A general release does not extend to claims that the creditor or releasing party does not know or suspect to exist in his or her favor at the time of executing the release and that, if known by him or her, would have materially affected his or her settlement to the debtor or released party.” Pursuant to Section 1542, you acknowledge that you may hereafter discover facts different from or in addition to facts which you now know or believe to be true with regard to the released claims, and further agree that this Agreement shall remain effective in all respects notwithstanding such discovery of new or different facts, including any such facts which may give rise to currently unknown claims, including but not limited to any claims or rights which you may have under Section 1542 of the California Civil Code or similar law or doctrine of any other state.
(c)You acknowledge and agree that, but for providing this waiver and release, you would not be receiving the Separation Pay and Benefits provided for in this Agreement.
(d)Because you are at least forty (40) years of age, you have specific rights under the federal Age Discrimination in Employment Act (“ADEA”) and Older Workers Benefits Protection Act (“OWBPA”), which prohibit discrimination on the basis of age. The release in Section 10 is intended to release any Claim you may have against the Company alleging discrimination on the basis of age under the ADEA, OWBPA and other Laws. Notwithstanding anything to the contrary in this Agreement, the release in Section 10 does not cover rights or Claims under the ADEA that arise from acts or omissions that occur after the date you sign this Agreement. In addition, consistent with the ADEA and OWBPA, the Company is providing you with the time to review and accept this Agreement and the ability to revoke your acceptance as described in Section 11(h) below.
(e)This release does not apply to any claims concerning a breach of this Agreement, claims for any vested ERISA benefits under employee benefit plans of the Company, any rights to benefits under applicable workers’ compensation statutes or government-provided unemployment benefits, or any claims wherein the cause of action arises after the date you sign this Agreement. Further, consistent with federal discrimination laws, nothing in this release shall be deemed to prohibit you from challenging the validity of this release under federal discrimination laws or from filing a charge or complaint of employment related discrimination with the Equal Employment Opportunity Commission (“EEOC”), or from participating in any investigation or proceeding
conducted by the EEOC. However, the release in Section 10 does prohibit you from seeking or receiving monetary damages or other individual-specific relief in connection with any such charge or complaint of work-related discrimination. Further, nothing in this release or Agreement shall be deemed to limit any Released Party’s right to seek immediate dismissal of such charge or complaint on the basis that your signing of this Agreement constitutes a full release of any individual rights under federal discrimination laws, or any Released Party’s right to seek restitution or other legal remedies to the extent permitted by law of the economic benefits provided to you under this Agreement in the event that you successfully challenge the validity of this release and prevail in any claim under federal discrimination laws.
11.Miscellaneous.
(a)Successors and Assigns. This Agreement shall be binding upon the respective legal representatives, heirs and successors of the parties, hereto and their respective successors andto the extent permitted assigns.
(b)Notices. Any notices required to be given in connection with this Agreement shall be given by certified mail to Ronan O’Brien, General Counsel, Pear Therapeutics, Inc., 200 State Street, Floor #13, Boston, MA 02109 with a copy via email to ronan.obrien@peartherapeutics.com.
(f) c)Counterparts, Electronic Signatures, etc. This Agreement may be executed in any numberEntire Agreement. You acknowledge that this Agreement sets forth the entire agreement between you and Pear concerning your separation and, except as expressly provided for herein (e.g., your obligations set forth in the agreement referenced in Section 9(b)), and except for the Plan, fully supersedes any and all prior agreements or understanding between you and Pear pertaining to the subject matter thereof. This Agreement may only be modified in a written document signed by you and an authorized representative of Pear. In the event any provision of this Agreement is held invalid, all remaining provisions of the Agreement shall remain in full force and effect.
(d)Severability. Should any provision of this Agreement be declared or be determined by any court of competent jurisdiction to be illegal or invalid, the validity of the remaining parts, terms or provisions shall not be affected thereby and said illegal or invalid part, term or provision shall be deemed not to be a part of this Agreement.
(e)Execution. This Agreement may be executed in one or ofmore counterparts, alleach of which takenwhen so executed shall be deemed to be an original, and all such counterparts together shall constitute but one and the same instrument and any of the parties hereto may execute this Agreement by signing any such counterpart. Delivery of an executed. Your signature page of this Agreement by facsimile transmission or electronic transmission (in PDF format) shall be effective as delivery of a manually executed counterpart hereof. Any signature (including, without limitation, (x) any electronic symbol or process attached to, or associated with, a contract or other record and adopted by a Person with the intent to sign, authenticate or accept such contract or record and (y) any facsimile transmission or PDF format signature) hereto or to any other certificate, agreement or document related to the transactions contemplated hereby, and any contract formation or record-keeping, in each case, through electronic means, shall have the same legal validity and enforceability as a manually executed signature or use of a paper-based record-keeping system to the fullest extent permitted by applicable law, including the Federal Electronic Signatures in Global and National Commerce Act, the New York State Electronic Signatures and Records Act, or any similar state law based on the Uniform Electronic Transactions Act, and the parties hereto hereby waive any objection to the contrary.
(g) Governing Law. THIS AGREEMENT AND ANY CLAIMS, CONTROVERSY, DISPUTE OR CAUSE OF ACTION (WHETHER IN CONTRACT OR TORT OR OTHERWISE) BASED UPON, ARISING OUT OF OR RELATING TO THIS AGREEMENT SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH, THE INTERNAL LAWS OF THE STATE OF NEW YORK (INCLUDING FOR SUCH PURPOSE SECTIONS 5-1401 AND 5-1402 OF THE GENERAL OBLIGATIONS LAW OF THE STATE OF NEW YORK).
[Signature pages to follow]
IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be duly executed and delivered as of the day and year first above written.
| | | | | | | | |
| HOLDINGS: |
| | |
| PEAR THERAPEUTICS, INC. |
| By | /s/ Ronan O’Brien |
| Name: Ronan O’Brien |
| Title: Secretary |
| | |
| | |
| BORROWER: |
| | |
| PEAR THERAPEUTICS (US), INC. |
| | |
| By | /s/ Ronan O’Brien |
| Name: Ronan O’Brien |
| Title: General Counsel & Chief Compliance Officer |
| | | | | | | | |
| ADMINISTRATIVE AGENT AND LENDER: |
| | |
| PERCEPTIVE CREDIT HOLDINGS III, LP |
| | |
By: PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner |
| | |
| | |
| By | /s/ Sandeep Dixit |
| Name: | Sandeep Dixit |
| Title: | Chief Credit Officer |
| | |
| | |
| By | /s/ Sam Chawla |
| Name: | Sam Chawla |
| Title: | Portfolio Manager |
EXHIBIT A
Amended and Restated Credit Agreement
(See attached.)
AMENDED AND RESTATED CREDIT AGREEMENT AND GUARANTY
dated as of
March 25, 2022
by and among
PEAR THERAPEUTICS, INC.,
as Holdings,
PEAR THERAPEUTICS (US), INC.,
as the Borrower,
THE SUBSIDIARY GUARANTORS FROM TIME TO TIME PARTY HERETO,
as the Subsidiary Guarantors,
THE LENDERS FROM TIME TO TIME PARTY HERETO,
as the Lenders,
and
PERCEPTIVE CREDIT HOLDINGS III, LP
as the Administrative Agent
U.S. $50,000,000
| | | | | | | | | | | |
| | | Page |
| | | |
| SECTION 1 DEFINITIONS | 1 |
| | | |
| 1.01 | Certain Defined Terms | 1 |
| | | |
| 1.02 | Accounting Terms and Principles | 30 |
| | | |
| 1.03 | Interpretation | 31 |
| | | |
| 1.04 | Divisions | 32 |
| | | |
| SECTION 2 THE COMMITMENTS AND THE LOANS | 32 |
| | | |
| 2.01 | Loans | 32 |
| | | |
| 2.02 | Borrowing Procedures | 32 |
| | | |
| 2.03 | Notes | 32 |
| | | |
| 2.04 | Use of Proceeds | 32 |
| | | |
| SECTION 3 PAYMENTS OF PRINCIPAL AND INTEREST | 33 |
| | | |
| 3.01 | Repayments and Prepayments Generally; Application | 33 |
| | | |
| 3.02 | Interest | 33 |
| | | |
| 3.03 | Prepayments; Early Prepayment Fee | 33 |
| | | |
| 3.04 | Closing Fee | 35 |
| | | |
| SECTION 4 PAYMENTS, ETC. | 35 |
| | | |
| 4.01 | Payments | 35 |
| | | |
| 4.02 | Computations | 36 |
| | | |
| 4.03 | Set-Off | 36 |
| | | |
| SECTION 5 YIELD PROTECTION, ETC. | 37 |
| | | |
| 5.01 | Additional Costs | 37 |
| | | |
| 5.02 | Illegality | 38 |
| | | |
| 5.03 | Taxes | 38 |
| | | |
| 5.04 | Mitigation Obligations | 42 |
| | | |
| SECTION 6 CONDITIONS PRECEDENT | 42 |
| | | |
| 6.01 | Conditions to the Borrowing of the Tranche 1 Loans | 42 |
| | | |
| 6.02 | Conditions to Effectiveness | 47 |
| | | |
| SECTION 7 REPRESENTATIONS AND WARRANTIES | 49 |
| | | |
| 7.01 | Power and Authority | 49 |
| | | |
| 7.02 | Authorization; Enforceability | 49 |
| | | |
TABLE OF CONTENTS
(continued)
| | | | | | | | | | | |
| | | Page |
| | | |
| 7.03 | Governmental and Other Approvals; No Conflicts | 50 |
| | | |
| 7.04 | Financial Statements; Material Adverse Change | 50 |
| | | |
| 7.05 | Properties | 50 |
| | | |
| 7.06 | No Actions or Proceedings | 54 |
| | | |
| 7.07 | Compliance with Laws and Agreements | 55 |
| | | |
| 7.08 | Taxes | 56 |
| | | |
| 7.09 | Full Disclosure | 56 |
| | | |
| 7.10 | Investment Company Act and Margin Stock Regulation | 57 |
| | | |
| 7.11 | Solvency | 57 |
| | | |
| 7.12 | Equity Holders, Subsidiaries and Other Investments | 57 |
| | | |
| 7.13 | Indebtedness and Liens | 57 |
| | | |
| 7.14 | Material Agreements | 57 |
| | | |
| 7.15 | Restrictive Agreements | 58 |
| | | |
| 7.16 | Real Property | 58 |
| | | |
| 7.17 | Pension Matters | 58 |
| | | |
| 7.18 | Priority of Obligations; Collateral; Security Interest | 58 |
| | | |
| 7.19 | Regulatory Approvals | 59 |
| | | |
| 7.20 | Transactions with Affiliates | 59 |
| | | |
| 7.21 | Sanctions | 59 |
| | | |
| 7.22 | Anti-Corruption | 59 |
| | | |
| 7.23 | Deposit and Disbursement Accounts | 60 |
| | | |
| 7.24 | Royalties and Other Payments | 60 |
| | | |
| 7.25 | Non-Competes | 60 |
| | | |
| 7.26 | Internal Controls | 60 |
| | | |
| SECTION 8 AFFIRMATIVE COVENANTS | 60 |
| | | |
| 8.01 | Financial Statements and Other Information | 60 |
| | | |
| 8.02 | Notices of Material Events | 62 |
| | | |
| 8.03 | Existence; Conduct of Business | 64 |
| | | |
| 8.04 | Payment of Obligations | 64 |
| | | |
| 8.05 | Insurance | 65 |
| | | |
TABLE OF CONTENTS
(continued)
| | | | | | | | | | | |
| | | Page |
| | | |
| 8.06 | Books and Records; Inspection Rights | 65 |
| | | |
| 8.07 | Compliance with Laws and Other Obligations | 65 |
| | | |
| 8.08 | Maintenance of Properties, Etc | 65 |
| | | |
| 8.09 | Licenses | 66 |
| | | |
| 8.10 | Action under Environmental Laws | 66 |
| | | |
| 8.11 | Use of Proceeds | 66 |
| | | |
| 8.12 | Certain Obligations Respecting Subsidiaries; Further Assurances | 66 |
| | | |
| 8.13 | Termination of Non-Permitted Liens | 68 |
| | | |
| 8.14 | Intellectual Property | 68 |
| | | |
| 8.15 | Maintenance of Regulatory Approvals, Contracts, Intellectual Property, Etc | 68 |
| | | |
| 8.16 | ERISA and Foreign Pension Plan Compliance | 68 |
| | | |
| 8.17 | Cash Management | 68 |
| | | |
| 8.18 | Litigation Cooperation | 69 |
| | | |
| 8.19 | Board Observation Rights | 69 |
| | | |
| SECTION 9 NEGATIVE COVENANTS | 70 |
| | | |
| 9.01 | Indebtedness | 70 |
| | | |
| 9.02 | Liens | 72 |
| | | |
| 9.03 | Fundamental Changes, Acquisitions, Etc | 73 |
| | | |
| 9.04 | Lines of Business | 74 |
| | | |
| 9.05 | Investments | 74 |
| | | |
| 9.06 | Restricted Payments | 75 |
| | | |
| 9.07 | Payments of Indebtedness | 76 |
| | | |
| 9.08 | Change in Fiscal Year | 76 |
| | | |
| 9.09 | Sales of Assets, Etc | 76 |
| | | |
| 9.10 | Transactions with Affiliates | 77 |
| | | |
| 9.11 | Restrictive Agreements | 77 |
| | | |
| 9.12 | Modifications and Terminations of Material Agreements and Organic Documents | 77 |
| | | |
| 9.13 | Sales and Leasebacks | 78 |
| | | |
| 9.14 | Hazardous Material | 78 |
| | | |
TABLE OF CONTENTS
(continued)
| | | | | | | | | | | |
| | | Page |
| | | |
| 9.15 | Accounting Changes | 78 |
| | | |
| 9.16 | Compliance with ERISA | 78 |
| | | |
| 9.17 | Sanctions; Anti-Corruption Use of Proceeds | 78 |
| | | |
| 9.18 | Inbound and Outbound Licenses | 78 |
| | | |
| 9.19 | Activities of the Massachusetts Securities Subsidiary | 79 |
| | | |
| SECTION 10 FINANCIAL COVENANTS | 79 |
| | | |
| 10.01 | Minimum Liquidity | 79 |
| | | |
| 10.02 | Minimum Revenue | 79 |
| | | |
| SECTION 11 EVENTS OF DEFAULT | 80 |
| | | |
| 11.01 | Events of Default | 80 |
| | | |
| 11.02 | Remedies | 83 |
| | | |
| 11.03 | Additional Remedies | 84 |
| | | |
| SECTION 12 THE ADMINISTRATIVE AGENT | 84 |
| | | |
| 12.01 | Appointment and Duties | 84 |
| | | |
| 12.02 | Binding Effect | 85 |
| | | |
| 12.03 | Use of Discretion | 86 |
| | | |
| 12.04 | Delegation of Rights and Duties | 86 |
| | | |
| 12.05 | Reliance and Liability | 86 |
| | | |
| 12.06 | Administrative Agent Individually | 87 |
| | | |
| 12.07 | Lender Credit Decision | 87 |
| | | |
| 12.08 | Expenses; Indemnities | 87 |
| | | |
| 12.09 | Resignation of the Administrative Agent | 88 |
| | | |
| 12.10 | Release of Collateral or Guarantors | 89 |
| | | |
| 12.11 | Additional Secured Parties | 89 |
| | | |
| SECTION 13 GUARANTEE | 90 |
| | | |
| 13.01 | The Guarantee | 90 |
| | | |
| 13.02 | Obligations Unconditional | 90 |
| | | |
| 13.03 | Reinstatement | 91 |
| | | |
| 13.04 | Subrogation | 91 |
| | | |
| 13.05 | Remedies | 91 |
| | | |
TABLE OF CONTENTS
(continued)
| | | | | | | | | | | |
| | | Page |
| | | |
| 13.06 | Instrument for the Payment of Money | 92 |
| | | |
| 13.07 | Continuing Guarantee | 92 |
| | | |
| 13.08 | General Limitation on Guarantee Obligations | 92 |
| | | |
| SECTION 14 MISCELLANEOUS | 92 |
| | | |
| 14.01 | No Waiver | 92 |
| | | |
| 14.02 | Notices | 92 |
| | | |
| 14.03 | Expenses, Indemnification, Etc | 93 |
| | | |
| 14.04 | Amendments, Etc | 94 |
| | | |
| 14.05 | Successors and Assigns | 94 |
| | | |
| 14.06 | Survival | 97 |
| | | |
| 14.07 | Captions | 97 |
| | | |
| 14.08 | Counterparts; Electronic Signatures | 97 |
| | | |
| 14.09 | Governing Law | 97 |
| | | |
| 14.10 | Jurisdiction, Service of Process and Venue | 97 |
| | | |
| 14.11 | Waiver of Jury Trial | 98 |
| | | |
| 14.12 | Waiver of Immunity | 98 |
| | | |
| 14.13 | Entire Agreement | 98 |
| | | |
| 14.14 | Severability | 99 |
| | | |
| 14.15 | No Fiduciary Relationship | 99 |
| | | |
| 14.16 | Confidentiality | 99 |
| | | |
| 14.17 | Interest Rate Limitation | 100 |
| | | |
| 14.18 | Early Prepayment Fee | 100 |
| | | |
| 14.19 | Judgment Currency | 100 |
| | | |
| 14.20 | USA PATRIOT Act | 101 |
| | | |
| 14.21 | Acknowledgement and Consent to Bail-In of EEA Financial Institutions | 101 |
| | | |
| 14.22 | Amendment and Restatement; No Novation | 101 |
SCHEDULES AND EXHIBITS
| | | | | | | | |
| Schedule 1 | - | Commitments |
| Schedule 2 | - | Deposit Accounts, Securities Accounts and Commodity Accounts of the Massachusetts Securities Subsidiary |
| Schedule 7.05(b) | - | Products |
| Schedule 7.05(c) | - | Obligor Intellectual Property |
| Schedule 7.06(a) | - | Certain Litigation |
| Schedule 7.06(c) | - | Labor Matters |
| Schedule 7.07(d) | - | Healthcare Law Matters |
| Schedule 7.08 | - | Taxes |
| Schedule 7.12(a) | - | Holders of Equity Interests of the Borrower |
| Schedule 7.12(b) | - | Subsidiaries of the Borrower |
| Schedule 7.12(c) | - | Other Equity Interests Owned by Borrower or its Subsidiaries |
| Schedule 7.13(a) | - | Existing Indebtedness |
| Schedule 7.13(b) | - | Existing Indebtedness to be Repaid on the Closing Date |
| Schedule 7.13(c) | - | Existing Liens |
| Schedule 7.14 | - | Material Agreements |
| Schedule 7.15 | - | Restrictive Agreements |
| Schedule 7.16 | - | Real Property |
| Schedule 7.17 | - | Pension Matters |
| Schedule 7.19(b) | - | Regulatory Approvals |
| Schedule 7.20 | - | Transactions with Affiliates |
| Schedule 7.23 | - | Deposit and Disbursement Accounts |
| Schedule 7.24 | - | Royalties and Other Payments |
| Schedule 7.25 | - | Non-Competes |
| Schedule 9.05 | - | Existing Investments |
| Schedule 9.13 | - | Permitted Sales and Leasebacks |
| | |
| Exhibit A | - | Form of Note |
| Exhibit B | - | Form of Borrowing Notice |
| Exhibit C | - | Form of Guarantee Assumption Agreement |
| Exhibit D-1 | - | Form of U.S. Tax Compliance Certificate (For Foreign Lenders That Are Not Partnerships for U.S. Federal Income Tax Purposes) |
| Exhibit D-2 | - | Form of U.S. Tax Compliance Certificate (For Foreign Participants That Are Not Partnerships for U.S. Federal Income Tax Purposes) |
| Exhibit D-3 | - | Form of U.S. Tax Compliance Certificate (For Foreign Participants That Are Partnerships for U.S. Federal Income Tax Purposes) |
| Exhibit D-4 | - | Form of U.S. Tax Compliance Certificate (For Foreign Lenders That Are Partnerships for U.S. Federal Income Tax Purposes) |
| Exhibit E | - | Form of Compliance Certificate |
| Exhibit F | - | Form of Assignment and Assumption |
| Exhibit G | - | Form of Information Certificate |
| Exhibit H | - | Form of Intercompany Subordination Agreement |
| Exhibit I | - | Form of Solvency Certificate |
AMENDEDED AND RESTATED CREDIT AGREEMENT AND GUARANTY
This Amended and Restated Credit Agreement and Guaranty, dated as of March 25, 2022 (this “Agreement”), by and among (i) Pear Therapeutics, Inc., a Delaware corporation (f/k/a Thimble Point Acquisition Corp.) (“Holdings”), (ii) Pear Therapeutics (US), Inc., a Delaware corporation (f/k/a Pear Therapeutics, Inc.) (the “Borrower”), whose direct parent is Holdings as of the date hereof, (iii) certain Subsidiaries of Holdings that may be required to provide Guarantees from time to time hereunder, (iv) Perceptive Credit Holdings III, LP (the “Closing Date Lender”) and each other lender that may from time to time become a party hereto (each, including the Closing Date Lender, a “Lender” and collectively, the “Lenders”), and (v) Perceptive Credit Holdings III, LP, as administrative agent for the Lenders (in such capacity, the “Administrative Agent”).
WITNESSETH:
WHEREAS, pursuant to the Credit Agreement and Guaranty, dated as of June 30, 2020, by and among the Borrower, the Subsidiary guarantors party thereto, the Lenders party thereto and the Administrative Agent (as amended or otherwise modified prior to the Effective Date, the “Original Credit Agreement”), the Lenders have provided a senior secured delayed draw term loan facility to the Borrower in an aggregate principal amount of $50,000,000, with up to $30,000,000 in aggregate principal amount of Loans to be available on the Closing Date, up to $10,000,000 in aggregate principal amount of Loans to be available after the Closing Date but prior to December 31, 2021, and up to $10,000,000 in aggregate principal amount of Loans to be available after the Closing Date but prior to December 31, 2021, of which, as of the date hereof, the Borrower has borrowed $30,000,000 in aggregate principal amount of Loans; and
WHEREAS, pursuant to the Third Amendment to Credit Agreement and Guaranty, dated as of the date hereof (the “Restatement Amendment”), Holdings, the Borrower, the Administrative Agent and the Lenders have agreed, subject to the terms and conditions set forth herein and in the Restatement Amendment, to enter into this Agreement and to amend and restate the Original Credit Agreement in its entirety as set forth herein.
NOW, THEREFORE, the parties hereto agree as follows:
SECTION 1
DEFINITIONS
1.01 Certain Defined Terms. As used herein (including the preamble and recitals), the following terms have the following respective meanings:
“510(k)” means (i) any premarket notification and corresponding FDA clearance for a Device pursuant to FDA regulations, (ii) all substantially equivalent or similar notifications, applications and clearances with respect to any non-U.S. Regulatory Authority, including the EMA, and (iii) all amendments, supplements and other additions and modifications thereto, and all documents, data and other information concerning any applicable Device which are necessary for, filed with, incorporated by reference in or otherwise support any of the foregoing.
“Acquisition” means any transaction, or any series of related transactions, by which any Person directly or indirectly, by means of an amalgamation, consolidation, merger, purchase of Equity Interests or other assets, tender offer, or similar transaction having the same effect as any of the foregoing, (i) acquires any business or all or substantially all of the assets of any other Person, (ii) acquires all or substantially all of a business line or unit or division of any other Person, (iii) acquires control of Equity Interests of another Person representing more than fifty percent (50%) of the ordinary voting power for the election of directors or other governing body if the business affairs of such Person are managed by a board of directors or other governing body, determined on a fully-diluted, as-if-converted or exercised basis, or (iv) acquires control of more than fifty percent (50%) of the Equity Interests in any Person engaged in any business that is not managed by a board of directors or other governing body, determined on a fully-diluted, as-if-converted or exercised basis.
“Administrative Agent” has the meaning set forth in the preamble hereto.
“Advanced Development” means, with respect to a proposed Product, that such proposed Product is in either a pivotal clinical trial or later stage of development.
“Adverse Regulatory Event” means the occurrence of any of the following events or circumstances:
(a) the failure of Holdings or any of its Subsidiaries to hold, directly or through licensees or agents, in full force and effect, all Regulatory Approvals necessary or required for Holdings, the Borrower or any such Subsidiary to conduct its respective operations and businesses;
(b) if required by any applicable Law, the failure of Holdings or any of its Subsidiaries to make or file with the FDA or any other applicable Regulatory Authority, in compliance with such applicable Law, any required notice, registration, listing, supplemental application or notification or report;
(c) in connection with any clinical, preclinical, safety or other studies or tests being conducted by (or on behalf of) Holdings or any of its Subsidiaries for purposes of obtaining regulatory clearance of, or any Regulatory Approval for, any Product or any Product Commercialization and Development Activities (i) the failure of any clinical, pre-clinical, safety or other required trial, study or test to be conducted in material compliance with any applicable Law or Regulatory Approval; (ii) the failure of any related clinical trial site to be monitored in material compliance with all applicable Laws and Regulatory Approvals; or (iii) the receipt by Holdings or any of its Subsidiaries of written notice from the FDA or any other Regulatory Authority requiring the termination or suspension of any such clinical, preclinical, safety or other study or test;
(d) Holdings or any of its Subsidiaries or, to the knowledge of Holdings, the Borrower, any agent, supplier, licensor or licensee of Holdings, the Borrower or any of its Subsidiaries, receives from any Regulatory Authority any written notice with respect to any Product or any Product Commercialization and Development Activities from any Regulatory Authority asserting (i) that such Person lacks a required Regulatory Approval
with respect to such Product or Product Commercialization and Development Activity, (ii) a material lack of compliance by such Person with any applicable Laws or Regulatory Approvals (or any similar order, injunction or decree) or (iii) that such Regulatory Authority has commenced any regulatory action, investigation or inquiry (other than non-material routine or periodic inspections or reviews) with respect to any Product or any Product Commercialization and Development Activities, in each case other than items contested in good faith by the Borrower by appropriate action; or
(e) with respect to any Product or Product Commercialization and Development Activity, (i) any Regulatory Authority commences any criminal, injunctive, seizure, detention or civil penalty action or (ii) Holdings, the Borrower or any Subsidiary enters into any consent decree, plea agreement or other settlement with any Regulatory Authority with respect to any of the foregoing.
“Affected Financial Institution” means (a) any EEA Financial Institution or (b) any UK Financial Institution.
“Affiliate” means, with respect to a specified Person, another Person that directly, or indirectly through one or more intermediaries, Controls or is Controlled by or is under common Control with the Person specified; provided that with respect to any Lender, an Affiliate of such Lender shall include, without limitation, all of such Lender’s Related Funds.
“Agreement” has the meaning set forth in the preamble hereto.
“Amendment No. 2” means that certain Amendment No. 2 to Credit Agreement, dated as of December 3, 2021, among the Borrower, certain Subsidiaries of the Borrower, the Majority Lenders and the Administrative Agent for the Majority Lender.
“Amendment No. 2 Effective Date” means December 3, 2021.
“Applicable Margin” means eleven percent (11.0%), as such percentage may be increased pursuant to Section 3.02(b).
“Asset Sale” has the meaning set forth in Section 9.09.
“Assignment and Assumption” means an assignment and assumption entered into by a Lender and an assignee of such Lender in substantially the form of Exhibit F.
“Bail-In Action” means the exercise of any Write-Down and Conversion Powers by the applicable Resolution Authority in respect of any liability of an Affected Financial Institution.
“Bail-In Legislation” means, (a) with respect to any EEA Member Country implementing Article 55 of Directive 2014/59/EU of the European Parliament and of the Council of the European Union, the implementing law for such EEA Member Country from time to time which is described in the EU Bail-In Legislation Schedule and (b) with respect to the United Kingdom, Part I of the United Kingdom Banking Act 2009 (as amended from time to time) and any other law, regulation or rule applicable in the United Kingdom relating to the resolution of unsound or failing banks,
investment firms or other financial institutions or their affiliates (other than through liquidation, administration or other insolvency proceedings).
“Bankruptcy Code” means Title 11 of the United States Code entitled “Bankruptcy.”
“Beneficial Ownership Regulation” means 31 C.F.R. § 1010.230.
“Benefit Plan” means any employee benefit plan as defined in Section 3(3) of ERISA (whether governed by the laws of the United States or otherwise) that is subject to Title I of ERISA or Section 4975 of the Code and to which any Obligor or Subsidiary thereof incurs or otherwise has any obligation or liability, contingent or otherwise.
“Board” means, with respect to any Person, the board of directors (or equivalent management or oversight body) of such Person or any committee thereof duly authorized to act on behalf of such board or equivalent body.
“Borrower” has the meaning set forth in the preamble hereto.
“Borrowing” means the borrowing of the Tranche 1 Loans on the Closing Date.
“Borrowing Date” means the Closing Date (for Tranche 1 Loans).
“Borrowing Notice” means a written notice substantially in the form of Exhibit B.
“Business Day” means a day (other than a Saturday or Sunday) on which commercial banks are not authorized or required to close in New York, New York.
“Capital Lease Obligation” means, as to any Person, any obligation of such Person to pay rent or other amounts under a lease of (or other agreement conveying the right to use) real and/or personal property, which obligation is required to be classified and accounted for as a capital lease on a balance sheet of such Person under GAAP and, for purposes of this Agreement, the amount of any such obligation shall be the capitalized amount thereof, determined in accordance with GAAP.
“Casualty Event” means the damage, destruction or condemnation, as the case may be, of any property of any Person.
“Change of Control” means an event or series of events (including any Acquisition) that causes or results in any of the following: (i) any Person or “group” (as defined above) (other than the Specified Holders) acting in concert shall have acquired beneficial ownership, directly or indirectly, of, or shall have acquired by Contract or otherwise, or shall have entered into a Contract or arrangement that, upon consummation, will result in its or their acquisition of, control over, voting stock of Holdings representing more than fifty percent (50%) of the combined voting power of all voting stock of Holdings, determined on a fully-diluted, as if converted or exercised basis, (ii) Holdings shall cease to own, directly, beneficially and of record, one hundred percent (100%) of the issued and outstanding Equity Interests of the Borrower, free and clear of all Liens, (iii) the Borrower shall cease to own, directly or indirectly, beneficially and of record, one hundred percent (100%) of the issued and outstanding Equity Interests of each of its Subsidiaries, free and clear of
all Liens, or (iv) the sale of all or substantially all of the property or business of Holdings, the Borrower and its Subsidiaries, taken as a whole.
“Claim” means any written claim, demand, complaint, grievance, action, application, suit, cause of action, order, charge, indictment, prosecution, judgment or other similar process, assessment or reassessment, whether made, converted or assessed in connection with a debt, liability, dispute, breach, failure or otherwise.
“Closing Date” means June 30, 2020.
“Closing Date Lender” has the meaning set forth in the preamble hereto.
“Closing Fee” has the meaning set forth in Section 3.04.
“Code” means the Internal Revenue Code of 1986, as amended from time to time, and the rules and regulations promulgated thereunder from time to time.
“Collateral” means any asset or property in which a Lien is purported to be granted under any Loan Document, including future acquired or created assets or property (or all such assets or property, as the context may require).
“Commitment” means, with respect to each Lender, the obligation of such Lender to make Loans to the Borrower on the applicable Borrowing Date in accordance with the terms and conditions of this Agreement, which commitments are in the amounts set forth opposite such Lender’s name on Schedule 1 hereto, as such Schedule may be amended from time to time pursuant to an Assignment and Assumption or otherwise; provided that the aggregate Commitments of all Lenders on the Closing Date equal $50,000,000.
“Commodity Account” means any commodity account, as such term is defined in Section 9-102 of the NY UCC.
“Competitor” means (i) any competitor of the Borrower or any of its Subsidiaries primarily operating in the same line of business as the Borrower or any of its Subsidiaries and (ii) any of its Affiliates (other than any Person that is a bona fide debt fund primarily engaged in the making, purchasing, holding or other investing in commercial loans, notes, bonds or similar extensions of credit or securities in the ordinary course of its business) that are either (x) identified by name in writing by the Borrower to the Administrative Agent from time to time or (y) clearly identifiable on the basis of such Affiliate’s name.
“Connection Income Taxes” means Other Connection Taxes that are imposed on or measured by net income (however denominated) or that are franchise Taxes or branch profits Taxes.
“Contract” means any contract, license, lease, agreement, obligation, promise, undertaking, understanding, arrangement, document, commitment, entitlement, indenture, instrument, or engagement under which a Person has, or will have, any liability or contingent liability (in each case, whether written or oral, express or implied, and whether in respect of
monetary or payment obligations, performance obligations or otherwise), in each case, other than the Loan Documents.
“Control” means, in respect of a particular Person, the possession, by one or more other Persons, directly or indirectly, of the power to direct or cause the direction of the management or policies of such particular Person, whether through the ability to exercise voting power, by contract or otherwise. “Controlling” and “Controlled” (and similar derivatives) have meanings correlative thereto.
“Controlled Account” has the meaning set forth in Section 8.17(a).
“Controlled Investment Affiliates” means, as to any Person (the “Controlling Person”), any Affiliate of such Controlling Person that (i) is Controlled, directly or indirectly by such Controlling Person, or (ii) was organized by such Controlling Person (or any Person Controlled by such Controlling Person) for the purpose of making equity or debt investments in Holdings or the Borrower or other portfolio companies of such Controlling Person.
“Copyright” means all copyrights, copyright registrations and applications for copyright registrations, including all renewals and extensions thereof, all rights to recover for past, present or future infringements thereof, and all other rights whatsoever accruing thereunder or pertaining thereto throughout the world.
“Coverage Sponsor” means any commercial entity, including health plans, self-insured employers, unions, accountable care organizations, special needs plans, worker’s compensation insurance plans, behavioral health organizations, pharmacy benefit managers, formulary optimization managers, medical benefit managers, third party administrators or any governmental entity, including Medicare, Medicaid, the State Children’s Health Insurance Program, the Department of Defense TRICARE and TRICARE for Life programs, the Veterans Health Administration program and the Indian Health Service program.
“Covered Insured Lives” means all individuals, employees and dependents for whom the Coverage Sponsor has an obligation to manage, adjudicate, pay or disburse claim payments.
“Default” means any Event of Default and any event that, upon the giving of notice, the lapse of time or both, would constitute an Event of Default.
“Default Rate” has the meaning set forth in Section 3.02(b).
“Deposit Account” means any deposit account, as such term is defined in Section 9-102 of the NY UCC.
“Designated Jurisdiction” means any country or territory to the extent that such country or territory is the subject of any Sanction.
“Device” means any medical instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent or other similar or related item, including any component, part or accessory, that (i) is intended for use in connection with the diagnosis of disease, malady or other conditions or in the cure, mitigation, treatment or prevention of disease or malady, in man or other
animals, or is intended to affect the structure or any function of the body of man or other animals, (ii) does not achieve its primary intended purpose or purposes through chemical action within or on the body of man or other animals and (iii) is not dependent upon being metabolized for the achievement of its primary intended purpose or purposes.
“Device Clearance Application” means any de novo application or request submitted under Section 513(f) of the FD&C Act (21 U.S.C. § 360c(f)), or any 510(k) submitted under Section 510(k) of the FD&C Act (21 U.S.C. § 360(k)) seeking clearance from the FDA for a Device that is substantially equivalent to a legally marketed predicate Device, as defined in the FD&C Act, or any corresponding or similar non-U.S. application or request in any non-U.S. jurisdiction, including, with respect to the European Union, any equivalent submission to a Standard Body pursuant to an applicable directive of the European Council with respect to any CE marking (or, if applicable, a self-certification of conformity with respect to any such directive through a “declaration of conformity”).
“Disqualified Equity Interests” means, with respect to any Person, any Equity Interest of such Person that, by its terms (or by the terms of any security or other Equity Interest into which it is convertible or for which it is exchangeable upon exercise or otherwise), or upon the happening of any event or condition (i) matures or is mandatorily redeemable (other than solely for Qualified Equity Interests), including pursuant to a sinking fund obligation or otherwise, (ii) is redeemable at the option of the holder thereof (other than solely for Qualified Equity Interests), in whole or in part, (iii) provides for the scheduled payments of dividends or other distributions in cash or other securities that would constitute Disqualified Equity Interests, or (iv) is or becomes convertible into or exchangeable for Indebtedness or any other Equity Interests that would constitute Disqualified Equity Interests, in each case, prior to the date that is ninety-one (91) days after the Maturity Date.
“Dollars” and “$” means lawful money of the United States of America.
“Early Prepayment Fee” means, with respect to any prepayment of all or any portion of the outstanding principal amount of the Loans on any Prepayment Date, whether pursuant to clause (a) or (b) of Section 3.03 or otherwise (including as a result of acceleration, an Insolvency Proceeding or other Event of Default), the positive difference (if any) of (i) the product of (A) the principal amount of such prepayment multiplied by (B) 1.50, less (ii) the sum of (A) the principal amount of such prepayment, plus (B) the amount of interest on such prepaid principal amount paid to the Lenders prior to such Prepayment Date, plus (C) the amount of interest on such prepaid principal amount to be paid to the Lenders on such Prepayment Date, plus (D) an amount equal to the product of (x) the total amount of the Closing Fee paid hereunder prior to such Prepayment Date multiplied by (y) a fraction (expressed as a percentage) having a numerator equal to such prepaid principal amount and a denominator equal to the aggregate original principal amount of the Loans made as of such Prepayment Date; provided that for purposes of calculating the amounts described in clauses (ii) (B) and (C) above, no portion of interest applicable to the Default Rate, whether paid or payable, shall be included in such calculation.
“EEA Financial Institution” means (i) any credit institution or investment firm established in any EEA Member Country which is subject to the supervision of an EEA Resolution Authority, (ii) any entity established in an EEA Member Country which is a parent of an institution described in clause (i) of this definition, or (iii) any financial institution established in an EEA
Member Country which is a subsidiary of an institution described in clauses (i) or (ii) of this definition and is subject to consolidated supervision with its parent.
“EEA Member Country” means any of the member states of the European Union, Iceland, Liechtenstein, and Norway.
“EEA Resolution Authority” means any public administrative authority or any person entrusted with public administrative authority of any EEA Member Country (including any delegee) having responsibility for the resolution of any EEA Financial Institution.
“Effective Date” means March 25, 2022.
“Eligible Transferee” means and includes (i) any commercial bank, (ii) any insurance company, (iii) any finance company, (iv) any financial institution, (v) any investment fund that invests in loans or other obligations for borrowed money, (vi) with respect to any Lender, any of its Affiliates, and (vii) any other “accredited investor” (as defined in Regulation D of the Securities Act) that is principally in the business of managing investments or holding assets for investment purposes; provided that, an Eligible Transferee shall not include (x) any Competitor, or (y) any Person that primarily invests in distressed debt or other distressed financial assets; provided further that (A) neither clause (x) or (y) above shall apply retroactively to any Person that previously acquired an assignment or participation interest hereunder to the extent such Person was not a Competitor or a Person of the type described in clause (y) above at the time of the applicable assignment or participation, as the case may be, and (B) with respect to both clauses (x) and (y) above, the Administrative Agent shall not have any duty or obligation to carry out due diligence in order to identify or determine whether a Person would be excluded as an Eligible Transferee as a result of the application of either such clause.
“Environmental Law” means any Law or Governmental Approval relating to pollution or protection of the environment or the treatment, storage, disposal, release, threatened release or handling of hazardous materials, and all local laws and regulations, whether U.S. or non-U.S., related to environmental matters and any specific agreements entered into with any competent authorities which include commitments related to environmental matters.
“Equity Interests” means, with respect to any Person (for purposes of this defined term, an “issuer”), all shares of, interests or participations in, or other equivalents in respect of such issuer’s capital stock, including all membership interests, partnership interests or equivalent, and all debt or other securities (including warrants, options and similar rights) directly or indirectly exchangeable, exercisable or otherwise convertible into, such issuer’s capital stock, whether now outstanding or issued after the Closing Date, and in each case, however classified or designated and whether voting or non-voting.
“Equivalent Amount” means, with respect to an amount denominated in a single currency, the amount in another currency that could be purchased by the amount in the former currency determined by reference to the Exchange Rate at the time of determination.
“ERISA” means the United States Employee Retirement Income Security Act of 1974, as amended.
“ERISA Affiliate” means, collectively, any Obligor, Subsidiary thereof, and any Person under common control, or treated as a single employer, with any Obligor or Subsidiary thereof, within the meaning of Section 414(b), (c), (m) or (o) of the Code.
“ERISA Event” means (i) a reportable event as defined in Section 4043 of ERISA with respect to a Title IV Plan, excluding, however, such events as to which the PBGC by regulation has waived the requirement of Section 4043(a) of ERISA that it be notified within thirty (30) days of the occurrence of such event; (ii) the applicability of the requirements of Section 4043(b) of ERISA with respect to a contributing sponsor, as defined in Section 4001(a)(13) of ERISA, to any Title IV Plan where an event described in paragraph (9), (10), (11), (12) or (13) of Section 4043(c) of ERISA is reasonably expected to occur with respect to such plan within the following thirty (30) days; (iii) a withdrawal by any Obligor or any ERISA Affiliate thereof from a Title IV Plan or the termination of any Title IV Plan resulting in liability under Sections 4063 or 4064 of ERISA; (iv) the withdrawal of any Obligor or any ERISA Affiliate thereof in a complete or partial withdrawal (within the meaning of Section 4203 and 4205 of ERISA) from any Multiemployer Plan if there is any potential liability therefor, or the receipt by any Obligor or any ERISA Affiliate thereof of notice from any Multiemployer Plan that it is insolvent pursuant to Section 4241 or 4245 of ERISA; (v) the filing of a notice of intent to terminate, the treatment of a plan amendment as a termination under Section 4041 or 4041A of ERISA, or the commencement of proceedings by the PBGC to terminate a Title IV Plan or Multiemployer Plan; (vi) the imposition of liability on any Obligor or any ERISA Affiliate thereof pursuant to Sections 4062(e) or 4069 of ERISA or by reason of the application of Section 4212(c) of ERISA; (vii) the failure by any Obligor or any ERISA Affiliate thereof to make any required contribution to a Title IV Plan, or the failure to meet the minimum funding standard of Section 412 of the Code with respect to any Title IV Plan (whether or not waived in accordance with Section 412(c) of the Code) or the failure to make by its due date a required installment under Section 430 of the Code with respect to any Title IV Plan or the failure to make any required contribution to a Multiemployer Plan; (viii) the determination that any Title IV Plan is considered an at-risk plan or the receipt of notice that a Multiemployer Plan is in endangered to critical status, within the meaning of Sections 430, 431 and 432 of the Code or Sections 303, 304 and 305 of ERISA, as applicable; (ix) an event or condition which might reasonably be expected to constitute grounds under Section 4042 of ERISA for the termination of, or the appointment of a trustee to administer, any Title IV Plan or Multiemployer Plan; (x) the imposition of any liability under Title I or Title IV of ERISA, other than PBGC premiums due but not delinquent under Section 4007 of ERISA, upon any Obligor or any ERISA Affiliate thereof; (xi) an application for a funding waiver under Section 303 of ERISA or an extension of any amortization period pursuant to Section 412 of the Code with respect to any Title IV Plan; (xii) the occurrence of a non-exempt prohibited transaction under Sections 406 or 407 of ERISA for which any Obligor or any Subsidiary thereof may be directly or indirectly liable; (xiii) a violation of the applicable requirements of Section 404 or 405 of ERISA or the exclusive benefit rule under Section 401(a) of the Code by any fiduciary or disqualified person for which any Obligor or any ERISA Affiliate thereof may be directly or indirectly liable; (xiv) the occurrence of an act or omission which could reasonably be expected to give rise to the imposition on any Obligor or any ERISA Affiliate thereof of fines, penalties, taxes or related charges under Chapter 43 of the Code or under Sections 409, 502(c), (i) or (1) or 4071 of ERISA; (xv) the assertion of a material claim (other than routine claims for benefits) against any Plan or the assets thereof, or against any Obligor or any Subsidiary thereof in connection with any such Plan; (xvi) receipt from the IRS of notice of the failure of any Qualified Plan to qualify under Section 401(a) of the Code,
or the failure of any trust forming part of any Qualified Plan to fail to qualify for exemption from taxation under Section 501(a) of the Code; (xvii) the imposition of any Lien (or the fulfillment of the conditions for the imposition of any Lien) on any of the rights, properties or assets of any Obligor or any ERISA Affiliate thereof, in either case pursuant to Title I or Title IV of ERISA, including Section 302(f) or 303(k) of ERISA or to Section 401(a)(29) or 430(k) of the Code; (xviii) the establishment or amendment by any Obligor or any Subsidiary thereof of any “welfare plan”, as such term is defined in Section 3(1) of ERISA, that provides post-employment welfare benefits in a manner that would increase the liability of any Obligor; or (xix) any Foreign Benefit Event.
“ERISA Funding Rules” means the rules regarding minimum required contributions (including any installment payment thereof) to Title IV Plans, as set forth in Sections 412, 430, 431, 432 and 436 of the Code and Sections 302, 303, 304 and 305 of ERISA.
“EU Bail-In Legislation Schedule” means the EU Bail-In Legislation Schedule published by the Loan Market Association (or any successor Person), as in effect from time to time.
“Event of Default” has the meaning set forth in Section 11.01.
“Exchange Act” means the Securities Exchange Act of 1934, as amended, and the rules and regulations promulgated thereunder.
“Exchange Rate” means, as of any date of determination, the rate at which any currency may be exchanged into another currency, as set forth on the relevant Reuters screen at or about 11:00 a.m. (New York City time) on such date. In the event that such rate does not appear on the Reuters screen, the “Exchange Rate” shall be determined by reference to such other publicly available service for displaying exchange rates as may be reasonably designated by the Administrative Agent.
“Excluded Accounts” means (x) the Deposit Accounts, Securities Accounts and Commodity Accounts of the Massachusetts Securities Subsidiary identified on Schedule 2 hereto, and (y) any other Deposit Account of Holdings or any of its Subsidiaries (other than the Massachusetts Securities Subsidiary or any Immaterial Subsidiary) (i) into which there are no funds on deposit other than those intended solely to cover wages and payroll for employees of the Borrower or any such Subsidiaries for a period of service no longer than one month at any time (and related contributions to be made on behalf of such employees to health and benefit plans) plus balances for outstanding checks for wages and payroll from prior periods; (ii) constituting employee withholding accounts and contain only funds deducted from pay otherwise due to employees for services rendered to be applied toward the Tax obligations of such employees; (iii) constituting escrow accounts into which there are deposited contractually escrowed funds in connection with Permitted Acquisitions; and (iv) constituting trust or fiduciary accounts into which there are no funds on deposit other than deposits held in trust for the benefit of a third-party (other than any Affiliate of the Borrower).
“Excluded Subsidiary” means (i) the Massachusetts Securities Subsidiary and (ii) any Immaterial Subsidiary.
“Excluded Taxes” means any of the following Taxes imposed on or with respect to a Recipient or required to be withheld or deducted from a payment to a Recipient: (i) Taxes imposed on or measured by net income (however denominated), franchise Taxes and branch profits Taxes, in each case, (x) imposed by the United States as a result of such Recipient being organized under the laws of, or having its principal office or, in the case of any Lender, its applicable lending office located in, the jurisdiction imposing such Tax (or any political subdivisions thereof) or (y) that are Other Connection Taxes, (ii) in the case of a Lender, U.S. federal withholding Taxes imposed on amounts payable to or for the account of such Lender with respect to an applicable interest in a Loan or Commitment pursuant to a law in effect on the date on which (1) such Lender acquires such interest in the Loan or Commitment (other than pursuant to an assignment request by the Borrower under Section 5.03(h)) or (2) such Lender changes its lending office, except in each case to the extent that, pursuant to Section 5.03, amounts with respect to such Taxes were payable either to such Lender's assignor immediately before such Lender became a party hereto or to such Lender immediately before it changed its lending office, (iii) Taxes attributable to such Recipient’s failure to comply with Section 5.03(f), and (iv) any U.S. federal withholding Taxes imposed under FATCA.
“Exclusive License” (and its derivatives) means and refers to any outbound license of Intellectual Property that is exclusive (whether as to use, geography or otherwise) and is not terminable by the licensor at any time upon ninety (90) days’ (or less) prior written notice.
“Expense Deposit” means the cash deposit referenced in the Proposal Letter.
“FATCA” means Sections 1471 through 1474 of the Code, as of the date of this Agreement (or any amended or successor version that is substantively comparable and not materially more onerous to comply with), any current or future regulations or official interpretations thereof and any agreements entered into pursuant to Section 1471(b)(1) of the Code and any fiscal or regulatory legislation, rules or practices adopted pursuant to any intergovernmental agreement, treaty or convention among Governmental Authorities and implementing such Sections of the Code.
“FD&C Act” means the U.S. Food, Drug and Cosmetic Act of 1938 (or any successor thereto), as amended from time to time, and the rules, regulations, guidelines, guidance documents and compliance policy guides issued or promulgated thereunder.
“FDA” means the U.S. Food and Drug Administration and any successor entity.
“Federal Funds Effective Rate” means, for any day, the greater of (i) the rate calculated by the Federal Reserve Bank of New York based on such day’s federal funds transactions by depositary institutions (as determined in such manner as the Federal Reserve Bank of New York sets forth on its public website from time to time) and published on the next succeeding Business Day by the Federal Reserve Bank of New York as the federal funds effective rate and (ii) zero percent (0%).
“Foreign Benefit Event” means, with respect to any Foreign Pension Plan, (a) the existence of unfunded liabilities in excess of the amount permitted under any applicable Law, or in excess of the amount that would be permitted absent a waiver from a Governmental Authority,
(b) the failure to make the required contributions or payments, under any applicable Law, on or before the due date (and any applicable grace period) for such contributions or payments, (c) the receipt of a notice by a Governmental Authority relating to the intention to terminate any such Foreign Pension Plan or to appoint a trustee or similar official to administer any such Foreign Pension Plan, or alleging the insolvency of any such Foreign Pension Plan, (d) the incurrence of any liability in excess of $1,500,000 by Holdings or any of its Subsidiaries under applicable Law on account of the complete or partial termination of such Foreign Pension Plan or the complete or partial withdrawal of any participating employer therein, or (e) the occurrence of any transaction that is prohibited under any applicable Law and that could reasonably be expected to result in the incurrence of any liability by Holdings or any of its Subsidiaries, or the imposition on Holdings or any of its Subsidiaries of any fine, excise tax or penalty resulting from any noncompliance with any applicable Law, in each case in excess of $1,500,000.
“Foreign Lender” means a Lender that is not a U.S. Person.
“Foreign Pension Plan” means any benefit plan that under applicable Law, other than the Laws of the United States or any political subdivision thereof, is required to be funded through a trust or other funding vehicle other than a trust or funding vehicle maintained exclusively by a Governmental Authority.
“GAAP” means generally accepted accounting principles in the United States, as in effect from time to time, set forth in the opinions and pronouncements of the Accounting Principles Board and the American Institute of Certified Public Accountants, in the statements and pronouncements of the Financial Accounting Standards Board and in such other statements by such other entity as may be in general use by significant segments of the accounting profession that are applicable to the circumstances as of the date of determination. All references to “GAAP” used herein shall be to GAAP applied consistently with the principles used in the preparation of the financial statements delivered pursuant to Section 6.01(e)(i).
“Governmental Approval” means any consent, authorization, approval, order, license, franchise, permit, certification, accreditation, registration, clearance, exemption, filing or notice that is issued or granted by or from (or pursuant to any act of) any Governmental Authority, including any application or submission related to any of the foregoing.
“Governmental Authority” means any nation, government, branch of power (whether executive, legislative or judicial), state, province or municipality or other political subdivision thereof and any entity exercising executive, legislative, judicial, monetary, regulatory or administrative functions of or pertaining to government, including without limitation regulatory authorities, governmental departments, agencies, commissions, bureaus, officials, ministers, courts, bodies, boards, tribunals and dispute settlement panels, and other law-, rule- or regulation-making organizations or entities of any state, territory, county, city or other political subdivision of any country, in each case whether U.S. or non-U.S., including the FDA and any other agency, branch or other governmental body that has regulatory, supervisory or administrative authority or oversight over, or is charged with the responsibility or vested with the authority to administer or enforce, any Healthcare Laws.
“Guarantee” of or by any Person (the “guarantor”) means any obligation, contingent or otherwise, of the guarantor guaranteeing or having the economic effect of guaranteeing any Indebtedness or other monetary obligation of any other Person (the “primary obligor”) in any manner, whether directly or indirectly, and including any obligation of the guarantor, direct or indirect, (i) to purchase or pay (or advance or supply funds for the purchase or payment of) such Indebtedness or other monetary obligation or to purchase (or to advance or supply funds for the purchase of) any security for the payment thereof, (ii) to purchase or lease property, securities or services for the purpose of assuring the owner of such Indebtedness or other monetary obligation of the payment thereof, (iii) to maintain working capital, equity capital or any other financial statement condition or liquidity of the primary obligor so as to enable the primary obligor to pay such Indebtedness or other monetary obligation or (iv) as an account party in respect of any letter of credit or letter of guaranty issued to support such Indebtedness or monetary obligation; provided that the term Guarantee shall not include endorsements for collection or deposit in the ordinary course of business.
“Guarantee Assumption Agreement” means a Guarantee Assumption Agreement substantially in the form of Exhibit C, executed by any entity that, pursuant to Section 8.12 is required to become a “Subsidiary Guarantor”.
“Guaranteed Obligations” has the meaning set forth in Section 13.01.
“Hazardous Material” means any substance, element, chemical, compound, product, solid, gas, liquid, waste, by-product, pollutant, contaminant or material which is hazardous or toxic, and includes, without limitation, (i) asbestos, polychlorinated biphenyls and petroleum (including crude oil or any fraction thereof) and (ii) any material classified or regulated as “hazardous” or “toxic” or words of like import pursuant to an Environmental Law.
“Healthcare Laws” means, collectively, all Laws and Regulatory Approvals applicable to any Product, the ownership or use of any Product or the regulation of any Product Commercialization and Development Activities conducted by or on behalf of Holdings or any of its Subsidiaries, whether U.S. or non-U.S., federal, state, local or equivalent, relating to the provision of medical or other professional healthcare services or supplies, billing and collection practices relating to the payment for healthcare services or supplies, insurance law (including law related to payment for “no-fault” claims) and workers compensation law as they relate to the provision of, and billing and payment for, healthcare services, patient healthcare, patient healthcare information, patient abuse, the quality and adequacy of medical care, rate-setting, equipment, personnel, operating policies, fee splitting, including, without limitation, the federal Anti-kickback Statute (42 U.S.C. § 1320a-7b(b)) (the “Federal Anti-Kickback Statute”), the Physician Self-Referral Statute (42 U.S.C. § 1395nn) (the “Stark Law”), the Physician Payments Sunshine Act (42 U.S.C. § 1320a-7h), the civil False Claims Act (31 U.S.C. §§ 3729 et seq.), the criminal False Claims Act (42 U.S.C. § 1320a-7b(a)), all criminal laws relating to health care fraud and abuse, including but not limited to 18 U.S.C. Sections 286, 287, 1035, 1347 and 1349, and the health care fraud criminal provisions under the Health Insurance Portability and Accountability Act of 1996 (42 U.S.C. §§ 1320d et seq.), the exclusion law (42 U.S.C. § 1320a-7), the civil monetary penalties law (42 U.S.C. § 1320a-7a), HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act (42 U.S.C. §§ 17921 et seq.), the FD&C Act, the statutes, regulations and binding directives of applicable federal healthcare programs, including but not
limited to Medicare (Title XVIII of the Social Security Act) and Medicaid (Title XIX of the Social Security Act), any binding collection and reporting requirements relating to applicable federal health care programs, the statutes, regulations, and binding directives relating to the processing of any applicable rebate, chargeback or adjustment, under applicable rules and regulations pursuant to the Medicaid Drug Rebate Program (42 U.S.C. § 1396r-8), any state supplemental rebate program, Medicare average sales price reporting (42 U.S.C. § 1395w-3a), the Public Health Service Act (42 U.S.C. § 256b), the VA Federal Supply Schedule (38 U.S.C. § 8126) or under any state pharmaceutical assistance program or U.S. Department of Veterans Affairs agreement, and any successor government programs, and any rules and regulations promulgated pursuant to the statutes listed herein.
“Healthcare Permit” means, with respect to any Person and its ordinary course business activities, any Regulatory Approval (i) issued or required under any Healthcare Laws applicable to such activities of such Person, including activities related to the provision of billing or invoicing for the sale of goods or services regulated or administered under any Healthcare Laws, or (ii) issued to such Person or required to be held by such Person under any Healthcare Laws.
“Hedging Agreement” means any interest rate exchange agreement, foreign currency exchange agreement, commodity price protection agreement or other interest or currency exchange rate or commodity price hedging arrangement.
“Holdings” has the meaning set forth in the preamble hereto.
“IDE” means an application, including an application filed with any Regulatory Authority, for authorization to commence human clinical studies with respect to any Device, including (i) an Investigational Device Exemption as defined in the FD&C Act or any successor application or procedure filed with the FDA, (ii) an abbreviated Investigational Device Exemption as specified in FDA regulations in 21 C.F.R. § 812.2(b), (iii) any equivalent of any of the foregoing pursuant to or under any non-U.S. country or regulatory jurisdiction, (iv) all amendments, variations, extensions and renewals of any of the foregoing that may be filed with respect thereto, and (v) all related documents and correspondence thereto, including documents and correspondence with Institutional Review Boards, whether U.S. or the non-U.S. equivalent.
“Immaterial Subsidiary” means, as of any time of determination, any Subsidiary of Holdings that (i) individually constitutes or holds less than $1,000,000 in total assets and generates less than $1,000,000 of annual Revenue, and (ii) when taken together with all other then existing Immaterial Subsidiaries, such Subsidiary and such Immaterial Subsidiaries, in the aggregate, constitute or hold less than $2,000,000 in total assets and generate less than $2,000,000 of annual Revenue.
“Indebtedness” of any Person means, without duplication, (i) all obligations of such Person for borrowed money, (ii) all obligations of such Person evidenced by bonds, debentures, notes, loan agreements or similar instruments, (iii) all obligations of such Person upon which interest charges are customarily paid, (iv) all obligations of such Person under conditional sale or other title retention agreements relating to property acquired by such Person, (v) all obligations of such Person in respect of the deferred purchase price of property or services (excluding accounts payable incurred in the ordinary course of business), (vi) all Indebtedness of others secured by (or
for which the holder of such Indebtedness has an existing right, contingent or otherwise, to be secured by) any Lien on property owned or acquired by such Person, whether or not the Indebtedness secured thereby has been assumed, (vii) all Guarantees by such Person of Indebtedness of others, (viii) all Capital Lease Obligations of such Person, (ix) all obligations, contingent or otherwise, of such Person as an account party in respect of letters of credit and letters of guaranty, (x) obligations under any Hedging Agreement, currency swaps, forwards, futures or derivatives transactions, (xi) all obligations, contingent or otherwise, of such Person in respect of bankers’ acceptances, (xii) any Disqualified Equity Interests of such Person, and (xiii) all other obligations required to be classified as indebtedness of such Person under GAAP. The Indebtedness of any Person shall include the Indebtedness of any other entity (including any partnership in which such Person is a general partner) to the extent such Person is liable therefor as a result of such Person’s ownership interest in or other relationship with such entity, except to the extent the terms of such Indebtedness provide that such Person is not liable therefor.
“Indemnified Party” has the meaning set forth in Section 14.03(b).
“Indemnified Taxes” means (i) Taxes, other than Excluded Taxes, imposed on or with respect to any payment made by or on account of any Obligation and (ii) to the extent not otherwise described in clause (i), Other Taxes.
“Information Certificate” means an Information and Collateral Certificate, in substantially the form set forth in Exhibit G.
“Insolvency Proceeding” means (i) any case, action or proceeding before any court or other Governmental Authority relating to bankruptcy, reorganization, insolvency, liquidation, receivership, dissolution, winding-up or relief of debtors, or (ii) any general assignment for the benefit of creditors, composition, marshaling of assets for creditors, or other, similar arrangement in respect of any Person’s creditors generally or any substantial portion of such Person’s creditors, in each case undertaken under U.S. federal, state or foreign law, including the Bankruptcy Code.
“Intellectual Property” means all Patents, Trademarks, Copyrights and Technical Information, whether registered or not, U.S. or non-U.S., including (without limitation) all of the following:
(i) applications, registrations, amendments and extensions relating to such Intellectual Property;
(ii) rights and privileges arising under any applicable Laws with respect to such Intellectual Property;
(iii) rights to sue for or collect any damages for any past, present or future infringements of such Intellectual Property; and
(iv) rights of the same or similar effect or nature in any jurisdiction corresponding to such Intellectual Property throughout the world.
“Intercompany Subordination Agreement” means the Amended and Restated Intercompany Subordination Agreement, dated as of the Effective Date, by and among Holdings,
the Borrower and each of Holdings’ Subsidiaries, pursuant to which all obligations in respect of any Indebtedness owing to any such Person by Holdings, the Borrower or any of Holdings’ Subsidiaries shall be subordinated to the prior payment in full in cash of all Obligations, such agreement to be substantially in the form attached hereto as Exhibit H, as amended or otherwise modified from time to time.
“Interest Period” means, with respect to any Borrowing, (i) initially, the period commencing on (and including) the Borrowing Date on which such Borrowing occurred and ending on (and including) the last day of the calendar month in which such Borrowing was made, and (ii) thereafter, the period beginning on (and including) the first day of each succeeding calendar month and ending on the earlier of (and including) (x) the last day of such calendar month and (y) the Maturity Date.
“Interest Rate” means, for any Interest Period, the sum of (i) the Applicable Margin plus (ii) the greater of (x) the Reference Rate as of the second Business Day immediately preceding the first day of such Interest Period and (y) one percent (1.00%).
“Invention” means any novel, inventive or useful art, apparatus, method, process, machine (including any article or device), manufacture or composition of matter, or any novel, inventive and useful improvement in any art, method, process, machine (including article or device), manufacture or composition of matter.
“Investment” means, for any Person: (i) the acquisition (whether for cash, property, services or securities or otherwise) of Equity Interests, bonds, notes, debentures, partnership or other ownership interests or other securities of any other Person (including any “short sale” or sale of any securities at a time when such securities are not owned by the Person entered into such sale); (ii) the making of any deposit with, or advance, loan, assumption of debt, or other extension of credit to, or capital contribution in any other Person (including the purchase of property from another Person subject to an understanding or agreement, contingent or otherwise, to resell such property to such Person), but excluding any such advance, loan or extension of credit having a term not exceeding ninety (90) days arising in connection with the sale of inventory or supplies by such Person in the ordinary course of business; (iii) the entering into of any Guarantee of, or other contingent obligation with respect to, Indebtedness of any other Person and (without duplication) any amount advanced, lent or extended to such Person; or (iv) the entering into of any Hedging Agreement. The amount of an Investment will be determined at the time the Investment is made without giving effect to any subsequent changes in value.
“IRS” means the U.S. Internal Revenue Service.
“Law” means any U.S. or non-U.S. federal, state, provincial, territorial, municipal or local statute, treaty, rule, guideline, regulation, ordinance, code or administrative or judicial precedent or authority, including any interpretation or administration thereof by any Governmental Authority charged with the enforcement, interpretation or administration thereof, and all applicable administrative orders, directed duties, requests, licenses, authorizations and permits of, and agreements with, any Governmental Authority, in each case whether or not having the force of law.
“Lenders” has the meaning set forth in the preamble hereto.
“Lien” means any mortgage, lien, pledge, charge or other security interest, or any lease, title retention agreement, mortgage, restriction, easement, right-of-way, option or adverse claim (of ownership or possession) or other encumbrance of any kind or character whatsoever or any preferential arrangement that has the practical effect of creating a security interest.
“Loan” means any Tranche 1 Loan.
“Loan Documents” means, collectively, this Agreement, the Notes, the Security Documents, the Proposal Letter, each Warrant Certificate, any Guarantee Assumption Agreement, any Information Certificate, the Intercompany Subordination Agreement, and any other guaranty, security agreement, subordination agreement, intercreditor agreement or other present or future document, instrument, agreement, certificate or other amendment, waiver or modification of the foregoing delivered to the Administrative Agent or any Lender in connection with this Agreement (including, without limitation, in connection with Section 8.12) or any of the other Loan Documents, in each case, as amended or otherwise modified.
“Loss” means judgments, debts, liabilities, expenses, costs, damages or losses, contingent or otherwise, whether liquidated or unliquidated, matured or unmatured, disputed or undisputed, contractual, legal or equitable, including loss of value, reasonable professional fees, including reasonable fees and disbursements of legal counsel on a full indemnity basis, and all costs incurred in investigating or pursuing any Claim or any proceeding relating to any Claim.
“Majority Lenders” means, at any time, Lenders having at such time in excess of fifty percent (50%) of the aggregate Commitments (or, if such Commitments are terminated, the outstanding principal amount of the Loans) then in effect.
“Margin Stock” means “margin stock” within the meaning of Regulation U and Regulation X.
“Massachusetts Securities Subsidiary” means Pear Therapeutics Securities Corporation, a Massachusetts corporation.
“Material Adverse Change” and “Material Adverse Effect” mean any event, occurrence, fact, development or circumstance that has had, or could be reasonably expected to have, a material adverse change in or effect on (i) the business condition (financial or otherwise), operations, performance or property of Holdings and its Subsidiaries taken as a whole, (ii) the ability of any Obligor to perform its obligations under the Loan Documents, as and when due, or (iii) the legality, validity, binding effect or enforceability against any Obligor of the Loan Documents or the rights and remedies of the Administrative Agent or the Lenders under any Loan Document.
“Material Agreement” means any Contract to which Holdings or any of its Subsidiaries is a party or a beneficiary from time to time, or to which any assets or properties of Holdings or any of its Subsidiaries are bound (i) the absence or termination of which could reasonably be expected to result in a Material Adverse Effect or (ii) without duplication during any period of twelve (12) consecutive months is reasonably expected to, directly or indirectly, (x) result in payments or receipts (including royalty, licensing or similar payments) made to Holdings or any of its
Subsidiaries in an aggregate amount in excess of $1,500,000, or (y) require payments or expenditures (including royalty, licensing or similar payments) to be made by Holdings or any of its Subsidiaries in an aggregate amount in excess of $1,500,000.
“Material Indebtedness” means, at any time, any Indebtedness of Holdings or any of its Subsidiaries, the outstanding principal amount of which, individually or in the aggregate, exceeds $1,000,000 (or the Equivalent Amount in other currencies).
“Material Intellectual Property” means all Obligor Intellectual Property currently owned or exclusively licensed in connection with Product Commercialization and Development Activities (i) necessary for the operation of the business of Holdings and its Subsidiaries as currently conducted or as currently contemplated to be conducted, (ii) the loss of which could reasonably be expected to result in a Material Adverse Effect, or (iii) that has a fair market value in excess of $1,500,000.
“Material Regulatory Event” means an Adverse Regulatory Event that (i) individually has resulted in, or could reasonably be expected to result in, a fine, penalty or Loss (including a loss of Revenue) in excess of $2,500,000 or (ii) when taken together with each other Adverse Regulatory Event that has occurred since the Closing Date, has resulted in, or could reasonably be expected to result in, a fine, penalty or Loss (including a loss of Revenue) in excess of $2,500,000.
“Maturity Date” means the earliest to occur of (i) June 30, 2025 and (ii) the acceleration of the Obligations pursuant to Section 11.02, when used herein, the term “scheduled Maturity Date” means the date set forth in clause (i) above.
“Medicaid” means that government-sponsored entitlement program under Title XIX, P.L. 89-97 of the Social Security Act, which provides federal grants to states for medical assistance based on specific eligibility criteria, as set forth on Section 1396, et seq. of Title 42 of the United States Code.
“Medicare” means that government-sponsored insurance program under Title XVIII, P.L. 89-97, of the Social Security Act, which provides for a health insurance system for eligible elderly and disabled individuals, as set forth at Section 1395, et seq. of Title 42 of the United States Code.
“Multiemployer Plan” means any multiemployer plan, as defined in Section 400l(a)(3) of ERISA, to which any ERISA Affiliate incurs or otherwise has any obligation or liability, contingent or otherwise.
“Net Cash Proceeds”, means, (i) with respect to any Casualty Event experienced or suffered by Holdings, the Borrower or any of its Subsidiaries, the amount of cash proceeds received (directly or indirectly) including, without limitation, in the form of insurance proceeds or condemnation awards in respect of such Casualty Event, from time to time by or on behalf of such Person after deducting therefrom only (x) reasonable costs and expenses related thereto incurred by Holdings, the Borrower or such Subsidiary in connection therewith, and (y) Taxes (including transfer Taxes or net income Taxes) paid or payable in connection therewith; and (ii) with respect to any Asset Sale by Holdings, the Borrower or any of its Subsidiaries, the amount of cash proceeds received (directly or indirectly) from time to time by or on behalf of such Person after deducting therefrom only (x) reasonable costs and expenses related thereto incurred by Holdings, the
Borrower or such Subsidiary in connection therewith, (y) Taxes (including transfer Taxes or net income Taxes) paid or payable in connection therewith and (z) reserves for future indemnity obligations and losses only to the extent actually applied; provided that, in each case of clauses (i) and (ii), costs and expenses shall only be deducted to the extent, that the amounts so deducted are (x) actually paid to a Person that is not an Affiliate of Holdings, the Borrower or any of its Subsidiaries and (y) properly attributable to such Casualty Event or Asset Sale, as the case may be.
“Note” means a promissory note, in substantially the form of Exhibit A hereto, executed and delivered by the Borrower to any Lender in accordance with Section 2.03.
“NY UCC” means the UCC as in effect from time to time in New York.
“Obligations” means, all amounts, obligations, liabilities, covenants and duties of every type and description (including all Guaranteed Obligations and Warrant Obligations) owing by any Obligor to any Secured Party, any indemnitee hereunder or any participant, arising out of, under, or in connection with, any Loan Document, whether direct or indirect (regardless of whether acquired by assignment), absolute or contingent, due or to become due, whether liquidated or not, now existing or hereafter arising and however acquired, and whether or not evidenced by any instrument or for the payment of money, including, without duplication, (i) all Loans, (ii) all interest, whether or not accruing after the filing of any petition in bankruptcy or after the commencement of any insolvency, reorganization or similar proceeding, and whether or not a claim for post-filing or post-petition interest is allowed in any such proceeding, and (iii) all other fees, expenses (including reasonable fees, charges and disbursement of counsel), interest, commissions, charges, costs, disbursements, indemnities and reimbursement of amounts paid and other sums chargeable to such Obligor under any Loan Document.
“Obligor Intellectual Property” means, at any time of determination, Intellectual Property owned by, licensed to or otherwise held by Holdings, the Borrower or any Subsidiary Guarantor at such time including, without limitation, the Intellectual Property listed on Schedule 7.05(c).
“Obligor Party” has the meaning set forth in Section 14.03(b).
“Obligors” means, collectively, Holdings, the Borrower, the Subsidiary Guarantors and any Subsidiary of the Borrower required to become a Subsidiary Guarantor or execute and deliver or become a party to a Security Document pursuant to Section 8.12, and their respective successors and permitted assigns.
“Observer” has the meaning set forth in Section 8.20(a).
“OFAC” means the U.S. Department of the Treasury’s Office of Foreign Assets Control.
“One-Month LIBOR” means, with respect to any applicable Interest Period hereunder, the one-month London Interbank Offered Rate for deposits in Dollars at approximately 11:00 a.m. (London, England time), as determined by the Administrative Agent from the appropriate Bloomberg or Telerate page selected by the Administrative Agent (or any successor thereto or similar source reasonably determined by the Administrative Agent from time to time), which shall be that one-month London Interbank Offered Rate for deposits in Dollars in effect two (2) Business
Days prior to the first day of such Interest Period rounded up to the nearest one hundredth (1/100) of one percent (1%). The Administrative Agent’s determination of interest rates shall be binding on all parties to the Loan Documents in the absence of manifest error.
“Organic Document” means, for any Person, such Person’s formation documents, including, as applicable its certificate of incorporation, by-laws, certificate of partnership, partnership agreement, certificate of formation, limited liability agreement, operating agreement and all shareholder agreements, voting trusts and similar arrangements applicable to such Person’s Equity Interests, or any equivalent document of any of the foregoing.
“Original Credit Agreement” has the meaning set forth in the first recital hereto.
“Other Connection Taxes” means, with respect to any Recipient, Taxes imposed as a result of a present or former connection between such Recipient and the jurisdiction imposing such Tax (other than connections arising from such Recipient having executed, delivered, become a party to, performed its obligations under, received payments under, received or perfected a security interest under, engaged in any other transaction pursuant to or enforced any Loan Document, or sold or assigned an interest in any Loan or Loan Document).
“Other Taxes” means all present or future stamp, court or documentary, intangible, recording, filing or similar Taxes that arise from any payment made under, from the execution, delivery, performance, enforcement or registration of, from the receipt or perfection of a security interest under, or otherwise with respect to, any Loan Document, except any such Taxes that are Other Connection Taxes imposed with respect to an assignment (other than an assignment made pursuant to Section 5.03(h)).
“Participant” has the meaning set forth in Section 14.05(e).
“Participant Register” has the meaning set forth in Section 14.05(g).
“Patents” means all patents and patent applications, including (i) the Inventions and improvements described and claimed therein, (ii) the reissues, divisions, continuations, renewals, extensions, and continuations in part thereof, and (iii) all income, royalties, damages and payment now or hereafter due and payable with respect thereto, (iv) all damages and payment for past or future infringements thereof, and rights to sue thereof, and (v) all rights whatsoever accruing thereunder or pertaining thereto throughout the world.
“Patriot Act” has the meaning set forth in Section 14.20.
“Payment Date” means (i) the last day of each Interest Period (provided that if such last day of any Interest Period is not a Business Day, then the Payment Date shall be the next succeeding Business Day) and (ii) the Maturity Date.
“PBGC” means the United States Pension Benefit Guaranty Corporation referred to and defined in ERISA and any successor entity performing similar functions.
“Permitted Acquisition” means (x) the Waypoint Product Acquisition; provided that such Acquisition is consummated in accordance with the terms and conditions of the Waypoint Product
Acquisition Agreement without any material waiver, amendment or other modification thereto (unless approved in writing by the Administrative Agent prior to the effectiveness thereof, which approval may not be unreasonably withheld), and (y) any other Acquisition by the Borrower or any of its Subsidiaries; provided that, in the case of any such other Acquisition made pursuant to this clause (y):
(a) immediately prior to, and after giving effect to such Acquisition, (i) all representations and warranties contained in this Agreement and the other Loan Documents that are qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct, (ii) all representations and warranties contained in this Agreement and the other Loan Documents that are not qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct in all material respects, and (iii) no Default shall have occurred and be continuing or could reasonably be expected to result therefrom;
(b) all transactions in connection therewith shall be consummated in accordance with all applicable Laws;
(c) in the case of an Acquisition of Equity Interests of any Person, all of such Equity Interests (except for any such securities in the nature of directors’ qualifying shares required pursuant to any applicable Law) shall be owned by Holdings or a wholly-owned, direct or indirect Subsidiary of the Borrower, and, in the event of an Acquisition that results in the creation or acquisition of a new Subsidiary of Holdings, the Borrower shall have taken, or caused to be taken, as of the date such Person becomes a Subsidiary of the Borrower, each of the actions set forth in Section 8.12(a), if applicable;
(d) such Person (in the case of an Acquisition of Equity Interests of such Person) or assets (in the case of an Acquisition of assets or a division of such Person) shall be engaged or used, as the case may be, in businesses or lines of business that would be permitted pursuant to Section 9.04;
(e) on a pro forma basis after giving effect to such Acquisition, the Obligors and their Subsidiaries shall be in compliance with the financial covenants set forth in Section 10;
(f) to the extent that the purchase price for any such Acquisition is paid in cash, the amount thereof, when taken together with the purchase price paid in cash for all other Acquisitions consummated or effected since the Closing Date, does not exceed $5,000,000 in the aggregate (orbelow reflects your understanding of, and agreement to, the terms and conditions set forth above.
(f)Modifications in writing. No variations or modifications hereof shall be deemed valid unless reduced to writing and signed by the Equivalent Amount thereof);Pear and you.
(g) the fair market value of the consideration paid in such Acquisition (exclusive of Qualified Equity Interests of Holdings or the Borrower), when taken together with the fair market value of consideration paid in connection with all other Permitted Acquisitions consummated or effected since the Closing Date (inclusive of cash, deferred purchase price payments, whether in respect of earn-out payments, post-closing adjustments, payments on “seller notes” or otherwise, to the extent actually paid, but excluding Qualified Equity Interests of Holdings or the Borrower), does not exceed $10,000,000 in the aggregate;
(h) at least five (5) Business Days prior to the proposed date of the Acquisition, the Administrative Agent shall have received a certificate of a Responsible Officer of the Borrower (prepared in reasonable detail), certifying that the Acquisition complies with the requirements of this definition, and which certificate shall include a summary (prepared in reasonable detail), certifying as to any contingent liabilities and prospective research and development costs associated with the Person, business or assets being acquired;
(i) to the extent that the purchase price for any such Acquisition is paid in Equity Interests, all such Equity Interests shall be Qualified Equity Interests;
(j) in the case of any Acquisition that has a purchase price in excess of $5,000,000, the Borrower shall have provided the Administrative Agent with at least fifteen (15) calendar days’ prior written notice of any such Acquisition, together with (i) a copy of the draft purchase agreement related to the proposed Acquisition (and any related documents requested by the Administrative Agent), (ii) any available quarterly and annual financial statements of the Person whose Equity Interests or assets are being acquired for the twelve (12) month period ending thirty (30) days immediately prior to such Acquisition, including any audited financial statements that are available, (iii) a summary of due diligence conducted by or on behalf of Holdings, the Borrower or its applicable Subsidiary, as applicable, prior to such Acquisition, (iv) summary information regarding any contingent liabilities or prospective research and development costs associated with the Person, business or assets being acquired and (v) any other information reasonably requested by the Administrative Agent and available to the Obligors; and
(k) neither Holdings nor any of its Subsidiaries shall, in connection with (and upon giving effect to) any such Acquisition, assume or remain liable with respect to, or be subject to (x) any Indebtedness of the related seller or the business, Person or properties acquired, except to the extent permitted pursuant to Section 9.01(g), (y) any Lien on any business, Person or assets acquired, except to the extent permitted pursuant to Section 9.02, or (z) any other liability (including Tax, ERISA or environmental liabilities) in excess of $1,500,000 in the aggregate since the Closing Date.
“Permitted Cash Equivalent Investments” means (i) marketable direct obligations issued or unconditionally guaranteed by the United States or any agency or any state thereof having maturities of not more than one year from the date of acquisition, (ii) commercial paper maturing no more than two hundred and seventy (270) days after the date of its creation and having the highest rating from either Standard & Poor’s Ratings Group or Moody’s Investors Service, Inc. and (iii) registered money market funds at least ninety five percent (95%) of the assets of which constitute Permitted Cash Equivalent Investments of the kinds described in in clauses (i) and (ii) above.
“Permitted Indebtedness” means any Indebtedness permitted under Section 9.01.
“Permitted Liens” means any Liens permitted under Section 9.02.
“Permitted Refinancing” means, with respect to any Indebtedness permitted to be refinanced, extended, renewed or replaced hereunder, any refinancings, extensions, renewals and
replacements of such Indebtedness; provided that such refinancing, extension, renewal or replacement shall not (i) increase the outstanding principal amount of the Indebtedness being refinanced, extended, renewed or replaced (except in the amount of accrued interest, upfront fees and transaction costs), (ii) contain terms relating to outstanding principal amount, amortization, maturity, collateral security (if any) or subordination (if any), or other material terms that, taken as a whole, are less favorable in any material respect to Holdings and its Subsidiaries or the Secured Parties than the terms of any agreement or instrument governing the Indebtedness being refinanced, (iii) have an applicable interest rate or equivalent yield that exceeds the interest rate or equivalent yield of the Indebtedness being refinanced, (iv) contain any new requirement to grant any Lien or to give any Guarantee that was not an existing requirement of the Indebtedness being refinanced and (v) after giving effect to such refinancing, extension, renewal or replacement, no Default shall have occurred (or could reasonably be expected to occur) as a result thereof.
“Person” means any individual, corporation, company, voluntary association, partnership, limited liability company, joint venture, trust, unincorporated organization or Governmental Authority or other entity of whatever nature.
“Plan” means any employee pension benefit plan (other than a Multiemployer Plan) subject to the provisions of Title IV of ERISA or Section 412 of the Code or Section 302 of ERISA, and in respect of which Holdings, the Borrower or any ERISA Affiliate is (or, if such plan were terminated, would under Section 4069 of ERISA be deemed to be) an “employer” as defined in Section 3(5) of ERISA.
“Positive Reimbursement Coverage” means a Coverage Sponsor has announced the decision to place a Product on its formulary or to cover a Product as a medically necessary service as part of its benefits.
“Prepayment Date” has the meaning set forth in Section 3.03(a)(i).
“Prepayment Price” has the meaning set forth in Section 3.03(a)(i).
“Product” means (i) those products and Devices set forth on Schedule 7.05(b) and (ii) any other products or Devices developed, distributed, imported, exported, labeled, promoted, licensed, marketed, sold or otherwise commercialized by Holdings or any of its Subsidiaries, but excluding any such products or Devices that, if in development or which may be developed, have not yet reached the stage of Advanced Development.
“Product Commercialization and Development Activities” means, with respect to any Product, any combination of (i) research, development, manufacturing, quality compliance, use, sale, licensing, importation, exportation, shipping, storage, handling, designing, labeling, marketing, promotion, supply, dispensing, distribution, testing, packaging, purchasing or other commercialization activity, (ii) receipt of payment or other remuneration in respect of any of the foregoing (including, without limitation, in respect of licensing, royalty or similar payments) or (iii) any similar or other activities the purpose of which is to commercially exploit such Product.
“Product Related Information” means, with respect to any Product, all books, records, lists, ledgers, files, manuals, Contracts, correspondence, reports, plans, drawings, data and other information of every kind (in any form or medium), including related to Intellectual Property, and
all techniques and other know-how, that is necessary or useful for any Product Commercialization and Development Activities relating to such Product, including (i) branding materials, packaging and other marketing, promotion and sales materials and information, (ii) clinical data, information included or supporting any Regulatory Approval and all other documents, records, files, data and other information relating to Product Commercialization and Development Activities, (iii) litigation and dispute records, and accounting records, and (iv) all other information, techniques and know-how necessary or useful in connection with the Product Commercialization and Development Activities for any Product.
“Product Standards” means all safety, quality and other specifications and standards applicable to any Product, including all medical device and other standards promulgated by Standards Bodies.
“Prohibited Payment” means any bribe, rebate, payoff, influence payment, kickback or other payment or gift of money or anything of value (including meals or entertainment) to any officer, employee or ceremonial office holder of any government or instrumentality thereof, political party or supra-national organization (such as the United Nations), any political candidate, any royal family member or any other person who is connected or associated personally with any of the foregoing that is prohibited under any applicable Law for the purpose of influencing any act or decision of such payee in such payee’s official capacity, inducing such payee to do or omit to do any act in violation of such payee’s lawful duty, securing any improper advantage or inducing such payee to use such payee’s influence with a government or instrumentality thereof to affect or influence any act or decision of such government or instrumentality.
“Proportionate Share” means, with respect to each Lender, the percentage obtained by dividing (i) the sum of all Commitments (or, if the Commitments are terminated, the outstanding principal amount of the Loans) of such Lender then in effect by (ii) the sum of all Commitments (or, if the Commitments are terminated, the outstanding principal amount of the Loans) of all Lenders then in effect.
“Proposal Letter” means the Proposal Letter, dated May 19, 2020, between the Borrower and Perceptive Advisors LLC (as supplemented by the outline of proposed terms and conditions attached thereto).
“Qualified Equity Interest” means, with respect to any Person, any Equity Interest of such Person that is not a Disqualified Equity Interest.
“Qualified Plan” means an employee benefit plan (as defined in Section 3(3) of ERISA) other than a Multiemployer Plan (i) that is or was at any time maintained or sponsored by any Obligor or any ERISA Affiliate thereof or to which any Obligor or any ERISA Affiliate thereof has ever made, or was ever obligated to make, contributions, and (ii) that is intended to be tax qualified under Section 401(a) of the Code.
“Real Property Security Documents” means any landlord consents, bailee letters, any mortgage or deed of trust or any other real property security document executed or required hereunder to be executed by any Obligor and granting a security interest in real property owned by any Obligor in favor of the Secured Parties for purposes of securing the Obligations.
“Recipient” means any Lender, the Administrative Agent or any other recipient of any payment to be made by or on account of any Obligation, as applicable.
“Reference Rate” means One-Month LIBOR; provided that if One-Month LIBOR can no longer be determined by the Administrative Agent (in its sole but reasonable discretion) or the Governmental Authority having jurisdiction over the quotation or determination of London Interbank Offered Rates ceases to supervise or sanction such rates for purposes of interest rates on loans, then the Administrative Agent and Holdings shall endeavor, in good faith, to establish an alternate rate of interest to One-Month LIBOR that gives due consideration to the then prevailing market convention for determining a rate of interest for middle-market loans in the United States at such time, and shall enter into an amendment to this Agreement to reflect such alternate rate of interest and such other related changes to this Agreement as may be applicable; provided, further that, until such alternate rate of interest is agreed upon by the Administrative Agent and the Borrower, the Reference Rate for purposes hereof and of each other Loan Document shall be the Wall Street Journal Prime Rate.
“Referral Source” has the meaning set forth in Section 7.07(c).
“Refinanced Debt” means the Loan and Security Agreement dated December 22, 2016 between the Borrower and Silicon Valley Bank, as amended.
“Register” has the meaning set forth in Section 14.05(d).
“Regulation T” means Regulation T of the Board of Governors of the Federal Reserve System, as amended.
“Regulation U” means Regulation U of the Board of Governors of the Federal Reserve System, as amended.
“Regulation X” means Regulation X of the Board of Governors of the Federal Reserve System, as amended.
“Regulatory Approval” means, with respect to any Product or Product Commercialization and Development Activities, any Governmental Approval, whether U.S. or non-U.S. that is required to be held or maintained by, or for the benefit of, Holdings, the Borrower or any of its Subsidiaries with respect thereto, including all applicable IDEs, 510(k)s, Device Clearance Applications, Product Standards, supplements, amendments, pre-approvals and post-approvals, governmental price, reimbursement approvals and approvals of applications for regulatory exclusivity, clearances, licenses, notifications, registrations or authorizations of any Regulatory Authority, in each case necessary for the ownership, use or other commercialization of such Product or for such Product Commercialization and Development Activities.
“Regulatory Authority” means any Governmental Authority, whether U.S. or non-U.S., that is concerned with or has regulatory or supervisory oversight with respect to any Product or any Product Commercialization and Development Activities relating to any Product, including the FDA and all equivalent Governmental Authorities, whether U.S. or non-U.S.
“Related Fund” means, with respect to any Lender, a fund which is managed or advised by the same investment manager or investment adviser as such Lender or, if it is managed by a different investment manager or investment adviser, a fund whose investment manager or investment adviser is an Affiliate of the investment manager or investment adviser of such Lender.
“Related Parties” has the meaning set forth in Section 14.16.
“Resolution Authority” means an EEA Resolution Authority or, with respect to any UK Financial Institution, a UK Resolution Authority.
“Responsible Officer” of any Person means each of the president, chief executive officer, chief financial officer, controller and similar officer of such Person.
“Restatement Amendment” has the meaning set forth in the first recital hereto.
“Restricted Payment” means any dividend or other distribution (whether in cash, Equity Interests or other property) with respect to any Equity Interests of Holdings or any of its Subsidiaries, any payment of interest, principal or fees in respect of any Indebtedness owed by Holdings or any of its Subsidiaries to any holder of any Equity Interests of Holdings or any of its Subsidiaries, or any payment (whether in cash, Equity Interests or other property), including any sinking fund or similar deposit, on account of the purchase, redemption, retirement, acquisition, cancellation or termination of any such Equity Interests of Holdings or any of its Subsidiaries, or any option, warrant or other right to acquire any such Equity Interests of Holdings or any of its Subsidiaries.
“Restrictive Agreement” means any Contract or other arrangement that prohibits, restricts or imposes any condition upon (i) the ability of Holdings or any of its Subsidiaries (other than any Excluded Subsidiary for so long as it remains an Excluded Subsidiary) to create, incur or permit to exist any Lien upon any of its properties or assets (other than (x) customary provisions in Contracts (including without limitation leases and licenses of Intellectual Property) restricting the assignment thereof and (y) restrictions or conditions imposed by any Contract governing secured Permitted Indebtedness permitted under Section 9.01(g), to the extent that such restrictions or conditions apply only to the property or assets securing such Indebtedness), or (ii) the ability of Holdings or any of its Subsidiaries (other than any Excluded Subsidiary for so long as it remains an Excluded Subsidiary) to make Restricted Payments with respect to any of their respective Equity Interests, to make or repay loans or advances to Holdings, Borrower or any such Subsidiary, or to Guarantee Indebtedness of Holdings, the Borrower or any such Subsidiary.
“Revenue” means, for any relevant fiscal period, the consolidated total net revenues of Holdings and its Subsidiaries for such fiscal period generated in the ordinary course of business, as recognized on the income statement of Holdings and its Subsidiaries for such fiscal period, determined on a consolidated basis in accordance with GAAP; provided that no more than $5,000,000 of Revenue received from or in connection with contingent milestone payments under licensing and collaborations agreements shall be included in the calculation of Revenue for all purposes hereunder.
“Sanction” means any international economic sanction administered or enforced by the United States government (including, without limitation, OFAC), the United Nations Security
Council, the European Union or its Member States, Her Majesty’s Treasury or other relevant sanctions authority.
“Secured Party” means each Lender, the Administrative Agent, each other Indemnified Party, any other holder of any Obligation, and any of their respective permitted transferees or assigns.
“Securities Account” means any securities account, as such term is defined in Section 8-501 of the NY UCC.
“Securities Act” means the Securities Act of 1933, as amended, and the rules and regulations promulgated thereunder.
“Security Agreement” means the Amended and Restated Security Agreement, dated as of the Effective Date, among the Borrower as the grantor party thereto and the Administrative Agent, granting a security interest in all of Holdings’ and the Borrower’s personal property in favor of the Administrative Agent.
“Security Documents” means, collectively, the Security Agreement, each Real Property Security Document, each Short-Form IP Security Agreement, and each other security agreement, control agreement or financing statement, registration, recordation, filing, instrument or approval required, entered into or recommended to grant, perfect and otherwise render enforceable Liens in favor of the Secured Parties for purposes of securing the Obligations, including (without limitation) pursuant to Section 8.12.
“Short-Form IP Security Agreements” means short-form copyright, patent or trademark (as the case may be) security agreements, substantially in the form Exhibit C, Exhibit D or Exhibit E to the Security Agreement, entered into by one or more Obligors in favor of the Secured Parties, each in form and substance reasonably satisfactory to the Administrative Agent (and as amended, modified or replaced from time to time).
“Solvent” means, with respect to any Person at any time, that (i) the present fair saleable value of the property of such Person is greater than the total amount of liabilities (including contingent liabilities) of such Person, (ii) the present fair saleable value of the property of such Person is not less than the amount that will be required to pay the probable liability of such Person on its debts as they become absolute and matured, (iii) such Person has not incurred and does not intend to, and does not believe that it will, incur debts or liabilities beyond such Person’s ability to pay as such debts and liabilities mature and (iv) such Person would be able to obtain a letter from its auditors that did not contain a going concern qualification.
“Specified Asset Sale” means any Asset Sale of the type described in any of clauses (d), (e), (g), (h), (i) or (j) of Section 9.09.
“Specified Holders” means (i) 5AM Ventures IV, LP, (ii) 5AM Co-Investors IV, LP, (iii) 5AM Opportunities I, L.P., (iv) Arboretum Ventures IV, L.P. and (v) Temasek Capital Management Pte. Ltd., and each Controlled Investment Affiliate of any Person specified in clauses (i) through (v) of this definition.
“Specified Intellectual Property” means any Intellectual Property to be licensed through an Exclusive License or sold by Holdings, the Borrower or its Subsidiaries on arm’s length terms to a non-affiliated pharmaceutical or life science company, or a joint venture or similar arrangement with such a company.
“Standard Bodies” means any of the organizations that create, sponsor or maintain safety, quality or other standards, including ISO, ANSI, CEN and SCC and the like.
“Subsidiary” means, with respect to any Person (for purposes of this definition, the “parent”) at any date, any corporation, limited liability company, partnership, association or other entity the accounts of which would be consolidated with those of the parent in the parent’s consolidated financial statements if such financial statements were prepared in accordance with GAAP as of such date, as well as any other corporation, limited liability company, partnership, association or other entity (i) of which securities or other ownership interests representing more than fifty percent (50%) of the equity or more than fifty percent (50%) of the ordinary voting power or, in the case of a partnership, more than fifty percent (50%) of the general partnership interests are, as of such date, owned, controlled or held, directly or indirectly or (ii) that is, as of such date, otherwise Controlled, by the parent or one or more direct or indirect subsidiaries of the parent or by the parent and one or more subsidiaries of the parent. Unless otherwise specified, all references herein to a “Subsidiary” or to “Subsidiaries” shall refer to a Subsidiary or Subsidiaries of the Borrower.
“Subsidiary Guarantor” means, initially as of the Closing Date, each Subsidiary of the Borrower identified under the caption “SUBSIDIARY GUARANTORS” on the signature pages hereto and, thereafter, each Subsidiary of the Borrower that becomes, or is required to become, a “Subsidiary Guarantor” after the Closing Date pursuant to Section 8.12.
“Taxes” means all present or future taxes, levies, imposts, duties, deductions, withholdings (including backup withholding), assessments, fees or other charges imposed by any Governmental Authority, including any interest, additions to tax or penalties applicable thereto.
“Technical Information” means all trade secrets and other proprietary or confidential information of a scientific, technical or commercial nature related to Product Commercialization and Development Activities.
“Title IV Plan” means an employee benefit plan (as defined in Section 3(3) of ERISA) other than a Multiemployer Plan (i) that is or was at any time maintained or sponsored by any Obligor or any ERISA Affiliate thereof or to which any Obligor or any ERISA Affiliate thereof has ever made, or was obligated to make, contributions, and (ii) that is or was subject to Section 412 of the Code, Section 302 of ERISA or Title IV of ERISA.
“Trademarks” means all trade names, trademarks and service marks, logos, trademark and service mark registrations, and applications for trademark and service mark registrations, including (i) all renewals of trademark and service mark registrations, (ii) all rights to recover for all past, present and future infringements thereof and all rights to sue therefor, and (iii) all rights whatsoever accruing thereunder or pertaining thereto throughout the world, together, in each case, with the goodwill of the business connected with the use thereof.
“Tranche 1 Loan” has the meaning set forth in the first recital hereto.
“Transactions” means the negotiation, preparation, execution, delivery and performance by each Obligor of this Agreement and the other Loan Documents to which such Obligor is (or is intended to be) a party, the making of the Loans hereunder, the repayment of the Refinanced Debt, and all other transactions contemplated pursuant to this Agreement and the other Loan Documents.
“UCC” means, with respect to any applicable jurisdictions, the Uniform Commercial Code as in effect in such jurisdiction, as may be modified from time to time.
“UK Financial Institution” means any BRRD Undertaking (as such term is defined under the PRA Rulebook (as amended from time to time) promulgated by the United Kingdom Prudential Regulation Authority) or any person falling within IFPRU 11.6 of the FCA Handbook (as amended from time to time) promulgated by the United Kingdom Financial Conduct Authority, which includes certain credit institutions and investment firms, and certain affiliates of such credit institutions or investment firms.
“UK Resolution Authority” means the Bank of England or any other public administrative authority having responsibility for the resolution of any UK Financial Institution.
“United States” or “U.S.” means the United States of America, its fifty (50) states and the District of Columbia.
“U.S. Person” means a “United States person” as defined in Section 7701(a)(30) of the Code.
“U.S. Tax Compliance Certificate” has the meaning set forth in Section 5.03(f)(ii)(B)(3).
“Wall Street Journal Prime Rate” means the Wall Street Journal Prime Rate, as published and defined in The Wall Street Journal.
“Warrant Certificate” means a Warrant Certificate in substantially the form of Exhibit J to the Original Credit Agreement, to be delivered pursuant to Section 6.01(g) (as a condition precedent to the Borrowing of Tranche 1 Loans), as amended or otherwise modified pursuant to the terms thereof.
“Warrant Obligations” means all Obligations of the Borrower owing to Perceptive Advisors LLC or one of its managed funds (and no other Person) arising out of, under or in connection with the Warrant Certificates; provided that, any term or provision hereof or of any other Loan Document to the contrary notwithstanding, neither the Borrower nor any other Person is providing (or intends to provide) the holder of the Warrant Certificate with any Guarantee or assurance of economic return or against risk of loss in respect of the Warrant Certificate or any Equity Interests issuable upon exercise thereof, and neither the term “Obligations” nor the term “Warrant Obligations” shall be deemed to include, directly or indirectly, any such Guarantee or assurance.
“Waypoint Product Acquisition” means the Acquisition by the Borrower of the Assets (as defined in the Waypoint Product Acquisition Agreement) from Waypoint Health Innovations, LLC.
“Waypoint Product Acquisition Agreement” means that certain Assignment Agreement, dated as of November 30, 2021, between the Borrower and Waypoint Health Innovations, LLC, pursuant to which the Borrower has agreed, among other things, to acquire the Assets (as defined in the Waypoint Product Acquisition Agreement.
“Withdrawal Liability” means, at any time, any liability incurred (whether or not assessed) by any ERISA Affiliate and not yet satisfied or paid in full at such time with respect to any Multiemployer Plan pursuant to Section 4201 of ERISA.
“Withholding Agent” means the Borrower, any other Obligor and the Administrative Agent, as applicable.
“Write-Down and Conversion Powers” means, (a) with respect to any EEA Resolution Authority, the write-down and conversion powers of such EEA Resolution Authority from time to time under the Bail-In Legislation for the applicable EEA Member Country, which write-down and conversion powers are described in the EU Bail-In Legislation Schedule, and (b) with respect to the United Kingdom, any powers of the applicable Resolution Authority under the Bail-In Legislation to cancel, reduce, modify or change the form of a liability of any UK Financial Institution or any contract or instrument under which that liability arises, to convert all or part of that liability into shares, securities or obligations of that person or any other person, to provide that any such contract or instrument is to have effect as if a right had been exercised under it or to suspend any obligation in respect of that liability or any of the powers under that Bail-In Legislation that are related to or ancillary to any of those powers.
1.02 Accounting Terms and Principles. Unless otherwise specified, all accounting terms used in each Loan Document shall be interpreted, and all accounting determinations and computations thereunder (including under Section 10 and any definitions used in such calculations) shall be made, in accordance with GAAP. Unless otherwise expressly provided, all financial covenants and defined financial terms shall be computed on a consolidated basis for Holdings and its Subsidiaries, in each case without duplication. If the Borrower requests an amendment to any provision hereof to eliminate the effect of (a) any change in GAAP or the application thereof or (b) the issuance of any new accounting rule or guidance or in the application thereof, in either case, occurring after the date of this Agreement, then the Lenders and Borrower agree that they will negotiate in good faith amendments to the provisions of this Agreement that are directly affected by such change or issuance with the intent of having the respective positions of the Lenders and Borrower after such change or issuance conform as nearly as possible to their respective positions as of the date of this Agreement and, until any such amendments have been agreed upon, (i) the provisions in this Agreement shall be calculated as if no such change or issuance has occurred and (ii) the Borrower shall provide to the Lenders a written reconciliation in form and substance reasonably satisfactory to the Lenders, between calculations of any baskets and other requirements hereunder before and after giving effect to such change or issuance.
1.03 Interpretation. For all purposes of this Agreement, except as otherwise expressly provided herein or unless the context otherwise requires,
(a) the terms defined in this Agreement include the plural as well as the singular and vice versa;
(b) words importing gender include all genders;
(c) any reference to a Section, Annex, Schedule or Exhibit refers to a Section of, or Annex, Schedule or Exhibit to, this Agreement;
(d) any reference to “this Agreement” refers to this Agreement, including all Annexes, Schedules and Exhibits hereto, and the words herein, hereof, hereto and hereunder and words of similar import refer to this Agreement and its Annexes, Schedules and Exhibits as a whole and not to any particular Section, Annex, Schedule, Exhibit or any other subdivision;
(e) references to days, months and years refer to calendar days, months and years, respectively;
(f) all references herein to “include” or “including” shall be deemed to be followed by the words “without limitation”;
(g) the word “from” when used in connection with a period of time means “from and including” and the word “until” means “to but not including”;
(h) the words “asset” and “property” shall be construed to have the same meaning and effect and to refer broadly to any and all assets and properties, whether tangible or intangible, real or personal, including cash, securities, rights under contractual obligations and permits and any right or interest in any such assets or property;
(i) accounting terms not specifically defined herein (other than “property” and “asset”) shall be construed in accordance with GAAP;
(j) where any provision in this Agreement or any other Loan Document refers to an action to be taken by any Person, or an action which such Person is prohibited from taking, such provision shall be applicable whether such action is taken directly or indirectly;
(k) the word “will” shall have the same meaning as the word “shall”;
(l) references to any Lien granted or created hereunder or pursuant to any other Loan Document securing any Obligations shall be deemed to be a Lien for the benefit of the Secured Parties; and
(m) references to any Law will include all statutory and regulatory provisions amending, consolidating, replacing, supplementing or interpreting such Law from time to time.
Unless otherwise expressly provided herein, references to organizational documents, agreements (including the Loan Documents) and other contractual instruments shall be deemed to include all
subsequent amendments, restatements, extensions, supplements and other modifications thereto permitted by the Loan Documents.
If any obligation to pay any amount pursuant to the terms and conditions of any Loan Document falls due on a day which is not a Business Day, then such required payment date shall be extended to the immediately following Business Day. For the purposes of calculations made pursuant to the terms of this Agreement or otherwise for purposes of compliance herewith, GAAP will be deemed to treat operating leases in a manner consistent with their current treatment under GAAP as in effect on the date of this Agreement, notwithstanding any modifications or interpretive changes thereto that may occur thereafter.
1.04 Divisions. For all purposes under the Loan Documents, in connection with any division or plan of division under Delaware law (or any comparable event under a different jurisdiction’s laws): (i) if any asset, right, obligation or liability of any Person becomes the asset, right, obligation or liability of a different Person, then it shall be deemed to have been transferred from the original Person to the subsequent Person, and (ii) if any new Person comes into existence, such new Person shall be deemed to have been organized on the first date of its existence by the holders of its Equity Interests at such time.
SECTION 2
The Commitments and the Loans
2.01 Loans.
(a) On the terms and subject to the conditions of this Agreement, each Lender agrees to make a Tranche 1 Loan to the Borrower, in a single Borrowing on the Closing Date, in an aggregate principal amount for all Lenders not to exceed $30,000,000.
(b) No amounts repaid or prepaid with respect to any Loan may be reborrowed.
(c) Any term or provision hereof (or of any other Loan Document) to the contrary notwithstanding, Loans made to the Borrower will be denominated solely in Dollars and will be repayable solely in Dollars and no other currency.
2.02 Borrowing Procedures. At least three (3), but not more than seven (7), Business Days prior to any proposed Borrowing Date (or at least one (1), but not more than five (5) Business Day(s) prior to the Borrowing on the Closing Date), the Borrower shall deliver to the Administrative Agent an irrevocable Borrowing Notice, which notice, if received by the Administrative Agent on a day that is not a Business Day or after 12:00 noon (New York City time) on a Business Day, shall be deemed to have been delivered on the next Business Day.
2.03 Notes. If requested by any Lender, any Loan of such Lender shall be evidenced by one or more Notes. The Borrower shall prepare, execute and deliver to the Lender such Notes in the form attached hereto as Exhibit A.
2.04 Use of Proceeds. The Borrower shall use the proceeds of the Loans for purposes of (i) in the case of Tranche 1 Loans, the repayment of the Refinanced Debt and certain other Indebtedness, (ii) working capital and general corporate purposes, and (iii) without duplication, the payment of
fees and expenses associated with this Agreement and the other Loan Documents and the transactions contemplated hereby and thereby.
SECTION 3
PAYMENTS OF PRINCIPAL AND INTEREST
3.01 Repayments and Prepayments Generally; Application.
(a) There will be no scheduled repayments of principal on the Loans prior to May 31, 2024. Thereafter, on each Payment Date occurring prior to the scheduled Maturity Date (and commencing on the fourth anniversary of the Closing Date), the Borrower shall make a payment of principal on the Loans in an amount equal to three percent (3.00%) of the aggregate original principal amount of all Loans made on and after the Closing Date and as of such Payment Date. On the Maturity Date the Borrower shall repay the entire remaining outstanding balance of the Loans in full and in cash.
(b) The Borrower agrees that all amounts payable hereunder or under any other Loan Document, in respect of any Loans, fees or interest accrued or accruing thereon, or any other Obligations, shall be repaid and prepaid solely in Dollars pursuant to the terms of this Section 3. Except as otherwise provided in this Agreement, proceeds of each payment (including each repayment and prepayment of Loans) by the Borrower shall be deemed to be made ratably to the Lenders in accordance with their respective Proportionate Shares of the Loans being repaid or prepaid.
3.02 Interest.
(a) Interest Generally. The outstanding principal amount of the Loans, as well as the amount of all other outstanding Obligations, shall accrue interest at the Interest Rate on and from the Closing Date. The Administrative Agent’s determination of the Interest Rate shall be binding on the Borrower, its Subsidiaries and the Lenders in the absence of manifest error.
(b) Default Interest. Notwithstanding the foregoing, upon notice from the Administrative Agent upon the occurrence and during the continuance of any Event of Default, the Applicable Margin shall increase automatically by four percent (4.0%) per annum (the Interest Rate, as increased pursuant to this Section 3.02(b), being the “Default Rate”). If any Obligation is not paid when due under any applicable Loan Document, the amount thereof shall accrue interest at the Default Rate.
(c) Interest Payment Dates. Accrued interest on the Loans shall be payable in cash, in arrears, on each Payment Date with respect to the most recently completed Interest Period, and upon the payment or prepayment of the Loans (on the principal amount being so paid or prepaid); provided that interest payable at the Default Rate, or any accrued interest not paid on the Maturity Date, shall be payable from time to time in cash on demand by the Administrative Agent until paid in full.
3.03 Prepayments; Early Prepayment Fee.
(a) Optional Prepayments.
(i) Subject to prior written notice pursuant to clause (a)(ii) below and the payment of the Early Prepayment Fee pursuant to clause (c) below, the Borrower shall have the right to optionally prepay, in whole or in part, the outstanding principal amount of the Loans on any Business Day (a “Prepayment Date”); provided that in addition to such prepaid principal amount and the Early Prepayment Fee, the Borrower shall also make payment in full in cash on such Prepayment Date of all accrued but unpaid interest on the principal amount of the Loans being prepaid (such aggregate amount, the “Prepayment Price”).
(ii) A notice of optional prepayment shall be effective only if received by the Administrative Agent not later than 2:00 p.m. (New York City time) on a date not less than three (3) (nor more than five (5)) Business Days prior to the proposed Prepayment Date. Each notice of optional prepayment shall specify the proposed Prepayment Date, the principal amount of the Loans to be prepaid, the amount of accrued and unpaid interest that will be paid on the Prepayment Date, and, in reasonable detail, a calculation of the Early Prepayment Fee payable on such Prepayment Date in connection with such proposed prepayment.
(b) Mandatory Prepayments. Within five (5) Business Days of the receipt of Net Cash Proceeds from the occurrence of any Casualty Event or Specified Asset Sale, Holdings and the Borrower shall apply an amount equal to one hundred percent (100%) of the Net Cash Proceeds received by Holdings or any of its Subsidiaries with respect to such Casualty Event or Specified Asset Sale, as the case may be, to (i) the prepayment of outstanding Loans and (ii) the payment of accrued and unpaid interest on the principal amount of the Loans being prepaid and the payment of the Early Prepayment Fee. Such Net Cash Proceeds shall be allocated to such prepayment and payments such that the full amount of principal, interest and prepayment fees payable hereunder shall be paid with such Net Cash Proceeds. Notwithstanding the foregoing, so long as no Default has occurred and is continuing or shall immediately result therefrom, if, within three (3) Business Days following the occurrence of any such Casualty Event or Specified Asset Sale, a Responsible Officer of the Borrower delivers to the Administrative Agent a notice to the effect that the Borrower or the applicable Subsidiary intends to apply the Net Cash Proceeds from such Casualty Event or Specified Asset Sale, to repair, refurbish, restore, replace or rebuild the asset subject to such Casualty Event or Specified Asset Sale or to the cost of purchase or constructing other assets useful in the business of the Borrower or its Subsidiaries, then such Net Cash Proceeds of such Casualty Event or Specified Asset Sale may be applied for such purpose in lieu of such mandatory prepayment otherwise required pursuant to this clause (b) to the extent such Net Cash Proceeds of such Casualty Event or Asset Sale are actually applied for such purpose; provided that, in the event that Net Cash Proceeds have not been so applied within one hundred and eighty (180) days following the occurrence of such Casualty Event or Specified Asset Sale (or, if within such 180-day period the Borrower enters into a binding commitment to purchase or acquire such assets, within ninety (90) days from entering into such binding commitment), the Borrower shall make a mandatory prepayment of the Loans in an aggregate amount equal to one hundred percent (100%) of the unused balance of such Net Cash Proceeds received by Holdings or any of its Subsidiaries with respect to such Casualty Event or Specified Asset Sale, as the case may be, together with payment of accrued and unpaid interest on the principal amount of the Loans being so prepaid and the applicable Early Prepayment Fee, with such amount of Net Cash Proceeds being allocated to the prepayment of principal, the payment of accrued and unpaid interest on such principal amount of the Loans being prepaid and the payment of the Early Prepayment Fee such that the full payable with respect to such mandatory prepayment is paid with such unused balance of Net Cash Proceeds.
(c) Early Prepayment Fee. Without limiting the foregoing, whenever any prepayment of Loans is made hereunder pursuant to Section 3.03(a) or Section 3.03(b) or otherwise, whether voluntary, involuntary, as a result of a Default, acceleration or otherwise, the Early Prepayment Fee shall be payable in full in cash on the applicable Prepayment Date for such prepayment.
(d) Application. Proceeds of any prepayment made pursuant to clauses (a) or (b) above shall be applied in the following order of priority, with proceeds being applied to a succeeding level of priority only if amounts owing pursuant to the immediately preceding level of priority have been paid in full in cash; provided that all such applications to Lenders shall be made in accordance with their respective Proportionate Shares:
(i) first, to the payment of that portion of the Obligations payable to the Administrative Agent constituting fees, indemnities, costs, expenses, and other amounts then due and owing (including fees and disbursements and other charges of counsel payable under Section 14.03);
(ii) second, to the payment of that portion of the Obligations payable to the Lenders constituting fees (other than the Early Prepayment Fee), indemnities, expenses, and other amounts then due and owing including fees and disbursements and other charges of counsel payable under Section 14.03 ratably among them in proportion to the respective amounts described in this clause (ii) payable to them;
(iii) third, to the payment of any accrued and unpaid interest and any fees then due and owing;
(iv) fourth, to the payment of unpaid principal of the Loans;
(v) fifth, to the payment of any Early Prepayment Fee then due and payable;
(vi) sixth, to the payment in full of all other Obligations then due and payable to the Administrative Agent and the Lenders, ratably among them in proportion to the respective amounts described in this clause (vi) payable to them; and
(vii) seventh, to Holdings, the Borrower or such other Persons as may lawfully be entitled to or directed by the Borrower to receive the remainder.
3.04 Closing Fee. On the Closing Date, the Borrower shall pay to the Administrative Agent (for the benefit of the Lenders) a fee equal to 1.50% of the aggregate Commitments of all Lenders (the “Closing Fee”). Upon receipt of payment from the Borrower, the Administrative Agent will promptly thereafter distribute like funds relating to any such payment to the Lenders pro rata on the basis of each Lender’s Proportionate Share. Once paid by the Borrower, the Closing Fee shall be non-refundable.
SECTION 4
PAYMENTS, ETC.
4.01 Payments.
(a) Payments Generally. Each payment of principal, interest and other amounts to be made by the Obligors under this Agreement or any other Loan Document shall be made (i) in
Dollars, in immediately available funds, without deduction, set off or counterclaim, to the Administrative Agent, for the account of the respective Lenders to which such payment is owed, to the deposit account of the Administrative Agent designated by the Administrative Agent by notice to Holdings, the Borrower, and (ii) not later than 2:00 p.m. (New York City time) on the date on which such payment is due (each such payment made after such time on such due date shall be deemed to have been made on the next succeeding Business Day).
(b) Application of Payments. All such payments referenced in clause (a) above shall be applied as set forth in Section 3.03(d) above.
(c) Non-Business Days. If the due date of any payment under this Agreement (whether in respect of principal, interest, fees, costs or otherwise) would otherwise fall on a day that is not a Business Day, such date shall be extended to the next succeeding Business Day; provided that if such next succeeding Business Day would fall after the Maturity Date, payment shall be made on the immediately preceding Business Day.
4.02 Computations. All computations of interest and fees hereunder shall be computed on the basis of a year of three hundred and sixty (360) days and actual days elapsed during the period for which payable.
4.03 Set-Off.
(a) Set-Off Generally. Upon the occurrence and during the continuance of any Event of Default, the Administrative Agent, each of the Lenders and each of their Affiliates is hereby authorized at any time and from time to time, to the fullest extent permitted by law, to set off and apply any and all deposits (general or special, time or demand, provisional or final) at any time held and other indebtedness at any time owing by the Administrative Agent, any Lender and any of their Affiliates to or for the credit or the account of any Obligor against any and all of the Obligations, whether or not such Person shall have made any demand and although such obligations may be unmatured. Any Person exercising rights of set off hereunder agrees promptly to notify the Borrower after any such set-off and application, provided that the failure to give such notice shall not affect the validity of such set-off and application. The rights of the Administrative Agent, the Lenders and each of their Affiliates under this Section 4.03 are in addition to other rights and remedies (including other rights of set-off) that such Persons may have.
(b) Exercise of Rights Not Required. Nothing contained in Section 4.03(a) shall require the Administrative Agent, any Lender or any of their Affiliates to exercise any such right or shall affect the right of such Persons to exercise, and retain the benefits of exercising, any such right with respect to any other indebtedness or obligation of any Obligor.
(c) Payments Set Aside. To the extent that any payment by or on behalf of any Obligor is made to the Administrative Agent or any Lender, or the Administrative Agent, any Lender or any Affiliate of the foregoing exercises its right of setoff, and such payment or the proceeds of such setoff or any part thereof is subsequently invalidated, declared to be fraudulent or preferential, set aside or required (including pursuant to any settlement entered into by the Administrative Agent, such Lender or such Affiliate in its discretion) to be repaid to a trustee, receiver or any other party, in connection with any Insolvency Proceeding or otherwise, then (i) to
the extent of such recovery, the obligation or part thereof originally intended to be satisfied shall be revived and continued in full force and effect as if such payment had not been made or such setoff had not occurred, and (ii) each Lender severally agrees to pay to the Administrative Agent upon demand its applicable share (without duplication) of any amount so recovered from or repaid by the Administrative Agent, plus interest thereon from the date of such demand to the date such payment is made at a rate per annum equal to the Federal Funds Effective Rate from time to time in effect.
SECTION 5
YIELD PROTECTION, ETC.
5.01 Additional Costs.
(a) Changes in Law Generally. If, on or after the date hereof (or, with respect to any Lender, such later date on which such Lender becomes party to this Agreement), the adoption of any Law, or any change in any Law, or any change in the interpretation or administration thereof by any court or other Governmental Authority charged with the interpretation or administration thereof, or compliance by the Administrative Agent or any of the Lenders (or its lending office) with any request or directive (whether or not having the force of law) of any such Governmental Authority, shall impose, modify or deem applicable any reserve (including any such requirement imposed by the Board of Governors of the Federal Reserve System), special deposit, contribution, insurance assessment or similar requirement, in each case that becomes effective after the date hereof (or, with respect to any Lender, such later date on which such Lender becomes party to this Agreement), against assets of, deposits with or for the account of, or credit extended by, a Lender (or its lending office) or other Recipient or shall impose on a Lender (or its lending office) or other Recipient any other condition affecting the Loans or the Commitment, and the result of any of the foregoing is to increase the cost to such Lender or such other Recipient of making or maintaining the Loans, or to reduce the amount of any sum received or receivable by such Lender or other Recipient under this Agreement or any other Loan Document, or subject any Lender or other Recipient to any Taxes on its loans, loan principal, commitments or other obligations, or its deposits, reserves, other liabilities or capital (if any) attributable thereto (other than (i) Indemnified Taxes, (ii) Taxes described in clauses (ii) through (iv) of the definition of “Excluded Taxes” and (iii) Connection Income Taxes), then the Borrower shall pay to such Lender or other Recipient within five (5) Business Days after demand such additional amount or amounts as will compensate such Lender for such increased cost or reduction.
(b) Change in Capital Requirements. If a Lender shall have reasonably determined that, on or after the date hereof (or, with respect to any Lender, such later date on which such Lender becomes party to this Agreement), the adoption of any applicable Law regarding capital adequacy, or any change therein, or any change in the interpretation or administration thereof by any Governmental Authority charged with the interpretation or administration thereof, or any request or directive regarding capital adequacy (whether or not having the force of law) of any such Governmental Authority, in each case that becomes effective after the date hereof (or, with respect to any Lender, such later date on which such Lender becomes party to this Agreement), has or would have the effect of reducing the rate of return on capital of a Lender (or its parent) as a consequence of a Lender’s obligations hereunder or the Loans to a level below that which a Lender (or its parent) could have achieved but for such adoption, change, request or directive by
an amount reasonably deemed by it to be material, then the Borrower shall pay to such Lender within five (5) Business Days after demand such additional amount or amounts as will compensate such Lender (or its parent) for such reduction.
(c) Notification by Lender. Each Lender shall promptly notify the Borrower of any event of which it has knowledge, occurring after the date hereof (or, with respect to any Lender, such later date on which such Lender becomes party to this Agreement), which will entitle such Lender to compensation pursuant to this Section 5.01. Before giving any such notice pursuant to this Section 5.01(c) such Lender shall designate a different lending office if such designation (x) will, in the reasonable judgment of such Lender, avoid the need for, or reduce the amount of, such compensation and (y) will not, in the reasonable judgment of such Lender, be materially disadvantageous to such Lender. A certificate of such Lender claiming compensation under this Section 5.01, setting forth in reasonable detail the computation of the additional amount or amounts to be paid to it hereunder, shall be conclusive and binding on the Borrower in the absence of manifest error.
(d) Delays in Requests. Failure or delay on the part of any Lender to demand compensation pursuant to the foregoing provisions of this Section shall not constitute a waiver of such Lender’s right to demand such compensation; provided that the Borrower shall not be required to compensate a Lender pursuant to this Section 5.01 for any increased costs that occur or arise in any fiscal year of such Lender unless such Lender notifies the Borrower thereof within sixty (60) days following the receipt by such Lender of its annual audited financial statements for such fiscal year.
(e) Other Changes. Notwithstanding anything herein to the contrary, (x) the Dodd-Frank Wall Street Reform and Consumer Protection Act and all requests, rules, guidelines or directives thereunder or issued in connection therewith and (y) all requests, rules, guidelines or directives promulgated by the Bank for International Settlements, the Basel Committee on Banking Supervision (or any successor or similar authority) or the United States or foreign regulatory authorities, in each case pursuant to Basel III, shall in each case be deemed to constitute a change in Law for all purposes of this Section 5.01, regardless of the date enacted, adopted or issued.
5.02 Illegality. Notwithstanding any other provision of this Agreement, if, on or after the date hereof (or, with respect to any Lender, such later date on which such Lender becomes party to this Agreement), the adoption of or any change in any applicable Law or in the interpretation or application thereof by any competent Governmental Authority shall make it unlawful for a Lender or its lending office to make or maintain the Loans (and, in the reasonable opinion of such Lender, the designation of a different lending office would either not avoid such unlawfulness or would be disadvantageous to such Lender), then such Lender shall promptly notify the Borrower thereof, following which (i) such Lender’s Commitment shall be suspended until such time as such Lender may again make and maintain the Loans hereunder and (ii) if such Law shall so mandate, the Loans shall be prepaid by the Borrower on or before such date as shall be mandated by such Law in an amount equal to the Prepayment Price applicable on such Prepayment Date in accordance with Section 3.03(a).
5.03 Taxes.
(a) Payments Free of Taxes. Any and all payments by or on account of any Obligation shall be made without deduction or withholding for any Taxes, except as required by applicable Law. If any applicable Law (as determined in the reasonable discretion of an applicable Withholding Agent) requires the deduction or withholding of any Tax from any such payment by a Withholding Agent, then the applicable Withholding Agent shall be entitled to make such deduction or withholding and shall timely pay the full amount deducted or withheld to the relevant Governmental Authority in accordance with applicable Law and, if such Tax is an Indemnified Tax, then the sum payable by such Obligor shall be increased as necessary so that after such deduction or withholding has been made (including such deductions and withholdings applicable to additional sums payable under this Section 5.03) the applicable Recipient receives an amount equal to the sum it would have received had no such deduction or withholding been made.
(b) Payment of Other Taxes by the Borrower. The Borrower shall timely pay to the relevant Governmental Authority in accordance with applicable Law, or at the option of the Administrative Agent or each Lender, timely reimburse it for the payment of any Other Taxes.
(c) Evidence of Payments. As soon as practicable after any payment of Taxes by the Borrower to a Governmental Authority pursuant to this Section 5, the Borrower shall deliver to the Administrative Agent the original or a certified copy of a receipt issued by such Governmental Authority evidencing such payment, a copy of the return reporting such payment or other evidence of such payment reasonably satisfactory to the Administrative Agent.
(d) Indemnification by Holdings and the Borrower. Holdings and the Borrower shall indemnify and reimburse each Recipient, within three (3) Business Days after demand therefor, for the full amount of any Indemnified Taxes (including Indemnified Taxes imposed or asserted on or attributable to amounts payable under this Section 5) payable or paid by such Recipient or required to be withheld or deducted from a payment to such Recipient and any reasonable expenses arising therefrom or with respect thereto, whether or not such Indemnified Taxes were correctly or legally imposed or asserted by the relevant Governmental Authority. A certificate as to the amount of such payment or liability delivered to the Borrower by a Lender (with a copy to the Administrative Agent), or by the Administrative Agent on its own behalf or on behalf of a Lender shall be conclusive absent manifest error.
(e) Indemnification by the Lenders. Each Lender shall severally indemnify the Administrative Agent, within ten (10) days after demand therefor, for (i) any Indemnified Taxes attributable to such Lender (but only to the extent that the Borrower has not already indemnified the Administrative Agent for such Indemnified Taxes and without limiting the obligation of the Borrower to do so), (ii) any Taxes attributable to such Lender’s failure to comply with the provisions of Section 14.05(g) relating to the maintenance of a Participant Register, and (iii) any Excluded Taxes attributable to such Lender, in each case, that are payable or paid by the Administrative Agent in connection with any Loan Document, and any reasonable expenses arising therefrom or with respect thereto, whether or not such Taxes were correctly or legally imposed or asserted by the relevant Governmental Authority. A certificate as to the amount of such payment or liability delivered to any Lender by the Administrative Agent shall be conclusive absent manifest error. Each Lender hereby authorizes the Administrative Agent to set off and apply any and all amounts at any time owing to such Lender under any Loan Document or
otherwise payable by the Administrative Agent to the Lender from any other source against any amount due to the Administrative Agent under this clause (e).
(f) Status of Lenders.
(i) Any Lender that is entitled to an exemption from or reduction of withholding Tax with respect to payments made under any Loan Document shall deliver to the Borrower and the Administrative Agent at the time or times reasonably requested by the Borrower or the Administrative Agent, such properly completed and executed documentation reasonably requested by the Borrower or the Administrative Agent as will permit such payments to be made without withholding or at a reduced rate of withholding; provided that, other than in the case of U.S. federal withholding Taxes, such Lender has received written notice from the Borrower advising it of the availability of such exemption or reduction and containing all applicable documentation. In addition, any Lender, if reasonably requested by the Borrower or the Administrative Agent shall deliver such other documentation prescribed by applicable Law as reasonably requested by the Borrower or the Administrative Agent as will enable the Borrower or the Administrative Agent to determine whether or not such Lender is subject to backup withholding or information reporting requirements. Notwithstanding anything to the contrary in the preceding two sentences, the completion, execution and submission of such documentation (other than such documentation set forth in Section 5.03(f)(ii)(A), (ii)(B), and (ii)(D)) shall not be required if in such Lender’s reasonable judgment such completion, execution or submission would subject such Lender to any material unreimbursed cost or expense or would materially prejudice the legal or commercial position of such Lender.
