UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16 OF THE
SECURITIES EXCHANGE ACT OF 1934
For the month of September 2025
Commission File Number
(Translation of registrant’s name into English)
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F:
Form 20-F x Form 40-F ¨
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ¨
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ¨
On September 12, 2025, Biodexa Pharmaceuticals PLC (the “Company”) issued a press release announcing its financial and operating results for the six months ended June 30, 2025. A copy of the press release is attached hereto as Exhibit 99.1 and is incorporated herein by reference.
The information set forth in the press release under the headings “Chief Executive’s Review,” “1H25 Financial Review” and the Consolidated Unaudited Interim Financial Statements for the six month period ended June 30, 2025 and the notes thereto shall be deemed to be incorporated by reference into the registration statements on Form S-8 (Registration Number 333-209365) and Form F-3 (Registration Number 333-267932) of the Company (including any prospectuses forming part of such registration statements) and be a part thereof from the date on which this report is filed, to the extent not superseded by documents or reports subsequently filed or furnished.
Other than as set forth herein, the information in the attached Exhibit 99.1 is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that Section, nor shall it be deemed incorporated by reference in any filing made by the Company under the Securities Act of 1933, as amended, or the Exchange Act, except as otherwise set forth herein or as shall be expressly set forth by specific reference in such a filing.
SUBMITTED HEREWITH
Attached to the Registrant’s Form 6-K filing for the month of September 2025, and incorporated by reference herein, is:
|
Exhibit No. |
Description | |
| 99.1 | Press Release, dated September 12, 2025 | |
| 101.INS | XBRL Instance Document - the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document. | |
| 101.SCH | INLINE XBRL Taxonomy Extension Schema Document. | |
| 101.DEF | INLINE XBRL Taxonomy Extension Calculation Linkbase Document. | |
| 101.CAL | INLINE XBRL Taxonomy Extension Definition Linkbase Document. | |
| 101.LAB | INLINE XBRL Taxonomy Extension Label Linkbase Document. | |
| 101.PRE | INLINE XBRL Taxonomy Extension Presentation Linkbase Document. | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| Biodexa Pharmaceuticals PLC | ||
| Date: September 12, 2025 | By: | /s/ Stephen Stamp |
| Stephen Stamp | ||
| Chief Executive Officer and Chief Financial Officer | ||
Exhibit 99.1
September 12, 2025
Biodexa Pharmaceuticals PLC
(“Biodexa” or the “Company”)
Interim results for the six months ended June 30, 2025
Biodexa Pharmaceuticals PLC (Nasdaq: BDRX), a clinical stage biopharmaceutical company developing a pipeline of innovative products for the treatment of diseases with unmet medical needs, today announces its unaudited interim results for the six months ended June 30, 2025 which will also be made available on the Company’s website at www.biodexapharma.com.
OPERATIONAL HIGHLIGHTS
The Company announced the following in the six months ended June 30, 2025:
| · | Allowance by the US Patent and Trademark Office of patent application No. 17/391-495 “Oral Rapamycin Nanoparticle Preparations and Use”, exclusively licensed to the Company by Emtora Biosciences . |
| · | Appointment of Precision for Medicine, LLC as the clinical research organization to conduct the European component of the upcoming registrational Phase 3 study of eRapa in FAP. |
| · | A successful Type C meeting with the US Food and Drug Administration regarding the protocol for the Company’s registrational Phase 3 study of eRapa in FAP. |
| · | Orphan Drug Designation granted for eRapa in FAP by the European Commission. |
| · | Recruitment of the first patient in a Phase 2a study of tolimidone in Type 1 Diabetes in an Investigator Initiated Trial conducted by the University of Alberta Diabetes Institute. |
| · | Selection of ‘Serenta’ as the brand name for its Phase 3 clinical study of eRapa in FAP together with launch of a dedicated website, www.serentatrial.com, to provide information and resources for patients, caregivers, and healthcare professionals. |
| · | Activation of the first clinical study site in the US for its Serenta trial in patients with FAP. |
Post period end:
| · | Filing of a Clinical Trial Application with the European Medicines Agency for its Serenta trial in patients with FAP. |
| · | Enrolment of first patients in the Serenta trial by the Pan American Center for Oncology Trials in San Juan, Puerto Rico. |
| 2 |
FINANCIAL HIGHLIGHTS
| · | Signing of a $35 million Equity Line of Credit with C/M Capital Master Fund LP, or C/M, pursuant to which the Company has the right, but not the obligation, to sell to C/M, and C/M is obligated to purchase newly issued ADSs for a period of 36 months. |
| · | The Company’s collaboration partner, Emtora Biosciences, was awarded an additional grant of $3.0 million from the Cancer Prevention & Research Institute of Texas, bringing the total of non-dilutive grant funding to $20.0 million in support of the registrational Phase 3 program of eRapa in FAP. |
| · | R&D costs decreased to £1.67 million in 1H25 (1H24: £2.19 million) reflecting a reduction in spend on the MAGIC-G1 study in recurrent glioblastoma and pre-clinical studies offset by an increase in expenditure (net of CPRIT grant income) on MTX230 (eRapa). |
| · | Administrative costs increased to £2.38 million (1H24: £2.03 million) as a result of a foreign exchange charge offset by a reduction in transaction related costs. |
| · | Net cash used in operating activities (after changes in working capital) in 1H25 was £3.30 million (1H24: £4.81 million). |
| · | The Company’s cash balance at June 30, 2025 was £4.04 million. |
Commenting, Stephen Stamp, CEO and CFO, said “The first half was extremely productive. Having secured Fast Track designation and successfully negotiated our way through a Type C meeting with the FDA, in August we enrolled the first patients into our pivotal Serenta trial of eRapa in FAP. In parallel, we secured orphan designation from the European Commission for eRapa in FAP in Europe and filed a Clinical Trial Application with the EMA, which sets us up to begin enrolment in the Serenta trial in Europe in the fourth quarter”.
| 3 |
CHIEF EXECUTIVE’S REVIEW
Our main focus in the first half of 2025 was on preparatory activities for the start of our registrational Phase 3 trial of eRapa in Familial Adenomatous Polyposis (“FAP”).
R&D update
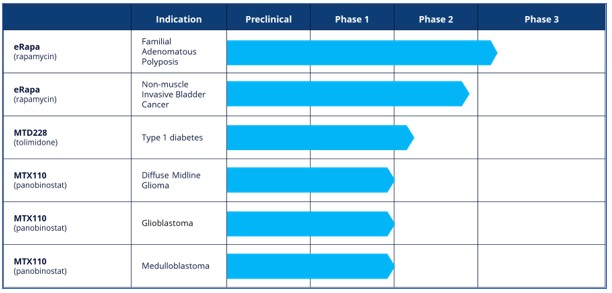
In the first half of 2025 we materially advanced our R&D pipeline, moving eRapa in FAP into Phase 3 and tolimidone for Type 1 Diabetes (“T1D) into Phase 2:

eRapa
eRapa is a proprietary oral formulation of rapamycin, also known as sirolimus. Rapamycin is an mTOR (mammalian Target Of Rapamycin) inhibitor. mTOR has been shown to have a significant role in the signalling pathway that regulates cellular metabolism, growth and proliferation and is activated during tumorgenesis. Rapamycin is approved in the US for organ rejection in renal transplantation as Rapamune®(Pfizer). Through the use of nanotechnology and pH sensitive polymers, eRapa is designed to address the poor bioavailability, variable pharmacokinetics and toxicity generally associated with the currently available forms of rapamycin. eRapa is protected by a number of issued patents which extend through 2035, with other pending applications potentially providing further protection beyond 2035.
Familial Adenomatous Polyposis (“FAP”)
FAP is an orphan indication characterized by a proliferation of polyps in the colon and/or rectum, usually occurring in mid-teens. There is no approved therapeutic option for treating FAP patients, for whom active surveillance and surgical resection of the colon and/or rectum remain the standard of care. If untreated, FAP almost always leads to cancer of the colon and/or rectum. There is a significant hereditary component to FAP with a reported incidence of one in 5,000 to 10,000 in the US and one in 11,300 to 37,600 in Europe. eRapa has received Orphan Designation in the US and in Europe. Importantly, mTOR has been shown to be over-expressed in FAP polyps – thereby underscoring the rationale for using a potent and safe mTOR inhibitor like eRapa to treat FAP.
An open-label Phase 2 study (NCT04230499) was conducted by Emtora in seven U.S. centres of excellence in 30 adult patients. Patients were sequentially enrolled into three dosing cohorts of 10 patients each for a 12-month treatment period: 0.5mg every other day (Cohort 1), 0.5mg daily every other week (Cohort 2), and 0.5mg daily (Cohort 3). Upper and lower endoscopic surveillance occurred at baseline and after six months. Primary endpoints were safety and tolerability of eRapa and percentage change from baseline in polyp burden, as measured by the aggregate of all polyp diameters.
| 4 |
In May 2024 and June 2024, results of the Phase 2 study at six months and 12 months, respectively, were presented at prestigious scientific meetings by Carol Burke, MD, the Principal Investigator. In summary, at six months, eRapa appeared safe and well-tolerated with a significant 24% reduction in the total polyp burden at six months compared with baseline and an overall 83% non-progression rate. At 12 months, 21 of 28 (75%) patients were deemed to be non-progressors with a median reduction in polyp burden of 17%. In Cohort 2, the dosage regimen for Phase 3, eight of nine (89)% of patients were deemed non-progressors at 12 months with a median reduction in polyp burden of 29%. Over the course of 12 months, there were four related Grade 3 or higher and one related Serious Adverse Event reported during the trial and 95% compliance rate at 12 months. One patient was removed from the trial due to non-compliance.
The Phase 3 registrational study (NCT06950385) is a double-blind placebo-controlled design, recruiting 168 high risk patients diagnosed with germline or phenotypic FAP. The primary clinical endpoint is first progression free survival event which will comprise composite endpoints including major surgery. We had a successful ‘Type C’ meeting with the FDA in January 2025 to finalise the protocol. The first clinical site was initiated in June 2025 and the first patients enrolled by the Pan American Center for Oncology Trials in San Juan, Puerto Rico in August 2025. Europe is following closely behind; our contract research organisation, Precision for Medicine, was appointed in March 2025. Orphan Drug Designation for eRapa in FAP was granted by the European Commission in May 2025. A Clinical Trial Application was filed with the European Medicines Agency for the Serenta trial in July 2025 which, if approved, will facilitate the start of patient recruitment in Europe in 4Q25.
Non-muscle Invasive Bladder Cancer (“NMIBC”)
NMIBC refers to tumors found in the tissue that lines the inner surface of the bladder. The most common treatment is transurethral resection of the bladder tumor followed by intravesical Bacillus Calmette-Guerin (“BCG”) with chemotherapy depending upon assessment of risk of recurrence. NMIBC is the fourth most common cancer in men with an incidence of 10.1 per 100,000 and 2.5 per 100,000 in women.
The ongoing multi-centre, double-blind, placebo-controlled Phase 2 study in NMIBC (NCT04375813) is fully enrolled at 166 patients with primary endpoints of safety/tolerability and relapse free survival after 12 months of treatment. The study, which is supported by a $3.0 million non-dilutive grant from the National Cancer Institute, part of the National Institutes of Health, was transferred to the University of Texas, San Antonio as an Investigator Initiated Trial and is expected to read out in mid-2026.
MTD228 - Tolimidone
Tolimidone was originally discovered by Pfizer and was developed through Phase 2 for the treatment of gastric ulcers. Pfizer undertook a broad pre-clinical program to characterize the pharmacology, pharmacokinetics, metabolism and toxicology of tolimidone. Pfizer discontinued development of the drug due to lack of efficacy for that indication.
Tolimidone is a selective activator of the enzyme Lyn kinase which increases phosphorylation of insulin substrate-1, thereby amplifying the signalling cascade initiated by the binding of insulin to its receptor.
| 5 |
Type 1 Diabetes (“T1D”)
Tolimidone’s potential utility in T1D has been demonstrated by several preclinical studies conducted by the University of Alberta, where Lyn kinase was identified as a key factor for beta cell survival and proliferation in in vitro and in vivo models. Most importantly, tolimidone appeared to induce proliferation in beta cells isolated from human cadavers. From a mechanism of action perspective, tolimidone has been shown to both prevent beta cell degradation and to stimulate beta cell proliferation. In a meta analysis of 1,202 articles and 193 studies, the incidence of T1D was shown to be 15 per 100,000 with a prevalence of 9.5 per 10,000 of the population.
As a first step in the continued clinical development of tolimidone, a Phase 2a Investigator Initiated Trial (IIT) at the University of Alberta Diabetes Institute (NCT06474598) is designed to establish the minimum effective dose of tolimidone in patients with T1D. The study, enrolled the first patient in June 2025 and is expected to recruit 12 patients initially across three dose groups. The study will measure C-peptide levels (a marker for insulin) and HbA1c (a marker for blood glucose) after three months compared with baseline and the number of hyperglycemic events.
MTX110
MTX110 is a solubilised formulation of the histone deacetylase (HDAC) inhibitor, panobinostat. This proprietary formulation enables delivery of the product via convection-enhanced delivery (CED) at chemotherapeutic doses directly to the site of the brain tumor, by-passing the blood-brain barrier and potentially avoiding systemic toxicity. All three types of brain cancer being studied are orphan.
Recurrent Glioblastoma (“rGBM”)
Our Phase 1 MAGIC-G1 study (NCT05324501) of MTX110 in rGBM has completed the dose escalation part of the study with the recruitment of the fourth patient in Cohort A. Overall survival was reported as between 11 and 12 months. Glioblastoma virtually always recurs with median Progression Free Survival of 1.5–6.0 months and median Overall Survival of 2.0–9.0 months.
Diffuse Midline Glioma (“DMG”)
In February 2024 we announced headline data from a Phase 1 IIT study conducted by Columbia University in newly diagnosed patients with DMG. As this was the first ever study of repeated infusions to the pons via an implanted CED catheter, the primary objective of the study was safety and tolerability and, accordingly, the number of infusions was limited to two, each of 48 hours, seven days apart in nine patients. One patient suffered a severe adverse event assessed by the investigators as not related to the study drug. Although not powered to reliably demonstrate efficacy, median Overall Survival of patients in the study was 16.5 months compared with median survival rate in a cohort of 316 cases of 10.0 months.
Study investigators subsequently presented the results of the trial at the 21st International Symposium on Pediatric Neuro-Oncology (ISPNO 2024) in Philadelphia, PA.
Medulloblastoma
An IIT Phase I study of MTX110 in medulloblastoma remains ongoing at the University of Texas.
Due to resource constraints, MTX110 has been de-prioritised and there are no current development activities being undertaken.
| 6 |
Financing
Equity Line of Credit
In January 2025, we entered into a securities purchase agreement, or equity line of credit (“ELOC”), with the newly formed C/M. Under the terms of the ELOC, we have the right, but not the obligation, to sell to C/M from time to time up to $35 million of newly issued ADSs over a 36-month period, unless the ELOC is terminated. As consideration for the execution and delivery of the ELOC, we agreed to pay a commitment fee (“Commitment Fee”) of $875,000 in cash, of which (i) $612,500 was to be paid to C/M on signing the ELOC and (ii) the balance was to be paid pro rata, simultaneously with the delivery of any ADSs sold under the ELOC. We had the right to issue ADSs representing the value of the applicable portion of the Commitment Fee. We paid the initial Commitment Fee of $612,500 through the issuance of 140,080 Depositary Shares to the Purchaser.
We may direct C/M to purchase a specified number of ADSs not to exceed $2.5 million on any given day, at a price based on a formula, typically 95% of the closing price on the prior day. As of June 30, 2025, the Company had raised gross proceeds of $8.56 million from the ELOC.
Warrant Inducement
In May 2025 we entered into letter agreements with certain holders of outstanding Series E, Series H, Series J and Series K warrants to reduce the exercise price of such warrants to $0.31 per share. The holders exercised an aggregate of 200,433 warrants representing the same number of ADSs. We received gross proceeds of approximately $62,000, before offering expenses. The Company did not issue new warrants to replace the exercised warrants and did not engage a placement agent to facilitate the transaction.
| 7 |
1H25 FINANCIAL REVIEW
The unaudited results for the six months ended June 30, 2025 are discussed below:
Key performance indicators (KPIs):
| 1H 2025 | 1H 2024 | Change | |
| R&D costs | £1.67m | £2.19m | (24)% |
| R&D as % of operating costs | 41% | 52% | (11)% |
| Net cash inflow/(outflow) for the period | £2.37m | £(0.92)m | N/M |
Biodexa’s KPIs focus on the key areas of operating results, R&D spend and cash management. These measures provide information on the core R&D operations. Additional financial and non-financial KPIs may be adopted in due course.
Revenues
Revenue for both periods was £Nil. The last of the Company’s R&D collaborations concluded in September 2023.
Research and Development
R&D costs for 1H25 and 1H24, analysed by development project indication were as follows:
| Six months ended June 30 | 2025 | 2024 | |||||||
| £’000 | £’000 | ||||||||
| eRapa | |||||||||
| Familial Adenomatous Polyposis | 251 | 175 | |||||||
| Non-muscle Invasive Bladder Cancer | 127 | 95 | |||||||
| Total eRapa | 378 | 270 | |||||||
| Tolimidone | |||||||||
| Type 1 Diabetes | 270 | 383 | |||||||
| Total tolimidone | 270 | 383 | |||||||
| MTX110 (panobinostat) | |||||||||
| Diffuse Midline Glioma | - | - | |||||||
| Recurrent Glioblastoma | 14 | 360 | |||||||
| Medulloblastoma | - | - | |||||||
| Total MTX110 (panobinostat) | 14 | 360 | |||||||
| Other preclinical | 1 | 104 | |||||||
| R&D overheads | 1,002 | 1,072 | |||||||
| Total R&D | 1,665 | 2,189 | |||||||
R&D costs in 1H25 reduced by £0.52 million, or 24%, to £1.67 million compared with £2.19 million in 1H24. The percentage of R&D costs as a percentage of total operating costs decreased to 41% in the period from 52%. The reduction in R&D costs in 1H25 reflects a reduction in spend of £0.56 million on the MAGIC-G1 study in rGBM and preclinical studies offset by an increase in expenditure (net of CPRIT grant income) on MTX230 (eRapa). The percentage of MTX230 (eRapa) costs that were able to be offset against grant funding during the period was 82% and we anticipate that over the life of the grant this will be 67%.
| 8 |
Administrative Costs
Administrative costs in 1H25 increased by £0.34 million, or 17%, to £2.38 million from £2.03 million in 1H24. The increase in administrative costs in 1H25 is driven by a foreign exchange charge of £0.40 million in the period compared to a £0.06 million gain in 2024. This was offset by a reduction in transaction related costs of £0.13 million. Transaction costs in the period included a £0.37 million non- cash upfront commitment fee for the ELOC.