(ii) Without limiting the generality of the foregoing, if the Borrower is a U.S. Person:
(A) any Lender that is a U.S. Person shall deliver to the Borrower and the Administrative Agent on or prior to the date on which such Lender becomes a Lender under this Agreement (and from time to time thereafter upon the reasonable request of the Borrower or the Administrative Agent), executed copies of IRS Form W-9 (or successor form) certifying that such Lender is exempt from U.S. federal backup withholding tax;
(B) any Foreign Lender shall, to the extent it is legally entitled to do so, deliver to the Borrower and the Administrative Agent (in such number of copies as shall be requested by the recipient) on or prior to the date on which such Foreign Lender becomes a Lender under this Agreement (and from time to time thereafter upon the reasonable request of the Borrower or the Administrative Agent), whichever of the following is applicable:
(1) in the case of a Foreign Lender claiming the benefits of an income tax treaty to which the United States is a party (x) with respect to payments of interest under any Loan Document, executed copies of IRS Form W-8BEN or IRS Form W-8BEN-E as applicable (or successor forms) establishing an exemption from, or reduction of, U.S. federal withholding Tax pursuant to the “interest” article of such tax treaty and (y) with respect to any other applicable payments under any Loan Document, IRS Form W-8BEN or IRS Form W-8BEN-E as applicable (or successor forms) establishing an exemption from, or reduction of, U.S. federal withholding Tax pursuant to the “business profits” or “other income” article of such tax treaty;
(2) executed copies of IRS Form W-8ECI (or successor form);
(3) in the case of a Foreign Lender claiming the benefits of the exemption for portfolio interest under Section 881(c) of the Code, (x) a certificate substantially in the form of Exhibit D-1 to the effect that such Foreign Lender is not a “bank” within the meaning of Section 881(c)(3)(A) of the Code, a “10 percent shareholder” of the Borrower within the meaning of Section 871(h)(3)(B) of the Code, or a “controlled foreign corporation” related to the Borrower as described in Section 881(c)(3)(C) of the Code (a “U.S. Tax Compliance Certificate”) and (y) executed copies of IRS Form W-8BEN or IRS Form W-8BEN-E as applicable (or successor forms); or
(4) to the extent a Foreign Lender is not the beneficial owner, executed copies of IRS Form W-8IMY (or successor form), accompanied by IRS Form W-8ECI (or successor form), IRS Form W-8BEN or IRS Form W-8BEN-E (or successor form), a U.S. Tax Compliance Certificate, substantially in the form of Exhibit D-2 or Exhibit D-3, IRS Form W-9 (or successor form), and/or other certification documents from each beneficial owner, as applicable; provided that if the Foreign Lender is a partnership and one or more direct or indirect partners of such Foreign Lender are claiming the portfolio interest exemption, such Foreign Lender may provide a U.S. Tax Compliance Certificate substantially in the form of Exhibit D-4 on behalf of each such direct and indirect partner.
(C) any Foreign Lender shall, to the extent it is legally entitled to do so, deliver to the Borrower and the Administrative Agent (in such number of copies as shall be requested by the recipient) on or prior to the date on which such Foreign Lender becomes a Lender under this Agreement (and from time to time thereafter upon the reasonable request of the Borrower or the Administrative Agent), executed copies of any other form prescribed by applicable Law as a basis for claiming exemption from or a reduction in U.S. federal withholding Tax, duly completed, together with such supplementary documentation as may be prescribed by such applicable Law to permit the Borrower or the Administrative Agent to determine the withholding or deduction required to be made; and
(D) if a payment made to a Lender under any Loan Document would be subject to U.S. federal withholding Tax imposed by FATCA if such Lender were to fail to comply with the applicable reporting requirements of FATCA (including those contained in Section 1471(b) or 1472(b) of the Code, as applicable), such Lender shall deliver to the Borrower and the Administrative Agent at the time or times prescribed by law and at such time or times reasonably requested by the Borrower or the Administrative Agent such documentation prescribed by applicable Law (including as prescribed by Section 1471(b)(3)(C)(i) of the Code) and such additional documentation reasonably requested by the Borrower or the Administrative Agent as may be necessary for the Borrower and the Administrative Agent to comply with their obligations under FATCA and to determine that such Lender has complied with such Recipient’s obligations under FATCA or to determine the amount, if any, to deduct and withhold from such payment under FATCA. Solely for purposes of this clause (D), “FATCA” shall include any amendments made to FATCA after the date of this Agreement.
Each Recipient agrees that if any form or certification it previously delivered expires or becomes obsolete or inaccurate in any respect, it shall update such form or certification or promptly notify the Borrower and the Administrative Agent in writing of its legal inability to do so.
(g) Treatment of Certain Tax Benefits. If any party to this Agreement determines, in its sole discretion exercised in good faith, that it has received a refund of any Taxes as to which it has been indemnified pursuant to this Section 5.03 (including by the payment of additional amounts pursuant to this Section 5.03), it shall pay to the indemnifying party an amount equal to such refund (but only to the extent of indemnity payments made under this Section 5.03 with respect to the Taxes giving rise to such refund), net of all out-of-pocket expenses (including Taxes) of such indemnified party and without interest (other than any interest paid by the relevant Governmental Authority with respect to such refund). Such indemnifying party, upon the request of such indemnified party, shall repay to such indemnified party the amount paid over pursuant to this Section 5.03(g) (plus any penalties, interest or other charges imposed by the relevant Governmental Authority) if such indemnified party is required to repay such refund to such Governmental Authority. Notwithstanding anything to the contrary in this Section 5.03(g), in no event will the indemnified party be required to pay any amount to an indemnifying party pursuant to this Section 5.03(g) the payment of which would place the indemnified party in a less favorable net after-Tax position than the indemnified party would have been in if the Tax subject to indemnification and giving rise to such refund had not been deducted, withheld or otherwise imposed and the indemnification payments or additional amounts with respect to such Tax had never been paid. This Section 5.03(g) shall not be construed to require any indemnified party to make available its Tax returns (or any other information relating to its Taxes that it deems confidential) to the indemnifying party or any other Person.
(h) Survival. Each party’s obligations under this Section 5.03 shall survive the resignation or replacement of the Administrative Agent or any assignment of rights by, or the replacement of, a Lender, the termination of the Commitments and the repayment, satisfaction or discharge of all Obligations under any Loan Document.
5.04 Mitigation Obligations. If the Borrower is required to pay any Indemnified Taxes or additional amounts to any Lender or to any Governmental Authority for the account of any Lender pursuant to Section 5.01 or Section 5.03, then such Lender shall (at the request of the Borrower) use commercially reasonable efforts to designate a different lending office for funding or booking its Loans hereunder or to assign and delegate its rights and obligations hereunder to another of its offices, branches or Affiliates if, in the sole, reasonable judgment of such Lender, such designation or assignment and delegation would (i) eliminate or reduce amounts payable pursuant to Section 5.01 or this Section 5.03, as the case may be, in the future, (ii) not subject such Lender to any unreimbursed cost or expense and (iii) not otherwise be disadvantageous to such Lender. The Borrower hereby agrees to pay all reasonable costs and expenses incurred by any Lender in connection with any such designation or assignment and delegation.
SECTION 6
CONDITIONS PRECEDENT
6.01 Conditions to the Borrowing of the Tranche 1 Loans. The obligation of the Closing Date Lender to make its Tranche 1 Loan on the Closing Date shall be subject to the execution and
delivery of this Agreement by the parties hereto, the delivery of a Borrowing Notice as required pursuant to Section 2.02, the delivery of a funds flow memorandum summarizing, in reasonable detail, the use of proceeds of the Tranche 1 Loans, and the prior or concurrent satisfaction (or waiver thereof by the Administrative Agent) of each of the conditions precedent set forth below in this Section 6.01.
(a) Secretary’s Certificate, Etc. The Administrative Agent shall have received from each Obligor party to a Loan Document on the Closing Date:
(i) a copy of a good standing certificate or the equivalent thereof, dated a date reasonably close to the Closing Date, for each such Person and
(ii) a certificate, dated as of the Closing Date, duly executed and delivered by such Person’s secretary or assistant secretary, managing member, general partner or equivalent, as to:
(A) resolutions of each such Person’s Board then in full force and effect authorizing the execution, delivery and performance of each Loan Document and the Transactions, to be executed and delivered by such Person;
(B) the incumbency and signatures of those of its officers, managing member or general partner or equivalent authorized to act with respect to each Loan Document to be executed and delivered by such Person; and
(C) true and complete copies of each Organic Document of such Person and copies thereof;
which certificates shall be in form and substance reasonably satisfactory to the Administrative Agent and upon which the Administrative Agent and the Lenders may conclusively rely until they shall have received a further certificate of the secretary, assistant secretary, managing member, general partner or equivalent of any such Person cancelling or amending the prior certificate of such Person.
(b) Information Certificate. The Administrative Agent shall have received a fully completed Information Certificate, in form and substance reasonably satisfactory to the Administrative Agent, dated as of the Closing Date, duly executed and delivered by a Responsible Officer of the Borrower, which is true and correct as of the Closing Date. All documents and agreements required to be appended to the Information Certificate, if any, shall be in form and substance reasonably satisfactory to the Administrative Agent and the Lenders, shall have been executed and delivered by the requisite parties and shall be in full force and effect.
(c) Closing Date Certificate. The following statements shall be true and correct, and the Administrative Agent shall have received a certificate, dated as of the Closing Date and in form and substance reasonably satisfactory to the Administrative Agent, duly executed and delivered by a Responsible Officer of the Borrower certifying that: (i) both immediately before and after giving effect to the Borrowing on the Closing Date, (x) the representations and warranties set forth in each Loan Document that are qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct; provided that to the extent that such representations and warranties
specifically refer to an earlier date, they shall be true and correct as of such earlier date, (y) the representations and warranties set forth in each Loan Document that are not qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct in all material respects; provided that to the extent that such representations and warranties specifically refer to an earlier date, they shall be true and correct in all material respects as of such earlier date, and (z) no Default or Event of Default has occurred and is continuing, or could reasonably be expected to result from the Borrowing of the Tranche 1 Loans, or the consummation of any Transactions contemplated to occur on the Closing Date, and (ii) all of the conditions set forth in this Section 6.01 have been satisfied (or waived in writing by the Administrative Agent). All documents and agreements required to be appended to the certificate delivered pursuant to this Section 6.01(c), if any, shall be in form and substance reasonably satisfactory to the Administrative Agent, shall have been executed and delivered by the requisite parties, and shall be in full force and effect.
(d) Delivery of Notes. The Administrative Agent shall have received a Note in favor of each Lender evidencing such Lender’s Tranche 1 Loan, duly executed and delivered by a Responsible Officer of the Borrower.
(e) Financial Information, Etc. The Administrative Agent shall have received:
(i) audited consolidated financial statements of the Borrower and its Subsidiaries for the fiscal year ended December 31, 2019; and
(ii) unaudited consolidated balance sheets of the Borrower and its Subsidiaries for each fiscal quarter ended after December 31, 2019 and at least twenty (20) Business Days prior to the Closing Date, together with the related consolidated statement of operations for each such fiscal quarter.
(f) Minimum Liquidity Compliance. The Administrative Agent shall have received evidence reasonably satisfactory to it that, immediately after giving effect to the Borrowing on the Closing Date, the Borrower will be in compliance with the covenant set forth in Section 10.01.
(g) Closing Date Warrant Certificate. The Closing Date Lender shall have received an executed counterpart of a Warrant Certificate, exercisable into 775,000 shares of the Borrower’s Series C preferred stock (or, in accordance with the terms of the Warrant Certificate, the Borrower’s Series D preferred stock), duly executed and delivered by the Borrower.
(h) Insurance. The Administrative Agent shall have received certificates of insurance evidencing that the insurance required to be maintained pursuant to Section 8.05 is in full force and effect, together with endorsements naming the Administrative Agent, for the benefit of the Lenders, as additional insured and loss payee thereunder, in each case, in form and substance reasonably satisfactory to the Administrative Agent.
(i) Solvency. The Administrative Agent shall have received a solvency certificate substantially in the form of Exhibit I, duly executed and delivered by the chief financial or accounting Responsible Officer of the Borrower, dated as of the Closing Date, in form and substance reasonably satisfactory to the Administrative Agent.
(j) Security Documents. The Administrative Agent shall have received executed counterparts of all Security Documents of all Obligors, each dated as of the Closing Date, duly executed and delivered by each such Obligor, together with:
(i) The delivery of all certificates (in the case of Equity Interests that are securities (as defined in the UCC)) evidencing the issued and outstanding capital securities owned by the Borrower and each Subsidiary that are required to be pledged under such Security Documents, which certificates in each case shall be accompanied by undated instruments of transfer duly executed in blank, or, in the case of Equity Interests that are uncertificated securities (as defined in the UCC), confirmation and evidence reasonably satisfactory to the Administrative Agent and the Lenders that the security interest required to be pledged therein under such Security Documents has been transferred to and perfected by the Administrative Agent for the benefit of the Secured Parties in accordance with Articles 8 and 9 of the NY UCC and all Laws otherwise applicable to the perfection of the pledge of such Equity Interests;
(ii) financing statements naming each Obligor as a debtor and the Administrative Agent as the secured party, or other similar instruments, registrations, or documents, in each case suitable for filing, filed under the UCC (or equivalent law) of all jurisdictions as may be necessary or, in the reasonable opinion of the Administrative Agent, desirable to perfect the Liens of the Secured Parties pursuant to such Security Documents;
(iii) UCC-3 termination statements, as may be necessary to release all Liens (other than Permitted Liens) and other rights of any Person in any collateral described in the Security Documents previously granted by any Person;
(iv) all Short-Form IP Security Agreements and Real Property Security Documents required to be provided under the Security Agreement, each dated as of the Closing Date, duly executed and delivered by the Borrower.
(k) Lien Searches. The Administrative Agent shall be reasonably satisfied with Lien searches regarding the Borrower and its Subsidiaries made within thirty (30) days prior to the Borrowing of the Tranche 1 Loans.
(l) Controlled Accounts. The Administrative Agent shall have received evidence reasonably satisfactory to it that all Deposit Accounts, Securities Accounts, Commodities Accounts, lockboxes or other similar accounts of each Obligor are Controlled Accounts.
(m) Opinions of Counsel. The Administrative Agent shall have received one or more legal opinions, dated as of the Closing Date and addressed to the Administrative Agent and the Lenders, from independent legal counsel to the Borrower and if necessary, other legal counsel satisfactory to the Administrative Agent, in each case, in form and substance reasonably acceptable to the Administrative Agent.
(n) Payoff of Indebtedness. To the extent applicable, all outstanding Indebtedness not permitted hereby of the Borrower and its Subsidiaries, including the Refinanced Debt, together with all accrued and unpaid interest and related fees, costs and expenses, shall be indefeasibly paid in full, and the Administrative Agent shall have received executed payoff letters, in form and substance reasonably satisfactory to the Administrative Agent, evidencing such indefeasible payment in full (and irrevocable termination) of such outstanding Indebtedness (including the
Refinanced Debt) and the termination and release of all guarantees and Liens, if any, in connection therewith.
(o) Material Adverse Change. Since December 31, 2019, no Material Adverse Change shall have occurred.
(p) Anti-Terrorism Laws. The Administrative Agent shall have received, as applicable, all documentation and other information that it has requested at least three (3) Business Days prior to the Closing Date and that is required by bank regulatory authorities under applicable “know your customer” and anti-money laundering rules and regulations, including the Patriot Act and the Beneficial Ownership Regulation.
(q) All Other Loan Documents. The Administrative Agent shall have received all other Loan Documents in form and substance reasonably satisfactory to the Administrative Agent and its counsel, and the Administrative Agent and its counsel shall have received all information, approvals, resolutions, opinions, documents or instruments as the Administrative Agent and its counsel may reasonably request.
(r) Satisfactory Legal Form. All documents, including any attachments or appendices thereto, executed, delivered or submitted pursuant hereto by or on behalf of the Borrower or any of its respective Subsidiary Guarantors shall be reasonably satisfactory in form and substance to the Administrative Agent and its counsel, and the Administrative Agent and its counsel shall have received all information, approvals, resolutions, opinions, documents or instruments as the Administrative Agent or its counsel may reasonably request.
(s) Governmental Approvals and Third Party Consents. The Administrative Agent shall have received evidence that the Borrower and the applicable Subsidiaries have obtained all Governmental Approvals and third party permits, licenses, approvals and consents necessary in connection with the execution, delivery and performance of the Loan Documents, the consummation of the Transactions or the operation and conduct of its business and ownership of its properties (including its Product Commercialization and Development Activities).
(t) Closing Fee, Expenses, Etc. The Administrative Agent shall have received for its account and the account of each Lender, the Closing Fee, together with all other fees, costs and expenses due and payable pursuant to the Proposal Letter and Section 14.03, including all closing costs and fees and all unpaid reasonable expenses of the Administrative Agent and the Lenders incurred in connection with the Transactions (including the Administrative Agent’s legal fees and expenses) in excess of the Expense Deposit.
(u) Material Agreement. The Administrative Agent shall have received true and complete executed copies of all Material Agreements, together with all amendments, waivers, exhibits, schedules and annexes thereto. The Administrative Agent shall have received a certificate from a Responsible Officer of the Borrower certifying that each such Material Agreement, together with all amendments, waivers, exhibits, schedules and annexes thereto, are true and complete and in full force and effect as of the Closing Date. The Administrative Agent shall be satisfied in its reasonable discretion with the terms and conditions of each Material Agreement (together with all amendments, waivers, exhibits, schedules and annexes thereto).
(v) Option to Invest. The Borrower shall have entered into a binding agreement with the Lenders, in form and substance reasonably satisfactory to the Lenders, pursuant to which the Borrower shall grant to the Lenders an irrevocable option to invest up to $10,000,000 in the Equity Interests of the Borrower.
6.02 Conditions to Effectiveness. The effectiveness of this Agreement shall be subject to the execution and delivery of this Agreement by the parties hereto, and the prior or concurrent satisfaction (or waiver thereof by the Administrative Agent) of each of the conditions precedent in the Restatement Amendment and set forth below in this Section 6.02.
(a) Secretary’s Certificate, Etc. The Administrative Agent shall have received from each Obligor party to a Loan Document on the Effective Date:
(i) a copy of a good standing certificate or the equivalent thereof, dated a date reasonably close to the Effective Date, for each such Person and
(ii) a certificate, dated as of the Effective Date, duly executed and delivered by such Person’s secretary or assistant secretary, managing member, general partner or equivalent, as to:
(A) resolutions of each such Person’s Board then in full force and effect authorizing the execution, delivery and performance of each Loan Document and the Transactions, to be executed and delivered by such Person;
(B) the incumbency and signatures of those of its officers, managing member or general partner or equivalent authorized to act with respect to each Loan Document to be executed and delivered by such Person; and
(C) true and complete copies of each Organic Document of such Person and copies thereof;
which certificates shall be in form and substance reasonably satisfactory to the Administrative Agent and upon which the Administrative Agent and the Lenders may conclusively rely until they shall have received a further certificate of the secretary, assistant secretary, managing member, general partner or equivalent of any such Person cancelling or amending the prior certificate of such Person.
(b) Information Certificate. The Administrative Agent shall have received a fully completed Information Certificate, in form and substance reasonably satisfactory to the Administrative Agent, dated as of the Effective Date, duly executed and delivered by a Responsible Officer of Holdings and the Borrower, which is true and correct as of the Effective Date. All documents and agreements required to be appended to the Information Certificate, if any, shall be in form and substance reasonably satisfactory to the Administrative Agent and the Lenders, shall have been executed and delivered by the requisite parties and shall be in full force and effect.
(c) Effective Date Certificate. The following statements shall be true and correct, and the Administrative Agent shall have received a certificate, dated as of the Effective Date and in
form and substance reasonably satisfactory to the Administrative Agent, duly executed and delivered by a Responsible Officer of Holdings and the Borrower certifying that: (i) both immediately before and after giving effect to Effective Date, (x) the representations and warranties set forth in each Loan Document that are qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct; provided that to the extent that such representations and warranties specifically refer to an earlier date, they shall be true and correct as of such earlier date, (y) the representations and warranties set forth in each Loan Document that are not qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct in all material respects; provided that to the extent that such representations and warranties specifically refer to an earlier date, they shall be true and correct in all material respects as of such earlier date, and (z) no Default has occurred and is continuing, or could reasonably be expected to result from the consummation of any Transactions contemplated to occur on the Effective Date, and (ii) all of the conditions set forth in this Section 6.02 have been satisfied (or waived in writing by the Administrative Agent). All documents and agreements required to be appended to the certificate delivered pursuant to this Section 6.02(c), if any, shall be in form and substance reasonably satisfactory to the Administrative Agent, shall have been executed and delivered by the requisite parties, and shall be in full force and effect.
(d) Solvency. The Administrative Agent shall have received a solvency certificate substantially in the form of Exhibit I, duly executed and delivered by the chief financial or accounting Responsible Officer of Holdings, dated as of the Effective Date, in form and substance reasonably satisfactory to the Administrative Agent.
(e) Lien Searches. The Administrative Agent shall be reasonably satisfied with Lien searches regarding Holdings and its Subsidiaries made within a recent date prior to the Effective Date.
(f) Controlled Accounts. The Administrative Agent shall have received evidence reasonably satisfactory to it that all Deposit Accounts, Securities Accounts, Commodities Accounts, lockboxes or other similar accounts of each Obligor are Controlled Accounts.
(g) Amended and Restated Security Agreement and Intercompany Subordination Agreement. The Administrative Agent shall have received executed counterparts of the Security Agreement and the Intercompany Subordination Agreement, together with:
(i) the delivery of all certificates (in the case of Equity Interests that are securities (as defined in the UCC)) evidencing the issued and outstanding capital securities owned by Holdings and each Subsidiary that are required to be pledged under such Security Documents, which certificates in each case shall be accompanied by undated instruments of transfer duly executed in blank, or, in the case of Equity Interests that are uncertificated securities (as defined in the UCC), confirmation and evidence reasonably satisfactory to the Administrative Agent and the Lenders that the security interest required to be pledged therein under such Security Documents has been transferred to and perfected by the Administrative Agent for the benefit of the Secured Parties in accordance with Articles 8 and 9 of the NY UCC and all Laws otherwise applicable to the perfection of the pledge of such Equity Interests; and
(ii) financing statements naming each Obligor as a debtor and the Administrative Agent as the secured party, or other similar instruments, registrations, or documents, in each case suitable for filing, filed under the UCC (or equivalent law) of all jurisdictions as may be necessary or, in the
reasonable opinion of the Administrative Agent, desirable to perfect the Liens of the Secured Parties pursuant to such Security Documents.
(h) Opinions of Counsel. The Administrative Agent shall have received one or more legal opinions, dated as of the Effective Date and addressed to the Administrative Agent and the Lenders, from independent legal counsel to Holdings and the Borrower and if necessary, other legal counsel satisfactory to the Administrative Agent, in each case, in form and substance reasonably acceptable to the Administrative Agent.
(i) Costs and Expenses, Etc. The Administrative Agent shall have received for its account and the account of each Lender all reasonable and documented fees, costs and expenses due and payable to them pursuant to Section 14.03 (including, in each case, the Administrative Agent’s and each Lender’s reasonable and documented legal fees and out-of-pocket expenses).
(j) Satisfactory Legal Form. All documents, including any attachments or appendices thereto, executed, delivered or submitted pursuant hereto by or on behalf of any Obligor shall be reasonably satisfactory in form and substance to the Administrative Agent and its counsel, and the Administrative Agent and its counsel shall have received all information, approvals, resolutions, opinions, documents or instruments as the Administrative Agent or its counsel may reasonably request.
SECTION 7
REPRESENTATIONS AND WARRANTIES
The Obligors hereby jointly and severally represent and warrant to the Administrative Agent and each Lender that:
7.01 Power and Authority. Each Obligor and each of its Subsidiaries (i) is duly organized and validly existing under the laws of its jurisdiction of organization, (ii) has all requisite corporate or other power, and has all Governmental Approvals necessary to own its assets and carry on its business as now being or as proposed to be conducted, including all requisite Regulatory Approvals, (iii) is qualified to do business and is in good standing in all jurisdictions in which the nature of the business conducted by it makes such qualification necessary and where failure so to qualify, individually or in the aggregate, could reasonably be expected to result in a Material Adverse Effect, and (iv) has full power, authority and legal right to enter into and perform its obligations under each of the Loan Documents to which it is a party and, in the case of the Borrower, to borrow the Loans hereunder.
7.02 Authorization; Enforceability. Each Transaction to which an Obligor or any of its Subsidiaries is a party (or to which it or any of its assets or properties is subject) are within such Person’s corporate or other powers and have been duly authorized by all necessary corporate action including, if required, approval by all necessary holders of Equity Interests. This Agreement has been duly executed and delivered by each Obligor and constitutes, and each of the other Loan Documents to which it is a party when executed and delivered by such Obligor, will constitute, a legal, valid and binding obligation of such Person, enforceable against such Person in accordance with its terms, except as such enforceability may be limited by (i) bankruptcy, insolvency, reorganization, moratorium or similar laws of general applicability affecting the enforcement of
creditors’ rights and (ii) the application of general principles of equity (regardless of whether such enforceability is considered in a proceeding in equity or at law).
7.03 Governmental and Other Approvals; No Conflicts. No authorization or approval or other action by, and no notice to or filing with, any Governmental Authority or any other Person (other than those that have been duly obtained or made and which are in full force and effect) is required for the due execution, delivery or performance by any Obligor of each Loan Document to which it is a party, except for filings and recordings in respect of perfecting or recording the Liens created pursuant to the Security Documents. The execution, delivery and performance by each Obligor of each Loan Document to which it is a party will not (i) violate or conflict with any Law, (ii) violate or conflict with any Organic Document of any Obligor, (iii) violate or conflict with any Governmental Approval of any Governmental Authority, (iv) violate or result in a default under any Material Agreement binding upon an Obligor or any of its Subsidiaries that, individually or in the aggregate, could reasonably be expected to result in a Material Adverse Effect or (v) result in the creation or imposition of any Lien (other than Permitted Liens) on any asset of such Obligor. Each Obligor, its Subsidiaries and their respective properties and businesses are in compliance in all material respects with all applicable Laws (including Healthcare Laws) and Governmental Approvals applicable to such Person and its properties or businesses, as the case may be. The Loan Documents and the Transactions contemplated therein do not conflict with or violate any Organic Documents, Material Agreements or other Contracts, or any Governmental Approval of any Governmental Authority binding upon Holdings, the Borrower or any its Subsidiaries. None of the Transactions will result in the creation or imposition of any Lien (other than Permitted Liens and Liens in favor of the Administrative Agent and the Lenders) on any asset of Holdings or any of its Subsidiaries.
7.04 Financial Statements; Material Adverse Change.
(a) Financial Statements. The Borrower has heretofore furnished to the Administrative Agent and the Lenders certain consolidated financial statements as provided for in Section 6.01(e). Such financial statements, and all other financial statements delivered by the Borrower pursuant hereto (whether prior to the Closing Date or otherwise) present fairly, in all material respects, the consolidated financial position and results of operations and cash flows of Holdings and its Subsidiaries as of such dates and for such periods in accordance with GAAP, subject to year-end audit adjustments and the absence of footnotes in the case of the statements of the type described in Section 8.01(b). Neither Holdings nor any of its Subsidiaries has any material contingent liabilities or unusual forward or long-term commitments not disclosed in the aforementioned financial statements.
(b) No Material Adverse Change. Since December 31, 2019, there has been no Material Adverse Change.
7.05 Properties.
(a) Property Generally. With respect to all real and personal assets and properties of each Obligor and each of its Subsidiaries (other than Intellectual Property which is covered in clause (c) below), such Obligor and each of its Subsidiaries has good and marketable fee simple title to, or valid leasehold interests in, all its real and personal property, whether tangible or
intangible, material to its business, including all Products and all properties and assets owned by such Obligor and its Subsidiaries relating to its Products or Product Commercialization and Development Activities, subject only to Permitted Liens and except as could not reasonably be expected to (i) interfere with its ability to conduct its business as currently conducted or to utilize such properties for their intended purposes or (ii) prevent or interfere with the ability of such Obligor or any of its Subsidiaries to conduct its business in the ordinary course.
(b) Products. Schedule 7.05(b) contains a complete and accurate list and description (in reasonable detail) of all Products (set forth on an Obligor-by-Obligor or Subsidiary-by-Subsidiary basis, as the case may be).
(c) Intellectual Property.
(i) Schedule 7.05(c) contains, with respect to each Obligor and each of its Subsidiaries (set for forth on a Subsidiary-by-Subsidiary basis):
(A) a complete and accurate list of all applied for, issued, or registered Patents, owned by each Obligor or any of its Subsidiaries or exclusively licensed to such Obligor any of its Subsidiaries to conduct all Product Commercialization and Development Activities relating to the Products, including the jurisdiction and patent number;
(B) a complete and accurate list of all applied for or registered Trademarks, owned by each Obligor or any of its Subsidiaries or exclusively licensed to such Obligor or any of its Subsidiaries to conduct all Product Commercialization and Development Activities relating to the Products, including the jurisdiction, trademark application or registration number and the application or registration date, which would qualify as Material Intellectual Property;
(C) a complete and accurate list of all applied for or registered Copyrights, owned by each Obligor or any of its Subsidiaries or exclusively licensed to such Obligor or any of its Subsidiaries to conduct all Product Commercialization and Development Activities relating to the Products, which would qualify as Material Intellectual Property; and
(D) a summary of Technical Information necessary for all Product Commercialization and Development Activities relating to the Products, in each case which would qualify as Material Intellectual Property.
(ii) An Obligor or one of its Subsidiaries, as applicable, is the absolute registered beneficial owner of all right, title and interest in and to such Person’s Intellectual Property that it owns (including, without limitation, the Intellectual Property indicated on Schedule 7.05(c) as being owned solely by an Obligor or one of its Subsidiaries), with no breaks in chain of title and with good and marketable title, free and clear of any Liens or Claims of any kind whatsoever other than Permitted Liens, and, to the extent any such Intellectual Property qualifies as Material Intellectual Property, Holdings and its applicable Subsidiaries have the right to exercise its rights under such Intellectual Property in the
ordinary course of their respective businesses as currently conducted or proposed to be conducted. Without limiting the foregoing, and except as set forth on Schedule 7.05(c):
(A) other than as permitted by Section 9.09, no Obligor or any of its Subsidiaries has transferred ownership of any of its Material Intellectual Property, in whole or in part, to any Person who is not an Obligor;
(B) other than (1) customary restrictions in in-bound licenses of Intellectual Property and non-disclosure agreements, (2) as set forth on Schedule 7.05(c) or (3) as would have been or is permitted by Section 9.18, there are no judgments, covenants not to sue, permits, grants, licenses, Liens (other than Permitted Liens), Claims, or other agreements or arrangements relating to or otherwise adversely affecting any Material Intellectual Property, including any development, submission, services, research, license or support agreements, which bind, obligate or otherwise restrict an Obligor or any of its Subsidiaries with respect to any Material Intellectual Property;
(C) other than as set forth on Schedule 7.05(c), the use by an Obligor or any of its Subsidiaries of any of its respective Material Intellectual Property in the ordinary course of such Person’s businesses does not breach, violate infringe or interfere with or constitute a misappropriation of any valid rights arising under any Intellectual Property of any other Person;
(D) other than as set forth on Schedule 7.05(c), (1) there are no pending or, to Holdings’ or the Borrower’s knowledge, threatened Claims against an Obligor or any of its Subsidiaries relating to any Material Intellectual Property, including any Claims of adverse ownership, invalidity, infringement, misappropriation, violation or other opposition to or conflict with such Intellectual Property; and (2) during the five (5) years preceding the date this representation is being made no Obligor nor any of its Subsidiaries has received any written notice from, or Claim by, any other Person asserting that the business of such Obligor or any of its Subsidiaries, the use of Material Intellectual Property by such Obligor or any of its Subsidiaries, or any Product Commercialization and Development Activities with respect to any Product, infringes upon, violates or constitutes a misappropriation of, or may infringe upon, violate or constitute a misappropriation of, or otherwise interfere with, or otherwise offer a license with respect to, any Intellectual Property of any such other Person, in each case, in any material respect, which have not been finally resolved;
(E) none of Holdings or the Borrower has knowledge that any Material Intellectual Property is being infringed, violated, misappropriated or otherwise used by any other Person without the express authorization of Holdings, the Borrower or their applicable Subsidiary; and, without limiting the foregoing, no Obligor nor any of its Subsidiaries has put any other Person on notice of actual or potential infringement, violation or misappropriation of any Material Intellectual Property, and no Obligor nor
any of its Subsidiaries has initiated the enforcement of any Claim with respect to any Material Intellectual Property;
(F) all relevant current and former employees and contractors of an Obligor and each of its Subsidiaries have executed written confidentiality and invention assignment Contracts with such Obligor or its Subsidiary, as applicable, that irrevocably assigns to such Obligor or such Subsidiary, as applicable, or its designee all rights of such employees and contractors to any Inventions relating to the business of such Obligor or such Subsidiary, as applicable;
(G) the Obligor Intellectual Property, together with the lawful use of open source, freeware and third party licensed Intellectual Property by the Obligors and their Subsidiaries, is all the Intellectual Property necessary for the operation of the business of the Obligors and their Subsidiaries as it is currently conducted;
(H) each Obligor and each of its Subsidiaries have made available to the Administrative Agent accurate and complete copies of all Material Agreements relating to Material Intellectual Property; and
(I) each Obligor and each of its Subsidiaries have taken reasonable precautions to protect the secrecy, confidentiality and value of its Material Intellectual Property consisting of trade secrets and confidential information.
(iii) With respect to the Material Intellectual Property consisting of Patents, except as set forth on Schedule 7.05(c), and without limiting the representations and warranties in Section 7.05(c)(ii):
(A) none of Holdings, the Borrower or any Subsidiary is aware of any Claim asserted in writing by any other Person that any of the issued claims in such Patents owned by any Obligor or its applicable Subsidiary or exclusively licensed to any Obligor or any of its Subsidiaries to conduct all Product Commercialization and Development Activities relating to the Products, is not valid or enforceable;
(B) each inventor, who was an employee of an Obligor or any of its Subsidiaries, named in such Patents has executed written Contracts with an Obligor or its predecessor-in-interest that properly and irrevocably assigns to such Obligor or its predecessor-in-interest all of such inventor’s rights, title and interest to any of the Inventions claimed in such Patents;
(C) all such Patents are in good standing and none of the Patents, or the Inventions claimed in any such Patent, have been dedicated to the public;
(D) to Holdings’ and Borrower’s knowledge as of the date of this Agreement, all prior art material to such Patents was adequately disclosed to or considered by the respective patent offices during prosecution of such Patents;
(E) subsequent to the issuance of such Patents, no Obligor nor any of its Subsidiaries nor any of their respective predecessors-in-interest, has filed any disclaimer or made or permitted any other voluntary reduction in the scope of the claims set forth in such Patents;
(F) to the best of Holdings’ or Borrower’s knowledge, neither Holdings nor the Borrower is aware of any Claim by any Person that any allowable or allowed claim in such Patents is subject to any competing conception claims of allowable or allowed subject matter of any patent applications or patents of any third party, and such claims have not been the subject of any interference, and are not and have not been the subject of any re-examination, opposition or any other post-grant proceedings, and none of Holdings nor any of its Subsidiaries has knowledge of any Claim by any Person for any such interference, re-examination, opposition, inter partes review, post grant review, or any other post-grant proceedings;
(G) none of the Patents owned by or exclusively licensed to an Obligor or any of its Subsidiaries have ever been finally adjudicated to be invalid, unpatentable or unenforceable for any reason in any administrative, arbitration, judicial or other proceeding, and, with the exception of publicly available documents in the applicable patent office recorded with respect to any such Patents, no Obligor nor any of its Subsidiaries has received any notice asserting that such Patents are invalid, unpatentable or unenforceable; if any of such Patents is terminally disclaimed to another patent or patent application, all patents and patent applications subject to such terminal disclaimer are included in the Collateral;
(H) no Obligor nor any of its Subsidiaries has received a written opinion from patent counsel which concludes that a challenge to the validity or enforceability of any Patents is more likely than not to succeed;
(I) to Holdings’ or Borrower’s knowledge as of the date of this Agreement, no Obligor nor any of its Subsidiaries or any prior owner of any Patent, or any of their respective agents or representatives, have engaged in any conduct, or omitted to perform any necessary act, the result of which would invalidate or render unpatentable or unenforceable any Patent; and
(J) all maintenance fees, annuities, and the like due or payable on or with respect to any Patents have been timely paid or the failure to so pay could not, individually or in the aggregate, reasonably be expected to result in a Material Adverse Change.
(iv) The Obligors own or hold rights to all Material Intellectual Property to conduct all Product Commercialization and Development Activities relating to the Products.
7.06 No Actions or Proceedings.
(a) Litigation. Except as specified on Schedule 7.06(a), there is no litigation, investigation or proceeding pending or, to the knowledge of each Obligor, threatened in writing, with respect to each Obligor or any of its Subsidiaries by or before any Governmental Authority or arbitrator that (i) could, individually or in the aggregate, reasonably be expected to result in a Material Adverse Effect or a Material Regulatory Event, or (ii) involves this Agreement, any other Loan Document, or the Transactions.
(b) Environmental Matters. The operations and property of each Obligor comply with all applicable Environmental Laws, except to the extent the failure to so comply (either individually or in the aggregate) could not reasonably be expected to result in Material Adverse Effect.
(c) Labor Matters. There are no strikes, lockouts or other material labor disputes against each Obligor or any of its Subsidiaries or, to each Obligor’s knowledge, threatened in writing against or affecting such Obligor or any of its Subsidiaries, and no significant unfair labor practice complaint is pending against such Obligor or any of its Subsidiaries or, to the knowledge of the Borrower, threatened against any of them before any Governmental Authority. Except as set forth on Schedule 7.06(c), no Obligor nor any of its Subsidiaries is a party to any collective bargaining agreements or similar Contracts, no union representation exists on any facilities of such Obligor or any of its Subsidiaries and no Obligor nor any of its Subsidiaries has any knowledge of any union organizing activities that are taking place.
7.07 Compliance with Laws and Agreements.
(a) Each Obligor and each of its Subsidiaries is in compliance with all applicable Laws and all Contracts binding upon it or its property, except (other than with respect to Material Intellectual Property, as covered in Section 7.05(c)) where the failure to do so could not, individually or in the aggregate, reasonably be expected to result in a Material Adverse Effect or a Material Regulatory Event. No Default has occurred and is continuing, or will occur as a result of any Borrowing hereunder.
(b) Without limiting the generality of the foregoing, each Obligor and each of its Subsidiaries is in material compliance with all applicable Healthcare Laws and Healthcare Permits, and no Obligor nor any such Subsidiary has received written notice by a Governmental Authority of any material violation (or of any investigation, audit, or other proceeding involving allegations of any violation) of any Healthcare Laws, and no such investigation, inspection, audit or other proceeding involving allegations of any such violation has been, to the knowledge of such Obligor or any Subsidiary, threatened in writing.
(c) Each physician, other licensed healthcare professional, or any other Person who is in a position to refer patients or other business to an Obligor or any of its Subsidiaries (collectively, a “Referral Source”) who has a direct ownership, investment, or financial interest in such Obligor or any such Subsidiary paid fair market value for such ownership, investment or financial interest; any ownership or investment returns distributed to any Referral Source is in proportion to such Referral Source’s ownership, investment or financial interest; and no preferential treatment or more favorable terms were or are offered to such Referral Source compared to investors or owners who are not in a position to refer patients or other business. No Obligor nor any of its Subsidiaries,
directly or indirectly, has or will guarantee a loan, make a payment toward a loan or otherwise subsidize a loan for any Referral Source including, without limitation, any loans related to financing the Referral Source’s ownership, investment or financial interest in such Obligor or any such Subsidiary.
(d) Without limiting the generality of the foregoing, except where noncompliance individually or in the aggregate could not reasonably be expected to result in a Material Regulatory Event:
(i) all financial relationships between or among an Obligor and its Subsidiaries, on the one hand, and any Referral Source, on the other hand (A) comply with all applicable Healthcare Laws including, without limitation, the Federal Anti-Kickback Statute, the Stark Law and other applicable anti-kickback and self-referral laws, whether U.S. or non-U.S.; (B) reflect fair market value, have commercially reasonable terms, and were negotiated at arm’s length; and (C) do not obligate the Referral Source to purchase, use, recommend or arrange for the use of any products or services of such Obligor or any of its Subsidiaries; and
(ii) without limiting the generality of the foregoing, the Obligor and each of its Subsidiaries is in material compliance with all applicable Healthcare Laws, and none of the Obligors nor any of their Subsidiaries has received written notice by a Governmental Authority of any material violation (or of any investigation, audit, or other proceeding involving allegations of any violation) of any Healthcare Laws, and except as set forth on Schedule 7.07(d), no such investigation, inspection, audit or other proceeding involving allegations of any such violation has been, to the knowledge of Holdings or the Borrower, threatened in writing.
(e) No Obligor nor any of its Subsidiaries is debarred or excluded from participation under any state or federal health care program, including any state or federal workers’ compensation programs.
(f) No Obligor nor any of its Subsidiaries is a party to any corporate integrity agreements, deferred prosecution agreements, monitoring agreements, consent decrees, settlement orders, or similar agreements with, or imposed by, any Governmental Authority.
7.08 Taxes. Except as set forth on Schedule 7.08, each Obligor and each of its Subsidiaries has timely filed or caused to be filed all tax returns and reports required to have been filed and has paid or caused to be paid all Taxes required to have been paid by it, except Taxes that are being contested in good faith by appropriate proceedings diligently conducted and for which such Borrower or such Subsidiary, as applicable, has set aside on its books adequate reserves with respect thereto in accordance with GAAP.
7.09 Full Disclosure. None of the reports, financial statements, certificates or other written information furnished by or on behalf of the Obligors to the Administrative Agent or any Lender in connection with the negotiation of this Agreement and the other Loan Documents or delivered hereunder or thereunder (as modified or supplemented by other information so furnished), taken as a whole, contains any material misstatement of material fact or omits to state any material fact necessary to make the statements therein, in the light of the circumstances under which they were made, not misleading; provided that, with respect to projected financial information, each Obligor represents only that such information was prepared in good faith based upon assumptions believed to be reasonable at the time.
7.10 Investment Company Act and Margin Stock Regulation.
(a) Investment Company Act. No Obligor nor any of its Subsidiaries is an “investment company” as defined in, or subject to regulation under, the Investment Company Act of 1940, as amended.
(b) Margin Stock. No Obligor nor any of its Subsidiaries is engaged principally, or as one of its important activities, in the business of extending credit for the purpose, whether immediate, incidental or ultimate, of buying or carrying Margin Stock, and no part of the proceeds of the Loans will be used to buy or carry any Margin Stock in violation of Regulation T, Regulation U or Regulation X.
7.11 Solvency. Each Obligor and its Subsidiaries, on a consolidated basis, are, and, immediately after giving effect to the Borrowing and the use of proceeds thereof, will be, Solvent. Each Subsidiary is, and immediately after giving effect to the Borrowing and the use of proceeds thereof, will be Solvent.
7.12 Equity Holders, Subsidiaries and Other Investments.
(a) Set forth on Schedule 7.12(a) is a complete and correct list of all holders of Equity Interests of each Obligor, setting forth the name of such holder, the series or class of Equity Interest of such Obligor held by such holder, and the fully diluted percentage ownership of such Obligor beneficially held by such holder.
(b) Set forth on Schedule 7.12(b) is a complete and correct list of all direct and indirect Subsidiaries of each Obligor. Each such Subsidiary is duly organized and validly existing under the jurisdiction of its organization shown in Schedule 7.12(b), and the percentage ownership by such Obligor of each such Subsidiary thereof is as shown in Schedule 7.12(b).
(c) Set forth on Schedule 7.12(c) is a complete and correct list of all other Equity Interests owned or held by each Obligor or any of its direct or indirect Subsidiaries in any Person that does not qualify as a direct or indirect Subsidiary of such Obligor. Schedule 7.12(c) also sets forth, in reasonable detail, the type of Equity Interest held by each Obligor in such other Person and the fully-diluted percentage ownership held beneficially by each Obligor or one or more of its Subsidiaries, as the case may be, in such other Person.
7.13 Indebtedness and Liens. Set forth on Schedule 7.13(a) is a complete and correct list of all outstanding Indebtedness of each Obligor and each of its Subsidiaries. Set forth on Schedule 7.13(b) is a complete and correct list (in reasonable detail) of all Indebtedness outstanding on the Closing Date that will be repaid and satisfied in full on the Closing Date with proceeds of the Tranche 1 Loan. Schedule 7.13(c) is a complete and correct list of all Liens granted by the Obligors and their Subsidiaries with respect to their respective property and outstanding as of the Effective Date other than the Liens granted pursuant to the Loan Documents.
7.14 Material Agreements. Set forth on Schedule 7.14 is a complete and correct list of (i) each Material Agreement and (ii) each Contract creating or evidencing any Material Indebtedness. Accurate and complete copies of each Contract disclosed on such schedule have been made available to the Administrative Agent. No Obligor nor any of its Subsidiaries is in default under
any such Material Agreement or Contract creating or evidencing any Material Indebtedness, and no Obligor has knowledge of any default by any counterparty to such Material Agreement or Contract and there are no pending or, to the Obligor’s knowledge, threatened Claims against any Obligor or any of its Subsidiaries asserted in writing by any other Person relating to any Material Agreements, including any Claims of breach or default under any such Material Agreements. No Obligor nor any of its Subsidiaries has received any written information from, or Claim by, any Person that any Material Agreement is breached or is in default. There are no outstanding (and, the Obligors do not have knowledge of, any threatened in writing) disputes or disagreements with respect to any Material Agreement. Except as otherwise disclosed on Schedule 7.14, all material vendor purchase agreements and provider Contracts of each Obligor and its Subsidiaries, and all Material Agreements including a grant of rights under any Intellectual Property to each Obligor or any of its Subsidiaries are in full force and effect without material modification from the form in which the same were disclosed to the Lenders.
7.15 Restrictive Agreements. Except as set forth on Schedule 7.15, no Obligor nor any of its Subsidiaries is subject to any Restrictive Agreement, except those permitted under Section 9.11.
7.16 Real Property. Except as set forth on Schedule 7.16, no Obligor nor any of its Subsidiaries owns or leases (as tenant thereof) any real property.
7.17 Pension Matters. Schedule 7.17 sets forth a complete and correct list of, and that separately identifies, (i) all Title IV Plans, (ii) all Multiemployer Plans and (iii) all material Benefit Plans. Each Benefit Plan, and each trust thereunder, intended to qualify for tax exempt status under Section 401 or 501 of the Code or other applicable Law so qualifies. Except for those that could not, in the aggregate, reasonably be expected to have a Material Adverse Effect, (x) each Benefit Plan and Foreign Pension Plan is in compliance with all applicable provisions of ERISA, the Code or other applicable Law, (y) there are no existing or pending or, to the knowledge of Holdings or the Borrower, threatened Claims (other than routine claims for benefits in the normal course of business), sanctions, actions, lawsuits or other proceedings or investigation involving any Benefit Plan to which an Obligor or any Subsidiary thereof incurs or otherwise has or could reasonably be expected to have an obligation or any liability or Claim and (z) no ERISA Event is reasonably expected to occur. Each Obligor and each of its ERISA Affiliates has met all applicable requirements under the ERISA Funding Rules with respect to each Title IV Plan, and no waiver of the minimum funding standards under the ERISA Funding Rules has been applied for or obtained. No ERISA Event has occurred or is reasonably expected to occur in connection with which obligations and liabilities (contingent or otherwise) remain outstanding. No ERISA Affiliate would have any Withdrawal Liability as a result of a complete withdrawal from any Multiemployer Plan on the date this representation is made.
7.18 Priority of Obligations; Collateral; Security Interest. Except as set forth on Schedule 7.13(a), the Obligations constitute the sole senior secured obligations and sole Indebtedness of Holdings and its Subsidiaries. No monetary Obligation arising hereunder or under any Loan Document, or arising in connection herewith or therewith, is subordinated to any other Indebtedness. Each Security Document is effective to create in favor of the Secured Parties a legal, valid and enforceable security interest in the Collateral subject to such Security Document, each such security interest is legal, valid and enforceable, and each such security interest is perfected
on a first-priority basis (subject to Permitted Liens that may apply to specific items of Collateral permitted pursuant to Section 9.02) and secures the Obligations.
7.19 Regulatory Approvals.
(a) Each Obligor and each of its Subsidiaries hold, and will continue to hold, either directly or through licensees and agents, all Regulatory Approvals, including all Healthcare Permits, necessary or required for such Obligor and each of its Subsidiaries to conduct their respective operations and businesses, including all Product Commercialization and Development Activities, in the manner currently conducted and proposed to be conducted in the ordinary course of business.
(b) Set forth on Schedule 7.19(b) is a complete and accurate list of all Regulatory Approvals of the type described in Section 7.19(a) above, which schedule sets forth the applicable Obligor or Subsidiary that holds such Regulatory Approval. All such Regulatory Approvals are (i) legally and beneficially owned or held exclusively by such Obligor or one of its Subsidiaries, as the case may be, free and clear of all Liens other than Permitted Liens, (ii) validly registered and on file with the applicable Regulatory Authority, in compliance with all registration, filing and maintenance requirements (including any fee requirements) thereof, and (iii) valid and in full force and effect with the applicable Regulatory Authority. All required notices, registrations, listing, supplemental applications or notification reports (including field alerts or other reports of adverse experiences) and other required filings have been filed with the appropriate Regulatory Authority, and all such filings are in compliance with all applicable Laws.
7.20 Transactions with Affiliates. Except as set forth on Schedule 7.20, no Obligor nor any of its Subsidiaries has entered into, renewed, extended or been a party to, any transaction (including the purchase, sale, lease, transfer or exchange of property or assets of any kind or the rendering of services of any kind, other than services as a director, officer or employee) with any Affiliate during the three-year period prior to the Closing Date except on an arm’s length basis.
7.21 Sanctions. No Obligor nor any of its Subsidiaries, nor, to the knowledge of Holdings, the Borrower and each other Obligor, any of their respective directors, officers, or employees nor, to the knowledge of Holdings or the Borrower, any agents or other Persons acting on behalf of any of the foregoing (i) is currently the target of any Sanctions, (ii) is located, organized or residing in any Designated Jurisdiction, (iii) is or has been (within the previous five (5) years) engaged in any transaction with, or for the benefit of, any Person who is now or was then the target of Sanctions or who is located, organized or residing in any Designated Jurisdiction or (iv) is or has ever been in violation of or subject to an investigation relating to Sanctions. No Loan, nor the proceeds from any Loan, has been or will be used, directly or indirectly, to lend, contribute or provide to, or has been or will be otherwise made available to fund, any activity or business in any Designated Jurisdiction or to fund any activity or business of any Person located, organized or residing in any Designated Jurisdiction or who is the subject of any Sanctions, or in any other manner that will result in any violation by any Person (including the Administrative Agent, the Lenders and their Affiliates) of Sanctions.
7.22 Anti-Corruption. No Obligor nor any of its Subsidiaries, nor, to the knowledge of Holdings, the Borrower, any of their respective directors, officers or employees nor, to the
knowledge of Holdings, the Borrower, any agents or other Persons acting on behalf of any of the foregoing, directly or indirectly, has (i) violated or is in violation of any applicable anti-corruption Law, (ii) made, offered to make, promised to make or authorized the payment or giving of, directly or indirectly, any Prohibited Payment or (iii) been subject to any investigation by any Governmental Authority with regard to any actual or alleged Prohibited Payment.
7.23 Deposit and Disbursement Accounts. Schedule 7.23 contains a list of all banks and other financial institutions at which each Obligor or any of its Subsidiaries maintains Deposit Accounts, Securities Accounts, Commodity Accounts, lockboxes, or other similar accounts, and such Schedule correctly identifies the name and address of each bank or financial institution, the name in which the account is held, the type of account, and the complete account number therefor.
7.24 Royalties and Other Payments. Except as set forth on Schedule 7.24, no Obligor nor any of its Subsidiaries is obligated to pay any royalty, milestone payment, deferred payment or any other contingent payment in respect of any Product in an amount in excess of $1,500,000 in the aggregate.
7.25 Non-Competes. No Obligor nor any of its Subsidiaries nor any of their respective directors, officers or employees is subject to a non-compete agreement that prohibits or will interfere with any of the Product Commercialization and Development Activities, including the development, commercialization or marketing of any Product, except for non-compete covenants that protect an exclusive license or otherwise as set forth on Schedule 7.25.
7.26 Internal Controls. Holdings and the Borrower each acknowledge that its management is responsible for the preparation and fair presentation of the financial statements of Holdings, the Borrower and each of its Subsidiaries provided to the Administrative Agent and the Lenders pursuant to Sections 8.01(a), 8.01(b) and 8.01(c), in each case, in accordance with GAAP. Holdings and the Borrower have, suitable for a company of their size and stage of development, designed, implemented and maintained internal controls relevant to the preparation and fair presentation of financial statements that are free from material misstatement, whether due to fraud or error.
SECTION 8
AFFIRMATIVE COVENANTS
The Obligors jointly and severally covenant and agree with the Administrative Agent and the Lenders that, until the Commitments have expired or been terminated and all Obligations (other than Warrant Obligations and inchoate indemnification and expense reimbursement obligations for which no Claim has been made) have been indefeasibly paid in full in cash:
8.01 Financial Statements and Other Information. Holdings shall furnish to the Administrative Agent (with sufficient copies for each Lender):
(a) Any monthly or other periodic financial statements (other than those described in clauses (b) and (c) below) if and to the extent Holdings provides the same to its Board.
(b) As soon as available and in any event within forty-five (45) days after the end of each fiscal quarter of each fiscal year, (i) an unaudited consolidated balance sheet of Holdings and
its Subsidiaries as of the end of such fiscal quarter, and (ii) the related unaudited consolidated statements of income and cash flows of Holdings and its Subsidiaries for each such quarter and the portion of the fiscal year through the end of such fiscal quarter, in each case, prepared in accordance with GAAP consistently applied, all in reasonable detail and setting forth in comparative form the figures for the corresponding period in the preceding fiscal year, together with (iii) a certificate of a Responsible Officer of Holdings stating that such financial statements (x) fairly present in all material respects the financial condition of Holdings and its Subsidiaries as at such date and the results of operations of Holdings and its Subsidiaries for the period ended on such date, and (y) have been prepared in accordance with GAAP consistently applied, subject to changes resulting from normal, year-end audit adjustments and the addition of notes.
(c) As soon as available and in any event within one hundred and twenty (120) days after the end of each fiscal year (i) the audited consolidated balance sheet of Holdings and its Subsidiaries as of the end of such fiscal year, and (ii) the related audited consolidated statements of income, shareholders’ equity and cash flows of Holdings and its Subsidiaries for such fiscal year, in each case prepared in accordance with GAAP consistently applied, all in reasonable detail and setting forth in comparative form the figures for the previous fiscal year, accompanied by a report and opinion thereon of Deloitte & Touche Ross LLP or another firm of independent certified public accountants of recognized national standing reasonably acceptable to the Administrative Agent, which report and opinion shall be prepared in accordance with generally accepted auditing standards and shall not be subject to any “going concern” or like qualification or exception (other than (i) with respect to the Maturity Date and (ii) in connection with the audited financial statements of Holdings and its Subsidiaries for the fiscal year 2021) or any qualification or exception as to the scope of such audit, and in the case of such consolidated financial statements, certified by a Responsible Officer of Holdings.
(d) (i) At the time of delivery of financial statements delivered pursuant to Sections 8.01(b) and 8.01(c), a compliance certificate delivered by the chief financial or accounting Responsible Officer of Holdings as of the end of each such calendar month referenced in clause (x) above or the applicable accounting period referenced in clause (y) above, as the case may be, substantially in the form of Exhibit E including, with respect to the financial statement delivered pursuant to Section 8.01(c), details of any issues that are material that are raised by Holdings’ auditors, and setting forth reasonably detailed calculations demonstrating compliance with Section 10; (ii) together with the financial statements required pursuant to Sections 8.01(b) and 8.01(c), a management discussion and analysis in the format provided by Holdings to its Board; and (iii) within five (5) Business Days following the end of each calendar month, Holding’s chief financial officer shall provide the Administrative Agent with proof of compliance with Section 10.01, which proof may be in the form of copies of one or more bank statements demonstrating such compliance, accompanied by a certification thereof from the chief financial officer.
(e) As soon as available and in any event no later than ninety (90) days following the end of each fiscal year of Holdings, copies of an annual budget (or equivalent) for Holdings and its Subsidiaries, approved by Holdings Board, for the then current fiscal year, in form reasonably satisfactory to the Administrative Agent, accompanied by a certificate of the chief financial officer of Holdings certifying (in his or her capacity as an officer of Holdings and not in his or her individual capacity) that (i) such budget was prepared by Holdings in good faith, (ii) Holdings had at the time of preparation of the budget, and at all times thereafter (including on and as of the date
of delivery to the Administrative Agent of such budget) has continued to have, what Holdings in good faith believes is a reasonable basis for all of the assumptions contained in such budget and (iii) such budget was prepared in accordance with, and based upon, such assumptions.
(f) Copies of all letters of representatives signed by an Obligor to its auditors and, promptly upon receipt thereof, copies of all auditor reports delivered for each fiscal quarter.
(g) Promptly after the same are released, copies of all press releases.
(h) Promptly, and in any event within five (5) Business Days after receipt thereof by Holdings or any of its Subsidiaries, copies of each notice or other correspondence received from any securities regulator or exchange to the authority of which Holdings may become subject from time to time concerning any investigation or possible investigation or other similar inquiry by such agency regarding financial or other operational results of Holdings or any such Subsidiary.
(i) Promptly after the same are available, copies of each annual report, proxy or financial statement or other report or communication sent to all the stockholders of Holdings or any of its Subsidiaries, and copies of all annual, regular, periodic and special reports and registration statements which Holdings or any of its Subsidiaries may file or be required to file with any securities regulator or exchange to the authority of which Holdings or any such Subsidiary, as applicable, may become subject from time to time.
(j) The information regarding insurance maintained by Holdings and its Subsidiaries as required under Section 8.05.
(k) As soon as possible and in any event within five (5) Business Days after an Obligor obtains knowledge of any Claim related to any Product or inventory involving more than $1,500,000, written notice thereof from a Responsible Officer of such Obligor which notice shall include a statement setting forth details of such Claim.
(l) Within five (5) days of delivery, copies of all statements, reports and notices (including board kits) made available to Holding’s Board; provided that any such material may be redacted by Holdings to exclude information relating to the Lenders (including Holdings’ strategy regarding the Loans).
(m) Such other information respecting the operations, properties, business, liabilities or condition (financial or otherwise) of Holdings and its Subsidiaries (including with respect to the Collateral) as the Administrative Agent or any Lender may from time to time reasonably request.
Information required to be delivered pursuant to Sections 8.01 (b), (c) or (i) shall be deemed to have been delivered if such information, or one or more annual or quarterly reports containing such information, shall be publicly available on “EDGAR” (provided, in each case, that Holdings has notified the Administrative Agent (including by e-mail) that such information is available in such a manner and, if requested by the Administrative Agent, shall have provided hard copies to the Administrative Agent).
8.02 Notices of Material Events. On or within five (5) Business Days (or such longer or shorter period as may be expressly set forth below) after a Responsible Officer of Holdings first learns of
or acquires knowledge with respect to any of the below events or circumstances, Holdings shall furnish to the Administrative Agent written notice thereof (prepared in reasonable detail).
(a) The occurrence of any Default.
(b) The occurrence of any event with respect to any property or assets of Holdings or any of its Subsidiaries resulting in a Loss aggregating $1,500,000 (or the Equivalent Amount in other currencies) or more.
(c) Any Claim, action, suit, notice of violation, hearing, investigation or other proceedings pending, or to Holdings’ or the Borrower’s knowledge, threatened in writing against or affecting Holdings or the Borrower or any of its Subsidiaries or with respect to the ownership, use, maintenance and operation of their respective businesses, operations or properties, whether made by a Governmental Authority or other Person that could reasonably be expected to result in a Loss of $1,500,000 or more.
(d) (i) On or prior to the date of any filing by any ERISA Affiliate of any notice of intent to terminate any Title IV Plan, a copy of such notice and (ii) promptly, and in any event within ten (10) days, after any Responsible Officer of any ERISA Affiliate knows or has reason to know (A) that an ERISA Event has occurred or is reasonably expected to occur or (B) that a request for a minimum funding waiver under Section 412 of the Code has been filed with respect to any Title IV Plan or Multiemployer Plan, a notice (which may be made by telephone if promptly confirmed in writing) describing such waiver request and any action that any ERISA Affiliate proposes to take with respect to either of the foregoing, together with a copy of any notice filed with the PBGC or the IRS pertaining thereto.
(e) (i) The termination of any Material Agreement other than in accordance with its terms, including as a result of a breach or default; (ii) the receipt by Holdings or any of its Subsidiaries of any material notice under any Material Agreement (and a copy thereof); (iii) the entering into of any new Material Agreement by Holdings or any of its Subsidiaries (and a copy thereof); or (iv) any material amendment to a Material Agreement (and a copy thereof).
(f) The reports and notices as required by the Security Documents.
(g) Within thirty (30) days of the date thereof, or, if earlier, on the date of delivery of any financial statements pursuant to Section 8.01, notice of any material change in accounting policies or financial reporting practices by the Obligors.
(h) Notice of any labor controversy resulting in or threatening to result in any strike, work stoppage, boycott, shutdown or other material labor disruption against or involving Holdings or any of its Subsidiaries.
(i) Any licensing agreements or similar arrangement entered into by Holdings or any of its Subsidiaries in connection with any infringement or alleged infringement of any Intellectual Property of another Person.
(j) Concurrently with the delivery of financial statements under Section 8.01(b) or (c), notice of the creation, development or other acquisition of any Intellectual Property by Holdings
or any of its Subsidiaries after the Closing Date and during such prior fiscal quarter for which such financial statements were delivered, which is registered or becomes registered or the subject of an application for registration with the U.S. Copyright Office or the U.S. Patent and Trademark Office, as applicable, or with any other equivalent foreign Governmental Authority.
(k) Any change to Holding’s or any of its Subsidiary Guarantors’ ownership of Deposit Accounts, Securities Accounts and Commodity Accounts, by delivering to the Administrative Agent, a notice setting forth a complete and correct list of all such accounts as of the date of such change.
(l) The acquisition by Holdings or any of its Subsidiaries, in a single or series or related transactions, of any fee interest in any real property having a fair market value in excess of $1,000,000.
(m) Any other development that results in, or could reasonably be expected to result in, a Material Adverse Effect or a Material Regulatory Event.
Each notice delivered under this Section 8.02 shall be accompanied by a statement of a Responsible Officer of Holdings setting forth the details of the event or development requiring such notice and any action taken or proposed to be taken with respect thereto. Nothing in this Section 8.02 is intended to waive, consent to or otherwise permit any action or omission that is otherwise prohibited by this Agreement or any other Loan Document.
Notwithstanding the foregoing, so long as Holdings or the Borrower is subject to the reporting requirements of Section 13 or Section 15 of the Exchange Act, Holdings covenants and agrees that neither it, nor any other Person acting on its behalf, will provide, or be obligated to provide, the Administrative Agent or any Lender or their respective representatives or agents with any information that Holdings reasonably believes constitutes material non-public information, unless prior thereto such receiving Person shall have confirmed to Holdings in writing that it consents to receive such information. Holdings acknowledges and confirms that each Secured Party shall be relying on the foregoing covenant in effecting transactions in securities of Holdings and the Borrower
8.03 Existence; Conduct of Business. Each Obligor shall, and shall cause each of its Subsidiaries to, do or cause to be done all things necessary to preserve, renew and maintain in full force and effect its legal existence and all Governmental Approvals necessary or material to the conduct of its business; provided that the foregoing shall not prohibit any merger, amalgamation, consolidation, liquidation or dissolution permitted under Section 9.03.
8.04 Payment of Obligations. Each Obligor shall, and shall cause each of its Subsidiaries to, pay and discharge its obligations, including (i) all Taxes, fees, assessments and governmental charges or levies imposed upon it or upon its properties or assets prior to the date on which penalties attach thereto, and all lawful Claims for labor, materials and supplies which, if unpaid, might become a Lien upon any properties or assets such Obligor or any of its Subsidiaries, except to the extent such Taxes, fees, assessments or governmental charges or levies, or such claims are being contested in good faith by appropriate proceedings and are adequately reserved against in accordance with
GAAP, and (ii) all other lawful Claims which, if unpaid, would by Law become a Lien upon any properties or assets of such Obligor or any of its Subsidiaries, other than any Permitted Lien.
8.05 Insurance. Each Obligor shall, and shall cause each of its Subsidiaries to maintain, with financially sound and reputable insurance companies, insurance in such amounts and against such risks as are customarily maintained by companies engaged in the same or similar businesses operating in the same or similar locations. Upon the reasonable request of the Administrative Agent, such Obligor shall furnish to the Administrative Agent from time to time: (i) information as to the insurance carried by such Obligor and each of its Subsidiaries and, if so requested, copies of all such insurance policies and (ii) a certificate from such Obligor’s insurance broker or other insurance specialist stating that all premiums then due on the policies relating to insurance on the Collateral have been paid and that such policies are in full force and effect. The Obligors shall use commercially reasonable efforts to ensure, or cause others to ensure, that all insurance policies required under this Section 8.05 shall provide that they shall not be terminated or cancelled nor shall any such policy be materially changed in a manner adverse to the insured Person without at least thirty (30) days’ (ten (10) days for nonpayment) prior written notice to such Obligor and the Administrative Agent. Receipt of notice of cancellation or modification of any such insurance policies or reduction of coverage or amounts thereunder shall entitle any Secured Party to renew any such policies, cause the coverage and amounts thereof to be maintained at levels required pursuant to the first sentence of this Section 8.05 or otherwise to obtain similar insurance in place of such policies, in each case at the expense of such Obligor (payable on demand). The amount of any such expenses shall accrue interest at the Default Rate if not paid on demand and shall constitute “Obligations.”
8.06 Books and Records; Inspection Rights. Each Obligor shall, and shall cause each of its Subsidiaries to, keep proper books of record and account in which full, true and correct entries are made of all dealings and transactions in relation to its business and activities. Each Obligor shall, and shall cause each of its Subsidiaries to, permit any representatives designated by the Administrative Agent or any Lender, upon reasonable prior notice, to visit and reasonably inspect its properties, to reasonably examine and make extracts from its books and records, and to discuss its affairs, finances and condition (financial or otherwise) with its officers and independent accountants, all at such reasonable times (but not more often than once per year unless an Event of Default has occurred and is continuing) as the Administrative Agent or the Lenders may reasonably request. Each Obligor shall pay all reasonable costs and expenses of all such inspections.
8.07 Compliance with Laws and Other Obligations. Each Obligor shall, and shall cause each of its Subsidiaries to, (i) comply in all material respects with all applicable Laws and Governmental Approvals (including Environmental Laws and all Healthcare Laws) and (ii) maintain in full force and effect, remain in compliance with, and perform all terms of outstanding Material Indebtedness and all Material Agreements.
8.08 Maintenance of Properties, Etc. Each Obligor shall, and shall cause each of its Subsidiaries to, maintain and preserve all of its assets and properties, whether tangible or intangible, relating to its Products or Product Commercialization and Development Activities or otherwise, necessary or useful in the proper conduct of its business in good working order and condition in accordance
with the general practice of other Persons of similar character and size, ordinary wear and tear and damage from casualty or condemnation excepted.
8.09 Licenses. Each Obligor shall, and shall cause each of its Subsidiaries to, obtain and maintain all Governmental Approvals necessary in connection with the execution, delivery and performance of the Loan Documents, the consummation of the Transactions or the operation and conduct of its business and ownership of its properties (including its Product Commercialization and Development Activities).
8.10 Action under Environmental Laws. Each Obligor shall, and shall cause each of its Subsidiaries to, upon becoming aware of the release of any Hazardous Materials or the existence of any environmental liability under applicable Environmental Laws with respect to their respective businesses, operations or properties, take all actions, at their cost and expense, as shall be necessary or advisable to investigate and clean up the condition of their respective businesses, operations or properties, including all required removal, containment and remedial actions, to restore their respective businesses, operations and properties to a condition in compliance with applicable Environmental Laws.
8.11 Use of Proceeds. The proceeds of the Loans shall be used only as provided in Section 2.04. Without limiting the foregoing, no part of the proceeds of the Loans shall be used, whether directly or indirectly, for any purpose that entails a violation of any of the Regulations of the Board of Governors of the Federal Reserve System, including Regulation T, Regulation U and Regulation X.
8.12 Certain Obligations Respecting Subsidiaries; Further Assurances. Subsidiary Guarantors. Holdings and the Borrower shall take such action from time to time as shall be necessary to ensure that (x) each of its Subsidiaries that is a party to this Agreement as of the Closing Date will be and will remain an Obligor and Subsidiary Guarantor hereunder (except as otherwise permitted by Section 9.03), and (y) each of its other Subsidiaries, whether direct or indirect, now existing or hereafter created (other than any Excluded Subsidiary), will, within ten (10) Business Days of becoming a Subsidiary, become a “Subsidiary Guarantor” pursuant to this Section 8.12. Without limiting the generality of the foregoing, if Holdings or any of its Subsidiaries form or acquire any new Subsidiary that is not an Excluded Subsidiary, then Holdings concurrently shall (unless otherwise agreed by the Administrative Agent in its sole discretion), within ten (10) Business Days of such event:
(i) cause such Subsidiary to become an “Obligor” and a “Subsidiary Guarantor” hereunder, a “Grantor” (or the equivalent thereof) under the applicable Security Document, and a “Subsidiary Party” under the Intercompany Subordination Agreement;
(ii) take such action or cause such Subsidiary to take such action (including joining the Security Agreement or the applicable Security Documents and delivering certificated Equity Interests together with undated transfer powers executed in blank, applicable control agreements, and other instruments) as shall be deemed reasonably necessary or desirable by the Administrative Agent to create and perfect, in favor of the Administrative Agent, for the benefit of the Secured Parties, valid and enforceable first priority Liens on substantially all of the personal property of such new Subsidiary as collateral security for the Obligations hereunder;
(iii) cause the parent of such Subsidiary to execute and deliver a pledge agreement in favor of the Administrative Agent, for the benefit of the Secured Parties, in respect of all outstanding issued Equity Interests of such Subsidiary; and
(iv) deliver such proof of corporate action, incumbency of officers, opinions of counsel and other documents as is consistent with those delivered by each Obligor pursuant to Section 6.01 or as the Administrative Agent shall have reasonably requested.
(b) Further Assurances.
(i) Each Obligor shall, and shall cause each of its direct or indirect Subsidiaries (including any newly formed or newly acquired Subsidiaries, but excluding Excluded Subsidiaries) to take such action from time to time as shall reasonably be requested by the Administrative Agent to effectuate the purposes and objectives of this Agreement and the applicable Security Documents.
(ii) In the event that Holdings or any of its Subsidiaries holds or acquires Obligor Intellectual Property during the term of this Agreement, then, upon the request of the Administrative Agent, Holdings or any such Subsidiary shall take any action as shall be reasonably necessary and reasonably requested by the Administrative Agent to ensure that the provisions of this Agreement and the Security Agreement shall apply thereto and any such Obligor Intellectual Property shall constitute part of the Collateral under the Security Documents.
(iii) Without limiting the generality of the foregoing, within ten (10) Business Days following written request from the Administrative Agent, Holdings shall cause each Person that is required to be a Subsidiary Guarantor or an Obligor hereunder to take such action from time to time (including executing and delivering such assignments, security agreements, control agreements and other instruments, and delivering certificated Equity Interests together with undated transfer powers executed in blank) as shall be reasonably requested by the Administrative Agent to create, in favor of the Secured Parties, perfected security interests and Liens in substantially all of the property of such Person as collateral security for the Obligations; provided that any such security interest or Lien shall be subject to the relevant requirements of the applicable Security Documents.
(iv) In the event that Holdings or any of its Subsidiaries acquires any real property with a value in excess of $1,000,000 during the term of this Agreement, Holdings shall promptly notify the Administrative Agent and provide the Administrative Agent with a description of such real property, the acquisition date thereof and the purchase price therefor. Upon the request of the Administrative Agent, Holdings or any such Subsidiary shall execute and deliver a Mortgage with respect to such acquired real property to secure the Obligations.
(c) Costs and Benefits. Notwithstanding any term or provision of this Section 8.12 to the contrary, without limiting the right of the Administrative Agent or the Lenders to require a Lien or a security interest in the Equity Interests of, or guaranty from, any newly acquired or created Subsidiary of Holdings (or any Subsidiary of Holdings that ceases to be an Immaterial Subsidiary), or a Lien or security interest on any assets or properties of Holdings or any of its Subsidiaries, so long as no Event of Default has occurred and is continuing, Holdings may request in writing to the Lenders that the Lenders waive the requirements of this Section 8.12 to provide a Lien, security interest or guaranty, as the case may be, due to the cost or burden thereof to Holdings and its Subsidiaries (when taken as a whole) being unreasonably excessive relative to the benefit that would inure to the Secured Parties, and describing such cost or burden in reasonable detail. Upon receipt of any such written notice, the Lenders shall review and consider such request
in good faith and, within five (5) Business Days of receipt of such request, shall determine in their sole but commercially reasonable discretion, and notify Holdings of such determination, whether the Lenders will grant such request for a waiver.
8.13 Termination of Non-Permitted Liens. In the event that an Obligor or any of its Subsidiaries shall obtain knowledge of, or be notified by the Administrative Agent or any Lender of the existence of, any outstanding Lien against any assets or property of such Obligor or any of its Subsidiaries, which Lien is not a Permitted Lien, such Obligor shall use its best commercially reasonable efforts to promptly terminate or cause the termination of such Lien.
8.14 Intellectual Property. In the event that an Obligor or any of its Subsidiaries creates, develops or acquires Obligor Intellectual Property during the term of this Agreement, then the provisions of this Agreement shall automatically apply thereto and any such Obligor Intellectual Property shall automatically constitute part of the Collateral under the Security Documents, without further action by any party, in each case from and after the date of such creation, development, or acquisition (except that any representations or warranties of any Obligor shall apply to any such Obligor Intellectual Property only from and after the date, if any, subsequent to such acquisition that such representations and warranties are brought down or made anew as provided herein).
8.15 Maintenance of Regulatory Approvals, Contracts, Intellectual Property, Etc. Each Obligor shall, and shall cause each of its Subsidiaries (to the extent applicable) to, (i) maintain in full force and effect all material Regulatory Approvals, Material Agreements, Material Intellectual Property and other rights, interests or assets (whether tangible or intangible) reasonably necessary for the operations of such Person’s business, including any Product Commercialization and Development Activities, (ii) maintain in full force and effect, and pay all costs and expenses relating to, such Regulatory Approvals, Material Agreements and Material Intellectual Property owned, used or controlled by such Obligor or any such Subsidiary that are used in or necessary for any related Product Commercialization and Development Activities, (iii) promptly after obtaining knowledge thereof, notify the Administrative Agent of any material infringement or violation by any Person of such Obligor’s or any such Subsidiaries’ Material Intellectual Property, and take commercially reasonable efforts to pursue any such infringement or other violation, (iv) use commercially reasonable efforts to pursue and maintain in full force and effect legal protection for all new Material Intellectual Property created, developed or acquired by such Obligor or any of its Subsidiaries, as the case may be, that is used in or necessary for the operations of the business of such Person, or in connection with any Product Commercialization and Development Activities relating to any Product, and (v) promptly after obtaining knowledge thereof, notify the Administrative Agent of any Claim by any Person that the conduct of the business of such Obligor or any of its Subsidiaries, including in connection with any Product Commercialization and Development Activities, has infringed upon any Intellectual Property of such Person.
8.16 ERISA and Foreign Pension Plan Compliance. Such Obligor shall comply, and shall cause each of its Subsidiaries to comply, in all material respects with the provisions of ERISA or applicable Law with respect to any Benefit Plans or Foreign Pension Plans to which such Obligor or any such Subsidiary is a party as an employer.
8.17 Cash Management. Each Obligor shall, and shall cause each of its Subsidiary to:
(a) maintain at all times all Deposit Accounts, Securities Accounts, Commodity Accounts, lockboxes and similar accounts (other than Excluded Accounts) held by each Obligor with a bank or financial institution that has executed and delivered to and in favor of the Administrative Agent an account control agreement, in form and substance reasonably acceptable to the Administrative Agent (each such Deposit Account, Securities Account, Commodity Account, lockbox or similar account, a “Controlled Account”);
(b) maintain each such Controlled Account as a cash collateral account, with all cash, checks and other similar items of payment in such account securing payment of the Obligations, and each Obligor shall have granted a Lien to the Administrative Agent, for the benefit of the Secured Parties, over such Controlled Accounts;
(c) deposit promptly, and in any event no later than five (5) Business Days after the date of receipt thereof, all cash, checks, drafts or other similar items of payment relating to or constituting payments made in respect of any and all accounts receivable, Contracts or any other rights and interests into one or more Controlled Accounts; and
(d) at any time after the occurrence and during the continuance of an Event of Default, at the request of the Administrative Agent, direct all payments constituting proceeds of accounts receivable to be directed into lockbox accounts pursuant to agreements in form and substance reasonably satisfactory to the Administrative Agent.
8.18 Litigation Cooperation. To the extent consistent with the preservation of attorney-client privilege, each Obligor shall, and shall cause each of its Subsidiaries to make reasonably available to the Administrative Agent, its (and its Subsidiaries’) officers, employees, agents, without expense to the Administrative Agent, books and records, to the extent that the Administrative Agent may deem them reasonably necessary to prosecute or defend against any third-party suit or proceeding instituted by or against the Administrative Agent or any Secured Party with respect to any Collateral, the subject of any Loan Document or relating to such Obligor or any of its Subsidiaries.
8.19 Board Observation Rights.
(a) Holdings shall permit one individual representing the Lenders and appointed by the Administrative Agent (the “Observer”) to attend and observe (but not vote) at all meetings of Holdings’ Board, whether in person, by telephone or otherwise as reasonably requested by the Observer. Holdings shall notify the Observer in writing at least five (5) Business Days in advance (or, if a shorter notice period is reasonably necessary given the circumstances, as soon as possible and in all circumstances at least twenty-four (24) hours in advance) of (i) the date and time for each general or special meeting of Holdings’ Board and (ii) the adoption of any resolutions or actions by Holdings’ Board or any committee thereof by written consent (describing, in reasonable detail, the nature and substance of such action). Concurrently with notice to the member of Holdings’ Board of any such meeting, Holdings shall deliver or cause to be delivered to the Observer all notices and any material delivered to any members of such Board in connection with the applicable meeting or action to be taken by written consent, including a draft of any material resolutions or actions proposed to be adopted by written consent. Prior to such meeting or adoption by written consent, the Observer shall be free to contact one or more members of Holdings’ Board
or committee and discuss the pending actions to be taken. Holdings’ Board may meet in executive session without the Observer present to the extent such Board or committee determines in good faith that the issue to be discussed at such session is not appropriate to be discussed with the Observer because (i) such issue directly involves the Loan Documents and discussion thereof would result in a conflict of interest with the Lenders with respect thereto or (ii) the discussion of such issue in the presence of the Observer would result in the loss of attorney-client privilege. The Observer may only be excluded from meetings or executive sessions to the extent the matter being discussed is of a type described in clauses (i) or (ii) above.
(b) Holdings reimburse the Observer for its reasonable out-of-pocket expenses (including the cost of travel, meals and lodging) in connection with the Observer’s attendance of such meetings in accordance with Holdings’ policies for reimbursement of directors.
SECTION 9
NEGATIVE COVENANTS
The Obligors jointly and severally covenant and agree with the Administrative Agent and the Lenders that, until the Commitments have expired or been terminated and all Obligations (other than Warrant Obligations and inchoate indemnification and expense reimbursement obligations for which no Claim has been made) have been indefeasibly paid in full in cash:
9.01 Indebtedness. The Obligors shall not, and shall not permit any of its Subsidiaries to, create, incur, assume or permit to exist any Indebtedness, whether directly or indirectly, except for the following:
(a) the Obligations;
(b) Indebtedness existing on the Closing Date and set forth on Schedule 7.13(a) and Permitted Refinancings thereof;
(c) Indebtedness of an Obligor owing to another Obligor; provided that, in each case, such Indebtedness shall be subordinated to the Obligations pursuant to the Intercompany Subordination Agreement;
(d) Guarantees by an Obligor of the Indebtedness of another Obligor to the extent such Indebtedness is otherwise permitted hereunder; provided that any subrogation claims of any such guarantying Obligor shall be subordinated to the Obligations pursuant to the Intercompany Subordination Agreement;
(e) ordinary course of business equipment financing and leasing; provided that (i) if secured, the collateral therefor consists solely of the assets being financed, the products and proceeds thereof and books and records related thereto, and (ii) the aggregate outstanding principal amount of such Indebtedness shall not exceed $1,500,000 (or the Equivalent Amount in other currencies) at any time;
(f) Indebtedness under Hedging Agreements permitted by Section 9.05(e);
(g) (i) to the extent constituting or qualifying as Indebtedness, all obligations of the Borrower to pay Earnout Payments (as defined in, and to the extent payable pursuant to, Exhibit D to the Waypoint Product Acquisition Agreement as in effect on the Amendment No. 2 Effective Date), and (ii) any additional Indebtedness assumed pursuant to any Permitted Acquisition; provided that, and without regard to any Indebtedness of the type described in subclause (i) of this Section 9.01(g), (x) the aggregate amount of such additional Indebtedness permitted pursuant to this Section 9.01(g)(ii) shall not exceed $1,500,000 at any time outstanding and (y) no such additional Indebtedness shall have been created or incurred in connection with, or in contemplation of, such Permitted Acquisition;
(h) Indebtedness in respect of any agreement providing for treasury, depositary or cash management services, including in connection with any automated clearing house transfers of funds or any similar transfers, netting services, overdraft protections and other cash management and similar arrangements, in each case in the ordinary course of business;
(i) advances or deposits from customers or vendors received in the ordinary course of business;
(j) workers’ compensation claims, payment obligations in connection with health, disability or other types of social security benefits, unemployment or other insurance obligations and reclamation and statutory obligations, in each case incurred in the ordinary course of business;
(k) Indebtedness not to exceed $1,500,000 at any time outstanding in respect of advances received under licensing and collaboration agreements;
(l) Indebtedness consisting of the deferred obligations to pay insurance premiums in respect of insurance policies pursuant to Section 8.05 insuring assets or businesses of an Obligor written or arranged in such Obligor’s ordinary course of business and which are payable within one (1) year; and
(m) Indebtedness incurred in connection with letters of credit that are secured solely by cash or cash equivalents and issued on behalf of the Borrower in the ordinary course of business in an aggregate amount outstanding not to exceed $1,000,000 at any time;
(n) Indebtedness of non-U.S. Subsidiaries of Holdings borrowed from Persons that are not Obligors, which Indebtedness is incurred for the purpose of financing working capital needs of such Subsidiaries (including leases of real and personal property used in the ordinary course), not to exceed (i) $750,000 in aggregate principal amount for all such Subsidiaries that are Obligors at any time outstanding and (iii) $1,000,000 in aggregate principal amount for all such Subsidiaries that are not Obligors at any time outstanding;
(o) unsecured Indebtedness of Obligors resulting from Guarantees of Indebtedness incurred by non-U.S. Subsidiaries pursuant to Section 9.01(n) above, not to exceed $500,000 in aggregate principal amount at any time outstanding;
(p) Indebtedness of non-U.S. Subsidiaries borrowed from Obligors, which Indebtedness, when taken together with any Investments made pursuant to Section 9.05(l)(ii)
below, shall not exceed $500,000 in aggregate principal amount for all such Subsidiaries at any time outstanding; and
(q) unsecured Indebtedness incurred by Obligors in an aggregate amount not to exceed $500,000 at any time outstanding.
9.02 Liens. The Obligors shall not, and shall not permit any of its Subsidiaries to, create, incur, assume or permit to exist any Lien on any property now owned by it or such Subsidiary, or assign or sell any income or revenues (including accounts receivable) or rights in respect of any thereof, except for the following:
(a) Liens securing the Obligations;
(b) any Lien on any property or asset of Holdings or any of its Subsidiaries existing on the Closing Date and set forth on Schedule 7.13(c); provided that (i) no such Lien shall extend to any other property or asset of Holdings or any of its Subsidiaries and (ii) any such Lien shall secure only those obligations which it secures on the Closing Date and extensions, renewals and replacements thereof that do not increase the outstanding principal amount thereof;
(c) Liens securing Indebtedness permitted under Section 9.01(g); provided that such Liens are restricted solely to the collateral permitted to be secured pursuant to Section 9.01(g);
(d) Liens imposed by any applicable Law arising in the ordinary course of business, including (but not limited to) carriers’, warehousemen’s, lessor’s and mechanics’ liens and other similar Liens arising in the ordinary course of business and which (x) do not in the aggregate materially detract from the value of the property subject thereto or materially impair the use thereof in the operations of the business of such Person or (y) are being contested in good faith by appropriate proceedings, which proceedings have the effect of preventing the forfeiture or sale of the property subject to such Liens and for which adequate reserves have been made if required in accordance with GAAP;
(e) pledges or deposits made in the ordinary course of business in connection with workers’ compensation, unemployment insurance or other similar social security legislation;
(f) Liens securing Taxes, assessments and other governmental charges, the payment of which is not yet due or is being contested in good faith by appropriate proceedings promptly initiated and diligently conducted and for which such reserve or other appropriate provisions, if any, as shall be required GAAP shall have been made;
(g) servitudes, easements, rights of way, restrictions and other similar encumbrances on real property imposed by any applicable Law and Liens consisting of zoning or building restrictions, easements, licenses, restrictions on the use of property or minor imperfections in title thereto which, in the aggregate, are not material, and which do not in any case materially detract from the value of the property subject thereto or interfere with the ordinary conduct of the business of any of the Obligors or any of their Subsidiaries;
(h) with respect to any real property, (i) such defects or encroachments as might be revealed by an up-to-date survey of such real property; (ii) the reservations, limitations, provisos
and conditions expressed in the original grant, deed or patent of such property by the original owner of such real property pursuant to applicable Law; (iii) rights of expropriation, access or user or any similar right conferred or reserved by or in any applicable Law, which, in the aggregate for clauses (i), (ii) and (iii), are not material, and which do not in any case materially detract from the value of the property subject thereto or interfere with the ordinary conduct of the business of any of the Obligors or any of their Subsidiaries and (iv) leases or subleases in the ordinary course of business;
(i) Liens securing Indebtedness permitted under Section 9.01(i); provided that (i) such Lien is not created in contemplation of or in connection with such Permitted Acquisition, (ii) such Lien shall not apply to any other property or assets of Holdings or any of its Subsidiaries other than the property or assets being acquired pursuant to such Permitted Acquisition, and (iii) such Lien shall secure only those obligations that it secured immediately prior to the consummation of such Permitted Acquisition and extensions, renewals and replacements thereof that do not increase the outstanding principal amount thereof;
(j) bankers’ liens, rights of setoff and similar Liens incurred on deposits made in the ordinary course of business;
(k) (i) licenses permitted pursuant to Section 9.18 and (ii) any ordinary course interest or title of a licensor, sublicensor, collaborator, lessor or sublessor with respect to any assets under any inbound license, collaboration agreement or lease agreement permitted pursuant to Section 9.18 (including Liens on Products that include Intellectual Property licensed to Holdings or its Subsidiaries to secure their payment obligations with respect to such license or collaboration agreement);
(l) Liens consisting of cash collateral securing Indebtedness under Hedging Agreements permitted by Section 9.01(f) and letters of credit permitted by Section 9.01(m);
(m) with respect to any Indebtedness incurred by a non-U.S. Subsidiary of Holdings pursuant to Section 9.01(n) above, Liens on assets and properties of such Subsidiary securing such Indebtedness; and
(n) other Liens securing other Indebtedness to the extent permitted hereby not to exceed $500,000 in an aggregate principal amount at any time outstanding.
Any term or provision of this Section 9.02 to the contrary notwithstanding, no Lien otherwise permitted under any of the foregoing clauses (b) through (n) shall apply to any Material Intellectual Property.
9.03 Fundamental Changes, Acquisitions, Etc. The Obligors shall not, and shall not permit any of its Subsidiaries to, (i) enter into any transaction of merger, amalgamation or consolidation, (ii) liquidate, wind up or dissolve itself (or suffer any liquidation or dissolution), (iii) sell or issue any Disqualified Equity Interests, or (iv) other than Permitted Acquisitions, make any Acquisition, except for the following (in each case to the extent that no Event of Default has occurred and is continuing, or could reasonably be expected to result therefrom):
(a) the merger, amalgamation or consolidation of any Subsidiary with or into any Obligor; provided that with respect to any such transaction involving Holdings or the Borrower, Holdings or the Borrower must be the surviving or successor entity of such transaction and with respect to any such transaction involving any other Obligor and a non-Obligor Subsidiary, such Obligor must be the surviving or successor entity of such transaction;
(b) the sale, lease, transfer or other disposition by any Subsidiary of any or all of its property (upon voluntary liquidation or otherwise) to any Obligor; and
(c) the sale, transfer or other disposition of the Equity Interests of any Subsidiary to any Obligor.
9.04 Lines of Business. The Obligors shall not, and shall not permit any of its Subsidiaries to, engage in any business other than the business engaged in on the Closing Date by such Persons or a business reasonably related thereto and reasonable extensions thereof, including the business of operating a pharmacy.
9.05 Investments. The Obligors shall not, and shall not permit any of its Subsidiaries to, make, directly or indirectly, or permit to remain outstanding any Investments, except for the following:
(a) Investments outstanding on the Closing Date and identified on Schedule 9.05;
(b) operating Deposit Accounts, Securities Accounts or Commodity Accounts with banks or financial institutions that are Controlled Accounts;
(c) extensions of credit in the nature of accounts receivable or notes receivable arising from the sales of goods or services in the ordinary course of business and prepaid royalties in the ordinary course of business;
(d) Permitted Cash Equivalent Investments in Controlled Accounts or Excluded Accounts;
(e) Hedging Agreements entered into in Holdings’ or any of its Subsidiaries’ ordinary course of business for the purpose of hedging currency risks or interest rate risks (but not for speculative purposes) and in an aggregate net exposure amount for all such Hedging Agreements not in excess of $1,500,000 (or the Equivalent Amount in other currencies);
(f) Investments consisting of security deposits with utilities and landlords to secure office space and other like Persons made in the ordinary course of business;
(g) employee loans, travel advances and guarantees in accordance with Holdings’ usual and customary practices with respect thereto (if permitted by applicable Law) which in the aggregate shall not exceed $1,500,000 outstanding at any time (or the Equivalent Amount in other currencies plus any amounts consisting of any employee’s deferred payment obligation for the purchase of Equity Interests in Holdings);
(h) Investments received in connection with any Insolvency Proceedings in respect of any customers, suppliers or clients and in settlement of delinquent obligations of, and other disputes with, customers, suppliers or clients;
(i) Investments in the form of Indebtedness owing by Obligors or any of their Subsidiaries to other Obligors or their Subsidiaries to the extent such Indebtedness is permitted pursuant to Section 9.01;
(j) Investments not to exceed $5,000,000 at any time outstanding in joint ventures and counterparties to collaboration and license agreements;
(k) Permitted Acquisitions;
(l) Investments by an Obligor in (i) any other Obligor and (ii) any non-U.S. Subsidiary described in Section 9.01(p); provided that the aggregate amount of such Investments made pursuant to this clause (l)(ii), when taken together with any Indebtedness incurred pursuant to Section 9.01(p) above, shall not exceed $500,000 in aggregate amount for all such Subsidiaries at any time outstanding; and
(m) other Investments not to exceed $1,500,000 in the aggregate at any time outstanding.
9.06 Restricted Payments. The Obligors shall not, and shall not permit any of its Subsidiaries to, declare or make, directly or indirectly, any Restricted Payment; provided that, notwithstanding the foregoing, the following Restricted Payments shall be permitted so long as no Event of Default could reasonably be expected to occur or result from the making of such Restricted Payment:
(a) dividends with respect to Holdings’ Equity Interests payable solely in shares of its Qualified Equity Interests;
(b) dividends paid by any Subsidiary to any Obligor;
(c) dividends paid by any foreign Subsidiary to another foreign Subsidiary;
(d) upon the death, incapacity or termination of any natural person that is a holder of Qualified Equity Interests of Holdings or the exercise of a right of first refusal or similar right in respect of any such holder, Holdings may repurchase the stock of such Qualified Equity Interests of such holder or such holder’s family, trusts, estates and heirs pursuant to stock repurchase agreements in an amount not to exceed $500,000 per fiscal year;
(e) the payment by Holdings or any of its Subsidiaries of cash in lieu of the issuance of fractional shares to the extent required by the applicable Organic Document of Holdings or such Subsidiary and in an aggregate amount not to exceed $25,000 per fiscal year for Holdings and all such Subsidiaries;
(f) Holdings may honor any non-cash (other than cash in lieu of fractional shares) conversion or exercise requests in respect of any convertible securities, options or warrants of
Holdings into Qualified Equity Interests of Holdings pursuant to the terms of such convertible securities, options or warrants or otherwise in exchange therefor; and
(g) the repurchase or other acquisition of Qualified Equity Interests of Holdings deemed to occur (i) upon the exercise of stock options, warrants, restricted stock units or other rights to purchase Qualified Equity Interests of Holdings if such Equity Interests represent a portion of the exercise price thereof or conversion price thereof and (ii) in connection with any tax withholding required upon the grant of or any exercise or vesting of any Qualified Equity Interests of Holdings (or options in respect thereof).
9.07 Payments of Indebtedness. The Obligors shall not, and shall not permit any of its Subsidiaries to, make any payments in respect of any Indebtedness other than (i) payments of the Obligations, (ii) scheduled payments and Permitted Refinancings of other Indebtedness to the extent such Indebtedness is permitted pursuant to Section 9.01 and (iii) voluntary prepayments of Indebtedness pursuant to Section 9.01(e) and, to the extent relating to the payment of the deferred purchase price of a Permitted Acquisition, Section 9.01(g).
9.08 Change in Fiscal Year. The Obligors shall not, and shall not permit any of its Subsidiaries to, change the last day of its fiscal year from that in effect on the Closing Date, except to change the fiscal year of a Subsidiary acquired in connection with an Acquisition to conform its fiscal year to that of the Borrower.
9.09 Sales of Assets, Etc. The Obligors shall not, and shall not permit any of its Subsidiaries to sell, lease, transfer, or otherwise dispose of any of its assets or property (including accounts receivable, Intellectual Property or Equity Interests of Subsidiaries), grant or enter into any Exclusive License, forgive, release or compromise any amount owed to such Obligor or such Subsidiary, in each case, in one transaction or series of transactions (any thereof, an “Asset Sale”), except for the following (provided that, in the case of any Asset Sale of the type described in clauses (c), (i) or (j) below, the Obligors shall not, and shall not permit any of its Subsidiaries to, allow any such Asset Sale to occur if any Event of Default has occurred and is continuing or could reasonably be expected to occur as a result of such Asset Sale):
(a) sales of inventory in the ordinary course of its business on ordinary business terms;
(b) the forgiveness, release or compromise of any amount owed to any Obligor or Subsidiary in the ordinary course of business;
(c) transfers of assets or property (other than any Material Intellectual Property) by any Obligor to any other Obligor;
(d) dispositions of any assets or property (including leaseholds, but other than any Material Intellectual Property) that is obsolete or worn out or no longer used or useful in the Business;
(e) in connection with any transaction permitted under Sections 9.03 or 9.05;
(f) the use of cash and Permitted Cash Equivalent Investments in the ordinary course of business or in connection with other business activities not prohibited or otherwise restricted hereby or by any other Loan Document;
(g) dispositions consisting of the sale, transfer, assignment or other disposition of unpaid and overdue accounts receivable in connection with the collection, compromise or settlement thereof;
(h) dispositions of property to the extent that such property is exchanged for credit against the purchase price of similar replacement property;
(i) dispositions in the form of any Exclusive License of Specified Intellectual Property permitted hereunder; and
(j) other dispositions of property not to exceed $1,500,000 in the aggregate in any fiscal year.
9.10 Transactions with Affiliates. The Obligors shall not, and shall not permit any of its Subsidiaries to, sell, lease, license or otherwise transfer any assets to, or purchase, lease, license or otherwise acquire any assets from, or otherwise engage in any other transactions with, any of its Affiliates, except:
(a) transactions between or among Obligors;
(b) customary compensation and indemnification of, and other employment arrangements with, directors, officers and employees of Holdings or any of its Subsidiaries in the ordinary course of business; and
(c) any other transaction that is (i) on fair and reasonable terms that are no less favorable (including with respect to the amount of cash or other consideration receivable or payable in connection therewith) to such Obligor or any such Subsidiary than it could obtain in an arm’s-length transaction with a Person that is not an Affiliate of such Obligor or any such Subsidiary, as applicable, and (ii) of the kind which would be entered into by a prudent Person in the position of such Obligor or any such Subsidiary with another Person that is not an Affiliate of such Obligor or any such Subsidiary, as applicable.
9.11 Restrictive Agreements. The Obligors shall not, and shall not permit any of its Subsidiaries to, directly or indirectly, enter into, incur or permit to exist any Restrictive Agreement other than (i) restrictions and conditions imposed by applicable Laws or by the Loan Documents and (ii) Restrictive Agreements listed on Schedule 7.15.
9.12 Modifications and Terminations of Material Agreements and Organic Documents. The Obligors shall not, and shall not permit any of its Subsidiaries to:
(a) waive, amend, modify, terminate, replace or otherwise modify any term or provision of any Organic Document in any manner materially adverse to the interests of the Administrative Agent or to the Lenders; or
(b) (x) take or omit to take any action that results in the termination of, or permits any other Person to terminate, any Material Agreement or Material Intellectual Property, or (y) waive, amend, terminate, replace or otherwise modify any term or provision of any Material Agreement in any manner materially adverse to the interests of the Secured Parties.
9.13 Sales and Leasebacks. Except as disclosed on Schedule 9.13, the Obligors shall not, and shall not permit any of its Subsidiaries to, become liable, directly or indirectly, with respect to any lease, whether an operating lease or a Capital Lease Obligation, of any property (whether real, personal, or mixed), whether now owned or hereafter acquired, (i) which such Person has sold or transferred or is to sell or transfer to any other Person and (ii) which such Person intends to use for substantially the same purposes as property which has been or is to be sold or transferred.
9.14 Hazardous Material. The Obligors shall not, and shall not permit any of its Subsidiaries to, use, generate, manufacture, install, treat, release, store or dispose of any Hazardous Material, except in compliance with all applicable Environmental Laws or where the failure to comply could not, individually or in the aggregate, reasonably be expected to result in a Material Adverse Effect.
9.15 Accounting Changes. The Obligors shall not, and shall not permit any of its Subsidiaries to, make any significant change in accounting treatment or reporting practices, except as required or permitted by GAAP.
9.16 Compliance with ERISA. No ERISA Affiliate shall cause or suffer to exist (i) any event that could result in the imposition of a Lien with respect to any Title IV Plan or Multiemployer Plan or (ii) any other ERISA Event that, in the aggregate, could reasonably be expected to result in a Material Adverse Effect. No Obligor nor any of its Subsidiaries shall cause or suffer to exist any event that could result in the imposition of a Lien with respect to any Benefit Plan (other than Permitted Liens).
9.17 Sanctions; Anti-Corruption Use of Proceeds. The Obligors shall not, directly or indirectly, use the proceeds of the Loans, or lend, contribute or otherwise make available such proceeds to any Subsidiary, joint venture partner or other Person, (i) in furtherance of an offer, payment, promise to pay, or authorization of the payment or giving of money, or anything else of value, to any Person in violation of any applicable anti-corruption Law, or (ii) (A) to fund any activities or business of or with any Person, or in any country or territory, that, at the time of such funding, is, or whose government is, the subject of Sanctions, or (B) in any other manner that would result in a violation of Sanctions by any Person (including any Person participating in the Loans, whether as Administrative Agent, Lender, underwriter, advisor, investor, or otherwise).
9.18 Inbound and Outbound Licenses.
(a) Inbound Licenses. The Obligors shall not, and shall not permit any of its Subsidiaries to, enter into or become or remain bound by any inbound license agreement requiring any Obligor or any of its Subsidiaries, as the case may be, during any twelve (12) month period during the term of such license agreement, to make aggregate payments in excess of $1,000,000 unless such Obligor has provided prior written notice to the Administrative Agent of the material terms of such license or agreement with a description of its anticipated and projected impact on such Obligor’s or such Subsidiary’s, as applicable, business or financial condition; provided that
inbound license agreements in the nature of over the counter or “shrink wrap” software that are commercially available to the public shall not be prohibited by this clause (a).
(b) Outbound Licenses. The Obligors shall not, and shall not permit any of its Subsidiaries to, enter into or become or remain bound by any outbound license of Intellectual Property unless such outbound license: (i) has been entered into on an arm’s-length basis, on commercially reasonable terms and in the ordinary course of business, (ii) is entered into for the purpose of Product Commercialization and Development Activities with respect to a Product, and (iii) subject to the terms of any customary non-disturbance or similar agreement relating to such license, to the extent such Intellectual Property constitutes Collateral, the terms of such license do not impair the Administrative Agent or the Lenders from fully exercising their rights under any of the Loan Documents in the event of a disposition or liquidation (including in connection with a foreclosure so long as any transfer of ownership is made subject to such license) of the rights, assets or property that is the subject of such license.
9.19 Activities of the Massachusetts Securities Subsidiary. The Massachusetts Securities Subsidiary shall not incur any Indebtedness or Liens, not make any Investments and shall not engage in any business activities and shall not own any property other than (a) activities permitted by, and Investments made in accordance with, Massachusetts General Law, Chapter 63, Section 38B, (b) activities and contractual rights incidental to the maintenance of its corporate existence, and (c) the performance of its obligations in its Organic Documents.
SECTION 10
FINANCIAL COVENANTS
10.01 Minimum Liquidity. The Borrower shall at all times maintain a minimum aggregate balance of five million dollars ($5,000,000) in cash in one or more Controlled Accounts maintained with one or more commercial banks or similar deposit-taking institutions in the U.S. that are free and clear of all Liens, other than Liens granted under the Loan Documents in favor of the Secured Parties.
10.02 Minimum Revenue. As of the last day of each fiscal quarter set forth below (each, a “Calculation Date”), Holdings and its Subsidiaries shall have received Revenue, for the trailing twelve (12) month consecutive period ending on such Calculation Date, in an aggregate amount not less than the corresponding amount set forth opposite such Calculation Date:
| | | | | |
| Calculation Date | Revenue |
| March 31, 2022 | $5,750,000 |
| June 30, 2022 | $7,750,000 |
| September 30, 2022 | $11,000,000 |
| December 31, 2022 | $18,000,000 |
| March 31, 2023 | $26,000,000 |
| | | | | |
| Calculation Date | Revenue |
| June 30, 2023 | $44,000,000 |
| September 30, 2023 | $69,000,000 |
| December 31, 2023 | $100,000,000 |
| March 31, 2024 | $125,000,000 |
| June 30, 2024 | $125,000,000 |
| September 30, 2024 | $125,000,000 |
| December 31, 2024 | $125,000,000 |
| March 31, 2025 | $125,000,000 |
10.03 Required Equity Raise. On or prior to December 31, 2020, the Borrower shall have consummated the issuance and sale of its Series D preferred stock resulting in gross proceeds to the Borrower of at least $50,000,000. Promptly after the consummation of such issuance and sale, the Borrower shall deliver to the Administrative Agent, (x) true and complete copies of all agreements (including amendments to Organic Documents) entered into in connection therewith, (y) evidence that the Borrower has consummated such issuance and sale pursuant to such agreements, and (z) evidence that all necessary corporate actions, including approval by the Borrower’s Board and by all necessary holders of Equity Interests, have been obtained in connection with the foregoing clauses (x) and (y), in each case, in form and substance reasonably satisfactory to the Administrative Agent.
SECTION 11
EVENTS OF DEFAULT
11.01 Events of Default. Each of the following events shall constitute an “Event of Default”:
(a) Principal or Interest Payment Default. The Borrower shall fail to pay any principal of or interest on the Loans, when and as the same shall become due and payable, whether at the due date thereof, at a date fixed for prepayment thereof or otherwise.
(b) Other Payment Defaults. Any Obligor shall fail to pay any Obligation (other than an amount referred to in Section 11.01(a)) when and as the same shall become due and payable, and such failure shall continue unremedied for a period of three (3) Business Days.
(c) Representations and Warranties. Any representation or warranty made in writing or deemed made by or on behalf of Holdings or any of its Subsidiaries in or in connection with this Agreement or any other Loan Document or any amendment or modification hereof or thereof, or in any report, certificate, financial statement or other document furnished pursuant to
or in connection with this Agreement or any other Loan Document or any amendment or modification thereof, shall: (i) prove to have been incorrect when made or deemed made to the extent that such representation or warranty contains any materiality or Material Adverse Effect qualifier; or (ii) prove to have been incorrect in any material respect when made or deemed made to the extent that such representation or warranty does not otherwise contain any materiality or Material Adverse Effect qualifier.
(d) Certain Covenants. Any Obligor shall fail to observe or perform any covenant, condition or agreement contained in Sections 8.02, 8.03 (with respect to the Borrower’s existence), 8.11, 8.12, 8.17, Section 9 or Section 10.
(e) Other Covenants. Any Obligor shall fail to observe or perform any covenant, condition or agreement contained in this Agreement (other than those specified in Section 11.01(a), 11.01(b) or 11.01(d)) or any other Loan Document, and, in the case of any failure that is capable of cure, such failure shall continue unremedied for a period of thirty (30) or more days after actual knowledge by a Responsible Officer or receipt of notice from the Administrative Agent.
(f) Payment Default on Other Indebtedness. Any Obligor or any of its Subsidiaries shall fail to make any payment (whether of principal or interest and regardless of amount) in respect of any Material Indebtedness, when and as the same shall become due and payable after giving effect to any applicable grace or cure period as originally provided by the terms of such Indebtedness.
(g) Other Defaults on Other Indebtedness. (i) Any material breach of, or “event of default” or similar event under, any Contract governing any Material Indebtedness shall occur, or (ii) any event or condition occurs (x) that results in any Material Indebtedness becoming due prior to its scheduled maturity or (y) that enables or permits (with or without the giving of notice, the lapse of time or both) the holder or holders of such Material Indebtedness or any trustee or agent on its or their behalf to cause such Material Indebtedness to become due, or to require the prepayment, repurchase, redemption or defeasance thereof, prior to its scheduled maturity; provided that this Section 11.01(g) shall not apply to secured Indebtedness that becomes due as a result of the voluntary sale or transfer of the property or assets securing such Material Indebtedness.
(h) Insolvency, Bankruptcy, Etc.
(i) Holdings or any of its Subsidiaries (other than Immaterial Subsidiaries) becomes insolvent, or generally does not or becomes unable to pay its debts or meet its liabilities as the same become due, or admits in writing its inability to pay its debts generally, or declares any general moratorium on its indebtedness, or proposes a compromise or arrangement or deed of company arrangement between it and any class of its creditors.
(ii) Holdings or any of its Subsidiaries (other than Immaterial Subsidiaries) commits an act of bankruptcy or makes an assignment of its property for the general benefit of its creditors or makes a proposal (or files a notice of its intention to do so).
(iii) Holdings or any of its Subsidiaries (other than Immaterial Subsidiaries) institutes any proceeding seeking to adjudicate it an insolvent, or seeking liquidation, dissolution, winding-up, reorganization, compromise, arrangement, adjustment, protection, moratorium, relief, stay of proceedings of creditors generally (or any class of creditors), or composition of it or its debts or any other relief, under any applicable Law, whether U.S. or non-U.S., now or hereafter in effect relating to bankruptcy, winding-up, insolvency, reorganization, receivership, plans of arrangement or relief or protection of debtors or at common law or in equity, or files an answer admitting the material allegations of a petition filed against it in any such proceeding.
(iv) Holdings or any of its Subsidiaries (other than Immaterial Subsidiaries) applies for the appointment of, or the taking of possession by, a receiver, interim receiver, receiver/manager, sequestrator, conservator, custodian, administrator, trustee, liquidator, voluntary administrator, receiver and manager or other similar official for it or any substantial part of its property.
(v) Holdings or any of its Subsidiaries (other than Immaterial Subsidiaries) takes any action, corporate or otherwise, to approve, effect, consent to or authorize any of the actions described in this Section 11.01(h), or otherwise acts in furtherance thereof or fails to act in a timely and appropriate manner in defense thereof.
(vi) Any petition is filed, application made or other proceeding instituted in a court of competent jurisdiction against or in respect of Holdings or any of its Subsidiaries (other than Immaterial Subsidiaries):
(A) seeking to adjudicate it as insolvent;
(B) seeking a receiving order against it;
(C) seeking liquidation, dissolution, winding-up, reorganization, compromise, arrangement, adjustment, protection, moratorium, relief, stay of proceedings of creditors generally (or any class of creditors), deed of company arrangement or composition of it or its debts or any other relief under any applicable Law, whether U.S. or non-U.S., now or hereafter in effect relating to bankruptcy, winding-up, insolvency, reorganization, receivership, plans of arrangement or relief or protection of debtors or at common law or in equity; or
(D) seeking the entry of an order for relief or the appointment of, or the taking of possession by, a receiver, interim receiver, receiver/manager, sequestrator, conservator, custodian, administrator, trustee, liquidator, voluntary administrator, receiver and manager or other similar official for it or any substantial part of its property,
and such petition, application or proceeding continues undismissed, or unstayed and in effect, for a period of sixty (60) days after the institution thereof; provided that if an order, decree or judgment is granted or entered (whether or not entered or subject to appeal) against Holdings or any of its Subsidiaries (other than Immaterial Subsidiaries) thereunder in the interim, such grace period will cease to apply; provided, further, that if Holdings or such Subsidiary files an answer admitting the material allegations of a petition filed against it in any such proceeding, such grace period will cease to apply.
(vii) Any other event occurs which, under the applicable Law of any applicable jurisdiction, has an effect equivalent to any of the events referred to in Section 11.01(h).
(i) Judgments. One or more judgments for the payment of money in an aggregate amount in excess of $1,500,000 (or the Equivalent Amount in other currencies) not covered by insurance shall be rendered against Holdings or any of its Subsidiaries or any combination thereof and the same shall remain undismissed, unsatisfied or undischarged for a period of thirty (30) calendar days during which execution shall not be effectively stayed, or any action shall be legally taken by a judgment creditor to attach or levy upon any assets of any Obligor to enforce any such judgment.
(j) ERISA and Pension Plans. An ERISA Event shall have occurred that, in the reasonable opinion of the Administrative Agent, when taken together with all other ERISA Events that have occurred, could reasonably be expected to result in liability of Holdings and its Subsidiaries in an aggregate amount exceeding $1,500,000 in the aggregate since the Closing Date.
(k) Change of Control. A Change of Control shall have occurred.
(l) Material Adverse Change, Etc. A Material Adverse Change, Material Adverse Effect or Material Regulatory Event shall have occurred.
(m) [Reserved].
(n) Hazardous Materials, Etc. Any Claim shall be asserted against Holdings or any of its Subsidiaries, whether accrued, absolute or contingent, based on or arising from the generation, storage, transport, handling or disposal of Hazardous Material by Holdings or any such Subsidiary, as applicable, or their predecessors, that are reasonably likely to be determined adversely to Holdings or any such Subsidiary, as applicable, and the amount thereof could, individually or in the aggregate, reasonably be expected to result in a Material Adverse Effect (insofar as such amount is payable by Holdings or any such Subsidiary, as applicable, but after deducting any portion thereof that is reasonably expected to be paid by insurance or other creditworthy Persons jointly and severally liable therefor).
(o) Impairment of Security, Etc. If any of the following events occurs, and with respect to the following clause (i), other than as a result of the acts or omissions of the Administrative Agent or any Lender: (i) any Lien created by any of the Security Documents shall at any time not constitute a valid and perfected Lien on the applicable Collateral in favor of the Secured Parties, free and clear of all other Liens (other than Permitted Liens), (ii) except for expiration in accordance with its terms, any of the Security Documents or any Guarantee of any of the Obligations (including that contained in Section 13) shall for whatever reason cease to be in full force and effect, or (iii) any Obligor shall, directly or indirectly, contest in any manner such effectiveness, validity, binding nature or enforceability of any such Lien or any Loan Document.
11.02 Remedies. Upon the occurrence and continuance of any Event of Default, then, and in every such event (other than an Event of Default described in Section 11.01(h)), and at any time thereafter during the continuance of such event, the Administrative Agent may, by notice to Holdings or the Borrower, declare the Loans then outstanding to be due and payable in whole (or in part, in which case any principal not so declared to be due and payable may thereafter be
declared to be due and payable), and thereupon the principal of the Loans so declared to be due and payable, together with accrued interest thereon and all fees and other Obligations, shall become due and payable immediately (in the case of the Loans, at the Prepayment Price therefor), without presentment, demand, protest or other notice of any kind, all of which are hereby waived by each Obligor; and in case of an Event of Default described in Section 11.01(h), the principal of the Loans then outstanding, together with accrued interest thereon and all fees and other Obligations, shall automatically become due and payable immediately (in the case of the Loans, at the Prepayment Price therefor), without presentment, demand, protest or other notice of any kind, all of which are hereby waived by each Obligor.
11.03 Additional Remedies. Upon the occurrence and during the continuance of an Event of Default, if Holdings or any of its Subsidiaries shall be in uncured default under a Material Agreement, the Administrative Agent or the Lenders shall have the right (but not the obligation) to cause the default or defaults under such Material Agreement to be remedied (including without limitation by paying any unpaid amount thereunder) and otherwise exercise any and all rights of Holdings or such Subsidiary, as the case may be, thereunder, as may be necessary to prevent or cure any default. Without limiting the foregoing, upon any such default, Holdings and each of its Subsidiaries shall promptly execute, acknowledge and deliver to the Administrative Agent such instruments as may reasonably be required of Holdings or such Subsidiary to permit the Administrative Agent and the Lenders to cure any default under the applicable Material Agreement or permit the Administrative Agent and the Lenders to take such other action required to enable the Administrative Agent and the Lenders to cure or remedy the matter in default and preserve the interests of the Administrative Agent or Lenders. Any amounts paid by the Administrative Agent or Lenders pursuant to this Section 11.03 shall be payable on demand by Obligors, shall accrue interest at the Default Rate if not paid on demand, and shall constitute “Obligations.”
SECTION 12
THE ADMINISTRATIVE AGENT
12.01 Appointment and Duties. Subject in all cases to clause (c) below:
(a) Appointment of the Administrative Agent. Each of the Lenders hereby irrevocably appoints Perceptive Credit Holdings III, LP (together with any successor the Administrative Agent pursuant to Section 12.09) as the Administrative Agent hereunder and authorizes the Administrative Agent to (i) execute and deliver the Loan Documents and accept delivery thereof on its behalf from Holdings or any of its Subsidiaries, (ii) take such action on its behalf and to exercise all rights, powers and remedies and perform the duties as are expressly delegated to the Administrative Agent under such Loan Documents and (iii) exercise such powers as are reasonably incidental thereto.
(b) Duties as Collateral and Disbursing Agent. Without limiting the generality of Section 12.01(a), the Administrative Agent shall have the sole and exclusive right and authority (to the exclusion of the Lenders), and is hereby authorized, to (i) act as the disbursing and collecting agent for the Lenders with respect to all payments and collections arising in connection with the Loan Documents (including in any proceeding described in Section 11.01(h) or any other bankruptcy, insolvency or similar proceeding), and each Person making any payment in connection with any Loan Document to any Secured Party is hereby authorized to make such
payment to the Administrative Agent, (ii) file and prove claims and file other documents necessary or desirable to allow the claims of the Secured Parties with respect to any Obligation in any proceeding described in Section 11.01(h) or any other bankruptcy, insolvency or similar proceeding (but not to vote, consent or otherwise act on behalf of such Secured Party), (iii) act as collateral agent for each Secured Party for purposes of the perfection of all Liens created by such agreements and all other purposes stated therein, (iv) manage, supervise and otherwise deal with the Collateral, (v) take such other action as is necessary or desirable to maintain the perfection and priority of the Liens created or purported to be created by the Loan Documents, (vi) except as may be otherwise specified in any Loan Document, exercise all remedies given to the Administrative Agent and the other Secured Parties with respect to the Collateral, whether under the Loan Documents, applicable Laws or otherwise and (vii) execute any amendment, consent or waiver under the Loan Documents on behalf of any Lender that has consented in writing to such amendment, consent or waiver; provided that the Administrative Agent hereby appoints, authorizes and directs each Lender to act as collateral sub-agent for the Administrative Agent and the Lenders for purposes of the perfection of all Liens with respect to the Collateral, including any deposit account maintained by any Obligor with, and cash and Permitted Cash Equivalent Investments held by, such Lender, and may further authorize and direct the Lenders to take further actions as collateral sub-agents for purposes of enforcing such Liens or otherwise to transfer the Collateral subject thereto to the Administrative Agent, and each Lender hereby agrees to take such further actions to the extent, and only to the extent, so authorized and directed.
(c) Limited Duties. The Lenders and the Obligors hereby each acknowledge and agree that the Administrative Agent (i) has undertaken its role hereunder purely as an accommodation to the parties hereto and the Transactions, (ii) is receiving no compensation for undertaking such role and (iii) subject only to the notice provisions set forth in Section 12.09, may resign from such role at any time for any reason or no reason whatsoever. Without limiting the foregoing, the parties hereto further acknowledge and agree that under the Loan Documents, the Administrative Agent (i) is acting solely on behalf of the Lenders (except to the limited extent provided in Section 12.11), with duties that are entirely administrative in nature and do not (and are not intended to) create any fiduciary obligations, notwithstanding the use of the defined term “the Administrative Agent”, the terms “agent”, “administrative agent” and “collateral agent” and similar terms in any Loan Document to refer to the Administrative Agent, which terms are used for title purposes only, (ii) is not assuming any obligation under any Loan Document other than as expressly set forth therein or any role as agent, fiduciary or trustee of or for any Lender or any other Secured Party and (iii) shall have no implied functions, responsibilities, duties, obligations or other liabilities under any Loan Document (fiduciary or otherwise), and each Lender hereby waives and agrees not to assert any claim against the Administrative Agent based on the roles, duties and legal relationships expressly disclaimed in this clause (c).
12.02 Binding Effect. Each Lender agrees that (i) any action taken by the Administrative Agent or the Majority Lenders (or, if expressly required hereby, a greater proportion of the Lenders) in accordance with the provisions of the Loan Documents, (ii) any action taken by the Administrative Agent in reliance upon the instructions of the Majority Lenders (or, where so required, such greater proportion) and (iii) the exercise by the Administrative Agent or the Majority Lenders (or, where so required, such greater proportion) of the powers set forth herein or therein, together with such other powers as are reasonably incidental thereto, shall be authorized and binding upon all of the Secured Parties.
12.03 Use of Discretion.
(a) No Action without Instructions. The Administrative Agent shall not be required to exercise any discretion or take, or to omit to take, any action, including with respect to enforcement or collection, except (subject to clause (b) below) any action it is required to take or omit to take (i) under any Loan Document or (ii) pursuant to instructions from the Majority Lenders (or, where expressly required by the terms of this Agreement, a greater proportion of the Lenders).
(b) Right Not to Follow Certain Instructions. Notwithstanding Section 12.03(a) or any other term or provision of this Section 12, the Administrative Agent shall not be required to take, or to omit to take, any action (i) unless, upon demand, the Administrative Agent receives an indemnification satisfactory to it from the Lenders (or, to the extent applicable and acceptable to the Administrative Agent, any other Secured Party) against all liabilities that, by reason of such action or omission, may be imposed on, incurred by or asserted against the Administrative Agent or any Related Parties thereof or (ii) that is, in the opinion of the Administrative Agent, in its sole and absolute discretion, contrary to any Loan Document, applicable Law or the best interests of the Administrative Agent or any of its Affiliates or Related Parties.
12.04 Delegation of Rights and Duties. The Administrative Agent may, upon any term or condition it specifies, delegate or exercise any of its rights, powers and remedies under, and delegate or perform any of its duties or any other action with respect to, any Loan Document by or through any trustee, co-agent, employee, attorney-in-fact and any other Person (including any Secured Party). Any such Person shall benefit from this Section 12 to the extent provided by the Administrative Agent.
12.05 Reliance and Liability.
(a) The Administrative Agent may, without incurring any liability hereunder, (i) consult with any of its Related Parties and, whether or not selected by it, any other advisors, accountants and other experts (including advisors to, and accountants and experts engaged by, any Obligor) and (ii) rely and act upon any document and information and any telephone message or conversation, in each case believed by it to be genuine and transmitted, signed or otherwise authenticated by the appropriate parties.
(b) Neither the Administrative Agent nor any of its Related Parties shall be liable for any action taken or omitted to be taken by any of them under or in connection with any Loan Document, and each Lender and Holdings and the Borrower hereby waives and shall not assert (and Holdings and the Borrower shall cause each other Obligor to waive and agree not to assert) any right, claim or cause of action based thereon, except to the extent of liabilities resulting primarily from the fraudulent conduct or behavior of the Administrative Agent or, as the case may be, such Related Party (each as determined in a final, non-appealable judgment or order by a court of competent jurisdiction) in connection with the duties expressly set forth herein. Without limiting the foregoing, the Administrative Agent:
(i) shall not be responsible or otherwise incur liability for any action or omission taken in reliance upon the instructions of the Majority Lenders or for the actions or omissions of any of their Related Parties selected with reasonable care (other than employees, officers and directors of the Administrative Agent, when acting on behalf of the Administrative Agent);
(ii) shall not be responsible to any Secured Party for the due execution, legality, validity, enforceability, effectiveness, genuineness, sufficiency or value of, or the attachment, perfection or priority of any Lien created or purported to be created under or in connection with, any Loan Document;
(iii) makes no warranty or representation, and shall not be responsible, to any Secured Party for any statement, document, information, representation or warranty made or furnished by or on behalf of any Related Party, in or in connection with any Loan Document or any transaction contemplated therein, whether or not transmitted by the Administrative Agent, including as to completeness, accuracy, scope or adequacy thereof, or for the scope, nature or results of any due diligence performed by the Administrative Agent in connection with the Loan Documents; and
(iv) shall not have any duty to ascertain or to inquire as to the performance or observance of any provision of any Loan Document, whether any condition set forth in any Loan Document is satisfied or waived, as to the financial condition of any Obligor or as to the existence or continuation or possible occurrence or continuation of any Default or Event of Default and shall not be deemed to have notice or knowledge of such occurrence or continuation unless it has received a notice from the Borrower, any Lender describing such Default or Event of Default clearly labeled “notice of default” (in which case the Administrative Agent shall promptly give notice of such receipt to all Lenders);
and, for each of the items set forth in clauses (i) through (iv) above, each Lender and the Borrower hereby waives and agrees not to assert (and the Borrower shall cause each other Obligor to waive and agree not to assert) any right, claim or cause of action it might have against the Administrative Agent based thereon.
12.06 Administrative Agent Individually. The Administrative Agent and its Affiliates may make loans and other extensions of credit to, acquire stock and stock equivalents of, engage in any kind of business with, any Obligor or Affiliate thereof as though it were not acting as the Administrative Agent and may receive separate fees and other payments therefor. To the extent the Administrative Agent or any of its Affiliates makes any Loan or otherwise becomes a Lender hereunder, it shall have and may exercise the same rights and powers hereunder and shall be subject to the same obligations and liabilities as any other Lender and the terms “Lender”, “Majority Lender”, and any similar terms shall, except where otherwise expressly provided in any Loan Document, include, without limitation, the Administrative Agent or such Affiliate, as the case may be, in its individual capacity as Lender or as one of the Majority Lenders, respectively.
12.07 Lender Credit Decision. Each Lender acknowledges that it has, independently and without reliance upon the Administrative Agent, any Lender or any of their Related Parties or upon any document solely or in part because such document was transmitted by the Administrative Agent or any of its Related Parties, conducted its own independent investigation of the financial condition and affairs of each Obligor and has made and continues to make its own credit decisions in connection with entering into, and taking or not taking any action under, any Loan Document or with respect to any transaction contemplated in any Loan Document, in each case based on such documents and information as it shall deem appropriate.
12.08 Expenses; Indemnities.
(a) Each Lender agrees to reimburse the Administrative Agent and each of its Related Parties (to the extent not reimbursed by any Obligor) promptly upon demand for such Lender’s Proportionate Share of any costs and expenses (including fees, charges and disbursements of
financial, legal and other advisors and Other Taxes paid in the name of, or on behalf of, any Obligor) that may be incurred by the Administrative Agent or any of its Related Parties in connection with the preparation, syndication, execution, delivery, administration, modification, consent, waiver or enforcement (whether through negotiations, through any work-out, bankruptcy, restructuring or other legal or other proceeding or otherwise) of, or legal advice in respect of its rights or responsibilities under, any Loan Document.
(b) Each Lender further agrees to indemnify the Administrative Agent and each of its Related Parties (to the extent not reimbursed by any Obligor), from and against such Lender’s aggregate Proportionate Share of the liabilities (including Taxes, interests and penalties imposed for not properly withholding or backup withholding on payments made to on or for the account of any Lender) that may be imposed on, incurred by or asserted against the Administrative Agent or any of its Related Parties in any matter relating to or arising out of, in connection with or as a result of any Loan Document or any other act, event or transaction related, contemplated in or attendant to any such Loan Document, or, in each case, any action taken or omitted to be taken by the Administrative Agent or any of its Related Parties under or with respect to any of the foregoing; provided that no Lender shall be liable to the Administrative Agent or any of its Related Parties to the extent such liability has resulted primarily from the gross negligence or willful misconduct of the Administrative Agent or, as the case may be, such Related Party, as determined by a court of competent jurisdiction in a final non-appealable judgment or order.
12.09 Resignation of the Administrative Agent.
(a) At any time upon not less than ten (10) Business Days prior written notice, the Administrative Agent may resign as the “the Administrative Agent” hereunder, in whole or in part (in the sole and absolute discretion of the Administrative Agent), effective on the date set forth in such notice, which effective date shall not be less than ten (10) Business Days following delivery of such notice. If the Administrative Agent delivers any such notice, the Majority Lenders shall have the right to appoint a successor to the Administrative Agent reasonably satisfactory to Holdings; provided that if a successor to the Administrative Agent has not been appointed on or before the effectiveness of the resignation of the resigning Administrative Agent, then the resigning Administrative Agent may, on behalf of the Lenders, appoint any Person reasonably chosen by it as the successor to the Administrative Agent.
(b) Effective immediately upon its resignation, (i) the resigning Administrative Agent shall be discharged from its duties and obligations under the Loan Documents to the extent set forth in the applicable resignation notice, (ii) the Lenders shall assume and perform all of the duties of the Administrative Agent until a successor the Administrative Agent shall have accepted a valid appointment hereunder, (iii) the resigning Administrative Agent and its Related Parties shall no longer have the benefit of any provision of any Loan Document other than with respect to (x) any actions taken or omitted to be taken while such resigning Administrative Agent was, or because the Administrative Agent had been, validly acting as the Administrative Agent under the Loan Documents or (y) any continuing duties such resigning Administrative Agent continues to perform, and (iv) subject to its rights under Section 12.04, the resigning Administrative Agent shall take such action as may be reasonably necessary to assign to the successor the Administrative Agent its rights as the Administrative Agent under the Loan Documents. Effective immediately upon its acceptance of a valid appointment as the Administrative Agent, a successor the Administrative
Agent shall succeed to, and become vested with, all the rights, powers, privileges and duties of the resigning Administrative Agent under the Loan Documents.
12.10 Release of Collateral or Guarantors.
(a) Each Lender hereby consents to the release and hereby directs the Administrative Agent to release (or, in the case of Section 12.10(a)(ii)(B), release or subordinate) the following:
(i) any Subsidiary of Holdings from its guaranty of any Obligation of any Obligor if all of the Equity Interests in such Subsidiary owned by any Obligor or any of its Subsidiaries are disposed of in an Asset Sale permitted under the Loan Documents (including pursuant to a waiver or consent), to the extent that, after giving effect to such Asset Sale, such Subsidiary would not be required to guaranty any Obligations pursuant to Section 8.12(a); and
(ii) any Lien held by the Administrative Agent for the benefit of the Secured Parties against (A) any Collateral that is disposed of by an Obligor in an Asset Sale permitted by the Loan Documents (including pursuant to a valid waiver or consent), (B) any property subject to a Lien described in Section 9.02(c), and (C) all of the Collateral and all Obligors, upon (w) termination of the Commitments, (x) payment and satisfaction in full of all Loans and all other Obligations (other than Warrant Obligations and inchoate indemnification and expense reimbursement obligations for which no Claim has been made) that the Administrative Agent has been notified in writing are then due and payable, (y) deposit of cash collateral with respect to all contingent Obligations (other than Warrant Obligations and inchoate indemnification and expense reimbursement obligations for which no Claim has been made), in amounts and on terms and conditions and with parties satisfactory to the Administrative Agent and each Indemnified Party that is owed such Obligations, and (z) to the extent requested by the Administrative Agent, receipt by the Secured Parties of liability releases from the Obligors, each in form and substance acceptable to the Administrative Agent.
(b) So long as no Event of Default has occurred and is continuing, Holdings and the Borrower may from time to time request that the Administrative Agent and the Lenders consent to the release of the Liens created pursuant to the Security Documents on the Specified Intellectual Property in order to permit Holdings and the Borrower to enter into or become bound by an outbound license of such Specified Intellectual Property that is permitted hereunder (such consent not to be unreasonably conditioned, delayed or withheld); provided that, notwithstanding the foregoing, the Administrative Agent shall continue to have a Lien on Holdings’ and the Borrower’s rights as a licensor under such outbound license including any amounts received by or other proceeds payable to Holdings and the Borrower in respect thereof; provided further that promptly upon request therefor, Holdings and the Borrower shall furnish to the Administrative Agent such information, documents and agreements relating to such outbound license as the Administrative Agent may reasonably request.
Each Lender hereby directs the Administrative Agent, and the Administrative Agent hereby agrees, upon receipt of reasonable advance notice from Holdings and the Borrower, to execute and deliver or file such documents and to perform other actions reasonably necessary to release the guarantees and Liens when and as directed in this Section 12.10.
12.11 Additional Secured Parties. The benefit of the provisions of the Loan Documents directly relating to the Collateral or any Lien granted thereunder shall extend to and be available to any Secured Party that is not a Lender so long as, by accepting such benefits, such Secured Party
agrees, as among the Administrative Agent and all other Secured Parties, that such Secured Party is bound by (and, if requested by the Administrative Agent, shall confirm such agreement in a writing in form and substance acceptable to the Administrative Agent) this Section 12 and the decisions and actions of the Administrative Agent and the Majority Lenders (or, where expressly required by the terms of this Agreement, a greater proportion of the Lenders) to the same extent a Lender is bound; provided that, notwithstanding the foregoing, (i) such Secured Party shall be bound by Section 12.08 only to the extent of liabilities, costs and expenses with respect to or otherwise relating to the Collateral held for the benefit of such Secured Party, in which case the obligations of such Secured Party thereunder shall not be limited by any concept of Proportionate Share or similar concept, (ii) each of the Administrative Agent and each Lender shall be entitled to act at its sole discretion, without regard to the interest of such Secured Party, regardless of whether any Obligation to such Secured Party thereafter remains outstanding, is deprived of the benefit of the Collateral, becomes unsecured or is otherwise affected or put in jeopardy thereby, and without any duty or liability to such Secured Party or any such Obligation and (iii) such Secured Party shall not have any right to be notified of, consent to, direct, require or be heard with respect to, any action taken or omitted in respect of the Collateral or under any Loan Document.
SECTION 13
GUARANTEE
13.01 The Guarantee. Holdings and the Subsidiary Guarantors hereby jointly and severally guarantee to the Administrative Agent and the Lenders, and their successors and assigns, the prompt payment in full when due (whether at stated maturity, by acceleration or otherwise) of the principal of and interest on the Loans, all fees and other amounts and Obligations from time to time owing to the Administrative Agent and the Lenders by the Borrower and each other Obligor under this Agreement or under any other Loan Document, in each case strictly in accordance with the terms hereof and thereof (such obligations being herein collectively called the “Guaranteed Obligations”). Holdings and the Subsidiary Guarantors hereby further jointly and severally agree that if the Borrower or any other Obligor shall fail to pay in full when due (whether at stated maturity, by acceleration or otherwise) any of the Guaranteed Obligations, Holdings and the Subsidiary Guarantors shall promptly pay the same, without any demand or notice whatsoever, and that in the case of any extension of time of payment or renewal of any of the Guaranteed Obligations, the same shall be promptly paid in full when due (whether at extended maturity, by acceleration or otherwise) in accordance with the terms of such extension or renewal.
13.02 Obligations Unconditional. The obligations of Holdings and the Subsidiary Guarantors under Section 13.01 are absolute and unconditional, joint and several, irrespective of the value, genuineness, validity, regularity or enforceability of the obligations of Holdings, the Borrower or any other Subsidiary Guarantor under this Agreement or any other agreement or instrument referred to herein, or any substitution, release or exchange of any other guarantee of or security for any of the Guaranteed Obligations, and, to the fullest extent permitted by all applicable Laws, irrespective of any other circumstance whatsoever that might otherwise constitute a legal or equitable discharge or defense of a surety or guarantor, it being the intent of this Section 13.02 that the obligations of Holdings and the Subsidiary Guarantors hereunder shall be absolute and unconditional, joint and several, under any and all circumstances. Without limiting the generality of the foregoing, it is agreed that the occurrence of any one or more of the following shall not alter
or impair the liability of Holdings and the Subsidiary Guarantors hereunder, which shall remain absolute and unconditional as described above:
(a) at any time or from time to time, without notice to Holdings and the Subsidiary Guarantors, the time for any performance of or compliance with any of the Guaranteed Obligations shall be extended, or such performance or compliance shall be waived;
(b) any of the acts mentioned in any of the provisions of this Agreement or any other agreement or instrument referred to herein shall be done or omitted;
(c) the maturity of any of the Guaranteed Obligations shall be accelerated, or any of the Guaranteed Obligations shall be modified, supplemented or amended in any respect, or any right under this Agreement or any other agreement or instrument referred to herein shall be waived or any other guarantee of any of the Guaranteed Obligations or any security therefor shall be released or exchanged in whole or in part or otherwise dealt with; or
(d) any lien or security interest granted to, or in favor of, the Secured Parties as security for any of the Guaranteed Obligations shall fail to be perfected.
Holdings and the Subsidiary Guarantors hereby expressly waive diligence, presentment, demand of payment, protest and all notices whatsoever, and any requirement that the Administrative Agent or any Lender exhaust any right, power or remedy or proceed against the Borrower or any other Subsidiary Guarantor under this Agreement or any other agreement or instrument referred to herein, or against any other Person under any other guarantee of, or security for, any of the Guaranteed Obligations.
13.03 Reinstatement. The obligations of Holdings and the Subsidiary Guarantors under this Section 13 shall be automatically reinstated if and to the extent that for any reason any payment by or on behalf of the Borrower in respect of the Guaranteed Obligations is rescinded or must be otherwise restored by any holder of any of the Guaranteed Obligations, whether as a result of any proceedings in bankruptcy or reorganization or otherwise, and Holdings and the Subsidiary Guarantors jointly and severally agree that they shall indemnify the Secured Parties on demand for all reasonable costs and expenses (including fees of counsel) incurred by such Persons in connection with such rescission or restoration, including any such costs and expenses incurred in defending against any claim alleging that such payment constituted a preference, fraudulent transfer or similar payment under any bankruptcy, insolvency or similar law.
13.04 Subrogation. Holdings and the Subsidiary Guarantors hereby jointly and severally agree that, until the payment and satisfaction in full of all Guaranteed Obligations and the expiration and termination of the Commitments, they shall not exercise any right or remedy arising by reason of any performance by them of their guarantee in Section 13.01, whether by subrogation or otherwise, against the Borrower or any other guarantor of any of the Guaranteed Obligations or any security for any of the Guaranteed Obligations.
13.05 Remedies. Holdings and the Subsidiary Guarantors jointly and severally agree that, as between Holdings and the Subsidiary Guarantors, on one hand, and the Administrative Agent and the Lenders, on the other hand, the obligations of the Borrower under this Agreement and under the other Loan Documents may be declared to be forthwith due and payable as provided in Section
11 (and shall be deemed to have become automatically due and payable in the circumstances provided in Section 11) for purposes of Section 13.01 notwithstanding any stay, injunction or other prohibition preventing such declaration (or such obligations from becoming automatically due and payable) as against the Borrower and that, in the event of such declaration (or such obligations being deemed to have become automatically due and payable), such obligations (whether or not due and payable by the Borrower) shall forthwith become due and payable by Holdings and the Subsidiary Guarantors for purposes of Section 13.01.
13.06 Instrument for the Payment of Money. Holdings and each Subsidiary Guarantor hereby acknowledges that the guarantee in this Section 13 constitutes an instrument for the payment of money, and consents and agrees that the Administrative Agent and the Lenders, at their sole option, in the event of a dispute by Holdings and such Subsidiary Guarantor in the payment of any moneys due hereunder, shall have the right to proceed by motion for summary judgment in lieu of complaint pursuant to N.Y. Civ. Prac. L&R § 3213.
13.07 Continuing Guarantee. The guarantee in this Section 13 is a continuing guarantee, and shall apply to all Guaranteed Obligations whenever arising.
13.08 General Limitation on Guarantee Obligations. In any action or proceeding involving any provincial, territorial or state corporate law, or any state or federal bankruptcy, insolvency, reorganization or other law affecting the rights of creditors generally, if the obligations of Holdings or any Subsidiary Guarantor under Section 13.01 would otherwise be held or determined to be void, invalid or unenforceable, or subordinated to the claims of any other creditors, on account of the amount of its liability under Section 13.01, then, notwithstanding any other provision hereof to the contrary, the amount of such liability shall, without any further action by Holdings or such Subsidiary Guarantor, the Administrative Agent, any Lender or any other Person, be automatically limited and reduced to the highest amount that is valid and enforceable and not subordinated to the claims of other creditors as determined in such action or proceeding.
SECTION 14
MISCELLANEOUS
14.01 No Waiver. No failure on the part of the Administrative Agent or the Lenders to exercise and no delay in exercising, and no course of dealing with respect to, any right, power or privilege under any Loan Document shall operate as a waiver thereof, nor shall any single or partial exercise of any right, power or privilege under any Loan Document preclude any other or further exercise thereof or the exercise of any other right, power or privilege. The remedies provided herein are cumulative and not exclusive of any remedies provided by law.
14.02 Notices. All notices, requests, instructions, directions and other communications provided for herein (including any modifications of, or waivers, requests or consents under, this Agreement) or in the other Loan Documents shall be given or made in writing (including by telecopy or email) delivered, if to Holdings, the Borrower, another Obligor, the Administrative Agent or any Lender, to its address specified on the signature pages hereto or its Guarantee Assumption Agreement, as the case may be, or at such other address as shall be designated by such party in a written notice to the other parties. Except as otherwise provided in this Agreement or therein, all such communications shall be deemed to have been duly given upon receipt of a legible copy thereof,
in each case given or addressed as aforesaid. All such communications provided for herein by telecopy shall be confirmed in writing promptly after the delivery of such communication (it being understood that non-receipt of written confirmation of such communication shall not invalidate such communication). Notwithstanding anything to the contrary in this Agreement or any other Loan Document, notices, documents, certificates and other deliverables to the Lenders by any Obligor may be made solely to the Administrative Agent and the Administrative Agent shall promptly deliver such notices, documents, certificates and other deliverables to the Lenders.
14.03 Expenses, Indemnification, Etc.
(a) Expenses. Each Obligor, jointly and severally, agrees to pay or reimburse (i) the Administrative Agent for all of its reasonable costs and expenses (including the reasonable fees and expenses of Morrison & Foerster LLP, counsel to the Administrative Agent, and printing, reproduction, document delivery, communication and travel costs) in connection with (x) the negotiation, preparation, execution and delivery of this Agreement and the other Loan Documents and the making of the Loans (exclusive of post-closing costs), and the Administrative Agent agrees to apply the Expense Deposit to such costs and expenses, (y) post-closing costs and (z) the negotiation or preparation of any modification, supplement or waiver of any of the terms of this Agreement or any of the other Loan Documents (whether or not consummated) and (ii) the Administrative Agent and the Lenders for all of their reasonable out-of-pocket costs and expenses (including the reasonable out-of-pocket fees and expenses of one legal counsel for the Administrative Agent and one legal counsel for all other Lenders in the aggregate) in connection with any enforcement or collection proceedings resulting from the occurrence and continuance of an Event of Default.
(b) Indemnification. Each Obligor, jointly and severally, hereby indemnifies the Administrative Agent, the Lenders and their respective Affiliates, directors, officers, employees, attorneys, agents, advisors and controlling parties (each, an “Indemnified Party”) from and against, and agrees to hold them harmless against, any and all Claims and Losses of any kind (including reasonable fees and disbursements of counsel), joint or several, that may be incurred by or asserted or awarded against any Indemnified Party, in each case arising out of or in connection with or relating to any investigation, litigation or proceeding or the preparation of any defense with respect thereto arising out of or in connection with or relating to this Agreement or any of the other Loan Documents or the Transactions or any use made or proposed to be made with the proceeds of the Loans, whether or not such investigation, litigation or proceeding is brought by any Obligor, any of its Subsidiaries, any of its shareholders or creditors, an Indemnified Party or any other Person, or an Indemnified Party is otherwise a party thereto, and whether or not any of the conditions precedent set forth in Section 6 are satisfied or the other transactions contemplated by this Agreement are consummated, except to the extent such Claim or Loss (x) is found in a final, non-appealable judgment by a court of competent jurisdiction to have resulted from an Indemnified Party’s gross negligence or willful misconduct, (y) results from a Claim brought by an Obligor against an Indemnified Party for breach of such Indemnified Party’s obligations hereunder or under any other Loan Document, if such Obligor has obtained a final and non-appealable judgment in its favor on such Claim as determined by a court of competent jurisdiction or (z) results from a Claim not involving an act or omission of an Obligor and that is brought by an Indemnified Party against another Indemnified Party (other than against the Administrative Agent in its capacity as such). No Obligor shall assert any Claim against any Indemnified Party, on any theory of liability, for
consequential, indirect, special or punitive damages arising out of or otherwise relating to this Agreement or any of the other Loan Documents or any of the Transactions or the actual or proposed use of the proceeds of the Loans. Holdings, its Subsidiaries and Affiliates and their respective directors, officers, employees, attorneys, agents, advisors and controlling parties are each sometimes referred to in this Agreement as an “Obligor Party.” No Lender shall assert any Claim against any Obligor Party, on any theory of liability, for consequential, indirect, special or punitive damages arising out of or otherwise relating to this Agreement or any of the other Loan Documents or any of the Transactions or the actual or proposed use of the proceeds of the Loans. This Section 14.03 shall not apply with respect to Taxes other than any Taxes that represent losses, claims or damages arising from any non-Tax Claim.
14.04 Amendments, Etc. Except as otherwise expressly provided in this Agreement, any provision of this Agreement and any other Loan Document may be modified or supplemented only by an instrument in writing signed by Holdings, the Borrower, the Administrative Agent and the Majority Lenders; provided that:
(a) any such modification or supplement that is disproportionately adverse to any Lender as compared to other Lenders or subjects any Lender to any additional obligation shall not be effective without the consent of such affected Lender;
(b) the consent of all of the Lenders directly affected thereby shall be required to:
(i) amend, modify, discharge, terminate or waive any of the terms of this Agreement or any other Loan Agreement if such amendment, modification, discharge, termination or waiver would increase the amount of the Loans or Commitment of such Lender, reduce the fees payable to such Lender hereunder, reduce interest rates or other amounts payable with respect to the Loans held by such Lender, extend any date fixed for payment of principal, interest or other amounts payable relating to the Loans held by such Lender or extend the repayment dates of the Loans held by such Lender;
(ii) amend, modify, discharge, terminate or waive any Security Document if the effect is to release a material part of the Collateral subject thereto other than pursuant to the terms hereof or thereof; or
(iii) amend this Section 14.04 or the definition of “Majority Lenders”; and
(c) if the Administrative Agent and Holdings shall have jointly identified an obvious error or any error or omission of a technical nature, in each case, in any provision of the Loan Documents, then the Administrative Agent and Holdings shall be permitted to amend such provision, and, in each case, such amendment shall become effective without any further action or consent of any other party to any Loan Document if the same is not objected to in writing by the Majority Lenders to the Administrative Agent within ten (10) Business Days following receipt of notice thereof.
14.05 Successors and Assigns.
(a) General. The provisions of this Agreement and the other Loan Documents shall be binding upon and shall inure to the benefit of the parties hereto or thereto and their respective successors and assigns permitted hereby or thereby, except that no Obligor may assign or otherwise
transfer any of its rights or obligations hereunder without the prior written consent of the Administrative Agent. Any Lender may assign or otherwisetransfer any of its rights or obligations hereunder or under any of the other Loan Documents (i) to an assignee in accordance with the provisions of Section 14.05(b), (ii) by way of participation in accordance with the provisions of Section 14.05(e), or (iii) by way of pledge or assignment of a security interest subject to the restrictions of Section 14.05(h). Nothing in this Agreement, expressed or implied, shall be construed to confer upon any Person (other than the parties hereto, their respective successors and assigns permitted hereby, Participants to the extent provided in Section 14.05(e) and, to the extent expressly contemplated hereby, the Related Parties of each of the Administrative Agent and the Lenders) any legal or equitable right, remedy or claim under or by reason of this Agreement.
(b) Assignments by Lender. Any Lender may at any time assign to one or more Eligible Transferees (or, if an Event of Default has occurred and is continuing, to any Person) all or a portion of its rights and obligations under this Agreement (including all or a portion of the Loans at the time owing to it) and the other Loan Documents; provided that (i) no such assignment shall be made to any Obligor, any Affiliate of any Obligor, or any employees or directors of any Obligor at any time, and (ii) no such assignment shall be made without the prior written consent of the Administrative Agent. Subject to the recording thereof by the Lender pursuant to Section 14.05(d), from and after the effective date specified in each Assignment and Assumption, the assignee thereunder shall be a party to this Agreement and, to the extent of the interest assigned by such Assignment and Assumption, have the rights and obligations of the Lender under this Agreement and the other Loan Documents, and correspondingly the assigning Lender shall, to the extent of the interest assigned by such Assignment and Assumption, be released from its obligations under this Agreement (and, in the case of an Assignment and Assumption covering all of the Lender’s rights and obligations under this Agreement, such Lender shall cease to be a party hereto) and the other Loan Documents but shall continue to be entitled to the benefits of Section 5 and Section 14.03. Any assignment or transfer by the Lender of rights or obligations under this Agreement that does not comply with this Section 14.05(b) shall be treated for purposes of this Agreement as a sale by such Lender of a participation in such rights and obligations in accordance with Section 14.05(e).
(c) Amendments to Loan Documents. Each of the Administrative Agent, the Lenders, Holdings and its Subsidiaries agrees to enter into such amendments to the Loan Documents, and such additional Security Documents and other instruments and agreements, in each case in form and substance reasonably acceptable to the Administrative Agent, the Lenders, Holdings and its Subsidiaries, as shall reasonably be necessary to implement and give effect to any assignment made under this Section 14.05.
(d) Register. The Administrative Agent, acting solely for this purpose as a non-fiduciary agent of the Borrower, shall maintain at one of its offices in the United States a copy of each Assignment and Assumption delivered to it and a register for the recordation of the names and addresses of the Lenders, and the Commitments of, and principal amounts (and stated interest) of the Loans owing to, each Lender pursuant to the terms hereof from time to time (the “Register”). The entries in the Register shall be conclusive absent manifest error, and the Borrower, the Administrative Agent and the Lenders shall treat each Person whose name is recorded in the Register pursuant to the terms hereof as a Lender hereunder for all purposes of this Agreement.
The Register shall be available for inspection by the Borrower and any Lender, at any reasonable time and from time to time upon reasonable prior notice.
(e) Participations. Any Lender may at any time, without the consent of, or notice to, Holdings or the Borrower, sell participations to any Person (other than a natural person or any Obligor or any of its Subsidiaries or Affiliates) (each, a “Participant”) in all or a portion of the Lender’s rights and/or obligations under this Agreement (including all or a portion of the Commitment and/or the Loans owing to it); provided that (i) such Lender’s obligations under this Agreement shall remain unchanged, (ii) such Lender shall remain solely responsible to the other parties hereto for the performance of such obligations and (iii) the Borrower shall continue to deal solely and directly with such Lender in connection therewith. Any agreement or instrument pursuant to which any Lender sells such a participation shall provide that such Lender shall retain the sole right to enforce the Loan Documents and to approve any amendment, modification or waiver of any provision of the Loan Documents; provided that such agreement or instrument may provide that such Lender shall not, without the consent of the Participant, agree to any amendment, modification or waiver that would (i) increase or extend the term of such Lender’s Commitment, (ii) extend the date fixed for the payment of principal of or interest on the Loans or any portion of any fee hereunder payable to the Participant, (iii) reduce the amount of any such payment of principal, or (iv) reduce the rate at which interest is payable thereon to a level below the rate at which the Participant is entitled to receive such interest. Subject to Section 14.05(f), the Borrower agrees that each Participant shall be entitled to the benefits of Section 5 (subject to the requirements and limitations therein including the requirements under Section 5.03(f) (it being understood that the documentation required under Section 5.03(f) shall be delivered to the participating Lender)) to the same extent as if it were a Lender and had acquired its interest by assignment pursuant to Section 14.05(b); provided that such Participant agrees to be subject to the provisions of Section 5.04 as if it were an assignee under Section 14.05(b) above. To the extent permitted by applicable Law, each Participant also shall be entitled to the benefits of Section 4.03(a) as though it were a Lender.
(f) Limitations on Rights of Participants. A Participant shall not be entitled to receive any greater payment under Sections 5.01 or 5.03 with respect to any participation than its participating Lender would have been entitled to receive, except to the extent such entitlement to receive a greater payment results from a change in Law that occurs after the Participant acquired the applicable participation.
(g) Participant Register. Each Lender that sells a participation shall, acting solely for this purpose as a non-fiduciary agent of the Borrower, maintain a register on which it enters the name and address of each Participant and the principal amounts (and stated interest) of each Participant’s interest in the Loans or other Obligations under the Loan Documents (the “Participant Register”); provided that no Lender shall have any obligation to disclose all or any portion of the Participant Register (including the identity of any Participant or any information relating to a Participant’s interest in any Commitments, Loans, or its other Obligations under any Loan Document) to any Person except to the extent that such disclosure is necessary to establish that such Commitment, Loan, or other Obligation is in registered form under Section 5f.103-1(c) of the United States Treasury Regulations. The entries in the Participant Register shall be conclusive absent manifest error, and such Lender shall treat each Person whose name is recorded in the Participant Register as the owner of such participation for all purposes of this Agreement
notwithstanding any notice to the contrary. For the avoidance of doubt, the Administrative Agent (in its capacity as Administrative Agent) shall have no responsibility for maintaining a Participant Register.
(h) Certain Pledges. Any Lender may at any time pledge or assign a security interest in all or any portion of its rights under the Loan Documents to secure obligations of such Lender, including any pledge or assignment to secure obligations to a Federal Reserve Bank; provided that no such pledge or assignment shall release such Lender from any of its obligations hereunder or substitute any such pledgee or assignee for such Lender as a party hereto.
14.06 Survival. The obligations of the Obligors under Sections 5.01, 5.02, 5.03, 14.03, 14.05, 14.06, 14.09, 14.10, 14.11, 14.12, 14.13, 14.14 and the obligations of Holdings and the Subsidiary Guarantors under Section 13 (solely to the extent guaranteeing any of the obligations under the foregoing Sections) shall survive the repayment of the Obligations and the termination of the Commitment and, in the case of the Lenders’ assignment of any interest in the Commitment or the Loans hereunder, shall survive, in the case of any event or circumstance that occurred prior to the effective date of such assignment, the making of such assignment, notwithstanding that the Lenders may cease to be “Lenders” hereunder.
14.07 Captions. The table of contents and captions and section headings appearing herein are included solely for convenience of reference and are not intended to affect the interpretation of any provision of this Agreement.
14.08 Counterparts; Electronic Signatures. This Agreement may be executed in any number of counterparts, all of which taken together shall constitute one and the same instrument and any of the parties hereto may execute this Agreement by signing any such counterpart. Delivery of an executed signature page of this Agreement by facsimile transmission or electronic transmission (in PDF format) shall be effective as delivery of a manually executed counterpart hereof. Any signature (including, without limitation, (x) any electronic symbol or process attached to, or associated with, a contract or other record and adopted by a Person with the intent to sign, authenticate or accept such contract or record and (y) any facsimile or .pdf signature) hereto or the other Loan Documents or to any other certificate, agreement or document related to any Loan Document or the Transactions, and any contract formation or record-keeping, in each case, through electronic means, shall have the same legal validity and enforceability as a manually executed signature or use of a paper-based record-keeping system to the fullest extent permitted by applicable law, including the Federal Electronic Signatures in Global and National Commerce Act, the New York State Electronic Signatures and Records Act, or any similar state law based on the Uniform Electronic Transactions Act, and the parties hereto hereby waive any objection to the contrary.
14.09 Governing Law. This Agreement and the rights and obligations of the parties hereunder shall be governed by, and construed in accordance with, the lawChoice of Law; Forum; Jury Waiver. The validity, interpretation and performance of this Agreement shall be governed by, and construed in accordance with, the internal laws of the StateCommonwealth of New YorkMassachusetts, without regardgiving effect to principles of conflicts of laws that would result in the application of the laws of any other jurisdiction; provided that Section 5-1401 of the New York General Obligations Law shall apply.
14.10 Jurisdiction, Service of Process and Venue.conflict of law principles. Both parties agree that any action, demand, claim or counterclaim relating to the terms and provisions of this
(a) Submission to Jurisdiction. Each Obligor agreesAgreement or to its breach, shall be commenced in the Commonwealth of Massachusetts in a court of competent jurisdiction. Both parties agree that any suit, action or proceeding with respect to this Agreementaction, demand, claim or counterclaim concerning your employment and separation of employment, and/or this Agreement, shall be tried by a judge alone, and both parties hereby waive and forever renounce the right to a trial before a civil jury.
(h)OWBPA. It is Pear’s desire and intent to make certain that you fully understand the provisions and effects of this Agreement. To that end, you acknowledge that you are hereby advised to consult with an attorney before signing this Agreement, particularly the release of ADEA claims, and that you have been given an opportunity to do so. You also acknowledge that, consistent with the OWBPA the Company has given you forty-five (45) days (the “Review Period”) to consider signing this Agreement. You further acknowledge that the Review Period is not and will not be affected or extended by any revisions, whether material or immaterial, that might be made to this Agreement. orIn anythe other Loan Document to which it is a party or any judgment entered by any court in respect thereof may be brought initially in the federal or state courtsevent you sign this Agreement before the Review Period has expired, you acknowledge that it is your intent to voluntarily waive the remainder of the Review Period. This Agreement will become effective seven (7) days after it is signed by you (the “Revocation Period”). Also consistent with the OWBPA, you may revoke this Agreement within the Revocation Period, and you understand that it shall not become effective until the expiration of the Revocation Period. To be effective, such rescission must be hand delivered or postmarked within the Revocation Period and sent by certified mail, return receipt requested, to Pear Therapeutics, Inc., attn. Ronan O’Brien, 200 State Street, Boston, MA 02109 with a copy via email to ronan.obrien@peartherapeutics.com. If you choose to revoke this Agreement, you understand that the Agreement will become null and void and that the Company will have no obligation to provide you with the Separation Pay and Benefits as set forth in this Agreement.
In addition, in accordance with OWBPA, you are receiving with this Agreement a list of
(i) the job titles and ages of all employees in the relevant decisional unit who in New York, New York and irrevocably submits to the non-exclusive jurisdiction of each such court for the purpose of any such suit, action, proceeding or judgment. This Section 14.10(a) iswere selected for the severance pay and benefits outlined in this Agreement; and (ii) the job titles and ages of all employees in the relevant decisional unit who were not selected for the benefit of the Administrative Agent and the Lenders only and, as a result, no Lender shall be prevented from taking proceedings in any other courts with jurisdiction. To the extent allowed by any applicable Law, the Lenders may take concurrent proceedings in any number of jurisdictions.
(b) Alternative Process. Nothing herein shall in any way be deemed to limit the ability of the Administrative Agent and the Lenders to serve any process or summons in any manner permitted by any applicable Law.severance pay and benefits as outlined in this Agreement.
(c) i)Waiver of Venue, Etc. Each Obligor irrevocably waives to the fullest extent permitted by law any objection that itYou acknowledge that (a) may now or hereafter have to the laying of the venue of any suit, action or proceeding arising out of or relating to this Agreement or any other Loan Document and hereby further irrevocably waives to the fullest extent permitted by law any claim that any such suit, action or proceeding brought in any such court has been brought in an inconvenient forum. A final judgment (in respect of which time for all appeals has elapsed) in any such suit, action or proceeding shall be conclusive and may be enforced in any court to the jurisdiction of which such Obligor is or may be subject, by suit upon judgment.
14.11 Waiver of Jury Trial. EACH PARTY HERETO HEREBY IRREVOCABLY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY SUIT, ACTION OR PROCEEDING ARISING OUT OF OR RELATING TO THIS AGREEMENT, THE OTHER LOAN DOCUMENTS OR THE TRANSACTIONS CONTEMPLATED HEREBY OR THEREBY.
14.12 Waiver of Immunity. To the extent that any Obligor may be or become entitled to claim for itself or its property or revenues any immunity on the ground of sovereignty or the like from suit, court jurisdiction, attachment prior to judgment, attachment in aid of execution of a judgment or execution of a judgment, and to the extent that in any such jurisdiction there may be attributed such an immunity (whether or not claimed), such Obligor hereby irrevocably agrees not to claim and hereby irrevocably waives such immunity with respect to its obligations under this Agreement andyour agreements and obligations under this Agreement are the other Loan Documents.
14.13 Entire Agreement. This Agreement and the other Loan Documents constitute the entire agreement among the parties with respectmade voluntarily, knowingly and without duress; (b) neither Pear nor its agents or representatives have made any representations to the subject matter hereof and thereof and supersede any and all previous agreements and understandings, oral or written, relating to the subject matter hereof, including any confidentiality (or similar) agreements. EACH OBLIGOR ACKNOWLEDGES, REPRESENTS AND WARRANTS THAT IN DECIDING TO ENTER INTO THIS AGREEMENT AND THE OTHER LOAN DOCUMENTS OR IN TAKING OR NOT TAKING ANY ACTION HEREUNDER OR THEREUNDER, IT HAS NOT RELIED, AND SHALL NOT RELY, ON ANY STATEMENT, REPRESENTATION, WARRANTY, COVENANT, AGREEMENT OR UNDERSTANDING, WHETHER WRITTEN OR ORAL, OF
OR WITH ADMINISTRATIVE AGENT OR THE LENDERS OTHER THAN THOSE EXPRESSLY SET FORTH IN THIS AGREEMENT AND THE OTHER LOAN DOCUMENTS.
14.14 Severability. If any provision hereof is found by a court to be invalid or unenforceable, to the fullest extent permitted by any applicable Law the parties agree that such invalidity or unenforceability shall not impair the validity or enforceability of any other provision hereof.
14.15 No Fiduciary Relationship. Holdings and the Borrower acknowledge that the Administrative Agent and the Lenders have no fiduciary relationship with, or fiduciary duty to, Holdings and the Borrower arising out of or in connection with this Agreement or the other Loan Documents, and the relationship between the Lenders and the Borrower is solely that of creditor and debtor. This Agreement and the other Loan Documents do not create a joint venture among the parties.
14.16 Confidentiality. The Administrative Agent and each Lender agree to keep confidential all non-public and other confidential information provided to them in writing by any Obligor pursuant to this Agreement in accordance with its customary procedures for handling its own confidential information; provided that nothing herein shall prevent the Administrative Agent or any Lender from disclosing any such information (i) to the Administrative Agent, any other Lender or, subject to an agreement to comply with the provisions of this Section 14.16, any Affiliate of a Lender or any Eligible Transferee or other assignee permitted under Section 14.05(b), (ii) subject to an agreement to comply with the provisions of this Section, to any actual or prospective direct or indirect counterparty to any Hedging Agreement (or any professional advisor to such counterparty), (iii) on a confidential basis to its employees, officers, directors, agents, attorneys, accountants, trustees and other professional advisors or those of any of its affiliates (collectively, its “Related Parties”); provided that the applicable Lender shall remain liable hereunder for any breach of this Section 14.16 by any of its Related Parties, (iv) upon the request or demand of any Governmental Authority or any Regulatory Authority purporting to have jurisdiction over such Person or its Related Parties (including any self-regulatory authority, such as the National Association of Insurance Commissioners), (v) in response to any order of any court or other Governmental Authority or as may otherwise be required pursuant to any applicable Law, (vi) if requested or required to do so in connection with any litigation or similar proceeding, (vii) that has been publicly disclosed (other than as a result of a disclosure in violation of this Section 14.16), (viii) to the National Association of Insurance Commissioners or any similar organization or any nationally recognized rating agency that requires access to information about a Lender’s investment portfolio in connection with ratings issued with respect to such Lender, (ix) in connection with the exercise of any remedy permitted hereunder or under any other Loan Document, (x) on a confidential basis to (A) any rating agency in connection with rating Holdings or any of its Subsidiaries or the Loans or (B) the CUSIP Service Bureau or any similar agency in connection with the issuance and monitoring of CUSIP numbers of other market identifiers with respect to the Loans or (xi) to any other party hereto; provided, further that, unless specifically prohibited by applicable law or court order, each Lender shall notify Holdings of any request by any Governmental Authority or representative thereof (other than any such request in connection with any examination of the financial condition or other routine examination of such Lender by such Governmental Authority) for disclosure of any such non-public information prior to disclosure of such information.
14.17 Interest Rate Limitation. Notwithstanding anything herein to the contrary, if at any time the interest rate applicable to any Loan, together with all fees, charges and other amounts that are treated as interest on such Loan under applicable Law (collectively, “charges”), shall exceed the maximum lawful rate (the “Maximum Rate”) that may be contracted for, charged, taken, received or reserved by the Administrative Agent and the Lender holding such Loan in accordance with applicable Law, the rate of interest payable in respect of such Loan hereunder, together with all charges payable in respect thereof, shall be limited to the Maximum Rate. To the extent lawful, the interest and charges that would have been paid in respect of such Loan but were not paid as a result of the operation of this Section shall be cumulated and the interest and charges payable to such Lender in respect of other Loans or periods shall be increased (but not above the amount collectible at the Maximum Rate therefor) until such cumulated amount, together with interest thereon at the Federal Funds Effective Rate for each day to the date of repayment, shall have been received by such Lender. Any amount collected by such Lender that exceeds the maximum amount collectible at the Maximum Rate shall be applied to the reduction of the principal balance of such Loan so that at no time shall the interest and charges paid or payable in respect of such Loan exceed the maximum amount collectible at the Maximum Rate.
14.18 Early Prepayment Fee. The parties hereto acknowledge and agree that, to the extent the Early Prepayment Fee is applicable to any repayment or prepayment of principal of any Loan at any time, such Early Prepayment Fee is not intended to be a penalty assessed as a result of any such repayment or prepayment of the Loans, but rather is the product of a good faith, arm’s length commercial negotiation between Holdings and the Borrower and the Lenders relating to the mutually satisfactory compensation payable to the Lenders by Holdings and the Borrower in respect of the Loans made hereunder. In furtherance of the foregoing, to the fullest extent permitted by applicable Law, the Obligors hereby jointly and severally waive any rights or Claims any of them may have under any such applicable Law (whether or not in effect on the Closing Date) that would prohibit or restrict the payment of the Early Prepayment Fee under any of the circumstances provided herein or in any other Loan Document, including payment after acceleration of the Loans.
14.19 Judgment Currency.
(a) If, for the purposes of obtaining judgment in any court, it is necessary to convert a sum due hereunder in Dollars into another currency, the parties hereto agree, to the fullest extent permitted by Law, that the rate of exchange used shall be that at which, in accordance with normal banking procedures, the Administrative Agent could purchase Dollars with such other currency at the buying spot rate of exchange in the New York foreign exchange market on the Business Day immediately preceding that on which any such judgment, or any relevant part thereof, is given.
(b) The obligations of the Obligors in respect of any sum due to the Administrative Agent hereunder and under the other Loan Documents shall, notwithstanding any judgment in a currency other than Dollars, be discharged only to the extent that on the Business Day following receipt by the Administrative Agent of any sum adjudged to be so due in such other currency the Administrative Agent may, in accordance with normal banking procedures, purchase Dollars with such other currency. If the amount of Dollars so purchased is less than the sum originally due to the Administrative Agent in Dollars, Holdings and the Borrower agree, to the fullest extent that they may effectively do so, as a separate obligation and notwithstanding any such judgment, to
indemnify the Administrative Agent against such loss. If the amount of Dollars so purchased exceeds the sum originally due to the Administrative Agent in Dollars, the Administrative Agent shall remit such excess to Holdings and the Borrower.
14.20 USA PATRIOT Act. The Administrative Agent and the Lenders hereby notify Holdings and its Subsidiaries that pursuant to the requirements of the USA PATRIOT Act (Title III of Pub. L. 107-56 (signed into law October 26, 2001)) (the “Patriot Act”) and the Beneficial Ownership Regulation, they are required to obtain, verify and record information that identifies Holdings and its Subsidiaries, which information includes the name and address of Holdings and its Subsidiaries and other information that will allow such Person to identify Holdings, the Borrower or such Subsidiary in accordance with the Patriot Act and the Beneficial Ownership Regulation.
14.21 Acknowledgement and Consent to Bail-In of EEA Financial Institutions. Notwithstanding anything to the contrary in any Loan Document or in any other agreement, arrangement or understanding among any such parties, each party hereto acknowledges that any liability of any Affected Institution arising under any Loan Document, to the extent such liability is unsecured, may be subject to the Write-Down and Conversion Powers of the applicable Resolution Authority and agrees and consents to, and acknowledges and agrees to be bound by:
(a) the application of any Write-Down and Conversion Powers by the applicable Resolution Authority to any such liabilities arising hereunder which may be payable to it by any party hereto that is an Affected Financial Institution; and
(b) the effects of any Bail-In Action on any such liability, including, if applicable:
(i) a reduction in full or in part or cancellation of any such liability;
(ii) a conversion of all, or a portion of, such liability into shares or other instruments of ownership in such Affected Financial Institution, its parent undertaking, or a bridge institution that may be issued to it or otherwise conferred on it, and that such shares or other instruments of ownership will be accepted by it in lieu of any rights with respect to any such liability under this Agreement or any other Loan Document; or
(iii) the variation of the terms of such liability in connection with the exercise of the Write-Down and Conversion Powers of the applicable Resolution Authority.
14.22 Amendment and Restatement; No Novation. On the Effective Date, upon the effectiveness of this Agreement, the Original Credit Agreement shallyou inconsistent with the provisions of this Agreement, and (c) in signing this Agreement, you are not relying on be amended and restated in its entiretyany other statements or explanations made to you by this Agreement. This Agreement shall not extinguish the obligations for the payment of money outstanding under the Original Credit Agreement or discharge or release any Loan Document. Nothing herein contained shall be construed as a substitution, novation, accord or satisfaction of the obligations outstanding under the Original Credit Agreement or the other Loan Documents. Nothing in this Agreement shall be construed as a release or other discharge of the Borrower or any other Obligor from its obligations and liabilities under the Original Credit Agreement or the other Loan Documents. On the Effective Date, any and all references in any Loan Documents to the Original Credit Agreement shall be deemed to be amended to refer to this Agreement. The Borrower hereby ratifies and reaffirms all of its liabilities and obligations under the Loan Documents and agrees that all such liabilities and obligations are carried forward in this Agreement
Pear that are not reflected in this Agreement.
andIf the other Loan Documents. The guaranties, liens, security interests, pledges, covenants and agreements set forth inforegoing correctly sets forth our arrangement, please sign, date and return the Loan Documents entered into in connection with the Original Credit Agreement are made and granted to secure and support the Obligations under this Agreement as if the same were made or granted on the Effective Date and the Loan Documents entered into in connection with the Original Credit Agreement shall continue in full force and effect. The Borrower agrees to cooperate and use commercially reasonably efforts to promptly take, or cause to be taken, all actions and to promptly do, or cause to be done, all things reasonably necessary, proper or advisable to fulfill its obligations hereunder.
[Signature Pages Follow]
IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be duly executed and delivered as ofenclosed copy of this Agreement to me within the day and year firsttimeframe set forth above written.
| | | | | | | | |
| BORROWER: |
| | |
| PEAR THERAPEUTICS (US), INC. |
| | |
| By | /s/ Ronan O’Brien |
| | Name: Ronan O’Brien |
| |
|
Very truly yours,
/s/ Christopher D.T. Guiffre
Christopher D.T. Guiffre
Title: General CounselChief Financial Officer & Chief Compliance Officer | | |
| | |
| Address for Notices: |
| | |
| 200 State Street, 13th Floor Boston, MA 02109 |
| Attn: General Counsel |
| Email: ronan.obrien@peartherapeutics.com |
| | |
| | |
| HOLDINGS: |
| | |
| PEAR THERAPEUTICS, INC. |
| | |
| By | /s/ Ronan O’Brien |
| | Name: Ronan O’Brien |
| | Title: Secretary |
| | |
| | |
| Address for Notices: |
| | |
| 200 State Street, 13th Floor Boston, MA 02109 |
| Attn: General Counsel |
| Email: ronan.obrien@peartherapeutics.com |
[SIGNATURE PAGE TO AMENDED AND RESTATED CREDIT AGREEMENT AND GUARANTY]Operating Officer
| | | | | | | | |
| ADMINISTRATIVE AGENT: |
| | |
| PERCEPTIVE CREDIT HOLDINGS III, LP |
| | |
| By: PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC,
Accepted and Agreed To: Katherine Jeffery its general partner |
| | |
| | |
| By | /s/ Sandeep Dixit |
| | Name: Sandeep Dixit |
| | Title: Chief Credit Officer |
| | |
| | |
| By | /s/ Sam Chawla |
| | Name: Sam Chawla |
| | Title: Portfolio Manager |
| | |
| | |
| Address for Notices: |
| Perceptive Credit Holdings III, LP
c/o Perceptive Advisors LLC
51 Astor Place, 10th Floor
New York, NY 10003 |
| Attn: Sandeep Dixit |
| Email: Sandeep@perceptivelife.com |
| PCOFReporting@perceptivelife.com |
/s/ Katherine Jeffery
Dated: 12/19/2022
| | | | | | | | |
| LENDERS: |
| | |
| PERCEPTIVE CREDIT HOLDINGS III, LP |
| | |
| By: PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner |
| | |
| | |
| By | /s/ Sandeep Dixit |
| | Name: Sandeep Dixit |
| | Title: Chief Credit Officer |
| | |
| | |
| By | /s/ Sam Chawla |
| | Name: Sam Chawla |
| | Title: Portfolio Manager |
| | |
| | |
| Address for Notices: |
| Perceptive Credit Holdings III, LP
c/o Perceptive Advisors LLC
51 Astor Place, 10th Floor
New York, NY 10003 |
| Attn: Sandeep Dixit |
| Email: Sandeep@perceptivelife.com |
| PCOFReporting@perceptivelife.com |
[SIGNATURE PAGE TO AMENDED AND RESTATED CREDIT AGREEMENT AND GUARANTY]
EXHIBIT A
TO CREDIT AGREEMENT AND GUARANTY
FORM OF NOTEDeclaration Regarding Return of Company Property
[DATE]
U.S. $ [30,000,000] [10,000,000] [10,000,000]I, Kathy Jeffery, declare under penalty of perjury as follows:
FOR VALUE RECEIVED, the undersigned, Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), hereby promises to pay to Perceptive Credit Holdings III, LP (the “Lender
1.I have returned to Pear all data (including work product, financial data, documents and computer data, regardless of form or medium), and all equipment (including keys, devices and computer software), in my possession, custody or control that relate to the business of Pear.
2.This includes, but is not limited to, all things and data identified in Section 8, return of property, of my employee non-disclosure, non-solicitation and assignment of intellectual property agreement, which states:
7. Return of property. Upon termination of my employment with the company, or at any other time upon request of the company, I shall return promptly any and all customer or prospective customer or business partner or prospective business partner lists, other customer or prospective customer or business partner or prospective business partner information or related materials, computer programs, software, electronic data, specifications, drawings, blueprints, data storage devices, reproductions, sketches, notes, notebooks, memoranda, reports, records, proposals, business plans, or copies of them, other documents or materials, tools, equipment, or other property belonging to the company or its customers or business partners which I may then possess or have under my control, in any form, and including all copies of the same. I further agree that upon termination of employment I shall not take with me any documents or data in any form or of any description containing or pertaining to confidential information or inventions.
3.I confirm that I have complied with that Section 9.
4.I further agree that I have provided Pear with access to all usernames and passwords maintained by me on behalf of Pear.
5.After returning all data that relate to the business of Pear, I deleted/destroyed all copies or abstracts of such data in my possession, custody or control. I also confirm that if I discover any other company materials in my possession after the Separation Date, I will immediately return such materials to Pear and thereafter delete the same.
I declare under penalty of perjury that the foregoing is true and correct.
Signature: /s/ Katherine Jeffery Dated: 02/21/2023
Katherine Jeffery
EXHIBIT B
Employee Nondisclosure, Non-solicitation, and
Assignment of Intellectual Property Agreement
I, Katherine Jeffery, acknowledge and agree that in consideration and as a condition of my employment by Pear Therapeutics, Inc., a Delaware corporation (the “Company”), in immediately available funds,and of the aggregate principal sum set forth above, or,compensation to be paid to me, and in recognition of the fact that as an employee of the Company I will have access to confidential information, trade secrets and goodwill belonging to the Company, I agree with the Company as follows:
1.Acknowledgments.
a.By signing this Agreement I confirm my understanding and acknowledgement that the Company has expended substantial time, expense and effort in developing and maintaining its trade secrets and other Confidential Information (as defined below) and its goodwill with its Customers, employees, contractors, and Business Partners, which goodwill I acknowledge and agree belongs exclusively to the Company. I specifically acknowledge and agree that the nature, scope and period of the covenants set forth in Section if less, the aggregate unpaid principal amount of the [Tranche 1] [Tranche 2] [Tranche 3] Loan made by the Lender pursuant to Section 2.01(a) of the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time6 below are reasonable and necessary to protect the Company’s trade secrets and other Confidential Information and goodwill. I also acknowledge and agree that in my position with the Company, I will be performing services for and have access to and use in the performance of my duties the Company’s Confidential Information and/or trade secrets regarding its products, processes, procedures, research and development, strategies and/or financial information. I further acknowledge that I have been offered substantial consideration for my agreements and covenants set forth in this Agreement, including my continued employment. “Customers” include, without limitation, current or prospective patients and/or users of the Company’s products or product candidates. “Business Partners” include, without limitation, Alliance Partners, Channel Partners, Clinical Partners and Healthcare Providers. “Alliance Partners” include, without limitation, developers or commercialization partners that develop or commercialize the Company’s products or product candidates. “Channel Partners” include, without limitation, any distribution platform or operator or other third party who is authorized to make the Company’s products or product candidates available for sale or distribution. “Clinical Partners” include, without limitation, hospitals, clinics, practices, other medical groups or healthcare systems that have contracted with the Company. “Healthcare Providers” include, without limitation, physicians, patient advocates, counselors, practitioners, recovery coaches, or other persons who provide healthcare or related services to Customers.
a.I hereby represent, warrant and agree that (i) I have disclosed to the Company any and all continuing obligations to previous employers or others that do or may restrict or purport to restrict in any manner my ability to provide services to the Company and/or require me not to disclose any information to the Company, by providing a copy of all documents containing such continuing obligations to the Company; (ii) in the course of performing services for the Company, I will not infringe or wrongfully appropriate any patents, copyrights, trade secrets or other intellectual property rights of any person or entity anywhere in the world; and (iii) I have not and will not disclose or use during my employment by the Company any confidential information that I acquired as a result of any previous employment or consulting arrangement or under a previous obligation of confidentiality.
1.Confidential Information. While employed by the Company and thereafter, I shall not, directly or indirectly, use any Confidential Information (as hereinafter defined) other than pursuant to my employment by and for the benefit of the Company, or disclose any Confidential Information to time, the “Credit Agreement”), among the Borrower, certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”), on the date or dates specified in the Credit Agreement, together with interest on the principal amount of such Loans from time to time outstanding thereunder at the rates, and payable in the manner and on the dates, specified in the Credit Agreement.
This Note is a Note issued pursuant to the terms of Section 2.03 of the Credit Agreement, and this Note and the holder hereof are entitled to all the benefits and security provided for thereby or referred to therein, to which Credit Agreement reference is hereby made for a statement thereof. All defined terms used in this Note, except terms otherwise defined herein, shall have the same meaning as in the Credit Agreement.
THIS NOTE AND THE RIGHTS AND OBLIGATIONS OF THE PARTIES HEREUNDER SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH, THE LAW OF THE STATE OF NEW YORK, WITHOUT REGARD TO PRINCIPLES OF CONFLICTS OF LAWS THAT WOULD RESULT IN THE APPLICATION OF THE LAWS OF ANY OTHER JURISDICTION; PROVIDED THAT SECTION 5-1401 OF THE NEW YORK GENERAL OBLIGATIONS LAW SHALL APPLY.
The Borrower hereby waives demand, presentment, protest or notice of any kind hereunder, other than notices provided for in the Loan Documents. The non-exercise by the holder hereof of any of its rights hereunder in any particular instance shall not constitute a waiver thereof in such particular or any subsequent instance.
THIS NOTE MAY NOT BE TRANSFERRED EXCEPT IN COMPLIANCE WITH THE TERMS OF THE CREDIT AGREEMENT.
[THIS NOTE HAS BEEN ISSUED WITH “ORIGINAL ISSUE DISCOUNT” (WITHIN THE MEANING OF SECTION 1273 OF THE INTERNAL REVENUE CODE OF 1986, AS AMENDED). UPON WRITTEN REQUEST, THE BORROWER WILL PROMPTLY MAKE AVAILABLE TO ANY HOLDER OF THIS NOTE THE FOLLOWING INFORMATION: (1)anyone outside of the Company,
whether by private communication, public address, publication or otherwise, or disclose any Confidential Information to anyone within the Company who has not been authorized to receive such information, in each case except as directed in writing by an authorized representative of the Company. The term “Confidential Information” as used throughout this Agreement shall mean all trade secrets, proprietary information, know-how, data, designs, specifications, processes, Customer lists, Business Partner lists, and other technical or business information (and any evidence, record or representation thereof, as well as all metadata associated with any electronic files that constitute or contain the same), whether prepared, conceived or developed by a consultant or employee of the Company (including myself) or received by the Company from an outside source, which is in the possession of the Company (whether or not the property of the Company), which in any way relates to the present or future business of the Company, which is maintained in confidence by the Company, or which might permit the Company or its Business Partners to obtain a competitive advantage over competitors who do not have access to such trade secrets, proprietary information, or other data or information. Without limiting the generality of the foregoing, Confidential Information shall include:
a.any idea, improvement, invention, innovation, development, technical data, design, formula, device, pattern, concept, art, method, process, procedure, form, strategy, technique, machine, manufacturing method, composition of matter, computer program, software, firmware, source code, object code, algorithm, subroutine, object module, schematic, model, diagram, flow chart, user manual, training or service manual, product specification, plan for a new or revised product, compilation of information, or work in process, and any and all revisions and improvements relating to any of the foregoing (in each case whether or not reduced to tangible form);
a.any sales or marketing material, plan, or survey, business plan or opportunity, product or development plan or specification, business proposal, budget, fee schedule, financial data or record, or business data or record, or other record or information relating to the present or proposed business of the Company or any Customer or Business Partner (or prospective Customer or Business Partner);
a.information in any form, whether in a list or otherwise, concerning Customers, prospective Customers, Business Partners, or prospective Business Partners, including but not limited to names or lists of names, personal or financial information, requirements, and financial data of any of the foregoing, and any and all contracts, agreements or other arrangements with the same; and
a.the education and employment history, personal data and information, resumes, wages, compensation, and compensation and/or incentive plans of the Company’s current, former or prospective employees, contractors or interns and any lists, directories or organizational charts listing the Company’s employees, contractors or interns (to the extent the same are not made public in violation of any legal obligation).
Notwithstanding the foregoing, the term Confidential Information shall not apply to information which the Company has voluntarily disclosed to the public without restriction, or which has otherwise lawfully entered the public domain.
I understand that the Company from time to time has in its possession information (including product and development plans and specifications) which is claimed by Customers or Business Partners and others to be proprietary and which the Company has agreed to keep confidential. I agree that all such information shall be Confidential Information for purposes of this Agreement.
Consistent with applicable law, the Company is providing you with notice of the following provisions of the Defend Trade Secrets Act of 2016: “Immunity from Liability for Confidential Disclosure of a Trade Secret to the Government or in a Court Filing— (1) Immunity.—An individual shall not be held criminally or civilly liable under
THE ISSUE PRICEany Federal or AND ISSUE DATE OFState trade secret law THIS NOTE, (2) THE AMOUNTfor the disclosure of a trade secret that—(A) is made –(i) in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney; and (ii) solely for the purpose of reporting or investigating a suspected violation of law; or (B) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. (2) Use of OF ORIGINAL ISSUE DISCOUNT ON THIS NOTE ANDTrade Secret Information in Anti-Retaliation Lawsuit—An individual who (3)files THEa YIELD TO MATURITY OFlawsuit for retaliation by THIS NOTE. HOLDERS SHOULD CONTACT THE CHIEF FINANCIAL OFFICER OF THE BORROWER AT [ADDRESS OF THE BORROWER]].
[Signature Page Follows]
| | | | | | | | |
| PEAR THERAPEUTICS, INC. |
| | |
| | |
| By | |
| Name: | |
| Title: | |
EXHIBIT B
TO CREDIT AGREEMENT AND GUARANTY
FORM OF BORROWING NOTICE
Date: [__________]
To: Perceptive Credit Holdings III, LP, as Administrative Agent
c/o Perceptive Advisors LLC
51 Astor Place, 10th Floor
New York, NY 10003
Attn: Sandeep Dixit
Email: Sandeep@perceptivelife.com and PCOF@perceptivelife.com
Re: Borrowing under the Credit Agreement
Ladies and Gentlemen:
The undersigned, Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), refers to the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among the Borrower, certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”). Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Credit Agreement.
The Borrower hereby gives you irrevocable notice, pursuant to Section 2.02 of the Credit Agreement, ofan employer for reporting a suspected violation of law may disclose the trade secret to the attorney of the individual and use the trade secret information in the court proceeding, if the individual— (A) files any document containing the borrowing of the Loan specified herein:
1. The Borrowing Date is [__________].1
2. The amount of the proposed Borrowing is $[____].
3. The payment instructions with respect to the funds to be made available to the Borrower are as follows:
Bank name:
Bank Address:
Routing Number:
Account Number:
The Borrower hereby certifies that, both immediately before and after giving effect to
1 Pursuant to Section 2.02 of the Credit Agreement, this Borrowing Notice must be delivered to the Administrative Agent at least three (3), but not more than seven (7), Business Days prior to the Borrowing Date. If received by the Administrative Agent on a day that is not a Business Day or after 12:00 p.m. (New York City time) on a Business Day, this Borrowing Notice will be deemed to have been delivered on the next Business Day.
both the Borrowing and the application of proceeds thereof on the Borrowing Date, the following statements are true and correct:
a) both immediately before and after giving effect to the Borrowing, (x) all representations and warranties set forth in each Loan Document that are qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct as of the date hereof, (y) the representations and warranties set forth in each Loan Document that are not qualified by materiality, Material Adverse Effect or the like are, in each case, true and correct in all material respects as of the date hereof, and (z) no Default has occurred and is continuing, or could reasonably be expected to result from the making of the Loans being advanced, or the consummation of any Transactions contemplated to occur, on the Borrowing Date; and
b) on and astrade secret under seal; and
(B) does not disclose the trade secret, except pursuant to court order.”
1.Ownership and Assignment of the Borrowing Date, there has occurred no Material Adverse Change since December 31, 2019.
[Signature Page Follows]
IN WITNESS WHEREOF, the Borrower has caused this Borrowing Notice to be duly executed and delivered as of the day and year first above written.
| | | | | | | | |
| BORROWER: |
| | |
| PEAR THERAPEUTICS, INC. |
| | |
| | |
| By | |
| Name: | |
| Title: | |
EXHIBIT C
TO CREDIT AGREEMENT AND GUARANTY
FORM OF GUARANTEE ASSUMPTION AGREEMENT
GUARANTEE ASSUMPTION AGREEMENT, dated as of [DATE] (this “Intellectual Property.
a.Guarantee and Assumption Agreement”), by [NAME OF ADDITIONAL SUBSIDIARY GUARANTOR], a [state of organization] [corporation][limited liability company] (the “Additional Subsidiary Guarantor”), under that certain Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”). Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Credit Agreement.
Pursuant to Section 8.12(a)(i) of the Credit Agreement, the Additional Subsidiary Guarantor hereby agrees to become a “Subsidiary Guarantor” for all purposes of the Credit Agreement, a “Grantor” for all purposes of the Security Agreement. Without limiting the foregoing, the Additional Subsidiary Guarantor hereby, jointly and severally with the other Subsidiary Guarantors, guarantees to the Lenders and their respective successors and assigns the prompt payment in full when due (whether at stated maturity, by acceleration or otherwise) of all Guaranteed ObligationsI agree that all originals and all copies of all manuscripts, drawings, prints, manuals, diagrams, letters, notes, notebooks, reports, models, records, files, memoranda, plans, sketches and all other documents and materials containing, representing, evidencing, recording, or constituting any Confidential Information (as defined in Section 13.012 of the Credit Agreement) in above), however and whenever produced (whether by myself or others) during the course of my employment, shall be the sole property of the Company.
a.I agree that all Confidential Information and all other discoveries, inventions, ideas, concepts, trademarks, service marks, logos, processes, products, formulas, computer programs or software, source codes, object codes, algorithms, machines, apparatuses, items of manufacture or composition of matter, or any new uses therefor or improvements thereon, or any new designs or modifications or configurations of any kind, or works of authorship of any kind, including, without limitation, compilations and derivative works, whether or not patentable or copyrightable, conceived, developed, reduced to practice or otherwise made by me, either alone or with others, and in any way related to the same manner and to the same extent as is provided in Section 13 of the Credit Agreement. In addition, as of the date hereof,business of the Company or to tasks assigned to me during the course of my employment, whether or not made during my regular working hours, whether or not conceived, developed, reduced to practice or made on the Company’s premises and whether or not disclosed by me to the Company (collectively “Inventions”), and any and all services and products which embody, emulate or employ any such Invention or Confidential Information shall be the sole property of the Company and all copyrights, patents, patent rights, trademarks and reproduction rights to, and
other proprietary rights in, each such Invention or Confidential Information, whether or not patentable or copyrightable, shall belong exclusively to the Company.
a.Subject to subsections (d) and, if applicable, (e) below, I agree to, and hereby do, assign to the Company all my right, title and interest throughout the world in and to all Inventions and to anything tangible which evidences, incorporates, constitutes, represents or records any Invention. I agree that all Inventions shall constitute works made for hire under the copyright laws of the United States and hereby assign and, to the extent any such assignment cannot be made at present, I hereby agree to assign to the Company all copyrights, patents and other proprietary rights I may have in any Inventions, together with the right to file for and/or own wholly without restriction United States and foreign patents, trademarks, and copyrights. I agree to waive, and hereby waive, all moral rights or proprietary rights in or to any Inventions and, to the extent that such rights may not be waived, agree not to assert such rights against the Company or its licensees, successors or assigns.
a.I hereby certify that Exhibit A sets forth any and all confidential information and intellectual property that I claim as my own or otherwise intend to exclude from this Agreement because it was developed by me prior to the date of this Agreement. I understand that after execution of this Agreement I shall have no right to exclude Confidential Information or Inventions from this Agreement.
a.For California Employees Only: I understand that the provisions of this Agreement requiring assignment of Inventions to the Company do not apply to any invention which qualifies fully under the provisions of California Labor Code Section 2870, which states: “(a) Any provision in an employment agreement which provides that an employee shall assign, or offer to assign, any of his or her rights in an invention to his or her employer shall not apply to an invention that the employee developed entirely on his or her own time without using the employer’s equipment, supplies, facilities, or trade secret information except for those inventions that either: (1) Relate at the time of conception or reduction to practice of the invention to the Additional Subsidiary Guarantor hereby makes the representations and warranties set forth in Section 7 of the Credit Agreement and in Section 4 of the Security Agreement, with respect to itself and its obligations under this Guarantee and Assumption Agreement and the other Loan Documents, as if each reference in such Sections to the Loan Documents included reference to this Guarantee and Assumption Agreement, such representations and warranties to be made as of the date hereof. [Attached asemployer’s business, or actual or demonstrably anticipated research or development of the employer; or (2) Result from any work performed by the employee for the employer.” I will advise the Company promptly in writing of any inventions that I believe meet the criteria in California Labor Code Section 2870 and are not otherwise disclosed on Exhibit A hereto are supplemental to the Schedules set forth in Section 7 to the Credit Agreement and Section 4 of the Security Agreement, updated to include the Additional Subsidiary Guarantor.]2
The Additional Subsidiary Guarantor hereby instructs its counsel to deliver the opinions referred to in .
1.Employee’s Obligation to Keep Records. I shall make and maintain adequate and current written records of all Inventions, including notebooks and invention disclosures, which records shall be available to and remain the property of the Company at all times. I shall disclose all Inventions promptly, fully and in writing to the Company immediately upon production or development of the same and at any time upon request.
1.Employee’s Obligation to CooperateSection 8.12(a)(iv) of the Credit Agreement to the Administrative Agent. I will, at any time during my employment, or after it terminates, upon request of the Company, execute all documents and perform all lawful acts which the Company considers necessary or advisable to secure its rights hereunder and the Lenders.
THIS GUARANTEE AND ASSUMPTION AGREEMENT AND THE RIGHTS AND OBLIGATIONS OF THE PARTIES HEREUNDER SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH, THE LAWS OF THE STATE OF NEW YORK, WITHOUT REGARD TO PRINCIPLES OF CONFLICTS OF LAWS THAT WOULD RESULT IN THE APPLICATION OF THE LAWS OF ANY OTHER JURISDICTION; PROVIDED THAT SECTION 5-1401 OF THE NEW YORK GENERAL OBLIGATIONS
2 [To be included if any Schedules set forth in Section 7 of the Credit Agreement or Section 4 of the Security Agreement need to be updated in connection with the making of representations and warranties with respect to the Additional Subsidiary Guarantor.]
to carry out the intent of this Agreement. Without limiting the generality of the foregoing, I will assist the Company in any reasonable manner to obtain for its own benefit patents or copyrights in any and all countries with respect to all Inventions assigned pursuant to Section 3, and I will execute, when requested, patent and other applications and assignments thereof to the Company,
LAW SHALL APPLY.
[Signature Page Follows]
IN WITNESS WHEREOF, the Additional Subsidiary Guarantor has causedor persons designated by it, and any other lawful documents deemed necessary by the Company to carry out the purposes of this Guarantee Assumption Agreement to be duly executedAgreement, and I will further assist the Company in every way to enforce any patents and delivered as of the day and year first above written.
| | | | | | | | |
| [ADDITIONAL SUBSIDIARY GUARANTOR] |
| | |
| By | |
| Name: | |
| Title: | |
| | |
| Address for Notices: |
| [__________] |
| [__________] |
| Attn: | [__________] |
| Tel.: | [__________] |
| Fax: | [__________] |
| Email: | [__________] |
EXHIBIT A TO GUARANTEE ASSUMPTION AGREEMENT
[ATTACH UPDATED SCHEDULES, IF NEEDED]
EXHIBIT D-1
TO CREDIT AGREEMENT AND GUARANTY
FORM OF U.S. TAX COMPLIANCE CERTIFICATE
(For Foreign Lenders That Are Not Partnerships for U.S. Federal Income Tax Purposes)
Reference is made to the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc. (the “Borrower”), a Delaware corporation, certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”).
Pursuant to the provisions of Section 5.03(f) of the Credit Agreement, the undersigned hereby certifies that (i) it is the sole record and beneficial owner of the Loan(s) (as well as any Note(s) evidencing such Loan(s)) in respect of which it is providing this certificate, (ii) it is not a “bank” within the meaning of Section 881(c)(3)(A) of the Code, (iii) it is not a “ten percent shareholder” of the Borrower within the meaning of Section 871(h)(3)(B) of the Code and (iv) it is not a “controlled foreign corporation” related to the Borrower as described in Section 881(c)(3)(C) of the Code.
The undersigned has furnished the Administrative Agent and the Borrower with a certificate of its non-U.S. Person status on IRS Form W-8BEN or IRS Form W-8BEN-E (or applicable successor form). By executing this certificate, the undersigned agrees that (1) if the information provided in this certificate changes, the undersigned shall promptly so inform the Borrower and the Administrative Agent, and (2) the undersigned shall have at all times furnished the Borrower and the Administrative Agent with a properly completed and currently effective certificate in either the calendar year in which each payment is to be made to the undersigned, or in either of the two calendar years preceding such payments.
Unless otherwise defined herein, terms defined in the Credit Agreement and used herein shall have the meanings given to them in the Credit Agreement.
[Signature Page Follows]
| | | | | | | | |
| [NAME OF LENDER] | |
| | |
| By: | | |
| Name: | |
| Title: | |
| | |
| Date: | [________ __], 20[ ] | |
EXHIBIT D-2
TO CREDIT AGREEMENT AND GUARANTY
FORM OF U.S. TAX COMPLIANCE CERTIFICATE
(For Foreign Participants That Are Not Partnerships for U.S. Federal Income Tax Purposes)
Reference is made to the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”).
Pursuant to the provisions of Section 5.03(f) of the Credit Agreement, the undersigned hereby certifies that (i) it is the sole record and beneficial owner of the participation in respect of which it is providing this certificate, (ii) it is not a “bank” within the meaning of Section 881(c)(3)(A) of the Code, (iii) it is not a “ten percent shareholder” of the Borrower within the meaning of Section 871(h)(3)(B) of the Code, and (iv) it is not a “controlled foreign corporation” related to the Borrower as described in Section 881(c)(3)(C) of the Code.
The undersigned has furnished its participating Lender with a certificate of its non-U.S. Person status on IRS Form W-8BEN or IRS Form W-8BEN-E (or applicable successor form). By executing this certificate, the undersigned agrees that (1) if the information provided in this certificate changes, the undersigned shall promptly so inform such Lender in writing, and (2) the undersigned shall have at all times furnished such Lender with a properly completed and currently effective certificate in either the calendar year in which each payment is to be made to the undersigned, or in either of the two calendar years preceding such payments.
Unless otherwise defined herein, terms defined in the Credit Agreement and used herein shall have the meanings given to them in the Credit Agreement.
[Signature Page Follows]
| | | | | | | | |
| [NAME OF PARTICIPANT] | |
| | |
| By: | | |
| Name: | |
| Title: | |
| | |
| Date: | [________ __], 20[ ] | |
EXHIBIT D-3
TO CREDIT AGREEMENT AND GUARANTY
FORM OF U.S. TAX COMPLIANCE CERTIFICATE
(For Foreign Participants That Are Partnerships for U.S. Federal Income Tax Purposes)
Reference is made to the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”).
Pursuant to the provisions of Section 5.03(f) of the Credit Agreement, the undersigned hereby certifies that (i) it is the sole record owner of the participation in respect of which it is providing this certificate, (ii) its direct or indirect partners/members are the sole beneficial owners of such participation, (iii) with respect such participation, neither the undersigned nor any of its direct or indirect partners/members is a “bank” extending credit pursuant to a loan agreement entered into in the ordinary course of its trade or business within the meaning of Section 881(c)(3)(A) of the Code, (iv) none of its direct or indirect partners/members is a “ten percent shareholder” of the Borrower within the meaning of Section 871(h)(3)(B) of the Code and (v) none of its direct or indirect partners/members is a “controlled foreign corporation” related to the Borrower as described in Section 881(c)(3)(C) of the Code.
The undersigned has furnished its participating Lender with an IRS Form W-8IMY (or applicable successor form) accompanied by one of the following forms from each of its partners/members that is claiming the portfolio interest exemption: (i) an IRS Form W-8BEN or IRS Form W-8BEN-E (or applicable successor form) or (ii) an IRS Form W-8IMY (or applicable successor form) accompanied by an IRS Form W-8BEN or IRS Form W-8BEN-E (or applicable successor form) from each of such partner’s/member’s beneficial owners that is claiming the portfolio interest exemption. By executing this certificate, the undersigned agrees that (1) if the information provided in this certificate changes, the undersigned shall promptly so inform such Lender and (2) the undersigned shall have at all times furnished such Lender with a properly completed and currently effective certificate in either the calendar year in which each payment is to be made to the undersigned, or in either of the two calendar years preceding such payments.
Unless otherwise defined herein, terms defined in the Credit Agreement and used herein shall have the meanings given to them in the Credit Agreement.
[Signature Page Follows]
| | | | | | | | |
| [NAME OF PARTICIPANT] | |
| | |
| By: | | |
| Name: | |
| Title: | |
| | |
| Date: | [________ __], 20[ ] | |
EXHIBIT D-4
TO CREDIT AGREEMENT AND GUARANTY
FORM OF U.S. TAX COMPLIANCE CERTIFICATE
(For Foreign Lenders That Are Partnerships for U.S. Federal Income Tax Purposes)
Reference is made to the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”).
Pursuant to the provisions of Section 5.03(f) of the Credit Agreement, the undersigned hereby certifies that (i) it is the sole record owner of the Loan(s) (as well as any Note(s) evidencing such Loan(s)) in respect of which it is providing this certificate, (ii) its direct or indirect partners/members are the sole beneficial owners of such Loan(s) (as well as any Note(s) evidencing such Loan(s)), (iii) with respect to the extension of credit pursuant to this Credit Agreement orcopyrights obtained, including, without limitation, testifying in any suit or proceeding involving any of said patents or copyrights or executing any documents deemed necessary by the Company, all without further consideration than provided for herein. It is understood that reasonable out-of-pocket expenses of my assistance incurred at the request of the Company under this Section will be reimbursed by the Company. In the event the Company is unable after reasonable effort to obtain my signature on any document which I may be required to sign pursuant to this Agreement, whether because of my physical or mental incapacity or for any other Loan Document, neither the undersigned nor any of its direct or indirect partners/members is a “bank” extending credit pursuant to a loan agreement entered into in the ordinary course of its trade or business within the meaning of Section 881(c)(3)(A) of the Code, (iv) none of its direct or indirect partners/members is a “ten percent shareholder” of the Borrower within the meaning of Section 871(h)(3)(B) of the Code and (v) none of its direct or indirect partners/members is a “controlled foreign corporation” related to the Borrower as described in Section 881(c)(3)(C) of the Code.
The undersigned has furnished the Administrative Agent and the Borrower with an IRS Form W-8IMY (or applicable successor form) accompanied by one of the following forms from each of its partners/members that is claiming the portfolio interest exemption: (i) an IRS Form W-8BEN or IRS Form W-8BEN-E (or applicable successor form) or (ii) an IRS Form W-8IMY (or applicable successor form) accompanied by an IRS Form W-8BEN or IRS Form W-8BEN-E (or applicable successor form) from each of such partner’s/member’s beneficial owners that is claiming the portfolio interest exemption. By executing this certificate, the undersigned agrees that (1) if the information provided in this certificate changes, the undersigned shall promptly so inform the Borrower and the Administrative Agent, and (2) the undersigned shall have at all times furnished the Borrower and the Administrative Agent with a properly completed and currently effective certificate in either the calendar year in which each payment is to be made to the undersigned, or in either of the two calendar years preceding such payments.
Unless otherwise defined herein, terms defined in the Credit Agreement and used herein shall have the meanings given to them in the Credit Agreement.
[Signature Page Follows]
| | | | | | | | |
| [NAME OF LENDER] | |
| | |
| By: | | |
| Name: | |
| Title: | |
| | |
| Date: | [________ __], 20[ ] | |
EXHIBIT E
TO CREDIT AGREEMENT AND GUARANTY
FORM OF COMPLIANCE CERTIFICATE
[DATE]
This certificate is delivered pursuant to Section 8.01(d) of, and in connection with the consummation of the Transactions contemplated in, the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”). Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Credit Agreement.
The undersigned, a duly authorized Responsible Officer of the Borrower having the name and title set forth below under his or her signature, hereby certifies (in his or her capacity as an officer of the Borrower and not in his or her individual capacity), on behalf of the Borrower for the benefit of the Secured Parties, that such Responsible Officer of the Borrower is familiar with the Credit Agreement and that, in accordance with each of the following sectionsreason whatsoever, I hereby irrevocably appoint each of the President and the Secretary of the Credit Agreement, eachCompany (whether now or hereafter in office) as my attorney-in-fact to execute any such document on my behalf.
1.Non-solicitation. I acknowledge and agree that any and all “goodwill” associated with any Customers, Business Partners and employees of the following is true on the date hereof, both before and after giving effect to any Loan to be made on or before the date hereof:
In accordance with Section 8.01[(b)/(c)] of the Credit Agreement, attached hereto as Annex A are the financial statements for the [fiscal quarter/fiscal year] ended [__________] required to be delivered pursuant to Section 8.01[(b)/(c)] of the Credit Agreement. [Such financial statements fairly present in all material respects the financial condition of the Borrower and its Subsidiaries as at the dates indicated therein and the results of operations of the Borrower and its Subsidiaries for the periods indicated therein and have been prepared in accordance with GAAP consistently applied, subject to changes resulting from normal, year-end audit adjustments and except for the absence of footnotes.]3
Attached hereto as Annex B are the calculations used to determine compliance with each financial covenant contained in Section 10 of the Credit Agreement, together with evidence based upon the Borrower’s bank account statements that the Borrower has met its minimum liquidity requirement set forth in Section 10.01 of the Credit Agreement.
No Default or Event of Default is continuing as of the date hereof[, except as provided for on Annex C attached hereto, with respect to each of which the Borrower proposes to take the actions set forth on Annex C].
[Pursuant to Section 8.02(j) of the Credit Agreement, attached hereto as Annex D is a list of all Intellectual Property created, developed or acquired by the Borrower or of its Subsidiaries
3 Insert bracketed language only for certifications pursuant to Section 8.01(b) of the Credit Agreement.
in the fiscal quarter ended [_________], 20[__].]4
[Signature Page Follows]
4 If applicable, insert bracketed language only for certifications pursuant to Sections 8.01(b) and 8.01(c) of the Credit Agreement.
IN WITNESS WHEREOF, the undersigned has executed this certificate on the date first written above.
| | | | | | | | |
| PEAR THERAPEUTICS, INC. |
| | |
| | |
| By | |
| Name: | |
| Title: | |
EXHIBIT F
TO CREDIT AGREEMENT AND GUARANTY
FORM OF ASSIGNMENT AND ASSUMPTION
This Assignment and Assumption (this “Assignment and Assumption”) is dated as of the Effective Date set forth below and is entered into by and between [______________] (the “Assignor”) and [______________] (the “Assignee”). Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”), receipt of a copy of which is hereby acknowledged by the Assignee. The Standard Terms and Conditions set forth in Annex 1 attached hereto are hereby agreed to and incorporated herein by reference and made a part of this Assignment and Assumption as if set forth herein in full.
For an agreed consideration, the Assignor hereby irrevocably sells and assigns to the Assignee, and the Assignee hereby irrevocably purchases and assumes from the Assignor, subject to and in accordance with the Standard Terms and Conditions and the Credit Agreement, as of the Effective Date inserted by the Administrative Agent as contemplated below (i) all of the Assignor’s rights and obligations in its capacity as a Lender under the Credit Agreement and any other documents or instruments delivered pursuant thereto to the extent related to the amount and percentage interest identified below of all of such outstanding rights and obligations of the Assignor under the Credit Agreement and (ii) to the extent permitted to be assigned under applicable law, all claims, suits, causes of action and any other right of the Assignor (in its capacity as a Lender) against any Person, whether known or unknown, arising under or in connection with the Credit Agreement, any other documents or instruments delivered pursuant thereto or the loan transactions governed thereby or in any way based on or related to any of the foregoing, including, but not limited to, contract claims, tort claims, malpractice claims, statutory claims and all other claims at lawCompany belongs exclusively to the Company including, but not limited to, any goodwill created as a result of direct or indirect contacts or relationships between me and any Customers, Business Partners or in equity related to the rights and obligations sold and assigned pursuant to clause (i) above (the rights and obligations soldemployees of the Company. I also acknowledge and agree that my employment with and services for the Company will require the access to and use of Confidential Information. I further acknowledge and agree that the Company makes reasonable efforts to maintain the confidentiality of its Confidential Information, and that it derives independent economic value, actual or potential, from the Confidential Information not being generally known to the public or to others who can obtain economic value from its disclosure. I also acknowledge and agree that the Company would be irreparably damaged if I were to engage in the conduct described below using the Company’s Confidential Information (including but not limited to its trade secrets). Accordingly, I hereby agree that:
a.During my employment with the Company and for a period of twelve (12) months after the termination of my employment with the Company for any reason, I shall not, directly or indirectly, for myself or for any other person or entity, solicit, induce, hire, or attempt to do any of the foregoing, any employee of the Company (or any person who may have and assigned by the Assignor to the Assignee pursuant to clauses (i) and (ii) above being referred to herein collectively as the “been employed by the Company during the last twelve (12) months of my employment with the Company), or assist in such attempted or actual solicitation, inducement or hiring by any other person or entity or encourage any such employee to terminate his or her employment with the Company.
a.I acknowledge that the Company has valuable Trade Secrets (as defined by applicable law from time to time) to which I will have access during the term of my employment with the Company. I understand that the Company intends to vigorously pursue its rights under applicable Trade Secrets law if, during a period of twelve (12) months immediately following the termination of my employment for any reason, whether with or without cause, I solicit or influence or attempt to influence any Customer, Business Partner or other person either directly or indirectly, to direct any purchase of products and/or services to any person, firm, corporation, institution or other entity in competition with the business of the Company. Thereafter, the Company intends to vigorously pursue its rights under applicable Trade Secrets law as the circumstances warrant.
a.Assigned InterestFor Non-California Employees Only: ”). Such saleDuring my employment with the Company and assignment is without recourse to the Assignor and, except as expressly provided in this Assignment and Assumption, without representation or warranty by the Assignor.for a period of twelve (12) months after the termination of my employment with the Company for any reason, I shall not, for myself or for any other person or entity, within the geographic area within which the Company did business at any time during the preceding two
(2) years, directly or indirectly, encourage or induce (or attempt to encourage or induce) any Customer, prospective Customer, Business Partner, or prospective Business Partner of the Company to terminate, cease, or reduce the scope of its relationship with the Company, or to refrain from entering into a business relationship with the Company.
I expressly agree that in the event of my breach of any of the foregoing, the period of the restriction shall be extended by at least an amount of time reflecting the period of time during which I was in violation of any of the provisions set forth in this Section.
1. Assignor[s]: _________________________________________
_________________________________________
2. Assignee[s]: _________________________________________
_________________________________________
[Assignee is an Affiliate of [Return of Propertyidentify Lender]]
. Upon termination of my employment with the Company, or at any other time upon request of the Company, I shall return promptly any and all Customer or prospective Customer or Business Partner or Prospective Business Partner lists, other Customer or prospective Customer or Business Partner or prospective Business Partner information or related materials, computer programs, software, electronic data, specifications, drawings, blueprints, data storage devices, reproductions, sketches, notes, notebooks, memoranda, reports, records, proposals, business plans, or copies of them, other documents or materials, tools, equipment, or other property belonging to the Company or its Customers or Business Partners which I may then possess or have under my control, in any form, and including all copies of the same. I further agree that upon termination of employment I shall not take with me any documents or data in any form or of any description containing or pertaining to Confidential Information or Inventions.
3. Borrower: Pear Therapeutics, Inc.
1.4. Administrative Agent: Perceptive Credit Holdings III, LP
5. Credit Agreement: Credit Agreement and Guaranty, dated as of June 30, 2020, among the Borrower, certain Subsidiaries of the Borrower that may be required to provide GuaranteesOther Obligations. I acknowledge that the Company from time to time thereunder, the lenders from time to time party thereto (the “Lendersmay have agreements with other persons, including other corporations, the government of the United States or other countries and agencies thereof, which impose obligations or restrictions on the Company regarding inventions made during the course of work thereunder or regarding the confidential nature of such work or the confidential nature of information disclosed to the Company by such persons. I agree to be bound by all such obligations and restrictions and to take all action necessary to discharge the obligations of the Company thereunder.
1.Enforcement. I acknowledge and agree that (i) the provisions of Sections 2 and 6 are necessary and reasonable to protect the Company’s Confidential Information (including trade secrets) and goodwill; (ii) the specific time, geography and scope provisions set forth in Section 6 are reasonable and necessary to protect the Company’s business interests; and (iii) violation of this Agreement by me would cause irreparable harm to the Company not adequately compensable by money damages alone. In recognition of the foregoing, I agree that, in addition to all other remedies available to the Company at law, in equity or otherwise, the Company shall be entitled to specific performance, or temporary, preliminary or permanent injunctive relief, or any other equitable remedy which then may be available, to prevent an actual or threatened violation of this Agreement and to enforce the provisions hereof, without showing or proving any actual damage to the Company or posting any bond in connection therewith. The seeking of such injunction or order shall not affect the Company’s right to seek and obtain damages or other equitable relief on account of any such actual or threatened breach. I further agree that the
Company shall be entitled to its costs and fees, including attorneys’ fees, incurred by it should it prevail in enforcing the provisions of this Agreement.
1.Miscellaneous
a.”)This Agreement contains the entire and only agreement between me and the Administrative Agent.
6. Assigned Interest[s]:Company with respect to the subject matter hereof, superseding any previous oral or written communications, representations, understandings, or agreements with the Company or any officer or representative thereof. In the event of any inconsistency between this Agreement and any other contract between me and the Company, the provisions of this Agreement shall prevail.
a.My obligations under this Agreement shall survive the termination of my employment with the Company regardless of the manner of or reasons for such termination, and regardless of whether such termination constitutes a breach of any other agreement I may have with the Company. My obligations under this Agreement also shall continue should I be promoted or reassigned to functions other than my present functions, or should my compensation and benefit package change, or should I be transferred between or among Company offices or any Company affiliates that may exist. My obligations under this Agreement shall be binding upon my heirs, assigns, executors, administrators and representatives, and the provisions of this Agreement shall inure to the benefit of and be binding on the successors and assigns of the Company, and the Company shall have the right to assign this Agreement to another party, whether by agreement or by operation of law, and whether resulting from a merger, sale (of assets or securities or otherwise), or any other transaction or event.
a.If any provision of this Agreement shall be determined to be unenforceable by any court of competent jurisdiction by reason of its extending for too great a period of time or over too large a geographic area or over too great a range of activities, it shall be interpreted to extend only over the maximum period of time, geographic area or range of activities as to which it may be enforceable. If, after application of the immediately preceding sentence, any provision of this Agreement shall be determined to be invalid, illegal or otherwise unenforceable by any court of competent jurisdiction, the validity, legality and enforceability of the other provisions of this Agreement shall not be affected thereby. Except as otherwise provided in this paragraph, any invalid, illegal or unenforceable provision of this Agreement shall be severable, and after any such severance, all other provisions hereof shall remain in full force and effect.
a.No failure by the Company to insist upon strict compliance with any of the terms, covenants, or conditions hereof, and no delay or omission by the Company in exercising any right under this Agreement, will operate as a waiver of such terms, covenants, conditions or rights. A waiver or
consent given by the Company on any one occasion is effective only in that instance and will not be construed as a bar to or waiver of any right on any other occasion.
a.This Agreement may not be changed, modified, released, discharged, abandoned, or otherwise amended, in whole or in part, except by an instrument in writing signed by me and the Company.
a.This Agreement shall be governed by, and construed and enforced in accordance with, the laws of state in which I reside and primarily provide services to the Company, without regard to the principles of conflicts of laws of such state. This Agreement is executed under seal.
BY PLACING MY SIGNATURE HEREUNDER, I ACKNOWLEDGE AND AGREE THAT I HAVE READ ALL THE PROVISIONS OF THIS EMPLOYEE NON-DISCLOSURE, NON- SOLICITATION AND ASSIGNMENT OF INTELLECTUAL PROPERTY AGREEMENT, THAT I HAVE HAD ADEQUATE OPPORTUNITY TO REVIEW THE TERMS HEREIN AND REFLECT UPON AND CONSIDER THE SAME, AND TO CONSULT A LAWYER REGARDING THE SAME, AND THAT I FULLY UNDERSTAND AND VOLUNTARILY AGREE TO ALL OF ITS TERMS.
Effective Date is Start Date: June 10, 2019
EMPLOYEE:
/s/ Katherine Jeffery
Employee's Signature
Name: Katherine Jeffery
Address:
[***]
Accepted and Agreed:
PEAR THERAPEUTICS, INC.
By: /s/ Christopher Guiffre
Title: CFO & COO
EXHIBIT A
Excluded Confidential Information and Inventions
| | | | | | | | | | | | | | |
| Title | | Date | | Identifying Number or Brief Description |
| Assignor[s]5n/a | | Assignee[s]6n/a | Type of Commitment/Loans Assigned | Aggregate Amount of Commitment/Loans for all Lenders | Amount of Commitment/Loans Assigned8 | Percentage Assigned of Commitment/
Loans7 |
| | [Tranche 1 Loan][Tranche 2 Loan][Tranche 3 Loan] | $ | $ | % |
| | [Tranche 1 Loan][Tranche 2 Loan][Tranche 3 Loan] | $ | $ | % |
| | [Tranche 1 Loan][Tranche 2 Loan][Tranche 3 Loan] | $ | $ | % |
[Signature Page Follows]
5 List each Assignor, as appropriate.
6 List each Assignee, as appropriate.
7 Set forth, to at least 9 decimals, as a percentage of the Commitment/Loans of all Lenders thereunder.
Effective Date: _____________ ___, 20___ [TO BE INSERTED BY ADMINISTRATIVE AGENT AND WHICH SHALL BE THE EFFECTIVE DATE OF RECORDATION OF TRANSFER IN THE REGISTER THEREFOR.]
The terms set forth in this Assignment and Assumption are hereby agreed to:
| | | | | | | | | | | |
| | ASSIGNOR |
| | | |
| | [NAME OF ASSIGNOR] |
| | | |
| | | |
| | By: | |
| | Name: | |
| | Title: | |
| | | |
| | | |
| | ASSIGNEE |
| | | |
| | [NAME OF ASSIGNEE] |
| | | |
| | | |
| | By: | |
| | Name: | |
| | Title: | |
| | | |
| | | |
| Consented to and Accepted: | | |
| | | |
PERCEPTIVE CREDIT HOLDINGS III, LP,
as Administrative Agent | | |
| | | |
| By: | PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner |
| | | |
| By: | | | |
| Name: | | | |
| Title: | | | |
| | | |
| | | |
| By: | | | |
| Name: | | | |
| Title: | | | |
ANNEX 1
STANDARD TERMS AND CONDITIONS
1. Representations and Warranties.
1.1 Assignor. The Assignor (a) represents and warrants that (i) it is the legal and beneficial owner of the Assigned Interest, (ii) the Assigned Interest is free and clear of any lien, encumbrance or other adverse claim and (iii) it has full power and authority, and has taken all action necessary, to execute and deliver this Assignment and Assumption and to consummate the transactions contemplated hereby; and (b) assumes no responsibility with respect to (i) any statements, warranties or representations made in or in connection with the Credit Agreement or any other Loan Document, (ii) the execution, legality, validity, enforceability, genuineness, sufficiency or value of the Loan Documents or any collateral thereunder, (iii) the financial condition of the Borrower, any of its Subsidiaries or Affiliates or any other Person obligated in respect of any Loan Document or (iv) the performance or observance by the Borrower, any of its Subsidiaries or Affiliates or other Person of any of their respective obligations under any Loan Document.
1.2 Assignee. The Assignee (a) represents and warrants that (i) it has full power and authority, and has taken all action necessary, to execute and deliver this Assignment and Assumption and to consummate the transactions contemplated hereby and to become a Lender under the Credit Agreement, (ii) it meets all requirements of an Eligible Transferee under the Credit Agreement, (iii) from and after the Effective Date, it shall be bound by the provisions of the Credit Agreement as a Lender thereunder and, to the extent of the Assigned Interest, shall have the obligations of a Lender thereunder, (iv) it is sophisticated with respect to decisions to acquire assets of the type represented by the Assigned Interest and is experienced in acquiring assets of such type, (v) it has received a copy of the Credit Agreement, and has received or has been accorded the opportunity to receive copies of the most recent financial statements delivered pursuant to Section 8.01(a), (b) and (c) thereof, as applicable, and such other documents and information as it has deemed appropriate to make its own credit analysis and decision to enter into this Assignment and Assumption and to purchase the Assigned Interest, (vi) it has, independently and without reliance upon the Administrative Agent or any other Lender and based on such documents and information as it has deemed appropriate, made its own credit analysis and decision to enter into this Assignment and Assumption and to purchase the Assigned Interest, and (vii) attached to the Assignment and Assumption is any documentation required to be delivered by it pursuant to the terms of the Credit Agreement, duly completed and executed by the Assignee; and (b) agrees that (i) it will, independently and without reliance on the Administrative Agent, the Assignor or any other Lender, and based on such documents and information as it shall deem appropriate at the time, continue to make its own credit decisions in taking or not taking action under the Loan Documents, and (ii) it will perform in accordance with their terms all of the obligations which by the terms of the Loan Documents are required to be performed by it as a Lender.
2. Payments. From and after the Effective Date, the Administrative Agent shall make all payments in respect of the Assigned Interest (including payments of principal, interest, fees and other amounts) to the Assignor for amounts which have accrued to but excluding the Effective
Date and to the Assignee for amounts which have accrued from and after the Effective Date. Notwithstanding the foregoing, the Administrative Agent shall make all payments of interest, fees or other amounts paid or payable in kind from and after the Effective Date to the Assignee.
3. General Provisions. This Assignment and Assumption shall be binding upon, and inure to the benefit of, the parties hereto and their respective successors and assigns. This Assignment and Assumption may be executed in any number of counterparts, which together shall constitute one instrument. Delivery of an executed counterpart of a signature page of this Assignment and Assumption by facsimile or in electronic (PDF) format shall be effective as delivery of a manually executed counterpart of this Assignment and Assumption.
4. THIS ASSIGNMENT AND ASSUMPTION SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH, THE LAW OF THE STATE OF NEW YORK, WITHOUT REGARD TO PRINCIPLES OF CONFLICTS OF LAWS THAT WOULD RESULT IN THE APPLICATION OF THE LAWS OF ANY OTHER JURISDICTION; PROVIDED THAT SECTION 5-1401 OF THE NEW YORK GENERAL OBLIGATIONS LAW SHALL APPLY.
EXHIBIT G
TO CREDIT AGREEMENT AND GUARANTY
FORM OF INFORMATION CERTIFICATE
(See attached.)
EXHIBIT H
TO CREDIT AGREEMENT AND GUARANTY
FORM OF INTERCOMPANY SUBORDINATION AGREEMENT
This INTERCOMPANY SUBORDINATION AGREEMENT, dated as of [_________], 20[__] (this “Subordination Agreement”), is entered into by and among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that are parties hereto, and certain other Subsidiaries of the Borrower that may, from time to time in the future, become parties hereto by executing and delivering a joinder agreement in substantially the form of Exhibit A hereto (any such Subsidiary being herein, individually, a “Subsidiary Party” and collectively the “Subsidiary Parties”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, in its capacity as Administrative Agent for the Lenders under the Credit Agreement (as defined below) (in such capacity, together with its successors and assigns, the “Administrative Agent”).
Reference is made to that certain Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”) among the Borrower, certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the Lenders from time to time party thereto and the Administrative Agent. Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Credit Agreement.
One or more of the Borrower and the Subsidiary Parties (individually, a “Pear Party” and collectively, the “Pear Parties”), in their capacities as lenders (each such entity, together with its successors, assigns and transferees in such capacity, individually, a “Junior Creditor”, and, collectively, “Junior Creditors”) has made, or may from time to time may make, loans or extend other financings to one or more of the Pear Parties that is an Obligor (each such Obligor, in its capacity as a borrower from any Junior Creditor (together with its successors, assigns and transferees) being herein, individually, a “Debtor Obligor”, and, collectively, “Debtor Obligors”) pursuant to Section 9.01(e) of the Credit Agreement. All such Indebtedness resulting from the making of any such loan or financing, together with all principal, interest, fees, premiums, costs, expenses, liabilities, obligations and other amounts of any type or nature at any time owing or arising in respect thereof, including but not limited to any such items or amounts as may accrue or be incurred before or after default or workout or the commencement of any liquidation, dissolution, bankruptcy, receivership or reorganization case by or against the Borrower, is herein collectively referred to as the “Junior Obligations”.
Each of the Junior Creditors and each of the Debtor Obligors, for the benefit of the Secured Parties and each of their permitted successors, transferees and assigns, hereby irrevocably and unconditionally agree as follows:
1. All payment obligations and other monetary obligations of any Debtor Obligor arising from time to time under or in connection with any Junior Obligations to any Junior Creditor are, and shall at all times be, subordinated in right of payment and performance to the prior Payment in Full (as defined below) of all Obligations owing under or in
connection with the Credit Agreement and the other Loan Documents, whether in respect of principal, interest, fees or other monetary obligations or liabilities of any type or nature, including costs and expenses of enforcement, if any (collectively, the “Senior Obligations”), notwithstanding the maturity date or amortization date of any Junior Obligations or any acceleration of the maturity date related thereto, any default by or insolvency of any Junior Creditor or any other Person, or otherwise.
2. The parties hereto agree that this Subordination Agreement is for the benefit of, and shall be enforceable by the Administrative Agent on behalf of the Secured Parties.
3. At all times from and after the date of this Subordination Agreement until Payment in Full of all Senior Obligations and the termination of all Senior Liens (as defined below), (i) no Debtor Obligor shall make, and no Junior Creditor shall accept, receive or collect from or on behalf of any Debtor Obligor, any direct or indirect payment or distribution of any kind or character whatsoever (whether in cash, securities, other property, by set-off, forgiveness of any Indebtedness of any Secured Party, or otherwise) on account of any of the Junior Obligations, and (ii) under no circumstance shall any payment of any of the Junior Obligations be accelerated, or any other remedy, enforcement action or other action be taken by any Junior Creditor against any Debtor Obligor or any property of any Debtor Obligor or of any other Person, in each case with respect to any of the Junior Obligations (including to assert, enforce or collect any of the Junior Obligations), in each case, except to the extent permitted by the Credit Agreement or with the prior written consent of the Administrative Agent, which consent may not be unreasonably withheld.
4. No Junior Creditor shall, directly or indirectly, independently or with any other Person, take any action that would be in violation of, or inconsistent with, or result in a breach of this Subordination Agreement or challenge or contest (i) the validity, perfection, priority or enforceability of this Subordination Agreement, any Senior Obligations or any Liens securing the Senior Obligations (“Senior Liens”), (ii) any of the rights of any Secured Party set forth in the Credit Agreement or any other Loan Document (including with respect to the Senior Liens), or (iii) the validity or enforceability of the Credit Agreement or any other Loan Document or any portion thereof.
5. In the event that, prior to Payment in Full of the Senior Obligations, any Junior Creditor shall receive any payment or distribution of any kind or character whatsoever (whether in cash, securities, other property, by set-off, forgiveness of any Indebtedness of any Secured Party, or otherwise) on or in respect of all or any portion of the Junior Obligations in violation of any of the provisions of this Subordination Agreement, then such payment or distribution shall be held in trust by such Junior Creditor for the benefit of, and promptly (and in any event within one (1) Business Day) paid over by such Junior Creditor to the Administrative Agent for application of such payment or distribution to repay the Obligations in accordance with the terms thereof, until Payment in Full of the Obligations as confirmed in writing by the Administrative Agent to the Borrower.
6. For purposes of this Subordination Agreement, “Payment in Full” means, with respect to the Obligations, that all such obligations and other amounts payable constituting Obligations have been indefeasibly paid in full in cash.
7. Neither any Junior Creditor nor any Debtor Obligor may assign or transfer any of its rights or obligations hereunder, except to another Obligor that becomes bound by the terms of this Subordination Agreement.
8. This Subordination Agreement is a Loan Document executed pursuant to the Credit Agreement and shall (unless otherwise expressly indicated therein) be construed, administered and applied in accordance with all of the terms and provisions of the Credit Agreement, as amended hereby, including Section 14 thereof. The provisions of this Subordination Agreement shall be binding upon and inure to the benefit of the parties hereto and their respective permitted successors and assigns.
9. This Subordination Agreement may be executed in any number of counterparts, all of which taken together shall constitute one and the same instrument and any of the parties hereto may execute this Subordination Agreement by signing any such counterpart.
10. This Subordination Agreement and the rights and obligations of the parties hereunder shall be governed by, and construed in accordance with, the law of the State of New York, without regard to principles of conflicts of laws that would result in the application of the laws of any other jurisdiction; provided that Section 5-1401 of the New York General Obligations Law shall apply.
11. Any Subsidiary of the Borrower may, without the consent of any other party to Subordination Agreement, become an Pear Party under this Subordination Agreement by executing and delivering to the Administrative Agent a joinder agreement in substantially the form of Exhibit A hereto.
12. Except as modified in accordance with Section 11 to add any Subsidiary of the Borrower as an additional Pear Party to this Subordination Agreement, this Subordination Agreement may not be amended, waived or otherwise modified without the prior written consent of each of the parties hereto.
[SIGNATURE PAGE FOLLOWS]
IN WITNESS WHEREOF, the parties have caused this Subordination Agreement to be duly executed and delivered as of the date first above written.
| | | | | |
| | | | | |
| x | | No inventions or improvements |
| PEAR THERAPEUTICS, INC. |
| | |
| | |
| By: | |
| Name: | |
| Title: | |
Exhibit H-4Additional Sheets Attached
| | | | | | | | |
| [SUBSIDIARY PARTY] |
| | |
| | |
| By: | |
| Name: | |
| Title: | |
| | | | | | | | |
|
PERCEPTIVE CREDIT HOLDINGS III, LP,Signature: as the Administrative Agent |
| | |
| /s/ Katherine Jeffery Dated: 02/21/2023 By Perceptive Credit Opportunities GP, LLC, its general partner |
| | |
| | |
| By: | |
| Name: | |
| Title: | |
| | |
| | |
| By: | |
| Name: | |
| Title: | |
Exhibit A
FORM OF INTERCOMPANY SUBORDINATION AGREEMENT JOINDER
This INTERCOMPANY SUBORDINATION AGREEMENT JOINDER, dated as of [DATE] by [NAME OF ADDITIONAL SUBSIDIARY], a [state of organization] [corporation][limited liability company] (the “Additional Pear Party”), under that certain Intercompany Subordination Agreement, dated as of [__________], 20[__] (as amended or otherwise modified from time to time, the “Subordination Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower from time to time party thereto and Perceptive Credit Holdings III, LP, a Delaware limited partnership, in its capacity as Administrative Agent for the Lenders under the Credit Agreement (in such capacity, together with its successors and assigns, the “Administrative Agent”). Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Subordination Agreement.
Pursuant to Section 11 of the Subordination Agreement, the Additional Pear Party hereby agrees to become a “Pear Party” for all purposes of the Subordination Agreement.
IN WITNESS WHEREOF, the Additional Pear Party has caused this Subordination Agreement Joinder to be duly executed and delivered as of the day and year first above written.
| | | | | | | | |
| [ADDITIONAL PEAR PARTY] |
| | |
| By | |
| Name: | |
| Title: | |
EXHIBIT I
TO CREDIT AGREEMENT AND GUARANTY
FORM OF SOLVENCY CERTIFICATE
[DATE]
This Solvency Certificate (this “Certificate”) is delivered pursuant to Section 6.01(j) of that certain Credit Agreement and Guaranty, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among Pear Therapeutics, Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of the Borrower that may be required to provide Guarantees from time to time thereunder, the lenders from time to time party thereto (the “Lenders”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, as administrative agent for the Lenders (in such capacity, together with its successors and assigns, the “Administrative Agent”). Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Credit Agreement.
The undersigned hereby certifies on behalf of the Borrower (in his or her capacity as an officer of the Borrower and not in his or her individual capacity) as of the date hereof as follows:
1. I am the chief [financial][accounting] Responsible Officer of the Borrower and am duly authorized to execute and deliver this Certificate on behalf of the Borrower. I have been employed in positions involving responsibility for the management of the financial affairs of the Borrower. I have, together with other officers of the Borrower, acted on behalf of the Borrower in connection with the transactions contemplated by the Credit Agreement and the other Loan Documents.
2. I have carefully reviewed the contents of this Certificate and have conferred with legal counsel for the Borrower for the purpose of discussing the meaning of its contents.
3. In connection with preparing for the transactions contemplated by the Credit Agreement and the other Loan Documents, I have reviewed audited consolidated financial statements of the Borrower and its Subsidiaries for the fiscal year ended December 31, 2019, and the unaudited consolidated balance sheets of the Borrower and its Subsidiaries for each fiscal quarter ended after December 31, 2019 together with the related consolidated statement of operations, shareholder’s equity and cash flows for such fiscal quarter (the information set forth in this paragraph being the “Financial Statements”).
4. The Financial Statements fairly present in all material respects the consolidated financial condition of the Borrower and its Subsidiaries as of such dates and for such periods covered by the Financial Statements. The assumptions stated in the Financial Statements were prepared in good faith based on assumptions believed to be reasonable.
The undersigned has concluded, in good faith and to the best of his or her knowledge and belief, that as of the date hereof and after giving effect to all the transactions contemplated by the Credit Agreement and the other Loan Documents and the incurrence of any other Indebtedness contemplated thereunder, and after giving effect to all rights of indemnity, obligation and contribution, as follows:
Katherine Jeffery
EXHIBIT C
(i) The Borrower and its Subsidiaries taken as a whole, are, and immediately after giving effect to the Borrowing being made on the date hereof and the use of proceeds thereof, will be, Solvent.
(ii) No transfer of property is being made by the Borrower or any of its Subsidiaries and no obligation is being incurred by the Borrower or any of its Subsidiaries in connection with the transactions contemplated by the Credit Agreement or the other Loan Documents with the intent to hinder, delay, or defraud either present or future creditors of the Borrower or any of its Subsidiaries.
The undersigned understands that the Administrative Agent and the Lenders are relying on the truth and accuracy of this Certificate and that the delivery of this Certificate is a material inducement for the Administrative Agent and the Lenders to enter into the Credit Agreement and consummate the transactions contemplated thereby, and the undersigned hereby consents to such reliance.
[Signature Page Follows]
IN WITNESS WHEREOF, the undersigned has executed this certificate on the date first written above.
| | | | | | | | | | | |
| PEAR THERAPEUTICS, INC. |
| | | |
| | | |
| By | |
| | Name: | |
| | Title: | |
EXHIBIT J
TO CREDIT AGREEMENT AND GUARANTY
FORM OF WARRANT CERTIFICATE
(See attached.)
FORM OF WARRANT CERTIFICATE
THIS WARRANT CERTIFICATE AND THE SECURITIES ISSUABLE UPON EXERCISE OF THIS WARRANT CERTIFICATE HAVE NOT BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR QUALIFIED UNDER ANY STATE OR FOREIGN SECURITIES LAWS AND MAY NOT BE OFFERED FOR SALE, SOLD, PLEDGED, HYPOTHECATED OR OTHERWISE TRANSFERRED OR ASSIGNED UNLESS (I) A REGISTRATION STATEMENT COVERING SUCH SHARES IS EFFECTIVE UNDER THE SECURITIES ACT AND IS QUALIFIED UNDER APPLICABLE STATE AND FOREIGN LAW OR (II) THE TRANSACTION IS EXEMPT FROM THE REGISTRATION AND PROSPECTUS DELIVERY REQUIREMENTS UNDER THE SECURITIES ACT AND THE QUALIFICATION REQUIREMENTS UNDER APPLICABLE STATE AND FOREIGN LAW AND, IN EACH CASE, IF THE COMPANY REQUESTS, AN OPINION SATISFACTORY TO THE COMPANY TO SUCH EFFECT HAS BEEN RENDERED BY COUNSEL.
Warrant Shares Issuable: [ ]
Warrant Certificate No.: [ ]
Issue Date: [ ], 2020 (the “Issue Date”)
FOR VALUE RECEIVED, PEAR THERAPEUTICS, INC., a Delaware corporation (the “Company”), hereby certifies that PERCEPTIVE CREDIT HOLDINGS III, LP, a Delaware limited partnership (the “Initial Holder” and, together with its successors and permitted transferees and assigns, a “Holder”) is entitled to purchase, at the per share Exercise Price, up to [ ] ([ ]) fully paid and nonassessable shares of the Company’s Series C Preferred Stock all subject to the terms, conditions and adjustments set forth below in this Warrant Certificate. Capitalized terms used herein have the meanings ascribed thereto in Section 1 below.
This Warrant Certificate has been issued as a condition precedent to the making of the [Tranche 1 Loan] [Tranche 2 Loan] [Tranche 3 Loan] under and pursuant to of the Credit and Guaranty Agreement, dated as of June 30, 2020 (as amended or otherwise modified from time to time, the “Credit Agreement”), among the Company, as borrower, certain Subsidiaries of the Company from time to time party thereto, as guarantors, the lenders from time to time party thereto, and Perceptive Credit Holdings III, LP, as the administrative agent for the lenders.
Section 1. Definitions. Capitalized terms used in this Warrant Certificate but not otherwise defined herein will have the meanings ascribed thereto in the Credit Agreement as in effect on the Issue Date. The following terms when used herein have the following meanings:
“Advanced Development” means, with respect to a proposed Product, that such proposed Product is in either a pivotal clinical trial or later stage of development.
“Aggregate Exercise Price” means, with respect to any exercise of this Warrant Certificate for Warrant Shares pursuant to Section 3, an amount equal to the product of (i) the number of Warrant Shares in respect of which this Warrant Certificate is then being exercised pursuant to Section 3, multiplied by (ii) the Exercise Price.
“Assignment” has the meaning set forth in Section 7.
“Bloomberg” has the meaning set forth within the definition of “VWAP”.
“Cashless Exercise” has the meaning set forth in Section 3(b).
“Certificate of Incorporation” means the Fourth Amended and Restated Certificate of Incorporation of the Company, filed with the Secretary of State of Delaware on December 21, 2018, as amended to date and as further amended, amended or restated, or subsequently modified.
“Common Stock” means the Company’s common stock, par value $0.0001 per share, having ordinary voting rights, as provided in the Company’s Certificate of Incorporation.
“Company” has the meaning set forth in the preamble.
“Convertible Securities” means any Equity Interests that, directly or indirectly, are convertible into or exchangeable for Series C Preferred Stock or Common Stock.
“Credit Agreement” has the meaning set forth in the preamble. “Determination Date” has the meaning set forth in the definition of “VWAP”.
“Device” means any medical instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent or other similar or related item, including any component, part or accessory, that (i) is intended for use in connection with the diagnosis of disease, malady or other conditions or in the cure, mitigation, treatment or prevention of disease or malady, in man or other animals, or is intended to affect the structure or any function of the body of man or other animals, (ii) does not achieve its primary intended purpose or purposes through chemical action within or on the body of man or other animals and (iii) is not dependent upon being metabolized for the achievement of its primary intended purpose or purposes.
“Equity Interests” means, with respect to any Person (for purposes of this defined term, an “issuer”), all shares of, interests or participations in, or other equivalents in respect of such issuer’s capital stock, including all membership interests, partnership interests or equivalent, and all debt or other securities (including warrants, options and similar rights) directly or indirectly exchangeable, exercisable or otherwise convertible into, such issuer’s capital stock, whether now outstanding or issued after the Issue Date, and in each case, however classified or designated and whether voting or non-voting.
“Exchange Act” means the Securities Exchange Act of 1934, as amended.
“Excluded Securities” means any issuance or sale (whether an original issuance or a sale from treasury) by the Company after the Issue Date of: (i) equity securities upon the exercise of this Warrant Certificate; (ii) equity securities (as such number of shares is equitably adjusted for subsequent stock splits, stock combinations, stock dividends and recapitalizations) issued directly or upon the exercise of Options to directors, officers, employees, or consultants of the Company
in connection with their service as directors or officers of the Company, their employment by the Company or their retention as consultants by the Company, in each case authorized by the Board and issued pursuant to a stock incentive plan (including all such equity securities outstanding prior to the Issue Date); (iii) equity securities issued upon the conversion or exercise of Options (other than Options covered by clause (ii) above) or Convertible Securities issued prior to the Issue Date, provided that such securities are not amended after the date hereof to increase the number of shares of equity securities issuable thereunder or to lower the exercise or conversion price thereof; (iv) equity securities issued (a) to Persons in connection with a joint venture, strategic alliance or other commercial relationship with such Person (including Persons that are customers, suppliers and strategic partners of the Company) relating to Product Commercialization and Development Activities and not for the primary purpose of raising equity capital, or (b) in connection with a transaction in which the Company, directly or indirectly, acquires another business substantially in its entirely, in each case where such issuance and transaction has been unanimously approved by the Board; (v) equity securities offered and sold pursuant to a Public Offering; or (vi) equity securities issued to the lessor or vendor in any office lease or equipment lease or similar equipment financing transaction in which the Company obtains the use of such office space or equipment for its business.
“Exercise Certificate” has the meaning set forth in Section 3(a)(i).
“Exercise Documents” has the meaning set forth in Section 3(a)(i).
“Exercise Date” means, for any given exercise of this Warrant Certificate, whether in whole or in part, a Business Day on which the conditions to such exercise as set forth in Section 3 have been satisfied at or prior to 5:00 p.m., New York City time.
“Exercise Period” means the period from (and including) the Issue Date to (and including) 5:00 p.m., New York City time, on June 30, 2030.
“Exercise Price” means 7.1935 per Warrant Share, as adjusted from time to time as provided herein.
“Expiration Date” means June 30, 2030.
“Fair Market Value” means, if the Warrant Shares are traded on a Trading Market, (i) the VWAP of such Warrant Shares for such day or (ii) if there have been no sales of such Warrant Shares on any Trading Market on any such day, the average of the highest bid and lowest asked prices for such Warrant Shares on all applicable Trading Markets at the end of such day; provided that if at any time the Warrant Shares are not listed, quoted or otherwise available for trading on any Trading Market (so that no Trading Date shall have occurred), the “Fair Market Value” of such Warrant Shares shall be the fair market value per share of such Warrant Shares (including, if applicable, any Common Stock or other Equity Interests issuable upon conversion of the Warrant Shares) as reasonably determined in good faith by the Company’s Board, such determination to be subject to Section 10(a) or Section 10(b), as applicable.
“Holder” has the meaning set forth in the preamble.
“Independent Advisor” has the meaning set forth in Section 10(a). “Initial Holder” has the meaning set forth in the preamble.
“Investors’ Rights Agreement” means that certain Second Amended and Restated Investors’ Rights Agreement, dated December 21, 2018, among the Company, certain holders of the Company’s Common Shares and certain holders of the preferred shares, as amended, amended and restated or subsequently modified.
“Issue Date” means the date designated as such on the first page of this Warrant Certificate.
“Marketable Securities” means Equity Interests meeting each of the following requirements: (i) the issuer thereof is subject to the reporting requirements of Section 13 or Section 15(d) of the Exchange Act, and is current in its filing of all required reports and other information under the Securities Act and the Exchange Act; (ii) such Equity Interests are traded on a Trading Market; and (iii) if delivered (or to be delivered) as payment or compensation to the Holder in connection with an automatic Cashless Exercise resulting from a Sale of the Company pursuant to Section 3(c), following the closing of the such Sale of the Company the Holder would not be restricted from publicly re-selling any or all of such Equity Interests delivered to it, except to the extent that any such restriction (x) arises solely under federal or state securities laws, rules or regulations, or (y) arising in connection with a customary market stand-off provision (or similar underwriters’ lock-up) and does not extend beyond six (6) months from the closing of such Sale of the Company to the extent such restrictions may be lifted at such time under the applicable federal or state securities laws, rules or regulations.
“Nasdaq” means The Nasdaq Stock Market, Inc.
“NYSE” means the New York Stock Exchange.
“Options” means any warrants, options or similar rights to subscribe for or purchase Equity Interests of the Company, including its Common Stock, Series C Preferred Stock or Convertible Securities.
“OTC Bulletin Board” means the National Association of Securities Dealers, Inc. OTC Bulletin Board.
“Person” means any individual, corporation, company, voluntary association, partnership, limited liability company, joint venture, trust, unincorporated organization or Governmental Authority or other entity of whatever nature.
“Product” means (i) those products and Devices set forth on Schedule 7.05(b) attached to the Credit Agreement, as amended, updated or otherwise modified from time to time pursuant to the terms thereof, and (ii) any other products or Devices developed, distributed, imported, exported, labeled, promoted, licensed, marketed, sold or otherwise commercialized by the Borrower or any of its Subsidiaries, but excluding any such products or Devices that, if in development or which may be developed, have not yet reached the stage of Advanced Development.
“Product Commercialization and Development Activities” means, with respect to any Product, any combination of (i) research, development, manufacturing, quality compliance, use, sale, licensing, importation, exportation, shipping, storage, handling, designing, labeling, marketing, promotion, supply, dispensing, distribution, testing, packaging, purchasing or other commercialization activity, (ii) receipt of payment or other remuneration in respect of any of the foregoing (including, without limitation, in respect of licensing, royalty or similar payments) or (iii) any similar or other activities the purpose of which is to commercially exploit such Product.
“Public Offering” means, with respect to any Person, any sale of Equity Interests of such Person pursuant to an offering that is underwritten on a firm commitment basis by a nationally recognized investment banking firm (or through the merger of such Person with a special purpose acquisition company) and, as a result of which, such Person becomes subject to the reporting requirements of Section 13 or Section 15 of the Exchange Act immediately following such offering.
“Qualified IPO” means an initial Public Offering by the Company of Equity Interests having ordinary voting rights that results in (i) such Equity Interests being listed on either the New York Stock Exchange or the NASDAQ National Market and (ii) the receipt by the Company of gross proceeds from such offering that equal or exceed $50,000,000.
“Registration Statement” means, with respect to any Public Offering by the Company of its Common Stock, a registration statement of the Company that covers such Common Stock, including any prospectus, amendments or supplements to such Registration Statement, including post-effective amendments and all exhibits and all materials incorporated by reference in such Registration Statement.
“Right of First Refusal and Co-Sale Agreement” means that certain Second Amended and Restated Right of First Refusal and Co-Sale Agreement, dated December 21, 2018, among the Company, certain holders of the Company’s Common Shares and certain holders of the preferred shares, as amended, amended and restated, or subsequently modified.
“Rule 144” means Rule 144 promulgated under the Securities Act.
“Sale of the Company” means an event or transaction or series of related events or transactions pursuant to which, directly or indirectly, either (x) any Person or group of Persons acting jointly or otherwise in concert (other than the Holder) acquires ownership, directly or indirectly, beneficially or of record, of Equity Interests of the Company having more than fifty percent (50%) of the aggregate ordinary voting power, determined on a fully-diluted, as-if-converted or exercised basis, whether such right is exercisable immediately or only after the passage of time, or (y) (i) all or substantially all of the assets or businesses of the Company and its Subsidiaries, taken as a whole, are transferred or sold, including by way of lease, transfer, conveyance or other disposition (including, without limitation, by way of irrevocable, exclusive license arrangements), and (ii) as a result of, and pursuant to the terms governing, such event, transaction or series of related events or transactions, all Obligations outstanding under the Credit Agreement are to be paid in full in cash, Marketable Securities or a combination thereof.
“SEC” means the Securities and Exchange Commission or any successor thereto.
“Securities Act” means the Securities Act of 1933, as amended.
“Series C Preferred Stock” means, as designated and defined in the Certificate of Incorporation, the Company’s Series C Preferred Stock having a par value $0.0001 per share.
“Stockholders’ Agreements” means the Investors’ Rights Agreement, Voting Agreement and Right of First Refusal and Co-Sale Agreement.
“Trading Market” means, with respect to the Warrant Shares, the principal US exchange or market on which such Warrant Shares are quoted or available for trading, including the Nasdaq, the NYSE, the OTC Bulletin Board or otherwise.
“Unrestricted Conditions” has the meaning set forth in Section 11(a)(ii).
“Voting Agreement” means that certain Second Amended and Restated Voting Agreement, dated December 21, 2018, among the Company, certain holders of the Company’s Common Shares and certain holders of the preferred shares, as amended, amended and restated, or subsequently modified.
“VWAP” means, with respect to any Warrant Shares, as of any day of determination (a “Determination Date”), the volume weighted average sale price for the period of ten (10) consecutive trading days immediately preceding such Determination Date on the Trading Market for such Warrant Shares as reported by, or based upon data reported by, Bloomberg Financial Markets or an equivalent, reliable reporting service reasonably acceptable to the Holder and the Company (collectively, “Bloomberg”) or, if the volume weighted average sale price has not been reported for such security by Bloomberg for such ten (10) day period, then the last closing trade price of such security as reported by Bloomberg, or, if no last closing trade price is reported for such security by Bloomberg, the average of the bid prices of any market makers for such security that are listed in the over the counter market by the Financial Industry Regulatory Authority, Inc. or on the OTC Bulletin Board (or any successor) or in the “pink sheets” (or any successor) by the OTC Markets Group, Inc.; provided that if VWAP cannot be calculated for such security on such date in the manner provided above (including because the applicable security is not listed or publicly traded), the VWAP shall be the Fair Market Value as mutually determined by the Board and the Holder; provided further that, in the event the Board and Holder are unable to so mutually agree, Fair Market Value shall be determined pursuant to Section 10(a).
“Warrant Certificate” means this Warrant Certificate and all subsequent warrant certificates issued upon division, combination or transfer of, or in substitution for, this Warrant Certificate.
“Warrant Register” has the meaning set forth in Section 6.
“Warrant Shares” means the shares of Series C Preferred Stock purchasable upon exercise of this Warrant Certificate in accordance with the terms of this Warrant Certificate and any capital stock into which such Series C Preferred Stock shall have been converted, exchanged or reclassified following the Issue Date.
“Warrant Shares Deemed Outstanding” means, at any given time, the sum of (i) the number of shares of Warrant Shares actually outstanding at such time, plus (ii) the number of shares of Warrant Shares issuable upon exercise of Options actually outstanding at such time, plus (iii) the number of shares of Warrant Shares issuable upon conversion or exchange of Convertible Securities actually outstanding at such time (treating as actually outstanding any Convertible Securities issuable upon exercise of Options actually outstanding at such time), in each case, regardless of whether the Options or Convertible Securities are actually exercisable at such time; provided that Warrant Shares Deemed Outstanding at any given time shall not include shares owned or held by or for the account of the Company or any or its wholly owned Subsidiaries.
Section 2. Term of Warrant Certificate. Subject to the terms and conditions hereof, from
time to time during the Exercise Period, the Holder of this Warrant Certificate may exercise this Warrant Certificate for all or any part of the Warrant Shares purchasable hereunder (subject to adjustment as provided herein).
Section 3. Exercise of Warrant Certificate.
(a) Exercise Procedure. This Warrant Certificate may be exercised from time to time on any Business Day during the Exercise Period, for all or any part of the unexercised Warrant Shares, upon:
(i) delivery to the Company at its then principle executive office of a duly completed and executed Exercise Certificate in the form attached hereto as Exhibit A (each, an “Exercise Certificate”), which certificate will specify the number of Warrant Shares to be purchased and the Aggregate Exercise Price, together with this Warrant Certificate (collectively with the Exercise Certificate, the “Exercise Documents”); and
(ii) substantially contemporaneously with the delivery of the Exercise Certificate, payment to the Company of the Aggregate Exercise Price in accordance with Section 3(b); provided that, notwithstanding anything to the contrary herein, in no event shall the Exercise Price be lower than the par value of a Warrant Share.
(b) Payment of the Aggregate Exercise Price. Payment of the Aggregate Exercise Price shall be made, at the option of the Holder as expressed in the Exercise Certificate, by any of the following methods:
(i) by wire transfer of immediately available funds to an account designated in writing by the Company, in the amount of such Aggregate Exercise Price;
(ii) by instructing the Company to withhold a number of Warrant Shares then issuable upon exercise of this Warrant Certificate with an aggregate Fair Market Value as of the Exercise Date equal to such Aggregate Exercise Price; or
(iii) any combination of the foregoing.
In the event of any withholding of Warrant Shares pursuant to Section 3(b)(ii) or (iii) (solely to the extent of such withholding, a “Cashless Exercise”) where the number of such shares whose value is equal to the Aggregate Exercise Price is not a whole number, the number of such shares withheld by the Company shall be rounded up to the nearest whole share and the Company shall make a cash payment to the Holder (by wire transfer of immediately available funds) in an amount calculated as provided pursuant to Section 3(e) below.