Finance Income and Expense
Finance income in 1H25 and 1H24 included gains in respect of an equity settled derivative financial liability of £0.15 million (1H24: £0.75 million). The gains arose as a result of the fall in the Biodexa share price. In addition, the Company earned interest on cash deposits.
Finance expense in the period related to lease liabilities, discounted interest on deferred consideration and interest on the promissory note issued in December 2024.
Cash Flows
Cash outflows from operating activities in 1H25 were £3.30 million compared to £4.81 million in 1H24, driven by a net loss of £3.81 million (1H24: £3.31 million) and after negative working capital of £0.04 million (1H24: negative £0.87 million) and other positive non-cash items totalling £0.24 million (1H24: negative £0.63 million).
Net cash used in investing activities in 1H25 of £0.34 million (1H24: outflow £0.75 million) resulted from the payment of deferred consideration on the eRapa licence of £0.37 million (1H24: £0.85 million) offset by £0.04 million of interest received (1H24: £0.10 million).
Net cash generated from financing activities in 1H25 was £6.01 million (1H24: inflow £4.65 million), which was driven by £6.20 million proceeds from share issuances under the ELOC, £0.05 million net proceeds from a warrant inducement, offset by loan repayments (including interest) of £0.15 million and payments on lease liabilities of £0.10 million.
Overall, cash increased by £2.37 million in 1H25 compared to a decrease of £0.92 million in 1H24. This resulted in a cash balance at June 30, 2025 of £4.04 million compared with £5.05 million at June 30, 2024 and £1.67 million at December 31, 2024.
Going concern
Biodexa has experienced net losses and significant cash outflows from cash used in operating activities over the past years as it develops its portfolio. For the six months to June 30, 2025, the Group incurred a consolidated loss from operations of £3.81 million (1H24: £3.31 million) and negative cash flows from operating activities of £3.30 million (1H24: £4.81 million). As of June 30, 2025, the Group had accumulated deficit of £154.13 million.
The Group’s future viability is dependent on its ability to raise cash from financing activities to finance its development plans until commercialisation, generate cash from operating activities and to successfully obtain regulatory approval to allow marketing of its development products. The Group’s failure to raise capital as and when needed could have a negative impact on its financial condition and ability to pursue its business strategies.
| 9 |
The Directors believe there are adequate options and time available to secure additional financing for the Group and after considering the uncertainties, the Directors consider it is appropriate to continue to adopt the going concern basis in preparing these financial statements. The Group’s consolidated financial statements have therefore been presented on a going concern basis, which contemplates the realisation of assets and the satisfaction of liabilities in the normal course of business.
As at June 30, 2025, the Group had cash and cash equivalents of £4.03 million. The Directors have prepared cash flow forecasts and considered the cash flow requirement for the Group for the next three years including the period 12 months from the date of approval of this interim financial information. These forecasts show that further financing will be required before the second quarter of 2026 assuming, inter alia, that certain development programs and other operating activities continue as currently planned. Provided certain conditions are met, including the price of the Company’s ADSs quoted on NASDAQ being above $1.00, the Company may direct C/M to purchase ADSs and receive proceeds in accordance with a formula price for up to 36 months from January 2025. There is no guarantee that the Company will be able to use the ELOC to the extent necessary to finance the Company’s operations.
In the Directors’ opinion, the environment for financing of small and micro-cap biotech companies remains challenging. While this may present acquisition and/or merger opportunities with other companies with limited or no access to financing, as noted above, any attendant financings by Biodexa are likely to be dilutive. The Directors continue to evaluate financing options, including those connected to acquisitions and/or mergers, potentially available to the Group. Any alternatives considered are contingent upon the agreement of counterparties and accordingly, there can be no assurance that any of alternative courses of action to finance the Group would be successful.
This requirement for additional financing in the short term represents a material uncertainty that may cast significant doubt upon the Group’s ability to continue as a going concern. Should it become evident in the future that there are no realistic financing options available to the Group which are actionable before its cash resources run out then the Group will no longer be a going concern. In such circumstances, we would no longer be able to prepare financial statements under paragraph 25 of IAS 1. Instead, the financial statements would be prepared on a liquidation basis and assets would be stated at net realizable value and all liabilities would be accelerated to current liabilities.
Stephen Stamp
Chief Executive Officer and Chief Financial Officer
| 10 |
Consolidated Statements of Comprehensive Income
For the six month period ended June 30
| Note | 2025 unaudited £’000 | 2024 unaudited £’000 | ||||||||||
| Revenue | ||||||||||||
| Other income | ||||||||||||
| Research and development costs | ( | ) | ( | ) | ||||||||
| Administrative costs | ( | ) | ( | ) | ||||||||
| Loss from operations | ( | ) | ( | ) | ||||||||
| Finance income | 3 | |||||||||||
| Finance expense | 3 | ( | ) | ( | ) | |||||||
| Loss before tax | ( | ) | ( | ) | ||||||||
| Taxation | ||||||||||||
| Loss for the period attributable to the owners of the parent | ( | ) | ( | ) | ||||||||
Items that will or may be reclassified subsequently to profit or loss: | ||||||||||||
Exchange gain arising on translation of foreign operations | ||||||||||||
| Total comprehensive loss attributable to the owners of the parent | ( | ) | ( | ) | ||||||||
| Loss per share | ||||||||||||
| Basic and diluted loss per ordinary share – £ | 4 | £ | ) | £ | ) | |||||||
The accompanying notes form part of these financial statements
| 11 |
Consolidated Statements of Financial Position
| Note | As at June 30, 2025 £’000 | As at December 31, 2024 £’000 | ||||||||||
| Assets | ||||||||||||
| Non-current assets | ||||||||||||
| Property, plant and equipment | ||||||||||||
| Intangible assets | ||||||||||||
| Total Non-Current Assets | ||||||||||||
| Current assets | ||||||||||||
| Trade and other receivables | ||||||||||||
| Taxation | ||||||||||||
| Cash and cash equivalents | ||||||||||||
| Total Current Assets | ||||||||||||
| Total assets | ||||||||||||
| Liabilities | ||||||||||||
| Non-current liabilities | ||||||||||||
| Deferred consideration | ||||||||||||
| Borrowings | ||||||||||||
| Total Non-Current Liabilities | ||||||||||||
| Current liabilities | ||||||||||||
| Trade and other payables | ||||||||||||
| Deferred consideration | ||||||||||||
| Borrowings | ||||||||||||
| Derivative financial liability | ||||||||||||
| Total current Liabilities | ||||||||||||
| Total liabilities | ||||||||||||
| Issued capital and reserves attributable to owners of the parent | ||||||||||||
| Share capital | 5 | |||||||||||
| Share premium | ||||||||||||
| Merger reserve | ||||||||||||
| Warrant reserve | ||||||||||||
| Foreign exchange reserve | ||||||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||||||
| Total equity | ||||||||||||
| Total equity and liabilities | ||||||||||||
The accompanying notes form part of these financial statements
| 12 |
Consolidated Statements of Cash Flows
For the six month period ended June 30
| Note | 2025 unaudited £’000 | 2024 unaudited £’000 | ||||||||||
| Cash flows from operating activities | ||||||||||||
| Loss for the period | ( | ) | ( | ) | ||||||||
| Adjustments for: | ||||||||||||
| Depreciation of property, plant and equipment | ||||||||||||
| Depreciation of right of use asset | ||||||||||||
| Amortisation of intangible fixed asset | ||||||||||||
| Impairment of commission paid in advance on ELOC | ||||||||||||
| Finance income | 3 | ( | ) | ( | ) | |||||||
| Finance expense | 3 | |||||||||||
| Share-based payment expense | ||||||||||||
| Taxation | 4 | ( | ) | ( | ) | |||||||
| Foreign exchange losses | ( | ) | ||||||||||
| Cash flows from operating activities before changes in working capital | ( | ) | ( | ) | ||||||||
| Decrease/(Increase) in trade and other receivables | ( | ) | ||||||||||
| (Decrease)/Increase in trade and other payables | ( | ) | ||||||||||
| Cash used in operations | ( | ) | ( | ) | ||||||||
| Taxes receipts | ||||||||||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||||||
| 13 |
Consolidated Statements of Cash Flows (continued)
For the six month period ended June 30
| Note | 2025 unaudited £’000 | 2024 unaudited £’000 | ||||||||||
| Investing activities | ||||||||||||
| Purchases of property, plant and equipment | ( | ) | ||||||||||
| Purchase of intangible assets | ( | ) | ( | ) | ||||||||
| Interest received | ||||||||||||
| Net cash generated from/(used in) investing activities | ( | ) | ( | ) | ||||||||
| Financing activities | ||||||||||||
| Interest paid | ( | ) | ||||||||||
| Amounts paid on lease liabilities | ( | ) | ( | ) | ||||||||
| Repayment of Promissory Note | ( | ) | ||||||||||
| Share issues including warrants, net of costs | 5 | |||||||||||
| Net cash generated from/(used in) financing activities | ||||||||||||
| Net increase/(decrease) in cash and cash equivalents | ( | ) | ||||||||||
| Cash and cash equivalents at beginning of period | ||||||||||||
| Cash and cash equivalents at end of period | ||||||||||||
The accompanying notes form part of these financial statements
| 14 |
Consolidated Statements of Changes in Equity (unaudited)
| Note | Share capital £’000 | Share premium £’000 | Merger reserve £’000 | Warrant reserve £’000 | Foreign exchange reserve £’000 | Accumulated deficit £’000 | Total equity £’000 | |||||||||||||||||||||||||
| At January 1, 2025 | ( | ) | ||||||||||||||||||||||||||||||
| Loss for the period | ( | ) | ( | ) | ||||||||||||||||||||||||||||
| Foreign exchange translation | ||||||||||||||||||||||||||||||||
| Total comprehensive loss | ( | ) | ( | ) | ||||||||||||||||||||||||||||
| Transactions with owners: | ||||||||||||||||||||||||||||||||
| Shares issued under ELOC agreement | 5 | |||||||||||||||||||||||||||||||
| Costs associated with ELOC agreement | ( | ) | ||||||||||||||||||||||||||||||
| Shares issued on 15 May 2024 | 5 | |||||||||||||||||||||||||||||||
| Costs associated with share issue on 15 May 2024 | ( | ) | ( | ) | ||||||||||||||||||||||||||||
| Share-based payment charge | ||||||||||||||||||||||||||||||||
| Total contribution by and distributions to owners | ||||||||||||||||||||||||||||||||
| At June 30, 2025 | ( | ) | ||||||||||||||||||||||||||||||
| Note | Share capital £’000 | Share premium £’000 | Merger reserve £’000 | Warrant reserve £’000 | Accumulated deficit £’000 | Total equity £’000 | ||||||||||||||||||||||
| At January 1, 2024 | ( | ) | ||||||||||||||||||||||||||
| Loss for the period | ( | ) | ( | ) | ||||||||||||||||||||||||
| Total comprehensive loss | ( | ) | ( | ) | ||||||||||||||||||||||||
| Transactions with owners: | ||||||||||||||||||||||||||||
| Shares issued on February 15, 2023 | 5 | |||||||||||||||||||||||||||
| Costs associated with share issue on February 15, 2023 | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||
| Shares issued on May 26, 2023 | 5 | ( | ) | |||||||||||||||||||||||||
| Costs associated with share issue on May 26, 2023 | ||||||||||||||||||||||||||||
| Share-based payment charge | ||||||||||||||||||||||||||||
| Total contribution by and distributions to owners | ||||||||||||||||||||||||||||
| At June 30, 2024 | ( | ) | ||||||||||||||||||||||||||
The accompanying notes form part of these financial statements
| 15 |
Notes Forming Part of The Consolidated Unaudited Interim Financial Information
For the six month period ended June 30, 2025
| 1. | Basis of preparation |
The unaudited interim consolidated financial information for the six months ended June 30, 2025 has been prepared following the recognition and measurement principles of the International Financial Reporting Standards, International Accounting Standards and Interpretations (collectively IFRS) issued by the International Accounting Standards Board (IASB), and as adopted by the UK and in accordance with International Accounting Standard 34 Interim Financial Reporting (‘IAS 34’). The interim consolidated financial information does not include all the information and disclosures required in the annual financial information and should be read in conjunction with the audited financial statements for the year ended December 31, 2024.
The accounting policies adopted are consistent with those of the previous financial year and corresponding interim reporting periods.
Book values approximate to fair value at June 30, 2025, June 30, 2024 and 31 December 31, 2024.
The condensed interim financial information contained in this interim statement does not constitute statutory financial statements as defined by section 434(3) of the Companies Act 2006. The condensed interim financial information has not been audited. The comparative financial information for the six months ended June 30, 2024 and the year ended December 31, 2024 in this interim financial information does not constitute statutory financial statements for that period or year. The statutory financial statements for December 31, 2024 have been delivered to the UK Registrar of Companies. The auditor’s report on those accounts was unqualified and did not contain a statement under section 498(2) or 498(3) of the Companies Act 2006. The auditor’s report did draw attention to a material uncertainty related to going concern and the requirement, as of the date of the report, for additional funding to be raised by the Company by the fourth quarter of 2025.
Biodexa Pharmaceutical’s annual reports may be downloaded from the Company’s website at https://biodexapharma.com/investors/financial-reports-and-presentations/#financial-reports or a copy may be obtained from 1 Caspian Point, Caspian Way, Cardiff CF10 4DQ.
Going Concern – material uncertainty
Biodexa has experienced
net losses and significant cash outflows from cash used in operating activities over the past years as it develops its portfolio. For
the six months to June 30, 2025, the Group incurred a consolidated loss from operations of £
The Group’s future viability is dependent on its ability to raise cash from financing activities to finance its development plans until commercialisation, generate cash from operating activities and to successfully obtain regulatory approval to allow marketing of its development products. The Group’s failure to raise capital as and when needed could have a negative impact on its financial condition and ability to pursue its business strategies.
The Directors believe there are adequate options and time available to secure additional financing for the Group and after considering the uncertainties, the Directors consider it is appropriate to continue to adopt the going concern basis in preparing these financial statements. The Group's consolidated financial statements have been presented on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business.
As at June 30,
2025, the Group had cash and cash equivalents of £
| 16 |
In the Directors’ opinion, the environment for financing of small and micro-cap biotech companies remains challenging. While this may present acquisition and/or merger opportunities with other companies with limited or no access to financing, as noted above, any attendant financings by Biodexa are likely to be dilutive. The Directors continue to evaluate financing options, including those connected to acquisitions and/or mergers, potentially available to the Group. Any alternatives considered are contingent upon the agreement of counterparties and accordingly, there can be no assurance that any of alternative courses of action to finance the Company would be successful.
This requirement for additional financing in the short term represents a material uncertainty that may cast significant doubt upon the Group’s ability to continue as a going concern. Should it become evident in the future that there are no realistic financing options available to the Group which are actionable before its cash resources run out then the Group will no longer be a going concern. In such circumstances, we would no longer be able to prepare financial statements under paragraph 25 of IAS 1. Instead, the financial statements would be prepared on a liquidation basis and assets would be stated at net realizable value and all liabilities would be accelerated to current liabilities.
| 2. | Accounting for eRapa and CPRIT grant |
The License and Collaboration Agreement (“LCA”) entered into with Emtora in April 2024 meets the definition of a Joint Arrangement under IFRS 11, specifically related to the FAP program.
A jointly controlled escrow account was established on completion of the LCA. All FAP program transactions are processed through the escrow account, including the Company’s deposits of matching funds, as set out in the agreement, the receipt of grant funding from CPRIT and the payment of eligible R&D expenses. Although the CPRIT grant and R&D supplier contracts are with Emtora, the joint arrangement nature of the LCA results in Emtora being deemed to be acting as the Company’s agent. Accordingly, the Company recognises 100% of the grant and 100% of the R&D expenditure. The CPRIT grant recognised is on a 1 for 2 match. In accordance with the Company’s accounting policy, the grant, as it is the re-imbursement of directly related costs, is credited to R&D costs in the same period in The Statements of Comprehensive Income. The escrow account is recognised within prepayments, CPRIT grant received in advance is recognised within deferred revenue and any grant not yet received is recognised in accrued income.
In 1H25 the company
recognised R&D costs of £
| 17 |
The balances in relation to the FAP project as at June 30 were as follows:
June 30, 2025 unaudited £’000 | June 30, 2024 unaudited £’000 | |||||||
| Prepayments* | ||||||||
| Accrued revenue | ||||||||
| Deferred revenue | ( | ) | ||||||
* prepayment reflects only the escrow account balance
| 3. | Finance income and expense |
Six months ended June 30, 2025 unaudited £’000 | Six months ended June 30, 2024 unaudited £’000 | |||||||
| Finance income | ||||||||
| Interest received on bank deposits | ||||||||
| Other interest | ||||||||
| Gain on equity settled derivative financial liability | ||||||||
| Total finance income | ||||||||
The gain on the equity settled derivative financial liability in 1H25 and 1H24 arose as a result of the fall in the Biodexa share price.
Six months ended June 30, 2025 unaudited £’000 | Six months ended June 30, 2024 unaudited £’000 | |||||||
| Finance expense | ||||||||
| Interest expense on lease liabilities | ||||||||
| Interest expense on deferred consideration | ||||||||
| Other loans | ||||||||
| Total finance expense | ||||||||
| 18 |
| 4. | Loss per share |
Basic loss per share amounts are calculated by dividing the net loss for the period from continuing operations, attributable to ordinary equity holders of the parent company, by the weighted average number of ordinary shares outstanding during the period. As the Group made a loss for the period the diluted loss per share is equal to the basic loss per share.
Six months ended June 30, 2025 unaudited £’000 | Six months ended June 30, 2024 unaudited £’000 | |||||||
| Numerator | ||||||||
| Loss used in basic EPS and diluted EPS: | ( | ) | ( | ) | ||||
| Denominator | ||||||||
| Weighted average number of ordinary shares used in basic EPS | ||||||||
| Basic and diluted loss per share: | ) | £ | ) | |||||
At a General Meeting
on 11 June 2025, shareholders approved the subdivision and redesignation of the
The Company has considered the guidance set out in IAS 33 in calculating the denominator in connection with the issuance of Pre-Funded and Abeyance Shares. Management have recognised the warrants from the date of grant rather than the date of issue of the corresponding Ordinary Shares when calculating the denominator.
The Group has made a loss in the current and previous periods presented, and therefore the options and warrants are anti-dilutive. As a result, diluted earnings per share is presented on the same basis as basic earnings per share.
| 5. | Share capital and reserves |
| Authorised, allotted and fully paid – classified as equity | As at June 30, 2025 unaudited Number | As at June 30, 2025 unaudited £ | As at December 31, 2024 Number | As at December 31, 2024 £ | ||||||||||||
| Ordinary shares of £0.000001 each | ||||||||||||||||
| ‘A’ Deferred shares of £1 each | ||||||||||||||||
| ‘B’ Deferred shares of £0.001 each | ||||||||||||||||
| ‘C’ Deferred shares of £0.00005 each | ||||||||||||||||
| ‘D’ Deferred shares of £0.000001 each | ||||||||||||||||
| Total | ||||||||||||||||
Ordinary and deferred shares were recorded as equity.