To the extent permitted by applicable Law, for purposes of Rule 144, it is acknowledged and agreed that (i) the Warrant Shares issuable upon any exercise of this Warrant Certificate in any Cashless Exercise transaction shall be deemed to have been acquired on the Issue Date, and (ii) the holding period for any Warrant Shares issuable upon the exercise of this Warrant Certificate in any Cashless Exercise transaction shall be deemed to have commenced on the Issue Date; provided that the Company makes no representation or warranty regarding the commencement of the holding period of any Warrant Share.
(c) Automatic Cashless Exercise. To the extent this Warrant Certificate has not been exercised in full by the Holder prior to the date of any of the following events or circumstances, any portion of this Warrant Certificate that remains unexercised on such date shall be deemed to have been exercised automatically pursuant to a Cashless Exercise, in whole (and not in part), on the Business Day immediately preceding the earlier of (i) the occurrence of the Expiration Date, (ii) the occurrence of a Qualified IPO and (iii) the consummation of a Sale of the Company in which the consideration to be received by the Company or is shareholders consists solely of cash, Marketable Securities or a combination thereof.
(d) Delivery of Stock Certificates. With respect to any exercise of this Warrant Certificate by the Holder, upon receipt by the Company of the Exercise Documents and delivery of the Aggregate Exercise Price, the Company shall, within three (3) Business Days, deliver in accordance with the terms hereof to or upon the order of the Holder that number of Warrant Shares for the portion of this Warrant Certificate so exercised on such date, together with cash in lieu of any fraction of a share, as provided in Section 3(e) below. If such Warrant Shares are issued in certificated form, the Company shall deliver a certificate or certificates, to the extent possible, representing the number of Warrant Shares as the Holder shall request in the Exercise Certificate. If such Warrant Shares are issued in uncertificated form, the Company shall deliver upon request a confirmation evidencing the registration of such shares. Except as otherwise provided herein, upon any exercise hereof this Warrant Certificate shall be deemed to have been exercised and such certificate or certificates of Warrant Shares shall be deemed to have been issued, and the Holder shall be deemed to have become a holder of record of such Warrant Shares for all purposes, as of the Exercise Date.
(e) Fractional Shares. The Company shall not be required to issue a fractional Warrant Share upon exercise of any Warrant Certificate. As to any fraction of a Warrant Share that the Holder would otherwise be entitled upon such exercise, the Company shall pay to such Holder an amount in cash (by wire transfer of immediately available funds) equal to the product of (i) such fraction multiplied by (ii) the Fair Market Value of one Warrant Share on the Exercise Date.
(f) Stockholders’ Agreements. The Company acknowledges and agrees that simultaneously with the execution and delivery of this Warrant Certificate the Holder has executed and delivered to the Company, in escrow, an Adoption Agreement (as defined in the Voting Agreement) as required to be delivered pursuant to Section 6.1 of the Voting Agreement upon the issuance of additional shares of Series C Preferred Stock. Upon the initial exercise of this Warrant Certificate, such Adoption Agreement shall automatically (and without need of any further or other approval, consent or other action by any Person) be released from escrow; provided that no release from escrow shall occur or be deemed to occur until and unless the initial exercise of this Warrant Certificate shall have occurred pursuant to Section 3 hereof. The Company and the Holder each confirms and agrees that upon (but at no time prior to) such automatic release of the Adoption Agreement as provided in this clause (f) the Holder shall become an Investor and Stockholder (each as defined in the Voting Agreement) as contemplated by such Section 6.1 of the Voting Agreement. The Company further confirms and agrees that upon such release of the Adoption Agreement from escrow all conditions to issuance of the Warrant Shares upon exercise hereof required pursuant to the Voting Agreement shall have been satisfied. In addition, in connection with any other exercise of this Warrant Certificate pursuant to Section 3 hereof (including the initial exercise), upon the request of either party hereto the parties shall promptly (but in any event within three (3) Business Days of the applicable Exercise Date) enter into the other Stockholders’ Agreements, pursuant to which the Holder will become a party thereto; provided that, any term or provision hereof to the contrary notwithstanding, without limiting the obligations of the parties set forth in Section 3(d), the execution and delivery of such other Stockholders’ Agreements by the Holder shall not be a condition precedent to any exercise of this Warrant Certificate or the delivery of Warrant Shares in connection therewith pursuant to Section 3(d).
(g) Delivery of New Warrant Certificate. Unless the purchase rights represented by this Warrant shall have expired or shall have been fully exercised, the Company shall, at the time of delivery of the certificate or certificates representing the Warrant Shares being issued in accordance with Section 3(d) hereof, deliver to the Holder a new Warrant evidencing the rights of the Holder to purchase the unexpired and unexercised Warrant Shares called for by this Warrant. Such new Warrant shall in all other respects be identical to this Warrant.
(h) Valid Issuance of Warrant Certificate and Warrant Shares; Payment of Taxes. With respect to the exercise of this Warrant Certificate, the Company hereby represents, warrants, covenants and agrees as follows:
(i) This Warrant Certificate is, and any Warrant Certificate issued in substitution for or replacement of this Warrant Certificate shall be, upon issuance, duly authorized.
(ii) All Warrant Shares issuable upon the exercise of this Warrant Certificate (or any substitute or replacement Warrant Certificate) in accordance with its terms including payment of the Exercise Price, shall be, upon issuance, and the Company shall take all such actions as may be necessary or appropriate in order that such Warrant Shares are, validly issued, fully paid and non-assessable, issued without violation of any pre-emptive or similar rights of any shareholder of the Company and free and clear of all liens and charges.
(iii) The Company shall take all such actions as may be necessary to ensure that all such Warrant Shares are issued without violation by the Company of any applicable Law or any requirements of any U.S. or non-U.S. securities exchange upon which Warrant Shares may be listed at the time of such exercise.
(iv) The Company shall pay all expenses in connection with, and all governmental charges that may be imposed with respect to, the issuance or delivery of Warrant Shares issuable upon exercise of their Warrant Certificate.
(v) The Company is a corporation duly organized and validly existing under the Laws of the State of Delaware and has the capacity and corporate power and authority to enter into this Warrant Certificate.
(vi) The Company has taken or caused to be taken all action required to be taken by it, its Board, any of its shareholders or any other Person to authorize the execution, delivery and performance of this Warrant Certificate and the issuance of the Warrant Shares.
(vii) This Warrant Certificate has been duly executed and delivered by the Company.
(viii) The obligations of the Company under this Warrant Certificate are legal, valid and binding obligations, enforceable against the Company in accordance with the terms hereof.
(ix) The Company has complied with all obligations set forth in Section 3(i), below.
(i) Conditional Exercise. Notwithstanding any other provision hereof, if an exercise of all or any portion of this Warrant Certificate is to be made in connection with a Public Offering, a Sale of the Company or other possible liquidity transaction or event, such exercise may, at the election of the Holder, be conditioned upon the consummation of such transaction, in which case such exercise shall not be deemed to be effective until immediately prior to the consummation of such transaction or event.
(j) Reservation of Shares. The Company shall at all times during the Exercise Period reserve and keep available out of its authorized but unissued Series C Preferred Stock or (if applicable) other Equity Interests constituting Warrant Shares (including its Common Stock or other Equity Interests) or into which Series C Preferred Stock may be exercised for or converted into, solely for the purpose of issuance upon the exercise of this Warrant Certificate,
the maximum number of Warrant Shares or other Equity Interests issuable upon the exercise of this Warrant Certificate or the conversion or exchange of Warrant Shares issuable upon such exercise. The Company shall not increase the par value of any Warrant Shares receivable upon the exercise of this Warrant Certificate above the Exercise Price then in effect, and shall take all such actions within its power as may be necessary or appropriate in order that the Company may validly and legally issue fully paid and nonassessable Warrant Shares upon the exercise of this Warrant Certificate.
(k) Rule 144 Compliance. At all times following the closing of a Public Offering, and with a view to making available to the Holder the benefits of Rule 144 and any other rule or regulation of the SEC that may at any time permit a holder to sell securities of the Company to the public without registration or pursuant to a Registration Statement, the Company shall:
(i) use reasonable commercial efforts to make and keep adequate public information available, as required by clause (c) of Rule 144;
(ii) use reasonable commercial efforts to file with the SEC in a timely manner all reports and other documents required of the Company under the Securities Act and the Exchange Act; and
(iii) furnish, or otherwise make available to the Holder so long as the Holder owns Warrant Shares, promptly upon request, a written statement by the Company as to its compliance with the reporting requirements of Rule 144 and the Exchange Act.
(l) Ownership Cap. At all times following the occurrence of a Qualified IPO, the Company shall not knowingly effect the exercise of this Warrant Certificate, and the Initial Holder shall not have the right to exercise this Warrant Certificate to the extent that, after giving effect to such exercise, the Initial Holder (together with its Affiliates) would beneficially own in excess of 9.99% of the Equity Interests of the Company having ordinary voting rights outstanding immediately after giving effect to such exercise. For purposes of the foregoing sentence, the aggregate number of such voting Equity Interests beneficially owned by the Initial Holder and its Affiliates shall include the number of Warrant Shares issuable upon exercise of this Warrant Certificate with respect to which the determination of such aggregate number is being made, but shall exclude Warrant Shares that do not have ordinary voting rights or that would be issuable upon (i) exercise of the remaining, unexercised portion of this Warrant Certificate beneficially owned by the Initial Holder and its Affiliates and (ii) exercise or conversion of the unexercised or unconverted portion of any other Equity Interests of the Company beneficially owned by the Initial Holder and its Affiliates (including, without limitation, any Convertible Securities) subject to a limitation on conversion or exercise analogous to the limitations contained herein. Except as set forth in the preceding sentence, for purposes of this Section 3(k), beneficial ownership shall be calculated in accordance with Section 13(d) of the Exchange Act, it being acknowledged by the Holder that the Company is not representing to such Holder that such calculation is in compliance with Section 13(d) of the Exchange Act and such Holder is solely responsible for any schedules required to be filed in accordance therewith. To the extent that the limitation contained in this Section 3(l) applies, the determination of whether this Warrant is exercisable (in relation to other securities owned by such Holder) and of which a portion of this Warrant is exercisable shall be in the sole discretion of the Holder, and the submission of a Notice of Exercise shall be deemed to be the Holder’s determination of whether this Warrant is exercisable (in relation to other securities owned by such Holder) and of which portion of this Warrant is exercisable, in each case subject to the ownership cap, and the Company shall have no obligation to verify or confirm the accuracy of such determination. For purposes of this Warrant Certificate, in determining the number of outstanding Equity Interests of the Company having ordinary voting rights the Initial Holder of this Warrant Certificate may rely on the number of such outstanding Equity Interests as reflected in the most recent of (i) if available, the Company’s Form 10-K, Form 10-Q or other public filing with the SEC, as the case may be, (ii) a more recent public announcement by the Company, or (iii) any other notice by the Company or its transfer agent setting forth the number of such Equity Interests outstanding. In addition, upon the written request of the Initial Holder, the Company shall, within three (3) Business Days, confirm to the Initial Holder the number of its shares of Equity Interests having ordinary voting rights then outstanding.
(m) Market Stand-off Agreement. The Holder hereby agrees that it will not, without the prior written consent of the managing underwriter, during the period commencing on the date of the final prospectus relating to the initial registration by the Company of shares of its Common Stock or any other equity securities under the Securities Act on a registration statement on Form S-1 or Form S-3, and ending on the date specified by the Company and the managing underwriter (such period not to exceed one hundred eighty (180) days, or such other period as may be requested by the Company or an underwriter to accommodate regulatory restrictions on (1) the publication or other distribution of research reports, and (2) analyst recommendations and opinions, including, but not limited to, the restrictions contained in FINRA Rule 2241, or any successor provisions or amendments thereto), (i) lend; offer; pledge; sell; contract to sell; sell any option or contract to purchase; purchase any option or contract to sell; grant any option, right, or warrant to purchase; or otherwise transfer or dispose of, directly or indirectly, any Warrant Shares or any securities convertible into or exercisable or exchangeable (directly or indirectly) for Common Stock held immediately before the effective date of the registration statement for such offering or (ii) enter into any swap or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership of such securities, whether any such transaction described in clause (i) or (ii) above is to be settled by delivery of Common Stock or other securities, in cash, or otherwise. The foregoing provisions of this Section 3(m) shall only apply to a Qualified IPO, shall not apply to the sale of any shares to an underwriter pursuant to an underwriting agreement, or the transfer of any shares to any trust for the direct or indirect benefit of the Holder or the immediate family of the Holder; provided that the trustee of the trust agrees to be bound in writing by the restrictions set forth herein; and provided further that any such transfer shall not involve a disposition for value, and shall be applicable to the Holder only if all officers and directors are subject to the same restrictions and the Company uses commercially reasonable efforts to obtain a similar agreement from all stockholders individually owning more than one percent (1%) of the Company’s outstanding Common Stock (after giving effect to conversion into Common Stock of all outstanding preferred stock). The underwriters in connection with such registration are intended third-party beneficiaries of this Section 3(m) and shall have the right, power and authority to enforce the provisions hereof as though they were a party hereto. The Holder further agrees to execute such customary agreements as may be reasonably requested by the underwriters in connection with such registration that are consistent with this Section 3(m) or that are necessary to give further effect thereto. Any discretionary waiver or termination of the restrictions of any or all of such agreements by the Company or the underwriters shall apply pro rata to all holders of the Company’s equity securities subject to such agreements, based on the number of shares subject to such agreements. Upon execution and delivery by the Holder of the other Stockholders’ Agreements referenced in Section 3(f) above, this Section 3(m) shall, automatically and without need of any further action by any Person, terminate and no longer be of any force and effect.
Section 4. Adjustment to Number of Warrant Shares, Exercise Price, etc. The number of Warrant Shares issuable upon exercise of this Warrant Certificate shall be subject to adjustment from time to time as provided in this Section 4.
(a) Adjustment to Number of Warrant Shares Upon Reorganizations, Reclassifications, etc. In the event of any changes in the number of Warrant Shares Deemed Outstanding by reason of redemptions, recapitalizations, reclassifications, combinations or exchanges of shares, splits or reverse splits, separations, reorganizations, liquidations, substitutions, replacements or the like, the number and class of Warrant Shares available upon exercise of this Warrant Certificate in the aggregate and the Exercise Price per share shall be correspondingly adjusted as may be necessary in order to give the Holder of this Warrant Certificate, upon exercise, the total number, class, and kind of shares as the Holder would have owned (or would have had the right to own upon exercise hereof) had this Warrant Certificate been exercised prior to any such event and had the Holder continued to hold such Warrant Shares until after the event requiring adjustment. The form of this Warrant Certificate need not be changed because of any adjustment in the number of Warrant Shares subject to this Warrant Certificate.
(b) Changes to Conversion Rate or Conversion Price of Series C Preferred Stock. Without limiting the foregoing, but without duplication of any adjustment to be made pursuant to Section 4(a) above or Section 4(c) below, in the event of any changes to the Series C Conversion Price pursuant to the terms of (and as defined in) the Certificate of Incorporation, then upon exercise of this Warrant Certificate each Warrant Share acquired as a result of such exercise, without additional cost to the Holder, shall be subject to the Series C Conversion Price last in effect for the Series C Preferred Stock, as if the Holder had owned the Warrant Shares of record as of the date of such change.
(c) Adjustment to Number of Warrant Shares Upon Certain Dividends, etc. If the Company declares or pays a dividend or distribution on the outstanding shares of its Series C Preferred Stock payable in cash, Equity Interests or other property (or prior to the exercise of this Warrant Certificate in full the holders of Series C Preferred Stock become entitled to receive any such dividend or distribution payable in respect of any Equity Interests into which the Series C Preferred Stock is convertible or exchangeable), then upon exercise of this Warrant Certificate, for each Warrant Share acquired, the Holder shall receive, without additional cost to the Holder, the total amount, number and kind of cash, Equity Interests or other property which the Holder would have received had the Holder owned the Warrant Shares of record as of the date such dividend or distribution occurred.
(d) Mandatory Conversion of Series C Preferred Stock. Without limiting the foregoing, but without duplication of any adjustment to be made pursuant to Section 4(a), Section (b) or Section 4(c) above, in the event that, pursuant to the provisions of the Certificate of Incorporation, all outstanding shares of Series C Preferred Stock are converted, automatically or by action of the holders thereof, into Common Stock (or other Equity Interests), then from and after the date on which all outstanding shares of Series C Preferred Stock have been so converted, this Warrant Certificate shall be exercisable for such number of shares of Common Stock (or other Equity Interests) into which the Warrant Shares would have been converted had the Holder held such Warrant Shares on the date of such conversion, and the Exercise Price shall equal the Exercise Price in effect as of immediately prior to such conversion divided by the number of shares of Common Stock (or other Equity Interests) into which one Warrant Share would have been converted, all subject to further adjustment thereafter from time to time in accordance with the provisions of this Warrant Certificate.
(e) No Adjustment for Excluded Securities. Notwithstanding the foregoing, no adjustments shall be made under this Section 4 as the result of the issuance or deemed issuance of Excluded Securities.
(f) Certificate as to Adjustment.
(i) As promptly as reasonably practicable following any change or adjustment of the type described above in this Section 4, but in any event not later than five (5) Business Days thereafter, the Company shall furnish to the Holder a certificate of a Responsible Officer setting forth in reasonable detail such change or adjustment and the facts upon which it is based and certifying the calculation thereof.
(ii) As promptly as reasonably practicable following the receipt by the Company of a written request by the Holder, but in any event not later than five (5) Business Days thereafter, the Company shall furnish to the Holder a certificate of a Responsible Officer certifying the number of Warrant Shares or the amount, if any, of other Equity Interests, securities or assets then issuable upon exercise of the Warrant Certificate.
(g) Notices. In the event that, at any time during the Exercise Period the Company shall take a record of the holders of its outstanding capital stock (or other Equity Interests at the time issuable upon exercise of this Warrant Certificate) for the purpose of:
(i) entitling or enabling such holders to receive any dividend or other distribution, to receive any right to subscribe for or purchase any shares of capital stock of any class or any other securities, or to receive any other security;
(ii) (x) any capital reorganization of the Company, any reclassification of any outstanding securities, any consolidation or merger of the Company with or into another Person, any Public Offering of the Company’s Equity Interests, (y) a sale of all or substantially all of the Company’s assets to another Person or (z) any liquidation, bankruptcy, dissolution, winding-up or any similar event of the Company; or
(iii) the voluntary or involuntary dissolution, liquidation or winding-up of the Company (including by way of a bankruptcy or equivalent insolvency proceeding);
then, and in each such case, the Company shall send or cause to be sent to the Holder at least five (5) Business Days prior to the applicable record date or the applicable expected effective date, as the case may be, for the event, a written notice specifying, as the case may be, (A) the record date for such dividend, distribution or other right or action, and a description of such dividend, distribution or other right or action, or (B) the effective date on which such reorganization, reclassification, consolidation, merger, sale, dissolution, liquidation or winding-up is proposed to take place, and the date, if any is to be fixed, as of which the books of the Company shall close or a record shall be taken with respect to which the holders of record of its capital stock (or such other Equity Interests at the time issuable upon exercise of the Warrant Certificate) shall be entitled to exchange their shares of capital stock (or such other Equity Interests), for securities or other property deliverable upon such reorganization, reclassification, consolidation, merger, sale, dissolution, liquidation or winding-up, and the amount per share and character of such exchange applicable to the Warrant Certificate and the Warrant Shares.
(h) Election to Convert Warrant Certificate in Respect of an Issuance of Senior Equity Interests. In the event that at any time after the date hereof the Company issues Series D preferred stock or another class or series of its Equity Interests having rights, preferences, priorities or the like that are senior or otherwise prior to the Series C Preferred Stock (collectively, “Senior Equity Interests”), the Holder shall have the option, exercisable in its sole discretion by written notice to the Company, to convert this Warrant Certificate (and the related rights and interests hereunder), to the extent this Warrant Certificate is unexercised at the time the Holder exercises such option, into a warrant to purchase such Senior Equity Interests; provided that in furtherance of the foregoing the parties agree to amend or otherwise modify this Warrant Certificate in all ways necessary and appropriate to effect such conversion of this Warrant Certificate into a warrant to purchase such Senior Equity Interests, including as follows: (i) all references herein to Series C Preferred Stock shall be changed to refer to the description of the Senior Equity Interests provided in the Certificate of Incorporation (as amended or modified); (ii) the Exercise Price of the Warrant Shares shall be automatically adjusted to equal the original per share issue price of the Senior Equity Interests as provided in the applicable sale or subscription agreement related thereto (the “Senior Equity Sales Price”), subject to further adjustment from time to time in accordance with Section 4, as applicable; and (iii) after giving effect to the conversion of the Warrant Certificate contemplated by this clause (h), the number of Warrant Shares issuable upon exercise of this Warrant Certificate shall be automatically adjusted to the product of (x) the total number of shares of Series C Preferred Stock that, immediately prior to the conversion of the Warrant Certificate contemplated by this clause (h), remain issuable upon exercise in full of this Warrant Certificate, multiplied by (y) a fraction having a numerator equal to Exercise Price in effect at the time referenced in clause (x) above and a denominator equal to the Senior Equity Sales Price, subject to further adjustment from time to time in accordance with Section 4; provided further that the option granted to the Holder pursuant to this clause (h) shall only apply to the immediate next issuance of Senior Equity Interests, if any, issued after the date hereof, and not any issuance of Senior Equity Interests, if any, that follows such immediate next issuance.
Section 5. [Reserved].
Section 6. Warrant Register. The Company shall keep and properly maintain at its principal executive offices a register (the “Warrant Register”) for the registration of this Warrant Certificate and any transfers thereof. The Company may deem and treat the Person in whose name this Warrant Certificate is registered on such register as the Holder thereof for all purposes, and the Company shall not be affected by any notice to the contrary, except any assignment, division, combination or other transfer of this Warrant Certificate effected in accordance with the provisions of this Warrant Certificate.
Section 7. Transfer of Warrant Certificate. Subject to the transfer conditions referred to in the legend endorsed hereon and Section 11, this Warrant Certificate and all rights hereunder are transferable, in whole or in part, by the Holder without charge to the Holder, upon surrender of this Warrant Certificate to the Company at its then principal executive offices with a properly completed and duly executed instrument of assignment in the form attached hereto as Exhibit B (an “Assignment”). Upon such compliance, surrender and delivery, the Company shall execute and deliver a new Warrant Certificate or Warrant Certificates in the name of the assignee or assignees and in the denominations specified in such Assignment, and shall issue to the assignor a new Warrant Certificate evidencing the portion of this Warrant Certificate, if any, not so assigned, and this Warrant Certificate shall promptly be cancelled.
Section 8. The Holder Not Deemed a Shareholder; Limitations on Liability. Except as otherwise specifically provided herein (including in Section 4(c) above), (i) prior to the Exercise Date, the Holder shall not be entitled to receive dividends, nor shall anything contained in this Warrant Certificate be construed to confer upon the Holder, as such, any of the rights of a shareholder of the Company or any right to receive dividends or subscription rights, and (ii) prior to the registration of the Holder in the share register of the Company with respect to the Warrant Shares to which the Holder is then entitled to receive upon the due exercise of this Warrant Certificate, the Holder shall not be entitled to vote, nor shall anything contained in this Warrant Certificate be construed to confer upon the Holder, as such, any right to vote, give or withhold consent to any corporate action (whether any reorganization, issue of stock, reclassification of stock, consolidation, merger, conveyance or otherwise) or receive notice of meetings. In addition, nothing contained in this Warrant Certificate shall be construed as imposing any liabilities on the Holder to purchase any securities (upon exercise of this Warrant Certificate or otherwise) or as a shareholder of the Company, whether such liabilities are asserted by the Company or by creditors of the Company. Notwithstanding this Section 8, the Company shall provide the Holder with copies of the same notices and other information given to all stockholders of the Company generally, contemporaneously with the giving thereof to such stockholders.
Section 9. Replacement on Loss; Division and Combination.
(a) Replacement of Warrant Certificate on Loss. Subject to any further requirements in relation to the cancellation of this Warrant Certificate pursuant to applicable Law, upon receipt of evidence reasonably satisfactory to the Company of the loss, theft, destruction or mutilation of this Warrant Certificate and upon delivery of an indemnity reasonably satisfactory to it (it being understood that a written indemnification agreement or affidavit of loss of the Holder shall be a sufficient indemnity) and, in case of mutilation, upon surrender of such Warrant Certificate for cancellation to the Company, the Company at its own expense shall execute and deliver to the Holder, in lieu hereof, a new Warrant Certificate of like tenor and exercisable for an equivalent number of Warrant Shares as this Warrant Certificate so lost, stolen, mutilated or destroyed; provided that, in the case of mutilation, no indemnity shall be required if this Warrant Certificate in identifiable form is surrendered to the Company for cancellation.
(b) Division and Combination of Warrant Certificate. Subject to compliance with the applicable provisions of this Warrant Certificate as to any transfer or other assignment which may be involved in such division or combination, this Warrant Certificate may be divided or, following any such division of this Warrant Certificate, subsequently combined with other Warrant Certificates, upon the surrender of this Warrant Certificate or Warrant Certificates to the Company at its then principal executive offices, together with a written notice specifying the names and denominations in which new Warrant Certificates are to be issued, signed by each applicable Holder or its agents or attorneys. Subject to compliance with the applicable provisions of this Warrant Certificate as to any transfer or assignment which may be involved in such division or combination, the Company shall at its own expense execute and deliver a new Warrant Certificate or Warrant Certificates in exchange for this Warrant Certificate or Warrant Certificates so surrendered in accordance with such notice. Such new Warrant Certificate or Warrant Certificates shall be of like tenor to the surrendered Warrant Certificate or Warrant Certificates and shall be exercisable in the aggregate for an equivalent number of Warrant Shares as this Warrant Certificate or Warrant Certificates so surrendered in accordance with such notice.
Section 10. Disputes; No Impairment, etc. The parties hereto agree as follows:
(a) Disputes. In the event of any dispute which arises between the Holder and the Company (including the Board of the Company) with respect to the calculation or determination of the adjusted Exercise Price, the number of Warrant Shares, other Equity Interests, Fair Market Value, cash or other property issuable upon exercise of this Warrant Certificate, the amount or type of consideration due to the Holder in connection with any event, transaction or other matter described in Section 4 above or any other matter involving this Warrant Certificate or the Warrant Shares that is not resolved by the parties after good faith discussions and efforts to reach resolution, upon the request of the Holder the disputed issue(s) shall be submitted to a firm of independent investment bankers or public accountants of recognized national standing, which (i) shall be chosen by the Company and be reasonably satisfactory to the Holder and (ii) shall be completely independent of the Company (an “Independent Advisor”), for determination, and such determination by the Independent Advisor shall be binding upon the Company and the Holder with respect to this Warrant Certificate, any Warrant Shares issued or issuable in connection herewith, the Exercise Price therefor, or any other matter in dispute, as the case may be, absent manifest error. Costs and expenses of the Independent Advisor shall be shared 50/50 by the Company and the Holder.
(b) Equitable Equivalent. In case any event shall occur as to which the provisions of Section 10(a) above are not strictly applicable but the failure to make any adjustment would not, in the reasonable, good faith opinion of the Holder, fairly protect the rights and benefits of the Holder represented by this Warrant Certificate in accordance with the essential intent and principles of Sections 4 and 10(a), then, in any such case, at the request of the Holder, the Company shall submit the matter and issues raised by the Holder to an Independent Advisor, which shall give its opinion upon the adjustment, if any, on a basis consistent with the essential intent and principles established in Sections 4 and 10(a), to the extent necessary to preserve the rights and benefits represented by this Warrant Certificate. Upon receipt of such opinion, the Company will promptly mail a copy thereof to the Holder and shall make the adjustments described therein, if any. Costs and expenses of the Independent Advisor shall be shared 50/50 by the Company and the Holder.
(c) No Avoidance. The Company shall not, by way of amendment of any of its Organic Documents or through any consolidation, merger, reorganization, transfer of assets, dissolution, issue or sale of securities or any other voluntary action, avoid or seek to avoid the observance, performance or intended results of any of the terms or provisions of this Warrant Certificate, but will at all times in good faith assist in the carrying out of all such terms or provisions and in the taking of all such action as may be necessary or appropriate in order to protect the rights of the Holder against dilution or other impairment as if the Holder was a shareholder of the Company entitled to the benefit of fiduciary duties afforded to shareholders under Delaware law.
Section 11. Compliance with the Securities Act.
(a) Agreement to Comply with the Securities Act, etc.
(i) Legend. The Holder, by acceptance of this Warrant Certificate, agrees to comply in all respects with the provisions of this Section 11 and the restrictive legend requirements set forth on the face of this Warrant Certificate and further agrees that it shall not offer, sell or otherwise dispose of this Warrant Certificate or any Warrant Shares to be issued upon exercise hereof except under circumstances that will not result in a violation of the Securities Act. Subject to clause (ii) below, this Warrant Certificate and all Warrant Shares issued upon exercise of this Warrant Certificate (unless registered under the Securities Act) shall be stamped or imprinted with a legend in substantially the form as set forth on the face hereof.
(ii) Removal of Restrictive Legends. Neither this Warrant Certificate nor any certificates evidencing Warrant Shares issuable or deliverable under or in connection with this Warrant Certificate shall contain any legend restricting the transfer of such Warrant Certificate or Warrant Shares as applicable (including the legend required above in clause (i)) in any of the following circumstances: (A) following any sale of this Warrant Certificate or any Warrant Shares issued or delivered to the Holder under or in connection herewith pursuant to Rule 144 or (B) if such legend is not required under applicable requirements of the Securities Act (including judicial interpretations and pronouncements issued by the staff of the SEC), including clause (b)(1) of Rule 144, or any applicable state or foreign securities laws; provided that, in the case of clause (B) above, if the Company requests, a customary opinion, in form and substance reasonably satisfactory to the Company to such effect, will be delivered to the Company by counsel to the Holder (collectively, the “Unrestricted Conditions”). This Warrant Certificate or Warrant Shares, as the case may be, shall be issued free of all legends if the Unrestricted Conditions are met at the time of issuance
(iii) Replacement Warrant Certificate. The Company agrees that at such time as the Unrestricted Conditions have been satisfied it shall promptly (but in any event within five (5) Business Days) following written request from the Holder issue a replacement Warrant Certificate or replacement Warrant Shares, as the case may be, free of all restrictive legends.
(iv) Sale of Unlegended Shares. The Holder agrees that the removal of the restrictive legend from this Warrant Certificate and any certificates representing securities as set forth in Section 11(a)(ii) above is predicated upon the Company’s reliance that the Holder will sell this Warrant Certificate or any such securities pursuant to either an effective Registration Statement or otherwise pursuant to the requirements of the Securities Act, including any applicable prospectus delivery requirements, or an exemption therefrom, and that if such securities are sold pursuant to a Registration Statement, they will be sold in compliance with the plan of distribution set forth therein.
(b) Representations of the Holder. In connection with the issuance of this Warrant Certificate, the Holder represents, as of the Issue Date, to the Company by acceptance of this Warrant Certificate as follows:
(i) The Holder is an “accredited investor” as defined in Rule 501 of Regulation D promulgated under the Securities Act. The Holder is acquiring this Warrant Certificate and the Warrant Shares to be issued upon exercise hereof for investment for its own account and not with a view towards, or for resale in connection with, the public sale or distribution of this Warrant Certificate or the Warrant Shares, except pursuant to sales registered or exempted under the Securities Act.
(ii) The Holder understands and acknowledges that this Warrant Certificate and the Warrant Shares to be issued upon exercise hereof are “restricted securities” under the Securities Act inasmuch as they are being acquired from the Company in a transaction not involving a public offering and that, under such Laws and applicable regulations, such securities may be resold without registration under the Securities Act only in certain limited circumstances. In addition, the Holder represents that it is familiar with Rule 144, as presently in effect, and understands the resale limitations imposed thereby and by the Securities Act.
(iii) The Holder acknowledges that it can bear the economic and financial risk of its investment for an indefinite period and has such knowledge and experience in financial or business matters that it is capable of evaluating the merits and risks of the investment in this Warrant Certificate and the Warrant Shares. The Holder has had an opportunity to ask questions and receive answers from the Company regarding the terms and conditions of the offering of this Warrant Certificate and the business, properties, prospects and financial condition of the Company.
Section 12. Notices. All notices, requests, consents, claims, demands, waivers and other communications hereunder shall be in writing and shall be deemed to have been given: (i) when delivered by hand (with written confirmation of receipt); (ii) when received by the addressee if sent by a nationally recognized overnight courier (receipt requested); (iii) on the date sent by facsimile or e-mail of a PDF document (with confirmation of transmission) if sent during normal business hours of the recipient, and on the next Business Day if sent after normal business hours of the recipient; or (iv) on the third day after the date mailed, by certified or registered mail, return receipt requested, postage prepaid. Such communications must be sent to the respective parties at the addresses indicated below (or at such other address for a party as shall be specified in a notice given in accordance with this Section 12).
| | | | | | | | |
| If to the Company: | Pear Therapeutics, Inc. |
| | 200 State Street, 13th Floor |
| | Boston, MA 02109 |
| | Attention: Ronan O’Brien |
| | E-mail: ronan.obrien@peartherapeutics.com |
| | |
| with a copy to: | Foley Hoag LLP |
| | 55 Seaport Boulevard |
| | Boston, MA 02210 |
| | Attention: David Broadwin, Esq. |
| | E-mail: DAB@foleyhoag.com |
| | |
| If to the Holder: | Perceptive Credit Holdings III, LP |
| | c/o Perceptive Advisors LLC |
| | 51 Astor Place, 10th Floor |
| | New York, NY 10003 |
| | Attention: Sandeep Dixit |
| | E-mail: Sandeep@perceptivelife.com |
| | |
| with a copy to: | Morrison & Foerster LLP |
| | 250 West 55th Street |
| | New York, NY 10019 |
| | Attention: Mark Wojciechowski, Esq. |
| | E-mail: mwojciechowski@mofo.com |
Section 13. Cumulative Remedies. Except to the extent expressly provided in Section 7 to the contrary, the rights and remedies provided in this Warrant Certificate are cumulative and are not exclusive of, and are in addition to and not in substitution for, any other rights or remedies available at Law, in equity or otherwise.
Section 14. Entire Agreement. This Warrant Certificate constitutes the sole and entire agreement of the parties to this Warrant Certificate with respect to the subject matter contained herein and supersedes all prior and contemporaneous understandings and agreements, both written and oral, with respect to such subject matter.
Section 15. Successor and Assigns. This Warrant Certificate and the rights evidenced hereby shall be binding upon and shall inure to the benefit of the parties hereto and the successors of the Company and the successors and permitted assigns of the Holder. Such successor or permitted assign of the Holder shall be deemed to be the “Holder” for all purposes hereunder.
Section 16. No Third-Party Beneficiaries. Except to the extent expressly provided in Section 3(m) above, this Warrant Certificate is for the sole benefit of the Company and the Holder and their respective successors and, in the case of the Holder, permitted assigns, and nothing herein, express or implied, is intended to or shall confer upon any other Person any legal or equitable right, benefit or remedy of any nature whatsoever, under or by reason of this Warrant Certificate.
Section 17. Headings. The headings in this Warrant Certificate are for reference only and shall not affect the interpretation of this Warrant Certificate.
Section 18. Amendment and Modification; Waiver. Except as otherwise provided herein, this Warrant Certificate may only be amended, modified or supplemented by an agreement in writing signed by each party hereto. No waiver by the Company or the Holder of any of the provisions hereof shall be effective unless explicitly set forth in writing and signed by the party so waiving. No waiver by any party shall operate or be construed as a waiver in respect of any failure, breach or default not expressly identified by such written waiver, whether of a similar or different character, and whether occurring before or after that waiver. No failure to exercise, or delay in exercising, any rights, remedy, power or privilege arising from this Warrant Certificate shall operate or be construed as a waiver thereof, nor shall any single or partial exercise of any right, remedy, power or privilege hereunder preclude any other or further exercise thereof or the exercise of any other right, remedy, power or privilege.
Section 19. Severability. If any term or provision of this Warrant Certificate is invalid, illegal or unenforceable in any jurisdiction, such invalidity, illegality or unenforceability shall not affect any other term or provision of this Warrant Certificate or invalidate or render unenforceable such term or provision in any other jurisdiction.
Section 20. Governing Law. This Warrant Certificate shall be governed by and construed in accordance with the internal Laws of the State of New York without effect to any choice or conflict of Law provision or rule (whether of the State of New York or any other jurisdiction) that would cause the application of Laws of any jurisdiction other than those of the State of New York.
Section 21. Submission to Jurisdiction. Any legal suit, action or proceeding arising out of or based on this Warrant Certificate or the transactions contemplated hereby may be instituted in the federal courts of the United States or the courts of the State of New York, in each case located in the city and county of New York. Each party irrevocably submits to the exclusive jurisdiction of such courts in any such suit, action or proceeding. Service of process, summons, notice or other document by certified or registered mail to such party’s address set forth in Section 12 shall be effective service of process for any suit, action or other proceeding, and the parties irrevocably and unconditionally waive any objection to the laying of venue of any suit, action or other proceeding in such courts and irrevocably waive and agree not to plead or claim in any such court that any such suit, action or proceeding has been brought in an inconvenient forum.
Section 22. Counterparts. This Warrant Certificate may be executed in counterparts, each of which shall be deemed an original, but all of which together shall be deemed to be one and the same agreement. A signed copy of this Warrant Certificate delivered by facsimile, e-mail or other means of electronic transmission shall be deemed to have the same legal effect as delivery of an original signed copy of this Warrant Certificate.
Section 23. No Strict Construction. This Warrant Certificate shall be construed without regard to any presumption or rule requiring construction or interpretation against the party drafting an instrument or causing any instrument to be drafted.
[SIGNATURE PAGE FOLLOWS]
IN WITNESS WHEREOF, the Company has duly executed this Warrant Certificate on the Issue Date.
| | | | | | | | | | | |
| PEAR THERAPEUTICS, INC. |
| | |
| By | | |
| | Name: | |
| | Title: | |
Exhibit A
to Warrant Certificate
FORM OF EXERCISE CERTIFICATE
(To be signed only upon exercise of Warrant Certificate)
To: Pear Therapeutics, Inc.
200 State Street, 13th Floor
Boston, MA 02109
Attention: [____]
Reference is made to that certain Warrant Certificate, having an issue date of [______] and bearing Warrant Certificate No. [___] (the “Warrant Certificate”), issued by Pear Therapeutics, Inc. (the “Company”) to [Name of Holder] (the “HolderOLDER WORKERS BENEFIT PROTECTION ACT NOTICE
Pear Therapeutics (US), Inc. (the “Company”) has decided that to better align its operations and business plans, it needs to conduct a reduction in force that will affect some of its employees, effective November 15, 2022. This decision will impact your position and, as a result, you are being offered a separation package including Severance Pay in exchange for signing a Separation Agreement. In accordance with the Older Workers Benefit Protection Act, the Company is obligated to provide you with certain information, provided below, relating to individuals selected for termination in the appropriate decisional unit and therefore, eligible for the offered Severance Pay, and any individuals in the same decisional unit not selected. This information is confidential and should not be shared with anyone other than your spouse or legal advisor.
This group termination program is being undertaken following the Company’s assessment of its business and financial needs and capabilities at present and going forward. Determination of which individuals would be terminated with eligibility for separation pay was made on the basis of a variety of factors, including (without limitation) whether individuals possess the necessary backgrounds, experiences and skills to meet the Company’s needs and goals going forward, the scope of the work performed by the individuals, and in some cases, factors specific to an individual. The decisional unit for this program was all Pear Therapeutic employees, wherever located, in its three offices and remote locations.
Individuals eligible for Severance Pay who are 40 years of age or older are being given at least 45 days to consider the Agreement. Individuals eligible for Severance Pay who are less than 40 years of age are being given fourteen (14) days to consider the Agreement.
Personnel Selected for Termination and Eligible for Severance Pay and Benefits in Exchange for a Release
[***]
Personnel Not Selected for Termination and Not Eligible for Severance Pay and Benefits in Exchange for a Release
[***]
EXHIBIT D
Option Amendment Agreement
This Option Amendment Agreement (this “Option Amendment Agreement”) is entered
into by Pear Therapeutics, Inc., (the “Company”). Unless otherwise defined herein, capitalized terms used herein have the meanings ascribed thereto in the Warrant Certificate.
The undersigned, as holder of a right to purchase Warrant Shares (as defined in the Warrant Certificate) of the Company pursuant to the terms of the Warrant Certificate, a copy of which is attached to this Exercise Certificate, hereby irrevocably elects, and Kathy Jeffery, an employee of the Company’s subsidiary, Pear Therapeutics (US), Inc. (the “Holder”). In order to be effective, this Option Amendment Agreement must be signed by the Holder and returned to Ronan O’Brien via email (ronan.obrien@peartherapeutics.com) no later than December 24, 2022.
WHEREAS, the Company has granted to the Holder, under the Company’s 2021 Stock Option and Incentive Plan (the “Plan”), one or more stock options (the “Option” or “Options”) to purchase shares of the Company’s class A common stock, pursuant to the terms of the Plan and the applicable option agreement granting the Options (the “Option Agreements”);
WHEREAS, the parties now desire to amend the Options to provide for a longer period of time to exercise the purchase right represented byOptions following termination of such Warrant Certificate for, and to purchase thereunder, [__________ (___________)] Warrant Sharesthe Holder’s employment with the Company; and
WHEREAS, the Holder understands that as a result of this Option Amendment Agreement any Option that qualified as an Incentive Stock Option, or ISO, shall no longer qualify as an Incentive Stock Option, and shall as of the date of execution of this Option Amendment Agreement automatically become a non-statutory Option; and the Holder understands further that upon exercise of this Option they shall have an obligation to satisfy any tax withholding obligation of the Company and herewith makes payment with respect to this Exercise Certificate of [____________ Dollars ($___________)] therefor by the following method.
(Check all that apply):
☐ The undersigned hereby elects to make payment of the Aggregate Exercise Price of [_________Dollars ($_________)] for [(_________)] shares of Series C Preferred Stock using the method described in Section 3(b)(i).
☐ The undersigned hereby elects to make payment of the Aggregate Exercise Price of [_________Dollars ($_________)] for [(_________)] shares of Series C Preferred Stock using the method described in Section 3(b)(ii).
☐ The undersigned hereby elects to make payment of the Aggregate Exercise Price of [_________Dollars ($_________)] for [(_________)] shares of Series C Preferred Stock using the method described in Section 3(b)(iii).
DATED:___________________
| | | | | | | | |
| [NAME OF HOLDER] |
| | |
| By | |
| | Name: |
| | Title: |
Exhibit B
to Warrant Certificate
FORM OF ASSIGNMENT
[DATE OF ASSIGNMENT]
Reference is made to that certain Warrant Certificate, having an issue date of [ __________] and bearing Warrant Certificate No. [____] (the “Warrant Certificate”), issued by Pear Therapeutics, Inc. (the “Company”) to the undersigned (the “Holder”). Unless otherwise defined herein, capitalized terms used herein have the meanings ascribed thereto in the Warrant Certificate.
THE UNDERSIGNED, [NAME OF HOLDER], is the holder of the Warrant Certificate and is entitled to purchase up to [__________] Warrant Shares pursuant to the terms thereof. A copy of the Warrant Certificate is attached hereto.
FOR VALUE RECEIVED, the Holder hereby sells, assigns and transfers to [NAME OF ASSIGNEE] (the “Assignee”) the right to acquire [all Warrant Shares entitled to be purchased upon exercise of the Warrant Certificate] [___________ of the Warrant Shares entitled to be purchased upon exercise of the Warrant Certificate] (the “ Assignment”). In furtherance of the foregoing assignment, the Holder hereby irrevocably instructs the Company to (i) memorialize such assignment in the Warrant Register as required pursuant to Section 6 of the Warrant Certificate, and (ii) pursuant to Section 7 of the Warrant Certificate, execute and deliver to the Assignee [and the Holder][a new Warrant Certificate][new Warrant Certificates] reflecting the foregoing assignment ([each] a “Substitute Warrant Certificate”). The Assignee acknowledges and agrees that its Substitute Warrant Certificate and the Warrant Shares to be issued upon exercise thereof are being acquired for investment and that the Assignee will not offer, sell or otherwise dispose of its Substitute Warrant Certificate or any Warrant Shares to be issued upon exercise or conversion thereof except under circumstances which will not result in a violation of the Securities Act or any applicable state securities Laws. The Assignee represents and warrants for the benefit of the Company that the Assignee is an “accredited investor” within the meaning of Rule 501 of Regulation D promulgated under the Securities Act.
To the extent required pursuant to Section 11(a) of the Warrant Certificate, the Assignee acknowledges and agrees that a restrictive legend shall be applied to the Assignee’s Substitute Warrant Certificate and the Warrant Shares issuable upon exercise of such certificate substantially consistent with the legend required pursuant to Section 11(a) of the Warrant Certificate.
The Assignee acknowledges and agrees that simultaneously with the execution and delivery of this Assignment, the Assignee has executed and delivered to the Company, in escrow, an Adoption Agreement to the Voting Agreement. Upon the initial exercise of the Substitute Warrant Certificate, such Adoption Agreement shall automatically (and without need of any further or other approval, consent or other action by any Person) be released from escrow.
such exercise, pursuant to the terms of the Plan.
Exhibit B-1
The Assignee further confirms, acknowledges and agrees to its obligation to enter into and become bound by the other Stockholders’ Agreements upon any exercise by it of the Substitute Warrant Certificate.
[SIGNATURE PAGE FOLLOWS]NOW, THEREFORE, the parties agree as follows:
1.Notwithstanding anything to the contrary in the applicable Option Agreement or the Plan, (i) your stock options will continue to vest through February 29, 2024 (notwithstanding your termination of employment), (ii) the period for exercising your stock options, to the extent that they have vested as of February 29, 2024, will be extended until the earlier of March 31, 2024, or the expiration of the applicable Option, as stated in the applicable Option Agreement, and (iii) any of your remaining stock options that would not vest on or before February 29, 2024 will terminate immediately.
1.Except as expressly amended by this Option Amendment Agreement, all other terms and conditions of the Options shall remain in full force and effect without any modification or amendment, provided however that as a result of this Option Amendment Agreement, any Option that was an Incentive Stock Option (as defined in the Plan) shall no longer qualify as an Incentive Stock Option, and shall as of the date of execution of this Option Amendment Agreement automatically become a non-statutory Option.
IN WITNESS WHEREOF, the parties hereto agree as setCompany has caused this Option Amendment Agreement to be executed by its duly authorized officer.
Pear Therapeutics, Inc.
By: /s/ Christopher D.T. Guiffre
Name: Christopher D.T. Guiffre
Title: CFO & COO
Dated: 12/17/2022
The Holder understands that as a forth aboveresult of this Option Amendment Agreement any Option that qualified as an ISO shall no longer qualify as an ISO, and shall as of the date first written above.
| | | | | | | | |
| [NAME OF HOLDER] |
| | |
| By | |
| Name: |
| Title: |
| | | | | | | | |
| Accepted and agreed, | |
| | |
| [NAME OF ASSIGNEE] | |
| | |
| By | | |
| Name: | |
| Title: | |
| | |
| PEAR THERAPEUTICS, INC. | |
| | |
| By | | |
| Name: | |
| Title: | |
Exhibit B-3of execution of this Option Amendment Agreement automatically become a non-statutory Option. The Holder understands further that upon exercise of this Option they shall have an obligation to satisfy any tax withholding obligation of the Company with respect to such exercise, pursuant to the terms of the Plan.
ACCEPTED AND AGREED:
Name: /s/ Katherine Jeffery
By: Katherine Jeffery
Date: 12/19/2022
Exhibit 10.35
Execution Version
AMENDED AND RESTATED SECURITYFIRST AMENDMENT TO EXCLUSIVE LICENSE AGREEMENT OF June 28, 2018
THIS AMENDED AND RESTATED SECURITY AGREEMENT (this “Agreement”), dated as of March [_], 2022 is made by and among PEAR THERAPEUTICS, INC., a Delaware corporation (“Holdings”), PEAR THERAPEUTICS (US), INC., a Delaware corporation (the “Borrower”), certain Subsidiaries of Holdings party hereto or having acceded hereto pursuant to Section 22 (each a “Subsidiary Guarantor” and, together with Holdings and the Borrower, each a “Grantor” and, collectively, the “Grantors”), and PERCEPTIVE CREDIT HOLDINGS III, LP, as administrative agent for the Lenders referred to below (in such capacity, together with its successors and assigns, the “Administrative Agent”).
WHEREAS, the Borrower and the Administrative Agent entered into that certain Security Agreement, dated as of June 30, 2020 (the “Existing Security Agreement”), pursuant to the terms of that certain Credit Agreement and Guaranty, dated as of June 30, 2020 (the “Existing Credit Agreement”), by and among the Borrower, certain Subsidiaries of the Borrower party thereto from time to time, the lenders from time to time party thereto (the “Lenders”) and the Administrative Agent;
WHEREAS, the parties hereto have agreed to amend and restate the Existing Credit Agreement pursuant to the terms of that certain Amended and Restated Credit Agreement and Guaranty, dated as of the date hereof (as amended or otherwise modified from time to time, the “Credit Agreement”), by and among Holdings, the Borrower, the other Subsidiaries of Holdings from time to time party thereto, the Lenders from time to time party thereto and the Administrative Agent; and
WHEREAS, as a condition precedent to the effectiveness of the Credit Agreement, the parties hereto desire to amend University of Virginia Licensing & Ventures Group – Pear Therapeutics (US), Inc.
This First Amendment (“First Amendment”) is made effective this [day] day of [month], 2022 (“First Amendment Date”) by and between the University of Virginia Patent Foundation d/b/a University of Virginia Licensing & Ventures Group (“UVA LVG”), a Virginia non-profit corporation having a principal place of business at 722 Preston Avenue, Suite 107, Charlottesville, Virginia 22903, and Pear Therapeutics, Inc. n/k/a Pear Therapeutics (US), Inc., (“Pear”), a Delaware for-profit corporation with offices at 200 State Street, 13th Floor, Boston, MA 02109 (each a “Party”, and collectively the “Parties”).
WITNESSETH
WHEREAS, UVA LVG and BeHealth Solutions, LLC (“BeHealth”) entered into an Exclusive License Agreement dated April 1, 2011, previously amended on March 19, 2015 (collectively the “Prior Agreement”);
WHEREAS, UVA LVG and BeHealth previously agreed to supersede the Prior Agreement with an amended and reinstated license agreement, dated June 28, 2018, related to the UVA LVG Tech ID RITTERBAND-SHUTI (“SHUTI Amended and Restated Agreement”);
WHEREAS, as part of the Assignment, License & Services Agreement effective as of March 24, 2018, by and between BeHealth and Pear, BeHealth transferred to Pear, among other things, all of BeHealth’s rights and obligations under the SHUTI Amended and Restated Agreement;
WHEREAS, the Parties agree to amend the SHUTI Amended and restate the Existing SecurityRestated Agreement in its entirety onto revise and update certain provisions in accordance with the terms and conditions set forth herein.below; and
NOW, THEREFORE, the parties, in consideration of the premises set forth above and the mutual covenants set forth below, the Parties hereto agree that the Existing Security Agreement is amended and restated in its entirety to read as follows:
SECTION 1 AMENDMENT
1.Definitions; Interpretation.Section 5.3 of the SHUTI Amended and Restated Agreement shall be amended as follows:
(a) The fifth sentence of Section 5.3 which states, “Any amounts due hereunder which are unpaid thirty (30) days after the end of the calendar quarter shall bear simple interest accrued at the annual rate of twelve percent (12%).” is replaced with the sentence, “Any amounts due hereunder which are unpaid sixty (60) days after the end of the calendar quarter shall bear simple interest accrued at the annual rate of twelve percent (12%).”
2.Terms Defined in Credit Agreement. All capitalized terms used in this Agreement (including inAll other terms and conditions of the Agreement remain in full force and effect for the recitals hereof) and not otherwise defined herein shall have the meanings assigned to them in the Creditterm of the Agreement.
(b) 3.Certain Defined Terms. As used in this Agreement, the following terms shall have the following meanings:
“Acceding Grantor” has the meaning set forth in Section 22.
“Accession Agreement” has the meaning set forth in Section 22.
“Agreement” has the meaning assigned to such term in the preamble hereto.
“Books” means all books, records and other written, electronic or other documentation in whatever form maintained now or hereafter by or for any Grantor in connection with the ownership of its assets or the conduct of its business or evidencing or containing information relating to the Collateral, including: (i) ledgers; (ii) records indicating, summarizing, or evidencing any Grantor’s assets (including Inventory and Rights to Payment), business operations or financial condition; (iii) computer programs and software; (iv) computer discs, tapes, files, manuals, spreadsheets; (v) computer printouts and output of whatever kind; (vi) any other computer prepared or electronically stored, collected or reported information and equipment of any kind; and (vii) any and all other rights now or hereafter arising out of any Contract between any Grantor and any service bureau, computer or data processing company or other Person charged with preparing or maintaining any of any Grantor’s books or records or with credit reporting, including with regard to any such Grantor’s Accounts.
“Borrower” has the meaning assigned to such term in the preamble hereto.
“Collateral” has the meaning provided in Section 2(a) hereof.
“Control Agreement” means any control agreement or other agreement with any securities intermediary, bank, depository or other Person establishing the Administrative Agent’s control with respect to any Deposit Accounts, Securities Accounts, lockboxes, disbursement accounts, investment accounts or similar accounts, Letter-of-Credit Rights or Investment Property, for purposes of Article 8 or Sections 9-104, 9-106 and 9-107 of the NY UCC.
“Credit Agreement” has the meaning assigned to such term in the first recital hereto.
“Excluded Equity” means Equity Interest in excess of 65% of the outstanding voting stock of each Excluded Subsidiary.
“Excluded Property” shall mean, with respect to a Grantor, (i) any application for registration of a Trademark filed on an intent-to-use basis, solely to the extent that the grant of a security interest in any such trademark application would materially adversely affect the validity or enforceability of the resulting trademark registration or result in cancellation of such trademark application; provided that immediately upon the time at which the consequences described in the foregoing clause (i) shall no longer exist, the Collateral shall include, and the applicable Grantor shall be deemed to have granted a security interest in, all of such Grantor’s right, title and interest in such Trademark; (ii) General Intangibles to which such Grantor is a party or under which such Grantor has any right, title or interest to the extent that such General Intangible contains a term or is subject to a rule of law, statute or regulation that restricts, prohibits, or requires a consent (that has not been obtained) of a Person (other than such Grantor) to, the creation, attachment or perfection of the security interest granted herein, and any such restriction, prohibition and/or requirement of consent is effective and enforceable under applicable law and is not rendered ineffective by applicable law (including, without limitation, pursuant to Sections 9-406, 9-407, 9-408 or 9-409 of the UCC), (iii) assets and property to the extent security interests therein are prohibited by enforceable provisions of contracts or applicable law or require governmental consent, approval, license or authorization that has not been obtained or, in the case of assets
consisting of licenses, permits, agreements or similar contracts, to the extent the grant of security therein or in such Grantor’s rights thereunder would violate the terms of such license, permit, agreement or similar contract relating to such asset (in each case, after giving effect to the applicable anti-assignment provisions of the UCC or other applicable law), (iv) any leasehold interest in real property (v) Excluded Equity, (vi) Excluded Accounts, (vii) any Equity Interests in partnerships, joint ventures or other entities that, in each case, are not Subsidiaries of the Grantors to the extent that such Equity Interests may not be pledged without the consent of one or more third parties (other than the Grantors or any of their Subsidiaries) pursuant to a binding contractual obligation after giving effect to the applicable anti-assignment provisions of the UCC or any other requirement of law, and (viii) motor vehicles and other assets subject to certificates of title, in each case other than to the extent a Lien thereon can be perfected by the filing of a financing statement under the UCC; provided, however, that in each of the foregoing cases Proceeds, Accounts and Payment Intangibles arising from the such items shall not constitute Excluded Property.
“Excluded Subsidiary” shall mean (i) any Foreign Subsidiary that is a “controlled foreign corporation” as defined in Section 957 of the Code, as amended and (ii) any Subsidiary substantially all the assets of which are Equity Interests in one or more Subsidiaries described in clause (i).
“Foreign Subsidiary” shall mean any Subsidiary that is not a “United States person” within the meaning of Section 7701(a)(30) of the Internal Revenue Code.
“Grantors” has the meaning set forth in the preamble hereto.
“Guarantee” has the meaning assigned to such term in the first recital hereto.
“Intellectual Property Collateral” means the following properties and assets owned or held by any Grantor or in which any Grantor otherwise has any interest, now existing or hereafter acquired or arising:
(i) all Patents, including domestic and foreign Patents, all licenses relating to any of the foregoing and all income and royalties with respect to any licenses (including such Patents and licenses described in Schedule 2), all rights to sue for past, present or future infringement thereof, all rights arising therefrom and pertaining thereto and all reissues, divisions, continuations, renewals, extensions and continuations-in-part thereof;
(ii) all Copyrights, including domestic and foreign Copyrights, together with underlying works of authorship (including titles), whether or not the underlying works of authorship have been published and whether said Copyrights are statutory or arise under the common law, and all other rights and works of authorship, all licenses relating to any of the foregoing and all income and royalties with respect to any licenses (including the Copyrights and licenses described in Schedule 2), and all income and royalties with respect thereto, and all other rights, Claims and demands in any way relating to any such Copyrights or works, including royalties and rights to sue for past, present or future infringement, and all rights of renewal and extension of such Copyright;
(iii) all Trademarks, including state (including common law), federal and foreign trademarks, service marks and trade names, and applications for registration of such Trademarks, all licenses relating to any of the foregoing and all income and royalties with respect to any licenses (including the Trademarks and licenses described in Schedule 2), whether registered or unregistered and wherever registered, all rights to sue for past, present or future infringement or unconsented use thereof, all rights arising therefrom and pertaining thereto and all reissues, extensions and renewals thereof, other than any “intent to use” application for registration of a Trademark filed pursuant to Section 1(b) of the Lanham Act, 15 U.S.C. § 1051, prior to the filing of a “Statement of Use” pursuant to Section 1(d) of the Lanham Act or an “Amendment to Allege Use” pursuant to Section 1(c) of the Lanham Act with respect thereto, to the extent that, and during the period in which, the grant of a security interest therein would impair the validity or enforceability of any registration that issues from such intent-to-use application under applicable federal law;
(iv) all Technical Information;
(v) the entire goodwill of or associated with the businesses now or hereafter conducted by such Grantor connected with and symbolized by any of the aforementioned properties and assets; and
(vi) all other Intellectual Property, all other proprietary rights, or other similar property and all other general intangibles associated with or arising out of any of the aforementioned properties and assets and not otherwise described above, including rights to sue for or collect damages for any past, present or future infringement of any of the foregoing.
“Intellectual Property Security Agreement” means each Copyright Security Agreement in substantially the form of Exhibit C, each Trademark Security Agreement in substantially the form of Exhibit D, each Patent Security Agreement in substantially the form of Exhibit E or any amendment thereto, in form and substance reasonably satisfactory to the Administrative Agent, supplementary to this Agreement and prepared for purposes of recordation with the U.S. Copyright Office or the U.S. Patent and Trademark Office, as applicable.
“Lender” has the meaning assigned to such term in the recital.
“Partnership and LLC Collateral” means any and all limited, limited liability and general partnership interests and limited liability company interests of any type or nature (including any such interests in Holdings’ direct or indirect Subsidiaries now or hereafter owned by any Grantor), whether now existing or hereafter acquired or arising, including any such interests specified in Schedule 3.
“Pledge Supplement” has the meaning specified in Section 3(h).
“Pledged Collateral” means any and all (i) Pledged Shares; (ii) additional capital stock or other Equity Interests of the direct or indirect Subsidiaries of Holdings, whether certificated or uncertificated, owned by any Grantor; (iii) other Investment Property of any Grantor; (iv) warrants, options or other rights entitling any Grantor to acquire any interest in Equity Interests or other securities of such Subsidiaries or any other Person; (v) Partnership and
LLC Collateral; (vi) Instruments; (vii) securities, property, interest, dividends and other payments and distributions issued as an addition to, in redemption of, in renewal or exchange for, in substitution or upon conversion of, or otherwise on account of, any of the foregoing; (viii) certificates and instruments now or hereafter representing or evidencing any of the foregoing; (ix) rights, interests and Claims with respect to the foregoing, including under any and all related agreements, instruments and other documents, and (x) cash and non-cash proceeds of any of the foregoing, in each case whether presently existing or owned or hereafter arising or acquired and wherever located, and as from time to time received or receivable by, or otherwise paid or distributed to or acquired by, any Grantor.
“Pledged Collateral Agreements” has the meaning specified in Section 5.
“Pledged Shares” means all of the issued and outstanding Equity Interests, whether certificated or uncertificated, of Holdings’ direct or indirect Subsidiaries now or hereafter owned by any Grantor, including each Subsidiary identified on Schedule 3 (as amended or supplemented from time to time).
“Proceeds Account” has the meaning set forth in Section 10(c).
“Rights to Payment” means any and all of any Grantor’s Accounts and any and all of any Grantor’s rights and Claims to the payment or receipt of money or other forms of consideration of any kind in, to and under or with respect to its Chattel Paper, Documents, General Intangibles, Instruments, Investment Property, Letter-of-Credit Rights, Proceeds and Supporting Obligations.
“Secured Obligations” means all Obligations, whether in respect of Indebtedness or other obligations of the Grantors to any Secured Party under the Credit Agreement, the Notes, the Guarantee or any of the other Loan Documents.
“Secured Parties” means the Administrative Agent and the Lenders.
“Supporting Obligations” means all supporting obligations, as such term is defined in Section 9-102 of the UCC.
“Threshold Amount” means $250,000.
(c) Terms Defined in the NY UCC. Where applicable and except as otherwise defined herein, terms used in this Agreement shall have the meanings assigned to them in the NY UCC; provided, however, that to the extent that the NY UCC is used to define any term herein and such term is defined differently in different Articles of the NY UCC, the definition of such term contained in Article 9 shall govern.
(d) Interpretation. The rules of interpretation set forth in Section 1.03 of the Credit Agreement shall be applicable to this Agreement and are incorporated herein by this reference.
SECTION 2 Security Interest.
(a) Grant of Security Interest. As security for the payment and performance of the Secured Obligations, each Grantor (1) hereby reaffirms the grant of security interest in the Collateral (as defined in the Existing Security Agreement) by each Grantor to the Administrative Agent pursuant to the Existing Security Agreement and (2) hereby grants to the Administrative Agent, for itself and on behalf of and for the ratable benefit of the other Secured Parties, a security interest in all of such Grantor’s right, title and interest in, to and under all of such Grantor’s personal property, wherever located and whether now existing or owned or hereafter acquired or arising, including the following property (collectively, the “Collateral”): (i) all Accounts; (ii) all Chattel Paper; (iii) all Commercial Tort Claims; (iv) all Deposit Accounts; (v) all Books and Documents; (vi) all Equipment; (vii) all General Intangibles; (viii) all Instruments; (ix) all Inventory; (x) all Investment Property; (xi) all Letter-of-Credit Rights; (xii) all other Goods; (xiii) all Intellectual Property Collateral; (xiv) all money; (xv) all Pledged Collateral; (xvi) all products and Proceeds of any and all of the foregoing; and (xvii) all Supporting Obligations of any and all of the foregoing; provided, however, that the Collateral shall not include Excluded Property.
(b) Grantors Remain Liable. Notwithstanding anything herein to the contrary, (i) each Grantor shall remain liable under any Contracts included in the Collateral, to the extent set forth therein, to perform all of its duties and obligations thereunder to the same extent as if this Agreement had not been executed, (ii) the exercise by the Administrative Agent of any of the rights granted to the Administrative Agent hereunder shall not release any Grantor from any of its duties or obligations under any such Contracts included in the Collateral, and (iii) neither the Administrative Agent nor any other Secured Party shall have any obligation or liability under any such Contracts included in the Collateral by reason of this Agreement, nor shall the Administrative Agent or any other Secured Party be obligated to perform any of the obligations or duties of any Grantor thereunder or to take any action to collect or enforce any such Contract included in the Collateral hereunder.
(c) Continuing Security Interest. Each Grantor agrees that this Agreement shall create a continuing security interest in the Collateral which shall remain in effect until terminated in accordance with Section 23.
SECTION 3 Perfection and Priority.
(a) Financing Statements, Etc. Each Grantor hereby authorizes the Administrative Agent (or its designee) to file at any time and from time to time any financing statements describing the Collateral, and each Grantor shall execute and deliver to the Administrative Agent, and each Grantor hereby authorizes the Administrative Agent (or its designee) to file (with or without such Grantor’s signature), at any time and from time to time, all amendments to financing statements, continuation financing statements, termination statements, Intellectual Property Security Agreements, assignments, fixture filings, affidavits, reports, notices and all other documents and instruments, in form reasonably satisfactory to the Administrative Agent, as the Administrative Agent may reasonably request, to perfect, continue the perfection of, maintain the priority of or provide notice of the Administrative Agent’s security interest in the Collateral and to accomplish the purposes of this Agreement. Without limiting the generality of the foregoing, each Grantor (i) ratifies the filing by the Administrative Agent of any financing
statements filed with respect to the Collateral prior to the date hereof and (ii) shall from time to time take the actions specified in subsections (b) through (i) below.
(b) Delivery of Pledged Collateral. Each Grantor shall, on the date hereof or promptly after becoming entitled to receive or receiving in accordance with Section 8.12 of the Credit Agreement, deliver to or for the account of the Administrative Agent, at the address and to the Person to be designated by the Administrative Agent, the certificates, instruments and other writings representing any Pledged Collateral, which shall be in suitable form for transfer by delivery and accompanied by duly executed instruments of transfer or assignment in blank, in form reasonably satisfactory to the Administrative Agent. If any Grantor becomes entitled to receive or receives certificates or instruments evidencing any Pledged Collateral after the date hereof, such Grantor shall hold it in trust for the Administrative Agent, segregate it from other property or funds of such Grantor and promptly deliver the same and all certificates, instruments and other writings representing such Pledged Collateral to or for the account of the Administrative Agent, at the address and to the Person to be designated by the Administrative Agent, which shall be in suitable form for transfer by delivery and accompanied by duly executed instruments of transfer or assignment in blank, in form reasonably satisfactory to the Administrative Agent.
(c) Transfer of Security Interest Other Than by Delivery. If for any reason Pledged Collateral cannot be delivered to or for the account of the Administrative Agent as provided in Section 3(b), each applicable Grantor shall promptly take such other steps as may be necessary or as shall be reasonably requested from time to time by the Administrative Agent to effect a transfer of a perfected first priority security interest in and pledge of the Pledged Collateral to the Administrative Agent for the ratable benefit of the Secured Parties pursuant to the NY UCC. Such Grantor shall deliver the Pledged Collateral to or for the account of the Administrative Agent as provided in Section 3(b) as soon as such Grantor becomes able to do so.
(d) Intellectual Property Collateral. (i) Each Grantor shall execute and deliver to the Administrative Agent, concurrently with the execution of this Agreement, such Intellectual Property Security Agreements as the Administrative Agent may reasonably request, and record or cause to be recorded (including by giving authorization to the Administrative Agent to so record) such Intellectual Property Security Agreements with the U.S. Patent and Trademark Office or the U.S. Copyright Office, as applicable, and take any such further or other action as may be necessary or as the Administrative Agent may reasonably request, to perfect the Administrative Agent’s security interest in such Intellectual Property Collateral with the U.S. Patent and Trademark Office or the U.S. Copyright Office or pursuant to the NY UCC, (ii) concurrently with the delivery of a Compliance Certificate pursuant to Section 8.01(d) of the Credit Agreement, for any Intellectual Property Collateral created or acquired by any Grantor after the date hereof which is registered or becomes registered or the subject of an application for registration with the U.S. Patent and Trademark Office or the U.S. Copyright Office, as applicable, such Grantor shall modify this Agreement by amending Schedule 2 to include any Intellectual Property Collateral which becomes part of the Collateral and which was not included on Schedule 2 as of the date hereof and record such Intellectual Property Security Agreement with the U.S. Patent and Trademark Office or the U.S. Copyright Office, as applicable, and take (or cause to be taken) such other action as may be necessary, or as the Administrative Agent may reasonably request, to perfect the Administrative Agent’s security interest in such Intellectual Property Collateral.
(e) Documents, Etc. Each Grantor shall deliver to the Administrative Agent, or an agent designated by it, appropriately endorsed or accompanied by appropriate instruments of transfer or assignment, all Documents and Chattel Paper, and all other Rights to Payment at any time evidenced by promissory notes, trade acceptances or other instruments, not already delivered hereunder pursuant to this Section 3, in each case for an amount in excess of the Threshold Amount (provided that in no event shall the aggregate amount of any such Documents Chattel Paper not delivered to the Administrative Agent exceed $500,000). Upon the reasonable request of the Administrative Agent, the Grantors shall mark all Documents and Chattel Paper with such legends as the Administrative Agent shall reasonably specify.
(f) Bailees. Any Person (other than the Administrative Agent) at any time and from time to time holding all or any portion of the Collateral shall be deemed to, and shall, hold the Collateral as the agent of, and as pledge holder for, the Administrative Agent. At any time and from time to time, the Administrative Agent may give notice to any such Person holding all or any portion of the Collateral that such Person is holding the Collateral as the agent and bailee of, and as pledge holder for, the Administrative Agent, and, in the case of Collateral having a value in excess of the Threshold Amount, obtain such Person’s written acknowledgment thereof, in form and substance reasonably satisfactory to the Administrative Agent. Without limiting the generality of the foregoing, each Grantor will, upon the Administrative Agent’s request, join with the Administrative Agent in notifying any Person who has possession of any Collateral of the Administrative Agent’s security interest therein and, in the case of Collateral having a value in excess of the Threshold Amount, obtaining an acknowledgment from such Person that it is holding the Collateral for the benefit of the Administrative Agent (provided that in no event shall the aggregate value of any such Collateral for which such Grantor has not obtained such acknowledgement exceed $500,000).
(g) Control. Each Grantor will cooperate with the Administrative Agent in obtaining control (as defined in the NY UCC) of or otherwise obtaining a perfected Lien in Collateral consisting of any Deposit Accounts (other than Excluded Accounts), Electronic Chattel Paper, Investment Property or Letter-of-Credit Rights, including delivery of Control Agreements in form and substance reasonably satisfactory to the Administrative Agent, as the Administrative Agent may reasonably request, to perfect, continue the perfection of, maintain the priority of or provide notice of the Administrative Agent’s security interest in such Collateral.
(h) After-Acquired Equity Interests. In the event that any Grantor acquires any Pledged Collateral after the date hereof, such Grantor shall deliver to the Administrative Agent a pledge supplement, duly executed by such Grantor and substantially in the form of Exhibit B (the “Pledge Supplement”), together with all schedules thereto, reflecting such additional Pledged Collateral, and the certificates and other documents required to be delivered pursuant to Section 3(b) hereof in respect of such additional Pledged Collateral. Notwithstanding the foregoing, it is understood and agreed that the security interest of the Administrative Agent shall attach to such Pledged Collateral immediately upon any Grantor’s acquisition of rights therein and shall not be affected by the failure of any Grantor to deliver a Pledge Supplement.
(i) Further Assurances Each Grantor agrees that, at its own expense, it will promptly execute and deliver all further instruments and documents and take all other
commercially reasonable actions as the Administrative Agent may reasonably request in order to perfect, preserve and protect any security interest granted or purported to be granted hereby or enable the Administrative Agent to exercise and enforce its rights and remedies hereunder with respect to any Collateral.
SECTION 4 Representations and Warranties. In addition to the representations and warranties of each Grantor set forth in the Credit Agreement, each Grantor represents and warrants to each Secured Party that:
(a) Location of Chief Executive Office and Collateral. Such Grantor’s chief executive office and principal place of business is located at the address set forth in Schedule 1, and all other locations where such Grantor owns or leases real property or Collateral with a value in excess of the Threshold Amount is kept are set forth in Schedule 1.
(b) Locations of Books. All locations where Books pertaining to the Rights to Payment of such Grantor are kept, including all equipment necessary for accessing such Books and the names and addresses of all service bureaus, computer or data processing companies and other Persons keeping any Books or collecting Rights to Payment for such Grantor, are set forth in Schedule 1.
(c) Jurisdiction of Organization and Names. Such Grantor’s jurisdiction of organization is set forth in Schedule 1; and such Grantor’s exact legal name is as set forth in the signature pages of this Agreement. All trade names and trade styles under which such Grantor presently conducts its business operations are set forth in Schedule 1, and, except as set forth in Schedule 1, such Grantor has not, at any time in the five years prior to the date of this Agreement: (i) been known as or used any other corporate, trade or fictitious name; or (ii) changed its nameAll terms capitalized herein maintain their definition as set forth in the Agreement.
(d) Collateral. Such Grantor has rights in or the power to transfer the Collateral, and such Grantor is the sole and complete owner of the Collateral (or, in the case of after-acquired Collateral, at the time such Grantor acquires rights in such Collateral, will be the sole and complete owner thereof), free from any Lien other than Permitted Liens.
(e) Enforceability; Priority of Security Interest. (i) This Agreement creates a security interest which is enforceable against the Collateral in which such Grantor now has rights and will create a security interest which is enforceable against the Collateral in which such Grantor hereafter acquires rights at the time such Grantor acquires any such rights; and (ii) the Administrative Agent has a perfected and first priority security interest in the Collateral in which such Grantor now has rights and will have a perfected and first priority security interest in the Collateral in which such Grantor hereafter acquires rights at the time such Grantor acquires any such rights, in each case, for the Administrative Agent’s own benefit and for the ratable benefit of the other Secured Parties, subject to Permitted Liens and securing the payment and performance of the Secured Obligations.
(f) Other Financing Statements. Other than (i) financing statements filed in connection with any Permitted Lien; (ii) financing statements in favor of the Administrative Agent for itself and on behalf of the other Secured Parties and (iii) precautionary financing statements filed in respect of leased, consigned or other third-party property, no effective financing statement
naming such Grantor as debtor, assignor, grantor, mortgagor, pledgor or the like and covering all or any part of the Collateral is on file in any filing or recording office in any jurisdiction.
(g) Rights to Payment. Such Grantor has not assigned any of its rights under any of its Rights to Payment except as provided in this Agreement or as set forth in the other Loan Documents.
(h) Inventory. No Inventory of such Grantor with a value in excess of the Threshold Amount is stored with any bailee, warehouseman or similar Person or on any premises leased to such Grantor, no such Inventory has been consigned to such Grantor or consigned by such Grantor to any Person, nor is any such Inventory held by such Grantor for any Person under any “bill and hold” or other arrangement, except as set forth in Schedule 1 or, after the date hereof, with any bailee at any other location that is duly disclosed to the Administrative Agent pursuant to Section 5(n).
(i) Intellectual Property. Except as set forth in Schedule 2, such Grantor does not own, co-own or use under any exclusive license agreement any Intellectual Property registered under the federal laws of the United States.
(j) Equipment. None of the Equipment that constitutes Collateral with a value in excess of the Threshold Amount is leased from or to any Person, except as set forth in Schedule 1 or as otherwise disclosed to the Administrative Agent and the Lenders.
(k) Deposit Accounts. The names and addresses of all financial institutions at which such Grantor maintains its Deposit Accounts (other than Excluded Accounts), and the account numbers and account names of such Deposit Accounts, are set forth in Schedule 1.
(l) Instrument Collateral
. (i) Such Grantor has not previously assigned any interest in any Instruments held by such Grantor (other than such interests as will be released on or before the date hereof), and (ii) no Person other than such Grantor owns an interest in such Instruments (whether as joint holders, participants or otherwise).
(m) Pledged Shares, Partnership and LLC Collateral and other Pledged Collateral
(i) All of the Pledged Shares and Partnership and LLC Collateral of such Grantor have been, and, upon issuance any additional Pledged Collateral consisting of Pledged Shares, Partnership and LLC Collateral or any other securities of such Grantor, will be, duly and validly issued, and are and will be fully paid and non-assessable, subject in the case of Partnership and LLC Collateral to future assessments required under applicable Law and any applicable partnership or operating agreement, (ii) such Grantor is or, in the case of any such additional Pledged Collateral, will be the legal record and beneficial owner thereof, (iii) in the case of Subsidiaries, there are no restrictions on the transferability of such Pledged Collateral or such additional Pledged Collateral to the Administrative Agent or with respect to the foreclosure, transfer or disposition thereof by the Administrative Agent, except as provided under applicable securities or “Blue Sky” laws, (iv) the Pledged Shares and Partnership and LLC Collateral of such Grantor constitute 100% of the issued and outstanding Equity Interests of all directly and indirectly owned Subsidiaries of such Grantor (other than with respect to Excluded Subsidiaries) and no securities convertible into or exchangeable for any Equity Interest of any such Subsidiary, or any
options, warrants or other commitments entitling any Person to purchase or otherwise acquire any Equity Interest of any such Subsidiary, are issued and outstanding, (v) any and all Pledged Collateral Agreements which affect or relate to the voting or giving of written consents with respect to any of the Pledged Shares pledged by such Grantor, and any and all other Pledged Collateral Agreements relating to the Partnership and LLC Collateral of such Grantor, have been disclosed in writing to the Administrative Agent and the Lenders, and (vi) as to each such Pledged Collateral Agreement relating to the Partnership and LLC Collateral of such Grantor, (A) such agreement contains the entire agreement between the parties thereto with respect to the subject matter thereof, has not been amended or modified, and is in full force and effect in accordance with its terms, (B) there exists no material violation or material default under any such agreement by such Grantor, and (C) such Grantor has not knowingly waived or released any of its material rights under or otherwise consented to a material departure from the terms and provisions of any such agreement.
(n) Other Investment Property; Instruments; and Chattel Paper. All Securities Accounts of such Grantor and other Investment Property of such Grantor are set forth in Schedule 1, and all Instruments and Chattel Paper for an amount in excess of the Threshold Amount held by such Grantor are also set forth in Schedule 1.
(o) Control Agreements. No Control Agreements exist with respect to any Collateral held by such Grantor other than any Control Agreements in favor of the Administrative Agent.
(p) Letter-of-Credit Rights. Such Grantor does not have any Letter-of-Credit Rights in excess of the Threshold Amount except as set forth in Schedule 1.
(q) Commercial Tort Claims. Such Grantor does not have any Commercial Tort Claims in excess of the Threshold Amount except as set forth in Schedule 1
(r) Leases. Such Grantor is not and will not become a lessee under any real property lease or other agreement governing the location of Collateral with a value in excess of the Threshold Amount at the premises of another Person pursuant to which the lessor or such other Person may obtain any rights in any of the Collateral, and no such lease or other such agreement now prohibits, restrains, impairs or will prohibit, restrain or impair such Grantor’s right to remove any Collateral from the premises at which such Collateral is situated, except for the usual and customary restrictions contained in such leases of real property.
SECTION 5 Covenants. Until the Commitments have expired or been terminated and all Secured Obligations (other than inchoate indemnification and expense reimbursement obligations for which no Claim has been made) have been indefeasibly paid in full in cash, each Grantor agrees that:
(a) Defense of Collateral. Such Grantor will appear in and defend any action, suit or proceeding which may affect to a material extent its title to, or right or interest in, or the Administrative Agent’s right or interest in, the Collateral.
(b) Preservation of Collateral. Such Grantor will do and perform all reasonable acts that may be necessary and appropriate to maintain, preserve and protect the Collateral.
(c) Compliance with Laws, Etc. Such Grantor will comply with all Laws and all policies of insurance, relating in a material way to the possession, operation, maintenance and control of the Collateral.
(d) Location of Books and Chief Executive Office. Such Grantor will: (i) keep all Books pertaining to the Rights to Payment of such Grantor at the locations set forth in Schedule 1 (or at such other locations as may be disclosed in writing to the Administrative Agent pursuant to the following clause (ii)); and (ii) give at least 15 days’ prior written notice to the Administrative Agent of (A) any changes in any location where Books pertaining to the Rights to Payment of such Grantor are kept, including any change of name or address of any service bureau, computer or data processing company or other Person preparing or maintaining any such Books or collecting Rights to Payment for such Grantor or (B) any changes in the location of such Grantor’s chief executive office or principal place of business.
(e) Location of Collateral. Such Grantor will: (i) keep the Collateral with a value in excess of the Threshold Amount held by such Grantor at the locations set forth in Schedule 1 (or, so long as no Default has occurred and is continuing, at such other locations as may be disclosed in writing to the Administrative Agent pursuant to the following clause (ii)) and will not remove any such Collateral from such locations (other than in connection with sales of Inventory in the ordinary course of such Grantor’s business, other dispositions permitted hereby or by the Credit Agreement and movements of Collateral from one disclosed location to another disclosed location within the United States), except upon at least 15 days’ prior written notice of any removal to the Administrative Agent; and (ii) give the Administrative Agent at least 15 days’ prior written notice of any change in the locations set forth in Schedule 1.
(f) Change in Name, Identity or Structure. Such Grantor will give at least 15 days’ prior written notice to the Administrative Agent of (i) any change in name, (ii) any change in its jurisdiction of organization, (iii) any change in its registration as an organization (or any new registration); and (iv) any changes in its identity or structure in any manner which might make any financing statement filed hereunder incorrect or misleading; provided that such Grantor shall not change its jurisdiction of organization to a jurisdiction outside of the United States.
(g) Maintenance of Records. Such Grantor will keep separate, accurate and complete Books with respect to the Collateral held by or owned by such Grantor, disclosing the Administrative Agent’s security interest hereunder.
(h) Disposition of Collateral. Such Grantor will not surrender or lose possession of, sell, lease, rent, or otherwise dispose of or transfer any of the Collateral having an aggregate value in excess of the Threshold Amount held or owned by such Grantor or any right or interest therein, except to the Administrative Agent or to the extent otherwise permitted by the Loan Documents; provided that no such disposition or transfer of Investment Property or Instruments shall be permitted if so directed by the Administrative Agent after an Event of Default has occurred and is continuing.
(i) Liens. Such Grantor will keep the Collateral held by such Grantor free of all Liens except Permitted Liens.
(j) Leased Premises; Collateral Held by Warehouseman, Bailee, Etc. At the Administrative Agent’s request, such Grantor will use commercially reasonable efforts to obtain from each Person from whom such Grantor leases any premises where any Collateral with a value in excess of the Threshold Amount is at any time maintained, and from each other Person at whose premises such Collateral is maintained (including any bailee, warehouseman or similar Person), any collateral access, subordination, landlord waiver, bailment, consent and estoppel agreements as the Administrative Agent may reasonably require, in form and substance reasonably satisfactory to the Administrative Agent.
(k) Rights to Payment. Such Grantor will:
(i)as may be required under the Credit Agreement, furnish to the Administrative Agent full and complete reports, in form and substance reasonably satisfactory to the Administrative Agent, with respect to the Accounts.
(ii)if any Accounts of such Grantor in excess of the Threshold Amount arise from Contracts with the United States or any department, agency or instrumentality thereof, promptly notify the Administrative Agent thereof and execute any documents and instruments and take any other steps reasonably requested by the Administrative Agent in order that all monies due and to become due thereunder shall be assigned to the Administrative Agent and notice thereof given to the Federal authorities under the Federal Assignment of Claims Act;
(iii)upon the request of the Administrative Agent upon the occurrence and during the continuance of an Event of Default, notify the account debtors and other obligors on the Rights to Payment or any designated portion thereof that payment shall be made directly to the Administrative Agent or to such other Person or location as the Administrative Agent shall specify; and
(iv)upon the occurrence and during the continuance of any Event of Default, establish such lockbox or similar arrangements for the payment of such Grantor’s Accounts and other Rights to Payment as the Administrative Agent shall require.
(l) Instruments, Investment Property, Etc. Upon the request of the Administrative Agent, such Grantor will (i) promptly deliver to the Administrative Agent, or an agent designated by it, appropriately endorsed or accompanied by appropriate instruments of transfer or assignment, all Instruments, Documents and Chattel Paper held by such Grantor in an amount in excess of the Threshold Amount, all letters of credit of such Grantor in an amount in excess of the Threshold Amount, and all other Rights to Payment held by such Grantor at any time evidenced by promissory notes, trade acceptances or other instruments in an amount in excess of the Threshold Amount, (ii) cause any securities intermediaries to show on their books that the Administrative Agent is the entitlement holder with respect to any Investment Property held by such securities intermediary on behalf of such Grantor, and/or obtain Control Agreements in favor of the Administrative Agent from such securities intermediaries, in form and substance reasonably satisfactory to the Administrative Agent, with respect to any such Investment Property, as
reasonably requested by the Administrative Agent, and (iii) provide such notice, obtain such acknowledgments and take all such other action, with respect to any Chattel Paper, Documents and Letter-of-Credit Rights held by such Grantor in an amount in excess of the Threshold Amount, as the Administrative Agent shall reasonably specify.
(l) Deposit Accounts and Securities Accounts. Such Grantor will give the Administrative Agent notice of the establishment of any new Deposit Account (other than any Excluded Account) and of any new Securities Account established by such Grantor with respect to any Investment Property held by such Grantor.
(m) Inventory. Except as provided for herein, such Grantor will not store any Inventory with an aggregate value in excess of the Threshold Amount with a bailee, warehouseman or similar Person or on premises leased to such Grantor other than those locations identified in Schedule 1, and will not dispose of any such Inventory on a bill-and-hold, guaranteed sale, sale and return, sale on approval, consignment or similar basis, and will not acquire any such Inventory from any Person on any such basis.
(n) Intellectual Property Collateral. Such Grantor:
(i)except as permitted under the Credit Agreement, will not allow or suffer any Intellectual Property Collateral that constitutes Material Intellectual Property held by such Grantor to become abandoned, nor any registration thereof to be terminated, forfeited, expired or dedicated to the public, except as shall be reasonable and appropriate in accordance with prudent business practice; and
(ii)will diligently prosecute all applications for Patents and Trademarks, and file and prosecute any and all continuations, continuations-in-part, applications for reissue, applications for certificate of correction and like matters as shall be reasonable and appropriate in accordance with prudent business practice, and promptly and timely pay any and all maintenance, license, registration and other fees, taxes and expenses incurred in connection with any Intellectual Property Collateral held by such Grantor.
(p) Notices, Reports and Information. Such Grantor will (i) promptly notify the Administrative Agent of any other modifications of or additions to the information contained in Schedule 1 (including any acquisition or holding of an interest in any Chattel Paper, Commercial Tort Claims and Letter-of-Credit Rights); (ii) promptly notify the Administrative Agent of any material Claim made or asserted against the Collateral by any Person and of any material change in the composition of the Collateral or other event which could materially adversely affect the value of the Collateral or the Administrative Agent’s Lien thereon; (iii) promptly furnish to the Administrative Agent such listings, descriptions and schedules with respect to such Grantor’s Equipment and Inventory, and such other reports and other information in connection with the Collateral, as the Administrative Agent may reasonably request, all in reasonable detail; and (iv) upon the reasonable request of the Administrative Agent make such demands and requests for information and reports as such Grantor is entitled to make in respect of the Collateral.
(q) Shareholder Agreements and Other Agreements. Such Grantor will:
(i)comply in all material respects with all of its obligations under any shareholders agreement, operating agreement, partnership agreement, voting trust, proxy agreement or other agreement or understanding (collectively, the “Pledged Collateral Agreements”) to which it is a party and shall enforce all of its rights thereunder;
(ii)take all actions necessary to cause each such Pledged Collateral Agreement relating to Partnership and LLC Collateral of Subsidiaries to provide specifically at all times that: (A) no such Partnership and LLC Collateral shall be a security governed by Article 8 of the applicable UCC; and (B) no consent of any member, manager, partner or other Person shall be a condition to the admission as a member or partner of any transferee (including the Administrative Agent) that acquires ownership of such Partnership and LLC Collateral as a result of the exercise by the Administrative Agent of any remedy hereunder or under applicable Law;
(iii)not take vote or enable to take any other action to certificate any Pledged Shares or Partnership and LLC Collateral of any Grantor to the extent such Pledged Shares or Partnership and LLC Collateral is not certificated as of the Closing Date, unless such Grantor delivers a certificate evidencing such Equity Interest to the Administrative Agent in accordance with this Agreement; and
(iv)not vote to enable or take any other action to: (A) amend or terminate, or waive compliance with any of the terms of, any such Pledged Collateral Agreement, certificate or articles of incorporation, bylaws or other organizational documents in any way that materially changes the rights of such Grantor with respect to any such Pledged Collateral in a manner adverse to the Administrative Agent or the other Secured Parties or that adversely affects the validity, perfection or priority of the Administrative Agent’s security interest therein.
(v)Additionally, such Grantor agrees that no such Partnership and LLC Collateral of Subsidiaries (A) shall be dealt in or traded on any securities exchange or in any securities market, (B) shall constitute an investment company security, or (C) shall be held by such Grantor in a Securities Account.
(r) Insurance
(i)Such Grantor shall carry and maintain in full force and effect, at the expense of such Grantor and with financially sound and reputable insurance companies, insurance with respect to the Collateral held by such Grantor in such amounts, with such deductibles and covering such risks in accordance with Section 8.05 of the Credit Agreement.
(ii)If Collateral having a value in excess of the Threshold Amount held by such Grantor is materially damaged or destroyed, in whole or in part, by fire or other casualty, such Grantor shall give immediate notice thereof to the Administrative Agent. After the occurrence and during the continuance of an Event of Default, or as otherwise required under the Loan Documents, all sums payable to any Grantor by any insurer with respect to a casualty relating to all or any part of the Collateral shall be paid to the Administrative Agent. If any Grantor receives any insurance proceeds which are to be paid to the Administrative Agent pursuant to the previous sentence, such Grantor shall hold such proceeds in trust for the Administrative Agent, segregate such proceeds from other funds of such Grantor, and promptly forward such proceeds in the form received to the Administrative Agent (appropriately endorsed
by such Grantor to the order of the Administrative Agent or in such other manner as satisfactory to the Administrative Agent). All such insurance proceeds may be retained by the Administrative Agent as part of Collateral hereunder and held in the Proceeds Account, applied by the Administrative Agent toward payment of all or part of the Secured Obligations in such order as is provided herein, or released to such Grantor upon its request with the consent of the Administrative Agent.
SECTION 6 Rights to Payment and Pledged Collateral.
(a) Collection of Rights to Payment. Until the Administrative Agent exercises its rights hereunder to collect any Rights to Payment of any Grantor, each such Grantor shall endeavor in the first instance diligently to collect all amounts due or to become due on or with respect to the Rights to Payment held by such Grantor. At the request of the Administrative Agent, upon the occurrence and during the continuance of any Event of Default, all remittances received by such Grantor shall be held in trust for the Administrative Agent and, in accordance with the Administrative Agent’s instructions, remitted to the Administrative Agent or deposited to an account with the Administrative Agent in the form received (with any necessary endorsements or instruments of assignment or transfer).
(b) Pledged Collateral. Unless and until an Event of Default shall have occurred and be continuing, each Grantor shall be entitled to receive and retain for its own account any cash dividend on or other cash distribution or payment, if any, in respect of the Pledged Collateral, to the extent consistent with the Credit Agreement or the Guarantee, as applicable; provided, however, that, except in connection with transactions permitted under Section 9.09 or Section 9.05 of the Credit Agreement, such Grantor shall not be entitled to receive (i) cash paid, payable or otherwise distributed in redemption of, or in exchange for or in substitution of, any Pledged Collateral held by such Grantor, or (ii) dividends and other distributions paid or payable in cash in respect of any such Pledged Collateral in connection with a partial or total liquidation or dissolution of any Person whose ownership interests constitute Pledged Collateral or in connection with a reduction of capital, capital surplus or paid-in-surplus or any other type of recapitalization involving any such Person. At the request of the Administrative Agent, upon the occurrence and during the continuance of any Event of Default, the Administrative Agent shall be entitled to receive all distributions and payments of any nature with respect to any Pledged Collateral, and all such distributions or payments received by such Grantor shall be held in trust for the Administrative Agent and, in accordance with the Administrative Agent’s instructions, remitted to the Administrative Agent or deposited to an account with the Administrative Agent in the form received (with any necessary endorsements or instruments of assignment or transfer). Following the occurrence and during the continuance of an Event of Default any such distributions and payments with respect to any such Pledged Collateral held in any Securities Account shall be held and retained in such Securities Account, in each case as part of the Collateral hereunder. Additionally, the Administrative Agent shall have the right, upon the occurrence and during the continuance of an Event of Default, following prior written notice to any applicable Grantor, to vote and to give consents, ratifications and waivers with respect to any Pledged Collateral held by such Grantor, and to exercise all rights of conversion, exchange, subscription or any other rights, privileges or options pertaining thereto, as if the Administrative Agent were the absolute owner thereof; provided that the Administrative Agent shall have no duty to exercise any of the foregoing
rights afforded to it and shall not be responsible to such Grantor or any other Person for any failure to do so or delay in doing so.
(c) Voting Prior to an Event of Default. Unless and until an Event of Default shall have occurred and is continuing, each Grantor shall have the right to vote the Pledged Collateral held by such Grantor and to give consents, ratifications and waivers in respect thereof, and shall retain the power to control the direction, management and policies of any Person comprising such Pledged Collateral to the same extent as such Grantor would if such Pledged Collateral were not pledged to the Administrative Agent pursuant to this Agreement; provided, however, that no vote shall be cast or consent, waiver or ratification given or action taken which would have the effect of materially impairing the position or interest of the Administrative Agent and the other Secured Parties in respect of such Pledged Collateral or which would alter the voting rights with respect to the stock or other ownership interest in or of any such Person or be inconsistent with or violate any provision of this Agreement, the Credit Agreement, or any other Loan Documents. If applicable, such Grantor shall be deemed the beneficial owner of all such Pledged Collateral for purposes of Sections 13 and 16 of the Exchange Act and agrees to file all reports required to be filed by beneficial owners of securities thereunder. The Administrative Agent shall execute and deliver (or cause to be executed and delivered) to each Grantor all such proxies and other instruments as such Grantor may reasonably request for the purpose of enabling such Grantor to exercise the voting and other rights which it is entitled to exercise pursuant to this subsection (c) and to receive the distributions which it is authorized to receive and retain pursuant to this subsection (c).
(d) Certain Other Administrative Matters. The Administrative Agent may cause any of the Pledged Collateral to be transferred into its name or into the name of its nominee or nominees (subject to the revocable rights specified in this Section 6). The Administrative Agent shall at all times have the right to exchange uncertificated Pledged Collateral for certificated Pledged Collateral, and to exchange certificated Pledged Collateral for certificates of larger or smaller denominations, for any purpose consistent with this Agreement.
SECTION 7 Authorization; Administrative Agent Appointed Attorney-in-Fact. In addition to (and not in limitation of) any other right or remedy provided to the Administrative Agent hereunder, the Administrative Agent shall have the right to, in the name of any Grantor, or in the name of the Administrative Agent or otherwise, without notice to or assent by any such Grantor, and each Grantor hereby constitutes and appoints the Administrative Agent (and any of the Administrative Agent’s officers or employees or agents designated by the Administrative Agent) as such Grantor’s true and lawful attorney-in-fact, with full power and authority to:
(a) file any of the financing statements which must be filed to perfect or continue to perfect, maintain the priority of or provide notice of the Administrative Agent’s Lien in the Collateral;
(b) take possession of and endorse any notes, acceptances, checks, drafts, money orders or other forms of payment or security and collect any Proceeds of any Collateral;
(c) sign and endorse any invoice or bill of lading relating to any of the Collateral, warehouse or storage receipts, drafts against customers or other obligors, assignments, notices of assignment, verifications and notices to customers or other obligors;
(d) notify the U.S. Postal Service and other postal authorities to change the address for delivery of mail addressed to such Grantor to such address as the Administrative Agent may designate; and, without limiting the generality of the foregoing, establish with any Person lockbox or similar arrangements for the payment of the Rights to Payment of such Grantor;
(e) receive, open and dispose of all mail addressed to such Grantor;
(f) send requests for verification of Rights to Payment to the customers or other obligors of such Grantor;
(g) contact, or direct such Grantor to contact, all account debtors and other obligors on the Rights to Payment of such Grantor and instruct such account debtors and other obligors to make all payments directly to the Administrative Agent;
(h) assert, adjust, sue for, compromise or release any Claims under any policies of insurance;
(i) exercise dominion and control over, and refuse to permit further withdrawals from, any Deposit Accounts of such Grantor maintained with the Administrative Agent, any Lender or any other bank, financial institution or other Person;
(j) notify each Person maintaining lockbox or similar arrangements for the payment of the Rights to Payment of such Grantor to remit all amounts representing collections on such Rights to Payment directly to the Administrative Agent;
(k) ask, demand, collect, receive and give acquittances and receipts for any and all Rights to Payment of such Grantor, enforce payment or any other rights in respect of the Rights to Payment and other Collateral, grant consents, agree to any amendments, modifications or waivers of the agreements and documents governing such Rights to Payment and other Collateral, and otherwise file any Claims, take any action or institute, defend, settle or adjust any actions, suits or proceedings with respect to the Collateral, as the Administrative Agent may deem necessary or desirable to maintain, preserve and protect the Collateral, to collect the Collateral or to enforce the rights of the Administrative Agent with respect to the Collateral;
(l) execute any and all applications, documents, papers and instruments necessary for the Administrative Agent to use the Intellectual Property Collateral and grant or issue any exclusive or non-exclusive license or sublicense with respect to any Intellectual Property Collateral;
(m) execute any and all endorsements, assignments or other documents and instruments necessary to sell, lease, assign, convey or otherwise transfer title in or dispose of the Collateral;
(n) execute and deliver to any securities intermediary or other Person any entitlement order or other notice, document or instrument which the Administrative Agent may deem necessary or advisable to maintain, protect, realize upon and preserve the Deposit Accounts and Investment Property of such Grantor and the Administrative Agent’s security interest therein; and
(o) execute any and all such other documents and instruments, and do any and all acts and things for and on behalf of such Grantor, which the Administrative Agent may deem necessary or advisable to maintain, protect, realize upon and preserve the Collateral and the Administrative Agent’s security interest therein and to accomplish the purposes of this Agreement.
The Administrative Agent agrees that, except upon the occurrence and during the continuation of an Event of Default, it shall not exercise the power of attorney, or any rights granted to the Administrative Agent, pursuant to clauses (b) through (o). The foregoing power of attorney is coupled with an interest and irrevocable so long as the Lenders have any Commitments or the Secured Obligations (other than inchoate indemnification and expense reimbursement obligations for which no Claim has been made) have not been indefeasibly paid in full in cash. Each Grantor hereby ratifies, to the extent permitted by applicable Law, all that the Administrative Agent shall lawfully and in good faith do or cause to be done by virtue of and in compliance with this Section 7.
SECTION 8 Administrative Agent Performance of Grantor Obligations. The Administrative Agent may perform or pay any obligation which any Grantor has agreed to perform or pay under or in connection with this Agreement, and such Grantor shall reimburse the Administrative Agent on demand for any amounts paid, or costs incurred, by the Administrative Agent pursuant to this Section 8.
SECTION 9 Administrative Agent’s Duties. Notwithstanding any provision contained in this Agreement, the Administrative Agent shall have no duty to exercise any of the rights, privileges or powers afforded to it and shall not be responsible to any Grantor or any other Person for any failure to do so or delay in doing so. Beyond the exercise of reasonable care to assure the safe custody of Collateral in the Administrative Agent’s possession and the accounting for moneys actually received by the Administrative Agent hereunder, the Administrative Agent shall have no duty or liability to exercise or preserve any rights, privileges or powers pertaining to the Collateral. The Administrative shall not deliver any notice of exclusive control under a Control Agreement with respect to any Collateral unless an Event of Default has occurred and is continuing.
SECTION 10 Remedies.
(a) Remedies. Upon the occurrence and during the continuation of any Event of Default, the Administrative Agent shall have, in addition to all other rights and remedies granted to it in this Agreement, the Credit Agreement, the Guarantee or any other Loan Document, all rights and remedies of a secured party under the NY UCC and other applicable Laws. Without limiting the generality of the foregoing, each Grantor agrees that:
(i)The Administrative Agent may peaceably and without notice enter any premises of such Grantor, take possession of any Collateral, remove or dispose of all or part of the
Collateral on any premises of such Grantor or elsewhere, or, in the case of Equipment, render it nonfunctional, and otherwise collect, receive, appropriate and realize upon all or any part of the Collateral, and demand, give receipt for, settle, renew, extend, exchange, compromise, adjust, or sue for all or any part of the Collateral, as the Administrative Agent may determine.
(i)The Administrative Agent may require such Grantor to assemble all or any part of the Collateral and make it available to the Administrative Agent, at any place and time designated by the Administrative Agent.
(ii)The Administrative Agent may use or transfer any of such Grantor’s rights and interests in any Intellectual Property Collateral, by license, by sublicense (to the extent permitted by an applicable license) or otherwise, on such conditions and in such manner as the Administrative Agent may determine.
(iii)The Administrative Agent may secure the appointment of a receiver of the Collateral or any part thereof (to the extent and in the manner provided by applicable Law).
(iv)The Administrative Agent may withdraw (or cause to be withdrawn) any and all funds from any Deposit Accounts (other than Excluded Accounts), Securities Accounts or Commodity Accounts.
(v)The Administrative Agent may sell, resell, lease, use, assign, transfer or otherwise dispose of any or all of the Collateral in its then condition or following any commercially reasonable preparation or processing (utilizing in connection therewith any of such Grantor’s assets, without charge or liability to the Administrative Agent therefor) at public or private sale, by one or more Contracts, in one or more parcels, at the same or different times, for cash or credit or for future delivery without assumption of any credit risk, all as the Administrative Agent deems advisable; provided, however, that such Grantor shall be credited with the net proceeds of sale only when such proceeds are finally collected by the Administrative Agent. The Administrative Agent and each of the other Secured Parties shall have the right upon any such public sale, and, to the extent permitted by applicable Law, upon any such private sale, to purchase the whole or any part of the Collateral so sold, free of any right or equity of redemption, which right or equity of redemption such Grantor hereby releases, to the extent permitted by applicable Law. The Administrative Agent shall give such Grantor such notice of any public or private sale as may be required by the NY UCC or other applicable Law. Such Grantor recognizes that the Administrative Agent may be unable to make a public sale of any or all of the Pledged Collateral, by reason of prohibitions contained in applicable securities laws or otherwise, and expressly agrees that a private sale to a restricted group of purchasers for investment and not with a view to any distribution thereof shall be considered a commercially reasonable sale.
(vii) Neither the Administrative Agent nor any other Secured Party shall have any obligation to clean up or otherwise prepare the Collateral for sale. The Administrative Agent has no obligation to attempt to satisfy the Secured Obligations by collecting them from any other Person liable for them and the Administrative Agent and the other Secured Parties may release, modify or waive any Collateral provided by any other Person to secure any of the Secured Obligations, all without affecting the Administrative Agent’s or any other Secured Party’s rights
against such Grantor. Such Grantor waives any right it may have to require the Administrative Agent or any other Secured Party to pursue any third Person for any of the Secured Obligations. The Administrative Agent and the other Secured Parties may comply with any applicable state or federal law requirements in connection with a disposition of the Collateral and compliance will not be considered adversely to affect the commercial reasonableness of any sale of the Collateral. The Administrative Agent may sell the Collateral without giving any warranties as to the Collateral. The Administrative Agent may specifically disclaim any warranties of title or the like. This procedure will not be considered adversely to affect the commercial reasonableness of any sale of the Collateral. If the Administrative Agent sells any of the Collateral upon credit, such Grantor will be credited only with payments actually made by the purchaser, received by the Administrative Agent and applied to the indebtedness of the purchaser. In the event the purchaser fails to pay for the Collateral, the Administrative Agent may resell the Collateral and the Grantors shall be credited with the proceeds of the sale.
(b) License. For the purpose of enabling the Administrative Agent to exercise its rights and remedies under this Section 10 or otherwise in connection with this Agreement, each Grantor hereby grants to the Administrative Agent an irrevocable, non-exclusive and assignable license (exercisable without payment or royalty or other compensation to such Grantor) to use, license or sublicense any Intellectual Property Collateral.
(c) Application of Proceeds. The cash proceeds actually received from the sale or other disposition or collection of any Grantor’s Collateral, and any other amounts received in respect of such Collateral the application of which is not otherwise provided for herein, shall be applied as provided in Section 3.03(d) of the Credit Agreement. Any surplus thereof which exists after payment and performance in full of the Secured Obligations shall be promptly paid over to such Grantor or otherwise disposed of in accordance with the NY UCC or other applicable Law. Each Grantor shall remain liable to the Administrative Agent and the other Secured Parties for any deficiency which exists after any sale or other disposition or collection of Collateral.
SECTION 11 Certain Waivers. Each Grantor waives, to the fullest extent permitted by applicable Law, (a) any right of redemption with respect to the Collateral, whether before or after sale hereunder, and all rights, if any, of marshalling of the Collateral or other collateral or security for the Secured Obligations; (b) any right to require the Administrative Agent or the other Secured Parties (i) to proceed against any Person, (ii) to exhaust any other collateral or security for any of the Secured Obligations, (iii) to pursue any remedy in the Administrative Agent’s or any of the other Secured Parties’ power, or (iv) to make or give any presentments, demands for performance, notices of nonperformance, protests, notices of protests or notices of dishonor in connection with any of the Collateral; and (c) all Claims, damages, and demands against the Administrative Agent or the other Secured Parties arising out of the repossession, retention, sale or application of the proceeds of any sale of the Collateral.
SECTION 12 Notices. All notices or other communications hereunder shall be given in the manner and to the addresses specified in Section 14.02 of the Credit Agreement.
SECTION 13 No Waiver; Cumulative Remedies. No failure on the part of the Administrative Agent or any other Secured Party to exercise, and no delay in exercising, any right,
remedy, power or privilege hereunder shall operate as a waiver thereof, nor shall any single or partial exercise of any such right, remedy, power or privilege preclude any other or further exercise thereof or the exercise of any other right, remedy, power or privilege. The rights and remedies under this Agreement are cumulative and not exclusive of any rights, remedies, powers and privileges that may otherwise be available to the Administrative Agent or any other Secured Party.
SECTION 14 Binding Effect. This Agreement shall be binding upon, inure to the benefit of and be enforceable by each Grantor, the Administrative Agent, each Secured Party and their respective successors and permitted assigns and shall bind any Person who becomes bound as a debtor, agent or secured party to this Agreement.
SECTION 15 Governing Law. This Agreement and the rights and obligations of the parties hereunder shall be governed by, and construed in accordance with, the law of the State of New York, without regard to principles of conflicts of laws that would result in the application of the laws of any other jurisdiction; provided that Section 5-1401 of the New York General Obligations Law shall apply.
SECTION 16 Submission to Jurisdiction
(a) . Each Grantor agrees that any suit, action or proceeding with respect to this Agreement or any judgment entered by any court in respect thereof may be brought initially in the federal or state courts in New York, New York or in the courts of its own corporate domicile and irrevocably submits to the non-exclusive jurisdiction of each such court for the purpose of any such suit, action, proceeding or judgment. This Section 16 is for the benefit of the Secured Parties only and, as a result, no Secured Party shall be prevented from taking proceedings in any other courts with jurisdiction. To the extent allowed by any Law, the Secured Parties may take concurrent proceedings in any number of jurisdictions.
(b) Waiver of Venue. Each Grantor irrevocably waives to the fullest extent permitted by applicable Law any objection that it may now or hereafter have to the laying of the venue of any suit, action or proceeding arising out of or relating to this Agreement and hereby further irrevocably waives to the fullest extent permitted by applicable Law any Claim that any such suit, action or proceeding brought in any such court has been brought in an inconvenient forum. A final judgment (in respect of which time for all appeals has elapsed) in any such suit, action or proceeding shall be conclusive and may be enforced in any court to the jurisdiction of which such Obligor is or may be subject, by suit upon judgment.
(c) Service of Process. Each party hereto irrevocably consents to service of process in the manner provided for notices in Section 12. Nothing in this Agreement will affect the right of any party hereto to serve process in any other manner permitted by applicable Law.
SECTION 17 Waiver of Jury Trial. EACH PARTY HERETO HEREBY IRREVOCABLY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY SUIT, ACTION OR PROCEEDING ARISING OUT OF OR RELATING TO THIS AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY OR THEREBY.
SECTION 18 Entire Agreement; Amendment. This Agreement and the other Loan Documents constitute the entire agreement among the parties with respect to the subject
matter hereof and thereof and supersede any and all previous agreements and understandings, oral or written, relating to the subject matter hereof, including any confidentiality (or similar) agreements. Each Grantor acknowledges, represents and warrants that in deciding to enter into this Agreement and the other Loan Documents and in taking or not taking any action hereunder or thereunder, it has not relied, and will not rely, on any statement, representation, warranty, covenant, agreement or understanding, whether written or oral, of or with the Lenders other than those expressly set forth in this Agreement and the other Loan Documents. This Agreement shall not be amended except by the written agreement of the parties as provided in the Credit Agreement.
SECTION 19 Severability. If any provision hereof is found by a court to be invalid or unenforceable, to the fullest extent permitted by any Law the parties agree that such invalidity or unenforceability shall not impair the validity or enforceability of any other provision hereof.
SECTION 20 Counterparts. This Agreement may be executed in any number of counterparts, all of which taken together shall constitute one and the same instrument and any of the parties hereto may execute this Agreement by signing any such counterpart. Delivery of an executed signature page of this Agreement by facsimile transmission or electronic transmission (in PDF format) shall be effective as delivery of a manually executed counterpart hereof.
SECTION 21 No Inconsistent Requirements. Each Grantor acknowledges that this Agreement and the other Loan Documents may contain covenants and other terms and provisions variously stated regarding the same or similar matters, and agrees that all such covenants, terms and provisions are cumulative and all shall be performed and satisfied in accordance with their respective terms.
SECTION 22 Accession. At such time following the date hereof as any Person (an “Acceding Grantor”) is required to accede hereto pursuant to the terms of Section 8.12 of the Credit Agreement, such Acceding Grantor shall execute and deliver to the Administrative Agent an accession agreement substantially in the form of Exhibit A (an “Accession Agreement”), signifying its agreement to be bound by the provisions of this Agreement as a Grantor to the same extent as if such Acceding Grantor had originally executed this Agreement as of the date hereof.
SECTION 23 Termination. Upon the termination of the Commitments of the Lenders and payment and performance in full in cash of all Secured Obligations (other than Warrant Obligations and inchoate indemnification and expense reimbursement obligations for which no Claim has been made), the security interests created by this Agreement shall terminate and the Administrative Agent shall promptly execute and deliver to each Grantor such documents and instruments reasonably requested by such Grantor as shall be necessary to evidence the termination of all security interests given by such Grantor to the Administrative Agent hereunder.
SECTION 24 Effect of Amendment and Restatement. This Agreement amends and restates the Existing Security Agreement but does not constitute and, is not intended to create, a novation or accord and satisfaction. All security interests and other Liens granted or conveyed with respect to the Collateral (as defined in the Existing Security Agreement) pursuant to the Existing Security Agreement shall continue in full force and effect and shall constitute Collateral hereunder, and nothing in this Agreement shall be construed to constitute a termination, release or extinguishment of any Lien in favor of the Administrative Agent that was in effect immediately prior to the date hereof.
[Remainder of page intentionally left blank; signature pages follow]
IN WITNESS WHEREOF, the parties hereto have duly executed this Agreement as of the date first above written.
| | | | | | | | |
| GRANTORS: |
| | |
| PEAR THERAPEUTICS (US), INC. |
| | |
| | |
| By: | /s/ Ronan O'Brien |
| Name: | Ronan O'Brien |
| Title: | General Counsel & Chief Compliance |
| | Officer |
| | | | | | | | |
| PEAR THERAPEUTICS, INC. |
| | |
| | |
| By: | /s/ Ronan O'Brien |
| Name: | Ronan O'Brien |
| Title: | Secretary |
[Signature Page to Security Agreement]
| | | | | | | | |
| ADMINISTRATIVE AGENT: |
| | |
| PERCEPTIVE CREDIT HOLDINGS III, LP |
| | |
| By: PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner |
| | |
| | |
| By: | /s/ Sandeep Dixit |
| Name: Sandeep Dixit |
| Title: Chief Credit Officer |
| | |
| | |
| By: | /s/ Sandeep Dixit |
| Name: | Sam Chawla |
| Title: | Portfolio Manager |
[Signature Page to Security Agreement]
EXHIBIT A
TO THE SECURITY AGREEMENT
FORM OF ACCESSION AGREEMENT
| | | | | |
| To: | Perceptive Credit Holdings III, LP, as Administrative Agent |
| |
| Re: | PEAR THERAPEUTICS, INC. |
Ladies and Gentlemen:
This Accession Agreement is made and delivered as of [__________], 20[__] pursuant to Section 22 of that certain Amended and Restated Security Agreement, dated as of March [_], 2022 (as amended or otherwise modified from time to time, the “Security Agreement”), between each Grantor from time to time party thereto (each a “Grantor” and collectively, the “Grantors”), and Perceptive Credit Holdings III, LP (in such capacity, together with its successors and assigns, the “Administrative Agent”). All capitalized terms used in this Accession Agreement and not otherwise defined herein shall have the meanings assigned to them in either the Security Agreement or the Credit Agreement (as defined in the Security Agreement), as the context may require.
The undersigned, ___________________________ [insert name of Acceding Grantor], a _____________________ [corporation, partnership, limited liability company, etc.], hereby acknowledges for the benefit of the Secured Parties that it shall be a “Grantor” for all purposes of the Security Agreement effective from the date hereof. The undersigned confirms that the representations and warranties set forth in Section 4 of the Security Agreement are true and correct as to the undersigned as of the date hereof.
Without limiting the foregoing, the undersigned hereby agrees to perform all of the obligations of a Grantor under, and to be bound in all respects by the terms of, the Security Agreement, including Section 5 thereof, to the same extent and with the same force and effect as if the undersigned were an original signatory thereto. The undersigned hereby grants to the Administrative Agent, for itself and on behalf of and for the ratable benefit of the other Secured Parties, a security interest in all of the undersigned’s right, title and interest in, to and under all of its personal property, wherever located and whether now existing or owned or hereafter acquired or arising, including all Collateral, as security for the payment and performance of the Secured Obligations.
Schedules 1 through 3 to the Security Agreement are hereby amended by adding Schedules 1 through 3 attached hereto to the Security Agreement. [Attach hereto completed Schedules 1 through 3 in the form of Schedules 1 through 3 attached to the Security Agreement.]
This Accession Agreement shall constitute a Loan Document under the Credit Agreement.
THIS ACCESSION AGREEMENT SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH, THE LAW OF THE STATE OF NEW YORK.
IN WITNESS WHEREOF, the undersigned hasThe Balance of This Page Intentionally Left Blank – Signature Page to Follow)
IN WITNESS WHEREOF, the Parties hereto have caused this First Amendment to be duly executed this Accession Agreementin duplicate counterparts, each of which shall be deemed to constitute an original, effective as of the date first above writtenFirst Amendment Date.
| | | | | |
[ACCEDING GRANTOR] |
| By: | |
| Name: |
| Title: |
| | | | | | | | | | | |
| Address: | |
| | | |
| | | |
| | | |
Email: | |
| Fax No.: | |
| Attention: | |
EXHIBIT B
TO THE SECURITY AGREEMENT
FORM OF PLEDGE SUPPLEMENT
| | | | | |
| To: | Perceptive Credit Holdings III, LP, as Administrative Agent |
| |
| Re: | PEAR THERAPEUTICS, INC. |
Ladies and Gentlemen:
This Pledge Supplement (this “Pledge Supplement”) is made and delivered as of [__________], 20[__] pursuant to Section 3(h) of that certain Amended and Restated Security Agreement, dated as of March [_], 2022 (as amended or otherwise modified from time to time, the “Security Agreement”), among each Grantor from time to time party thereto (each a “Grantor” and collectively, the “Grantors”), and Perceptive Credit Holdings III, LP (in such capacity, together with its successors and assigns, the “Administrative Agent”). All capitalized terms used in this Pledge Supplement and not otherwise defined herein shall have the meanings assigned The undersigned verify that they have the authority to thembind in eitherto this First Amendment the Security Agreement or the Credit Agreement (as defined in the Security Agreement), as the context may require.
The undersigned, ___________________________ [insert name of Grantor], a _____________________ [corporation, partnership, limited liability company, etc.], confirms and agrees that all Pledged Collateral of the undersigned, including the property described on the supplemental schedule attached hereto, shall be and become part of the Pledged Collateral and shall secure all Secured Obligations.
Schedule 3 to the Security Agreement is hereby amended by adding to such Schedule 3 the information set forth in the supplement attached hereto.
This Pledge Supplement shall constitute a Loan Document under the Credit Agreement.
THIS PLEDGE SUPPLEMENT SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH, THE LAW OF THE STATE OF NEW YORK.
IN WITNESS WHEREOF, the undersigned has executed this Pledge Supplement, as of the date first above writtenparty on behalf of which they are executing below.
| | | | | | | | | | | |
[____________________] |
| | | |
| By: | |
| Name: | |
| Title: | |
EXHIBIT C
TO THE SECURITY AGREEMENT
FORM OF COPYRIGHT SECURITY AGREEMENT
This COPYRIGHT SECURITY AGREEMENT, dated as of [__________], 20[__] (this “Copyright Security Agreement”), made by each of the signatories hereto (together with any other entity that may become a party hereto as provided herein, the “Copyright Grantors”), is in favor of Perceptive Credit Holdings III, LP, as administrative agent for the Secured Parties (in such capacity, together with its successors and assigns, the “Administrative Agent”).
W I T N E S S E T H:
WHEREAS, the Copyright Grantors are party to the Amended and Restated Security Agreement, dated as of March [_], 2022 (as amended or otherwise modified from time to time, the “Security Agreement”) in favor of the Administrative Agent, pursuant to which the Copyright Grantors are required to execute and deliver this Copyright Security Agreement (capitalized terms used but not otherwise defined herein shall have the meanings given to them in the Security Agreement);
WHEREAS, pursuant to the terms of the Security Agreement, each Copyright Grantor has created in favor of the Administrative Agent a security interest in, and the Administrative Agent has become a secured creditor with respect to, the Copyright Collateral (as defined below);
NOW, THEREFORE, in consideration of the premises and to induce the Administrative Agent and the Lender to enter into the Credit Agreement and to induce the Lender to make their respective extensions of credit to the Borrower thereunder, each Copyright Grantor hereby grants to the Administrative Agent, for the ratable benefit of the Secured Parties, a security interest in all of the following property now owned or at any time hereafter acquired by such Copyright Grantor or in which such Copyright Grantor now has or at any time in the future may acquire any right, title or interest (collectively, the “Copyright Collateral”), as collateral security for the complete payment and performance when due (whether at the stated maturity, by acceleration or otherwise) of all Secured Obligations:
(a) all Copyrights of such Copyright Grantor, including, without limitation, the registered and applied-for Copyrights of such Copyright Grantor listed on Schedule 1 attached hereto;
(b) to the extent not covered by clause (a), all Proceeds of any of the foregoing; and
(c) to the extent not covered by clause (a), all causes of action arising prior to or after the date hereof for infringement of any of the Copyrights.
The security interest granted pursuant to this Copyright Security Agreement is granted in conjunction with the security interest granted to the Administrative Agent pursuant to the Security Agreement, and the Copyright Grantors hereby acknowledge and affirm that the rights and remedies of the Administrative Agent with respect to the security interest in the Copyrights made and granted hereby are more fully set forth in the Security Agreement. In the event that any provision of this Copyright Security Agreement is deemed to conflict with the Security Agreement, the provisions of the Security Agreement shall govern.
Each Copyright Grantor hereby authorizes and requests that the United States Copyright Office record this Copyright Security Agreement.
THIS COPYRIGHT SECURITY AGREEMENT AND THE RIGHTS AND OBLIGATIONS OF THE PARTIES UNDER THIS COPYRIGHT SECURITY AGREEMENT SHALL BE GOVERNED BY, AND CONSTRUED AND INTERPRETED IN ACCORDANCE WITH, THE LAW OF THE STATE OF NEW YORK.
This Copyright Security Agreement may be executed by one or more of the parties to this Copyright Security Agreement on any number of separate counterparts, and all of said counterparts taken together shall be deemed to constitute one and the same instrument. Delivery of an executed signature page of this Copyright Security Agreement by facsimile transmission or electronic transmission (in PDF format) shall be effective as delivery of a manually executed counterpart hereof.
[Signature Pages Follow]
IN WITNESS WHEREOF, each Copyright Grantor has caused this Copyright Security Agreement to be executed and delivered by its duly authorized officer as of the date first above written.
| | | | | | | | |
| | | | | | | | | | | | |
PEAR THERAPEUTICS, INC. |
| University of Virginia Patent Foundation d/b/a University of Virginia Licensing & Ventures Group |
| Pear Therapeutics (US), Inc. | | |
| |
|
| By: | |
/s/ Richard W. Chylla | Name:
By: |
/s/ Corey McCann | |
| Title:Richard W. Chylla
Executive Director | | | Corey McCann, MD, PhD
President & CEO | |
| | |
| | |
| AddressDate: | 200 State Street 13th Floor |
| 12/8/2022 | | Boston, MA 02109 |
Accepted and Agreed:
PERCEPTIVE CREDIT HOLDINGS III, LP, as the Administrative Agent
By: PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner
Date:
12/8/2022 | | | | | | | |
| By: | |
| Name: |
| Title: |
| |
| |
| By | |
| Name: |
| Title: |
Perceptive Credit Holdings III/s/ Peter M. Grant, LP
c/o Perceptive Advisors LLC
51 Astor Place, 10th Floor
New York, NY 10003
Attn: Sandeep Dixit
Email: Sandeep@perceptivelife.com and PCOFReporting@perceptivelife.com
EXHIBIT D
TO THE SECURITY AGREEMENT
FORM OF TRADEMARK SECURITY AGREEMENT
This TRADEMARK SECURITY AGREEMENT, dated as of [__________], 20[__] (this “Trademark Security Agreement”), made by each of the signatories hereto (together with any other entity that may become a party hereto as provided herein, the “Trademark Grantors”), is in favor of Perceptive Credit Holdings III, LP, as administrative agent for the Secured Parties (in such capacity, together with its successors and assigns, the “Administrative Agent”).
W I T N E S S E T H:
WHEREAS, the Trademark Grantors are party to the Amended and Restated Security Agreement, dated as of March [_], 2022 (as amended or otherwise modified from time to time, the “Security Agreement”) in favor of the Administrative Agent, pursuant to which the Trademark Grantors are required to execute and deliver this Trademark Security Agreement (capitalized terms used but not otherwise defined herein shall have the meanings given to them in the Security Agreement);
WHEREAS, pursuant to the terms of the Security Agreement, each Trademark Grantor has created in favor of the Administrative Agent a security interest in, and the Administrative Agent has become a secured creditor with respect to, the Trademark Collateral (as defined below);
NOW, THEREFORE, in consideration of the premises and to induce the Administrative Agent and the Lender to enter into the Credit Agreement and to induce the Lender to make their respective extensions of credit to the Borrower thereunder, each Trademark Grantor hereby grants to the Administrative Agent, for the ratable benefit of the Secured Parties, a security interest in all of the following property now owned or at any time hereafter acquired by such Grantor or in which such Grantor now has or at any time in the future may acquire any right, title or interest (collectively, the “Trademark Collateral”), as collateral security for the complete payment and performance when due (whether at the stated maturity, by acceleration or otherwise) of all Secured Obligations:
(a) all Trademarks of such Trademark Grantor, including, without limitation, the registered and applied-for Trademarks of such Grantor listed on Schedule 1 attached hereto (excluding any application for registration of a trademark filed on an intent-to-use (or equivalent) basis solely to the extent that the grant of a security interest in any such trademark application would materially adversely affect the validity or enforceability of such application or the resulting registration, or result in abandonment of application or cancellation of the resulting registration);
(b) to the extent not covered by clause (a), all Proceeds of any of the foregoing;
(c) to the extent not covered by clause (a), the goodwill of the businesses with which the Trademarks are associated; and
(d) to the extent not covered by clause (a), all causes of action arising prior to or after the date hereof for infringement of any of the Trademarks or unfair competition regarding the same.
The security interest granted pursuant to this Trademark Security Agreement is granted in conjunction with the security interest granted to the Administrative Agent pursuant to the Security Agreement, and the Trademark Grantors hereby acknowledge and affirm that the rights and remedies of the Administrative Agent with respect to the security interest in the Trademarks made and granted hereby are more fully set forth in the Security Agreement. In the event that any provision of this Trademark Security Agreement is deemed to conflict with the Security Agreement, the provisions of the Security Agreement shall govern.
Each Trademark Grantor hereby authorizes and requests that the Commissioner of Patents and Trademarks record this Trademark Security Agreement.