At a General Meeting
on 11 June 2025, shareholders approved the subdivision and redesignation of the
| 19 |
As at June 30, 2025
and December 31, 2024 the Company had
In accordance with the Articles of Association for the Company adopted on 11 June 2025, the share capital of the Company consists of an unlimited number of ordinary shares of nominal value £ each. Ordinary and deferred shares were recorded as equity.
| 6. | Related party transaction |
The Directors consider there to be no related party transactions during the periods reported other than Directors Remuneration.
| 7. | Contingent liabilities |
The Company is in
a dispute with a former advisor over fees.
| 8. | Events after the reporting date |
On July 15, 2025,
the
20
Cover |
6 Months Ended |
|---|---|
Jun. 30, 2025 | |
| Cover [Abstract] | |
| Document Type | 6-K |
| Amendment Flag | false |
| Document Period End Date | Jun. 30, 2025 |
| Current Fiscal Year End Date | --12-31 |
| Entity File Number | 001-37652 |
| Entity Registrant Name | Biodexa Pharmaceuticals PLC |
| Entity Central Index Key | 0001643918 |
| Entity Address, Address Line One | 1 Caspian Point |
| Entity Address, Address Line Two | Caspian Way |
| Entity Address, City or Town | Cardiff |
| Entity Address, Country | GB |
| Entity Address, Postal Zip Code | CF10 4DQ |
Consolidated Statements of Comprehensive Income (Unaudited) - GBP (£) £ in Thousands |
6 Months Ended | |
|---|---|---|
Jun. 30, 2025 |
Jun. 30, 2024 |
|
| Profit or loss [abstract] | ||
| Revenue | ||
| Other income | 27 | |
| Research and development costs | (1,665) | (2,189) |
| Administrative costs | (2,378) | (2,034) |
| Loss from operations | (4,016) | (4,223) |
| Finance income | 180 | 839 |
| Finance expense | (135) | (49) |
| Loss before tax | (3,971) | (3,433) |
| Taxation | 165 | 125 |
| Loss for the period attributable to the owners of the parent | (3,806) | (3,308) |
| Items that will or may be reclassified subsequently to profit or loss: | ||
| Exchange gain arising on translation of foreign operations | 3 | |
| Total comprehensive loss attributable to the owners of the parent | £ (3,803) | £ (3,308) |
| Loss per share | ||
| Basic loss per share | £ (0.0002) | £ (0.001) |
| Diluted loss per share | £ (0.0002) | £ (0.001) |
Basis of preparation |
6 Months Ended | |||
|---|---|---|---|---|
Jun. 30, 2025 | ||||
| Basis Of Preparation | ||||
| Basis of preparation |
The unaudited interim consolidated financial information for the six months ended June 30, 2025 has been prepared following the recognition and measurement principles of the International Financial Reporting Standards, International Accounting Standards and Interpretations (collectively IFRS) issued by the International Accounting Standards Board (IASB), and as adopted by the UK and in accordance with International Accounting Standard 34 Interim Financial Reporting (‘IAS 34’). The interim consolidated financial information does not include all the information and disclosures required in the annual financial information and should be read in conjunction with the audited financial statements for the year ended December 31, 2024.
The accounting policies adopted are consistent with those of the previous financial year and corresponding interim reporting periods.
Book values approximate to fair value at June 30, 2025, June 30, 2024 and 31 December 31, 2024.
The condensed interim financial information contained in this interim statement does not constitute statutory financial statements as defined by section 434(3) of the Companies Act 2006. The condensed interim financial information has not been audited. The comparative financial information for the six months ended June 30, 2024 and the year ended December 31, 2024 in this interim financial information does not constitute statutory financial statements for that period or year. The statutory financial statements for December 31, 2024 have been delivered to the UK Registrar of Companies. The auditor’s report on those accounts was unqualified and did not contain a statement under section 498(2) or 498(3) of the Companies Act 2006. The auditor’s report did draw attention to a material uncertainty related to going concern and the requirement, as of the date of the report, for additional funding to be raised by the Company by the fourth quarter of 2025.
Biodexa Pharmaceutical’s annual reports may be downloaded from the Company’s website at https://biodexapharma.com/investors/financial-reports-and-presentations/#financial-reports or a copy may be obtained from 1 Caspian Point, Caspian Way, Cardiff CF10 4DQ.
Going Concern – material uncertainty
Biodexa has experienced net losses and significant cash outflows from cash used in operating activities over the past years as it develops its portfolio. For the six months to June 30, 2025, the Group incurred a consolidated loss from operations of £3.81million (1H24: loss £3.31 million) and negative cash flows from operating activities of £3.30 million (1H24 £4.81 million). As of June 30, 2025, the Group had accumulated deficit of £154.13 million.
The Group’s future viability is dependent on its ability to raise cash from financing activities to finance its development plans until commercialisation, generate cash from operating activities and to successfully obtain regulatory approval to allow marketing of its development products. The Group’s failure to raise capital as and when needed could have a negative impact on its financial condition and ability to pursue its business strategies.
The Directors believe there are adequate options and time available to secure additional financing for the Group and after considering the uncertainties, the Directors consider it is appropriate to continue to adopt the going concern basis in preparing these financial statements. The Group's consolidated financial statements have been presented on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business.
As at June 30, 2025, the Group had cash and cash equivalents of £4.04 million. The Directors have prepared cash flow forecasts and considered the cash flow requirement for the Group for the next three years including the period 12 months from the date of approval of this interim financial information. These forecasts show that further financing will be required before the second quarter of 2026 assuming, inter alia, that certain development programs and other operating activities continue as currently planned. Provided certain conditions are met, including the price of the Company’s ADSs quoted on NASDAQ being above $1.00, the Company may direct C/M to purchase ADSs and receive proceeds in accordance with a formula price for up to 36 months from January 2025. There is no guarantee that the Company will be able to use the ELOC to the extent necessary to finance the Company’s operations.
In the Directors’ opinion, the environment for financing of small and micro-cap biotech companies remains challenging. While this may present acquisition and/or merger opportunities with other companies with limited or no access to financing, as noted above, any attendant financings by Biodexa are likely to be dilutive. The Directors continue to evaluate financing options, including those connected to acquisitions and/or mergers, potentially available to the Group. Any alternatives considered are contingent upon the agreement of counterparties and accordingly, there can be no assurance that any of alternative courses of action to finance the Company would be successful.
This requirement for additional financing in the short term represents a material uncertainty that may cast significant doubt upon the Group’s ability to continue as a going concern. Should it become evident in the future that there are no realistic financing options available to the Group which are actionable before its cash resources run out then the Group will no longer be a going concern. In such circumstances, we would no longer be able to prepare financial statements under paragraph 25 of IAS 1. Instead, the financial statements would be prepared on a liquidation basis and assets would be stated at net realizable value and all liabilities would be accelerated to current liabilities. |
Accounting for eRapa and CPRIT grant |
6 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Accounting For Erapa And Cprit Grant | |||||||||||||||||||||||||||||||||||||||||||||||||
| Accounting for eRapa and CPRIT grant |
The License and Collaboration Agreement (“LCA”) entered into with Emtora in April 2024 meets the definition of a Joint Arrangement under IFRS 11, specifically related to the FAP program.
A jointly controlled escrow account was established on completion of the LCA. All FAP program transactions are processed through the escrow account, including the Company’s deposits of matching funds, as set out in the agreement, the receipt of grant funding from CPRIT and the payment of eligible R&D expenses. Although the CPRIT grant and R&D supplier contracts are with Emtora, the joint arrangement nature of the LCA results in Emtora being deemed to be acting as the Company’s agent. Accordingly, the Company recognises 100% of the grant and 100% of the R&D expenditure. The CPRIT grant recognised is on a 1 for 2 match. In accordance with the Company’s accounting policy, the grant, as it is the re-imbursement of directly related costs, is credited to R&D costs in the same period in The Statements of Comprehensive Income. The escrow account is recognised within prepayments, CPRIT grant received in advance is recognised within deferred revenue and any grant not yet received is recognised in accrued income.
In 1H25 the company recognised R&D costs of £0.5 million (1H24: £0.2 million) on the FAP project, made up of expenditure of £2.6 million (1H24: £0.5 million) netted against CPRIT grant of £2.1 million (1H24: £0.3 million).
The balances in relation to the FAP project as at June 30 were as follows:
* prepayment reflects only the escrow account balance |
||||||||||||||||||||||||||||||||||||||||||||||||
Finance income and expense |
6 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes and other explanatory information [abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Finance income and expense |
The gain on the equity settled derivative financial liability in 1H25 and 1H24 arose as a result of the fall in the Biodexa share price.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Loss per share |
6 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loss Per Share | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loss per share |
Basic loss per share amounts are calculated by dividing the net loss for the period from continuing operations, attributable to ordinary equity holders of the parent company, by the weighted average number of ordinary shares outstanding during the period. As the Group made a loss for the period the diluted loss per share is equal to the basic loss per share.
At a General Meeting on 11 June 2025, shareholders approved the subdivision and redesignation of the Company’s Issued Ordinary Shares of £0.00005 each into to one Ordinary Share of £0.000001 each and 49 ‘D’ Deferred Shares of £0.000001 each. The ‘D’ Deferred Shares have limited rights and are effectively valueless. The share sub-division and redesignation did not impact the calculation of the denominator as the number of Issued Ordinary Shares did not change.
The Company has considered the guidance set out in IAS 33 in calculating the denominator in connection with the issuance of Pre-Funded and Abeyance Shares. Management have recognised the warrants from the date of grant rather than the date of issue of the corresponding Ordinary Shares when calculating the denominator.
The Group has made a loss in the current and previous periods presented, and therefore the options and warrants are anti-dilutive. As a result, diluted earnings per share is presented on the same basis as basic earnings per share.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Share capital and reserves |
6 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share Capital And Reserves | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share capital and reserves |
Ordinary and deferred shares were recorded as equity.
At a General Meeting on 11 June 2025, shareholders approved the subdivision and redesignation of the Company’s Issued Ordinary Shares of £0.00005 each into to one Ordinary Share of £0.000001 each and 49 ‘D’ Deferred Shares of £0.000001 each. The ‘D’ Deferred Shares have limited rights and are effectively valueless.
As at June 30, 2025 and December 31, 2024 the Company had 17,415 pre-funded warrants outstanding over ADS’s. These are recognised in the warrant reserve until exercise.
In accordance with the Articles of Association for the Company adopted on 11 June 2025, the share capital of the Company consists of an unlimited number of ordinary shares of nominal value £ each. Ordinary and deferred shares were recorded as equity. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Related party transaction |
6 Months Ended | |||
|---|---|---|---|---|
Jun. 30, 2025 | ||||
| Notes and other explanatory information [abstract] | ||||
| Related party transaction |
The Directors consider there to be no related party transactions during the periods reported other than Directors Remuneration. |
Contingent liabilities |
6 Months Ended | |||
|---|---|---|---|---|
Jun. 30, 2025 | ||||
| Notes and other explanatory information [abstract] | ||||
| Contingent liabilities |
The Company is in a dispute with a former advisor over fees. In the event the Company is unsuccessful in its dispute the amount due to the vendor is approximately $1.16 million, of which $0.82 million would be payable in cash and the remainder amount in warrants exercisable for our ADSs. The Directors note that in the event of an unfavourable resolution the Company would not be able to recoup the loss from another party. |
Events after the reporting date |
6 Months Ended | |||
|---|---|---|---|---|
Jun. 30, 2025 | ||||
| Notes and other explanatory information [abstract] | ||||
| Events after the reporting date |
On July 15, 2025, the Company announced a ratio change on its ADSs from one (1) ADS representing ten thousand (10,000) ordinary shares, to the new ratio of one (1) ADS representing one hundred thousand (100,000) ordinary shares (the "Ratio Change"). The effective date of the Ratio Change was July 31, 2025. |
Accounting for eRapa and CPRIT grant (Tables) |
6 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | ||||||||||||||||||||||||||||||||||||||||||||||
| Accounting For Erapa And Cprit Grant | ||||||||||||||||||||||||||||||||||||||||||||||
| Schedule of relation the FAP |
|
|||||||||||||||||||||||||||||||||||||||||||||
Finance income and expense (Tables) |
6 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes and other explanatory information [abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Schedule of finance income |
The gain on the equity settled derivative financial liability in 1H25 and 1H24 arose as a result of the fall in the Biodexa share price.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Loss per share (Tables) |
6 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loss Per Share | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Schedule of loss per share |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Share capital and reserves (Tables) |
6 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Jun. 30, 2025 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share Capital And Reserves | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Schedule of detailed information about share capital |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Basis of preparation (Details Narrative) - GBP (£) £ in Thousands |
6 Months Ended | |
|---|---|---|
Jun. 30, 2025 |
Jun. 30, 2024 |
|
| Basis Of Preparation | ||
| Loss from operations | £ 3,810 | £ 3,310 |
| Negative cash flows from operating activities | 3,300 | £ 4,810 |
| Accumulated deficit | 154,130 | |
| Cash and cash equivalents | £ 4,040 | |
Accounting for eRapa and CPRIT grant (Details) - GBP (£) £ in Thousands |
Jun. 30, 2025 |
Jun. 30, 2024 |
|---|---|---|
| Accounting For Erapa And Cprit Grant | ||
| Prepayments* | £ 2,751 | £ 6,114 |
| Accrued revenue | 599 | |
| Deferred revenue | £ (1,468) |
Accounting for eRapa and CPRIT grant (Details Narrative) - GBP (£) £ in Thousands |
6 Months Ended | |
|---|---|---|
Jun. 30, 2025 |
Jun. 30, 2024 |
|
| Accounting For Erapa And Cprit Grant | ||
| Research and devlopment cost | £ 500 | £ 200 |
| Capital expenditure | 2,600 | 500 |
| Against CIPRIT grant value | £ 2,100 | £ 300 |
Finance income and expense (Details) - GBP (£) £ in Thousands |
6 Months Ended | |
|---|---|---|
Jun. 30, 2025 |
Jun. 30, 2024 |
|
| Finance income | ||
| Interest received on bank deposits | £ 32 | £ 86 |
| Other interest | 3 | 2 |
| Gain on equity settled derivative financial liability | 145 | 751 |
| Total finance income | 180 | 839 |
| Finance expense | ||
| Interest expense on lease liabilities | 6 | 11 |
| Interest expense on deferred consideration | 86 | 38 |
| Other loans | 43 | |
| Total finance expense | £ 135 | £ 49 |
Loss per share (Details) - GBP (£) £ / shares in Units, £ in Thousands |
6 Months Ended | |
|---|---|---|
Jun. 30, 2025 |
Jun. 30, 2024 |
|
| Numerator | ||
| Loss used in basic EPS and diluted EPS: | £ (3,806) | £ (3,308) |
| Denominator | ||
| Weighted average number of ordinary shares used in basic EPS | 25,267,266,823 | 3,280,798,115 |
| Basic loss per share | £ (0.0002) | £ (0.001) |
| Diluted loss per share | £ (0.0002) | £ (0.001) |
Loss per share (Details Narrative) |
Jun. 11, 2025 |
|---|---|
| Loss Per Share | |
| Description of stock split | Company’s Issued Ordinary Shares of £0.00005 each into to one Ordinary Share of £0.000001 each and 49 ‘D’ Deferred Shares of £0.000001 each. The ‘D’ Deferred Shares have limited rights and are effectively valueless. |
Share capital and reserves (Details Narrative) - £ / shares |
Jun. 11, 2025 |
Jun. 30, 2025 |
Dec. 31, 2024 |
|---|---|---|---|
| Share Capital And Reserves | |||
| Description of stock split | Company’s Issued Ordinary Shares of £0.00005 each into to one Ordinary Share of £0.000001 each and 49 ‘D’ Deferred Shares of £0.000001 each. The ‘D’ Deferred Shares have limited rights and are effectively valueless. | ||
| Warrants outstanding | 17,415 | 17,415 | |
| Ordinary shares par value | £ 0.000001 |
Contingent liabilities (Details Narrative) |
6 Months Ended |
|---|---|
Jun. 30, 2025 | |
| Notes and other explanatory information [abstract] | |
| Contingent liability description | In the event the Company is unsuccessful in its dispute the amount due to the vendor is approximately $1.16 million, of which $0.82 million would be payable in cash and the remainder amount in warrants exercisable for our ADSs. |
Events after the reporting date (Details Narrative) |
Jul. 15, 2025 |
|---|---|
| Notes and other explanatory information [abstract] | |
| Ratio change description | Company announced a ratio change on its ADSs from one (1) ADS representing ten thousand (10,000) ordinary shares, to the new ratio of one (1) ADS representing one hundred thousand (100,000) ordinary shares (the "Ratio Change"). The effective date of the Ratio Change was July 31, 2025. |
{
"version": "2.2",
"instance": {
"y982506k.htm ex99_1.htm": {
"nsprefix": "bdrx",
"nsuri": "http://biodexapharma.com/20250630",
"dts": {
"schema": {
"local": [
"bdrx-20250630.xsd"
],
"remote": [
"http://www.xbrl.org/2003/xbrl-instance-2003-12-31.xsd",
"http://www.xbrl.org/2003/xbrl-linkbase-2003-12-31.xsd",
"http://www.xbrl.org/2003/xl-2003-12-31.xsd",
"http://www.xbrl.org/2003/xlink-2003-12-31.xsd",
"http://www.xbrl.org/2005/xbrldt-2005.xsd",
"http://www.xbrl.org/2006/ref-2006-02-27.xsd",
"http://www.xbrl.org/lrr/role/negated-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/net-2009-12-16.xsd",
"http://www.xbrl.org/lrr/role/reference-2009-12-16.xsd",
"https://www.xbrl.org/2020/extensible-enumerations-2.0.xsd",
"https://www.xbrl.org/dtr/type/2020-01-21/types.xsd",
"https://www.xbrl.org/dtr/type/2022-03-31/types.xsd",
"https://xbrl.fasb.org/srt/2024/elts/srt-2024.xsd",
"https://xbrl.fasb.org/srt/2024/elts/srt-roles-2024.xsd",
"https://xbrl.fasb.org/srt/2024/elts/srt-types-2024.xsd",
"https://xbrl.ifrs.org/taxonomy/2024-03-27/full_ifrs/dimensions/rol_full_ifrs-dim_2024-03-27.xsd",
"https://xbrl.ifrs.org/taxonomy/2024-03-27/full_ifrs/full_ifrs-cor_2024-03-27.xsd",
"https://xbrl.org/2020/extensible-enumerations-2.0.xsd",
"https://xbrl.sec.gov/country/2024/country-2024.xsd",
"https://xbrl.sec.gov/dei/2024/dei-2024.xsd"
]
},
"calculationLink": {
"local": [
"bdrx-20250630_cal.xml"
]
},
"definitionLink": {
"local": [
"bdrx-20250630_def.xml"
],
"remote": [
"https://xbrl.ifrs.org/taxonomy/2024-03-27/full_ifrs/dimensions/dim_full_ifrs_2024-03-27_role-995000.xml"
]
},
"labelLink": {
"local": [
"bdrx-20250630_lab.xml"
]
},
"presentationLink": {
"local": [
"bdrx-20250630_pre.xml"
]
},
"inline": {
"local": [
"ex99_1.htm",
"y982506k.htm"
]
}
},
"keyStandard": 86,
"keyCustom": 37,
"axisStandard": 2,
"axisCustom": 0,
"memberStandard": 1,
"memberCustom": 10,
"hidden": {
"total": 81,
"http://xbrl.sec.gov/dei/2024": 5,
"https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full": 37,
"http://biodexapharma.com/20250630": 39
},
"contextCount": 52,
"entityCount": 1,
"segmentCount": 11,
"elementCount": 214,
"unitCount": 6,
"baseTaxonomies": {
"https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full": 237,
"http://xbrl.sec.gov/dei/2024": 13
},
"report": {
"R1": {
"role": "http://biodexapharma.com/role/Cover",
"longName": "00000001 - Document - Cover",
"shortName": "Cover",
"isDefault": "true",
"groupType": "document",
"subGroupType": "",
"menuCat": "Cover",
"order": "1",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "dei:DocumentType",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"b",
"p",
"body",
"html"
],
"reportCount": 1,
"baseRef": "y982506k.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "dei:DocumentType",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"b",
"p",
"body",
"html"
],
"reportCount": 1,
"baseRef": "y982506k.htm",
"first": true,
"unique": true
}
},
"R2": {
"role": "http://biodexapharma.com/role/StatementsOfComprehensiveIncome",
"longName": "00000002 - Statement - Consolidated Statements of Comprehensive Income (Unaudited)",
"shortName": "Consolidated Statements of Comprehensive Income (Unaudited)",
"isDefault": "false",
"groupType": "statement",
"subGroupType": "",
"menuCat": "Statements",
"order": "2",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:OtherIncomes",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:OtherIncomes",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R3": {
"role": "http://biodexapharma.com/role/StatementsOfFinancialPosition",
"longName": "00000003 - Statement - Consolidated Statements of Financial Position (Unaudited)",
"shortName": "Consolidated Statements of Financial Position (Unaudited)",
"isDefault": "false",
"groupType": "statement",
"subGroupType": "",
"menuCat": "Statements",
"order": "3",
"firstAnchor": {
"contextRef": "AsOf2025-06-30",
"name": "ifrs-full:PropertyPlantAndEquipment",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "AsOf2025-06-30",
"name": "ifrs-full:PropertyPlantAndEquipment",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R4": {
"role": "http://biodexapharma.com/role/StatementsOfCashFlows",
"longName": "00000004 - Statement - Consolidated Statements of Cash Flows (Unaudited)",
"shortName": "Consolidated Statements of Cash Flows (Unaudited)",
"isDefault": "false",
"groupType": "statement",
"subGroupType": "",
"menuCat": "Statements",
"order": "4",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:ProfitLoss",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:ProfitLoss",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R5": {
"role": "http://biodexapharma.com/role/StatementsOfChangesInEquity",
"longName": "00000005 - Statement - Consolidated Statements of Changes in Equity (Unaudited)",
"shortName": "Consolidated Statements of Changes in Equity (Unaudited)",
"isDefault": "false",
"groupType": "statement",
"subGroupType": "",
"menuCat": "Statements",
"order": "5",
"firstAnchor": {
"contextRef": "AsOf2023-12-31_custom_ShareCapitalMember",
"name": "ifrs-full:Equity",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "AsOf2023-12-31_custom_ShareCapitalMember",
"name": "ifrs-full:Equity",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R6": {
"role": "http://biodexapharma.com/role/BasisOfPreparation",
"longName": "999006 - Disclosure - Basis of preparation",
"shortName": "Basis of preparation",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "6",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfBasisOfPreparationExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfBasisOfPreparationExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R7": {
"role": "http://biodexapharma.com/role/AccountingForErapaAndCpritGrant",
"longName": "999007 - Disclosure - Accounting for eRapa and CPRIT grant",
"shortName": "Accounting for eRapa and CPRIT grant",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "7",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R8": {
"role": "http://biodexapharma.com/role/FinanceIncomeAndExpense",
"longName": "999008 - Disclosure - Finance income and expense",
"shortName": "Finance income and expense",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "8",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfFinanceIncomeExpenseExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfFinanceIncomeExpenseExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R9": {
"role": "http://biodexapharma.com/role/LossPerShare",
"longName": "999009 - Disclosure - Loss per share",
"shortName": "Loss per share",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "9",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfLossPerShareExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfLossPerShareExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R10": {
"role": "http://biodexapharma.com/role/ShareCapitalAndReserves",
"longName": "999010 - Disclosure - Share capital and reserves",
"shortName": "Share capital and reserves",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "10",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfShareCapitalReservesExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfShareCapitalReservesExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R11": {
"role": "http://biodexapharma.com/role/RelatedPartyTransaction",
"longName": "999011 - Disclosure - Related party transaction",
"shortName": "Related party transaction",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "11",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfRelatedPartyExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfRelatedPartyExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R12": {
"role": "http://biodexapharma.com/role/ContingentLiabilities",
"longName": "999012 - Disclosure - Contingent liabilities",
"shortName": "Contingent liabilities",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "12",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfContingentLiabilitiesExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfContingentLiabilitiesExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R13": {
"role": "http://biodexapharma.com/role/EventsAfterReportingDate",
"longName": "999013 - Disclosure - Events after the reporting date",
"shortName": "Events after the reporting date",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "",
"menuCat": "Notes",
"order": "13",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfEventsAfterReportingPeriodExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:DisclosureOfEventsAfterReportingPeriodExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R14": {
"role": "http://biodexapharma.com/role/AccountingForErapaAndCpritGrantTables",
"longName": "999014 - Disclosure - Accounting for eRapa and CPRIT grant (Tables)",
"shortName": "Accounting for eRapa and CPRIT grant (Tables)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "tables",
"menuCat": "Tables",
"order": "14",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:ScheduleOfAccountingForERapaAndCIPRITGrantTableTextBlock",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:ScheduleOfAccountingForERapaAndCIPRITGrantTableTextBlock",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R15": {
"role": "http://biodexapharma.com/role/FinanceIncomeAndExpenseTables",
"longName": "999015 - Disclosure - Finance income and expense (Tables)",
"shortName": "Finance income and expense (Tables)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "tables",
"menuCat": "Tables",
"order": "15",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfDetailedInformationAboutFinanceIncomeExpenseExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"ifrs-full:DisclosureOfFinanceIncomeExpenseExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfDetailedInformationAboutFinanceIncomeExpenseExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"ifrs-full:DisclosureOfFinanceIncomeExpenseExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R16": {
"role": "http://biodexapharma.com/role/LossPerShareTables",
"longName": "999016 - Disclosure - Loss per share (Tables)",
"shortName": "Loss per share (Tables)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "tables",
"menuCat": "Tables",
"order": "16",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfDetailedInformationAboutEarningsPerShareExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"bdrx:DisclosureOfLossPerShareExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfDetailedInformationAboutEarningsPerShareExplanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"bdrx:DisclosureOfLossPerShareExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R17": {
"role": "http://biodexapharma.com/role/ShareCapitalAndReservesTables",
"longName": "999017 - Disclosure - Share capital and reserves (Tables)",
"shortName": "Share capital and reserves (Tables)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "tables",
"menuCat": "Tables",
"order": "17",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfIssuedCapital1Explanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"bdrx:DisclosureOfShareCapitalReservesExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:DisclosureOfIssuedCapital1Explanatory",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"bdrx:DisclosureOfShareCapitalReservesExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R18": {
"role": "http://biodexapharma.com/role/BasisOfPreparationDetailsNarrative",
"longName": "999018 - Disclosure - Basis of preparation (Details Narrative)",
"shortName": "Basis of preparation (Details Narrative)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "18",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:LossFromOperations",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"span",
"p",
"bdrx:DisclosureOfBasisOfPreparationExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:LossFromOperations",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"span",
"p",
"bdrx:DisclosureOfBasisOfPreparationExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R19": {
"role": "http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetails",
"longName": "999019 - Disclosure - Accounting for eRapa and CPRIT grant (Details)",
"shortName": "Accounting for eRapa and CPRIT grant (Details)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "19",
"firstAnchor": {
"contextRef": "AsOf2025-06-30",
"name": "ifrs-full:Prepayments",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"bdrx:ScheduleOfAccountingForERapaAndCIPRITGrantTableTextBlock",
"bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "AsOf2025-06-30",
"name": "ifrs-full:Prepayments",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"bdrx:ScheduleOfAccountingForERapaAndCIPRITGrantTableTextBlock",
"bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R20": {
"role": "http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetailsNarrative",
"longName": "999020 - Disclosure - Accounting for eRapa and CPRIT grant (Details Narrative)",
"shortName": "Accounting for eRapa and CPRIT grant (Details Narrative)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "20",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:ResearchAndDevlopmentCost",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"span",
"p",
"bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:ResearchAndDevlopmentCost",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"span",
"p",
"bdrx:DisclosureAccountingForERapaAndCIPRITGrant",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R21": {
"role": "http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails",
"longName": "999021 - Disclosure - Finance income and expense (Details)",
"shortName": "Finance income and expense (Details)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "21",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:InterestIncomeOnDeposits",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"bdrx:DisclosureOfDetailedInformationAboutFinanceIncomeExpenseExplanatory",
"ifrs-full:DisclosureOfFinanceIncomeExpenseExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:InterestIncomeOnDeposits",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"bdrx:DisclosureOfDetailedInformationAboutFinanceIncomeExpenseExplanatory",
"ifrs-full:DisclosureOfFinanceIncomeExpenseExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R22": {
"role": "http://biodexapharma.com/role/LossPerShareDetails",
"longName": "999022 - Disclosure - Loss per share (Details)",
"shortName": "Loss per share (Details)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "22",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:ProfitLossFromContinuingOperations",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"bdrx:DisclosureOfDetailedInformationAboutEarningsPerShareExplanatory",
"bdrx:DisclosureOfLossPerShareExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "ifrs-full:ProfitLossFromContinuingOperations",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "-3",
"ancestors": [
"td",
"tr",
"table",
"bdrx:DisclosureOfDetailedInformationAboutEarningsPerShareExplanatory",
"bdrx:DisclosureOfLossPerShareExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R23": {
"role": "http://biodexapharma.com/role/LossPerShareDetailsNarrative",
"longName": "999023 - Disclosure - Loss per share (Details Narrative)",
"shortName": "Loss per share (Details Narrative)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "23",
"firstAnchor": {
"contextRef": "From2025-06-012025-06-11",
"name": "bdrx:DescriptionOStockholdersEquityNoteStockSplit",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"p",
"bdrx:DisclosureOfLossPerShareExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true
},
"uniqueAnchor": null
},
"R24": {
"role": "http://biodexapharma.com/role/ShareCapitalAndReservesDetails",
"longName": "999024 - Disclosure - Share capital and reserves (Details)",
"shortName": "Share capital and reserves (Details)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "24",
"firstAnchor": {
"contextRef": "AsOf2025-06-30",
"name": "bdrx:ValueOfShares",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "0",
"ancestors": [
"span",
"td",
"tr",
"table",
"bdrx:DisclosureOfIssuedCapital1Explanatory",
"bdrx:DisclosureOfShareCapitalReservesExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "AsOf2025-06-30",
"name": "bdrx:ValueOfShares",
"unitRef": "GBP",
"xsiNil": "false",
"lang": null,
"decimals": "0",
"ancestors": [
"span",
"td",
"tr",
"table",
"bdrx:DisclosureOfIssuedCapital1Explanatory",
"bdrx:DisclosureOfShareCapitalReservesExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R25": {
"role": "http://biodexapharma.com/role/ShareCapitalAndReservesDetailsNarrative",
"longName": "999025 - Disclosure - Share capital and reserves (Details Narrative)",
"shortName": "Share capital and reserves (Details Narrative)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "25",
"firstAnchor": {
"contextRef": "From2025-06-012025-06-11",
"name": "bdrx:DescriptionOStockholdersEquityNoteStockSplit",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"p",
"bdrx:DisclosureOfLossPerShareExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true
},
"uniqueAnchor": {
"contextRef": "AsOf2025-06-30",
"name": "bdrx:WarrantsOutstanding",
"unitRef": "Shares",
"xsiNil": "false",
"lang": null,
"decimals": "INF",
"ancestors": [
"span",
"span",
"p",
"bdrx:DisclosureOfShareCapitalReservesExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"unique": true
}
},
"R26": {
"role": "http://biodexapharma.com/role/ContingentLiabilitiesDetailsNarrative",
"longName": "999026 - Disclosure - Contingent liabilities (Details Narrative)",
"shortName": "Contingent liabilities (Details Narrative)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "26",
"firstAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:ContingentLiabilityDescription",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"p",
"ifrs-full:DisclosureOfContingentLiabilitiesExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-01-01to2025-06-30",
"name": "bdrx:ContingentLiabilityDescription",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"p",
"ifrs-full:DisclosureOfContingentLiabilitiesExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
},
"R27": {
"role": "http://biodexapharma.com/role/EventsAfterReportingDateDetailsNarrative",
"longName": "999027 - Disclosure - Events after the reporting date (Details Narrative)",
"shortName": "Events after the reporting date (Details Narrative)",
"isDefault": "false",
"groupType": "disclosure",
"subGroupType": "details",
"menuCat": "Details",
"order": "27",
"firstAnchor": {
"contextRef": "From2025-07-012025-07-15",
"name": "bdrx:RatioChangeDescription",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"p",
"ifrs-full:DisclosureOfEventsAfterReportingPeriodExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
},
"uniqueAnchor": {
"contextRef": "From2025-07-012025-07-15",
"name": "bdrx:RatioChangeDescription",
"unitRef": null,
"xsiNil": "false",
"lang": "en-US",
"decimals": null,
"ancestors": [
"span",
"p",
"ifrs-full:DisclosureOfEventsAfterReportingPeriodExplanatory",
"body",
"html"
],
"reportCount": 1,
"baseRef": "ex99_1.htm",
"first": true,
"unique": true
}
}
},
"tag": {
"bdrx_ADeferredSharesMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ADeferredSharesMember",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "A Deferred Shares [Member]"
}
}
},
"auth_ref": []
},
"bdrx_AccruedRevenue": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "AccruedRevenue",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Accrued revenue"
}
}
},
"auth_ref": []
},
"bdrx_AccumulatedDeficit": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "AccumulatedDeficit",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/BasisOfPreparationDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Accumulated deficit",
"label": "AccumulatedDeficit"
}
}
},
"auth_ref": []
},
"ifrs-full_AdjustmentsForAmortisationExpense": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForAmortisationExpense",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 4.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Amortisation of intangible fixed asset"
}
},
"en": {
"role": {
"documentation": "Adjustments for amortisation expense to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Profit (loss); Depreciation and amortisation expense]"
}
}
},
"auth_ref": [
"r119"
]
},
"ifrs-full_AdjustmentsForDecreaseIncreaseInTradeAndOtherReceivables": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForDecreaseIncreaseInTradeAndOtherReceivables",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInOperations",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Decrease/(Increase) in trade and other receivables"
}
},
"en": {
"role": {
"documentation": "Adjustments for decrease (increase) in trade and other receivables to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Trade and other receivables; Profit (loss)]"
}
}
},
"auth_ref": [
"r119"
]
},
"ifrs-full_AdjustmentsForDepreciationExpense": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForDepreciationExpense",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Depreciation of property, plant and equipment"
}
},
"en": {
"role": {
"documentation": "Adjustments for depreciation expense to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Profit (loss)]"
}
}
},
"auth_ref": [
"r119"
]
},
"ifrs-full_AdjustmentsForIncomeTaxExpense": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForIncomeTaxExpense",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 9.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Taxation",
"label": "Adjustments for income tax expense"
}
},
"en": {
"role": {
"documentation": "Adjustments for income tax expense to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Profit (loss)]"
}
}
},
"auth_ref": [
"r45"
]
},
"ifrs-full_AdjustmentsForIncreaseDecreaseInTradeAndOtherPayables": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForIncreaseDecreaseInTradeAndOtherPayables",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInOperations",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "(Decrease)/Increase in trade and other payables"
}
},
"en": {
"role": {
"documentation": "Adjustments for increase (decrease) in trade and other payables to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Trade and other payables; Profit (loss)]"
}
}
},
"auth_ref": [
"r119"
]
},
"ifrs-full_AdjustmentsForReconcileProfitLoss": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForReconcileProfitLoss",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInOperations",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Cash flows from operating activities before changes in working capital",
"label": "Adjustments to reconcile profit (loss)"
}
},
"en": {
"role": {
"documentation": "Adjustments to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Profit (loss)]"
}
}
},
"auth_ref": [
"r43"
]
},
"ifrs-full_AdjustmentsForReconcileProfitLossAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForReconcileProfitLossAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Adjustments for:"
}
}
},
"auth_ref": []
},
"ifrs-full_AdjustmentsForSharebasedPayments": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdjustmentsForSharebasedPayments",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 8.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Share-based payment expense"
}
},
"en": {
"role": {
"documentation": "Adjustments for share-based payments to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Profit (loss)]"
}
}
},
"auth_ref": [
"r118"
]
},
"ifrs-full_AdministrativeExpense": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AdministrativeExpense",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossFromOperatingActivities",
"weight": -1.0,
"order": 4.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Administrative costs",
"label": "Administrative expenses"
}
},
"en": {
"role": {
"documentation": "The amount of expenses that the entity classifies as being administrative."
}
}
},
"auth_ref": [
"r26",
"r34",
"r77"
]
},
"bdrx_AgainstCIPRITGrantValue": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "AgainstCIPRITGrantValue",
"crdr": "debit",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Against CIPRIT grant value"
}
}
},
"auth_ref": []
},
"dei_AmendmentDescription": {
"xbrltype": "stringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "AmendmentDescription",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Amendment Description",
"documentation": "Description of changes contained within amended document."
}
}
},
"auth_ref": []
},
"dei_AmendmentFlag": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "AmendmentFlag",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Amendment Flag",
"documentation": "Boolean flag that is true when the XBRL content amends previously-filed or accepted submission."