THIS TRADEMARK SECURITY AGREEMENT AND THE RIGHTS AND OBLIGATIONS OF THE PARTIES UNDER THIS TRADEMARK SECURITY AGREEMENT SHALL BE GOVERNED BY, AND CONSTRUED AND INTERPRETED IN ACCORDANCE WITH, THE LAW OF THE STATE OF NEW YORK.
This Trademark Security Agreement may be executed by one or more of the parties to this Trademark Security Agreement on any number of separate counterparts, and all of said counterparts taken together shall be deemed to constitute one and the same instrument. Delivery of an executed signature page of this Trademark Security Agreement by facsimile transmission or electronic transmission (in PDF format) shall be effective as delivery of a manually executed counterpart hereof.
[Signature Pages Follow]
IN WITNESS WHEREOF, each Trademark Grantor has caused this Trademark Security Agreement to be executed and delivered by its duly authorized officer as of the date first above written.
| | | | | | | | |
| II | | | | |
| PEAR THERAPEUTICS, INC. |
Peter M. Grant, II
Chair, Board of Directors | | | |
Date: |
12/8/2022 | | | | |
| | |
| By: | |
| Name: | |
| Title: | |
| | |
| | |
| Address: | 200 State Street 13th Floor |
| | Boston, MA 02109 |
Accepted and Agreed:
PERCEPTIVE CREDIT HOLDINGS III, LP, as the Administrative Agent
By: PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner
| | | | | |
| By | |
| Name: |
| Title: |
| |
| |
| By | |
| Name: |
| Title: |
Perceptive Credit Holdings III, LP
c/o Perceptive Advisors LLC
51 Astor Place, 10th Floor
New York, NY 10003
Attn: Sandeep Dixit
Email: Sandeep@perceptivelife.com and PCOFReporting@perceptivelife.com
EXHIBIT E
TO THE SECURITY AGREEMENT
FORM OF PATENT SECURITY AGREEMENT
This PATENT SECURITY AGREEMENT, dated as of [__________], 20[__] (this “Patent Security Agreement”), made by each of the signatories hereto (together with any other entity that may become a party hereto as provided herein, the “Patent Grantors”), is in favor of Perceptive Credit Holdings III, LP, as administrative agent for the Secured Parties (in such capacity, together with its successors and assigns, the “Administrative Agent”).
W I T N E S S E T H:
WHEREAS, the Patent Grantors are party to the Amended and Restated Security Agreement, dated as of March [_], 2022 (as amended or otherwise modified from time to time, the “Security Agreement”) in favor of the Administrative Agent, pursuant to which the Patent Grantors are required to execute and deliver this Patent Security Agreement (capitalized terms used but not otherwise defined herein shall have the meanings given to them in the Security Agreement);
WHEREAS, pursuant to the terms of the Security Agreement, each Patent Grantor has created in favor of the Administrative Agent a security interest in, and the Administrative Agent has become a secured creditor with respect to, the Patent Collateral (as defined below);
NOW, THEREFORE, in consideration of the premises and to induce the Administrative Agent and the Lender to enter into the Credit Agreement and to induce the Lender to make their respective extensions of credit to the Borrower thereunder, each Patent Grantor hereby grants to the Administrative Agent, for the ratable benefit of the Secured Parties, a security interest in all of the following property now owned or at any time hereafter acquired by such Patent Grantor or in which such Patent Grantor now has or at any time in the future may acquire any right, title or interest (collectively, the “Patent Collateral”), as collateral security for the complete payment and performance when due (whether at the stated maturity, by acceleration or otherwise) of all Secured Obligations:
(a) all Patents of such Patent Grantor, including, without limitation, the registered and applied-for Patents of such Grantor listed on Schedule 1 attached hereto;
(b) to the extent not covered by clause (a), all Proceeds of any of the foregoing; and
(c) to the extent not covered by clause (a), all causes of action arising prior to or after the date hereof for infringement of any of the Patents.
The security interest granted pursuant to this Patent Security Agreement is granted in conjunction with the security interest granted to the Administrative Agent pursuant to the
Security Agreement, and the Patent Grantors hereby acknowledge and affirm that the rights and remedies of the Administrative Agent with respect to the security interest in the Patents made and granted hereby are more fully set forth in the Security Agreement. In the event that any provision of this Patent Security Agreement is deemed to conflict with the Security Agreement, the provisions of the Security Agreement shall govern.
Each Patent Grantor hereby authorizes and requests that the Commissioner of Patents and Trademarks record this Patent Security Agreement.
THIS PATENT SECURITY AGREEMENT AND THE RIGHTS AND OBLIGATIONS OF THE PARTIES UNDER THIS PATENT SECURITY AGREEMENT SHALL BE GOVERNED BY, AND CONSTRUED AND INTERPRETED IN ACCORDANCE WITH, THE LAW OF THE STATE OF NEW YORK.
This Patent Security Agreement may be executed by one or more of the parties to this Patent Security Agreement on any number of separate counterparts, and all of said counterparts taken together shall be deemed to constitute one and the same instrument. Delivery of an executed signature page of this Patent Security Agreement by facsimile transmission or electronic transmission (in PDF format) shall be effective as delivery of a manually executed counterpart hereof.
[Signature Pages Follow]
IN WITNESS WHEREOF, each Patent Grantor has caused this Patent Security Agreement to be executed and delivered by its duly authorized officer as of the date first above written.
| | | | | | | | |
| PEAR THERAPEUTICS, INC. |
| | |
| | |
| By: | |
| Name: | |
| Title: | |
| | |
| | |
| Address: | 200 State Street 13th Floor |
| | Boston, MA 02109 |
Accepted and Agreed:
PERCEPTIVE CREDIT HOLDINGS III, LP, as Administrative Agent
By: PERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner
| | | | | |
| By | |
| Name: |
| Title: |
| |
| |
| By | |
| Name: |
| Title: |
Perceptive Credit Holdings III, LP
c/o Perceptive Advisors LLC
51 Astor Place, 10th Floor
New York, NY 10003
Attn: Sandeep Dixit
Email: Sandeep@perceptivelife.com and PCOFReporting@perceptivelife.com
Exhibit 10.36
Execution Version
FIRST AMENDMENT TO AMENDED AND RESTATED INTERCOMPANY SUBORDINATION
CREDIT AGREEMENT AND GUARANTY
This FIRST AMENDMENT TO AMENDED AND RESTATED INTERCOMPANY SUBORDINATIONCREDIT AGREEMENT AND GUARANTY, dated as of March 25January 13, 20222023 (this “Subordination AgreementAmendment”), is entered into by and among Pear TherapeuticsPEAR THERAPETUICS, IncINC., a Delaware corporation (f/k/a THIMBLE POINT ACQUISITION CORP.) (“Holdings”), Pear TherapeuticsPEAR THERAPEUTICS (US), IncINC., a Delaware corporation (f/k/a PEAR THERAPEUTICS, INC.) (the “Borrower”), certain Subsidiaries of the Borrower that are parties hereto, and certain other Subsidiaries of Holdings that may, from time to time in the future, become parties hereto by executing and delivering a joinder agreement in substantially the form of Exhibit A hereto (any such Subsidiary being herein, individually, a “Subsidiary Party” and collectively the “Subsidiary Parties”) and Perceptive Credit Holdings III, LP, a Delaware limited partnership, in its capacity as Administrative Agentand PERCEPTIVE CREDIT HOLDINGS III, LP, a Delaware limited partnership, as administrative agent for the Lenders under the Credit Agreement (as defined below) (in such capacity, together with its successors and assigns, the “Administrative Agent”).
Reference is made to that certain Intercompany Subordination Agreement, dated as of June 30, 2022 (the “Existing Intercompany Subordination Agreement”), which was entered into by the Borrower, each Subsidiary Party party thereto and the Administrative Agent pursuant to the terms of that certain and as the Majority Lender. Reference is made to the Amended and Restated Credit Agreement and Guaranty, dated as of JuneMarch 3025, 2020 (2022 (as amended, supplemented or otherwise modified and in effect prior to the date hereof, the “Existing Credit Agreement”, and as further amended by this Amendment, the “Credit Agreement”), by and among the Borrower, certain Subsidiariessubsidiaries of the Borrower party thereto from time to time party thereto, the lenders from time to time party thereto (the “Lenders”) and the Administrative Agent.Lenders (as defined therein) from time to time party thereto and the Administrative Agent. Capitalized terms used herein without definition shall have the same meanings as set forth in the Credit Agreement.
The parties hereto have agreed to amend and restateRECITALS
WHEREAS, the Loans under the Existing Credit Agreement pursuant to the terms of that certainbear interest based on London interbank offered rate (“LIBOR”) in accordance with the terms of the Existing Credit Agreement;
WHEREAS, on March 5, 2021, the U.K. Financial Conduct Authority confirmed the Governmental Authority having jurisdiction over the quotation or determination of LIBOR will either cease to supervise or sanction such rates for purposes of interest rates on loans or no longer be representative and the Administrative Agent has determined that One-Month LIBOR (as defined in the Existing Credit Agreement) can no longer be determined by the Administrative Agent; and
WHEREAS, the parties to the Existing Credit Agreement have agreed to establish an alternate rate of interest to One-Month LIBOR and to make such other related changes to the Existing Credit Agreement to reflect such alternate rate of interest;
NOW, THEREFORE, the parties hereto hereby agree as follows:
ARTICLE I
AMENDMENTS TO THE EXISTING CREDIT AGREEMENT
SECTION 1.01. Amendments to the Existing Credit Agreement. As of the Amendment Effective Date (defined below), the Existing Credit Agreement is hereby amended as set forth below:
(a)Section 1.01 of the Existing Credit Agreement is hereby amended by inserting the following new definitions in their respective alphabetically correct places:
“Amendment No. 1” means that certain First Amendment to Amended and Restated Credit Agreement and Guaranty, dated as of the date hereof (as amended or otherwise modified from time to time, the “Credit Agreement”), by and among Holdings, the Borrower, the other Subsidiaries of Holdings from time to time party thereto, the Lenders from time to time party thereto and the Administrative AgentJanuary 13, 2023.
As a condition precedent to the effectiveness“Conforming Changes” means, with respect to either the use or administration of One-Month Term SOFR, any technical, administrative or operational changes (including changes to the definition of the Credit Agreement,“Business Day,” the parties hereto desire to amend and restatedefinition of “U.S. Government Securities Business Day,” the Existing Intercompany Subordination Agreement indefinition of “Interest Period” or its entirety on the terms and conditions set forth herein.any
Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Credit Agreement.
One or more of Holdings, the Borrower and the Subsidiary Parties (individually, a “Pear Party” and collectively, the “Pear Parties”), in their capacities as lenders (each such entity, together with its successors, assigns and transferees in such capacity, individually, a “Junior Creditor”, and, collectively, “Junior Creditors”) has made, or may from time to time may make, loans or extend other financings to one or more of the Pear Parties that is an Obligor (each such Obligor, in its capacity as a borrower from any Junior Creditor (together with its successors, assigns and transferees) being herein, individually, a “Debtor Obligor”, and, collectively, “Debtor Obligors”) pursuant to Section 9.01(e) of the Credit Agreement. All such Indebtedness resulting from the making of any such loan or financing, together with all principal, interest, fees, premiums, costs, expenses, liabilities, obligations and other amounts of any type or nature at any time owing or arising in respect thereof, including but not limited to any such items or amounts as may accrue or be incurred before or after default or workout or the commencement of any liquidation, dissolution, bankruptcy, receivership or reorganization case by or against the Borrower, is herein collectively referred to as the “Junior Obligations”.
Each of the Junior Creditorssimilar or analogous definition (or the addition of a concept of “interest period”), with respect to the timing and each of the Debtor Obligors, for the benefitfrequency of determining rates and making payments of interest, the timing of borrowing requests, prepayments or conversion or continuation notices, the applicability and length of lookback periods, the applicability of Section 3.02(e) and other technical, administrative or operational matters) that the Administrative Agent reasonably decides may be appropriate to reflect the adoption and implementation of such rate or to permit the use and administration thereof by the Administrative Agent in a manner substantially consistent with market practice (or, if the Administrative Agent decides that adoption of any portion of such market practice is not administratively feasible or if the Administrative Agent determines that no market practice for the administration of the Secured Parties and each of their permitted successors, transferees and assigns, hereby irrevocably and unconditionally agree as follows:
1.such rate exists, in such other manner of administration as the Administrative Agent reasonablyAll payment obligations and other monetary obligations of any Debtor Obligor arising from time to time under or in connection with any Junior Obligations to any Junior Creditor are, and shall at all times be, subordinated in right of payment and performance to the prior Payment in Full (as defined below) of all Obligations owing under or in connection with the Credit Agreement and the other Loan Documents, whether in respect of principal, interest, fees or other monetary obligations or liabilities of any type or nature, including costs and expenses of enforcement, if any (collectively decides is necessary in connection with the administration of this Agreement and the other Loan Documents).
“One-Month Term SOFR” means, the Term SOFR Reference Rate (expressed, as a decimal, rounded upwards, if necessary, to the nearest 1/100th of 1%) for a one month tenor on the day (such day, the “Senior ObligationsPeriodic Term SOFR Determination Day”), notwithstanding the maturity date or amortization date of any Junior Obligations or any acceleration of the maturity date related thereto, any default by or insolvency that is two (2) U.S. Government Securities Business Days prior to the first day of the applicable Interest Period, as such rate is published by the Term SOFR Administrator; provided that if as of 5:00 p.m. (New York City time) on any Periodic Term SOFR Determination Day the Term SOFR Reference Rate for the applicable tenor has not been published by the Term SOFR Administrator, then Term SOFR will be the Term SOFR Reference Rate for such tenor as published by the Term SOFR Administrator on the first preceding U.S. Government Securities Business Day for which such Term SOFR Reference Rate for such tenor was published by the Term SOFR Administrator so long as such first preceding U.S. Government Securities Business Day is not more than three (3) U.S. Government Securities Business Days prior to such Periodic Term SOFR Determination Day.
“Periodic Term SOFR Determination Day” has the meaning set forth in the definition of “One-Month Term SOFR”.
“Reference Rate Transition Event” means the occurrence of one or more of the following events with respect to the Reference Rate then in effect:
(a) a public statement or publication of information by or on behalf of any Junior Creditor or any other Person, or otherwisethe administrator of such Reference Rate (or the published component used in the calculation thereof) stating that such administrator has ceased or will cease to provide such Reference Rate (or such component thereof), permanently or indefinitely; provided that, at the time of such statement or publication, there is no successor administrator that will continue to provide such Reference Rate (or such component thereof);
(b) a public statement or publication of information by the Governmental Authority governing or regulating the administrator of such Reference Rate (or the published component used in the calculation thereof), the U.S. Federal Reserve System, an insolvency official with jurisdiction over the then-current administrator for such Reference Rate (or such component thereof), a resolution authority with jurisdiction over the then-current administrator for such Reference Rate (or such component thereof) or a court or an entity with similar insolvency or resolution authority over the administrator for such Reference Rate (or such
component thereof), which in any case states that the then-current administrator of such Reference Rate (or such component thereof) has ceased or will cease to provide such Reference Rate (or such component thereof) permanently or indefinitely; provided that, at the time of such statement or publication, there is no successor administrator that will continue to provide such Reference Rate (or such component thereof); or
(c) a public statement or publication of information by the Governmental Authority governing or regulating the then-current administrator of such Reference Rate stating that such Reference Rate (or the published component used in the calculation thereof) is no longer, or as a of a specified future date will not be, representative.
2.The parties hereto agree that this Subordination Agreement is for the benefit of,A “Reference Rate Transition Event” will be deemed to have occurred with respect to any Reference Rate if a public statement or publication of information set forth above has occurred with respect to each then-current available tenor of such Reference Rate (or the published component used in the calculation thereof).
“SOFR” means a rate equal to the secured overnight financing rate as administered by the SOFR Administrator.
“SOFR Administrator” means the Federal Reserve Bank of andNew shallYork be enforceable by the Administrative Agent on behalf of the Secured Parties.
(or a successor administrator of the secured overnight financing rate).
“Term SOFR Administrator” means CME Group Benchmark Administration Limited (CBA) (or a successor administrator of the Term SOFR Reference Rate selected by the Administrative Agent in its reasonable discretion).
“Term SOFR Reference Rate” means the forward-looking term rate based on SOFR.
“U.S. Government Securities Business Day” means any day except for (i) a Saturday, (ii) a Sunday or (iii) a day on which the Securities Industry and Financial Markets Association recommends that the fixed income departments of its members be closed for the entire day for purposes of trading in United States government securities.”
(b)The following defined term set forth in Section 1.01 of the Existing Credit Agreement is hereby amended and restated in its entirety to read as follows:
“Reference Rate” means One-Month Term SOFR; provided that if One-Month Term SOFR can no longer be determined by the Administrative Agent for any reason (in its sole discretion, which determination shall be conclusive absent manifest or demonstrable error), including as a result of the Term SOFR Reference Rate not being available or published on a current basis or as a result of the occurrence of a Reference Rate Transition Event, then the Administrative Agent and the Borrower shall endeavor, in good faith, to establish an alternate rate of interest to One-Month Term SOFR that gives due consideration to the then prevailing market convention for determining a rate of interest for middle-market loans in the United States at such time, and shall enter into an amendment to this Agreement to reflect such alternate rate of interest and such other related changes to this Agreement as may be applicable; provided, further that, until such alternate rate of interest is agreed upon by the Administrative Agent and the Borrower, the Reference Rate for purposes hereof and of each other Loan Document shall be the Wall Street Journal Prime Rate.
(c)The following defined term set forth in Section 1.01 of the Existing Credit Agreement is hereby deleted in their entirety: “One-Month LIBOR”.
(d)Section 1 of the Existing Credit Agreement is hereby amended by inserting new Sections 1.05 and 1.06 at the end of such section, which shall read as follows:
“1.05 Reference Rate Replacement. Upon the occurrence of an event of the type described in the first proviso of the definition of “Reference Rate”, the Administrative Agent will promptly notify the Borrower thereof and, as set forth in such proviso, the Administrative Agent and the Borrower shall endeavor, in good faith, to establish an alternate rate of interest to One-Month Term SOFR, provided that there is no assurance that the composition or characteristics of any such alternative, successor or replacement Reference Rate will be similar to or produce the same value or economic equivalence as One-Month Term SOFR or that it will have the same volume or liquidity as did One-Month Term SOFR prior to its discontinuance or unavailability.
1.06 Rates.
3.At all times from and afterThe Administrative Agent does not warrant or accept responsibility for, and shall thenot datehave of this Subordination Agreement until Payment in Full of all Senior Obligations and the termination of all Senior Liens (as defined below), (i) no Debtor Obligor shall make, and no Junior Creditor shall accept, receive or collect from or on behalf of any Debtor Obligor, any direct or indirect payment or distribution of any kind or character whatsoever (whether in cash, securities, other property, by set-off, forgiveness of any Indebtedness of any Secured Party, or otherwise) on account of any of the Junior Obligations, and (ii) under no circumstance shall any payment of any of the Junior Obligations be accelerated, or any other remedy, enforcement action or other action be taken by any Junior Creditor against any Debtor Obligorany liability with respect to (i) the continuation of, administration of, submission of, calculation of or any other matter related to the Term SOFR Reference Rate or One-Month Term SOFR, or any component definition thereof or rates referred to in the definition thereof, or any alternative, successor or replacement rate thereto, including whether the composition or characteristics of any such alternative, successor or replacement rate will be similar to, or produce the same value or economic equivalence of, or have the same volume or liquidity as, the Term SOFR Reference Rate or One-Month Term SOFR prior to its discontinuance or unavailability, or (ii) the effect, implementation or composition of any Conforming Changes. The Administrative Agent and its Affiliates or other related entities may engage in transactions that affect the calculation of the Term SOFR Reference Rate or One-Month Term SOFR, any alternative, successor or replacement rate or any property of any Debtor Obligor or of any other Person, in each case with respect to any of the Junior Obligations (including to assert, enforce or collect any of the Junior Obligations), in each case, exceptrelevant adjustments thereto, in each case, in a manner adverse to the extent permitted by the Credit Agreement or with the prior written consent of the Administrative Agent, which consent may notBorrower. The Administrative Agent may select information sources or services in its reasonable discretion to ascertain the Term SOFR Reference Rate or One-Month Term SOFR, in each case pursuant to the terms of this Agreement, and shall have no liability to the Borrower, any Lender or any other Person or entity for damages of any kind, including direct or indirect, special, punitive, incidental or consequential damages, costs, losses or expenses (whether in tort, contract or otherwise and whether at law or in equity), for any error or calculation of any such rate (or component thereof) provided by any such information source or service.”
(e)Section 3.02 of the Existing Credit Agreement is hereby amended by adding new clauses (d) and (e) at the end of such section, which shall read as follows:
“(d) Conforming Changes. In connection with the use or administration of One-Month Term SOFR, the Administrative Agent will have the right to make Conforming Changes from time to time and, notwithstanding anything to the contrary herein or in any other Loan Document, any amendments implementing such Conforming Changes will become effective without any further action or consent of any other party to this Agreement or any other Loan Document. The Administrative Agent will promptly notify the Borrower and the Lenders of the effectiveness of any Conforming Changes in connection with the use or administration of One-Month Term SOFR.
(e) Compensation for Losses. In the event of the payment of any principal of any Loan other than on the last day of the Interest Period applicable thereto (including as a result of an Event of Default), then, in any such event, the Borrower shall compensate each Lender for any loss, cost or expense attributable to such event and actually incurred by such Lender, including any loss, cost or expense arising from the liquidation or redeployment of funds, but excluding lost profits or the loss of any Applicable Margin. A certificate of any Lender setting forth any amount or amounts that such Lender is entitled to receive pursuant to this Section 3.02(e) shall be delivered to the Borrower and shall be conclusive absent manifest error. The Borrower shall pay such Lender the amount shown as due on any such certificate within ten (10) days after receipt thereof. This Section 3.02(e) shall not apply with respect to Taxes other than any Taxes that represent Losses arising from any non-Tax loss, cost or expense.”
SECTION 1.02. Existing LIBOR Loans. Notwithstanding the amendments contemplated by this Amendment, interest due and payable under the Credit Agreement on January 31, 2023, shall accrue and be unreasonably withheld.
4.No Junior Creditor shall, directly or indirectly, independently or with any other Person, take any action that would be in violation of, or inconsistent with, or result in a breach of this Subordination Agreement or challenge or contest (i) the validity, perfection, priority or enforceability of this Subordination Agreement, any Senior Obligations or any Liens securing the Senior Obligations (“Senior Liens”), (ii) any of the rights of any Secured Party set forth in the Credit Agreement or any other Loan Document (including with respect to the Senior Liens), or (iii) the validity or enforceability of the Credit Agreement or any other Loan Document or payable as set forth in the Existing Credit Agreement without giving effect to the amendments contemplated by this Amendment.
ARTICLE II
ACKNOWLEDGEMENT, AGREEMENT AND CONSENT AND
REPRESENTATIONS AND WARRANTIES
SECTION 1.01. Each Obligor confirms, agrees, represents and warrants that, notwithstanding the effectiveness of this Amendment, the Obligations of such Obligor under the Credit Agreement and each other Loan Document to which such Obligor is a party shall not be and have not been impaired or limited and the Credit Agreement and each other Loan Document to which such Obligor is a party is, and shall continue to be, in full force and effect and is hereby confirmed and ratified in all respects by such Obligor.
SECTION 1.02. Each Obligor hereby acknowledges, agrees, represents and warrants that, except as expressly provided in this Amendment, no term or provision of the Existing Credit Agreement or any other Loan Document to which such Obligor is a party is amended or otherwise modified in any respect.
SECTION 1.03. To induce the Administrative Agent and the Lenders to execute and deliver this Amendment, each Obligor party hereto further represents and warrants to the Administrative Agent and the Lenders that as of the date hereof each of the following statements are true and correct:
(a)Such Obligor has full power, authority and legal right to enter into this Amendment and perform its obligations under this Amendment and each Loan Document to which it is a party as amended hereby.
(b)The transactions contemplated by this Amendment and the Credit Agreement are within such Obligor’s corporate or other powers and have been duly authorized by all necessary corporate action including, if required, approval by all necessary holders of Equity Interests. This Amendment has been duly executed and delivered by such Obligor and this Amendment, the Credit Agreement and each other Loan Document to which such Obligor is a party each constitutes a legal, valid and binding obligation of such Obligor, enforceable against such Obligor in accordance with its terms, except as such enforceability may be limited by (i) bankruptcy, insolvency, reorganization, moratorium or similar laws of general applicability affecting the enforcement of creditors’ rights and (ii) the application of general principles of equity (regardless of whether such enforceability is considered in a proceeding in equity or at law).
(c)No authorization or approval or other action by, and no notice to or filing with, any Governmental Authority or any other Person (other than those that have been duly obtained or made and which are in full force and effect as of the Amendment Effective Date) is required for the due execution or delivery by such Obligor of this Amendment, or performance by such Obligor of its obligations under this Amendment and each Loan Document to which it is a party as amended hereby. The execution, delivery and performance by such Obligor of this Amendment and the Loan Documents to which it is a party as amended hereby, will not (i) violate or conflict with any Law, (ii) violate or conflict with any Organic Document of any Obligor, (iii) violate or conflict with any Governmental Approval of any Governmental Authority, (iv) violate or result in a default under any Material Agreement binding upon an Obligor or any of its Subsidiaries that, individually or in the aggregate, could reasonably be expected to result in a Material Adverse Effect or (v) result in the creation or imposition of any Lien (other than Permitted Liens) on any asset of such Obligor.
(d)Both immediately before and after giving effect to this Amendment, no Default or Event of Default shall have then occurred and be continuing, or could reasonably be expected to result from the execution, delivery and performance of this Amendment or the transactions contemplated hereby.
ARTICLE III
CONDITIONS PRECEDENT
SECTION 1.01. Conditions to Effectiveness of this Amendment. This Amendment shall become effective only upon, and shall be subject to, the prior or simultaneous satisfaction or waiver of each of the following conditions precedent in a manner reasonably satisfactory to the Administrative Agent (the date of satisfaction of such conditions being referred to as the “Amendment Effective Date”):
(a)Executed Amendment. The Administrative Agent shall have received this Amendment, duly executed by the Borrower, the Subsidiary Guarantors, the Administrative Agent and each of the Lenders party hereto.
(b)Representations and Warranties. The statements, representations and warranties contained in Article II above shall each be true and correct, both immediately before and after giving effect to this Amendment, and the Administrative Agent shall have received a certificate executed by a Responsible Officer of the Borrower, in form and substance reasonably satisfactory to the Administrative Agent, addressed to it and the Lenders and certifying as to the foregoing.
(c)Costs and Expenses, Etc. The Administrative Agent shall have received for its account and the account of each Lender all reasonable and documented fees, costs and expenses due and payable to them pursuant to Section 14.03 of the Credit Agreement (including the Administrative Agent’s and each Lender’s reasonable and documented legal fees and out-of-pocket expenses).
ARTICLE IV
MISCELLANEOUS
SECTION 1.01. Governing Law; Jurisdiction; Jury Trial. This Amendment and the rights and obligations of the parties hereunder shall be governed by, and construed in accordance with, the law of the State of New York. The jurisdiction and waiver of jury trial provisions set forth in Sections 14.10 and 14.11 of the Credit Agreement, respectively, are incorporated herein by reference mutatis mutandis.
SECTION 1.02. Effect of this Amendment.
(a)On and after the Amendment Effective Date, each reference in any Loan Document (other than this Amendment) to the Credit Agreement shall mean and be a reference to the Existing Credit Agreement as amended by this Amendment.
(b)This Amendment shall constitute a Loan Document for all purposes of the Credit Agreement and each other Loan Documents. The Obligors party hereto agree that all of the representations, warranties, terms, covenants, conditions and other provisions of the Existing Credit Agreement and other Loan Documents shall, except as expressly set forth in this Amendment, remain unchanged and shall continue to be, and shall remain, in full force and effect in accordance with their respective terms. The amendments set forth herein shall be limited precisely as provided for herein to the provisions expressly amended herein and shall not be deemed to be an amendment to or modification of any other term or provision of the Existing Credit Agreement or any other Loan Document or of any portion thereof.
5.In the event that, prior to Payment in Fulltransaction or further or future action on the part of any Obligor which would require the consent of the Senior Obligations, any Junior Creditor shall receive any payment or distribution of any kind or character whatsoever (whether in cash, securities, other property, by set-off, forgiveness of any Indebtedness of any Secured Party, or otherwise) on or in respect of all or any portion of the Junior Obligations in violation of any of the provisions of this Subordination Agreement, then such payment or distribution shall be held in trust by such Junior Creditor for the benefit of, and promptly (and in any event within one (1) Business Day) paid over by such Junior Creditor to the Administrative Agent for application of such payment or distribution to repay the Obligations in accordance with the terms thereof, until Payment in Full of the Obligations as confirmed in writing by the Administrative Agent to the Borrower.
6.For purposes of this Subordination Agreement, “Payment in Full” means, with respect to the Obligations, that all such obligations and other amounts payable constituting Obligations have been indefeasibly paid in full in cash.
7.Neither any Junior Creditor nor any Debtor Obligor may assign or transfer any of its rights or obligations hereunder, except to another Obligor that becomes bound by the terms of this Subordination Agreement.
8.This Subordination Agreement is a Loan Document executed pursuant to the Credit Agreement and shall (unless otherwise expressly indicated therein) be construed, administered and applied in accordance with allLenders or the Administrative Agent under the Credit Agreement or any other Loan Document. Except as expressly amended by this Amendment, the Existing Credit Agreement and the other Loan Documents are and shall continue to be in full force and effect and are hereby in all respects ratified and confirmed.
(c)The execution, delivery and effectiveness of this Amendment shall not operate as a waiver of any right, power or remedy of any holder of the Administrative Agent or any Lender under any Loan Document or applicable Law, nor constitute a waiver of the terms and provisions of the Credit Agreement, as amended hereby, including Section 14 thereof. The provisions of this Subordination Agreement shall any provision of the Credit Agreement.
SECTION 1.03. No Novation. This Amendment is not intended by the parties to be, and shall not be binding upon and inureconstrued to the benefitbe, a novation of the parties hereto and their respective permitted successors and assignsExisting Credit Agreement, any other Loan Document or any Obligation thereunder.
9SECTION 1.04.This Subordination Agreement Counterparts; Electronic Signatures. This Amendment may be executed in any number of counterparts, all of which taken together shall constitute one and the same instrument and any of the parties hereto may execute this Subordination AgreementAmendment by signing any such counterpart.
10.This Subordination Agreement and the rights and obligations Delivery of an executed signature page of this Amendment by facsimile transmission or electronic transmission (in PDF format) shall be effective as delivery of a manually executed counterpart hereof. Any signature (including, without limitation, (x) any electronic symbol or process attached to, or associated with, a contract or other record and adopted by a person with the intent to sign, authenticate or accept such contract or record and (y) any facsimile or .pdf signature) hereto or to any other certificate, agreement or document related to this transaction, and any contract formation or record-keeping, in each case, through electronic means, shall have the same legal validity and enforceability as a manually executed signature or use of a paper-based record-keeping system to the fullest extent permitted by applicable law, including the Federal Electronic Signatures in Global and National Commerce Act, the New York State Electronic Signatures and Records Act, or any similar state law based on the Uniform Electronic Transactions Act, and the parties hereto hereby waive any objection to the contrary.
SECTION 1.05. Binding Nature. The provisions of this Amendment shall be binding upon and shall inure to the benefit of the parties hereunder shall be governed by,hereto and construed in accordance with, the law of the State of New York, without regard to principles of conflicts of laws that would result in the application of the laws of any other jurisdictiontheir respective successors and permitted assigns; provided that Section 5-1401 of the New York General Obligations Law shall apply.
11.Any Subsidiary of Holdings may, without the consent of any other party to Subordination Agreement, become an Pear Party under this Subordination Agreement by executing and delivering to the Administrative Agent a joinder agreement in substantially the form of Exhibit A hereto.
12.Except as modified in accordance with Section 11 to add any Subsidiary of Holdings as an additional Pear Party to this Subordination Agreement, this Subordination Agreement may not be amended, waivedno Obligor may assign or otherwise modified without the prior written consent of each of the parties hereto.
transfer any of its rights or obligations hereunder without the prior written consent of the Administrative Agent.SECTION 1.06. Captions. The captions and section headings appearing herein are included solely for convenience of reference and are not intended to affect the interpretation of any provision of this Amendment.
SECTION 1.07.This Subordination Agreement amends and restates the Existing Intercompany Subordination Agreement but does not constitute and, is not intended Severability. If any provision hereof is found by a court to create, a novation or accord and satisfaction. All obligations pursuantbe invalid or unenforceable, to the Existing Intercompany Subordination Agreement shall continue in full force and effect pursuant tofullest extent permitted by any applicable Law the parties agree that such invalidity or unenforceability shall not impair the validity or enforceability of any this Subordination Agreementother provision hereof.
SECTION 1.08.
Integration. This Amendment constitutes the entire agreement among the parties with respect to the subject matter hereof and supersedes any and all previous agreements and understanding, oral or written, relating to the subject matter hereof.
[SIGNATURE PAGE FOLLOWSSignature pages to follow]
IN WITNESS WHEREOF, the parties hereto have caused this Subordination AgreementAmendment to be duly executed and delivered as of the date first above written.hereof.
BORROWER:
PEAR THERAPEUTICS (US), INC.
By:____
| | | | | |
By | /s/ Ronan O'Brien______________’Brien |
Name: Ronan O'’Brien
Title: General Counsel & Chief Compliance Officer
PEAR THERAPEUTICS, INC.
By:______/s/ Ronan O'Brien____________
Name: Ronan O'Brien
Title: Secretary
PEAR THERAPEUTICS SECURITIES CORPORATION
By:_____/s/ Ronan O'Brien______________
Name: Ronan O'Brien
Title: General Counsel & Chief Compliance Officer
ny-2326299 General Counsel & Chief Compliance Officer
HOLDINGS:
PEAR THERAPEUTICS, INC.
Name: Ronan O’Brien
Title: Secretary
PERCEPTIVE CREDIT HOLDINGS III, LP, , as the Administrative Agent and Majority Lender
By: Perceptive Credit OpportunitiesPERCEPTIVE CREDIT OPPORTUNITIES GP, LLC, its general partner
| | | | | |
By:_ | /s/ Sandeep Dixit_________________ |
Name: Sandeep Dixit
Title: Chief Credit Officer
| | | | | |
By:___ | /s/ Sam Chawla_________________ |
Name: Sam Chawla
Title: Portfolio Manager
Exhibit A
FORM OF INTERCOMPANY SUBORDINATION AGREEMENT JOINDER
This INTERCOMPANY SUBORDINATION AGREEMENT JOINDER, dated as of [DATE] by [NAME OF ADDITIONAL SUBSIDIARY], a [state of organization] [corporation][limited liability company] (the “Additional Pear Party”), under that certain Amended and Restated Intercompany Subordination Agreement, dated as of March 25, 2022 (as amended and restated, amended or otherwise modified from time to time, the “Subordination Agreement”), among Pear Therapeutics (US), Inc., a Delaware corporation (“Holdings”), Pear Therapeutics (US), Inc., a Delaware corporation (the “Borrower”), certain Subsidiaries of Holdings from time to time party thereto and Perceptive Credit Holdings III, LP, a Delaware limited partnership, in its capacity as Administrative Agent for the Lenders under the Credit Agreement (in such capacity, together with its successors and assigns, the “Administrative Agent”). Capitalized terms used herein and not otherwise defined herein are used herein as defined in the Subordination Agreement.
Pursuant to Section 11 of the Subordination Agreement, the Additional Pear Party hereby agrees to become a “Pear Party” for all purposes of the Subordination Agreement.
IN WITNESS WHEREOF, the Additional Pear Party has caused this Intercompany Subordination Agreement Joinder to be duly executed and delivered as of the day and year first above written.
[ADDITIONAL PEAR PARTY]
By _______________________________________
Name:
Title:
ny-2326299 Portfolio Manager
CONSENT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
We consent to the incorporation by reference in Registration Statement NoNos. 333-261876 and 333-269097 on Form S-3 and Registration Statement Nos. 333-262568 and 333-269667 on Form S-8 of our report dated March 2831, 20222023, relating to the financial statements of Pear Therapeutics, Inc., appearing in this Annual Report on Form 10-K for the year ended December 31, 2021.2022.
/s/ Deloitte & Touche LLP
Boston, Massachusetts
March 28, 202231, 2023
CERTIFICATION PURSUANT TO
RULES 13a-14(a) AND 15d-14(a) UNDER THE SECURITIES EXCHANGE ACT OF 1934,
AS ADOPTED PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002
I, Corey M. McCann, certify that:
1.I have reviewed this Annual Report on Form 10-K of Pear Therapeutics, Inc.;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statementstatements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
(a)Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b)[omitted]Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c)Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
(d)Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5.The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
(a)All significant deficiencies and material weaknesses in the design or operation of internal controlscontrol over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
(b)Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal controlscontrol over financial reporting.
Date: March 2831, 20222023
| | |
|
| /s/ Corey M. McCann |
| Dr. Corey M. McCann |
Chief Executive Officer (Principal Executive Officer) |
CERTIFICATION PURSUANT TO
RULES 13a-14(a) AND 15d-14(a) UNDER THE SECURITIES EXCHANGE ACT OF 1934,
AS ADOPTED PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002
I, Christopher D.T. Guiffre, certify that:
1.I have reviewed this Annual Report on Form 10-K of Pear Therapeutics, Inc.;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statementstatements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
(a)Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b)[omitted]Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c)Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
(d)Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5.The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
(a)All significant deficiencies and material weaknesses in the design or operation of internal controlscontrol over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
(b)Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal controlscontrol over financial reporting.
Date: March 2831, 20222023
| | |
|
| /s/ Christopher D.T. Guiffre |
| Christopher D.T. Guiffre |
Chief Financial Officer (Principal Financial Officer) |
Certification of Periodic Financial Report
Pursuant to 18 U.S.C. Section 1350
as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
Each of the undersigned officers of Pear Therapeutics, Inc. (the “Company”) certifies, to his knowledge and solely for the purposes of 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that the Annual Report on Form 10-K of the Company for the year ended December 31, 20212022 complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934 and that information contained in the Form 10-K fairly presents, in all material respects, the financial condition and results of operations of the Company.
Dated: March 2831, 20222023
| | |
|
| /s/ Corey M. McCann |
| Dr. Corey M. McCann |
Chief Executive Officer (Principal Executive Officer) |
Dated: March 2831, 20222023
| | |
|
| /s/ Christopher D.T. Guiffre |
| Christopher D.T. Guiffre |
Chief Financial Officer (Principal Financial Officer) |
{
"instance": {
"pear-2021123120221231.htm": {
"axisCustom": 3,
"axisStandard": 4140,
"baseTaxonomies": {
"http://fasb.org/us-gaap/2022": 759,
"http://xbrl.sec.gov/dei/2022": 42
},
"contextCount": 293248,
"dts": {
"calculationLink": {
"local": [
"pear-20211231_cal20221231_cal.xml"
]
},
"definitionLink": {
"local": [
"pear-20211231_def20221231_def.xml"
]
},
"inline": {
"local": [
"pear-2021123120221231.htm"
]
},
"labelLink": {
"local": [
"pear-20211231_lab20221231_lab.xml"
]
},
"presentationLink": {
"local": [
"pear-20211231_pre20221231_pre.xml"
]
},
"schema": {
"local": [
"pear-2021123120221231.xsd"
],
"remote": [
"http://www.xbrl.org/2003/xbrl-instance-2003-12-31.xsd",
"http://www.xbrl.org/2003/xbrl-linkbase-2003-12-31.xsd",
"http://www.xbrl.org/2003/xl-2003-12-31.xsd",
"http://www.xbrl.org/2003/xlink-2003-12-31.xsd",
"http://www.xbrl.org/2005/xbrldt-2005.xsd",
"http://www.xbrl.org/2006/ref-2006-02-27.xsd",
"http://www.xbrl.org/lrr/role/negated-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/net-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/reference-2009-12-16.xsd",
"https://www.xbrl.org/2020/extensible-enumerations-2.0.xsd",
"https://www.xbrl.org/dtr/type/2020-01-21/types.xsd",
"https://xbrl.fasb.org/srt/20212022/elts/srt-2021-01-312022.xsd",
"https://xbrl.fasb.org/srt/20212022/elts/srt-roles-2021-01-312022.xsd",
"https://xbrl.fasb.org/srt/20212022/elts/srt-types-2021-01-312022.xsd",
"https://xbrl.fasb.org/us-gaap/20212022/elts/us-gaap-2021-01-312022.xsd",
"https://xbrl.fasb.org/us-gaap/20212022/elts/us-roles-2021-01-312022.xsd",
"https://xbrl.fasb.org/us-gaap/20212022/elts/us-types-2021-01-312022.xsd",
"https://xbrl.sec.gov/country/20212022/country-20212022.xsd",
"https://xbrl.sec.gov/dei/2021q42022/dei-2021q42022.xsd"
]
}
},
"elementCount": 680702,
"entityCount": 1,
"hidden": {
"http://fasb.org/us-gaap/2022": 1,
"http://xbrl.sec.gov/dei/2021q42022": 4,
"total": 45
},
"keyCustom": 6976,
"keyStandard": 328350,
"memberCustom": 5848,
"memberStandard": 5655,
"nsprefix": "pear",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"report": {
"R1": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:DocumentType",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "document",
"isDefault": "true",
"longName": "00010010000001 - Document - Cover",
"menuCat": "Cover",
"order": "1",
"role": "http://www.peartherapeutics.com/role/Cover",
"shortName": "Cover",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:DocumentType",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R10": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SignificantAccountingPoliciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000010 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES",
"menuCat": "Notes",
"order": "10",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIES",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SignificantAccountingPoliciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R11": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21121030000011 - Disclosure - BUSINESS COMBINATION",
"menuCat": "Notes",
"order": "11",
"role": "http://www.peartherapeutics.com/role/BUSINESSCOMBINATION",
"shortName": "BUSINESS COMBINATION",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R11R12": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueDisclosuresTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21171040000012 - Disclosure - FAIR VALUE MEASUREMENTS",
"menuCat": "Notes",
"order": "12",
"role": "http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTS",
"shortName": "FAIR VALUE MEASUREMENTS",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueDisclosuresTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R12R13": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21231050000013 - Disclosure - PROPERTY AND EQUIPMENT",
"menuCat": "Notes",
"order": "13",
"role": "http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENT",
"shortName": "PROPERTY AND EQUIPMENT",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R13R14": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:AccountsPayableAccruedLiabilitiesAndOtherLiabilitiesDisclosureCurrentTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21261060000014 - Disclosure - ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES",
"menuCat": "Notes",
"order": "14",
"role": "http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIES",
"shortName": "ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:AccountsPayableAccruedLiabilitiesAndOtherLiabilitiesDisclosureCurrentTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R14R15": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DebtDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21291070000015 - Disclosure - INDEBTEDNESS",
"menuCat": "Notes",
"order": "15",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESS",
"shortName": "INDEBTEDNESS",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DebtDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R15R16": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeasesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000016 - Disclosure - LEASES",
"menuCat": "Notes",
"order": "16",
"role": "http://www.peartherapeutics.com/role/LEASES",
"shortName": "LEASES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeasesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R17": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CommitmentsAndContingenciesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21351080000017 - Disclosure - COMMITMENTS AND CONTINGENCIES",
"menuCat": "Notes",
"order": "17",
"role": "http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIES",
"shortName": "COMMITMENTS AND CONTINGENCIES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CommitmentsAndContingenciesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R16R18": {
"firstAnchor": {
"ancestors": [
"us-gaap:RevenueFromContractWithCustomerTextBlock",
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i42ccea9f083b410fb9c7e5330a688a9b_D20190101-20191231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CollaborativeArrangementDisclosureTextBlockRevenueFromContractWithCustomerTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21391090000018 - Disclosure - PAST COLLABORATIONREVENUE AND LICENSE AGREEMENTCONTRACT BALANCES",
"menuCat": "Notes",
"order": "18",
"role": "http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTREVENUEANDCONTRACTBALANCES",
"shortName": "PAST COLLABORATIONREVENUE AND LICENSE AGREEMENTCONTRACT BALANCES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"us-gaap:RevenueFromContractWithCustomerTextBlock",
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i42ccea9f083b410fb9c7e5330a688a9b_D20190101-20191231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:CollaborativeArrangementDisclosureTextBlockRevenueFromContractWithCustomerTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R17R19": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:StockholdersEquityNoteDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21411100000019 - Disclosure - CAPITAL STOCK",
"menuCat": "Notes",
"order": "19",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCK",
"shortName": "CAPITAL STOCK",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:StockholdersEquityNoteDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R18R2": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2148111 - Disclosure - STOCK-BASED COMPENSATION",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATION",
"shortName": "STOCK-BASED COMPENSATION",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R19": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ic5469074105941a9a11743c0a2455c58_D20210101-20210331",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2155112 - Disclosure - INCOME TAXES",
"role": "http://www.peartherapeutics.com/role/INCOMETAXES",
"shortName": "INCOME TAXES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ic5469074105941a9a11743c0a2455c58_D20210101-20210331",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R2": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:AuditorName",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "document",
"isDefault": "false",
"longName": "00020020000002 - Document - Audit Information",
"menuCat": "Cover",
"order": "2",
"role": "http://www.peartherapeutics.com/role/AuditInformation",
"shortName": "Audit Information",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "dei:AuditorName",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R20": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000020 - Disclosure - STOCK-BASED COMPENSATION",
"menuCat": "Notes",
"order": "20",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATION",
"shortName": "STOCK-BASED COMPENSATION",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R21": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1f8f3040a25e4449b4ae0604a2067c17_D20220101-20220331",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000021 - Disclosure - INCOME TAXES",
"menuCat": "Notes",
"order": "21",
"role": "http://www.peartherapeutics.com/role/INCOMETAXES",
"shortName": "INCOME TAXES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1f8f3040a25e4449b4ae0604a2067c17_D20220101-20220331",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R22": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:EarningsPerShareTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21601130000022 - Disclosure - NET LOSS PER SHARE",
"menuCat": "Notes",
"order": "22",
"role": "http://www.peartherapeutics.com/role/NETLOSSPERSHARE",
"shortName": "NET LOSS PER SHARE",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:EarningsPerShareTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R21R23": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:RelatedPartyTransactionsDisclosureTextBlockRestructuringAndRelatedActivitiesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21641140000023 - Disclosure - RELATED-PARTY TRANSACTIONSRESTRUCTURING CHARGES",
"menuCat": "Notes",
"order": "23",
"role": "http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSRESTRUCTURINGCHARGES",
"shortName": "RELATED-PARTY TRANSACTIONSRESTRUCTURING CHARGES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:RelatedPartyTransactionsDisclosureTextBlockRestructuringAndRelatedActivitiesDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R22R24": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SubsequentEventsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21661150000024 - Disclosure - SUBSEQUENT EVENTS",
"menuCat": "Notes",
"order": "24",
"role": "http://www.peartherapeutics.com/role/SUBSEQUENTEVENTS",
"shortName": "SUBSEQUENT EVENTS",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SubsequentEventsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R23R25": {
"firstAnchor": {
"ancestors": [
"us-gaap:ConsolidationPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:BasisOfAccountingPolicyPolicyTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "22052010000025 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Policies)",
"menuCat": "Policies",
"order": "25",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Policies)",
"subGroupType": "policies",
"uniqueAnchor": {
"ancestors": [
"us-gaap:ConsolidationPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:BasisOfAccountingPolicyPolicyTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R24R26": {
"firstAnchor": {
"ancestors": [
"us-gaap:ScheduleOfRestrictedCashAndCashEquivalentsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23063010000026 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Tables)",
"menuCat": "Tables",
"order": "26",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESTables",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"us-gaap:ScheduleOfRestrictedCashAndCashEquivalentsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R25R27": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "pear:ScheduleOfReverseCapitalizationTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23133020000027 - Disclosure - BUSINESS COMBINATION (Tables)",
"menuCat": "Tables",
"order": "27",
"role": "http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONTables",
"shortName": "BUSINESS COMBINATION (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "pear:ScheduleOfReverseCapitalizationTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R26R28": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueByBalanceSheetGroupingTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23183030000028 - Disclosure - FAIR VALUE MEASUREMENTS (Tables)",
"menuCat": "Tables",
"order": "28",
"role": "http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSTables",
"shortName": "FAIR VALUE MEASUREMENTS (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueByBalanceSheetGroupingTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R27R29": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"us-gaap:PropertyPlantAndEquipmentPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentTextBlock",
"reportCount": 1,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23243040000029 - Disclosure - PROPERTY AND EQUIPMENT (Tables)",
"role": "http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTTables",
"shortName": "PROPERTY AND EQUIPMENT (Tables)",
"subGroupType": "tables",
"uniqueAnchor": null
},
"R28": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2327305 - Disclosure - ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES (Tables)",
"role": "http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESTables",
"shortName": "ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R29": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfDebtInstrumentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2330306 - Disclosure - INDEBTEDNESS (Tables)",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESSTables",
"shortName": "INDEBTEDNESS (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfDebtInstrumentsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R3": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"",
"menuCat": "Tables",
"order": "29",
"role": "http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTTables",
"shortName": "PROPERTY AND EQUIPMENT (Tables)",
"subGroupType": "tables",
"uniqueAnchor": null
},
"R3": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"us-gaap:ScheduleOfRestrictedCashAndCashEquivalentsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:CashAndCashEquivalentsAtCarryingValue",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "10010030000003 - Statement - UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OFBALANCE SHEETS",
FINANCIAL CONDITION"menuCat": "Statements",
"order": "3",
"role": "http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS",
"shortName": "UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF FINANCIAL CONDITIONBALANCE SHEETS",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:ShortTermInvestments",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R30": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfFutureMinimumRentalPaymentsForOperatingLeasesTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2336307 - Disclosure - COMMITMENTS AND CONTINGENCIES (Tables)ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000030 - Disclosure - ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES (Tables)",
"menuCat": "Tables",
"order": "30",
"role": "http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESTables",
"shortName": "ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R31": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "pear:DebtInstrumentDebtCovenantMinimumTotalRevenueTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000031 - Disclosure - INDEBTEDNESS (Tables)",
"menuCat": "Tables",
"order": "31",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESSTables",
"shortName": "INDEBTEDNESS (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "pear:DebtInstrumentDebtCovenantMinimumTotalRevenueTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R32": {
"firstAnchor": {
"ancestors": [
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000032 - Disclosure - LEASES (Tables)",
"menuCat": "Tables",
"order": "32",
"role": "http://www.peartherapeutics.com/role/LEASESTables",
"shortName": "LEASES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R33": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:RecordedUnconditionalPurchaseObligationsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000033 - Disclosure - COMMITMENTS AND CONTINGENCIES (Tables)",
"menuCat": "Tables",
"order": "33",
"role": "http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESTables",
"shortName": "COMMITMENTS AND CONTINGENCIES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfFutureMinimumRentalPaymentsForOperatingLeasesTableTextBlockRecordedUnconditionalPurchaseObligationsTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R31R34": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:TemporaryEquityTableTextBlockContractWithCustomerAssetAndLiabilityTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23423080000034 - Disclosure - CAPITAL STOCKREVENUE AND CONTRACT BALANCES (Tables)",
"menuCat": "Tables",
"order": "34",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCKTablesREVENUEANDCONTRACTBALANCESTables",
"shortName": "CAPITAL STOCKREVENUE AND CONTRACT BALANCES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:TemporaryEquityTableTextBlockContractWithCustomerAssetAndLiabilityTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R32R35": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlockScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23493090000035 - Disclosure - STOCK-BASED COMPENSATION (Tables)",
"menuCat": "Tables",
"order": "35",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONTables",
"shortName": "STOCK-BASED COMPENSATION (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlockScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R33R36": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "ic5469074105941a9a11743c0a2455c58_D20210101-20210331i1f8f3040a25e4449b4ae0604a2067c17_D20220101-20220331",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23563100000036 - Disclosure - INCOME TAXES (Tables)",
"menuCat": "Tables",
"order": "36",
"role": "http://www.peartherapeutics.com/role/INCOMETAXESTables",
"shortName": "INCOME TAXES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "ic5469074105941a9a11743c0a2455c58_D20210101-20210331i1f8f3040a25e4449b4ae0604a2067c17_D20220101-20220331",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R34R37": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "23613110000037 - Disclosure - NET LOSS PER SHARE (Tables)",
"menuCat": "Tables",
"order": "37",
"role": "http://www.peartherapeutics.com/role/NETLOSSPERSHARETables",
"shortName": "NET LOSS PER SHARE (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R35": {
"firstAnchor": {
"ancestors": [
"us-gaap:CommonStockParOrStatedValuePerShare",
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:CommonStockParOrStatedValuePerShare",
"reportCount": 1,
"unitRef": "usdPerShare",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2402401 - Disclosure - NATURE OF BUSINESS - Organization (Details)",
"role": "http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"shortName": "NATURE OF BUSINESS - Organization (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ia14ba0d74f86407999129ba32c975eb3_I20211203",
"decimals": "INF",
"lang": "en-US",
"name": "pear:BusinessCombinationConversionOfStockConversionRatio",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R36R38": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3"null,
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLoss",
"reportCount": 1,
"unitRef": "usdScheduleOfCostsOfRetirementPlansTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000038 - Disclosure - RESTRUCTURING CHARGES (Tables)",
"menuCat": "Tables",
"order": "38",
"role": "http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESTables",
"shortName": "RESTRUCTURING CHARGES (Tables)",
"subGroupType": "tables",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:ScheduleOfCostsOfRetirementPlansTableTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R39": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "pear:NumberOfProducts",
"reportCount": 1,
"unique": true,
"unitRef": "product",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24034020000039 - Disclosure - NATURE OF BUSINESS - Narrative (Details)",
"menuCat": "Details",
"order": "39",
"role": "http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails",
"shortName": "NATURE OF BUSINESS - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3"INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:CashCashEquivalentsAndShortTermInvestmentspear:NumberOfProducts",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R37": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"product",
"xsiNil": "false"
}
},
"R4": {
"firstAnchor": {
"ancestors": [
"us-gaap:PreferredStockParOrStatedValuePerShare",
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:PreferredStockParOrStatedValuePerShare",
"reportCount": 1,
"unitRef": "usdPerShare",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "0000004 - Statement - UNAUDITED CONDENSED CONSOLIDATED BALANCE SHEETS (Parenthetical)",
"menuCat": "Statements",
"order": "4",
"role": "http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical",
"shortName": "UNAUDITED CONDENSED CONSOLIDATED BALANCE SHEETS (Parenthetical)",
"subGroupType": "parenthetical",
"uniqueAnchor": null
},
"R40": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfCashAndCashEquivalentsTableTextBlock",
"us-gaap:ScheduleOfRestrictedCashAndCashEquivalentsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:CashAndCashEquivalentsAtCarryingValue",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24074030000040 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Cash, Cash Equivalents and Restricted Cash (Details)",
"menuCat": "Details",
"order": "40",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Cash, Cash Equivalents and Restricted Cash (Details)",
"subGroupType": "details",
"uniqueAnchor": null
},
"R38": {
"firstAnchor": {
"ancestors": [
"ix:continuation",
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AdvertisingExpense",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2408404 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Narrative (Details)",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AdvertisingExpense",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R39R41": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"us-gaap:PropertyPlantAndEquipmentPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i1e2fada15ec843a7abe31f0af39dabad_D20210101-20211231i2dab8e6da94c4e699d2a916cea670e86_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentUsefulLife",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24094050000041 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Property and Equipment (Details)",
"menuCat": "Details",
"order": "41",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Property and Equipment (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"us-gaap:PropertyPlantAndEquipmentPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i1e2fada15ec843a7abe31f0af39dabad_D20210101-20211231i2dab8e6da94c4e699d2a916cea670e86_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentUsefulLife",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R4R42": {
"firstAnchor": {
"ancestors": [
"span",
"divtd",
"ix:continuationtr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:PreferredStockParOrStatedValuePerShare",
"reportCount": 1,
"unitRef": "usdPerShare",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "1002004 - Statement - CONSOLIDATED STATEMENTS OF FINANCIAL CONDITION (Parenthetical)",
"role": "http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParenthetical",
"shortName": "CONSOLIDATED STATEMENTS OF FINANCIAL CONDITION (Parenthetical)",
"subGroupType": "parenthetical",
"uniqueAnchor": null
},
"R40": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:ClassOfWarrantOrRightOutstanding",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2410406 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Warrants and Revenue (Details)",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetails",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - Warrants and Revenue (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ia2d8e6f7855342a88decb74d426e9d78_D20210101-20211231",
"decimals": "2",
"lang": "en-US",
"name": "us-gaap:ConcentrationRiskPercentage1",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R41": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"us-gaap:NewAccountingPronouncementsPolicyPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i7482fb0f369844e5b669b10a459aecd6_I20220101",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeaseRightOfUseAsseti6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ImpairmentOfIntangibleAssetsExcludingGoodwill",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24114070000042 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - New Accounting PronouncementsNarrative (Details)",
"menuCat": "Details",
"order": "42",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES - New Accounting PronouncementsNarrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"span",
"div",
"us-gaap:NewAccountingPronouncementsPolicyPolicyTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7482fb0f369844e5b669b10a459aecd6_I20220101i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeaseRightOfUseAssetAdvertisingExpense",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R42R43": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"ix:continuationspan",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "pear:RecapitalizationExchangeRatious-gaap:CommonStockParOrStatedValuePerShare",
"reportCount": 1,
"unitRef": "numberusdPerShare",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24144080000043 - Disclosure - BUSINESS COMBINATION (Details)",
"menuCat": "Details",
"order": "43",
"role": "http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"shortName": "BUSINESS COMBINATION (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "ia4bee40d66e5440ab3df94ef33c59b47_D20211203i2cd8cf6133fb4dc884fb739d0bda50bf_D20211203-20211203",
"decimals": "INF-3",
"lang": "en-US",
"name": "us-gaap:BusinessAcquisitionEquityInterestsIssuedOrIssuableNumberOfSharesIssuedpear:ReverseRecapitalizationAccruedTransactionCosts",
"reportCount": 1,
"unique": true,
"unitRef": "sharesusd",
"xsiNil": "false"
}
},
"R43R44": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuationpear:ScheduleOfReverseCapitalizationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101i889587d81ccc44dcb00239a1b71f668a_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "pear:CashAcquiredThroughReverseRecapitalization",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24154090000044 - Disclosure - BUSINESS COMBINATION - Schedule of Reverse Capitalization (Details)",
"menuCat": "Details",
"order": "44",
"role": "http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"shortName": "BUSINESS COMBINATION - Schedule of Reverse Capitalization (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuationpear:ScheduleOfReverseCapitalizationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101i889587d81ccc44dcb00239a1b71f668a_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "pear:CashAcquiredThroughReverseRecapitalization",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R44R45": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "INF-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ClassOfWarrantOrRightOutstandingWarrantsAndRightsOutstanding",
"reportCount": 1,
"unitRef": "sharesusd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24164100000045 - Disclosure - BUSINESS COMBINATION - Warrants (Details)",
"menuCat": "Details",
"order": "45",
"role": "http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetails",
"shortName": "BUSINESS COMBINATION - Warrants (Details)",
"subGroupType": "details",
"uniqueAnchor": null
},
"R45R46": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueByBalanceSheetGroupingTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AvailableForSaleSecuritiesDebtSecurities",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24194110000046 - Disclosure - FAIR VALUE MEASUREMENTS - Assets and Fair Value (Details)",
"menuCat": "Details",
"order": "46",
"role": "http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails",
"shortName": "FAIR VALUE MEASUREMENTS - Assets and Fair Value (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueByBalanceSheetGroupingTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AvailableForSaleSecuritiesDebtSecurities",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R46R47": {
"firstAnchor": {
"ancestors": [
"span",
"divtd",
"ix:continuationtr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:OtherComprehensiveIncomeLossAvailableForSaleSecuritiesAdjustmentNetOfTax",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24204120000047 - Disclosure - FAIR VALUE MEASUREMENTS - Narrative (Details)",
"menuCat": "Details",
"order": "47",
"role": "http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"shortName": "FAIR VALUE MEASUREMENTS - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "id2f2687c5e5042fd9b97fa82b46b895c_I20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "INF-3",
"lang": "en-US",
"name": "us-gaap:DebtInstrumentMeasurementInputEmbeddedDerivativeGainOnEmbeddedDerivative",
"reportCount": 1,
"unique": true,
"unitRef": "numberusd",
"xsiNil": "false"
}
},
"R47R48": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuationus-gaap:FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iaa693b928ca4462b86cce094569e67da_I20211231ia1bd62e47a124b3c9876e29f4337072c_I20221231",
"decimals": "2",
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationContingentConsiderationLiabilityMeasurementInput",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24214130000048 - Disclosure - FAIR VALUE MEASUREMENTS - Valuation Assumptions (Details)",
"menuCat": "Details",
"order": "48",
"role": "http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails",
"shortName": "FAIR VALUE MEASUREMENTS - Valuation Assumptions (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuationus-gaap:FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iaa693b928ca4462b86cce094569e67da_I20211231ia1bd62e47a124b3c9876e29f4337072c_I20221231",
"decimals": "2",
"first": true,
"lang": "en-US",
"name": "us-gaap:BusinessCombinationContingentConsiderationLiabilityMeasurementInput",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R48R49": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7734b5067e0442e9bd6c788f9ddcba51_I20201231i94fe4c912bd34e1ba39e58165967fa4d_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueMeasurementWithUnobservableInputsReconciliationsRecurringBasisLiabilityValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24224140000049 - Disclosure - FAIR VALUE MEASUREMENTS - Level 3 Roll Forward (Details)",
"menuCat": "Details",
"order": "49",
"role": "http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails",
"shortName": "FAIR VALUE MEASUREMENTS - Level 3 Roll Forward (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i44cc880942254aa19111f721cad9e405_I20191231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:FairValueMeasurementWithUnobservableInputsReconciliationsRecurringBasisLiabilityValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R49": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentGross",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2425415 - Disclosure - PROPERTY AND EQUIPMENT (Details)",
"role": "http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"shortName": "PROPERTY AND EQUIPMENT (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentGross20221231.htm",
"contextRef": "i94fe4c912bd34e1ba39e58165967fa4d_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:FairValueMeasurementWithUnobservableInputsReconciliationsRecurringBasisLiabilityValue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R5": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:RevenueFromContractWithCustomerExcludingAssessedTax",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "10030050000005 - Statement - UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSSINCOME (LOSS)",
"menuCat": "Statements",
"order": "5",
"role": "http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS",
"shortName": "UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:RevenueFromContractWithCustomerExcludingAssessedTax",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R50": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentGross",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000050 - Disclosure - PROPERTY AND EQUIPMENT (Details)",
"menuCat": "Details",
"order": "50",
"role": "http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"shortName": "PROPERTY AND EQUIPMENT (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:PropertyPlantAndEquipmentGross",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R51": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:EmployeeRelatedLiabilitiesCurrent",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24284160000051 - Disclosure - ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES (Details)",
"menuCat": "Details",
"order": "51",
"role": "http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails",
"shortName": "ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:EmployeeRelatedLiabilitiesCurrent",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R51R52": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:FairValueByBalanceSheetGroupingTextBlockdiv",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:EmbeddedDerivativeFairValueOfEmbeddedDerivativeLiabilityDebtInstrumentFeeAmount",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24314170000052 - Disclosure - INDEBTEDNESS - Perceptive Credit Facility (Details)",
"menuCat": "Details",
"order": "52",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"shortName": "INDEBTEDNESS - Perceptive Credit Facility (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "ifea931feaa03413697518da188792ecf_I20211130i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "INF"-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ClassOfWarrantOrRightNumberOfSecuritiesCalledByWarrantsOrRightsDebtInstrumentFeeAmount",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R52usd",
"xsiNil": "false"
}
},
"R53": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"pear:DebtInstrumentDebtCovenantMinimumTotalRevenueTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i8081bffe17eb41f994d09411a8fcd062_D20231001-20231231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "pear:DebtInstrumentDebtCovenantMinimumTotalRevenue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000053 - Disclosure - INDEBTEDNESS - Minimum Revenue Covenant (Details)",
"menuCat": "Details",
"order": "53",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESSMinimumRevenueCovenantDetails",
"shortName": "INDEBTEDNESS - Minimum Revenue Covenant (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"pear:DebtInstrumentDebtCovenantMinimumTotalRevenueTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i8081bffe17eb41f994d09411a8fcd062_D20231001-20231231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "pear:DebtInstrumentDebtCovenantMinimumTotalRevenue",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R54": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:LongTermDebt",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24324180000054 - Disclosure - INDEBTEDNESS - Schedule of Debt (Details)",
"menuCat": "Details",
"order": "54",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails",
"shortName": "INDEBTEDNESS - Schedule of Debt (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i3e9cf9ae342243fa8f82e57678e87039_I20211231i6934df2b39ba47b7b262cc75fc0d7a18_I20221231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:DebtInstrumentCarryingAmount",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R53R55": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24334190000055 - Disclosure - INDEBTEDNESS - Future Minimum Payments Due (Details)",
"menuCat": "Details",
"order": "55",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails",
"shortName": "INDEBTEDNESS - Future Minimum Payments Due (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R54": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:GainsLossesOnExtinguishmentOfDebt",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2434420 - Disclosure - INDEBTEDNESS - SVB Term Loan (Details)",
"role": "http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"shortName": "INDEBTEDNESS - SVB Term Loan (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"ix:continuation",
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i34151ce77cec4273ba9b8f63f5be8b09_I20211202",
"decimals": "INF",
"lang": "en-US",
"name": "us-gaap:SharesIssued",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R55R56": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3"null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LeaseAndRentalExpenseLesseeOperatingLeaseRenewalTerm",
"reportCount": 1,
"unique": true,
"unitRef": "usd"null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24374210000056 - Disclosure - COMMITMENTS AND CONTINGENCIES - NarrativeLEASE - (Details)",
"menuCat": "Details",
"order": "56",
"role": "http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails",
"shortName": "COMMITMENTS AND CONTINGENCIES - NarrativeLEASE - (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:LeaseAndRentalExpensei1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseRenewalTerm",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R57": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"us-gaap:LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000057 - Disclosure - LEASES - Maturity Table (Details)",
"menuCat": "Details",
"order": "57",
"role": "http://www.peartherapeutics.com/role/LEASESMaturityTableDetails",
"shortName": "LEASES - Maturity Table (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"us-gaap:LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R56R58": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"divix:continuation",
"us-gaap:ScheduleOfFutureMinimumRentalPaymentsForOperatingLeasesTableTextBlockdiv",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i5359e8cc6c724c1783cb31077b7672a0_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeasesFutureMinimumPaymentsDueCurrent",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24384220000058 - Disclosure - COMMITMENTS AND CONTINGENCIESLEASES - Operating Leases (Details)",
"menuCat": "Details",
"order": "58",
"role": "http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails",
"shortName": "COMMITMENTS AND CONTINGENCIESLEASES - Operating Leases (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"divix:continuation",
"us-gaap:ScheduleOfFutureMinimumRentalPaymentsForOperatingLeasesTableTextBlockdiv",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i5359e8cc6c724c1783cb31077b7672a0_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:OperatingLeasesFutureMinimumPaymentsDueCurrent",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R57R59": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i463ddfce35014df1b3bca85573252a6e_D20180301-20180331i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:SaleOfStockNumberOfSharesIssuedInTransactionpear:NumberOfLicenseAgreements",
"reportCount": 1,
"unique": true,
"unitRef": "shareslicense_agreement",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24404230000059 - Disclosure - PAST COLLABORATIONCOMMITMENTS AND LICENSE AGREEMENTCONTINGENCIES - NovartisNarrative (Details)",
"menuCat": "Details",
"order": "59",
"role": "http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsCOMMITMENTSANDCONTINGENCIESNarrativeDetails",
"shortName": "PAST COLLABORATIONCOMMITMENTS AND LICENSE AGREEMENTCONTINGENCIES - NovartisNarrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"ix:continuationbody",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i525aa77976dd40d09ff91a8b7cc0edd3_D20220101-20221231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:ImpairmentOfIntangibleAssetsExcludingGoodwill",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R6": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i463ddfce35014df1b3bca85573252a6e_D20180301-20180331",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:SaleOfStockNumberOfSharesIssuedInTransaction",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R58": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231ic147dd7878c64739a85d46d84292c40c_I20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:SharesOutstanding",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "0000006 - Statement - UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS\u2019 EQUITY",
"menuCat": "Statements",
"order": "6",
"role": "http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"shortName": "UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS\u2019 EQUITY",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "ic147dd7878c64739a85d46d84292c40c_I20201231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:SharesOutstanding",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R60": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:PurchaseObligationDueInNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000060 - Disclosure - COMMITMENTS AND CONTINGENCIES - Unconditional Purchase Commitment (Details)",
"menuCat": "Details",
"order": "60",
"role": "http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESUnconditionalPurchaseCommitmentDetails",
"shortName": "COMMITMENTS AND CONTINGENCIES - Unconditional Purchase Commitment (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:PurchaseObligationDueInNextTwelveMonths",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R61": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ContractWithCustomerAssetNet",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000061 - Disclosure - REVENUE AND CONTRACT BALANCES (Details)",
"menuCat": "Details",
"order": "61",
"role": "http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails",
"shortName": "REVENUE AND CONTRACT BALANCES (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:ContractWithCustomerAssetNet",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R62": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:CommonStockSharesAuthorized",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24434240000062 - Disclosure - CAPITAL STOCK (Details)",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"shortName": "CAPITAL STOCK (Details)",
"subGroupType": "details",
"uniqueAnchor": null
},
"R59": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ie03abf4b7549420fbcd4e9b4389dafab_I20211202",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:TemporaryEquitySharesAuthorized",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2444425 - Disclosure - CAPITAL STOCK - Schedule of Convertible Stock (Details)",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"shortName": "CAPITAL STOCK - Schedule of Convertible Stock (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"us-gaap:TemporaryEquitySharesIssued",
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ie03abf4b7549420fbcd4e9b4389dafab_I20211202",
"decimals": "INF",
"lang": "en-US",
"name": "us-gaap:TemporaryEquitySharesOutstanding",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R6": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i6371d804e77e4dd7a6bf28b15f56146d_I20191231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:TemporaryEquitySharesOutstanding",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "1004006 - Statement - CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS\u2019 DEFICIT",
"role": "http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",
"shortName": "CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS\u2019 DEFICIT",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i6371d804e77e4dd7a6bf28b15f56146d_I20191231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:TemporaryEquitySharesOutstanding",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R60": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ie03abf4b7549420fbcd4e9b4389dafab_I20211202",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:TemporaryEquityLiquidationPreferencePerShare",
"reportCount": 1,
"unitRef": "usdPerShare",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2445426 - Disclosure - CAPITAL STOCK - Additional Information (Details)",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"shortName": "CAPITAL STOCK - Additional Information (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231",
"menuCat": "Details",
"order": "62",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"shortName": "CAPITAL STOCK (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "ie03abf4b7549420fbcd4e9b4389dafab_I20211202",
"decimals": "INF",
"lang": "en-US",
"name": "pear:PreferredStockConversionTriggeringNetProceedsMinimum",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R61": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"ix:continuation",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ie03abf4b7549420fbcd4e9b4389dafab_I20211202",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:TemporaryEquitySharesAuthorized",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2446427 - Disclosure - CAPITAL STOCK - Convertible Preferred Stock Issuance (Details)",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"shortName": "CAPITAL STOCK - Convertible Preferred Stock Issuance (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i18ea874c05da4ccc9495a6d3835a15f5_I20201102",
"decimals": "INF",
"lang": "en-US",
"name": "pear:TenderOfferSharesAvailableToPurchase",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R62": {
"firstAnchor": {
"ancestors": [
"us-gaap:CommonStockParOrStatedValuePerShare",
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:CommonStockParOrStatedValuePerShare",
"reportCount": 1,
"unitRef": "usdPerShare",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2447428 - Disclosure - CAPITAL STOCK - Warrants (Details)",
"role": "http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails",
"shortName": "CAPITAL STOCK - Warrants (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "ibbfa06a200d145a58920d2dc357ac7ac_D20210101-20211231i8749a4ae968549a2b747b31568e905e8_D20211203-20211203",
"decimals": "INF",
"lang": "en-US",
"name": "pear:ClassOfWarrantOrRightsIssuePricePerWarrant",
"reportCount": 1,
"unique": true,
"unitRef": "usdPerShare",
"xsiNil": "false"
}
},
"R63": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AllocatedShareBasedCompensationExpense",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24504290000063 - Disclosure - STOCK-BASED COMPENSATION - Narrative (Details)",
"menuCat": "Details",
"order": "63",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"shortName": "STOCK-BASED COMPENSATION - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "2",
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageGrantDateFairValue",
"reportCount": 1,
"unique": true,
"unitRef": "usdPerShare",
"xsiNil": "false"
}
},
"R64null,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardRequisiteServicePeriod1",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R64": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i63613787540047419b4acbdb3d2c179a_D20220101-20221231",
"decimals": "4",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000064 - Disclosure - STOCK-BASED COMPENSATION - Valuation Assumptions (Details)",
"menuCat": "Details",
"order": "64",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails",
"shortName": "STOCK-BASED COMPENSATION - Valuation Assumptions (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i63613787540047419b4acbdb3d2c179a_D20220101-20221231",
"decimals": "4",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R65": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "ic8435b1060a84f60855dec1d15e3a8c1_I20201231i5359e8cc6c724c1783cb31077b7672a0_I20211231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber",
"reportCount": 1,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24514300000065 - Disclosure - STOCK-BASED COMPENSATION - Common Stock Options (Details)",
"menuCat": "Details",
"order": "65",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails",
"shortName": "STOCK-BASED COMPENSATION - Common Stock Options (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "INF",
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresAndExpirationsInPeriod",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R65R66": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i320e4d2c79b24ee4ac145b68ecf9c778_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "4INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross",
"reportCount": 1,
"unique": true,
"unitRef": "numbershares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24524310000066 - Disclosure - STOCK-BASED COMPENSATION - Valuation AssumptionsRestricted Stock Units (Details)",
"menuCat": "Details",
"order": "66",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetailsSTOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails",
"shortName": "STOCK-BASED COMPENSATION - Valuation AssumptionsRestricted Stock Units (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"divus-gaap:ScheduleOfSharebasedCompensationRestrictedStockAndRestrictedStockUnitsActivityTableTextBlock",
"us-gaap:ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlockdiv",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i320e4d2c79b24ee4ac145b68ecf9c778_D20210101-20211231ifdb9e7ee2dd74323b60954440d310710_D20220101-20221231",
"decimals": "4",
"first": trueINF",
"lang": "en-US",
"name": "us-gaap:ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeitedInPeriod",
"reportCount": 1,
"unique": true,
"unitRef": "numbershares",
"xsiNil": "false"
}
},
"R66R67": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AllocatedShareBasedCompensationExpense",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2453432 - Disclosure - STOCK-BASED COMPENSATION - Stock Based Compensation Expense (Details)0000067 - Disclosure - STOCK-BASED COMPENSATION - ESPP (Details)",
"menuCat": "Details",
"order": "67",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"shortName": "STOCK-BASED COMPENSATION - ESPP (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "id82451edf1bb4857b4d7bf26aa356ed0_D20220101-20221231",
"decimals": "INF",
"lang": "en-US",
"name": "us-gaap:SharebasedCompensationArrangementBySharebasedPaymentAwardPurchasePriceOfCommonStockPercent",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R68": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AllocatedShareBasedCompensationExpense",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000068 - Disclosure - STOCK-BASED COMPENSATION - Stock Based Compensation Expense (Details)",
"menuCat": "Details",
"order": "68",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails",
"shortName": "STOCK-BASED COMPENSATION - Stock Based Compensation Expense (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "ie12ef8a78f6b49e39d595a7d9a729a4e_D20210101-20211231i525aa77976dd40d09ff91a8b7cc0edd3_D20220101-20221231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:AllocatedShareBasedCompensationExpense",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R67": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i54fdb71991ac4cb581e23c5e6cec7d21_D20210101-20211231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "us-gaap:SharebasedCompensationArrangementBySharebasedPaymentAwardPurchasePriceOfCommonStockPercent",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2454433 - Disclosure - STOCK-BASED COMPENSATION - ESPP (Details)",
"role": "http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"shortName": "STOCK-BASED COMPENSATION - ESPP (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i54fdb71991ac4cb581e23c5e6cec7d21_D20210101-20211231",
"decimals": "3",
"first": true,
"lang": "en-US",
"name": "us-gaap:SharebasedCompensationArrangementBySharebasedPaymentAwardPurchasePriceOfCommonStockPercent",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R68R69": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24574340000069 - Disclosure - INCOME TAXES - Reconciliation of Tax Rate (Details)",
"menuCat": "Details",
"order": "69",
"role": "http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails",
"shortName": "INCOME TAXES - Reconciliation of Tax Rate (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate",
"reportCount": 1,
"unique": true,
"unitRef": "number",
"xsiNil": "false"
}
},
"R69": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuationR7": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i26e6e0f021cc4affaa9c28e1c7a7ff0b_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:AdjustmentsToAdditionalPaidInCapitalStockIssuedIssuanceCosts",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "0000007 - Statement - UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS\u2019 EQUITY (Parenthetical)",
"menuCat": "Statements",
"order": "7",
"role": "http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical",
"shortName": "UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS\u2019 EQUITY (Parenthetical)",
"subGroupType": "parenthetical",
"uniqueAnchor": null
},
"R70": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"us-gaap:ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:DeferredTaxAssetsOperatingLossCarryforwards",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24584350000070 - Disclosure - INCOME TAXES - Deferred Tax Assets (Details)",
"menuCat": "Details",
"order": "70",
"role": "http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails",
"shortName": "INCOME TAXES - Deferred Tax Assets (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuationus-gaap:ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i7fc056c5d63a414b8c64487754d32f20_I20211231i1ddf60ff01eb488a80fd575c0d2e6e99_I20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:DeferredTaxAssetsOperatingLossCarryforwards",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R7R71": {
"firstAnchor": {
"ancestors": [
"us-gaap:IncomeTaxExpenseBenefit",
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLoss",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "1006008 - Statement - CONSOLIDATED STATEMENTS OF CASH FLOWS",
"role": "http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"shortName": "CONSOLIDATED STATEMENTS OF CASH FLOWS",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:AmortizationOfIntangibleAssets",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R70": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxExpenseBenefit",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24594360000071 - Disclosure - INCOME TAXES - Additional Information (Details)",
"menuCat": "Details",
"order": "71",
"role": "http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails",
"shortName": "INCOME TAXES - Additional Information (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"us-gaap:IncomeTaxExpenseBenefit",
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:IncomeTaxExpenseBenefit",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R71R72": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"divix:continuation",
"ix:continuationdiv",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "0",
"first": true,
"lang": "en-US",
"name": "us-gaap:AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareAmount",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "24624370000072 - Disclosure - NET LOSS PER SHARE - AntidilituveAntidilutive Securities (Details)",
"menuCat": "Details",
"order": "72",
"role": "http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails",
"shortName": "NET LOSS PER SHARE - AntidilituveAntidilutive Securities (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"divix:continuation",
"ix:continuationdiv",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "0",
"first": true,
"lang": "en-US",
"name": "us-gaap:AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareAmount",
"reportCount": 1,
"unique": true,
"unitRef": "shares",
"xsiNil": "false"
}
},
"R72": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231R73": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLossAvailableToCommonStockholdersBasic",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000073 - Disclosure - NET LOSS PER SHARE - Basic and Diluted Per Share (Details)",
"menuCat": "Details",
"order": "73",
"role": "http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails",
"shortName": "NET LOSS PER SHARE - Basic and Diluted Per Share (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLossAvailableToCommonStockholdersBasic",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R74": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLoss",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosurei075323ce068a4cbbbbba3fdf34b29749_D20221114-20221114",
"decimals": "INF",
"first": true,
"lang": "en-US",
"name": "us-gaap:RestructuringAndRelatedCostNumberOfPositionsEliminated",
"reportCount": 1,
"unitRef": "employee",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000074 - Disclosure - RESTRUCTURING CHARGES - Narrative (Details)",
"menuCat": "Details",
"order": "74",
"role": "http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESNarrativeDetails",
"shortName": "RESTRUCTURING CHARGES - Narrative (Details)",
"subGroupType": "details",
"uniqueAnchor": null
},
"R75": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:CompensationExpenseExcludingCostOfGoodAndServiceSold",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000075 - Disclosure - RESTRUCTURING CHARGES - Schedule of Severance, Employee Benefits, and Related Costs (Details)",
"menuCat": "Details",
"order": "75",
"role": "http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails",
"shortName": "RESTRUCTURING CHARGES - Schedule of Severance, Employee Benefits, and Related Costs (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"ix:continuation",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:CompensationExpenseExcludingCostOfGoodAndServiceSold",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R76": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfRestructuringReserveByTypeOfCostTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i5359e8cc6c724c1783cb31077b7672a0_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:RestructuringReserve",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "0000076 - Disclosure - RESTRUCTURING CHARGES - Restructuring Liability Roll forward (Details)",
"menuCat": "Details",
"order": "76",
"role": "http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESRestructuringLiabilityRollforwardDetails",
"shortName": "RESTRUCTURING CHARGES - Restructuring Liability Roll forward (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"us-gaap:ScheduleOfRestructuringReserveByTypeOfCostTextBlock",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i5359e8cc6c724c1783cb31077b7672a0_I20211231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:RestructuringReserve",
"reportCount": 1,
"unique": true,
"unitRef": "usd",
"xsiNil": "false"
}
},
"R8": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20221231.htm",
"contextRef": "i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:NetIncomeLoss",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "statement",
"isDefault": "false",
"longName": "24634380000008 - DisclosureStatement - NET LOSS PER SHARE -UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF Basic andCASH FLOWS",
Diluted Per"menuCat": "Statements",
Share (Details)"order": "8",
"role": "http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"shortName": "NET LOSS PER SHARE -UNAUDITED CONDENSED CONSOLIDATED STATEMENTS OF Basic and Diluted Per Share (Details)CASH FLOWS",
"subGroupType": "details",
"uniqueAnchor": null
},
"R73": {
"firstAnchor": {
"ancestors": [
"span",
"td",
"tr",
"table",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "i365d1f181c614fb6935bfa00b721a728_D20200101-20201231",
"decimals": "-3",
"first": true,
"lang": "en-US",
"name": "us-gaap:StockRepurchasedDuringPeriodValue",
"reportCount": 1,
"unitRef": "usd",
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2465439 - Disclosure - RELATED-PARTY TRANSACTIONS (Details)",
"role": "http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"shortName": "RELATED-PARTY TRANSACTIONS (Details)",
"subGroupType": "details",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"ix:continuation",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "i75a6000e9e7f47dd9990c37a17404cfe_D20201201-20201231",
"decimals": "INF",
"lang": "en-US",
"name": "us-gaap:StockRepurchasedDuringPeriodSharesi6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": "-3",
"lang": "en-US",
"name": "us-gaap:AmortizationOfIntangibleAssets",
"reportCount": 1,
"unique": true,
"unitRef": "sharesusd",
"xsiNil": "false"
}
},
"R8R9": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:OrganizationConsolidationAndPresentationOfFinancialStatementsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "21011010000009 - Disclosure - NATURE OF BUSINESS",
"menuCat": "Notes",
"order": "9",
"role": "http://www.peartherapeutics.com/role/NATUREOFBUSINESS",
"shortName": "NATURE OF BUSINESS",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-2021123120221231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231i6ae7b90e50804df59757cb3e12caa92d_D20220101-20221231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:OrganizationConsolidationAndPresentationOfFinancialStatementsDisclosureTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
},
"R9": {
"firstAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SignificantAccountingPoliciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
},
"groupType": "disclosure",
"isDefault": "false",
"longName": "2104102 - Disclosure - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES",
"role": "http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIES",
"shortName": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES",
"subGroupType": "",
"uniqueAnchor": {
"ancestors": [
"span",
"div",
"body",
"html"
],
"baseRef": "pear-20211231.htm",
"contextRef": "iee3b22b8ea4f4e898451482b7f0241ff_D20210101-20211231",
"decimals": null,
"first": true,
"lang": "en-US",
"name": "us-gaap:SignificantAccountingPoliciesTextBlock",
"reportCount": 1,
"unique": true,
"unitRef": null,
"xsiNil": "false"
}
}
},
"segmentCount": 126
},
"segmentCount": 109,
"tag": {
"dei_AmendmentFlag": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the XBRL content amends previously-filed or accepted submission.",
"label": "Amendment Flag",
"terseLabel": "Amendment Flag"
}
}
},
"localname": "AmendmentFlag",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_AuditorFirmId": {
"auth_ref": [
"r699r687",
"r700r688",
"r701r689"
],
"lang": {
"en-us": {
"role": {
"documentation": "PCAOB issued Audit Firm Identifier",
"label": "Auditor Firm ID",
"terseLabel": "Auditor Firm ID"
}
}
},
"localname": "AuditorFirmId",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/AuditInformation"
],
"xbrltype": "nonemptySequenceNumberItemType"
},
"dei_AuditorLocation": {
"auth_ref": [
"r699r687",
"r700r688",
"r701r689"
],
"lang": {
"en-us": {
"role": {
"label": "Auditor Location",
"terseLabel": "Auditor Location"
}
}
},
"localname": "AuditorLocation",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/AuditInformation"
],
"xbrltype": "internationalNameItemType"
},
"dei_AuditorName": {
"auth_ref": [
"r699r687",
"r700r688",
"r701r689"
],
"lang": {
"en-us": {
"role": {
"label": "Auditor Name",
"terseLabel": "Auditor Name"
}
}
},
"localname": "AuditorName",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/AuditInformation"
],
"xbrltype": "internationalNameItemType"
},
"dei_CityAreaCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Area code of city",
"label": "City Area Code",
"terseLabel": "City Area Code"
}
}
},
"localname": "CityAreaCode",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_CoverAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Cover page.",
"label": "Cover [Abstract]"
}
}
},
"localname": "CoverAbstract",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"xbrltype": "stringItemType"
},
"dei_CurrentFiscalYearEndDate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "End date of current fiscal year in the format --MM-DD.",
"label": "Current Fiscal Year End Date",
"terseLabel": "Current Fiscal Year End Date"
}
}
},
"localname": "CurrentFiscalYearEndDate",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "gMonthDayItemType"
},
"dei_DocumentAnnualReport": {
"auth_ref": [
"r699r687",
"r700r688",
"r701r689"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as an annual report.",
"label": "Document Annual Report",
"terseLabel": "Document Annual Report"
}
}
},
"localname": "DocumentAnnualReport",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentFiscalPeriodFocus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Fiscal period values are FY, Q1, Q2, and Q3. 1st, 2nd and 3rd quarter 10-Q or 10-QT statements have value Q1, Q2, and Q3 respectively, with 10-K, 10-KT or other fiscal year statements having FY.",
"label": "Document Fiscal Period Focus",
"terseLabel": "Document Fiscal Period Focus"
}
}
},
"localname": "DocumentFiscalPeriodFocus",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "fiscalPeriodItemType"
},
"dei_DocumentFiscalYearFocus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "This is focus fiscal year of the document report in YYYY format. For a 2006 annual report, which may also provide financial information from prior periods, fiscal 2006 should be given as the fiscal year focus. Example: 2006.",
"label": "Document Fiscal Year Focus",
"terseLabel": "Document Fiscal Year Focus"
}
}
},
"localname": "DocumentFiscalYearFocus",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "gYearItemType"
},
"dei_DocumentInformationLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Document Information [Line Items]",
"terseLabel": "Document Information [Line Items]"
}
}
},
"localname": "DocumentInformationLineItems",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "stringItemType"
},
"dei_DocumentInformationTable": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Container to support the formal attachment of each official or unofficial, public or private document as part of a submission package.",
"label": "Document Information [Table]",
"terseLabel": "Document Information [Table]"
}
}
},
"localname": "DocumentInformationTable",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "stringItemType"
},
"dei_DocumentPeriodEndDate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "For the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.",
"label": "Document Period End Date",
"terseLabel": "Document Period End Date"
}
}
},
"localname": "DocumentPeriodEndDate",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "dateItemType"
},
"dei_DocumentTransitionReport": {
"auth_ref": [
"r702r690"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true only for a form used as a transition report.",
"label": "Document Transition Report",
"terseLabel": "Document Transition Report"
}
}
},
"localname": "DocumentTransitionReport",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_DocumentType": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.",
"label": "Document Type",
"terseLabel": "Document Type"
}
}
},
"localname": "DocumentType",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "submissionTypeItemType"
},
"dei_DocumentsIncorporatedByReferenceTextBlock": {
"auth_ref": [
"r697r685"
],
"lang": {
"en-us": {
"role": {
"documentation": "Documents incorporated by reference.",
"label": "Documents Incorporated by Reference [Text Block]",
"terseLabel": "Documents Incorporated by Reference"
}
}
},
"localname": "DocumentsIncorporatedByReferenceTextBlock",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "textBlockItemType"
},
"dei_EntityAddressAddressLine1": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Address Line 1 such as Attn, Building Name, Street Name",
"label": "Entity Address, Address Line One",
"terseLabel": "Entity Address, Address Line One"
}
}
},
"localname": "EntityAddressAddressLine1",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressAddressLine2": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Address Line 2 such as Street or Suite number",
"label": "Entity Address, Address Line Two",
"terseLabel": "Entity Address, Address Line Two"
}
}
},
"localname": "EntityAddressAddressLine2",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressCityOrTown": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the City or Town",
"label": "Entity Address, City or Town",
"terseLabel": "Entity Address, City or Town"
}
}
},
"localname": "EntityAddressCityOrTown",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressPostalZipCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Code for the postal or zip code",
"label": "Entity Address, Postal Zip Code",
"terseLabel": "Entity Address, Postal Zip Code"
}
}
},
"localname": "EntityAddressPostalZipCode",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityAddressStateOrProvince": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the state or province.",
"label": "Entity Address, State or Province",
"terseLabel": "Entity Address, State or Province"
}
}
},
"localname": "EntityAddressStateOrProvince",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "stateOrProvinceItemType"
},
"dei_EntityCentralIndexKey": {
"auth_ref": [
"r696r684"
],
"lang": {
"en-us": {
"role": {
"documentation": "A unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK.",
"label": "Entity Central Index Key",
"terseLabel": "Entity Central Index Key"
}
}
},
"localname": "EntityCentralIndexKey",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "centralIndexKeyItemType"
},
"dei_EntityCommonStockSharesOutstanding": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate number of shares or other units outstanding of each of registrant's classes of capital or common stock or other ownership interests, if and as stated on cover of related periodic report. Where multiple classes or units exist define each class/interest by adding class of stock items such as Common Class A [Member], Common Class B [Member] or Partnership Interest [Member] onto the Instrument [Domain] of the Entity Listings, Instrument.",
"label": "Entity Common Stock, Shares Outstanding",
"terseLabel": "Entity Common Stock, Shares Outstanding"
}
}
},
"localname": "EntityCommonStockSharesOutstanding",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "sharesItemType"
},
"dei_EntityCurrentReportingStatus": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' whether registrants (1) have filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that registrants were required to file such reports), and (2) have been subject to such filing requirements for the past 90 days. This information should be based on the registrant's current or most recent filing containing the related disclosure.",
"label": "Entity Current Reporting Status",
"terseLabel": "Entity Current Reporting Status"
}
}
},
"localname": "EntityCurrentReportingStatus",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "All the names of the entities being reported upon in a document. Any legal structure used to conduct activities or to hold assets. Some examples of such structures are corporations, partnerships, limited liability companies, grantor trusts, and other trusts. This item does not include business and geographical segments which are included in the geographical or business segments domains.",
"label": "Entity [Domain]",
"terseLabel": "Entity [Domain]"
}
}
},
"localname": "EntityDomain",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "domainItemType"
},
"dei_EntityEmergingGrowthCompany": {
"auth_ref": [
"r696r684"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate if registrant meets the emerging growth company criteria.",
"label": "Entity Emerging Growth Company",
"terseLabel": "Entity Emerging Growth Company"
}
}
},
"localname": "EntityEmergingGrowthCompany",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntityExTransitionPeriod": {
"auth_ref": [
"r712r693"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate if an emerging growth company has elected not to use the extended transition period for complying with any new or revised financial accounting standards.",
"label": "Entity Ex Transition Period",
"terseLabel": "Entity Ex Transition Period"
}
}
},
"localname": "EntityExTransitionPeriod",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntityFileNumber": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Commission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.",
"label": "Entity File Number",
"terseLabel": "Entity File Number"
}
}
},
"localname": "EntityFileNumber",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "fileNumberItemType"
},
"dei_EntityFilerCategory": {
"auth_ref": [
"r696r684"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate whether the registrant is one of the following: Large Accelerated Filer, Accelerated Filer, Non-accelerated Filer. Definitions of these categories are stated in Rule 12b-2 of the Exchange Act. This information should be based on the registrant's current or most recent filing containing the related disclosure.",
"label": "Entity Filer Category",
"terseLabel": "Entity Filer Category"
}
}
},
"localname": "EntityFilerCategory",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "filerCategoryItemType"
},
"dei_EntityIncorporationStateCountryCode": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Two-character EDGAR code representing the state or country of incorporation.",
"label": "Entity Incorporation, State or Country Code",
"terseLabel": "Entity Incorporation, State or Country Code"
}
}
},
"localname": "EntityIncorporationStateCountryCode",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "edgarStateCountryItemType"
},
"dei_EntityInteractiveDataCurrent": {
"auth_ref": [
"r710r691"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).",
"label": "Entity Interactive Data Current",
"terseLabel": "Entity Interactive Data Current"
}
}
},
"localname": "EntityInteractiveDataCurrent",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityPublicFloat": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and asked price of such common equity, as of the last business day of the registrant's most recently completed second fiscal quarter.",
"label": "Entity Public Float",
"terseLabel": "Entity Public Float"
}
}
},
"localname": "EntityPublicFloat",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "monetaryItemType"
},
"dei_EntityRegistrantName": {
"auth_ref": [
"r696r684"
],
"lang": {
"en-us": {
"role": {
"documentation": "The exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC.",
"label": "Entity Registrant Name",
"terseLabel": "Entity Registrant Name"
}
}
},
"localname": "EntityRegistrantName",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_EntityShellCompany": {
"auth_ref": [
"r696r684"
],
"lang": {
"en-us": {
"role": {
"documentation": "Boolean flag that is true when the registrant is a shell company as defined in Rule 12b-2 of the Exchange Act.",
"label": "Entity Shell Company",
"terseLabel": "Entity Shell Company"
}
}
},
"localname": "EntityShellCompany",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntitySmallBusiness": {
"auth_ref": [
"r696r684"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicates that the company is a Smaller Reporting Company (SRC).",
"label": "Entity Small Business",
"terseLabel": "Entity Small Business"
}
}
},
"localname": "EntitySmallBusiness",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_EntityTaxIdentificationNumber": {
"auth_ref": [
"r696r684"
],
"lang": {
"en-us": {
"role": {
"documentation": "The Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS.",
"label": "Entity Tax Identification Number",
"terseLabel": "Entity Tax Identification Number"
}
}
},
"localname": "EntityTaxIdentificationNumber",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "employerIdItemType"
},
"dei_EntityVoluntaryFilers": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.",
"label": "Entity Voluntary Filers",
"terseLabel": "Entity Voluntary Filers"
}
}
},
"localname": "EntityVoluntaryFilers",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_EntityWellKnownSeasonedIssuer": {
"auth_ref": [
"r711r692"
],
"lang": {
"en-us": {
"role": {
"documentation": "Indicate 'Yes' or 'No' if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Is used on Form Type: 10-K, 10-Q, 8-K, 20-F, 6-K, 10-K/A, 10-Q/A, 20-F/A, 6-K/A, N-CSR, N-Q, N-1A.",
"label": "Entity Well-known Seasoned Issuer",
"terseLabel": "Entity Well-known Seasoned Issuer"
}
}
},
"localname": "EntityWellKnownSeasonedIssuer",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "yesNoItemType"
},
"dei_IcfrAuditorAttestationFlag": {
"auth_ref": [
"r699r687",
"r700r688",
"r701r689"
],
"lang": {
"en-us": {
"role": {
"label": "ICFR Auditor Attestation Flag",
"terseLabel": "ICFR Auditor Attestation Flag"
}
}
},
"localname": "IcfrAuditorAttestationFlag",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "booleanItemType"
},
"dei_LegalEntityAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The set of legal entities associated with a report.",
"label": "Legal Entity [Axis]",
"terseLabel": "Legal Entity [Axis]"
}
}
},
"localname": "LegalEntityAxis",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"dei_LocalPhoneNumber": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Local phone number for entity.",
"label": "Local Phone Number",
"terseLabel": "Local Phone Number"
}
}
},
"localname": "LocalPhoneNumber",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "normalizedStringItemType"
},
"dei_Security12bTitle": {
"auth_ref": [
"r695r683"
],
"lang": {
"en-us": {
"role": {
"documentation": "Title of a 12(b) registered security.",
"label": "Title of 12(b) Security",
"terseLabel": "Title of 12(b) Security"
}
}
},
"localname": "Security12bTitle",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "securityTitleItemType"
},
"dei_SecurityExchangeName": {
"auth_ref": [
"r698r686"
],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the Exchange on which a security is registered.",
"label": "Security Exchange Name",
"terseLabel": "Security Exchange Name"
}
}
},
"localname": "SecurityExchangeName",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "edgarExchangeCodeItemType"
},
"dei_TradingSymbol": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Trading symbol of an instrument as listed on an exchange.",
"label": "Trading Symbol",
"terseLabel": "Trading Symbol"
}
}
},
"localname": "TradingSymbol",
"nsuri": "http://xbrl.sec.gov/dei/2021q42022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover"
],
"xbrltype": "tradingSymbolItemType"
},
"pear_A2013StockIncentivePlanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A2013 Stock Incentive Plan",
"label": "A2013 Stock Incentive Plan [Member]",
"terseLabel": "2013 Stock Incentive Plan"
}
}
},
"localname": "A2013StockIncentivePlanMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_A2021IncentiveAwardPlanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "2021 Incentive Award Plan",
"label": "2021 Incentive Award Plan [Member]",
"terseLabel": "2021 Incentive Award Plan"
}
}
},
"localname": "A2021IncentiveAwardPlanMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_APICBusinessCombinationContingentConsiderationLiability": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "APIC, Business Combination, Contingent Consideration, Liability",
"label": "APIC, Business Combination, Contingent Consideration, Liability",
"negatedLabelnegatedTerseLabel": "Recognition of earnEarn-out liabilitiesliability (Note 3)"
}
}
},
"localname": "APICBusinessCombinationContingentConsiderationLiability",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"pear_APICBusinessCombinationWarrantLiabilitiesAssumed": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "APIC, Business Combination, Warrant Liabilities Assumed",
"label": "APIC, Business Combination, Warrant Liabilities Assumed",
"negatedTerseLabel": "Recognition of warrantWarrant liabilities assumed in connection with the Business CombinationMerger (Note 3)"
}
}
},
"localname": "APICBusinessCombinationWarrantLiabilitiesAssumed",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "monetaryItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"pear_ATMOfferingMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "ATM Offering",
"label": "ATM Offering [Member]",
"terseLabel": "ATM Offering"
}
}
},
"localname": "ATMOfferingMember",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"pear_AccruedCompensationAndRelatedBenefitsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Accrued Compensation And Related Benefits",
"label": "Accrued Compensation And Related Benefits [Member]",
"terseLabel": "Accrued Compensation andAnd Related Benefits"
}
}
},
"localname": "AccruedCompensationAndRelatedBenefitsMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_AnchorInvestorMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Anchor investor member.",
"label": "Anchor Investor [Member]",
"terseLabel": "Anchor Investor [Member]"
}
}
},
"localname": "AnchorInvestorMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsBUSINESSCOMBINATIONDetails"
],
"xbrltype": "domainItemType"
},
"pear_AuditInformationAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Audit Information",
"label": "Audit Information [Abstract]"
}
}
},
"localname": "AuditInformationAbstract",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"xbrltype": "stringItemType"
},
"pear_BeHealthSolutionsLLCMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "BeHealth Solutions,\u00a0LLC",
"label": "BeHealth Solutions,\u00a0LLC [Member]",
"terseLabel": "BeHealth Solutions,\u00a0LLC"
}
}
},
"localname": "BeHealthSolutionsLLCMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_BostonMassachusettsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Boston, Massachusetts",
"label": "Boston, Massachusetts [Member]",
"terseLabel": "Boston, Massachusetts"
}
}
},
"localname": "BostonMassachusettsMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "domainItemType"
},
"pear_BusinessAcquisitionEquityInterestIssuedOrIssuableNumberOfTranches": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Business Acquisition, Equity Interest Issued or Issuable, Number of Tranches",
"label": "Business Acquisition, Equity Interest Issued or Issuable, Number of Tranches",
"terseLabel": "Number of tranches"
}
}
},
"localname": "BusinessAcquisitionEquityInterestIssuedOrIssuableNumberOfTranches",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "integerItemType"
},
"pear_BusinessCombinationConversionOfStockConversionRatio": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Business Combination, Conversion Of Stock, Conversion Ratio",
"label": "Business Combination, Conversion Of Stock, Conversion Ratio",
"terseLabel": "Conversion ratio"
}
}
},
"localname": "BusinessCombinationConversionOfStockConversionRatio",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsBUSINESSCOMBINATIONDetails"
],
"xbrltype": "sharesItemType"
},
"pear_BusinessCombinationThresholdConsecutiveTradingDays": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Business Combination, Threshold Consecutive Trading Days",
"label": "Business Combination, Threshold Consecutive Trading Days",
"terseLabel": "Consecutive trading days"
}
}
},
"localname": "BusinessCombinationThresholdConsecutiveTradingDays",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "integerItemType"
},
"pear_BusinessCombinationThresholdTradingDays": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Business Combination, Threshold Trading Days",
"label": "Business Combination, Threshold Trading Days",
"terseLabel": "Threshold trading days"
}
}
},
"localname": "BusinessCombinationThresholdTradingDays",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "integerItemType"
},
"pear_BusinessCombinationTrancheAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Business Combination, Tranche",
"label": "Business Combination, Tranche [Axis]",
"terseLabel": "Business Combination, Tranche [Axis]"
}
}
},
"localname": "BusinessCombinationTrancheAxis",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "stringItemType"
},
"pear_BusinessCombinationTrancheDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Business Combination, Tranche [Domain]",
"label": "Business Combination, Tranche [Domain]",
"terseLabel": "Business Combination, Tranche [Domain]"
}
}
},
"localname": "BusinessCombinationTrancheDomain",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "domainItemType"
},
"pear_COVID19Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "COVID-19",
"label": "COVID-19 [Member]",
"terseLabel": "COVID-19"
}
}
},
"localname": "COVID19Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_CapitalizedMilestonePaymentLiabilityUsefulLife": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Capitalized Milestone Payment Liability, Useful Life",
"label": "Capitalized Milestone Payment Liability, Useful Life",
"terseLabel": "Useful life of capitalized milestone payments"
}
}
},
"localname": "CapitalizedMilestonePaymentLiabilityUsefulLife",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails20221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"pear_CapitalizedResearchAndDevelopmentExpenseAmortization": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Capitalized Research and Development Expense, Amortization",
"label": "Capitalized Research and Development Expense, Amortization",
"terseLabel": "Capitalized research and development expense"
}
}
},
"localname": "CapitalizedResearchAndDevelopmentExpenseAmortization",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_CapitalizedResearchAndDevelopmentExpenseAmortizationPeriod": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Capitalized Research and Development Expense, Amortization Period",
"label": "Capitalized Research and Development Expense, Amortization Period",
"terseLabel": "Amortization period for capitalized research and development expense"
}
}
},
"localname": "CapitalizedResearchAndDevelopmentExpenseAmortizationPeriod",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "durationItemType"
},
"pear_CashAcquiredThroughReverseRecapitalization": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails": {
"order": 3.0,
"parentTag": "pear_ProceedsFromReverseRecapitalizationTransactionGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Cash Acquired Through Reverse Recapitalization",
"label": "Cash Acquired Through Reverse Recapitalization",
"terseLabel": "Cash - THMA trust and cash (net of redemptions)"
}
}
},
"localname": "CashAcquiredThroughReverseRecapitalization",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_CertainEligibleEmployeesMember": {
"auth_ref": []pear_ChangeInFairValueAdjustmentOfWarrants": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 9.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Certain Eligible EmployeesChange In Fair Value Adjustment Of Warrants",
"label": "Certain Eligible Employees [Member]Change In Fair Value Adjustment Of Warrants",
"terseLabelverboseLabel": "Certain Eligible EmployeesChange in fair value of warrants"
}
}
},
"localname": "CertainEligibleEmployeesMemberChangeInFairValueAdjustmentOfWarrants",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemType"
},
"pear_ChiefExecutiveOfficerAndCertainEligibleEmployeesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Chief Executive Officer And Certain Eligible Employees",
"label": "Chief Executive Officer And Certain Eligible Employees [Member]",
"terseLabel": "Chief Executive Officer And Certain Eligible Employees"
}
}
},
"localname": "ChiefExecutiveOfficerAndCertainEligibleEmployeesMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_ClassOfWarrantOrRightsIssuePricePerWarrant": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Class Of Warrant Or Rights Issue Price Per Warrant.",
"label": "Class Of Warrant Or Rights Issue Price Per Warrant",
"terseLabel": "Class of warrant or rights issue price per warrant"
}
}
},
"localname": "ClassOfWarrantOrRightsIssuePricePerWarrant",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "perShareItemType"
},
"pear_ClassOfWarrantsRedemptionNoticePeriod": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Class of warrants, redemption notice period.",
"label": "Class of warrants redemption notice period",
"terseLabel": "Class of warrants, redemption notice period"
}
}
},
"localname": "ClassOfWarrantsRedemptionNoticePeriod",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "durationItemType"
},
"pear_ClassOfWarrantsRedemptionPricePerUnit": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Class of warrants, redemption price per unit.",
"label": "Class of warrants redemption price per unit",
"terseLabel": "Class of warrants, redemption price per unit"
}
}
},
"localname": "ClassOfWarrantsRedemptionPricePerUnit",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "perShareItemType"
},
"pear_CollaborationAndLicenseRevenueMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Collaboration And License Revenue",
"label": "Collaboration And License Revenue [Member]",
"terseLabel": "Collaboration and license revenue"
}
}
},
"localname": "CollaborationAndLicenseRevenueMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "domainItemType"
},
"pear_ContingentConsiderationLiabilityMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Contingent Consideration, Liability",
"label": "Contingent Consideration, Liability [Member]",
"terseLabel": "Earn-out Liabilities"
}
}
},
"localname": "ContingentConsiderationLiabilityMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "domainItemType"
},
"pear_ContingentConsiderationLiabilityPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Contingent Consideration, Liability, Policy",
"label": "Contingent Consideration, Liability, Policy [Policy Text Block]",
"terseLabel": "Earn-outOut Liabilities"
}
}
},
"localname": "ContingentConsiderationLiabilityPolicyPolicyTextBlock",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"pear_ConvertiblePreferredStockPolicyPolicyTextBlockpear_ContractWithCustomerLiabilityCancellationNotice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Convertible Preferred Stock, PolicyContract with Customer, Liability, Cancellation Notice",
"label": "Convertible Preferred Stock, Policy [Policy Text Block]Contract with Customer, Liability, Cancellation Notice",
"terseLabel": "Legacy Pear Convertible Preferred StockCancellation notice"
}
}
},
"localname": "ConvertiblePreferredStockPolicyPolicyTextBlockContractWithCustomerLiabilityCancellationNotice",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemTypeREVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "durationItemType"
},
"pear_ContractWithCustomerLiabilityTransactionCosts": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Contract with Customer, Liability, Transaction Costs",
"label": "Contract with Customer, Liability, Transaction Costs",
"terseLabel": "Transaction costs"
}
}
},
"localname": "ContractWithCustomerLiabilityTransactionCosts",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_Customer1Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Customer 1",
"label": "Customer 1 [Member]",
"terseLabel": "Customer 1"
}
}
},
"localname": "Customer1Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_Customer2Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Customer 2",
"label": "Customer 2 [Member]",
"terseLabel": "Customer 2"
}
}
},
"localname": "Customer2Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_Customer3Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Customer 3",
"label": "Customer 3 [Member]",
"terseLabel": "Customer 3"
}
}
},
"localname": "Customer3Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_Customer4Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Customer 4",
"label": "Customer 4 [Member]",
"terseLabel": "Customer 4"
}
}
},
"localname": "Customer4Member",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_DataFoundryCloudSubscriptionMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Data Foundry Cloud Subscription",
"label": "Data Foundry Cloud Subscription [Member]",
"terseLabel": "Data Foundry Cloud Subscription"
}
}
},
"localname": "DataFoundryCloudSubscriptionMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_DebtCovenant1Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Covenant 1",
"label": "Debt Covenant 1 [Member]",
"terseLabel": "Debt Covenant 1"
}
}
},
"localname": "DebtCovenant1Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_DebtCovenant2Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Covenant 2",
"label": "Debt Covenant 2 [Member]",
"terseLabel": "Debt Covenant 2"
}
}
},
"localname": "DebtCovenant2Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_DebtInstrumentCovenantComplianceAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Covenant Compliance",
"label": "Debt Instrument, Covenant Compliance [Axis]",
"terseLabel": "Debt Instrument, Covenant Compliance [Axis]"
}
}
},
"localname": "DebtInstrumentCovenantComplianceAxis",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "stringItemType"
},
"pear_DebtInstrumentCovenantComplianceDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Covenant Compliance [Domain]",
"label": "Debt Instrument, Covenant Compliance [Domain]",
"terseLabel": "Debt Instrument, Covenant Compliance [Domain]"
}
}
},
"localname": "DebtInstrumentCovenantComplianceDomain",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_DebtInstrumentCovenantComplianceMinimumAggregateCashBalance": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Covenant Compliance, Minimum Aggregate Cash Balance",
"label": "Debt Instrument, Covenant Compliance, Minimum Aggregate Cash Balance",
"terseLabel": "Minimum aggregate cash balance"
}
}
},
"localname": "DebtInstrumentCovenantComplianceMinimumAggregateCashBalance",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_DebtInstrumentCovenantComplianceRevenuesForTheTrailing12MonthPeriod": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Covenant Compliance, Revenues For The Trailing 12-Month Period",
"label": "Debt Instrument, Covenant Compliance, Revenues For The Trailing 12-Month Period",
"terseLabel": "Revenues for trailing 12-month period"
}
}
},
"localname": "DebtInstrumentCovenantComplianceRevenuesForTheTrailing12MonthPeriod",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_DebtInstrumentExtinguishmentOfDebtPercentageOfBorrowingspear_DebtInstrumentDebtCovenantMinimumTotalRevenue": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Debt Covenant, Minimum Total Revenue",
"label": "Debt Instrument, Debt Covenant, Minimum Total Revenue",
"terseLabel": "Debt Instrument, Debt Covenant, Minimum Total Revenue"
}
}
},
"localname": "DebtInstrumentDebtCovenantMinimumTotalRevenue",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSMinimumRevenueCovenantDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_DebtInstrumentDebtCovenantMinimumTotalRevenueTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Extinguishment Of DebtDebt Covenant, Percentage Of BorrowingsMinimum Total Revenue",
"label": "Debt Instrument, Extinguishment Of DebtDebt Covenant, Percentage Of BorrowingsMinimum Total Revenue [Table Text Block]",
"terseLabel": "Percentage of borrowings extinguishedTotal Minimum Revenue Covenant"
}
}
},
"localname": "DebtInstrumentExtinguishmentOfDebtPercentageOfBorrowingsDebtInstrumentDebtCovenantMinimumTotalRevenueTableTextBlock",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetailsINDEBTEDNESSTables"
],
"xbrltype": "percentItemTypetextBlockItemType"
},
"pear_DebtInstrumentFloorInterestRate": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Floor Interest Rate",
"label": "Debt Instrument, Floor Interest Rate",
"terseLabel": "Floor rate"
}
}
},
"localname": "DebtInstrumentFloorInterestRate",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "percentItemType"
},
"pear_DebtInstrumentPeriodicPaymentPercentageOfPrincipal": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Periodic Payment, Percentage Of Principal",
"label": "Debt Instrument, Periodic Payment, Percentage Of Principal",
"terseLabel": "Percentage of principal, payment"
}
}
},
"localname": "DebtInstrumentPeriodicPaymentPercentageOfPrincipal",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "percentItemType"
},
"pear_DebtInstrumentPeriodicPaymentPercentageOfVotingStockTriggeringRepayment": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Periodic Payment, Percentage Of Voting Stock Triggering Repayment",
"label": "Debt Instrument, Periodic Payment, Percentage Of Voting Stock Triggering Repayment",
"terseLabel": "Percentage of voting stock triggering repayment"
}
}
},
"localname": "DebtInstrumentPeriodicPaymentPercentageOfVotingStockTriggeringRepayment",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "percentItemType"
},
"pear_DebtInstrumentPeriodicPaymentReturnOnPrepaymentAmount": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Periodic Payment, Return On Prepayment Amount",
"label": "Debt Instrument, Periodic Payment, Return On Prepayment Amount",
"terseLabel": "Return on prepaid amount"
}
}
},
"localname": "DebtInstrumentPeriodicPaymentReturnOnPrepaymentAmount",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "pureItemType"
},
"pear_DebtInstrumentTerminationFee": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Debt Instrument, Termination Fee",
"label": "Debt Instrument, Termination Fee",
"terseLabel": "Termination fee"
}
}
},
"localname": "DebtInstrumentTerminationFee",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_DeferredOfferingCostsIncurredDuringNoncashOrPartialNoncashTransaction": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred Offering Costs Incurred During Noncash Or Partial Noncash Transaction",
"label": "Deferred Offering Costs Incurred During Noncash Or Partial Noncash Transaction",
"terseLabel": "Deferred offering and equity issuance costs included in accounts payable and accrued expenses"
}
}
},
"localname": "DeferredOfferingCostsIncurredDuringNoncashOrPartialNoncashTransaction",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS20221231",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_DeferredTaxAssetsCapitalizedResearchAndDevelopmentCosts": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 2.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred Tax Assets, Capitalized Research and Development Costs",
"label": "Deferred Tax Assets, Capitalized Research and Development Costs",
"terseLabel": "Capitalized R&D expenses"
}
}
},
"localname": "DeferredTaxAssetsCapitalizedResearchAndDevelopmentCosts",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_DeferredTaxAssetsLeasingArrangements": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 6.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Deferred Tax Assets, Leasing Arrangements",
"label": "Deferred Tax Assets, Leasing Arrangements",
"terseLabel": "Lease liabilities"
}
}
},
"localname": "DeferredTaxAssetsLeasingArrangements",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_EarnOutSharesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Earn-Out Shares",
"label": "Earn-Out Shares [Member]",
"terseLabel": "Earn-Out Shares"
}
}
},
"localname": "EarnOutSharesMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetails"
],
"xbrltype": "domainItemTypeNETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"pear_EffectiveIncomeTaxRateReconciliationChangeInFairValueOfEarnOutLiabilityPercent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 5.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Effective Income Tax Rate Reconciliation, Change in Fair Value of Earn-Out Liability, Percent",
"label": "Effective Income Tax Rate Reconciliation, Change in Fair Value of Earn-Out Liability, Percent",
"terseLabel": "Change in fair value of earn-out liability"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationChangeInFairValueOfEarnOutLiabilityPercent",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"pear_EffectiveIncomeTaxRateReconciliationProvisionToReturnAdjustmentPercent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 2.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Effective Income Tax Rate Reconciliation, Provision To Return Adjustment, Percent",
"label": "Effective Income Tax Rate Reconciliation, Provision To Return Adjustment, Percent",
"terseLabel": "Provision to return adjustment"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationProvisionToReturnAdjustmentPercent",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"pear_GainLossOnIssuanceOfConvertiblePreferredStock": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 15.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
},
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"pear_ForwardPurchaseAgreementToAnchorInvestorMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Forward Purchase Agreement To Anchor Investor",
"label": "Forward Purchase Agreement To Anchor Investor [Member]",
"terseLabel": "Forward Purchase Agreement To Anchor Investor"
}
}
},
"localname": "ForwardPurchaseAgreementToAnchorInvestorMember",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "domainItemType"
},
"pear_GainLossOnConvertiblePreferredStock": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 14.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Gain (Loss) On Convertible Preferred Stock",
"label": "Gain (Loss) On Convertible Preferred Stock",
"negatedTerseLabel": "Loss on issuance of Legacy Pear convertible preferred stock"
}
}
},
"localname": "GainLossOnConvertiblePreferredStock",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_GainLossOnIssuanceOfConvertiblePreferredStock": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 4.0,
"parentTag": "us-gaap_OtherNonoperatingIncomeExpense",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Gain (Loss) On Issuance Of Convertible Preferred Stock",
"label": "Gain (Loss) On Issuance Of Convertible Preferred Stock",
"negatedTerseLabel": "Loss on issuance of legacy convertible preferred stock",
"terseLabel": "Loss on issuance of legacyLegacy Pear convertible preferred stock"
}
}
},
"localname": "GainLossOnIssuanceOfConvertiblePreferredStock",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS"
],
"xbrltype": "monetaryItemType"
},
"pear_GainLossOnRepurchaseOfConvertiblePreferredStock": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails": {
"order": 2.0,
"parentTag": "us-gaap_NetIncomeLossAvailableToCommonStockholdersDiluted",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Gain (Loss) On Repurchase Of Convertible Preferred Stock",
"label": "Gain (Loss) On Repurchase Of Convertible Preferred Stock",
"negatedTerseLabel": "Loss on repurchase of convertible preferred stock",
"terseLabel": "Loss on repurchase of convertible preferred stock"
}
}
},
"localname": "GainLossOnRepurchaseOfConvertiblePreferredStock",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails20221231",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"pear_InventionScienceFundILLCMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Invention Science Fund I, LLC",
"label": "Invention Science Fund I, LLC [Member]",
"terseLabel": "Invention Science Fund I, LLC"
}
}
},
"localname": "InventionScienceFundILLCMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_LegacyPearEquityholdersMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Legacy Pear Equityholders",
"label": "Legacy Pear Equityholders [Member]",
"terseLabel": "Legacy Pear Equityholders"
}
}
},
"localname": "LegacyPearEquityholdersMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "domainItemType"
},
"pear_LesseeOperatingLeaseRenewalTermPercentageIncrease": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Lessee, Operating Lease, Renewal Term, Percentage Increase",
"label": "Lessee, Operating Lease, Renewal Term, Percentage Increase",
"terseLabel": "Rate increase for lease renewal"
}
}
},
"localname": "LesseeOperatingLeaseRenewalTermPercentageIncrease",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "percentItemType"
},
"pear_LiabilitiesRecognizedContingentConsiderationLiability": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Liabilities Recognized, Contingent Consideration, Liability",
"label": "Liabilities Recognized, Contingent Consideration, Liability",
"terseLabel": "Recognition of earn-out liabilities"
}
}
},
"localname": "LiabilitiesRecognizedContingentConsiderationLiability",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_LongTermPurchaseCommitment": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Long Term Purchase Commitment",
"label": "Long Term Purchase Commitment",
"terseLabel": "Purchase liability"
}
}
},
"localname": "LongTermPurchaseCommitment",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_MilestonePaymentsPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Milestone Payments, Policy",
"label": "Milestone Payments, Policy [Policy Text Block]",
"terseLabel": "Milestone Payments"
}
}
},
"localname": "MilestonePaymentsPolicyPolicyTextBlock",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"pear_NetOperatingLossCarryforwardsResearchAndDevelopmentTaxCreditsAndAccruedExpensesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Net Operating Loss Carryforwards, Research And Development Tax Credits And Accrued Expenses",
"label": "Net Operating Loss Carryforwards, Research And Development Tax Credits And Accrued Expenses [Member]",
"terseLabel": "Net operating loss carryforwards, research and development tax credits and accrued expenses"
}
}
},
"localname": "NetOperatingLossCarryforwardsResearchAndDevelopmentTaxCreditsAndAccruedExpensesMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"pear_NoncashOrPartNoncashAcquisitionAssetsAssumedReceivables": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Noncash Or Part Noncash Acquisition, Assets Assumed, Receivables",
"label": "Noncash Or Part Noncash Acquisition, Assets Assumed, Receivables",
"terseLabel": "Plus: cash receivable from Trust"
}
}
},
"localname": "NoncashOrPartNoncashAcquisitionAssetsAssumedReceivables",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_NoncashOrPartNoncashAcquisitionLiabilitiesAssumedWarrants": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Noncash or Part Noncash Acquisition, Liabilities Assumed, Warrants",
"label": "Noncash or Part Noncash Acquisition, Liabilities Assumed, Warrants",
"negatedTerseLabel": "Less: warrant liabilities assumed",
"terseLabel": "Recognition of warrant liability"
}
}
},
"localname": "NoncashOrPartNoncashAcquisitionLiabilitiesAssumedWarrants",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_NovartisMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Novartis",
"label": "Novartis [Member]",
"terseLabel": "Novartis"
}
}
},
"localname": "NovartisMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "domainItemType"
},
"pear_NumberOfAdditionalWarrantsIssued": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number Of Additional Warrants Issued",
"label": "Number Of Additional Warrants Issued",
"terseLabel": "Additional warrants"
}
}
},
"localname": "NumberOfAdditionalWarrantsIssued",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "integerItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_NumberOfConsecutiveTradingDaysForDeterminingSharePrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of consecutive trading days for determining share price.",
"label": "Number of consecutive trading days for determining share price",
"terseLabel": "Number of consecutive trading days for determining share price"
}
}
},
"localname": "NumberOfConsecutiveTradingDaysForDeterminingSharePrice",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "durationItemType"
},
"pear_NumberOfLicenseAgreements": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number Of License Agreements",
"label": "Number Of License Agreements",
"terseLabel": "Number of license agreements"
}
}
},
"localname": "NumberOfLicenseAgreements",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"pear_NumberOfProducts": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number Of Products",
"label": "Number Of Products",
"terseLabel": "Number of products"
}
}
},
"localname": "NumberOfProducts",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"pear_NumberOfTradingDaysForDeterminingSharePrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of trading days for determining share price.",
"label": "Number of trading days for determining share price",
"terseLabel": "Number of trading days for determining share price"
}
}
},
"localname": "NumberOfTradingDaysForDeterminingSharePrice",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "durationItemType"
},
"pear_OperatingLeasesFutureMinimumPaymentsDueInFiveYearsAndThereafter": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails": {
"order": 2.0,
"parentTag": "us-gaap_OperatingLeasesFutureMinimumPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Operating Leases, Future Minimum Payments, Due In Five Years And Thereafter",
"label": "Operating Leases, Future Minimum Payments, Due In Five Years And Thereafter",
"terseLabel": "2026 and thereafter"
}
}
},
"localname": "OperatingLeasesFutureMinimumPaymentsDueInFiveYearsAndThereafter",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_OperatingLossCarryforwardsNotSubjectToExpiration": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Operating Loss Carryforwards, Not Subject to Expiration",
"label": "Operating Loss Carryforwards, Not Subject to Expiration",
"terseLabel": "NOLs not subject to expiration"
}
}
},
"localname": "OperatingLossCarryforwardsNotSubjectToExpiration",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_OtherShareholdersMemberpear_OtherCapitalExpendituresIncurredButNotYetPaid": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Other ShareholdersCapital Expenditures Incurred but Not yet Paid",
"label": "Other Shareholders [Member]Capital Expenditures Incurred but Not yet Paid",
"terseLabel": "Other ShareholdersPurchases of other assets in accounts payable and accrued expenses"
}
}
},
"localname": "OtherShareholdersMemberOtherCapitalExpendituresIncurredButNotYetPaid",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "domainItemTypemonetaryItemType"
},
"pear_PIPEInvestorsAndForwardPurchaseAgreementMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "PIPE Investors And Forward Purchase Agreement",
"label": "PIPE Investors And Forward Purchase Agreement [Member]",
"terseLabel": "PIPE Investors And Forward Purchase Agreement"
}
}
},
"localname": "PIPEInvestorsAndForwardPurchaseAgreementMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "domainItemType"
},
"pear_PatentCostsPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Patent Costs, Policy",
"label": "Patent Costs, Policy [Policy Text Block]",
"terseLabel": "Patent Costs"
}
}
},
"localname": "PatentCostsPolicyPolicyTextBlock",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"pear_PaymentsForMilestonePayment": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 1.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Payments For Milestone Payment",
"label": "Payments For Milestone Payment",
"negatedTerseLabel": "Milestone based license fee payment",
"terseLabel": "Milestone basedPayment of milestone license fee paymentin intangibles and accrued expenses"
}
}
},
"localname": "PaymentsForMilestonePayment",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_PaymentsOfReverseRecapitalizationTransactionCosts": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails": {
"order": 2.0,
"parentTag": "pear_ProceedsFromReverseRecapitalizationTransaction",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Payments of Reverse Recapitalization Transaction Costs",
"label": "Payments of Reverse Recapitalization Transaction Costs",
"negatedTerseLabel": "Less: transaction costs and advisory fees paid"
}
}
},
"localname": "PaymentsOfReverseRecapitalizationTransactionCosts",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_PearTherapeuticsIncMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Pear Therapeutics, Inc",
"label": "Pear Therapeutics, Inc [Member]",
"terseLabel": "Pear Therapeutics, Inc"
}
}
},
"localname": "PearTherapeuticsIncMember",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "domainItemType"
},
"pear_PerceptiveCreditFacilityMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Perceptive Credit Facility",
"label": "Perceptive Credit Facility [Member]",
"terseLabel": "Perceptive Credit Facility"
}
}
},
"localname": "PerceptiveCreditFacilityMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "domainItemType"
},
"pear_PerceptiveCreditFacilityTranche1Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Perceptive Credit Facility, Tranche 1",
"label": "Perceptive Credit Facility, Tranche 1 [Member]",
"terseLabel": "Debt, Tranche 1"
}
}
},
"localname": "PerceptiveCreditFacilityTranche1Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_PerceptiveCreditFacilityTranche2Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Perceptive Credit Facility, Tranche 2",
"label": "Perceptive Credit Facility, Tranche 2 [Member]",
"terseLabel": "Debt, Tranche 2"
}
}
},
"localname": "PerceptiveCreditFacilityTranche2Member",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_PerceptiveCreditFacilityTranche3Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Perceptive Credit Facility, Tranche 3",
"label": "Perceptive Credit Facility, Tranche 3 [Member]",
"terseLabel": "Debt, Tranche 3"
}
}
},
"localname": "PerceptiveCreditFacilityTranche3Member",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_PreferredStockConversionTriggeringNetProceedsMinimum": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Preferred Stock, Conversion Triggering, Net Proceeds Minimum",
"label": "Preferred Stock, Conversion Triggering, Net Proceeds Minimum",
"terseLabel": "Triggering event, minimum net proceeds"
}
}
},
"localname": "PreferredStockConversionTriggeringNetProceedsMinimum",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_PreferredStockConversionTriggeringPricePricePerShare": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Preferred Stock, Conversion Triggering Price, Price Per Share",
"label": "Preferred Stock, Conversion Triggering Price, Price Per Share",
"terseLabel": "Triggering price (in dollars per share)"
}
}
},
"localname": "PreferredStockConversionTriggeringPricePricePerShare",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails"
],
"xbrltype": "perShareItemType20221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_PrivatePlacementWarrantsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Private placement warrants .",
"label": "Private Placement Warrants [Member]",
"terseLabel": "Private placement warrants to purchase common stock"
}
}
},
"localname": "PrivatePlacementWarrantsMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_ProceedsFromReverseRecapitalizationTransaction": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 23.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Proceeds From Reverse Recapitalization Transaction",
"label": "Proceeds From Reverse Recapitalization Transaction",
"terseLabel": "Gross proceedsProceeds from Business Combination, net of transactions costs paid",
"totalLabel": "Net proceeds from the Business Combination",
"verboseLabel": "Proceeds from Business Combination, net of transactions costs paid"
}
}
},
"localname": "ProceedsFromReverseRecapitalizationTransaction",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"pear_ProceedsFromReverseRecapitalizationTransactionGross": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails": {
"order": 1.0,
"parentTag": "pear_ProceedsFromReverseRecapitalizationTransaction",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Proceeds From Reverse Recapitalization Transaction, Gross",
"label": "Proceeds From Reverse Recapitalization Transaction, Gross",
"terseLabel": "Gross proceeds",
"totalLabel": "Gross proceeds (excluding cash due at December 31, 2021 from the Trust Account)"
}
}
},
"localname": "ProceedsFromReverseRecapitalizationTransactionGross",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_PublicAndPrivatePlacementWarrantsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Public And Private Placement Warrants",
"label": "Public And Private Placement Warrants [Member]",
"terseLabel": "Public And Private Placement Warrants"
}
}
},
"localname": "PublicAndPrivatePlacementWarrantsMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_PublicWarrantMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Public Warrant.",
"label": "Public Warrant [Member]",
"terseLabel": "Public Warrant [Member]"
}
}
},
"localname": "PublicWarrantMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"pear_PublicWarrantsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Public Warrants [Member]",
"label": "Public Warrants [Member]",
"terseLabel": "Public warrants to purchase common stock"
}
}
},
"localname": "PublicWarrantsMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetails"
],
"xbrltype": "domainItemTypeSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_PurchaseObligationCurrent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails": {
"order": 5.0,
"parentTag": "us-gaap_AccountsPayableAndOtherAccruedLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Purchase Obligation, Current",
"label": "Purchase Obligation, Current",
"terseLabel": "Amount due under non-cancelable purchase obligation"
}
}
},
"localname": "PurchaseObligationCurrent",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_RaleighNorthCarolinaMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Raleigh, North Carolina",
"label": "Raleigh, North Carolina [Member]",
"terseLabel": "Raleigh, North Carolina"
}
}
},
"localname": "RaleighNorthCarolinaMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "domainItemType"
},
"pear_RecapitalizationExchangeRatio": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Recapitalization Exchange Ratio",
"label": "Recapitalization Exchange Ratio",
"terseLabel": "Exchange ratio"
}
}
},
"localname": "RecapitalizationExchangeRatio",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "pureItemType"
},
"pear_Red5GroupLLCMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Red 5 Group, LLC",
"label": "Red 5 Group, LLC [Member]",
"terseLabel": "Red 5 Group, LLC"
}
}
},
"localname": "Red5GroupLLCMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"pear_ReimbursementMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Reimbursement",
"label": "Reimbursement [Member]",
"terseLabel": "Reimbursement"
}
}
},
"localname": "ReimbursementMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "domainItemType"
},
"pear_ResearchAndDevelopmentArrangementContractToPerformForOthersMaximumPaymentMilestoneAchievement": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Research and Development Arrangement, Contract to Perform for Others, Maximum Payment, Milestone Achievement",
"label": "Research and Development Arrangement, Contract to Perform for Others, Maximum Payment, Milestone Achievement",
"terseLabel": "Maximum payment, milestone achievement"
}
}
},
"localname": "ResearchAndDevelopmentArrangementContractToPerformForOthersMaximumPaymentMilestoneAchievement",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_ResearchAndDevelopmentLiabilitiesCurrent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails": {
"order": 36.0,
"parentTag": "us-gaap_AccountsPayableAndOtherAccruedLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Research And Development Liabilities, Current",
"label": "Research And Development Liabilities, Current",
"terseLabel": "Research and development costs"
}
}
},
"localname": "ResearchAndDevelopmentLiabilitiesCurrent",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_ReverseRecapitalizationAccruedTransactionCosts": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Reverse Recapitalization, Accrued Transaction Costs",
"label": "Reverse Recapitalization, Accrued Transaction Costs",
"negatedTerseLabel": "Less: accrued transaction costs at December 31, 2021",
"terseLabel": "Transaction costs"
}
}
},
"localname": "ReverseRecapitalizationAccruedTransactionCosts",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_ReverseRecapitalizationNet": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Reverse Recapitalization, Net",
"label": "Reverse Recapitalization, Net",
"terseLabel": "Reverse merger, net of transaction costs"
}
}
},
"localname": "ReverseRecapitalizationNet",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_RevisionOfPriorPeriodAdjustedBalanceMemberpear_SaleOfStockGrossProceedsAuthorizedAmount": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Sale of Stock, Gross Proceeds Authorized Amount",
"label": "Sale of Stock, Gross Proceeds Authorized Amount",
"terseLabel": "Authorized gross proceeds"
}
}
},
"localname": "SaleOfStockGrossProceedsAuthorizedAmount",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_SaleOfStockGrossProceedsAuthorizedAmountCommissionFeePercentage": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Revision Of Prior Period, Adjusted BalanceSale of Stock, Gross Proceeds Authorized Amount, Commission Fee Percentage",
"label": "Revision Of Prior Period, Adjusted Balance [Member]Sale of Stock, Gross Proceeds Authorized Amount, Commission Fee Percentage",
"terseLabel": "Revision Of Prior Period, Adjusted BalanceCommission fee percentage"
}
}
},
"localname": "RevisionOfPriorPeriodAdjustedBalanceMemberSaleOfStockGrossProceedsAuthorizedAmountCommissionFeePercentage",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "domainItemType"
},
"pear_SVBTermLoanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "SVB Term Loan",
"label": "SVB Term Loan [Member]",
"terseLabel": "SVB Term Loan"
}
}
},
"localname": "SVBTermLoanMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "domainItemTypeCAPITALSTOCKDetails"
],
"xbrltype": "percentItemType"
},
"pear_SanFranciscoCaliforniaMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "San Francisco, California",
"label": "San Francisco, California [Member]",
"terseLabel": "San Francisco, California"
}
}
},
"localname": "SanFranciscoCaliforniaMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "domainItemType"
},
"pear_ScheduleOfReverseCapitalizationTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule Of Reverse Capitalization",
"label": "Schedule Of Reverse Capitalization [Table Text Block]",
"terseLabel": "Schedule Of Reverse Capitalization"
}
}
},
"localname": "ScheduleOfReverseCapitalizationTableTextBlock",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONTables"
],
"xbrltype": "textBlockItemType"
},
"pear_ScheduleOfReverseRecapitalizationLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule Of Reverse Recapitalization [Line Items]",
"label": "Schedule Of Reverse Recapitalization [Line Items]",
"terseLabel": "Schedule Of Reverse Recapitalization [Line Items]"
}
}
},
"localname": "ScheduleOfReverseRecapitalizationLineItems",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "stringItemType"
},
"pear_ScheduleOfReverseRecapitalizationTable": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule Of Reverse Recapitalization",
"label": "Schedule Of Reverse Recapitalization [Table]",
"terseLabel": "Schedule Of Reverse Recapitalization [Table]"
}
}
},
"localname": "ScheduleOfReverseRecapitalizationTable",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "stringItemType"
},
"pear_SeriesAAndBPreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series A and B Preferred Stock",
"label": "Series A and B Preferred Stock [Member]",
"terseLabel": "Series A and B Preferred Stock"
}
}
},
"localname": "SeriesAAndBPreferredStockMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails"
],
"xbrltype": "domainItemType"
},
"pear_SeriesAWarrantsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series A Warrants",
"label": "Series A Warrants [Member]",
"terseLabel": "Series A Warrants"
}
}
},
"localname": "SeriesAWarrantsMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"pear_SeriesCPreferredStockWarrant1Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series C Preferred Stock, Warrant 1",
"label": "Series C Preferred Stock, Warrant 1 [Member]",
"terseLabel": "Series C Preferred Stock, Warrant 1"
}
}
},
"localname": "SeriesCPreferredStockWarrant1Member",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"pear_SeriesCPreferredStockWarrant2Member": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series C Preferred Stock, Warrant 2",
"label": "Series C Preferred Stock, Warrant 2 [Member]",
"terseLabel": "Series C Preferred Stock, Warrant 2"
}
}
},
"localname": "SeriesCPreferredStockWarrant2Member",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType20221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "stringItemType"
},
"pear_SeriesD1PreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series D-1 Preferred Stock",
"label": "Series D-1 Preferred Stock [Member]",
"terseLabel": "Series D-1 Preferred Stock"
}
}
},
"localname": "SeriesD1PreferredStockMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "domainItemType"
},
"pear_SeriesD2PreferredStockMemberpear_ShareBasedCompensationArrangementByShareBasedPaymentAwardGrantDateFairValue": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series D-2 Preferred StockShare-Based Compensation Arrangement by Share-Based Payment Award, Grant Date Fair Value",
"label": "Series D-2 Preferred Stock [Member]Share-Based Compensation Arrangement by Share-Based Payment Award, Grant Date Fair Value",
"terseLabel": "Series D-2 Preferred StockFair value at grant date (in dollars per share)"
}
}
},
"localname": "SeriesD2PreferredStockMemberShareBasedCompensationArrangementByShareBasedPaymentAwardGrantDateFairValue",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails"
],
"xbrltype": "domainItemType"
},
"pear_SeriesDWarrantsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Series D Warrants",
"label": "Series D Warrants [Member]",
"terseLabel": "Series D Warrants"
}
}
},
"localname": "SeriesDWarrantsMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemTypeSTOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "perShareItemType"
},
"pear_ShareBasedCompensationArrangementByShareBasedPaymentAwardMaximumEmployeeSubscriptionAmount": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Share-based Compensation Arrangement by Share-based Payment Award, Maximum Employee Subscription Amount",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Maximum Employee Subscription Amount",
"terseLabel": "Maximum purchase amount"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardMaximumEmployeeSubscriptionAmount",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfAdditionalSharesAllowableUnderThePlan": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share-based Compensation Arrangement by Share-based Payment Award, Number of Additional Shares Allowable Under The Plan",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Number of Additional Shares Allowable Under The Plan",
"terseLabel": "Number of additional shares allowed (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfAdditionalSharesAllowableUnderThePlan",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "sharesItemType"
},
"pear_ShareBasedCompensationArrangementByShareBasedPaymentAwardPurchasePeriod": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share-Based Compensation Arrangement By Share-based Payment Award, Purchase Period",
"label": "Share-Based Compensation Arrangement By Share-based Payment Award, Purchase Period",
"terseLabel": "Purchase period"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardPurchasePeriod",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "durationItemType"
},
"pear_SharePriceLessThanOrEqualsToUsdEighteenMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Price Less Than Or Equals To USD Eighteen.",
"label": "Share Price Less Than Or Equals To USD Eighteen [Member]",
"terseLabel": "Share price lessPrice Less thanThan orOr equals to 18Equals To USD [Member]Eighteen"
}
}
},
"localname": "SharePriceLessThanOrEqualsToUsdEighteenMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"pear_SharePriceMoreThanOrEqualsToUsdEighteenMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Price More Than Or Equals To USD Eighteen.",
"label": "Share Price More Than Or Equals To USD Eighteen [Member]",
"terseLabel": "Share price morePrice More thanThan orOr equals to 18Equals To USD [Member]Eighteen"
}
}
},
"localname": "SharePriceMoreThanOrEqualsToUsdEighteenMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"pear_SharePriceRangeAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Price Range [Axis]",
"label": "Share Price Range [Axis]",
"terseLabel": "Share Price Range [Axis]"
}
}
},
"localname": "SharePriceRangeAxis",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "stringItemType"
},
"pear_SharePriceRangeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share Price Range [Domain]",
"label": "Share Price Range [Domain]",
"terseLabel": "Share Price Range [Domain]"
}
}
},
"localname": "SharePriceRangeDomain",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"pear_SiliconValleyBankMemberpear_SoftBankCorpMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Silicon Valley BankSoftBank Corp.",
"label": "Silicon Valley BankSoftBank Corp. [Member]",
"terseLabel": "Silicon Valley BankSoftBank Corp."