}
}
},
"auth_ref": []
},
"dei_AnnualInformationForm": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "AnnualInformationForm",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Annual Information Form",
"documentation": "Boolean flag with value true on a form if it is an annual report containing an annual information form."
}
}
},
"auth_ref": [
"r107"
]
},
"ifrs-full_Assets": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "Assets",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": null,
"weight": null,
"order": null,
"root": true
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total assets",
"label": "Assets [Default Label]"
}
},
"en": {
"role": {
"documentation": "The amount of a present economic resource controlled by the entity as a result of past events. Economic resource is a right that has the potential to produce economic benefits."
}
}
},
"auth_ref": [
"r11",
"r60",
"r61",
"r62",
"r68",
"r71"
]
},
"ifrs-full_AssetsAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "AssetsAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Assets"
}
}
},
"auth_ref": []
},
"dei_AuditedAnnualFinancialStatements": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "AuditedAnnualFinancialStatements",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Audited Annual Financial Statements",
"documentation": "Boolean flag with value true on a form if it is an annual report containing audited financial statements."
}
}
},
"auth_ref": [
"r107"
]
},
"bdrx_BDeferredSharesMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "BDeferredSharesMember",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "B Deferred Shares [Member]"
}
}
},
"auth_ref": []
},
"ifrs-full_BasicEarningsLossPerShare": {
"xbrltype": "perShareItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "BasicEarningsLossPerShare",
"presentation": [
"http://biodexapharma.com/role/LossPerShareDetails",
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Basic loss per share"
}
},
"en": {
"role": {
"documentation": "The amount of profit (loss) attributable to ordinary equity holders of the parent entity (the numerator) divided by the weighted average number of ordinary shares outstanding during the period (the denominator)."
}
}
},
"auth_ref": [
"r36",
"r37"
]
},
"ifrs-full_Borrowings": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "Borrowings",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_CurrentLiabilities",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Borrowings",
"label": "Borrowings [Default Label]"
}
},
"en": {
"role": {
"documentation": "The amount of outstanding funds that the entity is obligated to repay."
}
}
},
"auth_ref": [
"r115"
]
},
"bdrx_CDeferredSharesMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "CDeferredSharesMember",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "C Deferred Shares [Member]"
}
}
},
"auth_ref": []
},
"ifrs-full_CapitalCommitments": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CapitalCommitments",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Capital expenditure"
}
},
"en": {
"role": {
"documentation": "The amount of future capital expenditures that the entity is committed to make."
}
}
},
"auth_ref": [
"r114"
]
},
"ifrs-full_Cash": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "Cash",
"crdr": "debit",
"presentation": [
"http://biodexapharma.com/role/BasisOfPreparationDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Cash and cash equivalents",
"label": "Cash"
}
},
"en": {
"role": {
"documentation": "The amount of cash on hand and demand deposits. [Refer: Cash on hand]"
}
}
},
"auth_ref": [
"r120"
]
},
"ifrs-full_CashAndCashEquivalents": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashAndCashEquivalents",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_CurrentAssets",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows",
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Cash and cash equivalents",
"periodStartLabel": "Cash and cash equivalents at beginning of period",
"periodEndLabel": "Cash and cash equivalents at end of period"
}
},
"en": {
"role": {
"documentation": "The amount of cash on hand and demand deposits, along with short-term, highly liquid investments that are readily convertible to known amounts of cash and that are subject to an insignificant risk of changes in value. [Refer: Cash; Cash equivalents]"
}
}
},
"auth_ref": [
"r8",
"r46",
"r58"
]
},
"ifrs-full_CashFlowsFromUsedInFinancingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashFlowsFromUsedInFinancingActivities",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_IncreaseDecreaseInCashAndCashEquivalents",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Net cash generated from/(used in) financing activities",
"label": "Cash flows from (used in) financing activities"
}
},
"en": {
"role": {
"documentation": "The cash flows from (used in) financing activities, which are activities that result in changes in the size and composition of the contributed equity and borrowings of the entity, from continuing and discontinued operations."
}
}
},
"auth_ref": [
"r41",
"r47"
]
},
"ifrs-full_CashFlowsFromUsedInFinancingActivitiesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashFlowsFromUsedInFinancingActivitiesAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Financing activities"
}
}
},
"auth_ref": []
},
"ifrs-full_CashFlowsFromUsedInInvestingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashFlowsFromUsedInInvestingActivities",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_IncreaseDecreaseInCashAndCashEquivalents",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Net cash generated from/(used in) investing activities",
"label": "Cash flows from (used in) investing activities"
}
},
"en": {
"role": {
"documentation": "The cash flows from (used in) investing activities, which are the acquisition and disposal of long-term assets and other investments not included in cash equivalents, from continuing and discontinued operations."
}
}
},
"auth_ref": [
"r41",
"r47"
]
},
"ifrs-full_CashFlowsFromUsedInInvestingActivitiesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashFlowsFromUsedInInvestingActivitiesAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Investing activities"
}
}
},
"auth_ref": []
},
"ifrs-full_CashFlowsFromUsedInOperatingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashFlowsFromUsedInOperatingActivities",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_IncreaseDecreaseInCashAndCashEquivalents",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Net cash used in operating activities",
"label": "Cash flows from (used in) operating activities"
}
},
"en": {
"role": {
"documentation": "The cash flows from (used in) operating activities, which are the principal revenue-producing activities of the entity and other activities that are not investing or financing activities, from continuing and discontinued operations. [Refer: Revenue]"
}
}
},
"auth_ref": [
"r41",
"r47"
]
},
"ifrs-full_CashFlowsFromUsedInOperatingActivitiesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashFlowsFromUsedInOperatingActivitiesAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Cash flows from operating activities"
}
}
},
"auth_ref": []
},
"ifrs-full_CashFlowsFromUsedInOperations": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CashFlowsFromUsedInOperations",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInOperatingActivities",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Cash used in operations",
"label": "Cash flows from (used in) operations"
}
},
"en": {
"role": {
"documentation": "The cash from (used in) the entity's operations."
}
}
},
"auth_ref": [
"r87",
"r88"
]
},
"ifrs-full_ChangesInEquity": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ChangesInEquity",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Total contribution by and distributions to owners"
}
},
"en": {
"role": {
"documentation": "The increase (decrease) in equity. [Refer: Equity]"
}
}
},
"auth_ref": [
"r3"
]
},
"dei_CityAreaCode": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "CityAreaCode",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "City Area Code",
"documentation": "Area code of city"
}
}
},
"auth_ref": []
},
"ifrs-full_ClassesOfShareCapitalAxis": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ClassesOfShareCapitalAxis",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Classes of share capital [axis]"
}
},
"en": {
"role": {
"documentation": "The axis of a table defines the relationship between the members in the table and the line items or concepts that complete the table."
}
}
},
"auth_ref": [
"r18"
]
},
"ifrs-full_ClassesOfShareCapitalDomain": {
"xbrltype": "domainItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ClassesOfShareCapitalDomain",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en": {
"role": {
"documentation": "This member stands for share capital of the entity. It also represents the standard value for the 'Classes of share capital' axis if no other member is used."
}
}
},
"auth_ref": [
"r18"
]
},
"ifrs-full_ComponentsOfEquityAxis": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ComponentsOfEquityAxis",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Components of equity [axis]"
}
},
"en": {
"role": {
"documentation": "The axis of a table defines the relationship between the members in the table and the line items or concepts that complete the table."
}
}
},
"auth_ref": [
"r4"
]
},
"ifrs-full_ComponentsOfEquityDomain": {
"xbrltype": "domainItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ComponentsOfEquityDomain",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en": {
"role": {
"documentation": "This member stands for the residual interest in the assets of the entity after deducting all its liabilities. It also represents the standard value for the 'Components of equity' axis if no other member is used."
}
}
},
"auth_ref": [
"r4"
]
},
"ifrs-full_ComprehensiveIncomeAttributableToOwnersOfParent": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ComprehensiveIncomeAttributableToOwnersOfParent",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": null,
"weight": null,
"order": null,
"root": true
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity",
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total comprehensive loss attributable to the owners of the parent",
"label": "Total comprehensive loss"
}
},
"en": {
"role": {
"documentation": "The amount of comprehensive income attributable to owners of the parent. [Refer: Comprehensive income]"
}
}
},
"auth_ref": [
"r0",
"r21"
]
},
"bdrx_ContingentLiabilityDescription": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ContingentLiabilityDescription",
"presentation": [
"http://biodexapharma.com/role/ContingentLiabilitiesDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Contingent liability description"
}
}
},
"auth_ref": []
},
"bdrx_CostsAssociatedWithElocAgreement": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "CostsAssociatedWithElocAgreement",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Costs associated with ELOC agreement"
}
}
},
"auth_ref": []
},
"bdrx_CostsAssociatedWithShareIssue": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "CostsAssociatedWithShareIssue",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Costs associated with share issue\u00a0\u00a0on\u00a0\u00a015 May 2024"
}
}
},
"auth_ref": []
},
"bdrx_CostsAssociatedWithShareIssue1": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "CostsAssociatedWithShareIssue1",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Costs associated with share issue on February 15, 2023"
}
}
},
"auth_ref": []
},
"bdrx_CostsAssociatedWithShareIssue2": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "CostsAssociatedWithShareIssue2",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Costs associated with share issue on May 26, 2023"
}
}
},
"auth_ref": []
},
"dei_CountryRegion": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "CountryRegion",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Country Region",
"documentation": "Region code of country"
}
}
},
"auth_ref": []
},
"dei_CoverAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "CoverAbstract",
"lang": {
"en-us": {
"role": {
"label": "Cover [Abstract]",
"documentation": "Cover page."
}
}
},
"auth_ref": []
},
"ifrs-full_CurrentAssets": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CurrentAssets",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Assets",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total Current Assets",
"label": "Current assets [Default Label]"
}
},
"en": {
"role": {
"documentation": "The amount of assets that the entity (a) expects to realise or intends to sell or consume in its normal operating cycle; (b) holds primarily for the purpose of trading; (c) expects to realise within twelve months after the reporting period; or (d) classifies as cash or cash equivalents (as defined in IAS 7) unless the asset is restricted from being exchanged or used to settle a liability for at least twelve months after the reporting period. [Refer: Assets]"
}
}
},
"auth_ref": [
"r12",
"r52",
"r89"
]
},
"ifrs-full_CurrentAssetsAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CurrentAssetsAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Current assets"
}
}
},
"auth_ref": []
},
"ifrs-full_CurrentDerivativeFinancialLiabilities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CurrentDerivativeFinancialLiabilities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_CurrentLiabilities",
"weight": 1.0,
"order": 4.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Derivative financial liability"
}
},
"en": {
"role": {
"documentation": "The amount of current derivative financial liabilities. [Refer: Derivative financial liabilities]"
}
}
},
"auth_ref": [
"r115"
]
},
"dei_CurrentFiscalYearEndDate": {
"xbrltype": "gMonthDayItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "CurrentFiscalYearEndDate",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Current Fiscal Year End Date",
"documentation": "End date of current fiscal year in the format --MM-DD."
}
}
},
"auth_ref": []
},
"ifrs-full_CurrentLiabilities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CurrentLiabilities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Liabilities",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total current Liabilities",
"label": "Current liabilities [Default Label]"
}
},
"en": {
"role": {
"documentation": "The amount of liabilities that: (a) the entity expects to settle in its normal operating cycle; (b) the entity holds primarily for the purpose of trading; (c) are due to be settled within twelve months after the reporting period; or (d) the entity does not have the right at the end of the reporting period to defer settlement for at least twelve months after the reporting period."
}
}
},
"auth_ref": [
"r13",
"r54",
"r89"
]
},
"ifrs-full_CurrentLiabilitiesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CurrentLiabilitiesAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Current liabilities"
}
}
},
"auth_ref": []
},
"ifrs-full_CurrentTaxAssetsCurrent": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "CurrentTaxAssetsCurrent",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_CurrentAssets",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Taxation"
}
},
"en": {
"role": {
"documentation": "The current amount of current tax assets. [Refer: Current tax assets]"
}
}
},
"auth_ref": [
"r10"
]
},
"bdrx_DDeferredSharesMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DDeferredSharesMember",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "D Deferred Shares [Member]"
}
}
},
"auth_ref": []
},
"bdrx_DeferredConsiderationCurrent": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DeferredConsiderationCurrent",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_CurrentLiabilities",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Deferred consideration",
"label": "DeferredConsiderationCurrent"
}
}
},
"auth_ref": []
},
"bdrx_DeferredConsiderationNonCurrent": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DeferredConsiderationNonCurrent",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_NoncurrentLiabilities",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Deferred consideration"
}
}
},
"auth_ref": []
},
"bdrx_DeferredRevenues": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DeferredRevenues",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Deferred revenue"
}
}
},
"auth_ref": []
},
"ifrs-full_DepreciationRightofuseAssets": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "DepreciationRightofuseAssets",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Depreciation of right of use asset"
}
},
"en": {
"role": {
"documentation": "The amount of depreciation of right-of-use assets. [Refer: Depreciation and amortisation expense; Right-of-use assets]"
}
}
},
"auth_ref": [
"r63"
]
},
"bdrx_DescriptionOStockholdersEquityNoteStockSplit": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DescriptionOStockholdersEquityNoteStockSplit",
"presentation": [
"http://biodexapharma.com/role/LossPerShareDetailsNarrative",
"http://biodexapharma.com/role/ShareCapitalAndReservesDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Description of stock split"
}
}
},
"auth_ref": []
},
"ifrs-full_DilutedEarningsLossPerShare": {
"xbrltype": "perShareItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "DilutedEarningsLossPerShare",
"presentation": [
"http://biodexapharma.com/role/LossPerShareDetails",
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Diluted loss per share"
}
},
"en": {
"role": {
"documentation": "The amount of profit (loss) attributable to ordinary equity holders of the parent entity (the numerator), divided by the weighted average number of ordinary shares outstanding during the period (the denominator), both adjusted for the effects of all dilutive potential ordinary shares. [Refer: Ordinary shares [member]; Weighted average [member]]"
}
}
},
"auth_ref": [
"r36",
"r37"
]
},
"bdrx_DisclosureAccountingForERapaAndCIPRITGrant": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureAccountingForERapaAndCIPRITGrant",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrant"
],
"lang": {
"en-us": {
"role": {
"label": "Accounting for eRapa and CPRIT grant"
}
}
},
"auth_ref": []
},
"bdrx_DisclosureAccountingForErapaAndCpritGrantAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureAccountingForErapaAndCpritGrantAbstract",
"lang": {
"en-us": {
"role": {
"label": "Accounting For Erapa And Cprit Grant"
}
}
},
"auth_ref": []
},
"bdrx_DisclosureBasisOfPreparationAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureBasisOfPreparationAbstract",
"lang": {
"en-us": {
"role": {
"label": "Basis Of Preparation"
}
}
},
"auth_ref": []
},
"bdrx_DisclosureLossPerShareAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureLossPerShareAbstract",
"lang": {
"en-us": {
"role": {
"label": "Loss Per Share"
}
}
},
"auth_ref": []
},
"bdrx_DisclosureOfBasisOfPreparationExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureOfBasisOfPreparationExplanatory",
"presentation": [
"http://biodexapharma.com/role/BasisOfPreparation"
],
"lang": {
"en-us": {
"role": {
"label": "Basis of preparation"
}
}
},
"auth_ref": []
},
"ifrs-full_DisclosureOfContingentLiabilitiesExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "DisclosureOfContingentLiabilitiesExplanatory",
"presentation": [
"http://biodexapharma.com/role/ContingentLiabilities"
],
"lang": {
"en-us": {
"role": {
"label": "Contingent liabilities"
}
},
"en": {
"role": {
"documentation": "The disclosure of contingent liabilities. [Refer: Classes of contingent liabilities [domain]]"
}
}
},
"auth_ref": [
"r39"
]
},
"bdrx_DisclosureOfDetailedInformationAboutEarningsPerShareExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureOfDetailedInformationAboutEarningsPerShareExplanatory",
"presentation": [
"http://biodexapharma.com/role/LossPerShareTables"
],
"lang": {
"en-us": {
"role": {
"label": "Schedule of loss per share"
}
}
},
"auth_ref": []
},
"bdrx_DisclosureOfDetailedInformationAboutFinanceIncomeExpenseExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureOfDetailedInformationAboutFinanceIncomeExpenseExplanatory",
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseTables"
],
"lang": {
"en-us": {
"role": {
"label": "Schedule of finance income"
}
}
},
"auth_ref": []
},
"ifrs-full_DisclosureOfEventsAfterReportingPeriodExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "DisclosureOfEventsAfterReportingPeriodExplanatory",
"presentation": [
"http://biodexapharma.com/role/EventsAfterReportingDate"
],
"lang": {
"en-us": {
"role": {
"label": "Events after the reporting date"
}
},
"en": {
"role": {
"documentation": "The entire disclosure for events after the reporting period."
}
}
},
"auth_ref": [
"r27"
]
},
"ifrs-full_DisclosureOfFinanceIncomeExpenseExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "DisclosureOfFinanceIncomeExpenseExplanatory",
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpense"
],
"lang": {
"en-us": {
"role": {
"label": "Finance income and expense"
}
},
"en": {
"role": {
"documentation": "The disclosure of finance income (cost). [Refer: Finance income (cost)]"
}
}
},
"auth_ref": [
"r113"
]
},
"bdrx_DisclosureOfIssuedCapital1Explanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureOfIssuedCapital1Explanatory",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesTables"
],
"lang": {
"en-us": {
"role": {
"label": "Schedule of detailed information about share capital"
}
}
},
"auth_ref": []
},
"bdrx_DisclosureOfLossPerShareExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureOfLossPerShareExplanatory",
"presentation": [
"http://biodexapharma.com/role/LossPerShare"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Loss per share",
"label": "DisclosureOfLossPerShareExplanatory"
}
}
},
"auth_ref": []
},
"ifrs-full_DisclosureOfRelatedPartyExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "DisclosureOfRelatedPartyExplanatory",
"presentation": [
"http://biodexapharma.com/role/RelatedPartyTransaction"
],
"lang": {
"en-us": {
"role": {
"label": "Related party transaction"
}
},
"en": {
"role": {
"documentation": "The entire disclosure for related parties."
}
}
},
"auth_ref": [
"r33"
]
},
"bdrx_DisclosureOfShareCapitalReservesExplanatory": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureOfShareCapitalReservesExplanatory",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReserves"
],
"lang": {
"en-us": {
"role": {
"label": "Share capital and reserves"
}
}
},
"auth_ref": []
},
"bdrx_DisclosureShareCapitalAndReservesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "DisclosureShareCapitalAndReservesAbstract",
"lang": {
"en-us": {
"role": {
"label": "Share Capital And Reserves"
}
}
},
"auth_ref": []
},
"dei_DocumentAccountingStandard": {
"xbrltype": "accountingStandardItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentAccountingStandard",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Accounting Standard",
"documentation": "The basis of accounting the registrant has used to prepare the financial statements included in this filing This can either be 'U.S. GAAP', 'International Financial Reporting Standards', or 'Other'."