}
}
},
"localname": "SiliconValleyBankMemberSoftBankCorpMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetailsREVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "domainItemType"
},
"pear_StockAndWarrantsIssuedDuringPeriodSharesPreferredStockAndWarrants": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Stock And Warrants Issued During Period, Shares, Preferred Stock And Warrants",
"label": "Stock And Warrants Issued During Period, Shares, Preferred Stock And Warrants",
"terseLabel": "Warrants exercised (in shares)"
}
}
},
"localname": "StockAndWarrantsIssuedDuringPeriodSharesPreferredStockAndWarrants",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "sharesItemType"
},
"pear_StockIssuedDuringPeriodSharesCommonStockWarrantsExercised": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Stock Issued During Period, Shares, Common Stock Warrants Exercised",
"label": "Stock Issued During Period, Shares, Common Stock Warrants Exercised",
"terseLabel": "Exercise of common stock warrants (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesCommonStockWarrantsExercised",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "sharesItemType"
},
"pear_StockIssuedDuringPeriodSharesReverseCapitalizationNetOfCosts": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Stock Issued During Period, Shares, Reverse Capitalization, Net Of Costs",
"label": "Stock Issued During Period, Shares, Reverse Capitalization, Net Of Costs",
"verboseLabelterseLabel": "Reverse recapitalization, net of transaction costs (Note 1) (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesReverseCapitalizationNetOfCosts",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "sharesItemType"
},
"pear_StockIssuedDuringPeriodValuePreferredStockWarrantsExercised": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Stock Issued During Period, Value, Preferred Stock Warrants Exercised",
"label": "Stock Issued During Period, Value, Preferred Stock Warrants Exercised",
"terseLabel": "Exercise of Legacy Pear warrants"
}
}
},
"localname": "StockIssuedDuringPeriodValuePreferredStockWarrantsExercised",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"pear_StockIssuedDuringPeriodValueReverseCapitalizationNetOfCosts": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Stock Issued During Period, Value, Reverse Capitalization, Net Of Costs",
"label": "Stock Issued During Period, Value, Reverse Capitalization, Net Of Costs",
"terseLabel": "Reverse recapitalization, net of transaction costs (Note 1)"
}
}
},
"localname": "StockIssuedDuringPeriodValueReverseCapitalizationNetOfCosts",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"pear_StockRepurchasedDuringPeriodPricePerSharepear_SubleaseMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Stock Repurchased During Period, Price Per ShareSublease",
"label": "Stock Repurchased During Period, Price Per ShareSublease [Member]",
"terseLabel": "Shares repurchased (in dollars per share)Sublease"
}
}
},
"localname": "StockRepurchasedDuringPeriodPricePerShareSubleaseMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails20221231",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "domainItemType"
},
"pear_SubscriptionSupportAndProfessionalServicesRevenueMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Subscription, Support, and Professional Services Revenue",
"label": "Subscription, Support, and Professional Services Revenue [Member]",
"terseLabel": "Subscription, support, and professional services revenue"
}
}
},
"localname": "SubscriptionSupportAndProfessionalServicesRevenueMember",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "perShareItemTypedomainItemType"
},
"pear_THMAInitialStockholdersMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "THMA Initial Stockholders",
"label": "THMA Initial Stockholders [Member]",
"terseLabel": "THMA Initial Stockholders"
}
}
},
"localname": "THMAInitialStockholdersMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "domainItemType"
},
"pear_THMAPublicMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "THMA Public",
"label": "THMA Public [Member]",
"terseLabel": "THMA Public"
}
}
},
"localname": "THMAPublicMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "domainItemType"
},
"pear_THMASponsorMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "THMA Sponsor",
"label": "THMA Sponsor [Member]",
"terseLabel": "THMA Sponsor"
}
}
},
"localname": "THMASponsorMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "domainItemType"
},
"pear_TenderOfferAggregateCost": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Tender Offer, Aggregate Cost",
"label": "Tender Offer, Aggregate Cost",
"terseLabel": "Aggregate cost of tender offer"
}
}
},
"localname": "TenderOfferAggregateCost",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_TenderOfferFeesAndExpenses": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Tender Offer, Fees And Expenses",
"label": "Tender Offer, Fees And Expenses",
"terseLabel": "Tender offer fees and expenses"
}
}
},
"localname": "TenderOfferFeesAndExpenses",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_TenderOfferPricePerShare": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tender Offer, Price Per Share",
"label": "Tender Offer, Price Per Share",
"terseLabel": "Tender offer price per share (in dollars per share)"
}
}
},
"localname": "TenderOfferPricePerShare",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails"
],
"xbrltype": "perShareItemType"
},
"pear_TenderOfferSharesAvailableToPurchase": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tender Offer, Shares Available To Purchase",
"label": "Tender Offer, Shares Available To Purchase",
"terseLabel": "Shares to purchase under tender offer (in shares)"
}
}
},
"localname": "TenderOfferSharesAvailableToPurchase",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails"
],
"xbrltype": "sharesItemType"
},
"pear_TermLoanMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Term Loan",
"label": "Term Loan [Member]",
"terseLabel": "Term Loan"
}
}
},
"localname": "TermLoanMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails20221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "domainItemType"
},
"pear_ThimblePointAcquisitionCorpMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Thimble Point Acquisition Corp.",
"label": "Thimble Point Acquisition Corp. [Member]",
"terseLabel": "Thimble Point Acquisition Corp."
}
}
},
"localname": "ThimblePointAcquisitionCorpMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"pear_TrancheOneMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tranche One",
"label": "Tranche One [Member]",
"terseLabel": "Tranche One"
}
}
},
"localname": "TrancheOneMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "domainItemType"
},
"pear_TrancheThreeMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tranche Three",
"label": "Tranche Three [Member]",
"terseLabel": "Tranche Three"
}
}
},
"localname": "TrancheThreeMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "domainItemType"
},
"pear_TrancheTwoMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tranche Two",
"label": "Tranche Two [Member]",
"terseLabel": "Tranche Two"
}
}
},
"localname": "TrancheTwoMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "domainItemType"
},
"pear_UnusualOrInfrequentItemsOrBothDeferredPaymentLiability": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Unusual Or Infrequent Items, Or Both, Deferred Payment, Liability",
"label": "Unusual Or Infrequent Items, Or Both, Deferred Payment, Liability",
"terseLabel": "Deferred payment liability"
}
}
},
"localname": "UnusualOrInfrequentItemsOrBothDeferredPaymentLiability",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_UpfrontPayment": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Upfront Payment",
"label": "Upfront Payment",
"terseLabel": "Upfront payment"
}
}
},
"localname": "UpfrontPayment",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_UpfrontPaymentAmortizationPeriod": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Upfront Payment, Amortization Period",
"label": "Upfront Payment, Amortization Period",
"terseLabel": "Amortization period"
}
}
},
"localname": "UpfrontPaymentAmortizationPeriod",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"pear_UpfrontPaymentAnnualPayment": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Upfront Payment, Annual Payment",
"label": "Upfront Payment, Annual Payment",
"terseLabel": "Upfront annual payment"
}
}
},
"localname": "UpfrontPaymentAnnualPayment",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_UpfrontPaymentLiability": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Upfront Payment, Liability",
"label": "Upfront Payment, Liability",
"terseLabel": "Upfront payment liability"
}
}
},
"localname": "UpfrontPaymentLiability",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"pear_UpfrontPaymentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Upfront Payment",
"label": "Upfront Payment [Member]",
"terseLabel": "Upfront Payment"
}
}
},
"localname": "UpfrontPaymentMember",
"nsuri": "http://www.peartherapeutics.com/20211231",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "domainItemType"
},
"pear_WarrantLiabilityNoncurrent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 56.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Warrant Liability, Noncurrent",
"label": "Warrant Liability, Noncurrent",
"terseLabel": "Warrant liabilities"
}
}
},
"localname": "WarrantLiabilityNoncurrent",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION"
],
"xbrltype": "monetaryItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"pear_WarrantsAndRightsOutstandingExpirationPeriod": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Warrants And Rights Outstanding, Expiration Period",
"label": "Warrants And Rights Outstanding, Expiration Period",
"terseLabel": "Expiration period"
}
}
},
"localname": "WarrantsAndRightsOutstandingExpirationPeriod",
"nsuri": "http://www.peartherapeutics.com/20221231",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "durationItemType"
},
"pear_WarrantsPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Warrants, Policy",
"label": "Warrants, Policy [Policy Text Block]",
"terseLabel": "WarrantWarrants Liabilities"
}
}
},
"localname": "WarrantsPolicyPolicyTextBlock",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"pear_WaypointHealthInnovationsLLCMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Waypoint Health Innovations, LLC",
"label": "Waypoint Health Innovations, LLC [Member]",
"terseLabel": "Waypoint Health Innovations, LLC"
}
}
},
"localname": "WaypointHealthInnovationsLLCMember",
"nsuri": "http://www.peartherapeutics.com/2021123120221231",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_ChiefExecutiveOfficerMember": {
"auth_ref": [
"r207"
],
"lang": {
"en-us": {
"role": {
"label": "Chief Executive Officer [Member]",
"terseLabel": "Chief Executive Officer"
}
}
},
"localname": "ChiefExecutiveOfficerMember",
"nsuri": "http://fasb.org/srt/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemType"
},
"srt_CounterpartyNameAxis": {
"auth_ref": [
"r58srt_CounterpartyNameAxis": {
"auth_ref": [
"r229",
"r60r230",
"r120r349",
"r121r379",
"r277r651",
"r320r653"
],
"lang": {
"en-us": {
"role": {
"label": "Counterparty Name [Axis]",
"terseLabel": "Counterparty Name [Axis]"
}
}
},
"localname": "CounterpartyNameAxis",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetailsBUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "stringItemType"
},
"srt_MajorCustomersAxis": {
"auth_ref": [
"r206REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"srt_CumulativeEffectPeriodOfAdoptionAdjustmentMember": {
"auth_ref": [
"r186",
"r236",
"r243",
"r249",
"r306",
"r467",
"r468",
"r469",
"r488",
"r489",
"r515",
"r518",
"r520",
"r521",
"r566"
],
"lang": {
"en-us": {
"role": {
"label": "Cumulative Effect, Period of Adoption, Adjustment [Member]",
"terseLabel": "Cumulative Effect, Period of Adoption, Adjustment"
}
}
},
"localname": "CumulativeEffectPeriodOfAdoptionAdjustmentMember",
"nsuri": "http://fasb.org/srt/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_CumulativeEffectPeriodOfAdoptionAxis": {
"auth_ref": [
"r186",
"r236",
"r243",
"r249",
"r306",
"r467",
"r468",
"r469",
"r488",
"r489",
"r515",
"r518",
"r520",
"r521",
"r566"
],
"lang": {
"en-us": {
"role": {
"label": "Cumulative Effect, Period of Adoption [Axis]",
"terseLabel": "Cumulative Effect, Period of Adoption [Axis]"
}
}
},
"localname": "CumulativeEffectPeriodOfAdoptionAxis",
"nsuri": "http://fasb.org/srt/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"srt_CumulativeEffectPeriodOfAdoptionDomain": {
"auth_ref": [
"r186",
"r236",
"r243",
"r249",
"r306",
"r467",
"r468",
"r469",
"r488",
"r489",
"r515",
"r518",
"r520",
"r521",
"r566"
],
"lang": {
"en-us": {
"role": {
"label": "Cumulative Effect, Period of Adoption [Domain]",
"terseLabel": "Cumulative Effect, Period of Adoption [Domain]"
}
}
},
"localname": "CumulativeEffectPeriodOfAdoptionDomain",
"nsuri": "http://fasb.org/srt/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_MajorCustomersAxis": {
"auth_ref": [
"r294",
"r364r669",
"r370r739",
"r661r795"
],
"lang": {
"en-us": {
"role": {
"label": "Customer [Axis]",
"terseLabel": "Customer [Axis]"
}
}
},
"localname": "MajorCustomersAxis",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"srt_MaximumMember": {
"auth_ref": [
"r276r333",
"r319r334",
"r384r335",
"r387r336",
"r579r427",
"r580r602",
"r581r617",
"r582r647",
"r583r648",
"r584r666",
"r603r675",
"r658r682",
"r662r735",
"r692r786",
"r693r787",
"r788",
"r789",
"r790",
"r791"
],
"lang": {
"en-us": {
"role": {
"label": "Maximum [Member]",
"terseLabel": "Maximum"
}
}
},
"localname": "MaximumMember",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "domainItemType"
},
"srt_MinimumMember": {
"auth_ref": [
"r276r333",
"r319r334",
"r384r335",
"r387r336",
"r579r427",
"r580r602",
"r581r617",
"r582r647",
"r583r648",
"r584r666",
"r603r675",
"r658r682",
"r662r735",
"r692r786",
"r693r787",
"r788",
"r789",
"r790",
"r791"
],
"lang": {
"en-us": {
"role": {
"label": "Minimum [Member]",
"terseLabel": "Minimum"
}
}
},
"localname": "MinimumMember",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "domainItemType"
},
"srt_NameOfMajorCustomerDomain": {
"auth_ref": [
"r206r294",
"r364r669",
"r370r739",
"r661r795"
],
"lang": {
"en-us": {
"role": {
"label": "Customer [Domain]",
"terseLabel": "Customer [Domain]"
}
}
},
"localname": "NameOfMajorCustomerDomain",
"nsuri": "http://fasb.org/srt/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetails2022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"srt_OwnershipAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Ownership [Axis]",
"terseLabel": "Ownership [Axis]"
}
}
},
"localname": "OwnershipAxis",
"nsuri": "http://fasb.org/srt/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"srt_OwnershipDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Ownership [Domain]",
"terseLabel": "Ownership [Domain]"
}
}
},
"localname": "OwnershipDomain",
"nsuri": "http://fasb.org/srt/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "domainItemType"
},
"srt_ProductOrServiceAxis": {
"auth_ref": [
"r203r291",
"r364r605",
"r368r667",
"r606r680",
"r657r730",
"r659r731",
"r739",
"r794"
],
"lang": {
"en-us": {
"role": {
"label": "Product and Service [Axis]",
"terseLabel": "Product and Service [Axis]"
}
}
},
"localname": "ProductOrServiceAxis",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSREVENUEANDCONTRACTBALANCESDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "stringItemType"
},
"srt_ProductsAndServicesDomain": {
"auth_ref": [
"r203r291",
"r364r605",
"r368r667",
"r606r680",
"r657r730",
"r659r731",
"r739",
"r794"
],
"lang": {
"en-us": {
"role": {
"label": "Product and Service [Domain]",
"terseLabel": "Product and Service [Domain]"
}
}
},
"localname": "ProductsAndServicesDomain",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSREVENUEANDCONTRACTBALANCESDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "domainItemType"
},
"srt_RangeAxis": {
"auth_ref": [
"r276r333",
"r319r334",
"r373r335",
"r384r336",
"r387r420",
"r579r427",
"r580r458",
"r581r459",
"r582r460",
"r583r578",
"r584r602",
"r603r617",
"r658r647",
"r662r648",
"r692",
"r693r666",
"r675",
"r682",
"r728",
"r735",
"r787",
"r788",
"r789",
"r790",
"r791"
],
"lang": {
"en-us": {
"role": {
"label": "Statistical Measurement [Axis]",
"terseLabel": "Statistical Measurement [Axis]"
}
}
},
"localname": "RangeAxis",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "stringItemType"
},
"srt_RangeMember": {
"auth_ref": [
"r276r333",
"r319r334",
"r373r335",
"r384r336",
"r387r420",
"r579r427",
"r580r458",
"r581r459",
"r582r460",
"r583r578",
"r584r602",
"r603r617",
"r658r647",
"r662r648",
"r692",
"r693r666",
"r675",
"r682",
"r728",
"r735",
"r787",
"r788",
"r789",
"r790",
"r791"
],
"lang": {
"en-us": {
"role": {
"label": "Statistical Measurement [Domain]",
"terseLabel": "Statistical Measurement [Domain]"
}
}
},
"localname": "RangeMember",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "domainItemType"
},
"srt_RepurchaseAgreementCounterpartyNameDomain": {
"auth_ref": [
"r59r229",
"r60r230",
"r120r349",
"r121r379",
"r277r652",
"r320r653"
],
"lang": {
"en-us": {
"role": {
"label": "Counterparty Name [Domain]",
"terseLabel": "Counterparty Name [Domain]"
}
}
},
"localname": "RepurchaseAgreementCounterpartyNameDomain",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "domainItemType"
},
"srt_RestatementAdjustmentMember": {
"auth_ref": [
"r125",
"r126",
"r127",
"r129",
"r130",
"r134",
"r135",
"r137",
"r139",
"r140",
"r142",
"r143",
"r163",
"r508",
"r509"
],
"lang": {
"en-us": {
"role": {
"label": "Revision of Prior Period, Adjustment [Member]",
"terseLabel": "Revision of Prior Period, Adjustment"
}
}
},
"localname": "RestatementAdjustmentMember",
"nsuri": "http://fasb.org/srt/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "domainItemType"
},
"srt_RestatementAxis": {
"auth_ref": [
"r3",
"r125",
"r126",
"r127",
"r129",
"r130",
"r134",
"r135",
"r136",
"r137",
"r139",
"r140",
"r141",
"r142",
"r143",
"r144",
"r163",
"r226",
"r227",
"r424",
"r456",
"r504",
"r508",
"r509",
"r510",
"r665",
"r666",
"r667",
"r668",
"r669",
"r670",
"r671",
"r672",
"r673",
"r674",
"r716",
"r717"
],
"lang": {
"en-us": {
"role": {
"label": "Revision of Prior Period [Axis]",
"terseLabel": "Revision of Prior Period [Axis]"
}
}
},
"localname": "RestatementAxis",
"nsuri": "http://fasb.org/srt/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "stringItemType"
},
"srt_RestatementDomain": {
"auth_ref": [
"r3",
"r125",
"r126",
"r127",
"r129",
"r130",
"r134",
"r135",
"r136",
"r137",
"r139",
"r140",
"r141",
"r142",
"r143",
"r144",
"r163",
"r226",
"r227",
"r424",
"r456",
"r504",
"r508",
"r509",
"r510",
"r665",
"r666",
"r667",
"r668",
"r669",
"r670",
"r671",
"r672",
"r673",
"r674",
"r716",
"r717"
],
"lang": {
"en-us": {
"role": {
"label": "Revision of Prior Period [Domain]",
"terseLabel": "Revision of Prior Period [Domain]"
}
}
},
"localname": "RestatementDomain",
"nsuri": "http://fasb.org/srt/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "domainItemType"
},
"srt_ScenarioForecastMember": {
"auth_ref": [
"r138BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "domainItemType"
},
"srt_ScenarioForecastMember": {
"auth_ref": [
"r428",
"r385r719"
],
"lang": {
"en-us": {
"role": {
"label": "Forecast [Member]",
"terseLabel": "Forecast"
}
}
},
"localname": "ScenarioForecastMember",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsINDEBTEDNESSMinimumRevenueCovenantDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"srt_ScenarioUnspecifiedDomain": {
"auth_ref": [
"r138r249",
"r144r428",
"r385r695",
"r719"
],
"lang": {
"en-us": {
"role": {
"label": "Scenario [Domain]",
"terseLabel": "Scenario [Domain]"
}
}
},
"localname": "ScenarioUnspecifiedDomain",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsINDEBTEDNESSMinimumRevenueCovenantDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"srt_SegmentGeographicalDomain": {
"auth_ref": [
"r204r292",
"r205r293",
"r364r638",
"r369r639",
"r660r640",
"r683r641",
"r684r642",
"r685r643",
"r686r644",
"r687r645",
"r688r646",
"r689r668",
"r690r681",
"r691r739"
],
"lang": {
"en-us": {
"role": {
"label": "Geographical [Domain]",
"terseLabel": "Geographical [Domain]"
}
}
},
"localname": "SegmentGeographicalDomain",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "domainItemType"
},
"srt_StatementGeographicalAxis": {
"auth_ref": [
"r204r292",
"r205r293",
"r364r631",
"r369r638",
"r660r639",
"r676r640",
"r683r641",
"r684r642",
"r685r643",
"r686r644",
"r687r645",
"r688r646",
"r689r668",
"r690r681",
"r691r739"
],
"lang": {
"en-us": {
"role": {
"label": "Geographical [Axis]",
"terseLabel": "Geographical [Axis]"
}
}
},
"localname": "StatementGeographicalAxis",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "stringItemType"
},
"srt_StatementScenarioAxis": {
"auth_ref": [
"r138r249",
"r144r428",
"r254r695",
"r385r696",
"r571r719"
],
"lang": {
"en-us": {
"role": {
"label": "Scenario [Axis]",
"terseLabel": "Scenario [Axis]"
}
}
},
"localname": "StatementScenarioAxis",
"nsuri": "http://fasb.org/srt/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsINDEBTEDNESSMinimumRevenueCovenantDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AccountingPoliciesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Accounting Policies [Abstract]"
}
}
},
"localname": "AccountingPoliciesAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_AccountingStandardsUpdate201602Member": {
"auth_ref": [
"r551r548"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accounting Standards Update 2016-02 Leases (Topic 842).",
"label": "Accounting Standards Update 2016-02 [Member]",
"terseLabel": "Accounting Standards Update 2016-02"
}
}
},
"localname": "AccountingStandardsUpdate201602Member",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccountsPayableAccruedLiabilitiesAndOtherLiabilitiesDisclosureCurrentTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for accounts payable, accrued expenses, and other liabilities that are classified as current at the end of the reporting period.",
"label": "Accounts Payable, Accrued Liabilities, and Other Liabilities Disclosure, Current [Text Block]",
"terseLabel": "ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES"
}
}
},
"localname": "AccountsPayableAccruedLiabilitiesAndOtherLiabilitiesDisclosureCurrentTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIES"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_AccountsPayableAndOtherAccruedLiabilitiesCurrent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 32.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liabilities incurred to vendors for goods and services received, and accrued liabilities classified as other, payable within one year or the normal operating cycle, if longer.",
"label": "Accounts Payable and Other Accrued Liabilities, Current",
"terseLabel": "Accrued expenses and other current liabilities",
"totalLabel": "Total"
}
}
},
"localname": "AccountsPayableAndOtherAccruedLiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccountsPayableCurrent": {
"auth_ref": [
"r43r10",
"r568r679"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 43.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Accounts Payable, Current",
"terseLabel": "Accounts payable"
}
}
},
"localname": "AccountsPayableCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION"
],
"xbrltype": "monetaryItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccountsReceivableMember": {
"auth_ref": [
"r633"
],
"lang": {
"en-us": {
"role": {
"documentation": "Due from customers or clients for goods or services that have been delivered or sold.",
"label": "Accounts Receivable [Member]",
"terseLabel": "Accounts Receivable"
}
}
},
"localname": "AccountsReceivableMember",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AccountsReceivableNetCurrent": {
"auth_ref": [
"r9r295",
"r29",
"r208",
"r209r296"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 13.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after allowance for credit loss, of right to consideration from customer for product sold and service rendered in normal course of business, classified as current.",
"label": "Accounts Receivable, after Allowance for Credit Loss, Current",
"terseLabel": "Accounts receivable"
}
}
},
"localname": "AccountsReceivableNetCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccretionAmortizationOfDiscountsAndPremiumsInvestments": {
"auth_ref": [
"r102r46"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 710.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The sum of the periodic adjustments of the differences between securities' face values and purchase prices that are charged against earnings. This is called accretion if the security was purchased at a discount and amortization if it was purchased at premium. As a noncash item, this element is an adjustment to net income when calculating cash provided by or used in operations using the indirect method.",
"label": "Accretion (Amortization) of Discounts and Premiums, Investments",
"negatedTerseLabel": "Accretion and amortization of interest income"
}
}
},
"localname": "AccretionAmortizationOfDiscountsAndPremiumsInvestments",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccruedMarketingCostsCurrent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails": {
"order": 21.0,
"parentTag": "us-gaap_AccountsPayableAndOtherAccruedLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of obligations incurred through that date and payable for the marketing, trade and selling of the entity's goods and services. Marketing costs would include expenditures for planning and executing the conception, pricing, promotion, and distribution of ideas, goods, and services; costs of public relations and corporate promotions; and obligations incurred and payable for sales discounts, rebates, price protection programs, etc. offered to customers and under government programs. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Accrued Marketing Costs, Current",
"terseLabel": "Commercial and marketing related costs"
}
}
},
"localname": "AccruedMarketingCostsCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccruedProfessionalFeesCurrent": {
"auth_ref": [
"r13",
"r14",
"r47r14"
],
"calculation": {
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails": {
"order": 53.0,
"parentTag": "us-gaap_AccountsPayableAndOtherAccruedLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying value as of the balance sheet date of obligations incurred through that date and payable for professional fees, such as for legal and accounting services received. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Accrued Professional Fees, Current",
"terseLabel": "Professional services"
}
}
},
"localname": "AccruedProfessionalFeesCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccumulatedDepreciationDepletionAndAmortizationPropertyPlantAndEquipment": {
"auth_ref": [
"r41r73",
"r244r194"
],
"calculation": {
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails": {
"order": 2.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentNet",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of accumulated depreciation, depletion and amortization for physical assets used in the normal conduct of business to produce goods and services.",
"label": "Accumulated Depreciation, Depletion and Amortization, Property, Plant, and Equipment",
"negatedTerseLabel": "Less: accumulated depreciation"
}
}
},
"localname": "AccumulatedDepreciationDepletionAndAmortizationPropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccumulatedOtherComprehensiveIncomeLossNetOfTax": {
"auth_ref": [
"r32r25",
"r64r26",
"r65r27",
"r66r201",
"r647r613",
"r670r625",
"r674r629"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 5.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated change in equity from transactions and other events and circumstances from non-owner sources, net of tax effect, at period end. Excludes Net Income (Loss), and accumulated changes in equity from transactions resulting from investments by owners and distributions to owners. Includes foreign currency translation items, certain pension adjustments, unrealized gains and losses on certain investments in debt and equity securities, other than temporary impairment (OTTI) losses related to factors other than credit losses on available-for-sale and held-to-maturity debt securities that an entity does not intend to sell and it is not more likely than not that the entity will be required to sell before recovery of the amortized cost basis, as well as changes in the fair value of derivatives related to the effective portion of a designated cash flow hedge.",
"label": "Accumulated Other Comprehensive Income (Loss), Net of Tax",
"terseLabel": "Accumulated other comprehensive (loss) income"
}
}
},
"localname": "AccumulatedOtherComprehensiveIncomeLossNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AccumulatedOtherComprehensiveIncomeMember": {
"auth_ref": [
"r63r24",
"r66r27",
"r74r129",
"r75r568",
"r76r620",
"r125r621",
"r126r703",
"r127r704",
"r486r705",
"r665",
"r666r716",
"r717",
"r718"
],
"lang": {
"en-us": {
"role": {
"documentation": "Accumulated increase (decrease) in equity from transactions and other events and circumstances from non-owner sources, attributable to the parent. Excludes net income (loss), and accumulated changes in equity from transactions resulting from investments by owners and distributions to owners.",
"label": "AOCI Attributable to Parent [Member]",
"terseLabel": "Accumulated Other Comprehensive IncomeLoss"
}
}
},
"localname": "AccumulatedOtherComprehensiveIncomeMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "domainItemType"
},
"us-gaap_AdditionalPaidInCapital": {
"auth_ref": [
"r30r5",
"r424",
"r568r679"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 4.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of excess of issue price over par or stated value of stock and from other transaction involving stock or stockholder. Includes, but is not limited to, additional paid-in capital (APIC) for common and preferred stock.",
"label": "Additional Paid in Capital",
"terseLabel": "Additional paid-in capital"
}
}
},
"localname": "AdditionalPaidInCapital",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AdditionalPaidInCapitalMember": {
"auth_ref": [
"r125r467",
"r126r468",
"r127r469",
"r421r716",
"r422r717",
"r423r718",
"r508r775"
],
"lang": {
"en-us": {
"role": {
"documentation": "Excess of issue price over par or stated value of the entity's capital stock and amounts received from other transactions involving the entity's stock or stockholders.",
"label": "Additional Paid-in Capital [Member]",
"terseLabel": "Additional Paid-In Capital"
}
}
},
"localname": "AdditionalPaidInCapitalMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "domainItemType"
},
"us-gaap_AdjustmentsForNewAccountingPronouncementsAxis": {
"auth_ref": [
"r0r184",
"r1r185",
"r2r186",
"r3r187",
"r4r188",
"r129r237",
"r130r238",
"r131r239",
"r132r240",
"r144r249",
"r211r297",
"r212r298",
"r222r303",
"r223r304",
"r224r305",
"r225r306",
"r226r307",
"r227r308",
"r260r467",
"r417r468",
"r418r469",
"r419r486",
"r420r487",
"r421r488",
"r422",
"r423",
"r424",
"r453",
"r454",
"r455",
"r456r489",
"r500",
"r501",
"r502",
"r503r512",
"r504r513",
"r505r514",
"r506r515",
"r507r516",
"r508r517",
"r509r518",
"r510r519",
"r526r520",
"r527r521",
"r528r522",
"r529r523",
"r530",
"r531",
"r532",
"r533",
"r557r534",
"r535",
"r536",
"r537",
"r545",
"r546",
"r549",
"r550",
"r551",
"r552",
"r563",
"r564",
"r565",
"r566",
"r567",
"r568",
"r606",
"r607",
"r608",
"r609r618",
"r663r619",
"r664r620",
"r665r621",
"r666r622",
"r667r623",
"r668r624",
"r669r625",
"r670r626",
"r671r627",
"r672r628",
"r673",
"r674",
"r713",
"r714",
"r715",
"r716",
"r717r629"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by amendment to accounting standards.",
"label": "Accounting Standards Update [Axis]",
"terseLabel": "Accounting Standards Update [Axis]"
}
}
},
"localname": "AdjustmentsForNewAccountingPronouncementsAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AdjustmentsToAdditionalPaidInCapitalSharebasedCompensationRequisiteServicePeriodRecognitionValue": {
"auth_ref": [
"r388r105",
"r390r106",
"r427",
"r428r430"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase to additional paid-in capital (APIC) for recognition of cost for award under share-based payment arrangement.",
"label": "APIC, Share-basedBased Payment Arrangement, Increase for Cost Recognition",
"terseLabel": "Stock-based compensation expense"
}
}
},
"localname": "AdjustmentsToAdditionalPaidInCapitalSharebasedCompensationRequisiteServicePeriodRecognitionValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AdjustmentsToAdditionalPaidInCapitalStockIssuedIssuanceCosts": {
"auth_ref": [
"r333r86",
"r340r91"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of decrease in additional paid in capital (APIC) resulting from direct costs associated with issuing stock. Includes, but is not limited to, legal and accounting fees and direct costs associated with stock issues under a shelf registration.",
"label": "Adjustments to Additional Paid in Capital, Stock Issued, Issuance Costs",
"terseLabel": "Adjustments to Additional Paid in Capital, Stock Issued, Issuance Costscosts"
}
}
},
"localname": "AdjustmentsToAdditionalPaidInCapitalStockIssuedIssuanceCosts",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AdjustmentsToAdditionalPaidInCapitalWarrantIssued": {
"auth_ref": [
"r272",
"r333",
"r340"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase in additional paid in capital (APIC) resulting from the issuance of warrants. Includes allocation of proceeds of debt securities issued with detachable stock purchase warrants.",
"label": "Adjustments to Additional Paid in Capital, Warrant Issued",
"terseLabel": "Issuance of warrants"
}
}
},
"localname": "AdjustmentsToAdditionalPaidInCapitalWarrantIssued",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AdjustmentsToReconcileNetIncomeLossToCashProvidedByUsedInOperatingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Adjustments to Reconcile Net Income (Loss) to Cash Provided by (Used in) Operating Activities [Abstract]",
"terseLabel": "Adjustments to reconcile net loss to net cash used in operating activities:"
}
}
},
"localname": "AdjustmentsToReconcileNetIncomeLossToCashProvidedByUsedInOperatingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "stringItemType"
},
"us-gaap_AdvertisingExpense": {
"auth_ref": [
"r431r472"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount charged to advertising expense for the period, which are expenses incurred with the objective of increasing revenue for a specified brand, product or product line.",
"label": "Advertising Expense",
"terseLabel": "Advertising expenses"
}
}
},
"localname": "AdvertisingExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AllocatedShareBasedCompensationExpense": {
"auth_ref": [
"r390",
"r413",
"r426r462"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expense for award under share-based payment arrangement. Excludes amount capitalized.",
"label": "Share-basedBased Payment Arrangement, Expense",
"terseLabel": "Stock based compensation expenses"
}
}
},
"localname": "AllocatedShareBasedCompensationExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetailsSTOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AmortizationOfDebtDiscountPremium": {
"auth_ref": [
"r84r34",
"r101r45",
"r296r149",
"r542r368"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 85.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of noncash expense included in interest expense to amortize debt discount and premium associated with the related debt instruments. Excludes amortization of financing costs. Alternate captions include noncash interest expense.",
"label": "Amortization of Debt Discount (Premium)",
"terseLabel": "Amortization of debt discount"
}
}
},
"localname": "AmortizationOfDebtDiscountPremium",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AmortizationOfIntangibleAssets": {
"auth_ref": [
"r101r45",
"r238r66",
"r240r68"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 1.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate expense charged against earnings to allocate the cost of intangible assets (nonphysical assets not used in production) in a systematic and rational manner to the periods expected to benefit from such assets. As a noncash expense, this element is added back to net income when calculating cash provided by or used in operations using the indirect method.",
"label": "Amortization of Intangible Assets",
"terseLabel": "Amortization of intangible asset"
}
}
},
"localname": "AmortizationOfIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareAmount": {
"auth_ref": [
"r160r260"
],
"lang": {
"en-us": {
"role": {
"documentation": "Securities (including those issuable pursuant to contingent stock agreements) that could potentially dilute basic earnings per share (EPS) or earnings per unit (EPU) in the future that were not included in the computation of diluted EPS or EPU because to do so would increase EPS or EPU amounts or decrease loss per share or unit amounts for the period presented.",
"label": "Antidilutive Securities Excluded from Computation of Earnings Per Share, Amount",
"terseLabel": "Antidilutive securities (in shares)"
}
}
},
"localname": "AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareAmount",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis": {
"auth_ref": [
"r160r55"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of antidilutive security.",
"label": "Antidilutive Securities [Axis]",
"terseLabel": "Antidilutive Securities [Axis]"
}
}
},
"localname": "AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Antidilutive Securities Excluded from Computation of Earnings Per Share [Line Items]",
"terseLabel": "Antidilutive Securities Excluded from Computation of Earnings Per Share [Line Items]"
}
}
},
"localname": "AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_AntidilutiveSecuritiesNameDomain": {
"auth_ref": [
"r160r55"
],
"lang": {
"en-us": {
"role": {
"documentation": "Incremental common shares attributable to securities that were not included in diluted earnings per share (EPS) because to do so would increase EPS amounts or decrease loss per share amounts for the period presented.",
"label": "Antidilutive Securities, Name [Domain]",
"terseLabel": "Antidilutive Securities, Name [Domain]"
}
}
},
"localname": "AntidilutiveSecuritiesNameDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_AreaOfRealEstateProperty": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Area of a real estate property.",
"label": "Area of Real Estate Property",
"terseLabel": "Area of office space (in sqft)"
}
}
},
"localname": "AreaOfRealEstateProperty",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "areaItemType"
},
"us-gaap_ArrangementsAndNonarrangementTransactionsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Collaborative arrangement and arrangement other than collaborative applicable to revenue-generating activity or operations.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Domain]",
"terseLabel": "Collaborative Arrangement and Arrangement Other than Collaborative [Domain]"
}
}
},
"localname": "ArrangementsAndNonarrangementTransactionsMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsREVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_Assets": {
"auth_ref": [
"r117r154",
"r186r169",
"r195r196",
"r201r226",
"r221r276",
"r261r285",
"r262r289",
"r263r302",
"r264r337",
"r265r338",
"r266r339",
"r267r340",
"r268r341",
"r269r342",
"r270r343",
"r271r344",
"r482r345",
"r487r506",
"r525r508",
"r566r529",
"r568r679",
"r623r733",
"r643r734",
"r784"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying amounts as of the balance sheet date of all assets that are recognized. Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events.",
"label": "Assets",
"totalLabel": "Total assets"
}
}
},
"localname": "Assets",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AssetsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Assets [Abstract]",
"verboseLabel": "Assets"
}
}
},
"localname": "AssetsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "stringItemType"
},
"us-gaap_AssetsCurrent": {
"auth_ref": [
"r10r190",
"r11r202",
"r57r226",
"r117r302",
"r221r337",
"r261r338",
"r262r339",
"r263r340",
"r264r341",
"r265r342",
"r266r343",
"r267r344",
"r268r345",
"r269r506",
"r270r508",
"r271r529",
"r482r679",
"r487r733",
"r525r734",
"r566",
"r568r784"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 35.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying amounts as of the balance sheet date of all assets that are expected to be realized in cash, sold, or consumed within one year (or the normal operating cycle, if longer). Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events.",
"label": "Assets, Current",
"totalLabel": "Total current assets"
}
}
},
"localname": "AssetsCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AssetsCurrentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Assets, Current [Abstract]",
"verboseLabel": "Current assets:"
}
}
},
"localname": "AssetsCurrentAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "stringItemType"
},
"us-gaap_AssetsFairValueDisclosure": {
"auth_ref": [
"r511r139"
],
"calculation": {
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of probable future economic benefits obtained or controlled by an entity as a result of past transactions or events.",
"label": "Assets, Fair Value Disclosure",
"totalLabel": "Total assets"
}
}
},
"localname": "AssetsFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AvailableForSaleSecuritiesDebtSecurities": {
"auth_ref": [
"r213r65",
"r215r299",
"r233r314",
"r630r609"
],
"calculation": {
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails": {
"order": 2.0,
"parentTag": "us-gaap_AssetsFairValueDisclosure",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Debt Securities, Available-for-saleSale",
"verboseLabel": "Debt investments"
}
}
},
"localname": "AvailableForSaleSecuritiesDebtSecurities",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_AwardTypeAxis": {
"auth_ref": [
"r391",
"r415r433",
"r434",
"r435",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r450",
"r451",
"r452",
"r453",
"r454",
"r457",
"r458",
"r459",
"r460",
"r461"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of award under share-based payment arrangement.",
"label": "Award Type [Axis]",
"terseLabel": "Award Type [Axis]"
}
}
},
"localname": "AwardTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BalanceSheetLocationAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by location on balance sheet (statement of financial position).",
"label": "Balance Sheet Location [Axis]",
"terseLabel": "Balance Sheet Location [Axis]"
}
}
},
"localname": "BalanceSheetLocationAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BalanceSheetLocationDomain": {
"auth_ref": [
"r494r135",
"r496r137"
],
"lang": {
"en-us": {
"role": {
"documentation": "Location in the balance sheet (statement of financial position).",
"label": "Balance Sheet Location [Domain]",
"terseLabel": "Balance Sheet Location [Domain]"
}
}
},
"localname": "BalanceSheetLocationDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_BasisOfAccountingPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for basis of accounting, or basis of presentation, used to prepare the financial statements (for example, US Generally Accepted Accounting Principles, Other Comprehensive Basis of Accounting, IFRS).",
"label": "Basis of Accounting, Policy [Policy Text Block]",
"terseLabel": "Basis of Presentation"
}
}
},
"localname": "BasisOfAccountingPolicyPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_BusinessAcquisitionAcquireeDomain": {
"auth_ref": [
"r383r498",
"r386r673",
"r674"
],
"lang": {
"en-us": {
"role": {
"documentation": "Identification of the acquiree in a material business combination (or series of individually immaterial business combinations), which may include the name or other type of identification of the acquiree.",
"label": "Business Acquisition, Acquiree [Domain]",
"terseLabel": "Business Acquisition, Acquiree [Domain]"
}
}
},
"localname": "BusinessAcquisitionAcquireeDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_BusinessAcquisitionAxis": {
"auth_ref": [
"r383r118",
"r386r119",
"r465r498",
"r466r673",
"r674"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by business combination or series of individually immaterial business combinations.",
"label": "Business Acquisition [Axis]",
"terseLabel": "Business Acquisition [Axis]"
}
}
},
"localname": "BusinessAcquisitionAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BusinessAcquisitionEquityInterestsIssuedOrIssuableNumberOfSharesIssued": {
"auth_ref": [
"r470r123"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares of equity interests issued or issuable to acquire entity.",
"label": "Business Acquisition, Equity Interest Issued or Issuable, Number of Shares",
"terseLabel": "Shares issuable (in shares)"
}
}
},
"localname": "BusinessAcquisitionEquityInterestsIssuedOrIssuableNumberOfSharesIssued",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_BusinessAcquisitionLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Business Acquisition [Line Items]",
"terseLabel": "Business Acquisition [Line Items]"
}
}
},
"localname": "BusinessAcquisitionLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_BusinessCombinationAndAssetAcquisitionAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Business Combination and Asset Acquisition [Abstract]"
}
}
},
"localname": "BusinessCombinationAndAssetAcquisitionAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationArrangementsChangeInAmountOfContingentConsiderationLiability1": {
"auth_ref": [
"r100r504",
"r472r709"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 6.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 1.0,
"parentTag": "us-gaap_OtherNonoperatingIncomeExpense",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in the value of a contingent consideration liability, including, but not limited to, differences arising upon settlement.",
"label": "Business Combination, Contingent Consideration Arrangements, Change in Amount of Contingent Consideration, Liability",
"negatedTerseLabel": "Change in estimated fair value of earn-out liabilities",
"verboseLabel": "Change in fair value of earn-out liabilities"
}
}
},
"localname": "BusinessCombinationContingentConsiderationArrangementsChangeInAmountOfContingentConsiderationLiability1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSFAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationLiability": {
"auth_ref": [
"r467r120",
"r468r121",
"r471r503"
],
"calculation": {
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails": {
"order": 3.0,
"parentTag": "us-gaap_FinancialLiabilitiesFairValueDisclosure",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liability recognized arising from contingent consideration in a business combination.",
"label": "Business Combination, Contingent Consideration, Liability",
"terseLabel": "Earn-out liabilities"
}
}
},
"localname": "BusinessCombinationContingentConsiderationLiability",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetailsBUSINESSCOMBINATIONWarrantsDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationLiabilityMeasurementInput": {
"auth_ref": [
"r514r526"
],
"lang": {
"en-us": {
"role": {
"documentation": "Value of input used to measure contingent consideration liability from business combination.",
"label": "Business Combination, Contingent Consideration, Liability, Measurement Input",
"terseLabel": "Earn-out liability"
}
}
},
"localname": "BusinessCombinationContingentConsiderationLiabilityMeasurementInput",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "decimalItemType"
},
"us-gaap_BusinessCombinationContingentConsiderationLiabilityNoncurrent": {
"auth_ref": [
"r467r120",
"r469r122"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 43.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liability recognized arising from contingent consideration in a business combination, expected to be settled beyond one year or the normal operating cycle, if longer.",
"label": "Business Combination, Contingent Consideration, Liability, Noncurrent",
"terseLabel": "Earn-out liabilities"
}
}
},
"localname": "BusinessCombinationContingentConsiderationLiabilityNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_BusinessCombinationDisclosureTextBlock": {
"auth_ref": [
"r473r124",
"r499"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for a business combination (or series of individually immaterial business combinations) completed during the period, including background, timing, and recognized assets and liabilities. The disclosure may include leverage buyout transactions (as applicable).",
"label": "Business Combination Disclosure [Text Block]",
"terseLabel": "BUSINESS COMBINATION"
}
}
},
"localname": "BusinessCombinationDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATION"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CapitalExpendituresIncurredButNotYetPaid": {
"auth_ref": [
"r106r49",
"r107r50",
"r108r51"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Future cash outflow to pay for purchases of fixed assets that have occurred.",
"label": "Capital Expenditures Incurred but Not yet Paid",
"terseLabel": "Purchases of property and equipment in accounts payable and accrued expenses"
}
}
},
"localname": "CapitalExpendituresIncurredButNotYetPaid",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashAndCashEquivalentsAtCarryingValue": {
"auth_ref": [
"r8r47",
"r38r192",
"r103r650"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION": {
"order": 4.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails": {
"order": 2.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 1.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation.",
"label": "Cash and Cash Equivalents, at Carrying Value",
"terseLabel": "Cash and cash equivalents"
}
}
},
"localname": "CashAndCashEquivalentsAtCarryingValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetailsUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashAndCashEquivalentsAxis": {
"auth_ref": [
"r192"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of cash and cash equivalent balance.",
"label": "Cash and Cash Equivalents [Axis]",
"terseLabel": "Cash and Cash Equivalents [Axis]"
}
}
},
"localname": "CashAndCashEquivalentsAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CashAndCashEquivalentsFairValueDisclosure": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails": {
"order": 1.0,
"parentTag": "us-gaap_AssetsFairValueDisclosure",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value portion of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Cash and Cash Equivalents, Fair Value Disclosure",
"terseLabel": "Cash and cash Equivalents"
}
}
},
"localname": "CashAndCashEquivalentsFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashAndCashEquivalentsRestrictedCashAndCashEquivalentsPolicy": {
"auth_ref": [
"r16r48",
"r104",
"r622r153"
],
"lang": {
"en-us": {
"role": {
"documentation": "Entity's cash and cash equivalents accounting policy with respect to restricted balances. Restrictions may include legally restricted deposits held as compensating balances against short-term borrowing arrangements, contracts entered into with others, or company statements of intention with regard to particular deposits; however, time deposits and short-term certificates of deposit are not generally included in legally restricted deposits.",
"label": "Cash and Cash Equivalents, Restricted Cash and Cash Equivalents, Policy [Policy Text Block]",
"terseLabel": "Cash, Cash Equivalents and Restricted Cash"
}
}
},
"localname": "CashAndCashEquivalentsRestrictedCashAndCashEquivalentsPolicy",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CashCashEquivalentsAndShortTermInvestments": {
"auth_ref": [
"r38r702"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Cash includes currency on hand as well as demand deposits with banks or financial institutions. It also includes other kinds of accounts that have the general characteristics of demand deposits in that the customer may deposit additional funds at any time and effectively may withdraw funds at any time without prior notice or penalty. Cash equivalents, excluding items classified as marketable securities, include short-term, highly liquid Investments that are both readily convertible to known amounts of cash, and so near their maturity that they present minimal risk of changes in value because of changes in interest rates. Generally, only investments with original maturities of three months or less qualify under that definition. Original maturity means original maturity to the entity holding the investment. For example, both a three-month US Treasury bill and a three-year Treasury note purchased three months from maturity qualify as cash equivalents. However, a Treasury note purchased three years ago does not become a cash equivalent when its remaining maturity is three months. Short-term investments, exclusive of cash equivalents, generally consist of marketable securities intended to be sold within one year (or the normal operating cycle if longer) and may include trading securities, available-for-sale securities, or held-to-maturity securities (if maturing within one year), as applicable.",
"label": "Cash, Cash Equivalents, and Short-termTerm Investments",
"terseLabel": "Cash and cash equivalents and short-term investments"
}
}
},
"localname": "CashCashEquivalentsAndShortTermInvestments",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents": {
"auth_ref": [
"r97r41",
"r103r47",
"r109r52"
],
"calculation": {
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash and cash equivalents, and cash and cash equivalents restricted to withdrawal or usage. Excludes amount for disposal group and discontinued operations. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Cash, Cash Equivalents, Restricted Cash, and Restricted Cash Equivalents",
"periodEndLabel": "Cash, cash equivalents and restricted cash\u2014end of period",
"periodStartLabel": "Cash, cash equivalents and restricted cash\u2014beginning of period",
"totalLabel": "Total cash, cash equivalents, and restricted cash"
}
}
},
"localname": "CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseIncludingExchangeRateEffect": {
"auth_ref": [
"r97r41",
"r534r148"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in cash, cash equivalents, and cash and cash equivalents restricted to withdrawal or usage; including effect from exchange rate change. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Cash, Cash Equivalents, Restricted Cash, and Restricted Cash Equivalents, Period Increase (Decrease), Including Exchange Rate Effect",
"totalLabel": "Net (decrease) increase in cash, cash equivalents and restricted cash"
}
}
},
"localname": "CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseIncludingExchangeRateEffect",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CashFlowNoncashInvestingAndFinancingActivitiesDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Cash Flow, Noncash Investing and Financing Activities Disclosure [Abstract]",
"terseLabel": "Supplemental disclosure of non-cash investing and financing information:"
}
}
},
"localname": "CashFlowNoncashInvestingAndFinancingActivitiesDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "stringItemType"
},
"us-gaap_ClassOfStockDomain": {
"auth_ref": [
"r114r198",
"r117r199",
"r148r200",
"r149r226",
"r154r253",
"r157r254",
"r159r257",
"r167",
"r168",
"r169",
"r221",
"r261",
"r265r259",
"r266",
"r267",
"r270r302",
"r271r337",
"r316r339",
"r317r340",
"r322r341",
"r326r344",
"r525",
"r703r345",
"r376",
"r377",
"r381",
"r385",
"r392",
"r529",
"r649",
"r694",
"r712",
"r720"
],
"lang": {
"en-us": {
"role": {
"documentation": "Share of stock differentiated by the voting rights the holder receives. Examples include, but are not limited to, common stock, redeemable preferred stock, nonredeemable preferred stock, and convertible stock.",
"label": "Class of Stock [Domain]",
"terseLabel": "Class of Stock [Domain]"
}
}
},
"localname": "ClassOfStockDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/Cover",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParenthetical"
],
"xbrltype": "domainItemType"
},
"us-gaap_ClassOfStockLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Class of Stock [Line Items]",
"terseLabel": "Class of Stock [Line Items]"
}
}
},
"localname": "ClassOfStockLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ClassOfWarrantOrRightAxis": {
"auth_ref": [
"r341UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "domainItemType"
},
"us-gaap_ClassOfWarrantOrRightAxis": {
"auth_ref": [
"r92",
"r389r95"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of warrant or right issued.",
"label": "Class of Warrant or Right [Axis]",
"terseLabel": "Class of Warrant or Right [Axis]"
}
}
},
"localname": "ClassOfWarrantOrRightAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ClassOfWarrantOrRightDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Name of the class or type of warrant or right outstanding. Warrants and rights represent derivative securities that give the holder the right to purchase securities (usually equity) from the issuer at a specific price within a certain time frame. Warrants are often included in a new debt issue to entice investors by a higher return potential. The main difference between warrants and call options is that warrants are issued and guaranteed by the company, whereas options are exchange instruments and are not issued by the company. Also, the lifetime of a warrant is often measured in years, while the lifetime of a typical option is measured in months.",
"label": "Class of Warrant or Right [Domain]",
"terseLabel": "Class of Warrant or Right [Domain]"
}
}
},
"localname": "ClassOfWarrantOrRightDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ClassOfWarrantOrRightExercisePriceOfWarrantsOrRights1": {
"auth_ref": [
"r334r393"
],
"lang": {
"en-us": {
"role": {
"documentation": "Exercise price per share or per unit of warrants or rights outstanding.",
"label": "Class of Warrant or Right, Exercise Price of Warrants or Rights",
"terseLabel": "Stock issued, exercise price",
"verboseLabel": "Exercise price (in dollars per share)"
}
}
},
"localname": "ClassOfWarrantOrRightExercisePriceOfWarrantsOrRights1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails
],
"xbrltype": "perShareItemType"
},
"us-gaap_ClassOfWarrantOrRightLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Class of Warrant or Right [Line Items]",
"terseLabel": "Class of Warrant or Right [Line Items]"
}
}
},
"localname": "ClassOfWarrantOrRightLineItems",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails"
],
"xbrltype": "perShareItemTypestringItemType"
},
"us-gaap_ClassOfWarrantOrRightNumberOfSecuritiesCalledByEachWarrantOrRight": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of securities into which each warrant or right may be converted. For example, but not limited to, each warrant may be converted into two shares.",
"label": "Class of Warrant or Right, Number of Securities Called by Each Warrant or Right",
"terseLabel": "Number of securities called by each warrant or right"
}
}
},
"localname": "ClassOfWarrantOrRightNumberOfSecuritiesCalledByEachWarrantOrRight",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ClassOfWarrantOrRightNumberOfSecuritiesCalledByWarrantsOrRights": {
"auth_ref": [
"r334r393"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of securities into which the class of warrant or right may be converted. For example, but not limited to, 500,000 warrants may be converted into 1,000,000 shares.",
"label": "Class of Warrant or Right, Number of Securities Called by Warrants or Rights",
"terseLabel": "Number of common stock into which the class of warrant or right converted (in warrants)"
}
}
},
"localname": "ClassOfWarrantOrRightNumberOfSecuritiesCalledByWarrantsOrRights",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ClassOfWarrantOrRightOutstanding": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of warrants or rights outstanding.",
"label": "Class of Warrant or Right, Outstanding",
"terseLabel": "Warrants outstanding (in shares)"
}
}
},
"localname": "ClassOfWarrantOrRightOutstanding",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CollaborativeArrangementDisclosureTextBlockgaap_ClassOfWarrantOrRightTable": {
"auth_ref": [
"r475r92",
"r476",
"r479r95"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosureDisclosure for collaborative arrangements in which the entity is a participant, including a) information about the nature and purpose of such arrangements; b) its rights and obligations thereunder; c) the accounting policy for collaborative arrangements; and d) the income statement classification and amounts attributable to transactions arising from the collaborative arrangement between participants.",
"label": "Collaborative Arrangement Disclosure [Text Blockwarrants or rights issued, which includes the title of issue of securities called for by warrants and rights outstanding, the aggregate amount of securities called for by warrants and rights outstanding, the date from which the warrants or rights are exercisable, and the price at which the warrant or right is exercisable.",
"label": "Class of Warrant or Right [Table]",
"terseLabel": "PAST COLLABORATION AND LICENSE AGREEMENTClass of Warrant or Right [Table]"
}
}
},
"localname": "CollaborativeArrangementDisclosureTextBlockClassOfWarrantOrRightTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTCAPITALSTOCKDetails"
],
"xbrltype": "textBlockItemTypestringItemType"
},
"us-gaap_CollaborativeArrangementMember": {
"auth_ref": [
"r478r505"
],
"lang": {
"en-us": {
"role": {
"documentation": "Contractual arrangement that involves two or more parties that both: (i) actively participate in a joint operating activity and (ii) are exposed to significant risks and rewards that depend on the commercial success of the joint operating activity.",
"label": "Collaborative Arrangement [Member]",
"terseLabel": "Collaborative Arrangement"
}
}
},
"localname": "CollaborativeArrangementMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsREVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CollaborativeArrangementsAndNoncollaborativeArrangementTransactionsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Line Items]",
"terseLabel": "Collaborative Arrangement and Arrangement Other than Collaborative [Line Items]"
}
}
},
"localname": "CollaborativeArrangementsAndNoncollaborativeArrangementTransactionsLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsREVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CommercialPaperMember": {
"auth_ref": [
"r257r81"
],
"lang": {
"en-us": {
"role": {
"documentation": "Unsecured promissory note (generally negotiable) that provides institutions with short-term funds.",
"label": "Commercial Paper [Member]",
"terseLabel": "Commercial paper"
}
}
},
"localname": "CommercialPaperMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CommitmentsAndContingencies": {
"auth_ref": [
"r52r20",
"r253r161",
"r632",
"r651r176"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 32.0,
"parentTag": "us-gaap_LiabilitiesAndStockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Represents the caption on the face of the balance sheet to indicate that the entity has entered into (1) purchase or supply arrangements that will require expending a portion of its resources to meet the terms thereof, and (2) is exposed to potential losses or, less frequently, gains, arising from (a) possible claims against a company's resources due to future performance under contract terms, and (b) possible losses or likely gains from uncertainties that will ultimately be resolved when one or more future events that are deemed likely to occur do occur or fail to occur.",
"label": "Commitments and Contingencies",
"verboseLabel": "Commitments and contingencies (Note 89)"
}
}
},
"localname": "CommitmentsAndContingencies",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CommitmentsAndContingenciesDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Commitments and Contingencies Disclosure [Abstract]"
}
}
},
"localname": "CommitmentsAndContingenciesDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_CommitmentsAndContingenciesDisclosureTextBlock": {
"auth_ref": [
"r250r79",
"r251r331",
"r252r332",
"r255r634",
"r679r732"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for commitments and contingencies.",
"label": "Commitments and Contingencies Disclosure [Text Block]",
"terseLabel": "COMMITMENTS AND CONTINGENCIES"
}
}
},
"localname": "CommitmentsAndContingenciesDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIES"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CommonClassAMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Classification of common stock representing ownership interest in a corporation.",
"label": "Common Class A [Member]",
"terseLabel": "Class A Common Stock"
}
}
},
"localname": "CommonClassAMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/Cover",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CommonStockCapitalSharesReservedForFutureIssuance": {
"auth_ref": [
"r54r21"
],
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate number of common shares reserved for future issuance.",
"label": "Common Stock, Capital Shares Reserved for Future Issuance",
"terseLabel": "Total number of shares (in shares)"
}
}
},
"localname": "CommonStockCapitalSharesReservedForFutureIssuance",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CommonStockMember": {
"auth_ref": [
"r125r716",
"r126r717",
"r508r775"
],
"lang": {
"en-us": {
"role": {
"documentation": "Stock that is subordinate to all other stock of the issuer.",
"label": "Common Stock [Member]",
"terseLabel": "Common\u00a0Stock"
}
}
},
"localname": "CommonStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",2022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "domainItemType"
},
"us-gaap_CommonStockParOrStatedValuePerShare": {
"auth_ref": [
"r28r4"
],
"lang": {
"en-us": {
"role": {
"documentation": "Face amount or stated value per share of common stock.",
"label": "Common Stock, Par or Stated Value Per Share",
"terseLabel": "Common stock, par value (in dollars per share)"
}
}
},
"localname": "CommonStockParOrStatedValuePerShare",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParenthetical",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "perShareItemType"
},
"us-gaap_CommonStockSharesAuthorized": {
"auth_ref": [
"r28r4"
],
"lang": {
"en-us": {
"role": {
"documentation": "The maximum number of common shares permitted to be issued by an entity's charter and bylaws.",
"label": "Common Stock, Shares Authorized",
"terseLabel": "Common stock, authorized (in shares)"
}
}
},
"localname": "CommonStockSharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CommonStockSharesIssued": {
"auth_ref": [
"r28r4"
],
"lang": {
"en-us": {
"role": {
"documentation": "Total number of common shares of an entity that have been sold or granted to shareholders (includes common shares that were issued, repurchased and remain in the treasury). These shares represent capital invested by the firm's shareholders and owners, and may be all or only a portion of the number of shares authorized. Shares issued include shares outstanding and shares held in the treasury.",
"label": "Common Stock, Shares, Issued",
"terseLabel": "Common stock, issued (in shares)"
}
}
},
"localname": "CommonStockSharesIssued",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CommonStockSharesOutstanding": {
"auth_ref": [
"r28r4",
"r333r86"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares of common stock outstanding. Common stock represent the ownership interest in a corporation.",
"label": "Common Stock, Shares, Outstanding",
"terseLabel": "Common stock, outstanding (in shares)"
}
}
},
"localname": "CommonStockSharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "sharesItemType"
},
"us-gaap_CommonStockValue": {
"auth_ref": [
"r28r4",
"r568r679"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 1.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate par or stated value of issued nonredeemable common stock (or common stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable common shares, par value and other disclosure concepts are in another section within stockholders' equity.",
"label": "Common Stock, Value, Issued",
"verboseLabel": "Common stock, $0.0001 par value; 690,000,000 shares authorized as of December\u00a031, 2021;2022; and 140,454,086 and 137,836,028 and 106,721,864 shares issued and outstanding as of December\u00a031, 20212022 and December\u00a031, 20202021, respectively"
}
}
},
"localname": "CommonStockValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CompensationExpenseExcludingCostOfGoodAndServiceSold": {
"auth_ref": [
"r706"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expense for salary, wage, profit sharing; incentive and equity-based compensation; and other employee benefit. Other employee benefit expense includes, but is not limited to, service component of net periodic benefit cost for defined benefit plan. Excludes compensation cost in cost of good and service sold.",
"label": "Compensation Expense, Excluding Cost of Good and Service Sold",
"terseLabel": "Compensation Expense, Excluding Cost of Good and Service Sold"
}
}
},
"localname": "CompensationExpenseExcludingCostOfGoodAndServiceSold",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ComprehensiveIncomeNetOfTax": {
"auth_ref": [
"r69r28",
"r71r208",
"r72r210",
"r78r215",
"r634r610",
"r654r614"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of increase (decrease) in equity from transactions and other events and circumstances from net income and other comprehensive income, attributable to parent entity. Excludes changes in equity resulting from investments by owners and distributions to owners.",
"label": "Comprehensive Income (Loss), Net of Tax, Attributable to Parent",
"totalLabel": "Comprehensive loss"
}
}
},
"localname": "ComprehensiveIncomeNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ComprehensiveIncomePolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for comprehensive income.",
"label": "Comprehensive Income, Policy [Policy Text Block]",
"terseLabel": "Comprehensive Loss"
}
}
},
"localname": "ComprehensiveIncomePolicyPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ConcentrationRiskBenchmarkDomain": {
"auth_ref": [
"r177r58",
"r178r59",
"r206r146",
"r522r147",
"r523r294",
"r678r633"
],
"lang": {
"en-us": {
"role": {
"documentation": "The denominator in a calculation of a disclosed concentration risk percentage.",
"label": "Concentration Risk Benchmark [Domain]",
"terseLabel": "Concentration Risk Benchmark [Domain]"
}
}
},
"localname": "ConcentrationRiskBenchmarkDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ConcentrationRiskByBenchmarkAxis": {
"auth_ref": [
"r177r58",
"r178r59",
"r206r146",
"r522r147",
"r523r294",
"r675r630",
"r678r633"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by benchmark of concentration risk.",
"label": "Concentration Risk Benchmark [Axis]",
"terseLabel": "Concentration Risk Benchmark [Axis]"
}
}
},
"localname": "ConcentrationRiskByBenchmarkAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ConcentrationRiskByTypeAxis": {
"auth_ref": [
"r177r58",
"r178r59",
"r206r146",
"r522r147",
"r523r294",
"r675r633",
"r678r796"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of concentration risk, for example, but not limited to, asset, liability, net assets, geographic, customer, employees, supplier, lender.",
"label": "Concentration Risk Type [Axis]",
"terseLabel": "Concentration Risk Type [Axis]"
}
}
},
"localname": "ConcentrationRiskByTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ConcentrationRiskCreditRisk": {
"auth_ref": [
"r172r166",
"r640r270"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for credit risk.",
"label": "Concentration Risk, Credit Risk, Policy [Policy Text Block]",
"verboseLabelterseLabel": "Concentration of Credit Risk"
}
}
},
"localname": "ConcentrationRiskCreditRisk",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ConcentrationRiskLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Concentration Risk [Line Items]",
"terseLabel": "Concentration Risk [Line Items]"
}
}
},
"localname": "ConcentrationRiskLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ConcentrationRiskPercentage1": {
"auth_ref": [
"r177gaap_ConcentrationRiskPercentage1": {
"auth_ref": [
"r58",
"r178r59",
"r206r146",
"r522r147",
"r523r294"
],
"lang": {
"en-us": {
"role": {
"documentation": "For an entity that discloses a concentration risk in relation to quantitative amount, which serves as the \"benchmark\" (or denominator) in the equation, this concept represents the concentration percentage derived from the division.",
"label": "Concentration Risk, Percentage",
"terseLabel": "Concentration percentage"
}
}
},
"localname": "ConcentrationRiskPercentage1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ConcentrationRiskTable": {
"auth_ref": [
"r175",
"r177",
"r178",
"r179",
"r522",
"r524",
"r678"
],
"lang": {
"en-us": {
"role": {
"documentation": "Describes the nature of a concentration, a benchmark to which it is compared, and the percentage that the risk is to the benchmark.",
"label": "Concentration Risk [Table]",
"terseLabel": "Concentration Risk [Table]"
}
}
},
"localname": "ConcentrationRiskTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ConcentrationRiskTypeDomain": {
"auth_ref": [
"r177SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ConcentrationRiskTypeDomain": {
"auth_ref": [
"r58",
"r178r59",
"r206r146",
"r522r147",
"r523r294",
"r678r633"
],
"lang": {
"en-us": {
"role": {
"documentation": "For an entity that discloses a concentration risk as a percentage of some financial balance or benchmark, identifies the type (for example, asset, liability, net assets, geographic, customer, employees, supplier, lender) of the concentration.",
"label": "Concentration Risk Type [Domain]",
"terseLabel": "Concentration Risk Type [Domain]"
}
}
},
"localname": "ConcentrationRiskTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ConsolidationPolicyTextBlock": {
"auth_ref": [
"r111r127",
"r484r654"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy regarding (1) the principles it follows in consolidating or combining the separate financial statements, including the principles followed in determining the inclusion or exclusion of subsidiaries or other entities in the consolidated or combined financial statements and (2) its treatment of interests (for example, common stock, a partnership interest or other means of exerting influence) in other entities, for example consolidation or use of the equity or cost methods of accounting. The accounting policy may also address the accounting treatment for intercompany accounts and transactions, noncontrolling interest, and the income statement treatment in consolidation for issuances of stock by a subsidiary.",
"label": "Consolidation, Policy [Policy Text Block]",
"verboseLabel": "Principles of Consolidation"
}
}
},
"localname": "ConsolidationPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ConstructionInProgressMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Structure or a modification to a structure under construction. Includes recently completed structures or modifications to structures that have not been placed into service.",
"label": "Construction in Progress [Member]",
"terseLabel": "Construction inIn process"
}
}
},
"localname": "ConstructionInProgressMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ContractWithCustomerLiabilityCumulativeCatchUpAdjustmentToRevenueChangeInEstimateOfTransactionPrice": {
"auth_ref": [
"r345"
],
"crdr": "creditgaap_ContractWithCustomerAssetAndLiabilityTableTextBlock": {
"auth_ref": [
"r738"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of receivable, contract asset, and contract liability from contract with customer. Includes, but is not limited to, change in contract asset and contract liability.",
"label": "Contract with Customer, Contract Asset, Contract Liability, and Receivable [Table Text Block]",
"terseLabel": "Contract assets and liabilities"
}
}
},
"localname": "ContractWithCustomerAssetAndLiabilityTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ContractWithCustomerAssetNet": {
"auth_ref": [
"r395",
"r397",
"r416"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease), after allowance for credit loss, of right to consideration in revenue recognized for cumulative catch-up adjustment from change in estimate of transaction price which (increases) decreases obligation to transferexchange for good or service transferred to customer for which consideration from customer has been received or is due. Includes, but is not limited to, change in assessment of whether estimate of variable consideration is constrainedwhen right is conditioned on something other than passage of time.",
"label": "Contract with Customer, Liability, Cumulative Catch-up Adjustment to Revenue, Change in Estimate of Transaction PriceAsset, after Allowance for Credit Loss",
"terseLabel": "Change in transaction priceTrade accounts receivable"
}
}
},
"localname": "ContractWithCustomerLiabilityCumulativeCatchUpAdjustmentToRevenueChangeInEstimateOfTransactionPriceContractWithCustomerAssetNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ContractWithCustomerLiabilityCurrent": {
"auth_ref": [
"r343REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ContractWithCustomerAssetNetAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Contract with Customer, Asset, after Allowance for Credit Loss [Abstract]",
"terseLabel": "Contract assets"
}
}
},
"localname": "ContractWithCustomerAssetNetAbstract",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ContractWithCustomerLiabilityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Contract with Customer, Liability [Abstract]",
"terseLabel": "Contract liabilities"
}
}
},
"localname": "ContractWithCustomerLiabilityAbstract",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ContractWithCustomerLiabilityCurrent": {
"auth_ref": [
"r395",
"r344r396",
"r365r416"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 14.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of obligation to transfer good or service to customer for which consideration has been received or is receivable, classified as current.",
"label": "Contract with Customer, Liability, Current",
"terseLabel": "Deferred revenues",
"verboseLabel": "Deferred revenue - current"
}
}
},
"localname": "ContractWithCustomerLiabilityCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ContractWithCustomerLiabilityNoncurrent": {
"auth_ref": [
"r395",
"r396",
"r416"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of obligation to transfer good or service to customer for which consideration has been received or is receivable, classified as noncurrent.",
"label": "Contract with Customer, Liability, Noncurrent",
"terseLabel": "Deferred revenue - non-current"
}
}
},
"localname": "ContractWithCustomerLiabilityNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ContractWithCustomerLiabilityRevenueRecognized": {
"auth_ref": [
"r366r417"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of revenue recognized that was previously included in balance of obligation to transfer good or service to customer for which consideration from customer has been received or is due.",
"label": "Contract with Customer, Liability, Revenue Recognized",
"terseLabel": "Revenue recognized"
}
}
},
"localname": "ContractWithCustomerLiabilityRevenueRecognized",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsREVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ConvertiblePreferredStockMember": {
"auth_ref": [
"r316r376",
"r317r377",
"r322r381"
],
"lang": {
"en-us": {
"role": {
"documentation": "Preferred stock that may be exchanged into common shares or other types of securities at the owner's option.",
"label": "Convertible Preferred Stock [Member]",
"terseLabel": "Convertible preferred stock (as converted to common stock)",
"verboseLabel": "Convertible Preferred Stock"
}
}
},
"localname": "ConvertiblePreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "domainItemType"
},
"us-gaap_CorporateBondSecuritiesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "This category includes information about long-term debt securities that are issued by either a domestic or foreign corporate business entity with a date certain promise of repayment and a return to the holder for the time value of money (for example, variable or fixed interest, original issue discount).",
"label": "Corporate Bond Securities [Member]",
"terseLabel": "Corporate bonds"
}
}
},
"localname": "CorporateBondSecuritiesMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CostOfRevenue": {
"auth_ref": [
"r81r31",
"r117r226",
"r221r302",
"r261r337",
"r262r338",
"r263r339",
"r265r340",
"r266r341",
"r267r342",
"r268r343",
"r269r344",
"r270r345",
"r271r529",
"r525r733"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 3.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate cost of goods produced and sold and services rendered during the reporting period.",
"label": "Cost of Revenue",
"terseLabel": "Cost of Revenuerevenue"
}
}
},
"localname": "CostOfRevenue",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CostOfSalesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing cost of sales.",
"label": "Cost of Sales [Member]",
"terseLabel": "Cost of product revenue"
}
}
},
"localname": "CostOfSalesMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CostOfSalesPolicyTextBlock": {
"auth_ref": [
"r372r740"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for cost of product sold and service rendered.",
"label": "Cost of Goods and Service [Policy Text Block]",
"terseLabel": "Cost of Product Revenue"
}
}
},
"localname": "CostOfSalesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_CostsAndExpenses": {
"auth_ref": [
"r80r30"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 1.0,
"parentTag": "us-gaap_OperatingIncomeLoss",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Total costs of sales and operating expenses for the period.",
"label": "Costs and Expenses",
"totalLabel": "Total cost and operating expenses"
}
}
},
"localname": "CostsAndExpenses",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_CostsAndExpensesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Costs and Expenses [Abstract]",
"terseLabel": "Cost and operating expenses"
}
}
},
"localname": "CostsAndExpensesAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "stringItemType"
},
"us-gaap_CreditFacilityAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of credit facility. Credit facilities provide capital to borrowers without the need to structure a loan for each borrowing.",
"label": "Credit Facility [Axis]",
"terseLabel": "Credit Facility [Axis]"
}
}
},
"localname": "CreditFacilityAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_CreditFacilityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Type of credit facility. Credit facilities provide capital to borrowers without the need to structure a loan for each borrowing.",
"label": "Credit Facility [Domain]",
"terseLabel": "Credit Facility [Domain]"
}
}
},
"localname": "CreditFacilityDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_CustomerConcentrationRiskMember": {
"auth_ref": [
"r176r57",
"r206r294"
],
"lang": {
"en-us": {
"role": {
"documentation": "Reflects the percentage that revenues in the period from one or more significant customers is to net revenues, as defined by the entity, such as total net revenues, product line revenues, segment revenues. The risk is the materially adverse effects of loss of a significant customer.",
"label": "Customer Concentration Risk [Member]",
"terseLabel": "Customer Concentration Risk"
}
}
},
"localname": "CustomerConcentrationRiskMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DebtDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Debt Disclosure [Abstract]"
}
}
},
"localname": "DebtDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_DebtDisclosureTextBlock": {
"auth_ref": [
"r113r84",
"r279r224",
"r280r351",
"r281r352",
"r282r353",
"r283r354",
"r284r355",
"r285r356",
"r290r357",
"r297r362",
"r298r369",
"r300r370",
"r310r372"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for information about short-term and long-term debt arrangements, which includes amounts of borrowings under each line of credit, note payable, commercial paper issue, bonds indenture, debenture issue, own-share lending arrangements and any other contractual agreement to repay funds, and about the underlying arrangements, rationale for a classification as long-term, including repayment terms, interest rates, collateral provided, restrictions on use of assets and activities, whether or not in compliance with debt covenants, and other matters important to users of the financial statements, such as the effects of refinancing and noncompliance with debt covenants.",
"label": "Debt Disclosure [Text Block]",
"terseLabel": "INDEBTEDNESS"
}
}
},
"localname": "DebtDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESS"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DebtInstrumentAxis": {
"auth_ref": [
"r20",
"r22",
"r23",
"r116",
"r123",
"r273",
"r274",
"r275",
"r276",
"r277",
"r278",
"r280",
"r286",
"r287",
"r288",
"r289",
"r291",
"r292",
"r293",
"r294",
"r295",
"r296",
"r304",
"r305",
"r306",
"r307",
"r545",
"r624",
"r625",
"r641"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of debt instrument, including, but not limited to, draws against credit facilities.",
"label": "Debt Instrument [Axis]",
"terseLabel": "Debt Instrument [Axis]"
}
}
},
"localname": "DebtInstrumentAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DebtInstrumentBasisSpreadOnVariableRate1": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Percentage points added to the reference rate to compute the variable rate on the debt instrument.",
"label": "Debt Instrument, Basis Spread on Variable Rate",
"terseLabel": "Variable rate"
}
}
},
"localname": "DebtInstrumentBasisSpreadOnVariableRate1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_DebtInstrumentCarryingAmount": {
"auth_ref": [
"r23r1",
"r301r155",
"r625r167",
"r641r373"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails": {
"order": 1.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before unamortized (discount) premium and debt issuance costs, of long-term debt. Includes, but is not limited to, notes payable, bonds payable, commercial loans, mortgage loans, convertible debt, subordinated debt and other types of debt.",
"label": "Long-termTerm Debt, Gross",
"terseLabel": "Principal"
}
}
},
"localname": "DebtInstrumentCarryingAmount",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DebtInstrumentInterestRateDuringPeriodgaap_DebtInstrumentFeeAmount": {
"auth_ref": [
"r49",
"r293",
"r543r17"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of the fee that accompanies borrowing money under the debt instrument.",
"label": "Debt Instrument, Fee Amount",
"terseLabel": "Prepayment penalty"
}
}
},
"localname": "DebtInstrumentFeeAmount",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DebtInstrumentInterestRateStatedPercentage": {
"auth_ref": [
"r16",
"r347"
],
"lang": {
"en-us": {
"role": {
"documentation": "The average effectiveContractual interest rate duringfor funds borrowed, under the reporting perioddebt agreement.",
"label": "Debt Instrument, Interest Rate, During PeriodStated Percentage",
"terseLabel": "Interest rate"
}
}
},
"localname": "DebtInstrumentInterestRateDuringPeriodDebtInstrumentInterestRateStatedPercentage",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_DebtInstrumentLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Debt Instrument [Line Items]",
"terseLabel": "Debt Instrument [Line Items]"
}
}
},
"localname": "DebtInstrumentLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetailsINDEBTEDNESSMinimumRevenueCovenantDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetailsINDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DebtInstrumentMeasurementInput": {
"auth_ref": [
"r514r526"
],
"lang": {
"en-us": {
"role": {
"documentation": "Value of input used to measure debt instrument, including, but not limited to, convertible and non-convertible debt.",
"label": "Debt Instrument, Measurement Input",
"terseLabel": "Debt measurement input"
}
}
},
"localname": "DebtInstrumentMeasurementInput",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails"
],
"xbrltype": "decimalItemType"
},
"us-gaap_DebtInstrumentNameDomain": {
"auth_ref": [
"r50",
"r116",
"r123",
"r273",
"r274",
"r275",
"r276",
"r277",
"r278",
"r280",
"r286",
"r287",
"r288",
"r289",
"r291",
"r292",
"r293",
"r294",
"r295",
"r296",
"r304",
"r305",
"r306",
"r307",
"r545"
],
"lang": {
"en-us": {
"role": {
"documentation": "The name for the particular debt instrument or borrowing that distinguishes it from other debt instruments or borrowings, including draws against credit facilities.",
"label": "Debt Instrument, Name [Domain]",
"terseLabel": "Debt Instrument, Name [Domain]"
}
}
},
"localname": "DebtInstrumentNameDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DebtInstrumentTable": {
"auth_ref": [
"r50gaap_DebtInstrumentTable": {
"auth_ref": [
"r18",
"r116r87",
"r123r88",
"r273r89",
"r274r90",
"r275r149",
"r276r150",
"r277r152",
"r278r165",
"r280r232",
"r286r346",
"r287r347",
"r288r348",
"r289r349",
"r291r350",
"r292r352",
"r293r358",
"r294r359",
"r295r360",
"r296r361",
"r299r363",
"r304r364",
"r305r365",
"r306r366",
"r307r367",
"r334r368",
"r337r371",
"r338r547",
"r339r661",
"r542r662",
"r543r663",
"r545r664",
"r546r665",
"r639r713"
],
"lang": {
"en-us": {
"role": {
"documentation": "A table or schedule providing information pertaining to long-term debt instruments or arrangements, including identification, terms, features, collateral requirements and other information necessary to a fair presentation. These are debt arrangements that originally required repayment more than twelve months after issuance or greater than the normal operating cycle of the company, if longer.",
"label": "Schedule of Long-termTerm Debt Instruments [Table]",
"terseLabel": "Schedule of Long-term Debt Instruments [Table]"
}
}
},
"localname": "DebtInstrumentTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetailsINDEBTEDNESSMinimumRevenueCovenantDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetailsINDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_DebtInstrumentUnamortizedDiscountPremiumAndDebtIssuanceCostsNet": {
"auth_ref": [
"r286r151",
"r302r358",
"r304r374",
"r305r662",
"r544r663"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails": {
"order": 2.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of unamortized debt discount (premium) and debt issuance costs.",
"label": "Debt Instrument, Unamortized Discount (Premium) and Debt Issuance Costs, Net",
"negatedTerseLabel": "Less: Debt issuance costs and discount at issuance"
}
}
},
"localname": "DebtInstrumentUnamortizedDiscountPremiumAndDebtIssuanceCostsNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredChargesPolicyTextBlock": {
"auth_ref": [
"r42"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for deferral and amortization of significant deferred charges.",
"label": "Deferred Charges, Policy [Policy Text Block]",
"terseLabel": "Deferred Offering Costs"
}
}
},
"localname": "DeferredChargesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DeferredFinanceCostsGross": {
"auth_ref": [
"r544gaap_DeferredFinanceCostsGross": {
"auth_ref": [
"r151"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before accumulated amortization, of debt issuance costs. Includes, but is not limited to, legal, accounting, underwriting, printing, and registration costs.",
"label": "Debt Issuance Costs, Gross",
"terseLabel": "Debt fees and expenses"
}
}
},
"localname": "DeferredFinanceCostsGross",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredFinanceCostsNet": {
"auth_ref": [
"r42r151",
"r286",
"r544r736"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after accumulated amortization, of debt issuance costs. Includes, but is not limited to, legal, accounting, underwriting, printing, and registration costs.",
"label": "Debt Issuance Costs, Net",
"terseLabel": "Issuance costs"
}
}
},
"localname": "DeferredFinanceCostsNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredRentCredit": {
"auth_ref": [
"r548r181",
"r556r783"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of excess of rental payment required by lease over rental income recognized.",
"label": "Deferred Rent Credit",
"terseLabel": "Deferred rent"
}
}
},
"localname": "DeferredRentCredit",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetDomain": {
"auth_ref": [
"r444r113"
],
"lang": {
"en-us": {
"role": {
"documentation": "Identification of the deferred tax asset for which a valuation reserve exists.",
"label": "Deferred Tax Asset [Domain]",
"terseLabel": "Deferred Tax Asset [Domain]"
}
}
},
"localname": "DeferredTaxAssetDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_DeferredTaxAssetsDeferredIncome": {
"auth_ref": [
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 47.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences from deferred income.",
"label": "Deferred Tax Assets, Deferred Income",
"terseLabel": "Deferred revenues"
}
}
},
"localname": "DeferredTaxAssetsDeferredIncome",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsGross": {
"auth_ref": [
"r443r480"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 25.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences and carryforwards.",
"label": "Deferred Tax Assets, Gross",
"totalLabel": "Total deferred tax assets"
}
}
},
"localname": "DeferredTaxAssetsGross",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsNet": {
"auth_ref": [
"r445r770"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences and carryforwards.",
"label": "Deferred Tax Assets, Net of Valuation Allowance",
"totalLabel": "Net deferred tax assets"
}
}
},
"localname": "DeferredTaxAssetsNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsOperatingLossCarryforwards": {
"auth_ref": [
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 74.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible operating loss carryforwards.",
"label": "Deferred Tax Assets, Operating Loss Carryforwards",
"terseLabel": "Net operating loss carryforwards"
}
}
},
"localname": "DeferredTaxAssetsOperatingLossCarryforwards",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsOther": {
"auth_ref": [
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 39.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, before allocation of valuation allowance, of deferred tax asset attributable to deductible temporary differences, classified as other.",
"label": "Deferred Tax Assets, Other",
"terseLabel": "Other"
}
}
},
"localname": "DeferredTaxAssetsOther",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsPropertyPlantAndEquipment": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 3.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences from property, plant, and equipment.",
"label": "Deferred Tax Assets, Property, Plant and Equipment",
"terseLabel": "Depreciation"
}
}
},
"localname": "DeferredTaxAssetsPropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsTaxCreditCarryforwardsResearch": {
"auth_ref": [
"r448r115",
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 61.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible research tax credit carryforwards.",
"label": "Deferred Tax Assets, Tax Credit Carryforwards, Research",
"terseLabel": "Research and developmentR&D credit carryforwards"
}
}
},
"localname": "DeferredTaxAssetsTaxCreditCarryforwardsResearch",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsTaxDeferredExpenseCompensationAndBenefitsShareBasedCompensationCost": {
"auth_ref": [
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 58.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences from share-based compensation.",
"label": "Deferred Tax Assets, Tax Deferred Expense, Compensation and Benefits, Share-basedBased Compensation Cost",
"terseLabel": "Stock-based compensation"
}
}
},
"localname": "DeferredTaxAssetsTaxDeferredExpenseCompensationAndBenefitsShareBasedCompensationCost",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsTaxDeferredExpenseReservesAndAccruals": {
"auth_ref": [
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 15.0,
"parentTag": "us-gaap_DeferredTaxAssetsGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences from reserves and accruals.",
"label": "Deferred Tax Assets, Tax Deferred Expense, Reserves and Accruals",
"terseLabel": "Accrued expenses and other"
}
}
},
"localname": "DeferredTaxAssetsTaxDeferredExpenseReservesAndAccruals",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxAssetsValuationAllowance": {
"auth_ref": [
"r444r481"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 12.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax assets for which it is more likely than not that a tax benefit will not be realized.",
"label": "Deferred Tax Assets, Valuation Allowance",
"negatedTerseLabel": "Less: valuation allowance"
}
}
},
"localname": "DeferredTaxAssetsValuationAllowance",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilitiesGoodwillAndIntangibleAssetsIntangibleAssets": {
"auth_ref": [
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 21.0,
"parentTag": "us-gaap_DeferredTaxAssetsGrossgaap_DeferredTaxAssetsNet",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax liability attributable to taxable temporary differences from intangible assets other than goodwill.",
"label": "Deferred Tax Liabilities, Intangible Assets",
"negatedTerseLabel": "Amortization"
}
}
},
"localname": "DeferredTaxLiabilitiesGoodwillAndIntangibleAssetsIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilitiesLeasingArrangements": {
"auth_ref": [
"r117",
"r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 4.0,
"parentTag": "us-gaap_DeferredTaxAssetsNet",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax liability attributable to taxable temporary differences from leasing arrangements.",
"label": "Deferred Tax Liabilities, Leasing Arrangements",
"negatedTerseLabel": "Lease right-of-use assets"
}
}
},
"localname": "DeferredTaxLiabilitiesLeasingArrangements",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DeferredTaxLiabilitiesPropertyPlantAndEquipment": {
"auth_ref": [
"r450r117",
"r451r771"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails": {
"order": 83.0,
"parentTag": "us-gaap_DeferredTaxAssetsGrossgaap_DeferredTaxAssetsNet",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of deferred tax liability attributable to taxable temporary differences from property, plant, and equipment.",
"label": "Deferred Tax Liabilities, Property, Plant and Equipment",
"negatedTerseLabel": "Depreciation"
}
}
},
"localname": "DeferredTaxLiabilitiesPropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESDeferredTaxAssetsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_Depreciation": {
"auth_ref": [
"r101r45",
"r242r71"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 11.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of expense recognized in the current period that reflects the allocation of the cost of tangible assets over the assets' useful lives. Includes production and non-production related depreciation.",
"label": "Depreciation",
"terseLabel": "Depreciation expense"
}
}
},
"localname": "Depreciation",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSPROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_DerivativesPolicyTextBlock": {
"auth_ref": [
"r122r131",
"r490r132",
"r491r133",
"r492r134",
"r493r138",
"r497r231"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for its derivative instruments and hedging activities.",
"label": "Derivatives, Policy [Policy Text Block]",
"terseLabel": "Derivative Liabilities"
}
}
},
"localname": "DerivativesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock": {
"auth_ref": [
"r429r432",
"r463",
"r464",
"r466",
"r471",
"r676"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for share-based payment arrangement.",
"label": "Share-basedBased Payment Arrangement [Text Block]",
"terseLabel": "STOCK-BASED COMPENSATION"
}
}
},
"localname": "DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATION"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_DisclosureOfCompensationRelatedCostsSharebasedPaymentsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Share-basedBased Payment Arrangement [Abstract]"
}
}
},
"localname": "DisclosureOfCompensationRelatedCostsSharebasedPaymentsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_DomesticCountryMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Designated tax department of the government that is entitled to levy and collect income taxes from the entity in its country of domicile.",
"label": "Domestic Tax Authority [Member]",
"terseLabel": "Federal"
}
}
},
"localname": "DomesticCountryMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_EarningsPerShareAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Earnings Per Share [Abstract]",
"terseLabel": "Net loss per share:"
}
}
},
"localname": "EarningsPerShareAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "stringItemType"
},
"us-gaap_EarningsPerShareBasic": {
"auth_ref": [
"r79r216",
"r134r241",
"r135r242",
"r137r243",
"r138r244",
"r139r245",
"r145r250",
"r148r253",
"r157r257",
"r158r258",
"r159r259",
"r163r263",
"r164r521",
"r509r522",
"r510r611",
"r635r615",
"r655r657"
],
"lang": {
"en-us": {
"role": {
"documentation": "The amount of net income (loss) for the period per each share of common stock or unit outstanding during the reporting period.",
"label": "Earnings Per Share, Basic",
"terseLabel": "Net loss per share attributable to common stockholders, basic (in dollars per share)",
"verboseLabel": "Basic (in dollars per share)"
}
}
},
"localname": "EarningsPerShareBasic",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSNETLOSSPERSHAREBasicandDilutedPerShareDetails",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "perShareItemType"
},
"us-gaap_EarningsPerShareDiluted": {
"auth_ref": [
"r79r216",
"r134r241",
"r135r242",
"r137r243",
"r138r244",
"r139r245",
"r148r253",
"r157r257",
"r158r258",
"r159r259",
"r163r263",
"r164r521",
"r509r522",
"r510r611",
"r635r615",
"r655r657"
],
"lang": {
"en-us": {
"role": {
"documentation": "The amount of net income (loss) for the period available to each share of common stock or common unit outstanding during the reporting period and to each share or unit that would have been outstanding assuming the issuance of common shares or units for all dilutive potential common shares or units outstanding during the reporting period.",
"label": "Earnings Per Share, Diluted",
"terseLabel": "Net loss per share attributable to common stockholders, diluted (in dollars per share)",
"verboseLabel": "Diluted (in dollars per share)"
}
}
},
"localname": "EarningsPerShareDiluted",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSNETLOSSPERSHAREBasicandDilutedPerShareDetails",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "perShareItemType"
},
"us-gaap_EarningsPerSharePolicyTextBlock": {
"auth_ref": [
"r160r55",
"r161r56"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for computing basic and diluted earnings or loss per share for each class of common stock and participating security. Addresses all significant policy factors, including any antidilutive items that have been excluded from the computation and takes into account stock dividends, splits and reverse splits that occur after the balance sheet date of the latest reporting period but before the issuance of the financial statements.",
"label": "Earnings Per Share, Policy [Policy Text Block]",
"verboseLabelterseLabel": "Net Loss Per Share"
}
}
},
"localname": "EarningsPerSharePolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_EarningsPerShareTextBlock": {
"auth_ref": [
"r160r260",
"r161r261",
"r162r262",
"r165r264"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for earnings per share.",
"label": "Earnings Per Share [Text Block]",
"terseLabel": "NET LOSS PER SHARE"
}
}
},
"localname": "EarningsPerShareTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHARE"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_EffectiveIncomeTaxRateContinuingOperations": {
"auth_ref": [
"r438r476"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of current income tax expense (benefit) and deferred income tax expense (benefit) pertaining to continuing operations.",
"label": "Effective Income Tax Rate Reconciliation, Percent",
"totalLabel": "Effective income tax rate"
}
}
},
"localname": "EffectiveIncomeTaxRateContinuingOperations",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate": {
"auth_ref": [
"r118r227",
"r438r476",
"r459r492"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 73.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of domestic federal statutory tax rate applicable to pretax income (loss).",
"label": "Effective Income Tax Rate Reconciliation, at Federal Statutory Income Tax Rate, Percent",
"terseLabel": "Federal statutory income tax rate"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationAtFederalStatutoryIncomeTaxRate",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationChangeInDeferredTaxAssetsValuationAllowance": {
"auth_ref": [
"r438r769",
"r459r772"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 54.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to changes in the valuation allowance for deferred tax assets.",
"label": "Effective Income Tax Rate Reconciliation, Change in Deferred Tax Assets Valuation Allowance, Percent",
"terseLabel": "Change in deferred tax asset valuation allowance"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationChangeInDeferredTaxAssetsValuationAllowance",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationNondeductibleExpense": {
"auth_ref": [
"r438r769",
"r459r772"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 6.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to nondeductible expenses.",
"label": "Effective Income Tax Rate Reconciliation, Nondeductible Expense, Percent",
"terseLabel": "Permanent differences"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationNondeductibleExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationOtherReconcilingItemsPercent": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 41.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference, between reported income tax expense (benefit) and the expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations, that is attributable to tax exempt income, equity in earnings (loss) of an unconsolidated subsidiary, minority interest income (expense), tax holiday, disposition of a business, disposition of an asset, repatriation of foreign earnings, repatriation of foreign earnings jobs creation act of 2004, change in enacted tax rate, prior year income taxes, change in deferred tax asset valuation allowance, and other adjustments.",
"label": "Effective Income Tax Rate Reconciliation, Other Reconciling Items, Percent",
"terseLabel": "Other"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationOtherReconcilingItemsPercent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationStateAndLocalIncomeTaxes": {
"auth_ref": [
"r438r769",
"r459r772"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 17.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations applicable to state and local income tax expense (benefit), net of federal tax expense (benefit).",
"label": "Effective Income Tax Rate Reconciliation, State and Local Income Taxes, Percent",
"terseLabel": "State income taxes, net of federal benefit"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationStateAndLocalIncomeTaxes",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EffectiveIncomeTaxRateReconciliationTaxCreditsResearch": {
"auth_ref": [
"r438r769",
"r459r772"
],
"calculation": {
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails": {
"order": 38.0,
"parentTag": "us-gaap_EffectiveIncomeTaxRateContinuingOperations",
"weight": -1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Percentage of the difference between reported income tax expense (benefit) and expected income tax expense (benefit) computed by applying the domestic federal statutory income tax rates to pretax income (loss) from continuing operations attributable to research tax credit.",
"label": "Effective Income Tax Rate Reconciliation, Tax Credit, Research, Percent",
"negatedTerseLabel": "Federal and state research and development tax credits"
}
}
},
"localname": "EffectiveIncomeTaxRateReconciliationTaxCreditsResearch",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESReconciliationofTaxRateDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_EmbeddedDerivativeFairValueOfEmbeddedDerivativeLiability": {
"auth_ref": [
"r495r136"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION": {
"order": 2.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails": {
"order": 1.0,
"parentTag": "us-gaap_FinancialLiabilitiesFairValueDisclosure",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 4.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value as of the balance sheet date of the embedded derivative or group of embedded derivatives classified as a liability.",
"label": "Embedded Derivative, Fair Value of Embedded Derivative Liability",
"terseLabel": "Embedded debt derivative"
}
}
},
"localname": "EmbeddedDerivativeFairValueOfEmbeddedDerivativeLiability",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetailsINDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetailsUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmbeddedDerivativeGainLossOnEmbeddedDerivativeNet": {
"auth_ref": [
"r498"
]r774"
],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 18.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Net Increase or Decrease in the fair value of the embedded derivative or group of embedded derivatives included in earnings in the period.",
"label": "Embedded Derivative, Gain (Loss) on Embedded Derivative, Net",
"terseLabelnegatedTerseLabel": "Embedded debtChange in fair value of embedded derivative"
}
}
},
"localname": "EmbeddedDerivativeGainLossOnEmbeddedDerivativeNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmbeddedDerivativeGainOnEmbeddedDerivative": {
"auth_ref": [
"r774"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Increase in the fair value of the embedded derivative or group of embedded derivatives included in earnings in the period.",
"label": "Embedded Derivative, Gain on Embedded Derivative",
"terseLabel": "Gain on embedded derivative"
}
}
},
"localname": "EmbeddedDerivativeGainOnEmbeddedDerivative",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeRelatedLiabilitiesCurrent": {
"auth_ref": [
"r47r14"
],
"calculation": {
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails": {
"order": 42.0,
"parentTag": "us-gaap_AccountsPayableAndOtherAccruedLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Total of the carrying values as of the balance sheet date of obligations incurred through that date and payable for obligations related to services received from employees, such as accrued salaries and bonuses, payroll taxes and fringe benefits. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer).",
"label": "Employee-related Liabilities, Current",
"terseLabel": "Compensation and related benefits"
}
}
},
"localname": "EmployeeRelatedLiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Share-basedBased Payment Arrangement, Expensed and Capitalized, Amount [Line Items]",
"terseLabel": "Share-based Payment Arrangement, Expensed and Capitalized, Amount [Line Items]"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedPeriodForRecognition1": {
"auth_ref": [
"r414r465"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted-average period over which cost not yet recognized is expected to be recognized for award under share-based payment arrangement, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Share-basedBased Payment Arrangement, Nonvested Award, Cost Not yet Recognized, Period for Recognition",
"terseLabel": "Unrecognized compensation costs, period of recognition"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedPeriodForRecognition1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedShareBasedAwardsOtherThanOptions": {
"auth_ref": [
"r768"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cost to be recognized for nonvested award under share-based payment arrangement. Excludes share and unit options.",
"label": "Share-Based Payment Arrangement, Nonvested Award, Excluding Option, Cost Not yet Recognized, Amount",
"terseLabel": "Unrecognized compensation costs"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedShareBasedAwardsOtherThanOptions",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedStockOptions": {
"auth_ref": [
"r414r768"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cost to be recognized for option under share-based payment arrangement.",
"label": "Share-basedBased Payment Arrangement, Nonvested Award, Option, Cost Not yet Recognized, Amount",
"terseLabel": "Unrecognized compensation costs"
}
}
},
"localname": "EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedStockOptions",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_EmployeeStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "An Employee Stock Purchase Plan is a tax-efficient means by which employees of a corporation can purchase the corporation's stock.",
"label": "Employee Stock [Member]",
"terseLabel": "Employee StockESPP"
}
}
},
"localname": "EmployeeStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_EmployeeStockOptionMember": {
"auth_ref": [
"r411"
],
"lang": {
"en-us": {
"role": {
"documentation": "Share-based payment arrangement granting right, subject to vesting and other restrictions, to purchase or sell certain number of shares at predetermined price for specified period of time.",
"label": "Share-basedBased Payment Arrangement, Option [Member]",
"terseLabel": "Outstanding common stock options"
}
}
},
"localname": "EmployeeStockOptionMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_EquipmentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tangible personal property used to produce goods and services.",
"label": "Equipment [Member]",
"terseLabel": "Equipment"
}
}
},
"localname": "EquipmentMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_EquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Equity [Abstract]"
}
}
},
"localname": "EquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_EquityComponentDomain": {
"auth_ref": [
"r2r86",
"r74r186",
"r75r212",
"r76r213",
"r125r214",
"r126r233",
"r127r234",
"r130r235",
"r140r238",
"r143r246",
"r166r248",
"r225r265",
"r333r306",
"r340r394",
"r421r467",
"r422r468",
"r423r469",
"r455r488",
"r456r489",
"r508",
"r535",
"r536",
"r537r520",
"r538",
"r539",
"r540",
"r665r541",
"r666r542",
"r667r543",
"r717r568",
"r620",
"r621",
"r622"
],
"lang": {
"en-us": {
"role": {
"documentation": "Components of equity are the parts of the total Equity balance including that which is allocated to common, preferred, treasury stock, retained earnings, etc.",
"label": "Equity Component [Domain]",
"terseLabel": "Equity Component [Domain]"
}
}
},
"localname": "EquityComponentDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",2022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParenthetical"
],
"xbrltype": "domainItemType"
},
"us-gaap_ExtinguishmentOfDebtAmount": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Gross amount of debt extinguished.",
"label": "Extinguishment of Debt, Amount",
"terseLabel": "Extinguishment of debt"
}
}
},
"localname": "ExtinguishmentOfDebtAmount",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "monetaryItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueAdjustmentOfWarrants": {
"auth_ref": [
"r101r45",
"r311r85"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 10.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS": {
"order": 5UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 2.0,
"parentTag": "us-gaap_OtherNonoperatingIncomeExpense",
"weight": -1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expense (income) related to adjustment to fair value of warrant liability.",
"label": "Fair Value Adjustment of Warrants",
"negatedTerseLabel": "Change in estimated fair value of warrant liabilities",
"verboseLabel": "Change in fair value of warrants"
}
}
},
"localname": "FairValueAdjustmentOfWarrants",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Fair Value Measurement Inputs and Valuation Techniques [Line Items]",
"terseLabel": "Fair Value Measurement Inputs and Valuation Techniques [Line Items]"
}
}
},
"localname": "FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTable": {
"auth_ref": [
"r513r140"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about input and valuation technique used to measure fair value and change in valuation approach and technique for each separate class of asset and liability measured on recurring and nonrecurring basis.",
"label": "Fair Value Measurement Inputs and Valuation Techniques [Table]",
"terseLabel": "Fair Value Measurement Inputs and Valuation Techniques [Table]"
}
}
},
"localname": "FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock": {
"auth_ref": [
"r513r140"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of input and valuation technique used to measure fair value and change in valuation approach and technique for each separate class of asset and liability measured on recurring and nonrecurring basis.",
"label": "Fair Value Measurement Inputs and Valuation Techniques [Table Text Block]",
"terseLabel": "Schedule of Valuation Assumptions"
}
}
},
"localname": "FairValueAssetsAndLiabilitiesMeasuredOnRecurringAndNonrecurringBasisValuationTechniquesTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueBalanceSheetGroupingFinancialStatementCaptionsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Fair Value, Balance Sheet Grouping, Financial Statement Captions [Line Items]",
"terseLabel": "Fair Value, Balance Sheet Grouping, Financial Statement Captions [Line Items]"
}
}
},
"localname": "FairValueBalanceSheetGroupingFinancialStatementCaptionsLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueByBalanceSheetGroupingTable": {
"auth_ref": [
"r511r139",
"r520r144",
"r521r145"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about the fair value of financial instruments, including financial assets and financial liabilities, and the measurements of those instruments, assets, and liabilities.",
"label": "Fair Value, by Balance Sheet Grouping [Table]",
"terseLabel": "Fair Value, by Balance Sheet Grouping [Table]"
}
}
},
"localname": "FairValueByBalanceSheetGroupingTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueByBalanceSheetGroupingTextBlock": {
"auth_ref": [
"r511r139",
"r520r144"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the fair value of financial instruments, including financial assets and financial liabilities, and the measurements of those instruments, assets, and liabilities.",
"label": "Fair Value, by Balance Sheet Grouping [Table Text Block]",
"terseLabel": "Schedule of Assets and Liabilities at Fair Value"
}
}
},
"localname": "FairValueByBalanceSheetGroupingTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueByFairValueHierarchyLevelAxis": {
"auth_ref": [
"r288r360",
"r304r421",
"r305r422",
"r374r423",
"r375r424",
"r376r425",
"r377r426",
"r378r524",
"r379",
"r380",
"r382",
"r512r575",
"r576",
"r577",
"r578r662",
"r663",
"r670",
"r671",
"r672"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by level within fair value hierarchy and fair value measured at net asset value per share as practical expedient.",
"label": "Fair Value Hierarchy and NAV [Axis]",
"terseLabel": "Fair Value Hierarchy and NAV [Axis]"
}
}
},
"localname": "FairValueByFairValueHierarchyLevelAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueByLiabilityClassAxis": {
"auth_ref": [
"r518r142",
"r519r143"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by class of liability.",
"label": "Liability Class [Axis]",
"terseLabel": "Liability Class [Axis]"
}
}
},
"localname": "FairValueByLiabilityClassAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueDisclosuresAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Fair Value Disclosures [Abstract]"
}
}
},
"localname": "FairValueDisclosuresAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_FairValueDisclosuresTextBlock": {
"auth_ref": [
"r518r528"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for the fair value of financial instruments (as defined), including financial assets and financial liabilities (collectively, as defined), and the measurements of those instruments as well as disclosures related to the fair value of non-financial assets and liabilities. Such disclosures about the financial instruments, assets, and liabilities would include: (1) the fair value of the required items together with their carrying amounts (as appropriate); (2) for items for which it is not practicable to estimate fair value, disclosure would include: (a) information pertinent to estimating fair value (including, carrying amount, effective interest rate, and maturity, and (b) the reasons why it is not practicable to estimate fair value; (3) significant concentrations of credit risk including: (a) information about the activity, region, or economic characteristics identifying a concentration, (b) the maximum amount of loss the entity is exposed to based on the gross fair value of the related item, (c) policy for requiring collateral or other security and information as to accessing such collateral or security, and (d) the nature and brief description of such collateral or security; (4) quantitative information about market risks and how such risks are managed; (5) for items measured on both a recurring and nonrecurring basis information regarding the inputs used to develop the fair value measurement; and (6) for items presented in the financial statement for which fair value measurement is elected: (a) information necessary to understand the reasons for the election, (b) discussion of the effect of fair value changes on earnings, (c) a description of [similar groups] items for which the election is made and the relation thereof to the balance sheet, the aggregate carrying value of items included in the balance sheet that are not eligible for the election; (7) all other required (as defined) and desired information.",
"label": "Fair Value Disclosures [Text Block]",
"terseLabel": "FAIR VALUE MEASUREMENTS"
}
}
},
"localname": "FairValueDisclosuresTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTS"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueInputsLevel1Member": {
"auth_ref": [
"r288r360",
"r374r421",
"r375r426",
"r380r524",
"r382r575",
"r512r670",
"r576r671",
"r672"
],
"lang": {
"en-us": {
"role": {
"documentation": "Quoted prices in active markets for identical assets or liabilities that the reporting entity can access at the measurement date.",
"label": "Fair Value, Inputs, Level 1 [Member]",
"terseLabel": "Level\u00a01"
}
}
},
"localname": "FairValueInputsLevel1Member",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueInputsLevel2Member": {
"auth_ref": [
"r288r360",
"r304r421",
"r305r426",
"r374r524",
"r375r576",
"r380r662",
"r382r663",
"r512r670",
"r577r671",
"r672"
],
"lang": {
"en-us": {
"role": {
"documentation": "Inputs other than quoted prices included within level 1 that are observable for an asset or liability, either directly or indirectly, including, but not limited to, quoted prices for similar assets or liabilities in active markets, or quoted prices for identical or similar assets or liabilities in inactive markets.",
"label": "Fair Value, Inputs, Level 2 [Member]",
"terseLabel": "Level\u00a02"
}
}
},
"localname": "FairValueInputsLevel2Member",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueInputsLevel3Member": {
"auth_ref": [
"r288r360",
"r304r421",
"r305r422",
"r374r423",
"r375r424",
"r376r425",
"r377r426",
"r378r524",
"r379r577",
"r380r662",
"r382r663",
"r512r670",
"r578r671",
"r672"
],
"lang": {
"en-us": {
"role": {
"documentation": "Unobservable inputs that reflect the entity's own assumption about the assumptions market participants would use in pricing.",
"label": "Fair Value, Inputs, Level 3 [Member]",
"terseLabel": "Level\u00a03"
}
}
},
"localname": "FairValueInputsLevel3Member",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationByLiabilityClassDomain": {
"auth_ref": [
"r515r141"
],
"lang": {
"en-us": {
"role": {
"documentation": "Represents classes of liabilities measured and disclosed at fair value.",
"label": "Fair Value by Liability Class [Domain]",
"terseLabel": "Fair Value by Liability Class [Domain]"
}
}
},
"localname": "FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationByLiabilityClassDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationCalculationRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation, Calculation [Roll Forward]",
"terseLabel": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation, Calculation [Roll Forward]"
}
}
},
"localname": "FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationCalculationRollForward",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation [Line Items]",
"terseLabel": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation [Line Items]"
}
}
},
"localname": "FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTable": {
"auth_ref": [
"r515r141",
"r519r143"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of information required and determined to be provided for purposes of reconciling beginning and ending balances of fair value measurements of liabilities using significant unobservable inputs (level 3). Separately presenting changes during the period, attributable to: (1) total gains or losses for the period (realized and unrealized) and location reported in the statement of income (or activities); (2) purchases, sales, issuances, and settlements (net); (3) transfers in and/or out of Level 3.",
"label": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation [Table]",
"terseLabel": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation [Table]"
}
}
},
"localname": "FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTextBlock": {
"auth_ref": [
"r515r141",
"r519r143"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the fair value measurement of liabilities using significant unobservable inputs (Level 3), a reconciliation of the beginning and ending balances, separately presenting changes attributable to the following: (1) total gains or losses for the period (realized and unrealized), segregating those gains or losses included in earnings (or changes in net assets), and gains or losses recognized in other comprehensive income (loss) and a description of where those gains or losses included in earnings (or changes in net assets) are reported in the statement of income (or activities); (2) purchases, sales, issues, and settlements (each type disclosed separately); and (3) transfers in and transfers out of Level 3 (for example, transfers due to changes in the observability of significant inputs) by class of liability.",
"label": "Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation [Table Text Block]",
"terseLabel": "Summary of Changes in the Fair Value of Level 3 Liabilities"
}
}
},
"localname": "FairValueLiabilitiesMeasuredOnRecurringBasisUnobservableInputReconciliationTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueMeasurementPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for fair value measurements of financial and non-financial assets, liabilities and instruments classified in shareholders' equity. Disclosures include, but are not limited to, how an entity that manages a group of financial assets and liabilities on the basis of its net exposure measures the fair value of those assets and liabilities.",
"label": "Fair Value Measurement, Policy [Policy Text Block]",
"terseLabel": "Fair Value Measurements"
}
}
},
"localname": "FairValueMeasurementPolicyPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityGainLossIncludedInEarnings": {
"auth_ref": [
"r516r527"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of gain (loss) recognized in income from liability measured at fair value on recurring basis using unobservable input (level 3).",
"label": "Fair Value, Measurement with Unobservable Inputs Reconciliation, Recurring Basis, Liability, Gain (Loss) Included in Earnings",
"terseLabel": "Change in fair value"
}
}
},
"localname": "FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityGainLossIncludedInEarnings",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityIssues": {
"auth_ref": [
"r517"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of issuances of financial instrument classified as a liability measured using unobservable inputs that reflect the entity's own assumption about the assumptions market participants would use in pricing.",
"label": "Fair Value, Measurement with Unobservable Inputs Reconciliation, Recurring Basis, Liability, Issuances",
"verboseLabel": "Issuance of warrants"
}
}
},
"localname": "FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilityIssues",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilitySettlements": {
"auth_ref": [
"r517"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of settlements of financial instrument classified as a liability measured using unobservable inputs that reflect the entity's own assumption about the assumptions market participants would use in pricing.",
"label": "Fair Value, Measurement with Unobservable Inputs Reconciliation, Recurring Basis, Liability, Settlements",
"negatedTerseLabel": "Exercise of warrants"
}
}
},
"localname": "FairValueMeasurementWithUnobservableInputsReconciliationRecurringBasisLiabilitySettlements",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueMeasurementWithUnobservableInputsReconciliationsRecurringBasisLiabilityValue": {
"auth_ref": [
"r515r141"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value of financial instrument classified as a liability measured using unobservable inputs that reflect the entity's own assumption about the assumptions market participants would use in pricing.",
"label": "Fair Value, Measurement with Unobservable Inputs Reconciliation, Recurring Basis, Liability Value",
"periodEndLabel": "Ending balance",
"periodStartLabel": "Beginning balance"
}
}
},
"localname": "FairValueMeasurementWithUnobservableInputsReconciliationsRecurringBasisLiabilityValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_FairValueMeasurementsFairValueHierarchyDomain": {
"auth_ref": [
"r288r360",
"r304r421",
"r305r422",
"r374r423",
"r375r424",
"r376r425",
"r377r426",
"r378",
"r379",
"r380",
"r382r575",
"r576",
"r577",
"r578r662",
"r663",
"r670",
"r671",
"r672"
],
"lang": {
"en-us": {
"role": {
"documentation": "Categories used to prioritize the inputs to valuation techniques to measure fair value.",
"label": "Fair Value Hierarchy and NAV [Domain]",
"terseLabel": "Fair Value Hierarchy and NAV [Domain]"
}
}
},
"localname": "FairValueMeasurementsFairValueHierarchyDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FinancialInstrumentAxis": {
"auth_ref": [
"r216r300",
"r217r301",
"r218r309",
"r219r310",
"r220r311",
"r228r312",
"r229r313",
"r230r315",
"r231r316",
"r232r317",
"r234r371",
"r235r390",
"r236r510",
"r237",
"r299",
"r331",
"r499r572",
"r573",
"r574",
"r575",
"r576",
"r577",
"r578",
"r579",
"r580",
"r581",
"r582",
"r583",
"r584",
"r585",
"r586",
"r587",
"r588",
"r589",
"r590",
"r591",
"r592",
"r593",
"r594",
"r595",
"r596",
"r597",
"r598",
"r599",
"r600",
"r601",
"r602r660",
"r703r724",
"r704r725",
"r705r726",
"r706r797",
"r707r798",
"r708r799",
"r709r800",
"r801",
"r802",
"r803"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of financial instrument.",
"label": "Financial Instrument [Axis]",
"terseLabel": "Financial Instrument [Axis]"
}
}
},
"localname": "FinancialInstrumentAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_FinancialLiabilitiesFairValueDisclosure": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value of financial obligations, including, but not limited to, debt instruments, derivative liabilities, federal funds purchased and sold under agreements to repurchase, securities loaned or sold under agreements to repurchase, financial instruments sold not yet purchased, guarantees, line of credit, loans and notes payable, servicing liability, and trading liabilities.",
"label": "Financial Liabilities Fair Value Disclosure",
"totalLabel": "Total liabilities"
}
}
},
"localname": "FinancialLiabilitiesFairValueDisclosure",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ForeignCountryMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Designated tax departments of governments entitled to levy and collect income taxes from the entity outside the entity's country of domicile.",
"label": "Foreign Tax Authority [Member]",
"terseLabel": "Foreign Tax Authority"
}
}
},
"localname": "ForeignCountryMember",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_FurnitureAndFixturesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Equipment commonly used in offices and stores that have no permanent connection to the structure of a building or utilities. Examples include, but are not limited to, desks, chairs, tables, and bookcases.",
"label": "Furniture and Fixtures [Member]",
"terseLabel": "Furniture and fixtures"
}
}
},
"localname": "FurnitureAndFixturesMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_GainsLossesOnExtinguishmentOfDebt": {
"auth_ref": [
"r101",
"r308",
"r309"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 13.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
},
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS": {
"order": 3.0,
"parentTag": "us-gaap_OtherNonoperatingIncomeExpense",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Difference between the fair value of payments made and the carrying amount of debt which is extinguished prior to maturity.",
"label": "Gain (Loss) on Extinguishment of Debt",
"negatedLabel": "Loss on extinguishment of debt",
"negatedTerseLabel": "Loss on extinguishment of debt",
"terseLabel": "Loss on extinguishment of debt"
}
}
},
"localname": "GainsLossesOnExtinguishmentOfDebt",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_GoodwillAndIntangibleAssetsIntangibleAssetsPolicy": {
"auth_ref": [
"r239gaap_GoodwillAndIntangibleAssetsIntangibleAssetsPolicy": {
"auth_ref": [
"r67"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for intangible assets. This accounting policy may address both intangible assets subject to amortization and those that are not. The following also may be disclosed: (1) a description of intangible assets (2) the estimated useful lives of those assets (3) the amortization method used (4) how the entity assesses and measures impairment of such assets (5) how future cash flows are estimated (6) how the fair values of such asset are determined.",
"label": "Goodwill and Intangible Assets, Intangible Assets, Policy [Policy Text Block]",
"terseLabel": "Intangible Assets"
}
}
},
"localname": "GoodwillAndIntangibleAssetsIntangibleAssetsPolicy",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_IPOMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "First sale of stock by a private company to the public.",
"label": "IPO [Member]",
"terseLabel": "IPO [Member]"
}
}
},
"localname": "IPOMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ImpairmentOrDisposalOfLongLivedAssetsPolicyTextBlock": {
"auth_ref": [
"r241CAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ImpairmentOfIntangibleAssetsExcludingGoodwill": {
"auth_ref": [
"r45",
"r69"
],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 15.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of impairment loss recognized in the period resulting from the write-down of the carrying amount of an intangible asset (excluding goodwill) to fair value.",
"label": "Impairment of Intangible Assets (Excluding Goodwill)",
"terseLabel": "Impairment of intangible asset"
}
}
},
"localname": "ImpairmentOfIntangibleAssetsExcludingGoodwill",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ImpairmentOrDisposalOfLongLivedAssetsPolicyTextBlock": {
"auth_ref": [
"r70",
"r247r75"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for recognizing and measuring the impairment of long-lived assets. An entity also may disclose its accounting policy for long-lived assets to be sold. This policy excludes goodwill and intangible assets.",
"label": "Impairment or Disposal of Long-Lived Assets, Policy [Policy Text Block]",
"terseLabel": "Impairment of Long-Lived Assets and Intangible Assets Subject to Amortization"
}
}
},
"localname": "ImpairmentOrDisposalOfLongLivedAssetsPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_IncomeStatementAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Income Statement [Abstract]"
}
}
},
"localname": "IncomeStatementAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_IncomeStatementLocationAxis": {
"auth_ref": [
"r246r318",
"r249r322"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by location in the income statement.",
"label": "Income Statement Location [Axis]",
"terseLabel": "Income Statement Location [Axis]"
}
}
},
"localname": "IncomeStatementLocationAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeStatementLocationDomain": {
"auth_ref": [
"r249r322"
],
"lang": {
"en-us": {
"role": {
"documentation": "Location in the income statement.",
"label": "Income Statement Location [Domain]",
"terseLabel": "Income Statement Location [Domain]"
}
}
},
"localname": "IncomeStatementLocationDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_IncomeTaxAuthorityAxis": {
"auth_ref": [
"r440r112"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by tax jurisdiction.",
"label": "Income Tax Authority [Axis]",
"terseLabel": "Income Tax Authority [Axis]"
}
}
},
"localname": "IncomeTaxAuthorityAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncomeTaxAuthorityDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Agency, division or body classification that levies income taxes, examines tax returns for compliance, or grants exemptions from or makes other decisions pertaining to income taxes.",
"label": "Income Tax Authority [Domain]",
"terseLabel": "Income Tax Authority [Domain]"
}
}
},
"localname": "IncomeTaxAuthorityDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_IncomeTaxDisclosureAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Income Tax Disclosure [Abstract]"
}
}
},
"localname": "IncomeTaxDisclosureAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_IncomeTaxDisclosureTextBlock": {
"auth_ref": [
"r118r227",
"r439r477",
"r441r478",
"r447r484",
"r457r490",
"r460r493",
"r462r495",
"r463r496",
"r464r497"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for income taxes. Disclosures may include net deferred tax liability or asset recognized in an enterprise's statement of financial position, net change during the year in the total valuation allowance, approximate tax effect of each type of temporary difference and carryforward that gives rise to a significant portion of deferred tax liabilities and deferred tax assets, utilization of a tax carryback, and tax uncertainties information.",
"label": "Income Tax Disclosure [Text Block]",
"terseLabel": "INCOME TAXES"
}
}
},
"localname": "IncomeTaxDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXES"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_IncomeTaxExpenseBenefit": {
"auth_ref": [
"r119r228",
"r142r247",
"r143r248",
"r185r275",
"r437r475",
"r458r491",
"r461r494",
"r656r616"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of current income tax expense (benefit) and deferred income tax expense (benefit) pertaining to continuing operations.",
"label": "Income Tax Expense (Benefit)",
"terseLabel": "Income tax expense"
}
}
},
"localname": "IncomeTaxExpenseBenefit",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncomeTaxPolicyTextBlock": {
"auth_ref": [
"r73r211",
"r435r473",
"r436r474",
"r441r478",
"r442r479",
"r446r483",
"r452r485"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for income taxes, which may include its accounting policies for recognizing and measuring deferred tax assets and liabilities and related valuation allowances, recognizing investment tax credits, operating loss carryforwards, tax credit carryforwards, and other carryforwards, methodologies for determining its effective income tax rate and the characterization of interest and penalties in the financial statements.",
"label": "Income Tax, Policy [Policy Text Block]",
"terseLabel": "Income Taxes"
}
}
},
"localname": "IncomeTaxPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_IncreaseDecreaseInAccountsPayable": {
"auth_ref": [
"r100r44"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 916.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The increase (decrease) during the reporting period in the aggregate amount of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business.",
"label": "Increase (Decrease) in Accounts Payable",
"terseLabel": "Accounts payable"
}
}
},
"localname": "IncreaseDecreaseInAccountsPayable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInAccountsReceivable": {
"auth_ref": [
"r100r44"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 24.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The increase (decrease) during the reporting period in amount due within one year (or one business cycle) from customers for the credit sale of goods and services.",
"label": "Increase (Decrease) in Accounts Receivable",
"negatedTerseLabel": "Accounts receivable"
}
}
},
"localname": "IncreaseDecreaseInAccountsReceivable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInAccruedLiabilitiesAndOtherOperatingLiabilities": {
"auth_ref": [
"r100r709"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 313.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in accrued expenses, and obligations classified as other.",
"label": "Increase (Decrease) in Accrued Liabilities and Other Operating Liabilities",
"terseLabel": "Accrued expenses, other liabilities and non-current liabilities"
}
}
},
"localname": "IncreaseDecreaseInAccruedLiabilitiesAndOtherOperatingLiabilities",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInContractWithCustomerLiability": {
"auth_ref": [
"r100r603",
"r604r709"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 147.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in obligation to transfer good or service to customer for which consideration has been received or is receivable.",
"label": "Increase (Decrease) in Contract with Customer, Liability",
"terseLabel": "Deferred revenues"
}
}
},
"localname": "IncreaseDecreaseInContractWithCustomerLiability",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInOperatingCapitalAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Increase (Decrease) in Operating Capital [Abstract]",
"terseLabel": "Changes in operating assets and liabilities:"
}
}
},
"localname": "IncreaseDecreaseInOperatingCapitalAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncreaseDecreaseInPrepaidDeferredExpenseAndOtherAssetsgaap_IncreaseDecreaseInOperatingLeaseLiability": {
"auth_ref": [
"r100r709",
"r781"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 5UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 12.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in obligation for operating lease.",
"label": "Increase (Decrease) in Operating Lease Liability",
"terseLabel": "Lease liabilities"
}
}
},
"localname": "IncreaseDecreaseInOperatingLeaseLiability",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInPrepaidDeferredExpenseAndOtherAssets": {
"auth_ref": [
"r44"
],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 17.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in prepaid expenses, and assets classified as other.",
"label": "Increase (Decrease) in Prepaid Expense and Other Assets",
"negatedTerseLabel": "Prepaid expenses and other assets"
}
}
},
"localname": "IncreaseDecreaseInPrepaidDeferredExpenseAndOtherAssets",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_IncreaseDecreaseInStockholdersEquityRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Increase (Decrease) in Stockholders' Equity [Roll Forward]",
"terseLabel": "Increase (Decrease) in Stockholders' Equity [Roll Forward]"
}
}
},
"localname": "IncreaseDecreaseInStockholdersEquityRollForward",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "stringItemType"
},
"us-gaap_IncreaseDecreaseInTemporaryEquityRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Increase (Decrease) in Temporary Equity [Roll Forward]",
"terseLabel": "Increase (Decrease) in Temporary Equity [Roll Forward]"
}
}
},
"localname": "IncreaseDecreaseInTemporaryEquityRollForward",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "stringItemType"
},
"us-gaap_InterestIncomeExpenseNet": {
"auth_ref": [
"r636r163"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 23.0,
"parentTag": "us-gaap_OtherNonoperatingIncomeExpense",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The net amount of operating interest income (expense).",
"label": "Interest Income (Expense), Net",
"terseLabel": "Interest and other (expense) income, net"
}
}
},
"localname": "InterestIncomeExpenseNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InterestPaidNet": {
"auth_ref": [
"r96r219",
"r98r221",
"r105r222"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash paid for interest, excluding capitalized interest, classified as operating activity. Includes, but is not limited to, payment to settle zero-coupon bond for accreted interest of debt discount and debt instrument with insignificant coupon interest rate in relation to effective interest rate of borrowing attributable to accreted interest of debt discount.",
"label": "Interest Paid, Excluding Capitalized Interest, Operating Activities",
"terseLabel": "Cash paid for interest"
}
}
},
"localname": "InterestPaidNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InterestPayableCurrentAndNoncurrent": {
"auth_ref": [
"r631r160",
"r652"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": 2.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
}r177"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of interest payable on debt, including, but not limited to, trade payables.",
"label": "Interest Payable",
"negatedTerseLabel": "Interest payable"
}
}
},
"localname": "InterestPayableCurrentAndNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_InvestorMembergaap_LeaseContractualTermAxis": {
"auth_ref": [
"r560",
"r561r780"
],
"lang": {
"en-us": {
"role": {
"documentation": "Business entity or individual that puts money, by purchase or expenditure, in something offering potential profitable returns, such as interest income or appreciation in valueInformation by contractual term of lease arrangement.",
"label": "Investor [MemberLease Contractual Term [Axis]",
"terseLabel": "InvestorLease Contractual Term [Axis]"
}
}
},
"localname": "InvestorMemberLeaseContractualTermAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetailsLEASEDetails"
],
"xbrltype": "domainItemTypestringItemType"
},
"us-gaap_LeaseAndRentalExpensegaap_LeaseContractualTermDomain": {
"auth_ref": [],
"crdrr780":
"debit"],
"lang": {
"en-us": {
"role": {
"documentation": "AmountContractual term of rent expense incurred for leased assets, including but not limited to, furniture and equipment, that is not directly or indirectly associated with the manufacture, sale or creation of a product or product line.",
"label": "Operating Leases, Rent Expenselease arrangement.",
"label": "Lease Contractual Term [Domain]",
"terseLabel": "Rent expenseLease Contractual Term [Domain]"
}
}
},
"localname": "LeaseAndRentalExpenseLeaseContractualTermDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetailsLEASEDetails"
],
"xbrltype": "monetaryItemTypedomainItemType"
},
"us-gaap_LeaseholdImprovementsMember": {
"auth_ref": [
"r243r72"
],
"lang": {
"en-us": {
"role": {
"documentation": "Additions or improvements to assets held under a lease arrangement.",
"label": "Leasehold Improvements [Member]",
"terseLabel": "Leasehold improvements"
}
}
},
"localname": "LeaseholdImprovementsMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LesseeLeasesPolicyTextBlock": {
"auth_ref": [
"r5542022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LeasesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Leases [Abstract]"
}
}
},
"localname": "LeasesAbstract",
"nsuri": "http://fasb.org/us-gaap/2022",
"xbrltype": "stringItemType"
},
"us-gaap_LesseeLeaseDescriptionLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Lessee, Lease, Description [Line Items]",
"terseLabel": "Lessee, Lease, Description [Line Items]"
}
}
},
"localname": "LesseeLeaseDescriptionLineItems",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LesseeLeaseDescriptionTable": {
"auth_ref": [
"r557"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about lessee's leases.",
"label": "Lessee, Lease, Description [Table]",
"terseLabel": "Lessee, Lease, Description [Table]"
}
}
},
"localname": "LesseeLeaseDescriptionTable",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LesseeLeasesPolicyTextBlock": {
"auth_ref": [
"r556"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for leasing arrangement entered into by lessee.",
"label": "Lessee, Leases [Policy Text Block]",
"terseLabel": "Leases"
}
}
},
"localname": "LesseeLeasesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_LesseeOperatingLeaseRenewalTerm": {
"auth_ref": [
"r5552022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityMaturityTableTextBlock": {
"auth_ref": [
"r782"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of undiscounted cash flows of lessee's operating lease liability. Includes, but is not limited to, reconciliation of undiscounted cash flows to operating lease liability recognized in statement of financial position.",
"label": "Lessee, Operating Lease, Liability, Maturity [Table Text Block]",
"terseLabel": "Schedule of Lease Payments"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityMaturityTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails_1": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease.",
"label": "Lessee, Operating Lease, Liability, to be Paid",
"terseLabel": "Future minimum lease payments",
"totalLabel": "Total lease payments"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDue",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails",
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueAfterYearFive": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails_1": {
"order": 6.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease due after fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, after Year Five",
"terseLabel": "2028 and thereafter"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueAfterYearFive",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails_1": {
"order": 4.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year One",
"terseLabel": "2023"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearFive": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails_1": {
"order": 5.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in fifth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Five",
"terseLabel": "2027"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearFive",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearFour": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails_1": {
"order": 2.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in fourth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Four",
"terseLabel": "2026"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearFour",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearThree": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails_1": {
"order": 3.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in third fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Three",
"terseLabel": "2025"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearThree",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearTwo": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails_1": {
"order": 1.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payment for operating lease to be paid in second fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Lessee, Operating Lease, Liability, to be Paid, Year Two",
"terseLabel": "2024"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityPaymentsDueYearTwo",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseLiabilityUndiscountedExcessAmount": {
"auth_ref": [
"r561"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails": {
"order": 2.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's undiscounted obligation for lease payments in excess of discounted obligation for lease payments for operating lease.",
"label": "Lessee, Operating Lease, Liability, Undiscounted Excess Amount",
"negatedTerseLabel": "Less: present value adjustment"
}
}
},
"localname": "LesseeOperatingLeaseLiabilityUndiscountedExcessAmount",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LesseeOperatingLeaseRenewalTerm": {
"auth_ref": [
"r779"
],
"lang": {
"en-us": {
"role": {
"documentation": "Term of lessee's operating lease renewal, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Lessee, Operating Lease, Renewal Term",
"terseLabel": "Renewal term"
}
}
},
"localname": "LesseeOperatingLeaseRenewalTerm",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_Liabilities": {
"auth_ref": [
"r46LEASEDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_LesseeOperatingLeaseTermOfContract": {
"auth_ref": [
"r779"
],
"lang": {
"en-us": {
"role": {
"documentation": "Term of lessee's operating lease, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Lessee, Operating Lease, Term of Contract",
"terseLabel": "Lease term"
}
}
},
"localname": "LesseeOperatingLeaseTermOfContract",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_LesseeOperatingLeasesTextBlock": {
"auth_ref": [
"r562"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for operating leases of lessee. Includes, but is not limited to, description of operating lease and maturity analysis of operating lease liability.",
"label": "Lessee, Operating Leases [Text Block]",
"terseLabel": "Lessee, Operating Leases"
}
}
},
"localname": "LesseeOperatingLeasesTextBlock",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASES"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_Liabilities": {
"auth_ref": [
"r13",
"r117r226",
"r196r302",
"r221r337",
"r261r338",
"r262r339",
"r263r340",
"r265r341",
"r266r342",
"r267r343",
"r268r344",
"r269r345",
"r270r507",
"r271r508",
"r483r509",
"r487r529",
"r488r658",
"r525r733",
"r566r784",
"r567r785"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 1.0,
"parentTag": "us-gaap_LiabilitiesAndStockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Sum of the carrying amounts as of the balance sheet date of all liabilities that are recognized. Liabilities are probable future sacrifices of economic benefits arising from present obligations of an entity to transfer assets or provide services to other entities in the future.",
"label": "Liabilities",
"totalLabel": "Total liabilities"
}
}
},
"localname": "Liabilities",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesAndStockholdersEquity": {
"auth_ref": [
"r35r9",
"r117r157",
"r221r174",
"r525r679",
"r568r714",
"r627r727",
"r649r777"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liabilities and equity items, including the portion of equity attributable to noncontrolling interests, if any.",
"label": "Liabilities and Equity",
"totalLabel": "Total liabilities and stockholders\u2019 equity"
}
}
},
"localname": "LiabilitiesAndStockholdersEquity",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesAndStockholdersEquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Liabilities and Equity [Abstract]",
"verboseLabel": "Liabilities and stockholders\u2019 equity"
}
}
},
"localname": "LiabilitiesAndStockholdersEquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "stringItemType"
},
"us-gaap_LiabilitiesAssumed1": {
"auth_ref": [
"r106r49",
"r107r50",
"r108r51"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The fair value of liabilities assumed in noncash investing or financing activities.",
"label": "Liabilities Assumed",
"terseLabel": "Note payable and net liabilities assumed in the Business Combination (excluding warrant liabilities)"
}
}
},
"localname": "LiabilitiesAssumed1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesCurrent": {
"auth_ref": [
"r48r15",
"r117r191",
"r221r226",
"r261r302",
"r262r337",
"r263r338",
"r265r339",
"r266r340",
"r267r341",
"r268r342",
"r269r343",
"r270r344",
"r271r345",
"r483r507",
"r487r508",
"r488r509",
"r525r529",
"r566r679",
"r567r733",
"r568r784",
"r785"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 12.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Total obligations incurred as part of normal operations that are expected to be paid during the following twelve months or within one business cycle, if longer.",
"label": "Liabilities, Current",
"totalLabel": "Total current liabilities"
}
}
},
"localname": "LiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LiabilitiesCurrentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Liabilities, Current [Abstract]",
"verboseLabel": "Current liabilities:"
}
}
},
"localname": "LiabilitiesCurrentAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "stringItemType"
},
"us-gaap_LicenseAgreementTermsMember": {
"auth_ref": [
"r433r109",
"r434r110"
],
"lang": {
"en-us": {
"role": {
"documentation": "Terms of the license agreements under research and development arrangements accounted for as a contract to perform research and development for others.",
"label": "License Agreement Terms [Member]",
"terseLabel": "License Agreement Terms"
}
}
},
"localname": "LicenseAgreementTermsMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LineOfCreditFacilityMaximumBorrowingCapacity": {
"auth_ref": [
"r44r11"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Maximum borrowing capacity under the credit facility without consideration of any current restrictions on the amount that could be borrowed or the amounts currently outstanding under the facility.",
"label": "Line of Credit Facility, Maximum Borrowing Capacity",
"terseLabel": "Maximum borrowing capacity"
}
}
},
"localname": "LineOfCreditFacilityMaximumBorrowingCapacity",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LineOfCreditMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A contractual arrangement with a lender under which borrowings can be made up to a specific amount at any point in time, and under which borrowings outstanding may be either short-term or long-term, depending upon the particulars.",
"label": "Line of Credit [Member]",
"terseLabel": "Line of Credit"
}
}
},
"localname": "LineOfCreditMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LondonInterbankOfferedRateLIBORMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Interest rate at which a bank borrows funds from other banks in the London interbank market.",
"label": "London Interbank Offered Rate (LIBOR) [Member]",
"terseLabel": "LIBOR"
}
}
},
"localname": "LondonInterbankOfferedRateLIBORMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LongTermDebt": {
"auth_ref": [
"r23r1",
"r287r155",
"r303r170",
"r304r359",
"r305r375",
"r625r662",
"r644r663"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails_1": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, afterexcluding unamortized premium (discount) premium and debt issuance costscost, of long-term debt. Includes, but not limited to, notes payable, bonds payable, debentures, mortgage loans and commercial paper. Excludes capitalExcludes lease obligationsobligation.",
"label": "Long-termTerm Debt",
"totalLabel": "Total"
}
}
},
"localname": "LongTermDebt",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtCurrent": {
"auth_ref": [
"r45r12"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 25.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": 1.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after unamortized (discount) premium and debt issuance costs, of long-term debt, classified as current. Includes, but not limited to, notes payable, bonds payable, debentures, mortgage loans and commercial paper. Excludes capital lease obligations.",
"label": "Long-termTerm Debt, Current Maturities",
"negatedLabel": "Current portion of long-term debt",
"terseLabel": "Debt"
}
}
},
"localname": "LongTermDebtCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONINDEBTEDNESSFutureMinimumPaymentsDueDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetailsUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths": {
"auth_ref": [
"r123r82",
"r258r232",
"r292r364"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails_1INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": 1.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year One",
"terseLabel": "20222023"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInNextTwelveMonths",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInYearFour": {
"auth_ref": [
"r123",
"r258",
"r292"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails_1": {
"order": 3.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in fourth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year Four",
"terseLabel": "2025"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInYearFour",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInYearThree": {
"auth_ref": [
"r123r82",
"r258r232",
"r292r364"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails_1INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": 43.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in third fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year Three",
"terseLabel": "20242025"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInYearThree",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtMaturitiesRepaymentsOfPrincipalInYearTwo": {
"auth_ref": [
"r123r82",
"r258r232",
"r292r364"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails_1INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": 2.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of long-term debt payable, sinking fund requirement, and other securities issued that are redeemable by holder at fixed or determinable price and date, maturing in second fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Long-Term Debt, Maturity, Year Two",
"terseLabel": "20232024"
}
}
},
"localname": "LongTermDebtMaturitiesRepaymentsOfPrincipalInYearTwo",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermDebtNoncurrent": {
"auth_ref": [
"r50"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": 3.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": 1.0
}
}r197"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after, excluding unamortized premium (discount) premium and debt issuance costscost, of long-term debt classified as noncurrent and excluding amounts to be repaid within one year or the normal operating cycle, if longer. Includes, but not limited to, notes payable, bonds payable, debentures, mortgage loans and commercial paper. Excludes capital . Excludes lease obligationsobligation.",
"label": "Long-termTerm Debt, Excluding Current Maturities",
"verboseLabelterseLabel": "Long-term debt"
}
}
},
"localname": "LongTermDebtNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermPurchaseCommitmentAmount": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The minimum amount the entity agreed to spend under the long-term purchase commitment.",
"label": "Long-termTerm Purchase Commitment, Amount",
"terseLabel": "Purchase commitment amount"
}
}
},
"localname": "LongTermPurchaseCommitmentAmount",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_LongTermPurchaseCommitmentByCategoryOfItemPurchasedAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by category of items purchased under a long-term purchase commitment.",
"label": "Category of Item Purchased [Axis]",
"terseLabel": "Category of Item Purchased [Axis]"
}
}
},
"localname": "LongTermPurchaseCommitmentByCategoryOfItemPurchasedAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LongTermPurchaseCommitmentCategoryOfItemPurchasedDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "General description of the goods or services to be purchased from the counterparty to the long-term purchase commitment.",
"label": "Long-termTerm Purchase Commitment, Category of Item Purchased [Domain]",
"terseLabel": "Long-term Purchase Commitment, Category of Item Purchased [Domain]"
}
}
},
"localname": "LongTermPurchaseCommitmentCategoryOfItemPurchasedDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LongtermDebtTypeAxis": {
"auth_ref": [
"r50r18"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of long-term debt.",
"label": "Long-termTerm Debt, Type [Axis]",
"terseLabel": "Long-term Debt, Type [Axis]"
}
}
},
"localname": "LongtermDebtTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_LongtermDebtTypeDomain": {
"auth_ref": [
"r50r18",
"r259r83"
],
"lang": {
"en-us": {
"role": {
"documentation": "Type of long-term debt arrangement, such as notes, line of credit, commercial paper, asset-based financing, project financing, letter of credit financing. These are debt arrangements that originally required repayment more than twelve months after issuance or greater than the normal operating cycle of the company, if longer.",
"label": "Long-termTerm Debt, Type [Domain]",
"terseLabel": "Long-term Debt, Type [Domain]"
}
}
},
"localname": "LongtermDebtTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_LongtermPurchaseCommitmentPeriod": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Period covered by the long-term purchase commitment, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Long-term Purchase Commitment, Period",
"terseLabel": "Purchase commitment period"
}
}
},
"localname": "LongtermPurchaseCommitmentPeriod",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_MarketableSecuritiesPolicy": {
"auth_ref": [
"r637INDEBTEDNESSScheduleofDebtDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MarketableSecuritiesPolicy": {
"auth_ref": [
"r164"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for investment classified as marketable security.",
"label": "Marketable Securities, Policy [Policy Text Block]",
"terseLabel": "Investments"
}
}
},
"localname": "MarketableSecuritiesPolicy",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_MeasurementInputExpectedDividendRateMember": {
"auth_ref": [
"r513r776"
],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input using expected dividend rate to be paid to holder of share per year.",
"label": "Measurement Input, Expected Dividend Rate [Member]",
"terseLabel": "Dividend yield"
}
}
},
"localname": "MeasurementInputExpectedDividendRateMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MeasurementInputExpectedTermMember": {
"auth_ref": [
"r513r776"
],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input using period financial instrument is expected to be outstanding. Excludes maturity date.",
"label": "Measurement Input, Expected Term [Member]",
"terseLabel": "Expected lifeterm (in years)"
}
}
},
"localname": "MeasurementInputExpectedTermMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MeasurementInputPriceVolatilityMember": {
"auth_ref": [
"r513r776"
],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input using rate at which price of security will increase (decrease) for given set of returns.",
"label": "Measurement Input, Price Volatility [Member]",
"terseLabel": "Expected volatility"
}
}
},
"localname": "MeasurementInputPriceVolatilityMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MeasurementInputRiskFreeInterestRateMember": {
"auth_ref": [
"r513r776"
],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input using interest rate on instrument with zero risk of financial loss.",
"label": "Measurement Input, Risk Free Interest Rate [Member]",
"terseLabel": "Risk-free interest rate"
}
}
},
"localname": "MeasurementInputRiskFreeInterestRateMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MeasurementInputSharePriceMember": {
"auth_ref": [
"r513r776"
],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input using share price of saleable stock.",
"label": "Measurement Input, Share Price [Member]",
"terseLabel": "Stock price"
}
}
},
"localname": "MeasurementInputSharePriceMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MeasurementInputTypeAxis": {
"auth_ref": [
"r513r525"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of measurement input used to determine value of asset and liability.",
"label": "Measurement Input Type [Axis]",
"terseLabel": "Measurement Input Type [Axis]"
}
}
},
"localname": "MeasurementInputTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_MeasurementInputTypeDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Measurement input used to determine value of asset and liability.",
"label": "Measurement Input Type [Domain]",
"terseLabel": "Measurement Input Type [Domain]"
}
}
},
"localname": "MeasurementInputTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_MoneyMarketFundsMember": {
"auth_ref": [
"r374gaap_MinorityInterestOwnershipPercentageByNoncontrollingOwners": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The equity interest of noncontrolling shareholders, partners or other equity holders in consolidated entity.",
"label": "Noncontrolling Interest, Ownership Percentage by Noncontrolling Owners",
"terseLabel": "Ownership percentage"
}
}
},
"localname": "MinorityInterestOwnershipPercentageByNoncontrollingOwners",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_MoneyMarketFundsMember": {
"auth_ref": [
"r741"
],
"lang": {
"en-us": {
"role": {
"documentation": "Fund that invests in short-term money-market instruments, for example, but not limited to, commercial paper, banker's acceptances, repurchase agreements, government securities, certificates of deposit, and other highly liquid securities.",
"label": "Money Market Funds [Member]",
"terseLabel": "Money market funds"
}
}
},
"localname": "MoneyMarketFundsMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_NetCashProvidedByUsedInFinancingActivities": {
"auth_ref": [
"r97r220"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 2.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseIncludingExchangeRateEffect",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow (outflow) from financing activities, including discontinued operations. Financing activity cash flows include obtaining resources from owners and providing them with a return on, and a return of, their investment; borrowing money and repaying amounts borrowed, or settling the obligation; and obtaining and paying for other resources obtained from creditors on long-term credit.",
"label": "Net Cash Provided by (Used in) Financing Activities",
"totalLabel": "Net cash provided by financing activities"
}
}
},
"localname": "NetCashProvidedByUsedInFinancingActivities",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetCashProvidedByUsedInFinancingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Net Cash Provided by (Used in) Financing Activities [Abstract]",
"terseLabel": "Financing Activities:"
}
}
},
"localname": "NetCashProvidedByUsedInFinancingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "stringItemType"
},
"us-gaap_NetCashProvidedByUsedInInvestingActivities": {
"auth_ref": [
"r97r220"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 3.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseIncludingExchangeRateEffect",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow (outflow) from investing activities, including discontinued operations. Investing activity cash flows include making and collecting loans and acquiring and disposing of debt or equity instruments and property, plant, and equipment and other productive assets.",
"label": "Net Cash Provided by (Used in) Investing Activities",
"totalLabel": "Net cash (used in) provided by investing activities"
}
}
},
"localname": "NetCashProvidedByUsedInInvestingActivities",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetCashProvidedByUsedInInvestingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Net Cash Provided by (Used in) Investing Activities [Abstract]",
"terseLabel": "Investing Activities:"
}
}
},
"localname": "NetCashProvidedByUsedInInvestingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "stringItemType"
},
"us-gaap_NetCashProvidedByUsedInOperatingActivities": {
"auth_ref": [
"r97r41",
"r99r43",
"r102r46"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 1.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseIncludingExchangeRateEffect",
"weight": 1.0
}
},
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow (outflow) from operating activities, including discontinued operations. Operating activity cash flows include transactions, adjustments, and changes in value not defined as investing or financing activities.",
"label": "Net Cash Provided by (Used in) Operating Activities",
"totalLabel": "Net cash used in operating activities"
}
}
},
"localname": "NetCashProvidedByUsedInOperatingActivities",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetCashProvidedByUsedInOperatingActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Net Cash Provided by (Used in) Operating Activities [Abstract]",
"terseLabel": "Operating Activities:"
}
}
},
"localname": "NetCashProvidedByUsedInOperatingActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "stringItemType"
},
"us-gaap_NetIncomeLoss": {
"auth_ref": [
"r6r29",
"r67r46",
"r70r162",
"r76r178",
"r77r189",
"r102r206",
"r117r209",
"r129r214",
"r134r226",
"r135r237",
"r137r241",
"r138r242",
"r142r243",
"r143r244",
"r155r247",
"r186r248",
"r194r255",
"r197r276",
"r200r284",
"r202r288",
"r221r290",
"r261r302",
"r262r337",
"r263r338",
"r265r339",
"r266r340",
"r267r341",
"r268r342",
"r269r343",
"r270r344",
"r271r345",
"r510r522",
"r525r529",
"r633r659",
"r653r733"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 123.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 2.0,
"parentTag": "us-gaap_ComprehensiveIncomeNetOfTax",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails": {
"order": 2.0,
"parentTag": "us-gaap_NetIncomeLossAvailableToCommonStockholdersBasic",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The portion of profit or loss for the period, net of income taxes, which is attributable to the parent.",
"label": "Net Income (Loss) Attributable to Parent",
"negatedLabel": "Net Income (Loss) Attributable to Parentloss",
"terseLabel": "Net loss",
"totalLabel": "Net loss"
}
}
},
"localname": "NetIncomeLoss",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS",2022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetIncomeLossAvailableToCommonStockholdersBasic": {
"auth_ref": [
"r134r241",
"r135r242",
"r137r243",
"r138r244",
"r145r250",
"r146r251",
"r156r256",
"r159r259",
"r186r276",
"r194r284",
"r197r288",
"r200r290",
"r202"
],
"calculation": {
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
}r659"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after deduction of tax, noncontrolling interests, dividends on preferred stock and participating securities; of income (loss) available to common shareholders.",
"label": "Net Income (Loss) Available to Common Stockholders, Basic",
"totalLabelterseLabel": "Net loss attributable to common shareholders"
}
}
},
"localname": "NetIncomeLossAvailableToCommonStockholdersBasic",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NetIncomeLossAvailableToCommonStockholdersDiluted": {
"auth_ref": [
"r147",
"r150",
"r151",
"r152",
"r153",
"r156",
"r159"
],
"calculation": {
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after deduction of tax, noncontrolling interests, dividends on preferred stock and participating securities, and addition from assumption of issuance of common shares for dilutive potential common shares; of income (loss) available to common shareholders.",
"label": "Net Income (Loss) Available to Common Stockholders, Diluted",
"totalLabel": "Net loss attributable to common shareholders"
}
}
},
"localname": "NetIncomeLossAvailableToCommonStockholdersDiluted",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS",2022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NewAccountingPronouncementsOrChangeInAccountingPrincipleLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "New Accounting Pronouncements or Change in Accounting Principle [Line Items]",
"terseLabel": "New Accounting Pronouncements or Change in Accounting Principle [Line Items]"
}
}
},
"localname": "NewAccountingPronouncementsOrChangeInAccountingPrincipleLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_NewAccountingPronouncementsOrChangeInAccountingPrincipleTable": {
"auth_ref": [
"r0r54",
"r1r184",
"r2r185",
"r3r186",
"r4r187",
"r128r188",
"r129r236",
"r130r237",
"r131r238",
"r132r239",
"r133r240",
"r137r243",
"r144r249",
"r163r263",
"r211r297",
"r212r298",
"r222r303",
"r223r304",
"r224r305",
"r225r306",
"r226r307",
"r227r308",
"r260r467",
"r417r468",
"r418r469",
"r419r486",
"r420r487",
"r421r488",
"r422",
"r423",
"r424",
"r453",
"r454",
"r455",
"r456r489",
"r500",
"r501",
"r502",
"r503r512",
"r504r513",
"r505r514",
"r506r515",
"r507r516",
"r508r517",
"r509r518",
"r510r519",
"r526r520",
"r527r521",
"r528r522",
"r529r523",
"r530",
"r531",
"r532",
"r533",
"r557r534",
"r535",
"r536",
"r537",
"r545",
"r546",
"r549",
"r550",
"r551",
"r552",
"r563",
"r564",
"r565",
"r566",
"r567",
"r568",
"r606",
"r607",
"r608",
"r609r618",
"r663r619",
"r664r620",
"r665r621",
"r666r622",
"r667r623",
"r668r624",
"r669r625",
"r670r626",
"r671r627",
"r672r628",
"r673",
"r674",
"r713",
"r714",
"r715",
"r716",
"r717r629"
],
"lang": {
"en-us": {
"role": {
"documentation": "Summarization of the changes in an accounting principle or a new accounting pronouncement, including the line items affected by the change and the financial effects of the change on those particular line items.",
"label": "Accounting Standards Update and Change in Accounting Principle [Table]",
"terseLabel": "Accounting Standards Update and Change in Accounting Principle [Table]"
}
}
},
"localname": "NewAccountingPronouncementsOrChangeInAccountingPrincipleTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_NewAccountingPronouncementsPolicyPolicyTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy pertaining to new accounting pronouncements that may impact the entity's financial reporting. Includes, but is not limited to, quantification of the expected or actual impact.",
"label": "New Accounting Pronouncements, Policy [Policy Text Block]",
"terseLabel": "Recently IssuedAdopted Accounting Pronouncements Not Yet Adopted"
}
}
},
"localname": "NewAccountingPronouncementsPolicyPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_NoncashOrPartNoncashAcquisitionIntangibleAssetsAcquired1": {
"auth_ref": [
"r106r49",
"r107r50",
"r108r51"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of intangibles that an Entity acquires in a noncash (or part noncash) acquisition. Noncash is defined as information about all investing and financing activities of an enterprise during a period that affect recognized assets or liabilities but that do not result in cash receipts or cash payments in the period. \"Part noncash\" refers to that portion of the transaction not resulting in cash receipts or cash payments in the period.",
"label": "Noncash or Part Noncash Acquisition, Intangible Assets Acquired",
"terseLabel": "Purchase of intangible asset with seller financing"
}
}
},
"localname": "NoncashOrPartNoncashAcquisitionIntangibleAssetsAcquired1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_NoncashOrPartNoncashAcquisitionValueOfLiabilitiesAssumed1": {
"auth_ref": [
"r106r49",
"r107r50",
"r108r51"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The total amount of [all] liabilities that an Entity assumes in acquiring a business or in consideration for an asset received in a noncash (or part noncash) acquisition. Noncash is defined as transactions during a period that affect recognized assets or liabilities but that do not result in cash receipts or cash payments in the period. \"Part noncash\" refers to that portion of the transaction not resulting in cash receipts or cash payments in the period.",
"label": "Noncash or Part Noncash Acquisition, Value of Liabilities Assumed",
"negatedTerseLabel": "Less: net liabilities assumed in the Business Combination"
}
}
},
"localname": "NoncashOrPartNoncashAcquisitionValueOfLiabilitiesAssumed1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingIncomeLoss": {
"auth_ref": [
"r186gaap_NumberOfOperatingSegments": {
"auth_ref": [
"r723"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of operating segments. An operating segment is a component of an enterprise: (a) that engages in business activities from which it may earn revenues and incur expenses (including revenues and expenses relating to transactions with other components of the same enterprise), (b) whose operating results are regularly reviewed by the enterprise's chief operating decision maker to make decisions about resources to be allocated to the segment and assess its performance, and (c) for which discrete financial information is available. An operating segment may engage in business activities for which it has yet to earn revenues, for example, start-up operations may be operating segments before earning revenues.",
"label": "Number of Operating Segments",
"terseLabel": "Number of operating segments"
}
}
},
"localname": "NumberOfOperatingSegments",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"us-gaap_OperatingIncomeLoss": {
"auth_ref": [
"r276",
"r194r284",
"r197r288",
"r200r290",
"r202r659"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 2.0,
"parentTag": "us-gaap_NetIncomeLoss",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The net result for the period of deducting operating expenses from operating revenues.",
"label": "Operating Income (Loss)",
"totalLabel": "Loss from operations"
}
}
},
"localname": "OperatingIncomeLoss",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseExpense": {
"auth_ref": [
"r778"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of operating lease expense. Excludes sublease income.",
"label": "Operating Lease, Expense",
"terseLabel": "Rent expense"
}
}
},
"localname": "OperatingLeaseExpense",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseLiability": {
"auth_ref": [
"r553"
]r554"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails": {
"order": 1.0,
"parentTag": "us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Present value of lessee's discounted obligation for lease payments from operating lease.",
"label": "Operating Lease, Liability",
"terseLabel": "Lease liabilities",
"totalLabel": "Present value of total lease liabilities"
}
}
},
"localname": "OperatingLeaseLiability",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseRightOfUseAsset": {
"auth_ref": [
"r552"
]2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseLiabilityCurrent": {
"auth_ref": [
"r554"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails": {
"order": 2.0,
"parentTag": "us-gaap_OperatingLeaseLiability",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 1.0,
"parentTag": "us-gaap_LiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Present value of lessee's discounted obligation for lease payments from operating lease, classified as current.",
"label": "Operating Lease, Liability, Current",
"negatedLabel": "Less: lease liabilities - current",
"terseLabel": "Operating lease liabilities - current"
}
}
},
"localname": "OperatingLeaseLiabilityCurrent",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseLiabilityNoncurrent": {
"auth_ref": [
"r554"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails": {
"order": 1.0,
"parentTag": "us-gaap_OperatingLeaseLiability",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 1.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Present value of lessee's discounted obligation for lease payments from operating lease, classified as noncurrent.",
"label": "Operating Lease, Liability, Noncurrent",
"terseLabel": "Operating lease liabilities - non-current",
"verboseLabel": "Lease liabilities non-current"
}
}
},
"localname": "OperatingLeaseLiabilityNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASESMaturityTableDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeasePayments": {
"auth_ref": [
"r555",
"r558"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow from operating lease, excluding payments to bring another asset to condition and location necessary for its intended use.",
"label": "Operating Lease, Payments",
"terseLabel": "Cash paid for measurement of operating lease liabilities"
}
}
},
"localname": "OperatingLeasePayments",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseRightOfUseAsset": {
"auth_ref": [
"r553"
],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 1.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of lessee's right to use underlying asset under operating lease.",
"label": "Operating Lease, Right-of-Use Asset",
"terseLabel": "ROUs",
"verboseLabel": "Lease right-of-use assets"
}
}
},
"localname": "OperatingLeaseRightOfUseAsset",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeasesFutureMinimumPaymentsDuegaap_OperatingLeaseRightOfUseAssetAmortizationExpense": {
"auth_ref": [
"r547",
"r549"
],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsr710"
],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 8.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of amortization expense for right-of-use asset from operating lease.",
"label": "Operating Lease, Right-of-Use Asset, Amortization Expense",
"terseLabel": "Amortization of right-of-use asset"
}
}
},
"localname": "OperatingLeaseRightOfUseAssetAmortizationExpense",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeaseWeightedAverageDiscountRatePercent": {
"auth_ref": [
"r560",
"r678"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average discount rate for operating lease calculated at point in time.",
"label": "Operating Lease, Weighted Average Discount Rate, Percent",
"terseLabel": "Weighted average discount rate"
}
}
},
"localname": "OperatingLeaseWeightedAverageDiscountRatePercent",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_OperatingLeaseWeightedAverageRemainingLeaseTerm1": {
"auth_ref": [
"r559",
"r678"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average remaining lease term for operating lease, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Operating Lease, Weighted Average Remaining Lease Term",
"terseLabel": "Weighted average remaining lease term"
}
}
},
"localname": "OperatingLeaseWeightedAverageRemainingLeaseTerm1",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/LEASEDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_OperatingLeasesFutureMinimumPaymentsDue": {
"auth_ref": [
"r180",
"r182"
],
"calculation": {
"http://www.peartherapeutics.com/role/LEASESOperatingLeasesDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of required minimum rental payments for leases having an initial or remaining non-cancelable letter-terms in excess of one year.",
"label": "Operating Leases, Future Minimum Payments Due",
"totalLabel": "Total lease payments"
}
}
},
"localname": "OperatingLeasesFutureMinimumPaymentsDue",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeasesFutureMinimumPaymentsDueCurrent": {
"auth_ref": [
"r547r180",
"r549r182"
],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails": {
"order": 4.0,
"parentTag": "us-gaap_OperatingLeasesFutureMinimumPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of required minimum rental payments for operating leases having an initial or remaining non-cancelable lease term in excess of one year due in the next fiscal year following the latest fiscal year. Excludes interim and annual periods when interim periods are reported on a rolling approach, from latest balance sheet date.",
"label": "Operating Leases, Future Minimum Payments Due, Next Twelve12 Months",
"terseLabel": "2022"
}
}
},
"localname": "OperatingLeasesFutureMinimumPaymentsDueCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeasesFutureMinimumPaymentsDueInFourYears": {
"auth_ref": [
"r547r180",
"r549r182"
],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails": {
"order": 3.0,
"parentTag": "us-gaap_OperatingLeasesFutureMinimumPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of required minimum rental payments for operating leases having an initial or remaining non-cancelable lease term in excess of one year due in the fourth fiscal year following the latest fiscal year. Excludes interim and annual periods when interim periods are reported on a rolling approach, from latest balance sheet date.",
"label": "Operating Leases, Future Minimum Payments, Due in Four Years",
"terseLabel": "2025"
}
}
},
"localname": "OperatingLeasesFutureMinimumPaymentsDueInFourYears",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeasesFutureMinimumPaymentsDueInThreeYears": {
"auth_ref": [
"r547r180",
"r549r182"
],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails": {
"order": 5.0,
"parentTag": "us-gaap_OperatingLeasesFutureMinimumPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of required minimum rental payments for operating leases having an initial or remaining non-cancelable lease term in excess of one year due in the third fiscal year following the latest fiscal year. Excludes interim and annual periods when interim periods are reported on a rolling approach, from latest balance sheet date.",
"label": "Operating Leases, Future Minimum Payments, Due in Three Years",
"terseLabel": "2024"
}
}
},
"localname": "OperatingLeasesFutureMinimumPaymentsDueInThreeYears",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLeasesFutureMinimumPaymentsDueInTwoYears": {
"auth_ref": [
"r547r180",
"r549r182"
],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails": {
"order": 1.0,
"parentTag": "us-gaap_OperatingLeasesFutureMinimumPaymentsDue",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of required minimum rental payments for operating leases having an initial or remaining non-cancelable lease term in excess of one year due in the second fiscal year following the latest fiscal year. Excludes interim and annual periods when interim periods are reported on a rolling approach, from latest balance sheet date.",
"label": "Operating Leases, Future Minimum Payments, Due in Two Years",
"terseLabel": "2023"
}
}
},
"localname": "OperatingLeasesFutureMinimumPaymentsDueInTwoYears",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESOperatingLeasesDetailsLEASESOperatingLeasesDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLossCarryforwards": {
"auth_ref": [
"r448r115"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of operating loss carryforward, before tax effects, available to reduce future taxable income under enacted tax laws.",
"label": "Operating Loss Carryforwards",
"terseLabel": "Net operating loss carryforwards"
}
}
},
"localname": "OperatingLossCarryforwards",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OperatingLossCarryforwardsLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Operating Loss Carryforwards [Line Items]",
"terseLabel": "Operating Loss Carryforwards [Line Items]"
}
}
},
"localname": "OperatingLossCarryforwardsLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_OperatingLossCarryforwardsTable": {
"auth_ref": [
"r449r116"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule reflecting pertinent information, such as tax authority, amounts, and expiration dates, of net operating loss carryforwards, including an assessment of the likelihood of utilization.",
"label": "Operating Loss Carryforwards [Table]",
"terseLabel": "Operating Loss Carryforwards [Table]"
}
}
},
"localname": "OperatingLossCarryforwardsTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Organization, Consolidation and Presentation of Financial Statements [Abstract]"
}
}
},
"localname": "OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsDisclosureTextBlock": {
"auth_ref": [
"r7r0",
"r489r130"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for organization, consolidation and basis of presentation of financial statements disclosure.",
"label": "Organization, Consolidation and Presentation of Financial Statements Disclosure [Text Block]",
"terseLabel": "NATURE OF BUSINESS"
}
}
},
"localname": "OrganizationConsolidationAndPresentationOfFinancialStatementsDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESS"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_OtherAssetsNoncurrent": {
"auth_ref": [
"r42r195"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 24.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of noncurrent assets classified as other.",
"label": "Other Assets, Noncurrent",
"terseLabel": "Other long-term assets"
}
}
},
"localname": "OtherAssetsNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossAvailableForSaleSecuritiesAdjustmentNetOfTax": {
"auth_ref": [
"r61r203",
"r62r204",
"r64r205"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 1.0,
"parentTag": "us-gaap_ComprehensiveIncomeNetOfTax",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, after tax and adjustment, of unrealized gain (loss) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale) and unrealized gain (loss) on investment in debt security measured at amortized cost (held-to-maturity) from transfer to available-for-sale.",
"label": "OCI, Debt Securities, Available-for-Sale, Gain (Loss), after Adjustment and Tax",
"terseLabel": "Unrealized (loss) gain on short-term investments"
}
}
},
"localname": "OtherComprehensiveIncomeLossAvailableForSaleSecuritiesAdjustmentNetOfTax",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSFAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent": {
"auth_ref": [
"r68r125",
"r71r126",
"r480r128",
"r481r207",
"r485r210"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after tax of other comprehensive income (loss) attributable to parent entity.",
"label": "Other Comprehensive Income (Loss), Net of Tax, Portion Attributable to Parent",
"terseLabel": "Other comprehensive income (loss)"
}
}
},
"localname": "OtherComprehensiveIncomeLossNetOfTaxPortionAttributableToParent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherLiabilitiesNoncurrent": {
"auth_ref": [
"r51r19"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 35.0,
"parentTag": "us-gaap_Liabilities",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of liabilities classified as other, due after one year or the normal operating cycle, if longer.",
"label": "Other Liabilities, Noncurrent",
"terseLabel": "Other long-term liabilities"
}
}
},
"localname": "OtherLiabilitiesNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherNoncurrentLiabilitiesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing other noncurrent liabilities.",
"label": "Other Noncurrent Liabilities [Member]",
"terseLabel": "Other Long-Term Liabilities"
}
}
},
"localname": "OtherNoncurrentLiabilitiesMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "domainItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherNonoperatingIncomeExpense": {
"auth_ref": [
"r85r35"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 1.0,
"parentTag": "us-gaap_NetIncomeLoss",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of income (expense) related to nonoperating activities, classified as other.",
"label": "Other Nonoperating Income (Expense)",
"totalLabel": "Total other income (expense)"
}
}
},
"localname": "OtherNonoperatingIncomeExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_OtherNonoperatingIncomeExpenseAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Other Nonoperating Income (Expense) [Abstract]",
"terseLabel": "Other income (expensesexpense):"
}
}
},
"localname": "OtherNonoperatingIncomeExpenseAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "stringItemType"
},
"us-gaap_OtherSundryLiabilitiesCurrent": {
"auth_ref": [
"r47r14",
"r256r80"
],
"calculation": {
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails": {
"order": 14.0,
"parentTag": "us-gaap_AccountsPayableAndOtherAccruedLiabilitiesCurrent",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Obligations not otherwise itemized or categorized in the footnotes to the financial statements that are due within one year or operating cycle, if longer, from the balance sheet date.",
"label": "Other Sundry Liabilities, Current",
"terseLabel": "Other"
}
}
},
"localname": "OtherSundryLiabilitiesCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PayablesAndAccrualsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Payables and Accruals [Abstract]"
}
}
},
"localname": "PayablesAndAccrualsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_PaymentsOfDebtExtinguishmentCostsgaap_PaymentsForRestructuring": {
"auth_ref": [
"r94r321",
"r708"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow forpayments made costas from early extinguishment and prepayment of debt. Includes, but is not limited to, third-party cost, premium paid, and other fee paid to lender directly for debt extinguishment or debt prepaymentthe result of exit or disposal activities. Excludes accrued interestpayments associated with a discontinued operation or an asset retirement obligation.",
"label": "Payment for Debt Extinguishment or Debt Prepayment CostPayments for Restructuring",
"terseLabelnegatedTerseLabel": "Payment for extinguishment of debtCash payments"
}
}
},
"localname": "PaymentsOfDebtExtinguishmentCostsPaymentsForRestructuring",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsOfFinancingCosts": {
"auth_ref": [
"r95"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 7.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow for loan and debt issuance costs.",
"label": "Payments of Financing Costs",
"negatedTerseLabel": "Deferred financing fees and related debt issuance costs"
}
}
},
"localname": "PaymentsOfFinancingCosts",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsOfStockIssuanceCosts": {
"auth_ref": [
"r95"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow for cost incurred directly with the issuance of an equity security.",
"label": "Payments of Stock Issuance Costs",
"terseLabel": "Stock issuance costs"
}
}
},
"localname": "PaymentsOfStockIssuanceCosts",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParenthetical"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireAvailableForSaleSecuritiesDebt": {
"auth_ref": [
"r872022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESRestructuringLiabilityRollforwardDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireAvailableForSaleSecuritiesDebt": {
"auth_ref": [
"r37",
"r89r64",
"r214r217"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 53.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash outflow to acquire investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Payments to Acquire Debt Securities, Available-for-saleSale",
"negatedTerseLabel": "Purchases of short-term investments"
}
}
},
"localname": "PaymentsToAcquireAvailableForSaleSecuritiesDebt",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquireIntangibleAssets": {
"auth_ref": [
"r90r38"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 34.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow to acquire asset without physical form usually arising from contractual or other legal rights, excluding goodwill.",
"label": "Payments to Acquire Intangible Assets",
"negatedTerseLabel": "Purchases of intangible assets"
}
}
},
"localname": "PaymentsToAcquireIntangibleAssets",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PaymentsToAcquirePropertyPlantAndEquipment": {
"auth_ref": [
"r90r38"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 25.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow associated with the acquisition of long-lived, physical assets that are used in the normal conduct of business to produce goods and services and not intended for resale; includes cash outflows to pay for construction of self-constructed assets.",
"label": "Payments to Acquire Property, Plant, and Equipment",
"negatedTerseLabel": "Purchases of property and equipment"
}
}
},
"localname": "PaymentsToAcquirePropertyPlantAndEquipment",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PlanNameAxis": {
"auth_ref": [
"r391",
"r415r742",
"r743",
"r744",
"r745",
"r746",
"r747",
"r748",
"r749",
"r750",
"r751",
"r752",
"r753",
"r754",
"r755",
"r756",
"r757",
"r758",
"r759",
"r760",
"r761",
"r762",
"r763",
"r764",
"r765",
"r766",
"r767"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by plan name for share-based payment arrangement.",
"label": "Plan Name [Axis]",
"terseLabel": "Plan Name [Axis]"
}
}
},
"localname": "PlanNameAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PlanNameDomain": {
"auth_ref": [
"r742",
"r743",
"r744",
"r745",
"r746",
"r747",
"r748",
"r749",
"r750",
"r751",
"r752",
"r753",
"r754",
"r755",
"r756",
"r757",
"r758",
"r759",
"r760",
"r761",
"r762",
"r763",
"r764",
"r765",
"r766",
"r767"
],
"lang": {
"en-us": {
"role": {
"documentation": "Plan name for share-based payment arrangement.",
"label": "Plan Name [Domain]",
"terseLabel": "Plan Name [Domain]"
}
}
},
"localname": "PlanNameDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_PreferredStockConvertibleConversionRatio": {
"auth_ref": [
"r318r378"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of common shares issuable upon conversion for each share of preferred stock to be converted.",
"label": "Preferred Stock, Convertible, Conversion Ratio",
"terseLabel": "Conversion ratio"
}
}
},
"localname": "PreferredStockConvertibleConversionRatio",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "pureItemType"
},
"us-gaap_PreferredStockDividendRatePercentage": {
"auth_ref": [
"r317"
],
"lang": {
"en-us": {
"role": {
"documentation": "The percentage rate used to calculate dividend payments on preferred stock.",
"label": "Preferred Stock, Dividend Rate, Percentage",
"terseLabel": "Dividend rate"
}
}
},
"localname": "PreferredStockDividendRatePercentage",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_PreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Preferred shares may provide a preferential dividend to the dividend on common stock and may take precedence over common stock in the event of a liquidation. Preferred shares typically represent an ownership interest in the company.",
"label": "Preferred Stock [Member]",
"terseLabel": "Preferred Stock"
}
}
},
"localname": "PreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "domainItemType"
},
"us-gaap_PreferredStockParOrStatedValuePerShare": {
"auth_ref": [
"r27r3",
"r316r376"
],
"lang": {
"en-us": {
"role": {
"documentation": "Face amount or stated value per share of preferred stock nonredeemable or redeemable solely at the option of the issuer.",
"label": "Preferred Stock, Par or Stated Value Per Share",
"terseLabel": "Preferred stock, Parpar value (in dollars per share)"
}
}
},
"localname": "PreferredStockParOrStatedValuePerShare",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "perShareItemType"
},
"us-gaap_PreferredStockSharesAuthorized": {
"auth_ref": [
"r27r3"
],
"lang": {
"en-us": {
"role": {
"documentation": "The maximum number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) permitted to be issued by an entity's charter and bylaws.",
"label": "Preferred Stock, Shares Authorized",
"terseLabel": "Preferred stock, authorized (in shares)"
}
}
},
"localname": "PreferredStockSharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "sharesItemType"
},
"us-gaap_PreferredStockSharesIssued": {
"auth_ref": [
"r27r3",
"r316r376"
],
"lang": {
"en-us": {
"role": {
"documentation": "Total number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) issued to shareholders (includes related preferred shares that were issued, repurchased, and remain in the treasury). May be all or portion of the number of preferred shares authorized. Excludes preferred shares that are classified as debt.",
"label": "Preferred Stock, Shares Issued",
"terseLabel": "Preferred stock, issued (in shares)"
}
}
},
"localname": "PreferredStockSharesIssued",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "sharesItemType"
},
"us-gaap_PreferredStockSharesOutstanding": {
"auth_ref": [
"r27r3"
],
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate share number for all nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer) held by stockholders. Does not include preferred shares that have been repurchased.",
"label": "Preferred Stock, Shares Outstanding",
"terseLabel": "Preferred stock, outstanding (in shares)"
}
}
},
"localname": "PreferredStockSharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETSParenthetical"
],
"xbrltype": "sharesItemType"
},
"us-gaap_PreferredStockValue": {
"auth_ref": [
"r27r3",
"r568r679"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 3.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate par or stated value of issued nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable preferred shares, par value and other disclosure concepts are in another section within stockholders' equity.",
"label": "Preferred Stock, Value, Issued",
"terseLabel": "Preferred stock, $0.0001 par value; 10,000,000 shares authorized as of December\u00a031, 20212022; and no shares issued and outstanding as of December\u00a031, 20212022 and December\u00a031, 20202021"
}
}
},
"localname": "PreferredStockValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PrepaidExpenseAndOtherAssetsCurrent": {
"auth_ref": [
"r10",
"r36",
"r37r702"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 32.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of asset related to consideration paid in advance for costs that provide economic benefits in future periods, and amount of other assets that are expected to be realized or consumed within one year or the normal operating cycle, if longer.",
"label": "Prepaid Expense and Other Assets, Current",
"terseLabel": "Prepaid expenses and other current assets"
}
}
},
"localname": "PrepaidExpenseAndOtherAssetsCurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PriorPeriodReclassificationAdjustmentDescription": {
"auth_ref": [
"r5"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for reclassification affecting comparability of financial statement. Excludes amendment to accounting standards, other change in accounting principle, and correction of error.",
"label": "Reclassification, Comparability Adjustment [Policy Text Block]",
"terseLabel": "Reclassification of Prior Year Presentation"
}
}
},
"localname": "PriorPeriodReclassificationAdjustmentDescription",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemTypeUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PrivatePlacementMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A private placement is a direct offering of securities to a limited number of sophisticated investors such as insurance companies, pension funds, mezzanine funds, stock funds and trusts.",
"label": "Private Placement [Member]",
"terseLabel": "Private Placement [Member]"
}
}
},
"localname": "PrivatePlacementMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsBUSINESSCOMBINATIONDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ProceedsFromIssuanceOfCommonStock": {
"auth_ref": [
"r91r39"
],
"calculation": {
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails": {
"order": 1.0,
"parentTag": "pear_ProceedsFromReverseRecapitalizationTransactionGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow from the additional capital contribution to the entity.",
"label": "Proceeds from Issuance of Common Stock",
"terseLabel": "Cash - Forward Purchase Agreement (Anchor Investor)"
}
}
},
"localname": "ProceedsFromIssuanceOfCommonStock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromIssuanceOfConvertiblePreferredStock": {
"auth_ref": [
"r91r39"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 14.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow from issuance of preferred stocks identified as being convertible into another form of financial instrument, typically the entity's common stock.",
"label": "Proceeds from Issuance of Convertible Preferred Stock",
"terseLabel": "Proceeds from issuance of Legacy Pear convertible preferred stock, net"
}
}
},
"localname": "ProceedsFromIssuanceOfConvertiblePreferredStock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromIssuanceOfDebt": {
"auth_ref": [
"r92"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 3.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
}r707"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow during the period from additional borrowings in aggregate debt. Includes proceeds from short-term and long-term debt.",
"label": "Proceeds from Issuance of Debt",
"terseLabel": "Proceeds from issuance of debt"
}
}
},
"localname": "ProceedsFromIssuanceOfDebt",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromIssuanceOfPrivatePlacement": {
"auth_ref": [
"r91r39"
],
"calculation": {
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails": {
"order": 2.0,
"parentTag": "pear_ProceedsFromReverseRecapitalizationTransactionGross",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow associated with the amount received from entity's raising of capital via private rather than public placement.",
"label": "Proceeds from Issuance of Private Placement",
"terseLabel": "Cash - PIPE Investors and Forward Purchase Assignment"
}
}
},
"localname": "ProceedsFromIssuanceOfPrivatePlacement",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsBUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromIssuanceOfWarrants": {
"auth_ref": [
"r91r39"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow from issuance of rights to purchase common shares at predetermined price (usually issued together with corporate debt).",
"label": "Proceeds from Issuance of Warrants",
"terseLabel": "Proceeds from issuance of Private Placement Warrants"
}
}
},
"localname": "ProceedsFromIssuanceOfWarrants",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromMaturitiesPrepaymentsAndCallsOfAvailableForSaleSecurities": {
"auth_ref": [
"r87r64",
"r88r217",
"r214r218"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 42.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInInvestingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow from maturity, prepayment and call of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale).",
"label": "Proceeds from Maturities, Prepayments and Calls of Debt Securities, Available-for-saleSale",
"terseLabel": "Proceeds from maturities of short-term investments"
}
}
},
"localname": "ProceedsFromMaturitiesPrepaymentsAndCallsOfAvailableForSaleSecurities",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromRoyaltiesReceived": {
"auth_ref": [
"r42"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Cash received for royalties during the current period.",
"label": "Proceeds from Royalties Received",
"terseLabel": "Proceeds from royalties"
}
}
},
"localname": "ProceedsFromRoyaltiesReceived",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromStockOptionsExercised": {
"auth_ref": [
"r91r39",
"r416r103"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 5.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash inflow from exercise of option under share-based payment arrangement.",
"label": "Proceeds from Stock Options Exercised",
"terseLabel": "Proceeds from exercise of stock options"
}
}
},
"localname": "ProceedsFromStockOptionsExercised",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProceedsFromStockPlans": {
"auth_ref": [
"r39"
],
"calculation": {
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 1.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash inflow associated with the amount received from the stock plan during the period.",
"label": "Proceeds from Stock Plans",
"terseLabel": "Proceeds from employee stock purchase plan"
}
}
},
"localname": "ProceedsFromStockPlans",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ProductMember": {
"auth_ref": [
"r368r667"
],
"lang": {
"en-us": {
"role": {
"documentation": "Article or substance produced by nature, labor or machinery.",
"label": "Product [Member]",
"terseLabel": "Product revenue"
}
}
},
"localname": "ProductMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "domainItemType"
},
"us-gaap_PropertyPlantAndEquipmentAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Property, Plant and Equipment [Abstract]"
}
}
},
"localname": "PropertyPlantAndEquipmentAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_PropertyPlantAndEquipmentByTypeAxis": {
"auth_ref": [
"r41",
"r245r74"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of long-lived, physical assets used to produce goods and services and not intended for resale.",
"label": "Long-Lived Tangible Asset [Axis]",
"terseLabel": "Long-Lived Tangible Asset [Axis]"
}
}
},
"localname": "PropertyPlantAndEquipmentByTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PropertyPlantAndEquipmentDisclosureTextBlock": {
"auth_ref": [
"r248r76",
"r680r635",
"r681r636",
"r682r637"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for long-lived, physical asset used in normal conduct of business and not intended for resale. Includes, but is not limited to, work of art, historical treasure, and similar asset classified as collections.",
"label": "Property, Plant and Equipment Disclosure [Text Block]",
"terseLabel": "PROPERTY AND EQUIPMENT"
}
}
},
"localname": "PropertyPlantAndEquipmentDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENT"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_PropertyPlantAndEquipmentGross": {
"auth_ref": [
"r40r72",
"r243r193"
],
"calculation": {
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails": {
"order": 1.0,
"parentTag": "us-gaap_PropertyPlantAndEquipmentNet",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount before accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures.",
"label": "Property, Plant and Equipment, Gross",
"terseLabel": "Total property and equipment"
}
}
},
"localname": "PropertyPlantAndEquipmentGross",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PropertyPlantAndEquipmentLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Property, Plant and Equipment [Line Items]",
"terseLabel": "Property, Plant and Equipment [Line Items]"
}
}
},
"localname": "PropertyPlantAndEquipmentLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PropertyPlantAndEquipmentNet": {
"auth_ref": [
"r17r74",
"r18r175",
"r245r612",
"r568",
"r638",
"r650r679"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION": {
"order": 4.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
},
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 3.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount after accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business to produce goods and services and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures.",
"label": "Property, Plant and Equipment, Net",
"terseLabel": "Property and equipment, net",
"totalLabel": "Property and equipment, net"
}
}
},
"localname": "PropertyPlantAndEquipmentNet",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONPROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetailsUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PropertyPlantAndEquipmentPolicyTextBlock": {
"auth_ref": [
"r39r74",
"r245r635",
"r680",
"r681r636"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for long-lived, physical asset used in normal conduct of business and not intended for resale. Includes, but is not limited to, work of art, historical treasure, and similar asset classified as collections.",
"label": "Property, Plant and Equipment, Policy [Policy Text Block]",
"terseLabel": "Property and Equipment"
}
}
},
"localname": "PropertyPlantAndEquipmentPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_PropertyPlantAndEquipmentTextBlock": {
"auth_ref": [
"r17",
"r245r74"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of physical assets used in the normal conduct of business and not intended for resale. Includes, but is not limited to, balances by class of assets, depreciation and depletion expense and method used, including composite depreciation, and accumulated deprecation.",
"label": "Property, Plant and Equipment [Table Text Block]",
"terseLabel": "Schedule of Property and Equipment"
}
}
},
"localname": "PropertyPlantAndEquipmentTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTTables",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_PropertyPlantAndEquipmentTypeDomain": {
"auth_ref": [
"r17",
"r243r72"
],
"lang": {
"en-us": {
"role": {
"documentation": "Listing of long-lived, physical assets that are used in the normal conduct of business to produce goods and services and not intended for resale. Examples include land, buildings, machinery and equipment, and other types of furniture and equipment including, but not limited to, office equipment, furniture and fixtures, and computer equipment and software.",
"label": "Long-Lived Tangible Asset [Domain]",
"terseLabel": "Long-Lived Tangible Asset [Domain]"
}
}
},
"localname": "PropertyPlantAndEquipmentTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_PropertyPlantAndEquipmentUsefulLife": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Useful life of long lived, physical assets used in the normal conduct of business and not intended for resale, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days. Examples include, but not limited to, land, buildings, machinery and equipment, office equipment, furniture and fixtures, and computer equipment.",
"label": "Property, Plant and Equipment, Useful Life",
"terseLabel": "Estimated useful life"
}
}
},
"localname": "PropertyPlantAndEquipmentUsefulLife",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_PurchaseCommitmentExcludingLongtermCommitmentLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Purchase Commitment, Excluding Long-termTerm Commitment [Line Items]",
"terseLabel": "Purchase Commitment, Excluding Long-term Commitment [Line Items]"
}
}
},
"localname": "PurchaseCommitmentExcludingLongtermCommitmentLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PurchaseCommitmentExcludingLongtermCommitmentTable": {
"auth_ref": [
"r24r2",
"r626r156",
"r645r171"
],
"lang": {
"en-us": {
"role": {
"documentation": "Summarization of information required or determined to be disclosed about arrangements in which the entity has agreed to expend funds to procure goods or services from one or more suppliers. Such disclosure may include identification of the goods or services to be purchased, identity of the seller, pricing, effects on pricing for failing to reach minimum quantities required to be purchased (such as penalties), cancellation rights, and termination provisions.",
"label": "Purchase Commitment, Excluding Long-termTerm Commitment [Table]",
"terseLabel": "Purchase Commitment, Excluding Long-term Commitment [Table]"
}
}
},
"localname": "PurchaseCommitmentExcludingLongtermCommitmentTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RelatedPartyDomain": {
"auth_ref": [
"r3812022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_PurchaseObligation": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESUnconditionalPurchaseCommitmentDetails": {
"order": null,
"parentTag": null,
"root": true,
"weight": null
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Minimum amount of purchase arrangement in which the entity has agreed to expend funds to procure goods or services from a supplier.",
"label": "Purchase Obligation",
"totalLabel": "Total"
}
}
},
"localname": "PurchaseObligation",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESUnconditionalPurchaseCommitmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PurchaseObligationDueInNextTwelveMonths": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESUnconditionalPurchaseCommitmentDetails": {
"order": 2.0,
"parentTag": "us-gaap_PurchaseObligation",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of purchase arrangement to be paid in next fiscal year following current fiscal year. Includes, but is not limited to, recorded and unrecorded purchase obligations, long-term purchase commitment, and short-term purchase commitment. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Purchase Obligation, to be Paid, Year One",
"terseLabel": "2023"
}
}
},
"localname": "PurchaseObligationDueInNextTwelveMonths",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESUnconditionalPurchaseCommitmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_PurchaseObligationDueInSecondYear": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESUnconditionalPurchaseCommitmentDetails": {
"order": 1.0,
"parentTag": "us-gaap_PurchaseObligation",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of purchase arrangement to be paid in second fiscal year following current fiscal year. Includes, but is not limited to, recorded and unrecorded purchase obligations, long-term purchase commitment, and short-term purchase commitment. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach).",
"label": "Purchase Obligation, to be Paid, Year Two",
"terseLabel": "2024"
}
}
},
"localname": "PurchaseObligationDueInSecondYear",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESUnconditionalPurchaseCommitmentDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RecordedUnconditionalPurchaseObligationsTextBlock": {
"auth_ref": [
"r82",
"r560",
"r561r729"
],
"lang": {
"en-us": {
"role": {
"documentation": "Related parties include affiliates; other entities for which investments are accounted for by the equity method by the entity; trusts for benefit of employees; and principal owners, management, and members of immediate families. It also may include other parties with which the entity may control or can significantly influence the management or operating policies of the other to an extent that one of the transacting parties might be prevented from fully pursuing its own separate interests.",
"label": "Related Party [DomainTabular disclosure of unconditional purchase obligation recognized as liability.",
"label": "Recorded Unconditional Purchase Obligations [Table Text Block]",
"terseLabel": "Related Party [Domain]Unconditional Purchase Commitments"
}
}
},
"localname": "RelatedPartyDomainRecordedUnconditionalPurchaseObligationsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RelatedPartyTransactionLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Related Party Transaction [Line Items]",
"terseLabel": "Related Party Transaction [Line Items]"
}
}
},
"localname": "RelatedPartyTransactionLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RelatedPartyTransactionsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Related Party Transactions [Abstract]"
}
}
},
"localname": "RelatedPartyTransactionsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"xbrltype": "stringItemType"
},
"us-gaap_RelatedPartyTransactionsByRelatedPartyAxis": {
"auth_ref": [
"r381",
"r560",
"r563",
"r610",
"r611",
"r612",
"r613",
"r614",
"r615",
"r616",
"r617",
"r618",
"r619",
"r620",
"r621"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of related party. Related parties include, but not limited to, affiliates; other entities for which investments are accounted for by the equity method by the entity; trusts for benefit of employees; and principal owners, management, and members of immediate families. It also may include other parties with which the entity may control or can significantly influence the management or operating policies of the other to an extent that one of the transacting parties might be prevented from fully pursuing its own separate interests.",
"label": "Related Party [Axis]",
"terseLabel": "Related Party [Axis]"
}
}
},
"localname": "RelatedPartyTransactionsByRelatedPartyAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RelatedPartyTransactionsDisclosureTextBlock": {
"auth_ref": [
"r558",
"r559",
"r561",
"r564",
"r565"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for related party transactions. Examples of related party transactions include transactions between (a) a parent company and its subsidiary; (b) subsidiaries of a common parent; (c) and entity and its principal owners; and (d) affiliates.",
"label": "Related Party Transactions Disclosure [Text Block]",
"terseLabel": "RELATED-PARTY TRANSACTIONS"
}
}
},
"localname": "RelatedPartyTransactionsDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONS"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RepaymentsOfAssumedDebt": {
"auth_ref": [
"r932022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RepaymentsOfAssumedDebt": {
"auth_ref": [
"r40"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 42.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow from the repayments of a long-term debt originally issued by another party but is assumed by the entity.",
"label": "Repayments of Assumed Debt",
"negatedLabelnegatedTerseLabel": "Repayment of note assumed in the Business Combination",
"negatedTerseLabel": "Less: repayment of note assumed in the Business Combination"
}
}
},
"localname": "RepaymentsOfAssumedDebt",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RepaymentsOfDebt": {
"auth_ref": [
"r93"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 6.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInFinancingActivities",
"weight": -1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cash outflow during the period from the repayment of aggregate short-term and long-term debt. Excludes payment of capital lease obligations.",
"label": "Repayments of Debt",
"negatedTerseLabel": "Principal payments on debt"
}
}
},
"localname": "RepaymentsOfDebt",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ResearchAndDevelopmentArrangementContractToPerformForOthersByTypeAxis": {
"auth_ref": [
"r433r109",
"r434r110"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by form of arrangement related to research and development.",
"label": "Research and Development Arrangement, Contract to Perform for Others, Type [Axis]",
"terseLabel": "Research and Development Arrangement, Contract to Perform for Others, Type [Axis]"
}
}
},
"localname": "ResearchAndDevelopmentArrangementContractToPerformForOthersByTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ResearchAndDevelopmentArrangementContractToPerformForOthersTypeDomain": {
"auth_ref": [
"r433r109",
"r434r110"
],
"lang": {
"en-us": {
"role": {
"documentation": "Listing of significant agreements under research and development arrangements accounted for as a contract to perform research and development for others.",
"label": "Research and Development Arrangement, Contract to Perform for Others, Type [Domain]",
"terseLabel": "Research and Development Arrangement, Contract to Perform for Others, Type [Domain]"
}
}
},
"localname": "ResearchAndDevelopmentArrangementContractToPerformForOthersTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ResearchAndDevelopmentExpense": {
"auth_ref": [
"r432r108",
"r605r179",
"r694r792"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 1.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate costs incurred (1) in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or (2) to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process whether intended for sale or the entity's use, during the reporting period charged to research and development projects, including the costs of developing computer software up to the point in time of achieving technological feasibility, and costs allocated in accounting for a business combination to in-process projects deemed to have no alternative future use.",
"label": "Research and Development Expense",
"terseLabel": "Research and development"
}
}
},
"localname": "ResearchAndDevelopmentExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ResearchAndDevelopmentExpenseMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption in which the reported facts about research and development expense have been included.",
"label": "Research and Development Expense [Member]",
"terseLabel": "Research and development"
}
}
},
"localname": "ResearchAndDevelopmentExpenseMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails",
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ResearchAndDevelopmentExpensePolicy": {
"auth_ref": [
"r432r108"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for costs it has incurred (1) in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or (2) to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process.",
"label": "Research and Development Expense, Policy [Policy Text Block]",
"terseLabel": "Research and Development Costs"
}
}
},
"localname": "ResearchAndDevelopmentExpensePolicy",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ResearchMember": {
"auth_ref": [
"r449r116"
],
"lang": {
"en-us": {
"role": {
"documentation": "Research tax credit carryforwards arising from certain qualifying expenditures incurred to develop new products and processes.",
"label": "Research Tax Credit Carryforward [Member]",
"terseLabel": "Research and Development"
}
}
},
"localname": "ResearchMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RestrictedCashAndCashEquivalentsCashAndCashEquivalentsMember": {
"auth_ref": [
"r192"
],
"lang": {
"en-us": {
"role": {
"documentation": "Type of cash and cash equivalent. Cash is currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.",
"label": "Cash and Cash Equivalents [Domain]",
"terseLabel": "Cash and Cash Equivalents [Domain]"
}
}
},
"localname": "RestrictedCashAndCashEquivalentsCashAndCashEquivalentsMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RestrictedCashNoncurrentgaap_RestrictedCashCurrent": {
"auth_ref": [
"r12r697",
"r19",
"r109",
"r677r711"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails": {
"order": 13.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
},gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 4.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash restricted as to withdrawal or usage, classified as current. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits.",
"label": "Restricted Cash, Current",
"terseLabel": "Restricted cash - short-term"
}
}
},
"localname": "RestrictedCashCurrent",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RestrictedCashNoncurrent": {
"auth_ref": [
"r632",
"r698",
"r711"
],
"calculation": {
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails": {
"order": 1.0,
"parentTag": "us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents",
"weight": 1.0
},
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 2.0,
"parentTag": "us-gaap_Assets",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of cash restricted as to withdrawal or usage, classified as noncurrent. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits.",
"label": "Restricted Cash, Noncurrent",
"terseLabel": "Restricted cash - long-term"
}
}
},
"localname": "RestrictedCashNoncurrent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESCashCashEquivalentsandRestrictedCashDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RetainedEarningsAccumulatedDeficit": {
"auth_ref": [
"r31UNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RestrictedStockMember": {
"auth_ref": [
"r55"
],
"lang": {
"en-us": {
"role": {
"documentation": "Stock including a provision that prohibits sale or substantive sale of an equity instrument for a specified period of time or until specified performance conditions are met.",
"label": "Restricted Stock [Member]",
"terseLabel": "Unvested restricted stock units"
}
}
},
"localname": "RestrictedStockMember",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RestrictedStockUnitsRSUMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Share instrument which is convertible to stock or an equivalent amount of cash, after a specified period of time or when specified performance conditions are met.",
"label": "Restricted Stock Units (RSUs) [Member]",
"terseLabel": "RSUs"
}
}
},
"localname": "RestrictedStockUnitsRSUMember",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RestructuringAndRelatedActivitiesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Restructuring and Related Activities [Abstract]"
}
}
},
"localname": "RestructuringAndRelatedActivitiesAbstract",
"nsuri": "http://fasb.org/us-gaap/2022",
"xbrltype": "stringItemType"
},
"us-gaap_RestructuringAndRelatedActivitiesDisclosureTextBlock": {
"auth_ref": [
"r319",
"r321",
"r324",
"r329",
"r330"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for restructuring and related activities. Description of restructuring activities such as exit and disposal activities, include facts and circumstances leading to the plan, the expected plan completion date, the major types of costs associated with the plan activities, total expected costs, the accrual balance at the end of the period, and the periods over which the remaining accrual will be settled.",
"label": "Restructuring and Related Activities Disclosure [Text Block]",
"terseLabel": "RESTRUCTURING CHARGES"
}
}
},
"localname": "RestructuringAndRelatedActivitiesDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGES"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RestructuringAndRelatedCostNumberOfPositionsEliminated": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The number of positions eliminated during the period as a result of restructuring activities.",
"label": "Restructuring and Related Cost, Number of Positions Eliminated",
"terseLabel": "Number of positions eliminated"
}
}
},
"localname": "RestructuringAndRelatedCostNumberOfPositionsEliminated",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "integerItemType"
},
"us-gaap_RestructuringCostAndReserveLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Restructuring Cost and Reserve [Line Items]",
"terseLabel": "Restructuring Cost and Reserve [Line Items]"
}
}
},
"localname": "RestructuringCostAndReserveLineItems",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RestructuringReserve": {
"auth_ref": [
"r321",
"r325"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying amount (including both current and noncurrent portions of the accrual) as of the balance sheet date pertaining to a specified type of cost associated with exit from or disposal of business activities or restructuring pursuant to a duly authorized plan.",
"label": "Restructuring Reserve",
"periodEndLabel": "Ending balance",
"periodStartLabel": "Beginning balance"
}
}
},
"localname": "RestructuringReserve",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESRestructuringLiabilityRollforwardDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RestructuringReserveAccrualAdjustment1": {
"auth_ref": [
"r321",
"r327"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expense (reversal of expense) which increases (decreases) the restructuring reserve from an adjustment to a previously accrued restructuring liability.",
"label": "Restructuring Reserve, Accrual Adjustment",
"terseLabel": "Accrual and accrual adjustments"
}
}
},
"localname": "RestructuringReserveAccrualAdjustment1",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESRestructuringLiabilityRollforwardDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RestructuringReserveRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Restructuring Reserve [Roll Forward]",
"terseLabel": "Restructuring Reserve [Roll Forward]"
}
}
},
"localname": "RestructuringReserveRollForward",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESRestructuringLiabilityRollforwardDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_RetainedEarningsAccumulatedDeficit": {
"auth_ref": [
"r6",
"r340r91",
"r424r173",
"r568r624",
"r648r629",
"r669",
"r674r679"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 2.0,
"parentTag": "us-gaap_StockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The cumulative amount of the reporting entity's undistributed earnings or deficit.",
"label": "Retained Earnings (Accumulated Deficit)",
"negatedLabel": "Retained Earnings (Accumulated Deficit)deficit",
"terseLabel": "Accumulated deficit"
}
}
},
"localname": "RetainedEarningsAccumulatedDeficit",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONNATUREOFBUSINESSNarrativeDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetailsUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RetainedEarningsMember": {
"auth_ref": [
"r2r186",
"r125r233",
"r126r234",
"r127r235",
"r130r238",
"r140r246",
"r143r248",
"r225r306",
"r421r467",
"r422r468",
"r423r469",
"r455r488",
"r456r489",
"r508r520",
"r665r620",
"r667r622"
],
"lang": {
"en-us": {
"role": {
"documentation": "The cumulative amount of the reporting entity's undistributed earnings or deficit.",
"label": "Retained Earnings [Member]",
"terseLabel": "Accumulated Deficit"
}
}
},
"localname": "RetainedEarningsMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "domainItemType"
},
"us-gaap_RevenueFromCollaborativeArrangementExcludingRevenueFromContractWithCustomergaap_RevenueFromContractWithCustomerAbstract": {
"auth_ref": [
"r474",
"r477"
],
"crdr": "credit"],
"lang": {
"en-us": {
"role": {
"documentation": "Amount of revenue from collaborative arrangement. Excludes revenue from contract with customer under Topic 606.",
"label": "Revenue from Collaborative Arrangement, Excluding Revenue from Contract with Customer",
"terseLabel": "Revenue from arrangement [Abstract]"
}
}
},
"localname": "RevenueFromCollaborativeArrangementExcludingRevenueFromContractWithCustomerRevenueFromContractWithCustomerAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
]2022",
"xbrltype": "monetaryItemTypestringItemType"
},
"us-gaap_RevenueFromContractWithCustomerExcludingAssessedTax": {
"auth_ref": [
"r183r273",
"r184r274",
"r193r283",
"r198r286",
"r199r287",
"r203r291",
"r204r292",
"r206r294",
"r363r414",
"r364r415",
"r606r605"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 2.0,
"parentTag": "us-gaap_OperatingIncomeLoss",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount, excluding tax collected from customer, of revenue from satisfaction of performance obligation by transferring promised good or service to customer. Tax collected from customer is tax assessed by governmental authority that is both imposed on and concurrent with specific revenue-producing transaction, including, but not limited to, sales, use, value added and excise.",
"label": "Revenue from Contract with Customer, Excluding Assessed Tax",
"terseLabel": "Total revenue"
}
}
},
"localname": "RevenueFromContractWithCustomerExcludingAssessedTax",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_RevenueFromContractWithCustomerMember": {
"auth_ref": [
"r177r294",
"r206r722"
],
"lang": {
"en-us": {
"role": {
"documentation": "Revenue from satisfaction of performance obligation by transferring promised product and service to customer, when it serves as benchmark in concentration of risk calculation.",
"label": "Revenue from Contract with Customer Benchmark [Member]",
"terseLabel": "Revenue from Contract with Customer Benchmark"
}
}
},
"localname": "RevenueFromContractWithCustomerMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESWarrantsandRevenueDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_RevenueFromContractWithCustomerPolicyTextBlock": {
"auth_ref": [
"r112r406",
"r355r407",
"r356r408",
"r357r409",
"r358r410",
"r359r411",
"r360r412",
"r361r413",
"r362r419",
"r371r655"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for revenue from contract with customer.",
"label": "Revenue from Contract with Customer [Policy Text Block]",
"terseLabel": "Revenue Recognition"
}
}
},
"localname": "RevenueFromContractWithCustomerPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RevenueFromContractWithCustomerTextBlock": {
"auth_ref": [
"r346r398",
"r347r399",
"r348r400",
"r349r401",
"r350r402",
"r351r403",
"r353r404",
"r354r405",
"r367r418",
"r371r419"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure of revenue from contract with customer to transfer good or service and to transfer nonfinancial asset. Includes, but is not limited to, disaggregation of revenue, credit loss recognized from contract with customer, judgment and change in judgment related to contract with customer, and asset recognized from cost incurred to obtain or fulfill contract with customer. Excludes insurance and lease contracts.",
"label": "Revenue from Contract with Customer [Text Block]",
"terseLabel": "PAST COLLABORATIONREVENUE AND LICENSE AGREEMENTCONTRACT BALANCES"
}
}
},
"localname": "RevenueFromContractWithCustomerTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENT"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RevenueRemainingPerformanceObligationExpectedTimingOfSatisfactionPeriod1": {
"auth_ref": [
"r352"
],
"lang": {
"en-us": {
"role": {
"documentation": "Period in which remaining performance obligation is expected to be recognized as revenue, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Revenue, Remaining Performance Obligation, Expected Timing of Satisfaction, Period",
"terseLabel": "Period of satisfaction"
}
}
},
"localname": "RevenueRemainingPerformanceObligationExpectedTimingOfSatisfactionPeriod1",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_RevenueRemainingPerformanceObligationExpectedTimingOfSatisfactionStartDateAxis": {
"auth_ref": [
"r352"
],
"lang": {
"en-us": {
"role": {
"documentation": "Start date of time band for expected timing of satisfaction of remaining performance obligation, in YYYY-MM-DD format.",
"label": "Revenue, Remaining Performance Obligation, Expected Timing of Satisfaction, Start Date [Axis]",
"terseLabel": "Revenue, Remaining Performance Obligation, Expected Timing of Satisfaction, Start Date [Axis]"
}
}
},
"localname": "RevenueRemainingPerformanceObligationExpectedTimingOfSatisfactionStartDateAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "stringItemTypeREVENUEANDCONTRACTBALANCES"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_RevenuesAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Revenues [Abstract]",
"terseLabel": "Revenue"
}
}
},
"localname": "RevenuesAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "stringItemType"
},
"us-gaap_RoyaltyExpense": {
"auth_ref": [
"r82r32"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of expense related to royalty payments under a contractual arrangement such as payment for mineral and drilling rights and use of technology or intellectual property.",
"label": "Royalty Expense",
"terseLabel": "Royalty and maintenance expense"
}
}
},
"localname": "RoyaltyExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SaleOfStockConsiderationReceivedOnTransaction": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Cash received on stock transaction after deduction of issuance costs.",
"label": "Sale of Stock, Consideration Received on Transaction",
"terseLabel": "Consideration received on transaction"
}
}
},
"localname": "SaleOfStockConsiderationReceivedOnTransaction",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SaleOfStockNameOfTransactionDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Sale of the entity's stock, including, but not limited to, initial public offering (IPO) and private placement.",
"label": "Sale of Stock [Domain]",
"terseLabel": "Sale of Stock [Domain]"
}
}
},
"localname": "SaleOfStockNameOfTransactionDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsCAPITALSTOCKDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SaleOfStockNumberOfSharesIssuedInTransaction": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The number of shares issued or sold by the subsidiary or equity method investee per stock transaction.",
"label": "Sale of Stock, Number of Shares Issued in Transaction",
"terseLabel": "Sale of stock (in shares)"
}
}
},
"localname": "SaleOfStockNumberOfSharesIssuedInTransaction",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsBUSINESSCOMBINATIONDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_SaleOfStockPricePerShare": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Per share amount received by subsidiary or equity investee for each share of common stock issued or sold in the stock transaction.",
"label": "Sale of Stock, Price Per Share",
"terseLabel": "Sale of stock (in dollars per share)"
}
}
},
"localname": "SaleOfStockPricePerShare",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsBUSINESSCOMBINATIONDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the (a) carrying value as of the balance sheet date of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business (accounts payable); (b) other payables; and (c) accrued liabilities. Examples include taxes, interest, rent and utilities. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer). An alternative caption includes accrued expenses.",
"label": "Schedule of Accounts Payable and Accrued Liabilities [Table Text Block]",
"terseLabel": "Schedule of Accounts Payable and Accrued Liabilities"
}
}
},
"localname": "ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/ACCRUEDEXPENSESANDOTHERCURRENTLIABILITIESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTable": {
"auth_ref": [
"r160r55"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule for securities (including those issuable pursuant to contingent stock agreements) that could potentially dilute basic earnings per share (EPS) in the future that were not included in the computation of diluted EPS because to do so would increase EPS amounts or decrease loss per share amounts for the period presented, by Antidilutive Securities.",
"label": "Schedule of Antidilutive Securities Excluded from Computation of Earnings Per Share [Table]",
"terseLabel": "Schedule of Antidilutive Securities Excluded from Computation of Earnings Per Share [Table]"
}
}
},
"localname": "ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTextBlock": {
"auth_ref": [
"r160r55"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of securities (including those issuable pursuant to contingent stock agreements) that could potentially dilute basic earnings per share (EPS) in the future that were not included in the computation of diluted EPS because to do so would increase EPS amounts or decrease loss per share amounts for the period presented, by antidilutive securities.",
"label": "Schedule of Antidilutive Securities Excluded from Computation of Earnings Per Share [Table Text Block]",
"terseLabel": "Schedule of Antidilutive Securities"
}
}
},
"localname": "ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHARETables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfBusinessAcquisitionsByAcquisitionTable": {
"auth_ref": [
"r465r118",
"r466r119",
"r498"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule reflecting each material business combination (or series of individually immaterial business combinations) completed during the period, including background, timing, and recognized assets and liabilities.",
"label": "Schedule of Business Acquisitions, by Acquisition [Table]",
"terseLabel": "Schedule of Business Acquisitions, by Acquisition [Table]"
}
}
},
"localname": "ScheduleOfBusinessAcquisitionsByAcquisitionTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfCashAndCashEquivalentsTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of cash and cash equivalents.",
"label": "Schedule of Cash and Cash Equivalents [Table Text Block]",
"terseLabel": "Schedule of Cash and Cash Equivalents"
}
}
},
"localname": "ScheduleOfCashAndCashEquivalentsTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfCollaborativeArrangementsAndNoncollaborativeArrangementTransactionsTable": {
"auth_ref": [
"r478r773"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about collaborative arrangement and arrangement other than collaborative applicable to revenue-generating activity or operations.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Table]",
"terseLabel": "Collaborative Arrangement and Arrangement Other than Collaborative [Table]"
}
}
},
"localname": "ScheduleOfCollaborativeArrangementsAndNoncollaborativeArrangementTransactionsTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfDebtInstrumentsTextBlock": {
"auth_ref": [
"r50REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfCostsOfRetirementPlansTableTextBlock": {
"auth_ref": [
"r94"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the costs related to the various types of retirement plans including defined benefit pension plan cost, defined contribution plan cost, other postretirement benefit plan cost, and net periodic benefit cost.",
"label": "Schedule of Costs of Retirement Plans [Table Text Block]",
"terseLabel": "Schedule of Severance, Employee Benefits, and Related Costs"
}
}
},
"localname": "ScheduleOfCostsOfRetirementPlansTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfDebtInstrumentsTextBlock": {
"auth_ref": [
"r18",
"r123r87",
"r304r88",
"r306r89",
"r334r90",
"r337r149",
"r338r150",
"r339r152",
"r542r165",
"r543r662",
"r546r664",
"r639r715"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of long-debt instruments or arrangements, including identification, terms, features, collateral requirements and other information necessary to a fair presentation. These are debt arrangements that originally required repayment more than twelve months after issuance or greater than the normal operating cycle of the entity, if longer.",
"label": "Schedule of Long-termTerm Debt Instruments [Table Text Block]",
"terseLabel": "Schedule of Long Term Debt"
}
}
},
"localname": "ScheduleOfDebtInstrumentsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock": {
"auth_ref": [
"r445r114"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the components of net deferred tax asset or liability recognized in an entity's statement of financial position, including the following: the total of all deferred tax liabilities, the total of all deferred tax assets, the total valuation allowance recognized for deferred tax assets.",
"label": "Schedule of Deferred Tax Assets and Liabilities [Table Text Block]",
"terseLabel": "Schedule of Deferred Tax Assets and Liabilities"
}
}
},
"localname": "ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock": {
"auth_ref": [
"r159r721"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of an entity's basic and diluted earnings per share calculations, including a reconciliation of numerators and denominators of the basic and diluted per-share computations for income from continuing operations.",
"label": "Schedule of Earnings Per Share, Basic and Diluted [Table Text Block]",
"terseLabel": "Schedule of Net Loss Per Share"
}
}
},
"localname": "ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NETLOSSPERSHARETables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock": {
"auth_ref": [
"r438r111"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the reconciliation using percentage or dollar amounts of the reported amount of income tax expense attributable to continuing operations for the year to the amount of income tax expense that would result from applying domestic federal statutory tax rates to pretax income from continuing operations.",
"label": "Schedule of Effective Income Tax Rate Reconciliation [Table Text Block]",
"terseLabel": "Schedule of Effective Income Tax Rate Reconciliation"
}
}
},
"localname": "ScheduleOfEffectiveIncomeTaxRateReconciliationTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTable": {
"auth_ref": [
"r390r101",
"r412",
"r426r104"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about amount recognized for award under share-based payment arrangement. Includes, but is not limited to, amount expensed in statement of income or comprehensive income, amount capitalized in statement of financial position, and corresponding reporting line item in financial statements.",
"label": "Share-basedBased Payment Arrangement, Expensed and Capitalized, Amount [Table]",
"terseLabel": "Share-based Payment Arrangement, Expensed and Capitalized, Amount [Table]"
}
}
},
"localname": "ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock": {
"auth_ref": [
"r390",
"r412",
"r426r101"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of allocation of amount expensed and capitalized for award under share-based payment arrangement to statement of income or comprehensive income and statement of financial position. Includes, but is not limited to, corresponding line item in financial statement.",
"label": "Share-basedBased Payment Arrangement, Expensed and Capitalized, Amount [Table Text Block]",
"terseLabel": "Schedule of Stock Based Compensation Expense"
}
}
},
"localname": "ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfFutureMinimumRentalPaymentsForOperatingLeasesTableTextBlock": {
"auth_ref": [
"r550r183"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of future minimum payments required in the aggregate and for each of the five succeeding fiscal years for operating leases having initial or remaining noncancelable lease terms in excess of one year and the total minimum rentals to be received in the future under noncancelable subleases as of the balance sheet date.",
"label": "Schedule of Future Minimum Rental Payments for Operating Leases [Table Text Block]",
"terseLabel": "Schedule of Lease Payments"
}
}
},
"localname": "ScheduleOfFutureMinimumRentalPaymentsForOperatingLeasesTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/COMMITMENTSANDCONTINGENCIESTablesLEASESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfMaturitiesOfLongTermDebtTableTextBlock": {
"auth_ref": [
"r258r82"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of maturity and sinking fund requirement for long-term debt.",
"label": "Schedule of Maturities of Long-termTerm Debt [Table Text Block]",
"terseLabel": "Schedule of Future Minimum Payments"
}
}
},
"localname": "ScheduleOfMaturitiesOfLongTermDebtTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfPropertyPlantAndEquipmentTable": {
"auth_ref": [
"r41",
"r245r74"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about physical assets used in the normal conduct of business and not intended for resale. Includes, but is not limited to, balances by class of assets, depreciation and depletion expense and method used, including composite depreciation, and accumulated deprecation.",
"label": "Property, Plant and Equipment [Table]",
"terseLabel": "Property, Plant and Equipment [Table]"
}
}
},
"localname": "ScheduleOfPropertyPlantAndEquipmentTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfRelatedPartyTransactionsByRelatedPartyTable": {
"auth_ref": [
"r562",
"r563"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule of quantitative and qualitative information pertaining to related party transactions. Examples of related party transactions include transactions between (a) a parent company and its subsidiary; (b) subsidiaries of a common parent; (c) and entity and its principal owners; and (d) affiliates.",
"label": "Schedule of Related Party Transactions, by Related Party [Table]",
"terseLabel": "Schedule of Related Party Transactions, by Related Party [Table]"
}
}
},
"localname": "ScheduleOfRelatedPartyTransactionsByRelatedPartyTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfRestrictedCashAndCashEquivalentsTextBlock": {
"auth_ref": [
"r16gaap_ScheduleOfRestrictedCashAndCashEquivalentsTextBlock": {
"auth_ref": [
"r52",
"r109r153",
"r622",
"r646r172"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of cash and cash equivalents restricted as to withdrawal or usage.",
"label": "Restrictions on Cash and Cash Equivalents [Table Text Block]",
"terseLabel": "Schedule of Restricted Cash"
}
}
},
"localname": "ScheduleOfRestrictedCashAndCashEquivalentsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfShareBasedCompensationArrangementsByShareBasedPaymentAwardTable": {
"auth_ref": [
"r391",
"r4152022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfRestructuringAndRelatedCostsTable": {
"auth_ref": [
"r320",
"r321",
"r322",
"r323",
"r326",
"r327",
"r328"
],
"lang": {
"en-us": {
"role": {
"documentation": "Table presenting the description of the restructuring costs, such as the expected cost; the costs incurred during the period; the cumulative costs incurred as of the balance sheet date; the income statement caption within which the restructuring charges recognized for the period are included; and the amount of and periodic changes to an entity's restructuring reserve that occurred during the period associated with the exit from or disposal of business activities or restructurings for each major type of cost by type of restructuring.",
"label": "Schedule of Restructuring and Related Costs [Table]",
"terseLabel": "Schedule of Restructuring and Related Costs [Table]"
}
}
},
"localname": "ScheduleOfRestructuringAndRelatedCostsTable",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfRestructuringReserveByTypeOfCostTextBlock": {
"auth_ref": [
"r77",
"r78"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of an entity's restructuring reserve that occurred during the period associated with the exit from or disposal of business activities or restructurings for each major type of cost. This element may also include a description of any reversal and other adjustment made during the period to the amount of an accrued liability for restructuring activities. This element may be used to encapsulate the roll forward presentations of an entity's restructuring reserve by type of cost and in total, and explanation of changes that occurred in the period.",
"label": "Schedule of Restructuring Reserve by Type of Cost [Table Text Block]",
"terseLabel": "Restructuring Reserve Roll Forward"
}
}
},
"localname": "ScheduleOfRestructuringReserveByTypeOfCostTextBlock",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfShareBasedCompensationArrangementsByShareBasedPaymentAwardTable": {
"auth_ref": [
"r429",
"r431",
"r433",
"r434",
"r435",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r450",
"r451",
"r452",
"r453",
"r454",
"r457",
"r458",
"r459",
"r460",
"r461"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about share-based payment arrangement.",
"label": "Schedule of Share-basedBased Compensation Arrangements by Share-basedBased Payment Award [Table]",
"terseLabel": "Schedule of Share-based Compensation Arrangements by Share-based Payment Award [Table]"
}
}
},
"localname": "ScheduleOfShareBasedCompensationArrangementsByShareBasedPaymentAwardTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock": {
"auth_ref": [
"r396r96",
"r402r97",
"r404r98"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure for stock option plans. Includes, but is not limited to, outstanding awards at beginning and end of year, grants, exercises, forfeitures, and weighted-average grant date fair value.",
"label": "Share-basedBased Payment Arrangement, Option, Activity [Table Text Block]",
"terseLabel": "Schedule of Stock Option Activity"
}
}
},
"localname": "ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock": {
"auth_ref": [
"r406r100"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of the significant assumptions used during the year to estimate the fair value of stock options, including, but not limited to: (a) expected term of share options and similar instruments, (b) expected volatility of the entity's shares, (c) expected dividends, (d) risk-free rate(s), and (e) discount for post-vesting restrictions.",
"label": "Schedule of Share-basedBased Payment Award, Stock Options, Valuation Assumptions [Table Text Block]",
"terseLabel": "Schedule of Valuation Assumptions"
}
}
},
"localname": "ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ScheduleOfStockByClassTable": {
"auth_ref": [
"r53",
"r114",
"r167",
"r168",
"r312",
"r314",
"r315",
"r316",
"r317",
"r319",
"r320",
"r322",
"r326",
"r331",
"r334",
"r335",
"r336",
"r337",
"r338",
"r339",
"r340"
gaap_ScheduleOfSharebasedCompensationRestrictedStockAndRestrictedStockUnitsActivityTableTextBlock": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule detailing information related to equity by class of stock. Class ofDisclosure of the number and weighted-average grant date fair value for restricted stock includes common, convertible, and preferred stocks which areand restricted stock units that not redeemable or redeemable solelywere outstanding at the optionbeginning and end of the issuer. Ityear, and also includes preferredthe number of restricted stock with redemption featuresand restricted stock units that are solely within the control of the issuer and mandatorily redeemable stock if redemption is required to occur only upon liquidation or termination of the reporting entitywere granted, vested, or forfeited during the year.",
"label": "Schedule ofShare-Based Payment Arrangement, Restricted Stock by Classand Restricted Stock Unit, Activity [Table Text Block]",
"terseLabel": "Schedule of Stock by Class [Table]RSU Activity"
}
}
},
"localname": "ScheduleOfStockByClassTableScheduleOfSharebasedCompensationRestrictedStockAndRestrictedStockUnitsActivityTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails"
],
"xbrltype": "stringItemTypeSTOCKBASEDCOMPENSATIONTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SegmentReportingPolicyPolicyTextBlock": {
"auth_ref": [
"r187r277",
"r188r278",
"r189r279",
"r190r280",
"r191r281",
"r192r282",
"r204r292"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for segment reporting.",
"label": "Segment Reporting, Policy [Policy Text Block]",
"terseLabel": "Segment and Geographic Information"
}
}
},
"localname": "SegmentReportingPolicyPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SellingGeneralAndAdministrativeExpense": {
"auth_ref": [
"r83r33"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS": {
"order": 2.0,
"parentTag": "us-gaap_CostsAndExpenses",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The aggregate total costs related to selling a firm's product and services, as well as all other general and administrative expenses. Direct selling expenses (for example, credit, warranty, and advertising) are expenses that can be directly linked to the sale of specific products. Indirect selling expenses are expenses that cannot be directly linked to the sale of specific products, for example telephone expenses, Internet, and postal charges. General and administrative expenses include salaries of non-sales personnel, rent, utilities, communication, etc.",
"label": "Selling, General and Administrative Expense",
"terseLabel": "Selling, general, and administrative"
}
}
},
"localname": "SellingGeneralAndAdministrativeExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SellingGeneralAndAdministrativeExpensesMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Primary financial statement caption encompassing selling, general and administrative expense.",
"label": "Selling, General and Administrative Expenses [Member]",
"terseLabel": "Selling, general, and administrative (1)"
}
}
},
"localname": "SellingGeneralAndAdministrativeExpensesMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/RESTRUCTURINGCHARGESScheduleofSeveranceEmployeeBenefitsandRelatedCostsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SellingGeneralAndAdministrativeExpensesPolicyTextBlock": {
"auth_ref": [
"r430r107"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for inclusion of significant items in the selling, general and administrative (or similar) expense report caption.",
"label": "Selling, General and Administrative Expenses, Policy [Policy Text Block]",
"terseLabel": "Selling, General and Administrative Expenses"
}
}
},
"localname": "SellingGeneralAndAdministrativeExpensesPolicyTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SeriesAPreferredStockMembergaap_SeriesCPreferredStockMember": {
"auth_ref": [
"r699",
"r700",
"r737"
],
"lang": {
"en-us": {
"role": {
"documentation": "Outstanding nonredeemable series A preferred stock or outstanding series A preferred stock. Classified within stockholders' equity if nonredeemable or redeemable solely at the option of the issuer. Classified within temporary equity if redemption is outside the control of the issuer.",
"label": "Series A Preferred Stock [Member]",
"terseLabel": "Series A Preferred Stock"
}
}
},
"localname": "SeriesAPreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SeriesBPreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Outstanding nonredeemable series B preferred stock or outstanding series B preferred stock. Classified within stockholders' equity if nonredeemable or redeemable solely at the option of the issuer. Classified within temporary equity if redemption is outside the control of the issuer.",
"label": "Series B Preferred Stock [Member]",
"terseLabel": "Series B Preferred Stock"
}
}
},
"localname": "SeriesBPreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SeriesCPreferredStockMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Outstanding nonredeemable series C preferred stock or outstanding series C preferred stock. Classified within stockholders' equity if nonredeemable or redeemable solely at the option of the issuer. Classified within temporary equity if redemption is outside the control of the issuerSeries C preferred stock.",
"label": "Series C Preferred Stock [Member]",
"terseLabel": "Series C Preferred Stock"
}
}
},
"localname": "SeriesCPreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",2022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SeriesDPreferredStockMember": {
"auth_ref": [
"r699",
"r700",
"r737"
],
"lang": {
"en-us": {
"role": {
"documentation": "Outstanding nonredeemable series D preferred stock or outstanding series D preferred stock. Classified within stockholders' equity if nonredeemable or redeemable solely at the option of the issuer. Classified within temporary equity if redemption is outside the control of the issuerSeries D preferred stock.",
"label": "Series D Preferred Stock [Member]",
"terseLabel": "Series D Preferred Stock"
}
}
},
"localname": "SeriesDPreferredStockMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ShareBasedCompensation": {
"auth_ref": [
"r100r44"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS": {
"order": 42.0,
"parentTag": "us-gaap_NetCashProvidedByUsedInOperatingActivities",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of noncash expense for share-based payment arrangement.",
"label": "Share-basedBased Payment Arrangement, Noncash Expense",
"terseLabel": "Stock-based compensation expense"
}
}
},
"localname": "ShareBasedCompensation",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardDiscountFromMarketPricePurchaseDate": {
"auth_ref": [
"r415UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardRequisiteServicePeriod1": {
"auth_ref": [
"r676"
],
"lang": {
"en-us": {
"role": {
"documentation": "Estimated period over which an employee is required to provide service in exchange for the equity-based payment award, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Award Requisite Service Period",
"terseLabel": "Award requisite service period"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardRequisiteServicePeriod1",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardVestingPeriod1": {
"auth_ref": [
"r676"
],
"lang": {
"en-us": {
"role": {
"documentation": "Period over which grantee's right to exercise award under share-based payment arrangement is no longer contingent on satisfaction of service or performance condition, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Includes, but is not limited to, combination of market, performance or service condition.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Award Vesting Period",
"terseLabel": "Vesting period"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardVestingPeriod1",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardDiscountFromMarketPricePurchaseDate": {
"auth_ref": [
"r102"
],
"lang": {
"en-us": {
"role": {
"documentation": "Discount rate from fair value on purchase date that participants pay for shares.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Discount from Market Price, Purchase Date",
"terseLabel": "Discount percentage"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardDiscountFromMarketPricePurchaseDate",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate": {
"auth_ref": [
"r4092022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeitedInPeriod": {
"auth_ref": [
"r450"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of equity-based payment instruments, excluding stock (or unit) options, that were forfeited during the reporting period.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Equity Instruments Other than Options, Forfeited in Period",
"negatedTerseLabel": "Forfeited and canceled (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeitedInPeriod",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeituresWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r450"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average fair value as of the grant date of equity-based award plans other than stock (unit) option plans that were not exercised or put into effect as a result of the occurrence of a terminating event.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Equity Instruments Other than Options, Forfeitures, Weighted Average Grant Date Fair Value",
"terseLabel": "Forfeited and canceled (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeituresWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriodWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r448"
],
"lang": {
"en-us": {
"role": {
"documentation": "The weighted average fair value at grant date for nonvested equity-based awards issued during the period on other than stock (or unit) option plans (for example, phantom stock or unit plan, stock or unit appreciation rights plan, performance target plan).",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Equity Instruments Other than Options, Grants in Period, Weighted Average Grant Date Fair Value",
"terseLabel": "Granted (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriodWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber": {
"auth_ref": [
"r445",
"r446"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of non-vested equity-based payment instruments, excluding stock (or unit) options, that validly exist and are outstanding as of the balance sheet date.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Equity Instruments Other than Options, Nonvested, Number",
"periodEndLabel": "Ending balance (in shares)",
"periodStartLabel": "Beginning balance (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Equity Instruments Other than Options, Nonvested, Number of Shares [Roll Forward]",
"terseLabel": "Number of Shares"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedRollForward",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r445",
"r446"
],
"lang": {
"en-us": {
"role": {
"documentation": "Per share or unit weighted-average fair value of nonvested award under share-based payment arrangement. Excludes share and unit options.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Equity Instruments Other than Options, Nonvested, Weighted Average Grant Date Fair Value",
"periodEndLabel": "Ending balance (in dollars per share)",
"periodStartLabel": "Beginning balance (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValueRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Equity Instruments Other than Options, Nonvested, Weighted Average Grant Date Fair Value [Abstract]",
"terseLabel": "Weighted Average Fair Value"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValueRollForward",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate": {
"auth_ref": [
"r459"
],
"lang": {
"en-us": {
"role": {
"documentation": "The estimated dividend rate (a percentage of the share price) to be paid (expected dividends) to holders of the underlying shares over the option's term.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Fair Value Assumptions, Expected Dividend Rate",
"terseLabel": "Expected dividend yield"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedVolatilityRate": {
"auth_ref": [
"r408r458"
],
"lang": {
"en-us": {
"role": {
"documentation": "The estimated measure of the percentage by which a share price is expected to fluctuate during a period. Volatility also may be defined as a probability-weighted measure of the dispersion of returns about the mean. The volatility of a share price is the standard deviation of the continuously compounded rates of return on the share over a specified period. That is the same as the standard deviation of the differences in the natural logarithms of the stock prices plus dividends, if any, over the period.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Fair Value Assumptions, Expected Volatility Rate",
"terseLabel": "Expected volatility"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedVolatilityRate",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRate": {
"auth_ref": [
"r410r460"
],
"lang": {
"en-us": {
"role": {
"documentation": "The risk-free interest rate assumption that is used in valuing an option on its own shares.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Fair Value Assumptions, Risk Free Interest Rate",
"terseLabel": "Risk-free interest rate"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRate",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award [Line Items]",
"terseLabel": "Share-based Compensation Arrangement by Share-based Payment Award [Line Items]"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "stringItemTypeSTOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfAdditionalSharesAuthorized": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of additional shares authorized for issuance under share-based payment arrangement.",
"label": "Share-Based Compensation Arrangement by Share-Based Payment Award, Number of Additional Shares Authorized",
"terseLabel": "Number of additional shares authorized (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfAdditionalSharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfSharesAuthorized": {
"auth_ref": [
"r392r677"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares authorized for issuance under share-based payment arrangement.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Number of Shares Authorized",
"terseLabel": "Shares authorized for grant (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfSharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableNumber": {
"auth_ref": [
"r399r439"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of shares into which fully or partially vested stock options outstanding as of the balance sheet date can be currently converted under the option plan.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Exercisable, Number",
"terseLabel": "Options exercisable (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableNumber",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableWeightedAverageExercisePrice": {
"auth_ref": [
"r399r439"
],
"lang": {
"en-us": {
"role": {
"documentation": "The weighted-average price as of the balance sheet date at which grantees can acquire the shares reserved for issuance on vested portions of options outstanding and currently exercisable under the stock option plan.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Exercisable, Weighted Average Exercise Price",
"terseLabel": "Options exercisable (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresAndExpirationsInPeriod": {
"auth_ref": [
"r401r748"
],
"lang": {
"en-us": {
"role": {
"documentation": "For presentations that combine terminations, the number of shares under options that were cancelled during the reporting period as a result of occurrence of a terminating event specified in contractual agreements pertaining to the stock option plan or that expired.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Forfeitures and Expirations in Period",
"negatedTerseLabel": "Canceled and forfeited (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresAndExpirationsInPeriod",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresAndExpirationsInPeriodWeightedAverageExercisePrice": {
"auth_ref": [
"r401r748"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average price of options that were either forfeited or expired.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Forfeitures and Expirations in Period, Weighted Average Exercise Price",
"terseLabel": "Canceled and forfeited (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresAndExpirationsInPeriodWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresAndExpirationsInPeriodWeightedAverageExercisePriceAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Forfeitures and Expirations in Period, Weighted Average Exercise Price [Abstract]",
"terseLabel": "Weighted Average Exercise Price (1)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresAndExpirationsInPeriodWeightedAverageExercisePriceAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross": {
"auth_ref": [
"r441"
],
"lang": {
"en-us": {
"role": {
"documentation": "Gross number of share options (or share units) granted during the period.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Grants in Period, Gross",
"terseLabel": "Granted in period (in shares)",
"verboseLabel": "Granted (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageGrantDateFairValue": {
"auth_ref": [
"r403"
],
"lang": {
"en-us": {
"role": {
"documentation": "The weighted average grant-date fair value of options granted during the reporting period as calculated by applying the disclosed option pricing methodology.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Options, Grants in Period, Weighted Average Grant Date Fair Value",
"terseLabel": "Weighted average grant date fair value of stock options (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageGrantDateFairValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails,
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails"
],
"xbrltype": "perShareItemTypesharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingIntrinsicValue": {
"auth_ref": [
"r415r102"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount by which the current fair value of the underlying stock exceeds the exercise price of options outstanding.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Outstanding, Intrinsic Value",
"terseLabel": "Options outstanding, aggregate intrinsic value"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingIntrinsicValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber": {
"auth_ref": [
"r398r437",
"r415r438"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of options outstanding, including both vested and non-vested options.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Outstanding, Number",
"periodEndLabel": "Ending balance, options outstanding (in shares)",
"periodStartLabel": "Beginning balance, options outstanding (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingRollForward": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "A roll forward is a reconciliation of a concept from the beginning of a period to the end of a period.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Outstanding [Roll Forward]",
"terseLabel": "Stock Options"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingRollForward",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePrice": {
"auth_ref": [
"r397r437",
"r438"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average price at which grantees can acquire the shares reserved for issuance under the stock option plan.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Outstanding, Weighted Average Exercise Price",
"periodEndLabel": "Ending balance, options outstanding (in dollars per share)",
"periodStartLabel": "Beginning balance, options outstanding (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardPercentageOfOutstandingStockMaximum": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Maximum number of shares that may be issued in accordance with the plan as a proportion of outstanding capital stock.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Percentage of Outstanding Stock Maximum",
"terseLabel": "Percentage of outstanding stock maximum"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardPercentageOfOutstandingStockMaximum",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardSharesIssuedInPeriod": {
"auth_ref": [
"r415"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares issued under share-based payment arrangement.",
"label": "Share-based Compensation Arrangement by Share-based Payment Award, Shares Issued in Period",
"terseLabel": "Shares issued (in shares)"
}
}
},
"localname": "ShareBasedCompensationArrangementByShareBasedPaymentAwardSharesIssuedInPeriod",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardAwardTypeAndPlanNameDomain": {
"auth_ref": [
"r389",
"r393r433",
"r434",
"r435",
"r437",
"r438",
"r439",
"r440",
"r441",
"r442",
"r443",
"r444",
"r445",
"r446",
"r447",
"r448",
"r449",
"r450",
"r451",
"r452",
"r453",
"r454",
"r457",
"r458",
"r459",
"r460",
"r461"
],
"lang": {
"en-us": {
"role": {
"documentation": "Award under share-based payment arrangement.",
"label": "Award Type [Domain]",
"terseLabel": "Award Type [Domain]"
}
}
},
"localname": "ShareBasedCompensationArrangementsByShareBasedPaymentAwardAwardTypeAndPlanNameDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONRestrictedStockUnitsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsExercisesInPeriodWeightedAverageExercisePrice": {
"auth_ref": [
"r442"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average price at which option holders acquired shares when converting their stock options into shares.",
"label": "Share-basedBased Compensation Arrangements by Share-basedBased Payment Award, Options, Exercises in Period, Weighted Average Exercise Price",
"terseLabel": "Exercised (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsExercisesInPeriodWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageExercisePrice": {
"auth_ref": [
"r441"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average per share amount at which grantees can acquire shares of common stock by exercise of options.",
"label": "Share-basedBased Compensation Arrangements by Share-basedBased Payment Award, Options, Grants in Period, Weighted Average Exercise Price",
"terseLabel": "Granted (in dollars per share)"
}
}
},
"localname": "ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageExercisePrice",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_ShareBasedCompensationOptionAndIncentivePlansPolicy": {
"auth_ref": [
"r391r436",
"r394r455",
"r456",
"r457",
"r458",
"r461",
"r470",
"r471"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for award under share-based payment arrangement. Includes, but is not limited to, methodology and assumption used in measuring cost.",
"label": "Share-basedBased Payment Arrangement [Policy Text Block]",
"terseLabel": "Stock-Based Compensation"
}
}
},
"localname": "ShareBasedCompensationOptionAndIncentivePlansPolicy",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SharePrice": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Price of a single share of a number of saleable stocks of a company.",
"label": "Share Price",
"terseLabel": "Price per shareFair value at grant date (in dollars per share)"
}
}
},
"localname": "SharePrice",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetailsCAPITALSTOCKDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardFairValueAssumptionsExpectedTerm1": {
"auth_ref": [
"r407",
"r425r457"
],
"lang": {
"en-us": {
"role": {
"documentation": "Expected term of award under share-based payment arrangement, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Fair Value Assumptions, Expected Term",
"terseLabel": "Expected term (years)"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardFairValueAssumptionsExpectedTerm1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONValuationAssumptionsDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsExercisableIntrinsicValue1": {
"auth_ref": [
"r415r102"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of difference between fair value of the underlying shares reserved for issuance and exercise price of vested portions of options outstanding and currently exercisable.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Exercisable, Intrinsic Value",
"terseLabel": "Options exercisable, aggregate intrinsic value"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsExercisableIntrinsicValue1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsExercisableWeightedAverageRemainingContractualTerm1": {
"auth_ref": [
"r415r102"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average remaining contractual term for vested portions of options outstanding and currently exercisable or convertible, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Exercisable, Weighted Average Remaining Contractual Term",
"terseLabel": "Options exercisable, weighted average remaining contractual life"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsExercisableWeightedAverageRemainingContractualTerm1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsOutstandingWeightedAverageRemainingContractualTerm2": {
"auth_ref": [
"r405r99"
],
"lang": {
"en-us": {
"role": {
"documentation": "Weighted average remaining contractual term for option awards outstanding, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Outstanding, Weighted Average Remaining Contractual Term",
"terseLabel": "Options outstanding, weighted average remaining contractual life"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsOutstandingWeightedAverageRemainingContractualTerm2",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetails"
],
"xbrltype": "durationItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsVestedInPeriodFairValue1": {
"auth_ref": [
"r395r452"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Fair value of options vested. Excludes equity instruments other than options, for example, but not limited to, share units, stock appreciation rights, restricted stock.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Vested in Period, Fair Value",
"terseLabel": "Fair value of vested stock options"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsVestedInPeriodFairValue1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardPurchasePriceOfCommonStockPercent": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Purchase price of common stock expressed as a percentage of its fair value.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Purchase Price of Common Stock, Percent",
"terseLabel": "Purchase price percentage"
}
}
},
"localname": "SharebasedCompensationArrangementBySharebasedPaymentAwardPurchasePriceOfCommonStockPercent",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails"
],
"xbrltype": "percentItemType"
},
"us-gaap_SharesIssued": {
"auth_ref": [
"r333"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares of stock issued as of the balance sheet date, including shares that had been issued and were previously outstanding but which are now held in the treasury.",
"label": "Shares, Issued",
"terseLabel": "Founder shares issued"
}
}
},
"localname": "SharesIssued",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_SharesIssuedPricePerShare": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Per share or per unit amount of equity securities issued.",
"label": "Shares Issued, Price Per Share",
"terseLabel": "Price per share (in dollars per share)"
}
}
},
"localname": "SharesIssuedPricePerShare",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetailsCAPITALSTOCKDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_SharesOutstanding": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares issued which are neither cancelled nor held in the treasury.",
"label": "Shares, Outstanding",
"periodEndLabel": "Ending balance (in shares)",
"periodStartLabel": "Beginning balance (in shares)"
}
}
},
"localname": "SharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "sharesItemType"
},
"us-gaap_ShortTermInvestments": {
"auth_ref": [
"r21r158",
"r628r159",
"r629r168",
"r642r701"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 25.0,
"parentTag": "us-gaap_AssetsCurrent",
"weight": 1.0
}
},
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of investments including trading securities, available-for-sale securities, held-to-maturity securities, and short-term investments classified as other and current.",
"label": "Short-termTerm Investments",
"terseLabel": "Short-term investments"
}
}
},
"localname": "ShortTermInvestments",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_SignificantAccountingPoliciesTextBlock": {
"auth_ref": [
"r110r53",
"r124r223"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for all significant accounting policies of the reporting entity.",
"label": "Significant Accounting Policies [Text Block]",
"terseLabel": "SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES"
}
}
},
"localname": "SignificantAccountingPoliciesTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIES"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SoftwareAndSoftwareDevelopmentCostsMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Purchased software applications and internally developed software for sale, licensing or long-term internal use.",
"label": "Software and Software Development Costs [Member]",
"terseLabel": "Software and Software Development CostsInternal-use software"
}
}
},
"localname": "SoftwareAndSoftwareDevelopmentCostsMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SoftwareDevelopmentMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Internally developed software for sale, licensing or long-term internal use.",
"label": "Software Development [Member]",
"terseLabel": "Internal-use software"
}
}
},
"localname": "SoftwareDevelopmentMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PROPERTYANDEQUIPMENTDetails",
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPropertyandEquipmentDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_StateAndLocalJurisdictionMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Designated tax department of a state or local government entitled to levy and collect income taxes from the entity.",
"label": "State and Local Jurisdiction [Member]",
"terseLabel": "State"
}
}
},
"localname": "StateAndLocalJurisdictionMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_StatementClassOfStockAxis": {
"auth_ref": [
"r26r198",
"r27r199",
"r28r200",
"r114r226",
"r117r253",
"r148r254",
"r149",
"r154",
"r157",
"r159",
"r167",
"r168",
"r169",
"r221",
"r261",
"r265r257",
"r259",
"r266",
"r267",
"r270r302",
"r271r337",
"r316r339",
"r317r340",
"r322r341",
"r326r344",
"r333r345",
"r525",
"r703r376",
"r377",
"r381",
"r385",
"r392",
"r529",
"r649",
"r694",
"r712",
"r720"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by the different classes of stock of the entity.",
"label": "Class of Stock [Axis]",
"terseLabel": "Class of Stock [Axis]"
}
}
},
"localname": "StatementClassOfStockAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONScheduleofReverseCapitalizationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/Cover",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails",
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "stringItemType"
},
"us-gaap_StatementEquityComponentsAxis": {
"auth_ref": [
"r2r23",
"r56r86",
"r74r186",
"r75r212",
"r76r213",
"r125r214",
"r126r233",
"r127r234",
"r130r235",
"r140r238",
"r143r246",
"r166r248",
"r225r265",
"r333r306",
"r340r394",
"r421r467",
"r422r468",
"r423r469",
"r455r488",
"r456r489",
"r508",
"r535",
"r536",
"r537r520",
"r538",
"r539",
"r540",
"r665r541",
"r666r542",
"r667r543",
"r717r568",
"r620",
"r621",
"r622"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by component of equity.",
"label": "Equity Components [Axis]",
"terseLabel": "Equity Components [Axis]"
}
}
},
"localname": "StatementEquityComponentsAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",2022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "stringItemType"
},
"us-gaap_StatementLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Statement [Line Items]",
"terseLabel": "Statement [Line Items]"
}
}
},
"localname": "StatementLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "stringItemType"
},
"us-gaap_StatementOfCashFlowsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Statement of Cash Flows [Abstract]"
}
}
},
"localname": "StatementOfCashFlowsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_StatementOfFinancialPositionAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Statement of Financial Position [Abstract]"
}
}
},
"localname": "StatementOfFinancialPositionAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_StatementOfStockholdersEquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Statement of Stockholders' Equity [Abstract]"
}
}
},
"localname": "StatementOfStockholdersEquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_StatementTable": {
"auth_ref": [
"r125r233",
"r126r234",
"r127r235",
"r166r265",
"r606r605"
],
"lang": {
"en-us": {
"role": {
"documentation": "Schedule reflecting a Statement of Income, Statement of Cash Flows, Statement of Financial Position, Statement of Shareholders' Equity and Other Comprehensive Income, or other statement as needed.",
"label": "Statement [Table]",
"terseLabel": "Statement [Table]"
}
}
},
"localname": "StatementTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITParentheticalUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITYParenthetical"
],
"xbrltype": "stringItemType"
},
"us-gaap_StockGrantedDuringPeriodValueSharebasedCompensationGross": {
"auth_ref": [],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Value, before forfeiture, of shares granted under share-based payment arrangement. Excludes employee stock ownership plan (ESOP).",
"label": "Shares Granted, Value, Share-basedBased Payment Arrangement, before Forfeiture",
"terseLabel": "Granted in period"
}
}
},
"localname": "StockGrantedDuringPeriodValueSharebasedCompensationGross",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockIssued1": {
"auth_ref": [
"r106r49",
"r107r50",
"r108r51"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The fair value of stock issued in noncash financing activities.",
"label": "Stock Issued",
"terseLabel": "Issuance of stock upon exercise of Legacy Pear warrants"
}
}
},
"localname": "StockIssued1",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockIssuedDuringPeriodSharesAcquisitions": {
"auth_ref": [
"r27r3",
"r28r4",
"r340r91"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares of stock issued during the period pursuant to acquisitions.",
"label": "Stock Issued During Period, Shares, Acquisitions",
"terseLabel": "Issuance of common stock in connection with Business Combination and PIPE offering (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesAcquisitions",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsBUSINESSCOMBINATIONDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockIssuedDuringPeriodSharesConversionOfConvertibleSecurities": {
"auth_ref": [
"r55r22",
"r291r86",
"r333r87",
"r334r91",
"r340r363"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares issued during the period as a result of the conversion of convertible securities.",
"label": "Stock Issued During Period, Shares, Conversion of Convertible Securities",
"terseLabel": "Conversion of convertible preferred stock to common stock (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesConversionOfConvertibleSecurities",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockIssuedDuringPeriodSharesEmployeeStockPurchasePlans": {
"auth_ref": [
"r3",
"r4",
"r86",
"r91"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares issued during the period as a result of an employee stock purchase plan.",
"label": "Stock Issued During Period, Shares, Employee Stock Purchase Plans",
"terseLabel": "Issuance of common stock under employee stock purchase plan (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesEmployeeStockPurchasePlans",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockIssuedDuringPeriodSharesStockOptionsExercised": {
"auth_ref": [
"r27r3",
"r28r4",
"r333r86",
"r340r91",
"r400r442"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of share options (or share units) exercised during the current period.",
"label": "Share-basedBased Compensation Arrangement by Share-basedBased Payment Award, Options, Exercises in Period",
"negatedTerseLabel": "Exercised (in shares)",
"terseLabel": "Exercise of common stock options (in shares)"
}
}
},
"localname": "StockIssuedDuringPeriodSharesStockOptionsExercised",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITSTOCKBASEDCOMPENSATIONCommonStockOptionsDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONCommonStockOptionsDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockIssuedDuringPeriodValueConversionOfConvertibleSecurities": {
"auth_ref": [
"r56r23",
"r333r86",
"r340r91"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "The gross value of stock issued during the period upon the conversion of convertible securities.",
"label": "Stock Issued During Period, Value, Conversion of Convertible Securities",
"terseLabel": "Conversion of convertible preferred stock to common stock"
}
}
},
"localname": "StockIssuedDuringPeriodValueConversionOfConvertibleSecurities",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockIssuedDuringPeriodValueEmployeeStockPurchasePlan": {
"auth_ref": [
"r3",
"r4",
"r86",
"r91"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Aggregate change in value for stock issued during the period as a result of employee stock purchase plan.",
"label": "Stock Issued During Period, Value, Employee Stock Purchase Plan",
"terseLabel": "Issuance of common stock under employee stock purchase plan"
}
}
},
"localname": "StockIssuedDuringPeriodValueEmployeeStockPurchasePlan",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockIssuedDuringPeriodValueStockOptionsExercised": {
"auth_ref": [
"r56r23",
"r333r86",
"r340r91"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Value of stock issued as a result of the exercise of stock options.",
"label": "Stock Issued During Period, Value, Stock Options Exercised",
"terseLabel": "Exercise of common stock options"
}
}
},
"localname": "StockIssuedDuringPeriodValueStockOptionsExercised",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockRepurchasedDuringPeriodShares": {
"auth_ref": [
"r27",
"r28",
"r333",
"r340"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of shares that have been repurchased during the period and have not been retired and are not held in treasury. Some state laws may govern the circumstances under which an entity may acquire its own stock and prescribe the accounting treatment therefore. This element is used when state law does not recognize treasury stock.",
"label": "Stock Repurchased During Period, Shares",
"negatedTerseLabel": "Repurchase of preferred and common shares (in shares)",
"terseLabel": "Shares repurchased (in shares)"
}
}
},
"localname": "StockRepurchasedDuringPeriodShares",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_StockRepurchasedDuringPeriodValue": {
"auth_ref": [
"r27",
"r28",
"r333",
"r340"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Equity impact of the value of stock that has been repurchased during the period and has not been retired and is not held in treasury. Some state laws may mandate the circumstances under which an entity may acquire its own stock and prescribe the accounting treatment therefore. This element is used when state law does not recognize treasury stock.",
"label": "Stock Repurchased During Period, Value",
"negatedTerseLabel": "Repurchase of preferred and common shares",
"terseLabel": "Shares repurchased"
}
}
},
"localname": "StockRepurchasedDuringPeriodValue",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",
"http://www.peartherapeutics.com/role/RELATEDPARTYTRANSACTIONSDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONStockBasedCompensationExpenseDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockholdersEquity": {
"auth_ref": [
"r282022",
"presentation": [
"http://www.peartherapeutics.com/role/UNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockholdersEquity": {
"auth_ref": [
"r4",
"r33r7",
"r34r8",
"r117r63",
"r210r679",
"r221r714",
"r525r727",
"r568r777"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS": {
"order": 43.0,
"parentTag": "us-gaap_LiabilitiesAndStockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Total of all stockholders' equity (deficit) items, net of receivables from officers, directors, owners, and affiliates of the entity which are attributable to the parent. The amount of the economic entity's stockholders' equity attributable to the parent excludes the amount of stockholders' equity which is allocable to that ownership interest in subsidiary equity which is not attributable to the parent (noncontrolling interest, minority interest). This excludes temporary equity and is sometimes called permanent equity.",
"label": "Stockholders' Equity Attributable to Parent",
"periodEndLabel": "Ending balance",
"periodStartLabel": "Beginning balance",
"totalLabel": "Total stockholders\u2019 equity"
}
}
},
"localname": "StockholdersEquity",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICITUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFSTOCKHOLDERSEQUITY"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_StockholdersEquityAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Stockholders' Equity Attributable to Parent [Abstract]",
"verboseLabel": "Stockholders\u2019 equity:"
}
}
},
"localname": "StockholdersEquityAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITIONUNAUDITEDCONDENSEDCONSOLIDATEDBALANCESHEETS"
],
"xbrltype": "stringItemType"
},
"us-gaap_StockholdersEquityNoteDisclosureTextBlock": {
"auth_ref": [
"r115r93",
"r317r225",
"r321r377",
"r322r380",
"r323r381",
"r324r382",
"r325r383",
"r326r384",
"r327r385",
"r328r386",
"r329r387",
"r330r388",
"r332r389",
"r340r391",
"r342r394",
"r511"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for shareholders' equity comprised of portions attributable to the parent entity and noncontrolling interest, including other comprehensive income. Includes, but is not limited to, balances of common stock, preferred stock, additional paid-in capital, other capital and retained earnings, accumulated balance for each classification of other comprehensive income and amount of comprehensive income.",
"label": "Stockholders' Equity Note Disclosure [Text Block]",
"terseLabel": "CAPITAL STOCK"
}
}
},
"localname": "StockholdersEquityNoteDisclosureTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCK"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SubsequentEventMember": {
"auth_ref": [
"r541r544",
"r570"
],
"lang": {
"en-us": {
"role": {
"documentation": "Identifies event that occurred after the balance sheet date but before financial statements are issued or available to be issued.",
"label": "Subsequent Event [Member]",
"terseLabel": "Subsequent Event"
}
}
},
"localname": "SubsequentEventMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/LEASEDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SubsequentEventTypeAxis": {
"auth_ref": [
"r541r544",
"r570"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by event that occurred after the balance sheet date but before financial statements are issued or available to be issued.",
"label": "Subsequent Event Type [Axis]",
"terseLabel": "Subsequent Event Type [Axis]"
}
}
},
"localname": "SubsequentEventTypeAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/LEASEDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_SubsequentEventTypeDomain": {
"auth_ref": [
"r541r544",
"r570"
],
"lang": {
"en-us": {
"role": {
"documentation": "Event that occurred after the balance sheet date but before financial statements are issued or available to be issued.",
"label": "Subsequent Event Type [Domain]",
"terseLabel": "Subsequent Event Type [Domain]"
}
}
},
"localname": "SubsequentEventTypeDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsCAPITALSTOCKDetails",
"http://www.peartherapeutics.com/role/LEASEDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONESPPDetails",
"http://www.peartherapeutics.com/role/STOCKBASEDCOMPENSATIONNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_SubsequentEventsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Subsequent Events [Abstract]"
}
}
},
"localname": "SubsequentEventsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"xbrltype": "stringItemType"
},
"us-gaap_SubsequentEventsTextBlock": {
"auth_ref": [
"r569",
"r572r571"
],
"lang": {
"en-us": {
"role": {
"documentation": "The entire disclosure for significant events or transactions that occurred after the balance sheet date through the date the financial statements were issued or the date the financial statements were available to be issued. Examples include: the sale of a capital stock issue, purchase of a business, settlement of litigation, catastrophic loss, significant foreign exchange rate changes, loans to insiders or affiliates, and transactions not in the ordinary course of business.",
"label": "Subsequent Events [Text Block]",
"terseLabel": "SUBSEQUENT EVENTS"
}
}
},
"localname": "SubsequentEventsTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUBSEQUENTEVENTS"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_SubsidiaryOrEquityMethodInvesteeSaleOfStockBySubsidiaryOrEquityInvesteeTable": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Different names of stock transactions and the different attributes of each transaction.",
"label": "Subsidiary or Equity Method Investee, Sale of Stock by Subsidiary or Equity Investee [Table]",
"terseLabel": "Subsidiary or Equity Method Investee, Sale of Stock by Subsidiary or Equity Investee [Table]"
}
}
},
"localname": "SubsidiaryOrEquityMethodInvesteeSaleOfStockBySubsidiaryOrEquityInvesteeTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_SubsidiarySaleOfStockAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of sale of the entity's stock.",
"label": "Sale of Stock [Axis]",
"terseLabel": "Sale of Stock [Axis]"
}
}
},
"localname": "SubsidiarySaleOfStockAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKWarrantsDetails",
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_SubsidiarySaleOfStockLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Subsidiary, Sale of Stock [Line Items]",
"terseLabel": "Subsidiary, Sale of Stock [Line Items]"
}
}
},
"localname": "SubsidiarySaleOfStockLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSOrganizationDetailsBUSINESSCOMBINATIONDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_SupplementalCashFlowInformationAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Supplemental Cash Flow Information [Abstract]",
"terseLabel": "Supplemental disclosure of cash flow information:"
}
}
},
"localname": "SupplementalCashFlowInformationAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCASHFLOWSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFCASHFLOWS"
],
"xbrltype": "stringItemType"
},
"us-gaap_TaxCreditCarryforwardAmount": {
"auth_ref": [
"r448r115"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The amount of the tax credit carryforward, before tax effects, available to reduce future taxable income under enacted tax laws.",
"label": "Tax Credit Carryforward, Amount",
"terseLabel": "Tax credit carryforwards"
}
}
},
"localname": "TaxCreditCarryforwardAmount",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TaxCreditCarryforwardAxis": {
"auth_ref": [
"r449r116"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by specific tax credit related to an unused tax credit.",
"label": "Tax Credit Carryforward [Axis]",
"terseLabel": "Tax Credit Carryforward [Axis]"
}
}
},
"localname": "TaxCreditCarryforwardAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_TaxCreditCarryforwardNameDomain": {
"auth_ref": [
"r449r116"
],
"lang": {
"en-us": {
"role": {
"documentation": "The name of the tax credit carryforward.",
"label": "Tax Credit Carryforward, Name [Domain]",
"terseLabel": "Tax Credit Carryforward, Name [Domain]"
}
}
},
"localname": "TaxCreditCarryforwardNameDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_TemporaryEquityCarryingAmountAttributableToParent": {
"auth_ref": [
"r261",
"r265",
"r266",
"r267",
"r270",
"r271"
],
"calculation": {
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION": {
"order": 2.0,
"parentTag": "us-gaap_LiabilitiesAndStockholdersEquity",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Carrying amount, attributable to parent, of an entity's issued and outstanding stock which is not included within permanent equity. Temporary equity is a security with redemption features that are outside the control of the issuer, is not classified as an asset or liability in conformity with GAAP, and is not mandatorily redeemable. Includes any type of security that is redeemable at a fixed or determinable price or on a fixed or determinable date or dates, is redeemable at the option of the holder, or has conditions for redemption which are not solely within the control of the issuer. Includes stock with a put option held by an ESOP and stock redeemable by a holder only in the event of a change in control of the issuer.",
"label": "Temporary Equity, Carrying Amount, Attributable to Parent",
"periodEndLabel": "Ending Balance",
"periodStartLabel": "Beginning Balance",
"terseLabel": "Carrying Value",
"verboseLabel": "Legacy convertible preferred stock (Note 10)"
}
}
},
"localname": "TemporaryEquityCarryingAmountAttributableToParent",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFFINANCIALCONDITION"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_TemporaryEquityLiquidationPreferencePerShare": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "The per share liquidation preference (or restrictions) of stock classified as temporary equity that has a preference in involuntary liquidation considerably in excess of the par or stated value of the shares. The liquidation preference is the difference between the preference in liquidation and the par or stated values of the share. Temporary equity is a security with redemption features that are outside the control of the issuer, is not classified as an asset or liability in conformity with GAAP, and is not mandatorily redeemable. Includes any type of security that is redeemable at a fixed or determinable price or on a fixed or determinable date or dates, is redeemable at the option of the holder, or has conditions for redemption which are not solely within the control of the issuer. If convertible, the issuer does not control the actions or events necessary to issue the maximum number of shares that could be required to be delivered under the conversion option if the holder exercises the option to convert the stock to another class of equity. If the security is a warrant or a rights issue, the warrant or rights issue is considered to be temporary equity if the issuer cannot demonstrate that it would be able to deliver upon the exercise of the option by the holder in all cases. Includes stock with put option held by ESOP and stock redeemable by holder only in the event of a change in control of the issuer.",
"label": "Temporary Equity, Liquidation Preference Per Share",
"terseLabel": "Conversion Price/Share (in dollars per share)"
}
}
},
"localname": "TemporaryEquityLiquidationPreferencePerShare",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKAdditionalInformationDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_TemporaryEquityParOrStatedValuePerShare": {
"auth_ref": [
"r15",
"r313"
],
"lang": {
"en-us": {
"role": {
"documentation": "Per share amount of par value or stated value of stock classified as temporary equity. Temporary equity is a security with redemption features that are outside the control of the issuer, is not classified as an asset or liability in conformity with GAAP, and is not mandatorily redeemable.",
"label": "Temporary Equity, Par or Stated Value Per Share",
"terseLabel": "Par Value (in dollars per share)"
}
}
},
"localname": "TemporaryEquityParOrStatedValuePerShare",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails"
],
"xbrltype": "perShareItemType"
},
"us-gaap_TemporaryEquitySharesAuthorized": {
"auth_ref": [
"r25"
],
"lang": {
"en-us": {
"role": {
"documentation": "The maximum number of securities classified as temporary equity that are permitted to be issued by an entity's charter and bylaws. Temporary equity is a security with redemption features that are outside the control of the issuer, is not classified as an asset or liability in conformity with GAAP, and is not mandatorily redeemable. Includes any type of security that is redeemable at a fixed or determinable price or on a fixed or determinable date or dates, is redeemable at the option of the holder, or has conditions for redemption which are not solely within the control of the issuer. If convertible, the issuer does not control the actions or events necessary to issue the maximum number of shares that could be required to be delivered under the conversion option if the holder exercises the option to convert the stock to another class of equity. If the security is a warrant or a rights issue, the warrant or rights issue is considered to be temporary equity if the issuer cannot demonstrate that it would be able to deliver upon the exercise of the option by the holder in all cases. Includes stock with put option held by ESOP and stock redeemable by holder only in the event of a change in control of the issuer.",
"label": "Temporary Equity, Shares Authorized",
"terseLabel": "Authorized (in shares)"
}
}
},
"localname": "TemporaryEquitySharesAuthorized",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_TemporaryEquitySharesIssued": {
"auth_ref": [
"r25"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of securities classified as temporary equity that have been sold (or granted) to the entity's shareholders. Securities issued include securities outstanding and securities held in treasury. Temporary equity is a security with redemption features that are outside the control of the issuer, is not classified as an asset or liability in conformity with GAAP, and is not mandatorily redeemable. Includes any type of security that is redeemable at a fixed or determinable price or on a fixed or determinable date or dates, is redeemable at the option of the holder, or has conditions for redemption which are not solely within the control of the issuer. If convertible, the issuer does not control the actions or events necessary to issue the maximum number of shares that could be required to be delivered under the conversion option if the holder exercises the option to convert the stock to another class of equity. If the security is a warrant or a rights issue, the warrant or rights issue is considered to be temporary equity if the issuer cannot demonstrate that it would be able to deliver upon the exercise of the option by the holder in all cases. Includes stock with put option held by ESOP and stock redeemable by holder only in the event of a change in control of the issuer.",
"label": "Temporary Equity, Shares Issued",
"terseLabel": "Issued (in shares)"
}
}
},
"localname": "TemporaryEquitySharesIssued",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKConvertiblePreferredStockIssuanceDetails",
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails"
],
"xbrltype": "sharesItemType"
},
"us-gaap_TemporaryEquitySharesOutstanding": {
"auth_ref": [
"r25"
],
"lang": {
"en-us": {
"role": {
"documentation": "The number of securities classified as temporary equity that have been issued and are held by the entity's shareholders. Securities outstanding equals securities issued minus securities held in treasury. Temporary equity is a security with redemption features that are outside the control of the issuer, is not classified as an asset or liability in conformity with GAAP, and is not mandatorily redeemable. Includes any type of security that is redeemable at a fixed or determinable price or on a fixed or determinable date or dates, is redeemable at the option of the holder, or has conditions for redemption which are not solely within the control of the issuer. If convertible, the issuer does not control the actions or events necessary to issue the maximum number of shares that could be required to be delivered under the conversion option if the holder exercises the option to convert the stock to another class of equity. If the security is a warrant or a rights issue, the warrant or rights issue is considered to be temporary equity if the issuer cannot demonstrate that it would be able to deliver upon the exercise of the option by the holder in all cases. Includes stock with put option held by ESOP and stock redeemable by holder only in the event of a change in control of the issuer.",
"label": "Temporary Equity, Shares Outstanding",
"periodEndLabel": "Ending Balance (in shares)",
"periodStartLabel": "Beginning Balance (in shares)",
"terseLabel": "Outstanding (in shares)"
}
}
},
"localname": "TemporaryEquitySharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKScheduleofConvertibleStockDetails",
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFCONVERTIBLEPREFERREDSTOCKANDSTOCKHOLDERSDEFICIT"
],
"xbrltype": "sharesItemType"
},
"us-gaap_TemporaryEquityTableTextBlock": {
"auth_ref": [
"r15",
"r313"
],
"lang": {
"en-us": {
"role": {
"documentation": "Tabular disclosure of temporary equity. Temporary equity is a security with redemption features that are outside the control of the issuer, is not classified as an asset or liability in conformity with GAAP, and is not mandatorily redeemable. Includes any type of security that is redeemable at a fixed or determinable price or on a fixed or determinable date or dates, is redeemable at the option of the holder, or has conditions for redemption which are not solely within the control of the issuer. If convertible, the issuer does not control the actions or events necessary to issue the maximum number of shares that could be required to be delivered under the conversion option if the holder exercises the option to convert the stock to another class of equity. If the security is a warrant or a rights issue, the warrant or rights issue is considered to be temporary equity if the issuer cannot demonstrate that it would be able to deliver upon the exercise of the option by the holder in all cases. Includes stock with put option held by ESOP and stock redeemable by holder only in the event of a change in control of the issuer.",
"label": "Temporary Equity [Table Text Block]",
"terseLabel": "Schedule of Convertible Preferred Stock"
}
}
},
"localname": "TemporaryEquityTableTextBlock",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/CAPITALSTOCKTables"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_TransfersAndServicingOfFinancialInstrumentsTypesOfFinancialInstrumentsDomain": {
"auth_ref": [
"r2162022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_TransfersAndServicingOfFinancialInstrumentsTypesOfFinancialInstrumentsDomain": {
"auth_ref": [
"r300",
"r217r301",
"r218r371",
"r219r390",
"r220r510",
"r299",
"r331",
"r499r572",
"r573",
"r574",
"r575",
"r576",
"r577",
"r578",
"r579",
"r580",
"r581",
"r582",
"r583",
"r584",
"r585",
"r586",
"r587",
"r588",
"r589",
"r590",
"r591",
"r592",
"r593",
"r594",
"r595",
"r596",
"r597",
"r598",
"r599",
"r600",
"r601",
"r602r724",
"r703r725",
"r704r726",
"r705r797",
"r706r798",
"r707r799",
"r708r800",
"r709r801",
"r802",
"r803"
],
"lang": {
"en-us": {
"role": {
"documentation": "Instrument or contract that imposes a contractual obligation to deliver cash or another financial instrument or to exchange other financial instruments on potentially unfavorable terms and conveys a contractual right to receive cash or another financial instrument or to exchange other financial instruments on potentially favorable terms.",
"label": "Financial Instruments [Domain]",
"terseLabel": "Financial Instruments [Domain]"
}
}
},
"localname": "TransfersAndServicingOfFinancialInstrumentsTypesOfFinancialInstrumentsDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_TypeOfAdoptionMember": {
"auth_ref": [
"r0r184",
"r1r185",
"r2r186",
"r3r187",
"r4r188",
"r129r237",
"r130r238",
"r131r239",
"r132r240",
"r144r249",
"r211r297",
"r212r298",
"r222r303",
"r223r304",
"r224r305",
"r225r306",
"r226r307",
"r227r308",
"r260r467",
"r417r468",
"r418r469",
"r419r486",
"r420r487",
"r421r488",
"r422",
"r423",
"r424",
"r453",
"r454",
"r455",
"r456r489",
"r500",
"r501",
"r502",
"r503r512",
"r504r513",
"r505r514",
"r506r515",
"r507r516",
"r508r517",
"r509r518",
"r510r519",
"r526r520",
"r527r521",
"r528r522",
"r529r523",
"r530",
"r531",
"r532",
"r533",
"r557r534",
"r535",
"r536",
"r537",
"r545",
"r546",
"r549",
"r550",
"r551",
"r552",
"r563",
"r564",
"r565",
"r566",
"r567",
"r568",
"r606",
"r607",
"r608",
"r609r618",
"r663r619",
"r664r620",
"r665r621",
"r666r622",
"r667r623",
"r668r624",
"r669r625",
"r670r626",
"r671r627",
"r672r628",
"r673",
"r674",
"r713",
"r714",
"r715",
"r716",
"r717r629"
],
"lang": {
"en-us": {
"role": {
"documentation": "Amendment to accounting standards.",
"label": "Accounting Standards Update [Domain]",
"terseLabel": "Accounting Standards Update [Domain]"
}
}
},
"localname": "TypeOfAdoptionMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNewAccountingPronouncementsDetailsSUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_TypeOfArrangementAxis": {
"auth_ref": [
"r478r773"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by collaborative arrangement and arrangement other than collaborative applicable to revenue-generating activity or operations.",
"label": "Collaborative Arrangement and Arrangement Other than Collaborative [Axis]",
"terseLabel": "Collaborative Arrangement and Arrangement Other than Collaborative [Axis]"
}
}
},
"localname": "TypeOfArrangementAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/PASTCOLLABORATIONANDLICENSEAGREEMENTNovartisDetailsREVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_UnamortizedDebtIssuanceExpensegaap_USTreasurySecuritiesMember": {
"auth_ref": [
"r42r656",
"r670",
"r672",
"r793"
],
"calculation": {
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails": {
"order": 4.0,
"parentTag": "us-gaap_LongTermDebt",
"weight": -1.0
}
}lang": {
"en-us": {
"role": {
"documentation": "This category includes information about debt securities issued by the United States Department of the Treasury and backed by the United States government. Such securities primarily consist of treasury bills (short-term maturities - one year or less), treasury notes (intermediate term maturities - two to ten years), and treasury bonds (long-term maturities - ten to thirty years).",
"label": "US Treasury Securities [Member]",
"terseLabel": "US Treasury bills"
}
}
},
"localname": "USTreasurySecuritiesMember",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_UnamortizedDebtIssuanceExpense": {
"auth_ref": [],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "The remaining balance of debt issuance expenses that were capitalized and are being amortized against income over the lives of the respective bond issues. This does not include the amounts capitalized as part of the cost of the utility plant or asset.",
"label": "Unamortized Debt Issuance Expense",
"negatedTerseLabel": "Unamortized debt issuance costs"
}
}
},
"localname": "UnamortizedDebtIssuanceExpense",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSFutureMinimumPaymentsDueDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnusualOrInfrequentItemAxis": {
"auth_ref": [
"r86gaap_UnbilledContractsReceivable": {
"auth_ref": [
"r604"
],
"crdr": "debit",
"lang": {
"en-us": {
"role": {
"documentation": "Unbilled amounts due for services rendered or to be rendered, actions taken or to be taken, or a promise to refrain from taking certain actions in accordance with the terms of a legally binding agreement between the entity and, at a minimum, one other party. An example would be amounts associated with contracts or programs where the recognized revenue for performance thereunder exceeds the amounts billed under the terms thereof as of the date of the balance sheet.",
"label": "Unbilled Contracts Receivable",
"terseLabel": "Unbilled receivables"
}
}
},
"localname": "UnbilledContractsReceivable",
"nsuri": "http://fasb.org/us-gaap/2022",
"presentation": [
"http://www.peartherapeutics.com/role/REVENUEANDCONTRACTBALANCESDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_UnusualOrInfrequentItemAxis": {
"auth_ref": [
"r36"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by an event or transaction that is unusual in nature or infrequent in occurrence, or both.",
"label": "Unusual or Infrequent Item, or Both [Axis]",
"terseLabel": "Unusual or Infrequent Item, or Both [Axis]"
}
}
},
"localname": "UnusualOrInfrequentItemAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_UnusualOrInfrequentItemDomain": {
"auth_ref": [
"r86r36"
],
"lang": {
"en-us": {
"role": {
"documentation": "Event or transaction that is unusual in nature or infrequent in occurrence, or both.",
"label": "Unusual or Infrequent Item, or Both [Domain]",
"terseLabel": "Unusual or Infrequent Item, or Both [Domain]"
}
}
},
"localname": "UnusualOrInfrequentItemDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_UnusualOrInfrequentItemLineItems": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Line items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.",
"label": "Unusual or Infrequent Item, or Both [Line Items]",
"terseLabel": "Unusual or Infrequent Item, or Both [Line Items]"
}
}
},
"localname": "UnusualOrInfrequentItemLineItems",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_UnusualOrInfrequentItemTable": {
"auth_ref": [
"r86r36"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of information about the nature and financial statement effect of an event or transaction that is unusual in nature or infrequent in occurrence, or both.",
"label": "Unusual or Infrequent Item, or Both [Table]",
"terseLabel": "Unusual or Infrequent Item, or Both [Table]"
}
}
},
"localname": "UnusualOrInfrequentItemTable",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/NATUREOFBUSINESSNarrativeDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_UseOfEstimates": {
"auth_ref": [
"r170r60",
"r171r61",
"r173r62",
"r174r268",
"r180r269",
"r181r271",
"r182r272"
],
"lang": {
"en-us": {
"role": {
"documentation": "Disclosure of accounting policy for the use of estimates in the preparation of financial statements in conformity with generally accepted accounting principles.",
"label": "Use of Estimates, Policy [Policy Text Block]",
"terseLabel": "Use of Estimates"
}
}
},
"localname": "UseOfEstimates",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/SUMMARYOFSIGNIFICANTACCOUNTINGPOLICIESPolicies"
],
"xbrltype": "textBlockItemType"
},
"us-gaap_ValuationAllowanceByDeferredTaxAssetAxis": {
"auth_ref": [
"r444r113"
],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of deferred tax consequences attributable to deductible temporary differences.",
"label": "Valuation Allowance by Deferred Tax Asset [Axis]",
"terseLabel": "Valuation Allowance by Deferred Tax Asset [Axis]"
}
}
},
"localname": "ValuationAllowanceByDeferredTaxAssetAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_ValuationAllowanceDeferredTaxAssetChangeInAmount": {
"auth_ref": [
"r445r482"
],
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Amount of increase (decrease) in the valuation allowance for a specified deferred tax asset.",
"label": "Valuation Allowance, Deferred Tax Asset, Increase (Decrease), Amount",
"terseLabel": "Increase in valuation allowance"
}
}
},
"localname": "ValuationAllowanceDeferredTaxAssetChangeInAmount",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INCOMETAXESAdditionalInformationDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_VariableRateAxis": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Information by type of variable rate.",
"label": "Variable Rate [Axis]",
"terseLabel": "Variable Rate [Axis]"
}
}
},
"localname": "VariableRateAxis",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "stringItemType"
},
"us-gaap_VariableRateDomain": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Interest rate that fluctuates over time as a result of an underlying benchmark interest rate or index.",
"label": "Variable Rate [Domain]",
"terseLabel": "Variable Rate [Domain]"
}
}
},
"localname": "VariableRateDomain",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_WarrantMember": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"documentation": "Security that gives the holder the right to purchase shares of stock in accordance with the terms of the instrument, usually upon payment of a specified amount.",
"label": "Warrant [Member]",
"terseLabel": "Warrants to purchase Legacy Pear common stock",
"verboseLabel": "Warrants"
}
}
},
"localname": "WarrantMember",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/Cover",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSLevel3RollForwardDetails",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREAntidilituveSecuritiesDetailsNETLOSSPERSHAREAntidilutiveSecuritiesDetails"
],
"xbrltype": "domainItemType"
},
"us-gaap_WarrantsAndRightsOutstanding": {
"auth_ref": [],
"calculation": {
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails": {
"order": 2.0,
"parentTag": "us-gaap_FinancialLiabilitiesFairValueDisclosure",
"weight": 1.0
}
},
"crdr": "credit",
"lang": {
"en-us": {
"role": {
"documentation": "Value of outstanding derivative securities that permit the holder the right to purchase securities (usually equity) from the issuer at a specified price.",
"label": "Warrants and Rights Outstanding",
"terseLabel": "Warrant liabilities"
}
}
},
"localname": "WarrantsAndRightsOutstanding",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/BUSINESSCOMBINATIONWarrantsDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSAssetsandFairValueDetails",
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSNarrativeDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSPerceptiveCreditFacilityDetails",
"http://www.peartherapeutics.com/role/INDEBTEDNESSSVBTermLoanDetails"
],
"xbrltype": "monetaryItemType"
},
"us-gaap_WarrantsAndRightsOutstandingMeasurementInput": {
"auth_ref": [
"r514"
],
"lang": {
"en-us": {
"role": {
"documentation": "Value of input used to measure outstanding warrant and right embodying unconditional obligation requiring redemption by transferring asset at specified or determinable date or upon event certain to occur.",
"label": "Warrants and Rights Outstanding, Measurement Input",
"terseLabel": "Warrants"
}
}
},
"localname": "WarrantsAndRightsOutstandingMeasurementInput",
"nsuri": "http://fasb.org/us-gaap/2021-01-31",
"presentation": [
"http://www.peartherapeutics.com/role/FAIRVALUEMEASUREMENTSValuationAssumptionsDetails"
],
"xbrltype": "decimalItemType
],
"xbrltype": "monetaryItemType"
},
"us-gaap_WeightedAverageNumberOfDilutedSharesOutstanding": {
"auth_ref": [
"r147r252",
"r159r259"
],
"lang": {
"en-us": {
"role": {
"documentation": "The average number of shares or units issued and outstanding that are used in calculating diluted EPS or earnings per unit (EPU), determined based on the timing of issuance of shares or units in the period.",
"label": "Weighted Average Number of Shares Outstanding, Diluted",
"terseLabel": "Weighted-average common shares outstanding, diluted (in shares)"
}
}
},
"localname": "WeightedAverageNumberOfDilutedSharesOutstanding",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSS",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "sharesItemType"
},
"us-gaap_WeightedAverageNumberOfSharesOutstandingBasic": {
"auth_ref": [
"r145r250",
"r159r259"
],
"lang": {
"en-us": {
"role": {
"documentation": "Number of [basic] shares or units, after adjustment for contingently issuable shares or units and other shares or units not deemed outstanding, determined by relating the portion of time within a reporting period that common shares or units have been outstanding to the total time in that period.",
"label": "Weighted Average Number of Shares Outstanding, Basic",
"terseLabel": "Weighted-average common shares outstanding, basic (in shares)"
}
}
},
"localname": "WeightedAverageNumberOfSharesOutstandingBasic",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSNETLOSSPERSHAREBasicandDilutedPerShareDetails",
"http://www.peartherapeutics.com/role/NETLOSSPERSHAREBasicandDilutedPerShareDetailsUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "sharesItemType"
},
"us-gaap_WeightedAverageNumberOfSharesOutstandingDilutedDisclosureItemsAbstract": {
"auth_ref": [],
"lang": {
"en-us": {
"role": {
"label": "Weighted Average Number of Shares Outstanding Reconciliation [Abstract]",
"terseLabel": "Weighted average common shares outstanding:"
}
}
},
"localname": "WeightedAverageNumberOfSharesOutstandingDilutedDisclosureItemsAbstract",
"nsuri": "http://fasb.org/us-gaap/2021-01-312022",
"presentation": [
"http://www.peartherapeutics.com/role/CONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVELOSSUNAUDITEDCONDENSEDCONSOLIDATEDSTATEMENTSOFOPERATIONSANDCOMPREHENSIVEINCOMELOSS"
],
"xbrltype": "stringItemType"
}
},
"unitCount": 10
}
},
"std_ref": {
"r0": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "105",
"URI": "http12
}
},
"std_ref": {
"r0": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "205",
"URI": "https://asc.fasb.org/topic&trid=2122149",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r1": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(22))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r10": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19(a))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r100": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r101": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r102": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r103": {
"Name": "Accounting Standards Codification",
"Paragraph": "2A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=SL79508275-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r104": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 14.F)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=122041274&loc=d3e301413-122809",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r105": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=126964447&loc=d3e11149-113907",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r106": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=126964447&loc=d3e11178-113907",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r107": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "35",
"Subparagraph": "(a)",
"Topic": "720",
"URI": "https://asc.fasb.org/extlink&oid=6420018&loc=d3e36677-107848",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r108": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "730",
"URI": "https://asc.fasb.org/extlink&oid=6420194&loc=d3e21568-108373",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r109": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "730",
"URI": "https://asc.fasb.org/extlink&oid=6420387&loc=d3e23199-108380",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r11": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19(b),22(b))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r110": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "730",
"URI": "https://asc.fasb.org/extlink&oid=6420387&loc=d3e23221-108380",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r111": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32687-109319",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r112": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32718-109319",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r113": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r114": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r115": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32559-109319",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r116": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32559-109319",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r117": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32632-109319",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r118": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=79982066&loc=d3e1392-128463",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r119": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=79982066&loc=d3e1486-128463",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r12": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19,20)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r120": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "30",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=6911189&loc=d3e6408-128476",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r121": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=126966325&loc=d3e6819-128478",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r122": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "30",
"Subparagraph": "b",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=126966325&loc=d3e6819-128478",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r123": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(b)(4)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=126975305&loc=d3e6927-128479",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r124": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "805",
"URI": "https://asc.fasb.org/topic&trid=2303972",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r125": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=126929396&loc=SL4569616-111683",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r126": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=126929396&loc=SL4569643-111683",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r127": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=109239629&loc=d3e5614-111684",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r128": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(3)",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r129": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c),(3)",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r13": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19-26)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r130": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "810",
"URI": "https://asc.fasb.org/topic&trid=2197479",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r131": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=SL5579240-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r132": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=SL5579245-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r133": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=d3e41620-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r134": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=d3e41638-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r135": {
"Name": "Accounting Standards Codification",
"Paragraph": "4A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=SL5618551-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r136": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=SL5624163-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r137": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=SL5624163-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r138": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=125515794&loc=d3e41675-113959",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r139": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r14": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.20)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r140": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r141": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r142": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r143": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19279-110258",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r144": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=123594938&loc=d3e13467-108611",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r145": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=123594938&loc=d3e13476-108611",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r146": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=123594938&loc=d3e13531-108611",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r147": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=123594938&loc=d3e13537-108611",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r148": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "230",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=123444420&loc=d3e33268-110906",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r149": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "https://asc.fasb.org/extlink&oid=124435984&loc=d3e28541-108399",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r15": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.21)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r150": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "https://asc.fasb.org/extlink&oid=124435984&loc=d3e28551-108399",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r151": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "https://asc.fasb.org/extlink&oid=124435984&loc=d3e28555-108399",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r152": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "30",
"Topic": "835",
"URI": "https://asc.fasb.org/extlink&oid=114775985&loc=d3e28878-108400",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r153": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(1)(a))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r154": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(11))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r155": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(16))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r156": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(17))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r157": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(23))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r158": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(4))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r159": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(5))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r16": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.22(a)(1))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r160": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.15(5))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r161": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.17)",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126897435&loc=d3e534808-122878",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r162": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(22))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r163": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04.10)",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r164": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126980459&loc=d3e62652-112803",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r165": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "470",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=123599511&loc=d3e64711-112823",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r166": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "825",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126941378&loc=d3e61044-112788",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r167": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(16))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r168": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(1)(g))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r169": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(12))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r17": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.22(b))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r170": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(16))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r171": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(19))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r172": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(2))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r173": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(23)(a)(4))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r174": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(25))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r175": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(8))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r176": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.(a),19)",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r177": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.15(a))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126734703&loc=d3e572229-122910",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r178": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(18))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r179": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "985",