}
}
},
"auth_ref": [
"r106"
]
},
"dei_DocumentAnnualReport": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentAnnualReport",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Annual Report",
"documentation": "Boolean flag that is true only for a form used as an annual report."
}
}
},
"auth_ref": [
"r104",
"r106",
"r107"
]
},
"dei_DocumentFiscalPeriodFocus": {
"xbrltype": "fiscalPeriodItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentFiscalPeriodFocus",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Fiscal Period Focus",
"documentation": "Fiscal period values are FY, Q1, Q2, and Q3. 1st, 2nd and 3rd quarter 10-Q or 10-QT statements have value Q1, Q2, and Q3 respectively, with 10-K, 10-KT or other fiscal year statements having FY."
}
}
},
"auth_ref": []
},
"dei_DocumentFiscalYearFocus": {
"xbrltype": "gYearItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentFiscalYearFocus",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Fiscal Year Focus",
"documentation": "This is focus fiscal year of the document report in YYYY format. For a 2006 annual report, which may also provide financial information from prior periods, fiscal 2006 should be given as the fiscal year focus. Example: 2006."
}
}
},
"auth_ref": []
},
"dei_DocumentPeriodEndDate": {
"xbrltype": "dateItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentPeriodEndDate",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Period End Date",
"documentation": "For the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD."
}
}
},
"auth_ref": []
},
"dei_DocumentPeriodStartDate": {
"xbrltype": "dateItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentPeriodStartDate",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Period Start Date",
"documentation": "The start date of the period covered in the document, in YYYY-MM-DD format."
}
}
},
"auth_ref": []
},
"dei_DocumentQuarterlyReport": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentQuarterlyReport",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Quarterly Report",
"documentation": "Boolean flag that is true only for a form used as an quarterly report."
}
}
},
"auth_ref": [
"r105"
]
},
"dei_DocumentRegistrationStatement": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentRegistrationStatement",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Registration Statement",
"documentation": "Boolean flag that is true only for a form used as a registration statement."
}
}
},
"auth_ref": [
"r93"
]
},
"dei_DocumentShellCompanyEventDate": {
"xbrltype": "dateItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentShellCompanyEventDate",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Shell Company Event Date",
"documentation": "Date of event requiring a shell company report."
}
}
},
"auth_ref": [
"r106"
]
},
"dei_DocumentShellCompanyReport": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentShellCompanyReport",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Shell Company Report",
"documentation": "Boolean flag that is true for a Shell Company Report pursuant to section 13 or 15(d) of the Exchange Act."
}
}
},
"auth_ref": [
"r106"
]
},
"dei_DocumentTransitionReport": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentTransitionReport",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Transition Report",
"documentation": "Boolean flag that is true only for a form used as a transition report."
}
}
},
"auth_ref": [
"r108"
]
},
"dei_DocumentType": {
"xbrltype": "submissionTypeItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentType",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Document Type",
"documentation": "The type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'."
}
}
},
"auth_ref": []
},
"dei_DocumentsIncorporatedByReferenceTextBlock": {
"xbrltype": "textBlockItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "DocumentsIncorporatedByReferenceTextBlock",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Documents Incorporated by Reference [Text Block]",
"documentation": "Documents incorporated by reference."
}
}
},
"auth_ref": [
"r96"
]
},
"ifrs-full_EarningsPerShareAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "EarningsPerShareAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Loss per share"
}
}
},
"auth_ref": []
},
"dei_EntityAddressAddressLine1": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityAddressAddressLine1",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Address, Address Line One",
"documentation": "Address Line 1 such as Attn, Building Name, Street Name"
}
}
},
"auth_ref": []
},
"dei_EntityAddressAddressLine2": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityAddressAddressLine2",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Address, Address Line Two",
"documentation": "Address Line 2 such as Street or Suite number"
}
}
},
"auth_ref": []
},
"dei_EntityAddressAddressLine3": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityAddressAddressLine3",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Address, Address Line Three",
"documentation": "Address Line 3 such as an Office Park"
}
}
},
"auth_ref": []
},
"dei_EntityAddressCityOrTown": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityAddressCityOrTown",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Address, City or Town",
"documentation": "Name of the City or Town"
}
}
},
"auth_ref": []
},
"dei_EntityAddressCountry": {
"xbrltype": "countryCodeItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityAddressCountry",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Address, Country",
"documentation": "ISO 3166-1 alpha-2 country code."
}
}
},
"auth_ref": []
},
"dei_EntityAddressPostalZipCode": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityAddressPostalZipCode",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Address, Postal Zip Code",
"documentation": "Code for the postal or zip code"
}
}
},
"auth_ref": []
},
"dei_EntityAddressStateOrProvince": {
"xbrltype": "stateOrProvinceItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityAddressStateOrProvince",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Address, State or Province",
"documentation": "Name of the state or province."
}
}
},
"auth_ref": []
},
"dei_EntityBankruptcyProceedingsReportingCurrent": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityBankruptcyProceedingsReportingCurrent",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Bankruptcy Proceedings, Reporting Current",
"documentation": "For registrants involved in bankruptcy proceedings during the preceding five years, the value Yes indicates that the registrant has filed all documents and reports required to be filed by Section 12, 13 or 15(d) of the Securities Exchange Act of 1934 subsequent to the distribution of securities under a plan confirmed by a court; the value No indicates the registrant has not. Registrants not involved in bankruptcy proceedings during the preceding five years should not report this element."
}
}
},
"auth_ref": [
"r99"
]
},
"dei_EntityCentralIndexKey": {
"xbrltype": "centralIndexKeyItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityCentralIndexKey",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Central Index Key",
"documentation": "A unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK."
}
}
},
"auth_ref": [
"r95"
]
},
"dei_EntityCommonStockSharesOutstanding": {
"xbrltype": "sharesItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityCommonStockSharesOutstanding",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Common Stock, Shares Outstanding",
"documentation": "Indicate number of shares or other units outstanding of each of registrant's classes of capital or common stock or other ownership interests, if and as stated on cover of related periodic report. Where multiple classes or units exist define each class/interest by adding class of stock items such as Common Class A [Member], Common Class B [Member] or Partnership Interest [Member] onto the Instrument [Domain] of the Entity Listings, Instrument."
}
}
},
"auth_ref": []
},
"dei_EntityCurrentReportingStatus": {
"xbrltype": "yesNoItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityCurrentReportingStatus",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Current Reporting Status",
"documentation": "Indicate 'Yes' or 'No' whether registrants (1) have filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that registrants were required to file such reports), and (2) have been subject to such filing requirements for the past 90 days. This information should be based on the registrant's current or most recent filing containing the related disclosure."
}
}
},
"auth_ref": []
},
"dei_EntityEmergingGrowthCompany": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityEmergingGrowthCompany",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Emerging Growth Company",
"documentation": "Indicate if registrant meets the emerging growth company criteria."
}
}
},
"auth_ref": [
"r95"
]
},
"dei_EntityExTransitionPeriod": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityExTransitionPeriod",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Elected Not To Use the Extended Transition Period",
"documentation": "Indicate if an emerging growth company has elected not to use the extended transition period for complying with any new or revised financial accounting standards."
}
}
},
"auth_ref": [
"r112"
]
},
"dei_EntityFileNumber": {
"xbrltype": "fileNumberItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityFileNumber",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity File Number",
"documentation": "Commission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen."
}
}
},
"auth_ref": []
},
"dei_EntityFilerCategory": {
"xbrltype": "filerCategoryItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityFilerCategory",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Filer Category",
"documentation": "Indicate whether the registrant is one of the following: Large Accelerated Filer, Accelerated Filer, Non-accelerated Filer. Definitions of these categories are stated in Rule 12b-2 of the Exchange Act. This information should be based on the registrant's current or most recent filing containing the related disclosure."
}
}
},
"auth_ref": [
"r95"
]
},
"dei_EntityIncorporationStateCountryCode": {
"xbrltype": "edgarStateCountryItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityIncorporationStateCountryCode",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Incorporation, State or Country Code",
"documentation": "Two-character EDGAR code representing the state or country of incorporation."
}
}
},
"auth_ref": []
},
"dei_EntityInteractiveDataCurrent": {
"xbrltype": "yesNoItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityInteractiveDataCurrent",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Interactive Data Current",
"documentation": "Boolean flag that is true when the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files)."
}
}
},
"auth_ref": [
"r109"
]
},
"dei_EntityPrimarySicNumber": {
"xbrltype": "sicNumberItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityPrimarySicNumber",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Primary SIC Number",
"documentation": "Primary Standard Industrial Classification (SIC) Number for the Entity."
}
}
},
"auth_ref": [
"r107"
]
},
"dei_EntityPublicFloat": {
"xbrltype": "monetaryItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityPublicFloat",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Public Float",
"documentation": "The aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and asked price of such common equity, as of the last business day of the registrant's most recently completed second fiscal quarter."
}
}
},
"auth_ref": []
},
"dei_EntityRegistrantName": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityRegistrantName",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Registrant Name",
"documentation": "The exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC."
}
}
},
"auth_ref": [
"r95"
]
},
"dei_EntityShellCompany": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityShellCompany",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Shell Company",
"documentation": "Boolean flag that is true when the registrant is a shell company as defined in Rule 12b-2 of the Exchange Act."
}
}
},
"auth_ref": [
"r95"
]
},
"dei_EntitySmallBusiness": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntitySmallBusiness",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Small Business",
"documentation": "Indicates that the company is a Smaller Reporting Company (SRC)."
}
}
},
"auth_ref": [
"r95"
]
},
"dei_EntityTaxIdentificationNumber": {
"xbrltype": "employerIdItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityTaxIdentificationNumber",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Tax Identification Number",
"documentation": "The Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS."
}
}
},
"auth_ref": [
"r95"
]
},
"dei_EntityVoluntaryFilers": {
"xbrltype": "yesNoItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityVoluntaryFilers",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Voluntary Filers",
"documentation": "Indicate 'Yes' or 'No' if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act."
}
}
},
"auth_ref": []
},
"dei_EntityWellKnownSeasonedIssuer": {
"xbrltype": "yesNoItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "EntityWellKnownSeasonedIssuer",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Entity Well-known Seasoned Issuer",
"documentation": "Indicate 'Yes' or 'No' if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Is used on Form Type: 10-K, 10-Q, 8-K, 20-F, 6-K, 10-K/A, 10-Q/A, 20-F/A, 6-K/A, N-CSR, N-Q, N-1A."
}
}
},
"auth_ref": [
"r110"
]
},
"ifrs-full_Equity": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "Equity",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_EquityAndLiabilities",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity",
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total equity",
"periodStartLabel": "At January 1, 2024",
"periodEndLabel": "At June 30, 2024",
"label": "Equity"
}
},
"en": {
"role": {
"documentation": "The amount of residual interest in the assets of the entity after deducting all its liabilities."
}
}
},
"auth_ref": [
"r11",
"r15",
"r48",
"r50",
"r60",
"r61",
"r62"
]
},
"ifrs-full_EquityAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "EquityAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Issued capital and reserves attributable to owners of the parent"
}
}
},
"auth_ref": []
},
"ifrs-full_EquityAndLiabilities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "EquityAndLiabilities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": null,
"weight": null,
"order": null,
"root": true
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total equity and liabilities",
"label": "Equity and liabilities"
}
},
"en": {
"role": {
"documentation": "The amount of the entity's equity and liabilities. [Refer: Equity; Liabilities]"
}
}
},
"auth_ref": [
"r11"
]
},
"dei_Extension": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "Extension",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Extension",
"documentation": "Extension number for local phone number."
}
}
},
"auth_ref": []
},
"ifrs-full_FinanceCosts": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "FinanceCosts",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossBeforeTax",
"weight": -1.0,
"order": 3.0
},
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 7.0
},
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": null,
"weight": null,
"order": null,
"root": true
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails",
"http://biodexapharma.com/role/StatementsOfCashFlows",
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Finance expense",
"label": "Finance expense",
"totalLabel": "Total finance expense"
}
},
"en": {
"role": {
"documentation": "The amount of costs associated with financing activities of the entity."
}
}
},
"auth_ref": [
"r23"
]
},
"ifrs-full_FinanceIncome": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "FinanceIncome",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossBeforeTax",
"weight": 1.0,
"order": 2.0
},
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": -1.0,
"order": 6.0
},
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": null,
"weight": null,
"order": null,
"root": true
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails",
"http://biodexapharma.com/role/StatementsOfCashFlows",
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Finance income",
"negatedLabel": "Finance income",
"totalLabel": "Total finance income"
}
},
"en": {
"role": {
"documentation": "The amount of income associated with interest and other financing activities of the entity."
}
}
},
"auth_ref": [
"r117"
]
},
"bdrx_FinanceIncomeAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "FinanceIncomeAbstract",
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Finance income"
}
}
},
"auth_ref": []
},
"ifrs-full_ForeignExchangeLoss": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ForeignExchangeLoss",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 10.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Foreign exchange losses"
}
},
"en": {
"role": {
"documentation": "The gross loss arising from exchange differences recognised in profit or loss, excluding those arising on financial instruments measured at fair value through profit or loss in accordance with IFRS 9. [Refer: Foreign exchange gain (loss)]"
}
}
},
"auth_ref": [
"r5",
"r32"
]
},
"bdrx_ForeignExchangeReserve": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ForeignExchangeReserve",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Equity",
"weight": 1.0,
"order": 5.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Foreign exchange reserve"
}
}
},
"auth_ref": []
},
"bdrx_ForeignExchangeReserveMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ForeignExchangeReserveMember",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Foreign Exchange Reserve [Member]"
}
}
},
"auth_ref": []
},
"bdrx_ForeignExchangeTranslation": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ForeignExchangeTranslation",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Foreign exchange translation"
}
}
},
"auth_ref": []
},
"ifrs-full_GainsLossesOnExchangeDifferencesOnTranslationNetOfTax": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "GainsLossesOnExchangeDifferencesOnTranslationNetOfTax",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ComprehensiveIncomeAttributableToOwnersOfParent",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Exchange gain arising on translation of foreign operations"
}
},
"en": {
"role": {
"documentation": "The gains (losses) recognised in other comprehensive income on exchange differences on the translation of financial statements of foreign operations, net of tax, before reclassification adjustments. [Refer: Other comprehensive income]"
}
}
},
"auth_ref": [
"r25"
]
},
"ifrs-full_GainsOnChangeInFairValueOfDerivatives": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "GainsOnChangeInFairValueOfDerivatives",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": "ifrs-full_FinanceIncome",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Gain on equity settled derivative financial liability"
}
},
"en": {
"role": {
"documentation": "The gains resulting from change in the fair value of derivatives recognised in profit or loss. [Refer: Derivatives [member]]"
}
}
},
"auth_ref": [
"r117"
]
},
"bdrx_IfrsStatementLineItems": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "IfrsStatementLineItems",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails",
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "IfrsStatementLineItems [Line Items]"
}
}
},
"auth_ref": []
},
"bdrx_ImpairmentOfCommissionPaidInAdvanceOnEloc": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ImpairmentOfCommissionPaidInAdvanceOnEloc",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": 1.0,
"order": 5.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Impairment of commission paid in advance on ELOC"
}
}
},
"auth_ref": []
},
"ifrs-full_IncomeStatementAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "IncomeStatementAbstract",
"lang": {
"en-us": {
"role": {
"label": "Profit or loss [abstract]"
}
}
},
"auth_ref": []
},
"ifrs-full_IncomeTaxExpenseContinuingOperations": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "IncomeTaxExpenseContinuingOperations",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossAttributableToOwnersOfParent",
"weight": -1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Taxation",
"label": "Tax expense (income)"
}
},
"en": {
"role": {
"documentation": "The aggregate amount included in the determination of profit (loss) for the period in respect of current tax and deferred tax. [Refer: Current tax expense (income); Deferred tax expense (income)]"
}
}
},
"auth_ref": [
"r24",
"r28",
"r29",
"r30",
"r35",
"r59",
"r67"
]
},
"ifrs-full_IncomeTaxesPaidRefundClassifiedAsOperatingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "IncomeTaxesPaidRefundClassifiedAsOperatingActivities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInOperatingActivities",
"weight": -1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Taxes receipts",
"label": "Income taxes paid (refund), classified as operating activities"
}
},
"en": {
"role": {
"documentation": "The cash flows from income taxes paid or refunded, classified as operating activities. [Refer: Income taxes paid (refund)]"
}
}
},
"auth_ref": [
"r45",
"r83"
]
},
"ifrs-full_IncreaseDecreaseInCashAndCashEquivalents": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "IncreaseDecreaseInCashAndCashEquivalents",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": null,
"weight": null,
"order": null,
"root": true
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Net increase/(decrease) in cash and cash equivalents",
"label": "Increase (decrease) in cash and cash equivalents after effect of exchange rate changes"
}
},
"en": {
"role": {
"documentation": "The increase (decrease) in cash and cash equivalents after the effect of exchange rate changes on cash and cash equivalents held in foreign currencies. [Refer: Cash and cash equivalents; Effect of exchange rate changes on cash and cash equivalents]"
}
}
},
"auth_ref": [
"r46"
]
},
"ifrs-full_IncreaseDecreaseThroughSharebasedPaymentTransactions": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "IncreaseDecreaseThroughSharebasedPaymentTransactions",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Share-based payment charge"
}
},
"en": {
"role": {
"documentation": "The increase (decrease) in equity resulting from share-based payment transactions. [Refer: Equity]"
}
}
},
"auth_ref": [
"r2"
]
},
"ifrs-full_IntangibleAssetsAndGoodwill": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "IntangibleAssetsAndGoodwill",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_NoncurrentAssets",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Intangible assets"
}
},
"en": {
"role": {
"documentation": "The amount of intangible assets and goodwill held by the entity. [Refer: Goodwill; Intangible assets other than goodwill]"
}
}
},
"auth_ref": [
"r115"
]
},
"ifrs-full_InterestCostsAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "InterestCostsAbstract",
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"verboseLabel": "Finance expense"
}
}
},
"auth_ref": []
},
"bdrx_InterestExpenseOnDeferredConsideration": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "InterestExpenseOnDeferredConsideration",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": "ifrs-full_FinanceCosts",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Interest expense on deferred consideration"
}
}
},
"auth_ref": []
},
"ifrs-full_InterestExpenseOnLeaseLiabilities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "InterestExpenseOnLeaseLiabilities",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": "ifrs-full_FinanceCosts",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Interest expense on lease liabilities"
}
},
"en": {
"role": {
"documentation": "The amount of interest expense on lease liabilities. [Refer: Lease liabilities]"
}
}
},
"auth_ref": [
"r64"
]
},
"ifrs-full_InterestExpenseOnOtherFinancialLiabilities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "InterestExpenseOnOtherFinancialLiabilities",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": "ifrs-full_FinanceCosts",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Other loans"
}
},
"en": {
"role": {
"documentation": "The amount of interest expense on other financial liabilities. [Refer: Interest expense; Other financial liabilities]"
}
}
},
"auth_ref": [
"r114"
]
},
"ifrs-full_InterestIncomeOnDeposits": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "InterestIncomeOnDeposits",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": "ifrs-full_FinanceIncome",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Interest received on bank deposits"
}
},
"en": {
"role": {
"documentation": "The amount of interest income on deposits. [Refer: Interest income]"
}
}
},
"auth_ref": [
"r114"
]
},
"ifrs-full_InterestPaidClassifiedAsFinancingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "InterestPaidClassifiedAsFinancingActivities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInFinancingActivities",
"weight": -1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Interest paid",
"label": "Interest paid, classified as financing activities"
}
},
"en": {
"role": {
"documentation": "The cash outflow for interest paid, classified as financing activities."