"URI": "https://asc.fasb.org/extlink&oid=6501960&loc=d3e128462-111756",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r18": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.22)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r180": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(Note 3)",
"Topic": "840",
"URI": "https://asc.fasb.org/extlink&oid=123403562&loc=d3e38371-112697",
"role": "http://fasb.org/us-gaap/role/ref/otherTransitionRef"
},
"r181": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "20",
"Topic": "840",
"URI": "https://asc.fasb.org/extlink&oid=123415192&loc=d3e39896-112707",
"role": "http://fasb.org/us-gaap/role/ref/otherTransitionRef"
},
"r182": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "840",
"URI": "https://asc.fasb.org/extlink&oid=123406913&loc=d3e41502-112717",
"role": "http://fasb.org/us-gaap/role/ref/otherTransitionRef"
},
"r183": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "840",
"URI": "https://asc.fasb.org/extlink&oid=123406913&loc=d3e41502-112717",
"role": "http://fasb.org/us-gaap/role/ref/otherTransitionRef"
},
"r184": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "105",
"URI": "https://asc.fasb.org/extlink&oid=124434974126987489&loc=SL124442142-165695",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r1r185": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "105",
"URI": "httphttps://asc.fasb.org/extlink&oid=124434974126987489&loc=SL124442142-165695"
},
"r10": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r186": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "105",
"URI": "https://asc.fasb.org/extlink&oid=126987489&loc=SL124442142-165695",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r187": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "105",
"URI": "https://asc.fasb.org/extlink&oid=126987489&loc=SL124442142-165695",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r188": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "105",
"URI": "https://asc.fasb.org/extlink&oid=126987489&loc=SL124442142-165695",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r189": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "https://asc.fasb.org/extlink&oid=109222650&loc=SL51721683-107760",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r19": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.24)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r190": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=124098289&loc=d3e6676-107765",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r100": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230r191": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=124098289&loc=d3e6904-107765",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r192": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(1))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r193": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(13))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r194": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(14))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r195": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(17))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r196": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(18))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r197": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(22))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r198": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(27)(b))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r199": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(28))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r2": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(25))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r20": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.25)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r200": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(29))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r201": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(4))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r202": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(9))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r203": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126968391&loc=SL7669646-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r204": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126968391&loc=SL7669646-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r205": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126968391&loc=d3e637-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r206": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "220",
"URI": "httphttps://asc.fasb.org/extlink&oid=123570139126968391&loc=d3e3602-108585SL7669619-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r101r207": {
"Name": "Accounting Standards Codification",
"Paragraph": "281A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230220",
"URI": "httphttps://asc.fasb.org/extlink&oid=123570139126968391&loc=d3e3602-108585SL7669619-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r102r208": {
"Name": "Accounting Standards Codification",
"Paragraph": "281A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "230220",
"URI": "httphttps://asc.fasb.org/extlink&oid=123570139126968391&loc=d3e3602-108585SL7669619-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r103r209": {
"Name": "Accounting Standards Codification",
"Paragraph": "41B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230220",
"URI": "httphttps://asc.fasb.org/extlink&oid=123570139126968391&loc=d3e3044-108585"
},
"r104SL7669625-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r21": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r210": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126968391&loc=SL7669625-108580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r211": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230220",
"URI": "httphttps://asc.fasb.org/extlink&oid=123431023124431353&loc=d3e4273-108586"
},
"r105": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "httpSL116659661-227067",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r212": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=124431353&loc=SL124442407-227067",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r213": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=124431353&loc=SL124442411-227067",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r214": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=124431353&loc=SL124452729-227067",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r215": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(24))",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126953954&loc=SL114868664-224227",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r216": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(25))",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126953954&loc=SL114868664-224227",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r217": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3151-108585",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r218": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3179-108585",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r219": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3367-108585",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r22": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29-30)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r220": {
"Name": "Accounting Standards Codification",
"Paragraph": "24",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3521-108585",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r221": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3536-108585",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r222": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=123431023126999549&loc=d3e4297-108586"
},
"r106": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=123431023&loc=d3e4304-108586"
},
"r107": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123431023&loc=d3e4313-108586"
},
"r108": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123431023&loc=d3e4332-108586"
},
"r109": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123431023&loc=SL98516268-108586"
},
"r11": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=124098289&loc=d3e6801-107765"
},
"r1102003/role/disclosureRef"
},
"r223": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=123372394126899994&loc=d3e18726-107790"
},
"r111": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "235",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=123372394&loc=d3e18823-107790"
},
"r112": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "235",
"URI": "http://asc.fasb.org/extlink&oid=123372394&loc=d3e18823-107790"
},
"r113xbrl.org/2003/role/disclosureRef"
},
"r224": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(c))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r114": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(d))",
"Topic": "235",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120395691&loc=d3e23780-122690xbrl.org/2003/role/disclosureRef"
},
"r115r225": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(e)(1))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r116": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(f))",
"Topic": "235",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120395691&loc=d3e23780-122690xbrl.org/2003/role/disclosureRef"
},
"r117r226": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(g)(1)(ii))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r118r227": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(h)(2))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r119r228": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(h))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r12": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=124098289&loc=d3e6812-1077652003/role/disclosureRef"
},
"r120r229": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(m)(1)(iii))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690"
},
"r121,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r23": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29-31)",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r230": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(m)(2)(ii))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r122r231": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(n))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r123r232": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-04(a))",
"Topic": "235",
"URI": "httphttps://asc.fasb.org/extlink&oid=120395691&loc=d3e24072-122690"
},
"r124": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "235",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/topic&trid=21223692003/role/disclosureRef"
},
"r125r233": {
"Name": "Accounting Standards Codification",
"Paragraph": "23",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124436220&loc=d3e21914-107793",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r126r234": {
"Name": "Accounting Standards Codification",
"Paragraph": "24",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124436220&loc=d3e21930-107793",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r127r235": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124436220&loc=d3e21711-107793",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r128r236": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124436220&loc=d3e21728-107793",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r129r237": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22499-107794"
},
"r13": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=124098289&loc=d3e6911-1077652003/role/disclosureRef"
},
"r130r238": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(3)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22499-107794",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r131r239": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(4)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22499-107794"
},
"r132,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r24": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126968391&loc=SL7669646-108580",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r240": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22499-107794"
},
"r133": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124431687&loc=d3e22499-107794xbrl.org/2003/role/disclosureRef"
},
"r134r241": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22694-107794",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r135r242": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22694-107794"
},
"r136": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124431687&loc=SL124452830-107794xbrl.org/2003/role/disclosureRef"
},
"r137r243": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22583-107794",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r138r244": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22595-107794",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r139r245": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22644-107794"
},
"r14": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=124098289&loc=d3e6935-1077652003/role/disclosureRef"
},
"r140r246": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22644-107794"
},
"r141": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124431687&loc=d3e22644-107794xbrl.org/2003/role/disclosureRef"
},
"r142r247": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22658-107794",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r143r248": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=124431687&loc=d3e22663-107794",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r144r249": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 11.M.Q2)",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=122038215&loc=d3e31137-122693"
},
"r145,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r25": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126968391&loc=d3e637-108580",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r250": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=d3e1448-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r146r251": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=d3e1377-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r147r252": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=d3e1505-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r148r253": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=d3e1252-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r149r254": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=d3e1278-109256"
},
"r15": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(27)",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r150": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=125511455&loc=d3e1930-109256"
},
"r151": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)(1)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=125511455&loc=d3e1930-109256"
},
"r152": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=125511455&loc=d3e1930-109256"
},
"r153": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)(3)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=125511455&loc=d3e1930-109256"
},
"r154": {
"Name": "Accounting Standards Codification",
"Paragraph": "55",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "260",
"URI": "http://asc.fasb.org/extlink&oid=125511455&loc=d3e2626-109256"
},
"r155xbrl.org/2003/role/disclosureRef"
},
"r255": {
"Name": "Accounting Standards Codification",
"Paragraph": "60B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=SL5780133-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r156r256": {
"Name": "Accounting Standards Codification",
"Paragraph": "60B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=SL5780133-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r157r257": {
"Name": "Accounting Standards Codification",
"Paragraph": "60B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=SL5780133-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r158r258": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125511455126958026&loc=d3e1337-109256",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r159r259": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=124432515&loc=d3e3550-109257",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r16r26": {
"Name": "Accounting Standards Codification",
"Paragraph": "114",
"Publisher": "FASB",
"Section": "S9945",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(1))",
"Topic": "210220",
"URI": "httphttps://asc.fasb.org/extlink&oid=120391452126968391&loc=d3e13212-122682d3e681-108580",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r160r260": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=124432515&loc=d3e3550-109257",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r161r261": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=124432515&loc=d3e3630-109257",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r162r262": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=109243012&loc=SL65017193-207537",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r163r263": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/extlink&oid=125512782128363288&loc=d3e3842-109258"
},
"r164": {
"Name": "Accounting Standards Codification",
"Paragraph": "52",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "260",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=125512782&loc=d3e4984-1092582003/role/disclosureRef"
},
"r165r264": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "260",
"URI": "httphttps://asc.fasb.org/topic&trid=2144383",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r166r265": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "272",
"URI": "httphttps://asc.fasb.org/extlink&oid=125520817&loc=d3e70191-108054"
},
"r167": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "272",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=125520817&loc=d3e70229-108054xbrl.org/2003/role/disclosureRef"
},
"r168r266": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "272",
"URI": "httphttps://asc.fasb.org/extlink&oid=6373374&loc=d3e70434-108055",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r169r267": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "272",
"URI": "httphttps://asc.fasb.org/extlink&oid=6373374&loc=d3e70478-108055"
},
"r17": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(13))",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120391452&loc=d3e13212-122682xbrl.org/2003/role/disclosureRef"
},
"r170r268": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "275",
"URI": "httphttps://asc.fasb.org/extlink&oid=99393423&loc=d3e5967-108592",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r171r269": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "275",
"URI": "httphttps://asc.fasb.org/extlink&oid=99393423&loc=d3e5967-108592"
},
"r172,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r27": {
"Name": "Accounting Standards Codification",
"Paragraph": "14A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126968391&loc=SL7669686-108580",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r270": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "275",
"URI": "httphttps://asc.fasb.org/extlink&oid=99393423&loc=d3e5967-108592",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r173r271": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "httphttps://asc.fasb.org/extlink&oid=99393423&loc=d3e6161-108592",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r174r272": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "httphttps://asc.fasb.org/extlink&oid=99393423&loc=d3e6191-108592"
},
"r175": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=99393423&loc=d3e6327-108592"
},
"r176": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6351-108592"
},
"r177": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6351-108592"
},
"r178": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6404-108592"
},
"r179": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6442-108592"
},
"r18": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(14))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r180": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6061-108592"
},
"r181": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6132-108592"
},
"r182": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "http://asc.fasb.org/extlink&oid=99393423&loc=d3e6143-108592"
},
"r183": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "http2003/role/disclosureRef"
},
"r273": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "https://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8736-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r184r274": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8736-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r185r275": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8736-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r186r276": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8736-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r187r277": {
"Name": "Accounting Standards Codification",
"Paragraph": "29",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8864-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r188r278": {
"Name": "Accounting Standards Codification",
"Paragraph": "29",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8864-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r189r279": {
"Name": "Accounting Standards Codification",
"Paragraph": "29",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8864-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r19r28": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "S9945",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(17))",
"Topic": "210220",
"URI": "httphttps://asc.fasb.org/extlink&oid=120391452126968391&loc=d3e13212-122682d3e557-108580",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r190r280": {
"Name": "Accounting Standards Codification",
"Paragraph": "29",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8864-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r191r281": {
"Name": "Accounting Standards Codification",
"Paragraph": "29",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8864-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r192r282": {
"Name": "Accounting Standards Codification",
"Paragraph": "29",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8864-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r193r283": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8906-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r194r284": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8906-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r195r285": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8906-108599"
},
"r196": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "280",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=123359005&loc=d3e8906-108599"
},
"r197": {
"Name": "Accounting Standards Codification",
"Paragraph": "31",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "http://asc.fasb.org/extlink&oid=123359005&loc=d3e8924-108599"
},
"r1982003/role/disclosureRef"
},
"r286": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8933-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r199r287": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8933-108599"
},
"r2": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "105",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124434974&loc=SL124442142-165695"
},
"r20": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(19))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r200xbrl.org/2003/role/disclosureRef"
},
"r288": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8933-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r201r289": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8933-108599"
},
"r202,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r29": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(20))",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126953954&loc=SL114868664-224227",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r290": {
"Name": "Accounting Standards Codification",
"Paragraph": "32",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e8933-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r203r291": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e9031-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r204r292": {
"Name": "Accounting Standards Codification",
"Paragraph": "41",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e9038-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r205r293": {
"Name": "Accounting Standards Codification",
"Paragraph": "41",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e9038-108599",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r206r294": {
"Name": "Accounting Standards Codification",
"Paragraph": "42",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "httphttps://asc.fasb.org/extlink&oid=123359005126901519&loc=d3e9054-108599"
},
"r207": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "310",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=124259787&loc=d3e4647-1115222003/role/disclosureRef"
},
"r208r295": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "310",
"URI": "httphttps://asc.fasb.org/extlink&oid=124259787&loc=d3e4428-111522",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r209r296": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "310",
"URI": "httphttps://asc.fasb.org/extlink&oid=124259787&loc=d3e4531-111522"
},
"r21": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(2))",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r210": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 4.E)",
"Topic": "310",
"URI": "http://asc.fasb.org/extlink&oid=122038336&loc=d3e74512-122707"
},
"r211xbrl.org/2003/role/disclosureRef"
},
"r297": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "310",
"URI": "httphttps://asc.fasb.org/extlink&oid=124402435126986314&loc=SL124402458-218513",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r212r298": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "310",
"URI": "httphttps://asc.fasb.org/extlink&oid=124402435126986314&loc=SL124402458-218513",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r213r299": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "320",
"URI": "httphttps://asc.fasb.org/extlink&oid=124260329&loc=d3e26610-111562",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r214r3": {
"Name": "Accounting Standards Codification",
"Paragraph": "111",
"Publisher": "FASB",
"Section": "45S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(28))",
"Topic": "320210",
"URI": "httphttps://asc.fasb.org/extlink&oid=124260329120391452&loc=d3e26853-111562d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r215r30": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50S99",
"SubTopic": "10",
"Subparagraph": "(aaSX 210.5-03)",
"Topic": "320220",
"URI": "httphttps://asc.fasb.org/extlink&oid=123581744126953954&loc=d3e27161-111563SL114868664-224227",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r216r300": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "httphttps://asc.fasb.org/extlink&oid=123581744126970911&loc=d3e27232-111563",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r217r301": {
"Name": "Accounting Standards Codification",
"Paragraph": "5A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "320",
"URI": "httphttps://asc.fasb.org/extlink&oid=123581744126970911&loc=SL120269820-111563"
},
"r218": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "321",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=123583765&loc=SL75117539-209714"
},
"r219": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "321",
"URI": "http://asc.fasb.org/extlink&oid=123583765&loc=SL75117539-209714"
},
"r22": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(20))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r220": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "321",
"URI": "http://asc.fasb.org/extlink&oid=123583765&loc=SL75117539-209714"
},
"r221xbrl.org/2003/role/disclosureRef"
},
"r302": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "323",
"URI": "httphttps://asc.fasb.org/extlink&oid=114001798&loc=d3e33918-111571",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r222r303": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=122640432&loc=SL121648383-210437",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r223r304": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=122640432&loc=SL121648383-210437",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r224r305": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=122640432&loc=SL121648383-210437",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r225r306": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=122640432&loc=SL121648383-210437",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r226r307": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(e)(3)",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=122640432&loc=SL121648383-210437",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r227r308": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(e)(4)",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=122640432&loc=SL121648383-210437",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r228r309": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124255953&loc=SL82919244-210447"
},
"r229,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r31": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.2)",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126953954&loc=SL114868664-224227",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r310": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124255953&loc=SL82919249-210447"
},
"r23": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(22))",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120391452&loc=d3e13212-122682xbrl.org/2003/role/disclosureRef"
},
"r230r311": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124255953&loc=SL82919253-210447",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r231r312": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124255953&loc=SL82919258-210447",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r232r313": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124255953&loc=SL82919230-210447",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r233r314": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124258926&loc=SL82898722-210454",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r234r315": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124269663&loc=SL82922888-210455",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r235r316": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124269663&loc=SL82922895-210455",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r236r317": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "326",
"URI": "httphttps://asc.fasb.org/extlink&oid=124269663&loc=SL82922900-210455"
},
"r237": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "30",
"Topic": "326",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=121590138&loc=SL82922954-210456"
},
"r238": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=6388964&loc=d3e16225-109274"
},
"r239": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16265-109275"
},
"r24": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(25))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r240": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(2)",
"Topic": "350",
"URI": "http://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275"
},
"r241": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "05",
"SubTopic": "10",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=109226317&loc=d3e202-110218"
},
"r242": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r243": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r244": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r245": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "360",
"URI": "http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229"
},
"r246": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "360",
"URI": "httpxbrl.org/2003/role/disclosureRef"
},
"r318": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "360",
"URI": "https://asc.fasb.org/extlink&oid=109226691&loc=d3e2941-110230"
},
"r247,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r319": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=6394359&loc=d3e17939-110869",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r32": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic SX 210.5-03.CC3)",
"Topic": "360220",
"URI": "httphttps://asc.fasb.org/extlink&oid=27011434126953954&loc=d3e125687-122742SL114868664-224227",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r248r320": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(1)",
"Topic": "360420",
"URI": "httphttps://asc.fasb.org/topic&trid=2155823"
},
"r249extlink&oid=6394359&loc=d3e17939-110869",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r321": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=6394359&loc=d3e17939-110869",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r322": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "420",
"URI": "httphttps://asc.fasb.org/extlink&oid=6394359&loc=d3e17939-110869",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r25": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(27)(b)r323": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=6394359&loc=d3e17939-110869",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r324": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=6394359&loc=d3e17939-110869",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r325": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB TOPIC 5.P.4(b)(2))",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=115931487&loc=d3e140904-122747",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r326": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.P.4(b)(1))",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=115931487&loc=d3e140904-122747",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r327": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.P.4(b)(2))",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=115931487&loc=d3e140904-122747",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r328": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.P.4(d))",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=115931487&loc=d3e140904-122747",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r329": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.P.4(e))",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=115931487&loc=d3e140904-122747",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r33": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.4)",
"Topic": "210220",
"URI": "httphttps://asc.fasb.org/extlink&oid=120391452126953954&loc=d3e13212-122682"
},
"r250SL114868664-224227",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r330": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "420",
"URI": "https://asc.fasb.org/topic&trid=2175745",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r331": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "440",
"URI": "httphttps://asc.fasb.org/extlink&oid=123406679&loc=d3e25336-109308",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r251r332": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "440",
"URI": "httphttps://asc.fasb.org/extlink&oid=123406679&loc=d3e25336-109308"
},
"r252": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "440",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/topic&trid=21446482003/role/disclosureRef"
},
"r253r333": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "450",
"URI": "httphttps://asc.fasb.org/extlink&oid=121557415&loc=d3e14326-108349d3e14435-108349",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r254r334": {
"Name": "Accounting Standards Codification",
"Paragraph": "109",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "450",
"URI": "http://asc.fasb.org/extlink&oid=121557415&loc=d3e14615-108349"
},
"r255": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "450",
"URI": "http://asc.fasb.org/topic&trid=2127136"
},
"r256": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "470",
"URI": "httpSubparagraph": "(b)",
"Topic": "450",
"URI": "https://asc.fasb.org/extlink&oid=99376301121557415&loc=d3e1243-112600"
},
"r257": {
"Name": "Accounting Standards Codification",
"Paragraph": "12A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "470",
"URI": "http://ascd3e14557-108349",
"role": "http://www.fasb.org/extlink&oid=99376301&loc=SL5988623-112600"
},
"r258": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123465755&loc=d3e1835-112601"
},
"r259": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123465755&loc=SL6230698-112601"
},
"r26xbrl.org/2003/role/disclosureRef"
},
"r335": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "1020",
"Subparagraph": "(SX 210.5-02(27)SAB Topic 5.Y.Q2)",
"Topic": "210450",
"URI": "httphttps://asc.fasb.org/extlink&oid=12039145227011672&loc=d3e13212-122682d3e149879-122751",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r260r336": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S65S99",
"SubTopic": "1020",
"Subparagraph": "(SAB Topic 5.Y.Q4)",
"Topic": "470450",
"URI": "httphttps://asc.fasb.org/extlink&oid=12435987227011672&loc=SL124427846-239511d3e149879-122751",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r261r337": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(4)(i))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442526-122756"
},
"r262": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(4)(ii))",
"Topic": "470",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124359900&loc=SL124442526-122756xbrl.org/2003/role/disclosureRef"
},
"r263r338": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(4)(iii)(A))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442526-122756"
},
"r264": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(4)(iii))",
"Topic": "470",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124359900&loc=SL124442526-122756xbrl.org/2003/role/disclosureRef"
},
"r265r339": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(4)(iv))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442526-122756"
},
"r266,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r34": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.8)",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126953954&loc=SL114868664-224227",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r340": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(5))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442526-122756",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r267r341": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-02(a)(4)(i))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442552-122756",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r268r342": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-02(a)(4)(iii)(A))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442552-122756",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r269r343": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-02(a)(4)(iii)(B))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442552-122756"
},
"r27": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(28))",
"Topic": "210",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120391452&loc=d3e13212-122682xbrl.org/2003/role/disclosureRef"
},
"r270r344": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-02(a)(4)(iv))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442552-122756",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r271r345": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-02(a)(5))",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=124359900126975872&loc=SL124442552-122756"
},
"r272": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "20",
"Topic": "470",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=123466302&loc=d3e4724-1126062003/role/disclosureRef"
},
"r273r346": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r274r347": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r275r348": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611"
},
"r276": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "470",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=123466505&loc=SL123495323-112611xbrl.org/2003/role/disclosureRef"
},
"r277r349": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(e)",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611"
},
"r278,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r35": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.9)",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=126953954&loc=SL114868664-224227",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r350": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(f)",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r279r351": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611"
},
"r28": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(29))",
"Topic": "210",
"URI": "http,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r352": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(h)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r353": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(i)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r354": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r355": {
"Name": "Accounting Standards Codification",
"Paragraph": "1C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495334-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r356": {
"Name": "Accounting Standards Codification",
"Paragraph": "1C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495334-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r357": {
"Name": "Accounting Standards Codification",
"Paragraph": "1C",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495334-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r358": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495340-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r359": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495340-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r36": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=114868883&loc=SL114871943-224233",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r360": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495340-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r361": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495348-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r362": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495348-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r363": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495348-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r364": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495348-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r365": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r366": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r367": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r368": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r369": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495371-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r37": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3213-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r370": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495371-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r371": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495371-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r372": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495371-112611",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r373": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466204&loc=SL6031897-161870",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r374": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466204&loc=SL6031897-161870",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r375": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466204&loc=SL6031897-161870",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r376": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r377": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r378": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r379": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r38": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3213-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r380": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r381": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r382": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(i)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r383": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r384": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496171-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r385": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496171-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r386": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496171-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r387": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496180-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r388": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496189-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r389": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496189-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r39": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3255-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r390": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496189-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r391": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496189-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r392": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21463-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r393": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21475-112644",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r394": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.3-04)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=120397183&loc=d3e187085-122770",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r395": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126919976&loc=SL49130531-203044",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r396": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126919976&loc=SL49130532-203044",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r397": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126919976&loc=SL49130533-203044",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r398": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130551-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r399": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130554-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r4": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(29))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r280": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(hr40": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3291-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r400": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130554-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r401": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130554-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r402": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130554-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r403": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130554-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r404": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130556-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r405": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130558-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r406": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130561-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r407": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130563-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r408": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130563-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r409": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130564-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r41": {
"Name": "Accounting Standards Codification",
"Paragraph": "24",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3521-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r410": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130566-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r411": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130566-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r412": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130566-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r413": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130566-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r414": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130543-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r415": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130545-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r416": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130549-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r417": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130549-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r418": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130550-203045",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r419": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "606",
"URI": "https://asc.fasb.org/topic&trid=49130388",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r42": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a),(c)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3536-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r420": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(i)",
"Topic": "470715",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505123447040&loc=SL123495323-112611d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r281r421": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(id)(iv)(01)",
"Topic": "470715",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505123447040&loc=SL123495323-112611d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r282r422": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611"
},
"r283": {
"Name": "Accounting Standards Codification",
"Paragraph": "1C1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(ad)(iv)(02)",
"Topic": "470715",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505123447040&loc=SL123495334-112611d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r284r423": {
"Name": "Accounting Standards Codification",
"Paragraph": "1C1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bd)(iv)(02)(A)",
"Topic": "470715",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505123447040&loc=SL123495334-112611d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r285r424": {
"Name": "Accounting Standards Codification",
"Paragraph": "1C1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(cd)(iv)(02)(B)",
"Topic": "470715",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505123447040&loc=SL123495334-112611"
},
"r286": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D",
"Publisher": "FASB",
"Section": "50d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r425": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)(C)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r426": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(03)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r427": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(d)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=65877416&loc=SL14450657-114947",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r428": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(f)(3)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=65877416&loc=SL14450657-114947",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r429": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "20",
"Subparagraph": "(a)10",
"Topic": "470718",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505126961718&loc=SL123495340-112611SL116886442-113899",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r287": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20r43": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3536-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r430": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "10",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=126961718&loc=d3e4534-113899",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r431": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "10",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=126961718&loc=d3e4549-113899",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r432": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(ba)",
"Topic": "470718",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505128089324&loc=SL123495340-112611d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r288r433": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "2010",
"Subparagraph": "(ca)(1)",
"Topic": "470718",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505128089324&loc=SL123495340-112611d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r289r434": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "4702",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r435": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(3)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r436": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r437": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r438": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(ii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r439": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r44": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505126954810&loc=SL123495348-112611"
},
"r29": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(3)d3e3602-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r440": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r441": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(01)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r442": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(02)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r443": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(03)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r444": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(04)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r445": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r446": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(ii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r447": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r448": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(01)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r449": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(02)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r45": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3602-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r450": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(03)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r451": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r452": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r453": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r454": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r455": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r456": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r457": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r458": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(ii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r459": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(iii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r46": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3602-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r460": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(iv)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r461": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(v)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r462": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(1)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r463": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r464": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(2)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r465": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r466": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(l)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r467": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128097895&loc=SL121327923-165333",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r468": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(f)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128097895&loc=SL121327923-165333",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r469": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(f)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128097895&loc=SL121327923-165333",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r47": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3044-108585",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r470": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 14.D.2.Q6)",
"Topic": "210718",
"URI": "httphttps://asc.fasb.org/extlink&oid=120391452122041274&loc=d3e13212-122682"
},
"r290": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470d3e301413-122809",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r471": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "718",
"URI": "https://asc.fasb.org/topic&trid=2228938",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r472": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "35",
"Subparagraph": "(b)",
"Topic": "720",
"URI": "https://asc.fasb.org/extlink&oid=6420018&loc=d3e36677-107848",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r473": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=123427490&loc=d3e32247-109318",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r474": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=123427490&loc=d3e32280-109318",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r475": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32672-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r476": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32687-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r477": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32705-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r478": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32809-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r479": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32840-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r48": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126999549&loc=d3e4273-108586",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r480": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r481": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r482": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r483": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32847-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r484": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32857-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r485": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32639-109319",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r486": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=126983759&loc=SL121830611-158277",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r487": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=126983759&loc=SL121830611-158277",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r488": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)(2)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=126983759&loc=SL121830611-158277",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r489": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)(3)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=126983759&loc=SL121830611-158277",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r49": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126999549&loc=d3e4304-108586",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r490": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB TOPIC 6.I.5.Q1)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=122134291&loc=d3e330036-122817",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r491": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB TOPIC 6.I.7)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=122134291&loc=d3e330036-122817",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r492": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.Fact.4)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=122134291&loc=d3e330036-122817",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r493": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 11.C)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=122134291&loc=d3e330215-122817",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r494": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=123586238&loc=d3e38679-109324",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r495": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "270",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=6424409&loc=d3e44925-109338",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r496": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=6424122&loc=d3e41874-109331",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r497": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "740",
"URI": "https://asc.fasb.org/topic&trid=2144680",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r498": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=128092470&loc=d3e4946-128472",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r499": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "805",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505128092470&loc=SL123495348-112611"
},
"r291": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "470",
"URI": "http://ascd3e4946-128472",
"role": "http://www.fasb.org/extlink&oid=123466505&loc=SL123495348-112611"
},
"r292": {
"Name": "Accounting Standards Codification",
"Paragraph": "1E",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495348-112611"
},
"r293": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611"
},
"r294": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611"
},
"r295": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611"
},
"r296": {
"Name": "Accounting Standards Codification",
"Paragraph": "1F",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495355-112611"
},
"r297": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495371-112611"
},
"r298": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495371-112611"
},
"r299": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466505&loc=SL123495371-112611"
},
"r3": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "105",
"URI": "http://asc.fasb.org/extlink&oid=124434974&loc=SL124442142-165695"
},
"r30": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(1))",
"Topic": "210",
"URI": "httpxbrl.org/2003/role/disclosureRef"
},
"r5": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(1))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r300": {
"Name": "Accounting Standards Codification",
"Paragraph": "1I",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(dr50": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126999549&loc=d3e4313-108586",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r500": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=128092918&loc=SL128093175-196984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r501": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=128092918&loc=SL128093175-196984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r502": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=128092918&loc=SL128093175-196984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r503": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(c)(1)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=126975305&loc=d3e6927-128479",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r504": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(1)",
"Topic": "805",
"URI": "https://asc.fasb.org/extlink&oid=126975305&loc=d3e7008-128479",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r505": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "808",
"URI": "https://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r506": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=116870748&loc=SL6758485-165988",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r507": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=116870748&loc=SL6758485-165988",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r508": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bb)",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=123419778&loc=d3e5710-111685",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r509": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "810",
"URI": "https://asc.fasb.org/extlink&oid=123419778&loc=d3e5710-111685",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r51": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126999549&loc=d3e4332-108586",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r510": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Subparagraph": "(f)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126731327&loc=d3e90205-114008",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r511": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Subparagraph": "(a)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126731327&loc=SL126733271-114008",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r512": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)(1)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r513": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)(2)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r514": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)(3)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r515": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(b)(1)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r516": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(b)(2)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r517": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(c)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r518": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(d)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r519": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)(2)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r52": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126999549&loc=SL98516268-108586",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r520": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)(3)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r521": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)(4)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r522": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(f)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r523": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL126732908-238011",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r524": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r525": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r526": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)(2)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r527": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r528": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r529": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=123596393&loc=d3e14064-108612",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r53": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "235",
"URI": "https://asc.fasb.org/topic&trid=2122369",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r530": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL120254526-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r531": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL120254526-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r532": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL121967933-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r533": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL121967933-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r534": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL121967933-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r535": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)(2)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL121967933-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r536": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL122642865-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r537": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=126983955&loc=SL122642865-165497",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r538": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=118261656&loc=d3e32136-110900",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r539": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r54": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "https://asc.fasb.org/extlink&oid=124431687&loc=d3e22499-107794",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r540": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r541": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(c)",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r542": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(d)",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r543": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=6450520&loc=d3e32583-110901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r544": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "830",
"URI": "https://asc.fasb.org/extlink&oid=6450520&loc=d3e32618-110901",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r545": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "832",
"URI": "https://asc.fasb.org/extlink&oid=128342835&loc=SL128342857-244232",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r546": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "832",
"URI": "https://asc.fasb.org/extlink&oid=128342835&loc=SL128342857-244232",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r547": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "835",
"URI": "https://asc.fasb.org/extlink&oid=124429444&loc=SL124452920-239629",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r548": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128293352&loc=SL77916155-209984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r549": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128293352&loc=SL126838806-209984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r55": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "260",
"URI": "https://asc.fasb.org/extlink&oid=124432515&loc=d3e3550-109257",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r550": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128293352&loc=SL126838806-209984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r551": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128293352&loc=SL126838806-209984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r552": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)(1)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128293352&loc=SL126838806-209984",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r553": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=123391704&loc=SL77918627-209977",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r554": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=123391704&loc=SL77918627-209977",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r555": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=123391704&loc=SL77918643-209977",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r556": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "470842",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466505128292326&loc=SL123495371-112611"
},
"r301": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "470SL77918666-209980",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r557": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918673-209980",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r558": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(1)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918686-209980",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r559": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(3)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918686-209980",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r56": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "260",
"URI": "https://asc.fasb.org/extlink&oid=124432515&loc=d3e3630-109257",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r560": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(4)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918686-209980",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r561": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918701-209980",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r562": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"SubTopic": "20",
"Topic": "842",
"URI": "https://asc.fasb.org/subtopic&trid=77888251",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r563": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "848",
"URI": "https://asc.fasb.org/extlink&oid=125980421&loc=SL122150809-237846",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r564": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "848",
"URI": "https://asc.fasb.org/extlink&oid=125980421&loc=SL125981372-237846",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r565": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "848",
"URI": "https://asc.fasb.org/extlink&oid=125980421&loc=SL125981372-237846",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r566": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "848",
"URI": "https://asc.fasb.org/extlink&oid=125980421&loc=SL125981372-237846",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r567": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(3)(iii)(01)",
"Topic": "848",
"URI": "https://asc.fasb.org/extlink&oid=125980421&loc=SL125981372-237846",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r568": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(3)(iii)(03)",
"Topic": "848",
"URI": "https://asc.fasb.org/extlink&oid=125980421&loc=SL125981372-237846",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r569": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "855",
"URI": "https://asc.fasb.org/extlink&oid=6842918&loc=SL6314017-165662",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r57": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "275",
"URI": "https://asc.fasb.org/extlink&oid=99393423&loc=d3e6351-108592",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r570": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "855",
"URI": "https://asc.fasb.org/extlink&oid=6842918&loc=SL6314017-165662",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r571": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "855",
"URI": "https://asc.fasb.org/topic&trid=2122774",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r572": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)(i)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r573": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)(ii)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r574": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r575": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(1)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r576": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(2)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r577": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(3)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r578": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(1)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r579": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(2)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r58": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "https://asc.fasb.org/extlink&oid=99393423&loc=d3e6351-108592",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r580": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(3)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r581": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "860",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466204121570589&loc=SL6031897-161870d3e107314-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r302r582": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "470860",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466204121570589&loc=SL6031897-161870d3e107314-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r303r583": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "470860",
"URI": "httphttps://asc.fasb.org/extlink&oid=123466204121570589&loc=SL6031897-161870"
},
"r304": {
"Name": "Accounting Standards Codification",
"Paragraph": "69B",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "http://ascd3e107314-111719",
"role": "http://www.fasb.org/extlink&oid=123466577&loc=SL123495735-112612"
},
"r305": {
"Name": "Accounting Standards Codification",
"Paragraph": "69C",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466577&loc=SL123495737-112612"
},
"r306": {
"Name": "Accounting Standards Codification",
"Paragraph": "69E",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466577&loc=SL123495743-112612"
},
"r307": {
"Name": "Accounting Standards Codification",
"Paragraph": "69F",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123466577&loc=SL123495745-112612"
},
"r308": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "40",
"SubTopic": "50",
"Topic": "470",
"URI": "http://asc.fasb.org/extlink&oid=123467658&loc=d3e12317-112629"
},
"r309xbrl.org/2003/role/disclosureRef"
},
"r584": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "40",
"SubTopic": "50",
"50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r585": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=66007379&loc=d3e113888-111728",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r586": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=109249958&loc=SL34722452-111729",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r587": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(1)",
"Topic": "470860",
"URI": "httphttps://asc.fasb.org/extlink&oid=123467658128311188&loc=d3e12355-112629"
},
"r31": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(3))",
"Topic": "210",
"URI": "httpd3e122625-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r588": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(2)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122625-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r589": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(3)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122625-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r59": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "https://asc.fasb.org/extlink&oid=99393423&loc=d3e6404-108592",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r590": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(4)(i)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122625-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r591": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(1)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r592": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(2)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r593": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(3)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r594": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(4)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r595": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(5)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r596": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(6)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r597": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(7)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r598": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(b)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r599": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(e)(1)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r6": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(3))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r310": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "470",
"URI": "http://asc.,
"role": "http://fasb.org/topic&trid=2208564"
},
"r311": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "10",
"Topic": "480",
"URI": "http://asc.fasb.org/extlink&oid=109262497&loc=d3e20148-110875"
},
"r312": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(CFRR 211.02)",
"Topic": "480",
"URI": "http://asc.fasb.org/extlink&oid=122040564&loc=d3e177068-122764"
},
"r313": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Topic": "480",
"URI": "http://asc.fasb.org/extlink&oid=122040564&loc=d3e177068-122764"
},
"r314": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=65888546&loc=d3e21300-112643"
},
"r315": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=d3e21553-112644"
},
"r316": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r317": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r318": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r319": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r32": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30)(a)(4))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r320": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r321": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r322": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r323": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(i)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r324": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496158-112644"
},
"r325": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496171-112644"
},
"r326": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496171-112644"
},
"r327": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496171-112644"
},
"r328": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496180-112644"
},
"r329": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496189-112644"
},
"r33": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r330": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496189-112644"
},
"r331": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496189-112644"
},
"r332": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=SL123496189-112644"
},
"r333": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=d3e21463-112644"
},
"r334": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=d3e21475-112644"
},
"r335": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505us-gaap/role/ref/legacyRef"
},
"r60": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "httphttps://asc.fasb.org/extlink&oid=12346781799393423&loc=d3e21484-112644"
},
"r336": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.d3e6061-108592",
"role": "http://fasb.org/extlink&oid=123467817&loc=d3e21488-112644"
},
"r337": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=d3e21506-112644"
},
"r338": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=d3e21521-112644"
},
"r339": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=123467817&loc=d3e21538-112644"
},
"r34": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(31))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r340": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.3-04)",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=120397183&loc=d3e187085-122770"
},
"r341": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "50",
"Topic": "505",
"URI": "http://asc.fasb.org/extlink&oid=6784392&loc=d3e188667-122775"
},
"r342": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "505",
"URI": "http://asc.fasb.org/topic&trid=2208762"
},
"r343": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123360276&loc=SL49130531-203044"
},
"r344": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123360276&loc=SL49130532-203044"
},
"r345": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130551-203045"
},
"r346": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130551-203045"
},
"r347": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130554-203045"
},
"r348": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130554-203045"
},
"r349": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130554-203045"
},
"r35": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(32))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r350": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130554-203045"
},
"r351": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130554-203045"
},
"r352": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(1)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130556-203045"
},
"r353": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130556-203045"
},
"r354": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130558-203045"
},
"r355": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130561-203045"
},
"r356": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130563-203045"
},
"r357": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130563-203045"
},
"r358": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130564-203045"
},
"r359": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130566-203045"
},
"r36": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(7))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r360": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130566-203045"
},
"r361": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130566-203045"
},
"r362": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130566-203045"
},
"r363": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130543-203045"
},
"r364": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130545-203045"
},
"r365": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130549-203045"
},
"r366": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130549-203045"
},
"r367": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123351226&loc=SL49130550-203045"
},
"r368": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123410239&loc=SL49130690-203046-203046"
},
"r369": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123410239&loc=SL49130690-203046-203046"
},
"r37": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(8))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r370": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "http://asc.fasb.org/extlink&oid=123410239&loc=SL49130690-203046-203046"
},
"r371": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "606",
"URI": "http://asc.fasb.org/topic&trid=49130388"
},
"r372": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "705",
"URI": "http://asc.fasb.org/topic&trid=2122478"
},
"r373": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(i)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r374": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(ii)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r375": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(01)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r376": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r377": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)(A)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r378": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)(B)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r379": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(02)(C)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r38": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.1)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r380": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(03)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r381": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(n)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920"
},
"r382": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=123450688&loc=d3e4179-114921"
},
"r383": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450702-114947"
},
"r384": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(d)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450657-114947"
},
"r385": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(f)(3)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450657-114947"
},
"r386": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=65877416&loc=SL14450673-114947"
},
"r387": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "80",
"Topic": "715",
"URI": "http://asc.fasb.org/extlink&oid=35742348&loc=SL14450788-114948"
},
"r388": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "10",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=123468992&loc=d3e4534-113899"
},
"r389": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5047-113901"
},
"r39": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.13(a))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r390": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5047-113901"
},
"r391": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5047-113901"
},
"r392": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(3)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r393": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a),(g)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r394": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b),(f)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r395": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r396": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r397": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(i)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r398": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(i)-(ii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r399": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r4": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "105",
"URI": "http://asc.fasb.org/extlink&oid=124434974&loc=SL124442142-165695"
},
"r40": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.13)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r400": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r401": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(3)-(4)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r402": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r403": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r404": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r405": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r406": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r407": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(i)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r408": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(ii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r409": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(iii)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r41": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.14)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r410": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(iv)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r411": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r412": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r413": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(h)(1)(i)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r414": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(i)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r415": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=d3e5070-113901"
},
"r416": {
"Name": "Accounting Standards Codification",
"Paragraph": "2A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=120381028&loc=SL79508275-113901"
},
"r417": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r418": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r419": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r42": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.17)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r420": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r421": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r422": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(f)(1)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r423": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(f)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r424": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(g)(2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=121322162&loc=SL121327923-165333"
},
"r425": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 14.D.2)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=122041274&loc=d3e301413-122809"
},
"r426": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 14.F)",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=122041274&loc=d3e301413-122809"
},
"r427": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=122142933&loc=d3e11149-113907"
},
"r428": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "718",
"URI": "http://asc.fasb.org/extlink&oid=122142933&loc=d3e11178-113907"
},
"r429": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "718",
"URI": "http://asc.fasb.org/topic&trid=2228938"
},
"r43": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19(a))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r430": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "35",
"Subparagraph": "(a)",
"Topic": "720",
"URI": "http://asc.fasb.org/extlink&oid=6420018&loc=d3e36677-107848"
},
"r431": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "35",
"Subparagraph": "(b)",
"Topic": "720",
"URI": "http://asc.fasb.org/extlink&oid=6420018&loc=d3e36677-107848"
},
"r432": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "730",
"URI": "http://asc.fasb.org/extlink&oid=6420194&loc=d3e21568-108373"
},
"r433": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "730",
"URI": "http://asc.fasb.org/extlink&oid=6420387&loc=d3e23199-108380"
},
"r434": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "730",
"URI": "http://asc.fasb.org/extlink&oid=6420387&loc=d3e23221-108380"
},
"r435": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=123427490&loc=d3e32247-109318"
},
"r436": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=123427490&loc=d3e32280-109318"
},
"r437": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32672-109319"
},
"r438": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32687-109319"
},
"r439": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32705-109319"
},
"r44": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19(b),22(b))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r440": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32718-109319"
},
"r441": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32809-109319"
},
"r442": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32840-109319"
},
"r443": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319"
},
"r444": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319"
},
"r445": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319"
},
"r446": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32847-109319"
},
"r447": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32857-109319"
},
"r448": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32559-109319"
},
"r449": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32559-109319"
},
"r45": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19,20)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r450": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32621-109319"
},
"r451": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32632-109319"
},
"r452": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=121826272&loc=d3e32639-109319"
},
"r453": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=123459177&loc=SL121830611-158277"
},
"r454": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=123459177&loc=SL121830611-158277"
},
"r455": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)(2)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=123459177&loc=SL121830611-158277"
},
"r456": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(d)(3)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=123459177&loc=SL121830611-158277"
},
"r457": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB TOPIC 6.I.5.Q1)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=122134291&loc=d3e330036-122817"
},
"r458": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB TOPIC 6.I.7)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=122134291&loc=d3e330036-122817"
},
"r459": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.Fact.4)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=122134291&loc=d3e330036-122817"
},
"r46": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.19-26)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r460": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 11.C)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=122134291&loc=d3e330215-122817"
},
"r461": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=123586238&loc=d3e38679-109324"
},
"r462": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "270",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=6424409&loc=d3e44925-109338"
},
"r463": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "740",
"URI": "http://asc.fasb.org/extlink&oid=6424122&loc=d3e41874-109331"
},
"r464": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "740",
"URI": "http://asc.fasb.org/topic&trid=2144680"
},
"r465": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=79982066&loc=d3e1392-128463"
},
"r466": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=79982066&loc=d3e1486-128463"
},
"r467": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "30",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=6911189&loc=d3e6408-128476"
},
"r468": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=116859824&loc=d3e6819-128478"
},
"r469": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "35",
"SubTopic": "30",
"Subparagraph": "b",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=116859824&loc=d3e6819-128478"
},
"r47": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.20)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r470": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(b)(4)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e6927-128479"
},
"r471": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(c)(1)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e6927-128479"
},
"r472": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(1)",
"Topic": "805",
"URI": "http://asc.fasb.org/extlink&oid=120321790&loc=d3e7008-128479"
},
"r473": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "805",
"URI": "http://asc.fasb.org/topic&trid=2303972"
},
"r474": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=123385629&loc=SL5834089-161433"
},
"r475": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434"
},
"r476": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434"
},
"r477": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434"
},
"r478": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "808",
"URI": "http://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434"
},
"r479": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "808",
"URI": "http://asc.fasb.org/topic&trid=5833765"
},
"r48": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.21)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r480": {
"Name": "Accounting Standards Codification",
"Paragraph": "19",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=123454820&loc=SL4569616-111683"
},
"r481": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=123454820&loc=SL4569643-111683"
},
"r482": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=116870748&loc=SL6758485-165988"
},
"r483": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=116870748&loc=SL6758485-165988"
},
"r484": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=d3e5614-111684"
},
"r485": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(3)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684"
},
"r486": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c),(3)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=109239629&loc=SL4573702-111684"
},
"r487": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bb)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=123419778&loc=d3e5710-111685"
},
"r488": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "810",
"URI": "http://asc.fasb.org/extlink&oid=123419778&loc=d3e5710-111685"
},
"r489": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "810",
"URI": "http://asc.fasb.org/topic&trid=2197479"
},
"r49": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.22(a)(1))",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r490": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=SL5579240-113959"
},
"r491": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=SL5579245-113959"
},
"r492": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=d3e41620-113959"
},
"r493": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=d3e41638-113959"
},
"r494": {
"Name": "Accounting Standards Codification",
"Paragraph": "4A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=SL5618551-113959"
},
"r495": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=SL5624163-113959"
},
"r496": {
"Name": "Accounting Standards Codification",
"Paragraph": "4B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=SL5624163-113959"
},
"r497": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=125515794&loc=d3e41675-113959"
},
"r498": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"SubTopic": "15",
"Topic": "815",
"URI": "http://asc.fasb.org/subtopic&trid=2229187"
},
"r499": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Subparagraph": "(f)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123477628&loc=d3e90205-114008"
},
"r5": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=124429488&loc=d3e326-107755"
},
"r50": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.22)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r500": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)(1)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r501": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)(2)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r502": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)(3)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r503": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(b)(1)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r504": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(b)(2)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r505": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(c)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r506": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(d)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r507": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)(2)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r508": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)(3)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r509": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)(4)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r51": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.24)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r510": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(f)",
"Topic": "815",
"URI": "http://asc.fasb.org/extlink&oid=123482062&loc=SL123482106-238011"
},
"r511": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r512": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r513": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r514": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)(2)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r515": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r516": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r517": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r518": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19207-110258"
},
"r519": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "820",
"URI": "http://asc.fasb.org/extlink&oid=123874694&loc=d3e19279-110258"
},
"r52": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.25)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r520": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123594938&loc=d3e13467-108611"
},
"r521": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123594938&loc=d3e13476-108611"
},
"r522": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123594938&loc=d3e13531-108611"
},
"r523": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123594938&loc=d3e13537-108611"
},
"r524": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123594938&loc=d3e13537-108611"
},
"r525": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123596393&loc=d3e14064-108612"
},
"r526": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL120254526-165497"
},
"r527": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL120254526-165497"
},
"r528": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL121967933-165497"
},
"r529": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL121967933-165497"
},
"r53": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.28,29)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r530": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL121967933-165497"
},
"r531": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(c)(2)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL121967933-165497"
},
"r532": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL122642865-165497"
},
"r533": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "825",
"URI": "http://asc.fasb.org/extlink&oid=123597120&loc=SL122642865-165497"
},
"r534": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "230",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=123444420&loc=d3e33268-110906"
},
"r535": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32136-110900"
},
"r536": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r537": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r538": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(c)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r539": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Subparagraph": "(d)",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=118261656&loc=d3e32211-110900"
},
"r54": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r540": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=6450520&loc=d3e32583-110901"
},
"r541": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "830",
"URI": "http://asc.fasb.org/extlink&oid=6450520&loc=d3e32618-110901"
},
"r542": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=124435984&loc=d3e28541-108399"
},
"r543": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=124435984&loc=d3e28551-108399"
},
"r544": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=124435984&loc=d3e28555-108399"
},
"r545": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=124429444&loc=SL124452920-239629"
},
"r546": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "30",
"Topic": "835",
"URI": "http://asc.fasb.org/extlink&oid=114775985&loc=d3e28878-108400"
},
"r547": {
"Name": "Accounting Standards Codification",
"Paragraph": "40",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(Note 3)",
"Topic": "840",
"URI": "http://asc.fasb.org/extlink&oid=123403562&loc=d3e38371-112697"
},
"r548": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "20",
"Topic": "840",
"URI": "http://asc.fasb.org/extlink&oid=123415192&loc=d3e39896-112707"
},
"r549": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "840",
"URI": "http://asc.fasb.org/extlink&oid=123406913&loc=d3e41502-112717"
},
"r55": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29-30)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r550": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "840",
"URI": "http://asc.fasb.org/extlink&oid=123406913&loc=d3e41502-112717"
},
"r551": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=123341672&loc=SL77916155-209984"
},
"r552": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=123391704&loc=SL77918627-209977"
},
"r553": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=123391704&loc=SL77918627-209977"
},
"r554": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=123408670&loc=SL77918666-209980"
},
"r555": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(3)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=123408670&loc=SL77918673-209980"
},
"r556": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "842",
"URI": "http://asc.fasb.org/extlink&oid=123408481&loc=SL77919140-209958"
},
"r557": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "848",
"URI": "http://asc.fasb.org/extlink&oid=122150657&loc=SL122150809-237846"
},
"r558": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864"
},
"r559": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864"
},
"r56": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.29-31)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r560": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864"
},
"r561": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864"
},
"r562": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864"
},
"r563": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39603-107864"
},
"r564": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "850",
"URI": "http://asc.fasb.org/extlink&oid=6457730&loc=d3e39691-107864"
},
"r565": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "850",
"URI": "http://asc.fasb.org/topic&trid=2122745"
},
"r566": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "852",
"URI": "http://asc.fasb.org/extlink&oid=124433192&loc=SL2890621-112765"
},
"r567": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "852",
"URI": "http://asc.fasb.org/extlink&oid=124433192&loc=SL2890621-112765"
},
"r568": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "852",
"URI": "http://asc.fasb.org/extlink&oid=84165509&loc=d3e56426-112766"
},
"r569": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "855",
"URI": "http://asc.fasb.org/extlink&oid=6842918&loc=SL6314017-165662"
},
"r57": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02.9)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682"
},
"r570": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "855",
"URI": "http://asc.fasb.org/extlink&oid=6842918&loc=SL6314017-165662"
},
"r571": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "855",
"URI": "http://asc.fasb.org/extlink&oid=6842918&loc=SL6314020-165662"
},
"r572": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "855",
"URI": "http://asc.fasb.org/topic&trid=2122774"
},
"r573": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)(i)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r574": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)(ii)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r575": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r576": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r577": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r578": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(bb)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r579": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r58": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=99393222&loc=SL20226024-175313"
},
"r580": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r581": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719"
},
"r582": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r583": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r584": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r585": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719"
},
"r586": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=66007379&loc=d3e113888-111728"
},
"r587": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=109249958&loc=SL34722452-111729"
},
"r588": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122625-111746"
},
"r589": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122625-111746"
},
"r59": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=99393222&loc=SL20226049-175313"
},
"r590": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122625-111746"
},
"r591": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(4)(i)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122625-111746"
},
"r592": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r593": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(2)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r594": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(3)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r595": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(4)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r596": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(5)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r597": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(6)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r598": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(a)(7)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r599": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(b)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r6": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "205",
"URI": "http://asc.fasb.org/extlink&oid=109222650&loc=SL51721683-107760"
},
"r60": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=99393222&loc=SL20226052-175313"
},
"r600": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(e)(1)",
"Topic": "860",
"URI": "http://asc.fasb.org/extlink&oid=125521744&loc=d3e122739-111746"
},
"r601us-gaap/role/ref/legacyRef"
},
"r600": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(e)(2)",
"Topic": "860",
"URI": "httphttps://asc.fasb.org/extlink&oid=125521744128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r602r601": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "50",
"Subparagraph": "(e)(3)",
"Topic": "860",
"URI": "httphttps://asc.fasb.org/extlink&oid=125521744128311188&loc=d3e122739-111746",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r603r602": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "910",
"URI": "httphttps://asc.fasb.org/extlink&oid=123353855126937589&loc=SL119991595-234733",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r604r603": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "310",
"Subparagraph": "(b)",
"Topic": "912",
"URI": "httphttps://asc.fasb.org/extlink&oid=123371682126938201&loc=d3e55415-109406",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r605r604": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "2545",
"SubTopic": "730310",
"Topic": "912",
"URI": "httphttps://asc.fasb.org/extlink&oid=6472174126938201&loc=d3e58812-109433d3e55302-109406",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r606r605": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 11.L)",
"Topic": "924",
"URI": "httphttps://asc.fasb.org/extlink&oid=6472922&loc=d3e499488-122856",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r607r606": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "926",
"URI": "httphttps://asc.fasb.org/extlink&oid=120154821&loc=SL120154904-197079",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r608r607": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "926",
"URI": "httphttps://asc.fasb.org/extlink&oid=120154821&loc=SL120154904-197079",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r609r608": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "20",
"Subparagraph": "(c)",
"Topic": "926",
"URI": "httphttps://asc.fasb.org/extlink&oid=120154821&loc=SL120154904-197079"
},
"r61": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "220",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124509347&loc=SL7669646-108580"
},
"r610": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(a)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e61929-109447"
},
"r611": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(b)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e61929-109447"
},
"r612": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(a)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e62059-109447"
},
"r613": {
"Name": "Accounting Standards Codification",
"Paragraph": "20",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(b)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e62059-109447"
},
"r614": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(a)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e62395-109447"
},
"r615": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(b)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e62395-109447"
},
"r616": {
"Name": "Accounting Standards Codification",
"Paragraph": "33",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(a)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e62479-109447"
},
"r617": {
"Name": "Accounting Standards Codification",
"Paragraph": "33",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(b)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e62479-109447"
},
"r618": {
"Name": "Accounting Standards Codification",
"Paragraph": "35A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(a)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=SL6807758-109447"
},
"r619": {
"Name": "Accounting Standards Codification",
"Paragraph": "35A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(b)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=SL6807758-109447"
},
"r62": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124509347&loc=SL7669646-108580"
},
"r620": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(c)(1)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e61872-109447"
},
"r621": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "235",
"Subparagraph": "(c)(2)",
"Topic": "932",
"URI": "http://asc.fasb.org/extlink&oid=123377692&loc=d3e61872-109447"
},
"r622": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(1)(a))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r623": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(11))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r624": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(13))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r625": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(16))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r626": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(17))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r627": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(23))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r628": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(4))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r629": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(5))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r63": {
"Name": "Accounting Standards Codification",
"Paragraph": "10A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124509347&loc=SL7669646-108580"
},
"r630": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(6))",
"Topic": "942",
"URI": "httpxbrl.org/2003/role/disclosureRef"
},
"r609": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03(6))",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=120398452126897435&loc=d3e534808-122878",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r631r61": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "S9950",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.15(5))10",
"Topic": "942275",
"URI": "httphttps://asc.fasb.org/extlink&oid=12039845299393423&loc=d3e534808-122878"
},
"r632": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.9-03.17)",
"Topic": "942",
"URI": "http://asc.d3e6132-108592",
"role": "http://fasb.org/extlink&oid=120398452&loc=d3e534808-122878"
},
"r633": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(22))",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r634us-gaap/role/ref/legacyRef"
},
"r610": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(26))",
"Topic": "942",
"URI": "httphttps://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r635r611": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04(27))",
"Topic": "942",
"URI": "httphttps://asc.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r636": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.9-04.10)",
"Topic": "942",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120399700&loc=SL114874048-224260"
},
"r637": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=123599081&loc=d3e62652-112803"
},
"r638xbrl.org/2003/role/disclosureRef"
},
"r612": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "360",
"Topic": "942",
"URI": "httphttps://asc.fasb.org/extlink&oid=124429447&loc=SL124453093-239630"
},
"r639": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "470",
"Topic": "942",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=123599511&loc=d3e64711-112823"
},
"r64": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124509347&loc=d3e637-108580"
},
"r640": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "825",
"Topic": "942",
"URI": "http://asc.fasb.org/extlink&oid=123345438&loc=d3e61044-112788"
},
"r641": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(16))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r642": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(1)(g))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r643": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(12))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r644": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(16))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r645": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(19))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r646": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(2))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r647": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(23)(a)(3))",
"Topic": "944",
"URI": "httpxbrl.org/2003/role/disclosureRef"
},
"r613": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(23)(a)(3))",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=120400017126734703&loc=d3e572229-122910"
},
"r648": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(23)(a)(4))",
"Topic": "944",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r649": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(25))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r65": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124509347&loc=d3e681-108580"
},
"r650": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03(a)(8))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r651": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.(a),19)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r652": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "210",
"Subparagraph": "(SX 210.7-03.15(a))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400017&loc=d3e572229-122910"
},
"r653": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(18))",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r654xbrl.org/2003/role/disclosureRef"
},
"r614": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(22))",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r655r615": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(23))",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r656r616": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "220",
"Subparagraph": "(SX 210.7-04(9))",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=120400993&loc=SL114874131-224263"
},
"r657": {
"Name": "Accounting Standards Codification",
"Paragraph": "4H",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Topic": "944",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=116884468&loc=SL65671331-158438xbrl.org/2003/role/disclosureRef"
},
"r658r617": {
"Name": "Accounting Standards Codification",
"Paragraph": "7A",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Subparagraph": "(d)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124506351&loc=SL117782755-158439"
},
"r659": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(a)",
"Topic": "944",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124504033&loc=SL117783719-158441"
},
"r66": {
"Name": "Accounting Standards Codification",
"Paragraph": "14A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124509347&loc=SL7669686-108580"
},
"r660": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(b)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=124504033&loc=SL117783719-158441"
},
"r661": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(c)",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=124504033&loc=SL117783719-158441"
},
"r662": {
"Name": "Accounting Standards Codification",
"Paragraph": "29F",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Topic": "944",
"URI": "http://asc.fasb.org/extlink&oid=124504033&loc=SL117819544-158441"
},
"r663xbrl.org/2003/role/disclosureRef"
},
"r618": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(a)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r664r619": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(b)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641"
},
"r665,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r62": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "https://asc.fasb.org/extlink&oid=99393423&loc=d3e6143-108592",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r620": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r666r621": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(f)(1)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r667r622": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(f)(2)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r668r623": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(g)(1)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r669r624": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(g)(2)(i)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641"
},
"r67": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "220",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124509347&loc=SL7669619-108580xbrl.org/2003/role/disclosureRef"
},
"r670r625": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(g)(2)(ii)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r671r626": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(g)(2)(iii)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r672r627": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(g)(2)(iv)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r673r628": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(h)(1)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r674r629": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(h)(2)",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=124501264&loc=SL117420844-207641"
},
"r675,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r63": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 4.E)",
"Topic": "310",
"URI": "https://asc.fasb.org/extlink&oid=122038336&loc=d3e74512-122707",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r630": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "825",
"Topic": "944",
"URI": "httphttps://asc.fasb.org/extlink&oid=123600520&loc=SL75241803-196195",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r676r631": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "310",
"Subparagraph": "(SX 210.12-29(Footnote 4))",
"Topic": "948",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402547&loc=d3e617274-123014",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r677r632": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "210",
"Topic": "954",
"URI": "httphttps://asc.fasb.org/extlink&oid=120413173&loc=SL116631458-115580",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r678r633": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "310",
"Topic": "954",
"URI": "httphttps://asc.fasb.org/extlink&oid=123364037126942805&loc=d3e3115-115594",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r679r634": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "440",
"Subparagraph": "(a)",
"Topic": "954",
"URI": "httphttps://asc.fasb.org/extlink&oid=6491277&loc=d3e6429-115629"
},
"r68": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "220",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124509347&loc=SL7669619-108580xbrl.org/2003/role/disclosureRef"
},
"r680r635": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "360",
"Subparagraph": "(d)",
"Topic": "958",
"URI": "httphttps://asc.fasb.org/extlink&oid=120429125126982197&loc=d3e99779-112916",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r681r636": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "360",
"Topic": "958",
"URI": "httphttps://asc.fasb.org/extlink&oid=120429125126982197&loc=d3e99893-112916",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r682r637": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "360",
"Topic": "958",
"URI": "httphttps://asc.fasb.org/extlink&oid=120429125126982197&loc=SL120174063-112916",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r683r638": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column B))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r684r639": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column C))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r685,
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r64": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "320",
"URI": "https://asc.fasb.org/extlink&oid=124260329&loc=d3e26853-111562",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r640": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column D))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r686r641": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column E))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r687r642": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column F))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r688r643": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column G))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r689r644": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column H))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024"
},
"r69": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "220",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=124509347&loc=SL7669619-108580xbrl.org/2003/role/disclosureRef"
},
"r690r645": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Column I))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r691r646": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "360",
"Subparagraph": "(SX 210.12-28(Footnote 2))",
"Topic": "970",
"URI": "httphttps://asc.fasb.org/extlink&oid=120402810&loc=d3e638233-123024",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r692r647": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "310",
"Subparagraph": "(c)",
"Topic": "976",
"URI": "httphttps://asc.fasb.org/extlink&oid=6497875&loc=d3e22274-108663",
"role": "http://www.xbrl.org/2003/role/disclosureRef"
},
"r693r648": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "310",
"Subparagraph": "(b)",
"Topic": "978",
"URI": "httphttps://asc.fasb.org/extlink&oid=123360121126945304&loc=d3e27327-108691"
},
"r694": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "985",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=6501960&loc=d3e128462-111756"
},
"r695": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b"
},
"r696": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-2"
},
"r697": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-23"
},
"r698": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "d1-1"
},
"r699": {
"Name": "Form 10-K",
"Number": "249",
"Publisher": "SEC",
"Section": "310"
},
"r7": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "205",
"URI": "http://asc.fasb.org/topic&trid=2122149"
},
"r70": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124509347&loc=SL7669625-108580"
},
"r700": {
"Name": "Form 20-F",
"Number": "249",
"Publisher": "SEC",
"Section": "220",
"Subsection": "f"
},
"r701": {
"Name": "Form 40-F",
"Number": "249",
"Publisher": "SEC",
"Section": "240",
"Subsection": "f"
},
"r702": {
"Name": "Forms 10-K, 10-Q, 20-F",
"Number": "240",
"Publisher": "SEC",
"Section": "13",
"Subsection": "a-1"
},
"r703": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(a)",
"Publisher": "SEC",
"Section": "1402"
},
"r704": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(b)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(1)"
},
"r705": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(b)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)"
},
"r706": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(b)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(3)"
},
"r707": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(c)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)(i)"
},
"r708": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(c)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)(ii)"
},
"r709": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(c)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)(iii)"
},
"r71": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "220",
"URI": "http://asc2003/role/disclosureRef"
},
"r649": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(a)",
"Publisher": "SEC",
"Section": "1402",
"role": "http://www.fasb.org/extlink&oid=124509347&loc=SL7669625-108580"
},
"r710": {
"Name": "Regulation S-T",
"Number": "232",
"Publisher": "SEC",
"Section": "405"
},
"r711": {
"Name": "Securities Act",
"Number": "230",
"Publisher": "SEC",
"Section": "405"
},
"r712": {
"Name": "Securities Act",
"Number": "7A",
"Publisher": "SEC",
"Section": "B",
"Subsection": "2"
},
"r713xbrl.org/2003/role/disclosureRef"
},
"r65": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "6550",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "848"
},
"r714": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "848"
},
"r715": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "848"
},
"r716": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(3)(iii)(01)",
"Topic": "848"
},
"r717": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "10",
"Subparagraph": "(a)(3)(iii)(03)",
"Topic": "848"
},
"r72": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "220",
"URI": "httpaa)",
"Topic": "320",
"URI": "https://asc.fasb.org/extlink&oid=124509347126970911&loc=d3e557-108580"
},
"r73": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.d3e27161-111563",
"role": "http://fasb.org/extlink&oid=124431353&loc=SL116659661-227067"
},
"r74": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124431353&loc=SL124442407-227067"
},
"r75": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124431353&loc=SL124442411-227067"
},
"r76": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=124431353&loc=SL124452729-227067"
},
"r77": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(20))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r78": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(24))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r79": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(25))",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r8us-gaap/role/ref/legacyRef"
},
"r650": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "210",
"URI": "httphttps://asc.fasb.org/extlink&oid=124098289&loc=d3e6676-107765"
},
"r80": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03,
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r651": {
"Name": "Accounting Standards Codification",
"Paragraph": "16",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=99393222&loc=SL20226024-175313",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r652": {
"Name": "Accounting Standards Codification",
"Paragraph": "21",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=99393222&loc=SL20226049-175313",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r653": {
"Name": "Accounting Standards Codification",
"Paragraph": "22",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=99393222&loc=SL20226052-175313",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r654": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "235",
"URI": "https://asc.fasb.org/extlink&oid=126899994&loc=d3e18823-107790",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r655": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "235",
"URI": "https://asc.fasb.org/extlink&oid=126899994&loc=d3e18823-107790",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r656": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(m)(1)(ii)(A))",
"Topic": "235",
"URI": "https://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r657": {
"Name": "Accounting Standards Codification",
"Paragraph": "52",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "260",
"URI": "https://asc.fasb.org/extlink&oid=128363288&loc=d3e4984-109258",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r658": {
"Name": "Accounting Standards Codification",
"Paragraph": "30",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "280",
"URI": "https://asc.fasb.org/extlink&oid=126901519&loc=d3e8906-108599",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r659": {
"Name": "Accounting Standards Codification",
"Paragraph": "31",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "https://asc.fasb.org/extlink&oid=126901519&loc=d3e8924-108599",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r66": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "30",
"Topic": "350",
"URI": "https://asc.fasb.org/extlink&oid=6388964&loc=d3e16225-109274",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r660": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "30",
"Topic": "326",
"URI": "https://asc.fasb.org/extlink&oid=121590138&loc=SL82922954-210456",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r661": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r662": {
"Name": "Accounting Standards Codification",
"Paragraph": "69B",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466577&loc=SL123495735-112612",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r663": {
"Name": "Accounting Standards Codification",
"Paragraph": "69C",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466577&loc=SL123495737-112612",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r664": {
"Name": "Accounting Standards Codification",
"Paragraph": "69E",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466577&loc=SL123495743-112612",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r665": {
"Name": "Accounting Standards Codification",
"Paragraph": "69F",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466577&loc=SL123495745-112612",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r666": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=SL123496158-112644",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r667": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920602&loc=SL49130690-203046-203046",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r668": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920602&loc=SL49130690-203046-203046",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r669": {
"Name": "Accounting Standards Codification",
"Paragraph": "91",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920602&loc=SL49130690-203046-203046",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r67": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Topic": "350",
"URI": "https://asc.fasb.org/extlink&oid=66006027&loc=d3e16265-109275",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r670": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(ii)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r671": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(iv)(01)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r672": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=123450688&loc=d3e4179-114921",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r673": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=65877416&loc=SL14450702-114947",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r674": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "80",
"Subparagraph": "(a)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=65877416&loc=SL14450673-114947",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r675": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "80",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=35742348&loc=SL14450788-114948",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r676": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r677": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(3)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r678": {
"Name": "Accounting Standards Codification",
"Paragraph": "53",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "20",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=123414884&loc=SL77918982-209971",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r679": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "10",
"Topic": "852",
"URI": "https://asc.fasb.org/extlink&oid=84165509&loc=d3e56426-112766",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r68": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(a)(2)",
"Topic": "350",
"URI": "https://asc.fasb.org/extlink&oid=66006027&loc=d3e16323-109275",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r680": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(a)",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126561865&loc=SL117783719-158441",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r681": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(b)",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126561865&loc=SL117783719-158441",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r682": {
"Name": "Accounting Standards Codification",
"Paragraph": "29F",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126561865&loc=SL117819544-158441",
"role": "http://www.xbrl.org/2003/role/exampleRef"
},
"r683": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r684": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-2",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r685": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "b-23",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r686": {
"Name": "Exchange Act",
"Number": "240",
"Publisher": "SEC",
"Section": "12",
"Subsection": "d1-1",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r687": {
"Name": "Form 10-K",
"Number": "249",
"Publisher": "SEC",
"Section": "310",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r688": {
"Name": "Form 20-F",
"Number": "249",
"Publisher": "SEC",
"Section": "220",
"Subsection": "f",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r689": {
"Name": "Form 40-F",
"Number": "249",
"Publisher": "SEC",
"Section": "240",
"Subsection": "f",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r69": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(b)",
"Topic": "350",
"URI": "https://asc.fasb.org/extlink&oid=66006027&loc=d3e16373-109275",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r690": {
"Name": "Forms 10-K, 10-Q, 20-F",
"Number": "240",
"Publisher": "SEC",
"Section": "13",
"Subsection": "a-1",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r691": {
"Name": "Regulation S-T",
"Number": "232",
"Publisher": "SEC",
"Section": "405",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r692": {
"Name": "Securities Act",
"Number": "230",
"Publisher": "SEC",
"Section": "405",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r693": {
"Name": "Securities Act",
"Number": "7A",
"Publisher": "SEC",
"Section": "B",
"Subsection": "2",
"role": "http://www.xbrl.org/2003/role/presentationRef"
},
"r694": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "272",
"URI": "https://asc.fasb.org/extlink&oid=125520817&loc=d3e70229-108054",
"role": "http://www.xbrl.org/2003/role/recommendedDisclosureRef"
},
"r695": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "450",
"URI": "https://asc.fasb.org/extlink&oid=121557415&loc=d3e14615-108349",
"role": "http://www.xbrl.org/2003/role/recommendedDisclosureRef"
},
"r696": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "855",
"URI": "https://asc.fasb.org/extlink&oid=6842918&loc=SL6314020-165662",
"role": "http://www.xbrl.org/2003/role/recommendedDisclosureRef"
},
"r697": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(1))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r698": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(17))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r699": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(27)(b))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r7": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(30))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r70": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "05",
"SubTopic": "10",
"Topic": "360",
"URI": "https://asc.fasb.org/extlink&oid=109226317&loc=d3e202-110218",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r700": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(28))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r701": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(8))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r702": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(9))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r703": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=124431353&loc=SL124442407-227067",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r704": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=124431353&loc=SL124442411-227067",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r705": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "220",
"URI": "https://asc.fasb.org/extlink&oid=124431353&loc=SL124452729-227067",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r706": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03(b)(4))",
"Topic": "220",
"URI": "httphttps://asc.fasb.org/extlink&oid=123367319126953954&loc=SL114868664-224227"
},
"r81": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.2)",
"Topic": "220",
"URI": "http://asc,
"role": "http://www.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r82": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.3)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r83": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.4)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r84": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.8)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r85": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-03.9)",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=123367319&loc=SL114868664-224227"
},
"r86": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "220",
"URI": "http://asc.fasb.org/extlink&oid=114868883&loc=SL114871943-224233"
},
"r87": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123570139&loc=d3e3151-108585"
},
"r88": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123570139&loc=d3e3179-108585"
},
"r89": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123570139&loc=d3e3213-108585"
},
"r9": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "210",
"URI": "http://asc.fasb.org/extlink&oid=124098289&loc=d3e6676-107765"
},
"r90": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123570139&loc=d3e3213-108585"
},
"r91": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123570139&loc=d3e3255-108585"
},
"r92": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "httpxbrl.org/2009/role/commonPracticeRef"
},
"r707": {
"Name": "Accounting Standards Codification",
"Paragraph": "14",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=123570139126954810&loc=d3e3255-108585"
},
"r93": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "http://asc,
"role": "http://www.fasbxbrl.org/extlink&oid=123570139&loc=d3e3291-108585"
},
"r94": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(g)",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123570139&loc=d3e3291-108585"
},
"r95": {
"Name": "Accounting Standards Codification",
"Paragraph": "15",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230",
"URI": "http://asc.fasb.org/extlink&oid=123570139&loc=d3e3291-108585"
},
"r962009/role/commonPracticeRef"
},
"r708": {
"Name": "Accounting Standards Codification",
"Paragraph": "17",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(df)",
"Topic": "230",
"URI": "httphttps://asc.fasb.org/extlink&oid=123570139126954810&loc=d3e3367-108585"
},
"r97": {
"Name": "Accounting Standards Codification",
"Paragraph": "24",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230,
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r709": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3602-108585",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r71": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "360",
"URI": "https://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r710": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126954810&loc=d3e3602-108585",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r711": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "230",
"URI": "https://asc.fasb.org/extlink&oid=126999549&loc=SL98516268-108586",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r712": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(d))",
"Topic": "235",
"URI": "https://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r713": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(f))",
"Topic": "235",
"URI": "https://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r714": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.4-08(g)(1)(ii))",
"Topic": "235",
"URI": "https://asc.fasb.org/extlink&oid=120395691&loc=d3e23780-122690",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r715": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.12-04(a))",
"Topic": "235",
"URI": "https://asc.fasb.org/extlink&oid=120395691&loc=d3e24072-122690",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r716": {
"Name": "Accounting Standards Codification",
"Paragraph": "23",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "250",
"URI": "https://asc.fasb.org/extlink&oid=124436220&loc=d3e21914-107793",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r717": {
"Name": "Accounting Standards Codification",
"Paragraph": "24",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "250",
"URI": "httphttps://asc.fasb.org/extlink&oid=123570139124436220&loc=d3e3521-108585d3e21930-107793",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r98r718": {
"Name": "Accounting Standards Codification",
"Paragraph": "255",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(eb)",
"Topic": "230250",
"URI": "httphttps://asc.fasb.org/extlink&oid=123570139124436220&loc=d3e3536-108585d3e21711-107793",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r99": {
"Name": "Accounting Standards Codification",
"Paragraph": "25",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "230r719": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "250",
"URI": "https://asc.fasb.org/extlink&oid=124431687&loc=d3e22595-107794",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r72": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "360",
"URI": "https://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r720": {
"Name": "Accounting Standards Codification",
"Paragraph": "55",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "260",
"URI": "https://asc.fasb.org/extlink&oid=126958026&loc=d3e2626-109256",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r721": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "260",
"URI": "https://asc.fasb.org/extlink&oid=124432515&loc=d3e3550-109257",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r722": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "275",
"URI": "https://asc.fasb.org/extlink&oid=99393423&loc=d3e6351-108592",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r723": {
"Name": "Accounting Standards Codification",
"Paragraph": "18",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "280",
"URI": "https://asc.fasb.org/extlink&oid=126901519&loc=d3e8672-108599",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r724": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "321",
"URI": "https://asc.fasb.org/extlink&oid=126980263&loc=SL75117539-209714",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r725": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "321",
"URI": "https://asc.fasb.org/extlink&oid=126980263&loc=SL75117539-209714",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r726": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "321",
"URI": "https://asc.fasb.org/extlink&oid=126980263&loc=SL75117539-209714",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r727": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "323",
"URI": "https://asc.fasb.org/extlink&oid=114001798&loc=d3e33918-111571",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r728": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "30",
"Subparagraph": "(c)",
"Topic": "410",
"URI": "https://asc.fasb.org/extlink&oid=6393242&loc=d3e13237-110859",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r729": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "440",
"URI": "https://asc.fasb.org/extlink&oid=123406679&loc=d3e25383-109308",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r73": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)",
"Topic": "360",
"URI": "https://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r730": {
"Name": "Accounting Standards Codification",
"Paragraph": "9",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "450",
"URI": "https://asc.fasb.org/extlink&oid=121557415&loc=d3e14557-108349",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r731": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "20",
"Subparagraph": "(SAB Topic 5.Y.Q2)",
"Topic": "450",
"URI": "https://asc.fasb.org/extlink&oid=27011672&loc=d3e149879-122751",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r732": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "450",
"URI": "https://asc.fasb.org/topic&trid=2127136",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r733": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(4)(ii))",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=126975872&loc=SL124442526-122756",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r734": {
"Name": "Accounting Standards Codification",
"Paragraph": "1A",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.13-01(a)(4)(iii))",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=126975872&loc=SL124442526-122756",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r735": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495323-112611",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r736": {
"Name": "Accounting Standards Codification",
"Paragraph": "1D",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123466505&loc=SL123495340-112611",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r737": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21463-112644",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r738": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130551-203045",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r739": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "606",
"URI": "https://asc.fasb.org/extlink&oid=126920106&loc=SL49130545-203045",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r74": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "360",
"URI": "https://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r740": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "705",
"URI": "https://asc.fasb.org/topic&trid=2122478",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r741": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(d)(ii)",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r742": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r743": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r744": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)(3)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r745": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r746": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(ii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r747": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r748": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r749": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(01)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r75": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.CC)",
"Topic": "360",
"URI": "https://asc.fasb.org/extlink&oid=27011434&loc=d3e125687-122742",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r750": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(02)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r751": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(03)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r752": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)(iv)(04)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r753": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r754": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(ii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r755": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r756": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(01)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r757": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(02)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r758": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(2)(iii)(03)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r759": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r76": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "360",
"URI": "https://asc.fasb.org/topic&trid=2155823",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r760": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r761": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r762": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)(2)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r763": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r764": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(ii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r765": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(iii)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r766": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(iv)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r767": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)(2)(v)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r768": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(i)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r769": {
"Name": "Accounting Standards Codification",
"Paragraph": "12",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32687-109319",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r77": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)(2)",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=6394359&loc=d3e17939-110869",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r770": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32537-109319",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r771": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=121826272&loc=d3e32621-109319",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r772": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 6.I.Fact.4)",
"Topic": "740",
"URI": "https://asc.fasb.org/extlink&oid=122134291&loc=d3e330036-122817",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r773": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "808",
"URI": "https://asc.fasb.org/extlink&oid=6931272&loc=SL5834143-161434",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r774": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"SubTopic": "15",
"Topic": "815",
"URI": "https://asc.fasb.org/subtopic&trid=2229187",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r775": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "65",
"SubTopic": "40",
"Subparagraph": "(e)(3)",
"Topic": "815",
"URI": "https://asc.fasb.org/extlink&oid=126732423&loc=SL123482106-238011",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r776": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(bbb)",
"Topic": "820",
"URI": "https://asc.fasb.org/extlink&oid=126976982&loc=d3e19207-110258",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r777": {
"Name": "Accounting Standards Codification",
"Paragraph": "28",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(f)",
"Topic": "825",
"URI": "https://asc.fasb.org/extlink&oid=123596393&loc=d3e14064-108612",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r778": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "20",
"Subparagraph": "(b)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=123391704&loc=SL77918638-209977",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r779": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(a)(3)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918673-209980",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r78": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SAB Topic 5.P.4(b)(2))",
"Topic": "420",
"URI": "https://asc.fasb.org/extlink&oid=115931487&loc=d3e140904-122747",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r780": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918673-209980",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r781": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(g)(1)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918686-209980",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r782": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=128292326&loc=SL77918701-209980",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r783": {
"Name": "Accounting Standards Codification",
"Paragraph": "11",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "30",
"Subparagraph": "(a)",
"Topic": "842",
"URI": "https://asc.fasb.org/extlink&oid=123408481&loc=SL77919140-209958",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r784": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "852",
"URI": "https://asc.fasb.org/extlink&oid=124433192&loc=SL2890621-112765",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r785": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(b)",
"Topic": "852",
"URI": "https://asc.fasb.org/extlink&oid=124433192&loc=SL2890621-112765",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r786": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(1)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r787": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(2)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r788": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(c)(3)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107207-111719",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r789": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(1)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r79": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "440",
"URI": "https://asc.fasb.org/topic&trid=2144648",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r790": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(2)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r791": {
"Name": "Accounting Standards Codification",
"Paragraph": "4",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Subparagraph": "(b)(3)",
"Topic": "860",
"URI": "https://asc.fasb.org/extlink&oid=121570589&loc=d3e107314-111719",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r792": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "730",
"Topic": "912",
"URI": "https://asc.fasb.org/extlink&oid=6472174&loc=d3e58812-109433",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r793": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "320",
"Subparagraph": "(b)",
"Topic": "942",
"URI": "https://asc.fasb.org/extlink&oid=126980459&loc=d3e62557-112803",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r794": {
"Name": "Accounting Standards Codification",
"Paragraph": "4H",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "40",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=116884468&loc=SL65671331-158438",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r795": {
"Name": "Accounting Standards Codification",
"Paragraph": "13H",
"Publisher": "FASB",
"Section": "55",
"SubTopic": "40",
"Subparagraph": "(c)",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=126561865&loc=SL117783719-158441",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r796": {
"Name": "Accounting Standards Codification",
"Paragraph": "1B",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "825",
"Topic": "944",
"URI": "https://asc.fasb.org/extlink&oid=123600520&loc=SL75241803-196195",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r797": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(a)",
"Publisher": "SEC",
"Section": "1402",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r798": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(b)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(1)",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r799": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(b)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r8": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(31))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r80": {
"Name": "Accounting Standards Codification",
"Paragraph": "10",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "470",
"URI": "httphttps://asc.fasb.org/extlink&oid=12357013999376301&loc=d3e3536-108585"
}
},
"version": "2.1d3e1243-112600",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r800": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(b)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(3)",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r801": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(c)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)(i)",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r802": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(c)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)(ii)",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r803": {
"Name": "Regulation S-K (SK)",
"Number": "229",
"Paragraph": "(c)",
"Publisher": "SEC",
"Section": "1402",
"Subparagraph": "(2)(iii)",
"role": "http://www.xbrl.org/2009/role/commonPracticeRef"
},
"r81": {
"Name": "Accounting Standards Codification",
"Paragraph": "12A",
"Publisher": "FASB",
"Section": "45",
"SubTopic": "10",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=99376301&loc=SL5988623-112600",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r82": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123465755&loc=d3e1835-112601",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r83": {
"Name": "Accounting Standards Codification",
"Paragraph": "5",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "470",
"URI": "https://asc.fasb.org/extlink&oid=123465755&loc=SL6230698-112601",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r84": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "470",
"URI": "https://asc.fasb.org/topic&trid=2208564",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r85": {
"Name": "Accounting Standards Codification",
"Paragraph": "13",
"Publisher": "FASB",
"Section": "25",
"SubTopic": "10",
"Topic": "480",
"URI": "https://asc.fasb.org/extlink&oid=109262497&loc=d3e20148-110875",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r86": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21463-112644",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r87": {
"Name": "Accounting Standards Codification",
"Paragraph": "3",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21475-112644",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r88": {
"Name": "Accounting Standards Codification",
"Paragraph": "6",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21506-112644",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r89": {
"Name": "Accounting Standards Codification",
"Paragraph": "7",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21521-112644",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r9": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.5-02(32))",
"Topic": "210",
"URI": "https://asc.fasb.org/extlink&oid=120391452&loc=d3e13212-122682",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r90": {
"Name": "Accounting Standards Codification",
"Paragraph": "8",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=126973232&loc=d3e21538-112644",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r91": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "10",
"Subparagraph": "(SX 210.3-04)",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=120397183&loc=d3e187085-122770",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r92": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "S99",
"SubTopic": "50",
"Topic": "505",
"URI": "https://asc.fasb.org/extlink&oid=6784392&loc=d3e188667-122775",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r93": {
"Name": "Accounting Standards Codification",
"Publisher": "FASB",
"Topic": "505",
"URI": "https://asc.fasb.org/topic&trid=2208762",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r94": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "20",
"Topic": "715",
"URI": "https://asc.fasb.org/extlink&oid=123447040&loc=d3e1928-114920",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r95": {
"Name": "Accounting Standards Codification",
"Paragraph": "1",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(a)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5047-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r96": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(c)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r97": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(d)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r98": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
},
"r99": {
"Name": "Accounting Standards Codification",
"Paragraph": "2",
"Publisher": "FASB",
"Section": "50",
"SubTopic": "10",
"Subparagraph": "(e)(1)",
"Topic": "718",
"URI": "https://asc.fasb.org/extlink&oid=128089324&loc=d3e5070-113901",
"role": "http://fasb.org/us-gaap/role/ref/legacyRef"
}
},
"version": "2.2"
}






 Similar to pharmaceuticals, novel PDTs undergo rigorous clinical development via clinical trials designed to seek FDA authorization to safely and effectively treat disease. Similar to wellness apps, PDTs utilize digital technology to
Similar to pharmaceuticals, novel PDTs undergo rigorous clinical development via clinical trials designed to seek FDA authorization to safely and effectively treat disease. Similar to wellness apps, PDTs utilize digital technology to