}
}
},
"auth_ref": [
"r44"
]
},
"ifrs-full_InterestReceivedClassifiedAsInvestingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "InterestReceivedClassifiedAsInvestingActivities",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInInvestingActivities",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Interest received"
}
},
"en": {
"role": {
"documentation": "The cash inflow from interest received, classified as investing activities."
}
}
},
"auth_ref": [
"r44"
]
},
"ifrs-full_IssuedCapital": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "IssuedCapital",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Equity",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Share capital"
}
},
"en": {
"role": {
"documentation": "The nominal value of capital issued."
}
}
},
"auth_ref": [
"r80"
]
},
"bdrx_ItemsThatWillOrMayBeReclassifiedSubsequentlyToProfitOrLossAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ItemsThatWillOrMayBeReclassifiedSubsequentlyToProfitOrLossAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Items that will or may be reclassified subsequently to profit or loss:"
}
}
},
"auth_ref": []
},
"ifrs-full_Liabilities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "Liabilities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_EquityAndLiabilities",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total liabilities",
"label": "Liabilities [Default Label]"
}
},
"en": {
"role": {
"documentation": "The amount of a present obligation of the entity to transfer an economic resource as a result of past events. Economic resource is a right that has the potential to produce economic benefits."
}
}
},
"auth_ref": [
"r11",
"r60",
"r61",
"r62",
"r68",
"r72"
]
},
"ifrs-full_LiabilitiesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "LiabilitiesAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Liabilities"
}
}
},
"auth_ref": []
},
"dei_LocalPhoneNumber": {
"xbrltype": "normalizedStringItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "LocalPhoneNumber",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Local Phone Number",
"documentation": "Local phone number for entity."
}
}
},
"auth_ref": []
},
"ifrs-full_LongtermBorrowings": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "LongtermBorrowings",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_NoncurrentLiabilities",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Borrowings"
}
},
"en": {
"role": {
"documentation": "The non-current portion of non-current borrowings. [Refer: Borrowings]"
}
}
},
"auth_ref": [
"r115"
]
},
"bdrx_LossFromOperations": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "LossFromOperations",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/BasisOfPreparationDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Loss from operations"
}
}
},
"auth_ref": []
},
"ifrs-full_MergerReserve": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "MergerReserve",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Equity",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Merger reserve"
}
},
"en": {
"role": {
"documentation": "A component of equity that may result in relation to a business combination outside the scope of IFRS 3."
}
}
},
"auth_ref": [
"r115"
]
},
"bdrx_MergersReserveMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "MergersReserveMember",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Mergers Reserve [Member]"
}
}
},
"auth_ref": []
},
"bdrx_NegativeCashFlowsFromOperatingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "NegativeCashFlowsFromOperatingActivities",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/BasisOfPreparationDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Negative cash flows from operating activities"
}
}
},
"auth_ref": []
},
"dei_NoTradingSymbolFlag": {
"xbrltype": "trueItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "NoTradingSymbolFlag",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "No Trading Symbol Flag",
"documentation": "Boolean flag that is true only for a security having no trading symbol."
}
}
},
"auth_ref": []
},
"ifrs-full_NoncurrentAssets": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "NoncurrentAssets",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Assets",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total Non-Current Assets",
"label": "Non-current assets [Default Label]"
}
},
"en": {
"role": {
"documentation": "The amount of assets that do not meet the definition of current assets. [Refer: Current assets]"
}
}
},
"auth_ref": [
"r12",
"r53",
"r89"
]
},
"ifrs-full_NoncurrentAssetsAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "NoncurrentAssetsAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Non-current assets"
}
}
},
"auth_ref": []
},
"ifrs-full_NoncurrentLiabilities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "NoncurrentLiabilities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Liabilities",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Total Non-Current Liabilities",
"label": "Non-current liabilities [Default Label]"
}
},
"en": {
"role": {
"documentation": "The amount of liabilities that do not meet the definition of current liabilities. [Refer: Current liabilities]"
}
}
},
"auth_ref": [
"r13",
"r55",
"r89"
]
},
"ifrs-full_NoncurrentLiabilitiesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "NoncurrentLiabilitiesAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Non-current liabilities"
}
}
},
"auth_ref": []
},
"ifrs-full_NotesAndOtherExplanatoryInformationAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "NotesAndOtherExplanatoryInformationAbstract",
"lang": {
"en-us": {
"role": {
"label": "Notes and other explanatory information [abstract]"
}
}
},
"auth_ref": []
},
"ifrs-full_NumberOfSharesAuthorised": {
"xbrltype": "sharesItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "NumberOfSharesAuthorised",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Number of shares authorised, allotted and fully paid - classified as equity"
}
},
"en": {
"role": {
"documentation": "The number of shares authorised."
}
}
},
"auth_ref": [
"r16"
]
},
"bdrx_OrdinaryShareMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "OrdinaryShareMember",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Ordinary Share [Member]"
}
}
},
"auth_ref": []
},
"bdrx_OtherIncomes": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "OtherIncomes",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossFromOperatingActivities",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Other income"
}
}
},
"auth_ref": []
},
"bdrx_OtherInterest": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "OtherInterest",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails": {
"parentTag": "ifrs-full_FinanceIncome",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/FinanceIncomeAndExpenseDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Other interest"
}
}
},
"auth_ref": []
},
"dei_OtherReportingStandardItemNumber": {
"xbrltype": "otherReportingStandardItemNumberItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "OtherReportingStandardItemNumber",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Other Reporting Standard Item Number",
"documentation": "\"Item 17\" or \"Item 18\" specified when the basis of accounting is neither US GAAP nor IFRS."
}
}
},
"auth_ref": [
"r106"
]
},
"ifrs-full_ParValuePerShare": {
"xbrltype": "perShareItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ParValuePerShare",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Ordinary shares par value"
}
},
"en": {
"role": {
"documentation": "The nominal value per share."
}
}
},
"auth_ref": [
"r17"
]
},
"ifrs-full_PaymentsOfLeaseLiabilitiesClassifiedAsFinancingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "PaymentsOfLeaseLiabilitiesClassifiedAsFinancingActivities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInFinancingActivities",
"weight": -1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Amounts paid on lease liabilities",
"label": "Payments of lease liabilities, classified as financing activities"
}
},
"en": {
"role": {
"documentation": "The cash outflow for payment of lease liabilities, classified as financing activities. [Refer: Lease liabilities]"
}
}
},
"auth_ref": [
"r86"
]
},
"dei_PreCommencementIssuerTenderOffer": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "PreCommencementIssuerTenderOffer",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Pre-commencement Issuer Tender Offer",
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act."
}
}
},
"auth_ref": [
"r100"
]
},
"dei_PreCommencementTenderOffer": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "PreCommencementTenderOffer",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Pre-commencement Tender Offer",
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act."
}
}
},
"auth_ref": [
"r102"
]
},
"ifrs-full_Prepayments": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "Prepayments",
"crdr": "debit",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Prepayments*"
}
},
"en": {
"role": {
"documentation": "Receivables that represent amounts paid for goods and services before they have been delivered."
}
}
},
"auth_ref": [
"r79"
]
},
"ifrs-full_ProceedsFromIssuingShares": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ProceedsFromIssuingShares",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInFinancingActivities",
"weight": 1.0,
"order": 4.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Share issues including warrants, net of costs"
}
},
"en": {
"role": {
"documentation": "The cash inflow from issuing shares."
}
}
},
"auth_ref": [
"r85"
]
},
"ifrs-full_ProfitLoss": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ProfitLoss",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_AdjustmentsForReconcileProfitLoss",
"weight": -1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Loss for the period",
"label": "Profit (loss)"
}
},
"en": {
"role": {
"documentation": "The total of income less expenses from continuing and discontinued operations, excluding the components of other comprehensive income. [Refer: Other comprehensive income]"
}
}
},
"auth_ref": [
"r1",
"r19",
"r42",
"r49",
"r51",
"r68",
"r70",
"r89",
"r90"
]
},
"ifrs-full_ProfitLossAttributableToOrdinaryEquityHoldersOfParentEntityAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ProfitLossAttributableToOrdinaryEquityHoldersOfParentEntityAbstract",
"presentation": [
"http://biodexapharma.com/role/LossPerShareDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Numerator"
}
}
},
"auth_ref": []
},
"ifrs-full_ProfitLossAttributableToOwnersOfParent": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ProfitLossAttributableToOwnersOfParent",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ComprehensiveIncomeAttributableToOwnersOfParent",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity",
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Loss for the period attributable to the owners of the parent",
"label": "Loss for the period"
}
},
"en": {
"role": {
"documentation": "The profit (loss) from continuing and discontinued operations attributable to owners of the parent. [Refer: Profit (loss)]"
}
}
},
"auth_ref": [
"r20"
]
},
"ifrs-full_ProfitLossBeforeTax": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ProfitLossBeforeTax",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossAttributableToOwnersOfParent",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Loss before tax",
"label": "Profit (loss) before tax"
}
},
"en": {
"role": {
"documentation": "The profit (loss) before tax expense or income. [Refer: Profit (loss)]"
}
}
},
"auth_ref": [
"r65",
"r76",
"r77",
"r91",
"r92"
]
},
"ifrs-full_ProfitLossFromContinuingOperations": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ProfitLossFromContinuingOperations",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/LossPerShareDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Loss used in basic EPS and diluted EPS:"
}
},
"en": {
"role": {
"documentation": "The profit (loss) from continuing operations. [Refer: Continuing operations, unless line item indicates otherwise [member]; Profit (loss)]"
}
}
},
"auth_ref": [
"r19",
"r57",
"r68",
"r70"
]
},
"ifrs-full_ProfitLossFromOperatingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ProfitLossFromOperatingActivities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossBeforeTax",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"totalLabel": "Loss from operations",
"label": "Profit (loss) from operating activities"
}
},
"en": {
"role": {
"documentation": "The profit (loss) from operating activities of the entity. [Refer: Profit (loss)]"
}
}
},
"auth_ref": [
"r82",
"r117"
]
},
"ifrs-full_PropertyPlantAndEquipment": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "PropertyPlantAndEquipment",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_NoncurrentAssets",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Property, plant and equipment"
}
},
"en": {
"role": {
"documentation": "The amount of tangible assets that: (a) are held for use in the production or supply of goods or services, for rental to others, or for administrative purposes; and (b) are expected to be used during more than one period. Note that right-of-use assets are not included. [Contrast: Property, plant and equipment including right-of-use assets]"
}
}
},
"auth_ref": [
"r6",
"r31"
]
},
"ifrs-full_PurchaseOfIntangibleAssetsClassifiedAsInvestingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "PurchaseOfIntangibleAssetsClassifiedAsInvestingActivities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInInvestingActivities",
"weight": -1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Purchase of intangible assets",
"label": "Purchase of intangible assets, classified as investing activities"
}
},
"en": {
"role": {
"documentation": "The cash outflow for the purchases of intangible assets, classified as investing activities. [Refer: Intangible assets other than goodwill]"
}
}
},
"auth_ref": [
"r84"
]
},
"ifrs-full_PurchaseOfPropertyPlantAndEquipmentClassifiedAsInvestingActivities": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "PurchaseOfPropertyPlantAndEquipmentClassifiedAsInvestingActivities",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInInvestingActivities",
"weight": -1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Purchases of property, plant and equipment",
"label": "Purchase of property, plant and equipment, classified as investing activities"
}
},
"en": {
"role": {
"documentation": "The cash outflow for the purchases of property, plant and equipment, classified as investing activities. [Refer: Property, plant and equipment]"
}
}
},
"auth_ref": [
"r84"
]
},
"bdrx_RatioChangeDescription": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "RatioChangeDescription",
"presentation": [
"http://biodexapharma.com/role/EventsAfterReportingDateDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Ratio change description"
}
}
},
"auth_ref": []
},
"bdrx_RepaymentOfPromissoryNote": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "RepaymentOfPromissoryNote",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfCashFlows": {
"parentTag": "ifrs-full_CashFlowsFromUsedInFinancingActivities",
"weight": 1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfCashFlows"
],
"lang": {
"en-us": {
"role": {
"label": "Repayment of Promissory Note"
}
}
},
"auth_ref": []
},
"ifrs-full_ResearchAndDevelopmentExpense": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "ResearchAndDevelopmentExpense",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossFromOperatingActivities",
"weight": -1.0,
"order": 3.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"negatedLabel": "Research and development costs",
"label": "Research and development expense"
}
},
"en": {
"role": {
"documentation": "The amount of expenditure directly attributable to research or development activities, recognised in profit or loss."
}
}
},
"auth_ref": [
"r40"
]
},
"bdrx_ResearchAndDevlopmentCost": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ResearchAndDevlopmentCost",
"crdr": "debit",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrantDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Research and devlopment cost"
}
}
},
"auth_ref": []
},
"ifrs-full_RetainedEarnings": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "RetainedEarnings",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Equity",
"weight": 1.0,
"order": 6.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Accumulated deficit"
}
},
"en": {
"role": {
"documentation": "A component of equity representing the entity's cumulative undistributed earnings or deficit."
}
}
},
"auth_ref": [
"r80",
"r81"
]
},
"ifrs-full_RetainedEarningsMember": {
"xbrltype": "domainItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "RetainedEarningsMember",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Retained earnings [member]"
}
},
"en": {
"role": {
"documentation": "This member stands for a component of equity representing an entity's cumulative undistributed earnings or deficit."
}
}
},
"auth_ref": [
"r4",
"r78"
]
},
"ifrs-full_Revenue": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "Revenue",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome": {
"parentTag": "ifrs-full_ProfitLossFromOperatingActivities",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfComprehensiveIncome"
],
"lang": {
"en-us": {
"role": {
"label": "Revenue"
}
},
"en": {
"role": {
"documentation": "The income arising in the course of an entity's ordinary activities. Income is increases in assets, or decreases in liabilities, that result in increases in equity, other than those relating to contributions from holders of equity claims."
}
}
},
"auth_ref": [
"r22",
"r56",
"r65",
"r66",
"r69",
"r73",
"r74",
"r75",
"r76",
"r77",
"r89"
]
},
"bdrx_ScheduleOfAccountingForERapaAndCIPRITGrantTableTextBlock": {
"xbrltype": "textBlockItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ScheduleOfAccountingForERapaAndCIPRITGrantTableTextBlock",
"presentation": [
"http://biodexapharma.com/role/AccountingForErapaAndCpritGrantTables"
],
"lang": {
"en-us": {
"role": {
"label": "Schedule of relation the FAP"
}
}
},
"auth_ref": []
},
"dei_Security12bTitle": {
"xbrltype": "securityTitleItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "Security12bTitle",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Title of 12(b) Security",
"documentation": "Title of a 12(b) registered security."
}
}
},
"auth_ref": [
"r94"
]
},
"dei_Security12gTitle": {
"xbrltype": "securityTitleItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "Security12gTitle",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Title of 12(g) Security",
"documentation": "Title of a 12(g) registered security."
}
}
},
"auth_ref": [
"r98"
]
},
"dei_SecurityExchangeName": {
"xbrltype": "edgarExchangeCodeItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "SecurityExchangeName",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Security Exchange Name",
"documentation": "Name of the Exchange on which a security is registered."
}
}
},
"auth_ref": [
"r97"
]
},
"dei_SecurityReportingObligation": {
"xbrltype": "securityReportingObligationItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "SecurityReportingObligation",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Security Reporting Obligation",
"documentation": "15(d), indicating whether the security has a reporting obligation under that section of the Exchange Act."
}
}
},
"auth_ref": [
"r103"
]
},
"bdrx_ShareCapitalMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ShareCapitalMember",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Share Capital [Member]"
}
}
},
"auth_ref": []
},
"bdrx_ShareIssued": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ShareIssued",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Shares issued on 15 May 2024"
}
}
},
"auth_ref": []
},
"ifrs-full_SharePremium": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "SharePremium",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Equity",
"weight": 1.0,
"order": 2.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Share premium"
}
},
"en": {
"role": {
"documentation": "The amount received or receivable from the issuance of the entity's shares in excess of nominal value."
}
}
},
"auth_ref": [
"r80"
]
},
"bdrx_SharesIssued1": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "SharesIssued1",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Shares issued on February 15, 2023"
}
}
},
"auth_ref": []
},
"bdrx_SharesIssued2": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "SharesIssued2",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Shares issued on May 26, 2023"
}
}
},
"auth_ref": []
},
"bdrx_SharesIssuedUnderElocAgreement": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "SharesIssuedUnderElocAgreement",
"crdr": "credit",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Shares issued under ELOC agreement"
}
}
},
"auth_ref": []
},
"bdrx_SharesPremiumMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "SharesPremiumMember",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Shares Premium [Member]"
}
}
},
"auth_ref": []
},
"dei_SolicitingMaterial": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "SolicitingMaterial",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Soliciting Material",
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act."
}
}
},
"auth_ref": [
"r101"
]
},
"ifrs-full_StatementOfCashFlowsAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "StatementOfCashFlowsAbstract",
"lang": {
"en-us": {
"role": {
"label": "Statement of cash flows [abstract]"
}
}
},
"auth_ref": []
},
"ifrs-full_StatementOfChangesInEquityAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "StatementOfChangesInEquityAbstract",
"auth_ref": []
},
"ifrs-full_StatementOfChangesInEquityTable": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "StatementOfChangesInEquityTable",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails",
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Statement of changes in equity [table]"
}
},
"en": {
"role": {
"documentation": "Schedule disclosing information related to changes in equity."
}
}
},
"auth_ref": [
"r4"
]
},
"ifrs-full_StatementOfFinancialPositionAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "StatementOfFinancialPositionAbstract",
"lang": {
"en-us": {
"role": {
"label": "Statement of financial position [abstract]"
}
}
},
"auth_ref": []
},
"ifrs-full_TradeAndOtherCurrentPayables": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "TradeAndOtherCurrentPayables",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_CurrentLiabilities",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Trade and other payables"
}
},
"en": {
"role": {
"documentation": "The amount of current trade payables and current other payables. [Refer: Current trade payables; Other current payables]"
}
}
},
"auth_ref": [
"r9"
]
},
"ifrs-full_TradeAndOtherCurrentReceivables": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "TradeAndOtherCurrentReceivables",
"crdr": "debit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_CurrentAssets",
"weight": 1.0,
"order": 1.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Trade and other receivables"
}
},
"en": {
"role": {
"documentation": "The amount of current trade receivables and current other receivables. [Refer: Current trade receivables; Other current receivables]"
}
}
},
"auth_ref": [
"r7",
"r14"
]
},
"dei_TradingSymbol": {
"xbrltype": "tradingSymbolItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "TradingSymbol",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Trading Symbol",
"documentation": "Trading symbol of an instrument as listed on an exchange."
}
}
},
"auth_ref": []
},
"bdrx_TransactionsWithOwnersAbstract": {
"xbrltype": "stringItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "TransactionsWithOwnersAbstract",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Transactions with owners:"
}
}
},
"auth_ref": []
},
"bdrx_ValueOfShares": {
"xbrltype": "monetaryItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "ValueOfShares",
"crdr": "debit",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Total"
}
}
},
"auth_ref": []
},
"ifrs-full_WarrantReserve": {
"xbrltype": "monetaryItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "WarrantReserve",
"crdr": "credit",
"calculation": {
"http://biodexapharma.com/role/StatementsOfFinancialPosition": {
"parentTag": "ifrs-full_Equity",
"weight": 1.0,
"order": 4.0
}
},
"presentation": [
"http://biodexapharma.com/role/StatementsOfFinancialPosition"
],
"lang": {
"en-us": {
"role": {
"label": "Warrant reserve"
}
},
"en": {
"role": {
"documentation": "A component of equity resulting from issuing share purchase warrants, other than those resulting from share-based payment arrangements. [Refer: Reserve of share-based payments]"
}
}
},
"auth_ref": [
"r116"
]
},
"bdrx_WarrantsOutstanding": {
"xbrltype": "sharesItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "WarrantsOutstanding",
"presentation": [
"http://biodexapharma.com/role/ShareCapitalAndReservesDetailsNarrative"
],
"lang": {
"en-us": {
"role": {
"label": "Warrants outstanding"
}
}
},
"auth_ref": []
},
"bdrx_WarrantsReserveMember": {
"xbrltype": "domainItemType",
"nsuri": "http://biodexapharma.com/20250630",
"localname": "WarrantsReserveMember",
"presentation": [
"http://biodexapharma.com/role/StatementsOfChangesInEquity"
],
"lang": {
"en-us": {
"role": {
"label": "Warrants Reserve [Member]"
}
}
},
"auth_ref": []
},
"ifrs-full_WeightedAverageShares": {
"xbrltype": "sharesItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "WeightedAverageShares",
"presentation": [
"http://biodexapharma.com/role/LossPerShareDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Weighted average number of ordinary shares used in basic EPS"
}
},
"en": {
"role": {
"documentation": "The number of ordinary shares outstanding at the beginning of the period, adjusted by the number of ordinary shares bought back or issued during the period multiplied by a time-weighting factor."
}
}
},
"auth_ref": [
"r38"
]
},
"ifrs-full_WeightedAverageSharesAndAdjustedWeightedAverageSharesAbstract": {
"xbrltype": "stringItemType",
"nsuri": "https://xbrl.ifrs.org/taxonomy/2024-03-27/ifrs-full",
"localname": "WeightedAverageSharesAndAdjustedWeightedAverageSharesAbstract",
"presentation": [
"http://biodexapharma.com/role/LossPerShareDetails"
],
"lang": {
"en-us": {
"role": {
"label": "Denominator"
}
}
},
"auth_ref": []
},
"dei_WrittenCommunications": {
"xbrltype": "booleanItemType",
"nsuri": "http://xbrl.sec.gov/dei/2024",
"localname": "WrittenCommunications",
"presentation": [
"http://biodexapharma.com/role/Cover"
],
"lang": {
"en-us": {
"role": {
"label": "Written Communications",
"documentation": "Boolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act."
}
}
},
"auth_ref": [
"r111"
]
}
}
}
},
"std_ref": {
"r0": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "106",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_106_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r1": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "106",
"Subparagraph": "d",
"Clause": "i",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_106_d_i&doctype=Standard",
"URIDate": "2024-03-27"
},
"r2": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "106",
"Subparagraph": "d",
"Clause": "iii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_106_d_iii&doctype=Standard",
"URIDate": "2024-03-27"
},
"r3": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "106",
"Subparagraph": "d",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_106_d&doctype=Standard",
"URIDate": "2024-03-27"
},
"r4": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "106",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_106&doctype=Standard",
"URIDate": "2024-03-27"
},
"r5": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "35",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_35&doctype=Standard",
"URIDate": "2024-03-27"
},
"r6": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "54",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_54_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r7": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "54",
"Subparagraph": "h",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_54_h&doctype=Standard",
"URIDate": "2024-03-27"
},
"r8": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "54",
"Subparagraph": "i",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_54_i&doctype=Standard",
"URIDate": "2024-03-27"
},
"r9": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "54",
"Subparagraph": "k",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_54_k&doctype=Standard",
"URIDate": "2024-03-27"
},
"r10": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "54",
"Subparagraph": "n",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_54_n&doctype=Standard",
"URIDate": "2024-03-27"
},
"r11": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "55",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_55&doctype=Standard",
"URIDate": "2024-03-27"
},
"r12": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "66",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_66&doctype=Standard",
"URIDate": "2024-03-27"
},
"r13": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "69",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_69&doctype=Standard",
"URIDate": "2024-03-27"
},
"r14": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "78",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_78_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r15": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "78",
"Subparagraph": "e",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_78_e&doctype=Standard",
"URIDate": "2024-03-27"
},
"r16": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "79",
"Subparagraph": "a",
"Clause": "i",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_79_a_i&doctype=Standard",
"URIDate": "2024-03-27"
},
"r17": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "79",
"Subparagraph": "a",
"Clause": "iii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_79_a_iii&doctype=Standard",
"URIDate": "2024-03-27"
},
"r18": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "79",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_79_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r19": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "81A",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_81A_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r20": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "81B",
"Subparagraph": "a",
"Clause": "ii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_81B_a_ii&doctype=Standard",
"URIDate": "2024-03-27"
},
"r21": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "81B",
"Subparagraph": "b",
"Clause": "ii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_81B_b_ii&doctype=Standard",
"URIDate": "2024-03-27"
},
"r22": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "82",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_82_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r23": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "82",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_82_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r24": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "82",
"Subparagraph": "d",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_82_d&doctype=Standard",
"URIDate": "2024-03-27"
},
"r25": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "91",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_91_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r26": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "99",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_99&doctype=Standard",
"URIDate": "2024-03-27"
},
"r27": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "10",
"IssueDate": "2024-01-01",
"Section": "Disclosure",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=10&code=ifrs-tx-2024-en-r&doctype=Standard&dita_xref=IAS10_g17-22_TI",
"URIDate": "2024-03-27"
},
"r28": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "79",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=12&code=ifrs-tx-2024-en-r&anchor=para_79&doctype=Standard",
"URIDate": "2024-03-27"
},
"r29": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "81",
"Subparagraph": "c",
"Clause": "i",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=12&code=ifrs-tx-2024-en-r&anchor=para_81_c_i&doctype=Standard",
"URIDate": "2024-03-27"
},
"r30": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "81",
"Subparagraph": "c",
"Clause": "ii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=12&code=ifrs-tx-2024-en-r&anchor=para_81_c_ii&doctype=Standard",
"URIDate": "2024-03-27"
},
"r31": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "16",
"IssueDate": "2024-01-01",
"Paragraph": "73",
"Subparagraph": "e",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=16&code=ifrs-tx-2024-en-r&anchor=para_73_e&doctype=Standard",
"URIDate": "2024-03-27"
},
"r32": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "21",
"IssueDate": "2024-01-01",
"Paragraph": "52",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=21&code=ifrs-tx-2024-en-r&anchor=para_52_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r33": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "24",
"IssueDate": "2024-01-01",
"Section": "Disclosures",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=24&code=ifrs-tx-2024-en-r&doctype=Standard&dita_xref=IAS24_g13-24_TI",
"URIDate": "2024-03-27"
},
"r34": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "26",
"IssueDate": "2024-01-01",
"Paragraph": "35",
"Subparagraph": "b",
"Clause": "vi",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=26&code=ifrs-tx-2024-en-r&anchor=para_35_b_vi&doctype=Standard",
"URIDate": "2024-03-27"
},
"r35": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "26",
"IssueDate": "2024-01-01",
"Paragraph": "35",
"Subparagraph": "b",
"Clause": "viii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=26&code=ifrs-tx-2024-en-r&anchor=para_35_b_viii&doctype=Standard",
"URIDate": "2024-03-27"
},
"r36": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "33",
"IssueDate": "2024-01-01",
"Paragraph": "66",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2024-en-r&anchor=para_66&doctype=Standard",
"URIDate": "2024-03-27"
},
"r37": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "33",
"IssueDate": "2024-01-01",
"Paragraph": "67",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2024-en-r&anchor=para_67&doctype=Standard",
"URIDate": "2024-03-27"
},
"r38": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "33",
"IssueDate": "2024-01-01",
"Paragraph": "70",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2024-en-r&anchor=para_70_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r39": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "37",
"IssueDate": "2024-01-01",
"Paragraph": "86",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=37&code=ifrs-tx-2024-en-r&anchor=para_86&doctype=Standard",
"URIDate": "2024-03-27"
},
"r40": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "38",
"IssueDate": "2024-01-01",
"Paragraph": "126",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=38&code=ifrs-tx-2024-en-r&anchor=para_126&doctype=Standard",
"URIDate": "2024-03-27"
},
"r41": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "10",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_10&doctype=Standard",
"URIDate": "2024-03-27"
},
"r42": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "18",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_18_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r43": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "20",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_20&doctype=Standard",
"URIDate": "2024-03-27"
},
"r44": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "31",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_31&doctype=Standard",
"URIDate": "2024-03-27"
},
"r45": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "35",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_35&doctype=Standard",
"URIDate": "2024-03-27"
},
"r46": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "45",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_45&doctype=Standard",
"URIDate": "2024-03-27"
},
"r47": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "50",
"Subparagraph": "d",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_50_d&doctype=Standard",
"URIDate": "2024-03-27"
},
"r48": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "24",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2024-en-r&anchor=para_24_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r49": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "24",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2024-en-r&anchor=para_24_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r50": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "32",
"Subparagraph": "a",
"Clause": "i",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2024-en-r&anchor=para_32_a_i&doctype=Standard",
"URIDate": "2024-03-27"
},
"r51": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "32",
"Subparagraph": "a",
"Clause": "ii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2024-en-r&anchor=para_32_a_ii&doctype=Standard",
"URIDate": "2024-03-27"
},
"r52": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B12",
"Subparagraph": "b",
"Clause": "i",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B12_b_i&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r53": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B12",
"Subparagraph": "b",
"Clause": "ii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B12_b_ii&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r54": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B12",
"Subparagraph": "b",
"Clause": "iii",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B12_b_iii&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r55": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B12",
"Subparagraph": "b",
"Clause": "iv",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B12_b_iv&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r56": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B12",
"Subparagraph": "b",
"Clause": "v",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B12_b_v&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r57": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B12",
"Subparagraph": "b",
"Clause": "vi",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B12_b_vi&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r58": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B13",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B13_a&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r59": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B13",
"Subparagraph": "g",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B13_g&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r60": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "13",
"IssueDate": "2024-01-01",
"Paragraph": "93",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2024-en-r&anchor=para_93_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r61": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "13",
"IssueDate": "2024-01-01",
"Paragraph": "93",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2024-en-r&anchor=para_93_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r62": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "13",
"IssueDate": "2024-01-01",
"Paragraph": "93",
"Subparagraph": "e",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2024-en-r&anchor=para_93_e&doctype=Standard",
"URIDate": "2024-03-27"
},
"r63": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "16",
"IssueDate": "2024-01-01",
"Paragraph": "53",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=16&code=ifrs-tx-2024-en-r&anchor=para_53_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r64": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "16",
"IssueDate": "2024-01-01",
"Paragraph": "53",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=16&code=ifrs-tx-2024-en-r&anchor=para_53_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r65": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "5",
"IssueDate": "2024-01-01",
"Paragraph": "33",
"Subparagraph": "b",
"Clause": "i",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=5&code=ifrs-tx-2024-en-r&anchor=para_33_b_i&doctype=Standard",
"URIDate": "2024-03-27"
},
"r66": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "23",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_23_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r67": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "23",
"Subparagraph": "h",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_23_h&doctype=Standard",
"URIDate": "2024-03-27"
},
"r68": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "23",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_23&doctype=Standard",
"URIDate": "2024-03-27"
},
"r69": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "28",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_28_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r70": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "28",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_28_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r71": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "28",
"Subparagraph": "c",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_28_c&doctype=Standard",
"URIDate": "2024-03-27"
},
"r72": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "28",
"Subparagraph": "d",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_28_d&doctype=Standard",
"URIDate": "2024-03-27"
},
"r73": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "32",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_32&doctype=Standard",
"URIDate": "2024-03-27"
},
"r74": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "33",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_33_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r75": {
"role": "http://www.xbrl.org/2003/role/disclosureRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "34",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_34&doctype=Standard",
"URIDate": "2024-03-27"
},
"r76": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "102",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_102&doctype=Standard",
"URIDate": "2024-03-27"
},
"r77": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "103",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_103&doctype=Standard",
"URIDate": "2024-03-27"
},
"r78": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "108",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_108&doctype=Standard",
"URIDate": "2024-03-27"
},
"r79": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "78",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_78_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r80": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "78",
"Subparagraph": "e",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_78_e&doctype=Standard",
"URIDate": "2024-03-27"
},
"r81": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "IG6",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_IG6&doctype=Implementation%20Guidance",
"URIDate": "2024-03-27"
},
"r82": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "32",
"IssueDate": "2024-01-01",
"Paragraph": "IE33",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=32&code=ifrs-tx-2024-en-r&anchor=para_IE33&doctype=Illustrative%20Examples",
"URIDate": "2024-03-27"
},
"r83": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "14",
"Subparagraph": "f",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_14_f&doctype=Standard",
"URIDate": "2024-03-27"
},
"r84": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "16",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_16_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r85": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "17",
"Subparagraph": "a",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_17_a&doctype=Standard",
"URIDate": "2024-03-27"
},
"r86": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "17",
"Subparagraph": "e",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_17_e&doctype=Standard",
"URIDate": "2024-03-27"
},
"r87": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "20",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_20&doctype=Standard",
"URIDate": "2024-03-27"
},
"r88": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Section": "A Statement of cash flows for an entity other than a financial institution",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&doctype=Illustrative%20Examples&dita_xref=IAS07_IE_A_TI",
"URIDate": "2024-03-27"
},
"r89": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IFRS",
"Number": "12",
"IssueDate": "2024-01-01",
"Paragraph": "B10",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2024-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B",
"URIDate": "2024-03-27"
},
"r90": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IFRS",
"Number": "17",
"IssueDate": "2024-01-01",
"Paragraph": "113",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=17&code=ifrs-tx-2024-en-r&anchor=para_113_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r91": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "23",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_23&doctype=Standard",
"URIDate": "2024-03-27"
},
"r92": {
"role": "http://www.xbrl.org/2003/role/exampleRef",
"Name": "IFRS",
"Number": "8",
"IssueDate": "2024-01-01",
"Paragraph": "28",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2024-en-r&anchor=para_28_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r93": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "12"
},
"r94": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "12",
"Subsection": "b"
},
"r95": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "12",
"Subsection": "b-2"
},
"r96": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "12",
"Subsection": "b-23"
},
"r97": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "12",
"Subsection": "d1-1"
},
"r98": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "12",
"Subsection": "g"
},
"r99": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "12, 13, 15d"
},
"r100": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "13e",
"Subsection": "4c"
},
"r101": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "14a",
"Subsection": "12"
},
"r102": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "14d",
"Subsection": "2b"
},
"r103": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Exchange Act",
"Number": "240",
"Section": "15",
"Subsection": "d"
},
"r104": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Form 10-K",
"Number": "249",
"Section": "310"
},
"r105": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Form 10-Q",
"Number": "240",
"Section": "308",
"Subsection": "a"
},
"r106": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Form 20-F",
"Number": "249",
"Section": "220",
"Subsection": "f"
},
"r107": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Form 40-F",
"Number": "249",
"Section": "240",
"Subsection": "f"
},
"r108": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Forms 10-K, 10-Q, 20-F",
"Number": "240",
"Section": "13",
"Subsection": "a-1"
},
"r109": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Regulation S-T",
"Number": "232",
"Section": "405"
},
"r110": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Securities Act",
"Number": "230",
"Section": "405"
},
"r111": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Securities Act",
"Number": "230",
"Section": "425"
},
"r112": {
"role": "http://www.xbrl.org/2003/role/presentationRef",
"Publisher": "SEC",
"Name": "Securities Act",
"Number": "7A",
"Section": "B",
"Subsection": "2"
},
"r113": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "10",
"Subparagraph": "e",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_10_e&doctype=Standard",
"URIDate": "2024-03-27"
},
"r114": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "112",
"Subparagraph": "c",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_112_c&doctype=Standard",
"URIDate": "2024-03-27"
},
"r115": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "55",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_55&doctype=Standard",
"URIDate": "2024-03-27"
},
"r116": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "78",
"Subparagraph": "e",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_78_e&doctype=Standard",
"URIDate": "2024-03-27"
},
"r117": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "1",
"IssueDate": "2024-01-01",
"Paragraph": "85",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2024-en-r&anchor=para_85&doctype=Standard",
"URIDate": "2024-03-27"
},
"r118": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "20",
"Subparagraph": "b",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_20_b&doctype=Standard",
"URIDate": "2024-03-27"
},
"r119": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "20",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_20&doctype=Standard",
"URIDate": "2024-03-27"
},
"r120": {
"role": "http://www.xbrl.org/2009/role/commonPracticeRef",
"Name": "IAS",
"Number": "7",
"IssueDate": "2024-01-01",
"Paragraph": "45",
"URI": "https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2024-en-r&anchor=para_45&doctype=Standard",
"URIDate": "2024-03-27"
}
}
}